Production of α,ω diols from Biomass
|
|
|
- Julius Rose
- 6 years ago
- Views:
Transcription
1 Production of α,ω diols from Biomass Thermal and Catalytic Sciences 216 November 3, 216 Jiayue He, Kefeng Huang, Pranav Karanjkar, Kevin J Barnett, Zachary Brentzal, Theodore Walker, Siddarth H Krishna, Sam Burt, Ive Hermans, Christos Maravelias, James A Dumesic, George W. Huber University of Wisconsin-Madison Department of Chemical & Biological Engineering 1
2 Catalytic Processes for Production of α,ω-diols from Lignocellulosic Biomass Support from Bioenergy Technology Office (BETO) Chris Marshall Michael Tspastsis, Ilja Siepmann
3 Price ($/MT) Price ($/MMBTU) High value infrastructure compatible commodity chemicals from biomass 1,6-Hexanediol (~13, MT/yr) ,6-hexanediol ($4,4/ton) 3 2 Gasoline ($2.5/gal) Volume (Million MT/yr.) 1 Source: ICIS, IEA Crude oil ($5/bbl) Biomass ($8/ton) Natural gas Source: Lux Research, Bio-based Materials and Chemical Intelligence Service, 3
4 There are several (aqueous-phase) acid catalyzed reactions in biomass conversion Hydrolysis Isomerization 1 Dehydration 2 Rehydration 1) Y. Roman-Leshkov, M. Moliner, J.A. Labinger, and M.E. Davis, Angewandte Chemie International Edition 49 (21) ) Y. Roman-Leshkov, J.N. Chheda, and J.A. Dumesic, Science 312 (26)
5 Low product selectivity is a key challenge in biomass conversion 5
6 Proposed reaction scheme 6
7 2 Activity of Acid sites are influenced by Solvent due to Solvation Effects Based on total detectable products Based on HMF Turnover frequency / hr T=17 C P=1 psig Cellulose=5wt% Acid= 5 mm H 2 SO 4 Solvent = THF Initial water content in solvent / vol % R. Weingarten, A Rodriguez-Beuerman, F Cai, JS Luterbacher, DM Alonso, JA Dumesic, GW Huber, Selective Conversion of Cellulose to Hydroxymethylfurfural in Polar Aprotic Solvents; ChemCatChem; (214)
8 HMF carbon yield (%) High HMF yields are obtained in Water THF GVL Ethyl Acetate Acetone Ethanol polar aprotic solvents Polar aprotic solvents T=17 C P=1 psig Cellulose=3 gr Acid=5 mm H 2 SO 4 Reaction vol.=6 ml time (min.) R. Weingarten, A Rodriguez-Beuerman, F Cai, JS Luterbacher, DM Alonso, JA Dumesic, GW Huber, Selective Conversion of Cellulose to Hydroxymethylfurfural in Polar Aprotic Solvents; ChemCatChem; (214) M A Mellmer, C Sener, JMR Gallo, JS Luterbachher, DM Alonso, JA Dumesic, Solvent Effects in Acid Catalyzed Biomass Conversion Reactions,, Ang Chemie (214)
9 Presence of water changes the product distribution SOLVENT: 11.6% water, bal. THF 2.7% water, bal. THF Pure THF 4 2 Yield (% Carbon) LGA Yield (% Carbon) Glucose Time (min) Time (min) Yield (% Carbon) LGO Yield (% Carbon) HMF Time (min) Time (min) Reaction Conditions: 1 wt% cellulose, 7.5 mm H 2 SO 4, 19 C 1 psig He, 6 ml total volume Source: Cao, F.; Schwartz, T. J.; McClelland, D. J.; Krishna, S. H.; Dumesic, J. A.; Huber, G. W., Dehydration of cellulose to levoglucosenone using polar aprotic solvents Energy & Environmental Science 215, 8 (6),
10 Levoglucosenone (LGO) Chiral carbon Sometimes referred to as the next HMF F Cao, TJ Schwartz, D McClelland, S Krishna, JA Dumesic, GW Huber, Dehydration of Cellulose to Levoglucosenone using Polar Aprotic Solvents, EES, (215)
11 Levoglucosenone (LGO) Chiral carbon Double bond conjugated with a ketone Sometimes referred to as the next HMF F Cao, TJ Schwartz, D McClelland, S Krishna, JA Dumesic, GW Huber, Dehydration of Cellulose to Levoglucosenone using Polar Aprotic Solvents, EES, (215)
12 Levoglucosenone (LGO) Chiral carbon Double bond conjugated with a ketone Protected aldehyde Sometimes referred to as the next HMF F Cao, TJ Schwartz, D McClelland, S Krishna, JA Dumesic, GW Huber, Dehydration of Cellulose to Levoglucosenone using Polar Aprotic Solvents, EES, (215)
13 Levoglucosenone (LGO) Chiral carbon Double bond conjugated with a ketone Protected aldehyde Two protected hydroxyl groups Sometimes referred to as the next HMF F Cao, TJ Schwartz, D McClelland, S Krishna, JA Dumesic, GW Huber, Dehydration of Cellulose to Levoglucosenone using Polar Aprotic Solvents, EES, (215)
14 Reaction Pathway for Dehydration of Cellulose in Polar Aprotic Solvents 14
15 LGO is hydrogenated to levoglucosanol over a metal catalyst (Pd/Al 2 O 3 ) High yields of Cyrene or Lgol achievable using monometallic catalysts Excess of exo-lgol produced over endo-lgol S.H. Krishna, D.J. McClelland, Q.R. Rashke, J.A. Dumesic, G.W. Huber, Catalytic hydrogenation of levoglucosenone to value-added chemicals. Submitted.
16 Selective Hydrogenolysis of Cyclic Ethers: Literature Bifunctional catalyst with a reducible metal and an oxophilic promoter; Low temperatures Reactant Transition state Product Proposed reaction pathway in literature with a RhRe/C catalyst through formation of a stable oxocarbenium ion [1] Tetrahydrofurfuryl alcohol Hydride transfer creates a stable oxocarbenium ion 97% selectivity to α,ω-diols Proposed reaction scheme for THP-2M hydrogenolysis with a IrRe/SiO 2 catalyst [2] THP-2M 1,6-HDO (>95% selectivity) 1,2-HDO 1-hexanol 2-hexanol 2-methyltetrahydropyran (2-MTHP) C-C cracking products Sources: [1] Chia et al., Journal of the American Chemical Society, 211, 133, [2] Chen et al., ChemCatChem, 21, 2,
17 Synergy between the reducible metal and oxophilic promoter is important Batch reactor activity data by Chia et al. [1] : THP-2M 1,6-HDO Catalyst Conversion (%) Rate (μmol g -1 min -1 ) 1,6-HDO selectivity (%) Byproducts selectivity (%) Rh/VXC (4 wt% Rh) ,2-hexanediol (11%), 1-hexanol (3%), others (42%) Re/VXC (3.6 wt% Re) RhRe/VXC (4 wt% Rh, 3.6 wt% Re) No reaction hexanols (3%), VXC = Vulcan XC-72, a carbon black by Cabot Corporation other products include over-hydrogenolysis and C-C cracking products Additional reaction conditions and details: Feedstock: 5% aq. THP-2M feedstock, Catalyst:Feedstock (g/g) = 2:7, Temperature = 12 o C, Pressure = 34 bar H 2, Time = 4 h Source: [1] Chia et al., Journal of the American Chemical Society, 211, 133,
18 High selectivity to 1,6-HDO is achievable using RhRe/VXC bifunctional catalysts separator THP-2M conversion (%) 1,6-HDO selectivity (%) To back-pressure regulator Reactor Gas-Liquid Hydrogen THP-2M solution through HPLC pump Time (h) Byproducts include small amounts of 1-hexanol, 2-MTHP, 1-pentanol Reaction conditions for continuous reaction: 5% aq. THP-2M, catalyst: RhRe/VXC, WHSV = 1.44 h -1, 12 o C, 34 bar H 2, 4 sccm H 2 Karanjkar et al.; Effect of carbon support on RhRe bifunctional catalysts for selective hydrogenolysis of tetrahydropyran-2- methanol RSC Catalysis Science and Technology, in press. 18
19 High selectivity to 1,6-HDO is achievable using RhRe/VXC bifunctional catalysts THP-2M conversion (%) 1,6-HDO selectivity (%) RhRe/VXC RhRe/NDC Time (h) 2 Byproducts include small amounts of 1-hexanol, 2-MTHP, 1-pentanol with RhRe/VXC catalyst Reaction conditions: 5% aq. THP-2M (initial reaction volume = 5 ml), catalyst: RhRe/VXC, catalyst:feedstock (g/g) = 1:7, 12 o C, 34 bar H 2. Karanjkar et al.; Effect of carbon support on RhRe bifunctional catalysts for selective hydrogenolysis of tetrahydropyran-2- methanol RSC Catalysis Science and Technology, in press. 19
