Preparation of stir bars for sorptive extraction using sol gel technology
|
|
|
- Maximilian Bruce
- 6 years ago
- Views:
Transcription
1 Journal of Chromatography A, 1045 (2004) Preparation of stir bars for sorptive extraction using sol gel technology Wenmin Liu, Hanwen Wang, Yafeng Guan Department of Analytical Chemistry and Micro-Instrumentation, Dalian Institute of Chemical Physics, Chinese Academy of Sciences, P.O. Box 100, Dalian , China Received 25 February 2004; received in revised form 9 June 2004; accepted 16 June 2004 Abstract A sol gel coating method for the preparation of extractive phase on bars used in sorptive microextraction is described. The extraction phase of poly(dimethylsiloxane) is partially crosslinked with the sol gel network, and the most part is physically incorporated in the network. Three aging steps at different temperatures are applied to complete the crosslinking process. Thirty-micrometer-thick coating layer is obtained by one coating process. The improved coating shows good thermal stability up to 300 C. Spiked aqueous samples containing n-alkanes, polycyclic aromatic hydrocarbons and organophosphorus pesticides were analyzed by using the sorptive bars and GC. The results demonstrate that it is suitable for both aploar and polar analytes. The detection limit for chrysene is 7.44 ng/l, 0.74 ng/l for C 19 and 0.9 ng/l for phorate. The extraction equilibration can be reached in less than 15 min by supersonic extraction with the bars of 30 m coating layer Elsevier B.V. All rights reserved. Keywords: Sol gel; Stir bar sorptive extraction; Supersonic sorptive extraction; Extraction methods; Alkanes; Polynuclear aromatic hydrocrabons; Pesticides; Organophosphorus compounds 1. Introduction Sorptive extraction (SE) provides a simple, effective, and solvent-free sample preparation technique for selective adsorption and enrichment of target compounds in sample matrix [1,2]. Many modes of SE have been developed to date, such as fiber-se [3 6], in-tube SE [7 10] and stir bar sorptive extraction (SBSE) [11 15], that have been applied to a great variety of matrices: gas [3,4], liquid [5 7] and solid [16 19], and extended to a wide range of analytes from volatile to nonvolatile compounds [3 9,16 19]. Because the recovery of an analyte from the sample is determined by the partitioning coefficient and the phase ratio between the extraction phase and the aqueous sample, low recoveries are obtained for compounds with low coefficients, which is improved by SBSE technique using stir bars of 10 mm and 40 mm length coated with L of poly(dimethyl siloxane) (PDMS) [11 15]. The phase ratio of SBSE is about Corresponding author. Tel.: ; fax: address: kfguan@mail.dlptt.ln.cn (Y. Guan) times lower than that in other modes of solid phase microextraction, resulting in much higher recoveries, especially for volatile compounds with higher octanol water partition coefficients (K o/w ) [12]. In SE, the coating technology of extraction phase is vitally important to the performance of the device. To date, static coating technology [20,21] and sol gel coating technology [22,23] are commonly used in SE. Compared to static coating technology, sol gel technology may be much more suitable for the preparation of thick film. Because of strong adhesion between the coating and the surface of bare fused silica or glass by chemical bonding, the phase exhibits low bleeding, good repeatability and long lifetime in SE [24,25]. In conventional SBSE, a slice of special PDMS tubing, which covers on a glass tube with a magnetic core, has been used as the extraction phase. To date there is no satisfaction method for the preparation of SBSE coating directly on the glass bar. In this paper, an improved sol gel method for the preparation of coating on the bars was investigated and two modes of extraction were examined. One mode is SBSE and the other is supersonication sorptive extraction (SSE) /$ see front matter 2004 Elsevier B.V. All rights reserved. doi: /j.chroma
2 16 W. Liu et al. / J. Chromatogr. A 1045 (2004) Experimental 2.1. Equipment A Trace GC 2000 capillary GC system (ThermoQuest CE Instruments, USA) equipped with a capillary split/splitless injector and flame ionization detection (FID) system was used to analyze n-alkanes and polycyclic aromatic hydrocarbons (PAHs). The analysis of organophosphorus pesticides (OPPs) was performed on a CP 3800 GC system (Varian, USA) system equipped with thermal specified detector. A home-made thermal desorption system (TDS), consists of a quartz liner and heaters, two gas flow controllers, temperature control units, and a sample transfer line. A 90 mm long stainless steel cylinder of 6.7 mm i.d. and 12 mm o.d. is used to hold the desorption liner and the transfer line. One of the gas flow controllers provides purge gas for the desorption from the top of the liner, and another flow controller provides sweep gas from the bottom of the liner to avoid diffusion and adsorption of the sample outside the quartz liner. The transfer line was inserted directly through the injector septum into the GC pre-column about 20 cm deep. A JEM-1200EX scanning electron microscope (JEOL, Japan) was used to examine the sol gel PDMS coatings (including the surface and thickness of the sol gel coating). A SK-1 quick blend machine (Zhengji Machine Co., Jiangsu, China) was used for thorough mixing of various ingredients in the sol solution. SB3200 ultrasonic bath (Shanghai Branson, China) was used in SSE. The bared glass bar (30 mm 1.8 mm o.d.) used for the preparation of sorptive stir bar contains an iron bar inside the glass tube, and the one used for supersonic sorptive bar contains no iron bar. The water used in this experiment was Wahaha purified water (Wahaha, Hangzhou, China) Reagents Hydroxy-terminated PDMS was obtained from Keguang New Material Co. (Nantong, Jiangsu Province, China). Methyltrimethoxane (MTMS) was purchased from Danyang Organic Silane Co. (Jiangsu Province, China). Poly(methylhydrosiloxane) (PMHS) was obtained from the Chemical Plant of Wuhan University (Wuhan, China). Trifluoroacetic acid (TFA) was purchased from Shanghai Chemical Plant (Chinese Medicine Group, China). HPLC-grade methylene chloride was purchased from Tedia (USA). Chromatography grade n-alkanes (C ) were from National Research Center for CRMs (Beijing, China). The PAH mixture and OPPs were purchased from J&K Acros Organics (Beijing, China) Preparation of sorptive bars Pretreatment of bars The bars were sequentially cleaned by water and methylene chloride, followed by 1 mol/l NaOH and 0.1 mol/l HCl for 8 12 h, respectively. After being washed by water, the bars were purged with N 2 at 120 Cina40mm 10 mm i.d. stainless steel tube Preparation of sol solution The sol solution was prepared as follows: 200 mg of hydroxy-terminated PDMS was thoroughly dissolved in 300 L methylene chloride; then 50 L MTMS and 50 mg PMHS were added. The mixture was vortexed thoroughly by quick blend machine. After this, 50 L TFA (containing 5% water) was added, and vortexed quickly until the mixture was a clear solution which was used for bar coating. The bars were immersed into the solution for a given time that controlled the thickness of coating. For coatings over 30 m, the above coating procedure should be repeated with fresh sol. The coated bars were then placed into a vacuum desiccator for 8 h for coating gelation Conditioning of the bars The sol gel PDMS coated bars were conditioned in the stainless steel tube described in Section under N 2 purge. The tube was put into a GC oven, and was heated under temperature programming. The oven temperature is from 40 Cto120 Cat1 C/min, held for 180 min, then to 240 C at the same ramp and held for 180 min, and finally to 300 Cat1 C/min and held for 240 min. The bars were then extracted by boiling methylene chloride for 4 h. Before being used for extraction, the bars were purged under a N 2 stream at 300 C for 2 h Extraction mode Stirring extraction and supersonic extraction modes were studied. For SBSE the stir bar was in a 50 ml sample vial at room temperature. For SSE the sorptive bar was in 50 ml sample vial, which was put into an ultrasonic bath Thermal desorption of extracted analytes in thermal desorption systems Thermal desorption of the extracted analytes from the sol gel bars was performed in the laboratory-made desorption unit. Brief drying of the bars with tissue paper before desorption is recommended. Desorption temperature was 260 C and 280 C, respectively, for n-alkanes and PAHs. The set temperature of desorption unit was reached in 2 min and held for 5 min for complete desorption. The thermally desorbed analytes were focused at the head of the analytical column, which was kept at 40 C during the thermal desorption process Preparation of aqueous standards A 10- L portion of the stock solutions of all PAHs was diluted with methanol in a 10-mL volumetric flask at room
