Bicyclic helical peptides as dual inhibitors selective. for Bcl2A1 and Mcl-1 proteins
|
|
|
- Tamsyn Ross
- 5 years ago
- Views:
Transcription
1 Supporting Inormation Bicyclic helical peptides as dual inhibitors selective or Bcl2A1 and Mcl-1 proteins de Araujo A.D.; Lim, J.; Wu, K-C.; Xiang, Y.; Good, A. C.; Skerlj, R.; Fairlie, D. P.* Contents Page Figure S1. Synthetic solid phase route or preparing synthetic cyclic peptides S2-3 Figure S2. Inhibition o A1/FBid complex measured by FP assay. Figure S3. Denaturing SDS-PAGE analysis or covalent adduct ormation with A1. Figure S4. Mass spectra o A1-peptide conjugates. S4 S4 S5 Figure S5. MS/MS analysis o trypsin-digested electrophoresis gel band containing protein ragment bearing Cys55 covalently bonded to peptide 5. Figure S6. Stability o BIM, 1, 5 and 8 in rat plasma. Figure S7. Stability o BIM, 1 and 5 with trypsin or chymotrypsin. S6 S7 S7 Figure S8. Western blot showing covalent bonding o intracellular A1 to peptides 5 and 10 in live SKMel28 cells ater 24h incubation. Figure S9. SKMel28 cell viability ater incubation with BIM or BimSAB A or 48 h Figure S10. Analytical PLC showing retention times o puriied peptides 5 and 8. Figure S11. UV spectra or puriied peptides 2-10 measured by UPLC. Figure S12. ESI-MS mass spectra or puriied peptides Table S1. IC 50 and Ki values or inhibition o Bcl2 protein/fbid complexes. Table S2. Structure and MS analysis o peptides synthesized in this study. S8 S8 S9 S10 S11 S12 S13 S1
2 A Step Reagents and conditions B SPPS a,d a b c d e SPPS: a) 30% piperidine in DMF (2 x 3 min); b) Fmoc-AA- (4 equiv), CTU (4 equiv), DIPEA (8 equiv), 2 x 30 min 10 mm Grubbs catalyst 1 st generation in dry DCE, 2 x 2 h 2% TFA in DCM, 10 x 1 min a) 30% piperidine in DMF (2 x 3 min); b) 2 (1 M): DIPEA (0.4M), 2 x 5 min acrylic acid (4 equiv), ATU (4 equiv), DIPEA (4 equiv) in DMF, 15 min I A Q E L R S 5 I G D S 5 F * b Mtt Mtt c,e g h TFA:TIS: 2 (95:2.5:2.5), 2 h 30 % piperidine in DMF, 2 x 3 min PyBP (4 equiv), DIPEA (8 equiv) in DMF, 4-6 h * i j k acrylic acid (1.2 equiv), ATU (1.2 equiv), DIPEA (2.4 equiv) in DMF, 30 min propiolic acid (1.2 equiv), ATU (1.2 equiv), DIPEA (2.4 equiv) in DMF, 30 min FITC (2 equiv), DIPEA (4 equiv) in DMF, overnight 2 Cycle 2 C SPPS a D SPPS a,d Mtt Pip Fmoc Fmoc E L R S 5 I G D S 5 F b E L R S 5 I G D S 5 F a,d SI 5 X I A SQ 5 E L R I G D F Mtt b Mtt * * c,e * K I A QD E L R S 5 I G D S 5 F Boc c,h I A Q E L R S 5 I G D S 5 F Boc b Boc 2 Bicycle i 2 Bicycle 4 S2
3 E Fmoc I A Q E L R K I G D D F c,h Fmoc I A SQ 5 E L R I G D F a,d SI 5 X I A SQ 5 E L R I G D F Mtt b Mtt c,e 2 SPPS a S 5 Mtt Pip Bicycle 5 c,j 2 Bicycle 7 F SPPS a I A Q E L R * * Boc I A Q E L R Mtt Pip Fmoc E L R K I G D D F c,h Fmoc E L R I G D F a,d Mtt Pip K I A QD E L R I G D F Boc c,h 2 i I A Q E L R Bicycle 6 I G D * F I G D F I G D F 2 2 G SPPS a Fmoc I A Q E L R K I G D D F c,h Fmoc I A SQ 5 E L R I G D F a Fmoc-βAla SI 5 X I A SQ 5 E L R I G D F Mtt b Fmoc-βAla Mtt c,j Fmoc-βAla g,k FITC-βAla FITC-βAla 2 S 5 Mtt Pip Bicycle 9 Figure S1. Synthetic solid phase route or preparing cyclic truncated peptides A. Reaction conditions and reagents used or each synthetic step depicted in B (peptide 2), C (peptide 3), D (peptide 4), E (peptides 5 and 7, similar route or peptide 8 and 10), F (peptide 6) and G (peptide 9). Asterisks denote that a standard protecting group was used or the amino acid side chain. S3
4 IC50 (nm) BIM time (min) Figure S2. Inhibition o A1/FBid complex measured by FP assay. Time-course o changing IC 50 or inhibition o the FBid/A1 complex measured by competitive FP assay. Assay conditions: FBid (5 nm) was combined with A1 (15 nm) or 30 min in running buer (50 mm Tris, 150mM acl, 1 mm EDTA, 1 mm TCEP, 0.005% Tween-20, p 7.2.) and combined with a serial dilution o the peptide (ranging rom 8 µm to 0.1 nm). Ater 15, 30, 60, 90 and 120 min incubation at rt, luorescence polarization was measured and IC 50 values were determined by nonlinear regression analysis using Prism sotware 7.0. A Buer BIM kda Figure S3. Denaturing SDS-PAGE analysis or covalent adduct ormation with A1. rylamide-containing peptides (1-6) covalently bind to A1 (showing an upper band shit or the protein ater incubation with peptide), whereas those without an electrophile do not orm a covalent adduct with A1 (BIM and 7). S4
5 20492 A A A A A m/z Figure S4. Mass spectra o A1-peptide conjugates. Incubation o A1 (1 µm, MW: 18756) with peptides 2-6 (10 µm) or 2-6 h in buer p 7.2, showing the expected molecular weight (within ±1 Da o calculated mass). o evidence o multiple addition o the peptide to the protein was ound. Conditions: A1 and peptides were combined in buer 50 mm Tris, 150mM acl, 1 mm EDTA, 2 mm TCEP, p 7.2 to a inal concentration o 1 µm A1 and 10 µm peptide in a total o 20 µl reaction volume. The reaction was analyzed by LC-MS on a Shimadzu exera uplc system coupled to a Triple TF 5600 mass spectrometer (ABSCIEX) using Zorbax C18 column (Agilent). S5
6 Bicycle 5/Bl-1 covalent conjugate in gel trypsin digestion LK I A Q E L R S SC 55 LDVVVSLDTAR TLF I G D F Intensity, cps Data Set ame: 3000 MS/MS or ragment [M+3] +3 R LR ELR SLDTAR VSLDTAR VVSLDTAR I A Q E L R 2500 S TAR VVSLDTAR LDVVVSLDTAR LDTAR DVVVSLDTAR DTAR TF Product (880.1): Exp 3, [M+3]+3 min rom Sample 1 (Aline AR VVVSLDTAR 0.1% o tallest A 47 D) peak o Aline A 47 D.wi m/z, Da Scan or combined ELR and LDVVVSLDTAR sequences S SCLDVVVSLDTAR only ragment containing sequence ELR +TF Product (880.1): Exp 3, min rom Sample 1 (Aline A 47 D) o Aline A 47 D.wi General Precursor MW: Precursor Charge: 3 Mass tolerance: 0.06 Da -terminal Modiication: (none) Cysteine Modiication: (none) C-terminal Modiication: (none) Methionine Modiication: (none) Digest Agent: Trypsin Present AAs: (none speciied) Missing AAs: (none speciied) Fragment Ions Data Mass Set Type: ame: Monoisotopic m/z Tolerance: 0.06 Da Minimum Intensity: General Peptide Precursor Sequence MW: Results: (none) Precursor Charge: 3 Mass tolerance: 0.06 Da -terminal Modiication: (none) Partial Cysteine Sequence Modiication: Tag Results: (none) (none) C-terminal Modiication: (none) Methionine Modiication: (none) Digest Agent: Trypsin Matches: Present AAs: 28 (none