Advances in Colloid and Interface Science
|
|
|
- Ginger Shelton
- 5 years ago
- Views:
Transcription
1 Advances in Colloid and Interface Science (2009) Contents lists available at ScienceDirect Advances in Colloid and Interface Science journal homepage: Air at hydrophobic surfaces and kinetics of three phase contact formation M. Krasowska a,b, J. Zawala a, K. Malysa a, a Institute of Catalysis and Surface Chemistry Polish Academy of Sciences, ul. Niezapominajek 8, Cracow, Poland b Ian Wark Research Institute, University of South Australia, Mawson Lakes SA 5095, Australia article info abstract Available online 1 November 2008 Keywords: Nanobubbles Air entrapment Hydrophobicity Surface roughness Three phase contact Bubble velocity and collision Wetting film This review focuses on the importance of air presence at hydrophobic solid surfaces for wetting film rupture and kinetics of three phase contact formation. Affinity to air is a typical feature of hydrophobic surfaces, but it has been often either overlooked or not taken into consideration. When the hydrophobic surface, contacted earlier with air, is immersed into water then air can stay attached to the surface. The origin of long range hydrophobic forces and data showing that these interactions were due to the bridging of nanobubbles attached to the hydrophobic surfaces are discussed. A major part of the review is devoted to the description and analysis of data showing that air (nano-, micro-bubbles and/or air film) present at a hydrophobic surface facilitated rupture of the liquid film and three phase contact formation during bubble collisions with flat Teflon plates of different surface roughness. Although all Teflon plates were highly hydrophobic (contact angles ca ) the time of the three phase contact (TPC) formation and attachment of the colliding bubble was strongly affected by the plate surface roughness. The time of the TPC formation was shortened from over 80 down to 2 3 ms when the roughness was increased from below 1 µm to over 50 µm. Higher surface roughness means that larger amounts of air was entrapped during the Teflon plates' immersion in water. Additional experimental evidence is given, showing that facilitation of the TPC formation and the bubble attachment was due to air presence and re-distribution over the Teflon surfaces: i) prolonging the plate immersion time resulted in quicker attachment; ii) irregular and disappearing air pockets were recorded at a Teflon surface; iii) a satellite bubble left at a Teflon surface during the first collision facilitated the attachment; iv) attachment always occurred during the first collision in the case of a very rough Teflon V surface, but in highly concentrated n-octanol and n-heptanol solutions there was bouncing and attachment occurred during the second collision, moreover; v) the degree of bubble kinetic energy transferred into surface energy was significantly smaller during collisions with hydrophobic (Teflon) surfaces than with the hydrophilic ones. The mechanism of air entrapment and redistribution over Teflon plates immersed in water is presented Elsevier B.V. All rights reserved. Contents 1. Introduction Long range interactions between hydrophobic surfaces Nanobubbles at hydrophobic surfaces Stability of nanobubbles Effect of surface roughness from hydrophobic to superhydrophobic surfaces Influence of gas presence on stability of the wetting films Effect of surface roughness on air presence and the TPC formation Air facilitated rupture of the wetting film and attachment of the colliding bubble Prolongation of the TPC formation time in surfactant solutions Air entrapment and redistribution at a hydrophobic surface immersed into an aqueous phase Effect of the bubble impact velocity on the size of the wetting film formed Degree of energy exchange during bubble collision with hydrophilic and hydrophobic surfaces Concluding remarks Corresponding author. address: ncmalysa@cyf-kr.edu.pl (K. Malysa) /$ see front matter 2008 Elsevier B.V. All rights reserved. doi: /j.cis
2 156 M. Krasowska et al. / Advances in Colloid and Interface Science (2009) List of symbols Acknowledgements References Introduction Interactions of gas bubbles with various interfaces and attachment to solid surfaces play an important role in many applications. For example flotation is an industrial process the outcome of which depends on interactions between bubbles and solid particles. The flotation separation is due to differences in wettability of solid surfaces (different degree of hydrophobicity) and stability of the wetting films. In classic flotation process only particles having a hydrophobic surface can easily be attached to the gas bubbles. The interaction between bubble and particle can be divided into three sub-processes: i) collision, ii) attachment and iii) detachment. Attachment is a very fast process and therefore, it has not fully been explored yet. In industrial flotation processes the attachment of mineral grains and formation of a stable bubble-grain aggregate need to occur during the very short collision time. During this time a liquid film, separating the colliding bubble and hydrophobic grain surface, must drain and rupture. Moreover, a hole of the three phase contact, formed as the film ruptures, needs to expand to ensure a high enough magnitude of the surface forces to prevent bubble-particle detachment. Properties of the solid surface (hydrophobic/hydrophilic character, roughness, heterogeneity, surface electric charge, etc.) are of crucial importance for stability and rupture kinetics of the liquid (wetting) film, and so are the compositions of the surrounding continuous phases (liquid and gas). The importance of the solution composition has always been recognized and used to modify the properties of the liquid/solid and liquid/gas interfaces. However, the fact that these processes, studied and carried out in an ambient atmosphere, can be affected by the air presence, have been overlooked till recent years. All experimental studies of colloid chemistry are, as a rule, carried out in a gas atmosphere either in air or an inert gas. Affinity to air is a typical feature of hydrophobic surfaces. Even though this is rather obvious and everyone is aware of it, this fact has very often been neglected and not taken into account in the analysis of experimental results. The most striking illustration of this fact is a concept of socalled long range (up to hundreds nanometers) hydrophobic forces LRHF [1 4] between hydrophobic bodies immersed into aqueous solutions. These long range interactions were first found in Surface Force Apparatus (SFA) experiments and their existence was proved, beyond any doubt, in various laboratories and published in a large number of papers [1 4]. However, as shown recently [5 9], the explanation of the origin of this interaction as a kind of new force was incorrect and originated from not taking into account that the ambient gas atmosphere can be a factor in these interactions. First, it was believed that the long range attraction between hydrophobic bodies immersed into aqueous solutions was caused by structural changes of boundary layers of water. However, to obtain such long range attractions this special structure of water should encompass thousands of molecular layers, which is unrealistic [10]. Later, it was believed the effect was caused by cavitation (separationinduced phase transition). The effect was also attributed to chargefluctuation correlations [11,12], electrostatic origins [13] and eventually to bridging of submicron bubbles [14]. The last mechanism is now rather commonly accepted and it is believed that the measured additional hydrophobic interactions are in reality due to a coalescence of nanobubbles attached to the hydrophobic surfaces immersed into aqueous solution [5 8]. Despite that nanobubbles were observed experimentally by many researchers [5 9] there is still an unsolved dispute whether they can be thermodynamically stable. Since the gas pressure inside such tiny objects should be very high, the question is whether they can really exist and be in equilibrium with the surrounding liquid phase. One explanation of the long lifetime of such nano- or submicro-bubbles refers to the effect of line tension [8] and the necessity of modification of the Young equation. Moreover, physical properties of nanobubbles were reported recently [15] to be comparable to those of macroscopic bubbles, except for the water contact angle values, which were significantly higher than the macroscopic ones. Thus, due to high contact angle the nanobubbles showed flatter shapes resulting in a lower gas pressure inside. Since the nanobubbles presence was detected mainly using tapping mode AFM, which is a quite invasive technique, the question arose whether the nanobubbles could be created (nucleated) by the AFM tip. However, the fact that nanobubbles are more likely to form on a heterogeneous than on a smooth homogeneous hydrophobic surface is a quite convincing indication that it is chemistry and topography of the surface which affect the nanobubble formation and presence. Appearance of the nano- and submicroscopic-bubbles at the surface is strongly related to its hydrophobicity (either natural or due to surfactant adsorption [16] or methylation [8]), morphology (roughness, heterogeneity) [17,18] and moreover, the solution composition for example the solution ph [6,7] and/or level of the liquid phase saturation with gas [9,18 20]). It was also reported recently [21] that air can be entrapped during immersion of hydrophobic solid into water and the submicroscopic bubbles adhering to hydrophobic surfaces can be of great importance in rupture of the wetting films and formation of the three phase contact (TPC) under dynamic conditions. The paper focuses on the importance of gas presence at hydrophobic solid surfaces for the long range interactions and kinetics of the TPC formation. Recent results documenting the existence of nanobubbles and hypotheses about their origin and reasons of stability are reviewed. The importance of gas presence, roughness of the hydrophobic solid surface and kinetic energy of the colliding bubbles for the time-scale of the three phase contact formation and the bubble attachment are described. 2. Long range interactions between hydrophobic surfaces The classical DLVO [22,23] theory states that the force between similar particles immersed in a liquid phase is given by the algebraic sum of the electrical double layer repulsion and the van der Waals attraction. Later on, as reviewed in [24], there were many experimental findings showing that in thin liquid layers the additional surface forces of different origin steric and/or structural forces, called non-dlvo components can be operative under certain conditions. Steric forces acting as a stiffer repellency between the two contacting adsorption layers of polymeric surfactants, called brush-to-brush repellency, were described by de Gennes' scaling theory [25]. Softer steric repulsions can exist at distances longer than the chain length of the polymeric surfactant and they depend on the bulk concentration [24,26]. Structural forces are considered to arise from the confinement of the fluid between two interfaces, which may induce a particular layering of molecules or particles [27 29]. Similarly, the depletion interactions [30,31], observed when nonadsorbing polymers or polyelectrolytes [30] are added to colloidal suspensions, are due to the excluded-volume effects [31] in thin liquid layers. A transition from electrostatic to steric stabilization of foam films was reported to occur under conditions of screening (due to increased ionic strength) of the electrostatic repulsion.