3 W. Liu et al. / J. Chromatogr. A 1045 (2004) Table 1 Contents of PAHs stock solution and diluted methanol solution Component Content of stock solution ( g/ml) Content of diluted methanol solution (ng/ml) Acenaphthene Acenaphthylene Anthracene Benz[a]anthracene Benzo[a]pyrene Benzo[b]fluoranthene Benzo[ghi]perylene Benzo[k]fluroanthene Chrysene Dibenz[a,h]anthracene Fluoranthene Fluorene Indeno[1,2,3-cd]pyrene Naphthalene Phenanthrene Pyrene Content of spiked water sample solution (pg/ml) temperature. The content of the stock solution and diluted methanol solution was showed in Table 1.A50 L portion of the diluted solution was diluted further with 50 ml purified water. Stock solution of n-alkanes was prepared by dissolving 2.00 mg of each compound in a 100-mL volumetric flask methanol at room temperature to give a 20 g/ml solution. A1- L portion of which was dissolved in 50 ml purified water to give 400 pg/ml n-alkanes sample. A 1-mL portion of 10 g/ml OPPs was diluted to 1 g/ml by methanol, which was then diluted further to 200 pg/l spiked water sample Chromatographic conditions Conditions for the analysis of n-alkanes and PAHs A 5 m 0.53 mm i.d. deactivated capillary column was connected to a 30 m 0.53 mm i.d. 0.6 m SE-54 analytical column by a press fit connector. The temperatures of injector and detector were all 300 C for n-alkanes analysis, and were 260 C and 300 C, respectively, for PAHs analysis. Hydrogen was used as the carrier gas at a linear velocity of 30 cm/s. The injector was maintained in the splitless mode. Column temperature is as follows: for analysis of n-alkanes, 30 C for 5 min, then to 280 C at a rate of 15 C/min; For PAHs analysis, 40 C for 5 min, program to 120 Cata rate of 30 C/min, then to 150 C at a rate of 15 C/min, and finally to 280 C at a rate of 8 C/min. The column temperature for the bar test is the same as that of n-alkanes Conditions for the analysis of OPPs A 5 m 0.53 mm i.d. deactivated capillary column was connected to a 30 m 0.25 mm i.d m SE-54 analytical column by a press fit connector. The temperatures of injector and detector were 260 C and 300 C, respectively. Nitrogen was used as the carrier gas at a linear velocity of 13.6 cm/s. The injector was maintained at splitless mode. Column temperature ramp is as follows: 40 C for 5 min, then to 110 C at a rate of 30 C/min, and finally to 260 C at a rate of 10 C and held for 20 min. 3. Results and discussion Because the coating layer of sorptive stir bar/supersonic sorptive bar is rather thick, it is very important to have a stable and long lasting stationary phase having a low bleeding at high temperature. Conventional coating technologies used in the preparation of fiber SPME, in-tube SPME, and GC columns are not fit for sorptive stir bar/supersonic sorptive bar. Sol gel approach to column technology for analytical microseparations is highlighted in recent years. This technology can be applied to a wide range of microseparation and sample preparation techniques including capillary gas chromatography, solid phase microextraction, capillary electrophoresis, and capillary electrochromatography. The main obstacle in the creation of film by sol gel method is cracking. The use of alkyl or aryl derivatives of tetraalkoxysilanes as precursors and drying control chemical additives (DCCAs) can release the capillary stress generated during drying of the coated surface The sol gel chemistry involved in the creation of coating layer The sol gel process starts with the hydrolysis of the sol gel precursor, followed by polycondensation of hydrolyzed products into a sol gel network. At the same time the hydroxyterminated PDMS is partially bonded to the network because of the large molecular size (M W 40,000), which prohibits complete bonding of OH-PDMS. The sol gel polymer is bonded to the silanol groups on the
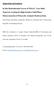
4 18 W. Liu et al. / J. Chromatogr. A 1045 (2004) Fig. 2. The scanning electron microscopy image of the surface structures of a sol gel PDMS coating on the wall of glass bar. Magnification 30,000. Fig. 1. (A) Physical incorporation of hydroxy-teminated PDMS into the sol gel network. (B) Deactivation of sol gel network with PMHS and its incorporation into the sol gel network. glass surface of bars. The major part of OH-PDMS is physically incorporated and entangled with sol gel network, as depicted in Fig. 1A. The mechanism of the coating process, therefore, is not the same as reported before [21,22,27]. In order to increase the surface area and expose most of the surface silanol groups, the bars are treated with 1 M NaOH and 0.1 M HCl [29]. The precursor in this sol gel process is much less than that of reported previously [22,26,28], in which a compact sol gel is created and vortexed thoroughly before adding catalyst TFA, resulting molecular level mixing. A loose sol gel network is formed because of the incorporation of more OH-PDMS after adding TFA. The pore diameters are relatively large and the degree of cracking is much less during the drying process. The role of PMHS is deactivation of surface and network. The active hydrogen atoms react with silanol groups at elevated temperatures to end-cap remaining silanol groups. Post-thermal conditioning of coating layer leads to Table 2 Peak height repeatability data for analytes and their detection limits Analytes Peak height repeatability (n = 5) Detection limit, S/N = 3 (pg/ml) Mean peak height RSD (%) C C C C C C C C Acenaphthene Acenaphthylene Anthracene Benz[a]anthracene Benzo[a]pyrene Benzo[b]fluoranthene Benzo[ghi]perylene Benzo[k]fluroanthene Chrysene Dibenz[a,h]anthracene Fluoranthene Fluorene Indeno[1,2,3-cd]pyrene Naphthalene Phenanthrene Pyrene
5 W. Liu et al. / J. Chromatogr. A 1045 (2004) Fig. 3. Chromatograms of spiked n-alkanes aqueous sample. (A) SSE cgc FID; (B) SBSE cgc FID. Conditions see text. The concentrations of all analytes were 400 ng/l.
6 20 W. Liu et al. / J. Chromatogr. A 1045 (2004) Fig. 4. Chromatogram of PAHs from the extraction of sorptive bar. Conditions: column conditions as in Fig. 3; injector: 260 C; detector: 300 C; column temperature: 40 C for 5 min, programmed to 120 C at a rate of 30 C/min, then to 150 Cat15 C/min, and finally to 280 Cat8 C/min. Peak identities: 1, naphthalene; 2, acenaphthylene; 3, acenaphthene; 4, fluorene; 5, phenanthrene; 6, anthracene; 7, fluoranthene; 8, pyrene; 9, benzo[a]pyrene; 10, chrysene; 11, benzo[b]fluoranthene; 12, benzo[k]fluroanthene; 13, benz[a]anthracene; 14, indeno[1,2,3-cd]pyrene; 15, dibenz[a,h]anthracene; 16, benzo[ghi]perylene. The concentrations of the analytes were shown in Table 1. deactivation of the bars. In the sol gel network PMHS has very similar chemical structure to that of PDMS, its incorporation in the coating structure as shown in Fig. 1B has a little effect on the characteristics of PDMS. The methyl group in the precursor of MTMS also plays the similar role as PMHS after polycondensation of hydrolysis [22]. Appropriate amounts of PMHS and OH-terminated PDMS in the solid phase improve the selectivity for polar compounds. Because of the thick coating layer on sorptive bars, rather high temperature for thermal desorption has to be applied especially for apolar analytes. To achieve high thermal stability, the post-coating thermal conditioning is optimized to guarantee sufficient conditioning and avoid cracking of the coating layer. Three temperature ramps at a rate of 1 C/min and maintaining at 120 C, 240 C, 300 C for several hours was finally adopted. Fig. 2 shows the coating layer structure by scanning electron microscopy (SEM). It shows that the sol gel coating layer possesses porous structure, which enhances the surface area for solute solid phase interactions and the extraction rate. The cross-section SEM image of the coated bar shows that the sol gel PDMS coating layer is about 30 m in thickness, and is very uniform. The thermal stability of the coating of the bars was examined by thermal desorption in the TDS unit at 300 C for Table 3 The peak height repeatability of two extraction modes Extraction mode RSD (%) (n = 5) C 12 C 13 C 14 C 15 C 16 C 17 C 18 C 19 Supersonic extraction for 10 min Stir bar sorptive extraction for 90 min
7 W. Liu et al. / J. Chromatogr. A 1045 (2004) Table 4 The precision and detection limits of OPPs by SBSE GC TSD Analyze Peak area repeatability (n = 5) Detection limit, S/N = 3 (pg/ml) Mean peak area RSD (%) Methamidophos Dichlorvos Acephate Monocrotophos Phorate Parathionmethyl Malathion Fenitrothion Fenthion a Chlorpyrifos a Parathion a Methidathion Triazophos Ethaion a These three peaks overlapped. 