Supplementary Figure 1 Irregular arrangement of E,E-8-mer on TMA. STM height images formed when
|
|
|
- Dana Chase
- 5 years ago
- Views:
Transcription
1 Supplementary Figure 1 Irregular arrangement of E,E-8-mer on TMA. STM height images formed when a 5 µl heptanoic acid solution of E,E-8-mer is applied on: (a) a TMA templated HOPG substrate, U s = V, I t = 50 pa; (b) HOPG, U s = V and I t = 58 pa. Unit cell: a = (2.93 ± 0.06) nm, b = (2.95 ± 0.07) nm, θ = (60 ± 3). 1
2 Supplementary Figure 2 Long-range STM height image of E,E-8-mer C 60 on TMA. Tunneling conditions: U s = V and I t = 56 pa. Two unit cells can be determined from the fast Fourier transform (FFT), shown as inset, both corresponding to hexagonal packing, with the axes rotated by 30. The smaller one with two complexes at opposite corners: a 1 = (1.69 ± 0.08) nm, b 1 = (1.70 ± 0.07) nm and 1 = (61 ± 2) ; and a bigger one with four complexes at the four corners: a 2 = (3.0 ± 0.1) nm, b 2 = (3.0 ± 0.2) nm and 2 = (60 ± 1). 2
3 Supplementary Figure 3 Bias dependent imaging of a templated bilayer of E,E-8-mer C 60. STM height images showing a progression of the bias set-point imaging of a templated bilayer of E,E-8-mer C 60. Scan time of each image is approximately one minute. Bright spots assigned to C 60 (b) are not visible in the following image (c). Switching back to positive substrate bias (d), restore the arrangement of moieties as in (a). 3
4 Supplementary Figure 4 Density Functional Theory molecular orbitals of E,E-8-mer C 60. (a) Calculated frontier molecular orbitals. (b) Isosurfaces showing the electron probability distribution 1 ev below and above the HOMO and LUMO respectively. 4
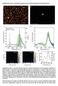
5 Supplementary Figure 5 STS measurements on TMA honeycomb-like network. STS on the bright spots (TMA molecules) and in the pores. The curves are lognormal averaged for measurements taken at the bright spot of the image inset (red curve), which we attributed to higher tunneling regions of the sample, named the aromatic cores of the TMA 1, while the black curve is taken on the dark spot of the image, which corresponds to the pores. The current response to opposite biases is symmetric in the case of the black curve, because the transport occurs direct to the HOPG substrate and/or through the solvent molecules, while in the case of the benzene core of the TMA, the current tends to be higher in absolute values at negative bias. In the scope of electronic transport through an asymmetric gap, this means that the higher occupied molecular orbital (HOMO) of the TMA lies closer to the Fermi-level of HOPG than the lowest occupied molecular orbital (LUMO) 2. Straight lines connect the limits of the error bars for each averaged curve. (STS conditions U s = 0.86 V and I t = 100 pa). 5
6 Supplementary Figure 6 Scattering of averaged I-Vs in Fig. 3. (a) 13 different I-V curves through the rim of the complex E,E-8-mer C 60 (color lines) and the average value (thick black line). (b) 8 different I-V curves through the center of the complex E,E-8-mer C 60 (color lines) and the average value (thick black line). (c) 12 different I-V curves through a monolayer of the macrocycle E,E-8-mer on HOPG (color lines) and the average value (thick black line). A dashed line connects the limits of the error bars for each graph. 6
7 Supplementary Figure 7 Time evolution of the coverage of E,E-8-mer C 60 on TMA. STM height images showing the progression of the coverage with approximately two minutes between each figure. U s = V and I t =65 pa. The lower part of the image is HOPG covered with TMA; after some minutes the islands reached a maximum size. 7
8 Supplementary Figure 8 Island of templated bilayer of E,E-8-mer C 60. (a) STM height image of a double layer from highly concentrated heptanoic acid solution (~10-4 M) of E,E-8-mer C 60 on TMA. (b) Histogram of the image in (a) with its mean values and standard errors. 8
9 Supplementary Figure 9 Scattering of averaged I-Vs in Fig different I-V curves through (a) one monolayer and (b) two monolayers of complex E,E-8-mer C 60 (color lines) and the average value (thick black line). A dashed line connects the limits of the error bars for each graph. 9
10 Supplementary Note 1: Validation of force field parameters for E,E-8-mer The potential energy function of the CGenff follows that of CHARMM 3, which consists of bonded and non-bonded terms, Supplementary Equation (1), where the force constants K for bond, angles and torsions; the equilibrium constants b 0, 0 ; the dihedral phase ; the partial atomic charges q; and the Lennard Jones parameters and need to be determined., 1 cos 4 2 (1) Molecule E,E-8-mer is a novel type of thiophene derivative macrocycle, therefore most of the force field parameters of Supplementary Equation (1) have to be calculated. An automatic assignment of the parameters could be done at paramchem.org 4,5, together with the correspondent penalty for using those parameters by analogy to other molecules. We did the fitting of the missing parameters using the force field toolkit 6 (fftk) implemented in VMD visualization software 7. The molecule E,E-8- mer is divided into reduced molecule for ease of the calculation, especially those involved quantum mechanical calculations of torsional scans, Supplementary Fig. 10 shows the reduced molecules employed. Saturated and unsaturated aliphatic and aromatic hydrogens were automatically assigned the standard charges 0.09, and Similarly, the partial charges of saturated carbons in the groups -CH 3 and -CH 2 were assigned to the standard value 0.27 and 0.18, except for the carbon attached to thiophene units. Reduced compound I and II were used to determine the partial charges of the atoms labeled in Supplementary Fig. 10b. These charges were assigned by analogy to equivalent atoms in reduced compounds III and IV, which were then used to calculate the partial charges of the remaining atoms. Equivalent charges were assigned to chemically equivalent atoms of thiophene, ethylene and acethylene units; for example C3-C5 and C6-C7 in Supplementary Fig. 10. Remaining bond, angle and dihedral parameters were fitted using reduced compounds III and IV. Supplementary Table 1 summarizes the CGenff force field parameter fitting using fftk, only those 10
11 parameters not assigned by analogy are reported. Lennard Jones parameters for each atom type are assigned by analogy. C 60 molecules were assigned atom type CG2R61 (aromatic) and no partial charge. To validate the assigned parameters we calculated the binding energy of the complex E,E-8- mer C 60 by computing the quantity E bind = E 3 (E 2 + E C60 ), where E 2, E C60 and E 3 are the total energies after a 2000 steps minimization of the macrocycle E,E-8-mer, C 60 and the complex E,E-8- mer C 60 respectively. The formation of the complex is favored by (10±5) kcal/mol, which is fair agreement with enthalpy of complexation estimated by van t Hoff plot from binding constant in toluene-d 8 solutions, (5.5±0.4) kcal/mol 8. 11
12 Supplementary Figure 10 Reduced molecules used for the parametrization of molecule E,E-8-mer. 12
13 Supplementary Table 1 Partial atomic charges of selected atoms in Supplementary Fig. 11 and CGenFF force field parameters using fftk. Force constants K b, K and K are given in kcal mol -1, b 0 in Å, 0 and in degrees. Partial atomic charges Bonds Angles Dihedrals Atom charge Bond K b b 0 Angle K 0 Dihedral K n S C C C C3 C S C6 C C8 C C C C CG2R51 CG1T1 CG2R51 CG2DC SG2R50 CG2R51 CG1T1 CG2R51 CG2R51 CG1T1 CG2R51 CG1T1 CG1T1 SG2R50 CG2R51 CGDC1 CG2R51 CG2R51 CGDC1 CG2R51 CG2DC1 HGA4 CG2R51 CG2DC1 CG2DC1 CG2R51 CG321 CG CG2R51 CG2DC1 CG2DC1 HGA4 CG2R51 CG2R51 CG2R51 CG2DC1 CG2R51 SG2R50 CG2R51 CG2DC1 CG2R51 CG2R51 CG2DC1 HGA4 SG2R50 CG2R51 CG2DC1 HGA4 SG2R50 CG2R51 CG2DC1 CG2DC1 CG2R51 CG2R51 CG2DC1 CGDC1 CG2R51 CG2DC1 CG2DC1 CG2R51 CG2DC1 CG2R51 CG2R51 CG321 CG2R51 CG2R51 CG2R51 CG1T1 CG2R51 SG2R50 CG2R51 CG1T1 CG321 CG2R51 CG2R51 SG2R50 CG2R51 CG2R51 CG2R51 CG321 CG321 CG2R51 CG2R51 CG1T1 CG321 CG2R51 CG2R51 CG