3 M. Krasowska et al. / Advances in Colloid and Interface Science (2009) The development of the surface force apparatus (SFA) had a great impact on studies of surface interactions. The original version invented by Tabor and co-workers [32 34] was meant to investigate van der Waals forces between smooth mica surfaces in air, however, a modified version of the SFA developed by Israelachvili and Adams [35,36] enabled the measurements of forces between mica surfaces immersed into liquid. In many cases, when the surfaces of both bodies were hydrophobic, some additional long range attractive interactions of unknown origin were observed and a concept of so-called longrange hydrophobic forces (LRHF) was formulated [2,4,37]. The origin of this long range attractive interaction was not clear but the concept was rather widely spread. There were a lot of attempts to explain the physical nature of the hydrophobic interactions [38], but none of them succeeded in a consistent explanation of various experimental results. Amongst a number of ideas proposed to explain the origin of the long range interaction forces the following four hypotheses have been most propagated [38]: (i) solvent structuring, (ii) electrostatic correlations, (iii) approach to spinodal separation, and (iv) bridging by nanobubbles. Currently, the explanation that the LRHF were due to the bridging by nanobubbles is gaining ground and the three first hypotheses seem to have only historical meaning. It was firstly postulated [39] that the long range hydrophobic interactions were due to structural changes of the boundary layers of water in vicinity of hydrophobic surfaces. A decreased density of water near a hydrophobic surface (so-called depletion layer) was revealed by X-ray reflectivity measurements of Jensen et al. [40]. A region with density about 10% lower than the bulk water density, having a half-width in order of 1 nm was observed. A similar density reduction in layers of 2 5 nm thick was reported also by Steitz et al. [41]. Although the existence of such a depletion layer next to the hydrophobic surface does not explain the long-range hydrophobic forces observed within distances up to two orders of magnitude greater than the structural changes of the boundary layer, this fact can be closely related to the nanobubbles observed on hydrophobic surfaces. The decrease in density next to a hydrophobic surface may be a result of not only a change in solvent structure but also an increased concentration of these bubbles close to the interface [42]. The second hypothesis assumed that the LRHF existence was related to electrostatic correlations [38], a form of van der Waals force with the electrostatic response induced by the surface with the attraction forces decaying exponentially as a function of half the Debye length [43,44]. It is also worth noting that solids used for the experiments devoted to the measurements of LRHF were usually hydrophobized due to the electrostatically driven adsorption of cationic surfactants. If the hydrophobized surfaces were not completely neutralized afterwards, some electrostatic interactions could be responsible for the attraction [43]. The magnitude of attractive forces between the surfaces modified due to the cationic surfactant adsorption showed the electrolyte dependence. Kekicheff and Spalla [44] determined a decrease in the attraction forces between the glass surfaces (hydrophobized due to adsorption of cationic surfactant CTAB) with increasing the KBr concentration. This effect can be related, however, to an increase in cationic surfactant adsorption at the negatively charged glass surface enhanced by decrease of electric double layer (EDL) thickness (i.e. increasing electrolyte concentration) and therefore a decrease in electrostatic repulsion in between CTAB molecules rather than due to the electrostatic correlation forces of the LRHF [43]. The third hypothesis attributed the LRHF to a separation induced cavitation and density depression in a liquid between the hydrophobic surfaces [45]. Yaminsky et al. [46,47] evaluated theoretically that if the contact angle of a hydrophobic surface exceeds 90, the formation of a water-vapour cavity can become thermodynamically favoured at small separations. Cavitation has been observed experimentally in different systems [48,49]. The LRHF were related to the cavity formation using as evidence the existence of hysteresis in the force measured between hydrophobic surfaces upon approach and retraction. It was assumed that the water cavities were formed between the surfaces brought to the contact and the cavities persisted, bridging the surfaces until they were separated by hundreds of nanometers [48,50]. Long range attraction forces leading to destabilization of the wetting film at hydrophobic solid surfaces have been of special interest for flotation separation, because attachment of hydrophobic particles to the bubble is the fundamental step of the flotation process. The very crucial step in this heterocoagulation process is the thinning and the rupture of the thin liquid (wetting) film between the particle and the bubble [51 54]. In most of the cases, for hydrophilic particles, the van der Waals force is repulsive and it becomes attractive only when the refractive index of the solid is smaller than for water [55].If the electrostatic component is also repulsive then the wetting films should be stable and the attachment of the particle to the bubble surface should not happen. On the other hand it is well known that the liquid film between two hydrophobic surfaces (like for example the hydrophobic grain and the bubble) is in an unstable state [53]. That is why the origin and nature of these attractive forces causing destabilization of the wetting films have been extensively studied. Currently, there is an increasing amount of findings indicating the crucial role of air presence at the hydrophobic surface for the wetting film rupture and the three phase contact formation. 3. Nanobubbles at hydrophobic surfaces The first suggestion that the long-ranged attraction between hydrophobic surfaces is due to bridging of submicroscopic bubbles adhered to the solids was published by Parker et al. [14]. Using the SFA technique they measured the long range attraction forces between fluorosilane treated glass surfaces and noted steps (discontinuities) in the measured forces at separations of the order of nm. They stated that these steps indicated that the long-range attraction were caused by the bridging of submicroscopic cavities or bubbles and presented a theoretical model to fit the force data [14]. First AFM images of nano-bubbles were published in 2000 by Ishida et al. [5] and later on the number of papers documenting existence of nanobubbles at hydrophobic surfaces has been increasing rapidly [7,8,16,19,56 61]. While most reports show isolated bubbles with a spherical appearance [8,15,56,62] the AFM images of Tyrell and Attard [6,57] showed closely-packed irregularly spaced bubbles, similar to a continuous gas film at the interface. The absence or presence of the nanobubbles is currently a topic of considerable interest and there is still a lot of controversy, especially regarding origin and stability of the nanobubbles. Steitz et al. [41] carried out neutron reflectivity experiments on the interface of spin-coated polystyrene (d-ps) film onto silicon blocks immersed into pure D 2 O and concluded that there was a depletion of water in the boundary layer at the hydrophobic surface. According to the authors [41] this depletion layer, the thickness of which was estimated to be 2 5 nm, represented a precursor layer of the submicroscopic gas bubbles. However, later Seo and Satija [63] concluded, on the basis of their neutron reflectivity measurements, that there was no intrinsic depletion layer on a polystyrene film at water and the observed low scattering length density (SLD) layer originated rather from hydrogenated contamination and/or oxidation of the d-ps surface during the sample treatment. Gong et al. [19] using attenuated total internal reflectance Fourier transform infrared spectroscopy (ATR-FTIR) indicated that CO 2 dissolved in solution forms tiny, submicron size bubbles on the surface of the methylated quartz particles. Ishida et al. [5] pointed out the importance of the manner of the hydrophobic surface preparation for the nanobubbles presence. They used silicon wafers, the surfaces of which were hydrophobized with octadecyltrichlorosilane (OTS) using two different procedures. Type-I surface was obtained when the silicon wafer
4 158 M. Krasowska et al. / Advances in Colloid and Interface Science (2009) surface was exposed to air to dry after the hydrophobizing. Type-II surface was prepared by hydrophobizing inside the AFM cell without exposure to air. Nanobubble presence was observed only in the case of the type-i surfaces, i.e. the hydrophobic surfaces which were in contact with air prior to their immersion into water. The authors [5] underlined also that lack of nanobubbles at type-ii surfaces is evidence that the nanobubbles were present on type I surfaces from the beginning. If nanobubbles were generated by the AFM tip contacting the hydrophobic surface then they should be observed both for the type-i and type-ii surfaces. Higashistani and co-workers showed [16,60,61], rather convincingly, that the long-ranged interaction force between hydrophobic surfaces in water is due to the bridging of nanobubbles attached to the interacting surfaces. They stated [60] that when the hydrophobized surfaces were exposed to air then the microscopic bubbles were not removed completely from the surfaces being immersed into water because of high affinity between the bubbles and surfaces. Moreover, it was showed in [16] that air can be introduced into degassed water during dissolution of surfactant powder and the usual degassing procedure (boiling, frizzing in a vessel with liquid nitrogen, evacuating by a vacuum pump, and then melting under reduced pressure) do not guarantee that surfactant solutions are degassed completely. Recently, Meyer et al. [64] studied an influence of dissolved gas on the hydrophobic attraction between surfactant-coated surfaces using the SFA. They reported that removal of dissolved gas reduced the range of the attractive interaction; the short-range attraction (i.e. shorter than 250 Å) remained unchanged. It is worth noting that their de-gassing procedure was rather mild (vacuum pumping, which assured removal of only 90% of dissolved oxygen). Meyer et al. [64] did not find evidence of preexisting nanobubbles either in the case of fully aerated or partially deaerated water. Nevertheless, they concluded [64] that the hydrophobic interaction may be the combination of a truly hydrophobic interaction at short range and a distinctly different interaction at larger separations, which is not in fact related to the hydrophobicity of the surfaces. Thus, it is rather commonly accepted now that the long range additional hydrophobic forces are due to coalescence of the bubbles adhering to hydrophobic surfaces [5,6,57,58,65,66]. The number and size of nanobubbles (usually 30 nm high with curvature radius of the order nm) depends on the solid surface wettability and morphology [8]. Surface roughness is a crucial parameter which can affect the number and size of the bubbles present at a hydrophobic surface [17,18]. Other parameters affecting number, size and distribution of the nanobubbles over the hydrophobic surfaces are the solution composition, ionic strength, ph [6,7] and amount and kind of gases dissolved in aqueous phase [9,18 20]. Pashley et al. [67 69] demonstrated that de-gassed water was a better medium for dispersing hydrophobic particles such as Teflon and oil droplets (in other words hydrophobic dirt ) enhancing in the same way a cleaning process. Rupture of the wetting films at the hydrophobic surfaces can be due to a nucleation of very small (submicroscopic) gas bubbles present there. The gas nucleation can take place at any film thickness because the size of the gas nuclei can vary within a wide range. It is also considered [70 74] that density fluctuation inside the wetting film in vicinity of these nanobubbles can be responsible for the film rupture. The film rupture governed by nucleation mechanism starts from a single hole, probably in the place where the biggest submicroscopic bubbles are located, and leads to a complete dewetting of the whole film area in a very short time [73] Stability of nanobubbles The question of the nanobubble stability is still unresolved and intriguing because the excess (capillary) pressure inside the nanobubbles should be extremely high. For instance in water the capillary pressure for nanobubbles of radius 50 nm is over 28 atm and so they should immediately dissolve into the water. The various proposed and discussed explanations of nanobubble stability seem to point to the differences in nanoscopic and macroscopic contact angle values and to flattening of the nanobubbles. Yang et al. [8] pointed out that the water contact angle of the submicroscopic bubbles on the methylated substrate were much higher than the contact angles measured by sessile drop at a macroscopic scale. This difference between microscopic and macroscopic contact angles was attributed to the influence of the line tension. Therefore, they stated that the Young equation must be modified by the line tension additional term. Thus, the microscopic contact angle θ micro was given as [8]: cosθ micro = cosθ Y τ σ lv R where: θ Y is the Young contact angle, σ lv is the liquid/vapour surface tension, 1/R is the local curvature of the nanobubble base on solid surface and τ is the line tension. Yang et al. [8] evaluated that the line tension value was N and caused stabilization of the submicroscopic bubbles due to their flattering (increase in microscopic water contact angle) and reduction of the Laplace (capillary) pressure. In other words the shape of nanobubbles attached to the surface is not spherical and the transformation into flat segments is the reason of their high stability [10]. Similar conclusions were presented by Zhang et al. [15] who studied physical properties of nanobubbles on hydrophobic surfaces (octadecyltrichlorosilane layer on silicon and highly ordered pyrolytic graphite) in water and surfactant solutions. They found that physical properties of nanobubbles were comparable to those of macroscopic bubbles, besides the contact angle values, which were much larger for nanobubbles. They evaluated [15] from tapping mode AFM measurements that the base radius of nanobubbles was 5 20 times larger than the height, i.e. the nanobubbles shape at hydrophobic surfaces was very flat. According to [15] the nanoscopic contact angles were 168±9 on OTS coated silicon while the macroscopic advancing contact angles of water droplets were only 108±5 really a big difference. Thus, a smaller curvature of the nanobubbles due to high values of the nanoscopic contact angles is currently considered as a main reason of their long life. Presence of gas (nano- and sub-microscopic bubbles) at hydrophobic surfaces can be of significant importance in dewetting, three phase contact formation and stability of various dispersed systems. 4. Effect of surface roughness from hydrophobic to superhydrophobic surfaces Hydrophilicity and hydrophobicity are general terms introduced to describe relative affinity of solids to water spreading on their surfaces. Wettability is due to an unbalance in molecular interactions when at least two materials are brought into contact and the contact angle (θ) is a measure of this unbalance of forces. The contact angle is related to surface energy values of the materials being contacted. It is commonly, but rather arbitrarily assumed that θb90 indicates that the solid surface has some affinity to water and is partially wetted by water. Such surfaces are generally called hydrophilic or weakly hydrophobic, while the surfaces having a contact angle θ 90 are called hydrophobic. A schematic illustration of water spreading and forming drops of various contact angles with variation of the surface hydrophilic/ hydrophobic properties is shown in Fig. 1. Hydrophobic surfaces having a water contact angle above 140 are termed superhydrophobic. Superhydrophobic surfaces can be obtained from hydrophobic ones by appropriate modifications such as roughening, chemical modification, micro patterning, machining, etching or electrospinning [75 80]. ð1þ
5 M. Krasowska et al. / Advances in Colloid and Interface Science (2009) Fig. 1. Water drop shapes and contact angle values at solids of different hydrophobicity. It needs to be mentioned here that when the contact angle is higher than zero then the work of spreading, W S, is negative: W S = σ LV ð1+cosθþ 2σ LV = σ LV ðcosθ 1Þ ð2þ where: σ LV is the liquid/vapor surface tension. A negative value of W S is the necessary condition for the flotation process to occur. Therefore, theoretically at any contact angle higher then zero the flotation of a mineral can proceed [81,82] and therefore in flotation the surfaces having contact angles below 90 are termed as weakly hydrophobic [81], while those of θn90 are called strongly hydrophobic. Recently, it was also shown [83] in laboratory flotation experiments that for a 50% floatability of a mineral in a Hallimond tube the work of spreading should be 20 mj/m 2. It means that for an efficient flotation the contact angle of water has to be at least 48. The Young equation [84] describes the equilibrium in a solid/ liquid/vapor system as the mechanical balance at the line of the three phase contact (TPC) on an ideal (smooth, homogeneous, rigid and insoluble) solid surface (see Fig. 2): σ SV = σ SL + σ LV cosθ where: σ SL is the solid/liquid surface energy, and σ SV is the solid/ vapor surface energy. The fundamental problem associated with the equilibrium contact angle is related to the structure and topography of the solid surface, as real solids are rough. Surface roughness can strongly affect wettability and the measured values of contact angles. To characterize the geometrical non-ideality of a surface, the roughness parameter r was introduced and defined as: r = A real A geometrical where: (A real ) and (A geometrical ) are the real and geometrical areas of the surface. For rn1 the Young Eq. (3) was modified by Wenzel [85] into: cosθ W = r cosθ where θ W is the Wenzel contact angle. The Wenzel equation refers to the so-called homogeneous wetting regime [86,87], i.e. when the ð3þ ð4þ ð5þ liquid completely penetrates scratches, grooves and cavities (Fig. 2). Another situation, termed heterogeneous wetting (see Fig. 2), refers to the cases when air is entrapped inside the grooves underneath the liquid. This is described by the Cassie-Baxter equation [88]: cosθ CB = r f 0 f 0 cosθ + f 0 1 where: θ CS is the Cassie-Baxter contact angle, f is the fraction of the projected area that is wetted by a liquid and r f is the roughness ratio of the wetted area. When f =1 then r f is equal r and Cassie-Baxter Eq. (6) simplifies to the Wenzel Eq. (5). As recently discussed by Extrand [89] and Sedev et al. [90], the phenomenon of higher values of contact angle on modified (roughened, micro patterned, machined or etched [75 77] hydrophobic surfaces is caused by inhibition of the liquid spreading into grooves, scratches and/or cavities on the rough surface. Moreover, a spreading of such drops can be arrested by the edges of the grooves and therefore leads to superhydrophobicity. 5. Influence of gas presence on stability of the wetting films It is well-known that wetting films at hydrophobic solids are not stable. The mechanism governing instability of such films is mainly due to nucleation inside the film [91]. Recently, Stöckelhuber et al. [72 74] have proposed a model which can explain the destabilizing effect of the nanobubble presence. The destabilization is due to density fluctuation inside the wetting film in vicinity of the nanobubbles, which eventually can lead to film rupture. The rupture thickness in such systems can reach very high values, up to hundreds of nanometers, and is strongly scattered since gas nucleation can take place at any film thickness and the size of the gas nuclei can vary within a wide range. Stöckelhuber et al. pointed out [73] that during drainage of the wetting film the apex of the largest nanobubble attached to a hydrophobic solid comes most closely to the solution/gas interface. At this local area a foam film (both solution/gas interfaces) is formed and in the foam films the van der Waals forces are always attractive leading to the film rupture. Moreover, when the three phase contact is formed after the film rupture then the perimeter of the contact formed is equal to the nanobubble base (attachment) ð6þ Fig. 2. Water drop shape and contact angle on smooth and rough hydrophobic surface.
