Naming Chemical Compounds And above all these put on love, which binds everything together in perfect harmony. Col 3:14
|
|
|
- Jewel Wilkerson
- 5 years ago
- Views:
Transcription
1 Naming Chemical Compounds And above all these put on love, which binds everything together in perfect harmony. Col 3:14 Introduction Chemical Compounds are placed into one of two categories, ionic compounds and molecular compounds. Ionic compounds are formed between a metal and a nonmetal or between a polyatomic ion and a nonmetal. Molecular compounds are formed between two or more nonmetals held together by covalent bonds. As they differ in the bonds that hold them, so too are they distinguished by their naming. In the lab Ions and Ionic Bond Puzzle, you practiced labeling the charges of ions of metals and nonmetals, reviewed polyatomic ions, and performed an exercise to predict the ratios in which metal anions and nonmetal cations combine. This knowledge is necessary in naming ionic compounds, particularly in going from the ionic compound name to its chemical formula. If necessary, review the information in that lab now. Oxidation numbers are assigned to elements within a compound and indicate the element s positive or negative nature. Oxidation numbers are needed to: A) write the chemical formula from the ionic compound name, and; B) assign the Roman Numeral on the transition metal cation for ionic compounds with variable oxidation numbers. Molecular compounds may be divided into three categories. Binary molecular compounds contain two nonmetals held by covalent bonds. Ternary molecular compounds contain more than two covalently bound nonmetals. Acids are compounds in which hydrogen is bound to either a nonmetal (a binary acid) or to a polyatomic acid (a ternary oxyacid). Given a formula can you recognize whether the compound is ionic or molecular? After learning how to name ionic and covalent compounds, it is imperative to be able to distinguish between them given either just the name or the formula. The final portion of this lab will be an exercise in putting all the information together. Learning Objectives: Proficiency in naming ionic and covalent compounds from the formula Competence in providing an ionic or covalent formula given the compound name Identify a compound as ionic or covalent Materials Required: From Chemistry Kit Student Supplied Periodic Table Text or Reference book, if desired Part 1 Ionic Compounds We will practice naming binary ionic compounds consisting of one metal and one nonmetal. We will also practice naming ternary ionic compounds which contain polyatomic ions (a group of more than one nonmetals held together by covalent bonds and holding a charge). Here is a 2016 Catholic Initiatives in Math and Science, LLC All Rights Reserved 1
2 short summary of the rules for naming, although more details may be found in your textbook or a reference book: The cation keeps its name and the anion uses the root name plus an ide ending. For example, Rb2O is named rubidium oxide. Metals with variable charges are written with Roman numerals following the name. For example, FeBr2 is named Iron(II) bromide. Polyatomic ions keep their names in naming ionic compounds in which they are present. For example, NH4F is ammonium fluoride and LiCN is lithium cyanide In compounds, each element has an oxidation number that indicates its positive or negative character. When going from the name to an ionic compound formula, oxidation numbers must be found so that the ratio of cation to anion may be determined. The oxidation number is the same as the charge on that atom. This is how the charge on iron in FeBr2 was determined, and the compound named Iron(II) bromide. Contrast that with charge determination on iron of FeBr3 and naming this ionic compound, Iron(III) bromide. (See the lab titled: Ions and Ionic Bond Puzzle.) Experiment Naming Ionic Compounds 1. Fill out the empty boxes in Table 1: Where there is a name, place the corresponding formula Where there is a formula, place the corresponding name. 2. Complete the questions under Part I: Data Analysis 3. Proceed to Part 2 Naming Molecular Compounds 2016 Catholic Initiatives in Math and Science, LLC All Rights Reserved 2
3 Part 2 Molecular Compounds Molecular compounds may be binary (containing two nonmetals held together by covalent bonds) or ternary (containing more than two different nonmetals). In naming binary molecular compounds, follow the rules below For binary molecular compounds that are NOT acids: The first atom is named first with its full name The second atom is named with its root plus an ide ending The number of atoms of each element are indicated by Greek prefixes The prefix mono is never used for the first atom For example, SF6 is sulfur hexafluoride and S2F10 is disulfur decafluoride Binary acids (compound with hydrogen) Follow the above rule, but do not use the prefix since hydrogen can only form one bond with any other atom For example, H2Se is hydrogen chloride Binary acids as a pure compound may be written Binary compounds with hydrogen become acids in their aqueous form The term (aq) is used to denote the acid is dissolved in water For example, HCl (aq) is hydrochloric acid But, HCl as a pure compound is hydrogen chloride To name ternary molecular compounds: Ternary acids (oxyacids) have a hydrogen and a polyatomic ion A per ate ion is named as a per ic acid HCLO4 (aq) has the perchlorate ion and is named perchloric acid An ate ion is named as an ic acid HCLO3 (aq) has the chlorate ion and is named chloric acid An ite ion is named as an ous acid HCLO2 (aq) has the chlorite ion and is named chlorous acid A hypo ite complex is named as a hypo ous acid HCLO (aq) has the hypochlorite ion and is named hypochlorous acid Experiment Molecular Compounds 1. Fill out the empty boxes in Table 2: Where there is a name, place the corresponding formula Where there is a formula, place the corresponding name. 2. Perform Data Analyses Part 2 3. Proceed to Part Catholic Initiatives in Math and Science, LLC All Rights Reserved 3
4 Part 3 Putting It All Together When confronted with a compound and asked to name it or to write the formula, you must first determine whether this is an ionic compound or a molecular compound. This is easily answered by determining whether the compound begins with a nonmetal or with ammonium ion. Once this question is answered, you can name the compound or write its formula by following a follow the flow of logical questions. If it is an Ionic Compound: 2016 Catholic Initiatives in Math and Science, LLC All Rights Reserved 4
5 If it is a Covalent Compound: Experiment Putting It All Together 1. Fill out the empty boxes in Table 3: Identify the compound as ionic or covalent Where there is a name, place the corresponding formula Where there is a formula, place the corresponding name. 2. Perform Data Analyses Part Catholic Initiatives in Math and Science, LLC All Rights Reserved 5
6 TABLE 1 Ionic Compounds FORMULA NAME SrI2 Lithium oxide CaCl2 NaCl Silver oxide FePO4 K2S Aluminum fluoride CuCl Copper(II) chloride FeCl2 Iron(III) chloride Mn3N4 Aluminum sulfide Data Analysis Part 1 1. What are the oxidation numbers for manganese and nitrogen in Mn3N4? 2. Give the name and formula for any ionic compound having a polyatomic ion Catholic Initiatives in Math and Science, LLC All Rights Reserved 6
7 TABLE 2 Molecular Compounds FORMULA NAME Xenon difluoride SO3 Diboron difluoride P4O10 H2Se Hydrogen chloride Hydroiodic acid HCl (aq) Sulfurous acid H2SO4 (aq) Nitrous acid HNO3 (aq) Hydrogen cyanide Data Analysis Part 2 3. Name the first ten Greek prefixes used in naming molecular compounds: # of Atoms Greek Prefix # of Atoms Greek Prefix 1 atom 6 atoms 2 atoms 7 atoms 3 atoms 8 atoms 4 atoms 9 atoms 5 atoms 10 atoms 2016 Catholic Initiatives in Math and Science, LLC All Rights Reserved 7
8 TABLE 3 Putting It All Together Ionic or Covalent? FORMULA NAME NI3 Sodium hydride BaI2 Sulfur hexafluoride CsBr Tin(II) iodide Co(C2H3O2)2 Potassium nitride Ag3PO4 Sulfur dioxide PO3 Ammonium nitrate CCl4 Iron(III) perchlorate N2O4 Data Analysis Part 3 Tetrasulfur tetranitride KCN 4. How do you determine whether a compound is molecular (covalent) or ionic? 2016 Catholic Initiatives in Math and Science, LLC All Rights Reserved 8
Chemical Formulas Types of chemical formulas [X m Y n ]
![Chemical Formulas Types of chemical formulas [X m Y n ] Chemical Formulas Types of chemical formulas [X m Y n ]](/thumbs/89/98748683.jpg) 1 Chemical Formulas Types of chemical formulas [X m Y n ] Molecular Actual # of atoms of each element Empirical Structural Relative # of atoms of each element Actual # of atoms and the bonds between them
1 Chemical Formulas Types of chemical formulas [X m Y n ] Molecular Actual # of atoms of each element Empirical Structural Relative # of atoms of each element Actual # of atoms and the bonds between them
Chemical Nomenclature
 Chemical Nomenclature! The first names for chemicals were common names: Sugar, quicklime, Epsom salts, milk of magnesia, gypsom, laughing gas Simple, but not practical, the tell us little about the chemicals
Chemical Nomenclature! The first names for chemicals were common names: Sugar, quicklime, Epsom salts, milk of magnesia, gypsom, laughing gas Simple, but not practical, the tell us little about the chemicals
Experiment #4. Chemical Nomenclature
 Experiment #4. Chemical Nomenclature Many everyday and historically important chemical compounds have common names. For example, water is the common name for H 2 O, baking soda is the common name for NaHCO
Experiment #4. Chemical Nomenclature Many everyday and historically important chemical compounds have common names. For example, water is the common name for H 2 O, baking soda is the common name for NaHCO
Chemical Formulas & Chemical Compounds. Chemical formula indicates the relative number of atoms of each kind in a chemical compound.
