Prof. Jason D. Kahn Your Signature: Exams written in pencil or erasable ink will not be re-graded under any circumstances.
|
|
|
- Frederica Robbins
- 5 years ago
- Views:
Transcription
1 1 Biochemistry 461, Section I February 27, 1997 Exam #1 Prof. Jason D. Kahn Your Printed ame: Your SS#: Your Signature: You have 80 minutes for this exam. Exams written in pencil or erasable ink will not be re-graded under any circumstances. Some information which may be useful is provided on the bottom half of the next page. Explanations should be concise, a couple of sentences. You will need a calculator for this exam, and no other study aids or materials are permitted. Useful Information: K a = [ + ][A - ]/[A] for the dissociation of A, so p = pk a + log ([A - ]/[A]) K w = = [ - ][ + ] RT = 2476 J/mole today DG = DG + RTlnQ, where Q has the form of an equilibrium constant DG = -nfde, where F = J/(V mole), n = number of electrons transferred A = particles per mole
2 2 1. (18 pts) Redox reactions and the ernst equation. The oxidation of AD by sulfate can be used to create a proton gradient, which ATP synthase then taps to make ATP, as in the sketch to the right. The relevant standard reduction potentials at p 7 are given below. AD e - AD E = V S e - S E = 0.48 V (a; 8 pts) Write the balanced equation for this redox reaction, calculate DE and DG, and calculate the equilibrium constant for the reaction. (b; 6 pts) If the p is 5 on the inside of the compartment and 7 on the outside, what is DG for moving a proton into the compartment? (Ignore any membrane potentials.) ow many protons can be pumped per AD oxidized?
3 3 (c; 4 pts) If the volume of the compartment is liters, how many protons are in it at p 5? If the single ATP synthase in the membrane can allow 10 protons per second to exit the compartment, how long can ATP synthesis continue once AD oxidation is shut off? 2. (20 points) Bioenergetics. (a; 4 pts) List two ways that cells store energy. The hydrolysis of the b-g phosphoanhydride bond of adenosine triphosphate (ATP) is accompanied by a large negative free energy change: ATP ADP + P i DG = kj/mole (b; 7 pts) Draw the triphosphate moiety of ATP in its predominant ionic form at p 7, adding onto the structure for adenosine given below. Indicate the high-energy phosphoanhydride bonds. Give two reasons for the large negative DG for ATP hydrolysis. In other words, why are phosphoanhydride bonds high-energy? There is space on the next page if you need it.
4 4 (c; 5 pts) Calculate DG for ATP hydrolysis at [ADP] = 2 mm, [ATP] = 8 mm, [P i ] = 5 mm. Assume the biochemical standard state. (d; 4 pts) The ATP hydrolysis reaction actually liberates a proton, as below. ormally we simply ignore this as part of working at the biochemical standard state (p 7). ATP ADP -3 + P DG = kj/mole The equation shows that decreasing the p reduces the driving force for hydrolysis. In spite of this fact, it is found that decreasing the p increases the rate of hydrolysis of ATP. ow can you explain this apparent paradox?
5 5 3. (20 points) Amino acids, peptides, and hydrogen bonding. (a; 10 pts) Draw the covalent structure of the tripeptide Tyr-Lys-Leu, with all functional groups in their predominant ionic forms at p 7. Indicate which protons would be removed at p 12, which is above the pk a of all ionizable side chains except Arg. Write the 1-letter code for each of the three amino acids below the structure. (b; 3 pts) Which amino acid provides biological buffering capacity? Why? ame the two sulfurcontaining amino acids (full names for all three, please). (c; 7 pts) n the picture of adenosine at the right, circle the s which can be donated in hydrogen bonds. Draw in a plausible structure for the asparagine side chain making two hydrogen bonds to the adenine ring. Adenine Ring
6 6 4. (22 points) Secondary structure and the Ramachandran plot. (a; 4 pts) n the left extended polypeptide below, indicate which four atoms define the Y angle for amino acid i, which is bracketed by the dashed lines. n the right side indicate which four atoms define the F angle. n the left-hand diagram, draw two parallelograms indicating which sets of atoms are constrained to lie in a plane. Cai-1 Cai Cai+1 Cai-1 Cai Cai+1 R R Y F (b; 8 pts) Draw in three residues of the antiparallel b-sheet partner below the extended polypeptide chain drawn here. Include backbone hydrogen bonds and indicate where the next strand (i.e. if you were to draw a third one) would -bond. Indicate side chains with R. Indicate the terminus and the C terminus of the strand you draw. R R R R
7 7 (c; 8 pts) Discuss how a-helices and b-sheets can exhibit sidedness, i.e. asymmetric chemical properties, in different ways. Draw sketches to illustrate. (d; 2 pts) ame the 3-D molecule viewer program used in the in class computer demonstrations of secondary structure.
8 8 5. (20 points) Acid-Base Chemistry. Tris(hydroxymethyl)aminomethane or Tris, (C 2 ) 3 C 2, is a commonly used buffer in biochemical experiments. The pk a for the reaction below is 8.06 at 25 C. Tris + Tris + + (a; 7 pts) Using the information above and the equation for water self-dissociation (K w ), calculate the p of a 0.1 M solution of Tris (i.e. c 0 = 0.1 M), assuming that what you spoon out of the bottle is the unprotonated form ( Tris base ). (b; 6 pts) ow much of a 10 M stock of the strong acid Cl would you have to add to 100 ml of the above solution to bring the p down to 7.5? Ignore the volume change of the solution.
9 9 (c; 4 pts) Tris has the unfortunate property that its pk a is quite temperature dependent, decreasing to 6.66 at 75 C. What would the p of the solution you made in (b) become at 75 C, ignoring any change in K w? (int: imagine you had added the acid at 75 C.) If you could not answer (b), assume 1 ml of Cl was added. (d; 3 pts) The pk a s of many weak acids decrease with increasing temperature, i.e. they become stronger acids. Speculate as to why this might be. Do ot Write Below This Line Score: Question 1: out of 18: ernst equation Question 2: out of 20: Thermodynamics and bioenergetics Question 3: out of 20: Amino acids and peptides Question 4: out of 22: Secondary structure Question 5: out of 20: Acid-Base Total: out of 100
Prof. Jason Kahn Your Signature: Exams written in pencil or erasable ink will not be re-graded under any circumstances.
