Državni izpitni center. Izpitna pola 1. Sobota, 27. avgust 2016 / 90 minut
|
|
|
- Brittany Carmel Barker
- 6 years ago
- Views:
Transcription
1 Š i f r a k a n d i d a t a : Državni izpitni center *M * JESENSKI IZPITNI OK Izpitna pola 1 Sobota, 7. avgust 016 / 90 minut Dovoljeno gradivo in pripomočki: Kandidat prinese nalivno pero ali kemični svinčnik, svinčnik, radirko, šestilo, trikotnika in računalo. Kandidat dobi dva konceptna lista in ocenjevalni obrazec. Priloga s konstantami in enačbami ter magnetilnimi krivuljami je na perforiranem listu, ki ga kandidat pazljivo iztrga. SPLOŠNA MATUA NAVODILA KANDIDATU Pazljivo preberite ta navodila. Ne odpirajte izpitne pole in ne začenjajte reševati nalog, dokler vam nadzorni učitelj tega ne dovoli. Prilepite kodo oziroma vpišite svojo šifro (v okvirček desno zgoraj na tej strani in na ocenjevalni obrazec). Svojo šifro vpišite tudi na konceptna lista. Izpitna pola vsebuje 8 nalog s kratkimi odgovori in 3 strukturirane naloge. Število točk, ki jih lahko dosežete, je 40. Za posamezno nalogo je število točk navedeno v izpitni poli. Pri reševanju si lahko pomagate z zbirko konstant in enačb v prilogi. ešitve, ki jih pišite z nalivnim peresom ali s kemičnim svinčnikom, vpisujte v izpitno polo v za to predvideni prostor, slike in diagrame pa rišite prostoročno s svinčnikom. Pišite čitljivo. Če se zmotite, napisano prečrtajte in rešitev zapišite na novo. Nečitljivi zapisi in nejasni popravki bodo ocenjeni z 0 točkami. Osnutki rešitev, ki jih lahko naredite na konceptna lista, se pri ocenjevanju ne upoštevajo. Zaupajte vase in v svoje zmožnosti. Želimo vam veliko uspeha. Ta pola ima 16 strani, od tega prazni. IC 016
2 /16 *M *
3 *M * 3/16 Konstante in enačbe Elektrina in električni tok 19 e 0 1, C Q ne0 DQ i = D t I JA m cit Električno polje 1 As 0 8, Vm QQ 1 F 4 d F QE Q E 4 r q E r E D E E U Ed 0 r UAB VA VB F e Q DA Q C U W CU Enosmerna vezja I 0 k m k U 0 m U 1 I G P UI W Pt l l A A C A d w ED Magnetno polje 7 Vs Am miil 1 F = p d F BIl F B A o I B r Ir B r 0 NI B l F=BA M IABsin Hl B H H m l A 0 r Inducirano električno polje Y = N F u i Y t vbl ui U m N F m L Y N A L i l W Li F B A 0 w BH Trifazni sistemi Y1U Y U Y3U V 0 Y Y Y Izmenična električna vezja f Tf 1 u U sin t i I sin t u j = a - a j e cos jsin U Z 1 I Y Z jx Y G jb Z Z L Z C jl 1 jc S P jq U I Q tan 1 0 LC 1 Q L 0 i 1 C Prehodni pojavi u i u L di d t i C du d t 0 u U 1 e t / / u Ue t C i I 1 e t / / i Ie t L i u 1 0 C 0 P P izh vh P perforiran list
4 4/16 *M *
5 *M * 5/16 1. Fizikalna veličina ima enoto Vs/Am. Poimenujte fizikalno veličino in zapišite njeno oznako. ( točki). Dovoljena gostota toka v vodniku je J = A/mm. Izračunajte dopustni tok I, če je premer vodnika d = mm. ( točki)
6 6/16 *M * 3. Postopek elektrolize bakra poteka pri gostoti toka A = 5 m J = 500 A/m, površina elektrod je, elektrokemijski ekvivalent bakra je c = 0,39 10 kg/c. Izračunajte, koliko bakra se izloči na elektrodah v času -6 t = 10 h. ( točki) 4. Z ampermetrom, ki ima merilno območje I A = 50 ma in notranjo upornost A = 3,6 W, želimo meriti toke do I = 500 ma. Izračunajte potrebno upornost S soupora. ( točki)
7 *M * 7/16 5. Dano je enosmerno vezje V V U U - Določite označeni napetosti U 1 in U. ( točki) 6. Na uporu sta dana harmonična napetost ut ( ) = 0sin(400t + 30 ) V in tok it ( ) = 40sin(400t + 30 ) ma. Izračunajte moč P na uporu. ( točki)
8 8/16 *M * 7. Zaporedno L-vezje smo priključili na vir harmonične napetosti efektivne vrednosti U = 50 V. Na uporu smo izmerili efektivno vrednost napetosti U = 30 V. Izračunajte efektivno vrednost napetosti U L, ki bi jo izmerili na tuljavi. ( točki) 8. Za narisano vezje S i L U = 0 V L = 4 H izračunajte upornost, da se bo prehodni pojav praktično končal v času t = s. ( točki)
9 *M * 9/16 Prazna stran OBNITE LIST.
10 10/16 *M * 9. Bakrena žica ima prerez A = 0, mm in dolžino l = 50 m -6-1 ( r = 0, W m, a = 0,0039 K ) Izračunajte upornost žice 0 pri sobni temperaturi T = 0 C. ( točki) 9.. Izračunajte upornost žice 60 pri delovni temperaturi T = 60 C. ( točki)
11 *M * 11/ Izračunajte toploto Q, ki se sprosti v žici v času t = 5 min, če jo priključimo na napetost U = 0 V. ( točki) 9.4. Izračunajte dolžino l 1, ki bi jo morala imeti žica, da bi imela pri delovni temperaturi T = 60 C enako upornost kakor 50-metrska žica pri sobni temperaturi. ( točki)
12 1/16 *M * 10. Na sliki je vezje s podatki: = 0 W, C = 0 m F, L = 40 mh, U = j0 V in w = 1000 rad/s. I I 1 I L C U Izračunajte impedanco Z 1 zgornje veje vezja. ( točki) 10.. Izračunajte kazalca I 1 in I. ( točki)
13 *M * 13/ Izračunajte kompleksno moč S. ( točki) Izračunajte elementa nadomestnega zaporednega vezja. ( točki)
14 14/16 *M * 11. V narisanem vezju (z U = 1 V, 1 = = 1 MW in C = 1 m F ) ob času t = 0 izklopimo stikalo S. U = 1 V 1 = 1 MW t = 0 S C = 1 mf u C = 1 MW Določite napetost U C0 na kondenzatorju C v trenutku pred razklenitvijo stikala. ( točki) 11.. Izračunajte časovno konstanto t prehodnega pojava. ( točki)
15 *M * 15/ Izračunajte energijo v kondenzatorju C po končanem prehodnem pojavu. ( točki) Skicirajte časovni diagram napetosti na kondenzatorju C med prehodnim pojavom. ( točki)
16 16/16 *M * Prazna stran
Državni izpitni center. Izpitna pola 1. Četrtek, 4. junij 2015 / 90 minut
 Š i f r a k a n d i d a t a : Državni izpitni center *M15177111* SPOMLADANSKI IZPITNI ROK Izpitna pola 1 Četrtek, 4. junij 015 / 90 minut Dovoljeno gradivo in pripomočki: Kandidat prinese nalivno pero