5 min, followed by a capillary (cgc) run and FID. No ghost peak was observed in the chromatogram (chromatogram not shown) Extraction modes In our work, two modes of extraction were applied: SBSE and SSE. In supersonic extraction mode, the intensive supersonic vibration destroys the boundary layer between extraction phase and sample matrix. The control step of extraction rate, therefore, becomes the mass transfer rate from the surface to the body of the extraction phase. The lower the diffusion coefficient, or the thicker the extraction phase, the longer the extraction time will be. Table 2 shows the results of run-to-run repeatability in peak height, retention time and the detection limits by using sol gel sorptive stir bar and supersonic sorptive bar. The detection limit of n-alkanes is 0.74 ng/l for nc 19, and of PAHs 0.19 ng/l for chrysene. Fig. 3A and B shows the GC chromatogram of n-alkanes by the extraction of sol gel coated sorptive stir bar and supersonic sorptive bar. The extraction time for the stir bar and the sorptive bar is 90 min and 10 min, respectively. It can be seen from the two figures that peak heights in SSE mode are higher than those in SBSE, implying that the extraction efficiency of supersonic extraction is higher than that of stir bar sorptive extraction. The boundary layer in SBSE limited the mass transfer rates of the analytes with relative large molecular weight. Table 3 compares the repeatability data of peak heights by the two extraction modes, proving that both extraction modes give satisfaction results. The carryover of the extraction bar was also examined by second TDS of the same bar followed by chromatographic analysis. We found no peaks in the chromatogram (chromatogram not shown), demonstrating complete desorption of analytes. Fig. 4 represents the GC chromatogram of PAHs by the extraction of sol gel coated sorptive stir bar for 120 min. Supersonic sorptive extraction was also tested, and the same phenomena as those of n-alkanes are also observed. It proved that for the stable compounds being tested, supersonic extraction mode is prior to SBSE. No carryover problem is found at the desorption temperature of 280 C for compounds tested. Polar analytes were also tested in our study. The OPPs spiked water sample was extracted by stir bar for 120 min, followed by TDS-cGC analysis. The chromatogram of OPPs was shown in Fig. 5, and results were shown in Table 4. The detection limit of OPPs are in the pg/ml range, and is 0.9 pg/ml for phorate. Fig. 5. Chromatogram of organophosphorus pesticides by SBSE cgc TSD. Conditions see text. Peak identities: 1, methamidophos; 2, dichlorvos; 3, acephate; 4, momocrotophos; 5, phorate; 6, parathion-methyl; 7, malathion; 8, fenitrothion; 9, fenthion; 10, chlorpyrifos; 11, parathion; 12, methidathion; 13, triazophos; 14, ethaion; concentration of each analyze was at 200 pg/ml. 4. Conclusion A sol gel method is investigated for the preparation of extraction phase on bars used in SBSE and SSE. The extraction solid phase (hydroxy-terminated PDMS) not only
8 22 W. Liu et al. / J. Chromatogr. A 1045 (2004) crosslinked with the sol gel network created by the hydrolysis products of the sol gel precursor but entangled within it. The aging process for the thick coating was also developed to avoid cracking of the film. Supersonic extraction mode was investigated and compared to SBSE mode, showing that the former has much higher mass transfer rate and shorter equilibration time, and higher extraction efficiency. References [1] C.L. Arthur, J. Pawliszyn, Anal. Chem. 62 (1987) [2] H. Lord, J. Pawliszyn, J. Chromatogr. A 885 (2000) 153. [3] J.J. Langenfeld, S.B. Hawthorne, D.J. Miller, Anal. Chem. 68 (1996) 144. [4] R.J. Bartelt, Anal. Chem. 69 (1997) 364. [5] K. Jinno, M. Kawazoe, Chromatographia 52 (2000) 309. [6] C. Rivasseau, G. Vanhoenacker, P. Sandra, M.C. Hennion, J. Microcol. Sep. 12 (2000) 323. [7] L. Nardi, Am. Lab. 34 (2002) 30. [8] Y. Saito, M. Kawazoe, M. Hayashida, K. Jinno, Analyst 125 (2000) 807. [9] Y. Gou, R. Eisert, J. Pawliszyn, J. Chromatogr. A 873 (2000) 137. [10] H.W. Wang, W.M. Liu, Y.F. Guan, LC:GC North Am. 22 (2004) 16. [11] E. Baltussen, P. Sandra, F. David, C. Cramers, J. Microcol. Sep. 11 (1999) 737. [12] D. Frank, B. Tienpont, P. Sandra, LC:GC North Am. 21 (2003) 1. [13] J. Vercauteren, C. Peres, C. Devos, P. Sandra, F. Vanhaecke, L. Moens, Anal. Chem. 73 (2001) [14] P. Popp, C. Bauer, L. Wennrich, Anal. Chim. Acta 436 (2001) 1. [15] G.J. Andreas, H.H.L. Lauer, T. Heideman, P. Sandra, J. High Resolut. Chromatogr. 23 (2000) 644. [16] J.J. Jimenez, F.J. Lopez, O. Cepria, F. Hernandez, J. Chromatogr. A 829 (1998) 269. [17] M. Moder, P. Popp, R. Eisert, J. Pawliszyn, Fresenius J. Anal. Chem. 363 (1999) 680. [18] A.A. Boyd-Boland, J. Pawliszyn, Analyst 121 (1996) 929. [19] R. Hu, B. Hennion, L. Urruty, M. Montury, Food Addit. Contam. 16 (1999) 111. [20] C. Haberhauer-Troyer, M. Crnoja, E. Rosenberg, Fresenius J. Anal. Chem. 366 (2000) 329. [21] B.W. Wright, P.A. Peaden, M.L. Lee, T.J. Stark, J. Chromatogr. 248 (1982) 17. [22] S. Chong, A. Malik, Anal. Chem. 69 (1997) [23] T.P. Gbatu, Anal. Chim. Acta 402 (1999) 67. [24] S.A. Rodriguez, L.A. Colon, Anal. Chim. Acta 397 (1999) 207. [25] A.M. Siouffi, J. Chromatogr. A 1000 (2003) 801. [26] Z.R. Zeng, W.L. Qiu, Z.F. Huang, Anal. Chem. 73 (2001) [27] C. Shende, A. Kabir, E. Townsend, A. Malik, Anal. Chem. 75 (2003) [28] A. Malik, Electrophoresis 23 (2002) [29] Y. Guo, L.A. Colon, Anal. Chem. 67 (1995) 2511.
AppNote 2/2000. Stir Bar Sorptive Extraction (SBSE) applied to Environmental Aqueous Samples
 AppNote 2/2 Stir Bar Sorptive Extraction (SBSE) applied to Environmental Aqueous Samples Pat Sandra Department of Organic Chemistry, University of Gent, Krijgslaan 281 S4, B-9 Gent, Belgium Erik Baltussen
AppNote 2/2 Stir Bar Sorptive Extraction (SBSE) applied to Environmental Aqueous Samples Pat Sandra Department of Organic Chemistry, University of Gent, Krijgslaan 281 S4, B-9 Gent, Belgium Erik Baltussen
anthracene Figure 1: Structures of Selected Polyaromatic Hydrocarbons (PAHs)
 Stir Bar Sorptive Extraction Analysis of PAHs in Aqueous Solution: Using a polymer based technique to pre-concentrate polyaromatic hydrocarbons for analysis by GC-MS. Background: Polyaromatic hydrocarbons
Stir Bar Sorptive Extraction Analysis of PAHs in Aqueous Solution: Using a polymer based technique to pre-concentrate polyaromatic hydrocarbons for analysis by GC-MS. Background: Polyaromatic hydrocarbons
AppNote 2/2000. Stir Bar Sorptive Extraction (SBSE) applied to En vi ron men tal Aqueous Samples INTRODUCTION
 AppNote 2/2 Stir Bar Sorptive Extraction (SBSE) applied to En vi ron men tal Aqueous Samples Pat Sandra Department of Organic Chemistry, University of Gent, Krijgslaan 281 S4, B-9 Gent, Belgium Erik Baltussen
AppNote 2/2 Stir Bar Sorptive Extraction (SBSE) applied to En vi ron men tal Aqueous Samples Pat Sandra Department of Organic Chemistry, University of Gent, Krijgslaan 281 S4, B-9 Gent, Belgium Erik Baltussen
In-Tube Solid-Phase Microextraction and On-Line Coupling with High-Resolution GC
 In-Tube Solid-Phase Microextraction and On-Line Coupling with High-Resolution GC Hanwen Wang, Wenmin Liu and Yafeng Guan, Dalian Institute of Chemical Physics, Dalian, China. The guest authors validated
In-Tube Solid-Phase Microextraction and On-Line Coupling with High-Resolution GC Hanwen Wang, Wenmin Liu and Yafeng Guan, Dalian Institute of Chemical Physics, Dalian, China. The guest authors validated
Screening of Pesticide Residues in Water by Sequential Stir Bar Sorptive Extraction-Thermal Desorption with GC/MS
 Screening of Pesticide Residues in Water by Sequential Stir Bar Sorptive Extraction-Thermal Desorption with GC/MS Application Note Food Authors Nobuo Ochiai and Kikuo Sasamoto GERSTEL K.K. 2-13-18 Nakane
Screening of Pesticide Residues in Water by Sequential Stir Bar Sorptive Extraction-Thermal Desorption with GC/MS Application Note Food Authors Nobuo Ochiai and Kikuo Sasamoto GERSTEL K.K. 2-13-18 Nakane
Selection of a Capillary
 Selection of a Capillary GC Column - Series 3 Mark Sinnott Application Engineer March 19, 2009 Page 1 Typical Gas Chromatographic System Mol-Sieve Traps Fixed Restrictors Regulators Injection Port Detector
Selection of a Capillary GC Column - Series 3 Mark Sinnott Application Engineer March 19, 2009 Page 1 Typical Gas Chromatographic System Mol-Sieve Traps Fixed Restrictors Regulators Injection Port Detector
Accelerated Solvent Extraction GC-MS Analysis and Detection of Polycyclic Aromatic Hydrocarbons in Soil
 Accelerated Solvent Extraction GC-MS Analysis and Detection of Polycyclic Aromatic Hydrocarbons in Soil Che Jinshui, 1 Deng Guifeng, 1 Liang Lina, 1 and Aaron Kettle, 2 1 Thermo Fisher Scientific (China)
Accelerated Solvent Extraction GC-MS Analysis and Detection of Polycyclic Aromatic Hydrocarbons in Soil Che Jinshui, 1 Deng Guifeng, 1 Liang Lina, 1 and Aaron Kettle, 2 1 Thermo Fisher Scientific (China)
Selection of a Capillary GC Column
 Selection of a Capillary GC Column Mark Sinnott Application Engineer March 13, 2008 Page 1 Typical Gas Chromatographic System Mol-Sieve Traps Fixed Restrictors Regulators Injection Port Detector Electrometer
Selection of a Capillary GC Column Mark Sinnott Application Engineer March 13, 2008 Page 1 Typical Gas Chromatographic System Mol-Sieve Traps Fixed Restrictors Regulators Injection Port Detector Electrometer
Volatile organic compounds (VOCs):
 Volatile organic compounds (VOCs): Organic chemicals with a high vapour pressure at room temperature. High vapour pressure results from a low boiling point. The World Health Organization (WHO) defined
Volatile organic compounds (VOCs): Organic chemicals with a high vapour pressure at room temperature. High vapour pressure results from a low boiling point. The World Health Organization (WHO) defined
METHOD 8100 POLYNUCLEAR AROMATIC HYDROCARBONS
 METHOD 8100 POLYNUCLEAR AROMATIC HYDROCARBONS 1.0 SCOPE AND APPLICATION 1.1 Method 8100 is used to determine the concentration of certain polynuclear aromatic hydrocarbons (PAH). Table 1 indicates compounds
METHOD 8100 POLYNUCLEAR AROMATIC HYDROCARBONS 1.0 SCOPE AND APPLICATION 1.1 Method 8100 is used to determine the concentration of certain polynuclear aromatic hydrocarbons (PAH). Table 1 indicates compounds
