Functional Group Fingerprints CNS Chemistry Wilmington, USA
|
|
|
- Martin Warner
- 6 years ago
- Views:
Transcription
1 Functional Group Fingerprints CS Chemistry Wilmington, USA James R. Arnold Charles L. Lerman William F. Michne James R. Damewood American Chemical Society ational Meeting August, 2004 Philadelphia, PA
2 Functional Group Fingerprint bjectives Develop a 2D fingerprint searching method that uses medicinally relevant, custom defined, functional groups. Validate the method on a large dataset of biological targets and chemical classes. Examine approaches for enhancing accuracy, and reducing false positive rates, of 2D searches. Deploy method company-wide. Rapidly expand SAR in it/lead Identification. Subset the corporate collection for screening. Factor of 2 improvement in every 2D similarity search.
3 Many Target Classes Approached by Ligand-Based Methods The Impetus for Developing Functional Group Fingerprints Biochemical Classes of Drug Targets of Current Therapies: (Perhaps 70% of Targets Approached by Ligand-Based Methods) Receptors, 45% Enzymes, 28% Unknown, 7% DA, 2% uclear Receptors, 2% ormones & Factors, 11% Ion Channels, 5% Drews, J. Science 2000, 287,
4 Functional Groups are ne Aspect of Medicinal Chemistry Reasoning in Ligand-Based Design 2 Express one aspect of the knowledge of our most experienced people for wide use. Design is often partially driven by functional group features. These can be warheads, linkers, substituents for interaction with receptors - and they influence many molecular properties. Functional Group Fingerprints capitalize on proven Medicinal Chemistry approaches, and two-dimensional searches are widely used
5 Functional Group Fingerprint Based Classification and Similarity Searching -Classification based on 400 medicinally relevant functional groups -Classification translated into bit strings Imigran (1): GSK, 1.07 billion dollar treatment for migraine in 2000.
6 Functional groups are recognized algorithmically using SMARTS The exclusions make the functional group definitions specific and make the entire set as orthogonal as possible.
7 Functional groups are Defined to Minimize verlap Between Definitions The above imide is defined as an imide.. not two carbonyls, two amides and an amine. rthogonal Functional group definitions allow specific functional groups to be related to activity.
8 Most common functional group classes in 2003 MDDR Functional Group Frequency Functional Group Frequency aromatic nitrogen 9,609 3 o amine, not a arom. 8,493 2 o alcohol 8,104 aryl halide 7,896 2 o amide 7,805 acyclic ether, a arom. 7,262 carboxylic acid 6,464 alkene 6,044 carboxylic ester aromatic alcohol 5,199 3,691 cyclic ether aromatic sulfur, thio. 4,778 3,485 2 o amine, not a arom. 3,281 aromatic sulfur 3,185 3 o amide 3,113 acyclic ether 3,018 3 o amine, a arom 2,938 imidazole, fused, no 2,737 1 o alcohol 2,662 1 o amine, a arom. 2,488 1 o amine, not a arom. 2,474 acetal ketal 2,177 beta lactam, fused 1,931 3 o alcohol 1,921 3 o lactam 1,814 aromatic -, no 1,709 a b unsaturated acid 1,666 2 o amine, a arom. 1,634 lactone 1,586 cyclic ether a 1 arom 1,552 cyclic thioether 1,552 imidazole, fused, w/ 1,533 ketone 1,513 ketone, a arom. 1,476 aromatic ketone 1,385 aromatic w/ 1,326 aromatic oxygen 1,283 acyclic thioether a arom 1,240 a b unsaturated ester 1,188 acyclic thioether 1,166 1 o amide 1,146 2 o lactam 1,136 oxime ether 1,135 trihalide 1,135 nitrile 1,122 sulfonamide, 1,075 urethane, 1,071 urea 864 General categories are shown, actual functional group classifications are more specific.
9 Classification Quality: Coverage and verlap of Functional Group Definitions Coverage: All heteroatoms in molecule are classified. verlap: A heteroatom in molecule classified in > 1 functional group. % Coverage and verlap Ideal Coverage CMC = 8,545 MDDR = 135,342 MedCh = 145, CMC MDDR MedChem Testing in medicinally relevant databases. Roughly 90% coverage and 10% overlap. Ideal verlap
10 Biological Validation: 538 Target Classes in MDDR Active compounds randomly divided into test and training Each Target Class had > 10 actives, or not included n average: 473 actives in 94 clusters* (Daylight) for each class Compounds in MDDR: 4.5 functional groups is median % Compounds # Functional Groups MDDR (Cumulative) # Cpds and # Clust in Tgt. Classes # clusters at Tanimoto # Functional Groups # Compounds: 537 Target Classes * Clusters generated with Daylight fingerprints at Tanimoto = 0.3
11 Tanimoto Scores From Functional Groups Tanimoto based on presence of functional group (binary) or counts (count) Count Tanimoto (C. Lerman) S S 1 2 B1 = FG in mol 1 B2 = FG in mol 2 BC = FG common to mol 1 and mol2 T dist = (B1 + B2-2 * BC) / (B1 + B2 - BC) S 3 Distance Matrix S ne functional group difference = distance
12 Average Percentage Actives Recovered 538 Target Classes in MDDR 2003 Actives in each target class randomly divided test & training. Recovery of test set using training set is graphed. % Actives Retreived Binary Counts Daylight Consensus Random Recovery Rates Top Top Top Top ,000 5,000 Bin Ct Day Cons > 60% Actives in top 1% DBase Ranked MDDR MDDR 2003 > 135,000 cpds.
13 Tanimoto Enrichment Rate Analysis 538 Target Classes in MDDR 2003 Actives in each target class randomly divided test & training Recovery of test set using training set is graphed Enrichment Rate Equation A = # actives at Tanimoto B = # cpds total at Tanimoto ADB = total actives in DBase DB = total cpds in Dbase E = (A / B) / (ADB / DB) Enrichments normalized for the number of actives in target class.
14 Example Biological Categories: MDDR 2003 umber Cpds With Biology In Test Set umber Cpds With Biology Retrieved Daylight 0.3 umber Cpds Total Retrieved Daylight 0.3 En h an ce Ratio 0.3 Daylight umber Cpds With Biology Retrieved FGroup 0.3 umber Cpds Total Retrieved FGroup 0.3 En h an ce Ratio 0.3 FGroup umber Cpds With Biology Retrieved Consens 0.3 umber Cpds Total Retrieved Consens 0.3 Enhance Ratio 0.3 Consens Biology eurokinin K2 Antagonist eurokinin K3 Antagonist Protein Kinase C Inhibitor IV-1 Protease Inhibitor T1B Agonist mglur1 Antagonist Thrombin Inhibitor Factor Xa Inhibitor GABA-B Receptor Antagonis Adrenergic_beta_Blocker Potassium_Channel_Blocke Sodium_Channel_Blocker ACE_Inhibitor Estrogen_Receptor_Modulat Dopamine_D2_Agonist Dopamine_D2_Antagonist Thymidylate_Synthetase_Inh Dihydrofolate_Reductase_Inh Renin_Inhibitor Trypsin_Inhibitor Antiviral Antiinflammatory
15 Consensus Approach: verlap of True Positives from FG Count and Daylight The circles are drawn to scale.
16 Performance of the FG Count, Daylight and Consensus Approaches in Terms of True and False Positives umber of Compounds FG = FG Count D = Daylight C = Consensus FG, 0.0 D, 0.0 C, 0.0 FG, 0.1 D, 0.1 C, 0.1 FG, 0.2 D, 0.2 C, 0.2 FG, 0.3 D, 0.3 C, 0.3 Tanimoto Distance for Methods umber of true and false positives for the Functional Group Fingerprint counts, Daylight fingerprint and consensus (logical AD ) approaches for the five hundred and thirty eight biological target classes at Tanimoto distances of 0.0, 0.1, 0.2, and 0.3. The three methods are binned at the various Tanimoto distances and are reported in the order of counts, Daylight, consensus, and are listed as C, D and A, respectively.
