From Trifluoroacetic acid to triflic derivatives : RHODIA s Fluorination tree. François METZ. Rhodia Research Centre Lyon - France
|
|
|
- Piers Hubbard
- 5 years ago
- Views:
Transcription
1 From Trifluoroacetic acid to triflic derivatives : RHDIA s Fluorination tree François METZ Rhodia Research Centre Lyon - France CHALLENGING BUNDARIES
2 Fluorination : a key technology for Rhodia For the past 20 years, fluorinated intermediates have known a great success in Life Science actives, as the introduction of one or several fluorine atoms can increase the active s biological activity. As a result, fluorination is a technology in demand with 20% of Life Science actives containing one or several fluorine atoms (C&EN, 84(23), 2006, pp15-23) Rhodia is recognised as a worldwide leader, with an extensive industrial experience and innovative chemistry in several fluorination technologies, to produce both fluoroaliphatic and fluoroaromatic compounds. 2 CHALLENGING BUNDARIES
3 Rhodia s Fluorination Technologies F (X) R R CF 2 X R = H,, F HALEX reaction HF liquid technology HF gas technology Direct trifluoromethylation Facilities Laboratory Pilot Industrial 3 CHALLENGING BUNDARIES
4 Fluorination sites location Industrial scale fluorination reactions are performed on dedicated and fully-integrated sites, designed for multi-products manufacturing. Research & Development teams are based in our central Research Center in Lyon. Avonmouth Lyon Salindres Main Research Center Production sites 4 CHALLENGING BUNDARIES
5 Fluoro-aliphatic intermediates tree A solid and stable trunk : F 3 C C 2 H TFA allowing a vigorous tree to grow 5 CHALLENGING BUNDARIES
6 TFA : a three stages process Photo-oxidation : C 2 = C 2 + 1/2 h 2 C 3 C PER TCAC Unique CFC-free process Fluorination : C 3 - C + 4HF - CF + 4H TFAF Hydrolysis : - CF + H 2 - CH + HF TFA 6 CHALLENGING BUNDARIES
7 TFA : a three stages process process Photo-oxidation : Reaction mechanism (supposed) h. +. Initiation 2 by-products propagation. by-products To fluorination CTCA 7 CHALLENGING BUNDARIES
8 TFA : a three stages process Fluorination : successive -F exchanges HF HF C 3 C C 3 CF C 2 FCF -H -H (TCAC) (TCAF) (DCFAF) HF HF C 2 FCF CF 2 CF CF -H -H (CDFAF) (TFAF) 8 CHALLENGING BUNDARIES
9 TFA : a three stages process Hydrolysis : access to both TFA & CDFA + F CF 2 F H 2 H 2 H CF 2 H trifluoroacetic acid (TFA) chlorodifluoroacetic acid (CDFA) TFA and CDFA are key starting raw materials, which open the way to a extensive range of aliphatic fluorinated compounds. 9 CHALLENGING BUNDARIES
10 Rhodia s advantages : Large capacity Unique and competitive process Environmentally friendly process : CFC free Experience in handling hazardous reagents Rhodia s integration in other technologies 10 TFA : specificities Cost competitive process Sustainable technology CHALLENGING BUNDARIES
11 TFA : applications Some examples : Protection and deprotection reactions (of alcohols, amides, nucleosides, peptides, phenols, sugars, cleavage of dithioacetals.) Catalyst and solvent (Ring substitution, aromatic nitration, rearrangements, isomerization ) Heterocyclization reactions Reaction with halogenated compounds 11 CHALLENGING BUNDARIES
12 TFA chain F 3 C H F 3 C H TFE TFA F 3 C TFAA Anesthetics Celecoxib (Pfizer) F 3 C H Fluoral Efavirenz (BMS) F 3 C Et ETFA S 3 H triflic acid chain Dithiopyr (Dow) F 3 C Et ETFAA Starting from these building blocks and thanks to an extensive portfolio of technologies, Rhodia offers the widest range of TFA derivatives and advanced downstream intermediates. 12 CHALLENGING BUNDARIES
13 CDFA chain F 2 C F H 2 -HF CF 2 H MeH CF - H 2 2 CDFAF CDFA MeCDFA F F MeCDFA Activated Zn CH H F F H 2 N N N F F H H Eli Lilly Gemcitabine (anticancer) 13 CHALLENGING BUNDARIES
14 Triflic acid (TA) : a unique chemical pathway F 3 C H S 3 H EP S 2 CK [ K] S 2 K -C 2 Direct nucleophilic trifluoromethylation : a new process using CFC free innovative technology cation effect : K>Na>Li Influence of temperature and choice of solvent are key Process optimisation allows for reliable and competitive manufacturing conditions. 14 CHALLENGING BUNDARIES
15 Anionic trifluoromethylation : scope &limitations S C 6 H 5 C 6 H 5 S + C 2 K C 6 H 5 S 80% H C 6 H 5 CH + C 2 K C 6 H % EP C 6 H 5 SCN + C 2 K C 6 H 5 S 40% 15 CHALLENGING BUNDARIES
16 Anionic trifluoromethylation : scope & limitations H C 6 H 5 + C 2 K 2 C 6 H 5 + C 6 H 5 F 3 C 10% 20% H + C 2 K C 6 H 5 C 6 H 5 F 3 C 25% EP C 2 K + crotonisation H 10% 16 CHALLENGING BUNDARIES
17 Acilys TA Triflic Acid CAS # MW 150 g.mol -1 BP 162 C Density 1.7 kg.l -1 Inventories All F F C S H F Trifluoromethanesulfonic acid, also known as triflic acid, is the most acidic of all the commercial chemicals available at the industrial scale. Its unique chemical structure gives triflic acid an outstanding acid strength as well as chemical stability. It is extremely resistant towards thermal degradation, oxidation and reduction, and is also non-oxidizing. Triflic acid is widely used for specialty and fine chemical productions, for acid catalyzed reactions such as : Cationic polymerizations Aromatic alkylations Esterifications Acylations 17 CHALLENGING BUNDARIES
18 Examples of reactions using Acilys-TA H H F F S H F + Me Me F F S H F + Rhodia commercializes triflic acid under the tradename Acilys-TA. It is readily available from few kilograms to multitons scale. 18 Moreover, a large range of triflic acid derivatives such as triflic anhydride, organic and silyl esters as well as metal triflates, Rhodia. are also available from CHALLENGING BUNDARIES
