Salt Bridges: Aggregation, Hydration, and. Fragmentation of Peptides and Oligonucleotides
|
|
|
- Brice Day
- 6 years ago
- Views:
Transcription
1 Salt Bridges: Aggregation, Hydration, and Fragmentation of Peptides and ligonucleotides UCSB: Thomas Wyttenbach, Perdita Barran, Jennifer Gidden, Summer Bernstein, Dengfeng Liu, Mike Bowers U. Arizona: Linda Breci, Vicki Wysocki He idelberg: Be la P a izs $$ NSF (UCSB)
2 Structure of small peptides H/D exchange vs. ion mobility Aggregation of peptides Salt bridges in oligonucleotides?
3
4 Building accurate computer models for protein identification from MS/MS data requires knowledge of fragmentation mechanisms. Is the structure/conformation related to fragmentation pattern/mechanism? Does H/D exchange give information on peptide structure/conformation? Can H/D exchange and ion mobility data be structurally correlated?
5 Look at several series of peptides where important groups are systematically varied. Do H/D exchange and ion mobility studies on each group. Measure hydration energies. Do detailed ab initio/dft calculations on selected systems. Here we will discuss RAAAA, AARAA, AAAAR but focus on AARAA.
6 University of Arizona, Tucson University of California, Santa Barbara German Cancer Research Center Heidelberg
7 2N H 2 N NH different residue NH NH + H 2 N NH 2 NH NH NH NH NH DvsL NH NH H H NH
8 MH + MD + Relative Intensity MH + H/D m/z
9 MH + 3 fast exchanges AARAA H 2 N HN H N H Relative Intensity NH H 2 N MH + MH NH NH NH H 75 AARAA--CH 3 75 acetyl-aaraa m/z
10 ' 25HD\0HFKDQLVPIRU+'H[FKDQJH NH H NH NH D D NH H D NH D NH H &DPSEH5RGJHUV0DU]XII%HDXFKDPS -$&6
11 AARAA AARAA-Me Ac-AARAA Exchange YES N N %RWKWHUPLQLLQYRYHG 6DWEULGJH"
12 /RZHVWHQHUJ\VWUXFWXUH$0%( &KDUJHVRYDWLRQ =ZLWWHULRQ VDWEULGJH
13 Ion mobility method (UCSB) Ion Source MS1 Drift Cell MS2 Detector
14 Ion mobility method in out
15 ,RQ0RELLW\8&6% Experiment Calculated (zwitterion) Calculated (charge solvation) [Ac-AARAA]H+ [AARAA]H Cross section (Å 2 )
16 Peptide σ (Experiment) σ (AMBER Calculation) Neutral Terminus Zwitterions Terminus [RAAAA]H [AARAA]H [AAAAR]H [AcRAAAA]H [AcAARAA]H [AcAAAAR]H ( )
17 (AARAA)H + + H 2 (AARAA)H +
18 (Heidelberg) B3LYP/6-31g(d)
19 Structure Energy (kcal/mol) (AARAA)H + (AARAA)H + + H 2 Charge solvation Zwitterion ( NH 3+ ) Zwitterion (>C=H + )
20 (AARAA)H + + H 2 Zwitterion setup for relay mechanism: 14.1 kcal/mol + C-terminus + N-terminus H 2
21 (AARAA)H + + D 2 (AARAA)D + + HD 7 kcal/mol 16 kcal/mol Relay ZW Relay TS Relay CS ZW 2 kcal/mol 5 kcal/mol CS (AARAA)H + D 2
22 (AARAA)H + H 2 Theory (Heidelberg): 16 kcal/mol Experiment (UCSB): 10 kcal/mol (BSSE correction? Larger basis set?)
23 H/D-exchange: salt bridge (?) (AARAA)H + H 2 Ion mobility: no salt bridge (AARAA)H + Theory: no salt bridge for (AARAA)H + salt bridge for (AARAA)H + H 2 low TS for H/D-exchange for all 3 N-terminus hydrogens from salt bridge form no exchange possible for blocked termini since TS for proton transfer to >C= groups high in energy
24 Important in certain diseases -Alzheimers -Mad Cow (TSE) - Diabetes type II Important for chaperone formation bserved as (common) occurrence in electrospray of peptides Energetics and mechanism not yet known
25 Beauchamp et al.: penta-, hexa-peptides Clemmer et al.: BK, Insulin chain A, ala 12 Jarrold et al.: ac-lys-ala 19, ac-(gly-ala) 7 -lys Bowers et al.: BK, LHRH, neurotensin But little detailed understanding of factors that control this aggregation
26 Bradykinin 9 residue peptide 2 Arg 1 Acidic functional group Zwitterion formation H2 N PR3 PR2 C N ARG1 CH C N C GLY4 H N CH C H N H PHE5 CH C CH 2 SER6 H N CH C CH 2 H PR7 PHE8 ARG9 N C H N H 2 C CH C H N H 2 C CH C CH 2 CH 2 HN C NH 2 NH H CH 2 CH 2 CH 2 NH PGLU & &+ & + & & C NH PR9 NH HIS2 TRP3 SER4 TYR5 GLY6 LEU7 ARG8 & &+ & 1 &+ & 1 &+ & 1 &+ & 1 &+ & 1 &+ & 1 &+ & 1 + &+ &+ &+ &+ + &+ & &+ &+ &+ &+ + & 1+ & GLY10 2 &+ & 1+ + LHRH 10 residue peptide 1 Arg no acidic group Blocked Termini
27 Ion Arrival Time Distribution at m/z = 354 Ion Arrival Time Distribution at m/z = 531 Ion Arrival Time Distribution at m/z = 1061 Bradykinin Time Time Time (M+2H) 2+ (M+3H) 3+ (M+H) m/z Mass Spectrum
28 Injection Voltage 105 V 75 V Conclude: A = (3BK+3H) 3+ B = (2BK+2H) 2+ C = (BK+H) + 60 V 45 V B A C Arrival time
29 Increased Cell Temperature (2M+2H) 2+ 2(M+H) + dissociation Fitting the ATDs yield rate constants Binding energies (E a ) are obtained from an Arrhenius type of analysis
30 [2BK+2H] 2+ 2[BK+H] K [BK+M] K 2[BK+2M] 2+ Arrival time 310 K
31 Fit ---- Data time (µs)
32 2+ + ln(k) Bradykinin E a = 35.1±1 Kcal/mol A = 5.1 *10 19 s -1 S = 28.8 cal mol -1 K /T (K -1 )
33 LHRH [M+H] 2+ [M+H] + Arrival Time m/z
34 2+ + ln(k) K Bradykinin E a = 35.1 ± 1 Kcal/mol S = 28.8 cal mol -1 K K LHRH E a = 51.7 ± 2 Kcal/mol S = 69.0 cal mol -1 K K 435 K /T (K -1 )
35 Simulated annealing Dynamics simulations (300 K)
36 (2M+2H) 2+ (TS) 2(M+H) + Bradykinin (TS) + LHRH (TS) +
37 9.8 Å Monomers only slightly rearranged in dimer Electrostatic binding between charged groups dominates 9.9 Å 7.2 Å 9.1 Å 12.7 Å
38 3.7 Å N C 6.0 Å N C 13.4 Å Intermolecular interactions in dimer force open the backbone of the monomer Causes repulsion of the 2 N-termini and attraction between opposing N and C termini N 9.6 Å C
39 Energy [2M+2H] 2+ E a = 35.1 kcal/mol (exp) E a = 51.7 kcal/mol (exp) 2[BK+H] kcal/mol (AMBR) 2[LHRH+H] kcal/mol (AMBER) Rxn Coordinate
40 Do our calculated structures agree with experiment? Compare Cross Sections (Å 2 ) Experiment Theory* σ (%) [LHRH+H] [2LHRH+2H] [BK+H] [2BK+2H] * minimized structures (lowest 10 kcal/mol)
41 Peptides tend to form aggregates of the form (zm+zh) z+. Energy barriers for dimer dissociation are of the order of kcal/mol both for zwitterion (BK) and charge solvation structures (LHRH). Entropies (and energies) of activation indicate a major structural change occurs in TS for LHRH (i.e. a loose TS) while the TS for BK is much more dimer like (i.e. tighter).
