Laser-cooled fluorescence mass spectrometry using laser-cooled barium ions in a tandem linear ion trap
|
|
|
- Pierce Maximilian Ball
- 6 years ago
- Views:
Transcription
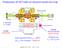
1 JOURNAL OF APPLIED PHYSICS VOLUME 89, NUMBER 8 15 APRIL 001 Laser-cooled fluorescence mass spectrometry using laser-cooled barium ions in a tandem linear ion trap Takashi Baba a) and Izumi Waki Central Research Laboratory, Hitachi, Ltd., Hatoyama, Saitama, , Japan Received 7 October 000; accepted for publication 18 January 001 We report on a mass spectrometry that uses laser-cooled barium ions to optically detect sample ions in a linear radio-frequency-quadrupole ion trap. To obtain a mass spectrum, we observe the intensity modulation of laser-induced fluorescence emitted by the barium ions, as we electrically excite the secular motions of sample ions that are sympathetically cooled by the barium ions. In a fluorescence mass spectrum of sample xenon ions, we identified the major isotopes of xenon. To estimate the absolute amount of the sample ions that contribute to the mass spectrum, we detect the ions using an electron multiplier that is absolutely calibrated by crystallized laser-cooled barium ions. We enhance transfer efficiency to the electron multiplier using a linear radio-frequency-quadrupole ion guide that is placed in tandem with the ion trap. Typically 30 ions of xenon isotope 131 Xe were detected with a signal-to-noise ratio of 30 in the fluorescence mass spectrum. 001 American Institute of Physics. DOI: / I. INTRODUCTION Sympathetic cooling 1 was proposed as a tool to cool ions which are not directly laser cooled. In atomic physics, several experimental and theoretical works were reported for frequency standards and for quantum computers. 3 In molecules, 4,5 we reported the detection of sympathetically cooled molecular ions using a mass spectrometric technique, which we call sympathetically laser-cooled fluorescence mass spectrometry or LCFMS. 6 To sympathetically cool molecular ions that originate from the vacuum residual gas, we stored them in a radio-frequency-quadrupole RFQ linear ion trap,6 8 simultaneously with laser-cooled magnesium ions 4 Mg. When we excited secular motions 9 of the molecular ions, the intensity of laser-induced fluorescence emitted by the magnesium ions was modulated. We identified the molecular ions H 3 O, NH 4, COH, and CO H and atomic ions of magnesium isotopes that were sympathetically cooled. We crudely estimated that about 80 5 Mg ions contributed to the fluorescence mass spectrum. Because most of the ions remained in the ion trap after several consecutive analyses, we expect that LCFMS can be used as an in situ tracing multistage mass spectroscopy MS/MS for a small amount of sample. In this article we present LCFMS using laser-cooled barium ions, 10 1 which are about six times heavier than the laser-cooled magnesium ions, where the amounts of sample and barium ions are absolutely calibrated. To be usable for mass spectrometry, it is necessary to understand the mechanism of LCFMS, which is determined by energy transfer from the molecular ions to the laser-cooled ions through Coulomb collisions when we excite the secular motion of the sample ions. Since mass and the number of ions are key parameters in such a multiparticle Coulomb system, it is necessary to understand how they affect the line profiles, such as a Electronic mail: baba@harl.hitachi.co.jp the asymmetry of the profiles. 6 In a previous report, 6 we did not have the means to measure the absolute amount of the molecular ions. Though we placed an electron multiplier by the ion trap miniature racetrack RFQ storage ring, the transfer efficiency to the electron multiplier was too poor for estimation of the trapped ion number because we extracted the trapped ions from a narrow gap space between the linear quadrupole electrodes. In this article, we perform LCFMS using a tandem linear radio-frequency-quadrupole ion trap. Here we measure the absolute amount of sample ions that contribute to the fluorescence mass spectrum using an absolutely calibrated electron multiplier that is calibrated by crystallized laser-cooled barium ions, as a standard of the absolute amount of trapped ions. We achieve high transfer efficiency from the ion trap to the electron multiplier using an ion guide placed between the ion trap and the electron multiplier. II. TANDEM LINEAR ION TRAP A. Configuration The ion trap that we constructed is a four-sectioned tandem linear ion trap Fig. 1, which has an end section a, an ion source section b, a linear ion trap section c, and an ion guide section d with an absolutely calibrated electron multiplier tube. All sections have the same quadrupole cross section. The distance from the quadrupole center to the electrodes r 0 is 3 mm. We made the electrodes of copper and plated the surface with gold to prevent oxidization. The ion trap section c is 50 mm in length. Laser-cooling beams pass along the center axis of the ion trap section. An ac dipole electric field for mass analysis is applied to the section. Other sections are curved so that the electron multiplier would not obstruct the laser beam path. Each section is separated with a1mmgap in order to electrostatistically insulate each other. The mechanical precision is 10 m /001/89(8)/459/7/$ American Institute of Physics
2 J. Appl. Phys., Vol. 89, No. 8, 15 April 001 T. Baba and I. Waki 4593 The equation of motion of an ion with mass m and electric charge e in the potential (x,y,z,t) is expressed by the well-known Mathieu equation 14 using dc parameters a x and a y and rf parameters q x and q y, a x a y 4eU m r 0 a, q x q y ev m r 0 q. 3 FIG. 1. The tandem ion trap is composed of an end section a, anion source section b, an ion trap section c, and an ion guide section d. The barium and xenon ions are created in the ion source section b using an electron gun and a barium oven. We perform mass analysis in the ion trap section c. When we measure the amount of trapped ions, the trapped ions are transferred to the electron multiplier through the ion guide section d. Figure shows the electric circuit to drive the foursectioned tandem linear ion trap. The circuit contains an rf circuit for ion trapping, dc bias circuits for control of the ion position, and an ac circuit for mass analysis. We apply nearly the same rf voltage to every section, whose frequencies are identical and whose phases and amplitudes are nearly the same. B. Operating parameters Since the theory of the linear ion trap is reported elsewhere,,7,8,13 we note some parameters used in this article. In the formulation, we define the z axis along the quadrupole centerline of the linear electrode. When a quadrupole rf voltage with amplitude V and frequency / and a quadrupole dc voltage U are applied to the linear quadrupole electrodes, the ideal linear quadrupole potential is given by x,y,z,t U V cos t x y. 1 r 0 FIG.. The driving electric circuit of the tandem ion trap is shown. When q 0.5 we can approximate (x,y,z,t) by a timeaveraged harmonic potential, i.e., pseudopotential, 9 x,y,z q 8 V x y U x y. 4 r 0 r 0 The depth of the pseudopotential D is given by D qv 8, when U is set to 0. The harmonic motions in the pseudopotential are called secular motions. The frequency of the secular motion, called the secular frequency, is given by 5 x a q, 6 y a q. 7 We set a 0 in the LCFMS, so that two secular frequencies x / and y / degenerate to the same value, ev m r. 8 0 In LCFMS we measure the secular frequency of sample ions, which is proportional to the specific charge of the ion Eq. 8, by detecting laser-induced fluorescence emitted by the laser-cooled ions as we oscillate the sample ions by a supplemental ac electric field. We fix the rf frequency at 1.76 MHz with an amplitude of 16 V so that q 0., pseudopotential depth is 3.6 V, and secular frequency is 10 khz for barium ions. C. Operation We create sample ions and barium ions in the ion source section b by impact of a 150 ev electron beam. The creation speed of xenon ions is controlled by the partial pressure of the xenon gas in the vacuum chamber, which we introduce through a leakage valve. We control creation speed of barium ions by the temperature of the atomic oven that generates the barium atomic vapor. The total amount of the trapped ions is controlled by the electron beam duration. We prevent ion loss from the end of the ion source by applying a dc voltage to the end electrode a. The dc bias V b of the ion source section is kept at 0.1 V higher than the ion trap section V c ( 0) so that the ions are transferred easily to the ion trap section. After the ion creation, V b is kept at 4 V, nearly
3 4594 J. Appl. Phys., Vol. 89, No. 8, 15 April 001 T. Baba and I. Waki FIG. 3. The storage lifetime was measured. After the ions are stored in the ion trap section for a certain storage period, they are transferred to the electron multiplier. The storage lifetime 1/ decay time is 160 s. the pseudopotential depth, higher than V c, so that ions are localized in the ion trap section. By separating the ion source from the ion trap section, we prevent the coating of barium metal onto the surface of the ion trap section c, because contact potential.7 V between gold and barium distorts the pseudopotential of the ion trap section. The ion guide section d functions as an end electrode of the ion trap section c as well. During ion storage, we keep the dc bias of the section the same as the pseudopotential depth 4 V. When we eject the trapped ions, we decrease the dc bias to 1 V. The ions are transferred to the electron multiplier through the ion guide section. The effect of curvature of the section can be neglected because the kinetic energy of the transferred ions is much less than the pseudopotential depth of the ion guide section. We place the electron multiplier at the end of the ion guide section with a 5 mm gap space. The window of the electron multiplier is 6 8 mm, which is enough to cover the exit area 6 mm 6 mm of the ion guide section. We apply kv to the first dynode of the electron multiplier, which accelerates and focuses the ejected ions, giving nearly 100% detection efficiency. We remove impurity ions lighter than xenon ions, such as Ba and CO, by increasing the rf voltage so that q reaches 0.7 for xenon ions. We did not remove ions heavier than barium ions, because such impurity ions are negligible in our vacuum. D. Basic performances of the tandem linear ion trap We measured storage lifetime, which is a basic performance of the tandem linear ion trap. We define the storage lifetime as the period during which the total amount of trapped ions decreases to half, which needs to be much longer than the time scale of LCFMS measurements, which is currently about 100 s. After storage of the ions for a certain time, we count the stored ions by the electron multiplier. We did not apply laser cooling or sympathetic cooling to the trapped ions in this storage lifetime measurement. Figure 3 shows the decay of the relative amount of stored ions after the storage time. In the figure, we show an exponential fit, whose decay constant, i.e., storage lifetime, is 160 s, which is long enough to FIG. 4. The transfer efficiency between sections was measured. We repeat round trip transfers between the sections c and b. The remaining ions are counted by the electron multiplier. Transfer efficiency per single transfer is 94%. perform LCFMS. The storage lifetime of the ion source section b was the same as the ion trap section c. Figure 4 shows a measurement of ejection efficiency from the ion trap section, which is another basic performance of the tandem linear ion trap. The efficiency affects the detection efficiency of trapped ions. We repeat round trip transfers between the ion trap section c and the ion source section b for N times. The remaining ions detected by the electron multiplier decreases by a factor N, where is the single transfer efficiency. Since each stay in a section takes 5 s, ion loss within a section is negligible because the stay is much shorter than the storage lifetime. The figure shows an exponential fit with 94% single transfer efficiency. Therefore, we assume that we can transfer the ions trapped in the ion trap section c with 94% efficiency through the ion guide section d toward the electron multiplier. III. LASER COOLING OF BARIUM IONS Figure 5 shows the system for laser cooling of barium ions Our transition for laser cooling is 6 S 1/ 6 P 1/ : GHz blue-green. The pump back transition from 5 D 3/ to 6 P 1/ is GHz red. The bluegreen laser beam is generated by a standing wave dye laser modified Coherent with coumarin 101 dye pumped by a long UV beam 70 nm multiline of an argon-ion laser Coherent Innova 400. The red laser beam is generated by a ring dye laser Coherent with kiton red dye pumped by a nm beam of an argon ion laser Coherent Innova 100. We stabilized the frequencies of the lasers by locking to molecular hyperfine spectra measured by Doppler free spectroscopy. 15 The blue-green reference is tellurium vapor and the red reference is iodine vapor. We tune the laser frequencies by acousto-optical frequency shifters so that the frequencies match the barium ion transitions. We apply a magnetic field parallel to the laser beam in order to eliminate the optical pumping to the D-state sublevels. 11 We observe the blue-green fluorescence either by a photomultiplier tube or by an image-intensified charge coupled device CCD camera.
