Catalytic oxidations: finding the optimum composition of AuPd coreshell nanoparticle catalysts
|
|
|
- Tyrone Hood
- 5 years ago
- Views:
Transcription
1 Brazilian ChemComm Symposium Chemistry and Sustainable Energy 5 th November 2012, São Paulo, Brazil Catalytic oxidations: finding the optimum composition of AuPd coreshell nanoparticle catalysts Prof. Dr. Liane M. Rossi Laboratory of Nanomaterials and Catalysis Instituto de Química Universidade de São Paulo Av. Prof. Lineu Prestes 748 São Paulo , SP Brasil lrossi@iq.usp.br
2 Catalytic oxidations are widely used in the manufacture of bulk petrochemicals, but are not a commonplace in the fine chemicals and pharmaceutical industry, and at the organic laboratory level: Stoichiometric Oxidations are very far from being ideal from the green point of view! hazardous or toxic chemicals volatile organic solvents large amounts of toxic wastes Oxidizing reagent Residue KMnO 4 Mn 2+ /MnO 2 K 2 CrO 4 Cr 3+ CH 3 COOOH t-buooh CH 3 COOH t-buoh ClO - Cl - H 2 O 2 O 2 H 2 O H 2 O
3 Green oxidizing agents, O 2 and H 2 O 2, do not readily react in a selective way with organic substrates, unless a catalyst is present. Catalytic Oxidations Control the reactivity of oxygen species to obtain valuable organic oxygenates and avoid overoxidation. R-CH 2 -OH R-CHO R-COOH CO 2 + H 2 O Discriminate functional groups in the same molecule. Oxidations in fine chemicals is generally more difficult, however, owing to the multifunctional nature of the molecules of interest.
4 Development of metal nanoparticle catalyst *Gold was discovered as an active catalyst in the late 80s after the seminal contribution of Haruta and Hutching.
5 Metal nanoparticle catalyst Soluble NPs high control on particle size, size distribution and surface chemistry Supported NPs Future Targets Control on particle size, size distribution and uniform dispersion of NPs on solid supports - dial up the active sites Prevent metal leaching Improve metal recovery Understand the role of stabilizers on metal NP catalysis
6 Supported metal NPs Immobilization of pre-formed metal NPs Metal salt impregnation and reduction method Catalyst Support M x+ M x+ M x+ M x+ M x+ M x+ M x+ Poor control on metal dispersion Particles size and sized distribution
7 Supported metal NPs Ligand-assisted method Nanoscale, 2012, 4, Catalyst Support M x+ M x+ M x+ M x+ M x+ M M x+ x+ R= NH 2 R= NH NH 2 None Si(OR) 3 = NH 2, en, COOH, SH, PR 2,... Tune NPs size with uniform dispersion of NPs on supports Inorganic Chemistry, 2009, 48, 4640.
8 Supported metal NPs Ligand-assisted method Nanoscale, 2012, 4, Catalyst Support M x+ M x+ M x+ M x+ M x+ M M x+ x+ Si(OR) 3 = NH 2, en, COOH, SH, PR 2,... Improve metal recovery using magnetic support Applied Catalysis. A, General, 2008, 338, 52.
9 Supported metal NPs Ligand-assisted method Nanoscale, 2012, 4, Catalyst Support M x+ M x+ M x+ M x+ M x+ M M x+ x+ Si(OR) 3 = NH 2, en, COOH, SH, PR 2,... Low metal leaching ChemCatChem, 2012, 4, 698.
10 Supported metal NPs Ligand-assisted method: metal support interaction Chemistry A European Journal, 2011, 17, 4626.
11 t Supported metal NPs Ligand-assisted method: metal support interaction X-ray absorption fine structure spectroscopy studies Au 3+ Au 3+ Au 3+ Au (a) (b) (c) Au L 3 -edge SiO 2 -Au (d) Au σ+ H 2 N H 2 N Au σ+ NH 2 NH 2 NH 2 O O Si O Si O O O O Si O O O O Si O Au σ+ NH 2 NH 2 Au σ+ SiO 2 -NH 2 -Au 3+ NH (e) Energy / ev (a) Au(CH 3 COO) 3 (b) SiO 2 -Au 3+ (c) HAuCl 4 (d) SiO 2 -NH 2 -Au (e) Au foil Chemistry A European Journal, 2011, 17, 4626.
12 Selected examples of magnetically recoverable catalysts Rh NPs PtNPs Ir NPs Ru NPs Applied Catalysis. A, General, 2008, 338, 52. Catalysis Communications, 2009, 10, ChemCatChem, 2012, 4, 698. Applied Catalysis. A, General, 2009, 360, 177. Applied Catalysis. B, Environmental, 2009, 90, 688.
13 Selected examples of magnetically recoverable catalysts NiNPs PdNPs AuNPs ACS Catalysis, 2012, 2, 925. Inorganic Chemistry., 2009, 48, Appl. Catal. B, Environ., 2010, 100, 42. Journal of Catalysis, 2010, 276, 382. Chemistry A European Journal, 2011, 17, Green Chemistry, 2010, 12, 144. Green Chemistry, 2009, 11, 1366.
14 Conversion (%) Supported Au NP catalysts AuNPs Oxidation of benzyl alcohol K 2 CO 3 = high selectivity and conversion rates, but low catalyst stability In the search for a more stable catalysts, we first chose to adhere to the literature by adding Pd to our supported gold catalyst 100 Selectivity =96% Chemistry A European Journal, 2011, 17, Green Chemistry, 2010, 12, 144. Green Chemistry, 2009, 11, K 2 CO 3 KOH Et 3 N absence of base
15 Considerations Open question Supported Au NP catalysts AuNPs... AuPdNPs Bimetallic NPs = metallic domain distributions: alloys (AB) or core-shell (A@B or B@A) NPs How much Pd should be added to activate Au NPs? Chemistry A European Journal, 2011, 17, Green Chemistry, 2010, 12, 144. Green Chemistry, 2009, 11, AuPd alloy NPs have received special attention in catalytic applications. However, the surface of an AuPd alloy NP differs from its corresponding bulk concentration Core-shell NPs can be obtained by the reduction of palladium over pre-formed gold NPs, and vice versa
16 Supported AuPd NP catalysts Oxidation of benzyl alcohol Chemistry A European Journal, 2011, 17, 4626.
17 Supported AuPd NP catalysts Catalytic performance of the catalysts in the oxidation reaction with benzyl alcohol. The amount of Au is fixed (3.4 mol), while the amount of Pd varies from 0 to 40 mol % (i.e., 0 to 1.4 mol). Reaction conditions: 1 ml (10 mmol) benzyl alcohol, 75 mg catalyst (3.4 µmolau), 0 to 1.4 µmol Pd(OAc) 2, 6 bar O 2, 2.5 h, 100 C.
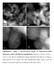
18 Supported AuPd NP catalysts morphologically structured Au-rich core and a Pd-rich shell TEM and HAADF-STEM image of a Au:Pd = 10:1 supported catalyst particle and the respective Au and Pd maps. The particle compositional distribution is observed in the line scans, measured from the regions delimited by the lines indicated in both maps. The hemispherical shape observed in the Au line scan contrasts with the flat distribution measured for Pd, which shows its concentration at the particle shell.
19 Supported AuPd NP catalysts morphologically structured Au-rich core and a Pd-rich shell (a) BF-STEM image of a supported catalyst particle Au:Pd = 5:2 (b) HAADF-STEM image of the supported catalyst particle and the respective Au and Pd maps. The particle compositional distribution is observed in the line scans, measured from the regions delimited by the lines indicated in both maps. The hemispherical shape observed in the Au line scan contrasts with the flat distribution measured for Pd, which shows its concentration at the particle shell.
