Introduction to Interfacial Segregation. Xiaozhe Zhang 10/02/2015
|
|
|
- Aron Black
- 6 years ago
- Views:
Transcription
1 Introducton to Interfacal Segregaton Xaozhe Zhang 10/02/2015
2 Interfacal egregaton Segregaton n materal refer to the enrchment of a materal conttuent at a free urface or an nternal nterface of a materal. In a polycrytallne old, a egregaton te can be a dlocaton, gran boundary, tackng fault, or an nterface wth a precptate or econdary phae wthn the old. There are two recognzed type of egregaton: equlbrum egregaton and non-equlbrum egregaton
3 Interfacal Thermodynamc Follow Gbb. Interfacal energy denoted by (wth typcal unt of [mj/m 2 ]). It defned a the reverble work needed to create unt area of urface, at contant temperature, volume (or preure), and chemcal potental. Ue lqud-vapor urface a an example,.e., conder a ytem compoed of a lqud and a vapor phae at equlbrum, eparated by an nterface.
4 Interfacal exce nternal energy, E : E = E - E' E'' and nterfacal exce no. of mole of component : n = n - n ' - n " Excepton: no nterfacal exce volume, V = V' + V'' Internal energe of phae ' and '' are wrtten (a uual): de de TdS PdV TdS PdV dn dn and nterfacal exce nternal energy (no volume) wrtten: de T Interfacal exce properte de TdS da dn ds ds ds P dv dv da dn dn dn de TdS PdV da dn
5 Interfacal Thermodynamc All other thermodynamc properte can be wrtten from the prevou relaton: e.g. Gbb free energy, G = E + PV - TS dg de PdV VdP TdS SdT SdT VdP da Note! The urface exce quantte are arbtrary, nce they depend on precely where the dvdng urface located wthn the dffue regon aocated wth the nterface. We hall ee later how th ue may be addreed. dn
6 Gbb adorpton equaton and otherm The expreon for de, wrtten only n term of extenve ndependent varable (ds, da, dn ), whle the ntenve varable (T,, ) are contant n the equlbrated ytem. Th make ntegraton of de traghtforward: Re-dfferentatng yeld: E TS A n de TdS S dt da Ad Comparng wth the expreon for de, we conclude: S dt Ad n d 0 We now defne the followng pecfc nterfacal exce quantte: S / A and n / A where referred to a the adorpton of component dn n d
7 and obtan: d dt d Gbb adorpton equaton At contant temperature, th mplfe to the Gbb adorpton otherm. For a twocomponent ytem, t can be wrtten: d 1 d 1 2 d 2 Th may be mplfed further by the ue of the Gbb-Duhem equaton: n 1 d 1 n 2 d 2 0 It conventonal to take component 1 and 2 to be the olvent and olute, repectvely, and to elmnate 1 from the adorpton equaton: d 1 d 2 n 2 n 1 2 The l.h.. of the above equaton meaurable, therefore the r.h.. cannot be arbtrary.
8 Approxmate form of the Gbb adorpton otherm The chemcal potental may be expreed a: 2 = 2 + RT ln a 2, where a, the actvty and the tandard tate chemcal potental. For deal oluton, a 2 = x 2, where x 2 the mole or atom fracton. For dlute oluton, a 2 = k o x 2, where k o Henry' Law contant. In both of thee cae, d 2 = RT d(ln x 2 ). Thu, n both thoe cae the Gbb otherm may be wrtten: 1 RT d 1 d lnx 2 n 2 n 1 2 In partcular, n dlute oluton, n' 2 << n' 1, o that we can wrte: 1 RT d d lnx 2 2 Th the mot commonly ued form of the Gbb adorpton otherm.
9 Stattcal Thermodynamc Model of The Gbban approach not alway eay to ue Nether, nor t varaton wth compoton, are eay to meaure n old. 2 dffcult to meaure drectly, becaue nterfacal compoton profle can extend ome dtance from the nterface, and one mut determne the compoton profle of both component n the mot general cae. Gbban thermodynamc do not provde a relatonhp between 2 and 2. Wthout uch nformaton, the Gbb adorpton otherm cannot be ntegrated. Thee factor have led to the development of varou model whch can overcome ome of thee problem. Here we hall brefly decrbe one of the mplet of thee model. Segregaton
10 Monolayer (urface) egregaton model (regular oluton approxmaton) x 1 x x 1 x exp E eg RT x and x are the atom fracton of the olute n the urface monolayer and the bulk, repectvely, and eg the energy of egregaton,.e. the energy change reultng from exchangng a olute atom n the bulk wth a olvent atom at the urface. E eg ( B A ) 2 z (x x ) z v (x 1 ) 2 E el A and B are the urface energe of the pure component (B=olute), the area per mole n the monolayer, = AB ( AA + BB )/2 the regular oluton contant, j are bond energe between -j par of atom, the n-plane coordnaton and z z v half of the out-of plane bond made by an atom, and E el the elatc tran energy of a olute atom.
11 Typcal reult from th type of model 1 monolayer multlayer 15 x = 0.01; E el = 2727 J/mol A = 1650 mj/m 2 ; B = 918 mj/m layer atom fracton x (bulk) = 0.1 ln(x /1-x ) 5 0 = 562 J/mol = 0 J/mol = -562 J/mol layer number 1/T(K) Alo, the relatonhp between x and : (x x)/ From th relaton, and the one between x and x, t poble to ntegrate the Gbb adorpton otherm and obtan:
12 RT where k a urface enrchment factor gven by ln(1 kx) x x kx /(1 kx) (mj/m 2 ) T=1000 K T=800 K T=600 K T=400 K Note! The urface energy not proportonal to the adorpton, rather t the lope, d/d, that proportonal to adorpton. x
13 Thank you for your tme!
Open Systems: Chemical Potential and Partial Molar Quantities Chemical Potential
 Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
You must not circulate this book in any other binding or cover and you must impose this same condition on any acquirer.
 6 Interfacal thermodynamc: Gbb equaton Luuk K. Koopal Chapter 6, Interfacal thermodynamc: Gbb equaton n Interface Scence, Second edton, 008, Wagenngen Unverty, Wagenngen, The Netherland. Avalable va: http://www.reearchgate.net/profle/luuk_koopal
6 Interfacal thermodynamc: Gbb equaton Luuk K. Koopal Chapter 6, Interfacal thermodynamc: Gbb equaton n Interface Scence, Second edton, 008, Wagenngen Unverty, Wagenngen, The Netherland. Avalable va: http://www.reearchgate.net/profle/luuk_koopal
Appendix II Summary of Important Equations
 W. M. Whte Geochemstry Equatons of State: Ideal GasLaw: Coeffcent of Thermal Expanson: Compressblty: Van der Waals Equaton: The Laws of Thermdynamcs: Frst Law: Appendx II Summary of Important Equatons
W. M. Whte Geochemstry Equatons of State: Ideal GasLaw: Coeffcent of Thermal Expanson: Compressblty: Van der Waals Equaton: The Laws of Thermdynamcs: Frst Law: Appendx II Summary of Important Equatons
No! Yes! Only if reactions occur! Yes! Ideal Gas, change in temperature or pressure. Survey Results. Class 15. Is the following possible?
 Survey Reult Chapter 5-6 (where we are gong) % of Student 45% 40% 35% 30% 25% 20% 15% 10% 5% 0% Hour Spent on ChE 273 1-2 3-4 5-6 7-8 9-10 11+ Hour/Week 2008 2009 2010 2011 2012 2013 2014 2015 2017 F17
Survey Reult Chapter 5-6 (where we are gong) % of Student 45% 40% 35% 30% 25% 20% 15% 10% 5% 0% Hour Spent on ChE 273 1-2 3-4 5-6 7-8 9-10 11+ Hour/Week 2008 2009 2010 2011 2012 2013 2014 2015 2017 F17
NAME and Section No. it is found that 0.6 mol of O
 NAME and Secton No. Chemstry 391 Fall 7 Exam III KEY 1. (3 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). In the reacton 3O O3 t s found that.6 mol of O are consumed. Fnd
NAME and Secton No. Chemstry 391 Fall 7 Exam III KEY 1. (3 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). In the reacton 3O O3 t s found that.6 mol of O are consumed. Fnd
Not at Steady State! Yes! Only if reactions occur! Yes! Ideal Gas, change in temperature or pressure. Yes! Class 15. Is the following possible?