14 Supplementary Note 2: MD simulation of monolayer and bilayer of templated E,E-8- mer C 60 on graphene A graphene slab was constructed to simulate the HOPG substrate as it has been shown to describe well the adsorption of polyaromatics onto this substrate 9. One unit cell of templated complex E,E-8-mer C 60 was built up on top of the 24 TMA honeycomb network as shown in Supplementary Fig. 11a. The hexagonal unit cell has equal cell constants a 3 = b 3 = 3 a TMA = Å, making the ( 3 3)R30 superstructure. In the initial configuration, all the alkyl chains are parallel to the substrate plane. After running 2 ns of MD simulation at 300 K, the system adopts the configuration showed in Supplementary Fig. 11b. Analysis of the last 50 frames of the simulation (1 ns) showed that the alkyl chain are uneven distributed between backfolded upwards (41%), downwards (26%) and in-plane (33%). Similarly, a bilayer of complex E,E-8-mer C 60 is built above the first monolayer as depicted in Supplementary Fig. 12a. After 2 ns of MD simulation at 300 K, the distribution of the alkyl chains changes to more butyl groups in-plane, 40% on the first ML and 41% on the second ML; backfolded upwards, 28% on first ML and 29% on second ML; and downwards, 33% and 30% for first and second ML respectively. The redistribution of the orientation of alkyl chains is due to steric effects by the proximity of the macrocycles of the two monolayers, 4.8 Å (see Fig. 5d in main text), while the mean distance between C 60 molecules is ~7.1 Å. Supplementary Fig. 13 show the evolution of the non-bonded terms of Supplementary Equation (1) for the MD simulations of 1ML and 2ML of complexes E,E-8-mer C 60 on TMA graphene. The decrease of the energy upon formation of the second monolayer is obtained by the difference E vdw2ml + E Elect2ML (E vdw1ml + E Elect1ML ), which for the last nanosecond is (169±30) kcal/mol. Supplementary Fig. 14 shows a STM simulated MD trajectory image by averaging 100 snapshots taking every 20 ps (2 ns MD simulation). Each frame is rendered according to the contribution of each atom to the tunneling current 10, proportional to the expression, where is the current attenuation factor and the height of the molecule/atom. 14
15 Supplementary Figure 11. a. Top and side view of the initial guess for a one monolayer of E,E-8-mer C 60 on top of a TMA network on a graphene slab. b. Top and side view after 2 ns of MD simulations at 300 K. 15
16 Supplementary Figure 12. a. Top and side view of the initial guess for two monolayers of E,E-8-mer C 60 on top of a TMA network on a graphene slab. b. Top and side view after 2 ns of MD simulations at 300 K. 16
17 Supplementary Figure 13. Evolution of non-bonded potential terms for the MD simulations depicted in Supplementary Figs 11 and
18 Supplementary Figure 14. STM simulated images averaged from 2 ns of MD simulation of two templated monolayers of E,E-8-mer C 60. a. First monolayer, unit cell a = (3.1±0.8) nm, b = (3.1±0.9) nm and = (60.5±0.6) b. Second monolayer, unit cell a = (3.1±0.8) nm, b = (3.1±0.8) nm and = (60.7±0.8) 18
19 Supplementary Methods Density functional theory (DFT) calculations The molecular geometries and molecular orbitals of the molecules have been computed by using density functional theory (DFT). Becke three-parameter 11 hybrid exchange functional combined with the Lee Yang Parr 12 correlation functional (B3LYP) was used with the 6-31G++(d,p) basis set for geometry optimization. Gaussian09 13 program, running on an Debian 7 Linux server in 1 Intel Core i processor at 3.40 GHz was used for calculations. Molecular frontier orbitals images were visualized with Gaussview5 software and isosurface here (Supplementary Fig. 4b) and in the main text (Fig. 2a) with VMD 7 software. 19
20 Supplementary References 1. Jäckel, F., Wang, Z., Watson, M. D., Müllen, K. & Rabe, J. P. Nanoscale array of inversely biased molecular rectifiers. Chem. Phys. Lett. 387, (2004). 2. Böhme, T., Simpson, C. D., Müllen, K. & Rabe, J. P. Current Voltage Characteristics of a Homologous Series of Polycyclic Aromatic Hydrocarbons. Chem. Eur. J. 13, (2007). 3. Brooks, B. R. et al. CHARMM: A program for macromolecular energy, minimization, and dynamics calculations. J. Comput. Chem. 4, (1983). 4. Vanommeslaeghe, K. & MacKerell, A. D. Automation of the CHARMM General Force Field (CGenFF) I: Bond Perception and Atom Typing. J. Chem. Inf. Model. 52, (2012). 5. Vanommeslaeghe, K., Raman, E. P. & MacKerell, A. D. Automation of the CHARMM General Force Field (CGenFF) II: Assignment of Bonded Parameters and Partial Atomic Charges. J. Chem. Inf. Model. 52, (2012). 6. Mayne, C. G., Saam, J., Schulten, K., Tajkhorshid, E. & Gumbart, J. C. Rapid parameterization of small molecules using the force field toolkit. J. Comput. Chem. 34, (2013). 7. Humphrey, W., Dalke, A. & Schulten, K. VMD: Visual molecular dynamics. J. Mol. Graph. 14, (1996). 8. Shimizu, H. et al. Synthesis, Structures, and Photophysical Properties of π-expanded Oligothiophene 8-mers and Their Saturn-Like C 60 Complexes. J. Am. Chem. Soc. 137, (2015). 9. Björk, J. et al. Adsorption of Aromatic and Anti-Aromatic Systems on Graphene through π π Stacking. J. Phys. Chem. Lett. 1, (2010). 10. Palma, C.-A. et al. Two-dimensional soft supramolecular networks. Chem. Commun. 51, (2015). 11. Becke, A. D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, (1993). 20
21 12. Lee, C., Yang, W. & Parr, R. G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 37, (1988). 13. Frisch, M. J. et al. Gaussian 09 Revision D
CHAPTER-IV. FT-IR and FT-Raman investigation on m-xylol using ab-initio HF and DFT calculations
 4.1. Introduction CHAPTER-IV FT-IR and FT-Raman investigation on m-xylol using ab-initio HF and DFT calculations m-xylol is a material for thermally stable aramid fibers or alkyd resins [1]. In recent
4.1. Introduction CHAPTER-IV FT-IR and FT-Raman investigation on m-xylol using ab-initio HF and DFT calculations m-xylol is a material for thermally stable aramid fibers or alkyd resins [1]. In recent
SUPPLEMENTAL MATERIAL
 SUPPLEMENTAL MATERIAL Systematic Coarse-Grained Modeling of Complexation between Small Interfering RNA and Polycations Zonghui Wei 1 and Erik Luijten 1,2,3,4,a) 1 Graduate Program in Applied Physics, Northwestern
SUPPLEMENTAL MATERIAL Systematic Coarse-Grained Modeling of Complexation between Small Interfering RNA and Polycations Zonghui Wei 1 and Erik Luijten 1,2,3,4,a) 1 Graduate Program in Applied Physics, Northwestern
Supplementary Fig. 1. Progress of the surface mediated Ullmann coupling reaction using STM at 5 K. Precursor molecules
 Supplementary Fig. 1. Progress of the surface mediated Ullmann coupling reaction using STM at 5 K. Precursor molecules (4-bromo-1-ethyl-2-fluorobenzene) are dosed on a Cu(111) surface and annealed to 80
Supplementary Fig. 1. Progress of the surface mediated Ullmann coupling reaction using STM at 5 K. Precursor molecules (4-bromo-1-ethyl-2-fluorobenzene) are dosed on a Cu(111) surface and annealed to 80
1+2 on GHD (5 µl) Volume 1+2 (µl) 1 on GHD 1+2 on GHD
 1+2 on GHD (20 µl) 1+2 on GHD (15 µl) 1+2 on GHD (10 µl) 1+2 on GHD (5 µl) Volume 1+2 (µl) 1 on GHD 1+2 on GHD Supplementary Figure 1 UV-Vis measurements a. UV-Vis spectroscopy of drop-casted volume of
1+2 on GHD (20 µl) 1+2 on GHD (15 µl) 1+2 on GHD (10 µl) 1+2 on GHD (5 µl) Volume 1+2 (µl) 1 on GHD 1+2 on GHD Supplementary Figure 1 UV-Vis measurements a. UV-Vis spectroscopy of drop-casted volume of
Supplementary Figure 1 Experimental setup for crystal growth. Schematic drawing of the experimental setup for C 8 -BTBT crystal growth.