Š i f r a k a n d i d a t a : Državni izpitni center *M15177111* SPOMLADANSKI IZPITNI ROK Izpitna pola 1 Četrtek, 4. junij 015 / 90 minut Dovoljeno gradivo in pripomočki: Kandidat prinese nalivno pero
Državni izpitni center. Izpitna pola 1. Četrtek, 27. avgust 2015 / 90 minut
 Š i f r a k a n d i d a t a : Državni izpitni center *M1577111* JESENSKI IZPITNI OK Izpitna pola 1 Četrtek, 7. avgust 015 / 90 minut Dovoljeno gradivo in pripomočki: Kandidat prinese nalivno pero ali kemični
Š i f r a k a n d i d a t a : Državni izpitni center *M1577111* JESENSKI IZPITNI OK Izpitna pola 1 Četrtek, 7. avgust 015 / 90 minut Dovoljeno gradivo in pripomočki: Kandidat prinese nalivno pero ali kemični
Državni izpitni center ELEKTROTEHNIKA. Izpitna pola 1. Četrtek, 1. junij 2017 / 90 minut
 Š i f r a k a n d i d a t a : Državni izpitni center *M17177111* SPOMLADANSKI IZPITNI ROK ELEKTROTEHNIKA Izpitna pola 1 Četrtek, 1. junij 017 / 90 minut Dovoljeno gradivo in pripomočki: Kandidat prinese
Š i f r a k a n d i d a t a : Državni izpitni center *M17177111* SPOMLADANSKI IZPITNI ROK ELEKTROTEHNIKA Izpitna pola 1 Četrtek, 1. junij 017 / 90 minut Dovoljeno gradivo in pripomočki: Kandidat prinese
Državni izpitni center. Izpitna pola 2. Ponedeljek, 28. avgust 2017 / 90 minut
 Š i f r a k a n d i d a t a : Državni izpitni center *M177711* JESENSKI IZPITNI ROK Izpitna pola Ponedeljek, 8. avgust 017 / 90 minut Dovoljeno gradivo in pripomočki: Kandidat prinese nalivno pero ali
Š i f r a k a n d i d a t a : Državni izpitni center *M177711* JESENSKI IZPITNI ROK Izpitna pola Ponedeljek, 8. avgust 017 / 90 minut Dovoljeno gradivo in pripomočki: Kandidat prinese nalivno pero ali
Državni izpitni center MATEMATIKA. Izpitna pola. Sobota, 3. junij 2017 / 120 minut
 Š i f r a k a n d i d a t a : Državni izpitni center *P171C10111* SPOMLADANSKI IZPITNI ROK MATEMATIKA Izpitna pola Sobota, 3. junij 017 / 10 minut Dovoljeno gradivo in pripomočki: Kandidat prinese nalivno
Š i f r a k a n d i d a t a : Državni izpitni center *P171C10111* SPOMLADANSKI IZPITNI ROK MATEMATIKA Izpitna pola Sobota, 3. junij 017 / 10 minut Dovoljeno gradivo in pripomočki: Kandidat prinese nalivno
Državni izpitni center. Osnovna raven ANGLEŠČINA. Izpitna pola 1. A) Bralno razumevanje B) Poznavanje in raba jezika
 Š i f r a k a n d i d a t a : Državni izpitni center *M17224111* Osnovna raven ANGLEŠČINA Izpitna pola 1 A) Bralno razumevanje B) Poznavanje in raba jezika JESENSKI IZPITNI ROK Sobota, 26. avgust 2017
Š i f r a k a n d i d a t a : Državni izpitni center *M17224111* Osnovna raven ANGLEŠČINA Izpitna pola 1 A) Bralno razumevanje B) Poznavanje in raba jezika JESENSKI IZPITNI ROK Sobota, 26. avgust 2017
Državni izpitni center. Izpitna pola 2. Četrtek, 28. avgust 2014 / 90 minut
 Š i f r a k a n d i d a t a : Državni izpitni center *M14244112* JESENSKI IZPITNI ROK Izpitna pola 2 Četrtek, 28. avgust 2014 / 90 minut Dovoljeno gradivo in pripomočki: Kandidat prinese nalivno pero ali
Š i f r a k a n d i d a t a : Državni izpitni center *M14244112* JESENSKI IZPITNI ROK Izpitna pola 2 Četrtek, 28. avgust 2014 / 90 minut Dovoljeno gradivo in pripomočki: Kandidat prinese nalivno pero ali
*M * ANGLE[^INA. Vi{ja raven. A: Bralno razumevanje B: Poznavanje in raba jezika. Sobota, 29. maj 2004 / 80 minut ( ) [ifra kandidata:
 [ifra kandidata: *M04124211* SPOMLADANSKI ROK Vi{ja raven ANGLE[^INA A: Bralno razumevanje B: Poznavanje in raba jezika Sobota, 29. maj 2004 / 80 minut (40 + 40) Dovoljeno dodatno gradivo in pripomočki:
[ifra kandidata: *M04124211* SPOMLADANSKI ROK Vi{ja raven ANGLE[^INA A: Bralno razumevanje B: Poznavanje in raba jezika Sobota, 29. maj 2004 / 80 minut (40 + 40) Dovoljeno dodatno gradivo in pripomočki:
Državni izpitni center ZGODOVINA. Izpitna pola 1. Obča zgodovina. Četrtek, 4. junij 2009 / 90 minut
 Š i f r a k a n d i d a t a : Državni izpitni center *M09151111* SPOMLADANSKI IZPITNI ROK ZGODOVINA Izpitna pola 1 Obča zgodovina Četrtek, 4. junij 2009 / 90 minut Dovoljeno gradivo in pripomočki: Kandidat
Š i f r a k a n d i d a t a : Državni izpitni center *M09151111* SPOMLADANSKI IZPITNI ROK ZGODOVINA Izpitna pola 1 Obča zgodovina Četrtek, 4. junij 2009 / 90 minut Dovoljeno gradivo in pripomočki: Kandidat
Izmenični signali moč (17)
 Izenicni_signali_MOC(17c).doc 1/7 8.5.007 Izenični signali oč (17) Zania nas potek trenutne oči v linearne dvopolne (dve zunanji sponki) vezju, kjer je napetost na zunanjih sponkah enaka u = U sin( ωt),
Izenicni_signali_MOC(17c).doc 1/7 8.5.007 Izenični signali oč (17) Zania nas potek trenutne oči v linearne dvopolne (dve zunanji sponki) vezju, kjer je napetost na zunanjih sponkah enaka u = U sin( ωt),
Državni izpitni center MATEMATIKA PREIZKUS ZNANJA. Sreda, 4. maj 2011 / 60 minut. NACIONALNO PREVERJANJE ZNANJA ob koncu 2. obdobja NAVODILA UČENCU
 Š i f r a u č e n c a: Državni izpitni center *N11140121* REDNI ROK 2. obdobje MATEMATIKA PREIZKUS ZNANJA Sreda, 4. maj 2011 / 60 minut Dovoljeno gradivo in pripomočki: Učenec prinese modro/črno nalivno
Š i f r a u č e n c a: Državni izpitni center *N11140121* REDNI ROK 2. obdobje MATEMATIKA PREIZKUS ZNANJA Sreda, 4. maj 2011 / 60 minut Dovoljeno gradivo in pripomočki: Učenec prinese modro/črno nalivno
Električne lastnosti vodov. Ohmske upornosti. Induktivnost vodov. Kapacitivnost vodov. Odvodnost vodov. Vod v svetlobi telegrafske enačbe.
