Shaggy, the Homolog of Glycogen Synthase Kinase 3, Controls Neuromuscular Junction Growth in Drosophila
|
|
|
- June Woods
- 5 years ago
- Views:
Transcription
1 The Journal of Neuroscience, July 21, (29): Brief Communication Shaggy, the Homolog of Glycogen Synthase Kinase 3, Controls Neuromuscular Junction Growth in Drosophila Bénédicte Franco, 1 Laurent Bogdanik, 1 Yves Bobinnec, 2 Alain Debec, 2 Joël Bockaert, 1 Marie-Laure Parmentier, 1 and Yves Grau 1 1 Laboratoire de Génomique Fonctionnelle, Unité Propre de Recherche Centre National de la Recherche Scientifique 2580, Montpellier Cedex 5, France, and 2 Biologie du Développement, Unité Mixte de Recherche 7009, Observatoire Océanologique, Villefranche sur Mer, France A protein-trap screen using the Drosophila neuromuscular junction (NMJ) as a model synapse was performed to identify genes that control synaptic structure or plasticity. We found that Shaggy (Sgg), the Drosophila homolog of the mammalian glycogen synthase kinases 3 and, two serine threonine kinases, was concentrated at this synapse. Using various combinations of mutant alleles of shaggy, we found that Shaggy negatively controlled the NMJ growth. Moreover, tissue-specific expression of a dominant-negative Sgg indicated that this kinase is required in the motoneuron, but not in the muscle, to control NMJ growth. Finally, we show that Sgg controlled the microtubule cytoskeleton dynamics in the motoneuron and that Futsch, a microtubule-associated protein, was required for Shaggy function on synaptic growth. Key words: motoneuron; morphological plasticity; microtubule loop; microtubule-associated protein; glycogen synthase kinase; synaptic plasticity Received Nov. 12, 2003; revised June 1, 2004; accepted June 1, This work was supported by the Centre National de la Recherche Scientifique, Institut National de la Santé etde la Recherche Médicale, and the French Ministry of Research and Technology. We thank V. Budnik, E. Buchner, G. Davis, P. Heitzler, C. Klämbt, C. O Kane, C. Schuster, and D. Woods for providing fly stocks or antibodies. We thank M. Bourouis and V. Homburger for discussions and J.-P. Pin, F. Maschat, J.-M. Dura, and C. Mitri for critical reading of this manuscript. Correspondence should be addressed to Marie-Laure Parmentier, Unité Propre de Recherche Centre National de la Recherche Scientifique 2580, 141 rue de la Cardonille Montpellier Cedex 5, France. mlparmentier@ccipe.cnrs.fr. DOI: /JNEUROSCI Copyright 2004 Society for Neuroscience /04/ $15.00/0 Introduction Once established, synaptic connections can be modified throughout animal life. Physiological tuning of synapses may be accompanied by morphological changes of these synapses. Such a synaptic plasticity is thought to be the cellular basis of information processing and storage in the brain (Muller et al., 2002). To identify the molecular mechanisms controlling synaptic structure and plasticity, we undertook a genetic approach in Drosophila melanogaster, focusing on the larval neuromuscular junction (NMJ). This synapse is functional at the end of embryogenesis and is required for the contraction of the body-wall muscles, leading to larval hatching and larval locomotion. This synapse shares important features with central excitatory synapses of the mammalian brain: it is a glutamatergic synapse containing ionotropic glutamate receptors, the postsynaptic density protein Discs-Large (DLG; homologous to PSD-95) and other proteins also found in mammalian central synapses (Gramates and Budnik, 1999; Broadie and Richmond, 2002). Finally, the Drosophila NMJ is a dynamic structure with morphological plasticity that depends on its activity as well as on the size of the target muscles. For example, during larval development, there is a 10-fold increase in the number of synaptic boutons in correlation with the growth of the target muscle (Schuster et al., 1996a). Furthermore, the hyperactive eag Sh mutants show a twofold increase in the number of boutons compared with wild-type larvae (Schuster et al., 1996b). Protein kinases are responsible for the phosphorylation of proteins, a major post-translational modification involved in numerous signaling and regulation pathways. Protein kinases represent one of the largest families of proteins, all possessing a common catalytic core. There are 450 kinases in the human genome and 255 kinases in the Drosophila genome. Many of these kinases play a role from early development onward, and mutants for the corresponding genes die during embryonic development. Thus, it is difficult to genetically screen for the role of such proteins in synaptic structure and plasticity at late stages of development when the synapses are established. Here, we used a protein-trap screen (Morin et al., 2001) where a green fluorescent protein (GFP) exon is inserted within genes, leading to fusion fluorescent proteins. We selected a line where this exon is inserted in the shaggy (sgg) gene. Shaggy is the Drosophila homolog of mammalian glycogen synthase kinase (GSK) 3. We observed that Sgg is concentrated in the presynaptic side of the NMJ. Using a hypomorph mutation of sgg, we discovered that this kinase negatively controls NMJ growth. Motoneuronspecific expression of a dominant-negative isoform of Sgg revealed that this kinase is required presynaptically for this NMJ growth control. Finally, inhibition of Sgg leads to an increase in the number of microtubule loops, suggesting that part of the growth control of the NMJ by Sgg occurs via changes in the microtubule cytoskeleton dynamics. This is supported by epistasis experiments between sgg and futsch, a gene encoding a
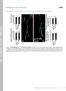
2 6574 J. Neurosci., July 21, (29): Franco et al. Shaggy Negatively Controls NMJ Growth microtubule-associated protein (MAP), showing that futsch is indeed downstream of sgg. Materials and Methods Fly stocks. The sgg K22 mutant and the sgg mini10 transgene were obtained from P. Heitzler (Institut de Génétique et de Biologie Moléculaire et Cellulaire, Illkirch, France). The sgg mini10 construct carries the cdna sequence of the SGG10 isoform fused to genomic untranslated region sequences of sgg (Ruel et al., 1993). Upstream activating sequence (UAS)-sgg DN stocks (on chromosomes 2 and 3) and deficiency Df(1)64j4 were obtained from the Bloomington Stock Center (Bloomington, IN). We used the Gal4 drivers elavc155 and myosin heavy chain (MHC)-Gal4 (C. Goodman, University of California, Berkeley, CA), BG380 (V. Budnik, University of Massachusetts Medical School, Worcester, MA), and OK6 (C. O Kane, University of Cambridge, Cambridge, UK). We obtained stronger phenotypes with the chromosome 2 UAS-sgg DN (see Fig. 3C) compared with chromosome 3 UAS-sgg DN (see Fig. 4 D). The futsch N94 and futsch K68 alleles were obtained from C. Klämbt (Universität of Münster, Münster, Germany) and G. Davis (University of California, San Francisco, CA). All stocks were reared at 25 C, except crosses with the UASsgg DN that were reared at 29 C during larval stages and larvae used for Figure 4D. Wild-type flies were Canton S (CS) or yw CS. To perform epistasis experiments between futsch and sgg, we constructed a (OK6-Gal4; UAS-sgg DN )/SM5:TM6B stock, which was crossed with futsch mutant females to obtain futsch N94 or futsch K68 /Y; OK6/ ; UAS-sgg DN / larvae. Immunocytochemistry and confocal microscopy. Wandering third instar larvae were dissected in 4% paraformaldehyde and 1 PBS. The following antibodies were used: mouse monoclonal anti-gsk-3, raised against an epitope conserved in Sgg (1:100; Upstate Biotechnology, Lake Placid, NY), guinea-pig anti-dlg (1:1000; D. Woods, University of California, Irvine, CA), mouse monoclonal anti-synapsin (1:100; E. Buchner, Universität Würzburg, Würzburg, Germany), mouse monoclonal anti-cystein string protein (CSP) (1:10, K. Zinsmaier, University of Arizona, Tucson, AZ), mouse monoclonal anti-futsch (22C10, 1:100) (Roos et al., 2000), and rabbit polyclonal anti-horse radish peroxydase (HRP) (1:1000; Sigma, St. Louis, MO). 