6 160 M. Krasowska et al. / Advances in Colloid and Interface Science (2009) perimeter, i.e. generally it can be larger then a hole formed on the wetted solid surface. A larger perimeter of the TPC formed at the moment of the film rupture means a lower probability of bubble detachment from the hydrophobic surface under dynamic conditions. Surface roughness is a parameter, which can affect the number and size of such bubbles present at a hydrophobic surface [17,18]. Thus, size and number of submicroscopic bubbles attached to hydrophobic surfaces can be tuned by controlling the surface roughness. Rupturing of the wetting film can be considered as the de-wetting process starting from a hole formation and followed by an expansion of the three phase contact till reaching an equilibrium or quasi-equilibrium perimeter. Formation of the primary hole is a rapid process occurring at the film spot of the lowest stability, which can be caused for example by solid surface roughness, non-homogeneity and/or presence of nanobubbles. The formation of primary hole means a rapid change of the wetting film state (equilibrium, quasi-equilibrium and/or non-equilibrium) and therefore is immediately followed by an expansion of the three phase contact till a new state of equilibrium or quasi-equilibrium is attained. The influence of the surface heterogeneity and gas presence on the kinetics of the wetting film rupture and the bubble attachment have been studied in a series of our recent papers [21,92 98]. It was observed there that air presence and roughness of hydrophobic surfaces were the factors of crucial importance for the kinetics of the three phase contact formation by the colliding bubble. 6. Effect of surface roughness on air presence and the TPC formation The influence of gas presence on coagulation of fine coal (a case of natural hydrophobicity) and silica particles was reported by Zhou et al. [99]. A silica surface was hydrophobized by dodecylamine hydrochloride adsorption. They observed different behavior depending whether or not the suspensions were degassed. After degassing, the apparent size of both coal and silica flocs were smaller and this effect was attributed [99] to removal of gas nuclei from the hydrophobic surfaces. Recently, we have observed [21,92 98] that the time of three phase contact formation on Teflon surfaces was strongly affected by air presence. Teflon was used as a model hydrophobic solid (contact angle ) and its surface roughness was varied by mechanical modification (see Table 1). The kinetics of the three phase contact (TPC) formation by the colliding bubble was monitored using a highspeed camera (1182 Hz). Details of the experimental procedures have been given elsewhere [21,92 98]. The roughness of the Teflon plates given in Table 1 was evaluated from optical microscope photos. A collection of the sequences of photos showing the course of the bubble collisions with Teflon plates of different roughness is presented in Table 2. As can be noted the bubbles colliding with Teflon I, Teflon II and Teflon III surfaces did not attach during the first collision, but instead they bounced backwards. In the case of the roughest Teflon IV and Teflon V surfaces the bubble attached during the first collision. Thus, the results presented in Table 2 show the crucial importance of surface roughness for the time-scale of the TPC formation and bubble attachment. With increasing surface roughness Table 1 Teflon plates Plate Surface roughness Teflon I Below 1 µm Teflon II Below 1 µm Teflon IIA µm Teflon III µm Teflon IV µm Teflon V µm the time needed for the TPC formation and bubble attachment was significantly shortened, from over 80 ms to ca. 2.5 ms. As seen in Table 2 the time of the TPC formation was identical for Teflon IV and Teflon V, which indicates that roughness of the order of 50 μm and higher was sufficient to assure the bubble attachment during the first collision. It is quite astonishing that an increase in the Teflon surface roughness from ca. 1 to 80 µm (Table 2) caused such significant reduction in time needed for the TPC formation (from 83.6 to 2.5 ms). Variations of the local bubble velocity during collisions with Teflon plates of different roughness are presented in Fig. 3. As seen the bubble velocity prior to the first collision was identical in all cases (34.8 cm/s). Differentiation in profiles of the bubble local velocity started from the moment of the first collision. That moment was denoted as t= 0 and was always used as the starting point in measurements of the times of the TPC formation and the bubble attachment. The time t=0 ms was chosen [92] as the moment at which the horizontal diameter of the colliding bubble started to increase. Enlargement of the horizontal diameter is an indication that the bubble approach to the solid surface was halted and the kinetic energy of the motion started to be transferred into surface energy of the bubble deformation. The attachment of the colliding bubble to Teflon IV and Teflon V surfaces at time t =2.5 ms, i.e. during the first collision, indicates that this short time period was sufficient for the wetting film drainage, rupture and formation of the TPC perimeter assuring the bubble attachment. The fact that surface roughness has such a crucial effect on the time of the three phase contact formation (i.e. cut the time from over 80 ms down to 2 3 ms) was discussed in [21,92,95] to be related to: i) larger asperities at a rougher surface (i.e. higher probability that the critical thickness of the wetting film is locally attained much faster), and/or ii) higher surface roughness, which enabled larger amounts of air entrapment. It was concluded in [21] that facilitation of the TPC formation and the bubble attachment at rougher Teflon surface was due to larger amount of air present. A careful analysis of the sequence of photos showing the first collision with a Teflon III surface shows that also in this case the wetting film was ruptured during the first collision (see the photos for t=3.4, 4.2 and 5.1 ms in Table 2). However, in the case of the Teflon III surface the perimeter of the TPC created was not sufficiently long to prevent the bubble detaching. It is seen that a neck was formed (photo for t=4.2 ms), which ruptured and a satellite-bubble attached to the Teflon III surface was left by the detaching bubble (photo for t=5.1 ms). It is worth to underline here that there was no visible microbubble attached to the Teflon III surface prior to the first collision. When during the second collision the bubble hit the spot with the satellite bubble left then the stable TPC was formed immediately (within less than 1 ms) because there was already the three phase contact formed by the attached satellite-bubble. There was, most probably, a rupture of the foam film formed between the top pole of the colliding bubble and the bottom pole of the attached satellite-bubble. In the case of collision with the Teflon plates of roughness below 1µm( Teflon I and Teflon II ) the bubble bounced four times with a decreasing velocity (see Fig. 3) due to energy dissipation. During the fifth approach the bubble velocity was reduced to ca. 4.5 cm/s. Such significant lowering in the bubble velocity caused prolongation of the contact time and was advantageous for the wetting film syneresis and the TPC formation. The effect of impact velocity (kinetic energy) of the colliding bubble on kinetics of the TPC formation and bubble attachment is described below Air facilitated rupture of the wetting film and attachment of the colliding bubble For the bubble attachment the wetting film must drain until its critical thickness of rupture is obtained. Then a hole of the TPC must be formed and expanded until the perimeter of the TPC will be large
7 M. Krasowska et al. / Advances in Colloid and Interface Science (2009) Table 2 Sequences of photos showing shape pulsation and bouncing of the bubble colliding with Teflon plates of different roughness in distilled water Bubble equivalent diameter 1.48±0.03 mm.
8 162 M. Krasowska et al. / Advances in Colloid and Interface Science (2009) Table 3 Photos of the bubble attached to Teflon surfaces of different roughness and diameters of the three phase contact (TPC) perimeter Fig. 3. Variations of the bubble local velocity during collisions with Teflon surfaces of different roughness ( Teflon I (black crosses); Teflon II (gray diamonds); Teflon III (white circles); Teflon IV (dark grey triangles); Teflon V (black squares)) and times of the three phase contact formation. enough to prevent the bubble detachment. The diameters of the TPC perimeter formed by the bubbles attached to Teflon surfaces of different roughness are listed in Table 3. As can be noticed the diameters of the TPC perimeter formed were increasing with the surface roughness and the effect is quite significant, from 1.71 to 2.29 mm. Let us discuss reasons for the variations of the attached bubble perimeter size with Teflon surface roughness. It was reported by Ryan and Hemmingsen [17] and by Snoswell et al. [18] that during immersion of dry hydrophobic surfaces into solution some microscopic air bubbles can get entrapped in the grooves, scratches and gaps. Results of our studies reported in [21,94 98] also show that presence of micro-bubbles at the hydrophobic solid surface can be one of the most important parameters facilitating attachment of the colliding bubble. Higher surface roughness means bigger cavities and scratches and therefore more gas can be entrapped during immersion of such surfaces into an aqueous phase. This is confirmed by the data presented in Table 3. As in all cases the size of the colliding bubble was identical (d b =1.48 mm), the larger diameter of the attached bubble perimeter indicates that there was more air at rougher Teflon surfaces. The largest diameters of the attached bubbles were at the roughest surfaces and there was also the quickest TPC formation and bubble attachment. This is an indication that this facilitation of the TPC formation was due to larger volumes of the gas entrapped in larger cavities of the rough surface. Fig. 4 presents further experimental data on the importance of air presence and its redistribution over the hydrophobic surface for the kinetics of bubble attachment. The collision numbers are presented during which the bubble attachment to the Teflon III surface occurred as a function of the immersion time into distilled water. The immersion time was defined [92] as the time elapsed from the plate immersion into water till the bubble first collided with the Teflon surface. As seen in Fig. 4 for immersion times shorter than 10 s the bubble attachment occurred either during the third or fourth collision only. At longer immersion times (20 s and longer) the attachment was quicker, even during the first collision. A longer immersion time means that there was a longer time available for redistribution of the air entrapped during the Teflon plate immersion into water. A quicker attachment of the colliding bubble indicates that there were more three phase contact spots (nano- and/or micro-bubbles) due to the entrapped air redistribution over the Teflon surface. Microscope photos presented in Fig. 5 illustrate that the entrapped air can undergo redistribution over the Teflon surface. The photos show the Teflon V surface immersed into water and irregular, white Fig. 4. Collision number of the bubble colliding with Teflon III surface, during which the attachment occurred, as a function of the time of the plate immersion into distilled water (redrawn from Ref. [96]).
