Modification of aerosol mass and size distribution due to aqueous-phase SO 2 oxidation in clouds: Comparisons of several models
|
|
|
- Laurence Murphy
- 5 years ago
- Views:
Transcription
1 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 108, NO. D7, 4213, doi: /2002jd002697, 2003 Modification of aerosol mass and size distribution due to aqueous-phase SO 2 oxidation in clouds: Comparisons of several models Sonia M. Kreidenweis, 1 Chris J. Walcek, 2 Graham Feingold, 3 Wanmin Gong, 4 Mark Z. Jacobson, 5 Cheol-Hee Kim, 6 Xiaohong Liu, 7 Joyce E. Penner, 7 Athanasios Nenes, 8,9 and John H. Seinfeld 8 Received 25 June 2002; revised 20 September 2002; accepted 27 November 2002; published 8 April [1] Models of aerosol scavenging and aqueous-phase oxidation of SO 2 by H 2 O 2 and O 3 in a cloud updraft are compared. Bulk models considering only a single droplet size are compared with size-resolved models that explicitly simulate multiple aerosol and drop sizes. All models simulate growth of cloud drops on a lognormal ammonium bisulfate aerosol distribution, and subsequent aqueous-phase chemistry during adiabatic ascent. In agreement with earlier published studies, it is found that relative to bulk models, the sizeresolved cloud chemical models consistently calculate 2 3 times more oxidation via the SO 2 +O 3 pathway, due to calculated variability of cloud water ph among cloud drops. All models calculate high scavenging of the input dry aerosol mass, but the calculated number of cloud drops formed varies from drops cm 3. Differences in the calculated number of cloud drops formed result from the treatment of gaseous species uptake, solution thermodynamics, applied water condensation mass accommodation coefficient, and bin size range definitions over which the input aerosol distribution is numerically approximated. The difference in calculated cloud drop number can under many conditions propagate to appreciable variations in cloud albedo. It is found that the modifications to the aerosol size and mass spectrum are sensitive to the number of cloud drops formed, and differences in the processed aerosol spectra were found to induce up to 13% differences in calculated light extinction properties of the modified particle distributions. These significant discrepancies among cloud aerosol chemistry interaction models, even when used to simulate relatively simple conditions, suggest that parameterizations of these processes used in larger-scale cloud, regional and longer-term climate models can contain high levels of uncertainty. INDEX TERMS: 0305 Atmospheric Composition and Structure: Aerosols and particles (0345, 4801); 0320 Atmospheric Composition and Structure: Cloud physics and chemistry; 0317 Atmospheric Composition and Structure: Chemical kinetic and photochemical properties; 0365 Atmospheric Composition and Structure: Troposphere composition and chemistry; 0368 Atmospheric Composition and Structure: Troposphere constituent transport and chemistry; KEYWORDS: aerosol size distribution, aqueous chemistry, sulfur oxidation, cloud chemistry Citation: Kreidenweis, S. M., C. J. Walcek, G. Feingold, W. Gong, M. Z. Jacobson, C.-H. Kim, X. Liu, J. E. Penner, A. Nenes, and J. H. Seinfeld, Modification of aerosol mass and size distribution due to aqueous-phase SO 2 oxidation in clouds: Comparisons of several models, J. Geophys. Res., 108(D7), 4213, doi: /2002jd002697, Introduction [2] Atmospheric particles containing sulfate are a dominant component of acidic deposition, and also contribute to fine particle pollution associated with regional haze [Malm et al., 1994] and health problems [Health Effects Institute, 2000]. Also, sulfate aerosol pollution induces an appreciable radiative cooling impact on global climate that 1 Department of Atmospheric Science, Colorado State University, Fort Collins, Colorado, USA. 2 Atmospheric Science Research Center, State University of New York, Albany, New York, USA. 3 National Oceanographic and Atmospheric Administration, Boulder, Colorado, USA. 4 Meteorological Service of Canada, Downsview, Ontario, Canada. Copyright 2003 by the American Geophysical Union /03/2002JD002697$ Department of Civil and Environmental Engineering, Stanford University, Palo Alto, California, USA. 6 National Institute of Environmental Research (NIER), Seo-gu, Incheon, Korea. 7 Atmospheric, Oceanic and Space Sciences, University of Michigan, Ann Arbor, Michigan, USA. 8 Division of Chemistry and Chemical Engineering, California Institute of Technology, Pasadena, California, USA. 9 Now at School of Earth and Atmospheric Sciences, Georgia Institute of Technology, Atlanta, Georgia, USA. AAC 4-1
2 AAC 4-2 KREIDENWEIS ET AL.: CLOUD CHEMISTRY COMPARISON counters the potential CO 2 -induced warming influences, but considerable uncertainties persist about quantifying the direct and indirect radiative effects of sulfate aerosols on climate [Intergovernmental Panel on Climate Change, 2001]. [3] Sulfate is a significant and frequently the dominant component of aerosol mass in the atmosphere, and is produced primarily from the oxidation of SO 2. This oxidation occurs in the gas phase through reaction with the hydroxyl radical, and the characteristic rate of the gas-phase reaction is on the order of a few percent per hour during the day [Stockwell et al., 1990]. In cloudy environments containing sufficient oxidants, SO 2 is rapidly oxidized by aqueous reactions with hydrogen peroxide, ozone, organic peroxides and a number of other oxidants. It is generally recognized that the dominant mechanism of sulfate formation in the troposphere is via reactions of SO 2 with H 2 O 2 and O 3 in clouds [Walcek and Taylor, 1986; Barth et al., 2000; Rasch et al., 2000]. Therefore it is important for atmospheric models that include sulfate to simulate accurately the oxidation of SO 2 in clouds. Most larger-scale models of atmospheric sulfur simulate in-cloud SO 2 oxidation using a simplified bulk modeling approach, where oxidation rates are calculated using a single characteristic water content, droplet size and acidity level [e.g., Chang et al., 1987; Iribarne and Cho, 1989]. [4] The aqueous-phase addition of mass to particles has direct and indirect radiative effects. Because of the global importance of sulfur gases and sulfate aerosol, this species has been the focus of the most study in the literature. The direct radiative effects are sensitive to the details of the initial aerosol distribution and how mass is added to the aerosol spectrum [e.g., Lelieveld and Heintzenberg, 1992; Hegg et al., 1996]. In turn, aqueous chemistry depends on the cloud drop spectrum. Indirect radiative effects arise from variations in drop spectra induced by changes in the input aerosol, and subsequent changes in cloud albedo. The concept of susceptibility of warm clouds to such albedo changes was first proposed by Twomey [1991], and later studies focused specifically on the link between sulfate aerosols, cloud condensation nuclei, and cloud droplet spectra in indirect radiative effects [e.g., Hegg, 1994; Jones et al., 1994; Boucher and Lohmann, 1995]. [5] Here several bulk and size-resolved aerosol/cloud/ chemistry models are compared under a well-defined set of conditions in order to assess the uncertainties associated with different modeling approaches. The results presented here summarize the findings of the aerosol parcel model component of the World Meteorological Organization 5th International Cloud Modeling Workshop held in Glenwood Springs, Colorado, United States, in August The problem statement was first designed and disseminated in early 2000, and discussions and revisions of results continued before and after the August 2000 workshop. 2. Models Compared [6] Seven participating modeling groups submitted twelve simulations for comparison. Table 1 identifies the models divided into simpler bulk models and more sophisticated, size-resolved models. The five groups contributing size-resolved calculations also submitted bulk versions of Table 1. Summary of Participating Models Name (Institution) Bulk Model Size-Resolved (Number Sizes) References Feingold (NOAA/ETL) x 20 Feingold et al. [1998] Gong (MSC) x 20 Gong [2002] Jacobson (Stanford) x 50 Jacobson [2002] Liu (Michigan) x 30 Liu and Seidl [1998] Nenes (CalTech) x 60 Nenes et al. [2001] Kim (CSU) x Kim et al. [2002] Walcek (SUNY) x Walcek and Taylor [1986] their models, and two groups contributed only bulk calculations. In subsequent figures, each model is identified using the first letter of the name identified on Table 1, and model results are further identified as bulk or sizeresolved. Many of the cloud chemistry models used here are extracted from larger-scale atmospheric models, and the references to related applications are listed in the right column of Table 1. [7] Further characteristics of the size-resolved models are summarized in Table 2. All of the models employ a socalled discrete, Lagrangian or moving bin approach. Although the models represent the aerosol and drop spectra as moving (in size space), discrete points, we will use the term bin here when discussing these points. All of the size-resolved models also solve gas-liquid transfer simultaneously with aqueous chemistry, and all treat this transfer as a nonequilibrium process. [8] All models used the identical simulation conditions presented in the next section, and differences between the models arise from different numerical integration methods, different aerosol and droplet size resolutions used by the size-resolved models, and different methods for assessing cloud drop activation and water activity. In addition, the transfer of soluble HNO 3 and NH 3 gases before and during droplet growth influences the growth calculations for the size-resolved models. 3. Comparison Conditions [9] All models calculate the aqueous chemical composition of cloud water within a cloud updraft. Dissolved constituents in cloud water result from the scavenging of a specified lognormal dry aerosol size distribution and dissolution of gases, and concentrations are modified by the oxidation of SO 2 by H 2 O 2 and O 3. The physical conditions simulated are listed in the top half of Table 3. The chemical composition of cloud water is simulated for an air parcel lifted adiabatically at 0.5 m s 1 starting from a height slightly below cloud base. Cloud base is reached 196 s into the simulation, and simulations continue for an additional 2400 s (1.2 km) above cloud base. At the end of the simulation, the cloud water is evaporated and the resulting dry aerosol distributions are compared. [10] The relatively unpolluted chemical conditions chosen for the simulation are listed in the bottom portion of Table 3. The initial ammonium and sulfate concentrations represent a slightly acidic ammonium bisulfate aerosol. The initial ammonia gas concentration is present in excess with respect to equilibrium with this aerosol composition, which can have consequences for aerosol-cloud interactions [Seidl, 1989], as
3 KREIDENWEIS ET AL.: CLOUD CHEMISTRY COMPARISON AAC 4-3 Table 2. Characteristics of Size-Resolving Models Feingold Gong Jacobson Liu Nenes moving discrete moving discrete moving discrete Bin structure moving discrete Lagrangian, remapped to initial grid gear-type (VODE) gear-type (SMVGEAR II) McRae solver (semi-implicit method) Numerical solver gear-type (VODE) Young-Boris hybrid predictor- corrector Maximum time step 2 s 0.2 s 900 s (not reached) 0.2 s 0.02 s Number of size bins Start of aqueous chemistry Conc < 2 M at activation prior to activation prior to activation prior to activation NH3 and HNO3 are taken up before activation, but not re-equilibrated before start of run uptake of gases before activation allowed; initial re-equilibration with gas-phase NH 3 performed NH3 and HNO3 taken up before activation, but not re-equilibrated before start none; activation is parameterized according to critical radius for input distribution Influence of gas uptake on activation none; initial composition is assumed Köhler equations used, computing n from number of ions, but setting osmotic coefficient to 1 Köhler equations used, computing n from ion number, osmotic coefficient set to 1 not computed ZSR for aerosol; for drops, Köhler equations used, computing n from number of ions, but setting osmotic coefficient to 1 Aerosol liquid water content ZSR, using binary data from known ammoniated sulfate solutions, for both aerosols and drops Bin activation entire bin activated at once Partial bin activation allowed Entire bin activated at once Entire bin activated at once Entire bin activated at once N/A Water condensation accommodation coefficient Droplet number concentration at cloud base (cm 3, initial T and P) discussed in section 5. Initial H 2 O 2 and O 3 concentrations are well in excess of SO 2 available for oxidation. [11] The initial number of particles per cm 3 (N) is defined as a function of particle diameter D, using a lognormal, monomodal dry aerosol size distribution where dn dðln DÞ ¼ N T pffiffiffiffiffi 2p ln sg " exp lnðdþ D 2 # g 2 : ð1þ 2lns g The median diameter D g = 0.08 mm, the geometric standard deviation s g = 2, and the total aerosol number concentration N T = 566 cm 3. A dry particle density of 1.8 g cm 3 is assumed. Input mass and number concentrations expressed in absolute concentration units (mg m 3, particles cm 3 )are defined at the initial (t = 0) temperature and pressure listed in Table 3. The assumption of a monomodal, singlecomposition aerosol is made in order to deal with as simple a system as possible when diagnosing differences among models. Many previous studies have been initialized with more realistic bimodal and externally mixed aerosol distributions and have found that model predictions vary with the relative sizes and compositions of the two modes [e.g., Ayers and Larson, 1990; Hegg et al., 1992; Yuen et al., 1994; Hoppel et al., 1994; Hegg et al., 1996]. [12] All models use the identical aqueous-phase equilibria listed in Table 4. Oxidation of SO 2 by O 3 and H 2 O 2 in the cloud water is specified using the ph-dependent reaction formulations and temperature dependencies shown in Table 5. The ozone reaction varies inversely with approximately the square of the H + concentration, while the H 2 O 2 reaction is largely insensitive to ph. 4. Sulfur Chemistry [13] Figure 1 shows the general meteorological and chemical conditions of the parcel as it rises. Figure 1a shows a nearly linear increase in condensed liquid water content above cloud base. All models calculate that 75 80% of the initial SO 2 is oxidized to sulfate during the 40 min Table 3. Cloud Conditions Factor Value (Units) Physical Conditions Height above surface at time = (m) Temperature at t = (K) Relative humidity at t = 0 95 (%) Pressure at t = (mbar) Cloud base height above surface 698 (m) Cloud base pressure 939 (mbar) Cloud base temperature (K) Air density at cloud base 1.15 (kg m 3 ) Updraft velocity 0.5 (m s 1 ) Cloud water mixing ratio 1200 m 2.17 (g kg 1 ) (2400 s) above cloud base Chemical Conditions SO 2 at t = (ppt-v) NH 3 (g) at t = (ppt-v) H 2 O 2 at t = (ppt-v) HNO 3 at t = (ppt-v) O 3 at t = 0 50 (ppb-v) CO 2 at t = (ppm-v) SO = 4 (particulate) at t = 0 2 (mg m 3 ) NH + 4 (particulate) at t = (mg m 3 )