20 Supported AuPd NP catalysts morphologically structured Au-rich core and a Pd-rich shell Catalytic performance of the Au@Pd catalysts in the oxidation reaction with benzyl alcohol. The amount of Au is fixed (3.4 mol), while the amount of Pd varies from 0 to 40 mol % (i.e., 0 to 1.4 mol). Reaction conditions: 1 ml (10 mmol) benzyl alcohol, 75 mg catalyst (3.4 µmolau), 0 to 1.4 µmol Pd(OAc) 2, 6 bar O 2, 2.5 h, 100 C.
21 Supported AuPd NP catalysts Full-shell cluster model: The most active catalyst, with 89.9% Au and 9.1% Pd, is very close to the nominal composition for the complete coverage of Au cores ( nm) with one atomic layer of Pd.
22 Activity Final Remarks Hypothesis based on morphological and catalytic studies: the deposition of one atomic layer of Pd on Au resulted in a Au core-pd-rich shell catalyst of maximum activity Pd added
23 Final Remarks The Au:Pd molar ratio needed to form a monolayer of Pd might change as a function of Au core size. Consequently, one can expect the maximum activity to occur at differing AuPd compositions when using Au core size or size distribution other than the one we used in our study. ~3 nm AuNP ~45% surface atoms ~40 mol% Pd for monolayer ~10 nm AuNP ~16% surface atoms ~15 mol% Pd for monolayer ~20 nm AuNP ~8% surface atoms ~8 mol% Pd for monolayer Experiments in progress!
24 Final Remarks Au core Pd-rich shell: the distribution of metal domains and the Au:Pd ratio are both important for the synergistic effect observed. high selectivity of Au high activity of Pd High activity and selectivity meeting the increasing demand for environmentally friendly chemical processes
25 ACKNOWLEDGMENTS Group Tiago Artur da Silva - PhD Fernanda Parra da Silva -PhD Lucas L. R. Vono - PhD Marco Aurélio S. Garcia - PhD Natália J. S. Costa Post Doc Jean-Claudio Costa Post Doc Leonardo Gomes Santos Undergrad Bruna Julio - Undergrad Rafael L. Oliveira Inna M. Nangoi Marcos J. Jacinto Fernando B. Effenberger Collaboration Pedro K. Kiyohara (IF/USP) Renato F. Jardim (IF/USP) Richard Landers (Unicamp) Daniela Zanchet (Unicamp) Érico Teixeira-Neto (IQ-USP) Elena Goussevskaia (UFMG) Paulo A. Z. Suarez (UnB) Joel C. Rubim (UnB) Karine Philippot (LCC/CNRS, Toulouse, France)
PROJECT 20: SUPPORTED METALS NANOPARTICLES AS CATALYST FOR THE PROX REACTION
 PROJECT 20: SUPPORTED METALS NANOPARTICLES AS CATALYST FOR THE PROX REACTION Prof. Elisabete M. Assaf, PhD IQSC - USP Prof. José M. Assaf, PhD; Janaina F. Gomes, PhD; Aline R. L. Miranda, Ms DEQ - UFSCar
PROJECT 20: SUPPORTED METALS NANOPARTICLES AS CATALYST FOR THE PROX REACTION Prof. Elisabete M. Assaf, PhD IQSC - USP Prof. José M. Assaf, PhD; Janaina F. Gomes, PhD; Aline R. L. Miranda, Ms DEQ - UFSCar
Strategies to Synthesize Supported Bimetallic Catalysts
 Strategies to Synthesize Supported Bimetallic Catalysts M. Sankar Cardiff Catalysis Institute School of Chemistry Cardiff University, Cardiff United Kingdom sankar@cardiff.ac.uk Outline Ø Supported Bimetallic
Strategies to Synthesize Supported Bimetallic Catalysts M. Sankar Cardiff Catalysis Institute School of Chemistry Cardiff University, Cardiff United Kingdom sankar@cardiff.ac.uk Outline Ø Supported Bimetallic
The Curious Case of Au Nanoparticles
 The Curious Case of Au Nanoparticles Industrial reactions performed by metals 1 Low Au reactivity Predictions are typically based on d-band model Hold well for polycrystalline materials Coinage metals
The Curious Case of Au Nanoparticles Industrial reactions performed by metals 1 Low Au reactivity Predictions are typically based on d-band model Hold well for polycrystalline materials Coinage metals
Magnetic Silica Particles for Catalysis
 4 Magnetic Silica Particles for atalysis Abstract Monodisperse magnetizable colloidal silica particles in a stable dispersion have been functionalized with a homogeneous catalyst: a PP-pincer Pd-complex.
4 Magnetic Silica Particles for atalysis Abstract Monodisperse magnetizable colloidal silica particles in a stable dispersion have been functionalized with a homogeneous catalyst: a PP-pincer Pd-complex.
One pot synthesis and systematic study of photophysical, magnetic properties and
 Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2018 One pot synthesis and systematic
Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2018 One pot synthesis and systematic
AP Chemistry. 9. Which of the following species CANNOT function as an oxidizing agent? (A) Cr 2 O 72 (B) MnO 4 (C) NO 3 (D) S (E) I
 Name AP Chemistry AP Chemistry Exam Part I: 40 Questions, 40 minutes, Multiple Choice, No Calculator Allowed Bubble the correct answer on your scantron for each of the following. Use the following answers
Name AP Chemistry AP Chemistry Exam Part I: 40 Questions, 40 minutes, Multiple Choice, No Calculator Allowed Bubble the correct answer on your scantron for each of the following. Use the following answers
A Simple and Green Method for Synthesis of Ag and Au Nanoparticles Using Biopolymers and Sugars as Reducing Agents.
 A Simple and Green Method for Synthesis of Ag and Au Nanoparticles Using Biopolymers and Sugars as Reducing Agents. German Ayala, Luci Cristina O. Vercik, Leticcia G. Ferreira, Andres Vercik and Thiago
A Simple and Green Method for Synthesis of Ag and Au Nanoparticles Using Biopolymers and Sugars as Reducing Agents. German Ayala, Luci Cristina O. Vercik, Leticcia G. Ferreira, Andres Vercik and Thiago
Modern Alchemy : Catalysis by Gold Nano-particles: Part 1
 Modern Alchemy : Catalysis by Gold Nano-particles: Part 1 PIRE-ECCI/ICMR Summer Conference SantaBarbara 17 August, 2006 Masatake Haruta Tokyo Metropolitan University 1. Overview on Gold 2. CO Oxidation
Modern Alchemy : Catalysis by Gold Nano-particles: Part 1 PIRE-ECCI/ICMR Summer Conference SantaBarbara 17 August, 2006 Masatake Haruta Tokyo Metropolitan University 1. Overview on Gold 2. CO Oxidation
TYPES OF CHEMICAL REACTIONS
 TYPES OF CHEMICAL REACTIONS Precipitation Reactions Compounds Soluble Ionic Compounds 1. Group 1A cations and NH 4 + 2. Nitrates (NO 3 ) Acetates (CH 3 COO ) Chlorates (ClO 3 ) Perchlorates (ClO 4 ) Solubility
TYPES OF CHEMICAL REACTIONS Precipitation Reactions Compounds Soluble Ionic Compounds 1. Group 1A cations and NH 4 + 2. Nitrates (NO 3 ) Acetates (CH 3 COO ) Chlorates (ClO 3 ) Perchlorates (ClO 4 ) Solubility
Theodore E. Madey. Department of Physics and Astronomy, and Laboratory for Surface Modification
 The Science of Catalysis at the Nanometer Scale Theodore E. Madey Department of Physics and Astronomy, and Laboratory for Surface Modification http://www.physics.rutgers.edu/lsm/ Rutgers, The State University
The Science of Catalysis at the Nanometer Scale Theodore E. Madey Department of Physics and Astronomy, and Laboratory for Surface Modification http://www.physics.rutgers.edu/lsm/ Rutgers, The State University
often display a deep green color due to where the SPR occurs (i.e., the wavelength of light that interacts with this specific morphology).
