Combining low- and high-energy tandem mass spectra for optimized peptide quantification with isobaric tags
|
|
|
- Gwenda Elliott
- 6 years ago
- Views:
Transcription
1 available at Combining low- and high-energy tandem mass spectra for optimized peptide quantification with isobaric tags Loïc Dayon a, Carla Pasquarello b, Christine Hoogland c, Jean-Charles Sanchez a,b, Alexander Scherl b, a Biomedical Proteomics Group, Department of Structural Biology and Bioinformatics, Faculty of Medicine, University of Geneva, Geneva, Switzerland b Proteomics Core Facility, Faculty of Medicine, University of Geneva, Geneva, Switzerland c Proteome Informatics Group, Swiss Institute of Bioinformatics, Geneva, Switzerland ARTICLE INFO Article history: Received 31 August 2009 Accepted 31 October 2009 Keywords: Quantitation ESI itraq Mass spectrometry Orbitrap Proteomics Tandem mass tags TMT ABSTRACT Isobaric tagging, via TMT or itraq, is widely used in quantitative proteomics. To date, tandem mass spectrometric analysis of isobarically-labeled peptides with hybrid ion trap orbitrap (LTQ-OT) instruments has been mainly carried out with higher-energy C-trap dissociation (HCD) or pulsed q dissociation (PQD). HCD provides good fragmentation of the reporter-ions, but peptide sequence-ion recovery is generally poor compared to collisioninduced dissociation (CID). Herein, we describe an approach where CID and HCD spectra are combined. The approach ensures efficiently both identification and relative quantification of proteins. Tandem mass tags (TMTs) were used to label digests of human plasma and LC- MS/MS was performed with an LTQ-OT instrument. Different HCD collision energies were tested. The benefits to use CID and HCD with respect to HCD alone were demonstrated in terms of number of identifications, subsequent number of quantifiable proteins, and quantification accuracy. A program was developed to merge the peptide sequence-ion m/z range from CID spectra and the reporter-ion m/z range from HCD spectra, and alternatively to separate both spectral data into different files. As parallel CID in the LTQ almost doesn't affect the analysis duty cycle, the procedure should become a standard for quantitative analyses of proteins with isobaric tagging using LTQ-OT instruments Elsevier B.V. All rights reserved. 1. Introduction Isotopic dilution combined with mass spectrometry (MS) has become the method of choice for discovery-based proteomics. Different strategies are used to introduce an isotopic label into a complex biological sample. Typically, the label can be introduced during cell growth [1 3] or during the sample preparation at the protein level (as, for example, ICAT [4]) or at the peptide level. Labeling peptides after proteolytic digestion is mainly performed with isobaric tags such as tandem mass tags (TMTs) or itraq [5 7]. These isobaric compounds are synthesized with heavy and light isotopes to have the same total mass, but to give rise to reporter-ions at different masses after activation and subsequent tandem mass spectrometry (MS/MS or MS2). Activation is generally done using collision-induced dissociation (CID). Relative quantification between protein content of Corresponding author. Proteomics Core Facility, Centre Medical Universitaire, Rue Michel-Servet 1, 1211 Genève 4, Switzerland. Tel.: ; fax: address: Alexander.Scherl@unige.ch (A. Scherl) /$ see front matter 2009 Elsevier B.V. All rights reserved. doi: /j.jprot
2 770 JOURNAL OF PROTEOMICS 73 (2010) different samples is then performed according to the ratio of the reporter-ion abundances in the MS2 spectrum. Isobaric tags have two major advantages compared to quantification in the first stage of mass spectrometry (MS1). First, this mode of operation provides an increased signal to noise ratio of reporter-ions used for quantification due to the removal of chemical noise in the second stage of mass spectrometry. Second, all differentially labeled precursor ions have the same mass. Thus, the MS1 spectrum is less complex, improving data-dependent precursor ion selection for indepth characterization of the sample. On the other hand, relatively high collision energy has to be applied in order to obtain good reporter-ion statistics. However, higher collision energy is not suitable to obtain peptidic backbone ions used to match experimental MS2 spectra to peptides from protein sequence databases. In addition, the relatively low reporterion masses (114 to 117 Da for itraq and 126 to 131 for TMT) are not suited for ion trap instruments. Indeed, these lowmass ions are not stable during the activation step, a principle known as the 1/3rd rule [8]. Recently, hybrid instruments combining the high sensitivity and versatility of linear ion traps with high performance orbitrap (OT) analyzers became available [9,10]. They have rapidly become popular for proteomics experiments. Basically, two activation methods using CID can be used with hybrid OT instruments for the analysis of low-mass fragment ions such as isobaric tag reporter-ions. The so-called pulsed q dissociation or PQD [11,12] consists of activating the precursor ion for a very short time at a high q value, where q is the Mathieu parameter, proportional to the applied radiofrequency voltage in the ion trap. The q value is then lowered before the dissociation of the precursor ion, allowing low-mass ions to be trapped as well. However, the sensitivity of this mode of operation is low compared to traditional CID [13]. Also, the relatively low resolution of the ion trap does not allow precise quantification on closely separated ions such as isobaric tag reporter-ions. Higher-energy C-trap dissociation (HCD) is the second activation method for the analysis of low-mass fragment ions. Activation and dissociation take place between the C-trap, used for the ion storage between the linear ion trap and the OT analyzer, and a supplementary hexapole placed after the C-trap [14]. This fragmentation mode produces higher collision energy that efficiently generates the reporter fragments for quantification. However, this higher collision energy is associated with a decreasing sensitivity due to ion scattering and a lack of backbone fragments useful for peptide identification [15]. Optimized quantitative analysis of phosphopeptides with itraq labeling has been recently reported and it has been shown that classical CID yielded better phosphopeptide sequence identification compared to HCD [16]. The authors proposed to combine both modes for the quantitative analysis of phosphopeptides with itraq. In the same way, this strategy has been applied very recently to study the changes in protein expression in mouse hearts upon transverse aortic constriction with CID followed by HCD of the precursor ions [17]. Better performance with a CID/HCD hybrid acquisition strategy was demonstrated relative to PQD acquisition. The authors demonstrated the superior sensitivity and limit of quantification on digested bovine serum albumin (BSA) and applied their quantification strategy to a complex protein mixture. In this report, we demonstrate that combining CID and HCD activation modes in a linear ion trap orbitrap (LTQ-OT) hybrid instrument allows precise peptide quantification without compromising peptide identification with the linear ion trap analyzer. To do so, a higher-energy HCD spectrum and a low energy CID spectrum were acquired on each selected precursor ion. The CID spectrum was then used for peptide identification at highest sensitivity, and the HCD spectrum was used for precise peptide quantification. TMTs were used as isobaric labeling reagents. Different HCD collision energies were evaluated and their impact on peptide/protein identification and quantification was carefully assessed. A program, accessible over the internet, and compatible with all database search tools for the creation of composite spectra containing both qualitative and quantitative information was developed. Because this program can also dissociate CID and HCD spectra, it can be used for different data analysis schemes, as for example a separated analysis pipeline for CID and HCD spectra. Finally, we showed how this combined acquisition mode affected the instrument duty cycle and the number of quantifiable peptides. 2. Experimental section 2.1. Materials Human plasma, iodoacetamide (IAA, 99%) and tris(2- carboxyethyl) phosphine hydrochloride (TCEP) were purchased from Sigma (St. Louis, MO, USA). Triethylammonium hydrogen carbonate buffer (TEAB) 1 M ph=8.5 and sodium dodecyl sulphate (SDS, 98%) were from Fluka (Buchs, Switzerland). Formic acid (FA, 99%) was from Biosolve (Valkenswaard, the Netherlands). Hydroxylamine solution 50 wt.% in H 2 O(99.999%) was from Aldrich (Milwaukee, WI, USA). Hydrochloric acid (25%) was from Merck (Darmstadt, Germany). Water for chromatography LiChrosolv and acetonitrile Chromasolv for HPLC ( 99.9%) were respectively from Merck and Sigma- Aldrich (Büchs, Switzerland). The duplex Tandem Mass Tags (TMT ) were kindly provided by Proteome Sciences (Frankfurt am Main, Germany) and can be purchased from Thermo Scientific (Rockford, IL, USA). Sequencing grade modified trypsin was from Promega (Madison, WI, USA) Sample preparation Ten µl of plasma was dissolved in 990 µl of TEAB 100 mm adjusted to ph=8 with diluted HCl. To 200 µl of this solution, 2 µl of SDS 1% and 4 µl TCEP 50 mm were added. The reduction was carried out at 60 C for 1 h. Alkylation was performed by adding 2 µl of IAA 400 mm during 30 min in the dark. Twenty µl trypsin 0.2 µg µl 1 (freshly prepared in the above-mentioned TEAB solution) was added and the digestion was proceeded overnight at 37 C. The sample was divided into two identical aliquots. Duplex TMT labeling was achieved for 1 h, after addition of 40.3 µl of TMT duplex reagent in CH 3 CN (i.e., 0.83 mg, mol). One plasma aliquot was labeled with TMT with reporter-ion at m/z=126.1 and the other with TMT with reporter-ion at m/z= Then, 8 µl of hydroxylamine 5% was added in each tube for reaction during 15 min. The differentially TMT-labeled samples were pooled
3 771 in a new tube and the resulting mixture was dried. The sample was desalted with Oasis HLB 1cm 3 (30 mg) extraction cartridges from Waters (Milford, MA, USA) following the manufacturer's instructions. After drying, the sample was cleaned again with a C18 ultramicrospin column (Harvard Apparatus, Holliston, MA, USA), and dried. The sample was dissolved in H 2 O/CH 3 CN/FA 96.9/3/0.1 for MS analysis LC-MS/MS LC-MS/MS was performed on a LTQ orbitrap XL from Thermo Electron (San Jose, CA, USA) equipped with a NanoAcquity HPLC system from Waters. Tryptic peptides (either 2 µg or 0.1 µg) were trapped on a home-made 5 µm 200 Å Magic C18 AQ (Michrom) mm pre-column. Following washing, they were separated on a home-made 5 µm 100 Å Magic C18 AQ (Michrom) mm column with a gravity-pulled emitter. The analytical separation was run for 85 min using a gradient of H 2 O/FA 99.9/0.1 (solvent A) and CH 3 CN/FA 99.9/0.1 (solvent B). The gradient was run as follows: 0 1 min 95% A and 5% B, then to 65% A and 35% B at 55 min, and 20% A and 80% B at 65 min at a flow rate of 220 nl min 1, followed by re-equilibration of the column. For MS1 survey scans, the OT resolution was 60,000 and the ion population with an m/z window from 400 to For MS/MS in the LTQ, the ion population was (isolation width of 2 m/z units). For MS/MS detection in the OT with HCD activation, ion population was set to (isolation width of 4 m/z units), with a resolution of 7500, first mass at m/z=100, and a maximum injection time of 750 ms. A maximum of three precursor ions (most intense) were selected for activation and subsequent MS/MS analysis. CID was performed at 35% of the normalized collision energy (NCE) in all cases. Different collision energies were evaluated for HCD, i.e., 30%, 40%, 50%, 60% and 70% NCE. Control experiments were carried out with CID alone. In this case, the five most intense precursor ions were selected to maximize peptide/ protein identification. The different operating modes are detailed precisely in the Results and discussion part. Briefly, one acquisition mode used HCD alone as fragmentation method and the other performed parallel fragmentation of a given precursor by CID and HCD (CID/HCD mode). All analyses were performed in triplicates. The resulting.mgf files were searched against UniProt- Swiss-Prot database (57.4 of 16-Jun-2009) using Mascot (version 2.2, Matrix Sciences, London, UK) and Phenyx (version 2.6, GeneBio, Geneva, Switzerland). Homo sapiens taxonomy was specified for database searching. Variable amino acid modifications were oxidized methionine. TMT-labeled peptide amino terminus and TMT-labeled