4 J. Appl. Phys., Vol. 89, No. 8, 15 April 001 T. Baba and I. Waki 4595 FIG. 5. We use two dye lasers for laser cooling. Frequencies of the lasers are stabilized by referring to the hyperfine spectra of tellurium and iodine vapor. The laser frequencies are fine-tuned by acousto-optical frequency shifters AOM. Both laser beams are coupled coaxially and focused at the center of the linear ion trap section c. We observed laser-induced fluorescence by a photomultiplier tube or an image intensified CCD camera. IV. CALIBRATION OF THE ION DETECTION SYSTEM Figure 6 shows schematically how we measure the absolute amount of trapped ions destructively by the electron multiplier, which is absolutely calibrated by crystallized laser-cooled barium ions, whose absolute amount can be counted optically Fig. 7 inset. When a stable ion crystal is formed, we assume that the absolute amount of the trapped ions is equal to the number of the crystal lattice points in the crystal image. This assumption is justified because ions in a rf trap are known to exhibit a clear phase transition between a crystal phase and a gaseous phase. 0 Though the lattice contains 135,137 Ba ions that do not emit laser-induced fluorescence, all lattice points emit nearly equal intensity of fluorescence when we integrate the frames of CCD video pictures for a few seconds, because the trapped ions swap their positions within a fraction of a second due to collisions with neutral particles. 17,1 It is difficult to extract laser-cooled crystallized ions from the ion trap section to calibrate the electron multiplier because cooled ions can be bound in a weak stray field in the trap. It would be, in principle, possible to take the lasercooled ion string, heat them to approximate one electron volt energy, and then dump these ions into the electron multiplier. The ions could be heated either by a supplemental ac field or the blue-detuned laser beam. We found it, however, difficult to control the amount of heating, so the heated ions were often lost. Therefore the cooled ionic crystal was first used to calibrate the absolute rate of barium ion creation. The absolute rate of creation was then used to calibrate the absolute FIG. 6. Method for the measurement of the absolute amount of trapped ions. a Linear ion trap with an ion guide and an electron multiplier SEMT. In b e, the solid lines represent dc potential along the ion trap electrodes. b Since laser-cooled ions are crystallized when we laser cool them strongly, we are able to estimate the absolute amount of trapped ions when we count the number of lattice points. Here, we calibrate the ionization rate of barium ions. c Another set of freshly created barium ions are transferred to the electron multiplier. We calibrate the electron multiplier by using the ions whose absolute amount is estimated using the above measurement in b d and e. To examine the absolute amount of trapped ions, we transfer the ions to the electron multiplier that is absolutely calibrated. sensitivity of the electron multiplier using noncooled ions. Once the electron multiplier was calibrated, it was used to estimate the amount of trapped sample ions that cannot be laser cooled, as described below. Figure 7 shows the calibration of the absolute rate of barium ion creation. We plot the absolute amount of the trapped ions as a function of the ionization time, which is the duration of the electron beam. We observed crystals when the ionization time was between and 4 s. When the ioniza- FIG. 7. Image of the crystallized barium ion inset, and ionization rate of barium ions. The number of the lattice points of the crystallized ions is approximately equal to the amount of trapped ions. The ionization rate derived from a linear fit is derived ions/s by linear fitting of the figure. The error bars in the figure show the standard error of measurements of 10 0 sets of crystals.
5 4596 J. Appl. Phys., Vol. 89, No. 8, 15 April 001 T. Baba and I. Waki FIG. 8. Linearity of amount of the trapped ions to the ionization time of barium ions. The coefficient of the electron multiplier signal per unit ionization time is 11 9 counts/s. The error bars in the figure show the standard deviation of ten measurements. tion time was shorter than 1 s, we did not observe any fluorescence after laser cooling for a few minutes. Our imaging system did not resolve individual ions when the ionization time was longer than 4 s. A linear fit of Fig. 7 gives barium ion creation of ions/s. Then, we count the trapped barium ions, which are not laser cooled, by the electron multiplier for absolute calibration of its sensitivity Fig. 6 c. After storage of barium ions using the same ion-creation condition as the above calibration of absolute ionization rate, they were ejected from the ion trap section c, transferred through the ion guide section d and detected by the electron multiplier. Figure 8 shows a linear fit of the electron multiplier signal as a function of the ionization time. The coefficient of the electron multiplier signal per unit ionization time was 11 9 counts/s. We calibrated the absolute sensitivity of the ion-guided ion detection system using Figs. 7 and 8, where the condition of ionization and ion trapping were identical in both measurements. The calibrated absolute efficiency of the total detection system was counts/ion, which is reasonable because the kinetic energy of injected ions is kev. FIG. 9. Fine tuning of the secular frequencies. Using quadrupole dc voltage Va, we compensated the quadrupole component of the dc stray potential in order to make single dip of the secular frequency of barium ions (V a 0.13). Without compensation, the dip was split in two (V a 0). condition, the trap contained barium ions and xenon ions. The errors are mainly determined by Poisson statistics. After we laser cooled the ions and observed strong fluorescence, we decreased the laser-cooling efficiency by detuning the blue-green laser to 10 MHz in order to make a gas phase of ions. When we applied the analyzing ac voltage with 1 mv amplitude and scanned the frequency from 1.5 to 13.5 khz, we observed intensity modulations of laser-induced fluorescence Fig. 10. Since the secular frequency of 138 Ba was 1.3 khz Fig. 9, we calculated the masses of the observed dips using the inverse relation between mass and the secular frequency Eq. 8. V. LASER-COOLED FLUORESCENCE MASS SPECTROMETRY When we apply the mass analyzing ac voltage to lasercooled 138 Ba ions, we observed two dips that correspond to the secular frequencies in the spectrum (V a 0 in Fig. 9. When we apply a quadrupole dc voltage V a ( V1 V, V3 V, V4 V1 to the ion trap section c, the dip frequencies shift and overlap at V a 0.13 V. Thus the origin of the difference is a weak stray dc potential in the ion trap, which makes a 0 in Eq. 7. After the compensation, the secular frequency of 138 Ba is measured as 1.3 khz. We mass analyzed xenon isotope ions using LCFMS, in which the absolute amount of the sample ions was estimated using the calibrated electron multiplier. We created xenon and barium ions with the electron gun with a duration of 10sin 10 7 Pa partial pressure of xenon gas. In this FIG. 10. A laser-cooled fluorescence mass spectrum of xenon isotopes. The dips are identified as xenon isotopes compared with natural abundance of xenon. The estimated amount of 131 Xe ions is
