Organic Synthesis: The Disconnection Approach
|
|
|
- Augustus Robinson
- 5 years ago
- Views:
Transcription
1
2
3 rganic Synthesis: The Disconnection Approach
4
5 rganic Synthesis: The Disconnection Approach 2 nd Edition Stuart Warren Chemistry Department, Cambridge University, UK and Paul Wyatt School of Chemistry, University of Bristol, UK A John Wiley and Sons, Ltd., Publication
6 This edition first published John Wiley & Sons, Inc. egistered office John Wiley & Sons Ltd, The Atrium, Southern Gate, Chichester, West Sussex, P19 8SQ, United Kingdom For details of our global editorial offices, for customer services and for information about how to apply for permission to reuse the copyright material in this book please see our website at The right of the author to be identified as the author of this work has been asserted in accordance with the Copyright, Designs and Patents Act All rights reserved. No part of this publication may be reproduced, stored in a retrieval system, or transmitted, in any form or by any means, electronic, mechanical, photocopying, recording or otherwise, except as permitted by the UK Copyright, Designs and Patents Act 1988, without the prior permission of the publisher. Wiley also publishes its books in a variety of electronic formats. Some content that appears in print may not be available in electronic books. Designations used by companies to distinguish their products are often claimed as trademarks. All brand names and product names used in this book are trade names, service marks, trademarks or registered trademarks of their respective owners. The publisher is not associated with any product or vendor mentioned in this book. This publication is designed to provide accurate and authoritative information in regard to the subject matter covered. It is sold on the understanding that the publisher is not engaged in rendering professional services. If professional advice or other expert assistance is required, the services of a competent professional should be sought. The publisher and the author make no representations or warranties with respect to the accuracy or completeness of the contents of this work and specifically disclaim all warranties, including without limitation any implied warranties of fitness for a particular purpose. This work is sold with the understanding that the publisher is not engaged in rendering professional services. The advice and strategies contained herein may not be suitable for every situation. In view of ongoing research, equipment modifications, changes in governmental regulations, and the constant flow of information relating to the use of experimental reagents, equipment, and devices, the reader is urged to review and evaluate the information provided in the package insert or instructions for each chemical, piece of equipment, reagent, or device for, among other things, any changes in the instructions or indication of usage and for added warnings and precautions. The fact that an organization or Website is referred to in this work as a citation and/or a potential source of further information does not mean that the author or the publisher endorses the information the organization or Website may provide or recommendations it may make. Further, readers should be aware that Internet Websites listed in this work may have changed or disappeared between when this work was written and when it is read. No warranty may be created or extended by any promotional statements for this work. Neither the publisher nor the author shall be liable for any damages arising herefrom. Library of Congress Cataloging-in-Publication Data Warren, Stuart G. rganic synthesis : the disconnection approach / Stuart Warren and Paul Wyatt. 2nd ed. p. cm. Includes bibliographical references and index. ISBN (cloth) ISBN (pbk. : alk. paper) 1. rganic compounds Synthesis. I. Wyatt, Paul. II. Title. QD262.W dc A catalogue record for this book is available from the British Library. ISBN (HBK) (PBK)
7 The first edition was written with the active participation of Denis Marrian who died in We dedicate this second edition to Denis Haigh Marrian, , a great teacher and friend.
8
9 Contents Preface General eferences ix xi 1. The Disconnection Approach 1 2. Basic Principles: Synthons and eagents Synthesis of Aromatic Compounds 7 3. Strategy I: The rder of Events ne-group C X Disconnections Strategy II: Chemoselectivity Two-Group C X Disconnections Strategy III: eversal of Polarity, Cyclisations, Summary of Strategy Amine Synthesis Strategy IV: Protecting Groups ne Group C C Disconnections I: Alcohols General Strategy A: Choosing a Disconnection Strategy V: Stereoselectivity A ne Group C C Disconnections II: Carbonyl Compounds Strategy VI: egioselectivity Alkene Synthesis Strategy VII: Use of Acetylenes (Alkynes) Two-Group C C Disconnections I: Diels-Alder eactions Strategy VIII: Introduction to Carbonyl Condensations Two-Group C C Disconnections II: 1,3-Difunctionalised Compounds Strategy IX: Control in Carbonyl Condensations Two-Group C C Disconnections III: 1,5-Difunctionalised Compounds Conjugate (Michael) Addition and obinson Annelation Strategy X: Aliphatic Nitro Compounds in Synthesis Two-Group Disconnections IV: 1,2-Difunctionalised Compounds Strategy XI: adical eactions in Synthesis Two-Group Disconnections V: 1,4-Difunctionalised Compounds Strategy XII: econnection Two-Group C C Disconnections VI: 1,6-diCarbonyl Compounds General Strategy B: Strategy of Carbonyl Disconnections Strategy XIII: Introduction to ing Synthesis: Saturated Heterocycles Three-Membered ings Strategy XIV: earrangements in Synthesis Four-Membered ings: Photochemistry in Synthesis 245
10 viii Contents 33. Strategy XV: The Use of Ketenes in Synthesis Five-Membered ings Strategy XVI: Pericyclic eactions in Synthesis: Special Methods for Five-Membered ings Six-Membered ings General Strategy C: Strategy of ing Synthesis Strategy XVII: Stereoselectivity B Aromatic Heterocycles General Strategy D: Advanced Strategy 313 Index 325
11 Preface In the 26 years since Wiley published rganic Synthesis: The Disconnection Approach by Stuart Warren, this approach to the learning of synthesis has become widespread while the book itself is now dated in content and appearance. In 2007, Wiley published rganic Synthesis: Strategy and Control by Paul Wyatt and Stuart Warren. This much bigger book is designed as a sequel for fourth year undergraduates and research workers in universities and industry. The accompanying workbook was published in This new book made the old one look very dated in style and content and exposed gaps between what students were expected to understand in the 1980s and what they are expected to understand now. This second edition is intended to fill some of those gaps. The plan of the original book is the same in the second edition. It alternates chapters presenting new concepts with strategy chapters that put the new work in the context of overall planning. The 40 chapters have the same titles: some chapters have hardly been changed while others have undergone a thorough revision with considerable amounts of new material. In most cases examples from recent years are included. ne source of new material is the courses that the authors give in the pharmaceutical industry. ur basic course is The Disconnection Approach and the material we have gathered for this course has reinforced our attempts to give reasons for the synthesis of the various compounds which we believe enlivens the book and makes it more interesting for students. We hope to complete a second edition of the workbook shortly after the publication of the main text. The first edition of the textbook was in fact the third in a series of books on organic chemistry published by Wiley. The first: The Carbonyl Group: an Introduction to rganic Mechanisms, published in 1974, is a programmed book asking for a degree of interaction with the reader who was expected to solve problems while reading. People rarely use programmed learning now as the method has been superseded by interactive programmes on computers. Paul Wyatt is writing an electronic book to replace The Carbonyl Group which will complete a package of an electronic book and books with associated workbooks in a uniform format that we hope will prove of progressive value as students of organic chemistry develop their careers. Stuart Warren and Paul Wyatt March 2008.
12
13 General eferences Full details of important books referred to by abbreviated titles in the chapters to avoid repetition. Clayden, Lithium: J. Clayden, rganolithiums: Selectivity for Synthesis, Pergamon, Clayden, rganic Chemistry: J. Clayden, N. Greeves, S. Warren and P. Wothers, rganic Chemistry, xford University Press, xford, Comp. rg. Synth.: eds. Ian Fleming and B. M. Trost, Comprehensive rganic Synthesis, Pergamon, xford, 1991, six volumes. Corey, Logic: E. J. Corey and X.-M. Cheng, The Logic of Chemical Synthesis, Wiley, New York, Fieser, eagents: L. Fieser and M. Fieser, eagents for rganic Synthesis, Wiley, New York, 20 volumes, , later volumes by T.-L. Ho. Fleming, rbitals: Ian Fleming, Frontier rbitals and rganic Chemical eactions, Wiley, London, Fleming, Syntheses: Ian Fleming, Selected rganic Syntheses, Wiley, London, Houben-Weyl; Methoden der rganischen Chemie, ed.e.müller, and Methods of rganic Chemistry, ed. H.-G. Padeken, Thieme, Stuttgart, many volumes House: H.. House, Modern Synthetic eactions, Benjamin, Menlo Park, Second Edition, Nicolaou and Sorensen: K. C. Nicolaou and E. Sorensen, Classics in Total Synthesis: Targets, Strategies, Methods. VCH, Weinheim, Second volume now published. Saunders, Top Drugs: J. Saunders, Top Drugs: Top Synthetic outes, xford University Press, xford, Strategy and Control; P. Wyatt and S. Warren, rganic Synthesis: Strategy and Control, Wiley, Chichester, 2007 and Workbook, Vogel: B. S. Furniss, A. J. Hannaford, P. W. G. Smith, and A.. Tatchell, Vogel s Textbook of Practical rganic Chemistry, Fifth Edition, Longman, Harlow, 1989.