42
43 Mass spe ctromet ric anal ysis of DNA crucial for obt a ining rapi d informat ion o n sma ll s amp les Problem: DNA fragment s du ring the sa mpling p roces s Solution: Unde rs tand th e fra gmentat ion me chan ism Propo sed mecha nis m: 1. proton ation of bas e 2. wea ken / b rea k ba se suga r bond 3. eliminate b ase a nd induce bac kbone fragmen tatio n
44 Proto n affinitie s of t he 4 DNA base s: G > A C >> T Hillenkam p, G ross: Look at f ragm e ntat ion of o ligonuc leot ides with T and G ba s es shoul d see pre fer e ntia l loss of G dgttt, dtgtt, dttgt, dtttg po s itive ion s : s e e lo s s of G in a ll case s but for negat ive ions
45 dtttg (dgttt )
46 ossible explanation: dtgtt and - dttgt form - salt bridges H CH 2 HN N CH 3 P - CH 2 + H N N N NH NH 2 P - HN N CH 3 CH 2 P H CH 2 HN N CH 3 H J. Gross, F. Hillenkamp, K.X. Wan, M.L. Gross J. Am. Soc. Mass Spectrom. 2001, 12, 180
47 Look at a simpler model with same possibility of salt bridge formation Is dtgt - a salt bridge? H H N CH 3 CH 2 N P - CH 2 H + N N N NH NH 2 P - HN CH 3 N CH 2 H
48 ATDs for dtgt K 80 K σ = 17 Å 2 arrival time
49 Scatter Plot for dtgt (salt bridge) - Cross-Section ( expt Å ) Relative Energy (kcal/mol)
50 Scatter Plot for dtgt (singly deprotonated) Cross-Section (Å open 2 ) expt folded Relative Energy (kcal/mol) 2 families of low-energy conformers "folded" smaller cross-section "open" larger cross-section σ ~ 18 Å 2 σ expt ~ 17 Å 2
51 Similar ATDs for dgtt -and dttg - (which should not be salt bridges) dgtt - dtgt - dttg - σ = 194 Å 2 σ = 196 Å 2 σ = 195 Å K folded open folded open folded 80 K open arrival time arrival time arrival time
52 Con cl us ion s : dtgt - not a sa lt bridge dtgtt - an d dttgt - a lso not sa lt bridges Gros s / Hillen kamp me chan ism may not be correct (or sa lt bridge is type of tra ns ition stat e ) Prelimina ry calcula tions indicate salt b ridge s tructure is highe r in e ne rgy Hydrat ion Studie s b e ing initiated
53 Small peptides and oligonucleotides are generally not salt bridges. However, salt bridges can be energetically close to charge solvation structures. Salt bridges are stabilized (compared to CS) by additional solvation. Salt bridges are important intermediates (H/D exchange, fragmentation). Little structural change occurs within a salt bridge unit when units aggregate.
The Structure of Small Protonated Peptides Containing Arginine and the Effect of Hydration
 The Structure of Small Protonated Peptides Containing Arginine and the Effect of Hydration Thomas Wyttenbach, Denfeng Liu, Perdita Barran, Michael T. Bowers University of California, Santa Barbara Vicki
The Structure of Small Protonated Peptides Containing Arginine and the Effect of Hydration Thomas Wyttenbach, Denfeng Liu, Perdita Barran, Michael T. Bowers University of California, Santa Barbara Vicki
Gas-Phase DNA Helix Conformations
 Gas-Phase DNA Helix Conformations Erin Shammel Baker, Jennifer Gidden, Alessandra Ferzoco, Thomas Wyttenbach and Michael Bowers utline Experimental Method Theoretical Method Instrumentation DNA Background
Gas-Phase DNA Helix Conformations Erin Shammel Baker, Jennifer Gidden, Alessandra Ferzoco, Thomas Wyttenbach and Michael Bowers utline Experimental Method Theoretical Method Instrumentation DNA Background
Hydration of Nucleotides Thomas Wyttenbach, Dengfeng Liu, and Michael T. Bowers
 http://bowers.chem.ucsb.edu/ ydration of ucleotides Thomas Wyttenbach, Dengfeng Liu, and Michael T. Bowers ASMS 2006 Why study hydration? Is a certain property of a molecule (e.g. conformation) inherent
http://bowers.chem.ucsb.edu/ ydration of ucleotides Thomas Wyttenbach, Dengfeng Liu, and Michael T. Bowers ASMS 2006 Why study hydration? Is a certain property of a molecule (e.g. conformation) inherent
Zwitterionic i-motif structures are preserved in DNA negatively charged ions. produced by electrospray mass spectrometry
 S1 Zwitterionic i-motif structures are preserved in DNA negatively charged ions produced by electrospray mass spectrometry Frederic Rosu 1*, Valérie Gabelica 1, Laure Joly 1, Gilles Grégoire 2, Edwin De
S1 Zwitterionic i-motif structures are preserved in DNA negatively charged ions produced by electrospray mass spectrometry Frederic Rosu 1*, Valérie Gabelica 1, Laure Joly 1, Gilles Grégoire 2, Edwin De
Structure and Modeling of Polyhedral Oligomeric Silsesquioxane (POSS) Systems
 Structure and Modeling of Polyhedral ligomeric lsesquioxane (PSS) Systems by Stanley Anderson (Westmont College) Michael Bowers (UCSB) Erin Shammel Baker, Jennifer Gidden (UCSB) Shawn Phillips, Tim Haddad,
Structure and Modeling of Polyhedral ligomeric lsesquioxane (PSS) Systems by Stanley Anderson (Westmont College) Michael Bowers (UCSB) Erin Shammel Baker, Jennifer Gidden (UCSB) Shawn Phillips, Tim Haddad,
Chapter 1. Introduction
 1-1 Chapter 1. Introduction 1.1. Background Non covalent interactions are important for the structures and reactivity of biological molecules in the gas phase as well as in the solution phase. 1 It is
1-1 Chapter 1. Introduction 1.1. Background Non covalent interactions are important for the structures and reactivity of biological molecules in the gas phase as well as in the solution phase. 1 It is
Peptides And Proteins
 Kevin Burgess, May 3, 2017 1 Peptides And Proteins from chapter(s) in the recommended text A. Introduction B. omenclature And Conventions by amide bonds. on the left, right. 2 -terminal C-terminal triglycine
Kevin Burgess, May 3, 2017 1 Peptides And Proteins from chapter(s) in the recommended text A. Introduction B. omenclature And Conventions by amide bonds. on the left, right. 2 -terminal C-terminal triglycine
Molecular Structure Prediction by Global Optimization
 Molecular Structure Prediction by Global Optimization K.A. DILL Department of Pharmaceutical Chemistry, University of California at San Francisco, San Francisco, CA 94118 A.T. PHILLIPS Computer Science
Molecular Structure Prediction by Global Optimization K.A. DILL Department of Pharmaceutical Chemistry, University of California at San Francisco, San Francisco, CA 94118 A.T. PHILLIPS Computer Science
Biological Macromolecules
 Introduction for Chem 493 Chemistry of Biological Macromolecules Dr. L. Luyt January 2008 Dr. L. Luyt Chem 493-2008 1 Biological macromolecules are the molecules of life allow for organization serve a
Introduction for Chem 493 Chemistry of Biological Macromolecules Dr. L. Luyt January 2008 Dr. L. Luyt Chem 493-2008 1 Biological macromolecules are the molecules of life allow for organization serve a
Other Methods for Generating Ions 1. MALDI matrix assisted laser desorption ionization MS 2. Spray ionization techniques 3. Fast atom bombardment 4.
