II: Nomenclature. III: Charcteristics R SH. R S R' sulfide R SOH R S OH HO S O S OH. soft base easily oxidized. thiol. sulfenic acid.
|
|
|
- Annis Hill
- 5 years ago
- Views:
Transcription
1 ( ) I Introduction 3 ( 3 ) = 2 Allicin II omenclature 2 thiol ' sulfide oa ysteine Acetyl coenzyme A Adenosyl methionine Gliotoxin &!lactams Lenthionine Mitrasulgynine sulfenic acid sulfinic acid sulfonic acid ' ' sulfoxide sulfone III harcteristics ii. Low affinity to ; trong nucleophilicity pka pka orbital P As 1s 2s 3s e 2p 3p F l Br 3d Et basisity nucleophilicity Et highly polarizable soft base easily oxidized ' l 3 dx 2, dx 2 y 2 dxy, dyz, dxz "! Bulky Et / All 3 DM y. quant i. Less electronegativity compared with xygen Mitragynine
2 iii. Low bond energy ' (89 Kcal) (100 Kcal) v. Electronaccepting conjugative effect iv. Electronreleasing conjugative effect l l 3dxy 2p 2p 3p i. base Ph 3 Ph 2 ii. Br IV Application to the rganic yntheses base l (MTM) ' formation ( 2 ) n BF 3 Et 2, 2 l 2 TM 2 2 TM, ZnI 2, Et 2 ' ' cleavage ( 2 ) n gl 2 or X I or, etc cleavage ( 2 ) n E 2 ' 2 Base E ' ' '
3 PfitznerMoffatt DM, D, (pyridinef 3 ) 2. AlbrightGoldman DMAc wern DM, (F 3 ) 2, Et3, 2 l 2 DM, (l) 2, Et3, 2 l 2 l 4. ParikhDoering DM, 3 pyridine 3 ex.2 ex.1 1. Ph2 2. (Ph) 2 1. Ph Br Ph 1. nbuli 2. Li, 3 ' 2 Ph Li, 3 aney i Al(g), etc Ph oreykim DM, l 2 DM, l ex.3 Et Et 3 i Pd Application U7,8 2. Ar Ar heat (syn elimination) Ar 1. base Ar oxid. 2. ArAr heat 4. Pummere eaction Ac 2 ' ' ( 3 ) 2 ' Ac ' Ac ' 3. Ar Evans earrangement 2 dipoledipole repulsion (Allyl sulfoxide) 87% yield Ph 3 Ph X (F 3 ) 2 Ph Ph 3 Ph Ph Ph Ph Ar Ar () 3 P
4 Biosynthesis of Penicillins 3 Enzyme 2 2 Arnstein tripeptide 2 Fe Fe 2 Fe 2 5. Dimethylsulfonium methylide 2 2 Dimethyloxosulfonium methylide 2 2 isopenicillin Biomimetic ynthesis of Penicillin Deriv. i t 2 Bu () Ph Ph i 2 Bu t Ph ZnI Ph 2 cis (~111) 2 PPh 3 PPh 3 lefine 6. eebach, orey Umpolung X nbuli Li a b a ' b D1. hiral ulfoxide () () FAM 2 MTsulfone
5 D2. Preparation of chiral sulfoxide (Andersen ) MgX 2 lmenthyl ()()ptolylsulfinate 2 P () () 2 P 2 1 'AlX 2 ZnBr 2 'AlX 2 a b Zn ' AlX 2 ' AlX 2 a b ' (1) ' (2) (1) (2) tbu 2 ZnBr 2 tbu 2 (n 7 15 ) 3 Al n 7 15 tbu 2 D4. Asymmetric cycloaddition DielsAlder reaction (hiral Auxiliary) n 9 19 n 9 19 MgX ZnBr 2 MgX t 97% ee >98% ee Dienophile ( 2 )n Diene ( 2 )n eactivity tereoselectivity Enantioselectivity
6 2 Et 2 Et Diene Et 2 Et endo exo 2 Et Ln eactivity as dienophile t Ln 2Pyridyl group EWG strong inductive electronwithdrawing group 2 n 1. s 4 2. acetone PPT TB 3. LiAl( t Bu) 3 4. TBl 1. Py, cat. Et 3 2. mpba 3. recrystalization 2 n 2 Py 2 n () DBU 2 n 1. D, DM 2. 2 =PPh 3 3. aq. F 3 2 n Dhowdomycin Py 2 n 1. s 4 2. acetone PPT 1. cat. Et 3 2 n 2. mpba F 3 3. recrystalization 2 n Ar Ar F 3 1. Til 3 2. LiAl 4 aq. F n () 0 Ar (982) 1. ()Glyoxalase I Inhibitor Ar 2. mpba 2 n 2, py
7 Asymmetric eduction of Ketones with AD Model ompounds arrying a hiral ulfoxide Functin Proton Pump Inhibitor PPI) (=,K ATPase ) prazole benzimidazole Pr 2 P P Ph X= (AD) X=P 2 2 (ADP) X 2 Mg 2 (AD, ADP) Ph ' methyl ()()mandelate 97% ee Ph si F 2 2 F 3 AstraZeneca AstraZeneca (1988, EU 1991, JP) (2000.8, EU 2001, U, , JP) WyethAyerst ( 2 ) 3 (2000.2, U) (1991, EU , JP 1995,U) ( , U 1997, JP) ynthesis of meprazole PPI * Enzyme Enzyme ) 3, 3 3 Ac , ) 3 1) a, Et(=) K mpba, 2 l 2 p 8.6, 5, 10 min ) l 2 l a, 2 Et reflux, 2 h 3 3 3
8 PAB (Pottasiumcompetitive acid blockers) = omeprazole (racemate) = 2 l PL F 3 ( ) harpless Asymmetric xid. Ti(iPr) 4, (,)DET ipr 2 Et, Ph() 2 > 94 % ee. ottn et al.(astrazeneca), TetrahedronAsymmetry, 11, 3819 (2000) a/ibu(mibk) rystal. from MIBK esomeprazole a a salt > 99.5 % ee,k ATPase
Structure and Reactivity: Prerequired Knowledge
 Structure and eactivity: Prerequired Knowledge!!! The concepts presented in this summary are required for lecture and examination!!! 1. Important Principles in rganic Chemistry In general, structures which
Structure and eactivity: Prerequired Knowledge!!! The concepts presented in this summary are required for lecture and examination!!! 1. Important Principles in rganic Chemistry In general, structures which
Carbon-heteroatom single bonds basic C N C X. X= F, Cl, Br, I Alkyl Halide C O. epoxide Chapter 14 H. alcohols acidic H C S C. thiols.
 hapter 13: Alcohols and Phenols 13.1 Structure and Properties of Alcohols Alkanes arbon - arbon Multiple Bonds arbon-heteroatom single bonds basic Alkenes X X= F, l,, I Alkyl alide amines hapter 23 nitro
hapter 13: Alcohols and Phenols 13.1 Structure and Properties of Alcohols Alkanes arbon - arbon Multiple Bonds arbon-heteroatom single bonds basic Alkenes X X= F, l,, I Alkyl alide amines hapter 23 nitro
Chiral Catalyst II. Palladium Catalysed Allylic Displacement ( -allyl complexes) 1. L n Pd(0) 2. Nuc
 Chiral Catalyst II ast lecture we looked at asymmetric catalysis for oxidation and reduction Many other organic transformations, this has led to much investigation Today we will look at some others...
Chiral Catalyst II ast lecture we looked at asymmetric catalysis for oxidation and reduction Many other organic transformations, this has led to much investigation Today we will look at some others...