 Električne lastnosti vodov Ohmske upornosti. Induktivnost vodov. Kapacitivnost vodov. Odvodnost vodov. Vod v svetlobi telegrafske enačbe. Primarne konstante vodov Če opazujemo električni vod iz istega
Električne lastnosti vodov Ohmske upornosti. Induktivnost vodov. Kapacitivnost vodov. Odvodnost vodov. Vod v svetlobi telegrafske enačbe. Primarne konstante vodov Če opazujemo električni vod iz istega
P a g e 5 1 of R e p o r t P B 4 / 0 9
 P a g e 5 1 of R e p o r t P B 4 / 0 9 J A R T a l s o c o n c l u d e d t h a t a l t h o u g h t h e i n t e n t o f N e l s o n s r e h a b i l i t a t i o n p l a n i s t o e n h a n c e c o n n e
P a g e 5 1 of R e p o r t P B 4 / 0 9 J A R T a l s o c o n c l u d e d t h a t a l t h o u g h t h e i n t e n t o f N e l s o n s r e h a b i l i t a t i o n p l a n i s t o e n h a n c e c o n n e
Baroklina nestabilnost
 Baroklina nestabilnost Navodila za projektno nalogo iz dinamične meteorologije 2012/2013 Januar 2013 Nedjeljka Zagar in Rahela Zabkar Naloga je zasnovana na dvoslojnem modelu baroklinega razvoja, napisana
Baroklina nestabilnost Navodila za projektno nalogo iz dinamične meteorologije 2012/2013 Januar 2013 Nedjeljka Zagar in Rahela Zabkar Naloga je zasnovana na dvoslojnem modelu baroklinega razvoja, napisana
Executive Committee and Officers ( )
 Gifted and Talented International V o l u m e 2 4, N u m b e r 2, D e c e m b e r, 2 0 0 9. G i f t e d a n d T a l e n t e d I n t e r n a t i o n a2 l 4 ( 2), D e c e m b e r, 2 0 0 9. 1 T h e W o r
Gifted and Talented International V o l u m e 2 4, N u m b e r 2, D e c e m b e r, 2 0 0 9. G i f t e d a n d T a l e n t e d I n t e r n a t i o n a2 l 4 ( 2), D e c e m b e r, 2 0 0 9. 1 T h e W o r
Reševanje problemov in algoritmi
 Reševanje problemov in algoritmi Vhod Algoritem Izhod Kaj bomo spoznali Zgodovina algoritmov. Primeri algoritmov. Algoritmi in programi. Kaj je algoritem? Algoritem je postopek, kako korak za korakom rešimo
Reševanje problemov in algoritmi Vhod Algoritem Izhod Kaj bomo spoznali Zgodovina algoritmov. Primeri algoritmov. Algoritmi in programi. Kaj je algoritem? Algoritem je postopek, kako korak za korakom rešimo
CATAVASII LA NAȘTEREA DOMNULUI DUMNEZEU ȘI MÂNTUITORULUI NOSTRU, IISUS HRISTOS. CÂNTAREA I-A. Ήχος Πα. to os se e e na aș te e e slă ă ă vi i i i i
 CATAVASII LA NAȘTEREA DOMNULUI DUMNEZEU ȘI MÂNTUITORULUI NOSTRU, IISUS HRISTOS. CÂNTAREA I-A Ήχος α H ris to os s n ș t slă ă ă vi i i i i ți'l Hris to o os di in c ru u uri, în tâm pi i n ți i'l Hris
CATAVASII LA NAȘTEREA DOMNULUI DUMNEZEU ȘI MÂNTUITORULUI NOSTRU, IISUS HRISTOS. CÂNTAREA I-A Ήχος α H ris to os s n ș t slă ă ă vi i i i i ți'l Hris to o os di in c ru u uri, în tâm pi i n ți i'l Hris
Attempt to prepare seasonal weather outlook for Slovenia
 Attempt to prepare seasonal weather outlook for Slovenia Main available sources (ECMWF, EUROSIP, IRI, CPC.NCEP.NOAA,..) Two parameters (T and RR anomally) Textual information ( Met Office like ) Issued
Attempt to prepare seasonal weather outlook for Slovenia Main available sources (ECMWF, EUROSIP, IRI, CPC.NCEP.NOAA,..) Two parameters (T and RR anomally) Textual information ( Met Office like ) Issued
T h e C S E T I P r o j e c t
 T h e P r o j e c t T H E P R O J E C T T A B L E O F C O N T E N T S A r t i c l e P a g e C o m p r e h e n s i v e A s s es s m e n t o f t h e U F O / E T I P h e n o m e n o n M a y 1 9 9 1 1 E T
T h e P r o j e c t T H E P R O J E C T T A B L E O F C O N T E N T S A r t i c l e P a g e C o m p r e h e n s i v e A s s es s m e n t o f t h e U F O / E T I P h e n o m e n o n M a y 1 9 9 1 1 E T
OA07 ANNEX 4: SCOPE OF ACCREDITATION IN CALIBRATION
 OA07 ANNEX 4: SCOPE OF ACCREDITATION IN CALIBRATION Table of contents 1 TECHNICAL FIELDS... 2 2 PRESENTING THE SCOPE OF A CALIBRATION LABOORATORY... 2 3 CONSIDERING CHANGES TO SCOPES... 6 4 CHANGES WITH
OA07 ANNEX 4: SCOPE OF ACCREDITATION IN CALIBRATION Table of contents 1 TECHNICAL FIELDS... 2 2 PRESENTING THE SCOPE OF A CALIBRATION LABOORATORY... 2 3 CONSIDERING CHANGES TO SCOPES... 6 4 CHANGES WITH
Note 11: Alternating Current (AC) Circuits
 Note 11: Alternating Current (AC) Circuits V R No phase difference between the voltage difference and the current and max For alternating voltage Vmax sin t, the resistor current is ir sin t. the instantaneous
Note 11: Alternating Current (AC) Circuits V R No phase difference between the voltage difference and the current and max For alternating voltage Vmax sin t, the resistor current is ir sin t. the instantaneous
Multipla korelacija in regresija. Multipla regresija, multipla korelacija, statistično zaključevanje o multiplem R
 Multipla koelacia in egesia Multipla egesia, multipla koelacia, statistično zaklučevane o multiplem Multipla egesia osnovni model in ačunane paametov Z multiplo egesio napoveduemo vednost kiteia (odvisne
Multipla koelacia in egesia Multipla egesia, multipla koelacia, statistično zaklučevane o multiplem Multipla egesia osnovni model in ačunane paametov Z multiplo egesio napoveduemo vednost kiteia (odvisne
HANDOUT SET GENERAL CHEMISTRY II
 HANDOUT SET GENERAL CHEMISTRY II Periodic Table of the Elements 1 2 3 4 5 6 7 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 IA VIIIA 1 2 H He 1.00794 IIA IIIA IVA VA VIA VIIA 4.00262 3 Li 6.941 11 Na 22.9898
HANDOUT SET GENERAL CHEMISTRY II Periodic Table of the Elements 1 2 3 4 5 6 7 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 IA VIIIA 1 2 H He 1.00794 IIA IIIA IVA VA VIA VIIA 4.00262 3 Li 6.941 11 Na 22.9898
A L A BA M A L A W R E V IE W
 A L A BA M A L A W R E V IE W Volume 52 Fall 2000 Number 1 B E F O R E D I S A B I L I T Y C I V I L R I G HT S : C I V I L W A R P E N S I O N S A N D TH E P O L I T I C S O F D I S A B I L I T Y I N
A L A BA M A L A W R E V IE W Volume 52 Fall 2000 Number 1 B E F O R E D I S A B I L I T Y C I V I L R I G HT S : C I V I L W A R P E N S I O N S A N D TH E P O L I T I C S O F D I S A B I L I T Y I N
Software Process Models there are many process model s in th e li t e ra t u re, s om e a r e prescriptions and some are descriptions you need to mode
 Unit 2 : Software Process O b j ec t i ve This unit introduces software systems engineering through a discussion of software processes and their principal characteristics. In order to achieve the desireable
Unit 2 : Software Process O b j ec t i ve This unit introduces software systems engineering through a discussion of software processes and their principal characteristics. In order to achieve the desireable
ORBITAL DIAGRAM - A graphical representation of the quantum number "map" of electrons around an atom.