17 Functional Group Fingerprint Conclusions Developed a 2D fingerprint searching method that uses medicinally relevant, custom defined, functional groups. Validated the method on a large dataset of biological targets and chemical classes (538 target classes, 473 cpds 90 clust per class). Factor of 2 gain in accuracy through reduction of false positives. Deploy method company-wide. Rapidly expand SAR in it/lead Identification. Subset the corporate collection for screening. Factor of 2 improvement in 2D searches. Acknowledgement: Dave Cosgrove AstraZeneca
Table 8.2 Detailed Table of Characteristic Infrared Absorption Frequencies
 Table 8.2 Detailed Table of Characteristic Infrared Absorption Frequencies The hydrogen stretch region (3600 2500 cm 1 ). Absorption in this region is associated with the stretching vibration of hydrogen
Table 8.2 Detailed Table of Characteristic Infrared Absorption Frequencies The hydrogen stretch region (3600 2500 cm 1 ). Absorption in this region is associated with the stretching vibration of hydrogen
Molecular Similarity Searching Using Inference Network
 Molecular Similarity Searching Using Inference Network Ammar Abdo, Naomie Salim* Faculty of Computer Science & Information Systems Universiti Teknologi Malaysia Molecular Similarity Searching Search for
Molecular Similarity Searching Using Inference Network Ammar Abdo, Naomie Salim* Faculty of Computer Science & Information Systems Universiti Teknologi Malaysia Molecular Similarity Searching Search for
Introduction. OntoChem
 Introduction ntochem Providing drug discovery knowledge & small molecules... Supporting the task of medicinal chemistry Allows selecting best possible small molecule starting point From target to leads
Introduction ntochem Providing drug discovery knowledge & small molecules... Supporting the task of medicinal chemistry Allows selecting best possible small molecule starting point From target to leads
General Infrared Absorption Ranges of Various Functional Groups
 General Infrared Absorption Ranges of Various Functional Groups Frequency Range Bond Type of Compound cm -1 Intensity C Alkanes 2850-2970 Strong 1340-1470 Strong C Alkenes 3010-3095 Medium 675-995 Strong
General Infrared Absorption Ranges of Various Functional Groups Frequency Range Bond Type of Compound cm -1 Intensity C Alkanes 2850-2970 Strong 1340-1470 Strong C Alkenes 3010-3095 Medium 675-995 Strong
ORGANIC CHEMISTRY. Fifth Edition. Stanley H. Pine
 ORGANIC CHEMISTRY Fifth Edition Stanley H. Pine Professor of Chemistry California State University, Los Angeles McGraw-Hill, Inc. New York St. Louis San Francisco Auckland Bogota Caracas Lisbon London
ORGANIC CHEMISTRY Fifth Edition Stanley H. Pine Professor of Chemistry California State University, Los Angeles McGraw-Hill, Inc. New York St. Louis San Francisco Auckland Bogota Caracas Lisbon London
Aromatic Hydrocarbons
 Aromatic Hydrocarbons Aromatic hydrocarbons contain six-membered rings of carbon atoms with alternating single and double carbon-carbon bonds. The ring is sometimes shown with a circle in the center instead
Aromatic Hydrocarbons Aromatic hydrocarbons contain six-membered rings of carbon atoms with alternating single and double carbon-carbon bonds. The ring is sometimes shown with a circle in the center instead
Identifying Functional Groups. Why is this necessary? Alkanes. Why is this so important? What is a functional group? 2/1/16
 Identifying Functional Groups The Key to Survival Why is this so important? ver and over again, you will be asked to do reactions, the details to which you will receive in lecture and via your textbook.
Identifying Functional Groups The Key to Survival Why is this so important? ver and over again, you will be asked to do reactions, the details to which you will receive in lecture and via your textbook.
GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR (preparation of carboxylic acid esters by telomerisation C07C 67/47; telomerisation C08F)
 CPC - C07B - 2017.08 C07B GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR (preparation of carboxylic acid esters by telomerisation C07C 67/47; telomerisation C08F) General methods for the preparation
CPC - C07B - 2017.08 C07B GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR (preparation of carboxylic acid esters by telomerisation C07C 67/47; telomerisation C08F) General methods for the preparation
Look for absorption bands in decreasing order of importance:
 1. Match the following to their IR spectra (30 points) Look for absorption bands in decreasing order of importance: a e a 2941 1716 d f b 3333 c b 1466 1.the - absorption(s) between 3100 and 2850 cm-1.
1. Match the following to their IR spectra (30 points) Look for absorption bands in decreasing order of importance: a e a 2941 1716 d f b 3333 c b 1466 1.the - absorption(s) between 3100 and 2850 cm-1.
The Basics of General, Organic, and Biological Chemistry
 The Basics of General, Organic, and Biological Chemistry By Ball, Hill and Scott Download PDF at https://open.umn.edu/opentextbooks/bookdetail.aspx?bookid=40 Page 5 Chapter 1 Chemistry, Matter, and Measurement
The Basics of General, Organic, and Biological Chemistry By Ball, Hill and Scott Download PDF at https://open.umn.edu/opentextbooks/bookdetail.aspx?bookid=40 Page 5 Chapter 1 Chemistry, Matter, and Measurement
More information can be found in Chapter 12 in your textbook for CHEM 3750/ 3770 and on pages in your laboratory manual.
 CHEM 3780 rganic Chemistry II Infrared Spectroscopy and Mass Spectrometry Review More information can be found in Chapter 12 in your textbook for CHEM 3750/ 3770 and on pages 13-28 in your laboratory manual.
CHEM 3780 rganic Chemistry II Infrared Spectroscopy and Mass Spectrometry Review More information can be found in Chapter 12 in your textbook for CHEM 3750/ 3770 and on pages 13-28 in your laboratory manual.
