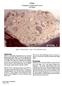
DRAFT Feldspathic Basalt? 3439 grams
|
|
|
- Jack Whitehead
- 5 years ago
- Views:
Transcription
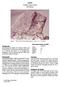
1 14310 Feldspathic Basalt? 3439 grams DRAFT Figure 1: Photograph of before cutting. Scale is 1 cm, cube is 1 inch. NASA photo # S Introduction Sample was found as a separate sample without adhering matrix and thus, may or may not have been a clast within the Fra Mauro Formation. It was apparently not photographed on the lunar surface, but it was apparently half buried and the soil line separating the cratered and uncratered surfaces can be easily distinguished. The following observations are from Hörz et al. (1972): Approximately 60% of the rock surfaces are completely uncratered, extremely fresh and essentially dust free (figure 1). On these surfaces small-scale surface relief features, such as cracks, depressions, and protruding mineral grains are well preserved. One surface of the rock is heavily cratered with features rounded off (figure 2, also see S ). The cratered surface displays a multitude of unambiguous cratering events with central, glass-lined pits and halo and spall zones. The overall shape of the rock and clear-cut relationship between cratered and uncratered areas is highly indicative of a simple surface history. It is suggested that the rock never tumbled after is was deposited on the lunar surface. That is to say, 60% of the rock was buried in the soil and protected from micrometeorite bombardment. Lunar sample has received more study than any other KREEP basalt (Brown and Peckett 1971, Gancarz et al. 1971, Ridley et al. 1972, Longhi et al. 1972, Hollister et al. 1972, Bence and Papike 1972, James 1973, Crawford and Hollister 1974, Meyer 1977 and others). It is a holocrystalline igneous basalt with finegrained subophitic to intergranular texture. However, has high Ni and Ir contents as well as Fe-Ni-P- S melt globules indicating that it is not of pristine igneous origin. In fact, Schonfeld and Meyer 1972 showed that the composition of can be modeled as a mixture of other lunar components (~8% mare, ~65% KREEP, ~25% anorthosite, ~3% meteorite), in a manor similar to a lunar soil!
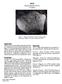
2 Figure 2: PET photograph of rounded and cratered top surface of Scale is 1 cm. NASA# S Petrography Thin sections of show that it is a fine-grained feldspathic basalt with intersertal texture consisting of lath-like plagioclase and anhedral pyroxene (LSPET 1972, Kushiro et al. 1972, Ridley et al. 1972, James 1973 and others). Numerous large (2 mm) blocky phenocrysts of plagioclase are found in the interlocking network of randomly-oriented laths (~200 micron) of plagioclase (figure 3). Pyroxene is found interstitial to the plagioclase. The cores of pyroxene crystals are orthopyroxene which zone to pigeonite (figure 4). Augite sometimes forms epitaxial overgrowths on the pigeonite. Ilmenite occurs in the interstices and is intergrown with the outer margins of the pyroxene. The mesostasis contains globules of Fe-Ni-schreibersitetroilite, Ba-K feldspar, baddeleyite, tranquillityite, Caphosphate and patches of devitrified silica-rich glass. Several investigators report cognate inclusions within LSPET (1972) initially illustrated one such fine-grained cognate inclusion. Kushiro et al. (1972) found that one small patch (~1 mm) with distinct boundary, consisted of plagioclase and pyroxene crystals about one-fifth to one-tenth the size of the main portion. James (1973) also reported cognate inclusions. Mineralogical Mode Gancarz Carlson Ridley et Brown et Longhi et et al et al al al al Plagioclase 59 % vol Pyroxene Ortho Pigeonite Augite 6.9 Opaque Mesostasis Phosphate 0.3
3 Figure 3 : Photomicrograph of thin section (crossed Nicols) of Field of view ~ 5 mm. The wide modal variation almost certainly confirms that is not a homogeneous rock, which should also apply to chemistry (Ridley et al. 1972). Mineralogy Pyroxene: Kushiro et al. (1972), Brown et al. (1972), Ridley et al. (1972), Gancarz et al. (1972), Bence and Papike (1972) have determined the composition of pyroxene in (figure 4). The crystal structure of pyroxenes in were determined by Takeda and Ridley (1972) and orthopyroxene was confirmed. Ghose et al. (1972) studied the structure and exsolution of clinopyroxene. Plagioclase: Ridley et al. (1972) reported unusual zoning patterns in plagioclase in (An ) and suggested that many of the crystals are broken fragments. Wenk et al. (1972) reported the structure of plagioclase in Brown and Peckett (1971) determined that experienced alkali loss during crystallization of plagioclase. Opaque: Ridley et al. (1972) estimate that ~72% of the opaques in are ilmenite, 8% ulvöspinel, 16% troilite, and 4% Fe-Ni. Globules of Fe-Ni-P-S containing schreibersite and troilite have been reported (El Goresy et al. 1972, James 1973). El Goresy et al. give analyses of Fe-Ni (figure 5), ilmenite and ulvöspinel. Phosphates: Whitlockite in was analyzed by Gancarz et al. (1971). Tranquillityite: Brown et al. (1972) reported the Zrrich minerals in El Goresy et al. (1972) give an analysis of tranquillityite in (12% ZrO 2 ). Chemistry A large number of chemical analyses have been made of (tables 1a, 1b and 2). Figure 6 shows the rare-earth-element pattern. Kushiro et al. (1972) reported an analysis of by Haramura using the conventional wet-chemical method (table 1b). Ni and Ir are high, indicating a meteoritic component (nonpristine).
4 Brown 72 Bence & Papike 72 Di Hd Kushiro 72 Ridley 72 En Fs Fo compiled by C Meyer Figure 4: Pyroxene composition of from Brown et al. 1972; Ridley et al. 1972, Kushiro et al and Bence and papike Additional pyroxene data can be found in Gancarz et al (not plotted). Fa Radiogenic age dating Numerous investigators determined the age of with considerable agreement (see table). However, proved difficult to date by the U-Pb method (Tatsumoto et al. 1972, Tera and Wasserburg 1972). Cosmogenic isotopes and exposure ages Rancitelli et al. (1972) and others determined the radionuclide concentrations of as soon as it was available, because of the large Solar Flare Event of 25 January Indeed, the top surface of was found to have significant 56 Co (half life, 77 days) 1000 sample/chondrites La Ce Nd Sm Eu Gd Dy Er Yb Lu Figure 5: Composition of metal grains in (from El Goresy et al. 1972). Figure 6: Normalized rare-earth-element diagram for (isotope dilution mass spectroscopy data only, from Wiesmann and Hubbard 1972 and Phillpotts et al. 1972).
5 Figure 7: Rb-Sr isochron diagram for (from Compston et al. 1972). Figure 11: Rb-Sr isochron diagram for (from Murthy et al. 1972). Figure 9: Internal Rb-Sr isochron for by Mark et al Figure 10: Internal Rb-Sr diagram for determinded by Papanastassiou and Wasserburg Summary of Age Data for Rb-Sr Ar-Ar figure # Murthy et al. (1972) 3.93 ± 0.06 m.y. 8 Compston et al. (1972), DeLaeter et al. (1972) 3.93 ± Papanastassiou and Wasserburg (1972) 3.87 ± Tatsumoto et al. (1972) 3.84 ± 0.04 Mark et al. (1974) 3.94 ± Turner et al. (1971) 3.89 ± 0.04 m.y. 12 York et al. (1972) 3.91 ± Stettler et al. (1973) 3.88 ±
6 Figure 10: Ar-Ar plateau age for (from Stettler et al. 1973). Figure 13: Exposure age of (from Husain et al. 1972). LSPET (1971). Wahlen et al. (1972) reported the 55 Fe, 22 Na, 26 Al, 53 Mn, 36 Cl and 10 Be activity of an interior piece of The depth profile of 53 Mn is reported in Imamura et al. (1974). Turner et al. (1971), York et al. (1972) and Stettler et al. (1973) determined 38 Ar exposure ages of 300 m.y., 333 m.y., 347 m.y. and 250 m.y. Husain et al. (1972) determined 210 m.y. (figure 13). Lugmair and Marti (1972) determined an 81 Kr exposure age of 259 ± 7 m.y. Figure 11: Ar-Ar plateau age for from York et al. (1972). Other Studies Noble gas concentrations of were determined by Husain et al. (1972). Green et al. (1972) and Walker et al. (1972) conducted experiments with powders prepared from to see what phases coexist at various temperatures and pressures (figure 14). Ford et al. (1972) studied the effect of H 2 O and oxygen fugacity on the crystallization of Berdot et al. (1972), Yuhas et al. (1972) and Crozaz et al. (1972) determined the density (number) of nuclear tracks near the surface of The suntan age appears to be only 2-3 m.y. Figure 12: Ar-Ar plateau diagram for (from Turner et al. 1973). Processing Two slabs were cut thru sample (figures 15 and 16). An exploded parts diagram for is also illustrated in Rancitelli et al. (1972) and Hörz et al. (1972).
