The Journal of Physiology
|
|
|
- August Webster
- 5 years ago
- Views:
Transcription
1 J Physiol (2012) pp Do calcium waves propagate between cells and synchronize alternating calcium release in rat ventricular myocytes? Y. Li, D. A. Eisner and S. C. O Neill Unit of Cardiac Physiology, Core Technology Facility, University of Manchester, 46 Grafton St, Manchester M13 9NT, UK The Journal of Physiology Key points In cell pairs isolated from rat ventricular muscle, spontaneous waves of Ca 2+ -induced Ca 2+ release are much more likely to propagate between cells connected side to side than end to end. Investigation of this difference using fluorescence techniques shows that the end-to-end connection probably contains the intercalated disc. It is this structure that accounts for the greater distance between sarcoplasmic reticulum Ca 2+ release units being so large (about 2.3μm). At side-to-side connections, this distance is much less (about 1.6 μm). This variation may be the cause of the difference in propagation properties. In cell pairs isolated from rat ventricular muscle, we have been able to induce systolic Ca 2+ alternans in-phase between the cells in a pair. This is not due to propagation of Ca 2+ waves between cells. In some cases, wave propagation can interfere with synchrony. Abstract The aim was to investigate the propagation of Ca 2+ waves between cells and determine whether this synchronizes alternating Ca 2+ release between cells. Experiments were carried out on electrically coupled cell pairs; spontaneous Ca 2+ waveswereproducedbyelevatingexternal Ca 2+. There was a significant difference in the ability of these waves to propagate between cells depending on the orientation of the pairs. Although almost all pairs connected by side-to-side contacts showed propagating Ca 2+ release, this was very uncommon in end-to-end cell pairs. Confocal studies showed that there was a gap at the intercalated disc consisting of cell membranes and a region of cytoplasm devoid of sarcoplasmic reticulum. This gap was 2.3 μm in length and is suggested to interfere with Ca 2+ wave propagation. The gap measured was much smaller between side-to-side contacts: 1.5 μm and so much less likely to interfere with propagation. Subsequent experiments investigated the synchronization between cells of Ca 2+ alternans produced by small depolarizing pulses. Although this alternation results from beat-to-beat alternation of intracellular Ca 2+ wave propagation, there was no evidence that propagation of Ca 2+ waves between cells contributed to synchronization of this alternans. (Resubmitted 20 September 2012; accepted 17 October 2012; first published online 22 October 2012) Corresponding author D. A. Eisner: Division of Cardiovascular and Endocrine Sciences, Core Technology Facility (3rd Floor), University of Manchester, 46 Grafton Street, Manchester M13 9NT, UK. eisner@man.ac.uk Abbreviations CICR, Ca 2+ -induced Ca 2+ release; NCX, Na + Ca 2+ exchange; RyR, ryanodine receptor; SR, sarcoplasmic reticulum. To the sorrow of his colleagues, Stephen O Neill died on 9 August DOI: /jphysiol
2 6354 Y. Li and others J Physiol Introduction Ca 2+ release from the cardiac sarcoplasmic reticulum (SR) normally occurs by the process of Ca 2+ -induced Ca 2+ release (CICR) in which calcium entering the cell via the L-type Ca 2+ channel binds to the SR Ca 2+ release channel (ryanodine receptor, RyR) resulting in openingoftheryrandfurtherreleaseofca 2+ from thesr.underconditionswhenthecellandsrareoverloaded with Ca 2+, release from the SR can occur in the absence of the triggering L-type channel. This occurs as a wave of CICR that propagates along the cell. These Ca 2+ waves activate the electrogenic Na + Ca 2+ exchange (NCX) and the resulting depolarization has been shown to trigger arrhythmias (see Venetucci et al for review). Given that Ca 2+ waves can propagate along a cell, it is of interest to know whether this can also occur via gap junctions between cells. In several studies Ca 2+ waves pass between cells although rather unreliably (Takamatsu et al. 1991; Minamikawa et al. 1997; Lamont et al. 1998; Miura et al. 1999; Kanekoet al. 2000; Klaukeet al. 2007). The major aim of this paper was therefore to investigate propagation ofwavesbetweencellsinapair. Recent work shows that propagation of Ca 2+ waves within a single cell can contribute to instability in controlling the amplitude of the systolic Ca 2+ transient. When cells are stimulated with small depolarizing pulses, there is a beat-to-beat alternation in the amplitude of the Ca 2+ transient. This arises because the larger responses involve propagation of Ca 2+ waves whereas propagation failsonthesmallerones(díaz et al. 2004; Li et al. 2009). As well as its relevance to the control of stability per se, this alternation of SR Ca 2+ release may play a role in the generation of pulsus alternans, which is a complication of heart failure that has a poor prognosis for the patient as it is strongly associated with sudden cardiac death (see Weiss et al for review). Given that propagation of Ca 2+ waves within a cell can result in alternans, the question arises as to whether propagation between cells can lead to co-ordinated alternans of multicellular structures, and answering this was the other aim of the paper. In this paper, we therefore attempt to define whether Ca 2+ waves can propagate between cells. We find that the ease with which Ca 2+ waves can propagate from cell to cell depends critically on whether the cells are connected side to side or end to end. In turn, the ease of propagation depends on the ultrastructure of the connections with the gap between SR Ca 2+ release units being critical. We also find that the propagation of Ca 2+ waves does not contribute to the synchronization of alternans between cells. Methods Cell pairs were isolated from male Wistar rats using a modification of a published method (Eisner et al. 1989). This modification involved adding bovine serum albumin (1 mg ml 1 ) to the isolation solution and increasing times in the enzyme solution. Rats were grown to a weight above 350 g before they were killed; at this weight the success ofisolatingcellpairswasgreaterthanatlowerweights. All procedures were in accordance with the Animals (Scientific Procedures) Act Voltage clamp experiments Cellswerebathedinacontrolsolution(inmM): NaCl 134, Hepes 10, glucose 11.1, MgCl 2 1.2, KCl 4 and CaCl 2 1. For voltage clamp experiments we used the perforated patch clamp technique (Rae et al. 1991) with the switch clamp mode of the Axoclamp 2A voltage clamp amplifier (Axon Instrument, Union City, CA, USA). Pipettes (<5M ) were filled with the following solution (mm): KCH 3 O 3 S 125, KCl 12, NaCl 10, Hepes 10, MgCl 2 5, EGTA 0.1; titrated to ph 7.2 with KOH; and a final concentration of amphotericin B of 240 μgml 1.ExternalCa 2+ was raised to 4 mm; 0.1 mm Ba 2+ and 5 mm 4-aminopyridine were added to block K + currents. Cells were stimulated by depolarizations from 40mV to the required level between 30 and 20 mv to produce alternans of systolic release. To measure intracellular Ca 2+, cells were loaded with the acetoxymethyl ester of fluo-3 (5 μm for 10 min at room temperature). Once a cell in a pair was patched, the voltage clamp was applied and measurement of images began. Light from the xenon arc lamp was filtered at 488 nm and fluorescence images (>500 nm) recorded at 30 Hz using a Luca EM Andor Camera System (Belfast, UK). Images were analysed using Andor software and ImageJ (rsbweb.nih.gov/ij/). Visualization of membranes and sarcoplasmic reticulum Visualization of cell membranes and SR was performed using a Leica SP2 confocal microscope. Cell pairs were incubated with Di-8 ANEPPS (5 μm for 5 min at 20 C) to show surface membranes and transverse tubules and ER-Tracker Green (500 nm for 30 min at 37 C) to show SR location. Both dyes were obtained from Invitrogen (Paisley, UK). Fluorescence from both dyes was excited at 488 nm. Emitted light was measured between 500 and 540 nm for ER-Tracker and 625 and 700 nm for Di-8 ANEPPS.
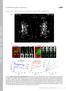
3 J Physiol Ca 2+ waves and synchronization of alternating systolic release 6355 Statistics All statistics reported are mean ± SEM. We have used Student s paired or unpaired t tests, chi squared or Mann Whitney tests, as appropriate. Results All data in this paper come from electrically coupled cell pairs. We tested for electrical coupling by voltage clamping onecellofapairanddepolarizingittodetermineifaca 2+ release took place in both cells. Figure 1 shows typical data recorded from a pair of cells connected side to side. Here cell 1 was voltage clamped with a voltage pulse to 0 mv from 40 mv. The systolic Ca 2+ transient was identical in both cells of the pair (Fig. 1B). Do calcium waves pass between cells? In this series of experiments, we investigated whether spontaneous Ca 2+ waves could propagate between cells. Experiments showed that the result depended on the physical relationship between the two cells. Some cells (e.g. Fig. 2A) were connected side to side and the actual degree of overlap of the lateral membranes was variable; others (e.g. Fig. 2B) were connected via their ends. The images in Fig. 2A show an example of a spontaneous wave of CICR passing from one cell to another via a side-to-side configuration junction. The wave begins in the lower part of the upper cell but very quickly passes to the lower cell with barely any detectable delay. However, wave propagation occurred only very rarely between cells connected end to end (e.g. Fig. 2B). In this case, two independent waves begin in each cell but both stop at the same point, presumably the intercalated disc between the cells. Summary data for this propagation behaviour are shown in the histogram in Fig. 2C. Fluorescence movies of the cell pairs in Fig. 2 can be seen in the supplementary data; these show the real-time propagation of CICR in these cells. In subsequent experiments, we investigated whether the probability of intercellular wave propagation in either configuration depended on the degree of Ca 2+ overload. The experiment in Fig. 3 shows recordings of Ca 2+ waves from regions of two cells connected side to side. In the Figure 1. Ca 2+ transients recorded from a pair of coupled cells Voltage clamping one cell effectively clamps both cells. A, image of cell pair in the side-to-side configuration. B, corresponding fluorescence traces taken from each of cells in the image. Depolarizing pulse was from 40 to 0 mv for 100 ms. Black trace is from cell 1 and red from cell 2. Figure 2. Wave propagation between cells A, cells connected side to side. Spontaneous Ca 2+ wave starts in the lower part of upper cell then spreads to the lower cell and propagates through both. The full-time course can be seen in Supplementary Movie 1. B, cells connected end to end. In this example, independent, spontaneous waves start in the two cells but do not propagate between them. The full-time course can be seen in Supplementary Movie 2. C, summary data showing proportion of cells showing propagation. Times under each image show when image was recorded.
