SUPPLEMENTARY INFORMATION
|
|
|
- Kristin Chase
- 6 years ago
- Views:
Transcription
1 DOI: 1.138/NGEO1825 Hydrogen generation from low temperature water-rock reactions L.E. Mayhew* 1, E.T. Ellison 1, T.M. McCollom 2, T.P. Trainor 3, and A.S. Templeton 1 *Corresponding author tel: (33) ; fax (33) ; mayhewl@colorado.edu 1 Department of Geological Sciences, UCB 399, University of Colorado - Boulder, Boulder, Colorado Laboratory for Atmospheric and Space Physics, UCB 392, University of Colorado - Boulder, Boulder, Colorado Department of Chemistry and Biochemistry, PO Box 75616, University of Alaska - Fairbanks, Fairbanks, Alaska pages (including cover page) 5 Figures 2 Tables NATURE GEOSCIENCE Macmillan Publishers Limited. All rights reserved.
2 DOI: 1.138/NGEO1825 Table S1. Major mineral constituents of the starting geologic substrates as identified by electron microprobe and/or XRD analyses. Chemical formulas were calculated from average normalized weight percent oxides of each phase as measured by electron microprobe. Starting material/phases Calculated mineral formula Forsythe mine fayalite fayalite fraction fayalite Fe 1.84 Mg.14 SiO 4 - Fa 88 magnetite Fe 3 O 4 clinopyroxene (Ca.92 Na.4 )(Mg 4 Fe.78 Al.4 )(Si 1.94 Al.6 )O 6 magnetite fraction magnetite Fe 3 O 4 fayalite Fe 1.84 Mg.14 SiO 4 clinopyroxene (Ca.92 Na.4 )(Mg 4 Fe.78 Al.4 )(Si 1.94 Al.6 )O 6 San Carlos olivine Mg 1.8 Fe SiO 4 - Fo 9 Hedenbergite CaFe Mn.1 Mg.1 Si 2 O 6 Petedunnite Ca.9 Zn.3 Mn.3 Fe.3 Mg Si 2 O 6 gahnite NA* San Carlos peridotite olivine Mg 1.8 Fe SiO 4 - Fo 9 clinopyroxene (Ca Na.1 )(Mg Fe.1 Al )(Si 1.9 Al.1 )O 6 orthopyroxene Ca.2 Mg 5 Fe.8 Si.97 Al.7 O 3 * phase was not analyzed by microprobe theoretical formula: ZnAl 2 O 4 Table S2. Geochemical parameters at the time* of sampling for synchrotron and XRD analyses. Values are averages of replicate vials. Experiment ph log fh 2 CO 2(aq) log fco 2 Aqueous species (µm) um Si Fe Mg Ca Fayalite fraction Magnetite fraction San Carlos olivine BDL Hedenbergite BDL Petedunnite BDL San Carlos peridotite BDL Media-only * San Carlos peridotite, fayalite, San Carlos olivine, and petedunnite were sampled at ~12 hours. Hedenbergite was sampled at ~23 hours. BDL below detection limit 2 NATURE GEOSCIENCE Macmillan Publishers Limited. All rights reserved.
3 DOI: 1.138/NGEO1825 SUPPLEMENTARY INFORMATION a. b. c. d. e. Figure S1. Mineral distribution maps of the unreacted starting substrates. (a.) San Carlos peridotite (red = pyroxene, green = olivine, blue = olivine + brucite). Scale bar = 1 um (b.) Petedunnite (red = Fe(III)-oxides, green = pyroxene, blue = gahnite). Scale bar = 1 um. (c.) Fayalite fraction (red = magnetite, green = fayalite, blue = pyroxene). Scale bar = 1 um. (d.) San Carlos olivine (red = San Carlos olivine > brucite, green = San Carlos olivine, blue = brucite > San Carlos olivine). Scale bar = 2 um. (e.) Hedenbergite (red = Fe(III)-(hydr)oxides, green = pyroxene, blue = altered pyroxene). Scale bar = 1 um. NATURE GEOSCIENCE Macmillan Publishers Limited. All rights reserved.
4 DOI: 1.138/NGEO1825 oxidized Fe a. b. pyroxene c. Fe(III) oxides Figure S2. (a.) Distribution of minerals in San Carlos olivine reacted at 1 C. (red = Fe(III)- (hydr)oxide, green = San Carlos olivine, blue = brucite > San Carlos olivine). (b.) Plane light photomicrograph of the mapped area. Scale bars = 2 um. (c.) Distribution of minerals in hedenbergite reacted at 1 C. (red = Fe(III)-(hydr)oxide, green = pyroxene, blue = Fe-bearing talc). Fe(III)-(hydr)oxides are not preferentially associated with any specific primary phase as seen in (d.) the plane light photomicrograph of the mapped area. d. 4 NATURE GEOSCIENCE Macmillan Publishers Limited. All rights reserved.
5 DOI: 1.138/NGEO1825 SUPPLEMENTARY INFORMATION XANES 1 XANES 11 XANES 16 XANES 12 XANES 6 XANES 1 XANES 24 XANES 9 fayalite unreacted fayalite reacted XANES 14 XANES XANES 24 XANES 16 XANES 13 XANES 3 XANES 7 XANES 6 XANES 2 XANES 3 San Carlos peridotite unreacted San Carlos peridotite reacted XANES 7 XANES 5 XANES 14 XANES 4 XANES 15 XANES 16 XANES 9 petedunnite unreacted petedunnite reacted Figure S3. Plots of representative XANES spectra from the geologic substrates both unreacted and reacted at 1ºC. NATURE GEOSCIENCE Macmillan Publishers Limited. All rights reserved.
6 DOI: 1.138/NGEO1825 XANES 3 XANES 1 XANES 9 San Carlos olivine unreacted XANES 26 XANES 24 XANES 3 XANES 1 San Carlos olivine reacted XANES 6 XANES 16 XANES 1 XANES 21 hedenbergite unreacted XANES 5 XANES 19 XANES 4 XANES 7 hedenbergite reacted Figure S3 continued. Plots of representative XANES spectra from the geologic substrates both unreacted and reacted at 1ºC. 6 NATURE GEOSCIENCE Macmillan Publishers Limited. All rights reserved.
7 DOI: 1.138/NGEO1825 SUPPLEMENTARY INFORMATION Fe-bearing brucite Fe-bearing talc ferrihydrite normalized intensity chromite magnetite hedenbergite San Carlos olivine Figure S4. Stack plot of spectra of key Fe-bearing model compunds. Vertical lines at 7127, 713, and 7133 ev show the peak position for models that are predominantely Fe(II)-bearing, mixed valent, and Fe(III)-bearing, respectively NATURE GEOSCIENCE Macmillan Publishers Limited. All rights reserved.
8 DOI: 1.138/NGEO1825 log a SiO 2 (aq) Fe ++ fayalite fraction magnetite fraction hedenbergite San Carlos peridotite Fe(II) bearing talc petedunnite San Carlos olivine Mg 2.7 Fe.3 Si 4 O 1 (OH) 2 1 C value at time of sampling for synchroton analysis for each substrate Fe(II) bearing pyroxene CaMg.95 Fe.5 Si 2 O ph Figure S5. Mineral stability diagram showing the thermodynamically stable Fe-bearing phases under average bulk aqueous and gaseous chemical conditions of the water-rock reaction systems at the time of sampling for synhcrotron analyses. The bulk aqueous chemistry at the time of sampling each experiment is plotted on the diagram. The uncertainity in the ph measurements was +/-.5. The model parameters were [Fe 2+ ]=1-7 M, [Ca 2+ ]=1-2.9 M, [Mg 2+ ]=1-2.4, log fh 2aq = and log fco 2 = NATURE GEOSCIENCE Macmillan Publishers Limited. All rights reserved.