19 Triflic acid chain : An extensive range of products for a wide range of applications Pharma S 3 CH 2 F 3 C Pharma Ac N. Roques FR (2000) C 2 H C 2 K S 2 K S 2 (Br) Ionic liquids ( S 2 ) 2 NH Battery salt ( S 2 ) 2 NLi S 3 SiMe 3 S 3 SiMe 2 t-bu Pharma Electronics H 2 2 Pharma buiding blocks Catalysis S 3 R (Me, Et, ) Pharma ( S 2 ) 2 Catalysis [( S 2 ) 2 N] 3 M Catalysis ( S 3 ) 3 M S 3 H In blue : development products M : rare earths e.g. Yb, La 19 CHALLENGING BUNDARIES
20 New technology: radical trifluoromethylation New technology: radical trifluoromethylation A new range of building blocks Ac F 3 C Ac F 3 C * F 3 C H * S 2 X F 3 C Ac Ac F 3 C Ac F 3 C H Ac F 3 C CH 3 Lab scale Kilo Lab 20 CHALLENGING BUNDARIES
21 Rhodia rganics : a key player in fluorination Rhodia associates all these fluoroaliphatic building blocks with an extensive portfolio of technologies. We are your innovative partner in developing more advanced downstream intermediates. Building blocks + our technologies portfolio Advanced fluorinated intermediates With you, let Rhodia s fluoroaliphatics tree grow and blossom! For more information, come and see us on Booth C11 21 CHALLENGING BUNDARIES
22 Acknowledgments This work is the result of a constant commitment of many teams : Synthesis chemistry, Analysis, Process development, Manufacturing, Marketing & Sales, Health & Safety, Regulatory THANKS T EVERYBDY!!! 22 CHALLENGING BUNDARIES
ORGANICS. Organic Fluorinated Compounds: Raw materials, Intermediates & Active ingredients
 RGANICS rganic Fluorinated Compounds: Raw materials, Intermediates & Active ingredients MARKETS & APPLICATINS rganic fluorinated compounds: raw materials, intermediates & active ingredients The use of
RGANICS rganic Fluorinated Compounds: Raw materials, Intermediates & Active ingredients MARKETS & APPLICATINS rganic fluorinated compounds: raw materials, intermediates & active ingredients The use of
ORGANIC - BROWN 8E CH. 22- REACTIONS OF BENZENE AND ITS DERIVATIVES
 !! www.clutchprep.com CONCEPT: ELECTROPHILIC AROMATIC SUBSTITUTION GENERAL MECHANISM Benzene reacts with very few reagents. It DOES NOT undergo typical addition reactions. Why? If we can get benzene to
!! www.clutchprep.com CONCEPT: ELECTROPHILIC AROMATIC SUBSTITUTION GENERAL MECHANISM Benzene reacts with very few reagents. It DOES NOT undergo typical addition reactions. Why? If we can get benzene to
TOK: The relationship between a reaction mechanism and the experimental evidence to support it could be discussed. See
 Option G: Further organic chemistry (15/22 hours) SL students study the core of these options and HL students study the whole option (the core and the extension material). TOK: The relationship between
Option G: Further organic chemistry (15/22 hours) SL students study the core of these options and HL students study the whole option (the core and the extension material). TOK: The relationship between
Organic Chemistry 112 A B C - Syllabus Addendum for Prospective Teachers
 Chapter Organic Chemistry 112 A B C - Syllabus Addendum for Prospective Teachers Ch 1-Structure and bonding Ch 2-Polar covalent bonds: Acids and bases McMurry, J. (2004) Organic Chemistry 6 th Edition
Chapter Organic Chemistry 112 A B C - Syllabus Addendum for Prospective Teachers Ch 1-Structure and bonding Ch 2-Polar covalent bonds: Acids and bases McMurry, J. (2004) Organic Chemistry 6 th Edition
Chapter 10: Carboxylic Acids and Their Derivatives
 Chapter 10: Carboxylic Acids and Their Derivatives The back of the white willow tree (Salix alba) is a source of salicylic acid which is used to make aspirin (acetylsalicylic acid) The functional group
Chapter 10: Carboxylic Acids and Their Derivatives The back of the white willow tree (Salix alba) is a source of salicylic acid which is used to make aspirin (acetylsalicylic acid) The functional group
Class XII: Chemistry Chapter 13: Amines Top concepts
 Class XII: Chemistry Chapter 13: Amines Top concepts 1. Amines are regarded as derivatives of ammonia in which one, two or all three hydrogen atoms are replaced by alkyl or aryl group 2. Classification
Class XII: Chemistry Chapter 13: Amines Top concepts 1. Amines are regarded as derivatives of ammonia in which one, two or all three hydrogen atoms are replaced by alkyl or aryl group 2. Classification
Detailed Course Content
 Detailed Course Content Chapter 1: Carbon Compounds and Chemical Bonds The Structural Theory of Organic Chemistry 4 Chemical Bonds: The Octet Rule 6 Lewis Structures 8 Formal Charge 11 Resonance 14 Quantum
Detailed Course Content Chapter 1: Carbon Compounds and Chemical Bonds The Structural Theory of Organic Chemistry 4 Chemical Bonds: The Octet Rule 6 Lewis Structures 8 Formal Charge 11 Resonance 14 Quantum
Advanced Analytical Chemistry Lecture 19. Chem 4631
 Advanced Analytical Chemistry Lecture 19 Chem 4631 Organic Electrochemistry is a multidisciplinary science overlapping the fields of organic chemistry, biochemistry, physical chemistry and electrochemistry.
Advanced Analytical Chemistry Lecture 19 Chem 4631 Organic Electrochemistry is a multidisciplinary science overlapping the fields of organic chemistry, biochemistry, physical chemistry and electrochemistry.
Synthesis of Nitriles a. dehydration of 1 amides using POCl 3 : b. SN2 reaction of cyanide ion on halides:
 I. Nitriles Nitriles consist of the CN functional group, and are linear with sp hybridization on C and N. Nitriles are non-basic at nitrogen, since the lone pair exists in an sp orbital (50% s character
I. Nitriles Nitriles consist of the CN functional group, and are linear with sp hybridization on C and N. Nitriles are non-basic at nitrogen, since the lone pair exists in an sp orbital (50% s character
Alkyl phenyl ketones are usually named by adding the acyl group as prefix to phenone.