14
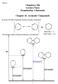
15 1 The Disconnection Approach This book is about making molecules. r rather it is to help you design your own syntheses by logical and sensible thinking. This is not a matter of guesswork but requires a way of thinking backwards that we call the disconnection approach. When you plan the synthesis of a molecule, all you know for certain is the structure of the molecule you are trying to make. It is made of atoms but we don t make molecules from atoms: we make them from smaller molecules. But how to choose which ones? If you wanted to make, say, a wooden joint, you would look in a do-it-yourself book on furniture and you would find an exploded diagram showing which pieces you would need and how they would fit together. screw screw The disconnection approach to the design of synthesis is essentially the same: we explode the molecule into smaller starting materials on paper and then combine these by chemical reactions. It isn t as easy as making wooden joints because we have to use logic based on our chemical knowledge to choose these starting materials. The first chemist to suggest the idea was obert obinson who published his famous tropinone synthesis 1 in His term was imaginary rganic Synthesis: The Disconnection Approach. Second Edition 2008 John Wiley & Sons, Ltd Stuart Warren and Paul Wyatt
16 2 1 The Disconnection Approach hydrolysis and he put dashed lines across a tropinone structure. Tropinone: obinson's Analysis NMe imaginary hydrolysis use symmetry CH CH + MeNH 2 + This was a famous synthesis because it is so short and simple and also because it makes a natural product in a way that imitates nature. The reaction is carried out at ph 7 in water. In fact obinson didn t use acetone, as suggested by his imaginary hydrolysis, but acetone dicarboxylic acid. This procedure is an improved one invented by Schöpf 2 in Tropinone: Synthesis CH + MeNH 2 + CH C 2 H C 2 H ph 7 water NMe 92.5% yield Amazingly, nobody picked up the idea until the 1960s when E. J. Corey at Harvard was considering how to write a computer program to plan organic syntheses. 3 He needed a systematic logic and he chose the disconnection approach, also called retrosynthetic analysis. All that is in this book owes its origin to his work. The computer program is called LHASA and the logic survives as a way of planning syntheses used by almost all organic chemists. It is more useful to humans than to machines. The Synthesis of Multistriatin Multistriatin 1 is a pheromone of the elm bark beetle. This beetle distributes the fungus responsible for Dutch elm disease and it was hoped that synthetic multistriatin might trap the beetle and prevent the spread of the disease. It is a cyclic compound with two oxygen atoms both joined to the same carbon atom (C-6 in 1) and we call such ethers acetals ; multistriatin acetal functional group We know one good way to make acetals: the reliable acid-catalysed reaction between two alcohols or one diol and an aldehyde or ketone. H + H H Intending to use this reliable reaction for our acetal we must disconnect the two C bonds to C 6 and reveal the starting material 2, drawn first in a similar way to 1, and then straightened
17 1 The Synthesis of Multistriatin 3 out to look more natural 2a. Numbering the carbon atoms helps to make sure 2 and 2a are the same. disconnect the acetal H H redraw a H 1 H We now have a continuous piece of carbon skeleton with two H groups and a ketone. No doubt we shall make this by forming a C C bond. But which one? We know that ketones can form nucleophilic enolates so disconnecting the bond between C 4 and C 5 is a good choice because one starting material 3 is symmetrical. As we plan to use an enolate we need to make 3 nucleophilic and therefore 4 must be electrophilic so we write plus and minus charges to show that. 5 4 H + H H 2b 3 H 4 Anion 3 can be made from the available ketone 5 but the only sensible way to make 4 electrophilic is to add a leaving group X, such as a halogen, deciding later exactly what to use. base H H X H H 6; X = leaving group Compound 6 has three functional groups. ne is undefined but the other two must be alcohols and must be on adjacent carbon atoms. There is an excellent reaction to make such a combination: the dihydroxylation of an alkene with a hydroxylating agent such as s 4. A good starting material becomes the unsaturated alcohol 7a as that is known. X H X H H 6a; X = leaving group 7 7a In one synthesis 4 the alcohol 7a was made from the available acid 8 and the leaving group (X in 6) was chosen as tosylate (Ts; toluene-p-sulfonate). H 2 C 8 LiAlH 4 H 7a TsCl pyridine Ts 7; X = Ts The two pieces were joined together by making the enolate of 5 and reacting it with 7; X= Ts. The unsaturated ketone 9 was then oxidised with a peroxyacid to give the epoxide 10 and
18 4 1 The Disconnection Approach cyclisation with the Lewis acid SnCl 4 gave the target molecule (TM) multistriatin base 2. 7; X = Ts 9 C 3 H SnCl 4 10 TM 1 You may have noticed that the synthesis does not exactly follow the analysis. We had planned to use the keto-diol 2b but in the event this was a less practical intermediate than the keto-epoxide 10. It often turns out that experience in the laboratory reveals alternatives that are better than the original plan. The basic idea the strategy remains the same. Summary: outine for Designing a Synthesis 1. Analysis (a) ecognise the functional groups in the target molecule. (b) Disconnect with known reliable reactions in mind. (c) epeat as necessary to find available starting materials. 2. Synthesis (a) Write out the plan adding reagents and conditions. (b) Modify the plan according to unexpected failures or successes in the laboratory. We shall develop and continue to use this routine throughout the book. What the est of the Book Contains The synthesis of multistriatin just described has one great fault: no attempt was made to control the stereochemistry at the four chiral centres (black blobs in 11). nly the natural stereoisomer attracts the beetle and stereoselective syntheses of multistriatin have now been developed. 11; chiral centres in multistriatin We must add stereochemistry to the list of essential background knowledge an organic chemist must have to design syntheses effectively. That list is now: 1. An understanding of reaction mechanisms. 2. A working knowledge of reliable reactions. 3. An appreciation that some compounds are readily available. 4. An understanding of stereochemistry. Don t be concerned if you feel you are weak in any of these areas. The book will strengthen your understanding as you progress. Each chapter will build on whichever of the four points are relevant. If a chapter demands the understanding of some basic chemistry, there is a list of references at the start to chapters in Clayden rganic Chemistry to help you revise. Any other textbook of organic chemistry will have similar chapters.
19 1 eferences 5 The elm bark beetle pheromone contains three compounds: multistriatin, the alcohol 12 and α-cubebene 13. At first we shall consider simple molecules like 12 but by the end of the book we shall have thought about molecules at least as complex as multistriatin and cubebene. H H 1; multistriatin 12 13; -cubebene Multistriatin has been made many times by many different strategies. Synthesis is a creative science and there is no correct synthesis for a molecule. We shall usually give only one synthesis for each target in this book: you may well be able to design shorter, more stereochemically controlled, higher yielding, more versatile in short better syntheses than those already published. If so, you are using the book to advantage. eferences 1.. obinson, J. Chem. Soc., 1917, 111, C. Schöpf and G. Lehmann, Liebig s Annalen, 1935, 518, E. J. Corey, Quart. ev., 1971, 25, 455; E. J. Corey and X.-M. Cheng, The Logic of Chemical Synthesis, Wiley, New York, G. T. Pearce, W. E. Gore and. M. Silverstein, J. rg. Chem., 1976, 41, 2797.
20
21 2 Basic Principles: Synthons and eagents Synthesis of Aromatic Compounds Background Needed for this Chapter eferences to Clayden, rganic Chemistry: Electrophilic aromatic substitution; chapter 22. (Electrophilic Aromatic Substitution) Nucleophilic aromatic substitution: chapter 23 (Electrophilic Alkenes). eduction: chapter 24 (Chemoselectivity: Selective eactions and Protection). Synthesis of Aromatic Compounds The benzene ring is a very stable structural unit. Making aromatic compounds usually means adding something(s) to a benzene ring. The disconnection is therefore almost always of a bond joining a side chain to the benzene ring. All we have to decide is when to make the disconnection and which reagents to use. You will meet the terms synthon and functional group interconversion (FGI) in this chapter. Disconnection and FGI You already know that disconnections are the reverse of known reliable reactions so you should not make a disconnection unless you have such a reaction in mind. In designing a synthesis for the local anaesthetic benzocaine 1, we see an ester group and know that esters are reliably made from some derivative of an acid (here 2) and an alcohol (here ethanol). We should disconnect the C ester bond. From now on we will usually write the reason for a disconnection or the name of the forward reaction above the arrow. Et Et C ester H + EtH H 2 N 1; benzocaine H 2 N 1a H 2 N 2 The sign for a disconnection on a molecule is some sort of wiggly line across the bond being disconnected. You can draw this line in any way you like within reason. The reaction arrow is the implies arrow from logic. The argument is that the existence of any ester implies that it can be made from an acid and an alcohol. rganic Synthesis: The Disconnection Approach. Second Edition 2008 John Wiley & Sons, Ltd Stuart Warren and Paul Wyatt
22 8 2 Basic Principles: Synthons and eagents Synthesis of Aromatic Compounds We should now like to disconnect either the NH 2 or the C 2 H group but we know of no good reactions corresponding to those disconnections. We need to change both groups into some other groups that can be added to a benzene ring by a known reliable reaction. This process is called functional group interconversion or FGI for short and is an imaginary process, just like a disconnection. It is the reverse of a real reaction. Here we know that we can make amino groups by reduction of nitro groups and aryl carboxylic acids by oxidation of alkyl groups. The FGIs are the reverse of these reactions. H FGI H FGI CH 3 H 2 N 2 N 2 N We oxidised the amino group first and reduced the acid second. The order is unimportant but is something we come back to in the forward reaction. What matters is that we have found a starting material 4 that we know how to make. If we disconnect the nitro group 4a we shall be left with toluene 5 and toluene can be nitrated in the para-position with a mixture of nitric and sulfuric acids. CH 3 C N CH 3 2 N 4a nitration H 5; toluene Now we should write out the synthesis. You cannot of course predict exactly which reagents and conditions will be successful and no sensible organic chemist would attempt to do this without studying related published work. It is enough to make suggestions for the type of reagent needed. We shall usually give the reagents used in the published work and conditions where they seem to matter. Here it is important to nitrate first and oxidise second to get the right substitution pattern. 1 HN 3 5 H 2 S 4 2 N CH 3 KMn 4 2 N 4 3 C 2 H H 2 Pd/C H 2 N 2 C 2 H EtH H TM 1 Synthons Illustrated by Friedel-Crafts Acylation The useful disconnection 6a corresponds to Friedel-Crafts acylation of aromatic rings and is the obvious one on the ketone 6 having the perfume of hawthorn blossom. eaction 2 of ether 7 with MeCCl and AlCl 3 gives 6 in 94 96% yield a good reaction indeed. Me Me 6 6a C C Friedel -Crafts Me 7 H + Cl