LAB 6: COMMON MINERALS IN IGNEOUS ROCKS
 GEOLOGY 17.01: Mineralogy LAB 6: COMMON MINERALS IN IGNEOUS ROCKS Part 2: Minerals in Gabbroic Rocks Learning Objectives: Students will be able to identify the most common silicate minerals in gabbroic
GEOLOGY 17.01: Mineralogy LAB 6: COMMON MINERALS IN IGNEOUS ROCKS Part 2: Minerals in Gabbroic Rocks Learning Objectives: Students will be able to identify the most common silicate minerals in gabbroic
Supporting Information
 Supporting Information Bindi et al. 10.1073/pnas.1111115109 Fig. S1. Electron microprobe X-ray elemental maps for the grain reported in Fig. 1B. Experimental details are given in Experimental Methods.
Supporting Information Bindi et al. 10.1073/pnas.1111115109 Fig. S1. Electron microprobe X-ray elemental maps for the grain reported in Fig. 1B. Experimental details are given in Experimental Methods.
Common non-silicate planetary minerals
 Common non-silicate planetary minerals Many of the non-silicate minerals are simple oxides. Corundum Al2O3 Al2+3 O3-2 Rutile Ti2O3 Ti2+3 O3-2 Ilmenite FeTiO3 Fe+3Ti+3O3-2 Hematite Fe2O3 Fe2+3 O3-2 Families
Common non-silicate planetary minerals Many of the non-silicate minerals are simple oxides. Corundum Al2O3 Al2+3 O3-2 Rutile Ti2O3 Ti2+3 O3-2 Ilmenite FeTiO3 Fe+3Ti+3O3-2 Hematite Fe2O3 Fe2+3 O3-2 Families
Serpentinization, Abiogenic Methane, and Extremophilic Archaea within the Seafloor
 Serpentinization, Abiogenic Methane, and Extremophilic Archaea within the Seafloor Michael J. Mottl Department of Oceanography NAI Winter School, Jan. 2005 Earth s mantle is 68% of its mass and 83% of
Serpentinization, Abiogenic Methane, and Extremophilic Archaea within the Seafloor Michael J. Mottl Department of Oceanography NAI Winter School, Jan. 2005 Earth s mantle is 68% of its mass and 83% of
ENVIRONMENTAL FACTORS IMPACTING THE FORMATION AND KINETICS OF FE(II) LAYERED HYDROXIDES ON MINERALS AND SOILS. Autumn Nichole Starcher
 ENVIRONMENTAL FACTORS IMPACTING THE FORMATION AND KINETICS OF FE(II) LAYERED HYDROXIDES ON MINERALS AND SOILS by Autumn Nichole Starcher A dissertation submitted to the Faculty of the University of Delaware
ENVIRONMENTAL FACTORS IMPACTING THE FORMATION AND KINETICS OF FE(II) LAYERED HYDROXIDES ON MINERALS AND SOILS by Autumn Nichole Starcher A dissertation submitted to the Faculty of the University of Delaware
WORKING WITH ELECTRON MICROPROBE DATA FROM A HIGH PRESSURE EXPERIMENT CALCULATING MINERAL FORMULAS, UNIT CELL CONTENT, AND GEOTHERMOMETRY
 WORKING WITH ELECTRON MICROPROBE DATA FROM A HIGH PRESSURE EXPERIMENT CALCULATING MINERAL FORMULAS, UNIT CELL CONTENT, AND GEOTHERMOMETRY Brandon E. Schwab Department of Geology Humboldt State University
WORKING WITH ELECTRON MICROPROBE DATA FROM A HIGH PRESSURE EXPERIMENT CALCULATING MINERAL FORMULAS, UNIT CELL CONTENT, AND GEOTHERMOMETRY Brandon E. Schwab Department of Geology Humboldt State University
rinds for 8 Loihi rocks. Sample spectra, fits and model compounds are shown in Figure S5. All
 SUPPLEMENTARY INFORMATION Supplementary Table 1 Sample Location %Mats (M34, M39) 2L-FH (III)- smectite Vivianite Basalt glass red chi 2 J2-369-R2 Marker 17a 30% 21% - 11% 38% 0.06 J2-369-R5 Marker 17a
SUPPLEMENTARY INFORMATION Supplementary Table 1 Sample Location %Mats (M34, M39) 2L-FH (III)- smectite Vivianite Basalt glass red chi 2 J2-369-R2 Marker 17a 30% 21% - 11% 38% 0.06 J2-369-R5 Marker 17a
How many molecules? Pyrite FeS 2. Would there be any other elements in there???
 How many molecules? Pyrite FeS 2 Would there be any other elements in there??? Goldschmidt s rules of Substitution 1. The ions of one element can extensively replace those of another in ionic crystals
How many molecules? Pyrite FeS 2 Would there be any other elements in there??? Goldschmidt s rules of Substitution 1. The ions of one element can extensively replace those of another in ionic crystals
Geogenic versus Anthropogenic Metals and Metalloids
 Geogenic versus Anthropogenic Metals and Metalloids Geochemical methods for evaluating whether metals and metalloids are from geogenic versus anthropogenic sources 1 Definitions Geogenic from natural geological
Geogenic versus Anthropogenic Metals and Metalloids Geochemical methods for evaluating whether metals and metalloids are from geogenic versus anthropogenic sources 1 Definitions Geogenic from natural geological
Breeding et al., Data Repository Material Figure DR1. Athens. Study Area
 Breeding, Ague, and Brocker 1 Figure DR1 21 o 24 Greece o A 38 o Athens Tinos 37 o Syros Attic-Cycladic Blueschist Belt Syros Kampos B Study Area Ermoupoli N Vari Unit Cycladic HP-LT Unit Marble horizons
Breeding, Ague, and Brocker 1 Figure DR1 21 o 24 Greece o A 38 o Athens Tinos 37 o Syros Attic-Cycladic Blueschist Belt Syros Kampos B Study Area Ermoupoli N Vari Unit Cycladic HP-LT Unit Marble horizons
CO 2 sequestration via direct mineral carbonation of Mg-silicates. Natalie Johnson GCEP Symposium 4 October 2011
 CO 2 sequestration via direct mineral carbonation of Mg-silicates Natalie Johnson GCEP Symposium 4 October 2011 CO 2 release/year (Gt) 2 CCS: Part of climate change mitigation Projection based on current
CO 2 sequestration via direct mineral carbonation of Mg-silicates Natalie Johnson GCEP Symposium 4 October 2011 CO 2 release/year (Gt) 2 CCS: Part of climate change mitigation Projection based on current
LOMONOSOV MOSCOW STATE UNIVERSITY FACULTY OF GEOLOGY «ROCK-FORMING MINERALS OF OLIVINE GROUP»
 LOMONOSOV MOSCOW STATE UNIVERSITY FACULTY OF GEOLOGY «ROCK-FORMING MINERALS OF OLIVINE GROUP» A.B. Shefel GOALS AND OBJECTIVES Goal: Review of modern literature in order to summarize information about
LOMONOSOV MOSCOW STATE UNIVERSITY FACULTY OF GEOLOGY «ROCK-FORMING MINERALS OF OLIVINE GROUP» A.B. Shefel GOALS AND OBJECTIVES Goal: Review of modern literature in order to summarize information about
Supplementary Information
 Supplementary Information Supplementary Tables Major element compositions of host and daughter crystals: Table 1 reports composition for host-olivine. No daughter olivine has been found. The number for
Supplementary Information Supplementary Tables Major element compositions of host and daughter crystals: Table 1 reports composition for host-olivine. No daughter olivine has been found. The number for
Geos 306, Mineralogy Final Exam, Dec 12, pts
 Name: Geos 306, Mineralogy Final Exam, Dec 12, 2014 200 pts 1. (9 pts) What are the 4 most abundant elements found in the Earth and what are their atomic abundances? Create a reasonable hypothetical charge-balanced
Name: Geos 306, Mineralogy Final Exam, Dec 12, 2014 200 pts 1. (9 pts) What are the 4 most abundant elements found in the Earth and what are their atomic abundances? Create a reasonable hypothetical charge-balanced
muscovite PART 4 SHEET SILICATES
 muscovite PART 4 SHEET SILICATES SHEET SILICATES = PHYLLOSILICATES Phyllon = leaf Large group of mineral including many common minerals: muscovite, biotite, serpentine, chlorite, talc, clay minerals Structure:
muscovite PART 4 SHEET SILICATES SHEET SILICATES = PHYLLOSILICATES Phyllon = leaf Large group of mineral including many common minerals: muscovite, biotite, serpentine, chlorite, talc, clay minerals Structure:
