8.4 Changes in Matter: Hot Ice and the Carbon Snake. Grade 8 Activity Plan
|
|
|
- Lambert Price
- 6 years ago
- Views:
Transcription
1 8.4 Changes in Matter: Hot Ice and the Carbon Snake Grade 8 Activity Plan 1
2 Reviews and Updates - Hot Ice and Carbon Snake Activity add by Fola Akpan in July
3 8.4 Changes in Matter: Hot Ice and the Carbon Snake Objectives: 1. To know the science behind, and be acquainted with, natural and artificial occurrences of phase changes. 2. To understand the differences between physical changes and chemical changes 3. To understand combustion, supercooling and supersaturation Keywords/concepts: combustion, phase change, heat, temperature, supercooling, supersaturation Take-home product: A piece of hot ice 3
4 Segment African Proverb and Cultural Relevance (5 min.) Details Don t take another mouthful before you have swallowed what is in your mouth. Madagascar Pre-test (5 min.) Ask probing questions on students knowledge of heat, water, and steam together? Define and illustrate key concepts where applicable. Background (15 min.) What is heat energy, how it is produced, harnessed and used in everyday life. Activity 1 Students will get to see how supersaturation and supercooling can create a reaction that causes Hot Ice Activity 2 Make a carbon snake using sugar and baking soda Post-test (10 min.) Oral Question and answer session Suggested interpretation of proverb: One needs to always have their priorities right. It is not good to rush. Concentrate on what is present and necessary. As seen in the activity, the engine would eject steam before sucking in more water into it. 4
5 BACKGROUND INFORMATION Heat energy is the energy produced by the moving atoms or molecules. Heat is produced from the movement of molecules. The faster the molecules move the more heat is produced. The heat energy created can be used to produce other forms of energy, such as steam energy, kinetic energy or chemical energy. For more information on heat energy see activity 7.7 When heat is applied to a water molecule, the atoms begin to move very rapidly around it. The change of state from a liquid to a gas takes place when the energized, moving molecule of water takes to the air because of its quickly moving electrons. Physical changes of matter are changes in the form of a substance, but not the chemical composition Chemical changes of matter are changes in the chemical composition of matter In the hot ice experiment, the sodium acetate is used to show how supercooling and supersaturation work together. A supersaturated is a solution in which the solute is more than the solvent. In this case the solute is the Sodium Acetate and the solvent is the water. A supercooled solution is a liquid is chilled below its freezing point. This experiment is an example of a physical change and a phase change. In the black carbon snake experiment, the sugar and baking soda demonstrate the concept of combustion. Combustion is the process of burning matter. When the sugar and baking soda in the experiment combusts, they is chemically changed into water vapour, sodium carbonate and carbon dioxide. The pressure created by the carbon dioxide pushes the sodium carbonate out, which creates the black carbon snake. This reaction is referred to as the dehydration of sugar. Both experiments are examples of exothermic reactions, which produce heat. In the hot ice experiment, bond formation causes the exothermic reaction. In the carbon snake experiment, combustion causes the exothermic reaction because energy is released. Additional Information
6 Activity 1- Hot Ice: Sodium Acetate Source: Purpose: To understand the science behind, and be acquainted with, natural and artificial occurrences of phase changes, supersaturation and supercooling Note: It is advised that the mentor prepare and test the solution beforehand Item Quantity (10 students) Baking Soda 3 Tablespoons Vinegar 1 Liter Hot Plate 1 Pot 1 Mason Jar 2 Gloves 10 Procedure: NOTE: Make sure that all of your equipment is clean since other particles can affect the outcome of the solution 1. Measure out 3 tablespoons of Baking Soda and put it into the pot 2. Measure out 1 Liter of Vinegar and pout it slowly into the pot 3. Stir the baking soda and vinegar together until there are no undissolved baking soda particles (NOTE: It is very important that the mixture is completely clear) 4. Boil the solution until the first trace of film appears on the surface of the solution, then take the pot off heat immediately (NOTE: It is very important that the mixture is not overcooked and does not burn but there is a way to fix the solution if a problem does occur) 5. As the water is boiling, scrape off the crystals that form on the side of the pot. These crystals will be used to activate the hot ice 6. Pour the solution into a mason jar and seal the mason jar 7. Chill the solution in the fridge or in an ice water bath until the temperature is slightly below room temperature 8. Place the crystals into the solution. This is when the hot ice starts to form. NOTE: If the hot ice doesn t form, then the solution was made incorrectly. To fix this, add more vinegar to your original solution and redo steps 4-8. Additional Information Contains Hot Ice troubleshooting information 6
7 Activity 2 Carbon Snake Source: Purpose: To understand combustion, exothermic reactions and dehydration reactions Suggested Format: Have the kids work in groups or 2 or three Item Quantity (10 students) Powdered Sugar 1 package Baking Soda 1 package Bowl or Foil Pan 5 100% Ethanol 250 ml Safety Goggles 10 Lighter 1 Sand 20 cups Gloves 11 Procedure: 1. Fill the bowl about 4/5 th with sand 2. Mix together 1 tsp of baking soda and 4 tsp of powdered sugar 3. Make a hole in the sand and put about 50 ml of ethanol into the bowl 4. Place the sugar mixture into the hole in the sand 5. Light it on fire and watch the carbon snake form! 7
Activity Sheet Transferring thermal energy by dissolving salts
 Student Name: Date: Activity Sheet Transferring thermal energy by dissolving salts 1) Define Thermal energy and temperature in the boxes below. Thermal Energy Temperature Practice Experiment: Aim: To practice
Student Name: Date: Activity Sheet Transferring thermal energy by dissolving salts 1) Define Thermal energy and temperature in the boxes below. Thermal Energy Temperature Practice Experiment: Aim: To practice
Chapter 6, Lesson 7: Energy Changes in Chemical Reactions
 Chapter 6, Lesson 7: Energy Changes in Chemical Reactions Key Concepts If two substances react and the temperature of the mixture decreases, the reaction is endothermic. If two substances react and the
Chapter 6, Lesson 7: Energy Changes in Chemical Reactions Key Concepts If two substances react and the temperature of the mixture decreases, the reaction is endothermic. If two substances react and the
SPOOKY SCIENCE GUIDE
 GUIDE TABLE OF CONTENTS PACING GUIDE 2 SCIENCE SAFETY 3 SELF CARVING PUMPKIN 4 GHOST BUBBLES 7 SPOOKY CLOUD 9 TEA BAG GHOST 11 BLOODY HAND PRINT 13 QUIZ 15 SPOOKY SCIENCE GUIDE l 1 PACING GUIDE TEACHER
GUIDE TABLE OF CONTENTS PACING GUIDE 2 SCIENCE SAFETY 3 SELF CARVING PUMPKIN 4 GHOST BUBBLES 7 SPOOKY CLOUD 9 TEA BAG GHOST 11 BLOODY HAND PRINT 13 QUIZ 15 SPOOKY SCIENCE GUIDE l 1 PACING GUIDE TEACHER
What Do You Think? Investigate GOALS. Part A: Freezing Water
 Activity 5 Freezing Water GOALS In this activity you will: Determine the freezing point of water. Show graphically what happens to the temperature as water is cooled to freezing and while it is freezing.
