SUPPLEMENTARY INFORMATION
|
|
|
- Clement Powers
- 5 years ago
- Views:
Transcription
1 In the format provided by the authors and unedited. DOI: /NMAT4868 Antifogging abilities of model nanotextures Timothée Mouterde 1,2, Gaëlle Lehoucq 3, Stéphane Xavier 3, Antonio Checco 4, Charles T. Black 5, Atikur Rahman 5, Thierry Midavaine 6, Christophe Clanet 1,2 & David Quéré 1,2 1. Physique et Mécanique des Milieux Hétérogènes, UMR 7636 du CNRS, ESPCI, Paris, France. 2. LadHyX, UMR 7646 du CNRS, École polytechnique, Palaiseau, France. 3. Thales Research & Technology, Route Départementale 128, Palaiseau, France. 4. Condensed Matter Physics and Materials Science Department, Brookhaven National Laboratory, Upton NY 11973, USA. 5. Center for Functional Nanomaterials, Brookhaven National Laboratory, Upton NY 11973, USA. 6. Thales Optronique SAS, 2 avenue Gay-Lussac, Élancourt, France. We present in these supplementary materials additional experiments, and follow the development of the accompanying paper. 1) We compare the wetting characteristics of our samples. 2) We discuss the influence of the injection rate of hot water on the various substrates. 3) We show that drops still bounce on substrate A even when hot, but stick to substrates B (in agreement with results on both detaching drops and bouncing jets in the accompanying paper). 4) We model and discuss the possibility of leakage as a nucleus of water grows in a cell constituted of hydrophobic pillars. 5) We carefully discuss the model for the condensation-induced force, leading to Eq. (1). 6) We describe the measurement of the radius of departing drops. 7) We compare the antifogging ability on material C (nanocones) to that on a disordered nanotextured material for which jumps at condensation were reported in the literature (ref. 22 in the accompanying paper). 8) We provide captions for the movies. 1. Wetting characteristics of our samples For each sample, we measure advancing and receding contact angles of water at room temperature. A drop of volume Ω = 5 µl is dispensed from a needle on substrates horizontally displaced at V = 300 µm/s, while water remains attached to the needle (Figure S1). Denoting η and γ as the liquid viscosity and surface tension, the corresponding capillary number ηv/γ is , a value much smaller than unity: measurements are quasi-static. This experiment allows us to scan the surface of the substrates by centimetre-size distances, and we checked that there is no wetting anomaly along the explored paths. Typical photos of NATURE MATERIALS 1
2 drops displayed in Figure S1 are similar on all substrates, and corresponding contact angles are reported in the enclosed table. For each sample, ten experiments were performed, and the reported value in each case is the average over these measurements. Standard deviations are 2 for the advancing angle, and 3 for the receding angle. θa θr θr θa v A B1 A B B B2 B3 B Figure S1. Water droplets of volume Ω = 5 µl on the different substrates used in our study (and defined in the main text). The substrate is moved at V = 300 µm/s, while water is maintained by a needle, and we measure quasi-static advancing and receding contact angles whose mean values are reported in the table. Advancing and receding angles are very similar for the B family, and the corresponding mean values are 168 ± 2, and 143 ± 3. These values are characteristic of a Cassie state. The A sample also has a similar wettability, with advancing and receding angles of 167 ± 2 and 140 ± 2, respectively. The Cassie formula would predict smaller contact angles for the A substrate, of higher pillar density. We observe a very slight decrease of contact angle, yet not significant enough to agree with the Cassie model (which anyway is rarely a quantitative tool for superhydrophobic contact angles, owing to the existence of hysteresis). It is interesting to note that A has pillars with a size smaller than the range of van der Waals forces (around 100 nm), so that wetting and pinning at this scale might be modified by long range effects: the liquids tails that appear in the receding stage are van der Waals-unstable, so that the density of actual pinning might be smaller than assumed if there is one tail per pillar. For example, if there is one tail per pair of pillars, the effective pillar density is divided by 2 a large effect, indeed, which brings us back to the typical pillar density of samples B. In addition, nanopillars edges are often less sharp than micropillars edges, which also decreases the strength of pinning. These two effects generate the possibility of having larger contact angles than expected from a macroscopic formula such as Cassie equation. Anyway, the similarity of the wetting characteristics for our four samples appear to be a fortunate coincidence, allowing us to make quantitative comparisons between materials having comparable adhesion towards NATURE MATERIALS 2
3 water without condensation. It is finally worth noting that the A-sample, which is very slightly more adhesive without condensation, is by far less adhesive as water condenses. This confirms the difference in nature between intrinsic adhesion and condensation adhesion. 2. Effect of injection rate on our measurements In order to characterize water adhesion induced by condensation in textured materials, we measure the critical mass m above which a drop slides on an incline, as explained in the accompanying paper. We compare the mass when there is no temperature difference between water and its substrate, to the situation where the substrate is colder than the liquid by a quantity ΔT. Here we discuss the sensitivity of our measurements to the rate Q at which drops are dispensed. We report in Figure S2a the mass of drops detaching from surface A (p = 52 nm) for ΔT = 0 C and ΔT = 15 C, for flow rates varying between 0.01 ml/min to 0.3 ml/min. The critical mass is (slightly yet systematically) larger when there is condensation (ΔT > 0), in agreement with the (blue) data in Figure 2d of the accompanying paper. In addition, mass at detachment is found here to be quite independent of the injection rate for both ΔT. m (mg) T = 0 C T = 15 C Q (ml/min) (a) B2 A B2 A V (m/s) T = 15 C V (m/s) T = 0 C bouncing non-bouncing (b) Figure S2. (a) Mass m of water drop detaching from the surface A (p = 52 nm) as a function of the flow rate Q (varying from 0.01 ml/min to 0.3 ml/min) at which drops are dispensed. Black data represent the reference case when there is no temperature difference, that is, no condensation-induced adhesion; blue data are used when there is a temperature difference T = 15 C between water and the substrate. (b) Diagram showing the behaviour of a water jet of velocity V between 1.6 and 6.3 m/s impacting sample A (p = 52 nm) or B2 (p = 840 nm) for T = 0 C and T = 15 C. A green spot means that the jet bounces, a red spot that it sticks to the surface. However, we observe in Figure S2a two small effects bounding the rates at which measurements can be done. At very low injection rate (Q = 0.01 ml/min), the mass difference (and consequently the adhesion) is found to be smaller than that at higher injection rate. At such NATURE MATERIALS 3
4 slow injection, drops are filled in more than 2 minutes. During this time, heat transfer can cool the drop, leading to a smaller contrast of temperature between the drop and the surface hence the observed decrease of condensation-induced adhesion. We can estimate the cooling time τ 0 of a water drop warmer by ΔT than the surrounding. It is typically τ 0 ρc p R 2 /λ, where ρ = 1000 kg/m 3 is the density of water, C p = 4100 J/kg/K its heat capacity, R = 1 mm the typical size of the drop and λ = W/m/K the thermal conductivity of the surrounding air at 25 C. τ 0 is around 150 s, so that temperature can indeed equilibrate at a flow rate on the order of 0.01 ml/min. The effect becomes hardly visible if Q = 0.05 ml/min (that is, a filling time smaller than 30 s), above which we indeed observe that blue data plateau in Figure S2a. At high injection rate (Q = 0.3 ml/min), the mass m simultaneously decreases for both series of data. For needles with diameter 200 µm, the corresponding injection velocity V is about 0.14 m/s. The (local) dynamic pressure ρv 2 /2 associated to the flow is on the order of 10 Pa, that is, comparable to the global hydrostatic pressure difference in the drop, of about 10 Pa at detachment: a high flow rate produces an inertial force that participates to the drop departure which leads to smaller values for m, independently of ΔT. At even higher Q, inertia produces a jet out of the needle, instead of regular droplets. We wonder whether our samples can maintain repellency when a high flux of warm water (ΔT > 0) is continuously brought by such jets. We can think of two effects. 1) Whatever the jet temperature, the impact speed can itself induce a sticky behavior, due to the dynamic impalement of liquid in the textures. We describe in Figure S2b the behavior of a water jet with various velocities and various temperatures impacting materials A (p = 52 nm) and B2 (p = 840 nm). Without temperature difference (bottom of the figure), water jets bounce on both surfaces for all explored velocities, which is expected: the dynamic pressure of the jet remains smaller than 40 kpa, a value itself smaller than the typical Laplace pressure γ/p needed to penetrate the texture, respectively 83 kpa for B2 and 1300 kpa for A. This confirms the ability of the nanoscale textures in A to repel water even for fast impacts. 2) When the temperature difference is increased (ΔT = 15 C, top of the figure), condensation occurs and water jets behave differently on both surfaces, as reported in the accompanying paper (Figures 2b and 2c). While bouncing is systematically observed on A, hot jets all stick to surface B2, and then creep along it (Figure 2b). This diagram finally confirms the ability of material A to keep repellency in broad conditions of temperature and impact velocity. NATURE MATERIALS 4