 178 (MAGNETIC) SPIN QUANTUM NUMBER: "spin down" or "spin up" - An ORBITAL (region with fixed "n", "l" and "ml" values) can hold TWO electrons. ORBITAL DIAGRAM - A graphical representation of the quantum
178 (MAGNETIC) SPIN QUANTUM NUMBER: "spin down" or "spin up" - An ORBITAL (region with fixed "n", "l" and "ml" values) can hold TWO electrons. ORBITAL DIAGRAM - A graphical representation of the quantum
Last 4 Digits of USC ID:
 Chemistry 05 B Practice Exam Dr. Jessica Parr First Letter of last Name PLEASE PRINT YOUR NAME IN BLOCK LETTERS Name: Last 4 Digits of USC ID: Lab TA s Name: Question Points Score Grader 8 2 4 3 9 4 0
Chemistry 05 B Practice Exam Dr. Jessica Parr First Letter of last Name PLEASE PRINT YOUR NAME IN BLOCK LETTERS Name: Last 4 Digits of USC ID: Lab TA s Name: Question Points Score Grader 8 2 4 3 9 4 0
Solutions and Ions. Pure Substances
 Class #4 Solutions and Ions CHEM 107 L.S. Brown Texas A&M University Pure Substances Pure substance: described completely by a single chemical formula Fixed composition 1 Mixtures Combination of 2 or more
Class #4 Solutions and Ions CHEM 107 L.S. Brown Texas A&M University Pure Substances Pure substance: described completely by a single chemical formula Fixed composition 1 Mixtures Combination of 2 or more
CHEM 10113, Quiz 5 October 26, 2011
 CHEM 10113, Quiz 5 October 26, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
CHEM 10113, Quiz 5 October 26, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
Mathematics 22: Lecture 7
 Mathematics 22: Lecture 7 Separation of Variables Dan Sloughter Furman University January 15, 2008 Dan Sloughter (Furman University) Mathematics 22: Lecture 7 January 15, 2008 1 / 8 Separable equations
Mathematics 22: Lecture 7 Separation of Variables Dan Sloughter Furman University January 15, 2008 Dan Sloughter (Furman University) Mathematics 22: Lecture 7 January 15, 2008 1 / 8 Separable equations
(please print) (1) (18) H IIA IIIA IVA VA VIA VIIA He (2) (13) (14) (15) (16) (17)
 CHEM 10113, Quiz 3 September 28, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
CHEM 10113, Quiz 3 September 28, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
Lewis dot structures for molecules
 1 Lewis dot structures for molecules In the dot structure of a molecule, - SHARED valence electrons are shown with dashes - one per pair. - UNSHARED valence electrons ("lone pairs") are represented by
1 Lewis dot structures for molecules In the dot structure of a molecule, - SHARED valence electrons are shown with dashes - one per pair. - UNSHARED valence electrons ("lone pairs") are represented by
CHEMICAL COMPOUNDS MOLECULAR COMPOUNDS
 48 CHEMICAL COMPOUNDS - Dalton's theory does not mention this, but there is more than one way for atoms to come together to make chemical compounds! - There are TWO common kinds of chemical compound, classified
48 CHEMICAL COMPOUNDS - Dalton's theory does not mention this, but there is more than one way for atoms to come together to make chemical compounds! - There are TWO common kinds of chemical compound, classified
176 5 t h Fl oo r. 337 P o ly me r Ma te ri al s
 A g la di ou s F. L. 462 E l ec tr on ic D ev el op me nt A i ng er A.W.S. 371 C. A. M. A l ex an de r 236 A d mi ni st ra ti on R. H. (M rs ) A n dr ew s P. V. 326 O p ti ca l Tr an sm is si on A p ps
A g la di ou s F. L. 462 E l ec tr on ic D ev el op me nt A i ng er A.W.S. 371 C. A. M. A l ex an de r 236 A d mi ni st ra ti on R. H. (M rs ) A n dr ew s P. V. 326 O p ti ca l Tr an sm is si on A p ps
ORBITAL DIAGRAM - A graphical representation of the quantum number "map" of electrons around an atom.
 160 ORBITAL DIAGRAM - A graphical representation of the quantum number "map" of electrons around an atom. 4p 3d 4s 3p 3s 2p 2s 1s Each blank represents an ORBITAL, and can hold two electrons. The 4s subshell
160 ORBITAL DIAGRAM - A graphical representation of the quantum number "map" of electrons around an atom. 4p 3d 4s 3p 3s 2p 2s 1s Each blank represents an ORBITAL, and can hold two electrons. The 4s subshell
CHEMICAL COMPOUNDS MOLECULAR COMPOUNDS
 48 CHEMICAL COMPOUNDS - Dalton's theory does not mention this, but there is more than one way for atoms to come together to make chemical compounds! - There are TWO common kinds of chemical compound, classified
48 CHEMICAL COMPOUNDS - Dalton's theory does not mention this, but there is more than one way for atoms to come together to make chemical compounds! - There are TWO common kinds of chemical compound, classified
-"l" also contributes ENERGY. Higher values for "l" mean the electron has higher energy.
 175 - Giving the four parameters will uniquely identify an electron around an atom. No two electrons in the same atom can share all four. These parameters are called QUANTUM NUMBERS. PRINCIPAL QUANTUM
175 - Giving the four parameters will uniquely identify an electron around an atom. No two electrons in the same atom can share all four. These parameters are called QUANTUM NUMBERS. PRINCIPAL QUANTUM
8. Relax and do well.
 CHEM 1515 Exam II John II. Gelder October 14, 1993 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 8 different pages. The last two pages include a periodic table, a
CHEM 1515 Exam II John II. Gelder October 14, 1993 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 8 different pages. The last two pages include a periodic table, a
CHEM 10123/10125, Exam 2
 CHEM 10123/10125, Exam 2 March 7, 2012 (50 minutes) Name (please print) Please box your answers, and remember that significant figures, phases (for chemical equations), and units do count! 1. (13 points)
CHEM 10123/10125, Exam 2 March 7, 2012 (50 minutes) Name (please print) Please box your answers, and remember that significant figures, phases (for chemical equations), and units do count! 1. (13 points)
REACTANCE. By: Enzo Paterno Date: 03/2013
 REACTANCE REACTANCE By: Enzo Paterno Date: 03/2013 5/2007 Enzo Paterno 1 RESISTANCE - R i R (t R A resistor for all practical purposes is unaffected by the frequency of the applied sinusoidal voltage or
REACTANCE REACTANCE By: Enzo Paterno Date: 03/2013 5/2007 Enzo Paterno 1 RESISTANCE - R i R (t R A resistor for all practical purposes is unaffected by the frequency of the applied sinusoidal voltage or
VIIIA H PREDICTING CHARGE
 58 IA PREDICTING CHARGE VIIIA H IIA IIIA IVA VA VIA VIIA You can reliably determine the charge using our method for Groups IA, IIA, IIIB, Aluminum, and the Group VA, VIA, and VIIA NONMETALS Li Be B C N
58 IA PREDICTING CHARGE VIIIA H IIA IIIA IVA VA VIA VIIA You can reliably determine the charge using our method for Groups IA, IIA, IIIB, Aluminum, and the Group VA, VIA, and VIIA NONMETALS Li Be B C N
Scripture quotations marked cev are from the Contemporary English Version, Copyright 1991, 1992, 1995 by American Bible Society. Used by permission.