speciied) Missing AAs: (none speciied) ypertag: MS/MS Fragment Ions or (none) ragment ELR [M+3] +3 (none) : matching MS ingerprint or sequence ELR Ion Charge: Mass Type: 1 Monoisotopic m/z Tolerance: 0.06 Da Fragments: Minimum Intensity: 0.1% o tallest peak Residue Mass Immo a a-3 b b-2 b-3 b-3 b- c x y y-2 y-3 y-3 z E, Glu Peptide Sequence Results: (none) L, Leu R, Arg Partial Sequence Tag Results: (none) Matches: 131 ypertag: (none) LDVVVSLDTAR (none) Ion Charge: 1 Fragments: Residue Mass Immo a a-3 b b-2 b-3 b-3 b- c x y y-2 y-3 y-3 z L, Leu D, Asp , Asn V, Val , Asn V, Val V, Val S, Ser L, Leu D, Asp T, Thr A, Ala R, Arg MS/MS or ragment [M+3] +3 : matching MS ingerprint or sequence LDVVVSLDTAR Figure S5. MS/MS analysis o trypsin-digested electrophoresis gel band containing protein ragment bearing Cys55 covalently bonded to peptide 5. The MS/MS ingerprint or m/z ion revealed the expected y ion ragmentation pattern or the A1 derived-sequence LDVVVSVDTAR and the peptide 5 derived-sequence ELR. S6
7 Figure S6. Stability o BIM, 1, 5 and 8 in rat plasma. Time-course degradation o BIM and peptides 1, 5 and 8 (10 µm) in resh rat plasma (containing 1% DMS) at 37 C over 24 h (n = 3). TRYPSI CYMTRYPSI XIC o +TF MS: 400 to 1800 Da rom Sample 1 (J51A) o J51A.wi (DuoSpray ()) Max. 4.1e7 cps. 1.7e7 1.6e I W I A Q E L R R I G D E F A Y Y A R R 1.4e7 I W I A Q E L R R I G D E F A Y Y A R R 1.2e7 BIM Intensity, cps 1.0e7 8.0e6 6.0e6 BIM e6 2.0e6 1.08e7 1.00e XIC o +TF MS: to Da rom Sample (J51B) 4.0 o J51B.wi 5.0 (DuoSpray 6.0()) Max. 3.7e cps. Time, min e7 3.0e7 Intensity, cps Intensity, cps 9.00e6 8.00e6 7.00e6 6.00e6 5.00e6 4.00e6 3.00e6 2.00e6 1.00e XIC 0.00 o +TF MS: to Da rom 3.0 Sample 4.01 (Aline S F 6) o Aline Max e7 cps Time, min e7 1.8e7 1.6e7 1.4e7 1.2e7 1.0e7 8.0e6 A Y Y A R R 1 5 Intensity, cps Intensity, cps 2.5e7 2.0e7 1.5e7 1.0e7 5.0e6 0.0 XIC o +TF MS: to Da rom Sample (J51E) 4.0 o J51E.wi 5.0 (DuoSpray 6.0()) Max. 3.4e7 cps Time, min e7 3.0e7 2.5e7 2.0e7 1.5e7 A Y Y A R R e e7 4.0e6 2.0e e Time, min Time, min Figure S7. Stability o BIM, 1 and 5 with trypsin or chymotrypsin. Stability o BIM, 1 and 5 (10 µm) in the presence o trypsin (1:150, protease:peptide ratio) or chymotrypsin (1:50, protease:peptide ratio) in 50 mm ammonium bicarbonate buer ater 1h incubation at 37 0 C. The integrity o the peptides in the presence o the proteases was analysed by LC-MS, and ragmentation determined by MS. Yellow arrows pointed to retention time o intact peptide. Dotted red lines indicated site o proteolytic cleavage. S7
8 Bicycle 5 (µm) Bicycle 10 (µm) Bl GAPD Figure S8. Western blot showing covalent bonding o intracellular A1 to peptides 5 and 10 in live SKMel28 cells ater 24 h incubation. 125 % cell viability BimSAB A BIM log [peptide] (M) Figure S9. SKMel28 cell viability ater incubation with BIM or BimSAB A or 48 h. BIM is not active up to 50 µm while BimSAB A displayed a dose-dependent activity with a IC 50 o 15.7 µm. S8
9 mau 120 bicycle min mau bicycle min Figure S10. Analytical PLC showing the retention times o puriied peptides 5 and 8. Conditions: Phenomenex Luna C18 5 um (250 x 4.60 mm) column eluting at a low rate o 1 ml/min and a gradient o 30 to 100 % buer B (90% C 3 C/10% 2 /0.1% TFA in buer A, 0.1% TFA in water) over 20 min. S9
10 Figure S11. UV spectra or puriied peptides 2-10 measured by UPLC. Conditions: Shimadzu UPLC system connected to LCMS-2020 single quadrupole mass spectrometer using an Agilent Zorbax R-DS III column (Fig.S9) and and a gradient o 20 to 80 % buer B (90% C 3 C/10% 2 /0.1% ormic acid in buer A, 0.1% ormic acid in water) over 6 min. For peptide 9, gradient was 30 to 100% B over 6 min. S10
11 9 Figure S12. ESI-UPLC-MS mass spectra or puriied peptides Calculated expected mass ound in Table S2. S11
12 Table S1. IC 50 and Ki values or inhibition o Bcl2 protein/fbid complexes measured by competitive FP assays ater 2h incubation with peptide. Ki values were calculated rom the IC 50 values accordingly to reerence 30. (nd = not determined). IC 50 and K i or A1/FBid inhibition o these peptides is time-dependent, here are the calculated values ater 2h incubation. IC 50 (nm) Bcl-w Bcl-xL Bcl-2 Mcl-1 A1 BIM 38.1 ± ± ± ± ± ± ± ± ± ± 0.7* ± ± ± ± ± 4.2* 3 > 3200 >32000 > ± ± 1.5* 4 > ± ± 1.1* ± ± ± ± ± 0.2* ± ± ± ± ± 0.3* ± ± ± ± ± ± ± ± ± ± nd nd nd 63.0 ± ± 0.2* * IC 50 or A1/FBid inhibition o these peptides is time-dependent. ere is IC 50 ater 2h incubation. Ki (nm) Bcl-w Bcl-xL Bcl-2 Mcl-1 A1 BIM < ± 0.3 < 1 < ± < ± ± 0.6 < 1 < 1* ± ± ± ± ± 1.2* 3 > 1000 > 1000 > 1000 < 1 < 1* 4 > ± ± 24 < ± 0.3* ± ± ± 2.0 < 1 < 1* ± ± ± 4.0 < 1 < 1* ± ± ± 6 < 1 < ± ± ± 7 < ± nd nd nd < 1 < 1* * K i or A1/FBid inhibition o these peptides is time-dependent. ere is K i ater 2h incubation. S12
13 Table S2. Structure and MS analysis o synthetic peptides synthesized in this study. Cycle 1 was synthesized as described (re 22). Compound Sequence Expected mass bserved mass I A Q L L R I G D F FITC-βAla I A Q L L R I G D F I A Q L L R I G D F BIM -IWIAQELRRIGDEFAYYARR S13
Other Methods for Generating Ions 1. MALDI matrix assisted laser desorption ionization MS 2. Spray ionization techniques 3. Fast atom bombardment 4.
 Other Methods for Generating Ions 1. MALDI matrix assisted laser desorption ionization MS 2. Spray ionization techniques 3. Fast atom bombardment 4. Field Desorption 5. MS MS techniques Matrix assisted
Other Methods for Generating Ions 1. MALDI matrix assisted laser desorption ionization MS 2. Spray ionization techniques 3. Fast atom bombardment 4. Field Desorption 5. MS MS techniques Matrix assisted
Self-Assembly of Single Amino acid-pyrene Conjugates with Unique Structure-Morphology Relationship
 Electronic Supplementary Material (ESI) for Soft Matter. This journal is The Royal Society of Chemistry 2017 Self-Assembly of Single Amino acid-pyrene Conjugates with Unique Structure-Morphology Relationship
Electronic Supplementary Material (ESI) for Soft Matter. This journal is The Royal Society of Chemistry 2017 Self-Assembly of Single Amino acid-pyrene Conjugates with Unique Structure-Morphology Relationship
1 Supporting Information. 2 3 Materials and methods: 4 Chemicals: Fmoc-amino acids were obtained from GL Biochem (Shanghai).
