Table 1. ODEs used in the model based on mass balances.
|
|
|
- Sharon McCoy
- 5 years ago
- Views:
Transcription
1 Table. ODEs used in the model based on mass balances. ODE s _ = _ 6 = + _ 6 = + + = +! 3 = +2!!$ = +!$ %&' ( = _ + %&' ) *+ = + _, - +2 ),$.(/0 = +,!%!-.(023 = + - $!. 56..(023_ = +! ,3 8.3(20 = + 9 = + + _; = + ; _ = _; + ; ( =+,$ (h.30 = +!%.(.( = +!- 2 = +! +2 6= 56.!$ + 2_ = 6= 56.. =! +!- = %&' + ) 3. = +2!% + $ +!$ +,$ /0 = +!-, +!% 0+9.( = +,
2 Table 2. Initial and optimized kinetic model parameters. Enzyme Index Parameter Initial PTS_Glc ATPase 8 0 pi Trsp PGI PFK FBA GAPDH ENO _.. _!! "# $ "$ % &' &' % () ( *+, *+, value Source [3] [4] [3] [4] [5] [5] Estimated value 3.7 mmol/s 0.07 mm.7 mm 0.28 mm mm 0.3 mm.96 mm 3.29 mm/s 4.34 mm 3.60 mmol/s 6 mm 0.9 mm 2 mm 0.75 mm 0.30 mm 2.68 mm/s 3.4 mm 0.20 mm 8.36 mm/s 0.74 mm mm.02 mm mm 56.3 mm/s 0.30 mm 0. mm 30.0 mm/s mm 0.8 mm 0.64 mm mm 6.75 mm 29.3 mm/s 0.4 mm 0.75 mm mm.39 mm
3 PYK LDH PFL AE ACK PDC # -#.&' () ( ". /0 ". % /0 0 * /0 /0 )(0 () ( /# /0 /0 &/ &/ 0( [7] [8] [9] [] [0] 2.22 mm/s mm 3.70 mm 3.08 mm mm 0.33 mm mm mmol/s 0.08 mm mm 94.2 mm 0.4 mm 3.08 mm 0.0 mm 0.2 mm mmol/s mm 7.34 mm mm 5.78 mm 2.2 mmol/s 6.28 mm 7.38 mm mm 2.28 mm 0.43 mm.3 mm 3.84 mmol/s.7 mm 4.6 mm 6 mm 5 mm 0.7 mm 0.35 mm/s mm 0.26 mm
4 BDH 82 $&' $&' 0( (0 () ( mmol/s 5.62 mm.8 mm 3.55 mm.30 mm MPD 88 2& 200 [2] & 3 () ( [2] [2] [2] [2] [2] [2].33 mm/s mm 0.32 mm mm mm 0.37 mm MP ((0 0.2 [3] 3.49 mm/s 3.52 mm 0.24 mm PTS_Mtl ((0_* 4.45 mmol/s 0.36 mm 0.03 mm 2.2 mm 0.34 mm Ace. Trsp (.60 mmol/s.89 mm Mtl. Trsp (_* 2.. ((0_* (( mm.62 mmol/s 0.94 mm mm FBPase "$ mm/s.9 mm 0.4 mm 0.0 mm Parameters with missing reference or not estimated represented by. Here the initial V max values units are given in mm/s and the initial K m values in mm. All the abbreviations for the enzymes are explained in nomenclature. Reference List. Levering J, Musters MWJM, Bekker M, Bellomo D, Fiedler T, de Vos WM et al.: Role of phoshate in the central metabolism of two lactic acid bacteria - a comparative systems biology approach. FEBS J 202, 279:
5 2. Brown AT, Bowles RD: Polyol Metabolism by A Caries-Conducive Streptococcus - Purification and Properties of A Nicotinamide Adenine Dinucleotide-Dependent Mannitol--Phosphate Dehydrogenase. Infection and Immunity 977, 6: Pettersson G: What Metabolite Levels May be Evolutionarily Reached in the Glycolytic Pathway. European Journal of Biochemistry 990, 94: Gao H, Chen Y, Leary JA: Kinetic measurements of phosphoglucose isomerase and phosphomannose isomerase by direct analysis of phosphorylated aldose-ketose isomers using tandem mass spectrometry. International Journal of Mass Spectrometry 2005, 240: Hansen T, Schonheit P: Purification and properties of the first-identified, archaeal, ATP-dependent 6-phosphofructokinase, an extremely thermophilic non-allosteric enzyme, from the hyperthermophile Desulfurococcus amylolyticus. Archives of Microbiology 2000, 73: Hoefnagel MHN, Starrenburg MJC, Martens DE, Hugenholtz J, Kleerebezem M, Van Swam II et al.: Metabolic engineering of lactic acid bacteria, the combined approach: kinetic modelling, metabolic control and experimental analysis. Microbiology 2002, 48: Andersen AZ, Carvalho AL, Neves AR, Santos H, Kummer U, Olsen LF: The metabolic ph response in Lactococcus lactis: An integrative experimental and modelling approach. Computational Biology and Chemistry 2009, 33: Takahashi S, Abbe K, Yamada T: Purification of Pyruvate Formate-Lyase from Streptococcus-Mutans and Its Regulatory Properties. Journal of Bacteriology 982, 49: Asanuma N, Hino T: Effects of ph and energy supply on activity and amount of pyruvate formate-lyase in Streptococcus bovis. Applied and Environmental Microbiology 2000, 66: Tittmann K, Vyazmensky M, Hubner G, Barak Z, Chipman DM: The carboligation reaction of acetohydroxyacid synthase II: Steady-state intermediate distributions in wild type and mutants by NMR. Proceedings of the National Academy of Sciences of the United States of America 2005, 02: Hoefnagel MHN, van der Burgt A, Martens DE, Hugenholtz J, Snoep JL: Time dependent responses of glycolytic intermediates in a detailed glycolytic model of Lactococcus lactis during glucose run-out experiments. Molecular Biology Reports 2002, 29: Wolff JB, Kaplan NO: D-Mannitol -Phosphate Dehydrogenase from Escherichia-Coli. Journal of Biological Chemistry 956, 28: Schomburg I, Chang A, Schomburg D: BRENDA, enzyme data and metabolic information. Nucleic Acids Research 2002, 30:
6 Table 3. Initial conditions of state variables (metabolites concentrations) for 40mM glucose pulse in the kinetic model. Metabolite Initial concentration Source (mm) glc_ext 40 experimental g6p 0 assumed f6p 0 assumed fbp 5.3 experimental g3p 0 experimental bpg.26 estimated pep 2.48 estimated pyr 0 experimental lactate 0 experimental acetoin 0 experimental acetoin_ext 0 experimental 2,3-butanediol 0 experimental acetcoa 0 experimental CoA ethanol 0 experimental formate 0 experimental acetate 0 experimental mp 0 experimental mannitol 0 experimental mannitol_ext 0 experimental atp 4.89 estimated adp estimated nad 4.67 experimental nadh estimated pi experimental pi_ext 50 experimental All the abbreviations for the metabolites are explained in nomenclature. Reference List. Hoefnagel MHN, van der Burgt A, Martens DE, Hugenholtz J, Snoep JL: Time dependent responses of glycolytic intermediates in a detailed glycolytic model of Lactococcus lactis during glucose run-out experiments. Molecular Biology Reports 2002, 29: 57-6.