 Chapter 5-6 (where we are gong) Ideal gae and lqud (today) Dente Partal preure Non-deal gae (next tme) Eqn. of tate Reduced preure and temperature Compreblty chart (z) Vapor-lqud ytem (Ch. 6) Vapor preure
Chapter 5-6 (where we are gong) Ideal gae and lqud (today) Dente Partal preure Non-deal gae (next tme) Eqn. of tate Reduced preure and temperature Compreblty chart (z) Vapor-lqud ytem (Ch. 6) Vapor preure
Lecture. Polymer Thermodynamics 0331 L Chemical Potential
 Prof. Dr. rer. nat. habl. S. Enders Faculty III for Process Scence Insttute of Chemcal Engneerng Department of Thermodynamcs Lecture Polymer Thermodynamcs 033 L 337 3. Chemcal Potental Polymer Thermodynamcs
Prof. Dr. rer. nat. habl. S. Enders Faculty III for Process Scence Insttute of Chemcal Engneerng Department of Thermodynamcs Lecture Polymer Thermodynamcs 033 L 337 3. Chemcal Potental Polymer Thermodynamcs
...Thermodynamics. If Clausius Clapeyron fails. l T (v 2 v 1 ) = 0/0 Second order phase transition ( S, v = 0)
 If Clausus Clapeyron fals ( ) dp dt pb =...Thermodynamcs l T (v 2 v 1 ) = 0/0 Second order phase transton ( S, v = 0) ( ) dp = c P,1 c P,2 dt Tv(β 1 β 2 ) Two phases ntermngled Ferromagnet (Excess spn-up
If Clausus Clapeyron fals ( ) dp dt pb =...Thermodynamcs l T (v 2 v 1 ) = 0/0 Second order phase transton ( S, v = 0) ( ) dp = c P,1 c P,2 dt Tv(β 1 β 2 ) Two phases ntermngled Ferromagnet (Excess spn-up
3. Be able to derive the chemical equilibrium constants from statistical mechanics.
 Lecture #17 1 Lecture 17 Objectves: 1. Notaton of chemcal reactons 2. General equlbrum 3. Be able to derve the chemcal equlbrum constants from statstcal mechancs. 4. Identfy how nondeal behavor can be
Lecture #17 1 Lecture 17 Objectves: 1. Notaton of chemcal reactons 2. General equlbrum 3. Be able to derve the chemcal equlbrum constants from statstcal mechancs. 4. Identfy how nondeal behavor can be
CHAPTER X PHASE-CHANGE PROBLEMS
 Chapter X Phae-Change Problem December 3, 18 917 CHAPER X PHASE-CHANGE PROBLEMS X.1 Introducton Clacal Stefan Problem Geometry of Phae Change Problem Interface Condton X. Analytcal Soluton for Soldfcaton
Chapter X Phae-Change Problem December 3, 18 917 CHAPER X PHASE-CHANGE PROBLEMS X.1 Introducton Clacal Stefan Problem Geometry of Phae Change Problem Interface Condton X. Analytcal Soluton for Soldfcaton
a for save as PDF Chemistry 163B Introduction to Multicomponent Systems and Partial Molar Quantities
 a for save as PDF Chemstry 163B Introducton to Multcomponent Systems and Partal Molar Quanttes 1 the problem of partal mmolar quanttes mx: 10 moles ethanol C 2 H 5 OH (580 ml) wth 1 mole water H 2 O (18
a for save as PDF Chemstry 163B Introducton to Multcomponent Systems and Partal Molar Quanttes 1 the problem of partal mmolar quanttes mx: 10 moles ethanol C 2 H 5 OH (580 ml) wth 1 mole water H 2 O (18
Introduction to Vapor/Liquid Equilibrium, part 2. Raoult s Law:
 CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
3) Thermodynamic equation to characterize interfaces
 3) Thermodynamc equaton to characterze nterfaces 3.1) Gbbs Model Realty: rapd contnuous change of chemcal and thermodynamc propertes Replaced by model (constant propertes up to the surface) uv bulk uv
3) Thermodynamc equaton to characterze nterfaces 3.1) Gbbs Model Realty: rapd contnuous change of chemcal and thermodynamc propertes Replaced by model (constant propertes up to the surface) uv bulk uv
Problem #1. Known: All required parameters. Schematic: Find: Depth of freezing as function of time. Strategy:
 BEE 3500 013 Prelm Soluton Problem #1 Known: All requred parameter. Schematc: Fnd: Depth of freezng a functon of tme. Strategy: In thee mplfed analy for freezng tme, a wa done n cla for a lab geometry,
BEE 3500 013 Prelm Soluton Problem #1 Known: All requred parameter. Schematc: Fnd: Depth of freezng a functon of tme. Strategy: In thee mplfed analy for freezng tme, a wa done n cla for a lab geometry,
Supplementary Notes for Chapter 9 Mixture Thermodynamics
 Supplementary Notes for Chapter 9 Mxture Thermodynamcs Key ponts Nne major topcs of Chapter 9 are revewed below: 1. Notaton and operatonal equatons for mxtures 2. PVTN EOSs for mxtures 3. General effects
Supplementary Notes for Chapter 9 Mxture Thermodynamcs Key ponts Nne major topcs of Chapter 9 are revewed below: 1. Notaton and operatonal equatons for mxtures 2. PVTN EOSs for mxtures 3. General effects
Lecture 2 Thermodynamics and stability October 30, 2009
 The tablty of thn (oft) flm Lecture 2 Thermodynamc and tablty October 30, 2009 Ian Morron 2009 Revew of Lecture 1 The djonng preure a jump n preure at the boundary. It doe not vary between the plate. 1
The tablty of thn (oft) flm Lecture 2 Thermodynamc and tablty October 30, 2009 Ian Morron 2009 Revew of Lecture 1 The djonng preure a jump n preure at the boundary. It doe not vary between the plate. 1
CHEMICAL REACTIONS AND DIFFUSION
 CHEMICAL REACTIONS AND DIFFUSION A.K.A. NETWORK THERMODYNAMICS BACKGROUND Classcal thermodynamcs descrbes equlbrum states. Non-equlbrum thermodynamcs descrbes steady states. Network thermodynamcs descrbes
CHEMICAL REACTIONS AND DIFFUSION A.K.A. NETWORK THERMODYNAMICS BACKGROUND Classcal thermodynamcs descrbes equlbrum states. Non-equlbrum thermodynamcs descrbes steady states. Network thermodynamcs descrbes
Review of Classical Thermodynamics
 Revew of Classcal hermodynamcs Physcs 4362, Lecture #1, 2 Syllabus What s hermodynamcs? 1 [A law] s more mpressve the greater the smplcty of ts premses, the more dfferent are the knds of thngs t relates,
Revew of Classcal hermodynamcs Physcs 4362, Lecture #1, 2 Syllabus What s hermodynamcs? 1 [A law] s more mpressve the greater the smplcty of ts premses, the more dfferent are the knds of thngs t relates,
4.2 Chemical Driving Force
 4.2. CHEMICL DRIVING FORCE 103 4.2 Chemcal Drvng Force second effect of a chemcal concentraton gradent on dffuson s to change the nature of the drvng force. Ths s because dffuson changes the bondng n a
4.2. CHEMICL DRIVING FORCE 103 4.2 Chemcal Drvng Force second effect of a chemcal concentraton gradent on dffuson s to change the nature of the drvng force. Ths s because dffuson changes the bondng n a
Harmonic oscillator approximation
 armonc ocllator approxmaton armonc ocllator approxmaton Euaton to be olved We are fndng a mnmum of the functon under the retrcton where W P, P,..., P, Q, Q,..., Q P, P,..., P, Q, Q,..., Q lnwgner functon