Chiral Auxiliaries. attach auxiliary Substrate Substrate Auxiliary
 Chiral Auxiliaries Previously on Advanced ynthesis... Discussed the need for stereoselective synthesis Looked at the use of resolution, the chiral pool and substrate control t there are some potential
Chiral Auxiliaries Previously on Advanced ynthesis... Discussed the need for stereoselective synthesis Looked at the use of resolution, the chiral pool and substrate control t there are some potential
O + k 2. H(D) Ar. MeO H(D) rate-determining. step?
 ame: CEM 633: Advanced rganic Chem: ysical Problem Set 6 (Due Thurs, 12/8/16) Please do not look up references until after you turn in the problem set unless otherwise noted. For the following problems,
ame: CEM 633: Advanced rganic Chem: ysical Problem Set 6 (Due Thurs, 12/8/16) Please do not look up references until after you turn in the problem set unless otherwise noted. For the following problems,
Electrophilic Carbenes
 Electrophilic Carbenes The reaction of so-called stabilized diazo compounds with late transition metals produces a metal carbene intermediate that is electrophilic. The most common catalysts are Cu(I)
Electrophilic Carbenes The reaction of so-called stabilized diazo compounds with late transition metals produces a metal carbene intermediate that is electrophilic. The most common catalysts are Cu(I)
The aldol reaction with metal enolates proceeds by a chair-like, pericyclic process: favored. disfavored. favored. disfavored
 The aldol reaction with metal enolates proceeds by a chair-like, pericyclic process: Z-enolates: M 2 M 2 syn 2 C 2 favored 2 M 2 anti disfavored E-enolates: M 2 2 C 3 C 3 C 2 favored 2 M M disfavored In
The aldol reaction with metal enolates proceeds by a chair-like, pericyclic process: Z-enolates: M 2 M 2 syn 2 C 2 favored 2 M 2 anti disfavored E-enolates: M 2 2 C 3 C 3 C 2 favored 2 M M disfavored In
Organocopper Reagents
 rganocopper eagents General Information!!! why organocopper reagents? - Efficient method of C-C bond formation - Cu less electropositive than Li or Mg, so -Cu bond less polarized - consequences: 1. how
rganocopper eagents General Information!!! why organocopper reagents? - Efficient method of C-C bond formation - Cu less electropositive than Li or Mg, so -Cu bond less polarized - consequences: 1. how
Stable gold(iii) catalysts by oxidative addition of a carboncarbon
 Stable gold(iii) catalysts by oxidative addition of a carboncarbon bond Chung-Yeh Wu, Takahiro oribe, Christian Borch Jacobsen & F. Dean Toste ature, 517, 449-454 (2015) presented by Ian Crouch Literature
Stable gold(iii) catalysts by oxidative addition of a carboncarbon bond Chung-Yeh Wu, Takahiro oribe, Christian Borch Jacobsen & F. Dean Toste ature, 517, 449-454 (2015) presented by Ian Crouch Literature
Reactivity Umpolung-1 Ready
 eactivity Umpolung-1 eady eactivity Umpolung: reversal of normal polarity electrophiles become nucleophiles nucleophiles become electrophiles complimentary disconnections ormal reactivity: x = heteroatom
eactivity Umpolung-1 eady eactivity Umpolung: reversal of normal polarity electrophiles become nucleophiles nucleophiles become electrophiles complimentary disconnections ormal reactivity: x = heteroatom
Asymmetric Catalysis by Lewis Acids and Amines
 Asymmetric Catalysis by Lewis Acids and Amines Asymmetric Lewis acid catalysis - Chiral (bisooxazoline) copper (II) complexes - Monodentate Lewis acids: the formyl -bond Amine catalysed reactions Asymmetric
Asymmetric Catalysis by Lewis Acids and Amines Asymmetric Lewis acid catalysis - Chiral (bisooxazoline) copper (II) complexes - Monodentate Lewis acids: the formyl -bond Amine catalysed reactions Asymmetric
Answers To Chapter 7 Problems.
 Answers To Chapter Problems.. Most of the Chapter problems appear as end-of-chapter problems in later chapters.. The first reaction is an ene reaction. When light shines on in the presence of light and
Answers To Chapter Problems.. Most of the Chapter problems appear as end-of-chapter problems in later chapters.. The first reaction is an ene reaction. When light shines on in the presence of light and
JACS 1982: A Survey of Papers with a Focus on Synthetic Organic Chemistry
 JAC 1982: A urvey of Papers with a Focus on ynthetic rganic Chemistry Baran Lab Group eting 15 ctober 2003 Carlos A. Guerrero eagents and thods 1 2 [] 2 2 1 1 PhC, Et 3 2 [] 2 1 2 1 By 1982, the [3 + 2]
JAC 1982: A urvey of Papers with a Focus on ynthetic rganic Chemistry Baran Lab Group eting 15 ctober 2003 Carlos A. Guerrero eagents and thods 1 2 [] 2 2 1 1 PhC, Et 3 2 [] 2 1 2 1 By 1982, the [3 + 2]
Structure and Reactivity
 Structure and eactivity Fall Semester 2008 Supplementary Material Prof. Jérôme Waser BC 4306 021 693 93 88 jerome.waser@epfl.ch Assistant: avinia FernandezGonzalez BC 4409 021 693 94 49 davinia.fernandez@epfl.ch
Structure and eactivity Fall Semester 2008 Supplementary Material Prof. Jérôme Waser BC 4306 021 693 93 88 jerome.waser@epfl.ch Assistant: avinia FernandezGonzalez BC 4409 021 693 94 49 davinia.fernandez@epfl.ch
Asymmetric Radical Reactions. Zhen Liu 08/30/2018
 Asymmetric adical eactions Zhen Liu 08/30/2018 Contents Introduction eactions Using Chiral Auxiliary Chiral Lewis Acid-diated eactions Transition tal-catalyzed eactions eactions Using Chiral rganocatalysts
Asymmetric adical eactions Zhen Liu 08/30/2018 Contents Introduction eactions Using Chiral Auxiliary Chiral Lewis Acid-diated eactions Transition tal-catalyzed eactions eactions Using Chiral rganocatalysts
OC 2 (FS 2013) Lecture 3 Prof. Bode. Redox Neutral Reactions and Rearrangements
 C 2 (F 203) Lecture 3 Prof. Bode edox eutral eactions and earrangements Types of edox eutral rganic eactions. eactions with no external reducing or oxidizing agent In this case, one part of the starting
C 2 (F 203) Lecture 3 Prof. Bode edox eutral eactions and earrangements Types of edox eutral rganic eactions. eactions with no external reducing or oxidizing agent In this case, one part of the starting
Dienes & Polyenes: An overview and two key reactions (Ch )
 Dienes & Polyenes: An overview and two key reactions (h. 14.1-14.5) Polyenes contain more than one double bond and are very common in natural products (ex: carotene). Diene chemistry applies to trienes,
Dienes & Polyenes: An overview and two key reactions (h. 14.1-14.5) Polyenes contain more than one double bond and are very common in natural products (ex: carotene). Diene chemistry applies to trienes,
Alchohols, Phenols, Ethers
