Unit 1: Analyzing Data 1. Measure the following using the appropriate number of significant digits. Name Hour Date. b. o C
|
|
|
- Tobias Baldwin
- 5 years ago
- Views:
Transcription
1 Name Hour Date Chemistry Semester 1 Review!!! If you have completed ALL of the review on the assigned days you are allowed to use a cheat sheet that is created on the back of the periodic table found on the last page of this review. Unit 1: Analyzing Data 1. Measure the following using the appropriate number of significant digits a. cm b. o C Measurement in cm for metric ruler on the left. Measurement in ml of graduate cylinder. Measurement in ⁰C. Measurement in ml of graduate cylinder. Measurement in inches. ⁰C Page 1 of 15
2 Draw a cube. Indicate the length of one side in centimeters (cm)? What is the volume of this cube represented by symbols? What is the volume of this cube? What volume (in ml) of water could this box hold if it was filled with water? What is the volume of this cube? What volume (in ml) of water could this box hold if it was filled with water? What is the volume of this cube? What volume (in ml) of water could this box hold if it was filled with water? 2. Does the objects below have the same mass or volume? Explain your answer. In your explanation make sure to include how mass and volume differ. 3. A. Explain the law of conservation of mass. B. Provide an example for the labs that we did to support your explanation. C. Sulfur and Iron are mixed together and heated until they form a compound. Using the picture below determine the mass of the product that was formed. g Page 2 of 15
3 4. What is density? Describe how substances with different densities behave using the pictures below. Use the graph below the answer the following 3 questions. 5. What does the slope of the graph mean? Ma ss (g) Substance A Calculate the slope for substance A. Substance B 6. Which object has a larger density? How do you know? 7. The density of water is 1g/cm 3. Draw the line for water on the graph above. 8. What is the mass of the ethyl alcohol that exactly fills a ml container? The density of ethyl alcohol is g/ml. 9. Calculate the density of sulfuric acid if 35.4 ml of the acid weighs g. 10. A rectangular block of copper metal weighs 1896 g. The dimensions of the block are 8.4 cm by 5.5 cm by 4.6 cm. From this data, what is the density of copper? 11. The diagram below depicts there different gases. Rank the gasses from lowest to highest density. Explain your ranking. Page 3 of 15
4 Unit 2: Gases 12. As the volume of a gas decreases, the pressure (increases/decreases). Particle diagram: Sketch a graph to illustrate this relationship 13. As the temperature of a gas increases, the volume (increases/decreases). Particle diagram: Sketch a graph to illustrate this relationship. 14. As the temperature of a gas decreases, the pressure (increases/decreases). Particle diagram: Sketch a graph to illustrate this relationship. 15. What is the conversion from Celsius to Kelvin: 16. A gas in a 2.0 L container has a pressure of 4.0 atm. When injected into a 1.5 L container, what is the pressure? Initial Final Change Effect P T V n 17. A gas in a closed container is pressurized from 7.00 atm to 10.0 atm. If the original temperature is 10.0⁰C, what is the final temperature of the gas? Initial Final Change Effect P T V n Page 4 of 15
5 18. If 5.0 L of CO2 is heated from K to 150 K, what is the new volume? Initial Final Change Effect P T V n 19. What would be the new volume if 150 cm 3 of gas at 25 C and 0.92 atm pressure were changed to standard conditions of temperature (0 C ) and pressure (1 atm)? Initial Final Change Effect P T V n Unit 3: Energy and States of matter 20. Ice cream is left out and melts. 21. In the previous problem did the system gain or lose energy? Explain your answer. 22. When water is boiling what are in the bubbles? Useful Constants 1 calorie =4.18 joules 1000 joules = I kilojoule 1000 calories = 1 kilocalorie = 1 nutritional calorie 23. A candy bar contains 250 nutritional calories determine how many calories it contains. 24. To boil water it requires 351 kilojoules of energy determine how many calories are required. Page 5 of 15
6 25. How many joules of energy would be required to change the temperature of g of water from 25 o C to 75 o C? 26. Exactly 850 J will raise the temperature of 30.0g of a metal from 25.0⁰C to 60.0⁰C. What is the specific heat capacity of the metal? g of ice at 0.0 C sits on the counter and begins to melt to liquid water. How much energy must be absorbed by the ice if it all melted? 28. While sweating water vapor evaporates from your body, absorbing 125,000 J of energy. (assume no temperature change). What mass of water evaporates? 29. On the graph below label the following items for each region. a. Label the state(s) of matter and describe what is happening during each region. b. Identify how the energy is being stored (Eth or Eph). A B C c. Draw a particle diagram for each region in the boxes below. 29. A 25.0 g sample of water at 20.0⁰C changes to steam at 140⁰C. a. Draw the heating curve for this sample. b. Calculate the total amount of energy this process requires. Page 6 of 15
7 Unit 4: Describing Substances 30. Give an example of a mixture that could be separated using the apparatus shown to the right. 31. Explain how the apparatus below works to separate solutions? 32. Classify the following as Chemical or Physical changes. Verbal Description of Change C or P? a. Melting Iron b. Burning paper c. Metal rusting d. Aluminum foil is cut into smaller pieces. e. Sand is mixed with pebbles and water. 33. Describe and draw a particle drawing of the different categories of matter. ELEMENT: COMPOUND: MIXTURE: 34. Explain how a mixture is different from a compound. Provide an example of each. 35. Consider the four containers below a. Which of these are mixtures? pure substances? b. Which contain only compounds? only elements Page 7 of 15
8 Unit 5: Atomic Structure and Electrons in the Atom 36. Determine whether the statements describe Protons (p + ), Electrons (e - ), Neutrons (n 0 ), or Nucleus (N). There may be more than one answer. Smallest mass Found in the nucleus Positively charged Negatively charged 37. Fill in the chart below Neutral Most dense part of the atom Mass of 1 amu Identifying characteristic of an atom Sc Isotopic Symbol Atomic # Mass # # of protons # of Neutrons # of Electrons What is an isotope? 39. What is the average atomic mass, in amu, for element X? Isotope Mass of the Isotope Percent Abundance X % Identify the element X % Is the average atomic mass from the periodic table closer to or ? EXPLAIN why. 40. Pictured below is the mass spectrogram of a transition metal. a. Based on the experimentally obtained values of mass numbers and percent abundance, calculate the average atomic mass of the element. Show your work below. b. Which isotope is the most abundant? c. Which isotope will deviate the most from the straight line? Page 8 of 15