12 Chemistry (Mg,Fe) 2 SiO 4 Olivine is forms what is called an isomorphous solid solution series that ranges between two end members: Forsterite Mg
 11 Olivine Structure Olivine is a common green or brown rock forming minerals which consists of a solid-solution series between Forsterite (Fo) and Fayalite (Fa). It is an orthorhombic orthosilicate with
11 Olivine Structure Olivine is a common green or brown rock forming minerals which consists of a solid-solution series between Forsterite (Fo) and Fayalite (Fa). It is an orthorhombic orthosilicate with
Accelerated reaction-driven carbon storage: Insights from laboratory-scale studies for field-scale geologic carbon storage
 Accelerated reaction-driven carbon storage: Insights from laboratory-scale studies for field-scale geologic carbon storage Greeshma Gadikota Department of Civil and Environmental Engineering Environmental
Accelerated reaction-driven carbon storage: Insights from laboratory-scale studies for field-scale geologic carbon storage Greeshma Gadikota Department of Civil and Environmental Engineering Environmental
Balancing Reaction Equations Oxidation State Reduction-oxidation Reactions. OCN 623 Chemical Oceanography
 Balancing Reaction Equations Oxidation State Reductionoxidation Reactions OCN 623 Chemical Oceanography Balanced chemical reactions are the math of chemistry They show the relationship between the reactants
Balancing Reaction Equations Oxidation State Reductionoxidation Reactions OCN 623 Chemical Oceanography Balanced chemical reactions are the math of chemistry They show the relationship between the reactants
Alkaline Seafloor Hydrothermal Systems: Experimental Simulation of CO 2 -Peridotite-Seawater Reactions
 Alkaline Seafloor Hydrothermal Systems: Experimental Simulation of CO 2 -Peridotite-Seawater Reactions Thomas M. Carpenter John P. Kaszuba Melissa Fittipaldo Michael Rearick Los Alamos National Laboratory
Alkaline Seafloor Hydrothermal Systems: Experimental Simulation of CO 2 -Peridotite-Seawater Reactions Thomas M. Carpenter John P. Kaszuba Melissa Fittipaldo Michael Rearick Los Alamos National Laboratory
Computational support for a pyrolitic lower mantle containing ferric iron
 SUPPLEMENTARY INFORMATION DOI: 1.138/NGEO2458 Computational support for a pyrolitic lower mantle containing ferric iron Xianlong Wang, Taku Tsuchiya and Atsushi Hase NATURE GEOSCIENCE www.nature.com/naturegeoscience
SUPPLEMENTARY INFORMATION DOI: 1.138/NGEO2458 Computational support for a pyrolitic lower mantle containing ferric iron Xianlong Wang, Taku Tsuchiya and Atsushi Hase NATURE GEOSCIENCE www.nature.com/naturegeoscience
Structures and Chemistry of silicate Silicates are classified on the basis of Si-O polymerism The culprit: the [SiO 4 ] 4 - tetrahedron
![Structures and Chemistry of silicate Silicates are classified on the basis of Si-O polymerism The culprit: the [SiO 4 ] 4 - tetrahedron Structures and Chemistry of silicate Silicates are classified on the basis of Si-O polymerism The culprit: the [SiO 4 ] 4 - tetrahedron](/thumbs/87/95554203.jpg) Structures and Chemistry of silicate by: Seyed mohsen hoseini zade Structures and Chemistry of silicate Silicates are classified on the basis of Si-O polymerism The culprit: the [SiO 4 ] 4 - tetrahedron
Structures and Chemistry of silicate by: Seyed mohsen hoseini zade Structures and Chemistry of silicate Silicates are classified on the basis of Si-O polymerism The culprit: the [SiO 4 ] 4 - tetrahedron
Redox transformation of arsenic. by Fe(II)-activated goethite (α-feooh)
 -SUPPORTING INFORMATION- Redox transformation of arsenic by Fe(II)-activated goethite (α-feooh) Katja Amstaetter 1,2, Thomas Borch 3, Philip Larese-Casanova 1, Andreas Kappler 1 * 1 Geomicrobiology, Center
-SUPPORTING INFORMATION- Redox transformation of arsenic by Fe(II)-activated goethite (α-feooh) Katja Amstaetter 1,2, Thomas Borch 3, Philip Larese-Casanova 1, Andreas Kappler 1 * 1 Geomicrobiology, Center
Redox reactions.
 Redox reactions http://eps.mcgill.ca/~courses/c220/ Redox reactions In a similar way that acids and bases have been defined as proton donors and proton acceptors, reductants and oxidants are defined as
Redox reactions http://eps.mcgill.ca/~courses/c220/ Redox reactions In a similar way that acids and bases have been defined as proton donors and proton acceptors, reductants and oxidants are defined as
SIC CONCEPTS TS OF CHEMISTRY. Unit. I. Multiple Choice Questions (Type-I)
 Unit 1 SOME BASIC B SIC CONCEPTS CONCEP TS OF CHEMISTRY CHEMIS I. Multiple Choice Questions (Type-I) 1. Two students performed the same experiment separately and each one of them recorded two readings
Unit 1 SOME BASIC B SIC CONCEPTS CONCEP TS OF CHEMISTRY CHEMIS I. Multiple Choice Questions (Type-I) 1. Two students performed the same experiment separately and each one of them recorded two readings
doi: /nature09369
 doi:10.1038/nature09369 Supplementary Figure S1 Scanning electron microscope images of experimental charges with vapour and vapour phase quench. Experimental runs are in the order of added water concentration
doi:10.1038/nature09369 Supplementary Figure S1 Scanning electron microscope images of experimental charges with vapour and vapour phase quench. Experimental runs are in the order of added water concentration
Environments of Mineral Formation. Stability Diagrams
 Environments of Mineral Formation Unary, Binary, and Ternary Mineral Stability Diagrams Minerals of differing composition (or polymorphs of the same mineral) that coexist at a set of pressure (P) temperature
Environments of Mineral Formation Unary, Binary, and Ternary Mineral Stability Diagrams Minerals of differing composition (or polymorphs of the same mineral) that coexist at a set of pressure (P) temperature
Q. WANG, Q-K. XIA, S. Y. O REILLY, W. L. GRIFFIN, E. E. BEYER AND H. K. BRUECKNER
 Pressure- and stress-induced fabric transition in olivine from peridotites in the Western Gneiss Region (Norway): implications for mantle seismic anisotropy Q. WANG, Q-K. XIA, S. Y. O REILLY, W. L. GRIFFIN,
Pressure- and stress-induced fabric transition in olivine from peridotites in the Western Gneiss Region (Norway): implications for mantle seismic anisotropy Q. WANG, Q-K. XIA, S. Y. O REILLY, W. L. GRIFFIN,
Earth and Planetary Materials
 Earth and Planetary Materials Spring 2013 Lecture 4 2013.01.16 Example Beryl Be 3 Al 2 (SiO 3 ) 6 Goshenite Aquamarine Emerald Heliodor Red beryl Morganite pure Fe 2+ & Fe 3+ Cr 3+ Fe 3+ Mn 3+ Mn 2+ Rules
Earth and Planetary Materials Spring 2013 Lecture 4 2013.01.16 Example Beryl Be 3 Al 2 (SiO 3 ) 6 Goshenite Aquamarine Emerald Heliodor Red beryl Morganite pure Fe 2+ & Fe 3+ Cr 3+ Fe 3+ Mn 3+ Mn 2+ Rules
Sedimentary Rocks and Processes
 Sedimentary Rocks and Processes Weathering Sedimentary Processes Breakdown of pre-existing rock by physical and chemical processes Transport Movement of sediments from environments of relatively high potential
Sedimentary Rocks and Processes Weathering Sedimentary Processes Breakdown of pre-existing rock by physical and chemical processes Transport Movement of sediments from environments of relatively high potential
Create custom rock (Rock1) and fluid (Fluid1) compositions. 1. Copy the folder Module3 to your project folder located in Library\Gems3\projects.