4 AAC 4-4 KREIDENWEIS ET AL.: CLOUD CHEMISTRY COMPARISON Table 4. Aqueous-Phase Equilibria Equilibrium a K 298 H/R b Accommodation Coefficient O 3 (g) () O 3 (aq) H 2 O 2 (g) () H 2 O 2 (aq) HNO 3 (g) () HNO 3 (aq) c 0.05 CO 2 (g) () CO 2 (aq) SO 2 (g) () SO 2 (aq) NH 3 (g) () NH 3 (aq) H 2 O 2 (aq) () HO 2 +H CO 2 (aq) () HCO 3 +H HCO 3 () CO 2 3 +H SO 2 (aq) () HSO 3 +H HSO 3 () SO 2 3 +H NH 3 (aq) () NH + 4 +OH HNO 3 (aq) () NO 3 +H c a 1 Units of K are moles liter (water) atm 1 1 or moles liter (water). b Equilibrium for other temperatures (T) calculated as K(T) = K 298 exp [ H/R(1/T 1/298)]. c Value for HNO 3 (g) () NO 3 +H +. simulation (Figure 1b). In Figures 1a and 1b, the solid black and the gray lines denote the maximum and minimum of the range of quantities calculated by the 12 models. [14] Figure 1c shows the calculated water-mass-weighted average cloud water ph as a function of distance above cloud base according to the models. Again, the two curves in Figure 1c denote the range of the 12 models calculations. Due to the low water content near cloud base, and the acidic nature of the aerosols, cloud water ph starts off very low, less than ph 3.6, but rapidly increases as the acidic material is diluted with increasing amounts of condensing water. Although ammonia gas is absorbed during the initial stages of cloud formation, HNO 3 is also absorbed, so that the observed increase in ph is due primarily to the effects of dilution. All models calculate that greater than 97% of the initial dry aerosol mass is incorporated into the cloud water. [15] Figure 2 plots the calculated total sulfate production as a function of the liquid-water-weighted ph 1.2 km above cloud base, showing approximately 20% range in the calculated sulfate production, and a corresponding deviation in the calculated acidity [H + ] in the cloud water. The thick gray line in Figure 2 shows the slope of relationship between ph and sulfate assuming H + is linearly related to sulfate concentration in the cloud water. This figure clearly shows that the size-resolved models disagree significantly from the bulk models, and calculate about 17% more sulfate production, leading to higher acidity and thus slightly lower cloud-water-averaged phs. The seven bulk models agree with each other to ±4%, slightly larger than the discrepancies of the five size resolved models (±3%). [16] Figure 3 provides a further breakdown of the calculated SO 2 oxidation showing the sulfate produced by O 3 and via H 2 O 2, plotted against each other. Here there is a distinct clustering of most of the size-resolved models showing about 45 parts per trillion (ppt) more (a factor of 2 3 more) sulfate formation via the ph-dependent O 3 pathway relative to the bulk models, which consistently calculate about 30 ppt sulfate production via O 3 oxidation. This extra sulfate production from ozone by the size-resolved models is somewhat countered by a decrease in sulfate formation from H 2 O 2, which is about ppt less in the size resolved models relative to the bulk models. Generally there is an inverse relationship between the magnitude of the H 2 O 2 versus O 3 oxidation: As more sulfate is produced via the O 3 reaction, less SO 2 is available for the H 2 O 2 reaction, and sulfate production from H 2 O 2 decreases. Therefore the size-resolved models calculate less sulfate production via the H 2 O 2 pathway, even though this reaction rate is largely independent of ph. [17] Figures 2 and 3 demonstrate that there is a systematic and significant difference between the calculations of aqueous-phase SO 2 oxidation by bulk and size-resolved models of cloud water chemistry, as has been shown previously [e.g., Hegg and Larson, 1990; Gurciullo and Pandis, 1997; Ogren and Charlson, 1992]. This results primarily from the strong ph dependence of the aqueous-phase reaction between SO 2 and O 3 in cloud water, and the ph differences that can exist among numerous drop categories in a cloud. Any differences in ph among cloud droplets will lead to different average rates of the reaction between SO 2 and O 3 in clouds. [18] Figure 4 shows examples of the calculated distribution of ph among the cloud water mass 1.2 km above cloud base according to three of the size-resolved aqueous chemistry models. In this figure, the ph of each liquid water fraction is plotted cumulatively, and the liquid-waterweighted average ph is shown below each model label. In all models, cloud drops forming on smaller aerosols are calculated to have a higher ph, and the largest activated aerosols grow into the most acidic cloud drops. Figure 4 shows that the Gong and Liu models have a small fraction of their water mass (10% 20%) with the highest ph (5.2), despite calculating a more acidic average ph. In contrast, the less acidic Jacobson model contains no water with ph >5.2. Since the ozone oxidation rate is approximately inversely proportional to the square of the H + concentration in cloud water, a change in 1 ph point represents a 100- fold change in the reaction rate of SO 2 and O 3 in cloud water. A doubling of the SO 2 +O 3 oxidation rate requires only a 0.3 ph point increase. The Gong and Liu sizeresolved models calculate appreciably greater SO 2 oxidation than the Jacobson model, primarily because appreciable fractions of the cloud water have a higher ph than the highest ph calculated by Jacobson s model. Despite these Table 5. Aqueous-Phase Reactions Reaction Rate Expression a k 298 E/R O 3 +SO 2 (aq) ) S(VI) b k 298 exp[ E/R(1/T 1/298)] O 3 + HSO 3 ) S(VI) b k 298 exp[ E/R(1/T 1/298)] O 3 +SO 2 3 ) S(VI) b k 298 exp[ E/R(1/T 1/298)] H 2 O 2 (aq) + HSO 3 ) S(VI) b k298½hþšexp½ E=Rð1=T 1=298ÞŠ ½ Š a Units of k are liter (water) moles 1 s 1. b S(VI)= SO HSO 4. 1þ13 H þ
5 KREIDENWEIS ET AL.: CLOUD CHEMISTRY COMPARISON AAC 4-5 Figure 1. Vertical variation of various meteorological and chemical factors in cloud updraft according to models considered in this study. (a) Condensed liquid water mixing ratio. (b) Total gas + aqueous SO 2 concentration. (c) Average cloud water ph. Simulation starts 196 s (0.1 km) below cloud base and continues for 2400 s (1.2 km) above cloud base. Grey and black lines denote lower and upper bounds of values calculated by models use in this study. considerable ranges and differences, the average ph calculated by the size-resolved models agrees to within 0.03 ph units (Figure 1). 5. Cloud Drop Number Concentrations and Modification of Aerosol Size Distributions [19] The number concentration of cloud droplets computed in each of the five size-resolved models is shown in the last row of Table 2, from 275 cm 3 for Jacobson s model to 358 cm 3 for Liu s model. This variation can be traced to a number of assumptions made in the models. Although, as shown in Table 2, different numerical solvers are used in the models, we did not find any appreciable differences in results that could be traced to the solvers, but found that other factors were important. First, while the Feingold, Jacobson, Liu, and Nenes models explicitly treat the growth of droplets by water condensation and calculate the time history of ambient water saturation ratio, the Gong model uses a parameterization as implemented in the regional model AURAMS [Gong, 2002]. This parameterization is based on a simple empirical relation between the number density of cloud condensation nuclei (and thus cloud drops) and that of the ambient atmospheric aerosols [Jones et al., 1994]. The largest particles are assumed to activate first; the smallest size of activated particle is determined by counting downward from the large end of the aerosol spectrum until the specified total number of CCN is accumulated. The adiabatic liquid water content, computed from thermodynamic considerations alone, is distributed evenly among the activated particles to determine drop size, resulting in a relatively narrow cloud droplet spectrum. Differences in drop size arise only at the large end of the drop spectrum, and originate from the chosen step size for updating LWC (60 s for this study). Despite the simplicity of this treatment, the number of drops computed in the Gong model is similar to that computed by the other models. [20] Differences between the Feingold, Jacobson, Liu and Nenes models derive from a number of factors. The water condensation mass accommodation coefficient (a) used in each model is shown in Table 2, and varies from to 1. The higher the applied value of a, the more effective is condensational growth at scavenging water vapor that would otherwise go toward activation of more droplets. The corresponding variation in droplet number concentration (N D ) is shown in Figure 5, where Feingold, Liu and Nenes varied the value of a from 1 to 0.042, resulting in 12 35% increases in N D. The relationship between droplet number concentration (N D ) and maximum supersaturation for the Feingold, Jacobson, Liu and Nenes models, using the base-case values of a assumed in each as indicated on the plot, is shown in Figure 6. From this figure it is apparent that differences in treatment of water condensation cannot alone account for the differences in predicted drop number concentrations, since there should be a positive, monotonic
6 AAC 4-6 KREIDENWEIS ET AL.: CLOUD CHEMISTRY COMPARISON Figure 2. Calculated net sulfate formation plotted against cloud water ph after parcel lifted 1.2 km above cloud base according to all models. Filled squares denote bulk aqueous chemistry models. Open circles denote size-resolved aerosol/cloud water models. Gray line denotes the slope of the ph-sulfate relationship assuming H + is proportional to total sulfate. Letters next to each point refer to the first letter of the contributing model shown in Table 1. Fine stippled lines connect models of the same authors. relationship between S max and N D with decreasing a. Those models using a = 1 (Nenes and Liu) did compute the lowest maximum supersaturation (S max ), but predicted higher number concentrations than the other models. The reasons for these differences are twofold: First, the droplet solution thermodynamics is treated differently among these models, and second, each model has a different bin structure. This affects the calculated N D because none of these models treats activation of partial bins. [21] None of the models adjusted chemical reactions (except gas-liquid equilibria, and reversible liquid-liquid equilibria in the Jacobson model) for nonideal conditions under high ionic strengths. The Feingold model does not allow for transfer of gases or chemical reaction until droplets are sufficiently dilute (ionic strength <2 M). Thus the water content of drops before activation is based primarily on the initial aerosol composition. During all phases of aerosol and drop growth, the water vapor pressure over the drop is computed from a Köhler-type equation, but without the dilute approximation. Drop size is solved for iteratively, using water activity as a function of composition from the ZSR relationship, and using binary ammoniated sulfate solution data from Tang and Munkelwitz [1994]. The Jacobson, Liu, and Nenes models use the Köhler equations with the dilute approximation to compute droplet size, with the number of ions, n, set according to the evolving composition of the drops. These latter three models all permit gas transfer to the aqueous phase from the start of the simulation, so the aerosol in general contains more soluble material than the initial conditions suggest and is therefore easier to activate, tending to produce higher N D. This explanation was confirmed by Nenes, who found drop number concentrations of 290 cm 3 when uptake of gases was ignored, compared with 301 cm 3 when gas transfer prior to and during activation was included. In addition, the Liu model re-equilibrates the aerosol composition with the specified initial ammonia gas concentration, so that this case has essentially a different input aerosol spectrum that leads to significantly larger N D. This interpretation was borne out by a sensitivity study performed by Jacobson [2002], wherein he re-ran his model with the same initial reequilibration assumption as used by Liu and Seidl [1998] and computed 321 drops cm 3, compared with 275 drops cm 3 in the model run without this assumption. [22] The grids that are used by the various size resolving models also play a role in determining the number concentration of droplets. Figure 7 shows the superposition of initial dry aerosol size distribution, approximate critical supersaturation for this spectrum, and bin structure for the five sizeresolved models (arbitrary y axis). The range of S max predicted by the models is indicated by the shaded region, and corresponds to particles with diameters from about mm. Since the maximum supersaturations fall near the peak of the aerosol size distribution, it is clear that small deviations in predicted S max will translate into relatively large changes in N D. Cumulative number concentrations (computed from the large end of the spectrum) vary from 375 to 235 cm 3 between these diameters; the models have 2 4 bins across this range. Thus, depending on the Figure 3. Cumulative sulfate formation via H 2 O 2 versus sulfate formation via O 3 at 1.2 km above cloud base. Filled squares denote bulk aqueous chemistry models. Open circles denote size-resolved aerosol/cloud water models. Letters next to each point refer to the first letter of the contributing model shown in Table 1. Fine stippled lines connect models of the same authors.