9 41. Sketch the visible light spectrum below. Include the energy trend. Energy Describe this diagram and indicate which color of visible light has the highest energy? Describe this diagram and indicate which color of visible light has the lowest energy? What energy drop is required to produce a red photon of light? Discuss energy levels. What energy drop is required to produce a violet photon of light? Discuss energy levels. What color of visible light has the lowest energy? What color of visible light has the highest energy? 42. Explain the following rules for electron configuration. a. Aufbau Principle: Page 9 of 15
10 b. Hund s Rule: c. Pauli Exclusion Principle: 43. Rank the following orbitals from lowest to highest energy. 4s, 2p, 3s, 3d, 4p 44. Please study the water flooding buildings analogy below. What sublevel fills after the 3s? The 3s sublevel contains what two elements. What sublevel fills after the 3p? What sublevel fills after the 4s? The 4s sublevel contains what two elements. What sublevel fills after the 5s? This is not shown on the diagram. 45. Write electron configurations and orbital diagrams for the following elements: Be Ni 46. Write the noble gas configuration for the following elements: a. P b. Br Page 10 of 15
11 47. Write the electron configuration for the following elements: a. Kr b. F Unit 6: Periodic Table 48. List common properties of metals and nonmetals. Metals Nonmetals 49. Explain the characteristic that determines the chemical properties of an element. Which characteristic is the most important to determining the chemical properties? 50. Label and the following: alkali metals, alkaline earth metals, halogens, noble gases metals, nonmetals, metalloids # of valence electrons for groups 1-8A s, p, d, and f blocks 51. Identify the trends in atomic size on the periodic table and why each trend exists. Label them on the periodic table above. Page 11 of 15
12 Period Trend: Group Trend: 52. Describe the ionization energy of an atom. 53. Explain why the ionization energy would increase based on the number of protons in the nucleus. The diagram for sodium (Na). The diagram for magnesium (Mg). Write the number of protons in the blank above. Write the electron configuration below. Label the core electrons and valence electrons on the diagram above. Write the number of protons in the blank above. Write the electron configuration below. Label the core electrons and valence electrons on the diagram above. Discuss the force of attraction between the protons in the nucleus and valence (outer electrons). Discuss the force of attraction between the protons in the nucleus and valence (outer electrons). Page 12 of 15
13 54. Identify the trends in ionization energy on the periodic table and why each trend exists. Label them on the periodic table above. Period Trend: Group Trend: 55. Order the following from smallest to largest ionization energy: Ar, He, Rn, Xe 56. Order the following from smallest to largest atomic radius: Ca, Sc, Fe, Br Page 13 of 15
14 57. Explain why the following elements would decrease as you move from left to right on the periodic table. The diagram for sulfur (S). The diagram for chlorine (Cl). Write the number of protons in the blank above. Write the electron configuration below. Write the number of protons in the blank above. Write the electron configuration below. Label the core electrons and valence electrons on the diagram above. Label the core electrons and valence electrons on the diagram above. Discuss the force of attraction between the protons in the nucleus and valence (outer electrons). Discuss the force of attraction between the protons in the nucleus and valence (outer electrons). 58. Draw the electron (Lewis) dot structure for the following elements. a. Sulfur b. Neon 59. What do the dots represent in the above drawing? Page 14 of 15
15 Page 15 of 15
Name Hour Date. B. Provide an example for the labs that we did to support your explanation.
 Name Hour Date Chemistry Semester 1 Review!!! If you have completed ALL of the review on the assigned days you are allowed to use a cheat sheet that is created on the back and of the periodic table found
Name Hour Date Chemistry Semester 1 Review!!! If you have completed ALL of the review on the assigned days you are allowed to use a cheat sheet that is created on the back and of the periodic table found
Ch. 1: Introduction to Chemistry. Ch. 2: Matter and Change
 Review Sheet for Chemistry First Semester Final Refer to your class notes, worksheets, and the textbook to complete this review sheet. Study early so that you will have time to ask questions about what
Review Sheet for Chemistry First Semester Final Refer to your class notes, worksheets, and the textbook to complete this review sheet. Study early so that you will have time to ask questions about what
CP Chemistry Semester 1 Final Test Review 1. Know the symbol and the power of 10 for the following metric prefixes: A. Mega B.
 Mass in grams 10 20 30 40 50 CP Chemistry Semester 1 Final Test Review 1. Know the symbol and the power of 10 for the following metric prefixes: A. Mega B. kilo D. deka E. deci G. milli H. micro C. hecto
Mass in grams 10 20 30 40 50 CP Chemistry Semester 1 Final Test Review 1. Know the symbol and the power of 10 for the following metric prefixes: A. Mega B. kilo D. deka E. deci G. milli H. micro C. hecto
Chemistry Final Exam Study Guide Fall Semester
 Chemistry Final Exam Study Guide Fall Semester Name: Date: Class: Basics of Science (Ch 1) 1. Briefly describe what occurs in each step of the scientific method: 1. Problem/Question 2. Research 3. Hypothesis
Chemistry Final Exam Study Guide Fall Semester Name: Date: Class: Basics of Science (Ch 1) 1. Briefly describe what occurs in each step of the scientific method: 1. Problem/Question 2. Research 3. Hypothesis
Questions 1 to 58 must be answered on the Scantron sheets.
 Questions 1 to 58 must be answered on the Scantron sheets. Base your answers to questions 1 to 5 on the heating curve for a pure substance that is shown below. 1. The freezing point of the substance is
Questions 1 to 58 must be answered on the Scantron sheets. Base your answers to questions 1 to 5 on the heating curve for a pure substance that is shown below. 1. The freezing point of the substance is
new experimental data, and can be modified
 Mass in grams 10 20 30 40 50 Name: Date: Period: CP Chemistry Semester 1 Final Test Review CHAPTERS 1 & 2: Scientific Method, Density, Metric Conversions, Accuracy/Precision, Significant Figures 1. Know
Mass in grams 10 20 30 40 50 Name: Date: Period: CP Chemistry Semester 1 Final Test Review CHAPTERS 1 & 2: Scientific Method, Density, Metric Conversions, Accuracy/Precision, Significant Figures 1. Know
Name: Date: Chemistry ~ Ms. Hart Class: Anions or Cations. Station Review Midterm January 2014 STATION 1: Chemical/physical properties and change
 Name: Date: STATION 1: Chemical/physical properties and change Physical changes are changes in matter in which the appearance of a substance changes but the identity of the compound remains the same Chemical
Name: Date: STATION 1: Chemical/physical properties and change Physical changes are changes in matter in which the appearance of a substance changes but the identity of the compound remains the same Chemical
Unit 2 Review Please note that this does not start on question 1.