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 1 Supporting Information 2 3 Materials and methods: 4 Chemicals: Fmoc-amino acids were obtained
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 1 Supporting Information 2 3 Materials and methods: 4 Chemicals: Fmoc-amino acids were obtained
Supporting Information. A fluorogenic assay for screening Sirt6 modulators
 This journal is The Royal Society of Chemistry 213 Supporting Information A fluorogenic assay for screening Sirt6 modulators Jing Hu, Bin He, Shiva Bhargava, Hening Lin* Department of Chemistry and Chemical
This journal is The Royal Society of Chemistry 213 Supporting Information A fluorogenic assay for screening Sirt6 modulators Jing Hu, Bin He, Shiva Bhargava, Hening Lin* Department of Chemistry and Chemical
Postsynthetic modification of unprotected peptides via S-tritylation reaction
 Supporting Information Postsynthetic modification of unprotected peptides via S-tritylation reaction Masayoshi Mochizuki, Hajime Hibino and Yuji Nishiuchi *,, SAITO Research Center, Peptide Institute,
Supporting Information Postsynthetic modification of unprotected peptides via S-tritylation reaction Masayoshi Mochizuki, Hajime Hibino and Yuji Nishiuchi *,, SAITO Research Center, Peptide Institute,
Supporting Information
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting Information Tuning the Thermosensitive Properties of Hybrid Collagen Peptide-Polymer
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting Information Tuning the Thermosensitive Properties of Hybrid Collagen Peptide-Polymer
Strategies for the Analysis of Therapeutic Peptides in Biofluids by LC-MS/MS. Lee Goodwin
 Strategies for the Analysis of Therapeutic Peptides in Biofluids by LC-MS/MS Lee Goodwin Sample Preparation Chromatography Detection General Strategies Examples New Approaches Summary Outline ABUNDANCE
Strategies for the Analysis of Therapeutic Peptides in Biofluids by LC-MS/MS Lee Goodwin Sample Preparation Chromatography Detection General Strategies Examples New Approaches Summary Outline ABUNDANCE
Supporting Information
 Supporting Information Syntheses and characterizations: Compound 1 was synthesized according to Scheme S-1. Scheme S-1 2 N N 5 i N 4 P Et Et iii N 6 ii P Et Et iv v, vi N N i) Fmoc-Su, DIPEA, Acetone;
Supporting Information Syntheses and characterizations: Compound 1 was synthesized according to Scheme S-1. Scheme S-1 2 N N 5 i N 4 P Et Et iii N 6 ii P Et Et iv v, vi N N i) Fmoc-Su, DIPEA, Acetone;
CH 395G Lecture 5 Fall 2009
 CH 395G Lecture 5 Fall 2009 Voet Biochemistry 3e Chapter 6 : Separation of Proteins We will discuss a liitle more re protein purification and proteomics and the Protein Center facilities at UT before moving
CH 395G Lecture 5 Fall 2009 Voet Biochemistry 3e Chapter 6 : Separation of Proteins We will discuss a liitle more re protein purification and proteomics and the Protein Center facilities at UT before moving
Novel Water-Soluble Near-Infrared Cyanine Dyes: Synthesis, Spectral Properties, and Use in the Preparation of Internally Quenched Fluorescent Probes
 ovel Water-Soluble ear-infrared Cyanine Dyes: Synthesis, Spectral Properties, and Use in the Preparation of Internally Quenched Fluorescent Probes Cédric Bouteiller,,, Guillaume Clavé,,, Aude Bernardin,,
ovel Water-Soluble ear-infrared Cyanine Dyes: Synthesis, Spectral Properties, and Use in the Preparation of Internally Quenched Fluorescent Probes Cédric Bouteiller,,, Guillaume Clavé,,, Aude Bernardin,,
Exam I Answer Key: Summer 2006, Semester C
 1. Which of the following tripeptides would migrate most rapidly towards the negative electrode if electrophoresis is carried out at ph 3.0? a. gly-gly-gly b. glu-glu-asp c. lys-glu-lys d. val-asn-lys
1. Which of the following tripeptides would migrate most rapidly towards the negative electrode if electrophoresis is carried out at ph 3.0? a. gly-gly-gly b. glu-glu-asp c. lys-glu-lys d. val-asn-lys
Peptides And Proteins
 Kevin Burgess, May 3, 2017 1 Peptides And Proteins from chapter(s) in the recommended text A. Introduction B. omenclature And Conventions by amide bonds. on the left, right. 2 -terminal C-terminal triglycine
Kevin Burgess, May 3, 2017 1 Peptides And Proteins from chapter(s) in the recommended text A. Introduction B. omenclature And Conventions by amide bonds. on the left, right. 2 -terminal C-terminal triglycine
SUPPORTING INFORMATION
 SUPPRTIG IFRMATI Salicylaldehyde Ester-Induced Chemoselective Peptide Ligations Enabling Generation of atural Peptidic Linkages at the Serine/Threonine Sites Xuechen Li,* iu Yung Lam, Yinfeng Zhang, Chun
SUPPRTIG IFRMATI Salicylaldehyde Ester-Induced Chemoselective Peptide Ligations Enabling Generation of atural Peptidic Linkages at the Serine/Threonine Sites Xuechen Li,* iu Yung Lam, Yinfeng Zhang, Chun
Biochemistry 3100 Sample Problems Chemical and Physical Methods
 (1) Describe the function of the following compounds in chemical sequencing, synthesis and modification: (a) Dithiothreitol (c) t-butyloxycarbonyl chloride (b) Dicyclohexyl carbodiimide (d) Cyanogen Bromide
(1) Describe the function of the following compounds in chemical sequencing, synthesis and modification: (a) Dithiothreitol (c) t-butyloxycarbonyl chloride (b) Dicyclohexyl carbodiimide (d) Cyanogen Bromide
SRM assay generation and data analysis in Skyline
 in Skyline Preparation 1. Download the example data from www.srmcourse.ch/eupa.html (3 raw files, 1 csv file, 1 sptxt file). 2. The number formats of your computer have to be set to English (United States).
in Skyline Preparation 1. Download the example data from www.srmcourse.ch/eupa.html (3 raw files, 1 csv file, 1 sptxt file). 2. The number formats of your computer have to be set to English (United States).
NMR study of complexes between low molecular mass inhibitors and the West Nile virus NS2B-NS3 protease
 University of Wollongong Research Online Faculty of Science - Papers (Archive) Faculty of Science, Medicine and Health 2009 NMR study of complexes between low molecular mass inhibitors and the West Nile
University of Wollongong Research Online Faculty of Science - Papers (Archive) Faculty of Science, Medicine and Health 2009 NMR study of complexes between low molecular mass inhibitors and the West Nile
Separation of Large and Small Peptides by Supercritical Fluid Chromatography and Detection by Mass Spectrometry
 Separation of Large and Small Peptides by Supercritical Fluid Chromatography and Detection by Mass Spectrometry Application Note Biologics and Biosimilars Author Edgar Naegele Agilent Technologies, Inc.
Separation of Large and Small Peptides by Supercritical Fluid Chromatography and Detection by Mass Spectrometry Application Note Biologics and Biosimilars Author Edgar Naegele Agilent Technologies, Inc.
Rapid and Sensitive Fluorescent Peptide Quantification Using LavaPep
 Rapid and Sensitive Fluorescent Peptide Quantification Using LavaPep Purpose To develop a fast, sensitive and robust fluorescent-based assay, to quantify peptides that is compatible with downstream proteomics
Rapid and Sensitive Fluorescent Peptide Quantification Using LavaPep Purpose To develop a fast, sensitive and robust fluorescent-based assay, to quantify peptides that is compatible with downstream proteomics
Tutorial 1: Setting up your Skyline document
 Tutorial 1: Setting up your Skyline document Caution! For using Skyline the number formats of your computer have to be set to English (United States). Open the Control Panel Clock, Language, and Region
Tutorial 1: Setting up your Skyline document Caution! For using Skyline the number formats of your computer have to be set to English (United States). Open the Control Panel Clock, Language, and Region
Supporting Information
 Supporting Information Wiley-VC 2005 69451 Weinheim, Germany Charge Interactions do the Job: A Combined Statistical and Combinatorial Approach to Find Artificial Receptors for Tetrapeptide Binding in Water.
Supporting Information Wiley-VC 2005 69451 Weinheim, Germany Charge Interactions do the Job: A Combined Statistical and Combinatorial Approach to Find Artificial Receptors for Tetrapeptide Binding in Water.