7 KREIDENWEIS ET AL.: CLOUD CHEMISTRY COMPARISON AAC 4-7 Figure 5. Dependence of computed drop number concentration on water condensation mass accommodation coefficient. Figure 4. The ph of various fractions of the liquid water mass in cloud updraft according to three size-resolved models (listed in Table 1) 1.2 km above cloud base. Waterweighted average ph is noted under each model name. model size resolution, if the predicted S max falls just inside or just outside one of the bins, a difference of 30 or more droplets cm 3 can be computed, since these models treat the contents of each bin as represented by a single size particle. The effects of resolution on predicted N D are explored further in Figure 8. In most (but not all) cases, N D decreased when resolution was increased, suggesting that S max was falling just inside a relatively large bin in the base cases. However, it is difficult to generalize the relationship between N D and size resolution, since it depends on the maximum supersaturation, the aerosol size distribution, and the setup of the grid in diameter space. [23] Figures 9a 9e show the modifications to the dry sulfate aerosol mass distribution calculated by the five sizeresolved models. The cloud drops 1.2 km above cloud base are evaporated, and the resulting dry aerosol size distributions are compared with the initial input spectrum. Within the cloud updraft, dry aerosols are activated in the lowest several tens of meters above cloud base, and all models activate from the largest aerosols down to a cutoff size determined by the simulated maximum in-cloud supersaturation. Only aerosols larger than the cutoff size are incorporated into the cloud water and therefore only these larger particles have the potential to accumulate more sulfate due to aqueous-phase chemistry. For the conditions considered here, Figure 2 shows that the size-resolved models calculated that SO 2 oxidation increased total sulfate aerosol mass by ppt from the initial 2 mg m 3 (520 ppt), which represents a 33 35% increase in sulfate mass. Figure 6. Relationship between maximum supersaturation predicted by the models and droplet number concentration. The value of water condensation mass accommodation coefficient is shown next to the data points.
8 AAC 4-8 KREIDENWEIS ET AL.: CLOUD CHEMISTRY COMPARISON Figure 7. Superposition of initial dry aerosol number distribution, estimated critical supersaturation (Sc) as a function of particle size, and grid structure of the five size-resolved models, shown as bin endpoints. The shaded area indicates the size range spanned by the upper and lower estimates of critical supersaturation, according to whether n =2orn = 3 is assumed in the Köhler equation. The heavy horizontal lines show the largest and smallest maximum supersaturations computed by the five models, and define the width of the shaded area. [24] In Figure 9, aerosol size distributions are plotted on logarithmic scales (top) and linear scales (bottom) for each model, since each plotting method shows different pertinent details of the changes to the initial spectrum. Figures 9a 9e are sorted in order of increasing number of cloud drops formed at cloud base. All models calculate essentially negligible changes to the larger portion of the aerosol spectra, since the relatively small amount of added sulfate mass does not appreciably modify the diameter of the larger particles. Particles that are smaller than the smallest particle activated are also largely unmodified by cloud chemical processes, since either chemistry in unactivated particles is neglected, or only extremely small amounts of aqueous-phase sulfate formation are calculated within the small size bins. [25] Particles equal to and larger than the smallest particle activated show the largest increases in particle size due to the mass addition, creating a minimum in the processed aerosol spectrum [Hoppel et al., 1990; Yuen et al., 1994; Hoppel et al., 1994]. For the updraft speed, humidity conditions, aerosol composition, and initial size distribution used in this study, the smallest dry diameter activated ranges from mm, in agreement with the range of maximum supersaturations shown in Figure 7. The minimum is less pronounced in the Gong model, because in that model the grown particles are re-mapped onto the original grid, creating some loss of resolution, whereas the other modelers report the grown sizes for the moving discrete points and a modified grid is constructed from that infor- Figure 8. Variation of drop number concentration with number of sizes ( bins ). Applied values of water condensation mass accommodation coefficient are shown alongside each name.
9 KREIDENWEIS ET AL.: CLOUD CHEMISTRY COMPARISON AAC 4-9 Figure 9. Modification of the dry aerosol sulfate mass distribution according to the five size-resolved models listed in Table 1. Distributions plotted on a log (top) and linear (bottom) scales. Gray curve shows initial input aerosol mass spectrum. Number of cloud drops formed is shown in the lower linear panel of each figure. mation. The Jacobson and Gong models, forming the fewest number of cloud drops, show significant aerosol mass depletion near aerosol diameters mm, and increases in the mass concentration distribution in the size range from mm diameter. In contrast, Liu s model, forming the greatest number of cloud drops (358 cm 3 ), induces appreciable mass concentration depletions down to 0.06 mm diameter, and aerosol mass increases primarily in the mm diameter range. 6. Potential Radiative Effects [26] In this study, the largest discrepancies among the sizeresolved models are the 25 30% differences in the calculated
10 AAC 4-10 KREIDENWEIS ET AL.: CLOUD CHEMISTRY COMPARISON number of cloud drops formed. These differences will induce appreciable microphysical and radiative impacts [e.g., Feingold and Kreidenweis, 2000; Hegg et al., 1996]. Here we crudely estimate the potential direct and indirect radiative impacts. We emphasize that although the relationships between variations in processed aerosol spectra, cloud drop number concentrations, and radiative properties have been explored in many previous studies, some of which are cited earlier, our focus in this work is to examine how differences arising from models treating an identical, simple case propagate into uncertainties in estimates of such relationships Indirect Radiative Effects [27] Indirect radiative impacts of aerosols refer to the influence of aerosols on the radiative properties of clouds forming on aerosols. To examine the potential impact of model differences on indirect aerosol forcing, we consider the simple Twomey [1991] model as summarized by Seinfeld and Pandis [1998]. This model proposes that cloud optical depth is proportional to cloud depth, cloud liquid water content to the 2/3 power, and cloud drop number concentration to the 1/3 power. Therefore the range in cloud droplet number calculated in this study ( drops cm 3 ) translates into approximately a 9% range [(358/275) 1/3 1] in calculated cloud optical depth. [28] Twomey s [1991] definition of the susceptibility, dr c / dn, at constant liquid water content, shows that the cloud albedo, R c, for optically thin clouds is most sensitive to changes in drop number concentration when cloud albedo is 0.5. Under these conditions the approximation R c ln(n) is valid. Using this expression, the 25 30% spread in the calculated droplet number translates into an albedo change of 0.02 [= 0.075ln(358/275)], which is a significant uncertainty in the influence of clouds on the atmospheric energy budget in cloudy areas Direct Radiative Effects [29] Direct radiative impacts of aerosols refer to the scattering and absorption of light as it passes through an aerosolladen atmosphere. Here we examine changes in the light scattering properties of the aerosol distributions shown in Figure 9 caused by the addition of sulfate mass by calculating radiative extinction efficiencies from the size distributions. [30] For this extinction calculation, sulfate is assumed to be associated with ammonium bisulfate aerosol having a refractive index of 1.5, no absorption, and a dry particle density r of 1.8 g cm 3. The extinction efficiencies computed for each size distribution are normalized by the mass concentration of ammonium bisulfate aerosol. The single particle light extinction efficiency was calculated for a representative visible wavelength (530 nm) using a Mie theory model of light absorption and scattering by spherical particles [Bohren and Huffman, 1983]. Integrations are performed by numerically summing particle diameter bins of the size-resolved models, assuming each bin is represented by its midpoint diameter. [31] Figure 10 shows the changes to the extinction efficiency induced by the addition of sulfate mass to the initial distribution by in-cloud SO 2 chemistry. Extinction efficiencies are calculated for dry particles, and for those same particles under high humidity conditions (RH = 90%), where the particles absorb water and swell in size and mass Figure 10. Change in aerosol radiative extinction efficiencies under dry and humidified conditions due to the addition of sulfate mass to the initial lognormal aerosol. Letters along horizontal axis refers to first initial of model name identified in Table 1. Scattering calculation uses the size distributions shown in Figure 9. LogNrml model assumes additional mass on lognormal distribution. relative to the dry particles. Recall that all models calculate 34% sulfate mass increases from 520 ppt to ppt. However, since the models calculate different number concentrations of droplets, the particle diameters experiencing the most significant growth by mass addition change from model to model. [32] Under dry conditions, the initial dry lognormal aerosol distribution is calculated to have an extinction efficiency E =3.23m 2 g 1. According to all of the sizeresolving models, the addition of sulfate mass under dry conditions decreases the normalized extinction efficiency. This effect results from the fact that all models add sulfate mass to aerosols in the mm diameter range, which is smaller than the aerosol diameter that most efficiently scatters visible radiation (0.7 mm diameter). The magnitude of this decrease ranges from less than 5% to about 13% relative to the initial lognormal distribution. [33] Under humid conditions, a wet-to-dry particle diameter ratio of 1.8 is assumed, a reasonable approximation for ammonium bisulfate aerosol at 90% RH [Tang and Munkelwitz, 1994]. The initial and final size distributions are
11 KREIDENWEIS ET AL.: CLOUD CHEMISTRY COMPARISON AAC 4-11 shifted by this factor, and the aerosol mass is increased by the corresponding amount of water required to increase the diameter by 1.8. The computed extinction efficiencies are again normalized by the dry ammonium bisulfate mass. The initial humidified lognormal size distribution now has E = 20.5 m 2 g 1, and all of the modified size distributions show 1 11% increases over this value of E. Under higher humidity conditions, the peak in the sulfate mass distribution is shifted into more optically active diameter ranges. [34] In Figure 10 one additional simplified calculation (labeled LogNrml ) is included. In this case, the additional sulfate mass is uniformly added to the initial distribution in a manner that preserves the lognormal shape of the mass distribution. Such simplifications are often employed in larger-scale models where an aerosol size distribution shape is assumed. The extinction efficiencies computed under this assumption increase under both dry and humid conditions, since mass is added to all particle sizes including those particles that are in the size range of high extinction efficiency. Clearly the assumption of a fixed aerosol size distribution shape limits the ability to accurately characterize radiative impacts associated with changes in mass of the aerosol distribution; in this study, even the sign of the changes in the extinction properties could not be reproduced. This dependence of the scattering efficiency of secondary sulfate on the initial aerosol spectrum, and on the mechanism producing the sulfate, has been pointed out by a number of authors [e.g., Lelieveld and Heintzenberg, 1992; Hegg et al., 1993; Tang, 1996; Hegg et al., 1996]. Here, we have shown that model-to-model variations for a simple case study, for which all models predicted similar total sulfate formation, can also contribute to variations in computed scattering efficiencies. 7. Conclusions [35] Larger-scale atmospheric chemistry models that include the formation of sulfate usually use a simplified bulk approach to account for aqueous-phase SO 2 oxidation. Results for the simple case simulated here indicate a 15 25% difference in the calculated sulfate formation between bulk and size resolving models. These findings are in agreement with prior measurement and modeling work that showed that there is considerable uncertainty introduced in quantifying the oxidation of SO 2 by ozone, induced by ph variations among different cloud drops [Collett et al., 1994; Reilly et al., 2001; Pandis et al., 1990; Gurciullo and Pandis, 1997; Yuen et al., 1996; Zhang et al., 1999]. This study extends this analysis to explore differences among modeling approaches as an element of the total uncertainty. Among models of the same type (bulk or size-resolved), the <4% differences in total SO 2 -to-sulfate conversion are not very large and would certainly be acceptable in estimates of the contribution of aqueous-phase chemistry to the global sulfur cycle. For the conditions considered here, the underestimate of the SO 2 +O 3 reaction induces only a minor change in total sulfate formation despite a factor of 3 underestimate of the SO 2 +O 3 contribution. Under other atmospheric conditions, the O 3 reaction may be much more important than in this study. [36] Detailed analysis of the size-resolved aerosol/cloud chemistry models suggests appreciable uncertainty in the number of dry aerosols that are scavenged in a cloud updraft, even for the relatively simple ammonium bisulfate aerosol distribution considered here. All models calculate approximately consistent and high scavenging of the input dry aerosol mass, but the calculated number of cloud drops formed varies from drops cm 3. These differences were found to depend on different treatment of uptake of gases, assumptions regarding solution water content, and different values of water condensation mass accommodation coefficient. Among these, the value of the water condensation mass accommodation coefficient appears to be the single largest source of uncertainty that could be addressed experimentally; in a recent study, Shaw and Lamb [1999] report a value of 0.05 as the best fit to their data. Another important factor is the model resolution in diameter space, especially in the critical size range near the smallest particles activated, which is generally not known a priori from updraft and dry aerosol size distribution information. Finally, unlike the conditions chosen for this study, the composition of ambient aerosol is extremely variable, and is an additional source of uncertainty in N D. It is most likely the largest source of uncertainty in regions affected by anthropogenic pollution, where particles are not as hygroscopic as in more pristine regions. [37] Using a simple model of cloud radiative properties, the cloud drop number deviations among models were estimated to induce a 9% change in calculated cloud optical depths, and a corresponding change in absolute cloud albedo of up to 2%. For optically thin clouds, this represents an appreciable relative change in cloud albedo. Furthermore, such discrepancies arising in a simple parcel model with well-characterized aerosol composition suggest that radiative and microphysical processes within clouds are highly uncertain within more complex atmospheric cloud models, regional-scale meteorological models, and longerterm climate models where many factors in addition to the uncertainties shown here can arise. [38] The direct radiative effects of aerosols are also sensitive to how mass is added to an initial aerosol spectrum. Using a simple extinction calculation, we show that the changes to the radiative extinction efficiency, relevant to direct aerosol radiative forcing estimates, can be as large as 13% among model simulations of the same case. [39] The simulation carried out in this study is inherently limited in scope. Only a single chemical and aerosol condition is specified, with one updraft velocity and vertical displacement. Clearly more general conclusions can only be arrived at with a much wider range of physical and chemical conditions. Variations such as polluted versus clean, oxidant limited versus oxidant rich, overall acidity level and updraft speed are all factors that will influence the results presented here. However, a key factor regulating the magnitudes of the differences found is the relatively large variation in absolute number of cloud drops formed. This discrepancy would probably occur under many environmental conditions, given the reasons leading to it that were diagnosed from this study. This work suggests that an important next step in a cloud-aerosol model comparison series would be a more focused study of the cloud drop activation behavior in the lowest few tens of meters above cloud base in a cloud updraft.