 Unit 2 Review Please note that this does not start on question 1. 21. Of the three particles; protons, neutrons, and electrons, which one(s) are responsible for most of the mass of an atom? a) the protons
Unit 2 Review Please note that this does not start on question 1. 21. Of the three particles; protons, neutrons, and electrons, which one(s) are responsible for most of the mass of an atom? a) the protons
CP Chemistry Semester 1 Final Test Review
 Mass in grams 10 20 30 40 50 CP Chemistry Semester 1 Final Test Review 1. Know the symbol and the power of 10 for the following metric prefixes: A. Mega M 10 6 D. deka da 10 1 G. milli m 10 6 B. kilo k
Mass in grams 10 20 30 40 50 CP Chemistry Semester 1 Final Test Review 1. Know the symbol and the power of 10 for the following metric prefixes: A. Mega M 10 6 D. deka da 10 1 G. milli m 10 6 B. kilo k
AE Chemistry Midterm Study Guide
 Name Date Define Chemistry AE Chemistry Midterm Study Guide Since chemistry studies matter what is the definition of matter. What is the Law of Conservation of Matter? What is energy, what are the two
Name Date Define Chemistry AE Chemistry Midterm Study Guide Since chemistry studies matter what is the definition of matter. What is the Law of Conservation of Matter? What is energy, what are the two
CP Chemistry Semester 1 Final Review KEY. Unit 1
 CP Chemistry Semester 1 Final Review KEY Unit 1 Practice Problems 1. What tool do you use to measure volume of water? Describe how to make a proper measurement of a volume of water using a 50 ml graduated
CP Chemistry Semester 1 Final Review KEY Unit 1 Practice Problems 1. What tool do you use to measure volume of water? Describe how to make a proper measurement of a volume of water using a 50 ml graduated
Chapter 4 Atoms Practice Problems
 Chapter 4 Atoms Practice Problems 1) The primary substances of which all other things are composed are A) molecules. B) compounds. C) elements. D) electrons. E) protons. 2) Which of the following is a
Chapter 4 Atoms Practice Problems 1) The primary substances of which all other things are composed are A) molecules. B) compounds. C) elements. D) electrons. E) protons. 2) Which of the following is a
CHM 100 CHEMISTRY MAN & ENVIRONMENT Atoms and Elements Sample Test
 CHM 100 CHEMISTRY MAN & ENVIRONMENT Atoms and Elements Sample Test Multiple Choice: Identify the choice that best completes the statement or answers the question. 1. Which of these elements has two valence
CHM 100 CHEMISTRY MAN & ENVIRONMENT Atoms and Elements Sample Test Multiple Choice: Identify the choice that best completes the statement or answers the question. 1. Which of these elements has two valence
Honors Chemistry: Chapter 4- Problem Set (with some 6)
 Honors Chemistry: Chapter 4- Problem Set (with some 6) All answers and work on a separate sheet of paper! Classify the following as always true (AT), sometimes true (ST), or never true (NT) 1. Atoms of
Honors Chemistry: Chapter 4- Problem Set (with some 6) All answers and work on a separate sheet of paper! Classify the following as always true (AT), sometimes true (ST), or never true (NT) 1. Atoms of
What are the isotopes of hydrogen? H, 2 H, and 3 H
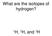 What are the isotopes of hydrogen? 1 H, 2 H, and 3 H The average mass of a boron atom is 10.81. Assuming you were able to isolate only one boron atom, the chance that you would randomly get one with a
What are the isotopes of hydrogen? 1 H, 2 H, and 3 H The average mass of a boron atom is 10.81. Assuming you were able to isolate only one boron atom, the chance that you would randomly get one with a
Part A. Answer all questions in this part.
 Part A Directions (1-20): For each statement or question, record on your separate answer sheet the number of the word or expression that, of those given, best completes the statement or answers the question.
Part A Directions (1-20): For each statement or question, record on your separate answer sheet the number of the word or expression that, of those given, best completes the statement or answers the question.
Periodic Table Practice 11/29
 Periodic Table Practice 11/29 1. The arrangement of the elements from left to right in Period 4 on the Periodic Table is based on A) atomic mass B) atomic number C) the number of electron shells D) the
Periodic Table Practice 11/29 1. The arrangement of the elements from left to right in Period 4 on the Periodic Table is based on A) atomic mass B) atomic number C) the number of electron shells D) the
There is more here than would be on the test, but this is a good example of the types of questions you will encounter.
 Test Study Materials There is more here than would be on the test, but this is a good example of the types of questions you will encounter. Chapter 1 38. Define matter. Explain what is meant by mass and
Test Study Materials There is more here than would be on the test, but this is a good example of the types of questions you will encounter. Chapter 1 38. Define matter. Explain what is meant by mass and
Test Topics: Periodic Table, Atomic Theory, Physical/Chemical Properties, Atom, Isotopes, Average Atomic Mass
 Elemental Properties Review Worksheet Test Topics: Periodic Table, Atomic Theory, Physical/Chemical Properties, Atom, Isotopes, Average Atomic Mass Periodic Table 1. List the element symbols for the following
Elemental Properties Review Worksheet Test Topics: Periodic Table, Atomic Theory, Physical/Chemical Properties, Atom, Isotopes, Average Atomic Mass Periodic Table 1. List the element symbols for the following
Chemistry Mid-Term Exam Review Spring 2017
 Unit 1 Measurement & Math Accuracy & Precision (recognizing given lab data) Density calculations Number of SFs in a measurement, Round answers to correct number of SFs Percent Error Unit conversions in
Unit 1 Measurement & Math Accuracy & Precision (recognizing given lab data) Density calculations Number of SFs in a measurement, Round answers to correct number of SFs Percent Error Unit conversions in
Units 1, 2 study guide- atomic structure
 Name: Units 1, 2 study guide- atomic structure 1) Complete the required information for each subatomic particle (T1.3) symbol name charge location Mass (AMU) p + e - n 0 2) Define the following terms:
Name: Units 1, 2 study guide- atomic structure 1) Complete the required information for each subatomic particle (T1.3) symbol name charge location Mass (AMU) p + e - n 0 2) Define the following terms:
ATOMIC MATH HOMEWORK
 Name: Block: ATOMIC MATH HOMEWORK True/False: Indicate if each of the following is true or false. If it is false, CHANGE the underlined portion to make it true. 1. In a neutral atom of an element, the
Name: Block: ATOMIC MATH HOMEWORK True/False: Indicate if each of the following is true or false. If it is false, CHANGE the underlined portion to make it true. 1. In a neutral atom of an element, the
1. The arrangement of the elements from left to right in Period 4 on the Periodic Table is based on
 1. The arrangement of the elements from left to right in Period 4 on the Periodic Table is based on A) atomic mass B) atomic number C) the number of electron shells D) the number of oxidation states 2.
1. The arrangement of the elements from left to right in Period 4 on the Periodic Table is based on A) atomic mass B) atomic number C) the number of electron shells D) the number of oxidation states 2.
Lab safety and equipment: Sig Figs and Metric Conversions. Matter. Chemistry Review
 Lab safety and equipment: Chemistry Review Know: How to handle chemicals and laboratory equipment safely. Basic laboratory equipment Sig Figs and Metric Conversions Significant Figures How many significant
Lab safety and equipment: Chemistry Review Know: How to handle chemicals and laboratory equipment safely. Basic laboratory equipment Sig Figs and Metric Conversions Significant Figures How many significant
Solid Gas Liquid Plasma
 Unit 1: MATTER 1. Define CHEMISTRY: 2. Define MATTER: Use one of the states of matter to complete each statement. (Words will be used more than once.) Solid Gas Liquid Plasma 3. A has definite volume and
Unit 1: MATTER 1. Define CHEMISTRY: 2. Define MATTER: Use one of the states of matter to complete each statement. (Words will be used more than once.) Solid Gas Liquid Plasma 3. A has definite volume and
UNIT (2) ATOMS AND ELEMENTS
 UNIT (2) ATOMS AND ELEMENTS 2.1 Elements An element is a fundamental substance that cannot be broken down by chemical means into simpler substances. Each element is represented by an abbreviation called
UNIT (2) ATOMS AND ELEMENTS 2.1 Elements An element is a fundamental substance that cannot be broken down by chemical means into simpler substances. Each element is represented by an abbreviation called
Chemistry Midterm Review
 Chemistry Midterm Review Name To Do: 1) Make Note/Summary Sheet for each unit 2) Complete problems in this packet. As part of our review process, you will make a note sheet (max 1 SHEET per UNIT) that
Chemistry Midterm Review Name To Do: 1) Make Note/Summary Sheet for each unit 2) Complete problems in this packet. As part of our review process, you will make a note sheet (max 1 SHEET per UNIT) that
CP Physical Science Chemistry: Bell Work, Notes, Study Guides
 CP Physical Science Chemistry: Bell Work, Notes, Study Guides Mr. Banker Fall 2014 ian_banker@charleston.k12.sc.us http://wandohigh.ccsdschools.com/directory/science/banker_ian/physical_science/ Remind101.com
CP Physical Science Chemistry: Bell Work, Notes, Study Guides Mr. Banker Fall 2014 ian_banker@charleston.k12.sc.us http://wandohigh.ccsdschools.com/directory/science/banker_ian/physical_science/ Remind101.com
What is the smallest particle of the element gold (Au) that can still be classified as gold? A. atom B. molecule C. neutron D.
