REACTION STUDY SHEET. NAME OF REACTION: Antarafacial (anti) Addition of an Electrophile (:E Nu:) to an Alkene
|
|
|
- Mae Wheeler
- 5 years ago
- Views:
Transcription
1 REACTONSTUDYSEET NAMEOFREACTON: Antarafacial(anti)AdditionofanElectrophile(:E Nu:)toan Alkene mportantfeatureofthisreaction:theelectrophilicatomcarriesanon bondingpairofelectrons. Generalformofthereaction: Reactant Reagent Product(s) R 1 R 4 R 2 R 3 R 1 R 2 R 4 Aspecificexample Reactant Reagent Product(s) R 3 2 /Li/TF AcO Whatwayscanthisspecificexamplebeaskedonanexam(i.e.whatflashcardsdoneed?) 2 /Li/TF?? 2 /Li/TF AcO (±)? AcO Whatdoesthisreagentdo? t adds :E Nu: across a π bond of an alkene to give a bromohydrin. The addition is stereospecific to give the antarafacial(anti) adduct. This stereochemistry is a direct result of the involvement of a three membered cyclic onium ion intermediate, which is made possible by the lonepairontheelectrophilicatom.without this lone pair, the stereospecificity is lost because the three memberedcycliconiumionbecomesimpossibletoform.
2 Whatisthemechanismofthisreaction? R 1 R 4 R 2 R 3 R 1 R2 E R 4 R3 R 1 R 4 R 2 R 3 :Nu Whatisthestereochemistryofthisreaction?The addition is stereospecific. t occurs with antarafacial(anti)additionofthetwogroupstotheπbond(i.e.theelectrophilicatom,:e,the nucleophilicatom,nu:,areaddedtooppositefacesoftheπbond). Whatistheregiochemistryofthisreaction? The addition occurs with Markovnikov regiochemistry: the:e atom becomes bonded to the end of the π bond that is less able to carry a positivecharge(i.e.wouldmakethelessstablecarbocation),thenu:atombecomesbondedto theendoftheπbondthatisbetterabletosupportapositivecharge(i.e.wouldbethemorestable carbocation) sthereanintermediateinvolvedinthisreaction? There is a three membered cyclic onium ionintermediate.thisintermediatehaseveryatomwithacompleteouter shelloctet,whichmakes itlowerenergythanasimplecarbocation(stabilizedbyhyperconjugationorconjugationwithaπ bond),whichhasonecarbonatomthatiselectrondeficient(i.e.lacksacompleteoctet). Does this intermediate lead to any special reactivity or observations? Yes: this threemembered ring must open anti, so the overall addition is always antarafacial. When the double bond is part of a cyclohexene ring, the E Nu groups both end up axial, although the ring may then flip to make them both equatorial. Antarafacial addition will supersede the regiochemical preferenceformarkovnikovaddition. Whatreagentorcombinationsofreagentscanbeusedtomaketheactivereagentinthis reaction? AllreagentsareseparatedintotheE Nusegments. a.addingx O:X2/2O;2/K/2O;2/K/2O;NaO3/NaSO3/2SO4/2O;NaO/2O; g()2/2o/tf;etc. b.addingx OR:2/RO;g(OCOCF3)2/RO;etc. c.addingx OCOR[thisreactionisespeciallyusefulinanintramolecularversionbecauseit givescyclicesters,orlactones]:2/rco2li/tf;g()2/tf;g()2/eto;etc. d.adding N3:N3/C22 e.addingrs(e) X:PhS/C22;PhSe/C22;etc. f.addingx Y:2/Li/MeCN;/C22;etc. g.addingx NCO2R[thisreactionismostusefulintheintramolecularversion]: 2/ROCON2;g(OCOCF3)2/ROCON2;etc.
3 PracticeProblems: Whatisthemajororganicproductformedfromeachofthefollowingalkeneswiththe reagentspecified? (1)2/2O (2)g(OCOCF3)2/TF/MeO (3)C65Se/C22 (4)2/LiOCOC3/TF. (5)N3/C22 (6)C3S/C22 (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) (m) (n) (o) (p) (q) (r) Whatalkenereagentcombinationshouldbeusedtoprepareeachofthefollowing additionproducts?[onlyoneenantiomerisshown,althoughsomeproductsmayberacemic] fmorethanonediastereoisomerorregioisomerwillbeformedasamajorproduct,givethe structureofthatotherproduct. (a) O (b) (c) O (d) (e) (f) go 2 CCF 3 (g) (h) O
4 (i) (j) g (k) S (l) O (m) AcOg (n) O (o) (p) O (q) (r) O SELECTEDANSWERS What is the major organic product formed from each of the following alkenes with the reagent specified? (s) O (t) MeO go 2 CCF 3 (b) The starting alkene is symmetrical not chiral; the product is racemic, but there is no regiochemistry involved in this addition. The antarafacial addition may result in addition of the electrophilic atom to the top face or the bottom face of the double bond with equal facility, but note how the two new chiral centers have stereochemistry that reflects the stereochemistry of the original double bond (Z). (1) (2) (3) (4) (5) (6) O MeO g OCOCF 3 SeC 6 5 SC 3 (f) The starting alkene is not symmetrical not chiral; the product is racemic, this addition will occur with Markovnikov regiochemistry. The antarafacial addition may result in addition of the electrophilic atom to the top face or the bottom face of the double bond with equal facility, but note how the two new chiral centers have stereochemistry that reflects the E stereochemistry of the original double bond. (1) (2) (3) go 2 CCF 3 O
5 (4) (5) (6) (i) The starting alkene is not symmetrical is chiral, the tert-butyl group prevernts the ring from inverting. Antarafacial addition in cyclohexanes forces the two new σ bonds to be formed in a trans-diaxial relationship. This supersedes the Markovnikov rule for regiochemistry of the addition. The upper product results from initial addition of the electrophilic atom to the top face of the π bond; the lower product results from initial addition of the electrophilic atom to the bottom face of the π bond. The products are regioisomers, but are both E isomers. The result of the reaction may be better understood seen in the chair projection formulas. (1) (2) (3) O gococf 3 O O gococf 3 gococf 3 O gococf 3 (4) (5) (6) What alkene reagent combination should be used to prepare each of the following addition products? [Only one enantiomer is shown, although some products may be racemic] R (a) The functional groups in this molecule are the bromine the hydroxy groups, which are on adjacent carbon atoms. This locates the O position of the double bond; these groups can be incorporated into a O R molecule by the antarafacial addition of O to an alkene through a three-membered cyclic bromonium ion intermediate. When we examine the Newman projection of this product, we see that when the bromine the hydroxy groups are anti to each other, so are the hydrogen atoms the alkyl groups. This means that in the starting alkene, the alkyl groups must be anti to each other, so must the hydrogen atoms. The starting alkene is therefore E-4-octene.