4 6356 Y. Li and others J Physiol top panel of Fig. 3A the superfusion solution contained 1mMCa 2+. Here the first wave did not propagate whereas the second one did. In the lower panel, external Ca 2+ was increased to 6 mm. The most obvious effect is an increase in the frequency of Ca 2+ waves (Díaz et al. 1997). Here the first wave arose in the cell indicated with the dashed line and then propagated to the other. The third wave propagated in the other direction. The second wave arose at the same time in both cells. Figure 3B shows the fraction of waves that propagated during 66 s observation periods in six cells over a range of Ca 2+ from 1to6mM. There is no significant effect of external Ca 2+ concentration on the fraction of waves that propagate (one-way ANOVA). We performed similar experiments on seven cells connected end to end. In none of these cells did wave propagation occur at any of the Ca 2+ concentrations used (not shown). In these end-to-end cells, we also investigated whether potentiating the RyR would allow propagation. We found, however, that 100 μm caffeine Figure 3. Lack of effect of changing external Ca 2+ concentration on the probability of propagation of waves between side-to-side connected cells A, original data. Solid trace shows fluorescence from a region of interest in one cell and dashed from the other cell. Trace a was recorded in 1 mm Ca 2+ and b in 6 mm. B, mean probability of propagation as a function of external Ca 2+ concentration for six cell pairs. did not promote propagation. Similarly, beta-adrenergic stimulation with 1 μm isoprenaline did not result in propagation. Why does propagation fail in the end-to-end configuration? We investigated the reason for the different CICR wave propagation properties of the two pair configurations. Our hypothesis was that the intercalated disc formed a barrier to propagation of CICR preventing passage of the wave. We used ER-Tracker Green to show the distribution of SR and the sarcolemma was identified by the membrane-staining dye Di-8 ANEPPS. A sample (end-to-end) cell pair loaded with both dyes is illustrated in Fig. 4A. Thelowerpart of Fig. 4A shows the fluorescence from Di-8 ANEPPS indicating staining of the intercalated disc with other surface membranes also visible. The graph in Fig. 4Ba shows the profile of the stain intensity along the cell interior, from the white rectangle on the left, showing a sarcomeric pattern in both stains, with a periodicity of 1.81 ± 0.02 μm(n = 18). However, it is clear that the two stains are exactly out of phase with each other. This is to be expected as the membrane dye stains t-tubules and the ER dye shows the location of the SR; however, these two structures are never found in the same place. In the right-hand plots (Fig. 4Bb), we see the spatial location of fluorescence across the end-to-end junction in the cell pair showninthegreenrectanglesinthecentreoftheimages in Fig. 4A. Here the two dyes are again found in different parts of the images. The membrane dye shows the location of the intercalated disc, the SR is not associated with this part of the image. In Fig. 4C the region of membrane staining is smaller than the gap between cells stained with ER-Tracker; on average the gap between the SR staining had a half distance of 2.35 ± 0.19 μm (n = 12) and the membrane half distance was 1.48 ± 0.17 μm in the same cell pairs. This is a statistically significant difference (P = 0.001). Such an area lacking SR would be a barrier to propagation. Figure 5A shows that the side-to-side junction between cells is narrower than the intercalated disc observed in end-to-end junctions. Staining is quantified in Fig. 5B from the regions indicated by rectangles in Fig. 5A.Again, the membrane dye shows a narrower staining than the ER-Tracker dye. There is also a significant difference between end membranes and side-to-side membranes, i.e. the side-to-side membrane is thinner at 0.86 ± 0.08μm than the membrane at the end of the cell (1.48 ± 0.17 μm; Mann Whitney test P < 0.002). In addition, the gap measured using ER-Tracker is also significantly greater at the intercalated disc (2.35 ± 0.19 μm) as against the thickness measured at the side-to-side
5 J Physiol Ca 2+ waves and synchronization of alternating systolic release 6357 Figure 4. The area of the intercalated disc lacks sarcoplasmic reticulum A, images of ER-Tracker (above) and Di-8 ANEPPS (below). Ba, plots showing fluorescence measured from the region of interest marked by white rectangle along the sarcomeric pattern to the left in each image. b, the spatial profiles of fluorescence across the intercalated disc at the points indicated by the green rectangles. C, summary data for 12 cell pairs of gap widths at end-to-end junctions in both membrane and ER dyes. P = Figure 5. Thicknesses of membranes between cells A, Di-8 ANEPPS and ER-Tracker-labelled cell pair showing the measurements made of side to side. B, these plots show the fluorescence intensities for the two regions contained within rectangles on image above. NB The distances shown in B represent the vertical dimension in A. C, summary data for nine cell pairs, P =
6 6358 Y. Li and others J Physiol junctions (1.59 ± 0.14 μm; P = 0.006). We will discuss the appropriateness of our side-to-side membrane measurements in the discussion but the physiological significance of these measurements is clear: the ability of propagation to cross from side to side is very likely due to the much smaller gap between the SR units either side of the membranes. Alternans of systolic calcium release in cell pairs The aim of this series of experiments was to see to what extent the alternation in the amplitude of the systolic Ca 2+ transient produced by small depolarizing pulses was co-ordinated between cells in a pair, and whether there was evidence for propagation of Ca 2+ waves between cells playing a role. The data in Fig. 6 show that alternans occurs in cell pairs under appropriate conditions. Both panels show recordings from a pair of cells; on the left, the cells are connected end to end whereas on the right they are side to side. In both cases, one cell is patched and depolarized under conditions of slight Ca 2+ overload (4 mm external Ca 2+ concentration). In both cells in the pair, one stimulus produces a large release of calcium and the next a small one. The larger release also has a much longer duration than the smaller one (cf. Fig. 8) indicating wave propagation. In other words the alternans observed in cell pairs is very similartothatseenpreviouslyinsinglecells(díaz et al. 2004; Li et al. 2009) Is calcium passing through the gap junction required for alternans? The next series of experiments was designed to investigate whether movement of Ca 2+ across the gap junctions between cells was required to maintain Ca 2+ alternans in pairs of cells. One line of evidence casting doubt on an obligatory role for intercellular Ca 2+ waves in synchronizing alternans comes from data in Fig. 7. In this case the only Ca 2+ wave recorded propagated from cell 1intocell2duringtheCa 2+ transient marked with the asterisk (see also Supplementary Movie 3). However, this wave did not help enforce the phase of alternans rather it interferes with and decreases the magnitude of alternans in cell 2. Another means of testing this question is shown in Fig. 8. Here the alternans is in-phase as shown in Fig. 8B; these traces indicate changes of fluorescence taken from regions that occupy the majority of each of the two cells. However, images in Fig. 8A show how these traces arose. The coloured boxes are regions of interest shown in the same colour code as the traces in Fig. 8C. It is clear that each of the two waves that produce the large release initiates at sites that are not in contact with each other (i.e. the two regions cannot be expected to influence each other). In addition, the waves do not cross from one cell into the other. Figure 6. Alternans produced by small depolarizing pulses A and B, the upper panel shows an image of the cells and patch pipette and the bottom shows superimposed recordings of fluo-3 fluorescence from the two cells. A, a pair of cells connected end to end, stimulated by depolarizing pulses from 40 to 23 mv (bottom trace shows when each stimulus was applied). B, a pair of cells connected side to side stimulated by a pulse from 40 mv to 25 mv. The red trace is from cell 1 and black line from cell 2. Figure 7. Changing pattern of alternans in the same cell pair A, bright field image of the cell pair. B, fluorescence traces measured from cell 1 (Top) and from cell 2 (Bottom). Cell 1 shows classical systolic Ca 2+ alternans. However, cell 2 shows that when there is propagation of Ca 2+ -induced Ca 2+ release between the two cells (marked by the asterisk and in Supplementary Movie 3), this interferes with the alternans phase.