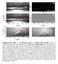
5 3. Behaviour of water drops impacting the substrates in various conditions Water drops in adhesion experiments are dispensed in a time ranging between 6 s and 24 s, and we assumed in our paper that the time τ needed to fill the cells (when condensation takes place) is much smaller than the typical duration of an experiment. (This assumption is justified in ref. 31 in the accompanying paper and it arises from the very small size of the cells, whose volume is always much smaller than 1 fl.) We show in Figure S3 chronophotographies of drops impacting sample A and B2, for ΔT = 0 C and ΔT = 15 C (interval between successive images, 6 ms). Colours are used to improve the readability of the figure, in particular in cases where drops stop and superimpose in the successive images. 15 T ( C) p (nm) Figure S3. Coloured chronophotographies of water drops with radius R = 1.1 mm impacting surfaces A (p = 52 nm) and B2 (p = 840 nm) at a velocity of 0.37 m/s. For the sake of clarity (in particular in the case where water sticks at impact), drops have been coloured differently on successive frames separated by 6 ms. It is observed that drops systematically bounce on material A, and on material B2 only for ΔT = 0 C: the impacting water with ΔT = 15 C sticks to B2. These experiments thus support the observations reported in the accompanying paper (Figures 2a, 2b and 2c), when condensation is present (loss of repellency for microtextures, upholding of repellency for nanotextures). They also extend these observations to the important practical case of impact. Finally, contact time in Figure S3 is 12 ± 1 ms, a time found to be large enough to generate water bridges on material B2, which leads to sticking (top right in the figure). This confirms that the time needed to condense water in the solid cavities is very short (smaller than 10 ms). NATURE MATERIALS 5
6 4. Configuration of water droplets growing in a network of pillars We assume in our model that water droplets stay enclosed within cells as they grow by vapour condensation. A cell being defined by the space between four proximal pillars, a liquid nucleus remains a spherical cap until its size becomes comparable to the spacing between pillars, as shown in Figure S4a. If the pillar height h is comparable to the spacing p, the nucleus directly merges with the drop above (not represented in Figure S4); if it is larger, the liquid adopts the shape of the cell (as sketched in Figure 1c) and keeps on filling. We propose here a simple energy argument to evaluate the condition under which water is likely to leak from the cell in this stage. h1 p θo state 1 (b) h2 state 2 (a) ε A 3.1 B1 1.4 B2 1.4 B3 1.4 (c) Figure S4. (a) Two wetting states for a large nucleus of water in a cell constituted by four proximal pillars. Either the nucleus remains trapped in the cell (state 1), or it leaks and invades the next cell (state 2). When leakage takes place, we consider the configuration of minimum energy, that is, with a constant thickness between both cells. (b) A water drop deposited on a flat silicon surface coated with 1H,1H,2H,2H-perfluorooctyltrichlorosilane exhibits a contact angle θ o = 120 ± 2. (c) The table gives the value of the number ε = p(1 cosθ o )/h (or 2 3p(1 cosθ o )/h for a rhombus) for the samples used in this study. When ε is larger than 1, it is energetically favourable for the liquid to stay within a cell up to its complete filling. As water is attached to the surrounding pillars, liquid interfaces are close to flat, as represented in Figure S4a. State 1 corresponds to a cell filled with water up to a height h 1. We consider the possibility of leakage to the next cell (state 2 in Figure S4a), where conservation of volume imposes a constant thickness h 2 = h 1 /2. The energy difference between both states is ΔE = 6γph 2 + 2γp 2 γp 2 cosθ o 4γph 1 γp 2, that is, ΔE = γp 2 (1 cosθ o ) γph 1, which is minimum for h 1 = h, as the liquid reaches the pillars tops and the drop above. Leakage does not happen provided we have ΔE(h) < 0, that is, ε = p(1 cosθ o )/h > 1. The same reasoning with a rhombic cell leads to a similar criterion: ε = 2 3p(1 cosθ o )/h > 1. NATURE MATERIALS 6
7 In order to be quantitative, we need to specify the Young contact angle θ o on a flat surface of the same chemical nature as our samples. We show in Figure S4b a water drop on smooth silicon coated with 1H,1H,2H,2H-perfluoro-octyltrichlorosilane (coating done the same way as for our structured samples). The contact angle θ o is observed to be 120 ± 2. This allows us to calculate the number ε for samples A, B1, B2 and B3, which is reported in the table in Figure S4c. We can notice that ε is always larger than unity, showing that cells should not leak as condensation takes place. The only cases where we expect adjacent cells to be filled (and merged) are the ones where nuclei appear and grow in proximal cells. 5. Condensation induced adhesion We characterize the first stage of condensation by the number n( T) of nuclei per unit area. As sketched in Figure 1c of the main text, substrates (seen from the top) are made of elementary cells delimited by four pillars. The projected surface area of a cell is p 2 but accounting for the pillar sides provides an actual surface area rp 2 hence an average number P of nuclei per cell P = np 2 r. At small ΔT (bottom left in Figure 1c of the main text), an elementary cell contains on average less than one nucleus (P < 1). As time progresses, the nuclei grow and eventually fill cells of height comparable to their size, until they come in contact after a time τ with the large drop resting on the pillars top. Even in soft condensation ( T = 0.04 C under the dew point), cells (always smaller than 1 µm) fill rapidly, with τ ~ ms (ref. 31 in the main text), as also discussed in the Supplementary Note 3. The design of pillars is such that water stays enclosed within each cell (see Supplementary Note 4). Then, as sketched in Figure 1c (top left), the breath figure is a collection of water cells (whose number increases with P) connected to the drop above. Figure S5. (a) Sketch of a triple line contacting a wet patch enclosed by four pillars. (b) The detachment of the line from the wet patch produces two liquid interfaces with area p 2, as coloured in red. NATURE MATERIALS 7
8 The elementary force f needed to detach this drop from a filled cell can be estimated as follows. A receding liquid leaves behind the (strongly pinned) wet patch: as shown in Figure S5, two water/air interfaces of surface area p 2 and surface energy γ are created in the process, which implies an elementary increase of surface energy by 2γp 2. This energy can be seen as the work of the detachment force f by a distance p, which yields f 2γp. The number N w of wet patches under the trailing edge of the drop is obtained by multiplying the cell number under the contact line N t = πr/p by the probability P = np 2 r for a cell to contain a nucleus. We get: N w πrnpr. F is simply obtained by adding the N w elementary tensions f acting on the triple line. We find F 2πRγrp 2 n, that is, Eq. 1 in the main text. 6. Radius of the trailing edge of detaching drops Here we discuss the value of the radius R of the trailing edge of drops detaching from samples A and B1-3 as described in the accompanying paper. Our method consists of dispensing drops of hot water until they detach, and taking top views 0.2 s after water started to slide. As seen in Figure S6 for ΔT = 15 C and samples A and B2, we observe (in white) the breath figure formed on the substrate during the filling time, and below a black area corresponding to the footprint of the pinned drop, and to its trail once moving. R R A 3 mm T = 15 C B2 Figure S6. Top view of the mark remaining just after a drop detaches. The drop is hotter by ΔT = 15 C than its substrate tilted by 20. Photos are taken on substrates A and B2. The white halos correspond to the dew formed during the growth of the drop, while the black area indicates the place where water pinned and then slid. We show with a red dashed line a circle of radius R = 1.2 mm that highlights the radius of the trailing edge of the drop as it was pinned. The scale bar is the same for the two photos. NATURE MATERIALS 8