Chapter 2. Molecular Representations
 hapter 2. Molecular Representations 3 () 3 ( 3 ) 2 3 3 3 8 Lewis (Kekule) structure ondensed and par6ally condensed structure Skeletal (bond- line) structure Molecular formula Amoxicillin a widely prescribed
hapter 2. Molecular Representations 3 () 3 ( 3 ) 2 3 3 3 8 Lewis (Kekule) structure ondensed and par6ally condensed structure Skeletal (bond- line) structure Molecular formula Amoxicillin a widely prescribed
PHARMACEUTICAL CHEMISTRY EXAM #1 Februrary 21, 2008
 PHARMACEUTICAL CHEMISTRY EXAM #1 Februrary 21, 2008 1 Name SECTION B. Answer each question in this section by writing the letter corresponding to the best answer on the line provided (2 points each; 60
PHARMACEUTICAL CHEMISTRY EXAM #1 Februrary 21, 2008 1 Name SECTION B. Answer each question in this section by writing the letter corresponding to the best answer on the line provided (2 points each; 60
Patrick: An Introduction to Medicinal Chemistry 5e Chapter 01
 Questions Patrick: An Introduction to Medicinal Chemistry 5e 01) Which of the following molecules is a phospholipid? a. i b. ii c. iii d. iv 02) Which of the following statements is false regarding the
Questions Patrick: An Introduction to Medicinal Chemistry 5e 01) Which of the following molecules is a phospholipid? a. i b. ii c. iii d. iv 02) Which of the following statements is false regarding the
Synthesis of Nitriles a. dehydration of 1 amides using POCl 3 : b. SN2 reaction of cyanide ion on halides:
 I. Nitriles Nitriles consist of the CN functional group, and are linear with sp hybridization on C and N. Nitriles are non-basic at nitrogen, since the lone pair exists in an sp orbital (50% s character
I. Nitriles Nitriles consist of the CN functional group, and are linear with sp hybridization on C and N. Nitriles are non-basic at nitrogen, since the lone pair exists in an sp orbital (50% s character
Molecular Graphics. Molecular Graphics Expt. 1 1
 Molecular Graphics Expt. 1 1 Molecular Graphics The study of organic chemistry has for more than a century and a half focussed on the relationship between the structure of an organic molecule (its three-dimensional
Molecular Graphics Expt. 1 1 Molecular Graphics The study of organic chemistry has for more than a century and a half focussed on the relationship between the structure of an organic molecule (its three-dimensional
Jonathan S. Mason,, Isabelle Morize, Paul R. Menard,*, Daniel L. Cheney, Christopher Hulme, and Richard F. Labaudiniere
 J. Med. Chem. 1999, 42, 3251-3264 3251 New 4-Point Pharmacophore Method for Molecular Similarity and Diversity Applications: Overview of the Method and Applications, Including a Novel Approach to the Design
J. Med. Chem. 1999, 42, 3251-3264 3251 New 4-Point Pharmacophore Method for Molecular Similarity and Diversity Applications: Overview of the Method and Applications, Including a Novel Approach to the Design
Using NMR and IR Spectroscopy to Determine Structures Dr. Carl Hoeger, UCSD
 Using NMR and IR Spectroscopy to Determine Structures Dr. Carl Hoeger, UCSD The following guidelines should be helpful in assigning a structure from NMR (both PMR and CMR) and IR data. At the end of this
Using NMR and IR Spectroscopy to Determine Structures Dr. Carl Hoeger, UCSD The following guidelines should be helpful in assigning a structure from NMR (both PMR and CMR) and IR data. At the end of this
Machine Learning Concepts in Chemoinformatics
 Machine Learning Concepts in Chemoinformatics Martin Vogt B-IT Life Science Informatics Rheinische Friedrich-Wilhelms-Universität Bonn BigChem Winter School 2017 25. October Data Mining in Chemoinformatics
Machine Learning Concepts in Chemoinformatics Martin Vogt B-IT Life Science Informatics Rheinische Friedrich-Wilhelms-Universität Bonn BigChem Winter School 2017 25. October Data Mining in Chemoinformatics
Chem 1075 Chapter 19 Organic Chemistry Lecture Outline
 Chem 1075 Chapter 19 Organic Chemistry Lecture Outline Slide 2 Introduction Organic chemistry is the study of and its compounds. The major sources of carbon are the fossil fuels: petroleum, natural gas,
Chem 1075 Chapter 19 Organic Chemistry Lecture Outline Slide 2 Introduction Organic chemistry is the study of and its compounds. The major sources of carbon are the fossil fuels: petroleum, natural gas,
How to Interpret an Infrared (IR) Spectrum
 How to Interpret an Infrared (IR) Spectrum Infrared (IR) Spectroscopy allows the identification of particular bonds present within molecules. In this class we have simplified IR analysis by only focusing
How to Interpret an Infrared (IR) Spectrum Infrared (IR) Spectroscopy allows the identification of particular bonds present within molecules. In this class we have simplified IR analysis by only focusing
MOLECULAR REPRESENTATIONS AND INFRARED SPECTROSCOPY
 MOLEULAR REPRESENTATIONS AND INFRARED SPETROSOPY A STUDENT SOULD BE ABLE TO: 1. Given a Lewis (dash or dot), condensed, bond-line, or wedge formula of a compound draw the other representations. 2. Give
MOLEULAR REPRESENTATIONS AND INFRARED SPETROSOPY A STUDENT SOULD BE ABLE TO: 1. Given a Lewis (dash or dot), condensed, bond-line, or wedge formula of a compound draw the other representations. 2. Give
ORGANIC - BROWN 8E CH INFRARED SPECTROSCOPY.
 !! www.clutchprep.com CONCEPT: PURPOSE OF ANALYTICAL TECHNIQUES Classical Methods (Wet Chemistry): Chemists needed to run dozens of chemical reactions to determine the type of molecules in a compound.
!! www.clutchprep.com CONCEPT: PURPOSE OF ANALYTICAL TECHNIQUES Classical Methods (Wet Chemistry): Chemists needed to run dozens of chemical reactions to determine the type of molecules in a compound.
Infrared Characteristic Group Frequencies
 Infrared Characteristic Group Frequencies Tables and Charts Second Edition GEORGE SOCRATES Brunei, The University of West London, Middlesex, United Kingdom JOHN WILEY & SONS Chichester New York Brisbane
Infrared Characteristic Group Frequencies Tables and Charts Second Edition GEORGE SOCRATES Brunei, The University of West London, Middlesex, United Kingdom JOHN WILEY & SONS Chichester New York Brisbane
GENERAL, ORGANIC AND BIOLOGICAL CHEMISTRY. JOHN R. AMEND Montana State University. BRADFORD P. MUNDY Colby College
 GENERAL, ORGANIC AND BIOLOGICAL CHEMISTRY JOHN R. AMEND Montana State University BRADFORD P. MUNDY Colby College MELVIN T. ARMOLD Adams State College # Saunders Golden Sunburst Series SAUNDERS COLLEGE
GENERAL, ORGANIC AND BIOLOGICAL CHEMISTRY JOHN R. AMEND Montana State University BRADFORD P. MUNDY Colby College MELVIN T. ARMOLD Adams State College # Saunders Golden Sunburst Series SAUNDERS COLLEGE
Chemistry 11. Unit 10 Organic Chemistry Part I Introduction
 Chemistry 11 Unit 10 Organic Chemistry Part I Introduction 2 1. Prelude In this chapter we will be looking at organic chemistry. By definition, organic chemistry is the study of structure, properties and
Chemistry 11 Unit 10 Organic Chemistry Part I Introduction 2 1. Prelude In this chapter we will be looking at organic chemistry. By definition, organic chemistry is the study of structure, properties and
Ligand-receptor interactions
 University of Silesia, Katowice, Poland 11 22 March 2013 Ligand-receptor interactions Dr. Pavel Polishchuk A.V. Bogatsky Physico-Chemical Institute of National Academy of Sciences of Ukraine Odessa, Ukraine
University of Silesia, Katowice, Poland 11 22 March 2013 Ligand-receptor interactions Dr. Pavel Polishchuk A.V. Bogatsky Physico-Chemical Institute of National Academy of Sciences of Ukraine Odessa, Ukraine
CHEM 203. Midterm Exam 1 October 31, 2008 ANSWERS. This a closed-notes, closed-book exam. You may use your set of molecular models
 CEM 203 Midterm Exam 1 ctober 31, 2008 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This exam contains 8 pages Time: 1h 30 min 1. / 15 2. / 16 3. /
CEM 203 Midterm Exam 1 ctober 31, 2008 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This exam contains 8 pages Time: 1h 30 min 1. / 15 2. / 16 3. /
Assessing Synthetic Accessibility of Chemical Compounds Using Machine Learning Methods
 J. Chem. Inf. Model. 2010, 50, 979 991 979 Assessing Synthetic Accessibility of Chemical Compounds Using Machine Learning Methods Yevgeniy Podolyan, Michael A. Walters, and George Karypis*, Department
J. Chem. Inf. Model. 2010, 50, 979 991 979 Assessing Synthetic Accessibility of Chemical Compounds Using Machine Learning Methods Yevgeniy Podolyan, Michael A. Walters, and George Karypis*, Department
Bio-elements. Living organisms requires only 27 of the 90 common chemical elements found in the crust of the earth, to be as its essential components.