12 AAC 4-12 KREIDENWEIS ET AL.: CLOUD CHEMISTRY COMPARISON [40] Acknowledgments. The authors thank two anonymous reviewers for helpful comments which greatly improved the manuscript. S. K. and G. F. acknowledge support of NOAA Office of Global Programs grants NA67RJ0152 and NA17RJ1228. A. N. acknowledges the support of Office of Naval Research grant N C. W. acknowledges the U.S. Environmental Protection Agency (EPA) for support under grant R M. J. acknowledges support from the National Science Foundation (grant ATM ), National Aeronautics and Space Administration (grant NAG5-8645) and the EPA. References Ayers, G. P., and T. V. Larson, Numerical study of droplet size dependent chemistry in oceanic, wintertime stratus cloud at southern mid-latitudes, J. Atmos. Chem., 11, , Barth, M. C., P. J. Rasch, J. T. Kiehl, C. M. Benkovitz, and S. E. Schwartz, Sulfur chemistry in the National Center for Atmospheric Research Community Climate Model: Description, evaluation, features and sensitivity to aqueous chemistry, J. Geophys. Res., 105, , Bohren, C. F., and D. R. Huffman, Absorption and scattering of light by small particles, 544 pp., John Wiley, New York, Boucher, O., and U. Lohmann, The sulfate-ccn-cloud albedo effect, Tellus, Ser. B, 47, , Chang, J. S., R. A. Brost, I. S. A. Isaksen, S. Madronich, P. Middleton, W. R. Stockwell, and C. J. Walcek, A three-dimensional Eulerian acid deposition model: Physical concepts and formulation, J. Geophys. Res., 92, 14,681 14,700, Collett, J. L., Jr., A. Bator, X. Rao, and B. Demoz, Acidity variations across the cloud drop size spectrum and their influence on rates of atmospheric sulfate production, Geophys. Res. Lett., 21, , Feingold, G., and S. M. Kreidenweis, Does cloud processing of aerosol enhance droplet concentrations, J. Geophys. Res., 105, 24,351 24,361, Feingold, G., S. M. Kreidenweis, and Y. Zhang, Stratocumulus processing of gases and cloud condensation nuclei, part I, Trajectory ensemble model, J. Geophys. Res., 103, 19,527 19,542, Gong, W., Modelling cloud chemistry in a regional aerosol model: Bulk vs. size resolved representation, in Air Pollution Modeling and Its Applications, vol. 15, edited by C. Borrego and G. Schayes, pp , Plenum, New York, Gurciullo, C. S., and S. N. Pandis, Effect of composition variations in cloud droplet populations on aqueous-phase chemistry, J. Geophys. Res, 102, , Health Effects Institute, Reanalysis of the Harvard Six Cities Study and the American Cancer Society Study of particulate air pollution and mortality, Health Effects Inst. spec. rep., Cambridge, Mass., July 2000 (available at Hegg, D. A., Cloud condensation nucleus-sulfate mass relationship and cloud albedo, J. Geophys. Res., 99, 25,903 25,907, Hegg, D. A., and T. V. Larson, The effects of microphysical parameterization on model predictions of sulfate production in clouds, Tellus, Ser. B, 42, , Hegg, D. A., P. F. Yuen, and T. V. Larson, Modeling the effects of heterogeneous cloud chemistry on the marine particle size distribution, J. Geophys. Res., 97, 12,927 12,933, Hegg, D., T. Larson, and P. F. Yuen, A theoretical study of the effect of relative humidity on light scattering by tropospheric aerosols, J. Geophys. Res., 98, 18,435 18,439, Hegg, D. A., R. Majeed, P. F. Yuen, M. B. Baker, and T. V. Larson, The impacts of SO 2 oxidation in cloud drops and in haze particles on aerosol light scattering and CCN activity, Geophys. Res. Lett., 23, , Hoppel, W. A., J. W. Fitzgerald, G. M. Frick, and R. E. Larson, Aerosol size distributions and optical properties found in the marine boundary layer over the Atlantic Ocean, J. Geophys. Res., 95, , Hoppel, W. A., G. M. Frick, J. W. Fitzgerald, and B. J. Wattle, A cloud chamber study of the effect that nonprecipitating water clouds have on the aerosol-size distribution, Aerosol Sci. Technol., 20(1), 1 30, Intergovernmental Panel on Climate Change, Third assessment report, technical summary, IPCC Secr., World Meteorol. Org., Geneva, Switzerland, Iribarne, J. V., and H. R. Cho, Models of cloud chemistry, Tellus, Ser. B, 41, 2 23, Jacobson, M. Z., Analysis of aerosol interactions with numerical techniques for solving coagulation, nucleation, condensation, dissolution, and reversible chemistry among multiple size distributions, J. Geophys. Res., 107(D19), 4366, doi: /2001jd002044, Jones, A., D. L. Roberts, and A. Slingo, A climate model study of indirect radiative forcing by anthropogenic sulfate aerosols, Nature, 370, , Kim, C.-H., S. M. Kreidenweis, G. Feingold, G. J. Frost, and M. K. Trainer, Modeling cloud effects on hydrogen peroxide and methylhydroperoxide in the marine atmosphere, J. Geophys. Res, 107(D2), 4018, doi: / 2000JD000285, Lelieveld, J., and J. Heintzenberg, Sulfate cooling effect on climate through in-cloud oxidation of anthropogenic SO 2, Science, 258, , Liu, X., and W. Seidl, Modeling study of cloud droplet nucleation and incloud sulfate production during the SANA 2 Campaign, J. Geophys. Res, 103, 16,145 16,158, Malm, W. C., J. F. Sisler, D. Huffman, R. A. Eldred, and T. A. Cahill, Spatial and seasonal trends in particle concentration and optical extinction in the United States, J. Geophys. Res., 99, , Nenes, A., S. Gahn, H. Abdul-Razzak, P. Y. Chuang, and J. H. Seinfeld, Kinetic limitations on cloud droplet formation and impact on cloud albedo, Tellus, Ser. B, 53, , Ogren, J. A., and R. J. Charlson, Implications for models and measurements of chemical inhomogeneities among cloud droplets, Tellus, Ser. B, 44, , Pandis, S. N., J. H. Seinfeld, and C. Pilinis, Chemical composition differences in fog and cloud droplets of different sizes, Atmos. Environ., Part A, 24, , Rasch, P. J., M. C. Barth, J. T. Kiehl, S. E. Schwartz, and C. M. Benkovitz, A description of the global sulfur cycle and its controlling processes in the National Center for Atmospheric Research Community Climate Model Version 3, J. Geophys. Res., 105, , Reilly, J. E., O. V. Rattigan, K. F. Moore, C. Judd, D. E. Sherman, V. A. Dutkiewicz, S. M. Kreidenweis, L. Husain, and J. L. Collett Jr., Drop size-dependent S(IV) oxidation in chemically heterogeneous California radiation fogs, Atmos. Environ., 35, , Seidl, W., Ionic concentrations and initial S(IV) oxidation rates in droplets during the condensational stage of cloud, Tellus, Ser. B, 41, 32 50, Seinfeld, J. H., and S. N. Pandis, Atmospheric Chemistry and Physics, 1326 pp., Wiley-Interscience, New York, Shaw, R. A., and D. Lamb, Experimental determination of the thermal accommodation and condensation coefficients of water, J. Chem. Phys., 111, 10,659 10,663, Stockwell, W. R., P. Middeleton, J. S. Chang, and X. Tang, The second generation regional acid deposition model chemical mechanism for regional air quality modeling, J. Geophys. Res., 95, 16,343 16,367, Tang, I. N., Chemical and size effects of hygroscopic aerosols on light scattering coefficients, J. Geophys. Res., 101, 19,245 19,250, Tang, I. N., and H. R. Munkelwitz, Water activities, densities, and refractive indices of aqueous sulfates and sodium nitrate droplets of atmospheric importance, J. Geophys. Res., 99, 18,801 18,808, Twomey, S., Aerosols, clouds and radiation, Atmos. Environ, Part A, 25, , Walcek, C. J., and G. Taylor, A theoretical method for computing vertical distributions of acidity and sulfate production within cumulus clouds, J. Atmos. Sci., 43, , Yuen, P.-F., D. A. Hegg, and T. V. Larson, The effects of in-cloud sulfate production on light-scattering properties of continental aerosol, J. Appl. Meteorol., 33, , Yuen, P.-F., D. A. Hegg, T. V. Larson, and M. C. Barth, Parameterization of heterogeneous droplet chemistry for use in bulk cloud models, J. Appl. Meteorol., 35, , Zhang, Y., S. M. Kreidenweis, and G. Feingold, Stratocumulus processing of gases and cloud condensation nuclei, 2, Chemistry sensitivity analyses, J. Geophys. Res., 104, 16,061 16,080, G. Feingold, National Oceanographic and Atmospheric Administration, 325 Broadway, Boulder, CO 80305, USA. W. Gong, Meteorological Service of Canada, 4905 Dufferin Street, Downsview, Ontario, Canada M3H 5T4. M. Z. Jacobson, Department of Civil and Environmental Engineering, Stanford University, Terman Engineering Center, Room M-13, Stanford, CA , USA. C.-H. Kim, National Institute of Environmental Research (NIER), Kyong-Seo-dong, 2-1, Seo-gu, Incheon , Korea. S. M. Kreidenweis, Department of Atmospheric Science, Colorado State University, Fort Collins, CO 80523, USA. (sonia@chem.atmos.colostate. edu) X. Liu and J. E. Penner, Atmospheric, Oceanic and Space Sciences, University of Michigan, 2455 Hayward, Ann Arbor, MI , USA. A. Nenes, School of Earth and Atmospheric Sciences, Georgia Institute of Technology, Atlanta, GA , USA. J. H. Seinfeld, Division of Chemistry and Chemical Engineering, , California Institute of Technology, 1200 E. California Blvd., Pasadena, CA 91125, USA. C. J. Walcek, Atmospheric Science Research Center, State University of New York, 251 Fuller Road, Albany, NY 12203, USA. (walcek@asrc. cestm.albany.edu)
Parameterization of the nitric acid effect on CCN activation
 Atmos. Chem. Phys., 5, 879 885, 25 SRef-ID: 168-7324/acp/25-5-879 European Geosciences Union Atmospheric Chemistry and Physics Parameterization of the nitric acid effect on CCN activation S. Romakkaniemi,
Atmos. Chem. Phys., 5, 879 885, 25 SRef-ID: 168-7324/acp/25-5-879 European Geosciences Union Atmospheric Chemistry and Physics Parameterization of the nitric acid effect on CCN activation S. Romakkaniemi,
CHAPTER 8. AEROSOLS 8.1 SOURCES AND SINKS OF AEROSOLS
 1 CHAPTER 8 AEROSOLS Aerosols in the atmosphere have several important environmental effects They are a respiratory health hazard at the high concentrations found in urban environments They scatter and
1 CHAPTER 8 AEROSOLS Aerosols in the atmosphere have several important environmental effects They are a respiratory health hazard at the high concentrations found in urban environments They scatter and
A cloud microphysics model including trace gas condensation and sulfate chemistry
 BOREAL ENVIRONMENT RESEARCH 8: 43 424 ISSN 239-695 Helsinki December 23 23 A cloud microphysics model including trace gas condensation and sulfate chemistry Harri Kokkola ), Sami Romakkaniemi ), Markku
BOREAL ENVIRONMENT RESEARCH 8: 43 424 ISSN 239-695 Helsinki December 23 23 A cloud microphysics model including trace gas condensation and sulfate chemistry Harri Kokkola ), Sami Romakkaniemi ), Markku
Sensitivity of cloud condensation nuclei activation processes to kinetic parameters
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 111,, doi:10.1029/2005jd006529, 2006 Sensitivity of cloud condensation nuclei activation processes to kinetic parameters P. Y. Chuang 1 Received 25 July 2005; revised
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 111,, doi:10.1029/2005jd006529, 2006 Sensitivity of cloud condensation nuclei activation processes to kinetic parameters P. Y. Chuang 1 Received 25 July 2005; revised
Black carbon radiative heating effects on cloud microphysics and implications for the aerosol indirect effect 2. Cloud microphysics
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 107, NO. D21, 4605, doi:10.1029/2002jd002101, 2002 Black carbon radiative heating effects on cloud microphysics and implications for the aerosol indirect effect 2.