The Depsipeptide Methodology for Solid Phase Peptide Synthesis: Circumventing Side Reactions and Development of an Automated Technique via
 Supporting Information The Depsipeptide Methodology for Solid Phase Peptide Synthesis: Circumventing Side Reactions and Development of an Automated Technique via Depsidipeptide Units. Irene Coin, Rudolf
Supporting Information The Depsipeptide Methodology for Solid Phase Peptide Synthesis: Circumventing Side Reactions and Development of an Automated Technique via Depsidipeptide Units. Irene Coin, Rudolf
Supporting Information
 Electronic upplementary Material (EI) for ChemComm. This journal is The Royal ociety of Chemistry 2014 upporting Information Materials and methods: Chemicals: Fmoc-amino acids were obtained from GL Biochem
Electronic upplementary Material (EI) for ChemComm. This journal is The Royal ociety of Chemistry 2014 upporting Information Materials and methods: Chemicals: Fmoc-amino acids were obtained from GL Biochem
Lecture 8 (9/29/17) Lecture 8 (9/29/17)
 Reading: Ch3; 97-102 Lecture 8 (9/29/17) Problems: Ch3 (text); 18, 19, 20, 23 Ch3 (Study guide); 9 Ch4 (Study guide); 3 NEXT Reading: Ch4; 119-122, 125-126, 131-133 Ch4; 123-124, 130-131, 133, 137-138
Reading: Ch3; 97-102 Lecture 8 (9/29/17) Problems: Ch3 (text); 18, 19, 20, 23 Ch3 (Study guide); 9 Ch4 (Study guide); 3 NEXT Reading: Ch4; 119-122, 125-126, 131-133 Ch4; 123-124, 130-131, 133, 137-138
Solutions In each case, the chirality center has the R configuration
 CAPTER 25 669 Solutions 25.1. In each case, the chirality center has the R configuration. C C 2 2 C 3 C(C 3 ) 2 D-Alanine D-Valine 25.2. 2 2 S 2 d) 2 25.3. Pro,, Trp, Tyr, and is, Trp, Tyr, and is Arg,
CAPTER 25 669 Solutions 25.1. In each case, the chirality center has the R configuration. C C 2 2 C 3 C(C 3 ) 2 D-Alanine D-Valine 25.2. 2 2 S 2 d) 2 25.3. Pro,, Trp, Tyr, and is, Trp, Tyr, and is Arg,
Tracking down protein-protein interaction via FRET-system using site-specific thiol-labeling
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2018 Tracking down protein-protein interaction via FRET-system using site-specific
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2018 Tracking down protein-protein interaction via FRET-system using site-specific
Introduction to Proteomics: Fragmentation of protonated peptides and manual sequencing
 Introduction to Proteomics: ragmentation of protonated peptides and manual sequencing Árpád Somogyi CCIC SP SU Summer Workshop S/S hod using ESI Ion rap (Bottom up) 1 An alternative strategy for complex
Introduction to Proteomics: ragmentation of protonated peptides and manual sequencing Árpád Somogyi CCIC SP SU Summer Workshop S/S hod using ESI Ion rap (Bottom up) 1 An alternative strategy for complex
How proteins separate on reverse-phase HPLC
 1 Reverse Phase How proteins separate on reverse-phase HPLC RP chromatography separates proteins through the interaction of the hydrophobic foot of the protein with a nonpolar surface of the particle RP
1 Reverse Phase How proteins separate on reverse-phase HPLC RP chromatography separates proteins through the interaction of the hydrophobic foot of the protein with a nonpolar surface of the particle RP
Supporting Information
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 214 Supporting Information Rapid and sensitive detection of acrylic acid using a novel fluorescence
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 214 Supporting Information Rapid and sensitive detection of acrylic acid using a novel fluorescence
Breaking the speed limit: Fast, high-resolution peptide and tryptic digest separations using fused-core particles
 Breaking the speed limit: Fast, high-resolution peptide and tryptic digest separations using fused-core particles Stephanie Schuster 1, Barry Boyes 1,2, and Darryl Johnson 2 1 Advanced Materials Technology,
Breaking the speed limit: Fast, high-resolution peptide and tryptic digest separations using fused-core particles Stephanie Schuster 1, Barry Boyes 1,2, and Darryl Johnson 2 1 Advanced Materials Technology,
Biotin-guided fluorescent-peptide drug delivery system for cancer
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting Information For Biotin-guided fluorescent-peptide drug delivery system for cancer Taeyoung
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting Information For Biotin-guided fluorescent-peptide drug delivery system for cancer Taeyoung
Lecture 15: Realities of Genome Assembly Protein Sequencing
 Lecture 15: Realities of Genome Assembly Protein Sequencing Study Chapter 8.10-8.15 1 Euler s Theorems A graph is balanced if for every vertex the number of incoming edges equals to the number of outgoing
Lecture 15: Realities of Genome Assembly Protein Sequencing Study Chapter 8.10-8.15 1 Euler s Theorems A graph is balanced if for every vertex the number of incoming edges equals to the number of outgoing
Backbone modification of a parathyroid hormone receptor-1 antagonist/inverse agonist
 SUPPORTING INFORMATION Backbone modification of a parathyroid hormone receptor-1 antagonist/inverse agonist Ross W. Cheloha, Tomoyuki Watanabe, Thomas Dean, Samuel H. Gellman*, Thomas J. Gardella* *Corresponding
SUPPORTING INFORMATION Backbone modification of a parathyroid hormone receptor-1 antagonist/inverse agonist Ross W. Cheloha, Tomoyuki Watanabe, Thomas Dean, Samuel H. Gellman*, Thomas J. Gardella* *Corresponding
Collision Cross Section: Ideal elastic hard sphere collision:
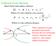 Collision Cross Section: Ideal elastic hard sphere collision: ( r r 1 ) Where is the collision cross-section r 1 r ) ( 1 Where is the collision distance r 1 r These equations negate potential interactions
Collision Cross Section: Ideal elastic hard sphere collision: ( r r 1 ) Where is the collision cross-section r 1 r ) ( 1 Where is the collision distance r 1 r These equations negate potential interactions
C a h p a t p e t r e r 6 E z n y z m y e m s
 Chapter 6 Enzymes 4. Examples of enzymatic reactions acid-base catalysis: give and take protons covalent catalysis: a transient covalent bond is formed between the enzyme and the substrate metal ion catalysis:
Chapter 6 Enzymes 4. Examples of enzymatic reactions acid-base catalysis: give and take protons covalent catalysis: a transient covalent bond is formed between the enzyme and the substrate metal ion catalysis:
SUPPORTING INFORMATION Antimicrobial activity of chlorinated amino acids and peptides
 SUPPORTING INFORMATION Antimicrobial activity of chlorinated amino acids and peptides Melanie S. A. Coker*, Wan-Ping Hu, Senti T Senthilmohan and Anthony J. Kettle Free Radical Research Group, University
SUPPORTING INFORMATION Antimicrobial activity of chlorinated amino acids and peptides Melanie S. A. Coker*, Wan-Ping Hu, Senti T Senthilmohan and Anthony J. Kettle Free Radical Research Group, University
Rapid Distinction of Leucine and Isoleucine in Monoclonal Antibodies Using Nanoflow. LCMS n. Discovery Attribute Sciences
 Rapid Distinction of Leucine and Isoleucine in Monoclonal Antibodies Using Nanoflow LCMS n Dhanashri Bagal *, Eddie Kast, Ping Cao Discovery Attribute Sciences Amgen, South San Francisco, California, United
Rapid Distinction of Leucine and Isoleucine in Monoclonal Antibodies Using Nanoflow LCMS n Dhanashri Bagal *, Eddie Kast, Ping Cao Discovery Attribute Sciences Amgen, South San Francisco, California, United
Diastereomeric resolution directed towards chirality. determination focussing on gas-phase energetics of coordinated. sodium dissociation
 Supplementary Information Diastereomeric resolution directed towards chirality determination focussing on gas-phase energetics of coordinated sodium dissociation Authors: samu Kanie*, Yuki Shioiri, Koji
Supplementary Information Diastereomeric resolution directed towards chirality determination focussing on gas-phase energetics of coordinated sodium dissociation Authors: samu Kanie*, Yuki Shioiri, Koji
1. Draw the structure of oxazolone formed upon activation of N-Acetylvaline
 Peptide quiz 1 (5 questions for 10 minutes) 10 points max 1. Draw the structure of oxazolone formed upon activation of -Acetylvaline 3 C 3 C 2. The following DKP (1) is prepared by cyclisation of dipeptide
Peptide quiz 1 (5 questions for 10 minutes) 10 points max 1. Draw the structure of oxazolone formed upon activation of -Acetylvaline 3 C 3 C 2. The following DKP (1) is prepared by cyclisation of dipeptide
Research Article Synthesis of Hemopressin Peptides by Classical Solution Phase Fragment Condensation
 Peptides Volume 2012, Article ID 186034, 5 pages doi:10.1155/2012/186034 Research Article Synthesis of Hemopressin Peptides by Classical Solution Phase Fragment Condensation P. Anantha Reddy, Sean T. Jones,
Peptides Volume 2012, Article ID 186034, 5 pages doi:10.1155/2012/186034 Research Article Synthesis of Hemopressin Peptides by Classical Solution Phase Fragment Condensation P. Anantha Reddy, Sean T. Jones,
-1- HO H O H. β-d-galactopyranose (A) OMe CH 3 I. MeO. Ag 2 O. O Me HNO 3. OAc (CH 3 CO) 2 O. AcO. pyridine. O Ac. NaBH 4 H 2 O. MeOH. dry HCl.