6 2 / 2 O (±) O R (b) The functional groups in this molecule are the iodine R R the azide groups, which are on adjacent carbon atoms. This locates the position of the double bond; these groups R can be incorporated into a molecule by the antarafacial addition of to an alkene through a three-membered cyclic iodonium ion intermediate. When we examine the Newman projection of this product, we see that when the iodine the azide group are syn to each other, so the first thing we must do is to re-write the Newman projection so that these two groups are anti to each other. n this conformation, the two alkyl groups are syn to each other, so are the hydrogen atoms. This means that in the starting alkene, the alkyl groups must be syn to each other, so must the hydrogen atoms. The starting alkene is therefore Z-4-octene. /C 2 2 (±) (f) The functional groups in this molecule are the methoxy group the mercury group, which are on adjacent carbon atoms. This locates the position of the double bond. They are both axial, which places them in an anti relationship; they can be go 2 CCF 3 incorporated into a molecule by the antarafacial addition of MeO gococf 3 to an alkene in methanol solvent through a three-membered cyclic mercurinium ion intermediate. The reagent itself is formed by using mervury () trifluoroacetate in methanol. g(ococf 3 ) 2 /MeO go 2 CCF 3 AcOg Ph (m) The functional groups in this molecule are the acetate the g mercuriacetate groups, which are on adjacent carbon atoms. This AcO locates the position of the double bond; these groups can be R incorporated into a molecule by the antarafacial addition of AcOg to an alkene through a three-membered cyclic mercurinium ion intermediate. The mercury atom is attached to a secondary carbon, the acetate is secondary benzylic, which corresponds to Markovnikov regiochemistry in the addition. When we examine the Newman projection of this product, we see that the bromine sulfur atoms are anti to each other, as are the two alkyl groups the two hydrogen atoms. This means that in the starting alkene, the alkyl groups must be anti to each other, so must the hydrogen atoms. The starting alkene is therefore the E isomer. The other chiral center present in the product is not affected during the reaction, so it must have been present already in the starting alkene. g() 2 /TF AcOg AcOg
Chapter 8 Alkenes and Alkynes II: Addition Reactions
 Chapter 8 Alkenes and Alkynes II: Addition Reactions Introduction: Additions to Alkenes Generally the reaction is exothermic because one π and one σ bond are converted to two σ bonds The π electrons of
Chapter 8 Alkenes and Alkynes II: Addition Reactions Introduction: Additions to Alkenes Generally the reaction is exothermic because one π and one σ bond are converted to two σ bonds The π electrons of
Chapter 8. Alkenes and Alkynes II: Addition Reactions. Ch. 8-1
 hapter 8 Alkenes and Alkynes II: Addition Reactions h. 8-1 1. Addition Reactions of Alkenes E + E Nu Nu h. 8-2 1A. ow To Understand Additions to Alkenes This is an addition reaction: E Nu added across
hapter 8 Alkenes and Alkynes II: Addition Reactions h. 8-1 1. Addition Reactions of Alkenes E + E Nu Nu h. 8-2 1A. ow To Understand Additions to Alkenes This is an addition reaction: E Nu added across
Chapter 8 Alkenes and Alkynes II: Addition Reactions. Alkenes are electron rich. Additions to Alkenes
 Additions to Alkenes Chapter 8 Alkenes and Alkynes II: Addition Reactions Generally the reaction is exothermic because one p and one s bond are converted to two s bonds Alkenes are electron rich The carbocation
Additions to Alkenes Chapter 8 Alkenes and Alkynes II: Addition Reactions Generally the reaction is exothermic because one p and one s bond are converted to two s bonds Alkenes are electron rich The carbocation
2/26/18. Practice Questions. Practice Questions B F. How many steps are there in this reaction?
 Practice Questions Practice Questions D B F C E A G How many steps are there in this reaction? 1 Practice Questions D B F C E A G What is the highest-energy transitions state? Practice Questions D B F
Practice Questions Practice Questions D B F C E A G How many steps are there in this reaction? 1 Practice Questions D B F C E A G What is the highest-energy transitions state? Practice Questions D B F
Chapter 8 Alkenes and Alkynes II: Addition Reactions "
 Chapter 8 Alkenes and Alkynes II: Addition Reactions Additions to Alkenes Generally the reaction is exothermic because one π and one σ bond are converted to two σ bonds Alkenes are electron rich The π
Chapter 8 Alkenes and Alkynes II: Addition Reactions Additions to Alkenes Generally the reaction is exothermic because one π and one σ bond are converted to two σ bonds Alkenes are electron rich The π
Preparation of alkenes
 Lecture 11 אלקנים הכנה ותגובות של אלקנים: הידרוגנציה, סיפוח הידרוהלוגנים )כלל מארקובניקוב(, סיפוח הלוגנים והסטראוכימיה של תוצרי הסיפוח, הידרובורציה, אפוקסידציה, אוזונוליזה. 1 Preparation of alkenes 1.
Lecture 11 אלקנים הכנה ותגובות של אלקנים: הידרוגנציה, סיפוח הידרוהלוגנים )כלל מארקובניקוב(, סיפוח הלוגנים והסטראוכימיה של תוצרי הסיפוח, הידרובורציה, אפוקסידציה, אוזונוליזה. 1 Preparation of alkenes 1.
1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound?
 CEM 331: Chapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N C 2 C N C 2 C N 1 2 3 4 1: three sigma bonds and
CEM 331: Chapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N C 2 C N C 2 C N 1 2 3 4 1: three sigma bonds and
75. A This is a Markovnikov addition reaction. In these reactions, the pielectrons in the alkene act as a nucleophile. The strongest electrophile will
 71. B SN2 stands for substitution nucleophilic bimolecular. This means that there is a bimolecular rate-determining step. Therefore, the reaction will follow second-order kinetics based on the collision
71. B SN2 stands for substitution nucleophilic bimolecular. This means that there is a bimolecular rate-determining step. Therefore, the reaction will follow second-order kinetics based on the collision
LECTURE #14 Thurs., Oct.20, Midterm exam: Tues.Oct.25 during class Ch.1, , 7.10, 2, Sections
 CHEM 221 section 01 LECTURE #14 Thurs., Oct.20, 2005 Midterm exam: Tues.Oct.25 during class Ch.1, 7.2-7.5, 7.10, 2, 3.1-3.5 ASSIGNED READINGS: TODAY S CLASS: NEXT LECTURE: Sections 4.7-4.10 finish Ch.4,
CHEM 221 section 01 LECTURE #14 Thurs., Oct.20, 2005 Midterm exam: Tues.Oct.25 during class Ch.1, 7.2-7.5, 7.10, 2, 3.1-3.5 ASSIGNED READINGS: TODAY S CLASS: NEXT LECTURE: Sections 4.7-4.10 finish Ch.4,
NAME: STUDENT NUMBER: Page 1 of 4
 NAME: STUDENT NUMBER: Page 1 of 4 Chemistry 2.339 Midterm Test Friday ctober 28, 2005 This test is worth 40 Marks. You must complete all work within the class period. Put all answers in the space provided.
NAME: STUDENT NUMBER: Page 1 of 4 Chemistry 2.339 Midterm Test Friday ctober 28, 2005 This test is worth 40 Marks. You must complete all work within the class period. Put all answers in the space provided.