 Synthesis-Dependent Catalytic Properties of Gold Nanoparticles Nanoscience is the study of materials that have dimensions, intuitively, on the nanoscale, typically between 1 100 nm. This field has received
Synthesis-Dependent Catalytic Properties of Gold Nanoparticles Nanoscience is the study of materials that have dimensions, intuitively, on the nanoscale, typically between 1 100 nm. This field has received
Supplementary Figure 2. (a) XRD patterns of the MOF and the simulated Ni-MOF-74
 Supplementary Figure 1. Low-magnification TEM image of Pt-Ni frame @ MOF. The scale bar is 200 nm. Supplementary Figure 2. (a) XRD patterns of the Pt-Ni @ MOF and the simulated Ni-MOF-74 pattern. (b) XRD
Supplementary Figure 1. Low-magnification TEM image of Pt-Ni frame @ MOF. The scale bar is 200 nm. Supplementary Figure 2. (a) XRD patterns of the Pt-Ni @ MOF and the simulated Ni-MOF-74 pattern. (b) XRD
Student Manual for Aerobic Alcohol Oxidation Using a Copper(I)/TEMPO Catalyst System
 Student Manual for Aerobic Alcohol Oxidation Using a Copper(I)/TEMPO Catalyst System icholas J. Hill, Jessica M. Hoover and Shannon S. Stahl* Department of Chemistry, University of Wisconsin-Madison, 1101
Student Manual for Aerobic Alcohol Oxidation Using a Copper(I)/TEMPO Catalyst System icholas J. Hill, Jessica M. Hoover and Shannon S. Stahl* Department of Chemistry, University of Wisconsin-Madison, 1101
State of São Paulo. Brasil. Localization. Santo André
 Brasil State of São Paulo Localization Santo André The Group www.crespilho.com Frank N. Crespilho Pablo R. S. Abdias Group Leader Synthesis of nanostructured materials using polyelectrolytes Andressa R.
Brasil State of São Paulo Localization Santo André The Group www.crespilho.com Frank N. Crespilho Pablo R. S. Abdias Group Leader Synthesis of nanostructured materials using polyelectrolytes Andressa R.
One-pot Solvent-free Synthesis of Sodium Benzoate from the Oxidation of Benzyl Alcohol over Novel Efficient AuAg/TiO 2 Catalysts
 Electronic Supplementary Information One-pot Solvent-free Synthesis of Sodium Benzoate from the Oxidation of Benzyl Alcohol over Novel Efficient AuAg/TiO 2 Catalysts Ying Wang, Jia-Min Zheng, Kangnian
Electronic Supplementary Information One-pot Solvent-free Synthesis of Sodium Benzoate from the Oxidation of Benzyl Alcohol over Novel Efficient AuAg/TiO 2 Catalysts Ying Wang, Jia-Min Zheng, Kangnian
Green Oxidations with Tungsten Catalysts. by Mike Kuszpit Michigan State University
 Green xidations with Tungsten Catalysts by Mike Kuszpit Michigan State University xidations in rganic Chemistry [] [] R 1 R 1 R 1 [] R 1 R 2 R 1 R 2 [] R 1 R 2 R 1 R 2 R 1 R 2 [] R 1 R 2 Essential as building
Green xidations with Tungsten Catalysts by Mike Kuszpit Michigan State University xidations in rganic Chemistry [] [] R 1 R 1 R 1 [] R 1 R 2 R 1 R 2 [] R 1 R 2 R 1 R 2 R 1 R 2 [] R 1 R 2 Essential as building
Chapter 3: Stoichiometry
 Chapter 3: Stoichiometry Chem 6A Michael J. Sailor, UC San Diego 1 Announcements: Thursday (Sep 29) quiz: Bring student ID or we cannot accept your quiz! No notes, no calculators Covers chapters 1 and
Chapter 3: Stoichiometry Chem 6A Michael J. Sailor, UC San Diego 1 Announcements: Thursday (Sep 29) quiz: Bring student ID or we cannot accept your quiz! No notes, no calculators Covers chapters 1 and
Synthesis and Characterization of Gold-Palladium Nanoparticles Catalyst For Improved Hydrogen Fuel Cell Performance
 Synthesis and Characterization of Gold-Palladium Nanoparticles Catalyst For Improved Hydrogen Fuel Cell Performance Adam Bennett a, Helen Liu a, Allen Tran a, Likun Wang b, Miriam Rafailovich a,b* a,b
Synthesis and Characterization of Gold-Palladium Nanoparticles Catalyst For Improved Hydrogen Fuel Cell Performance Adam Bennett a, Helen Liu a, Allen Tran a, Likun Wang b, Miriam Rafailovich a,b* a,b
Chapter Test B. Chapter: Chemical Equilibrium. following equilibrium system? 2CO(g) O 2 (g) ^ 2CO 2 (g)
 Assessment Chapter Test B Chapter: Chemical Equilibrium PART I In the space provided, write the letter of the term or phrase that best completes each statement or best answers each question. 1. What is
Assessment Chapter Test B Chapter: Chemical Equilibrium PART I In the space provided, write the letter of the term or phrase that best completes each statement or best answers each question. 1. What is
Depressing the hydrogenation and decomposition. nanoparticles on oxygen functionalized. carbon nanofibers. Supporting Information
 Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2015 Depressing the hydrogenation and decomposition reaction in H 2 O 2 synthesis
Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2015 Depressing the hydrogenation and decomposition reaction in H 2 O 2 synthesis
[Supplementary Information] One-Pot Synthesis and Electrocatalytic Activity of Octapodal Au-Pd Nanoparticles
![[Supplementary Information] One-Pot Synthesis and Electrocatalytic Activity of Octapodal Au-Pd Nanoparticles [Supplementary Information] One-Pot Synthesis and Electrocatalytic Activity of Octapodal Au-Pd Nanoparticles](/thumbs/78/77448401.jpg) [Supplementary Information] One-Pot Synthesis and Electrocatalytic Activity of Octapodal Au-Pd Nanoparticles Jong Wook Hong, Young Wook Lee, Minjung Kim, Shin Wook Kang, and Sang Woo Han * Department of
[Supplementary Information] One-Pot Synthesis and Electrocatalytic Activity of Octapodal Au-Pd Nanoparticles Jong Wook Hong, Young Wook Lee, Minjung Kim, Shin Wook Kang, and Sang Woo Han * Department of
Supporting information
 Supporting information imetallic AuRh nanodendrites consisting of Au icosahedron cores and atomically ultrathin Rh nanoplate shells: synthesis and light-enhanced catalytic activity Yongqiang Kang 1,2,
Supporting information imetallic AuRh nanodendrites consisting of Au icosahedron cores and atomically ultrathin Rh nanoplate shells: synthesis and light-enhanced catalytic activity Yongqiang Kang 1,2,
Sub-10-nm Au-Pt-Pd Alloy Trimetallic Nanoparticles with. High Oxidation-Resistant Property as Efficient and Durable
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information Sub-10-nm Au-Pt-Pd Alloy Trimetallic Nanoparticles with High
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information Sub-10-nm Au-Pt-Pd Alloy Trimetallic Nanoparticles with High
Supporting Information
 Supporting Information A Rational Solid-state Synthesis of Supported Au-Ni Bimetallic Nanoparticles with Enhanced Activity for Gas-phase Selective Oxidation of Alcohols Wuzhong Yi, a Wentao Yuan, b Ye
Supporting Information A Rational Solid-state Synthesis of Supported Au-Ni Bimetallic Nanoparticles with Enhanced Activity for Gas-phase Selective Oxidation of Alcohols Wuzhong Yi, a Wentao Yuan, b Ye
INSTRUCTIONS: Exam III. November 10, 1999 Lab Section
 CHEM 1215 Exam III John III. Gelder November 10, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
CHEM 1215 Exam III John III. Gelder November 10, 1999 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
TEM image of derivative 1 and fluorescence spectra of derivative 1 upon addition of
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2016 Supramolecular ensemble of PBI derivative and Cu 2 O NPs: Potential photo catalysts for
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2016 Supramolecular ensemble of PBI derivative and Cu 2 O NPs: Potential photo catalysts for
Ligand coated metal nanoparticles and quantum dots
 The Supramolecular Nano Materials Group Ligand coated metal nanoparticles and quantum dots Francesco Stellacci Department of Materials Science and Engineering frstella@mit.edu Outline Self-Assembled Monolayers
The Supramolecular Nano Materials Group Ligand coated metal nanoparticles and quantum dots Francesco Stellacci Department of Materials Science and Engineering frstella@mit.edu Outline Self-Assembled Monolayers
METHANOL OXIDATION OVER AU/ γ -AL 2 O 3 CATALYSTS
 Bajopas Volume 2 Number 2 December, 29 Bayero Journal of Pure and Applied Sciences, 2(2): 149-154 Received: May, 29 Accepted: July, 29 METHANOL OXIDATION OVER AU/ γ -AL 2 O 3 CATALYSTS Abdullahi Nuhu Kano
Bajopas Volume 2 Number 2 December, 29 Bayero Journal of Pure and Applied Sciences, 2(2): 149-154 Received: May, 29 Accepted: July, 29 METHANOL OXIDATION OVER AU/ γ -AL 2 O 3 CATALYSTS Abdullahi Nuhu Kano
Supplementary information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Supplementary information Real-time imaging and elemental mapping of AgAu nanoparticle transformations
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Supplementary information Real-time imaging and elemental mapping of AgAu nanoparticle transformations
CHAPTER 6. SOLVENT-FREE SELECTIVE OXIDATION OF -PINENE OVER Co-SBA-15 CATALYST
 135 CHAPTER 6 SOLVENT-FREE SELECTIVE OXIDATION OF -PINENE OVER Co-SBA-15 CATALYST 6.1 INTRODUCTION -Pinene is a terpenoid family of organic compound which is inexpensive, readily available and renewable
135 CHAPTER 6 SOLVENT-FREE SELECTIVE OXIDATION OF -PINENE OVER Co-SBA-15 CATALYST 6.1 INTRODUCTION -Pinene is a terpenoid family of organic compound which is inexpensive, readily available and renewable
M09/4/CHEMI/SPM/ENG/TZ1/XX+ CHEMISTRY. Monday 18 May 2009 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES
 M09/4/CHEMI/SPM/ENG/TZ1/XX+ 22096110 CHEMISTRY standard level Paper 1 Monday 18 May 2009 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES Do not open this examination paper until instructed to do so.