 -1-1. Draw structures for the products you would expect to obtain from reaction of β-dgalactopyranose () with each of the reagents below. Be sure to include all relevant stereochemistry. (40 pts). β-d-galactopyranose
-1-1. Draw structures for the products you would expect to obtain from reaction of β-dgalactopyranose () with each of the reagents below. Be sure to include all relevant stereochemistry. (40 pts). β-d-galactopyranose
Improved 6- Plex TMT Quantification Throughput Using a Linear Ion Trap HCD MS 3 Scan Jane M. Liu, 1,2 * Michael J. Sweredoski, 2 Sonja Hess 2 *
 Improved 6- Plex TMT Quantification Throughput Using a Linear Ion Trap HCD MS 3 Scan Jane M. Liu, 1,2 * Michael J. Sweredoski, 2 Sonja Hess 2 * 1 Department of Chemistry, Pomona College, Claremont, California
Improved 6- Plex TMT Quantification Throughput Using a Linear Ion Trap HCD MS 3 Scan Jane M. Liu, 1,2 * Michael J. Sweredoski, 2 Sonja Hess 2 * 1 Department of Chemistry, Pomona College, Claremont, California
(d, J = 7.0 Hz, 2H), 1.80 (m, 2H), 1.65 (m, 2H), 1.49 (m, 2H), 1.46 (s, 9H) ppm; 13 C NMR
 Site-Specific Protein Immobilization by Staudinger Ligation Matthew B. Soellner, Kimberly A. Dickson, Bradley L. ilsson, and Ronald T. Raines Departments of Chemistry and Biochemistry, University of Wisconsin,
Site-Specific Protein Immobilization by Staudinger Ligation Matthew B. Soellner, Kimberly A. Dickson, Bradley L. ilsson, and Ronald T. Raines Departments of Chemistry and Biochemistry, University of Wisconsin,
Computational Methods for Mass Spectrometry Proteomics
 Computational Methods for Mass Spectrometry Proteomics Eidhammer, Ingvar ISBN-13: 9780470512975 Table of Contents Preface. Acknowledgements. 1 Protein, Proteome, and Proteomics. 1.1 Primary goals for studying
Computational Methods for Mass Spectrometry Proteomics Eidhammer, Ingvar ISBN-13: 9780470512975 Table of Contents Preface. Acknowledgements. 1 Protein, Proteome, and Proteomics. 1.1 Primary goals for studying
A Plausible Model Correlates Prebiotic Peptide Synthesis with. Primordial Genetic Code
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 A Plausible Model Correlates Prebiotic Peptide Synthesis with Primordial Genetic Code Jianxi Ying,
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 A Plausible Model Correlates Prebiotic Peptide Synthesis with Primordial Genetic Code Jianxi Ying,
A High Sensitivity Dual Solid Phase Extraction LC-MS/MS Assay for the Determination of the Therapeutic Peptide Desmopressin in Human Plasma
 White Paper A High Sensitivity Dual Solid Phase Extraction LC-MS/MS Assay for the Determination of the Therapeutic Peptide Desmopressin in Human Plasma Lars Neudert, MSc, Senior Scientist Method Development
White Paper A High Sensitivity Dual Solid Phase Extraction LC-MS/MS Assay for the Determination of the Therapeutic Peptide Desmopressin in Human Plasma Lars Neudert, MSc, Senior Scientist Method Development
Identification of Human Hemoglobin Protein Variants Using Electrospray Ionization-Electron Transfer Dissociation Mass Spectrometry
 Identification of Human Hemoglobin Protein Variants Using Electrospray Ionization-Electron Transfer Dissociation Mass Spectrometry Jonathan Williams Waters Corporation, Milford, MA, USA A P P L I C AT
Identification of Human Hemoglobin Protein Variants Using Electrospray Ionization-Electron Transfer Dissociation Mass Spectrometry Jonathan Williams Waters Corporation, Milford, MA, USA A P P L I C AT
New Approaches in the Analysis of Amadori Compounds
 7 TH Wartburg Symposium on Flavor Chemistry and Biology Eisenach, April 21 st - April 23 rd, 2004 New Approaches in the Analysis of Amadori Compounds T. Davidek, I. Blank, K. Kraehenhuehl, J. Hau and S.
7 TH Wartburg Symposium on Flavor Chemistry and Biology Eisenach, April 21 st - April 23 rd, 2004 New Approaches in the Analysis of Amadori Compounds T. Davidek, I. Blank, K. Kraehenhuehl, J. Hau and S.
Rapid Microwave-Assisted CNBr Cleavage of Bead-Bound Peptides
 Rapid Microwave-Assisted CNBr Cleavage of Bead-Bound Peptides Su Seong Lee 1,*, Jaehong Lim 1, Junhoe Cha 1, Sylvia Tan 1, James R. Heath 1,2, * 1 Institute of Bioengineering and Nanotechnology, 31 Biopolis
Rapid Microwave-Assisted CNBr Cleavage of Bead-Bound Peptides Su Seong Lee 1,*, Jaehong Lim 1, Junhoe Cha 1, Sylvia Tan 1, James R. Heath 1,2, * 1 Institute of Bioengineering and Nanotechnology, 31 Biopolis
Synthesis of Peptide-Grafted Comb Polypeptides via Polymerisation of NCA-Peptides
 Supporting Information to Synthesis of Peptide-Grafted Comb Polypeptides via Polymerisation of NCA-Peptides Hiroshi Enomoto, Benjamin Nottelet, Soultan Al Halifa, Christine Enjalbal, Mathieu Dupré, Julien
Supporting Information to Synthesis of Peptide-Grafted Comb Polypeptides via Polymerisation of NCA-Peptides Hiroshi Enomoto, Benjamin Nottelet, Soultan Al Halifa, Christine Enjalbal, Mathieu Dupré, Julien
EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I Dr. Glen B Legge
 EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I 2009 Dr. Glen B Legge This is a Scantron exam. All answers should be transferred to the Scantron sheet using a #2 pencil. Write and bubble
EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I 2009 Dr. Glen B Legge This is a Scantron exam. All answers should be transferred to the Scantron sheet using a #2 pencil. Write and bubble
MS/MS of Peptides Manual Sequencing of Protonated Peptides
 S/S of Peptides anual Sequencing of Protonated Peptides Árpád Somogyi Associate irector CCIC, ass Spectrometry and Proteomics Laboratory SU July 11, 2018 Peptides Product Ion Scan Product ion spectra contain
S/S of Peptides anual Sequencing of Protonated Peptides Árpád Somogyi Associate irector CCIC, ass Spectrometry and Proteomics Laboratory SU July 11, 2018 Peptides Product Ion Scan Product ion spectra contain
Advanced Fragmentation Techniques for BioPharma Characterization
 Advanced Fragmentation Techniques for BioPharma Characterization Global BioPharma Summit The world leader in serving science Different modes of fragmentation to answer different questions or for different
Advanced Fragmentation Techniques for BioPharma Characterization Global BioPharma Summit The world leader in serving science Different modes of fragmentation to answer different questions or for different
Downloaded from:
 Withers-Martinez, C; Suarez, C; Fulle, S; Kher, S; Penzo, M; Ebejer, JP; Koussis, K; ackett, F; Jirgensons, A; Finn, P; Blackman, MJ (2012) Plasmodium subtilisin-like protease 1 (SUB1): insights into the
Withers-Martinez, C; Suarez, C; Fulle, S; Kher, S; Penzo, M; Ebejer, JP; Koussis, K; ackett, F; Jirgensons, A; Finn, P; Blackman, MJ (2012) Plasmodium subtilisin-like protease 1 (SUB1): insights into the
Electronic Supplementary Information. Zhen Zheng, Gongyu Li, Chengfan Wu, Miaomiao Zhang, Yue Zhao, and Gaolin Liang*
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Intracellular synthesis of D-aminoluciferin for bioluminescence
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Intracellular synthesis of D-aminoluciferin for bioluminescence
Full-length GlpG sequence was generated by PCR from E. coli genomic DNA. (with two sequence variations, D51E/L52V, from the gene bank entry aac28166),
 Supplementary Methods Protein expression and purification Full-length GlpG sequence was generated by PCR from E. coli genomic DNA (with two sequence variations, D51E/L52V, from the gene bank entry aac28166),