1. Addition of HBr to alkenes
 eactions of Alkenes I eading: Wade chapter 8, sections 8-1- 8-8 tudy Problems: 8-47, 8-48, 8-55, 8-66, 8-67, 8-70 Key Concepts and kills: Predict the products of additions to alkenes, including regiochemistry
eactions of Alkenes I eading: Wade chapter 8, sections 8-1- 8-8 tudy Problems: 8-47, 8-48, 8-55, 8-66, 8-67, 8-70 Key Concepts and kills: Predict the products of additions to alkenes, including regiochemistry
Chapter 8 Alkenes and Alkynes II: Addition Reactions. Alkenes are electron rich. Additions to Alkenes
 Additions to Alkenes hapter 8 Alkenes and Alkynes II: Addition eactions Generally the reaction is exothermic because one π and one σ bond are converted to two σ bonds Alkenes are electron rich The carbocation
Additions to Alkenes hapter 8 Alkenes and Alkynes II: Addition eactions Generally the reaction is exothermic because one π and one σ bond are converted to two σ bonds Alkenes are electron rich The carbocation
Chapter 9. Nucleophilic Substitution and ß-Elimination
 Chapter 9 Nucleophilic Substitution and ß-Elimination Nucleophilic Substitution Nucleophile: From the Greek meaning nucleus loving. A molecule or ion that donates a pair of electrons to another atom or
Chapter 9 Nucleophilic Substitution and ß-Elimination Nucleophilic Substitution Nucleophile: From the Greek meaning nucleus loving. A molecule or ion that donates a pair of electrons to another atom or
Organic Chemistry. Alkenes (2)
 For updated version, please click on http://ocw.ump.edu.my Organic Chemistry Alkenes (2) by Dr. Seema Zareen & Dr. Izan Izwan Misnon Faculty Industrial Science & Technology seema@ump.edu.my & iezwan@ump.edu.my
For updated version, please click on http://ocw.ump.edu.my Organic Chemistry Alkenes (2) by Dr. Seema Zareen & Dr. Izan Izwan Misnon Faculty Industrial Science & Technology seema@ump.edu.my & iezwan@ump.edu.my
Organic Halogen Compounds
 8 Organic alogen ompounds APTER SUMMARY 8.1 Introduction Although organic halogen compounds are rarely found in nature, they do have a variety of commercial applications including use as insecticides,
8 Organic alogen ompounds APTER SUMMARY 8.1 Introduction Although organic halogen compounds are rarely found in nature, they do have a variety of commercial applications including use as insecticides,
General Glossary. General Glossary
 General Glossary Absolute configuration The actual three-dimensional structure of a chiral molecule. Absolute configurations are specified verbally by the Cahn-Ingold-Prelog R,S convention and are represented
General Glossary Absolute configuration The actual three-dimensional structure of a chiral molecule. Absolute configurations are specified verbally by the Cahn-Ingold-Prelog R,S convention and are represented
COURSE OBJECTIVES / OUTCOMES / COMPETENCIES.
 COURSE OBJECTIVES / OUTCOMES / COMPETENCIES. By the end of the course, students should be able to do the following: See Test1-4 Objectives/Competencies as listed in the syllabus and on the main course
COURSE OBJECTIVES / OUTCOMES / COMPETENCIES. By the end of the course, students should be able to do the following: See Test1-4 Objectives/Competencies as listed in the syllabus and on the main course
REVIEW PROBLEMS Key. 1. Draw a complete orbital picture for the molecule shown below. Is this molecule chiral? Explain. H H.
 rganic hemistry II (E325) REVIEW PRBLEMS Key 1. Draw a complete orbital picture for the molecule shown below. Is this molecule chiral? Explain. 3 3 sp3 orbital p orbital sp2 orbital s orbital molecule
rganic hemistry II (E325) REVIEW PRBLEMS Key 1. Draw a complete orbital picture for the molecule shown below. Is this molecule chiral? Explain. 3 3 sp3 orbital p orbital sp2 orbital s orbital molecule
Organic Chemistry Lecture 2 - Hydrocarbons, Alcohols, Substitutions
 ALKANES Water-insoluble, low density C-C single bonds Higher MW -> higher BP, higher MP Branching -> lower BP, higher MP Forms cycloalkanes which can have ring strain Cyclohexane: chair vs. boat configuration
ALKANES Water-insoluble, low density C-C single bonds Higher MW -> higher BP, higher MP Branching -> lower BP, higher MP Forms cycloalkanes which can have ring strain Cyclohexane: chair vs. boat configuration
1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound?
 EM 331: hapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N 2 N 2 N 1 2 3 4 2. What hybrid orbitals are used to
EM 331: hapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N 2 N 2 N 1 2 3 4 2. What hybrid orbitals are used to
STEREOGENIC CENTER (Chiral Center,Asymmetric Center)
 STEREOGENI ENTER (hiral enter,asymmetric enter) Atom (usually carbon) to which 4 different groups are attached: W Z X Y Many, but not all, molecules which contain a stereogenic center are chiral. (A molecule
STEREOGENI ENTER (hiral enter,asymmetric enter) Atom (usually carbon) to which 4 different groups are attached: W Z X Y Many, but not all, molecules which contain a stereogenic center are chiral. (A molecule
Detailed Course Content
 Detailed Course Content Chapter 1: Carbon Compounds and Chemical Bonds The Structural Theory of Organic Chemistry 4 Chemical Bonds: The Octet Rule 6 Lewis Structures 8 Formal Charge 11 Resonance 14 Quantum
Detailed Course Content Chapter 1: Carbon Compounds and Chemical Bonds The Structural Theory of Organic Chemistry 4 Chemical Bonds: The Octet Rule 6 Lewis Structures 8 Formal Charge 11 Resonance 14 Quantum
Chem 232. Problem Set 9. Question 1. D. J. Wardrop
 Chem 232 D. J. Wardrop wardropd@uic.edu Problem Set 9 Question 1. a. Rank in order of increasing number of chirality centers (1 = fewest chirality centers; 5 = most chirality). 3 C C 3 b. Rank in order
Chem 232 D. J. Wardrop wardropd@uic.edu Problem Set 9 Question 1. a. Rank in order of increasing number of chirality centers (1 = fewest chirality centers; 5 = most chirality). 3 C C 3 b. Rank in order
Ether, NaOH. This is a two-step process, but is most commonly done as a one-pot procedure (upper right).
 CAPTE 8. ALKENES: EACTINS AND SYNTESIS ydration of Alkenes by xymercuration-eduction - concerted formation of three-membered ring intermediate - addition of water occurs through anti stereoselectivity
CAPTE 8. ALKENES: EACTINS AND SYNTESIS ydration of Alkenes by xymercuration-eduction - concerted formation of three-membered ring intermediate - addition of water occurs through anti stereoselectivity
CHEM 2312 practice final. Version - II
 EM 2312 practice final Version - II The following standardized final examination covers the entire introductory year of organic chemistry (EM 2311 and 2312 at Georgia Tech). The exam consists of 70 multiple
EM 2312 practice final Version - II The following standardized final examination covers the entire introductory year of organic chemistry (EM 2311 and 2312 at Georgia Tech). The exam consists of 70 multiple
Homework - Review of Chem 2310
 omework - Review of Chem 2310 Chapter 1 - Atoms and Molecules Name 1. What is organic chemistry? 2. Why is there an entire one year course devoted to the study of organic compounds? 3. Give 4 examples
omework - Review of Chem 2310 Chapter 1 - Atoms and Molecules Name 1. What is organic chemistry? 2. Why is there an entire one year course devoted to the study of organic compounds? 3. Give 4 examples
Essentials of Chapter 6
 Essentials of Chapter 6 (Videos: conformational Analysis, Conformational Analysis of Cycloalkanes, Chirality, /S Nomenclature [Basic Advanced], ptical Activity) A. Stereochemical Structures Wedge Bond
Essentials of Chapter 6 (Videos: conformational Analysis, Conformational Analysis of Cycloalkanes, Chirality, /S Nomenclature [Basic Advanced], ptical Activity) A. Stereochemical Structures Wedge Bond
OChem1 Old Exams. Chemistry 3719 Practice Exams
 OChem1 Old Exams Chemistry 3719 Practice Exams Fall 2017 Chemistry 3719 Practice Exam A1 This exam is worth 100 points out of a total of 600 points for Chemistry 3719/3719L. You have 50 minutes to complete
OChem1 Old Exams Chemistry 3719 Practice Exams Fall 2017 Chemistry 3719 Practice Exam A1 This exam is worth 100 points out of a total of 600 points for Chemistry 3719/3719L. You have 50 minutes to complete
Double and Triple Bonds. The addition of an electrophile and a
 Chapter 11 Additions to Carbon-Carbon Double and Triple Bonds The addition of an electrophile and a nucleophile to a C-C C double or triple bonds 11.1 The General Mechanism Pi electrons of the double bond
Chapter 11 Additions to Carbon-Carbon Double and Triple Bonds The addition of an electrophile and a nucleophile to a C-C C double or triple bonds 11.1 The General Mechanism Pi electrons of the double bond
Alkenes are prepared by the reverse of the electrophilic reactions in the last chapter. That is, they are prepared by elimination reactions.