 Supplementary Figure 1 Experimental setup for crystal growth. Schematic drawing of the experimental setup for C 8 -BTBT crystal growth. Supplementary Figure 2 AFM study of the C 8 -BTBT crystal growth
Supplementary Figure 1 Experimental setup for crystal growth. Schematic drawing of the experimental setup for C 8 -BTBT crystal growth. Supplementary Figure 2 AFM study of the C 8 -BTBT crystal growth
Quantum chemical studies on the structures of some heterocyclic azo disperse dyes
 Quantum chemical studies on the structures of some heterocyclic azo disperse dyes Nesrin Tokay, a* Zeynel Seferoğlu, b Cemil Öğretir, c and Nermin Ertan b a Hacettepe University, Faculty of Science, Chemistry
Quantum chemical studies on the structures of some heterocyclic azo disperse dyes Nesrin Tokay, a* Zeynel Seferoğlu, b Cemil Öğretir, c and Nermin Ertan b a Hacettepe University, Faculty of Science, Chemistry
Structure, Electronic and Nonlinear Optical Properties of Furyloxazoles and Thienyloxazoles
 Journal of Physics: Conference Series PAPER OPEN ACCESS Structure, Electronic and Nonlinear Optical Properties of Furyls and Thienyls To cite this article: Ozlem Dagli et al 6 J. Phys.: Conf. Ser. 77 View
Journal of Physics: Conference Series PAPER OPEN ACCESS Structure, Electronic and Nonlinear Optical Properties of Furyls and Thienyls To cite this article: Ozlem Dagli et al 6 J. Phys.: Conf. Ser. 77 View
A critical approach toward resonance-assistance in the intramolecular hydrogen bond
 Electronic Supplementary Material (ESI) for Photochemical & Photobiological Sciences. This journal is The Royal Society of Chemistry and Owner Societies 2015 Supplementary Information for A critical approach
Electronic Supplementary Material (ESI) for Photochemical & Photobiological Sciences. This journal is The Royal Society of Chemistry and Owner Societies 2015 Supplementary Information for A critical approach
Supporting Information
 Supporting Information Constant ph molecular dynamics reveals ph-modulated binding of two small-molecule BACE1 inhibitors Christopher R. Ellis 1,, Cheng-Chieh Tsai 1,, Xinjun Hou 2, and Jana Shen 1, 1
Supporting Information Constant ph molecular dynamics reveals ph-modulated binding of two small-molecule BACE1 inhibitors Christopher R. Ellis 1,, Cheng-Chieh Tsai 1,, Xinjun Hou 2, and Jana Shen 1, 1
Physisorption Controls the Conformation and. Density of States of an Adsorbed Porphyrin
 Physisorption Controls the Conformation and Density of States of an Adsorbed Porphyrin S.P. Jarvis, S. Taylor, J. D. Baran, D. Thompson, A. Saywell, B. Mangham, N. R. Champness, J. A. Larsson, and P. Moriarty
Physisorption Controls the Conformation and Density of States of an Adsorbed Porphyrin S.P. Jarvis, S. Taylor, J. D. Baran, D. Thompson, A. Saywell, B. Mangham, N. R. Champness, J. A. Larsson, and P. Moriarty
The Force Field Toolkit (fftk)
 Parameterizing Small Molecules Using: The Force Field Toolkit (fftk) Christopher G. Mayne, Emad Tajkhorshid Beckman Institute for Advanced Science and Technology University of Illinois, Urbana-Champaign
Parameterizing Small Molecules Using: The Force Field Toolkit (fftk) Christopher G. Mayne, Emad Tajkhorshid Beckman Institute for Advanced Science and Technology University of Illinois, Urbana-Champaign
Supplementary Note 1 Cleanliness during FM-TERS measurement Initial cleanliness of the whole system (before TERS measurement) can be assured using
 Supplementary Note 1 Cleanliness during FM-TERS measurement Initial cleanliness of the whole system (before TERS measurement) can be assured using the protocol in the Methods sections concerning tip and
Supplementary Note 1 Cleanliness during FM-TERS measurement Initial cleanliness of the whole system (before TERS measurement) can be assured using the protocol in the Methods sections concerning tip and
Excited States Calculations for Protonated PAHs
 52 Chapter 3 Excited States Calculations for Protonated PAHs 3.1 Introduction Protonated PAHs are closed shell ions. Their electronic structure should therefore be similar to that of neutral PAHs, but
52 Chapter 3 Excited States Calculations for Protonated PAHs 3.1 Introduction Protonated PAHs are closed shell ions. Their electronic structure should therefore be similar to that of neutral PAHs, but
Universal Repulsive Contribution to the. Solvent-Induced Interaction Between Sizable, Curved Hydrophobes: Supporting Information
 Universal Repulsive Contribution to the Solvent-Induced Interaction Between Sizable, Curved Hydrophobes: Supporting Information B. Shadrack Jabes, Dusan Bratko, and Alenka Luzar Department of Chemistry,
Universal Repulsive Contribution to the Solvent-Induced Interaction Between Sizable, Curved Hydrophobes: Supporting Information B. Shadrack Jabes, Dusan Bratko, and Alenka Luzar Department of Chemistry,
Supplementary Figure 1: Change of scanning tunneling microscopy (STM) tip state. a, STM tip transited from blurred (the top dark zone) to orbital
 Supplementary Figure 1: Change of scanning tunneling microscopy (STM) tip state. a, STM tip transited from blurred (the top dark zone) to orbital resolvable (the bright zone). b, Zoomedin tip-state changing
Supplementary Figure 1: Change of scanning tunneling microscopy (STM) tip state. a, STM tip transited from blurred (the top dark zone) to orbital resolvable (the bright zone). b, Zoomedin tip-state changing
The experimental work seems to be well carried out and the DFT calculations carefully crafted.
 Reviewers' comments: Reviewer #1 (Remarks to the Author): The manuscript by Maier et al. reports on the on-surface synthesis of 1D and 2D polymers in ultra-high vacuum. A halogenated triphenylamine precursor
Reviewers' comments: Reviewer #1 (Remarks to the Author): The manuscript by Maier et al. reports on the on-surface synthesis of 1D and 2D polymers in ultra-high vacuum. A halogenated triphenylamine precursor
Formation of Halogen Bond-Based 2D Supramolecular Assemblies by Electric Manipulation
 Formation of Halogen Bond-Based 2D Supramolecular Assemblies by Electric Manipulation Qing-Na Zheng, a,b Xuan-He Liu, a,b Ting Chen, a Hui-Juan Yan, a Timothy Cook, c Dong Wang* a, Peter J. Stang, c Li-Jun
Formation of Halogen Bond-Based 2D Supramolecular Assemblies by Electric Manipulation Qing-Na Zheng, a,b Xuan-He Liu, a,b Ting Chen, a Hui-Juan Yan, a Timothy Cook, c Dong Wang* a, Peter J. Stang, c Li-Jun
Supporting Information for Ultra-narrow metallic armchair graphene nanoribbons
 Supporting Information for Ultra-narrow metallic armchair graphene nanoribbons Supplementary Figure 1 Ribbon length statistics. Distribution of the ribbon lengths and the fraction of kinked ribbons for
Supporting Information for Ultra-narrow metallic armchair graphene nanoribbons Supplementary Figure 1 Ribbon length statistics. Distribution of the ribbon lengths and the fraction of kinked ribbons for
Supplementary Information for Solution-Synthesized Chevron Graphene Nanoribbons Exfoliated onto H:Si(100)
 Supplementary Information for Solution-Synthesized Chevron Graphene Nanoribbons Exfoliated onto H:Si(100) Adrian Radocea,, Tao Sun,, Timothy H. Vo, Alexander Sinitskii,,# Narayana R. Aluru,, and Joseph
Supplementary Information for Solution-Synthesized Chevron Graphene Nanoribbons Exfoliated onto H:Si(100) Adrian Radocea,, Tao Sun,, Timothy H. Vo, Alexander Sinitskii,,# Narayana R. Aluru,, and Joseph
Supplementary information
 Supplementary information Supplementary Figure S1STM images of four GNBs and their corresponding STS spectra. a-d, STM images of four GNBs are shown in the left side. The experimental STS data with respective
Supplementary information Supplementary Figure S1STM images of four GNBs and their corresponding STS spectra. a-d, STM images of four GNBs are shown in the left side. The experimental STS data with respective
Self assembly of Carbon Atoms in Interstellar Space and Formation Mechanism of Naphthalene from Small Precursors: A Molecular Dynamics Study
 Self assembly of Carbon Atoms in Interstellar Space and Formation Mechanism of Naphthalene from Small Precursors: A Molecular Dynamics Study Niladri Patra UIC Advisor: Dr. H. R. Sadeghpour ITAMP Large
Self assembly of Carbon Atoms in Interstellar Space and Formation Mechanism of Naphthalene from Small Precursors: A Molecular Dynamics Study Niladri Patra UIC Advisor: Dr. H. R. Sadeghpour ITAMP Large
Self-Assembly of Two-Dimensional Organic Networks Containing Heavy Metals (Pb, Bi) and Preparation of Spin-Polarized Scanning Tunneling Microscope
 MPhil Thesis Defense Self-Assembly of Two-Dimensional Organic Networks Containing Heavy Metals (Pb, Bi) and Preparation of Spin-Polarized Scanning Tunneling Microscope Presented by CHEN Cheng 12 th Aug.