 Use the Periodic Table of Elements to answer the following question(s). Which sentence about the periodic table of elements is true? A. All elements in period 2 are metals. B. All elements in group 18
Use the Periodic Table of Elements to answer the following question(s). Which sentence about the periodic table of elements is true? A. All elements in period 2 are metals. B. All elements in group 18
Chapter 1 Introduction to Chemistry 1. What is chemistry?
 FIRST SEMESTER EXAM REVIEW Chapter 1 Introduction to Chemistry 1. What is chemistry? 2. Identify the independent and dependent variables using the graph above. IV: DV: 3. Write a hypothesis for this experiment.
FIRST SEMESTER EXAM REVIEW Chapter 1 Introduction to Chemistry 1. What is chemistry? 2. Identify the independent and dependent variables using the graph above. IV: DV: 3. Write a hypothesis for this experiment.
Assessment Chapter 5 Pre-Test Chapter: The Periodic Law Use the periodic table below to answer the questions in this Chapter Test.
 Assessment Chapter 5 Pre-Test Chapter: The Periodic Law Use the periodic table below to answer the questions in this Chapter Test. In the space provided, write the letter of the term or phrase that best
Assessment Chapter 5 Pre-Test Chapter: The Periodic Law Use the periodic table below to answer the questions in this Chapter Test. In the space provided, write the letter of the term or phrase that best
Chapter 2: Atoms and the Periodic Table
 1. Which element is a nonmetal? A) K B) Co C) Br D) Al Ans: C Difficulty: Easy 2. Which element is a metal? A) Li B) Si C) Cl D) Ar E) More than one of the elements above are metals. 3. Which element is
1. Which element is a nonmetal? A) K B) Co C) Br D) Al Ans: C Difficulty: Easy 2. Which element is a metal? A) Li B) Si C) Cl D) Ar E) More than one of the elements above are metals. 3. Which element is
Unit 2: Atoms and the Periodic Table
 Unit 2: Atoms and the Periodic Table Name Block Learning Goals A. Use the periodic table to identify the group and the period of an element. Your Prior Understanding of Learning Goals Excellent Good Okay
Unit 2: Atoms and the Periodic Table Name Block Learning Goals A. Use the periodic table to identify the group and the period of an element. Your Prior Understanding of Learning Goals Excellent Good Okay
Chemistry Chapter 9 Review. 2. Calculate the wavelength of a photon of blue light whose frequency is 6.3 x s -1.
 Chemistry Chapter 9 Review 1. What is the frequency of radiation that has a wavelength of 4.7 x 10-5 cm? 2. Calculate the wavelength of a photon of blue light whose frequency is 6.3 x 10 14 s -1. 3. The
Chemistry Chapter 9 Review 1. What is the frequency of radiation that has a wavelength of 4.7 x 10-5 cm? 2. Calculate the wavelength of a photon of blue light whose frequency is 6.3 x 10 14 s -1. 3. The
1. The elements on the Periodic Table are arranged in order of increasing A atomic mass C molar mass
 1. The elements on the Periodic Table are arranged in order of increasing A atomic mass C molar mass A Br, Ga, Hg C O, S, Se B atomic number D oxidation number 2. Which list includes elements with the
1. The elements on the Periodic Table are arranged in order of increasing A atomic mass C molar mass A Br, Ga, Hg C O, S, Se B atomic number D oxidation number 2. Which list includes elements with the
b. Na. d. So. 1 A basketball has more mass than a golf ball because:
 Chem I Semester Review All of the following are general characteristics of a substance in the liquid state except a. definite volume. c. not easily compressed. b. able to flow. d. definite shape. In the
Chem I Semester Review All of the following are general characteristics of a substance in the liquid state except a. definite volume. c. not easily compressed. b. able to flow. d. definite shape. In the
Period: Chemistry Semester 1 Final Exam Review Packet. 1. What is the difference between a hypothesis and a theory?
 Chemistry Name: Period: Chemistry Semester 1 Final Exam Review Packet 1. What is the difference between a hypothesis and a theory? 2. Distinguish between quantitative and qualitative observations. States
Chemistry Name: Period: Chemistry Semester 1 Final Exam Review Packet 1. What is the difference between a hypothesis and a theory? 2. Distinguish between quantitative and qualitative observations. States
Unit 2 Atomic Theory and Periodicity Review
 Unit 2 Atomic Theory and Periodicity Review Section I: History In each box, write the name of the scientist(s) associated with the statement. Choose from among the following: Democritus Thomson Bohr Schroedinger
Unit 2 Atomic Theory and Periodicity Review Section I: History In each box, write the name of the scientist(s) associated with the statement. Choose from among the following: Democritus Thomson Bohr Schroedinger
1. The elements on the Periodic Table are arranged in order of increasing A atomic mass C molar mass
 1. The elements on the Periodic Table are arranged in order of increasing A atomic mass C molar mass A Br, Ga, Hg C O, S, Se B atomic number D oxidation number 2. Which list includes elements with the
1. The elements on the Periodic Table are arranged in order of increasing A atomic mass C molar mass A Br, Ga, Hg C O, S, Se B atomic number D oxidation number 2. Which list includes elements with the
Chemistry B11 Chapter 3 Atoms
 Chapter 3 Atoms Element: is a substance that consists of identical atoms (hydrogen, oxygen, and Iron). 116 elements are known (88 occur in nature and chemist have made the others in the lab). Compound:
Chapter 3 Atoms Element: is a substance that consists of identical atoms (hydrogen, oxygen, and Iron). 116 elements are known (88 occur in nature and chemist have made the others in the lab). Compound:
1. What is the difference between a qualitative and quantitative observation? Give at least one example of each.
 1 st 9wks Exam Review Name Per. Chemistry 1H Scientific Skills Unit - Lab Safety & Equipment: KEY EQUIPMENT TO KNOW: +beaker +Erlenmeyer flask +beaker tongs +balance +graduated cylinder +test tube +test
1 st 9wks Exam Review Name Per. Chemistry 1H Scientific Skills Unit - Lab Safety & Equipment: KEY EQUIPMENT TO KNOW: +beaker +Erlenmeyer flask +beaker tongs +balance +graduated cylinder +test tube +test
Unit 3: Atoms and Periodic Table Retake Review Packet
 Name Date Block Unit 3: Atoms and Periodic Table Retake Review Packet Completing each section of the retake review packet will fulfill one of the requirements for retaking the unit 3 test. Section 1: Vocabulary
Name Date Block Unit 3: Atoms and Periodic Table Retake Review Packet Completing each section of the retake review packet will fulfill one of the requirements for retaking the unit 3 test. Section 1: Vocabulary
6. How many nonmetal atoms are there in the formula: NaH 2 PO 4? a. 1 b. 2 c. 4 d. 6 e. 7
 C101 Chapters 1 and 2 90 representative questions from old exams The questions are grouped by topic. Do these problems as homework to prepare for Exam 1. 1. Carbon is a metal/nonmetal that has the symbol.