 Other Methods for Generating Ions 1. MALDI matrix assisted laser desorption ionization MS 2. Spray ionization techniques 3. Fast atom bombardment 4. Field Desorption 5. MS MS techniques Matrix assisted
Other Methods for Generating Ions 1. MALDI matrix assisted laser desorption ionization MS 2. Spray ionization techniques 3. Fast atom bombardment 4. Field Desorption 5. MS MS techniques Matrix assisted
Assessing Intrinsic Side Chain Interactions between i and i + 4 Residues in Solvent-Free Peptides: A Combinatorial Gas-Phase Approach
 10566 J. Phys. Chem. A 2003, 107, 10566-10579 Assessing Intrinsic Side Chain Interactions between i and i + 4 Residues in Solvent-Free Peptides: A Combinatorial Gas-Phase Approach Catherine A. Srebalus
10566 J. Phys. Chem. A 2003, 107, 10566-10579 Assessing Intrinsic Side Chain Interactions between i and i + 4 Residues in Solvent-Free Peptides: A Combinatorial Gas-Phase Approach Catherine A. Srebalus
What binds to Hb in addition to O 2?
 Reading: Ch5; 158-169, 162-166, 169-174 Problems: Ch5 (text); 3,7,8,10 Ch5 (study guide-facts); 1,2,3,4,5,8 Ch5 (study guide-apply); 2,3 Remember Today at 5:30 in CAS-522 is the second chance for the MB
Reading: Ch5; 158-169, 162-166, 169-174 Problems: Ch5 (text); 3,7,8,10 Ch5 (study guide-facts); 1,2,3,4,5,8 Ch5 (study guide-apply); 2,3 Remember Today at 5:30 in CAS-522 is the second chance for the MB
Zwitterions in Neutral Arginine Clusters
 Chapter 3 43 Cooperative Salt Bridge Stabilization of Gas Phase Zwitterions in Neutral Arginine Clusters Portions published previously in: Julian R. R.; Beauchamp J. L.; Goddard W. A. J. Phys. Chem. A
Chapter 3 43 Cooperative Salt Bridge Stabilization of Gas Phase Zwitterions in Neutral Arginine Clusters Portions published previously in: Julian R. R.; Beauchamp J. L.; Goddard W. A. J. Phys. Chem. A
Problem Set 1
 2006 7.012 Problem Set 1 Due before 5 PM on FRIDAY, September 15, 2006. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. For each of the following parts, pick
2006 7.012 Problem Set 1 Due before 5 PM on FRIDAY, September 15, 2006. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. For each of the following parts, pick
Solutions and Non-Covalent Binding Forces
 Chapter 3 Solutions and Non-Covalent Binding Forces 3.1 Solvent and solution properties Molecules stick together using the following forces: dipole-dipole, dipole-induced dipole, hydrogen bond, van der
Chapter 3 Solutions and Non-Covalent Binding Forces 3.1 Solvent and solution properties Molecules stick together using the following forces: dipole-dipole, dipole-induced dipole, hydrogen bond, van der
What makes a good graphene-binding peptide? Adsorption of amino acids and peptides at aqueous graphene interfaces: Electronic Supplementary
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry B. This journal is The Royal Society of Chemistry 21 What makes a good graphene-binding peptide? Adsorption of amino acids and
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry B. This journal is The Royal Society of Chemistry 21 What makes a good graphene-binding peptide? Adsorption of amino acids and
Figure 1. Molecules geometries of 5021 and Each neutral group in CHARMM topology was grouped in dash circle.
 Project I Chemistry 8021, Spring 2005/2/23 This document was turned in by a student as a homework paper. 1. Methods First, the cartesian coordinates of 5021 and 8021 molecules (Fig. 1) are generated, in
Project I Chemistry 8021, Spring 2005/2/23 This document was turned in by a student as a homework paper. 1. Methods First, the cartesian coordinates of 5021 and 8021 molecules (Fig. 1) are generated, in
Lecture 2 and 3: Review of forces (ctd.) and elementary statistical mechanics. Contributions to protein stability
 Lecture 2 and 3: Review of forces (ctd.) and elementary statistical mechanics. Contributions to protein stability Part I. Review of forces Covalent bonds Non-covalent Interactions: Van der Waals Interactions
Lecture 2 and 3: Review of forces (ctd.) and elementary statistical mechanics. Contributions to protein stability Part I. Review of forces Covalent bonds Non-covalent Interactions: Van der Waals Interactions
Supplementary Figure 3 a. Structural comparison between the two determined structures for the IL 23:MA12 complex. The overall RMSD between the two
 Supplementary Figure 1. Biopanningg and clone enrichment of Alphabody binders against human IL 23. Positive clones in i phage ELISA with optical density (OD) 3 times higher than background are shown for
Supplementary Figure 1. Biopanningg and clone enrichment of Alphabody binders against human IL 23. Positive clones in i phage ELISA with optical density (OD) 3 times higher than background are shown for
Ranjit P. Bahadur Assistant Professor Department of Biotechnology Indian Institute of Technology Kharagpur, India. 1 st November, 2013
 Hydration of protein-rna recognition sites Ranjit P. Bahadur Assistant Professor Department of Biotechnology Indian Institute of Technology Kharagpur, India 1 st November, 2013 Central Dogma of life DNA
Hydration of protein-rna recognition sites Ranjit P. Bahadur Assistant Professor Department of Biotechnology Indian Institute of Technology Kharagpur, India 1 st November, 2013 Central Dogma of life DNA
NMR parameters intensity chemical shift coupling constants 1D 1 H spectra of nucleic acids and proteins
 Lecture #2 M230 NMR parameters intensity chemical shift coupling constants Juli Feigon 1D 1 H spectra of nucleic acids and proteins NMR Parameters A. Intensity (area) 1D NMR spectrum: integrated intensity
Lecture #2 M230 NMR parameters intensity chemical shift coupling constants Juli Feigon 1D 1 H spectra of nucleic acids and proteins NMR Parameters A. Intensity (area) 1D NMR spectrum: integrated intensity
Supplementary information Silver (I) as DNA glue: Ag + - mediated guanine pairing revealed by removing Watson- Crick constraints
 Supplementary information Silver (I) as DNA glue: Ag + - mediated guanine pairing revealed by removing Watson- Crick constraints Steven M. Swasey [b], Leonardo Espinosa Leal [c], Olga Lopez- Acevedo [c],
Supplementary information Silver (I) as DNA glue: Ag + - mediated guanine pairing revealed by removing Watson- Crick constraints Steven M. Swasey [b], Leonardo Espinosa Leal [c], Olga Lopez- Acevedo [c],
Supplementary figure 1. Comparison of unbound ogm-csf and ogm-csf as captured in the GIF:GM-CSF complex. Alignment of two copies of unbound ovine
 Supplementary figure 1. Comparison of unbound and as captured in the GIF:GM-CSF complex. Alignment of two copies of unbound ovine GM-CSF (slate) with bound GM-CSF in the GIF:GM-CSF complex (GIF: green,
Supplementary figure 1. Comparison of unbound and as captured in the GIF:GM-CSF complex. Alignment of two copies of unbound ovine GM-CSF (slate) with bound GM-CSF in the GIF:GM-CSF complex (GIF: green,
CHMI 2227 EL. Biochemistry I. Test January Prof : Eric R. Gauthier, Ph.D.