9 Images in Figure S6 correspond to the maximum contrast in size of departing drops in our experiments. However, the radius of the trailing edge is found to be almost the same, R = 1.2 ± 0.1 mm. Whatever the intensity of adhesion (either low as on A, where drops are quite axisymmetric, or high as on B, where they are strongly anisotropic), gravity thins the drops so that the size of the trailing edge can be roughly constant. 7. Comparison of the antifogging efficiency on Glaco and nanocones Material D is a silicon wafer treated three times by dip-coating it from a colloidal suspension (Glaco Mirror Coat Zero; Soft99) of hydrophobic silica beads of 15 nm radius dispersed in isopropanol. The resulting texture is a porous layer having a height h of typically 300 nm; atomic force microscopy shows cavities of maximum depth 100 nm. Advancing and receding contact angles of water on material D are θ a = 170 ± 2 and θ r = 164 ± 2, respectively. We performed with sample D a condensation experiment in the same conditions as for samples A and C, corresponding to Figure 4 in the accompanying paper. We plot in Figure S7 the percentage N of drops leaving the surface after a coalescence event on both samples D and C (nanocones). Ejections are observed on D (black data), with an average probability of 6%, hence much smaller than on C (red data). Droplets can be pinned inside the porous structure of material D, which explains a much smaller anti-fogging efficiency N (%) C D Figure S7. Time evolution of the percentage N of coalescence resulting in droplet ejection for sample C (red squares), corresponding to the nanocones of the accompanying paper (Figures 3 and 4), and for sample D (black diamonds) corresponding to the Glaco textures. Supersaturation S = 1.7 is the same in both cases. t (s) NATURE MATERIALS 9
10 8. Captions for the supplementary movies Supplementary Movie 1 Water adhesion on surfaces A (p = 52 nm) and B2 (p = 840 nm) for T = 0 C. Supplementary Movie 2 Water adhesion on surfaces A (p = 52 nm) and B2 (p = 840 nm) for T = 15 C. Supplementary Movie 3 Resistance to hot water jet on surface A (p = 52 nm) and B2 (p = 840 nm) for T = 0 and T = 15 C. Supplementary Movie 4 Water drops bouncing on surface A (p = 52 nm) and B2 (p = 840 nm) for T = 0 and T = 15 C. Supplementary Movie 5 Breath figure on sample A and sample C. Supersaturation S is maintained at a constant value 1.7 ± 0.1. The real duration of the experiment is 45 minutes. NATURE MATERIALS 10
Drop friction on liquid-infused materials
 Electronic Supplementary Material (ESI) for Soft Matter. This journal is The Royal Society of Chemistry 207 Drop friction on liquid-infused materials Armelle Gas,2, Ludovic Keiser,3, Christophe Clanet,2
Electronic Supplementary Material (ESI) for Soft Matter. This journal is The Royal Society of Chemistry 207 Drop friction on liquid-infused materials Armelle Gas,2, Ludovic Keiser,3, Christophe Clanet,2
On supercooled water drops impacting on superhydrophobic textures
 of On supercooled water drops impacting on superhydrophobic textures Tanmoy Maitra, Carlo Antonini, Manish K. Tiwari a, Adrian Mularczyk, Zulkufli Imeri, Philippe Schoch and imos Poulikakos * Laboratory
of On supercooled water drops impacting on superhydrophobic textures Tanmoy Maitra, Carlo Antonini, Manish K. Tiwari a, Adrian Mularczyk, Zulkufli Imeri, Philippe Schoch and imos Poulikakos * Laboratory
Microfluidics 2 Surface tension, contact angle, capillary flow
 MT-0.6081 Microfluidics and BioMEMS Microfluidics 2 Surface tension, contact angle, capillary flow 28.1.2017 Ville Jokinen Surface tension & Surface energy Work required to create new surface = surface
MT-0.6081 Microfluidics and BioMEMS Microfluidics 2 Surface tension, contact angle, capillary flow 28.1.2017 Ville Jokinen Surface tension & Surface energy Work required to create new surface = surface
Maximal deformation of an impacting drop
 J. Fluid Mech. (24), vol. 57, pp. 99 28. c 24 Cambridge University Press DOI:.7/S222494 Printed in the United Kingdom 99 Maximal deformation of an impacting drop By CHRISTOPHE CLANET, CÉDRIC BÉGUIN, DENIS
J. Fluid Mech. (24), vol. 57, pp. 99 28. c 24 Cambridge University Press DOI:.7/S222494 Printed in the United Kingdom 99 Maximal deformation of an impacting drop By CHRISTOPHE CLANET, CÉDRIC BÉGUIN, DENIS
Superhydrophobic surfaces. José Bico PMMH-ESPCI, Paris
 Superhydrophobic surfaces José Bico PMMH-ESPCI, Paris Superhydrophobic surfaces José Bico PMMH-ESPCI, Paris? Rain droplet on a window film pinning tear 180? mercury calefaction Leidenfrost point, T = 150
Superhydrophobic surfaces José Bico PMMH-ESPCI, Paris Superhydrophobic surfaces José Bico PMMH-ESPCI, Paris? Rain droplet on a window film pinning tear 180? mercury calefaction Leidenfrost point, T = 150
Droplet Migration during Condensation on Chemically Patterned. Micropillars
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry Please do 2016 not adjust margins RSC Advances ELECTRONIC SUPPORTING INFORMATION (ESI) Droplet Migration
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry Please do 2016 not adjust margins RSC Advances ELECTRONIC SUPPORTING INFORMATION (ESI) Droplet Migration
Supplementary Material
 1 2 3 Topological defects in confined populations of spindle-shaped cells by G. Duclos et al. Supplementary Material 4 5 6 7 8 9 10 11 12 13 Supplementary Note 1: Characteristic time associated with the
1 2 3 Topological defects in confined populations of spindle-shaped cells by G. Duclos et al. Supplementary Material 4 5 6 7 8 9 10 11 12 13 Supplementary Note 1: Characteristic time associated with the
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa.
 Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
MICRO GROOVED SURFACE IMPROVE DEW COLLECTION
 MICRO GROOVED SURFACE IMPROVE DEW COLLECTION Royon L.* Matière et Systèmes Complexes, CNRS UMR 7057, Univ. Paris Diderot, Paris Sorbonne Cité, F-75013, Paris, E-mail : laurent.royon@univ-paris-diderot.fr
MICRO GROOVED SURFACE IMPROVE DEW COLLECTION Royon L.* Matière et Systèmes Complexes, CNRS UMR 7057, Univ. Paris Diderot, Paris Sorbonne Cité, F-75013, Paris, E-mail : laurent.royon@univ-paris-diderot.fr
Supplementary table I. Table of contact angles of the different solutions on the surfaces used here. Supplementary Notes
 1 Supplementary Figure 1. Sketch of the experimental setup (not to scale) : it consists of a thin mylar sheet (0, 02 4 3cm 3 ) held fixed vertically. The spacing y 0 between the glass plate and the upper
1 Supplementary Figure 1. Sketch of the experimental setup (not to scale) : it consists of a thin mylar sheet (0, 02 4 3cm 3 ) held fixed vertically. The spacing y 0 between the glass plate and the upper
Contact time of a bouncing drop
 Contact time of a bouncing drop Denis Richard, Christophe Clanet (*) & David Quéré Laboratoire de Physique de la Matière Condensée, URA 792 du CNRS, Collège de France, 75231 Paris Cedex 05, France (*)
Contact time of a bouncing drop Denis Richard, Christophe Clanet (*) & David Quéré Laboratoire de Physique de la Matière Condensée, URA 792 du CNRS, Collège de France, 75231 Paris Cedex 05, France (*)
Supplementary Information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supplementary Information Visualization of equilibrium position of colloidal particles at fluid-water
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supplementary Information Visualization of equilibrium position of colloidal particles at fluid-water
Experimental and Theoretical Study of Motion of Drops on Horizontal Solid Surfaces with a Wettability Gradient Nadjoua Moumen
 Experimental and Theoretical Study of Motion of Drops on Horizontal Solid Surfaces with a Wettability Gradient Nadjoua Moumen Department of Chemical and Biomolecular Engineering Clarkson University Outline
Experimental and Theoretical Study of Motion of Drops on Horizontal Solid Surfaces with a Wettability Gradient Nadjoua Moumen Department of Chemical and Biomolecular Engineering Clarkson University Outline
Supporting information: Morphing and vectoring impacting droplets
 Supporting information: Morphing and vectoring impacting droplets by means of wettability-engineered surfaces Thomas M. Schutzius 1,2 Gustav Graeber 3 Mohamed Elsharkawy 1 James Oreluk 4 Constantine M.
Supporting information: Morphing and vectoring impacting droplets by means of wettability-engineered surfaces Thomas M. Schutzius 1,2 Gustav Graeber 3 Mohamed Elsharkawy 1 James Oreluk 4 Constantine M.
Supplementary Figure 1 Surface tension of polyelectrolyte solutions. Experimentally measured values of surface tension of the solutions that were
 Supplementary Figure 1 Surface tension of polyelectrolyte solutions. Experimentally measured values of surface tension of the solutions that were used in experiments throughout the paper. All surface tensions
Supplementary Figure 1 Surface tension of polyelectrolyte solutions. Experimentally measured values of surface tension of the solutions that were used in experiments throughout the paper. All surface tensions
Citation for published version (APA): Wal, B. P. V. D. (2006). Static and dynamic wetting of porous Teflon surfaces s.n.