7 J Physiol Ca 2+ waves and synchronization of alternating systolic release 6359 What evidence is there that alternans is in-phase in cell pairs? Of the 14 cell pairs in which we observed alternans, nine were in-phase in each cell (i.e. as in Fig. 2); in two out of phase. Finally, in three cell pairs there was one cell alternating while the other cell was giving uniform systolic Ca 2+ transients. If we perform a chi-squared test to determine if there is a preference for in-phase as opposed to any other pattern of alternans the value we get is 1.92 (i.e. not sufficiently high enough to reject the null hypothesis that the alternans phase is random between the two cells in the pair; P = 0.18). Discussion In this paper, we have studied interactions between Ca 2+ wavesinpairsofcells.wefindthatca 2+ waves do not propagate between pairs of cells connected end to end but, in contrast, can propagate between pairs connected side to side. The lack of propagation between cells connected end to end is associated with the gap junction providing a substantial gap between the SR of the two cells. Furthermore, we find that changes of Ca 2+ in individual cells in a cell pair during systolic Ca 2+ alternans do not reinforce alternans of systolic Ca 2+ alternans in the other cell. Figure 8. A, fluorescence images of cells in a side-to-side pair Ca 2+ -induced Ca 2+ release initiation does not need to take place where cells meet. B, fluorescence traces taken from the cell pair shown in A. Each of the two cells is showing in-phase systolic alternans. C, fluorescence traces taken from the two coloured regions of interest shown in images in A. Both show clear wave propagation. Propagation of waves of calcium-induced calcium release between cells It has been previously reported in multicellular tissues that the probability of CICR waves passing between cells is low (Takamatsu et al. 1991; Minamikawa et al. 1997; Lamont et al. 1998; Miura et al. 1999; Kaneko et al. 2000; Klauke et al. 2007). In our study on isolated cell pairs we confirm the observation that end-to-end propagation is relatively unlikely (see Fig. 2) but, in contrast to previous work, we find that waves pass readily between cells arranged in the side-by-side configuration. We have investigated why this might be by looking at the junctions between cell pairs that can propagate waves and those that cannot. We have used the lipid marking dye Di-8 ANEPPS to show where membranes intervene between cells and find that the intercalateddiscbetweencellsformsabarrierabout1.5μm wide between end-to-end connected cells. Furthermore, by staining with ER-Tracker Green in the same cells to visualize SR, we find a gap about 2.3 μm wideinthe same location. Previous electron microscopic studies of the intercalated disc region show that the majority of gap junctions in that region are associated with sparse junctional SR (Forbes & Sperelakis, 1977, 1980). This suggests that around the intercalated disc region there ought to be a gap between cells where less SR is present. Our measurements suggest that this region extends for about 0.4 μm on either side of the disc region. The region will also include the thickness of the intercalated disc itself. On the other hand, at the side to-side junctions the gap due to the membrane is about 0.9 μm wide. The important point, however, is the gap between release units of the SR; we measure this as 1.6 μm. The location of the gap junction in these cell pairs is unknown as we do not measure Ca 2+ in these cells but the gap between SR release units seems to be within the distance that CICR can bridge. It is possible that it is the large length of the intercalated disc at end-to-end junctions that interferes with cell-to-cell propagation. Modelling of wave propagation predicts that propagation will fail when the sarcomere spacing is increased above normal sarcomere lengths (i.e. 1.9 μm). It is thought that propagation of spontaneous activity is prevented by the diffusion distance from one Ca 2+ release unit to the next (Chen-Izu et al. 2006; Izu et al. 2006). Our measurements show that the gap between SR Ca 2+ release units at the intercalated disc always exceeds this value (2.3μm); however, the SR gap between side-to-side contacts is always much less (1.6 μm). This could explain the lack of end-to-end propagation and the much higher probability of propagation in the side-to-side contacting cells. We cannot rule out, however, the possibility that other differences in the intercalated disc region contribute to preventing propagation. For example, it is possible that Ca 2+ buffering is greater in this region. Our measurements
8 6360 Y. Li and others J Physiol of the width of side-to-side contacts between cells are almost certainly an overestimate of the gap junction, as they will be affected by the finite depth of the images. However, the only important region for measurement is thegapjunction.atthispointthemembraneisverythin (10 20 nm) (Sommer & Waugh, 1976; Severs, 1990) and well below the spatial resolution of the light microscope. We have selected the thinnest portion of the side-to-side membrane we could find in all the examples we use. This may well not be the gap junction but it is still a lot thinner than the membrane at the intercalated disc region and, according to the models published for wave propagation (Chen-Izu et al. 2006; Izu et al. 2006), should offer little resistance to wave propagation. Low-voltage-induced alternans in cell pairs Stimulation of cell pairs with small depolarizing pulses results in alternans of the Ca 2+ transient. The mechanism of this alternans has previously been demonstrated (Díaz et al. 2004; Li et al. 2009). In particular (see Fig. 8) there are two phases of calcium release. The first of these represents direct CICR from the small L-type Ca 2+ current and the later phase is the propagating wave of CICR that leads to the larger of the two transients. Two lines of evidence lead us to conclude that propagation of CICR between cells is not required to maintain systolic Ca 2+ alternans. First, the fact that end-to-end cell pairs do not generally allow propagation of CICR but do allow in-phase alternans (see Fig. 6A). Secondly cell pairs can also show in-phase alternans when sites of initiation of the CICR waves are not close to the points of contact of the cells (see Fig. 8). If in-phase alternans between two cells in a pair is possible without propagation of CICR there remain only two other possible mechanisms: membrane potential/ membrane current or random chance. We know that conditions favouring small releases of Ca 2+ can lead to alternans. Thus, a small release can then trigger a larger propagating release; however, many cells do not show alternans in this situation. If the two cells are electrically effectively one (meaning that the small L-type Ca 2+ current stimulation of Ca 2+ release from the SR will be present in both cells) then it may simply be that both cells spontaneously begin to show systolic alternans at the same time. Statistical analysis of the data shows that there is no reason to believe that cells in a pair influence each other to produce systolic Ca 2+ alternans. Relationship of low-voltage-induced alternans to pulsus alternans Clinically, pulsus alternans involves the synchronization of many cells. The fact that alternans produced by low-voltage depolarization is not synchronized between cells suggests that it is unlikely to be the major mechanism of the clinical condition. This study has shown that systolic Ca 2+ alternans is present in cell pairs isolated from rat ventricular muscle. In addition, we find no strong evidence for any influence between cells in a cell pair that can alter the phase of systolic Ca 2+ alternans. We also report that propagation of CICR between cells is very frequent in side-to-side cell pairs and much less frequent between end-to-end pairs. We also propose that the intercalated disc, by excluding SR, prevents propagation of CICR while side-to-side junctions do not as the gap between SR units is much lower. References Chen-Izu Y, McCulle SL, Ward CW, Soeller C, Allen BM, Rabang C, Cannell MB, Balke CW & Izu LT (2006). Three-dimensional distribution of ryanodine receptor clusters in cardiac myocytes. Biophys J 91,1 13. Díaz ME, Trafford AW, O Neill SC & Eisner DA (1997). Measurement of sarcoplasmic reticulum Ca 2+ content and sarcolemmal Ca 2+ fluxes in isolated rat ventricular myocytes during spontaneous Ca 2+ release. JPhysiol501, Díaz ME, O Neill SC & Eisner DA (2004). Sarcoplasmic reticulum calcium content fluctuation is the key to cardiac alternans. Circ Res 94, Eisner DA, Nichols CG, O Neill SC, Smith GL & Valdeolmillos M (1989). The effects of metabolic inhibition on intracellular calcium and ph in isolated rat ventricular cells. JPhysiol411, Forbes MS & Sperelakis N (1977). Myocardial couplings: their structural variations in the mouse. J Ultrastruct Res 58, Forbes MS & Sperelakis N (1980). Structures located at the levels of the Z bands in mouse ventricular myocardial cells. Tissue Cell 12, Izu LT, Means SA, Shadid JN, Chen-Izu Y & Balke CW (2006). Interplay of ryanodine receptor distribution and calcium dynamics. Biophys J 91, Kaneko T, Tanaka H, Oyamada M, Kawata S & Takamatsu T (2000). Three distinct types of Ca 2+ waves in Langendorff-perfused rat heart revealed by real-time confocal microscopy. Circ Res 86, Klauke N, Smith GL & Cooper JM (2007). Microfluidic systems to examine intercellular coupling of pairs of cardiac myocytes. Lab Chip 7, Lamont C, Luther PW, Balke CW & Wier WG (1998). Intercellular Ca 2+ waves in rat heart muscle. JPhysiol512, Li Y, Diaz ME, Eisner DA & O Neill SC (2009). The effects of membrane potential, SR Ca content and RyR responsiveness on systolic Ca alternans in rat ventricular myocytes. JPhysiol 587, Minamikawa T, Cody SH & Williams DA (1997). In situ visualization of spontaneous calcium waves within perfused whole rat heart by confocal imaging. Am J Physiol Heart Circ Physiol 272, H236 H243.
9 J Physiol Ca 2+ waves and synchronization of alternating systolic release 6361 Miura M, Boyden PA & ter Keurs HE (1999). Ca 2+ waves during triggered propagated contractions in intact trabeculae. Determinants of the velocity of propagation. Circ Res 84, Rae J, Cooper K, Gates P & Watsky M (1991). Low access resistance perforated patch recordings using amphotericin B. JNeurosciMethods37, Severs NJ (1990). The cardiac gap junction and intercalated disc. Int J Cardiol 26, Sommer JR & Waugh RA (1976). The ultrastructure of the mammalian cardiac muscle cell, with special emphasis on the tubular membrane systems. A review. Am J Pathol 82, Takamatsu T, Minamikawa T, Kawachi H & Fujita S (1991). Imaging of calcium wave propagation in guinea-pig ventricular cell pairs by confocal laser scanning microscopy. Cell Struct Funct 16, Venetucci LA, Trafford AW, O Neill SC & Eisner DA (2008). The sarcoplasmic reticulum and arrhythmogenic calcium release. Cardiovasc Res 77, Weiss JN, Karma A, Shiferaw Y, Chen PS, Garfinkel A & Qu Z (2006). From pulsus to pulseless: the saga of cardiac alternans. Circ Res 98, Author contributions The work was performed in the Unit of Cardiac Physiology, University of Manchester. D.A.E. and S.C.O. designed the project and wrote the manuscript. Y.L. performed the experiments and, with D.A.E. and S.C.O., analysed the data. Acknowledgements This work was supported by the British Heart Foundation.