6 J. Appl. Phys., Vol. 89, No. 8, 15 April 001 T. Baba and I. Waki 4597 VI. DISCUSSION In Fig. 10 the dip positions and the depths in the spectrum agreed with the natural abundance of xenon isotopes. Since the estimated total amount of xenon ions is ions in Fig. 10, the amount of each isotope ion according to the natural abundance of xenon should be: Xe ions, Xe, Xe, Xe, Xe, Xe, and Xe ions. Significant dips at m/e 134 and 136 in Fig. 10 may contain a contribution from barium isotope ions 134 Ba and 136 Ba, because we did not purify the barium isotope. Moreover, since 134 Ba and 136 Ba may emit laser-induced fluorescence by the laser-cooling beam tuned to 138 Ba, the dips at m/e 134 and 136 might be enhanced when excited by the ac analyzing field. Other dips should be pure xenon isotope ions. To estimate the absolute detection limit, we focus on the 131 Xe isotope. The noise one standard deviation of fluorescence fluctuation around m/e 131 is about onethirtieth of the equivalent width of the dip. When we define the absolute detection limit of LCFMS as three standard deviations of the noise, the absolute detection limit is 3 ions. Since the amount of 18 Xe and 130 Xe ions were about the same as this absolute detection limit, it is reasonable that we did not observe evident dips in Fig. 10 at m/e 18 and 130. Thus the position and the depth of the dips are consistent to the masses and the abundance of the xenon isotope ions. Asymmetry of the line profiles is observed in the figure, i.e., the lighter mass side is steeper than the heavy mass side. This asymmetry is independent of the sweep direction of the mass analyzing ac frequency. This asymmetry can be explained by space charge. 3 Space charge makes the pseudopotential shallower and the secular frequency of the sample ions shift to a lower frequency. Figure 10 shows a gradual increase of the fluorescence intensity in the direction of frequency scan. A probable reason is the loss of the sample ions when the ions are on resonance with the supplemental ac voltage due to insufficient damping by the sympathetic cooling. When the total amount of trapped ions decrease due to the ion loss, the fluorescence intensity can be increased because rf heating 0,4,5 is decreased. Shimizu et al. suggested that ions lighter than the lasercooled ions are not cooled by sympathetic cooling in a threedimensional RFQ ion trap Paul trap when the temperature of the trapped ions is higher than 1 K. 6,7 In their model of ion ion collision in the rf field, the one-dimensional sympathetic heating rate is proportional to m m s m, 9 m s m where, m s is the mass of the sample ion, m is the mass of laser-cooled ions, and 1 for q 0.5. When m m s, i.e., the sample ions are lighter than the laser-cooled ions, the one-dimensional heating rate is positive and the sample ions are sympathetically heated although the laser-cooled ions are strongly cooled. In contrast, we observed sympathetic cooling of ions lighter than laser-cooled ions in a linear RFQ ion trap. Xenon ions were sympathetically cooled by barium ions in this article, and H 3 O and NH 4 were sympathetically cooled by magnesium ions in the previous report. 6 The inconsistency may be solved by the difference between a Paul trap Shimizu s case and our linear ion trap. In the z direction of the linear ion trap, parameter is always 0 because of the absence of a radio-frequency field. When 0, the sample ions can be cooled even when m m s, because the z-direction heating rate is always negative i.e., cooling though the x- and y-direction heating rate is positive. In order to understand the mechanism of LCFMS further, such as the difference between linear traps and Paul traps, the asymmetry in line profiles, and the loss of ions during mass scan, we should perform experiments in which we change masses and the absolute amounts of the sample and the laser-cooled ions. These LCFMS characteristics should be compared with molecular dynamical simulation and modeling of LCFMS. We expect several advantages of laser-cooled barium ions for future applications of LCFMS. When we take q 0.4 for barium ions, for example, we can trap sample ions whose masses extend from m/e 50 to Mass range is important for organic molecules. In contrast, it is difficult to simultaneously trap the sample ions whose mass is 1000 together with lighter laser-cooled ions, such as Mg, because space charge effect 3 can limit the mass range. In addition, barium ions can be laser cooled using diode lasers, 1 which provide better stability of power and frequency as well as smaller size and easier operation than conventional dye lasers. A LCFMS mass spectrometer may be useful for low temperature molecular chemistry. 8,9 VII. CONCLUSIONS We successfully mass analyzed isotopes of xenon ions by laser-cooled fluorescence mass spectrometry using lasercooled barium ions in a four-sectioned tandem linear ion trap. We measured the absolute amount of the sample ions that contributed to a mass spectrum using an ion detection system composed of an electron multiplier and an ion guide section, which was calibrated using crystallized laser-cooled barium ions as a standard amount of trapped ions. Thirty ions of a xenon isotope were observed with a signal-to-noise ratio of 30. ACKNOWLEDGMENT We acknowledge valuable discussions with Dr. Yasuharu Hirai. 1 D. J. Larson, J. C. Bergquist, J. J. Bollinger, W. M. Itano, and D. J. Wineland, Phys. Rev. Lett. 57, D. J. Wineland et al., IEEE Trans. Ultrason. Ferroelectr. Freq. Control 37, D. Kielpinski et al., Phys. Rev. A 61, D. J. Wineland, R. E. Drullinger, and F. L. Walls, Phys. Rev. Lett. 40, K. Mølhave and M. Drewsen, Phys. Rev. A 6, T. Baba and I. Waki, Jpn. J. Appl. Phys., Part 35, L D. A. Church, J. Appl. Phys. 40, M. Welling, H. A. Schuessler, R. I. Thompson, and H. Walther, Int. J. Mass Spectrom. Ion Processes 17, H. G. Dehmelt, Adv. At. Mol. Phys. 3, W. Neuhauser, M. Hohenstatt, P. E. Toschek, and H. Dehmelt, Appl. Phys. 17,
7 4598 J. Appl. Phys., Vol. 89, No. 8, 15 April 001 T. Baba and I. Waki 11 G. Janik, W. Nagourney, and H. Dehmelt, J. Opt. Soc. Am. B, C. Rabb et al., Appl. Phys. B: Lasers Opt. B67, J. D. Prestage, G. R. Janik, G. J. Dick, and L. Maleki, IEEE Trans. Ultrason. Ferroelectr. Freq. Control 37, E. Fischer, Z. Phys. 156, T. W. Hänsch, M. D. Levenson, and A. L. Schawlow, Phys. Rev. Lett. 6, R. Brümel et al., Nature London 334, I. Waki, S. Kassner, G. Birkl, and H. Walther, Phys. Rev. Lett. 68, G. Birkl, S. Kassner, and H. Walther, Nature London 357, M. G. Raizen, J. M. Gilligan, J. C. Bergquist, W. M. Itano, and D. J. Wineland, Phys. Rev. A 45, R. Brümel, C. Kappler, W. Quint, and H. Walther, Phys. Rev. A 40, P. Bowe et al., Phys. Rev. Lett. 8, S. A. Fields, L. J. Burch, and W. A. Oran, Rev. Sci. Instrum. 48, J. F. J. Todd, R. M. Waldren, and R. E. Mather, Int. J. Mass Spectrom. Ion Processes 34, J. D. Prestage, A. Williams, L. Maleki, M. J. Djomehri, and E. Harabetian, Phys. Rev. Lett. 66, Y. Maeno, M. Tachikawa, Y. Moriwaki, and T. Shimizu, Jpn. J. Appl. Phys., Part 34, L T. Shimizu, Y. Moriwaki, Y. Maeno, Y. Ohsima, and M. Tachikawa, Genshikaku Kenkyu 41, Y. Moriwaki, M. Tachikawa, Y. Maeno, and T. Shimizu, Jpn. J. Appl. Phys., Part 31, L A. N. Nikolov et al., Phys. Rev. Lett. 84, R. Wynar, R. S. Freeland, D. J. Han, C. Ryu, and D. J. Heinzen, Science 87,
Sympathetic cooling rate of gas-phase ions in a radio-frequency-quadrupole ion trap
 Appl. Phys. B 74, 375 382 (22) DOI: 1.17/s342829 Applied Physics B Lasers and Optics t. baba i. waki Sympathetic cooling rate of gas-phase ions in a radio-frequency-quadrupole ion trap Central Research
Appl. Phys. B 74, 375 382 (22) DOI: 1.17/s342829 Applied Physics B Lasers and Optics t. baba i. waki Sympathetic cooling rate of gas-phase ions in a radio-frequency-quadrupole ion trap Central Research
Fluorescence profiles and cooling dynamics of laser-cooled Mg + ions in a linear rf ion trap
 Fluorescence profiles and cooling dynamics of laser-cooled Mg + ions in a linear rf ion trap XianZhen Zhao,* Vladimir L. Ryjkov, and Hans A. Schuessler Department of Physics, Texas A&M University, College
Fluorescence profiles and cooling dynamics of laser-cooled Mg + ions in a linear rf ion trap XianZhen Zhao,* Vladimir L. Ryjkov, and Hans A. Schuessler Department of Physics, Texas A&M University, College
Mass Analyzers. Principles of the three most common types magnetic sector, quadrupole and time of flight - will be discussed herein.
 Mass Analyzers After the production of ions in ion sources, the next critical step in mass spectrometry is to separate these gas phase ions according to their mass-to-charge ratio (m/z). Ions are extracted
Mass Analyzers After the production of ions in ion sources, the next critical step in mass spectrometry is to separate these gas phase ions according to their mass-to-charge ratio (m/z). Ions are extracted
Isotope shifts of the 4s 2 1 S 0 \4s5p 1 P 1 transition and hyperfine splitting of the 4s5p 1 P 1 state in calcium
 PHYSICAL REVIEW A 69, 042502 (2004) Isotope shifts of the 4s 2 1 S 0 \4s5p 1 P 1 transition and hyperfine splitting of the 4s5p 1 P 1 state in calcium A. Mortensen,* J. J. T. Lindballe, I. S. Jensen, P.
PHYSICAL REVIEW A 69, 042502 (2004) Isotope shifts of the 4s 2 1 S 0 \4s5p 1 P 1 transition and hyperfine splitting of the 4s5p 1 P 1 state in calcium A. Mortensen,* J. J. T. Lindballe, I. S. Jensen, P.
