Chemistry Lab. Regina Zibuck
|
|
|
- Madeline Margaret Mosley
- 6 years ago
- Views:
Transcription
1 Chemistry Lab Regina Zibuck
2 Description One or more tasks/experiments Answer questions involving the science processes of chemistry Kinetics Gases (new)
3 Routine Information Interpretation of experimental data (tabular and/or graphic) Observation of an experiment set up and running Computer or calculator sensors/probes Stoichiometry : mole conversions and percentage yield
4 Routine Information Nomenclature and formula writing - symbols and charges for the polyatomic ions by memory: nitrate, carbonate, phosphate, acetate, sulfate, ammonium, bicarbonate, and hydroxide ( ite forms of ates listed)
5 Safety Students must wear: Closed shoes Slacks or skirts that come to the ankles Lab coat or lab apron Long-Sleeved Shirt (if wearing a lab apron) Indirect vent or unvented chemical splash proof goggles. Eye protection C. Long hair must be tied back Gloves are optional
6 What Students Should Bring Safety gear Something to write with FIVE 8.5 X 11 sheets of paper (double sided, sheet protectors are allowed) TWO non-camera calculators
7 What the Supervisor Provides Everything the student will need This may include: Glassware Reagents Balances Hot plates Thermometers Probes Stirrers A periodic table and any constants
8 Changes for new topic & 1 old topic: Kinetics Gases
9 How to prepare participants Make sure students read the directions and pay particular attention to the description of the event (The Competition) Have them do many experiments together Have them determine their individual strengths Divide (and conquer) tasks during competition Check each other s work
10 How to Prepare Use a college level General (Freshman) Chemistry textbook Chemistry by Zumdahl and Zumdahl Start with a Prep-Gen Chem book if lower level background material needed Introductory Chemistry by Zumdahl
11 How to Prepare Kinetics Reaction rates Rate Laws The integrated rate law Reaction mechanisms Descriptive kinetics Catalysis
12 How to Prepare Gases The ideal gas law and all variations Boyle s Law Charles Law Avogadro s Law Collection of gas over water
13 Gases Other gas laws: Dalton s Law of Partial Pressures: The total pressure of a mixture of gases equals the sum of the individual partial pressures of each gas in the mixture. P total = P 1 + P 2 + P 3 + Gay-Lussac s Law (the law of combining volumes): At a given pressure and temperature, the volumes of gases reacting with each other are in the ratios of small whole numbers.
14 Gases Determining the density of a gas: D = m/v = nm/v = PM/RT Determining the molar mass of a gas: M = DRT/P = mrt/pv Determining the partial pressure of a gas: P 1 = X 1 P total where X 1 is the mole fraction of a gas. X 1 = n 1 /n total
15 How to prepare Search for experiments that fit the topics Classic kinetics Classic gas laws
16 Kinetics Activities Students can perform iodine clock reactions to determine the effect of concentration on the rate of the reaction. Students can look at reactions that produce a gas and either use soap bubbles or a gas probe to determine the concentration of the gas. This reaction can look at the effect of varying concentration of reactants It can look at varying the temperature of either one of the reactants.
17 Kinetics Activities Students can look at the reaction of an acid with metal and see how the size of the metal effects the rate. Students can look at the breakdown of starch by salivary amylase to see the effects of a catalyst Students can look at the breakdown of fats by pancreatic lipase with and without bile salts
18 Kinetics Activities t Science, chemistry, kinetics
19 Gas Activities Determine the density of a gas Partial pressure of a gas Molar mass of a gas Relative rates of diffusion
20 Gas Activities Examine relationship between pressure and volume Examine relationship between pressure and temperature Examine relationship between temperature and volume
21 Gas Activities ubject Science, chemistry, gases
22 Scoring 50% Kinetics 50% Gases
Chemistry Lab. By Lin Wozniewski
 Chemistry Lab By Lin Wozniewski lwoz@iun.edu Disclaimer This presentation was prepared using draft rules. There may be some changes in the final copy of the rules. The rules which will be in your Coaches
Chemistry Lab By Lin Wozniewski lwoz@iun.edu Disclaimer This presentation was prepared using draft rules. There may be some changes in the final copy of the rules. The rules which will be in your Coaches
Chemistry Lab Division C. Presenters: Phoebe Shih & Arya Pontula North Carolina Science Olympiad
 Chemistry Lab-2019 Division C Presenters: Phoebe Shih & Arya Pontula pshih130@cccc.edu Required for the event: Category C Eye Protection: Safety goggles with Indirect Vent such as the two shown on above
Chemistry Lab-2019 Division C Presenters: Phoebe Shih & Arya Pontula pshih130@cccc.edu Required for the event: Category C Eye Protection: Safety goggles with Indirect Vent such as the two shown on above
Kinetics of an Iodine Clock Reaction Lab_Student Copy
 Kinetics of an Iodine Clock Reaction Lab_Student Copy Purpose: Purpose: In this lab, you will find the reaction rate, rate law,, and observe the effects of a catalyst for the oxidation of iodide ions by
Kinetics of an Iodine Clock Reaction Lab_Student Copy Purpose: Purpose: In this lab, you will find the reaction rate, rate law,, and observe the effects of a catalyst for the oxidation of iodide ions by
Kinetics of an Iodine Clock Reaction Lab_ Teacher s Key
 Kinetics of an Iodine Clock Reaction Lab_ Teacher s Key Purpose: In this lab, you will find the reaction rate, rate law,, and observe the effects of a catalyst for the oxidation of iodide ions by bromate
Kinetics of an Iodine Clock Reaction Lab_ Teacher s Key Purpose: In this lab, you will find the reaction rate, rate law,, and observe the effects of a catalyst for the oxidation of iodide ions by bromate
General Chemistry 201 Section ABC Harry S. Truman College Spring Semester 2014
 Instructor: Michael Davis Office: 3226 Phone: 773 907 4718 Office Hours: Tues 9:00 12:00 Wed 1:00 3:00 Thurs 9:00 12:00 Email: mdavis@ccc.edu Website: http://faradaysclub.com http://ccc.blackboard.com
Instructor: Michael Davis Office: 3226 Phone: 773 907 4718 Office Hours: Tues 9:00 12:00 Wed 1:00 3:00 Thurs 9:00 12:00 Email: mdavis@ccc.edu Website: http://faradaysclub.com http://ccc.blackboard.com
8 Enthalpy of Reaction
 E x p e r i m e n t Enthalpy of Reaction Lecture and Lab Skills Emphasized Calculating the heat and enthalpy of reactions. Writing net ionic equations. Using Hess s law to determine the enthalpy of a reaction.