22C10 was obtained from the Developmental Studies Hybridoma Bank (University of Iowa, Iowa City, IA). Fluorescent-conjugated antibodies were from Jackson ImmunoResearch (West Grove, PA) and Molecular Probes (Eugene, OR). Confocal images were acquired using a Bio-Rad (Hercules, CA) MRC 1024 laser-scanning confocal microscope (Centre Régional d Imagerie Cellulaire, Montpellier, France). Quantification. Synaptic boutons (only type Ib) on muscles 6/7 of the A3 segment were counted from HRP-stained preparations on a wide-field fluorescence microscope (Zeiss Axiophot; Zeiss, Thornwood, NY) at 100 magnification. A picture of the inner surface of muscle 6/7 was taken at 20 magnification to quantify the muscle area. The normalized bouton number was calculated by dividing bouton numbers by muscle surface area (Schuster et al., 1996a). Data were expressed as a percentage of the control of each experiment. We observed no major differences in muscle size between the genotypes. Hence, rough bouton number quantification yielded the same phenotypes as normalized bouton number. To correct for variations in total bouton number, we quantified the amount of satellite boutons as a percentage of the mean total bouton number for each genotype. Statistical analysis was performed using a two-tailed Student s t test. Results SGG is enriched in the presynaptic side at the NMJ To visualize proteins concentrated at the NMJ, we took advantage of a GFP transposon-based screen, in which a GFP sequence engineered as an exon is randomly inserted in genes (Morin et al., Figure 1. Synaptic localization of Sgg::GFP and Shaggy. GFP fluorescence (A, D, G) and anti-sgg ( B), anti-dlg ( E), and anti-synapsin (SYN) ( H ) immunostainings on protein trap line. All NMJs shown are located on muscle 4, but the same results could be observed on any other body-wall muscle. Color merges are shown (C, F, I), with higher magnification of one varicosity in C and I. Small arrows indicate type Ib boutons, and arrowheads indicate type Is boutons. Large arrows indicate the nerve branch. Scale bars: A C, G I,20 m; D F,10 m. 2001). Insertions of the GFP exon in frame with an expressed gene were recovered by screening larvae for fluorescence. We selected lines that presented a clear fluorescence at the third instar larvae NMJs. In line 105-1, the GFP exon was inserted in the second intron of the sgg gene (position 8665 of scaffold AE ), thus producing an Sgg::GFP fusion protein. We confirmed the in-frame insertion of the GFP sequence by 5 and 3 rapid amplification of cdna ends experiments (data not shown). Null mutations of sgg are lethal at the embryonic stage or the first instar larval stage (Ruel et al., 1993). Here, the transgenic line was homozygous viable and fertile, suggesting that the Sgg::GFP fusion protein was functional. This transgenic line presented a clear fluorescence at the NMJs of all body-wall muscles. Both type Ib and Is synaptic boutons, which correspond to different glutamatergic motoneurons innervating the muscles, expressed Sgg::GFP (Fig. 1A). At the NMJ, all of the GFP signal overlapped with an anti-sgg immunostaining, indicating that the GFP signal indeed corresponded to tagged Shaggy proteins (Fig. 1A C). We obtained qualitatively the same anti-sgg signal on wild-type CS flies, indicating that Sgg::GFP localization was the same as the wild-type Sgg localization (data not shown). Comparison of Sgg::GFP fluorescence with the postsynaptic marker DLG (Lahey et al., 1994) revealed that Sgg::GFP was not localized in the postsynaptic side but was presynaptic (Fig. 1D F). We confirmed this presynaptic localization by observing that Sgg::GFP colocalized with the vesicle marker Synapsin (Klagges et al., 1996) (Fig. 1G I). Moreover, we also detected some clear Sgg::GFP signal in axonal branches (Fig. 1A, G, arrows). Altogether, these results show that Sgg is enriched in the presynaptic side of the NMJ. Mutation in sgg alters the growth of the NMJ To examine sgg function during the development of the NMJ, we used the hypomorph mutation sgg K22. This mutation is viable until the third instar larval stage. We did not detect any Sgg immunoreactivity at the NMJs of sgg K22 third instar larvae (Fig. 2A D) nor any Sgg product on Western blot from third instar larva protein extracts (data not shown). In all of our experiments using sgg mutants, we chose crawling larvae, indicating that the NMJs were functional. During larval development, there is constant adaptation of the NMJ size to the increase in muscle size. This includes an increase in the number and size of synaptic boutons as well as in the
3 Franco et al. Shaggy Negatively Controls NMJ Growth J. Neurosci., July 21, (29): Figure 2. Morphological phenotype of sgg mutants. A D, Anti-HRP (A, C) and anti-sgg (B, D) immunostainings on a CS male larva (A, B)orsgg K22 /Y larva (C, D). E H, Illustration of the overgrowth phenotype in sgg K22 /Y ( F)orsgg K22 /Df(1)64j4 ( G) mutant larvae. This phenotype is rescued to the wild-type range ( E) by the addition of an sgg minigene (mini10) in the genome of sgg K22 mutants ( H ). The NMJs of muscle 6/7 are visualized with an anti-hrp staining. I, Quantification of the total number of Ib boutons and the percentage of satellite boutons at muscles 6/7 NMJs of CS male (n 12), sgg K22 /Y (n 25), sgg K22 /Df(1)64j4 (n 29), sgg K22 /Y;mini10/ (n 14), and yw CS / ;mini10/ (n 13) larvae. Statistical comparisons to the CS control: *p 0.05; **p 0.01; ***p J, Anti-SYN (green) and anti-hrp (red) double staining on an sgg K22 /Y muscle 13 NMJ. K, Anti-DLG (green) and anti-hrp (red) double staining on an sgg K22 /Y muscle 13 NMJ. In all images, arrowheads indicate satellite boutons. Scale bars: A D,20 m; F H,11 m; J, K,14 m. number of active zones (Schuster et al., 1996a; Lnenicka and Keshishian, 2000). We examined synaptic morphology by labeling presynaptic terminals using anti-hrp, an insect neuronal marker. Analysis of NMJs in sgg K22 mutants showed that the loss of sgg function had clear consequences for the growth of the synapse. Indeed, NMJ expansion was dramatically increased, resulting in an 80% increase in the number of type Ib boutons on muscle 6/7 in sgg K22 mutant larvae compared with wild-type controls (Fig. 2E I) ( for CS; for sgg K22 /Y). To ensure that this phenotype was caused by shaggy loss of function and not to a second site mutation, we used the deficiency Df(1)64j4 known to delete the entire sgg gene (Ruel et al., 1993). sgg K22 /Df(1)64j4 larvae also displayed a 60% increase in bouton number ( ). Interestingly, in both sgg loss-offunction genotypes, the increase in bouton number was accompanied by a twofold increase in the number of satellite boutons (Torroja et al., 1999) [ for CS; for sgg K22 /Y; for sgg K22 /Df(1)64j4] (i.e., boutons budding off from a bouton present in the axis of the branch, or boutons budding off from neuronal connections between two boutons) (Fig. 2F I). Both phenotypes of sgg K22 mutant larvae were rescued to wildtype range by adding one copy of a wild-type sgg minigene (mini10) in the genome ( for type Ib boutons; for satellite boutons) (Fig. 2H, I). This demonstrates that these phenotypes were attributable to the loss of shaggy function. Because sgg loss-of-function mutants displayed an overdeveloped NMJ, we would expect to observe the opposite phenotype in the presence of a gain of sgg function. We tested this hypothesis by quantifying the number of synaptic boutons in larvae possessing, in addition to the wild-type sgg gene on the X chromosome, an additional autosomal copy of sgg. In these larvae, the total number of Ib boutons ( ) was decreased compared with the control larvae, whereas the percentage of satellite boutons ( ) was not significantly modified. These data show that a dose increase of sgg is sufficient to slightly shrink the NMJ. To determine whether all boutons in sgg mutant synapses had normal features of synaptic boutons, we studied the localization of presynaptic and postsynaptic markers. Analysis of presynaptic synapsin (Fig. 2J) and CSP (data not shown) revealed that each bouton, and notably the satellite ones, was immunopositive for these markers. Similarly, we found that all boutons were surrounded by postsynaptic DLG immunoreactivity (Fig. 2K). Thus, the mutant boutons, showing a wild-type localization of some elements of the presynaptic and postsynaptic apparatus, are probably functional. Altogether, these results show that Sgg has an inhibitory role in expanding the NMJ but may not play an important role in bouton differentiation. Presynaptic Sgg controls the NMJ growth Anti-Sgg immunostaining as well as Sgg::GFP fluorescence were concentrated in the presynaptic side, suggesting a role of the presynaptic Sgg in the phenotypes described above. To examine the function of presynaptic Sgg, we used a dominant-negative version of the Sgg10 isoform (Bourouis, 2002). The mutation introduced in the coding sequence is the substitution A81T leading to a kinase-null enzyme. We overexpressed this transgene (sgg DN ) under the control of different presynaptic (or postsynaptic) Gal4 drivers and quantified the bouton number. For presynaptic expression, we used the panneuronal elav-gal4 driver as well as the BG380-Gal4 and OK6-Gal4 drivers, which have patterns of expression more restricted to motoneurons. For postsynaptic expression, we used the MHC-Gal4 driver, which drives expression in all body-wall muscles, as checked with UAS-mCD8GFP (data not shown). In all genotypes where Sgg DN is expressed presynaptically, we observed a % increase in the number of synaptic boutons (Fig. 3A C) ( for CS; for BG380-Gal4; for elav-gal4; for OK6-Gal4). Interestingly, the number of boutons did not change when the dominant-negative construct was expressed postsynaptically (Fig. 3C) ( for MHC-Gal4). The results were similar when we considered the satellite boutons: presynaptic inhibition of Sgg led to a twofold to threefold increase in the percentage of satellite boutons ( for CS; for BG380-Gal4; for elav-gal4; for OK6-Gal4), whereas postsynaptic inhibition of Sgg led to no changes (Fig. 3C) ( for MHC-Gal4). Again, immunostainings against the presynaptic markers CSP, Synapsin, and the postsynaptic marker DLG revealed that all boutons were immunoreactive for these markers, suggesting that they are all functional (data not shown). Altogether, our
4 6576 J. Neurosci., July 21, (29): Franco et al. Shaggy Negatively Controls NMJ Growth Figure 3. Overexpression of a dominant-negative Sgg with different Gal4 drivers. A, B, Illustration of the overgrowth phenotype when Sgg DN is expressed presynaptically: anti-hrp staining of a muscle 6/7 NMJ of a control female larva (CS/UAS-sgg DN )(A) or a female overexpressing Sgg DN presynaptically (OK6-Gal4/UAS-sgg DN )(B). Arrowheads indicate satellite boutons. Scale bar, 11 m. C, Quantification of the total bouton number and the percentage of satellite boutons of control and Sgg DN overexpressing flies. n 25, 26, 18, 31, and 14 for crosses with CS, MHC-Gal4, BG380-Gal4, elav-gal4, and OK6-Gal4, respectively. **p 0.01; ***p results show that presynaptic but not postsynaptic kinase activity of Sgg is required for normal synaptic growth. Sgg controls the microtubule cytoskeleton dynamics One feature of the Sgg inhibition phenotype was the increase in the percentage of satellite boutons. No such increase is found in other mutants displaying an NMJ overgrowth phenotype, like the eag Sh hyperactive mutants (Budnik et al., 1990; Torroja et al., 1999). Satellite boutons can be considered potential ramifications because they are not in line with the other boutons. Ramifications of the NMJ have recently been shown to occur when the microtubule cytoskeleton forms a loop within a bouton (Roos et al., 2000). We wondered whether the increase in satellite boutons observed in sgg loss-of-function conditions was related to modifications in the microtubule cytoskeleton dynamics. We therefore performed stainings directed against the Drosophila MAP1B homolog, Futsch, and quantified the number of microtubule loops when the dominant-negative form of Sgg was expressed in the motoneurons or in the muscle. There were effectively more loops when the percentage of satellite boutons increased in larvae expressing Sgg DN presynaptically (Fig. 4A C) ( for CS; for BG380-Gal4; for elav-gal4; for OK6-Gal4). In larvae expressing Sgg DN postsynaptically, the amount of microtubule loops ( for MHC-Gal4), like the percentage of satellite boutons, remained unchanged. In Figure 4B, we show that many loops were localized in satellite boutons (arrows). Altogether, these results show that Sgg controls the dynamics of the microtubule cytoskeleton, such that an inhibition of the kinase leads to an increase in the number of microtubule loops, which is possibly the cause of the increase in the amount of satellite boutons. Futsch is downstream of Shaggy To find out whether microtubule effectors are actually downstream of shaggy, we performed epistasis experiments. We used two futsch hypomorph loss-of-function mutations, futsch N94 and futsch K68, which respectively display a partially or strongly disorganized microtubule cytoskeleton. Moreover, microtubule loop Figure 4. Effect of Sgg DN overexpression on microtubule dynamics and genetic interaction with futsch. A, B, Illustration of the increase in the number of microtubule loops when Sgg DN is expressed presynaptically: anti-futsch staining of a muscle 13 NMJ on a control female larva (CS/UAS-sgg DN )(A) or a female overexpressing Sgg DN presynaptically (OK6-Gal4/UAS-sgg DN ) ( B). Arrows indicate loops within satellite boutons. Scale bar, 10 m. C, Quantification of the number of microtubule loops in control larvae versus larvae expressing the Sgg DN protein. n 15, 9, 9, 15, and 6 for UAS-sgg DN crosses with CS, MHC-Gal4, BG380-Gal4, elav-gal4, and OK6- Gal4. **p 0.01; ***p D, Genetic interaction between Sgg DN expressed presynaptically and futsch N94 or futsch K68. Quantification of boutons number and percentage of satellite boutons of larvae expressing Sgg DN in the presynapse in a wild-type, futsch N94, and futsch K68 genetic background. n 29 for yw CS /Y, 22 for futsch N94 /Y and futsch K68 /Y, 28 for yw CS /Y;OK6/ ;sgg DN /, 30forfutsch N94 /Y;OK6/ ;sgg DN /, and 22 for futsch K68 /Y;OK6/ ;sgg DN /. ***p for statistical significance versus yw CS /Y; OK6/ ; sgg DN / ; # p 0.05; ## p 0.01; ### p for statistical significance versus yw CS /Y. formation is absent in both mutants (Roos et al., 2000). We verified that the two futsch mutants displayed a 20% decreased number of synaptic boutons compared with CS larvae, as shown by Roos et al. (2000) (Fig. 4D) ( for yw CS ; for futsch N94 ; for futsch K68 ). We also detected a significant decrease in the percentage of satellite boutons in these mutants (Fig. 4D) ( for yw CS ; for futsch N94 ; for futsch K68 ). These phenotypes are opposite of those observed in sgg loss-of-function conditions. Hence, if the NMJ overgrowth observed in sgg mutants requires intact microtubule effectors, we should not see this phenotype when the microtubule cytoskeleton is affected. To test this hypothesis, we presynaptically overexpressed the dominant-negative form of Sgg in wild-type or futsch mutant background (see Materials and Methods). Although NMJs of larvae expressing Sgg DN presynaptically in a wild-type background presented a 30% increase of bouton number ( ), NMJs of larvae expressing Sgg DN in either futsch N94 or futsch K68 background did not display any increase in bouton number ( and , respectively) but displayed the futsch phenotype (Fig. 4D). The situation was similar when we looked at the satellite bouton phenotype (Fig. 4D). Larvae expressing Sgg DN in the futsch K68 background displayed the futsch K68 phenotype ( ). Interestingly, the phenotype was intermediate in the futsch N94 background ( ). This
5 Franco et al. Shaggy Negatively Controls NMJ Growth J. Neurosci., July 21, (29): may be attributable to the remaining Futsch protein detected in these mutants. We also performed Futsch immunostaining of NMJs of futsch N94 larvae either expressing or not expressing the Sgg DN transgene. In both cases, Futsch protein was diffusely localized in the synaptic boutons, indicating that the inhibition of Sgg does not affect the localization of this mutant Futsch protein (data not shown). Altogether, our data demonstrate that the NMJ overgrowth observed when Sgg is inhibited presynaptically requires the microtubule-associated protein Futsch. Discussion The gene sgg is known to play an important role in embryo morphogenesis and cell fate determination (Ruel et al., 1993). Here, our screen let us find that the kinase encoded by this gene was highly concentrated at larval motoneuron terminals. This suggested a role for this protein in these differentiated neurons. In accordance with this hypothesis, we showed, using the mutant sgg K22 and a dominant-negative construct of Sgg, that this kinase negatively controlled the NMJ growth during the larval stages. These data are the first to reveal a role of this kinase in the growth of a differentiated and functional synapse. Using different presynaptic and postsynaptic markers, we could not detect any obvious defect in synapse differentiation. However, the sgg K22 allele is not a null allele. Thus, we cannot exclude that shaggy plays other roles than growth control at the NMJ. The function of GSK-3 in neuronal development, and notably in synapse differentiation had been studied previously on neuronal cultures using lithium chloride (LiCl) as a GSK-3 inhibitor (Lucas and Salinas, 1997; Hall et al., 