lysine ( Da) were set as fixed modifications as well as carbamidomethylation of cysteines. Trypsin was selected as the enzyme, with one potential missed cleavage. As to Mascot, peptide and fragment ion tolerance was respectively 20 ppm and either 0.6 or 0.02 Da when fragments were recorded in the LTQ or in the OT. The significance threshold (p) was set to The Mascot ion score cutoff value was then automatically derived to 30 by the software. In Phenyx, turbo scoring was selected. Falsepositive ratios were estimated using a reverse decoy database [18]. All datasets where searched once in the forward and once in the reverse database. Separate searches were used to keep the database size constant. Protein and peptide score were then set up to maintain the false-positive peptide ratio below 1%. This resulted in a slight overestimation of the false-positive ratio [18]. Parent ion tolerance was set to 20 ppm. The scoring model was selected depending on fragment analysis, either in the LTQ or in the OT. For LTQ scans, the peptide z-score cutoff was set to 5.2. For HCD scans, the peptide z-score cutoffs were set to 9.9, 8.6, 8.2, 7.3, and 7.3 for respective NCE of 30%, 40%, 50%, 60%, and 70% Protein and peptide quantification Reporter-ion abundances (I 126 and I 127 ) were extracted directly from peak lists using dedicated Phenyx exports. No individual protein quantification was carried out. I 126 and I 127 were corrected from isotopic impurities [7,19]. Relative quantification ratios were calculated by division of the corrected abundances of reporter-ions at m/z=127.1 (i 127 ) over those of reporter-ions at m/z=126.1 (i 126 ). Average ratio and mean relative standard deviation (RSD) were calculated on the normalized data i 126 / (i 126 +i 127 )andi 127 /(i 126 +i 127 ) according to the Libra module used in the trans-proteomic pipeline (TPP). In addition, geometric average ratio and RSD were also calculated on the ratios i 126 /i 127 according to the Mascot quantification module Protein and peptide identification Peak lists files (.dta) were generated using the extract_msn. exe embedded in the data analysis software provided by the instrument vendor. Then, the.dta files were merged into a.mgf( mascot generic format ) file using an in-house perl script. CID and HCD data were merged using an in-house perl program as well. This script basically kept, for a given precursor ion, the CID fragments (m/z and ion abundance), and added to their mass list the HCD fragments (m/z and ion abundance) comprised in a specified m/z window (typically in the zone of reporter-ions). An m/z window between 124 and 129 was specified using the duplex TMTs. An additional function of this software was to separate peak lists whose fragments were originally acquired in the LTQ or in the OT in order to extract data coming from HCD fragmentation only. 3. Results and discussion In order to optimize both peptide/protein identification and quantification when analyzing isobarically-tagged peptides with LTQ-OT MS, two MS/MS acquisition modes were evaluated. One was carried out using HCD alone with analysis of the fragment ions in the OT. Different collision energies were assessed. The other mode was performed with concomitant fragmentation by CID and HCD (CID/HCD mode) with fragment ion analysis respectively in the LTQ and OT. In both cases, all MS1 survey scans were acquired in the OT analyzer. A human plasma sample was taken as test sample. After reduction, alkylation and digestion with trypsin, identical plasma samples were labeled with different versions of the duplex TMT and mixed. The pooled sample was analyzed with RP-LC LTQ- OT MS.
4 772 JOURNAL OF PROTEOMICS 73 (2010) CID vs. HCD regarding peptide and protein identification Peptide and protein identification from MS2 spectra depends mainly on mass measurement accuracy of precursor and fragment ions, peptide sequence coverage by fragment ion series and the algorithm and parameters used for the database search. The identification performance of TMT-labeled peptides was evaluated according to the experimental set-up. HCD fragmentation was performed with increasing collision energies ranging from 30% to 70% NCE. The number of unique peptides identified with Mascot was reported as a function of the collision energy in Fig. 1a. This number increased to reach a plateau for energies of 40% and 50% NCE and then it decreased drastically. At 70% NCE, only 8.9% of unique peptides were identified with respect to the maximum number at 50% NCE (i.e., 719 unique peptides). An identical behavior was obtained with 20-times less sample to analyze (i.e., 0.1 µg). The results were slightly different in comparison to a very recent report [17] where collision energies of 30% and 40% were optimal for the analysis of itraq-labeled tryptic peptides of BSA. The data obtained by acquiring CID and HCD spectra together (CID/HCD mode) were merged using a program written in-house. The program added the HCD spectrum window containing the reporter-ion m/z region into the corresponding CID spectrum for each precursor ion. Ion abundance was then normalized so that the base peak of the CID spectrum corresponded to the base peak of the reporter-ion region of the HCD spectrum. Using this procedure the number of identified unique peptides was slightly higher (respectively 745 and 763 unique peptides for HCD collision energy of 50% and 60%) compared to HCD alone at 40% and 50% collision energy (Fig. 1a). Because identifications were obtained from CID data, this number was reproducible whatever HCD collision energy was used. A control comparison with the gold standard method for peptide identification (i.e., CID alone, see Experimental section) stressed that the quantification procedure presented herein reduced the total number of potential identifications as a consequence of the increased scanning duty cycle [15]. With the database search engine Phenyx, HCD spectra acquired at 40% and 50% NCE provided slightly more identifications than those obtained by CID/HCD (whatever HCD collision energy was used; see Supplementary information SI1). Indeed, the optimized Phenyx algorithm was previously shown to take good advantage of the high measured mass accuracy of fragment ions [15]. The total number of matched peptides was also evaluated for both HCD and CID/HCD approaches (Fig. 1b). This number was actually relevant as all these peptides served for protein quantification. In general, the higher number of matched peptides per protein increases the value of the quantitative measurements in terms of accuracy, precision, and overall Fig. 1 Number of unique peptides (a) and total number of peptides (b) identified with Mascot according to the acquisition mode used for the RP-LC LTQ-OT MS analysis. Several collision energies were tested for HCD, ranging from 30% to 70%. Either 2 µg (light bars) or 0.1 µg (dark bars) of TMT-labeled human plasma digest were loaded on LC column. Control CID experiments were carried out and were reported in the upper figure. Fig. 2 Venn diagram of unique peptides identified with Mascot when performing the search on the merged CID/HCD data (i.e., identification carried out on CID spectra) or on the dissociated (i.e., extracted) HCD spectra. Using the CID/HCD acquisition mode with the LTQ-OT mass spectrometer, collision energies of 50% (a) and 60% (b) for HCD were evaluated. Two µg of TMT-labeled human plasma digest was loaded on LC column. The calculation was performed from a common database search of the triplicate MS analyses.
5 773 statistics. A 10% increase of matched peptides was obtained using the CID/HCD mode with respect to HCD at 50% collision energy. The concomitant fragmentation of peptides using the CID/ HCD mode with respective analysis in the LTQ and OT analyzer allowed performing two independent database searches and subsequent relative quantification (i.e., on the merged CID/HCD spectra, and on the extracted HCD spectra). After extracting the HCD peak list from the whole dataset with our program, and performing the database search, different peptides were identified. This is illustrated in the Venn diagram of Fig. 2. Comparison of Fig. 2a and b obtained at respectively 50% and 60% HCD collision energy showed again that HCD performed at 60% was detrimental to the identification process when CID mass spectra were not acquired in parallel. Proportionally more peptide identifications were obtained from the extracted HCD spectra with Phenyx (see Supplementary information SI2). At 50% collision energy, 94 and 98 unique peptides were respectively identified only using the merged CID/HCD and the extracted HCD data. Ultimately, successive database searches with different search engines are therefore a relevant option to increase the total number of peptide/protein identification. The situation where a different peptide sequence was identified from the two CID and HCD scans did not occur in this dataset. Also, the number of accidental CID events [20] (e.g., MS2 spectra with more than one peptide sequence identified after co-isolation and cofragmentation) was low in both CID and HCD spectra (below 10), although a wider isolation window was used for the HCD spectra. In practice, a wider isolation window is necessary to increase the sensitivity of the HCD mode with OT acquisition to a similar level as the CID mode with linear ion trap acquisition [15] Influence of collision energy on the quantification Regarding peptide identification, HCD and CID/HCD could provide equivalent results. Quantification results enabled to refine the selection of an accurate acquisition mode for the analysis of samples modified with isobaric tags such as TMT. Quantification performances were first assessed using HCD alone (collision energy ranging from 30% to 70% NCE). From 30% to 50%, the best quantitative results were obtained at collision energy of 50% in terms of reporter-ion abundance, ratio accuracy and precision (Table 1 and Fig. 3a). At this energy value, the relative standard deviation (RSD) calculated on the relative abundance according to the Libra module used in the trans-proteomic pipeline (TPP) was 3.8%. The RSD value at 30% collision energy was 26.0%, rendering precise quantification impossible (median and average ratios were 1.02, and 1.01 respectively). Although 40% NCE displayed efficient identification results (Fig. 1), the RSD was 17.7%. The results for collision energy of 50%, 60% and 70% are reported in Fig. 3b. Although less data points were obtained when performing HCD alone at 60% and 70% collision energy (as less peptide matches were obtained; see Fig. 1 and Supplementary information SI1), the precision of the relative quantification was superior with respective RSD values of 3.0% and 2.9%. As shown in Table 1, average ratios calculated according to the Libra or Mascot quantification module were identical. The associated RSD and geometric RSD followed similar tendencies. We also investigated if absolute reporter-ion abundance influenced the quantification accuracy. Table 2 shows the Table 1 Summary of the performances of HCD and CID/HCD acquisition modes tested on a human plasma tryptic digest sample labeled with duplex TMT and analyzed with RP-LC LTQ-OT MS (2 µg on column). Data were obtained from the triplicate MS analyses. Number of MS2 spectra d Missing values c /% Ratio geometric RSD b /% Geometric average ratio b ð i127=i126þ Relative abundance mean RSD a /% Abbreviation Median ratio Average ratio a ð i127=ði126 + i127þ= i 126=ði126 + i127þþ Collision energy for HCD/% NCE MS/MS acquisition mode HCD 30 HCD ±85 40 HCD ±55 50 HCD ±76 60 HCD ±77 70 HCD ±7 CID/HCD 50 CID/HCD ±37 60 CID/HCD ±35 Average ratio and mean RSD values were calculated on the relative abundance i126/(i126+i127) and i127/(i126+i127) according to the Libra module used in the trans-proteomic pipeline (TPP). Geometric average ratio and RSD values were calculated on the ratios i127/i126 according to the Mascot quantification module. Missing values corresponded to the percentage of reporter-ion without reporter signal over the total number of reporter-ions. d For CID/HCD, the number of MS2 spectra corresponded to the number of MS2 spectra after combination of CID and HCD spectra together. During acquisition, the actual number of MS/MS events was therefore twice more. a b c
6 774 JOURNAL OF PROTEOMICS 73 (2010) Fig. 3 Distribution representation of Log2(i 127 /i 126 ) as a function of the HCD collision energy. The MS/MS experiments were carried out with HCD alone. The collision energy was varied between 30 and 50% (a), and 50 and 70% (b). Two µg of TMT-labeled human plasma digest was loaded on LC column. Data were obtained from the triplicate MS analyses.