Activity 5 Freezing Water GOALS In this activity you will: Determine the freezing point of water. Show graphically what happens to the temperature as water is cooled to freezing and while it is freezing.
Characteristics of Chemical Change
 Section 2 Characteristics of Chemical Change What Do You See? Learning Outcomes In this section you will Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Section 2 Characteristics of Chemical Change What Do You See? Learning Outcomes In this section you will Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
MIXTURES, COMPOUNDS, & SOLUTIONS
 MIXTURES, COMPOUNDS, & SOLUTIONS As with elements, few compounds are found pure in nature and usually found as mixtures with other compounds. A mixture is a combination of two or more substances that are
MIXTURES, COMPOUNDS, & SOLUTIONS As with elements, few compounds are found pure in nature and usually found as mixtures with other compounds. A mixture is a combination of two or more substances that are
1 st Semester Exam Study Guide 1.) Which of the following is NOT a compound? Explain why. a. H2O b. O2
 1 st Semester Exam Study Guide 1.) Which of the following is NOT a compound? Explain why. a. H2O b. O2 2.) A chemist has discovered what she thinks is a new molecule. In order for it to be a molecule,
1 st Semester Exam Study Guide 1.) Which of the following is NOT a compound? Explain why. a. H2O b. O2 2.) A chemist has discovered what she thinks is a new molecule. In order for it to be a molecule,
ANSWER KEY. Introduction to Matter
 Chapter Project Worksheet 1 1. The object to be massed is placed on one side of the balance. Objects with known masses are placed on the other side of the arm until the balance arm is level. 2. Sample
Chapter Project Worksheet 1 1. The object to be massed is placed on one side of the balance. Objects with known masses are placed on the other side of the arm until the balance arm is level. 2. Sample
What Do You Think? Investigate GOALS
 Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations about the combinations of materials
Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations about the combinations of materials
AP Chemistry Lab #5- Synthesis and Analysis of Alum (Big Idea 1 & 2)
 www.pedersenscience.com AP Chemistry Lab #5- Synthesis and Analysis of Alum (Big Idea 1 & 2) 1.A.1: Molecules are composed of specific combinations of atoms; different molecules are composed of combinations
www.pedersenscience.com AP Chemistry Lab #5- Synthesis and Analysis of Alum (Big Idea 1 & 2) 1.A.1: Molecules are composed of specific combinations of atoms; different molecules are composed of combinations
5 th Grade Lesson Plan: Matter and Chemical Reactions
 5 th Grade Lesson Plan: Matter and Chemical Reactions Objective: Teach students that matter is neither created nor destroyed during a chemical reaction, rather, it is transformed. Identify evidence that
5 th Grade Lesson Plan: Matter and Chemical Reactions Objective: Teach students that matter is neither created nor destroyed during a chemical reaction, rather, it is transformed. Identify evidence that
2. Synthesis of Aspirin
 This is a two-part laboratory experiment. In part one, you will synthesize (make) the active ingredient in aspirin through a reaction involving a catalyst. The resulting product will then be purified through
This is a two-part laboratory experiment. In part one, you will synthesize (make) the active ingredient in aspirin through a reaction involving a catalyst. The resulting product will then be purified through
Physical and Chemical Properties and Changes Lab
 Name: Date: Period: Group Members Physical and Chemical Properties and Changes Lab Station 1 Color Station Instruction: Describe the color of the following substances. Substance Color 1. Sulfur 2. Ammonium
Name: Date: Period: Group Members Physical and Chemical Properties and Changes Lab Station 1 Color Station Instruction: Describe the color of the following substances. Substance Color 1. Sulfur 2. Ammonium
Chemical Reactions. Teachers Guide.
 Chemical Reactions. Teachers Guide. Introduction: Almost everything you will ever see in this universe is made of atoms. Each different type of atom is known as a chemical element there are currently 118
Chemical Reactions. Teachers Guide. Introduction: Almost everything you will ever see in this universe is made of atoms. Each different type of atom is known as a chemical element there are currently 118
Chemical versus Physical Change
 Chemical versus Physical Change Overall Objective: By the end of the session pupils should understand the difference between a physical and chemical change and have a basic understanding of the states
Chemical versus Physical Change Overall Objective: By the end of the session pupils should understand the difference between a physical and chemical change and have a basic understanding of the states
Name: 1. Which of the following is probably true about 300 ml of sand and 300 ml of water?
 Name: Jaguar Review #2 Physical Sciences Benchmark A: Relate uses, properties and chemical processes to the behavior and/or arrangement of the small particles that compose matter. 1. Which of the following
Name: Jaguar Review #2 Physical Sciences Benchmark A: Relate uses, properties and chemical processes to the behavior and/or arrangement of the small particles that compose matter. 1. Which of the following
Moles Lab Activity 2: Elements Copper
 Materials Sample of copper Balance Pre-1982 penny Moles Lab Activity 2: Elements Copper Procedure Take the necessary measurements, and record them with units. Show all your calculations, rounding your
Materials Sample of copper Balance Pre-1982 penny Moles Lab Activity 2: Elements Copper Procedure Take the necessary measurements, and record them with units. Show all your calculations, rounding your
Physical and Chemical Properties of Matter Lab
 Physical and Chemical Properties of Matter Lab Purpose To introduce the student to physical and chemical properties of matter and their use for the identification and separation of compounds. Each student
Physical and Chemical Properties of Matter Lab Purpose To introduce the student to physical and chemical properties of matter and their use for the identification and separation of compounds. Each student
Page 1 / 12. Chemistry Exam. Name: Matter Properties, Structure. Question 1 (1 point) The atomic number of an atom is. A. The mass of the atom.