Modeling 3-D Calcium Waves from Stochastic Calcium sparks in a Sarcomere Using COMSOL
 Modeling 3-D Calcium Waves from Stochastic Calcium sparks in a Sarcomere Using COMSOL Zana Coulibaly 1, Leighton T. Izu 2 and Bradford E. Peercy 1 1 University of Maryland, Baltimore County 2 University
Modeling 3-D Calcium Waves from Stochastic Calcium sparks in a Sarcomere Using COMSOL Zana Coulibaly 1, Leighton T. Izu 2 and Bradford E. Peercy 1 1 University of Maryland, Baltimore County 2 University
Measurement of sarcoplasmic reticulum Ca content and sarcolemmal Ca fluxes in isolated rat ventricular myocytes during spontaneous Ca release
 Keywords: Calcium, Sarcoplasmic reticulum, Ventricular myocyte 6508 Journal of Physiology (1997), 501.1, pp. 3 16 3 Measurement of sarcoplasmic reticulum Ca content and sarcolemmal Ca fluxes in isolated
Keywords: Calcium, Sarcoplasmic reticulum, Ventricular myocyte 6508 Journal of Physiology (1997), 501.1, pp. 3 16 3 Measurement of sarcoplasmic reticulum Ca content and sarcolemmal Ca fluxes in isolated
Computer Science Department Technical Report. University of California. Los Angeles, CA
 Computer Science Department Technical Report University of California Los Angeles, CA 995-1596 COMPUTER SIMULATION OF THE JAFRI-WINSLOW ACTION POTENTIAL MODEL Sergey Y. Chernyavskiy November 1998 Boris
Computer Science Department Technical Report University of California Los Angeles, CA 995-1596 COMPUTER SIMULATION OF THE JAFRI-WINSLOW ACTION POTENTIAL MODEL Sergey Y. Chernyavskiy November 1998 Boris
Basic mechanisms of arrhythmogenesis and antiarrhythmia
 EHRA EDUCATIONAL REVIEW AND PREPARATORY COURSE ON INVASIVE CARDIAC ELECTROPHYSIOLOGY EUROPEAN HEART HOUSE, February 2011 Basic mechanisms of arrhythmogenesis and antiarrhythmia Antonio Zaza Università
EHRA EDUCATIONAL REVIEW AND PREPARATORY COURSE ON INVASIVE CARDIAC ELECTROPHYSIOLOGY EUROPEAN HEART HOUSE, February 2011 Basic mechanisms of arrhythmogenesis and antiarrhythmia Antonio Zaza Università
Cellular Electrophysiology and Biophysics
 BIOEN 6003 Cellular Electrophysiology and Biophysics Modeling of Force Development in Myocytes II Frank B. Sachse, University of Utah Overview Experimental Studies Sliding Filament Theory Group work Excitation-Contraction
BIOEN 6003 Cellular Electrophysiology and Biophysics Modeling of Force Development in Myocytes II Frank B. Sachse, University of Utah Overview Experimental Studies Sliding Filament Theory Group work Excitation-Contraction
Nonuniformity of Sarcomere Shortenings in the Isolated Rat Ventricular Myocyte
 Japanese Journal of Physiology, 52, 371 381, 2002 Nonuniformity of Sarcomere Shortenings in the Isolated Rat Ventricular Myocyte Nobuaki SARAI, Yasuki KIHARA*, Toshiaki IZUMI*, Tamotsu MITSUIYE, Satoshi
Japanese Journal of Physiology, 52, 371 381, 2002 Nonuniformity of Sarcomere Shortenings in the Isolated Rat Ventricular Myocyte Nobuaki SARAI, Yasuki KIHARA*, Toshiaki IZUMI*, Tamotsu MITSUIYE, Satoshi
PHYSIOLOGY CHAPTER 9 MUSCLE TISSUE Fall 2016
 PHYSIOLOGY CHAPTER 9 MUSCLE TISSUE Fall 2016 2 Chapter 9 Muscles and Muscle Tissue Overview of Muscle Tissue types of muscle: are all prefixes for muscle Contractility all muscles cells can Smooth & skeletal
PHYSIOLOGY CHAPTER 9 MUSCLE TISSUE Fall 2016 2 Chapter 9 Muscles and Muscle Tissue Overview of Muscle Tissue types of muscle: are all prefixes for muscle Contractility all muscles cells can Smooth & skeletal
Modeling. EC-Coupling and Contraction
 Bioeng 6460 Electrophysiology and Bioelectricity Modeling of EC-Coupling and Contraction Frank B. Sachse fs@cvrti.utah.edu Overview Quiz Excitation-Contraction Coupling Anatomy Cross Bridge Binding Coupling
Bioeng 6460 Electrophysiology and Bioelectricity Modeling of EC-Coupling and Contraction Frank B. Sachse fs@cvrti.utah.edu Overview Quiz Excitation-Contraction Coupling Anatomy Cross Bridge Binding Coupling
Role of the Na -Ca 2 Exchanger as an Alternative Trigger of CICR in Mammalian Cardiac Myocytes
 Biophysical Journal Volume 82 March 2002 1483 1496 1483 Role of the Na -Ca 2 Exchanger as an Alternative Trigger of CICR in Mammalian Cardiac Myocytes Chunlei Han, Pasi Tavi, and Matti Weckström Department
Biophysical Journal Volume 82 March 2002 1483 1496 1483 Role of the Na -Ca 2 Exchanger as an Alternative Trigger of CICR in Mammalian Cardiac Myocytes Chunlei Han, Pasi Tavi, and Matti Weckström Department
Our patient for the day...
 Muscles Ch.12 Our patient for the day... Name: Eddy Age: Newborn Whole-body muscle contractions No relaxation Severe difficulty breathing due to inadequate relaxation of breathing muscles Diagnosed with
Muscles Ch.12 Our patient for the day... Name: Eddy Age: Newborn Whole-body muscle contractions No relaxation Severe difficulty breathing due to inadequate relaxation of breathing muscles Diagnosed with
Inhibition of S532C by MTSET at intracellular ph 6.8 indicates accessibility in the closed
 Supplementary Text Inhibition of S532C by MTSET at intracellular ph 6.8 indicates accessibility in the closed state It is difficult to examine accessibility of cysteine-substituted mutants in the fully
Supplementary Text Inhibition of S532C by MTSET at intracellular ph 6.8 indicates accessibility in the closed state It is difficult to examine accessibility of cysteine-substituted mutants in the fully
Coupled dynamics of voltage and calcium in paced cardiac cells
 Coupled dynamics of voltage and calcium in paced cardiac cells Yohannes Shiferaw, 1,2 Daisuke Sato, 1 and Alain Karma 1 1 Department of Physics and Center for Interdisciplinary Research on Complex Systems,
Coupled dynamics of voltage and calcium in paced cardiac cells Yohannes Shiferaw, 1,2 Daisuke Sato, 1 and Alain Karma 1 1 Department of Physics and Center for Interdisciplinary Research on Complex Systems,
Introduction to Physiology IV - Calcium Dynamics
 Introduction to Physiology IV - Calcium Dynamics J. P. Keener Mathematics Department Introduction to Physiology IV - Calcium Dynamics p.1/26 Introduction Previous lectures emphasized the role of sodium
Introduction to Physiology IV - Calcium Dynamics J. P. Keener Mathematics Department Introduction to Physiology IV - Calcium Dynamics p.1/26 Introduction Previous lectures emphasized the role of sodium
A Model Based Analysis of Steady-State versus Dynamic Elements in the Relationship between Calcium and Force
 A Model Based Analysis of Steady-State versus Dynamic Elements in the Relationship between Calcium and Force Casey L. Overby, Sanjeev G. Shroff BACKGROUND Cardiac contraction and calcium. Intracellular
A Model Based Analysis of Steady-State versus Dynamic Elements in the Relationship between Calcium and Force Casey L. Overby, Sanjeev G. Shroff BACKGROUND Cardiac contraction and calcium. Intracellular
Role of coupled gating between cardiac ryanodine receptors in the genesis of triggered arrhythmias
 Am J Physiol Heart Circ Physiol 297: H171 H180, 2009. First published May 8, 2009; doi:10.1152/ajpheart.00098.2009. Role of coupled gating between cardiac ryanodine receptors in the genesis of triggered
Am J Physiol Heart Circ Physiol 297: H171 H180, 2009. First published May 8, 2009; doi:10.1152/ajpheart.00098.2009. Role of coupled gating between cardiac ryanodine receptors in the genesis of triggered
Rapid Report. Fundamental calcium release events revealed by two-photon excitation photolysis of caged calcium in guinea-pig cardiac myocytes
 Keywords: Calcium release; Cardiac myocyte; Sarcoplasmic reticulum 7700 Journal of Physiology (1998), 508.3, pp. 801 809 801 Rapid Report Fundamental calcium release events revealed by two-photon excitation
Keywords: Calcium release; Cardiac myocyte; Sarcoplasmic reticulum 7700 Journal of Physiology (1998), 508.3, pp. 801 809 801 Rapid Report Fundamental calcium release events revealed by two-photon excitation
A model for diffusion waves of calcium ions in a heart cell is given by a system of coupled, time-dependent reaction-diffusion equations
 Long-Time Simulation of Calcium Waves in a Heart Cell to Study the Effects of Calcium Release Flux Density and of Coefficients in the Pump and Leak Mechanisms on Self-Organizing Wave Behavior Zana Coulibaly,
Long-Time Simulation of Calcium Waves in a Heart Cell to Study the Effects of Calcium Release Flux Density and of Coefficients in the Pump and Leak Mechanisms on Self-Organizing Wave Behavior Zana Coulibaly,
Muscle tissue. Types. Functions. Cardiac, Smooth, and Skeletal
 Types Cardiac, Smooth, and Skeletal Functions movements posture and body position Support soft tissues Guard openings body temperature nutrient reserves Muscle tissue Special Characteristics of Muscle
Types Cardiac, Smooth, and Skeletal Functions movements posture and body position Support soft tissues Guard openings body temperature nutrient reserves Muscle tissue Special Characteristics of Muscle
Introduction to electrophysiology. Dr. Tóth András
 Introduction to electrophysiology Dr. Tóth András Topics Transmembran transport Donnan equilibrium Resting potential Ion channels Local and action potentials Intra- and extracellular propagation of the
Introduction to electrophysiology Dr. Tóth András Topics Transmembran transport Donnan equilibrium Resting potential Ion channels Local and action potentials Intra- and extracellular propagation of the
UNIT 6 THE MUSCULAR SYSTEM
 UNIT 6 THE MUSCULAR SYSTEM I. Functions of Muscular System A. Produces Movement Internal vs. External «locomotion & manipulation «circulate blood & maintain blood pressure «move fluids, food, baby B. Maintaining
UNIT 6 THE MUSCULAR SYSTEM I. Functions of Muscular System A. Produces Movement Internal vs. External «locomotion & manipulation «circulate blood & maintain blood pressure «move fluids, food, baby B. Maintaining
Microscopic Spiral Waves Reveal Positive Feedback in Subcellular
 2272 Biophysical Journal Volume 65 December 1993 2272-2276 Microscopic Spiral Waves Reveal Positive Feedback in Subcellular Calcium Signaling Peter Lipp and Ernst Niggli Department of Physiology, University
2272 Biophysical Journal Volume 65 December 1993 2272-2276 Microscopic Spiral Waves Reveal Positive Feedback in Subcellular Calcium Signaling Peter Lipp and Ernst Niggli Department of Physiology, University
Quantitative Electrophysiology
 ECE 795: Quantitative Electrophysiology Notes for Lecture #1 Wednesday, September 13, 2006 1. INTRODUCTION TO EXCITABLE CELLS Historical perspective: Bioelectricity first discovered by Luigi Galvani in
ECE 795: Quantitative Electrophysiology Notes for Lecture #1 Wednesday, September 13, 2006 1. INTRODUCTION TO EXCITABLE CELLS Historical perspective: Bioelectricity first discovered by Luigi Galvani in
Physiology Unit 2. MEMBRANE POTENTIALS and SYNAPSES
 Physiology Unit 2 MEMBRANE POTENTIALS and SYNAPSES Neuron Communication Neurons are stimulated by receptors on dendrites and cell bodies (soma) Ligand gated ion channels GPCR s Neurons stimulate cells
Physiology Unit 2 MEMBRANE POTENTIALS and SYNAPSES Neuron Communication Neurons are stimulated by receptors on dendrites and cell bodies (soma) Ligand gated ion channels GPCR s Neurons stimulate cells
لجنة الطب البشري رؤية تنير دروب تميزكم
 1) Hyperpolarization phase of the action potential: a. is due to the opening of voltage-gated Cl channels. b. is due to prolonged opening of voltage-gated K + channels. c. is due to closure of the Na +
1) Hyperpolarization phase of the action potential: a. is due to the opening of voltage-gated Cl channels. b. is due to prolonged opening of voltage-gated K + channels. c. is due to closure of the Na +
The Journal of Physiology
 J Physiol 591.9 (2013) pp 2287 2306 2287 Sarcolemmal localisation of Na + /H + exchange and Na + HCO 3 co-transport influences the spatial regulation of intracellular ph in rat ventricular myocytes Carolina
J Physiol 591.9 (2013) pp 2287 2306 2287 Sarcolemmal localisation of Na + /H + exchange and Na + HCO 3 co-transport influences the spatial regulation of intracellular ph in rat ventricular myocytes Carolina
Lecture 13, 05 October 2004 Chapter 10, Muscle. Vertebrate Physiology ECOL 437 University of Arizona Fall instr: Kevin Bonine t.a.