9 M. Krasowska et al. / Advances in Colloid and Interface Science (2009) Fig. 5. Microscope photographs of the Teflon V surface immersed into water. Photographs A and B show the same area at different times from the plate immersion into water (redrawn from Ref. [21]). shapes (air pockets) can be easily spotted there. Photos A and B show the same area of the Teflon surface at different times after the plate immersion into water. As seen the shapes of these white spots are different, i.e. they were evolving with time (photo B was taken after longer immersion time). Thus, these images document that indeed, air entrapped during the Teflon plate immersion can undergo redistribution over the hydrophobic surface Prolongation of the TPC formation time in surfactant solutions Studies on the kinetics of the three phase contact formation in n-hexanol and n-octanol solutions supplied additional evidence on the importance of air presence at hydrophobic surfaces [21,94 96].As described above (see Table 2 and Fig. 3), in the case of bubble collisions with the roughest Teflon surface ( Teflon V ) in distilled water, the TPC was formed within a time shorter than 3 ms. It needs to be underlined here that in this case the reproducibility was excellent and the bubble was always attached during the first collision. In surfactant solutions the terminal velocity of the rising bubbles is lowered due to retardation of the gas/liquid interface mobility by the adsorbed layer. Depending on solution concentration (adsorption coverage) it can be lowered even over twofold [93]. A smaller terminal velocity means a lower kinetic energy of the colliding bubble and a longer time of contact with solid surface. The bubble terminal velocity decreases rapidly at low surfactant concentration, but it is almost constant starting from a given concentration (adsorption coverage). At this characteristic adsorption coverage the bubble interface is fully immobilized and therefore the bubble velocity decreases no further [93]. Thus, one would expect that in surfactant solutions there should always be attachment during the first bubble collision with a rough Teflon surface. Indeed, this was observed at low concentration of n-hexanol [94] and n-octanol solutions [21], but quite a different situation was found at high solution concentrations. This is illustrated in Figs. 6 and 7. Fig. 6 presents sequences of photos showing bubble collisions with the Teflon V plate in n-heptanol solutions of concentration of and M. At lower n-heptanol concentration the TPC was formed at t=2.5 ms during the first collision (see top part of Fig. 6), i.e. similarly as in distilled water. However, at higher n-heptanol concentration ( M) there was no immediate TPC formation and bubble attachment during the first collision. The bubble bounced a bit and the time of the three phase contact formation was prolonged till ca. 19 ms, i.e. was over 6 times longer than in distilled water, despite the bubble impact velocity being lower. Fig. 7 presents a similar effect observed [21] in n-octanol solutions. This shows the bubble velocity variations during collisions with a Teflon V surface in distilled water and n-octanol solutions of concentrations and M. As can be seen in Fig. 7 the bubble impact velocity during the first collision was identical at both n-octanol concentrations and significantly lower than in distilled water. However, again the time of the TPC formation and the bubble attachment was prolonged to ca. 18 ms at higher n-octanol concentrations. The fact that the TPC was formed and the bubble was always attached to the Teflon V surface during the first collision in distilled water shows that the wetting film was unstable and ruptured very quickly. A similar low stability of the intervening liquid was observed in n-heptanol and n-octanol solutions of low concentration. Fig. 6. Sequences of the photos (time interval=0.845 ms) documenting bouncing and attachment of the bubble to the Teflon V surface in M and M n-heptanol solutions.
10 164 M. Krasowska et al. / Advances in Colloid and Interface Science (2009) of the wetting (non-symmetric) film [21]. It is well known that stability of symmetric foam films increases with surfactant concentration. Thus, prolongation of the time of the TPC formation at high n- heptanol and n-octanol concentrations is strong evidence that indeed there was air adhering to hydrophobic Teflon surfaces immersed into an aqueous phase Air entrapment and redistribution at a hydrophobic surface immersed into an aqueous phase Fig. 7. Variations of the bubble local velocity with time during collision with the Teflon V plate in distilled water (gray diamonds), M n-octanol (black triangles), and M n-octanol (white circles) solutions (redrawn from Ref. [21]). However, at high n-hexanol and n-octanol concentrations the time of the TPC formation was prolonged, which indicates the increased stability of the liquid film separating the colliding bubble from the rough Teflon V surface. As discussed above the increased amount of air (nano- and/or sub-microscopic bubbles) adhering to the rougher Teflon surface can be the most important factor responsible for facilitating the TPC formation and the bubble attachment. It also means that the intervening film rupture would happen as a result of bridging between the colliding bubble and nano- or sub-microscopic bubbles already attached to the hydrophobic surface. Thus, a foam (symmetric) film would locally be formed between bubbles instead As discussed above, when hydrophobic surfaces being in contact with air are immersed into an aqueous phase then air can be entrapped, due to its high affinity to the hydrophobic surfaces. Data presented in Figs. 4 7 illustrate the importance of the air presence and redistribution at a hydrophobic surface on the kinetics of attachment of the colliding bubble. The possible mechanism of the air entrapment and redistribution over the surface of the Teflon plate being immersed in a water phase [21] is showed schematically in Fig. 8. When the Teflon plate is being immersed into water then air is entrapped inside its surface cavities (pores, holes, scratches) and amounts of the air entrapped are increasing with surface roughness. It can be assumed that at the moment of the plate immersion and its positioning horizontally there was approximately a planar interface between water and air entrapped inside the cavities. However, this interface shape is definitely a non-equilibrium state (parts A and B of Fig. 8). The equilibrium state of the interface between water and air pockets inside the Teflon cavities is determined by the Young-Laplace equation: Δp =2ðσ lv =R cav Þcosθ ð7þ where R cav is the radius of the cavity (pore), and Δ p is the pressure difference. Since the equilibrium advancing contact advancing angle Fig. 8. Mechanism of air entrapment and redistribution during immersion of the Teflon plate (redrawn from Ref. [21]).
11 M. Krasowska et al. / Advances in Colloid and Interface Science (2009) values for the Teflon plates were above 100, therefore cosθb0 and Δpb0 (part C of Fig. 8). Thus, at equilibrium there should be a convex meniscus inside the cavities and the excessive amounts of air entrapped had to be transferred elsewhere. As the aqueous phase was already air saturated, so these excessive amounts of air could not be dissolved so a spreading over the Teflon surface was the most probable option. The air transferred from the cavities to the surface of neighboring asperities and/or pillars formed either a film or submicroscopic bubbles (part D of Fig. 8). When the bubble collided with a Teflon surface having air films and/or sub-microscopic bubbles at its pillars (asperities), i.e. at the outmost elements of the surface, then the intervening liquid film was ruptured at these local areas. Thus, there was bridging (coalescence) of the colliding bubble (macroscopic) with a number of sub-microscopic bubbles (already being in contact with Teflon surface) or de-wetted areas (air films at Teflon asperities) [21]. As a result of the gas presence at the outmost elements of the Teflon plate a long enough perimeter (a large area) of the three phase contact was obtained immediately during the collision. Due to the long perimeter of the three phase contact the surface forces were high enough to prevent the bubble detachment. The bubble backward motion was arrested and the bubble stayed attached to the Teflon surface. 7. Effect of the bubble impact velocity on the size of the wetting film formed The kinetics of liquid film drainage depends strongly on the film size. As shown by Zawala et al. [100] the size of the liquid films formed by the bubbles colliding with liquid/solid and liquid/air interfaces depended on the bubble impact velocity (kinetic energy). The bubble kinetic energy at the moment of collision was varied in two ways: (i) the diameter of the bubble was varied, and (ii) the distance (L) between a point of the bubble detachment and the interface location was varied (the bubble was at the acceleration stage). The bubble kinetic energy, E k, at the moment of the collision was calculated [100] as: E k =0:5C m ρ l V b U 2 b where: V b is the bubble volume, ρ l is the liquid density, U b is a bubble impact velocity and C m is the added mass coefficient. The added mass is an additional force related to the deformation of the bubble geometry. The added mass coefficient is a function of the bubbledeformation ratio and it can be expressed [101,102] as: C m = α ð9þ 2 α where! α = 2χ2 χ p ffiffiffiffiffiffiffiffiffiffiffi cos 1 1 χ 2 1 χ ð8þ ð10þ and χ is the ratio of the bubble horizontal (d h ) and vertical (d v ) diameters. After linearization the C m value is given [102] as: C m =0:62χ 0:12: ð11þ For non-deformed bubbles the added mass coefficient calculated on the basis of Eq. (11) is equal 0.5, i.e., as the C m values reported in the literature for spherical particles and bubbles [ ]. Fig. 9 presents a comparison of velocity variations during the bubble (R b =0.74) collisions with the water/teflon interface located at the distances L=1.8, 3.6 and 300 mm above the capillary. In these experiments [100] the Teflon plate of surface roughness ca µm was used and it will be called further Teflon IIA. As can be noted in Fig. 9, prior to the first collision at location L=300 mm, the bubble approached the water/ Teflon IIA interface with the constant terminal velocity (34.7 cm/s). For the interface locations at L=1.8 Fig. 9. Velocity variations of the bubble colliding with water/ Teflon IIA interface located at different distances form the bubble formation point [redrawn from Ref. [100]). and L=3.6 mm the bubble impact velocities were significantly smaller, because the bubble was still at the acceleration stage. The different location of the interface corresponded to different collision kinetic energies, namely: , , J for L=300, 3.6 and 1.8 mm, respectively, and it had a significant influence on the time of the three-phase contact formation, t TPC. The time of the TPC formation was increasing with the bubble kinetic energy, from t TPC =35 to 84 ms when the bubble kinetic energy increased from to J. This prolongation of the time of the TPC formation was due to the larger size of the intervening films formed during the bubble collision with the Teflon surface [100]. Higher kinetic energy means larger deformation of the colliding bubble shape and, as a consequence, a larger radius of the wetting film formed. To confirm the effect of the film size on time of the TPC formation a series of experiments were carried out, in which bubbles of different radius (0.61, 0.74 and 0.88 mm) were used [100]. The Teflon II plate was located always at distance L = 1.8 mm. The bubble impact velocities were ca. 8 cm/s, but there were small differences in the bubble kinetic energy due to increasing bubble size. To eliminate the influence of these differences in kinetic energy the time of the TPC formation was measured from the moment when the bubble stayed practically motionless beneath the Teflon IIA plate [100]. Fig. 10 presents three sets of photos; i) at the moment of the first collision, ii) after the energy dissipation, i.e. when the bubbles stays captured beneath the Teflon IIA plate, and iii) after the TPC formation. The time intervals from the moment when the bubble stayed motionless (Fig. 10B) to the moment of the TPC formation (Fig. 10C) was measured and interpreted [100] as the time available for the wetting film drainage to its critical thickness of rupture (t drain ). These t drain values were equal to 29.4±8.7, 41±7 and 57.7±12.6 ms for the bubbles of radius R b =0.61, 0.74, 0.88 mm, respectively. Thus, the film drainage time was increasing with the bubble size. The radius of the wetting film formed can be calculated [105] as: R 2 F = F br b 2πσ LV ð12þ where F b is the buoyancy force. Taking into account that for water σ LV =72.4 mn/m, the radii of the wetting films formed (R F ) were 0.11,
12 166 M. Krasowska et al. / Advances in Colloid and Interface Science (2009) Fig. 10. Collisions and attachment of the bubbles of different radii to Teflon II surface. (A) - first collision (B) - after dissipation of the kinetic energy, (C) - after the TPC formation. Fig. 11. Thickness of the wetting film of different radii as a function of drainage time. Points show the experimental values of the film drainage time and 0.23 in the case of the bubbles radii 0.61, 0.74, 0.88 mm, respectively. To evaluate the rupture thicknesses of the wetting films formed at Teflon IIA surface the Scheludko equation [106] was used: d 1=h2 = 4 dt 3 n Δp ηr 2 F ð13þ where n is the factor for interfacial mobility of the film (n=4 when the film is created between the solid wall with no-slip condition and a completely mobile liquid/gas interface), η is dynamic viscosity and h is the film thickness. After integration and rearrangement the Scheludko equation can be rewritten as: 1 h 1 2 h 2 0 = 16 3 Δp t ηr 2 F ð14þ liquid film formed, and (ii) the increase of the free energy of the system resulting from the bubble's surface area increase due to the shape deformation. The bubble rebounds if the thinning liquid layer did not reach a critical rupture thickness before the entire bubble kinetic energy is transferred into surface energy of the system. The amplitude of the bubble bouncing is decreasing with every collision [94,110], due to energy dissipation. Comparison of the velocity variations during the bubble (R b =0.74 mm) collisions with hydrophobic ( Teflon IIA ) and hydrophilic (mica) surfaces is presented in Fig. 12. The velocity variations were similar and three distinct bounces were recorded (see Fig. 12). Photos inserted in Fig. 12 show that at a hydrophobic Teflon surface the TPC was formed after the bouncing was damped, while at a hydrophilic mica surface the wetting film was stable and there was no TPC formation. Analysis of the degree of kinetic energy exchange into surface energy of the deformed bubble published in [100] supplied data indicating the presence of air at hydrophobic solid surfaces. An where h 0 is an initial film thickness. Variations of the wetting film thickness as a function of the syneresis time are presented in Fig. 11 for the R F =0.61, 0.74 and 0.88 mm. The thicknesses of the liquid films calculated on the basis of experimentally determined t drain values are presented there as well. It can be observed in Fig. 11, that the film with smaller radius needs less time to drain to the given thickness. Moreover, the experimentally determined values of t drain correspond to the rupture thickness of 0.854, 1.03 and 1.22 µm. Thus, the rupture thicknesses of the larger films were higher even though the drainage time was longer. It shows that the probability of rupture of the film having a larger radius was higher, due to, most probably, larger thickness fluctuations and non-homogeneities [107,108], especially at the liquid/solid interface. 8. Degree of energy exchange during bubble collision with hydrophilic and hydrophobic surfaces Bouncing of the bubble colliding with various interfaces is a consequence of competition of two processes [109]: (i) thinning of the Fig. 12. Velocity variations of the bubble colliding with water/ Teflon IIA and water/ mica interfaces.