E x p e r i m e n t Enthalpy of Reaction Lecture and Lab Skills Emphasized Calculating the heat and enthalpy of reactions. Writing net ionic equations. Using Hess s law to determine the enthalpy of a reaction.
Research Experiences for Teachers (RET) 2015 LESSON PLAN TEMPLATE
 LESSON PLAN TEMPLATE LESSON TOPIC: Molarity of a solution STANDARD(S) & INDICATOR(S): CCSS Mathematics: HSF-IF. Interpret functions that arise in applications in terms of the context. OBJECTIVE(S): Students
LESSON PLAN TEMPLATE LESSON TOPIC: Molarity of a solution STANDARD(S) & INDICATOR(S): CCSS Mathematics: HSF-IF. Interpret functions that arise in applications in terms of the context. OBJECTIVE(S): Students
Chemistry: Final Exam Review. June, 2017 Mrs. Barbarito, Mrs. Corcoran, Ms. Guglielmo
 Chemistry: Final Exam Review June, 2017 Mrs. Barbarito, Mrs. Corcoran, Ms. Guglielmo 1 CHEMISTRY FINAL EXAM FORMAT TIME: 1.5 HOURS 1. MULTIPLE CHOICE 2. BRIEF ANSWERS AND PROBLEMS 3. CONTENT READING WITH
Chemistry: Final Exam Review June, 2017 Mrs. Barbarito, Mrs. Corcoran, Ms. Guglielmo 1 CHEMISTRY FINAL EXAM FORMAT TIME: 1.5 HOURS 1. MULTIPLE CHOICE 2. BRIEF ANSWERS AND PROBLEMS 3. CONTENT READING WITH
Apply the ideal gas law (PV = nrt) to experimentally determine the number of moles of carbon dioxide gas generated
 Teacher Information Ideal Gas Law Objectives Determine the number of moles of carbon dioxide gas generated during a reaction between hydrochloric acid and sodium bicarbonate. Through this investigation,
Teacher Information Ideal Gas Law Objectives Determine the number of moles of carbon dioxide gas generated during a reaction between hydrochloric acid and sodium bicarbonate. Through this investigation,
Chemistry 1
 WTCS Repository 10-806-127 Chemistry 1 Course Outcome Summary Course Information Description Total Credits 4.00 Fundamental concepts of inorganic chemistry. Emphasizes learning the basic principles and
WTCS Repository 10-806-127 Chemistry 1 Course Outcome Summary Course Information Description Total Credits 4.00 Fundamental concepts of inorganic chemistry. Emphasizes learning the basic principles and
Credits: 4 Lecture Hours: 3 Lab/Studio Hours: 3
 Code: CHEM-100 Title: Principles of Chemistry Institute: STEM Department: Chemistry Course Description: The student will be able to identify and interpret the basic concepts of inorganic chemistry including
Code: CHEM-100 Title: Principles of Chemistry Institute: STEM Department: Chemistry Course Description: The student will be able to identify and interpret the basic concepts of inorganic chemistry including
Kinetics of Crystal Violet Fading AP Chemistry Big Idea 4, Investigation 11 An Advanced Inquiry Lab (adapted by Flinn Scientific, Inc.
 Introduction Kinetics of Crystal Violet Fading AP Chemistry Big Idea 4, Investigation 11 An Advanced Inquiry Lab (adapted by Flinn Scientific, Inc.) Crystal violet is a common, beautiful purple dye. In
Introduction Kinetics of Crystal Violet Fading AP Chemistry Big Idea 4, Investigation 11 An Advanced Inquiry Lab (adapted by Flinn Scientific, Inc.) Crystal violet is a common, beautiful purple dye. In
Unit 1: Matter, Chemical Trends, and Chemical Bonding
 Unit : Matter, Chemical Trends, and Chemical Bonding Every element has predictable chemical and physical properties determined by its structure. The type of chemical bond in a compound determines the physical
Unit : Matter, Chemical Trends, and Chemical Bonding Every element has predictable chemical and physical properties determined by its structure. The type of chemical bond in a compound determines the physical
Kinetics of Crystal Violet Fading AP* Chemistry Big Idea 4, Investigation 11 An Advanced Inquiry Lab
 Introduction Kinetics of Crystal Violet Fading AP* Chemistry Big Idea 4, Investigation 11 An Advanced Inquiry Lab Catalog o. AP7644S Publication o. 7644S Crystal violet is a common, beautiful purple dye.
Introduction Kinetics of Crystal Violet Fading AP* Chemistry Big Idea 4, Investigation 11 An Advanced Inquiry Lab Catalog o. AP7644S Publication o. 7644S Crystal violet is a common, beautiful purple dye.
POTIONS AND POISONS. Lin Wozniewski
 POTIONS AND POISONS Lin Wozniewski lwoz@iun.edu DISCLAIMER This presentation was prepared using draft rules. There may be some changes in the final copy of the rules. The rules which will be posted on
POTIONS AND POISONS Lin Wozniewski lwoz@iun.edu DISCLAIMER This presentation was prepared using draft rules. There may be some changes in the final copy of the rules. The rules which will be posted on
LAGUARDIA COMMUNITY COLLEGE CITY UNIVERSITY OF NEW YORK NATURAL SCIENCES DEPARTMENT. SCC105: Introduction to Chemistry Fall I 2014
 LAGUARDIA COMMUNITY COLLEGE CITY UNIVERSITY OF NEW YORK NATURAL SCIENCES DEPARTMENT SCC105: Introduction to Chemistry Fall I 2014 Your Instructor's name Your Instructor's contact information Course Description:
LAGUARDIA COMMUNITY COLLEGE CITY UNIVERSITY OF NEW YORK NATURAL SCIENCES DEPARTMENT SCC105: Introduction to Chemistry Fall I 2014 Your Instructor's name Your Instructor's contact information Course Description:
CHEM 1B Chemistry I, Second Semester PR-10288, BK (v.3.0) To the Student:
 CHEM 1B Chemistry I, Second Semester PR-10288, BK-10289 (v.3.0) To the Student: After your registration is complete and your proctor has been approved, you may take the Credit by Examination for CHEM 1B.
CHEM 1B Chemistry I, Second Semester PR-10288, BK-10289 (v.3.0) To the Student: After your registration is complete and your proctor has been approved, you may take the Credit by Examination for CHEM 1B.