armonc ocllator approxmaton armonc ocllator approxmaton Euaton to be olved We are fndng a mnmum of the functon under the retrcton where W P, P,..., P, Q, Q,..., Q P, P,..., P, Q, Q,..., Q lnwgner functon
University of Washington Department of Chemistry Chemistry 452/456 Summer Quarter 2014
 Lecture 12 7/25/14 ERD: 7.1-7.5 Devoe: 8.1.1-8.1.2, 8.2.1-8.2.3, 8.4.1-8.4.3 Unversty o Washngton Department o Chemstry Chemstry 452/456 Summer Quarter 2014 A. Free Energy and Changes n Composton: The
Lecture 12 7/25/14 ERD: 7.1-7.5 Devoe: 8.1.1-8.1.2, 8.2.1-8.2.3, 8.4.1-8.4.3 Unversty o Washngton Department o Chemstry Chemstry 452/456 Summer Quarter 2014 A. Free Energy and Changes n Composton: The
Thermodynamics II. Department of Chemical Engineering. Prof. Kim, Jong Hak
 Thermodynamcs II Department of Chemcal Engneerng Prof. Km, Jong Hak Soluton Thermodynamcs : theory Obectve : lay the theoretcal foundaton for applcatons of thermodynamcs to gas mxture and lqud soluton
Thermodynamcs II Department of Chemcal Engneerng Prof. Km, Jong Hak Soluton Thermodynamcs : theory Obectve : lay the theoretcal foundaton for applcatons of thermodynamcs to gas mxture and lqud soluton
Non-Ideality Through Fugacity and Activity
 Non-Idealty Through Fugacty and Actvty S. Patel Deartment of Chemstry and Bochemstry, Unversty of Delaware, Newark, Delaware 19716, USA Corresondng author. E-mal: saatel@udel.edu 1 I. FUGACITY In ths dscusson,
Non-Idealty Through Fugacty and Actvty S. Patel Deartment of Chemstry and Bochemstry, Unversty of Delaware, Newark, Delaware 19716, USA Corresondng author. E-mal: saatel@udel.edu 1 I. FUGACITY In ths dscusson,
Problem Free Expansion of Ideal Gas
 Problem 4.3 Free Expanon o Ideal Ga In general: ds ds du P dv P dv NR V dn Snce U o deal ga ndependent on olume (du=), and N = cont n the proce: dv In a ere o nntemal ree expanon, entropy change by: S
Problem 4.3 Free Expanon o Ideal Ga In general: ds ds du P dv P dv NR V dn Snce U o deal ga ndependent on olume (du=), and N = cont n the proce: dv In a ere o nntemal ree expanon, entropy change by: S
Chapter 11. Supplemental Text Material. The method of steepest ascent can be derived as follows. Suppose that we have fit a firstorder
 S-. The Method of Steepet cent Chapter. Supplemental Text Materal The method of teepet acent can be derved a follow. Suppoe that we have ft a frtorder model y = β + β x and we wh to ue th model to determne
S-. The Method of Steepet cent Chapter. Supplemental Text Materal The method of teepet acent can be derved a follow. Suppoe that we have ft a frtorder model y = β + β x and we wh to ue th model to determne
APPLICATIONS: CHEMICAL AND PHASE EQUILIBRIA
 5.60 Sprn 2007 Lecture #28 pae PPLICTIOS: CHMICL D PHS QUILIBRI pply tattcal mechanc to develop mcrocopc model for problem you ve treated o far wth macrocopc thermodynamc 0 Product Reactant Separated atom
5.60 Sprn 2007 Lecture #28 pae PPLICTIOS: CHMICL D PHS QUILIBRI pply tattcal mechanc to develop mcrocopc model for problem you ve treated o far wth macrocopc thermodynamc 0 Product Reactant Separated atom
Improvements on Waring s Problem
 Imrovement on Warng Problem L An-Png Bejng 85, PR Chna al@nacom Abtract By a new recurve algorthm for the auxlary equaton, n th aer, we wll gve ome mrovement for Warng roblem Keyword: Warng Problem, Hardy-Lttlewood
Imrovement on Warng Problem L An-Png Bejng 85, PR Chna al@nacom Abtract By a new recurve algorthm for the auxlary equaton, n th aer, we wll gve ome mrovement for Warng roblem Keyword: Warng Problem, Hardy-Lttlewood
Introduction to Statistical Methods
 Introducton to Statstcal Methods Physcs 4362, Lecture #3 hermodynamcs Classcal Statstcal Knetc heory Classcal hermodynamcs Macroscopc approach General propertes of the system Macroscopc varables 1 hermodynamc
Introducton to Statstcal Methods Physcs 4362, Lecture #3 hermodynamcs Classcal Statstcal Knetc heory Classcal hermodynamcs Macroscopc approach General propertes of the system Macroscopc varables 1 hermodynamc
NAME and Section No.
 Chemstry 391 Fall 2007 Exam I KEY (Monday September 17) 1. (25 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). Defne the terms: open system, closed system and solated system
Chemstry 391 Fall 2007 Exam I KEY (Monday September 17) 1. (25 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). Defne the terms: open system, closed system and solated system
Energy, Entropy, and Availability Balances Phase Equilibria. Nonideal Thermodynamic Property Models. Selecting an Appropriate Model
 Lecture 4. Thermodynamcs [Ch. 2] Energy, Entropy, and Avalablty Balances Phase Equlbra - Fugactes and actvty coeffcents -K-values Nondeal Thermodynamc Property Models - P-v-T equaton-of-state models -
Lecture 4. Thermodynamcs [Ch. 2] Energy, Entropy, and Avalablty Balances Phase Equlbra - Fugactes and actvty coeffcents -K-values Nondeal Thermodynamc Property Models - P-v-T equaton-of-state models -
Chapter 18, Part 1. Fundamentals of Atmospheric Modeling
 Overhead Sldes for Chapter 18, Part 1 of Fundamentals of Atmospherc Modelng by Mark Z. Jacobson Department of Cvl & Envronmental Engneerng Stanford Unversty Stanford, CA 94305-4020 January 30, 2002 Types
Overhead Sldes for Chapter 18, Part 1 of Fundamentals of Atmospherc Modelng by Mark Z. Jacobson Department of Cvl & Envronmental Engneerng Stanford Unversty Stanford, CA 94305-4020 January 30, 2002 Types
( ) 1/ 2. ( P SO2 )( P O2 ) 1/ 2.
 Chemstry 360 Dr. Jean M. Standard Problem Set 9 Solutons. The followng chemcal reacton converts sulfur doxde to sulfur troxde. SO ( g) + O ( g) SO 3 ( l). (a.) Wrte the expresson for K eq for ths reacton.
Chemstry 360 Dr. Jean M. Standard Problem Set 9 Solutons. The followng chemcal reacton converts sulfur doxde to sulfur troxde. SO ( g) + O ( g) SO 3 ( l). (a.) Wrte the expresson for K eq for ths reacton.
Osmotic pressure and protein binding
 Osmotc pressure and proten bndng Igor R. Kuznetsov, KochLab Symposum talk 5/15/09 Today we take a closer look at one of the soluton thermodynamcs key ponts from Steve s presentaton. Here t s: d[ln(k off
Osmotc pressure and proten bndng Igor R. Kuznetsov, KochLab Symposum talk 5/15/09 Today we take a closer look at one of the soluton thermodynamcs key ponts from Steve s presentaton. Here t s: d[ln(k off
If two volatile and miscible liquids are combined to form a solution, Raoult s law is not obeyed. Use the experimental data in Table 9.