 1 Biochemistry 461 February 16, 1995 Exam #1 Prof. Jason Kahn Your Printed Name: Your SS#: Your Signature: You have 75 minutes for this exam. Exams written in pencil or erasable ink will not be re-graded
1 Biochemistry 461 February 16, 1995 Exam #1 Prof. Jason Kahn Your Printed Name: Your SS#: Your Signature: You have 75 minutes for this exam. Exams written in pencil or erasable ink will not be re-graded
Biochemistry and Physiology ID #:
 BCHM 463 Your Name: Biochemistry and Physiology ID #: Exam II, November 4, 2002 Prof. Jason Kahn You have 50 minutes for this exam. Exams written in pencil or erasable ink will not be re-graded under any
BCHM 463 Your Name: Biochemistry and Physiology ID #: Exam II, November 4, 2002 Prof. Jason Kahn You have 50 minutes for this exam. Exams written in pencil or erasable ink will not be re-graded under any
S H/T 0 ph = log([h + ]) E = mc 2 S = klnw G = H T S ph = pk a + log([a ]/[HA]) K a = [H + ][A ]/[HA] G = RTlnK eq e iπ + 1 = 0
![S H/T 0 ph = log([h + ]) E = mc 2 S = klnw G = H T S ph = pk a + log([a ]/[HA]) K a = [H + ][A ]/[HA] G = RTlnK eq e iπ + 1 = 0 S H/T 0 ph = log([h + ]) E = mc 2 S = klnw G = H T S ph = pk a + log([a ]/[HA]) K a = [H + ][A ]/[HA] G = RTlnK eq e iπ + 1 = 0](/thumbs/90/101592193.jpg) Biochemistry 463, Summer II Your Name: University of Maryland, College Park Your SID #: Biochemistry and Physiology Prof. Jason Kahn Exam I (100 points total) July 27, 2007 You have 80 minutes for this
Biochemistry 463, Summer II Your Name: University of Maryland, College Park Your SID #: Biochemistry and Physiology Prof. Jason Kahn Exam I (100 points total) July 27, 2007 You have 80 minutes for this
(6 pts) List three general characteristics shared by stable secondary structures.
 Biochemistry 461, Section I Your Name: March 19, 1998 Exam #2 Your SS#: Prof. Jason D. Kahn Your Signature: Please have photo ID available. You have 80 minutes for this exam. Exams written in pencil or
Biochemistry 461, Section I Your Name: March 19, 1998 Exam #2 Your SS#: Prof. Jason D. Kahn Your Signature: Please have photo ID available. You have 80 minutes for this exam. Exams written in pencil or
General Chemistry and Energetics Your Section #: Exam I (100 points total) October 6, 2010
 Chemistry 271, Section 23xx Your Name: Prof. Jason Kahn University of Maryland, College Park Your SID #: General Chemistry and Energetics Your Section #: Exam I (100 points total) October 6, 2010 You have
Chemistry 271, Section 23xx Your Name: Prof. Jason Kahn University of Maryland, College Park Your SID #: General Chemistry and Energetics Your Section #: Exam I (100 points total) October 6, 2010 You have
General Chemistry and Energetics Your Section #: Exam I (100 points total) October 6, 2010
 Chemistry 271, Section 22xx Your Name: Prof. Jason Kahn University of Maryland, College Park Your SID #: General Chemistry and Energetics Your Section #: Exam I (100 points total) October 6, 2010 You have
Chemistry 271, Section 22xx Your Name: Prof. Jason Kahn University of Maryland, College Park Your SID #: General Chemistry and Energetics Your Section #: Exam I (100 points total) October 6, 2010 You have
Prof. Jason Kahn University of Maryland, College Park Your SID #: General Chemistry and Energetics Final Exam (150 points total) December 19, 2016
 Chemistry 134 Your Name: Prof. Jason Kahn University of Maryland, College Park Your SID #: General Chemistry and Energetics Final Exam (150 points total) December 19, 2016 You have 120 minutes for this
Chemistry 134 Your Name: Prof. Jason Kahn University of Maryland, College Park Your SID #: General Chemistry and Energetics Final Exam (150 points total) December 19, 2016 You have 120 minutes for this
Chemical standard state: 1 M solutes, pure liquids, 1 atm gases Biochemical standard state: ph 7, all species in the ionic form found at ph 7
 Chemistry 271, Section 22xx Your Name: Prof. Jason Kahn University of Maryland, College Park Your SID #: General Chemistry and Energetics Exam II (100 points total) Your Section #: November 4, 2009 You
Chemistry 271, Section 22xx Your Name: Prof. Jason Kahn University of Maryland, College Park Your SID #: General Chemistry and Energetics Exam II (100 points total) Your Section #: November 4, 2009 You
Chemical standard state: 1 M solutes, pure liquids, 1 atm gases Biochemical standard state: ph 7, all species in the ionic form found at ph 7
 Chemistry 271, Section 22xx Your Name: Prof. Jason Kahn University of Maryland, College Park Your SID #: General Chemistry and Energetics Exam II (100 points total) Your Section #: November 4, 2009 You
Chemistry 271, Section 22xx Your Name: Prof. Jason Kahn University of Maryland, College Park Your SID #: General Chemistry and Energetics Exam II (100 points total) Your Section #: November 4, 2009 You
K a = [H + ][A ]/[HA] ph = log([h + ]) K b = [HA][HO ]/[A ]
![K a = [H + ][A ]/[HA] ph = log([h + ]) K b = [HA][HO ]/[A ] K a = [H + ][A ]/[HA] ph = log([h + ]) K b = [HA][HO ]/[A ]](/thumbs/93/114497464.jpg) Chemistry 271 23XX Prof. Jason Kahn Your Name: University of Maryland, College Park Your SID #: General Chemistry and Energetics Final Exam (200 points total) You have 120 minutes for this exam. Your Section
Chemistry 271 23XX Prof. Jason Kahn Your Name: University of Maryland, College Park Your SID #: General Chemistry and Energetics Final Exam (200 points total) You have 120 minutes for this exam. Your Section
Chapter 2 The Chemistry of Biology. Dr. Ramos BIO 370
 Chapter 2 The Chemistry of Biology Dr. Ramos BIO 370 2 Atoms, Bonds, and Molecules Matter - all materials that occupy space and have mass Matter is composed of atoms. Atom simplest form of matter not divisible
Chapter 2 The Chemistry of Biology Dr. Ramos BIO 370 2 Atoms, Bonds, and Molecules Matter - all materials that occupy space and have mass Matter is composed of atoms. Atom simplest form of matter not divisible
Practice Midterm Exam 200 points total 75 minutes Multiple Choice (3 pts each 30 pts total) Mark your answers in the space to the left:
 MITES ame Practice Midterm Exam 200 points total 75 minutes Multiple hoice (3 pts each 30 pts total) Mark your answers in the space to the left: 1. Amphipathic molecules have regions that are: a) polar
MITES ame Practice Midterm Exam 200 points total 75 minutes Multiple hoice (3 pts each 30 pts total) Mark your answers in the space to the left: 1. Amphipathic molecules have regions that are: a) polar
EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I Dr. Glen B Legge
 EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I 2009 Dr. Glen B Legge This is a Scantron exam. All answers should be transferred to the Scantron sheet using a #2 pencil. Write and bubble
EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I 2009 Dr. Glen B Legge This is a Scantron exam. All answers should be transferred to the Scantron sheet using a #2 pencil. Write and bubble
+3 for pka = 14 pkb = 9.25 [+1 for the idea that pka and pkb are related.]