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 107, NO. D21, 4605, doi:10.1029/2002jd002101, 2002 Black carbon radiative heating effects on cloud microphysics and implications for the aerosol indirect effect 2.
Updated H 2 SO 4 -H 2 O binary homogeneous nucleation look-up tables
 Click Here for Full Article JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 113,, doi:10.1029/2008jd010527, 2008 Updated H 2 SO 4 -H 2 O binary homogeneous nucleation look-up tables Fangqun Yu 1 Received 2 June
Click Here for Full Article JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 113,, doi:10.1029/2008jd010527, 2008 Updated H 2 SO 4 -H 2 O binary homogeneous nucleation look-up tables Fangqun Yu 1 Received 2 June
Modeling of cloud microphysics: from simple concepts to sophisticated parameterizations. Part I: warm-rain microphysics
 Modeling of cloud microphysics: from simple concepts to sophisticated parameterizations. Part I: warm-rain microphysics Wojciech Grabowski National Center for Atmospheric Research, Boulder, Colorado parameterization
Modeling of cloud microphysics: from simple concepts to sophisticated parameterizations. Part I: warm-rain microphysics Wojciech Grabowski National Center for Atmospheric Research, Boulder, Colorado parameterization
Black carbon radiative heating effects on cloud microphysics and implications for the aerosol indirect effect: 2. Cloud microphysics
 Black carbon radiative heating effects on cloud microphysics and implications for the aerosol indirect effect: 2. Cloud microphysics Athanasios Nenes, William C. Conant and John H. Seinfeld Departments
Black carbon radiative heating effects on cloud microphysics and implications for the aerosol indirect effect: 2. Cloud microphysics Athanasios Nenes, William C. Conant and John H. Seinfeld Departments
APPLICATION OF KOHLER THEORY: MODELING CLOUD CONDENSATION NUCLEI ACTIVITY
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 APPLICATION OF KOHLER THEORY: MODELING CLOUD CONDENSATION NUCLEI ACTIVITY Gavin Cornwell, Katherine Nadler, Alex Nguyen, and Steven Schill Department of
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 APPLICATION OF KOHLER THEORY: MODELING CLOUD CONDENSATION NUCLEI ACTIVITY Gavin Cornwell, Katherine Nadler, Alex Nguyen, and Steven Schill Department of
Implications of Sulfate Aerosols on Clouds, Precipitation and Hydrological Cycle
 Implications of Sulfate Aerosols on Clouds, Precipitation and Hydrological Cycle Source: Sulfate aerosols are produced by chemical reactions in the atmosphere from gaseous precursors (with the exception
Implications of Sulfate Aerosols on Clouds, Precipitation and Hydrological Cycle Source: Sulfate aerosols are produced by chemical reactions in the atmosphere from gaseous precursors (with the exception
Chemical and Dynamical Effects on Cloud Droplet Number: Implications for Estimates of the Aerosol Indirect Effect
 Chemical and Dynamical Effects on Cloud Droplet Number: Implications for Estimates of the Aerosol Indirect Effect SARA LANCE 1, A. NENES 1,2*, T. RISSMAN 3 1 School of Earth and Atmospheric Sciences and
Chemical and Dynamical Effects on Cloud Droplet Number: Implications for Estimates of the Aerosol Indirect Effect SARA LANCE 1, A. NENES 1,2*, T. RISSMAN 3 1 School of Earth and Atmospheric Sciences and
The Atmospheric Chemistry and Physics of Ammonia
 The Atmospheric Chemistry and Physics of Ammonia Russell Dickerson Dept. Meteorology, The University of Maryland Presented at the National Atmospheric Deposition Program Ammonia Workshop October 23, 2003
The Atmospheric Chemistry and Physics of Ammonia Russell Dickerson Dept. Meteorology, The University of Maryland Presented at the National Atmospheric Deposition Program Ammonia Workshop October 23, 2003
Importance of vertical velocity variations in the cloud droplet nucleation process of marine stratus clouds
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 110,, doi:10.1029/2004jd004922, 2005 Importance of vertical velocity variations in the cloud droplet nucleation process of marine stratus clouds Yiran Peng, 1,2 Ulrike
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 110,, doi:10.1029/2004jd004922, 2005 Importance of vertical velocity variations in the cloud droplet nucleation process of marine stratus clouds Yiran Peng, 1,2 Ulrike
Aerosol Dynamics. Antti Lauri NetFAM Summer School Zelenogorsk, 9 July 2008
 Aerosol Dynamics Antti Lauri NetFAM Summer School Zelenogorsk, 9 July 2008 Department of Physics, Division of Atmospheric Sciences and Geophysics, University of Helsinki Aerosol Dynamics: What? A way to
Aerosol Dynamics Antti Lauri NetFAM Summer School Zelenogorsk, 9 July 2008 Department of Physics, Division of Atmospheric Sciences and Geophysics, University of Helsinki Aerosol Dynamics: What? A way to
DEVELOPMENT AND TESTING OF AN AEROSOL / STRATUS CLOUD PARAMETERIZATION SCHEME FOR MIDDLE AND HIGH LATITUDES
 DOE/ER/6195&3 DEVELOPMENT AND TESTING OF AN AEROSOL / STRATUS CLOUD PARAMETERIZATION SCHEME FOR MIDDLE AND HIGH LATITUDES Year 3 Technical Progress Report For Period of Activity, Year 3: November 1,1996
DOE/ER/6195&3 DEVELOPMENT AND TESTING OF AN AEROSOL / STRATUS CLOUD PARAMETERIZATION SCHEME FOR MIDDLE AND HIGH LATITUDES Year 3 Technical Progress Report For Period of Activity, Year 3: November 1,1996
Ammonium Bisulfate/Water Equilibrium and Metastability Phase Diagrams
 J. Phys. Chem. A 1997, 101, 4191-4195 4191 Ammonium Bisulfate/Water Equilibrium and Metastability Phase Diagrams Dan G. Imre,*, Jun Xu, I. N. Tang, and R. McGraw EnVironmental Chemistry DiVision, Department
J. Phys. Chem. A 1997, 101, 4191-4195 4191 Ammonium Bisulfate/Water Equilibrium and Metastability Phase Diagrams Dan G. Imre,*, Jun Xu, I. N. Tang, and R. McGraw EnVironmental Chemistry DiVision, Department
Aerosol Basics: Definitions, size distributions, structure
 Aerosol Basics: Definitions, size distributions, structure Antti Lauri NetFAM Summer School Zelenogorsk, 9 July 2008 Department of Physics, Division of Atmospheric Sciences and Geophysics, University of
Aerosol Basics: Definitions, size distributions, structure Antti Lauri NetFAM Summer School Zelenogorsk, 9 July 2008 Department of Physics, Division of Atmospheric Sciences and Geophysics, University of
Droplet Nucleation: Physically-Based Parameterizations and Comparative
 Droplet Nucleation: Physically-Based Parameterizations and Comparative Evaluation 1 1 1 1 1 1 1 1 0 1 0 1 Steven J. Ghan 1, Hayder Abdul-Razzak, Athanasios Nenes, Yi Ming, Xiaohong Liu 1, Mikhail Ovchinnikov
Droplet Nucleation: Physically-Based Parameterizations and Comparative Evaluation 1 1 1 1 1 1 1 1 0 1 0 1 Steven J. Ghan 1, Hayder Abdul-Razzak, Athanasios Nenes, Yi Ming, Xiaohong Liu 1, Mikhail Ovchinnikov
ATOC 3500/CHEM 3152 Week 9, March 8, 2016
 ATOC 3500/CHEM 3152 Week 9, March 8, 2016 Hand back Midterm Exams (average = 84) Interaction of atmospheric constituents with light Haze and Visibility Aerosol formation processes (more detail) Haze and
ATOC 3500/CHEM 3152 Week 9, March 8, 2016 Hand back Midterm Exams (average = 84) Interaction of atmospheric constituents with light Haze and Visibility Aerosol formation processes (more detail) Haze and
Consistent estimates from satellites and models for the first aerosol indirect forcing
 GEOPHYSICAL RESEARCH LETTERS, VOL. 39,, doi:10.1029/2012gl051870, 2012 Consistent estimates from satellites and models for the first aerosol indirect forcing Joyce E. Penner, 1 Cheng Zhou, 1 and Li Xu
GEOPHYSICAL RESEARCH LETTERS, VOL. 39,, doi:10.1029/2012gl051870, 2012 Consistent estimates from satellites and models for the first aerosol indirect forcing Joyce E. Penner, 1 Cheng Zhou, 1 and Li Xu
9 Condensation. Learning Goals. After studying this chapter, students should be able to:
 9 Condensation Learning Goals After studying this chapter, students should be able to: 1. explain the microphysical processes that operate in clouds to influence the formation and growth of cloud droplets
9 Condensation Learning Goals After studying this chapter, students should be able to: 1. explain the microphysical processes that operate in clouds to influence the formation and growth of cloud droplets
Slides partly by Antti Lauri and Hannele Korhonen. Liquid or solid particles suspended in a carrier gas Described by their
 Atmospheric Aerosols Slides partly by Antti Lauri and Hannele Korhonen Aerosol particles Liquid or solid particles suspended in a carrier gas Described by their Size Concentration - Number - Surface -
Atmospheric Aerosols Slides partly by Antti Lauri and Hannele Korhonen Aerosol particles Liquid or solid particles suspended in a carrier gas Described by their Size Concentration - Number - Surface -
P = x i. P i. = y i. Aerosol and Aqueous Chemistry. Raoult s Law. Raoult s Law vs. Henry s Law. or C i. = HC i. = k H
 The Great Smog Aerosol and Aqueous Chemistry Equilibrium Partitioning Oxidation and Oxidants Other Surface-driven Fogs in London were a common occurrence, but the events that began on the 5th of December
The Great Smog Aerosol and Aqueous Chemistry Equilibrium Partitioning Oxidation and Oxidants Other Surface-driven Fogs in London were a common occurrence, but the events that began on the 5th of December
An investigation into the aerosol dispersion effect through the activation process in marine stratus clouds
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 112,, doi:10.1029/2006jd007401, 2007 An investigation into the aerosol dispersion effect through the activation process in marine stratus clouds Yiran Peng, 1 Ulrike
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 112,, doi:10.1029/2006jd007401, 2007 An investigation into the aerosol dispersion effect through the activation process in marine stratus clouds Yiran Peng, 1 Ulrike
Influence of Organic-Containing Aerosols on Marine Boundary Layer Processes
 DISTRIBUTION STATEMENT A. Approved for public release; distribution is unlimited. Influence of Organic-Containing Aerosols on Marine Boundary Layer Processes John H. Seinfeld California Institute of Technology,
DISTRIBUTION STATEMENT A. Approved for public release; distribution is unlimited. Influence of Organic-Containing Aerosols on Marine Boundary Layer Processes John H. Seinfeld California Institute of Technology,
Evaluation of a new Cloud Droplet Activation Parameterization with In Situ Data from CRYSTAL-FACE and CSTRIPE
 Evaluation of a new Cloud Droplet Activation Parameterization with In Situ Data from CRYSTAL-FACE and CSTRIPE Nicholas Meskhidze 1, Athanasios Nenes 1,2*, William C. Conant 3, John H. Seinfeld 3,4 1 School
Evaluation of a new Cloud Droplet Activation Parameterization with In Situ Data from CRYSTAL-FACE and CSTRIPE Nicholas Meskhidze 1, Athanasios Nenes 1,2*, William C. Conant 3, John H. Seinfeld 3,4 1 School
