Intro to the Course. CHEM-5181 Mass Spectrometry, Chromatography, and Computer Programming
|
|
|
- Jerome Goodman
- 6 years ago
- Views:
Transcription
1 Intro to the Course CHEM-5181 Mass Spectrometry, Chromatography, and Computer Programming Profs. Paul J. Ziemann & Jose L. Jimenez University of Colorado-Boulder Last update: Aug Introduction to CHEM-5181 Distribute Clickers as people come in If you borrow a clicker, you are responsible for returning a working clicker to me (@ end, or if you drop) or an Incomplete Grade will be assigned until you do Round of introductions Name, background, interests related to class Are you registered? Let me know about adds / drops Go through the Syllabus Boot camp for 1 st years: skills for research History of course, updates for this year Register clickers ( roll call ) 2 1
2 Precision vs. Accuracy I Y X Measure two variables E.g. concentration of Na + and Cl - in seawater Accepted value at origin 3 Clicker Test: Precision vs. Accuracy A B C D CQ: Which is the most precise? A B C D I don t know 4 2
3 Precision vs. Accuracy III A B C D CQ: Which is the most accurate? A B C D I don t know Follow up: Which is better, A or B? 5 Precision Precision vs. Accuracy IV Agreement between two or more measurements made in an identical fashion Accuracy Accuracy is the nearness of a measurement to the accepted value CQ: This measurement is A. Accurate B. Precise C. Precise and Accurate D. Neither E. I don t know 6 3
4 Action Items for you Get tetbooks (by today) Fill Doodle Poll for office hrs (by Thu) Piazza bulletin board: sign up (by today) ALL class announcements will go there ALL questions should go there unless they are of a personal nature (e.g. you are sick) Igor and Labview software Install IGOR in your laptops (by today) Go through IGOR tutorials (by Thu) Bring laptops to class on Thu and for all programming-related lectures Make sure you can connect to VGA display, and set resolution to Do reading and HW for Thu 7 Other Items Please erase board & turn on projector before class Silence your cell phone & laptop (and remind me to silence mine!) Suggestions during the course Questions? 8 4
5 Random errors are often distributed according to the normal error law Probability Density Normal Probability Density Function Absorbance (unitless) CQ: the probability that the absorbance is eactly 0.49 is: A B. 25 C. infinity D. zero E. I don t know CLT: 9 PDF vs CDF Probability Density vs. Cumulative Probability Probability Density Normal Probability Density Function Cumulative Probability Normal Cumulative Probability Function Absorbance (unitless) Absorbance (unitless) CQ: the probability that the absorbance is between 0.48 and 0.49 is: A. zero B. It is not defined C D E. I don t know 10 5
6 What is the strength of the electric field between two parallel plates at 1000 and 1000 V with respect to ground, if they are separated by 5 mm? A Tesla B. 400,000 Volts/meter C. Zero D. 400,000 Newtons/Coulomb E Coulombs 11 Clicker Q What is the strength of the electric field between two parallel plates at and V with respect to ground, if they are separated by 5 mm? A. -400,000 Newtons/Coulomb B. +400,000 Newtons/Coulomb C. +200,000 Newtons/Coulomb D. Zero E. I don t know 12 6
7 What is the force acting on a singly charged (+) potassium ion with zero velocity in a 1 Tesla magnetic field A. 1 Newton B Newtons C. Zero D. It depends on the relative alignment E. I don t know Clicker Q What is the net electric force on a neutral mercury atom in an electric field of 10 6 V/m? A. It depends on the polarizability of Hg B Newtons C Newtons / meter D. Zero E. I don t know 14 7
8 What is true for the distribution of molecular speeds of N 2 at ambient T & P A. The average speed is ~472 m/s B. The minimum speed is 0 m/s C. The maimum speed is not bound D. None of the above E. A, B, and C 15 Clicker Q The average thermal speed of SF 6 at ambient T & P is A. Smaller than that of N 2 B. Larger than that of N 2 C. Same as that of N 2 D. None of the above E. A, B, and C 16 8
9 Each air molecule in this classroom is colliding with others approimately: A. Once a second B. 100 times per second C. 10,000 times per second D times per second E times per second 17 Clicker Q The time between collisions for an individual air molecule in an ionizer at a pressure of 10-4 mbar is approimately: A. 0.1 ns B. 1 s C. 100 s D. 1 ms E. 3 s 18 9
10 What is the vapor pressure of water at 100 degrees C? A Torr B. 760 mbar C. 1 bar D. 1 atmosphere E. 15 Torr 19 Kinetic Theory of Gases
Lecture Interp-3: The Molecular Ion (McLafferty & Turecek 1993, Chapter 3)
 Lecture Interp-3: The Molecular Ion (McLafferty & Turecek 1993, Chapter 3) CU- Boulder CHEM-5181 Mass Spectrometry & Chromatography Prof. Jose-Luis Jimenez Last Updated: Oct. 2013 1 Business Items Delinquent
Lecture Interp-3: The Molecular Ion (McLafferty & Turecek 1993, Chapter 3) CU- Boulder CHEM-5181 Mass Spectrometry & Chromatography Prof. Jose-Luis Jimenez Last Updated: Oct. 2013 1 Business Items Delinquent
Data Analysis II. CU- Boulder CHEM-4181 Instrumental Analysis Laboratory. Prof. Jose-Luis Jimenez Spring 2007
 Data Analysis II CU- Boulder CHEM-48 Instrumental Analysis Laboratory Prof. Jose-Luis Jimenez Spring 007 Lecture will be posted on course web page based on lab manual, Skoog, web links Summary of Last
Data Analysis II CU- Boulder CHEM-48 Instrumental Analysis Laboratory Prof. Jose-Luis Jimenez Spring 007 Lecture will be posted on course web page based on lab manual, Skoog, web links Summary of Last
MS Interpretation-1: Introduction + Elemental Composition I
 MS Interpretation-1: Introduction + Elemental Composition I CU- Boulder CHEM 5181 Mass Spectrometry & Chromatography Prof. Jose-Luis Jimenez A few slides adapted from an earlier version of notes from Dr.
MS Interpretation-1: Introduction + Elemental Composition I CU- Boulder CHEM 5181 Mass Spectrometry & Chromatography Prof. Jose-Luis Jimenez A few slides adapted from an earlier version of notes from Dr.