7 Table 4. Kinetic rate expressions used in the model. Phosphotransferase System (PTS_Glc): = [pi] +[pi] +[fbp] _ [glc_ext] ' _() [pep] + + [glc_ext] ' _() +[pep] ++ + [g6p] '. +[pyr] 2 Phosphoglucose isomerase (PGI): Phosphofructokinase (PFK): 4 = 4 4 [g6p] ' [f6p] '. + [g6p] '. +[f6p]. 67 = 67 [f6p]. [atp] ) + [f6p]. +[atp] )+ + [fbp] +[adp] : Fructose-,6-bisphosphate phosphatase (FBPase): 6;<+ = 6;<+ [fbp] + [fbp] + +[f6p]. +[pi] Fructose-,6-bisphosphate aldolase (FBA): 6;= = 6;=[fbp] 6;= 6;= +5 [>3@] A + [fbp] +[g3p] 'B + [g3p] A 'B Glyceraldehyde-3-phosphate dehydrogenase (GAPDH): =CD = =CD[g3p] [nad][pi] 'B F: +5 =CD =CD [bpg] 'B [nadh] F: + [g3p] 'B +[pi] H+[nad] F: I+ +[bpg] ' H+[nadh] F:J I Enolase (ENO): (KL = (KL[bpg][adp] F: 'B (KL (KL +5 [pep][atp] ' : + [bpg] ' +[adp] :+ +[pep] + + [atp] )
8 Pyruvate kinase (PYK): M7 = [fbp] +[fbp] N O P FQRS V O P FQRS +T[pi]U FQRS M7 [adp] : [pep] + + [adp] : +[pep] ++ + [atp] ) + [pyr] 2 L-lactate dehydrogenase (LDH): WCD = [fbp] +[fbp] +[pi] WCD [pyr] 2H [nadh] F:J I + [pyr] 2H+ [nadh] F:J I+H+[lactate] ))+ IH+[nad] F: I Pyruvate dehydrogenase (PFL): 6W = 'B 'B +[g3p] 6W[pyr] [CoA] [\= 6W 6W 2 +5 [acetcoa][formate] 2 [\= + [pyr] 2H+ [CoA] [\= I+H+[acetCoA] ^_`abcd I +[formate] \2)+ Alcohol dehydrogenase (AE): =( = ) ) +[atp] =( H [acetcoa] A +)[\= IH[nadh] F:J I H+ [acetcoa] A +)[\= I +[nadh] F:J +H[nadh] F:J I +H+ [ethanol] A +)JF\ IH+[CoA] [\= I +[nad] F: +H[nad] F: I Acetate kinase (ACK): =[7 = =[7 H [acetcoa] +)[\= I [adp] : H+ [acetcoa] +)[\= I +[adp] : +H+[acetate] +))+I + [atp] )H+ [CoA] [\= I Pyruvate decarboxylase (PDC): C[ = C [pyr] A 2 C [acetoin] C [pyr] 2 + [pyr] A 2 +H+ [acetoin] +)\F I
9 2,3-Butanediol dehydrogenase (BDH): ;C H [acetoin] +)\F IH[nadh] F:J I [2,3 butanediol] [nad] ;C +5 +)\F F:J ;CD = H+ [acetoin] +)\F IH+[nadh] F:J I+H+[2,3 butanediol] h)f+:\ IH+ [nad] F: I ;C Acetoin transport (Ace. Trsp.): = +.2<. = = +.2<. H [acetoin] +)\F I H+ [acetoin] +)\F I+ +[acetoin_ext] +)\F_() Mannitol--Phosphate dehydrogenase (MPD):. jc =. +[f6p] jc[f6p] [nadh] F:J. jc +5 jc [mp]. [nad] F:J + [f6p]. H+[nadh] F:JI+ +[mp] k H+[nad] F: I Mannitol--Phosphatase (MP): j = j [mp] k + [mp] k +H+[mannitol] FF)\ I Phosphotransferase System (PTS_Mtl): _j) = _j) [mannitol_ext] FF)\_() [pep] + + [mannitol_ext] FF)\_() +[pep] ++ + [mp] k +[pyr] 2 Mannitol transport (Mtl. Trsp.): j).2<. = Phosphate transport (pi Trsp.): j).2<. H [mannitol] FF)\ I H+ [mannitol] FF)\ I+ +[mannitol_ext] FF)\_() 2<. = +[pi] 2<. [atp] ) [pi_ext] _() + [atp] ) + [pi_ext] _() + +[adp] : +[pi] + [pi] A
10 ATP phosphatase (ATPase): =<+ = =<+ o n m F [atp] pqqrst ) [atp] ) F pqqrst + w v u
11 Figure. Variance analysis of the parameter set values of the kinetic model. For a better visualization the variance of every parameter are plotted with a logarithmic scale. The parameter indices correspond to the reaction numbers shown in Additional File 2.
12 Figure 2. Simulation results for the initial metabolite concentrations and estimated kinetic parameter values listed in Additional File 3 and 2, respectively.
13 Figure 3. Correlation values of sensitivities between all pairs of fits from wilt-type strain for 2,3-butanediol (plots in lower triangle) and mannitol (plots in upper triangle).
14 Figure 4. Correlation values of sensitivities between all pairs of fits from LDH mutant strain for 2,3-butanediol (plots in lower triangle) and mannitol (plots in upper triangle).
15 Figure 5. Correlation values of sensitivities between all pairs of fits from LDH/PTS Mtl mutant strain for 2,3-butanediol (plots in lower triangle) and mannitol (plots in upper triangle).
16
17
18 Figure 6. Heat maps showing the integrated response coefficients to variation in V max parameters for all ten fits.
Parameter Estimation in Metabolic Reaction Networks
 HELSINKI UNIVERSITY OF TECHNOLOGY Engineering Physics and Mathematics Systems Analysis Laboratory Mat-2.108 Independent research projects in applied mathematics Parameter Estimation in Metabolic Reaction
HELSINKI UNIVERSITY OF TECHNOLOGY Engineering Physics and Mathematics Systems Analysis Laboratory Mat-2.108 Independent research projects in applied mathematics Parameter Estimation in Metabolic Reaction
Giving you the energy you need!
 Giving you the energy you need! Use your dominant hand Open and close the pin (with your thumb and forefinger) as many times as you can for 20 seconds while holding the other fingers straight out! Repeat
Giving you the energy you need! Use your dominant hand Open and close the pin (with your thumb and forefinger) as many times as you can for 20 seconds while holding the other fingers straight out! Repeat
Structural Analysis of Expanding Metabolic Networks
 Genome Informatics 15(1): 35 45 (24) 35 Structural Analysis of Expanding Metabolic Networks Oliver Ebenhöh oliver.ebenhoeh@rz.hu-berlin.de Reinhart Heinrich reinhart.heinrich@rz.hu-berlin.de Thomas Handorf
Genome Informatics 15(1): 35 45 (24) 35 Structural Analysis of Expanding Metabolic Networks Oliver Ebenhöh oliver.ebenhoeh@rz.hu-berlin.de Reinhart Heinrich reinhart.heinrich@rz.hu-berlin.de Thomas Handorf
CHAPTER 15 Metabolism: Basic Concepts and Design
 CHAPTER 15 Metabolism: Basic Concepts and Design Chapter 15 An overview of Metabolism Metabolism is the sum of cellular reactions - Metabolism the entire network of chemical reactions carried out by living
CHAPTER 15 Metabolism: Basic Concepts and Design Chapter 15 An overview of Metabolism Metabolism is the sum of cellular reactions - Metabolism the entire network of chemical reactions carried out by living
Biochemistry 3300 Problems (and Solutions) Metabolism I
 (1) Provide a reasonable systematic name for an enzyme that catalyzes the following reaction: fructose + ATP > fructose-1 phosphate + ADP (2) The IUBMB has a developed a set of rules for classifying enzymes
(1) Provide a reasonable systematic name for an enzyme that catalyzes the following reaction: fructose + ATP > fructose-1 phosphate + ADP (2) The IUBMB has a developed a set of rules for classifying enzymes
Enzymes I. Dr. Mamoun Ahram Summer semester,
 Enzymes I Dr. Mamoun Ahram Summer semester, 2017-2018 Resources Mark's Basic Medical Biochemistry Other resources NCBI Bookshelf: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=books The Medical Biochemistry
Enzymes I Dr. Mamoun Ahram Summer semester, 2017-2018 Resources Mark's Basic Medical Biochemistry Other resources NCBI Bookshelf: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=books The Medical Biochemistry
Chapter 15 part 2. Biochemistry I Introduction to Metabolism Bioenergetics: Thermodynamics in Biochemistry. ATP 4- + H 2 O ADP 3- + P i + H +
 Biochemistry I Introduction to Metabolism Bioenergetics: Thermodynamics in Biochemistry ATP 4- + 2 ADP 3- + P i 2- + + Chapter 15 part 2 Dr. Ray 1 Energy flow in biological systems: Energy Transformations