MPhil Thesis Defense Self-Assembly of Two-Dimensional Organic Networks Containing Heavy Metals (Pb, Bi) and Preparation of Spin-Polarized Scanning Tunneling Microscope Presented by CHEN Cheng 12 th Aug.
Supplementary Note 1: Dark field measurements and Scattering properties of NPoM geometries
 Supplementary Note 1: Dark field measurements and Scattering properties of NPoM geometries Supplementary Figure 1: Dark field scattering properties of individual nanoparticle on mirror geometries separated
Supplementary Note 1: Dark field measurements and Scattering properties of NPoM geometries Supplementary Figure 1: Dark field scattering properties of individual nanoparticle on mirror geometries separated
Supporting information for Chemical and Electrochemical. Surfaces: Insights into the Mechanism and Selectivity from DFT.
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supporting information for Chemical and Electrochemical Hydrogenation of CO 2 to hydrocarbons
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supporting information for Chemical and Electrochemical Hydrogenation of CO 2 to hydrocarbons
Trace Solvent as a Predominant Factor to Tune Dipeptide. Self-Assembly
 Trace Solvent as a Predominant Factor to Tune Dipeptide Self-Assembly Juan Wang,, Kai Liu,,, Linyin Yan,, Anhe Wang, Shuo Bai, and Xuehai Yan *,, National Key Laboratory of Biochemical Engineering, Institute
Trace Solvent as a Predominant Factor to Tune Dipeptide Self-Assembly Juan Wang,, Kai Liu,,, Linyin Yan,, Anhe Wang, Shuo Bai, and Xuehai Yan *,, National Key Laboratory of Biochemical Engineering, Institute
SUPPLEMENTARY INFORMATION
 Simultaneous and coordinated rotational switching of all molecular rotors in a network Y. Zhang, H. Kersell, R. Stefak, J. Echeverria, V. Iancu, U. G. E. Perera, Y. Li, A. Deshpande, K.-F. Braun, C. Joachim,
Simultaneous and coordinated rotational switching of all molecular rotors in a network Y. Zhang, H. Kersell, R. Stefak, J. Echeverria, V. Iancu, U. G. E. Perera, Y. Li, A. Deshpande, K.-F. Braun, C. Joachim,
Notes. Rza Abbasoglu*, Abdurrahman Atalay & Ahmet Şenocak. Indian Journal of Chemistry Vol. 53A, March, 2014, pp
 Indian Journal of Chemistry Vol. 53A, March, 2014, pp. 288-293 Notes DFT investigations of the Diels-Alder reaction of fulvene with tetracyclo[6.2.2.1 3,6.0 2,7 ]trideca-4,9,11-triene- 9,10-dicarboxylic
Indian Journal of Chemistry Vol. 53A, March, 2014, pp. 288-293 Notes DFT investigations of the Diels-Alder reaction of fulvene with tetracyclo[6.2.2.1 3,6.0 2,7 ]trideca-4,9,11-triene- 9,10-dicarboxylic
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/NCHEM.2491 Experimental Realization of Two-dimensional Boron Sheets Baojie Feng 1, Jin Zhang 1, Qing Zhong 1, Wenbin Li 1, Shuai Li 1, Hui Li 1, Peng Cheng 1, Sheng Meng 1,2, Lan Chen 1 and
DOI: 10.1038/NCHEM.2491 Experimental Realization of Two-dimensional Boron Sheets Baojie Feng 1, Jin Zhang 1, Qing Zhong 1, Wenbin Li 1, Shuai Li 1, Hui Li 1, Peng Cheng 1, Sheng Meng 1,2, Lan Chen 1 and
Ion-Gated Gas Separation through Porous Graphene
 Online Supporting Information for: Ion-Gated Gas Separation through Porous Graphene Ziqi Tian, Shannon M. Mahurin, Sheng Dai,*,, and De-en Jiang *, Department of Chemistry, University of California, Riverside,
Online Supporting Information for: Ion-Gated Gas Separation through Porous Graphene Ziqi Tian, Shannon M. Mahurin, Sheng Dai,*,, and De-en Jiang *, Department of Chemistry, University of California, Riverside,
Identifying and Visualizing the Edge Terminations of Single-Layer MoSe2 Island Epitaxially Grown on Au(111)
 Supporting Information Identifying and Visualizing the Edge Terminations of Single-Layer MoSe2 Island Epitaxially Grown on Au(111) Jianchen Lu, De-Liang Bao, Kai Qian, Shuai Zhang, Hui Chen, Xiao Lin*,
Supporting Information Identifying and Visualizing the Edge Terminations of Single-Layer MoSe2 Island Epitaxially Grown on Au(111) Jianchen Lu, De-Liang Bao, Kai Qian, Shuai Zhang, Hui Chen, Xiao Lin*,
Supplementary Figure S1. STM image of monolayer graphene grown on Rh (111). The lattice
 Supplementary Figure S1. STM image of monolayer graphene grown on Rh (111). The lattice mismatch between graphene (0.246 nm) and Rh (111) (0.269 nm) leads to hexagonal moiré superstructures with the expected
Supplementary Figure S1. STM image of monolayer graphene grown on Rh (111). The lattice mismatch between graphene (0.246 nm) and Rh (111) (0.269 nm) leads to hexagonal moiré superstructures with the expected
Supporting Information Dynamics and Structure of Monolayer Polymer Crystallites on Graphene
 Supporting Information Dynamics and Structure of Monolayer Polymer Crystallites on Graphene Max Gulde,*, Anastassia N. Rissanou, Vagelis Harmandaris,*,, Marcus Müller, Sascha Schäfer, Claus Ropers 4th
Supporting Information Dynamics and Structure of Monolayer Polymer Crystallites on Graphene Max Gulde,*, Anastassia N. Rissanou, Vagelis Harmandaris,*,, Marcus Müller, Sascha Schäfer, Claus Ropers 4th
Modeling Ultrafast Deactivation in Oligothiophenes via Nonadiabatic Dynamics
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2015 Supplementary Data for Modeling Ultrafast Deactivation in Oligothiophenes via Nonadiabatic
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2015 Supplementary Data for Modeling Ultrafast Deactivation in Oligothiophenes via Nonadiabatic
Department of Materials Science and Engineering, Research Institute of Advanced
 Supporting Information High Energy Organic Cathode for Sodium Rechargeable Batteries Haegyeom Kim 1, Ji Eon Kwon 2, Byungju Lee 1, Jihyun Hong 1, Minah Lee 3, Soo Young Park 2*, and Kisuk Kang 1,4 * 1.
Supporting Information High Energy Organic Cathode for Sodium Rechargeable Batteries Haegyeom Kim 1, Ji Eon Kwon 2, Byungju Lee 1, Jihyun Hong 1, Minah Lee 3, Soo Young Park 2*, and Kisuk Kang 1,4 * 1.
Theory of doping graphene
 H. Pinto, R. Jones School of Physics, University of Exeter, EX4 4QL, Exeter United Kingdom May 25, 2010 Graphene Graphene is made by a single atomic layer of carbon atoms arranged in a honeycomb lattice.
H. Pinto, R. Jones School of Physics, University of Exeter, EX4 4QL, Exeter United Kingdom May 25, 2010 Graphene Graphene is made by a single atomic layer of carbon atoms arranged in a honeycomb lattice.