Data Analysis I. CU- Boulder CHEM-4181 Instrumental Analysis Laboratory. Prof. Jose-Luis Jimenez Spring 2007
 Data Analysis I CU- Boulder CHEM-4181 Instrumental Analysis Laboratory Prof. Jose-Luis Jimenez Spring 2007 Presentation will be posted on course web page based on lab manual, Skoog, web links 5 Objective
Data Analysis I CU- Boulder CHEM-4181 Instrumental Analysis Laboratory Prof. Jose-Luis Jimenez Spring 2007 Presentation will be posted on course web page based on lab manual, Skoog, web links 5 Objective
PHYSICS 272 Electric & Magnetic Interactions
 PHYSICS 272 Electric & Magnetic Interactions Professor in Charge: Prof. Wei Xie WebAsssign Administrator: Dr. V.K. Saxena Office: PHYS 246 Office: PHYS 76 Email: wxie@purdue.edu E-Mail: saxena@purdue.edu
PHYSICS 272 Electric & Magnetic Interactions Professor in Charge: Prof. Wei Xie WebAsssign Administrator: Dr. V.K. Saxena Office: PHYS 246 Office: PHYS 76 Email: wxie@purdue.edu E-Mail: saxena@purdue.edu
Astronomy 1010: Survey of Astronomy. University of Toledo Department of Physics and Astronomy
 Astronomy 1010: Survey of Astronomy University of Toledo Department of Physics and Astronomy Information Kathy Shan Office: MH 4008 Phone: 530 2226 Email: kathy.shan@utoledo.edu Email is the best way to
Astronomy 1010: Survey of Astronomy University of Toledo Department of Physics and Astronomy Information Kathy Shan Office: MH 4008 Phone: 530 2226 Email: kathy.shan@utoledo.edu Email is the best way to
Energy. on this world and elsewhere. Instructor: Gordon D. Cates Office: Physics 106a, Phone: (434)
 Energy on this world and elsewhere Instructor: Gordon D. Cates Office: Physics 106a, Phone: (434) 924-4792 email: cates@virginia.edu Course web site available at www.phys.virginia.edu, click on classes
Energy on this world and elsewhere Instructor: Gordon D. Cates Office: Physics 106a, Phone: (434) 924-4792 email: cates@virginia.edu Course web site available at www.phys.virginia.edu, click on classes
Required Items. ACTIVE PARTICIPATION in class and lab. Use of iclicker
 Welcome to Chem103 Required Items Textbook: Chemistry: The Molecular Nature of Matter and Change, Martin S. Silberberg, 5 th Edition, McGraw-Hill, 2009 iclicker Scientific calculator ACTIVE PARTICIPATION
Welcome to Chem103 Required Items Textbook: Chemistry: The Molecular Nature of Matter and Change, Martin S. Silberberg, 5 th Edition, McGraw-Hill, 2009 iclicker Scientific calculator ACTIVE PARTICIPATION
CHEM 116 Phase Changes and Phase Diagrams
 CHEM 116 Phase Changes and Phase Diagrams Lecture 4 Prof. Sevian Please turn in extra credit assignments at the very beginning of class. Today s agenda Finish chapter 10 Partial pressures Vapor pressure
CHEM 116 Phase Changes and Phase Diagrams Lecture 4 Prof. Sevian Please turn in extra credit assignments at the very beginning of class. Today s agenda Finish chapter 10 Partial pressures Vapor pressure
Welcome to Physics 212
 Welcome to Physics 212 http://online.physics.uiuc.edu/courses/phys212 This lecture is VERY full. Please sit next to someone nice. Find out the best thing that happened to them during the winter break!
Welcome to Physics 212 http://online.physics.uiuc.edu/courses/phys212 This lecture is VERY full. Please sit next to someone nice. Find out the best thing that happened to them during the winter break!
Please turn cell phones off
 Welcome to Physics 102! Electricity Magnetism (at the heart of most processes around us: in atoms & molecules; living cells) Optics Atomic Physics Nuclear Physics Relativity http://www.communicationcurrents.com
Welcome to Physics 102! Electricity Magnetism (at the heart of most processes around us: in atoms & molecules; living cells) Optics Atomic Physics Nuclear Physics Relativity http://www.communicationcurrents.com
Lecture 2.1 :! Electric Field
 Lecture 2.1 :! Electric Field Lecture Outline:! Electric Field! Electric Field of Point Charges! Electric Field of Continuous Distribution of Charge! Textbook Reading:! Ch. 26.1-26.3 Jan. 20, 2015 1 Announcements
Lecture 2.1 :! Electric Field Lecture Outline:! Electric Field! Electric Field of Point Charges! Electric Field of Continuous Distribution of Charge! Textbook Reading:! Ch. 26.1-26.3 Jan. 20, 2015 1 Announcements
Gas Particle Partitioning to OA
 Gas Particle Partitioning to OA Advanced Atmospheric chemistry CHEM 5152 Prof. J.L. Jimenez Last updated: Spring 2017 1 Gas Phase vs Aerosol Compounds For monofunctional compounds, alkanes beyond C 20
Gas Particle Partitioning to OA Advanced Atmospheric chemistry CHEM 5152 Prof. J.L. Jimenez Last updated: Spring 2017 1 Gas Phase vs Aerosol Compounds For monofunctional compounds, alkanes beyond C 20
Physics 141 Course Information
 Physics 141 Course Information General Physics I - Mechanics Spring 2008 Instructors: Office Hours: Textbook: Online Homework: Disclaimer: Nikos Varelas 2134 SES (312) 996-3415 varelas@uic.edu Charles
Physics 141 Course Information General Physics I - Mechanics Spring 2008 Instructors: Office Hours: Textbook: Online Homework: Disclaimer: Nikos Varelas 2134 SES (312) 996-3415 varelas@uic.edu Charles
GG101 Dynamic Earth Fall 2015!
 GG101 Dynamic Earth Fall 2015! Instructor:! Dr. Bridget Smith-Konter!!!!! office: 819D POST!!! e-mail: brkonter@hawaii.edu! Syllabus Quick Guide! etext Physical Geology Fletcher, 2014 WileyPLUS Learning
GG101 Dynamic Earth Fall 2015! Instructor:! Dr. Bridget Smith-Konter!!!!! office: 819D POST!!! e-mail: brkonter@hawaii.edu! Syllabus Quick Guide! etext Physical Geology Fletcher, 2014 WileyPLUS Learning
Motion, Energy, and Gravity. Reminder to take out your clicker and turn it on!
 Motion, Energy, and Gravity Reminder to take out your clicker and turn it on! Attendance Quiz Are you here today? Here! (a) yes (b) no (c) Opening Day is here! x Clickers I have not been able to download
Motion, Energy, and Gravity Reminder to take out your clicker and turn it on! Attendance Quiz Are you here today? Here! (a) yes (b) no (c) Opening Day is here! x Clickers I have not been able to download
Statics - TAM 210 & TAM 211. Spring 2018
 Statics - TAM 210 & TAM 211 Spring 2018 Course distribution Required TAM 210 TAM 211 Aerospace Engineering 31 1 Agricultural & Biological 12 3 Bioengineering 2 6 Civil Engineering 41 Engineering Mechanics
Statics - TAM 210 & TAM 211 Spring 2018 Course distribution Required TAM 210 TAM 211 Aerospace Engineering 31 1 Agricultural & Biological 12 3 Bioengineering 2 6 Civil Engineering 41 Engineering Mechanics
Physics 141 Course Information
 Physics 141 Course Information General Physics I - Mechanics Spring 2009 Instructors: Office Hours: Textbook: Online Homework: Disclaimer: Nikos Varelas 2134 SES (312) 996-3415 varelas@uic.edu Adrian Barkan
Physics 141 Course Information General Physics I - Mechanics Spring 2009 Instructors: Office Hours: Textbook: Online Homework: Disclaimer: Nikos Varelas 2134 SES (312) 996-3415 varelas@uic.edu Adrian Barkan
PHYS 1020 General Physics. This image is from the wikimedia commons. File:Luna_Park_Melbourne_scenic_railway.
