Chem Hughbanks Exam 2, March 7, 2014
|
|
|
- Lynette McCoy
- 5 years ago
- Views:
Transcription
1 Chem Hughbanks Exam 2, March 7, 2014 Key UI # Section 504 Exam 2, Key for n the last page of this exam, you ve been given a periodic table and some physical constants. You ll probably want to tear that page off to use during the exam you don t need to turn it in with the rest of the exam. The exam contains 11 problems, with 4 numbered pages. You have 50 minutes to complete the exam. Please show ALL your work as clearly as possible this will help us award you partial credit if appropriate. Even correct answers without supporting work may not receive credit. You may use an approved calculator for the exam, one without extensive programmable capabilities or the ability to store alphanumeric information. Print your name above, provide your UI number, and sign the honor code statement below. n my honor as an Aggie, I will neither give nor receive unauthorized assistance on this exam. SIGATURE:
2 For questions 1 through 4, consider the following gas samples (assume all the gases behave ideally): A. 20 g of e (g) at 0 C in a 22.4 L container B. 300 torr of 2 (g) at 27 C in a L container C mol of Ar (g) at 27 C in a 5.0 L container For each question below, please indicate your choice and give a brief justification of your answer (i.e., an explanation of your reasoning). You shouldn t need detailed numerical calculations, although you may perform some if you feel that will help. In the event that more than one of the gases satisfies one of the statements, you should say so. (For example, if you think they all are at the same pressure, then for part (b) you would write All have the same pressure because... ) (1) (5 pts) Which gas sample contains the fewest molecules? (For grading) Scores 1 /5 2 /5 3 /5 4 /5 5 /7 6 /7 7 /7 8 /18 9 /10 10 /16 11 /15 Tot. /100 Ans. 1 c (2) (5 pts) Which gas sample is at the lowest pressure? Ans. 2 b (3) (5 pts) In which of the samples is the average kinetic energy of the molecules highest? K.E. of ideal gases depends only on temperature, b and c have equal temps. Ans. 3 b & c (4) (5 pts) In which of the samples is the average molecular speed slowest? Ans. 4 c 1
3 (5) (7 pts) Lithium metal has an electron binding energy of J. What is the longest wavelength of light (in nm) than can eject electrons from lithium? (Put your answer with the correct units in the space provided.) E = hc/λ λ = hc/e = ( J s)( m/s)/( J) = m Ans nm (6) (7 pts) Write out the full electronic configuration of a selenium atom (Se, no inert gas formalism). Ans. 6 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 4 (7) (7 pts) In a selenium atom, which electron is least effective in screening a 3p electron from the nucleus, (a) a 1s electron (b) a 3s electron (c) another 3p electron (d) a 4p electron (e) a d are all equally effective Ans. 7 d (8) (18 points) In (a) (c), BE SURE T READ EACH STATEMET VERY CAREFULLY! (a) (6 pts) From the following choices, circle the molecule with the strongest C bond(s) and put a box around the molecule with the longest C bond(s). H 3 CCH 3 F 2 HC()H C 2 C H 2 C (b) (6 pts) Put the atoms, molecules, or ions in order, starting with those with the fewest unpaired electrons to most unpaired electrons: a, i 2+, Zn 2+, 2, Fewest unpaired 2 = Zn 2+ < a < i 2+ < Most unpaired (c) (6 pts) Atoms of which one of the following elements has the highest ionization energy? Be B C F e 2
4 Questions 9-11 show all work (42 pts) (9) (10 points) 2 5 is an unstable gas that decomposes to form 2 and 2 gases (only). What would be the total pressure of gases present if a 10.0 L container at 27 C begins with atm of 2 5 and the gas completely decomposes? Assume the final temperature is also 27 C. Balanced equation: Since nothing else changes, the pressure change will be solely due to the change in the number of moles of gas. 2 moles of reactant gas yields 5 moles of product gases, therefore the pressure will increase by a factor of 5/2; the final pressure will be 2.5(0.800 atm) = 2.00 atm (10) (16 points) Excited mercury atoms emit light at many wavelengths, two of which are at and nm. Both of these transitions are to the same final state. (a) (5 pts) Give the energy difference between the initial excited state and the lower final state for the transition at nm. E = hc/λ = ( J s)( m/s)/( m) = J (b) (5 pts) Give the energy difference between the initial excited state and the lower final state for the transition at nm. E = hc/λ = ( J s)( m/s)/( m) = J (c) (6 pts) If a transition between the two higher energy excited states could be observed, what would be the frequency of the light? The energy difference between the two excited states is ( E) = ( J) ( J) = J Therefore, the frequency is ν = ( E)/h = ( J)/( J s) = s 1 3
5 (11) (15 points) Below are shown four ions and one neutral molecule containing nitrogen-oxygen bonds. The diagrams given show only the way the atoms are connected together, they are T complete Lewis diagrams. There are five distinct observed bond distances (in pm), in order of increasing length: 106.2, 112.8, 118.8, 122.2, pm for the bonds that are labeled below. (Some bonds have the same length as others, so there are more labeled bonds than there are bond distances given above.) Pay attention to the charges on the ions only 2 is a neutral molecule. a + [ ] c b b d >50% <50% Fill in the table below. Use Lewis diagrams, including any necessary resonance structures, to find the - bond orders and decide which observed bond distances belong to which molecules. If you can t assign an exact value to a bond order, give the best estimate you can by using inequalities (for example, if you think a bond order is greater than 2 but less than 2.5, write 2 < B.. < 2.5). e equivalent h g f i two equivalent Bond Bond order Bond distance* Bond distance* a pm pm b 1 < B.. < pm pm c pm pm d pm pm e 1½ pm pm f 1½ pm pm g 1⅓ pm pm h 1⅓ pm pm i 1⅓ pm pm *Bond distance choices are (in pm): 106.2, 112.8, 118.8, 122.2, Answers in either of the two bond distance columns will be given full credit. 4
6 0
Chem Hughbanks Exam 2, March 10, 2016
 Chem 107 - Hughbanks Exam 2, March 10, 2016 Name (Print) UIN # Section 502 Exam 2, On the last page of this exam, you ve been given a periodic table and some physical constants. You ll probably want to
Chem 107 - Hughbanks Exam 2, March 10, 2016 Name (Print) UIN # Section 502 Exam 2, On the last page of this exam, you ve been given a periodic table and some physical constants. You ll probably want to
Chem Hughbanks Final Exam, May 11, 2011
 Chem 107 - Hughbanks Final Exam, May 11, 2011 Name (Print) UIN # Section 503 Exam 3, Version # A On the last page of this exam, you ve been given a periodic table and some physical constants. You ll probably
Chem 107 - Hughbanks Final Exam, May 11, 2011 Name (Print) UIN # Section 503 Exam 3, Version # A On the last page of this exam, you ve been given a periodic table and some physical constants. You ll probably
Chem Hughbanks Final Exam, May 11, 2011