 Bio-elements Living organisms requires only 27 of the 90 common chemical elements found in the crust of the earth, to be as its essential components. Most of the chemical components of living organisms
Bio-elements Living organisms requires only 27 of the 90 common chemical elements found in the crust of the earth, to be as its essential components. Most of the chemical components of living organisms
Chapter 25: The Chemistry of Life: Organic and Biological Chemistry
 Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
An Enhancement of Bayesian Inference Network for Ligand-Based Virtual Screening using Features Selection
 American Journal of Applied Sciences 8 (4): 368-373, 2011 ISSN 1546-9239 2010 Science Publications An Enhancement of Bayesian Inference Network for Ligand-Based Virtual Screening using Features Selection
American Journal of Applied Sciences 8 (4): 368-373, 2011 ISSN 1546-9239 2010 Science Publications An Enhancement of Bayesian Inference Network for Ligand-Based Virtual Screening using Features Selection
Course Information. Instructor Information
 Jordan University of Science and Technology Department of Chemistry Course Syllabus Fall 2018/2019 Course Information Course Number: CHEM 108 Course Name: General and Organic Chemistry Credit Hours: 4
Jordan University of Science and Technology Department of Chemistry Course Syllabus Fall 2018/2019 Course Information Course Number: CHEM 108 Course Name: General and Organic Chemistry Credit Hours: 4
Chemogenomic: Approaches to Rational Drug Design. Jonas Skjødt Møller
 Chemogenomic: Approaches to Rational Drug Design Jonas Skjødt Møller Chemogenomic Chemistry Biology Chemical biology Medical chemistry Chemical genetics Chemoinformatics Bioinformatics Chemoproteomics
Chemogenomic: Approaches to Rational Drug Design Jonas Skjødt Møller Chemogenomic Chemistry Biology Chemical biology Medical chemistry Chemical genetics Chemoinformatics Bioinformatics Chemoproteomics
Infra-red Spectroscopy
 Molecular vibrations are associated with the absorption of energy (infrared activity) by the molecule as sets of atoms (molecular moieties) vibrate about the mean center of their chemical bonds. Infra-red
Molecular vibrations are associated with the absorption of energy (infrared activity) by the molecule as sets of atoms (molecular moieties) vibrate about the mean center of their chemical bonds. Infra-red
Chapter 2 Molecular Representations
 hapter 2 Molecular Representations Structural Formulas and ondensed Structures Because organic compounds are molecular in nature, the additional information conveyed in structural formulas is sometimes
hapter 2 Molecular Representations Structural Formulas and ondensed Structures Because organic compounds are molecular in nature, the additional information conveyed in structural formulas is sometimes
Lecture 11. IR Theory. Next Class: Lecture Problem 4 due Thin-Layer Chromatography
 Lecture 11 IR Theory Next Class: Lecture Problem 4 due Thin-Layer Chromatography This Week In Lab: Ch 6: Procedures 2 & 3 Procedure 4 (outside of lab) Next Week in Lab: Ch 7: PreLab Due Quiz 4 Ch 5 Final
Lecture 11 IR Theory Next Class: Lecture Problem 4 due Thin-Layer Chromatography This Week In Lab: Ch 6: Procedures 2 & 3 Procedure 4 (outside of lab) Next Week in Lab: Ch 7: PreLab Due Quiz 4 Ch 5 Final
ORGANIC - EGE 5E CH. 2 - COVALENT BONDING AND CHEMICAL REACTIVITY
 !! www.clutchprep.com CONCEPT: HYBRID ORBITAL THEORY The Aufbau Principle states that electrons fill orbitals in order of increasing energy. If carbon has only two unfilled orbitals, why does it like to
!! www.clutchprep.com CONCEPT: HYBRID ORBITAL THEORY The Aufbau Principle states that electrons fill orbitals in order of increasing energy. If carbon has only two unfilled orbitals, why does it like to
Chapter 12: Carbonyl Compounds II
 Chapter 12: Carbonyl Compounds II Learning bjectives: 1. Recognize and assign names to aldehydes and ketones. 2. Write the mechanism for nucleophilic addition and nucleophilic addition-elimination reactions
Chapter 12: Carbonyl Compounds II Learning bjectives: 1. Recognize and assign names to aldehydes and ketones. 2. Write the mechanism for nucleophilic addition and nucleophilic addition-elimination reactions
COMPARISON OF SIMILARITY METHOD TO IMPROVE RETRIEVAL PERFORMANCE FOR CHEMICAL DATA
 http://www.ftsm.ukm.my/apjitm Asia-Pacific Journal of Information Technology and Multimedia Jurnal Teknologi Maklumat dan Multimedia Asia-Pasifik Vol. 7 No. 1, June 2018: 91-98 e-issn: 2289-2192 COMPARISON
http://www.ftsm.ukm.my/apjitm Asia-Pacific Journal of Information Technology and Multimedia Jurnal Teknologi Maklumat dan Multimedia Asia-Pasifik Vol. 7 No. 1, June 2018: 91-98 e-issn: 2289-2192 COMPARISON
Chapter 20: Carboxylic Acids
 1 Chapter 20: Carboxylic Acids I. Introduction: Carboxylic acid structure: Classification of carboxylic acids: A carboxylic acid donates protons by the heterocyclic cleavage of the O-H bond, generating
1 Chapter 20: Carboxylic Acids I. Introduction: Carboxylic acid structure: Classification of carboxylic acids: A carboxylic acid donates protons by the heterocyclic cleavage of the O-H bond, generating
A Tiered Screen Protocol for the Discovery of Structurally Diverse HIV Integrase Inhibitors
 A Tiered Screen Protocol for the Discovery of Structurally Diverse HIV Integrase Inhibitors Rajarshi Guha, Debojyoti Dutta, Ting Chen and David J. Wild School of Informatics Indiana University and Dept.