 N Ra: E K B Da a a B a a, a-a- a aa, a a. T, a a. 2009 Ba P, I. ISBN 978-1-60260-296-0. N a a a a a, a,. C a a a Ba P, a 500 a a aa a. W, : F K B Da, Ba P, I. U. S a a a a K Ja V B. S a a a a N K Ja V.
N Ra: E K B Da a a B a a, a-a- a aa, a a. T, a a. 2009 Ba P, I. ISBN 978-1-60260-296-0. N a a a a a, a,. C a a a Ba P, a 500 a a aa a. W, : F K B Da, Ba P, I. U. S a a a a K Ja V B. S a a a a N K Ja V.
8. Relax and do well.
 CHEM 1225 Exam III John III. Gelder April 8, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last two pages includes a periodic table and
CHEM 1225 Exam III John III. Gelder April 8, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last two pages includes a periodic table and
7. Relax and do well.
 CHEM 1215 Exam II John II. Gelder October 7, 1998 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 5 different pages. The last page includes a periodic table and a solubility
CHEM 1215 Exam II John II. Gelder October 7, 1998 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 5 different pages. The last page includes a periodic table and a solubility
The exam must be written in ink. No calculators of any sort allowed. You have 2 hours to complete the exam. Periodic table 7 0
 Email: The exam must be written in ink. No calculators of any sort allowed. You have 2 hours to complete the exam. CEM 610B Exam 3 Spring 2002 Instructor: Dr. Brian Pagenkopf Page Points 2 6 3 7 4 9 5
Email: The exam must be written in ink. No calculators of any sort allowed. You have 2 hours to complete the exam. CEM 610B Exam 3 Spring 2002 Instructor: Dr. Brian Pagenkopf Page Points 2 6 3 7 4 9 5
K. 27 Co. 28 Ni. 29 Cu Rb. 46 Pd. 45 Rh. 47 Ag Cs Ir. 78 Pt.
 1 IA 1 ydrogen 1.01 Atomic number Element symbol Element name Atomic mass VIIIA 1 1.01 IIA IIIA IVA VA VIA VIIA 2 e 4.00 Metalloids 3 Li 6.94 4 Be 9.01 5 B 10.81 6 C 12.01 7 N 14.01 8 O 16.00 9 F 19.00
1 IA 1 ydrogen 1.01 Atomic number Element symbol Element name Atomic mass VIIIA 1 1.01 IIA IIIA IVA VA VIA VIIA 2 e 4.00 Metalloids 3 Li 6.94 4 Be 9.01 5 B 10.81 6 C 12.01 7 N 14.01 8 O 16.00 9 F 19.00
S U E K E AY S S H A R O N T IM B E R W IN D M A R T Z -PA U L L IN. Carlisle Franklin Springboro. Clearcreek TWP. Middletown. Turtlecreek TWP.
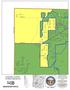 F R A N K L IN M A D IS O N S U E R O B E R T LE IC H T Y A LY C E C H A M B E R L A IN T W IN C R E E K M A R T Z -PA U L L IN C O R A O W E N M E A D O W L A R K W R E N N LA N T IS R E D R O B IN F
F R A N K L IN M A D IS O N S U E R O B E R T LE IC H T Y A LY C E C H A M B E R L A IN T W IN C R E E K M A R T Z -PA U L L IN C O R A O W E N M E A D O W L A R K W R E N N LA N T IS R E D R O B IN F
INSTRUCTIONS: CHEM Exam I. September 13, 1994 Lab Section
 CHEM 1314.05 Exam I John I. Gelder September 13, 1994 Name TA's Name Lab Section Please sign your name below to give permission to post, by the last 4 digits of your student I.D. number, your course scores
CHEM 1314.05 Exam I John I. Gelder September 13, 1994 Name TA's Name Lab Section Please sign your name below to give permission to post, by the last 4 digits of your student I.D. number, your course scores
WRITING AN IONIC FORMULA
 WRITING AN IONIC FORMULA - if you know the ions that make up a compound, all you need to do is find the smallest ratio of cation to anion the compound needs to have an overall charge of zero Example: If
WRITING AN IONIC FORMULA - if you know the ions that make up a compound, all you need to do is find the smallest ratio of cation to anion the compound needs to have an overall charge of zero Example: If
Example: If a simple ionic compound is made of these two ions, what is its formula? In the final formula, don't write the charges on the ions!
 88 WRITING AN IONIC FORMULA - if you know the ions that make up a compound, all you need to do is find the smallest ratio of cation to anion the compound needs to have an overall charge of zero Example:
88 WRITING AN IONIC FORMULA - if you know the ions that make up a compound, all you need to do is find the smallest ratio of cation to anion the compound needs to have an overall charge of zero Example:
8. Relax and do well.
 CHEM 1215 Exam III John III. Gelder November 11, 1998 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
CHEM 1215 Exam III John III. Gelder November 11, 1998 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
The Periodic Table of the Elements
 The Periodic Table of the Elements All matter is composed of elements. All of the elements are composed of atoms. An atom is the smallest part of an element which still retains the properties of that element.
The Periodic Table of the Elements All matter is composed of elements. All of the elements are composed of atoms. An atom is the smallest part of an element which still retains the properties of that element.
E4 Acids, Bases, and Salts
 E4 Acids, Bases, and Salts Session One of two session lab Complete Parts 1 and 2 in lab. If time allows, start or complete Part 3. Acids and Bases Q. Are acid-base properties of substances predictable
E4 Acids, Bases, and Salts Session One of two session lab Complete Parts 1 and 2 in lab. If time allows, start or complete Part 3. Acids and Bases Q. Are acid-base properties of substances predictable
CHEMICAL COMPOUNDS MOLECULAR COMPOUNDS
 48 CHEMICAL COMPOUNDS - Dalton's theory does not mention this, but there is more than one way for atoms to come together to make chemical compounds! - There are TWO common kinds of chemical compound, classified
48 CHEMICAL COMPOUNDS - Dalton's theory does not mention this, but there is more than one way for atoms to come together to make chemical compounds! - There are TWO common kinds of chemical compound, classified
CHEM Come to the PASS workshop with your mock exam complete. During the workshop you can work with other students to review your work.
 It is most beneficial to you to write this mock midterm UNDER EXAM CONDITIONS. This means: Complete the midterm in 1.5 hours. Work on your own. Keep your notes and textbook closed. Attempt every question.
It is most beneficial to you to write this mock midterm UNDER EXAM CONDITIONS. This means: Complete the midterm in 1.5 hours. Work on your own. Keep your notes and textbook closed. Attempt every question.