 CHMI 2227 EL Biochemistry I Test 1 26 January 2007 Prof : Eric R. Gauthier, Ph.D. Guidelines: 1) Duration: 55 min 2) 14 questions, on 7 pages. For 70 marks (5 marks per question). Worth 15 % of the final
CHMI 2227 EL Biochemistry I Test 1 26 January 2007 Prof : Eric R. Gauthier, Ph.D. Guidelines: 1) Duration: 55 min 2) 14 questions, on 7 pages. For 70 marks (5 marks per question). Worth 15 % of the final
Sequential resonance assignments in (small) proteins: homonuclear method 2º structure determination
 Lecture 9 M230 Feigon Sequential resonance assignments in (small) proteins: homonuclear method 2º structure determination Reading resources v Roberts NMR of Macromolecules, Chap 4 by Christina Redfield
Lecture 9 M230 Feigon Sequential resonance assignments in (small) proteins: homonuclear method 2º structure determination Reading resources v Roberts NMR of Macromolecules, Chap 4 by Christina Redfield
schematic diagram; EGF binding, dimerization, phosphorylation, Grb2 binding, etc.
 Lecture 1: Noncovalent Biomolecular Interactions Bioengineering and Modeling of biological processes -e.g. tissue engineering, cancer, autoimmune disease Example: RTK signaling, e.g. EGFR Growth responses
Lecture 1: Noncovalent Biomolecular Interactions Bioengineering and Modeling of biological processes -e.g. tissue engineering, cancer, autoimmune disease Example: RTK signaling, e.g. EGFR Growth responses
Observations of noncovalently bound complexes
 ARTICLES High-Order Structure and Dissociation of Gaseous Peptide Aggregates that are Hidden in Mass Spectra Anne E. Counterman, Stephen J. Valentine, Catherine A. Srebalus, Sheila C. Henderson, Cherokee
ARTICLES High-Order Structure and Dissociation of Gaseous Peptide Aggregates that are Hidden in Mass Spectra Anne E. Counterman, Stephen J. Valentine, Catherine A. Srebalus, Sheila C. Henderson, Cherokee
Conformations of Reaction Products: Polymers and Organic Phenyl/Vinyl Linkages
 Conformations of Reaction Products: Polymers and Organic Phenyl/Vinyl Linkages Erin Shammel Baker, Jennifer Gidden, Glenn Bartholomew, Guillermo Bazan, and Michael T. Bowers (UCSB) James Scrivens and Anthony
Conformations of Reaction Products: Polymers and Organic Phenyl/Vinyl Linkages Erin Shammel Baker, Jennifer Gidden, Glenn Bartholomew, Guillermo Bazan, and Michael T. Bowers (UCSB) James Scrivens and Anthony
Chapter 1. Introduction
 1-1 Chapter 1. Introduction 1.1. Background Non-covalent bonds play a critical role in determining the structure and behavior of biological molecules. 1 The tertiary structure of proteins is largely determined
1-1 Chapter 1. Introduction 1.1. Background Non-covalent bonds play a critical role in determining the structure and behavior of biological molecules. 1 The tertiary structure of proteins is largely determined
Protein Structure Bioinformatics Introduction
 1 Swiss Institute of Bioinformatics Protein Structure Bioinformatics Introduction Basel, 27. September 2004 Torsten Schwede Biozentrum - Universität Basel Swiss Institute of Bioinformatics Klingelbergstr
1 Swiss Institute of Bioinformatics Protein Structure Bioinformatics Introduction Basel, 27. September 2004 Torsten Schwede Biozentrum - Universität Basel Swiss Institute of Bioinformatics Klingelbergstr
Supplementary information
 Supplementary information doi: 10.1038/nchem.247 Amyloid!-Protein Oligomerization and the Importance of Tetramers and Dodecamers in the Aetiology of Alzheimer s Disease Summer L. Bernstein, Nicholas F.
Supplementary information doi: 10.1038/nchem.247 Amyloid!-Protein Oligomerization and the Importance of Tetramers and Dodecamers in the Aetiology of Alzheimer s Disease Summer L. Bernstein, Nicholas F.
Physiochemical Properties of Residues
 Physiochemical Properties of Residues Various Sources C N Cα R Slide 1 Conformational Propensities Conformational Propensity is the frequency in which a residue adopts a given conformation (in a polypeptide)
Physiochemical Properties of Residues Various Sources C N Cα R Slide 1 Conformational Propensities Conformational Propensity is the frequency in which a residue adopts a given conformation (in a polypeptide)
NMR study of complexes between low molecular mass inhibitors and the West Nile virus NS2B-NS3 protease
 University of Wollongong Research Online Faculty of Science - Papers (Archive) Faculty of Science, Medicine and Health 2009 NMR study of complexes between low molecular mass inhibitors and the West Nile
University of Wollongong Research Online Faculty of Science - Papers (Archive) Faculty of Science, Medicine and Health 2009 NMR study of complexes between low molecular mass inhibitors and the West Nile
Improving Protein Function Prediction with Molecular Dynamics Simulations. Dariya Glazer Russ Altman
 Improving Protein Function Prediction with Molecular Dynamics Simulations Dariya Glazer Russ Altman Motivation Sometimes the 3D structure doesn t score well for a known function. The experimental structure
Improving Protein Function Prediction with Molecular Dynamics Simulations Dariya Glazer Russ Altman Motivation Sometimes the 3D structure doesn t score well for a known function. The experimental structure
Proton Acidity. (b) For the following reaction, draw the arrowhead properly to indicate the position of the equilibrium: HA + K + B -
 Proton Acidity A01 Given that acid A has a pk a of 15 and acid B has a pk a of 10, then: (a) Which of the two acids is stronger? (b) For the following reaction, draw the arrowhead properly to indicate
Proton Acidity A01 Given that acid A has a pk a of 15 and acid B has a pk a of 10, then: (a) Which of the two acids is stronger? (b) For the following reaction, draw the arrowhead properly to indicate
Protein Folding experiments and theory
 Protein Folding experiments and theory 1, 2,and 3 Protein Structure Fig. 3-16 from Lehninger Biochemistry, 4 th ed. The 3D structure is not encoded at the single aa level Hydrogen Bonding Shared H atom
Protein Folding experiments and theory 1, 2,and 3 Protein Structure Fig. 3-16 from Lehninger Biochemistry, 4 th ed. The 3D structure is not encoded at the single aa level Hydrogen Bonding Shared H atom
Conformational Analysis
 Conformational Analysis C01 3 C C 3 is the most stable by 0.9 kcal/mole C02 K eq = K 1-1 * K 2 = 0.45-1 * 0.048 = 0.11 C04 The intermediate in the reaction of 2 has an unfavorable syn-pentane interaction,
Conformational Analysis C01 3 C C 3 is the most stable by 0.9 kcal/mole C02 K eq = K 1-1 * K 2 = 0.45-1 * 0.048 = 0.11 C04 The intermediate in the reaction of 2 has an unfavorable syn-pentane interaction,
NAME IV. /22. I. MULTIPLE CHOICE. (48 points; 2 pts each) Choose the BEST answer to the question by circling the appropriate letter.
 NAME Exam I I. /48 September 25, 2017 Biochemistry I II. / 4 BI/CH 421/621 III. /26 IV. /22 TOTAL /100 I. MULTIPLE CHOICE. (48 points; 2 pts each) Choose the BEST answer to the question by circling the
NAME Exam I I. /48 September 25, 2017 Biochemistry I II. / 4 BI/CH 421/621 III. /26 IV. /22 TOTAL /100 I. MULTIPLE CHOICE. (48 points; 2 pts each) Choose the BEST answer to the question by circling the
Catalytic Mechanism of the Glycyl Radical Enzyme 4-Hydroxyphenylacetate Decarboxylase from Continuum Electrostatic and QC/MM Calculations
 Catalytic Mechanism of the Glycyl Radical Enzyme 4-Hydroxyphenylacetate Decarboxylase from Continuum Electrostatic and QC/MM Calculations Supplementary Materials Mikolaj Feliks, 1 Berta M. Martins, 2 G.
