Transmutation of Carbon
|
|
|
- Philip Allison
- 5 years ago
- Views:
Transcription
1 This page was last modified 26 February Transmutation of Carbon In 1994, R. Sundaresan and J.Bockris (Texas A&M) reported that they had observed "Anomalous Reactions During Arcing Between Carbon Rods In Water: "Spectroscopically pure carbon rods were subjected to a carbon arc in highly purified water. The arc current varied from 20 to 25 A and was passed intermittently for several hours. The original carbon contained ~ 2 ppm Fe. The C rods remained cool to the touch at >2 cm from their tips. Absorption of iron from water or the surrounding atmosphere was established as not being the cause of the increase of iron. There is a weak correlation between the iron formed and the time of passage of current. "When dissolved O 2 was replaced by N 2 in the solution, no iron was formed. Hence, the mechanism 2 6 C O 18 = 26 Fe He 4 was suggested as the origin of the iron. The increase in temperature of the solution was consistent with expectation based on this reaction." The Fe which is produced by this transmutation is stainless. It does not rust easily. It has also much less reaction to heat than ordinary iron, due to its composition of 2 Si (silicon) atoms. This iron was name G.O.S. (George Ohsawa Steel), given the initials of George Ohsawa by the scientists who worked with this transmutation. All results of the transmutation of Fe have been carefully examined and analyzed by several methods including: magnetic inspection, spectroscopic (1 of 7)7/4/2006 1:19:07 PM
2 analysis, chemical analysis, and examination by reagent, confirmed by authoritative testing agencies. Also in 1994, another group of researchers (M. Singh, et al.) at the Bhabha Atomic Research Centre (Bombay) reported their "Verification of the G. Ohsawa Experiment for Anomalous Production of Iron from Carbon Arc in Water: "A direct current arc was run between ultrapure graphite electrodes dipped in ultrapure water for 1-20 hours. The graphite residue collected at the bottom of the water trough was analyzed for Fe content by a conventional spectrographic method... The Fe content was fairly high, depending on the duration of the arcing... The results showed large variations in Fe content (50 to 2000 ppm) in the C residue. In the second series of experiments... with the water trough fully covered, the amount of Fe in the carbon residue decreased significantly ( ppm). Here also there were large variations in the iron concentration in the residue, although the experiments were performed under identical conditions. Whether Fe is really being synthesized through transmutation from C and O as suggested by George Ohsawa or is getting concentrated to different degrees through some other phenomenon is not currently clear. The Fe in the C residue was also analyzed by mass spectroscopy for the abundance of various isotopes... Besides Fe, the presence of other elements like Si, Ni, Al, and Cr was also determined in the C residue, and it was found that the variation of their concentrations followed the same pattern as that of Fe." Anomalies of Generated Molecules Santilli's main hypothesis for the resulting gases anomalies is that, at the time of their formation under an electric arc, gases H2, CO, CO2, O2, etc. do not have a conventional structure because the orbits of their valence electrons, and maybe also their necleus shells are mostly polarized in a plane due to the very intense magnetic field surrounding the electric arc (of the order of 10 Tesla or more). In turn, such a polarization implies the creation of strong magnetic moments, resulting in new magnetic bonds constituting magnecules. The anomalous IR signature of Carbon Monoxide (top) compared to the conventional one (bottom), establishing a polarization of the orbits of valence and other electrons. (2 of 7)7/4/2006 1:19:07 PM
3 The experimental verification of the fuel gas requires the detection of a number of anomalies that can be summarized as follows. All these anomalies have been experimentally verified. Anomaly 1: Appearance of unexpected heavy MS peaks. Fuel gas molecules, referred as magnecules are generally heavier than the heaviest molecule in a given gas. Peaks in the GC-MS are therefore expected in macroscopic percentages with molecular weights bigger than the heaviest molecule. These heavy composites should not provide MS peaks according to quantum chemistry, thus constituting an anomaly. As an example, by ignoring heavy compounds in parts per million [ppm], Magnegases TM should have no large peak in the GC-MS with more than the CO 2 molecular weight of 44 a.m.u. The existence of heavier large peaks would establish this first anomaly. Anomaly 2: "Unknown" character of the unexpected heavy peaks. To provide the initial premises for magnecules, the peaks of Anomaly 1 should result in "unknown" in the search by the GC-MS computer in its memory banks of conventional molecules, usually including about 150,000 molecules. Anomaly 3: Lack of IR signature of the "unknown" peaks. Another necessary condition to have magnecules is that the "unknown" peaks of Anomaly 1 should have no infrared signature at all. According to established evidence, all gases with a valence bond must have a well defined infrared signature [with a few exceptions of spherically symmetric molecules, such as hydrogen]. In the event the peaks of Anomaly 1 do have an infrared signature, they can be constituted by new yet conventional molecules not identified before. The only infrared signatures of any given gas magnecule should be those of the conventional molecules and atoms constituting the cluster itself. As an illustration, the only admissible infrared signatures of magnecule {O2}x{O2} are those of the conventional molecules O-O and C-O. Anomaly 4: Mutation of conventional IR signatures. The infrared signatures of the molecules constituting a magnecules are expected to be mutated, in the sense that the shape of their peaks is not the established one. This is another anomaly of magnecules expected from the polarization of the orbits of the valence and other electrons. In fact, this polarization implies space distributions of the orbitals different than the conventional ones, thus resulting in a deformation of the shape of the IR peaks. Moreover, the same polarizations are expected to create additional strong bonds within a conventional molecule, that are expected to appear as new IR peaks. Still in turn, such an internal mutations of conventional molecules have far reaching scientific and technological implications, as will be shown. Anomaly 5: Mutation of magnecules. While molecules preserve their structure at conventional temperatures and pressures, this is not the case for magnecules, that are expected to mutate in time, that is, to change the shape of the MS peaks due to change in their constituents. Since we are referring to gases whose constituents notoriously collide, magnecules can break-down into parts during collisions, which parts can then recombine with other magnecules to form new clusters. Alternatively, magnecules are expected to experience accretion [or emission] of polarized conventional atoms or molecules without necessarily breaking down into parts. It follows that the peaks of Anomaly 1 are not expected to remain the same over a sufficient period of time for the same gas under the same conditions. Anomaly 6: Mutated physical characteristics. Magnetically polarized gases are expected to have mutated physical characteristics because the very notion of polarization of the orbits implies a smaller average molecular volume. Mutations of other physical characteristics are then consequential. Anomaly 7: Anomalous adhesion. (3 of 7)7/4/2006 1:19:07 PM