Parameterization of cloud droplet formation in global climate models
 1 Parameterization of cloud droplet formation in global climate models ATHANASIOS NENES 1 and JOHN H. SEINFELD Department of Chemical Engineering, Mail Code 210-41, California Institute of Technology,
1 Parameterization of cloud droplet formation in global climate models ATHANASIOS NENES 1 and JOHN H. SEINFELD Department of Chemical Engineering, Mail Code 210-41, California Institute of Technology,
MODELING THE FORMATION AND DEPOSITION OF ACIDIC POLLUTANTS
 Atmospheric Deposition (Proceedings of the Baltimore Symposium, May 1989). IAHS Pub], No. 179. MODELING THE FORMATION AND DEPOSITION OF ACIDIC POLLUTANTS Chris J. Walcek Atmospheric Sciences Research Center,
Atmospheric Deposition (Proceedings of the Baltimore Symposium, May 1989). IAHS Pub], No. 179. MODELING THE FORMATION AND DEPOSITION OF ACIDIC POLLUTANTS Chris J. Walcek Atmospheric Sciences Research Center,
Kinetic Limitations on Cloud Droplet Formation and Impact on Cloud Albedo
 1 Appeared in Tellus B, 53, 133 149, 2001. Kinetic Limitations on Cloud Droplet Formation and Impact on Cloud Albedo Athanasios Nenes Department of Chemical Engineering,Mail Code 210-41, California Institute
1 Appeared in Tellus B, 53, 133 149, 2001. Kinetic Limitations on Cloud Droplet Formation and Impact on Cloud Albedo Athanasios Nenes Department of Chemical Engineering,Mail Code 210-41, California Institute
Aerosol Effects on Water and Ice Clouds
 Aerosol Effects on Water and Ice Clouds Ulrike Lohmann Department of Physics and Atmospheric Science, Dalhousie University, Halifax, N. S., Canada Contributions from Johann Feichter, Johannes Hendricks,
Aerosol Effects on Water and Ice Clouds Ulrike Lohmann Department of Physics and Atmospheric Science, Dalhousie University, Halifax, N. S., Canada Contributions from Johann Feichter, Johannes Hendricks,
Satellite-based estimate of global aerosol-cloud radiative forcing by marine warm clouds
 SUPPLEMENTARY INFORMATION DOI: 10.1038/NGEO2214 Satellite-based estimate of global aerosol-cloud radiative forcing by marine warm clouds Y.-C. Chen, M. W. Christensen, G. L. Stephens, and J. H. Seinfeld
SUPPLEMENTARY INFORMATION DOI: 10.1038/NGEO2214 Satellite-based estimate of global aerosol-cloud radiative forcing by marine warm clouds Y.-C. Chen, M. W. Christensen, G. L. Stephens, and J. H. Seinfeld
Aerosol modeling with WRF/Chem
 Aerosol modeling with WRF/Chem Jan Kazil University of Colorado / NOAA Earth System Research Laboratory WRF/Chem Tutorial, 3 August 2015 (WRF/Chem 3.7) Part I - Introduction Overview of... Aerosol Aerosol
Aerosol modeling with WRF/Chem Jan Kazil University of Colorado / NOAA Earth System Research Laboratory WRF/Chem Tutorial, 3 August 2015 (WRF/Chem 3.7) Part I - Introduction Overview of... Aerosol Aerosol
Clouds, Haze, and Climate Change
 Clouds, Haze, and Climate Change Jim Coakley College of Oceanic and Atmospheric Sciences Earth s Energy Budget and Global Temperature Incident Sunlight 340 Wm -2 Reflected Sunlight 100 Wm -2 Emitted Terrestrial
Clouds, Haze, and Climate Change Jim Coakley College of Oceanic and Atmospheric Sciences Earth s Energy Budget and Global Temperature Incident Sunlight 340 Wm -2 Reflected Sunlight 100 Wm -2 Emitted Terrestrial
CHAPTER 1. MEASURES OF ATMOSPHERIC COMPOSITION
 1 CHAPTER 1. MEASURES OF ATMOSPHERIC COMPOSITION The objective of atmospheric chemistry is to understand the factors that control the concentrations of chemical species in the atmosphere. In this book
1 CHAPTER 1. MEASURES OF ATMOSPHERIC COMPOSITION The objective of atmospheric chemistry is to understand the factors that control the concentrations of chemical species in the atmosphere. In this book
Leo Donner GFDL/NOAA, Princeton University. EGU, Vienna, 18 April 2016
 Cloud Dynamical Controls on Climate Forcing by Aerosol-Cloud Interactions: New Insights from Observations, High- Resolution Models, and Parameterizations Leo Donner GFDL/NOAA, Princeton University EGU,
Cloud Dynamical Controls on Climate Forcing by Aerosol-Cloud Interactions: New Insights from Observations, High- Resolution Models, and Parameterizations Leo Donner GFDL/NOAA, Princeton University EGU,
Summary of the cloud chemistry modeling intercomparison: Photochemical box model simulation
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 108, NO. D7, 4214, doi:10.1029/2002jd002673, 2003 Summary of the cloud chemistry modeling intercomparison: Photochemical box model simulation M. C. Barth, 1 S. Sillman,
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 108, NO. D7, 4214, doi:10.1029/2002jd002673, 2003 Summary of the cloud chemistry modeling intercomparison: Photochemical box model simulation M. C. Barth, 1 S. Sillman,
Lecture 26. Regional radiative effects due to anthropogenic aerosols. Part 2. Haze and visibility.
 Lecture 26. Regional radiative effects due to anthropogenic aerosols. Part 2. Haze and visibility. Objectives: 1. Attenuation of atmospheric radiation by particulates. 2. Haze and Visibility. Readings:
Lecture 26. Regional radiative effects due to anthropogenic aerosols. Part 2. Haze and visibility. Objectives: 1. Attenuation of atmospheric radiation by particulates. 2. Haze and Visibility. Readings:
On the formation of radiation fogs under heavily polluted conditions
 Atmos. Chem. Phys.,, 581 589, 2 www.atmos-chem-phys.org/acp//581/ Atmospheric Chemistry and Physics On the formation of radiation fogs under heavily polluted conditions H. Kokkola, S. Romakkaniemi, and
Atmos. Chem. Phys.,, 581 589, 2 www.atmos-chem-phys.org/acp//581/ Atmospheric Chemistry and Physics On the formation of radiation fogs under heavily polluted conditions H. Kokkola, S. Romakkaniemi, and
Black carbon radiative heating effects on cloud microphysics and implications for the aerosol indirect effect 1. Extended Köhler theory
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 107, NO. D21, 4604, doi:10.1029/2002jd002094, 2002 Black carbon radiative heating effects on cloud microphysics and implications for the aerosol indirect effect 1.
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 107, NO. D21, 4604, doi:10.1029/2002jd002094, 2002 Black carbon radiative heating effects on cloud microphysics and implications for the aerosol indirect effect 1.
Aerosol Optical Properties
 ATM 507 Lecture 25 Text reading Chapter 15 Paper Due Dec. 9 Review Session Dec. 9 Final Dec. 12 (10:30 AM-12:30 PM) Today s topic Aerosol Optical Properties 1 Aerosol Optical Properties There are a number
ATM 507 Lecture 25 Text reading Chapter 15 Paper Due Dec. 9 Review Session Dec. 9 Final Dec. 12 (10:30 AM-12:30 PM) Today s topic Aerosol Optical Properties 1 Aerosol Optical Properties There are a number
An investigation into the aerosol dispersion effect through the activation process in marine stratus clouds
 1 An investigation into the aerosol dispersion effect through the activation process in marine stratus clouds Yiran Peng, 1, Ulrike Lohmann, 2 Richard Leaitch 3 and Markku Kulmala 4 Short title: 1 Max
1 An investigation into the aerosol dispersion effect through the activation process in marine stratus clouds Yiran Peng, 1, Ulrike Lohmann, 2 Richard Leaitch 3 and Markku Kulmala 4 Short title: 1 Max
Comparing Modal and Sectional Approaches in Modeling Particulate Matter in Northern California
 Comparing Modal and Sectional Approaches in Modeling Particulate Matter in Northern California K. Max Zhang* [1], Jinyou Liang [2], Anthony S. Wexler [1], and Ajith Kaduwela [1,2] 1. University of California,
Comparing Modal and Sectional Approaches in Modeling Particulate Matter in Northern California K. Max Zhang* [1], Jinyou Liang [2], Anthony S. Wexler [1], and Ajith Kaduwela [1,2] 1. University of California,
AEROCOM-Workshop,Paris, June 2-3, model. Øyvind Seland; Alf Kirkevåg
 An AGCM operated at University of Oslo (UiO) Norway Øyvind Seland; Alf Kirkevåg AEROCOM-Workshop,Paris, June 2-3, 2003 by Kirkevåg; Jón Egill Kristjánsson; ; Trond Iversen Basic: NCAR-CCM3.2 CCM3.2 (Kiehl,et
An AGCM operated at University of Oslo (UiO) Norway Øyvind Seland; Alf Kirkevåg AEROCOM-Workshop,Paris, June 2-3, 2003 by Kirkevåg; Jón Egill Kristjánsson; ; Trond Iversen Basic: NCAR-CCM3.2 CCM3.2 (Kiehl,et
Clouds associated with cold and warm fronts. Whiteman (2000)
 Clouds associated with cold and warm fronts Whiteman (2000) Dalton s law of partial pressures! The total pressure exerted by a mixture of gases equals the sum of the partial pressure of the gases! Partial
Clouds associated with cold and warm fronts Whiteman (2000) Dalton s law of partial pressures! The total pressure exerted by a mixture of gases equals the sum of the partial pressure of the gases! Partial
Progress on Application of Modal Aerosol Dynamics to CAM
 Progress on Application of Modal Aerosol Dynamics to CAM Xiaohong Liu, Steve Ghan, Richard Easter, Rahul Zaveri, Yun Qian (Pacific Northwest National Laboratory) Jean-Francois Lamarque, Peter Hess, Natalie
Progress on Application of Modal Aerosol Dynamics to CAM Xiaohong Liu, Steve Ghan, Richard Easter, Rahul Zaveri, Yun Qian (Pacific Northwest National Laboratory) Jean-Francois Lamarque, Peter Hess, Natalie
Direct radiative forcing due to aerosols in Asia during March 2002
 Direct radiative forcing due to aerosols in Asia during March 2002 Soon-Ung Park, Jae-In Jeong* Center for Atmospheric and Environmental Modeling *School of Earth and Environmental Sciences, Seoul National
Direct radiative forcing due to aerosols in Asia during March 2002 Soon-Ung Park, Jae-In Jeong* Center for Atmospheric and Environmental Modeling *School of Earth and Environmental Sciences, Seoul National
THE RELATIONSHIP BETWEEN CLOUD DROPLET AND AEROSOL NUMBER CONCENTRATIONS FOR CLIMATE MODELS
 INTERNATIONAL JOURNAL OF CLIMATOLOGY, VOL. 16, 94 1-946 (1 996) 551.521.1 l(4) THE RELATIONSHIP BETWEEN CLOUD DROPLET AND AEROSOL NUMBER CONCENTRATIONS FOR CLIMATE MODELS 1. GULTEPE and G. A. ISAAC Cloud
INTERNATIONAL JOURNAL OF CLIMATOLOGY, VOL. 16, 94 1-946 (1 996) 551.521.1 l(4) THE RELATIONSHIP BETWEEN CLOUD DROPLET AND AEROSOL NUMBER CONCENTRATIONS FOR CLIMATE MODELS 1. GULTEPE and G. A. ISAAC Cloud
Comparison of AERONET inverted size distributions to measured distributions from the Aerodyne Aerosol Mass Spectrometer
 Comparison of inverted size distributions to measured distributions from the Aerodyne Aerosol Mass Spectrometer Peter DeCarlo Remote Sensing Project April 28, 23 Introduction The comparison of direct in-situ
Comparison of inverted size distributions to measured distributions from the Aerodyne Aerosol Mass Spectrometer Peter DeCarlo Remote Sensing Project April 28, 23 Introduction The comparison of direct in-situ
Three-dimensional (3-D) radiative transfer codes are mainly used in cloud studies (e.g., LES models) and vegetation studies.