 Chemistry Exam Matter Properties, Structure Name: Question 1 (1 point) The atomic number of an atom is A. The mass of the atom. B. The number of protons added to the number of neutrons in the nucleus.
Chemistry Exam Matter Properties, Structure Name: Question 1 (1 point) The atomic number of an atom is A. The mass of the atom. B. The number of protons added to the number of neutrons in the nucleus.
Are Chemical Reactions Closed Systems?
 Measuring a Chemical Reaction 1. Place about 100 ml of vinegar in a large plastic cup. Carefully weigh the cup AND the vinegar all together, and write down the total mass. (Don t worry about subtracting
Measuring a Chemical Reaction 1. Place about 100 ml of vinegar in a large plastic cup. Carefully weigh the cup AND the vinegar all together, and write down the total mass. (Don t worry about subtracting
Chapter 2: Properties of Matter Student Outline 2.1 Classifying Matter A. Pure Substances
 Name: Date: Physical Science Period: Chapter 2: Properties of Matter Student Outline GA Performance Standards SPS1. Students will investigate our current understanding of the atom. SPS2. Students will
Name: Date: Physical Science Period: Chapter 2: Properties of Matter Student Outline GA Performance Standards SPS1. Students will investigate our current understanding of the atom. SPS2. Students will
MiSP CHEMICAL REACTIONS, L3 Teacher Guide. Introduction
 MiSP CHEMICAL REACTIONS, L3 Teacher Guide Introduction This weeklong unit should be included with other chemistry content teaching and learning. It is designed to follow Intermediate Level Science Core
MiSP CHEMICAL REACTIONS, L3 Teacher Guide Introduction This weeklong unit should be included with other chemistry content teaching and learning. It is designed to follow Intermediate Level Science Core
Chemical Change. Day 1: 70 minutes; Day 2: minutes
 Chemical Change Lesson Concept When one substance interacts with another substance, a chemical change may occur. There are five indicators that a chemical change has occurred: gas production (bubbles),
Chemical Change Lesson Concept When one substance interacts with another substance, a chemical change may occur. There are five indicators that a chemical change has occurred: gas production (bubbles),
Name: Section: Matter: Atoms and Properties Practice Test
 Name: Section: Matter: Atoms and Properties Practice Test Directions: For each of the questions or incomplete statements below, choose the best of the answer choices given and write your answer on the
Name: Section: Matter: Atoms and Properties Practice Test Directions: For each of the questions or incomplete statements below, choose the best of the answer choices given and write your answer on the
Lesson Plan: Stearic Acid
 Lesson Plan: Stearic Acid Created by: In this lesson, students investigate how stearic acid undergoes a 2014 AACT Middle School phase change from solid to liquid and back from liquid to solid. Content
Lesson Plan: Stearic Acid Created by: In this lesson, students investigate how stearic acid undergoes a 2014 AACT Middle School phase change from solid to liquid and back from liquid to solid. Content
What is Science? Science is both a collection of knowledge and the process for building that knowledge.
 Introduction to Science Junior Science What is Science? Science is both a collection of knowledge and the process for building that knowledge. Science asks questions about the natural world and looks for
Introduction to Science Junior Science What is Science? Science is both a collection of knowledge and the process for building that knowledge. Science asks questions about the natural world and looks for
6.7 Design Your Own Experiment: Factors
 6.7 Design Your Own Experiment: Factors That Affect the Rate of Dissolving Page 158 PRESCRIBED LEARNING OUTCOMES measure substances and solutions according to ph, solubility, and concentration conduct
6.7 Design Your Own Experiment: Factors That Affect the Rate of Dissolving Page 158 PRESCRIBED LEARNING OUTCOMES measure substances and solutions according to ph, solubility, and concentration conduct
Moles Lab Activity 1: PCU (Popcorn Counting Units)
 Moles Lab Activity 1: PCU (Popcorn Counting Units) Materials: A container of each of the following: Popcorn kernels Another type of beans A large unopened bag of popcorn Kernels Balance Safety goggles
Moles Lab Activity 1: PCU (Popcorn Counting Units) Materials: A container of each of the following: Popcorn kernels Another type of beans A large unopened bag of popcorn Kernels Balance Safety goggles
Post-Show HOT AND COLD. Gases. Liquids. Solids. After the Show. Traveling Science Shows
 Traveling Science Shows Post-Show HOT AND COLD After the Show We recently presented a Hot and Cold show at your school, and thought you and your students might like to continue investigating this topic.
Traveling Science Shows Post-Show HOT AND COLD After the Show We recently presented a Hot and Cold show at your school, and thought you and your students might like to continue investigating this topic.
8.2 Solubility and Concentration
 Fresh lemonade is a solution of water, lemon juice, and sugar. There is a limit to the amount of sugar that can dissolve in a given amount of water. Once that limit is reached, you cannot make the solution
Fresh lemonade is a solution of water, lemon juice, and sugar. There is a limit to the amount of sugar that can dissolve in a given amount of water. Once that limit is reached, you cannot make the solution
A simple equation of what happens when you add baking soda to vinegar:
 What s the Matter? Matter is anything that takes up space and has mass. Mass is the stuff that matter is made of, or the amount of particles in a substance or object. Matter has physical and chemical properties
What s the Matter? Matter is anything that takes up space and has mass. Mass is the stuff that matter is made of, or the amount of particles in a substance or object. Matter has physical and chemical properties
Grade 8 Science Unit 2 Test» Form A (Master Copy) Directions: Please choose the best answer choice for each of the following questions.