2000). These authors observed more synapsin-positive clusters along the axons in the presence of LiCl (Hall et al., 2000). This last observation was interpreted as an increase in accumulation of synapsin at the synapses (i.e., a modification in the differentiation of synapses). However, we can interpret this result as an increase in the number of synapses. This in vitro result would then be in accordance with our in vivo data at the Drosophila NMJ, where inhibition of Shaggy increases the number of synaptic boutons. Targeted expression of Sgg DN showed that the function of Shaggy on the growth of the NMJ was required presynaptically and that it requires the kinase function of Sgg. Some major presynaptic targets of GSK-3 known in vertebrates are microtubule-associated proteins like tau, MAP1B, and MAP2 (Cohen and Frame, 2001). Here, we show that when we inhibit Shaggy in the motoneuron, there is an increase in the number of microtubule loops. This change in the dynamics of the microtubule skeleton suggests that some microtubule-associated proteins like the Drosophila tau homolog or the MAP1B homolog, Futsch, may also be substrates of Shaggy. Of interest, we could identify consensus sites for GSK-3 like phosphorylation [SXXX(ST)(PR)XXS] in the sequence of these proteins (96 for Futsch; 1 for tau) but do not have evidence yet that these proteins are phosphorylated by Shaggy. Recently, an effect of LiCl treatment was observed on axonal transport defects resulting from human Tau overexpression in Drosophila larvae, suggesting a functional interaction between Shaggy and Tau (Mudher et al., 2004). Here, we demonstrated that the protein Futsch was required for the overgrowth phenotype observed in sgg loss-of-function conditions. How is the activity of Sgg controlled at the NMJ? The kinase Sgg is known to be inhibited in both the insulin and the WNT/ Wingless (Wg) signaling pathways (Cohen and Frame, 2001). There is little evidence of the presence of insulin-like peptides or insulin receptor-like proteins at the NMJ in Drosophila (Gorczyca et al., 1993). Wg has been shown to be released by larval motoneurons in Drosophila and to control the NMJ growth (Gramates and Budnik, 1999; Packard et al., 2002). The control of Sgg activity via different signaling pathways at the NMJ is still an open question. References Bourouis M (2002) Targeted increase in shaggy activity levels blocks wingless signaling. Genesis 34: Broadie KS, Richmond JE (2002) Establishing and sculpting the synapse in Drosophila and C. elegans. Curr Opin Neurobiol 12: Budnik V, Zhong Y, Wu CF (1990) Morphological plasticity of motor axons in Drosophila mutants with altered excitability. J Neurosci 10: Cohen P, Frame S (2001) The renaissance of GSK3. Nat Rev Mol Cell Biol 2: Gorczyca M, Augart C, Budnik V (1993) Insulin-like receptor and insulinlike peptide are localized at neuromuscular junctions in Drosophila. J Neurosci 13: Gramates LS, Budnik V (1999) Assembly and maturation of the Drosophila larval neuromuscular junction. Int Rev Neurobiol 43: Hall AC, Lucas FR, Salinas PC (2000) Axonal remodeling and synaptic differentiation in the cerebellum is regulated by WNT-7a signaling. Cell 100: Klagges BR, Heimbeck G, Godenschwege TA, Hofbauer A, Pflugfelder GO, Reifegerste R, Reisch D, Schaupp M, Buchner S, Buchner E (1996) Invertebrate synapsins: a single gene codes for several isoforms in Drosophila. J Neurosci 16: Lahey T, Gorczyca M, Jia XX, Budnik V (1994) The Drosophila tumor suppressor gene dlg is required for normal synaptic bouton structure. Neuron 13: Lnenicka GA, Keshishian H (2000) Identified motor terminals in Drosophila larvae show distinct differences in morphology and physiology. J Neurobiol 43: Lucas FR, Salinas PC (1997) WNT-7a induces axonal remodeling and increases synapsin I levels in cerebellar neurons. Dev Biol 192: Morin X, Daneman R, Zavortink M, Chia W (2001) A protein trap strategy to detect GFP-tagged proteins expressed from their endogenous loci in Drosophila. Proc Natl Acad Sci USA 98: Mudher A, Shepherd D, Newman TA, Mildren P, Jukes JP, Squire A, Mears A, Berg S, MacKay D, Asuni AA, Bhat R, Lovestone S (2004) GSK-3beta inhibition reverses axonal transport defects and behavioural phenotypes in Drosophila. Mol Psychiatry 9: Muller D, Nikonenko I, Jourdain P, Alberi S (2002) LTP, memory and structural plasticity. Curr Mol Med 2: Packard M, Koo ES, Gorczyca M, Sharpe J, Cumberledge S, Budnik V (2002) The Drosophila Wnt, wingless, provides an essential signal for pre- and postsynaptic differentiation. Cell 111: Roos J, Hummel T, Ng N, Klambt C, Davis GW (2000) Drosophila Futsch regulates synaptic microtubule organization and is necessary for synaptic growth. Neuron 26: Ruel L, Pantesco V, Lutz Y, Simpson P, Bourouis M (1993) Functional significance of a family of protein kinases encoded at the shaggy locus in Drosophila. EMBO J 12: Schuster CM, Davis GW, Fetter RD, Goodman CS (1996a) Genetic dissection of structural and functional components of synaptic plasticity. I. Fasciclin II controls synaptic stabilization and growth. Neuron 17: Schuster CM, Davis GW, Fetter RD, Goodman CS (1996b) Genetic dissection of structural and functional components of synaptic plasticity. II. Fasciclin II controls presynaptic structural plasticity. Neuron 17: Torroja L, Packard M, Gorczyca M, White K, Budnik V (1999) The Drosophila beta-amyloid precursor protein homolog promotes synapse differentiation at the neuromuscular junction. J Neurosci 19:
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10923 Supplementary Figure 1 Ten-a and Ten-m antibody and cell type specificities. a c, Representative single confocal sections of a Drosophila NMJ stained with antibodies to Ten-a (red),
doi:10.1038/nature10923 Supplementary Figure 1 Ten-a and Ten-m antibody and cell type specificities. a c, Representative single confocal sections of a Drosophila NMJ stained with antibodies to Ten-a (red),
Baz, Par-6 and apkc are not required for axon or dendrite specification in Drosophila
 Baz, Par-6 and apkc are not required for axon or dendrite specification in Drosophila Melissa M. Rolls and Chris Q. Doe, Inst. Neurosci and Inst. Mol. Biol., HHMI, Univ. Oregon, Eugene, Oregon 97403 Correspondence
Baz, Par-6 and apkc are not required for axon or dendrite specification in Drosophila Melissa M. Rolls and Chris Q. Doe, Inst. Neurosci and Inst. Mol. Biol., HHMI, Univ. Oregon, Eugene, Oregon 97403 Correspondence
Chapter 4 Evaluating a potential interaction between deltex and git in Drosophila: genetic interaction, gene overexpression and cell biology assays.
 Evaluating a potential interaction between deltex and git in Drosophila: genetic interaction, gene overexpression and cell biology assays. The data described in chapter 3 presented evidence that endogenous
Evaluating a potential interaction between deltex and git in Drosophila: genetic interaction, gene overexpression and cell biology assays. The data described in chapter 3 presented evidence that endogenous
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/6/301/ra98/dc1 Supplementary Materials for Regulation of Epithelial Morphogenesis by the G Protein Coupled Receptor Mist and Its Ligand Fog Alyssa J. Manning,
www.sciencesignaling.org/cgi/content/full/6/301/ra98/dc1 Supplementary Materials for Regulation of Epithelial Morphogenesis by the G Protein Coupled Receptor Mist and Its Ligand Fog Alyssa J. Manning,
Bypass and interaction suppressors; pathway analysis
 Bypass and interaction suppressors; pathway analysis The isolation of extragenic suppressors is a powerful tool for identifying genes that encode proteins that function in the same process as a gene of
Bypass and interaction suppressors; pathway analysis The isolation of extragenic suppressors is a powerful tool for identifying genes that encode proteins that function in the same process as a gene of
Supplementary Figures Supplementary Tables 1 and 2. Supplementary References
 The Nuclear Import of a Non-Canonical Frizzled2 Signal by Importins-β11 and α2 Promotes Postsynaptic Development Timothy J. Mosca and Thomas L. Schwarz * The F.M. Kirby Neurobiology Center, Children s
The Nuclear Import of a Non-Canonical Frizzled2 Signal by Importins-β11 and α2 Promotes Postsynaptic Development Timothy J. Mosca and Thomas L. Schwarz * The F.M. Kirby Neurobiology Center, Children s
Drosophila Spastin Regulates Synaptic Microtubule Networks and Is Required for Normal Motor Function
 Drosophila Spastin Regulates Synaptic Microtubule Networks and Is Required for Normal Motor Function Nina Tang Sherwood 1, Qi Sun 1 1, Mingshan Xue 2 2, Bing Zhang 2, Kai Zinn 1 * Open access, freely available
