(CH 3 ) 3 COH. CH 3 ONa
|
|
|
- Roy Byrd
- 6 years ago
- Views:
Transcription
1 1. Rank the following compounds in the trend requested. (15 points each) a. Rank by nucleophilicity. The strongest nucleophile is 1, while the weakest nucleophile is 5. C 3 PNa (C 3 ) 3 C C 3 Na C 3 C 2 Na C 3 NNa b. Rank by bond dissociation energy. Consider the bond dissociation energy for the C- bond indicated. The C- bond with the lowest bond dissociation energy (bond requiring the least amount of energy to break) is 1, while the C- bond with the highest bond dissociation energy is c. Rank by N 2 reactivity. Consider the rate of N 2 reaction for each substrate. The substrate that reacts the fastest in a N 2 reaction is 1, while the substrate that reacts the slowest is 5. I
2 2.(12) There are four isomers (A-D) that contain a benzene group with a methyl group attached para to a C 4 9 substituent. Para refers to when two substituents are placed 1,4 relative to each other on a benzene ring (similar to the structure shown). The C 4 9 substituent can have four different constitutional isomers thus yielding the four isomers A-D. In order to distinguish the four isomers a chlorination reaction ( 2, hν) was performed on each and the number of products obtained for monochlorination for each compound was determined. Compound A yielded 2 monochlorinated products, compound B yielded 4 monochlorinated products, and compounds C and D both yielded 5 monochlorinated products. To differentiate compounds C and D, it was determined that compound C has two 2 hydrogens, while compound D has six 2 hydrogens. Draw the structures for compounds A-D in the appropriate space. C 4 9 Compound A Compound B Compound C Compound D b.(12) Indicate the monobrominated product obtained if compounds A-D are reacted with 2 and hν c.(4) d.(4) Which compound (A-D) reacts the fastest in a bromination? C, generates 3 radical in resonance Which compound (A-D) reacts the slowest in a bromination? A, generates 1 radical in resonance
3 3. Indicate the major product for the following reactions. (7 points each) a. C 3 Na E2, strong base with 3 alkyl halide b. I Na N 2, intramolecular reaction c. C 3 K C 3 C 3 N 1, 3 alkyl halide with no adjacent hydrogens for elimination d. C 3 Na C 3 N 2, 2 alkyl halide with good nucleophile/weak base e. 2 obtain most stable 3 radical in resonance f. C 3 Na E2, strong base with 2 alkyl halide need anticoplanar elimination
4 g. C 3 C 3 Na 3 C C 3 C 3 C 3 N 2, good nucleophile/weak base h. C 3 AgN 3 C 3 N 1, rearrange carbocation i. 1) 2, 2) NaCN NC N 2 after halogenation j. Na N 2, good nucleophile/weak base k. C 3 Na E2, strong base with 2 adjacent to quarternary, need anticoplanar l. NaN 3 N 3 F F N 2, react at 2 bromide
5 4. Consider the following two-step reaction sequence. The first step is a bromination reaction, while the second step is a N 2 reaction. Answer the following rate questions by simply writing either faster, slower, or no change for each part. 2 NaC C 3 3 C 3 A B C a.(8) What would happen to the rate of each step if propane (A) is changed to 2- methylpropane? tep 1: tep 2: faster, obtain 3 radical in halogenation slower, N 2 reaction does not occur at 3 site b.(8) tep 1: tep 2: What would happen to the rate of each step if propane (A) is changed to ethane? slower, 1 radical obtained in halogenation faster, N 2 reactions are faster with less steric hindrance c.(8) What would happen to the rate of each step if 2 is changed to I 2? tep 1: tep 2: slower, iodination is slower than bromination faster, iodine is better leaving group in N 2 than bromine d.(5) What would happen to the rate of the second step if the solvent is changed from methanol (C 3 ) to acetonitrile (C 3 CN)? The rate would be FATER. Acetonitrile is an aprotic solvent, which will not hydrogen bond with nucleophile in N 2 reaction.
6 5. When an alcohol is reacted with bromide a different product is obtained depending upon whether acidic () or basic (Na) conditions are used. No reaction occurs under one set of conditions but a substitution occurs under the other. a.(5) Which conditions will allow a substitution to occur? (write acidic or basic) ACIDIC b.(5) What is the leaving group under this set of conditions? Water ( 2 ) Need to protonate alcohol first to make a good leaving group (hydroxide is a PR leaving group for N 2 reaction. c. In order to convert an alcohol into a better leaving group, the group can be reacted with a tosyl chloride to yield what is called a tosylate compound as shown. R Et 3 N Which of these compounds will react with potassium ethoxide with the fastest rate in a N 2 reaction? (Circle the substrate that would react the fastest) (5) R d.(12) The experiments shown below were carried out in 1935 as a way to understand the mechanism of the N 2 reaction. Indicate the product obtained under each set of conditions. 1) Et 3 N 2) C 3 C 2 K 1) Na 2) C 3 C 2
7 6. The cis and trans isomers of 1-bromo-4-tert-butylcyclohexane react at different rates with KC(C 3 ) 3 to yield the same mixture of enantiomers A and B. ( 3 C) 3 C ( 3 C) 3 C cis-1-bromo-4-tert-butylcyclohexane trans-1-bromo-4-tert-butylcyclohexane a.(8) Draw the structures of compounds A and B. ( 3 C) 3 C ( 3 C) 3 C Compound A Compound B b.(5) faster) c.(5) Which isomer (cis or trans) reacts faster with KC(C 3 ) 3? (circle the isomer that reacts CI TRAN Explain WY either cis or trans reacts faster. In the cis isomer, the bromine is in an axial position in the lowest energy conformation to allow the anticoplanar elimination to occur. d.(10) When NaC 3 is reacted with cis-1-bromo-4-tert-butylcyclohexane the major product is C, whereas the major product of NaC 3 reacting with trans-1-bromo-4-tert-butylcyclohexane is D. Draw the structures of compounds C and D. ( 3 C) 3 C C 3 ( 3 C) 3 C C 3 Compound C Compound D btain inversion of configuration during N 2 reaction. e.(5) Which isomer (cis or trans) reacts faster with NaC 3? CI TRAN
CHE1502. Tutorial letter 201/1/2016. General Chemistry 1B. Semester 1. Department of Chemistry CHE1502/201/1/2016
 CE1502/201/1/2016 Tutorial letter 201/1/2016 General Chemistry 1B CE1502 Semester 1 Department of Chemistry This tutorial letter contains the answers to the questions in assignment 1. FIRST SEMESTER: KEY
CE1502/201/1/2016 Tutorial letter 201/1/2016 General Chemistry 1B CE1502 Semester 1 Department of Chemistry This tutorial letter contains the answers to the questions in assignment 1. FIRST SEMESTER: KEY
1. When methane is photochlorinated a small amount of ethane is found in the product. Give a full mechanism to account for presence of the ethane.