 Unit 6: Chemical Formulas & Chemical Compounds Chemical Names & Formulas Chemical formula indicates the relative number of atoms of each kind in a chemical compound. Molecular compound - it s formula reveals
Unit 6: Chemical Formulas & Chemical Compounds Chemical Names & Formulas Chemical formula indicates the relative number of atoms of each kind in a chemical compound. Molecular compound - it s formula reveals
Naming and Formula Writing
 + Naming and Formula Writing + Chemical Formulas Shows the kind and number of atoms in the smallest piece of a substance Use subscripts to show the number of atoms per element Molecular formula- number
+ Naming and Formula Writing + Chemical Formulas Shows the kind and number of atoms in the smallest piece of a substance Use subscripts to show the number of atoms per element Molecular formula- number
Summary for Naming Compounds
 Summary for Naming Compounds 1. Group 1, 2, 3 metal with Group 15, 16, 17 nonmetals 2. Group 1, 2, 3, metal with polyatomic ions 3. Transition/other metal with Group 15, 16, 17 nonmetal 4. Transition/other
Summary for Naming Compounds 1. Group 1, 2, 3 metal with Group 15, 16, 17 nonmetals 2. Group 1, 2, 3, metal with polyatomic ions 3. Transition/other metal with Group 15, 16, 17 nonmetal 4. Transition/other
CHEMICAL FORMULAS - NOMENCLATURE (The Language of Chemistry)
 CHEMICAL FORMULAS - NOMENCLATURE (The Language of Chemistry) Systematic chemical names of inorganic compounds were developed by a group of scientists who were part of the International Union of Pure and
CHEMICAL FORMULAS - NOMENCLATURE (The Language of Chemistry) Systematic chemical names of inorganic compounds were developed by a group of scientists who were part of the International Union of Pure and
Big Idea: Matter & Atoms
 Big Idea: Matter & Atoms Naming Ionic Compounds Naming Covalent Compounds Naming Acids Naming Hydrates The cation (positive ion) is written first Takes the same name as the element if only forms one charge
Big Idea: Matter & Atoms Naming Ionic Compounds Naming Covalent Compounds Naming Acids Naming Hydrates The cation (positive ion) is written first Takes the same name as the element if only forms one charge
Books at Amazon.com on Nanotechnology
 Books at Amazon.com on Nanotechnology http://www.amazon.com/s/103-9040246- 0256655?ie=UTF8&keywords=NANOTECHNOLOGY%20&tag =thenanotecinscie&index=books&search=search&link%5fcode=qs Ions Ions are charged
Books at Amazon.com on Nanotechnology http://www.amazon.com/s/103-9040246- 0256655?ie=UTF8&keywords=NANOTECHNOLOGY%20&tag =thenanotecinscie&index=books&search=search&link%5fcode=qs Ions Ions are charged
Topic 5: The Language of Chemistry
 Topic 5: The Language of Chemistry Chemical Formulas & Chemical Compounds (Chapter 7 in Modern Chemistry) A Chemical Formula Recall that a chemical formula indicates the relative number of atoms of each
Topic 5: The Language of Chemistry Chemical Formulas & Chemical Compounds (Chapter 7 in Modern Chemistry) A Chemical Formula Recall that a chemical formula indicates the relative number of atoms of each
AP Chemistry (1 of 20) AP Chemistry (2 of 20) AP Chemistry (3 of 20) AP Chemistry (4 of 20) AP Chemistry (5 of 20) AP Chemistry (6 of 20)
 Ions Ionic Charges of Representative Elements (1 of 20) (2 of 20) Formulas and Names of Common Metal Ions with More than One Common Ionic Charge Some Common Polyatomic Ions (3 of 20) (4 of 20) Naming Monatomic
Ions Ionic Charges of Representative Elements (1 of 20) (2 of 20) Formulas and Names of Common Metal Ions with More than One Common Ionic Charge Some Common Polyatomic Ions (3 of 20) (4 of 20) Naming Monatomic
NOMENCLATURE AND WRITING FORMULAS
 NOMENCLATURE AND WRITING FORMULAS PART I--FORMULAS AND NOMENCLATURE OF IONIC COMPOUND Composed of Cations and Anions. Types of Cations (positive ions): A. Metals lose electrons to form positive ions. These
NOMENCLATURE AND WRITING FORMULAS PART I--FORMULAS AND NOMENCLATURE OF IONIC COMPOUND Composed of Cations and Anions. Types of Cations (positive ions): A. Metals lose electrons to form positive ions. These
Chapter 6 and 15 Ionic Compounds
 Chapter 6 and 15 Ionic Compounds Chapter 6 Ionic compounds 6.3, 6.4 6.1: Intro to Chemical Bonding A chemical bond is a mutual electrical attraction between the nuclei and valence electrons of different
Chapter 6 and 15 Ionic Compounds Chapter 6 Ionic compounds 6.3, 6.4 6.1: Intro to Chemical Bonding A chemical bond is a mutual electrical attraction between the nuclei and valence electrons of different
629 CER Modular Laboratory Program in Chemistry
 M I S C 629 CER Modular Laboratory Program in Chemistry editor: M. L. Gillette Naming Inorganic Chemical Substances prepared by M. L. Gillette, Indiana University Kokomo, and H. A. Neidig, Lebanon Valley
M I S C 629 CER Modular Laboratory Program in Chemistry editor: M. L. Gillette Naming Inorganic Chemical Substances prepared by M. L. Gillette, Indiana University Kokomo, and H. A. Neidig, Lebanon Valley
CHEM 1305: Introductory Chemistry
 CHEM 1305: Introductory Chemistry Naming Inorganic Compounds From Chapter 6 Textbook Introductory Chemistry: Concepts and Critical Thinking Seventh Edition by Charles H. Corwin Language of Chemistry By
CHEM 1305: Introductory Chemistry Naming Inorganic Compounds From Chapter 6 Textbook Introductory Chemistry: Concepts and Critical Thinking Seventh Edition by Charles H. Corwin Language of Chemistry By
Nomenclature. A Systematic Approach to Naming Chemical Compounds
 Nomenclature A Systematic Approach to Naming Chemical Compounds Nomenclature Ternary Ionic Compounds Binary and Ternary Acids Ternary Ionic Compounds >2 elements cation with an anion Metal cation with
Nomenclature A Systematic Approach to Naming Chemical Compounds Nomenclature Ternary Ionic Compounds Binary and Ternary Acids Ternary Ionic Compounds >2 elements cation with an anion Metal cation with
Chemical Formulas and Chemical Nomenclature. Mr. Matthew Totaro Legacy High School Honors Chemistry
 Chemical Formulas and Chemical Nomenclature Mr. Matthew Totaro Legacy High School Honors Chemistry 1 Molecular View of Elements and Compounds 2 Atomic Elements Atomic Elements = elements whose smallest
Chemical Formulas and Chemical Nomenclature Mr. Matthew Totaro Legacy High School Honors Chemistry 1 Molecular View of Elements and Compounds 2 Atomic Elements Atomic Elements = elements whose smallest