 9.9 Real Solutons Exhbt Devatons from Raoult s Law If two volatle and mscble lquds are combned to form a soluton, Raoult s law s not obeyed. Use the expermental data n Table 9.3: Physcal Chemstry 00 Pearson
9.9 Real Solutons Exhbt Devatons from Raoult s Law If two volatle and mscble lquds are combned to form a soluton, Raoult s law s not obeyed. Use the expermental data n Table 9.3: Physcal Chemstry 00 Pearson
Solution Thermodynamics
 CH2351 Chemcal Engneerng Thermodynamcs II Unt I, II www.msubbu.n Soluton Thermodynamcs www.msubbu.n Dr. M. Subramanan Assocate Professor Department of Chemcal Engneerng Sr Svasubramanya Nadar College of
CH2351 Chemcal Engneerng Thermodynamcs II Unt I, II www.msubbu.n Soluton Thermodynamcs www.msubbu.n Dr. M. Subramanan Assocate Professor Department of Chemcal Engneerng Sr Svasubramanya Nadar College of
Improvements on Waring s Problem
 Improvement on Warng Problem L An-Png Bejng, PR Chna apl@nacom Abtract By a new recurve algorthm for the auxlary equaton, n th paper, we wll gve ome mprovement for Warng problem Keyword: Warng Problem,
Improvement on Warng Problem L An-Png Bejng, PR Chna apl@nacom Abtract By a new recurve algorthm for the auxlary equaton, n th paper, we wll gve ome mprovement for Warng problem Keyword: Warng Problem,
Physics 607 Exam 1. ( ) = 1, Γ( z +1) = zγ( z) x n e x2 dx = 1. e x2
 Physcs 607 Exam 1 Please be well-organzed, and show all sgnfcant steps clearly n all problems. You are graded on your wor, so please do not just wrte down answers wth no explanaton! Do all your wor on
Physcs 607 Exam 1 Please be well-organzed, and show all sgnfcant steps clearly n all problems. You are graded on your wor, so please do not just wrte down answers wth no explanaton! Do all your wor on
Additional File 1 - Detailed explanation of the expression level CPD
 Addtonal Fle - Detaled explanaton of the expreon level CPD A mentoned n the man text, the man CPD for the uterng model cont of two ndvdual factor: P( level gen P( level gen P ( level gen 2 (.).. CPD factor
Addtonal Fle - Detaled explanaton of the expreon level CPD A mentoned n the man text, the man CPD for the uterng model cont of two ndvdual factor: P( level gen P( level gen P ( level gen 2 (.).. CPD factor
Chemical Equilibrium. Chapter 6 Spontaneity of Reactive Mixtures (gases) Taking into account there are many types of work that a sysem can perform
 Ths chapter deals wth chemcal reactons (system) wth lttle or no consderaton on the surroundngs. Chemcal Equlbrum Chapter 6 Spontanety of eactve Mxtures (gases) eactants generatng products would proceed
Ths chapter deals wth chemcal reactons (system) wth lttle or no consderaton on the surroundngs. Chemcal Equlbrum Chapter 6 Spontanety of eactve Mxtures (gases) eactants generatng products would proceed
and Statistical Mechanics Material Properties
 Statstcal Mechancs and Materal Propertes By Kuno TAKAHASHI Tokyo Insttute of Technology, Tokyo 15-855, JAPA Phone/Fax +81-3-5734-3915 takahak@de.ttech.ac.jp http://www.de.ttech.ac.jp/~kt-lab/ Only for
Statstcal Mechancs and Materal Propertes By Kuno TAKAHASHI Tokyo Insttute of Technology, Tokyo 15-855, JAPA Phone/Fax +81-3-5734-3915 takahak@de.ttech.ac.jp http://www.de.ttech.ac.jp/~kt-lab/ Only for
Statistical Properties of the OLS Coefficient Estimators. 1. Introduction
 ECOOMICS 35* -- OTE 4 ECO 35* -- OTE 4 Stattcal Properte of the OLS Coeffcent Etmator Introducton We derved n ote the OLS (Ordnary Leat Square etmator ˆβ j (j, of the regreon coeffcent βj (j, n the mple
ECOOMICS 35* -- OTE 4 ECO 35* -- OTE 4 Stattcal Properte of the OLS Coeffcent Etmator Introducton We derved n ote the OLS (Ordnary Leat Square etmator ˆβ j (j, of the regreon coeffcent βj (j, n the mple
ESCI 341 Atmospheric Thermodynamics Lesson 13 Phase Changes Dr. DeCaria
 ESCI 341 Atmopherc Thermodynamc Leon 13 Phae Change Dr. DeCara Reference: Thermodynamc and an Introducton to Thermotattc, Caen Phyca Chemtry, Lene GENERAL A phae change a change between od, qud, or apor
ESCI 341 Atmopherc Thermodynamc Leon 13 Phae Change Dr. DeCara Reference: Thermodynamc and an Introducton to Thermotattc, Caen Phyca Chemtry, Lene GENERAL A phae change a change between od, qud, or apor
Specification -- Assumptions of the Simple Classical Linear Regression Model (CLRM) 1. Introduction
 ECONOMICS 35* -- NOTE ECON 35* -- NOTE Specfcaton -- Aumpton of the Smple Clacal Lnear Regreon Model (CLRM). Introducton CLRM tand for the Clacal Lnear Regreon Model. The CLRM alo known a the tandard lnear
ECONOMICS 35* -- NOTE ECON 35* -- NOTE Specfcaton -- Aumpton of the Smple Clacal Lnear Regreon Model (CLRM). Introducton CLRM tand for the Clacal Lnear Regreon Model. The CLRM alo known a the tandard lnear
Thermodynamics and statistical mechanics in materials modelling II
 Course MP3 Lecture 8/11/006 (JAE) Course MP3 Lecture 8/11/006 Thermodynamcs and statstcal mechancs n materals modellng II A bref résumé of the physcal concepts used n materals modellng Dr James Ellott.1
Course MP3 Lecture 8/11/006 (JAE) Course MP3 Lecture 8/11/006 Thermodynamcs and statstcal mechancs n materals modellng II A bref résumé of the physcal concepts used n materals modellng Dr James Ellott.1
y i x P vap 10 A T SOLUTION TO HOMEWORK #7 #Problem
 SOLUTION TO HOMEWORK #7 #roblem 1 10.1-1 a. In order to solve ths problem, we need to know what happens at the bubble pont; at ths pont, the frst bubble s formed, so we can assume that all of the number
SOLUTION TO HOMEWORK #7 #roblem 1 10.1-1 a. In order to solve ths problem, we need to know what happens at the bubble pont; at ths pont, the frst bubble s formed, so we can assume that all of the number
TP A SOLUTION. For an ideal monatomic gas U=3/2nRT, Since the process is at constant pressure Q = C. giving ) =1000/(5/2*8.31*10)
 T A SOLUTION For an deal monatomc gas U/nRT, Snce the process s at constant pressure Q C pn T gvng a: n Q /( 5 / R T ) /(5/*8.*) C V / R and C / R + R 5 / R. U U / nr T (/ ) R T ( Q / 5 / R T ) Q / 5 Q
T A SOLUTION For an deal monatomc gas U/nRT, Snce the process s at constant pressure Q C pn T gvng a: n Q /( 5 / R T ) /(5/*8.*) C V / R and C / R + R 5 / R. U U / nr T (/ ) R T ( Q / 5 / R T ) Q / 5 Q
TEST 5 (phy 240) 2. Show that the volume coefficient of thermal expansion for an ideal gas at constant pressure is temperature dependent and given by
 ES 5 (phy 40). a) Wrte the zeroth law o thermodynamcs. b) What s thermal conductvty? c) Identyng all es, draw schematcally a P dagram o the arnot cycle. d) What s the ecency o an engne and what s the coecent
ES 5 (phy 40). a) Wrte the zeroth law o thermodynamcs. b) What s thermal conductvty? c) Identyng all es, draw schematcally a P dagram o the arnot cycle. d) What s the ecency o an engne and what s the coecent
Mass Transfer Processes
 Mass Transfer Processes S. Majd Hassanzadeh Department of Earth Scences Faculty of Geoscences Utrecht Unversty Outlne: 1. Measures of Concentraton 2. Volatlzaton and Dssoluton 3. Adsorpton Processes 4.