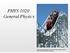 PHYS 1020 General Physics This image is from the wikimedia commons. http://commons.wikimedia.org/wiki/ File:Luna_Park_Melbourne_scenic_railway.jpg (Erich)Werner Ens 518 Allen Office Hours: MW 9:30-10:30
PHYS 1020 General Physics This image is from the wikimedia commons. http://commons.wikimedia.org/wiki/ File:Luna_Park_Melbourne_scenic_railway.jpg (Erich)Werner Ens 518 Allen Office Hours: MW 9:30-10:30
GEO 448 Plate Tectonics Fall 2014 Syllabus
 GEO 448 Plate Tectonics Fall 2014 Syllabus TH 4:00-8:10pm, Gillet Hall 324 Plate Tectonics as a unifying theory: the driving mechanisms of crustal deformation. Evidence supporting sea-floor spreading and
GEO 448 Plate Tectonics Fall 2014 Syllabus TH 4:00-8:10pm, Gillet Hall 324 Plate Tectonics as a unifying theory: the driving mechanisms of crustal deformation. Evidence supporting sea-floor spreading and
Math 103 Exam 3 Sample Exam. Name: Section: Date:
 Math 0 Eam Sample Eam Name: Section: Date: Signature: Do not start this test until your instructor says so. You will have eactly 0 minutes from this time to finish the test. Please show all work in the
Math 0 Eam Sample Eam Name: Section: Date: Signature: Do not start this test until your instructor says so. You will have eactly 0 minutes from this time to finish the test. Please show all work in the
PHYSICS 272 Electric & Magnetic Interactions
 PHYS 272: Matter and Interactions II Electric And Magnetic Interactions PHYSICS 272 Electric & Magnetic Interactions Lecture 5 Charging/Discharging; Efield of distributed charges: charged rod and ring
PHYS 272: Matter and Interactions II Electric And Magnetic Interactions PHYSICS 272 Electric & Magnetic Interactions Lecture 5 Charging/Discharging; Efield of distributed charges: charged rod and ring
CHEMISTRY 101 DETAILED WEEKLY TEXTBOOK HOMEWORK & READING SCHEDULE *
 CHEMISTRY 101 COURSE POLICIES 15 CHEMISTRY 101 DETAILED WEEKLY TEXTBOOK HOMEWORK & READING SCHEDULE * * Refer to textbook homework assignment and pre-lecture assignment for corresponding chapters to read.
CHEMISTRY 101 COURSE POLICIES 15 CHEMISTRY 101 DETAILED WEEKLY TEXTBOOK HOMEWORK & READING SCHEDULE * * Refer to textbook homework assignment and pre-lecture assignment for corresponding chapters to read.
Welcome. to Physics 2135.
 Welcome to Physics 2135. PHYSICS 2135 Engineering Physics II Dr. S. Thomas Vojta Instructor in charge Office: 204 Physics, Phone: 341-4793 vojtat@mst.edu www.mst.edu/~vojtat Office hours: Mon+ Wed 11am-12pm
Welcome to Physics 2135. PHYSICS 2135 Engineering Physics II Dr. S. Thomas Vojta Instructor in charge Office: 204 Physics, Phone: 341-4793 vojtat@mst.edu www.mst.edu/~vojtat Office hours: Mon+ Wed 11am-12pm
Intermediate Physics PHYS102
 Intermediate Physics PHYS102 Dr Richard H. Cyburt Assistant Professor of Physics My office: 402c in the Science Building My phone: (304) 384-6006 My email: rcyburt@concord.edu My webpage: www.concord.edu/rcyburt
Intermediate Physics PHYS102 Dr Richard H. Cyburt Assistant Professor of Physics My office: 402c in the Science Building My phone: (304) 384-6006 My email: rcyburt@concord.edu My webpage: www.concord.edu/rcyburt
Welcome to PHY212!! General Physics II:! Electricity, Magnetism, and Light!
 Welcome to PHY212!! General Physics II:! Electricity, Magnetism, and Light! Prof. Mitch Soderberg! msoderbe@syr.edu Lecture 1.1! Jan. 13, 2015 1 Announcements Register for PHY222 (labs) if you haven t
Welcome to PHY212!! General Physics II:! Electricity, Magnetism, and Light! Prof. Mitch Soderberg! msoderbe@syr.edu Lecture 1.1! Jan. 13, 2015 1 Announcements Register for PHY222 (labs) if you haven t
Lecture 1.2 :! Electric Force and Electric Field
 Lecture 1.2 :! Electric Force and Electric Field Lecture Outline:! Charging Objects! Coulomb s Law! Electric Field! Textbook Reading:! Ch. 25.3-25.5 Jan. 15, 2015 1 Announcements Quiz in class next Thu.
Lecture 1.2 :! Electric Force and Electric Field Lecture Outline:! Charging Objects! Coulomb s Law! Electric Field! Textbook Reading:! Ch. 25.3-25.5 Jan. 15, 2015 1 Announcements Quiz in class next Thu.
Mechanisms of Ion Fragmentation (McLafferty Chapter 4) Business Items
 Mechanisms of Ion Fragmentation (McLafferty Chapter 4) CU- Boulder CHEM 5181 Mass Spectrometry & Chromatography Prof. Jose-Luis Jimenez 1 Business Items Last real lecture is today Material from today,
Mechanisms of Ion Fragmentation (McLafferty Chapter 4) CU- Boulder CHEM 5181 Mass Spectrometry & Chromatography Prof. Jose-Luis Jimenez 1 Business Items Last real lecture is today Material from today,
SOLID 1. Make sure your state of matter is set on solid. Write your observations below:
 Chemistry Ms. Ye Name Date Block Properties of Matter: Particle Movement Part 1: Follow the instructions below to complete the activity. Click on the link to open the simulation for this activity: http://phet.colorado.edu/sims/states-of-matter/states-of-matterbasics_en.jnlp***note:
Chemistry Ms. Ye Name Date Block Properties of Matter: Particle Movement Part 1: Follow the instructions below to complete the activity. Click on the link to open the simulation for this activity: http://phet.colorado.edu/sims/states-of-matter/states-of-matterbasics_en.jnlp***note:
Ionization Techniques Part IV
 Ionization Techniques Part IV CU- Boulder CHEM 5181 Mass Spectrometry & Chromatography Presented by Prof. Jose L. Jimenez High Vacuum MS Interpretation Lectures Sample Inlet Ion Source Mass Analyzer Detector
Ionization Techniques Part IV CU- Boulder CHEM 5181 Mass Spectrometry & Chromatography Presented by Prof. Jose L. Jimenez High Vacuum MS Interpretation Lectures Sample Inlet Ion Source Mass Analyzer Detector
Everything in the universe can be classified as either matter or energy. Kinetic Energy Theory: All particles of matter are in constant motion.