A Tiered Screen Protocol for the Discovery of Structurally Diverse HIV Integrase Inhibitors Rajarshi Guha, Debojyoti Dutta, Ting Chen and David J. Wild School of Informatics Indiana University and Dept.
CHEMISTRY 1A Fall 2010 Final Exam Key
 CHEMISTRY 1A Fall 2010 Final Exam Key YOU MIGHT FIND THE FOLLOWING USEFUL; 0.008314 kj H E ( n)rt R = K mol 0.00418 kj q C cal m w T g C H rxn = H f (products) H f (reactants) Electronegativities H 2.2
CHEMISTRY 1A Fall 2010 Final Exam Key YOU MIGHT FIND THE FOLLOWING USEFUL; 0.008314 kj H E ( n)rt R = K mol 0.00418 kj q C cal m w T g C H rxn = H f (products) H f (reactants) Electronegativities H 2.2
Introduc)on to Func)onal Groups in Organic Molecules
 Introduc)on to Func)onal Groups in rganic Molecules CH 3 H 3 C N C C N C C N N CH CH 3 Caffeine Func)onal Group Func%onal group - collec)on of atoms at a site that have a characteris)c behavior in all
Introduc)on to Func)onal Groups in rganic Molecules CH 3 H 3 C N C C N C C N N CH CH 3 Caffeine Func)onal Group Func%onal group - collec)on of atoms at a site that have a characteris)c behavior in all
An alcohol is a compound obtained by substituting a hydoxyl group ( OH) for an H atom on a carbon atom of a hydrocarbon group.
 Derivatives of Hydrocarbons A functional group is a reactive portion of a molecule that undergoes predictable reactions. All other organic compounds can be considered as derivatives of hydrocarbons (i.e.,
Derivatives of Hydrocarbons A functional group is a reactive portion of a molecule that undergoes predictable reactions. All other organic compounds can be considered as derivatives of hydrocarbons (i.e.,
Exam 1 (Monday, July 6, 2015)
 Chem 231 Summer 2015 Assigned Homework Problems Last updated: Friday, July 24, 2015 Problems Assigned from Essential Organic Chemistry, 2 nd Edition, Paula Yurkanis Bruice, Prentice Hall, New York, NY,
Chem 231 Summer 2015 Assigned Homework Problems Last updated: Friday, July 24, 2015 Problems Assigned from Essential Organic Chemistry, 2 nd Edition, Paula Yurkanis Bruice, Prentice Hall, New York, NY,
Infrared Spectroscopy
 Infrared Spectroscopy IR Spectroscopy Used to identify organic compounds IR spectroscopy provides a 100% identification if the spectrum is matched. If not, IR at least provides information about the types
Infrared Spectroscopy IR Spectroscopy Used to identify organic compounds IR spectroscopy provides a 100% identification if the spectrum is matched. If not, IR at least provides information about the types
Similarity methods for ligandbased virtual screening
 Similarity methods for ligandbased virtual screening Peter Willett, University of Sheffield Computers in Scientific Discovery 5, 22 nd July 2010 Overview Molecular similarity and its use in virtual screening
Similarity methods for ligandbased virtual screening Peter Willett, University of Sheffield Computers in Scientific Discovery 5, 22 nd July 2010 Overview Molecular similarity and its use in virtual screening
QUALITATIVE ORGANIC CHEMICAL ANALYSIS
 QUALITATIVE ORGANIC CHEMICAL ANALYSIS QUALITATIVE ORGANIC CHEMICAL ANALYSIS W. J. Criddle, B.Sc., Ph.D. (Wales) G. P. Ellis, B.Sc., Ph.D. (Lond.), F.R.I.C. University of Wales Institute of Science and
QUALITATIVE ORGANIC CHEMICAL ANALYSIS QUALITATIVE ORGANIC CHEMICAL ANALYSIS W. J. Criddle, B.Sc., Ph.D. (Wales) G. P. Ellis, B.Sc., Ph.D. (Lond.), F.R.I.C. University of Wales Institute of Science and
2Dstructuredrawing Chem314 Beauchamp
 2Dstructuredrawing hem314 Beauchamp 3 2 3 3 2 2 3 2 2 3 2 2 (neutral) (cation) (anion) (free radical) use zig-zag drawing for sp 3 chains 1 o carbocation 1 o carbanion 1 o free radical 3 3 3 3 3 3 (cation)
2Dstructuredrawing hem314 Beauchamp 3 2 3 3 2 2 3 2 2 3 2 2 (neutral) (cation) (anion) (free radical) use zig-zag drawing for sp 3 chains 1 o carbocation 1 o carbanion 1 o free radical 3 3 3 3 3 3 (cation)
Chapter 9. Organic Chemistry: The Infinite Variety of Carbon Compounds. Organic Chemistry
 Chapter 9 Organic Chemistry: The Infinite Variety of Carbon Compounds Organic Chemistry Organic chemistry is defined as the chemistry of carbon compounds. Of tens of millions of known chemical compounds,
Chapter 9 Organic Chemistry: The Infinite Variety of Carbon Compounds Organic Chemistry Organic chemistry is defined as the chemistry of carbon compounds. Of tens of millions of known chemical compounds,
Computational Chemistry in Drug Design. Xavier Fradera Barcelona, 17/4/2007
 Computational Chemistry in Drug Design Xavier Fradera Barcelona, 17/4/2007 verview Introduction and background Drug Design Cycle Computational methods Chemoinformatics Ligand Based Methods Structure Based
Computational Chemistry in Drug Design Xavier Fradera Barcelona, 17/4/2007 verview Introduction and background Drug Design Cycle Computational methods Chemoinformatics Ligand Based Methods Structure Based
12.1 The Nature of Organic molecules
 12.1 The Nature of Organic molecules Organic chemistry: : The chemistry of carbon compounds. Carbon is tetravalent; it always form four bonds. Prentice Hall 2003 Chapter One 2 Organic molecules have covalent
12.1 The Nature of Organic molecules Organic chemistry: : The chemistry of carbon compounds. Carbon is tetravalent; it always form four bonds. Prentice Hall 2003 Chapter One 2 Organic molecules have covalent
Chapter 25 Organic and Biological Chemistry
 Chapter 25 Organic and Biological Chemistry Organic Chemistry The chemistry of carbon compounds. Carbon has the ability to form long chains. Without this property, large biomolecules such as proteins,
Chapter 25 Organic and Biological Chemistry Organic Chemistry The chemistry of carbon compounds. Carbon has the ability to form long chains. Without this property, large biomolecules such as proteins,
SAR. Structure - Activity Relationships (alkoholy, amíny, aldehydy, ketóny, estery, amidy, kyseliny, uhľovodíky) 2/28/2016
 Structure Activity elationships (SA) identifies which functional groups are important for binding and activity SA Structure - Activity elationships (alkoholy, amíny, aldehydy, ketóny, estery, amidy, kyseliny,
Structure Activity elationships (SA) identifies which functional groups are important for binding and activity SA Structure - Activity elationships (alkoholy, amíny, aldehydy, ketóny, estery, amidy, kyseliny,
Infrared Spectroscopy: How to use the 5 zone approach to identify functional groups
 Infrared Spectroscopy: How to use the 5 zone approach to identify functional groups Definition: Infrared Spectroscopy is the study of the Infrared Spectrum. An Infrared Spectrum is the plot of photon energy
Infrared Spectroscopy: How to use the 5 zone approach to identify functional groups Definition: Infrared Spectroscopy is the study of the Infrared Spectrum. An Infrared Spectrum is the plot of photon energy
2. Separate the ions based on their mass to charge (m/e) ratio. 3. Measure the relative abundance of the ions that are produced
 I. Mass spectrometry: capable of providing both quantitative and qualitative information about samples as small as 100 pg (!) and with molar masses in the 10 4-10 5 kdalton range A. The mass spectrometer
I. Mass spectrometry: capable of providing both quantitative and qualitative information about samples as small as 100 pg (!) and with molar masses in the 10 4-10 5 kdalton range A. The mass spectrometer
Montgomery County Community College CHE 132 Chemistry for Technology II 4-3-3
 Montgomery County Community College CHE 132 Chemistry for Technology II 4-3-3 COURSE DESCRIPTION: This course will present a brief overview of Nuclear Chemistry. The major portion of the semester will
Montgomery County Community College CHE 132 Chemistry for Technology II 4-3-3 COURSE DESCRIPTION: This course will present a brief overview of Nuclear Chemistry. The major portion of the semester will
OXFORD H i g h e r E d u c a t i o n Oxford University Press, All rights reserved.