Chemistry CP Putting It All Together II
 Chemistry CP Putting It All Together II Name: Date: Calculations in Chemistry It s time to pull out your calculators! In the first review sheet, you were able to write formulas of compounds when different
Chemistry CP Putting It All Together II Name: Date: Calculations in Chemistry It s time to pull out your calculators! In the first review sheet, you were able to write formulas of compounds when different
Worksheet 1.1. Chapter 1: Quantitative chemistry glossary
 Worksheet 1.1 Chapter 1: Quantitative chemistry glossary Amount The number of moles of a substance present in a sample. Aqueous solution A solution with water as the solvent. Atmosphere The unit atmosphere
Worksheet 1.1 Chapter 1: Quantitative chemistry glossary Amount The number of moles of a substance present in a sample. Aqueous solution A solution with water as the solvent. Atmosphere The unit atmosphere
Chemistry CP Lab: Additivity of Heats of Reaction (Hess Law)
 Chemistry CP Lab: Additivity of Heats of Reaction (Hess Law) Name: Date: The formation or destruction of chemical bonds is always accompanied by an energy exchange between the reactant molecules and the
Chemistry CP Lab: Additivity of Heats of Reaction (Hess Law) Name: Date: The formation or destruction of chemical bonds is always accompanied by an energy exchange between the reactant molecules and the
Thermal Energy and Temperature Lab. Experiment Question: How can the difference between thermal energy and temperature be experimentally observed?
 Thermal Energy and Temperature Lab Name 7 th Grade PSI Grade / 20 Experiment Question: How can the difference between thermal energy and temperature be experimentally observed? Hypothesis Starters: 1.
Thermal Energy and Temperature Lab Name 7 th Grade PSI Grade / 20 Experiment Question: How can the difference between thermal energy and temperature be experimentally observed? Hypothesis Starters: 1.
Designing a Hand Warmer AP* Chemistry Big Idea 5, Investigation 12 An Advanced Inquiry Lab
 Introduction Designing a Hand Warmer AP* Chemistry Big Idea 5, Investigation 12 An Advanced Inquiry Lab Catalog No. AP7654 Publication No. 7654 WEB Put your chemistry skills to commercial use! From instant
Introduction Designing a Hand Warmer AP* Chemistry Big Idea 5, Investigation 12 An Advanced Inquiry Lab Catalog No. AP7654 Publication No. 7654 WEB Put your chemistry skills to commercial use! From instant
Chem. I Notes Ch. 11 STOICHIOMETRY NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics.
 Chem. I Notes Ch. 11 STOICHIOMETRY NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics. 11.1 notes 1 MOLE = 6.02 x 10 23 representative particles representative particles
Chem. I Notes Ch. 11 STOICHIOMETRY NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics. 11.1 notes 1 MOLE = 6.02 x 10 23 representative particles representative particles
WEST LOS ANGELES COLLEGE Introduction to General Chemistry CHEMISTRY 60 SYLLABUS; 5 units
 WEST LOS ANGELES COLLEGE Introduction to General Chemistry CHEMISTRY 60 SYLLABUS; 5 units SPRING 2014; Section 3492 Instructor Information Instructor E-mail Lecture Room MSA 005 Laboratory Session Room
WEST LOS ANGELES COLLEGE Introduction to General Chemistry CHEMISTRY 60 SYLLABUS; 5 units SPRING 2014; Section 3492 Instructor Information Instructor E-mail Lecture Room MSA 005 Laboratory Session Room
University of International Business and Economics International Summer School
 University of International Business and Economics International Summer School CHE 110:Introduction to Chemistry (with Lab) Term: July 8 August 2, 2019 Instructor: Nan Chen Home Institution: Curry College
University of International Business and Economics International Summer School CHE 110:Introduction to Chemistry (with Lab) Term: July 8 August 2, 2019 Instructor: Nan Chen Home Institution: Curry College
Exam Review Sheet. Solutions and Solubility. Gases and Atmospheric Chemistry
 NORTH TORONTO C.I. SCH 3U Exam Review Sheet This is just a guideline of the possible topics covered on the final exam in June. Refer to your class notes, textbook, quizzes, lab notes, assignments, tests
NORTH TORONTO C.I. SCH 3U Exam Review Sheet This is just a guideline of the possible topics covered on the final exam in June. Refer to your class notes, textbook, quizzes, lab notes, assignments, tests
Biodiesel Fundamentals for High School Chemistry Classes. Laboratory 4: Chemical Equilibrium in Biodiesel
 Laboratory 4: Chemical Equilibrium in Biodiesel Production Topics Covered Forward chemical reactions vs. reverse reactions Chemical reactions in equilibrium Ways to stimulate a reaction to proceed towards
Laboratory 4: Chemical Equilibrium in Biodiesel Production Topics Covered Forward chemical reactions vs. reverse reactions Chemical reactions in equilibrium Ways to stimulate a reaction to proceed towards
Unit 1: Chemical Foundations: Lab Skills, Properties of Matter, Scientific Measurement, and Dimensional Analysis
 Curriculum at a Glance Chemistry Honors Level: 400 Grades 10-12 The curriculum for the Chemistry Honors 400 course is designed to provide higher performing students a strong, fundamental understanding
Curriculum at a Glance Chemistry Honors Level: 400 Grades 10-12 The curriculum for the Chemistry Honors 400 course is designed to provide higher performing students a strong, fundamental understanding
After your registration is complete and your proctor has been approved, you may take the Credit by Examination for Chemistry 1A.
 CHEM 1A Chemistry, First Semester #8437 (v.2.0) To the Student: After your registration is complete and your proctor has been approved, you may take the Credit by Examination for Chemistry 1A. WHAT TO
CHEM 1A Chemistry, First Semester #8437 (v.2.0) To the Student: After your registration is complete and your proctor has been approved, you may take the Credit by Examination for Chemistry 1A. WHAT TO
Thermodynamics Enthalpy of Reaction and Hess s Law
 P.O. Box 219 Batavia, Illinois 60510 1-800-452-1261 flinn@flinnsci.com Visit our website at: www.flinnsci.com 2003 Flinn Scientific, Inc. All Rights Reserved. Your Safer Source for Science Supplies Thermodynamics
P.O. Box 219 Batavia, Illinois 60510 1-800-452-1261 flinn@flinnsci.com Visit our website at: www.flinnsci.com 2003 Flinn Scientific, Inc. All Rights Reserved. Your Safer Source for Science Supplies Thermodynamics
The Eight Solution Problem Exploring Reactions of Aqueous Ionic Compounds
 The Eight Solution Problem Exploring Reactions of Aqueous Ionic Compounds About this Lesson This activity allows students to mix a variety of known ionic solutions while making careful observations. After
The Eight Solution Problem Exploring Reactions of Aqueous Ionic Compounds About this Lesson This activity allows students to mix a variety of known ionic solutions while making careful observations. After
Boyle s law states the relationship between the pressure and the volume of a sample of gas.