Supplementary Methods Protein expression and purification Full-length GlpG sequence was generated by PCR from E. coli genomic DNA (with two sequence variations, D51E/L52V, from the gene bank entry aac28166),
available 3,5-dihydroxybenzoic acid was reduced to afford 3,5-diketohexahydrobenzoic acid in
 Supporting Information Chemical Synthesis of DAz-1. DAz-1 was synthesized as shown in Supporting Information Figure 1. Commercially available 3,5-dihydroxybenzoic acid was reduced to afford 3,5-diketohexahydrobenzoic
Supporting Information Chemical Synthesis of DAz-1. DAz-1 was synthesized as shown in Supporting Information Figure 1. Commercially available 3,5-dihydroxybenzoic acid was reduced to afford 3,5-diketohexahydrobenzoic
SUPPORTING INFORMATION. Reversible and versatile on-tether modification of chiral. content
 UPPRTIG IFRMATI Reversible and versatile on-tether modification of chiral center-induced helical peptides Kuan u,,# Chengjie un,,# Zigang Li #,* # chool of Chemical Biology and Biotechnology, Peking University
UPPRTIG IFRMATI Reversible and versatile on-tether modification of chiral center-induced helical peptides Kuan u,,# Chengjie un,,# Zigang Li #,* # chool of Chemical Biology and Biotechnology, Peking University
Applying MRM Spectrum Mode and Library Searching for Enhanced Reporting Confidence in Routine Pesticide Residue Analysis
 PO-CON1768E Applying MRM Spectrum Mode and Library Searching for Enhanced Reporting Confidence in Routine Pesticide ASMS 2017 TP-194 David Baker 1, Christopher Titman 1, Neil Loftus 1, Jonathan Horner
PO-CON1768E Applying MRM Spectrum Mode and Library Searching for Enhanced Reporting Confidence in Routine Pesticide ASMS 2017 TP-194 David Baker 1, Christopher Titman 1, Neil Loftus 1, Jonathan Horner
Simplified platensimycin analogues as antibacterial agents
 Simplified platensimycin analogues as antibacterial agents Dragan Krsta, a Caron Ka, a Ian T. Crosby, a Ben Capuano a and David T. Manallack a * a Medicinal Chemistry and Drug Action, Monash Institute
Simplified platensimycin analogues as antibacterial agents Dragan Krsta, a Caron Ka, a Ian T. Crosby, a Ben Capuano a and David T. Manallack a * a Medicinal Chemistry and Drug Action, Monash Institute
Supporting Information
 Supporting Information Synthesis of the natural product Marthiapeptide A Yuqi Zhang 1, Md. Amirul Islam 1 and Shelli R. McAlpine 1* 1 School of Chemistry, University of New South Wales, Sydney, NSW 2052
Supporting Information Synthesis of the natural product Marthiapeptide A Yuqi Zhang 1, Md. Amirul Islam 1 and Shelli R. McAlpine 1* 1 School of Chemistry, University of New South Wales, Sydney, NSW 2052
Protein Quantitation II: Multiple Reaction Monitoring. Kelly Ruggles New York University
 Protein Quantitation II: Multiple Reaction Monitoring Kelly Ruggles kelly@fenyolab.org New York University Traditional Affinity-based proteomics Use antibodies to quantify proteins Western Blot RPPA Immunohistochemistry
Protein Quantitation II: Multiple Reaction Monitoring Kelly Ruggles kelly@fenyolab.org New York University Traditional Affinity-based proteomics Use antibodies to quantify proteins Western Blot RPPA Immunohistochemistry
Electronic Supplementary Information
 Electronic Supplementary Information A Sensitive Phosphorescent Thiol Chemosensor Based on an Iridium(III) Complex with α,β-unsaturated Ketone Functionalized 2,2 -Bipyridyl Ligand Na Zhao, a Yu-Hui Wu,
Electronic Supplementary Information A Sensitive Phosphorescent Thiol Chemosensor Based on an Iridium(III) Complex with α,β-unsaturated Ketone Functionalized 2,2 -Bipyridyl Ligand Na Zhao, a Yu-Hui Wu,
Supporting Information
 Supporting Information Experimental Section Azido Acids General: Triflyl anhydride and Cu II S pentahydrate 99.999% were obtained from Aldrich. All other solvents and chemicals were reagent grade or better
Supporting Information Experimental Section Azido Acids General: Triflyl anhydride and Cu II S pentahydrate 99.999% were obtained from Aldrich. All other solvents and chemicals were reagent grade or better
Quantitative Proteomics
 Quantitative Proteomics Quantitation AND Mass Spectrometry Condition A Condition B Identify and quantify differently expressed proteins resulting from a change in the environment (stimulus, disease) Lyse
Quantitative Proteomics Quantitation AND Mass Spectrometry Condition A Condition B Identify and quantify differently expressed proteins resulting from a change in the environment (stimulus, disease) Lyse
C H E M I S T R Y N A T I O N A L Q U A L I F Y I N G E X A M I N A T I O N SOLUTIONS GUIDE
 C H E M I S T R Y 2 0 0 0 A T I A L Q U A L I F Y I G E X A M I A T I SLUTIS GUIDE Answers are a guide only and do not represent a preferred method of solving problems. Section A 1B, 2A, 3C, 4C, 5D, 6D,
C H E M I S T R Y 2 0 0 0 A T I A L Q U A L I F Y I G E X A M I A T I SLUTIS GUIDE Answers are a guide only and do not represent a preferred method of solving problems. Section A 1B, 2A, 3C, 4C, 5D, 6D,
Supporting Information
 Supporting Information A synthetic strategy for conjugation of paromomycin to cell-penetrating Tat(48-60) for delivery and visualization into Leishmania parasites Sira Defaus 1, Maria Gallo 1, María A.
Supporting Information A synthetic strategy for conjugation of paromomycin to cell-penetrating Tat(48-60) for delivery and visualization into Leishmania parasites Sira Defaus 1, Maria Gallo 1, María A.
Reagents. Affinity Tag (Biotin) Acid Cleavage Site. Figure 1. Cleavable ICAT Reagent Structure.
 DATA SHEET Protein Expression Analysis Reagents Background The ultimate goal of proteomics is to identify and quantify proteins that are relevant to a given biological state; and to unearth networks of
DATA SHEET Protein Expression Analysis Reagents Background The ultimate goal of proteomics is to identify and quantify proteins that are relevant to a given biological state; and to unearth networks of
1-N-(3,4,6-tri-O-acetyl-2-deoxy-2-N-acetyl-β-D-glucopyranosylamide)-4-(N -methylidenyl -2 - bromoacetamido)-4,5-anhydro-triazole (5) AcO AcO
 upporting Information -(propargyl)-bromacetamide (4) ac 3, 2 2 Propargylamine (0.1 ml, 1.45 mmol) was dissolved in water (15.0 ml) and was treated with bromoacetic anhydride (1.85 g, 7.25 mmol) in the
upporting Information -(propargyl)-bromacetamide (4) ac 3, 2 2 Propargylamine (0.1 ml, 1.45 mmol) was dissolved in water (15.0 ml) and was treated with bromoacetic anhydride (1.85 g, 7.25 mmol) in the
Applications of Mass Spectrometry for Biotherapeutic Characterization
 Applications of Mass Spectrometry for Biotherapeutic Characterization Case Studies of Disulfide Characterization and Separation free Modes of Analysis Steven L. Cockrill Amgen Colorado Analytical Sciences
Applications of Mass Spectrometry for Biotherapeutic Characterization Case Studies of Disulfide Characterization and Separation free Modes of Analysis Steven L. Cockrill Amgen Colorado Analytical Sciences
Biochemistry Quiz Review 1I. 1. Of the 20 standard amino acids, only is not optically active. The reason is that its side chain.
 Biochemistry Quiz Review 1I A general note: Short answer questions are just that, short. Writing a paragraph filled with every term you can remember from class won t improve your answer just answer clearly,
Biochemistry Quiz Review 1I A general note: Short answer questions are just that, short. Writing a paragraph filled with every term you can remember from class won t improve your answer just answer clearly,
Table S1. Overview of used PDZK1 constructs and their binding affinities to peptides. Related to figure 1.