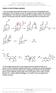 Patrick, An Introduction to dicinal hemistry 4e Answers to end-of-chapter questions 1) The mechanism below shows the release of one molecule of formaldehyde from methenamine. The mechanism can then be
Patrick, An Introduction to dicinal hemistry 4e Answers to end-of-chapter questions 1) The mechanism below shows the release of one molecule of formaldehyde from methenamine. The mechanism can then be
Fragment-based de novo Design
 ragment-based de novo Design Gisbert Schneider & Uli echner gisbert.schneider@modlab.de www.modlab.de Beilstein Endowed Chair for Cheminformatics Institute of rganic Chemistry & Chemical Biology Johann
ragment-based de novo Design Gisbert Schneider & Uli echner gisbert.schneider@modlab.de www.modlab.de Beilstein Endowed Chair for Cheminformatics Institute of rganic Chemistry & Chemical Biology Johann
Using Phase for Pharmacophore Modelling. 5th European Life Science Bootcamp March, 2017
 Using Phase for Pharmacophore Modelling 5th European Life Science Bootcamp March, 2017 Phase: Our Pharmacohore generation tool Significant improvements to Phase methods in 2016 New highly interactive interface
Using Phase for Pharmacophore Modelling 5th European Life Science Bootcamp March, 2017 Phase: Our Pharmacohore generation tool Significant improvements to Phase methods in 2016 New highly interactive interface
1.4A: Common functional groups in organic compounds
 Ashley Robison My Preferences Site Tools FAQ Sign Out If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out. ChemWiki BioWiki GeoWiki StatWiki
Ashley Robison My Preferences Site Tools FAQ Sign Out If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out. ChemWiki BioWiki GeoWiki StatWiki
Chemistry 11. Organic Chemistry
 hemistry 11 rganic hemistry Are you feeling like a happy rganic hemistry Pony fully of rainbows, sunshine and joy?...r are you more like this guy? (don't worry...this is the last booklet) Book 3 Name:
hemistry 11 rganic hemistry Are you feeling like a happy rganic hemistry Pony fully of rainbows, sunshine and joy?...r are you more like this guy? (don't worry...this is the last booklet) Book 3 Name:
Chapter 22. Organic and Biological Molecules
 Chapter 22 Organic and Biological Molecules The Bonding of Carbon Organic chemistry is the chemistry of compounds containing carbon. Because carbon can form single, double, and triple bonds, the following
Chapter 22 Organic and Biological Molecules The Bonding of Carbon Organic chemistry is the chemistry of compounds containing carbon. Because carbon can form single, double, and triple bonds, the following
10. Amines (text )
 2009, Department of Chemistry, The University of Western Ontario 10.1 10. Amines (text 10.1 10.6) A. Structure and omenclature Amines are derivatives of ammonia (H 3 ), where one or more H atoms has been
2009, Department of Chemistry, The University of Western Ontario 10.1 10. Amines (text 10.1 10.6) A. Structure and omenclature Amines are derivatives of ammonia (H 3 ), where one or more H atoms has been
Alkanes 3/27/17. Hydrocarbons: Compounds made of hydrogen and carbon only. Aliphatic (means fat ) - Open chain Aromatic - ring. Alkane Alkene Alkyne
 Alkanes EQ 1. How will I define Hydrocarbons? 2. Compare and contrast the 3 types of hydrocarbons (Alkanes, alkenes, alkynes). Hydrocarbons: Compounds made of hydrogen and carbon only. Aliphatic (means
Alkanes EQ 1. How will I define Hydrocarbons? 2. Compare and contrast the 3 types of hydrocarbons (Alkanes, alkenes, alkynes). Hydrocarbons: Compounds made of hydrogen and carbon only. Aliphatic (means
Chapter 20 Carboxylic Acid Derivatives Nucleophilic Acyl Substitution
 Chapter 20 Carboxylic Acid Derivatives Nucleophilic Acyl Substitution Nomenclature: In carboxylic acid chlorides, anhydrides, esters and amides, the parent is the carboxylic acid. In each case be sure
Chapter 20 Carboxylic Acid Derivatives Nucleophilic Acyl Substitution Nomenclature: In carboxylic acid chlorides, anhydrides, esters and amides, the parent is the carboxylic acid. In each case be sure
Similarity Search. Uwe Koch
 Similarity Search Uwe Koch Similarity Search The similar property principle: strurally similar molecules tend to have similar properties. However, structure property discontinuities occur frequently. Relevance
Similarity Search Uwe Koch Similarity Search The similar property principle: strurally similar molecules tend to have similar properties. However, structure property discontinuities occur frequently. Relevance
Name: Score: /100. Part I. Multiple choice. Write the letter of the correct answer for each problem. 3 points each
 ame: Score: /100 Part I. Multiple choice. Write the letter of the correct answer for each problem. 3 points each 1. When l is added to pure water, l molecules lose protons, while water molecules gain protons.
ame: Score: /100 Part I. Multiple choice. Write the letter of the correct answer for each problem. 3 points each 1. When l is added to pure water, l molecules lose protons, while water molecules gain protons.
Chemistry 1A Spring 1998 Exam #4 KEY Chapters 9 & 10
 Chemistry 1A Spring 1998 Exam #4 KEY Chapters 9 & 10 For each of the following, write the word, words, or number in each blank that best completes each sentence. (2 points each) 1. A(n) molecular orbital
Chemistry 1A Spring 1998 Exam #4 KEY Chapters 9 & 10 For each of the following, write the word, words, or number in each blank that best completes each sentence. (2 points each) 1. A(n) molecular orbital
ORGANIC - BROWN 8E CH.1 - COVALENT BONDING AND SHAPES OF MOLECULES
 !! www.clutchprep.com CONCEPT: WHAT IS ORGANIC CHEMISTRY? Organic Chemistry is the chemistry of life. It consists of the study of molecules that are (typically) created and used by biological systems.
!! www.clutchprep.com CONCEPT: WHAT IS ORGANIC CHEMISTRY? Organic Chemistry is the chemistry of life. It consists of the study of molecules that are (typically) created and used by biological systems.