20 Carbon Blacks vs. Activated Carbons Formation of carbon blacks (vapor phase) Hydrocarbons Gas-phase pyrolysis Formation of activated carbons (solid phase) Low-rank coals, wood, thermosetting polymers Thermal decomposition by charring Carbon black Glassy carbon Activation Activated carbons (chars) 1. Selective gasification 2. Chemical treatment 3. Carbon supports discussed here: Vulcan XC-72, Cabot Corp., carbon black VXC Source: L. R. Radovic, in Carbon Materials for Catalysis, John Wiley & Sons, Inc., 28, pp & Norit Darco 12X4, Cabot Corp., activated carbon NDC 2
21 Quantity adsorbed (cm 3 g -1 ) Pore volume (cm 3 g -1 Å -1 ) Characterization of Carbon Supports Carbon support NDC VXC Type of carbon support Activated (acid washed) Carbon black BET surface area (m 2 g -1 ) Quantity adsorbed (cm 3 g -1 STP) Nitrogen adsorption isotherm Adsorption Desorption NDC VXC Relative pressure (P/P o ) o ) Pore volume (cm 3 g -1 Å -1 ) VXC NDC Pore size distribution Pore width (Å) Karanjkar et al.; Effect of carbon support on RhRe bifunctional catalysts for selective hydrogenolysis of tetrahydropyran-2- methanol RSC Catalysis Science and Technology, in press. 21
22 Quantity adsorbed (mmol g -1 ) Quantity adsorbed (mmol g -1 ) NDC has more surface oxygen present XPS analysis of carbon supports Surface elemental composition (mol %) Carbon C O S Si Na Al VXC NDC Adsorption Desorption NDC 4 3 Water vapor adsorption isotherm Water adsorption capacity of VXC = 1.2 mmol g Water adsorption capacity of NDC = 17.5 mmol g VXC Relative pressure (P/P o ) Karanjkar et al.; Effect of carbon support on RhRe bifunctional catalysts for selective hydrogenolysis of tetrahydropyran-2- methanol RSC Catalysis Science and Technology, in press. 22
23 Signal (a.u.) Higher density of oxidized species on NDC than VXC more sites for interaction with metal and metal precursors Support characterization with DRIFTS NDC Band Position (cm -1 ) Assignment C-CO deformation of cyclic ketone C-O-C stretch of aromatic cyclic anhydride or O-H bend of carboxylic acid Acid Anhydride C-O-C stretch Relative abundance NDC VXC Phenol O-H bend Ether C-O-C stretch Phenol C-OH stretch.8.42 VXC 1215 Lactone C=O stretch Carb. Acid C=O stretch Carboxyl Carbonate C=O stretch Wavenumber (cm -1 ) C=C stretch (conjugated aromatic) O-H stretch of water or Sources: [1] Figueiredo et.al., Carbon, 1999, 37, carboxylic acid [2] Fanning et al., Carbon, 1993, 31, Abundance shown is relative to C=C stretch (the only signal due to [3] Collins et al., Carbon, 213, 57, a non-oxygenated bond) 23 Karanjkar et al.; Effect of carbon support on RhRe bifunctional catalysts for selective hydrogenolysis of tetrahydropyran-2-
24 Characterization of Bimetallic Catalysts Catalyst Rh/NDC RhRe/NDC Rh/VXC RhRe/VXC Rh loading (wt%) Rh:Re atomic ratio - 1:.5-1:.5 Rh atom density (atoms/nm 2 ) CO uptake (μmol/g) CO:Rh (mol:mol) Rate of THP-2M [ ] hydrogenolysis (μmol/g/min) 1 76 Reaction conditions for batch reactions: 12 o C, 34 bar H 2, 5% aq. THP-2M (Initial reaction volume = 25 ml), catalyst: RhRe/C, catalyst:feedstock (g/g) = 1:7, Reaction time = 4 h Karanjkar et al.; Effect of carbon support on RhRe bifunctional catalysts for selective hydrogenolysis of tetrahydropyran-2- methanol RSC Catalysis Science and Technology, in press. 24
25 Normalized absorption Normalized absorption X-ray Absorption Near Edge Structure Re catalysts RhRe catalysts Re 2 O 7 ReO 2 Re/NDC reduced at 2 o C Re/VXC reduced at 2 o C 3 ReO 2 Re 2 O 7 RhRe/NDC reduced at 2 o C RhRe/VXC reduced at 2 o C Re foil 1 Re foil Energy (kev) Energy (kev) Catalyst Treatment prior to scan in He XANES fit for Re edge Re(VII) Re(IV) Re() Catalyst Treatment prior to scan in He XANES fit for Re edge Re(VII) Re(IV) Re() Re/NDC 2 C reduction RhRe/NDC 2 C reduction Re/VXC 2 C reduction RhRe/VXC 2 C reduction Karanjkar et al.; Effect of carbon support on RhRe bifunctional catalysts for selective hydrogenolysis of tetrahydropyran-2- methanol RSC Catalysis Science and Technology, in press. 25
26 Fraction Fraction Scanning Transmission Electron Microscopy (STEM) 1..8 Average = 1.61±.61 nm 868 particles analyzed RhRe/NDC Particle size (nm).5.4 Average = 1.98±.73 nm 793 particles analyzed RhRe/VXC Particle size (nm) Karanjkar et al.; Effect of carbon support on RhRe bifunctional catalysts for selective hydrogenolysis of tetrahydropyran-2- methanol RSC Catalysis Science and Technology, in press. 26
27 STEM-EDS images for catalyst nanoparticles Rh Map RhRe/NDC Avg. particle size : 1.61±.61 nm Drift ref. Re Rh RhRe/VXC Avg. particle size : 1.98±.73 nm Map Re Drift ref. Karanjkar et al.; Effect of carbon support on RhRe bifunctional catalysts for selective hydrogenolysis of tetrahydropyran-2- methanol RSC Catalysis Science and Technology, in press. 27
28 Number of particles Number of particles STEM-EDS indicates segregation of Re species in NDC atomic% Re Re content (atomic %) RhRe/NDC Re content (% atomic) Rh:Re (mol:mol) Theoretical ICP EDS 33.3% 35.5% 61.3% 1:.5 1:.55 1: atomic% Re RhRe/VXC Theoretical ICP EDS Re content (atomic %) Re content (% atomic) Rh:Re (mol:mol) Note: The distribution considers composition of ~6 particles analyzed with EDS 33.3% 33.8% 36.7% 1:.5 1:.51 1:.58 28
29 Tiny Rh particles (<1 nm) Note: Schematic is for visualization purposes only! 1% Re 65% Re ~33% Re RhRe/NDC RhRe/VXC Nanoparticles with segrated particle composition Many small particles (<1 nm) rich in Rh are present that evade detection in STEM Many Re only particles also present and larger particles are rich in Re Higher surface oxygen content may restrict mobility of metals/metalprecursors resulting in segregation of Rh and Re Nanoparticles with relatively uniform composition Anchoring of metals/metal-precursors is relatively absent because of low surface oxygen Formation of small Rh particles (<1 nm) may be avoided because of smaller surface area of support 29
30 Conclusion New pathways for production of high value oxygenated commodity chemicals from biomass Cellulose can be converted into LGO and HMF in high yields in polar aprotic solvents LGO is a promising platform molecule LGO and HMF hydrogenated into THFDM which then undergoes hydrogenolysis into 1,6 hexanediol 1,5 pentanediol can be produced from biomass without expensive RhRe catalysts 3
31 31
A New Catalytic Pathway for the Production of Bio-based 1,5-Pentanediol
 A New Catalytic Pathway for the Production of Bio-based 1,5-Pentanediol Kevin J. Barnett, Kefeng Huang, and George W. Huber [ kjbarnett@pyranco.com ] MI Forest Bioeconomy Conference February 12, 2019 Oxidation
A New Catalytic Pathway for the Production of Bio-based 1,5-Pentanediol Kevin J. Barnett, Kefeng Huang, and George W. Huber [ kjbarnett@pyranco.com ] MI Forest Bioeconomy Conference February 12, 2019 Oxidation
Department of Chemical and Biological Engineering, University of Wisconsin-Madison, Madison, WI 53706, USA. *
 Mechanistic Insights into the Hydrogenolysis of Levoglucosanol over Bifunctional Platinum Silica-Alumina Catalysts Siddarth H. Krishna +, Rajeev S. Assary #, Quinn A. Rashke +, Zachary R. Schmidt +, Larry
Mechanistic Insights into the Hydrogenolysis of Levoglucosanol over Bifunctional Platinum Silica-Alumina Catalysts Siddarth H. Krishna +, Rajeev S. Assary #, Quinn A. Rashke +, Zachary R. Schmidt +, Larry