 Aromatic ompounds Aromaticity lectrophilic aromatic subst. epetition cleophilic aromatic subst. (McM 7th ed, 16.7) Benzyne (McM 7th ed, 16.8) eduction of aromatics (McM 7th ed, 16.10) Alchohols, Phenols,
Aromatic ompounds Aromaticity lectrophilic aromatic subst. epetition cleophilic aromatic subst. (McM 7th ed, 16.7) Benzyne (McM 7th ed, 16.8) eduction of aromatics (McM 7th ed, 16.10) Alchohols, Phenols,
2,3-Sigmatropic Rearrangements in Organic Synthesis
 2,3-igmatropic Rearrangements in rganic ynthesis ctober 25, 2006 Matt aley Crimmins roup igmatropic Rearrangements -concerted pericyclic reactions traditionally thought to be governed by orbital symmetry
2,3-igmatropic Rearrangements in rganic ynthesis ctober 25, 2006 Matt aley Crimmins roup igmatropic Rearrangements -concerted pericyclic reactions traditionally thought to be governed by orbital symmetry
Synthetic Developments Towards the Preparation of Erythromycin and Erythronolide Derivatives
 ynthetic Developments Towards the Preparation of Erythromycin and Erythronolide Derivatives Russell C. mith Denmark Group Meeting 8-9-2005 most extensive project in organic synthesis this phenomenon is
ynthetic Developments Towards the Preparation of Erythromycin and Erythronolide Derivatives Russell C. mith Denmark Group Meeting 8-9-2005 most extensive project in organic synthesis this phenomenon is
VINBLASTINE. H MeO 2 C MeO. OAc. CO 2 Me. Me H
 VIBLATIE 2 C 1 C 2 Ac a 3: catharanthine C 2 Ac C 2 2: ( )-vindoline xidation 5' 2 C 3' 16' 20' Ac C 2 1: (+)-vinblastine b 4 C 2 TFAA, -50 C Polonovski fragmentation 6' 5' 16' C 2 5 TFA 4' 3' 15' 16'
VIBLATIE 2 C 1 C 2 Ac a 3: catharanthine C 2 Ac C 2 2: ( )-vindoline xidation 5' 2 C 3' 16' 20' Ac C 2 1: (+)-vinblastine b 4 C 2 TFAA, -50 C Polonovski fragmentation 6' 5' 16' C 2 5 TFA 4' 3' 15' 16'
Stereoselective Organic Synthesis
 Stereoselective rganic Synthesis Prabhat Arya Professor and Leader, Chemical Biology Program Dean, Academic Affairs, Institute of Life Sciences (An Associate Institute of University of yderabad Supported
Stereoselective rganic Synthesis Prabhat Arya Professor and Leader, Chemical Biology Program Dean, Academic Affairs, Institute of Life Sciences (An Associate Institute of University of yderabad Supported
Chapter 16: Ethers, Epoxides, and Sulfides
 Chapter 16: Ethers, Epoxides, and Sulfides 16.1: omenclature of Ethers, Epoxides, and Sulfides (Please read) 16.2: Structure and Bonding in Ethers and Epoxides The ether oxygen is sp 3 -hybridized and
Chapter 16: Ethers, Epoxides, and Sulfides 16.1: omenclature of Ethers, Epoxides, and Sulfides (Please read) 16.2: Structure and Bonding in Ethers and Epoxides The ether oxygen is sp 3 -hybridized and
Chapter 20 Carboxylic Acid Derivatives. Nucleophilic Acyl Substitution
 ucleophilic Acyl Substitution hapter 20 arboxylic Acid Derivatives ucleophilic Acyl Substitution Y (1) need to have Y as a u Y u u + Y (2) could not happen with aldehydes or ketones as : and : are poor
ucleophilic Acyl Substitution hapter 20 arboxylic Acid Derivatives ucleophilic Acyl Substitution Y (1) need to have Y as a u Y u u + Y (2) could not happen with aldehydes or ketones as : and : are poor
Loudon Chapter 15 Review: Dienes and Aromaticity Jacquie Richardson, CU Boulder Last updated 1/28/2019
 This chapter looks at the behavior of carbon-carbon double bonds when several of them are in the same molecule. There are several possible ways they can be grouped. Conjugated dienes have a continuous
This chapter looks at the behavior of carbon-carbon double bonds when several of them are in the same molecule. There are several possible ways they can be grouped. Conjugated dienes have a continuous
Stereoselective reactions of the carbonyl group
 1 Stereoselective reactions of the carbonyl group We have seen many examples of substrate control in nucleophilic addition to the carbonyl group (Felkin-Ahn & chelation control) If molecule does not contain
1 Stereoselective reactions of the carbonyl group We have seen many examples of substrate control in nucleophilic addition to the carbonyl group (Felkin-Ahn & chelation control) If molecule does not contain
Chiral Brønsted Acid Catalysis
 Chiral Brønsted Acid Catalysis Aryl Aryl Aryl Aryl S CF 3 2 P Fe CF 3 CF 3 2 Jack Liu ov. 16, 2004 CF 3 Introduction Chiral Brønsted acid catalysis in nature: enzymes and peptides Chiral Brønsted acid
Chiral Brønsted Acid Catalysis Aryl Aryl Aryl Aryl S CF 3 2 P Fe CF 3 CF 3 2 Jack Liu ov. 16, 2004 CF 3 Introduction Chiral Brønsted acid catalysis in nature: enzymes and peptides Chiral Brønsted acid
Chapter 4 Electrophilic Addition to Carbon Carbon Multiple Bonds 1. Addition of H X 2. Addition of H OH and addition of Y X 3. Addition to allene and
 Chapter 4 Electrophilic Addition to Carbon Carbon Multiple Bonds 1. Addition of X 2. Addition of and addition of Y X 3. Addition to allene and alkyne 4. Substitution at α-carbon 5. eactions via organoborane
Chapter 4 Electrophilic Addition to Carbon Carbon Multiple Bonds 1. Addition of X 2. Addition of and addition of Y X 3. Addition to allene and alkyne 4. Substitution at α-carbon 5. eactions via organoborane
Chapter 15. Reactions of Aromatic Compounds. 1. Electrophilic Aromatic Substitution Reactions
 hapter 15 eactions of Aromatic ompounds 1. Electrophilic Aromatic Substitution eactions v verall reaction reated by Professor William Tam & Dr. Phillis hang opyright S 3 2 S 4 S 3 2. A General Mechanism
hapter 15 eactions of Aromatic ompounds 1. Electrophilic Aromatic Substitution eactions v verall reaction reated by Professor William Tam & Dr. Phillis hang opyright S 3 2 S 4 S 3 2. A General Mechanism
CEM 852 Final Exam. May 5, 2011
 CEM 852 Final Exam May 5, 2011 This exam consists of 8 pages. Please make certain that your exam has all of the necessary pages. Total points possible for this exam are 150. In answering your questions,
CEM 852 Final Exam May 5, 2011 This exam consists of 8 pages. Please make certain that your exam has all of the necessary pages. Total points possible for this exam are 150. In answering your questions,
CHEM 330. Final Exam December 8, 2010 ANSWERS. This a closed-notes, closed-book exam. The use of molecular models is allowed
 CEM 330 Final Exam December 8, 2010 Your name: ASWERS This a closed-notes, closed-book exam The use of molecular models is allowed This exam contains 10 pages Time: 2h 30 min 1. / 20 2. / 20 3. / 20 4.
CEM 330 Final Exam December 8, 2010 Your name: ASWERS This a closed-notes, closed-book exam The use of molecular models is allowed This exam contains 10 pages Time: 2h 30 min 1. / 20 2. / 20 3. / 20 4.