Measurement of storage time, estimation of ion number and study of absorption line profile in a Paul trap
 PRAMANA c Indian Academy of Sciences Vol. 67, No. 6 journal of December 2006 physics pp. 1087 1097 Measurement of storage time, estimation of ion number and study of absorption line profile in a Paul trap
PRAMANA c Indian Academy of Sciences Vol. 67, No. 6 journal of December 2006 physics pp. 1087 1097 Measurement of storage time, estimation of ion number and study of absorption line profile in a Paul trap
Coulomb crystal extraction from an ion trap for application to nano-beam source"
 Coulomb crystal extraction from an ion trap for application to nano-beam source" K. Ito, K. Izawa, H. Higaki and H. Okamoto,! Aadvanced Sciences of Matter, Hiroshima University,! 1-3-1 Kagamiyama, Higashi-Hiroshima,
Coulomb crystal extraction from an ion trap for application to nano-beam source" K. Ito, K. Izawa, H. Higaki and H. Okamoto,! Aadvanced Sciences of Matter, Hiroshima University,! 1-3-1 Kagamiyama, Higashi-Hiroshima,
Zero-point cooling and low heating of trapped 111 Cd + ions
 PHYSICAL REVIEW A 70, 043408 (2004) Zero-point cooling and low heating of trapped 111 Cd + ions L. Deslauriers, P. C. Haljan, P. J. Lee, K-A. Brickman, B. B. Blinov, M. J. Madsen, and C. Monroe FOCUS Center,
PHYSICAL REVIEW A 70, 043408 (2004) Zero-point cooling and low heating of trapped 111 Cd + ions L. Deslauriers, P. C. Haljan, P. J. Lee, K-A. Brickman, B. B. Blinov, M. J. Madsen, and C. Monroe FOCUS Center,
Formation process of large ion Coulomb crystals in linear Paul traps
 PHYSICAL REVIEW A 66, 013412 2002 Formation process of large ion Coulomb crystals in linear Paul traps L. Hornekær* and M. Drewsen Institute of Physics and Astronomy, Aarhus University, DK-8000 Aarhus,
PHYSICAL REVIEW A 66, 013412 2002 Formation process of large ion Coulomb crystals in linear Paul traps L. Hornekær* and M. Drewsen Institute of Physics and Astronomy, Aarhus University, DK-8000 Aarhus,
Design considerations for linear Paul trap mass spectrometer under development
 Design considerations for linear Paul trap mass spectrometer under development S.Sevugarajan and A.G.Menon Department of Instrumentation, Indian Institute of Science, Bangalore 561, India. Abstract This
Design considerations for linear Paul trap mass spectrometer under development S.Sevugarajan and A.G.Menon Department of Instrumentation, Indian Institute of Science, Bangalore 561, India. Abstract This
Observing a single hydrogen-like ion in a Penning trap at T = 4K
 Hyperfine Interactions 115 (1998) 185 192 185 Observing a single hydrogen-like ion in a Penning trap at T = 4K M. Diederich a,h.häffner a, N. Hermanspahn a,m.immel a,h.j.kluge b,r.ley a, R. Mann b,w.quint
Hyperfine Interactions 115 (1998) 185 192 185 Observing a single hydrogen-like ion in a Penning trap at T = 4K M. Diederich a,h.häffner a, N. Hermanspahn a,m.immel a,h.j.kluge b,r.ley a, R. Mann b,w.quint
Ion trap. 3D Quadrupole Trap. -Mass spectrometer -Ion trap (Quadrupol Ion Storage, Quistor) 18. April
 Ion trap 3D Quadrupole Trap -Mass spectrometer -Ion trap (Quadrupol Ion Storage, Quistor) 18. April 011 1 The trajectories of the ions can be given: They form Mathieu differential equations: with r + Ion
Ion trap 3D Quadrupole Trap -Mass spectrometer -Ion trap (Quadrupol Ion Storage, Quistor) 18. April 011 1 The trajectories of the ions can be given: They form Mathieu differential equations: with r + Ion
Fundamentals of Mass Spectrometry. Fundamentals of Mass Spectrometry. Learning Objective. Proteomics
 Mass spectrometry (MS) is the technique for protein identification and analysis by production of charged molecular species in vacuum, and their separation by magnetic and electric fields based on mass
Mass spectrometry (MS) is the technique for protein identification and analysis by production of charged molecular species in vacuum, and their separation by magnetic and electric fields based on mass
NEW ION TRAP FOR FREQUENCY STANDARD APPLICATIONS*
 NEW ION TRAP FOR FREQUENCY STANDARD APPLICATIONS* J. D. Prestage, G. J. Dick, and L. Maleki California Institute of Technology Jet Propulsion Laboratory 4800 Oak Grove Drive Pasadena, California 91109
NEW ION TRAP FOR FREQUENCY STANDARD APPLICATIONS* J. D. Prestage, G. J. Dick, and L. Maleki California Institute of Technology Jet Propulsion Laboratory 4800 Oak Grove Drive Pasadena, California 91109
Ion trap quantum processor
 Ion trap quantum processor Laser pulses manipulate individual ions row of qubits in a linear Paul trap forms a quantum register Effective ion-ion interaction induced by laser pulses that excite the ion`s
Ion trap quantum processor Laser pulses manipulate individual ions row of qubits in a linear Paul trap forms a quantum register Effective ion-ion interaction induced by laser pulses that excite the ion`s
1. Introduction. 2. New approaches
 New Approaches To An Indium Ion Optical Frequency Standard Kazuhiro HAYASAKA National Institute of Information and Communications Technology(NICT) e-mail:hayasaka@nict.go.jp ECTI200 . Introduction Outline
New Approaches To An Indium Ion Optical Frequency Standard Kazuhiro HAYASAKA National Institute of Information and Communications Technology(NICT) e-mail:hayasaka@nict.go.jp ECTI200 . Introduction Outline
Extrel Application Note
 Extrel Application Note Real-Time Plasma Monitoring and Detection of Trace H 2 O and HF Species in an Argon Based Plasma Jian Wei, 575 Epsilon Drive, Pittsburgh, PA 15238. (Presented at the 191st Electrochemical
Extrel Application Note Real-Time Plasma Monitoring and Detection of Trace H 2 O and HF Species in an Argon Based Plasma Jian Wei, 575 Epsilon Drive, Pittsburgh, PA 15238. (Presented at the 191st Electrochemical
Physics of and in Ion Traps
 Physics of and in Ion Traps Proposed Topics: TRIUMF, Vancouver June 01 Basics of Paul- and Penning-traps (equ. of motion, trap geometries, influence of trap imperfections,) Ion detection and cooling (Buffer
Physics of and in Ion Traps Proposed Topics: TRIUMF, Vancouver June 01 Basics of Paul- and Penning-traps (equ. of motion, trap geometries, influence of trap imperfections,) Ion detection and cooling (Buffer
CEE 772 Lecture #27 12/10/2014. CEE 772: Instrumental Methods in Environmental Analysis
 Updated: 10 December 2014 Print version CEE 772: Instrumental Methods in Environmental Analysis Lecture #21 Mass Spectrometry: Mass Filters & Spectrometers (Skoog, Chapt. 20, pp.511 524) (Harris, Chapt.
Updated: 10 December 2014 Print version CEE 772: Instrumental Methods in Environmental Analysis Lecture #21 Mass Spectrometry: Mass Filters & Spectrometers (Skoog, Chapt. 20, pp.511 524) (Harris, Chapt.
CEE 772: Instrumental Methods in Environmental Analysis
 Updated: 10 December 2014 Print version CEE 772: Instrumental Methods in Environmental Analysis Lecture #21 Mass Spectrometry: Mass Filters & Spectrometers (Skoog, Chapt. 20, pp.511-524) (Harris, Chapt.
Updated: 10 December 2014 Print version CEE 772: Instrumental Methods in Environmental Analysis Lecture #21 Mass Spectrometry: Mass Filters & Spectrometers (Skoog, Chapt. 20, pp.511-524) (Harris, Chapt.
Investigating a qubit candidate: Spectroscopy on the S 1/2 to D 5/2 transition of a trapped calcium ion in a linear Paul trap
 Investigating a qubit candidate: Spectroscopy on the S 1/2 to D 5/2 transition of a trapped calcium ion in a linear Paul trap H. C. Nägerl,* Ch. Roos, D. Leibfried, H. Rohde, G. Thalhammer, J. Eschner,
Investigating a qubit candidate: Spectroscopy on the S 1/2 to D 5/2 transition of a trapped calcium ion in a linear Paul trap H. C. Nägerl,* Ch. Roos, D. Leibfried, H. Rohde, G. Thalhammer, J. Eschner,
TANDEM MASS SPECTROSCOPY
 TANDEM MASS SPECTROSCOPY 1 MASS SPECTROMETER TYPES OF MASS SPECTROMETER PRINCIPLE TANDEM MASS SPECTROMETER INSTRUMENTATION QUADRAPOLE MASS ANALYZER TRIPLE QUADRAPOLE MASS ANALYZER TIME OF FLIGHT MASS ANALYSER
TANDEM MASS SPECTROSCOPY 1 MASS SPECTROMETER TYPES OF MASS SPECTROMETER PRINCIPLE TANDEM MASS SPECTROMETER INSTRUMENTATION QUADRAPOLE MASS ANALYZER TRIPLE QUADRAPOLE MASS ANALYZER TIME OF FLIGHT MASS ANALYSER
Chapter 4 Scintillation Detectors
 Med Phys 4RA3, 4RB3/6R03 Radioisotopes and Radiation Methodology 4-1 4.1. Basic principle of the scintillator Chapter 4 Scintillation Detectors Scintillator Light sensor Ionizing radiation Light (visible,
Med Phys 4RA3, 4RB3/6R03 Radioisotopes and Radiation Methodology 4-1 4.1. Basic principle of the scintillator Chapter 4 Scintillation Detectors Scintillator Light sensor Ionizing radiation Light (visible,
Opportunities with collinear laser spectroscopy at DESIR:
 Opportunities with collinear laser spectroscopy at DESIR: the LUMIERE facility GOALS of LUMIERE experiments: Gerda Neyens, K.U. Leuven, Belgium (1) measure ground state properties of exotic isotopes: (see
Opportunities with collinear laser spectroscopy at DESIR: the LUMIERE facility GOALS of LUMIERE experiments: Gerda Neyens, K.U. Leuven, Belgium (1) measure ground state properties of exotic isotopes: (see
Mass Spectrometry in MCAL
 Mass Spectrometry in MCAL Two systems: GC-MS, LC-MS GC seperates small, volatile, non-polar material MS is detection devise (Agilent 320-MS TQ Mass Spectrometer) Full scan monitoring SIM single ion monitoring
Mass Spectrometry in MCAL Two systems: GC-MS, LC-MS GC seperates small, volatile, non-polar material MS is detection devise (Agilent 320-MS TQ Mass Spectrometer) Full scan monitoring SIM single ion monitoring
Lecture 11, May 11, 2017
 Lecture 11, May 11, 2017 This week: Atomic Ions for QIP Ion Traps Vibrational modes Preparation of initial states Read-Out Single-Ion Gates Two-Ion Gates Introductory Review Articles: D. Leibfried, R.
Lecture 11, May 11, 2017 This week: Atomic Ions for QIP Ion Traps Vibrational modes Preparation of initial states Read-Out Single-Ion Gates Two-Ion Gates Introductory Review Articles: D. Leibfried, R.
Mass Spectrometry. What is Mass Spectrometry?
 Mass Spectrometry What is Mass Spectrometry? Mass Spectrometry (MS): The generation of gaseous ions from a sample, separation of these ions by mass-to-charge ratio, and measurement of relative abundance
Mass Spectrometry What is Mass Spectrometry? Mass Spectrometry (MS): The generation of gaseous ions from a sample, separation of these ions by mass-to-charge ratio, and measurement of relative abundance
CMSC 33001: Novel Computing Architectures and Technologies. Lecture 06: Trapped Ion Quantum Computing. October 8, 2018
 CMSC 33001: Novel Computing Architectures and Technologies Lecturer: Kevin Gui Scribe: Kevin Gui Lecture 06: Trapped Ion Quantum Computing October 8, 2018 1 Introduction Trapped ion is one of the physical
CMSC 33001: Novel Computing Architectures and Technologies Lecturer: Kevin Gui Scribe: Kevin Gui Lecture 06: Trapped Ion Quantum Computing October 8, 2018 1 Introduction Trapped ion is one of the physical
Lecture 22 Ion Beam Techniques
 Lecture 22 Ion Beam Techniques Schroder: Chapter 11.3 1/44 Announcements Homework 6/6: Will be online on later today. Due Wednesday June 6th at 10:00am. I will return it at the final exam (14 th June).