Homework #1 Chapter 2 Atoms, Molecules, and Ions
 Homework #1 Chapter 2 Atoms, Molecules, and Ions 19. 1 L (Cl 2) + 5 L (F 2) 2 L (?) For gases the volume is proportional to the number of participles. Since it is proportional, we can simplify the problem
Homework #1 Chapter 2 Atoms, Molecules, and Ions 19. 1 L (Cl 2) + 5 L (F 2) 2 L (?) For gases the volume is proportional to the number of participles. Since it is proportional, we can simplify the problem
Naming Chemical Compounds
 Naming Chemical Compounds Naming compounds is an important part of chemistry. Most compounds fall into one of four categories Ionic Compounds, Molecular Compounds, Acids and Bases, and Hydrates Part One:
Naming Chemical Compounds Naming compounds is an important part of chemistry. Most compounds fall into one of four categories Ionic Compounds, Molecular Compounds, Acids and Bases, and Hydrates Part One:
Chemical Nomenclature Chapter 2.5-8
 Chemical Nomenclature Chapter 2.5-8 Octet Rule An octet is 8 valence electrons is associated with the stability of the noble gases does not occur with He, which is stable with two valence electrons (duet)
Chemical Nomenclature Chapter 2.5-8 Octet Rule An octet is 8 valence electrons is associated with the stability of the noble gases does not occur with He, which is stable with two valence electrons (duet)
CHEM 1105 S10 January 21, 2014
 CHEM 1105 S10 January 21, 2014 Chapter 3: Compounds and Formulas Today: Types of compounds: Ionic vs. covalent Naming ionic compounds Naming binary covalent compounds (two elements only) Ionic Bonding
CHEM 1105 S10 January 21, 2014 Chapter 3: Compounds and Formulas Today: Types of compounds: Ionic vs. covalent Naming ionic compounds Naming binary covalent compounds (two elements only) Ionic Bonding
Naming Compounds. Part One: Naming Ionic Compounds. Identifying Ionic Compounds
 Naming Compounds Naming compounds is an important part of chemistry. Most compounds fall in to one of three categories ionic compounds, molecular compounds, or acids. Part One: Naming Ionic Compounds Identifying
Naming Compounds Naming compounds is an important part of chemistry. Most compounds fall in to one of three categories ionic compounds, molecular compounds, or acids. Part One: Naming Ionic Compounds Identifying
Nomenclature of inorganic compounds. = naming non carbon (mostly) compounds. Some definitions:
 1 Chemistry 047 Inorganic Nomenclature Nomenclature of inorganic compounds = naming non carbon (mostly) compounds Some definitions: Nomenclature = system used by chemists to name and identify compounds
1 Chemistry 047 Inorganic Nomenclature Nomenclature of inorganic compounds = naming non carbon (mostly) compounds Some definitions: Nomenclature = system used by chemists to name and identify compounds
Naming Inorganic Compounds. common names systematic names
 Naming Inorganic Compounds common names systematic names Molecular Common Systematic Formula name name AgCl Lunar caustic Silver chloride H 2 SO 4 Oil of vitriol Sulfuric acid MgSO 4 Epsom salts Magnesium
Naming Inorganic Compounds common names systematic names Molecular Common Systematic Formula name name AgCl Lunar caustic Silver chloride H 2 SO 4 Oil of vitriol Sulfuric acid MgSO 4 Epsom salts Magnesium
H 2 O. Chapter 9 Chemical Names and Formulas
 H 2 O Chapter 9 Chemical Names and Formulas Section 9.1 Naming Ions OBJECTIVES: Identify the charges on monatomic ions by using the periodic table, and name the ions. Section 9.1 Naming Ions OBJECTIVES:
H 2 O Chapter 9 Chemical Names and Formulas Section 9.1 Naming Ions OBJECTIVES: Identify the charges on monatomic ions by using the periodic table, and name the ions. Section 9.1 Naming Ions OBJECTIVES:
Chapter 5 Molecules and Compounds (Part 2)
 Introductory Chemistry, 3 rd Edition Nivaldo Tro Molecules and Compounds (Part 2) Roy Kennedy Massachusetts Bay Community College Wellesley Hills, MA 2009, Prentice Hall 5.6 Nomenclature: Naming Compounds
Introductory Chemistry, 3 rd Edition Nivaldo Tro Molecules and Compounds (Part 2) Roy Kennedy Massachusetts Bay Community College Wellesley Hills, MA 2009, Prentice Hall 5.6 Nomenclature: Naming Compounds
Nomenclature PO 4. phosphate ion. HC 2 H 3 O 2 Acetic Acid C 2 H 3 O 2. acetate ion. Chemistry 1 Honors: Chapter 7: pp
 Chemistry 1 Honors: Chapter 7: pp218-258 PO 4 3- phosphate ion Nomenclature HC 2 H 3 O 2 Acetic Acid C 2 H 3 O 2 - acetate ion SAVE PAPER AND INK!!! When you print out the notes on PowerPoint, print "Handouts"
Chemistry 1 Honors: Chapter 7: pp218-258 PO 4 3- phosphate ion Nomenclature HC 2 H 3 O 2 Acetic Acid C 2 H 3 O 2 - acetate ion SAVE PAPER AND INK!!! When you print out the notes on PowerPoint, print "Handouts"
CHEMISTRY 1: Chemical Bonding
 CHEMISTRY 1: Chemical Bonding Brown, T.L., LeMay, H.E., and B.E. Bursten. Chemistry: The Central Science 8 th Ed. New Jersey: PrenticeHall, 2002. CHEMICAL BONDS The forces that hold atoms or ions together
CHEMISTRY 1: Chemical Bonding Brown, T.L., LeMay, H.E., and B.E. Bursten. Chemistry: The Central Science 8 th Ed. New Jersey: PrenticeHall, 2002. CHEMICAL BONDS The forces that hold atoms or ions together
3/1/2017. Chapter 5. Nomenclature. Naming Compounds. Metal nonmetal. Section 5.1. Binary Compounds: Composed of two elements. Binary Ionic Compounds
 Chapter 5 Nomenclature Section 5.1 Naming Compounds Binary Compounds: Composed of two elements Binary Ionic Compounds Metal nonmetal Copyright Cengage Learning. All rights reserved 2 1 Binary Ionic Compounds
Chapter 5 Nomenclature Section 5.1 Naming Compounds Binary Compounds: Composed of two elements Binary Ionic Compounds Metal nonmetal Copyright Cengage Learning. All rights reserved 2 1 Binary Ionic Compounds
Chemical Nomenclature
 Chemical Nomenclature IUPAC International Union of Pure and Applied Chemistry. This is a group of chemists that determines, among other things, how chemicals will be named. IONIC COMPOUNDS 1. Binary Ionic
Chemical Nomenclature IUPAC International Union of Pure and Applied Chemistry. This is a group of chemists that determines, among other things, how chemicals will be named. IONIC COMPOUNDS 1. Binary Ionic
Chapter 5 Molecules and Compounds
 Chapter 5 Molecules and Compounds 1 Chemical Formulas Atomic/Molecular Elements Ionic/Molecular Compounds Naming Ionic Compounds Naming Molecular Compounds Naming Acids Formula Mass Atomic / Molecular