Biochemistry I Introduction to Metabolism Bioenergetics: Thermodynamics in Biochemistry ATP 4- + 2 ADP 3- + P i 2- + + Chapter 15 part 2 Dr. Ray 1 Energy flow in biological systems: Energy Transformations
Dynamic modeling and control of the main metabolism in Lactic Acid Bacteria
 Dynamic modeling and control of the main metabolism in Lactic Acid Bacteria Bhabuk Koirala Dissertation submitted to obtain Masters Degree in Biotechnology Examination Committee Chairperson: Supervisors:
Dynamic modeling and control of the main metabolism in Lactic Acid Bacteria Bhabuk Koirala Dissertation submitted to obtain Masters Degree in Biotechnology Examination Committee Chairperson: Supervisors:
Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Outer Glycolysis mitochondrial membrane Glucose ATP
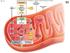 Fig. 7.5 uter Glycolysis mitochondrial membrane Glucose Intermembrane space xidation Mitochondrial matrix Acetyl-oA Krebs FAD e NAD + FAD Inner mitochondrial membrane e Electron e Transport hain hemiosmosis
Fig. 7.5 uter Glycolysis mitochondrial membrane Glucose Intermembrane space xidation Mitochondrial matrix Acetyl-oA Krebs FAD e NAD + FAD Inner mitochondrial membrane e Electron e Transport hain hemiosmosis
2015 AP Biology PRETEST Unit 3: Cellular Energetics Week of October
 Name: Class: _ Date: _ 2015 AP Biology PRETEST Unit 3: Cellular Energetics Week of 19-23 October Multiple Choice Identify the choice that best completes the statement or answers the question. 1) Which
Name: Class: _ Date: _ 2015 AP Biology PRETEST Unit 3: Cellular Energetics Week of 19-23 October Multiple Choice Identify the choice that best completes the statement or answers the question. 1) Which
Gene Regulatory Network Identification
 Gene Regulatory Network Identification Korkut Uygun and Yinlun Huang Department of Chemical Engineering and Materials Science Wayne State University AIChE Annual National Meeting San Francisco November
Gene Regulatory Network Identification Korkut Uygun and Yinlun Huang Department of Chemical Engineering and Materials Science Wayne State University AIChE Annual National Meeting San Francisco November
Lecture Series 9 Cellular Pathways That Harvest Chemical Energy
 Lecture Series 9 Cellular Pathways That Harvest Chemical Energy Reading Assignments Review Chapter 3 Energy, Catalysis, & Biosynthesis Read Chapter 13 How Cells obtain Energy from Food Read Chapter 14
Lecture Series 9 Cellular Pathways That Harvest Chemical Energy Reading Assignments Review Chapter 3 Energy, Catalysis, & Biosynthesis Read Chapter 13 How Cells obtain Energy from Food Read Chapter 14
37 Modelling and experimental evidence for two separate steady states in the photosynthetic Calvin cycle
 37 Modelling and experimental evidence for two separate steady states in the photosynthetic Calvin cycle M.G. Poolman and D.A. Fell School of Biology and Molecular Science, Oxford Brookes University, Headington,
37 Modelling and experimental evidence for two separate steady states in the photosynthetic Calvin cycle M.G. Poolman and D.A. Fell School of Biology and Molecular Science, Oxford Brookes University, Headington,
Welcome to Class 8! Introductory Biochemistry! Announcements / Reminders! Midterm TA led Review Sessions!
 Announcements / Reminders Midterm TA led Review Sessions Welcome to Class 8 Sunday, February 23 from 8-10pm Location: Science Center Main Room (315) Office Hours Prof Salomon: SFH 270 on Thursday Feb 20,
Announcements / Reminders Midterm TA led Review Sessions Welcome to Class 8 Sunday, February 23 from 8-10pm Location: Science Center Main Room (315) Office Hours Prof Salomon: SFH 270 on Thursday Feb 20,
Applications of Free Energy. NC State University
 Chemistry 433 Lecture 15 Applications of Free Energy NC State University Thermodynamics of glycolysis Reaction kj/mol D-glucose + ATP D-glucose-6-phosphate + ADP ΔG o = -16.7 D-glucose-6-phosphate p D-fructose-6-phosphate
Chemistry 433 Lecture 15 Applications of Free Energy NC State University Thermodynamics of glycolysis Reaction kj/mol D-glucose + ATP D-glucose-6-phosphate + ADP ΔG o = -16.7 D-glucose-6-phosphate p D-fructose-6-phosphate
Exam 3 Review (4/12/2011) Lecture note excerpt covering lectures (Exam 3 topics: Chapters 8, 12, 14 & 15)
 Exam 3 Review (4/12/2011) Lecture note excerpt covering lectures 17-23 (Exam 3 topics: Chapters 8, 12, 14 & 15) Enzyme Kinetics, Inhibition, and Regulation Chapter 12 Enzyme Kinetics When the concentration
Exam 3 Review (4/12/2011) Lecture note excerpt covering lectures 17-23 (Exam 3 topics: Chapters 8, 12, 14 & 15) Enzyme Kinetics, Inhibition, and Regulation Chapter 12 Enzyme Kinetics When the concentration
I. Flow of Energy in Living Things II. Laws of Thermodynamics & Free Energy III. Activation Energy IV. Enzymes V. Reaction Coupling VI.
 Chapter 6 Energy & Metabolism I. Flow of Energy in Living Things II. Laws of Thermodynamics & Free Energy III. Activation Energy IV. Enzymes V. Reaction Coupling VI. Metabolism I. Flow of Energy in Living
Chapter 6 Energy & Metabolism I. Flow of Energy in Living Things II. Laws of Thermodynamics & Free Energy III. Activation Energy IV. Enzymes V. Reaction Coupling VI. Metabolism I. Flow of Energy in Living
Description of the algorithm for computing elementary flux modes
 Description of the algorithm for computing elementary flux modes by Stefan Schuster, Thomas Dandekar,, and David Fell Department of Bioinformatics, Max Delbrück Centre for Molecular Medicine D-9 Berlin-Buch,
Description of the algorithm for computing elementary flux modes by Stefan Schuster, Thomas Dandekar,, and David Fell Department of Bioinformatics, Max Delbrück Centre for Molecular Medicine D-9 Berlin-Buch,
Evolutionary Optimization of Metabolic Pathways. Theoretical Reconstruction of the Stoichiometry of ATP and NADH Producing Systems
 Bulletin of Mathematical Biology (2001) 63, 21 55 doi:10.1006/bulm.2000.0197 Available online at http://www.idealibrary.com on Evolutionary Optimization of Metabolic Pathways. Theoretical Reconstruction
Bulletin of Mathematical Biology (2001) 63, 21 55 doi:10.1006/bulm.2000.0197 Available online at http://www.idealibrary.com on Evolutionary Optimization of Metabolic Pathways. Theoretical Reconstruction
Quantitative, scalable discrete event simulation of metabolic pathways
 From: ISMB-99 Proceedings. Copyright 1999, AAAI (www.aaai.org). All rights reserved. Quantitative, scalable discrete event simulation of metabolic pathways Peter A. Meric Basser Department of Computer
From: ISMB-99 Proceedings. Copyright 1999, AAAI (www.aaai.org). All rights reserved. Quantitative, scalable discrete event simulation of metabolic pathways Peter A. Meric Basser Department of Computer
2) This is a learning problem. You will learn something. You will appreciate this information. If not soon, than someday. Ready?