 Chem 107 - Hughbanks Final Exam, May 11, 2011 Name (Print) UIN # Section 503 Exam 3, Version # A On the last page of this exam, you ve been given a periodic table and some physical constants. You ll probably
Chem 107 - Hughbanks Final Exam, May 11, 2011 Name (Print) UIN # Section 503 Exam 3, Version # A On the last page of this exam, you ve been given a periodic table and some physical constants. You ll probably
CHEMISTRY 107 Section 501 Exam #2 Version A October 20, 2017 Dr. Larry Brown
 NAME: (print) UIN #: CHEMISTRY 107 Section 501 Exam #2 Version A October 20, 2017 Dr. Larry Brown This is a 50-minute exam, and contains 23 multiple-choice questions and 3 free response problems. Point
NAME: (print) UIN #: CHEMISTRY 107 Section 501 Exam #2 Version A October 20, 2017 Dr. Larry Brown This is a 50-minute exam, and contains 23 multiple-choice questions and 3 free response problems. Point
Chem Hughbanks Exam 3A, Solutions
 Chem 107 - Hughbanks Exam 3A, Solutions Name (Print) UIN # Section 503 Exam 3, Version # A On the last page of this exam, you ve been given a periodic table and some physical constants. You ll probably
Chem 107 - Hughbanks Exam 3A, Solutions Name (Print) UIN # Section 503 Exam 3, Version # A On the last page of this exam, you ve been given a periodic table and some physical constants. You ll probably
Chem Hughbanks Exam 3, April 19, 2012
 Chem 107 - Hughbanks Exam 3, April 19, 2012 Name (Print) UIN # Section 503 Exam 3, Version # A On the last page of this exam, you ve been given a periodic table and some physical constants. You ll probably
Chem 107 - Hughbanks Exam 3, April 19, 2012 Name (Print) UIN # Section 503 Exam 3, Version # A On the last page of this exam, you ve been given a periodic table and some physical constants. You ll probably
Chem Hughbanks Exam 1 - Spring 2013
 Chem 107 - Hughbanks Exam 1 - Spring 2013 UIN # Section 502 Exam 1, Version # A On the last page of this exam, you ve been given a periodic table and some physical constants. You ll probably want to tear
Chem 107 - Hughbanks Exam 1 - Spring 2013 UIN # Section 502 Exam 1, Version # A On the last page of this exam, you ve been given a periodic table and some physical constants. You ll probably want to tear
Chem Hughbanks Exam 3, April 21, 2015
 Chem 107 - ughbanks Exam 3, April 21, 2015 Name (Print) Solutions UIN # Section 502 Exam 3, Version A On the last page of this exam, you ve been given a periodic table and some physical constants. You
Chem 107 - ughbanks Exam 3, April 21, 2015 Name (Print) Solutions UIN # Section 502 Exam 3, Version A On the last page of this exam, you ve been given a periodic table and some physical constants. You
Chem Hughbanks Exam 1 - Spring 2012
 UIN # Section 502 Exam 1, Version # A Chem 107 - Hughbanks Exam 1 - Spring 2012 LAST YEAR (2012) WE DIDN T COVER GASES ON THE FIRST EXAM. THIS YEAR WE WILL. KEEP THIS IN MIND WHE USING THIS FOR STUDY PURPOSES.
UIN # Section 502 Exam 1, Version # A Chem 107 - Hughbanks Exam 1 - Spring 2012 LAST YEAR (2012) WE DIDN T COVER GASES ON THE FIRST EXAM. THIS YEAR WE WILL. KEEP THIS IN MIND WHE USING THIS FOR STUDY PURPOSES.
CHEMISTRY 107 Section 501 Final Exam Version A December 12, 2016 Dr. Larry Brown
 NAME: (print) UIN #: CHEMISTRY 107 Section 501 Final Exam Version A December 12, 2016 Dr. Larry Brown This is a 2-hour exam, and contains 11 problems. There should be 14 numbered pages, including this
NAME: (print) UIN #: CHEMISTRY 107 Section 501 Final Exam Version A December 12, 2016 Dr. Larry Brown This is a 2-hour exam, and contains 11 problems. There should be 14 numbered pages, including this
CHEMISTRY 107 Section 501 Exam #3 Version A November 16, 2016 Dr. Larry Brown
 NAME: (print) UIN #: CHEMISTRY 107 Section 501 Exam #3 Version A November 16, 2016 Dr. Larry Brown This is a 50-minute exam, and contains 7 problems. There should be 10 numbered pages, including this one.
NAME: (print) UIN #: CHEMISTRY 107 Section 501 Exam #3 Version A November 16, 2016 Dr. Larry Brown This is a 50-minute exam, and contains 7 problems. There should be 10 numbered pages, including this one.
CHEMISTRY 107 Section 501 Final Exam Version A December 11, 2017 Dr. Larry Brown
 NAME: (print) UIN #: CHEMISTRY 107 Section 501 Final Exam Version A December 11, 2017 Dr. Larry Brown This is a 2-hour exam and contains 21 multiple-choice questions and 5 free response problems. Point
NAME: (print) UIN #: CHEMISTRY 107 Section 501 Final Exam Version A December 11, 2017 Dr. Larry Brown This is a 2-hour exam and contains 21 multiple-choice questions and 5 free response problems. Point
(3 pts) 2. In which gas sample do the molecules have a lower average kinetic energy? (A) Gas A (B) Gas B (C) Neither
 Consider two samples of gas, A and B, as shown in the figure below. Both containers are at the same temperature and pressure. Gas A 1.0 L 0.32 g Gas B 1.0 L 0.48 g (3 pts) 1. Which gas sample contains
Consider two samples of gas, A and B, as shown in the figure below. Both containers are at the same temperature and pressure. Gas A 1.0 L 0.32 g Gas B 1.0 L 0.48 g (3 pts) 1. Which gas sample contains
CHEMISTRY 107 Section 501 Exam #3 Version A November 15, 2017 Dr. Larry Brown
 NAME: (print) UIN #: CHEMISTRY 107 Section 501 Exam #3 Version A November 15, 2017 Dr. Larry Brown This is a 50-minute exam and contains 22 multiple-choice questions and 3 free response problems. Point
NAME: (print) UIN #: CHEMISTRY 107 Section 501 Exam #3 Version A November 15, 2017 Dr. Larry Brown This is a 50-minute exam and contains 22 multiple-choice questions and 3 free response problems. Point
Partial Periodic Table
 Easily Legible Printed Name: CHEM 3451, Spring 2018 Professor Walba Second Hour Exam March 13, 2018 scores: 1) 2) 3) 4) 5) CU Honor Code Pledge: On my honor, as a University of Colorado at Boulder Student,
Easily Legible Printed Name: CHEM 3451, Spring 2018 Professor Walba Second Hour Exam March 13, 2018 scores: 1) 2) 3) 4) 5) CU Honor Code Pledge: On my honor, as a University of Colorado at Boulder Student,
CHEMISTRY 2A Exam II
 Name Student ID Number TA Name Lab Section Winter 016 Enderle CHEMISTRY A Exam II Instructions: CLOSED BOOK EXAM! No books, notes, or additional scrap paper are permitted. All information required is contained
Name Student ID Number TA Name Lab Section Winter 016 Enderle CHEMISTRY A Exam II Instructions: CLOSED BOOK EXAM! No books, notes, or additional scrap paper are permitted. All information required is contained
CHEM 107 (Spring-2005) Exam 3 (100 pts)
 CHEM 107 (Spring-2005) Exam 3 (100 pts) Name: ------------------------------------------------------------------------, Clid # ------------------------------ LAST NAME, First (Circle the alphabet segment
CHEM 107 (Spring-2005) Exam 3 (100 pts) Name: ------------------------------------------------------------------------, Clid # ------------------------------ LAST NAME, First (Circle the alphabet segment
Chem 6 Sample exam 1 (150 points total) NAME:
 Chem 6 Sample exam 1 (150 points total) @ This is a closed book exam to which the Honor Principle applies. @ The last page contains equations and physical constants; you can detach it for easy reference.