Forces across thin liquid film
 Forces across thin liquid film Effect of surface charge and hydrophobicity on wetting film stability Marta Krasowska, Daniel Fornasiero, John Ralston Bubble-particle interactions 1. Collision rce o f e
Forces across thin liquid film Effect of surface charge and hydrophobicity on wetting film stability Marta Krasowska, Daniel Fornasiero, John Ralston Bubble-particle interactions 1. Collision rce o f e
Lecture 7 Contact angle phenomena and wetting
 Lecture 7 Contact angle phenomena and Contact angle phenomena and wetting Young s equation Drop on the surface complete spreading Establishing finite contact angle γ cosθ = γ γ L S SL γ S γ > 0 partial
Lecture 7 Contact angle phenomena and Contact angle phenomena and wetting Young s equation Drop on the surface complete spreading Establishing finite contact angle γ cosθ = γ γ L S SL γ S γ > 0 partial
Solid-liquid interface
 Lecture Note #9 (Spring, 2017) Solid-liquid interface Reading: Shaw, ch. 6 Contact angles and wetting Wetting: the displacement from a surface of one fluid by another. A gas is displaced by a liquid at
Lecture Note #9 (Spring, 2017) Solid-liquid interface Reading: Shaw, ch. 6 Contact angles and wetting Wetting: the displacement from a surface of one fluid by another. A gas is displaced by a liquid at
DLVO interaction between the spheres
 DLVO interaction between the spheres DL-interaction energy for two spheres: D w ( x) 64c π ktrϕ e λ DL 2 x λ 2 0 0 D DLVO interaction w ( x) 64πkTRϕ e λ DLVO AR /12x 2 x λd 2 0 D Lecture 11 Contact angle
DLVO interaction between the spheres DL-interaction energy for two spheres: D w ( x) 64c π ktrϕ e λ DL 2 x λ 2 0 0 D DLVO interaction w ( x) 64πkTRϕ e λ DLVO AR /12x 2 x λd 2 0 D Lecture 11 Contact angle
Microfluidics 2 Surface tension, contact angle, capillary flow
 MT-0.6081 Microfluidics and BioMEMS Microfluidics 2 Surface tension, contact angle, capillary flow 28.1.2017 Ville Jokinen Surface tension & Surface energy Work required to create new surface = surface
MT-0.6081 Microfluidics and BioMEMS Microfluidics 2 Surface tension, contact angle, capillary flow 28.1.2017 Ville Jokinen Surface tension & Surface energy Work required to create new surface = surface
Contents. Preface XI Symbols and Abbreviations XIII. 1 Introduction 1
 V Contents Preface XI Symbols and Abbreviations XIII 1 Introduction 1 2 Van der Waals Forces 5 2.1 Van der Waals Forces Between Molecules 5 2.1.1 Coulomb Interaction 5 2.1.2 Monopole Dipole Interaction
V Contents Preface XI Symbols and Abbreviations XIII 1 Introduction 1 2 Van der Waals Forces 5 2.1 Van der Waals Forces Between Molecules 5 2.1.1 Coulomb Interaction 5 2.1.2 Monopole Dipole Interaction
*blood and bones contain colloids. *milk is a good example of a colloidal dispersion.
 Chap. 3. Colloids 3.1. Introduction - Simple definition of a colloid: a macroscopically heterogeneous system where one component has dimensions in between molecules and macroscopic particles like sand
Chap. 3. Colloids 3.1. Introduction - Simple definition of a colloid: a macroscopically heterogeneous system where one component has dimensions in between molecules and macroscopic particles like sand
Interfacial forces and friction on the nanometer scale: A tutorial
 Interfacial forces and friction on the nanometer scale: A tutorial M. Ruths Department of Chemistry University of Massachusetts Lowell Presented at the Nanotribology Tutorial/Panel Session, STLE/ASME International
Interfacial forces and friction on the nanometer scale: A tutorial M. Ruths Department of Chemistry University of Massachusetts Lowell Presented at the Nanotribology Tutorial/Panel Session, STLE/ASME International
Colloidal Suspension Rheology Chapter 1 Study Questions
 Colloidal Suspension Rheology Chapter 1 Study Questions 1. What forces act on a single colloidal particle suspended in a flowing fluid? Discuss the dependence of these forces on particle radius. 2. What
Colloidal Suspension Rheology Chapter 1 Study Questions 1. What forces act on a single colloidal particle suspended in a flowing fluid? Discuss the dependence of these forces on particle radius. 2. What
Praktikum zur. Materialanalytik
 Praktikum zur Materialanalytik Functionalized Surfaces B510 Stand: 20.10.2017 Table of contents Introduction 2 Basics 2 Surface tension 2 From wettability to the contact angle 4 The Young equation 5 Wetting
Praktikum zur Materialanalytik Functionalized Surfaces B510 Stand: 20.10.2017 Table of contents Introduction 2 Basics 2 Surface tension 2 From wettability to the contact angle 4 The Young equation 5 Wetting
Physics and Chemistry of Interfaces
 Hans Jürgen Butt, Karlheinz Graf, and Michael Kappl Physics and Chemistry of Interfaces Second, Revised and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Contents Preface XI 1 Introduction
Hans Jürgen Butt, Karlheinz Graf, and Michael Kappl Physics and Chemistry of Interfaces Second, Revised and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Contents Preface XI 1 Introduction
Probing the Hydrophobic Interaction between Air Bubbles and Partially. Hydrophobic Surfaces Using Atomic Force Microscopy
 Supporting Information for Probing the Hydrophobic Interaction between Air Bubbles and Partially Hydrophobic Surfaces Using Atomic Force Microscopy Chen Shi, 1 Derek Y.C. Chan, 2.3 Qingxia Liu, 1 Hongbo
Supporting Information for Probing the Hydrophobic Interaction between Air Bubbles and Partially Hydrophobic Surfaces Using Atomic Force Microscopy Chen Shi, 1 Derek Y.C. Chan, 2.3 Qingxia Liu, 1 Hongbo
Surface and Interfacial Tensions. Lecture 1
 Surface and Interfacial Tensions Lecture 1 Surface tension is a pull Surfaces and Interfaces 1 Thermodynamics for Interfacial Systems Work must be done to increase surface area just as work must be done
Surface and Interfacial Tensions Lecture 1 Surface tension is a pull Surfaces and Interfaces 1 Thermodynamics for Interfacial Systems Work must be done to increase surface area just as work must be done
emulsions, and foams March 21 22, 2009
 Wetting and adhesion Dispersions in liquids: suspensions, emulsions, and foams ACS National Meeting March 21 22, 2009 Salt Lake City Ian Morrison 2009 Ian Morrison 2009 Lecure 2 - Wetting and adhesion
Wetting and adhesion Dispersions in liquids: suspensions, emulsions, and foams ACS National Meeting March 21 22, 2009 Salt Lake City Ian Morrison 2009 Ian Morrison 2009 Lecure 2 - Wetting and adhesion
Specific ion effects on the interaction of. hydrophobic and hydrophilic self assembled
 Supporting Information Specific ion effects on the interaction of hydrophobic and hydrophilic self assembled monolayers T. Rios-Carvajal*, N. R. Pedersen, N. Bovet, S.L.S. Stipp, T. Hassenkam. Nano-Science
Supporting Information Specific ion effects on the interaction of hydrophobic and hydrophilic self assembled monolayers T. Rios-Carvajal*, N. R. Pedersen, N. Bovet, S.L.S. Stipp, T. Hassenkam. Nano-Science
Supplementary Information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supplementary Information Visualization of equilibrium position of colloidal particles at fluid-water
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supplementary Information Visualization of equilibrium position of colloidal particles at fluid-water
Contents. Preface XIII
 V Contents Preface XIII 1 General Introduction 1 1.1 Fundamental Knowledge Required for Successful Dispersion of Powders into Liquids 1 1.1.1 Wetting of Powder into Liquid 1 1.1.2 Breaking of Aggregates
V Contents Preface XIII 1 General Introduction 1 1.1 Fundamental Knowledge Required for Successful Dispersion of Powders into Liquids 1 1.1.1 Wetting of Powder into Liquid 1 1.1.2 Breaking of Aggregates
The Origins of Surface and Interfacial Tension
 The Origins of Surface and Interfacial Tension Imbalance of intermolecular forces exists at the liquid-air interface γ la= the surface tension that exists at the liquid-air interface Suppose we have a
The Origins of Surface and Interfacial Tension Imbalance of intermolecular forces exists at the liquid-air interface γ la= the surface tension that exists at the liquid-air interface Suppose we have a
DLVO Theory and Non-DLVO Forces
 NPTEL Chemical Engineering Interfacial Engineering Module 3: Lecture 5 DLVO Theory and Non-DLVO Forces Dr. Pallab Ghosh Associate Professor Department of Chemical Engineering IIT Guwahati, Guwahati 781039
NPTEL Chemical Engineering Interfacial Engineering Module 3: Lecture 5 DLVO Theory and Non-DLVO Forces Dr. Pallab Ghosh Associate Professor Department of Chemical Engineering IIT Guwahati, Guwahati 781039
Capillarity and Wetting Phenomena
 ? Pierre-Gilles de Gennes Frangoise Brochard-Wyart David Quere Capillarity and Wetting Phenomena Drops, Bubbles, Pearls, Waves Translated by Axel Reisinger With 177 Figures Springer Springer New York Berlin
? Pierre-Gilles de Gennes Frangoise Brochard-Wyart David Quere Capillarity and Wetting Phenomena Drops, Bubbles, Pearls, Waves Translated by Axel Reisinger With 177 Figures Springer Springer New York Berlin
Surface forces action in a vicinity of three phase contact line and other current problems in kinetics of wetting and spreading
 Loughborough University Institutional Repository Surface forces action in a vicinity of three phase contact line and other current problems in kinetics of wetting and spreading This item was submitted
Loughborough University Institutional Repository Surface forces action in a vicinity of three phase contact line and other current problems in kinetics of wetting and spreading This item was submitted
Applied Surfactants: Principles and Applications
 Applied Surfactants: Principles and Applications Tadros, Tharwat F. ISBN-13: 9783527306299 Table of Contents Preface. 1 Introduction. 1.1 General Classification of Surface Active Agents. 1.2 Anionic Surfactants.
Applied Surfactants: Principles and Applications Tadros, Tharwat F. ISBN-13: 9783527306299 Table of Contents Preface. 1 Introduction. 1.1 General Classification of Surface Active Agents. 1.2 Anionic Surfactants.