 MODULE 3: GREISEN ALTERATION In this tutorial we will use the GEMS project file Module3 in the examples to model the reaction path of a leucogranite during greisenization and evaluate the solubility of
MODULE 3: GREISEN ALTERATION In this tutorial we will use the GEMS project file Module3 in the examples to model the reaction path of a leucogranite during greisenization and evaluate the solubility of
Arsenic(V) incorporation in vivianite during microbial reduction of arsenic(v)-bearing biogenic Fe(III) (oxyhydr)oxides
 Arsenic(V) incorporation in vivianite during microbial reduction of arsenic(v)-bearing biogenic Fe(III) (oxyhydr)oxides E. Marie Muehe 1,2*, Guillaume Morin 3, Lukas Scheer 1, Pierre Le Pape 3, Imène Esteve
Arsenic(V) incorporation in vivianite during microbial reduction of arsenic(v)-bearing biogenic Fe(III) (oxyhydr)oxides E. Marie Muehe 1,2*, Guillaume Morin 3, Lukas Scheer 1, Pierre Le Pape 3, Imène Esteve
2. DATA REPORT: MICROPROBE ANALYSES (SERPENTINE, IOWAITE, TALC, MAGNETITE, OF PRIMARY PHASES (OLIVINE, PYROXENE, AND SPINEL) AND ALTERATION PRODUCTS
 Kelemen, P.., Kikawa, E., and Miller, D.J. (Eds.) Proceedings of the Ocean Drilling Program, Scientific Results Volume 9. DT REPORT: MICROPROE NLYSES OF PRIMRY PHSES (OLIVINE, PYROXENE, ND SPINEL) ND LTERTION
Kelemen, P.., Kikawa, E., and Miller, D.J. (Eds.) Proceedings of the Ocean Drilling Program, Scientific Results Volume 9. DT REPORT: MICROPROE NLYSES OF PRIMRY PHSES (OLIVINE, PYROXENE, ND SPINEL) ND LTERTION
Chromite and tourmaline chemical composition as a guide to mineral exploration
 Chromite and tourmaline chemical composition as a guide to mineral exploration Gabriel Voicu Université du Québec à Montréal, Canada and Cambior Management Services Content of presentation Part 1 Chromite
Chromite and tourmaline chemical composition as a guide to mineral exploration Gabriel Voicu Université du Québec à Montréal, Canada and Cambior Management Services Content of presentation Part 1 Chromite
Supplementary information. Jadeite in Chelyabinsk meteorite and the nature of an impact event on its parent. body
 Supplementary information Jadeite in Chelyabinsk meteorite and the nature of an impact event on its parent body Shin Ozawa 1*, Masaaki Miyahara 1, 2, Eiji Ohtani 1, 3, Olga N. Koroleva 4, Yoshinori Ito
Supplementary information Jadeite in Chelyabinsk meteorite and the nature of an impact event on its parent body Shin Ozawa 1*, Masaaki Miyahara 1, 2, Eiji Ohtani 1, 3, Olga N. Koroleva 4, Yoshinori Ito
Activity Balancing Oxidation-Reduction Reactions
 Activity 201 3 Balancing Oxidation-Reduction Reactions Directions: This GLA worksheet goes over the half-reaction method of balancing oxidation-reduction (redox) reactions. Part A introduces the oxidation
Activity 201 3 Balancing Oxidation-Reduction Reactions Directions: This GLA worksheet goes over the half-reaction method of balancing oxidation-reduction (redox) reactions. Part A introduces the oxidation
Mineralogy and petrology of two ordinary chondrites and their correlation with other meteorites
 MINERALOGIA, 40, No. 1 4: 107 116 (2009) DOI: 10.2478/v10002-009-0009-9 www.mineralogia.pl MINERALOGICAL SOCIETY OF POLAND POLSKIE TOWARZYSTWO MINERALOGICZNE Short note Mineralogy and petrology of two
MINERALOGIA, 40, No. 1 4: 107 116 (2009) DOI: 10.2478/v10002-009-0009-9 www.mineralogia.pl MINERALOGICAL SOCIETY OF POLAND POLSKIE TOWARZYSTWO MINERALOGICZNE Short note Mineralogy and petrology of two
(DO NOT WRITE ON THIS TEST)
 Final Prep Chap 8&9 (DO NOT WRITE ON THIS TEST) Multiple Choice Identify the choice that best completes the statement or answers the question. 1. After the correct formula for a reactant in an equation
Final Prep Chap 8&9 (DO NOT WRITE ON THIS TEST) Multiple Choice Identify the choice that best completes the statement or answers the question. 1. After the correct formula for a reactant in an equation
Thermodynamic constraints on fayalite formation on parent bodies of chondrites
 Meteoritics & Planetary Science 41, Nr 11, 1775 1796 (2006) Abstract available online at http://meteoritics.org Thermodynamic constraints on fayalite formation on parent bodies of chondrites Mikhail Yu.
Meteoritics & Planetary Science 41, Nr 11, 1775 1796 (2006) Abstract available online at http://meteoritics.org Thermodynamic constraints on fayalite formation on parent bodies of chondrites Mikhail Yu.
Class XI Chapter 1 Some Basic Concepts of Chemistry Chemistry
 Question 1.1: Calculate the molecular mass of the following: (i) H 2 O (ii) CO 2 (iii) CH 4 (i) H 2 O: The molecular mass of water, H 2 O = (2 Atomic mass of hydrogen) + (1 Atomic mass of oxygen) = [2(1.0084)
Question 1.1: Calculate the molecular mass of the following: (i) H 2 O (ii) CO 2 (iii) CH 4 (i) H 2 O: The molecular mass of water, H 2 O = (2 Atomic mass of hydrogen) + (1 Atomic mass of oxygen) = [2(1.0084)
GSA DATA REPOSITORY
 GSA DATA REPOSITORY 2013019 Supplemental information for The Solidus of Alkaline Carbonatite in the Deep Mantle Konstantin D. Litasov, Anton Shatskiy, Eiji Ohtani, and Gregory M. Yaxley EXPERIMENTAL METHODS
GSA DATA REPOSITORY 2013019 Supplemental information for The Solidus of Alkaline Carbonatite in the Deep Mantle Konstantin D. Litasov, Anton Shatskiy, Eiji Ohtani, and Gregory M. Yaxley EXPERIMENTAL METHODS
 Question 1.1: Calculate the molecular mass of the following: (i) H 2 O (ii) CO 2 (iii) CH 4 (i) H 2 O: The molecular mass of water, H 2 O = (2 Atomic mass of hydrogen) + (1 Atomic mass of oxygen) = [2(1.0084)
Question 1.1: Calculate the molecular mass of the following: (i) H 2 O (ii) CO 2 (iii) CH 4 (i) H 2 O: The molecular mass of water, H 2 O = (2 Atomic mass of hydrogen) + (1 Atomic mass of oxygen) = [2(1.0084)
The stability of arc lower crust: Insights from the Talkeetna Arc section, south-central Alaska and the seismic structure of modern arcs
 The stability of arc lower crust: Insights from the Talkeetna Arc section, south-central Alaska and the seismic structure of modern arcs Mark D. Behn,* and Peter B. Kelemen Department of Geology and Geophysics,
The stability of arc lower crust: Insights from the Talkeetna Arc section, south-central Alaska and the seismic structure of modern arcs Mark D. Behn,* and Peter B. Kelemen Department of Geology and Geophysics,
Solapur University, Solapur. Syllabus for B.Sc. II- Geochemistry - (IDS) Semester System - CGPA To be implemented from Academic Year
 Solapur University, Solapur Syllabus for B.Sc. II- Geochemistry - (IDS) Semester System - CGPA To be implemented from Academic Year- 2015-16 Course Structure Total Credit 16 - (Theory (4 x 3) = 12+Practical
Solapur University, Solapur Syllabus for B.Sc. II- Geochemistry - (IDS) Semester System - CGPA To be implemented from Academic Year- 2015-16 Course Structure Total Credit 16 - (Theory (4 x 3) = 12+Practical
Lecture 6. Physical Properties. Solid Phase. Particle Composition
 Lecture 6 Physical Properties Solid Phase Particle Composition 1 Questions What are tetrahedrons and octahedrons? How do silica tetrahedra bonds affect mineral weathering? Difference between primary and
Lecture 6 Physical Properties Solid Phase Particle Composition 1 Questions What are tetrahedrons and octahedrons? How do silica tetrahedra bonds affect mineral weathering? Difference between primary and
Mole: base unit for an amount of substance A mole contains Avogadro s number (N A ) of particles (atoms, molecules, ions, formula units )
 Mole: base unit for an amount of substance A mole contains Avogadro s number (N A ) of particles (atoms, molecules, ions, formula units ) N A 6.0 10 mol -1 1 mol substance contains N A Molar mass (g/mol)