Mass Transfer Processes S. Majd Hassanzadeh Department of Earth Scences Faculty of Geoscences Utrecht Unversty Outlne: 1. Measures of Concentraton 2. Volatlzaton and Dssoluton 3. Adsorpton Processes 4.
Chapter 6 The Effect of the GPS Systematic Errors on Deformation Parameters
 Chapter 6 The Effect of the GPS Sytematc Error on Deformaton Parameter 6.. General Beutler et al., (988) dd the frt comprehenve tudy on the GPS ytematc error. Baed on a geometrc approach and aumng a unform
Chapter 6 The Effect of the GPS Sytematc Error on Deformaton Parameter 6.. General Beutler et al., (988) dd the frt comprehenve tudy on the GPS ytematc error. Baed on a geometrc approach and aumng a unform
Small signal analysis
 Small gnal analy. ntroducton Let u conder the crcut hown n Fg., where the nonlnear retor decrbed by the equaton g v havng graphcal repreentaton hown n Fg.. ( G (t G v(t v Fg. Fg. a D current ource wherea
Small gnal analy. ntroducton Let u conder the crcut hown n Fg., where the nonlnear retor decrbed by the equaton g v havng graphcal repreentaton hown n Fg.. ( G (t G v(t v Fg. Fg. a D current ource wherea
AP Statistics Ch 3 Examining Relationships
 Introducton To tud relatonhp between varable, we mut meaure the varable on the ame group of ndvdual. If we thnk a varable ma eplan or even caue change n another varable, then the eplanator varable and
Introducton To tud relatonhp between varable, we mut meaure the varable on the ame group of ndvdual. If we thnk a varable ma eplan or even caue change n another varable, then the eplanator varable and
University of Washington Department of Chemistry Chemistry 452/456 Summer Quarter 2013
 Lecture 8/8/3 Unversty o Washngton Departent o Chestry Chestry 45/456 Suer Quarter 3 A. The Gbbs-Duhe Equaton Fro Lecture 7 and ro the dscusson n sectons A and B o ths lecture, t s clear that the actvty
Lecture 8/8/3 Unversty o Washngton Departent o Chestry Chestry 45/456 Suer Quarter 3 A. The Gbbs-Duhe Equaton Fro Lecture 7 and ro the dscusson n sectons A and B o ths lecture, t s clear that the actvty
8 Waves in Uniform Magnetized Media
 8 Wave n Unform Magnetzed Meda 81 Suceptblte The frt order current can be wrtten j = j = q d 3 p v f 1 ( r, p, t) = ɛ 0 χ E For Maxwellan dtrbuton Y n (λ) = f 0 (v, v ) = 1 πvth exp (v V ) v th 1 πv th
8 Wave n Unform Magnetzed Meda 81 Suceptblte The frt order current can be wrtten j = j = q d 3 p v f 1 ( r, p, t) = ɛ 0 χ E For Maxwellan dtrbuton Y n (λ) = f 0 (v, v ) = 1 πvth exp (v V ) v th 1 πv th
Electric and magnetic field sensor and integrator equations
 Techncal Note - TN12 Electrc and magnetc feld enor and ntegrator uaton Bertrand Da, montena technology, 1728 oen, Swtzerland Table of content 1. Equaton of the derate electrc feld enor... 1 2. Integraton
Techncal Note - TN12 Electrc and magnetc feld enor and ntegrator uaton Bertrand Da, montena technology, 1728 oen, Swtzerland Table of content 1. Equaton of the derate electrc feld enor... 1 2. Integraton
Numerical Solution of Ordinary Differential Equations
 Numercal Methods (CENG 00) CHAPTER-VI Numercal Soluton of Ordnar Dfferental Equatons 6 Introducton Dfferental equatons are equatons composed of an unknown functon and ts dervatves The followng are examples
Numercal Methods (CENG 00) CHAPTER-VI Numercal Soluton of Ordnar Dfferental Equatons 6 Introducton Dfferental equatons are equatons composed of an unknown functon and ts dervatves The followng are examples
Chapter 5. Simple Mixtures Fall Semester Physical Chemistry 1 (CHM2201)
 Chapter 5. Simple Mixtures 2011 Fall Semester Physical Chemistry 1 (CHM2201) Contents The thermodynamic description of mixtures 5.1 Partial molar quantities 5.2 The thermodynamic of Mixing 5.3 The chemical
Chapter 5. Simple Mixtures 2011 Fall Semester Physical Chemistry 1 (CHM2201) Contents The thermodynamic description of mixtures 5.1 Partial molar quantities 5.2 The thermodynamic of Mixing 5.3 The chemical
Solution Thermodynamics
 Soluton hermodynamcs usng Wagner Notaton by Stanley. Howard Department of aterals and etallurgcal Engneerng South Dakota School of nes and echnology Rapd Cty, SD 57701 January 7, 001 Soluton hermodynamcs
Soluton hermodynamcs usng Wagner Notaton by Stanley. Howard Department of aterals and etallurgcal Engneerng South Dakota School of nes and echnology Rapd Cty, SD 57701 January 7, 001 Soluton hermodynamcs
V T for n & P = constant
 Pchem 365: hermodynamcs -SUMMARY- Uwe Burghaus, Fargo, 5 9 Mnmum requrements for underneath of your pllow. However, wrte your own summary! You need to know the story behnd the equatons : Pressure : olume
Pchem 365: hermodynamcs -SUMMARY- Uwe Burghaus, Fargo, 5 9 Mnmum requrements for underneath of your pllow. However, wrte your own summary! You need to know the story behnd the equatons : Pressure : olume
HYDRODYNAMIC LIMIT FOR A GAS WITH CHEMICAL REACTIONS
 October 3, 003 8:48 WSPC/Trm Sze: 9n x 6n for Proceedng bp HYDRODYNAMIC LIMIT FOR A GAS WITH CHEMICAL REACTIONS M. BISI Dpartmento d Matematca, Unvertà d Mlano, Va Saldn 50, 0133 Mlano, Italy, E-mal: b@mat.unm.t
October 3, 003 8:48 WSPC/Trm Sze: 9n x 6n for Proceedng bp HYDRODYNAMIC LIMIT FOR A GAS WITH CHEMICAL REACTIONS M. BISI Dpartmento d Matematca, Unvertà d Mlano, Va Saldn 50, 0133 Mlano, Italy, E-mal: b@mat.unm.t
MULTIPLE REGRESSION ANALYSIS For the Case of Two Regressors
 MULTIPLE REGRESSION ANALYSIS For the Cae of Two Regreor In the followng note, leat-quare etmaton developed for multple regreon problem wth two eplanator varable, here called regreor (uch a n the Fat Food
MULTIPLE REGRESSION ANALYSIS For the Cae of Two Regreor In the followng note, leat-quare etmaton developed for multple regreon problem wth two eplanator varable, here called regreor (uch a n the Fat Food
FREE DIFFUSION INTRODUCTION
 2 FREE DIFFUSION INTRODUCTION In th chapter, we conder the mplet of tranport procee: the pave dffuon of a olute that occur when t electrochemcal potental on the two de of a permeable barrer are dfferent.