 Problem Set 2 Hello Class. This is a Big Round problem set. Lots of word problems. I do this because science is a language. Much of it, and medicine too, is transacted as spoken word: seminars, grand rounds,
Problem Set 2 Hello Class. This is a Big Round problem set. Lots of word problems. I do this because science is a language. Much of it, and medicine too, is transacted as spoken word: seminars, grand rounds,
Reconstruction and Analysis of Metabolic Networks
 1 Reconstruction and Analysis of Metabolic Networks 2 Outline What is a Reconstruction? Data Collection Interactions Between Network Components Special Considerations Applications Genome-scale Metabolic
1 Reconstruction and Analysis of Metabolic Networks 2 Outline What is a Reconstruction? Data Collection Interactions Between Network Components Special Considerations Applications Genome-scale Metabolic
Winter School in Mathematical & Computational Biology
 1 Network reconstruction, topology and feasible solution space From component to systems biology Component biology Systems biology Component view Systems view Needed homeostasis Function S+E X E+P Reaction
1 Network reconstruction, topology and feasible solution space From component to systems biology Component biology Systems biology Component view Systems view Needed homeostasis Function S+E X E+P Reaction
Cellular Energy: Respiration. Goals: Anaerobic respiration
 Cellular Energy: Respiration Anaerobic respiration Goals: Define and describe the 3 sets of chemical reactions that comprise aerobic cellular respiration Describe the types of anaerobic respiration Compare
Cellular Energy: Respiration Anaerobic respiration Goals: Define and describe the 3 sets of chemical reactions that comprise aerobic cellular respiration Describe the types of anaerobic respiration Compare
Assimilation of Carbon in Plants
 Module 0210101: Molecular Biology and Biochemistry of the Cell Lecture 18 Assimilation of Carbon in Plants Dale Sanders 17 March 2009 Objectives By the end of the lecture you should understand: 1. How
Module 0210101: Molecular Biology and Biochemistry of the Cell Lecture 18 Assimilation of Carbon in Plants Dale Sanders 17 March 2009 Objectives By the end of the lecture you should understand: 1. How
Constraint-Based Workshops
 Constraint-Based Workshops 2. Reconstruction Databases November 29 th, 2007 Defining Metabolic Reactions ydbh hslj ldha 1st level: Primary metabolites LAC 2nd level: Neutral Formulas C 3 H 6 O 3 Charged
Constraint-Based Workshops 2. Reconstruction Databases November 29 th, 2007 Defining Metabolic Reactions ydbh hslj ldha 1st level: Primary metabolites LAC 2nd level: Neutral Formulas C 3 H 6 O 3 Charged
Part II => PROTEINS and ENZYMES. 2.5 Enzyme Properties 2.5a Enzyme Nomenclature 2.5b Transition State Theory
 Part II => PROTEINS and ENZYMES 2.5 Enzyme Properties 2.5a Enzyme Nomenclature 2.5b Transition State Theory Section 2.5a: Enzyme Nomenclature Synopsis 2.5a - Enzymes are biological catalysts they are almost
Part II => PROTEINS and ENZYMES 2.5 Enzyme Properties 2.5a Enzyme Nomenclature 2.5b Transition State Theory Section 2.5a: Enzyme Nomenclature Synopsis 2.5a - Enzymes are biological catalysts they are almost
Transduction of Intracellular and Intercellular Dynamics in Yeast Glycolytic Oscillations
 Biophysical Journal Volume 78 March 2000 1145 1153 1145 Transduction of Intracellular and Intercellular Dynamics in Yeast Glycolytic Oscillations Jana Wolf,* Jutta Passarge,, Oscar J.G. Somsen, Jacky L.
Biophysical Journal Volume 78 March 2000 1145 1153 1145 Transduction of Intracellular and Intercellular Dynamics in Yeast Glycolytic Oscillations Jana Wolf,* Jutta Passarge,, Oscar J.G. Somsen, Jacky L.
1) What is the one letter and the three letter abbreviations for the poly peptide shown
 Lecture 1 Short problems 1) What is the one letter and the three letter abbreviations for the poly peptide shown 2) How many peptide bonds does this polypeptide possess? 3) Circle the peptide bonds 4)
Lecture 1 Short problems 1) What is the one letter and the three letter abbreviations for the poly peptide shown 2) How many peptide bonds does this polypeptide possess? 3) Circle the peptide bonds 4)
Enzymatic Assay of GLYCOGEN SYNTHASE (EC )
 PRINCIPLE: UDPG + (Glycogen) n Glycogen Synthase > UDP + (Glycogen) n+1 UDP + PEP PK > UTP + Pyruvate Pyruvate + ß-NADH LDH > Lactate + ß-NAD Abbreviations: UDPG = Uridine 5'-Diphosphoglucose UDP = Uridine
PRINCIPLE: UDPG + (Glycogen) n Glycogen Synthase > UDP + (Glycogen) n+1 UDP + PEP PK > UTP + Pyruvate Pyruvate + ß-NADH LDH > Lactate + ß-NAD Abbreviations: UDPG = Uridine 5'-Diphosphoglucose UDP = Uridine
Determining Enzyme Kinetics for Systems Biology with Nuclear Magnetic Resonance Spectroscopy
 Metabolites 2012, 2, 818-843; doi:10.3390/metabo2040818 Article OPEN ACCESS metabolites ISSN 2218-1989 www.mdpi.com/journal/metabolites Determining Enzyme Kinetics for Systems Biology with Nuclear Magnetic
Metabolites 2012, 2, 818-843; doi:10.3390/metabo2040818 Article OPEN ACCESS metabolites ISSN 2218-1989 www.mdpi.com/journal/metabolites Determining Enzyme Kinetics for Systems Biology with Nuclear Magnetic
Exam 4 April 15, 2005 CHEM 3511 Print Name: KEY Signature
 1) (8 pts) General Properties of Enzymes. Give four properties of enzymaticallycatalyzed reactions. The answers should indicate how enzymatic reactions differ from non-enzymatic reactions. Write four only
1) (8 pts) General Properties of Enzymes. Give four properties of enzymaticallycatalyzed reactions. The answers should indicate how enzymatic reactions differ from non-enzymatic reactions. Write four only
Mechanism of CO 2 Fixation: Rubisco Step 1
 dark means light-indendent! The Dark Reaction: Carbon Fixation Requires Rubisco: Ribulose-1,5-bisphosphate carboxylase RuBPi + C 6 x 3PG MW = 550, 000 g/mol Subunits eight small (14,000 g/mol) eight large
dark means light-indendent! The Dark Reaction: Carbon Fixation Requires Rubisco: Ribulose-1,5-bisphosphate carboxylase RuBPi + C 6 x 3PG MW = 550, 000 g/mol Subunits eight small (14,000 g/mol) eight large
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 AP Exam Chapters 9 and 10; Photosynthesis and Respiration Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Carbon dioxide (CO2) is released
AP Exam Chapters 9 and 10; Photosynthesis and Respiration Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Carbon dioxide (CO2) is released
Glycolysis and Fermentation. Chapter 8
 Glycolysis and Fermentation Chapter 8 Cellular Respiration and Photosynthesis 0 Things to know in these chapters 0 Names and order of the processes 0 Reactants and products of each process 0 How do they
Glycolysis and Fermentation Chapter 8 Cellular Respiration and Photosynthesis 0 Things to know in these chapters 0 Names and order of the processes 0 Reactants and products of each process 0 How do they
Oxidative Phosphorylation versus. Photophosphorylation
 Photosynthesis Oxidative Phosphorylation versus Photophosphorylation Oxidative Phosphorylation Electrons from the reduced cofactors NADH and FADH 2 are passed to proteins in the respiratory chain. In eukaryotes,
Photosynthesis Oxidative Phosphorylation versus Photophosphorylation Oxidative Phosphorylation Electrons from the reduced cofactors NADH and FADH 2 are passed to proteins in the respiratory chain. In eukaryotes,
This is an example of cellular respiration, which can be used to make beer and wine using different metabolic pathways For these reasons we call this
 Chapter 6 Carvings from ancient Egypt show barley being crushed and mixed with water (left) and then put into closed vessels (centre) where airless conditions are suitable for the production of alcohol
Chapter 6 Carvings from ancient Egypt show barley being crushed and mixed with water (left) and then put into closed vessels (centre) where airless conditions are suitable for the production of alcohol
Enzymatic Assay of HYPOXANTHINE-GUANINE PHOSPHORIBOSYL TRANSFERASE (EC )
 PRINCIPLE: PRPP + Guanine HGPRT > GMP + Pyrophosphate Mg ++ GMP + ATP GK > GDP + ADP ADP + PEP PK > ATP + Pyruvate GDP + PEP PK > GTP + Pyruvate 2 Pyruvate + 2 ß-NADH LDH > 2 Lactate + 2 ß-NAD Abbreviations
PRINCIPLE: PRPP + Guanine HGPRT > GMP + Pyrophosphate Mg ++ GMP + ATP GK > GDP + ADP ADP + PEP PK > ATP + Pyruvate GDP + PEP PK > GTP + Pyruvate 2 Pyruvate + 2 ß-NADH LDH > 2 Lactate + 2 ß-NAD Abbreviations
UvA-DARE (Digital Academic Repository)
 UvA-DARE Dgtal Academc Repostory onte-carlo modelng of the central carbon metabolsm of Lactococcus lacts: nsghts nto metabolc regulaton urabto, E.; Verma,.; Bekker,.; Bellomo, D.; Westerhoff, H.V.; Teusnk,
UvA-DARE Dgtal Academc Repostory onte-carlo modelng of the central carbon metabolsm of Lactococcus lacts: nsghts nto metabolc regulaton urabto, E.; Verma,.; Bekker,.; Bellomo, D.; Westerhoff, H.V.; Teusnk,
R-Biopharm AG RIDA CUBE SCAN. The smart analyser. Product Information. R-Biopharm for the safety of your analysis.