 The Ideal Gas Law Boyle s law states the relationship between the pressure and the volume of a sample of gas. Charles s law states the relationship between the volume and the absolute temperature of a
The Ideal Gas Law Boyle s law states the relationship between the pressure and the volume of a sample of gas. Charles s law states the relationship between the volume and the absolute temperature of a
Pacing Guide for 7-12 Curriculum
 Pacing Guide for 7-12 Curriculum Course Title: Length of Course: Week Number Week 1 Week 2 Chapter COS Objectives Orientation Lab Safety Lab Equipment Chapter 1: The Science of Chemistry AHSGE 1 COS 1
Pacing Guide for 7-12 Curriculum Course Title: Length of Course: Week Number Week 1 Week 2 Chapter COS Objectives Orientation Lab Safety Lab Equipment Chapter 1: The Science of Chemistry AHSGE 1 COS 1
HONORS CHEMISTRY Putting It All Together II
 NAME: SECTION: HONORS CHEMISTRY Putting It All Together II Calculations in Chemistry It s time to pull out your calculators! In the first review sheet, you were able to write formulas of compounds when
NAME: SECTION: HONORS CHEMISTRY Putting It All Together II Calculations in Chemistry It s time to pull out your calculators! In the first review sheet, you were able to write formulas of compounds when
Chemistry 112. Dr. Geldart.
 1 Chemistry 112 Dr. Geldart sgeldart@chm.uri.edu Material Covered in Course Raymond Chang: Chemistry: Essential Concepts 7 th Edition. Ch. 14-19 2 Ch. 14: Kinetics Ch. 15: Equilibrium Ch. 16: Acids and
1 Chemistry 112 Dr. Geldart sgeldart@chm.uri.edu Material Covered in Course Raymond Chang: Chemistry: Essential Concepts 7 th Edition. Ch. 14-19 2 Ch. 14: Kinetics Ch. 15: Equilibrium Ch. 16: Acids and
Reaction mixtures that have reached equilibrium are represted by chemical equaitions with a doubleheaded
 EXPERIMENT 24 LeChatelier s Principle INTRODUCTION Chemical reactions in which a product is essentially unionized, is given off as a gas, or is precipitated, may be thought of as running to completion.
EXPERIMENT 24 LeChatelier s Principle INTRODUCTION Chemical reactions in which a product is essentially unionized, is given off as a gas, or is precipitated, may be thought of as running to completion.
Interview Notes. m) calorimeter b) temperature. o) isolated system d) heat of reaction. p) enthalpy of formation e) heat capacity
 TOPIC 1: Heat of Neutralization - The Strong versus The Weak: The heats of neutralization for mixtures of strong and weak acids with strong and weak bases will be determined and compared. A calorimeter
TOPIC 1: Heat of Neutralization - The Strong versus The Weak: The heats of neutralization for mixtures of strong and weak acids with strong and weak bases will be determined and compared. A calorimeter
Chemistry (Master) Content Skills Learning Targets Assessment Resources & Technology
 St. Michael-Albertville High School Teacher: Joe Peatrowsky Chemistry (Master) August 2014 September 2014 CEQ Measurement, Matter, Measurement, Matter, and Measurement, Matter, and and Chemical Equations
St. Michael-Albertville High School Teacher: Joe Peatrowsky Chemistry (Master) August 2014 September 2014 CEQ Measurement, Matter, Measurement, Matter, and Measurement, Matter, and and Chemical Equations
1 (C) demonstrate an understanding of the use and conservation of resources and the proper disposal or recycling of materials.
 112.35. Chemistry, Beginning with School Year 2010-2011 (One Credit). (a) General requirements. Students shall be awarded one credit for successful completion of this course. Required prerequisites: one
112.35. Chemistry, Beginning with School Year 2010-2011 (One Credit). (a) General requirements. Students shall be awarded one credit for successful completion of this course. Required prerequisites: one
EXPERIMENT 20. Solutions INTRODUCTION
 EXPERIMENT 20 Solutions INTRODUCTION A solution is a homogeneous mixture. The solvent is the dissolving substance, while the solute is the dissolved substance. A saturated solution is one in which the
EXPERIMENT 20 Solutions INTRODUCTION A solution is a homogeneous mixture. The solvent is the dissolving substance, while the solute is the dissolved substance. A saturated solution is one in which the
Chemistry Curriculum Guide Scranton School District Scranton, PA
 Chemistry Scranton, PA Chemistry Prerequisite: Biology, concurrent with Algebra II This is a college preparatory chemistry course that is taken usually in grade 11. It is taken with or after Algebra II.
Chemistry Scranton, PA Chemistry Prerequisite: Biology, concurrent with Algebra II This is a college preparatory chemistry course that is taken usually in grade 11. It is taken with or after Algebra II.
Chemistry 20, Section 1204 Fundamentals of Chemistry, 5 Units El Camino College Spring 2008
 Chemistry 20, Section 1204 Fundamentals of Chemistry, 5 Units El Camino College Spring 2008 Instructor: Lida Latifzadeh Masoudipour, Ph.D. Lecture: M, 6:00 PM 9:10 PM, Room Chem. 133 W, 6:00 PM 7:00 PM,
Chemistry 20, Section 1204 Fundamentals of Chemistry, 5 Units El Camino College Spring 2008 Instructor: Lida Latifzadeh Masoudipour, Ph.D. Lecture: M, 6:00 PM 9:10 PM, Room Chem. 133 W, 6:00 PM 7:00 PM,
Miami Dade College CHM 1045 First Semester General Chemistry
 Miami Dade College CHM 1045 First Semester General Chemistry Course Description: CHM 1045 is the first semester of a two-semester general chemistry course for science, premedical science and engineering
Miami Dade College CHM 1045 First Semester General Chemistry Course Description: CHM 1045 is the first semester of a two-semester general chemistry course for science, premedical science and engineering
2007 Release Chemistry SOL Test
 Name: Score: 50 / 50 points (100%) 2007 Release Chemistry SOL Test Multiple Choice Identify the choice that best completes the statement or answers the question. B 1. What is the volume of the liquid in
Name: Score: 50 / 50 points (100%) 2007 Release Chemistry SOL Test Multiple Choice Identify the choice that best completes the statement or answers the question. B 1. What is the volume of the liquid in
Learning Objectives for Chemistry 173
 Learning Objectives for Chemistry 173 Glenbrook North High School Academic Year, 2017-2018 This outline provides a comprehensive list of the topics and concepts you will learn in this course. For each
Learning Objectives for Chemistry 173 Glenbrook North High School Academic Year, 2017-2018 This outline provides a comprehensive list of the topics and concepts you will learn in this course. For each
Texts: Chemistry: A Molecular Approach by Nivaldo Tro, (custom NCC edition) Prentice Hall 2008.