4 Magnetically polarized gases are expected to have anomalous adhesion to walls of disparate nature as compared to the same unpolarized gas. This is due to the well known property that magnetism can be propagated by induction, according to which a magnetically polarized molecule with a sufficiently intense magnetic moment can induce a corresponding polarization of valence [and_or other] electrons in the atoms or molecules constituting the walls surface. Once such a polarization is created by induction, magnecules can have rather strong magnetic bonds to said walls. Anomaly 8: Increased penetration through substances. Magnetically polarized gases are expected to have anomalous absorption or penetration through other substances. This is first due to the reduction of the average molecular volume with inherent increase of permeability, as compared to the same unpolarized gas. The second reason is the magnetic induction of the preceding anomaly. Anomaly 9: Increased energy release. Magnetically polarized gases are expected to have thermochemical reactions with macroscopic increases of energy releases, as compared to the same reactions among unpolarized gases, an expected anomaly that, alone, has large scientific and industrial significance. (4 of 7)7/4/2006 1:19:07 PM
5 (5 of 7)7/4/2006 1:19:07 PM
6 (6 of 7)7/4/2006 1:19:07 PM
7 (7 of 7)7/4/2006 1:19:07 PM
or other reasons. The use of hydrogen in fuel cells is afflicted by similar problems which are inherent in the low specific weight of conventional
 Preprint Institute for Basic Research IBR-TC-033, of December 29, 2001 in press at the International Journal of Hydrogen Energy, Pergamon Press, Oxford, England THE NOVEL MAGNECULAR SPECIES OF HYDROGEN
Preprint Institute for Basic Research IBR-TC-033, of December 29, 2001 in press at the International Journal of Hydrogen Energy, Pergamon Press, Oxford, England THE NOVEL MAGNECULAR SPECIES OF HYDROGEN
A STUDY OF POLYCARBONYL COMPOUNDS IN MAGNEGASES
 A STUDY F PLYARBNYL MPUNDS IN MAGNEGASES A.K. Aringazin 1,2 and R.M. Santilli 2 1 Institute for Basic Research, Department of Theoretical Physics, Eurasian National University, Astana 473021 Kazakstan
A STUDY F PLYARBNYL MPUNDS IN MAGNEGASES A.K. Aringazin 1,2 and R.M. Santilli 2 1 Institute for Basic Research, Department of Theoretical Physics, Eurasian National University, Astana 473021 Kazakstan
arxiv:physics/ v1 [physics.gen-ph] 20 Dec 2001
![arxiv:physics/ v1 [physics.gen-ph] 20 Dec 2001 arxiv:physics/ v1 [physics.gen-ph] 20 Dec 2001](/thumbs/95/122582611.jpg) arxiv:physics/0112066v1 [physics.gen-ph] 20 Dec 2001 STRUCTURE AND COMBUSTION OF MAGNEGASES tm R. M. Santilli 1 and A. K. Aringazin 1,2 1 Institute for Basic Research, P.O. Box 1577, Palm Harbor, FL 64382,
arxiv:physics/0112066v1 [physics.gen-ph] 20 Dec 2001 STRUCTURE AND COMBUSTION OF MAGNEGASES tm R. M. Santilli 1 and A. K. Aringazin 1,2 1 Institute for Basic Research, P.O. Box 1577, Palm Harbor, FL 64382,
7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text , , 12.10)
 2009, Department of Chemistry, The University of Western Ontario 7a.1 7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text 11.1 11.5, 12.1 12.5, 12.10) A. Electromagnetic Radiation Energy is
2009, Department of Chemistry, The University of Western Ontario 7a.1 7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text 11.1 11.5, 12.1 12.5, 12.10) A. Electromagnetic Radiation Energy is
NCSD HIGH SCHOOL CHEMISTRY SCOPE AND SEQUENCE
 UNIT Atomic Structure & Properties Periodic Table Bonding Chemical Reactions Moles / Stoichiometry Acids and Bases 15 Days 11 Days 16 Days 16 Days 26 Days 9 Days S T A N D A R D S H.1P.1 Explain how atomic
UNIT Atomic Structure & Properties Periodic Table Bonding Chemical Reactions Moles / Stoichiometry Acids and Bases 15 Days 11 Days 16 Days 16 Days 26 Days 9 Days S T A N D A R D S H.1P.1 Explain how atomic
Universe Now. 9. Interstellar matter and star clusters
 Universe Now 9. Interstellar matter and star clusters About interstellar matter Interstellar space is not completely empty: gas (atoms + molecules) and small dust particles. Over 10% of the mass of the
Universe Now 9. Interstellar matter and star clusters About interstellar matter Interstellar space is not completely empty: gas (atoms + molecules) and small dust particles. Over 10% of the mass of the
Nuclear Magnetic Resonance (NMR) Spectroscopy Introduction:
 Nuclear Magnetic Resonance (NMR) Spectroscopy Introduction: Nuclear magnetic resonance spectroscopy (NMR) is the most powerful tool available for organic structure determination. Like IR spectroscopy,
Nuclear Magnetic Resonance (NMR) Spectroscopy Introduction: Nuclear magnetic resonance spectroscopy (NMR) is the most powerful tool available for organic structure determination. Like IR spectroscopy,
Lecture 2 nmr Spectroscopy
 Lecture 2 nmr Spectroscopy Pages 427 430 and Chapter 13 Molecular Spectroscopy Molecular spectroscopy: the study of the frequencies of electromagnetic radiation that are absorbed or emitted by substances
Lecture 2 nmr Spectroscopy Pages 427 430 and Chapter 13 Molecular Spectroscopy Molecular spectroscopy: the study of the frequencies of electromagnetic radiation that are absorbed or emitted by substances
1. The most important aspects of the quantum theory.
 Lecture 5. Radiation and energy. Objectives: 1. The most important aspects of the quantum theory: atom, subatomic particles, atomic number, mass number, atomic mass, isotopes, simplified atomic diagrams,
Lecture 5. Radiation and energy. Objectives: 1. The most important aspects of the quantum theory: atom, subatomic particles, atomic number, mass number, atomic mass, isotopes, simplified atomic diagrams,
Chapter 8 notes. 8.1 Matter. 8.1 objectives. Earth Chemistry
 Chapter 8 notes Earth Chemistry 8.1 Matter 8.1 objectives Compare chemical properties and physical properties of matter. Describe the basic structure of an atom. Compare atomic number, mass number, and
Chapter 8 notes Earth Chemistry 8.1 Matter 8.1 objectives Compare chemical properties and physical properties of matter. Describe the basic structure of an atom. Compare atomic number, mass number, and
HE2131 MODC+ ARTICLE IN PRESS. A new gaseous and combustible form of water. Ruggero Maria Santilli
 PROD. TYPE: COM PP: -6 (col.fig.: nil) HE2 MODC+ ED: S.Latha PAGN: Usha.K -- SCAN: Agnes International Journal of Hydrogen Energy ( ) www.elsevier.com/locate/ijhydene A new gaseous and combustible form
PROD. TYPE: COM PP: -6 (col.fig.: nil) HE2 MODC+ ED: S.Latha PAGN: Usha.K -- SCAN: Agnes International Journal of Hydrogen Energy ( ) www.elsevier.com/locate/ijhydene A new gaseous and combustible form
Metallic Bonds MOLECULES AND REACTIONS. Metallic Properties. Metallic Alloys. Mobility of electrons results in many properties of metals.