![+3 for pka = 14 pkb = 9.25 [+1 for the idea that pka and pkb are related.] +3 for pka = 14 pkb = 9.25 [+1 for the idea that pka and pkb are related.]](/thumbs/82/86175751.jpg) Chemistry 271, Section 21xx Your Name: Key University of Maryland, College Park Your SID #: General Chemistry and Energetics Prof. Jason Kahn Exam I (100 points total) March 11, 2009 You have 50 minutes
Chemistry 271, Section 21xx Your Name: Key University of Maryland, College Park Your SID #: General Chemistry and Energetics Prof. Jason Kahn Exam I (100 points total) March 11, 2009 You have 50 minutes
Exam II (100 points total) November 5, 2012
 Chemistry 271, Section 22xx Your Name: Prof. Jason Kahn University of Maryland, College Park Your SID #: General Chemistry and Energetics Your Section #: (+1 point) Exam II (100 points total) November
Chemistry 271, Section 22xx Your Name: Prof. Jason Kahn University of Maryland, College Park Your SID #: General Chemistry and Energetics Your Section #: (+1 point) Exam II (100 points total) November
R = J/mole K = cal/mole K = N A k B K a K b = K w
 Chemistry 271 22XX Prof. Jason Kahn Your Name: University of Maryland, College Park Your SID #: General Chemistry and Energetics Exam II (100 points) You have 53 minutes for this exam. Your Section # or
Chemistry 271 22XX Prof. Jason Kahn Your Name: University of Maryland, College Park Your SID #: General Chemistry and Energetics Exam II (100 points) You have 53 minutes for this exam. Your Section # or
Homework - I. Hand draw the structure of an eukaryotic cell with all organelles clearly drawn and describe the function of each organelle.
 Homework - I Hand draw the structure of an eukaryotic cell with all organelles clearly drawn and describe the function of each organelle. Please get a book and read the chapter related to cell structure!!!
Homework - I Hand draw the structure of an eukaryotic cell with all organelles clearly drawn and describe the function of each organelle. Please get a book and read the chapter related to cell structure!!!
Energy and Cells. Appendix 1. The two primary energy transformations in plants are photosynthesis and respiration.
 Energy and Cells Appendix 1 Energy transformations play a key role in all physical and chemical processes that occur in plants. Energy by itself is insufficient to drive plant growth and development. Enzymes
Energy and Cells Appendix 1 Energy transformations play a key role in all physical and chemical processes that occur in plants. Energy by itself is insufficient to drive plant growth and development. Enzymes
Problem Set 1
 2006 7.012 Problem Set 1 Due before 5 PM on FRIDAY, September 15, 2006. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. For each of the following parts, pick
2006 7.012 Problem Set 1 Due before 5 PM on FRIDAY, September 15, 2006. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. For each of the following parts, pick
Biomolecules. Energetics in biology. Biomolecules inside the cell
 Biomolecules Energetics in biology Biomolecules inside the cell Energetics in biology The production of energy, its storage, and its use are central to the economy of the cell. Energy may be defined as
Biomolecules Energetics in biology Biomolecules inside the cell Energetics in biology The production of energy, its storage, and its use are central to the economy of the cell. Energy may be defined as
Prof. Jason D. Kahn Your Signature: Exams written in pencil or erasable ink will not be re-graded under any circumstances.
 Biochemistry 461, Section I May 6, 1997 Exam #3 Prof. Jason D. Kahn Your Printed Name: Your SS#: Your Signature: You have 80 minutes for this exam. Exams written in pencil or erasable ink will not be re-graded
Biochemistry 461, Section I May 6, 1997 Exam #3 Prof. Jason D. Kahn Your Printed Name: Your SS#: Your Signature: You have 80 minutes for this exam. Exams written in pencil or erasable ink will not be re-graded
Objective: Students will be able identify peptide bonds in proteins and describe the overall reaction between amino acids that create peptide bonds.
 Scott Seiple AP Biology Lesson Plan Lesson: Primary and Secondary Structure of Proteins Purpose:. To understand how amino acids can react to form peptides through peptide bonds.. Students will be able
Scott Seiple AP Biology Lesson Plan Lesson: Primary and Secondary Structure of Proteins Purpose:. To understand how amino acids can react to form peptides through peptide bonds.. Students will be able
LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor
 LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor Note: Adequate space is given for each answer. Questions that require a brief explanation should
LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor Note: Adequate space is given for each answer. Questions that require a brief explanation should
Exam I Answer Key: Summer 2006, Semester C
 1. Which of the following tripeptides would migrate most rapidly towards the negative electrode if electrophoresis is carried out at ph 3.0? a. gly-gly-gly b. glu-glu-asp c. lys-glu-lys d. val-asn-lys
1. Which of the following tripeptides would migrate most rapidly towards the negative electrode if electrophoresis is carried out at ph 3.0? a. gly-gly-gly b. glu-glu-asp c. lys-glu-lys d. val-asn-lys
Foundations in Microbiology Seventh Edition
 Lecture PowerPoint to accompany Foundations in Microbiology Seventh Edition Talaro Chapter 2 The Chemistry of Biology Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display.
Lecture PowerPoint to accompany Foundations in Microbiology Seventh Edition Talaro Chapter 2 The Chemistry of Biology Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display.
Biochemistry Quiz Review 1I. 1. Of the 20 standard amino acids, only is not optically active. The reason is that its side chain.