Lecture 22 Ion Beam Techniques Schroder: Chapter 11.3 1/44 Announcements Homework 6/6: Will be online on later today. Due Wednesday June 6th at 10:00am. I will return it at the final exam (14 th June).
Lecture 8: Mass Spectrometry
 intensity Lecture 8: Mass Spectrometry Relative abundance m/z 1 Ethylbenzene experiment CH 2 CH 3 + m/z = 106 CH 2 + m/z = 91 C 8 H 10 MW = 106 CH + m/z = 77 + 2 2 What information can we get from MS spectrum?
intensity Lecture 8: Mass Spectrometry Relative abundance m/z 1 Ethylbenzene experiment CH 2 CH 3 + m/z = 106 CH 2 + m/z = 91 C 8 H 10 MW = 106 CH + m/z = 77 + 2 2 What information can we get from MS spectrum?
Minimization of ion micromotion in a Paul trap
 JOURNAL OF APPLIED PHYSICS VOLUME 83, NUMBER 10 15 MAY 1998 Minimization of ion micromotion in a Paul trap D. J. Berkeland, a) J. D. Miller, b) J. C. Bergquist, W. M. Itano, and D. J. Wineland National
JOURNAL OF APPLIED PHYSICS VOLUME 83, NUMBER 10 15 MAY 1998 Minimization of ion micromotion in a Paul trap D. J. Berkeland, a) J. D. Miller, b) J. C. Bergquist, W. M. Itano, and D. J. Wineland National
Lecture 8: Mass Spectrometry
 intensity Lecture 8: Mass Spectrometry Relative abundance m/z 1 Ethylbenzene CH 2 CH 3 + m/z = 106 CH 2 + m/z = 91 C 8 H 10 MW = 106 CH + m/z = 77 + 2 2 What information can be obtained from a MS spectrum?
intensity Lecture 8: Mass Spectrometry Relative abundance m/z 1 Ethylbenzene CH 2 CH 3 + m/z = 106 CH 2 + m/z = 91 C 8 H 10 MW = 106 CH + m/z = 77 + 2 2 What information can be obtained from a MS spectrum?
3. Synchrotrons. Synchrotron Basics
 1 3. Synchrotrons Synchrotron Basics What you will learn about 2 Overview of a Synchrotron Source Losing & Replenishing Electrons Storage Ring and Magnetic Lattice Synchrotron Radiation Flux, Brilliance
1 3. Synchrotrons Synchrotron Basics What you will learn about 2 Overview of a Synchrotron Source Losing & Replenishing Electrons Storage Ring and Magnetic Lattice Synchrotron Radiation Flux, Brilliance
Types of Analyzers: Quadrupole: mass filter -part1
 16 Types of Analyzers: Sector or double focusing: magnetic and electric Time-of-flight (TOF) Quadrupole (mass filter) Linear ion trap Quadrupole Ion Trap (3D trap) FTICR fourier transform ion cyclotron
16 Types of Analyzers: Sector or double focusing: magnetic and electric Time-of-flight (TOF) Quadrupole (mass filter) Linear ion trap Quadrupole Ion Trap (3D trap) FTICR fourier transform ion cyclotron
Quadrupole Storage Mass Spectrometry
 Quadrupole Storage Mass Spectrometry RAYMOND E. MARCH AND RICHARD J. HUGHES Trent University Peterborough, Ontario, Canada with a historical review by John F. J. Tbdd University of Kent Canterbury, Kent,
Quadrupole Storage Mass Spectrometry RAYMOND E. MARCH AND RICHARD J. HUGHES Trent University Peterborough, Ontario, Canada with a historical review by John F. J. Tbdd University of Kent Canterbury, Kent,
Citation for published version (APA): Mollema, A. K. (2008). Laser cooling, trapping and spectroscopy of calcium isotopes s.n.
 University of Groningen Laser cooling, trapping and spectroscopy of calcium isotopes Mollema, Albert Kornelis IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you
University of Groningen Laser cooling, trapping and spectroscopy of calcium isotopes Mollema, Albert Kornelis IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you
Harris: Quantitative Chemical Analysis, Eight Edition
 Harris: Quantitative Chemical Analysis, Eight Edition CHAPTER 21: MASS SPECTROMETRY CHAPTER 21: Opener 21.0 Mass Spectrometry Mass Spectrometry provides information about 1) The elemental composition of
Harris: Quantitative Chemical Analysis, Eight Edition CHAPTER 21: MASS SPECTROMETRY CHAPTER 21: Opener 21.0 Mass Spectrometry Mass Spectrometry provides information about 1) The elemental composition of
Collinear laser spectroscopy of radioactive isotopes at IGISOL 4 Liam Vormawah
 Collinear laser spectroscopy of radioactive isotopes at IGISOL 4 Liam Vormawah University of Liverpool Introduction Collinear laser spectroscopy Nuclear properties from hyperfine structures Isotope shifts
Collinear laser spectroscopy of radioactive isotopes at IGISOL 4 Liam Vormawah University of Liverpool Introduction Collinear laser spectroscopy Nuclear properties from hyperfine structures Isotope shifts
ICPMS Doherty Lecture 1
 ICPMS Doherty Lecture 1 Mass Spectrometry This material provides some background on how to measure isotope abundances by means of mass spectrometry. Mass spectrometers create and separate ionized atoms
ICPMS Doherty Lecture 1 Mass Spectrometry This material provides some background on how to measure isotope abundances by means of mass spectrometry. Mass spectrometers create and separate ionized atoms
Chemistry Instrumental Analysis Lecture 35. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 35 Principle components: Inlet Ion source Mass analyzer Ion transducer Pumps Signal processor Mass analyzers Quadrupole Time of Flight Double Focusing Ion
Chemistry 4631 Instrumental Analysis Lecture 35 Principle components: Inlet Ion source Mass analyzer Ion transducer Pumps Signal processor Mass analyzers Quadrupole Time of Flight Double Focusing Ion
(Refer Slide Time 00:09) (Refer Slide Time 00:13)
 (Refer Slide Time 00:09) Mass Spectrometry Based Proteomics Professor Sanjeeva Srivastava Department of Biosciences and Bioengineering Indian Institute of Technology, Bombay Mod 02 Lecture Number 09 (Refer
(Refer Slide Time 00:09) Mass Spectrometry Based Proteomics Professor Sanjeeva Srivastava Department of Biosciences and Bioengineering Indian Institute of Technology, Bombay Mod 02 Lecture Number 09 (Refer
Energetic particles and their detection in situ (particle detectors) Part II. George Gloeckler
 Energetic particles and their detection in situ (particle detectors) Part II George Gloeckler University of Michigan, Ann Arbor, MI University of Maryland, College Park, MD Simple particle detectors Gas-filled
Energetic particles and their detection in situ (particle detectors) Part II George Gloeckler University of Michigan, Ann Arbor, MI University of Maryland, College Park, MD Simple particle detectors Gas-filled
HYPERFINE STRUCTURE CONSTANTS IN THE 102D3/2 AND 112D 3/2 STATES OF 85Rb M. GLOW
 Vol. 83 (1993) ACTA PHYSICA POLONICA A No. 2 HYPERFINE STRUCTURE CONSTANTS IN THE 102D3/2 AND 112D 3/2 STATES OF 85Rb M. GLOW Institute of Physics, Polish Academy of Sciences Al. Lotników 32/46, 02-668
Vol. 83 (1993) ACTA PHYSICA POLONICA A No. 2 HYPERFINE STRUCTURE CONSTANTS IN THE 102D3/2 AND 112D 3/2 STATES OF 85Rb M. GLOW Institute of Physics, Polish Academy of Sciences Al. Lotników 32/46, 02-668
Ultracold atoms and molecules
 Advanced Experimental Techniques Ultracold atoms and molecules Steven Knoop s.knoop@vu.nl VU, June 014 1 Ultracold atoms laser cooling evaporative cooling BEC Bose-Einstein condensation atom trap: magnetic
Advanced Experimental Techniques Ultracold atoms and molecules Steven Knoop s.knoop@vu.nl VU, June 014 1 Ultracold atoms laser cooling evaporative cooling BEC Bose-Einstein condensation atom trap: magnetic
Precision VUV spectroscopy of Ar I at 105 nm
 J. Phys. B: At. Mol. Opt. Phys. 32 (999) L5 L56. Printed in the UK PII: S0953-4075(99)05625-4 LETTER TO THE EDITOR Precision VUV spectroscopy of Ar I at 05 nm I Velchev, W Hogervorst and W Ubachs Vrije
J. Phys. B: At. Mol. Opt. Phys. 32 (999) L5 L56. Printed in the UK PII: S0953-4075(99)05625-4 LETTER TO THE EDITOR Precision VUV spectroscopy of Ar I at 05 nm I Velchev, W Hogervorst and W Ubachs Vrije
Mass Spectroscopy. Base peak. Molecular Ion peak. The positively charged fragments produced are separated, based on their mass/charge (m/z) ratio. M+.
 Mass spectrometry is the study of systems causing the formation of gaseous ions, with or without fragmentation, which are then characteried by their mass to charge ratios (m/) and relative abundances.
Mass spectrometry is the study of systems causing the formation of gaseous ions, with or without fragmentation, which are then characteried by their mass to charge ratios (m/) and relative abundances.