 Aldehydes, Ketones and Carboxylic Acids Nomenclature of aldehydes and ketones Aldehydes: Often called by their common names instead of IUPAC names. Ketones: Derived by naming two alkyl or aryl groups bonded
Aldehydes, Ketones and Carboxylic Acids Nomenclature of aldehydes and ketones Aldehydes: Often called by their common names instead of IUPAC names. Ketones: Derived by naming two alkyl or aryl groups bonded
GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR (preparation of carboxylic acid esters by telomerisation C07C 67/47; telomerisation C08F)
 CPC - C07B - 2017.08 C07B GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR (preparation of carboxylic acid esters by telomerisation C07C 67/47; telomerisation C08F) General methods for the preparation
CPC - C07B - 2017.08 C07B GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR (preparation of carboxylic acid esters by telomerisation C07C 67/47; telomerisation C08F) General methods for the preparation
Module9. Nuclear Magnetic Resonance Spectroscopy Nuclear Magnetic Resonance (NMR) spectroscopy - Chemical shift - Integration of signal area
 1 CHEMISTRY 263 HOME WORK Lecture Topics: Module7. Hydrogenation of Alkenes The Function of the Catalyst - Syn and anti- addition Hydrogenation of Alkynes - Syn- addition of hydrogen: Synthesis of cis-alkenes
1 CHEMISTRY 263 HOME WORK Lecture Topics: Module7. Hydrogenation of Alkenes The Function of the Catalyst - Syn and anti- addition Hydrogenation of Alkynes - Syn- addition of hydrogen: Synthesis of cis-alkenes
Option G: Further organic chemistry (15/22 hours)
 Option G: Further organic chemistry (15/) TOK: The relationship between a reaction mechanism and the experimental evidence to support it could be discussed. See 16... Core material: G1 G8 are core material
Option G: Further organic chemistry (15/) TOK: The relationship between a reaction mechanism and the experimental evidence to support it could be discussed. See 16... Core material: G1 G8 are core material
Chapter 20: Carboxylic Acids
 1 Chapter 20: Carboxylic Acids I. Introduction: Carboxylic acid structure: Classification of carboxylic acids: A carboxylic acid donates protons by the heterocyclic cleavage of the O-H bond, generating
1 Chapter 20: Carboxylic Acids I. Introduction: Carboxylic acid structure: Classification of carboxylic acids: A carboxylic acid donates protons by the heterocyclic cleavage of the O-H bond, generating
CHEMISTRY 263 HOME WORK
 Lecture Topics: CHEMISTRY 263 HOME WORK Module7: Hydrogenation of Alkenes Hydrogenation - syn and anti- addition - hydrogenation of alkynes - synthesis of cis-alkenes -synthesis of trans-alkenes Text sections:
Lecture Topics: CHEMISTRY 263 HOME WORK Module7: Hydrogenation of Alkenes Hydrogenation - syn and anti- addition - hydrogenation of alkynes - synthesis of cis-alkenes -synthesis of trans-alkenes Text sections:
OCR (A) Chemistry A-level. Module 6: Organic Chemistry and Analysis
 OCR (A) Chemistry A-level Module 6: Organic Chemistry and Analysis Organic Synthesis Notes by Adam Robertson DEFINITIONS Heterolytic fission: The breaking of a covalent bond when one of the bonded atoms
OCR (A) Chemistry A-level Module 6: Organic Chemistry and Analysis Organic Synthesis Notes by Adam Robertson DEFINITIONS Heterolytic fission: The breaking of a covalent bond when one of the bonded atoms
KOT 222 Organic Chemistry II
 KOT 222 Organic Chemistry II Course Objectives: 1) To introduce the chemistry of alcohols and ethers. 2) To study the chemistry of functional groups. 3) To learn the chemistry of aromatic compounds and
KOT 222 Organic Chemistry II Course Objectives: 1) To introduce the chemistry of alcohols and ethers. 2) To study the chemistry of functional groups. 3) To learn the chemistry of aromatic compounds and
FIVE MEMBERED AROMATIC HETEROCYCLES
 FIVE MEMBERED AROMATIC HETEROCYCLES 63 Electrophilic aromatic substitution reaction of five membered aromatic heterocycles X = O, S or NH a-substitution b-substitution The Substitution is regioselective
FIVE MEMBERED AROMATIC HETEROCYCLES 63 Electrophilic aromatic substitution reaction of five membered aromatic heterocycles X = O, S or NH a-substitution b-substitution The Substitution is regioselective
 Top concepts Chapter: Amines 1. Amines are regarded as derivatives of ammonia in which one, two or all three hydrogen atoms are replaced by alkyl or aryl group 2. Classification of amines: 3. Preparation
Top concepts Chapter: Amines 1. Amines are regarded as derivatives of ammonia in which one, two or all three hydrogen atoms are replaced by alkyl or aryl group 2. Classification of amines: 3. Preparation
CARBOXYLIC ACIDS and their Derivatives Nucleophilic Acyl substitution - Review the nomenclature for these compounds in your textbook
 CARBXYLIC ACIDS and their Derivatives Nucleophilic Acyl substitution - Review the nomenclature for these compounds in your textbook R Z R Z R Z - the basicity of Z determines the relative stability of
CARBXYLIC ACIDS and their Derivatives Nucleophilic Acyl substitution - Review the nomenclature for these compounds in your textbook R Z R Z R Z - the basicity of Z determines the relative stability of
REACTION AND SYNTHESIS REVIEW
 REACTION AND SYNTHESIS REVIEW A STUDENT SHOULD BE ABLE TO PREDICT PRODUCTS, IDENTIFY REACTANTS, GIVE REACTION CONDITIONS, PROPOSE SYNTHESES, AND PROPOSE MECHANISMS (AS LISTED BELOW). REVIEW THE MECHANISM
REACTION AND SYNTHESIS REVIEW A STUDENT SHOULD BE ABLE TO PREDICT PRODUCTS, IDENTIFY REACTANTS, GIVE REACTION CONDITIONS, PROPOSE SYNTHESES, AND PROPOSE MECHANISMS (AS LISTED BELOW). REVIEW THE MECHANISM
JEFFERSON COLLEGE COURSE SYLLABUS CHM201 ORGANIC CHEMISTRY II. 5 Credit Hours. Prepared by: Richard A. Pierce
 JEFFERSON COLLEGE COURSE SYLLABUS CHM201 ORGANIC CHEMISTRY II 5 Credit Hours Prepared by: Richard A. Pierce Revised Date: January 2008 by Ryan H. Groeneman Arts & Science Education Dr. Mindy Selsor, Dean
JEFFERSON COLLEGE COURSE SYLLABUS CHM201 ORGANIC CHEMISTRY II 5 Credit Hours Prepared by: Richard A. Pierce Revised Date: January 2008 by Ryan H. Groeneman Arts & Science Education Dr. Mindy Selsor, Dean
A. Loupy, B.Tchoubar. Salt Effects in Organic and Organometallic Chemistry
 A. Loupy, B.Tchoubar Salt Effects in Organic and Organometallic Chemistry 1 Introduction - Classification of Specific Salt Effects 1 1.1 Specific Salt Effects Involving the Salt's Lewis Acid or Base Character
A. Loupy, B.Tchoubar Salt Effects in Organic and Organometallic Chemistry 1 Introduction - Classification of Specific Salt Effects 1 1.1 Specific Salt Effects Involving the Salt's Lewis Acid or Base Character
CHEM 303 Organic Chemistry II Problem Set III Chapter 14 Answers
 CHEM 303 rganic Chemistry II Problem Set III Chapter 14 Answers 1) Give the major products of each of the following reactions. If a mixture is expected, identify the major product. + H 3 CHC CHCH 3 H 2
CHEM 303 rganic Chemistry II Problem Set III Chapter 14 Answers 1) Give the major products of each of the following reactions. If a mixture is expected, identify the major product. + H 3 CHC CHCH 3 H 2
12/27/2010. Chapter 15 Reactions of Aromatic Compounds
 Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen atom
Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen atom
CHM 292 Final Exam Answer Key
 CHM 292 Final Exam Answer Key 1. Predict the product(s) of the following reactions (5 points each; 35 points total). May 7, 2013 Acid catalyzed elimination to form the most highly substituted alkene possible
CHM 292 Final Exam Answer Key 1. Predict the product(s) of the following reactions (5 points each; 35 points total). May 7, 2013 Acid catalyzed elimination to form the most highly substituted alkene possible
UNIT 4 REVISION CHECKLIST CHEM 4 AS Chemistry
 UNIT 4 REVISION CHECKLIST CHEM 4 AS Chemistry Topic 4.1 Kinetics a) Define the terms: rate of a reaction, rate constant, order of reaction and overall order of reaction b) Deduce the orders of reaction
UNIT 4 REVISION CHECKLIST CHEM 4 AS Chemistry Topic 4.1 Kinetics a) Define the terms: rate of a reaction, rate constant, order of reaction and overall order of reaction b) Deduce the orders of reaction
ORGANIC - BROWN 8E CH CARBOXYLIC ACIDS.