Catalytic Mechanism of the Glycyl Radical Enzyme 4-Hydroxyphenylacetate Decarboxylase from Continuum Electrostatic and QC/MM Calculations Supplementary Materials Mikolaj Feliks, 1 Berta M. Martins, 2 G.
SUPPLEMENTARY INFORMATION
 In the format provided by the authors and unedited. SUPPLEMENTARY INFORMATION DOI: 10.1038/NCHEM.2720 1 2 3 Tuning underwater adhesion with cation-π interactions Matthew A. Gebbie, Wei Wei, Alex M. Schrader,
In the format provided by the authors and unedited. SUPPLEMENTARY INFORMATION DOI: 10.1038/NCHEM.2720 1 2 3 Tuning underwater adhesion with cation-π interactions Matthew A. Gebbie, Wei Wei, Alex M. Schrader,
Sunhats for plants. How plants detect dangerous ultraviolet rays
 Sunhats for plants How plants detect dangerous ultraviolet rays Anyone who has ever suffered sunburn will know about the effects of too much ultraviolet (UV) radiation, in particular UV-B (from 280-315
Sunhats for plants How plants detect dangerous ultraviolet rays Anyone who has ever suffered sunburn will know about the effects of too much ultraviolet (UV) radiation, in particular UV-B (from 280-315
Lecture 21 (11/3/17) Protein Stability, Folding, and Dynamics Hydrophobic effect drives protein folding
 Reading: Ch4; 142-151 Problems: Ch4 (text); 14, 16 Ch6 (text); 1, 4 NEXT (after exam) Reading: Ch8; 310-312, 279-285, 285-289 Ch24; 957-961 Problems: Ch8 (text); 1,2,22 Ch8 (study-guide:facts); 1,2,3,4,5,9,10
Reading: Ch4; 142-151 Problems: Ch4 (text); 14, 16 Ch6 (text); 1, 4 NEXT (after exam) Reading: Ch8; 310-312, 279-285, 285-289 Ch24; 957-961 Problems: Ch8 (text); 1,2,22 Ch8 (study-guide:facts); 1,2,3,4,5,9,10
Supplementary Information
 Supplementary Information Resveratrol Serves as a Protein-Substrate Interaction Stabilizer in Human SIRT1 Activation Xuben Hou,, David Rooklin, Hao Fang *,,, Yingkai Zhang Department of Medicinal Chemistry
Supplementary Information Resveratrol Serves as a Protein-Substrate Interaction Stabilizer in Human SIRT1 Activation Xuben Hou,, David Rooklin, Hao Fang *,,, Yingkai Zhang Department of Medicinal Chemistry
Lecture 15: Realities of Genome Assembly Protein Sequencing
 Lecture 15: Realities of Genome Assembly Protein Sequencing Study Chapter 8.10-8.15 1 Euler s Theorems A graph is balanced if for every vertex the number of incoming edges equals to the number of outgoing
Lecture 15: Realities of Genome Assembly Protein Sequencing Study Chapter 8.10-8.15 1 Euler s Theorems A graph is balanced if for every vertex the number of incoming edges equals to the number of outgoing
Identification of Human Hemoglobin Protein Variants Using Electrospray Ionization-Electron Transfer Dissociation Mass Spectrometry
 Identification of Human Hemoglobin Protein Variants Using Electrospray Ionization-Electron Transfer Dissociation Mass Spectrometry Jonathan Williams Waters Corporation, Milford, MA, USA A P P L I C AT
Identification of Human Hemoglobin Protein Variants Using Electrospray Ionization-Electron Transfer Dissociation Mass Spectrometry Jonathan Williams Waters Corporation, Milford, MA, USA A P P L I C AT
AN AB INITIO STUDY OF INTERMOLECULAR INTERACTIONS OF GLYCINE, ALANINE AND VALINE DIPEPTIDE-FORMALDEHYDE DIMERS
 Journal of Undergraduate Chemistry Research, 2004, 1, 15 AN AB INITIO STUDY OF INTERMOLECULAR INTERACTIONS OF GLYCINE, ALANINE AND VALINE DIPEPTIDE-FORMALDEHYDE DIMERS J.R. Foley* and R.D. Parra Chemistry
Journal of Undergraduate Chemistry Research, 2004, 1, 15 AN AB INITIO STUDY OF INTERMOLECULAR INTERACTIONS OF GLYCINE, ALANINE AND VALINE DIPEPTIDE-FORMALDEHYDE DIMERS J.R. Foley* and R.D. Parra Chemistry
Supplementary Information
 1 Supplementary Information Figure S1 The V=0.5 Harker section of an anomalous difference Patterson map calculated using diffraction data from the NNQQNY crystal at 1.3 Å resolution. The position of the
1 Supplementary Information Figure S1 The V=0.5 Harker section of an anomalous difference Patterson map calculated using diffraction data from the NNQQNY crystal at 1.3 Å resolution. The position of the
FOCUS: ION THERMOCHEMISTRY
 Gas-Phase Conformations of Cationized Poly(styrene) Oligomers FOCUS: ION THERMOCHEMISTRY Jennifer Gidden and Michael T. Bowers Department of Chemistry and Biochemistry, University of California, Santa
Gas-Phase Conformations of Cationized Poly(styrene) Oligomers FOCUS: ION THERMOCHEMISTRY Jennifer Gidden and Michael T. Bowers Department of Chemistry and Biochemistry, University of California, Santa
Biochemistry Quiz Review 1I. 1. Of the 20 standard amino acids, only is not optically active. The reason is that its side chain.
 Biochemistry Quiz Review 1I A general note: Short answer questions are just that, short. Writing a paragraph filled with every term you can remember from class won t improve your answer just answer clearly,
Biochemistry Quiz Review 1I A general note: Short answer questions are just that, short. Writing a paragraph filled with every term you can remember from class won t improve your answer just answer clearly,
Using Higher Calculus to Study Biologically Important Molecules Julie C. Mitchell
 Using Higher Calculus to Study Biologically Important Molecules Julie C. Mitchell Mathematics and Biochemistry University of Wisconsin - Madison 0 There Are Many Kinds Of Proteins The word protein comes
Using Higher Calculus to Study Biologically Important Molecules Julie C. Mitchell Mathematics and Biochemistry University of Wisconsin - Madison 0 There Are Many Kinds Of Proteins The word protein comes
ONETEP PB/SA: Application to G-Quadruplex DNA Stability. Danny Cole
 ONETEP PB/SA: Application to G-Quadruplex DNA Stability Danny Cole Introduction Historical overview of structure and free energy calculation of complex molecules using molecular mechanics and continuum
ONETEP PB/SA: Application to G-Quadruplex DNA Stability Danny Cole Introduction Historical overview of structure and free energy calculation of complex molecules using molecular mechanics and continuum
Oxygen Binding in Hemocyanin
 Supporting Information for Quantum Mechanics/Molecular Mechanics Study of Oxygen Binding in Hemocyanin Toru Saito and Walter Thiel* Max-Planck-Institut für Kohlenforschung, Kaiser-Wilhelm-Platz 1, D-45470
Supporting Information for Quantum Mechanics/Molecular Mechanics Study of Oxygen Binding in Hemocyanin Toru Saito and Walter Thiel* Max-Planck-Institut für Kohlenforschung, Kaiser-Wilhelm-Platz 1, D-45470
CHAPTER 29 HW: AMINO ACIDS + PROTEINS
 CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
NIH Public Access Author Manuscript Int J Mass Spectrom. Author manuscript; available in PMC 2015 September 15.