Effect of Spiral Microwave Antenna Configuration on the Production of Nano-crystalline Film by Chemical Sputtering in ECR Plasma
 THE HARRIS SCIENCE REVIEW OF DOSHISHA UNIVERSITY, VOL. 56, No. 1 April 2015 Effect of Spiral Microwave Antenna Configuration on the Production of Nano-crystalline Film by Chemical Sputtering in ECR Plasma
THE HARRIS SCIENCE REVIEW OF DOSHISHA UNIVERSITY, VOL. 56, No. 1 April 2015 Effect of Spiral Microwave Antenna Configuration on the Production of Nano-crystalline Film by Chemical Sputtering in ECR Plasma
Ion traps. Trapping of charged particles in electromagnetic. Laser cooling, sympathetic cooling, optical clocks
 Ion traps Trapping of charged particles in electromagnetic fields Dynamics of trapped ions Applications to nuclear physics and QED The Paul trap Laser cooling, sympathetic cooling, optical clocks Coulomb
Ion traps Trapping of charged particles in electromagnetic fields Dynamics of trapped ions Applications to nuclear physics and QED The Paul trap Laser cooling, sympathetic cooling, optical clocks Coulomb
LASER SPECTROSCOPIC STUDIES OF NEUTRON-DEFICIENT EUROPIUM AND GADOLINIUM ISOTOPES
 LASER SPECTROSCOPIC STUDIES OF NEUTRON-DEFICIENT EUROPIUM AND GADOLINIUM ISOTOPES A.E. Barzakh, D.V. Fedorov, A.M. Ionan, V.S. Ivanov, F.V. Moroz, K.A. Mezilev, S.Yu. Orlov, V.N. Panteleev, Yu.M. Volkov
LASER SPECTROSCOPIC STUDIES OF NEUTRON-DEFICIENT EUROPIUM AND GADOLINIUM ISOTOPES A.E. Barzakh, D.V. Fedorov, A.M. Ionan, V.S. Ivanov, F.V. Moroz, K.A. Mezilev, S.Yu. Orlov, V.N. Panteleev, Yu.M. Volkov
CHAPTER D4 ORTHOGONAL TIME OF FLIGHT OPTICS
 Back to Basics Section D: Ion Optics CHAPTER D4 ORTHOGONAL TIME OF FLIGHT OPTICS TABLE OF CONTENTS QuickGuide...413 Summary...415 Introduction...417 The physical basis of orthogonal TOF....... 419 Pulsedmainbeamsofions...421
Back to Basics Section D: Ion Optics CHAPTER D4 ORTHOGONAL TIME OF FLIGHT OPTICS TABLE OF CONTENTS QuickGuide...413 Summary...415 Introduction...417 The physical basis of orthogonal TOF....... 419 Pulsedmainbeamsofions...421
Because light behaves like a wave, we can describe it in one of two ways by its wavelength or by its frequency.
 Light We can use different terms to describe light: Color Wavelength Frequency Light is composed of electromagnetic waves that travel through some medium. The properties of the medium determine how light
Light We can use different terms to describe light: Color Wavelength Frequency Light is composed of electromagnetic waves that travel through some medium. The properties of the medium determine how light
Secondary Ion Mass Spectroscopy (SIMS)
 Secondary Ion Mass Spectroscopy (SIMS) Analyzing Inorganic Solids * = under special conditions ** = semiconductors only + = limited number of elements or groups Analyzing Organic Solids * = under special
Secondary Ion Mass Spectroscopy (SIMS) Analyzing Inorganic Solids * = under special conditions ** = semiconductors only + = limited number of elements or groups Analyzing Organic Solids * = under special
arxiv:quant-ph/ v1 15 Sep 2003
 Molecular Dynamics Simulation of Sympathetic Crystallization of Molecular Ions Stephan Schiller and Claus Lämmerzahl Institute for Experimental Physics, Heinrich Heine University Düsseldoerf, 40225 Düsseldorf,
Molecular Dynamics Simulation of Sympathetic Crystallization of Molecular Ions Stephan Schiller and Claus Lämmerzahl Institute for Experimental Physics, Heinrich Heine University Düsseldoerf, 40225 Düsseldorf,
Chemistry 311: Topic 3 - Mass Spectrometry
 Mass Spectroscopy: A technique used to measure the mass-to-charge ratio of molecules and atoms. Often characteristic ions produced by an induced unimolecular dissociation of a molecule are measured. These
Mass Spectroscopy: A technique used to measure the mass-to-charge ratio of molecules and atoms. Often characteristic ions produced by an induced unimolecular dissociation of a molecule are measured. These
Mass Analyzers. Ion Trap, FTICR, Orbitrap. CU- Boulder CHEM 5181: Mass Spectrometry & Chromatography. Prof. Jose-Luis Jimenez
 Mass Analyzers Ion Trap, FTICR, Orbitrap CU- Boulder CHEM 5181: Mass Spectrometry & Chromatography Prof. Jose-Luis Jimenez Last Update: Oct. 014 Some slides from Dr. Joel Kimmel (007) MS Interpretation
Mass Analyzers Ion Trap, FTICR, Orbitrap CU- Boulder CHEM 5181: Mass Spectrometry & Chromatography Prof. Jose-Luis Jimenez Last Update: Oct. 014 Some slides from Dr. Joel Kimmel (007) MS Interpretation
Microwave and optical spectroscopy in r.f. traps Application to atomic clocks
 Microwave and optical spectroscopy in r.f. traps Application to atomic clocks Microwave spectroscopy for hyperfine structure t measurements Energy of a hyperfine state Hyperfine coupling constants: A:
Microwave and optical spectroscopy in r.f. traps Application to atomic clocks Microwave spectroscopy for hyperfine structure t measurements Energy of a hyperfine state Hyperfine coupling constants: A:
arxiv:physics/ v1 [physics.plasm-ph] 5 Nov 2004
![arxiv:physics/ v1 [physics.plasm-ph] 5 Nov 2004 arxiv:physics/ v1 [physics.plasm-ph] 5 Nov 2004](/thumbs/72/67704246.jpg) Ion Resonance Instability in the ELTRAP electron plasma G. Bettega, 1 F. Cavaliere, 2 M. Cavenago, 3 A. Illiberi, 1 R. Pozzoli, 1 and M. Romé 1 1 INFM Milano Università, INFN Sezione di Milano, Dipartimento
Ion Resonance Instability in the ELTRAP electron plasma G. Bettega, 1 F. Cavaliere, 2 M. Cavenago, 3 A. Illiberi, 1 R. Pozzoli, 1 and M. Romé 1 1 INFM Milano Università, INFN Sezione di Milano, Dipartimento
Laser cooling of 173 Yb for isotope separation and precision hyperfine spectroscopy
 Laser cooling of 173 Yb for isotope separation and precision hyperfine spectroscopy Dipankar Das and Vasant Natarajan* Department of Physics, Indian Institute of Science, Bangalore 560 012, India Received
Laser cooling of 173 Yb for isotope separation and precision hyperfine spectroscopy Dipankar Das and Vasant Natarajan* Department of Physics, Indian Institute of Science, Bangalore 560 012, India Received
spectroscopy of cold molecular ions
 Workshop on an Optical Clock Mission in ESA s Cosmic Vision Program Düsseldorf 8. - 9. 3. 2007 High-resolution spectroscopy of cold molecular ions B. Roth, J. Koelemeij, I. Ernsting, A. Wicht, S. Schiller
Workshop on an Optical Clock Mission in ESA s Cosmic Vision Program Düsseldorf 8. - 9. 3. 2007 High-resolution spectroscopy of cold molecular ions B. Roth, J. Koelemeij, I. Ernsting, A. Wicht, S. Schiller
Photoelectron Spectroscopy using High Order Harmonic Generation
 Photoelectron Spectroscopy using High Order Harmonic Generation Alana Ogata Yamanouchi Lab, University of Tokyo ABSTRACT The analysis of photochemical processes has been previously limited by the short
Photoelectron Spectroscopy using High Order Harmonic Generation Alana Ogata Yamanouchi Lab, University of Tokyo ABSTRACT The analysis of photochemical processes has been previously limited by the short
Different ion-qubit choises. - One electron in the valence shell; Alkali like 2 S 1/2 ground state.
 Different ion-qubit choises - One electron in the valence shell; Alkali like 2 S 1/2 ground state. Electronic levels Structure n 2 P 3/2 n 2 P n 2 P 1/2 w/o D Be + Mg + Zn + Cd + 313 nm 280 nm 206 nm 226
Different ion-qubit choises - One electron in the valence shell; Alkali like 2 S 1/2 ground state. Electronic levels Structure n 2 P 3/2 n 2 P n 2 P 1/2 w/o D Be + Mg + Zn + Cd + 313 nm 280 nm 206 nm 226
Crystalline beam emulations in a pulse-excited linear Paul trap
 PHYSICS OF PLASMAS VOLUME 8, NUMBER 4 APRIL 200 Crystalline beam emulations in a pulse-excited linear Paul trap Niels Kjærgaard a) and Michael Drewsen Institute of Physics and Astronomy, Aarhus University,
PHYSICS OF PLASMAS VOLUME 8, NUMBER 4 APRIL 200 Crystalline beam emulations in a pulse-excited linear Paul trap Niels Kjærgaard a) and Michael Drewsen Institute of Physics and Astronomy, Aarhus University,
SRF GUN CHARACTERIZATION - PHASE SPACE AND DARK CURRENT MEASUREMENTS AT ELBE*
 SRF GUN CHARACTERIZATION - PHASE SPACE AND DARK CURRENT MEASUREMENTS AT ELBE* E. Panofski #, A. Jankowiak, T. Kamps, Helmholtz-Zentrum Berlin, Berlin, Germany P.N. Lu, J. Teichert, Helmholtz-Zentrum Dresden-Rossendorf,
SRF GUN CHARACTERIZATION - PHASE SPACE AND DARK CURRENT MEASUREMENTS AT ELBE* E. Panofski #, A. Jankowiak, T. Kamps, Helmholtz-Zentrum Berlin, Berlin, Germany P.N. Lu, J. Teichert, Helmholtz-Zentrum Dresden-Rossendorf,
PROGRESS TOWARDS CONSTRUCTION OF A FERMIONIC ATOMIC CLOCK FOR NASA S DEEP SPACE NETWORK
 PROGRESS TOWARDS CONSTRUCTION OF A FERMIONIC ATOMIC CLOCK FOR NASA S DEEP SPACE NETWORK Megan K. Ivory Advisor: Dr. Seth A. Aubin College of William and Mary Atomic clocks are the most accurate time and
PROGRESS TOWARDS CONSTRUCTION OF A FERMIONIC ATOMIC CLOCK FOR NASA S DEEP SPACE NETWORK Megan K. Ivory Advisor: Dr. Seth A. Aubin College of William and Mary Atomic clocks are the most accurate time and
Saturation Absorption Spectroscopy of Rubidium Atom
 Saturation Absorption Spectroscopy of Rubidium Atom Jayash Panigrahi August 17, 2013 Abstract Saturated absorption spectroscopy has various application in laser cooling which have many relevant uses in
Saturation Absorption Spectroscopy of Rubidium Atom Jayash Panigrahi August 17, 2013 Abstract Saturated absorption spectroscopy has various application in laser cooling which have many relevant uses in
Plasma Traps for Space-Charge Studies : Status and Perspectives. Hiromi Okamoto (Hiroshima Univ.)