C101 Chapters 1 and 2 90 representative questions from old exams The questions are grouped by topic. Do these problems as homework to prepare for Exam 1. 1. Carbon is a metal/nonmetal that has the symbol.
Lab safety and equipment: Sig Figs and Metric Conversions. Matter. Chemistry Review
 Lab safety and equipment: Chemistry Review Know: How to handle chemicals and laboratory equipment safely. Basic laboratory equipment Sig Figs and Metric Conversions Significant Figures How many significant
Lab safety and equipment: Chemistry Review Know: How to handle chemicals and laboratory equipment safely. Basic laboratory equipment Sig Figs and Metric Conversions Significant Figures How many significant
Unit 7 Study Guide: Name: KEY Atomic Concepts & Periodic Table
 Unit 7 Study Guide: Name: KEY Atomic Concepts & Periodic Table Focus Questions for the unit... How has the modern view of the atom changed over time? How does a chemist use symbols and notation to communicate
Unit 7 Study Guide: Name: KEY Atomic Concepts & Periodic Table Focus Questions for the unit... How has the modern view of the atom changed over time? How does a chemist use symbols and notation to communicate
Regents review Atomic & periodic
 2011-2012 1. The diagram below represents the nucleus of an atom. What are the atomic number and mass number of this atom? A) The atomic number is 9 and the mass number is 19. B) The atomic number is 9
2011-2012 1. The diagram below represents the nucleus of an atom. What are the atomic number and mass number of this atom? A) The atomic number is 9 and the mass number is 19. B) The atomic number is 9
CHEMISTRY I - HONORS MIDTERM REVIEW* *Test may cover other topics not included on this review, yet have been covered throughout the semester.
 Name Period CHEMISTRY I - HONORS MIDTERM REVIEW* *Test may cover other topics not included on this review, yet have been covered throughout the semester. Chapter 2 Measurement & Calculations Describe the
Name Period CHEMISTRY I - HONORS MIDTERM REVIEW* *Test may cover other topics not included on this review, yet have been covered throughout the semester. Chapter 2 Measurement & Calculations Describe the
Chemistry Final Review 2017
 Chemistry Final Review 2017 Atomic/Molecular Structure and Periodic Trends 1. What is the atomic number trend on the periodic table? 2. On the following periodic table label metals, nonmetals, Alkali metals,
Chemistry Final Review 2017 Atomic/Molecular Structure and Periodic Trends 1. What is the atomic number trend on the periodic table? 2. On the following periodic table label metals, nonmetals, Alkali metals,
A bit of review. Atoms are made of 3 different SUB-ATOMIC PARTICLES: 1. ELECTRONS 2. PROTONS 3. NEUTRONS
 Chemistry in Action A bit of review Chemistry is the study of MATTER and ENERGY. Matter is anything that has MASS. All matter is made of super small particles called ATOMS. Atoms are made of 3 different
Chemistry in Action A bit of review Chemistry is the study of MATTER and ENERGY. Matter is anything that has MASS. All matter is made of super small particles called ATOMS. Atoms are made of 3 different
Mr. Dolgos Regents Chemistry PRACTICE PACKET. Unit 3: Periodic Table
 *STUDENT* *STUDENT* Mr. Dolgos Regents Chemistry PRACTICE PACKET Unit 3: Periodic Table 2 3 It s Elemental DIRECTIONS: Use the reading below to answer the questions that follow. We all know by now that
*STUDENT* *STUDENT* Mr. Dolgos Regents Chemistry PRACTICE PACKET Unit 3: Periodic Table 2 3 It s Elemental DIRECTIONS: Use the reading below to answer the questions that follow. We all know by now that
Collegiate Institute for Math and Science Day 57: December 9, 2016 Room 427
 Unit 2: Atomic Concepts Outline Name: Period: Date: 1. The modern model of the atom has evolved over a long period of time through the work of many scientists. Dalton s Model: Elements are made of atoms
Unit 2: Atomic Concepts Outline Name: Period: Date: 1. The modern model of the atom has evolved over a long period of time through the work of many scientists. Dalton s Model: Elements are made of atoms
Fundamentals of General, Organic, and Biological Chemistry, 7e (McMurry) Chapter 2 Atoms and the Periodic Table
 Fundamentals of General, Organic, and Biological Chemistry, 7e (McMurry) Chapter 2 Atoms and the Periodic Table 1) The smallest amount of an element that retains that element's characteristics is the A)
Fundamentals of General, Organic, and Biological Chemistry, 7e (McMurry) Chapter 2 Atoms and the Periodic Table 1) The smallest amount of an element that retains that element's characteristics is the A)
FALL SEMESTER REVIEW NAME: PERIOD: 3. (Unit 2) Is the following picture representing accuracy, precision or both? Explain the difference.