 Physical Science Everything in the universe can be classified as either matter or energy. Kinetic Energy Theory: All particles of matter are in constant motion. State of Matter Bose- Einstein Condensate
Physical Science Everything in the universe can be classified as either matter or energy. Kinetic Energy Theory: All particles of matter are in constant motion. State of Matter Bose- Einstein Condensate
Physics 133 Electricity & Magnetism Class #2. Today s Concepts: A) Coulomb s Law B) Superposi<on
 Electricity & Magnetism Class #2 Today s Concepts: A) Coulomb s Law B) Superposi
Electricity & Magnetism Class #2 Today s Concepts: A) Coulomb s Law B) Superposi
PHYS 241 Electricity, Magnetism & Optics Syllabus
 PHYS 241 Electricity, Magnetism & Optics Syllabus Questions about grades, grade checks, absences, course withdrawal, etc please see Prof. Laura Pyrak-Nolte 1 PHYS 241 Electricity & Optics Physics & Astronomy
PHYS 241 Electricity, Magnetism & Optics Syllabus Questions about grades, grade checks, absences, course withdrawal, etc please see Prof. Laura Pyrak-Nolte 1 PHYS 241 Electricity & Optics Physics & Astronomy
CHEM 116 Phase Changes and Phase Diagrams
 CHEM 116 Phase Changes and Phase Diagrams Today is the first day that clicker questions count toward your grade. Remember to bring your i>clicker to class with you. There will be a clicker question at
CHEM 116 Phase Changes and Phase Diagrams Today is the first day that clicker questions count toward your grade. Remember to bring your i>clicker to class with you. There will be a clicker question at
Physics 2BL: Experiments in Mechanics and Electricity Summer Session I, 2012
 Physics BL: Experiments in Mechanics and Electricity Summer Session I, 01 Instructor: E-mail: Office: Office Hours: Phone: Tera (Bell) Austrum tbell@physics.ucsd.edu 164 Mayer Hall Addition TuTh 6-7 pm
Physics BL: Experiments in Mechanics and Electricity Summer Session I, 01 Instructor: E-mail: Office: Office Hours: Phone: Tera (Bell) Austrum tbell@physics.ucsd.edu 164 Mayer Hall Addition TuTh 6-7 pm
PHYS 122 Introductory Physics II Dr. Eric C. Anderson UMBC Fall 2016 Syllabus. Getting ready
 PHYS 122 Introductory Physics II Dr. Eric C. Anderson UMBC Fall 2016 Syllabus Getting ready Prerequisites You must have completed Phys 121, and Math 152 (or be enrolled in it this semester.) Workload Experience
PHYS 122 Introductory Physics II Dr. Eric C. Anderson UMBC Fall 2016 Syllabus Getting ready Prerequisites You must have completed Phys 121, and Math 152 (or be enrolled in it this semester.) Workload Experience
Start with known facts and reasonable guesses. More on order of magnitude Astronomy How many piano tuners are there in Boulder County?
 ASTR 101 Introduction to Astronomy: Stars & Galaxies Prof. Rosalba Perna TA: Cameron Clarke http://amalfi.astrosunysb.edu/rosalba/ast101/ast101.html MAKE SURE TO GO OVER THE SYLLABUS (if you haven t already
ASTR 101 Introduction to Astronomy: Stars & Galaxies Prof. Rosalba Perna TA: Cameron Clarke http://amalfi.astrosunysb.edu/rosalba/ast101/ast101.html MAKE SURE TO GO OVER THE SYLLABUS (if you haven t already
Chapter 11. Preview. Lesson Starter Objectives Pressure and Force Dalton s Law of Partial Pressures
 Preview Lesson Starter Objectives Pressure and Force Dalton s Law of Partial Pressures Section 1 Gases and Pressure Lesson Starter Make a list of gases you already know about. Separate your list into elements,
Preview Lesson Starter Objectives Pressure and Force Dalton s Law of Partial Pressures Section 1 Gases and Pressure Lesson Starter Make a list of gases you already know about. Separate your list into elements,
Chem Hughbanks Exam 2, March 10, 2016
 Chem 107 - Hughbanks Exam 2, March 10, 2016 Name (Print) UIN # Section 502 Exam 2, On the last page of this exam, you ve been given a periodic table and some physical constants. You ll probably want to
Chem 107 - Hughbanks Exam 2, March 10, 2016 Name (Print) UIN # Section 502 Exam 2, On the last page of this exam, you ve been given a periodic table and some physical constants. You ll probably want to
Agenda & Announcements
 CHEM 115 Review for Exam 3 Lecture 24 Prof. Sevian 1 Agenda & Announcements Reminder to take post-test next week during first hour of any lab period (next slide) Assignment 13 is to study for Exam 3. Attending
CHEM 115 Review for Exam 3 Lecture 24 Prof. Sevian 1 Agenda & Announcements Reminder to take post-test next week during first hour of any lab period (next slide) Assignment 13 is to study for Exam 3. Attending
Elements of Physics II. Agenda for Today
 Physics 132: Lecture e 18 Elements of Physics II Agenda for Today Magnets and the Magnetic Field Magnetic fields caused by charged particles B-field from a current-carrying wire Magnetic fields and forces
Physics 132: Lecture e 18 Elements of Physics II Agenda for Today Magnets and the Magnetic Field Magnetic fields caused by charged particles B-field from a current-carrying wire Magnetic fields and forces
PHYS 241 Electricity & Optics
 PHYS 41 Electricity & Optics Physics & Astronomy Department Home page: http://www.physics.purdue.edu/ Course Home page: http://www.physics.purdue.edu/phys41/ CHIP Home page: http://chip.physics.purdue.edu/public/41/fall016/
PHYS 41 Electricity & Optics Physics & Astronomy Department Home page: http://www.physics.purdue.edu/ Course Home page: http://www.physics.purdue.edu/phys41/ CHIP Home page: http://chip.physics.purdue.edu/public/41/fall016/
Lecture 31: Solutions 5
 Lecture 31: Solutions 5 Read: BLB 13.5 13.6 HW: BLB 13:9,58,61,67,69,75 Sup13:12 18 Know: colligative properties vapor pressure lowering boiling point elevation freezing point depression osmotic pressure
Lecture 31: Solutions 5 Read: BLB 13.5 13.6 HW: BLB 13:9,58,61,67,69,75 Sup13:12 18 Know: colligative properties vapor pressure lowering boiling point elevation freezing point depression osmotic pressure
EXPERIMENT 2-6. e/m OF THE ELECTRON GENERAL DISCUSSION
 Columbia Physics: Lab -6 (ver. 10) 1 EXPERMENT -6 e/m OF THE ELECTRON GENERAL DSCUSSON The "discovery" of the electron by J. J. Thomson in 1897 refers to the experiment in which it was shown that "cathode
Columbia Physics: Lab -6 (ver. 10) 1 EXPERMENT -6 e/m OF THE ELECTRON GENERAL DSCUSSON The "discovery" of the electron by J. J. Thomson in 1897 refers to the experiment in which it was shown that "cathode
Mass Analyzers. Ion Trap, FTICR, Orbitrap. CU- Boulder CHEM 5181: Mass Spectrometry & Chromatography. Prof. Jose-Luis Jimenez
 Mass Analyzers Ion Trap, FTICR, Orbitrap CU- Boulder CHEM 5181: Mass Spectrometry & Chromatography Prof. Jose-Luis Jimenez Last Update: Oct. 014 Some slides from Dr. Joel Kimmel (007) MS Interpretation
Mass Analyzers Ion Trap, FTICR, Orbitrap CU- Boulder CHEM 5181: Mass Spectrometry & Chromatography Prof. Jose-Luis Jimenez Last Update: Oct. 014 Some slides from Dr. Joel Kimmel (007) MS Interpretation
Chemistry 200: Basic Chemistry and Applications Course Syllabus: Spring
 Chemistry 200: Basic Chemistry and Applications Course Syllabus: Spring 2017 2018 Course Instructors Faraj Hasanayn; Office of Faraj Hasanayn: Chem Bldg. Rm 522 Office Hours: TBA Email: fh19@aub.edu.lb.
Chemistry 200: Basic Chemistry and Applications Course Syllabus: Spring 2017 2018 Course Instructors Faraj Hasanayn; Office of Faraj Hasanayn: Chem Bldg. Rm 522 Office Hours: TBA Email: fh19@aub.edu.lb.