Chapter 5 Molecules and Compounds 1 Chemical Formulas Atomic/Molecular Elements Ionic/Molecular Compounds Naming Ionic Compounds Naming Molecular Compounds Naming Acids Formula Mass Atomic / Molecular
Nomenclature. HC 2 H 3 O 2 Acetic Acid C 2 H 3 O 2. acetate ion
 Nomenclature C 2 H 3 O 2 - acetate ion HC 2 H 3 O 2 Acetic Acid 1 Forms of Chemical Bonds Most bonds are somewhere in between ionic There are 3 forms bonding atoms: Ionic complete transfer of 1 or more
Nomenclature C 2 H 3 O 2 - acetate ion HC 2 H 3 O 2 Acetic Acid 1 Forms of Chemical Bonds Most bonds are somewhere in between ionic There are 3 forms bonding atoms: Ionic complete transfer of 1 or more
DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE NAME
 DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE NAME - The name of an ionic compound is made of the names of the CATION and ANION in the compound. - To get the FORMULA, you must figure out the SMALLEST
DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE NAME - The name of an ionic compound is made of the names of the CATION and ANION in the compound. - To get the FORMULA, you must figure out the SMALLEST
Nomenclature Hint Sheet
 Nomenclature Hint Sheet The nomenclature for four different classes of compounds is covered in CH101: ionic, covalent, acid/base, and organic compounds. This document will cover ionic (chapter 4) and covalent
Nomenclature Hint Sheet The nomenclature for four different classes of compounds is covered in CH101: ionic, covalent, acid/base, and organic compounds. This document will cover ionic (chapter 4) and covalent
NAMING IONIC COMPOUNDS. ammonium sulfide. iron(ii) carbonate. barium phosphate. titanium(iv) sulfide. Spelling matters!
 67 NAMING IONIC COMPOUNDS ammonium sulfide iron(ii) carbonate titanium(iv) sulfide barium phosphate Spelling matters! barium phosphide 68 DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE NAME - The
67 NAMING IONIC COMPOUNDS ammonium sulfide iron(ii) carbonate titanium(iv) sulfide barium phosphate Spelling matters! barium phosphide 68 DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE NAME - The
Chemistry Chapter 2. Atoms, Molecules, and Ions Section Periodic Table Ions Chemical Bonds Nomenclature
 Chemistry Chapter 2 Atoms, Molecules, and Ions Section 2 2.6-2.8 Periodic Table Ions Chemical Bonds Nomenclature Organization of the Periodic Table Columns, Groups or Families elements in the same vertical
Chemistry Chapter 2 Atoms, Molecules, and Ions Section 2 2.6-2.8 Periodic Table Ions Chemical Bonds Nomenclature Organization of the Periodic Table Columns, Groups or Families elements in the same vertical
Naming Simple Compounds
 Naming Simple Compounds Ionic Compounds Ionic compounds consist of positive and negative ions. have attractions called ionic bonds between positively and negatively charged ions. have high melting and
Naming Simple Compounds Ionic Compounds Ionic compounds consist of positive and negative ions. have attractions called ionic bonds between positively and negatively charged ions. have high melting and
Nomenclature of Inorganic Compounds
 Nomenclature of Inorganic Compounds Chapter 6 Hein and Arena Version 1.1 Eugene Passer Chemistry Department Bronx Community 1 College John Wiley and Sons, Inc Elements and Ions 2 The formula for most elements
Nomenclature of Inorganic Compounds Chapter 6 Hein and Arena Version 1.1 Eugene Passer Chemistry Department Bronx Community 1 College John Wiley and Sons, Inc Elements and Ions 2 The formula for most elements
Inorganic Chemical Formulation. Ms. María Isern
 Inorganic Chemical Formulation Ms. María Isern Oxides: Combination of an element with an oxide O 2- anion To formulate: write the symbol of the element + O 2- and interchange o.n. as subscripts. To name:
Inorganic Chemical Formulation Ms. María Isern Oxides: Combination of an element with an oxide O 2- anion To formulate: write the symbol of the element + O 2- and interchange o.n. as subscripts. To name:
What is the major difference between a molecular formula and formula unit?
 Lecture 5 Molecular Substances versus Ionic Substances Molecular Substance Ionic Substance What are some examples of molecular substances? What are some examples of ionic substances? What is the major
Lecture 5 Molecular Substances versus Ionic Substances Molecular Substance Ionic Substance What are some examples of molecular substances? What are some examples of ionic substances? What is the major
Naming Inorganic Compounds. common names systematic names
 Naming Inorganic Compounds common names systematic names Molecular Common Systematic Formula name name AgCl Lunar caustic Silver chloride H 2 SO 4 Oil of vitriol Sulfuric acid MgSO 4 Epsom salts Magnesium
Naming Inorganic Compounds common names systematic names Molecular Common Systematic Formula name name AgCl Lunar caustic Silver chloride H 2 SO 4 Oil of vitriol Sulfuric acid MgSO 4 Epsom salts Magnesium
Let s Figure out how to name compounds
 Let s Figure out how to name compounds Covalent Bonds Atoms can combine to achieve an octet of valence electrons by sharing electrons. Two fluorine atoms, for example, can form a stable F 2 molecule in
Let s Figure out how to name compounds Covalent Bonds Atoms can combine to achieve an octet of valence electrons by sharing electrons. Two fluorine atoms, for example, can form a stable F 2 molecule in
O ( ) are only used with polyatomic ions and only when there is more than one of any group. a. Examples: HNO 3. ) is incorrect; Al 2 ) 3 (SO 4
 HONORS CHEMISTRY - CHAPTER 9 CHEMICAL NAMES AND FORMULAS NOMENCLATURE PACKET - V16 NAME: DATE: PAGE: I. Writing formulas of ionic compounds when given the component parts. 1. Many compounds are composed
HONORS CHEMISTRY - CHAPTER 9 CHEMICAL NAMES AND FORMULAS NOMENCLATURE PACKET - V16 NAME: DATE: PAGE: I. Writing formulas of ionic compounds when given the component parts. 1. Many compounds are composed
Tustin HS AP Chemistry SUMMER ASSIGNMENT
 Tustin HS AP Chemistry SUMMER ASSIGNMENT Instructor: Email: Ms. Abbey Zinsser azinsser@tustin.k12.ca.us or azinsser@mytusd.org Textbook: Chemistry: The Central Science, Brown, LeMay, Bursten, 11 th edition
Tustin HS AP Chemistry SUMMER ASSIGNMENT Instructor: Email: Ms. Abbey Zinsser azinsser@tustin.k12.ca.us or azinsser@mytusd.org Textbook: Chemistry: The Central Science, Brown, LeMay, Bursten, 11 th edition
Systematic Naming. Chapter 9. Two Types of Compounds. Two Types of Compounds 2 Ionic Compounds. Two Types of Compounds.