 Lecture 5. Composition and structure of the Earth s atmosphere. Basic properties of gases, aerosols, and clouds that are important for radiative transfer modeling. Objectives: 1. Structure of the Earth
Lecture 5. Composition and structure of the Earth s atmosphere. Basic properties of gases, aerosols, and clouds that are important for radiative transfer modeling. Objectives: 1. Structure of the Earth
Aerosols AP sizes AP types Sources Sinks Amount and lifetime Aerosol radiative effects. Aerosols. Trude Storelvmo Aerosols 1 / 21
 Aerosols Trude Storelvmo Aerosols 1 / 21 Aerosols: Definition Definition of an aerosol: disperse system with air as carrier gas and a solid or liquid or a mixture of both as disperse phases. Aerosol particles
Aerosols Trude Storelvmo Aerosols 1 / 21 Aerosols: Definition Definition of an aerosol: disperse system with air as carrier gas and a solid or liquid or a mixture of both as disperse phases. Aerosol particles
A novel model to predict the physical state of atmospheric H 2 SO 4 /NH 3 /H 2 O aerosol particles
 Atmos. Chem. Phys., 3, 909 924, 2003 Atmospheric Chemistry and Physics A novel model to predict the physical state of atmospheric H 2 SO 4 /NH 3 /H 2 O aerosol particles C. A. Colberg, B. P. Luo, H. Wernli,
Atmos. Chem. Phys., 3, 909 924, 2003 Atmospheric Chemistry and Physics A novel model to predict the physical state of atmospheric H 2 SO 4 /NH 3 /H 2 O aerosol particles C. A. Colberg, B. P. Luo, H. Wernli,
Precipitation Formation, and RADAR Equation by Dario B. Giaiotti and Fulvio Stel (1)
 PhD Environmental Fluid Mechanics Physics of the Atmosphere University of Trieste International Center for Theoretical Physics Precipitation Formation, and RADAR Equation by Dario B. Giaiotti and Fulvio
PhD Environmental Fluid Mechanics Physics of the Atmosphere University of Trieste International Center for Theoretical Physics Precipitation Formation, and RADAR Equation by Dario B. Giaiotti and Fulvio
Phase Transformations of the Ternary System (NH 4 ) 2 SO 4 - H 2 SO 4 -H 2 O and the Implications for Cirrus Cloud Formation
 Phase Transformations of the Ternary System (NH 4 ) 2 SO 4 - H 2 SO 4 -H 2 O and the Implications for Cirrus Cloud Formation Scot T. Martin Department of Environmental Sciences and Engineering, The University
Phase Transformations of the Ternary System (NH 4 ) 2 SO 4 - H 2 SO 4 -H 2 O and the Implications for Cirrus Cloud Formation Scot T. Martin Department of Environmental Sciences and Engineering, The University
Observed impacts of vertical velocity on cloud microphysics and implications for aerosol indirect effects
 GEOPHYSICAL RESEARCH LETTERS, VOL. 39,, doi:10.1029/2012gl053599, 2012 Observed impacts of vertical velocity on cloud microphysics and implications for aerosol indirect effects Chunsong Lu, 1,2 Yangang
GEOPHYSICAL RESEARCH LETTERS, VOL. 39,, doi:10.1029/2012gl053599, 2012 Observed impacts of vertical velocity on cloud microphysics and implications for aerosol indirect effects Chunsong Lu, 1,2 Yangang
Warm Rain Precipitation Processes
 Warm Rain Precipitation Processes Cloud and Precipitation Systems November 16, 2005 Jonathan Wolfe 1. Introduction Warm and cold precipitation formation processes are fundamentally different in a variety
Warm Rain Precipitation Processes Cloud and Precipitation Systems November 16, 2005 Jonathan Wolfe 1. Introduction Warm and cold precipitation formation processes are fundamentally different in a variety
Analysis of Cloud-Radiation Interactions Using ARM Observations and a Single-Column Model
 Analysis of Cloud-Radiation Interactions Using ARM Observations and a Single-Column Model S. F. Iacobellis, R. C. J. Somerville, D. E. Lane, and J. Berque Scripps Institution of Oceanography University
Analysis of Cloud-Radiation Interactions Using ARM Observations and a Single-Column Model S. F. Iacobellis, R. C. J. Somerville, D. E. Lane, and J. Berque Scripps Institution of Oceanography University
Problem Session (afternoon, 9/19/11)
 Problem Session (afternoon, 9/19/11) CCN growth; supersaturation in a updraft (finish up last few slides from Lecture 12) Atmospheric Aerosols (2 page notes + spreadsheet) Computing CCN spectra (spreadsheet)
Problem Session (afternoon, 9/19/11) CCN growth; supersaturation in a updraft (finish up last few slides from Lecture 12) Atmospheric Aerosols (2 page notes + spreadsheet) Computing CCN spectra (spreadsheet)
An adsorption model of insoluble particle activation: Application to black carbon
 Click Here for Full Article JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 112,, doi:10.1029/2007jd008549, 2007 An adsorption model of insoluble particle activation: Application to black carbon B. F. Henson 1 Received
Click Here for Full Article JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 112,, doi:10.1029/2007jd008549, 2007 An adsorption model of insoluble particle activation: Application to black carbon B. F. Henson 1 Received
Importance of Composition and Hygroscopicity of BC Particles to the Effect of BC Mitigation on Cloud Properties: Application to California Conditions
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 Importance of Composition and Hygroscopicity of BC Particles to the Effect of BC Mitigation on Cloud Properties:
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 Importance of Composition and Hygroscopicity of BC Particles to the Effect of BC Mitigation on Cloud Properties:
Aerosol-Cloud-Radiation Interactions in Atmospheric Forecast Models
 Aerosol-Cloud-Radiation Interactions in Atmospheric Forecast Models John H. Seinfeld, Principal Investigator California Institute of Technology 1200 E. California Blvd., M/C 210-41 Pasadena, CA 91125 (626)
Aerosol-Cloud-Radiation Interactions in Atmospheric Forecast Models John H. Seinfeld, Principal Investigator California Institute of Technology 1200 E. California Blvd., M/C 210-41 Pasadena, CA 91125 (626)
Step 1. Step 2. g l = g v. dg = 0 We have shown that over a plane surface of water. g v g l = ρ v R v T ln e/e sat. this can be rewritten
 The basic question is what makes the existence of a droplet thermodynamically preferable to the existence only of water vapor. We have already derived an expression for the saturation vapor pressure over
The basic question is what makes the existence of a droplet thermodynamically preferable to the existence only of water vapor. We have already derived an expression for the saturation vapor pressure over
Aerosols and climate. Rob Wood, Atmospheric Sciences
 Aerosols and climate Rob Wood, Atmospheric Sciences What are aerosols? Solid or liquid particles suspended in air Sizes range from a few nm to a few thousand nm Huge range of masses Where do aerosols come
Aerosols and climate Rob Wood, Atmospheric Sciences What are aerosols? Solid or liquid particles suspended in air Sizes range from a few nm to a few thousand nm Huge range of masses Where do aerosols come
The Atmosphere. All of it. In one hour. Mikael Witte 10/27/2010
 The Atmosphere All of it. In one hour. Mikael Witte 10/27/2010 Outline Structure Dynamics - heat transport Composition Trace constituent compounds Some Atmospheric Processes Ozone destruction in stratosphere
The Atmosphere All of it. In one hour. Mikael Witte 10/27/2010 Outline Structure Dynamics - heat transport Composition Trace constituent compounds Some Atmospheric Processes Ozone destruction in stratosphere
Urban background aerosols: Negative correlations of particle modes and fragmentation mechanism
 Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 34, L11811, doi:10.1029/2006gl029109, 2007 Urban background aerosols: Negative correlations of particle modes and fragmentation mechanism
Click Here for Full Article GEOPHYSICAL RESEARCH LETTERS, VOL. 34, L11811, doi:10.1029/2006gl029109, 2007 Urban background aerosols: Negative correlations of particle modes and fragmentation mechanism
Highly Biased Hygroscopicity Derived from Size-Resolved Cloud Condensation Nuclei Activation Ratios without Data Inversion
 ATMOSPHERIC AND OCEANIC SCIENCE LETTERS, 2014, VOL. 7, NO. 3, 254 259 Highly Biased Hygroscopicity Derived from Size-Resolved Cloud Condensation Nuclei Activation Ratios without Data Inversion DENG Zhao-Ze
ATMOSPHERIC AND OCEANIC SCIENCE LETTERS, 2014, VOL. 7, NO. 3, 254 259 Highly Biased Hygroscopicity Derived from Size-Resolved Cloud Condensation Nuclei Activation Ratios without Data Inversion DENG Zhao-Ze
Precipitation Processes
 Precipitation Processes Dave Rahn Precipitation formation processes may be classified into two categories. These are cold and warm processes, where cold processes can only occur below 0 C and warm processes
Precipitation Processes Dave Rahn Precipitation formation processes may be classified into two categories. These are cold and warm processes, where cold processes can only occur below 0 C and warm processes
Arctic Chemistry And Climate
 21 July 2016 Connaught Summer Institute 1 Arctic Chemistry And Climate Connaught Summer Institute 2016 William (Bill) Simpson Geophysical Institute and Department of Chemistry, University of Alaska Fairbanks
21 July 2016 Connaught Summer Institute 1 Arctic Chemistry And Climate Connaught Summer Institute 2016 William (Bill) Simpson Geophysical Institute and Department of Chemistry, University of Alaska Fairbanks
Supplement of Multiphase oxidation of SO 2 by NO 2 on CaCO 3 particles
 Supplement of Atmos. Chem. Phys., 18, 2481 2493, 2018 https://doi.org/10.5194/acp-18-2481-2018-supplement Author(s) 2018. This work is distributed under the Creative Commons Attribution 4.0 License. Supplement
Supplement of Atmos. Chem. Phys., 18, 2481 2493, 2018 https://doi.org/10.5194/acp-18-2481-2018-supplement Author(s) 2018. This work is distributed under the Creative Commons Attribution 4.0 License. Supplement
Use of In Situ Data to Test a Raman Lidar Based Cloud Condensation Nuclei Remote Sensing Method
 FEBRUARY 2004 NOTES AND CORRESPONDENCE 387 Use of In Situ Data to Test a Raman Lidar Based Cloud Condensation Nuclei Remote Sensing Method STEVEN J. GHAN Pacific Northwest National Laboratory, Richland,
FEBRUARY 2004 NOTES AND CORRESPONDENCE 387 Use of In Situ Data to Test a Raman Lidar Based Cloud Condensation Nuclei Remote Sensing Method STEVEN J. GHAN Pacific Northwest National Laboratory, Richland,
CONTENTS 1 MEASURES OF ATMOSPHERIC COMPOSITION
 i CONTENTS 1 MEASURES OF ATMOSPHERIC COMPOSITION 1 1.1 MIXING RATIO 1 1.2 NUMBER DENSITY 2 1.3 PARTIAL PRESSURE 6 PROBLEMS 10 1.1 Fog formation 10 1.2 Phase partitioning of water in cloud 10 1.3 The ozone
i CONTENTS 1 MEASURES OF ATMOSPHERIC COMPOSITION 1 1.1 MIXING RATIO 1 1.2 NUMBER DENSITY 2 1.3 PARTIAL PRESSURE 6 PROBLEMS 10 1.1 Fog formation 10 1.2 Phase partitioning of water in cloud 10 1.3 The ozone
climate change Contents CO 2 (ppm)
 climate change CO 2 (ppm) 2007 Joachim Curtius Institut für Physik der Atmosphäre Universität Mainz Contents 1. Summary 2. Background 3. Climate change: observations 4. CO 2 5. OtherGreenhouse Gases (GHGs):
climate change CO 2 (ppm) 2007 Joachim Curtius Institut für Physik der Atmosphäre Universität Mainz Contents 1. Summary 2. Background 3. Climate change: observations 4. CO 2 5. OtherGreenhouse Gases (GHGs):
Cloud Condensation Nuclei Hygroscopic Parameter Kappa
 Cloud Condensation Nuclei Hygroscopic Parameter Kappa Covers Reading Material in Chapter 17.5 Atmospheric Sciences 5200 Physical Meteorology III: Cloud Physics Cloud Condensation Nuclei (CCN) Form a cloud
Cloud Condensation Nuclei Hygroscopic Parameter Kappa Covers Reading Material in Chapter 17.5 Atmospheric Sciences 5200 Physical Meteorology III: Cloud Physics Cloud Condensation Nuclei (CCN) Form a cloud
Humidity impact on the aerosol effect in warm cumulus clouds
 GEOPHYSICAL RESEARCH LETTERS, VOL. 35, L17804, doi:10.1029/2008gl034178, 2008 Humidity impact on the aerosol effect in warm cumulus clouds O. Altaratz, 1 I. Koren, 1 and T. Reisin 2 Received 31 March 2008;
GEOPHYSICAL RESEARCH LETTERS, VOL. 35, L17804, doi:10.1029/2008gl034178, 2008 Humidity impact on the aerosol effect in warm cumulus clouds O. Altaratz, 1 I. Koren, 1 and T. Reisin 2 Received 31 March 2008;
An Annual Cycle of Arctic Cloud Microphysics
 An Annual Cycle of Arctic Cloud Microphysics M. D. Shupe Science and Technology Corporation National Oceanic and Atmospheric Administration Environmental Technology Laboratory Boulder, Colorado T. Uttal
An Annual Cycle of Arctic Cloud Microphysics M. D. Shupe Science and Technology Corporation National Oceanic and Atmospheric Administration Environmental Technology Laboratory Boulder, Colorado T. Uttal
representing in-cloud oxidation of sulfur in a particle-based cloud-microphysics scheme