OPTIMISATION OF SOLID PHASE MICROEXTRACTION (SPME) CONDITIONS FOR HEADSPACE ANALYSIS OF ORGANOPHOSPHATE PESTICIDES IN WHOLE BLOOD
 OPTIMISATION OF SOLID PHASE MICROEXTRACTION (SPME) CONDITIONS FOR HEADSPACE ANALYSIS OF ORGANOPHOSPHATE PESTICIDES IN WHOLE BLOOD Kamarruddin ASRI 1, Robert A. ANDERSON 2 1 Department of Chemistry, Jalan
OPTIMISATION OF SOLID PHASE MICROEXTRACTION (SPME) CONDITIONS FOR HEADSPACE ANALYSIS OF ORGANOPHOSPHATE PESTICIDES IN WHOLE BLOOD Kamarruddin ASRI 1, Robert A. ANDERSON 2 1 Department of Chemistry, Jalan
The Design of High Temperature Capillary Gas Chromatography Columns Based on Polydimethylsiloxane
 The Design of High Temperature Capillary Gas Chromatography Columns Based on Polydimethylsiloxane Jarl Snider, D. J. Hotnisky, Kristi Sellers, Dinesh V. Patwardhan Ph. D. Restek Corporation www.restekcorp.com
The Design of High Temperature Capillary Gas Chromatography Columns Based on Polydimethylsiloxane Jarl Snider, D. J. Hotnisky, Kristi Sellers, Dinesh V. Patwardhan Ph. D. Restek Corporation www.restekcorp.com
TechNote 5/2002 KEYWORDS ABSTRACT. SBSE, Twister, Thermal Desorption
 TechNote 5/2002 Design, Performance, and Applicability of a Newly Developed Sample Introduction System for Use with Stir Bar Sorptive Extraction (SBSE) Andreas Hoffmann, Bita Kolahgar, Christian Heinz,
TechNote 5/2002 Design, Performance, and Applicability of a Newly Developed Sample Introduction System for Use with Stir Bar Sorptive Extraction (SBSE) Andreas Hoffmann, Bita Kolahgar, Christian Heinz,
Chapter 27: Gas Chromatography
 Chapter 27: Gas Chromatography Gas Chromatography Mobile phase (carrier gas): gas (He, N 2, H 2 ) - do not interact with analytes - only transport the analyte through the column Analyte: volatile liquid
Chapter 27: Gas Chromatography Gas Chromatography Mobile phase (carrier gas): gas (He, N 2, H 2 ) - do not interact with analytes - only transport the analyte through the column Analyte: volatile liquid
Facility: Mansfield Revision 3. Title: PAHs by SPME Page 1 of 11
 Title: PAHs by SPME Page 1 of 11 Determination of Parent and Alkyl Polycyclic Aromatics in sediment Pore Water Using Solid-Phase Microextraction and Gas Chromatography/Mass Spectrometry in Selected Ion
Title: PAHs by SPME Page 1 of 11 Determination of Parent and Alkyl Polycyclic Aromatics in sediment Pore Water Using Solid-Phase Microextraction and Gas Chromatography/Mass Spectrometry in Selected Ion
Supporting information
 Supporting information In Situ Hydrothermal Grown of TiO 2 @C Core-Shell Nanowire Coating for High Sensitive Solid Phase Microextraction of Polycyclic Aromatic Hydrocarbons Fuxin Wang, Juan Zheng, Junlang
Supporting information In Situ Hydrothermal Grown of TiO 2 @C Core-Shell Nanowire Coating for High Sensitive Solid Phase Microextraction of Polycyclic Aromatic Hydrocarbons Fuxin Wang, Juan Zheng, Junlang
Determination of Pesticides in Aqueous Samples by On-Line Coupling Solid-Phase Extraction to Gas Chromatography with At-Column Concentrating Interface
 Application Note No. 033 Determination of Pesticides in Aqueous Samples by On-Line Coupling Solid-Phase Extraction to Gas Chromatography with At-Column Concentrating Interface Ryoichi Sasano*, Takayuki
Application Note No. 033 Determination of Pesticides in Aqueous Samples by On-Line Coupling Solid-Phase Extraction to Gas Chromatography with At-Column Concentrating Interface Ryoichi Sasano*, Takayuki
Sensitive and rapid determination of polycyclic aromatic hydrocarbons in tap water
 APPLICATION NOTE 70923 Sensitive and rapid determination of polycyclic aromatic hydrocarbons in tap water Authors Chen Jing, Dai Zhenyu, Xu Qun, and Liang Lina, Thermo Fisher Scientific, Shanghai, People
APPLICATION NOTE 70923 Sensitive and rapid determination of polycyclic aromatic hydrocarbons in tap water Authors Chen Jing, Dai Zhenyu, Xu Qun, and Liang Lina, Thermo Fisher Scientific, Shanghai, People
The Suite for Environmental GC Analysis
 The Suite for Environmental GC Analysis SGE Environmental GC Columns Performance Selectivity Delivery Promise www.sge.com SGE Environmental GC Columns The Suite for Environmental GC Analysis SGE GC Columns
The Suite for Environmental GC Analysis SGE Environmental GC Columns Performance Selectivity Delivery Promise www.sge.com SGE Environmental GC Columns The Suite for Environmental GC Analysis SGE GC Columns
Rapid Determination Method for 12 Pyrethroid Pesticide Residues in Tea by Stir Bar Sorptive Extraction-Thermal Desorption-Gas Chromatography*
 Available online at www.sciencedirect.com Physics Procedia 25 (2012 ) 1776 1780 2012 International Conference on Solid State Devices and Materials Science Rapid Determination Method for 12 Pyrethroid Pesticide
Available online at www.sciencedirect.com Physics Procedia 25 (2012 ) 1776 1780 2012 International Conference on Solid State Devices and Materials Science Rapid Determination Method for 12 Pyrethroid Pesticide
Determination of Polycyclic Aromatic Hydrocarbons (PAHs) in Seafood Using GC/MS
 Determination of Polycyclic Aromatic Hydrocarbons (PAHs) in Seafood Using GC/MS UCT Part Numbers: ECQUUS2-MP (4 g of muffled anh. MgSO 4 and 2 g of NaCl) ECPAHFR50CT (50 ml centrifuge tubes, PAHs removed)
Determination of Polycyclic Aromatic Hydrocarbons (PAHs) in Seafood Using GC/MS UCT Part Numbers: ECQUUS2-MP (4 g of muffled anh. MgSO 4 and 2 g of NaCl) ECPAHFR50CT (50 ml centrifuge tubes, PAHs removed)
Determination of Volatile Aromatic Compounds in Soil by Manual SPME and Agilent 5975T LTM GC/MSD
 Determination of Volatile Aromatic Compounds in Soil by Manual SPME and Agilent 5975T LTM GC/MSD Application Note Environmental Author Suli Zhao, Andy Zhai Agilent Technologies Co., Ltd. 412 Yinglun Road
Determination of Volatile Aromatic Compounds in Soil by Manual SPME and Agilent 5975T LTM GC/MSD Application Note Environmental Author Suli Zhao, Andy Zhai Agilent Technologies Co., Ltd. 412 Yinglun Road
Journal of Chromatography A, 1114 (2006)
 Journal of Chromatography A, 1114 (2006) 234 238 Fast and sensitive environmental analysis utilizing microextraction in packed syringe online with gas chromatography mass spectrometry Determination of
Journal of Chromatography A, 1114 (2006) 234 238 Fast and sensitive environmental analysis utilizing microextraction in packed syringe online with gas chromatography mass spectrometry Determination of
Choosing the Correct GC Column Dimensions and Stationary Phase
 Choosing the Correct GC Column Dimensions and Stationary Phase Daron Decker Chromatography Technical Specialist Page 1 Nothing is useless it can always serve as a bad example Custom Column: 150 m x 250
Choosing the Correct GC Column Dimensions and Stationary Phase Daron Decker Chromatography Technical Specialist Page 1 Nothing is useless it can always serve as a bad example Custom Column: 150 m x 250
637. Thiamethoxam. HPLC method
 637. Thiamethoxam HPLC method CIPAC Collaborative Trial according to CIPAC Information Sheet N o 293 Dr. Sven Adolph Syngenta Crop Protection Münchwilen AG CH-4333 Münchwilen Switzerland May 212 page 1
637. Thiamethoxam HPLC method CIPAC Collaborative Trial according to CIPAC Information Sheet N o 293 Dr. Sven Adolph Syngenta Crop Protection Münchwilen AG CH-4333 Münchwilen Switzerland May 212 page 1
Determination of Volatile Substances Proof of Food Adulteration
 ANALYSIS OF FOOD AND NATURAL PRODUCTS LABORATORY EXERCISE Determination of Volatile Substances Proof of Food Adulteration (method: gas chromatography with mass spectrometric detection) Exercise guarantor:
ANALYSIS OF FOOD AND NATURAL PRODUCTS LABORATORY EXERCISE Determination of Volatile Substances Proof of Food Adulteration (method: gas chromatography with mass spectrometric detection) Exercise guarantor:
Multi-residue Analysis for PAHs, PCBs and OCPs on Agilent J&W FactorFour VF-Xms
 Multi-residue Analysis for PAHs, PCBs and OCPs on Agilent J&W FactorFour VF-Xms Application Note Author Laura Provoost Agilent Technologies, Inc. Introduction In multi-residue analysis different groups
Multi-residue Analysis for PAHs, PCBs and OCPs on Agilent J&W FactorFour VF-Xms Application Note Author Laura Provoost Agilent Technologies, Inc. Introduction In multi-residue analysis different groups
Application Note GCMS-09 Evaluation of Rapid Extraction and Analysis Techniques for Polycyclic Aromatic Hydrocarbons (PAHs) in Seafood by GC/MS/MS
 Application Note GCMS-09 Evaluation of Rapid Extraction and Analysis Techniques for Polycyclic Aromatic Hydrocarbons (PAHs) in Seafood by GC/MS/MS Abstract Rapid sample preparation methods for the analysis
Application Note GCMS-09 Evaluation of Rapid Extraction and Analysis Techniques for Polycyclic Aromatic Hydrocarbons (PAHs) in Seafood by GC/MS/MS Abstract Rapid sample preparation methods for the analysis
Application Note # CA
 Application Note # CA-274101 Evaluation of Rapid Extraction and Analysis Techniques for Polycyclic Aromatic Hydrocarbons (PAHs) in Seafood by GC/MS/MS Rapid sample preparation methods for the analysis
Application Note # CA-274101 Evaluation of Rapid Extraction and Analysis Techniques for Polycyclic Aromatic Hydrocarbons (PAHs) in Seafood by GC/MS/MS Rapid sample preparation methods for the analysis
Gas Chromatography (GC)