SUPPORTING INFORMATION. Framework and Extraframework Tin Sites in Zeolite Beta React Glucose Differently
 SUPPORTING INFORMATION Framework and Extraframework Tin Sites in Zeolite Beta React Glucose Differently Ricardo Bermejo-Deval, Rajamani Gounder and Mark E. Davis* Chemical Engineering, California Institute
SUPPORTING INFORMATION Framework and Extraframework Tin Sites in Zeolite Beta React Glucose Differently Ricardo Bermejo-Deval, Rajamani Gounder and Mark E. Davis* Chemical Engineering, California Institute
Prediction and accelerated laboratory discovery of heterogeneous catalysts
 Prediction and accelerated laboratory discovery of heterogeneous catalysts Kenneth R. Poeppelmeier Charles E. and Emma H. Morrison Professor of Chemistry Department of Chemistry 2145 Sheridan Road Evanston,
Prediction and accelerated laboratory discovery of heterogeneous catalysts Kenneth R. Poeppelmeier Charles E. and Emma H. Morrison Professor of Chemistry Department of Chemistry 2145 Sheridan Road Evanston,
Tetrahydrofuran (THF) is a versatile chemical that has commercial application as a solvent for
 0 Supplementary Information: Tetrahydrofuran (THF) is a versatile chemical that has commercial application as a solvent for the manufacture of plastics and is closely related to,-butanediol (BDO) and γ-butyrolactone
0 Supplementary Information: Tetrahydrofuran (THF) is a versatile chemical that has commercial application as a solvent for the manufacture of plastics and is closely related to,-butanediol (BDO) and γ-butyrolactone
15.1: Hydrocarbon Reactions
 15.1: Hydrocarbon Reactions Halogenation An alkane will react with a halogen to produce a halalkane and the corresponding hydrogen halide. The catalyst is ultraviolet radiation. Reaction 1 methane chlorine
15.1: Hydrocarbon Reactions Halogenation An alkane will react with a halogen to produce a halalkane and the corresponding hydrogen halide. The catalyst is ultraviolet radiation. Reaction 1 methane chlorine
Synthesis of jet fuel range cycloalkanes with diacetone alcohol. from lignocellulose
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2016 Supporting Information Synthesis of jet fuel range cycloalkanes with diacetone alcohol from
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2016 Supporting Information Synthesis of jet fuel range cycloalkanes with diacetone alcohol from
Study on the Production of Gamma valerolactone from Hybrid Poplar
 Study on the Production of Gamma valerolactone from Hybrid Poplar Troy Runge, Chunhui Zhang March 16, 2011 Topics of my Talk Bioenergy Gamma valerolactone background Pentose & Hexose hydrolysis Levulinic
Study on the Production of Gamma valerolactone from Hybrid Poplar Troy Runge, Chunhui Zhang March 16, 2011 Topics of my Talk Bioenergy Gamma valerolactone background Pentose & Hexose hydrolysis Levulinic
Chapter 23 Aldehydes and Ketones
 Chapter 23 Aldehydes and Ketones Ketones are common solvents for quickdrying paints. Introduction to General, Organic, and Biochemistry, 10e John Wiley & Sons, Inc Morris Hein, Scott Pattison, and Susan
Chapter 23 Aldehydes and Ketones Ketones are common solvents for quickdrying paints. Introduction to General, Organic, and Biochemistry, 10e John Wiley & Sons, Inc Morris Hein, Scott Pattison, and Susan
Copper based catalysts in the selective dehydration of polyols
 Graduate Theses and Dissertations Iowa State University Capstones, Theses and Dissertations 2015 Copper based catalysts in the selective dehydration of polyols Anita Ntandoyenkosi Bejile Iowa State University
Graduate Theses and Dissertations Iowa State University Capstones, Theses and Dissertations 2015 Copper based catalysts in the selective dehydration of polyols Anita Ntandoyenkosi Bejile Iowa State University
Lecture 15. More Carbonyl Chemistry. Alcohols React with Aldehydes and Ketones in two steps first O R'OH, H + OR" 2R"OH R + H 2 O OR" 3/8/16
 Lecture 15 More Carbonyl Chemistry R" R C + R' 2R" R C R" R' + 2 March 8, 2016 Alcohols React with Aldehydes and Ketones in two steps first R', + R R 1 emiacetal reacts further in acid to yield an acetal
Lecture 15 More Carbonyl Chemistry R" R C + R' 2R" R C R" R' + 2 March 8, 2016 Alcohols React with Aldehydes and Ketones in two steps first R', + R R 1 emiacetal reacts further in acid to yield an acetal
Reactions of Chapter 10 Worksheet and Key
 1) Alcohol Fermentation Reactions of Chapter 10 Worksheet and Key Alcohol fermentation is a series of chemical reaction that convert sugar molecules, such a glucose, into ethanol and C 2. The overall reaction
1) Alcohol Fermentation Reactions of Chapter 10 Worksheet and Key Alcohol fermentation is a series of chemical reaction that convert sugar molecules, such a glucose, into ethanol and C 2. The overall reaction
Supplementary Information. ZIF-8 Immobilized Ni(0) Nanoparticles: Highly Effective Catalysts for Hydrogen Generation from Hydrolysis of Ammonia Borane
 Supplementary Information ZIF-8 Immobilized Ni() Nanoparticles: Highly Effective Catalysts for Hydrogen Generation from Hydrolysis of Ammonia Borane Pei-Zhou Li, a,b Kengo Aranishi, a and Qiang Xu* a,b
Supplementary Information ZIF-8 Immobilized Ni() Nanoparticles: Highly Effective Catalysts for Hydrogen Generation from Hydrolysis of Ammonia Borane Pei-Zhou Li, a,b Kengo Aranishi, a and Qiang Xu* a,b
Oxygen Reduction Reaction
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Oxygen Reduction Reaction Oxygen is the most common oxidant for most fuel cell cathodes simply
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Oxygen Reduction Reaction Oxygen is the most common oxidant for most fuel cell cathodes simply
Department of Chemistry and Chemical Biology, Cornell University, Ithaca 14853
 Supporting Information Synthesis of Structurally Ordered Pt 3 Ti and Pt 3 V Nanoparticles as Methanol Oxidation Catalysts Zhiming Cui, # Hao Chen, # Mengtian Zhao, Daniel Marshall, Yingchao Yu, Héctor
Supporting Information Synthesis of Structurally Ordered Pt 3 Ti and Pt 3 V Nanoparticles as Methanol Oxidation Catalysts Zhiming Cui, # Hao Chen, # Mengtian Zhao, Daniel Marshall, Yingchao Yu, Héctor
Chapter 9. Organic Chemistry: The Infinite Variety of Carbon Compounds. Organic Chemistry
 Chapter 9 Organic Chemistry: The Infinite Variety of Carbon Compounds Organic Chemistry Organic chemistry is defined as the chemistry of carbon compounds. Of tens of millions of known chemical compounds,
Chapter 9 Organic Chemistry: The Infinite Variety of Carbon Compounds Organic Chemistry Organic chemistry is defined as the chemistry of carbon compounds. Of tens of millions of known chemical compounds,
The lifetime of the catalyst, and therefore its stability, are measured in terms of its TN.
 A catalyst may be defined by its Turnover Number (TN). Each time the complete catalyst cycle occurs, we consider one catalytic turnover to have been completed (one mole of product formed per mole of catalyst).
A catalyst may be defined by its Turnover Number (TN). Each time the complete catalyst cycle occurs, we consider one catalytic turnover to have been completed (one mole of product formed per mole of catalyst).