7 Table 1a. Chemical composition of reference LSPET 71 Wiesmann 73 Hubbard 72 Brunfelt 72 Rose 72 Taylor 72 Willis 72 Helmke 72 Philpotts 72 weight SiO2 % (d) (f) (d) 48.3 (g) TiO (d) 1.22 (e) (f) 1.25 (d) 1.25 (g) Al2O (d) 20.8 (e) (f) (d) (g) FeO (d) 7.93 (e) (f) 8.31 (d) 7.78 (g) MnO (d) 0.11 (e) (f) (d) 0.1 (e) 0.11 (g) MgO (c ) 7.87 (d) 8.8 (e) (f) 7.83 (d) 8 (g) CaO (c ) 12.3 (d) 12.6 (e) (f) (d) (g) Na2O (c ) 0.63 (d) 0.73 (e) (f) 0.72 (d) 0.76 (g) K2O (c ) 0.49 (d) 0.46 (e) (f) (d) 0.52 (g) P2O (d) (f) (d) 0.38 (g) S % 0.02 (d) (d) sum Sc ppm 20 (a) 16.7 (e) (e) V 35 (a) 56 (e) (f) Cr 1100 (a) 1231 (d) 1160 (e) (f) 1163 (d) 1440 (e) 1163 (c ) Co 31 (a) 15.1 (e) (f) 16.1 (e) Ni 165 (a) 64 (d) (f) 150 (e) Cu 11 (a) 3.8 (e) 9 Zn 1.6 (e) <4 Ga 3.7 (e) (f) 4.3 (e) Ge ppb As 0.03 (e) Se Rb 15 (a) 12.8 (c ) 13 (d) 15 (e) (f) 12.1 (d) 12.7 (c ) Sr 250 (a) 188 (c ) 193 (d) 220 (e) (f) 177 (d) (c ) Y 180 (a) 174 (d) (f) 174 (d) Zr 930 (a) 842 (d) (f) 852 (d) 893 (c ) Nb 43 (a) 52 (d) (f) Mo Ru Rh Pd ppb Ag ppb Cd ppb In ppb 30 (e) Sn ppb Sb ppb 4 (e) Te ppb Cs ppm 0.4 (e) 0.7 (f) Ba 630 (a) 617 (c ) 595 (e) (f) 666 (d) 649 (c ) La 36 (a) 56.4 (c ) 53 (e) (f) 57 (e) Ce 144 (c ) 207 (f) 135 (e) 143 (c ) Pr 17 (e) 23 (f) Nd 87 (c ) 91 (f) 93 (e) 87.9 (c ) Sm 24 (c ) 22.7 (e) 23 (f) 25.6 (e) 24.6 (c ) Eu 2.15 (c ) 2.4 (e) 2.28 (f) 2.08 (e) 2.09 (c ) Gd 28 (c ) 29 (f) Tb 5.1 (e) 4.2 (f) 5.3 (e) Dy 32.7 (c ) 27.3 (e) 29 (f) 36.2 (e) 31.7 (c ) Ho 6.5 (e) 6.8 (f) 6.7 (e) Er 19.7 (c ) 16 (e) 19 (f) 20 (e) 19.3 (c ) Tm 3 (f) Yb 30 (a) 18.4 (c ) 12.5 (e) (f) 18.6 (e) 18.1 (c ) Lu 2.76 (e) 2.66 (c ) Hf 17.2 (e) 16 (f) 18 (e) 21 (c ) Ta 2.3 (e) W ppb 1200 (e) 600 (f) Re ppb Reed 72 Os ppb 12 Ir ppb Pt ppb Au ppb 0.3 (e) Th ppm 13.7 (b) Reed (d) 8.6 (e) 12 (f) U ppm 3.7 (b) (e) 3 (f) technique (a) emiss. spec., (b) radiation counting, (c ) IDMS, (d) XRF, (e) INAA, (f) spark source mass spec., (g) AA-Coul.
8 Table 1b. Chemical composition of reference Masuda 72 Baedecker 72 Morgan 72 Haramura 72 Longhi 72 Chi 73 weight Kushiro 72 SiO2 % (h) TiO (h) 1.27 Al2O (h) FeO 8.22 (h) 8.11 MnO 0.13 (h) MgO 7.42 (h) 7.76 CaO (h) Na2O 0.72 (h) 0.81 K2O 0.5 (h) 0.55 P2O (h) S % sum Sc ppm V Cr 1710 (h) Co Ni 210 (e) Cu Zn 1.5 (e) 2.3 (e) Ga 4.2 (e) Ge ppb 90 (e) 130 (e) As Se 120 (e) Rb 11.8 (e) Sr Y Zr 1230 Nb Mo Ru Rh Pd ppb Ag ppb Cd ppb 8.4 (e) 2.6 (e) In ppb 20 (e) 130 (e) Sn ppb Sb ppb 4.5 (e) Te ppb 4 (e) Cs ppm 0.54 (e) Ba La 54.9 (c ) Ce (c ) Pr Nd 88.8 (c ) Sm (c ) Eu 2.33 (c ) Gd (c ) Tb Dy (c ) Ho Er (c ) Tm Yb (c ) Lu 2.6 (c ) Hf 27 Ta W ppb Re ppb 1.2 (e) Os ppb Ir ppb 7.8 (e) 10.5 (e) Pt ppb Au ppb 4.31 (e) Th ppm U ppm technique (a) emiss. spec., (b) radiation counting, (c ) IDMS, (d) XRF, (e) INAA, (f) spark source mass spec., (g) AA-Coul., (h) wet chemistry
9 Figure 14: Pressure-temperature phase diagram for (from Walker et al. 1972). Figure 15: Cutting plan for Figure 16: Group processing photo of Compare with figures 1 and 15. List of Photos # S B & W photos, PET S B & W S color S color Table 2. Light and/or volatile elements for reference LSPET 71 Reed 72 Goel 72 Brunfelt 72 Rose 72 Taylor 72 Gibson 72 Morgan 72 Philpotts 72 Li ppm Be 4.2 B C S N ppm 21 F ppm Cl (e) Br ppb (e) I ppb 4.7 Pb ppm 13 Hg ppb 42 Tl 10 (e) Bi 2.5 (e)
10 References for Bence A.E. and Papike J.J. (1972) Pyroxenes as recorders of lunar basalt petrogenesis: Chemical trends due to crystalliquid interaction. Proc. 3 rd Lunar Sci. Conf Berdot J.L., Chetrit G.C., Lorin J.C., Pellas P. and Poupeau G. (1972) Track studies of Apollo 14 rocks and Apollo 14, Apollo 15 and Luna 16 soils. Proc. 3 rd Lunar Sci. Conf Brown G.M. and Peckett A. (1971) Selective volitization on the lunar surface: Evidence from Apollo 14 feldsparphyric basalts. Nature 234, Brown G.M. and Gay P. (1971) Lunar Antiperthites. Earth Planet. Sci. Lett. 11, Brown G.M., Emeleus C.H., Holland G.J., Peckett A. and Phillips R. (1972) Mineral-chemical variations in Apollo 14 and Apollo 15 basalts and granitic fractions. Proc. 3 rd Lunar Sci. Conf Carlson I.C. and Walton W.J.A. (1978) Apollo 14 Rock Samples. Curators Office. JSC Crawford M.L. and Hollister L.S. (1974) KREEP basalt: a possible partial melt from the lunar interior. Proc. 5 th Lunar Sci. Conf Compston W., Vernon M.J., Berry H. and Rudowski R. (1971) The age of the Fra Mauro Formation: A radiometric older limit. Earth Planet. Sci. Lett. 12, Compston W., Vernon M.J., Berry H., Rudowski R., Gray C.M. and Ware N. (1972) Age and petrogenesis of Apollo 14 basalts (abs). Lunar Planet. Sci. III, The Lunar Science Institute, Houston. Compston W., Vernon M.J., Berry H., Rudowski R., Gray C.M., Ware N., Chappell B.W. and Kaye M. (1972) Apollo 14 mineral ages and the thermal history of the Fra Mauro formation. Proc. 3 rd Lunar Sci. Conf Crozaz G., Drozd R., Hohenberg C.M., Hoyt H.P., Rajan D., Walker R.M. and Yuhas D. (1972b) Solar flare and galactic cosmic ray studies of Apollo,14 and 15 samples. Proc. 3 rd Lunar Sci. Conf de Laeter J.R., Vernon M.J. and Compston W. (1973) Revison of lunar Rb-Sr ages. Geochem. Cosmochim. Acta 37, El Goresy A., Taylor L.A. and Ramdohr P. (1972) Fra Mauro crystalline rocks: Mineralogy, geochemistry and subsolidus reduction of the opaque minerals. Proc. 3 rd Lunar Sci. Conf Eugster O., Eberhardt P., Geiss J., Grogler N., Jungck M., Meier F., Morgell M. and Niederer F. (1984a) Cosmic ray exposure histories of Apollo 14, Apollo 15 and Apollo 16 rocks. Proc. 14 th Lunar Planet. Sci. Conf. in J. Geophys. Res. 89, B498-B512. Ford C.E., Biggar G.M., O Hara M.J., Humphries D.J. and Spencer P.M. (1974) Origin of the lunar highlands (abs). Lunar Sci. V, Lunar Planetary Institute, Houston. Ford C.E., O Hara M.J. and Spencer P.M. (1977) The origin of lunar feldspathic liquids. Phil. Trans. Roy. Soc. London A285, Gancarz A.J., Albee A.L. and Chodos A.A. (1971) Petrologic and mineralogic investigation of some crystalline rocks returned by Apollo 14 mission. Earth Planet. Sci. Lett. 12, Gancarz A.J., Albee A.L. and Chodos A.A. (1972) Comparative petrology of Apollo 16 sample and Apollo 14 samples and Earth Planet. Sci. Lett. 16, Ghose S., Ng G. and Walter L.S. (1972) Cliopyroxenes from Apollo 12 and 14: Exsolution, domain structure and cation order. Proc. 3 rd Lunar Sci. Conf Green D.H., Ware N.G. and Hibberson W.O. (1972) Experimental evidence against the role of selective volatilization on the lunar surface. Nature 238, 450. Husain L., Sutter J.F. and Schaeffer O.A. (1971) Ages of crystalline rocks from Fra Mauro. Science 173, Husain L., Schaeffer O.A., Funkhouser J. and Sutter J. (1972b) The ages of lunar material from Fra Mauro, Hadley Rille and Spur Crater. Proc. 3 rd Lunar Sci. Conf Hörz F., Morrison D.A. and Hartung J.B. (1972) The surface orientation of some Apollo 14 rocks. Modern Geology 3, Imamura M., Nishiizumi K., Honda M., Finkle R.C., Arnold J.R. and Kohl C.P. (1974) Depth profiles of 53Mn in lunar rocks and soils. Proc. 5 th Lunar Sci. Conf James O.B. (1973) Crystallization history of lunar feldspathic basalt U.S. Geol. Survey Prof. Paper 841, 29 pages. Keith J.E., Clark R.S. and Richardson K.A. (1972) Gammaray measurements of Apollo 12, 14 and 15 lunar samples. Proc. 3 rd Lunar Sci. Conf