 Chemistry 51 DS Quiz 2 1. When methane is photochlorinated a small amount of ethane is found in the product. Give a full mechanism to account for presence of the ethane. 2. When 2,3-dimethylbutane is monochlorinated
Chemistry 51 DS Quiz 2 1. When methane is photochlorinated a small amount of ethane is found in the product. Give a full mechanism to account for presence of the ethane. 2. When 2,3-dimethylbutane is monochlorinated
Chapter 5. Nucleophilic aliphatic substitution mechanism. by G.DEEPA
 Chapter 5 Nucleophilic aliphatic substitution mechanism by G.DEEPA 1 Introduction The polarity of a carbon halogen bond leads to the carbon having a partial positive charge In alkyl halides this polarity
Chapter 5 Nucleophilic aliphatic substitution mechanism by G.DEEPA 1 Introduction The polarity of a carbon halogen bond leads to the carbon having a partial positive charge In alkyl halides this polarity
CHE1502. Tutorial letter 203/1/2016. General Chemistry 1B. Semester 1. Department of Chemistry
 E1502/203/1/2016 Tutorial letter 203/1/2016 General hemistry 1B E1502 Semester 1 Department of hemistry This tutorial letter contains the answers to the questions in assignment 3. FIRST SEMESTER: KEY T
E1502/203/1/2016 Tutorial letter 203/1/2016 General hemistry 1B E1502 Semester 1 Department of hemistry This tutorial letter contains the answers to the questions in assignment 3. FIRST SEMESTER: KEY T
Chemistry 210 Organic Chemistry I Fall Semester 2000 Dr. Rainer Glaser
 hemistry 210 rganic hemistry I Fall Semester 2000 Dr. Rainer Glaser Examination #2 Alkyl alides: Their Synthesis by alogenation of Alkanes and Their Nucleophilic Substitution Reactions. Friday, ctober
hemistry 210 rganic hemistry I Fall Semester 2000 Dr. Rainer Glaser Examination #2 Alkyl alides: Their Synthesis by alogenation of Alkanes and Their Nucleophilic Substitution Reactions. Friday, ctober
CHEMISTRY 341. Final Exam Tuesday, December 16, Problem 1 15 pts Problem 9 8 pts. Problem 2 5 pts Problem pts
 CEMISTRY 341 Final Exam Tuesday, December 16, 1997 Name NAID Problem 1 15 pts Problem 9 8 pts Problem 2 5 pts Problem 10 21 pts Problem 3 26 pts Problem 11 15 pts Problem 4 10 pts Problem 12 6 pts Problem
CEMISTRY 341 Final Exam Tuesday, December 16, 1997 Name NAID Problem 1 15 pts Problem 9 8 pts Problem 2 5 pts Problem 10 21 pts Problem 3 26 pts Problem 11 15 pts Problem 4 10 pts Problem 12 6 pts Problem
REACTIONS OF HALOALKANES - SUBSTITUTION AND ELIMINATION
 REACTIONS OF HALOALKANES - SUBSTITUTION AND ELIMINATION Haloalkanes (also known as halogenoalkanes and alkyl halides) are organic compounds where one of the hydrogens of an alkane or cycloalkane has been
REACTIONS OF HALOALKANES - SUBSTITUTION AND ELIMINATION Haloalkanes (also known as halogenoalkanes and alkyl halides) are organic compounds where one of the hydrogens of an alkane or cycloalkane has been
Chem 232. Problem Set 9. Question 1. D. J. Wardrop
 Chem 232 D. J. Wardrop wardropd@uic.edu Problem Set 9 Question 1. a. Rank in order of increasing number of chirality centers (1 = fewest chirality centers; 5 = most chirality). 3 C C 3 b. Rank in order
Chem 232 D. J. Wardrop wardropd@uic.edu Problem Set 9 Question 1. a. Rank in order of increasing number of chirality centers (1 = fewest chirality centers; 5 = most chirality). 3 C C 3 b. Rank in order
CHEMISTRY MIDTERM # 3 March 31, The total number of points in this midterm is 100. The total exam time is 120 min (2 h). Good luck!
 Name: CEMISTRY 1-01 MIDTERM # March 1, 2009 The total number of points in this midterm is 100. The total exam time is 120 min (2 h). Good luck! 1. (11 pts) Mark as true (T) false (F) the following statements.
Name: CEMISTRY 1-01 MIDTERM # March 1, 2009 The total number of points in this midterm is 100. The total exam time is 120 min (2 h). Good luck! 1. (11 pts) Mark as true (T) false (F) the following statements.
CHEM 2312 practice final. Version - II
 EM 2312 practice final Version - II The following standardized final examination covers the entire introductory year of organic chemistry (EM 2311 and 2312 at Georgia Tech). The exam consists of 70 multiple
EM 2312 practice final Version - II The following standardized final examination covers the entire introductory year of organic chemistry (EM 2311 and 2312 at Georgia Tech). The exam consists of 70 multiple
Homework problems Chapters 6 and Give the curved-arrow formalism for the following reaction: CH 3 OH + H 2 C CH +
 omework problems hapters 6 and 7 1. Give the curved-arrow formalism for the following reaction: : 3 - : 2 : 3 2-3 3 2. In each of the following sets, arrange the compounds in order of decreasing pka and
omework problems hapters 6 and 7 1. Give the curved-arrow formalism for the following reaction: : 3 - : 2 : 3 2-3 3 2. In each of the following sets, arrange the compounds in order of decreasing pka and
Chapter 5. 3-Chloro-2-methylpentane. Cl 2. 2-Chloro-2-methylpentane. 1-Chloro-2-methylpentane. Cl 2-Chloro-4-methylpentane. 1-Chloro-4-methylpentane
 hapter 5 5.1 lassify each of the following reactions as an addition, elimination, substitution, or rearrangement: (a) 3Br K 3 KBr (b) 3 2 2 2 2 (c) 2 2 2 3 3 a. substitution b. elimination c. addition
hapter 5 5.1 lassify each of the following reactions as an addition, elimination, substitution, or rearrangement: (a) 3Br K 3 KBr (b) 3 2 2 2 2 (c) 2 2 2 3 3 a. substitution b. elimination c. addition
Chapter 6 Ionic Reactions-Nucleophilic Substitution and Elimination Reactions of Alkyl Halides"
 Chapter 6 Ionic Reactions-Nucleophilic Substitution and Elimination Reactions of Alkyl Halides" t Introduction" The polarity of a carbon-halogen bond leads to the carbon having a partial positive charge"
Chapter 6 Ionic Reactions-Nucleophilic Substitution and Elimination Reactions of Alkyl Halides" t Introduction" The polarity of a carbon-halogen bond leads to the carbon having a partial positive charge"
Elimination Reactions. Chapter 6 1
 Elimination Reactions Chapter 6 1 E1 Mechanism Step 1: halide ion leaves, forming a carbocation. Step 2: Base abstracts H + from adjacent carbon forming the double bond. Chapter 6 2 E1 Energy Diagram E1:
Elimination Reactions Chapter 6 1 E1 Mechanism Step 1: halide ion leaves, forming a carbocation. Step 2: Base abstracts H + from adjacent carbon forming the double bond. Chapter 6 2 E1 Energy Diagram E1:
C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2
 C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2 CHM 321: Summary of Important Concepts Concepts for Chapter 7: Substitution Reactions I. Nomenclature of