Chem 6 Sample exam 1 (150 points total) @ This is a closed book exam to which the Honor Principle applies. @ The last page contains equations and physical constants; you can detach it for easy reference.
Identification Sticker
 Chemistry 1A Midterm Exam 1 September 20, 2001 Page 1 of 10. (Closed Book, 90 minutes, 150 points) Version D Name: TA: SID: Section: Identification Sticker Test-taking strategy: PLEASE READ THIS FIRST!
Chemistry 1A Midterm Exam 1 September 20, 2001 Page 1 of 10. (Closed Book, 90 minutes, 150 points) Version D Name: TA: SID: Section: Identification Sticker Test-taking strategy: PLEASE READ THIS FIRST!
Success means having the courage, the determination, and the will to become the person you believe you were meant to be.
 CHEMISTRY 101 Hour Exam III December 1, 2016 Adams/ Huynh Name Signature Section Success means having the courage, the determination, and the will to become the person you believe you were meant to be.
CHEMISTRY 101 Hour Exam III December 1, 2016 Adams/ Huynh Name Signature Section Success means having the courage, the determination, and the will to become the person you believe you were meant to be.
Chemistry 6 10:00 Section Time limit = 3 hours Spring There are two sections to this exam. Please read the instructions carefully.
 Chemistry 6 10:00 Section Final Exam Time limit = 3 hours Spring 2005 Name There are two sections to this exam. Please read the instructions carefully. In the first section, there are 13 multiple choice
Chemistry 6 10:00 Section Final Exam Time limit = 3 hours Spring 2005 Name There are two sections to this exam. Please read the instructions carefully. In the first section, there are 13 multiple choice
CHEM- 457: Inorganic Chemistry
 CHEM- 457: Inorganic Chemistry Midterm I March 13 th, 2014 NAME This exam is comprised of six questions and is ten pages in length. Please be sure that you have a complete exam and place your name on each
CHEM- 457: Inorganic Chemistry Midterm I March 13 th, 2014 NAME This exam is comprised of six questions and is ten pages in length. Please be sure that you have a complete exam and place your name on each
CH301 Fall 2012 Name: KEY VandenBout/LaBrake UNIT 2 READINESS ASSESSMENT QUIZ (RAQ) THIS QUIZ WILL BE PACED WITH CLICKER QUESTIONS
 CH301 Fall 2012 Name: KEY VandenBout/LaBrake UNIT 2 READINESS ASSESSMENT QUIZ (RAQ) THIS QUIZ WILL BE PACED WITH CLICKER QUESTIONS 1. A laser pulse shines for 10 s delivering a total energy of 4 mj of
CH301 Fall 2012 Name: KEY VandenBout/LaBrake UNIT 2 READINESS ASSESSMENT QUIZ (RAQ) THIS QUIZ WILL BE PACED WITH CLICKER QUESTIONS 1. A laser pulse shines for 10 s delivering a total energy of 4 mj of
CHEMISTRY 101 SPRING 2005 FINAL FORM B SECTIONS DR. KEENEY-KENNICUTT PART 1
 NAME CHEMISTRY 101 SPRING 2005 FINAL FORM B SECTIONS 501-511 DR. KEENEY-KENNICUTT Directions: (1) Put your name and signature on PART 1 and PART 2 of the exam where indicated. (2) Sign the Aggie Code on
NAME CHEMISTRY 101 SPRING 2005 FINAL FORM B SECTIONS 501-511 DR. KEENEY-KENNICUTT Directions: (1) Put your name and signature on PART 1 and PART 2 of the exam where indicated. (2) Sign the Aggie Code on
Practice Exam A Answer Key for Fall 2014
 Practice Exam A Answer Key for Fall 2014 First Hour Exam 5.111. Write your name and your TA's name below. Do not open the exam until the start of the exam is announced. The exam is closed notes and closed
Practice Exam A Answer Key for Fall 2014 First Hour Exam 5.111. Write your name and your TA's name below. Do not open the exam until the start of the exam is announced. The exam is closed notes and closed
CHEMISTRY 101 Hour Exam III. Andino/McCarren Section
 CHEMISTRY 101 Hour Exam III December 7, 2017 Andino/McCarren Name Signature Section "The ultimate measure of a man is not where he stands in moments of comfort and convenience, but where he stands at times
CHEMISTRY 101 Hour Exam III December 7, 2017 Andino/McCarren Name Signature Section "The ultimate measure of a man is not where he stands in moments of comfort and convenience, but where he stands at times
CHEMISTRY 101 SPRING 2010 FINAL FORM B DR. KEENEY-KENNICUTT PART 1
 NAME (Please print ) CHEMISTRY 101 SPRING 2010 FINAL FORM B DR. KEENEY-KENNICUTT Directions: (1) Put your name on PART 1 and your name and signature on PART 2 of the exam where indicated. (2) Sign the
NAME (Please print ) CHEMISTRY 101 SPRING 2010 FINAL FORM B DR. KEENEY-KENNICUTT Directions: (1) Put your name on PART 1 and your name and signature on PART 2 of the exam where indicated. (2) Sign the
YOUR NAME (last name, first name)...answers... FALL 2018 MIDTERM. (Total number of pages = 10) (Total points = 110) (Total time = 110 minutes)
 CHEM 14A Instructor: Dr. Laurence Lavelle YOUR NAME (last name, first name)...answers... STUDENT ID#... FALL 2018 MIDTERM (Total number of pages = 10) (Total points = 110) (Total time = 110 minutes) **Carefully
CHEM 14A Instructor: Dr. Laurence Lavelle YOUR NAME (last name, first name)...answers... STUDENT ID#... FALL 2018 MIDTERM (Total number of pages = 10) (Total points = 110) (Total time = 110 minutes) **Carefully
Chem 6 Practice Exam 2
 These problems are from past Chem 6 exams. Each exam contained a page of universal constant values and common equations; yours will, too, along with a Periodic Table! The answers follow after all the questions.
These problems are from past Chem 6 exams. Each exam contained a page of universal constant values and common equations; yours will, too, along with a Periodic Table! The answers follow after all the questions.