Drosophila Spastin Regulates Synaptic Microtubule Networks and Is Required for Normal Motor Function Nina Tang Sherwood 1, Qi Sun 1 1, Mingshan Xue 2 2, Bing Zhang 2, Kai Zinn 1 * Open access, freely available
T H E J O U R N A L O F C E L L B I O L O G Y
 Supplemental material Kasprowicz et al., http://www.jcb.org/cgi/content/full/jcb.201310090/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. NMJ morphology of shi 12-12B ; shi-4c not treated
Supplemental material Kasprowicz et al., http://www.jcb.org/cgi/content/full/jcb.201310090/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. NMJ morphology of shi 12-12B ; shi-4c not treated
A role for PS integrins in morphological growth and synaptic function at the postembryonic neuromuscular junction of Drosophila
 Development 126, 5833-5846 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV8655 5833 A role for PS integrins in morphological growth and synaptic function at the postembryonic
Development 126, 5833-5846 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV8655 5833 A role for PS integrins in morphological growth and synaptic function at the postembryonic
Synaptic Homeostasis Is Consolidated by the Cell Fate Gene gooseberry,adrosophila pax3/7 Homolog
 The Journal of Neuroscience, June 16, 2010 30(24):8071 8082 8071 Development/Plasticity/Repair Synaptic Homeostasis Is Consolidated by the Cell Fate Gene gooseberry,adrosophila pax3/7 Homolog Bruno Marie,
The Journal of Neuroscience, June 16, 2010 30(24):8071 8082 8071 Development/Plasticity/Repair Synaptic Homeostasis Is Consolidated by the Cell Fate Gene gooseberry,adrosophila pax3/7 Homolog Bruno Marie,
The BMP Ligand Gbb Gates the Expression of Synaptic Homeostasis Independent of Synaptic Growth Control
 Article The BMP Ligand Gbb Gates the Expression of Synaptic Homeostasis Independent of Synaptic Growth Control Carleton P. Goold 1 and Graeme W. Davis 1, * 1 Department of Biochemistry and Biophysics,
Article The BMP Ligand Gbb Gates the Expression of Synaptic Homeostasis Independent of Synaptic Growth Control Carleton P. Goold 1 and Graeme W. Davis 1, * 1 Department of Biochemistry and Biophysics,
Highwire Regulates Synaptic Growth in Drosophila
 Neuron, Vol. 26, 313 329, May, 2000, Copyright 2000 by Cell Press Highwire Regulates Synaptic Growth in Drosophila Hong I. Wan,* Aaron DiAntonio,* Richard D. Fetter,* Kendra Bergstrom,* Roland Strauss,
Neuron, Vol. 26, 313 329, May, 2000, Copyright 2000 by Cell Press Highwire Regulates Synaptic Growth in Drosophila Hong I. Wan,* Aaron DiAntonio,* Richard D. Fetter,* Kendra Bergstrom,* Roland Strauss,
Anterograde Activin Signaling Regulates Postsynaptic Membrane Potential and GluRIIA/B Abundance at the Drosophila Neuromuscular Junction
 Anterograde Activin Signaling Regulates Postsynaptic Membrane Potential and GluRIIA/B Abundance at the Drosophila Neuromuscular Junction Myung-Jun Kim, Michael B. O Connor* Department of Genetics, Cell
Anterograde Activin Signaling Regulates Postsynaptic Membrane Potential and GluRIIA/B Abundance at the Drosophila Neuromuscular Junction Myung-Jun Kim, Michael B. O Connor* Department of Genetics, Cell
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3267 Supplementary Figure 1 A group of genes required for formation or orientation of annular F-actin bundles and aecm ridges: RNAi phenotypes and their validation by standard mutations.
DOI: 10.1038/ncb3267 Supplementary Figure 1 A group of genes required for formation or orientation of annular F-actin bundles and aecm ridges: RNAi phenotypes and their validation by standard mutations.
Cells. Steven McLoon Department of Neuroscience University of Minnesota
 Cells Steven McLoon Department of Neuroscience University of Minnesota 1 Microscopy Methods of histology: Treat the tissue with a preservative (e.g. formaldehyde). Dissect the region of interest. Embed
Cells Steven McLoon Department of Neuroscience University of Minnesota 1 Microscopy Methods of histology: Treat the tissue with a preservative (e.g. formaldehyde). Dissect the region of interest. Embed
Drosophila VAP-33A Directs Bouton Formation at Neuromuscular Junctions in a Dosage-Dependent Manner
 Neuron, Vol. 35, 291 306, July 18, 2002, Copyright 2002 by Cell Press Drosophila VAP-33A Directs Bouton Formation at Neuromuscular Junctions in a Dosage-Dependent Manner Giuseppa Pennetta, 1,2 Peter Robin
Neuron, Vol. 35, 291 306, July 18, 2002, Copyright 2002 by Cell Press Drosophila VAP-33A Directs Bouton Formation at Neuromuscular Junctions in a Dosage-Dependent Manner Giuseppa Pennetta, 1,2 Peter Robin
Midterm 1. Average score: 74.4 Median score: 77
 Midterm 1 Average score: 74.4 Median score: 77 NAME: TA (circle one) Jody Westbrook or Jessica Piel Section (circle one) Tue Wed Thur MCB 141 First Midterm Feb. 21, 2008 Only answer 4 of these 5 problems.
Midterm 1 Average score: 74.4 Median score: 77 NAME: TA (circle one) Jody Westbrook or Jessica Piel Section (circle one) Tue Wed Thur MCB 141 First Midterm Feb. 21, 2008 Only answer 4 of these 5 problems.
Nature Biotechnology: doi: /nbt Supplementary Figure 1. Overexpression of YFP::GPR-1 in the germline.
 Supplementary Figure 1 Overexpression of YFP::GPR-1 in the germline. The pie-1 promoter and 3 utr were used to express yfp::gpr-1 in the germline. Expression levels from the yfp::gpr-1(cai 1.0)-expressing
Supplementary Figure 1 Overexpression of YFP::GPR-1 in the germline. The pie-1 promoter and 3 utr were used to express yfp::gpr-1 in the germline. Expression levels from the yfp::gpr-1(cai 1.0)-expressing
Dimorphism and Homeostasis of Neuromuscular Synaptic Function
 Dimorphism and Homeostasis of Neuromuscular Synaptic Function - Epp m ε Release + + φ - q Sensitivity The nature of the problem Size, Strength, Resistance.. The Power of Drosophila The nature of the problem
Dimorphism and Homeostasis of Neuromuscular Synaptic Function - Epp m ε Release + + φ - q Sensitivity The nature of the problem Size, Strength, Resistance.. The Power of Drosophila The nature of the problem
"Small Brains, Big Ideas: The value of model organisms to science. Dr. Yuly Fuentes-Medel Executive Director Descience
 "Small Brains, Big Ideas: The value of model organisms to science Dr. Yuly Fuentes-Medel Executive Director Descience Outline 1. What is a model organism? 2. Big questions solved by small-brained organisms
"Small Brains, Big Ideas: The value of model organisms to science Dr. Yuly Fuentes-Medel Executive Director Descience Outline 1. What is a model organism? 2. Big questions solved by small-brained organisms
Supporting Information
 Supporting Information Cao et al. 10.1073/pnas.1306220110 Gram - bacteria Hemolymph Cytoplasm PGRP-LC TAK1 signalosome Imd dfadd Dredd Dnr1 Ikk signalosome P Relish Nucleus AMP and effector genes Fig.
Supporting Information Cao et al. 10.1073/pnas.1306220110 Gram - bacteria Hemolymph Cytoplasm PGRP-LC TAK1 signalosome Imd dfadd Dredd Dnr1 Ikk signalosome P Relish Nucleus AMP and effector genes Fig.
Cell Death & Trophic Factors II. Steven McLoon Department of Neuroscience University of Minnesota
 Cell Death & Trophic Factors II Steven McLoon Department of Neuroscience University of Minnesota 1 Remember? Neurotrophins are cell survival factors that neurons get from their target cells! There is a
Cell Death & Trophic Factors II Steven McLoon Department of Neuroscience University of Minnesota 1 Remember? Neurotrophins are cell survival factors that neurons get from their target cells! There is a
Nature Neuroscience: doi: /nn.2662
 Supplementary Figure 1 Atlastin phylogeny and homology. (a) Maximum likelihood phylogenetic tree based on 18 Atlastin-1 sequences using the program Quicktree. Numbers at internal nodes correspond to bootstrap
Supplementary Figure 1 Atlastin phylogeny and homology. (a) Maximum likelihood phylogenetic tree based on 18 Atlastin-1 sequences using the program Quicktree. Numbers at internal nodes correspond to bootstrap
Open Syntaxin Docks Synaptic Vesicles
 Marc Hammarlund 1,2[, Mark T. Palfreyman 1,2[, Shigeki Watanabe 1,2[, Shawn Olsen 1,2, Erik M. Jorgensen 1,2* PLoS BIOLOGY 1 Department of Biology, University of Utah, Salt Lake City, Utah, United States
Marc Hammarlund 1,2[, Mark T. Palfreyman 1,2[, Shigeki Watanabe 1,2[, Shawn Olsen 1,2, Erik M. Jorgensen 1,2* PLoS BIOLOGY 1 Department of Biology, University of Utah, Salt Lake City, Utah, United States
A complementation test would be done by crossing the haploid strains and scoring the phenotype in the diploids.