CHAPTER 1 INTRODUCTION
 1 CHAPTER 1 INTRODUCTION Catalysis is an important process to improve the production of chemicals. This phenomenon can be employed in a chemical reaction that is favored thermodynamically but is very slow
1 CHAPTER 1 INTRODUCTION Catalysis is an important process to improve the production of chemicals. This phenomenon can be employed in a chemical reaction that is favored thermodynamically but is very slow
A green and efficient oxidation of alcohols by supported gold. conditions
 A green and efficient oxidation of alcohols by supported gold catalysts using aqueous H 2 O 2 under organic solvent-free conditions Ji Ni, Wen-Jian Yu, Lin He, Hao sun, Yong Cao,* He-Yong He, and Kang-Nian
A green and efficient oxidation of alcohols by supported gold catalysts using aqueous H 2 O 2 under organic solvent-free conditions Ji Ni, Wen-Jian Yu, Lin He, Hao sun, Yong Cao,* He-Yong He, and Kang-Nian
Supporting Information
 Supporting Information A Rational Solid-state Synthesis of Supported Au-Ni Bimetallic Nanoparticles with Enhanced Activity for Gas-phase Selective Oxidation of Alcohols Wuzhong Yi, a Wentao Yuan, b Ye
Supporting Information A Rational Solid-state Synthesis of Supported Au-Ni Bimetallic Nanoparticles with Enhanced Activity for Gas-phase Selective Oxidation of Alcohols Wuzhong Yi, a Wentao Yuan, b Ye
Chapter 25: The Chemistry of Life: Organic and Biological Chemistry
 Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
Chapter 10: Carboxylic Acids and Their Derivatives
 Chapter 10: Carboxylic Acids and Their Derivatives The back of the white willow tree (Salix alba) is a source of salicylic acid which is used to make aspirin (acetylsalicylic acid) The functional group
Chapter 10: Carboxylic Acids and Their Derivatives The back of the white willow tree (Salix alba) is a source of salicylic acid which is used to make aspirin (acetylsalicylic acid) The functional group
Electronic Supplementary Information (ESI)
 Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information (ESI) Multi-scale promoting effects
Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information (ESI) Multi-scale promoting effects
Bifunctional Catalysts for Upgrading of Biomass-Derived Oxygenates: A Review
 This is an open access article published under an ACS AuthorChoice License, which permits copying and redistribution of the article or any adaptations for non-commercial purposes. pubs.acs.org/acscatalysis
This is an open access article published under an ACS AuthorChoice License, which permits copying and redistribution of the article or any adaptations for non-commercial purposes. pubs.acs.org/acscatalysis
Carboxylic Acids O R C + H + O - Chemistry 618B
 arboxylic Acids R H R + H + - R - Nomenclature - IUPA IUPA names: drop the -e from the parent alkane and add the suffix -oic acid If the compound contains a carbon-carbon double bond, change the infix
arboxylic Acids R H R + H + - R - Nomenclature - IUPA IUPA names: drop the -e from the parent alkane and add the suffix -oic acid If the compound contains a carbon-carbon double bond, change the infix
Naming Organic Halides. Properties of Organic Halides
 Organic Compounds Organic Halides A hydrocarbon in which one or more hydrogen atoms have been replaced by halogen atoms Freons (chlorofluorocarbons) in refrigeration and air conditioning Teflon (polytetrafluoroethane)
Organic Compounds Organic Halides A hydrocarbon in which one or more hydrogen atoms have been replaced by halogen atoms Freons (chlorofluorocarbons) in refrigeration and air conditioning Teflon (polytetrafluoroethane)
Synthesis of mixed alcohols over K-Ni-MoS 2 catalysts
 Synthesis of mixed alcohols over K-Ni-MoS 2 catalysts Rodrigo Suárez París Supervisors: Magali Boutonnet, Sven Järås Division of Chemical Technology, KTH OUTLINE Introduction and objective Experimental
Synthesis of mixed alcohols over K-Ni-MoS 2 catalysts Rodrigo Suárez París Supervisors: Magali Boutonnet, Sven Järås Division of Chemical Technology, KTH OUTLINE Introduction and objective Experimental
Direct Methane Oxidation over Pt-modified Nitrogen-Doped Carbons
 Supporting Information Direct Methane Oxidation over Pt-modified Nitrogen-Doped Carbons Mario Soorholtz, a Robin J. White,* b,c Tobias Zimmermann, a Maria-Magdalena Titirici, b Markus Antonietti, b Regina
Supporting Information Direct Methane Oxidation over Pt-modified Nitrogen-Doped Carbons Mario Soorholtz, a Robin J. White,* b,c Tobias Zimmermann, a Maria-Magdalena Titirici, b Markus Antonietti, b Regina
Table 8.2 Detailed Table of Characteristic Infrared Absorption Frequencies
 Table 8.2 Detailed Table of Characteristic Infrared Absorption Frequencies The hydrogen stretch region (3600 2500 cm 1 ). Absorption in this region is associated with the stretching vibration of hydrogen
Table 8.2 Detailed Table of Characteristic Infrared Absorption Frequencies The hydrogen stretch region (3600 2500 cm 1 ). Absorption in this region is associated with the stretching vibration of hydrogen
Infrared Spectroscopy An Instrumental Method for Detecting Functional Groups
 Infrared Spectroscopy An Instrumental Method for Detecting Functional Groups 1 The Electromagnetic Spectrum Infrared Spectroscopy I. Physics Review Frequency, υ (nu), is the number of wave cycles that
Infrared Spectroscopy An Instrumental Method for Detecting Functional Groups 1 The Electromagnetic Spectrum Infrared Spectroscopy I. Physics Review Frequency, υ (nu), is the number of wave cycles that
21.1 Introduction Carboxylic Acids Nomenclature of Carboxylic Acids. Acids Structure and Properties of Carboxylic Acids.
 21.1 Introduction Carboxylic Acids Carboxylic acids are abundant in nature and in pharmaceuticals. 21.1 Introduction Carboxylic Acids The US produces over 2.5 million tons of acetic acid per year, which
21.1 Introduction Carboxylic Acids Carboxylic acids are abundant in nature and in pharmaceuticals. 21.1 Introduction Carboxylic Acids The US produces over 2.5 million tons of acetic acid per year, which
Chemical Reactions - Oxidation. Reactions Involving Aldehydes and Ketones. Learning Check. Learning Check. Chemical Reactions - Addition of Hydrogen
 Reactions Involving Aldehydes and Ketones Chemical Reactions - Oxidation When aldehydes are prepared by oxidizing primary alcohols with KMnO 4 or K 2 Cr 2 O 7, the reaction may continue and produce carboxylic
Reactions Involving Aldehydes and Ketones Chemical Reactions - Oxidation When aldehydes are prepared by oxidizing primary alcohols with KMnO 4 or K 2 Cr 2 O 7, the reaction may continue and produce carboxylic
Supplementary Figures
 Supplementary Figures Supplementary Figure 1: Temporal evolution of GVL (green triangles) and LA (black squares) during the catalytic hydrogenation of LA over 1% Au/TiO 2 (M Im ). Reaction conditions:
Supplementary Figures Supplementary Figure 1: Temporal evolution of GVL (green triangles) and LA (black squares) during the catalytic hydrogenation of LA over 1% Au/TiO 2 (M Im ). Reaction conditions:
Synthesis gas production via the biogas reforming reaction over Ni/MgO-Al 2 O 3 and Ni/CaO-Al 2 O 3 catalysts
 Synthesis gas production via the biogas reforming reaction over Ni/MgO-Al 2 O 3 and Ni/CaO-Al 2 O 3 catalysts N.D. Charisiou 1,2, A. Baklavaridis 1, V.G. Papadakis 2, M.A. Goula 1 1 Department of Environmental
Synthesis gas production via the biogas reforming reaction over Ni/MgO-Al 2 O 3 and Ni/CaO-Al 2 O 3 catalysts N.D. Charisiou 1,2, A. Baklavaridis 1, V.G. Papadakis 2, M.A. Goula 1 1 Department of Environmental
ACETONE. PRODUCT IDENTIFICATION CAS NO EINECS NO MOL WT H.S. CODE Oral rat LD50: 5800 mg/kg
 ACETONE www.pawarchemicals.com PRODUCT IDENTIFICATION CAS NO 67-64-1 EINECS NO. 200-662-2 FORMULA (CH3)2C=O MOL WT. 58.08 H.S. CODE 2914.11 TOXICITY SYNONYMS Oral rat LD50: 5800 mg/kg Dimethyl ketone;
ACETONE www.pawarchemicals.com PRODUCT IDENTIFICATION CAS NO 67-64-1 EINECS NO. 200-662-2 FORMULA (CH3)2C=O MOL WT. 58.08 H.S. CODE 2914.11 TOXICITY SYNONYMS Oral rat LD50: 5800 mg/kg Dimethyl ketone;
Supporting Information High Activity and Selectivity of Ag/SiO 2 Catalyst for Hydrogenation of Dimethyloxalate
 Supporting Information High Activity and Selectivity of Ag/SiO 2 Catalyst for Hydrogenation of Dimethyloxalate An-Yuan Yin, Xiao-Yang Guo, Wei-Lin Dai*, Kang-Nian Fan Shanghai Key Laboratory of Molecular