 General Chemistry II (CHE 132) Nassau Community College Dr. Charles Hicks Email: hicksc@ncc.edu Tel: 516-572-7969 Office: Cluster C - 2081 Office Hours: Tu & Th 10:00-11:15, or by appointment. Texts: Chemistry:
General Chemistry II (CHE 132) Nassau Community College Dr. Charles Hicks Email: hicksc@ncc.edu Tel: 516-572-7969 Office: Cluster C - 2081 Office Hours: Tu & Th 10:00-11:15, or by appointment. Texts: Chemistry:
Representing Alabama s Public Two-Year College System Alabama Department of Postsecondary Education
 Date Adopted: 1966 Date Reviewed: 1987, 2011 Date Revised: 1988, 1998, 2001, 2008, 2011, 2015 Representing Alabama s Public Two-Year College System Alabama Department of Postsecondary Education Jefferson
Date Adopted: 1966 Date Reviewed: 1987, 2011 Date Revised: 1988, 1998, 2001, 2008, 2011, 2015 Representing Alabama s Public Two-Year College System Alabama Department of Postsecondary Education Jefferson
Moorpark College Chemistry 11 Fall 2011 Instructor: Professor Gopal. Examination #2: Section Two October 17, Name: (print)
 Moorpark College Chemistry 11 Fall 2011 Instructor: Professor Gopal Examination #2: Section Two October 17, 2011 Name: (print) Directions: Make sure your examination contains ELEVEN total pages (including
Moorpark College Chemistry 11 Fall 2011 Instructor: Professor Gopal Examination #2: Section Two October 17, 2011 Name: (print) Directions: Make sure your examination contains ELEVEN total pages (including
Classifying Chemical Reactions Analyzing and Predicting Products
 Classifying Chemical Reactions Analyzing and Predicting Products Background A chemical reaction is defined as any process in which one or more substances are converted into new substances with different
Classifying Chemical Reactions Analyzing and Predicting Products Background A chemical reaction is defined as any process in which one or more substances are converted into new substances with different
Chemical Names and Formulas
 Cool Chemistry Show Section 3 Chemical Names and Formulas What Do You See? Learning Outcomes In this section you will Predict the charges of ions of some elements. Determine the formulas of ionic compounds.
Cool Chemistry Show Section 3 Chemical Names and Formulas What Do You See? Learning Outcomes In this section you will Predict the charges of ions of some elements. Determine the formulas of ionic compounds.
Bundle at a Glance Chemistry 2015/16
 Introduction: Scientific Investigation and Reasoning Skills (3 A/B days) Chemistry Process TEKS: 1A demonstrate safe practices during laboratory and field investigations, including the appropriate use
Introduction: Scientific Investigation and Reasoning Skills (3 A/B days) Chemistry Process TEKS: 1A demonstrate safe practices during laboratory and field investigations, including the appropriate use
CHEM 1413 Course Syllabus (CurricUNET) Course Syllabus
 Semester with Course Reference Number (CRN) CHEM 1413 Course Syllabus (CurricUNET) Course Syllabus College Chemistry I CHEM 1413 Instructor contact information (phone number and email address) Office Location
Semester with Course Reference Number (CRN) CHEM 1413 Course Syllabus (CurricUNET) Course Syllabus College Chemistry I CHEM 1413 Instructor contact information (phone number and email address) Office Location
CHEMISTRY Matter and Change. Chapter 13: Gases
 CHEMISTRY Matter and Change Chapter 13: Gases CHAPTER 13 Table Of Contents Section 13.1 Section 13.2 Section 13.3 The Gas Laws The Ideal Gas Law Gas Stoichiometry Click a hyperlink to view the corresponding
CHEMISTRY Matter and Change Chapter 13: Gases CHAPTER 13 Table Of Contents Section 13.1 Section 13.2 Section 13.3 The Gas Laws The Ideal Gas Law Gas Stoichiometry Click a hyperlink to view the corresponding
Ch. 10 Notes STOICHIOMETRY NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics.
 Ch. 10 Notes STOICHIOMETRY NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics. 1 MOLE = 6.02 x 10 23 representative particles representative particles = ATOMS, IONS,
Ch. 10 Notes STOICHIOMETRY NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics. 1 MOLE = 6.02 x 10 23 representative particles representative particles = ATOMS, IONS,
CHEMISTRY INTRODUCTORY CHEMISTRY
 BRAZOSPORT COLLEGE LAKE JACKSON, TEXAS SYLLABUS CHEMISTRY 1405 - INTRODUCTORY CHEMISTRY CATALOG DESCRIPTION: CHEM 1405 Introductory Chemistry. CIP 4005015103 A study of basic organic and inorganic chemistry
BRAZOSPORT COLLEGE LAKE JACKSON, TEXAS SYLLABUS CHEMISTRY 1405 - INTRODUCTORY CHEMISTRY CATALOG DESCRIPTION: CHEM 1405 Introductory Chemistry. CIP 4005015103 A study of basic organic and inorganic chemistry
Process Skills Review
 Process Skills Review Warm-Up Define function Match the following C. 1. Puts out fire 2. Curved line of liquid in a graduated cylinder 3. Used to observe insects A. B. Which of the following describes
Process Skills Review Warm-Up Define function Match the following C. 1. Puts out fire 2. Curved line of liquid in a graduated cylinder 3. Used to observe insects A. B. Which of the following describes
SL/HL 1 - Chemistry. Web: BrakkeIBchem1.wikispaces.com
 SL/HL 1 - Chemistry SL/HL 1 - Chemistry Instructor : Mr. Martin Brakke Email : martinb@eca.com.ve Web: BrakkeIBchem1.wikispaces.com Copy this web address and set a bookmark on your web browser. You will
SL/HL 1 - Chemistry SL/HL 1 - Chemistry Instructor : Mr. Martin Brakke Email : martinb@eca.com.ve Web: BrakkeIBchem1.wikispaces.com Copy this web address and set a bookmark on your web browser. You will
Total
 Student Performance by Question Chemistry (Multiple-Choice ONLY) Teacher: Core 1 / S-14 Scientific Investigation Atomic Structure and Periodic Relationships Analysis of Performance by Question of each
Student Performance by Question Chemistry (Multiple-Choice ONLY) Teacher: Core 1 / S-14 Scientific Investigation Atomic Structure and Periodic Relationships Analysis of Performance by Question of each
Chemistry Curriculum Map. Embedded in all standards. Chm Chm Chm Chm Writing:
 Six weeks Unit Unit Focus Chemistry Essential Standards Literacy in Science &Technical Subjects/ 8 Mathematical Practices First Introductory Chemistry Material - Density - Significant Digits - Metric Conversion
Six weeks Unit Unit Focus Chemistry Essential Standards Literacy in Science &Technical Subjects/ 8 Mathematical Practices First Introductory Chemistry Material - Density - Significant Digits - Metric Conversion
Montgomery County Community College CHE 131 Chemistry for Technology I 4-3-3