 Problem set H answers 1. To study DNA repair mechanisms, geneticists isolated yeast mutants that were sensitive to various types of radiation; for example, mutants that were more sensitive to UV light.
Problem set H answers 1. To study DNA repair mechanisms, geneticists isolated yeast mutants that were sensitive to various types of radiation; for example, mutants that were more sensitive to UV light.
Supplementary Information
 Supplementary Information Supplementary Figure 1. JAK/STAT in early wing development (a-f) Wing primordia of second instar larvae of the indicated genotypes labeled to visualize expression of upd mrna
Supplementary Information Supplementary Figure 1. JAK/STAT in early wing development (a-f) Wing primordia of second instar larvae of the indicated genotypes labeled to visualize expression of upd mrna
Integrins regulate DLG/FAS2 via a CaM kinase II-dependent pathway to mediate synapse elaboration and stabilization during postembryonic development
 Development 129, 3381-3391 (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV9843 3381 Integrins regulate DLG/FAS2 via a CaM kinase II-dependent pathway to mediate synapse elaboration
Development 129, 3381-3391 (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV9843 3381 Integrins regulate DLG/FAS2 via a CaM kinase II-dependent pathway to mediate synapse elaboration
Synaptic abnormalities in a Drosophila model of Alzheimer s disease
 2014. Published by The Company of Biologists Ltd (2014) 7, 373-385 doi:10.1242/dmm.012104 RESEARCH ARTICLE Synaptic abnormalities in a Drosophila model of Alzheimer s disease Siddhita D. Mhatre 1, Vivek
2014. Published by The Company of Biologists Ltd (2014) 7, 373-385 doi:10.1242/dmm.012104 RESEARCH ARTICLE Synaptic abnormalities in a Drosophila model of Alzheimer s disease Siddhita D. Mhatre 1, Vivek
Developmental Biology
 Developmental Biology 366 (2012) 204 217 Contents lists available at SciVerse ScienceDirect Developmental Biology journal homepage: www.elsevier.com/locate/developmentalbiology Developmental changes in
Developmental Biology 366 (2012) 204 217 Contents lists available at SciVerse ScienceDirect Developmental Biology journal homepage: www.elsevier.com/locate/developmentalbiology Developmental changes in
Supplementary Figure 1. Nature Genetics: doi: /ng.3848
 Supplementary Figure 1 Phenotypes and epigenetic properties of Fab2L flies. A- Phenotypic classification based on eye pigment levels in Fab2L male (orange bars) and female (yellow bars) flies (n>150).
Supplementary Figure 1 Phenotypes and epigenetic properties of Fab2L flies. A- Phenotypic classification based on eye pigment levels in Fab2L male (orange bars) and female (yellow bars) flies (n>150).
Single-Cell Analysis of Drosophila Larval Neuromuscular Synapses
 Developmental Biology 229, 55 70 (2001) doi:10.1006/dbio.2000.9983, available online at http://www.idealibrary.com on Single-Cell Analysis of Drosophila Larval Neuromuscular Synapses Bao Hoang 1 and Akira
Developmental Biology 229, 55 70 (2001) doi:10.1006/dbio.2000.9983, available online at http://www.idealibrary.com on Single-Cell Analysis of Drosophila Larval Neuromuscular Synapses Bao Hoang 1 and Akira
Monitoring neurite morphology and synapse formation in primary neurons for neurotoxicity assessments and drug screening
 APPLICATION NOTE ArrayScan High Content Platform Monitoring neurite morphology and synapse formation in primary neurons for neurotoxicity assessments and drug screening Suk J. Hong and Richik N. Ghosh
APPLICATION NOTE ArrayScan High Content Platform Monitoring neurite morphology and synapse formation in primary neurons for neurotoxicity assessments and drug screening Suk J. Hong and Richik N. Ghosh
Segment boundary formation in Drosophila embryos
 Segment boundary formation in Drosophila embryos Development 130, August 2003 Camilla W. Larsen, Elizabeth Hirst, Cyrille Alexandre and Jean Paul Vincent 1. Introduction: - Segment boundary formation:
Segment boundary formation in Drosophila embryos Development 130, August 2003 Camilla W. Larsen, Elizabeth Hirst, Cyrille Alexandre and Jean Paul Vincent 1. Introduction: - Segment boundary formation:
Overexpression of Cysteine-String Proteins in Drosophila Reveals Interactions with Syntaxin
 The Journal of Neuroscience, December 1, 1999, 19(23):10270 10279 Overexpression of Cysteine-String Proteins in Drosophila Reveals Interactions with Syntaxin Zhiping Nie, Ravi Ranjan, Julia J. Wenniger,
The Journal of Neuroscience, December 1, 1999, 19(23):10270 10279 Overexpression of Cysteine-String Proteins in Drosophila Reveals Interactions with Syntaxin Zhiping Nie, Ravi Ranjan, Julia J. Wenniger,
Modulation of central pattern generator output by peripheral sensory cells in Drosophila larvae. BioNB4910 Cornell University.
 Modulation of central pattern generator output by peripheral sensory cells in Drosophila larvae BioNB4910 Cornell University Goals 1) Observe the behavioral effects of remotely activating different populations
Modulation of central pattern generator output by peripheral sensory cells in Drosophila larvae BioNB4910 Cornell University Goals 1) Observe the behavioral effects of remotely activating different populations
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature12791 Supplementary Figure 1 (1/3) WWW.NATURE.COM/NATURE 1 RESEARCH SUPPLEMENTARY INFORMATION Supplementary Figure 1 (2/3) 2 WWW.NATURE.COM/NATURE SUPPLEMENTARY
SUPPLEMENTARY INFORMATION doi:10.1038/nature12791 Supplementary Figure 1 (1/3) WWW.NATURE.COM/NATURE 1 RESEARCH SUPPLEMENTARY INFORMATION Supplementary Figure 1 (2/3) 2 WWW.NATURE.COM/NATURE SUPPLEMENTARY
Supplementary Figure 1. Markedly decreased numbers of marginal zone B cells in DOCK8 mutant mice Supplementary Figure 2.
 Supplementary Figure 1. Markedly decreased numbers of marginal zone B cells in DOCK8 mutant mice. Percentage of marginal zone B cells in the spleen of wild-type mice (+/+), mice homozygous for cpm or pri
Supplementary Figure 1. Markedly decreased numbers of marginal zone B cells in DOCK8 mutant mice. Percentage of marginal zone B cells in the spleen of wild-type mice (+/+), mice homozygous for cpm or pri
1. Draw, label and describe the structure of DNA and RNA including bonding mechanisms.
 Practicing Biology BIG IDEA 3.A 1. Draw, label and describe the structure of DNA and RNA including bonding mechanisms. 2. Using at least 2 well-known experiments, describe which features of DNA and RNA
Practicing Biology BIG IDEA 3.A 1. Draw, label and describe the structure of DNA and RNA including bonding mechanisms. 2. Using at least 2 well-known experiments, describe which features of DNA and RNA
Reading: Chapter 5, pp ; Reference chapter D, pp Problem set F
 Mosaic Analysis Reading: Chapter 5, pp140-141; Reference chapter D, pp820-823 Problem set F Twin spots in Drosophila Although segregation and recombination in mitosis do not occur at the same frequency
Mosaic Analysis Reading: Chapter 5, pp140-141; Reference chapter D, pp820-823 Problem set F Twin spots in Drosophila Although segregation and recombination in mitosis do not occur at the same frequency
SUPPLEMENTARY INFORMATION
 Supplementary Figure 1 Sns and Duf co-localise in embryonic nephrocytes a-c, Wild-type stage 11 (a),14 (b) and 16 (c) embryos stained with anti-duf (green) and anti-sns (red). Higher magnification images
Supplementary Figure 1 Sns and Duf co-localise in embryonic nephrocytes a-c, Wild-type stage 11 (a),14 (b) and 16 (c) embryos stained with anti-duf (green) and anti-sns (red). Higher magnification images
Chapter 11. Development: Differentiation and Determination
 KAP Biology Dept Kenyon College Differential gene expression and development Mechanisms of cellular determination Induction Pattern formation Chapter 11. Development: Differentiation and Determination
KAP Biology Dept Kenyon College Differential gene expression and development Mechanisms of cellular determination Induction Pattern formation Chapter 11. Development: Differentiation and Determination
Cell biology: Death drags down the neighbourhood
 Cell biology: Death drags down the neighbourhood The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published Publisher Vasquez,
Cell biology: Death drags down the neighbourhood The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published Publisher Vasquez,
SUPPLEMENTARY INFORMATION
 med!1,2 Wild-type (N2) end!3 elt!2 5 1 15 Time (minutes) 5 1 15 Time (minutes) med!1,2 end!3 5 1 15 Time (minutes) elt!2 5 1 15 Time (minutes) Supplementary Figure 1: Number of med-1,2, end-3, end-1 and
med!1,2 Wild-type (N2) end!3 elt!2 5 1 15 Time (minutes) 5 1 15 Time (minutes) med!1,2 end!3 5 1 15 Time (minutes) elt!2 5 1 15 Time (minutes) Supplementary Figure 1: Number of med-1,2, end-3, end-1 and
Illegitimate translation causes unexpected gene expression from on-target out-of-frame alleles
 Illegitimate translation causes unexpected gene expression from on-target out-of-frame alleles created by CRISPR-Cas9 Shigeru Makino, Ryutaro Fukumura, Yoichi Gondo* Mutagenesis and Genomics Team, RIKEN
Illegitimate translation causes unexpected gene expression from on-target out-of-frame alleles created by CRISPR-Cas9 Shigeru Makino, Ryutaro Fukumura, Yoichi Gondo* Mutagenesis and Genomics Team, RIKEN
BIS &003 Answers to Assigned Problems May 23, Week /18.6 How would you distinguish between an enhancer and a promoter?