 Ch 8 Alkene Reactions and Syntheses Alkenes are prepared by the reverse of the electrophilic reactions in the last chapter. That is, they are prepared by elimination reactions. Dehydrohalogenation - Mechanism
Ch 8 Alkene Reactions and Syntheses Alkenes are prepared by the reverse of the electrophilic reactions in the last chapter. That is, they are prepared by elimination reactions. Dehydrohalogenation - Mechanism
Chapter 15: Conjugated Systems, Orbital Symmetry, and UV Spectroscopy
 Chapter 15: Conjugated Systems, Orbital Symmetry, and UV Spectroscopy Conjugated unsaturated systems have a p orbital on a carbon adjacent to a double bond The p orbital can come from another double (e.g.
Chapter 15: Conjugated Systems, Orbital Symmetry, and UV Spectroscopy Conjugated unsaturated systems have a p orbital on a carbon adjacent to a double bond The p orbital can come from another double (e.g.
Halogenation Of Alkenes
 Kevin Burgess, December 4, 2016 1 alogenation Of Alkenes from chapter(s) in the recommended text A. Introduction Kevin Burgess, December 4, 2016 2 B. Mechanism parallel polarized positively is Chlorination
Kevin Burgess, December 4, 2016 1 alogenation Of Alkenes from chapter(s) in the recommended text A. Introduction Kevin Burgess, December 4, 2016 2 B. Mechanism parallel polarized positively is Chlorination
CHEM 261 HOME WORK Lecture Topics: MODULE 1: The Basics: Bonding and Molecular Structure Text Sections (N0 1.9, 9-11) Homework: Chapter 1:
 CHEM 261 HOME WORK Lecture Topics: MODULE 1: The Basics: Bonding and Molecular Structure Atomic Structure - Valence Electrons Chemical Bonds: The Octet Rule - Ionic bond - Covalent bond How to write Lewis
CHEM 261 HOME WORK Lecture Topics: MODULE 1: The Basics: Bonding and Molecular Structure Atomic Structure - Valence Electrons Chemical Bonds: The Octet Rule - Ionic bond - Covalent bond How to write Lewis
Chapter 7 Cyclic Compounds. Stereochemistry of Reactions
 Instructor Supplemental Solutions to Problems 2010 Roberts and Company Publishers Chapter 7 Cyclic Compounds. Stereochemistry of Reactions Solutions to In-Text Problems 7.3 Following the procedure in the
Instructor Supplemental Solutions to Problems 2010 Roberts and Company Publishers Chapter 7 Cyclic Compounds. Stereochemistry of Reactions Solutions to In-Text Problems 7.3 Following the procedure in the
Loudon Chapter 7 Review: Cyclic Compounds Jacquie Richardson, CU Boulder Last updated 8/24/2017
 Compounds with a single ring are monocyclic. For example: Assuming they have no double or triple bonds, they each have one degree of unsaturation. This means that their formulas follow the pattern C nh
Compounds with a single ring are monocyclic. For example: Assuming they have no double or triple bonds, they each have one degree of unsaturation. This means that their formulas follow the pattern C nh
Course Outline For: Organic Chemistry I (CHM 270) Credits: 5 Contact Hours: Lecture: 3 Lab: 4
 Course Outline For: Organic Chemistry I (CHM 270) Credits: 5 Contact Hours: Lecture: 3 Lab: 4 NOTE on Laboratory: Both Lecture and Laboratory must be taken simultaneously; separate grades will not be given
Course Outline For: Organic Chemistry I (CHM 270) Credits: 5 Contact Hours: Lecture: 3 Lab: 4 NOTE on Laboratory: Both Lecture and Laboratory must be taken simultaneously; separate grades will not be given
CHEM 302 Organic Chemistry I Problem Set VII Chapter 7 Answers
 CEM 302 rganic Chemistry Problem Set V Chapter 7 Answers 1) The pk a values of acetic acid and trifluroacetic acid are 4.5 and 0.9 respectively. Based on this information, which anion, C 3 C - or CF 3
CEM 302 rganic Chemistry Problem Set V Chapter 7 Answers 1) The pk a values of acetic acid and trifluroacetic acid are 4.5 and 0.9 respectively. Based on this information, which anion, C 3 C - or CF 3
Organic Chemistry I Lesson Objectives, Lesson Problems, Course Outline Spring 2008
 Organic Chemistry I Lesson Objectives, Lesson Problems, Course Outline Spring 2008 Lesson Date Assignment Lesson Objective Description Lesson Problems 4 14-Jan Chapter 1 Quiz Describe how bond polarity
Organic Chemistry I Lesson Objectives, Lesson Problems, Course Outline Spring 2008 Lesson Date Assignment Lesson Objective Description Lesson Problems 4 14-Jan Chapter 1 Quiz Describe how bond polarity
Chapter 10 Lecture Outline
 Organic Chemistry, Fifth Edition Janice Gorzynski Smith University of Hawai i Chapter 10 Lecture Outline Modified by Dr. Juliet Hahn Copyright 2017 McGraw-Hill Education. All rights reserved. No reproduction
Organic Chemistry, Fifth Edition Janice Gorzynski Smith University of Hawai i Chapter 10 Lecture Outline Modified by Dr. Juliet Hahn Copyright 2017 McGraw-Hill Education. All rights reserved. No reproduction
Chem ORGANIC CHEMISTRY I
 ORGANIC CHEMISTRY I CHEM 221 /2 01 Final Examination December 20, 2005 1400-1700 Dr. Cerrie ROGERS x x molecular model kits (without instructions) programmable calculators must be reset Chem 221 --- ORGANIC
ORGANIC CHEMISTRY I CHEM 221 /2 01 Final Examination December 20, 2005 1400-1700 Dr. Cerrie ROGERS x x molecular model kits (without instructions) programmable calculators must be reset Chem 221 --- ORGANIC
Suggested solutions for Chapter 19
 s for Chapter 19 19 PRBLEM 1 Predict the orientation of addition to these alkenes. Simple examples of addition with regioselectivity. The first and last alkenes have different numbers of substituents at
s for Chapter 19 19 PRBLEM 1 Predict the orientation of addition to these alkenes. Simple examples of addition with regioselectivity. The first and last alkenes have different numbers of substituents at
p Bonds as Nucleophiles
 Chapter 8 p Bonds as Nucleophiles REACTIONS OF ALKENES, ALKYNES, DIENES, AND ENOLS Copyright 2018 by Nelson Education Limited 1 8.2.1 Orbital structure of alkenes Geometry: Electrostatic potential: Electron-rich
Chapter 8 p Bonds as Nucleophiles REACTIONS OF ALKENES, ALKYNES, DIENES, AND ENOLS Copyright 2018 by Nelson Education Limited 1 8.2.1 Orbital structure of alkenes Geometry: Electrostatic potential: Electron-rich
Chapter 8 Alkenes: Reactions
 Chamras Glendale Community College Organic Chemistry 105 Chapter 8 Alkenes: Reactions Name Type 1. alo-hydrogenation (Ionic & Radical) Electrophilic Addition 2. ydration (Acid-Catalyzed) Electrophilic
Chamras Glendale Community College Organic Chemistry 105 Chapter 8 Alkenes: Reactions Name Type 1. alo-hydrogenation (Ionic & Radical) Electrophilic Addition 2. ydration (Acid-Catalyzed) Electrophilic
There are several possible arrangements for a molecule which contains two double bonds (diene): 1. Isolated: (two or more single bonds between them)