 Directions: Please choose the best answer choice for each of the following questions. 1. Which of the following could be a molecule BUT not a compound? a pair of atoms of the same element a collection
Directions: Please choose the best answer choice for each of the following questions. 1. Which of the following could be a molecule BUT not a compound? a pair of atoms of the same element a collection
Matter: Chemical and Physical Changes
 PARAMUS ELEMENTARY SCHOOL PARAMUS, NEW JERSEY CURRICULUM OUTLINE DEPARTMENT: Science REVISION DATE: January, 2004 COURSE: Matter: Chemical and Physical Changes GRADE LEVEL: Grade 5 I. COURSE DESCRIPTION:
PARAMUS ELEMENTARY SCHOOL PARAMUS, NEW JERSEY CURRICULUM OUTLINE DEPARTMENT: Science REVISION DATE: January, 2004 COURSE: Matter: Chemical and Physical Changes GRADE LEVEL: Grade 5 I. COURSE DESCRIPTION:
The Next Generation Science Standards (NGSS)
 The Next Generation Science Standards (NGSS) CHAPTER 2, LESSON 1 HEAT, TEMPERATURE, AND CONDUCTION MS-PS1-4. Develop a model that predicts and describes changes in particle motion, temperature, and state
The Next Generation Science Standards (NGSS) CHAPTER 2, LESSON 1 HEAT, TEMPERATURE, AND CONDUCTION MS-PS1-4. Develop a model that predicts and describes changes in particle motion, temperature, and state
Classification of Matter
 Matter in Chemistry Classification of Matter Elements: substances that are made from one type of atom only. Compounds: substances that are made from more than one type of atom chemically bonded together.
Matter in Chemistry Classification of Matter Elements: substances that are made from one type of atom only. Compounds: substances that are made from more than one type of atom chemically bonded together.
Solids, liquids and gases
 Solids, liquids and gases Duration 60 minutes Lesson overview Students share what they know about the three states of matter solid, liquid and gas and consider some of their scientific properties. They
Solids, liquids and gases Duration 60 minutes Lesson overview Students share what they know about the three states of matter solid, liquid and gas and consider some of their scientific properties. They
Experiment 17 It s A Gas and More!
 Energy Energy Experiment 17 It s A Gas and More! OUTCOMES After completing this lab activity, the student should be able to: explain a simple method for distinguishing carbon dioxide gas from oxygen gas.
Energy Energy Experiment 17 It s A Gas and More! OUTCOMES After completing this lab activity, the student should be able to: explain a simple method for distinguishing carbon dioxide gas from oxygen gas.
CHM 130 Physical and Chemical Changes
 Objectives CHM 130 and Changes 1. List physical and chemical properties of substances and explain how they are used in identification and separation of substances. 2. Become familiar with the concepts
Objectives CHM 130 and Changes 1. List physical and chemical properties of substances and explain how they are used in identification and separation of substances. 2. Become familiar with the concepts
Exploring Acids & Bases
 Food Explorations Lab: Exploring Acids & Bases STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will use cabbage juice indicator to determine if two unknown samples are acids or
Food Explorations Lab: Exploring Acids & Bases STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will use cabbage juice indicator to determine if two unknown samples are acids or
KITCHEN CHEMISTRY Identifying acids and bases with red cabbage indicator
 KITCHEN CHEMISTRY Identifying acids and bases with red cabbage indicator By Darby Sloss and Marianne Smith Edited by Anne Starace Abstract Chemistry is an important part of our lives. Kitchen Chemistry
KITCHEN CHEMISTRY Identifying acids and bases with red cabbage indicator By Darby Sloss and Marianne Smith Edited by Anne Starace Abstract Chemistry is an important part of our lives. Kitchen Chemistry
Conservation of Mass in Chemical Reactions Lab. Experiment Question: What happens to the total mass of substances when a chemical reaction occurs?
 Conservation of Mass in Chemical Reactions Lab Name: 5 th Grade PSI Science Score: / 5 Experiment Question: What happens to the total mass of substances when a chemical reaction occurs? Hypothesis Starters:
Conservation of Mass in Chemical Reactions Lab Name: 5 th Grade PSI Science Score: / 5 Experiment Question: What happens to the total mass of substances when a chemical reaction occurs? Hypothesis Starters:
How can homogeneous and heterogeneous mixtures be. 1. classified? 2. separated?
 How can homogeneous and heterogeneous mixtures be 1. classified? 2. separated? 1. HETEROGENEOUS MIXTURE 2. COLLOID 3. EMULSION 4. SUSPENSION 5. FILTRATION 6. TYNDALL EFFECT 7. HOMOGENEOUS MIXTURE 8. SOLUTION
How can homogeneous and heterogeneous mixtures be 1. classified? 2. separated? 1. HETEROGENEOUS MIXTURE 2. COLLOID 3. EMULSION 4. SUSPENSION 5. FILTRATION 6. TYNDALL EFFECT 7. HOMOGENEOUS MIXTURE 8. SOLUTION
W X gas liquid solid Y Z. C X and Y D Y and Z X Y Z. C Z to X D Z to Y
 1 In which changes do the particles move further apart? W X gas liquid solid Y Z W and X W and Z X and Y Y and Z 2 iagrams X, Y and Z represent the three states of matter. X Y Z Which change occurs during
1 In which changes do the particles move further apart? W X gas liquid solid Y Z W and X W and Z X and Y Y and Z 2 iagrams X, Y and Z represent the three states of matter. X Y Z Which change occurs during
Physical and Chemical Changes
 Physical and Chemical Changes Textbook pages 96 105 Section 3.3 Summary Before You Read Cooking an egg is a chemical change. Boiling water is a physical change. What do you think is the difference between
Physical and Chemical Changes Textbook pages 96 105 Section 3.3 Summary Before You Read Cooking an egg is a chemical change. Boiling water is a physical change. What do you think is the difference between
Physical Changes can be observed without changing the identity of the substance (often states of matter changes).