 RGANIC - BRWN 8E CH. 17 - CARBXYLIC ACIDS!! www.clutchprep.com RGANIC - BRWN 8E CH. 17 - CARBXYLIC ACIDS CNCEPT: CARBXYLIC ACID NMENCLATURE IUPAC: Replace alkane -e with Substituents are located using
RGANIC - BRWN 8E CH. 17 - CARBXYLIC ACIDS!! www.clutchprep.com RGANIC - BRWN 8E CH. 17 - CARBXYLIC ACIDS CNCEPT: CARBXYLIC ACID NMENCLATURE IUPAC: Replace alkane -e with Substituents are located using
Aldehydes & Ketones LEVEL I. Phenol (enol form) Phenol is aromatic, so equiulibrium is shifted to the right hand side. b) O
 Subjective Problems Aldehydes & Ketones LEVEL I 1. a) 2,4cyclohexadiene-1-one (keto form) Phenol (enol form) Phenol is aromatic, so equiulibrium is shifted to the right hand side. b) Base This ketone is
Subjective Problems Aldehydes & Ketones LEVEL I 1. a) 2,4cyclohexadiene-1-one (keto form) Phenol (enol form) Phenol is aromatic, so equiulibrium is shifted to the right hand side. b) Base This ketone is
11/26/ Polycyclic Aromatic Compounds. Polycyclic Aromatic Compounds. Polycyclic Aromatic Compounds
 9.5 Polycyclic Aromatic Compounds The general concept of aromaticity can be extended to include polycyclic aromatic compounds Benzo[a]pyrene is one of the cancer-causing substances found in tobacco smoke
9.5 Polycyclic Aromatic Compounds The general concept of aromaticity can be extended to include polycyclic aromatic compounds Benzo[a]pyrene is one of the cancer-causing substances found in tobacco smoke
3.2.8 Haloalkanes. Nucleophilic Substitution. 267 minutes. 264 marks. Page 1 of 36
 3.2.8 Haloalkanes Nucleophilic Substitution 267 minutes 264 marks Page 1 of 36 Q1. (a) The equation below shows the reaction of 2-bromopropane with an excess of ammonia. CH 3 CHBrCH 3 + 2NH 3 CH 3 CH(NH
3.2.8 Haloalkanes Nucleophilic Substitution 267 minutes 264 marks Page 1 of 36 Q1. (a) The equation below shows the reaction of 2-bromopropane with an excess of ammonia. CH 3 CHBrCH 3 + 2NH 3 CH 3 CH(NH
Lecture Topics: I. Electrophilic Aromatic Substitution (EAS)
 Reactions of Aromatic Compounds Reading: Wade chapter 17, sections 17-1- 17-15 Study Problems: 17-44, 17-46, 17-47, 17-48, 17-51, 17-52, 17-53, 17-59, 17-61 Key Concepts and Skills: Predict and propose
Reactions of Aromatic Compounds Reading: Wade chapter 17, sections 17-1- 17-15 Study Problems: 17-44, 17-46, 17-47, 17-48, 17-51, 17-52, 17-53, 17-59, 17-61 Key Concepts and Skills: Predict and propose
Unit title: Organic Chemistry
 Unit title: Organic Chemistry Unit code: R/601/0352 QCF level: 4 Credit value: 15 Aim This unit develops the principles and practical techniques of organic chemistry. Rationalisation of structure and bonding
Unit title: Organic Chemistry Unit code: R/601/0352 QCF level: 4 Credit value: 15 Aim This unit develops the principles and practical techniques of organic chemistry. Rationalisation of structure and bonding
Fisika Polimer Ariadne L Juwono. Sem /2007
 Chapter 4. Ionic and coordination (addition) polymerization 4.1. Similarities and contrast on ionic polymerization 4.2. Cationic polymerization 4.3. Anionic polymerization 4.4. Coordination polymerization
Chapter 4. Ionic and coordination (addition) polymerization 4.1. Similarities and contrast on ionic polymerization 4.2. Cationic polymerization 4.3. Anionic polymerization 4.4. Coordination polymerization
ANSWER GUIDE APRIL/MAY 2006 EXAMINATIONS CHEMISTRY 249H
 AWER GUIDE APRIL/MAY 2006 EXAMIATI CEMITRY 249 1. (a) PDC / C 2 2 (b) t-bume 2 i (1 equiv) / imidazole (1 equiv) i TBDM protection of the less sterically hindered alcohol (c) BuLi (1 equiv) then (d) 2
AWER GUIDE APRIL/MAY 2006 EXAMIATI CEMITRY 249 1. (a) PDC / C 2 2 (b) t-bume 2 i (1 equiv) / imidazole (1 equiv) i TBDM protection of the less sterically hindered alcohol (c) BuLi (1 equiv) then (d) 2
Bio-elements. Living organisms requires only 27 of the 90 common chemical elements found in the crust of the earth, to be as its essential components.
 Bio-elements Living organisms requires only 27 of the 90 common chemical elements found in the crust of the earth, to be as its essential components. Most of the chemical components of living organisms
Bio-elements Living organisms requires only 27 of the 90 common chemical elements found in the crust of the earth, to be as its essential components. Most of the chemical components of living organisms
Chapter 15 Reactions of Aromatic Compounds
 Chapter 15 1 Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen
Chapter 15 1 Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen
An Introduction to Ionic Liquids. Michael Freemantle. RSC Publishing
 An to Ionic Liquids Michael Freemantle RSC Publishing Chapter 1 1 1.1 Definition of Ionic Liquids 1 1.2 Synonyms 1 1.3 Attraction of Ionic Liquids 2 1.4 Cations and Anions 3 1.5 Shorthand Notation for
An to Ionic Liquids Michael Freemantle RSC Publishing Chapter 1 1 1.1 Definition of Ionic Liquids 1 1.2 Synonyms 1 1.3 Attraction of Ionic Liquids 2 1.4 Cations and Anions 3 1.5 Shorthand Notation for
SURVEY ON ARYL COMPOUNDS
 Journal of Plastic and Polymer Technology (JPPT) Vol. 1, Issue 1, Jun 2015, 111-132 TJPRC Pvt. Ltd SURVEY ON ARYL COMPOUNDS NAGHAM MAHMOOD ALJAMALI Organic Chemistry, Department of Chemistry, College of
Journal of Plastic and Polymer Technology (JPPT) Vol. 1, Issue 1, Jun 2015, 111-132 TJPRC Pvt. Ltd SURVEY ON ARYL COMPOUNDS NAGHAM MAHMOOD ALJAMALI Organic Chemistry, Department of Chemistry, College of
16. Chemistry of Benzene: Electrophilic Aromatic Substitution جانشینی الکتروندوستی آروماتیک شیمی آلی 2
 16. Chemistry of Benzene: Electrophilic Aromatic Substitution جانشینی الکتروندوستی آروماتیک شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based
16. Chemistry of Benzene: Electrophilic Aromatic Substitution جانشینی الکتروندوستی آروماتیک شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based
16. Chemistry of Benzene: Electrophilic Aromatic Substitution جانشینی الکتروندوستی آروماتیک شیمی آلی 2
 16. Chemistry of Benzene: Electrophilic Aromatic Substitution جانشینی الکتروندوستی آروماتیک شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based
16. Chemistry of Benzene: Electrophilic Aromatic Substitution جانشینی الکتروندوستی آروماتیک شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based
Chapter 25: The Chemistry of Life: Organic and Biological Chemistry
 Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
E. Dithianes (S,S-Acetals)
 E. Dithianes (,-Acetals) bjectives By the end of this section you will be able to: 1) prepare,-acetals (dithianes) from aldehydes and ketones; 2) draw an arrow-pushing mechanism for the formation of dithianes
E. Dithianes (,-Acetals) bjectives By the end of this section you will be able to: 1) prepare,-acetals (dithianes) from aldehydes and ketones; 2) draw an arrow-pushing mechanism for the formation of dithianes
Chap 11. Carbonyl Alpha-Substitution Reactions and Condensation Reactions
 Chap 11. Carbonyl Alpha-Substitution eactions and Condensation eactions Four fundamental reactions of carbonyl compounds 1) Nucleophilic addition (aldehydes and ketones) ) Nucleophilic acyl substitution
Chap 11. Carbonyl Alpha-Substitution eactions and Condensation eactions Four fundamental reactions of carbonyl compounds 1) Nucleophilic addition (aldehydes and ketones) ) Nucleophilic acyl substitution
Molecular Geometry: VSEPR model stand for valence-shell electron-pair repulsion and predicts the 3D shape of molecules that are formed in bonding.