 University of Groningen Static and dynamic wetting of porous Teflon surfaces Wal, Bouwe Pieter van der IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to
University of Groningen Static and dynamic wetting of porous Teflon surfaces Wal, Bouwe Pieter van der IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to
Surface and Interfacial Tensions. Lecture 1
 Surface and Interfacial Tensions Lecture 1 Surface tension is a pull Surfaces and Interfaces 1 Thermodynamics for Interfacial Systems Work must be done to increase surface area just as work must be done
Surface and Interfacial Tensions Lecture 1 Surface tension is a pull Surfaces and Interfaces 1 Thermodynamics for Interfacial Systems Work must be done to increase surface area just as work must be done
The evaporation of sessile droplets onto solid surfaces : experiments and simulations of the contact line pinning-depinning
 The evaporation of sessile droplets onto solid surfaces : experiments and simulations of the contact line pinning-depinning L.Kabeya-Mukeba, N.Vandewalle and S.Dorbolo GRASP, Institut de Physique B5a,
The evaporation of sessile droplets onto solid surfaces : experiments and simulations of the contact line pinning-depinning L.Kabeya-Mukeba, N.Vandewalle and S.Dorbolo GRASP, Institut de Physique B5a,
Supporting Information
 Supporting Information Yanshen Li a,b, David Quéré c, Cunjing Lv a,d, Quanshui Zheng a,b,e,1 a Department of Engineering Mechanics, Tsinghua University, Beijing 100084, China. b Center for Nano and Micro
Supporting Information Yanshen Li a,b, David Quéré c, Cunjing Lv a,d, Quanshui Zheng a,b,e,1 a Department of Engineering Mechanics, Tsinghua University, Beijing 100084, China. b Center for Nano and Micro
Colloidal Particles at Liquid Interfaces: An Introduction
 1 Colloidal Particles at Liquid Interfaces: An Introduction Bernard P. Binks and Tommy S. Horozov Surfactant and Colloid Group, Department of Chemistry, University of Hull, Hull, HU6 7RX, UK 1.1 Some Basic
1 Colloidal Particles at Liquid Interfaces: An Introduction Bernard P. Binks and Tommy S. Horozov Surfactant and Colloid Group, Department of Chemistry, University of Hull, Hull, HU6 7RX, UK 1.1 Some Basic
Capillarity and Wetting Phenomena
 ? Pierre-Gilles de Gennes Frangoise Brochard-Wyart David Quere Capillarity and Wetting Phenomena Drops, Bubbles, Pearls, Waves Translated by Axel Reisinger With 177 Figures Springer Springer New York Berlin
? Pierre-Gilles de Gennes Frangoise Brochard-Wyart David Quere Capillarity and Wetting Phenomena Drops, Bubbles, Pearls, Waves Translated by Axel Reisinger With 177 Figures Springer Springer New York Berlin
The effects of gravity on the capillary instability in tubes
 J. Fluid Mech. (2006), vol. 556, pp. 217 226. c 2006 Cambridge University Press doi:10.1017/s0022112006009505 Printed in the United Kingdom 217 The effects of gravity on the capillary instability in tubes
J. Fluid Mech. (2006), vol. 556, pp. 217 226. c 2006 Cambridge University Press doi:10.1017/s0022112006009505 Printed in the United Kingdom 217 The effects of gravity on the capillary instability in tubes
Supplementary Material
 Mangili et al. Supplementary Material 2 A. Evaluation of substrate Young modulus from AFM measurements 3 4 5 6 7 8 Using the experimental correlations between force and deformation from AFM measurements,
Mangili et al. Supplementary Material 2 A. Evaluation of substrate Young modulus from AFM measurements 3 4 5 6 7 8 Using the experimental correlations between force and deformation from AFM measurements,
Numerical Simulation of Drops Impacting on Textured Surfaces
 Numerical Simulation of Drops Impacting on Textured Surfaces Abstract Y.X. Wang, *S. Chen School of Aerospace Engineering and Applied Mechanics, Tongji University, China. *Corresponding author: schen_tju@tongji.edu.cn
Numerical Simulation of Drops Impacting on Textured Surfaces Abstract Y.X. Wang, *S. Chen School of Aerospace Engineering and Applied Mechanics, Tongji University, China. *Corresponding author: schen_tju@tongji.edu.cn
Topography driven spreading. School of Biomedical & Natural Sciences, Nottingham Trent University. Clifton Lane, Nottingham NG11 8NS, UK.
 Postprint Version G. McHale, N. J. Shirtcliffe, S. Aqil, C. C. Perry and M. I. Newton, Topography driven spreading, Phys. Rev. Lett. 93, Art. No. 036102 (2004); DOI: 10.1103/PhysRevLett.93.036102. The
Postprint Version G. McHale, N. J. Shirtcliffe, S. Aqil, C. C. Perry and M. I. Newton, Topography driven spreading, Phys. Rev. Lett. 93, Art. No. 036102 (2004); DOI: 10.1103/PhysRevLett.93.036102. The
spreading of drops on soft surfaces
 Supplementary Material on Electrically modulated dynamic spreading of drops on soft surfaces Ranabir Dey 1, Ashish Daga 1, Sunando DasGupta 2,3, Suman Chakraborty 1,3 1 Department of Mechanical Engineering,
Supplementary Material on Electrically modulated dynamic spreading of drops on soft surfaces Ranabir Dey 1, Ashish Daga 1, Sunando DasGupta 2,3, Suman Chakraborty 1,3 1 Department of Mechanical Engineering,
Hydrodynamics of wetting phenomena. Jacco Snoeijer PHYSICS OF FLUIDS
 Hydrodynamics of wetting phenomena Jacco Snoeijer PHYSICS OF FLUIDS Outline 1. Creeping flow: hydrodynamics at low Reynolds numbers (2 hrs) 2. Thin films and lubrication flows (3 hrs + problem session
Hydrodynamics of wetting phenomena Jacco Snoeijer PHYSICS OF FLUIDS Outline 1. Creeping flow: hydrodynamics at low Reynolds numbers (2 hrs) 2. Thin films and lubrication flows (3 hrs + problem session
SUPPLEMENTARY INFORMATION
 In the format provided by the authors and unedited. Phagocytosis- inspired behaviour in synthetic protocell communities of compartmentalized colloidal objects Laura Rodríguez- Arco, Mei Li and Stephen
In the format provided by the authors and unedited. Phagocytosis- inspired behaviour in synthetic protocell communities of compartmentalized colloidal objects Laura Rodríguez- Arco, Mei Li and Stephen
Reaction at the Interfaces
 Reaction at the Interfaces Lecture 1 On the course Physics and Chemistry of Interfaces by HansJürgen Butt, Karlheinz Graf, and Michael Kappl Wiley VCH; 2nd edition (2006) http://homes.nano.aau.dk/lg/surface2009.htm
Reaction at the Interfaces Lecture 1 On the course Physics and Chemistry of Interfaces by HansJürgen Butt, Karlheinz Graf, and Michael Kappl Wiley VCH; 2nd edition (2006) http://homes.nano.aau.dk/lg/surface2009.htm
Capturing drops with a thin fiber
 Journal of Colloid and Interface Science 279 (2004) 192 197 www.elsevier.com/locate/jcis Capturing drops with a thin fiber Élise Lorenceau a, Christophe Clanet b, David Quéré a, a Laboratoire de Physique
Journal of Colloid and Interface Science 279 (2004) 192 197 www.elsevier.com/locate/jcis Capturing drops with a thin fiber Élise Lorenceau a, Christophe Clanet b, David Quéré a, a Laboratoire de Physique
Supplementary Figures
 Supplementary Figures Supplementary Figure S1. The effect of window size. The phonon MFP spectrum of intrinsic c-si (T=300 K) is shown for 7-point, 13-point, and 19-point windows. Increasing the window
Supplementary Figures Supplementary Figure S1. The effect of window size. The phonon MFP spectrum of intrinsic c-si (T=300 K) is shown for 7-point, 13-point, and 19-point windows. Increasing the window
CENG 501 Examination Problem: Estimation of Viscosity with a Falling - Cylinder Viscometer
 CENG 501 Examination Problem: Estimation of Viscosity with a Falling - Cylinder Viscometer You are assigned to design a fallingcylinder viscometer to measure the viscosity of Newtonian liquids. A schematic
CENG 501 Examination Problem: Estimation of Viscosity with a Falling - Cylinder Viscometer You are assigned to design a fallingcylinder viscometer to measure the viscosity of Newtonian liquids. A schematic
Supporting Information
 Supporting Information On the Minimal Size of Coffee Ring Structure Xiaoying Shen, Chih-Ming Ho and Tak-Sing Wong * Mechanical and Aerospace Engineering Department, University of California, Los Angeles,
Supporting Information On the Minimal Size of Coffee Ring Structure Xiaoying Shen, Chih-Ming Ho and Tak-Sing Wong * Mechanical and Aerospace Engineering Department, University of California, Los Angeles,
J. Bico, C. Tordeux and D. Quéré Laboratoire de Physique de la Matière Condensée, URA 792 du CNRS Collège de France Paris Cedex 05, France
 EUROPHYSICS LETTERS 15 July 2001 Europhys. Lett., 55 (2), pp. 214 220 (2001) Rough wetting J. Bico, C. Tordeux and D. Quéré Laboratoire de Physique de la Matière Condensée, URA 792 du CNRS Collège de France
EUROPHYSICS LETTERS 15 July 2001 Europhys. Lett., 55 (2), pp. 214 220 (2001) Rough wetting J. Bico, C. Tordeux and D. Quéré Laboratoire de Physique de la Matière Condensée, URA 792 du CNRS Collège de France
Surface Tension and its measurements
 Surface Tension and its measurements Surface Tension Surface tension is a fundamental property by which the gas liquid interfaces are characterized. The zone between a gaseous phase and a liquid phase
Surface Tension and its measurements Surface Tension Surface tension is a fundamental property by which the gas liquid interfaces are characterized. The zone between a gaseous phase and a liquid phase
emulsions, and foams March 21 22, 2009
 Wetting and adhesion Dispersions in liquids: suspensions, emulsions, and foams ACS National Meeting March 21 22, 2009 Salt Lake City Ian Morrison 2009 Ian Morrison 2009 Lecure 2 - Wetting and adhesion
Wetting and adhesion Dispersions in liquids: suspensions, emulsions, and foams ACS National Meeting March 21 22, 2009 Salt Lake City Ian Morrison 2009 Ian Morrison 2009 Lecure 2 - Wetting and adhesion
Praktikum zur. Materialanalytik
 Praktikum zur Materialanalytik Functionalized Surfaces B510 Stand: 20.10.2017 Table of contents Introduction 2 Basics 2 Surface tension 2 From wettability to the contact angle 4 The Young equation 5 Wetting
Praktikum zur Materialanalytik Functionalized Surfaces B510 Stand: 20.10.2017 Table of contents Introduction 2 Basics 2 Surface tension 2 From wettability to the contact angle 4 The Young equation 5 Wetting
Resonant Oscillations of Liquid Marbles
 Resonant Oscillations of Liquid Marbles Glen McHale*, Stephen J. Elliott*, Michael I. Newton*, Dale L. Herbertson* and Kadir Esmer $ *School of Science & Technology, Nottingham Trent University, UK $ Department
Resonant Oscillations of Liquid Marbles Glen McHale*, Stephen J. Elliott*, Michael I. Newton*, Dale L. Herbertson* and Kadir Esmer $ *School of Science & Technology, Nottingham Trent University, UK $ Department
Supplementary Figures
 Supplementary Figures 1 Supplementary Figure 1 Micro and nano-textured boiling surfaces. (a) A schematic of the textured boiling surfaces. (b) An isometric view of the square array of square micropillars.