11 Kushiro I., Ikeda Y. and Nakamura Y. (1972) Petrology of Apollo 14 high-alumina basalt. Proc. 3 rd Lunar Sci. Conf Longhi J., Walker D. and Hays J.F. (1972) Petrology and crystallization history of basalts and Proc. 3 rd Lunar Sci. Conf LSPET (1971) Preliminary examination of lunar samples from Apollo 14. Science 173, Lugmair G.W. and Marti K. (1972) Exposure ages and neutron capture record in lunar samples from Fra Mauro. Proc. 3 rd Lunar Sci. Conf Mark R.K., Lee-Hu C.-N. and Wetherill G.W. (1974) Rb-Sr age of lunar igneous rocks and Geochim. Cosmochim. Acta 38, Meyer C. (1977) Petrology, Mineralogy and Chemistry of KREEP basalt. In Physics and Chemistry of the Earth 10, (Ahrens and Runcorn, eds) Murthy V.R., Evensen N.M., Jahn B.-M. and Coscio M.R. (1972) Apollo 14 and 15 samples: Rb-Sr ages, trace elements, and lunar evolution. Proc. 3 rd Lunar Sci. Conf Neal C.R. and Kramer G.Y. (2006) The petrogenesis of the Apollo 14 high-al mare basalts. Am. Mineralogist 91, Papanastassiou D.A. and Wasserburg G.J. (1971b) Rb-Sr ages of igneous rocks from the Apollo 14 mission and the age of the Fra Mauro Formation. Earth Planet. Sci. Lett. 12, Rancitelli L.A., Perkins R.W., Felix W.D. and Wogman N.A. (1972) Lunar surface processes and cosmic ray characterization from Apollo lunar samples analyses. Proc. 3 rd Lunar Sci. Conf Schonfeld E. and Meyer C. (1972) The abundances of components of the lunar soils by a least-squares mixing model and the formation age of KREEP. Proc. 3 rd Lunar Sci. Conf Simonds C.H., Phinney W.C., Warner J.L., McGee P.E., Geeslin J., Brown R.W. and Rhodes J.M. (1977) Apollo 14 revisited, or breccias aren t so bad after all. Proc. 8 th Lunar Sci. Conf Stettler A., Eberhardt Peter, Geiss J., Grogler N. and Maurer P. (1973) Ar39-Ar40 ages and Ar37-Ar38 exposure ages of lunar rocks. Proc. 4 th Lunar Sci. Conf Sutton R.L., Hait M.H. and Swann G.A. (1972) Geology of the Apollo 14 landing site. Proc. 3 rd Lunar Sci. Conf Swann G.A., Trask N.J., Hait M.H. and Sutton R.L. (1971a) Geologic setting of the Apollo 14 samples. Science 173, Swann G.A., Bailey N.G., Batson R.M., Eggleton R.E., Hait M.H., Holt H.E., Larson K.B., Reed V.S., Schaber G.G., Sutton R.L., Trask N.J., Ulrich G.E. and Wilshire H.G. (1977) Geology of the Apollo 14 landing site in the Fra Mauro Highlands. U.S.G.S. Prof. Paper 880. Swann G.A., Bailey N.G., Batson R.M., Eggleton R.E., Hait M.H., Holt H.E., Larson K.B., McEwen M.C., Mitchell E.D., Schaber G.G., Schafer J.P., Shepard A.B., Sutton R.L., Trask N.J., Ulrich G.E., Wilshire H.G. and Wolfe E.W. (1972) 3. Preliminary Geologic Investigation of the Apollo 14 landing site. In Apollo 14 Preliminary Science Rpt. NASA SP-272. pages Tera F. and Wasserburg G.J. (1972a) U-Th-Pb systematics in three Apollo 14 basalts and the problem of initial Pb in lunar rocks. Earth Planet. Sci. Lett. 14, Tatsumoto M., Hedge C.E., Doe B.R. and Unruh D.M. (1972a) U-Th-Pb and Rb-Sr measurements on some Apollo 14 lunar samples. Proc. 3 rd Lunar Sci. Conf Turner G., Huneke J.C., Podosek F.A. and Wasserburg G.J. (1971) 40Ar-39Ar ages and cosmic ray exposure ages of Apollo 14 samples. Earth Planet. Sci. Lett. 12, Turner G., Huneke J.C., Podosek F.A. and Wasserburg G.J. (1972) Ar40-39 systematics in rocks and separated minerals from Apollo 14. Proc. 3 rd Lunar Sci. Conf Wahlen M., Honda M., Imamura M., Fruchter J.S., Finkel R.C., Kohl C.P., Arnold J.R. and Reedy R.C. (1972) Cosmogenic nuclides in football-sized rocks. Proc. 3 rd Lunar Sci. Conf Wahlen M., Finkel R.C., Imamura M., Kohl C.P. and Arnold J.R. (1973) 60Co in lunar samples. Earth Planet. Sci. Lett. 19, Walker D., Longhi J. and Hays J.F. (1972) Experimental petrology and origin of Fra Mauro rocks and soil. Proc. 3 rd Lunar Sci. Conf Warner J.L. (1972) Metamophism of Apollo 14 breccias. Proc. 3 rd Lunar Sci. Conf Wenk E., Glauser A., Schwander H. and Trommsdorff V. (1972) Twin laws, optic orientation and composition of plagioclase from rocks 12051, and Proc. 3 rd Lunar Sci. Conf
12 Willis J.P., Erlank A.J., Gurney J.J., Theil R.H. and Ahrens L.H. (1972) Major, minor, and trace element data for some Apollo 11, 12, 14 and 15 samples. Proc. 3 rd Lunar Sci. Conf Williams R.J. (1972) The lithification of metamorphism of lunar breccias. Earth Planet. Sci. Lett. 16, Wilshire H.G. and Jackson E.D. (1972) Petrology and stratigraphy of the Fra Mauro Formation at the Apollo 14 site. U.S. Geol. Survey Prof. Paper 785. York D., Kenyon W.J. and Doyle R.J. (1972) 40Ar-39Ar ages of Apollo 14 and 15 samples. Proc. 3 rd Lunar Sci. Conf
grams grams Crystalline matrix Breccia
 14069 24.87 grams 14070-36.56 grams Crystalline matrix Breccia Figure 1: Photo of 14069,0. Scale in cm and mm. S78-28808. 14070 Figure 2: Photo of 14070. Scale is in cm. S71-22168. Introduction 14069 and
14069 24.87 grams 14070-36.56 grams Crystalline matrix Breccia Figure 1: Photo of 14069,0. Scale in cm and mm. S78-28808. 14070 Figure 2: Photo of 14070. Scale is in cm. S71-22168. Introduction 14069 and
14041, and Regolith Breccia 166.3, and 65.2 grams
 14041, and 14045 Regolith Breccia 166.3, 103.2 and 65.2 grams Figure 1a: Sample 14041-14045 on lunar surface. MET and LM in distance. AS14-68-9409. Introduction Samples 14041 14046 are fragments from a
14041, and 14045 Regolith Breccia 166.3, 103.2 and 65.2 grams Figure 1a: Sample 14041-14045 on lunar surface. MET and LM in distance. AS14-68-9409. Introduction Samples 14041 14046 are fragments from a
14315 Unusual Regolith Breccia 115 grams
 Unusual Regolith Breccia 115 grams Figure 1: Photo of,0 after chipping and dusting. Sample is 5 cm across. NASA S86-36340. Introduction was collected as a grab sample from the North Boulder Field (station
Unusual Regolith Breccia 115 grams Figure 1: Photo of,0 after chipping and dusting. Sample is 5 cm across. NASA S86-36340. Introduction was collected as a grab sample from the North Boulder Field (station
74235 Vitrophric Basalt 59 grams
 74235 Vitrophric Basalt 59 grams Figure 1: Photo of 74235. Cube is 1 cm. S73-16017 Figure 2: Map of station 4, Apollo 17. Transcript (while the LMP was discussing the core with CC, the CDR made a discovery)
74235 Vitrophric Basalt 59 grams Figure 1: Photo of 74235. Cube is 1 cm. S73-16017 Figure 2: Map of station 4, Apollo 17. Transcript (while the LMP was discussing the core with CC, the CDR made a discovery)
65055 Basaltic Impact Melt grams
 65055 Basaltic Impact Melt 500.8 grams Figure 1: Photo of 65055 showing large zap pit. Cube is 1 cm. S72-43869. Introduction According to the Apollo 16 Catalog by Ryder and Norman, 65055 is an aluminous,
65055 Basaltic Impact Melt 500.8 grams Figure 1: Photo of 65055 showing large zap pit. Cube is 1 cm. S72-43869. Introduction According to the Apollo 16 Catalog by Ryder and Norman, 65055 is an aluminous,
12052 Pigeonite Basalt 1866 grams
 12052 Pigeonite Basalt 1866 grams Revised Figure 1: Photo of broken side of 12052 showing vugs. Sample is 6.5 cm high. NASA # S70-44633. Summary of Age Data for 12052 Ar/Ar Rb/Sr Nyquist 1977 (recalculated)
12052 Pigeonite Basalt 1866 grams Revised Figure 1: Photo of broken side of 12052 showing vugs. Sample is 6.5 cm high. NASA # S70-44633. Summary of Age Data for 12052 Ar/Ar Rb/Sr Nyquist 1977 (recalculated)
15595 and Vuggy Vitrophyric Pigeonite Basalt and grams
 and 15596 Vuggy Vitrophyric Pigeonite Basalt 237.6 and 224.8 grams Figure 1: Photo of. Sample is about 9 cm across. NASA S71-44491. Figure 2: Photo of 15596 showing surface that was exposed to micrometeorites.
and 15596 Vuggy Vitrophyric Pigeonite Basalt 237.6 and 224.8 grams Figure 1: Photo of. Sample is about 9 cm across. NASA S71-44491. Figure 2: Photo of 15596 showing surface that was exposed to micrometeorites.