 1 Chapter 15: Conjugation and Reactions of Dienes I. Introduction to Conjugation There are several possible arrangements for a molecule which contains two double bonds (diene): 1. Isolated: (two or more
1 Chapter 15: Conjugation and Reactions of Dienes I. Introduction to Conjugation There are several possible arrangements for a molecule which contains two double bonds (diene): 1. Isolated: (two or more
STEREOGENIC CENTER (Chiral Center,Asymmetric Center) Atom (usually carbon) to which 4 different groups are attached: W Z C X Y
 STEREOGENI ENTER (hiral enter,asymmetric enter) Atom (usually carbon) to which 4 different groups are attached: W Z X Y Many, but not all, molecules which contain a stereogenic center are chiral. (A molecule
STEREOGENI ENTER (hiral enter,asymmetric enter) Atom (usually carbon) to which 4 different groups are attached: W Z X Y Many, but not all, molecules which contain a stereogenic center are chiral. (A molecule
Chapter 4. Reactions of alkenes. Addition reactions Carbocations Selectivity of reactions
 Chapter 4 Reactions of alkenes Addition reactions Carbocations Selectivity of reactions Prob 47 p192. Give the reagents that would be required (including catalyst). Ch 4 #2 Electrophilic addition Ch 4
Chapter 4 Reactions of alkenes Addition reactions Carbocations Selectivity of reactions Prob 47 p192. Give the reagents that would be required (including catalyst). Ch 4 #2 Electrophilic addition Ch 4
C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2
 C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2 CHM 321: Summary of Important Concepts Concepts for Chapter 7: Substitution Reactions I. Nomenclature of
C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2 CHM 321: Summary of Important Concepts Concepts for Chapter 7: Substitution Reactions I. Nomenclature of
Chapter 8 - Alkenes and Alkynes II Addition Reactions of Alkenes - Electrons in the π bond of alkenes react with electrophiles
 Andrew Rosen Chapter 8 - Alkenes and Alkynes II 8.1 - Addition Reactions of Alkenes - Electrons in the π bond of alkenes react with electrophiles 8.2 - Electrophilic Addition of Hydrogen Halides to Alkenes:
Andrew Rosen Chapter 8 - Alkenes and Alkynes II 8.1 - Addition Reactions of Alkenes - Electrons in the π bond of alkenes react with electrophiles 8.2 - Electrophilic Addition of Hydrogen Halides to Alkenes:
6.10 Acid-Catalyzed Hydration of Alkenes. Acid-Catalyzed Hydration of Alkenes
 Acid-atalyzed ydration of Alkenes 6.10 Acid-atalyzed ydration of Alkenes O O reaction is acid catalyzed; typical hydration medium is 50% 2 SO 4-50% 2 O Follows Markovnikov's Rule Follows Markovnikov's
Acid-atalyzed ydration of Alkenes 6.10 Acid-atalyzed ydration of Alkenes O O reaction is acid catalyzed; typical hydration medium is 50% 2 SO 4-50% 2 O Follows Markovnikov's Rule Follows Markovnikov's
Chemistry 35 Exam 2 Answers - April 9, 2007
 Chemistry 35 Exam 2 Answers - April 9, 2007 Problem 1. (14 points) Provide the products for the reactions shown below. If stereochemistry is important, be sure to indicate stereochemistry clearly. If no
Chemistry 35 Exam 2 Answers - April 9, 2007 Problem 1. (14 points) Provide the products for the reactions shown below. If stereochemistry is important, be sure to indicate stereochemistry clearly. If no
Organic Chemistry I: Reactions and Overview
 Organic Chemistry I: Reactions and Overview Andrew Rosen Editor: Raghav Malik January 13, 2013 Contents I Library of Synthetic Reactions 3 II Organic Trends and Essentials 4 1 The Basics: Bonding and Molecular
Organic Chemistry I: Reactions and Overview Andrew Rosen Editor: Raghav Malik January 13, 2013 Contents I Library of Synthetic Reactions 3 II Organic Trends and Essentials 4 1 The Basics: Bonding and Molecular
1. Use appropriate curved arrows to indicate the complete mechanism of each of these reactions. KH (1 equiv.) + KCl THF. + HBr.
 1. Use appropriate curved arrows to indicate the complete mechanism of each of these reactions. K (1 equiv.) TF K 3 2 2 3 enantiomer While writing the mechanism, justify both the regiochemistry the relative
1. Use appropriate curved arrows to indicate the complete mechanism of each of these reactions. K (1 equiv.) TF K 3 2 2 3 enantiomer While writing the mechanism, justify both the regiochemistry the relative
CHEM 2311 HW1. COMPLETELY FILL THE BOXES IN DARK PENCIL, i.e.:, NOT or or STRUCTURE
 EM 2311 W1 Provide answers to the following multiple choice questions on the Scantron form provided in lecture. Make sure that you put your name and bubble in your GTID # on the answer form. MPLETELY FILL
EM 2311 W1 Provide answers to the following multiple choice questions on the Scantron form provided in lecture. Make sure that you put your name and bubble in your GTID # on the answer form. MPLETELY FILL
Chem 3719 Example Exams. Chemistry 3719 Practice Exams
 Chem 3719 Example Exams Chemistry 3719 Practice Exams Fall 2018 Chemistry 3719, Fall 2017 Exam 1 Student Name: Y Number: This exam is worth 100 points out of a total of 700 points for Chemistry 3719/3719L.
Chem 3719 Example Exams Chemistry 3719 Practice Exams Fall 2018 Chemistry 3719, Fall 2017 Exam 1 Student Name: Y Number: This exam is worth 100 points out of a total of 700 points for Chemistry 3719/3719L.
More tutorial at
 Answer all the questions. 1) (12 pts) Assign (R) or (S) to all the chiral centers in the following molecules and mark the priority of each group around a chiral center (just the top chiral carbon in molecule
Answer all the questions. 1) (12 pts) Assign (R) or (S) to all the chiral centers in the following molecules and mark the priority of each group around a chiral center (just the top chiral carbon in molecule
c. Cl H Page 1 of 7 major P (E > Z and more substituted over less substituted alkene) LG must be axial are the same Cl -
 CEM 109A 1. Predict the products of the following reactions (a-c E2, d-f E1 KEY focuses only on elimination products, in most cases there will also be substitution products.) a. - LG must be axial - are
CEM 109A 1. Predict the products of the following reactions (a-c E2, d-f E1 KEY focuses only on elimination products, in most cases there will also be substitution products.) a. - LG must be axial - are
Week 4. Even harder stuff!
 Week 4 Even harder stuff! Focus: SN1 and SN2 Two organic reactions Learn about two basic pathways for how these reactions happen Focus on stereochemistry Focus: SN1 and SN2 You need a couple of things
Week 4 Even harder stuff! Focus: SN1 and SN2 Two organic reactions Learn about two basic pathways for how these reactions happen Focus on stereochemistry Focus: SN1 and SN2 You need a couple of things
Alkyl Halides. Alkyl halides are a class of compounds where a halogen atom or atoms are bound to an sp 3 orbital of an alkyl group.