 R-Biopharm AG The smart analyser Product Information R-Biopharm for the safety of your analysis. Intended use The R-Biopharm is a new photometric system that allows biochemistry testing in small food laboratories
R-Biopharm AG The smart analyser Product Information R-Biopharm for the safety of your analysis. Intended use The R-Biopharm is a new photometric system that allows biochemistry testing in small food laboratories
RUBISCO > 2 moles of 3-phosphoglycerate Mg +2
 PRINCIPLE: RuDP + CO 2 RUBISCO > 2 moles of 3-phosphoglycerate Mg +2 3-Phosphoglycerate + ATP PGK > Glycerate 1,3-Diphosphate + ADP Glycerate 1,3-Diphosphate + ß-NADH GAPDH > Glyceraldehyde 3-Phosphate
PRINCIPLE: RuDP + CO 2 RUBISCO > 2 moles of 3-phosphoglycerate Mg +2 3-Phosphoglycerate + ATP PGK > Glycerate 1,3-Diphosphate + ADP Glycerate 1,3-Diphosphate + ß-NADH GAPDH > Glyceraldehyde 3-Phosphate
Cellular Respiration: Harvesting Chemical Energy. 9.1 Catabolic pathways yield energy by oxidizing organic fuels
 Cellular Respiration: Harvesting Chemical Energy 9.1 Catabolic pathways yield energy by oxidizing organic fuels 9.2 Glycolysis harvests chemical energy by oxidizing glucose to pyruvate 9.3 The citric acid
Cellular Respiration: Harvesting Chemical Energy 9.1 Catabolic pathways yield energy by oxidizing organic fuels 9.2 Glycolysis harvests chemical energy by oxidizing glucose to pyruvate 9.3 The citric acid
Biochemical Pathways
 Biochemical Pathways Living organisms can be divided into two large groups according to the chemical form in which they obtain carbon from the environment. Autotrophs can use carbon dioxide from the atmosphere
Biochemical Pathways Living organisms can be divided into two large groups according to the chemical form in which they obtain carbon from the environment. Autotrophs can use carbon dioxide from the atmosphere
Enzymatic Assay of GUANYLATE KINASE (EC )
 PRINCIPLE: GMP + ATP Guanylate Kinase > GDP + ADP ADP + PEP Pyruvate Kinase > ATP + Pyruvate GDP + PEP Pyruvate Kinase > GTP + Pyruvate 2 Pyruvate + 2 ß-NADH Lactic Dehydrogenase > 2 Lactate + 2 ß-NAD
PRINCIPLE: GMP + ATP Guanylate Kinase > GDP + ADP ADP + PEP Pyruvate Kinase > ATP + Pyruvate GDP + PEP Pyruvate Kinase > GTP + Pyruvate 2 Pyruvate + 2 ß-NADH Lactic Dehydrogenase > 2 Lactate + 2 ß-NAD
Towards integrated models of regulatory networks: the MetaGenoReg project
 Towards integrated models of regulatory networks: the MetaGenoReg project Hidde de Jong and Daniel Kahn INRIA Grenoble - Rhône-Alpes Hidde.de-Jong@inria.fr http://ibis.inrialpes.fr LBBE, Université de
Towards integrated models of regulatory networks: the MetaGenoReg project Hidde de Jong and Daniel Kahn INRIA Grenoble - Rhône-Alpes Hidde.de-Jong@inria.fr http://ibis.inrialpes.fr LBBE, Université de
An introduction to biochemical reaction networks
 Chapter 1 An introduction to biochemical reaction networks (This part is discussed in the slides). Biochemical reaction network distinguish themselves from general chemical reaction networks in that the
Chapter 1 An introduction to biochemical reaction networks (This part is discussed in the slides). Biochemical reaction network distinguish themselves from general chemical reaction networks in that the
AHL Topic 8 IB Biology Miss Werba
 CELL RESPIRATION & PHOTOSYNTHESIS AHL Topic 8 IB Biology Miss Werba TOPIC 8 CELL RESPIRATION & PHOTOSYNTHESIS 8.1 CELL RESPIRATION 1. STATE that oxidation involves the loss of electrons from an element,
CELL RESPIRATION & PHOTOSYNTHESIS AHL Topic 8 IB Biology Miss Werba TOPIC 8 CELL RESPIRATION & PHOTOSYNTHESIS 8.1 CELL RESPIRATION 1. STATE that oxidation involves the loss of electrons from an element,
Chemistry and Biochemistry 153A Winter Final Exam
 Chemistry and Biochemistry 153A Winter 2010 Final Exam Instructions: (Note that changes or additions to the usual instructions have been underlined.) You will have 3 hours to complete the exam. You may
Chemistry and Biochemistry 153A Winter 2010 Final Exam Instructions: (Note that changes or additions to the usual instructions have been underlined.) You will have 3 hours to complete the exam. You may
Photosynthesis and cellular respirations
 The Introduction of Biology Defining of life Basic chemistry, the chemistry of organic molecules Classification of living things History of cells and Cells structures and functions Photosynthesis and cellular
The Introduction of Biology Defining of life Basic chemistry, the chemistry of organic molecules Classification of living things History of cells and Cells structures and functions Photosynthesis and cellular
Cell population modelling of yeast glycolytic oscillations
 Biochem. J. (2002) 368, 433 446 (Printed in Great Britain) 433 Cell population modelling of yeast glycolytic oscillations Michael A. HENSON* 1, Dirk MU LLER and Matthias REUSS *Department of Chemical Engineering,
Biochem. J. (2002) 368, 433 446 (Printed in Great Britain) 433 Cell population modelling of yeast glycolytic oscillations Michael A. HENSON* 1, Dirk MU LLER and Matthias REUSS *Department of Chemical Engineering,
Feedback and reversibility in substrate-enzyme reactions as discrete event models van Zwieten, D.A.J.; Rooda, J.E.; Armbruster, H.D.; Nagy, J.D.