 FALL SEMESTER REVIEW NAME: PERIOD: 1. (Unit 7) When the equation Cu + AgNO 3 Cu(NO3) 2 + Ag is balanced the coefficient of Ag NO 3 is? 2. (Unit 7) Balance the following equation: AgNO 3 + BaCl 2 AgCl +
FALL SEMESTER REVIEW NAME: PERIOD: 1. (Unit 7) When the equation Cu + AgNO 3 Cu(NO3) 2 + Ag is balanced the coefficient of Ag NO 3 is? 2. (Unit 7) Balance the following equation: AgNO 3 + BaCl 2 AgCl +
Name Midterm Review Date
 Name Midterm Review Date 1. In which process does a solid change directly into a vapor? A) sublimation B) deposition C) condensation D) solidification 2. What is the molecular formula of a compound that
Name Midterm Review Date 1. In which process does a solid change directly into a vapor? A) sublimation B) deposition C) condensation D) solidification 2. What is the molecular formula of a compound that
Additional Problem 1.13
 Task 1 Due: 11:59pm on Friday, April 27, 2018 You will receive no credit for items you complete after the assignment is due. Grading Policy Additional Problem 1.13 Classify each of the following as a pure
Task 1 Due: 11:59pm on Friday, April 27, 2018 You will receive no credit for items you complete after the assignment is due. Grading Policy Additional Problem 1.13 Classify each of the following as a pure
Electron Configuration & Periodicity Unit 3
 Name: Electron Configuration & Periodicity Unit 3 (seven class periods) Unit 3.1: First Ionization Energy & Photoelectron Spectroscopy 1) Coulombs Law a) The force of attraction between two charged objects
Name: Electron Configuration & Periodicity Unit 3 (seven class periods) Unit 3.1: First Ionization Energy & Photoelectron Spectroscopy 1) Coulombs Law a) The force of attraction between two charged objects
Notes: Electrons and Periodic Table (text Ch. 4 & 5)
 Name Per. Notes: Electrons and Periodic Table (text Ch. 4 & 5) NOTE: This set of class notes is not complete. We will be filling in information in class. If you are absent, it is your responsibility to
Name Per. Notes: Electrons and Periodic Table (text Ch. 4 & 5) NOTE: This set of class notes is not complete. We will be filling in information in class. If you are absent, it is your responsibility to
Chemistry 9 Weeks Exam Review Guide
 Name Date: Chemistry 9 Weeks Exam Review Guide 9 Weeks Exam: Tuesday, October 15 th Topics Covered Unit 1 Measurement: Accuracy & Precision, metric system, Conversions, Significant Figures, Percent Error,
Name Date: Chemistry 9 Weeks Exam Review Guide 9 Weeks Exam: Tuesday, October 15 th Topics Covered Unit 1 Measurement: Accuracy & Precision, metric system, Conversions, Significant Figures, Percent Error,
VOCABULARY define the following terms:
 VOCABULARY define the following terms: Unit 1 Study Guide Honors Chem 1) Electron configuration- 2) Effective nuclear charge 3) Isotope 4) Ion 5) Anion 6) Cation - 7) Isoelectronic - 8) Halogens 9) Electronegativity
VOCABULARY define the following terms: Unit 1 Study Guide Honors Chem 1) Electron configuration- 2) Effective nuclear charge 3) Isotope 4) Ion 5) Anion 6) Cation - 7) Isoelectronic - 8) Halogens 9) Electronegativity
3.1 Classification of Matter. Copyright 2009 by Pearson Education, Inc.
 Chapter 3 Atoms and Elements 3.1 Classification of Matter Copyright 2009 by Pearson Education, Inc. 1 Matter Matter is the stuff that makes up all things. Copyright 2009 by Pearson Education, Inc. 2 Pure
Chapter 3 Atoms and Elements 3.1 Classification of Matter Copyright 2009 by Pearson Education, Inc. 1 Matter Matter is the stuff that makes up all things. Copyright 2009 by Pearson Education, Inc. 2 Pure
M T W R. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. (3 pts ea)
 Exam 1 A Fall 2016 a Seat # Name M T W R MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. (3 pts ea) 1) Which of the following is a chemical property?
Exam 1 A Fall 2016 a Seat # Name M T W R MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. (3 pts ea) 1) Which of the following is a chemical property?
Review. 8th grade science STAAR. Name Class. Underline your strong TEKS and circle your weak TEKS: 8.5A Atoms. 7.5C Energy Flow Through Ecosystems
 8th grade science STAAR Review Name Class Underline your strong TEKS and circle your weak TEKS: 8.5A Atoms 7.5C Energy Flow Through Ecosystems 8.5B Reactivity 8.5C Periodic Table 8.5D Chemical Formulas
8th grade science STAAR Review Name Class Underline your strong TEKS and circle your weak TEKS: 8.5A Atoms 7.5C Energy Flow Through Ecosystems 8.5B Reactivity 8.5C Periodic Table 8.5D Chemical Formulas
Test Review # 5. Chemistry: Form TR5-8A. Average Atomic Mass. Subatomic particles.
 Chemistry: Form TR5-8A REVIEW Name Date Period Test Review # 5 Subatomic particles. Type of Particle Location Mass Relative Mass Charge Proton Center 1.67 10-27 kg 1 +1 Electron Outside 9.11 10-31 kg 0-1
Chemistry: Form TR5-8A REVIEW Name Date Period Test Review # 5 Subatomic particles. Type of Particle Location Mass Relative Mass Charge Proton Center 1.67 10-27 kg 1 +1 Electron Outside 9.11 10-31 kg 0-1
Test Review # 4. Chemistry: Form TR4-9A
 Chemistry: Form TR4-9A REVIEW Name Date Period Test Review # 4 Location of electrons. Electrons are in regions of the atom known as orbitals, which are found in subdivisions of the principal energy levels
Chemistry: Form TR4-9A REVIEW Name Date Period Test Review # 4 Location of electrons. Electrons are in regions of the atom known as orbitals, which are found in subdivisions of the principal energy levels
Test 3: Lab Safety, Measurements, Matter and Periodic Table
 Name: Grade/Group: Subject: Chemistry-7 Teacher: Mrs. Raj Date: Test 3: Lab Safety, Measurements, Matter and Periodic Table Directions: Determine the best answer for each question. Circle your answer on
Name: Grade/Group: Subject: Chemistry-7 Teacher: Mrs. Raj Date: Test 3: Lab Safety, Measurements, Matter and Periodic Table Directions: Determine the best answer for each question. Circle your answer on
2. Which is NOT considered matter? (A) air (B) lead (C) sugar (D) sunlight. 4. Which of the following is a metalloid? (A) Se (B) Sr (C) Si (D) Sn
 Midterm Review Packet ANSWER KEY 01/26/2014 Multiple Choice Question 1. Which part of an atom is positively charged? (A) proton (B) neutron (C) electron (D) shells 2. Which is NOT considered matter? (A)
Midterm Review Packet ANSWER KEY 01/26/2014 Multiple Choice Question 1. Which part of an atom is positively charged? (A) proton (B) neutron (C) electron (D) shells 2. Which is NOT considered matter? (A)
AlCl3(aq) + 3 NaOH(aq) Al(OH)3(s) + 3 NaCl(aq)
 1. Under which conditions does a real gas behave most like an ideal gas? A) at low temperatures and high pressures B) at low temperatures and low pressures C) at high temperatures and high pressures D)
1. Under which conditions does a real gas behave most like an ideal gas? A) at low temperatures and high pressures B) at low temperatures and low pressures C) at high temperatures and high pressures D)
Classify each of these statements as always true, AT; sometimes true, ST; or never true, NT.
 ELECTRONS IN ATOMS Chapter Quiz Classify each of these statements as always true, AT; sometimes true, ST; or never true, NT. 1. The orbitals of a principal energy level are lower in energy than the orbitals
ELECTRONS IN ATOMS Chapter Quiz Classify each of these statements as always true, AT; sometimes true, ST; or never true, NT. 1. The orbitals of a principal energy level are lower in energy than the orbitals
CHE 105 Exam 1 Fall 2016
 CHE 105 Exam 1 Fall 2016 Your Name: Your ID: attachment_for_pubexamuid_lnxp114738117286913141xx_99.jpg Question #: 1 The state of matter in which atoms or molecules have a fixed volume but are free to
CHE 105 Exam 1 Fall 2016 Your Name: Your ID: attachment_for_pubexamuid_lnxp114738117286913141xx_99.jpg Question #: 1 The state of matter in which atoms or molecules have a fixed volume but are free to
Unit Five Practice Test (Part I) PT C U5 P1
 Unit Five Practice Test (Part I) PT C U5 P1 Name Period LPS Standard(s): --- State Standard(s): 12.3.1 Short Answers. Answer the following questions. (5 points each) 1. Write the electron configuration
Unit Five Practice Test (Part I) PT C U5 P1 Name Period LPS Standard(s): --- State Standard(s): 12.3.1 Short Answers. Answer the following questions. (5 points each) 1. Write the electron configuration
CH1410 Practice Exam #1
 CH1410 Practice Exam #1 Section 1 - Multiple Choice - Write the letter of the BEST CHOICE in the space provided. 1. Which state of matter has atomic spacing that is close together and indefinite shape?