Supplementary Figures 1 Supplementary Figure 1 Micro and nano-textured boiling surfaces. (a) A schematic of the textured boiling surfaces. (b) An isometric view of the square array of square micropillars.
AN OPTIMAL CURVE FOR FASTEST TRANSPROTATION OF LIQUID DROPS ON A SUPERHYDROPHOBIC SURFACE
 AN OPTIMAL CURVE FOR FASTEST TRANSPROTATION OF LIQUID DROPS ON A SUPERHYDROPHOBIC SURFACE ABSTRACT Kwangseok Seo, Minyoung Kim, Do Hyun Kim Department of Chemical and Biomolecular Engineering, Korea Advanced
AN OPTIMAL CURVE FOR FASTEST TRANSPROTATION OF LIQUID DROPS ON A SUPERHYDROPHOBIC SURFACE ABSTRACT Kwangseok Seo, Minyoung Kim, Do Hyun Kim Department of Chemical and Biomolecular Engineering, Korea Advanced
Supplementary Information on Thermally Enhanced Self-Propelled Droplet Motion on Gradient Surfaces
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supplementary Information on Thermally Enhanced Self-Propelled Droplet Motion on Gradient Surfaces
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supplementary Information on Thermally Enhanced Self-Propelled Droplet Motion on Gradient Surfaces
Thickness and Shape of Films Driven by a Marangoni Flow
 Langmuir 1996, 12, 5875-5880 5875 Thickness and Shape of Films Driven by a Marangoni Flow X. Fanton, A. M. Cazabat,* and D. Quéré Laboratoire de Physique de la Matière Condensée, Collège de France, 11
Langmuir 1996, 12, 5875-5880 5875 Thickness and Shape of Films Driven by a Marangoni Flow X. Fanton, A. M. Cazabat,* and D. Quéré Laboratoire de Physique de la Matière Condensée, Collège de France, 11
Relaxation Effects in the Modeling of Gradient Stresses
 Relaxation Effects in the Modeling of Gradient Stresses Daniel D. Joseph 1 The topics being discussed here are the physics and modeling of stresses due to gradients of composition volume fraction of solute
Relaxation Effects in the Modeling of Gradient Stresses Daniel D. Joseph 1 The topics being discussed here are the physics and modeling of stresses due to gradients of composition volume fraction of solute
L = I ω = const. I = 2 3 MR2. When the balloon shrinks (because the air inside it cools down), the moment of inertia decreases, R = 1. L = I ω = I ω.
 PHY 30 K. Solutions for mid-term test #3. Problem 1: Out in space, there are no forces acting on the balloon except gravity and hence no torques (with respect to an axis through the center of mass). Consequently,
PHY 30 K. Solutions for mid-term test #3. Problem 1: Out in space, there are no forces acting on the balloon except gravity and hence no torques (with respect to an axis through the center of mass). Consequently,
Tanmoy Maitra. Superhydrophobicity to Supericephobicity: A technological Challenge
 Superhydrophobicity to Supericephobicity: A technological Challenge Tanmoy Maitra Laboratory of Thermodynamics in Emerging Technologies Mechanical & Process Engineering 1 Icing in aeronautics www.staralliance.com/int/press/media_library/images/
Superhydrophobicity to Supericephobicity: A technological Challenge Tanmoy Maitra Laboratory of Thermodynamics in Emerging Technologies Mechanical & Process Engineering 1 Icing in aeronautics www.staralliance.com/int/press/media_library/images/
Universal Self-assembly of Organosilanes with Long Alkyl Groups
 Supporting Information for Universal Self-assembly of Organosilanes with Long Alkyl Groups into Silicone Nanofilaments Junping Zhang,, * Aiqin Wang, and Stefan Seeger Center of Eco-material and Green Chemistry,
Supporting Information for Universal Self-assembly of Organosilanes with Long Alkyl Groups into Silicone Nanofilaments Junping Zhang,, * Aiqin Wang, and Stefan Seeger Center of Eco-material and Green Chemistry,
WAVE STRUCTURES ON A JET ENTERING THE BULK LIQUID HE YIJUN; LI YOUXI; ZHU ZIJIA; LIN LIANG; CORKILL ASHLEY CHRISTINE
 WAVE STRUCTURES ON A JET ENTERING THE BULK LIQUID HE YIJUN; LI YOUXI; ZHU ZIJIA; LIN LIANG; CORKILL ASHLEY CHRISTINE INTRODUCTION When a jet impacts the liquid surface, there will be wave on the jet. Such
WAVE STRUCTURES ON A JET ENTERING THE BULK LIQUID HE YIJUN; LI YOUXI; ZHU ZIJIA; LIN LIANG; CORKILL ASHLEY CHRISTINE INTRODUCTION When a jet impacts the liquid surface, there will be wave on the jet. Such
PHYSICS OF FLUID SPREADING ON ROUGH SURFACES
 INTERNATIONAL JOURNAL OF NUMERICAL ANALYSIS AND MODELING Volume 5, Supp, Pages 85 92 c 2008 Institute for Scientific Computing and Information PHYSICS OF FLUID SPREADING ON ROUGH SURFACES K. M. HAY AND
INTERNATIONAL JOURNAL OF NUMERICAL ANALYSIS AND MODELING Volume 5, Supp, Pages 85 92 c 2008 Institute for Scientific Computing and Information PHYSICS OF FLUID SPREADING ON ROUGH SURFACES K. M. HAY AND
Supplementary material to On the rheology of pendular gels and morphological developments in paste- like ternary systems based on capillary attraction
 Electronic Supplementary Material (ESI) for Soft Matter. This journal is The Royal Society of Chemistry 214 Supplementary material to On the rheology of pendular gels and morphological developments in
Electronic Supplementary Material (ESI) for Soft Matter. This journal is The Royal Society of Chemistry 214 Supplementary material to On the rheology of pendular gels and morphological developments in
Lubricant Impregnated Nanotextured Surfaces
 Supporting Information: Enhanced Condensation on Lubricant Impregnated Nanotextured Surfaces Sushant Anand, Adam T. Paxson, Rajeev Dhiman, J. David Smith, Kripa K. Varanasi* Department of Mechanical Engineering,
Supporting Information: Enhanced Condensation on Lubricant Impregnated Nanotextured Surfaces Sushant Anand, Adam T. Paxson, Rajeev Dhiman, J. David Smith, Kripa K. Varanasi* Department of Mechanical Engineering,
arxiv: v1 [cond-mat.soft] 21 Oct 2016
![arxiv: v1 [cond-mat.soft] 21 Oct 2016 arxiv: v1 [cond-mat.soft] 21 Oct 2016](/thumbs/92/110492609.jpg) Contact angles of a drop pinned on an incline Joe l De Coninck Laboratoire de Physique des Surfaces et Interfaces Universite de Mons, Place du Parc, 7 Mons, Belgium Franc ois Dunlop and Thierry Huillet
Contact angles of a drop pinned on an incline Joe l De Coninck Laboratoire de Physique des Surfaces et Interfaces Universite de Mons, Place du Parc, 7 Mons, Belgium Franc ois Dunlop and Thierry Huillet
Lecture 7 Contact angle phenomena and wetting
 Lecture 7 Contact angle phenomena and Contact angle phenomena and wetting Young s equation Drop on the surface complete spreading Establishing finite contact angle γ cosθ = γ γ L S SL γ S γ > 0 partial
Lecture 7 Contact angle phenomena and Contact angle phenomena and wetting Young s equation Drop on the surface complete spreading Establishing finite contact angle γ cosθ = γ γ L S SL γ S γ > 0 partial
1) Institut d Electronique Fondamentale, CNRS, Univ. Paris- Sud, Université Paris- Saclay, Bâtiment 220, Rue André Ampère, F Orsay, France
 Supporting information Direct band gap germanium microdisks obtained with silicon nitride stressor layers Moustafa El Kurdi, 1 Mathias Prost, 1 Abdelhamid Ghrib, 1 Sébastien Sauvage, 1 Xavier Checoury,