 Plasma Traps for Space-Charge Studies : Status and Perspectives Hiromi Okamoto (Hiroshima Univ.) Acknowlegements Past and Present Contributors to the S-POD Project Hiroshima University Hiroyuki Higaki,
Plasma Traps for Space-Charge Studies : Status and Perspectives Hiromi Okamoto (Hiroshima Univ.) Acknowlegements Past and Present Contributors to the S-POD Project Hiroshima University Hiroyuki Higaki,
Longitudinal dynamics Yannis PAPAPHILIPPOU CERN
 Longitudinal dynamics Yannis PAPAPHILIPPOU CERN United States Particle Accelerator School, University of California - Santa-Cruz, Santa Rosa, CA 14 th 18 th January 2008 1 Outline Methods of acceleration
Longitudinal dynamics Yannis PAPAPHILIPPOU CERN United States Particle Accelerator School, University of California - Santa-Cruz, Santa Rosa, CA 14 th 18 th January 2008 1 Outline Methods of acceleration
DESIR. P.G. Thirolf Department f. Physik, LMU München. simultaneous relative measurement of individual ions in two precision traps
 MLLTRAP @ DESIR P.G. Thirolf Department f. Physik, LMU München concept of MLLTRAP: measurement with Penning trap of highly-charged ions simultaneous relative measurement of individual ions in two precision
MLLTRAP @ DESIR P.G. Thirolf Department f. Physik, LMU München concept of MLLTRAP: measurement with Penning trap of highly-charged ions simultaneous relative measurement of individual ions in two precision
Secondary Ion Mass Spectrometry (SIMS) Thomas Sky
 1 Secondary Ion Mass Spectrometry (SIMS) Thomas Sky Depth (µm) 2 Characterization of solar cells 0,0 1E16 1E17 1E18 1E19 1E20 0,2 0,4 0,6 0,8 1,0 1,2 P Concentration (cm -3 ) Characterization Optimization
1 Secondary Ion Mass Spectrometry (SIMS) Thomas Sky Depth (µm) 2 Characterization of solar cells 0,0 1E16 1E17 1E18 1E19 1E20 0,2 0,4 0,6 0,8 1,0 1,2 P Concentration (cm -3 ) Characterization Optimization
Application Note GA-301E. MBMS for Preformed Ions. Extrel CMS, 575 Epsilon Drive, Pittsburgh, PA I. SAMPLING A CHEMICAL SOUP
 Application Note MBMS for Preformed Ions, 575 Epsilon Drive, Pittsburgh, PA 15238 (Poster Presented at 45th ASMS Conference on Mass Spectrometry, June 1-5, 1997) In order to accurately characterize a plasma
Application Note MBMS for Preformed Ions, 575 Epsilon Drive, Pittsburgh, PA 15238 (Poster Presented at 45th ASMS Conference on Mass Spectrometry, June 1-5, 1997) In order to accurately characterize a plasma
Introduction to SIMS Basic principles Components Techniques Drawbacks Figures of Merit Variations Resources
 Introduction to SIMS Basic principles Components Techniques Drawbacks Figures of Merit Variations Resources New technique for surface chemical analysis. SIMS examines the mass of ions, instead of energy
Introduction to SIMS Basic principles Components Techniques Drawbacks Figures of Merit Variations Resources New technique for surface chemical analysis. SIMS examines the mass of ions, instead of energy
FOCUS: QUADRUPOLE ION TRAPS ACCOUNT AND PERSPECTIVE
 Matrix Methods for the Calculation of Stability Diagrams in Quadrupole Mass Spectrometry FOCUS: QUADRUPOLE ION TRAPS ACCOUNT AND PERSPECTIVE N. V. Konenkov and M. Sudakov* Department of General Physics,
Matrix Methods for the Calculation of Stability Diagrams in Quadrupole Mass Spectrometry FOCUS: QUADRUPOLE ION TRAPS ACCOUNT AND PERSPECTIVE N. V. Konenkov and M. Sudakov* Department of General Physics,
Fluctuation Suppression during the ECH Induced Potential Formation in the Tandem Mirror GAMMA 10
 EXC/P8-2 Fluctuation Suppression during the ECH Induced Potential Formation in the Tandem Mirror GAMMA M. Yoshikawa ), Y. Miyata ), M. Mizuguchi ), Y. Oono ), F. Yaguchi ), M. Ichimura ), T. Imai ), T.
EXC/P8-2 Fluctuation Suppression during the ECH Induced Potential Formation in the Tandem Mirror GAMMA M. Yoshikawa ), Y. Miyata ), M. Mizuguchi ), Y. Oono ), F. Yaguchi ), M. Ichimura ), T. Imai ), T.
Production of HCI with an electron beam ion trap
 Production of HCI with an electron beam ion trap I=450 ma E= 5 kev axially: electrodes radially: electron beam space charge total trap potential U trap 200 V (U trap ion charge) 10000 ev 15000 A/cm 2 n
Production of HCI with an electron beam ion trap I=450 ma E= 5 kev axially: electrodes radially: electron beam space charge total trap potential U trap 200 V (U trap ion charge) 10000 ev 15000 A/cm 2 n
Quantum Logic Spectroscopy and Precision Measurements
 Quantum Logic Spectroscopy and Precision Measurements Piet O. Schmidt PTB Braunschweig and Leibniz Universität Hannover Bad Honnef, 4. November 2009 Overview What is Quantum Metrology? Quantum Logic with
Quantum Logic Spectroscopy and Precision Measurements Piet O. Schmidt PTB Braunschweig and Leibniz Universität Hannover Bad Honnef, 4. November 2009 Overview What is Quantum Metrology? Quantum Logic with
PIC/MCC Simulation of Radio Frequency Hollow Cathode Discharge in Nitrogen
 PIC/MCC Simulation of Radio Frequency Hollow Cathode Discharge in Nitrogen HAN Qing ( ), WANG Jing ( ), ZHANG Lianzhu ( ) College of Physics Science and Information Engineering, Hebei Normal University,
PIC/MCC Simulation of Radio Frequency Hollow Cathode Discharge in Nitrogen HAN Qing ( ), WANG Jing ( ), ZHANG Lianzhu ( ) College of Physics Science and Information Engineering, Hebei Normal University,
Dense plasma formation on the surface of a ferroelectric cathode
 Vacuum ] (]]]]) ]]] ]]] www.elsevier.com/locate/vacuum Dense plasma formation on the surface of a ferroelectric cathode K. Chirko, Ya.E. Krasik, A. Sayapin, J. Felsteiner Physics Department, Technion Israel
Vacuum ] (]]]]) ]]] ]]] www.elsevier.com/locate/vacuum Dense plasma formation on the surface of a ferroelectric cathode K. Chirko, Ya.E. Krasik, A. Sayapin, J. Felsteiner Physics Department, Technion Israel
arxiv:physics/ v1 3 Aug 2006
 Gamma Ray Spectroscopy with Scintillation Light in Liquid Xenon arxiv:physics/6834 v1 3 Aug 26 K. Ni, E. Aprile, K.L. Giboni, P. Majewski, M. Yamashita Physics Department and Columbia Astrophysics Laboratory
Gamma Ray Spectroscopy with Scintillation Light in Liquid Xenon arxiv:physics/6834 v1 3 Aug 26 K. Ni, E. Aprile, K.L. Giboni, P. Majewski, M. Yamashita Physics Department and Columbia Astrophysics Laboratory
Formation of High-b ECH Plasma and Inward Particle Diffusion in RT-1
 J Fusion Energ (2010) 29:553 557 DOI 10.1007/s10894-010-9327-6 ORIGINAL RESEARCH Formation of High-b ECH Plasma and Inward Particle Diffusion in RT-1 H. Saitoh Z. Yoshida J. Morikawa Y. Yano T. Mizushima
J Fusion Energ (2010) 29:553 557 DOI 10.1007/s10894-010-9327-6 ORIGINAL RESEARCH Formation of High-b ECH Plasma and Inward Particle Diffusion in RT-1 H. Saitoh Z. Yoshida J. Morikawa Y. Yano T. Mizushima
arxiv:quant-ph/ v1 29 Apr 2003
 Atomic Qubit Manipulations with an Electro-Optic Modulator P. J. Lee, B. B. Blinov, K. Brickman, L. Deslauriers, M. J. Madsen, R. arxiv:quant-ph/0304188v1 29 Apr 2003 Miller, D. L. Moehring, D. Stick,
Atomic Qubit Manipulations with an Electro-Optic Modulator P. J. Lee, B. B. Blinov, K. Brickman, L. Deslauriers, M. J. Madsen, R. arxiv:quant-ph/0304188v1 29 Apr 2003 Miller, D. L. Moehring, D. Stick,
hν' Φ e - Gamma spectroscopy - Prelab questions 1. What characteristics distinguish x-rays from gamma rays? Is either more intrinsically dangerous?