 Metallic Bonds MOLECULES AND REACTIONS Nucleus of metal atoms only weakly hold outer Weak attraction allows the to move from one atom to another quite freely Chapter 15, 17 Metallic Properties Mobility
Metallic Bonds MOLECULES AND REACTIONS Nucleus of metal atoms only weakly hold outer Weak attraction allows the to move from one atom to another quite freely Chapter 15, 17 Metallic Properties Mobility
Dust in the Diffuse Universe
 Dust in the Diffuse Universe Obscuring Effects Chemical Effects Thermal Effects Dynamical Effects Diagnostic Power Evidence for Grains: Chemical Effects Catalyzes molecular hydrogen formation. Depletion
Dust in the Diffuse Universe Obscuring Effects Chemical Effects Thermal Effects Dynamical Effects Diagnostic Power Evidence for Grains: Chemical Effects Catalyzes molecular hydrogen formation. Depletion
Chapter 2 Notes The Chemistry of Life
 Name: Chapter 2 Notes The Chemistry of Life Section 2-1 The Nature of Matter Date: Atoms (p. 35) The study of chemistry begins with the basic unit of matter, the. Comes from the Greek word atomos, meaning
Name: Chapter 2 Notes The Chemistry of Life Section 2-1 The Nature of Matter Date: Atoms (p. 35) The study of chemistry begins with the basic unit of matter, the. Comes from the Greek word atomos, meaning
Experimental verification for Intermediate Controlled Nuclear Fusion
 Experimental verification for Intermediate Controlled Nuclear Fusion Leong Ying 1,*, Wei Cai 2, Jerdsay Kadeisvili 3, Chris Lynch 3, Scott Marton 3, Steve Elliot 3 and Yun Yang 3 1 Thermo Fisher Scientific,
Experimental verification for Intermediate Controlled Nuclear Fusion Leong Ying 1,*, Wei Cai 2, Jerdsay Kadeisvili 3, Chris Lynch 3, Scott Marton 3, Steve Elliot 3 and Yun Yang 3 1 Thermo Fisher Scientific,
Chemistry B11 Chapter 3 Atoms
 Chapter 3 Atoms Element: is a substance that consists of identical atoms (hydrogen, oxygen, and Iron). 116 elements are known (88 occur in nature and chemist have made the others in the lab). Compound:
Chapter 3 Atoms Element: is a substance that consists of identical atoms (hydrogen, oxygen, and Iron). 116 elements are known (88 occur in nature and chemist have made the others in the lab). Compound:
 CHAPTER 5 ATOMIC STRUCTURE SHORT QUESTIONS AND ANSWERS Q.1 Why it is necessary to decrease the pressure in the discharge tube to get the cathode rays? The current does not flow through the gas at ordinary
CHAPTER 5 ATOMIC STRUCTURE SHORT QUESTIONS AND ANSWERS Q.1 Why it is necessary to decrease the pressure in the discharge tube to get the cathode rays? The current does not flow through the gas at ordinary
Figure 1. Describe what happens when two atoms of potassium react with one atom of sulfur. Give your answer in terms of electron transfer
 Q1.Figure 1 shows the outer electrons in an atom of the Group 1 element potassium and in an atom of the Group 6 element sulfur. Figure 1 (a) Potassium forms an ionic compound with sulfur. Describe what
Q1.Figure 1 shows the outer electrons in an atom of the Group 1 element potassium and in an atom of the Group 6 element sulfur. Figure 1 (a) Potassium forms an ionic compound with sulfur. Describe what
ICSE Board Class IX Chemistry Paper 3 Solution
 ICSE Board Class IX Chemistry Paper 3 Solution SECTION I Answer 1 i. The number of electrons, that atom can lose, gain or share during a chemical reaction is called its valency. ii. Solute: A solute is
ICSE Board Class IX Chemistry Paper 3 Solution SECTION I Answer 1 i. The number of electrons, that atom can lose, gain or share during a chemical reaction is called its valency. ii. Solute: A solute is
CHAPTER 2. Atoms,Elements, Periodic Table
 CHAPTER Atoms,Elements, Periodic Table 1 Vocabulary Chemistry Science that describes matter its properties, the changes it undergoes, and the energy changes that accompany those processes Matter Anything
CHAPTER Atoms,Elements, Periodic Table 1 Vocabulary Chemistry Science that describes matter its properties, the changes it undergoes, and the energy changes that accompany those processes Matter Anything
Transmutation Reality or Macrobiotic Fairy Tale?
 Transmutation Reality or Macrobiotic Fairy Tale? A recent Quantum Rabbit experiment provides evidence that contamination, not transmutation played a major role in it, and seriously calls in question all
Transmutation Reality or Macrobiotic Fairy Tale? A recent Quantum Rabbit experiment provides evidence that contamination, not transmutation played a major role in it, and seriously calls in question all
y = (e/mv 2 ) A, z = (e/mv) B, Page 1 of 6
 Rays of positive electricity Joseph John Thomson Proceedings of the Royal Society A 89, 1-20 (1913) [as excerpted in Henry A. Boorse & Lloyd Motz, The World of the Atom, Vol. 1 (New York: Basic Books,
Rays of positive electricity Joseph John Thomson Proceedings of the Royal Society A 89, 1-20 (1913) [as excerpted in Henry A. Boorse & Lloyd Motz, The World of the Atom, Vol. 1 (New York: Basic Books,
Atomic structure Calculate the number of protons, electrons and neutrons in Important terms: quantum shells, principle quantum number, energy levels,
 Atomic structure Important terms: quantum shells, principle quantum number, energy levels, Calculate the number of protons, electrons and neutrons in Isotopes Definitions? Remember - isotopes have exactly
Atomic structure Important terms: quantum shells, principle quantum number, energy levels, Calculate the number of protons, electrons and neutrons in Isotopes Definitions? Remember - isotopes have exactly
EDULABZ INTERNATIONAL 3 STRUCTURE OF ATOMS
 3 STRUCTURE OF ATOMS I. Multiple choice questions (Tick the correct option). 1. Which one of the following proposed the atomic theory of matter? (a) John Dalton (b) J.J. Thomson (c) Rutherford (d) Niels
3 STRUCTURE OF ATOMS I. Multiple choice questions (Tick the correct option). 1. Which one of the following proposed the atomic theory of matter? (a) John Dalton (b) J.J. Thomson (c) Rutherford (d) Niels
ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY
 ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY All matter is made of atoms. There are a limited number of types of atoms; these are the elements. (EU 1.A) Development of Atomic Theory Atoms are so small
ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY All matter is made of atoms. There are a limited number of types of atoms; these are the elements. (EU 1.A) Development of Atomic Theory Atoms are so small
Materials Science. Atomic Structures and Bonding
 Materials Science Atomic Structures and Bonding 1 Atomic Structure Fundamental concepts Each atom consists of a nucleus composed of protons and neutrons which are encircled by electrons. Protons and electrons
Materials Science Atomic Structures and Bonding 1 Atomic Structure Fundamental concepts Each atom consists of a nucleus composed of protons and neutrons which are encircled by electrons. Protons and electrons
Mid-Term Review Multiple Choice: Ch. 3 Identify the letter of the choice that best completes the statement or answers the question.
 HONORS CHEMISTRY MID-TERM PRACTICE January 2013 Mrs. Allen from HS North put together a collection of multiple choice questions for you to use as a study tool. Not all of the midterm topics are covered
HONORS CHEMISTRY MID-TERM PRACTICE January 2013 Mrs. Allen from HS North put together a collection of multiple choice questions for you to use as a study tool. Not all of the midterm topics are covered
Unit Two Test Review. Click to get a new slide. Choose your answer, then click to see if you were correct.