 Gas Chromatography (GC) Ahmad Aqel Ifseisi Assistant Professor of Analytical Chemistry College of Science, Department of Chemistry King Saud University P.O. Box 2455 Riyadh 11541 Saudi Arabia Office: AA53
Gas Chromatography (GC) Ahmad Aqel Ifseisi Assistant Professor of Analytical Chemistry College of Science, Department of Chemistry King Saud University P.O. Box 2455 Riyadh 11541 Saudi Arabia Office: AA53
ChromTech options for PAL systems
 ChromTech options for PAL systems SPDE features full automation for dynamic sample extraction, desorption and analysis using the CTC Combi PAL. Most of our customers use SPDE for food and flavor research,
ChromTech options for PAL systems SPDE features full automation for dynamic sample extraction, desorption and analysis using the CTC Combi PAL. Most of our customers use SPDE for food and flavor research,
Chromatography. Gas Chromatography
 Chromatography Chromatography is essentially the separation of a mixture into its component parts for qualitative and quantitative analysis. The basis of separation is the partitioning of the analyte mixture
Chromatography Chromatography is essentially the separation of a mixture into its component parts for qualitative and quantitative analysis. The basis of separation is the partitioning of the analyte mixture
Application Note. Abstract. Introduction. Determination of Polycyclic Aromatic Hydrocarbons in Seafood by an Automated QuEChERS Solution
 Application Note Abstract Determination of Polycyclic Aromatic Hydrocarbons in Seafood by an Automated QuEChERS Solution Page 1 Polycyclic Aromatic Hydrocarbons (PAHs) are a large group of organic compounds
Application Note Abstract Determination of Polycyclic Aromatic Hydrocarbons in Seafood by an Automated QuEChERS Solution Page 1 Polycyclic Aromatic Hydrocarbons (PAHs) are a large group of organic compounds
Chapter 27: Gas Chromatography. Principles Instrumentation Detectors Columns and Stationary Phases Applications
 Chapter 27: Gas Chromatography Principles Instrumentation Detectors Columns and Stationary Phases Applications GC-MS Schematic Interface less critical for capillary columns Several types of Mass Specs
Chapter 27: Gas Chromatography Principles Instrumentation Detectors Columns and Stationary Phases Applications GC-MS Schematic Interface less critical for capillary columns Several types of Mass Specs
Determination of Off-Odor Compounds in Drinking Water Using an SPME Device with Gas Chromatography and Mass Spectrometry
 Application Note Environmental Determination of Off-Odor Compounds in Drinking Water Using an SPME Device with Gas Chromatography and Mass Spectrometry Authors Ye Kong and Zhe Cao Agilent Technologies,
Application Note Environmental Determination of Off-Odor Compounds in Drinking Water Using an SPME Device with Gas Chromatography and Mass Spectrometry Authors Ye Kong and Zhe Cao Agilent Technologies,
CHEM. RES. CHINESE UNIVERSITIES 2011, 27(2),
 CHEM. RES. CHINESE UNIVERSITIES 2011, 27(2), 198 202 Solid-phase Microextraction with Benzoxy-calix[6]arene Fiber Coupled to Gas Chromatography for the Analysis of Polycyclic Aromatic Hydrocarbons in Water
CHEM. RES. CHINESE UNIVERSITIES 2011, 27(2), 198 202 Solid-phase Microextraction with Benzoxy-calix[6]arene Fiber Coupled to Gas Chromatography for the Analysis of Polycyclic Aromatic Hydrocarbons in Water
Sulfotepp impurities in Chlorpyrifos EC formulations
 Page 1 of 16 Method DAS-AM-01-058 Sulfotepp impurities in Chlorpyrifos EC formulations A. ABSTRACT Method DAS-AM-01-058 has been validated for the analysis of the relevant impurity sulfotepp in chlorpyrifos
Page 1 of 16 Method DAS-AM-01-058 Sulfotepp impurities in Chlorpyrifos EC formulations A. ABSTRACT Method DAS-AM-01-058 has been validated for the analysis of the relevant impurity sulfotepp in chlorpyrifos
PAH Analyses with High Efficiency GC Columns: Column Selection and Best Practices
 PAH Analyses with High Efficiency GC Columns: Column Selection and Best Practices Food Quality and Environmental Author Ken Lynam Agilent Technologies, Inc. 280 Centerville Road Wilmington, DE 19808 Abstract
PAH Analyses with High Efficiency GC Columns: Column Selection and Best Practices Food Quality and Environmental Author Ken Lynam Agilent Technologies, Inc. 280 Centerville Road Wilmington, DE 19808 Abstract
H 3 CO H 3 CO S CH 3
 FENITROTHION 35 H 3 CO P H 3 CO S O CH 3 NO 2 ISO common name Chemical name Fenitrothion O,O-Dimethyl O-4-nitro-m-tolyl phosphorothioate (IUPAC) O,O-Dimethyl O-(3-methyl-4-nitrophenyl)- phosphorothioate
FENITROTHION 35 H 3 CO P H 3 CO S O CH 3 NO 2 ISO common name Chemical name Fenitrothion O,O-Dimethyl O-4-nitro-m-tolyl phosphorothioate (IUPAC) O,O-Dimethyl O-(3-methyl-4-nitrophenyl)- phosphorothioate
STANDARD OPERATING PROCEDURES
 PAGE: 1 of 12 CONTENTS 1.0 SCOPE AND APPLICATION 2.0 METHOD SUMMARY 3.0 SAMPLE PRESERVATION, CONTAINERS, HANDLING, AND STORAGE 4.0 INTERFERENCES AND POTENTIAL PROBLEMS 5.0 EQUIPMENT/APPARATUS 6.0 REAGENTS
PAGE: 1 of 12 CONTENTS 1.0 SCOPE AND APPLICATION 2.0 METHOD SUMMARY 3.0 SAMPLE PRESERVATION, CONTAINERS, HANDLING, AND STORAGE 4.0 INTERFERENCES AND POTENTIAL PROBLEMS 5.0 EQUIPMENT/APPARATUS 6.0 REAGENTS
STANDARD OPERATING PROCEDURES SOP: 1828 PAGE: 1 of 14 REV: 0.0 DATE: 05/12/95 ANALYSIS OF METHYL PARATHION IN CARPET SAMPLES BY GC/MS
 PAGE: 1 of 14 1.0 SCOPE AND APPLICATION 2.0 METHOD SUMMARY CONTENTS 3.0 SAMPLE PRESERVATION, CONTAINERS, HANDLING AND STORAGE 4.0 INTERFERENCES AND POTENTIAL PROBLEMS 5.0 EQUIPMENT/APPARATUS 6.0 REAGENTS
PAGE: 1 of 14 1.0 SCOPE AND APPLICATION 2.0 METHOD SUMMARY CONTENTS 3.0 SAMPLE PRESERVATION, CONTAINERS, HANDLING AND STORAGE 4.0 INTERFERENCES AND POTENTIAL PROBLEMS 5.0 EQUIPMENT/APPARATUS 6.0 REAGENTS
GC/MS Application Note
 GC/MS Application Note Determination of Odor Compounds in Water by SPME Arrow Gas Chromatography/Mass Spectrometry www.palsystem.com Determination of Odor Compounds in Water by SPME Arrow Gas Chromatography/Mass
GC/MS Application Note Determination of Odor Compounds in Water by SPME Arrow Gas Chromatography/Mass Spectrometry www.palsystem.com Determination of Odor Compounds in Water by SPME Arrow Gas Chromatography/Mass
TOTALLY INNOVATIVE MULTIMODE AUTOSAMPLER NEW KONIK ROBOKROM Laboratory Gas Generators An Overview +8 OPERATIONAL MODES MAIN FEATURES
 ROBOKROM 1 TOTALLY INNOVATIVE MULTIMODE AUTOSAMPLER NEW KONIK ROBOKROM Laboratory Gas Generators An Overview ROBOKROM 2 +8 OPERATIONAL MODES HRGC+HRGC-MS HRGC+HPLC-MS STATIC HEAD SPACE PURGE & TRAP SMPE
ROBOKROM 1 TOTALLY INNOVATIVE MULTIMODE AUTOSAMPLER NEW KONIK ROBOKROM Laboratory Gas Generators An Overview ROBOKROM 2 +8 OPERATIONAL MODES HRGC+HRGC-MS HRGC+HPLC-MS STATIC HEAD SPACE PURGE & TRAP SMPE
GC Instruments. GC Instruments - Sample Introduction
 GC Instruments 1 Fairly simple instrumentation Maintaining constant average pressure is important! Pressure controls flow rate T influences retention (k ) Flow rate monitoring Changing flow rate changes
GC Instruments 1 Fairly simple instrumentation Maintaining constant average pressure is important! Pressure controls flow rate T influences retention (k ) Flow rate monitoring Changing flow rate changes
So Many Columns! How Do I Choose? Daron Decker Chromatography Technical Specialist
 So Many Columns! How Do I Choose? Daron Decker Chromatography Technical Specialist GC Columns Wall Coated Open Tubulars Liquid phase coated capillaries Internal Diameter 0.05 0.53mm Length 5m 100m Porous
So Many Columns! How Do I Choose? Daron Decker Chromatography Technical Specialist GC Columns Wall Coated Open Tubulars Liquid phase coated capillaries Internal Diameter 0.05 0.53mm Length 5m 100m Porous
Ch24. Gas Chromatography (GC)
 Ch24. Gas Chromatography (GC) 24.1 What did they eat in the year 1000? From 13 C content of cholesterol in ancient bone 13 C : 1.1%, 12 C: 98.9% 13 C/ 12 C ratio types of plants Bones of 50 people in Barton-on-Humber
Ch24. Gas Chromatography (GC) 24.1 What did they eat in the year 1000? From 13 C content of cholesterol in ancient bone 13 C : 1.1%, 12 C: 98.9% 13 C/ 12 C ratio types of plants Bones of 50 people in Barton-on-Humber
AYOUB ET AL.: JOURNAL OF AOAC INTERNATIONAL VOL. 85, NO. 6,
 AYOUB ET AL.: JOURNAL OF AOAC INTERNATIONAL VOL. 85, NO. 6, 2002 1205 SPECIAL REPORT Determination of Low-Level Residual Ethylene Oxide by Using Solid-Phase Microextraction and Gas Chromatography KAMAL
AYOUB ET AL.: JOURNAL OF AOAC INTERNATIONAL VOL. 85, NO. 6, 2002 1205 SPECIAL REPORT Determination of Low-Level Residual Ethylene Oxide by Using Solid-Phase Microextraction and Gas Chromatography KAMAL
Polycyclic Aromatic Hydrocarbons in Water by GC/MS PBM
 Organics Revision Date: July 10, 2017 Polycyclic Aromatic Hydrocarbons in Water by GC/MS PBM Parameter Analytical Method Introduction Method Summary MDL(s) and EMS Analyte Codes Polycyclic Aromatic Hydrocarbons
Organics Revision Date: July 10, 2017 Polycyclic Aromatic Hydrocarbons in Water by GC/MS PBM Parameter Analytical Method Introduction Method Summary MDL(s) and EMS Analyte Codes Polycyclic Aromatic Hydrocarbons
Analysis of USP Method <467> Residual Solvents on the Agilent 8890 GC System
 Application Note Residual Solvent Analysis of USP Method Residual Solvents on the Agilent 889 GC System Author Lukas Wieder, Jie Pan, and Rebecca Veeneman Agilent Technologies, Inc. 8 Centerville Road