Chapter 12. Alcohols from Carbonyl Compounds Oxidation-Reduction & Organometallic Compounds. Structure
 Chapter 12 Alcohols from Carbonyl Compounds xidation-eduction & rganometallic Compounds Created by Professor William Tam & Dr. Phillis Chang Structure ~ 120 o ~ 120 o C ~ 120 o Carbonyl carbon: sp 2 hybridized
Chapter 12 Alcohols from Carbonyl Compounds xidation-eduction & rganometallic Compounds Created by Professor William Tam & Dr. Phillis Chang Structure ~ 120 o ~ 120 o C ~ 120 o Carbonyl carbon: sp 2 hybridized
Shi Asymmetric Epoxidation
 Shi Asymmetric Epoxidation Chiral dioxirane strategy: R 3 + 1 xone, ph 10.5, K 2 C 3, H 2, C R 3 formed in situ catalyst (10-20 mol%) is prepared from D-fructose, and its enantiomer from L-sorbose oxone,
Shi Asymmetric Epoxidation Chiral dioxirane strategy: R 3 + 1 xone, ph 10.5, K 2 C 3, H 2, C R 3 formed in situ catalyst (10-20 mol%) is prepared from D-fructose, and its enantiomer from L-sorbose oxone,
Total Synthesis of (+/-)-Goniomitine via a Formal Nitrile/Donor-Acceptor Cyclopropane [3 + 2] Cyclization
![Total Synthesis of (+/-)-Goniomitine via a Formal Nitrile/Donor-Acceptor Cyclopropane [3 + 2] Cyclization Total Synthesis of (+/-)-Goniomitine via a Formal Nitrile/Donor-Acceptor Cyclopropane [3 + 2] Cyclization](/thumbs/82/85235286.jpg) Total Synthesis of (+/-)-Goniomitine via a Formal itrile/donor-acceptor Cyclopropane [3 + 2] Cyclization (-)-Goniomitine Christian L. Morales and Brian Pagenkopf* rganic Letters, ASAP Current Literature
Total Synthesis of (+/-)-Goniomitine via a Formal itrile/donor-acceptor Cyclopropane [3 + 2] Cyclization (-)-Goniomitine Christian L. Morales and Brian Pagenkopf* rganic Letters, ASAP Current Literature
Chem 345 Reaction List: Chem 343 Reactions: Page 1 (You do not need to know the mechanism for the reactions in the boxes).
 Chem 345 eaction List: Chem 343 eactions: Page 1 (You do not need to know the mechanism for the reactions in the boxes). uc - optically active LG 2 optically active uc Alpha carbon (carbon attached to
Chem 345 eaction List: Chem 343 eactions: Page 1 (You do not need to know the mechanism for the reactions in the boxes). uc - optically active LG 2 optically active uc Alpha carbon (carbon attached to
CHEM 203. Final Exam December 15, 2010 ANSWERS. This a closed-notes, closed-book exam. You may use your set of molecular models
 CEM 203 Final Exam December 15, 2010 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This test contains 15 pages Time: 2h 30 min 1. / 16 2. / 15 3. / 24
CEM 203 Final Exam December 15, 2010 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This test contains 15 pages Time: 2h 30 min 1. / 16 2. / 15 3. / 24
water methanol dimethyl ether Ether can only act as a hydrogen bond acceptor H-bond acceptor O R
 Chapter 14: Ethers and Epoxides; Thiols and Sulfides 14.1 Introduction to Ethers An ether group is an oxygen atom that is bonded to two carbons. The ether carbons can be part of alkyl, aryl, or vinyl groups.
Chapter 14: Ethers and Epoxides; Thiols and Sulfides 14.1 Introduction to Ethers An ether group is an oxygen atom that is bonded to two carbons. The ether carbons can be part of alkyl, aryl, or vinyl groups.
Chem 263 Nov 24, Properties of Carboxylic Acids
 Chem 263 ov 24, 2009 Properties of Carboxylic Acids Since carboxylic acids are structurally related to both ketones and aldehydes, we would expect to see some similar structural properties. The carbonyl
Chem 263 ov 24, 2009 Properties of Carboxylic Acids Since carboxylic acids are structurally related to both ketones and aldehydes, we would expect to see some similar structural properties. The carbonyl
Organic Tutorials 3 rd Year Michaelmas Transition Metals in Organic Synthesis: (General paper level) ! 1! Reading
 rganic Tutorials 3 rd Year Michaelmas 2010 Transition Metals in rganic Synthesis: (General paper level) Reading 1. Lecture Course, and suggested references from this. 2. Clayden, Greaves, Warren and Wothers.
rganic Tutorials 3 rd Year Michaelmas 2010 Transition Metals in rganic Synthesis: (General paper level) Reading 1. Lecture Course, and suggested references from this. 2. Clayden, Greaves, Warren and Wothers.
TMSCl imidazole DMF. Ph Ph OTMS. Michael reaction. Michael reaction Ph R 3. epoxidation O R
 eaction using diarylprolinol silyl ether derivatives as catalyst 1) C Et K C 3, ) MgBr, TF TMS hexane, 0 o C TBS p- C 6 4, T C Et 85%, 99% ee Angew. Chem., nt. Ed., 44, 41 (005). rg. Synth., 017, 94, 5.
eaction using diarylprolinol silyl ether derivatives as catalyst 1) C Et K C 3, ) MgBr, TF TMS hexane, 0 o C TBS p- C 6 4, T C Et 85%, 99% ee Angew. Chem., nt. Ed., 44, 41 (005). rg. Synth., 017, 94, 5.
Problem session (3) Daiki Kuwana. Please fill in the blank and explain reaction mechanisms and stereoselectivities.
 Problem session (3) Daiki Kuwana Please fill in the blank and explain reaction mechanisms and stereoselectivities. 1. 1-1 1. (Ac) 2 (10 mol%), DPEphos (20 mol%) Et 3, toluene, 90 C 2. s 4 (14 mol%), M;
Problem session (3) Daiki Kuwana Please fill in the blank and explain reaction mechanisms and stereoselectivities. 1. 1-1 1. (Ac) 2 (10 mol%), DPEphos (20 mol%) Et 3, toluene, 90 C 2. s 4 (14 mol%), M;
CEM 852 Final Exam. May 6, 2010
 CEM 852 Final Exam May 6, 2010 This exam consists of 7 pages. Please make certain that your exam has all of the necessary pages. Total points possible for this exam are 150. n answering your questions,
CEM 852 Final Exam May 6, 2010 This exam consists of 7 pages. Please make certain that your exam has all of the necessary pages. Total points possible for this exam are 150. n answering your questions,
R or S? 2) oxidation numbers of the designated carbon atoms: note: An oxidation number must have a sign. F 3 C OCH 3 oxidation #: OH.