Supporting Information High Activity and Selectivity of Ag/SiO 2 Catalyst for Hydrogenation of Dimethyloxalate An-Yuan Yin, Xiao-Yang Guo, Wei-Lin Dai*, Kang-Nian Fan Shanghai Key Laboratory of Molecular
Isobutanol and Methanol Synthesis on Copper Supported on Alkali- Modified MgO and ZnO Supports
 NATURAL GAS CONVERSION V Studies in Surface Science and Catalysis, Vol. 119 A. Parmaliana et al. (Editors) o 1998 Elsevier Science B.V. All rights reserved. 509 Isobutanol and Methanol Synthesis on Copper
NATURAL GAS CONVERSION V Studies in Surface Science and Catalysis, Vol. 119 A. Parmaliana et al. (Editors) o 1998 Elsevier Science B.V. All rights reserved. 509 Isobutanol and Methanol Synthesis on Copper
cis,cis-muconic acid: Separation and catalysis to bioadipic acid for nylon-6,6 polymerization
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information for: cis,cis-muconic acid: Separation and catalysis
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information for: cis,cis-muconic acid: Separation and catalysis
Catalytic Hydrodesulfurisation
 CHAPTER 2 Catalytic Hydrodesulfurisation 1 The Process Although some of the organic sulfur compounds found in oil and other feedstocks can be removed by the absorption, adsorption and oxidation processes
CHAPTER 2 Catalytic Hydrodesulfurisation 1 The Process Although some of the organic sulfur compounds found in oil and other feedstocks can be removed by the absorption, adsorption and oxidation processes
9. Which compound is an alcohol? A) methanol C) butane B) ethyne D) propanal
 1. Given the structural formulas for two organic compounds: The differences in their physical and chemical properties are primarily due to their different A) number of hydrogen atoms B) number of carbon
1. Given the structural formulas for two organic compounds: The differences in their physical and chemical properties are primarily due to their different A) number of hydrogen atoms B) number of carbon
E (mol NO 2) L 1 s 1. C (mol NO 2) L 1 s 1. D (mol NO 2) L 1 s 1. Answer: B
 CH302 Spring 2008 Practice Exam 3 [Q1] Multiple Choice - 1 point 1) The rate of formation of oxygen in the reaction 2N 2 O 5 (g) 4NO 2 (g) + O 2 (g) is 2.28 (mol O 2 ) L 1 s 1. What is the rate of formation
CH302 Spring 2008 Practice Exam 3 [Q1] Multiple Choice - 1 point 1) The rate of formation of oxygen in the reaction 2N 2 O 5 (g) 4NO 2 (g) + O 2 (g) is 2.28 (mol O 2 ) L 1 s 1. What is the rate of formation
Reversible Additions to carbonyls: Weak Nucleophiles Relative Reactivity of carbonyls: Hydration of Ketones and Aldehydes
 Reversible Additions to carbonyls: Weak Nucleophiles Weak nucleophiles, such as water, alcohols, and amines, require acid or base catalysis to undergo addition to carbonyl compounds Relative Reactivity
Reversible Additions to carbonyls: Weak Nucleophiles Weak nucleophiles, such as water, alcohols, and amines, require acid or base catalysis to undergo addition to carbonyl compounds Relative Reactivity
First-Principles Kinetic Model for 2-Methyl- Tetrahydrofuran Pyrolysis and Combustion
 First-Principles Kinetic Model for 2-Methyl- Tetrahydrofuran Pyrolysis and Combustion Ruben De Bruycker a, Luc-Sy Tran a,b,*, Hans-Heinrich Carstensen a, Pierre-Alexandre Glaude b, Frédérique Battin-Leclerc
First-Principles Kinetic Model for 2-Methyl- Tetrahydrofuran Pyrolysis and Combustion Ruben De Bruycker a, Luc-Sy Tran a,b,*, Hans-Heinrich Carstensen a, Pierre-Alexandre Glaude b, Frédérique Battin-Leclerc
Lecture 18. Oxidation and Reduction. Oxidation. Reduction O CH 4 CH 3 OH H C H. Chemistry 328N
 Lecture 18 xidation and Reduction C 4 C 3 C C C xidation Reduction March 27, 2018 Suppose you want to make this compound????? C + BrC 2 C 2 C?? CC 2 C 2 C 4-ydroxy-4-phenylbutanal It s an alcohol. Use
Lecture 18 xidation and Reduction C 4 C 3 C C C xidation Reduction March 27, 2018 Suppose you want to make this compound????? C + BrC 2 C 2 C?? CC 2 C 2 C 4-ydroxy-4-phenylbutanal It s an alcohol. Use
Aqueous-Phase Reforming of Ethylene Glycol Over Supported Platinum Catalysts
 University of Massachusetts Amherst From the SelectedWorks of George W. Huber 2003 Aqueous-Phase Reforming of Ethylene Glycol Over Supported Platinum Catalysts George W Huber, University of Massachusetts
University of Massachusetts Amherst From the SelectedWorks of George W. Huber 2003 Aqueous-Phase Reforming of Ethylene Glycol Over Supported Platinum Catalysts George W Huber, University of Massachusetts
An Alternative Biofuel: LPG from Biomass- Derived Organic Acids TIE Graduate Research Fellowship Report
 An Alternative Biofuel: LPG from Biomass- Derived Organic Acids 2010-2011 TIE Graduate Research Fellowship Report Branko Zugic Department of Chemical and Biological Engineering Tufts University, Medford
An Alternative Biofuel: LPG from Biomass- Derived Organic Acids 2010-2011 TIE Graduate Research Fellowship Report Branko Zugic Department of Chemical and Biological Engineering Tufts University, Medford
Green Chemistry. A catalytic aldol condensation system enables one pot conversion of biomass saccharides to biofuel intermediates PAPER.
 Green Chemistry PAPER Cite this: Green Chem., 2017, 19, 1751 A catalytic aldol condensation system enables one pot conversion of biomass saccharides to biofuel intermediates Huixiang Li, a,b Zhanwei Xu,
Green Chemistry PAPER Cite this: Green Chem., 2017, 19, 1751 A catalytic aldol condensation system enables one pot conversion of biomass saccharides to biofuel intermediates Huixiang Li, a,b Zhanwei Xu,
Supplementary Information for Efficient catalytic conversion of fructose into hydroxymethylfurfural by a novel carbon based solid acid
 Supplementary Information for Efficient catalytic conversion of fructose into hydroxymethylfurfural by a novel carbon based solid acid Jianjian Wang, Wenjie Xu, Jiawen Ren*, Xiaohui Liu, Guanzhong Lu,
Supplementary Information for Efficient catalytic conversion of fructose into hydroxymethylfurfural by a novel carbon based solid acid Jianjian Wang, Wenjie Xu, Jiawen Ren*, Xiaohui Liu, Guanzhong Lu,
Chapter 1 IB Chemistry Warm Ups Stoichiometry. Mrs. Hilliard
 Chapter 1 IB Chemistry Warm Ups Stoichiometry Mrs. Hilliard Vocabulary 1. Atomic theory 2. Kelvin 3. Mole 4. Relative abundance 5. Molar Mass 6. Empirical formula 7. Molecular formula 8. Stoichiometry
Chapter 1 IB Chemistry Warm Ups Stoichiometry Mrs. Hilliard Vocabulary 1. Atomic theory 2. Kelvin 3. Mole 4. Relative abundance 5. Molar Mass 6. Empirical formula 7. Molecular formula 8. Stoichiometry
Silesian University of Technology, Poland Recoverable and recyclable catalysts for sustainable chemical processes
 Silesian University of Technology, Poland Recoverable and recyclable catalysts for sustainable chemical processes Anna Chrobok What are the challenges for the sustainable chemical industry today? reduce
Silesian University of Technology, Poland Recoverable and recyclable catalysts for sustainable chemical processes Anna Chrobok What are the challenges for the sustainable chemical industry today? reduce
Facile synthesis of polymer and carbon spheres decorated with highly dispersed metal nanoparticles
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 1 Facile synthesis of polymer and carbon spheres decorated with highly dispersed metal nanoparticles
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 1 Facile synthesis of polymer and carbon spheres decorated with highly dispersed metal nanoparticles
1. What is the letter of the alphabet in parentheses that follows EXAM I in the title above? a. a b. b c. c d. d e. e
 HEM 102, EXAM I ( a ) 1. What is the letter of the alphabet in parentheses that follows EXAM I in the title above? a. a b. b c. c d. d e. e 2. Which compound has the most constitutional isomers? a. 2 H
HEM 102, EXAM I ( a ) 1. What is the letter of the alphabet in parentheses that follows EXAM I in the title above? a. a b. b c. c d. d e. e 2. Which compound has the most constitutional isomers? a. 2 H
Aromatic Hydrocarbons
 Aromatic Hydrocarbons Aromatic hydrocarbons contain six-membered rings of carbon atoms with alternating single and double carbon-carbon bonds. The ring is sometimes shown with a circle in the center instead
Aromatic Hydrocarbons Aromatic hydrocarbons contain six-membered rings of carbon atoms with alternating single and double carbon-carbon bonds. The ring is sometimes shown with a circle in the center instead
Chemistry 106 Fall 2006 Exam 1 Form A 1. Does this molecule have both cis and trans isomers?