Name Chemistry 10 Dr. Kline 11 May 2004 Row Seat Lab 8:00 10:30
 Test 3 Name Chemistry 10 Dr. Kline 11 May 2004 Row Seat Lab 8:00 10:30 This test consists of a combination of multiple choice and other questions. There should be a total of 24 questions on 6 pages plus
Test 3 Name Chemistry 10 Dr. Kline 11 May 2004 Row Seat Lab 8:00 10:30 This test consists of a combination of multiple choice and other questions. There should be a total of 24 questions on 6 pages plus
Placement Exam for Exemption from Chemistry 120 Fall 2006
 Placement Exam for Exemption from Chemistry 120 Fall 2006 Name: By submitting this exam you affirm that you have not consulted any person, book, website or any other source for assistance. You may use
Placement Exam for Exemption from Chemistry 120 Fall 2006 Name: By submitting this exam you affirm that you have not consulted any person, book, website or any other source for assistance. You may use
CHEMISTRY 102 FALL 2009 EXAM 2 FORM B SECTION 501 DR. KEENEY-KENNICUTT PART 1
 NAME CHEMISTRY 102 FALL 2009 EXAM 2 FORM B SECTION 501 DR. KEENEY-KENNICUTT Directions: (1) Put your name on PART 1 and your name and signature on PART 2 of the exam where indicated. (2) Sign the Aggie
NAME CHEMISTRY 102 FALL 2009 EXAM 2 FORM B SECTION 501 DR. KEENEY-KENNICUTT Directions: (1) Put your name on PART 1 and your name and signature on PART 2 of the exam where indicated. (2) Sign the Aggie
Please read and sign the Honor Code statement below:
 CHEM 3311 Exam #1 Name Dr. Minger June 7, 2010 Please read and sign the Honor Code statement below: I pledge that on my honor, as a University of Colorado at Boulder student, I have neither given nor received
CHEM 3311 Exam #1 Name Dr. Minger June 7, 2010 Please read and sign the Honor Code statement below: I pledge that on my honor, as a University of Colorado at Boulder student, I have neither given nor received
Section A-Gases 1A. Circle the correct answer (18 pts total) (2 points each)
 Section A-Gases 1A. Circle the correct answer (18 pts total) (2 points each) (b) If a sample of gas is warmed in a rigid container (i.e., with fixed volume), which is true? i) the density of the gas remains
Section A-Gases 1A. Circle the correct answer (18 pts total) (2 points each) (b) If a sample of gas is warmed in a rigid container (i.e., with fixed volume), which is true? i) the density of the gas remains
Name: 1: /33 Grade: /100 2: /33 3: /33 +1 free point. Midterm Exam I. CHEM 181: Introduction to Chemical Principles September 20, 2012 Answer Key
 ame: 1: /33 Grade: /100 2: /33 3: /33 +1 free point Directions: Do all three problems. Midterm Exam I EM 181: Introduction to hemical Principles eptember 20, 2012 Answer Key how all of your work neatly
ame: 1: /33 Grade: /100 2: /33 3: /33 +1 free point Directions: Do all three problems. Midterm Exam I EM 181: Introduction to hemical Principles eptember 20, 2012 Answer Key how all of your work neatly
KEY: FREE RESPONSE. Question Ammonium carbamate, NH 4CO 2NH 2, reversibly decomposes into CO 2 and NH 3 as shown by the balanced equation below.
 KEY: FREE RESPONSE Question 1 1. Ammonium carbamate, NH 4CO 2NH 2, reversibly decomposes into CO 2 and NH 3 as shown by the balanced equation below. NH 4CO 2NH 2(s) 2 NH 3(g) + CO 2(g) K p =? A student
KEY: FREE RESPONSE Question 1 1. Ammonium carbamate, NH 4CO 2NH 2, reversibly decomposes into CO 2 and NH 3 as shown by the balanced equation below. NH 4CO 2NH 2(s) 2 NH 3(g) + CO 2(g) K p =? A student
Chem 105 Final Exam. Here is the summary of the total 225 points plus 10 bonus points. Carefully read the questions. Good luck!
 May 3 rd, 2012 Name: CLID: Score: Chem 105 Final Exam There are 50 multiple choices that are worth 3 points each. There are 4 problems and 1 bonus problem. Try to answer the questions, which you know first,
May 3 rd, 2012 Name: CLID: Score: Chem 105 Final Exam There are 50 multiple choices that are worth 3 points each. There are 4 problems and 1 bonus problem. Try to answer the questions, which you know first,
Partial Periodic Table
 CEM 33-00, Fall 203 Professor Walba First our Exam September 24, 203 scores: ) 20 2) 20 3) 20 4) 20 5) 20 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder Student, I have neither
CEM 33-00, Fall 203 Professor Walba First our Exam September 24, 203 scores: ) 20 2) 20 3) 20 4) 20 5) 20 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder Student, I have neither
Chem. 1B Final Practice
 Chem. 1B Final Practice Name Student Number All work must be shown on the exam for partial credit. Points will be taken off for incorrect or no units and for the incorrect number of significant figures.
Chem. 1B Final Practice Name Student Number All work must be shown on the exam for partial credit. Points will be taken off for incorrect or no units and for the incorrect number of significant figures.
Name: CHEMISTRY 101 SPRING 2005 EXAM 3 FORM A SECTIONS DR. KEENEY-KENNICUTT
 Name: CHEMISTRY 101 SPRING 2005 EXAM 3 FORM A SECTIONS 501511 DR. KEENEYKENNICUTT Directions: (1) Put your name and signature on PART 2 of the exam where indicated. (2) Sign the Aggie Code on PART 2 of
Name: CHEMISTRY 101 SPRING 2005 EXAM 3 FORM A SECTIONS 501511 DR. KEENEYKENNICUTT Directions: (1) Put your name and signature on PART 2 of the exam where indicated. (2) Sign the Aggie Code on PART 2 of
Chem 115 Sample Examination #3
 _ Chem 115 Sample Examination #3 Student Number: This exam consists of seven (7) pages, including this cover page. Be sure your copy is complete before beginning your work. If this test packet is defective,
_ Chem 115 Sample Examination #3 Student Number: This exam consists of seven (7) pages, including this cover page. Be sure your copy is complete before beginning your work. If this test packet is defective,
Chemistry 11, Fall 2006 Exam II November 16, :30 PM 9:30 PM
 Name: Section (circle): 1 2 3 4 5 Chemistry 11, Fall 2006 Exam II November 16, 2006 7:30 PM 9:30 PM As always, full credit will not be given unless you have written down the reasoning or calculations you
Name: Section (circle): 1 2 3 4 5 Chemistry 11, Fall 2006 Exam II November 16, 2006 7:30 PM 9:30 PM As always, full credit will not be given unless you have written down the reasoning or calculations you
"Let me tell you the secret that has led me to my goal. My strength lies solely in my tenacity." -Louis Pasteur-
 CHEMISTRY 101 Hour Exam III December 4, 2014 Adams/Esbenshade Name Signature Section "Let me tell you the secret that has led me to my goal. My strength lies solely in my tenacity." -Louis Pasteur- This
CHEMISTRY 101 Hour Exam III December 4, 2014 Adams/Esbenshade Name Signature Section "Let me tell you the secret that has led me to my goal. My strength lies solely in my tenacity." -Louis Pasteur- This
Chemistry 1A Fall 2000
 Chemistry 1A Fall 2000 Midterm Exam II, version A October 17, 2000 (Closed book, 75 minutes, 145 points) Name: SID: Section Number: T.A. Name: Exam information, extra directions, and useful hints to maximize
Chemistry 1A Fall 2000 Midterm Exam II, version A October 17, 2000 (Closed book, 75 minutes, 145 points) Name: SID: Section Number: T.A. Name: Exam information, extra directions, and useful hints to maximize
CHEMISTRY 101 SPRING 2009 EXAM 2 FORM A SECTION 501 DR. KEENEY-KENNICUTT PART 1
 NAME CHEMISTRY 101 SPRING 2009 EXAM 2 FORM A SECTION 501 DR. KEENEY-KENNICUTT Directions: (1) Put your name on PART 1 and your name and signature on PART 2 of the exam where indicated. (2) Sign the Aggie
NAME CHEMISTRY 101 SPRING 2009 EXAM 2 FORM A SECTION 501 DR. KEENEY-KENNICUTT Directions: (1) Put your name on PART 1 and your name and signature on PART 2 of the exam where indicated. (2) Sign the Aggie
High School Name: Individual Exam 1 Solutions: Chemical Structure and Properties
 Name (Last, First): ID Number: High School Name: Washington University Chemistry Tournament April 2, 2016 Individual Exam 1 Solutions: Chemical Structure and Properties Please write your full name, ID
Name (Last, First): ID Number: High School Name: Washington University Chemistry Tournament April 2, 2016 Individual Exam 1 Solutions: Chemical Structure and Properties Please write your full name, ID
CHEMISTRY 101 SPRING 2007 FI NAL FORM B SECTIONS DR. KEENEY-KENNICUTT
 NAME (Please print ) AM or PM CHEMISTRY 101 SPRING 2007 FI NAL FORM B SECTIONS 501-513 DR. KEENEY-KENNICUTT Directions: (1) Put your name (neatly) and signature on both parts of the exam where indicated.