INSTRUCTIONS: Exam III. November 10, 1999 Lab Section
 CHEM 1215 Exam III John III. Gelder November 10, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
CHEM 1215 Exam III John III. Gelder November 10, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
VIIIA H PREDICTING CHARGE
 58 IA PREDICTING CHARGE VIIIA H IIA IIIA IVA VA VIA VIIA You can reliably determine the charge using our method for Groups IA, IIA, IIIB, Aluminum, and the Group VA, VIA, and VIIA NONMETALS Li Be B C N
58 IA PREDICTING CHARGE VIIIA H IIA IIIA IVA VA VIA VIIA You can reliably determine the charge using our method for Groups IA, IIA, IIIB, Aluminum, and the Group VA, VIA, and VIIA NONMETALS Li Be B C N
CHEM 251 (Fall-2003) Final Exam (100 pts)
 CEM 251 (Fall-2003) Final Exam (100 pts) Name: -------------------------------------------------------------------------------, SSN -------------------------------- LAST NAME, First (Circle the alphabet
CEM 251 (Fall-2003) Final Exam (100 pts) Name: -------------------------------------------------------------------------------, SSN -------------------------------- LAST NAME, First (Circle the alphabet
CHM 101 PRACTICE TEST 1 Page 1 of 4
 CHM 101 PRACTICE TEST 1 Page 1 of 4 Please show calculations (stuffed equations) on all mathematical problems!! On the actual test, "naked answers, with no work shown, will receive no credit even if correct.
CHM 101 PRACTICE TEST 1 Page 1 of 4 Please show calculations (stuffed equations) on all mathematical problems!! On the actual test, "naked answers, with no work shown, will receive no credit even if correct.
Average and Instantaneous Velocity. p(a) p(b) Average Velocity on a < t < b =, where p(t) is the position a b
 Particle Motion Problems Particle motion problems deal with particles that are moving along the x or y axis. Thus, we are speaking of horizontal of vertical movement. The position, velocity or acceleration
Particle Motion Problems Particle motion problems deal with particles that are moving along the x or y axis. Thus, we are speaking of horizontal of vertical movement. The position, velocity or acceleration
EXAMPLES. He VIA VIIA Li Be B C N O F Ne
 59 IA EXAMPLES VIIIA H IIA IIIA IVA VA He VIA VIIA Li Be B C N O F Ne Na Mg IIIB IVB VB Al Si P VIB VIIB VIIIB IB IIB S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru
59 IA EXAMPLES VIIIA H IIA IIIA IVA VA He VIA VIIA Li Be B C N O F Ne Na Mg IIIB IVB VB Al Si P VIB VIIB VIIIB IB IIB S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru
-"l" also contributes ENERGY. Higher values for "l" mean the electron has higher energy.
 170 - Giving the four parameters will uniquely identify an electron around an atom. No two electrons in the same atom can share all four. These parameters are called QUANTUM NUMBERS. PRINCIPAL QUANTUM
170 - Giving the four parameters will uniquely identify an electron around an atom. No two electrons in the same atom can share all four. These parameters are called QUANTUM NUMBERS. PRINCIPAL QUANTUM
Atomic weight: This is a decimal number, but for radioactive elements it is replaced with a number in parenthesis.
 47 Blocks on the periodic table 11 Sodium 22.99 Atomic number: This is always a whole number. The periodic table is arranged by atomic number! Element symbol: A one or two letter abbreviation for the name
47 Blocks on the periodic table 11 Sodium 22.99 Atomic number: This is always a whole number. The periodic table is arranged by atomic number! Element symbol: A one or two letter abbreviation for the name
Circle the letters only. NO ANSWERS in the Columns!
 Chemistry 1304.001 Name (please print) Exam 5 (100 points) April 18, 2018 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Circle the letters only. NO ANSWERS in
Chemistry 1304.001 Name (please print) Exam 5 (100 points) April 18, 2018 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Circle the letters only. NO ANSWERS in
WRITING AN IONIC FORMULA
 55 WRITING AN IONIC FORMULA - if you know the ions that make up a compound, all you need to do is find the smallest ratio of cation to anion the compound needs to have an overall charge of zero Example:
55 WRITING AN IONIC FORMULA - if you know the ions that make up a compound, all you need to do is find the smallest ratio of cation to anion the compound needs to have an overall charge of zero Example:
8. Relax and do well.
 CHEM 15 Exam II John II. Gelder March 4, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last two pages includes a periodic table, a solubility
CHEM 15 Exam II John II. Gelder March 4, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last two pages includes a periodic table, a solubility
Revised October 6, EEL , Henry Zmuda. 2. Three-Phase Circuits 1
 Three Phase Circuitsit Revised October 6, 008. Three-Phase Circuits 1 Preliminary Comments and a quick review of phasors. We live in the time domain. We also assume a causal (nonpredictive) world. Real-world
Three Phase Circuitsit Revised October 6, 008. Three-Phase Circuits 1 Preliminary Comments and a quick review of phasors. We live in the time domain. We also assume a causal (nonpredictive) world. Real-world
Chapter 12 The Atom & Periodic Table- part 2
 Chapter 12 The Atom & Periodic Table- part 2 Electrons found outside the nucleus; negatively charged Protons found in the nucleus; positive charge equal in magnitude to the electron s negative charge Neutrons
Chapter 12 The Atom & Periodic Table- part 2 Electrons found outside the nucleus; negatively charged Protons found in the nucleus; positive charge equal in magnitude to the electron s negative charge Neutrons
E4 Acids, Bases, and Salts
 E4 Acids, Bases, and Salts Session One of two session lab Complete Parts 1 and 2 in lab. If time allows, start or complete Part 3. Reminder: Pre-lab report, page 112, due at start of lab. Acids and Bases
E4 Acids, Bases, and Salts Session One of two session lab Complete Parts 1 and 2 in lab. If time allows, start or complete Part 3. Reminder: Pre-lab report, page 112, due at start of lab. Acids and Bases
610B Final Exam Cover Page
 1 st Letter of Last Name NAME: 610B Final Exam Cover Page No notes or calculators of any sort allowed. You have 3 hours to complete the exam. CHEM 610B, 50995 Final Exam Fall 2003 Instructor: Dr. Brian
1 st Letter of Last Name NAME: 610B Final Exam Cover Page No notes or calculators of any sort allowed. You have 3 hours to complete the exam. CHEM 610B, 50995 Final Exam Fall 2003 Instructor: Dr. Brian
CMSC 313 Lecture 17 Postulates & Theorems of Boolean Algebra Semiconductors CMOS Logic Gates
 CMSC 313 Lecture 17 Postulates & Theorems of Boolean Algebra Semiconductors CMOS Logic Gates UMBC, CMSC313, Richard Chang Last Time Overview of second half of this course Logic gates &
CMSC 313 Lecture 17 Postulates & Theorems of Boolean Algebra Semiconductors CMOS Logic Gates UMBC, CMSC313, Richard Chang Last Time Overview of second half of this course Logic gates &
Equivalent Circuits with Multiple Damper Windings (e.g. Round rotor Machines)
 Equivalent Circuits with Multiple Damper Windings (e.g. Round rotor Machines) d axis: L fd L F - M R fd F L 1d L D - M R 1d D R fd R F e fd e F R 1d R D Subscript Notations: ( ) fd ~ field winding quantities
Equivalent Circuits with Multiple Damper Windings (e.g. Round rotor Machines) d axis: L fd L F - M R fd F L 1d L D - M R 1d D R fd R F e fd e F R 1d R D Subscript Notations: ( ) fd ~ field winding quantities
Formulas and Constants (you may remove this page)
 Formulas and Constants (you may remove this page) NA = 6.0x0 3 mol - h = 6.66x0-34 J s c =.99x0 m s - e =.60x0-9 C me = 9.09x0-3 kg Å = x0-0 m = 00 pm atm = 760 torr R = 0.006 L atm K - mol - =.34 J K
Formulas and Constants (you may remove this page) NA = 6.0x0 3 mol - h = 6.66x0-34 J s c =.99x0 m s - e =.60x0-9 C me = 9.09x0-3 kg Å = x0-0 m = 00 pm atm = 760 torr R = 0.006 L atm K - mol - =.34 J K
8. Relax and do well.
 CHEM 1314.03 Exam I John I. Gelder September 25, 1997 Name TA's Name Lab Section Please sign your name below to give permission to post, by the last 4 digits of your student I.D. number, your course scores
CHEM 1314.03 Exam I John I. Gelder September 25, 1997 Name TA's Name Lab Section Please sign your name below to give permission to post, by the last 4 digits of your student I.D. number, your course scores
... but using electron configurations to describe how aluminum bromide forms is a bit cumbersome! Can we simplify the picture a bit?