Module17: Intermolecular Force between Surfaces and Particles. Lecture 23: Intermolecular Force between Surfaces and Particles
 Module17: Intermolecular Force between Surfaces and Particles Lecture 23: Intermolecular Force between Surfaces and Particles 1 We now try to understand the nature of spontaneous instability in a confined
Module17: Intermolecular Force between Surfaces and Particles Lecture 23: Intermolecular Force between Surfaces and Particles 1 We now try to understand the nature of spontaneous instability in a confined
Colloidal Particles at Liquid Interfaces: An Introduction
 1 Colloidal Particles at Liquid Interfaces: An Introduction Bernard P. Binks and Tommy S. Horozov Surfactant and Colloid Group, Department of Chemistry, University of Hull, Hull, HU6 7RX, UK 1.1 Some Basic
1 Colloidal Particles at Liquid Interfaces: An Introduction Bernard P. Binks and Tommy S. Horozov Surfactant and Colloid Group, Department of Chemistry, University of Hull, Hull, HU6 7RX, UK 1.1 Some Basic
Abstract. The principles and applicability of surface structure and hydrophobicity of polymers (PS, PDMS),
 Contact Angle Goniometer: Hydrophobicity of Biomaterial Surfaces and Protein Coatings Eman Mousa Alhajji North Carolina State University Department of Materials Science and Engineering MSE 255 Lab Report
Contact Angle Goniometer: Hydrophobicity of Biomaterial Surfaces and Protein Coatings Eman Mousa Alhajji North Carolina State University Department of Materials Science and Engineering MSE 255 Lab Report
Reaction at the Interfaces
 Reaction at the Interfaces Lecture 1 On the course Physics and Chemistry of Interfaces by HansJürgen Butt, Karlheinz Graf, and Michael Kappl Wiley VCH; 2nd edition (2006) http://homes.nano.aau.dk/lg/surface2009.htm
Reaction at the Interfaces Lecture 1 On the course Physics and Chemistry of Interfaces by HansJürgen Butt, Karlheinz Graf, and Michael Kappl Wiley VCH; 2nd edition (2006) http://homes.nano.aau.dk/lg/surface2009.htm
Supplementary Information
 Supplementary Information Under-water Superoleophobic Glass: Unexplored role of the surfactant-rich solvent Authors: Prashant R. Waghmare, Siddhartha Das, Sushanta K. Mitra * Affiliation: Micro and Nano-scale
Supplementary Information Under-water Superoleophobic Glass: Unexplored role of the surfactant-rich solvent Authors: Prashant R. Waghmare, Siddhartha Das, Sushanta K. Mitra * Affiliation: Micro and Nano-scale
Lecture 12: Biomaterials Characterization in Aqueous Environments
 3.051J/20.340J 1 Lecture 12: Biomaterials Characterization in Aqueous Environments High vacuum techniques are important tools for characterizing surface composition, but do not yield information on surface
3.051J/20.340J 1 Lecture 12: Biomaterials Characterization in Aqueous Environments High vacuum techniques are important tools for characterizing surface composition, but do not yield information on surface
Foundations of. Colloid Science SECOND EDITION. Robert J. Hunter. School of Chemistry University of Sydney OXPORD UNIVERSITY PRESS
 Foundations of Colloid Science SECOND EDITION Robert J. Hunter School of Chemistry University of Sydney OXPORD UNIVERSITY PRESS CONTENTS 1 NATURE OF COLLOIDAL DISPERSIONS 1.1 Introduction 1 1.2 Technological
Foundations of Colloid Science SECOND EDITION Robert J. Hunter School of Chemistry University of Sydney OXPORD UNIVERSITY PRESS CONTENTS 1 NATURE OF COLLOIDAL DISPERSIONS 1.1 Introduction 1 1.2 Technological
INTERMOLECULAR AND SURFACE FORCES
 INTERMOLECULAR AND SURFACE FORCES SECOND EDITION JACOB N. ISRAELACHVILI Department of Chemical & Nuclear Engineering and Materials Department University of California, Santa Barbara California, USA ACADEMIC
INTERMOLECULAR AND SURFACE FORCES SECOND EDITION JACOB N. ISRAELACHVILI Department of Chemical & Nuclear Engineering and Materials Department University of California, Santa Barbara California, USA ACADEMIC
Thermodynamically Stable Emulsions Using Janus Dumbbells as Colloid Surfactants
 Thermodynamically Stable Emulsions Using Janus Dumbbells as Colloid Surfactants Fuquan Tu, Bum Jun Park and Daeyeon Lee*. Description of the term notionally swollen droplets When particles are adsorbed
Thermodynamically Stable Emulsions Using Janus Dumbbells as Colloid Surfactants Fuquan Tu, Bum Jun Park and Daeyeon Lee*. Description of the term notionally swollen droplets When particles are adsorbed
Module 8: "Stability of Colloids" Lecture 37: "" The Lecture Contains: DLVO Theory. Effect of Concentration. Objectives_template
 The Lecture Contains: DLVO Theory Effect of Concentration file:///e /courses/colloid_interface_science/lecture37/37_1.htm[6/16/2012 1:02:12 PM] Studying the stability of colloids is an important topic
The Lecture Contains: DLVO Theory Effect of Concentration file:///e /courses/colloid_interface_science/lecture37/37_1.htm[6/16/2012 1:02:12 PM] Studying the stability of colloids is an important topic
Experimental Classification of Matter
 Experimental Classification of Matter Sample Heterogeneous? Heterogeneous Can sample be separated by physical means? Homogeneous Substance (pure) Can sample be separated by chemical means? Element Does
Experimental Classification of Matter Sample Heterogeneous? Heterogeneous Can sample be separated by physical means? Homogeneous Substance (pure) Can sample be separated by chemical means? Element Does
Supplementary table I. Table of contact angles of the different solutions on the surfaces used here. Supplementary Notes
 1 Supplementary Figure 1. Sketch of the experimental setup (not to scale) : it consists of a thin mylar sheet (0, 02 4 3cm 3 ) held fixed vertically. The spacing y 0 between the glass plate and the upper
1 Supplementary Figure 1. Sketch of the experimental setup (not to scale) : it consists of a thin mylar sheet (0, 02 4 3cm 3 ) held fixed vertically. The spacing y 0 between the glass plate and the upper
Basic Laboratory. Materials Science and Engineering. Atomic Force Microscopy (AFM)
 Basic Laboratory Materials Science and Engineering Atomic Force Microscopy (AFM) M108 Stand: 20.10.2015 Aim: Presentation of an application of the AFM for studying surface morphology. Inhalt 1.Introduction...
Basic Laboratory Materials Science and Engineering Atomic Force Microscopy (AFM) M108 Stand: 20.10.2015 Aim: Presentation of an application of the AFM for studying surface morphology. Inhalt 1.Introduction...
Mohamed Daoud Claudine E.Williams Editors. Soft Matter Physics. With 177 Figures, 16 of them in colour
 Mohamed Daoud Claudine E.Williams Editors Soft Matter Physics With 177 Figures, 16 of them in colour Contents 1. Droplets: CapiUarity and Wetting 1 By F. Brochard-Wyart (With 35 figures) 1.1 Introduction
Mohamed Daoud Claudine E.Williams Editors Soft Matter Physics With 177 Figures, 16 of them in colour Contents 1. Droplets: CapiUarity and Wetting 1 By F. Brochard-Wyart (With 35 figures) 1.1 Introduction
Adsorption Processes. Ali Ahmadpour Chemical Eng. Dept. Ferdowsi University of Mashhad
 Adsorption Processes Ali Ahmadpour Chemical Eng. Dept. Ferdowsi University of Mashhad Contents Introduction Principles of adsorption Types of adsorption Definitions Brief history Adsorption isotherms Mechanism
Adsorption Processes Ali Ahmadpour Chemical Eng. Dept. Ferdowsi University of Mashhad Contents Introduction Principles of adsorption Types of adsorption Definitions Brief history Adsorption isotherms Mechanism
Review. Surfaces of Biomaterials. Characterization. Surface sensitivity
 Surfaces of Biomaterials Three lectures: 1.23.05 Surface Properties of Biomaterials 1.25.05 Surface Characterization 1.27.05 Surface and Protein Interactions Review Bulk Materials are described by: Chemical
Surfaces of Biomaterials Three lectures: 1.23.05 Surface Properties of Biomaterials 1.25.05 Surface Characterization 1.27.05 Surface and Protein Interactions Review Bulk Materials are described by: Chemical
Colloid Science Principles, methods and applications
 Colloid Science Principles, methods and applications Second Edition Edited by TERENCE COSGROVE School of Chemistry, University of Bristol, Bristol, UK WILEY A John Wiley and Sons, Ltd, Publication Contents
Colloid Science Principles, methods and applications Second Edition Edited by TERENCE COSGROVE School of Chemistry, University of Bristol, Bristol, UK WILEY A John Wiley and Sons, Ltd, Publication Contents
Instability & Patterning of Thin Polymer Films Prof. R. Mukherjee Department of Chemical Engineering Indian Institute Of Technology, Kharagpur
 Instability & Patterning of Thin Polymer Films Prof. R. Mukherjee Department of Chemical Engineering Indian Institute Of Technology, Kharagpur Lecture No. # 33 Spontaneous Instability & Dewetting of Thin
Instability & Patterning of Thin Polymer Films Prof. R. Mukherjee Department of Chemical Engineering Indian Institute Of Technology, Kharagpur Lecture No. # 33 Spontaneous Instability & Dewetting of Thin
Intermolecular and Surface Forces
 Intermolecular and Surface Forces ThirH FHitinn '' I I 111 \J& LM* КтЛ I Km I W I 1 Jacob N. Israelachvili UNIVERSITY OF CALIFORNIA SANTA BARBARA, CALIFORNIA, USA AMSTERDAM BOSTON HEIDELBERG LONDON NEW
Intermolecular and Surface Forces ThirH FHitinn '' I I 111 \J& LM* КтЛ I Km I W I 1 Jacob N. Israelachvili UNIVERSITY OF CALIFORNIA SANTA BARBARA, CALIFORNIA, USA AMSTERDAM BOSTON HEIDELBERG LONDON NEW
8.2 Surface phenomenon of liquid. Out-class reading: Levine p Curved interfaces
 Out-class reading: Levine p. 387-390 13.2 Curved interfaces https://news.cnblogs.com/n/559867/ 8.2.1 Some interesting phenomena 8.2.1 Some interesting phenomena Provided by Prof. Yu-Peng GUO of Jilin
Out-class reading: Levine p. 387-390 13.2 Curved interfaces https://news.cnblogs.com/n/559867/ 8.2.1 Some interesting phenomena 8.2.1 Some interesting phenomena Provided by Prof. Yu-Peng GUO of Jilin
Ultrafast water harvesting and transport in hierarchical microchannels
 SUPPLEMENTARY INFORMATION Articles https://doi.org/10.1038/s41563-018-0171-9 In the format provided by the authors and unedited. Ultrafast water harvesting and transport in hierarchical microchannels Huawei
SUPPLEMENTARY INFORMATION Articles https://doi.org/10.1038/s41563-018-0171-9 In the format provided by the authors and unedited. Ultrafast water harvesting and transport in hierarchical microchannels Huawei
The Wilhelmy balance. How can we measure surface tension? Surface tension, contact angles and wettability. Measuring surface tension.
 ow can we measure surface tension? Surface tension, contact angles and wettability www.wikihow.com/measure-surface-tension Measuring surface tension The Wilhelmy balance F Some methods: Wilhelmy plate
ow can we measure surface tension? Surface tension, contact angles and wettability www.wikihow.com/measure-surface-tension Measuring surface tension The Wilhelmy balance F Some methods: Wilhelmy plate
Biomaterial Scaffolds
 Biomaterial Scaffolds Biomaterial Properties Surface properties Bulk properties Biological properties Types of Biomaterials Biological materials Synthetic materials Surface Properties The body reads the
Biomaterial Scaffolds Biomaterial Properties Surface properties Bulk properties Biological properties Types of Biomaterials Biological materials Synthetic materials Surface Properties The body reads the
Surface interactions part 1: Van der Waals Forces
 CHEM-E150 Interfacial Phenomena in Biobased Systems Surface interactions part 1: Van der Waals Forces Monika Österberg Spring 018 Content Colloidal stability van der Waals Forces Surface Forces and their
CHEM-E150 Interfacial Phenomena in Biobased Systems Surface interactions part 1: Van der Waals Forces Monika Österberg Spring 018 Content Colloidal stability van der Waals Forces Surface Forces and their
Chapter 7. Pickering Stabilisation ABSTRACT
 Chapter 7 Pickering Stabilisation ABSTRACT In this chapter we investigate the interfacial properties of Pickering emulsions. Based upon findings that indicate these emulsions to be thermodynamically stable,
Chapter 7 Pickering Stabilisation ABSTRACT In this chapter we investigate the interfacial properties of Pickering emulsions. Based upon findings that indicate these emulsions to be thermodynamically stable,
Jacco Snoeijer PHYSICS OF FLUIDS
 Jacco Snoeijer PHYSICS OF FLUIDS dynamics dynamics freezing dynamics freezing microscopics of capillarity Menu 1. surface tension: thermodynamics & microscopics 2. wetting (statics): thermodynamics & microscopics
Jacco Snoeijer PHYSICS OF FLUIDS dynamics dynamics freezing dynamics freezing microscopics of capillarity Menu 1. surface tension: thermodynamics & microscopics 2. wetting (statics): thermodynamics & microscopics
Interfaces and interfacial energy
 Interfaces and interfacial energy 1/14 kinds: l/g }{{ l/l } mobile s/g s/l s/s Example. Estimate the percetage of water molecules on the surface of a fog droplet of diameter (i) 0.1 mm (naked eye visibility
Interfaces and interfacial energy 1/14 kinds: l/g }{{ l/l } mobile s/g s/l s/s Example. Estimate the percetage of water molecules on the surface of a fog droplet of diameter (i) 0.1 mm (naked eye visibility
Supplementary Figure 1 Surface tension of polyelectrolyte solutions. Experimentally measured values of surface tension of the solutions that were
 Supplementary Figure 1 Surface tension of polyelectrolyte solutions. Experimentally measured values of surface tension of the solutions that were used in experiments throughout the paper. All surface tensions
Supplementary Figure 1 Surface tension of polyelectrolyte solutions. Experimentally measured values of surface tension of the solutions that were used in experiments throughout the paper. All surface tensions
HYDROPHOBIC FORCES IN FLOTATION. Rajesh Pazhianur ABSTRACT
 HYDROPHOBIC FORCES IN FLOTATION Rajesh Pazhianur ABSTRACT An atomic force microscope (AFM) has been used to conduct force measurements to better understand the role of hydrophobic forces in flotation.