 Chapter 9 Naming Compounds Writing Formulas Systematic Naming There are too many compounds to remember the names of them all. Compound is made of two or more elements. Put together atoms. Name should tell
Chapter 9 Naming Compounds Writing Formulas Systematic Naming There are too many compounds to remember the names of them all. Compound is made of two or more elements. Put together atoms. Name should tell
nomenclature nomen name + clatura calling Download the naming reference charts today!
 nomenclature nomen name + clatura calling Download the naming reference charts today! first we learn about binary cmpds, cmpds made of two elements there are two classes of binaries: metal/nonmetal & nonmetal/nonmetal
nomenclature nomen name + clatura calling Download the naming reference charts today! first we learn about binary cmpds, cmpds made of two elements there are two classes of binaries: metal/nonmetal & nonmetal/nonmetal
Worksheet 5 Nomenclature 1 Ionic Compounds I. Naming compounds containing only two elements (binary compounds)
 Worksheet 5 Nomenclature 1 Ionic Compounds I. Naming compounds containing only two elements (binary compounds) 1. Name the first element. 2. Name the second element with an ide ending Example: NaCl A1
Worksheet 5 Nomenclature 1 Ionic Compounds I. Naming compounds containing only two elements (binary compounds) 1. Name the first element. 2. Name the second element with an ide ending Example: NaCl A1
Chemical Names & Formulas. Water Ammonia Methane 1
 Chemical Names & Formulas Water Ammonia Methane 1 Why Systematic Names? # atomic particles 3 (p, n, e) # elements 110+ # elements in 8 earth s crust (99%) # elements in all 25 living things # compounds
Chemical Names & Formulas Water Ammonia Methane 1 Why Systematic Names? # atomic particles 3 (p, n, e) # elements 110+ # elements in 8 earth s crust (99%) # elements in all 25 living things # compounds
Ch.2: Atoms, Molecules, and Ions
 Ch.2: Atoms, Molecules, and Ions Naming Recall Ionic Bond = electrostatic attraction due to the transfer of vse - s between a metal and nonmetal Covalent Bond = sharing of valence electrons between nonmetals
Ch.2: Atoms, Molecules, and Ions Naming Recall Ionic Bond = electrostatic attraction due to the transfer of vse - s between a metal and nonmetal Covalent Bond = sharing of valence electrons between nonmetals
World of Chemistry Notes for Students [Chapter 4, page 1] Chapter 4 Nomenclature
![World of Chemistry Notes for Students [Chapter 4, page 1] Chapter 4 Nomenclature World of Chemistry Notes for Students [Chapter 4, page 1] Chapter 4 Nomenclature](/thumbs/78/77609914.jpg) World of Chemistry Notes for Students [Chapter 4, page 1] Chapter 4 Nomenclature 1) The Periodic Table Review from Chapter The elements are arranged in rows and columns on the Periodic Table according
World of Chemistry Notes for Students [Chapter 4, page 1] Chapter 4 Nomenclature 1) The Periodic Table Review from Chapter The elements are arranged in rows and columns on the Periodic Table according
- Use the STEM NAME of the element, then add "-ide" suffix
 65 ANIONS 2 kinds Main-group nonmetals - Use the STEM NAME of the element, then add "-ide" suffix N : "nitride" ion P : "phosphide ion" O : "oxide ion" F : "fluoride ion" Polyatomic ions - Memorize list.(see
65 ANIONS 2 kinds Main-group nonmetals - Use the STEM NAME of the element, then add "-ide" suffix N : "nitride" ion P : "phosphide ion" O : "oxide ion" F : "fluoride ion" Polyatomic ions - Memorize list.(see
- Some MOLECULES can gain or lose electrons to form CATIONS or ANIONS. These are called POLYATOMIC IONS
 63 POLYATOMIC IONS - Some MOLECULES can gain or lose electrons to form CATIONS or ANIONS. These are called POLYATOMIC IONS - Polyatomic ions form ionic compounds in the same way that single-element ions
63 POLYATOMIC IONS - Some MOLECULES can gain or lose electrons to form CATIONS or ANIONS. These are called POLYATOMIC IONS - Polyatomic ions form ionic compounds in the same way that single-element ions
Extra Credit Directions
 Extra Credit Directions 1. Everything must be correct in order to receive extra credit. There is no partial extra credit. 2. Using the provided charts, you will end in one of the outlined boxes that have
Extra Credit Directions 1. Everything must be correct in order to receive extra credit. There is no partial extra credit. 2. Using the provided charts, you will end in one of the outlined boxes that have
Honors Chemistry - Unit 5 Chapter 7 - Nomenclature
 Honors Chemistry - Unit 5 Chapter 7 - Nomenclature Unit 5 Packet - Page 1 of 16 Vocab Due: Quiz(zes): 1) Test Date: UT Quest: 2) VOCABULARY: 1) monatomic ion 2) cation 3) anion 4) binary compound 4) ionic
Honors Chemistry - Unit 5 Chapter 7 - Nomenclature Unit 5 Packet - Page 1 of 16 Vocab Due: Quiz(zes): 1) Test Date: UT Quest: 2) VOCABULARY: 1) monatomic ion 2) cation 3) anion 4) binary compound 4) ionic
Personal Guided Inquiry: Naming Chemical Compounds and Writing Formulas
 Personal Guided Inquiry: Naming Chemical Compounds and Writing Formulas As you work through the online videos, make notes of important ideas and practice what you are learning. These skills are some of
Personal Guided Inquiry: Naming Chemical Compounds and Writing Formulas As you work through the online videos, make notes of important ideas and practice what you are learning. These skills are some of
Ionic Compounds. And Acids
 CHAPTER 7 LANGUAGE OF CHEMISTRY CLASSIFICATION OF COMPOUNDS Inorganic compounds does not contain the element carbon, but there are exception to this rule, CO 2 (carbon dioxide), CO 3 2 (carbonate), and
CHAPTER 7 LANGUAGE OF CHEMISTRY CLASSIFICATION OF COMPOUNDS Inorganic compounds does not contain the element carbon, but there are exception to this rule, CO 2 (carbon dioxide), CO 3 2 (carbonate), and