 Unit Two Test Review Click to get a new slide. Choose your answer, then click to see if you were correct. According to the law of definite proportions, any two samples of water, H2O, A. will be made up
Unit Two Test Review Click to get a new slide. Choose your answer, then click to see if you were correct. According to the law of definite proportions, any two samples of water, H2O, A. will be made up
Inorganic Spectroscopic and Structural Methods
 Inorganic Spectroscopic and Structural Methods Electromagnetic spectrum has enormous range of energies. Wide variety of techniques based on absorption of energy e.g. ESR and NMR: radiowaves (MHz) IR vibrations
Inorganic Spectroscopic and Structural Methods Electromagnetic spectrum has enormous range of energies. Wide variety of techniques based on absorption of energy e.g. ESR and NMR: radiowaves (MHz) IR vibrations
Chapter 8. Spectroscopy. 8.1 Purpose. 8.2 Introduction
 Chapter 8 Spectroscopy 8.1 Purpose In the experiment atomic spectra will be investigated. The spectra of three know materials will be observed. The composition of an unknown material will be determined.
Chapter 8 Spectroscopy 8.1 Purpose In the experiment atomic spectra will be investigated. The spectra of three know materials will be observed. The composition of an unknown material will be determined.
8. Which of the following could be an isotope of chlorine? (A) 37 Cl 17 (B) 17 Cl 17 (C) 37 Cl 17 (D) 17 Cl 37.5 (E) 17 Cl 37
 Electronic Structure Worksheet 1 Given the following list of atomic and ionic species, find the appropriate match for questions 1-4. (A) Fe 2+ (B) Cl (C) K + (D) Cs (E) Hg + 1. Has the electron configuration:
Electronic Structure Worksheet 1 Given the following list of atomic and ionic species, find the appropriate match for questions 1-4. (A) Fe 2+ (B) Cl (C) K + (D) Cs (E) Hg + 1. Has the electron configuration:
CHEM6416 Theory of Molecular Spectroscopy 2013Jan Spectroscopy frequency dependence of the interaction of light with matter
 CHEM6416 Theory of Molecular Spectroscopy 2013Jan22 1 1. Spectroscopy frequency dependence of the interaction of light with matter 1.1. Absorption (excitation), emission, diffraction, scattering, refraction
CHEM6416 Theory of Molecular Spectroscopy 2013Jan22 1 1. Spectroscopy frequency dependence of the interaction of light with matter 1.1. Absorption (excitation), emission, diffraction, scattering, refraction
Classification of spectroscopic methods
 Introduction Spectroscopy is the study of the interaction between the electromagnetic radiation and the matter. Spectrophotometry is the measurement of these interactions i.e. the measurement of the intensity
Introduction Spectroscopy is the study of the interaction between the electromagnetic radiation and the matter. Spectrophotometry is the measurement of these interactions i.e. the measurement of the intensity
Topic: Atoms and the Periodic Table
 Topic: Atoms and the Periodic Table Atoms the basic building blocks of all substances 92 different naturally-occurring types of atom the smallest amount of any element we can have Fun Facts: One cubic
Topic: Atoms and the Periodic Table Atoms the basic building blocks of all substances 92 different naturally-occurring types of atom the smallest amount of any element we can have Fun Facts: One cubic
Chapter 3 Nuclear Properties
 CHM 511 Chapter 3 page 1 of 9 Chapter 3 Nuclear Properties Chemistry typically deals with the consideration of electrons: where they are, where they want to go, where they are going. A typical chemical
CHM 511 Chapter 3 page 1 of 9 Chapter 3 Nuclear Properties Chemistry typically deals with the consideration of electrons: where they are, where they want to go, where they are going. A typical chemical
Chapter 29 Molecular and Solid-State Physics
 Chapter 29 Molecular and Solid-State Physics GOALS When you have mastered the content of this chapter, you will be able to achieve the following goals: Definitions Define each of the following terms, and
Chapter 29 Molecular and Solid-State Physics GOALS When you have mastered the content of this chapter, you will be able to achieve the following goals: Definitions Define each of the following terms, and
What is the smallest particle of the element gold (Au) that can still be classified as gold? A. atom B. molecule C. neutron D.
 Use the Periodic Table of Elements to answer the following question(s). Which sentence about the periodic table of elements is true? A. All elements in period 2 are metals. B. All elements in group 18
Use the Periodic Table of Elements to answer the following question(s). Which sentence about the periodic table of elements is true? A. All elements in period 2 are metals. B. All elements in group 18
ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY
 ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY All matter is made of atoms. There are a limited number of types of atoms; these are the elements. (EU 1.A) Development of Atomic Theory Atoms are so small
ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY All matter is made of atoms. There are a limited number of types of atoms; these are the elements. (EU 1.A) Development of Atomic Theory Atoms are so small
Chemistry Vocabulary. These vocabulary words appear on the Chemistry CBA in addition to being tested on the Chemistry Vocabulary Test.
 Chemistry Vocabulary These vocabulary words appear on the Chemistry CBA in addition to being tested on the Chemistry Vocabulary Test. atom the smallest unit of an element that still represents that element.
Chemistry Vocabulary These vocabulary words appear on the Chemistry CBA in addition to being tested on the Chemistry Vocabulary Test. atom the smallest unit of an element that still represents that element.
Chapter 7 Chemical Bonding and Molecular Geometry
 Chapter 7 Chemical Bonding and Molecular Geometry 347 Chapter 7 Chemical Bonding and Molecular Geometry Figure 7.1 Nicknamed buckyballs, buckminsterfullerene molecules (C60) contain only carbon atoms.
Chapter 7 Chemical Bonding and Molecular Geometry 347 Chapter 7 Chemical Bonding and Molecular Geometry Figure 7.1 Nicknamed buckyballs, buckminsterfullerene molecules (C60) contain only carbon atoms.