 Lecture 13, 05 October 2004 Chapter 10, Muscle Vertebrate Physiology ECOL 437 University of Arizona Fall 2004 instr: Kevin Bonine t.a.: Nate Swenson Vertebrate Physiology 437 18 1. Muscle A. Sarcomere
Lecture 13, 05 October 2004 Chapter 10, Muscle Vertebrate Physiology ECOL 437 University of Arizona Fall 2004 instr: Kevin Bonine t.a.: Nate Swenson Vertebrate Physiology 437 18 1. Muscle A. Sarcomere
Quantitative Electrophysiology
 ECE 795: Quantitative Electrophysiology Notes for Lecture #1 Tuesday, September 18, 2012 1. INTRODUCTION TO EXCITABLE CELLS Historical perspective: Bioelectricity first discovered by Luigi Galvani in 1780s
ECE 795: Quantitative Electrophysiology Notes for Lecture #1 Tuesday, September 18, 2012 1. INTRODUCTION TO EXCITABLE CELLS Historical perspective: Bioelectricity first discovered by Luigi Galvani in 1780s
Journal of Theoretical Biology
 Journal of Theoretical Biology 266 (21) 48 418 Contents lists available at ScienceDirect Journal of Theoretical Biology journal homepage: www.elsevier.com/locate/yjtbi Feedback-control induced pattern
Journal of Theoretical Biology 266 (21) 48 418 Contents lists available at ScienceDirect Journal of Theoretical Biology journal homepage: www.elsevier.com/locate/yjtbi Feedback-control induced pattern
According to the diagram, which of the following is NOT true?
 Instructions: Review Chapter 44 on muscular-skeletal systems and locomotion, and then complete the following Blackboard activity. This activity will introduce topics that will be covered in the next few
Instructions: Review Chapter 44 on muscular-skeletal systems and locomotion, and then complete the following Blackboard activity. This activity will introduce topics that will be covered in the next few
Electrophysiological Modeling of Membranes and Cells
 Bioeng 6460 Electrophysiology and Bioelectricity Electrophysiological Modeling of Membranes and Cells Frank B. Sachse fs@cvrti.utah.edu Overview Recapitulation Electrical Modeling of Membranes Cardiac
Bioeng 6460 Electrophysiology and Bioelectricity Electrophysiological Modeling of Membranes and Cells Frank B. Sachse fs@cvrti.utah.edu Overview Recapitulation Electrical Modeling of Membranes Cardiac
Spontaneous Calcium Release in Cardiac Myocytes: Store Overload and Electrical Dynamics
 Spora: A Journal of Biomathematics Volume 1 Issue 1 Article 6 215 Spontaneous Calcium Release in Cardiac Myocytes: Store Overload and Electrical Dynamics Amanda M. Alexander Western Washington University,
Spora: A Journal of Biomathematics Volume 1 Issue 1 Article 6 215 Spontaneous Calcium Release in Cardiac Myocytes: Store Overload and Electrical Dynamics Amanda M. Alexander Western Washington University,
Computational Modeling of the Cardiovascular and Neuronal System
 BIOEN 6900 Computational Modeling of the Cardiovascular and Neuronal System Modeling of Force Development in Myocytes Overview Recapitulation Modeling of Conduction Modeling of Force in Skeletal Muscle
BIOEN 6900 Computational Modeling of the Cardiovascular and Neuronal System Modeling of Force Development in Myocytes Overview Recapitulation Modeling of Conduction Modeling of Force in Skeletal Muscle
Biology September 2015 Exam One FORM G KEY
 Biology 251 17 September 2015 Exam One FORM G KEY PRINT YOUR NAME AND ID NUMBER in the space that is provided on the answer sheet, and then blacken the letter boxes below the corresponding letters of your
Biology 251 17 September 2015 Exam One FORM G KEY PRINT YOUR NAME AND ID NUMBER in the space that is provided on the answer sheet, and then blacken the letter boxes below the corresponding letters of your
Biology September 2015 Exam One FORM W KEY
 Biology 251 17 September 2015 Exam One FORM W KEY PRINT YOUR NAME AND ID NUMBER in the space that is provided on the answer sheet, and then blacken the letter boxes below the corresponding letters of your
Biology 251 17 September 2015 Exam One FORM W KEY PRINT YOUR NAME AND ID NUMBER in the space that is provided on the answer sheet, and then blacken the letter boxes below the corresponding letters of your
Membrane Physiology. Dr. Hiwa Shafiq Oct-18 1
 Membrane Physiology Dr. Hiwa Shafiq 22-10-2018 29-Oct-18 1 Chemical compositions of extracellular and intracellular fluids. 29-Oct-18 2 Transport through the cell membrane occurs by one of two basic processes:
Membrane Physiology Dr. Hiwa Shafiq 22-10-2018 29-Oct-18 1 Chemical compositions of extracellular and intracellular fluids. 29-Oct-18 2 Transport through the cell membrane occurs by one of two basic processes:
Frontiers in CardioVascular Biology
 Frontiers in CardioVascular Biology Young Investigator Award Mitofusin 2 controls calcium transmission between the SR and mitochondria and regulates the bioenergetic feedback response in cardiac myocytes
Frontiers in CardioVascular Biology Young Investigator Award Mitofusin 2 controls calcium transmission between the SR and mitochondria and regulates the bioenergetic feedback response in cardiac myocytes
Ion Channel Discovery Services
 www.essenbioscience.com Ion Channel Discovery Services Customer: Customer PO: Essen contact: Date of assay: Customer contact: Essen Quote No: Srini Kanumilli Lab Notebook Ref: Date of Report: LNB-2650
www.essenbioscience.com Ion Channel Discovery Services Customer: Customer PO: Essen contact: Date of assay: Customer contact: Essen Quote No: Srini Kanumilli Lab Notebook Ref: Date of Report: LNB-2650
SUPPLEMENTARY INFORMATION
 Cell viability rate 0.8 0.6 0 0.05 0.1 0.2 0.3 0.4 0.5 0.7 1 Exposure duration (s) Supplementary Figure 1. Femtosecond laser could disrupt and turn off GFP without photons at 473 nm and keep cells alive.
Cell viability rate 0.8 0.6 0 0.05 0.1 0.2 0.3 0.4 0.5 0.7 1 Exposure duration (s) Supplementary Figure 1. Femtosecond laser could disrupt and turn off GFP without photons at 473 nm and keep cells alive.
CALIFORNIA STATE UNIVERSITY, NORTHRIDGE. Cardiac Cells. For the degree of Master of Science in Physics. Gonzalo Hernandez Hernandez
 CALIFORNIA STATE UNIVERSITY, NORTHRIDGE Role of Network Connectivity and Fluctuations in the Nucleation of Calcium Waves in Cardiac Cells A thesis submitted in partial fulfillment of the requirements For
CALIFORNIA STATE UNIVERSITY, NORTHRIDGE Role of Network Connectivity and Fluctuations in the Nucleation of Calcium Waves in Cardiac Cells A thesis submitted in partial fulfillment of the requirements For
Effectiveness of numerical techniques for calculating the quantity of calcium ion species during calcium sparksin heart muscle
 Effectiveness of numerical techniques for calculating the quantity of calcium ion species during calcium sparksin heart muscle D. A. Lott Departments of Mathematics and Biotechnology, Delaware State University,
Effectiveness of numerical techniques for calculating the quantity of calcium ion species during calcium sparksin heart muscle D. A. Lott Departments of Mathematics and Biotechnology, Delaware State University,
Peripheral Nerve II. Amelyn Ramos Rafael, MD. Anatomical considerations
 Peripheral Nerve II Amelyn Ramos Rafael, MD Anatomical considerations 1 Physiologic properties of the nerve Irritability of the nerve A stimulus applied on the nerve causes the production of a nerve impulse,
Peripheral Nerve II Amelyn Ramos Rafael, MD Anatomical considerations 1 Physiologic properties of the nerve Irritability of the nerve A stimulus applied on the nerve causes the production of a nerve impulse,
Overview Organization: Central Nervous System (CNS) Peripheral Nervous System (PNS) innervate Divisions: a. Afferent
 Overview Organization: Central Nervous System (CNS) Brain and spinal cord receives and processes information. Peripheral Nervous System (PNS) Nerve cells that link CNS with organs throughout the body.
Overview Organization: Central Nervous System (CNS) Brain and spinal cord receives and processes information. Peripheral Nervous System (PNS) Nerve cells that link CNS with organs throughout the body.
the axons of the nerve meet with the muscle cell.