Type of file: PDF Title of file for HTML: Peer Review File Description:
 Type of file: PDF Title of file for HTML: Supplementary Information Description: Supplementary Figures, Supplementary Table, Supplementary Notes and Supplementary References. Type of file: PDF Title of
Type of file: PDF Title of file for HTML: Supplementary Information Description: Supplementary Figures, Supplementary Table, Supplementary Notes and Supplementary References. Type of file: PDF Title of
CHAPTER 2. Structure and Reactivity: Acids and Bases, Polar and Nonpolar Molecules
 CHAPTER 2 Structure and Reactivity: Acids and Bases, Polar and Nonpolar Molecules 2-1 Kinetics and Thermodynamics of Simple Chemical Processes Chemical thermodynamics: Is concerned with the extent that
CHAPTER 2 Structure and Reactivity: Acids and Bases, Polar and Nonpolar Molecules 2-1 Kinetics and Thermodynamics of Simple Chemical Processes Chemical thermodynamics: Is concerned with the extent that
Supplementary Figures
 Supplementary Figures Supplementary Figure 1: Microstructure, morphology and chemical composition of the carbon microspheres: (a) A SEM image of the CM-NFs; and EDS spectra of CM-NFs (b), CM-Ns (d) and
Supplementary Figures Supplementary Figure 1: Microstructure, morphology and chemical composition of the carbon microspheres: (a) A SEM image of the CM-NFs; and EDS spectra of CM-NFs (b), CM-Ns (d) and
Hydrogenated Graphene
 Hydrogenated Graphene Stefan Heun NEST, Istituto Nanoscienze-CNR and Scuola Normale Superiore Pisa, Italy Outline Epitaxial Graphene Hydrogen Chemisorbed on Graphene Hydrogen-Intercalated Graphene Outline
Hydrogenated Graphene Stefan Heun NEST, Istituto Nanoscienze-CNR and Scuola Normale Superiore Pisa, Italy Outline Epitaxial Graphene Hydrogen Chemisorbed on Graphene Hydrogen-Intercalated Graphene Outline
Experiment Section Fig. S1 Fig. S2
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Supplementary Materials Experiment Section The STM experiments were carried out in an ultrahigh
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Supplementary Materials Experiment Section The STM experiments were carried out in an ultrahigh
Learning to Use Scigress Wagner, Eugene P. (revised May 15, 2018)
 Learning to Use Scigress Wagner, Eugene P. (revised May 15, 2018) Abstract Students are introduced to basic features of Scigress by building molecules and performing calculations on them using semi-empirical
Learning to Use Scigress Wagner, Eugene P. (revised May 15, 2018) Abstract Students are introduced to basic features of Scigress by building molecules and performing calculations on them using semi-empirical
Supporting Information for Solid-liquid Thermal Transport and its Relationship with Wettability and the Interfacial Liquid Structure
 Supporting Information for Solid-liquid Thermal Transport and its Relationship with Wettability and the Interfacial Liquid Structure Bladimir Ramos-Alvarado, Satish Kumar, and G. P. Peterson The George
Supporting Information for Solid-liquid Thermal Transport and its Relationship with Wettability and the Interfacial Liquid Structure Bladimir Ramos-Alvarado, Satish Kumar, and G. P. Peterson The George
Visualizing the evolution from the Mott insulator to a charge-ordered insulator in lightly doped cuprates
 Visualizing the evolution from the Mott insulator to a charge-ordered insulator in lightly doped cuprates Peng Cai 1, Wei Ruan 1, Yingying Peng, Cun Ye 1, Xintong Li 1, Zhenqi Hao 1, Xingjiang Zhou,5,
Visualizing the evolution from the Mott insulator to a charge-ordered insulator in lightly doped cuprates Peng Cai 1, Wei Ruan 1, Yingying Peng, Cun Ye 1, Xintong Li 1, Zhenqi Hao 1, Xingjiang Zhou,5,
Name Date Class HYDROCARBONS
 22.1 HYDROCARBONS Section Review Objectives Describe the relationship between number of valence electrons and bonding in carbon Define and describe alkanes Relate the polarity of hydrocarbons to their
22.1 HYDROCARBONS Section Review Objectives Describe the relationship between number of valence electrons and bonding in carbon Define and describe alkanes Relate the polarity of hydrocarbons to their
SUPPLEMENTARY INFORMATION
 Real-space imaging of interfacial water with submolecular resolution Jing Guo, Xiangzhi Meng, Ji Chen, Jinbo Peng, Jiming Sheng, Xinzheng Li, Limei Xu, Junren Shi, Enge Wang *, Ying Jiang * International
Real-space imaging of interfacial water with submolecular resolution Jing Guo, Xiangzhi Meng, Ji Chen, Jinbo Peng, Jiming Sheng, Xinzheng Li, Limei Xu, Junren Shi, Enge Wang *, Ying Jiang * International
240 Chem. Aromatic Compounds. Chapter 6
 240 Chem Aromatic Compounds Chapter 6 1 The expressing aromatic compounds came to mean benzene and derivatives of benzene. Structure of Benzene: Resonance Description C 6 H 6 1.It contains a six-membered
240 Chem Aromatic Compounds Chapter 6 1 The expressing aromatic compounds came to mean benzene and derivatives of benzene. Structure of Benzene: Resonance Description C 6 H 6 1.It contains a six-membered
Chapter 6. Electronic spectra and HOMO-LUMO studies on Nickel, copper substituted Phthalocyanine for solar cell applications
 Chapter 6 Electronic spectra and HOMO-LUMO studies on Nickel, copper substituted Phthalocyanine for solar cell applications 6.1 Structures of Ni, Cu substituted Phthalocyanine Almost all of the metals
Chapter 6 Electronic spectra and HOMO-LUMO studies on Nickel, copper substituted Phthalocyanine for solar cell applications 6.1 Structures of Ni, Cu substituted Phthalocyanine Almost all of the metals
Supporting Information for
 Supporting Information for Odd-Even Effects in Chiral Phase Transition at the Liquid/Solid Interface Hai Cao, a Kazukuni Tahara, bc Shintaro Itano, b Yoshito Tobe b* and Steven De Feyter a* a Division
Supporting Information for Odd-Even Effects in Chiral Phase Transition at the Liquid/Solid Interface Hai Cao, a Kazukuni Tahara, bc Shintaro Itano, b Yoshito Tobe b* and Steven De Feyter a* a Division
Electronic Supporting Information for
 Electronic Supplementary Material (ESI) for Materials Horizons. This journal is The Royal Society of Chemistry 2015 Electronic Supporting Information for Probing the Energy Levels in Hole-doped Molecular
Electronic Supplementary Material (ESI) for Materials Horizons. This journal is The Royal Society of Chemistry 2015 Electronic Supporting Information for Probing the Energy Levels in Hole-doped Molecular
Macrocyclic Oligofurans: A Computational Study
 Supporting Information Macrocyclic Oligofurans: A Computational Study Or Dishi and Ori Gidron* Institute of Chemistry, The Hebrew University of Jerusalem, Edmond J. Safra Campus, Givat Ram, Jerusalem,
Supporting Information Macrocyclic Oligofurans: A Computational Study Or Dishi and Ori Gidron* Institute of Chemistry, The Hebrew University of Jerusalem, Edmond J. Safra Campus, Givat Ram, Jerusalem,
Quantum Mechanical Study on the Adsorption of Drug Gentamicin onto γ-fe 2
 ORIENTAL JOURNAL OF CHEMISTRY An International Open Free Access, Peer Reviewed Research Journal www.orientjchem.org ISSN: 0970-00 X CODEN: OJCHEG 015, Vol. 31, No. (3): Pg. 1509-1513 Quantum Mechanical
ORIENTAL JOURNAL OF CHEMISTRY An International Open Free Access, Peer Reviewed Research Journal www.orientjchem.org ISSN: 0970-00 X CODEN: OJCHEG 015, Vol. 31, No. (3): Pg. 1509-1513 Quantum Mechanical
CHAPTER 5. FORMATION OF SAMs CONRTOLLED BY STERIC EFFECTS. The steric effect is an important subject in chemistry. It arises from the fact that
 CHAPTER 5 FRMATIN F SAMs CNRTLLED BY STERIC EFFECTS 5.1 Motivation The steric effect is an important subject in chemistry. It arises from the fact that each atom within a molecule occupies a certain volume
CHAPTER 5 FRMATIN F SAMs CNRTLLED BY STERIC EFFECTS 5.1 Motivation The steric effect is an important subject in chemistry. It arises from the fact that each atom within a molecule occupies a certain volume
In order to determine the energy level alignment of the interface between cobalt and
 SUPPLEMENTARY INFORMATION Energy level alignment of the CuPc/Co interface In order to determine the energy level alignment of the interface between cobalt and CuPc, we have performed one-photon photoemission
SUPPLEMENTARY INFORMATION Energy level alignment of the CuPc/Co interface In order to determine the energy level alignment of the interface between cobalt and CuPc, we have performed one-photon photoemission
Electronic structures of one-dimension carbon nano wires and rings
 IOP Publishing Journal of Physics: Conference Series 61 (2007) 252 256 doi:10.1088/1742-6596/61/1/051 International Conference on Nanoscience and Technology (ICN&T 2006) Electronic structures of one-dimension
IOP Publishing Journal of Physics: Conference Series 61 (2007) 252 256 doi:10.1088/1742-6596/61/1/051 International Conference on Nanoscience and Technology (ICN&T 2006) Electronic structures of one-dimension
Help me understanding the effect of organic acceptor molecules on coinage metals.