CHAPTER 3. Crystallography
 CHAPTER 3 Crystallography Atomic Structure Atoms are made of Protons: mass 1.00728 amu, +1 positive charge Neutrons: mass of 1.00867 amu, neutral Electrons: mass of 0.00055 amu, -1 negative charge (1 amu
CHAPTER 3 Crystallography Atomic Structure Atoms are made of Protons: mass 1.00728 amu, +1 positive charge Neutrons: mass of 1.00867 amu, neutral Electrons: mass of 0.00055 amu, -1 negative charge (1 amu
Chemistry 104 Final Exam Content Evaluation and Preparation for General Chemistry I Material
 Chemistry 104 Final Exam Content Evaluation and Preparation for General Chemistry I Material What is 25 mph in mm s 1? Unit conversions What is 1025 K in o F? Which is larger 1 ft 3 or 0.1 m 3? What is
Chemistry 104 Final Exam Content Evaluation and Preparation for General Chemistry I Material What is 25 mph in mm s 1? Unit conversions What is 1025 K in o F? Which is larger 1 ft 3 or 0.1 m 3? What is
THE ATOMIC NATURE OF MATTER Chapter 11. Lecture 1
 THE ATOMIC NATURE OF MATTER Chapter 11 Lecture 1 Armen Kocharian Objectives: The Atomic Hypothesis Characteristics of Atoms Atomic Imagery Atomic Structure The Elements The Periodic Table of Elements Relative
THE ATOMIC NATURE OF MATTER Chapter 11 Lecture 1 Armen Kocharian Objectives: The Atomic Hypothesis Characteristics of Atoms Atomic Imagery Atomic Structure The Elements The Periodic Table of Elements Relative
Atomic Structure. Niels Bohr Nature, March 24, 1921
 1 of 5 4/26/2013 12:47 AM Atomic Structure Niels Bohr Nature, March 24, 1921 In a letter to NATURE of November 25 last Dr. Norman Campbell discusses the problem of the possible consistency of the assumptions
1 of 5 4/26/2013 12:47 AM Atomic Structure Niels Bohr Nature, March 24, 1921 In a letter to NATURE of November 25 last Dr. Norman Campbell discusses the problem of the possible consistency of the assumptions
January 29, 2019 Chemistry 328N
 Lecture 3 NMR Spectroscopy January 29, 2019 Molecular Spectroscopy Molecular spectroscopy: the study of the frequencies of electromagnetic radiation that are absorbed or emitted by substances and the correlation
Lecture 3 NMR Spectroscopy January 29, 2019 Molecular Spectroscopy Molecular spectroscopy: the study of the frequencies of electromagnetic radiation that are absorbed or emitted by substances and the correlation
Teacher: Mr. gerraputa. Name: Base your answer to the question on the information below. Given the electron dot diagram:
 Teacher: Mr. gerraputa Print Close Name: 1. Given the electron dot diagram: The valence electrons represented by the electron dot diagram could be those of atoms in Group 1. 13 3. 3 2. 15 4. 16 2. Which
Teacher: Mr. gerraputa Print Close Name: 1. Given the electron dot diagram: The valence electrons represented by the electron dot diagram could be those of atoms in Group 1. 13 3. 3 2. 15 4. 16 2. Which
Unit 1 Test A Atomic Theory & Nuclear Decay 1. Which of these BEST describes any two atoms of the same element? a. same number of protons
 1. Which of these BEST describes any two atoms of the same element? same number of protons same number of chemical bonds same number of neutrons same number of particles in the nucleus Self Assessment
1. Which of these BEST describes any two atoms of the same element? same number of protons same number of chemical bonds same number of neutrons same number of particles in the nucleus Self Assessment
The Claviature of Gas Analysis
 The Claviature of Gas Analysis Mass spectrometric gas analysis for quality assurance of gases and gas mixtures High purity and special gases are becoming increasingly important in the development of modern
The Claviature of Gas Analysis Mass spectrometric gas analysis for quality assurance of gases and gas mixtures High purity and special gases are becoming increasingly important in the development of modern
Lecture 3 NMR Spectroscopy. January 26, 2016 Chemistry 328N
 Lecture 3 NMR Spectroscopy January 26, 2016 Please see me after class Enrollment issue Hugo Nicolas Eichner Nurbol Kaliyev E = hn Energy per photon Frequency Wave Length Electromagnetic Radiation Important
Lecture 3 NMR Spectroscopy January 26, 2016 Please see me after class Enrollment issue Hugo Nicolas Eichner Nurbol Kaliyev E = hn Energy per photon Frequency Wave Length Electromagnetic Radiation Important
Principles of Molecular Spectroscopy: Electromagnetic Radiation and Molecular structure. Nuclear Magnetic Resonance (NMR)
 Principles of Molecular Spectroscopy: Electromagnetic Radiation and Molecular structure Nuclear Magnetic Resonance (NMR) !E = h" Electromagnetic radiation is absorbed when the energy of photon corresponds
Principles of Molecular Spectroscopy: Electromagnetic Radiation and Molecular structure Nuclear Magnetic Resonance (NMR) !E = h" Electromagnetic radiation is absorbed when the energy of photon corresponds
How does your eye form an Refraction
 Astronomical Instruments Eyes and Cameras: Everyday Light Sensors How does your eye form an image? How do we record images? How does your eye form an image? Refraction Refraction is the bending of light
Astronomical Instruments Eyes and Cameras: Everyday Light Sensors How does your eye form an image? How do we record images? How does your eye form an image? Refraction Refraction is the bending of light
Personalised Learning Checklists AQA Chemistry Paper 1
 AQA Chemistry (8462) from 2016 Topics C4.1 Atomic structure and the periodic table State that everything is made of atoms and recall what they are Describe what elements and compounds are State that elements
AQA Chemistry (8462) from 2016 Topics C4.1 Atomic structure and the periodic table State that everything is made of atoms and recall what they are Describe what elements and compounds are State that elements
Nuclear Magnetic Resonance Spectroscopy (NMR)
 OCR Chemistry A 432 Spectroscopy (NMR) What is it? An instrumental method that gives very detailed structural information about molecules. It can tell us - how many of certain types of atom a molecule
OCR Chemistry A 432 Spectroscopy (NMR) What is it? An instrumental method that gives very detailed structural information about molecules. It can tell us - how many of certain types of atom a molecule
AQA C1 Atomic Structure
 AQA C1 Atomic Structure What s in an atom? Elements in the periodic have different sizes of atoms. The size of the atoms depends on the number of protons, electrons and neutrons they have. Each element
AQA C1 Atomic Structure What s in an atom? Elements in the periodic have different sizes of atoms. The size of the atoms depends on the number of protons, electrons and neutrons they have. Each element
Additional Science Chemistry
 Additional Science Chemistry C2 Core Questions and Keywords and Definitions Question How did Mendeleev arrange the elements known at the time into a periodic table? How did Mendeleev use his table? Where