M09/4/CHEMI/SPM/ENG/TZ1/XX+ 22096110 CHEMISTRY standard level Paper 1 Monday 18 May 2009 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES Do not open this examination paper until instructed to do so.
Silesian University of Technology, Poland Recoverable and recyclable catalysts for sustainable chemical processes
 Silesian University of Technology, Poland Recoverable and recyclable catalysts for sustainable chemical processes Anna Chrobok What are the challenges for the sustainable chemical industry today? reduce
Silesian University of Technology, Poland Recoverable and recyclable catalysts for sustainable chemical processes Anna Chrobok What are the challenges for the sustainable chemical industry today? reduce
M10/4/CHEMI/SPM/ENG/TZ2/XX+ CHEMISTRY. Wednesday 12 May 2010 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES
 M10/4/CHEMI/SPM/ENG/TZ/XX+ 106116 CHEMISTRY standard level Paper 1 Wednesday 1 May 010 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES Do not open this examination paper until instructed to do so. Answer
M10/4/CHEMI/SPM/ENG/TZ/XX+ 106116 CHEMISTRY standard level Paper 1 Wednesday 1 May 010 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES Do not open this examination paper until instructed to do so. Answer
A nanoscale perspective on the effect of acid washing of carbon catalyst supports
 A nanoscale perspective on the effect of acid washing of carbon catalyst supports Emir Bouleghlimat, Philip R. Davies, Robert J. Davies, Jiri Kulahvy, David J. Morgan, School of Chemistry, Cardiff University
A nanoscale perspective on the effect of acid washing of carbon catalyst supports Emir Bouleghlimat, Philip R. Davies, Robert J. Davies, Jiri Kulahvy, David J. Morgan, School of Chemistry, Cardiff University
Chemistry Standard level Paper 1
 M15/4/CHEMI/SPM/ENG/TZ1/XX Chemistry Standard level Paper 1 Thursday 14 May 2015 (afternoon) 45 minutes Instructions to candidates Do not open this examination paper until instructed to do so. Answer all
M15/4/CHEMI/SPM/ENG/TZ1/XX Chemistry Standard level Paper 1 Thursday 14 May 2015 (afternoon) 45 minutes Instructions to candidates Do not open this examination paper until instructed to do so. Answer all
Ir-Re Alloy as a highly active catalyst for the hydrogenolysis
 Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2014 Supporting Information Ir-Re Alloy as a highly active catalyst for the hydrogenolysis
Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2014 Supporting Information Ir-Re Alloy as a highly active catalyst for the hydrogenolysis
Revision of Important Concepts. 1. Types of Bonding
 Revision of Important Concepts 1. Types of Bonding Electronegativity (EN) often molecular often ionic compounds Bonding in chemical substances Bond energy: Is the energy that is released when a bond is
Revision of Important Concepts 1. Types of Bonding Electronegativity (EN) often molecular often ionic compounds Bonding in chemical substances Bond energy: Is the energy that is released when a bond is
Chemistry Standard level Paper 1
 Chemistry Standard level Paper 1 Thursday 12 May 2016 (morning) 45 minutes Instructions to candidates Do not open this examination paper until instructed to do so. Answer all the questions. For each question,
Chemistry Standard level Paper 1 Thursday 12 May 2016 (morning) 45 minutes Instructions to candidates Do not open this examination paper until instructed to do so. Answer all the questions. For each question,
8. Relax and do well.
 CHEM 1215 Exam III John III. Gelder November 11, 1998 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
CHEM 1215 Exam III John III. Gelder November 11, 1998 Name TA's Name Lab Section INSTRUCTIONS: 1. This examination consists of a total of 7 different pages. The last page includes a periodic table and
**The partially (-) oxygen pulls apart and surrounds the (+) cation. The partially (+) hydrogen pulls apart and surrounds the (-) anion.
 #19 Notes Unit 3: Reactions in Solutions Ch. Reactions in Solutions I. Solvation -the act of dissolving (solute (salt) dissolves in the solvent (water)) Hydration: dissolving in water, the universal solvent.
#19 Notes Unit 3: Reactions in Solutions Ch. Reactions in Solutions I. Solvation -the act of dissolving (solute (salt) dissolves in the solvent (water)) Hydration: dissolving in water, the universal solvent.