Mole: base unit for an amount of substance A mole contains Avogadro s number (N A ) of particles (atoms, molecules, ions, formula units ) N A 6.0 10 mol -1 1 mol substance contains N A Molar mass (g/mol)
DIFFERENTIATION OF MAGMAS BY FRACTIONAL CRYSTALLIZATION THE M&M MAGMA CHAMBER
 Geol 2312 Igneous and Metamorphic Petrology Spring 2009 Name DIFFERENTIATION OF MAGMAS BY FRACTIONAL CRYSTALLIZATION THE M&M MAGMA CHAMBER Objective: This exercise is intended to improve understanding
Geol 2312 Igneous and Metamorphic Petrology Spring 2009 Name DIFFERENTIATION OF MAGMAS BY FRACTIONAL CRYSTALLIZATION THE M&M MAGMA CHAMBER Objective: This exercise is intended to improve understanding
VOCABULARY Define. 1. reactants. 2. products. 3. chemical equation. 4. precipitate. 5. word equation
 CHAPTER 8 HOMEWORK 8-1 (pp. 241 245) Define. 1. reactants 2. products 3. chemical equation 4. precipitate 5. word equation GRAPHIC ORGANIZER Complete the chart by listing three signs that suggest that
CHAPTER 8 HOMEWORK 8-1 (pp. 241 245) Define. 1. reactants 2. products 3. chemical equation 4. precipitate 5. word equation GRAPHIC ORGANIZER Complete the chart by listing three signs that suggest that
Predicting Mineral Transformations in Wet Supercritical CO 2 : The Critical Role of Water
 Predicting Mineral Transformations in Wet Supercritical CO 2 : The Critical Role of Water Andrew R. Felmy Eugene S. Ilton Andre Anderko Kevin M. Rosso Ja Hun Kwak Jian Zhi Hu 1 Outline Background Importance
Predicting Mineral Transformations in Wet Supercritical CO 2 : The Critical Role of Water Andrew R. Felmy Eugene S. Ilton Andre Anderko Kevin M. Rosso Ja Hun Kwak Jian Zhi Hu 1 Outline Background Importance
 Unit 1 SOME BASIC CONCEPTS OF CHEMISTRY I. Multiple Choice Questions (Type-I) 1. Two students performed the same experiment separately and each one of them recorded two readings of mass which are given
Unit 1 SOME BASIC CONCEPTS OF CHEMISTRY I. Multiple Choice Questions (Type-I) 1. Two students performed the same experiment separately and each one of them recorded two readings of mass which are given
Silicates. The most common group of minerals forming the silicate Earth
 Silicates The most common group of minerals forming the silicate Earth 25% of all minerals (~1000) 40% of rock forming minerals 90% of earth s crust i.e those minerals you are likely to find ~100 of earth
Silicates The most common group of minerals forming the silicate Earth 25% of all minerals (~1000) 40% of rock forming minerals 90% of earth s crust i.e those minerals you are likely to find ~100 of earth
Limiting Reactants. and Percentage Yield. Section 3
 GO ONLINE Section 3 8E Main Ideas One reactant limits the product of a reaction. Comparing the actual and theoretical yields helps chemists determine the reaction s efficiency. 8E perform stoichiometric
GO ONLINE Section 3 8E Main Ideas One reactant limits the product of a reaction. Comparing the actual and theoretical yields helps chemists determine the reaction s efficiency. 8E perform stoichiometric
Suzanne Picazo, Mathilde Cannat, Adelie Delacour, Javier Escartın, Stephane Roumejon, and Serguei Silantyev
 Correction Volume 14, Number 8 16 August 2013 doi: ISSN: 1525-2027 Correction to Deformation associated with the denudation of mantle-derived rocks at the Mid- Atlantic Ridge 13 15 N: The role of magmatic
Correction Volume 14, Number 8 16 August 2013 doi: ISSN: 1525-2027 Correction to Deformation associated with the denudation of mantle-derived rocks at the Mid- Atlantic Ridge 13 15 N: The role of magmatic
Hydration of Olivine and. Earth s Deep Water Cycle
 Hydration of Olivine and Earth s Deep Water Cycle IASPEI October 5, 2005 Olivine Hydration and Earth s Deep Water Cycle J. R. Smyth, (University of Colorado) Dan Frost and Fabrizio Nestola (Bayerisches
Hydration of Olivine and Earth s Deep Water Cycle IASPEI October 5, 2005 Olivine Hydration and Earth s Deep Water Cycle J. R. Smyth, (University of Colorado) Dan Frost and Fabrizio Nestola (Bayerisches
CHEMISTRY 13 Electrochemistry Supplementary Problems
 1. When the redox equation CHEMISTRY 13 Electrochemistry Supplementary Problems MnO 4 (aq) + H + (aq) + H 3 AsO 3 (aq) Mn 2+ (aq) + H 3 AsO 4 (aq) + H 2 O(l) is properly balanced, the coefficients will
1. When the redox equation CHEMISTRY 13 Electrochemistry Supplementary Problems MnO 4 (aq) + H + (aq) + H 3 AsO 3 (aq) Mn 2+ (aq) + H 3 AsO 4 (aq) + H 2 O(l) is properly balanced, the coefficients will
Chapter 3 The Mole and Stoichiometry
 Chapter 3 The Mole and Stoichiometry Chemistry, 7 th Edition International Student Version Brady/Jespersen/Hyslop Brady/Jespersen/Hyslop Chemistry7E, Copyright 015 John Wiley & Sons, Inc. All Rights Reserved
Chapter 3 The Mole and Stoichiometry Chemistry, 7 th Edition International Student Version Brady/Jespersen/Hyslop Brady/Jespersen/Hyslop Chemistry7E, Copyright 015 John Wiley & Sons, Inc. All Rights Reserved
Dunite A cost effective raw material in basic refractory mixes for steel making. 24 th September 2009
 Dunite A cost effective raw material in basic refractory mixes for steel making 24 th September 2009 Index Continuous casting in steel making Typical composition of tundish basic mixes Dunite PM Design
Dunite A cost effective raw material in basic refractory mixes for steel making 24 th September 2009 Index Continuous casting in steel making Typical composition of tundish basic mixes Dunite PM Design
Earth Materials I Crystal Structures
 Earth Materials I Crystal Structures Isotopes same atomic number, different numbers of neutrons, different atomic mass. Ta ble 1-1. Su mmar y of quantu m num bers Name Symbol Values Principal n 1, 2,
Earth Materials I Crystal Structures Isotopes same atomic number, different numbers of neutrons, different atomic mass. Ta ble 1-1. Su mmar y of quantu m num bers Name Symbol Values Principal n 1, 2,
it must be it must be it must have been formed by it must have it must have
 6. Minerals II (p. 78-87) What is a mineral? The five characteristics required in order for a compound to be a mineral are: it must be it must be it must have been formed by it must have it must have Characteristics
6. Minerals II (p. 78-87) What is a mineral? The five characteristics required in order for a compound to be a mineral are: it must be it must be it must have been formed by it must have it must have Characteristics
Tailings and Mineral Carbonation: The Potential for Atmospheric CO 2 Sequestration
 Tailings and Mineral Carbonation: The Potential for Atmospheric CO 2 Sequestration H. Andrew Rollo Lorax Environmental Services Ltd. Heather. E. Jamieson Department of Geological Sciences and Geological
Tailings and Mineral Carbonation: The Potential for Atmospheric CO 2 Sequestration H. Andrew Rollo Lorax Environmental Services Ltd. Heather. E. Jamieson Department of Geological Sciences and Geological
Average Composition of the Continental Crust. Table 3.4
 Minerlaogi II Average Composition of the Continental Crust Si O Weight Percent O Volume Percent Table 3.4 Ionic Radii of some geologically important ions Fig. 3.8 Silika Tetraederet Silicates are classified
Minerlaogi II Average Composition of the Continental Crust Si O Weight Percent O Volume Percent Table 3.4 Ionic Radii of some geologically important ions Fig. 3.8 Silika Tetraederet Silicates are classified
1 Some Basic Concepts of Chemistry
 1 Some Basic Concepts of Chemistry Multiple Choice Questions (MCQs) Q. 1 Two students performed the same experiment separately and each one of them recorded two readings of mass which are given below.