CH1410 Practice Exam #1 Section 1 - Multiple Choice - Write the letter of the BEST CHOICE in the space provided. 1. Which state of matter has atomic spacing that is close together and indefinite shape?
1 st Semester Final Review Sheet (Chapters 2-7)
 CP Chemistry Name: Period: 1 st Semester Final Review Sheet (Chapters 2-7) Matter (Chapter 2) 1. What is the definition of an element? 2. What is the definition of a compound? 3. What is the definition
CP Chemistry Name: Period: 1 st Semester Final Review Sheet (Chapters 2-7) Matter (Chapter 2) 1. What is the definition of an element? 2. What is the definition of a compound? 3. What is the definition
Multiple Choice Identify the choice that best completes the statement or answers the question.
 Chemi1100pretest1 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Accordingly to the periodic law the properties of elements repeat at regular intervals
Chemi1100pretest1 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Accordingly to the periodic law the properties of elements repeat at regular intervals
Chapter 6 The Periodic Table
 Chapter 6 The Periodic Table Section 6.1 Organizing the Elements OBJECTIVES: Explain how elements are organized in a periodic table. Section 6.1 Organizing the Elements OBJECTIVES: Compare early and modern
Chapter 6 The Periodic Table Section 6.1 Organizing the Elements OBJECTIVES: Explain how elements are organized in a periodic table. Section 6.1 Organizing the Elements OBJECTIVES: Compare early and modern
Chemistry CRT Study Guide First Quarter
 Number AL COS # 1. #1.0 Classify sodium chloride as an element, mixture, compound, or colloid. Compound 2. #1.0 Classify air as an element, mixture, compound, or colloid. Mixture 3. #1.0 Classify a blueberry
Number AL COS # 1. #1.0 Classify sodium chloride as an element, mixture, compound, or colloid. Compound 2. #1.0 Classify air as an element, mixture, compound, or colloid. Mixture 3. #1.0 Classify a blueberry
VOCABULARY define the following terms:
 VOCABULARY define the following terms: Unit 1 Study Guide Honors Chem 1) Electron configuration- 2) Effective nuclear charge 3) Isotope 4) Ion 5) Anion 6) Cation - 7) Isoelectronic - 8) Halogens 9) Electronegativity
VOCABULARY define the following terms: Unit 1 Study Guide Honors Chem 1) Electron configuration- 2) Effective nuclear charge 3) Isotope 4) Ion 5) Anion 6) Cation - 7) Isoelectronic - 8) Halogens 9) Electronegativity
 1. Dimensional Analysis: convert the following values a. 47,340 cm to m Unit 1: Chemistry Matters b. 40.64 km to m c. 58,700 ml to L 2. Calculate the number of significant figures a. 0.0210 b. 3.6056 c.
1. Dimensional Analysis: convert the following values a. 47,340 cm to m Unit 1: Chemistry Matters b. 40.64 km to m c. 58,700 ml to L 2. Calculate the number of significant figures a. 0.0210 b. 3.6056 c.
PERIODIC TRENDS AND THE PERIODIC TABLE
 PERIODIC TRENDS AND THE PERIODIC TABLE THE PERIODIC TABLE The row tells us how many energy levels are in that atom The row is also the group The column tells us how many electrons are in the outer energy
PERIODIC TRENDS AND THE PERIODIC TABLE THE PERIODIC TABLE The row tells us how many energy levels are in that atom The row is also the group The column tells us how many electrons are in the outer energy
Metric System: 1. The basic unit of length in the metric system is the (a) kilometer (b) mile (c) foot (d) meter (e) none of these 2.
 Metric System: 1. The basic unit of length in the metric system is the (a) kilometer (b) mile (c) foot (d) meter (e) none of these 2. The basic unit of mass in the metric system is the _ (a) gram (b) kilogram
Metric System: 1. The basic unit of length in the metric system is the (a) kilometer (b) mile (c) foot (d) meter (e) none of these 2. The basic unit of mass in the metric system is the _ (a) gram (b) kilogram
Matter and Energy. Chapter 3
 Matter and Energy Chapter 3 Matter Anything that has mass and takes up space Two categories Pure substances Mixtures Pure Substances Matter with a fixed composition Either an element or compound Element
Matter and Energy Chapter 3 Matter Anything that has mass and takes up space Two categories Pure substances Mixtures Pure Substances Matter with a fixed composition Either an element or compound Element
Name: Midterm Review Date:
 Name: Midterm Review Date: 1. Which statement concerning elements is true? A) Different elements must have different numbers of isotopes. B) Different elements must have different numbers of neutrons.
Name: Midterm Review Date: 1. Which statement concerning elements is true? A) Different elements must have different numbers of isotopes. B) Different elements must have different numbers of neutrons.
Chem 110 Fall 2014 Exam I Whelan
 Chem 110 Fall 2014 Exam I Whelan SID Last First Question 1 5 Points a) How many significant figures are there in each of the following numbers? 0.927790 0.060464 1.00x10 3 b) There are 12 eggs in a dozen.
Chem 110 Fall 2014 Exam I Whelan SID Last First Question 1 5 Points a) How many significant figures are there in each of the following numbers? 0.927790 0.060464 1.00x10 3 b) There are 12 eggs in a dozen.
Chapter 3: Electron Structure and the Periodic Law
 Chapter 3: Electron Structure and the Periodic Law PERIODIC LAW This is a statement about the behavior of the elements when they are arranged in a specific order. In its present form the statement is:
Chapter 3: Electron Structure and the Periodic Law PERIODIC LAW This is a statement about the behavior of the elements when they are arranged in a specific order. In its present form the statement is:
9/13/2011. The Greek Philosophers. Atomic Structure & The Periodic Table. Dalton s Atomic Theory. J. J. Thomson. Thomson s Experiment
 Atomic Structure & The Periodic Table The Greek Philosophers Democritus believed that all matter is made up of tiny particles that could not be divided Aristotle -- thought that matter was made of only
Atomic Structure & The Periodic Table The Greek Philosophers Democritus believed that all matter is made up of tiny particles that could not be divided Aristotle -- thought that matter was made of only
Chemistry Mid-Term Practice Exam
 Chemistry Mid-Term Practice Exam Multiple Choice. Identify the choice that best completes the statement or answers the question. 1. A measure of the 3-D space matter occupies is a. density. c. volume.
Chemistry Mid-Term Practice Exam Multiple Choice. Identify the choice that best completes the statement or answers the question. 1. A measure of the 3-D space matter occupies is a. density. c. volume.
3. (3) For each of the following measurements, state the number of significant figures, the uncertainty, and the range of possible values.