 Help me understanding the effect of organic acceptor molecules on coinage metals. Gerold M. Rangger, 1 Oliver T. Hofmann, 1 Anna M. Track, 1 Ferdinand Rissner, 1 Lorenz Romaner, 1,2 Georg Heimel, 2 Benjamin
Help me understanding the effect of organic acceptor molecules on coinage metals. Gerold M. Rangger, 1 Oliver T. Hofmann, 1 Anna M. Track, 1 Ferdinand Rissner, 1 Lorenz Romaner, 1,2 Georg Heimel, 2 Benjamin
CN NC. dha-7. dha-6 R' R. E-vhf (s-trans) E-vhf (s-cis) R CN. Z-vhf (s-cis) Z-vhf (s-trans) R = AcS R' = AcS
 R' R R' R dha-6 dha-7 R' R R' R E-vhf (s-cis) E-vhf (s-trans) R R' R R' Z-vhf (s-cis) Z-vhf (s-trans) R = R' = Supplementary Figure 1 Nomenclature of compounds. Supplementary Figure 2 500 MHz 1 H NMR spectrum
R' R R' R dha-6 dha-7 R' R R' R E-vhf (s-cis) E-vhf (s-trans) R R' R R' Z-vhf (s-cis) Z-vhf (s-trans) R = R' = Supplementary Figure 1 Nomenclature of compounds. Supplementary Figure 2 500 MHz 1 H NMR spectrum
Introduction to Nanotechnology Chapter 5 Carbon Nanostructures Lecture 1
 Introduction to Nanotechnology Chapter 5 Carbon Nanostructures Lecture 1 ChiiDong Chen Institute of Physics, Academia Sinica chiidong@phys.sinica.edu.tw 02 27896766 Carbon contains 6 electrons: (1s) 2,
Introduction to Nanotechnology Chapter 5 Carbon Nanostructures Lecture 1 ChiiDong Chen Institute of Physics, Academia Sinica chiidong@phys.sinica.edu.tw 02 27896766 Carbon contains 6 electrons: (1s) 2,
AB INITIO MODELING OF THE STRUCTURAL DEFECTS IN AMIDES
 Int. J. Chem. Sci.: 9(4), 2011, 1564-1568 ISSN 0972-768X www.sadgurupublications.com AB INITIO MODELING OF THE STRUCTURAL DEFECTS IN AMIDES M. FATHIMA BEGUM, HEMA TRESA VARGHESE a, Y. SHEENA MARY a, C.
Int. J. Chem. Sci.: 9(4), 2011, 1564-1568 ISSN 0972-768X www.sadgurupublications.com AB INITIO MODELING OF THE STRUCTURAL DEFECTS IN AMIDES M. FATHIMA BEGUM, HEMA TRESA VARGHESE a, Y. SHEENA MARY a, C.
Alkanes. Introduction
 Introduction Alkanes Recall that alkanes are aliphatic hydrocarbons having C C and C H bonds. They can be categorized as acyclic or cyclic. Acyclic alkanes have the molecular formula C n H 2n+2 (where
Introduction Alkanes Recall that alkanes are aliphatic hydrocarbons having C C and C H bonds. They can be categorized as acyclic or cyclic. Acyclic alkanes have the molecular formula C n H 2n+2 (where
Australian Journal of Basic and Applied Sciences
 AENSI Journals Australian Journal of Basic and Applied Sciences ISSN:1991-8178 Journal home page: www.ajbasweb.com Theoretical Study for the Effect of Hydroxyl Radical on the Electronic Properties of Cyclobutadiene
AENSI Journals Australian Journal of Basic and Applied Sciences ISSN:1991-8178 Journal home page: www.ajbasweb.com Theoretical Study for the Effect of Hydroxyl Radical on the Electronic Properties of Cyclobutadiene
State Key Laboratory of Molecular Reaction Dynamics, Dalian Institute of Chemical Physics, Chinese Academic of Sciences, Dalian , P. R. China.
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Supplementary Information for: Theoretical exploration of MgH 2 and graphene nano-flake in cyclohexane:
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Supplementary Information for: Theoretical exploration of MgH 2 and graphene nano-flake in cyclohexane:
Additive Perturbed Molecular Assembly in Two- Dimensional Crystals: Differentiating Kinetic and. Thermodynamic Pathways
 Additive Perturbed Molecular Assembly in Two- Dimensional Crystals: Differentiating Kinetic and Thermodynamic Pathways Seokhoon Ahn and Adam J. Matzger* Department of Chemistry and the Macromolecular Science
Additive Perturbed Molecular Assembly in Two- Dimensional Crystals: Differentiating Kinetic and Thermodynamic Pathways Seokhoon Ahn and Adam J. Matzger* Department of Chemistry and the Macromolecular Science
Direct Measurement of Electron Transfer through a Hydrogen Bond
 Supporting Information Direct Measurement of Electron Transfer through a Hydrogen Bond between Single Molecules Tomoaki Nishino,*, Nobuhiko Hayashi, and Phuc T. Bui Nanoscience and Nanotechnology Research
Supporting Information Direct Measurement of Electron Transfer through a Hydrogen Bond between Single Molecules Tomoaki Nishino,*, Nobuhiko Hayashi, and Phuc T. Bui Nanoscience and Nanotechnology Research
Supporting Information
 Supporting Information Ionic Liquid Designed for PEDOT:PSS Conductivity Enhancement Ambroise de Izarra, a,b,1 Seongjin Park, a,1 Jinhee Lee, a Yves Lansac, b,c, * Yun Hee Jang a, * a Department of Energy
Supporting Information Ionic Liquid Designed for PEDOT:PSS Conductivity Enhancement Ambroise de Izarra, a,b,1 Seongjin Park, a,1 Jinhee Lee, a Yves Lansac, b,c, * Yun Hee Jang a, * a Department of Energy
Supplementary Information
 Supplementary Information Supplementary Figure 1: Electronic Kohn-Sham potential profile of a charged monolayer MoTe 2 calculated using PBE-DFT. Plotted is the averaged electronic Kohn- Sham potential
Supplementary Information Supplementary Figure 1: Electronic Kohn-Sham potential profile of a charged monolayer MoTe 2 calculated using PBE-DFT. Plotted is the averaged electronic Kohn- Sham potential
1-amino-9-octadecene, HAuCl 4, hexane, ethanol 55 o C, 16h AuSSs on GO
 Supplementary Figures GO Supplementary Figure S1 1-amino-9-octadecene, HAuCl 4, hexane, ethanol 55 o C, 16h AuSSs on GO Schematic illustration of synthesis of Au square sheets on graphene oxide sheets.
Supplementary Figures GO Supplementary Figure S1 1-amino-9-octadecene, HAuCl 4, hexane, ethanol 55 o C, 16h AuSSs on GO Schematic illustration of synthesis of Au square sheets on graphene oxide sheets.
Supporting Information: Local Electronic Structure of a Single-Layer. Porphyrin-Containing Covalent Organic Framework
 Supporting Information: Local Electronic Structure of a Single-Layer Porphyrin-Containing Covalent Organic Framework Chen Chen 1, Trinity Joshi 2, Huifang Li 3, Anton D. Chavez 4,5, Zahra Pedramrazi 2,
Supporting Information: Local Electronic Structure of a Single-Layer Porphyrin-Containing Covalent Organic Framework Chen Chen 1, Trinity Joshi 2, Huifang Li 3, Anton D. Chavez 4,5, Zahra Pedramrazi 2,
Outline. Introduction: graphene. Adsorption on graphene: - Chemisorption - Physisorption. Summary
 Outline Introduction: graphene Adsorption on graphene: - Chemisorption - Physisorption Summary 1 Electronic band structure: Electronic properties K Γ M v F = 10 6 ms -1 = c/300 massless Dirac particles!
Outline Introduction: graphene Adsorption on graphene: - Chemisorption - Physisorption Summary 1 Electronic band structure: Electronic properties K Γ M v F = 10 6 ms -1 = c/300 massless Dirac particles!