 Molecular Geometry: VSEPR model stand for valence-shell electron-pair repulsion and predicts the 3D shape of molecules that are formed in bonding. Sigma and Pi Bonds: All single bonds are sigma(σ), that
Molecular Geometry: VSEPR model stand for valence-shell electron-pair repulsion and predicts the 3D shape of molecules that are formed in bonding. Sigma and Pi Bonds: All single bonds are sigma(σ), that
Ch 20 Carboxylic Acids and Nitriles
 Ch 20 Carboxylic Acids and Nitriles Carboxylic Acids (RCO 2 H) are compounds with an OH attached to a carbonyl. Nitriles (RC N) are compounds a carbon-nitrogen triple bond. Naming Carboxylic Acids 1. Replace
Ch 20 Carboxylic Acids and Nitriles Carboxylic Acids (RCO 2 H) are compounds with an OH attached to a carbonyl. Nitriles (RC N) are compounds a carbon-nitrogen triple bond. Naming Carboxylic Acids 1. Replace
Chapter 16: Aromatic Compounds
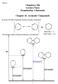 Chamras Chemistry 106 Lecture otes xamination 2 Materials Chapter 16: Aromatic Compounds Benzene, the Most Commonly Known Aromatic Compound: The aromatic nature of benzene stabilizes it 36 kcal.mol 1.
Chamras Chemistry 106 Lecture otes xamination 2 Materials Chapter 16: Aromatic Compounds Benzene, the Most Commonly Known Aromatic Compound: The aromatic nature of benzene stabilizes it 36 kcal.mol 1.
Exam 1 (Monday, July 6, 2015)
 Chem 231 Summer 2015 Assigned Homework Problems Last updated: Friday, July 24, 2015 Problems Assigned from Essential Organic Chemistry, 2 nd Edition, Paula Yurkanis Bruice, Prentice Hall, New York, NY,
Chem 231 Summer 2015 Assigned Homework Problems Last updated: Friday, July 24, 2015 Problems Assigned from Essential Organic Chemistry, 2 nd Edition, Paula Yurkanis Bruice, Prentice Hall, New York, NY,
AMINES. 3. Secondary When two hydrogen atoms are replaced by two alkyl or aryl groups.
 AMINES Amine may be regarded as derivative of ammonia formed by replacement of one or more hydrogen atoms by corresponding number of alkyl or aryl group CLASSIFICATION 1. Ammonia 2. Primary amine 3. Secondary
AMINES Amine may be regarded as derivative of ammonia formed by replacement of one or more hydrogen atoms by corresponding number of alkyl or aryl group CLASSIFICATION 1. Ammonia 2. Primary amine 3. Secondary
NANYANG TECHNOLOGICAL UNIVERSITY ENTRANCE EXAMINATION SYLLABUS FOR INTERNATIONAL STUDENTS CHEMISTRY
 NANYANG TECHNOLOGICAL UNIVERSITY ENTRANCE EXAMINATION SYLLABUS FOR INTERNATIONAL STUDENTS OAFA/01/07 STRUCTURE OF EXAMINATION PAPER CHEMISTRY 1. There will be one 2-hour paper consisting of two sections.
NANYANG TECHNOLOGICAL UNIVERSITY ENTRANCE EXAMINATION SYLLABUS FOR INTERNATIONAL STUDENTS OAFA/01/07 STRUCTURE OF EXAMINATION PAPER CHEMISTRY 1. There will be one 2-hour paper consisting of two sections.
Chapter 16 Chemistry of Benzene: Electrophilic Aromatic Substitution
 John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 16 Chemistry of Benzene: Electrophilic Aromatic Substitution Paul D. Adams University of Arkansas Substitution Reactions of Benzene and Its Derivatives
John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 16 Chemistry of Benzene: Electrophilic Aromatic Substitution Paul D. Adams University of Arkansas Substitution Reactions of Benzene and Its Derivatives
Chapter 19 Substitutions at the Carbonyl Group
 Chapter 19 Substitutions at the Carbonyl Group In Chapter 18 Additions to the Carbonyl Groups In Chapter 19 Substitutions at the Carbonyl Group O O - - O - O R Y R C+ Y R Y Nu -Ȳ R N u + Y=goodleavinggroup
Chapter 19 Substitutions at the Carbonyl Group In Chapter 18 Additions to the Carbonyl Groups In Chapter 19 Substitutions at the Carbonyl Group O O - - O - O R Y R C+ Y R Y Nu -Ȳ R N u + Y=goodleavinggroup
Importance of Carbohydrates
 Chapter 25 Importance of Carbohydrates Distributed widely in nature Key intermediates of metabolism (sugars) Structural components of plants (cellulose) Central to materials of industrial products: paper,
Chapter 25 Importance of Carbohydrates Distributed widely in nature Key intermediates of metabolism (sugars) Structural components of plants (cellulose) Central to materials of industrial products: paper,
Pyrrole reaction. Assis.Prof.Dr.Mohammed Hassan Lecture 4
 Pyrrole reaction Assis.Prof.Dr.Mohammed assan Lecture 4 Acidic properties of pyrrole Due to participation of lone pair in aromaticity), pyrrole has exceptionally strong acidic properties It can react with
Pyrrole reaction Assis.Prof.Dr.Mohammed assan Lecture 4 Acidic properties of pyrrole Due to participation of lone pair in aromaticity), pyrrole has exceptionally strong acidic properties It can react with
Ethers can be symmetrical or not:
 Chapter 14: Ethers, Epoxides, and Sulfides 175 Physical Properties Ethers can be symmetrical or not: linear or cyclic. Ethers are inert and make excellent solvents for organic reactions. Epoxides are very
Chapter 14: Ethers, Epoxides, and Sulfides 175 Physical Properties Ethers can be symmetrical or not: linear or cyclic. Ethers are inert and make excellent solvents for organic reactions. Epoxides are very
Mirabilis 2.0. Lhasa Limited vicgm. 11 January Martin Ott
 Mirabilis 2.0 Lhasa Limited vicgm 11 January 2017 Martin Ott Overview Introduction What is Mirabilis? Impurities Purge parameters and factors Scientific prioritisation Mirabilis reactivity knowledge matrix
Mirabilis 2.0 Lhasa Limited vicgm 11 January 2017 Martin Ott Overview Introduction What is Mirabilis? Impurities Purge parameters and factors Scientific prioritisation Mirabilis reactivity knowledge matrix
Keynotes in Organic Chemistry
 Keynotes in Organic Chemistry Second Edition ANDREW F. PARSONS Department of Chemistry, University of York, UK Wiley Contents Preface xi 1 Structure and bonding 1 1.1 Ionic versus covalent bonds 1 1.2
Keynotes in Organic Chemistry Second Edition ANDREW F. PARSONS Department of Chemistry, University of York, UK Wiley Contents Preface xi 1 Structure and bonding 1 1.1 Ionic versus covalent bonds 1 1.2
1. What is the major organic product obtained from the following sequence of reactions?
 CH320 N N_HW1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. Carefully record your answers on the Scantron
CH320 N N_HW1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. Carefully record your answers on the Scantron
N_HW1 N_HW1. 1. What is the purpose of the H 2 O in this sequence?