7 775 Table 2 Quantification performances of CID/HCD acquisition mode tested on a human plasma tryptic digest sample labeled with duplex TMT and analyzed with RP-LC LTQ-OT MS (2 µg on column). Data were obtained from the triplicate MS analyses. Experiment Reporter intensity/counts Number of peptides a Average ratio b ð i 127 =ði i 127 Þ= i126 =ði i 127 ÞÞ Relative abundance mean RSD b /% Geometric average ratio c ð i 127 =i 126 Þ Ratio geometric RSD c /% CID/HCD CID/HCD a The number of peptides corresponded to the sum of the peptides from the triplicate MS analyses. b Average ratio and mean RSD values were calculated on the relative abundance i 126 /(i 126 +i 127 ) and i 127 /(i 126 +i 127 ) according to the Libra module used in the trans-proteomic pipeline (TPP). c Geometric average ratio and RSD values were calculated on the ratios i 127 /i 126 according to the Mascot quantification module. measured average peptide ratio and RSD as a function of the absolute reporter-ion count. Basically, the accuracy of the measured ratio and the RSD respectively increased and decreased when the intensity of the reporter-ions increased. Reporter-ions with abundances between and counts provided excellent accuracy and RSD. Collision energy of 60% NCE provided more reporter-ions in this range of intensity. Above counts, the few numbers of data did not allow to evaluate the accuracy and potential saturation issues were observed. These results showed that a high absolute ion count was beneficial for precise quantification. Furthermore, sufficient collision energy prevented obtaining missing values (zero values), reflecting the lack of detection of TMT reporter-ions within the OT analyzer (Table 1). With a lower amount of sample to analyze (i.e., 0.1 µg), the quantification was also better when higher energy was used (see Supplementary information SI4 and SI5).Inthisreport,anionisolationwindowof4m/z units for HCD spectra was chosen. As discussed previously, a larger isolation window increases the number of ions ultimately detected in the Fourier-transform analyzer (in this case the OT). It also reduces the duty cycle by decreasing the ion injection times, and increases the ion statistics [15], and thus the quantification accuracy. On the other hand, opening the precursor ion isolation window might increase the number of accidental CID events and thus perturb the quantification accuracy. Sample fractionation prior to LC-MS/MS analysis should be considered in this case to reduce the number of accidental CID events and thus provide best quantification accuracy. As shown here, HCD energy had to be properly tailored in order to obtain valuable quantitative analysis. When using HCD alone, collision energy of 50% NCE was absolutely required to ensure both valuable identification and quantification Enhanced identification and quantification by merging CID and HCD spectra With the combined CID/HCD acquisition mode, peptide identification through CID spectra could be ensured whatever collision energy was used for HCD. Collision energy of 60% NCE could be implemented without being detrimental to the identification process (Fig. 4a). Besides, quantification performances were enhanced as well. Comparison of Figs. 3b and 4a clearly highlighted the combined benefit in peptide identification rate and quantification accuracy. Higher collision energy provided higher reporter-ion signals (Fig. 4b), preventing acquisition of missing values (Table 1) and potentially allowing higher limits of quantification (see Table 2 and Supplementary information SI3). The combined CID/HCD acquisition mode at 50% NCE offered a good compromise for proteomics discovery experiments. Quantitative data were sufficiently accurate and precise (RSD of 4.5%). Identification could be carried out sequentially on merged CID/HCD and extracted HCD spectra (Fig. 2a and Supplementary information SI2ba), both allowing accurate relative quantification. When accuracy, precision and/or limit of quantification become an issue, higher collision energy should be used for HCD in order to favor the release of TMT reporter-ions. This is especially relevant when targeted proteomics approaches are involved. The combined CID/HCD mode did almost not affect the instrument duty cycle. The parallel acquisition mode (e.g., CID scans were performed in the linear ion trap while the transient signal was recorded in the OT analyzer) allowed acquiring the 3 CID spectra during the MS1 survey scan. Average spectrum acquisition time for an MS1 survey scan was 1.06 s. Average scan time for an MS2 spectrum acquisition in the linear ion trap was 0.27 s. The acquisition of the 3 CID scans in the linear ion trap was therefore performed on the same timescale as the MS1 survey scan. In contrast, the average scan time for a HCD spectrum acquisition was 0.88 s. The 3 HCD acquisitions were performed after the MS1 survey scan. These HCD scans, necessary for quantification with isobaric tags, were therefore responsible for the largest loss in the duty cycle. The total number of selected precursor ions for each acquisition mode is shown in Table 1. In summary, the 3 added CID scans in the linear ion trap were coming almost for free with the
8 776 JOURNAL OF PROTEOMICS 73 (2010) Fig. 4 Distribution representation of Log2(i 127 /i 126 ) as a function of the HCD collision energy (a). The MS/MS experiments were carried out with the CID/HCD acquisition mode. Identification was obtained from CID fragments and quantification from the TMT reporter-ions under HCD. HCD collision energies of 50% and 60% were tested. Abundance of reporter-ions at m/z=127.1 (i 127 ) was represented as a function of reporter-ion abundance at m/z=126.1 (i 126 ) (b). A log scale was used. Two µg of TMT-labeled human plasma digest was loaded on LC column. Data were obtained from the triplicate MS analyses.
9 777 combined method presented here. In average, the number of fragmented precursor ions decreased by less than 20% and this number was easily compensated by the superior sensitivity of the CID spectrum used for peptide identification. 4. Conclusions Concomitant CID and HCD MS/MS acquisition mode was demonstrated to be valuable in terms of identification and quantification when analyzing TMT-labeled peptides with a hybrid linear ion trap orbitrap mass spectrometer. Because CID and HCD can be carried out in parallel respectively with analysis in the LTQ and in the OT, the CID/HCD operating mode almost doesn't affect the overall scanning duty cycle. There is therefore no disadvantage to use it instead of performing HCD alone. The appropriate HCD collision energies for accurate quantification have been emphasized. Informatics tools were developed to deal with the data obtained with the combined CID/HCD mode. One is dedicated to the merging of the CID spectra with corresponding reporter-ion signals from the HCD spectra. The other is used to separate peak lists from CID and HCD spectra in order to proceed to a second identification and quantification round on the extracted HCD data. These programs are available on the expasy website ( In conclusion, the described procedure should rationally become a standard for the quantitative analyses of proteins with isobaric tagging using LTQ-OT mass spectrometers. Acknowledgment The authors thank Proteome Sciences R&D GmbH & Co. KG for the financial support. Appendix A. Supplementary data Supplementary data associated with this article can be found, in the online version, at doi: /j.jprot REFERENCES [1] Ong SE, Blagoev B, Kratchmarova I, Kristensen DB, Steen H, Pandey A, et al. Stable isotope labeling by amino acids in cell culture, SILAC, as a simple and accurate approach to expression proteomics. Mol Cell Proteomics 2002;1: [2] Oda Y, Huang K, Cross FR, Cowburn D, Chait BT. Accurate quantitation of protein expression and site-specific phosphorylation. Proc Natl Acad Sci U S A 1999;96: [3] Zhu H, Pan S, Gu S, Bradbury EM, Chen X. Amino acid residue specific stable isotope labeling for quantitative proteomics. Rapid Commun Mass Spectrom 2002;16: [4]GygiSP,RistB,GerberSA,TurecekF,GelbMH,Aebersold R. Quantitative analysis of complex protein mixtures using isotope-coded affinity tags. Nat Biotechnol 1999;17: [5] Thompson A, Schafer J, Kuhn K, Kienle S, Schwarz J, Schmidt G, et al. Tandem mass tags: a novel quantification strategy for comparative analysis of complex protein mixtures by MS/MS. Anal Chem 2003;75: [6] Ross PL, Huang YN, Marchese JN, Williamson B, Parker K, Hattan S, et al. Multiplexed protein quantitation in Saccharomyces cerevisiae using amine-reactive isobaric tagging reagents. Mol Cell Proteomics 2004;3: [7] Dayon L, Hainard A, Licker V, Turck N, Kuhn K, Hochstrasser DF, et al. Relative quantification of proteins in human cerebrospinal fluids by MS/MS using 6-plex isobaric tags. Anal Chem 2008;80: [8] Louris JN, Cooks RG, Syka JEP, Kelley PE, Stafford GC, Todd JFJ. Instrumentation, applications, and energy deposition in quadrupole ion-trap tandem mass spectrometry. Anal Chem 2002;59: [9] Hu Q, Noll RJ, Li H, Makarov A, Hardman M, Graham Cooks R. The Orbitrap: a new mass spectrometer. J Mass Spectrom 2005;40: [10] Makarov A, Denisov E, Lange O, Horning S. Dynamic range of mass accuracy in LTQ Orbitrap hybrid mass spectrometer. J Am Soc Mass Spectrom 2006;17: [11] Schwartz JC, Syka JP, Quarmby ST. Improving the fundamentals of Msn on 2D ion traps: new ion activation and isolation techniques. 53rd ASMS conference on mass spectrometry, (June 5 9), San Antonio, Texas; [12] Cunningham J, Connell, Glish GL, Burinsky DJ. High amplitude short time excitation: a method to form and detect low mass product ions in a quadrupole ion trap mass spectrometer. J Am Soc Mass Spectrom 2006;17:81 4. [13] Bantscheff M, Boesche M, Eberhard D, Matthieson T, Sweetman G, Kuster B. Robust and sensitive itraq quantification on an LTQ Orbitrap mass spectrometer. Mol Cell Proteomics 2008;7: [14] Olsen JV, Macek B, Lange O, Makarov A, Horning S, Mann M. Higher-energy C-trap dissociation for peptide modification analysis. Nat Methods 2007;4: [15] Scherl A, Shaffer SA, Taylor GK, Hernandez P, Appel RD, Binz PA, et al. On the benefits of acquiring peptide fragment ions at high measured mass accuracy. J Am Soc Mass Spectrom 2008;19: [16] Zhang Y, Ficarro SB, Li S, Marto JA. Optimized Orbitrap HCD for quantitative analysis of phosphopeptides. J Am Soc Mass Spectrom 2009;20: [17] Köcher T, Pichler P, Schutzbier M, Stingl C, Kaul A, Hasenfuss G, et al. High precision quantitative proteomics using itraq on an LTQ Orbitrap: a new mass spectrometric method combining the benefits of all. J Proteome Res 2009;8: [18] Elias JE, Gygi SP. Target-decoy search strategy for increased confidence in large-scale protein identifications by mass spectrometry. Nat Methods 2007;4: [19] Shadforth IP, Dunkley TP, Lilley KS, Bessant C. i-tracker: for quantitative proteomics using itraq. BMC Genomics 2005;6:145. [20] Scherl A, Tsai YS, Shaffer SA, Goodlett DR. Increasing information from shotgun proteomic data by accounting for misassigned precursor ion masses. Proteomics 2008;8:
Improved 6- Plex TMT Quantification Throughput Using a Linear Ion Trap HCD MS 3 Scan Jane M. Liu, 1,2 * Michael J. Sweredoski, 2 Sonja Hess 2 *
 Improved 6- Plex TMT Quantification Throughput Using a Linear Ion Trap HCD MS 3 Scan Jane M. Liu, 1,2 * Michael J. Sweredoski, 2 Sonja Hess 2 * 1 Department of Chemistry, Pomona College, Claremont, California
Improved 6- Plex TMT Quantification Throughput Using a Linear Ion Trap HCD MS 3 Scan Jane M. Liu, 1,2 * Michael J. Sweredoski, 2 Sonja Hess 2 * 1 Department of Chemistry, Pomona College, Claremont, California
Increasing the Multiplexing of Protein Quantitation from 6- to 10-Plex with Reporter Ion Isotopologues
 Increasing the Multiplexing of Protein Quantitation from 6- to 1-Plex with Reporter Ion Isotopologues Rosa Viner, 1 Ryan Bomgarden, 2 Michael Blank, 1 John Rogers 2 1 Thermo Fisher Scientific, San Jose,
Increasing the Multiplexing of Protein Quantitation from 6- to 1-Plex with Reporter Ion Isotopologues Rosa Viner, 1 Ryan Bomgarden, 2 Michael Blank, 1 John Rogers 2 1 Thermo Fisher Scientific, San Jose,
Mass Spectrometry and Proteomics - Lecture 5 - Matthias Trost Newcastle University
 Mass Spectrometry and Proteomics - Lecture 5 - Matthias Trost Newcastle University matthias.trost@ncl.ac.uk Previously Proteomics Sample prep 144 Lecture 5 Quantitation techniques Search Algorithms Proteomics
Mass Spectrometry and Proteomics - Lecture 5 - Matthias Trost Newcastle University matthias.trost@ncl.ac.uk Previously Proteomics Sample prep 144 Lecture 5 Quantitation techniques Search Algorithms Proteomics
Modeling Mass Spectrometry-Based Protein Analysis
 Chapter 8 Jan Eriksson and David Fenyö Abstract The success of mass spectrometry based proteomics depends on efficient methods for data analysis. These methods require a detailed understanding of the information
Chapter 8 Jan Eriksson and David Fenyö Abstract The success of mass spectrometry based proteomics depends on efficient methods for data analysis. These methods require a detailed understanding of the information
Methods for proteome analysis of obesity (Adipose tissue)
 Methods for proteome analysis of obesity (Adipose tissue) I. Sample preparation and liquid chromatography-tandem mass spectrometric analysis Instruments, softwares, and materials AB SCIEX Triple TOF 5600
Methods for proteome analysis of obesity (Adipose tissue) I. Sample preparation and liquid chromatography-tandem mass spectrometric analysis Instruments, softwares, and materials AB SCIEX Triple TOF 5600
Quantitation of a target protein in crude samples using targeted peptide quantification by Mass Spectrometry
 Quantitation of a target protein in crude samples using targeted peptide quantification by Mass Spectrometry Jon Hao, Rong Ye, and Mason Tao Poochon Scientific, Frederick, Maryland 21701 Abstract Background:
Quantitation of a target protein in crude samples using targeted peptide quantification by Mass Spectrometry Jon Hao, Rong Ye, and Mason Tao Poochon Scientific, Frederick, Maryland 21701 Abstract Background:
Label-free protein quantifcation on tandem mass spectra in an ion trapping device. PAK, Hui Song, PASQUARELLO MOSIMANN, Carla, SCHERL, Alexander
 Article Label-free protein quantifcation on tandem mass spectra in an ion trapping device PAK, Hui Song, PASQUARELLO MOSIMANN, Carla, SCHERL, Alexander Abstract Label free quantification using liquid chromatography
Article Label-free protein quantifcation on tandem mass spectra in an ion trapping device PAK, Hui Song, PASQUARELLO MOSIMANN, Carla, SCHERL, Alexander Abstract Label free quantification using liquid chromatography
Quantitation of TMT-Labeled Peptides Using Higher-Energy Collisional Dissociation on the Velos Pro Ion Trap Mass Spectrometer
 Application Note: 520 Quantitation of TMT-Labeled Peptides Using Higher-Energy Collisional Dissociation on the Velos Pro Ion Trap Mass Spectrometer Roger G. Biringer, Julie A. Horner, Rosa Viner, Andreas
Application Note: 520 Quantitation of TMT-Labeled Peptides Using Higher-Energy Collisional Dissociation on the Velos Pro Ion Trap Mass Spectrometer Roger G. Biringer, Julie A. Horner, Rosa Viner, Andreas
NPTEL VIDEO COURSE PROTEOMICS PROF. SANJEEVA SRIVASTAVA
 LECTURE-25 Quantitative proteomics: itraq and TMT TRANSCRIPT Welcome to the proteomics course. Today we will talk about quantitative proteomics and discuss about itraq and TMT techniques. The quantitative
LECTURE-25 Quantitative proteomics: itraq and TMT TRANSCRIPT Welcome to the proteomics course. Today we will talk about quantitative proteomics and discuss about itraq and TMT techniques. The quantitative
Self-assembling covalent organic frameworks functionalized. magnetic graphene hydrophilic biocomposite as an ultrasensitive
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2017 Electronic Supporting Information for: Self-assembling covalent organic frameworks functionalized
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2017 Electronic Supporting Information for: Self-assembling covalent organic frameworks functionalized
Figure S1. Interaction of PcTS with αsyn. (a) 1 H- 15 N HSQC NMR spectra of 100 µm αsyn in the absence (0:1, black) and increasing equivalent
 Figure S1. Interaction of PcTS with αsyn. (a) 1 H- 15 N HSQC NMR spectra of 100 µm αsyn in the absence (0:1, black) and increasing equivalent concentrations of PcTS (100 µm, blue; 500 µm, green; 1.5 mm,
Figure S1. Interaction of PcTS with αsyn. (a) 1 H- 15 N HSQC NMR spectra of 100 µm αsyn in the absence (0:1, black) and increasing equivalent concentrations of PcTS (100 µm, blue; 500 µm, green; 1.5 mm,
Relative quantification using TMT11plex on a modified Q Exactive HF mass spectrometer
 POSTER NOTE 6558 Relative quantification using TMT11plex on a modified mass spectrometer Authors Tabiwang N. Arrey, 1 Rosa Viner, 2 Ryan D. Bomgarden, 3 Eugen Damoc, 1 Markus Kellmann, 1 Thomas Moehring,
POSTER NOTE 6558 Relative quantification using TMT11plex on a modified mass spectrometer Authors Tabiwang N. Arrey, 1 Rosa Viner, 2 Ryan D. Bomgarden, 3 Eugen Damoc, 1 Markus Kellmann, 1 Thomas Moehring,
HR/AM Targeted Peptide Quantification on a Q Exactive MS: A Unique Combination of High Selectivity, High Sensitivity, and High Throughput
 HR/AM Targeted Peptide Quantification on a Q Exactive MS: A Unique Combination of High Selectivity, High Sensitivity, and High Throughput Yi Zhang 1, Zhiqi Hao 1, Markus Kellmann 2 and Andreas FR. Huhmer
HR/AM Targeted Peptide Quantification on a Q Exactive MS: A Unique Combination of High Selectivity, High Sensitivity, and High Throughput Yi Zhang 1, Zhiqi Hao 1, Markus Kellmann 2 and Andreas FR. Huhmer
Peptide Labeling with Isobaric Tags Yields Higher Identification Rates Using itraq 4-Plex Compared to TMT 6-Plex and itraq 8-Plex on LTQ Orbitrap
 Anal. Chem. 2010, 82, 6549 6558 Peptide Labeling with Isobaric Tags Yields Higher Identification Rates Using itraq 4-Plex Compared to TMT 6-Plex and itraq 8-Plex on LTQ Orbitrap Peter Pichler,*, Thomas
Anal. Chem. 2010, 82, 6549 6558 Peptide Labeling with Isobaric Tags Yields Higher Identification Rates Using itraq 4-Plex Compared to TMT 6-Plex and itraq 8-Plex on LTQ Orbitrap Peter Pichler,*, Thomas
Mass spectrometry-based proteomics has become
 FOCUS: THE ORBITRAP Computational Principles of Determining and Improving Mass Precision and Accuracy for Proteome Measurements in an Orbitrap Jürgen Cox and Matthias Mann Proteomics and Signal Transduction,
FOCUS: THE ORBITRAP Computational Principles of Determining and Improving Mass Precision and Accuracy for Proteome Measurements in an Orbitrap Jürgen Cox and Matthias Mann Proteomics and Signal Transduction,
Key questions of proteomics. Bioinformatics 2. Proteomics. Foundation of proteomics. What proteins are there? Protein digestion
 s s Key questions of proteomics What proteins are there? Bioinformatics 2 Lecture 2 roteomics How much is there of each of the proteins? - Absolute quantitation - Stoichiometry What (modification/splice)
s s Key questions of proteomics What proteins are there? Bioinformatics 2 Lecture 2 roteomics How much is there of each of the proteins? - Absolute quantitation - Stoichiometry What (modification/splice)
Improved Precision of itraq and TMT Quantification by an Axial Extraction Field in an Orbitrap HCD Cell
 pubs.acs.org/ac Improved Precision of itraq and TMT Quantification by an Axial Extraction Field in an Orbitrap HCD Cell Peter Pichler,*, Thomas K ocher, Johann Holzmann, Thomas M ohring, Gustav Ammerer,,
pubs.acs.org/ac Improved Precision of itraq and TMT Quantification by an Axial Extraction Field in an Orbitrap HCD Cell Peter Pichler,*, Thomas K ocher, Johann Holzmann, Thomas M ohring, Gustav Ammerer,,
HOWTO, example workflow and data files. (Version )
 HOWTO, example workflow and data files. (Version 20 09 2017) 1 Introduction: SugarQb is a collection of software tools (Nodes) which enable the automated identification of intact glycopeptides from HCD
HOWTO, example workflow and data files. (Version 20 09 2017) 1 Introduction: SugarQb is a collection of software tools (Nodes) which enable the automated identification of intact glycopeptides from HCD
PC235: 2008 Lecture 5: Quantitation. Arnold Falick
 PC235: 2008 Lecture 5: Quantitation Arnold Falick falickam@berkeley.edu Summary What you will learn from this lecture: There are many methods to perform quantitation using mass spectrometry (any method
PC235: 2008 Lecture 5: Quantitation Arnold Falick falickam@berkeley.edu Summary What you will learn from this lecture: There are many methods to perform quantitation using mass spectrometry (any method
Analysis of Labeled and Non-Labeled Proteomic Data Using Progenesis QI for Proteomics
 Analysis of Labeled and Non-Labeled Proteomic Data Using Progenesis QI for Proteomics Lee Gethings, Gushinder Atwal, Martin Palmer, Chris Hughes, Hans Vissers, and James Langridge Waters Corporation, Wilmslow,
Analysis of Labeled and Non-Labeled Proteomic Data Using Progenesis QI for Proteomics Lee Gethings, Gushinder Atwal, Martin Palmer, Chris Hughes, Hans Vissers, and James Langridge Waters Corporation, Wilmslow,
Improved Throughput and Reproducibility for Targeted Protein Quantification Using a New High-Performance Triple Quadrupole Mass Spectrometer
 Improved Throughput and Reproducibility for Targeted Protein Quantification Using a New High-Performance Triple Quadrupole Mass Spectrometer Reiko Kiyonami, Mary Blackburn, Andreas FR Hühme: Thermo Fisher