Rh 3d. Co 2p. Binding Energy (ev) Binding Energy (ev) (b) (a)
 Co 2p Co(0) 778.3 Rh 3d Rh (0) 307.2 810 800 790 780 770 Binding Energy (ev) (a) 320 315 310 305 Binding Energy (ev) (b) Supplementary Figure 1 Photoemission features of a catalyst precursor which was
Co 2p Co(0) 778.3 Rh 3d Rh (0) 307.2 810 800 790 780 770 Binding Energy (ev) (a) 320 315 310 305 Binding Energy (ev) (b) Supplementary Figure 1 Photoemission features of a catalyst precursor which was
S 8 + F 2 SF 6 4/9/2014. iclicker Participation Question: Balance the following equation by inspection: H + + Cr 2 O 7 + C 2 H 5 OH
 Today: Redox Reactions Oxidations Reductions Oxidation Numbers Half Reactions Balancing in Acidic Solution Balancing in Basic Solution QUIZ 3 & EXAM 3 moved up by one day: Quiz 3 Wednesday/Thursday next
Today: Redox Reactions Oxidations Reductions Oxidation Numbers Half Reactions Balancing in Acidic Solution Balancing in Basic Solution QUIZ 3 & EXAM 3 moved up by one day: Quiz 3 Wednesday/Thursday next
Silica-Supported Cationic Gold(I) Complexes as Heterogeneous Catalysts for Regio- and Enantioselective Lactonization Reactions
 Silica-Supported Cationic Gold(I) Complexes as Heterogeneous Catalysts for Regio- and Enantioselective Lactonization Reactions Xing-Zhong Shu, Son C. Nguyen,Ying He, Fadekemi Oba, Qiao Zhang, Christian
Silica-Supported Cationic Gold(I) Complexes as Heterogeneous Catalysts for Regio- and Enantioselective Lactonization Reactions Xing-Zhong Shu, Son C. Nguyen,Ying He, Fadekemi Oba, Qiao Zhang, Christian
Chapter 4. The Major Classes of Chemical Reactions 4-1
 Chapter 4 The Major Classes of Chemical Reactions 4-1 The Major Classes of Chemical Reactions 4.1 The Role of Water as a Solvent 4.2 Writing Equations for Aqueous Ionic Reactions 4.3 Precipitation Reactions
Chapter 4 The Major Classes of Chemical Reactions 4-1 The Major Classes of Chemical Reactions 4.1 The Role of Water as a Solvent 4.2 Writing Equations for Aqueous Ionic Reactions 4.3 Precipitation Reactions
Mild and Efficient Oxidation of Primary and Secondary Alcohols Using NiO 2 /Silica Gel System (Solvent Free)
 ISSN: 0973-4945; CDEN ECJA E- Chemistry http://www.e-journals.net 2011, 8(2), 491-494 Mild and Efficient xidation of Primary and Secondary Alcohols Using Ni 2 /Silica Gel System (Solvent Free) MAMMAD KTI
ISSN: 0973-4945; CDEN ECJA E- Chemistry http://www.e-journals.net 2011, 8(2), 491-494 Mild and Efficient xidation of Primary and Secondary Alcohols Using Ni 2 /Silica Gel System (Solvent Free) MAMMAD KTI
Fuel Cells in Energy Technology. Tutorial 5 / SS solutions. Prof. W. Schindler, Jassen Brumbarov / Celine Rüdiger
 Fuel Cells in Energy Technology Tutorial 5 / SS 2013 - solutions Prof. W. Schindler, Jassen Brumbarov / Celine Rüdiger 05.06.2013 Homework 3: What hydrogen flow rate (g/hour) is required to generate 1
Fuel Cells in Energy Technology Tutorial 5 / SS 2013 - solutions Prof. W. Schindler, Jassen Brumbarov / Celine Rüdiger 05.06.2013 Homework 3: What hydrogen flow rate (g/hour) is required to generate 1
M11/4/CHEMI/SPM/ENG/TZ2/XX CHEMISTRY STANDARD LEVEL PAPER 1. Monday 9 May 2011 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES
 M11/4/CHEMI/SPM/ENG/TZ/XX 116116 CHEMISTRY STANDARD LEVEL PAPER 1 Monday 9 May 011 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES Do not open this examination paper until instructed to do so. Answer
M11/4/CHEMI/SPM/ENG/TZ/XX 116116 CHEMISTRY STANDARD LEVEL PAPER 1 Monday 9 May 011 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES Do not open this examination paper until instructed to do so. Answer
Unit-8 Equilibrium. Rate of reaction: Consider the following chemical reactions:
 Unit-8 Equilibrium Rate of reaction: Consider the following chemical reactions: 1. The solution of sodium chloride and silver nitrate when mixed, there is an instantaneous formation of a precipitate of
Unit-8 Equilibrium Rate of reaction: Consider the following chemical reactions: 1. The solution of sodium chloride and silver nitrate when mixed, there is an instantaneous formation of a precipitate of
NAME: SECOND EXAMINATION
 1 Chemistry 64 Winter 1994 NAME: SECOND EXAMINATION THIS EXAMINATION IS WORTH 100 POINTS AND CONTAINS 4 (FOUR) QUESTIONS THEY ARE NOT EQUALLY WEIGHTED! YOU SHOULD ATTEMPT ALL QUESTIONS AND ALLOCATE YOUR
1 Chemistry 64 Winter 1994 NAME: SECOND EXAMINATION THIS EXAMINATION IS WORTH 100 POINTS AND CONTAINS 4 (FOUR) QUESTIONS THEY ARE NOT EQUALLY WEIGHTED! YOU SHOULD ATTEMPT ALL QUESTIONS AND ALLOCATE YOUR
CHEMISTRY 1A SPRING 2011 EXAM 1 KEY CHAPTERS 1-4
 You might find the following useful. Electronegativities H 2.2 CHEMISTRY 1A SPRING 2011 EXAM 1 KEY CHAPTERS 1- Li Be B C N O F 0.98 1.57 2.0 2.55.0..98 Na Mg Al Si P S Cl 0.9 1.1 1.61 1.9 2.19 2.58.16
You might find the following useful. Electronegativities H 2.2 CHEMISTRY 1A SPRING 2011 EXAM 1 KEY CHAPTERS 1- Li Be B C N O F 0.98 1.57 2.0 2.55.0..98 Na Mg Al Si P S Cl 0.9 1.1 1.61 1.9 2.19 2.58.16
SCH4U: Practice Exam
 SCHU_07-08 SCHU: Practice Exam Energy in Chemistry 1. Which of the following correctly describes a reaction that absorbs heat from the surroundings? a. the reaction is endothermic b. H for this reaction
SCHU_07-08 SCHU: Practice Exam Energy in Chemistry 1. Which of the following correctly describes a reaction that absorbs heat from the surroundings? a. the reaction is endothermic b. H for this reaction
Chemistry 122 Wrap-Up Review Kundell
 Chapter 11 Chemistry 122 Wrap-Up Review Kundell 1. The enthalpy (heat) of vaporization for ethanol (C 2 H 5 OH) is 43.3 kj/mol. How much heat, in kilojoules, is required to vaporize 115 g of ethanol at
Chapter 11 Chemistry 122 Wrap-Up Review Kundell 1. The enthalpy (heat) of vaporization for ethanol (C 2 H 5 OH) is 43.3 kj/mol. How much heat, in kilojoules, is required to vaporize 115 g of ethanol at
Magnetic halloysite: an envirmental nanocatalyst for the synthesis of. benzoimidazole
 doi:10.3390/ecsoc-21-04726 Magnetic halloysite: an envirmental nanocatalyst for the synthesis of benzoimidazole Ali Maleki*, Zoleikha Hajizadeh Catalysts and Organic Synthesis Research Laboratory, Department
doi:10.3390/ecsoc-21-04726 Magnetic halloysite: an envirmental nanocatalyst for the synthesis of benzoimidazole Ali Maleki*, Zoleikha Hajizadeh Catalysts and Organic Synthesis Research Laboratory, Department
Direct Catalytic Conversion of Methane
 Direct Catalytic Conversion of Methane Xiulian Pan, Zengjian An, Ding Ma, Yide Xu, Xinhe Bao State Key Laboratory of Catlaysis, Dalian Institute of Chemical Physics, Chinese Academy of Sciences www.fruit.dicp.ac.cn