 193... but using electron configurations to describe how aluminum bromide forms is a bit cumbersome! Can we simplify the picture a bit? LEWIS NOTATION / ELECTRON-DOT NOTATION - Lewis notation represents
193... but using electron configurations to describe how aluminum bromide forms is a bit cumbersome! Can we simplify the picture a bit? LEWIS NOTATION / ELECTRON-DOT NOTATION - Lewis notation represents
8. Relax and do well.
 CHEM 1014 Exam I John I. Gelder September 16, 1999 Name TA's Name Lab Section Please sign your name below to give permission to post your course scores on homework, laboratories and exams. If you do not
CHEM 1014 Exam I John I. Gelder September 16, 1999 Name TA's Name Lab Section Please sign your name below to give permission to post your course scores on homework, laboratories and exams. If you do not
- A CHEMICAL BOND is a strong attractive force between the atoms in a compound. attractive forces between oppositely charged ions
 CHEMICAL BONDS - A CHEMICAL BOND is a strong attractive force between the atoms in a compound. 3 TYPES OF CHEMICAL BOND Ionic bonds attractive forces between oppositely charged ions sodium chloride Covalent
CHEMICAL BONDS - A CHEMICAL BOND is a strong attractive force between the atoms in a compound. 3 TYPES OF CHEMICAL BOND Ionic bonds attractive forces between oppositely charged ions sodium chloride Covalent
Atomic weight: This is a decimal number, but for radioactive elements it is replaced with a number in parenthesis.
 47 Blocks on the periodic table 11 Sodium 22.99 Atomic number: This is always a whole number. The periodic table is arranged by atomic number! Element symbol: A one or two letter abbreviation for the name
47 Blocks on the periodic table 11 Sodium 22.99 Atomic number: This is always a whole number. The periodic table is arranged by atomic number! Element symbol: A one or two letter abbreviation for the name
IZRAČUN MEMBRANSKE RAZTEZNE POSODE - "MRP" za HLADNOVODNE SISTEME (DIN 4807/2)
 IZPIS IZRAČUN MEMBRANSKE RAZTEZNE POSODE - "MRP" za HLADNOVODNE SISTEME Izhodiščni podatki: Objkt : Vrtc Kamnitnik Projkt : PZI Uporaba MRP : Črpalna vrtina Datum : 30.8.2017 Obdlal : Zupan Skupna hladilna
IZPIS IZRAČUN MEMBRANSKE RAZTEZNE POSODE - "MRP" za HLADNOVODNE SISTEME Izhodiščni podatki: Objkt : Vrtc Kamnitnik Projkt : PZI Uporaba MRP : Črpalna vrtina Datum : 30.8.2017 Obdlal : Zupan Skupna hladilna
Example: Helium has an atomic number of 2. Every helium atom has two protons in its nucleus.
 59 Atomic terms - ATOMIC NUMBER: The number of protons in the atomic nucleus. Each ELEMENT has the SAME NUMBER OF PROTONS in every nucleus. In neutral atoms, the number of ELECTRONS is also equal to the
59 Atomic terms - ATOMIC NUMBER: The number of protons in the atomic nucleus. Each ELEMENT has the SAME NUMBER OF PROTONS in every nucleus. In neutral atoms, the number of ELECTRONS is also equal to the
The Periodic Table. Periodic Properties. Can you explain this graph? Valence Electrons. Valence Electrons. Paramagnetism
 Periodic Properties Atomic & Ionic Radius Energy Electron Affinity We want to understand the variations in these properties in terms of electron configurations. The Periodic Table Elements in a column
Periodic Properties Atomic & Ionic Radius Energy Electron Affinity We want to understand the variations in these properties in terms of electron configurations. The Periodic Table Elements in a column
WRITING AN IONIC FORMULA
 55 WRITING AN IONIC FORMULA - if you know the ions that make up a compound, all you need to do is find the smallest ratio of cation to anion the compound needs to have an overall charge of zero Example:
55 WRITING AN IONIC FORMULA - if you know the ions that make up a compound, all you need to do is find the smallest ratio of cation to anion the compound needs to have an overall charge of zero Example:
Splošna teorija električnih strojev s preizkušanjem
 Univerza v Ljubljani Fakulteta za elektrotehniko Danilo Makuc Splošna teorija električnih strojev s preizkušanjem Laboratorijske vaje Danilo Makuc, FE UNI LJ, študijsko leto 2010/11 Kazalo UOD... III LABORATORIJSKA
Univerza v Ljubljani Fakulteta za elektrotehniko Danilo Makuc Splošna teorija električnih strojev s preizkušanjem Laboratorijske vaje Danilo Makuc, FE UNI LJ, študijsko leto 2010/11 Kazalo UOD... III LABORATORIJSKA
8. Relax and do well.
 CHEM 1314 3;30 pm Theory Exam III John III. Gelder November 13, 2002 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 8 different pages. The last page include a periodic
CHEM 1314 3;30 pm Theory Exam III John III. Gelder November 13, 2002 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 8 different pages. The last page include a periodic
Acta Chim. Slov. 2003, 50,
 771 IMPACT OF STRUCTURED PACKING ON BUBBE COUMN MASS TRANSFER CHARACTERISTICS EVAUATION. Part 3. Sensitivity of ADM Volumetric Mass Transfer Coefficient evaluation Ana akota Faculty of Chemistry and Chemical
771 IMPACT OF STRUCTURED PACKING ON BUBBE COUMN MASS TRANSFER CHARACTERISTICS EVAUATION. Part 3. Sensitivity of ADM Volumetric Mass Transfer Coefficient evaluation Ana akota Faculty of Chemistry and Chemical