 Physical Changes can be observed without changing the identity of the substance (often states of matter changes). Remember: States of matter changes are physical changes. The identify of the substance
Physical Changes can be observed without changing the identity of the substance (often states of matter changes). Remember: States of matter changes are physical changes. The identify of the substance
Science 14 Unit A: Investigating Properties of Matter Chapter 3 Mixtures and Their Uses pp WORKBOOK Name:
 Science 14 Unit A: Investigating Properties of Matter Chapter 3 Mixtures and Their Uses pp. 40-57 WORKBOOK Name: 3.1 Two Kinds of Mixtures pp. 42-44 Read pp. 42-43 Mixtures are represented on the right
Science 14 Unit A: Investigating Properties of Matter Chapter 3 Mixtures and Their Uses pp. 40-57 WORKBOOK Name: 3.1 Two Kinds of Mixtures pp. 42-44 Read pp. 42-43 Mixtures are represented on the right
What Do You Think? Investigate GOALS
 Activity 1 Chemical and Physical Changes GOALS In this activity you will: Learn to differentiate between chemical and physical changes. Make observations and cite evidence to identify changes as chemical
Activity 1 Chemical and Physical Changes GOALS In this activity you will: Learn to differentiate between chemical and physical changes. Make observations and cite evidence to identify changes as chemical
SOLUTIONS. Heterogeneous Mixtures. Section 8.1: Solutions and Other Mixtures. Heterogeneous Mixtures (cont d) CHAPTER 8.
 Section 8.1: Solutions and Other Mixtures CHAPTER 8 SOLUTIONS Key Questions What is a heterogeneous mixture? What is a homogeneous mixture? Heterogeneous Mixtures The tree of matter branches into two distinct
Section 8.1: Solutions and Other Mixtures CHAPTER 8 SOLUTIONS Key Questions What is a heterogeneous mixture? What is a homogeneous mixture? Heterogeneous Mixtures The tree of matter branches into two distinct
LEVEL ZERO VOICE CATALYST (10 minutes, individual work): 1. Counting atoms
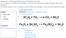 Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
2018 Version. Separating Mixtures Junior Science
 2018 Version Separating Mixtures Junior Science 6a Solutions are made from a solute dissolved in a solvent Solution (saltwater) Solute (salt) Solvent (water) A solution is made up of a solvent and a solute.
2018 Version Separating Mixtures Junior Science 6a Solutions are made from a solute dissolved in a solvent Solution (saltwater) Solute (salt) Solvent (water) A solution is made up of a solvent and a solute.
Lab Activity 3: Factors Affecting Reaction Rate
 Chemistry 3202 Lab #3 factors affecting Reaction Rate Page 1 of 5 Lab Activity 3: Factors Affecting Reaction Rate Introduction Several factors influence how fast a reaction proceeds. In this activity,
Chemistry 3202 Lab #3 factors affecting Reaction Rate Page 1 of 5 Lab Activity 3: Factors Affecting Reaction Rate Introduction Several factors influence how fast a reaction proceeds. In this activity,
Physical Science Review Sheet Matter & Physical Properties
 Name: Date: 1. The four diagrams below model the results of mixing atoms of different substances. Each atom is represented by a different symbol. Which diagram correctly models a chemical change? 4. Base
Name: Date: 1. The four diagrams below model the results of mixing atoms of different substances. Each atom is represented by a different symbol. Which diagram correctly models a chemical change? 4. Base
SNC2D CHEMISTRY 2/23/2013. CHEMICAL REACTIONS L Conservation of Mass (P ) Activity: Measuring Mass (Part 1) Activity: Measuring Mass (Part 1)
 SNC2D CHEMISTRY CHEMICAL REACTIONS L Conservation of Mass (P.176-177) Activity: Measuring Mass (Part 1) INSTRUCTIONS (CLOSED SYSTEM) A. Measure~ 30 ml of vinegar (acetic acid) into a small Erlenmeyer flask.
SNC2D CHEMISTRY CHEMICAL REACTIONS L Conservation of Mass (P.176-177) Activity: Measuring Mass (Part 1) INSTRUCTIONS (CLOSED SYSTEM) A. Measure~ 30 ml of vinegar (acetic acid) into a small Erlenmeyer flask.
CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED)
 CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED) Overview: Students investigate chemical reactions using given substances. Students identify chemical reactions in their daily lives. Objectives: The student
CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED) Overview: Students investigate chemical reactions using given substances. Students identify chemical reactions in their daily lives. Objectives: The student
Unit 4. Compounds & Mixtures
 Unit 4 Compounds & Mixtures Homework for Unit 4 1. Vocab for Unit 4; due: 2. Pg 81 (1-7), Pg 83 (1-7), Pg 85 (1-6) complete sentences; due: 3. Pg 149 (1-4), Pg 169 (1-4), Pg 173 (1-7) complete sentences;
Unit 4 Compounds & Mixtures Homework for Unit 4 1. Vocab for Unit 4; due: 2. Pg 81 (1-7), Pg 83 (1-7), Pg 85 (1-6) complete sentences; due: 3. Pg 149 (1-4), Pg 169 (1-4), Pg 173 (1-7) complete sentences;
Important Examples of Chemical Change
 Important Examples of Chemical Change chemical change: the change that occurs when substances react in a chemical reaction to create different substances When was the last time you watched an awesome display
Important Examples of Chemical Change chemical change: the change that occurs when substances react in a chemical reaction to create different substances When was the last time you watched an awesome display
2-1: Describing Matter. 8 th Grade Physical Sciences
 8 th Grade Physical Sciences What is Matter? Matter is anything that has mass and takes up space. Properties of Matter Matter can be described in many ways; hard, soft, heavy, light, rough, smooth,
8 th Grade Physical Sciences What is Matter? Matter is anything that has mass and takes up space. Properties of Matter Matter can be described in many ways; hard, soft, heavy, light, rough, smooth,
Experimentell kemi Gävle 2017 Part 2
 Experimentell kemi Gävle 2017 Part 2 Lynn Hogue, Lynn@TerrificScience.org Mickey Sarquis, Mickey@TerrificScience.org www.terrificscience.org What do whoopee cushions, potato guns, and exploding straws
Experimentell kemi Gävle 2017 Part 2 Lynn Hogue, Lynn@TerrificScience.org Mickey Sarquis, Mickey@TerrificScience.org www.terrificscience.org What do whoopee cushions, potato guns, and exploding straws
Performance script for sixth graders By Thomas Kuo and Kimberly Kline LEAPS Fellows, University of California, Santa Barbara
 Performance script for sixth graders By Thomas Kuo and Kimberly Kline LEAPS Fellows, 2007-08 University of California, Santa Barbara [Remember to get answers from a wide variety of students in the audience.