2 FREE DIFFUSION INTRODUCTION In th chapter, we conder the mplet of tranport procee: the pave dffuon of a olute that occur when t electrochemcal potental on the two de of a permeable barrer are dfferent.
University of Washington Department of Chemistry Chemistry 452/456 Summer Quarter 2014
 Lecture 16 8/4/14 Unversty o Washngton Department o Chemstry Chemstry 452/456 Summer Quarter 214. Real Vapors and Fugacty Henry s Law accounts or the propertes o extremely dlute soluton. s shown n Fgure
Lecture 16 8/4/14 Unversty o Washngton Department o Chemstry Chemstry 452/456 Summer Quarter 214. Real Vapors and Fugacty Henry s Law accounts or the propertes o extremely dlute soluton. s shown n Fgure
The influence of Stern layer conductance on the. dielectrophoretic behaviour of latex nanospheres
 The nfluence of Stern layer conductance on the delectrophoretc behavour of latex nanophere Mchael Pycraft Hughe* Bomedcal Engneerng Group, Unverty of Surrey, Guldford, GU2 7XH, UK Ncola Gavn Green Boelectronc
The nfluence of Stern layer conductance on the delectrophoretc behavour of latex nanophere Mchael Pycraft Hughe* Bomedcal Engneerng Group, Unverty of Surrey, Guldford, GU2 7XH, UK Ncola Gavn Green Boelectronc
Inductance Calculation for Conductors of Arbitrary Shape
 CRYO/02/028 Aprl 5, 2002 Inductance Calculaton for Conductors of Arbtrary Shape L. Bottura Dstrbuton: Internal Summary In ths note we descrbe a method for the numercal calculaton of nductances among conductors
CRYO/02/028 Aprl 5, 2002 Inductance Calculaton for Conductors of Arbtrary Shape L. Bottura Dstrbuton: Internal Summary In ths note we descrbe a method for the numercal calculaton of nductances among conductors
between standard Gibbs free energies of formation for products and reactants, ΔG! R = ν i ΔG f,i, we
 hermodynamcs, Statstcal hermodynamcs, and Knetcs 4 th Edton,. Engel & P. ed Ch. 6 Part Answers to Selected Problems Q6.. Q6.4. If ξ =0. mole at equlbrum, the reacton s not ery far along. hus, there would
hermodynamcs, Statstcal hermodynamcs, and Knetcs 4 th Edton,. Engel & P. ed Ch. 6 Part Answers to Selected Problems Q6.. Q6.4. If ξ =0. mole at equlbrum, the reacton s not ery far along. hus, there would
PHYS 705: Classical Mechanics. Newtonian Mechanics
 1 PHYS 705: Classcal Mechancs Newtonan Mechancs Quck Revew of Newtonan Mechancs Basc Descrpton: -An dealzed pont partcle or a system of pont partcles n an nertal reference frame [Rgd bodes (ch. 5 later)]
1 PHYS 705: Classcal Mechancs Newtonan Mechancs Quck Revew of Newtonan Mechancs Basc Descrpton: -An dealzed pont partcle or a system of pont partcles n an nertal reference frame [Rgd bodes (ch. 5 later)]
Estimation of the composition of the liquid and vapor streams exiting a flash unit with a supercritical component
 Department of Energ oltecnco d Mlano Va Lambruschn - 05 MILANO Eercses of Fundamentals of Chemcal rocesses rof. Ganpero Gropp Eercse 8 Estmaton of the composton of the lqud and vapor streams etng a unt
Department of Energ oltecnco d Mlano Va Lambruschn - 05 MILANO Eercses of Fundamentals of Chemcal rocesses rof. Ganpero Gropp Eercse 8 Estmaton of the composton of the lqud and vapor streams etng a unt
Scattering of two identical particles in the center-of. of-mass frame. (b)
 Lecture # November 5 Scatterng of two dentcal partcle Relatvtc Quantum Mechanc: The Klen-Gordon equaton Interpretaton of the Klen-Gordon equaton The Drac equaton Drac repreentaton for the matrce α and
Lecture # November 5 Scatterng of two dentcal partcle Relatvtc Quantum Mechanc: The Klen-Gordon equaton Interpretaton of the Klen-Gordon equaton The Drac equaton Drac repreentaton for the matrce α and
CHAPTER 9 LINEAR MOMENTUM, IMPULSE AND COLLISIONS
 CHAPTER 9 LINEAR MOMENTUM, IMPULSE AND COLLISIONS 103 Phy 1 9.1 Lnear Momentum The prncple o energy conervaton can be ued to olve problem that are harder to olve jut ung Newton law. It ued to decrbe moton
CHAPTER 9 LINEAR MOMENTUM, IMPULSE AND COLLISIONS 103 Phy 1 9.1 Lnear Momentum The prncple o energy conervaton can be ued to olve problem that are harder to olve jut ung Newton law. It ued to decrbe moton
Weierstraß-Institut. für Angewandte Analysis und Stochastik. Leibniz-Institut im Forschungsverbund Berlin e. V. Preprint ISSN
 Weertraß-Inttut für Angewandte Analy und Stochatk Lebnz-Inttut m Forchungverbund Berln e. V. Preprnt ISSN 2198-5855 A new perpectve on the electron tranfer: Recoverng the Butler-Volmer equaton n non-equlbrum
Weertraß-Inttut für Angewandte Analy und Stochatk Lebnz-Inttut m Forchungverbund Berln e. V. Preprnt ISSN 2198-5855 A new perpectve on the electron tranfer: Recoverng the Butler-Volmer equaton n non-equlbrum
Outlet temperature of a WGS reactor (Stage I) for the conversion of CO, applied for the abatement of CO to a fixed value.
 Department of Energy Poltecnco d Mlano Va Lambruschn 56 MILAN Exercses of Fundamentals of Chemcal Processes Prof. Ganpero Gropp Exercse utlet temperature of a WGS reactor (Stage I for the converson of
Department of Energy Poltecnco d Mlano Va Lambruschn 56 MILAN Exercses of Fundamentals of Chemcal Processes Prof. Ganpero Gropp Exercse utlet temperature of a WGS reactor (Stage I for the converson of
On the SO 2 Problem in Thermal Power Plants. 2.Two-steps chemical absorption modeling
 Internatonal Journal of Engneerng Reearch ISSN:39-689)(onlne),347-53(prnt) Volume No4, Iue No, pp : 557-56 Oct 5 On the SO Problem n Thermal Power Plant Two-tep chemcal aborpton modelng hr Boyadjev, P
Internatonal Journal of Engneerng Reearch ISSN:39-689)(onlne),347-53(prnt) Volume No4, Iue No, pp : 557-56 Oct 5 On the SO Problem n Thermal Power Plant Two-tep chemcal aborpton modelng hr Boyadjev, P
Name ID # For relatively dilute aqueous solutions the molality and molarity are approximately equal.