Supporting information Direct band gap germanium microdisks obtained with silicon nitride stressor layers Moustafa El Kurdi, 1 Mathias Prost, 1 Abdelhamid Ghrib, 1 Sébastien Sauvage, 1 Xavier Checoury,
Supplementary Information. In colloidal drop drying processes, multi-ring depositions are formed due to the stick-slip
 Electronic Supplementary Material (ESI for Soft Matter. This journal is The Royal Society of Chemistry 14 Supplementary Information A1. Contact line receding velocity of an evaporating drop In colloidal
Electronic Supplementary Material (ESI for Soft Matter. This journal is The Royal Society of Chemistry 14 Supplementary Information A1. Contact line receding velocity of an evaporating drop In colloidal
Vibration of submillimeter-size supported droplets
 PHYSICAL REVIEW E 73, 041602 2006 Vibration of submillimeter-size supported droplets Franck Celestini* and Richard Kofman Laboratoire de Physique de la Matière Condensée, UMR 6622, CNRS, Université de
PHYSICAL REVIEW E 73, 041602 2006 Vibration of submillimeter-size supported droplets Franck Celestini* and Richard Kofman Laboratoire de Physique de la Matière Condensée, UMR 6622, CNRS, Université de
Wetting contact angle
 Wetting contact angle Minh Do-Quang www.flow.kth.se Outline Statics; capillarity and wetting Dynamics; models describing dynamic wetting Hydrodynamics (Tanner-Cox-Voinov law) Molecular kinetics theory
Wetting contact angle Minh Do-Quang www.flow.kth.se Outline Statics; capillarity and wetting Dynamics; models describing dynamic wetting Hydrodynamics (Tanner-Cox-Voinov law) Molecular kinetics theory
Sta$s$cal mechanics of hystere$c capillary phenomena: predic$ons of contact angle on rough surfaces and liquid reten$on in unsaturated porous media
 Sta$s$cal mechanics of hystere$c capillary phenomena: predic$ons of contact angle on rough surfaces and liquid reten$on in unsaturated porous media Michel Louge h@p://grainflowresearch.mae.cornell.edu/
Sta$s$cal mechanics of hystere$c capillary phenomena: predic$ons of contact angle on rough surfaces and liquid reten$on in unsaturated porous media Michel Louge h@p://grainflowresearch.mae.cornell.edu/
A SCIENTIFIC APPROACH TO A STICKY PROBLEM
 A SCIENTIFIC APPROACH TO A STICKY PROBLEM Sticking, the adherence of granule to punch face or die bore, is one of the major issues affecting the manufacture of solid dose pharmaceuticals. As part of I
A SCIENTIFIC APPROACH TO A STICKY PROBLEM Sticking, the adherence of granule to punch face or die bore, is one of the major issues affecting the manufacture of solid dose pharmaceuticals. As part of I
Impalement of fakir drops
 January 2008 EPL, 81 (2008) 26006 doi: 10.1209/0295-5075/81/26006 www.epljournal.org Impalement of fakir drops M. Reyssat 1,J.M.Yeomans 2 and D. Quéré 1 1 Physique et Mécanique des Milieux Hétérogènes,
January 2008 EPL, 81 (2008) 26006 doi: 10.1209/0295-5075/81/26006 www.epljournal.org Impalement of fakir drops M. Reyssat 1,J.M.Yeomans 2 and D. Quéré 1 1 Physique et Mécanique des Milieux Hétérogènes,
Boiling crisis dynamics: low gravity experiments and modeling
 Boiling crisis dynamics: low gravity experiments and modeling V. Nikolayev (1), Y. Garrabos (2), C. Lecoutre (2), D. Hitz (3), D. Chatain (3), R. Guillaument (2), V. Janeček (4,5), D. Beysens (3,4) (1)
Boiling crisis dynamics: low gravity experiments and modeling V. Nikolayev (1), Y. Garrabos (2), C. Lecoutre (2), D. Hitz (3), D. Chatain (3), R. Guillaument (2), V. Janeček (4,5), D. Beysens (3,4) (1)
arxiv:cond-mat/ v1 [cond-mat.soft] 18 Sep 2000
![arxiv:cond-mat/ v1 [cond-mat.soft] 18 Sep 2000 arxiv:cond-mat/ v1 [cond-mat.soft] 18 Sep 2000](/thumbs/81/83362442.jpg) arxiv:cond-mat/0009259v1 [cond-mat.soft] 18 Sep 2000 Two remarks on wetting and emulsions P. G. de Gennes Collège de France, 11 place M. Berthelot, 75231 Paris Cedex, France June 12, 2018 Abstract This
arxiv:cond-mat/0009259v1 [cond-mat.soft] 18 Sep 2000 Two remarks on wetting and emulsions P. G. de Gennes Collège de France, 11 place M. Berthelot, 75231 Paris Cedex, France June 12, 2018 Abstract This
Driven large contact angle droplets on chemically heterogeneous substrates
 October 2012 EPL, 100 (2012) 16002 doi: 10.1209/0295-5075/100/16002 www.epljournal.org Driven large contact angle droplets on chemically heterogeneous substrates D. Herde 1,U.Thiele 2,S.Herminghaus 1 and
October 2012 EPL, 100 (2012) 16002 doi: 10.1209/0295-5075/100/16002 www.epljournal.org Driven large contact angle droplets on chemically heterogeneous substrates D. Herde 1,U.Thiele 2,S.Herminghaus 1 and
Generalized Cassie-Baxter equation for wetting of a spherical droplet within a smooth and heterogeneous conical cavity
 Science Front Publishers Journal for Foundations and pplications of Physics, vol. 4, No. (017) (sciencefront.org) ISSN 394-3688 Generalized Cassie-axter equation for wetting of a spherical droplet within
Science Front Publishers Journal for Foundations and pplications of Physics, vol. 4, No. (017) (sciencefront.org) ISSN 394-3688 Generalized Cassie-axter equation for wetting of a spherical droplet within
cavity (inner rectangular mark) in the frozen decane (dark background) is clearly visible.
 Supplementary Figure S1. Low magnification image showing one half of the sandwich holder from the top. The rectangular print left by the water droplet contained in the holder cavity (inner rectangular
Supplementary Figure S1. Low magnification image showing one half of the sandwich holder from the top. The rectangular print left by the water droplet contained in the holder cavity (inner rectangular
Chapter 10. Solids and Fluids
 Chapter 10 Solids and Fluids Surface Tension Net force on molecule A is zero Pulled equally in all directions Net force on B is not zero No molecules above to act on it Pulled toward the center of the
Chapter 10 Solids and Fluids Surface Tension Net force on molecule A is zero Pulled equally in all directions Net force on B is not zero No molecules above to act on it Pulled toward the center of the
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/3/10/eaao3530/dc1 Supplementary Materials for Topological liquid diode Jiaqian Li, Xiaofeng Zhou, Jing Li, Lufeng Che, Jun Yao, Glen McHale, Manoj K. Chaudhury,
advances.sciencemag.org/cgi/content/full/3/10/eaao3530/dc1 Supplementary Materials for Topological liquid diode Jiaqian Li, Xiaofeng Zhou, Jing Li, Lufeng Che, Jun Yao, Glen McHale, Manoj K. Chaudhury,
DLVO interaction between the spheres
 DLVO interaction between the spheres DL-interaction energy for two spheres: D w ( x) 64c π ktrϕ e λ DL 2 x λ 2 0 0 D DLVO interaction w ( x) 64πkTRϕ e λ DLVO AR /12x 2 x λd 2 0 D Lecture 11 Contact angle
DLVO interaction between the spheres DL-interaction energy for two spheres: D w ( x) 64c π ktrϕ e λ DL 2 x λ 2 0 0 D DLVO interaction w ( x) 64πkTRϕ e λ DLVO AR /12x 2 x λd 2 0 D Lecture 11 Contact angle
The Wilhelmy balance. How can we measure surface tension? Surface tension, contact angles and wettability. Measuring surface tension.