 Biochemistry Quiz Review 1I A general note: Short answer questions are just that, short. Writing a paragraph filled with every term you can remember from class won t improve your answer just answer clearly,
Biochemistry Quiz Review 1I A general note: Short answer questions are just that, short. Writing a paragraph filled with every term you can remember from class won t improve your answer just answer clearly,
CHEMISTRY OF THE HUMAN BODY
 CHEMISTRY OF THE HUMAN BODY (Sample Questions) WUCT 2018 The three questions below are meant to give a sense of the kinds of questions that might be asked on the exam in April 2018. The actual exam is
CHEMISTRY OF THE HUMAN BODY (Sample Questions) WUCT 2018 The three questions below are meant to give a sense of the kinds of questions that might be asked on the exam in April 2018. The actual exam is
Peptides And Proteins
 Kevin Burgess, May 3, 2017 1 Peptides And Proteins from chapter(s) in the recommended text A. Introduction B. omenclature And Conventions by amide bonds. on the left, right. 2 -terminal C-terminal triglycine
Kevin Burgess, May 3, 2017 1 Peptides And Proteins from chapter(s) in the recommended text A. Introduction B. omenclature And Conventions by amide bonds. on the left, right. 2 -terminal C-terminal triglycine
K a = [H + ][A ]/[HA] ph = log([h + ]) K b = [HA][HO ]/[A ]
![K a = [H + ][A ]/[HA] ph = log([h + ]) K b = [HA][HO ]/[A ] K a = [H + ][A ]/[HA] ph = log([h + ]) K b = [HA][HO ]/[A ]](/thumbs/93/114497428.jpg) Chemistry 271 23XX Prof. Jason Kahn Your Name: University of Maryland, College Park Your SID #: General Chemistry and Energetics Final Exam (200 points total) You have 120 minutes for this exam. Your Section
Chemistry 271 23XX Prof. Jason Kahn Your Name: University of Maryland, College Park Your SID #: General Chemistry and Energetics Final Exam (200 points total) You have 120 minutes for this exam. Your Section
MODULE 2: BIOLOGICAL MOLECULES
 PEER-LED TEAM LEARNING INTRDUCTRY BILGY MDULE 2: BILGICAL MLECULES JSEP GRISWLD, + DEAN STETLER,* AND MICAEL GAINES, ( + City College of New York, *University of Kansas, Univ. of Miami;) I. Introduction
PEER-LED TEAM LEARNING INTRDUCTRY BILGY MDULE 2: BILGICAL MLECULES JSEP GRISWLD, + DEAN STETLER,* AND MICAEL GAINES, ( + City College of New York, *University of Kansas, Univ. of Miami;) I. Introduction
CHEMISTRY OF THE HUMAN BODY
 CHEMISTRY OF THE HUMAN BODY (Sample Questions Key) WUCT 2018 The three questions below are meant to give a sense of the kinds of questions that might be asked on the exam in April 2018. The actual exam
CHEMISTRY OF THE HUMAN BODY (Sample Questions Key) WUCT 2018 The three questions below are meant to give a sense of the kinds of questions that might be asked on the exam in April 2018. The actual exam
Cell Energy Photosynthesis Study Guide
 Name Date Pd Circle the word or phrase that best completes the statement. Study all notes and read Ch 4.1-4.3. 1. All cells use adenosine triphosphate (ATP) for energy. ATP is a molecule / organelle that
Name Date Pd Circle the word or phrase that best completes the statement. Study all notes and read Ch 4.1-4.3. 1. All cells use adenosine triphosphate (ATP) for energy. ATP is a molecule / organelle that
Pathways that Harvest and Store Chemical Energy
 6 Pathways that Harvest and Store Chemical Energy Energy is stored in chemical bonds and can be released and transformed by metabolic pathways. Chemical energy available to do work is termed free energy
6 Pathways that Harvest and Store Chemical Energy Energy is stored in chemical bonds and can be released and transformed by metabolic pathways. Chemical energy available to do work is termed free energy
Discussion Section (Day, Time):
 Chemistry 27 Spring 2005 Exam 3 Chemistry 27 Professor Gavin MacBeath arvard University Spring 2005 our Exam 3 Friday April 29 th, 2005 11:07 AM 12:00 PM Discussion Section (Day, Time): TF: Directions:
Chemistry 27 Spring 2005 Exam 3 Chemistry 27 Professor Gavin MacBeath arvard University Spring 2005 our Exam 3 Friday April 29 th, 2005 11:07 AM 12:00 PM Discussion Section (Day, Time): TF: Directions:
NH 2. Biochemistry I, Fall Term Sept 9, Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter
 Biochemistry I, Fall Term Sept 9, 2005 Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter 3.1-3.4. Key Terms: ptical Activity, Chirality Peptide bond Condensation reaction ydrolysis
Biochemistry I, Fall Term Sept 9, 2005 Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter 3.1-3.4. Key Terms: ptical Activity, Chirality Peptide bond Condensation reaction ydrolysis
Chemistry 271, Section 21xx University of Maryland, College Park
 Chemistry 271, Section 21xx University of Maryland, College Park General Chemistry and Energetics Prof. Jason Kahn Final Exam (200 points total) May 15, 2009 You have 120 minutes for this exam. Exams written
Chemistry 271, Section 21xx University of Maryland, College Park General Chemistry and Energetics Prof. Jason Kahn Final Exam (200 points total) May 15, 2009 You have 120 minutes for this exam. Exams written
Discussion Section (Day, Time):
 Chemistry 27 pring 2005 Exam 3 Chemistry 27 Professor Gavin MacBeath arvard University pring 2005 our Exam 3 Friday April 29 th, 2005 11:07 AM 12:00 PM Discussion ection (Day, Time): TF: Directions: 1.
Chemistry 27 pring 2005 Exam 3 Chemistry 27 Professor Gavin MacBeath arvard University pring 2005 our Exam 3 Friday April 29 th, 2005 11:07 AM 12:00 PM Discussion ection (Day, Time): TF: Directions: 1.
Chemistry 271, Section 22xx University of Maryland, College Park. Final Exam (200 points total) May 14, 2010
 Chemistry 271, Section 22xx University of Maryland, College Park General Chemistry and Energetics Prof. Jason Kahn Final Exam (200 points total) May 14, 2010 You have 120 minutes for this exam. Exams written
Chemistry 271, Section 22xx University of Maryland, College Park General Chemistry and Energetics Prof. Jason Kahn Final Exam (200 points total) May 14, 2010 You have 120 minutes for this exam. Exams written
Biomolecules: lecture 9
 Biomolecules: lecture 9 - understanding further why amino acids are the building block for proteins - understanding the chemical properties amino acids bring to proteins - realizing that many proteins
Biomolecules: lecture 9 - understanding further why amino acids are the building block for proteins - understanding the chemical properties amino acids bring to proteins - realizing that many proteins
BA, BSc, and MSc Degree Examinations
 Examination Candidate Number: Desk Number: BA, BSc, and MSc Degree Examinations 2017-8 Department : BIOLOGY Title of Exam: Molecular Biology and Biochemistry Part I Time Allowed: 1 hour and 30 minutes
Examination Candidate Number: Desk Number: BA, BSc, and MSc Degree Examinations 2017-8 Department : BIOLOGY Title of Exam: Molecular Biology and Biochemistry Part I Time Allowed: 1 hour and 30 minutes
Section Week 3. Junaid Malek, M.D.
 Section Week 3 Junaid Malek, M.D. Biological Polymers DA 4 monomers (building blocks), limited structure (double-helix) RA 4 monomers, greater flexibility, multiple structures Proteins 20 Amino Acids,
Section Week 3 Junaid Malek, M.D. Biological Polymers DA 4 monomers (building blocks), limited structure (double-helix) RA 4 monomers, greater flexibility, multiple structures Proteins 20 Amino Acids,
Review Activity Module 1: Biological Chemistry
 Review Activity Module 1: Biological Chemistry Laroche: The picture above is of a molecule calle MC1R. Based on what you ve learned so far about the various biological macromolecules, what kind of macromolecule
Review Activity Module 1: Biological Chemistry Laroche: The picture above is of a molecule calle MC1R. Based on what you ve learned so far about the various biological macromolecules, what kind of macromolecule
Sample Questions for the Chemistry of Life Topic Test
 Sample Questions for the Chemistry of Life Topic Test 1. Enzymes play a crucial role in biology by serving as biological catalysts, increasing the rates of biochemical reactions by decreasing their activation
Sample Questions for the Chemistry of Life Topic Test 1. Enzymes play a crucial role in biology by serving as biological catalysts, increasing the rates of biochemical reactions by decreasing their activation
CHMI 2227 EL. Biochemistry I. Test January Prof : Eric R. Gauthier, Ph.D.