 Steps to Contraction 1. A nerve impulse travels to the neuromuscular junction on a muscle cell. The neuromuscular junction is the point where the axons of the nerve meet with the muscle cell. 2. Ach is
Steps to Contraction 1. A nerve impulse travels to the neuromuscular junction on a muscle cell. The neuromuscular junction is the point where the axons of the nerve meet with the muscle cell. 2. Ach is
Modeling of Action Potential Generation in NG cells
 Modeling of Action Potential Generation in NG108-15 cells NanoScience Technology Center University of Central Florida 144 Research Parkway, Suite 400 Orlando, FL 386 * Corresponding author Email: pmolnar@mail.ucf.edu
Modeling of Action Potential Generation in NG108-15 cells NanoScience Technology Center University of Central Florida 144 Research Parkway, Suite 400 Orlando, FL 386 * Corresponding author Email: pmolnar@mail.ucf.edu
Introduction to electrophysiology 1. Dr. Tóth András
 Introduction to electrophysiology 1. Dr. Tóth András Topics Transmembran transport Donnan equilibrium Resting potential Ion channels Local and action potentials Intra- and extracellular propagation of
Introduction to electrophysiology 1. Dr. Tóth András Topics Transmembran transport Donnan equilibrium Resting potential Ion channels Local and action potentials Intra- and extracellular propagation of
Chapter 2 Basic Cardiac Electrophysiology: Excitable Membranes
 Chapter Basic Cardiac Electrophysiology: Excitable Membranes Deborah A. Jaye, Yong-Fu Xiao, and Daniel C. Sigg Abstract Cardiomyocytes are excitable cells that have the ability to contract after excitation;
Chapter Basic Cardiac Electrophysiology: Excitable Membranes Deborah A. Jaye, Yong-Fu Xiao, and Daniel C. Sigg Abstract Cardiomyocytes are excitable cells that have the ability to contract after excitation;
3-dimensional simulation of long QT syndrome: early afterdepolarizations and reentry Ray Huffaker, Boris Kogan
 3-dimensional simulation of long QT syndrome: early afterdepolarizations and reentry Ray Huffaker, Boris Kogan Abstract Early afterdepolarizations (EADs), thought to be highly arrhythmogenic in the case
3-dimensional simulation of long QT syndrome: early afterdepolarizations and reentry Ray Huffaker, Boris Kogan Abstract Early afterdepolarizations (EADs), thought to be highly arrhythmogenic in the case
THE HIGH prevalence of heart failure is largely due to our
 IEEE TRANSACTIONS ON BIOMEDICAL ENGINEERING, VOL. 58, NO. 10, OCTOBER 2011 2947 Multiscale Modeling of Calcium Dynamics in Ventricular Myocytes With Realistic Transverse Tubules Zeyun Yu*, Guangming Yao,
IEEE TRANSACTIONS ON BIOMEDICAL ENGINEERING, VOL. 58, NO. 10, OCTOBER 2011 2947 Multiscale Modeling of Calcium Dynamics in Ventricular Myocytes With Realistic Transverse Tubules Zeyun Yu*, Guangming Yao,
GPU-Enabled Spatiotemporal Model of Stochastic Cardiac Calcium Dynamics and Arrhythmias
 GPU-Enabled Spatiotemporal Model of Stochastic Cardiac Calcium Dynamics and Arrhythmias M. Saleet Jafri Hoang Trong Minh Tuan Department of Bioinformatics and Computational Biology George Mason University
GPU-Enabled Spatiotemporal Model of Stochastic Cardiac Calcium Dynamics and Arrhythmias M. Saleet Jafri Hoang Trong Minh Tuan Department of Bioinformatics and Computational Biology George Mason University
Lecture Notes 8C120 Inleiding Meten en Modelleren. Cellular electrophysiology: modeling and simulation. Nico Kuijpers
 Lecture Notes 8C2 Inleiding Meten en Modelleren Cellular electrophysiology: modeling and simulation Nico Kuijpers nico.kuijpers@bf.unimaas.nl February 9, 2 2 8C2 Inleiding Meten en Modelleren Extracellular
Lecture Notes 8C2 Inleiding Meten en Modelleren Cellular electrophysiology: modeling and simulation Nico Kuijpers nico.kuijpers@bf.unimaas.nl February 9, 2 2 8C2 Inleiding Meten en Modelleren Extracellular
Introduction to Physiology V - Coupling and Propagation
 Introduction to Physiology V - Coupling and Propagation J. P. Keener Mathematics Department Coupling and Propagation p./33 Spatially Extended Excitable Media Neurons and axons Coupling and Propagation
Introduction to Physiology V - Coupling and Propagation J. P. Keener Mathematics Department Coupling and Propagation p./33 Spatially Extended Excitable Media Neurons and axons Coupling and Propagation
Nervous Systems: Neuron Structure and Function
 Nervous Systems: Neuron Structure and Function Integration An animal needs to function like a coherent organism, not like a loose collection of cells. Integration = refers to processes such as summation
Nervous Systems: Neuron Structure and Function Integration An animal needs to function like a coherent organism, not like a loose collection of cells. Integration = refers to processes such as summation
Reception The target cell s detection of a signal coming from outside the cell May Occur by: Direct connect Through signal molecules
 Why Do Cells Communicate? Regulation Cells need to control cellular processes In multicellular organism, cells signaling pathways coordinate the activities within individual cells that support the function
Why Do Cells Communicate? Regulation Cells need to control cellular processes In multicellular organism, cells signaling pathways coordinate the activities within individual cells that support the function
Membrane Potentials, Action Potentials, and Synaptic Transmission. Membrane Potential
 Cl Cl - - + K + K+ K + K Cl - 2/2/15 Membrane Potentials, Action Potentials, and Synaptic Transmission Core Curriculum II Spring 2015 Membrane Potential Example 1: K +, Cl - equally permeant no charge
Cl Cl - - + K + K+ K + K Cl - 2/2/15 Membrane Potentials, Action Potentials, and Synaptic Transmission Core Curriculum II Spring 2015 Membrane Potential Example 1: K +, Cl - equally permeant no charge
2013 NSF-CMACS Workshop on Atrial Fibrillation
 2013 NSF-CMACS Workshop on A Atrial Fibrillation Flavio H. Fenton School of Physics Georgia Institute of Technology, Atlanta, GA and Max Planck Institute for Dynamics and Self-organization, Goettingen,
2013 NSF-CMACS Workshop on A Atrial Fibrillation Flavio H. Fenton School of Physics Georgia Institute of Technology, Atlanta, GA and Max Planck Institute for Dynamics and Self-organization, Goettingen,
Physiology Unit 2. MEMBRANE POTENTIALS and SYNAPSES
 Physiology Unit 2 MEMBRANE POTENTIALS and SYNAPSES In Physiology Today Ohm s Law I = V/R Ohm s law: the current through a conductor between two points is directly proportional to the voltage across the
Physiology Unit 2 MEMBRANE POTENTIALS and SYNAPSES In Physiology Today Ohm s Law I = V/R Ohm s law: the current through a conductor between two points is directly proportional to the voltage across the
CSD-TR R. Samade, B. Kogan
 The properties of the cardiac cell mathematical model with a Markovian representation of potassium channel gating processes under high pacing rate (Computer simulation study) CSD-TR040007 R. Samade, B.
The properties of the cardiac cell mathematical model with a Markovian representation of potassium channel gating processes under high pacing rate (Computer simulation study) CSD-TR040007 R. Samade, B.
Effects of Partial Sarcoplasmic Reticulum Calcium Depletion on Calcium Release in Frog Cut Muscle Fibers Equilibrated with 20 mm EGTA
 Published Online: 1 September, 1998 Supp Info: http://doi.org/10.1085/jgp.112.3.263 Downloaded from jgp.rupress.org on August 16, 2018 Effects of Partial Sarcoplasmic Reticulum Calcium Depletion on Calcium
Published Online: 1 September, 1998 Supp Info: http://doi.org/10.1085/jgp.112.3.263 Downloaded from jgp.rupress.org on August 16, 2018 Effects of Partial Sarcoplasmic Reticulum Calcium Depletion on Calcium
Fundamentals of Neurosciences. Smooth Muscle. Dr. Kumar Sambamurti 613-SEI; ;
 Fundamentals of Neurosciences Smooth Muscle Dr. Kumar Sambamurti 613-SEI; 792-4315; sambak@musc.edu 1 Smooth Muscle Structure Cells much smaller than skeletal muscle (2-5µM diam, 100-400µM long) Single
Fundamentals of Neurosciences Smooth Muscle Dr. Kumar Sambamurti 613-SEI; 792-4315; sambak@musc.edu 1 Smooth Muscle Structure Cells much smaller than skeletal muscle (2-5µM diam, 100-400µM long) Single
MEMBRANE POTENTIALS AND ACTION POTENTIALS:
 University of Jordan Faculty of Medicine Department of Physiology & Biochemistry Medical students, 2017/2018 +++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++ Review: Membrane physiology
University of Jordan Faculty of Medicine Department of Physiology & Biochemistry Medical students, 2017/2018 +++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++ Review: Membrane physiology
Electrical Signaling. Lecture Outline. Using Ions as Messengers. Potentials in Electrical Signaling
 Lecture Outline Electrical Signaling Using ions as messengers Potentials in electrical signaling Action Graded Other electrical signaling Gap junctions The neuron Using Ions as Messengers Important things
Lecture Outline Electrical Signaling Using ions as messengers Potentials in electrical signaling Action Graded Other electrical signaling Gap junctions The neuron Using Ions as Messengers Important things
Periodic Behavior in Cardiac Tissue: Dynamics of Spatially Discordant Calcium Alternans
 University of Colorado, Boulder CU Scholar Applied Mathematics Graduate Theses & Dissertations Applied Mathematics Spring 1-1-213 Periodic Behavior in Cardiac Tissue: Dynamics of Spatially Discordant Calcium
University of Colorado, Boulder CU Scholar Applied Mathematics Graduate Theses & Dissertations Applied Mathematics Spring 1-1-213 Periodic Behavior in Cardiac Tissue: Dynamics of Spatially Discordant Calcium
Membrane Currents in Mammalian Ventricular Heart Muscle Fibers Using a Voltage-Clamp Technique
 Membrane Currents in Mammalian Ventricular Heart Muscle Fibers Using a Voltage-Clamp Technique GERHARD GIEBISCH and SILVIO WEIDMANN From the Department of Physiology, University of Berne, Berne, Switzerland.