 N_HW1 N_HW1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. 1. What is the purpose of the H 2 O in this
N_HW1 N_HW1 Multiple Choice Identify the choice that best completes the statement or answers the question. There is only one correct response for each question. 1. What is the purpose of the H 2 O in this
Organic Chemistry. M. R. Naimi-Jamal. Faculty of Chemistry Iran University of Science & Technology
 Organic Chemistry M. R. Naimi-Jamal Faculty of Chemistry Iran University of Science & Technology Chapter 5-2. Chemistry of Benzene: Electrophilic Aromatic Substitution Based on McMurry s Organic Chemistry,
Organic Chemistry M. R. Naimi-Jamal Faculty of Chemistry Iran University of Science & Technology Chapter 5-2. Chemistry of Benzene: Electrophilic Aromatic Substitution Based on McMurry s Organic Chemistry,
ORGANIC - CLUTCH CH ALDEHYDES AND KETONES: NUCLEOPHILIC ADDITION
 !! www.clutchprep.com CONCEPT: ALDEHYDE NOMENCLATURE Replace the suffix of the alkane -e with the suffix On the parent chain, the carbonyl is always terminal, and receive a location As substituents, they
!! www.clutchprep.com CONCEPT: ALDEHYDE NOMENCLATURE Replace the suffix of the alkane -e with the suffix On the parent chain, the carbonyl is always terminal, and receive a location As substituents, they
Carbonyl Chemistry V + C O C. Chemistry /30/02
 arbonyl hemistry V Ō - + Keto-enol enol Tautomerism H 3 H 3 Ketone H H R 2 H R' H H 3 H 2 H Enol H Acid atalyzed α Halogenation R 2 R' + X 2 H + R 2 R' + HX H X X 2 can be l 2, Br 2, or I 2. Substitution
arbonyl hemistry V Ō - + Keto-enol enol Tautomerism H 3 H 3 Ketone H H R 2 H R' H H 3 H 2 H Enol H Acid atalyzed α Halogenation R 2 R' + X 2 H + R 2 R' + HX H X X 2 can be l 2, Br 2, or I 2. Substitution
Benzene and Aromatic Compounds. Chapter 15 Organic Chemistry, 8 th Edition John McMurry
 Benzene and Aromatic Compounds Chapter 15 Organic Chemistry, 8 th Edition John McMurry 1 Background Benzene (C 6 H 6 ) is the simplest aromatic hydrocarbon (or arene). Four degrees of unsaturation. It
Benzene and Aromatic Compounds Chapter 15 Organic Chemistry, 8 th Edition John McMurry 1 Background Benzene (C 6 H 6 ) is the simplest aromatic hydrocarbon (or arene). Four degrees of unsaturation. It
16. Chemistry of Benzene: Electrophilic Aromatic Substitution. Based on McMurry s Organic Chemistry, 7 th edition
 16. Chemistry of Benzene: Electrophilic Aromatic Substitution Based on McMurry s Organic Chemistry, 7 th edition Substitution Reactions of Benzene and Its Derivatives Benzene is aromatic: a cyclic conjugated
16. Chemistry of Benzene: Electrophilic Aromatic Substitution Based on McMurry s Organic Chemistry, 7 th edition Substitution Reactions of Benzene and Its Derivatives Benzene is aromatic: a cyclic conjugated
Carboxylic Acids O R C + H + O - Chemistry 618B
 arboxylic Acids R H R + H + - R - Nomenclature - IUPA IUPA names: drop the -e from the parent alkane and add the suffix -oic acid If the compound contains a carbon-carbon double bond, change the infix
arboxylic Acids R H R + H + - R - Nomenclature - IUPA IUPA names: drop the -e from the parent alkane and add the suffix -oic acid If the compound contains a carbon-carbon double bond, change the infix
C H Activated Trifluoromethylation
 Literature report C H Activated Trifluoromethylation Reporter:Yan Fang Superior:Prof. Yong Huang Jun. 17 th 2013 Contents Background Trifluoromethylation of sp-hybridized C-H Bonds Trifluoromethylation
Literature report C H Activated Trifluoromethylation Reporter:Yan Fang Superior:Prof. Yong Huang Jun. 17 th 2013 Contents Background Trifluoromethylation of sp-hybridized C-H Bonds Trifluoromethylation
Lecture 27 Organic Chemistry 1
 CHEM 232 rganic Chemistry I at Chicago Lecture 27 rganic Chemistry 1 Professor Duncan Wardrop April 20, 2010 1 Self Test Question Nitrosonium (not nitronium) cations can be generated by treating sodium
CHEM 232 rganic Chemistry I at Chicago Lecture 27 rganic Chemistry 1 Professor Duncan Wardrop April 20, 2010 1 Self Test Question Nitrosonium (not nitronium) cations can be generated by treating sodium
Best-in-class API / HPAPI / Small Molecules Custom Manufacturing
 Best-in-class API / HPAPI / Small Molecules Custom Manufacturing Six production locations countless options. USA: TIPPECANOE World s largest HPAPI capacity Four cgmp plants and pilot plant, 140 vessels,
Best-in-class API / HPAPI / Small Molecules Custom Manufacturing Six production locations countless options. USA: TIPPECANOE World s largest HPAPI capacity Four cgmp plants and pilot plant, 140 vessels,
Chapter 15. Reactions of Aromatic Compounds. Electrophilic Aromatic Substitution on Arenes. The first step is the slow, rate-determining step
 Electrophilic Aromatic Substitution on Arenes Chapter 15 Reactions of Aromatic Compounds The characteristic reaction of aromatic rings is substitution initiated by an electrophile halogenation nitration
Electrophilic Aromatic Substitution on Arenes Chapter 15 Reactions of Aromatic Compounds The characteristic reaction of aromatic rings is substitution initiated by an electrophile halogenation nitration
There are two main electronic effects that substituents can exert:
 Substituent Effects There are two main electronic effects that substituents can exert: RESONANCE effects are those that occur through the π system and can be represented by resonance structures. These
Substituent Effects There are two main electronic effects that substituents can exert: RESONANCE effects are those that occur through the π system and can be represented by resonance structures. These
Chapter 9. Organic Chemistry: The Infinite Variety of Carbon Compounds. Organic Chemistry
 Chapter 9 Organic Chemistry: The Infinite Variety of Carbon Compounds Organic Chemistry Organic chemistry is defined as the chemistry of carbon compounds. Of tens of millions of known chemical compounds,
Chapter 9 Organic Chemistry: The Infinite Variety of Carbon Compounds Organic Chemistry Organic chemistry is defined as the chemistry of carbon compounds. Of tens of millions of known chemical compounds,
CHEM*2700 ORGANIC CHEMISTRY I (Spring/Summer Semester 2009) Information Sheet and Course Outline
 CHEM*2700 ORGANIC CHEMISTRY I (Spring/Summer Semester 2009) Information Sheet and Course Outline Instructor: Professor William Tam Office: MacN 332 Phone: 824-4120 (Ext.52268) E-mail: wtam@uoguelph.ca
CHEM*2700 ORGANIC CHEMISTRY I (Spring/Summer Semester 2009) Information Sheet and Course Outline Instructor: Professor William Tam Office: MacN 332 Phone: 824-4120 (Ext.52268) E-mail: wtam@uoguelph.ca
media), except those of aluminum and calcium
 1- Aspirin occurs as white crystals or as a white crystalline powder. 2- It is slightly soluble in water (1:300), soluble in alcohol (1 :5), chloroform (1:17) & ether (1:15). It dissolves easily in glycerin.