 CHMI 2227 EL Biochemistry I Test 1 26 January 2007 Prof : Eric R. Gauthier, Ph.D. Guidelines: 1) Duration: 55 min 2) 14 questions, on 7 pages. For 70 marks (5 marks per question). Worth 15 % of the final
CHMI 2227 EL Biochemistry I Test 1 26 January 2007 Prof : Eric R. Gauthier, Ph.D. Guidelines: 1) Duration: 55 min 2) 14 questions, on 7 pages. For 70 marks (5 marks per question). Worth 15 % of the final
2/25/2013. Electronic Configurations
 1 2 3 4 5 Chapter 2 Chemical Principles The Structure of Atoms Chemistry is the study of interactions between atoms and molecules The atom is the smallest unit of matter that enters into chemical reactions
1 2 3 4 5 Chapter 2 Chemical Principles The Structure of Atoms Chemistry is the study of interactions between atoms and molecules The atom is the smallest unit of matter that enters into chemical reactions
CHAPTER 29 HW: AMINO ACIDS + PROTEINS
 CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
W2. Chemical structures of protein and DNA
 W2. Chemical structures of protein and DNA Copyright Kang, Lin-Woo, Ph.D. Professor Department of Biological Sciences Konkuk University Seoul, Korea Lectures prepared by Christine L. Case The Structure
W2. Chemical structures of protein and DNA Copyright Kang, Lin-Woo, Ph.D. Professor Department of Biological Sciences Konkuk University Seoul, Korea Lectures prepared by Christine L. Case The Structure
BSc and MSc Degree Examinations
 Examination Candidate Number: Desk Number: BSc and MSc Degree Examinations 2018-9 Department : BIOLOGY Title of Exam: Molecular Biology and Biochemistry Part I Time Allowed: 1 hour and 30 minutes Marking
Examination Candidate Number: Desk Number: BSc and MSc Degree Examinations 2018-9 Department : BIOLOGY Title of Exam: Molecular Biology and Biochemistry Part I Time Allowed: 1 hour and 30 minutes Marking
Sample Question Solutions for the Chemistry of Life Topic Test
 Sample Question Solutions for the Chemistry of Life Topic Test 1. Enzymes play a crucial role in biology by serving as biological catalysts, increasing the rates of biochemical reactions by decreasing
Sample Question Solutions for the Chemistry of Life Topic Test 1. Enzymes play a crucial role in biology by serving as biological catalysts, increasing the rates of biochemical reactions by decreasing
Bio10 Cell and Molecular Lecture Notes SRJC
 Basic Chemistry Atoms Smallest particles that retain properties of an element Made up of subatomic particles: Protons (+) Electrons (-) Neutrons (no charge) Isotopes Atoms of an element with different
Basic Chemistry Atoms Smallest particles that retain properties of an element Made up of subatomic particles: Protons (+) Electrons (-) Neutrons (no charge) Isotopes Atoms of an element with different
BIOLOGY REVISION SHEET FINAL EXAM TERM-I GRADE 10 Session:
 BIOLOGY REVISION SHEET FINAL EXAM TERM-I GRADE 10 Session: 2018-19 Note: The students should first study from their textbook and then try to solve this revision sheet independently Materials included in
BIOLOGY REVISION SHEET FINAL EXAM TERM-I GRADE 10 Session: 2018-19 Note: The students should first study from their textbook and then try to solve this revision sheet independently Materials included in
Chapter 2. Chemical Principles
 Chapter 2 Chemical Principles Insert Fig CO 2 The Structure of Atoms Chemistry is the study of interactions between atoms and molecules The atom is the smallest unit of matter that enters into chemical
Chapter 2 Chemical Principles Insert Fig CO 2 The Structure of Atoms Chemistry is the study of interactions between atoms and molecules The atom is the smallest unit of matter that enters into chemical
P215 Basic Human Physiology Summer 2003 Lab Exam #1
 PLEASE BE AWARE CONTENT COVERED ON EXAMS VARIES FROM ONE SEMESTER TO ANOTHER. THIS EXAM MAY NOT CONTAIN MATERIAL THAT WILL BE ON YOUR EXAM THIS SEMESTER, AND/OR MAY CONTAIN MATERIAL THAT WILL NOT BE COVERED
PLEASE BE AWARE CONTENT COVERED ON EXAMS VARIES FROM ONE SEMESTER TO ANOTHER. THIS EXAM MAY NOT CONTAIN MATERIAL THAT WILL BE ON YOUR EXAM THIS SEMESTER, AND/OR MAY CONTAIN MATERIAL THAT WILL NOT BE COVERED
THE UNIVERSITY OF MANITOBA. PAPER NO: _1_ LOCATION: 173 Robert Schultz Theatre PAGE NO: 1 of 5 DEPARTMENT & COURSE NO: CHEM / MBIO 2770 TIME: 1 HOUR
 THE UNIVERSITY OF MANITOBA 1 November 1, 2016 Mid-Term EXAMINATION PAPER NO: _1_ LOCATION: 173 Robert Schultz Theatre PAGE NO: 1 of 5 DEPARTMENT & COURSE NO: CHEM / MBIO 2770 TIME: 1 HOUR EXAMINATION:
THE UNIVERSITY OF MANITOBA 1 November 1, 2016 Mid-Term EXAMINATION PAPER NO: _1_ LOCATION: 173 Robert Schultz Theatre PAGE NO: 1 of 5 DEPARTMENT & COURSE NO: CHEM / MBIO 2770 TIME: 1 HOUR EXAMINATION:
7.014 Quiz I Handout
 7.014 Quiz I andout Quiz I announcements: Quiz I: Friday, February 27 12:05 12:55 Walker Gym, rd floor (room 5040) **This will be a closed book exam** Quiz Review Session: Wednesday, February 25 7:00 9:00
7.014 Quiz I andout Quiz I announcements: Quiz I: Friday, February 27 12:05 12:55 Walker Gym, rd floor (room 5040) **This will be a closed book exam** Quiz Review Session: Wednesday, February 25 7:00 9:00
7.012 Problem Set 1. i) What are two main differences between prokaryotic cells and eukaryotic cells?
 ame 7.01 Problem Set 1 Section Question 1 a) What are the four major types of biological molecules discussed in lecture? Give one important function of each type of biological molecule in the cell? b)
ame 7.01 Problem Set 1 Section Question 1 a) What are the four major types of biological molecules discussed in lecture? Give one important function of each type of biological molecule in the cell? b)
Outline. Metabolism: Energy and Enzymes. Forms of Energy. Chapter 6
 Metabolism: Energy and Enzymes Chapter 6 Forms of Energy Outline Laws of Thermodynamics Metabolic Reactions ATP Metabolic Pathways Energy of Activation Enzymes Photosynthesis Cellular Respiration 1 2 Forms
Metabolism: Energy and Enzymes Chapter 6 Forms of Energy Outline Laws of Thermodynamics Metabolic Reactions ATP Metabolic Pathways Energy of Activation Enzymes Photosynthesis Cellular Respiration 1 2 Forms
Human Biology. The Chemistry of Living Things. Concepts and Current Issues. All Matter Consists of Elements Made of Atoms
 2 The Chemistry of Living Things PowerPoint Lecture Slide Presentation Robert J. Sullivan, Marist College Michael D. Johnson Human Biology Concepts and Current Issues THIRD EDITION Copyright 2006 Pearson
2 The Chemistry of Living Things PowerPoint Lecture Slide Presentation Robert J. Sullivan, Marist College Michael D. Johnson Human Biology Concepts and Current Issues THIRD EDITION Copyright 2006 Pearson
Full file at
 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following is an uncharged particle found in the nucleus of 1) an atom and which has
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following is an uncharged particle found in the nucleus of 1) an atom and which has
Final Exam Version 1. Chemistry 140C. Fall Good Luck! Dec 5, :30 am 2:30 pm This exam accounts for 50% of the final grade.