Improved Throughput and Reproducibility for Targeted Protein Quantification Using a New High-Performance Triple Quadrupole Mass Spectrometer Reiko Kiyonami, Mary Blackburn, Andreas FR Hühme: Thermo Fisher
High-Throughput Protein Quantitation Using Multiple Reaction Monitoring
 High-Throughput Protein Quantitation Using Multiple Reaction Monitoring Application Note Authors Ning Tang, Christine Miller, Joe Roark, Norton Kitagawa and Keith Waddell Agilent Technologies, Inc. Santa
High-Throughput Protein Quantitation Using Multiple Reaction Monitoring Application Note Authors Ning Tang, Christine Miller, Joe Roark, Norton Kitagawa and Keith Waddell Agilent Technologies, Inc. Santa
TOMAHAQ Method Construction
 TOMAHAQ Method Construction Triggered by offset mass accurate-mass high-resolution accurate quantitation (TOMAHAQ) can be performed in the standard method editor of the instrument, without modifications
TOMAHAQ Method Construction Triggered by offset mass accurate-mass high-resolution accurate quantitation (TOMAHAQ) can be performed in the standard method editor of the instrument, without modifications
Workflow concept. Data goes through the workflow. A Node contains an operation An edge represents data flow The results are brought together in tables
 PROTEOME DISCOVERER Workflow concept Data goes through the workflow Spectra Peptides Quantitation A Node contains an operation An edge represents data flow The results are brought together in tables Protein
PROTEOME DISCOVERER Workflow concept Data goes through the workflow Spectra Peptides Quantitation A Node contains an operation An edge represents data flow The results are brought together in tables Protein
HR/AM Targeted Peptide Quantitation on a Q Exactive MS: A Unique Combination of High Selectivity, Sensitivity and Throughput
 Application Note: 554 HR/AM Targeted Peptide Quantitation on a Q Exactive MS: A Unique Combination of High Selectivity, Sensitivity and Throughput Yi Zhang 1, Zhiqi Hao 1, Markus Kellmann 2 and Andreas
Application Note: 554 HR/AM Targeted Peptide Quantitation on a Q Exactive MS: A Unique Combination of High Selectivity, Sensitivity and Throughput Yi Zhang 1, Zhiqi Hao 1, Markus Kellmann 2 and Andreas
Relative Quantitation of TMT-Labeled Proteomes Focus on Sensitivity and Precision
 Relative Quantitation of TMT-Labeled Proteomes Focus on Sensitivity and Precision R. Viner 1, M. Scigelova 2, M. Zeller 2, M. Oppermann 2, T. Moehring 2 and V. Zabrouskov 1 1 Thermo Fisher Scientific,
Relative Quantitation of TMT-Labeled Proteomes Focus on Sensitivity and Precision R. Viner 1, M. Scigelova 2, M. Zeller 2, M. Oppermann 2, T. Moehring 2 and V. Zabrouskov 1 1 Thermo Fisher Scientific,
Reagents. Affinity Tag (Biotin) Acid Cleavage Site. Figure 1. Cleavable ICAT Reagent Structure.
 DATA SHEET Protein Expression Analysis Reagents Background The ultimate goal of proteomics is to identify and quantify proteins that are relevant to a given biological state; and to unearth networks of
DATA SHEET Protein Expression Analysis Reagents Background The ultimate goal of proteomics is to identify and quantify proteins that are relevant to a given biological state; and to unearth networks of
MS-based proteomics to investigate proteins and their modifications
 MS-based proteomics to investigate proteins and their modifications Francis Impens VIB Proteomics Core October th 217 Overview Mass spectrometry-based proteomics: general workflow Identification of protein
MS-based proteomics to investigate proteins and their modifications Francis Impens VIB Proteomics Core October th 217 Overview Mass spectrometry-based proteomics: general workflow Identification of protein
Overview - MS Proteomics in One Slide. MS masses of peptides. MS/MS fragments of a peptide. Results! Match to sequence database
 Overview - MS Proteomics in One Slide Obtain protein Digest into peptides Acquire spectra in mass spectrometer MS masses of peptides MS/MS fragments of a peptide Results! Match to sequence database 2 But
Overview - MS Proteomics in One Slide Obtain protein Digest into peptides Acquire spectra in mass spectrometer MS masses of peptides MS/MS fragments of a peptide Results! Match to sequence database 2 But
Protein Quantitation II: Multiple Reaction Monitoring. Kelly Ruggles New York University
 Protein Quantitation II: Multiple Reaction Monitoring Kelly Ruggles kelly@fenyolab.org New York University Traditional Affinity-based proteomics Use antibodies to quantify proteins Western Blot RPPA Immunohistochemistry
Protein Quantitation II: Multiple Reaction Monitoring Kelly Ruggles kelly@fenyolab.org New York University Traditional Affinity-based proteomics Use antibodies to quantify proteins Western Blot RPPA Immunohistochemistry
Translational Biomarker Core
 Translational Biomarker Core Instrumentation Thermo Scientific TSQ Quantum Triple Quadrupole Mass Spectrometers. There are two TSQ Quantum Ultra AM instruments available in the TBC. The TSQ Quantum Ultra
Translational Biomarker Core Instrumentation Thermo Scientific TSQ Quantum Triple Quadrupole Mass Spectrometers. There are two TSQ Quantum Ultra AM instruments available in the TBC. The TSQ Quantum Ultra
Tandem mass spectra were extracted from the Xcalibur data system format. (.RAW) and charge state assignment was performed using in house software
 Supplementary Methods Software Interpretation of Tandem mass spectra Tandem mass spectra were extracted from the Xcalibur data system format (.RAW) and charge state assignment was performed using in house
Supplementary Methods Software Interpretation of Tandem mass spectra Tandem mass spectra were extracted from the Xcalibur data system format (.RAW) and charge state assignment was performed using in house
Key Words Q Exactive, Accela, MetQuest, Mass Frontier, Drug Discovery
 Metabolite Stability Screening and Hotspot Metabolite Identification by Combining High-Resolution, Accurate-Mass Nonselective and Selective Fragmentation Tim Stratton, Caroline Ding, Yingying Huang, Dan
Metabolite Stability Screening and Hotspot Metabolite Identification by Combining High-Resolution, Accurate-Mass Nonselective and Selective Fragmentation Tim Stratton, Caroline Ding, Yingying Huang, Dan
High-Field Orbitrap Creating new possibilities
 Thermo Scientific Orbitrap Elite Hybrid Mass Spectrometer High-Field Orbitrap Creating new possibilities Ultrahigh resolution Faster scanning Higher sensitivity Complementary fragmentation The highest
Thermo Scientific Orbitrap Elite Hybrid Mass Spectrometer High-Field Orbitrap Creating new possibilities Ultrahigh resolution Faster scanning Higher sensitivity Complementary fragmentation The highest
Supporting Information
 Supporting Information Highly Efficient Ionization of Phosphopeptides at Low ph by Desorption Electrospray Ionization Mass Spectrometry Ning Pan, a, b Pengyuan Liu, b Weidong Cui, c Bo Tang, a Jingmin
Supporting Information Highly Efficient Ionization of Phosphopeptides at Low ph by Desorption Electrospray Ionization Mass Spectrometry Ning Pan, a, b Pengyuan Liu, b Weidong Cui, c Bo Tang, a Jingmin
Tutorial 1: Setting up your Skyline document
 Tutorial 1: Setting up your Skyline document Caution! For using Skyline the number formats of your computer have to be set to English (United States). Open the Control Panel Clock, Language, and Region
Tutorial 1: Setting up your Skyline document Caution! For using Skyline the number formats of your computer have to be set to English (United States). Open the Control Panel Clock, Language, and Region
SILAC and TMT. IDeA National Resource for Proteomics Workshop for Graduate Students and Post-docs Renny Lan 5/18/2017
 SILAC and TMT IDeA National Resource for Proteomics Workshop for Graduate Students and Post-docs Renny Lan 5/18/2017 UHPLC peak chosen at 26.47 min LC Mass at 571.36 chosen for MS/MS MS/MS MS This is a
SILAC and TMT IDeA National Resource for Proteomics Workshop for Graduate Students and Post-docs Renny Lan 5/18/2017 UHPLC peak chosen at 26.47 min LC Mass at 571.36 chosen for MS/MS MS/MS MS This is a
Thermo Scientific LTQ Orbitrap Velos Hybrid FT Mass Spectrometer
 IET International Equipment Trading Ltd. www.ietltd.com Proudly serving laboratories worldwide since 1979 CALL +847.913.0777 for Refurbished & Certified Lab Equipment Thermo Scientific LTQ Orbitrap Velos
IET International Equipment Trading Ltd. www.ietltd.com Proudly serving laboratories worldwide since 1979 CALL +847.913.0777 for Refurbished & Certified Lab Equipment Thermo Scientific LTQ Orbitrap Velos
Designed for Accuracy. Innovation with Integrity. High resolution quantitative proteomics LC-MS
 Designed for Accuracy High resolution quantitative proteomics Innovation with Integrity LC-MS Setting New Standards in Accuracy The development of mass spectrometry based proteomics approaches has dramatically
Designed for Accuracy High resolution quantitative proteomics Innovation with Integrity LC-MS Setting New Standards in Accuracy The development of mass spectrometry based proteomics approaches has dramatically
Protein Quantitation II: Multiple Reaction Monitoring. Kelly Ruggles New York University
 Protein Quantitation II: Multiple Reaction Monitoring Kelly Ruggles kelly@fenyolab.org New York University Traditional Affinity-based proteomics Use antibodies to quantify proteins Western Blot Immunohistochemistry
Protein Quantitation II: Multiple Reaction Monitoring Kelly Ruggles kelly@fenyolab.org New York University Traditional Affinity-based proteomics Use antibodies to quantify proteins Western Blot Immunohistochemistry
Workshop: SILAC and Alternative Labeling Strategies in Quantitative Proteomics
 Workshop: SILAC and Alternative Labeling Strategies in Quantitative Proteomics SILAC and Stable Isotope Dimethyl-Labeling Approaches in Quantitative Proteomics Ho-Tak Lau, Hyong-Won Suh, Shao-En Ong UW
Workshop: SILAC and Alternative Labeling Strategies in Quantitative Proteomics SILAC and Stable Isotope Dimethyl-Labeling Approaches in Quantitative Proteomics Ho-Tak Lau, Hyong-Won Suh, Shao-En Ong UW
Multi-residue analysis of pesticides by GC-HRMS
 An Executive Summary Multi-residue analysis of pesticides by GC-HRMS Dr. Hans Mol is senior scientist at RIKILT- Wageningen UR Introduction Regulatory authorities throughout the world set and enforce strict
An Executive Summary Multi-residue analysis of pesticides by GC-HRMS Dr. Hans Mol is senior scientist at RIKILT- Wageningen UR Introduction Regulatory authorities throughout the world set and enforce strict
Effective desalting and concentration of in-gel digest samples with Vivapure C18 Micro spin columns prior to MALDI-TOF analysis.