DRAFT Coarse-fines 569 grams
 10085 Coarse-fines 569 grams DRAFT Figure 1: Selected coarse fines from 10085. Scale is in mm. Photo from Wood et al. 1969. Introduction!0085 and 10084 were created during the Apollo 11 preliminary examination
10085 Coarse-fines 569 grams DRAFT Figure 1: Selected coarse fines from 10085. Scale is in mm. Photo from Wood et al. 1969. Introduction!0085 and 10084 were created during the Apollo 11 preliminary examination
15556 Vesicular Olivine-normative Basalt grams
 15556 Vesicular Olivine-normative Basalt 1542.3 grams Figure 1: Photo of 15556,0. Largest vesicles are about 6 mm. NASA S87-48187. Introduction 15556 was collected about 60 meters from the edge of Hadley
15556 Vesicular Olivine-normative Basalt 1542.3 grams Figure 1: Photo of 15556,0. Largest vesicles are about 6 mm. NASA S87-48187. Introduction 15556 was collected about 60 meters from the edge of Hadley
72335 Impact melt Breccia grams
 Impact melt Breccia 108.9 grams Figure 1: Location of on boulder #2 on landslide off of South Massif. Boulder is ~ 2-3 meters high. AS17-137-20918. Transcript Hey, that s a different rock, Gene (station
Impact melt Breccia 108.9 grams Figure 1: Location of on boulder #2 on landslide off of South Massif. Boulder is ~ 2-3 meters high. AS17-137-20918. Transcript Hey, that s a different rock, Gene (station
14049 Regolith Breccia grams
 Regolith Breccia 200.1 grams Figure 1: Photo of. Edge of cube is 1 inch. NASA S71-29143. Figure 2: Map of Apollo 14 showing traverse for EVA 2. Introduction The Apollo 14 regolith breccias (vitric matrix
Regolith Breccia 200.1 grams Figure 1: Photo of. Edge of cube is 1 inch. NASA S71-29143. Figure 2: Map of Apollo 14 showing traverse for EVA 2. Introduction The Apollo 14 regolith breccias (vitric matrix
,1,2,3,4,9, Impact Melt Breccia 65.4 grams
 63335 Impact Melt Breccia 65.4 grams,1,2,3,4,9,10,8 Figure 2: 63335,6. Cube is 1 cm. S75-33389.,7 Introduction 63335 is a sample chipped off of Shadow Rock (Ulrich 1973). It was collected as several fragments
63335 Impact Melt Breccia 65.4 grams,1,2,3,4,9,10,8 Figure 2: 63335,6. Cube is 1 cm. S75-33389.,7 Introduction 63335 is a sample chipped off of Shadow Rock (Ulrich 1973). It was collected as several fragments
grams g g g g Regolith Breccia
 15306 134.2 grams 15315 35.6 g 15324 32.3 g 15325 57.8 g 15330 57.8 g Regolith Breccia Figure 1a,b: Photo of dust-covered 15306. Sample is 7 cm across. S71-43064 and 067. Introduction 15306 is a regolith
15306 134.2 grams 15315 35.6 g 15324 32.3 g 15325 57.8 g 15330 57.8 g Regolith Breccia Figure 1a,b: Photo of dust-covered 15306. Sample is 7 cm across. S71-43064 and 067. Introduction 15306 is a regolith
70035 Ilmenite Basalt 5765 grams
 Ilmenite Basalt 5765 grams Figure 1: Photograph of top surface of illustrating vugs and vesicles. Note the smooth, rounded surface shaped by micrometeorite bombardment. Small cube is 1 cm. NASA photo #
Ilmenite Basalt 5765 grams Figure 1: Photograph of top surface of illustrating vugs and vesicles. Note the smooth, rounded surface shaped by micrometeorite bombardment. Small cube is 1 cm. NASA photo #
DRAFT Cataclastic Dunite grams
 72415-72418 Cataclastic Dunite 58.74 grams DRAFT Figure 1: Boulder 3 at Station 2, with large dunite clast (72415-72418). The bright spot is the location of samples. A sample of the boulder matrix (72435)
72415-72418 Cataclastic Dunite 58.74 grams DRAFT Figure 1: Boulder 3 at Station 2, with large dunite clast (72415-72418). The bright spot is the location of samples. A sample of the boulder matrix (72435)
61015 Dimict Breccia 1789 grams
 61015 Dimict Breccia 1789 grams Figure 1: Photo of 61015. NASA # S72-37758. Scale and cube in cm. Note the zap pits on the T1 surface and the residual black glass coating on the sides. Introduction Breccia
61015 Dimict Breccia 1789 grams Figure 1: Photo of 61015. NASA # S72-37758. Scale and cube in cm. Note the zap pits on the T1 surface and the residual black glass coating on the sides. Introduction Breccia
Ilmenite Basalt grams
 Ilmenite Basalt 737.6 grams Figure 1: Picute of 5 meter boulder on rim of Shorty Crater showing where was collected. Trench in forground is location of orange soil sample 74220. Photo number AS17-137-20990
Ilmenite Basalt 737.6 grams Figure 1: Picute of 5 meter boulder on rim of Shorty Crater showing where was collected. Trench in forground is location of orange soil sample 74220. Photo number AS17-137-20990
grams grams Poikilitic Impact Melt Breccia
 64567 13.8 grams 64569-14.3 grams Poikilitic Impact Melt Breccia Figure 1: Photo of 64567. Scale in mm. S72-55386 Mineralogical Mode by Simonds et al. (1973) 64567 64569 Plagioclase 69% 57 Pyroxene 10
64567 13.8 grams 64569-14.3 grams Poikilitic Impact Melt Breccia Figure 1: Photo of 64567. Scale in mm. S72-55386 Mineralogical Mode by Simonds et al. (1973) 64567 64569 Plagioclase 69% 57 Pyroxene 10
grams grams Regolith Breccia
 10019 297 grams 10066 60 grams Regolith Breccia Figure 1: Photo of 10019,1. Cube is 1 inch and scale is in cm. NASA S76-23354. Introduction Kramer et al. (1077) reported that 10019 and 10066 appeared to
10019 297 grams 10066 60 grams Regolith Breccia Figure 1: Photo of 10019,1. Cube is 1 inch and scale is in cm. NASA S76-23354. Introduction Kramer et al. (1077) reported that 10019 and 10066 appeared to
72320 Partially Shadowed Soil (portion frozen) grams. boulder # 2 station 2 South Massif
 Partially Shadowed Soil (portion ) 106.31 grams Nansen Crater boulder # 2 station 2 South Massif Figure 1: Location of soil sample in shadow of boulder 2, at station 2, Apollo 17. NASA photo #AS17-137-20925.
Partially Shadowed Soil (portion ) 106.31 grams Nansen Crater boulder # 2 station 2 South Massif Figure 1: Location of soil sample in shadow of boulder 2, at station 2, Apollo 17. NASA photo #AS17-137-20925.
IV. Governador Valadares clinopyroxenite, 158 grams find
 IV. Governador Valadares clinopyroxenite, 158 grams find Figure IV-1. Photograph of Governador Valadares (158 grams) from Dr. Fernanda Ferrucci via Dr. Giuseppe Cavarretta. Photo taken by L. Spinozzi.