 Week 9 Study Questions from the textbook: 6 th Edition: Chapter 19-19.6, 19.7, 19.15, 19.17 OR 7 th Edition: Chapter 18-18.6 18.7, 18.15, 18.17 19.6/18.6 How would you distinguish between an enhancer and
Week 9 Study Questions from the textbook: 6 th Edition: Chapter 19-19.6, 19.7, 19.15, 19.17 OR 7 th Edition: Chapter 18-18.6 18.7, 18.15, 18.17 19.6/18.6 How would you distinguish between an enhancer and
13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins
 13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins Molecular sorting: specific budding, vesicular transport, fusion 1. Why is this important? A. Form and
13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins Molecular sorting: specific budding, vesicular transport, fusion 1. Why is this important? A. Form and
The Molecular Mechanisms of Activity-Dependent Wingless (Wg)/Wnt Signaling at a Drosophila Glutamatergic Synapse: a Dissertation
 University of Massachusetts Medical School escholarship@umms GSBS Dissertations and Theses Graduate School of Biomedical Sciences 2-1-2008 The Molecular Mechanisms of Activity-Dependent Wingless (Wg)/Wnt
University of Massachusetts Medical School escholarship@umms GSBS Dissertations and Theses Graduate School of Biomedical Sciences 2-1-2008 The Molecular Mechanisms of Activity-Dependent Wingless (Wg)/Wnt
Nervous Systems: Neuron Structure and Function
 Nervous Systems: Neuron Structure and Function Integration An animal needs to function like a coherent organism, not like a loose collection of cells. Integration = refers to processes such as summation
Nervous Systems: Neuron Structure and Function Integration An animal needs to function like a coherent organism, not like a loose collection of cells. Integration = refers to processes such as summation
Solutions to Problem Set 4
 Question 1 Solutions to 7.014 Problem Set 4 Because you have not read much scientific literature, you decide to study the genetics of garden peas. You have two pure breeding pea strains. One that is tall
Question 1 Solutions to 7.014 Problem Set 4 Because you have not read much scientific literature, you decide to study the genetics of garden peas. You have two pure breeding pea strains. One that is tall
Time allowed: 2 hours Answer ALL questions in Section A, ALL PARTS of the question in Section B and ONE question from Section C.
 UNIVERSITY OF EAST ANGLIA School of Biological Sciences Main Series UG Examination 2017-2018 GENETICS BIO-5009A Time allowed: 2 hours Answer ALL questions in Section A, ALL PARTS of the question in Section
UNIVERSITY OF EAST ANGLIA School of Biological Sciences Main Series UG Examination 2017-2018 GENETICS BIO-5009A Time allowed: 2 hours Answer ALL questions in Section A, ALL PARTS of the question in Section
4) Please cite Dagda et al J Biol Chem 284: , for any publications or presentations resulting from use or modification of the macro.
 Supplement Figure S1. Algorithmic quantification of mitochondrial morphology in SH- SY5Y cells treated with known fission/fusion mediators. Parental SH-SY5Y cells were transiently transfected with an empty
Supplement Figure S1. Algorithmic quantification of mitochondrial morphology in SH- SY5Y cells treated with known fission/fusion mediators. Parental SH-SY5Y cells were transiently transfected with an empty
Role of Neural Activity during Synaptogenesis in Drosophila
 The Journal of Neuroscience, December 1995, 15( 12): 8177-8190 Role of Neural Activity during Synaptogenesis in Drosophila Jill Jareckil and Haig Keshishian* Departments of Genetics and 2Biology, Yale
The Journal of Neuroscience, December 1995, 15( 12): 8177-8190 Role of Neural Activity during Synaptogenesis in Drosophila Jill Jareckil and Haig Keshishian* Departments of Genetics and 2Biology, Yale
Lecture 2: mrna localisation and decay in cells and in tissues
 Lecture 2: mrna localisation and decay in cells and in tissues Prof. Ilan Davis, Department of Biochemistry. Senior Research Fellow, Jesus College ilan.davis@bioch.ox.ac.uk http://www.ilandavis.com References
Lecture 2: mrna localisation and decay in cells and in tissues Prof. Ilan Davis, Department of Biochemistry. Senior Research Fellow, Jesus College ilan.davis@bioch.ox.ac.uk http://www.ilandavis.com References
7.06 Problem Set #4, Spring 2005
 7.06 Problem Set #4, Spring 2005 1. You re doing a mutant hunt in S. cerevisiae (budding yeast), looking for temperaturesensitive mutants that are defective in the cell cycle. You discover a mutant strain
7.06 Problem Set #4, Spring 2005 1. You re doing a mutant hunt in S. cerevisiae (budding yeast), looking for temperaturesensitive mutants that are defective in the cell cycle. You discover a mutant strain
Chromosome Chr Duplica Duplic t a ion Pixley
 Chromosome Duplication Pixley Figure 4-6 Molecular Biology of the Cell ( Garland Science 2008) Figure 4-72 Molecular Biology of the Cell ( Garland Science 2008) Interphase During mitosis (cell division),
Chromosome Duplication Pixley Figure 4-6 Molecular Biology of the Cell ( Garland Science 2008) Figure 4-72 Molecular Biology of the Cell ( Garland Science 2008) Interphase During mitosis (cell division),
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY METHODS Mef2 primary screen. RNAi hairpins from the VDRC collection were crossed to Mef2-GAL4 at 27 C. After 2 weeks lethality rate and stage was scored, and if possible 20-30 males containing
SUPPLEMENTARY METHODS Mef2 primary screen. RNAi hairpins from the VDRC collection were crossed to Mef2-GAL4 at 27 C. After 2 weeks lethality rate and stage was scored, and if possible 20-30 males containing
Chapter 37 Active Reading Guide Neurons, Synapses, and Signaling
 Name: AP Biology Mr. Croft Section 1 1. What is a neuron? Chapter 37 Active Reading Guide Neurons, Synapses, and Signaling 2. Neurons can be placed into three groups, based on their location and function.
Name: AP Biology Mr. Croft Section 1 1. What is a neuron? Chapter 37 Active Reading Guide Neurons, Synapses, and Signaling 2. Neurons can be placed into three groups, based on their location and function.
SUPPLEMENTARY INFORMATION
 Supplementary Discussion Rationale for using maternal ythdf2 -/- mutants as study subject To study the genetic basis of the embryonic developmental delay that we observed, we crossed fish with different
Supplementary Discussion Rationale for using maternal ythdf2 -/- mutants as study subject To study the genetic basis of the embryonic developmental delay that we observed, we crossed fish with different
Green Fluorescent Protein (GFP) Today s Nobel Prize in Chemistry
 In the news: High-throughput sequencing using Solexa/Illumina technology The copy number of each fetal chromosome can be determined by direct sequencing of DNA in cell-free plasma from pregnant women Confession:
In the news: High-throughput sequencing using Solexa/Illumina technology The copy number of each fetal chromosome can be determined by direct sequencing of DNA in cell-free plasma from pregnant women Confession:
Neurite initiation. Neurite formation begins with a bud that sprouts from the cell body. One or several neurites can sprout at a time.