HYDROPHOBIC FORCES IN FLOTATION Rajesh Pazhianur ABSTRACT An atomic force microscope (AFM) has been used to conduct force measurements to better understand the role of hydrophobic forces in flotation.
Chapter 11 Properties of Solutions
 Chapter 11 Properties of Solutions Solutions Homogeneous mixtures of two or more substances Composition is uniform throughout the sample No chemical reaction between the components of the mixture Solvents
Chapter 11 Properties of Solutions Solutions Homogeneous mixtures of two or more substances Composition is uniform throughout the sample No chemical reaction between the components of the mixture Solvents
The Effect of Water and Confinement on Self-Assembly of
 Supporting Information: The Effect of Water and Confinement on Self-Assembly of Imidazolium Based Ionic Liquids at Mica Interface H.-W. Cheng, J.-N. Dienemann, P. Stock, C. Merola, Y.-J. Chen and M. Valtiner*
Supporting Information: The Effect of Water and Confinement on Self-Assembly of Imidazolium Based Ionic Liquids at Mica Interface H.-W. Cheng, J.-N. Dienemann, P. Stock, C. Merola, Y.-J. Chen and M. Valtiner*
Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces.
 Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák & István Bányai, University of Debrecen Dept of Colloid and Environmental Chemistry http://kolloid.unideb.hu/~kolloid/
Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák & István Bányai, University of Debrecen Dept of Colloid and Environmental Chemistry http://kolloid.unideb.hu/~kolloid/
Chapter 9 Generation of (Nano)Particles by Growth
 Chapter 9 Generation of (Nano)Particles by Growth 9.1 Nucleation (1) Supersaturation Thermodynamics assumes a phase change takes place when there reaches Saturation of vapor in a gas, Saturation of solute
Chapter 9 Generation of (Nano)Particles by Growth 9.1 Nucleation (1) Supersaturation Thermodynamics assumes a phase change takes place when there reaches Saturation of vapor in a gas, Saturation of solute
Particle Characterization Laboratories, Inc.
 Analytical services Particle size analysis Dynamic Light Scattering Static Light Scattering Sedimentation Diffraction Zeta Potential Analysis Single Point Titration Isoelectric point determination Aqueous
Analytical services Particle size analysis Dynamic Light Scattering Static Light Scattering Sedimentation Diffraction Zeta Potential Analysis Single Point Titration Isoelectric point determination Aqueous
SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES
 30 SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES * Gas molecules are small compared to the space between them. * Gas molecules move in straight lines
30 SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES * Gas molecules are small compared to the space between them. * Gas molecules move in straight lines
Chapter 7 Solid Surface
 Chapter 7 Solid Surface Definition of solid : A matter that is rigid and resists stress. Difference between solid and liquid surface : Liquid : always in equilibrium and equipotential. (Fig 7.1a,b) Solid
Chapter 7 Solid Surface Definition of solid : A matter that is rigid and resists stress. Difference between solid and liquid surface : Liquid : always in equilibrium and equipotential. (Fig 7.1a,b) Solid
Molecular Driving Forces
 Molecular Driving Forces Statistical Thermodynamics in Chemistry and Biology SUBGfittingen 7 At 216 513 073 / / Ken A. Dill Sarina Bromberg With the assistance of Dirk Stigter on the Electrostatics chapters
Molecular Driving Forces Statistical Thermodynamics in Chemistry and Biology SUBGfittingen 7 At 216 513 073 / / Ken A. Dill Sarina Bromberg With the assistance of Dirk Stigter on the Electrostatics chapters
Surface Tension and its measurements
 Surface Tension and its measurements Surface Tension Surface tension is a fundamental property by which the gas liquid interfaces are characterized. The zone between a gaseous phase and a liquid phase
Surface Tension and its measurements Surface Tension Surface tension is a fundamental property by which the gas liquid interfaces are characterized. The zone between a gaseous phase and a liquid phase
Chemical Potential. Combining the First and Second Laws for a closed system, Considering (extensive properties)
 Chemical Potential Combining the First and Second Laws for a closed system, Considering (extensive properties) du = TdS pdv Hence For an open system, that is, one that can gain or lose mass, U will also
Chemical Potential Combining the First and Second Laws for a closed system, Considering (extensive properties) du = TdS pdv Hence For an open system, that is, one that can gain or lose mass, U will also
Instabilities in Thin Polymer Films: From Pattern Formation to Rupture
 Instabilities in Thin Polymer Films: From Pattern Formation to Rupture John R. Dutcher*, Kari Dalnoki-Veress Η, Bernie G. Nickel and Connie B. Roth Department of Physics, University of Guelph, Guelph,
Instabilities in Thin Polymer Films: From Pattern Formation to Rupture John R. Dutcher*, Kari Dalnoki-Veress Η, Bernie G. Nickel and Connie B. Roth Department of Physics, University of Guelph, Guelph,
Physical Chemistry of Polymers (4)
 Physical Chemistry of Polymers (4) Dr. Z. Maghsoud CONCENTRATED SOLUTIONS, PHASE SEPARATION BEHAVIOR, AND DIFFUSION A wide range of modern research as well as a variety of engineering applications exist
Physical Chemistry of Polymers (4) Dr. Z. Maghsoud CONCENTRATED SOLUTIONS, PHASE SEPARATION BEHAVIOR, AND DIFFUSION A wide range of modern research as well as a variety of engineering applications exist
Capillarity. ESS5855 Lecture Fall 2010
 Capillarity ESS5855 Lecture Fall 2010 Capillarity: the tendency of a liquid in a narrow tube or pore to rise or fall as a result of surface tension (The concise Oxford Dictionary) Surface tension: the
Capillarity ESS5855 Lecture Fall 2010 Capillarity: the tendency of a liquid in a narrow tube or pore to rise or fall as a result of surface tension (The concise Oxford Dictionary) Surface tension: the
Instability and Patterning of Thin Polymer Films Prof. R. Mukherjee Department Of Chemical Engineering Indian Institute Of Technology, Kharagpur
 Instability and Patterning of Thin Polymer Films Prof. R. Mukherjee Department Of Chemical Engineering Indian Institute Of Technology, Kharagpur Lecture No. # 36 Spontaneous Instability & Dewetting of
Instability and Patterning of Thin Polymer Films Prof. R. Mukherjee Department Of Chemical Engineering Indian Institute Of Technology, Kharagpur Lecture No. # 36 Spontaneous Instability & Dewetting of
Fabrication of ordered array at a nanoscopic level: context
 Fabrication of ordered array at a nanoscopic level: context Top-down method Bottom-up method Classical lithography techniques Fast processes Size limitations it ti E-beam techniques Small sizes Slow processes
Fabrication of ordered array at a nanoscopic level: context Top-down method Bottom-up method Classical lithography techniques Fast processes Size limitations it ti E-beam techniques Small sizes Slow processes
Colloid & Interface Science Case Study Model Answers
 Colloid & Interface Science Case Study Model Answers Distance Learning Course in Cosmetic Science Society of Cosmetic Scientists Common Features Formulations were examples of lyophobic colloidal systems
Colloid & Interface Science Case Study Model Answers Distance Learning Course in Cosmetic Science Society of Cosmetic Scientists Common Features Formulations were examples of lyophobic colloidal systems
SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES
 30 SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES * Gas molecules are small compared to the space between them. * Gas molecules move in straight lines
30 SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES * Gas molecules are small compared to the space between them. * Gas molecules move in straight lines
TECHNICAL NOTES 9 FLOTATION
 TECHNICAL NOTES 9 FLOTATION Flotation is the most widely used mineral separation method. It is the preferred method of mineral recovery for many of the most important minerals that are recovered and large
TECHNICAL NOTES 9 FLOTATION Flotation is the most widely used mineral separation method. It is the preferred method of mineral recovery for many of the most important minerals that are recovered and large
Wettability of CaCO 3 surfaces
 Colloids and Surfaces A: Physicochemical and Engineering Aspects 157 (1999) 333 340 www.elsevier.nl/locate/colsurfa Wettability of CaCO 3 surfaces Malvina G. Orkoula a,b, Petros G. Koutsoukos a,b, *, Michel
Colloids and Surfaces A: Physicochemical and Engineering Aspects 157 (1999) 333 340 www.elsevier.nl/locate/colsurfa Wettability of CaCO 3 surfaces Malvina G. Orkoula a,b, Petros G. Koutsoukos a,b, *, Michel
SYNTHESIS OF INORGANIC MATERIALS AND NANOMATERIALS. Pr. Charles Kappenstein LACCO, Laboratoire de Catalyse en Chimie Organique, Poitiers, France
 SYNTHESIS OF INORGANIC MATERIALS AND NANOMATERIALS Pr. Charles Kappenstein LACCO, Laboratoire de Catalyse en Chimie Organique, Poitiers, France Outline IV - FORMATION OF SOLIDS FROM SOLUTIONS 1) Glass
SYNTHESIS OF INORGANIC MATERIALS AND NANOMATERIALS Pr. Charles Kappenstein LACCO, Laboratoire de Catalyse en Chimie Organique, Poitiers, France Outline IV - FORMATION OF SOLIDS FROM SOLUTIONS 1) Glass
Monolayers. Factors affecting the adsorption from solution. Adsorption of amphiphilic molecules on solid support
 Monolayers Adsorption as process Adsorption of gases on solids Adsorption of solutions on solids Factors affecting the adsorption from solution Adsorption of amphiphilic molecules on solid support Adsorption
Monolayers Adsorption as process Adsorption of gases on solids Adsorption of solutions on solids Factors affecting the adsorption from solution Adsorption of amphiphilic molecules on solid support Adsorption
Miami Dade College CHM Second Semester General Chemistry
 Miami Dade College CHM 1046 - Second Semester General Chemistry Course Description: CHM 1046 is the second semester of a two-semester general chemistry course for science, premedical science and engineering
Miami Dade College CHM 1046 - Second Semester General Chemistry Course Description: CHM 1046 is the second semester of a two-semester general chemistry course for science, premedical science and engineering
Chapter 10: Liquids, Solids, and Phase Changes
 Chapter 10: Liquids, Solids, and Phase Changes In-chapter exercises: 10.1 10.6, 10.11; End-of-chapter Problems: 10.26, 10.31, 10.32, 10.33, 10.34, 10.35, 10.36, 10.39, 10.40, 10.42, 10.44, 10.45, 10.66,
Chapter 10: Liquids, Solids, and Phase Changes In-chapter exercises: 10.1 10.6, 10.11; End-of-chapter Problems: 10.26, 10.31, 10.32, 10.33, 10.34, 10.35, 10.36, 10.39, 10.40, 10.42, 10.44, 10.45, 10.66,
Wettability of carbonate reservoir minerals under carbon storage conditions
 TCCS-9 Wettability of carbonate reservoir minerals under carbon storage conditions Dr Mihaela Stevar and Prof Martin Trusler 13 June 2017 1 Outline Background Objectives http://www.hydrobead.com/ Experimental
TCCS-9 Wettability of carbonate reservoir minerals under carbon storage conditions Dr Mihaela Stevar and Prof Martin Trusler 13 June 2017 1 Outline Background Objectives http://www.hydrobead.com/ Experimental
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa.