C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2 CHM 321: Summary of Important Concepts Concepts for Chapter 7: Substitution Reactions I. Nomenclature of
CHAPTER 7. Further Reactions of Haloalkanes: Unimolecular Substitution and Pathways of Elimination
 CHAPTER 7 Further Reactions of Haloalkanes: Unimolecular Substitution and Pathways of Elimination 7-1 Solvolysis of Tertiary and Secondary Haloalkanes The rate of S N 2 reactions decrease dramatically
CHAPTER 7 Further Reactions of Haloalkanes: Unimolecular Substitution and Pathways of Elimination 7-1 Solvolysis of Tertiary and Secondary Haloalkanes The rate of S N 2 reactions decrease dramatically
Chapter 7 Alkenes; Elimination Reactions
 hapter 7 Alkenes; Elimination Reactions Alkenes Alkenes contain a carbon-carbon double bond. They are named as derivatives of alkanes with the suffix -ane changed to -ene. The parent alkane is the longest
hapter 7 Alkenes; Elimination Reactions Alkenes Alkenes contain a carbon-carbon double bond. They are named as derivatives of alkanes with the suffix -ane changed to -ene. The parent alkane is the longest
Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions
 Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions Halogen compounds are important for several reasons. Simple alkyl and aryl halides, especially chlorides and bromides, are versatile
Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions Halogen compounds are important for several reasons. Simple alkyl and aryl halides, especially chlorides and bromides, are versatile
Homework - Review of Chem 2310
 omework - Review of Chem 2310 Chapter 1 - Atoms and Molecules Name 1. What is organic chemistry? 2. Why is there an entire one year course devoted to the study of organic compounds? 3. Give 4 examples
omework - Review of Chem 2310 Chapter 1 - Atoms and Molecules Name 1. What is organic chemistry? 2. Why is there an entire one year course devoted to the study of organic compounds? 3. Give 4 examples
Lecture 21 Organic Chemistry 1
 CEM 232 Organic Chemistry I at Chicago Lecture 21 Organic Chemistry 1 Professor Duncan Wardrop March 30, 2010 1 Self Test Question Ethanolysis of alkyl halide 1 gives ether 2 as one product; however, several
CEM 232 Organic Chemistry I at Chicago Lecture 21 Organic Chemistry 1 Professor Duncan Wardrop March 30, 2010 1 Self Test Question Ethanolysis of alkyl halide 1 gives ether 2 as one product; however, several
PRACTICE PROBLEMS UNIT 8
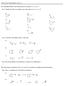 PRACTICE PROBLEMS UNIT 8 8A. Identify halides and carbocations as being 1 o, 2 o, or 3 o 8A.1 assify the following halides and carbocations as 1 o, 2 o, or 3 o. 8A.2 Circle the most stable cation in each
PRACTICE PROBLEMS UNIT 8 8A. Identify halides and carbocations as being 1 o, 2 o, or 3 o 8A.1 assify the following halides and carbocations as 1 o, 2 o, or 3 o. 8A.2 Circle the most stable cation in each
Part I. Multiple choice. (4 points each.) Choose the one best answer and mark your answer on the ScanTron sheet.
 Test 3 Chemistry 2321 April 25, 2003 McMurry, Chapters 9-11 103 Total Points Name Please Print Part I. Multiple choice. (4 points each.) Choose the one best answer and mark your answer on the ScanTron
Test 3 Chemistry 2321 April 25, 2003 McMurry, Chapters 9-11 103 Total Points Name Please Print Part I. Multiple choice. (4 points each.) Choose the one best answer and mark your answer on the ScanTron
Allylic and Benzylic Reactivity
 17 17 Allylic and Benzylic Reactivity An allylic group is a group on a carbon adjacent to a double bond. A benzylic group is a group on a carbon adjacent to a benzene ring or substituted benzene ring.
17 17 Allylic and Benzylic Reactivity An allylic group is a group on a carbon adjacent to a double bond. A benzylic group is a group on a carbon adjacent to a benzene ring or substituted benzene ring.
HALOALKANES. Structure Contain the functional group C-X where X is a halogen (F,Cl,Br or I)
 aloalkanes AS3 1 ALOALKANES Structure ontain the functional group X where X is a halogen (F,l, or I) Types aloalkanes halogen is attached to an aliphatic skeleton alkyl group aloarenes halogen is attached
aloalkanes AS3 1 ALOALKANES Structure ontain the functional group X where X is a halogen (F,l, or I) Types aloalkanes halogen is attached to an aliphatic skeleton alkyl group aloarenes halogen is attached
Organic Reactions Susbstitution S N. Dr. Sapna Gupta
 Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. A) I B) II C) III D) IV E) V
 Practice Questions : Chem 226 / Exam 3 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following statements about benzene is correct?
Practice Questions : Chem 226 / Exam 3 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following statements about benzene is correct?
B. (2 pts) The regiochemistry of alcohol dehydration is determined by the: a. Zaitsev rule; b. Hammond postulate;
 CEMISTRY 313-01 MIDTERM # answer key ctober 1, 008 Statistics: Average: 73 pts (73%); ighest: 98 pts (98%); Lowest: 3 pts (3%) Number of students performing at or above average: 36 (58%) Number of students
CEMISTRY 313-01 MIDTERM # answer key ctober 1, 008 Statistics: Average: 73 pts (73%); ighest: 98 pts (98%); Lowest: 3 pts (3%) Number of students performing at or above average: 36 (58%) Number of students
1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound?
 EM 331: hapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N 2 N 2 N 1 2 3 4 2. What hybrid orbitals are used to
EM 331: hapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N 2 N 2 N 1 2 3 4 2. What hybrid orbitals are used to
Chapter 8: Alkene Structure and Preparation via Elimination Reactions
 Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C 3 C C 3 bond length bond strength 2 C C 2 a C=C double bond is stronger than a C C single
Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C 3 C C 3 bond length bond strength 2 C C 2 a C=C double bond is stronger than a C C single
REACTIONS OF AROMATIC COMPOUNDS
 A STUDENT SHOULD BE ABLE TO: REACTIONS OF AROMATIC COMPOUNDS 1. Predict the product(s) of Electrophilic aromatic substitution (EAS): halogenation, sulfonation, nitration, Friedel- Crafts alkylation and
A STUDENT SHOULD BE ABLE TO: REACTIONS OF AROMATIC COMPOUNDS 1. Predict the product(s) of Electrophilic aromatic substitution (EAS): halogenation, sulfonation, nitration, Friedel- Crafts alkylation and
CHE 275 NUCLEOPHILIC SUBSTITUTUION CHAP 8 ASSIGN. 1. Which best depicts the partial charges on methyl bromide and sodium methoxide?