 1. Does this molecule have both cis and trans isomers? Cl A. No, it has only the cis isomer. B. Yes, this is the cis isomer. C. Yes, this is the trans isomer. D. No. E. No, it has only the trans isomer
1. Does this molecule have both cis and trans isomers? Cl A. No, it has only the cis isomer. B. Yes, this is the cis isomer. C. Yes, this is the trans isomer. D. No. E. No, it has only the trans isomer
Selective O-Alkylation Reaction of Hydroquinone with Methanol over Cs Ion-Exchanged Zeolites
 Korean J. Chem. Eng., 19(3), 406-410 (2002) Selective O-Alkylation Reaction of Hydroquinone with Methanol over Cs Ion-Exchanged Zeolites Sang Sung Lee, Soo Chool Lee and Jae Chang Kim Department of Chemical
Korean J. Chem. Eng., 19(3), 406-410 (2002) Selective O-Alkylation Reaction of Hydroquinone with Methanol over Cs Ion-Exchanged Zeolites Sang Sung Lee, Soo Chool Lee and Jae Chang Kim Department of Chemical
Supporting Information for. Selectivity and Activity in Catalytic Methanol Oxidation in the Gas Phase
 1 / 5 Supporting Information for The Influence of Size-Induced Oxidation State of Platinum Nanoparticles on Selectivity and Activity in Catalytic Methanol Oxidation in the Gas Phase Hailiang Wang, Yihai
1 / 5 Supporting Information for The Influence of Size-Induced Oxidation State of Platinum Nanoparticles on Selectivity and Activity in Catalytic Methanol Oxidation in the Gas Phase Hailiang Wang, Yihai
Infrared Spectroscopy
 Infrared Spectroscopy IR Spectroscopy Used to identify organic compounds IR spectroscopy provides a 100% identification if the spectrum is matched. If not, IR at least provides information about the types
Infrared Spectroscopy IR Spectroscopy Used to identify organic compounds IR spectroscopy provides a 100% identification if the spectrum is matched. If not, IR at least provides information about the types
Aqueous-phase reforming a pathway to chemicals and fuels
 Alexey Kirilin Aqueous-phase reforming a pathway to chemicals and fuels Laboratory of Industrial Chemistry and Reaction Engineering Process Chemistry Centre Åbo Akademi Agenda 2 Short Introduction, biomass,
Alexey Kirilin Aqueous-phase reforming a pathway to chemicals and fuels Laboratory of Industrial Chemistry and Reaction Engineering Process Chemistry Centre Åbo Akademi Agenda 2 Short Introduction, biomass,
Atovaquone: An Antipneumocystic Agent
 Atovaquone: An Antipneumocystic Agent Atovaquone is a pharmaceutical compound marketed in the United States under different combinations to prevent and treat pneumocystosis and malaria. In a report from
Atovaquone: An Antipneumocystic Agent Atovaquone is a pharmaceutical compound marketed in the United States under different combinations to prevent and treat pneumocystosis and malaria. In a report from
CO 2 and CO activation
 2 and activation Most organic chemicals are currently made commercially from ethylene, a product of oil refining. Itispossiblethatinthenextseveraldecadeswemayhavetoshifttowardothercarbonsources for these
2 and activation Most organic chemicals are currently made commercially from ethylene, a product of oil refining. Itispossiblethatinthenextseveraldecadeswemayhavetoshifttowardothercarbonsources for these
Chapter 7: Alcohols, Phenols and Thiols
 Chapter 7: Alcohols, Phenols and Thiols 45 -Alcohols have the general formula R-OH and are characterized by the presence of a hydroxyl group, -OH. -Phenols have a hydroxyl group attached directly to an
Chapter 7: Alcohols, Phenols and Thiols 45 -Alcohols have the general formula R-OH and are characterized by the presence of a hydroxyl group, -OH. -Phenols have a hydroxyl group attached directly to an
Supplementary Figure 1 HAADF-STEM images of 0.08%Pt/FeO x -R200 single-atom catalyst with different magnifications. Scale bar: a, 20 nm; b, 10 nm; c,
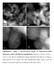 Supplementary Figure 1 HAADF-STEM images of 0.08%Pt/FeO x -R200 single-atom catalyst with different magnifications. Scale bar: a, 20 nm; b, 10 nm; c, 2 nm; d, 2 nm. The low magnification images demonstrate
Supplementary Figure 1 HAADF-STEM images of 0.08%Pt/FeO x -R200 single-atom catalyst with different magnifications. Scale bar: a, 20 nm; b, 10 nm; c, 2 nm; d, 2 nm. The low magnification images demonstrate
Chapter 17: Alcohols and Phenols. Based on McMurry s Organic Chemistry, 7 th edition
 Chapter 17: Alcohols and Phenols Based on McMurry s Organic Chemistry, 7 th edition Alcohols and Phenols Alcohols contain an OH group connected to a a saturated C (sp 3 ) They are important solvents and
Chapter 17: Alcohols and Phenols Based on McMurry s Organic Chemistry, 7 th edition Alcohols and Phenols Alcohols contain an OH group connected to a a saturated C (sp 3 ) They are important solvents and
Sodium Borohydride Reduction of Benzoin
 Sodium Borohydride Reduction of Benzoin Introduction The most common and useful reducing agents for reducing aldehydes, ketones, and other functional groups are metal hydride reagents. The two most common
Sodium Borohydride Reduction of Benzoin Introduction The most common and useful reducing agents for reducing aldehydes, ketones, and other functional groups are metal hydride reagents. The two most common
Catalytic Chemistry. Bruce C. Gates. John Wiley & Sons, Inc. New York Chichester Brisbane Toronto Singapore. University of Delaware ^.'-'.