 ame 5 F0-Exam o. Page I. ( points) The following compounds are those used in our study on the mechanism of the chemical carcinogenesis of benzo[a]pyrene. ) Designate in each of the boxes below the stereochemistry
ame 5 F0-Exam o. Page I. ( points) The following compounds are those used in our study on the mechanism of the chemical carcinogenesis of benzo[a]pyrene. ) Designate in each of the boxes below the stereochemistry
Chapter 22: Amines. Organic derivatives of ammonia, NH 3. Nitrogen atom have a lone pair of electrons, making the amine both basic and nucleophilic
 hapter 22: Amines. rganic derivatives of ammonia, 3. itrogen atom have a lone pair of electrons, making the amine both basic and nucleophilic 22.1: Amines omenclature. (please read) sp 3 Amines are classified
hapter 22: Amines. rganic derivatives of ammonia, 3. itrogen atom have a lone pair of electrons, making the amine both basic and nucleophilic 22.1: Amines omenclature. (please read) sp 3 Amines are classified
Organocatalysis Enabled by N-Heterocyclic Carbenes
 rganocatalysis Enabled by -eterocyclic Carbenes Acyl Anions X Y omoenolates Acylazolium Azolium enolate Base Catalysis Jiaming Li 2018/04/27 tability of -heterocyclic Carbenes 2p x,y,z p π 2p x,y,z p π
rganocatalysis Enabled by -eterocyclic Carbenes Acyl Anions X Y omoenolates Acylazolium Azolium enolate Base Catalysis Jiaming Li 2018/04/27 tability of -heterocyclic Carbenes 2p x,y,z p π 2p x,y,z p π
Suggested solutions for Chapter 41
 s for Chapter 41 41 PBLEM 1 Explain how this synthesis of amino acids, starting with natural proline, works. Explain the stereoselectivity of each step after the first. C 2 C 2 3 CF 3 C 2 2 Pd 2 C 2 +
s for Chapter 41 41 PBLEM 1 Explain how this synthesis of amino acids, starting with natural proline, works. Explain the stereoselectivity of each step after the first. C 2 C 2 3 CF 3 C 2 2 Pd 2 C 2 +
O or R E + R. - Keto-Enol Tautomerization (enol form usually very minor for simple ketones)
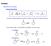 General eactivity base or acid or E + E - Keto-Enol Tautomerization (enol form usually very minor for simple ketones) - Can enhance rate / concentration by addition of acid or base + catalyzed + + + base
General eactivity base or acid or E + E - Keto-Enol Tautomerization (enol form usually very minor for simple ketones) - Can enhance rate / concentration by addition of acid or base + catalyzed + + + base
Chapter 5 Three and Four-Membered Ring Systems
 Chapter 5 Three and Four-mbered ing ystems 5.1 Aziridines Aziridines are good alkylating agents because of their tendency to undergo ring-opening reaction with nucleophiles 例 mitomycin C antibiotic and
Chapter 5 Three and Four-mbered ing ystems 5.1 Aziridines Aziridines are good alkylating agents because of their tendency to undergo ring-opening reaction with nucleophiles 例 mitomycin C antibiotic and
Chapter 7 Substitution Reactions 7.1 Introduction to Substitution Reactions Substitution Reactions: two reactants exchange parts to give new products
 hapter 7 Substitution eactions 7.1 Introduction to Substitution eactions Substitution eactions: two reactants exchange parts to give new products A-B + -D A-D + B- 3 2 + Br 3 2 Br + Elimination eaction:
hapter 7 Substitution eactions 7.1 Introduction to Substitution eactions Substitution eactions: two reactants exchange parts to give new products A-B + -D A-D + B- 3 2 + Br 3 2 Br + Elimination eaction:
Zr-Catalyzed Carbometallation
 -Catalyzed Carbometallation C C C C ML n C C ML n ML n C C C C ML n ML n C C ML n Wipf Group esearch Topic Seminar Juan Arredondo November 13, 2004 Juan Arredondo @ Wipf Group 1 11/14/2004 Carbometallation
-Catalyzed Carbometallation C C C C ML n C C ML n ML n C C C C ML n ML n C C ML n Wipf Group esearch Topic Seminar Juan Arredondo November 13, 2004 Juan Arredondo @ Wipf Group 1 11/14/2004 Carbometallation
Lecture 9: Addition to σ*
 hemistry 201: rganic eaction chanisms Lecture 9: Addition to σ* B fast A not so fast σ* π* δ - δ δ - E M p Addition to σ* orbital leads to bond cleavage ydrogen atoms are always attached to something.
hemistry 201: rganic eaction chanisms Lecture 9: Addition to σ* B fast A not so fast σ* π* δ - δ δ - E M p Addition to σ* orbital leads to bond cleavage ydrogen atoms are always attached to something.
Organometallics Study Meeting
 rganometallics Study eeting 04/21/2011.itsunuma 1. rystal field theory(ft) and ligand field theory(lft) FT: interaction between positively charged metal cation and negative charge on the non-bonding electrons
rganometallics Study eeting 04/21/2011.itsunuma 1. rystal field theory(ft) and ligand field theory(lft) FT: interaction between positively charged metal cation and negative charge on the non-bonding electrons
When something goes wrong. Goya: Mother showing her derformed child to two women Louvre, Paris
 1 ew Catalytic Asymmestric eactions Karl Anker Jørgensen Danish ational eserach Foundation: Center for Catalysis Department of Chemistry, Aarhus University Denmark kaj@chem.au.dk When something goes wrong
1 ew Catalytic Asymmestric eactions Karl Anker Jørgensen Danish ational eserach Foundation: Center for Catalysis Department of Chemistry, Aarhus University Denmark kaj@chem.au.dk When something goes wrong
Synthesis of Amphidinolide X and an Exploration of Key Reactions
 PJM 1/12/05 Synthesis of Amphidinolide X and an Exploration of Key eactions Lepage,.; Kattnig, E.; Furstner, A. JACS, 2004, 126, 15970-15971. 7 13 1 6 19 - Produced by marine dinoflagellates, Amphidinium
PJM 1/12/05 Synthesis of Amphidinolide X and an Exploration of Key eactions Lepage,.; Kattnig, E.; Furstner, A. JACS, 2004, 126, 15970-15971. 7 13 1 6 19 - Produced by marine dinoflagellates, Amphidinium
Scope: 1. S N 2' Displacements. 2. Michael additions. Not Covered:
 Is There a "Crams's ule" for lefins? Part A C=C cleophile Additions cleophilic lefin Additions?! Any reaction which proceeds through electron donation (pair or radical) into the π* orbital of the olefin
Is There a "Crams's ule" for lefins? Part A C=C cleophile Additions cleophilic lefin Additions?! Any reaction which proceeds through electron donation (pair or radical) into the π* orbital of the olefin
Ch.17 Alcohols and Phenols
 Ch.17 Alcohols and Phenols C An alcohol A phenol An enol - Me: wood alcohol: made from wood - industrial preparation of methanol: C + 2 2 400 o C Zinc oxide/chromia C 3 - Ethanol: fermentation of grains
Ch.17 Alcohols and Phenols C An alcohol A phenol An enol - Me: wood alcohol: made from wood - industrial preparation of methanol: C + 2 2 400 o C Zinc oxide/chromia C 3 - Ethanol: fermentation of grains
Advanced Organic Chemistry
 D. A. Evans, G. Lalic Question of the day: Chemistry 530A TBS Me 2 C Me toluene, 130 C 70% TBS C 2 Me H H Advanced rganic Chemistry Me Lecture 16 Cycloaddition Reactions Diels _ Alder Reaction Photochemical
D. A. Evans, G. Lalic Question of the day: Chemistry 530A TBS Me 2 C Me toluene, 130 C 70% TBS C 2 Me H H Advanced rganic Chemistry Me Lecture 16 Cycloaddition Reactions Diels _ Alder Reaction Photochemical
JOC: 1985 Year in Review