 NIH Public Access Author Manuscript Published in final edited form as: Int J Mass Spectrom. 2014 September 15; 370: 75 95. doi:10.1016/j.ijms.2014.06.016. Ion mobility spectrometry: A personal view of
NIH Public Access Author Manuscript Published in final edited form as: Int J Mass Spectrom. 2014 September 15; 370: 75 95. doi:10.1016/j.ijms.2014.06.016. Ion mobility spectrometry: A personal view of
Packing of Secondary Structures
 7.88 Lecture Notes - 4 7.24/7.88J/5.48J The Protein Folding and Human Disease Professor Gossard Retrieving, Viewing Protein Structures from the Protein Data Base Helix helix packing Packing of Secondary
7.88 Lecture Notes - 4 7.24/7.88J/5.48J The Protein Folding and Human Disease Professor Gossard Retrieving, Viewing Protein Structures from the Protein Data Base Helix helix packing Packing of Secondary
12/6/12. Dr. Sanjeeva Srivastava IIT Bombay. Primary Structure. Secondary Structure. Tertiary Structure. Quaternary Structure.
 Dr. anjeeva rivastava Primary tructure econdary tructure Tertiary tructure Quaternary tructure Amino acid residues α Helix Polypeptide chain Assembled subunits 2 1 Amino acid sequence determines 3-D structure
Dr. anjeeva rivastava Primary tructure econdary tructure Tertiary tructure Quaternary tructure Amino acid residues α Helix Polypeptide chain Assembled subunits 2 1 Amino acid sequence determines 3-D structure
Exam I Answer Key: Summer 2006, Semester C
 1. Which of the following tripeptides would migrate most rapidly towards the negative electrode if electrophoresis is carried out at ph 3.0? a. gly-gly-gly b. glu-glu-asp c. lys-glu-lys d. val-asn-lys
1. Which of the following tripeptides would migrate most rapidly towards the negative electrode if electrophoresis is carried out at ph 3.0? a. gly-gly-gly b. glu-glu-asp c. lys-glu-lys d. val-asn-lys
Supplementary Figure S1. Urea-mediated buffering mechanism of H. pylori. Gastric urea is funneled to a cytoplasmic urease that is presumably attached
 Supplementary Figure S1. Urea-mediated buffering mechanism of H. pylori. Gastric urea is funneled to a cytoplasmic urease that is presumably attached to HpUreI. Urea hydrolysis products 2NH 3 and 1CO 2
Supplementary Figure S1. Urea-mediated buffering mechanism of H. pylori. Gastric urea is funneled to a cytoplasmic urease that is presumably attached to HpUreI. Urea hydrolysis products 2NH 3 and 1CO 2
Gaseous Bradykinin and Its Singly, Doubly, and Triply Protonated Forms: A First-Principles Study
 7528 J. Phys. Chem. B 2006, 110, 7528-7537 Gaseous Bradykinin and Its Singly, Doubly, and Triply Protonated Forms: A First-Principles Study Christopher F. Rodriquez, Galina Orlova,, Yuzhu Guo, Xiaomao
7528 J. Phys. Chem. B 2006, 110, 7528-7537 Gaseous Bradykinin and Its Singly, Doubly, and Triply Protonated Forms: A First-Principles Study Christopher F. Rodriquez, Galina Orlova,, Yuzhu Guo, Xiaomao
Multi-scale approaches in description and design of enzymes
 Multi-scale approaches in description and design of enzymes Anastassia Alexandrova and Manuel Sparta UCLA & CNSI Catalysis: it is all about the barrier The inside-out protocol: Big Aim: development of
Multi-scale approaches in description and design of enzymes Anastassia Alexandrova and Manuel Sparta UCLA & CNSI Catalysis: it is all about the barrier The inside-out protocol: Big Aim: development of
EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I Dr. Glen B Legge
 EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I 2009 Dr. Glen B Legge This is a Scantron exam. All answers should be transferred to the Scantron sheet using a #2 pencil. Write and bubble
EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I 2009 Dr. Glen B Legge This is a Scantron exam. All answers should be transferred to the Scantron sheet using a #2 pencil. Write and bubble
BENG 183 Trey Ideker. Protein Sequencing
 BENG 183 Trey Ideker Protein Sequencing The following slides borrowed from Hong Li s Biochemistry Course: www.sb.fsu.edu/~hongli/4053notes Introduction to Proteins Proteins are of vital importance to biological
BENG 183 Trey Ideker Protein Sequencing The following slides borrowed from Hong Li s Biochemistry Course: www.sb.fsu.edu/~hongli/4053notes Introduction to Proteins Proteins are of vital importance to biological
Chemistry Chapter 22
 hemistry 2100 hapter 22 Proteins Proteins serve many functions, including the following. 1. Structure: ollagen and keratin are the chief constituents of skin, bone, hair, and nails. 2. atalysts: Virtually
hemistry 2100 hapter 22 Proteins Proteins serve many functions, including the following. 1. Structure: ollagen and keratin are the chief constituents of skin, bone, hair, and nails. 2. atalysts: Virtually
NAME. EXAM I I. / 36 September 25, 2000 Biochemistry I II. / 26 BICH421/621 III. / 38 TOTAL /100
 EXAM I I. / 6 September 25, 2000 Biochemistry I II. / 26 BIH421/621 III. / 8 TOTAL /100 I. MULTIPLE HOIE (6 points) hoose the BEST answer to the question by circling the appropriate letter. 1. An amino
EXAM I I. / 6 September 25, 2000 Biochemistry I II. / 26 BIH421/621 III. / 8 TOTAL /100 I. MULTIPLE HOIE (6 points) hoose the BEST answer to the question by circling the appropriate letter. 1. An amino
LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor
 LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor Note: Adequate space is given for each answer. Questions that require a brief explanation should
LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor Note: Adequate space is given for each answer. Questions that require a brief explanation should
Introduction to Comparative Protein Modeling. Chapter 4 Part I
 Introduction to Comparative Protein Modeling Chapter 4 Part I 1 Information on Proteins Each modeling study depends on the quality of the known experimental data. Basis of the model Search in the literature
Introduction to Comparative Protein Modeling Chapter 4 Part I 1 Information on Proteins Each modeling study depends on the quality of the known experimental data. Basis of the model Search in the literature
A. Two of the common amino acids are analyzed. Amino acid X and amino acid Y both have an isoionic point in the range of
 Questions with Answers- Amino Acids & Peptides A. Two of the common amino acids are analyzed. Amino acid X and amino acid Y both have an isoionic point in the range of 5.0-6.5 (Questions 1-4) 1. Which
Questions with Answers- Amino Acids & Peptides A. Two of the common amino acids are analyzed. Amino acid X and amino acid Y both have an isoionic point in the range of 5.0-6.5 (Questions 1-4) 1. Which