Chemistry 311: Topic 3 - Mass Spectrometry
 Mass Spectroscopy: A technique used to measure the mass-to-charge ratio of molecules and atoms. Often characteristic ions produced by an induced unimolecular dissociation of a molecule are measured. These
Mass Spectroscopy: A technique used to measure the mass-to-charge ratio of molecules and atoms. Often characteristic ions produced by an induced unimolecular dissociation of a molecule are measured. These
Syllabus. Physics 0847, How Things Work Section II Fall 2014
 Syllabus Physics 0847, How Things Work Section II Fall 2014 Class Schedule: Tuesday, Thursday 11:00 a.m.-12:20 p.m. Location: Barton Hall BA130. Instructor: Dr. Zameer Hasan (215) 638 7219 Phone: Office:
Syllabus Physics 0847, How Things Work Section II Fall 2014 Class Schedule: Tuesday, Thursday 11:00 a.m.-12:20 p.m. Location: Barton Hall BA130. Instructor: Dr. Zameer Hasan (215) 638 7219 Phone: Office:
Chemistry 110 General Chemistry, Course Lecture MWF 8:30 am 9:50 am Room NSM C221 Laboratory M or W 1:00 pm 3:50 pm Room NSM B340
 Course Syllabus Fall 2009 CSU Dominguez Hills Chemistry 110 General Chemistry, Course 40318 Lecture MWF 8:30 am 9:50 am Room NSM C221 Laboratory M or W 1:00 pm 3:50 pm Room NSM B340 Instructor: Dr. Kenneth
Course Syllabus Fall 2009 CSU Dominguez Hills Chemistry 110 General Chemistry, Course 40318 Lecture MWF 8:30 am 9:50 am Room NSM C221 Laboratory M or W 1:00 pm 3:50 pm Room NSM B340 Instructor: Dr. Kenneth
CHEM 30A: Introductory General Chemistry Fall 2017, Laney College. Welcome to Chem 30A!
 CHEM 30A: Introductory General Chemistry Fall 2017, Laney College Welcome to Chem 30A! Meeting Time/Location Lecture (Class Code 41591): Sa 10:00AM 12:50PM in A233 Lab (Class Code 41592): Sa 1:30PM 4:20PM
CHEM 30A: Introductory General Chemistry Fall 2017, Laney College Welcome to Chem 30A! Meeting Time/Location Lecture (Class Code 41591): Sa 10:00AM 12:50PM in A233 Lab (Class Code 41592): Sa 1:30PM 4:20PM
Atoms & Their Interactions
 Lecture 2 Atoms & Their Interactions Si: the heart of electronic materials Intel, 300mm Si wafer, 200 μm thick and 48-core CPU ( cloud computing on a chip ) Twin Creeks Technologies, San Jose, Si wafer,
Lecture 2 Atoms & Their Interactions Si: the heart of electronic materials Intel, 300mm Si wafer, 200 μm thick and 48-core CPU ( cloud computing on a chip ) Twin Creeks Technologies, San Jose, Si wafer,
Newton s Laws of Motion
 Newton s Laws of Motion Chapter 2: pages 37-53 Review questions 1, 5-10, 14, 17, 21-24, 30 Peer Led Team Learning ES105x PLTL CRN 21823 Tuesdays, 7:30 to 9 PM, NS 101 Jody Berg, senior ES major who had
Newton s Laws of Motion Chapter 2: pages 37-53 Review questions 1, 5-10, 14, 17, 21-24, 30 Peer Led Team Learning ES105x PLTL CRN 21823 Tuesdays, 7:30 to 9 PM, NS 101 Jody Berg, senior ES major who had
Unit 4: The Nature of Matter
 16 16 Table of Contents Unit 4: The Nature of Matter Chapter 16: Solids, Liquids, and Gases 16.1: Kinetic Theory 16.2: Properties and Fluids 16.3: Behavior of Gases 16.1 Kinetic Theory Kinetic Theory kinetic
16 16 Table of Contents Unit 4: The Nature of Matter Chapter 16: Solids, Liquids, and Gases 16.1: Kinetic Theory 16.2: Properties and Fluids 16.3: Behavior of Gases 16.1 Kinetic Theory Kinetic Theory kinetic
ASTR1010 Lecture 2 17 Jan 13. Reading Comprehension Question. Reading Comprehension Question. Your turns. Introduce yourself to 2 neighbors
 ASTR1010 Lecture 2 17 Jan 13 Today Why Astronomy? One more answer The Really Big Picture Announcements: Tuesday: please install SkyGazer and bring laptop! Clickers-for-credit starts Tuesday, 22 Jan JiTT
ASTR1010 Lecture 2 17 Jan 13 Today Why Astronomy? One more answer The Really Big Picture Announcements: Tuesday: please install SkyGazer and bring laptop! Clickers-for-credit starts Tuesday, 22 Jan JiTT
CHEMISTRY 101 DETAILED WEEKLY TEXTBOOK HOMEWORK & READING SCHEDULE*
 CHEMISTRY 101 COURSE POLICIES 15 CHEMISTRY 101 DETAILED WEEKLY TEXTBOOK HOMEWORK & READING SCHEDULE* *Refer to textbook homework assignment and pre-lecture assignment for corresponding chapters to read.
CHEMISTRY 101 COURSE POLICIES 15 CHEMISTRY 101 DETAILED WEEKLY TEXTBOOK HOMEWORK & READING SCHEDULE* *Refer to textbook homework assignment and pre-lecture assignment for corresponding chapters to read.
PHY 114 A General Physics II 11 AM-12:15 PM TR Olin 101. Plan for Lecture 1:
 PHY 4 A General Physics II AM-:5 PM TR Olin Plan for Lecture :. Welcome & overview. Class structure & announcements 3. Electrical charges and forces /8/0 PHY 4 A Spring 0 -- Lecture PHY 4 A General Physics
PHY 4 A General Physics II AM-:5 PM TR Olin Plan for Lecture :. Welcome & overview. Class structure & announcements 3. Electrical charges and forces /8/0 PHY 4 A Spring 0 -- Lecture PHY 4 A General Physics
Who SHOULD take this course? Course Goals. Beginning of Today s Class. Who am I. Course Goals (more general) 1/16/18
 ASTR 1040: Accel Intro Astron 2 Stars & Galaxies Two merging galaxies HST: NGC2207 / IC2163 Prof. Juri Toomre TAs: Peri Johnson, Ryan Horton Tues/Thur 11:00am, Duane G-130 Lecture 1 16 Jan 2018 Detailed
ASTR 1040: Accel Intro Astron 2 Stars & Galaxies Two merging galaxies HST: NGC2207 / IC2163 Prof. Juri Toomre TAs: Peri Johnson, Ryan Horton Tues/Thur 11:00am, Duane G-130 Lecture 1 16 Jan 2018 Detailed
ASTR1120L & 2030L Introduction to Astronomical Observations Fall 2018
 ASTR1120L & 2030L Introduction to Astronomical Observations Fall 2018 Professor: Loris Magnani Office: Physics 238 E-Mail: loris@physast.uga.edu Web Page: www.physast.uga.edu/~loris follow the link to
ASTR1120L & 2030L Introduction to Astronomical Observations Fall 2018 Professor: Loris Magnani Office: Physics 238 E-Mail: loris@physast.uga.edu Web Page: www.physast.uga.edu/~loris follow the link to
ORANGE COAST COLLEGE
 ORANGE COAST COLLEGE Chemistry 185: General Chemistry Spring 2017 16 weeks: 01/30 05/28/2016 Section: 35652 (Lecture) 5 Units Lecture: M/W 12:45 pm 2:10 pm Room: CHEM 207 Labs: 30475: M/W 7:50 am 11:00
ORANGE COAST COLLEGE Chemistry 185: General Chemistry Spring 2017 16 weeks: 01/30 05/28/2016 Section: 35652 (Lecture) 5 Units Lecture: M/W 12:45 pm 2:10 pm Room: CHEM 207 Labs: 30475: M/W 7:50 am 11:00
Lecture 2 [Chapter 21] Tuesday, Jan 17th
![Lecture 2 [Chapter 21] Tuesday, Jan 17th Lecture 2 [Chapter 21] Tuesday, Jan 17th](/thumbs/73/68267691.jpg) Lecture 2 [Chapter 21] Tuesday, Jan 17th Administrative Items Assignments this week: read Ch 21 and Ch 22 in the textbook complete Pre-Lecture Ch22 HW assignment complete Ch 21 HW assignment [Pre-Lecture
Lecture 2 [Chapter 21] Tuesday, Jan 17th Administrative Items Assignments this week: read Ch 21 and Ch 22 in the textbook complete Pre-Lecture Ch22 HW assignment complete Ch 21 HW assignment [Pre-Lecture
ASTR1010 Lecture 3 22 Jan 13. MA2 due Thursday re-read D2L>Content>MA on tutorial submission. Space News? Reminders.