 Exam 1 Chem 30A, Fall 11 Fossum Name: Note: there are 5 points of extra credit built in to this exam. 1. (2) Shown here is a close-up of part of a ruler. The units are centimeters. If you were measuring
Exam 1 Chem 30A, Fall 11 Fossum Name: Note: there are 5 points of extra credit built in to this exam. 1. (2) Shown here is a close-up of part of a ruler. The units are centimeters. If you were measuring
Atoms, molecules, bonding, periodic table
 Atoms, molecules, bonding, periodic table Atoms Modern Atom Model Nucleus-Protons and Neutrons Electrons around nucleus, never know the true location Protons Positively charged In nucleus Neutrons Neutral
Atoms, molecules, bonding, periodic table Atoms Modern Atom Model Nucleus-Protons and Neutrons Electrons around nucleus, never know the true location Protons Positively charged In nucleus Neutrons Neutral
A1: Atomic Structure Worksheet (Goals 1 3, Chapter 4)
 Unit 3 Assignment Packet Name: Period: A1: Atomic Structure Worksheet (Goals 1 3, Chapter 4) 1. Democritus, who lived in Greece during the 4 th century B.C., suggested that is made up of tiny particles
Unit 3 Assignment Packet Name: Period: A1: Atomic Structure Worksheet (Goals 1 3, Chapter 4) 1. Democritus, who lived in Greece during the 4 th century B.C., suggested that is made up of tiny particles
8. (5 pts) Draw a sketch of an atom. Label the nucleus, protons, neutrons. nucleus, containing protons and neutrons
 1. (2 pts) Atoms of the same element, regardless of charge, all have the same number of protons. 2. (2 pts) Comparing the mass of an electron to the mass of a proton, one could say that the electron is
1. (2 pts) Atoms of the same element, regardless of charge, all have the same number of protons. 2. (2 pts) Comparing the mass of an electron to the mass of a proton, one could say that the electron is
Unit 3: The Periodic Table and Atomic Theory
 Name: Period: Unit 3: The Periodic Table and Atomic Theory Day Page # Description IC/HW 1 2-3 Periodic Table and Quantum Model Notes IC 1 4-5 Orbital Diagrams Notes IC 1 14 3-A: Orbital Diagrams Worksheet
Name: Period: Unit 3: The Periodic Table and Atomic Theory Day Page # Description IC/HW 1 2-3 Periodic Table and Quantum Model Notes IC 1 4-5 Orbital Diagrams Notes IC 1 14 3-A: Orbital Diagrams Worksheet
Full file at
 16 Chapter 2: Atoms and the Periodic Table Solutions to In-Chapter Problems 2.1 Each element is identified by a one- or two-letter symbol. Use the periodic table to find the symbol for each element. a.
16 Chapter 2: Atoms and the Periodic Table Solutions to In-Chapter Problems 2.1 Each element is identified by a one- or two-letter symbol. Use the periodic table to find the symbol for each element. a.
Atomic Structure Practice Questions
 Atomic Structure Practice Questions 1. Experiments performed to reveal the structure of atoms led scientists to conclude that an atom s (1) positive charge is evenly distributed throughout its volume (2)
Atomic Structure Practice Questions 1. Experiments performed to reveal the structure of atoms led scientists to conclude that an atom s (1) positive charge is evenly distributed throughout its volume (2)
5. The outermost principal energy level electron configuration of the element bromine is: a. 4s 2 c. 4s 2 4p 5 b. 4p 5 d.
 1 c E = h 1. Sodium and potassium have similar properties because they have the same a. atomic radii. c. number of valence electrons. b. ionization energy. d. electronegativity. 2. Electrons must be added
1 c E = h 1. Sodium and potassium have similar properties because they have the same a. atomic radii. c. number of valence electrons. b. ionization energy. d. electronegativity. 2. Electrons must be added
HONORS CHEMISTRY. Chapter 1 Introduction to Chemistry 1. What is chemistry?
 HONORS CHEMISTRY FIRST SEMESTER EXAM REVIEW Chapter 1 Introduction to Chemistry 1. What is chemistry? 2. Identify the independent and dependent variables using the graph to the right. IV: DV: 3. Write
HONORS CHEMISTRY FIRST SEMESTER EXAM REVIEW Chapter 1 Introduction to Chemistry 1. What is chemistry? 2. Identify the independent and dependent variables using the graph to the right. IV: DV: 3. Write
Chapter 2: Atoms and the Periodic Table
 1. Which element is a nonmetal? A) K B) Co C) Br D) Al Ans: C Difficulty: Easy 2. Which element is a metal? A) Li B) Si C) Cl D) Ar E) More than one of the elements above is a metal. Ans: A Difficulty:
1. Which element is a nonmetal? A) K B) Co C) Br D) Al Ans: C Difficulty: Easy 2. Which element is a metal? A) Li B) Si C) Cl D) Ar E) More than one of the elements above is a metal. Ans: A Difficulty:
A.P. Chemistry Practice Test - Ch. 7, Atomic Structure and Periodicity
 A.P. Chemistry Practice Test - Ch. 7, Atomic Structure and Periodicity 1) Ham radio operators often broadcast on the 6-meter band. The frequency of this electromagnetic radiation is MHz. A) 50 B) 20 C)
A.P. Chemistry Practice Test - Ch. 7, Atomic Structure and Periodicity 1) Ham radio operators often broadcast on the 6-meter band. The frequency of this electromagnetic radiation is MHz. A) 50 B) 20 C)
Regular Chemistry - 1st Semester Final Practice Exam
 Regular Chemistry - 1st Semester Final Practice Exam Mr. Matthew Totaro MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which statement about the
Regular Chemistry - 1st Semester Final Practice Exam Mr. Matthew Totaro MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which statement about the
Chem 12 Unit 1 Review: Chapters 1-4, 9
 1 Chem 12 Unit 1 Review: Chapters 1-4, 9 1. What are the steps in the Scientific Method? Significant Figures: 2. Counting Significant Figures: Circle the significant figures in the following: a) 5280 feet
1 Chem 12 Unit 1 Review: Chapters 1-4, 9 1. What are the steps in the Scientific Method? Significant Figures: 2. Counting Significant Figures: Circle the significant figures in the following: a) 5280 feet
CP Chemistry Midterm Study Guide What is the proper way to measure a volume in a graduated cylinder?
 Name Date CP Chemistry Midterm Study Guide 2017 List 5 laboratory safety rules. What is the proper way to measure a volume in a graduated cylinder? Name 5 pieces of equipment used in the laboratory, and
Name Date CP Chemistry Midterm Study Guide 2017 List 5 laboratory safety rules. What is the proper way to measure a volume in a graduated cylinder? Name 5 pieces of equipment used in the laboratory, and
Chapter 2 Atoms and the Periodic Table
 Chapter 2 1 Chapter 2 Atoms and the Periodic Table Solutions to In-Chapter Problems 2.1 Each element is identified by a one- or two-letter symbol. Use the periodic table to find the symbol for each element.
Chapter 2 1 Chapter 2 Atoms and the Periodic Table Solutions to In-Chapter Problems 2.1 Each element is identified by a one- or two-letter symbol. Use the periodic table to find the symbol for each element.