1- Aspirin occurs as white crystals or as a white crystalline powder. 2- It is slightly soluble in water (1:300), soluble in alcohol (1 :5), chloroform (1:17) & ether (1:15). It dissolves easily in glycerin.
SAR. Structure - Activity Relationships (alkoholy, amíny, aldehydy, ketóny, estery, amidy, kyseliny, uhľovodíky) 2/28/2016
 Structure Activity elationships (SA) identifies which functional groups are important for binding and activity SA Structure - Activity elationships (alkoholy, amíny, aldehydy, ketóny, estery, amidy, kyseliny,
Structure Activity elationships (SA) identifies which functional groups are important for binding and activity SA Structure - Activity elationships (alkoholy, amíny, aldehydy, ketóny, estery, amidy, kyseliny,
Chemistry 254 Lab Experiment 1: Qualitative Organic Analysis Summer 2004
 Chemistry 254 Lab Experiment 1: Qualitative Organic Analysis Summer 2004 Introduction: Qualitative organic analysis, the identification and characterization of unknown compounds, in an important part of
Chemistry 254 Lab Experiment 1: Qualitative Organic Analysis Summer 2004 Introduction: Qualitative organic analysis, the identification and characterization of unknown compounds, in an important part of
24.4: Acidity of Phenols. Phenols are more acidic than aliphatic alcohols. + Electron-withdrawing groups make an O
 Chapter 24: Phenols. Alcohols contain an group bonded to an sp 3 -hybridized carbon. Phenols contain an group bonded to an sp 2 -hybridized carbon of a benzene ring 24.1: Nomenclature (please read) 24.2:
Chapter 24: Phenols. Alcohols contain an group bonded to an sp 3 -hybridized carbon. Phenols contain an group bonded to an sp 2 -hybridized carbon of a benzene ring 24.1: Nomenclature (please read) 24.2:
Carboxylic Acids and Nitriles
 Carboxylic Acids and Nitriles Why this Chapter? Carboxylic acids present in many industrial processes and most biological processes They are the starting materials from which other acyl derivatives are
Carboxylic Acids and Nitriles Why this Chapter? Carboxylic acids present in many industrial processes and most biological processes They are the starting materials from which other acyl derivatives are
Anderson Development Company. Almabor TM. Tailor Made Organoborates
 Anderson Development Company H Almabor TM Tailor Made rganoborates Almabor TM Tailor Made rganoborates 1415 E. Michigan Street Adrian, MI 49221 Phone: 517.263.2121 Fax: 517.263.1000 www.andersondevelopment.com
Anderson Development Company H Almabor TM Tailor Made rganoborates Almabor TM Tailor Made rganoborates 1415 E. Michigan Street Adrian, MI 49221 Phone: 517.263.2121 Fax: 517.263.1000 www.andersondevelopment.com
20.5 Preparation of Amines
 RGANIC CHEMISTRY 20.5 Preparation of Amines RGANIC CHEMISTRY 20.5A Through Nucleophilic Substitution Reactions Alkylation of Ammonia or Amines NaH NH 3 + R X RNH 3 X RNH2 R + R X R 2 X NaH R 2 NH R 2 NH
RGANIC CHEMISTRY 20.5 Preparation of Amines RGANIC CHEMISTRY 20.5A Through Nucleophilic Substitution Reactions Alkylation of Ammonia or Amines NaH NH 3 + R X RNH 3 X RNH2 R + R X R 2 X NaH R 2 NH R 2 NH
Chemistry of Benzene: Electrophilic Aromatic Substitution
 Chemistry of Benzene: Electrophilic Aromatic Substitution Why this Chapter? Continuation of coverage of aromatic compounds in preceding chapter focus shift to understanding reactions Examine relationship
Chemistry of Benzene: Electrophilic Aromatic Substitution Why this Chapter? Continuation of coverage of aromatic compounds in preceding chapter focus shift to understanding reactions Examine relationship
Mechanisms. . CCl2 F + Cl.
 Mechanisms 1) Free radical substitution Alkane à halogenoalkane Initiation: Propagation: Termination: Overall: 2) Ozone depletion UV light breaks the C Cl bond releasing chlorine radical CFCl 3 F à. CCl2
Mechanisms 1) Free radical substitution Alkane à halogenoalkane Initiation: Propagation: Termination: Overall: 2) Ozone depletion UV light breaks the C Cl bond releasing chlorine radical CFCl 3 F à. CCl2
Aldehydes, Ketones and Carboxylic acids
 Teacher Orientation Aldehydes, Ketones and Carboxylic Acids contains following topics: Nomenclature Preparation Properties Student Orientation Preparation and Properties Of Aldehydes, Ketones and Carboxylic
Teacher Orientation Aldehydes, Ketones and Carboxylic Acids contains following topics: Nomenclature Preparation Properties Student Orientation Preparation and Properties Of Aldehydes, Ketones and Carboxylic
1.1 Is the following molecule aromatic or not aromatic? Give reasons for your answer.
 Page 1 QUESTION ONE 1.1 Is the following molecule aromatic or not aromatic? Give reasons for your answer. 1.2 List four criteria which compounds must meet in order to be considered aromatic. Page 2 QUESTION
Page 1 QUESTION ONE 1.1 Is the following molecule aromatic or not aromatic? Give reasons for your answer. 1.2 List four criteria which compounds must meet in order to be considered aromatic. Page 2 QUESTION
Module No and Title. PAPER No: 5 ; TITLE : Organic Chemistry-II MODULE No: 25 ; TITLE: S E 1 reactions
 Subject Chemistry Paper No and Title Module No and Title Module Tag 5; Organic Chemistry-II 25; S E 1 reactions CHE_P5_M25 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction 3. S E 1 reactions 3.1
Subject Chemistry Paper No and Title Module No and Title Module Tag 5; Organic Chemistry-II 25; S E 1 reactions CHE_P5_M25 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction 3. S E 1 reactions 3.1
PAPER No. 5: REACTION MECHANISM MODULE No. 2: Types of Organic Reaction Mechanisms
 Subject Chemistry Paper No and Title Module No and Title Module Tag Paper No. 5:Organic Chemistry-II Module No. 2: Overview of different types of Organic Reaction Mechanisms CHE_P5_M2 TABLE OF CONTENTS
Subject Chemistry Paper No and Title Module No and Title Module Tag Paper No. 5:Organic Chemistry-II Module No. 2: Overview of different types of Organic Reaction Mechanisms CHE_P5_M2 TABLE OF CONTENTS
A-LEVEL A-LEVEL CHEMISTRY CHEMISTRY NOTES
 A-LEVEL A-LEVEL CHEMISTRY CHEMISTRY NOTES snaprevise.co.uk I have designed and compiled these beautiful notes to provide a detailed but concise summary of this module. I have spent a lot of time perfecting
A-LEVEL A-LEVEL CHEMISTRY CHEMISTRY NOTES snaprevise.co.uk I have designed and compiled these beautiful notes to provide a detailed but concise summary of this module. I have spent a lot of time perfecting
Chapter 9 Aldehydes and Ketones
 Chapter 9 Aldehydes and Ketones 9.1 Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al The aldehyde functional group is always carbon
Chapter 9 Aldehydes and Ketones 9.1 Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al The aldehyde functional group is always carbon
A. Reaction Mechanisms and Catalysis (1) proximity effect (2) acid-base catalysts (3) electrostatic (4) functional groups (5) structural flexibility
 (P&S Ch 5; Fer Ch 2, 9; Palm Ch 10,11; Zub Ch 9) A. Reaction Mechanisms and Catalysis (1) proximity effect (2) acid-base catalysts (3) electrostatic (4) functional groups (5) structural flexibility B.