23 2 Synthons Illustrated by Friedel-Crafts Alkylation 9 In both this reaction and the nitration of toluene we used to make benzocaine, the reagent is a cation: MeC + for the Friedel-Crafts and N + 2 for the nitration. ur first choice on disconnecting a bond to a benzene ring is to look for a cationic reagent so that we can use electrophilic aromatic substitution. We know not only which bond to break but also in which sense electronically to break it. In principle we could have chosen either polarity from the same disconnection: a (we actually chose) or b (we did not). b a Me Me Me 8 9 6a The four fragments 8 11 are synthons that is idealised ions that may or may not be involved in the actual reaction but help us to work out which reagent to choose. As it happens, synthon 11 is a real intermediate but the others are not. For an anionic synthon like 10 the reagent is often the corresponding hydrocarbon as H + is lost during the reaction. For a cationic synthon like 11 the reagent is often the corresponding halide as that will be lost as a leaving group during the reaction. It is a matter of personal choice in analysing a synthesis problem whether you draw the synthons or go direct to the reagents. As you become more proficient at retrosynthetic analysis, you will probably find that drawing the synthons becomes unnecessary and cumbersome. Me 6a C C Friedel -Crafts Me Cl Me 12 7 H synthons reagents Synthons Illustrated by Friedel-Crafts Alkylation Friedel-Crafts alkylation is also useful though less reliable than acylation. With that in mind, we could disconnect BHT 13 ( Butylated Hydroxy-Toluene ) at either bond b to remove the methyl group or bond a to remove both t-butyl groups. There are various reasons for preferring a. para-cresol 15 is available whereas 14 is not. The t-butyl cation is a much more stable intermediate than the methyl cation and t-alkylations are among the most reliable. Finally the H group is more powerfully ortho-directing than the methyl group. H b C C a H a a C C H Me Friedel -Crafts 14 13; BHT 15 b Friedel -Crafts t-butyl cation
24 10 2 Basic Principles: Synthons and eagents Synthesis of Aromatic Compounds We have a choice of reagents for the t-butyl cation: a halide with Lewis acid catalysis, and t-butanol or isobutene with protic acid catalysis. The least wasteful is the alkene as nothing is lost. Protonation gives the t-butyl cation and two t-butyl groups are added in one operation. 3 H H H 15 13; BHT Functional Group Addition Illustrated by Friedel-Crafts Alkylation Attempting Friedel-Crafts alkylation with primary halides often gives the wrong product by rearrangement of the intermediate cation. If we want to make i-butylbenzene 16, it seems obvious that we should alkylate benzene with an i-butyl halide, e.g. 18 and AlCl 3. 16; i-butylbenzene C C Friedel -Crafts H + Cl This reaction gives two products 21 and 22 but neither contains the i-butyl group. Both contain instead the t-butyl group. The intermediate complex rearranges by hydride shift 19 into the t-butyl cation 20 as the primary cation 17 is too unstable. 18 AlCl 3 Cl 3 Al Cl H Polyalkylation was an advantage in the synthesis of BHT 13: it is the rearrangement that is chiefly unacceptable here. Friedel-Crafts acylation avoids both problems. The acyl group does not rearrange and the product is deactivated towards further electrophilic attack by the electron-withdrawing carbonyl group. We have an extra step: reduction of the ketone to a CH 2 group. There are various ways to do this (see chapter 24) here the Clemmensen reduction is satisfactory. 4 + Cl AlCl 3 Zn, Hg conc. HCl TM 16 The preliminary to the corresponding disconnection is the addition (imaginary) of a functional group where there was none. We call this FGA (functional group addition). The corresponding
25 2 Changing the Polarity: Nucleophilic Aromatic Substitution 11 known reliable reaction is the removal of the functional group. We could put the carbonyl group anywhere but we put it next to the benzene ring as it then allows us to do a reliable disconnection. TM 16 FGA 24 C C Friedel -Crafts H + Cl 23 eliable eagents for Electrophilic Substitution Table 2.1 summarises the various reagents we have mentioned (and some we haven t). Full details of mechanisms, orientation and applications appear in Clayden chapter 22. TABLE 2.1 eagents for aromatic electrophilic substitution H X electrophilic reagent X Synthon eagent eaction Comments + Br + AlCl 3 Friedel-Crafts good for t-alkyl H or alkene +H + alkylation 5 K for s-alkyl C + CCl + AlCl 3 Friedel-Crafts acylation very general N + 2 HN 3 + H 2 S 4 nitration very vigorous Cl + Cl 2 + FeCl 3 chlorination other Lewis acids used too Br + Br 2 + Fe (=FeBr 3 ) bromination other Lewis acids used too + S 2 H H 2 S 4 sulfonation may need fuming H 2 S 4 + S 2 Cl ClS 2 H + H 2 S 4 chloro-sulfonation very vigorous + ArN 2 ArNH 2 + HN diazo-coupling product is Ar 1 N=NAr 2 Changing the Polarity: Nucleophilic Aromatic Substitution If we make the same disconnections as before 25 and 27 but change the polarity we need electrophilic aromatic rings and nucleophilic reagents. We shall need a leaving group X (might be a halogen) on the aromatic ring 26 and reagents such as alkoxides or amines. nucleophilic substitution X + + HN 2 nucleophilic substitution N The nucleophilic reagents are behaving normally for alcohols or amines but the aromatic electrophiles present a problem. Benzene rings are nucleophilic, if weakly, but are not electrophilic at all. There is no S N 2 reaction on an aryl halide. To get the reactions we want, we must have
26 12 2 Basic Principles: Synthons and eagents Synthesis of Aromatic Compounds ortho- orpara-electron-withdrawing groups such as N 2 or C= to accept the electrons as the nucleophile adds 28 to form 29. N Cl N Cl N Fortunately, nitro groups go in the right positions (i.e. ortho and para but not meta) by direct nitration of, say, chlorobenzene. So we can be guided in our choice of polarity by the nature of the target molecule. The Lilly pre-emergent herbicide trifluralin B 31 has three electron-withdrawing groups: two nitro and one CF 3, ortho- andpara- to the amine, ideal for nucleophilic substitution on 32. The nitro groups can be introduced by nitration as Cl directs ortho, para while CF 3 directs meta. i-pr 2 N N 2 nucleophilic substitution i-pr 2 NH + Cl N 2 electrophilic substitution Cl 2 N CF 3 2 N CF 3 CF The synthesis 5 is simplicity itself, as the synthesis of any agrochemical must be. The base in the second step is to remove the HCl produced in the reaction, not to deprotonate the amine. N 2 N 2 Cl HN 3 Cl i-pr 2 NH i-pr 2 N CF 3 H 2 S 4 2 N CF 3 base 2 N CF Thinking Mechanistically It is obvious that the choice between nucleophilic and electrophilic substitution must be mechanistically made but this is generally true of the choice of all disconnections, synthons and reagents. The formation of 31 was easy because the aryl chloride was activated by three groups. In the synthesis of fluoxetine (Prozac), a rather widely taken anti-depressant, aryl ether 34 is an essential intermediate. 6 Though disconnection b looks attractive, as a simple S N 2 reaction should work well, disconnection a was preferred because 34 must be a single enantiomer and enantiomerically pure alcohol 36 was available. F 3 C a b Ph 34 NHMe nucleophilic substitution a F 3 C F H + Ph NHMe
27 2 Changing the Polarity: Nucleophilic Aromatic Substitution by the S N 1 Mechanism 13 You should have been surprised to see fluoride as the leaving group. Fluoride is the worst leaving group among the halogens as the C F bond is very strong: it is rare to see an S N 2 reaction with fluoride as the leaving group. Yet it is the best choice for nucleophilic aromatic substitution especially when the ring is only weakly activated as here with just one CF 3 group. In this two-step reaction, the difficult step is the addition of the nucleophile: aromaticity is destroyed and the intermediate is an unstable anion. The second step 38 is fast. Fluorine accelerates the first step as it is so electronegative and it doesn t matter that it hinders the second step as that is fast anyway. F F NHMe NHMe 34 F 3 C Ph F 3 C Ph You may have noticed something else. The formation of trifluralin 31 showed that amines are good nucleophiles for nucleophilic aromatic substitution and the nucleophile here is an amino-alcohol 36. Direct reaction with 36 might lead to the formation of an amine instead of an ether. To avoid this, 36 is first treated with NaH to make the oxyanion and then added to 35. The alcohol is less nucleophilic but the oxyanion is more nucleophilic than the amine. We hope you now see why an understanding of reaction mechanisms is an essential preliminary to the designing of syntheses. Changing the Polarity: Nucleophilic Aromatic Substitution by the S N 1 Mechanism Though the S N 2 mechanism is not available for aromatic nucleophilic substitutions, the S N 1is providing we use the very best leaving group available. This is a molecule of nitrogen released from a diazonium salt 42 on gentle warming. A standard sequence is nitration of an aromatic compound 39 to give 40, reduction to the amine 41 and diazotisation with NaN 2 /HCl to give the diazonium salt 42. Nitrous acid HN is the true reagent giving N + that attacks at nitrogen. N 2 NH HN 2 3 H 2 /Pd/C NaN 2 H 2 S 4 or SnCl 2 HCl, 5 C N The diazonium salt 42 is stable at 0 5 C but decomposes to N 2 and an unstable aryl cation 43 on warming to room temperature. The empty orbital of 43 is in an sp 2 orbital in the plane of the aromatic ring, quite unlike the normal p orbital for cations like 20. eaction occurs with any available nucleophile, even water, and this is a route to phenols 45. N N >10 C H 2 H 2 H H 42a
28 14 2 Basic Principles: Synthons and eagents Synthesis of Aromatic Compounds This route is particularly valuable for substituents that cannot easily be added by electrophilic substitution such as H or CN. Table 2.2 gives you a selection of reagents. For the addition of CN, Cl or Br, copper (I) derivatives usually give the best results. So the aryl nitrile 46 might come from amine 47 via a diazonium salt and routine disconnections lead us back to toluene. Me nucleophilic substitution Me FGI Me electrophilic substitution Me CN NH 2 N ; toluene The synthesis is straightforward. 7 In the laboratory you would not have to carry out the first two steps as the amine 47 can be bought. Industry makes it on a large scale by this route. Notice that we do not draw the diazonium salt. You can if you want, but it is usual to show two steps carried out without isolation of the intermediate in this style: 1. reagent A, 2. reagent B. This makes it clear that all the reagents are not just mixed together. Another style is used in Table 2.2: the reactive intermediate is in square brackets. But it is helpful to show conditions for the diazotisation as temperature control is important. Me HN 3 Me H 2 /Pd/C Me 1. NaN 2 HCl, 5 C Me H 2 S 4 N 2 or SnCl 2 47 NH 2 2. Cu(I)CN CN 5; toluene TABLE 2.2 eagents for aromatic nucleophilic substitution on ArN 2 + NH 2 NaN 2 HCl, 5 C N 2 reagents Z Synthon eagents Comments H water probably S N 1 alcohol H probably S N 1 CN Cu(I)CN may be a radical reaction Cl Cu(I)Cl may be a radical reaction Br Cu(I)Br may be a radical reaction I KI best way to add iodine Ar ArH Friedel-Crafts arylation H H 3 P 2 or EtH/H + + reduction of ArN 2 ortho- and para- Product Mixtures We used the nitration of toluene to give both the para-nitro 4 and the ortho-nitro compounds 48. In fact the reaction gives a mixture. This is acceptable providing the compounds can be separated and especially so if industry does the job on a very large scale, as here. The synthesis
29 2 eferences 15 of the sweetener saccharine is a good example. Saccharine 50 is a cyclic imide: that is a double amide from one nitrogen atom and two acids. If we disconnect the C N and S N bonds the two acids one carboxylic and one sulfonic are revealed 51. Both groups are meta-directing so we must do FGI to convert one of them into an ortho,para-directing group and we can use the same oxidation reaction we met at the start of the chapter (4 to 3). Now 52 can be made by sulfonation. CH 3 C-N and S-N H FGI NH H H S amides S S 50; saccharine electrophilic substitution 5 toluene In practice chloro-sulfonic acid is used as this gives the sulfonyl chloride directly. You may be surprised at this, thinking that Cl might be the best leaving group. But there is no Lewis acid here. Instead the very strong chloro-sulfonic acid protonates itself to provide a molecule of water as leaving group (see workbook). Me ClS 2 H Me + Me 5; toluene S 2 Cl Cl 2 S The reaction gives a mixture of the ortho- 53 and para- 54 products. The ortho-compound is converted into saccharine by reaction with ammonia and oxidation and the para-compound toluene-p-sulfonyl chloride 54, or tosyl chloride, is sold as a reagent for converting alcohols into leaving groups. 53 NH 3 Me KMn 4 C 2 H S 2 NH 2 S 2 NH heat NH S 50; saccharine eferences 1. Drug Synthesis, vol. 1, page 9; H. Salkowski, Ber., 1895, 28, P. H. Gore in Friedel-Crafts and elated eactions, ed. G. A. lah, Vol III, Part 1, Interscience, New York, 1964, p W. Weinrich, Ind. Eng. Chem., 1943, 35, 264; S. H. Patinkin and B. S. Friedman in ref. 2, vol. II, part 1, p E. L. Martin, rg. eact., 1942, 1, Pesticides, p. 154; Pesticide Manual, p Saunders, Top Drugs, xford University Press, p H. T. Clarke and.. ead, rg. Synth. Coll., 1932, 1, 514.