 Introduction The identification of proteins plays an important role in today s pharmaceutical and proteomics research. Commonly used methods for separating proteins from complex samples are 1D or 2D gels.
Introduction The identification of proteins plays an important role in today s pharmaceutical and proteomics research. Commonly used methods for separating proteins from complex samples are 1D or 2D gels.
Spectrum-to-Spectrum Searching Using a. Proteome-wide Spectral Library
 MCP Papers in Press. Published on April 30, 2011 as Manuscript M111.007666 Spectrum-to-Spectrum Searching Using a Proteome-wide Spectral Library Chia-Yu Yen, Stephane Houel, Natalie G. Ahn, and William
MCP Papers in Press. Published on April 30, 2011 as Manuscript M111.007666 Spectrum-to-Spectrum Searching Using a Proteome-wide Spectral Library Chia-Yu Yen, Stephane Houel, Natalie G. Ahn, and William
LECTURE-11. Hybrid MS Configurations HANDOUT. As discussed in our previous lecture, mass spectrometry is by far the most versatile
 LECTURE-11 Hybrid MS Configurations HANDOUT PREAMBLE As discussed in our previous lecture, mass spectrometry is by far the most versatile technique used in proteomics. We had also discussed some of the
LECTURE-11 Hybrid MS Configurations HANDOUT PREAMBLE As discussed in our previous lecture, mass spectrometry is by far the most versatile technique used in proteomics. We had also discussed some of the
Purdue-UAB Botanicals Center for Age- Related Disease
 Purdue-UAB Botanicals Center for Age- Related Disease MALDI-TOF Mass Spectrometry Fingerprinting Technique Landon Wilson MALDI-TOF mass spectrometry is an advanced technique for rapid protein identification
Purdue-UAB Botanicals Center for Age- Related Disease MALDI-TOF Mass Spectrometry Fingerprinting Technique Landon Wilson MALDI-TOF mass spectrometry is an advanced technique for rapid protein identification
De novo Protein Sequencing by Combining Top-Down and Bottom-Up Tandem Mass Spectra. Xiaowen Liu
 De novo Protein Sequencing by Combining Top-Down and Bottom-Up Tandem Mass Spectra Xiaowen Liu Department of BioHealth Informatics, Department of Computer and Information Sciences, Indiana University-Purdue
De novo Protein Sequencing by Combining Top-Down and Bottom-Up Tandem Mass Spectra Xiaowen Liu Department of BioHealth Informatics, Department of Computer and Information Sciences, Indiana University-Purdue
Multiple Fragmentation Methods for Small Molecule Characterization on a Dual Pressure Linear Ion Trap Orbitrap Hybrid Mass Spectrometer
 Application ote: 54 Multiple Fragmentation Methods for Small Molecule Characterization on a Dual Pressure Linear Ion Trap rbitrap Hybrid Mass Spectrometer Kate Comstock, Yingying Huang; Thermo Fisher Scientific,
Application ote: 54 Multiple Fragmentation Methods for Small Molecule Characterization on a Dual Pressure Linear Ion Trap rbitrap Hybrid Mass Spectrometer Kate Comstock, Yingying Huang; Thermo Fisher Scientific,
Optimization and Use of Peptide Mass Measurement Accuracy in Shotgun Proteomics* S
 Research Optimization and Use of Peptide Mass Measurement Accuracy in Shotgun Proteomics* S Wilhelm Haas, Brendan K. Faherty, Scott A. Gerber, Joshua E. Elias, Sean A. Beausoleil, Corey E. Bakalarski,
Research Optimization and Use of Peptide Mass Measurement Accuracy in Shotgun Proteomics* S Wilhelm Haas, Brendan K. Faherty, Scott A. Gerber, Joshua E. Elias, Sean A. Beausoleil, Corey E. Bakalarski,
The Agilent 6495 Triple Quadrupole LC/MS: Peptide Quantitation Performance
 The Agilent 495 Triple Quadrupole LC/MS: Peptide Quantitation Performance Technical Overview Introduction Sample complexity and the low concentration of certain biomarkers are the major challenges encountered
The Agilent 495 Triple Quadrupole LC/MS: Peptide Quantitation Performance Technical Overview Introduction Sample complexity and the low concentration of certain biomarkers are the major challenges encountered
Isotopic-Labeling and Mass Spectrometry-Based Quantitative Proteomics
 Isotopic-Labeling and Mass Spectrometry-Based Quantitative Proteomics Xiao-jun Li, Ph.D. Current address: Homestead Clinical Day 4 October 19, 2006 Protein Quantification LC-MS/MS Data XLink mzxml file
Isotopic-Labeling and Mass Spectrometry-Based Quantitative Proteomics Xiao-jun Li, Ph.D. Current address: Homestead Clinical Day 4 October 19, 2006 Protein Quantification LC-MS/MS Data XLink mzxml file
Increasing Speed of UHPLC-MS Analysis Using Single-stage Orbitrap Mass Spectrometer
 Increasing Speed of UHPLC-MS Analysis Using Single-stage Orbitrap Mass Spectrometer Olaf Scheibner and Maciej Bromirski Thermo Fisher Scientific, Bremen, Germany Overview Purpose: Improve the performance
Increasing Speed of UHPLC-MS Analysis Using Single-stage Orbitrap Mass Spectrometer Olaf Scheibner and Maciej Bromirski Thermo Fisher Scientific, Bremen, Germany Overview Purpose: Improve the performance
Comprehensive support for quantitation
 Comprehensive support for quantitation One of the major new features in the current release of Mascot is support for quantitation. This is still work in progress. Our goal is to support all of the popular
Comprehensive support for quantitation One of the major new features in the current release of Mascot is support for quantitation. This is still work in progress. Our goal is to support all of the popular
(APTES), 3- mercaptopropyltrimethoxysilane (MPTMS), tetraethyl orthosilicate (TEOS), N,Ndimethylformamide
 SUPPRTING INFRMATIN Materials: Iron (II) chloride hydrate ( FeCl 2,4H 2, 99%) was purchased from Alfa Aesar ( Ward Hill, MA). Iron (III) chloride hexahydrate ( FeCl 3,6H 2, 97%), (3-aminopropyl)triethoxysilane
SUPPRTING INFRMATIN Materials: Iron (II) chloride hydrate ( FeCl 2,4H 2, 99%) was purchased from Alfa Aesar ( Ward Hill, MA). Iron (III) chloride hexahydrate ( FeCl 3,6H 2, 97%), (3-aminopropyl)triethoxysilane
Comparative Evaluation of Two Isobaric Labeling Tags, DiART and itraq
 pubs.acs.org/ac Comparative Evaluation of Two Isobaric Labeling Tags, DiART and itraq Zhen Chen,,, Quanhui Wang,, Liang Lin, Qi Tang, James L. Edwards,, Shuwei Li,*,, and Siqi Liu*,, Beijing Institute
pubs.acs.org/ac Comparative Evaluation of Two Isobaric Labeling Tags, DiART and itraq Zhen Chen,,, Quanhui Wang,, Liang Lin, Qi Tang, James L. Edwards,, Shuwei Li,*,, and Siqi Liu*,, Beijing Institute
DIA-Umpire: comprehensive computational framework for data independent acquisition proteomics
 DIA-Umpire: comprehensive computational framework for data independent acquisition proteomics Chih-Chiang Tsou 1,2, Dmitry Avtonomov 2, Brett Larsen 3, Monika Tucholska 3, Hyungwon Choi 4 Anne-Claude Gingras
DIA-Umpire: comprehensive computational framework for data independent acquisition proteomics Chih-Chiang Tsou 1,2, Dmitry Avtonomov 2, Brett Larsen 3, Monika Tucholska 3, Hyungwon Choi 4 Anne-Claude Gingras
A Quadrupole-Orbitrap Hybrid Mass Spectrometer Offers Highest Benchtop Performance for In-Depth Analysis of Complex Proteomes
 A Quadrupole-Orbitrap Hybrid Mass Spectrometer Offers Highest Benchtop Performance for In-Depth Analysis of Complex Proteomes Zhiqi Hao 1, Yi Zhang 1, Shannon Eliuk 1, Justin Blethrow 1, Dave Horn 1, Vlad
A Quadrupole-Orbitrap Hybrid Mass Spectrometer Offers Highest Benchtop Performance for In-Depth Analysis of Complex Proteomes Zhiqi Hao 1, Yi Zhang 1, Shannon Eliuk 1, Justin Blethrow 1, Dave Horn 1, Vlad
SERVA ICPL Kit (Cat.-No )
 INSTRUCTION MANUAL SERVA ICPL Kit (Cat.-No. 39230.01) SERVA Electrophoresis GmbH Carl-Benz-Str. 7 D-69115 Heidelberg Phone +49-6221-138400, Fax +49-6221-1384010 e-mail: info@serva.de http://www.serva.de
INSTRUCTION MANUAL SERVA ICPL Kit (Cat.-No. 39230.01) SERVA Electrophoresis GmbH Carl-Benz-Str. 7 D-69115 Heidelberg Phone +49-6221-138400, Fax +49-6221-1384010 e-mail: info@serva.de http://www.serva.de
Identification of proteins by enzyme digestion, mass
 Method for Screening Peptide Fragment Ion Mass Spectra Prior to Database Searching Roger E. Moore, Mary K. Young, and Terry D. Lee Beckman Research Institute of the City of Hope, Duarte, California, USA
Method for Screening Peptide Fragment Ion Mass Spectra Prior to Database Searching Roger E. Moore, Mary K. Young, and Terry D. Lee Beckman Research Institute of the City of Hope, Duarte, California, USA
The Power of LC MALDI: Identification of Proteins by LC MALDI MS/MS Using the Applied Biosystems 4700 Proteomics Analyzer with TOF/TOF Optics
 APPLICATION NOTE TOF MS The Power of LC MALDI: Identification of Proteins by LC MALDI MS/MS Using the Applied Biosystems 4700 Proteomics Analyzer with TOF/TOF Optics Purpose The Applied Biosystems 4700
APPLICATION NOTE TOF MS The Power of LC MALDI: Identification of Proteins by LC MALDI MS/MS Using the Applied Biosystems 4700 Proteomics Analyzer with TOF/TOF Optics Purpose The Applied Biosystems 4700
Rapid Distinction of Leucine and Isoleucine in Monoclonal Antibodies Using Nanoflow. LCMS n. Discovery Attribute Sciences
 Rapid Distinction of Leucine and Isoleucine in Monoclonal Antibodies Using Nanoflow LCMS n Dhanashri Bagal *, Eddie Kast, Ping Cao Discovery Attribute Sciences Amgen, South San Francisco, California, United
Rapid Distinction of Leucine and Isoleucine in Monoclonal Antibodies Using Nanoflow LCMS n Dhanashri Bagal *, Eddie Kast, Ping Cao Discovery Attribute Sciences Amgen, South San Francisco, California, United
Thermo Fisher Scientific, San Jose, CA; 2 Kelleher Lab, Northwestern University, Evanston, IL; 3
 Second-Generation Electron Transfer Dissociation (ETD) on the Thermo Scientific Orbitrap Fusion Mass Spectrometer with Improved Functionality, Increased Speed, Christopher Mullen, 1 Lee Earley, 1 Jean-Jacques