 Gamma spectroscopy - Prelab questions 1. What characteristics distinguish x-rays from gamma rays? Is either more intrinsically dangerous? 2. Briefly discuss dead time in a detector. What factors are important
Gamma spectroscopy - Prelab questions 1. What characteristics distinguish x-rays from gamma rays? Is either more intrinsically dangerous? 2. Briefly discuss dead time in a detector. What factors are important
Laser-cooling and trapping (some history) Theory (neutral atoms) Hansch & Schawlow, 1975
 Laser-cooling and trapping (some history) Theory (neutral atoms) Hansch & Schawlow, 1975 Laser-cooling and trapping (some history) Theory (neutral atoms) Hansch & Schawlow, 1975 (trapped ions) Wineland
Laser-cooling and trapping (some history) Theory (neutral atoms) Hansch & Schawlow, 1975 Laser-cooling and trapping (some history) Theory (neutral atoms) Hansch & Schawlow, 1975 (trapped ions) Wineland
MASS ANALYSER. Mass analysers - separate the ions according to their mass-to-charge ratio. sample. Vacuum pumps
 ION ANALYZERS MASS ANALYSER sample Vacuum pumps Mass analysers - separate the ions according to their mass-to-charge ratio MASS ANALYSER Separate the ions according to their mass-to-charge ratio in space
ION ANALYZERS MASS ANALYSER sample Vacuum pumps Mass analysers - separate the ions according to their mass-to-charge ratio MASS ANALYSER Separate the ions according to their mass-to-charge ratio in space
Ion crystallisation. computing
 Ion crystallisation and application to quantum computing Cooling with incrased laser power: (a) reduced Doppler width (b) Kink in the line profile (b) P=0.2 mw P=0.5 mw Excitation spectra of an ion cloud
Ion crystallisation and application to quantum computing Cooling with incrased laser power: (a) reduced Doppler width (b) Kink in the line profile (b) P=0.2 mw P=0.5 mw Excitation spectra of an ion cloud
Quantum Zeno effect
 PHYSICAL REVIEW A VOLUME 41, NUMBER 5 1 MARCH 1990 Quantum Zeno effect Wayne M. Itano, D. J. Heinzen, J. J. Bollinger, and D. J. Wineland Time and Frequency Division, National Institute of Standards and
PHYSICAL REVIEW A VOLUME 41, NUMBER 5 1 MARCH 1990 Quantum Zeno effect Wayne M. Itano, D. J. Heinzen, J. J. Bollinger, and D. J. Wineland Time and Frequency Division, National Institute of Standards and
Mass Selective Ejection by Axial Resonant Excitation from a Linear Ion Trap
 Mass Selective Ejection by Axial Resonant Excitation from a Linear Ion Trap Yuichiro Hashimoto, Hideki Hasegawa, Takashi Baba, and Izumi Waki Central Research Laboratory, Hitachi, Ld., Tokyo, Japan We
Mass Selective Ejection by Axial Resonant Excitation from a Linear Ion Trap Yuichiro Hashimoto, Hideki Hasegawa, Takashi Baba, and Izumi Waki Central Research Laboratory, Hitachi, Ld., Tokyo, Japan We
Building Blocks for Quantum Computing Part IV. Design and Construction of the Trapped Ion Quantum Computer (TIQC)
 Building Blocks for Quantum Computing Part IV Design and Construction of the Trapped Ion Quantum Computer (TIQC) CSC801 Seminar on Quantum Computing Spring 2018 1 Goal Is To Understand The Principles And
Building Blocks for Quantum Computing Part IV Design and Construction of the Trapped Ion Quantum Computer (TIQC) CSC801 Seminar on Quantum Computing Spring 2018 1 Goal Is To Understand The Principles And
Dynamics of Ions in an Electrostatic Ion Beam Trap
 Dynamics of Ions in an Electrostatic Ion Beam Trap Daniel Zajfman Dept. of Particle Physics Weizmann Institute of Science Israel and Max-Planck Institute for Nuclear Physics Heidelberg, Germany Charles
Dynamics of Ions in an Electrostatic Ion Beam Trap Daniel Zajfman Dept. of Particle Physics Weizmann Institute of Science Israel and Max-Planck Institute for Nuclear Physics Heidelberg, Germany Charles
David McIntyre. A slow beam of laser cooled rubidium atoms will be used as the matter-wave source. The atom
 AD-A282 483 R&T 3124128 MATTER-WAVE INTERFEROMETRY WITH LASER COOLED ATOMS David McIntyre Department of Physics, Oregon State University, Corvallis, OR 97331-6507 Grant No: N00014-91-J-1198 DTIC Annual
AD-A282 483 R&T 3124128 MATTER-WAVE INTERFEROMETRY WITH LASER COOLED ATOMS David McIntyre Department of Physics, Oregon State University, Corvallis, OR 97331-6507 Grant No: N00014-91-J-1198 DTIC Annual
Vacuum-Ultraviolet-Excited and CH 2 Cl 2 /H 2 O-Amplified Ionization- Coupled Mass Spectrometry for Oxygenated Organics Analysis
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 Supporting Information for Vacuum-Ultraviolet-Excited and CH 2 Cl 2 /H 2 O-Amplified Ionization- Coupled Mass Spectrometry
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 Supporting Information for Vacuum-Ultraviolet-Excited and CH 2 Cl 2 /H 2 O-Amplified Ionization- Coupled Mass Spectrometry
Association of H + with H 2 at Low Temperatures
 WDS'11 Proceedings of Contributed Papers, Part II, 175 179, 011. ISBN 978--7378-185-9 MATFYZPRESS Association of with at Low Temperatures I. Zymak, P. Jusko, S. Roučka, D. Mulin, R. Plašil, and J. Glosík
WDS'11 Proceedings of Contributed Papers, Part II, 175 179, 011. ISBN 978--7378-185-9 MATFYZPRESS Association of with at Low Temperatures I. Zymak, P. Jusko, S. Roučka, D. Mulin, R. Plašil, and J. Glosík
arxiv: v2 [physics.atom-ph] 10 Sep 2010
![arxiv: v2 [physics.atom-ph] 10 Sep 2010 arxiv: v2 [physics.atom-ph] 10 Sep 2010](/thumbs/93/112763879.jpg) Ion dynamics in a linear radio-frequency trap with a single cooling laser M. Marciante, C. Champenois, A. Calisti, J. Pedregosa-Gutierrez, and M. Knoop arxiv:1004.5202v2 [physics.atom-ph] 10 Sep 2010 Physique
Ion dynamics in a linear radio-frequency trap with a single cooling laser M. Marciante, C. Champenois, A. Calisti, J. Pedregosa-Gutierrez, and M. Knoop arxiv:1004.5202v2 [physics.atom-ph] 10 Sep 2010 Physique
Møller Polarimetry on Atomic Hydrogen
 E.Chudakov June 21, 2011 Møller Polarimetry on Atomic Hydrogen 1 Møller Polarimetry on Atomic Hydrogen E.Chudakov 1 1 JLab Meeting at UVA Outline E.Chudakov June 21, 2011 Møller Polarimetry on Atomic Hydrogen
E.Chudakov June 21, 2011 Møller Polarimetry on Atomic Hydrogen 1 Møller Polarimetry on Atomic Hydrogen E.Chudakov 1 1 JLab Meeting at UVA Outline E.Chudakov June 21, 2011 Møller Polarimetry on Atomic Hydrogen
Instrumental Analysis. Mass Spectrometry. Lecturer:! Somsak Sirichai
 303351 Instrumental Analysis Mass Spectrometry Lecturer:! Somsak Sirichai Mass Spectrometry What is Mass spectrometry (MS)? An analytic method that employs ionization and mass analysis of compounds in
303351 Instrumental Analysis Mass Spectrometry Lecturer:! Somsak Sirichai Mass Spectrometry What is Mass spectrometry (MS)? An analytic method that employs ionization and mass analysis of compounds in
The Aerosol Ion Trap Mass Spectrometer (AIMS): Instrument development and first experimental results
 The Aerosol Ion Trap Mass Spectrometer (AIMS): Instrument development and first experimental results A. Kürten 1, J. Curtius 1 and S. Borrmann 1,2 1 Johannes Gutenberg-University Mainz, Germany 2 Max Planck
The Aerosol Ion Trap Mass Spectrometer (AIMS): Instrument development and first experimental results A. Kürten 1, J. Curtius 1 and S. Borrmann 1,2 1 Johannes Gutenberg-University Mainz, Germany 2 Max Planck
Pulsed Photoacoustic Spectroscopy of I 2 and NO 2
 Int J Thermophys (2012) 33:2055 2061 DOI 10.1007/s10765-012-1323-9 Pulsed Photoacoustic Spectroscopy of I 2 and NO 2 Fahem Yehya Anil K. Chaudhary Received: 30 January 2012 / Accepted: 26 September 2012
Int J Thermophys (2012) 33:2055 2061 DOI 10.1007/s10765-012-1323-9 Pulsed Photoacoustic Spectroscopy of I 2 and NO 2 Fahem Yehya Anil K. Chaudhary Received: 30 January 2012 / Accepted: 26 September 2012
Quantum gates in rare-earth-ion doped crystals
 Quantum gates in rare-earth-ion doped crystals Atia Amari, Brian Julsgaard Stefan Kröll, Lars Rippe Andreas Walther, Yan Ying Knut och Alice Wallenbergs Stiftelse Outline Rare-earth-ion doped crystals
Quantum gates in rare-earth-ion doped crystals Atia Amari, Brian Julsgaard Stefan Kröll, Lars Rippe Andreas Walther, Yan Ying Knut och Alice Wallenbergs Stiftelse Outline Rare-earth-ion doped crystals
High-fidelity simulations of ion trajectories in miniature ion traps using the boundary-element method
 High-fidelity simulations of ion trajectories in miniature ion traps using the boundary-element method Boris Brkić,* Stephen Taylor, Jason F. Ralph, and Neil France Department of Electrical Engineering
High-fidelity simulations of ion trajectories in miniature ion traps using the boundary-element method Boris Brkić,* Stephen Taylor, Jason F. Ralph, and Neil France Department of Electrical Engineering
PROGRESS TOWARDS CONSTRUCTION OF A FERMION ATOMIC CLOCK FOR NASA S DEEP SPACE NETWORK
 PROGRESS TOWARDS CONSTRUCTION OF A FERMION ATOMIC CLOCK FOR NASA S DEEP SPACE NETWORK Megan K. Ivory Advisor: Dr. Seth A. Aubin College of William and Mary Abstract: The most accurate time and frequency
PROGRESS TOWARDS CONSTRUCTION OF A FERMION ATOMIC CLOCK FOR NASA S DEEP SPACE NETWORK Megan K. Ivory Advisor: Dr. Seth A. Aubin College of William and Mary Abstract: The most accurate time and frequency
The World s Smallest Extreme Laboratories:
 The World s Smallest Extreme Laboratories: Probing QED with Highly Charged Ions. Joan Marler Clemson University Outline About HCIs About Ion trapping Precision spectroscopy with HCIs Highly Charged Ions
The World s Smallest Extreme Laboratories: Probing QED with Highly Charged Ions. Joan Marler Clemson University Outline About HCIs About Ion trapping Precision spectroscopy with HCIs Highly Charged Ions
Measurement of the Hyperfine Structure and Isotope Shifts of the 3s 2 3p 2 3 P 2
 Measurement of the Hyperfine Structure and Isotope Shifts of the 3s 2 3p 2 3 P 2 3s3p 3 3 D o 3 Transition in Silicon S. A. Lee * and W. M. Fairbank, Jr. Department of Physics Colorado State University
Measurement of the Hyperfine Structure and Isotope Shifts of the 3s 2 3p 2 3 P 2 3s3p 3 3 D o 3 Transition in Silicon S. A. Lee * and W. M. Fairbank, Jr. Department of Physics Colorado State University