8. Relax and do well.
 CHEM 1225 Exam I John I. Gelder February 4, 1999 Name KEY TA's Name Lab Section Please sign your name below to give permission to post your course scores on homework, laboratories and exams. If you do
CHEM 1225 Exam I John I. Gelder February 4, 1999 Name KEY TA's Name Lab Section Please sign your name below to give permission to post your course scores on homework, laboratories and exams. If you do
E5 Lewis Acids and Bases: lab 2. Session two lab Parts 2B, 3, and 4. Session one lab Parts 1and 2A. Aquo Complex Ions
 E5 Lewis Acids and Bases: lab 2 Session one lab Parts 1and 2A Session two lab Parts 2B, 3, and 4 Part 2B. Complexation, Structure and Periodicity Compare the reactivity of aquo complex ions containing
E5 Lewis Acids and Bases: lab 2 Session one lab Parts 1and 2A Session two lab Parts 2B, 3, and 4 Part 2B. Complexation, Structure and Periodicity Compare the reactivity of aquo complex ions containing
Circle the letters only. NO ANSWERS in the Columns! (3 points each)
 Chemistry 1304.001 Name (please print) Exam 4 (100 points) April 12, 2017 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Circle the letters only. NO ANSWERS in
Chemistry 1304.001 Name (please print) Exam 4 (100 points) April 12, 2017 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Circle the letters only. NO ANSWERS in
CHEM 107 (Spring-2004) Exam 2 (100 pts)
 CHEM 107 (Spring-2004) Exam 2 (100 pts) Name: ------------------------------------------------------------------------, SSN -------------------------------- LAST NAME, First (Circle the alphabet segment
CHEM 107 (Spring-2004) Exam 2 (100 pts) Name: ------------------------------------------------------------------------, SSN -------------------------------- LAST NAME, First (Circle the alphabet segment
Chemistry 2 Exam Roane State Academic Festival. Name (print neatly) School
 Name (print neatly) School There are fifteen question on this exam. Each question is weighted equally. n the answer sheet, write your name in the space provided and your answers in the blanks provided.
Name (print neatly) School There are fifteen question on this exam. Each question is weighted equally. n the answer sheet, write your name in the space provided and your answers in the blanks provided.
Fun and Fascinating Bible Reference for Kids Ages 8 to 12. starts on page 3! starts on page 163!
 F a Faa R K 8 12 a a 3! a a 163! 2013 a P, I. ISN 978-1-62416-216-9. N a a a a a, a,. C a a a a P, a 500 a a aa a. W, : F G: K Fa a Q &, a P, I. U. L aa a a a Fa a Q & a. C a 2 (M) Ta H P M (K) Wa P a
F a Faa R K 8 12 a a 3! a a 163! 2013 a P, I. ISN 978-1-62416-216-9. N a a a a a, a,. C a a a a P, a 500 a a aa a. W, : F G: K Fa a Q &, a P, I. U. L aa a a a Fa a Q & a. C a 2 (M) Ta H P M (K) Wa P a
I M P O R T A N T S A F E T Y I N S T R U C T I O N S W h e n u s i n g t h i s e l e c t r o n i c d e v i c e, b a s i c p r e c a u t i o n s s h o
 I M P O R T A N T S A F E T Y I N S T R U C T I O N S W h e n u s i n g t h i s e l e c t r o n i c d e v i c e, b a s i c p r e c a u t i o n s s h o u l d a l w a y s b e t a k e n, i n c l u d f o l
I M P O R T A N T S A F E T Y I N S T R U C T I O N S W h e n u s i n g t h i s e l e c t r o n i c d e v i c e, b a s i c p r e c a u t i o n s s h o u l d a l w a y s b e t a k e n, i n c l u d f o l
Analogna elektronska vezja. Uvodna vaja
 Analogna elektronska vezja Uvodna vaja Povzetek Namen uvodne vaje je, da študenti spoznajo orodja, ki jih bojo uporabljali pri laboratorijskih vajah predmeta Analogna elektronska vezja in sicer: podatkovne
Analogna elektronska vezja Uvodna vaja Povzetek Namen uvodne vaje je, da študenti spoznajo orodja, ki jih bojo uporabljali pri laboratorijskih vajah predmeta Analogna elektronska vezja in sicer: podatkovne
K. 27 Co. 28 Ni. 29 Cu Rb. 46 Pd. 45 Rh. 47 Ag Cs Ir. 78 Pt.
 1 IA 1 H Hydrogen 1.01 Atomic number Element symbol Element name Atomic mass VIIIA 1 H 1.01 IIA IIIA IVA VA VIA VIIA 2 He 4.00 Metalloids 3 Li 6.94 4 Be 9.01 5 B 10.81 6 C 12.01 7 N 14.01 8 O 16.00 9 F
1 IA 1 H Hydrogen 1.01 Atomic number Element symbol Element name Atomic mass VIIIA 1 H 1.01 IIA IIIA IVA VA VIA VIIA 2 He 4.00 Metalloids 3 Li 6.94 4 Be 9.01 5 B 10.81 6 C 12.01 7 N 14.01 8 O 16.00 9 F
- Atomic line spectra are UNIQUE to each element. They're like atomic "fingerprints".
 - Atomic line spectra are UNIQUE to each element. They're like atomic "fingerprints". - Problem was that the current model of the atom completely failed to explain why atoms emitted these lines. An orbit
- Atomic line spectra are UNIQUE to each element. They're like atomic "fingerprints". - Problem was that the current model of the atom completely failed to explain why atoms emitted these lines. An orbit
7. Relax and do well.
 CHEM 1014 Exam III John III. Gelder November 18, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
CHEM 1014 Exam III John III. Gelder November 18, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
E5 Lewis Acids and Bases: lab 2. Session two lab Parts 2B, 3, and 4. Session one lab Parts 1and 2A. Aquo Complex Ions. Aquo Complex Ion Reactions
 E5 Lewis Acids and Bases: lab 2 Session one lab Parts 1and 2A Part 2B. Complexation, Structure and Periodicity Compare the reactivity of aquo complex ions containing pre-transition, transition, and post-transition
E5 Lewis Acids and Bases: lab 2 Session one lab Parts 1and 2A Part 2B. Complexation, Structure and Periodicity Compare the reactivity of aquo complex ions containing pre-transition, transition, and post-transition
1 2 U CV. K dq I dt J nqv d J V IR P VI
 o 5 o T C T F 3 9 T K T o C 73.5 L L T V VT Q mct nct Q F V ml F V dq A H k TH TC L pv nrt 3 Ktr nrt 3 CV R ideal monatomic gas 5 CV R ideal diatomic gas w/o vibration V W pdv V U Q W W Q e Q Q e Carnot
o 5 o T C T F 3 9 T K T o C 73.5 L L T V VT Q mct nct Q F V ml F V dq A H k TH TC L pv nrt 3 Ktr nrt 3 CV R ideal monatomic gas 5 CV R ideal diatomic gas w/o vibration V W pdv V U Q W W Q e Q Q e Carnot
Halogens HALOGENS. Parts 2A and 2B. Chem : Feb. 19, 20 and March 3. Compare the properties and reactivity of the halogens and halides
 Chem. 125-126: Feb. 19, 20 and March 3 Experiment 3 Session 2 (Three hour lab) Complete Experiment 3 Parts 2B and 3 Complete team report Complete discussion presentation Parts 2A and 2B Compare the properties
Chem. 125-126: Feb. 19, 20 and March 3 Experiment 3 Session 2 (Three hour lab) Complete Experiment 3 Parts 2B and 3 Complete team report Complete discussion presentation Parts 2A and 2B Compare the properties
BROOKLYN COLLEGE Department of Chemistry. Chemistry 1 Second Lecture Exam Nov. 27, Name Page 1 of 5
 BROOKLYN COLLEGE Department of Chemistry Chemistry 1 Second Lecture Exam Nov. 27, 2002 Name Page 1 of 5 Circle the name of your lab instructor Kobrak, Zhou, Girotto, Hussey, Du Before you begin the exam,
BROOKLYN COLLEGE Department of Chemistry Chemistry 1 Second Lecture Exam Nov. 27, 2002 Name Page 1 of 5 Circle the name of your lab instructor Kobrak, Zhou, Girotto, Hussey, Du Before you begin the exam,