 Chemistry 140C Final Exam Version 1 Fall 2006 Dec 5, 2006 11:30 am 2:30 pm This exam accounts for 50% of the final grade. Mark your final answer clearly. Completely erase irrelevant information! Exams
Chemistry 140C Final Exam Version 1 Fall 2006 Dec 5, 2006 11:30 am 2:30 pm This exam accounts for 50% of the final grade. Mark your final answer clearly. Completely erase irrelevant information! Exams
First Problem Set, Due 30 Sep. (1st midterm is 7 Oct.) If you want it graded by 6 Oct, turn in on or before 30 Sep. at 5 pm.
 First Problem Set, Due 30 Sep. (1st midterm is 7 ct.) If you want it graded by 6 ct, turn in on or before 30 Sep. at 5 pm. Show your work calculations / steps!! Problems from Chapter 1 1 EM of a eukaryotic
First Problem Set, Due 30 Sep. (1st midterm is 7 ct.) If you want it graded by 6 ct, turn in on or before 30 Sep. at 5 pm. Show your work calculations / steps!! Problems from Chapter 1 1 EM of a eukaryotic
BIOCHEMISTRY. František Vácha. JKU, Linz.
 BIOCHEMISTRY František Vácha http://www.prf.jcu.cz/~vacha/ JKU, Linz Recommended reading: D.L. Nelson, M.M. Cox Lehninger Principles of Biochemistry D.J. Voet, J.G. Voet, C.W. Pratt Principles of Biochemistry
BIOCHEMISTRY František Vácha http://www.prf.jcu.cz/~vacha/ JKU, Linz Recommended reading: D.L. Nelson, M.M. Cox Lehninger Principles of Biochemistry D.J. Voet, J.G. Voet, C.W. Pratt Principles of Biochemistry
CHEM 3653 Exam # 1 (03/07/13)
 1. Using phylogeny all living organisms can be divided into the following domains: A. Bacteria, Eukarya, and Vertebrate B. Archaea and Eukarya C. Bacteria, Eukarya, and Archaea D. Eukarya and Bacteria
1. Using phylogeny all living organisms can be divided into the following domains: A. Bacteria, Eukarya, and Vertebrate B. Archaea and Eukarya C. Bacteria, Eukarya, and Archaea D. Eukarya and Bacteria
Proton Acidity. (b) For the following reaction, draw the arrowhead properly to indicate the position of the equilibrium: HA + K + B -
 Proton Acidity A01 Given that acid A has a pk a of 15 and acid B has a pk a of 10, then: (a) Which of the two acids is stronger? (b) For the following reaction, draw the arrowhead properly to indicate
Proton Acidity A01 Given that acid A has a pk a of 15 and acid B has a pk a of 10, then: (a) Which of the two acids is stronger? (b) For the following reaction, draw the arrowhead properly to indicate
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. Figure 2.1
 Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. Figure 2.1 1) Which compound in Figure 2.1 is an ester? 1) A) a b c d e Answer: D 2) A scientist
Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. Figure 2.1 1) Which compound in Figure 2.1 is an ester? 1) A) a b c d e Answer: D 2) A scientist
BI/CH421 Biochemistry I Exam 1 09/29/2014
 Part I: Multiple choice for each question, circle the choice that best answers the question. Do not write the letter in the margin to indicate your answer, circle it. 3 points each. 1. For a reaction with
Part I: Multiple choice for each question, circle the choice that best answers the question. Do not write the letter in the margin to indicate your answer, circle it. 3 points each. 1. For a reaction with
Biochemistry Prof. S. DasGupta Department of Chemistry Indian Institute of Technology Kharagpur. Lecture - 06 Protein Structure IV
 Biochemistry Prof. S. DasGupta Department of Chemistry Indian Institute of Technology Kharagpur Lecture - 06 Protein Structure IV We complete our discussion on Protein Structures today. And just to recap
Biochemistry Prof. S. DasGupta Department of Chemistry Indian Institute of Technology Kharagpur Lecture - 06 Protein Structure IV We complete our discussion on Protein Structures today. And just to recap
UNIT TWELVE. a, I _,o "' I I I. I I.P. l'o. H-c-c. I ~o I ~ I / H HI oh H...- I II I II 'oh. HO\HO~ I "-oh
 UNT TWELVE PROTENS : PEPTDE BONDNG AND POLYPEPTDES 12 CONCEPTS Many proteins are important in biological structure-for example, the keratin of hair, collagen of skin and leather, and fibroin of silk. Other
UNT TWELVE PROTENS : PEPTDE BONDNG AND POLYPEPTDES 12 CONCEPTS Many proteins are important in biological structure-for example, the keratin of hair, collagen of skin and leather, and fibroin of silk. Other
Lecture 20. Chemical Potential
 Lecture 20 Chemical Potential Reading: Lecture 20, today: Chapter 10, sections A and B Lecture 21, Wednesday: Chapter 10: 10 17 end 3/21/16 1 Pop Question 7 Boltzmann Distribution Two systems with lowest
Lecture 20 Chemical Potential Reading: Lecture 20, today: Chapter 10, sections A and B Lecture 21, Wednesday: Chapter 10: 10 17 end 3/21/16 1 Pop Question 7 Boltzmann Distribution Two systems with lowest
Midterm Exam III. CHEM 181: Introduction to Chemical Principles November 25, 2014 Answer Key
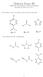 Midterm Exam III EM 181: Introduction to hemical Principles ovember 25, 2014 Answer Key 1. For reference, here are the pk a values for three weak acids: 3 pk a = 7.2 pk a = 4.8 pk a = 15 ow consider this
Midterm Exam III EM 181: Introduction to hemical Principles ovember 25, 2014 Answer Key 1. For reference, here are the pk a values for three weak acids: 3 pk a = 7.2 pk a = 4.8 pk a = 15 ow consider this
Chemistry 3351: Organic Chemistry Thursday: Sept. 7:00pm 9:00/1 st Exam. Name: (please print)
 Chemistry 3351: Organic Chemistry Thursday: Sept. 23 @ 7:00pm 9:00/1 st Exam 1 Name: (please print) Page Possible Points Score 2 9 3 9 4 8 5 10 6 10 7 4 8 9 9 10 10 10 11 10 12 10 13 10 TOTAL 109 2 1.