30
Workbook for Organic Synthesis: The Disconnection Approach
 Workbook for rganic Synthesis: The Disconnection Approach Workbook for rganic Synthesis: The Disconnection Approach Second Edition Stuart Warren Reader in rganic Chemistry, Department of Chemistry, University
Workbook for rganic Synthesis: The Disconnection Approach Workbook for rganic Synthesis: The Disconnection Approach Second Edition Stuart Warren Reader in rganic Chemistry, Department of Chemistry, University
Suggested solutions for Chapter 28
 s for Chapter 28 28 PBLEM 1 ow would you make these four compounds? Give your disconnections, explain why you chose them and then give reagents for the. 2 2 Me S Exercises in basic one- group C X disconnections.
s for Chapter 28 28 PBLEM 1 ow would you make these four compounds? Give your disconnections, explain why you chose them and then give reagents for the. 2 2 Me S Exercises in basic one- group C X disconnections.
Organic Chemistry. Second Edition. Chapter 19 Aromatic Substitution Reactions. David Klein. Klein, Organic Chemistry 2e
 Organic Chemistry Second Edition David Klein Chapter 19 Aromatic Substitution Reactions Copyright 2015 John Wiley & Sons, Inc. All rights reserved. Klein, Organic Chemistry 2e 19.1 Introduction to Electrophilic
Organic Chemistry Second Edition David Klein Chapter 19 Aromatic Substitution Reactions Copyright 2015 John Wiley & Sons, Inc. All rights reserved. Klein, Organic Chemistry 2e 19.1 Introduction to Electrophilic
16. Chemistry of Benzene: Electrophilic Aromatic Substitution. Based on McMurry s Organic Chemistry, 7 th edition
 16. Chemistry of Benzene: Electrophilic Aromatic Substitution Based on McMurry s Organic Chemistry, 7 th edition Substitution Reactions of Benzene and Its Derivatives Benzene is aromatic: a cyclic conjugated
16. Chemistry of Benzene: Electrophilic Aromatic Substitution Based on McMurry s Organic Chemistry, 7 th edition Substitution Reactions of Benzene and Its Derivatives Benzene is aromatic: a cyclic conjugated
Organic Chemistry. M. R. Naimi-Jamal. Faculty of Chemistry Iran University of Science & Technology
 Organic Chemistry M. R. Naimi-Jamal Faculty of Chemistry Iran University of Science & Technology Chapter 5-2. Chemistry of Benzene: Electrophilic Aromatic Substitution Based on McMurry s Organic Chemistry,
Organic Chemistry M. R. Naimi-Jamal Faculty of Chemistry Iran University of Science & Technology Chapter 5-2. Chemistry of Benzene: Electrophilic Aromatic Substitution Based on McMurry s Organic Chemistry,
16. Chemistry of Benzene: Electrophilic Aromatic Substitution جانشینی الکتروندوستی آروماتیک شیمی آلی 2
 16. Chemistry of Benzene: Electrophilic Aromatic Substitution جانشینی الکتروندوستی آروماتیک شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based
16. Chemistry of Benzene: Electrophilic Aromatic Substitution جانشینی الکتروندوستی آروماتیک شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based
16. Chemistry of Benzene: Electrophilic Aromatic Substitution جانشینی الکتروندوستی آروماتیک شیمی آلی 2
 16. Chemistry of Benzene: Electrophilic Aromatic Substitution جانشینی الکتروندوستی آروماتیک شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based
16. Chemistry of Benzene: Electrophilic Aromatic Substitution جانشینی الکتروندوستی آروماتیک شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based
Chapter 16 Chemistry of Benzene: Electrophilic Aromatic Substitution
 John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 16 Chemistry of Benzene: Electrophilic Aromatic Substitution Paul D. Adams University of Arkansas Substitution Reactions of Benzene and Its Derivatives
John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 16 Chemistry of Benzene: Electrophilic Aromatic Substitution Paul D. Adams University of Arkansas Substitution Reactions of Benzene and Its Derivatives
Chemistry of Benzene: Electrophilic Aromatic Substitution
 Chemistry of Benzene: Electrophilic Aromatic Substitution Why this Chapter? Continuation of coverage of aromatic compounds in preceding chapter focus shift to understanding reactions Examine relationship
Chemistry of Benzene: Electrophilic Aromatic Substitution Why this Chapter? Continuation of coverage of aromatic compounds in preceding chapter focus shift to understanding reactions Examine relationship
Synthesis Using Aromatic Materials
 Chapter 10 Synthesis Using Aromatic Materials ELECTROPHILIC AROMATIC SUBSTITUTION AND DIRECTED ORTHO METALATION Copyright 2018 by Nelson Education Limited 1 10.2 p Bonds Acting as Nucleophiles Copyright
Chapter 10 Synthesis Using Aromatic Materials ELECTROPHILIC AROMATIC SUBSTITUTION AND DIRECTED ORTHO METALATION Copyright 2018 by Nelson Education Limited 1 10.2 p Bonds Acting as Nucleophiles Copyright
KOT 222 Organic Chemistry II
 KOT 222 Organic Chemistry II Course Objectives: 1) To introduce the chemistry of alcohols and ethers. 2) To study the chemistry of functional groups. 3) To learn the chemistry of aromatic compounds and
KOT 222 Organic Chemistry II Course Objectives: 1) To introduce the chemistry of alcohols and ethers. 2) To study the chemistry of functional groups. 3) To learn the chemistry of aromatic compounds and
CHAPTER 16 - CHEMISTRY OF BENZENE: ELECTROPHILIC AROMATIC SUBSTITUTION
 CAPTR 16 - CMISTRY F BNZN: LCTRPILIC ARMATIC SUBSTITUTIN As stated in the previous chapter, benzene and other aromatic rings do not undergo electrophilic addition reactions of the simple alkenes but rather
CAPTR 16 - CMISTRY F BNZN: LCTRPILIC ARMATIC SUBSTITUTIN As stated in the previous chapter, benzene and other aromatic rings do not undergo electrophilic addition reactions of the simple alkenes but rather
08. Chemistry of Benzene: Electrophilic Aromatic Substitution. Based on McMurry s Organic Chemistry, 6 th edition, Chapter 16
 08. Chemistry of Benzene: Electrophilic Aromatic Substitution Based on McMurry s Organic Chemistry, 6 th edition, Chapter 16 Benzene is a nucleophile p electrons make benzene nucleophile, like alkenes.
08. Chemistry of Benzene: Electrophilic Aromatic Substitution Based on McMurry s Organic Chemistry, 6 th edition, Chapter 16 Benzene is a nucleophile p electrons make benzene nucleophile, like alkenes.
Electrophilic Aromatic Substitution. Dr. Mishu Singh Department of chemistry Maharana Pratap Govt.P.G.College Hardoi
 Electrophilic Aromatic Substitution Dr. Mishu Singh Department of chemistry Maharana Pratap Govt.P.G.College Hardoi 1 Recall the electophilic addition of HBr (or Br2) to alkenes H + nu cleophile H Br H
Electrophilic Aromatic Substitution Dr. Mishu Singh Department of chemistry Maharana Pratap Govt.P.G.College Hardoi 1 Recall the electophilic addition of HBr (or Br2) to alkenes H + nu cleophile H Br H
Lecture Topics: I. Electrophilic Aromatic Substitution (EAS)
 Reactions of Aromatic Compounds Reading: Wade chapter 17, sections 17-1- 17-15 Study Problems: 17-44, 17-46, 17-47, 17-48, 17-51, 17-52, 17-53, 17-59, 17-61 Key Concepts and Skills: Predict and propose
Reactions of Aromatic Compounds Reading: Wade chapter 17, sections 17-1- 17-15 Study Problems: 17-44, 17-46, 17-47, 17-48, 17-51, 17-52, 17-53, 17-59, 17-61 Key Concepts and Skills: Predict and propose
Suggested solutions for Chapter 41
 s for Chapter 41 41 PBLEM 1 Explain how this synthesis of amino acids, starting with natural proline, works. Explain the stereoselectivity of each step after the first. C 2 C 2 3 CF 3 C 2 2 Pd 2 C 2 +
s for Chapter 41 41 PBLEM 1 Explain how this synthesis of amino acids, starting with natural proline, works. Explain the stereoselectivity of each step after the first. C 2 C 2 3 CF 3 C 2 2 Pd 2 C 2 +
Organic Chemistry, 7 L. G. Wade, Jr. Chapter , Prentice Hall
 Organic Chemistry, 7 th Edition L. G. Wade, Jr. Chapter 17 Reactions of Aromatic Compounds 2010, Prentice Hall Electrophilic Aromatic Substitution Although h benzene s pi electrons are in a stable aromatic
Organic Chemistry, 7 th Edition L. G. Wade, Jr. Chapter 17 Reactions of Aromatic Compounds 2010, Prentice Hall Electrophilic Aromatic Substitution Although h benzene s pi electrons are in a stable aromatic
Chapter 17. Reactions of Aromatic Compounds
 Chapter 17 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Although benzene s pi electrons are in a stable aromatic system, they are available to attack a strong electrophile to give
Chapter 17 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Although benzene s pi electrons are in a stable aromatic system, they are available to attack a strong electrophile to give
12/27/2010. Chapter 15 Reactions of Aromatic Compounds
 Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen atom
Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen atom
Aromatic Compounds II
 2302272 Org Chem II Part I Lecture 2 Aromatic Compounds II Instructor: Dr. Tanatorn Khotavivattana E-mail: tanatorn.k@chula.ac.th Recommended Textbook: Chapter 17 in Organic Chemistry, 8 th Edition, L.