Second-Generation Electron Transfer Dissociation (ETD) on the Thermo Scientific Orbitrap Fusion Mass Spectrometer with Improved Functionality, Increased Speed, Christopher Mullen, 1 Lee Earley, 1 Jean-Jacques
Proteome-wide label-free quantification with MaxQuant. Jürgen Cox Max Planck Institute of Biochemistry July 2011
 Proteome-wide label-free quantification with MaxQuant Jürgen Cox Max Planck Institute of Biochemistry July 2011 MaxQuant MaxQuant Feature detection Data acquisition Initial Andromeda search Statistics
Proteome-wide label-free quantification with MaxQuant Jürgen Cox Max Planck Institute of Biochemistry July 2011 MaxQuant MaxQuant Feature detection Data acquisition Initial Andromeda search Statistics
AB SCIEX SelexION Technology Used to Improve Mass Spectral Library Searching Scores by Removal of Isobaric Interferences
 AB SCIEX SelexION Technology Used to Improve Mass Spectral Library Searching s by Removal of Isobaric Interferences Differential Mobility Used as a Tool to Address Selectivity Challenges Adrian M. Taylor
AB SCIEX SelexION Technology Used to Improve Mass Spectral Library Searching s by Removal of Isobaric Interferences Differential Mobility Used as a Tool to Address Selectivity Challenges Adrian M. Taylor
Yifei Bao. Beatrix. Manor Askenazi
 Detection and Correction of Interference in MS1 Quantitation of Peptides Using their Isotope Distributions Yifei Bao Department of Computer Science Stevens Institute of Technology Beatrix Ueberheide Department
Detection and Correction of Interference in MS1 Quantitation of Peptides Using their Isotope Distributions Yifei Bao Department of Computer Science Stevens Institute of Technology Beatrix Ueberheide Department
Pro tein quantification has become an important. Peptide and Protein Quantification Using itraq with Electron Transfer Dissociation
 Peptide and Protein Quantification Using itraq with Electron Transfer Dissociation Doug Phanstiel," Yi Zhang," [arrod A. Marte,'>' and Joshua J. Coon'':" a Department of Chemistry, University of Wisconsin,
Peptide and Protein Quantification Using itraq with Electron Transfer Dissociation Doug Phanstiel," Yi Zhang," [arrod A. Marte,'>' and Joshua J. Coon'':" a Department of Chemistry, University of Wisconsin,
Quantitative Proteomics
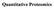 Quantitative Proteomics Quantitation AND Mass Spectrometry Condition A Condition B Identify and quantify differently expressed proteins resulting from a change in the environment (stimulus, disease) Lyse
Quantitative Proteomics Quantitation AND Mass Spectrometry Condition A Condition B Identify and quantify differently expressed proteins resulting from a change in the environment (stimulus, disease) Lyse
MS Based Proteomics: Recent Case Studies Using Advanced Instrumentation
 MS Based Proteomics: Recent Case Studies Using Advanced Instrumentation Chris Adams, PH.D. Stanford University Mass Spectrometry http://mass-spec.stanford.edu/ For personal use only. Please do not reuse
MS Based Proteomics: Recent Case Studies Using Advanced Instrumentation Chris Adams, PH.D. Stanford University Mass Spectrometry http://mass-spec.stanford.edu/ For personal use only. Please do not reuse
Protocol. Product Use & Liability. Contact us: InfoLine: Order per fax: www:
 Protocol SpikeTides Sets SpikeTides Sets_L heavy SpikeMix SpikeMix_L heavy Peptide Sets for relative quantification of Proteins in Mass Spectrometry Based Assays Contact us: InfoLine: +49-30-6392-7878
Protocol SpikeTides Sets SpikeTides Sets_L heavy SpikeMix SpikeMix_L heavy Peptide Sets for relative quantification of Proteins in Mass Spectrometry Based Assays Contact us: InfoLine: +49-30-6392-7878
Plasma Metanephrines and 3-Methoxytyramine by LC/MS/MS Using Agilent SimpliQ WCX SPE, 1290 Infi nity LC, and 6460 Triple Quadrupole LC/MS
 Plasma Metanephrines and 3-Methoxytyramine by LC/MS/MS Using Agilent SimpliQ WCX SPE, 129 Infi nity LC, and 646 Triple Quadrupole LC/MS Application Note Clinical Research Authors Linda Côté and Christophe
Plasma Metanephrines and 3-Methoxytyramine by LC/MS/MS Using Agilent SimpliQ WCX SPE, 129 Infi nity LC, and 646 Triple Quadrupole LC/MS Application Note Clinical Research Authors Linda Côté and Christophe
LECTURE-13. Peptide Mass Fingerprinting HANDOUT. Mass spectrometry is an indispensable tool for qualitative and quantitative analysis of
 LECTURE-13 Peptide Mass Fingerprinting HANDOUT PREAMBLE Mass spectrometry is an indispensable tool for qualitative and quantitative analysis of proteins, drugs and many biological moieties to elucidate
LECTURE-13 Peptide Mass Fingerprinting HANDOUT PREAMBLE Mass spectrometry is an indispensable tool for qualitative and quantitative analysis of proteins, drugs and many biological moieties to elucidate
Chemical Labeling Strategy for Generation of Internal Standards for Targeted Quantitative Proteomics
 Chemical Labeling Strategy for Generation of Internal Standards for Targeted Quantitative Proteomics mtraq Reagents Triplex Christie Hunter, Brian Williamson, Marjorie Minkoff AB SCIEX, USA The utility
Chemical Labeling Strategy for Generation of Internal Standards for Targeted Quantitative Proteomics mtraq Reagents Triplex Christie Hunter, Brian Williamson, Marjorie Minkoff AB SCIEX, USA The utility
Mass spectrometry has been used a lot in biology since the late 1950 s. However it really came into play in the late 1980 s once methods were
 Mass spectrometry has been used a lot in biology since the late 1950 s. However it really came into play in the late 1980 s once methods were developed to allow the analysis of large intact (bigger than
Mass spectrometry has been used a lot in biology since the late 1950 s. However it really came into play in the late 1980 s once methods were developed to allow the analysis of large intact (bigger than
Analytical determination of testosterone in human serum using an Agilent Ultivo Triple Quadrupole LC/MS
 Application Note Clinical Research Analytical determination of testosterone in human serum using an Agilent Ultivo Triple Quadrupole LC/MS Authors Yanan Yang 1, Victor Mandragon 2, and Peter Stone 1 1
Application Note Clinical Research Analytical determination of testosterone in human serum using an Agilent Ultivo Triple Quadrupole LC/MS Authors Yanan Yang 1, Victor Mandragon 2, and Peter Stone 1 1
Types of Analyzers: Quadrupole: mass filter -part1
 16 Types of Analyzers: Sector or double focusing: magnetic and electric Time-of-flight (TOF) Quadrupole (mass filter) Linear ion trap Quadrupole Ion Trap (3D trap) FTICR fourier transform ion cyclotron
16 Types of Analyzers: Sector or double focusing: magnetic and electric Time-of-flight (TOF) Quadrupole (mass filter) Linear ion trap Quadrupole Ion Trap (3D trap) FTICR fourier transform ion cyclotron
PeptideProphet: Validation of Peptide Assignments to MS/MS Spectra. Andrew Keller
 PeptideProphet: Validation of Peptide Assignments to MS/MS Spectra Andrew Keller Outline Need to validate peptide assignments to MS/MS spectra Statistical approach to validation Running PeptideProphet
PeptideProphet: Validation of Peptide Assignments to MS/MS Spectra Andrew Keller Outline Need to validate peptide assignments to MS/MS spectra Statistical approach to validation Running PeptideProphet
Effective Strategies for Improving Peptide Identification with Tandem Mass Spectrometry
 Effective Strategies for Improving Peptide Identification with Tandem Mass Spectrometry by Xi Han A thesis presented to the University of Waterloo in fulfillment of the thesis requirement for the degree
Effective Strategies for Improving Peptide Identification with Tandem Mass Spectrometry by Xi Han A thesis presented to the University of Waterloo in fulfillment of the thesis requirement for the degree
Identification and Characterization of an Isolated Impurity Fraction: Analysis of an Unknown Degradant Found in Quetiapine Fumarate
 Identification and Characterization of an Isolated Impurity Fraction: Analysis of an Unknown Degradant Found in Quetiapine Fumarate Michael D. Jones, Xiang Jin Song, Robert S. Plumb, Peter J. Lee, and
Identification and Characterization of an Isolated Impurity Fraction: Analysis of an Unknown Degradant Found in Quetiapine Fumarate Michael D. Jones, Xiang Jin Song, Robert S. Plumb, Peter J. Lee, and
Assay Robustness Improvement for Drug Urinalysis Using FAIMS and H-SRM on a Triple- Quadrupole Mass Spectrometer
 38 Current Trends in Mass Spectrometry November 6 Assay Robustness Improvement for Drug Urinalysis Using FAIMS and H-SRM on a Triple- Quadrupole Mass Spectrometer This article demonstrates the improved
38 Current Trends in Mass Spectrometry November 6 Assay Robustness Improvement for Drug Urinalysis Using FAIMS and H-SRM on a Triple- Quadrupole Mass Spectrometer This article demonstrates the improved
Rapid method development to study plasma stability of diverse pharmaceutical compounds using Rapid Resolution LC and triple quadrupole MS
 Rapid method development to study plasma stability of diverse pharmaceutical compounds using Rapid Resolution LC and triple quadrupole MS Application Note Drug Discovery Authors Srividya Kailasam Agilent
Rapid method development to study plasma stability of diverse pharmaceutical compounds using Rapid Resolution LC and triple quadrupole MS Application Note Drug Discovery Authors Srividya Kailasam Agilent
Computational Methods for Mass Spectrometry Proteomics
 Computational Methods for Mass Spectrometry Proteomics Eidhammer, Ingvar ISBN-13: 9780470512975 Table of Contents Preface. Acknowledgements. 1 Protein, Proteome, and Proteomics. 1.1 Primary goals for studying
Computational Methods for Mass Spectrometry Proteomics Eidhammer, Ingvar ISBN-13: 9780470512975 Table of Contents Preface. Acknowledgements. 1 Protein, Proteome, and Proteomics. 1.1 Primary goals for studying