IV. Governador Valadares clinopyroxenite, 158 grams find Figure IV-1. Photograph of Governador Valadares (158 grams) from Dr. Fernanda Ferrucci via Dr. Giuseppe Cavarretta. Photo taken by L. Spinozzi.
grams grams Double Drive tube 61 cm
 68002 583.5 grams 68001 840.7 grams Double Drive tube 61 cm Figure 1a: Surface photo for double drive tube 68001-2. AS16-108-17684. Introduction Station 8 was the closest to South Ray Crater so premission
68002 583.5 grams 68001 840.7 grams Double Drive tube 61 cm Figure 1a: Surface photo for double drive tube 68001-2. AS16-108-17684. Introduction Station 8 was the closest to South Ray Crater so premission
12013 Breccia with granite 82.3 grams
 12013 Breccia with granite 82.3 grams Figure 1: Close-up photo of 12013,11. Field of view is 4.5 cm. NASA# S70-43636 Introduction 12013 is a unique lunar sample, but it did not appear to be remarkable
12013 Breccia with granite 82.3 grams Figure 1: Close-up photo of 12013,11. Field of view is 4.5 cm. NASA# S70-43636 Introduction 12013 is a unique lunar sample, but it did not appear to be remarkable
10003 Ilmenite Basalt (low K) 213 grams
 10003 Ilmenite Basalt (low K) 213 grams Figure 1: Photo of 10003,25. Sample is 5 cm across. NASA S76-25545. Introduction Lunar sample 10003 is a low-k, high-ti basalt (figure 1). It is about 3.9 b.y. old,
10003 Ilmenite Basalt (low K) 213 grams Figure 1: Photo of 10003,25. Sample is 5 cm across. NASA S76-25545. Introduction Lunar sample 10003 is a low-k, high-ti basalt (figure 1). It is about 3.9 b.y. old,
62255 Anorthosite with melt 1239 grams
 62255 Anorthosite with melt 1239 grams Figure 1: Photo of 62255 showing glass splash on anorthosite. Cube is 1 cm. NASA S # S72-38309. Introduction Lunar sample 62255 is significant because it is largely
62255 Anorthosite with melt 1239 grams Figure 1: Photo of 62255 showing glass splash on anorthosite. Cube is 1 cm. NASA S # S72-38309. Introduction Lunar sample 62255 is significant because it is largely
10020 Ilmenite Basalt (low K) 425 grams
 10020 Ilmenite Basalt (low K) 425 grams Figure 1: Photo of 10020 taken in the F-201 during initial processing. Sample is 6 cm across. NASA S-69-45261. Introduction 10020 is a low-k variety of fine-grained
10020 Ilmenite Basalt (low K) 425 grams Figure 1: Photo of 10020 taken in the F-201 during initial processing. Sample is 6 cm across. NASA S-69-45261. Introduction 10020 is a low-k variety of fine-grained
DRAFT Regolith Breccia 155 grams
 12034 Regolith Breccia 155 grams DRAFT Figure 1: Lunar sample 12034 after first saw cut in 1970. NASA S75-34235. Cube is 1 cm. Introduction 12034 was collected from the bottom of the same trench as soil
12034 Regolith Breccia 155 grams DRAFT Figure 1: Lunar sample 12034 after first saw cut in 1970. NASA S75-34235. Cube is 1 cm. Introduction 12034 was collected from the bottom of the same trench as soil
15009 Single Drive Tube Station 6
 15009 Single Drive Tube Station 6 Figure 2: Location of soil samples, trench and drive tube at station 6, Apollo 15. Figure 1: Photo of drive tube 15009 driven in all the way. AS15-86-11565. Introduction
15009 Single Drive Tube Station 6 Figure 2: Location of soil samples, trench and drive tube at station 6, Apollo 15. Figure 1: Photo of drive tube 15009 driven in all the way. AS15-86-11565. Introduction
Element Cube Project (x2)
 Element Cube Project (x2) Background: As a class, we will construct a three dimensional periodic table by each student selecting two elements in which you will need to create an element cube. Helpful Links
Element Cube Project (x2) Background: As a class, we will construct a three dimensional periodic table by each student selecting two elements in which you will need to create an element cube. Helpful Links
7) Applications of Nuclear Radiation in Science and Technique (1) Analytical applications (Radiometric titration)
 7) Applications of Nuclear Radiation in Science and Technique (1) (Radiometric titration) The radioactive material is indicator Precipitation reactions Complex formation reactions Principle of a precipitation
7) Applications of Nuclear Radiation in Science and Technique (1) (Radiometric titration) The radioactive material is indicator Precipitation reactions Complex formation reactions Principle of a precipitation
76215 Vesicular Micropoikilitic Impact Melt Breccia 644 grams
 76215 Vesicular Micropoikilitic Impact Melt Breccia 644 grams 76215 76215 Figure 1: Photo of station 6 boulder with location of 76215 indicated. AS17-140-21421 Figure 2: Photo of 76215. Sample is about
76215 Vesicular Micropoikilitic Impact Melt Breccia 644 grams 76215 76215 Figure 1: Photo of station 6 boulder with location of 76215 indicated. AS17-140-21421 Figure 2: Photo of 76215. Sample is about
15016 Vesicular Olivine-normative Basalt grams
 15016 Vesicular Olivine-normative Basalt 923.7 grams Figure 1: Photograph of vesicular basalt 15016. NASA# S71-46632. Cube is 1 inch. Introduction Lunar Sample 15016 is a highly-vesicular, olivinenormative,
15016 Vesicular Olivine-normative Basalt 923.7 grams Figure 1: Photograph of vesicular basalt 15016. NASA# S71-46632. Cube is 1 inch. Introduction Lunar Sample 15016 is a highly-vesicular, olivinenormative,
10017 Ilmenite Basalt (high K) 973 grams
 10017 Ilmenite Basalt (high K) 973 grams Figure 1: Close-up photo of dusted surface of 10017,81 N1 face. Sample is 4 cm across. NASA #76-25451. Introduction 10017 is the largest rock sample returned by
10017 Ilmenite Basalt (high K) 973 grams Figure 1: Close-up photo of dusted surface of 10017,81 N1 face. Sample is 4 cm across. NASA #76-25451. Introduction 10017 is the largest rock sample returned by
Solutions and Ions. Pure Substances
 Class #4 Solutions and Ions CHEM 107 L.S. Brown Texas A&M University Pure Substances Pure substance: described completely by a single chemical formula Fixed composition 1 Mixtures Combination of 2 or more
Class #4 Solutions and Ions CHEM 107 L.S. Brown Texas A&M University Pure Substances Pure substance: described completely by a single chemical formula Fixed composition 1 Mixtures Combination of 2 or more
65015 Poikilitic Impact Melt Breccia 1802 grams
 65015 Poikilitic Impact Melt Breccia 1802 grams Figure 1: Photos of 65015 showing rounded exposed surface with numerous zap pits and angular portion without zap pits protected by burial in soil. NASA photos
65015 Poikilitic Impact Melt Breccia 1802 grams Figure 1: Photos of 65015 showing rounded exposed surface with numerous zap pits and angular portion without zap pits protected by burial in soil. NASA photos
15058 Pigeonite Basalt 2672 grams
 15058 Pigeonite Basalt 2672 grams Figure 1: Photo of mare basalt 15058 (before dusting). NASA S71-44205. Sample is shaped like a brick about 6 inches long and 3 inches high. Note: see also figures 17 and
15058 Pigeonite Basalt 2672 grams Figure 1: Photo of mare basalt 15058 (before dusting). NASA S71-44205. Sample is shaped like a brick about 6 inches long and 3 inches high. Note: see also figures 17 and
DRAFT. fillet grams grams grams (frozen) grams Vesicular Ilmenite Basalt
 71055 669.6 grams 71035 141.8 grams 71036 118.4 grams (frozen) 71037 14.39 grams Vesicular Ilmenite Basalt DRAFT 71040 71060 71055 71035 71036 71037 fillet Figure 1: This boulder of vesicular basalt from
71055 669.6 grams 71035 141.8 grams 71036 118.4 grams (frozen) 71037 14.39 grams Vesicular Ilmenite Basalt DRAFT 71040 71060 71055 71035 71036 71037 fillet Figure 1: This boulder of vesicular basalt from
Atoms and the Periodic Table
 Atoms and the Periodic Table Parts of the Atom Proton Found in the nucleus Number of protons defines the element Charge +1, mass 1 Parts of the Atom Neutron Found in the nucleus Stabilizes the nucleus
Atoms and the Periodic Table Parts of the Atom Proton Found in the nucleus Number of protons defines the element Charge +1, mass 1 Parts of the Atom Neutron Found in the nucleus Stabilizes the nucleus
The Periodic Table. Periodic Properties. Can you explain this graph? Valence Electrons. Valence Electrons. Paramagnetism
 Periodic Properties Atomic & Ionic Radius Energy Electron Affinity We want to understand the variations in these properties in terms of electron configurations. The Periodic Table Elements in a column
Periodic Properties Atomic & Ionic Radius Energy Electron Affinity We want to understand the variations in these properties in terms of electron configurations. The Periodic Table Elements in a column
Radiometric Dating (tap anywhere)
 Radiometric Dating (tap anywhere) Protons Neutrons Electrons Elements on the periodic table are STABLE Elements can have radioactive versions of itself called ISOTOPES!! Page 1 in your ESRT has your list!
Radiometric Dating (tap anywhere) Protons Neutrons Electrons Elements on the periodic table are STABLE Elements can have radioactive versions of itself called ISOTOPES!! Page 1 in your ESRT has your list!
12042 Soil 255 grams
 Soil 255 grams Figure 1: Photo of location of (Halo Crater). AS12-48-7072 Introduction are fines collected in documented bag 12. They were from the outer flank of Surveyor Crater (figure 2). Figure 2:
Soil 255 grams Figure 1: Photo of location of (Halo Crater). AS12-48-7072 Introduction are fines collected in documented bag 12. They were from the outer flank of Surveyor Crater (figure 2). Figure 2:
Lab Day and Time: Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Revised Impact Melt Rock with Shocked/Melted Anorthosite Cap 11,745 grams
 61016 Impact Melt Rock with Shocked/Melted Anorthosite Cap 11,745 grams Revised Figure 1: Close-up photo of a portion of the top surface of 61016,456 showing dimorphic nature of stone and numeraous zap
61016 Impact Melt Rock with Shocked/Melted Anorthosite Cap 11,745 grams Revised Figure 1: Close-up photo of a portion of the top surface of 61016,456 showing dimorphic nature of stone and numeraous zap
Chemistry 431 Practice Final Exam Fall Hours
 Chemistry 431 Practice Final Exam Fall 2018 3 Hours R =8.3144 J mol 1 K 1 R=.0821 L atm mol 1 K 1 R=.08314 L bar mol 1 K 1 k=1.381 10 23 J molecule 1 K 1 h=6.626 10 34 Js N A = 6.022 10 23 molecules mol
Chemistry 431 Practice Final Exam Fall 2018 3 Hours R =8.3144 J mol 1 K 1 R=.0821 L atm mol 1 K 1 R=.08314 L bar mol 1 K 1 k=1.381 10 23 J molecule 1 K 1 h=6.626 10 34 Js N A = 6.022 10 23 molecules mol
Earth Materials I Crystal Structures
 Earth Materials I Crystal Structures Isotopes same atomic number, different numbers of neutrons, different atomic mass. Ta ble 1-1. Su mmar y of quantu m num bers Name Symbol Values Principal n 1, 2,
Earth Materials I Crystal Structures Isotopes same atomic number, different numbers of neutrons, different atomic mass. Ta ble 1-1. Su mmar y of quantu m num bers Name Symbol Values Principal n 1, 2,
15555 Olivine-normative Basalt 9614 grams
 15555 Olivine-normative Basalt 9614 grams Figure 1: Photo of S1 surface of 15555, illustrating large mirometeorite crater (zap pit) and vuggy nature of rock. NASA S71-43954. Scale is in cm. Introduction
15555 Olivine-normative Basalt 9614 grams Figure 1: Photo of S1 surface of 15555, illustrating large mirometeorite crater (zap pit) and vuggy nature of rock. NASA S71-43954. Scale is in cm. Introduction
CHM 101 PRACTICE TEST 1 Page 1 of 4
 CHM 101 PRACTICE TEST 1 Page 1 of 4 Please show calculations (stuffed equations) on all mathematical problems!! On the actual test, "naked answers, with no work shown, will receive no credit even if correct.