2302272 Org Chem II Part I Lecture 2 Aromatic Compounds II Instructor: Dr. Tanatorn Khotavivattana E-mail: tanatorn.k@chula.ac.th Recommended Textbook: Chapter 17 in Organic Chemistry, 8 th Edition, L.
REACTIONS OF AROMATIC COMPOUNDS
 A STUDENT SHOULD BE ABLE TO: REACTIONS OF AROMATIC COMPOUNDS 1. Predict the product(s) of Electrophilic aromatic substitution (EAS): halogenation, sulfonation, nitration, Friedel- Crafts alkylation and
A STUDENT SHOULD BE ABLE TO: REACTIONS OF AROMATIC COMPOUNDS 1. Predict the product(s) of Electrophilic aromatic substitution (EAS): halogenation, sulfonation, nitration, Friedel- Crafts alkylation and
Benzenes & Aromatic Compounds
 Benzenes & Aromatic Compounds 1 Structure of Benzene H H C C C H C 6 H 6 H C C C H H A cyclic conjugate molecule Benzene is a colourless odourless liquid, boiling at 80 o C and melting at 5 o C. It is
Benzenes & Aromatic Compounds 1 Structure of Benzene H H C C C H C 6 H 6 H C C C H H A cyclic conjugate molecule Benzene is a colourless odourless liquid, boiling at 80 o C and melting at 5 o C. It is
Chapter 15 Reactions of Aromatic Compounds
 Chapter 15 1 Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen
Chapter 15 1 Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen
Reactions of Aromatic Compounds. Aromatic compounds do not react like other alkenes. With an appropriate catalyst, however, benzene will react
 Reactions of Aromatic Compounds Aromatic compounds do not react like other alkenes 2 Fe 3 2 Does not form A major part of the problem for this reaction is the product has lost all aromatic stabilization,
Reactions of Aromatic Compounds Aromatic compounds do not react like other alkenes 2 Fe 3 2 Does not form A major part of the problem for this reaction is the product has lost all aromatic stabilization,
Suggested solutions for Chapter 29
 s for Chapter 29 29 PRBLEM 1 or each of the following reactions (a) state what kind of substitution is suggested and (b) suggest what product might be formed if monosubstitution occured. Br 2 3 2 S 4 S
s for Chapter 29 29 PRBLEM 1 or each of the following reactions (a) state what kind of substitution is suggested and (b) suggest what product might be formed if monosubstitution occured. Br 2 3 2 S 4 S
Benzene and Aromatic Compounds. Chapter 15 Organic Chemistry, 8 th Edition John McMurry
 Benzene and Aromatic Compounds Chapter 15 Organic Chemistry, 8 th Edition John McMurry 1 Background Benzene (C 6 H 6 ) is the simplest aromatic hydrocarbon (or arene). Four degrees of unsaturation. It
Benzene and Aromatic Compounds Chapter 15 Organic Chemistry, 8 th Edition John McMurry 1 Background Benzene (C 6 H 6 ) is the simplest aromatic hydrocarbon (or arene). Four degrees of unsaturation. It
Advanced Organic Chemistry: Retrosynthesis
 123.312 Advanced rganic Chemistry: Retrosynthesis Tutorial Question 1. Propose a retrosynthetic analysis of the following two compounds. Your answer should include both the synthons, showing your thinking,
123.312 Advanced rganic Chemistry: Retrosynthesis Tutorial Question 1. Propose a retrosynthetic analysis of the following two compounds. Your answer should include both the synthons, showing your thinking,
Organic Spectroscopy Workbook
 Organic Spectroscopy Workbook Organic Spectroscopy Workbook TOM FORREST JEAN-PIERRE RABINE MICHEL ROUILLARD A John Wiley & Sons, Ltd., Publication This edition first published 2011 2011 John Wiley and
Organic Spectroscopy Workbook Organic Spectroscopy Workbook TOM FORREST JEAN-PIERRE RABINE MICHEL ROUILLARD A John Wiley & Sons, Ltd., Publication This edition first published 2011 2011 John Wiley and
Ch.16 Chemistry of Benzene: Electrophilic Aromatic Substitution
 Ch.16 Chemistry of Benzene: Electrophilic Aromatic Substitution Electrophilic aromatic substitution: E + E + + Some electrophilic aromatic substitution: X N 2 S 3 R C R alogenation Nitration Sulfonation
Ch.16 Chemistry of Benzene: Electrophilic Aromatic Substitution Electrophilic aromatic substitution: E + E + + Some electrophilic aromatic substitution: X N 2 S 3 R C R alogenation Nitration Sulfonation
Chapter 15. Reactions of Aromatic Compounds. Electrophilic Aromatic Substitution on Arenes. The first step is the slow, rate-determining step
 Electrophilic Aromatic Substitution on Arenes Chapter 15 Reactions of Aromatic Compounds The characteristic reaction of aromatic rings is substitution initiated by an electrophile halogenation nitration
Electrophilic Aromatic Substitution on Arenes Chapter 15 Reactions of Aromatic Compounds The characteristic reaction of aromatic rings is substitution initiated by an electrophile halogenation nitration
11/26/ Polycyclic Aromatic Compounds. Polycyclic Aromatic Compounds. Polycyclic Aromatic Compounds
 9.5 Polycyclic Aromatic Compounds The general concept of aromaticity can be extended to include polycyclic aromatic compounds Benzo[a]pyrene is one of the cancer-causing substances found in tobacco smoke
9.5 Polycyclic Aromatic Compounds The general concept of aromaticity can be extended to include polycyclic aromatic compounds Benzo[a]pyrene is one of the cancer-causing substances found in tobacco smoke
2016 Pearson Education, Inc. Isolated and Conjugated Dienes
 2016 Pearson Education, Inc. Isolated and Conjugated Dienes 2016 Pearson Education, Inc. Reactions of Isolated Dienes 2016 Pearson Education, Inc. The Mechanism Double Bonds can have Different Reactivities
2016 Pearson Education, Inc. Isolated and Conjugated Dienes 2016 Pearson Education, Inc. Reactions of Isolated Dienes 2016 Pearson Education, Inc. The Mechanism Double Bonds can have Different Reactivities
Hour Examination # 1
 CEM 347 rganic Chemistry II Spring 2015 Exam # 1 Solutions Key Page 1 of 11 CEM 347 rganic Chemistry II Spring 2015 Instructor: Paul Bracher our Examination # 1 Wednesday, February 11 th, 2015 6:00 8:00
CEM 347 rganic Chemistry II Spring 2015 Exam # 1 Solutions Key Page 1 of 11 CEM 347 rganic Chemistry II Spring 2015 Instructor: Paul Bracher our Examination # 1 Wednesday, February 11 th, 2015 6:00 8:00
Loudon Chapter 23 Review: Amines Jacquie Richardson, CU Boulder Last updated 4/22/2018
 This chapter is about the chemistry of nitrogen. We ve seen it before in several places, but now we can look at several reactions that are specific to nitrogen. Amines can be subdivided based on how many
This chapter is about the chemistry of nitrogen. We ve seen it before in several places, but now we can look at several reactions that are specific to nitrogen. Amines can be subdivided based on how many
CHM 292 Final Exam Answer Key
 CHM 292 Final Exam Answer Key 1. Predict the product(s) of the following reactions (5 points each; 35 points total). May 7, 2013 Acid catalyzed elimination to form the most highly substituted alkene possible
CHM 292 Final Exam Answer Key 1. Predict the product(s) of the following reactions (5 points each; 35 points total). May 7, 2013 Acid catalyzed elimination to form the most highly substituted alkene possible
Organic Synthesis: The Disconnection Approach
 Organic Synthesis: The Disconnection Approach Organic Synthesis: The Disconnection Approach 2 nd Edition Stuart Warren Chemistry Department, Cambridge University, UK and Paul Wyatt School of Chemistry,
Organic Synthesis: The Disconnection Approach Organic Synthesis: The Disconnection Approach 2 nd Edition Stuart Warren Chemistry Department, Cambridge University, UK and Paul Wyatt School of Chemistry,
R N R N R N. primary secondary tertiary
 Chapter 19 Amines omenclature o assification of amines Amines are classified as 1, 2, or 3 based on how many R groups are attached to the nitrogen R R R R R R primary secondary tertiary When there are
Chapter 19 Amines omenclature o assification of amines Amines are classified as 1, 2, or 3 based on how many R groups are attached to the nitrogen R R R R R R primary secondary tertiary When there are
FORENSIC ANALYTICAL TECHNIQUES
 FORENSIC ANALYTICAL TECHNIQUES Analytical Techniques in the Sciences (AnTS) Series Editor: David J. Ando, Consultant, Dartford, Kent, UK A series of open learning/distance learning books which covers all
FORENSIC ANALYTICAL TECHNIQUES Analytical Techniques in the Sciences (AnTS) Series Editor: David J. Ando, Consultant, Dartford, Kent, UK A series of open learning/distance learning books which covers all
CHEMISTRY 263 HOME WORK