4.0-Ionic Compounds Unit
 4.0-Ionic Compounds Unit Objectives: --Given formula, determine if compound is ionic, molecular, or an acid. --Given name, write formula --Given formula, write name --Understand how ionic compounds form
4.0-Ionic Compounds Unit Objectives: --Given formula, determine if compound is ionic, molecular, or an acid. --Given name, write formula --Given formula, write name --Understand how ionic compounds form
Chemical Nomenclature
 Name Period Date Chemical Nomenclature Fill-in the blanks during the PowerPoint presentation in class. Common Names A lot of chemicals have common names as well as the proper (International Union of Pure
Name Period Date Chemical Nomenclature Fill-in the blanks during the PowerPoint presentation in class. Common Names A lot of chemicals have common names as well as the proper (International Union of Pure
Chapter 3 Molecules, Compounds, and Chemical Equations
 Chapter 3 Molecules, Compounds, and Chemical Equations Molecular View of Elements and Compounds 2 How do atom join together to form a compound? compounds are made of atoms held together by chemical bonds
Chapter 3 Molecules, Compounds, and Chemical Equations Molecular View of Elements and Compounds 2 How do atom join together to form a compound? compounds are made of atoms held together by chemical bonds
Chapter 7, Sections 1-2
 Chapter 7, Sections 1-2 Chemical Formulas & Compounds Nomenclature and Oxidation numbers C. Goodman, Doral Preparatory Academy, 2011-2014 Based on a PowerPoint by Mrs. S. Temple, Essential Questions Section
Chapter 7, Sections 1-2 Chemical Formulas & Compounds Nomenclature and Oxidation numbers C. Goodman, Doral Preparatory Academy, 2011-2014 Based on a PowerPoint by Mrs. S. Temple, Essential Questions Section
Chapter 5: Nomenclature
 Chem 1025 Prof George W.J. Kenney, Jr Introductory Chemistry, Zumdahl Decoste, 6th ed Last Update: 21July09 Chapter 5: Nomenclature These Notes are to SUPPLIMENT the Text, They do NOT Replace reading the
Chem 1025 Prof George W.J. Kenney, Jr Introductory Chemistry, Zumdahl Decoste, 6th ed Last Update: 21July09 Chapter 5: Nomenclature These Notes are to SUPPLIMENT the Text, They do NOT Replace reading the
Molecules and Compounds
 Molecules and Compounds ed. Brad Collins Some images Copyright The McGraw-Hill Companies, Inc. A molecule is an aggregate of two or more atoms in a definite arrangement held together by chemical bonds
Molecules and Compounds ed. Brad Collins Some images Copyright The McGraw-Hill Companies, Inc. A molecule is an aggregate of two or more atoms in a definite arrangement held together by chemical bonds
Chemistry Worksheet. Name of Ion Metallic Ion or Nonmetallic ion? Formula. a. ions, also known as, are derived. from metals, and ions, also known as,
 Chemistry Worksheet Nomenclature Review Name: Block: Date: 1. Review of charges and names of ions. Use your periodic table to fill in the following table of monatomic ions. [Prerequisite reading/reference
Chemistry Worksheet Nomenclature Review Name: Block: Date: 1. Review of charges and names of ions. Use your periodic table to fill in the following table of monatomic ions. [Prerequisite reading/reference
Nomenclature. Common Names. Common Names COMPOUNDS FORMED FROM IONS. Binary Ionic Compounds
 PO 4 3- phosphate ion C 2 H 3 O 2 - acetate ion World of Chemistry: Chapter 4 Nomenclature HC 2 H 3 O 2 Acetic Acid Common Names Common Names A lot of chemicals have common lot of chemicals have common
PO 4 3- phosphate ion C 2 H 3 O 2 - acetate ion World of Chemistry: Chapter 4 Nomenclature HC 2 H 3 O 2 Acetic Acid Common Names Common Names A lot of chemicals have common lot of chemicals have common
WRITING FORMULAS AND NAMING COMPOUNDS IONIC COMPOUNDS
 WRITING FORMULAS AND NAMING COMPOUNDS IONIC COMPOUNDS One of the most important concepts to understand when naming and writing ionic compounds is the concept of charge. You must be able to predict which
WRITING FORMULAS AND NAMING COMPOUNDS IONIC COMPOUNDS One of the most important concepts to understand when naming and writing ionic compounds is the concept of charge. You must be able to predict which
Nomenclature. Symbols for Atoms, Molecules, and Ions. Ion Example: Ionic Bonding: Atoms:
 Symbols for Atoms, Molecules, and Ions Nomenclature (Naming) Chemistry 11 Atoms: Remember: the atomic number = # of protons The atomic number = # of electrons in a neutral element # of neutrons = mass
Symbols for Atoms, Molecules, and Ions Nomenclature (Naming) Chemistry 11 Atoms: Remember: the atomic number = # of protons The atomic number = # of electrons in a neutral element # of neutrons = mass
High School Chemistry - Problem Drill 08: Naming Chemical Compounds
 High School Chemistry - Problem Drill 08: Naming Chemical Compounds No. 1 of 10 1. Which of the following is correct for FeO? (A) Iron monoxide (B) Iron oxide (C) Iron (II) oxide (D) Iron (III) oxide (E)
High School Chemistry - Problem Drill 08: Naming Chemical Compounds No. 1 of 10 1. Which of the following is correct for FeO? (A) Iron monoxide (B) Iron oxide (C) Iron (II) oxide (D) Iron (III) oxide (E)
Chapter 5. Molecules and Compounds. Introductory Chemistry, 3 rd Edition Nivaldo Tro 2/21/2011
 Introductory Chemistry, 3 rd Edition Nivaldo Tro Chapter 5 Molecules and Compounds Based on notes of Roy Kennedy Massachusetts Bay Community College 2006, Prentice Hall Molecules and Compounds Salt Sodium
Introductory Chemistry, 3 rd Edition Nivaldo Tro Chapter 5 Molecules and Compounds Based on notes of Roy Kennedy Massachusetts Bay Community College 2006, Prentice Hall Molecules and Compounds Salt Sodium
Title: Chem Review 4 PART 1 TOPIC: TYPES OF BONDS. EQ: What is an Ionic Bond?