 Table S1. Overview of used PDZK1 constructs and their binding affinities to peptides. Related to figure 1. PDZK1 constru cts Amino acids MW [kda] KD [μm] PEPT2-CT- FITC KD [μm] NHE3-CT- FITC KD [μm] PDZK1-CT-
Table S1. Overview of used PDZK1 constructs and their binding affinities to peptides. Related to figure 1. PDZK1 constru cts Amino acids MW [kda] KD [μm] PEPT2-CT- FITC KD [μm] NHE3-CT- FITC KD [μm] PDZK1-CT-
Bacterial protease uses distinct thermodynamic signatures for substrate recognition
 Bacterial protease uses distinct thermodynamic signatures for substrate recognition Gustavo Arruda Bezerra, Yuko Ohara-Nemoto, Irina Cornaciu, Sofiya Fedosyuk, Guillaume Hoffmann, Adam Round, José A. Márquez,
Bacterial protease uses distinct thermodynamic signatures for substrate recognition Gustavo Arruda Bezerra, Yuko Ohara-Nemoto, Irina Cornaciu, Sofiya Fedosyuk, Guillaume Hoffmann, Adam Round, José A. Márquez,
Studies Leading to the Development of a Highly Selective. Colorimetric and Fluorescent Chemosensor for Lysine
 Supporting Information for Studies Leading to the Development of a Highly Selective Colorimetric and Fluorescent Chemosensor for Lysine Ying Zhou, a Jiyeon Won, c Jin Yong Lee, c * and Juyoung Yoon a,
Supporting Information for Studies Leading to the Development of a Highly Selective Colorimetric and Fluorescent Chemosensor for Lysine Ying Zhou, a Jiyeon Won, c Jin Yong Lee, c * and Juyoung Yoon a,
PC235: 2008 Lecture 5: Quantitation. Arnold Falick
 PC235: 2008 Lecture 5: Quantitation Arnold Falick falickam@berkeley.edu Summary What you will learn from this lecture: There are many methods to perform quantitation using mass spectrometry (any method
PC235: 2008 Lecture 5: Quantitation Arnold Falick falickam@berkeley.edu Summary What you will learn from this lecture: There are many methods to perform quantitation using mass spectrometry (any method
Supplementary information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2016 Supplementary information 1. Composition of the nanoparticles 2. Dynamic light scattering 3. Scanning
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2016 Supplementary information 1. Composition of the nanoparticles 2. Dynamic light scattering 3. Scanning
1. Amino Acids and Peptides Structures and Properties
 1. Amino Acids and Peptides Structures and Properties Chemical nature of amino acids The!-amino acids in peptides and proteins (excluding proline) consist of a carboxylic acid ( COOH) and an amino ( NH
1. Amino Acids and Peptides Structures and Properties Chemical nature of amino acids The!-amino acids in peptides and proteins (excluding proline) consist of a carboxylic acid ( COOH) and an amino ( NH
Supporting Information For:
 Supporting Information For: Peptidic α-ketocarboxylic Acids and Sulfonamides as Inhibitors of Protein Tyrosine Phosphatases Yen Ting Chen, Jian Xie, and Christopher T. Seto* Department of Chemistry, Brown
Supporting Information For: Peptidic α-ketocarboxylic Acids and Sulfonamides as Inhibitors of Protein Tyrosine Phosphatases Yen Ting Chen, Jian Xie, and Christopher T. Seto* Department of Chemistry, Brown
TANDEM MASS SPECTROSCOPY
 TANDEM MASS SPECTROSCOPY 1 MASS SPECTROMETER TYPES OF MASS SPECTROMETER PRINCIPLE TANDEM MASS SPECTROMETER INSTRUMENTATION QUADRAPOLE MASS ANALYZER TRIPLE QUADRAPOLE MASS ANALYZER TIME OF FLIGHT MASS ANALYSER
TANDEM MASS SPECTROSCOPY 1 MASS SPECTROMETER TYPES OF MASS SPECTROMETER PRINCIPLE TANDEM MASS SPECTROMETER INSTRUMENTATION QUADRAPOLE MASS ANALYZER TRIPLE QUADRAPOLE MASS ANALYZER TIME OF FLIGHT MASS ANALYSER
Total Synthesis of Cyclosporine: Access to N-Methylated Peptides via Isonitrile Coupling Reactions
 Total Synthesis of Cyclosporine: Access to -thylated Peptides via Isonitrile Coupling Reactions Xiangyang Wu, Jennifer L. Stockdill, Ping Wang, Samuel J. Danishefsky* J. Am. Chem. Soc. 2010,132, 4098-4100
Total Synthesis of Cyclosporine: Access to -thylated Peptides via Isonitrile Coupling Reactions Xiangyang Wu, Jennifer L. Stockdill, Ping Wang, Samuel J. Danishefsky* J. Am. Chem. Soc. 2010,132, 4098-4100
SERVA ICPL Kit (Cat.-No )
 INSTRUCTION MANUAL SERVA ICPL Kit (Cat.-No. 39230.01) SERVA Electrophoresis GmbH Carl-Benz-Str. 7 D-69115 Heidelberg Phone +49-6221-138400, Fax +49-6221-1384010 e-mail: info@serva.de http://www.serva.de
INSTRUCTION MANUAL SERVA ICPL Kit (Cat.-No. 39230.01) SERVA Electrophoresis GmbH Carl-Benz-Str. 7 D-69115 Heidelberg Phone +49-6221-138400, Fax +49-6221-1384010 e-mail: info@serva.de http://www.serva.de
BENG 183 Trey Ideker. Protein Sequencing
 BENG 183 Trey Ideker Protein Sequencing The following slides borrowed from Hong Li s Biochemistry Course: www.sb.fsu.edu/~hongli/4053notes Introduction to Proteins Proteins are of vital importance to biological
BENG 183 Trey Ideker Protein Sequencing The following slides borrowed from Hong Li s Biochemistry Course: www.sb.fsu.edu/~hongli/4053notes Introduction to Proteins Proteins are of vital importance to biological
MS-based proteomics to investigate proteins and their modifications
 MS-based proteomics to investigate proteins and their modifications Francis Impens VIB Proteomics Core October th 217 Overview Mass spectrometry-based proteomics: general workflow Identification of protein
MS-based proteomics to investigate proteins and their modifications Francis Impens VIB Proteomics Core October th 217 Overview Mass spectrometry-based proteomics: general workflow Identification of protein
Supporting Information
 Supporting Information Responsive Prodrug Self-Assembled Vesicles for Targeted Chemotherapy in Combination with Intracellular Imaging Hongzhong Chen, Huijun Phoebe Tham,, Chung Yen Ang, Qiuyu Qu, Lingzhi
Supporting Information Responsive Prodrug Self-Assembled Vesicles for Targeted Chemotherapy in Combination with Intracellular Imaging Hongzhong Chen, Huijun Phoebe Tham,, Chung Yen Ang, Qiuyu Qu, Lingzhi
Structure and evolution of the spliceosomal peptidyl-prolyl cistrans isomerase Cwc27
 Acta Cryst. (2014). D70, doi:10.1107/s1399004714021695 Supporting information Volume 70 (2014) Supporting information for article: Structure and evolution of the spliceosomal peptidyl-prolyl cistrans isomerase
Acta Cryst. (2014). D70, doi:10.1107/s1399004714021695 Supporting information Volume 70 (2014) Supporting information for article: Structure and evolution of the spliceosomal peptidyl-prolyl cistrans isomerase
Total chemical synthesis of a thermostable enzyme capable of polymerase chain reaction
 Supplementary Information Total chemical synthesis of a thermostable enzyme capable of polymerase chain reaction Weiliang Xu 1,2,3, Wenjun Jiang 1,3, Jiaxing Wang 2,3, Linping Yu 1,3, Ji Chen 1, Xianyu
Supplementary Information Total chemical synthesis of a thermostable enzyme capable of polymerase chain reaction Weiliang Xu 1,2,3, Wenjun Jiang 1,3, Jiaxing Wang 2,3, Linping Yu 1,3, Ji Chen 1, Xianyu
D-Leu-L-Phe-containing dipeptide inhibitors of α-chymotrypsin the role of the N- and C-termini in enzyme affinity
 General Papers ARKIVC 2008 (xii) 85-94 D-Leu-L-Phe-containing dipeptide inhibitors of α-chymotrypsin the role of the - and C-termini in enzyme affinity David Pearson* and Andrew D. Abell Department of