CHM 101 PRACTICE TEST 1 Page 1 of 4 Please show calculations (stuffed equations) on all mathematical problems!! On the actual test, "naked answers, with no work shown, will receive no credit even if correct.
Nucleus. Electron Cloud
 Atomic Structure I. Picture of an Atom Nucleus Electron Cloud II. Subatomic particles Particle Symbol Charge Relative Mass (amu) protons p + +1 1.0073 neutrons n 0 1.0087 electrons e - -1 0.00054858 Compare
Atomic Structure I. Picture of an Atom Nucleus Electron Cloud II. Subatomic particles Particle Symbol Charge Relative Mass (amu) protons p + +1 1.0073 neutrons n 0 1.0087 electrons e - -1 0.00054858 Compare
67016 Feldspathic Fragmental Breccia 4262 grams
 67016 Feldspathic Fragmental Breccia 4262 grams Figure 1: Photo of 67016,1. Cube is 1 inch. NASA # S81-26050. Introduction 67016 is a feldspathic fragmental breccia with both light and dark clasts (figures
67016 Feldspathic Fragmental Breccia 4262 grams Figure 1: Photo of 67016,1. Cube is 1 inch. NASA # S81-26050. Introduction 67016 is a feldspathic fragmental breccia with both light and dark clasts (figures
Spin Cut-off Parameter of Nuclear Level Density and Effective Moment of Inertia
 Commun. Theor. Phys. (Beijing, China) 43 (005) pp. 709 718 c International Academic Publishers Vol. 43, No. 4, April 15, 005 Spin Cut-off Parameter of Nuclear Level Density and Effective Moment of Inertia
Commun. Theor. Phys. (Beijing, China) 43 (005) pp. 709 718 c International Academic Publishers Vol. 43, No. 4, April 15, 005 Spin Cut-off Parameter of Nuclear Level Density and Effective Moment of Inertia
Oriented Ilmenite Basalt 1493 grams
 74275 Oriented Ilmenite Basalt 1493 grams 74275 Figure 1: Location of basalt sample 74275 at Shorty Crater - also see 74220. (falce color) AS17-137-20990 Introduction 74275 is a fine-grained, high-ti mare
74275 Oriented Ilmenite Basalt 1493 grams 74275 Figure 1: Location of basalt sample 74275 at Shorty Crater - also see 74220. (falce color) AS17-137-20990 Introduction 74275 is a fine-grained, high-ti mare
GSA DATA REPOSITORY
 GSA DATA REPOSITORY 2014135 Apatites in lunar KREEP basalts: The missing link to understanding the H isotope systematics of the Moon R. Tartèse, M. Anand, F. M. McCubbin, S. M. Elardo, C. K. Shearer, I.
GSA DATA REPOSITORY 2014135 Apatites in lunar KREEP basalts: The missing link to understanding the H isotope systematics of the Moon R. Tartèse, M. Anand, F. M. McCubbin, S. M. Elardo, C. K. Shearer, I.
Chapter 3: Stoichiometry
 Chapter 3: Stoichiometry Chem 6A Michael J. Sailor, UC San Diego 1 Announcements: Thursday (Sep 29) quiz: Bring student ID or we cannot accept your quiz! No notes, no calculators Covers chapters 1 and
Chapter 3: Stoichiometry Chem 6A Michael J. Sailor, UC San Diego 1 Announcements: Thursday (Sep 29) quiz: Bring student ID or we cannot accept your quiz! No notes, no calculators Covers chapters 1 and
The Periodic Table of Elements
 The Periodic Table of Elements 8 Uuo Uus Uuh (9) Uup (88) Uuq (89) Uut (8) Uub (8) Rg () 0 Ds (9) 09 Mt (8) 08 Hs (9) 0 h () 0 Sg () 0 Db () 0 Rf () 0 Lr () 88 Ra () 8 Fr () 8 Rn () 8 At (0) 8 Po (09)
The Periodic Table of Elements 8 Uuo Uus Uuh (9) Uup (88) Uuq (89) Uut (8) Uub (8) Rg () 0 Ds (9) 09 Mt (8) 08 Hs (9) 0 h () 0 Sg () 0 Db () 0 Rf () 0 Lr () 88 Ra () 8 Fr () 8 Rn () 8 At (0) 8 Po (09)
Ch. 9 NOTES ~ Chemical Bonding NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics.
 Ch. 9 NOTES ~ Chemical Bonding NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics. I. Review: Comparison of ionic and molecular compounds Molecular compounds Ionic
Ch. 9 NOTES ~ Chemical Bonding NOTE: Vocabulary terms are in boldface and underlined. Supporting details are in italics. I. Review: Comparison of ionic and molecular compounds Molecular compounds Ionic
Summary of test results for Daya Bay rock samples. by Patrick Dobson Celia Tiemi Onishi Seiji Nakagawa
 Summary of test results for Daya Bay rock samples by Patrick Dobson Celia Tiemi Onishi Seiji Nakagawa October 2004 Summary A series of analytical tests were conducted on a suite of granitic rock samples
Summary of test results for Daya Bay rock samples by Patrick Dobson Celia Tiemi Onishi Seiji Nakagawa October 2004 Summary A series of analytical tests were conducted on a suite of granitic rock samples
Chem Exam 1. September 26, Dr. Susan E. Bates. Name 9:00 OR 10:00
 Chem 1711 Exam 1 September 26, 2013 Dr. Susan E. Bates Name 9:00 OR 10:00 N A = 6.022 x 10 23 mol 1 I A II A III B IV B V B VI B VII B VIII I B II B III A IV A V A VI A VII A inert gases 1 H 1.008 3 Li
Chem 1711 Exam 1 September 26, 2013 Dr. Susan E. Bates Name 9:00 OR 10:00 N A = 6.022 x 10 23 mol 1 I A II A III B IV B V B VI B VII B VIII I B II B III A IV A V A VI A VII A inert gases 1 H 1.008 3 Li
Made the FIRST periodic table
 Made the FIRST periodic table 1869 Mendeleev organized the periodic table based on the similar properties and relativities of certain elements Later, Henri Moseley organized the elements by increasing
Made the FIRST periodic table 1869 Mendeleev organized the periodic table based on the similar properties and relativities of certain elements Later, Henri Moseley organized the elements by increasing
M10/4/CHEMI/SPM/ENG/TZ2/XX+ CHEMISTRY. Wednesday 12 May 2010 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES
 M10/4/CHEMI/SPM/ENG/TZ/XX+ 106116 CHEMISTRY standard level Paper 1 Wednesday 1 May 010 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES Do not open this examination paper until instructed to do so. Answer
M10/4/CHEMI/SPM/ENG/TZ/XX+ 106116 CHEMISTRY standard level Paper 1 Wednesday 1 May 010 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES Do not open this examination paper until instructed to do so. Answer
Faculty of Natural and Agricultural Sciences Chemistry Department. Semester Test 1. Analytical Chemistry CMY 283. Time: 120 min Marks: 100 Pages: 6
 Faculty of Natural and Agricultural Sciences Chemistry Department Semester Test 1 Analytical Chemistry CMY 283 Date: 5 September 2016 Lecturers : Prof P Forbes, Dr Laurens, Mr SA Nsibande Time: 120 min
Faculty of Natural and Agricultural Sciences Chemistry Department Semester Test 1 Analytical Chemistry CMY 283 Date: 5 September 2016 Lecturers : Prof P Forbes, Dr Laurens, Mr SA Nsibande Time: 120 min
DO NOW: Retrieve your projects. We will be reviewing them again today. Textbook pg 23, answer questions 1-3. Use the section 1.2 to help you.