Direct Catalytic Conversion of Methane Xiulian Pan, Zengjian An, Ding Ma, Yide Xu, Xinhe Bao State Key Laboratory of Catlaysis, Dalian Institute of Chemical Physics, Chinese Academy of Sciences www.fruit.dicp.ac.cn
Recoverable rhodium nanoparticles: Synthesis, characterization and catalytic performance in hydrogenation reactions
 Available online at www.sciencedirect.com Applied Catalysis A: General 338 (2008) 52 57 www.elsevier.com/locate/apcata Recoverable rhodium nanoparticles: Synthesis, characterization and catalytic performance
Available online at www.sciencedirect.com Applied Catalysis A: General 338 (2008) 52 57 www.elsevier.com/locate/apcata Recoverable rhodium nanoparticles: Synthesis, characterization and catalytic performance
Supporting Information
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2015 Supporting Information Experimental details 1) Preparation of the catalytic materials 1,
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2015 Supporting Information Experimental details 1) Preparation of the catalytic materials 1,
Supplementary Figure 1 HAADF-STEM images of 0.08%Pt/FeO x -R200 single-atom catalyst with different magnifications. Scale bar: a, 20 nm; b, 10 nm; c,
 Supplementary Figure 1 HAADF-STEM images of 0.08%Pt/FeO x -R200 single-atom catalyst with different magnifications. Scale bar: a, 20 nm; b, 10 nm; c, 2 nm; d, 2 nm. The low magnification images demonstrate
Supplementary Figure 1 HAADF-STEM images of 0.08%Pt/FeO x -R200 single-atom catalyst with different magnifications. Scale bar: a, 20 nm; b, 10 nm; c, 2 nm; d, 2 nm. The low magnification images demonstrate
XXI. Part I Synthesis, Characterization, and Evaluation of Nanocatalysts in Ionic Liquids 1
 V List of Contributors XI Preface XV Foreword XIX Symbols and Abbreviations XXI Part I Synthesis, Characterization, and Evaluation of Nanocatalysts in Ionic Liquids 1 1 Fe, Ru, and Os Nanoparticles 3 Madhu
V List of Contributors XI Preface XV Foreword XIX Symbols and Abbreviations XXI Part I Synthesis, Characterization, and Evaluation of Nanocatalysts in Ionic Liquids 1 1 Fe, Ru, and Os Nanoparticles 3 Madhu
Shaping Single-crystalline Trimetallic Pt Pd Rh Nanocrystals toward. High-efficiency C C Splitting of Ethanol in Conversion to CO 2
 Supporting Information Shaping Single-crystalline Trimetallic Pt Pd Rh Nanocrystals toward High-efficiency C C Splitting of Ethanol in Conversion to CO 2 Wei Zhu,, Jun Ke,, Si-Bo Wang, Jie Ren, Hong-Hui
Supporting Information Shaping Single-crystalline Trimetallic Pt Pd Rh Nanocrystals toward High-efficiency C C Splitting of Ethanol in Conversion to CO 2 Wei Zhu,, Jun Ke,, Si-Bo Wang, Jie Ren, Hong-Hui
Monitoring the Surface Chemistry of Functionalized Nanomaterials with a Microfluidic Electronic Tongue
 Supporting Information Monitoring the Surface Chemistry of Functionalized Nanomaterials with a Microfluidic Electronic Tongue Flavio M. Shimizu, Anielli M. Pasqualeti, Fagner R. Todão, Jessica F. A. de
Supporting Information Monitoring the Surface Chemistry of Functionalized Nanomaterials with a Microfluidic Electronic Tongue Flavio M. Shimizu, Anielli M. Pasqualeti, Fagner R. Todão, Jessica F. A. de
TRU Chemistry Contest Chemistry 12 May 21, 2003 Time: 90 minutes
 TRU Chemistry Contest Chemistry 12 May 21, 2003 Time: 90 minutes Last Name First name School Teacher Please follow the instructions below. We will send your teacher a report on your performance. Top performers
TRU Chemistry Contest Chemistry 12 May 21, 2003 Time: 90 minutes Last Name First name School Teacher Please follow the instructions below. We will send your teacher a report on your performance. Top performers
Supporting Information
 Supporting Information for "Size-Controlled Pd Nanoparticle Catalysts Prepared by Galvanic Displacement into a Porous Si-Iron Oxide Nanoparticle Host." Taeho Kim, a Xin Fu, b David Warther, a and Michael
Supporting Information for "Size-Controlled Pd Nanoparticle Catalysts Prepared by Galvanic Displacement into a Porous Si-Iron Oxide Nanoparticle Host." Taeho Kim, a Xin Fu, b David Warther, a and Michael
Magnetically-driven selective synthesis of Au clusters on Fe 3 O 4 Nanoparticles
 Electronic Supplementary Material (ESI) for Chemical Communications Magnetically-driven selective synthesis of Au clusters on Fe 3 O 4 Nanoparticles Víctor Sebastian, M. Pilar Calatayud, Gerardo F. Goya
Electronic Supplementary Material (ESI) for Chemical Communications Magnetically-driven selective synthesis of Au clusters on Fe 3 O 4 Nanoparticles Víctor Sebastian, M. Pilar Calatayud, Gerardo F. Goya
Arborescent Polymers as Templates for the Preparation of Metallic Nanoparticles
 Arborescent Polymers as Templates for the Preparation of Metallic Nanoparticles Jason Dockendorff Department of Chemistry University of Waterloo Outline 1. 1. Focus and Purpose of of Research 2. 2. The
Arborescent Polymers as Templates for the Preparation of Metallic Nanoparticles Jason Dockendorff Department of Chemistry University of Waterloo Outline 1. 1. Focus and Purpose of of Research 2. 2. The
Special Properties of Au Nanoparticles
 Special Properties of Au Nanoparticles Maryam Ebrahimi Chem 7500/750 March 28 th, 2007 1 Outline Introduction The importance of unexpected electronic, geometric, and chemical properties of nanoparticles
Special Properties of Au Nanoparticles Maryam Ebrahimi Chem 7500/750 March 28 th, 2007 1 Outline Introduction The importance of unexpected electronic, geometric, and chemical properties of nanoparticles
Titration a solution of known concentration, called a standard solution
 Acid-Base Titrations Titration is a form of analysis in which we measure the volume of material of known concentration sufficient to react with the substance being analyzed. Titration a solution of known
Acid-Base Titrations Titration is a form of analysis in which we measure the volume of material of known concentration sufficient to react with the substance being analyzed. Titration a solution of known
Modified from: Larry Scheffler Lincoln High School IB Chemistry 1-2.1
 Modified from: Larry Scheffler Lincoln High School IB Chemistry 1-2.1 The development of the periodic table brought a system of order to what was otherwise an collection of thousands of pieces of information.
Modified from: Larry Scheffler Lincoln High School IB Chemistry 1-2.1 The development of the periodic table brought a system of order to what was otherwise an collection of thousands of pieces of information.