Application Note Residual Solvent Analysis of USP Method Residual Solvents on the Agilent 889 GC System Author Lukas Wieder, Jie Pan, and Rebecca Veeneman Agilent Technologies, Inc. 8 Centerville Road
APPENDIX A TO PART 136 METHODS FOR ORGANIC CHEMICAL ANALYSIS OF MUNICIPAL AND INDUSTRIAL WASTEWATER METHOD 610 POLYNUCLEAR AROMATIC HYDROCARBONS
 APPENDIX A TO PART 136 METHODS FOR ORGANIC CHEMICAL ANALYSIS OF MUNICIPAL AND INDUSTRIAL WASTEWATER METHOD 610 POLYNUCLEAR AROMATIC HYDROCARBONS 1. Scope and Application 1.1 This method covers the determination
APPENDIX A TO PART 136 METHODS FOR ORGANIC CHEMICAL ANALYSIS OF MUNICIPAL AND INDUSTRIAL WASTEWATER METHOD 610 POLYNUCLEAR AROMATIC HYDROCARBONS 1. Scope and Application 1.1 This method covers the determination
Gas Chromatography. Introduction
 Gas Chromatography Introduction 1.) Gas Chromatography Mobile phase (carrier gas) is a gas - Usually N 2, He, Ar and maybe H 2 - Mobile phase in liquid chromatography is a liquid Requires analyte to be
Gas Chromatography Introduction 1.) Gas Chromatography Mobile phase (carrier gas) is a gas - Usually N 2, He, Ar and maybe H 2 - Mobile phase in liquid chromatography is a liquid Requires analyte to be
Chem 230, Fall, 2014 Homework Set # 3 Short Answer SOLUTIONS
 Chem 230, Fall, 2014 Homework Set # 3 Short Answer SOLUTIONS 1. List two advantages of temperature programming in GC. a) Allows separation of solutes with widely varying retention factors in a reasonable
Chem 230, Fall, 2014 Homework Set # 3 Short Answer SOLUTIONS 1. List two advantages of temperature programming in GC. a) Allows separation of solutes with widely varying retention factors in a reasonable
PAL SPME Arrow The Better SPME
 PAL The Better SPME PAL The new dimension for Solid-Phase Micro SPME has become one of the most widely used extraction technologies for environmental, food and clinical analyses. It is well suited for
PAL The Better SPME PAL The new dimension for Solid-Phase Micro SPME has become one of the most widely used extraction technologies for environmental, food and clinical analyses. It is well suited for
GUIDELINES FOR THE DESIGN OF CHROMATOGRAPHIC ANALYTICAL METHODS INTENDED FOR CIPAC COLLABORATIVE STUDY
 Page 1 of 13 CIPAC/4105/R GUIDELINES FOR THE DESIGN OF CHROMATOGRAPHIC ANALYTICAL METHODS INTENDED FOR CIPAC COLLABORATIVE STUDY Prepared for CIPAC by Dr M J Tandy*, P M Clarke and B White (UK) The rapid
Page 1 of 13 CIPAC/4105/R GUIDELINES FOR THE DESIGN OF CHROMATOGRAPHIC ANALYTICAL METHODS INTENDED FOR CIPAC COLLABORATIVE STUDY Prepared for CIPAC by Dr M J Tandy*, P M Clarke and B White (UK) The rapid
METHOD: 1403, Issue 3 EVALUATION: FULL Issue 1: 15 August 1990 Issue 3: 15 March 2003
 ALCOHOLS IV 1403 (1) HOCH 2 CH 2 OCH 3 MW: (1) 76.09 CAS: (1) 109-86-4 RTECS: (1) KL5775000 (2) HOCH 2 CH 2 OCH 2 CH 3 (2) 90.12 (2) 110-80-5 (2) KK8050000 (3) HOCH 2 CH 2 O(CH 2 ) 3 CH 3 (3) 118.17 (3)
ALCOHOLS IV 1403 (1) HOCH 2 CH 2 OCH 3 MW: (1) 76.09 CAS: (1) 109-86-4 RTECS: (1) KL5775000 (2) HOCH 2 CH 2 OCH 2 CH 3 (2) 90.12 (2) 110-80-5 (2) KK8050000 (3) HOCH 2 CH 2 O(CH 2 ) 3 CH 3 (3) 118.17 (3)
Determination of 24 PAHs in Drinking Water
 Determination of 24 PAHs in Drinking Water Application Note Food Testing and Agriculture Authors M.C. Díaz Ramos, A. Suárez, A. Rúbies, and R. Companyó Laboratori de l Agencia de Salut Publica de Barcelona
Determination of 24 PAHs in Drinking Water Application Note Food Testing and Agriculture Authors M.C. Díaz Ramos, A. Suárez, A. Rúbies, and R. Companyó Laboratori de l Agencia de Salut Publica de Barcelona
PAL SPME Arrow The Better SPME
 PAL The Better SPME PAL The new dimension for Solid-Phase Micro SPME has become one of the most widely used extraction technologies for environmental, food and clinical analyses. It is well suited for
PAL The Better SPME PAL The new dimension for Solid-Phase Micro SPME has become one of the most widely used extraction technologies for environmental, food and clinical analyses. It is well suited for
EVALUATION OF NEW POLAR FUNCTIONALISED SOL-GEL HYBRID SORBENT FOR STIR BAR SORPTIVE EXTRACTION OF ORGANIC DYES
 EVALUATIO OF EW POLAR FUCTIOALISED SOL-GEL HYBRID SORBET FOR STIR BAR SORPTIVE EXTRACTIO OF ORGAIC DYES 1 UR ATIQAH ABU BAKAR, 2* WA AII WA IBRAHIM, AD 3 MOHD. MARSI SAAGI 1, 2 Separation Science and Technology
EVALUATIO OF EW POLAR FUCTIOALISED SOL-GEL HYBRID SORBET FOR STIR BAR SORPTIVE EXTRACTIO OF ORGAIC DYES 1 UR ATIQAH ABU BAKAR, 2* WA AII WA IBRAHIM, AD 3 MOHD. MARSI SAAGI 1, 2 Separation Science and Technology
Supporting Information High Activity and Selectivity of Ag/SiO 2 Catalyst for Hydrogenation of Dimethyloxalate
 Supporting Information High Activity and Selectivity of Ag/SiO 2 Catalyst for Hydrogenation of Dimethyloxalate An-Yuan Yin, Xiao-Yang Guo, Wei-Lin Dai*, Kang-Nian Fan Shanghai Key Laboratory of Molecular
Supporting Information High Activity and Selectivity of Ag/SiO 2 Catalyst for Hydrogenation of Dimethyloxalate An-Yuan Yin, Xiao-Yang Guo, Wei-Lin Dai*, Kang-Nian Fan Shanghai Key Laboratory of Molecular
Reviewing Material Safety Data Sheets to Verify Significant Drinking Water Threats
 Reviewing Material Safety Data Sheets to Verify Significant Drinking Water Threats 1 INTRODUCTION When attempting to verify the existence of a Significant Drinking Water Threat (SDWT) related to the handling
Reviewing Material Safety Data Sheets to Verify Significant Drinking Water Threats 1 INTRODUCTION When attempting to verify the existence of a Significant Drinking Water Threat (SDWT) related to the handling
Laser induced fluorescence of polycyclic aromatic hydrocarbons: An approach to gas standards
 Laser induced fluorescence of polycyclic aromatic hydrocarbons: An approach to gas standards Patricia B. C. Forbes a,b*, Andreas Trüe a and Egmont R. Rohwer b a Natural Resources and the Environment, Council
Laser induced fluorescence of polycyclic aromatic hydrocarbons: An approach to gas standards Patricia B. C. Forbes a,b*, Andreas Trüe a and Egmont R. Rohwer b a Natural Resources and the Environment, Council
Chemistry Instrumental Analysis Lecture 28. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 28 Two types in general use: -packed (stationary phase) -open tubular or capillary determine selectivity and efficiency of the sample. Column Materials Column
Chemistry 4631 Instrumental Analysis Lecture 28 Two types in general use: -packed (stationary phase) -open tubular or capillary determine selectivity and efficiency of the sample. Column Materials Column
Determination of releasable 2,4,6-trichloroanisole in wine by cork stoppers (Resolution OIV-Oeno 296/2009)
 Method OIV-MA-AS315-16 Type IV method Determination of releasable in wine by cork stoppers (Resolution OIV-Oeno 296/2009) 1 SCOPE: The method of determination of releasable (TCA) by cork stoppers measures
Method OIV-MA-AS315-16 Type IV method Determination of releasable in wine by cork stoppers (Resolution OIV-Oeno 296/2009) 1 SCOPE: The method of determination of releasable (TCA) by cork stoppers measures
Agilent UltiMetal Plus Stainless Steel Deactivation for Tubing, Connectors, and Fittings
 Agilent UltiMetal Plus Stainless Steel Deactivation for Tubing, Connectors, and Fittings Technical Overview Introduction Modern GC and GC/MS instruments are important analytical tools for accurate and
Agilent UltiMetal Plus Stainless Steel Deactivation for Tubing, Connectors, and Fittings Technical Overview Introduction Modern GC and GC/MS instruments are important analytical tools for accurate and
Course goals: Course goals: Lecture 1 A brief introduction to chromatography. AM Quality parameters and optimization in Chromatography
 Emqal module: M0925 - Quality parameters and optimization in is a separation technique used for quantification of mixtures of analytes Svein.mjos@kj.uib.no Exercises and lectures can be found at www.chrombox.org/emq
Emqal module: M0925 - Quality parameters and optimization in is a separation technique used for quantification of mixtures of analytes Svein.mjos@kj.uib.no Exercises and lectures can be found at www.chrombox.org/emq
STANDARD OPERATING PROCEDURES SOP: 1826 PAGE: 1 of 18 REV: 0.0 DATE: 03/30/95 ANALYSIS OF METHYL PARATHION IN WIPE SAMPLES BY GC/MS
 PAGE: 1 of 18 CONTENTS 1.0 SCOPE AND APPLICATION 2.0 METHOD SUMMARY 3.0 SAMPLE PRESERVATION, CONTAINERS, HANDLING AND STORAGE 4.0 INTERFERENCES AND POTENTIAL PROBLEMS 5.0 EQUIPMENT/APPARATUS 6.0 REAGENTS
PAGE: 1 of 18 CONTENTS 1.0 SCOPE AND APPLICATION 2.0 METHOD SUMMARY 3.0 SAMPLE PRESERVATION, CONTAINERS, HANDLING AND STORAGE 4.0 INTERFERENCES AND POTENTIAL PROBLEMS 5.0 EQUIPMENT/APPARATUS 6.0 REAGENTS
Optimizing GC Parameters for Faster Separations with Conventional Instrumentation
 Optimizing GC Parameters for Faster Separations with Conventional Instrumentation Anila I. Khan, Thermo Fisher Scientific, Runcorn, Cheshire, UK Technical Note 243 Key Words TraceGOLD fast GC analysis
Optimizing GC Parameters for Faster Separations with Conventional Instrumentation Anila I. Khan, Thermo Fisher Scientific, Runcorn, Cheshire, UK Technical Note 243 Key Words TraceGOLD fast GC analysis
Rely on Rxi -PAH Columns
 Rely on Rxi -PAH Columns to Ensure Successful Polycyclic Aromatic Hydrocarbon (PAH) Analysis Optimized Efficiency, Selectivity, and Robustness Let You: Report accurate results with speed and confidence.