 : s / ; '.... ;. : : ^.'-'. Catalytic Chemistry Bruce C. Gates University of Delaware John Wiley & Sons, Inc. New York Chichester Brisbane Toronto Singapore Contents List of Notation xix 1 INTRODUCTION
: s / ; '.... ;. : : ^.'-'. Catalytic Chemistry Bruce C. Gates University of Delaware John Wiley & Sons, Inc. New York Chichester Brisbane Toronto Singapore Contents List of Notation xix 1 INTRODUCTION
Production of 5-Hydroxymethylfurfural from Glucose Using a Combination of Lewis and
 Supporting Information Production of 5-ydroxymethylfurfural from Glucose Using a Combination of Lewis and Brønsted Acid Catalysts in Water in a Biphasic Reactor with an Alkylphenol Solvent Yomaira J. Pagán-Torres,
Supporting Information Production of 5-ydroxymethylfurfural from Glucose Using a Combination of Lewis and Brønsted Acid Catalysts in Water in a Biphasic Reactor with an Alkylphenol Solvent Yomaira J. Pagán-Torres,
Alcohols and Phenols. Classification of Alcohols. Learning Check. Lecture 4 Alcohols, Phenols, and Thiols. Alcohols, Phenols, and Thiols
 Lecture 4 Alcohols, Phenols, and Thiols Alcohols, Phenols, and Thiols Alcohols and Phenols An alcohol contains A hydroxyl group ( ) attached to a carbon chain. A phenol contains A hydroxyl group ( ) attached
Lecture 4 Alcohols, Phenols, and Thiols Alcohols, Phenols, and Thiols Alcohols and Phenols An alcohol contains A hydroxyl group ( ) attached to a carbon chain. A phenol contains A hydroxyl group ( ) attached
Strategic use of CuAlO 2 as a sustained release catalyst for production of hydrogen from methanol steam reforming
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Strategic use of CuAlO 2 as a sustained release catalyst for
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Strategic use of CuAlO 2 as a sustained release catalyst for
Depressing the hydrogenation and decomposition. nanoparticles on oxygen functionalized. carbon nanofibers. Supporting Information
 Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2015 Depressing the hydrogenation and decomposition reaction in H 2 O 2 synthesis
Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2015 Depressing the hydrogenation and decomposition reaction in H 2 O 2 synthesis
University of Groningen. Levulinic acid from lignocellulosic biomass Girisuta, Buana
 University of Groningen Levulinic acid from lignocellulosic biomass Girisuta, Buana IMPRTANT NTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
University of Groningen Levulinic acid from lignocellulosic biomass Girisuta, Buana IMPRTANT NTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
High Performance Liquid Chromatography
 Updated: 3 November 2014 Print version High Performance Liquid Chromatography David Reckhow CEE 772 #18 1 HPLC System David Reckhow CEE 772 #18 2 Instrument Basics PUMP INJECTION POINT DETECTOR COLUMN
Updated: 3 November 2014 Print version High Performance Liquid Chromatography David Reckhow CEE 772 #18 1 HPLC System David Reckhow CEE 772 #18 2 Instrument Basics PUMP INJECTION POINT DETECTOR COLUMN
High Performance Liquid Chromatography
 Updated: 3 November 2014 Print version High Performance Liquid Chromatography David Reckhow CEE 772 #18 1 HPLC System David Reckhow CEE 772 #18 2 1 Instrument Basics PUMP INJECTION POINT DETECTOR COLUMN
Updated: 3 November 2014 Print version High Performance Liquid Chromatography David Reckhow CEE 772 #18 1 HPLC System David Reckhow CEE 772 #18 2 1 Instrument Basics PUMP INJECTION POINT DETECTOR COLUMN
Chapter 18: Ketones and Aldehydes. I. Introduction
 1 Chapter 18: Ketones and Aldehydes I. Introduction We have already encountered numerous examples of this functional group (ketones, aldehydes, carboxylic acids, acid chlorides, etc). The three-dimensional
1 Chapter 18: Ketones and Aldehydes I. Introduction We have already encountered numerous examples of this functional group (ketones, aldehydes, carboxylic acids, acid chlorides, etc). The three-dimensional
Alkenes are prepared by the reverse of the electrophilic reactions in the last chapter. That is, they are prepared by elimination reactions.
 Ch 8 Alkene Reactions and Syntheses Alkenes are prepared by the reverse of the electrophilic reactions in the last chapter. That is, they are prepared by elimination reactions. Dehydrohalogenation - Mechanism
Ch 8 Alkene Reactions and Syntheses Alkenes are prepared by the reverse of the electrophilic reactions in the last chapter. That is, they are prepared by elimination reactions. Dehydrohalogenation - Mechanism
ALCOHOLS AND PHENOLS
 ALCOHOLS AND PHENOLS ALCOHOLS AND PHENOLS Alcohols contain an OH group connected to a a saturated C (sp3) They are important solvents and synthesis intermediates Phenols contain an OH group connected to
ALCOHOLS AND PHENOLS ALCOHOLS AND PHENOLS Alcohols contain an OH group connected to a a saturated C (sp3) They are important solvents and synthesis intermediates Phenols contain an OH group connected to
Organic Chemistry. REACTIONS Grade 12 Physical Science Mrs KL Faling
 Organic Chemistry REACTIONS Grade 12 Physical Science Mrs KL Faling SUBSTITUTION REACTIONS This is a reaction where an atom or group of atoms is replaced by another atom or group of atoms Substitution
Organic Chemistry REACTIONS Grade 12 Physical Science Mrs KL Faling SUBSTITUTION REACTIONS This is a reaction where an atom or group of atoms is replaced by another atom or group of atoms Substitution
Chapter 13: Alcohols and Phenols
 Chapter 13: Alcohols and Phenols [ Chapter 9 Sections: 9.10; Chapter 13 Sections: 13.1-13.3, 13.9-13.10] 1. Nomenclature of Alcohols simple alcohols C3 C3C2 Eddie Sachs 1927-1964 larger alcohols find the
Chapter 13: Alcohols and Phenols [ Chapter 9 Sections: 9.10; Chapter 13 Sections: 13.1-13.3, 13.9-13.10] 1. Nomenclature of Alcohols simple alcohols C3 C3C2 Eddie Sachs 1927-1964 larger alcohols find the
Structural and Electronic properties of platinum nanoparticles studied by diffraction and absorption spectroscopy
 The 4 th SUNBEAM Workshop Structural and Electronic properties of platinum nanoparticles studied by in situ x-ray x diffraction and in situ x-ray x absorption spectroscopy Hideto Imai Fundamental and Environmental
The 4 th SUNBEAM Workshop Structural and Electronic properties of platinum nanoparticles studied by in situ x-ray x diffraction and in situ x-ray x absorption spectroscopy Hideto Imai Fundamental and Environmental
Organic Chemistry. Organic chemistry is the chemistry of compounds containing carbon.
 Organic Chemistry Organic Chemistry Organic chemistry is the chemistry of compounds containing carbon. In this chapter we will discuss the structural features of organic molecules, nomenclature, and a
Organic Chemistry Organic Chemistry Organic chemistry is the chemistry of compounds containing carbon. In this chapter we will discuss the structural features of organic molecules, nomenclature, and a
An alcohol is a compound obtained by substituting a hydoxyl group ( OH) for an H atom on a carbon atom of a hydrocarbon group.
 Derivatives of Hydrocarbons A functional group is a reactive portion of a molecule that undergoes predictable reactions. All other organic compounds can be considered as derivatives of hydrocarbons (i.e.,
Derivatives of Hydrocarbons A functional group is a reactive portion of a molecule that undergoes predictable reactions. All other organic compounds can be considered as derivatives of hydrocarbons (i.e.,
NO REDUCTION BY GRAPEFRUIT SKIN-BASED CARBON CATALYST. Abstract. Introduction. Experimental
 REDUCTION BY GRAPEFRUIT SKIN-BASED CARBON CATALYST Rosas Juana Mª, Bedia Jorge, Rodríguez-Mirasol José, Cordero Tomás Dept of Chemical Engineering, University of Málaga, Málaga, 2913 (SPAIN) Abstract reduction
REDUCTION BY GRAPEFRUIT SKIN-BASED CARBON CATALYST Rosas Juana Mª, Bedia Jorge, Rodríguez-Mirasol José, Cordero Tomás Dept of Chemical Engineering, University of Málaga, Málaga, 2913 (SPAIN) Abstract reduction
QuickTime and a TIFF (Uncompressed) decompressor are needed to see this picture. Organic Chemistry. QuickTime and a are needed to see this picture.