 Lecture Topics: CHEMISTRY 263 HOME WORK Module7: Hydrogenation of Alkenes Hydrogenation - syn and anti- addition - hydrogenation of alkynes - synthesis of cis-alkenes -synthesis of trans-alkenes Text sections:
Lecture Topics: CHEMISTRY 263 HOME WORK Module7: Hydrogenation of Alkenes Hydrogenation - syn and anti- addition - hydrogenation of alkynes - synthesis of cis-alkenes -synthesis of trans-alkenes Text sections:
Practical Statistics for Geographers and Earth Scientists
 Practical Statistics for Geographers and Earth Scientists Nigel Walford A John Wiley & Sons, Ltd., Publication Practical Statistics for Geographers and Earth Scientists Practical Statistics for Geographers
Practical Statistics for Geographers and Earth Scientists Nigel Walford A John Wiley & Sons, Ltd., Publication Practical Statistics for Geographers and Earth Scientists Practical Statistics for Geographers
Benzene and Aromatic Compounds
 1 Background Benzene and Aromatic Compounds Benzene (C 6 H 6 ) is the simplest aromatic hydrocarbon (or arene). Benzene has four degrees of unsaturation, making it a highly unsaturated hydrocarbon. Whereas
1 Background Benzene and Aromatic Compounds Benzene (C 6 H 6 ) is the simplest aromatic hydrocarbon (or arene). Benzene has four degrees of unsaturation, making it a highly unsaturated hydrocarbon. Whereas
Module9. Nuclear Magnetic Resonance Spectroscopy Nuclear Magnetic Resonance (NMR) spectroscopy - Chemical shift - Integration of signal area
 1 CHEMISTRY 263 HOME WORK Lecture Topics: Module7. Hydrogenation of Alkenes The Function of the Catalyst - Syn and anti- addition Hydrogenation of Alkynes - Syn- addition of hydrogen: Synthesis of cis-alkenes
1 CHEMISTRY 263 HOME WORK Lecture Topics: Module7. Hydrogenation of Alkenes The Function of the Catalyst - Syn and anti- addition Hydrogenation of Alkynes - Syn- addition of hydrogen: Synthesis of cis-alkenes
Chapter 17 Reactions of Aromatic Compounds
 Chapter 17 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution o General reaction - an electrophile replaces a hydrogen Electrons of pi system attack strong electrophile, generating resonancestabilized
Chapter 17 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution o General reaction - an electrophile replaces a hydrogen Electrons of pi system attack strong electrophile, generating resonancestabilized
Arrow Pushing in Organic Chemistry
 Arrow Pushing in Organic Chemistry An Easy Approach to Understanding Reaction Mechanisms Daniel E. Levy Arrow Pushing in Organic Chemistry Arrow Pushing in Organic Chemistry An Easy Approach to Understanding
Arrow Pushing in Organic Chemistry An Easy Approach to Understanding Reaction Mechanisms Daniel E. Levy Arrow Pushing in Organic Chemistry Arrow Pushing in Organic Chemistry An Easy Approach to Understanding
Chapter 17 Aromati ti S u stit tit t u i tion Reactions
 Chapter 17 Aromatic Substitution Reactions 1 17.1 Mechanism for Electricphilic Aromatic Substitution Arenium ion resonance stabilization 2 Example 1. Example 2. 3 Example 2. Mechanism of the nitration
Chapter 17 Aromatic Substitution Reactions 1 17.1 Mechanism for Electricphilic Aromatic Substitution Arenium ion resonance stabilization 2 Example 1. Example 2. 3 Example 2. Mechanism of the nitration
Ch 16 Electrophilic Aromatic Substitution
 Ch 16 Electrophilic Aromatic Substitution Mechanism - Aromatic rings typically undergo substitution, where an H is replaced with an electrophile (E+). - The rings do not typically undergo addition across
Ch 16 Electrophilic Aromatic Substitution Mechanism - Aromatic rings typically undergo substitution, where an H is replaced with an electrophile (E+). - The rings do not typically undergo addition across
C h a p t e r N i n e t e e n Aromatics II: Reactions of Benzene & Its Derivatives
 C h a p t e r N i n e t e e n Aromatics II: Reactions of Benzene & Its Derivatives Arenium ion from addition of tert-butyl cation to benzene (blue is δ+and red δ-) Note: Problems with italicized numbers
C h a p t e r N i n e t e e n Aromatics II: Reactions of Benzene & Its Derivatives Arenium ion from addition of tert-butyl cation to benzene (blue is δ+and red δ-) Note: Problems with italicized numbers
Suggested solutions for Chapter 30
 s for Chapter 30 30 PRBLEM 1 uggest a mechanism for this synthesis of a tricyclic aromatic heterocycle. 2 Cl base A simple exercise in the synthesis of a pyridine fused to a pyrrole (or an indole with
s for Chapter 30 30 PRBLEM 1 uggest a mechanism for this synthesis of a tricyclic aromatic heterocycle. 2 Cl base A simple exercise in the synthesis of a pyridine fused to a pyrrole (or an indole with
PRACTICAL RAMAN SPECTROSCOPY AN INTRODUCTION
 PRACTICAL RAMAN SPECTROSCOPY AN INTRODUCTION PRACTICAL RAMAN SPECTROSCOPY AN INTRODUCTION Peter Vandenabeele Ghent University, Belgium This edition first published 2013 2013 John Wiley & Sons, Ltd Registered
PRACTICAL RAMAN SPECTROSCOPY AN INTRODUCTION PRACTICAL RAMAN SPECTROSCOPY AN INTRODUCTION Peter Vandenabeele Ghent University, Belgium This edition first published 2013 2013 John Wiley & Sons, Ltd Registered
Treatment of cyclooctatetrene with potassium gives you a dianion. Classify the starting material and product as aromatic, antiaromatic or
 Treatment of cyclooctatetrene with potassium gives you a dianion. Classify the starting material and product as aromatic, antiaromatic or nonaromatic? 1 2 Classify cyclononatetrene and it s various ions
Treatment of cyclooctatetrene with potassium gives you a dianion. Classify the starting material and product as aromatic, antiaromatic or nonaromatic? 1 2 Classify cyclononatetrene and it s various ions
Cape Cod Community College
 Cape Cod Community College Departmental Syllabus Prepared by the Department of Natural Sciences & Applied Technology Date of Departmental Approval: February 3, 2014 Date Approved by Curriculum and Programs:
Cape Cod Community College Departmental Syllabus Prepared by the Department of Natural Sciences & Applied Technology Date of Departmental Approval: February 3, 2014 Date Approved by Curriculum and Programs:
CHEM Chapter 16. Chemistry of Benzene: Electrophilic Aromatic Substitution (homework) W
 CHEM 2425. Chapter 16. Chemistry of Benzene: Electrophilic Aromatic Substitution (homework) W Short Answer Exhibit 16-1 MATCH a structure or term from the following list with each description below. Place
CHEM 2425. Chapter 16. Chemistry of Benzene: Electrophilic Aromatic Substitution (homework) W Short Answer Exhibit 16-1 MATCH a structure or term from the following list with each description below. Place
Chapter 16. Chemistry of Benzene: Electrophilic Aromatic Substitution. Reactivity of Benzene
 hapter 16 hemistry of Benzene: Electrophilic Aromatic Substitution Reactivity of Benzene - stabilization due to aromaticity makes benzene significantly less reactive than isolated alkenes 2 no reaction
hapter 16 hemistry of Benzene: Electrophilic Aromatic Substitution Reactivity of Benzene - stabilization due to aromaticity makes benzene significantly less reactive than isolated alkenes 2 no reaction
Keynotes in Organic Chemistry
 Keynotes in Organic Chemistry Second Edition ANDREW F. PARSONS Department of Chemistry, University of York, UK Wiley Contents Preface xi 1 Structure and bonding 1 1.1 Ionic versus covalent bonds 1 1.2
Keynotes in Organic Chemistry Second Edition ANDREW F. PARSONS Department of Chemistry, University of York, UK Wiley Contents Preface xi 1 Structure and bonding 1 1.1 Ionic versus covalent bonds 1 1.2
Spring Term 2012 Dr. Williams (309 Zurn, ex 2386)
 Chemistry 242 Organic Chemistry II Spring Term 2012 Dr. Williams (309 Zurn, ex 2386) Web Page: http://math.mercyhurst.edu/~jwilliams/ jwilliams@mercyhurst.edu (or just visit Department web site and look
Chemistry 242 Organic Chemistry II Spring Term 2012 Dr. Williams (309 Zurn, ex 2386) Web Page: http://math.mercyhurst.edu/~jwilliams/ jwilliams@mercyhurst.edu (or just visit Department web site and look
Lecture 27 Organic Chemistry 1
 CHEM 232 rganic Chemistry I at Chicago Lecture 27 rganic Chemistry 1 Professor Duncan Wardrop April 20, 2010 1 Self Test Question Nitrosonium (not nitronium) cations can be generated by treating sodium
CHEM 232 rganic Chemistry I at Chicago Lecture 27 rganic Chemistry 1 Professor Duncan Wardrop April 20, 2010 1 Self Test Question Nitrosonium (not nitronium) cations can be generated by treating sodium
Synthesis and Structure of Alcohols Alcohols can be considered organic analogues of water.