 representing incloud oxidation of sulfur in a particlebased cloudmicrophysics scheme Anna Jaruga, Hanna Paw lowska, Sylwester Arabas Institute of Geophysics Faculty of Physics, University of Warsaw 8 th
representing incloud oxidation of sulfur in a particlebased cloudmicrophysics scheme Anna Jaruga, Hanna Paw lowska, Sylwester Arabas Institute of Geophysics Faculty of Physics, University of Warsaw 8 th
J. Schneider & Chr. Voigt - Physics and Chemistry of Aerosols and Ice Clouds
 Chapter 8 Contrails and contrail cirrus 8.1 Introduction - Terminology 8.2 Contrail formation conditions 8.3 Heterogeneous nucleation on volatile aerosol and soot 8.4 Indirect effect of soot on cirrus
Chapter 8 Contrails and contrail cirrus 8.1 Introduction - Terminology 8.2 Contrail formation conditions 8.3 Heterogeneous nucleation on volatile aerosol and soot 8.4 Indirect effect of soot on cirrus
P1.17 SENSITIVITY OF THE RETRIEVAL OF STRATOCUMULUS CLOUD LIQUID WATER AND PRECIPITATION FLUX TO DOPPLER RADAR PARAMETERS
 P1.17 SENSITIVITY OF THE RETRIEVAL OF STRATOCUMULUS CLOUD LIQUID WATER AND PRECIPITATION FLUX TO DOPPLER RADAR PARAMETERS Yefim L. Kogan*, Zena N. Kogan, and David B. Mechem Cooperative Institute for Mesoscale
P1.17 SENSITIVITY OF THE RETRIEVAL OF STRATOCUMULUS CLOUD LIQUID WATER AND PRECIPITATION FLUX TO DOPPLER RADAR PARAMETERS Yefim L. Kogan*, Zena N. Kogan, and David B. Mechem Cooperative Institute for Mesoscale
P2.21 THE IMPACT OF CLOUD PROCESSING BY TRADE-WIND CUMULUS ON THE LIGHT SCATTERING EFFICIENCY OF AEROSOL PARTICLES
 P2.21 THE IMPACT OF CLOUD PROCESSING BY TRADE-WIND CUMULUS ON THE LIGHT SCATTERING EFFICIENCY OF AEROSOL PARTICLES Justin R. Peter, Alan M. Blyth, J ørgen B. Jensen and Donald C. Thornton 1. Introduction
P2.21 THE IMPACT OF CLOUD PROCESSING BY TRADE-WIND CUMULUS ON THE LIGHT SCATTERING EFFICIENCY OF AEROSOL PARTICLES Justin R. Peter, Alan M. Blyth, J ørgen B. Jensen and Donald C. Thornton 1. Introduction
Atmospheric Aerosols: Size Distributions and Composition
 2.1 Atmospheric Aerosols: Size Distributions and Composition The Table below (from Seinfeld and Pandis) lists typical properties of different types of atmospheric model aerosols. Note that the characteristics
2.1 Atmospheric Aerosols: Size Distributions and Composition The Table below (from Seinfeld and Pandis) lists typical properties of different types of atmospheric model aerosols. Note that the characteristics
Ammonium Sulfate: Equilibrium and Metastability Phase Diagrams from 40 to -50 C
 7462 J. Phys. Chem. B 1998, 102, 7462-7469 Ammonium Sulfate: Equilibrium and Metastability Phase Diagrams from 40 to -50 C Jun Xu, Dan Imre,* Robert McGraw, and Ignatius Tang BrookhaVen National Laboratory,
7462 J. Phys. Chem. B 1998, 102, 7462-7469 Ammonium Sulfate: Equilibrium and Metastability Phase Diagrams from 40 to -50 C Jun Xu, Dan Imre,* Robert McGraw, and Ignatius Tang BrookhaVen National Laboratory,
The Effect of Future Climate Change on Aerosols: Biogenic SOA and Inorganics
 The Effect of Future Climate Change on Aerosols: Biogenic SOA and Inorganics GCAP Phase 2 Science Team Meeting October 12, 2007 Havala O. T. Pye 1, Hong Liao 2, John Seinfeld 1, Shiliang Wu 3, Loretta
The Effect of Future Climate Change on Aerosols: Biogenic SOA and Inorganics GCAP Phase 2 Science Team Meeting October 12, 2007 Havala O. T. Pye 1, Hong Liao 2, John Seinfeld 1, Shiliang Wu 3, Loretta
New Insights into Aerosol Asymmetry Parameter
 New Insights into Aerosol Asymmetry Parameter J.A. Ogren, E. Andrews, A. McComiskey, P. Sheridan, A. Jefferson, and M. Fiebig National Oceanic and Atmospheric Administration/ Earth System Research Laboratory
New Insights into Aerosol Asymmetry Parameter J.A. Ogren, E. Andrews, A. McComiskey, P. Sheridan, A. Jefferson, and M. Fiebig National Oceanic and Atmospheric Administration/ Earth System Research Laboratory
Effect of aging on cloud nucleating properties of atmospheric aerosols
 Effect of aging on cloud nucleating properties of atmospheric aerosols Yan Ma Nanjing University of Information Science & Technology The 1st Regional GEOS-Chem Asia Meeting 2015 年 5 月 8 日 Indirect effect
Effect of aging on cloud nucleating properties of atmospheric aerosols Yan Ma Nanjing University of Information Science & Technology The 1st Regional GEOS-Chem Asia Meeting 2015 年 5 月 8 日 Indirect effect
Warm Cloud Processes. Some definitions. Two ways to make big drops: Effects of cloud condensation nuclei
 Warm Cloud Processes Dr. Christopher M. Godfrey University of North Carolina at Asheville Warm clouds lie completely below the 0 isotherm 0 o C Some definitions Liquid water content (LWC) Amount of liquid
Warm Cloud Processes Dr. Christopher M. Godfrey University of North Carolina at Asheville Warm clouds lie completely below the 0 isotherm 0 o C Some definitions Liquid water content (LWC) Amount of liquid
Role of molecular size in cloud droplet activation
 GEOPHYSICAL RESEARCH LETTERS, VOL. 36, L22801, doi:10.1029/2009gl040131, 2009 Role of molecular size in cloud droplet activation M. D. Petters, 1,2 S. M. Kreidenweis, 1 A. J. Prenni, 1 R. C. Sullivan,
GEOPHYSICAL RESEARCH LETTERS, VOL. 36, L22801, doi:10.1029/2009gl040131, 2009 Role of molecular size in cloud droplet activation M. D. Petters, 1,2 S. M. Kreidenweis, 1 A. J. Prenni, 1 R. C. Sullivan,
CLIMATE CHANGE Albedo Forcing ALBEDO FORCING
 ALBEDO FORCING Albedo forcing is the hypothesis that variations in the Earth s reflectance of solar radiation can bring about global climate change. This hypothesis is undeniable in principle; since virtually
ALBEDO FORCING Albedo forcing is the hypothesis that variations in the Earth s reflectance of solar radiation can bring about global climate change. This hypothesis is undeniable in principle; since virtually
On-line Aerosols in the Oslo Version of CAM3: Some shortcomings. Seland,
 On-line Aerosols in the Oslo Version of CAM3: Some shortcomings Trond Iversen,, Alf Kirkevåg, Øyvind Seland, Jon Egill Kristjansson, Trude Storelvmo,, Jens Debernard Norwegian Meteorological Institute
On-line Aerosols in the Oslo Version of CAM3: Some shortcomings Trond Iversen,, Alf Kirkevåg, Øyvind Seland, Jon Egill Kristjansson, Trude Storelvmo,, Jens Debernard Norwegian Meteorological Institute
Impacts of Aerosol Direct/Indirect Effect Feedbacks and Forest Shading/Turbulence on Urban Air Quality Forecasts
 Impacts of Aerosol Direct/Indirect Effect Feedbacks and Forest Shading/Turbulence on Urban Air Quality Forecasts Paul A. Makar, Ayodeji Akingunola, Craig Stroud, Junhua Zhang, Wanmin Gong, Michael D. Moran,
Impacts of Aerosol Direct/Indirect Effect Feedbacks and Forest Shading/Turbulence on Urban Air Quality Forecasts Paul A. Makar, Ayodeji Akingunola, Craig Stroud, Junhua Zhang, Wanmin Gong, Michael D. Moran,
Response to Referee 2
 Response to Referee 2 S. Metzger et al. 10 August 2018 We thank the referee for the manuscript review. Please find our pointby-point reply below. Accordingly, the revised MS will include clarifications.
Response to Referee 2 S. Metzger et al. 10 August 2018 We thank the referee for the manuscript review. Please find our pointby-point reply below. Accordingly, the revised MS will include clarifications.
Cloud processing, cloud evaporation and Angström exponent
 Atmos. Chem. Phys., 9, 71 80, 2009 Author(s) 2009. This work is distributed under the Creative Commons Attribution 3.0 License. Atmospheric Chemistry and Physics Cloud processing, cloud evaporation and
Atmos. Chem. Phys., 9, 71 80, 2009 Author(s) 2009. This work is distributed under the Creative Commons Attribution 3.0 License. Atmospheric Chemistry and Physics Cloud processing, cloud evaporation and
References: Cloud Formation. ESCI Cloud Physics and Precipitation Processes Lesson 1 - Cloud Types and Properties Dr.
 ESCI 34 - Cloud Physics and Precipitation Processes Lesson 1 - Cloud Types and Properties Dr. DeCaria References: Glossary of Meteorology, 2nd ed., American Meteorological Society A Short Course in Cloud
ESCI 34 - Cloud Physics and Precipitation Processes Lesson 1 - Cloud Types and Properties Dr. DeCaria References: Glossary of Meteorology, 2nd ed., American Meteorological Society A Short Course in Cloud
Chasing Aerosol Particles Down to Nano Sizes
 Chasing Aerosol Particles Down to Nano Sizes ERC Seminar 13 June 2013 Armin Sorooshian Chemical & Environmental Engineering Atmospheric Sciences University of Arizona Outline of Talk 1. What are aerosol
Chasing Aerosol Particles Down to Nano Sizes ERC Seminar 13 June 2013 Armin Sorooshian Chemical & Environmental Engineering Atmospheric Sciences University of Arizona Outline of Talk 1. What are aerosol
Cloud Brightening and Climate Change
 Cloud Brightening and Climate Change 89 Hannele Korhonen and Antti-Ilari Partanen Contents Definitions... 778 Aerosols and Cloud Albedo... 778 Cloud Brightening with Sea-Salt Aerosol... 779 Climate Effects
Cloud Brightening and Climate Change 89 Hannele Korhonen and Antti-Ilari Partanen Contents Definitions... 778 Aerosols and Cloud Albedo... 778 Cloud Brightening with Sea-Salt Aerosol... 779 Climate Effects
The impact of microphysical parameters, ice nucleation mode, and habit growth on the ice/liquid partitioning in mixed phase Arctic clouds
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 116,, doi:10.1029/2011jd015729, 2011 The impact of microphysical parameters, ice nucleation mode, and habit growth on the ice/liquid partitioning in mixed phase Arctic
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 116,, doi:10.1029/2011jd015729, 2011 The impact of microphysical parameters, ice nucleation mode, and habit growth on the ice/liquid partitioning in mixed phase Arctic
Haze Communication using the CAMNET and IMPROVE Archives: Case Study at Acadia National Park
 Haze Communication using the CAMNET and IMPROVE Archives: Case Study at Acadia National Park Light absorption by fine Light scattering by fine Clear line of sight in absence of fine Light extinction is
Haze Communication using the CAMNET and IMPROVE Archives: Case Study at Acadia National Park Light absorption by fine Light scattering by fine Clear line of sight in absence of fine Light extinction is
Consequences for Climate Feedback Interpretations
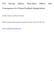 CO 2 Forcing Induces Semi-direct Effects with Consequences for Climate Feedback Interpretations Timothy Andrews and Piers M. Forster School of Earth and Environment, University of Leeds, Leeds, LS2 9JT,
CO 2 Forcing Induces Semi-direct Effects with Consequences for Climate Feedback Interpretations Timothy Andrews and Piers M. Forster School of Earth and Environment, University of Leeds, Leeds, LS2 9JT,
VOCALS REx: Aerosol Physics at the Ocean Surface On the NOAA RV Ronald H. Brown October, November 2008
 VOCALS REx: Aerosol Physics at the Ocean Surface On the NOAA RV Ronald H. Brown October, November 8 Catherine Hoyle, NOAA PMEL Derek Coffman, NOAA PMEL Tim Bates, NOAA PMEL Trish Quinn, NOAA PMEL Lelia
VOCALS REx: Aerosol Physics at the Ocean Surface On the NOAA RV Ronald H. Brown October, November 8 Catherine Hoyle, NOAA PMEL Derek Coffman, NOAA PMEL Tim Bates, NOAA PMEL Trish Quinn, NOAA PMEL Lelia
Aerosol chemistry and climate: Laboratory studies of the carbonate component of mineral dust and its reaction products
 GEOPHYSICAL RESEARCH LETTERS, VOL. 33, L13811, doi:10.1029/2006gl026386, 2006 Aerosol chemistry and climate: Laboratory studies of the carbonate component of mineral dust and its reaction products Elizabeth
GEOPHYSICAL RESEARCH LETTERS, VOL. 33, L13811, doi:10.1029/2006gl026386, 2006 Aerosol chemistry and climate: Laboratory studies of the carbonate component of mineral dust and its reaction products Elizabeth
Implementation of modules for wet and dry deposition into the ECMWF Integrated Forecast System
 Implementation of modules for wet and dry deposition into the ECMWF Integrated Forecast System Johannes Flemming (ECMWF), Vincent Huijnen (KNMI) and Luke Jones (ECMWF) Deliverable D G-RG 4.6 1 Abstract
Implementation of modules for wet and dry deposition into the ECMWF Integrated Forecast System Johannes Flemming (ECMWF), Vincent Huijnen (KNMI) and Luke Jones (ECMWF) Deliverable D G-RG 4.6 1 Abstract
STEPHEN M. SALEEBY AND WILLIAM R. COTTON
 18 JOURNAL OF APPLIED METEOROLOGY VOLUME 43 A Large-Droplet Mode and Prognostic Number Concentration of Cloud Droplets in the Colorado State University Regional Atmospheric Modeling System (RAMS). Part
18 JOURNAL OF APPLIED METEOROLOGY VOLUME 43 A Large-Droplet Mode and Prognostic Number Concentration of Cloud Droplets in the Colorado State University Regional Atmospheric Modeling System (RAMS). Part