Chemistry 3351: Organic Chemistry Thursday: Sept. 23 @ 7:00pm 9:00/1 st Exam 1 Name: (please print) Page Possible Points Score 2 9 3 9 4 8 5 10 6 10 7 4 8 9 9 10 10 10 11 10 12 10 13 10 TOTAL 109 2 1.
K w. Acids and bases 8/24/2009. Acids and Bases 9 / 03 / Ionization of water. Proton Jumping Large proton and hydroxide mobility
 Chapter 2 Water Acids and Bases 9 / 03 / 2009 1. How is the molecular structure of water related to physical and chemical behavior? 2. What is a Hydrogen Bond? 3Wh 3. What are Acids Aid and db Bases? 4.
Chapter 2 Water Acids and Bases 9 / 03 / 2009 1. How is the molecular structure of water related to physical and chemical behavior? 2. What is a Hydrogen Bond? 3Wh 3. What are Acids Aid and db Bases? 4.
Free Energy. because H is negative doesn't mean that G will be negative and just because S is positive doesn't mean that G will be negative.
 Biochemistry 462a Bioenergetics Reading - Lehninger Principles, Chapter 14, pp. 485-512 Practice problems - Chapter 14: 2-8, 10, 12, 13; Physical Chemistry extra problems, free energy problems Free Energy
Biochemistry 462a Bioenergetics Reading - Lehninger Principles, Chapter 14, pp. 485-512 Practice problems - Chapter 14: 2-8, 10, 12, 13; Physical Chemistry extra problems, free energy problems Free Energy
AMINO ACIDS ARE JOINED TOGETHER BY BONDS. FILE
 04 February, 2018 AMINO ACIDS ARE JOINED TOGETHER BY BONDS. FILE Document Filetype: PDF 401.79 KB 0 AMINO ACIDS ARE JOINED TOGETHER BY BONDS. FILE Understanding the chemistry and properties of the peptide
04 February, 2018 AMINO ACIDS ARE JOINED TOGETHER BY BONDS. FILE Document Filetype: PDF 401.79 KB 0 AMINO ACIDS ARE JOINED TOGETHER BY BONDS. FILE Understanding the chemistry and properties of the peptide
Chemistry of Life. Chapters 2 & 3. Credit: Larry Stepanowicz. Learning Objectives
 Chemistry of Life Chapters 2 & 3 Credit: Larry Stepanowicz Learning Objectives 1. Differentiate between the definitions of an atom, element, ion, and molecule. 2. Describe why and how atoms react chemically.
Chemistry of Life Chapters 2 & 3 Credit: Larry Stepanowicz Learning Objectives 1. Differentiate between the definitions of an atom, element, ion, and molecule. 2. Describe why and how atoms react chemically.
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1)
 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) 1) Which of the following statements about the atom A) It has 12 neutrons in its nucleus. B) It
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) 1) Which of the following statements about the atom A) It has 12 neutrons in its nucleus. B) It
Chemistry. Atomic and Molecular Structure
 Chemistry Atomic and Molecular Structure 1. The periodic table displays the elements in increasing atomic number and shows how periodicity of the physical and chemical properties of the elements relates
Chemistry Atomic and Molecular Structure 1. The periodic table displays the elements in increasing atomic number and shows how periodicity of the physical and chemical properties of the elements relates
Discussion Section (Day, Time): TF:
 ame: Chemistry 27 Professor Gavin MacBeath arvard University Spring 2004 Final Exam Thursday, May 28, 2004 2:15 PM - 5:15 PM Discussion Section (Day, Time): Directions: TF: 1. Do not write in red ink.
ame: Chemistry 27 Professor Gavin MacBeath arvard University Spring 2004 Final Exam Thursday, May 28, 2004 2:15 PM - 5:15 PM Discussion Section (Day, Time): Directions: TF: 1. Do not write in red ink.
CHEM J-2 June 2006 HCO 2. Calculate the osmotic pressure of a solution of 1.0 g of glucose (C 6 H 12 O 6 ) in 1500 ml of water at 37 C.
 CEM1405 2006-J-2 June 2006 Draw Lewis structures of ozone, 3, and the formate anion, C 2, including resonance hybrids where appropriate. 3 C 2 3 C C Calculate the osmotic pressure of a solution of 1.0
CEM1405 2006-J-2 June 2006 Draw Lewis structures of ozone, 3, and the formate anion, C 2, including resonance hybrids where appropriate. 3 C 2 3 C C Calculate the osmotic pressure of a solution of 1.0
MITOCW watch?v=vykadbjib8a
 MITOCW watch?v=vykadbjib8a The following content is provided under a Creative Commons license. Your support will help MIT OpenCourseWare continue to offer high-quality, educational resources for free.
MITOCW watch?v=vykadbjib8a The following content is provided under a Creative Commons license. Your support will help MIT OpenCourseWare continue to offer high-quality, educational resources for free.
Chemistry State Content Standards EXAM. from human beings! Explanations and Examples MUST be in Complete Sentences!
 Chemistry State Content Standards EXAM You may use your Notes, PowerPoint, or Text on this exam but NO help from human beings! You MUST HAND WRITE THESE EXAMS in INK!! NO TYPED or PENCIL PAPERS WILL BE
Chemistry State Content Standards EXAM You may use your Notes, PowerPoint, or Text on this exam but NO help from human beings! You MUST HAND WRITE THESE EXAMS in INK!! NO TYPED or PENCIL PAPERS WILL BE
Ch. 2 BASIC CHEMISTRY. Copyright 2010 Pearson Education, Inc.
 Ch. 2 BASIC CHEMISTRY Matter and Composition of Matter Definition: Anything that has mass and occupies space Matter is made up of elements An element cannot be broken down by ordinary chemical means Atoms
Ch. 2 BASIC CHEMISTRY Matter and Composition of Matter Definition: Anything that has mass and occupies space Matter is made up of elements An element cannot be broken down by ordinary chemical means Atoms
Human Anatomy & Physiology. Chapter 2: Chemistry Comes Alive. Copyright 2010 Pearson Education, Inc.