1 Some Basic Concepts of Chemistry Multiple Choice Questions (MCQs) Q. 1 Two students performed the same experiment separately and each one of them recorded two readings of mass which are given below.
10/8/15. Earth Materials Minerals and Rocks. I) Minerals. Minerals. (A) Definition: Topics: -- naturally occurring What are minerals?
 minerals Earth Materials Minerals and Rocks I) Minerals Minerals Topics: What are minerals? Basic Chemistry Amethysts in geode: minerals Characteristics of Minerals Types of Minerals -- orderly arrangement
minerals Earth Materials Minerals and Rocks I) Minerals Minerals Topics: What are minerals? Basic Chemistry Amethysts in geode: minerals Characteristics of Minerals Types of Minerals -- orderly arrangement
Chapter 16 Weathering, Erosion, Mass Wasting. Chapter 16 Weathering, Erosion, Mass Wasting. Mechanical Weathering
 Weathering, Erosion and Mass Wasting Weathering is the the breakdown of solid rock at or near the Earth's surface. Chapter 16 Weathering, Erosion, Mass Wasting Does weathering of rock remove or add CO
Weathering, Erosion and Mass Wasting Weathering is the the breakdown of solid rock at or near the Earth's surface. Chapter 16 Weathering, Erosion, Mass Wasting Does weathering of rock remove or add CO
Advances in Environmental Biology
 AENSI Journals Advances in Environmental Biology ISSN-1995-0756 EISSN-1998-1066 Journal home page: http://www.aensiweb.com/aeb/ Pplication of Principal Analysis Component and Mobility of Heavy Metals in
AENSI Journals Advances in Environmental Biology ISSN-1995-0756 EISSN-1998-1066 Journal home page: http://www.aensiweb.com/aeb/ Pplication of Principal Analysis Component and Mobility of Heavy Metals in
Chemistry 2000 Lecture 14: Redox reactions
 Chemistry 2000 Lecture 14: Redox reactions Marc R. Roussel February 8, 2018 Marc R. Roussel Chemistry 2000 Lecture 14: Redox reactions February 8, 2018 1 / 12 Review: Oxidation states The oxidation state
Chemistry 2000 Lecture 14: Redox reactions Marc R. Roussel February 8, 2018 Marc R. Roussel Chemistry 2000 Lecture 14: Redox reactions February 8, 2018 1 / 12 Review: Oxidation states The oxidation state
Figure S1. CRISM maps of modeled mineralogy projected over CTX imagery (same
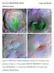 GSA DATA REPOSITORY 2015222 Cannon and Mustard Additional examples Figure S1. CRISM maps of modeled mineralogy projected over CTX imagery (same parameters as Figure 1). A: Taytay Crater (CRISM ID: HRL00005B77).
GSA DATA REPOSITORY 2015222 Cannon and Mustard Additional examples Figure S1. CRISM maps of modeled mineralogy projected over CTX imagery (same parameters as Figure 1). A: Taytay Crater (CRISM ID: HRL00005B77).
Cation Exchange Capacity, CEC
 Cation Exchange Capacity, CEC The basic building blocks of clay minerals are: silicon atoms surrounded by four oxygen atoms (tetrahedra), and aluminium atoms surrounded by six hydroxide groups (dioctahedra),
Cation Exchange Capacity, CEC The basic building blocks of clay minerals are: silicon atoms surrounded by four oxygen atoms (tetrahedra), and aluminium atoms surrounded by six hydroxide groups (dioctahedra),
F Ccp = (Cu wr )/ (Cu Ccp ) (1) In the first iteration all of the Ni was assigned to pentlandite. F Pn = (Ni wr )/ (Ni Pn ) (2)
 Online resource C: Method to determine the proportion (in wt.%) of each element hosted by pyrrhotite, pentlandite, chalcopyrite, pyrite and the precious metal minerals (PMM) used by Dare et al. in Chalcophile
Online resource C: Method to determine the proportion (in wt.%) of each element hosted by pyrrhotite, pentlandite, chalcopyrite, pyrite and the precious metal minerals (PMM) used by Dare et al. in Chalcophile
Topic 5 : Crystal chemistry
 GEOL360 LECTURE NOTES: T5 : CRYSTAL CHEMISTRY 1/15 GEOL360 Topic 5 : Crystal chemistry 5.1 Introduction what is a crystal? A crystal is a homogeneous, solid body of a chemical element, compound, or isomorphous
GEOL360 LECTURE NOTES: T5 : CRYSTAL CHEMISTRY 1/15 GEOL360 Topic 5 : Crystal chemistry 5.1 Introduction what is a crystal? A crystal is a homogeneous, solid body of a chemical element, compound, or isomorphous
*Corresponding author. Phone, x2920; Fax +34. Vicente del Raspeig, Alicante, Spain
 Green synthesis of thin shell carbon-encapsulated iron nanoparticles via hydrothermal carbonization B. Calderon a,*, F. Smith a, I. Aracil a, A. Fullana a a Chemical Engineering Department, University
Green synthesis of thin shell carbon-encapsulated iron nanoparticles via hydrothermal carbonization B. Calderon a,*, F. Smith a, I. Aracil a, A. Fullana a a Chemical Engineering Department, University
Topic 5.2 PERIODICITY. The oxides of period 3 elements The reaction of period 3 elements with water
 Topic 5.2 PERIODICITY The oxides of period 3 elements The reaction of period 3 elements with water 1. Formation of oxides THE OXIDES OF PERIOD 3 ELEMENTS All the elements in Period 3 except chlorine and
Topic 5.2 PERIODICITY The oxides of period 3 elements The reaction of period 3 elements with water 1. Formation of oxides THE OXIDES OF PERIOD 3 ELEMENTS All the elements in Period 3 except chlorine and
67. PHASE CHEMISTRY STUDIES ON GABBRO AND PERIDOTITE ROCKS FROM SITE 334, DSDP LEG 37
 67. PHASE CHEMISTRY STUDIES N GABBR AND PERIDTITE RCKS FRM SITE 334, DSDP LEG 37 R.F. Symes, J.C. Bevan, and R. Hutchison, Department of Mineralogy, British Museum (Natural History, London, England INTRDUCTIN
67. PHASE CHEMISTRY STUDIES N GABBR AND PERIDTITE RCKS FRM SITE 334, DSDP LEG 37 R.F. Symes, J.C. Bevan, and R. Hutchison, Department of Mineralogy, British Museum (Natural History, London, England INTRDUCTIN
MOSELEY and MODERN PERIODIC TABLE (designed by atomic numbers of elements)
 MOSELEY and MODERN PERIODIC TABLE (designed by atomic numbers of elements) 1 PERIODS: Period number = Number of basic energy levels = The principal quantum number The horizontal lines in the periodic system
MOSELEY and MODERN PERIODIC TABLE (designed by atomic numbers of elements) 1 PERIODS: Period number = Number of basic energy levels = The principal quantum number The horizontal lines in the periodic system
Notes for Use of the Cpx-Plag-Ol Thermobar Workbook Last Updated:
 Notes for Use of the Cpx-Plag-Ol Thermobar Workbook Last Updated: 7-22-05 Cpx-Plag-Ol Thermobar is an Excel workbook that can be used to calculate crystallization pressures and temperatures for clinopyroxene-
Notes for Use of the Cpx-Plag-Ol Thermobar Workbook Last Updated: 7-22-05 Cpx-Plag-Ol Thermobar is an Excel workbook that can be used to calculate crystallization pressures and temperatures for clinopyroxene-
Magma Evolution Activity. Magma Evolution Activity. Magma Evolution Activity. Magma Evolution Activity
 Evolution Activity You have grams of magma atoms (oxygen, the most abundant, is not included) Each poker chip is gram of a specific atom. Separate chips into colored stacks. What is the highest percentage
Evolution Activity You have grams of magma atoms (oxygen, the most abundant, is not included) Each poker chip is gram of a specific atom. Separate chips into colored stacks. What is the highest percentage