 DO NOW: Retrieve your projects. We will be reviewing them again today. Textbook pg, answer questions. Use the section. to help you. Chapter test is FRIDAY. The Periodic Table of Elements 8 Uuo Uus Uuh
DO NOW: Retrieve your projects. We will be reviewing them again today. Textbook pg, answer questions. Use the section. to help you. Chapter test is FRIDAY. The Periodic Table of Elements 8 Uuo Uus Uuh
60025 Ferroan Anorthosite 1836 grams
 60025 Ferroan Anorthosite 1836 grams Figure 1: 60025. NASA #S72-41586. Cube and scale are 1 cm. Note the thick black glass coating and numerous micrometeorite pits. Introduction Lunar sample 60025 is a
60025 Ferroan Anorthosite 1836 grams Figure 1: 60025. NASA #S72-41586. Cube and scale are 1 cm. Note the thick black glass coating and numerous micrometeorite pits. Introduction Lunar sample 60025 is a
CLASS TEST GRADE 11. PHYSICAL SCIENCES: CHEMISTRY Test 4: Matter and materials 1
 CLASS TEST GRADE PHYSICAL SCIENCES: CHEMISTRY Test 4: Matter and materials MARKS: 45 TIME: hour INSTRUCTIONS AND INFORMATION. Answer ALL the questions. 2. You may use non-programmable calculators. 3. You
CLASS TEST GRADE PHYSICAL SCIENCES: CHEMISTRY Test 4: Matter and materials MARKS: 45 TIME: hour INSTRUCTIONS AND INFORMATION. Answer ALL the questions. 2. You may use non-programmable calculators. 3. You
Last 4 Digits of USC ID:
 Chemistry 05 B Practice Exam Dr. Jessica Parr First Letter of last Name PLEASE PRINT YOUR NAME IN BLOCK LETTERS Name: Last 4 Digits of USC ID: Lab TA s Name: Question Points Score Grader 8 2 4 3 9 4 0
Chemistry 05 B Practice Exam Dr. Jessica Parr First Letter of last Name PLEASE PRINT YOUR NAME IN BLOCK LETTERS Name: Last 4 Digits of USC ID: Lab TA s Name: Question Points Score Grader 8 2 4 3 9 4 0
Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Part 2. Multiple choice (use answer card). 90 pts. total. 3 pts. each.
 1 Exam I CHEM 1303.001 Name (print legibly) Seat no. On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Part 1. Nomenclature. 10 pts. total. 2 pts. each. Fill in
1 Exam I CHEM 1303.001 Name (print legibly) Seat no. On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Part 1. Nomenclature. 10 pts. total. 2 pts. each. Fill in
Faculty of Natural and Agricultural Sciences Chemistry Department. Semester Test 1 MEMO. Analytical Chemistry CMY 283
 Faculty of Natural and Agricultural Sciences Chemistry Department Semester Test 1 MEMO Analytical Chemistry CMY 283 Date: 5 September 2016 Lecturers : Prof P Forbes, Dr Laurens, Mr SA Nsibande Time: 90
Faculty of Natural and Agricultural Sciences Chemistry Department Semester Test 1 MEMO Analytical Chemistry CMY 283 Date: 5 September 2016 Lecturers : Prof P Forbes, Dr Laurens, Mr SA Nsibande Time: 90
Guide to the Extended Step-Pyramid Periodic Table
 Guide to the Extended Step-Pyramid Periodic Table William B. Jensen Department of Chemistry University of Cincinnati Cincinnati, OH 452201-0172 The extended step-pyramid table recognizes that elements
Guide to the Extended Step-Pyramid Periodic Table William B. Jensen Department of Chemistry University of Cincinnati Cincinnati, OH 452201-0172 The extended step-pyramid table recognizes that elements
(please print) (1) (18) H IIA IIIA IVA VA VIA VIIA He (2) (13) (14) (15) (16) (17)
 CHEM 10113, Quiz 3 September 28, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
CHEM 10113, Quiz 3 September 28, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
The Periodic Table of the Elements
 The Periodic Table of the Elements All matter is composed of elements. All of the elements are composed of atoms. An atom is the smallest part of an element which still retains the properties of that element.
The Periodic Table of the Elements All matter is composed of elements. All of the elements are composed of atoms. An atom is the smallest part of an element which still retains the properties of that element.
February 20, Joe Cerniglia The International Group for Historic Aircraft Recovery (TIGHAR) Job Number: S0CHG688. Dear Joe:
 February 20, 2012 Joe Cerniglia The International Group for Historic Aircraft Recovery (TIGHAR) Subject: ICP-MS Report Job Number: S0CHG688 Dear Joe: Please find enclosed the procedure report for the analysis
February 20, 2012 Joe Cerniglia The International Group for Historic Aircraft Recovery (TIGHAR) Subject: ICP-MS Report Job Number: S0CHG688 Dear Joe: Please find enclosed the procedure report for the analysis
Circle the letters only. NO ANSWERS in the Columns!
 Chemistry 1304.001 Name (please print) Exam 5 (100 points) April 18, 2018 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Circle the letters only. NO ANSWERS in
Chemistry 1304.001 Name (please print) Exam 5 (100 points) April 18, 2018 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Circle the letters only. NO ANSWERS in
HANDOUT SET GENERAL CHEMISTRY I
 HANDOUT SET GENERAL CHEMISTRY I Periodic Table of the Elements 1 2 3 4 5 6 7 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 IA VIIIA 1 2 H He 1.00794 IIA IIIA IVA VA VIA VIIA 4.00262 3 Li 6.941 11 Na 22.9898
HANDOUT SET GENERAL CHEMISTRY I Periodic Table of the Elements 1 2 3 4 5 6 7 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 IA VIIIA 1 2 H He 1.00794 IIA IIIA IVA VA VIA VIIA 4.00262 3 Li 6.941 11 Na 22.9898
1 of 5 14/10/ :21
 X-ray absorption s, characteristic X-ray lines... 4.2.1 Home About Table of Contents Advanced Search Copyright Feedback Privacy You are here: Chapter: 4 Atomic and nuclear physics Section: 4.2 Absorption
X-ray absorption s, characteristic X-ray lines... 4.2.1 Home About Table of Contents Advanced Search Copyright Feedback Privacy You are here: Chapter: 4 Atomic and nuclear physics Section: 4.2 Absorption
Lab Day and Time: Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
EMMR25 Mineralogy: Ol + opx + chlorite + cpx + amphibole + serpentine + opaque
 GSA Data Repository 2017365 Marshall et al., 2017, The role of serpentinite derived fluids in metasomatism of the Colorado Plateau (USA) lithospheric mantle: Geology, https://doi.org/10.1130/g39444.1 Appendix
GSA Data Repository 2017365 Marshall et al., 2017, The role of serpentinite derived fluids in metasomatism of the Colorado Plateau (USA) lithospheric mantle: Geology, https://doi.org/10.1130/g39444.1 Appendix
70017 Ilmenite Basalt 2957 grams
 Ilmenite Basalt 2957 grams Figure 1: Photo of lunar basalt showing vugs and vesicles. Sample is about 10 cm across. Cube is 1 cm. NASA# S73-15723. Introduction Lunar sample is a vesicular, medium-grained,
Ilmenite Basalt 2957 grams Figure 1: Photo of lunar basalt showing vugs and vesicles. Sample is about 10 cm across. Cube is 1 cm. NASA# S73-15723. Introduction Lunar sample is a vesicular, medium-grained,
If anything confuses you or is not clear, raise your hand and ask!
 CHM 1045 Dr. Light s Section December 10, 2002 FINAL EXAM Name (please print) Recitation Section Meeting Time This exam consists of six pages. Make sure you have one of each. Print your name at the top
CHM 1045 Dr. Light s Section December 10, 2002 FINAL EXAM Name (please print) Recitation Section Meeting Time This exam consists of six pages. Make sure you have one of each. Print your name at the top
PHYSICAL SCIENCES GRADE : 10
 PHYSICAL SCIENCES GRADE : 0 TIME : hour TOTAL : 75 INSTRUCTIONS AND INFORMATION. Write your full name on your answer book in the appropriate place. 2. The question paper consists of SEVEN questions. Answer
PHYSICAL SCIENCES GRADE : 0 TIME : hour TOTAL : 75 INSTRUCTIONS AND INFORMATION. Write your full name on your answer book in the appropriate place. 2. The question paper consists of SEVEN questions. Answer
Using the Periodic Table
 MATH SKILLS TRANSPARENCY WORKSHEET Using the Periodic Table 6 Use with Chapter 6, Section 6.2 1. Identify the number of valence electrons in each of the following elements. a. Ne e. O b. K f. Cl c. B g.
MATH SKILLS TRANSPARENCY WORKSHEET Using the Periodic Table 6 Use with Chapter 6, Section 6.2 1. Identify the number of valence electrons in each of the following elements. a. Ne e. O b. K f. Cl c. B g.
Lunar Glossary. Note to the User: Glossary
 Lunar Glossary Note to the User: A number of terms are unique to lunar science or are at least used in a specialized sense. The following brief glossary is an attempt to define these unique terms plus
Lunar Glossary Note to the User: A number of terms are unique to lunar science or are at least used in a specialized sense. The following brief glossary is an attempt to define these unique terms plus
What monitoring techniques are appropriate and effective for detecting CO2 migration in groundwater: isotope-based monitoring Philippe Négrel
 What monitoring techniques are appropriate and effective for detecting CO2 migration in groundwater: isotope-based monitoring Philippe Négrel Acting in complicity with Pauline Humez. Results from Pauline
What monitoring techniques are appropriate and effective for detecting CO2 migration in groundwater: isotope-based monitoring Philippe Négrel Acting in complicity with Pauline Humez. Results from Pauline
Chemistry 1 First Lecture Exam Fall Abbasi Khajo Levine Mathias Mathias/Ortiz Metlitsky Rahi Sanchez-Delgado Vasserman
 Chemistry 1 First Lecture Exam Fall 2011 Page 1 of 9 NAME Circle the name of your recitation/lab instructor(s) Abbasi Khajo Levine Mathias Mathias/Ortiz Metlitsky Rahi Sanchez-Delgado Vasserman Before
Chemistry 1 First Lecture Exam Fall 2011 Page 1 of 9 NAME Circle the name of your recitation/lab instructor(s) Abbasi Khajo Levine Mathias Mathias/Ortiz Metlitsky Rahi Sanchez-Delgado Vasserman Before
5 questions, 3 points each, 15 points total possible. 26 Fe Cu Ni Co Pd Ag Ru 101.