 Title: Chem Review 4 PART 1 TOPIC: TYPES OF BONDS EQ: What is an Ionic Bond? Title and Highlight Topic: EQ: Date Reflect Question: Reflect on the material by asking a question (its not suppose to be answered
Title: Chem Review 4 PART 1 TOPIC: TYPES OF BONDS EQ: What is an Ionic Bond? Title and Highlight Topic: EQ: Date Reflect Question: Reflect on the material by asking a question (its not suppose to be answered
Compounds Element = 1 type of atom Compound = more than 1 type of atom (over 8 million) Chemical Bond = glue that links atoms together in a compound
 Compounds Element = 1 type of atom Compound = more than 1 type of atom (over 8 million) Chemical Bond = glue that links atoms together in a compound Ionic Compounds Ionic Bonds = atoms switch e- forming
Compounds Element = 1 type of atom Compound = more than 1 type of atom (over 8 million) Chemical Bond = glue that links atoms together in a compound Ionic Compounds Ionic Bonds = atoms switch e- forming
CHEM 103 Naming Compounds
 CHEM 103 Naming Compounds Lecture Notes February 9, 2006 Prof. Sevian Chem 103 Please sit with your groups today. We will be doing a group problem at the end of class. 2 2005 H. Sevian 1 Agenda How we
CHEM 103 Naming Compounds Lecture Notes February 9, 2006 Prof. Sevian Chem 103 Please sit with your groups today. We will be doing a group problem at the end of class. 2 2005 H. Sevian 1 Agenda How we
Nomenclature. Why, Learn it? History:
 Nomenclature Why, Learn it? Allow us to be able to name compound we have never seen before. If we know the name of a compound we can determine its formula. We can look at a formula, determine it name and
Nomenclature Why, Learn it? Allow us to be able to name compound we have never seen before. If we know the name of a compound we can determine its formula. We can look at a formula, determine it name and
Chapter 2. Chapter 2
 Chapter 2 Atoms, Molecules, and Ions Chapter 2 Atomic Theory Model for atom and ions that works well for chemistry Molecules Combinations of atoms Ways of representing molecules: formulas and models Names
Chapter 2 Atoms, Molecules, and Ions Chapter 2 Atomic Theory Model for atom and ions that works well for chemistry Molecules Combinations of atoms Ways of representing molecules: formulas and models Names
Nomenclature Report. GCC CHM 151LL: Nomenclature Report GCC, 2019 page 1 of 6
 Nomenclature Report Introduction Ionic Compound Nomenclature An ionic compound is described as an electrostatic attractive force between oppositely charged ions. Monoatomic Cations Metal cations are formed
Nomenclature Report Introduction Ionic Compound Nomenclature An ionic compound is described as an electrostatic attractive force between oppositely charged ions. Monoatomic Cations Metal cations are formed
DETERMINING IONIC FORMULAS strontium oxide
 69 sodium sulfate DETERMINING IONIC FORMULAS strontium oxide tin(ii) phosphate chromium(iii) nitrate barium hydroxide titanium(iv) chloride When adding a subscript to a polyatomic ion, put the polyatomic
69 sodium sulfate DETERMINING IONIC FORMULAS strontium oxide tin(ii) phosphate chromium(iii) nitrate barium hydroxide titanium(iv) chloride When adding a subscript to a polyatomic ion, put the polyatomic
Bonding and Nomenclature notes.notebook
 Chemical Bonding & Nomenclature Objectives: Distinguish between covalent and ionic bonding Explain the process of bonding Name ionic and covalent compounds and acids Write chemical formulas for ionic and
Chemical Bonding & Nomenclature Objectives: Distinguish between covalent and ionic bonding Explain the process of bonding Name ionic and covalent compounds and acids Write chemical formulas for ionic and
Chapter 9 Study Guide
 Name: Class: Date: Chapter 9 Study Guide Matching Match each item with the correct statement below. a. monatomic ion f. cation b. acid g. binary compound c. base h. anion d. law of definite proportions
Name: Class: Date: Chapter 9 Study Guide Matching Match each item with the correct statement below. a. monatomic ion f. cation b. acid g. binary compound c. base h. anion d. law of definite proportions
NAMING IONIC COMPOUNDS
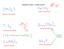 NAMING IONIC COMPOUNDS iron(ii) carbonate ammonium sulfide titanium (IV) sulfide barium phosphate Spelling matters! calcium nitrate sodium phosphide DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE
NAMING IONIC COMPOUNDS iron(ii) carbonate ammonium sulfide titanium (IV) sulfide barium phosphate Spelling matters! calcium nitrate sodium phosphide DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE
Chapter 9 Practice Test
 Name: Class: Date: Chapter 9 Practice Test Multiple Choice Identify the choice that best completes the statement or answers the question. ****You need to know all the rules for naming ionic, molecular
Name: Class: Date: Chapter 9 Practice Test Multiple Choice Identify the choice that best completes the statement or answers the question. ****You need to know all the rules for naming ionic, molecular
ATOMS, MOLECULES AND IONS
 2 ATOMS, MOLECULES AND IONS LECTURE NOTES Students find this material relatively easy to assimilate; it s almost entirely qualitative. On the other hand, there s a lot of memorizing (sorry, learning) to
2 ATOMS, MOLECULES AND IONS LECTURE NOTES Students find this material relatively easy to assimilate; it s almost entirely qualitative. On the other hand, there s a lot of memorizing (sorry, learning) to
Ionic Compound: Metal + Nonmetal
 Chemical Nomenclature: Writing Names and Formulas Bonding is the way atoms are attracted to each other to form compounds. During bonding atoms gain, lose, or share electrons to reach stable configurations.
Chemical Nomenclature: Writing Names and Formulas Bonding is the way atoms are attracted to each other to form compounds. During bonding atoms gain, lose, or share electrons to reach stable configurations.
Nomenclature Naming Ionic Compounds Worksheet #1
 Naming Ionic Compounds Worksheet #1 In forming ionic compounds with non-metals, the transition metals often exhibit more than one valence. For example, in the reaction between iron and chlorine, two products
Naming Ionic Compounds Worksheet #1 In forming ionic compounds with non-metals, the transition metals often exhibit more than one valence. For example, in the reaction between iron and chlorine, two products
AP Chemistry Unit 1 Review Guide: IUPAC Naming, Stoichiometry, Solution Chemistry
 I. IUPAC Naming AP Chemistry Unit 1 Review Guide: IUPAC Naming, Stoichiometry, Solution Chemistry For Ionic Compounds: Formula to Name: 1. Identify the cation (positive ion) by name, then identify the
I. IUPAC Naming AP Chemistry Unit 1 Review Guide: IUPAC Naming, Stoichiometry, Solution Chemistry For Ionic Compounds: Formula to Name: 1. Identify the cation (positive ion) by name, then identify the
Chemical Names and Formulas
 Chemical Names and Formulas ELECTRONS AND THE STRUCTURE OF ATOMS BONDING AND INTERACTIONS 91 Naming Ions For students using the Foundation edition, assign problems 4, 8 15 Essential Understanding Ions
Chemical Names and Formulas ELECTRONS AND THE STRUCTURE OF ATOMS BONDING AND INTERACTIONS 91 Naming Ions For students using the Foundation edition, assign problems 4, 8 15 Essential Understanding Ions
Chapter 4 Compounds and Their Bonds. Octet Rule. Metals Form Positive Ions. Ionic and Covalent Bonds. Formation of a Sodium Ion, Na +
 Chapter 4 Compounds and Their Bonds Octet Rule 4.1 Octet Rule and Ions An octet Is 8 valence electrons. Is associated with the stability of the noble gases. Helium (He) is stable with 2 valence electrons
Chapter 4 Compounds and Their Bonds Octet Rule 4.1 Octet Rule and Ions An octet Is 8 valence electrons. Is associated with the stability of the noble gases. Helium (He) is stable with 2 valence electrons
Chapter 9. Chemical Names and Formulas
 Chapter 9 Chemical Names and Formulas 9.1 - Naming Ions Monatomic ions: Single atom with a positive or negative charge resulting from the loss or gain of one or more valence electrons. - Cations: Groups
Chapter 9 Chemical Names and Formulas 9.1 - Naming Ions Monatomic ions: Single atom with a positive or negative charge resulting from the loss or gain of one or more valence electrons. - Cations: Groups
Honors Chemistry - Topic IV Part I Study Guide The Language of Chemistry
 Honors Chemistry - Topic IV Part I Study Guide The Language of Chemistry I. Chemical Nomenclature A. Inorganic Nomenclature (pp.11132; 349-350, 554-555; 213-220) 1. Types of formulas chart showing positions
Honors Chemistry - Topic IV Part I Study Guide The Language of Chemistry I. Chemical Nomenclature A. Inorganic Nomenclature (pp.11132; 349-350, 554-555; 213-220) 1. Types of formulas chart showing positions
Nomenclature PO 4. phosphate ion. HC 2 H 3 O 2 Acetic Acid C 2 H 3 O 2. acetate ion
 PO 4 3- phosphate ion Nomenclature HC 2 H 3 O 2 Acetic Acid C 2 H 3 O 2 - acetate ion SAVE PAPER AND INK!!! When you print out the notes on PowerPoint, print "Handouts" instead of "Slides" in the print
PO 4 3- phosphate ion Nomenclature HC 2 H 3 O 2 Acetic Acid C 2 H 3 O 2 - acetate ion SAVE PAPER AND INK!!! When you print out the notes on PowerPoint, print "Handouts" instead of "Slides" in the print
Naming Compounds Tutorial and Worksheet
 Naming Compounds Tutorial and Worksheet Since we use different methods in naming binary covalent (molecular) compounds and ionic compounds, the first step in naming or writing the formula of a compound
Naming Compounds Tutorial and Worksheet Since we use different methods in naming binary covalent (molecular) compounds and ionic compounds, the first step in naming or writing the formula of a compound
Ch2.0 Lesson.notebook. February 08, Bell Activity. How do you think chemicals get their name? Who gives them their name?