General Papers ARKIVC 2008 (xii) 85-94 D-Leu-L-Phe-containing dipeptide inhibitors of α-chymotrypsin the role of the - and C-termini in enzyme affinity David Pearson* and Andrew D. Abell Department of
Dental Biochemistry Exam The total number of unique tripeptides that can be produced using all of the common 20 amino acids is
 Exam Questions for Dental Biochemistry Monday August 27, 2007 E.J. Miller 1. The compound shown below is CH 3 -CH 2 OH A. acetoacetate B. acetic acid C. acetaldehyde D. produced by reduction of acetaldehyde
Exam Questions for Dental Biochemistry Monday August 27, 2007 E.J. Miller 1. The compound shown below is CH 3 -CH 2 OH A. acetoacetate B. acetic acid C. acetaldehyde D. produced by reduction of acetaldehyde
Mass Spectrometry. Hyphenated Techniques GC-MS LC-MS and MS-MS
 Mass Spectrometry Hyphenated Techniques GC-MS LC-MS and MS-MS Reasons for Using Chromatography with MS Mixture analysis by MS alone is difficult Fragmentation from ionization (EI or CI) Fragments from
Mass Spectrometry Hyphenated Techniques GC-MS LC-MS and MS-MS Reasons for Using Chromatography with MS Mixture analysis by MS alone is difficult Fragmentation from ionization (EI or CI) Fragments from
Electron Transfer Dissociation of N-linked Glycopeptides from a Recombinant mab Using SYNAPT G2-S HDMS
 Electron Transfer Dissociation of N-linked Glycopeptides from a Recombinant mab Using SYNAPT G2-S HDMS Jonathan P. Williams, Jeffery M. Brown, Stephane Houel, Ying Qing Yu, and Weibin Chen Waters Corporation,
Electron Transfer Dissociation of N-linked Glycopeptides from a Recombinant mab Using SYNAPT G2-S HDMS Jonathan P. Williams, Jeffery M. Brown, Stephane Houel, Ying Qing Yu, and Weibin Chen Waters Corporation,
Tandem mass spectra were extracted from the Xcalibur data system format. (.RAW) and charge state assignment was performed using in house software
 Supplementary Methods Software Interpretation of Tandem mass spectra Tandem mass spectra were extracted from the Xcalibur data system format (.RAW) and charge state assignment was performed using in house
Supplementary Methods Software Interpretation of Tandem mass spectra Tandem mass spectra were extracted from the Xcalibur data system format (.RAW) and charge state assignment was performed using in house
ml. ph 7.5 ph 6.5 ph 5.5 ph 4.5. β 2 AR-Gs complex + GDP β 2 AR-Gs complex + GTPγS
 a UV28 absorption (mau) 9 8 7 5 3 β 2 AR-Gs complex β 2 AR-Gs complex + GDP β 2 AR-Gs complex + GTPγS β 2 AR-Gs complex dissociated complex excess nucleotides b 9 8 7 5 3 β 2 AR-Gs complex β 2 AR-Gs complex
a UV28 absorption (mau) 9 8 7 5 3 β 2 AR-Gs complex β 2 AR-Gs complex + GDP β 2 AR-Gs complex + GTPγS β 2 AR-Gs complex dissociated complex excess nucleotides b 9 8 7 5 3 β 2 AR-Gs complex β 2 AR-Gs complex
Amino Acid Side Chain Induced Selectivity in the Hydrolysis of Peptides Catalyzed by a Zr(IV)-Substituted Wells-Dawson Type Polyoxometalate
 Amino Acid Side Chain Induced Selectivity in the Hydrolysis of Peptides Catalyzed by a Zr(IV)-Substituted Wells-Dawson Type Polyoxometalate Stef Vanhaecht, Gregory Absillis, Tatjana N. Parac-Vogt* Department
Amino Acid Side Chain Induced Selectivity in the Hydrolysis of Peptides Catalyzed by a Zr(IV)-Substituted Wells-Dawson Type Polyoxometalate Stef Vanhaecht, Gregory Absillis, Tatjana N. Parac-Vogt* Department
Discussion Section (Day, Time):
 Chemistry 27 Spring 2005 Exam 1 Chemistry 27 Professor Gavin MacBeath arvard University Spring 2005 our Exam 1 Friday, February 25, 2005 11:07 AM 12:00 PM Discussion Section (Day, Time): TF: Directions:
Chemistry 27 Spring 2005 Exam 1 Chemistry 27 Professor Gavin MacBeath arvard University Spring 2005 our Exam 1 Friday, February 25, 2005 11:07 AM 12:00 PM Discussion Section (Day, Time): TF: Directions:
Hemoglobin (Hb) exists in the blood cells of
 Prediction of Product Ion Isotope Ratios in the Tandem Electrospray Ionization Mass Spectra from the Second Isotope of Tryptic Peptides: Identification of the Variant 131 Gln Glu, Hemoglobin Camden Brian
Prediction of Product Ion Isotope Ratios in the Tandem Electrospray Ionization Mass Spectra from the Second Isotope of Tryptic Peptides: Identification of the Variant 131 Gln Glu, Hemoglobin Camden Brian
Serine-7 but not serine-5 phosphorylation primes RNA polymerase II CTD for P-TEFb recognition
 Supplementary Information to Serine-7 but not serine-5 phosphorylation primes RNA polymerase II CTD for P-TEFb recognition Nadine Czudnochowski 1,2, *, Christian A. Bösken 1, * & Matthias Geyer 1 1 Max-Planck-Institut
Supplementary Information to Serine-7 but not serine-5 phosphorylation primes RNA polymerase II CTD for P-TEFb recognition Nadine Czudnochowski 1,2, *, Christian A. Bösken 1, * & Matthias Geyer 1 1 Max-Planck-Institut
Ionization Methods in Mass Spectrometry at the SCS Mass Spectrometry Laboratory
 Ionization Methods in Mass Spectrometry at the SCS Mass Spectrometry Laboratory Steven L. Mullen, Ph.D. Associate Director SCS Mass Spectrometry Laboratory Contact Information 31 oyes Laboratory (8:00-5:00
Ionization Methods in Mass Spectrometry at the SCS Mass Spectrometry Laboratory Steven L. Mullen, Ph.D. Associate Director SCS Mass Spectrometry Laboratory Contact Information 31 oyes Laboratory (8:00-5:00
BCH 4053 Exam I Review Spring 2017
 BCH 4053 SI - Spring 2017 Reed BCH 4053 Exam I Review Spring 2017 Chapter 1 1. Calculate G for the reaction A + A P + Q. Assume the following equilibrium concentrations: [A] = 20mM, [Q] = [P] = 40fM. Assume
BCH 4053 SI - Spring 2017 Reed BCH 4053 Exam I Review Spring 2017 Chapter 1 1. Calculate G for the reaction A + A P + Q. Assume the following equilibrium concentrations: [A] = 20mM, [Q] = [P] = 40fM. Assume
Protein Quantitation II: Multiple Reaction Monitoring. Kelly Ruggles New York University
 Protein Quantitation II: Multiple Reaction Monitoring Kelly Ruggles kelly@fenyolab.org New York University Traditional Affinity-based proteomics Use antibodies to quantify proteins Western Blot Immunohistochemistry
Protein Quantitation II: Multiple Reaction Monitoring Kelly Ruggles kelly@fenyolab.org New York University Traditional Affinity-based proteomics Use antibodies to quantify proteins Western Blot Immunohistochemistry
Supplementary Information. Affinity Selection and sequence-activity relationships of HIV-1 membrane fusion inhibitors
 Supplementary Information Affinity Selection and sequence-activity relationships of HIV-1 membrane fusion inhibitors directed at the drug-resistant variants Shinya Oishi,* a Kentaro Watanabe, a Saori Ito,
Supplementary Information Affinity Selection and sequence-activity relationships of HIV-1 membrane fusion inhibitors directed at the drug-resistant variants Shinya Oishi,* a Kentaro Watanabe, a Saori Ito,
Quantitation of High Resolution MS Data Using UNIFI: Acquiring and Processing Full Scan or Tof-MRM (Targeted HRMS) Datasets for Quantitative Assays
 : Acquiring and Processing Full Scan or Tof-MRM (Targeted HRMS) Datasets for Quantitative Assays Mark Wrona, Jayne Kirk, and Yun Alelyunas Waters Corporation, Milford, MA, USA APPLICATION BENEFITS Ability
: Acquiring and Processing Full Scan or Tof-MRM (Targeted HRMS) Datasets for Quantitative Assays Mark Wrona, Jayne Kirk, and Yun Alelyunas Waters Corporation, Milford, MA, USA APPLICATION BENEFITS Ability