 Feedback and reversibility in substrate-enzyme reactions as discrete event models van Zwieten, D.A.J.; Rooda, J.E.; Armbruster, H.D.; Nagy, J.D. Published: 01/01/2010 Document Version Publisher s PDF,
Feedback and reversibility in substrate-enzyme reactions as discrete event models van Zwieten, D.A.J.; Rooda, J.E.; Armbruster, H.D.; Nagy, J.D. Published: 01/01/2010 Document Version Publisher s PDF,
Chapter 7: Metabolic Networks
 Chapter 7: Metabolic Networks 7.1 Introduction Prof. Yechiam Yemini (YY) Computer Science epartment Columbia University Introduction Metabolic flux analysis Applications Overview 2 1 Introduction 3 Metabolism:
Chapter 7: Metabolic Networks 7.1 Introduction Prof. Yechiam Yemini (YY) Computer Science epartment Columbia University Introduction Metabolic flux analysis Applications Overview 2 1 Introduction 3 Metabolism:
Be sure to understand:
 Learning Targets & Focus Questions for Unit 6: Bioenergetics Chapter 8: Thermodynamics Chapter 9: Cell Resp Focus Q Ch. 10: Photosynthesis Chapter 8 (141-150) 1. I can explain how living systems adhere
Learning Targets & Focus Questions for Unit 6: Bioenergetics Chapter 8: Thermodynamics Chapter 9: Cell Resp Focus Q Ch. 10: Photosynthesis Chapter 8 (141-150) 1. I can explain how living systems adhere
Review Questions - Lecture 5: Metabolism, Part 1
 Review Questions - Lecture 5: Metabolism, Part 1 Questions: 1. What is metabolism? 2. What does it mean to say that a cell has emergent properties? 3. Define metabolic pathway. 4. What is the difference
Review Questions - Lecture 5: Metabolism, Part 1 Questions: 1. What is metabolism? 2. What does it mean to say that a cell has emergent properties? 3. Define metabolic pathway. 4. What is the difference
Change to Office Hours this Friday and next Monday. Tomorrow (Abel): 8:30 10:30 am. Monday (Katrina): Cancelled (05/04)
 Change to Office Hours this Friday and next Monday Tomorrow (Abel): 8:30 10:30 am Monday (Katrina): Cancelled (05/04) Lecture 10 Proton Gradient-dependent ATP Synthesis Oxidative Phosphorylation Photo-Phosphorylation
Change to Office Hours this Friday and next Monday Tomorrow (Abel): 8:30 10:30 am Monday (Katrina): Cancelled (05/04) Lecture 10 Proton Gradient-dependent ATP Synthesis Oxidative Phosphorylation Photo-Phosphorylation
Supplementary Information
 1 Steady State Analysis of Genetic Regulatory Network incorporating underlying Molecular Mechanisms for Anaerobic Metabolism in Escherichia coli Sumana Srinivasan and K. V. Venkatesh Department of Chemical
1 Steady State Analysis of Genetic Regulatory Network incorporating underlying Molecular Mechanisms for Anaerobic Metabolism in Escherichia coli Sumana Srinivasan and K. V. Venkatesh Department of Chemical
Modelling and Optimisation of Central Metabolism Response to Glucose Pulse
 Modelling and Optimisation of Central Metabolism Response to Glucose Pulse Sonja Čerić, Želimir Kurtanjek University of Zagreb, Faculty of Food Technology and Biotechnology, Pierottijeva 6, 0000 Zagreb,
Modelling and Optimisation of Central Metabolism Response to Glucose Pulse Sonja Čerić, Želimir Kurtanjek University of Zagreb, Faculty of Food Technology and Biotechnology, Pierottijeva 6, 0000 Zagreb,
Sabrina Hoffmann, Andreas Hoppe and Hermann-Georg Holzhutter
 Beilstein-Institut ESCEC, March 19 th 23 rd, 2006, Rüdesheim/Rhein, Germany 91 Decomposition into Minimal Flux Modes: A Novel Flux-Balance Approach to Predict Flux Changes in Metabolic Networks from Changes
Beilstein-Institut ESCEC, March 19 th 23 rd, 2006, Rüdesheim/Rhein, Germany 91 Decomposition into Minimal Flux Modes: A Novel Flux-Balance Approach to Predict Flux Changes in Metabolic Networks from Changes
METABOLISM CHAPTER 04 BIO 211: ANATOMY & PHYSIOLOGY I. Dr. Lawrence G. Altman Some illustrations are courtesy of McGraw-Hill.
 BIO 211: ANATOMY & PHYSIOLOGY I CHAPTER 04 1 Please wait 20 seconds before starting slide show. Mouse click or Arrow keys to navigate. Hit ESCAPE Key to exit. CELLULAR METABOLISM Dr. Lawrence G. Altman
BIO 211: ANATOMY & PHYSIOLOGY I CHAPTER 04 1 Please wait 20 seconds before starting slide show. Mouse click or Arrow keys to navigate. Hit ESCAPE Key to exit. CELLULAR METABOLISM Dr. Lawrence G. Altman
CELL METABOLISM OVERVIEW Keep the big picture in mind as we discuss the particulars!
 BIO 211: ANATOMY & PHYSIOLOGY I CHAPTER 04 CELLULAR METABOLISM 1 Please wait 20 seconds before starting slide show. Mouse click or Arrow keys to navigate. Hit ESCAPE Key to exit. Dr. Lawrence G. Altman
BIO 211: ANATOMY & PHYSIOLOGY I CHAPTER 04 CELLULAR METABOLISM 1 Please wait 20 seconds before starting slide show. Mouse click or Arrow keys to navigate. Hit ESCAPE Key to exit. Dr. Lawrence G. Altman
All organisms require a constant expenditure of energy to maintain the living state - "LIFE".
 CELLULAR RESPIRATION All organisms require a constant expenditure of energy to maintain the living state - "LIFE". Where does the energy come from and how is it made available for life? With rare exception,
CELLULAR RESPIRATION All organisms require a constant expenditure of energy to maintain the living state - "LIFE". Where does the energy come from and how is it made available for life? With rare exception,
Biologic catalysts 1. Shared properties with chemical catalysts a. Enzymes are neither consumed nor produced during the course of a reaction. b.
 Enzyme definition Enzymes are protein catalysts that increase the velocity of a chemical reaction and are not consumed during the reaction they catalyze. [Note: Some types of RNA can act like enzymes,
Enzyme definition Enzymes are protein catalysts that increase the velocity of a chemical reaction and are not consumed during the reaction they catalyze. [Note: Some types of RNA can act like enzymes,
BBS2710 Microbial Physiology. Module 5 - Energy and Metabolism
 BBS2710 Microbial Physiology Module 5 - Energy and Metabolism Topics Energy production - an overview Fermentation Aerobic respiration Alternative approaches to respiration Photosynthesis Summary Introduction
BBS2710 Microbial Physiology Module 5 - Energy and Metabolism Topics Energy production - an overview Fermentation Aerobic respiration Alternative approaches to respiration Photosynthesis Summary Introduction
Department of Chemistry and Biochemistry University of Lethbridge. Biochemistry II. Bioenergetics
 Department of Chemistry and Biochemistry University of Lethbridge II. Bioenergetics Slide 1 Bioenergetics Bioenergetics is the quantitative study of energy relationships and energy conversion in biological
Department of Chemistry and Biochemistry University of Lethbridge II. Bioenergetics Slide 1 Bioenergetics Bioenergetics is the quantitative study of energy relationships and energy conversion in biological
3. Organic Geochemisty Organic Chemistry is the chemistry... of Carbon -Morrison and Boyd
 3. Organic Geochemisty Organic Chemistry is the chemistry... of Carbon -Morrison and Boyd Definitions, Nomenclature Organic Compound Solubility Octanol-Water Partition Coefficient Organic Compound Sorption
3. Organic Geochemisty Organic Chemistry is the chemistry... of Carbon -Morrison and Boyd Definitions, Nomenclature Organic Compound Solubility Octanol-Water Partition Coefficient Organic Compound Sorption