 Baran Group eting JC: 1985 Year in eview Syntheses discussed: thodoligies discussed: Quadrone 2 C (+)-irsutic Acid C (±) Coriolin (!)-Longifolene (!)-astanecine Manganese (III)-mediated "-lactone annulation
Baran Group eting JC: 1985 Year in eview Syntheses discussed: thodoligies discussed: Quadrone 2 C (+)-irsutic Acid C (±) Coriolin (!)-Longifolene (!)-astanecine Manganese (III)-mediated "-lactone annulation
Guideline 5: Tactical Bonds
 Guideline 5: Tactical Bonds f the molecule contains more than one functional group and hence there is a choice of which bonds to disconnect, how do you decide? Practice, but here are a few rough guidelines
Guideline 5: Tactical Bonds f the molecule contains more than one functional group and hence there is a choice of which bonds to disconnect, how do you decide? Practice, but here are a few rough guidelines
CHAPTER 22: Amines R 2 H N N N R 1. cadeverine N CH 3. nicotine
 Based on notes by JZ updated 4/2018 Amines are Biologically Important CAPTE 22: Amines Amino acids are the basis of all peptides and proteins. These are the tissue building blocks and nature's catalysts
Based on notes by JZ updated 4/2018 Amines are Biologically Important CAPTE 22: Amines Amino acids are the basis of all peptides and proteins. These are the tissue building blocks and nature's catalysts
Appendix A. Common Abbreviations, Arrows, and Symbols. Abbreviations
 smi97462_apps.qxd 12/6/04 1:09 PM Page A-1 Appendix A ommon Abbreviations, Arrows, and Symbols Abbreviations Ac acetyl, 3 BBN 9-borabicyclo[3.3.1]nonane BINAP 2,2'-bis(diphenylphosphino)-1,1'-binaphthyl
smi97462_apps.qxd 12/6/04 1:09 PM Page A-1 Appendix A ommon Abbreviations, Arrows, and Symbols Abbreviations Ac acetyl, 3 BBN 9-borabicyclo[3.3.1]nonane BINAP 2,2'-bis(diphenylphosphino)-1,1'-binaphthyl
Selective Oxidations SUBSTRATE REAGENT. Non-activated Carbon Atoms. Bugs, enzymes. X 2 / hv. X 2 / hv or ArICl 2
 Selective xidations SUBSTATE EAGET on-activated Carbon Atoms Bugs, enzymes X X 2 / hv X 2 / hv or ArICl 2 Activated on-functionalized Carbon Atoms Se 2 Se 2, PDC Se 2 K t Bu / (Et) 3 P / 2 or LDA / Mo(VI)(
Selective xidations SUBSTATE EAGET on-activated Carbon Atoms Bugs, enzymes X X 2 / hv X 2 / hv or ArICl 2 Activated on-functionalized Carbon Atoms Se 2 Se 2, PDC Se 2 K t Bu / (Et) 3 P / 2 or LDA / Mo(VI)(
Keisuke Suzuki. Baran lab Group Meeting 6/11/16. Shigenobu Umemiya. Akira Suzuki. Takanori Suzuki (Hokkaido University)
 197.D., Teruaki Mukaiyama, University of Tokyo 193 Assistant Professor, Keio University 197 Lecturer, Keio University 199 Assocate Professor, Keio University 1990 Visiting Professor, ET 1994 ull Professor,
197.D., Teruaki Mukaiyama, University of Tokyo 193 Assistant Professor, Keio University 197 Lecturer, Keio University 199 Assocate Professor, Keio University 1990 Visiting Professor, ET 1994 ull Professor,
Non-Metathesis Ruthenium-Catalyzed Reactions for Organic Synthesis
 on-tathesis thenium-catalyzed eactions for rganic Synthesis Tristan Lambert MacMillan Group eting May 23, 2002 I. Properties of thenium II. eductions III. xidations IV. Isomerizations V. C-C bond forming
on-tathesis thenium-catalyzed eactions for rganic Synthesis Tristan Lambert MacMillan Group eting May 23, 2002 I. Properties of thenium II. eductions III. xidations IV. Isomerizations V. C-C bond forming
Enolates: Z(O,R) (O,R)- and E(O,R) (O,R)-enolates
 Enolates: Z(,) (,)- and E(,) (,)-enolates egardless of other groups the encircled ' and - determine whether one is Z(,)- or E(,)- enolate. - - ' ' (E)-enolate (Z)-enolate Enolates: deprotonation 90 ' Most
Enolates: Z(,) (,)- and E(,) (,)-enolates egardless of other groups the encircled ' and - determine whether one is Z(,)- or E(,)- enolate. - - ' ' (E)-enolate (Z)-enolate Enolates: deprotonation 90 ' Most
11.5 PREPARATION AND OXIDATIVE CLEAVAGE OF GLYCOLS
 11.5 PEPAATIN AND XIDATIVE EAVAGE F GY 50 These reactions also provide methods for the formation of carbon carbon bonds. eactions that form carbon carbon bonds are especially important in organic chemistry
11.5 PEPAATIN AND XIDATIVE EAVAGE F GY 50 These reactions also provide methods for the formation of carbon carbon bonds. eactions that form carbon carbon bonds are especially important in organic chemistry
Chemistry of Carbonyl Compounds
 Chemistry of Carbonyl Compounds ucleophilic addition (1,2-add) / substitution Conjugate addition (1,4 add) obinson annulation (McM 23.12) Alkylation of enolate anions namines as enolate equivs. (McM 23.11)
Chemistry of Carbonyl Compounds ucleophilic addition (1,2-add) / substitution Conjugate addition (1,4 add) obinson annulation (McM 23.12) Alkylation of enolate anions namines as enolate equivs. (McM 23.11)
Chem 263 March 28, 2006
 Chem 263 March 28, 2006 Properties of Carboxylic Acids Since carboxylic acids are structurally related to both ketones and aldehydes, we would expect to see some similar structural properties. The carbonyl
Chem 263 March 28, 2006 Properties of Carboxylic Acids Since carboxylic acids are structurally related to both ketones and aldehydes, we would expect to see some similar structural properties. The carbonyl
Total Syntheses of Manzamine A.
 Total yntheses of Manzamine. R E Winkler, 1998 hv D B R akagawa, 2000 [4+2] B R Me P 2 Me B TBDP Pandit, 1991 [4+2] E manzamine Martin, 1999 [4+2] R Me 2 B R R Ramil Baiazitov. ED group meeting. February
Total yntheses of Manzamine. R E Winkler, 1998 hv D B R akagawa, 2000 [4+2] B R Me P 2 Me B TBDP Pandit, 1991 [4+2] E manzamine Martin, 1999 [4+2] R Me 2 B R R Ramil Baiazitov. ED group meeting. February
CHEM 330. Final Exam December 5, 2014 ANSWERS. This a closed-notes, closed-book exam. The use of molecular models is allowed
 CEM 330 Final Exam December 5, 2014 Your name: ASWERS This a closed-notes, closed-book exam The use of molecular models is allowed This exam consists of 12 pages Time: 2h 30 min 1. / 30 2. / 30 3. / 30
CEM 330 Final Exam December 5, 2014 Your name: ASWERS This a closed-notes, closed-book exam The use of molecular models is allowed This exam consists of 12 pages Time: 2h 30 min 1. / 30 2. / 30 3. / 30
Free Radical Reactions in Synthetic Organic Chemistry
 Free adical eactions in ynthetic rganic Chemistry C549.M. Williams Free radical intermediates have been utilized with varying degrees of success in the stereocontrolled formation of C-C bonds. Among the
Free adical eactions in ynthetic rganic Chemistry C549.M. Williams Free radical intermediates have been utilized with varying degrees of success in the stereocontrolled formation of C-C bonds. Among the
Chapter 17: Alcohols and Phenols
 hapter 17: Alcohols and Phenols sp 3 alcohol phenol (aromatic alcohol) pka~ 16-18 pka~ 10 Alcohols contain an group connected to a saturated carbon (sp 3 ) Phenols contain an group connected to a carbon
hapter 17: Alcohols and Phenols sp 3 alcohol phenol (aromatic alcohol) pka~ 16-18 pka~ 10 Alcohols contain an group connected to a saturated carbon (sp 3 ) Phenols contain an group connected to a carbon
Synthesis of the Phomoidrides (CP 225,917 & CP 263,114) Chem. Rev. 2003, 103, 2691.