 Name ID # 1 CHEMISTRY 212, Lect. Sect. 002 Dr. G. L. Roberts Exam #1/Sprng 2000 Thursday, February 24, 2000 CLOSED BOOK EXM No notes or books allowed. Calculators may be used. tomc masses of nterest are
Name ID # 1 CHEMISTRY 212, Lect. Sect. 002 Dr. G. L. Roberts Exam #1/Sprng 2000 Thursday, February 24, 2000 CLOSED BOOK EXM No notes or books allowed. Calculators may be used. tomc masses of nterest are
2. SINGLE VS. MULTI POLARIZATION SAR DATA
 . SINGLE VS. MULTI POLARIZATION SAR DATA.1 Scatterng Coeffcent v. Scatterng Matrx In the prevou chapter of th document, we dealt wth the decrpton and the characterzaton of electromagnetc wave. A t wa hown,
. SINGLE VS. MULTI POLARIZATION SAR DATA.1 Scatterng Coeffcent v. Scatterng Matrx In the prevou chapter of th document, we dealt wth the decrpton and the characterzaton of electromagnetc wave. A t wa hown,
Module 3: Element Properties Lecture 1: Natural Coordinates
 Module 3: Element Propertes Lecture : Natural Coordnates Natural coordnate system s bascally a local coordnate system whch allows the specfcaton of a pont wthn the element by a set of dmensonless numbers
Module 3: Element Propertes Lecture : Natural Coordnates Natural coordnate system s bascally a local coordnate system whch allows the specfcaton of a pont wthn the element by a set of dmensonless numbers
Module 5. Cables and Arches. Version 2 CE IIT, Kharagpur
 odule 5 Cable and Arche Veron CE IIT, Kharagpur Leon 33 Two-nged Arch Veron CE IIT, Kharagpur Intructonal Objectve: After readng th chapter the tudent wll be able to 1. Compute horzontal reacton n two-hnged
odule 5 Cable and Arche Veron CE IIT, Kharagpur Leon 33 Two-nged Arch Veron CE IIT, Kharagpur Intructonal Objectve: After readng th chapter the tudent wll be able to 1. Compute horzontal reacton n two-hnged
Lecture 7: Boltzmann distribution & Thermodynamics of mixing
 Prof. Tbbtt Lecture 7 etworks & Gels Lecture 7: Boltzmann dstrbuton & Thermodynamcs of mxng 1 Suggested readng Prof. Mark W. Tbbtt ETH Zürch 13 März 018 Molecular Drvng Forces Dll and Bromberg: Chapters
Prof. Tbbtt Lecture 7 etworks & Gels Lecture 7: Boltzmann dstrbuton & Thermodynamcs of mxng 1 Suggested readng Prof. Mark W. Tbbtt ETH Zürch 13 März 018 Molecular Drvng Forces Dll and Bromberg: Chapters
Weak McCoy Ore Extensions
 Internatonal Mathematcal Forum, Vol. 6, 2, no. 2, 75-86 Weak McCoy Ore Extenon R. Mohammad, A. Mouav and M. Zahr Department of Pure Mathematc, Faculty of Mathematcal Scence Tarbat Modare Unverty, P.O.
Internatonal Mathematcal Forum, Vol. 6, 2, no. 2, 75-86 Weak McCoy Ore Extenon R. Mohammad, A. Mouav and M. Zahr Department of Pure Mathematc, Faculty of Mathematcal Scence Tarbat Modare Unverty, P.O.
Thermodynamics of Materials
 Thermdynamcs f Materals 14th Lecture 007. 4. 8 (Mnday) FUGACITY dg = Vd SdT dg = Vd at cnstant T Fr an deal gas dg = (RT/)d = RT dln Ths s true fr deal gases nly, but t wuld be nce t have a smlar frm fr
Thermdynamcs f Materals 14th Lecture 007. 4. 8 (Mnday) FUGACITY dg = Vd SdT dg = Vd at cnstant T Fr an deal gas dg = (RT/)d = RT dln Ths s true fr deal gases nly, but t wuld be nce t have a smlar frm fr
Physics 5153 Classical Mechanics. D Alembert s Principle and The Lagrangian-1
 P. Guterrez Physcs 5153 Classcal Mechancs D Alembert s Prncple and The Lagrangan 1 Introducton The prncple of vrtual work provdes a method of solvng problems of statc equlbrum wthout havng to consder the
P. Guterrez Physcs 5153 Classcal Mechancs D Alembert s Prncple and The Lagrangan 1 Introducton The prncple of vrtual work provdes a method of solvng problems of statc equlbrum wthout havng to consder the
SOLUTION MANUAL ENGLISH UNIT PROBLEMS CHAPTER 9 SONNTAG BORGNAKKE VAN WYLEN. FUNDAMENTALS of. Thermodynamics. Sixth Edition
 SOLUTION MANUAL ENGLISH UNIT PROBLEMS CHAPTER 9 SONNTAG BORGNAKKE VAN WYLEN FUNDAMENTALS of Thermodynamc Sxth Edton CONTENT SUBSECTION PROB NO. Concept-Study Gude Problem 134-141 Steady Sngle Flow Devce
SOLUTION MANUAL ENGLISH UNIT PROBLEMS CHAPTER 9 SONNTAG BORGNAKKE VAN WYLEN FUNDAMENTALS of Thermodynamc Sxth Edton CONTENT SUBSECTION PROB NO. Concept-Study Gude Problem 134-141 Steady Sngle Flow Devce
1 Routh Array: 15 points
 EE C28 / ME34 Problem Set 3 Solution Fall 2 Routh Array: 5 point Conider the ytem below, with D() k(+), w(t), G() +2, and H y() 2 ++2 2(+). Find the cloed loop tranfer function Y () R(), and range of k
EE C28 / ME34 Problem Set 3 Solution Fall 2 Routh Array: 5 point Conider the ytem below, with D() k(+), w(t), G() +2, and H y() 2 ++2 2(+). Find the cloed loop tranfer function Y () R(), and range of k
Wilbur and Ague 4 WILBUR AND AGUE; APPENDIX DR1. Two-dimensional chemical maps as well as chemical profiles were done at 15 kv using
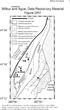 DR2006139 Wlbur and Ague 4 WILBUR AND AGUE; APPENDIX DR1 MINERAL ANALYSES Two-dmensonal chemcal maps as well as chemcal profles were done at 15 kv usng the JEOL JXA-8600 electron mcroprobe at Yale Unversty
DR2006139 Wlbur and Ague 4 WILBUR AND AGUE; APPENDIX DR1 MINERAL ANALYSES Two-dmensonal chemcal maps as well as chemcal profles were done at 15 kv usng the JEOL JXA-8600 electron mcroprobe at Yale Unversty
APPROXIMATE FUZZY REASONING BASED ON INTERPOLATION IN THE VAGUE ENVIRONMENT OF THE FUZZY RULEBASE AS A PRACTICAL ALTERNATIVE OF THE CLASSICAL CRI
 Kovác, Sz., Kóczy, L.T.: Approxmate Fuzzy Reaonng Baed on Interpolaton n the Vague Envronment of the Fuzzy Rulebae a a Practcal Alternatve of the Clacal CRI, Proceedng of the 7 th Internatonal Fuzzy Sytem
Kovác, Sz., Kóczy, L.T.: Approxmate Fuzzy Reaonng Baed on Interpolaton n the Vague Envronment of the Fuzzy Rulebae a a Practcal Alternatve of the Clacal CRI, Proceedng of the 7 th Internatonal Fuzzy Sytem
Effect of the adsorption component of the disjoining pressure on foam film drainage
 Collod J. 75 (2013) 176-180 [arxv 1209.4745] Effect of the adorpton component of the djonng preure on foam flm dranage Stoyan I. Karakahev 1, Anh V. Nguyen 2 and Roumen Tekov 1 1 Department of Phycal Chemtry,
Collod J. 75 (2013) 176-180 [arxv 1209.4745] Effect of the adorpton component of the djonng preure on foam flm dranage Stoyan I. Karakahev 1, Anh V. Nguyen 2 and Roumen Tekov 1 1 Department of Phycal Chemtry,
Thermodynamics Second Law Entropy
 Thermodynamcs Second Law Entropy Lana Sherdan De Anza College May 8, 2018 Last tme the Boltzmann dstrbuton (dstrbuton of energes) the Maxwell-Boltzmann dstrbuton (dstrbuton of speeds) the Second Law of
Thermodynamcs Second Law Entropy Lana Sherdan De Anza College May 8, 2018 Last tme the Boltzmann dstrbuton (dstrbuton of energes) the Maxwell-Boltzmann dstrbuton (dstrbuton of speeds) the Second Law of
2.3 Least-Square regressions