Rely on Rxi -PAH Columns to Ensure Successful Polycyclic Aromatic Hydrocarbon (PAH) Analysis Optimized Efficiency, Selectivity, and Robustness Let You: Report accurate results with speed and confidence.
The Pulsed Flame Photometric Detector for analysis of phosphorus pesticides in wastewater and food commodities
 The Pulsed Flame Photometric Detector for analysis of phosphorus pesticides in wastewater and food commodities Application Note Environmental Authors Agilent Technologies, Inc. Introduction Twenty-eight
The Pulsed Flame Photometric Detector for analysis of phosphorus pesticides in wastewater and food commodities Application Note Environmental Authors Agilent Technologies, Inc. Introduction Twenty-eight
Analysis of Trace (mg/kg) Thiophene in Benzene Using Two-Dimensional Gas Chromatography and Flame Ionization Detection Application
 Analysis of Trace (mg/kg) Thiophene in Using Two-Dimensional Gas Chromatography and Flame Ionization Detection Application Petrochemical Authors James D. McCurry and Bruce D. Quimby Agilent Technologies
Analysis of Trace (mg/kg) Thiophene in Using Two-Dimensional Gas Chromatography and Flame Ionization Detection Application Petrochemical Authors James D. McCurry and Bruce D. Quimby Agilent Technologies
Trace analysis of mesityl oxide and diacetone alcohol in pharmaceuticals by capillary gas chromatography with flame ionization detection
 Trade Science Inc. September 2009 Volume 8 Issue 3 ACAIJ, 8(3) 2009 [346-349] Trace analysis of mesityl oxide and diacetone alcohol in pharmaceuticals by capillary gas chromatography with flame ionization
Trade Science Inc. September 2009 Volume 8 Issue 3 ACAIJ, 8(3) 2009 [346-349] Trace analysis of mesityl oxide and diacetone alcohol in pharmaceuticals by capillary gas chromatography with flame ionization
Analysis of BTEX in Natural Water with SPME
 Analysis of BTEX in Natural Water with SPME Application Note Environmental Author Giordano Vassalli Sezione Protezione Aria Introduction Benzene, toluene, ethylbenzene and xylene (BTEX) isomers are monocyclic
Analysis of BTEX in Natural Water with SPME Application Note Environmental Author Giordano Vassalli Sezione Protezione Aria Introduction Benzene, toluene, ethylbenzene and xylene (BTEX) isomers are monocyclic
THE USE OF ORGANOCLAY IN MANAGING DISSOLVED ORGANIC CONTAMINANTS RELEVANT TO CONTAMINATED SEDIMENTS
 THE USE OF ORGANOCLAY IN MANAGING DISSOLVED ORGANIC CONTAMINANTS RELEVANT TO CONTAMINATED SEDIMENTS Organoclay has great potential as a permeable adsorptive barrier in treating contaminated groundwater
THE USE OF ORGANOCLAY IN MANAGING DISSOLVED ORGANIC CONTAMINANTS RELEVANT TO CONTAMINATED SEDIMENTS Organoclay has great potential as a permeable adsorptive barrier in treating contaminated groundwater
Application Note. Abstract. Introduction. Experimental-Instrument Conditions. By: Anne Jurek
 Automated Handling Techniques for the Analysis of Elevated Volatile Organic Compound (VOC) Concentrations in Soils Utilizing the Atomx Concentrator/Multimatrix Autosampler. Application Note By: Anne Jurek
Automated Handling Techniques for the Analysis of Elevated Volatile Organic Compound (VOC) Concentrations in Soils Utilizing the Atomx Concentrator/Multimatrix Autosampler. Application Note By: Anne Jurek
Pesticides Analysis Using the Agilent 5977A Series GC/MSD
 Pesticides Analysis Using the Agilent 5977A Series GC/MSD Application Note Food Testing and Agriculture Authors Jia-jia Wu and Wen-wen Wang Agilent Technologies Co. Ltd (China) Beijing, People s Republic
Pesticides Analysis Using the Agilent 5977A Series GC/MSD Application Note Food Testing and Agriculture Authors Jia-jia Wu and Wen-wen Wang Agilent Technologies Co. Ltd (China) Beijing, People s Republic
Developing Large Volume Injection (LVI) in Split / Splitless Inlets. Philip J. Koerner Phenomenex Inc. NEMC 2014
 Developing Large Volume Injection (LVI) in Split / Splitless Inlets Philip J. Koerner Phenomenex Inc. NEMC 2014 Goal To produce a method that provides the largest response of analytes that will produce
Developing Large Volume Injection (LVI) in Split / Splitless Inlets Philip J. Koerner Phenomenex Inc. NEMC 2014 Goal To produce a method that provides the largest response of analytes that will produce
High Performance Liquid Chromatography
 Updated: 3 November 2014 Print version High Performance Liquid Chromatography David Reckhow CEE 772 #18 1 HPLC System David Reckhow CEE 772 #18 2 Instrument Basics PUMP INJECTION POINT DETECTOR COLUMN
Updated: 3 November 2014 Print version High Performance Liquid Chromatography David Reckhow CEE 772 #18 1 HPLC System David Reckhow CEE 772 #18 2 Instrument Basics PUMP INJECTION POINT DETECTOR COLUMN
High Performance Liquid Chromatography
 Updated: 3 November 2014 Print version High Performance Liquid Chromatography David Reckhow CEE 772 #18 1 HPLC System David Reckhow CEE 772 #18 2 1 Instrument Basics PUMP INJECTION POINT DETECTOR COLUMN
Updated: 3 November 2014 Print version High Performance Liquid Chromatography David Reckhow CEE 772 #18 1 HPLC System David Reckhow CEE 772 #18 2 1 Instrument Basics PUMP INJECTION POINT DETECTOR COLUMN
VOC Analysis of Water-Based Coatings by Headspace-Gas Chromatography
 VOC Analysis of Water-Based Coatings by Headspace-Gas Chromatography Introduction ASTM International publishes a number of different methods for the determination of volatile compounds in water-based coatings.
VOC Analysis of Water-Based Coatings by Headspace-Gas Chromatography Introduction ASTM International publishes a number of different methods for the determination of volatile compounds in water-based coatings.
Simultaneous Estimation of Residual Solvents (Isopropyl Alcohol and Dichloromethane) in Dosage Form by GC-HS-FID
 Asian Journal of Chemistry Vol. 21, No. 3 (2009), 1739-1746 Simultaneous Estimation of Residual Solvents (Isopropyl Alcohol and Dichloromethane) in Dosage Form by GC-HS-FID PRAVEEN KUMAR BALIYAN*, R.P.
Asian Journal of Chemistry Vol. 21, No. 3 (2009), 1739-1746 Simultaneous Estimation of Residual Solvents (Isopropyl Alcohol and Dichloromethane) in Dosage Form by GC-HS-FID PRAVEEN KUMAR BALIYAN*, R.P.