 CHE 275 NUCLEPHILIC SUBSTITUTUIN CHAP 8 ASSIGN 1. Which best depicts the partial charges on methyl bromide and sodium methoxide? 2. Which of the following would be the best (most reactive) nucleophile
CHE 275 NUCLEPHILIC SUBSTITUTUIN CHAP 8 ASSIGN 1. Which best depicts the partial charges on methyl bromide and sodium methoxide? 2. Which of the following would be the best (most reactive) nucleophile
The Electrophile. S N 2 and E2 least stable most stable least hindered most hindered. S N 1 and E1. > x > >
 The Electrophile 1 Recall that electrophile means electron- loving. When considering substitution and elimination reactions we must consider the carbon attached to the leaving group. Is it a primary, secondary,
The Electrophile 1 Recall that electrophile means electron- loving. When considering substitution and elimination reactions we must consider the carbon attached to the leaving group. Is it a primary, secondary,
Elimination Reactions Heating an alkyl halide with a strong base causes elimination of a. molecule of HX
 Elimination eactions eating an alkyl halide with a strong base causes elimination of a molecule of X 1. Potassium hydroxide dissolved in ethanol and the sodium salts of alcohols (such as sodium ethoxide)
Elimination eactions eating an alkyl halide with a strong base causes elimination of a molecule of X 1. Potassium hydroxide dissolved in ethanol and the sodium salts of alcohols (such as sodium ethoxide)
Organic Chemistry CHM 314 Dr. Laurie S. Starkey, Cal Poly Pomona Alkyl Halides: Substitution Reactions - Chapter 6 (Wade)
 rganic Chemistry CM 314 Dr. Laurie S. Starkey, Cal Poly Pomona Alkyl alides: Substitution Reactions - Chapter 6 (Wade) Chapter utline I. Intro to RX (6-1 - 6-7) II. Substitution Reactions A) S N 2 (6-8,
rganic Chemistry CM 314 Dr. Laurie S. Starkey, Cal Poly Pomona Alkyl alides: Substitution Reactions - Chapter 6 (Wade) Chapter utline I. Intro to RX (6-1 - 6-7) II. Substitution Reactions A) S N 2 (6-8,
Chemistry 2321 Last name, First name (please print) April 22, 2008 McMurry, Chapters 10 and 11 Row # Seat #
 Test 4 Name Chemistry 2321 Last name, First name (please print) April 22, 2008 McMurry, Chapters 10 and 11 Row # Seat # Part 1. Multiple Choice. Choose the one best answer, and clearly mark that answer
Test 4 Name Chemistry 2321 Last name, First name (please print) April 22, 2008 McMurry, Chapters 10 and 11 Row # Seat # Part 1. Multiple Choice. Choose the one best answer, and clearly mark that answer
CHEM Chapter 16. Chemistry of Benzene: Electrophilic Aromatic Substitution (homework) W
 CHEM 2425. Chapter 16. Chemistry of Benzene: Electrophilic Aromatic Substitution (homework) W Short Answer Exhibit 16-1 MATCH a structure or term from the following list with each description below. Place
CHEM 2425. Chapter 16. Chemistry of Benzene: Electrophilic Aromatic Substitution (homework) W Short Answer Exhibit 16-1 MATCH a structure or term from the following list with each description below. Place
CHEM 241 ALCOHOLS AND ALKYL HALIDES CHAP 5 ASSIGN
 CEM 241 ALCOOLS AND ALKYL ALIDES CAP 5 ASSIGN 1. What is the IUPAC name of the compound below? A. 3-isobutyl-2-hexanol B. 2-methyl-5-propyl-6-heptanol C. 2-methyl-5-(1-hydroxyethyl)octane D. 6-methyl-3-propyl-2-heptanol
CEM 241 ALCOOLS AND ALKYL ALIDES CAP 5 ASSIGN 1. What is the IUPAC name of the compound below? A. 3-isobutyl-2-hexanol B. 2-methyl-5-propyl-6-heptanol C. 2-methyl-5-(1-hydroxyethyl)octane D. 6-methyl-3-propyl-2-heptanol
CHEM Exam 2 October 25, 2007
 CEM 3311-200 Exam 2 October 25, 2007 By printing my name below, I pledge that "On my honor, as a University of Colorado at Boulder student, I have neither given nor received unauthorized assistance on
CEM 3311-200 Exam 2 October 25, 2007 By printing my name below, I pledge that "On my honor, as a University of Colorado at Boulder student, I have neither given nor received unauthorized assistance on
Chapter 8. Substitution reactions of Alkyl Halides
 Chapter 8. Substitution reactions of Alkyl Halides There are two types of possible reaction in organic compounds in which sp 3 carbon is bonded to an electronegative atom or group (ex, halides) 1. Substitution
Chapter 8. Substitution reactions of Alkyl Halides There are two types of possible reaction in organic compounds in which sp 3 carbon is bonded to an electronegative atom or group (ex, halides) 1. Substitution
FOURTH EXAMINATION. (1)..20 pts... (2)..24 pts... (3)..18 pts... (4)..12 pts... (5)..12 pts... (6).. 6 pts... (7).. 8 pts... Bonus 4 pts...
 Name: CHEM 331 FURTH EXAMINATIN All answers should be written on the exam in the spaces provided. Clearly indicate your answers in the spaces provided; if I have to guess as to what or where your answer
Name: CHEM 331 FURTH EXAMINATIN All answers should be written on the exam in the spaces provided. Clearly indicate your answers in the spaces provided; if I have to guess as to what or where your answer
C h a p t e r S e v e n : Substitution Reactions S N 2 O H H H O H H. Br -
 C h a p t e r S e v e n : Substitution Reactions Br Br S N 2 CM 321: Summary of Important Concepts YConcepts for Chapter 7: Substitution Reactions I. Nomenclature of alkyl halides, R X A. Common name:
C h a p t e r S e v e n : Substitution Reactions Br Br S N 2 CM 321: Summary of Important Concepts YConcepts for Chapter 7: Substitution Reactions I. Nomenclature of alkyl halides, R X A. Common name:
OChem1 Old Exams. Chemistry 3719 Practice Exams
 OChem1 Old Exams Chemistry 3719 Practice Exams Fall 2017 Chemistry 3719 Practice Exam A1 This exam is worth 100 points out of a total of 600 points for Chemistry 3719/3719L. You have 50 minutes to complete
OChem1 Old Exams Chemistry 3719 Practice Exams Fall 2017 Chemistry 3719 Practice Exam A1 This exam is worth 100 points out of a total of 600 points for Chemistry 3719/3719L. You have 50 minutes to complete
Reaction mechanism 1/17/19. HCl. HCl. 3 radical can form here. Br 2. hint!