Additional Science Chemistry C2 Core Questions and Keywords and Definitions Question How did Mendeleev arrange the elements known at the time into a periodic table? How did Mendeleev use his table? Where
Collegiate Institute for Math and Science Day 57: December 9, 2016 Room 427
 Unit 2: Atomic Concepts Outline Name: Period: Date: 1. The modern model of the atom has evolved over a long period of time through the work of many scientists. Dalton s Model: Elements are made of atoms
Unit 2: Atomic Concepts Outline Name: Period: Date: 1. The modern model of the atom has evolved over a long period of time through the work of many scientists. Dalton s Model: Elements are made of atoms
Form A. Exam 1, Ch 1-4 September 23, Points
 Chem 130 Name Exam 1, Ch 1-4 September 23, 2011 100 Points Please follow the instructions for each section of the exam. Show your work on all mathematical problems. Provide answers with the correct units
Chem 130 Name Exam 1, Ch 1-4 September 23, 2011 100 Points Please follow the instructions for each section of the exam. Show your work on all mathematical problems. Provide answers with the correct units
U N I T T E S T P R A C T I C E
 South Pasadena AP Chemistry Name 8 Atomic Theory Period Date U N I T T E S T P R A C T I C E Part 1 Multiple Choice You should allocate 25 minutes to finish this portion of the test. No calculator should
South Pasadena AP Chemistry Name 8 Atomic Theory Period Date U N I T T E S T P R A C T I C E Part 1 Multiple Choice You should allocate 25 minutes to finish this portion of the test. No calculator should
The wavefunction that describes a bonding pair of electrons:
 4.2. Molecular Properties from VB Theory a) Bonding and Bond distances The wavefunction that describes a bonding pair of electrons: Ψ b = a(h 1 ) + b(h 2 ) where h 1 and h 2 are HAOs on adjacent atoms
4.2. Molecular Properties from VB Theory a) Bonding and Bond distances The wavefunction that describes a bonding pair of electrons: Ψ b = a(h 1 ) + b(h 2 ) where h 1 and h 2 are HAOs on adjacent atoms
Chem 3502/4502 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 2006 Christopher J. Cramer. Lecture 10, February 10, / 4
 Chem 350/450 Physical Chemistry II (Quantum Mechanics 3 Credits Spring Semester 006 Christopher J. Cramer Lecture 10, February 10, 006 Solved Homework We are asked to find and for the first two
Chem 350/450 Physical Chemistry II (Quantum Mechanics 3 Credits Spring Semester 006 Christopher J. Cramer Lecture 10, February 10, 006 Solved Homework We are asked to find and for the first two
T2-1P1 [187 marks] [1 mark] How many protons, neutrons and electrons are present in each atom of 31 P? [1 mark]
![T2-1P1 [187 marks] [1 mark] How many protons, neutrons and electrons are present in each atom of 31 P? [1 mark] T2-1P1 [187 marks] [1 mark] How many protons, neutrons and electrons are present in each atom of 31 P? [1 mark]](/thumbs/89/100541620.jpg) T2-1P1 [187 marks] 1. How many protons, neutrons and electrons are present in each atom of 31 P? 2. What is the atomic number of a neutral atom which has 51 neutrons and 40 electrons? A. 40 B. 51 C. 91
T2-1P1 [187 marks] 1. How many protons, neutrons and electrons are present in each atom of 31 P? 2. What is the atomic number of a neutral atom which has 51 neutrons and 40 electrons? A. 40 B. 51 C. 91
1 st shell holds 2 electrons. 2 nd shell holds 8 electrons
 ATOM INDIVISIBLE ELEMENTS - Nucleus = protons (+ charge) & neutrons (no charge ) - Electrons (- charge) orbit the nucleus in shells of 2, 8, 8 electrons (inner orbit outward) - Atomic number = number of
ATOM INDIVISIBLE ELEMENTS - Nucleus = protons (+ charge) & neutrons (no charge ) - Electrons (- charge) orbit the nucleus in shells of 2, 8, 8 electrons (inner orbit outward) - Atomic number = number of
William H. Brown & Christopher S. Foote
 Requests for permission to make copies of any part of the work should be mailed to:permissions Department, Harcourt Brace & Company, 6277 Sea Harbor Drive, Orlando, Florida 32887-6777 William H. Brown
Requests for permission to make copies of any part of the work should be mailed to:permissions Department, Harcourt Brace & Company, 6277 Sea Harbor Drive, Orlando, Florida 32887-6777 William H. Brown
CHAPTER 2 Building Blocks of Materials
 CHAPTER 2 Building Blocks of Materials Matter is everything that is around us that we interact with anything that occupies space and has mass. A classification scheme that you have likely seen. Chapter
CHAPTER 2 Building Blocks of Materials Matter is everything that is around us that we interact with anything that occupies space and has mass. A classification scheme that you have likely seen. Chapter
Chemical Reactions. Chapter 17
 Chemical Reactions Chapter 17 Chemical Equations C+O 2 CO 2 C (s) +O 2 (g) CO 2 (g) Reactants on left, products on right Each are balanced because same number of atoms of reactants as products Some equations
Chemical Reactions Chapter 17 Chemical Equations C+O 2 CO 2 C (s) +O 2 (g) CO 2 (g) Reactants on left, products on right Each are balanced because same number of atoms of reactants as products Some equations
Regents Chemistry. Topic Review Packet. Name:
 Regents Chemistry Topic Review Packet Name: 1 p. 5-7 Matter 2. 2 3. 3 4. 1 5. 4 6. 3 7. 1 Answer Key 8. 3 10. 3 11. 1 12. 4 13. 3 14. 4 15. density of neon gas = 0.827 grams/liter 16. one physical property
Regents Chemistry Topic Review Packet Name: 1 p. 5-7 Matter 2. 2 3. 3 4. 1 5. 4 6. 3 7. 1 Answer Key 8. 3 10. 3 11. 1 12. 4 13. 3 14. 4 15. density of neon gas = 0.827 grams/liter 16. one physical property
Specimen. Date Morning/Afternoon Time allowed: 1 hour 30 minutes. AS Level Chemistry B (Salters) H033/02 Chemistry in depth Sample Question Paper PMT
 AS Level Chemistry B (Salters) H033/02 Chemistry in depth Sample Question Paper Date Morning/Afternoon Time allowed: 1 hour 30 minutes You must have: the Data Sheet for Chemistry B (Salters) You may use:
AS Level Chemistry B (Salters) H033/02 Chemistry in depth Sample Question Paper Date Morning/Afternoon Time allowed: 1 hour 30 minutes You must have: the Data Sheet for Chemistry B (Salters) You may use:
Unit 11 Instrumentation. Mass, Infrared and NMR Spectroscopy
 Unit 11 Instrumentation Mass, Infrared and NMR Spectroscopy Spectroscopic identification of organic compounds Qualitative analysis: presence but not quantity (i.e. PEDs) Quantitative analysis: quantity
Unit 11 Instrumentation Mass, Infrared and NMR Spectroscopy Spectroscopic identification of organic compounds Qualitative analysis: presence but not quantity (i.e. PEDs) Quantitative analysis: quantity
Page 2. Q1.The electronic structure of the atoms of five elements are shown in the figure below. The letters are not the symbols of the elements.