 Human Anatomy & Physiology Chapter 2: Chemistry Comes Alive MATTER VS. ENERGY Which of the following is not an example of matter? 1) Blood plasma 2) The air we breathe 3) An arm bone 4) Electricity Which
Human Anatomy & Physiology Chapter 2: Chemistry Comes Alive MATTER VS. ENERGY Which of the following is not an example of matter? 1) Blood plasma 2) The air we breathe 3) An arm bone 4) Electricity Which
Unit 2: Basic Chemistry
 Unit 2: Basic Chemistry I. Matter and Energy A. Matter anything that occupies space and has mass (weight) B. Energy the ability to do work 1. Chemical 2. Electrical 3. Mechanical 4. Radiant C. Composition
Unit 2: Basic Chemistry I. Matter and Energy A. Matter anything that occupies space and has mass (weight) B. Energy the ability to do work 1. Chemical 2. Electrical 3. Mechanical 4. Radiant C. Composition
Thermodynamics is the study of energy and its effects on matter
 00Note Set 3 1 THE ENERGETICS OF LIFE Thermodynamics and Bioenergetics: Thermodynamics is the study of energy and its effects on matter Bioenergetics is the quantitative analysis of how organisms gain
00Note Set 3 1 THE ENERGETICS OF LIFE Thermodynamics and Bioenergetics: Thermodynamics is the study of energy and its effects on matter Bioenergetics is the quantitative analysis of how organisms gain
Chapter Two: The Chemistry of Biology. The molecules of life make up the structure of cells Chemistry of biological molecule
 Chapter Two: The Chemistry of Biology The molecules of life make up the structure of cells Chemistry of biological molecule Atoms and Elements: Atoms: The basic units of all matter, containing three major
Chapter Two: The Chemistry of Biology The molecules of life make up the structure of cells Chemistry of biological molecule Atoms and Elements: Atoms: The basic units of all matter, containing three major
BIOCHEMISTRY GUIDED NOTES - AP BIOLOGY-
 BIOCHEMISTRY GUIDED NOTES - AP BIOLOGY- ELEMENTS AND COMPOUNDS - anything that has mass and takes up space. - cannot be broken down to other substances. - substance containing two or more different elements
BIOCHEMISTRY GUIDED NOTES - AP BIOLOGY- ELEMENTS AND COMPOUNDS - anything that has mass and takes up space. - cannot be broken down to other substances. - substance containing two or more different elements
LS1a Midterm Exam 1 Review Session Problems
 LS1a Midterm Exam 1 Review Session Problems 1. n aqueous mixture of a weak acid and its conjugate base is often used in the laboratory to prepare solutions referred to as buffers. ne commonly used acid
LS1a Midterm Exam 1 Review Session Problems 1. n aqueous mixture of a weak acid and its conjugate base is often used in the laboratory to prepare solutions referred to as buffers. ne commonly used acid
2) Matter composed of a single type of atom is known as a(n) 2) A) element. B) mineral. C) electron. D) compound. E) molecule.
 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following is a particle found in the nucleus of an atom and that has no electrical
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following is a particle found in the nucleus of an atom and that has no electrical
Advanced Certificate in Principles in Protein Structure. You will be given a start time with your exam instructions
 BIRKBECK COLLEGE (University of London) Advanced Certificate in Principles in Protein Structure MSc Structural Molecular Biology Date: Thursday, 1st September 2011 Time: 3 hours You will be given a start
BIRKBECK COLLEGE (University of London) Advanced Certificate in Principles in Protein Structure MSc Structural Molecular Biology Date: Thursday, 1st September 2011 Time: 3 hours You will be given a start
Metabolism. AP Biology Chapter 8
 Metabolism AP Biology Chapter 8 Energy Energy management Bioenergetics is the study of how organisms manage their energy resources. Energy is the capacity to do work. Energy exists in various forms Cells
Metabolism AP Biology Chapter 8 Energy Energy management Bioenergetics is the study of how organisms manage their energy resources. Energy is the capacity to do work. Energy exists in various forms Cells
Secondary Structure. Bioch/BIMS 503 Lecture 2. Structure and Function of Proteins. Further Reading. Φ, Ψ angles alone determine protein structure
 Bioch/BIMS 503 Lecture 2 Structure and Function of Proteins August 28, 2008 Robert Nakamoto rkn3c@virginia.edu 2-0279 Secondary Structure Φ Ψ angles determine protein structure Φ Ψ angles are restricted
Bioch/BIMS 503 Lecture 2 Structure and Function of Proteins August 28, 2008 Robert Nakamoto rkn3c@virginia.edu 2-0279 Secondary Structure Φ Ψ angles determine protein structure Φ Ψ angles are restricted
BIRKBECK COLLEGE (University of London)
 BIRKBECK COLLEGE (University of London) SCHOOL OF BIOLOGICAL SCIENCES M.Sc. EXAMINATION FOR INTERNAL STUDENTS ON: Postgraduate Certificate in Principles of Protein Structure MSc Structural Molecular Biology
BIRKBECK COLLEGE (University of London) SCHOOL OF BIOLOGICAL SCIENCES M.Sc. EXAMINATION FOR INTERNAL STUDENTS ON: Postgraduate Certificate in Principles of Protein Structure MSc Structural Molecular Biology
EVPP 110 Lecture Exam #1 Study Questions Fall 2003 Dr. Largen
 EVPP 110 Lecture Exam #1 Study Questions Fall 2003 Dr. Largen These study questions are meant to focus your study of the material for the first exam. The absence here of a topic or point covered in lecture
EVPP 110 Lecture Exam #1 Study Questions Fall 2003 Dr. Largen These study questions are meant to focus your study of the material for the first exam. The absence here of a topic or point covered in lecture
Teacher Notes for How do biological organisms use energy? 1
 Teacher Notes for How do biological organisms use energy? 1 This analysis and discussion activity introduces students to the basic principles of how biological organisms use energy. The focus is on understanding
Teacher Notes for How do biological organisms use energy? 1 This analysis and discussion activity introduces students to the basic principles of how biological organisms use energy. The focus is on understanding
Properties of amino acids in proteins
 Properties of amino acids in proteins one of the primary roles of DNA (but not the only one!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids repeated
Properties of amino acids in proteins one of the primary roles of DNA (but not the only one!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids repeated
Chapter 1. Topic: Overview of basic principles
 Chapter 1 Topic: Overview of basic principles Four major themes of biochemistry I. What are living organism made from? II. How do organism acquire and use energy? III. How does an organism maintain its
Chapter 1 Topic: Overview of basic principles Four major themes of biochemistry I. What are living organism made from? II. How do organism acquire and use energy? III. How does an organism maintain its
Principles Of Acid-Base Balance
 Principles Of Acid-Base Balance I. Introduction A. For normal body function the free H+ concentration [H+] or ph must be kept within a narrow normal range. Some reasons why: 1. The proton "pump" within
Principles Of Acid-Base Balance I. Introduction A. For normal body function the free H+ concentration [H+] or ph must be kept within a narrow normal range. Some reasons why: 1. The proton "pump" within
Chapter 2. The Structure of Atoms. The Structure of Atoms. The Structure of Atoms
 1 The Structure of Atoms 2 Chapter 2 Chemical Principles Chemistry is the study of interactions between atoms and molecules The atom is the smallest unit of matter that enters into chemical reactions Atoms
1 The Structure of Atoms 2 Chapter 2 Chemical Principles Chemistry is the study of interactions between atoms and molecules The atom is the smallest unit of matter that enters into chemical reactions Atoms
BCH Graduate Survey of Biochemistry
 BCH 5045 Graduate Survey of Biochemistry Instructor: Charles Guy Producer: Ron Thomas Director: Marsha Derosier Lecture 6 Slide sets available at: http://hort.ifas.ufl.edu/teach/guyweb/bch5045/index.html
BCH 5045 Graduate Survey of Biochemistry Instructor: Charles Guy Producer: Ron Thomas Director: Marsha Derosier Lecture 6 Slide sets available at: http://hort.ifas.ufl.edu/teach/guyweb/bch5045/index.html