 If the reagents or reaction conditions favor radicals, then it is likely to be a radical reaction. Look for hints like peroxide, AIBN, NBS, hn (UV light). 3 radical can form here Reaction mechanism How
If the reagents or reaction conditions favor radicals, then it is likely to be a radical reaction. Look for hints like peroxide, AIBN, NBS, hn (UV light). 3 radical can form here Reaction mechanism How
3-chloro-1-propene 1-chloropropane 2-chloropropene
 ANSWERS #1. (from 50 minute exam #3, Fall 2000) 5. (6 points) For each group of 3 compounds, identify the compound that expresses the indicated property the MOST and the compound that expresses it the
ANSWERS #1. (from 50 minute exam #3, Fall 2000) 5. (6 points) For each group of 3 compounds, identify the compound that expresses the indicated property the MOST and the compound that expresses it the
Chem Final Examination August 7, 2004
 Chem 281 2004-2 Final Examination August 7, 2004 Name: Student Number: Note: You are allowed to use models for this exam. Notes, textbooks and calculators are strictly prohibited. Write your final answers
Chem 281 2004-2 Final Examination August 7, 2004 Name: Student Number: Note: You are allowed to use models for this exam. Notes, textbooks and calculators are strictly prohibited. Write your final answers
75. A This is a Markovnikov addition reaction. In these reactions, the pielectrons in the alkene act as a nucleophile. The strongest electrophile will
 71. B SN2 stands for substitution nucleophilic bimolecular. This means that there is a bimolecular rate-determining step. Therefore, the reaction will follow second-order kinetics based on the collision
71. B SN2 stands for substitution nucleophilic bimolecular. This means that there is a bimolecular rate-determining step. Therefore, the reaction will follow second-order kinetics based on the collision
Chapter 17. Reactions of Aromatic Compounds
 Chapter 17 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Although benzene s pi electrons are in a stable aromatic system, they are available to attack a strong electrophile to give
Chapter 17 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Although benzene s pi electrons are in a stable aromatic system, they are available to attack a strong electrophile to give
Detailed Course Content
 Detailed Course Content Chapter 1: Carbon Compounds and Chemical Bonds The Structural Theory of Organic Chemistry 4 Chemical Bonds: The Octet Rule 6 Lewis Structures 8 Formal Charge 11 Resonance 14 Quantum
Detailed Course Content Chapter 1: Carbon Compounds and Chemical Bonds The Structural Theory of Organic Chemistry 4 Chemical Bonds: The Octet Rule 6 Lewis Structures 8 Formal Charge 11 Resonance 14 Quantum
Lecture 18 Organic Chemistry 1
 CEM 232 rganic Chemistry I at Chicago Lecture 18 rganic Chemistry 1 Professor Duncan Wardrop March 9, 2010 1 Nucleophilicity nucleophilicity: measures the strength of the nucleophile ; more nucleophilic
CEM 232 rganic Chemistry I at Chicago Lecture 18 rganic Chemistry 1 Professor Duncan Wardrop March 9, 2010 1 Nucleophilicity nucleophilicity: measures the strength of the nucleophile ; more nucleophilic
Chapter 8: Alkene Structure and Preparation via Elimination Reactions
 1. Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C bond length bond strength 3 C C 3 3 C C 3 3 C C 3 3 C 2 C C 2 3 C a C=C double bond
1. Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C bond length bond strength 3 C C 3 3 C C 3 3 C C 3 3 C 2 C C 2 3 C a C=C double bond
Basic Organic Chemistry Course code : CHEM (Pre-requisites : CHEM 11122)
 Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
Homework for Chapter 17 Chem 2320
 Homework for Chapter 17 Chem 2320 I. Cumulated, isolated, and conjugated dienes Name 1. Draw structures which fit the following descriptions. Use correct geometry! a conjugated diene with the formula C
Homework for Chapter 17 Chem 2320 I. Cumulated, isolated, and conjugated dienes Name 1. Draw structures which fit the following descriptions. Use correct geometry! a conjugated diene with the formula C
Dehydrohalogenation of Alkyl Halides E2 and E1 Reactions in Detail
 Dehydrohalogenation of Alkyl Halides E2 and E1 Reactions in Detail b-elimination Reactions Overview dehydration of alcohols: X = H; Y = OH dehydrohalogenation of alkyl halides: X = H; Y = Br, etc. X C
Dehydrohalogenation of Alkyl Halides E2 and E1 Reactions in Detail b-elimination Reactions Overview dehydration of alcohols: X = H; Y = OH dehydrohalogenation of alkyl halides: X = H; Y = Br, etc. X C
Walden discovered a series of reactions that could interconvert (-)-malic acid and (+)-malic acid.
 Chapter 11: Reactions of alkyl halides: nucleophilic substitutions and eliminations Alkyl halides are polarized in the C-X bond, making carbon δ+ (electrophilic). A nucleophilecan attack this carbon, displacing
Chapter 11: Reactions of alkyl halides: nucleophilic substitutions and eliminations Alkyl halides are polarized in the C-X bond, making carbon δ+ (electrophilic). A nucleophilecan attack this carbon, displacing
Structure and Preparation of Alkenes: Elimination Reactions
 Structure and Preparation of Alkenes: Elimination Reactions Alkene Nomenclature First identify the longest continuous chain that includes the double bond. Replace the -ane ending of the corresponding unbranched
Structure and Preparation of Alkenes: Elimination Reactions Alkene Nomenclature First identify the longest continuous chain that includes the double bond. Replace the -ane ending of the corresponding unbranched
Halo Alkanes and Halo Arenes
 alo Alkanes and alo Arenes Short Answer Questions: **1. Write the isomers of the compound having formula C 4 9 Br? Sol. There are five isomers of C 4 9 Br. These are: 2-bromobutane is expected to exhibit
alo Alkanes and alo Arenes Short Answer Questions: **1. Write the isomers of the compound having formula C 4 9 Br? Sol. There are five isomers of C 4 9 Br. These are: 2-bromobutane is expected to exhibit
trans-cyclooctene 4-fluoro-1-butanol 2-cyclohexenol 4. (5 pts) Provide structural formula for each of the following molecules: Br OH Cl OH
 CEMISTRY 313-01 MIDTERM # 2 answer key March 12, 2009 Statistics: Average: 72 pts (72%); ighest: 98 pts (98%); Lowest: 21 pts (21%) umber of students performing at or above average: 22 (50%) umber of students