Supplementary Figure 1 Typical angles of the corners of the 2D compact MoSe2 islands.
 1 2 Supplementary Figure 1 Typical angles of the corners of the 2D compact MoSe2 islands. 3 The scale bar at the bottom represents 500 nm. 60, 90, 120 and 150, originated from the inter- 4 junctioning
1 2 Supplementary Figure 1 Typical angles of the corners of the 2D compact MoSe2 islands. 3 The scale bar at the bottom represents 500 nm. 60, 90, 120 and 150, originated from the inter- 4 junctioning
Supplementary Information
 Supplementary Information a b Supplementary Figure 1. Morphological characterization of synthesized graphene. (a) Optical microscopy image of graphene after transfer on Si/SiO 2 substrate showing the array
Supplementary Information a b Supplementary Figure 1. Morphological characterization of synthesized graphene. (a) Optical microscopy image of graphene after transfer on Si/SiO 2 substrate showing the array
Supplementary Information
 Supplementary Information Supplementary Figure S1: Structure and composition of Teflon tape. (a) XRD spectra of original Teflon tape and Teflon tape subjected to annealing at 150 o C under Ar atmosphere.
Supplementary Information Supplementary Figure S1: Structure and composition of Teflon tape. (a) XRD spectra of original Teflon tape and Teflon tape subjected to annealing at 150 o C under Ar atmosphere.
Supplementary information: Topological Properties Determined by Atomic Buckling in Self-Assembled Ultrathin Bi (110)
 Supplementary information: Topological Properties Determined by Atomic Buckling in Self-Assembled Ultrathin Bi (110) Yunhao Lu, *,, Wentao Xu, Mingang Zeng, Guanggeng Yao, Lei Shen, Ming Yang, Ziyu Luo,
Supplementary information: Topological Properties Determined by Atomic Buckling in Self-Assembled Ultrathin Bi (110) Yunhao Lu, *,, Wentao Xu, Mingang Zeng, Guanggeng Yao, Lei Shen, Ming Yang, Ziyu Luo,
2D surface confined nanoporous molecular networks at the liquid-solid interface: a scanning tunneling microscopy study
 2D surface confined nanoporous molecular networks at the liquid-solid interface: a scanning tunneling microscopy study Shengbin Lei Laboratory of Photochemistry and Spectroscopy and INPAC KULeuven Content
2D surface confined nanoporous molecular networks at the liquid-solid interface: a scanning tunneling microscopy study Shengbin Lei Laboratory of Photochemistry and Spectroscopy and INPAC KULeuven Content
Adsorption, desorption, and diffusion on surfaces. Joachim Schnadt Divsion of Synchrotron Radiation Research Department of Physics
 Adsorption, desorption, and diffusion on surfaces Joachim Schnadt Divsion of Synchrotron Radiation Research Department of Physics Adsorption and desorption Adsorption Desorption Chemisorption: formation
Adsorption, desorption, and diffusion on surfaces Joachim Schnadt Divsion of Synchrotron Radiation Research Department of Physics Adsorption and desorption Adsorption Desorption Chemisorption: formation
Theoretical Modeling of Tunneling Barriers in Carbon-based Molecular Electronic Junctions
 Second Revised version, jp-2014-09838e Supporting Information Theoretical Modeling of Tunneling Barriers in Carbon-based Molecular Electronic Junctions Mykola Kondratenko 1,2, Stanislav R. Stoyanov 1,3,4,
Second Revised version, jp-2014-09838e Supporting Information Theoretical Modeling of Tunneling Barriers in Carbon-based Molecular Electronic Junctions Mykola Kondratenko 1,2, Stanislav R. Stoyanov 1,3,4,
Supplementary Information for Electronic signature of the instantaneous asymmetry in the first coordination shell in liquid water
 Supplementary Information for Electronic signature of the instantaneous asymmetry in the first coordination shell in liquid water Thomas D. Kühne 1, 2 and Rustam Z. Khaliullin 1, 1 Institute of Physical
Supplementary Information for Electronic signature of the instantaneous asymmetry in the first coordination shell in liquid water Thomas D. Kühne 1, 2 and Rustam Z. Khaliullin 1, 1 Institute of Physical
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Method: Epitaxial graphene was prepared by heating an Ir(111) crystal to 550 K for 100 s under 2 x 10-5 Pa partial pressure of ethylene, followed by a flash anneal to 1420 K 1.
SUPPLEMENTARY INFORMATION Method: Epitaxial graphene was prepared by heating an Ir(111) crystal to 550 K for 100 s under 2 x 10-5 Pa partial pressure of ethylene, followed by a flash anneal to 1420 K 1.
Organic Electronic Devices
 Organic Electronic Devices Week 5: Organic Light-Emitting Devices and Emerging Technologies Lecture 5.5: Course Review and Summary Bryan W. Boudouris Chemical Engineering Purdue University 1 Understanding
Organic Electronic Devices Week 5: Organic Light-Emitting Devices and Emerging Technologies Lecture 5.5: Course Review and Summary Bryan W. Boudouris Chemical Engineering Purdue University 1 Understanding
Aliphatic Hydrocarbons Anthracite alkanes arene alkenes aromatic compounds alkyl group asymmetric carbon Alkynes benzene 1a
 Aliphatic Hydrocarbons Anthracite alkanes arene alkenes aromatic compounds alkyl group asymmetric carbon Alkynes benzene 1a Hard coal, which is high in carbon content any straight-chain or branched-chain
Aliphatic Hydrocarbons Anthracite alkanes arene alkenes aromatic compounds alkyl group asymmetric carbon Alkynes benzene 1a Hard coal, which is high in carbon content any straight-chain or branched-chain
Catalytic Activity of IrO 2 (110) Surface: A DFT study
 Catalytic Activity of IrO 2 (110) Surface: A DFT study Jyh-Chiang Jiang Department of Chemical Engineering, National Taiwan University of Science and Technology (NTUST) NCTS-NCKU 9/7, 2010 Computational
Catalytic Activity of IrO 2 (110) Surface: A DFT study Jyh-Chiang Jiang Department of Chemical Engineering, National Taiwan University of Science and Technology (NTUST) NCTS-NCKU 9/7, 2010 Computational
DFT STUDY OF Co 2+ AND Fe 2+ - URACIL COMPLEXES IN THE GAS PHASE
 INTERNATIONAL JOURNAL OF RESEARCH IN PHARMACY AND CHEMISTRY Available online at www.ijrpc.com Research Article DFT STUDY OF Co 2+ AND Fe 2+ - URACIL COMPLEXES IN THE GAS PHASE Raghab Parajuli* Department
INTERNATIONAL JOURNAL OF RESEARCH IN PHARMACY AND CHEMISTRY Available online at www.ijrpc.com Research Article DFT STUDY OF Co 2+ AND Fe 2+ - URACIL COMPLEXES IN THE GAS PHASE Raghab Parajuli* Department
Molecular Orbital Theory. Molecular Orbital Theory: Electrons are located in the molecule, not held in discrete regions between two bonded atoms
 Molecular Orbital Theory Valence Bond Theory: Electrons are located in discrete pairs between specific atoms Molecular Orbital Theory: Electrons are located in the molecule, not held in discrete regions
Molecular Orbital Theory Valence Bond Theory: Electrons are located in discrete pairs between specific atoms Molecular Orbital Theory: Electrons are located in the molecule, not held in discrete regions
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Transforming Fullerene into Graphene Quantum Dots Jiong Lu, Pei Shan Emmeline Yeo, Chee Kwan Gan, Ping Wu and Kian Ping Loh email: chmlohkp@nus.edu.sg Contents Figure S1 Transformation
SUPPLEMENTARY INFORMATION Transforming Fullerene into Graphene Quantum Dots Jiong Lu, Pei Shan Emmeline Yeo, Chee Kwan Gan, Ping Wu and Kian Ping Loh email: chmlohkp@nus.edu.sg Contents Figure S1 Transformation
Molecular Modeling and Conformational Analysis with PC Spartan
 Molecular Modeling and Conformational Analysis with PC Spartan Introduction Molecular modeling can be done in a variety of ways, from using simple hand-held models to doing sophisticated calculations on
Molecular Modeling and Conformational Analysis with PC Spartan Introduction Molecular modeling can be done in a variety of ways, from using simple hand-held models to doing sophisticated calculations on