 Montgomery County Community College CHE 131 Chemistry for Technology I 4-3-3 COURSE DESCRIPTION: A general course in physical-inorganic and organic chemistry. Emphasis will be placed on the theoretical
Montgomery County Community College CHE 131 Chemistry for Technology I 4-3-3 COURSE DESCRIPTION: A general course in physical-inorganic and organic chemistry. Emphasis will be placed on the theoretical
CHEM 1B General Chemistry 5 Unit(s)
 I. Catalog Information Credit- Degree applicable Effective Quarter: Fall 2014 CHEM 1B General Chemistry 5 Unit(s) (See general education pages for the requirement this course meets.) Prerequisite: Chemistry
I. Catalog Information Credit- Degree applicable Effective Quarter: Fall 2014 CHEM 1B General Chemistry 5 Unit(s) (See general education pages for the requirement this course meets.) Prerequisite: Chemistry
Percentage of Acetic Acid in Vinegar
 Microscale Percentage of Acetic Acid in Vinegar When sweet apple cider is fermented in the absence of oxygen, the product is an acid, vinegar. Most commercial vinegars are made by fermentation, but some,
Microscale Percentage of Acetic Acid in Vinegar When sweet apple cider is fermented in the absence of oxygen, the product is an acid, vinegar. Most commercial vinegars are made by fermentation, but some,
Curriculum Catalog
 2017-2018 Curriculum Catalog 2017 Glynlyon, Inc. Table of Contents CHEMISTRY COURSE OVERVIEW...1 UNIT 1: MEASUREMENT AND ANALYSIS... 1 UNIT 2: STARTING THE INVESTIGATION: HOW TO IDENTIFY ELEMENTS, COMPOUNDS,
2017-2018 Curriculum Catalog 2017 Glynlyon, Inc. Table of Contents CHEMISTRY COURSE OVERVIEW...1 UNIT 1: MEASUREMENT AND ANALYSIS... 1 UNIT 2: STARTING THE INVESTIGATION: HOW TO IDENTIFY ELEMENTS, COMPOUNDS,
generate testable Students will be able to investigations. Biology 1 2 (can be conclusions. reveal relationships identify sources of error higher.
 Honors Chemistry Essential Questions: 1. How can one explain the structure, properties, and interactions of matter? Communication of Scientific Information Anchor Standard 1: Students will be able to design,
Honors Chemistry Essential Questions: 1. How can one explain the structure, properties, and interactions of matter? Communication of Scientific Information Anchor Standard 1: Students will be able to design,
Lab- Properties of Acids and Bases. Name. PSI Chemistry
 Lab- Properties of Acids and Bases PSI Chemistry Name Introduction Acids and bases are useful reagents in the chemistry laboratory and play an important role in biology and nature. What are acids and bases?
Lab- Properties of Acids and Bases PSI Chemistry Name Introduction Acids and bases are useful reagents in the chemistry laboratory and play an important role in biology and nature. What are acids and bases?
Kinetics of an Iodine Clock Reaction
 Kinetics of an Iodine Clock Reaction Introduction: In this experiment, you will determine the rate law for a reaction and the effect of concentration on the rate of the reaction by studying the initial
Kinetics of an Iodine Clock Reaction Introduction: In this experiment, you will determine the rate law for a reaction and the effect of concentration on the rate of the reaction by studying the initial
Acidity of Beverages Lab
 Acidity of Beverages Lab Name: Introduction: Common beverages may be either acidic or basic. Fruit juices, for example, get their sweet taste from sugars and their sour or tart taste from weak acids such
Acidity of Beverages Lab Name: Introduction: Common beverages may be either acidic or basic. Fruit juices, for example, get their sweet taste from sugars and their sour or tart taste from weak acids such
Chemistry Assessment. Eligible Texas Essential Knowledge and Skills
 Chemistry Assessment Eligible Texas Essential Knowledge and Skills STAAR Chemistry Assessment Reporting Category 1: Matter and the Periodic Table The student will demonstrate an understanding of the properties
Chemistry Assessment Eligible Texas Essential Knowledge and Skills STAAR Chemistry Assessment Reporting Category 1: Matter and the Periodic Table The student will demonstrate an understanding of the properties
OFFICE Room 3268; Tel Chemistry for Changing Times, 14 th Edition, -- Fourth Custom Edition for CCRI - by Hill and McCreary Other Supplies
 COMMUNITY COLLEGE of RHODE ISLAND Chemistry Department Course Information Package CHEM-1000, Chemistry of Our Environment Section 001 T, Th; 9:00 10:25 Lecture, Room 3046 T 1:00 3:50 Lab (Room 3046) Spring,
COMMUNITY COLLEGE of RHODE ISLAND Chemistry Department Course Information Package CHEM-1000, Chemistry of Our Environment Section 001 T, Th; 9:00 10:25 Lecture, Room 3046 T 1:00 3:50 Lab (Room 3046) Spring,
De Anza College Chemistry Department Spring 2018
 De Anza College Chemistry Department Spring 2018 COURSE TITLE Chemistry 1A-01 & 02 General Chemistry Class 04/09/18 to 06/28/18 Meeting times: Sec 01/02 Lecture 11:20 AM 12:20 PM, MWF, Room SC1102 Sec
De Anza College Chemistry Department Spring 2018 COURSE TITLE Chemistry 1A-01 & 02 General Chemistry Class 04/09/18 to 06/28/18 Meeting times: Sec 01/02 Lecture 11:20 AM 12:20 PM, MWF, Room SC1102 Sec
EXPERIMENT 6. Properties of Buffers INTRODUCTION
 EXPERIMENT 6 Properties of Buffers INTRODUCTION A chemical buffer is any substance in a solution that tends to stabilize the hydronium ion concentration by neutralizing any added acid or base. Buffers
EXPERIMENT 6 Properties of Buffers INTRODUCTION A chemical buffer is any substance in a solution that tends to stabilize the hydronium ion concentration by neutralizing any added acid or base. Buffers
10 th Grade Honors Chemistry
 10 th Grade Honors Chemistry Scranton School District Scranton, PA 10 th Grade Honors Chemistry Prerequisite: Honors Biology, Geometry Be in compliance with the SSD Honors and AP Criteria Policy 10 th
10 th Grade Honors Chemistry Scranton School District Scranton, PA 10 th Grade Honors Chemistry Prerequisite: Honors Biology, Geometry Be in compliance with the SSD Honors and AP Criteria Policy 10 th
Virginia Standards of Learning Chemistry
 correlated to the Virginia Standards of Learning Chemistry CC2 5/2004 2003 Science Standard Correlation By Page Numbers CH.1 The student will investigate and understand that experiments in which variables
correlated to the Virginia Standards of Learning Chemistry CC2 5/2004 2003 Science Standard Correlation By Page Numbers CH.1 The student will investigate and understand that experiments in which variables
A stand alone calculator (not part of your cell phone) (preferably a scientific calculator).