C. Incorrect! Catalysts themselves are not altered or consumed during the reaction.
 Human Physiology - Problem Drill 04: Enzymes and Energy Question No. 1 of 10 Instructions: (1) Read the problem and answer choices carefully, (2) Work the problems on paper as needed, (3) Pick the answer,
Human Physiology - Problem Drill 04: Enzymes and Energy Question No. 1 of 10 Instructions: (1) Read the problem and answer choices carefully, (2) Work the problems on paper as needed, (3) Pick the answer,
Cell Energy: The Big Picture. So, What Exactly is ATP. Adenosine Triphosphate. Your turn to Practice converting ATP to ADP:
 Understanding How Living Things Obtain and Use Energy. Cell Energy: The Big Picture Most Autotrophs produce food (sugar) using light energy during Photosynthesis. Then, both Autotrophs and Heterotroph
Understanding How Living Things Obtain and Use Energy. Cell Energy: The Big Picture Most Autotrophs produce food (sugar) using light energy during Photosynthesis. Then, both Autotrophs and Heterotroph
Basic Concepts of Metabolism. Stages of Catabolism. Key intermediates 10/12/2015. Chapter 15, Stryer Short Course
 Basic Concepts of Metabolism Chapter 15, Stryer Short Course Digestion Formation of key intermediate small molecules Formation of ATP Stages of Catabolism Key intermediates 1 Fundamental Needs for Energy
Basic Concepts of Metabolism Chapter 15, Stryer Short Course Digestion Formation of key intermediate small molecules Formation of ATP Stages of Catabolism Key intermediates 1 Fundamental Needs for Energy
Energetics of metabolism
 Energetics of metabolism Dr. Bódis Emőke October 7, 2015 JJ9 Why do we study difficult thermodynamics? The laws and principles of the thermodynamics describe the characteristics of matter- and energy flow
Energetics of metabolism Dr. Bódis Emőke October 7, 2015 JJ9 Why do we study difficult thermodynamics? The laws and principles of the thermodynamics describe the characteristics of matter- and energy flow
of a comprehensive model (phosphofructokinase-pyruvate kinase reaction coupling/efficiency/atp/adp ratio)
 Proc. Nati. Acad. Sci. USA Vol. 78, No. 5, pp. 2952-2956, May 1981 Biophysics Oscillations and control features in glycolysis: Numerical analysis of a comprehensive model (phosphofructokinase-pyruvate
Proc. Nati. Acad. Sci. USA Vol. 78, No. 5, pp. 2952-2956, May 1981 Biophysics Oscillations and control features in glycolysis: Numerical analysis of a comprehensive model (phosphofructokinase-pyruvate
Enzymatic Assay of GALACTOSYLTRANSFERASE (EC )
 Enzymatic Assay of GALACTOSYLTRANSFERASE PRINCIPLE: UDP-Galactose + D-Glucose Galactosyltransferase > UDP + Lactose UDP + PEP PK > Pyruvate + UTP Pyruvate + ß-NADH LDH > Lactate + ß-NAD Abbreviations used:
Enzymatic Assay of GALACTOSYLTRANSFERASE PRINCIPLE: UDP-Galactose + D-Glucose Galactosyltransferase > UDP + Lactose UDP + PEP PK > Pyruvate + UTP Pyruvate + ß-NADH LDH > Lactate + ß-NAD Abbreviations used:
Something from nothing ) bridging the gap between constraint-based and kinetic modelling
 Something from nothing ) bridging the gap between constraint-based and kinetic modelling Kieran Smallbone,, Evangelos Simeonidis,3, David S. Broomhead, and Douglas B. Kell,4 Manchester Centre for Integrative
Something from nothing ) bridging the gap between constraint-based and kinetic modelling Kieran Smallbone,, Evangelos Simeonidis,3, David S. Broomhead, and Douglas B. Kell,4 Manchester Centre for Integrative
Bio102 Problems Photosynthesis
 Bio102 Problems Photosynthesis 1. Why is it advantageous for chloroplasts to have a very large (in surface area) thylakoid membrane contained within the inner membrane? A. This limits the amount of stroma
Bio102 Problems Photosynthesis 1. Why is it advantageous for chloroplasts to have a very large (in surface area) thylakoid membrane contained within the inner membrane? A. This limits the amount of stroma
G = (G C+ G D ) (G A + G B ) = ΣG P ΣG R Η = (H C+ H D ) (H A + H B ) = ΣΗ P ΣΗ S = (S C+ S D ) (S A + S B ) = ΣS P ΣS R BACKGROUND
 BACKGRUND BACKGRUND Α + Β = C + D G = Η Τ Τ S G = (G C+ G D ) (G A + G B ) = ΣG P ΣG R Η = (H C+ H D ) (H A + H B ) = ΣΗ P ΣΗ R S = (S C+ S D ) (S A + S B ) = ΣS P ΣS R 1 Standard versus Physiological
BACKGRUND BACKGRUND Α + Β = C + D G = Η Τ Τ S G = (G C+ G D ) (G A + G B ) = ΣG P ΣG R Η = (H C+ H D ) (H A + H B ) = ΣΗ P ΣΗ R S = (S C+ S D ) (S A + S B ) = ΣS P ΣS R 1 Standard versus Physiological
PARALLELS BETWEEN GLUCONEOGENESIS AND SYNCHRONOUS MACHINES
 PARALLELS BETWEEN GLUCONEOGENESIS AND SYNCHRONOUS MACHINES Monendra Grover Amity Institute of Biotechnology, Amity University, NOIDA Current Address : National Bureau of Plant Genetic Resources, Pusa Campus,
PARALLELS BETWEEN GLUCONEOGENESIS AND SYNCHRONOUS MACHINES Monendra Grover Amity Institute of Biotechnology, Amity University, NOIDA Current Address : National Bureau of Plant Genetic Resources, Pusa Campus,
Basic Concepts of Enzyme Action. Enzymes. Rate Enhancement 9/17/2015. Stryer Short Course Chapter 6
 Basic Concepts of Enzyme Action Stryer Short Course Chapter 6 Enzymes Biocatalysts Active site Substrate and product Catalyzed rate Uncatalyzed rate Rate Enhancement Which is a better catalyst, carbonic
Basic Concepts of Enzyme Action Stryer Short Course Chapter 6 Enzymes Biocatalysts Active site Substrate and product Catalyzed rate Uncatalyzed rate Rate Enhancement Which is a better catalyst, carbonic
Scientific paper. Ana Tu{ek Kurtanjek*
 52 Acta Chim. Slov. 2010, 57, 52 59 Scientific paper Lin-log Model of E. coli Central Metabolism Ana Tu{ek and @elimir Kurtanjek* University of Zagreb, Faculty of Food Technology and Biotechnology, Pierottijeva
52 Acta Chim. Slov. 2010, 57, 52 59 Scientific paper Lin-log Model of E. coli Central Metabolism Ana Tu{ek and @elimir Kurtanjek* University of Zagreb, Faculty of Food Technology and Biotechnology, Pierottijeva
Chapter 8: Energy and Metabolism
 Chapter 8: Energy and Metabolism Why do organisms need energy? How do organisms manage their energy needs? Defining terms and issues: energy and thermodynamics metabolic reactions and energy transfers
Chapter 8: Energy and Metabolism Why do organisms need energy? How do organisms manage their energy needs? Defining terms and issues: energy and thermodynamics metabolic reactions and energy transfers
Photosynthesis and Cellular Respiration Unit
 Photosynthesis and Cellular Respiration Unit All cellular activities require energy. Directly or indirectly nearly all energy for life comes from the sun. Autotrophs: organisms that can make their own
Photosynthesis and Cellular Respiration Unit All cellular activities require energy. Directly or indirectly nearly all energy for life comes from the sun. Autotrophs: organisms that can make their own
Cell and Molecular Biology
 Cell and Molecular Biology (3000719): academic year 2013 Content & Objective :Cell Chemistry and Biosynthesis 3 rd Edition, 1994, pp. 41-88. 4 th Edition, 2002, pp. 47-127. 5 th Edition, 2008, pp. 45-124.
Cell and Molecular Biology (3000719): academic year 2013 Content & Objective :Cell Chemistry and Biosynthesis 3 rd Edition, 1994, pp. 41-88. 4 th Edition, 2002, pp. 47-127. 5 th Edition, 2008, pp. 45-124.