Performance script for sixth graders By Thomas Kuo and Kimberly Kline LEAPS Fellows, 2007-08 University of California, Santa Barbara [Remember to get answers from a wide variety of students in the audience.
Stoichiometry: Baking Soda and Vinegar Reactions Teacher Version
 Stoichiometry: Baking Soda and Vinegar Reactions Teacher Version In this lab, students will examine the chemical reaction between baking soda and vinegar, and mix different amounts of these household chemicals
Stoichiometry: Baking Soda and Vinegar Reactions Teacher Version In this lab, students will examine the chemical reaction between baking soda and vinegar, and mix different amounts of these household chemicals
Chemistry: classifying chemical and physical changes in various materials/substances
 Chemistry: classifying chemical and physical changes in various materials/substances Nikki Schilling, Ames, St. Paul Mn Based on original activity from Cool Chemistry Concoctions by Joe Rhatigan and Veronika
Chemistry: classifying chemical and physical changes in various materials/substances Nikki Schilling, Ames, St. Paul Mn Based on original activity from Cool Chemistry Concoctions by Joe Rhatigan and Veronika
Experiment 2 - Using Physical Properties to Identify an Unknown Liquid
 Experiment 2 - Using Physical Properties to Identify an Unknown Liquid We usually think of chemists as scientists who do things with chemicals. We can picture a chemist's laboratory with rows of bottles
Experiment 2 - Using Physical Properties to Identify an Unknown Liquid We usually think of chemists as scientists who do things with chemicals. We can picture a chemist's laboratory with rows of bottles
Introduction to Work in Laboratory
 INSTITUTE OF MEDICAL BIOCHEMISTRY AND LABORATORY MEDICINE Introduction to Work in Measuring volumes, filtration, centrifugation, solubility, separation Practical in Medical Biochemistry General Medicine
INSTITUTE OF MEDICAL BIOCHEMISTRY AND LABORATORY MEDICINE Introduction to Work in Measuring volumes, filtration, centrifugation, solubility, separation Practical in Medical Biochemistry General Medicine
Experiment 15 - Heat of Fusion and Heat of Solution
 Experiment 15 - Heat of Fusion and Heat of Solution Phase changes and dissolving are physical processes that involve heat. In this experiment, you will determine the heat of fusion of ice (the energy required
Experiment 15 - Heat of Fusion and Heat of Solution Phase changes and dissolving are physical processes that involve heat. In this experiment, you will determine the heat of fusion of ice (the energy required
After you are finished, you can collect the salol, which should come off the plastic wrap easily. It can then be melted again and reused.
 IGNEOUS ROCK TEXTURES Teacher Information: Be sure to explain and use all standard laboratory safety practices and procedures. Be sure your students understand how to safely handle the chemicals and materials
IGNEOUS ROCK TEXTURES Teacher Information: Be sure to explain and use all standard laboratory safety practices and procedures. Be sure your students understand how to safely handle the chemicals and materials
6 th Grade Introduction to Chemistry
 Lesson 1 (Describing Matter) 6 th Grade Introduction to Chemistry Matter anything that has mass and takes up space All the stuff in the natural world is matter. Chapter 1: Introduction to Matter Chemistry
Lesson 1 (Describing Matter) 6 th Grade Introduction to Chemistry Matter anything that has mass and takes up space All the stuff in the natural world is matter. Chapter 1: Introduction to Matter Chemistry
5 th GRADE PHYSICAL AND CHEMICAL TESTS
 5 th GRADE PHYSICAL AND CHEMICAL TESTS Summary: Students investigate 5 unknown white powders. They gather clues by observing the physical and chemical changes of the powders. At the end of the activity,
5 th GRADE PHYSICAL AND CHEMICAL TESTS Summary: Students investigate 5 unknown white powders. They gather clues by observing the physical and chemical changes of the powders. At the end of the activity,
Experiment 8 Synthesis of Aspirin
 Experiment 8 Synthesis of Aspirin Aspirin is an effective analgesic (pain reliever), antipyretic (fever reducer) and anti-inflammatory agent and is one of the most widely used non-prescription drugs. The
Experiment 8 Synthesis of Aspirin Aspirin is an effective analgesic (pain reliever), antipyretic (fever reducer) and anti-inflammatory agent and is one of the most widely used non-prescription drugs. The
Safety and Types of Fires
 Cookin' Chem Activity 2 Safety and Types of Fires CHEM POETRY Oliver learned all those Don t s and Do s, The ones that get passed down through moles of O 2 s; Each new generation did soon learn to trust
Cookin' Chem Activity 2 Safety and Types of Fires CHEM POETRY Oliver learned all those Don t s and Do s, The ones that get passed down through moles of O 2 s; Each new generation did soon learn to trust
Copyright 2015 Edmentum - All rights reserved. During which of the following phase changes is there a gain in energy? I.
 Study Island Copyright 2015 Edmentum - All rights reserved. Generation Date: 03/16/2015 Generated By: Kristina Brown 1. Examine the phase-change diagram below. During which of the following phase changes
Study Island Copyright 2015 Edmentum - All rights reserved. Generation Date: 03/16/2015 Generated By: Kristina Brown 1. Examine the phase-change diagram below. During which of the following phase changes
4. Every CHANGE in matter includes a change in, which is conserved in a chemical reaction and. TRANSFORMED from one form to another.