Temperature induced structural phase transitions in a twodimensional
 Temperature induced structural phase transitions in a twodimensional self-assembled network Matthew O. Blunt*, Jinne Adisoejoso, Kazukuni Tahara, Keisuke Katayama, Mark Van der Auweraer*, Yoshito Tobe*,
Temperature induced structural phase transitions in a twodimensional self-assembled network Matthew O. Blunt*, Jinne Adisoejoso, Kazukuni Tahara, Keisuke Katayama, Mark Van der Auweraer*, Yoshito Tobe*,
Lesmahagow High School CfE Advanced Higher Chemistry. Unit 2 Organic Chemistry and Instrumental Analysis. Molecular Orbitals and Structure
 Lesmahagow High School CfE Advanced Higher Chemistry Unit 2 Organic Chemistry and Instrumental Analysis Molecular Orbitals and Structure 1 Molecular Orbitals Orbitals can be used to explain the bonding
Lesmahagow High School CfE Advanced Higher Chemistry Unit 2 Organic Chemistry and Instrumental Analysis Molecular Orbitals and Structure 1 Molecular Orbitals Orbitals can be used to explain the bonding
Supplementary material. From cellulose to kerogen: molecular simulation. of a geological process
 Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2017 Supplementary material From cellulose to kerogen: molecular simulation of a geological
Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2017 Supplementary material From cellulose to kerogen: molecular simulation of a geological
The Boron Buckyball has an Unexpected T h Symmetry
 The Boron Buckyball has an Unexpected T h Symmetry G. Gopakumar, Minh Tho Nguyen, and Arnout Ceulemans* Department of Chemistry and Institute for Nanoscale Physics and Chemistry, University of Leuven,
The Boron Buckyball has an Unexpected T h Symmetry G. Gopakumar, Minh Tho Nguyen, and Arnout Ceulemans* Department of Chemistry and Institute for Nanoscale Physics and Chemistry, University of Leuven,
Supporting Material for. Microscopic origin of gating current fluctuations in a potassium channel voltage sensor
 Supporting Material for Microscopic origin of gating current fluctuations in a potassium channel voltage sensor J. Alfredo Freites, * Eric V. Schow, * Stephen H. White, and Douglas J. Tobias * * Department
Supporting Material for Microscopic origin of gating current fluctuations in a potassium channel voltage sensor J. Alfredo Freites, * Eric V. Schow, * Stephen H. White, and Douglas J. Tobias * * Department
Quantum Chemistry. NC State University. Lecture 5. The electronic structure of molecules Absorption spectroscopy Fluorescence spectroscopy
 Quantum Chemistry Lecture 5 The electronic structure of molecules Absorption spectroscopy Fluorescence spectroscopy NC State University 3.5 Selective absorption and emission by atmospheric gases (source:
Quantum Chemistry Lecture 5 The electronic structure of molecules Absorption spectroscopy Fluorescence spectroscopy NC State University 3.5 Selective absorption and emission by atmospheric gases (source:
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/3/10/e1701661/dc1 Supplementary Materials for Defect passivation of transition metal dichalcogenides via a charge transfer van der Waals interface Jun Hong Park,
advances.sciencemag.org/cgi/content/full/3/10/e1701661/dc1 Supplementary Materials for Defect passivation of transition metal dichalcogenides via a charge transfer van der Waals interface Jun Hong Park,
STRUCTURAL DEFECTS IN IMIDATES : AN AB INITIO STUDY
 Int. J. Chem. Sci.: 9(4), 2011, 1763-1767 ISSN 0972-768X www.sadgurupublications.com STRUCTURAL DEFECTS IN IMIDATES : AN AB INITIO STUDY M. FATHIMA BEGUM, HEMA TRESA VARGHESE a, Y. SHEENA MARY a, C. YOHANNAN
Int. J. Chem. Sci.: 9(4), 2011, 1763-1767 ISSN 0972-768X www.sadgurupublications.com STRUCTURAL DEFECTS IN IMIDATES : AN AB INITIO STUDY M. FATHIMA BEGUM, HEMA TRESA VARGHESE a, Y. SHEENA MARY a, C. YOHANNAN
NH 3 inversion: Potential energy surfaces and transition states CH342L March 28, 2016
 N 3 inversion: Potential energy surfaces and transition states C342L March 28, 2016 Last week, we used the IR spectrum of ammonia to determine the splitting of energy levels due to inversion of the umbrella
N 3 inversion: Potential energy surfaces and transition states C342L March 28, 2016 Last week, we used the IR spectrum of ammonia to determine the splitting of energy levels due to inversion of the umbrella
Current address: Department of Chemistry, Hong Kong Baptist University, Kowloon Tong, Hong Kong,
 Hydrolysis of Cisplatin - A Metadynamics Study Supporting Information Justin Kai-Chi Lau a and Bernd Ensing* b Department of Chemistry and Applied Bioscience, ETH Zurich, USI Campus, Computational Science,
Hydrolysis of Cisplatin - A Metadynamics Study Supporting Information Justin Kai-Chi Lau a and Bernd Ensing* b Department of Chemistry and Applied Bioscience, ETH Zurich, USI Campus, Computational Science,
Supplementary Information. Reversible Spin Control of Individual Magnetic Molecule by. Hydrogen Atom Adsorption
 Supplementary Information Reversible Spin Control of Individual Magnetic Molecule by Hydrogen Atom Adsorption Liwei Liu 1, Kai Yang 1, Yuhang Jiang 1, Boqun Song 1, Wende Xiao 1, Linfei Li 1, Haitao Zhou
Supplementary Information Reversible Spin Control of Individual Magnetic Molecule by Hydrogen Atom Adsorption Liwei Liu 1, Kai Yang 1, Yuhang Jiang 1, Boqun Song 1, Wende Xiao 1, Linfei Li 1, Haitao Zhou
Supporting Information for. Predicting the Stability of Fullerene Allotropes Throughout the Periodic Table MA 02139
 Supporting Information for Predicting the Stability of Fullerene Allotropes Throughout the Periodic Table Qing Zhao 1, 2, Stanley S. H. Ng 1, and Heather J. Kulik 1, * 1 Department of Chemical Engineering,
Supporting Information for Predicting the Stability of Fullerene Allotropes Throughout the Periodic Table Qing Zhao 1, 2, Stanley S. H. Ng 1, and Heather J. Kulik 1, * 1 Department of Chemical Engineering,
CHEM 344 Molecular Modeling
 CHEM 344 Molecular Modeling The Use of Computational Chemistry to Support Experimental Organic Chemistry Part 1: Molecular Orbital Theory, Hybridization, & Formal Charge * all calculation data obtained
CHEM 344 Molecular Modeling The Use of Computational Chemistry to Support Experimental Organic Chemistry Part 1: Molecular Orbital Theory, Hybridization, & Formal Charge * all calculation data obtained
International Journal of Materials Science ISSN Volume 12, Number 2 (2017) Research India Publications
 HF, DFT Computations and Spectroscopic study of Vibrational frequency, HOMO-LUMO Analysis and Thermodynamic Properties of Alpha Bromo Gamma Butyrolactone K. Rajalakshmi 1 and A.Susila 2 1 Department of
HF, DFT Computations and Spectroscopic study of Vibrational frequency, HOMO-LUMO Analysis and Thermodynamic Properties of Alpha Bromo Gamma Butyrolactone K. Rajalakshmi 1 and A.Susila 2 1 Department of
3rd Advanced in silico Drug Design KFC/ADD Molecular mechanics intro Karel Berka, Ph.D. Martin Lepšík, Ph.D. Pavel Polishchuk, Ph.D.
 3rd Advanced in silico Drug Design KFC/ADD Molecular mechanics intro Karel Berka, Ph.D. Martin Lepšík, Ph.D. Pavel Polishchuk, Ph.D. Thierry Langer, Ph.D. Jana Vrbková, Ph.D. UP Olomouc, 23.1.-26.1. 2018
3rd Advanced in silico Drug Design KFC/ADD Molecular mechanics intro Karel Berka, Ph.D. Martin Lepšík, Ph.D. Pavel Polishchuk, Ph.D. Thierry Langer, Ph.D. Jana Vrbková, Ph.D. UP Olomouc, 23.1.-26.1. 2018
Journal of Computational Methods in Molecular Design, 2013, 3 (1):1-8. Scholars Research Library (
 Journal of Computational Methods in Molecular Design, 2013, 3 (1):1-8 Scholars Research Library (http://scholarsresearchlibrary.com/archive.html) ISSN : 2231-3176 CODEN (USA): JCMMDA Theoretical study
Journal of Computational Methods in Molecular Design, 2013, 3 (1):1-8 Scholars Research Library (http://scholarsresearchlibrary.com/archive.html) ISSN : 2231-3176 CODEN (USA): JCMMDA Theoretical study