 Physical Chemistry II Lab CHEM 4644 spring 2017 final exam KEY 5 questions, 3 points each, 15 points total possible h = 6.626 10-34 J s c = 3.00 10 8 m/s 1 GHz = 10 9 s -1. B= h 8π 2 I ν= 1 2 π k μ 6 P
Physical Chemistry II Lab CHEM 4644 spring 2017 final exam KEY 5 questions, 3 points each, 15 points total possible h = 6.626 10-34 J s c = 3.00 10 8 m/s 1 GHz = 10 9 s -1. B= h 8π 2 I ν= 1 2 π k μ 6 P
Figure of rare earth elemental abundances removed due to copyright restrictions.
 Figure of rare earth elemental abundances removed due to copyright restrictions. See figure 3.1 on page 26 of Tolstikhin, Igor and Jan Kramers. The Evolution of Matter: From the Big Bang to the Present
Figure of rare earth elemental abundances removed due to copyright restrictions. See figure 3.1 on page 26 of Tolstikhin, Igor and Jan Kramers. The Evolution of Matter: From the Big Bang to the Present
02/05/09 Last 4 Digits of USC ID: Dr. Jessica Parr
 Chemistry 05 B First Letter of PLEASE PRINT YOUR NAME IN BLOCK LETTERS Exam last Name Name: 02/05/09 Last 4 Digits of USC ID: Dr. Jessica Parr Lab TA s Name: Question Points Score Grader 2 2 9 3 9 4 2
Chemistry 05 B First Letter of PLEASE PRINT YOUR NAME IN BLOCK LETTERS Exam last Name Name: 02/05/09 Last 4 Digits of USC ID: Dr. Jessica Parr Lab TA s Name: Question Points Score Grader 2 2 9 3 9 4 2
Why all the repeating Why all the repeating Why all the repeating Why all the repeating
 Why all the repeating Why all the repeating Why all the repeating Why all the repeating Patterns What Patterns have you observed in your life? Where to Get Help If you don t understand concepts in chapter
Why all the repeating Why all the repeating Why all the repeating Why all the repeating Patterns What Patterns have you observed in your life? Where to Get Help If you don t understand concepts in chapter
CHEM 108 (Spring-2008) Exam. 3 (105 pts)
 CHEM 08 (Spring-008) Exam. (05 pts) Name: --------------------------------------------------------------------------, CLID # -------------------------------- LAST NAME, First (Circle the alphabet segment
CHEM 08 (Spring-008) Exam. (05 pts) Name: --------------------------------------------------------------------------, CLID # -------------------------------- LAST NAME, First (Circle the alphabet segment
PERIODIC TABLE OF THE ELEMENTS
 Useful Constants and equations: K = o C + 273 Avogadro's number = 6.022 x 10 23 d = density = mass/volume R H = 2.178 x 10-18 J c = E = h = hc/ h = 6.626 x 10-34 J s c = 2.998 x 10 8 m/s E n = -R H Z 2
Useful Constants and equations: K = o C + 273 Avogadro's number = 6.022 x 10 23 d = density = mass/volume R H = 2.178 x 10-18 J c = E = h = hc/ h = 6.626 x 10-34 J s c = 2.998 x 10 8 m/s E n = -R H Z 2
Chemistry 2 Exam Roane State Academic Festival. Name (print neatly) School
 Name (print neatly) School There are fifteen question on this exam. Each question is weighted equally. n the answer sheet, write your name in the space provided and your answers in the blanks provided.
Name (print neatly) School There are fifteen question on this exam. Each question is weighted equally. n the answer sheet, write your name in the space provided and your answers in the blanks provided.
CHEM 10113, Quiz 5 October 26, 2011
 CHEM 10113, Quiz 5 October 26, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
CHEM 10113, Quiz 5 October 26, 2011 Name (please print) All equations must be balanced and show phases for full credit. Significant figures count, show charges as appropriate, and please box your answers!
TABLE DR2. Lu-Hf ISOTOPIC DATA FOR WHOLE ROCK SAMPLES AND ZIRCONS [Lu] [Hf]
![TABLE DR2. Lu-Hf ISOTOPIC DATA FOR WHOLE ROCK SAMPLES AND ZIRCONS [Lu] [Hf] TABLE DR2. Lu-Hf ISOTOPIC DATA FOR WHOLE ROCK SAMPLES AND ZIRCONS [Lu] [Hf]](/thumbs/72/67409273.jpg) TABLE DR1. LOWER CRUSTAL GRANULITE XENOLITH DERIVATION AND MINERALOGY Sample Kimberlite Type Mineralogy KX1-1 Lace s gt + qz + sa + rt (sil, ky, gr, su, cor, zr, mz) KX1-2 Lace s gt + sa + qz + rt (sil,
TABLE DR1. LOWER CRUSTAL GRANULITE XENOLITH DERIVATION AND MINERALOGY Sample Kimberlite Type Mineralogy KX1-1 Lace s gt + qz + sa + rt (sil, ky, gr, su, cor, zr, mz) KX1-2 Lace s gt + sa + qz + rt (sil,
NAME: FIRST EXAMINATION
 1 Chemistry 64 Winter 1994 NAME: FIRST EXAMINATION THIS EXAMINATION IS WORTH 100 POINTS AND CONTAINS 4 (FOUR) QUESTIONS THEY ARE NOT EQUALLY WEIGHTED! YOU SHOULD ATTEMPT ALL QUESTIONS AND ALLOCATE YOUR
1 Chemistry 64 Winter 1994 NAME: FIRST EXAMINATION THIS EXAMINATION IS WORTH 100 POINTS AND CONTAINS 4 (FOUR) QUESTIONS THEY ARE NOT EQUALLY WEIGHTED! YOU SHOULD ATTEMPT ALL QUESTIONS AND ALLOCATE YOUR
(C) Pavel Sedach and Prep101 1
 (C) Pavel Sedach and Prep101 1 (C) Pavel Sedach and Prep101 1 (C) Pavel Sedach and Prep101 2 (C) Pavel Sedach and Prep101 2 (C) Pavel Sedach and Prep101 3 (C) Pavel Sedach and Prep101 3 (C) Pavel Sedach
(C) Pavel Sedach and Prep101 1 (C) Pavel Sedach and Prep101 1 (C) Pavel Sedach and Prep101 2 (C) Pavel Sedach and Prep101 2 (C) Pavel Sedach and Prep101 3 (C) Pavel Sedach and Prep101 3 (C) Pavel Sedach
Atomic Structure & Interatomic Bonding
 Atomic Structure & Interatomic Bonding Chapter Outline Review of Atomic Structure Atomic Bonding Atomic Structure Atoms are the smallest structural units of all solids, liquids & gases. Atom: The smallest
Atomic Structure & Interatomic Bonding Chapter Outline Review of Atomic Structure Atomic Bonding Atomic Structure Atoms are the smallest structural units of all solids, liquids & gases. Atom: The smallest
Dept., Univ. of ela as are, 'i?ewark, DE Approximately 130 low specific gravity ((2.601, high silica
 HIGH (760%) SiO LUNAR GLASS%. ir.f. Glass, Geoloey Dept., Univ. of ela as are, 'i?ewark, DE. 1971 1 Approximately 130 low specific gravity ((2.601, high silica p602) glass particles recovered from a 4.88
HIGH (760%) SiO LUNAR GLASS%. ir.f. Glass, Geoloey Dept., Univ. of ela as are, 'i?ewark, DE. 1971 1 Approximately 130 low specific gravity ((2.601, high silica p602) glass particles recovered from a 4.88
HANDOUT SET GENERAL CHEMISTRY II
 HANDOUT SET GENERAL CHEMISTRY II Periodic Table of the Elements 1 2 3 4 5 6 7 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 IA VIIIA 1 2 H He 1.00794 IIA IIIA IVA VA VIA VIIA 4.00262 3 Li 6.941 11 Na 22.9898
HANDOUT SET GENERAL CHEMISTRY II Periodic Table of the Elements 1 2 3 4 5 6 7 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 IA VIIIA 1 2 H He 1.00794 IIA IIIA IVA VA VIA VIIA 4.00262 3 Li 6.941 11 Na 22.9898
1 Genesis 1:1. Chapter 10 Matter. Lesson. Genesis 1:1 In the beginning God created the heavens and the earth. (NKJV)
 1 Genesis 1:1 Genesis 1:1 In the beginning God created the heavens and the earth. (NKJV) 1 Vocabulary Saturated having all the solute that can be dissolved at that temperature Neutron a particle with no
1 Genesis 1:1 Genesis 1:1 In the beginning God created the heavens and the earth. (NKJV) 1 Vocabulary Saturated having all the solute that can be dissolved at that temperature Neutron a particle with no
INSTRUCTIONS: CHEM Exam I. September 13, 1994 Lab Section
 CHEM 1314.05 Exam I John I. Gelder September 13, 1994 Name TA's Name Lab Section Please sign your name below to give permission to post, by the last 4 digits of your student I.D. number, your course scores
CHEM 1314.05 Exam I John I. Gelder September 13, 1994 Name TA's Name Lab Section Please sign your name below to give permission to post, by the last 4 digits of your student I.D. number, your course scores
Lab Day and Time: Instructions. 1. Do not open the exam until you are told to start.
 Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
Name: Lab Day and Time: Instructions 1. Do not open the exam until you are told to start. 2. This exam is closed note and closed book. You are not allowed to use any outside material while taking this