 Bell Activity How do you think chemicals get their name? Who gives them their name? 1 Introduction to Naming 2 IUPAC system ensures that each pure substance has a single unique name. This is called its
Bell Activity How do you think chemicals get their name? Who gives them their name? 1 Introduction to Naming 2 IUPAC system ensures that each pure substance has a single unique name. This is called its
Ion formation: Writing formulae and names for ionic compounds. Having fun with nomenclature! element 1 Ca 2 electrons lost. nonmetallic.
 Ion formation: Symbol of Change in electrons element 1 Ca 2 electrons lost Formula of ion Name of ion Metallic or nonmetallic 2 F F 1-3 Al 3+ 4 Zn 2 electrons lost 5 O 2-6 Cs 1 electron lost 7 Ba 2+ 8
Ion formation: Symbol of Change in electrons element 1 Ca 2 electrons lost Formula of ion Name of ion Metallic or nonmetallic 2 F F 1-3 Al 3+ 4 Zn 2 electrons lost 5 O 2-6 Cs 1 electron lost 7 Ba 2+ 8
CHAPTER 7: LANGUAGE OF CHEMISTRY
 CHAPTER 7: LANGUAGE OF CHEMISTRY Problems: 3-12, 13(a-c), 14(a,c,d), 15(a-c), 16(a,c), 17-19, 20c, 21, 22(a,c), 23-27, 33(a,c,d), 34(a,c,d), 35, 36(a,c), 37(a,b,d), 43-48, 49b, 50b, 51-52, 59-62 7.1 CLASSIFICATION
CHAPTER 7: LANGUAGE OF CHEMISTRY Problems: 3-12, 13(a-c), 14(a,c,d), 15(a-c), 16(a,c), 17-19, 20c, 21, 22(a,c), 23-27, 33(a,c,d), 34(a,c,d), 35, 36(a,c), 37(a,b,d), 43-48, 49b, 50b, 51-52, 59-62 7.1 CLASSIFICATION
Why does an element want to bond?
 Why does an element want to bond? State 3 differences between ionic vs. covalent compounds What is a chemical formula? It indicates the relative number of atoms of each kind in an ionic compound. Ex Al
Why does an element want to bond? State 3 differences between ionic vs. covalent compounds What is a chemical formula? It indicates the relative number of atoms of each kind in an ionic compound. Ex Al
Naming Ionic Compounds with Two Elements
 Chapter 6 Lecture Chapter 6 Ionic and Molecular Compounds 6.3 Naming and Writing Ionic Compounds Fifth Edition Naming of Ionic Compounds In the name of an ionic compound, the positive ion (first ion) is
Chapter 6 Lecture Chapter 6 Ionic and Molecular Compounds 6.3 Naming and Writing Ionic Compounds Fifth Edition Naming of Ionic Compounds In the name of an ionic compound, the positive ion (first ion) is
CHEMISTRY 103 Help Sheet #2 Atoms, Molecules, and Ions (Text: Ch 2: )
 CHEMISTRY 103 Help Sheet #2 Atoms, Molecules, and Ions (Text: Ch 2: 2.1-2.8) Do the topics appropriate for your lecture Prepared by Dr. Tony Jacob http://www.chem.wisc.edu/areas/clc (Resource page) Nuggets:
CHEMISTRY 103 Help Sheet #2 Atoms, Molecules, and Ions (Text: Ch 2: 2.1-2.8) Do the topics appropriate for your lecture Prepared by Dr. Tony Jacob http://www.chem.wisc.edu/areas/clc (Resource page) Nuggets:
Chemical Nomenclature
 Chemical Nomenclature I. The reason that we care about naming compounds a. Naming, a.k.a. nomenclature of compounds b. Significance: must know what we are talking about very clearly when we refer to substances
Chemical Nomenclature I. The reason that we care about naming compounds a. Naming, a.k.a. nomenclature of compounds b. Significance: must know what we are talking about very clearly when we refer to substances
CHAPTER 7: CHEMICAL FORMULAS AND CHEMICAL COMPOUNDS. Chemistry 1-2 Enriched Mr. Chumbley
 CHAPTER 7: CHEMICAL FORMULAS AND CHEMICAL COMPOUNDS Chemistry 1-2 Enriched Mr. Chumbley SECTION 1: CHEMICAL NAMES AND FORMULAS While the bulk of the content will come from the Naming Chemical Compounds
CHAPTER 7: CHEMICAL FORMULAS AND CHEMICAL COMPOUNDS Chemistry 1-2 Enriched Mr. Chumbley SECTION 1: CHEMICAL NAMES AND FORMULAS While the bulk of the content will come from the Naming Chemical Compounds
Chapter 6 Inorganic and Organic Compounds: Names and Formulas
 Chapter 6 Inorganic and Organic Compounds: Names and Formulas 6.1 Octet Rule and Ions 1 Octet Rule An octet is 8 valence electrons is associated with the stability of the noble gases does not occur with
Chapter 6 Inorganic and Organic Compounds: Names and Formulas 6.1 Octet Rule and Ions 1 Octet Rule An octet is 8 valence electrons is associated with the stability of the noble gases does not occur with
- Some MOLECULES can gain or lose electrons to form CATIONS or ANIONS. These are called POLYATOMIC IONS
 63 POLYATOMIC IONS - Some MOLECULES can gain or lose electrons to form CATIONS or ANIONS. These are called POLYATOMIC IONS - Polyatomic ions form ionic compounds in the same way that single-element ions
63 POLYATOMIC IONS - Some MOLECULES can gain or lose electrons to form CATIONS or ANIONS. These are called POLYATOMIC IONS - Polyatomic ions form ionic compounds in the same way that single-element ions
OWL Assignment #2 Study Sheet
 OWL Assignment #2 Study Sheet Binary Acid Nomenclature Binary compounds are composed of two elements. When one of the elements is a binary acid can be formed. Examples of this are HCl or H 2 S. When put
OWL Assignment #2 Study Sheet Binary Acid Nomenclature Binary compounds are composed of two elements. When one of the elements is a binary acid can be formed. Examples of this are HCl or H 2 S. When put