 Part C: Lesson 1.4 - Read pages 20-29 to answer these questions. Add to notes - 1. A physical change alters the form or appearance of matter, such as Change in size, a change in SIZE or. SHAPE shape, or
Part C: Lesson 1.4 - Read pages 20-29 to answer these questions. Add to notes - 1. A physical change alters the form or appearance of matter, such as Change in size, a change in SIZE or. SHAPE shape, or
Section 2: How Substances Dissolve. Preview Key Ideas Bellringer Water: A Common Solvent The Dissolving Process Surface Area
 : How Substances Dissolve Preview Key Ideas Bellringer Water: A Common Solvent The Dissolving Process Surface Area Key Ideas Why is water called the universal solvent? Why do substances dissolve? Bellringer
: How Substances Dissolve Preview Key Ideas Bellringer Water: A Common Solvent The Dissolving Process Surface Area Key Ideas Why is water called the universal solvent? Why do substances dissolve? Bellringer
Pre-Lab Exercises Lab 3: Chemical Properties
 Pre-Lab Exercises Lab 3: Chemical Properties 1. How is a chemical property different from a physical property? Name Date Section 2. How is a chemical change different from a physical change? 3. Give two
Pre-Lab Exercises Lab 3: Chemical Properties 1. How is a chemical property different from a physical property? Name Date Section 2. How is a chemical change different from a physical change? 3. Give two
Bay Area Scientists in Schools Presentation Plan
 Bay Area Scientists in Schools Presentation Plan Lesson Name Presenter(s) Grade Level 3rd Melting, Freezing, and More!: Phase Transitions Steven Scroggins, Ailey Crow, Tom Holcombe, and Terence Choy California
Bay Area Scientists in Schools Presentation Plan Lesson Name Presenter(s) Grade Level 3rd Melting, Freezing, and More!: Phase Transitions Steven Scroggins, Ailey Crow, Tom Holcombe, and Terence Choy California
Section 1: What is a Chemical Reaction
 Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
Cool Chemistry. Middle School TEKS. Vocabulary
 Cool Chemistry Middle School TEKS Sixth Grade: 6.5A, 6.5B, 6.5C, 6.5D Seventh Grade: 7.6A, 7.6B, 7.6C Eighth Grade: 8.5A, 8.5B, 8.5C, 8.5E Vocabulary chemical change, chemical reaction, cirrhosis of the
Cool Chemistry Middle School TEKS Sixth Grade: 6.5A, 6.5B, 6.5C, 6.5D Seventh Grade: 7.6A, 7.6B, 7.6C Eighth Grade: 8.5A, 8.5B, 8.5C, 8.5E Vocabulary chemical change, chemical reaction, cirrhosis of the
Experiment 8 - Chemical Changes
 Experiment 8 - Chemical Changes When a chemical change occurs, the chemicals that you start with are changed into different chemicals. We know when this happens because the new chemicals have different
Experiment 8 - Chemical Changes When a chemical change occurs, the chemicals that you start with are changed into different chemicals. We know when this happens because the new chemicals have different
Solutions CHAPTER Solution Formation. Ch.16 Notes with notations. April 17, 2018
 CHAPTER 16 Solutions 16.1 Solution Formation Solutions can be either solids, liquids, or gases Solutions are homogeneous mixtures that are grouped according to physical state. (mixtures = no bonding) The
CHAPTER 16 Solutions 16.1 Solution Formation Solutions can be either solids, liquids, or gases Solutions are homogeneous mixtures that are grouped according to physical state. (mixtures = no bonding) The
2. What is meant by Chemical State?. 3. Changing states of matter is about changing,,, and other.
 Name: Date: Period: Matter Mania! Online Computer Activity (3 pages) Part I: Go to http://www.chem4kids.com/ and answer the following questions in complete sentences. a. Click on MATTER (written in yellow)
Name: Date: Period: Matter Mania! Online Computer Activity (3 pages) Part I: Go to http://www.chem4kids.com/ and answer the following questions in complete sentences. a. Click on MATTER (written in yellow)
Solution formation. The nature (polarity, or composition) of the solute and the solvent will determine. Factors determining rate of solution...
 Solutions Solution formation The nature (polarity, or composition) of the solute and the solvent will determine 1. Whether a substance will dissolve 2. How much will dissolve Factors determining rate of
Solutions Solution formation The nature (polarity, or composition) of the solute and the solvent will determine 1. Whether a substance will dissolve 2. How much will dissolve Factors determining rate of
TEACHER S NOTES FOR UNIT 1: KAI/OCEAN (SCIENTIFIC METHOD PLUS)
 TEACHER S NOTES FOR UNIT 1: KAI/OCEAN (SCIENTIFIC METHOD PLUS) After introducing/reviewing the scientific method (see 2 handouts), try this sequence of lessons to allow students to experiment themselves,
TEACHER S NOTES FOR UNIT 1: KAI/OCEAN (SCIENTIFIC METHOD PLUS) After introducing/reviewing the scientific method (see 2 handouts), try this sequence of lessons to allow students to experiment themselves,
8.2 Solubility and Concentration
 Fresh lemonade is a solution of water, lemon juice, and sugar. There is a limit to the amount of sugar that can dissolve in a given amount of water. Once that limit is reached, you cannot make the solution
Fresh lemonade is a solution of water, lemon juice, and sugar. There is a limit to the amount of sugar that can dissolve in a given amount of water. Once that limit is reached, you cannot make the solution
AP Chemistry: Designing an Effective Hand Warmer Student Guide INTRODUCTION
 AP Chemistry: Designing an Effective Hand Warmer Student Guide INTRODUCTION AP and the Advanced Placement Program are registered trademarks of the College Entrance Examination Board. The activity and materials
AP Chemistry: Designing an Effective Hand Warmer Student Guide INTRODUCTION AP and the Advanced Placement Program are registered trademarks of the College Entrance Examination Board. The activity and materials
Final Review Graphs and Charts TWO Page 1 of 35
 TEST NAME:Final Review Graphs and Charts TWO TEST ID:1086907 GRADE:11 Eleventh Grade 12 Twelfth Grade SUBJECT:Life and Physical Sciences TEST CATEGORY:School Assessment Final Review Graphs and Charts TWO
TEST NAME:Final Review Graphs and Charts TWO TEST ID:1086907 GRADE:11 Eleventh Grade 12 Twelfth Grade SUBJECT:Life and Physical Sciences TEST CATEGORY:School Assessment Final Review Graphs and Charts TWO
Student Notes. Chemical Reactions LINK
 LCPS Core Experience Chemical Reactions Student Notes OBJECTIVES Students will: investigate the relationship between reactants and products. investigate an exothermic reaction. investigate an endothermic
LCPS Core Experience Chemical Reactions Student Notes OBJECTIVES Students will: investigate the relationship between reactants and products. investigate an exothermic reaction. investigate an endothermic
Name: Block: Date: Student Notes. OBJECTIVE Students will investigate the relationship between temperature and the change of the state of matter.