 Alkyl Halides Alkyl halides are a class of compounds where a halogen atom or atoms are bound to an sp 3 orbital of an alkyl group. CHCl 3 (Chloroform: organic solvent) CF 2 Cl 2 (Freon-12: refrigerant
Alkyl Halides Alkyl halides are a class of compounds where a halogen atom or atoms are bound to an sp 3 orbital of an alkyl group. CHCl 3 (Chloroform: organic solvent) CF 2 Cl 2 (Freon-12: refrigerant
Chem HH W13-Notes Dr. Masato Koreeda - Page 1 of 13 Date: February 20, 2013
 hem 215-216 W13-otes Dr. Masato Koreeda - Page 1 of 13 Date: February 20, 2013 hapter 13. Alcohols, Diols, and Ethers verview: hemistry and reactions of sp 3 oxygen groups, particularly oxidation of an
hem 215-216 W13-otes Dr. Masato Koreeda - Page 1 of 13 Date: February 20, 2013 hapter 13. Alcohols, Diols, and Ethers verview: hemistry and reactions of sp 3 oxygen groups, particularly oxidation of an
CHEM J-10 June The structure of ( )-linalool, a commonly occurring natural product, is shown below.
 CEM1102 2014-J-10 June 2014 The structure of ( )-linalool, a commonly occurring natural product, is shown below. 4 What is the molecular formula of ( )-linalool? C 10 18 O Which of the following best describes
CEM1102 2014-J-10 June 2014 The structure of ( )-linalool, a commonly occurring natural product, is shown below. 4 What is the molecular formula of ( )-linalool? C 10 18 O Which of the following best describes
CHEM 2312 practice final. Version I
 CEM 2312 practice final Version The following standardized final examination covers the entire introductory year of organic chemistry (CEM 2311 and 2312 at Georgia Tech). The exam consists of 70 multiple
CEM 2312 practice final Version The following standardized final examination covers the entire introductory year of organic chemistry (CEM 2311 and 2312 at Georgia Tech). The exam consists of 70 multiple
Chapter 7. dehydration
 hapter 7 7.1 ne problem with elimination reactions is that mixtures of products are often formed. For example, treatment of 2-bromo-2-methylbutane with K in ethanol yields a mixture of two alkene products.
hapter 7 7.1 ne problem with elimination reactions is that mixtures of products are often formed. For example, treatment of 2-bromo-2-methylbutane with K in ethanol yields a mixture of two alkene products.
Chapter 11: Nucleophilic Substitution and Elimination Walden Inversion
 hapter 11: Nucleophilic Substitution and Elimination Walden Inversion (S)-(-) Malic acid [a] D = -2.3 Ag 2, 2 Pl 5 l Ag 2, 2 ()-2-hlorosuccinic acid l (-)-2-hlorosuccinic acid Pl 5 ()-() Malic acid [a]
hapter 11: Nucleophilic Substitution and Elimination Walden Inversion (S)-(-) Malic acid [a] D = -2.3 Ag 2, 2 Pl 5 l Ag 2, 2 ()-2-hlorosuccinic acid l (-)-2-hlorosuccinic acid Pl 5 ()-() Malic acid [a]
Basic Organic Chemistry Course code : CHEM (Pre-requisites : CHEM 11122)
 Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
Preparation of Alkyl Halides, R-X. Reaction of alkanes with Cl 2 & Br 2 (F 2 is too reactive, I 2 is unreactive): R + X X 2.
 Preparation of Alkyl alides, R-X Reaction of alkanes with Cl 2 & Br 2 (F 2 is too reactive, I 2 is unreactive): UV R + X 2 R X or heat + X This mechanism involves a free radical chain reaction. A chain
Preparation of Alkyl alides, R-X Reaction of alkanes with Cl 2 & Br 2 (F 2 is too reactive, I 2 is unreactive): UV R + X 2 R X or heat + X This mechanism involves a free radical chain reaction. A chain
Organic Chemistry CHM 314 Dr. Laurie S. Starkey, Cal Poly Pomona Alkyl Halides: Substitution Reactions - Chapter 6 (Wade)
 rganic Chemistry CM 314 Dr. Laurie S. Starkey, Cal Poly Pomona Alkyl alides: Substitution Reactions - Chapter 6 (Wade) Chapter utline I. Intro to RX (6-1 - 6-7) II. Substitution Reactions A) S N 2 (6-8,
rganic Chemistry CM 314 Dr. Laurie S. Starkey, Cal Poly Pomona Alkyl alides: Substitution Reactions - Chapter 6 (Wade) Chapter utline I. Intro to RX (6-1 - 6-7) II. Substitution Reactions A) S N 2 (6-8,
1. Radical Substitution on Alkanes. 2. Radical Substitution with Alkenes. 3. Electrophilic Addition
 1. Radical Substitution on Alkanes Only Cl and Br are useful at the laboratory level. Alkane reactivity: tertiary > secondary > primary > methyl Numbers below products give their relative yield. Relative
1. Radical Substitution on Alkanes Only Cl and Br are useful at the laboratory level. Alkane reactivity: tertiary > secondary > primary > methyl Numbers below products give their relative yield. Relative
Time: 3 hours (180 minutes) Marking Scheme For The Exam
 hemistry 2320 S10 Fall 2004 FINAL EXAM Thursday, ecember 23, 2004 Name: Student Number This paper consists of 13 pages (10 questions) (including this title page). Ensure that you have a complete paper
hemistry 2320 S10 Fall 2004 FINAL EXAM Thursday, ecember 23, 2004 Name: Student Number This paper consists of 13 pages (10 questions) (including this title page). Ensure that you have a complete paper
CH 334. Tuesday, December 5, Name KEY
 CH 334 Final Exam Tuesday, December 5, 2017 Name KEY You may use model kits but no other material with chemical information without instructor approval. Tables of possibly useful data are included on the
CH 334 Final Exam Tuesday, December 5, 2017 Name KEY You may use model kits but no other material with chemical information without instructor approval. Tables of possibly useful data are included on the
Electrophilic Addition
 . Reactivity of = Electrons in pi bond are loosely held. Electrophiles are attracted to the pi electrons. arbocation intermediate forms. Nucleophile adds to the carbocation. Net result is addition to the
. Reactivity of = Electrons in pi bond are loosely held. Electrophiles are attracted to the pi electrons. arbocation intermediate forms. Nucleophile adds to the carbocation. Net result is addition to the
Hydrogen iodide is a strong acid and will drive the reverse reaction, meaning the forward reaction will not occur.
 EM 261 Oct 18, 2018 Photosynthesis and Related Reactions O 2 2 O 6 12 O 6 2 O N 3, S, Fe, u, o, other Natural Products D-Glucose R O R OR Ionic substitution S N 1 & R X X 2 hv Petroleum/ Alkanes R N 2
EM 261 Oct 18, 2018 Photosynthesis and Related Reactions O 2 2 O 6 12 O 6 2 O N 3, S, Fe, u, o, other Natural Products D-Glucose R O R OR Ionic substitution S N 1 & R X X 2 hv Petroleum/ Alkanes R N 2
CHEM 263 Oct 25, stronger base stronger acid weaker acid weaker base
 CEM 263 ct 25, 2016 Reactions and Synthesis (Preparation) of R- Breaking the - Bond of R- with Metals R + Li 0 or Na 0 or K 0 metal R Li + 1/2 2 alkoxide Breaking the - Bond of R- by Acid-Base Reaction
CEM 263 ct 25, 2016 Reactions and Synthesis (Preparation) of R- Breaking the - Bond of R- with Metals R + Li 0 or Na 0 or K 0 metal R Li + 1/2 2 alkoxide Breaking the - Bond of R- by Acid-Base Reaction
Part I. Multiple choice. (4 points each.) Choose the one best answer and mark your answer on the ScanTron sheet.