CEMISTRY 313-01 MIDTERM # 2 answer key March 12, 2009 Statistics: Average: 72 pts (72%); ighest: 98 pts (98%); Lowest: 21 pts (21%) umber of students performing at or above average: 22 (50%) umber of students
Chemistry 35 Exam 2 Answers - April 9, 2007
 Chemistry 35 Exam 2 Answers - April 9, 2007 Problem 1. (14 points) Provide the products for the reactions shown below. If stereochemistry is important, be sure to indicate stereochemistry clearly. If no
Chemistry 35 Exam 2 Answers - April 9, 2007 Problem 1. (14 points) Provide the products for the reactions shown below. If stereochemistry is important, be sure to indicate stereochemistry clearly. If no
HALOGENOALKANES (HALOALKANES) Structure Contain the functional group C-X where X is a halogen (F, Cl, Br or I)
 alogenoalkanes F322 1 ALOGENOALKANES (ALOALKANES) Structure ontain the functional group X where X is a halogen (F, l, or I) Types alogenoalkanes halogen is attached to an aliphatic skeleton alkyl group
alogenoalkanes F322 1 ALOGENOALKANES (ALOALKANES) Structure ontain the functional group X where X is a halogen (F, l, or I) Types alogenoalkanes halogen is attached to an aliphatic skeleton alkyl group
CHEM 302 Organic Chemistry I Problem Set VII Chapter 7 Answers
 CEM 302 rganic Chemistry Problem Set V Chapter 7 Answers 1) The pk a values of acetic acid and trifluroacetic acid are 4.5 and 0.9 respectively. Based on this information, which anion, C 3 C - or CF 3
CEM 302 rganic Chemistry Problem Set V Chapter 7 Answers 1) The pk a values of acetic acid and trifluroacetic acid are 4.5 and 0.9 respectively. Based on this information, which anion, C 3 C - or CF 3
Glendale Community College Chemistry 105 Exam. 3 Lecture Notes Chapters 6 & 7
 Sevada Chamras, Ph.D. Glendale Community College Chemistry 105 Exam. 3 Lecture Notes Chapters 6 & 7 Description: Examples: 3 Major Types of Organic Halides: 1. Alkyl Halides: Chapter 6 (Part 1/2) : Alkyl
Sevada Chamras, Ph.D. Glendale Community College Chemistry 105 Exam. 3 Lecture Notes Chapters 6 & 7 Description: Examples: 3 Major Types of Organic Halides: 1. Alkyl Halides: Chapter 6 (Part 1/2) : Alkyl
CHAPTER 8 HW SOLUTIONS: ELIMINATIONS REACTIONS
 APTER 8 W SLUTNS: ELMNATNS REATNS S-TRANS SMERSM 1. Use a discussion and drawing of orbitals to help explain why it is generally easy to rotate around single bonds at room temperature, while it is difficult
APTER 8 W SLUTNS: ELMNATNS REATNS S-TRANS SMERSM 1. Use a discussion and drawing of orbitals to help explain why it is generally easy to rotate around single bonds at room temperature, while it is difficult
CHE 321 Summer 2010 Exam 2 Form Choose the structure(s) that represent cis-1-sec-butyl-4-methylcyclohexane. I II III
 CE 321 Summer 2010 Exam 2 orm 2 Multiple Choice Questions: 60 points 1. Choose the structure(s) that represent cis-1-sec-butyl-4-methylcyclohexane. I II III (A) I only (B) II only (C) I and II only II
CE 321 Summer 2010 Exam 2 orm 2 Multiple Choice Questions: 60 points 1. Choose the structure(s) that represent cis-1-sec-butyl-4-methylcyclohexane. I II III (A) I only (B) II only (C) I and II only II
Organic Halogen Compounds
 8 Organic alogen ompounds APTER SUMMARY 8.1 Introduction Although organic halogen compounds are rarely found in nature, they do have a variety of commercial applications including use as insecticides,
8 Organic alogen ompounds APTER SUMMARY 8.1 Introduction Although organic halogen compounds are rarely found in nature, they do have a variety of commercial applications including use as insecticides,
CHEM 2311 HW1. COMPLETELY FILL THE BOXES IN DARK PENCIL, i.e.:, NOT or or STRUCTURE
 EM 2311 W1 Provide answers to the following multiple choice questions on the Scantron form provided in lecture. Make sure that you put your name and bubble in your GTID # on the answer form. MPLETELY FILL
EM 2311 W1 Provide answers to the following multiple choice questions on the Scantron form provided in lecture. Make sure that you put your name and bubble in your GTID # on the answer form. MPLETELY FILL
DAV CENTENARY PUBLIC SCHOOL, PASCHIM ENCLAVE, NEW DELHI - 87
 HYDROCARBONS 1. Why do alkenes prefer to undergo electrophilic addition reaction while arenes prefer electrophilic substitution reactions? Explain. 2. Alkynes on reduction with sodium in liquid ammonia
HYDROCARBONS 1. Why do alkenes prefer to undergo electrophilic addition reaction while arenes prefer electrophilic substitution reactions? Explain. 2. Alkynes on reduction with sodium in liquid ammonia
UCF - ORGANIC CHEMISTRY 1 - PROF. DAOUDI UCF PROF. DAOUDI EXAM 3 REVIEW.
 UCF PROF. DAOUDI EXAM 3 REVIEW www.clutchprep.com 1 PRACTICE: Identify the most stable and the least stable alkene PRACTICE: Create the full arrow pushing mechanism which shows all intermediates and all
UCF PROF. DAOUDI EXAM 3 REVIEW www.clutchprep.com 1 PRACTICE: Identify the most stable and the least stable alkene PRACTICE: Create the full arrow pushing mechanism which shows all intermediates and all
1. What is the letter designation given to dumbbell shaped orbitals like the one depicted below?
 1. What is the letter designation given to dumbbell shaped orbitals like the one depicted below? 2. Which of the following does not have an octet of electrons surrounding the central atom? A. B 3 B. C
1. What is the letter designation given to dumbbell shaped orbitals like the one depicted below? 2. Which of the following does not have an octet of electrons surrounding the central atom? A. B 3 B. C
Chapter 9:Nucleophiles & Substitution Reactions
 1. a. Place the following nucleophiles in order of strength (1= strongest; 3 = weakest). i. ii. b. Place the following in order of leaving group ability (1= best; 7 = worst). (A pka table may help you!)
1. a. Place the following nucleophiles in order of strength (1= strongest; 3 = weakest). i. ii. b. Place the following in order of leaving group ability (1= best; 7 = worst). (A pka table may help you!)
Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320
 Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320 I. Isolated, cumulated, and conjugated dienes A diene is any compound with two or C=C's is a diene. Compounds containing more than two
Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320 I. Isolated, cumulated, and conjugated dienes A diene is any compound with two or C=C's is a diene. Compounds containing more than two
The C-X bond gets longerand weakergoing down the periodic table.