NAME (Please print ) AM or PM CHEMISTRY 101 SPRING 2007 FI NAL FORM B SECTIONS 501-513 DR. KEENEY-KENNICUTT Directions: (1) Put your name (neatly) and signature on both parts of the exam where indicated.
BCIT Fall Chem Exam #1
 BCIT Fall 2012 Chem 3615 Exam #1 Name: Attempt all questions in this exam. Read each question carefully and give a complete answer in the space provided. Part marks given for wrong answers with partially
BCIT Fall 2012 Chem 3615 Exam #1 Name: Attempt all questions in this exam. Read each question carefully and give a complete answer in the space provided. Part marks given for wrong answers with partially
Chem. 1C Midterm 1 Version B April 26, 2017
 First initial of last name Chem. C Midterm Version B April 6, 07 Name: Print Neatly. You will lose point if I cannot read your name or perm number. Perm Number: All work must be shown on the exam for partial
First initial of last name Chem. C Midterm Version B April 6, 07 Name: Print Neatly. You will lose point if I cannot read your name or perm number. Perm Number: All work must be shown on the exam for partial
Chem. 1C Midterm 1 Version A April 26, 2017
 First initial of last name Chem. C Midterm Version A April 6, 07 Name: Print Neatly. You will lose point if I cannot read your name or perm number. Perm Number: All work must be shown on the exam for partial
First initial of last name Chem. C Midterm Version A April 6, 07 Name: Print Neatly. You will lose point if I cannot read your name or perm number. Perm Number: All work must be shown on the exam for partial
CHEM 121a Exam 3 Fall 1998
 Name SSN CHEM 121a Exam 3 Fall 1998 This exam consists of 8 true-false questions (each worth 2 pts), 20 multiple choice questions (each worth 3 pts), and 3 short problems (each worth 8 pts). There are
Name SSN CHEM 121a Exam 3 Fall 1998 This exam consists of 8 true-false questions (each worth 2 pts), 20 multiple choice questions (each worth 3 pts), and 3 short problems (each worth 8 pts). There are
Partial Periodic Table
 Easily Legible Printed Name: CEM 3311 (300), Fall 2014 Professor Walba First our Exam September 23, 2014 scores: 1) 2) 3) 4) 5) CU onor Code Pledge: n my honor, as a University of Colorado at Boulder Student,
Easily Legible Printed Name: CEM 3311 (300), Fall 2014 Professor Walba First our Exam September 23, 2014 scores: 1) 2) 3) 4) 5) CU onor Code Pledge: n my honor, as a University of Colorado at Boulder Student,
move on if you get stuck on one part
 Chem 105 Exam 2 Name This exam is schedule for 75 minutes and I anticipate it to take the full time allotted. You are free to leave if you finish. In multiple part problems, points awarded will not be
Chem 105 Exam 2 Name This exam is schedule for 75 minutes and I anticipate it to take the full time allotted. You are free to leave if you finish. In multiple part problems, points awarded will not be
4 Diatomic molecules
 s manual for Burrows et.al. Chemistry 3 Third edition 4 Diatomic molecules Answers to worked examples WE 4.1 The Lewis model (on p. 174 in Chemistry 3 ) Use the Lewis model to describe the bonding in (a)
s manual for Burrows et.al. Chemistry 3 Third edition 4 Diatomic molecules Answers to worked examples WE 4.1 The Lewis model (on p. 174 in Chemistry 3 ) Use the Lewis model to describe the bonding in (a)
McCarren Section. 16 (12 pts.) 17 (18 pts.) Assume atmospheric pressure is 1.00 atm (unless explicitly told otherwise).
 Page 1 CHEMISTRY 101 Hour Exam I February 12, 2019 McCarren Name KEY Signature Section Positive thinking is more than just a tagline. It changes the way we behave. And I firmly believe that when I am positive,
Page 1 CHEMISTRY 101 Hour Exam I February 12, 2019 McCarren Name KEY Signature Section Positive thinking is more than just a tagline. It changes the way we behave. And I firmly believe that when I am positive,
Chemical standard state: 1 M solutes, pure liquids, 1 atm gases Biochemical standard state: ph 7, all species in the ionic form found at ph 7
 Chemistry 271, Section 22xx Your Name: Prof. Jason Kahn University of Maryland, College Park Your SID #: General Chemistry and Energetics Exam II (100 points total) Your Section #: November 4, 2009 You
Chemistry 271, Section 22xx Your Name: Prof. Jason Kahn University of Maryland, College Park Your SID #: General Chemistry and Energetics Exam II (100 points total) Your Section #: November 4, 2009 You
Midterm Exam I. CHEM 181: Introduction to Chemical Principles September 24, 2015 Key
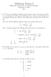 Midterm Exam I CHEM 8: Introduction to Chemical Principles September 24, 205 Key. A Li 2+ ion in an unknown, highly excited electronic state, first emits a photon at a wavelength of 4.36 µm, and following
Midterm Exam I CHEM 8: Introduction to Chemical Principles September 24, 205 Key. A Li 2+ ion in an unknown, highly excited electronic state, first emits a photon at a wavelength of 4.36 µm, and following
Lab Day and Time: Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Chem. 1A Final Practice Test 2
 Chem. 1A Final Practice Test 2 All work must be shown on the exam for partial credit. Points will be taken off for incorrect or missing units. Calculators are allowed. Cell phones may not be used as calculators.
Chem. 1A Final Practice Test 2 All work must be shown on the exam for partial credit. Points will be taken off for incorrect or missing units. Calculators are allowed. Cell phones may not be used as calculators.