 ow can we measure surface tension? Surface tension, contact angles and wettability www.wikihow.com/measure-surface-tension Measuring surface tension The Wilhelmy balance F Some methods: Wilhelmy plate
ow can we measure surface tension? Surface tension, contact angles and wettability www.wikihow.com/measure-surface-tension Measuring surface tension The Wilhelmy balance F Some methods: Wilhelmy plate
ESS 431 Principles of Glaciology ESS 505 The Cryosphere
 MID-TERM November 9, 2015 ESS 431 Principles of Glaciology ESS 505 The Cryosphere Instructions: Please answer the following 5 questions. [The actual 5 questions will be selected from these 12 questions
MID-TERM November 9, 2015 ESS 431 Principles of Glaciology ESS 505 The Cryosphere Instructions: Please answer the following 5 questions. [The actual 5 questions will be selected from these 12 questions
arxiv: v2 [physics.flu-dyn] 24 Oct 2013
![arxiv: v2 [physics.flu-dyn] 24 Oct 2013 arxiv: v2 [physics.flu-dyn] 24 Oct 2013](/thumbs/73/68817731.jpg) Supporting Information: Short and long time drop dynamics on lubricated substrates Andreas Carlson1,3,4,, Pilnam Kim2,4, Gustav Amberg3, and Howard A. Stone4, arxiv:1309.6339v2 [physics.flu-dyn] 24 Oct
Supporting Information: Short and long time drop dynamics on lubricated substrates Andreas Carlson1,3,4,, Pilnam Kim2,4, Gustav Amberg3, and Howard A. Stone4, arxiv:1309.6339v2 [physics.flu-dyn] 24 Oct
contact line dynamics
 contact line dynamics part 2: hydrodynamics dynamic contact angle? lubrication: Cox-Voinov theory maximum speed for instability corner shape? dimensional analysis: speed U position r viscosity η pressure
contact line dynamics part 2: hydrodynamics dynamic contact angle? lubrication: Cox-Voinov theory maximum speed for instability corner shape? dimensional analysis: speed U position r viscosity η pressure
Self-cleaning of hydrophobic rough surfaces by coalescence-induced wetting transition
 arxiv:1810.13073v1 [physics.comp-ph] 31 Oct 2018 Self-cleaning of hydrophobic rough surfaces by coalescence-induced wetting transition Kaixuan Zhang 1,2, Zhen Li 2,,, Martin Maxey 2, Shuo Chen 1, and George
arxiv:1810.13073v1 [physics.comp-ph] 31 Oct 2018 Self-cleaning of hydrophobic rough surfaces by coalescence-induced wetting transition Kaixuan Zhang 1,2, Zhen Li 2,,, Martin Maxey 2, Shuo Chen 1, and George
Supporting Information: On Localized Vapor Pressure Gradients Governing Condensation and Frost Phenomena
 Supporting Information: On Localized Vapor Pressure Gradients Governing Condensation and Frost Phenomena Saurabh Nath and Jonathan B. Boreyko Department of Biomedical Engineering and Mechanics, Virginia
Supporting Information: On Localized Vapor Pressure Gradients Governing Condensation and Frost Phenomena Saurabh Nath and Jonathan B. Boreyko Department of Biomedical Engineering and Mechanics, Virginia
Surface forces action in a vicinity of three phase contact line and other current problems in kinetics of wetting and spreading
 Loughborough University Institutional Repository Surface forces action in a vicinity of three phase contact line and other current problems in kinetics of wetting and spreading This item was submitted
Loughborough University Institutional Repository Surface forces action in a vicinity of three phase contact line and other current problems in kinetics of wetting and spreading This item was submitted
Multifunctionality and control of the crumpling and unfolding of
 Addendum notice Nature Mater. 12, 321 325 (2013) Multifunctionality and control of the crumpling and unfolding of large-area graphene Jianfeng Zang, Seunghwa Ryu, Nicola Pugno, QimingWang, Qing Tu, Markus
Addendum notice Nature Mater. 12, 321 325 (2013) Multifunctionality and control of the crumpling and unfolding of large-area graphene Jianfeng Zang, Seunghwa Ryu, Nicola Pugno, QimingWang, Qing Tu, Markus
Line Tension Effect upon Static Wetting
 Line Tension Effect upon Static Wetting Pierre SEPPECHER Université de Toulon et du Var, BP 132 La Garde Cedex seppecher@univ tln.fr Abstract. Adding simply, in the classical capillary model, a constant
Line Tension Effect upon Static Wetting Pierre SEPPECHER Université de Toulon et du Var, BP 132 La Garde Cedex seppecher@univ tln.fr Abstract. Adding simply, in the classical capillary model, a constant
Köhler Curve. Covers Reading Material in Chapter 10.3 Atmospheric Sciences 5200 Physical Meteorology III: Cloud Physics
 Köhler Curve Covers Reading Material in Chapter 10.3 Atmospheric Sciences 5200 Physical Meteorology III: Cloud Physics Review of Kelvin Effect Gibbs Energy for formation of a drop G = G &'()*+, G ).'+
Köhler Curve Covers Reading Material in Chapter 10.3 Atmospheric Sciences 5200 Physical Meteorology III: Cloud Physics Review of Kelvin Effect Gibbs Energy for formation of a drop G = G &'()*+, G ).'+
Supporting Information
 Supporting Information Superhydrophobic Coatings: Are They Really Ice-Repellent? S.A. Kulinich a,b, S. Farhadi a, K. Nose c, X.W. Du d a Department of Applied Sciences, University of Quebec, Saguenay,
Supporting Information Superhydrophobic Coatings: Are They Really Ice-Repellent? S.A. Kulinich a,b, S. Farhadi a, K. Nose c, X.W. Du d a Department of Applied Sciences, University of Quebec, Saguenay,
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10447 Supplementary Methods Materials. The lubricating fluids used for the experiment were perfluorinated fluids (e.g., 3M Fluorinert FC-70, Dupont Krytox 100 and 103). Unless otherwise
doi:10.1038/nature10447 Supplementary Methods Materials. The lubricating fluids used for the experiment were perfluorinated fluids (e.g., 3M Fluorinert FC-70, Dupont Krytox 100 and 103). Unless otherwise
Supplementary Information
 Supplementary Information Under-water Superoleophobic Glass: Unexplored role of the surfactant-rich solvent Authors: Prashant R. Waghmare, Siddhartha Das, Sushanta K. Mitra * Affiliation: Micro and Nano-scale
Supplementary Information Under-water Superoleophobic Glass: Unexplored role of the surfactant-rich solvent Authors: Prashant R. Waghmare, Siddhartha Das, Sushanta K. Mitra * Affiliation: Micro and Nano-scale
Physics 202 Exam 1. May 1, 2013
 Name: Physics 202 Exam 1 May 1, 2013 Word Problems Show all your work and circle your final answer. (Ten points each.) 1. If 2.4 m 3 of a gas initially at STP is compressed to 1.6 m 3 and its temperature
Name: Physics 202 Exam 1 May 1, 2013 Word Problems Show all your work and circle your final answer. (Ten points each.) 1. If 2.4 m 3 of a gas initially at STP is compressed to 1.6 m 3 and its temperature
Basic Laboratory. Materials Science and Engineering. Atomic Force Microscopy (AFM)
 Basic Laboratory Materials Science and Engineering Atomic Force Microscopy (AFM) M108 Stand: 20.10.2015 Aim: Presentation of an application of the AFM for studying surface morphology. Inhalt 1.Introduction...
Basic Laboratory Materials Science and Engineering Atomic Force Microscopy (AFM) M108 Stand: 20.10.2015 Aim: Presentation of an application of the AFM for studying surface morphology. Inhalt 1.Introduction...