 Chapter 10: Organohalides Organic molecules containing halogen atoms (X) bonded to carbon are useful compounds in synthesis and on their own. 10.2 Structure of alkyl halides The C-X bond gets longerand
Chapter 10: Organohalides Organic molecules containing halogen atoms (X) bonded to carbon are useful compounds in synthesis and on their own. 10.2 Structure of alkyl halides The C-X bond gets longerand
BSc. II 3 rd Semester. Submitted By Dr. Sangita Nohria Associate Professor PGGCG-11 Chandigarh 1
 BSc. II 3 rd Semester Submitted By Dr. Sangita Nohria Associate Professor PGGCG-11 Chandigarh 1 Introduction to Alkyl Halides Alkyl halides are organic molecules containing a halogen atom bonded to an
BSc. II 3 rd Semester Submitted By Dr. Sangita Nohria Associate Professor PGGCG-11 Chandigarh 1 Introduction to Alkyl Halides Alkyl halides are organic molecules containing a halogen atom bonded to an
HALOGENOALKANES (HALOALKANES) Structure Contain the functional group C-X where X is a halogen (F,Cl,Br or I)
 aloalkanes 2812 1 ALOGENOALKANES (ALOALKANES) Structure ontain the functional group X where X is a halogen (F,l, or I) Types alogenoalkanes halogen is attached to an aliphatic skeleton alkyl group aloarenes
aloalkanes 2812 1 ALOGENOALKANES (ALOALKANES) Structure ontain the functional group X where X is a halogen (F,l, or I) Types alogenoalkanes halogen is attached to an aliphatic skeleton alkyl group aloarenes
PAPER No. 5: REACTION MECHANISM MODULE No. 2: Types of Organic Reaction Mechanisms
 Subject Chemistry Paper No and Title Module No and Title Module Tag Paper No. 5:Organic Chemistry-II Module No. 2: Overview of different types of Organic Reaction Mechanisms CHE_P5_M2 TABLE OF CONTENTS
Subject Chemistry Paper No and Title Module No and Title Module Tag Paper No. 5:Organic Chemistry-II Module No. 2: Overview of different types of Organic Reaction Mechanisms CHE_P5_M2 TABLE OF CONTENTS
CHEMISTRY MIDTERM # 1 answer key October 05, 2010
 CEMISTRY 313-03 MIDTERM # 1 answer key ctober 05, 2010 Statistics: Average: 73 pts (73%); ighest: 99 pts (99%); Lowest: 31 pts (31%) Number of students performing at or above average: 61 (52%) Number of
CEMISTRY 313-03 MIDTERM # 1 answer key ctober 05, 2010 Statistics: Average: 73 pts (73%); ighest: 99 pts (99%); Lowest: 31 pts (31%) Number of students performing at or above average: 61 (52%) Number of
Exam 1 Chem 3045x Monday, October 1, 2001
 Exam 1 Chem 3045x Monday, October 1, 2001 Instructions: This is a closed book examination. Please print your name and social security number on the front page of the examination. Be sure to allot your
Exam 1 Chem 3045x Monday, October 1, 2001 Instructions: This is a closed book examination. Please print your name and social security number on the front page of the examination. Be sure to allot your
17.3 REACTIONS INVOLVING ALLYLIC AND BENZYLIC ANIONS
 798 HAPTER 17 ALLYLI AND BENZYLI REATIVITY Because the unpaired electron is shared by two different carbons, this radical can react in the final propagation step to give two different products. Reaction
798 HAPTER 17 ALLYLI AND BENZYLI REATIVITY Because the unpaired electron is shared by two different carbons, this radical can react in the final propagation step to give two different products. Reaction
What is the major product of the following reaction?
 What is the major product of the following reaction? Predict the major product of the following reaction: 2-methylbutane + Br 2 /light energy? A) 1-bromo-2-methylbutane B) 2-bromo-2-methylbutane C) 2-bromo-3-methylbutane
What is the major product of the following reaction? Predict the major product of the following reaction: 2-methylbutane + Br 2 /light energy? A) 1-bromo-2-methylbutane B) 2-bromo-2-methylbutane C) 2-bromo-3-methylbutane
Chapter 17 Reactions of Aromatic Compounds
 rganic Chemistry, 6 th Edition L. G. Wade, Jr. Chapter 17 Reactions of Aromatic Compounds Jo Blackburn Richland College, Dallas, TX Dallas County Community College District 2006, Prentice all Electrophilic
rganic Chemistry, 6 th Edition L. G. Wade, Jr. Chapter 17 Reactions of Aromatic Compounds Jo Blackburn Richland College, Dallas, TX Dallas County Community College District 2006, Prentice all Electrophilic
12/27/2010. Chapter 15 Reactions of Aromatic Compounds
 Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen atom
Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen atom
1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound?
 CEM 331: Chapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N C 2 C N C 2 C N 1 2 3 4 1: three sigma bonds and
CEM 331: Chapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N C 2 C N C 2 C N 1 2 3 4 1: three sigma bonds and
Reactions of Haloalkanes
 Reactions of aloalkanes N Goalby chemrevise.org aloalkanes ontain a halogen atom covalently bonded to a carbon atom General formula: R-X where X is a halogen atom (F, l,, I) and R is the carbon chain.
Reactions of aloalkanes N Goalby chemrevise.org aloalkanes ontain a halogen atom covalently bonded to a carbon atom General formula: R-X where X is a halogen atom (F, l,, I) and R is the carbon chain.
1) (100 pts) 5) (20 pts) 3) (35 pts) 4) (25pts. Total (200 pts) More Tutorial at
 Name: Perm number: Question 1) (100 pts) 2) (20 pts) 3) (35 pts) 4) (25pts 5) (20 pts) Total (200 pts) Your score 1. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers
Name: Perm number: Question 1) (100 pts) 2) (20 pts) 3) (35 pts) 4) (25pts 5) (20 pts) Total (200 pts) Your score 1. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers
Department of Chemistry SUNY/Oneonta. Chem Organic Chemistry I. Examination #4 - December 9, 2002 ANSWERS
 INSTRUCTINS Department of Chemistry SUNY/neonta Chem 221 - rganic Chemistry I Examination #4 - December 9, 2002 ANSWERS This examination is in multiple choice format; the questions are in this Exam Booklet
INSTRUCTINS Department of Chemistry SUNY/neonta Chem 221 - rganic Chemistry I Examination #4 - December 9, 2002 ANSWERS This examination is in multiple choice format; the questions are in this Exam Booklet
Chemistry 250B Final Exam Answer Key December 19, 2008
 Name: NSWER KEY 1 hemistry 250 Final Exam nswer Key ecember 19, 2008 Show non-zero formal charges on all atoms for all structures. There are 11 pages. 1. (42 pts) omplete the following reactions. Show
Name: NSWER KEY 1 hemistry 250 Final Exam nswer Key ecember 19, 2008 Show non-zero formal charges on all atoms for all structures. There are 11 pages. 1. (42 pts) omplete the following reactions. Show
How alkyl halides react
 Chapter 10 1 How alkyl halides react δ+ δ- RCH 2 -X X= halogen X = higher EN C = lower EN This polar carbon-halogen bond causes alkyl halide to undergo S N and elimination reaction. 2 The mechanism of
Chapter 10 1 How alkyl halides react δ+ δ- RCH 2 -X X= halogen X = higher EN C = lower EN This polar carbon-halogen bond causes alkyl halide to undergo S N and elimination reaction. 2 The mechanism of
Learning Guide for Chapter 15 - Alcohols (II)
 Learning Guide for Chapter 15 - Alcohols (II) I. Introduction to alcohol reactivity II. Reactions of alcohols with acids III. Reactions of alcohols with electrophiles alogenated phosphorus and sulfur compounds
Learning Guide for Chapter 15 - Alcohols (II) I. Introduction to alcohol reactivity II. Reactions of alcohols with acids III. Reactions of alcohols with electrophiles alogenated phosphorus and sulfur compounds
Aromatic Compounds II
 2302272 Org Chem II Part I Lecture 2 Aromatic Compounds II Instructor: Dr. Tanatorn Khotavivattana E-mail: tanatorn.k@chula.ac.th Recommended Textbook: Chapter 17 in Organic Chemistry, 8 th Edition, L.