 .3 Leat-Square regreon Eample.10 How do chldren grow? The pattern of growth vare from chld to chld, o we can bet undertandng the general pattern b followng the average heght of a number of chldren. Here
.3 Leat-Square regreon Eample.10 How do chldren grow? The pattern of growth vare from chld to chld, o we can bet undertandng the general pattern b followng the average heght of a number of chldren. Here
Inner Product. Euclidean Space. Orthonormal Basis. Orthogonal
 Inner Product Defnton 1 () A Eucldean space s a fnte-dmensonal vector space over the reals R, wth an nner product,. Defnton 2 (Inner Product) An nner product, on a real vector space X s a symmetrc, blnear,
Inner Product Defnton 1 () A Eucldean space s a fnte-dmensonal vector space over the reals R, wth an nner product,. Defnton 2 (Inner Product) An nner product, on a real vector space X s a symmetrc, blnear,
The multivariate Gaussian probability density function for random vector X (X 1,,X ) T. diagonal term of, denoted
 Appendx Proof of heorem he multvarate Gauan probablty denty functon for random vector X (X,,X ) px exp / / x x mean and varance equal to the th dagonal term of, denoted he margnal dtrbuton of X Gauan wth
Appendx Proof of heorem he multvarate Gauan probablty denty functon for random vector X (X,,X ) px exp / / x x mean and varance equal to the th dagonal term of, denoted he margnal dtrbuton of X Gauan wth
Module 1 : The equation of continuity. Lecture 1: Equation of Continuity
 1 Module 1 : The equaton of contnuty Lecture 1: Equaton of Contnuty 2 Advanced Heat and Mass Transfer: Modules 1. THE EQUATION OF CONTINUITY : Lectures 1-6 () () () (v) (v) Overall Mass Balance Momentum
1 Module 1 : The equaton of contnuty Lecture 1: Equaton of Contnuty 2 Advanced Heat and Mass Transfer: Modules 1. THE EQUATION OF CONTINUITY : Lectures 1-6 () () () (v) (v) Overall Mass Balance Momentum
MODULE 4: ABSORPTION
 MODULE 4: ABSORPTION LECTURE NO. 3 4.4. Deign of packed tower baed on overall ma tranfer coefficient * From overall ma tranfer equation, N K ( y y ) one can write for packed tower a N A K y (y-y*) Then,
MODULE 4: ABSORPTION LECTURE NO. 3 4.4. Deign of packed tower baed on overall ma tranfer coefficient * From overall ma tranfer equation, N K ( y y ) one can write for packed tower a N A K y (y-y*) Then,
Electromagnetic scattering. Graduate Course Electrical Engineering (Communications) 1 st Semester, Sharif University of Technology
 Electromagnetc catterng Graduate Coure Electrcal Engneerng (Communcaton) 1 t Semeter, 1390-1391 Sharf Unverty of Technology Content of lecture Lecture : Bac catterng parameter Formulaton of the problem
Electromagnetc catterng Graduate Coure Electrcal Engneerng (Communcaton) 1 t Semeter, 1390-1391 Sharf Unverty of Technology Content of lecture Lecture : Bac catterng parameter Formulaton of the problem
MODIFIED GENERAL TRANSFER MATRIX METHOD FOR LINEAR ROTOR BEARING SYSTEMS , Dhenkanal, Orissa, India
 IV arn Autrala 9- July, 007 Abtract MODIFIED GENERAL TRANFER MATRIX METHOD FOR LINEAR ROTOR BEARING YTEM B B Maharath, R R Dah and T Rout Department of Mechancal Engneerng, IGIT, arang 769 6, Dhenkanal,
IV arn Autrala 9- July, 007 Abtract MODIFIED GENERAL TRANFER MATRIX METHOD FOR LINEAR ROTOR BEARING YTEM B B Maharath, R R Dah and T Rout Department of Mechancal Engneerng, IGIT, arang 769 6, Dhenkanal,
Kinetic-Energy Density-Functional Theory on a Lattice
 h an open acce artcle publhed under an ACS AuthorChoce Lcene, whch permt copyng and redtrbuton of the artcle or any adaptaton for non-commercal purpoe. Artcle Cte h: J. Chem. heory Comput. 08, 4, 407 4087
h an open acce artcle publhed under an ACS AuthorChoce Lcene, whch permt copyng and redtrbuton of the artcle or any adaptaton for non-commercal purpoe. Artcle Cte h: J. Chem. heory Comput. 08, 4, 407 4087
Chemical Engineering Department University of Washington
 Chemcal Engneerng Department Unversty of Washngton ChemE 60 - Exam I July 4, 003 - Mass Flow Rate of Steam Through a Turbne (5 onts) Steam enters a turbne at 70 o C and.8 Ma and leaves at 00 ka wth a qualty
Chemcal Engneerng Department Unversty of Washngton ChemE 60 - Exam I July 4, 003 - Mass Flow Rate of Steam Through a Turbne (5 onts) Steam enters a turbne at 70 o C and.8 Ma and leaves at 00 ka wth a qualty
Envr 210, Chapter 3, Intermolecular forces and partitioning Free energies and equilibrium partitioning chemical potential fugacity activity coef.
 Envr 20, Chapter 3, Intermolecular forces and parttonng Free energes and equlbrum parttonng chemcal potental fugacty actvty coef. phase transfer- actvty coef and fugactes more on free energes and equlbrum
Envr 20, Chapter 3, Intermolecular forces and parttonng Free energes and equlbrum parttonng chemcal potental fugacty actvty coef. phase transfer- actvty coef and fugactes more on free energes and equlbrum
Thermodynamics General
 Thermodynamcs General Lecture 1 Lecture 1 s devoted to establshng buldng blocks for dscussng thermodynamcs. In addton, the equaton of state wll be establshed. I. Buldng blocks for thermodynamcs A. Dmensons,
Thermodynamcs General Lecture 1 Lecture 1 s devoted to establshng buldng blocks for dscussng thermodynamcs. In addton, the equaton of state wll be establshed. I. Buldng blocks for thermodynamcs A. Dmensons,
Estimation of Finite Population Total under PPS Sampling in Presence of Extra Auxiliary Information
 Internatonal Journal of Stattc and Analy. ISSN 2248-9959 Volume 6, Number 1 (2016), pp. 9-16 Reearch Inda Publcaton http://www.rpublcaton.com Etmaton of Fnte Populaton Total under PPS Samplng n Preence
Internatonal Journal of Stattc and Analy. ISSN 2248-9959 Volume 6, Number 1 (2016), pp. 9-16 Reearch Inda Publcaton http://www.rpublcaton.com Etmaton of Fnte Populaton Total under PPS Samplng n Preence
10.34 Numerical Methods Applied to Chemical Engineering Fall Homework #3: Systems of Nonlinear Equations and Optimization
 10.34 Numercal Methods Appled to Chemcal Engneerng Fall 2015 Homework #3: Systems of Nonlnear Equatons and Optmzaton Problem 1 (30 ponts). A (homogeneous) azeotrope s a composton of a multcomponent mxture
10.34 Numercal Methods Appled to Chemcal Engneerng Fall 2015 Homework #3: Systems of Nonlnear Equatons and Optmzaton Problem 1 (30 ponts). A (homogeneous) azeotrope s a composton of a multcomponent mxture
The Euler Equation. Using the additive property of the internal energy U, we can derive a useful thermodynamic relation the Euler equation.
 The Euler Equation Using the additive property of the internal energy U, we can derive a useful thermodynamic relation the Euler equation. Let us differentiate this extensivity condition with respect to
The Euler Equation Using the additive property of the internal energy U, we can derive a useful thermodynamic relation the Euler equation. Let us differentiate this extensivity condition with respect to