Extraction of Methylmalonic Acid from Serum Using ISOLUTE. SAX Prior to LC-MS/MS Analysis
 Application Note AN89.V.1 Extraction of Methylmalonic Acid from Serum Using ISOLUTE SAX Page 1 Extraction of Methylmalonic Acid from Serum Using ISOLUTE SAX Prior to LC-MS/MS Analysis Sample Preparation
Application Note AN89.V.1 Extraction of Methylmalonic Acid from Serum Using ISOLUTE SAX Page 1 Extraction of Methylmalonic Acid from Serum Using ISOLUTE SAX Prior to LC-MS/MS Analysis Sample Preparation
Isobaric Labeling-Based Relative Quantification in Shotgun Proteomics
 This is an open access article published under an ACS AuthorChoice License, which permits copying and redistribution of the article or any adaptations for non-commercial purposes. pubs.acs.org/jpr Isobaric
This is an open access article published under an ACS AuthorChoice License, which permits copying and redistribution of the article or any adaptations for non-commercial purposes. pubs.acs.org/jpr Isobaric
Protein analysis using mass spectrometry
 Protein analysis using mass spectrometry Michael Stadlmeier 2017/12/18 Literature http://www.carellgroup.de/teaching/master 3 What is Proteomics? The proteome is: the entire set of proteins in a given
Protein analysis using mass spectrometry Michael Stadlmeier 2017/12/18 Literature http://www.carellgroup.de/teaching/master 3 What is Proteomics? The proteome is: the entire set of proteins in a given
Yun W. Alelyunas, Mark D. Wrona, Russell J. Mortishire-Smith, Nick Tomczyk, and Paul D. Rainville Waters Corporation, Milford, MA, USA INTRODUCTION
 Quantitation by High Resolution Mass Spectrometry: Using Target Enhancement and Tof-MRM to Achieve Femtogram-level On-column Sensitivity for Quantitation of Drugs in Human Plasma Yun W. Alelyunas, Mark
Quantitation by High Resolution Mass Spectrometry: Using Target Enhancement and Tof-MRM to Achieve Femtogram-level On-column Sensitivity for Quantitation of Drugs in Human Plasma Yun W. Alelyunas, Mark
Bruker Daltonics. EASY-nLC. Tailored HPLC for nano-lc-ms Proteomics. Nano-HPLC. think forward
 Bruker Daltonics EASY-nLC Tailored HPLC for nano-lc-ms Proteomics think forward Nano-HPLC World-Class Performance with a Small Footprint Bruker Daltonics presents a nano-lc system, perfectly integrated
Bruker Daltonics EASY-nLC Tailored HPLC for nano-lc-ms Proteomics think forward Nano-HPLC World-Class Performance with a Small Footprint Bruker Daltonics presents a nano-lc system, perfectly integrated
Maximizing Triple Quadrupole Mass Spectrometry Productivity with the Agilent StreamSelect LC/MS System
 Maximizing Triple Quadrupole Mass Spectrometry Productivity with the Agilent StreamSelect LC/MS System Application Note Authors Kevin McCann, Sameer Nene, Doug McIntyre, Edmond Neo, Dennis Nagtalon, and
Maximizing Triple Quadrupole Mass Spectrometry Productivity with the Agilent StreamSelect LC/MS System Application Note Authors Kevin McCann, Sameer Nene, Doug McIntyre, Edmond Neo, Dennis Nagtalon, and
Electron Transfer Dissociation of N-linked Glycopeptides from a Recombinant mab Using SYNAPT G2-S HDMS
 Electron Transfer Dissociation of N-linked Glycopeptides from a Recombinant mab Using SYNAPT G2-S HDMS Jonathan P. Williams, Jeffery M. Brown, Stephane Houel, Ying Qing Yu, and Weibin Chen Waters Corporation,
Electron Transfer Dissociation of N-linked Glycopeptides from a Recombinant mab Using SYNAPT G2-S HDMS Jonathan P. Williams, Jeffery M. Brown, Stephane Houel, Ying Qing Yu, and Weibin Chen Waters Corporation,
Performing Peptide Bioanalysis Using High Resolution Mass Spectrometry with Target Enhancement MRM Acquisition
 Performing Peptide Bioanalysis Using High Resolution Mass Spectrometry with Target Enhancement MRM Acquisition Yun Wang Alelyunas, Mark D. Wrona, and Nick Tomczyk Waters Corporation, Milford, MA, USA GOAL
Performing Peptide Bioanalysis Using High Resolution Mass Spectrometry with Target Enhancement MRM Acquisition Yun Wang Alelyunas, Mark D. Wrona, and Nick Tomczyk Waters Corporation, Milford, MA, USA GOAL
Q Exactive TM : A True Qual-Quan HR/AM Mass Spectrometer for Routine Proteomics Applications. Yi Zhang, Ph.D. ThermoFisher Scientific
 Q Exactive TM : A True Qual-Quan HR/AM Mass Spectrometer for Routine Proteomics Applications Yi Zhang, Ph.D. ThermoFisher Scientific Outline Introduction of Q Exactive Performance in Discovery Proteomics
Q Exactive TM : A True Qual-Quan HR/AM Mass Spectrometer for Routine Proteomics Applications Yi Zhang, Ph.D. ThermoFisher Scientific Outline Introduction of Q Exactive Performance in Discovery Proteomics
Achieve confident synthesis control with the Thermo Scientific ISQ EC single quadrupole mass spectrometer
 APPLICATION NOTE 72385 Achieve confident synthesis control with the Thermo Scientific ISQ EC single quadrupole mass spectrometer Authors Stephan Meding, Katherine Lovejoy, Martin Ruehl Thermo Fisher Scientific,
APPLICATION NOTE 72385 Achieve confident synthesis control with the Thermo Scientific ISQ EC single quadrupole mass spectrometer Authors Stephan Meding, Katherine Lovejoy, Martin Ruehl Thermo Fisher Scientific,
Bioanalytical Chem: 4590: LC-MSMS of analgesics LC-MS Experiment Liquid Chromatography Mass Spectrometry (LC/MS)
 Liquid Chromatography Mass Spectrometry (LC/MS) Prelab Questions: Questions to be answered before doing the experiment. The answers are due at the beginning of each experiment without exception (the questions
Liquid Chromatography Mass Spectrometry (LC/MS) Prelab Questions: Questions to be answered before doing the experiment. The answers are due at the beginning of each experiment without exception (the questions
Strategies for the Analysis of Therapeutic Peptides in Biofluids by LC-MS/MS. Lee Goodwin
 Strategies for the Analysis of Therapeutic Peptides in Biofluids by LC-MS/MS Lee Goodwin Sample Preparation Chromatography Detection General Strategies Examples New Approaches Summary Outline ABUNDANCE
Strategies for the Analysis of Therapeutic Peptides in Biofluids by LC-MS/MS Lee Goodwin Sample Preparation Chromatography Detection General Strategies Examples New Approaches Summary Outline ABUNDANCE
Powerful Scan Modes of QTRAP System Technology
 Powerful Scan Modes of QTRAP System Technology Unique Hybrid Triple Quadrupole Linear Ion Trap Technology Provides Powerful Workflows to Answer Complex Questions with No Compromises While there are many
Powerful Scan Modes of QTRAP System Technology Unique Hybrid Triple Quadrupole Linear Ion Trap Technology Provides Powerful Workflows to Answer Complex Questions with No Compromises While there are many
Amine specific Labeling Reagents for Multiplexed Relative and Absolute Protein Quantitation
 Product Bulletin itraq Reagents itraq Reagents Amine specific Labeling Reagents for Multiplexed Relative and Absolute Protein Quantitation Background Proteomics research includes the characterization of
Product Bulletin itraq Reagents itraq Reagents Amine specific Labeling Reagents for Multiplexed Relative and Absolute Protein Quantitation Background Proteomics research includes the characterization of
Nature Methods: doi: /nmeth Supplementary Figure 1. Fragment indexing allows efficient spectra similarity comparisons.
 Supplementary Figure 1 Fragment indexing allows efficient spectra similarity comparisons. The cost and efficiency of spectra similarity calculations can be approximated by the number of fragment comparisons
Supplementary Figure 1 Fragment indexing allows efficient spectra similarity comparisons. The cost and efficiency of spectra similarity calculations can be approximated by the number of fragment comparisons
Atomic masses. Atomic masses of elements. Atomic masses of isotopes. Nominal and exact atomic masses. Example: CO, N 2 ja C 2 H 4
 High-Resolution Mass spectrometry (HR-MS, HRAM-MS) (FT mass spectrometry) MS that enables identifying elemental compositions (empirical formulas) from accurate m/z data 9.05.2017 1 Atomic masses (atomic
High-Resolution Mass spectrometry (HR-MS, HRAM-MS) (FT mass spectrometry) MS that enables identifying elemental compositions (empirical formulas) from accurate m/z data 9.05.2017 1 Atomic masses (atomic
Mass Selective Ejection by Axial Resonant Excitation from a Linear Ion Trap
 Mass Selective Ejection by Axial Resonant Excitation from a Linear Ion Trap Yuichiro Hashimoto, Hideki Hasegawa, Takashi Baba, and Izumi Waki Central Research Laboratory, Hitachi, Ld., Tokyo, Japan We
Mass Selective Ejection by Axial Resonant Excitation from a Linear Ion Trap Yuichiro Hashimoto, Hideki Hasegawa, Takashi Baba, and Izumi Waki Central Research Laboratory, Hitachi, Ld., Tokyo, Japan We
Supporting information Online HPLC-ESI-HRMS Method for the Analysis and Comparison of Different Dissolved Organic Matter Samples
 1 2 3 4 5 6 Supporting information Online HPLC-ESI-HRMS Method for the Analysis and Comparison of Different Dissolved Organic Matter Samples Claudia Patriarca a, *, Jonas Bergquist a, Per J. R. Sjöberg
1 2 3 4 5 6 Supporting information Online HPLC-ESI-HRMS Method for the Analysis and Comparison of Different Dissolved Organic Matter Samples Claudia Patriarca a, *, Jonas Bergquist a, Per J. R. Sjöberg
Mass Analyzers. Ion Trap, FTICR, Orbitrap. CU- Boulder CHEM 5181: Mass Spectrometry & Chromatography. Prof. Jose-Luis Jimenez
 Mass Analyzers Ion Trap, FTICR, Orbitrap CU- Boulder CHEM 5181: Mass Spectrometry & Chromatography Prof. Jose-Luis Jimenez Last Update: Oct. 014 Some slides from Dr. Joel Kimmel (007) MS Interpretation
Mass Analyzers Ion Trap, FTICR, Orbitrap CU- Boulder CHEM 5181: Mass Spectrometry & Chromatography Prof. Jose-Luis Jimenez Last Update: Oct. 014 Some slides from Dr. Joel Kimmel (007) MS Interpretation