 ASTR1010 Lecture 3 22 Jan 13 Today How the Sky Works Announcements: Need a SkyGazer CD? Come borrow one now, return Thurs If you brought a laptop, get SkyGazer running now Clickers-for-credit starts TODAY
ASTR1010 Lecture 3 22 Jan 13 Today How the Sky Works Announcements: Need a SkyGazer CD? Come borrow one now, return Thurs If you brought a laptop, get SkyGazer running now Clickers-for-credit starts TODAY
Shenandoah University. (PowerPoint) LESSON PLAN *
 Shenandoah University (PowerPoint) LESSON PLAN * NAME DATE 10/28/04 TIME REQUIRED 90 minutes SUBJECT Algebra I GRADE 6-9 OBJECTIVES AND PURPOSE (for each objective, show connection to SOL for your subject
Shenandoah University (PowerPoint) LESSON PLAN * NAME DATE 10/28/04 TIME REQUIRED 90 minutes SUBJECT Algebra I GRADE 6-9 OBJECTIVES AND PURPOSE (for each objective, show connection to SOL for your subject
Physics 1520, Fall 2011 Quiz 3, Form: A
 Physics 1520, Fall 2011 Quiz 3, Form: A Name: Date: Numeric answers must include units. Sketches must be labeled. All short-answer questions must include your reasoning, for full credit. A correct answer
Physics 1520, Fall 2011 Quiz 3, Form: A Name: Date: Numeric answers must include units. Sketches must be labeled. All short-answer questions must include your reasoning, for full credit. A correct answer
Chemistry 141 Samuel A. Abrash Chemical Reactions Lab Lecture 9/5/2011
 Chemistry 141 Samuel A. Abrash Chemical Reactions Lab Lecture 9/5/2011 Q: Before we start discussing this week s lab, can we talk about our lab notebooks? Sure. Q: What makes a lab notebook a good notebook?
Chemistry 141 Samuel A. Abrash Chemical Reactions Lab Lecture 9/5/2011 Q: Before we start discussing this week s lab, can we talk about our lab notebooks? Sure. Q: What makes a lab notebook a good notebook?
Test Wednesday, March 15 th 7pm, Bring your calculator and #2 pencil with a good eraser! 20 Multiple choice questions from:
 Test Wednesday, March 15 th 7pm, Bring your calculator and #2 pencil with a good eraser! 20 Multiple choice questions from: Chapter 4 Newton s Three Laws, Free Body Diagrams, Friction Chapter 5 (except
Test Wednesday, March 15 th 7pm, Bring your calculator and #2 pencil with a good eraser! 20 Multiple choice questions from: Chapter 4 Newton s Three Laws, Free Body Diagrams, Friction Chapter 5 (except
AP Chemistry. Free-Response Questions
 2018 AP Chemistry Free-Response Questions College Board, Advanced Placement Program, AP, AP Central, and the acorn logo are registered trademarks of the College Board. AP Central is the official online
2018 AP Chemistry Free-Response Questions College Board, Advanced Placement Program, AP, AP Central, and the acorn logo are registered trademarks of the College Board. AP Central is the official online
Pretest A. Mean=43% 2 year college physics profs (US) get 80% (26/32)
 Announcements: Lab schedule will be posted next week. The first labs will start the week after Ask FB questions in real time during lectures Assignment 1 is due January 18 Quiz 3 is due January 15 Tutorials
Announcements: Lab schedule will be posted next week. The first labs will start the week after Ask FB questions in real time during lectures Assignment 1 is due January 18 Quiz 3 is due January 15 Tutorials
(3 pts) 2. In which gas sample do the molecules have a lower average kinetic energy? (A) Gas A (B) Gas B (C) Neither
 Consider two samples of gas, A and B, as shown in the figure below. Both containers are at the same temperature and pressure. Gas A 1.0 L 0.32 g Gas B 1.0 L 0.48 g (3 pts) 1. Which gas sample contains
Consider two samples of gas, A and B, as shown in the figure below. Both containers are at the same temperature and pressure. Gas A 1.0 L 0.32 g Gas B 1.0 L 0.48 g (3 pts) 1. Which gas sample contains
LECTURE 15. Prof. Paul
 LECTURE 15 Prof. Paul Review Clicker Questions: Review Clicker Questions: (You can talk with others) Mark and David are loading identical cement blocks onto a truck. Mark lifts his block straight up from
LECTURE 15 Prof. Paul Review Clicker Questions: Review Clicker Questions: (You can talk with others) Mark and David are loading identical cement blocks onto a truck. Mark lifts his block straight up from
10/29/2018. Chapter 7. Atoms Light and Spectra. Reminders. Topics For Today s Class. Hydrogen Atom. The Atom. Phys1411 Introductory Astronomy
 Phys1411 Introductory Astronomy Instructor: Dr. Goderya Chapter 7 Atoms Light and Spectra Reminders Topics For Today s Class Project 1 due November 12 th after and during Lab. Extra-credit Homework online.
Phys1411 Introductory Astronomy Instructor: Dr. Goderya Chapter 7 Atoms Light and Spectra Reminders Topics For Today s Class Project 1 due November 12 th after and during Lab. Extra-credit Homework online.