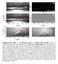 Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
Film Formation from Industrial Waterborne Latices
 Film Formation from Industrial Waterborne Latices I. Ludwig*, W. Schabel*, J.-C. Castaing**, P. Ferlin**, M. Kind* *Universität Karlsruhe (TH), Germany **Rhodia Recherches, Aubervilliers, France Abstract
Film Formation from Industrial Waterborne Latices I. Ludwig*, W. Schabel*, J.-C. Castaing**, P. Ferlin**, M. Kind* *Universität Karlsruhe (TH), Germany **Rhodia Recherches, Aubervilliers, France Abstract
CHEMICAL KINETICS (RATES OF REACTION)
 Kinetics F322 1 CHEMICAL KINETICS (RATES OF REACTION) Introduction Chemical kinetics is concerned with the dynamics of chemical reactions such as the way reactions take place and the rate (speed) of the
Kinetics F322 1 CHEMICAL KINETICS (RATES OF REACTION) Introduction Chemical kinetics is concerned with the dynamics of chemical reactions such as the way reactions take place and the rate (speed) of the
General Chemistry A
 General Chemistry 1140 - A May 5, 2005 (6 Pages, 48 Questions) ame 1. Which of the following properties is a general characteristic of solids? (A) Solids have a rigid shape and fixed volume (B) Solids
General Chemistry 1140 - A May 5, 2005 (6 Pages, 48 Questions) ame 1. Which of the following properties is a general characteristic of solids? (A) Solids have a rigid shape and fixed volume (B) Solids
8.2 Surface phenomena of liquid. Out-class reading: Levine p Curved interfaces
 Out-class reading: Levine p. 387-390 13.2 Curved interfaces 8.2.1 Some interesting phenomena Evolution of bubbles on porous surface. 8.2.1 Some interesting phenomena Addition of a seed in Supersaturated
Out-class reading: Levine p. 387-390 13.2 Curved interfaces 8.2.1 Some interesting phenomena Evolution of bubbles on porous surface. 8.2.1 Some interesting phenomena Addition of a seed in Supersaturated
Rationale: Phase diagrams are standard in all high school chemistry textbooks and therefore are considered prior knowledge.
 Big Idea 2: Chemical and physical properties of materials can be explained by the structure and the arrangement of atoms, ions, or molecules and the forces between them. Material Covered (Y or N) and Location
Big Idea 2: Chemical and physical properties of materials can be explained by the structure and the arrangement of atoms, ions, or molecules and the forces between them. Material Covered (Y or N) and Location
Module 5: "Adsoption" Lecture 25: The Lecture Contains: Definition. Applications. How does Adsorption occur? Physisorption Chemisorption.
 The Lecture Contains: Definition Applications How does Adsorption occur? Physisorption Chemisorption Energetics Adsorption Isotherms Different Adsorption Isotherms Langmuir Adsorption Isotherm file:///e
The Lecture Contains: Definition Applications How does Adsorption occur? Physisorption Chemisorption Energetics Adsorption Isotherms Different Adsorption Isotherms Langmuir Adsorption Isotherm file:///e
- As for the liquids, the properties of different solids often differ considerably. Compare a sample of candle wax to a sample of quartz.
 32 SOLIDS * Molecules are usually packed closer together in the solid phase than in the gas or liquid phases. * Molecules are not free to move around each other as in the liquid phase. Molecular/atomic
32 SOLIDS * Molecules are usually packed closer together in the solid phase than in the gas or liquid phases. * Molecules are not free to move around each other as in the liquid phase. Molecular/atomic
Sanitary Engineering. Coagulation and Flocculation. Week 3
 Sanitary Engineering Coagulation and Flocculation Week 3 1 Coagulation and Flocculation Colloidal particles are too small to be removed by sedimentation or by sand filtration processes. Coagulation: Destabilization
Sanitary Engineering Coagulation and Flocculation Week 3 1 Coagulation and Flocculation Colloidal particles are too small to be removed by sedimentation or by sand filtration processes. Coagulation: Destabilization
Supplementary Information on Thermally Enhanced Self-Propelled Droplet Motion on Gradient Surfaces
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supplementary Information on Thermally Enhanced Self-Propelled Droplet Motion on Gradient Surfaces
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supplementary Information on Thermally Enhanced Self-Propelled Droplet Motion on Gradient Surfaces
Surface Forces & Liquid Films (Answers to Exercise Problems)
 //5 Surface Forces & Liquid Films (nswers to Exercise Problems) Wuge H. Briscoe wuge.briscoe@bris.ac.uk URL: wugebrisco7.wix.com/briscoegroup Exercise : van der Waals forces & liquid films When octane
//5 Surface Forces & Liquid Films (nswers to Exercise Problems) Wuge H. Briscoe wuge.briscoe@bris.ac.uk URL: wugebrisco7.wix.com/briscoegroup Exercise : van der Waals forces & liquid films When octane
Chapter 11. Intermolecular forces. Chapter 11 1
 Chapter 11 Intermolecular Attractions and the Properties of Liquids and Solids 1 2 Intermolecular forces Forces of attraction between molecules Directly dependent on the distance between the molecules
Chapter 11 Intermolecular Attractions and the Properties of Liquids and Solids 1 2 Intermolecular forces Forces of attraction between molecules Directly dependent on the distance between the molecules
Chapter Intermolecular attractions
 Chapter 11 11.2 Intermolecular attractions Intermolecular Attractions and the Properties of Liquids and Solids Intermolecular forces control the physical properties of the substance. Intramolecular forces
Chapter 11 11.2 Intermolecular attractions Intermolecular Attractions and the Properties of Liquids and Solids Intermolecular forces control the physical properties of the substance. Intramolecular forces
Solvation, Structural and Hydration Forces. Chapter 13
 Solvation, Structural and Hydration Forces Chapter 13 Non-DLVO Forces When two surfaces or particles approach closer than a few nanometres, continuum theories of van der Waals and double layer forces often
Solvation, Structural and Hydration Forces Chapter 13 Non-DLVO Forces When two surfaces or particles approach closer than a few nanometres, continuum theories of van der Waals and double layer forces often
Supporting Information
 Electronic Supplementary Material (ESI) for Biomaterials Science. This journal is The Royal Society of Chemistry 2014 Supporting Information Biomimetic Honeycomb-patterned Surface as the Tunable Cell Adhesion
Electronic Supplementary Material (ESI) for Biomaterials Science. This journal is The Royal Society of Chemistry 2014 Supporting Information Biomimetic Honeycomb-patterned Surface as the Tunable Cell Adhesion
Module 3: "Thin Film Hydrodynamics" Lecture 12: "" The Lecture Contains: Linear Stability Analysis. Some well known instabilities. Objectives_template
 The Lecture Contains: Linear Stability Analysis Some well known instabilities file:///e /courses/colloid_interface_science/lecture12/12_1.htm[6/16/2012 1:39:16 PM] Linear Stability Analysis This analysis
The Lecture Contains: Linear Stability Analysis Some well known instabilities file:///e /courses/colloid_interface_science/lecture12/12_1.htm[6/16/2012 1:39:16 PM] Linear Stability Analysis This analysis
SOLUTIONS TO CHAPTER 5: COLLOIDS AND FINE PARTICLES
 SOLUTIONS TO CHAPTER 5: COLLOIDS AND FINE PARTICLES EXERCISE 5.1: Colloidal particles may be either dispersed or aggregated. (a) What causes the difference between these two cases? Answer in terms of interparticle
SOLUTIONS TO CHAPTER 5: COLLOIDS AND FINE PARTICLES EXERCISE 5.1: Colloidal particles may be either dispersed or aggregated. (a) What causes the difference between these two cases? Answer in terms of interparticle
Solution Formation. Copyright Houghton Mifflin Company.All rights reserved. Presentation of Lecture Outlines, 12 2
 Solutions Solution Formation A solution is a homogeneous mixture of two or more substances, consisting of ions or molecules. (See Animation: Solution Equilibrium). A colloid, although it also appears to
Solutions Solution Formation A solution is a homogeneous mixture of two or more substances, consisting of ions or molecules. (See Animation: Solution Equilibrium). A colloid, although it also appears to
CHEMISTRY PHYSICAL. of FOODS INTRODUCTION TO THE. CRC Press. Translated by Jonathan Rhoades. Taylor & Francis Croup
 Christos Ritzoulis Translated by Jonathan Rhoades INTRODUCTION TO THE PHYSICAL CHEMISTRY of FOODS CRC Press Taylor & Francis Croup Boca Raton London NewYork CRC Press is an imprint of the Taylor & Francis
Christos Ritzoulis Translated by Jonathan Rhoades INTRODUCTION TO THE PHYSICAL CHEMISTRY of FOODS CRC Press Taylor & Francis Croup Boca Raton London NewYork CRC Press is an imprint of the Taylor & Francis
CHAPTER 13. States of Matter. Kinetic = motion. Polar vs. Nonpolar. Gases. Hon Chem 13.notebook
 CHAPTER 13 States of Matter States that the tiny particles in all forms of matter are in constant motion. Kinetic = motion A gas is composed of particles, usually molecules or atoms, with negligible volume
CHAPTER 13 States of Matter States that the tiny particles in all forms of matter are in constant motion. Kinetic = motion A gas is composed of particles, usually molecules or atoms, with negligible volume
Module 3: "Thin Film Hydrodynamics" Lecture 11: "" The Lecture Contains: Micro and Nano Scale Hydrodynamics with and without Free Surfaces
 The Lecture Contains: Micro and Nano Scale Hydrodynamics with and without Free Surfaces Order of Magnitude Analysis file:///e /courses/colloid_interface_science/lecture11/11_1.htm[6/16/2012 1:39:56 PM]
The Lecture Contains: Micro and Nano Scale Hydrodynamics with and without Free Surfaces Order of Magnitude Analysis file:///e /courses/colloid_interface_science/lecture11/11_1.htm[6/16/2012 1:39:56 PM]
Thermodynamics of Surface Nanobubbles
 This is an open access article published under an ACS AuthorChoice icense, which permits copying and redistribution of the article or any adaptations for non-commercial purposes. pubs.acs.org/angmuir Thermodynamics
This is an open access article published under an ACS AuthorChoice icense, which permits copying and redistribution of the article or any adaptations for non-commercial purposes. pubs.acs.org/angmuir Thermodynamics
COMPLEX FLOW OF NANOCONFINED POLYMERS
 COMPLEX FLOW OF NANOCONFINED POLYMERS Connie B. Roth, Chris A. Murray and John R. Dutcher Department of Physics University of Guelph Guelph, Ontario, Canada N1G 2W1 OUTLINE instabilities in freely-standing
COMPLEX FLOW OF NANOCONFINED POLYMERS Connie B. Roth, Chris A. Murray and John R. Dutcher Department of Physics University of Guelph Guelph, Ontario, Canada N1G 2W1 OUTLINE instabilities in freely-standing
Module 4: "Surface Thermodynamics" Lecture 21: "" The Lecture Contains: Effect of surfactant on interfacial tension. Objectives_template
 The Lecture Contains: Effect of surfactant on interfacial tension file:///e /courses/colloid_interface_science/lecture21/21_1.htm[6/16/2012 1:10:36 PM] Surface Thermodynamics: Roles of Surfactants and
The Lecture Contains: Effect of surfactant on interfacial tension file:///e /courses/colloid_interface_science/lecture21/21_1.htm[6/16/2012 1:10:36 PM] Surface Thermodynamics: Roles of Surfactants and
Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák István Bányai
 Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák István Bányai Surfaces and Interfaces Defining of interfacial region Types of interfaces: surface vs interface Surface
Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák István Bányai Surfaces and Interfaces Defining of interfacial region Types of interfaces: surface vs interface Surface