 COMMUNITY COLLEGE of RHODE ISLAND Chemistry Department Course Information Package CHEM-1000, Chemistry of Our Environment Section 001 T, Th; 9:00 10:25 Lecture, Room 3046 T 1:00 3:50 Lab (Room 3046) Spring,
COMMUNITY COLLEGE of RHODE ISLAND Chemistry Department Course Information Package CHEM-1000, Chemistry of Our Environment Section 001 T, Th; 9:00 10:25 Lecture, Room 3046 T 1:00 3:50 Lab (Room 3046) Spring,
Unit 1 Lab Equipment. Chemistry Midterm Review
 Chemistry Midterm Review Topics: Unit 1: laboratory equipment and safety rules accuracy vs precision scientific method: observation, hypothesis. experimental design: independent vs dependent variables,
Chemistry Midterm Review Topics: Unit 1: laboratory equipment and safety rules accuracy vs precision scientific method: observation, hypothesis. experimental design: independent vs dependent variables,
This image cannot currently be displayed. Course Catalog. Chemistry Glynlyon, Inc.
 This image cannot currently be displayed. Course Catalog Chemistry 2016 Glynlyon, Inc. Table of Contents COURSE OVERVIEW... 1 UNIT 1: MEASUREMENT AND ANALYSIS... 1 UNIT 2: STARTING THE INVESTIGATION: HOW
This image cannot currently be displayed. Course Catalog Chemistry 2016 Glynlyon, Inc. Table of Contents COURSE OVERVIEW... 1 UNIT 1: MEASUREMENT AND ANALYSIS... 1 UNIT 2: STARTING THE INVESTIGATION: HOW
Important Examples of Chemical Change
 Important Examples of Chemical Change chemical change: the change that occurs when substances react in a chemical reaction to create different substances When was the last time you watched an awesome display
Important Examples of Chemical Change chemical change: the change that occurs when substances react in a chemical reaction to create different substances When was the last time you watched an awesome display
Experiment 8 - Double Displacement Reactions
 Experiment 8 - Double Displacement Reactions A double displacement reaction involves two ionic compounds that are dissolved in water. In a double displacement reaction, it appears as though the ions are
Experiment 8 - Double Displacement Reactions A double displacement reaction involves two ionic compounds that are dissolved in water. In a double displacement reaction, it appears as though the ions are
Completion Match each each piece of equipment with its description. Please only put one number in the blank.
 LAB SAFETY AND EQUIPMENT TEST Completion Match each each piece of equipment with its description. Please only put one number in the blank. 1. Used for filtering and for adding chemicals without spilling.
LAB SAFETY AND EQUIPMENT TEST Completion Match each each piece of equipment with its description. Please only put one number in the blank. 1. Used for filtering and for adding chemicals without spilling.
LEVEL ZERO VOICE CATALYST (10 minutes, individual work): 1. Counting atoms
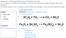 Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
The periodic table is the most recognized symbol in chemistry across the world. It is a valuable tool that allows scientists not only to classify the
 The periodic table is the most recognized symbol in chemistry across the world. It is a valuable tool that allows scientists not only to classify the elements but also to explain and predict their properties.
The periodic table is the most recognized symbol in chemistry across the world. It is a valuable tool that allows scientists not only to classify the elements but also to explain and predict their properties.
CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED)
 CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED) Overview: Students investigate chemical reactions using given substances. Students identify chemical reactions in their daily lives. Objectives: The student
CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED) Overview: Students investigate chemical reactions using given substances. Students identify chemical reactions in their daily lives. Objectives: The student
If you have a Mac do whatever you have to do to play it as a slide show I don t know Macs well. Dr. Buckley
 For best results please view this as a slide show. You can hit the F5 key or go to the Slide Show tab on the menu bar and click on From Beginning. Page Down and Page Up will move you through the presentation.
For best results please view this as a slide show. You can hit the F5 key or go to the Slide Show tab on the menu bar and click on From Beginning. Page Down and Page Up will move you through the presentation.
1) How are experiments designed and analyzed. 2) How are chemical formulas used? 1) How do you write and balance chemical equations
 GBCS Curriculum Guide GRADE: 11-12 Topic Pacing Unit Standards SUBJECT: AP Chemistry General Chemistry Review 1 Week Unit 1 CR1 AP At minimum, six of the required 16 labs are conducted in a guided-inquiry
GBCS Curriculum Guide GRADE: 11-12 Topic Pacing Unit Standards SUBJECT: AP Chemistry General Chemistry Review 1 Week Unit 1 CR1 AP At minimum, six of the required 16 labs are conducted in a guided-inquiry
Chemical Names and Formulas
 Cool Chemistry Show Activity 3 Chemical Names and Formulas GOALS In this activity you will: Predict the charges of ions of some elements. Determine the formulas of ionic compounds. Write the conventional
Cool Chemistry Show Activity 3 Chemical Names and Formulas GOALS In this activity you will: Predict the charges of ions of some elements. Determine the formulas of ionic compounds. Write the conventional
St. John s College High School Mr. Trubic AP Midterm Review Packet 1
 Name Date Directions: Read each question carefully and write your response in the space provided following each question. Your responses to these questions will be scored on the basis of the accuracy and
Name Date Directions: Read each question carefully and write your response in the space provided following each question. Your responses to these questions will be scored on the basis of the accuracy and
First Semester Review AP Chemistry 7 points DUE AT EXAM (Thurs., 1/25/18) Date:
 First Semester Review Name: AP Chemistry 7 points DUE AT EXAM (Thurs., 1/25/18) Date: The AP Chemistry Semester Exam on January 25, 2018, will cover textbook Chapters 3 through 10, our labs, and any additional
First Semester Review Name: AP Chemistry 7 points DUE AT EXAM (Thurs., 1/25/18) Date: The AP Chemistry Semester Exam on January 25, 2018, will cover textbook Chapters 3 through 10, our labs, and any additional