Membrane Currents in Mammalian Ventricular Heart Muscle Fibers Using a Voltage-Clamp Technique GERHARD GIEBISCH and SILVIO WEIDMANN From the Department of Physiology, University of Berne, Berne, Switzerland.
AD-" IONIC BASIS OF POTENTIAL REGULATION(U) BAYLOR COLLO / U U ijejmedicine L HOUSTON TX DEPT OF PHYSIOLOGY AND MOLECULAR 7 MEEE"..
 AD-"19 459 IONIC BASIS OF POTENTIAL REGULATION(U) BAYLOR COLLO / U U ijejmedicine L HOUSTON TX DEPT OF PHYSIOLOGY AND MOLECULAR 7 MEEE"..,E NCLA SIFIE BIOPHYSIC S D C CHANG 6 i N 1988 Neg@14-85-K-6424
AD-"19 459 IONIC BASIS OF POTENTIAL REGULATION(U) BAYLOR COLLO / U U ijejmedicine L HOUSTON TX DEPT OF PHYSIOLOGY AND MOLECULAR 7 MEEE"..,E NCLA SIFIE BIOPHYSIC S D C CHANG 6 i N 1988 Neg@14-85-K-6424
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Eisner et al., http://www.jcb.org/cgi/content/full/jcb.201312066/dc1 Figure S1. Mitochondrial continuity in adult skeletal muscle. (A)
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Eisner et al., http://www.jcb.org/cgi/content/full/jcb.201312066/dc1 Figure S1. Mitochondrial continuity in adult skeletal muscle. (A)
Calcium waves initiating from the anomalous subdiffusive calcium sparks
 Calcium waves initiating from the anomalous subdiffusive calcium sparks rsif.royalsocietypublishing.org Research Cite this article: Chen X, Guo L, Kang J, Huo Y, Wang S, Tan W. 2014 Calcium waves initiating
Calcium waves initiating from the anomalous subdiffusive calcium sparks rsif.royalsocietypublishing.org Research Cite this article: Chen X, Guo L, Kang J, Huo Y, Wang S, Tan W. 2014 Calcium waves initiating
Action Potential Propagation
 Action Potential Propagation 2 Action Potential is a transient alteration of transmembrane voltage (or membrane potential) across an excitable membrane generated by the activity of voltage-gated ion channels.
Action Potential Propagation 2 Action Potential is a transient alteration of transmembrane voltage (or membrane potential) across an excitable membrane generated by the activity of voltage-gated ion channels.
CELL BIOLOGY - CLUTCH CH. 9 - TRANSPORT ACROSS MEMBRANES.
 !! www.clutchprep.com K + K + K + K + CELL BIOLOGY - CLUTCH CONCEPT: PRINCIPLES OF TRANSMEMBRANE TRANSPORT Membranes and Gradients Cells must be able to communicate across their membrane barriers to materials
!! www.clutchprep.com K + K + K + K + CELL BIOLOGY - CLUTCH CONCEPT: PRINCIPLES OF TRANSMEMBRANE TRANSPORT Membranes and Gradients Cells must be able to communicate across their membrane barriers to materials
Lecture 2. Excitability and ionic transport
 Lecture 2 Excitability and ionic transport Selective membrane permeability: The lipid barrier of the cell membrane and cell membrane transport proteins Chemical compositions of extracellular and intracellular
Lecture 2 Excitability and ionic transport Selective membrane permeability: The lipid barrier of the cell membrane and cell membrane transport proteins Chemical compositions of extracellular and intracellular
Model of Intracellular Calcium Cycling in Ventricular Myocytes
 3666 Biophysical Journal Volume 85 December 2003 3666 3686 Model of Intracellular Calcium Cycling in Ventricular Myocytes Y. Shiferaw,* M. A. Watanabe,* A. Garfinkel, y J. N. Weiss, y and A. Karma* *Department
3666 Biophysical Journal Volume 85 December 2003 3666 3686 Model of Intracellular Calcium Cycling in Ventricular Myocytes Y. Shiferaw,* M. A. Watanabe,* A. Garfinkel, y J. N. Weiss, y and A. Karma* *Department
Ca 2 -induced Ca 2 Release in Chinese Hamster Ovary (CHO) Cells Co-expressing Dihydropyridine and Ryanodine Receptors
 Ca 2 -induced Ca 2 Release in Chinese Hamster Ovary (CHO) Cells Co-expressing Dihydropyridine and Ryanodine Receptors Norio Suda,* Dorothee Franzius,* Andrea Fleig,* Seiichiro Nishimura, Matthias Bödding,*
Ca 2 -induced Ca 2 Release in Chinese Hamster Ovary (CHO) Cells Co-expressing Dihydropyridine and Ryanodine Receptors Norio Suda,* Dorothee Franzius,* Andrea Fleig,* Seiichiro Nishimura, Matthias Bödding,*
BRIEF COMMUNICATION OF ASYMMETRY CURRENT SQUID AXON MEMBRANE FREQUENCY DOMAIN ANALYSIS
 FREQUENCY DOMAIN ANALYSIS OF ASYMMETRY CURRENT IN SQUID AXON MEMBRANE SHIRo TAKASHIMA, Department ofbioengineering D2, University of Pennsylvania, Philadelphia, Pennsylvania 19104 U.S.A. ABSTRACT The change
FREQUENCY DOMAIN ANALYSIS OF ASYMMETRY CURRENT IN SQUID AXON MEMBRANE SHIRo TAKASHIMA, Department ofbioengineering D2, University of Pennsylvania, Philadelphia, Pennsylvania 19104 U.S.A. ABSTRACT The change
Biomedical Instrumentation
 ELEC ENG 4BD4: Biomedical Instrumentation Lecture 5 Bioelectricity 1. INTRODUCTION TO BIOELECTRICITY AND EXCITABLE CELLS Historical perspective: Bioelectricity first discovered by Luigi Galvani in 1780s
ELEC ENG 4BD4: Biomedical Instrumentation Lecture 5 Bioelectricity 1. INTRODUCTION TO BIOELECTRICITY AND EXCITABLE CELLS Historical perspective: Bioelectricity first discovered by Luigi Galvani in 1780s
Membrane Protein Pumps
 Membrane Protein Pumps Learning objectives You should be able to understand & discuss: Active transport-na + /K + ATPase ABC transporters Metabolite transport by lactose permease 1. Ion pumps: ATP-driven
Membrane Protein Pumps Learning objectives You should be able to understand & discuss: Active transport-na + /K + ATPase ABC transporters Metabolite transport by lactose permease 1. Ion pumps: ATP-driven
Dynamical Systems for Biology - Excitability
 Dynamical Systems for Biology - Excitability J. P. Keener Mathematics Department Dynamical Systems for Biology p.1/25 Examples of Excitable Media B-Z reagent CICR (Calcium Induced Calcium Release) Nerve
Dynamical Systems for Biology - Excitability J. P. Keener Mathematics Department Dynamical Systems for Biology p.1/25 Examples of Excitable Media B-Z reagent CICR (Calcium Induced Calcium Release) Nerve
Neurons and Nervous Systems
 34 Neurons and Nervous Systems Concept 34.1 Nervous Systems Consist of Neurons and Glia Nervous systems have two categories of cells: Neurons, or nerve cells, are excitable they generate and transmit electrical
34 Neurons and Nervous Systems Concept 34.1 Nervous Systems Consist of Neurons and Glia Nervous systems have two categories of cells: Neurons, or nerve cells, are excitable they generate and transmit electrical
Neurons. The Molecular Basis of their Electrical Excitability
 Neurons The Molecular Basis of their Electrical Excitability Viva La Complexity! Consider, The human brain contains >10 11 neurons! Each neuron makes 10 3 (average) synaptic contacts on up to 10 3 other
Neurons The Molecular Basis of their Electrical Excitability Viva La Complexity! Consider, The human brain contains >10 11 neurons! Each neuron makes 10 3 (average) synaptic contacts on up to 10 3 other
Modeling action potential generation and propagation in NRK fibroblasts
 Am J Physiol Cell Physiol 287: C851 C865, 2004. First published May 12, 2004; 10.1152/ajpcell.00220.2003. Modeling action potential generation and propagation in NRK fibroblasts J. J. Torres, 1,2 L. N.
Am J Physiol Cell Physiol 287: C851 C865, 2004. First published May 12, 2004; 10.1152/ajpcell.00220.2003. Modeling action potential generation and propagation in NRK fibroblasts J. J. Torres, 1,2 L. N.
Multiple Choice Identify the letter of the choice that best completes the statement or answers the question.
 chapter 7 Test Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. Who was one of the first people to identify and see cork cells? a. Anton van
chapter 7 Test Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. Who was one of the first people to identify and see cork cells? a. Anton van
Cardiac-type EC-Coupling in Dysgenic Myotubes Restored with Ca 21 Channel Subunit Isoforms a 1C and a 1D Does not Correlate with Current Density
 3816 Biophysical Journal Volume 84 June 2003 3816 3828 Cardiac-type EC-Coupling in Dysgenic Myotubes Restored with Ca 21 Channel Subunit Isoforms a 1C and a 1D Does not Correlate with Current Density Nicole
3816 Biophysical Journal Volume 84 June 2003 3816 3828 Cardiac-type EC-Coupling in Dysgenic Myotubes Restored with Ca 21 Channel Subunit Isoforms a 1C and a 1D Does not Correlate with Current Density Nicole
Channels can be activated by ligand-binding (chemical), voltage change, or mechanical changes such as stretch.
 1. Describe the basic structure of an ion channel. Name 3 ways a channel can be "activated," and describe what occurs upon activation. What are some ways a channel can decide what is allowed to pass through?
1. Describe the basic structure of an ion channel. Name 3 ways a channel can be "activated," and describe what occurs upon activation. What are some ways a channel can decide what is allowed to pass through?