 Q1.The electronic structure of the atoms of five elements are shown in the figure below. The letters are not the symbols of the elements. Choose the element to answer the question. Each element can be
Q1.The electronic structure of the atoms of five elements are shown in the figure below. The letters are not the symbols of the elements. Choose the element to answer the question. Each element can be
In the Beginning. After about three minutes the temperature had cooled even further, so that neutrons were able to combine with 1 H to form 2 H;
 In the Beginning Obviously, before we can have any geochemistry we need some elements to react with one another. The most commonly held scientific view for the origin of the universe is the "Big Bang"
In the Beginning Obviously, before we can have any geochemistry we need some elements to react with one another. The most commonly held scientific view for the origin of the universe is the "Big Bang"
CHAPTER 2 Atoms and the Periodic Table
 CHAPTER 2 and the Periodic Table General, Organic, & Biological Chemistry Janice Gorzynski Smith CHAPTER 2: & the Periodic Table Learning Objectives:! Elemental Symbols! Metals vs Nonmetals vs Metalloids
CHAPTER 2 and the Periodic Table General, Organic, & Biological Chemistry Janice Gorzynski Smith CHAPTER 2: & the Periodic Table Learning Objectives:! Elemental Symbols! Metals vs Nonmetals vs Metalloids
Advances on Alternative Fuels with Santilli Magnecular Structure
 International Journal of Alternative Fuels, ISSN: 2051-5987, Vol.17, Issue.1 1132 ABSTRACT Advances on Alternative Fuels with Santilli Magnecular Structure Santilli, the Italian-American physicist, for
International Journal of Alternative Fuels, ISSN: 2051-5987, Vol.17, Issue.1 1132 ABSTRACT Advances on Alternative Fuels with Santilli Magnecular Structure Santilli, the Italian-American physicist, for
GENERAL CHEMISTRY FOR INTEGRATED SCIENCE 1 STUDY NOTES CHAPTER ONE
 GENERAL CHEMISTRY FOR INTEGRATED SCIENCE 1 STUDY NOTES CHAPTER ONE ELEMENTARY UNITS IN CHEMICAL REACTIONS CHAPTER ONE CONTENTS 0 Introduction 23 Objectives 0 Main Content 0 Elements, Compounds and Mixtures
GENERAL CHEMISTRY FOR INTEGRATED SCIENCE 1 STUDY NOTES CHAPTER ONE ELEMENTARY UNITS IN CHEMICAL REACTIONS CHAPTER ONE CONTENTS 0 Introduction 23 Objectives 0 Main Content 0 Elements, Compounds and Mixtures
Atomic Structure
 Answer Key Matter 1. 3 2. 2 3. 3 4. 1 5. 4 6. 3 7. 1 8. 3 9. 2 10. 3 11. 1 12. 4 13. 3 14. 4 15. density of neon gas = 0.827 grams/liter 16. one physical property is the melting of the wax 17. one indication
Answer Key Matter 1. 3 2. 2 3. 3 4. 1 5. 4 6. 3 7. 1 8. 3 9. 2 10. 3 11. 1 12. 4 13. 3 14. 4 15. density of neon gas = 0.827 grams/liter 16. one physical property is the melting of the wax 17. one indication
Introduction to Nanotechnology Chapter 5 Carbon Nanostructures Lecture 1
 Introduction to Nanotechnology Chapter 5 Carbon Nanostructures Lecture 1 ChiiDong Chen Institute of Physics, Academia Sinica chiidong@phys.sinica.edu.tw 02 27896766 Carbon contains 6 electrons: (1s) 2,
Introduction to Nanotechnology Chapter 5 Carbon Nanostructures Lecture 1 ChiiDong Chen Institute of Physics, Academia Sinica chiidong@phys.sinica.edu.tw 02 27896766 Carbon contains 6 electrons: (1s) 2,
Example: What is the number of electrons in an atom that has 3 protons and 4 neutrons? A. 3. B. 5. C. 7. D. 10.
 Structure of atom: PROTONS Protons are located in the nucleus of an atom. They carry a +1 electrical charge and have a mass of 1 atomic mass unit (u). NEUTRONS Neutrons are located in the nucleus of an
Structure of atom: PROTONS Protons are located in the nucleus of an atom. They carry a +1 electrical charge and have a mass of 1 atomic mass unit (u). NEUTRONS Neutrons are located in the nucleus of an
Q1. The electronic structure of the atoms of five elements are shown in the figure below.
 Q1. The electronic structure of the atoms of five elements are shown in the figure below. The letters are not the symbols of the elements. Choose the element to answer the question. Each element can be
Q1. The electronic structure of the atoms of five elements are shown in the figure below. The letters are not the symbols of the elements. Choose the element to answer the question. Each element can be
Creating Energy-Level Diagrams Aufbau (building up) Principle Electrons are added to the lowest energy orbital available.
 3.6 Atomic Structure and the Periodic Table Bohr's Theory Was Incorrect Because... Only explained the line spectrum of hydrogen Position and motion of an e cannot be specified (since the e is so small,
3.6 Atomic Structure and the Periodic Table Bohr's Theory Was Incorrect Because... Only explained the line spectrum of hydrogen Position and motion of an e cannot be specified (since the e is so small,
5 questions, 3 points each, 15 points total possible. 26 Fe Cu Ni Co Pd Ag Ru 101.
 Physical Chemistry II Lab CHEM 4644 spring 2017 final exam KEY 5 questions, 3 points each, 15 points total possible h = 6.626 10-34 J s c = 3.00 10 8 m/s 1 GHz = 10 9 s -1. B= h 8π 2 I ν= 1 2 π k μ 6 P
Physical Chemistry II Lab CHEM 4644 spring 2017 final exam KEY 5 questions, 3 points each, 15 points total possible h = 6.626 10-34 J s c = 3.00 10 8 m/s 1 GHz = 10 9 s -1. B= h 8π 2 I ν= 1 2 π k μ 6 P
Galaxies and the Universe. Our Galaxy - The Milky Way The Interstellar Medium
 Galaxies and the Universe Our Galaxy - The Milky Way The Interstellar Medium Our view of the Milky Way The Radio Sky COBE Image of our Galaxy The Milky Way Galaxy - The Galaxy By Visual Observation
Galaxies and the Universe Our Galaxy - The Milky Way The Interstellar Medium Our view of the Milky Way The Radio Sky COBE Image of our Galaxy The Milky Way Galaxy - The Galaxy By Visual Observation
Unit 8: Atomic Theory. Quantum Mechanics
 Unit 8: Atomic Theory Quantum Mechanics 1 Unit 8: Atomic Theory 1. Historical Views of the Atom 2. The 'New' Look Atom 3. Electron Configurations 4. Electron Configurations & the Periodic Table 5. Quantum
Unit 8: Atomic Theory Quantum Mechanics 1 Unit 8: Atomic Theory 1. Historical Views of the Atom 2. The 'New' Look Atom 3. Electron Configurations 4. Electron Configurations & the Periodic Table 5. Quantum
Topic 3: Periodic Trends and Atomic Spectroscopy
 Topic 3: Periodic Trends and Atomic Spectroscopy Introduction Valence Electrons are those in the outer most shell of an element and are responsible for the bonding characteristics of that element. Core
Topic 3: Periodic Trends and Atomic Spectroscopy Introduction Valence Electrons are those in the outer most shell of an element and are responsible for the bonding characteristics of that element. Core
Element. Molecule. atoms. They are organized. Alkali Metals. The basic building blocks of all matter. Can be found on the periodic table.
 Element The basic building blocks of all matter. Can be found on the periodic table. Molecule A unit of two or more atoms joined together by a chemical bond atoms The smallest particle of an element that
Element The basic building blocks of all matter. Can be found on the periodic table. Molecule A unit of two or more atoms joined together by a chemical bond atoms The smallest particle of an element that
Atomic and Nuclear Physics. Topic 7.3 Nuclear Reactions
 Atomic and Nuclear Physics Topic 7.3 Nuclear Reactions Nuclear Reactions Rutherford conducted experiments bombarding nitrogen gas with alpha particles from bismuth-214. He discovered that fast-moving particles
Atomic and Nuclear Physics Topic 7.3 Nuclear Reactions Nuclear Reactions Rutherford conducted experiments bombarding nitrogen gas with alpha particles from bismuth-214. He discovered that fast-moving particles
Welcome to Organic Chemistry II
 Welcome to Organic Chemistry II Erika Bryant, Ph.D. erika.bryant@hccs.edu Class Syllabus 3 CHAPTER 12: STRUCTURE DETERMINATION 4 What is this solution Soda Tea Coffee??? 5 What is this solution Soda Tea
Welcome to Organic Chemistry II Erika Bryant, Ph.D. erika.bryant@hccs.edu Class Syllabus 3 CHAPTER 12: STRUCTURE DETERMINATION 4 What is this solution Soda Tea Coffee??? 5 What is this solution Soda Tea
Formation of the Earth and Solar System
 Formation of the Earth and Solar System a. Supernova and formation of primordial dust cloud. NEBULAR HYPOTHESIS b. Condensation of primordial dust. Forms disk-shaped nubular cloud rotating counterclockwise.