 ynthesis of the Phomoidrides (P 225,917 & P 263,114) hem. Rev. 2003, 103, 2691. 15 7 26 2 7 16 14 1 2 5 7 2 7 1 2 1 4 2 6 1 5 9 1 7 1 9 2 9 1 5 (+)-P hom oidr ide A ; 7 = (P- 225,917) (+)-P hom oidr ide
ynthesis of the Phomoidrides (P 225,917 & P 263,114) hem. Rev. 2003, 103, 2691. 15 7 26 2 7 16 14 1 2 5 7 2 7 1 2 1 4 2 6 1 5 9 1 7 1 9 2 9 1 5 (+)-P hom oidr ide A ; 7 = (P- 225,917) (+)-P hom oidr ide
Protecting Groups in Organic Synthesis-1 Ready
 Protecting Groups in rganic Synthesis-1 eady Protecting Groups for Alcohols Protecting groups are a sad fact of synthetic chemistry They are usually needed, but rarely desired Many syntheses have stalled
Protecting Groups in rganic Synthesis-1 eady Protecting Groups for Alcohols Protecting groups are a sad fact of synthetic chemistry They are usually needed, but rarely desired Many syntheses have stalled
Chapter 3 Acids and Bases
 hapter 3 Acids and Bases Basic Definitions Associated with Acids and Bases Molecular Definitions of Acids and Bases Molecular Models of Selected Acids Brønsted-Lowry Theory 1. In a Brønsted-Lowry reaction,
hapter 3 Acids and Bases Basic Definitions Associated with Acids and Bases Molecular Definitions of Acids and Bases Molecular Models of Selected Acids Brønsted-Lowry Theory 1. In a Brønsted-Lowry reaction,
Alkenes. sp 2 C 120 o. Electrophilic addition to alkenes. Nucleophilic addition to alkenes (chapt. 23) Radical react. (Polymerization etc, Chapt 7.
 Alkenes Double bond: π and σ sp 2 120 o Electrophilic addition to alkenes E- E Alkene Funktionalized "alkane" Nucleophilic addition to alkenes (chapt. 23) EWG Nu- EWG Nu adical react. (Polymerization etc,
Alkenes Double bond: π and σ sp 2 120 o Electrophilic addition to alkenes E- E Alkene Funktionalized "alkane" Nucleophilic addition to alkenes (chapt. 23) EWG Nu- EWG Nu adical react. (Polymerization etc,
Total Syntheses of Nominine
 Total Syntheses of ominine i Literature t Group eting Lizzie Bryan ctober 17, 2007 Aconite Alkaloids Atidane V eatchane Cycloveatchane Aconitane etisan Muratake,. M.; atsume, M.; akai,. Tetrahedron, 2006,
Total Syntheses of ominine i Literature t Group eting Lizzie Bryan ctober 17, 2007 Aconite Alkaloids Atidane V eatchane Cycloveatchane Aconitane etisan Muratake,. M.; atsume, M.; akai,. Tetrahedron, 2006,
Synthetic Methodology. Using Tertiary Phosphines. as Nucleophilic Catalysts
 Synthetic Methodology Using Tertiary osphines as Nucleophilic Catalysts 1 3 2 u 2 (P 3 ) 3 4 1 2 D. Ma, X. Lu 1988 1 2 Pd 2 (dba) 3.CCl 3 /P 3 /Ac or Pd(Ac) 2 /P 3 1 2 B. M. Trost 1988 1 3 2 u 2 (P 3 )
Synthetic Methodology Using Tertiary osphines as Nucleophilic Catalysts 1 3 2 u 2 (P 3 ) 3 4 1 2 D. Ma, X. Lu 1988 1 2 Pd 2 (dba) 3.CCl 3 /P 3 /Ac or Pd(Ac) 2 /P 3 1 2 B. M. Trost 1988 1 3 2 u 2 (P 3 )
"-Amino Acids: Function and Synthesis
 "-Amino Acids: Function and Synthesis # Conformations of "-Peptides # Biological Significance # Asymmetric Synthesis Sean Brown MacMillan Group eting ovember 14, 2001 Lead eferences: Cheng,. P.; Gellman,
"-Amino Acids: Function and Synthesis # Conformations of "-Peptides # Biological Significance # Asymmetric Synthesis Sean Brown MacMillan Group eting ovember 14, 2001 Lead eferences: Cheng,. P.; Gellman,
Organic Chemistry I (Chem340), Spring Final Exam
 rganic Chemistry I (Chem340), pring 2005 Final Exam This is a closed-book exam. No aid is to be given to or received from another person. Model set and calculator may be used, but cannot be shared. Please
rganic Chemistry I (Chem340), pring 2005 Final Exam This is a closed-book exam. No aid is to be given to or received from another person. Model set and calculator may be used, but cannot be shared. Please
Highlights in the Progress of Biomimetic Strategies for the Construction of Complex Natural Products. Chris Galliford 26 th August 2003
 ighlights in the Progress of Biomimetic Strategies for the Construction of Complex Natural Products Chris Galliford 26 th August 2003 The Biomimetic Approach Inspiration for biomimetic synthesis of a target
ighlights in the Progress of Biomimetic Strategies for the Construction of Complex Natural Products Chris Galliford 26 th August 2003 The Biomimetic Approach Inspiration for biomimetic synthesis of a target
Amines Amines (e.g. RNH 2 ) are organic derivatives of ammonia, NH 3 similar to ammonia
 148Exam 2 notes rganic hemistry (e.g. 2 ) are organic derivatives of ammonia, 3 similar to ammonia omenclature: suffix 2 prefix 2 ( 3 ) 2 Types: Primary (1 o ), secondary (2 o ), tertiary (3 o ) or quaternary
148Exam 2 notes rganic hemistry (e.g. 2 ) are organic derivatives of ammonia, 3 similar to ammonia omenclature: suffix 2 prefix 2 ( 3 ) 2 Types: Primary (1 o ), secondary (2 o ), tertiary (3 o ) or quaternary
3.7. Pyridinium Chloro Chromate (PCC):
 xidation 3.7. Pyridinium hloro hromate (P): 97 l l r r 3 r l P is a very important oxidising reagent which will give controlled oxidation in the case of primary alcohol. It does not give over oxidation
xidation 3.7. Pyridinium hloro hromate (P): 97 l l r r 3 r l P is a very important oxidising reagent which will give controlled oxidation in the case of primary alcohol. It does not give over oxidation
Functional Group Transformations
 Ways to make esters Baeyer-Villiger oxidation Favorskii rearrangement C 2 Et aet/et mcpba Mitsunobu reaction C 2 + ' DEAD, PPh 3 C 2 ' Ph 3 P Et 2 C C 2 Et Ph 3 P Et 2 C C 2 Et Ph 3 P Et 2 C C 2 Et + C
Ways to make esters Baeyer-Villiger oxidation Favorskii rearrangement C 2 Et aet/et mcpba Mitsunobu reaction C 2 + ' DEAD, PPh 3 C 2 ' Ph 3 P Et 2 C C 2 Et Ph 3 P Et 2 C C 2 Et Ph 3 P Et 2 C C 2 Et + C
Chem 263 Nov 7, elimination reaction. There are many reagents that can be used for this reaction. Only three are given in this course:
 hem 263 Nov 7, 2013 Preparation of Ketones and Aldehydes from Alcohols xidation of Alcohols [] must have at least 1 E elimination reaction [] = oxidation; removal of electrons [] = reduction; addition
hem 263 Nov 7, 2013 Preparation of Ketones and Aldehydes from Alcohols xidation of Alcohols [] must have at least 1 E elimination reaction [] = oxidation; removal of electrons [] = reduction; addition
When H and OH add to the alkyne, an enol is formed, which rearranges to form a carbonyl (C=O) group:
 Next Up: Addition of, : The next two reactions are the Markovnikov and non-markovnikov additions of and to an alkyne But you will not see alcohols form in this reaction! When and add to the alkyne, an
Next Up: Addition of, : The next two reactions are the Markovnikov and non-markovnikov additions of and to an alkyne But you will not see alcohols form in this reaction! When and add to the alkyne, an
Organometallics: Hard to define usefully and completely at the same time, but generally: Compounds containing metal-carbon bond(s).