 QuickTime and a TIFF (Uncompressed) decompressor are needed to see this picture. Organic Chemistry QuickTime and a TIFF (Uncompressed) decompressor are needed to see this picture. Organic Chemistry Has
QuickTime and a TIFF (Uncompressed) decompressor are needed to see this picture. Organic Chemistry QuickTime and a TIFF (Uncompressed) decompressor are needed to see this picture. Organic Chemistry Has
CHEM 203. Final Exam December 15, 2010 ANSWERS. This a closed-notes, closed-book exam. You may use your set of molecular models
 CEM 203 Final Exam December 15, 2010 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This test contains 15 pages Time: 2h 30 min 1. / 16 2. / 15 3. / 24
CEM 203 Final Exam December 15, 2010 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This test contains 15 pages Time: 2h 30 min 1. / 16 2. / 15 3. / 24
Direct Synthesis of H 2 O 2 on AgPt Octahedra: The Importance of Ag-Pt Coordination for High H 2 O 2 Selectivity
 Supporting Information Direct Synthesis of H 2 O 2 on AgPt Octahedra: The Importance of Ag-Pt Coordination for High H 2 O 2 Selectivity Neil M. Wilson, 1 Yung-Tin Pan, 1 Yu-Tsun Shao, 2 Jian-Min Zuo, 2
Supporting Information Direct Synthesis of H 2 O 2 on AgPt Octahedra: The Importance of Ag-Pt Coordination for High H 2 O 2 Selectivity Neil M. Wilson, 1 Yung-Tin Pan, 1 Yu-Tsun Shao, 2 Jian-Min Zuo, 2
Sustainable Energy Technologies
 Sustainable Energy Technologies Molecular Heterogeneous Catalysis Hydrogen Technology Renewable feedstocks Fuel cell catalysis Prof. Dr. Emiel Hensen Prof. Dr. Peter Notten Prof. Dr. Jaap Schouten Chemical
Sustainable Energy Technologies Molecular Heterogeneous Catalysis Hydrogen Technology Renewable feedstocks Fuel cell catalysis Prof. Dr. Emiel Hensen Prof. Dr. Peter Notten Prof. Dr. Jaap Schouten Chemical
Class XI Chapter 1 Some Basic Concepts of Chemistry Chemistry
 Question 1.1: Calculate the molecular mass of the following: (i) H 2 O (ii) CO 2 (iii) CH 4 (i) H 2 O: The molecular mass of water, H 2 O = (2 Atomic mass of hydrogen) + (1 Atomic mass of oxygen) = [2(1.0084)
Question 1.1: Calculate the molecular mass of the following: (i) H 2 O (ii) CO 2 (iii) CH 4 (i) H 2 O: The molecular mass of water, H 2 O = (2 Atomic mass of hydrogen) + (1 Atomic mass of oxygen) = [2(1.0084)
Dr Panagiotis Kechagiopoulos. Lecturer in Chemical Engineering. School of Engineering
 Catalytic reforming of biomass derived oxygenates for sustainable hydrogen production: Experimental investigations, microkinetic modelling and reactor design Dr Panagiotis Kechagiopoulos Lecturer in Chemical
Catalytic reforming of biomass derived oxygenates for sustainable hydrogen production: Experimental investigations, microkinetic modelling and reactor design Dr Panagiotis Kechagiopoulos Lecturer in Chemical
Assistant Lecturer: Sahar Mohammed Shakir Assistant Lecturer: Sarah Sattar Jabbar
 Assistant Lecturer: Sahar Mohammed Shakir Assistant Lecturer: Sarah Sattar Jabbar Structure Aldehydes are cpd.s of the general formula R ; Ketones are cpd.s of the general formula RŔ. The groups R and
Assistant Lecturer: Sahar Mohammed Shakir Assistant Lecturer: Sarah Sattar Jabbar Structure Aldehydes are cpd.s of the general formula R ; Ketones are cpd.s of the general formula RŔ. The groups R and
Propylene: key building block for the production of important petrochemicals
 Propylene production from 11-butene and ethylene catalytic cracking: Study of the performance of HZSMHZSM-5 zeolites and silicoaluminophosphates SAPO--34 and SAPOSAPO SAPO-18 E. Epelde Epelde*, *, A.G.
Propylene production from 11-butene and ethylene catalytic cracking: Study of the performance of HZSMHZSM-5 zeolites and silicoaluminophosphates SAPO--34 and SAPOSAPO SAPO-18 E. Epelde Epelde*, *, A.G.
Conversion Technologies Chemical and Catalytical Processing: Gas phase route
 1 st Brazil-U.S. Biofuels Short Course: Providing Interdisciplinary Education in Biofuels Technology July 27 - August 7, 2009 University of Sao Paulo, Sao Paulo, Brazil Conversion Technologies Chemical
1 st Brazil-U.S. Biofuels Short Course: Providing Interdisciplinary Education in Biofuels Technology July 27 - August 7, 2009 University of Sao Paulo, Sao Paulo, Brazil Conversion Technologies Chemical
CHM 292 Final Exam Answer Key
 CHM 292 Final Exam Answer Key 1. Predict the product(s) of the following reactions (5 points each; 35 points total). May 7, 2013 Acid catalyzed elimination to form the most highly substituted alkene possible
CHM 292 Final Exam Answer Key 1. Predict the product(s) of the following reactions (5 points each; 35 points total). May 7, 2013 Acid catalyzed elimination to form the most highly substituted alkene possible
CHEM 203. Midterm Exam 1 October 31, 2008 ANSWERS. This a closed-notes, closed-book exam. You may use your set of molecular models
 CEM 203 Midterm Exam 1 ctober 31, 2008 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This exam contains 8 pages Time: 1h 30 min 1. / 15 2. / 16 3. /
CEM 203 Midterm Exam 1 ctober 31, 2008 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This exam contains 8 pages Time: 1h 30 min 1. / 15 2. / 16 3. /
Green Oxidations with Tungsten Catalysts. by Mike Kuszpit Michigan State University
 Green xidations with Tungsten Catalysts by Mike Kuszpit Michigan State University xidations in rganic Chemistry [] [] R 1 R 1 R 1 [] R 1 R 2 R 1 R 2 [] R 1 R 2 R 1 R 2 R 1 R 2 [] R 1 R 2 Essential as building
Green xidations with Tungsten Catalysts by Mike Kuszpit Michigan State University xidations in rganic Chemistry [] [] R 1 R 1 R 1 [] R 1 R 2 R 1 R 2 [] R 1 R 2 R 1 R 2 R 1 R 2 [] R 1 R 2 Essential as building
Ir-Re Alloy as a highly active catalyst for the hydrogenolysis
 Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2014 Supporting Information Ir-Re Alloy as a highly active catalyst for the hydrogenolysis
Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2014 Supporting Information Ir-Re Alloy as a highly active catalyst for the hydrogenolysis
One-Pot Reaction Cascades Catalyzed by Base and Acid Confined Inside Pores of Mesoporous Silica Nanospheres
 One-Pot Reaction Cascades Catalyzed by Base and Acid Confined Inside Pores of Mesoporous Silica Nanospheres Yulin Huang, Brian Trewyn, Hung-ting Chen, Shu Xu, Victor S.-Y. Lin* Department of Chemistry
One-Pot Reaction Cascades Catalyzed by Base and Acid Confined Inside Pores of Mesoporous Silica Nanospheres Yulin Huang, Brian Trewyn, Hung-ting Chen, Shu Xu, Victor S.-Y. Lin* Department of Chemistry
Supporting Information
 Supporting Information High Performance Electrocatalyst: Pt-Cu Hollow Nanocrystals Xiaofei Yu, a Dingsheng, a Qing Peng a and Yadong Li* a a Department of Chemistry, Tsinghua University, Beijing, 100084
Supporting Information High Performance Electrocatalyst: Pt-Cu Hollow Nanocrystals Xiaofei Yu, a Dingsheng, a Qing Peng a and Yadong Li* a a Department of Chemistry, Tsinghua University, Beijing, 100084
Supplementary Figure 1 Morpholigical properties of TiO 2-x SCs. The statistical particle size distribution (a) of the defective {001}-TiO 2-x SCs and
 Supplementary Figure 1 Morpholigical properties of TiO 2-x s. The statistical particle size distribution (a) of the defective {1}-TiO 2-x s and their typical TEM images (b, c). Quantity Adsorbed (cm 3
Supplementary Figure 1 Morpholigical properties of TiO 2-x s. The statistical particle size distribution (a) of the defective {1}-TiO 2-x s and their typical TEM images (b, c). Quantity Adsorbed (cm 3
Facile and selective hydrogenolysis of β-o-4 linkages in lignin catalyzed by Pd-Ni bimetallic nanoparticles supported on ZrO 2
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2016 S1 Supporting Information Facile and selective hydrogenolysis of β--4 linkages in lignin
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2016 S1 Supporting Information Facile and selective hydrogenolysis of β--4 linkages in lignin
CHEM Chapter 12 Infrared and Mass Spec (homework). Stafford. S18
 Exhibit 12-4 The following question(s) refer to the mass spectrum shown below. 1. Refer to Exhibit 12-4. This compound contains C, H, and one other atom. Identify the other atom from the mass spectrum
Exhibit 12-4 The following question(s) refer to the mass spectrum shown below. 1. Refer to Exhibit 12-4. This compound contains C, H, and one other atom. Identify the other atom from the mass spectrum
Chemistry 110. Bettelheim, Brown, Campbell & Farrell. Ninth Edition
 Chemistry 110 Bettelheim, Brown, Campbell & Farrell Ninth Edition Introduction to General, rganic and Biochemistry Chapter 14 Alcohols, Ethers and Thiols Alcohols have a ydroxyl Group, -, bonded to tetrahedral
Chemistry 110 Bettelheim, Brown, Campbell & Farrell Ninth Edition Introduction to General, rganic and Biochemistry Chapter 14 Alcohols, Ethers and Thiols Alcohols have a ydroxyl Group, -, bonded to tetrahedral