Date: Hybrid Chemistry Regents Prep Ms. Hart/Mr. Kuhnau
 Name: Period: Date: Hybrid Chemistry Regents Prep Ms. Hart/Mr. Kuhnau UNIT 5: Kinetics and Equilibrium Lesson 1: Collision theory and potential energy diagrams By the end of today, you will have an answer
Name: Period: Date: Hybrid Chemistry Regents Prep Ms. Hart/Mr. Kuhnau UNIT 5: Kinetics and Equilibrium Lesson 1: Collision theory and potential energy diagrams By the end of today, you will have an answer
Gases CHAPTER. Section 10.1 Properties of Gases
 CHAPTER Gases 10 Section 10.1 Properties of Gases 2. The following are observed properties of gases: (a) Gases have a variable volume. (b) Gases expand infinitely. (c) Gases compress uniformly. (d) Gases
CHAPTER Gases 10 Section 10.1 Properties of Gases 2. The following are observed properties of gases: (a) Gases have a variable volume. (b) Gases expand infinitely. (c) Gases compress uniformly. (d) Gases
Physical Science (SCI101) Final Exam
 Department of Mathematics and General Sciences Final Exam Second Semester, Term 132 Date: Wednesday 28/5/2014 Name: ID number: Section number or time: Instructor s name: Important instructions: 1. Examination
Department of Mathematics and General Sciences Final Exam Second Semester, Term 132 Date: Wednesday 28/5/2014 Name: ID number: Section number or time: Instructor s name: Important instructions: 1. Examination
Week 1 (8/25 8/29) Date Chapter Reading Topics Assigned Practice Problems (Zumdahl)
 Lecture Schedule, Chapter Readings and Practice Problems Chem 102 F&G Fall 2014 (Discussion dates and corresponding worksheets are noted in italics and in parenthesis) Week 1 (8/25 8/29) M, 8/25 Ch. 1:
Lecture Schedule, Chapter Readings and Practice Problems Chem 102 F&G Fall 2014 (Discussion dates and corresponding worksheets are noted in italics and in parenthesis) Week 1 (8/25 8/29) M, 8/25 Ch. 1:
Lecture 13.1 :! Electromagnetic Induction Continued
 Lecture 13.1 :! Electromagnetic Induction Continued Lecture Outline:! Faraday s Law! Induced Fields! Applications! Textbook Reading:! Ch. 33.5-33.7 April 7, 2015 1 Announcements! Homework #10 due on Tuesday,
Lecture 13.1 :! Electromagnetic Induction Continued Lecture Outline:! Faraday s Law! Induced Fields! Applications! Textbook Reading:! Ch. 33.5-33.7 April 7, 2015 1 Announcements! Homework #10 due on Tuesday,
General Chemistry I Office: Chem
 Chemistry 1A Section 1238 Instructor: Dr. Peter A. Doucette General Chemistry I Office: Chem 122 Spring 2010 email: pdoucette@elcamino.edu Office Hours: M, Tu & W: 1:00 2:00, or by appointment Meeting
Chemistry 1A Section 1238 Instructor: Dr. Peter A. Doucette General Chemistry I Office: Chem 122 Spring 2010 email: pdoucette@elcamino.edu Office Hours: M, Tu & W: 1:00 2:00, or by appointment Meeting
Physics 100. Reminder: All lecture notes posted, after lecture, follow link at:
 Physics 100 Please pick up a clicker! Reminder: All lecture notes posted, after lecture, follow link at: http://www.hunter.cuny.edu/physics/courses/physics100/spring-2016 Note: Before the actual lecture,
Physics 100 Please pick up a clicker! Reminder: All lecture notes posted, after lecture, follow link at: http://www.hunter.cuny.edu/physics/courses/physics100/spring-2016 Note: Before the actual lecture,
PHYS 3313 Section 001 Lecture #14
 PHYS 3313 Section 001 Lecture #14 Monday, March 6, 2017 The Classic Atomic Model Bohr Radius Bohr s Hydrogen Model and Its Limitations Characteristic X-ray Spectra 1 Announcements Midterm Exam In class
PHYS 3313 Section 001 Lecture #14 Monday, March 6, 2017 The Classic Atomic Model Bohr Radius Bohr s Hydrogen Model and Its Limitations Characteristic X-ray Spectra 1 Announcements Midterm Exam In class
CHE 105 EXAMINATION II October 13, 2011
 CHE 105 EXAMINATION II October 13, 2011 University of Kentucky Department of Chemistry READ THESE DIRECTIONS CAREFULLY BEFORE STARTING THE EXAMINATION! It is extremely important that you fill in the answer
CHE 105 EXAMINATION II October 13, 2011 University of Kentucky Department of Chemistry READ THESE DIRECTIONS CAREFULLY BEFORE STARTING THE EXAMINATION! It is extremely important that you fill in the answer
SPRING 2014 BIOC 4224 Physical Chemistry for Biologists SYLLABUS INSTRUCTORS:
 SPRING 2014 BIOC 4224 Physical Chemistry for Biologists SYLLABUS INSTRUCTORS: From Jan 13, 2014 - March 3, 2014 Dr. Jose L. Soulages, Professor E-mail: jose.soulages@okstate.edu Phone: 744-6212; Office:
SPRING 2014 BIOC 4224 Physical Chemistry for Biologists SYLLABUS INSTRUCTORS: From Jan 13, 2014 - March 3, 2014 Dr. Jose L. Soulages, Professor E-mail: jose.soulages@okstate.edu Phone: 744-6212; Office:
This should serve a s a study guide as you go on to do the problems in Sapling and take the quizzes and exams.
 CHM 111 Chapter 9 Worksheet and Study Guide Purpose: This is a guide for your as you work through the chapter. The major topics are provided so that you can write notes on each topic and work the corresponding
CHM 111 Chapter 9 Worksheet and Study Guide Purpose: This is a guide for your as you work through the chapter. The major topics are provided so that you can write notes on each topic and work the corresponding
LECTURE 5 SUMMARY OF KEY IDEAS
 LECTURE 5 SUMMARY OF KEY IDEAS Etching is a processing step following lithography: it transfers a circuit image from the photoresist to materials form which devices are made or to hard masking or sacrificial
LECTURE 5 SUMMARY OF KEY IDEAS Etching is a processing step following lithography: it transfers a circuit image from the photoresist to materials form which devices are made or to hard masking or sacrificial
Physics 1302W.400 Lecture 2 Introductory Physics for Scientists and Engineering II
 Physics 1302W.400 Lecture 2 Introductory Physics for Scientists and Engineering II In today s lecture, we will start to cover Chapter 23, with the goal to develop the force field concept for the long-range
Physics 1302W.400 Lecture 2 Introductory Physics for Scientists and Engineering II In today s lecture, we will start to cover Chapter 23, with the goal to develop the force field concept for the long-range
Required Textbook. Grade Determined by
 Physics 273 Honors (Spring 2015) (4 Credit Hours) Fundamentals of Physics II Syllabus available on BlackBoard http://webcourses.niu.edu/ under Course information Name: Prof. Omar Chmaissem (sha-my-sim)
Physics 273 Honors (Spring 2015) (4 Credit Hours) Fundamentals of Physics II Syllabus available on BlackBoard http://webcourses.niu.edu/ under Course information Name: Prof. Omar Chmaissem (sha-my-sim)
Welcome!! Chemistry 328N Organic Chemistry for Chemical Engineers. Professor: Grant Willson
 Welcome!! - 50750 Organic Chemistry for Chemical Engineers Professor: Grant Willson Teaching Assistants: Ji yeon Kim, Jai Hyun Koh, Paul Meyer, Qingjun Zhu http://willson.cm.utexas.edu January 19,2016
Welcome!! - 50750 Organic Chemistry for Chemical Engineers Professor: Grant Willson Teaching Assistants: Ji yeon Kim, Jai Hyun Koh, Paul Meyer, Qingjun Zhu http://willson.cm.utexas.edu January 19,2016
Physics 273 (Fall 2013) (4 Credit Hours) Fundamentals of Physics II