 Name: Block: Date: LCPS Core Experience Heat Transfer Student Notes OBJECTIVE Students will investigate the relationship between temperature and the change of the state of matter. LINK 1. Particles in
Name: Block: Date: LCPS Core Experience Heat Transfer Student Notes OBJECTIVE Students will investigate the relationship between temperature and the change of the state of matter. LINK 1. Particles in
Elements, Compounds, and Mixtures. Matter: Properties and Changes
 Elements, Compounds, and Mixtures Matter: Properties and Changes Warm up Observe the pictures and use the following terms to name them: element, compound, or mixture. Explain your answer. Warm up answers
Elements, Compounds, and Mixtures Matter: Properties and Changes Warm up Observe the pictures and use the following terms to name them: element, compound, or mixture. Explain your answer. Warm up answers
2 How Substances Dissolve
 CHAPTER 8 SECTION Solutions 2 How Substances Dissolve KEY IDEAS As you read this section, keep these questions in mind: Why is water called the universal solvent? How do substances dissolve? Why Do Substances
CHAPTER 8 SECTION Solutions 2 How Substances Dissolve KEY IDEAS As you read this section, keep these questions in mind: Why is water called the universal solvent? How do substances dissolve? Why Do Substances
Name: Class: Date: Topics in Chemistry Final Exam Study Guide
 Name: Class: _ Date: _ Topics in Chemistry Final Exam Study Guide Multiple Choice Identify the choice that best completes the statement or answers the question. 1. If the force applied to an object increases,
Name: Class: _ Date: _ Topics in Chemistry Final Exam Study Guide Multiple Choice Identify the choice that best completes the statement or answers the question. 1. If the force applied to an object increases,
100 C = 100 X = X = 218 g will fit in this solution. 25 C = 100 X = 3640 X = 36.4 g will fit in this solution.
 58 Questions for Solutions - You should be able to do ALL of these problems. Use a calculator, write all formulas, watch SF, and find the answers online at Arbuiso.com on the SOLUTIONS page. This is great
58 Questions for Solutions - You should be able to do ALL of these problems. Use a calculator, write all formulas, watch SF, and find the answers online at Arbuiso.com on the SOLUTIONS page. This is great
Physical Science SPS 6 Review Activity
 Properties of Acids & Bases: in this table, list everything that you know about acids, bases, & their properties. List specific things and general things. (how do you identify them? What makes them, them?
Properties of Acids & Bases: in this table, list everything that you know about acids, bases, & their properties. List specific things and general things. (how do you identify them? What makes them, them?
Post-Show. Chemistry. Periodic Table of the Elements. After the Show. Traveling Science Shows
 Traveling Science Shows Post-Show Chemistry After the Show We recently presented a Chemistry show at your school, and thought you and your students might like to continue investigating this topic. The
Traveling Science Shows Post-Show Chemistry After the Show We recently presented a Chemistry show at your school, and thought you and your students might like to continue investigating this topic. The
Physical Changes vs Chemical Changes Lab
 Physical Changes vs Chemical Changes Lab What was done? What can you observe? Is it a physical or chemical change and why? What else have you learned during the demonstration? #1) Crumpling Paper Notice
Physical Changes vs Chemical Changes Lab What was done? What can you observe? Is it a physical or chemical change and why? What else have you learned during the demonstration? #1) Crumpling Paper Notice
Chemical Energy Conversions. Vanderbilt Student Volunteers for Science VINSE/VSVS Rural
 Chemical Energy Conversions Vanderbilt Student Volunteers for Science 2018-2019 VINSE/VSVS Rural Important!!! Please use this resource to reinforce your understanding of the lesson! Make sure you have
Chemical Energy Conversions Vanderbilt Student Volunteers for Science 2018-2019 VINSE/VSVS Rural Important!!! Please use this resource to reinforce your understanding of the lesson! Make sure you have
LESSON 1. Chemical Reactions. Fireflies, also called lightning bugs, are small insects that generate their own light using chemical reactions.
 LESSON 1 Chemical Reactions Fireflies, also called lightning bugs, are small insects that generate their own light using chemical reactions. By the end of this lesson... you will be able to explain ways
LESSON 1 Chemical Reactions Fireflies, also called lightning bugs, are small insects that generate their own light using chemical reactions. By the end of this lesson... you will be able to explain ways
Chemistry Unit Test 1 Review
 Chemistry Unit Test 1 Review Name: S8P1a. Students will distinguish between atoms and molecules. 1. Which of the following particles combine to form molecules? a. Atoms b. Protons c. Electrons d. Compounds
Chemistry Unit Test 1 Review Name: S8P1a. Students will distinguish between atoms and molecules. 1. Which of the following particles combine to form molecules? a. Atoms b. Protons c. Electrons d. Compounds
Alternative Reaction Pathways
 Section 1 Energy and Entropy: Alternative Reaction Pathways What Do You See? Learning Outcomes In this section you will Apply the engineering-design process to scientific and everyday situations. Generate
Section 1 Energy and Entropy: Alternative Reaction Pathways What Do You See? Learning Outcomes In this section you will Apply the engineering-design process to scientific and everyday situations. Generate
More Chemical Changes
 Activity 2 More Chemical Changes Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Activity 2 More Chemical Changes Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
CHM101 Lab - Solutions Grading Rubric
 Spring 2017 Name Team Name CHM101 Lab - Solutions Grading Rubric To participate in this lab you must have splash- proof goggles, proper shoes and attire. Criteria Points possible Points earned Lab Performance
Spring 2017 Name Team Name CHM101 Lab - Solutions Grading Rubric To participate in this lab you must have splash- proof goggles, proper shoes and attire. Criteria Points possible Points earned Lab Performance
PROPERTIES OF MATTER STATION 1. Where did the water droplets on the outside of this cup come from? What phase change is this?
 PROPERTIES OF MATTER STATION 1 Where did the water droplets on the outside of this cup come from? What phase change is this? PROPERTIES OF MATTER STATION 2 Make a copy of this graph on your paper. Label
PROPERTIES OF MATTER STATION 1 Where did the water droplets on the outside of this cup come from? What phase change is this? PROPERTIES OF MATTER STATION 2 Make a copy of this graph on your paper. Label