Data Mining in the Chemical Industry. Overview of presentation
 Data Mining in the Chemical Industry Glenn J. Myatt, Ph.D. Partner, Myatt & Johnson, Inc. glenn.myatt@gmail.com verview of presentation verview of the chemical industry Example of the pharmaceutical industry
Data Mining in the Chemical Industry Glenn J. Myatt, Ph.D. Partner, Myatt & Johnson, Inc. glenn.myatt@gmail.com verview of presentation verview of the chemical industry Example of the pharmaceutical industry
antidisestablishmenttarianism an-ti-dis-es-tab-lish-ment-ta-ri-an-ism
 What do you do when you encounter a very long, difficult word? 1 antidisestablishmenttarianism break it up into syllables: an-ti-dis-es-tab-lish-ment-ta-ri-an-ism meaning: antidisestablishmenttarianism
What do you do when you encounter a very long, difficult word? 1 antidisestablishmenttarianism break it up into syllables: an-ti-dis-es-tab-lish-ment-ta-ri-an-ism meaning: antidisestablishmenttarianism
Chapter 20 Carboxylic Acid Derivatives. Nucleophilic Acyl Substitution
 ucleophilic Acyl Substitution hapter 20 arboxylic Acid Derivatives ucleophilic Acyl Substitution Y (1) need to have Y as a u Y u u + Y (2) could not happen with aldehydes or ketones as : and : are poor
ucleophilic Acyl Substitution hapter 20 arboxylic Acid Derivatives ucleophilic Acyl Substitution Y (1) need to have Y as a u Y u u + Y (2) could not happen with aldehydes or ketones as : and : are poor
The PhilOEsophy. There are only two fundamental molecular descriptors
 The PhilOEsophy There are only two fundamental molecular descriptors Where can we use shape? Virtual screening More effective than 2D Lead-hopping Shape analogues are not graph analogues Molecular alignment
The PhilOEsophy There are only two fundamental molecular descriptors Where can we use shape? Virtual screening More effective than 2D Lead-hopping Shape analogues are not graph analogues Molecular alignment
Chapter 17: Carbonyl Compounds II
 Chapter 17: Carbonyl Compounds II Learning bjectives: 1. ecognize and assign names to aldehydes and ketones. 2. Write the mechanism for nucleophilic addition and nucleophilic addition-elimination reactions
Chapter 17: Carbonyl Compounds II Learning bjectives: 1. ecognize and assign names to aldehydes and ketones. 2. Write the mechanism for nucleophilic addition and nucleophilic addition-elimination reactions
Receptor Based Drug Design (1)
 Induced Fit Model For more than 100 years, the behaviour of enzymes had been explained by the "lock-and-key" mechanism developed by pioneering German chemist Emil Fischer. Fischer thought that the chemicals
Induced Fit Model For more than 100 years, the behaviour of enzymes had been explained by the "lock-and-key" mechanism developed by pioneering German chemist Emil Fischer. Fischer thought that the chemicals
JBA 2018 Chemistry Exam 2. Name: Score: /100 = /80
 JBA 2018 hemistry Exam 2 ame: Score: /100 = /80 Multiple choice questions are worth two points each. 1. onstitutional isomers are compounds that have a. the same chemical formulas and molecular structures
JBA 2018 hemistry Exam 2 ame: Score: /100 = /80 Multiple choice questions are worth two points each. 1. onstitutional isomers are compounds that have a. the same chemical formulas and molecular structures
Structure-Activity Modeling - QSAR. Uwe Koch
 Structure-Activity Modeling - QSAR Uwe Koch QSAR Assumption: QSAR attempts to quantify the relationship between activity and molecular strcucture by correlating descriptors with properties Biological activity
Structure-Activity Modeling - QSAR Uwe Koch QSAR Assumption: QSAR attempts to quantify the relationship between activity and molecular strcucture by correlating descriptors with properties Biological activity
Unit 5: Organic Chemistry
 Unit 5: Organic Chemistry Organic chemistry: discipline in chemistry focussing strictly on the study of hydrocarbons compounds made up of carbon & hydrogen Organic compounds can contain other elements
Unit 5: Organic Chemistry Organic chemistry: discipline in chemistry focussing strictly on the study of hydrocarbons compounds made up of carbon & hydrogen Organic compounds can contain other elements
Complete Volume List for Science of Synthesis Volumes
 Complete List for Science of Synthesis s Knowledge Update s Publication Date 1 Complete List for Science of Synthesis, by Georg Thieme Verlag KG ISBN/DOI Knowledge Update 2018/1 2018 502 978-3-13-242313-8
Complete List for Science of Synthesis s Knowledge Update s Publication Date 1 Complete List for Science of Synthesis, by Georg Thieme Verlag KG ISBN/DOI Knowledge Update 2018/1 2018 502 978-3-13-242313-8
Chapter 18: Carbonyl Compounds II
 Chapter 18: Carbonyl Compounds II Learning bjectives: 1. ecognize and assign names to aldehydes and ketones. 2. Write the mechanism for nucleophilic addition and nucleophilic addition-elimination reactions
Chapter 18: Carbonyl Compounds II Learning bjectives: 1. ecognize and assign names to aldehydes and ketones. 2. Write the mechanism for nucleophilic addition and nucleophilic addition-elimination reactions
Chemistry 343- Spring 2008
 Chemistry 343- Spring 2008 27 Chapter 2- Representative Carbon Compounds: Functional Groups, Intermolecular Forces and IR Spectroscopy A. ydrocarbons: Compounds composed of only C and Four Basic Types:
Chemistry 343- Spring 2008 27 Chapter 2- Representative Carbon Compounds: Functional Groups, Intermolecular Forces and IR Spectroscopy A. ydrocarbons: Compounds composed of only C and Four Basic Types:
Sul Ross State University Syllabus for Organic Chemistry II: CHEM 3408 (Spring 2017)
 Sul Ross State University Syllabus for Organic Chemistry II: CHEM 3408 (Spring 2017) Class: Organic Chemistry II Instructor: Dr. David J. Leaver Room: WSB 307 Office: WSB 318 Time: MWF 9:00-9:50am Office
Sul Ross State University Syllabus for Organic Chemistry II: CHEM 3408 (Spring 2017) Class: Organic Chemistry II Instructor: Dr. David J. Leaver Room: WSB 307 Office: WSB 318 Time: MWF 9:00-9:50am Office
ORGANIC - BRUICE 8E CH.3 - AN INTRODUCTION TO ORGANIC COMPOUNDS
 !! www.clutchprep.com CONCEPT: INDEX OF HYDROGEN DEFICIENCY (STRUCTURAL) A saturated molecule is any molecule that has the maximum number of hydrogens possible for its chemical structure. The rule that
!! www.clutchprep.com CONCEPT: INDEX OF HYDROGEN DEFICIENCY (STRUCTURAL) A saturated molecule is any molecule that has the maximum number of hydrogens possible for its chemical structure. The rule that
Classifying Compounds in Public Databases
 Classifying Compounds in Public Databases ACS San Diego, March 15, 2016 OntoChem IT Solutions GmbH Blücherstr. 24 06120 Halle (Saale) Germany Tel. +49 345 4780470 Fax: +49 345 4780471 mail: info(at)ontochem.com
Classifying Compounds in Public Databases ACS San Diego, March 15, 2016 OntoChem IT Solutions GmbH Blücherstr. 24 06120 Halle (Saale) Germany Tel. +49 345 4780470 Fax: +49 345 4780471 mail: info(at)ontochem.com