7.012 Problem Set 1. i) What are two main differences between prokaryotic cells and eukaryotic cells?
 ame 7.01 Problem Set 1 Section Question 1 a) What are the four major types of biological molecules discussed in lecture? Give one important function of each type of biological molecule in the cell? b)
ame 7.01 Problem Set 1 Section Question 1 a) What are the four major types of biological molecules discussed in lecture? Give one important function of each type of biological molecule in the cell? b)
Computational Studies of the Photoreceptor Rhodopsin. Scott E. Feller Wabash College
 Computational Studies of the Photoreceptor Rhodopsin Scott E. Feller Wabash College Rhodopsin Photocycle Dark-adapted Rhodopsin hn Isomerize retinal Photorhodopsin ~200 fs Bathorhodopsin Meta-II ms timescale
Computational Studies of the Photoreceptor Rhodopsin Scott E. Feller Wabash College Rhodopsin Photocycle Dark-adapted Rhodopsin hn Isomerize retinal Photorhodopsin ~200 fs Bathorhodopsin Meta-II ms timescale
CHEM J-9 June 2014
 CEM1611 2014-J-9 June 2014 Alanine (ala) and lysine (lys) are two amino acids with the structures given below as Fischer projections. The pk a values of the conjugate acid forms of the different functional
CEM1611 2014-J-9 June 2014 Alanine (ala) and lysine (lys) are two amino acids with the structures given below as Fischer projections. The pk a values of the conjugate acid forms of the different functional
FOCUS: NOVEL APPROACHES TO PEPTIDE AND PROTEIN STRUCTURE
 FOCUS: NOVEL APPROACHES TO PEPTIDE AND PROTEIN STRUCTURE Evaluation of Ion Mobility Spectroscopy for Determining Charge-Solvated versus Salt-Bridge Structures of Protonated Trimers Richard L. Wong and
FOCUS: NOVEL APPROACHES TO PEPTIDE AND PROTEIN STRUCTURE Evaluation of Ion Mobility Spectroscopy for Determining Charge-Solvated versus Salt-Bridge Structures of Protonated Trimers Richard L. Wong and
Dominant Paths in Protein Folding
 Dominant Paths in Protein Folding Henri Orland SPhT, CEA-Saclay France work in collaboration with P. Faccioli, F. Pederiva, M. Sega University of Trento Henri Orland Annecy meeting 2006 Outline Basic notions
Dominant Paths in Protein Folding Henri Orland SPhT, CEA-Saclay France work in collaboration with P. Faccioli, F. Pederiva, M. Sega University of Trento Henri Orland Annecy meeting 2006 Outline Basic notions
1. What is an ångstrom unit, and why is it used to describe molecular structures?
 1. What is an ångstrom unit, and why is it used to describe molecular structures? The ångstrom unit is a unit of distance suitable for measuring atomic scale objects. 1 ångstrom (Å) = 1 10-10 m. The diameter
1. What is an ångstrom unit, and why is it used to describe molecular structures? The ångstrom unit is a unit of distance suitable for measuring atomic scale objects. 1 ångstrom (Å) = 1 10-10 m. The diameter
Structural and mechanistic insight into the substrate. binding from the conformational dynamics in apo. and substrate-bound DapE enzyme
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 215 Structural and mechanistic insight into the substrate binding from the conformational
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 215 Structural and mechanistic insight into the substrate binding from the conformational
Collision Cross Section: Ideal elastic hard sphere collision:
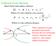 Collision Cross Section: Ideal elastic hard sphere collision: ( r r 1 ) Where is the collision cross-section r 1 r ) ( 1 Where is the collision distance r 1 r These equations negate potential interactions
Collision Cross Section: Ideal elastic hard sphere collision: ( r r 1 ) Where is the collision cross-section r 1 r ) ( 1 Where is the collision distance r 1 r These equations negate potential interactions
Central Dogma. modifications genome transcriptome proteome
 entral Dogma DA ma protein post-translational modifications genome transcriptome proteome 83 ierarchy of Protein Structure 20 Amino Acids There are 20 n possible sequences for a protein of n residues!
entral Dogma DA ma protein post-translational modifications genome transcriptome proteome 83 ierarchy of Protein Structure 20 Amino Acids There are 20 n possible sequences for a protein of n residues!
Supporting Information
 Supporting Information Boehr et al. 10.1073/pnas.0914163107 SI Text Materials and Methods. R 2 relaxation dispersion experiments. 15 NR 2 relaxation dispersion data measured at 1 H Larmor frequencies of
Supporting Information Boehr et al. 10.1073/pnas.0914163107 SI Text Materials and Methods. R 2 relaxation dispersion experiments. 15 NR 2 relaxation dispersion data measured at 1 H Larmor frequencies of
arxiv: v1 [physics.bio-ph] 26 Nov 2007
![arxiv: v1 [physics.bio-ph] 26 Nov 2007 arxiv: v1 [physics.bio-ph] 26 Nov 2007](/thumbs/90/104325462.jpg) On the Helix-coil Transition in Alanine-based Polypeptides in Gas Phase Yanjie Wei arxiv:0711.4087v1 [physics.bio-ph] 26 Nov 2007 Department of Physics, Michigan Technological University, Houghton, MI
On the Helix-coil Transition in Alanine-based Polypeptides in Gas Phase Yanjie Wei arxiv:0711.4087v1 [physics.bio-ph] 26 Nov 2007 Department of Physics, Michigan Technological University, Houghton, MI
On the Structure Elucidation Using Ion Mobility Spectrometry and Molecular Dynamics
 J. Phys. Chem. A 2009, 113, 8221 8234 8221 On the Structure Elucidation Using Ion Mobility Spectrometry and Molecular Dynamics F. A. Fernandez-Lima, H. Wei, Y. Q. Gao, and D. H. Russell* Department of
J. Phys. Chem. A 2009, 113, 8221 8234 8221 On the Structure Elucidation Using Ion Mobility Spectrometry and Molecular Dynamics F. A. Fernandez-Lima, H. Wei, Y. Q. Gao, and D. H. Russell* Department of
- Basic understandings: - Mapping interactions:
 NMR-lecture April 6th, 2009, FMP Berlin Outline: Christian Freund - Basic understandings: Relaxation Chemical exchange - Mapping interactions: -Chemical shift mapping (fast exchange) Linewidth analysis
NMR-lecture April 6th, 2009, FMP Berlin Outline: Christian Freund - Basic understandings: Relaxation Chemical exchange - Mapping interactions: -Chemical shift mapping (fast exchange) Linewidth analysis
Other types of liquid chromatography
 Other types of liquid chromatography Objectives: After this discussion you should be able to: Define IEC Basic mechanism Relationship between net charge and isoelectric point (pi) Relationship between
Other types of liquid chromatography Objectives: After this discussion you should be able to: Define IEC Basic mechanism Relationship between net charge and isoelectric point (pi) Relationship between
Programme Last week s quiz results + Summary Fold recognition Break Exercise: Modelling remote homologues
 Programme 8.00-8.20 Last week s quiz results + Summary 8.20-9.00 Fold recognition 9.00-9.15 Break 9.15-11.20 Exercise: Modelling remote homologues 11.20-11.40 Summary & discussion 11.40-12.00 Quiz 1 Feedback
Programme 8.00-8.20 Last week s quiz results + Summary 8.20-9.00 Fold recognition 9.00-9.15 Break 9.15-11.20 Exercise: Modelling remote homologues 11.20-11.40 Summary & discussion 11.40-12.00 Quiz 1 Feedback
Structure Investigation of Fam20C, a Golgi Casein Kinase
 Structure Investigation of Fam20C, a Golgi Casein Kinase Sharon Grubner National Taiwan University, Dr. Jung-Hsin Lin University of California San Diego, Dr. Rommie Amaro Abstract This research project
Structure Investigation of Fam20C, a Golgi Casein Kinase Sharon Grubner National Taiwan University, Dr. Jung-Hsin Lin University of California San Diego, Dr. Rommie Amaro Abstract This research project
SUPPLEMENTAL MATERIAL
 SUPPLEMENTAL MATERIAL Systematic Coarse-Grained Modeling of Complexation between Small Interfering RNA and Polycations Zonghui Wei 1 and Erik Luijten 1,2,3,4,a) 1 Graduate Program in Applied Physics, Northwestern