Gravitational effects on the deformation of a droplet adhering to a horizontal solid surface in shear flow
 PHYSICS OF FLUIDS 19, 122105 2007 Gravitational effects on the deformation of a droplet adhering to a horizontal solid surface in shear flow P. Dimitrakopoulos Department of Chemical and Biomolecular Engineering,
PHYSICS OF FLUIDS 19, 122105 2007 Gravitational effects on the deformation of a droplet adhering to a horizontal solid surface in shear flow P. Dimitrakopoulos Department of Chemical and Biomolecular Engineering,
FLOATING WITH SURFACE TENSION
 FLOATING WITH SURFACE TENSION FROM ARCHIMEDES TO KELLER Dominic Vella Mathematical Institute, University of Oxford JBK @ WHOI Joe was a regular fixture of the Geophysical Fluid Dynamics programme (run
FLOATING WITH SURFACE TENSION FROM ARCHIMEDES TO KELLER Dominic Vella Mathematical Institute, University of Oxford JBK @ WHOI Joe was a regular fixture of the Geophysical Fluid Dynamics programme (run
8.012 Physics I: Classical Mechanics Fall 2008
 MIT OpenCourseWare http://ocw.mit.edu 8.012 Physics I: Classical Mechanics Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. MASSACHUSETTS INSTITUTE
MIT OpenCourseWare http://ocw.mit.edu 8.012 Physics I: Classical Mechanics Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. MASSACHUSETTS INSTITUTE
Comptes Rendus Mecanique
 C. R. Mecanique 338 (2010) 12 17 Contents lists available at ScienceDirect Comptes Rendus Mecanique www.sciencedirect.com Vortex-induced vibration of a square cylinder in wind tunnel Xavier Amandolèse
C. R. Mecanique 338 (2010) 12 17 Contents lists available at ScienceDirect Comptes Rendus Mecanique www.sciencedirect.com Vortex-induced vibration of a square cylinder in wind tunnel Xavier Amandolèse
Brownian diffusion of a partially wetted colloid
 SUPPLEMENTARY INFORMATION DOI: 1.138/NMAT4348 Brownian diffusion of a partially wetted colloid Giuseppe Boniello, Christophe Blanc, Denys Fedorenko, Mayssa Medfai, Nadia Ben Mbarek, Martin In, Michel Gross,
SUPPLEMENTARY INFORMATION DOI: 1.138/NMAT4348 Brownian diffusion of a partially wetted colloid Giuseppe Boniello, Christophe Blanc, Denys Fedorenko, Mayssa Medfai, Nadia Ben Mbarek, Martin In, Michel Gross,
SUPPLEMENTARY INFORMATION
 SUPPLMNTARY INFORMATION Hindered rolling and friction anisotropy in supported carbon nanotubes Marcel Lucas 1, Xiaohua Zhang +, Ismael Palaci 1, Christian Klinke 3, rio Tosatti, 4 * and lisa Riedo 1 *
SUPPLMNTARY INFORMATION Hindered rolling and friction anisotropy in supported carbon nanotubes Marcel Lucas 1, Xiaohua Zhang +, Ismael Palaci 1, Christian Klinke 3, rio Tosatti, 4 * and lisa Riedo 1 *
Temperature ( o C)
 Viscosity (Pa sec) Supplementary Information 10 8 10 6 10 4 10 2 150 200 250 300 Temperature ( o C) Supplementary Figure 1 Viscosity of fibre components (PC cladding blue; As 2 Se 5 red; CPE black) as
Viscosity (Pa sec) Supplementary Information 10 8 10 6 10 4 10 2 150 200 250 300 Temperature ( o C) Supplementary Figure 1 Viscosity of fibre components (PC cladding blue; As 2 Se 5 red; CPE black) as
Ultrafast water harvesting and transport in hierarchical microchannels
 SUPPLEMENTARY INFORMATION Articles https://doi.org/10.1038/s41563-018-0171-9 In the format provided by the authors and unedited. Ultrafast water harvesting and transport in hierarchical microchannels Huawei
SUPPLEMENTARY INFORMATION Articles https://doi.org/10.1038/s41563-018-0171-9 In the format provided by the authors and unedited. Ultrafast water harvesting and transport in hierarchical microchannels Huawei
Mohamed Daoud Claudine E.Williams Editors. Soft Matter Physics. With 177 Figures, 16 of them in colour
 Mohamed Daoud Claudine E.Williams Editors Soft Matter Physics With 177 Figures, 16 of them in colour Contents 1. Droplets: CapiUarity and Wetting 1 By F. Brochard-Wyart (With 35 figures) 1.1 Introduction
Mohamed Daoud Claudine E.Williams Editors Soft Matter Physics With 177 Figures, 16 of them in colour Contents 1. Droplets: CapiUarity and Wetting 1 By F. Brochard-Wyart (With 35 figures) 1.1 Introduction
Droplet Coalescence and Freezing on Hydrophilic, Hydrophobic, and Biphilic Surfaces
 Droplet Coalescence and Freezing on Hydrophilic, Hydrophobic, and Biphilic Surfaces Abstract Alexander S. Van Dyke 1, Diane Collard 2, Melanie M. Derby 1, Amy Rachel Betz 1 * 1 Mechanical and Nuclear Engineering,
Droplet Coalescence and Freezing on Hydrophilic, Hydrophobic, and Biphilic Surfaces Abstract Alexander S. Van Dyke 1, Diane Collard 2, Melanie M. Derby 1, Amy Rachel Betz 1 * 1 Mechanical and Nuclear Engineering,
Capillarity induced folding of elastic sheets
 Eur. Phys. J. Special Topics 166, 67 71 (2009) c EDP Sciences, Springer-Verlag 2009 DOI: 10.1140/epjst/e2009-00880-4 Regular Article THE EUROPEAN PHYSICAL JOURNAL SPECIAL TOPICS Capillarity induced folding
Eur. Phys. J. Special Topics 166, 67 71 (2009) c EDP Sciences, Springer-Verlag 2009 DOI: 10.1140/epjst/e2009-00880-4 Regular Article THE EUROPEAN PHYSICAL JOURNAL SPECIAL TOPICS Capillarity induced folding
DROPWISE CONDENSATION
 DROPWISE CONDENSATION Davide Del Col Università di Padova Dipartimento di Ingegneria Industriale Via Venezia, 1-35131 Padova E-mail: davide.delcol@unipd.i http://stet.dii.unipd.it/ Outline Surface wettability
DROPWISE CONDENSATION Davide Del Col Università di Padova Dipartimento di Ingegneria Industriale Via Venezia, 1-35131 Padova E-mail: davide.delcol@unipd.i http://stet.dii.unipd.it/ Outline Surface wettability
Anti-icing surfaces based on enhanced self-propelled jumping of condensed water microdroplets
 Anti-icing surfaces based on enhanced self-propelled jumping of condensed water microdroplets Qiaolan Zhang, a,b Min He, a Jing Chen, a,b Jianjun Wang,* a Yanlin Song* a and Lei Jiang a a Beijing National
Anti-icing surfaces based on enhanced self-propelled jumping of condensed water microdroplets Qiaolan Zhang, a,b Min He, a Jing Chen, a,b Jianjun Wang,* a Yanlin Song* a and Lei Jiang a a Beijing National
Supplementary Figure 1: Micromechanical cleavage of graphene on oxygen plasma treated Si/SiO2. Supplementary Figure 2: Comparison of hbn yield.
 1 2 3 4 Supplementary Figure 1: Micromechanical cleavage of graphene on oxygen plasma treated Si/SiO 2. Optical microscopy images of three examples of large single layer graphene flakes cleaved on a single
1 2 3 4 Supplementary Figure 1: Micromechanical cleavage of graphene on oxygen plasma treated Si/SiO 2. Optical microscopy images of three examples of large single layer graphene flakes cleaved on a single
On the Landau-Levich Transition
 10116 Langmuir 2007, 23, 10116-10122 On the Landau-Levich Transition Maniya Maleki Institute for AdVanced Studies in Basic Sciences (IASBS), Zanjan 45195, P.O. Box 45195-1159, Iran Etienne Reyssat and
10116 Langmuir 2007, 23, 10116-10122 On the Landau-Levich Transition Maniya Maleki Institute for AdVanced Studies in Basic Sciences (IASBS), Zanjan 45195, P.O. Box 45195-1159, Iran Etienne Reyssat and
Supplementary Figure 1 Detailed illustration on the fabrication process of templatestripped
 Supplementary Figure 1 Detailed illustration on the fabrication process of templatestripped gold substrate. (a) Spin coating of hydrogen silsesquioxane (HSQ) resist onto the silicon substrate with a thickness
Supplementary Figure 1 Detailed illustration on the fabrication process of templatestripped gold substrate. (a) Spin coating of hydrogen silsesquioxane (HSQ) resist onto the silicon substrate with a thickness
Cohesion, Surface Tension, and Adhesion
 Cohesion, Surface Tension, and Adhesion Content Objectives SWBAT describe how hydrogen bonding allows water molecules to maintain strong cohesion, adhesion, and surface tension. Van der Waals Forces The
Cohesion, Surface Tension, and Adhesion Content Objectives SWBAT describe how hydrogen bonding allows water molecules to maintain strong cohesion, adhesion, and surface tension. Van der Waals Forces The
SUPPLEMENTARY INFORMATION
 DOI: 1.138/NMAT415 Giant Switchable Photovoltaic Effect in Organometal Trihalide Perovskite Devices Zhengguo Xiao 1,2, Yongbo Yuan 1,2, Yuchuan Shao 1,2, Qi Wang, 1,2 Qingfeng Dong, 1,2 Cheng Bi 1,2, Pankaj
DOI: 1.138/NMAT415 Giant Switchable Photovoltaic Effect in Organometal Trihalide Perovskite Devices Zhengguo Xiao 1,2, Yongbo Yuan 1,2, Yuchuan Shao 1,2, Qi Wang, 1,2 Qingfeng Dong, 1,2 Cheng Bi 1,2, Pankaj