Incontro per l orientamento alla tesi sperimentale
 Corso di Laurea Magistrale in Chimica e Tecnologia del armaco Corso di Laurea Magistrale in armacia Incontro per l orientamento alla tesi sperimentale Prof.ssa Daniela Lanari 22 ovembre 2017, Aula A via
Corso di Laurea Magistrale in Chimica e Tecnologia del armaco Corso di Laurea Magistrale in armacia Incontro per l orientamento alla tesi sperimentale Prof.ssa Daniela Lanari 22 ovembre 2017, Aula A via
Problem Set III Stoichiometry - Solutions
 Chem 121 Problem set III Solutions - 1 Problem Set III Stoichiometry - Solutions 1. 2. 3. molecular mass of ethane = 2(12.011) + 6(1.008) = 30.07 g 4. molecular mass of aniline = 6(12.011) + 7(1.008) +
Chem 121 Problem set III Solutions - 1 Problem Set III Stoichiometry - Solutions 1. 2. 3. molecular mass of ethane = 2(12.011) + 6(1.008) = 30.07 g 4. molecular mass of aniline = 6(12.011) + 7(1.008) +
Sacrifical Template-Free Strategy
 Supporting Information Core/Shell to Yolk/Shell Nanostructures by a Novel Sacrifical Template-Free Strategy Jie Han, Rong Chen and Rong Guo* School of Chemistry and Chemical Engineering, Yangzhou University,
Supporting Information Core/Shell to Yolk/Shell Nanostructures by a Novel Sacrifical Template-Free Strategy Jie Han, Rong Chen and Rong Guo* School of Chemistry and Chemical Engineering, Yangzhou University,
Photon Energy Threshold in Direct Photocatalysis with Metal Nanoparticles Key Evidence from Action Spectrum of the Reaction
 Supporting Information Photon Energy Threshold in Direct Photocatalysis with Metal Nanoparticles Key Evidence from Action Spectrum of the Reaction Sarina Sarina, a Esa Jaatinen, a Qi Xiao, a,b Yi Ming
Supporting Information Photon Energy Threshold in Direct Photocatalysis with Metal Nanoparticles Key Evidence from Action Spectrum of the Reaction Sarina Sarina, a Esa Jaatinen, a Qi Xiao, a,b Yi Ming
Chemical Reactions Chapter 12 Study Guide (Unit 9)
 Name: Hr: Understand and be able to explain all of the key concepts. Define and understand all of the survival words Memorize the names and symbols for these elements: (Ag, Al, Ar, As, Au, B, Ba, Be, Br,
Name: Hr: Understand and be able to explain all of the key concepts. Define and understand all of the survival words Memorize the names and symbols for these elements: (Ag, Al, Ar, As, Au, B, Ba, Be, Br,
Introduction to X-ray Photoelectron Spectroscopy (XPS) XPS which makes use of the photoelectric effect, was developed in the mid-1960
 Introduction to X-ray Photoelectron Spectroscopy (XPS) X-ray Photoelectron Spectroscopy (XPS), also known as Electron Spectroscopy for Chemical Analysis (ESCA) is a widely used technique to investigate
Introduction to X-ray Photoelectron Spectroscopy (XPS) X-ray Photoelectron Spectroscopy (XPS), also known as Electron Spectroscopy for Chemical Analysis (ESCA) is a widely used technique to investigate
Pharmaceutical Analytical Chemistry PHCM223 Lecture 9 REDOX REACTIONS (I) Dr. Nesrine El Gohary 9 th lecture
 Pharmaceutical Analytical Chemistry PHCM223 Lecture 9 REDOX REACTIONS (I) Dr. Nesrine El Gohary 9 th lecture Learning outcomes Define redox reactions. Apply redox titrations. Detect the end point of redox
Pharmaceutical Analytical Chemistry PHCM223 Lecture 9 REDOX REACTIONS (I) Dr. Nesrine El Gohary 9 th lecture Learning outcomes Define redox reactions. Apply redox titrations. Detect the end point of redox
Very low temperature CO oxidation over colloidally deposited gold nanoparticles on Mg(OH) 2 and MgO
 Supporing Information Very low temperature CO oxidation over colloidally deposited gold nanoparticles on Mg(OH) 2 and MgO Chun-Jiang Jia, Yong Liu, Hans Bongard, Ferdi Schüth* Max-Planck-Institut für Kohlenforschung,
Supporing Information Very low temperature CO oxidation over colloidally deposited gold nanoparticles on Mg(OH) 2 and MgO Chun-Jiang Jia, Yong Liu, Hans Bongard, Ferdi Schüth* Max-Planck-Institut für Kohlenforschung,
Compounds in Aqueous Solution
 1 Compounds in Aqueous Solution Many reactions involve ionic compounds, especially reactions in water KMnO 4 in water K + (aq) ) + MnO 4- (aq) 2 CCR, page 149 3 How do we know ions are present in aqueous
1 Compounds in Aqueous Solution Many reactions involve ionic compounds, especially reactions in water KMnO 4 in water K + (aq) ) + MnO 4- (aq) 2 CCR, page 149 3 How do we know ions are present in aqueous
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/NCHEM.1066 Facile removal of stabiliser-ligands from supported gold nanoparticles Jose A. Lopez-Sanchez 1, Nikolaos Dimitratos 1, Ceri Hammond 1, Gemma L. Brett 1, Lokesh Kesavan 1, Saul White
DOI: 10.1038/NCHEM.1066 Facile removal of stabiliser-ligands from supported gold nanoparticles Jose A. Lopez-Sanchez 1, Nikolaos Dimitratos 1, Ceri Hammond 1, Gemma L. Brett 1, Lokesh Kesavan 1, Saul White
Design of a new family of catalytic support based on thiol containing plasma polymer films
 Design of a new family of catalytic support based on thiol containing plasma polymer films Dr. D. Thiry damien.thiry@umons.ac.be Chimie des Interactions Plasma Surface (ChIPS), CIRMAP, University of Mons,
Design of a new family of catalytic support based on thiol containing plasma polymer films Dr. D. Thiry damien.thiry@umons.ac.be Chimie des Interactions Plasma Surface (ChIPS), CIRMAP, University of Mons,
Stoichiometry Practice Problems
 Name Period CRHS Academic Chemistry Stoichiometry Practice Problems Due Date Assignment On-Time (100) Late (70) 9.1 9.2 9.3 9.4 9.5 Warm-Up EC Notes, Homework, Exam Reviews and Their KEYS located on CRHS
Name Period CRHS Academic Chemistry Stoichiometry Practice Problems Due Date Assignment On-Time (100) Late (70) 9.1 9.2 9.3 9.4 9.5 Warm-Up EC Notes, Homework, Exam Reviews and Their KEYS located on CRHS
CHEMICAL OXIDATION. The use of oxidizing agents without the need of microorganisms for the reactions to proceed
 CHEMICAL OXIDATION The use of oxidizing agents without the need of microorganisms for the reactions to proceed oxidizing agents : O 3, H 2 O 2, Cl 2 or HOCl or O 2 etc catalysts : ph, transition metals,
CHEMICAL OXIDATION The use of oxidizing agents without the need of microorganisms for the reactions to proceed oxidizing agents : O 3, H 2 O 2, Cl 2 or HOCl or O 2 etc catalysts : ph, transition metals,
Basic Concepts of Chemistry and Chemical Calculations. The ratio of the average mass factor to one twelfth of the mass of an atom of carbon-12
 Basic Concepts of Chemistry and Chemical Calculations Relative Atomic mass: The relative atomic mass is defined as the ratio of the average atomic mass factor to the unified atomic mass unit. (Or) The
Basic Concepts of Chemistry and Chemical Calculations Relative Atomic mass: The relative atomic mass is defined as the ratio of the average atomic mass factor to the unified atomic mass unit. (Or) The
Chem 1A Dr. White Fall Handout 4
 Chem 1A Dr. White Fall 2014 1 Handout 4 4.4 Types of Chemical Reactions (Overview) A. Non-Redox Rxns B. Oxidation-Reduction (Redox) reactions 4.6. Describing Chemical Reactions in Solution A. Molecular
Chem 1A Dr. White Fall 2014 1 Handout 4 4.4 Types of Chemical Reactions (Overview) A. Non-Redox Rxns B. Oxidation-Reduction (Redox) reactions 4.6. Describing Chemical Reactions in Solution A. Molecular
Name AP Chemistry September 30, 2013
 Name AP Chemistry September 30, 2013 AP Chemistry Exam Part I: 40 Questions, 40 minutes, Multiple Choice, No Calculator Allowed Bubble the correct answer on the blue side of your scantron for each of the
Name AP Chemistry September 30, 2013 AP Chemistry Exam Part I: 40 Questions, 40 minutes, Multiple Choice, No Calculator Allowed Bubble the correct answer on the blue side of your scantron for each of the
IB Chemistry. Topic 3: Periodicity. Name
 IB Chemistry Topic 3: Periodicity Name Topic 3 and 13 Periodicity Alkali metals: Group 1 elements. Strength of metallic bond gets weaker as atoms get larger. Anion: A charged ion. Decrease in size across
IB Chemistry Topic 3: Periodicity Name Topic 3 and 13 Periodicity Alkali metals: Group 1 elements. Strength of metallic bond gets weaker as atoms get larger. Anion: A charged ion. Decrease in size across
Section 3: Etching. Jaeger Chapter 2 Reader
 Section 3: Etching Jaeger Chapter 2 Reader Etch rate Etch Process - Figures of Merit Etch rate uniformity Selectivity Anisotropy d m Bias and anisotropy etching mask h f substrate d f d m substrate d f
Section 3: Etching Jaeger Chapter 2 Reader Etch rate Etch Process - Figures of Merit Etch rate uniformity Selectivity Anisotropy d m Bias and anisotropy etching mask h f substrate d f d m substrate d f
Unit 4a: Solution Stoichiometry Last revised: October 19, 2011 If you are not part of the solution you are the precipitate.