Development of a Structure Generator to Explore Target Areas on Chemical Space
 Development of a Structure Generator to Explore Target Areas on Chemical Space Kimito Funatsu Department of Chemical System Engineering, This materials will be published on Molecular Informatics Drug Development
Development of a Structure Generator to Explore Target Areas on Chemical Space Kimito Funatsu Department of Chemical System Engineering, This materials will be published on Molecular Informatics Drug Development
Chemistry 234 Exam 3. The Periodic Table
 ame: Last First MI Chemistry 234 Exam 3 Fall 2017 Dr. J. sbourn Instructions: The first 24 questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in the Scantron
ame: Last First MI Chemistry 234 Exam 3 Fall 2017 Dr. J. sbourn Instructions: The first 24 questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in the Scantron
The reuse of structural data for fragment binding site prediction
 The reuse of structural data for fragment binding site prediction Richard Hall 1 Motivation many examples of fragments binding in a phenyl shaped pocket or a kinase slot good shape complementarity between
The reuse of structural data for fragment binding site prediction Richard Hall 1 Motivation many examples of fragments binding in a phenyl shaped pocket or a kinase slot good shape complementarity between
Suggested solutions for Chapter 29
 s for Chapter 29 29 PRBLEM 1 or each of the following reactions (a) state what kind of substitution is suggested and (b) suggest what product might be formed if monosubstitution occured. Br 2 3 2 S 4 S
s for Chapter 29 29 PRBLEM 1 or each of the following reactions (a) state what kind of substitution is suggested and (b) suggest what product might be formed if monosubstitution occured. Br 2 3 2 S 4 S
Chimica Farmaceutica (Insegnamento Integrato di Chimica e Biotecnologie Farmaceutiche) Drug design (2)
 (Insegnamento Integrato di Chimica e Biotecnologie Farmaceutiche) Drug design (2) Molecular Variations in Homologous Series: Vinylogues and Benzologues HOMOLOGOUS SERIES. Definition and classification
(Insegnamento Integrato di Chimica e Biotecnologie Farmaceutiche) Drug design (2) Molecular Variations in Homologous Series: Vinylogues and Benzologues HOMOLOGOUS SERIES. Definition and classification
Comprehensive Organic Functional Group Transformations
 Comprehensive Organic Functional Group Transformations Editors-in-Chief Alan R. Katritzky, FRS University of Florida, Gainesville, FL, USA Otto Meth-Cohn University of Sunderland, UK Charles W. Rees, FRS
Comprehensive Organic Functional Group Transformations Editors-in-Chief Alan R. Katritzky, FRS University of Florida, Gainesville, FL, USA Otto Meth-Cohn University of Sunderland, UK Charles W. Rees, FRS
Organic Chemistry SL IB CHEMISTRY SL
 Organic Chemistry SL IB CHEMISTRY SL 10.1 Fundamentals of organic chemistry Understandings: A homologous series is a series of compounds of the same family, with the same general formula, which differ
Organic Chemistry SL IB CHEMISTRY SL 10.1 Fundamentals of organic chemistry Understandings: A homologous series is a series of compounds of the same family, with the same general formula, which differ
A Deep Convolutional Neural Network for Bioactivity Prediction in Structure-based Drug Discovery
 AtomNet A Deep Convolutional Neural Network for Bioactivity Prediction in Structure-based Drug Discovery Izhar Wallach, Michael Dzamba, Abraham Heifets Victor Storchan, Institute for Computational and
AtomNet A Deep Convolutional Neural Network for Bioactivity Prediction in Structure-based Drug Discovery Izhar Wallach, Michael Dzamba, Abraham Heifets Victor Storchan, Institute for Computational and
Loudon Chapter 20 & 21 Review: Carboxylic Acids & Derivatives CHEM 3331, Jacquie Richardson, Fall Page 1
 Loudon Chapter 20 & 21 eview: Carboxylic Acids & Derivatives CEM 3331, Jacquie ichardson, Fall 2010 - Page 1 These two chapters cover compounds which are all at the three bonds to more electronegative
Loudon Chapter 20 & 21 eview: Carboxylic Acids & Derivatives CEM 3331, Jacquie ichardson, Fall 2010 - Page 1 These two chapters cover compounds which are all at the three bonds to more electronegative
Symmetric Stretch: allows molecule to move through space
 BACKGROUND INFORMATION Infrared Spectroscopy Before introducing the subject of IR spectroscopy, we must first review some aspects of the electromagnetic spectrum. The electromagnetic spectrum is composed
BACKGROUND INFORMATION Infrared Spectroscopy Before introducing the subject of IR spectroscopy, we must first review some aspects of the electromagnetic spectrum. The electromagnetic spectrum is composed
Organic Chemistry Unit Review Package
 Name: Worksheet 7.viii Organic Chemistry Unit Review Package Generalized Organic Chemistry Naming Procedure Grouped into three general phases. 1. Identification phase (finding all important/correct information)
Name: Worksheet 7.viii Organic Chemistry Unit Review Package Generalized Organic Chemistry Naming Procedure Grouped into three general phases. 1. Identification phase (finding all important/correct information)
Montgomery County Community College CHE 122 General Chemistry-Organic (For the Non-Science Major) 4-3-3
 Montgomery County Community College CHE 122 General Chemistry-Organic (For the Non-Science Major) 4-3-3 COURSE DESCRIPTION: This course emphasizes introductory Organic Chemistry and Biochemistry. The examination
Montgomery County Community College CHE 122 General Chemistry-Organic (For the Non-Science Major) 4-3-3 COURSE DESCRIPTION: This course emphasizes introductory Organic Chemistry and Biochemistry. The examination
ORGANIC CHEMISTRY. Wiley STUDY GUIDE AND SOLUTIONS MANUAL TO ACCOMPANY ROBERT G. JOHNSON JON ANTILLA ELEVENTH EDITION. University of South Florida
 STUDY GUIDE AND SOLUTIONS MANUAL TO ACCOMPANY ORGANIC CHEMISTRY ELEVENTH EDITION T. W. GRAHAM SOLOMONS University of South Florida CRAIG B. FRYHLE Pacific Lutheran University SCOTT A. SNYDER Columbia University
STUDY GUIDE AND SOLUTIONS MANUAL TO ACCOMPANY ORGANIC CHEMISTRY ELEVENTH EDITION T. W. GRAHAM SOLOMONS University of South Florida CRAIG B. FRYHLE Pacific Lutheran University SCOTT A. SNYDER Columbia University
Infrared Spectroscopy used to analyze the presence of functional groups (bond types) in organic molecules How IR spectroscopy works:
 Infrared Spectroscopy used to analyze the presence of functional groups (bond types) in organic molecules It is the study of the interaction of infrared energy with organic molecules; the process analyzes
Infrared Spectroscopy used to analyze the presence of functional groups (bond types) in organic molecules It is the study of the interaction of infrared energy with organic molecules; the process analyzes