4. What is the law of constant composition (also known as the law of definite proportion)?
 Name: Exercises #1: 1. What is the law of conservation of mass? 2. Show that the results of the following experiments illustrate the law of conservation of mass. Experiment #1: a 5.00-g sample of pure
Name: Exercises #1: 1. What is the law of conservation of mass? 2. Show that the results of the following experiments illustrate the law of conservation of mass. Experiment #1: a 5.00-g sample of pure
BUREAU OF MINERAL RESOURCES, GEOLOGY AND GEOPHYS-ICS
 BUREAU OF MINERAL RESOURCES, GEOLOGY AND GEOPHYS-ICS RECORD 1980/53 MINERAL ANALYSES AND ROCK DESCRIPTIONS FOR CUMULATE PERIDOTITES AND GABBROS FROM THE MARUM OPHIOLITE COMPLEX, NORTHERN PAPUA NE\v GUINEA
BUREAU OF MINERAL RESOURCES, GEOLOGY AND GEOPHYS-ICS RECORD 1980/53 MINERAL ANALYSES AND ROCK DESCRIPTIONS FOR CUMULATE PERIDOTITES AND GABBROS FROM THE MARUM OPHIOLITE COMPLEX, NORTHERN PAPUA NE\v GUINEA
amphibole PART 3 Pyroxene: augite CHAIN SILICATES
 amphibole PART 3 Pyroxene: augite CHAIN SILICATES CHAIN SILICATES = INOSILICATES inos = chains Basic structural group: Si 2 O 6 (each tetrahedra shared two corners) Simple or double chains linked by cations
amphibole PART 3 Pyroxene: augite CHAIN SILICATES CHAIN SILICATES = INOSILICATES inos = chains Basic structural group: Si 2 O 6 (each tetrahedra shared two corners) Simple or double chains linked by cations
Introductory Inorganic Chemistry
 Introductory Inorganic Chemistry What is Inorganic Chemistry? As: 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 3 Classes of Inorganic Substances Elements Ionic Compounds Covalent Compounds Atomic/Molecular
Introductory Inorganic Chemistry What is Inorganic Chemistry? As: 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 3 Classes of Inorganic Substances Elements Ionic Compounds Covalent Compounds Atomic/Molecular
26. N 2 + H 2 NH N 2 + O 2 N 2 O 28. CO 2 + H 2 O C 6 H 12 O 6 + O SiCl 4 + H 2 O H 4 SiO 4 + HCl 30. H 3 PO 4 H 4 P 2 O 7 + H 2 O
 Balance the following chemical equations: (Some may already be balanced.) 1. H 2 + O 2 H 2 O 2. S 8 + O 2 SO 3 3. HgO Hg + O 2 4. Zn + HCl ZnCl 2 + H 2 5. Na + H 2 O NaOH + H 2 6. C 10 H 16 + Cl 2 C +
Balance the following chemical equations: (Some may already be balanced.) 1. H 2 + O 2 H 2 O 2. S 8 + O 2 SO 3 3. HgO Hg + O 2 4. Zn + HCl ZnCl 2 + H 2 5. Na + H 2 O NaOH + H 2 6. C 10 H 16 + Cl 2 C +
Supplementary Figure 1 HAADF-STEM images of 0.08%Pt/FeO x -R200 single-atom catalyst with different magnifications. Scale bar: a, 20 nm; b, 10 nm; c,
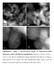 Supplementary Figure 1 HAADF-STEM images of 0.08%Pt/FeO x -R200 single-atom catalyst with different magnifications. Scale bar: a, 20 nm; b, 10 nm; c, 2 nm; d, 2 nm. The low magnification images demonstrate
Supplementary Figure 1 HAADF-STEM images of 0.08%Pt/FeO x -R200 single-atom catalyst with different magnifications. Scale bar: a, 20 nm; b, 10 nm; c, 2 nm; d, 2 nm. The low magnification images demonstrate
Unit IV: Stoichiometry
 Unit IV: Stoichiometry D. Calculations from chemical equations 1. Mol-mol calculations 2. Mol-gram calculations 3. Gram-gram calculations E. Limiting reactant (or reagent) F. Other applications of stoichiometry
Unit IV: Stoichiometry D. Calculations from chemical equations 1. Mol-mol calculations 2. Mol-gram calculations 3. Gram-gram calculations E. Limiting reactant (or reagent) F. Other applications of stoichiometry
1. [7 points] How many significant figures should there be in the answer to the following problem?
![1. [7 points] How many significant figures should there be in the answer to the following problem? 1. [7 points] How many significant figures should there be in the answer to the following problem?](/thumbs/89/97819589.jpg) 1 of 6 10/20/2009 3:54 AM Avogadro s Number = 6.022 10 23 1. [7 points] How many significant figures should there be in the answer to the following problem? (29.0025 + 0.2)/(6.1345 36.101) (a) 1 (b) 2
1 of 6 10/20/2009 3:54 AM Avogadro s Number = 6.022 10 23 1. [7 points] How many significant figures should there be in the answer to the following problem? (29.0025 + 0.2)/(6.1345 36.101) (a) 1 (b) 2
16+ ENTRANCE EXAMINATION
 ST EDWARD S OXFORD 16+ ENTRANCE EXAMINATION For entry in September 2015 CHEMISTRY Time: 1 hour Candidates Name: St Edward's School 1 1. Complete the table below. St Edward's School 2 Element calcium Symbol
ST EDWARD S OXFORD 16+ ENTRANCE EXAMINATION For entry in September 2015 CHEMISTRY Time: 1 hour Candidates Name: St Edward's School 1 1. Complete the table below. St Edward's School 2 Element calcium Symbol
Oxidation and Reduction in Photochromism. Oxidation and Reduction. Examples of Oxidized and Reduced Substances. Electron Loss and Gain.