(P&S Ch 5; Fer Ch 2, 9; Palm Ch 10,11; Zub Ch 9) A. Reaction Mechanisms and Catalysis (1) proximity effect (2) acid-base catalysts (3) electrostatic (4) functional groups (5) structural flexibility B.
ACS Green Chemistry Institute. ACS Green Chemistry Institute
 American Chemical Society Advancing Green Chemistry Innovation in the Pharmaceutical Industry: The ACS GCI Pharmaceutical Roundtable s Research Grant Program Julie B. Manley Guiding Green LLC Contracted
American Chemical Society Advancing Green Chemistry Innovation in the Pharmaceutical Industry: The ACS GCI Pharmaceutical Roundtable s Research Grant Program Julie B. Manley Guiding Green LLC Contracted
The Organic Acids. Carboxylic Acids * *
 arboxylic Acids The rganic Acids Some Notation: Acids and their conjugate bases pka ~ 15 - weak acid arboxylic Acid pka ~ 4 moderate acid - arboxylate Anion pka ~ -7 very strong acid l - l arboxylic acids
arboxylic Acids The rganic Acids Some Notation: Acids and their conjugate bases pka ~ 15 - weak acid arboxylic Acid pka ~ 4 moderate acid - arboxylate Anion pka ~ -7 very strong acid l - l arboxylic acids
TUTORIAL LIST 2017/18
 TUTORIAL LIST 2017/18 ATOMIC STRUCTURE & MOLE CALCULATIONS BONDING & INORGANIC CHEMISTRY PHYSICAL CHEMISTRY ORGANIC CHEMISTRY PRACTICAL CHEMISTRY PAST PAPER WALKTHROUGHS ATOMIC STRUCTURE & MOLE CALCULATIONS
TUTORIAL LIST 2017/18 ATOMIC STRUCTURE & MOLE CALCULATIONS BONDING & INORGANIC CHEMISTRY PHYSICAL CHEMISTRY ORGANIC CHEMISTRY PRACTICAL CHEMISTRY PAST PAPER WALKTHROUGHS ATOMIC STRUCTURE & MOLE CALCULATIONS
Intermediates. THF Solutions. Expanded Tetrahydrofuran Offer for Pharma Customers in Europe
 Intermediates THF Solutions Expanded Tetrahydrofuran Offer for Pharma Customers in Europe BASF We create chemistry About BASF At BASF, we create chemistry and have been doing so for 150 years. Our portfolio
Intermediates THF Solutions Expanded Tetrahydrofuran Offer for Pharma Customers in Europe BASF We create chemistry About BASF At BASF, we create chemistry and have been doing so for 150 years. Our portfolio
22.7 Reactions of Amines: A Review and a Preview
 22.7 Reactions of Amines: A Review and a Preview Preparation of Amines Two questions to answer: 1) How is the C N C N bond to be formed? 2) How do we obtain the correct oxidation state of nitrogen (and
22.7 Reactions of Amines: A Review and a Preview Preparation of Amines Two questions to answer: 1) How is the C N C N bond to be formed? 2) How do we obtain the correct oxidation state of nitrogen (and
2 Answer all the questions. 1 Alkenes and benzene both react with bromine but alkenes are much more reactive.
 2 Answer all the questions. 1 Alkenes and benzene both react with bromine but alkenes are much more reactive. (a) Explain the relative resistance to bromination of benzene compared with alkenes. In your
2 Answer all the questions. 1 Alkenes and benzene both react with bromine but alkenes are much more reactive. (a) Explain the relative resistance to bromination of benzene compared with alkenes. In your
CHEM 251 (4 credits): Description
 CHEM 251 (4 credits): Intermediate Reactions of Nucleophiles and Electrophiles (Reactivity 2) Description: An understanding of chemical reactivity, initiated in Reactivity 1, is further developed based
CHEM 251 (4 credits): Intermediate Reactions of Nucleophiles and Electrophiles (Reactivity 2) Description: An understanding of chemical reactivity, initiated in Reactivity 1, is further developed based
Chapter 8: Ethers and Epoxides. Diethyl ether in starting fluid
 Chapter 8: Ethers and Epoxides Diethyl ether in starting fluid 8.1 Nomenclature of Ethers Ethers are usually named by giving the name of each alkyl or aryl group, in alphabetical order, followed by the
Chapter 8: Ethers and Epoxides Diethyl ether in starting fluid 8.1 Nomenclature of Ethers Ethers are usually named by giving the name of each alkyl or aryl group, in alphabetical order, followed by the
Abstract Process Economics Program Report 37B ACETIC ACID AND ACETIC ANHYDRIDE (November 1994)
 Abstract Process Economics Program Report 37B ACETIC ACID AND ACETIC ANHYDRIDE (November 1994) This Report presents preliminary process designs and estimated economics for the manufacture of acetic acid
Abstract Process Economics Program Report 37B ACETIC ACID AND ACETIC ANHYDRIDE (November 1994) This Report presents preliminary process designs and estimated economics for the manufacture of acetic acid
INTRODUCTION... XVII
 INTRODUCTION... XVII CHAPTER ONE: ANTISEPTICS AND DISINFECTANTS (CHM052B)... 1 INTRODUCTION... 1 STUDY OBJECTIVES... 1 REASONS FOR DOING THIS STUDY... 1 INTENDED AUDIENCE... 2 SCOPE OF THE STUDY... 2 METHODOLOGY...
INTRODUCTION... XVII CHAPTER ONE: ANTISEPTICS AND DISINFECTANTS (CHM052B)... 1 INTRODUCTION... 1 STUDY OBJECTIVES... 1 REASONS FOR DOING THIS STUDY... 1 INTENDED AUDIENCE... 2 SCOPE OF THE STUDY... 2 METHODOLOGY...
Nucleophilic attack on ligand
 Nucleophilic attack on ligand Nucleophile "substitutes" metal hapticity usually decreases xidation state mostly unchanged Competition: nucleophilic attack on metal usually leads to ligand substitution
Nucleophilic attack on ligand Nucleophile "substitutes" metal hapticity usually decreases xidation state mostly unchanged Competition: nucleophilic attack on metal usually leads to ligand substitution
DEPARTMENT: Chemistry
 CODE CHEM 204 TITLE: Organic Chemistry II INSTITUTE: STEM DEPARTMENT: Chemistry COURSE DESCRIPTION: A continuation of CHEM-203, students will extend their studies into topics including aromatic hydrocarbons,
CODE CHEM 204 TITLE: Organic Chemistry II INSTITUTE: STEM DEPARTMENT: Chemistry COURSE DESCRIPTION: A continuation of CHEM-203, students will extend their studies into topics including aromatic hydrocarbons,