CHEMISTRY 101 SPRING 2010 EXAM 3 FORM B SECTION 502 DR. KEENEY-KENNICUTT PART 1
 NAME CHEMISTRY 101 SPRING 2010 EXAM 3 FORM B SECTION 502 DR. KEENEY-KENNICUTT Directions: (1) Put your name and signature on PART 2 of the exam where indicated. (2) Sign the Aggie Code on PART 2 of this
NAME CHEMISTRY 101 SPRING 2010 EXAM 3 FORM B SECTION 502 DR. KEENEY-KENNICUTT Directions: (1) Put your name and signature on PART 2 of the exam where indicated. (2) Sign the Aggie Code on PART 2 of this
Chemistry General Chemistry II Spring 2006 Test #1
 Name: KEY Chemistry 122-04 -- General Chemistry II Spring 2006 Test #1 Organic molecules, molecular structure and bonding theory, solubility, (breathe now) phase transitions, spectroscopy, and kinetics
Name: KEY Chemistry 122-04 -- General Chemistry II Spring 2006 Test #1 Organic molecules, molecular structure and bonding theory, solubility, (breathe now) phase transitions, spectroscopy, and kinetics
CHEMISTRY 107 Section 501 Mini-Exam Version A September 7, 2018 Dr. Larry Brown
 NAME: (print) KEY UIN #: CHEMISTRY 107 Section 501 Mini-Exam Version A September 7, 2018 Dr. Larry Brown This is a 25-minute exam, and contains 12 multiple choice questions. Point values for the different
NAME: (print) KEY UIN #: CHEMISTRY 107 Section 501 Mini-Exam Version A September 7, 2018 Dr. Larry Brown This is a 25-minute exam, and contains 12 multiple choice questions. Point values for the different
CHEMISTRY 102 FALL 2010 EXAM 2 FORM C SECTION 502 DR. KEENEY-KENNICUTT PART 1
 NAME CHEMISTRY 102 FALL 2010 EXAM 2 FORM C SECTION 502 DR. KEENEY-KENNICUTT Directions: (1) Put your name on PART 1 and your name and signature on PART 2 of the exam where indicated. (2) Sign the Aggie
NAME CHEMISTRY 102 FALL 2010 EXAM 2 FORM C SECTION 502 DR. KEENEY-KENNICUTT Directions: (1) Put your name on PART 1 and your name and signature on PART 2 of the exam where indicated. (2) Sign the Aggie
KEY Chem 1A First Midterm Examination February 7, 2005 Professor David Chandler
 KEY Chem 1A First Midterm Examination February 7, 2005 Professor David Chandler 1 2 3 4 5 6 7 8 9 10 Extra Name: Signature: Section: GSI: Instructions As indicated, either fill in blank space with appropriate
KEY Chem 1A First Midterm Examination February 7, 2005 Professor David Chandler 1 2 3 4 5 6 7 8 9 10 Extra Name: Signature: Section: GSI: Instructions As indicated, either fill in blank space with appropriate
1. Which of the following is the correct Lewis Structure for the molecule sulfur tetriodide, SI 4? A.
 CHM 1045-All ections Exam 3 Practice Exam Fall 2013 Unit 3 Thermochemistry, Atomic tructure, & Bonding Chapters 6-10 ======================================================================= 1. Which of
CHM 1045-All ections Exam 3 Practice Exam Fall 2013 Unit 3 Thermochemistry, Atomic tructure, & Bonding Chapters 6-10 ======================================================================= 1. Which of
CHEM 341: Organic Chemistry I at North Dakota State University Midterm Exam 01 - Fri, Feb 10, 2012!! Name:!
 CEM 341: rganic Chemistry I at North Dakota State University Midterm Exam 01 - Fri, Feb 10, 2012!! Name:! Please read through each question carefully and answer in the spaces provided. A good strategy
CEM 341: rganic Chemistry I at North Dakota State University Midterm Exam 01 - Fri, Feb 10, 2012!! Name:! Please read through each question carefully and answer in the spaces provided. A good strategy
CHEMISTRY 101 EXAM 3 FORM 3M
 CHEMISTRY 101 EXAM 3 SECTIONS 572-580 Dr. Joy Heising Directions: FORM 3M November 20, 2001 1. This examination consists of two parts: 17 multiple choice questions (6 points each) in Part 1 and 4 free
CHEMISTRY 101 EXAM 3 SECTIONS 572-580 Dr. Joy Heising Directions: FORM 3M November 20, 2001 1. This examination consists of two parts: 17 multiple choice questions (6 points each) in Part 1 and 4 free
Lynch 2017 Page 1 of 5. Math 150, Fall 2017 Exam 1 Form A Multiple Choice
 Lynch 017 Page 1 of 5 Math 150, Fall 017 Exam 1 Form A Multiple Choice Last Name: First Name: Section Number: Student ID number: Directions: 1. No calculators, cell phones, or other electronic devices
Lynch 017 Page 1 of 5 Math 150, Fall 017 Exam 1 Form A Multiple Choice Last Name: First Name: Section Number: Student ID number: Directions: 1. No calculators, cell phones, or other electronic devices
Chem 35 - Exam II November 2, 1999 Prof. W.R. Leenstra. Please take a minute to read these instructions:
 Chem 35 - Exam II November 2, 1999 Prof. W.R. Leenstra NAME: Please take a minute to read these instructions: This may be common sense, but I require that all answers be justified, i.e., show all your
Chem 35 - Exam II November 2, 1999 Prof. W.R. Leenstra NAME: Please take a minute to read these instructions: This may be common sense, but I require that all answers be justified, i.e., show all your
Chem Midterm 3 April 23, 2009
 Chem. 101 - Midterm 3 April 3, 009 Name All work must be shown on the exam for partial credit. Points will be taken off for incorrect or no units and for the incorrect number of significant figures. Only
Chem. 101 - Midterm 3 April 3, 009 Name All work must be shown on the exam for partial credit. Points will be taken off for incorrect or no units and for the incorrect number of significant figures. Only
Name (printed): Signature:
 CHEM Lab Section Number: Name (printed): Signature: This exam consists of 36 questions all of equal value for a total of 225 points. Make sure that your test has all of the pages. Please read each problem
CHEM Lab Section Number: Name (printed): Signature: This exam consists of 36 questions all of equal value for a total of 225 points. Make sure that your test has all of the pages. Please read each problem
Name (printed): Signature:
 CHEM Lab Section Number: Name (printed): Signature: This exam consists of 36 questions all of equal value for a total of 225 points. Make sure that your test has all of the pages. Please read each problem
CHEM Lab Section Number: Name (printed): Signature: This exam consists of 36 questions all of equal value for a total of 225 points. Make sure that your test has all of the pages. Please read each problem
BROOKLYN COLLEGE Department of Chemistry. Chemistry 1 Second Lecture Exam Nov. 27, Name Page 1 of 5
 BROOKLYN COLLEGE Department of Chemistry Chemistry 1 Second Lecture Exam Nov. 27, 2002 Name Page 1 of 5 Circle the name of your lab instructor Kobrak, Zhou, Girotto, Hussey, Du Before you begin the exam,
BROOKLYN COLLEGE Department of Chemistry Chemistry 1 Second Lecture Exam Nov. 27, 2002 Name Page 1 of 5 Circle the name of your lab instructor Kobrak, Zhou, Girotto, Hussey, Du Before you begin the exam,
CHEMISTRY. Section II (Total time 95 minutes) Part A Time 55 minutes YOU MAY USE YOUR CALCULATOR FOR PART A.