 Synthesis and Structure of Alcohols Alcohols can be considered organic analogues of water. Alcohols are usually classified as primary, secondary and tertiary. Alcohols with the hydroxyl bound directly
Synthesis and Structure of Alcohols Alcohols can be considered organic analogues of water. Alcohols are usually classified as primary, secondary and tertiary. Alcohols with the hydroxyl bound directly
Tips for taking exams in 852
 Comprehensive Tactical Methods in rganic Synthesis W. D. Wulff 1) Know the relative reactivity of carbonyl compounds Tips for taking exams in 852 Cl > > ' > > ' N2 eg: 'Mg Et ' 1equiv. 1equiv. ' ' Et 50%
Comprehensive Tactical Methods in rganic Synthesis W. D. Wulff 1) Know the relative reactivity of carbonyl compounds Tips for taking exams in 852 Cl > > ' > > ' N2 eg: 'Mg Et ' 1equiv. 1equiv. ' ' Et 50%
ORGANIC - BROWN 8E CH. 22- REACTIONS OF BENZENE AND ITS DERIVATIVES
 !! www.clutchprep.com CONCEPT: ELECTROPHILIC AROMATIC SUBSTITUTION GENERAL MECHANISM Benzene reacts with very few reagents. It DOES NOT undergo typical addition reactions. Why? If we can get benzene to
!! www.clutchprep.com CONCEPT: ELECTROPHILIC AROMATIC SUBSTITUTION GENERAL MECHANISM Benzene reacts with very few reagents. It DOES NOT undergo typical addition reactions. Why? If we can get benzene to
24.4: Acidity of Phenols. Phenols are more acidic than aliphatic alcohols. + Electron-withdrawing groups make an O
 Chapter 24: Phenols. Alcohols contain an group bonded to an sp 3 -hybridized carbon. Phenols contain an group bonded to an sp 2 -hybridized carbon of a benzene ring 24.1: Nomenclature (please read) 24.2:
Chapter 24: Phenols. Alcohols contain an group bonded to an sp 3 -hybridized carbon. Phenols contain an group bonded to an sp 2 -hybridized carbon of a benzene ring 24.1: Nomenclature (please read) 24.2:
THE CHEMISTRY OF THE CARBONYL GROUP
 TE CEMISTY F TE CABYL GUP Professor Tim Donohoe 8 lectures, T, weeks 1-4, 2007 andout A C C You will be able to download copies of the handouts from this course at http://users.ox.ac.uk/~magd1571/teaching/teaching.htm
TE CEMISTY F TE CABYL GUP Professor Tim Donohoe 8 lectures, T, weeks 1-4, 2007 andout A C C You will be able to download copies of the handouts from this course at http://users.ox.ac.uk/~magd1571/teaching/teaching.htm
ALCOHOLS AND PHENOLS
 ALCOHOLS AND PHENOLS ALCOHOLS AND PHENOLS Alcohols contain an OH group connected to a a saturated C (sp3) They are important solvents and synthesis intermediates Phenols contain an OH group connected to
ALCOHOLS AND PHENOLS ALCOHOLS AND PHENOLS Alcohols contain an OH group connected to a a saturated C (sp3) They are important solvents and synthesis intermediates Phenols contain an OH group connected to
There are two main electronic effects that substituents can exert:
 Substituent Effects There are two main electronic effects that substituents can exert: RESONANCE effects are those that occur through the π system and can be represented by resonance structures. These
Substituent Effects There are two main electronic effects that substituents can exert: RESONANCE effects are those that occur through the π system and can be represented by resonance structures. These
Chapter 22 Amines. Nomenclature Amines are classified according to the degree of substitution at nitrogen.
 CH. 22 Chapter 22 Amines Amines are very important in biological chemistry. Most of the bases in biological acid-base reactions are amines. They are also very important nucleophiles in biochemical reactions.
CH. 22 Chapter 22 Amines Amines are very important in biological chemistry. Most of the bases in biological acid-base reactions are amines. They are also very important nucleophiles in biochemical reactions.
Suggested solutions for Chapter 32
 s for Chapter 32 32 PBLEM 1 Explain how the stereo- and regio- chemistry of these reactions are controlled. Why is the epoxidation only moderately diastereoselective, and why does the amine attack where
s for Chapter 32 32 PBLEM 1 Explain how the stereo- and regio- chemistry of these reactions are controlled. Why is the epoxidation only moderately diastereoselective, and why does the amine attack where
How to make pyridines: the Hantzsch pyridine synthesis
 ow to make pyridines: the antzsch pyridine synthesis 1191 Zinc in acetic acid (Chapter 24) reduces the oxime to the amine and we can start the synthesis by doing the conjugate addition and then reducing
ow to make pyridines: the antzsch pyridine synthesis 1191 Zinc in acetic acid (Chapter 24) reduces the oxime to the amine and we can start the synthesis by doing the conjugate addition and then reducing
Chapter 17 Reactions of Aromatic Compounds
 rganic Chemistry, 6 th Edition L. G. Wade, Jr. Chapter 17 Reactions of Aromatic Compounds Jo Blackburn Richland College, Dallas, TX Dallas County Community College District 2006, Prentice all Electrophilic
rganic Chemistry, 6 th Edition L. G. Wade, Jr. Chapter 17 Reactions of Aromatic Compounds Jo Blackburn Richland College, Dallas, TX Dallas County Community College District 2006, Prentice all Electrophilic
JEFFERSON COLLEGE COURSE SYLLABUS CHM201 ORGANIC CHEMISTRY II. 5 Credit Hours. Prepared by: Richard A. Pierce
 JEFFERSON COLLEGE COURSE SYLLABUS CHM201 ORGANIC CHEMISTRY II 5 Credit Hours Prepared by: Richard A. Pierce Revised Date: January 2008 by Ryan H. Groeneman Arts & Science Education Dr. Mindy Selsor, Dean
JEFFERSON COLLEGE COURSE SYLLABUS CHM201 ORGANIC CHEMISTRY II 5 Credit Hours Prepared by: Richard A. Pierce Revised Date: January 2008 by Ryan H. Groeneman Arts & Science Education Dr. Mindy Selsor, Dean
Amines Reading Study Problems Key Concepts and Skills Lecture Topics: Amines: structure and nomenclature
 Amines Reading: Wade chapter 19, sections 19-1-19-19 Study Problems: 19-37, 19-39, 19-40, 19-41, 19-44, 19-46, 19-47, 19-48, 19-51, 19-54 Key Concepts and Skills: Explain how the basicity of amines varies
Amines Reading: Wade chapter 19, sections 19-1-19-19 Study Problems: 19-37, 19-39, 19-40, 19-41, 19-44, 19-46, 19-47, 19-48, 19-51, 19-54 Key Concepts and Skills: Explain how the basicity of amines varies
Chapter 15. Reactions of Aromatic Compounds. 1. Electrophilic Aromatic Substitution Reactions
 hapter 15 eactions of Aromatic ompounds 1. Electrophilic Aromatic Substitution eactions v verall reaction reated by Professor William Tam & Dr. Phillis hang opyright S 3 2 S 4 S 3 2. A General Mechanism
hapter 15 eactions of Aromatic ompounds 1. Electrophilic Aromatic Substitution eactions v verall reaction reated by Professor William Tam & Dr. Phillis hang opyright S 3 2 S 4 S 3 2. A General Mechanism
SURVEY ON ARYL COMPOUNDS
 Journal of Plastic and Polymer Technology (JPPT) Vol. 1, Issue 1, Jun 2015, 111-132 TJPRC Pvt. Ltd SURVEY ON ARYL COMPOUNDS NAGHAM MAHMOOD ALJAMALI Organic Chemistry, Department of Chemistry, College of
Journal of Plastic and Polymer Technology (JPPT) Vol. 1, Issue 1, Jun 2015, 111-132 TJPRC Pvt. Ltd SURVEY ON ARYL COMPOUNDS NAGHAM MAHMOOD ALJAMALI Organic Chemistry, Department of Chemistry, College of
11/30/ Substituent Effects in Electrophilic Substitutions. Substituent Effects in Electrophilic Substitutions
 Chapter 9 Problems: 9.1-29, 32-34, 36-37, 39-45, 48-56, 58-59, 61-69, 71-72. 9.8 Substituent effects in the electrophilic substitution of an aromatic ring Substituents affect the reactivity of the aromatic
Chapter 9 Problems: 9.1-29, 32-34, 36-37, 39-45, 48-56, 58-59, 61-69, 71-72. 9.8 Substituent effects in the electrophilic substitution of an aromatic ring Substituents affect the reactivity of the aromatic
ORGANIC CHEMISTRY. Fifth Edition. Stanley H. Pine
 ORGANIC CHEMISTRY Fifth Edition Stanley H. Pine Professor of Chemistry California State University, Los Angeles McGraw-Hill, Inc. New York St. Louis San Francisco Auckland Bogota Caracas Lisbon London
ORGANIC CHEMISTRY Fifth Edition Stanley H. Pine Professor of Chemistry California State University, Los Angeles McGraw-Hill, Inc. New York St. Louis San Francisco Auckland Bogota Caracas Lisbon London
Chapter 17: Reactions of Aromatic Compounds
 1 Chapter 17: Reactions of Aromatic Compounds I. Introduction to Electrophilic Aromatic Substitution (EAS) A. General Mechanism II. Reactions of Electrophilic Aromatic Substitution A. Halogenation (E =
1 Chapter 17: Reactions of Aromatic Compounds I. Introduction to Electrophilic Aromatic Substitution (EAS) A. General Mechanism II. Reactions of Electrophilic Aromatic Substitution A. Halogenation (E =
Loudon Chapter 23 Review: Amines CHEM 3331, Jacquie Richardson, Fall Page 1
 Loudon Chapter 23 eview: Amines CEM 3331, Jacquie ichardson, Fall 2010 - Page 1 This chapter is about the chemistry of nitrogen. We ve seen it before in several places, but now we can look at several reactions
Loudon Chapter 23 eview: Amines CEM 3331, Jacquie ichardson, Fall 2010 - Page 1 This chapter is about the chemistry of nitrogen. We ve seen it before in several places, but now we can look at several reactions
Class XII: Chemistry Chapter 13: Amines Top concepts
 Class XII: Chemistry Chapter 13: Amines Top concepts 1. Amines are regarded as derivatives of ammonia in which one, two or all three hydrogen atoms are replaced by alkyl or aryl group 2. Classification
Class XII: Chemistry Chapter 13: Amines Top concepts 1. Amines are regarded as derivatives of ammonia in which one, two or all three hydrogen atoms are replaced by alkyl or aryl group 2. Classification
Chapter 25: The Chemistry of Life: Organic and Biological Chemistry
 Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
CHEM 347 Organic Chemistry II Spring Instructor: Paul Bracher. Quiz # 2
 CHEM 347 Organic Chemistry II Spring 2015 Quiz # 2 Solutions Key Page 1 of 12 CHEM 347 Organic Chemistry II Spring 2015 Instructor: Paul Bracher Quiz # 2 Due: Monday, February 9 th, 2015 2:00 p.m. (in
CHEM 347 Organic Chemistry II Spring 2015 Quiz # 2 Solutions Key Page 1 of 12 CHEM 347 Organic Chemistry II Spring 2015 Instructor: Paul Bracher Quiz # 2 Due: Monday, February 9 th, 2015 2:00 p.m. (in
75. A This is a Markovnikov addition reaction. In these reactions, the pielectrons in the alkene act as a nucleophile. The strongest electrophile will
 71. B SN2 stands for substitution nucleophilic bimolecular. This means that there is a bimolecular rate-determining step. Therefore, the reaction will follow second-order kinetics based on the collision
71. B SN2 stands for substitution nucleophilic bimolecular. This means that there is a bimolecular rate-determining step. Therefore, the reaction will follow second-order kinetics based on the collision
Suggested solutions for Chapter 34
 s for Chapter 34 34 PRBLEM 1 Predict the structure of the product of this Diels- Alder reaction. C 2 +? 3 Si Can you deal with a moderately complicated Diels- Alder? The diene is electron- rich and will
s for Chapter 34 34 PRBLEM 1 Predict the structure of the product of this Diels- Alder reaction. C 2 +? 3 Si Can you deal with a moderately complicated Diels- Alder? The diene is electron- rich and will
Examples of Substituted Benzenes
 Organic Chemistry 5 th Edition Paula Yurkanis Bruice Examples of Substituted Benzenes Chapter 15 Reactions of Substituted Benzenes Irene Lee Case Western Reserve University Cleveland, OH 2007, Prentice
Organic Chemistry 5 th Edition Paula Yurkanis Bruice Examples of Substituted Benzenes Chapter 15 Reactions of Substituted Benzenes Irene Lee Case Western Reserve University Cleveland, OH 2007, Prentice
Chapter 16: Aromatic Compounds
 Chamras Chemistry 106 Lecture otes xamination 2 Materials Chapter 16: Aromatic Compounds Benzene, the Most Commonly Known Aromatic Compound: The aromatic nature of benzene stabilizes it 36 kcal.mol 1.