Formation of the Earth and Solar System a. Supernova and formation of primordial dust cloud. NEBULAR HYPOTHESIS b. Condensation of primordial dust. Forms disk-shaped nubular cloud rotating counterclockwise.
What is a theory? An organized system of accepted knowledge that applies in a variety of circumstances to explain a specific set of phenomena
 Atomic Structure What is a theory? An organized system of accepted knowledge that applies in a variety of circumstances to explain a specific set of phenomena Early Theories Democritus: 4 B.C.: atom He
Atomic Structure What is a theory? An organized system of accepted knowledge that applies in a variety of circumstances to explain a specific set of phenomena Early Theories Democritus: 4 B.C.: atom He
Na Na + +e - Cl+e - Cl -
 LAB-Ionic vs. Covalent Bonding Have you ever accidentally used salt instead of sugar? Drinking tea that has been sweetened with salt or eating vegetables that have been salted with sugar tastes awful!
LAB-Ionic vs. Covalent Bonding Have you ever accidentally used salt instead of sugar? Drinking tea that has been sweetened with salt or eating vegetables that have been salted with sugar tastes awful!
Electronic structure of atoms
 Chapter 1 Electronic structure of atoms light photons spectra Heisenberg s uncertainty principle atomic orbitals electron configurations the periodic table 1.1 The wave nature of light Much of our understanding
Chapter 1 Electronic structure of atoms light photons spectra Heisenberg s uncertainty principle atomic orbitals electron configurations the periodic table 1.1 The wave nature of light Much of our understanding
Interaction of the radiation with a molecule knocks an electron from the molecule. a. Molecule ¾ ¾ ¾ ion + e -
 Interaction of the radiation with a molecule knocks an electron from the molecule. radiation a. Molecule ¾ ¾ ¾ ion + e - This can destroy the delicate balance of chemical reactions in living cells. The
Interaction of the radiation with a molecule knocks an electron from the molecule. radiation a. Molecule ¾ ¾ ¾ ion + e - This can destroy the delicate balance of chemical reactions in living cells. The
Classification of Matter. Elements, Compounds, Mixtures
 Classification of Matter Elements, Compounds, Mixtures Introducing Little Miss Element Hi! I am Little Miss Element I am PURE SUBSTANCE I cannot be broken down into any simpler substance by means of a
Classification of Matter Elements, Compounds, Mixtures Introducing Little Miss Element Hi! I am Little Miss Element I am PURE SUBSTANCE I cannot be broken down into any simpler substance by means of a
Chapter 3-1. proton positive nucleus 1 amu neutron zero nucleus 1 amu electron negative on energy levels around the nucleus very small
 Chapter 3-1 Sub-atomic Charge Location Mass Particle proton positive nucleus 1 amu neutron zero nucleus 1 amu electron negative on energy levels around the nucleus very small The most mass of the atom
Chapter 3-1 Sub-atomic Charge Location Mass Particle proton positive nucleus 1 amu neutron zero nucleus 1 amu electron negative on energy levels around the nucleus very small The most mass of the atom
Ch(3)Matter & Change. John Dalton
 Ch(3)Matter & Change John Dalton What is Matter? Matter is anything that contains mass & volume (takes up space) Energy, such as light, heat, and sound, is NOT matter. The Particle Theory of Matter 1.
Ch(3)Matter & Change John Dalton What is Matter? Matter is anything that contains mass & volume (takes up space) Energy, such as light, heat, and sound, is NOT matter. The Particle Theory of Matter 1.
A. 24 B. 27 C. 30 D. 32 E. 33. A. It is impossible to tell from the information given. B. 294 mm C. 122 mm D. 10 mm E. 60 mm A. 1 H B. C. D. 19 F " E.
 CHEMISTRY 110 EXAM 1 Sept. 24, 2012 FORM A 1. A microwave oven uses 2.45! 10 9 Hz electromagnetic waves to heat food. What is the wavelength of this radiation in mm? A. It is impossible to tell from the
CHEMISTRY 110 EXAM 1 Sept. 24, 2012 FORM A 1. A microwave oven uses 2.45! 10 9 Hz electromagnetic waves to heat food. What is the wavelength of this radiation in mm? A. It is impossible to tell from the
Observation information obtained through the senses; observation in science often involves measurement
 Review Sheet Unit 1: The Atom Chemistry the study of the composition of matter and the changes matter undergoes Scientific Method Scientific method a logical, systematic approach to the solution of a scientific
Review Sheet Unit 1: The Atom Chemistry the study of the composition of matter and the changes matter undergoes Scientific Method Scientific method a logical, systematic approach to the solution of a scientific
Introduction to Atoms
 Introduction to Atoms Understanding Main Ideas Answer the following questions on a separate sheet of paper. 1. What three particles are found in an atom? 2. Which two particles are found in an atom s nucleus?
Introduction to Atoms Understanding Main Ideas Answer the following questions on a separate sheet of paper. 1. What three particles are found in an atom? 2. Which two particles are found in an atom s nucleus?
EARTH TAKES SHAPE 1. Define all vocabulary words. Crust: The thin and solid outermost layer of the Earth above the mantle. Mantle: The layer of rock
 EARTH TAKES SHAPE 1. Define all vocabulary words. Crust: The thin and solid outermost layer of the Earth above the mantle. Mantle: The layer of rock between the Earth s crust and core Core: The central
EARTH TAKES SHAPE 1. Define all vocabulary words. Crust: The thin and solid outermost layer of the Earth above the mantle. Mantle: The layer of rock between the Earth s crust and core Core: The central
Chapter 16 Lecture. The Cosmic Perspective Seventh Edition. Star Birth Pearson Education, Inc.
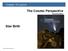 Chapter 16 Lecture The Cosmic Perspective Seventh Edition Star Birth 2014 Pearson Education, Inc. Star Birth The dust and gas between the star in our galaxy is referred to as the Interstellar medium (ISM).
Chapter 16 Lecture The Cosmic Perspective Seventh Edition Star Birth 2014 Pearson Education, Inc. Star Birth The dust and gas between the star in our galaxy is referred to as the Interstellar medium (ISM).
Stoichiometry Ratios of Combination
 Chapter 3 Stoichiometry Ratios of Combination Dr. A. Al-Saadi 1 Preview Concepts of atomic mass, molecular mass, mole, molar mass, and percent compositions. Balancing chemical equations. Stoichiometric
Chapter 3 Stoichiometry Ratios of Combination Dr. A. Al-Saadi 1 Preview Concepts of atomic mass, molecular mass, mole, molar mass, and percent compositions. Balancing chemical equations. Stoichiometric
The Chemistry of Global Warming
 The Chemistry of Global Warming Venus Atmospheric pressure is 90x that of Earth 96% CO 2 and sulfuric acid clouds Average temperature = 450 C Expected temperature based on solar radiation and distance
The Chemistry of Global Warming Venus Atmospheric pressure is 90x that of Earth 96% CO 2 and sulfuric acid clouds Average temperature = 450 C Expected temperature based on solar radiation and distance