 eady; Catalysis rganometallics: Definitions rganometallics: ard to define usefully and completely at the same time, but generally: Compounds containing metal-carbon bond(s). C C C C C C K 3 o question:????
eady; Catalysis rganometallics: Definitions rganometallics: ard to define usefully and completely at the same time, but generally: Compounds containing metal-carbon bond(s). C C C C C C K 3 o question:????
R N R N R N. primary secondary tertiary
 Chapter 19 Amines omenclature o assification of amines Amines are classified as 1, 2, or 3 based on how many R groups are attached to the nitrogen R R R R R R primary secondary tertiary When there are
Chapter 19 Amines omenclature o assification of amines Amines are classified as 1, 2, or 3 based on how many R groups are attached to the nitrogen R R R R R R primary secondary tertiary When there are
Hamari Chemicals, Ltd. Technology for Life
 amari Chemicals, Ltd. Technology for Life 1 Agenda What amari can offer your company amari s Technology The amari Advantage Core Technology unnatural amino acids and peptides; reduction; chiral technology;
amari Chemicals, Ltd. Technology for Life 1 Agenda What amari can offer your company amari s Technology The amari Advantage Core Technology unnatural amino acids and peptides; reduction; chiral technology;
Tips for taking exams in 852
 Comprehensive Tactical Methods in rganic Synthesis W. D. Wulff 1) Know the relative reactivity of carbonyl compounds Tips for taking exams in 852 Cl > > ' > > ' N2 eg: 'Mg Et ' 1equiv. 1equiv. ' ' Et 50%
Comprehensive Tactical Methods in rganic Synthesis W. D. Wulff 1) Know the relative reactivity of carbonyl compounds Tips for taking exams in 852 Cl > > ' > > ' N2 eg: 'Mg Et ' 1equiv. 1equiv. ' ' Et 50%
Synthetic possibilities Chem 315 Beauchamp 1
 Synthetic possibilities hem Beauchamp Propose reasonable syntheses f the following target molecules (TM-#). You can use the given starting materials and any typical ganic reagents studied in our course
Synthetic possibilities hem Beauchamp Propose reasonable syntheses f the following target molecules (TM-#). You can use the given starting materials and any typical ganic reagents studied in our course
+ + CH 11: Substitution and Elimination Substitution reactions
 C 11: Substitution and Elimination Substitution reactions Things to sort out: Nucleophile Electrophile -- > substrate Leaving Group S N 2 S N 1 E 1 E 2 Analysis Scheme Kinetics Reaction profile Substrates
C 11: Substitution and Elimination Substitution reactions Things to sort out: Nucleophile Electrophile -- > substrate Leaving Group S N 2 S N 1 E 1 E 2 Analysis Scheme Kinetics Reaction profile Substrates
Scandium-Catalyzed Asymmetric Reactions
 Scandium-Catalyzed Asymmetric eactions Jimmy Wu Evans Group Seminar February 11, 2005 I. Background II. eutral BIL Ligands III. Anionic BIL Ligands IV. Pybox Ligands V. Bip yridine Ligands VI. rganop hosp
Scandium-Catalyzed Asymmetric eactions Jimmy Wu Evans Group Seminar February 11, 2005 I. Background II. eutral BIL Ligands III. Anionic BIL Ligands IV. Pybox Ligands V. Bip yridine Ligands VI. rganop hosp
Chiral Catalysis. Chiral Catalyst. Substrate. Chiral Catalyst
 Chiral Catalysis Chiral (stoichiometric) reagents are a very important class of compound but... eed a stoichiometric quantity of the chiral component Unless it is cheap or recoverable this is not very
Chiral Catalysis Chiral (stoichiometric) reagents are a very important class of compound but... eed a stoichiometric quantity of the chiral component Unless it is cheap or recoverable this is not very
CHEM 234: Organic Chemistry II Reaction Sheets
 EM234:rganichemistry eactionsheets ucleophilic addition at carbonyl groups: Grignards and reducing agents u: u u u: u u = or = or l u u u ucleophilic addition at carbonyl groups: oxygen and nitrogen nucleophiles:
EM234:rganichemistry eactionsheets ucleophilic addition at carbonyl groups: Grignards and reducing agents u: u u u: u u = or = or l u u u ucleophilic addition at carbonyl groups: oxygen and nitrogen nucleophiles:
Stereoselective reactions of enolates
 1 Stereoselective reactions of enolates Chiral auxiliaries are frequently used to allow diastereoselective enolate reactions Possibly the most extensively studied are the Evan s oxazolidinones These are
1 Stereoselective reactions of enolates Chiral auxiliaries are frequently used to allow diastereoselective enolate reactions Possibly the most extensively studied are the Evan s oxazolidinones These are
Chapter 8: Nucleophilic Substitution 8.1: Functional Group Transformation By Nucleophilic Substitution
 hapter 8: Nucleophilic Substitution 8.1: Functional Group Transformation By Nucleophilic Substitution Nu: = l,, I Nu - Nucleophiles are Lewis bases (electron-pair donor) Nucleophiles are often negatively
hapter 8: Nucleophilic Substitution 8.1: Functional Group Transformation By Nucleophilic Substitution Nu: = l,, I Nu - Nucleophiles are Lewis bases (electron-pair donor) Nucleophiles are often negatively
Radical Reactions. Radical Stability!!! bond dissociation energies X Y X + Y. bond BDE (kcal/mol) bond BDE (kcal/mol) CH 3 CH 3 CH 2 95 O H R 2 C H
 adical eactions adical Stability!!! bond dissociation energies X Y X Y bond BDE (kcal/mol) bond BDE (kcal/mol) C 3 104 108 C 3 C 2 98 110 95 2 C 102 (-) 93 (C-) 92 C 3 C 3 36 89 85 C 3 C 3 80 adical eactions
adical eactions adical Stability!!! bond dissociation energies X Y X Y bond BDE (kcal/mol) bond BDE (kcal/mol) C 3 104 108 C 3 C 2 98 110 95 2 C 102 (-) 93 (C-) 92 C 3 C 3 36 89 85 C 3 C 3 80 adical eactions
2.222 Practice Problems 2003
 2.222 Practice Problems 2003 Set #1 1. Provide the missing starting compound(s), reagent/solvent, or product to correctly complete each of the following. Most people in the class have not done this type
2.222 Practice Problems 2003 Set #1 1. Provide the missing starting compound(s), reagent/solvent, or product to correctly complete each of the following. Most people in the class have not done this type
Predicting LA-LB reactions
 Predicting LA-LB reactions A chemical reaction is governed by G rxn
Predicting LA-LB reactions A chemical reaction is governed by G rxn