 Physics 273 (Fall 2013) (4 Credit Hours) Fundamentals of Physics II Syllabus available on BlackBoard http://webcourses.niu.edu/ under Course information Name: Prof. Omar Chmaissem (sha- my- sim) Email:
Physics 273 (Fall 2013) (4 Credit Hours) Fundamentals of Physics II Syllabus available on BlackBoard http://webcourses.niu.edu/ under Course information Name: Prof. Omar Chmaissem (sha- my- sim) Email:
Chem Hughbanks Exam 2, March 7, 2014
 Chem 107 - Hughbanks Exam 2, March 7, 2014 Key UI # Section 504 Exam 2, Key for n the last page of this exam, you ve been given a periodic table and some physical constants. You ll probably want to tear
Chem 107 - Hughbanks Exam 2, March 7, 2014 Key UI # Section 504 Exam 2, Key for n the last page of this exam, you ve been given a periodic table and some physical constants. You ll probably want to tear
Welcome to Physics 101! Basic Concepts of Physics
 Welcome to Physics 101! Basic Concepts of Physics Based on the book by Paul G. Hewitt: Instructor: Neepa Maitra Please pick up one handout for today, and a clicker Course information (on your handout)
Welcome to Physics 101! Basic Concepts of Physics Based on the book by Paul G. Hewitt: Instructor: Neepa Maitra Please pick up one handout for today, and a clicker Course information (on your handout)
PHYS 241 Electricity & Optics
 PHYS 241 Electricity & Optics Physics Department Home page: http://www.physics.purdue.edu/ Course Home page: http://www.physics.purdue.edu/phys241/ CHIP Home page: http://chip.physics.purdue.edu/public/241/spring2014/
PHYS 241 Electricity & Optics Physics Department Home page: http://www.physics.purdue.edu/ Course Home page: http://www.physics.purdue.edu/phys241/ CHIP Home page: http://chip.physics.purdue.edu/public/241/spring2014/
PHYS 241D Electricity & Optics
 PHYS 241D Electricity & Optics Physics Department Home page: http://www.physics.purdue.edu/ Course Home page: http://www.physics.purdue.edu/phys241d/ CHIP Home page: http://chip.physics.purdue.edu/public/241d/fall2014/
PHYS 241D Electricity & Optics Physics Department Home page: http://www.physics.purdue.edu/ Course Home page: http://www.physics.purdue.edu/phys241d/ CHIP Home page: http://chip.physics.purdue.edu/public/241d/fall2014/
Physics 211 (Spring 2016) (4 Credit Hours) General Physics II. Syllabus available on BlackBoard under Course information
 Physics 211 (Spring 2016) (4 Credit Hours) General Physics II Syllabus available on BlackBoard http://webcourses.niu.edu/ under Course information Name: Prof. Omar Chmaissem (sha-my-sim) (You can call
Physics 211 (Spring 2016) (4 Credit Hours) General Physics II Syllabus available on BlackBoard http://webcourses.niu.edu/ under Course information Name: Prof. Omar Chmaissem (sha-my-sim) (You can call
Chemistry 451. Prerequisites: CHEM 013, MATH 141, PHYS 202 or PHYS 212. Prof. Mueller/Sykes Chemistry 451 Spring 2004 Lecture 1-1
 Chemistry 451 CHEM 451 PHYSICAL CHEMISTRY ( 3 credits) Introduction to chemical principles, including properties of matter and fundamentals of chemical thermodynamics. Prerequisites: CHEM 013, MATH 141,
Chemistry 451 CHEM 451 PHYSICAL CHEMISTRY ( 3 credits) Introduction to chemical principles, including properties of matter and fundamentals of chemical thermodynamics. Prerequisites: CHEM 013, MATH 141,
The Measurement of e/m
 MSCD/UCD Physics Laboratories Lab II e/m The Measurement of e/m PURPOSE The objectives of this experiment are to measure the ratio between the charge and the mass of electrons, and then to find the mass
MSCD/UCD Physics Laboratories Lab II e/m The Measurement of e/m PURPOSE The objectives of this experiment are to measure the ratio between the charge and the mass of electrons, and then to find the mass
Unit 3 - Part 2: Gas Laws. Objective - learn the main gas laws that all molecules follow.
 Unit 3 - Part 2: Gas Laws Objective - learn the main gas laws that all molecules follow. Pressure - Pressure = Force / Area Created by collisions of the gas molecules with each other and with surfaces.
Unit 3 - Part 2: Gas Laws Objective - learn the main gas laws that all molecules follow. Pressure - Pressure = Force / Area Created by collisions of the gas molecules with each other and with surfaces.
CHEM 115 Precipitation Reactions and Witi Writing Net Ionic Equations
 CHEM 115 Precipitation React and Witi Writing Net Ionic Equat Important Announcement Exam #1 is next Thursday (February 26) Please read the information about exams on the website Important: You will sit
CHEM 115 Precipitation React and Witi Writing Net Ionic Equat Important Announcement Exam #1 is next Thursday (February 26) Please read the information about exams on the website Important: You will sit
AP Physics Ch 6 Day 4
 Textbook Reference: Goal/Objectives: Sections 6.3 (Collisions) - Understand how momentum and kinetic energy relate to the 3 types of collisions: elastic, inelastic, and perfectly inelastic - Use projectile
Textbook Reference: Goal/Objectives: Sections 6.3 (Collisions) - Understand how momentum and kinetic energy relate to the 3 types of collisions: elastic, inelastic, and perfectly inelastic - Use projectile
ASTR1120L & 2030L Introduction to Astronomical Observations Spring 2019
 ASTR1120L & 2030L Introduction to Astronomical Observations Spring 2019 Professor: Teaching Assistant: Office: Loris Magnani Jayne Dailey Physics 238 (Loris Magnani) Physics 241C (Jayne Dailey) E-Mail:
ASTR1120L & 2030L Introduction to Astronomical Observations Spring 2019 Professor: Teaching Assistant: Office: Loris Magnani Jayne Dailey Physics 238 (Loris Magnani) Physics 241C (Jayne Dailey) E-Mail:
PHYSICS 221 Fall 2007 EXAM 2: November 14, :00pm 10:00pm
 PHYSICS 221 Fall 2007 EXAM 2: November 14, 2007 8:00pm 10:00pm Name (printed): Recitation Instructor: Section # INSTRUCTIONS: This exam contains 25 multiple-choice questions plus 2 extra credit questions,
PHYSICS 221 Fall 2007 EXAM 2: November 14, 2007 8:00pm 10:00pm Name (printed): Recitation Instructor: Section # INSTRUCTIONS: This exam contains 25 multiple-choice questions plus 2 extra credit questions,
Chem 130 Name Exam 2 October 11, Points Part I: Complete all of problems 1-9
 Chem 130 Name Exam October 11, 017 100 Points Please follow the instructions for each section of the exam. Show your work on all mathematical problems. Provide answers with the correct units and significant
Chem 130 Name Exam October 11, 017 100 Points Please follow the instructions for each section of the exam. Show your work on all mathematical problems. Provide answers with the correct units and significant
Fig Note the three different types of systems based on the type of boundary between system and surroundings.
 CHAPTER 1 LECTURE NOTES System, Surroundings, and States Fig. 1.4 Note the three different types of systems based on the type of boundary between system and surroundings. Intensive and Extensive Properties
CHAPTER 1 LECTURE NOTES System, Surroundings, and States Fig. 1.4 Note the three different types of systems based on the type of boundary between system and surroundings. Intensive and Extensive Properties