13. PG3 + ATP + NADH => GA3P + ADP + Pi + NAD +
 Additional file 2. 1.1 Stochiometric model for P. pastoris containing some additional reactions from the 13 C model (section 1.2) Methanol metabolism 1. Metoh => Form 2. Form => FOR + NADH 3. FOR + NAD
Additional file 2. 1.1 Stochiometric model for P. pastoris containing some additional reactions from the 13 C model (section 1.2) Methanol metabolism 1. Metoh => Form 2. Form => FOR + NADH 3. FOR + NAD
Photosynthesis and Cellular Respiration
 Photosynthesis and Cellular Respiration Photosynthesis and Cellular Respiration All cellular activities require energy. Directly or indirectly nearly all energy for life comes from the sun. Autotrophs:
Photosynthesis and Cellular Respiration Photosynthesis and Cellular Respiration All cellular activities require energy. Directly or indirectly nearly all energy for life comes from the sun. Autotrophs:
Lecture 10. Proton Gradient-dependent ATP Synthesis. Oxidative. Photo-Phosphorylation
 Lecture 10 Proton Gradient-dependent ATP Synthesis Oxidative Phosphorylation Photo-Phosphorylation Model of the Electron Transport Chain (ETC) Glycerol-3-P Shuttle Outer Mitochondrial Membrane G3P DHAP
Lecture 10 Proton Gradient-dependent ATP Synthesis Oxidative Phosphorylation Photo-Phosphorylation Model of the Electron Transport Chain (ETC) Glycerol-3-P Shuttle Outer Mitochondrial Membrane G3P DHAP
CHEM-E3215 Advanced Biochemistry
 CHEM-E3215 Advanced Biochemistry 30. Jan. 2018 Prof. Silvan Scheller Lecture 10 Energy conservation general (some calculations) Energy conservation in anaerobes: e.g. methanogensis Life close to the thermodynamic
CHEM-E3215 Advanced Biochemistry 30. Jan. 2018 Prof. Silvan Scheller Lecture 10 Energy conservation general (some calculations) Energy conservation in anaerobes: e.g. methanogensis Life close to the thermodynamic
A hypothetical model of the influence of inorganic phosphate on the kinetics of pyruvate kinase
 BioSystems 54 (1999) 71 76 www.elsevier.com/locate/biosystems A hypothetical model of the influence of inorganic phosphate on the kinetics of pyruvate kinase Marian Kuczek * Institute of Biology and En
BioSystems 54 (1999) 71 76 www.elsevier.com/locate/biosystems A hypothetical model of the influence of inorganic phosphate on the kinetics of pyruvate kinase Marian Kuczek * Institute of Biology and En
QUALITATIVE PATH ANALYSIS OF METABOLIC PATHWAYS USING PETRI NETS FOR GENERIC MODELLING
 BRANDENBURG UNIVERSITY OF TECHNOLOGY AT COTTBUS Faculty of Mathematics, Natural Sciences and Computer Science Institute of Computer Science COMPUTER SCIENCE REPORTS Report 03/04 August 2004 QUALITATIVE
BRANDENBURG UNIVERSITY OF TECHNOLOGY AT COTTBUS Faculty of Mathematics, Natural Sciences and Computer Science Institute of Computer Science COMPUTER SCIENCE REPORTS Report 03/04 August 2004 QUALITATIVE
BCH Graduate Survey of Biochemistry
 BCH 5045 Graduate Survey of Biochemistry Instructor: Charles Guy Producer: Ron Thomas Director: Marsha Durosier Lecture 55 Slide sets available at: http://hort.ifas.ufl.edu/teach/guyweb/bch5045/index.html
BCH 5045 Graduate Survey of Biochemistry Instructor: Charles Guy Producer: Ron Thomas Director: Marsha Durosier Lecture 55 Slide sets available at: http://hort.ifas.ufl.edu/teach/guyweb/bch5045/index.html
Dr. Mallery Biology 150 Workshop Fall Semester ENERGY and METABOLISM ANSWERS
 Dr. Mallery Biology 150 Workshop Fall Semester ENERGY and METABOLISM ANSWERS IN THE GRAND SCHEME The purpose of this workshop is to get the Learning Communities to begin thinking about why certain chemical
Dr. Mallery Biology 150 Workshop Fall Semester ENERGY and METABOLISM ANSWERS IN THE GRAND SCHEME The purpose of this workshop is to get the Learning Communities to begin thinking about why certain chemical
Energy in Chemical and Biochemical Reactions
 Energy in Chemical and Biochemical Reactions Reaction Progress Diagram for Exothermic Reaction Reactants activated complex Products ENERGY A + B Reactants E a C + D Products Δ rxn Reaction coordinate The
Energy in Chemical and Biochemical Reactions Reaction Progress Diagram for Exothermic Reaction Reactants activated complex Products ENERGY A + B Reactants E a C + D Products Δ rxn Reaction coordinate The
Chapter 8 Metabolism: Energy, Enzymes, and Regulation
 Chapter 8 Metabolism: Energy, Enzymes, and Regulation Energy: Capacity to do work or cause a particular change. Thus, all physical and chemical processes are the result of the application or movement of
Chapter 8 Metabolism: Energy, Enzymes, and Regulation Energy: Capacity to do work or cause a particular change. Thus, all physical and chemical processes are the result of the application or movement of
RESPIRATION AND FERMENTATION: AEROBIC AND ANAEROBIC OXIDATION OF ORGANIC MOLECULES. Bio 107 Week 6
 RESPIRATION AND FERMENTATION: AEROBIC AND ANAEROBIC OXIDATION OF ORGANIC MOLECULES Bio 107 Week 6 Procedure 7.2 Label test tubes well, including group name 1) Add solutions listed to small test tubes 2)
RESPIRATION AND FERMENTATION: AEROBIC AND ANAEROBIC OXIDATION OF ORGANIC MOLECULES Bio 107 Week 6 Procedure 7.2 Label test tubes well, including group name 1) Add solutions listed to small test tubes 2)
AP Biology Exam Review 5: Enzymes & Metabolism (Photosynthesis & Respiration)
 Name: Date: AP Biology Exam Review 5: Enzymes & Metabolism (Photosynthesis & Respiration) Helpful Videos and Animations: 1. Bozeman Biology: Photosynthesis and Respiration 2. Bozeman Biology: Photosynthesis
Name: Date: AP Biology Exam Review 5: Enzymes & Metabolism (Photosynthesis & Respiration) Helpful Videos and Animations: 1. Bozeman Biology: Photosynthesis and Respiration 2. Bozeman Biology: Photosynthesis
Lecture 21 - Introduction to Metabolism: Bioenergetics
 Lecture 21 - Introduction to Metabolism: Bioenergetics Key Concepts Energy conversion in biological systems Metabolic redox reactions Review of thermodynamic principles and coupled reactions The adenylate
Lecture 21 - Introduction to Metabolism: Bioenergetics Key Concepts Energy conversion in biological systems Metabolic redox reactions Review of thermodynamic principles and coupled reactions The adenylate
Edexcel (B) Biology A-level
 Edexcel (B) Biology A-level Topic 5: Energy for Biological Processes Notes Aerobic Respiration Aerobic respiration as splitting of the respiratory substrate, to release carbon dioxide as a waste product
Edexcel (B) Biology A-level Topic 5: Energy for Biological Processes Notes Aerobic Respiration Aerobic respiration as splitting of the respiratory substrate, to release carbon dioxide as a waste product
Predicting rice (Oryza sativa) metabolism
 Predicting rice (Oryza sativa) metabolism Sudip Kundu Department of Biophysics, Molecular Biology & Bioinformatics, University of Calcutta, WB, India. skbmbg@caluniv.ac.in Collaborators: Mark G Poolman
Predicting rice (Oryza sativa) metabolism Sudip Kundu Department of Biophysics, Molecular Biology & Bioinformatics, University of Calcutta, WB, India. skbmbg@caluniv.ac.in Collaborators: Mark G Poolman
Transformation of Energy! Energy is the ability to do work.! Thermodynamics is the study of the flow and transformation of energy in the universe.
 Section 1 How Organisms Obtain Energy Transformation of Energy! Energy is the ability to do work.! Thermodynamics is the study of the flow and transformation of energy in the universe. Section 1 How Organisms
Section 1 How Organisms Obtain Energy Transformation of Energy! Energy is the ability to do work.! Thermodynamics is the study of the flow and transformation of energy in the universe. Section 1 How Organisms
Cellular Energy. How Organisms Obtain Energy Section 2: Photosynthesis Section 3: Cellular Respiration. Click on a lesson name to select.
 Section 1: How Organisms Obtain Energy Section 2: Photosynthesis Section 3: Cellular Respiration Click on a lesson name to select. Section 1 How Organisms Obtain Energy Transformation of Energy Energy
Section 1: How Organisms Obtain Energy Section 2: Photosynthesis Section 3: Cellular Respiration Click on a lesson name to select. Section 1 How Organisms Obtain Energy Transformation of Energy Energy
Chemistry 5.07SC Biological Chemistry I Fall Semester, 2013
 Chemistry 5.07SC Biological Chemistry I Fall Semester, 2013 Lecture 10. Biochemical Transformations II. Phosphoryl transfer and the kinetics and thermodynamics of energy currency in the cell: ATP and GTP.
Chemistry 5.07SC Biological Chemistry I Fall Semester, 2013 Lecture 10. Biochemical Transformations II. Phosphoryl transfer and the kinetics and thermodynamics of energy currency in the cell: ATP and GTP.
A NONLINEAR MODEL FOR GLYCOLYTIC OSCILLATIONS IN YEAST EXTRACTS
 A NONLINEAR MODEL FOR GLYCOLYTIC OSCILLATIONS IN YEAST EXTRACTS Item type text; Dissertation-Reproduction (electronic) Authors Singh, Ajeet, 1942- Publisher Rights The University of Arizona. Copyright
A NONLINEAR MODEL FOR GLYCOLYTIC OSCILLATIONS IN YEAST EXTRACTS Item type text; Dissertation-Reproduction (electronic) Authors Singh, Ajeet, 1942- Publisher Rights The University of Arizona. Copyright
Prediction of Enzyme Kinetic Parameters Based on Statistical Learning
 80 Genome Informatics 17(1): 80 87 (2006) Prediction of Enzyme Kinetic Parameters Based on Statistical Learning Simon Borger Wolfram Liebermeister Edda Klipp borger@molgen.mpg.de lieberme@molgen.mpg.de
80 Genome Informatics 17(1): 80 87 (2006) Prediction of Enzyme Kinetic Parameters Based on Statistical Learning Simon Borger Wolfram Liebermeister Edda Klipp borger@molgen.mpg.de lieberme@molgen.mpg.de