 Test 3 Chemistry 2321 April 25, 2003 McMurry, Chapters 9-11 103 Total Points Name Please Print Part I. Multiple choice. (4 points each.) Choose the one best answer and mark your answer on the ScanTron
Test 3 Chemistry 2321 April 25, 2003 McMurry, Chapters 9-11 103 Total Points Name Please Print Part I. Multiple choice. (4 points each.) Choose the one best answer and mark your answer on the ScanTron
Elimination Reactions The E2 Mechanism
 Elimination Reactions The E2 Mechanism The E2 Mechanism X X- B: B- δ- B:- δ+ R 1 δ- R 2 δ+ X δ- The E2 Mechanism R 3 R 4 transition state Free energy (G) Eact B:- B R 1 R 2 X R 1 R 2 R 3 R 4 R 4 R 3 X:-
Elimination Reactions The E2 Mechanism The E2 Mechanism X X- B: B- δ- B:- δ+ R 1 δ- R 2 δ+ X δ- The E2 Mechanism R 3 R 4 transition state Free energy (G) Eact B:- B R 1 R 2 X R 1 R 2 R 3 R 4 R 4 R 3 X:-
Organic Chemistry 1 CHM 2210 Exam 2 (October 10, 2001)
 Organic Chemistry 1 CM 2210 Exam 2 (October 10, 2001) Name (print): _ Signature: _ Student ID Number: _ There are 10 multiple choice problems (4 points each) on this exam. Record the answers to the multiple
Organic Chemistry 1 CM 2210 Exam 2 (October 10, 2001) Name (print): _ Signature: _ Student ID Number: _ There are 10 multiple choice problems (4 points each) on this exam. Record the answers to the multiple
Organic Chemistry, Fifth Edition
 Organic Chemistry, Fifth Edition Janice Gorzynski Smith Modified by Dr. Juliet Hahn Chapter 4 Alkanes Copyright 2017 McGraw-Hill Education. All rights reserved. No reproduction or distribution without
Organic Chemistry, Fifth Edition Janice Gorzynski Smith Modified by Dr. Juliet Hahn Chapter 4 Alkanes Copyright 2017 McGraw-Hill Education. All rights reserved. No reproduction or distribution without
11/5/ Conjugated Dienes. Conjugated Dienes. Conjugated Dienes. Heats of Hydrogenation
 8.12 Sites of unsaturation Many compounds have numerous sites of unsaturation If sites are well separated in molecule they react independently If sites are close together they may interact with one another
8.12 Sites of unsaturation Many compounds have numerous sites of unsaturation If sites are well separated in molecule they react independently If sites are close together they may interact with one another
Organic Chemistry, Second Edition. Janice Gorzynski Smith University of Hawai i. Chapter 10 Alkenes
 Organic Chemistry, Second Edition Janice Gorzynski Smith University of Hawai i Chapter 10 Alkenes Prepared by Rabi Ann Musah State University of New York at Albany Copyright The McGraw-Hill Companies,
Organic Chemistry, Second Edition Janice Gorzynski Smith University of Hawai i Chapter 10 Alkenes Prepared by Rabi Ann Musah State University of New York at Albany Copyright The McGraw-Hill Companies,
HO HO. 1) BH 3 HCl. + enantiomer either one may be drawn 4. + enantiomer either one may be drawn 4 1) O 3
 . (0 points) Page 1 A. Reaction analysis: provide the requested information in each of the following chemical transformations. how all missing starting materials and products unless otherwise indicated,
. (0 points) Page 1 A. Reaction analysis: provide the requested information in each of the following chemical transformations. how all missing starting materials and products unless otherwise indicated,
Enantiomers. nonsuperimposable mirror image Both Configuration will be opposite. Both Configuration will be opposite
 Optical Isomerism Isomerism of Organic Molecules: Two chiral centers Many organic compounds have more than one asymmetric carbon. The more asymmetric carbons a compound has, the more number of stereoisomers
Optical Isomerism Isomerism of Organic Molecules: Two chiral centers Many organic compounds have more than one asymmetric carbon. The more asymmetric carbons a compound has, the more number of stereoisomers
CHEM 2423 Unit 8 Homework Answers
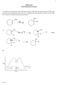 1 CEM 2423 Unit 8 omework Answers 1. Show the mechanism of the following reaction. Label the rate-limiting step. Draw the potential energy diagram for this reaction. (c) Explain the regiochemistry of this
1 CEM 2423 Unit 8 omework Answers 1. Show the mechanism of the following reaction. Label the rate-limiting step. Draw the potential energy diagram for this reaction. (c) Explain the regiochemistry of this
OChem 1 Mechanism Flashcards. Dr. Peter Norris, 2018
 OChem 1 Mechanism Flashcards Dr. Peter Norris, 2018 Mechanism Basics Chemical change involves bonds forming and breaking; a mechanism describes those changes using curved arrows to describe the electrons
OChem 1 Mechanism Flashcards Dr. Peter Norris, 2018 Mechanism Basics Chemical change involves bonds forming and breaking; a mechanism describes those changes using curved arrows to describe the electrons
Chapter 3 AN INTRODUCTION TO ORGANIC COMPOUNDS NOMENCLATURE, PHYSICAL PROPERTIES, REPRESENTATION OF STRUCTURE AND
 ORGANIC CHEMISTRY, 2 ND EDITION PAULA YURKANIS BRUICE Chapter 3 AN INTRODUCTION TO ORGANIC COMPOUNDS NOMENCLATURE, PHYSICAL PROPERTIES, AND REPRESENTATION OF STRUCTURE RAED M. AL-ZOUBI, ASSISTANT PROFESSOR
ORGANIC CHEMISTRY, 2 ND EDITION PAULA YURKANIS BRUICE Chapter 3 AN INTRODUCTION TO ORGANIC COMPOUNDS NOMENCLATURE, PHYSICAL PROPERTIES, AND REPRESENTATION OF STRUCTURE RAED M. AL-ZOUBI, ASSISTANT PROFESSOR
Organic Chemistry Practice Problems: Solutions
 rganic Chemistry Practice Problems: Solutions 1. 2. a. B, A b. D, B c. A, D d. D, A a. Resonance b. Electronegativity of fluorine atoms F F c. Neither is very acidic, but the oxygen will help stabilise
rganic Chemistry Practice Problems: Solutions 1. 2. a. B, A b. D, B c. A, D d. D, A a. Resonance b. Electronegativity of fluorine atoms F F c. Neither is very acidic, but the oxygen will help stabilise
Chapter 10 Radical Reactions"
 Chapter 10 Radical Reactions Radicals are intermediates with an unpaired electron H. Cl. Hydrogen radical t Often called free radicals What are radicals? Chlorine radical t Formed by homolytic bond cleavage
Chapter 10 Radical Reactions Radicals are intermediates with an unpaired electron H. Cl. Hydrogen radical t Often called free radicals What are radicals? Chlorine radical t Formed by homolytic bond cleavage
ELECTROPHILIC ADDITIONS OF ALKENES AS THE COUNTERPART OF ELIMINATIONS
 ELECTRPHILIC ADDITINS F ALKENES AS THE CUNTERPART F ELIMINATINS INTRDUCTIN - Chapter 8 is mostly about alkene reactions. That is, how one can transform alkenes into other functional groups. Most of these
ELECTRPHILIC ADDITINS F ALKENES AS THE CUNTERPART F ELIMINATINS INTRDUCTIN - Chapter 8 is mostly about alkene reactions. That is, how one can transform alkenes into other functional groups. Most of these