 CHEMISTRY Section II (Total time 95 minutes) Part A Time 55 minutes YOU MAY USE YOUR CALCULATOR FOR PART A. CLEARLY SHOW THE METHOD USED AND THE STEPS INVOLVED IN ARRIVING AT YOUR ANSWERS. It is to your
CHEMISTRY Section II (Total time 95 minutes) Part A Time 55 minutes YOU MAY USE YOUR CALCULATOR FOR PART A. CLEARLY SHOW THE METHOD USED AND THE STEPS INVOLVED IN ARRIVING AT YOUR ANSWERS. It is to your
CHEMISTRY 101 Hour Exam III. Dr. D. DeCoste T.A (30 pts.) 16 (12 pts.) 17 (18 pts.) Total (60 pts)
 CHEMISTRY 101 Hour Exam III April 27, 2017 Dr. D. DeCoste Name Signature T.A. This exam contains 17 questions on 6 numbered pages. Check now to make sure you have a complete exam. You have one hour and
CHEMISTRY 101 Hour Exam III April 27, 2017 Dr. D. DeCoste Name Signature T.A. This exam contains 17 questions on 6 numbered pages. Check now to make sure you have a complete exam. You have one hour and
Success means having the courage, the determination, and the will to become the person you believe you were meant to be.
 CHEMISTRY 101 Hour Exam III December 7, 2006 Adams/Le Name KEY Signature T.A./Section Success means having the courage, the determination, and the will to become the person you believe you were meant to
CHEMISTRY 101 Hour Exam III December 7, 2006 Adams/Le Name KEY Signature T.A./Section Success means having the courage, the determination, and the will to become the person you believe you were meant to
Practice Exam A for Fall 2014 Exam 1
 Practice Exam A for Fall 2014 Exam 1 Page 1 of 8 pages First Hour Exam 5.111 Write your name and your TA's name below. Do not open the exam until the start of the exam is announced. The exam is closed
Practice Exam A for Fall 2014 Exam 1 Page 1 of 8 pages First Hour Exam 5.111 Write your name and your TA's name below. Do not open the exam until the start of the exam is announced. The exam is closed
Chemical standard state: 1 M solutes, pure liquids, 1 atm gases Biochemical standard state: ph 7, all species in the ionic form found at ph 7
 Chemistry 271, Section 22xx Your Name: Prof. Jason Kahn University of Maryland, College Park Your SID #: General Chemistry and Energetics Exam II (100 points total) Your Section #: November 4, 2009 You
Chemistry 271, Section 22xx Your Name: Prof. Jason Kahn University of Maryland, College Park Your SID #: General Chemistry and Energetics Exam II (100 points total) Your Section #: November 4, 2009 You
. This atom contains unpaired electrons. The
 Name (PRINT clearly) Seat Number Exam 2 (100 points) Chemistry 1303.002 October 25, 2017 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Write your answer in the
Name (PRINT clearly) Seat Number Exam 2 (100 points) Chemistry 1303.002 October 25, 2017 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Write your answer in the
CHEMISTRY 107 Section 501 Exam #1 Version A September 22, 2017 Dr. Larry Brown
 NAME: (print) UIN #: CHEMISTRY 107 Section 501 Exam #1 Version A September 22, 2017 Dr. Larry Brown This is a 50-minute exam, and contains 14 multiple choice questions and 4 free response problems. Point
NAME: (print) UIN #: CHEMISTRY 107 Section 501 Exam #1 Version A September 22, 2017 Dr. Larry Brown This is a 50-minute exam, and contains 14 multiple choice questions and 4 free response problems. Point
Partial Periodic Table
 Easily Legible Printed Name: CEM 3351 (100), Fall 2015 Professor Walba First our Exam September 22, 2015 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder Student, I have neither
Easily Legible Printed Name: CEM 3351 (100), Fall 2015 Professor Walba First our Exam September 22, 2015 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder Student, I have neither
1 Your full name (PLEASE PRINT) First hour test page 1 of 5 September 25, 2008 Your scheduled Tuesday quiz section (please circle) B hr E hr
 1 CHEM 111 Your full name (PLEASE PRINT) First hour test page 1 of 5 September 25, 2008 Your scheduled Tuesday quiz section (please circle) B hr E hr Your scheduled Tuesday quiz instructor: Make sure your
1 CHEM 111 Your full name (PLEASE PRINT) First hour test page 1 of 5 September 25, 2008 Your scheduled Tuesday quiz section (please circle) B hr E hr Your scheduled Tuesday quiz instructor: Make sure your
Do NOT rely on this as your only preparation for the Chem 101A final. You will almost certainly get a bad grade if this is all you look at.
 Do NOT rely on this as your only preparation for the Chem 101A final. You will almost certainly get a bad grade if this is all you look at. This is an INCOMPLETE list of hints and topics for the Chem 101
Do NOT rely on this as your only preparation for the Chem 101A final. You will almost certainly get a bad grade if this is all you look at. This is an INCOMPLETE list of hints and topics for the Chem 101
Chemistry 12 Dr. Kline 28 September 2005 Name
 Test 1 first letter of last name Chemistry 12 Dr. Kline 28 September 2005 Name This test consists of a combination of multiple choice and other questions. There should be a total of 24 questions on 8 pages;
Test 1 first letter of last name Chemistry 12 Dr. Kline 28 September 2005 Name This test consists of a combination of multiple choice and other questions. There should be a total of 24 questions on 8 pages;
CHEMISTRY 102 FALL 2010 EXAM 2 FORM B SECTION 501 DR. KEENEY-KENNICUTT PART 1
 NAME CHEMISTRY 102 FALL 2010 EXAM 2 FORM B SECTION 501 DR. KEENEY-KENNICUTT Directions: (1) Put your name on PART 1 and PART 2 of the exam where indicated. (2) Sign the Aggie Code on PART 2 of this exam.
NAME CHEMISTRY 102 FALL 2010 EXAM 2 FORM B SECTION 501 DR. KEENEY-KENNICUTT Directions: (1) Put your name on PART 1 and PART 2 of the exam where indicated. (2) Sign the Aggie Code on PART 2 of this exam.
DO NOT TURN THE PAGE UNTIL INSTRUCTED TO DO SO!