Gas Volumes and the Ideal Gas Law
 SECTION 11.3 Gas Volumes and the Ideal Gas Law Section 2 presented laws that describe the relationship between the pressure, temperature, and volume of a gas. The volume of a gas is also related to the
SECTION 11.3 Gas Volumes and the Ideal Gas Law Section 2 presented laws that describe the relationship between the pressure, temperature, and volume of a gas. The volume of a gas is also related to the
The Kinetics of the Iodine Clock Reaction
 Experiment 2 Pre-lab Assignment Before coming to lab: Read the lab thoroughly. Answer the pre-lab questions that appear at the end of this lab exercise. The questions should be answered on a separate (new)
Experiment 2 Pre-lab Assignment Before coming to lab: Read the lab thoroughly. Answer the pre-lab questions that appear at the end of this lab exercise. The questions should be answered on a separate (new)
1. Determine the mass of water that can be produced when 10.0g of hydrogen is combined with excess oxygen. 2 H 2 + O 2 2 H 2 O
 Pre-AP Chemistry Spring 2016 Final Review Objective 6.1: Students will recognize indicators of chemical change write balanced chemical equations to describe them based on common reactivity patterns. [S.12.C.1,
Pre-AP Chemistry Spring 2016 Final Review Objective 6.1: Students will recognize indicators of chemical change write balanced chemical equations to describe them based on common reactivity patterns. [S.12.C.1,
Student Book links Specification links Links to prior learning Suggested teaching order
 Teaching plan 5.1.2 Molecular formulae Student Book links Specification links Links to prior learning Suggested teaching order 5.1.2 5.03 5.04 5.05 Core practical 1 Learning objectives Empirical formulae
Teaching plan 5.1.2 Molecular formulae Student Book links Specification links Links to prior learning Suggested teaching order 5.1.2 5.03 5.04 5.05 Core practical 1 Learning objectives Empirical formulae
Gases. Section 13.1 The Gas Laws Section 13.2 The Ideal Gas Law Section 13.3 Gas Stoichiometry
 Gases Section 13.1 The Gas Laws Section 13.2 The Ideal Gas Law Section 13.3 Gas Stoichiometry Click a hyperlink or folder tab to view the corresponding slides. Exit Section 13.1 The Gas Laws State the
Gases Section 13.1 The Gas Laws Section 13.2 The Ideal Gas Law Section 13.3 Gas Stoichiometry Click a hyperlink or folder tab to view the corresponding slides. Exit Section 13.1 The Gas Laws State the
Shown below is a sample titration curve for a diprotic acid. Note the two equivalence points.
 EXPERIMENT 9 Titration Curve for a Polyprotic Acid INTRODUCTION Other than by strength and concentration, another way of classifying acids involves the number of H + ions an acid can donate. A monoprotic
EXPERIMENT 9 Titration Curve for a Polyprotic Acid INTRODUCTION Other than by strength and concentration, another way of classifying acids involves the number of H + ions an acid can donate. A monoprotic
CHEM 30A: Introductory General Chemistry Fall 2017, Laney College. Welcome to Chem 30A!
 CHEM 30A: Introductory General Chemistry Fall 2017, Laney College Welcome to Chem 30A! Meeting Time/Location Lecture (Class Code 41591): Sa 10:00AM 12:50PM in A233 Lab (Class Code 41592): Sa 1:30PM 4:20PM
CHEM 30A: Introductory General Chemistry Fall 2017, Laney College Welcome to Chem 30A! Meeting Time/Location Lecture (Class Code 41591): Sa 10:00AM 12:50PM in A233 Lab (Class Code 41592): Sa 1:30PM 4:20PM
Student Review Packet Answer Key
 Student Review acket Answer Key. Convert the following temperatures as indicated. a 0 o C to K 73 K e atm to ka 0.3 ka (s.f. = 00 b -0 o C to K 63 K f 0.878 atm to ka 88.9 ka c 45 o C to K 38 K g 3. atm
Student Review acket Answer Key. Convert the following temperatures as indicated. a 0 o C to K 73 K e atm to ka 0.3 ka (s.f. = 00 b -0 o C to K 63 K f 0.878 atm to ka 88.9 ka c 45 o C to K 38 K g 3. atm
Chem 1110 and Chem 106 Exam 2 Study Guide
 Chem 1110 and Chem 106 Exam 2 Study Guide Chapter 4 4.1 From the periodic table, determine the number of valence electrons in a Lewis dot diagram for an element. Master Tutor Section 4.1 Review Section
Chem 1110 and Chem 106 Exam 2 Study Guide Chapter 4 4.1 From the periodic table, determine the number of valence electrons in a Lewis dot diagram for an element. Master Tutor Section 4.1 Review Section
Course: CP Chemistry Year: Teacher: L. Page. Unit 2: Matter and Energy Approximate Time Frame: # of Weeks 4
 Course: CP Chemistry Year: 2013 14 Teacher: L. Page Unit 1: Matter and Change Approximate Time Frame: # of Weeks 4 Biological, chemical and physical properties of matter result from the ability of atoms
Course: CP Chemistry Year: 2013 14 Teacher: L. Page Unit 1: Matter and Change Approximate Time Frame: # of Weeks 4 Biological, chemical and physical properties of matter result from the ability of atoms
Rate of Reaction. Introduction
 5 Rate of Reaction Introduction This experiment will allow you to study the effects of concentration, temperature, and catalysts on a reaction rate. The reaction whose rate you will study is the oxidation
5 Rate of Reaction Introduction This experiment will allow you to study the effects of concentration, temperature, and catalysts on a reaction rate. The reaction whose rate you will study is the oxidation
Use this dramatic iodine clock reaction to demonstrate the effect of concentration, temperature, and a catalyst on the rate of a chemical reaction.
 Clock Reaction Race Reaction Pathways SCIENTIFIC Introduction Use this dramatic iodine clock reaction to demonstrate the effect of concentration temperature and a catalyst on the rate of a chemical reaction.
Clock Reaction Race Reaction Pathways SCIENTIFIC Introduction Use this dramatic iodine clock reaction to demonstrate the effect of concentration temperature and a catalyst on the rate of a chemical reaction.
CURRICULUM CATALOG CHEMISTRY (34202X0) NC
 2018-19 CURRICULUM CATALOG CHEMISTRY (34202X0) NC Table of Contents CHEMISTRY (34202X0) NC COURSE OVERVIEW... 2 UNIT 1: MEASUREMENT AND ANALYSIS... 2 UNIT 2: STARTING THE INVESTIGATION: HOW TO IDENTIFY
2018-19 CURRICULUM CATALOG CHEMISTRY (34202X0) NC Table of Contents CHEMISTRY (34202X0) NC COURSE OVERVIEW... 2 UNIT 1: MEASUREMENT AND ANALYSIS... 2 UNIT 2: STARTING THE INVESTIGATION: HOW TO IDENTIFY