OChem1 Course Pack Practice Exams Practice Problems by Chapter Mechanism Flashcards
 Chem1 Course Pack Practice Exams Practice Problems by Chapter Mechanism Flashcards Chem1 Practice Exams Dr. Peter Norris, 2014 Chemistry 3719 Practice Exam A1 This exam is worth 100 points out of a total
Chem1 Course Pack Practice Exams Practice Problems by Chapter Mechanism Flashcards Chem1 Practice Exams Dr. Peter Norris, 2014 Chemistry 3719 Practice Exam A1 This exam is worth 100 points out of a total
NAME: SPRING 2015 MIDTERM
 page 1 pts NAME: SPRING 2015 MIDTERM hemistry 231 Professor: Dr. Gergens take-home portion (DUE at the beginning of the period, 4/6) Do your best on this take-home portion of your mid-term. I may grade
page 1 pts NAME: SPRING 2015 MIDTERM hemistry 231 Professor: Dr. Gergens take-home portion (DUE at the beginning of the period, 4/6) Do your best on this take-home portion of your mid-term. I may grade
CHEM 231 (Davis) Organic Chemistry FINAL EXAM May 15, YOUR NAME (Last, First, M.I.) DISCUSSION SECTION #53 (5 Points)
 CHEM 231 (Davis) rganic Chemistry FINAL EXAM May 15, 2006 YUR NAME (Last, First, M.I.) DISCUSSIN SECTIN #53 (5 Points) Initial of last name Instructions Please fill in your name in the space above and
CHEM 231 (Davis) rganic Chemistry FINAL EXAM May 15, 2006 YUR NAME (Last, First, M.I.) DISCUSSIN SECTIN #53 (5 Points) Initial of last name Instructions Please fill in your name in the space above and
3-chloro-1-propene 1-chloropropane 2-chloropropene
 ANSWERS #1. (from 50 minute exam #3, Fall 2000) 5. (6 points) For each group of 3 compounds, identify the compound that expresses the indicated property the MOST and the compound that expresses it the
ANSWERS #1. (from 50 minute exam #3, Fall 2000) 5. (6 points) For each group of 3 compounds, identify the compound that expresses the indicated property the MOST and the compound that expresses it the
Chemistry 210 Organic Chemistry I Summer Semester 1999 Dr. Somnath Sarkar
 Chemistry 210 rganic Chemistry I Summer Semester 1999 Dr. Somnath Sarkar Final Examination Name: Thursday, July 29, 1999, 6:30 8:30 Question 1 Stereochemistry(MC#1) 16 Question 2. Substitution(MC #2) 16
Chemistry 210 rganic Chemistry I Summer Semester 1999 Dr. Somnath Sarkar Final Examination Name: Thursday, July 29, 1999, 6:30 8:30 Question 1 Stereochemistry(MC#1) 16 Question 2. Substitution(MC #2) 16
Organic Chemistry Curriculum Content Outline
 Organic Chemistry 2014-15 Curriculum Content Outline CHEM 0203: Organic Structure and Reactivity 1. Structure & Bonding (Brief Review from General Chemistry) a. Ionic & Covalent Bonding b. Lewis Structures
Organic Chemistry 2014-15 Curriculum Content Outline CHEM 0203: Organic Structure and Reactivity 1. Structure & Bonding (Brief Review from General Chemistry) a. Ionic & Covalent Bonding b. Lewis Structures
Chemistry 123: Physical and Organic Chemistry Topic 1: Organic Chemistry
 Topic 1: Mechanisms and Curved Arrows etc Reactions of Alkenes:.Similar functional groups react the same way. Why? Winter 2009 Page 73 Topic 1: Mechanisms and Curved Arrows etc Reactivity:.Electrostatic
Topic 1: Mechanisms and Curved Arrows etc Reactions of Alkenes:.Similar functional groups react the same way. Why? Winter 2009 Page 73 Topic 1: Mechanisms and Curved Arrows etc Reactivity:.Electrostatic
Exam Analysis: Organic Chemistry, Midterm 1
 Exam Analysis: Organic Chemistry, Midterm 1 1) TEST BREAK DOWN: There are three independent topics covered in the first midterm, which are hybridization, structure and isomerism, and resonance. The test
Exam Analysis: Organic Chemistry, Midterm 1 1) TEST BREAK DOWN: There are three independent topics covered in the first midterm, which are hybridization, structure and isomerism, and resonance. The test
Chemistry 2030 Survey of Organic Chemistry Fall Semester 2014 Dr. Rainer Glaser
 Chemistry 2030 Survey of Organic Chemistry Fall Semester 2014 Dr. Rainer Glaser Examination #1 Bonding, Alkanes, Alkenes & Alkynes Thursday, September 18, 2014, 8:00 8:50 am Question 1. Atomic Structure
Chemistry 2030 Survey of Organic Chemistry Fall Semester 2014 Dr. Rainer Glaser Examination #1 Bonding, Alkanes, Alkenes & Alkynes Thursday, September 18, 2014, 8:00 8:50 am Question 1. Atomic Structure
ORGANIC - EGE 5E CH. 5 - ALKANES AND CYCLOALKANES.
 !! www.clutchprep.com CONCEPT: ALKANE NOMENCLATURE Before 1919, chemists literally had to memorize thousands of random (common) chemical names. IUPAC naming provides a systematic method to give every chemical
!! www.clutchprep.com CONCEPT: ALKANE NOMENCLATURE Before 1919, chemists literally had to memorize thousands of random (common) chemical names. IUPAC naming provides a systematic method to give every chemical
Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320
 Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320 I. Isolated, cumulated, and conjugated dienes A diene is any compound with two or C=C's is a diene. Compounds containing more than two
Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320 I. Isolated, cumulated, and conjugated dienes A diene is any compound with two or C=C's is a diene. Compounds containing more than two
CHEMISTRY 231 GENERAL ORGANIC CHEMISTRY I FALL 2014 List of Topics / Examination Schedule
 Page 1 of 5 CHEMISTRY 231 FALL 2014 List of Topics / Examination Schedule Unit Starts Topic of Study 20 Aug 2014 STRUCTURE AND BONDING Suggested Reading: Chapter 1 29 Aug 2014 ALKANES & CYCLOALKANES Suggested
Page 1 of 5 CHEMISTRY 231 FALL 2014 List of Topics / Examination Schedule Unit Starts Topic of Study 20 Aug 2014 STRUCTURE AND BONDING Suggested Reading: Chapter 1 29 Aug 2014 ALKANES & CYCLOALKANES Suggested
CH Organic Chemistry I (Katz) Practice Exam #3- Fall 2013
 C2710 - rganic Chemistry I (Katz) Practice Exam #3- all 2013 Name: Score: Part I - Choose the best answer and write the letter of your choice in the space provided. (32 pts) 1. f the following, which reaction
C2710 - rganic Chemistry I (Katz) Practice Exam #3- all 2013 Name: Score: Part I - Choose the best answer and write the letter of your choice in the space provided. (32 pts) 1. f the following, which reaction
We can review the concepts we have dealt with above by considering as a
 Text Related to Segment 5.06 2002 Claude E. Wintner We can review the concepts we have dealt with above by considering as a final exercise the dimethylcyclohexanes. First treating the problem as regards
Text Related to Segment 5.06 2002 Claude E. Wintner We can review the concepts we have dealt with above by considering as a final exercise the dimethylcyclohexanes. First treating the problem as regards