REVIEW 2: CELLS & CELL COMMUNICATION. A. Top 10 If you learned anything from this unit, you should have learned:
 Name AP Biology REVIEW 2: CELLS & CELL COMMUNICATION A. Top 10 If you learned anything from this unit, you should have learned: 1. Prokaryotes vs. eukaryotes No internal membranes vs. membrane-bound organelles
Name AP Biology REVIEW 2: CELLS & CELL COMMUNICATION A. Top 10 If you learned anything from this unit, you should have learned: 1. Prokaryotes vs. eukaryotes No internal membranes vs. membrane-bound organelles
Spatiotemporal Features of Ca 2 Buffering and Diffusion in Atrial Cardiac Myocytes with Inhibited Sarcoplasmic Reticulum
 3134 Biophysical Journal Volume 83 December 2002 3134 3151 Spatiotemporal Features of 2 Buffering and Diffusion in Atrial rdiac Myocytes with Inhibited Sarcoplasmic Reticulum Anushka Michailova,* Franco
3134 Biophysical Journal Volume 83 December 2002 3134 3151 Spatiotemporal Features of 2 Buffering and Diffusion in Atrial rdiac Myocytes with Inhibited Sarcoplasmic Reticulum Anushka Michailova,* Franco
EIGENVALUE BASED ALTERNANS PREDICTION AND THE EFFECTS OF HEART RATE VARIABILITY ON ALTERNANS FORMATION
 EIGENVALUE BASED ALTERNANS PREDICTION AND THE EFFECTS OF HEART RATE VARIABILITY ON ALTERNANS FORMATION A Thesis SUBMITTED TO THE FACULTY OF UNIVERSITY OF MINNESOTA BY VIRENDRA VILAS KAKADE IN PARTIAL FULFILLMENT
EIGENVALUE BASED ALTERNANS PREDICTION AND THE EFFECTS OF HEART RATE VARIABILITY ON ALTERNANS FORMATION A Thesis SUBMITTED TO THE FACULTY OF UNIVERSITY OF MINNESOTA BY VIRENDRA VILAS KAKADE IN PARTIAL FULFILLMENT
(Be sure to clearly state the principles addressed in your discussion.)
 CELL QUESTION 1992: AP BIOLOGY A laboratory assistant prepared solutions of 0.8 M, 0.6 M, 0.4 M, and 0.2 M sucrose, but forgot to label them. After realizing the error, the assistant randomly labeled the
CELL QUESTION 1992: AP BIOLOGY A laboratory assistant prepared solutions of 0.8 M, 0.6 M, 0.4 M, and 0.2 M sucrose, but forgot to label them. After realizing the error, the assistant randomly labeled the
Analysing cardiac excitation contraction coupling with mathematical models of local control
 Progress in Biophysics & Molecular Biology 85 (2004) 141 162 Analysing cardiac excitation contraction coupling with mathematical models of local control Christian Soeller*, Mark B. Cannell Department of
Progress in Biophysics & Molecular Biology 85 (2004) 141 162 Analysing cardiac excitation contraction coupling with mathematical models of local control Christian Soeller*, Mark B. Cannell Department of
Ionic Charge Conservation and Long-Term Steady State in the Luo Rudy Dynamic Cell Model
 3324 Biophysical Journal Volume 81 December 2001 3324 3331 Ionic Charge Conservation and Long-Term Steady State in the Luo Rudy Dynamic Cell Model Thomas J. Hund,* Jan P. Kucera,* Niels F. Otani,* and
3324 Biophysical Journal Volume 81 December 2001 3324 3331 Ionic Charge Conservation and Long-Term Steady State in the Luo Rudy Dynamic Cell Model Thomas J. Hund,* Jan P. Kucera,* Niels F. Otani,* and
Microsystems for Neuroscience and Medicine. Lecture 9
 1 Microsystems for Neuroscience and Medicine Lecture 9 2 Neural Microsystems Neurons - Structure and behaviour Measuring neural activity Interfacing with neurons Medical applications - DBS, Retinal Implants
1 Microsystems for Neuroscience and Medicine Lecture 9 2 Neural Microsystems Neurons - Structure and behaviour Measuring neural activity Interfacing with neurons Medical applications - DBS, Retinal Implants
Ch. 5. Membrane Potentials and Action Potentials
 Ch. 5. Membrane Potentials and Action Potentials Basic Physics of Membrane Potentials Nerve and muscle cells: Excitable Capable of generating rapidly changing electrochemical impulses at their membranes
Ch. 5. Membrane Potentials and Action Potentials Basic Physics of Membrane Potentials Nerve and muscle cells: Excitable Capable of generating rapidly changing electrochemical impulses at their membranes
(tolbutamide), Ca 2 + ( [ Ca 2 + ] i ). (MCh) [ Ca 2 + ] i, [ Ca 2 + ] i Tsinghua Tongfang Optical Disc Co., Ltd. All rights reserved.
![(tolbutamide), Ca 2 + ( [ Ca 2 + ] i ). (MCh) [ Ca 2 + ] i, [ Ca 2 + ] i Tsinghua Tongfang Optical Disc Co., Ltd. All rights reserved. (tolbutamide), Ca 2 + ( [ Ca 2 + ] i ). (MCh) [ Ca 2 + ] i, [ Ca 2 + ] i Tsinghua Tongfang Optical Disc Co., Ltd. All rights reserved.](/thumbs/93/111981675.jpg) 44 19 1999 10 mental Botany, 1987, 38 : 1174 1181 4 Zhang J, Daveis W J Abscisic acid produced in dehydrating roots may be the plant to measure the water status of the soil Plant Cell and Environment,
44 19 1999 10 mental Botany, 1987, 38 : 1174 1181 4 Zhang J, Daveis W J Abscisic acid produced in dehydrating roots may be the plant to measure the water status of the soil Plant Cell and Environment,
Particles with opposite charges (positives and negatives) attract each other, while particles with the same charge repel each other.
 III. NEUROPHYSIOLOGY A) REVIEW - 3 basic ideas that the student must remember from chemistry and physics: (i) CONCENTRATION measure of relative amounts of solutes in a solution. * Measured in units called
III. NEUROPHYSIOLOGY A) REVIEW - 3 basic ideas that the student must remember from chemistry and physics: (i) CONCENTRATION measure of relative amounts of solutes in a solution. * Measured in units called
The molecular basis of cardiac mechanics: Regulation of motor unit recruitment.
 1of 4 The molecular basis of cardiac mechanics: Regulation of motor unit recruitment. C. Levy, A. Landesberg Department of Biomedical Engineering, Technion-Israel Institute of Technology, Haifa, Israel.
1of 4 The molecular basis of cardiac mechanics: Regulation of motor unit recruitment. C. Levy, A. Landesberg Department of Biomedical Engineering, Technion-Israel Institute of Technology, Haifa, Israel.
Nervous System Organization
 The Nervous System Chapter 44 Nervous System Organization All animals must be able to respond to environmental stimuli -Sensory receptors = Detect stimulus -Motor effectors = Respond to it -The nervous
The Nervous System Chapter 44 Nervous System Organization All animals must be able to respond to environmental stimuli -Sensory receptors = Detect stimulus -Motor effectors = Respond to it -The nervous
Application of density estimation methods to quantal analysis
 Application of density estimation methods to quantal analysis Koichi Yoshioka Tokyo Medical and Dental University Summary There has been controversy for the quantal nature of neurotransmission of mammalian
Application of density estimation methods to quantal analysis Koichi Yoshioka Tokyo Medical and Dental University Summary There has been controversy for the quantal nature of neurotransmission of mammalian
Nerve Signal Conduction. Resting Potential Action Potential Conduction of Action Potentials
 Nerve Signal Conduction Resting Potential Action Potential Conduction of Action Potentials Resting Potential Resting neurons are always prepared to send a nerve signal. Neuron possesses potential energy
Nerve Signal Conduction Resting Potential Action Potential Conduction of Action Potentials Resting Potential Resting neurons are always prepared to send a nerve signal. Neuron possesses potential energy
Disulfonic Stilbene Derivatives Open the Ca2+ Release Channel of Sarcoplasmic Reticulum1
 J. Biochem. 106, 401-405 (1989) Disulfonic Stilbene Derivatives Open the Ca2+ Release Channel of Sarcoplasmic Reticulum1 Takashi Kawasaki and Michiki Kasai Department of Biophysical Engineering, Faculty
J. Biochem. 106, 401-405 (1989) Disulfonic Stilbene Derivatives Open the Ca2+ Release Channel of Sarcoplasmic Reticulum1 Takashi Kawasaki and Michiki Kasai Department of Biophysical Engineering, Faculty
6.3.4 Action potential
 I ion C m C m dφ dt Figure 6.8: Electrical circuit model of the cell membrane. Normally, cells are net negative inside the cell which results in a non-zero resting membrane potential. The membrane potential
I ion C m C m dφ dt Figure 6.8: Electrical circuit model of the cell membrane. Normally, cells are net negative inside the cell which results in a non-zero resting membrane potential. The membrane potential
This script will produce a series of pulses of amplitude 40 na, duration 1ms, recurring every 50 ms.
 9.16 Problem Set #4 In the final problem set you will combine the pieces of knowledge gained in the previous assignments to build a full-blown model of a plastic synapse. You will investigate the effects
9.16 Problem Set #4 In the final problem set you will combine the pieces of knowledge gained in the previous assignments to build a full-blown model of a plastic synapse. You will investigate the effects
TISSUE. A) Types. (i)
 MUSCLES & MUSCLE TISSUE I. OVERVIEW - Muscle ( little mouse ) - tissue designed to cause movementt thru contraction ( shortening ). A) Types - There are some SIMILARITIES between muscle types: (i) All
MUSCLES & MUSCLE TISSUE I. OVERVIEW - Muscle ( little mouse ) - tissue designed to cause movementt thru contraction ( shortening ). A) Types - There are some SIMILARITIES between muscle types: (i) All
Modeling. EC-Coupling and Contraction
 Bioeng 6460 Electrophysiology and Bioelectricity Modeling of EC-Coupling and Contraction Frank B. Sachse fs@cvrti.utah.edu Overview Recapitulation Group Work Excitation-Contraction Coupling Hill s Muscle
Bioeng 6460 Electrophysiology and Bioelectricity Modeling of EC-Coupling and Contraction Frank B. Sachse fs@cvrti.utah.edu Overview Recapitulation Group Work Excitation-Contraction Coupling Hill s Muscle