 Chapter 15 Lecture Chapter 15 15.1 Oxidation Reduction Reactions Oxidation and Reduction Fifth Edition Oxidation and Reduction An oxidation-reduction reaction provides us with energy from food provides
Chapter 15 Lecture Chapter 15 15.1 Oxidation Reduction Reactions Oxidation and Reduction Fifth Edition Oxidation and Reduction An oxidation-reduction reaction provides us with energy from food provides
Chem A Ch. 9 Practice Test
 Name: Class: Date: Chem A Ch. 9 Practice Test Matching Match each item with the correct statement below. a. product d. balanced equation b. reactant e. skeleton equation c. chemical equation 1. a new substance
Name: Class: Date: Chem A Ch. 9 Practice Test Matching Match each item with the correct statement below. a. product d. balanced equation b. reactant e. skeleton equation c. chemical equation 1. a new substance
CHEM 1423 Chapter 21 Homework Questions TEXTBOOK HOMEWORK
 CHEM 1423 Chapter 21 Homework Questions TEXTBOOK HOMEWORK 21.5 Consider the following balanced redox reaction: 16 H + (aq) + 2 MnO 4- (aq) + 10 Cl - (aq) 2 Mn 2+ (aq) + 5 Cl 2 (g) + 8 H 2 O(l) (a) Which
CHEM 1423 Chapter 21 Homework Questions TEXTBOOK HOMEWORK 21.5 Consider the following balanced redox reaction: 16 H + (aq) + 2 MnO 4- (aq) + 10 Cl - (aq) 2 Mn 2+ (aq) + 5 Cl 2 (g) + 8 H 2 O(l) (a) Which
1 - C Systems. The system H 2 O. Heat an ice at 1 atm from-5 to 120 o C. Heat vs. Temperature
 1 - C Systems The system H 2 O Heat an ice at 1 atm from-5 to 120 o C Heat vs. Temperature Fig. 6.7. After Bridgman (1911) Proc. Amer. Acad. Arts and Sci., 5, 441-513; (1936) J. Chem. Phys., 3, 597-605;
1 - C Systems The system H 2 O Heat an ice at 1 atm from-5 to 120 o C Heat vs. Temperature Fig. 6.7. After Bridgman (1911) Proc. Amer. Acad. Arts and Sci., 5, 441-513; (1936) J. Chem. Phys., 3, 597-605;
Chapter 6: The Phase Rule and One and Two-Component Systems aka Phase Equilibria
 Chapter 6: The Phase Rule and One and Two-Component Systems aka Phase Equilibria Makaopuhi Lava Lake Magma samples recovered from various depths beneath solid crust From Wright and Okamura, (1977) USGS
Chapter 6: The Phase Rule and One and Two-Component Systems aka Phase Equilibria Makaopuhi Lava Lake Magma samples recovered from various depths beneath solid crust From Wright and Okamura, (1977) USGS
ANALYSIS OF GEOLOGIC MATERIALS USING RIETVELD QUANTIATIVE X-RAY DIFFRACTION
 Copyright JCPDS - International Centre for Diffraction Data 2003, Advances in X-ray Analysis, Volume 46. 204 ANALYSIS OF GEOLOGIC MATERIALS USING RIETVELD QUANTIATIVE X-RAY DIFFRACTION Robin M. Gonzalez,
Copyright JCPDS - International Centre for Diffraction Data 2003, Advances in X-ray Analysis, Volume 46. 204 ANALYSIS OF GEOLOGIC MATERIALS USING RIETVELD QUANTIATIVE X-RAY DIFFRACTION Robin M. Gonzalez,
Name: Date: M O L A R M A S S & P E R C E N T C O M P O S I T I O N
 Name: Date: M O L A R M A S S & P E R C E N T C O M P O S I T I O N I. Molar Masses Given a periodic table, you should be able to calculate the molecular mass (in amu s) or the molar mass (in grams) for
Name: Date: M O L A R M A S S & P E R C E N T C O M P O S I T I O N I. Molar Masses Given a periodic table, you should be able to calculate the molecular mass (in amu s) or the molar mass (in grams) for
Oxidation-Reduction (Redox) Reactions
 Oxidation-Reduction (Redox) Reactions Def n: Reactions in which one or more electrons is shifted from one element to another (In acid/base, gas transfer, and precipitation reactions discussed previously,
Oxidation-Reduction (Redox) Reactions Def n: Reactions in which one or more electrons is shifted from one element to another (In acid/base, gas transfer, and precipitation reactions discussed previously,
Water-rock interaction ascribed to hyperalkaline mineral waters in the Cabeço de Vide serpentinized ultramafic intrusive massif (Central Portugal)
 Water-rock interaction ascribed to hyperalkaline mineral waters in the Cabeço de Vide serpentinized ultramafic intrusive massif (Central Portugal) José M. Marques a, Maria O. Neves a, Ana Z. Miller a,
Water-rock interaction ascribed to hyperalkaline mineral waters in the Cabeço de Vide serpentinized ultramafic intrusive massif (Central Portugal) José M. Marques a, Maria O. Neves a, Ana Z. Miller a,
Chapter 9: Trace Elements
 Lecture 13 Introduction to Trace Elements Wednesday, March 9, 2005 Chapter 9: Trace Elements Note magnitude of major element changes Figure 8-2. Harker variation diagram for 310 analyzed volcanic rocks
Lecture 13 Introduction to Trace Elements Wednesday, March 9, 2005 Chapter 9: Trace Elements Note magnitude of major element changes Figure 8-2. Harker variation diagram for 310 analyzed volcanic rocks
predictive mineral discovery*cooperative Research Centre A legacy for mineral exploration science Mineral Systems Q3 Fluid reservoirs
 Mineral Systems Q3 Fluid reservoirs 1 Key Parameter Mineral System Exploration is reflected in scale-dependent translation A. Gradient in hydraulic potential B. Permeability C. Solubility sensitivity to
Mineral Systems Q3 Fluid reservoirs 1 Key Parameter Mineral System Exploration is reflected in scale-dependent translation A. Gradient in hydraulic potential B. Permeability C. Solubility sensitivity to
General Chemistry Multiple Choice Questions Chapter 8
 1 Write the skeleton chemical equation for the following word equation: Hydrochloric acid plus magnesium yields magnesium chloride and hydrogen gas. a HClO 4 + Mg --> MgClO 4 + H 2 b HClO 4 + Mg --> MgClO
1 Write the skeleton chemical equation for the following word equation: Hydrochloric acid plus magnesium yields magnesium chloride and hydrogen gas. a HClO 4 + Mg --> MgClO 4 + H 2 b HClO 4 + Mg --> MgClO
WEATHERING. Weathering breakdown of rock materials Erosion transport of broken-down materials
 WEATHERING the interacting physical, chemical & biological processes that progressively alter the original lithologic character of rocks to produce secondary minerals (e.g. clays) & unconsolidated regolith
WEATHERING the interacting physical, chemical & biological processes that progressively alter the original lithologic character of rocks to produce secondary minerals (e.g. clays) & unconsolidated regolith
UV-V-NIR Reflectance Spectroscopy
 UV-V-NIR Reflectance Spectroscopy Methods and Results A. Nathues Naturally-occurring inorganic substances with a definite and predictable chemical composition and physical properties Major groups: Silicates
UV-V-NIR Reflectance Spectroscopy Methods and Results A. Nathues Naturally-occurring inorganic substances with a definite and predictable chemical composition and physical properties Major groups: Silicates
Radionuclide Immobilization in the Phases Formed by Corrosion of Spent Nuclear Fuel: The Long-Term Assessment
 Radionuclide Immobilization in the Phases Formed by Corrosion of Spent Nuclear Fuel: The Long-Term Assessment Rodney C. Ewing (P.I) Fanrong Chen and Donggao Zhao (Post-doctoral fellows) Department of Nuclear
Radionuclide Immobilization in the Phases Formed by Corrosion of Spent Nuclear Fuel: The Long-Term Assessment Rodney C. Ewing (P.I) Fanrong Chen and Donggao Zhao (Post-doctoral fellows) Department of Nuclear
Dust formation in AGB stars. David Gobrecht Sergio Cristallo Luciano Piersanti Stefan T. Bromley Isabelle Cherchneff & Arkaprabha Sarangi
 Dust formation in AGB stars David Gobrecht Sergio Cristallo Luciano Piersanti Stefan T. Bromley Isabelle Cherchneff & Arkaprabha Sarangi Evidence for dust In presolar meteoritic grains with particular
Dust formation in AGB stars David Gobrecht Sergio Cristallo Luciano Piersanti Stefan T. Bromley Isabelle Cherchneff & Arkaprabha Sarangi Evidence for dust In presolar meteoritic grains with particular