 The University of British Columbia Okanagan CHEM 121 ATOMIC & MOLECULAR CHEMISTRY Final Exam, Fall 2011 Wednesday, Dec 14, 9AM Name: Circle your Section: Student Number: McNeil / Neeland Signature: DO
The University of British Columbia Okanagan CHEM 121 ATOMIC & MOLECULAR CHEMISTRY Final Exam, Fall 2011 Wednesday, Dec 14, 9AM Name: Circle your Section: Student Number: McNeil / Neeland Signature: DO
CHEM 200/202 Exam 2 October 18, Lab Section #:
 Name: Lab Section #: Please mark your answers on the scantron sheet using a #2 pencil and also mark your answers on the exam itself. Mark Test From A on your scantron. 1. A 4.25 L container is filled with
Name: Lab Section #: Please mark your answers on the scantron sheet using a #2 pencil and also mark your answers on the exam itself. Mark Test From A on your scantron. 1. A 4.25 L container is filled with
CHEMISTRY 101 Hour Exam III. Dr. D. DeCoste T.A (30 pts.) 16 (12 pts.) 17 (18 pts.) Total (60 pts)
 CHEMISTRY 101 Hour Exam III April 27, 2017 Dr. D. DeCoste Name Signature T.A. This exam contains 17 questions on 6 numbered pages. Check now to make sure you have a complete exam. You have one hour and
CHEMISTRY 101 Hour Exam III April 27, 2017 Dr. D. DeCoste Name Signature T.A. This exam contains 17 questions on 6 numbered pages. Check now to make sure you have a complete exam. You have one hour and
1.) The answer to question 1 is A. Bubble in A on your Scantron form.
 Part 1: Multiple Choice. (5 pts each, 145 pts total) Instructions: Bubble in the correct answer on your Scantron form AND circle the answer on your exam. Each question has one correct answer. 1.) The answer
Part 1: Multiple Choice. (5 pts each, 145 pts total) Instructions: Bubble in the correct answer on your Scantron form AND circle the answer on your exam. Each question has one correct answer. 1.) The answer
Chemistry 110 Lecture Exam 4 Materials Chapter 11
 Chamras Chemistry 110 Lecture Exam 4 Materials Chapter 11 Modern Atomic Theory Rutherford s Atom: A theory developed to describe the structure of atoms, based on experimental evidence. The Experiment:
Chamras Chemistry 110 Lecture Exam 4 Materials Chapter 11 Modern Atomic Theory Rutherford s Atom: A theory developed to describe the structure of atoms, based on experimental evidence. The Experiment:
MATH 112 Final Exam, Spring Honor Statement
 NAME: QUIZ Section: STUDENT ID: MATH 112 Final Exam, Spring 2013 Honor Statement I affirm that my work upholds the highest standards of honesty and academic integrity at the University of Washington, and
NAME: QUIZ Section: STUDENT ID: MATH 112 Final Exam, Spring 2013 Honor Statement I affirm that my work upholds the highest standards of honesty and academic integrity at the University of Washington, and
1. My answers for this Chemistry 102 exam should be graded with the answer sheet associated with: a. Form A b. Form B c. Form C d. Form D e.
 EMISTRY 10 Fall 010 our Exam Page 1 1. My answers for this hemistry 10 exam should be graded with the answer sheet associated with: a. Form A b. Form B c. Form d. Form D e. Form E. Which of the statements
EMISTRY 10 Fall 010 our Exam Page 1 1. My answers for this hemistry 10 exam should be graded with the answer sheet associated with: a. Form A b. Form B c. Form d. Form D e. Form E. Which of the statements
CHEMISTRY 101 SPRING 2010 EXAM 1 FORM B SECTION 503 DR. KEENEY-KENNICUTT PART 1
 NAME CHEMISTRY 101 SPRING 2010 EXAM 1 FORM B SECTION 503 DR. KEENEY-KENNICUTT Directions: (1) Put your name and signature on PART 2 of the exam where indicated. (2) Sign the Aggie Code on PART 2 of this
NAME CHEMISTRY 101 SPRING 2010 EXAM 1 FORM B SECTION 503 DR. KEENEY-KENNICUTT Directions: (1) Put your name and signature on PART 2 of the exam where indicated. (2) Sign the Aggie Code on PART 2 of this
Page 1 of 22 Final Exam December 21, PAGE 2 1. TRANSITION METALS (35 points) PAGE 3 2. TRANSITION METALS (10 points)
 Page 1 of 22 Final Exam 5.111 December 21, 2011 Write your name and your TA's name below. Do not open the exam until the start of the exam is announced. 1) Read each part of a problem thoroughly. Many
Page 1 of 22 Final Exam 5.111 December 21, 2011 Write your name and your TA's name below. Do not open the exam until the start of the exam is announced. 1) Read each part of a problem thoroughly. Many
Chemistry 1A, Fall 2003 Midterm 1 Sept 16, 2003 (90 min, closed book)
 Name: SID: TA Name: Chemistry 1A, Fall 2003 Midterm 1 Sept 16, 2003 (90 min, closed book) This exam has 38 multiple choice questions. Fill in the Scantron form AND circle your answer on the exam. Each
Name: SID: TA Name: Chemistry 1A, Fall 2003 Midterm 1 Sept 16, 2003 (90 min, closed book) This exam has 38 multiple choice questions. Fill in the Scantron form AND circle your answer on the exam. Each
CHEM 237-Davis Organic Chemistry Examination 3 April 15, Instructions
 CHEM 237-Davis rganic Chemistry Examination 3 April 15, 2009 YUR NAME (Last, First) Initial of Last Name Instructions Fill in your name in the space above and on the next page Print the initial of your
CHEM 237-Davis rganic Chemistry Examination 3 April 15, 2009 YUR NAME (Last, First) Initial of Last Name Instructions Fill in your name in the space above and on the next page Print the initial of your
Chemistry 12 Name Exam II Form A Section
 hemistry 12 Name Exam II Form A Section July 21, 2005 Student No. IMPORTANT: On the scantron (answer sheet), you MUST clearly fill your name, your student number, section number, and test form (white cover
hemistry 12 Name Exam II Form A Section July 21, 2005 Student No. IMPORTANT: On the scantron (answer sheet), you MUST clearly fill your name, your student number, section number, and test form (white cover
CHEMISTRY 101 EXAM 3 FORM 3N
 CHEMISTRY 101 EXAM 3 SECTIONS 572-580 Dr. Joy Heising Directions: FORM 3N November 20, 2001 1. This examination consists of two parts: 17 multiple choice questions (6 points each) in Part 1 and 4 free
CHEMISTRY 101 EXAM 3 SECTIONS 572-580 Dr. Joy Heising Directions: FORM 3N November 20, 2001 1. This examination consists of two parts: 17 multiple choice questions (6 points each) in Part 1 and 4 free
On my honor as a Texas A&M University student, I will neither give nor receive unauthorized help on this exam.
 Physics 201, Exam 4 Name (printed) On my honor as a Texas A&M University student, I will neither give nor receive unauthorized help on this exam. Name (signed) The multiple-choice problems carry no partial
Physics 201, Exam 4 Name (printed) On my honor as a Texas A&M University student, I will neither give nor receive unauthorized help on this exam. Name (signed) The multiple-choice problems carry no partial
CHEM 101 Fall 08 Exam II(a)
 CHEM 101 Fall 08 Exam II(a) On the answer sheet (scantron) write you name, student ID number, and recitation section number. Choose the best (most correct) answer for each question and enter it on your
CHEM 101 Fall 08 Exam II(a) On the answer sheet (scantron) write you name, student ID number, and recitation section number. Choose the best (most correct) answer for each question and enter it on your