2302272 Org Chem II Part I Lecture 2 Aromatic Compounds II Instructor: Dr. Tanatorn Khotavivattana E-mail: tanatorn.k@chula.ac.th Recommended Textbook: Chapter 17 in Organic Chemistry, 8 th Edition, L.
Chapter 10. BrCH 2 CH 2 CH 2 CCH 2 Br CH 3. CH 3 CCH 2 CH 2 Cl CH 3 CHCH 2 CH 2 CHCH Give IUPAC names for the following alkyl halides:
 hapter 10 10.1 Give IUPA names for the following alkyl halides: (a), 3 2 2 2 I 1-iodobutane 3 (b), 3 2 2 l 1-chloro-3-methylbutane (c), 3 2 2 2 2 3 1,5-Dibromo-2,2-dimethylpentane 3 3 2 2 l (d), l 1,3-Dichloro-3-methylbutane
hapter 10 10.1 Give IUPA names for the following alkyl halides: (a), 3 2 2 2 I 1-iodobutane 3 (b), 3 2 2 l 1-chloro-3-methylbutane (c), 3 2 2 2 2 3 1,5-Dibromo-2,2-dimethylpentane 3 3 2 2 l (d), l 1,3-Dichloro-3-methylbutane
I C(CH 3 ) 3. (c)2ch 3 OH+(CH 3 ) 3 CCl (CH 3 ) 3 COCH 3 +CH 3 OH + 2 +Cl - (d) CH 3 CH 2 CH 2 Br+NaCN CH 3 CH 2 CH 2 CN+NaBr
 6.1 Writer the following as net ionic reactions and designate the nucleophile, substrate, and leaving group in each reaction. (a) 3 + 3 2 Na 3 2 3 +Na (b) Na+ 3 2 3 2 +Na (c) 2 3 +( 3 ) 3 ( 3 ) 3 3 + 3
6.1 Writer the following as net ionic reactions and designate the nucleophile, substrate, and leaving group in each reaction. (a) 3 + 3 2 Na 3 2 3 +Na (b) Na+ 3 2 3 2 +Na (c) 2 3 +( 3 ) 3 ( 3 ) 3 3 + 3
Learning Guide for Chapter 18 - Aromatic Compounds II
 Learning Guide for Chapter 18 Aromatic Compounds. lectrophilic aromatic substitution ntroduction Mechanism Reagents and Products lectrophiles ffects of stituents FriedelCrafts alkylation and acylation
Learning Guide for Chapter 18 Aromatic Compounds. lectrophilic aromatic substitution ntroduction Mechanism Reagents and Products lectrophiles ffects of stituents FriedelCrafts alkylation and acylation
Learning Guide for Chapter 17 - Dienes
 Learning Guide for Chapter 17 - Dienes I. Isolated, conjugated, and cumulated dienes II. Reactions involving allylic cations or radicals III. Diels-Alder Reactions IV. Aromaticity I. Isolated, Conjugated,
Learning Guide for Chapter 17 - Dienes I. Isolated, conjugated, and cumulated dienes II. Reactions involving allylic cations or radicals III. Diels-Alder Reactions IV. Aromaticity I. Isolated, Conjugated,
1. Complete the template below to show the stereochemistry of (3R,4R) 3-chloro-4,5,5- trimethyl-hexane-1,4-diol.
 Chemistry 51 Exam 1, Fall 2004 This is a closed book exam. The exam lasts 50 minutes. All answers must appear on the answer sheet. Only the answer sheet will be collected. Put your name on the answer sheet
Chemistry 51 Exam 1, Fall 2004 This is a closed book exam. The exam lasts 50 minutes. All answers must appear on the answer sheet. Only the answer sheet will be collected. Put your name on the answer sheet
Classes of Halides. Chapter 6 Alkyl Halides: Nucleophilic Substitution and Elimination. Polarity and Reactivity. Classes of Alkyl Halides
 rganic hemistry, 5 th Edition L. G. Wade, Jr. hapter 6 Alkyl alides: Nucleophilic Substitution and Elimination lasses of alides Alkyl: alogen, X, is directly bonded to sp 3 carbon. Vinyl: X is bonded to
rganic hemistry, 5 th Edition L. G. Wade, Jr. hapter 6 Alkyl alides: Nucleophilic Substitution and Elimination lasses of alides Alkyl: alogen, X, is directly bonded to sp 3 carbon. Vinyl: X is bonded to
Chapter 11, Part 1: Polar substitution reactions involving alkyl halides
 hapter 11, Part 1: Polar substitution reactions involving alkyl halides Overview: The nature of alkyl halides and other groups with electrophilic sp 3 hybridized leads them to react with nucleophiles and
hapter 11, Part 1: Polar substitution reactions involving alkyl halides Overview: The nature of alkyl halides and other groups with electrophilic sp 3 hybridized leads them to react with nucleophiles and
Name: Student Number:
 Page 1 of 5 Name: Student Number: l University of Manitoba - Department of Chemistry CEM 2220 - Introductory rganic Chemistry II - Term Test 1 Thursday, February 14, 2008 This is a 2-hour test, marked
Page 1 of 5 Name: Student Number: l University of Manitoba - Department of Chemistry CEM 2220 - Introductory rganic Chemistry II - Term Test 1 Thursday, February 14, 2008 This is a 2-hour test, marked
Organic Reactions Susbstitution S N. Dr. Sapna Gupta
 Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams.
 2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams. Disclaimer.: Use only to help learn what you need to know and don t expect the final to be in the same form. 1 1. Short
2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams. Disclaimer.: Use only to help learn what you need to know and don t expect the final to be in the same form. 1 1. Short
Chapter 15 Reactions of Aromatic Compounds
 Chapter 15 1 Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen
Chapter 15 1 Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen
Practice Multiple-Choice Questions for Ending Chem 21 and/or Starting Chem 22
 Practice Multiple-Choice Questions for Ending Chem 21 and/or Starting Chem 22 Includes information from chapters 5-10 of the third edition of Klein s Organic Chemistry text. 1. Which of the following compounds
Practice Multiple-Choice Questions for Ending Chem 21 and/or Starting Chem 22 Includes information from chapters 5-10 of the third edition of Klein s Organic Chemistry text. 1. Which of the following compounds