SUPPLEMENTAL MATERIAL Systematic Coarse-Grained Modeling of Complexation between Small Interfering RNA and Polycations Zonghui Wei 1 and Erik Luijten 1,2,3,4,a) 1 Graduate Program in Applied Physics, Northwestern
Protein Fragment Search Program ver Overview: Contents:
 Protein Fragment Search Program ver 1.1.1 Developed by: BioPhysics Laboratory, Faculty of Life and Environmental Science, Shimane University 1060 Nishikawatsu-cho, Matsue-shi, Shimane, 690-8504, Japan
Protein Fragment Search Program ver 1.1.1 Developed by: BioPhysics Laboratory, Faculty of Life and Environmental Science, Shimane University 1060 Nishikawatsu-cho, Matsue-shi, Shimane, 690-8504, Japan
Ligand-receptor interactions
 University of Silesia, Katowice, Poland 11 22 March 2013 Ligand-receptor interactions Dr. Pavel Polishchuk A.V. Bogatsky Physico-Chemical Institute of National Academy of Sciences of Ukraine Odessa, Ukraine
University of Silesia, Katowice, Poland 11 22 March 2013 Ligand-receptor interactions Dr. Pavel Polishchuk A.V. Bogatsky Physico-Chemical Institute of National Academy of Sciences of Ukraine Odessa, Ukraine
High-throughput, global proteomics assays are
 Evaluation of the Influence of Amino Acid Composition on the Propensity for Collision-Induced Dissociation of Model Peptides Using Molecular Dynamics Simulations William R. Cannon, a Danny Taasevigen,
Evaluation of the Influence of Amino Acid Composition on the Propensity for Collision-Induced Dissociation of Model Peptides Using Molecular Dynamics Simulations William R. Cannon, a Danny Taasevigen,
Lecture 10 (10/4/17) Lecture 10 (10/4/17)
 Lecture 10 (10/4/17) Reading: Ch4; 125, 138-141, 141-142 Problems: Ch4 (text); 7, 9, 11 Ch4 (study guide); 1, 2 NEXT Reading: Ch4; 125, 132-136 (structure determination) Ch4; 12-130 (Collagen) Problems:
Lecture 10 (10/4/17) Reading: Ch4; 125, 138-141, 141-142 Problems: Ch4 (text); 7, 9, 11 Ch4 (study guide); 1, 2 NEXT Reading: Ch4; 125, 132-136 (structure determination) Ch4; 12-130 (Collagen) Problems:
PROTEIN STRUCTURE AMINO ACIDS H R. Zwitterion (dipolar ion) CO 2 H. PEPTIDES Formal reactions showing formation of peptide bond by dehydration:
 PTEI STUTUE ydrolysis of proteins with aqueous acid or base yields a mixture of free amino acids. Each type of protein yields a characteristic mixture of the ~ 20 amino acids. AMI AIDS Zwitterion (dipolar
PTEI STUTUE ydrolysis of proteins with aqueous acid or base yields a mixture of free amino acids. Each type of protein yields a characteristic mixture of the ~ 20 amino acids. AMI AIDS Zwitterion (dipolar
Computational Protein Design
 11 Computational Protein Design This chapter introduces the automated protein design and experimental validation of a novel designed sequence, as described in Dahiyat and Mayo [1]. 11.1 Introduction Given
11 Computational Protein Design This chapter introduces the automated protein design and experimental validation of a novel designed sequence, as described in Dahiyat and Mayo [1]. 11.1 Introduction Given
Tridip Sheet, Raja Banerjee*
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Supplementary information The C NN motif: an intrinsic lover of sulfate and phosphate ions
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Supplementary information The C NN motif: an intrinsic lover of sulfate and phosphate ions
Structural Perspectives on Drug Resistance
 Structural Perspectives on Drug Resistance Irene Weber Departments of Biology and Chemistry Molecular Basis of Disease Program Georgia State University Atlanta, GA, USA What have we learned from 20 years
Structural Perspectives on Drug Resistance Irene Weber Departments of Biology and Chemistry Molecular Basis of Disease Program Georgia State University Atlanta, GA, USA What have we learned from 20 years
Prof. Jason Kahn Your Signature: Exams written in pencil or erasable ink will not be re-graded under any circumstances.
 1 Biochemistry 461 February 16, 1995 Exam #1 Prof. Jason Kahn Your Printed Name: Your SS#: Your Signature: You have 75 minutes for this exam. Exams written in pencil or erasable ink will not be re-graded
1 Biochemistry 461 February 16, 1995 Exam #1 Prof. Jason Kahn Your Printed Name: Your SS#: Your Signature: You have 75 minutes for this exam. Exams written in pencil or erasable ink will not be re-graded
Comparison between Bacteriorhodopsin and Halorhodopsin. Halorhodopsin (HR) and Bacteriorhodopsin (BR) belong to a subfamily of
 Comparison between Bacteriorhodopsin and Halorhodopsin Halorhodopsin (HR) and Bacteriorhodopsin (BR) belong to a subfamily of heptahelical membrane proteins, the archaeal rhodopsins. They are found in
Comparison between Bacteriorhodopsin and Halorhodopsin Halorhodopsin (HR) and Bacteriorhodopsin (BR) belong to a subfamily of heptahelical membrane proteins, the archaeal rhodopsins. They are found in
MS/MS of Peptides Manual Sequencing of Protonated Peptides
 S/S of Peptides anual Sequencing of Protonated Peptides Árpád Somogyi Associate irector CCIC, ass Spectrometry and Proteomics Laboratory SU July 11, 2018 Peptides Product Ion Scan Product ion spectra contain
S/S of Peptides anual Sequencing of Protonated Peptides Árpád Somogyi Associate irector CCIC, ass Spectrometry and Proteomics Laboratory SU July 11, 2018 Peptides Product Ion Scan Product ion spectra contain
Sensitive NMR Approach for Determining the Binding Mode of Tightly Binding Ligand Molecules to Protein Targets
 Supporting information Sensitive NMR Approach for Determining the Binding Mode of Tightly Binding Ligand Molecules to Protein Targets Wan-Na Chen, Christoph Nitsche, Kala Bharath Pilla, Bim Graham, Thomas
Supporting information Sensitive NMR Approach for Determining the Binding Mode of Tightly Binding Ligand Molecules to Protein Targets Wan-Na Chen, Christoph Nitsche, Kala Bharath Pilla, Bim Graham, Thomas
Potential Energy (hyper)surface
 The Molecular Dynamics Method Thermal motion of a lipid bilayer Water permeation through channels Selective sugar transport Potential Energy (hyper)surface What is Force? Energy U(x) F = " d dx U(x) Conformation
The Molecular Dynamics Method Thermal motion of a lipid bilayer Water permeation through channels Selective sugar transport Potential Energy (hyper)surface What is Force? Energy U(x) F = " d dx U(x) Conformation
BSc and MSc Degree Examinations
 Examination Candidate Number: Desk Number: BSc and MSc Degree Examinations 2018-9 Department : BIOLOGY Title of Exam: Molecular Biology and Biochemistry Part I Time Allowed: 1 hour and 30 minutes Marking
Examination Candidate Number: Desk Number: BSc and MSc Degree Examinations 2018-9 Department : BIOLOGY Title of Exam: Molecular Biology and Biochemistry Part I Time Allowed: 1 hour and 30 minutes Marking
NH 2. Biochemistry I, Fall Term Sept 9, Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter
 Biochemistry I, Fall Term Sept 9, 2005 Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter 3.1-3.4. Key Terms: ptical Activity, Chirality Peptide bond Condensation reaction ydrolysis
Biochemistry I, Fall Term Sept 9, 2005 Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter 3.1-3.4. Key Terms: ptical Activity, Chirality Peptide bond Condensation reaction ydrolysis
Retinal Proteins (Rhodopsins) Vision, Bioenergetics, Phototaxis. Bacteriorhodopsin s Photocycle. Bacteriorhodopsin s Photocycle
 Molecular chanisms of Photoactivation and Spectral Tuning in Retinal Proteins Emad Tajkhorshid Theoretical and Computational Biophysics Group Beckman Institute University of Illinois at Urbana-Champaign
Molecular chanisms of Photoactivation and Spectral Tuning in Retinal Proteins Emad Tajkhorshid Theoretical and Computational Biophysics Group Beckman Institute University of Illinois at Urbana-Champaign