Determination of Benzene, Toluene, Ethylbenzene and Xylene in River Water by Solid-Phase Extraction and Gas Chromatography
 2003 The Japan Society for Analytical Chemistry 1365 Determination of Benzene, Toluene, Ethylbenzene and Xylene in River Water by Solid-Phase Extraction and Gas Chromatography Mohammad A. MOTTALEB,* Mohammad
2003 The Japan Society for Analytical Chemistry 1365 Determination of Benzene, Toluene, Ethylbenzene and Xylene in River Water by Solid-Phase Extraction and Gas Chromatography Mohammad A. MOTTALEB,* Mohammad
Capillary GC Column Selection and Method Development A Primer on Column Parameters and Instrument Conditions
 Capillary GC Column Selection and Method Development A Primer on Column Parameters and Instrument Conditions Michael D. Buchanan September 11, 2014 sigma-aldrich.com/analytical 2012 Sigma-Aldrich Co. All
Capillary GC Column Selection and Method Development A Primer on Column Parameters and Instrument Conditions Michael D. Buchanan September 11, 2014 sigma-aldrich.com/analytical 2012 Sigma-Aldrich Co. All
A Tunable Process: Catalytic Transformation of Renewable Furfural with. Aliphatic Alcohols in the Presence of Molecular Oxygen. Supporting Information
 Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2015 A Tunable Process: Catalytic Transformation of Renewable Furfural with Aliphatic
Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2015 A Tunable Process: Catalytic Transformation of Renewable Furfural with Aliphatic
AppNote 8/2001 KEYWORDS ABSTRACT. PAH, SPME, SBSE, GERSTEL Twister, Thermal desorption, GC-MS, PCB
 AppNote 8/2001 Comparison of the Sensitivity of Solid Phase MicroExtraction (SPME) and Stir Bar Sorptive Extraction (SBSE) for the Determination of Polycyclic Aromatic Hydrocarbons (PAHs) in Water and
AppNote 8/2001 Comparison of the Sensitivity of Solid Phase MicroExtraction (SPME) and Stir Bar Sorptive Extraction (SBSE) for the Determination of Polycyclic Aromatic Hydrocarbons (PAHs) in Water and
1. A gas chromatograph equipped with flame ionization detection and a computing integrator.
 HYDRO.02-1 HYDROXYETHYL SUBSTITUTION LEVEL PRINCIPLE SCOPE The hydroxyethyl group is cleaved from the starch by hydrogen iodide in the presence of adipic acid and an organic solvent containing a known
HYDRO.02-1 HYDROXYETHYL SUBSTITUTION LEVEL PRINCIPLE SCOPE The hydroxyethyl group is cleaved from the starch by hydrogen iodide in the presence of adipic acid and an organic solvent containing a known
Supporting Information
 Supporting Information Dynamic Interaction between Methylammonium Lead Iodide and TiO 2 Nanocrystals Leads to Enhanced Photocatalytic H 2 Evolution from HI Splitting Xiaomei Wang,, Hong Wang,, Hefeng Zhang,,
Supporting Information Dynamic Interaction between Methylammonium Lead Iodide and TiO 2 Nanocrystals Leads to Enhanced Photocatalytic H 2 Evolution from HI Splitting Xiaomei Wang,, Hong Wang,, Hefeng Zhang,,
*Correspondence to:
 Supporting Information for Carbonate-promoted hydrogenation of carbon dioxide to multi-carbon carboxylates Aanindeeta Banerjee 1 and Matthew W. Kanan 1 * 1 Department of Chemistry, Stanford University,
Supporting Information for Carbonate-promoted hydrogenation of carbon dioxide to multi-carbon carboxylates Aanindeeta Banerjee 1 and Matthew W. Kanan 1 * 1 Department of Chemistry, Stanford University,
Residual Solvents in Pharmaceuticals by USP Chapter <467> Methodology
 APPLICATION NOTE Gas Chromatography Author: David Scott PerkinElmer, Inc. Shelton, CT Residual Solvents in Pharmaceuticals by USP Chapter Methodology Introduction The synthesis of active pharmaceutical
APPLICATION NOTE Gas Chromatography Author: David Scott PerkinElmer, Inc. Shelton, CT Residual Solvents in Pharmaceuticals by USP Chapter Methodology Introduction The synthesis of active pharmaceutical
Chemistry Instrumental Analysis Lecture 27. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 27 Gas Chromatography Introduction GC covers all chromatographic methods in which the mobile phase is gas. It may involve either a solid stationary phase (GSC)
Chemistry 4631 Instrumental Analysis Lecture 27 Gas Chromatography Introduction GC covers all chromatographic methods in which the mobile phase is gas. It may involve either a solid stationary phase (GSC)
Ultra-Inert chemistry for Trace Level Analysis
 Ultra-Inert chemistry for Trace Level Analysis Cikui Liang, Ph.D. Challenges and Needs of Today s Laboratories Challenges Qualification/quantification of trace samples Keep instrument up and running Needs
Ultra-Inert chemistry for Trace Level Analysis Cikui Liang, Ph.D. Challenges and Needs of Today s Laboratories Challenges Qualification/quantification of trace samples Keep instrument up and running Needs
Formation of artefacts during air analysis of volatile amines by solid-phase micro extraction
 Formation of artefacts during air analysis of volatile amines by solid-phase micro extraction François Lestremau,* Valérie Desauziers and Jean Louis Fanlo THE ANALYST FULL PAPER Laboratoire Génie de l
Formation of artefacts during air analysis of volatile amines by solid-phase micro extraction François Lestremau,* Valérie Desauziers and Jean Louis Fanlo THE ANALYST FULL PAPER Laboratoire Génie de l
Analyzing Residual Solvents in Pharmaceutical Products Using GC Headspace with Valve-and-Loop Sampling
 Analyzing Residual Solvents in Pharmaceutical Products Using GC Headspace with Valve-and-Loop Sampling Andrea Caruso and Massimo Santoro, Thermo Fisher Scientific, Milan, Italy Application Note 1316 Key
Analyzing Residual Solvents in Pharmaceutical Products Using GC Headspace with Valve-and-Loop Sampling Andrea Caruso and Massimo Santoro, Thermo Fisher Scientific, Milan, Italy Application Note 1316 Key
Application Note. Abstract. Introduction. Experimental-Instrument Conditions. By: Anne Jurek
 Requirements of an Automated Sample Delivery System in Today s Realm of Ever Increasing Sensitivity Demands Utilizing the Atomx Concentrator/Multimatrix Autosampler. Application Note By: Anne Jurek Abstract
Requirements of an Automated Sample Delivery System in Today s Realm of Ever Increasing Sensitivity Demands Utilizing the Atomx Concentrator/Multimatrix Autosampler. Application Note By: Anne Jurek Abstract
Chemical Analysis Problem
 Chemical Analysis Problem Hair analysis is frequently used for the long-term monitoring of drug and alcohol users. You are working at a forensics laboratory and have been given the task of developing a
Chemical Analysis Problem Hair analysis is frequently used for the long-term monitoring of drug and alcohol users. You are working at a forensics laboratory and have been given the task of developing a
Synthesis of jet fuel range cycloalkanes with diacetone alcohol. from lignocellulose
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2016 Supporting Information Synthesis of jet fuel range cycloalkanes with diacetone alcohol from
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2016 Supporting Information Synthesis of jet fuel range cycloalkanes with diacetone alcohol from
Synthesis of renewable diesel with hydroxyacetone and 2-methyl-furan
 Supporting Information Synthesis of renewable diesel with hydroxyacetone and 2-methyl-furan Guangyi Li, a,b Ning Li, a Shanshan Li, a,b Aiqin Wang, a Yu Cong, a Xiaodong Wang a and Tao Zhang a * a State
Supporting Information Synthesis of renewable diesel with hydroxyacetone and 2-methyl-furan Guangyi Li, a,b Ning Li, a Shanshan Li, a,b Aiqin Wang, a Yu Cong, a Xiaodong Wang a and Tao Zhang a * a State
Selective Formation of Benzo[c]cinnoline by Photocatalytic Reduction of 2,2 Dinitrobiphenyl with TiO 2 and UV light irradiation
![Selective Formation of Benzo[c]cinnoline by Photocatalytic Reduction of 2,2 Dinitrobiphenyl with TiO 2 and UV light irradiation Selective Formation of Benzo[c]cinnoline by Photocatalytic Reduction of 2,2 Dinitrobiphenyl with TiO 2 and UV light irradiation](/thumbs/90/104361197.jpg) Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2015 Content: Selective Formation of Benzo[c]cinnoline by Photocatalytic Reduction of
Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2015 Content: Selective Formation of Benzo[c]cinnoline by Photocatalytic Reduction of
Katherine K. Stenerson, Michael Ye, Michael Halpenny, Olga Shimelis, and Leonard M. Sidisky. Supelco, Div. of Sigma-Aldrich Bellefonte, PA USA
 New Analytical Tools for the Determination of Persistent Organic Pollutants (POPs) in Fatty Food and Beverage Matrices Using QuEChERS Extraction/Cleanup and Gas Chromatography (GC) Analysis Katherine K.
New Analytical Tools for the Determination of Persistent Organic Pollutants (POPs) in Fatty Food and Beverage Matrices Using QuEChERS Extraction/Cleanup and Gas Chromatography (GC) Analysis Katherine K.
METHOD 3600B CLEANUP
 METHOD 3600B CLEANUP 1.0 SCOPE AND APPLICATION 1.1 Method 3600 provides general guidance on selection of cleanup methods that are appropriate for the target analytes of interest. Cleanup methods are applied
METHOD 3600B CLEANUP 1.0 SCOPE AND APPLICATION 1.1 Method 3600 provides general guidance on selection of cleanup methods that are appropriate for the target analytes of interest. Cleanup methods are applied
Chemistry 311: Instrumental Analysis Topic 4: Basic Chromatography. Chemistry 311: Instrumental Analysis Topic 4: Basic Chromatography
 Introductory Theory, Basic Components, Qualitative and Quantitative applications. HPLC, GC, Ion Chromatography. Rouessac Ch. 1-7 Winter 2011 Page 1 Chromatography: The separation of analytes based on differences
Introductory Theory, Basic Components, Qualitative and Quantitative applications. HPLC, GC, Ion Chromatography. Rouessac Ch. 1-7 Winter 2011 Page 1 Chromatography: The separation of analytes based on differences