 1 Unit 4a: Solution Stoichiometry Last revised: October 19, 2011 If you are not part of the solution you are the precipitate. You should be able to: Vocabulary of water solubility Differentiate between
1 Unit 4a: Solution Stoichiometry Last revised: October 19, 2011 If you are not part of the solution you are the precipitate. You should be able to: Vocabulary of water solubility Differentiate between
Supporting Information
 Supporting Information Single Enzyme Direct Biomineralization of CdSe and CdSe-CdS Core-Shell Quantum Dots Zhou Yang 1, Li Lu 2, Christopher J. Kiely 1,2, Bryan W. Berger* 1,3, and Steven McIntosh* 1 1
Supporting Information Single Enzyme Direct Biomineralization of CdSe and CdSe-CdS Core-Shell Quantum Dots Zhou Yang 1, Li Lu 2, Christopher J. Kiely 1,2, Bryan W. Berger* 1,3, and Steven McIntosh* 1 1
Electrostatic interactions to modulate the reflective assembly of nanoparticles at the oilwater
 Supplemental Information Electrostatic interactions to modulate the reflective assembly of nanoparticles at the oilwater interface Mingxiang Luo, Gloria K. Olivier, and Joelle Frechette* Department of
Supplemental Information Electrostatic interactions to modulate the reflective assembly of nanoparticles at the oilwater interface Mingxiang Luo, Gloria K. Olivier, and Joelle Frechette* Department of
A green and efficient oxidation of alcohols by supported gold. conditions
 A green and efficient oxidation of alcohols by supported gold catalysts using aqueous H 2 O 2 under organic solvent-free conditions Ji Ni, Wen-Jian Yu, Lin He, Hao sun, Yong Cao,* He-Yong He, and Kang-Nian
A green and efficient oxidation of alcohols by supported gold catalysts using aqueous H 2 O 2 under organic solvent-free conditions Ji Ni, Wen-Jian Yu, Lin He, Hao sun, Yong Cao,* He-Yong He, and Kang-Nian
( ) SENIOR 4 CHEMISTRY FINAL PRACTICE REVIEW TEST VALUE: TOTAL 100 MARKS. Multiple Choice. Ca (PO ) 3Ca + 2PO. Name Student Number
 SENIOR 4 CHEMISTRY FINAL PRACTICE REVIEW TEST Name Student Number Attending Phone Number Address NonAttending ANSWER EY VALUE: TOTAL 100 MARS PART A Multiple Choice 1. (c) Using the solubility chart i)
SENIOR 4 CHEMISTRY FINAL PRACTICE REVIEW TEST Name Student Number Attending Phone Number Address NonAttending ANSWER EY VALUE: TOTAL 100 MARS PART A Multiple Choice 1. (c) Using the solubility chart i)
klm Mark Scheme Chemistry 6421 General Certificate of Education Thermodynamics and Further Inorganic Chemistry 2010 examination - January series
 Version.: 0502 klm General Certificate of Education Chemistry 642 CHM5 Thermodynamics and Further Inorganic Chemistry Mark Scheme 200 examination - January series Mark schemes are prepared by the Principal
Version.: 0502 klm General Certificate of Education Chemistry 642 CHM5 Thermodynamics and Further Inorganic Chemistry Mark Scheme 200 examination - January series Mark schemes are prepared by the Principal
International Journal of Scientific & Engineering Research, Volume 5, Issue 3, March-2014 ISSN
 156 Copper Nanoparticles: Green Synthesis Characterization Y.Suresh*1, S.Annapurna*2, G.Bhikshamaiah*3, A.K.Singh#4 Abstract Present work describes the synthesis nanoparticles using papaya extract as a
156 Copper Nanoparticles: Green Synthesis Characterization Y.Suresh*1, S.Annapurna*2, G.Bhikshamaiah*3, A.K.Singh#4 Abstract Present work describes the synthesis nanoparticles using papaya extract as a
Towards the pressure and material gap in heterogeneous catalysis: hydrogenation of acrolein over silver catalysts
 Towards the pressure and material gap in heterogeneous catalysis: hydrogenation of acrolein over silver catalysts M. Bron 1, M. Bonifer 1, A. Knop-Gericke 2, D. Teschner 2, J. Kröhnert 2, F.C. Jentoft
Towards the pressure and material gap in heterogeneous catalysis: hydrogenation of acrolein over silver catalysts M. Bron 1, M. Bonifer 1, A. Knop-Gericke 2, D. Teschner 2, J. Kröhnert 2, F.C. Jentoft
Cadiz-Paris Train Line; Catalysts, Nanostructures and EELS Spectroscopy. Susana Trasobares
 Cadiz-Paris Train Line; Catalysts, Nanostructures and EELS Spectroscopy Susana Trasobares 2010 Paris Odile Stéphan Mathieu Kociak Christian Colliex 1997 Cadiz Miguel López-Haro Juan C. Hernandez Jose A.
Cadiz-Paris Train Line; Catalysts, Nanostructures and EELS Spectroscopy Susana Trasobares 2010 Paris Odile Stéphan Mathieu Kociak Christian Colliex 1997 Cadiz Miguel López-Haro Juan C. Hernandez Jose A.
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/321/5894/1331/dc1 Supporting Online Material for Identification of Active Gold Nanoclusters on Iron Oxide Supports for CO Oxidation Andrew A. Herzing, Christopher J.
www.sciencemag.org/cgi/content/full/321/5894/1331/dc1 Supporting Online Material for Identification of Active Gold Nanoclusters on Iron Oxide Supports for CO Oxidation Andrew A. Herzing, Christopher J.
Selective Catalytic Dimethyl Disulphide Conversion into Dimethyl Sulphide
 Chemistry for Sustainable Development 21 (2013) 471 478 471 UDC 547.279.3:547.279.1:542.97:547.261 Selective Catalytic Dimethyl Disulphide Conversion into Dimethyl Sulphide A. V. MASHKINA and L. N. KHAIRULINA
Chemistry for Sustainable Development 21 (2013) 471 478 471 UDC 547.279.3:547.279.1:542.97:547.261 Selective Catalytic Dimethyl Disulphide Conversion into Dimethyl Sulphide A. V. MASHKINA and L. N. KHAIRULINA
J. Am. Chem. Soc. 2009, 131,
 Palladium Nanoparticles on Graphite Oxide and Its Functionalized Graphene Derivatives as Highly Active Catalysts for the Suzuki-Miyaura Coupling Reaction J. Am. Chem. Soc. 2009, 131, 8262 8270 Rolf Műlhaupt
Palladium Nanoparticles on Graphite Oxide and Its Functionalized Graphene Derivatives as Highly Active Catalysts for the Suzuki-Miyaura Coupling Reaction J. Am. Chem. Soc. 2009, 131, 8262 8270 Rolf Műlhaupt
Circle the letters only. NO ANSWERS in the Columns! (3 points each)
 Chemistry 1304.001 Name (please print) Exam 4 (100 points) April 12, 2017 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Circle the letters only. NO ANSWERS in
Chemistry 1304.001 Name (please print) Exam 4 (100 points) April 12, 2017 On my honor, I have neither given nor received unauthorized aid on this exam. Signed Date Circle the letters only. NO ANSWERS in
Permeable Silica Shell through Surface-Protected Etching
 Permeable Silica Shell through Surface-Protected Etching Qiao Zhang, Tierui Zhang, Jianping Ge, Yadong Yin* University of California, Department of Chemistry, Riverside, California 92521 Experimental Chemicals:
Permeable Silica Shell through Surface-Protected Etching Qiao Zhang, Tierui Zhang, Jianping Ge, Yadong Yin* University of California, Department of Chemistry, Riverside, California 92521 Experimental Chemicals:
Green nanoscience: Opportunities and challenges for innovation
 Green nanoscience: Opportunities and challenges for innovation Jim Hutchison Department of Chemistry, University of Oregon Director, UO Materials Science Institute Director, ONAMI Safer Nanomaterials and
Green nanoscience: Opportunities and challenges for innovation Jim Hutchison Department of Chemistry, University of Oregon Director, UO Materials Science Institute Director, ONAMI Safer Nanomaterials and