Chamras Chemistry 106 Lecture otes xamination 2 Materials Chapter 16: Aromatic Compounds Benzene, the Most Commonly Known Aromatic Compound: The aromatic nature of benzene stabilizes it 36 kcal.mol 1.
Exam 1 (Monday, July 6, 2015)
 Chem 231 Summer 2015 Assigned Homework Problems Last updated: Friday, July 24, 2015 Problems Assigned from Essential Organic Chemistry, 2 nd Edition, Paula Yurkanis Bruice, Prentice Hall, New York, NY,
Chem 231 Summer 2015 Assigned Homework Problems Last updated: Friday, July 24, 2015 Problems Assigned from Essential Organic Chemistry, 2 nd Edition, Paula Yurkanis Bruice, Prentice Hall, New York, NY,
Chemistry 3720 Old Exams. Practice Exams & Keys
 Chemistry 3720 ld Exams Practice Exams & Keys 2015-17 Spring 2017 Page File 3 Spring 2017 Exam 1 10 Spring 2017 Exam 1 Key 16 Spring 2017 Exam 2 23 Spring 2017 Exam 2 Key 29 Spring 2017 Exam 3 36 Spring
Chemistry 3720 ld Exams Practice Exams & Keys 2015-17 Spring 2017 Page File 3 Spring 2017 Exam 1 10 Spring 2017 Exam 1 Key 16 Spring 2017 Exam 2 23 Spring 2017 Exam 2 Key 29 Spring 2017 Exam 3 36 Spring
BENZENE AND AROMATIC COMPOUNDS
 BENZENE AND AROMATIC COMPOUNDS The discovery of benzene: 1825 - Michael Faraday, empirical formula of C 1834 - Eilhard Mitscherlich synthesized benzin from gum benzoin, empirical formula C Aromatic The
BENZENE AND AROMATIC COMPOUNDS The discovery of benzene: 1825 - Michael Faraday, empirical formula of C 1834 - Eilhard Mitscherlich synthesized benzin from gum benzoin, empirical formula C Aromatic The
Sp2002 Final Organic II 200pts (Weighted as 300) Good luck ( I believe in you, you can do it, etc.) and please read the questions!
 Sp2002 Final rganic II 200pts (Weighted as 300) NAME: To not have your graded script placed outside my office please check this box Good luck ( I believe in you, you can do it, etc.) and please read the
Sp2002 Final rganic II 200pts (Weighted as 300) NAME: To not have your graded script placed outside my office please check this box Good luck ( I believe in you, you can do it, etc.) and please read the
Learning Guide for Chapter 18 - Aromatic Compounds II
 Learning Guide for Chapter 18 Aromatic Compounds. lectrophilic aromatic substitution ntroduction Mechanism Reagents and Products lectrophiles ffects of stituents FriedelCrafts alkylation and acylation
Learning Guide for Chapter 18 Aromatic Compounds. lectrophilic aromatic substitution ntroduction Mechanism Reagents and Products lectrophiles ffects of stituents FriedelCrafts alkylation and acylation
Chapter 13 Reactions of Arenes Electrophilic Aromatic Substitution
 . 13 hapter 13 eactions of Arenes lectrophilic Aromatic ubstitution lectrophiles add to aromatic rings in a fashion somewhat similar to the addition of electrophiles to alkenes. ecall: 3 4 Y 1 4 2 1 δ
. 13 hapter 13 eactions of Arenes lectrophilic Aromatic ubstitution lectrophiles add to aromatic rings in a fashion somewhat similar to the addition of electrophiles to alkenes. ecall: 3 4 Y 1 4 2 1 δ
Chapter 16- Chemistry of Benzene: Electrophilic Aromatic Substitution
 Chapter 16- Chemistry of Benzene: Electrophilic Aromatic Substitution Ashley Piekarski, Ph.D. Substitution Reactions of Benzene and Its Derivatives Benzene is aroma%c What does aromatic mean? Reac9ons
Chapter 16- Chemistry of Benzene: Electrophilic Aromatic Substitution Ashley Piekarski, Ph.D. Substitution Reactions of Benzene and Its Derivatives Benzene is aroma%c What does aromatic mean? Reac9ons
(Neither an oxidation or reduction: Addition or loss of H +, H 2 O, HX).
 eactions of Alcohols Alcohols are versatile organic compounds since they undergo a wide variety of transformations the majority of which are either oxidation or reduction type reactions. xidation is a
eactions of Alcohols Alcohols are versatile organic compounds since they undergo a wide variety of transformations the majority of which are either oxidation or reduction type reactions. xidation is a
CHEM 303 Organic Chemistry II Problem Set III Chapter 14 Answers
 CHEM 303 rganic Chemistry II Problem Set III Chapter 14 Answers 1) Give the major products of each of the following reactions. If a mixture is expected, identify the major product. + H 3 CHC CHCH 3 H 2
CHEM 303 rganic Chemistry II Problem Set III Chapter 14 Answers 1) Give the major products of each of the following reactions. If a mixture is expected, identify the major product. + H 3 CHC CHCH 3 H 2
CHEM 242 REACTIONS OF ARENES: CHAP 12 ASSIGN ELECTROPHILIC AROMATIC SUBSTITUTION A B C D E
 CHEM 242 REACTIONS OF ARENES: CHAP 12 ASSIGN ELECTROPHILIC AROMATIC SUBSTITUTION 1. Consider carefully the mechanism of the following electrophilic aromatic substitution reaction and indicate which of
CHEM 242 REACTIONS OF ARENES: CHAP 12 ASSIGN ELECTROPHILIC AROMATIC SUBSTITUTION 1. Consider carefully the mechanism of the following electrophilic aromatic substitution reaction and indicate which of
PAPER No. 5: REACTION MECHANISM MODULE No. 2: Types of Organic Reaction Mechanisms
 Subject Chemistry Paper No and Title Module No and Title Module Tag Paper No. 5:Organic Chemistry-II Module No. 2: Overview of different types of Organic Reaction Mechanisms CHE_P5_M2 TABLE OF CONTENTS
Subject Chemistry Paper No and Title Module No and Title Module Tag Paper No. 5:Organic Chemistry-II Module No. 2: Overview of different types of Organic Reaction Mechanisms CHE_P5_M2 TABLE OF CONTENTS
Electrophilic Aromatic Substitution
 Chem 263 ct. 8, 2013 lectrophilic Aromatic Substitution Benzene appears to be a remarkably stable and unreactive compared to alkenes, such as cyclohexene or ethylene, or even alkanes, such as cyclohexane
Chem 263 ct. 8, 2013 lectrophilic Aromatic Substitution Benzene appears to be a remarkably stable and unreactive compared to alkenes, such as cyclohexene or ethylene, or even alkanes, such as cyclohexane
Aryl Halides. Structure
 Aryl Halides Structure Aryl halides are compounds containing halogen attached directly to an aromatic ring. They have the general formula ArX, where Ar is phenyl, substituted phenyl. X= F,Cl,Br,I An aryl
Aryl Halides Structure Aryl halides are compounds containing halogen attached directly to an aromatic ring. They have the general formula ArX, where Ar is phenyl, substituted phenyl. X= F,Cl,Br,I An aryl
Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320
 Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320 I. Isolated, cumulated, and conjugated dienes A diene is any compound with two or C=C's is a diene. Compounds containing more than two
Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320 I. Isolated, cumulated, and conjugated dienes A diene is any compound with two or C=C's is a diene. Compounds containing more than two
Principles and Applications of Photochemistry
 Principles and Applications of Photochemistry Brian Wardle Manchester Metropolitan University, Manchester, UK A John Wiley & Sons, Ltd., Publication Principles and Applications of Photochemistry Principles
Principles and Applications of Photochemistry Brian Wardle Manchester Metropolitan University, Manchester, UK A John Wiley & Sons, Ltd., Publication Principles and Applications of Photochemistry Principles
Lecture Notes Chem 51C S. King. Chapter 20 Introduction to Carbonyl Chemistry; Organometallic Reagents; Oxidation & Reduction
 Lecture Notes Chem 51C S. King Chapter 20 Introduction to Carbonyl Chemistry; rganometallic Reagents; xidation & Reduction I. The Reactivity of Carbonyl Compounds The carbonyl group is an extremely important
Lecture Notes Chem 51C S. King Chapter 20 Introduction to Carbonyl Chemistry; rganometallic Reagents; xidation & Reduction I. The Reactivity of Carbonyl Compounds The carbonyl group is an extremely important
More Nomenclature: Common Names for Selected Aromatic Groups. Aryl = Ar = aromatic group. It is a broad term, and includes any aromatic rings.
 More Nomenclature: Common Names for Selected Aromatic Groups Phenyl group = or Ph = C 6 H 5 = Aryl = Ar = aromatic group. It is a broad term, and includes any aromatic rings. Benzyl = Bn = It has a -CH
More Nomenclature: Common Names for Selected Aromatic Groups Phenyl group = or Ph = C 6 H 5 = Aryl = Ar = aromatic group. It is a broad term, and includes any aromatic rings. Benzyl = Bn = It has a -CH
Chapter 19: Amines. Introduction
 Chapter 19: Amines Chap 19 HW: (be able to name amines); 37, 39, 41, 42, 44, 46, 47, 48, 53-55, 57, 58 Introduction Organic derivatives of ammonia. Many are biologically active. Chap 19: Amines Slide 19-2
Chapter 19: Amines Chap 19 HW: (be able to name amines); 37, 39, 41, 42, 44, 46, 47, 48, 53-55, 57, 58 Introduction Organic derivatives of ammonia. Many are biologically active. Chap 19: Amines Slide 19-2