 Neurite initiation. Neuronal maturation initiation f-actin polarization and maturation tubulin stage 1: "spherical" neuron stage 2: neurons extend several neurites stage 3: one neurite accelerates its
Neurite initiation. Neuronal maturation initiation f-actin polarization and maturation tubulin stage 1: "spherical" neuron stage 2: neurons extend several neurites stage 3: one neurite accelerates its
Mitochondrial Dynamics Is a Distinguishing Feature of Skeletal Muscle Fiber Types and Regulates Organellar Compartmentalization
 Cell Metabolism Supplemental Information Mitochondrial Dynamics Is a Distinguishing Feature of Skeletal Muscle Fiber Types and Regulates Organellar Compartmentalization Prashant Mishra, Grigor Varuzhanyan,
Cell Metabolism Supplemental Information Mitochondrial Dynamics Is a Distinguishing Feature of Skeletal Muscle Fiber Types and Regulates Organellar Compartmentalization Prashant Mishra, Grigor Varuzhanyan,
 DOI: 10.1038/ncb2819 Gαi3 / Actin / Acetylated Tubulin Gαi3 / Actin / Acetylated Tubulin a a Gαi3 a Actin Gαi3 WT Gαi3 WT Gαi3 WT b b Gαi3 b Actin Gαi3 KO Gαi3 KO Gαi3 KO # # Figure S1 Loss of protein
DOI: 10.1038/ncb2819 Gαi3 / Actin / Acetylated Tubulin Gαi3 / Actin / Acetylated Tubulin a a Gαi3 a Actin Gαi3 WT Gαi3 WT Gαi3 WT b b Gαi3 b Actin Gαi3 KO Gαi3 KO Gαi3 KO # # Figure S1 Loss of protein
Neuronal Differentiation: Synapse Formation NMJ
 Neuronal Differentiation: Synapse Formation NMJ Approaches to study synapse formation Rodent NMJ (cholinergic) Drosophila NMJ (glutamatergic) Rodent CNS synapse (glutamatergic or GABAergic) C. elegans
Neuronal Differentiation: Synapse Formation NMJ Approaches to study synapse formation Rodent NMJ (cholinergic) Drosophila NMJ (glutamatergic) Rodent CNS synapse (glutamatergic or GABAergic) C. elegans
5/4/05 Biol 473 lecture
 5/4/05 Biol 473 lecture animals shown: anomalocaris and hallucigenia 1 The Cambrian Explosion - 550 MYA THE BIG BANG OF ANIMAL EVOLUTION Cambrian explosion was characterized by the sudden and roughly simultaneous
5/4/05 Biol 473 lecture animals shown: anomalocaris and hallucigenia 1 The Cambrian Explosion - 550 MYA THE BIG BANG OF ANIMAL EVOLUTION Cambrian explosion was characterized by the sudden and roughly simultaneous
Postsynaptic Rab GTPases and Exocyst: a screen at the Drosophila neuromuscular junction
 Postsynaptic Rab GTPases and Exocyst: a screen at the Drosophila neuromuscular junction Cátia F Patrício-Rodrigues, Rita O Teodoro CEDOC, Nova Medical School, Universidade Nova de Lisboa Correspondence
Postsynaptic Rab GTPases and Exocyst: a screen at the Drosophila neuromuscular junction Cátia F Patrício-Rodrigues, Rita O Teodoro CEDOC, Nova Medical School, Universidade Nova de Lisboa Correspondence
Drosophila melanogaster and D. simulans, two fruit fly species that are nearly
 Comparative Genomics: Human versus chimpanzee 1. Introduction The chimpanzee is the closest living relative to humans. The two species are nearly identical in DNA sequence (>98% identity), yet vastly different
Comparative Genomics: Human versus chimpanzee 1. Introduction The chimpanzee is the closest living relative to humans. The two species are nearly identical in DNA sequence (>98% identity), yet vastly different
18.4 Embryonic development involves cell division, cell differentiation, and morphogenesis
 18.4 Embryonic development involves cell division, cell differentiation, and morphogenesis An organism arises from a fertilized egg cell as the result of three interrelated processes: cell division, cell
18.4 Embryonic development involves cell division, cell differentiation, and morphogenesis An organism arises from a fertilized egg cell as the result of three interrelated processes: cell division, cell
Original Article Overexpression of ter94, Drosophila VCP, improves motor neuron degeneration induced by knockdown of TBPH, Drosophila TDP-43
 Am J Neurodegener Dis 2018;7(1):11-31 www.ajnd.us /ISSN:2165-591X/AJND0069303 Original Article Overexpression of ter94, Drosophila VCP, improves motor neuron degeneration induced by knockdown of TBPH,
Am J Neurodegener Dis 2018;7(1):11-31 www.ajnd.us /ISSN:2165-591X/AJND0069303 Original Article Overexpression of ter94, Drosophila VCP, improves motor neuron degeneration induced by knockdown of TBPH,
THE ability of an animal to adjust to its environment
 Copyright Ó 2008 by the Genetics Society of America DOI: 10.1534/genetics.107.085555 Overexpression Screen in Drosophila Identifies Neuronal Roles of GSK-3b/shaggy as a Regulator of AP-1-Dependent Developmental
Copyright Ó 2008 by the Genetics Society of America DOI: 10.1534/genetics.107.085555 Overexpression Screen in Drosophila Identifies Neuronal Roles of GSK-3b/shaggy as a Regulator of AP-1-Dependent Developmental
Supplementary Materials for
 www.sciencemag.org/cgi/content/full/science.1244624/dc1 Supplementary Materials for Cytoneme-Mediated Contact-Dependent Transport of the Drosophila Decapentaplegic Signaling Protein Sougata Roy, Hai Huang,
www.sciencemag.org/cgi/content/full/science.1244624/dc1 Supplementary Materials for Cytoneme-Mediated Contact-Dependent Transport of the Drosophila Decapentaplegic Signaling Protein Sougata Roy, Hai Huang,
Bio 3411, Fall 2006, Lecture 19-Cell Death.
 Types of Cell Death Questions : Apoptosis (Programmed Cell Death) : Cell-Autonomous Stereotypic Rapid Clean (dead cells eaten) Necrosis : Not Self-Initiated Not Stereotypic Can Be Slow Messy (injury can
Types of Cell Death Questions : Apoptosis (Programmed Cell Death) : Cell-Autonomous Stereotypic Rapid Clean (dead cells eaten) Necrosis : Not Self-Initiated Not Stereotypic Can Be Slow Messy (injury can
Anterograde Jelly belly ligand to Alk receptor signaling at developing synapses is regulated by Mind the gap
 RESEARCH ARTICLE 3523 Development 137, 3523-3533 (2010) doi:10.1242/dev.047878 2010. Published by The Company of Biologists Ltd Anterograde Jelly belly ligand to Alk receptor signaling at developing synapses
RESEARCH ARTICLE 3523 Development 137, 3523-3533 (2010) doi:10.1242/dev.047878 2010. Published by The Company of Biologists Ltd Anterograde Jelly belly ligand to Alk receptor signaling at developing synapses
Homeostatic Control of Presynaptic Release Is Triggered by Postsynaptic Membrane Depolarization
 Neuron, Vol. 30, 737 749, June, 2001, Copyright 2001 by Cell Press Homeostatic Control of Presynaptic Release Is Triggered by Postsynaptic Membrane Depolarization Suzanne Paradis, Sean T. Sweeney, and
Neuron, Vol. 30, 737 749, June, 2001, Copyright 2001 by Cell Press Homeostatic Control of Presynaptic Release Is Triggered by Postsynaptic Membrane Depolarization Suzanne Paradis, Sean T. Sweeney, and
Honors Biology Reading Guide Chapter 11
 Honors Biology Reading Guide Chapter 11 v Promoter a specific nucleotide sequence in DNA located near the start of a gene that is the binding site for RNA polymerase and the place where transcription begins
Honors Biology Reading Guide Chapter 11 v Promoter a specific nucleotide sequence in DNA located near the start of a gene that is the binding site for RNA polymerase and the place where transcription begins
7.06 Cell Biology EXAM #3 KEY
 7.06 Cell Biology EXAM #3 KEY May 2, 2006 This is an OPEN BOOK exam, and you are allowed access to books, a calculator, and notes BUT NOT computers or any other types of electronic devices. Please write
7.06 Cell Biology EXAM #3 KEY May 2, 2006 This is an OPEN BOOK exam, and you are allowed access to books, a calculator, and notes BUT NOT computers or any other types of electronic devices. Please write
10/2/2015. Chapter 4. Determination and Differentiation. Neuroanatomical Diversity
 Chapter 4 Determination and Differentiation Neuroanatomical Diversity 1 Neurochemical diversity: another important aspect of neuronal fate Neurotransmitters and their receptors Excitatory Glutamate Acetylcholine
Chapter 4 Determination and Differentiation Neuroanatomical Diversity 1 Neurochemical diversity: another important aspect of neuronal fate Neurotransmitters and their receptors Excitatory Glutamate Acetylcholine
Varicose and cheerio collaborate with pebble to mediate semaphorin-1a reverse signaling in Drosophila
 Varicose and cheerio collaborate with pebble to mediate semaphorin-1a reverse signaling in Drosophila Sangyun Jeong a,1, Da-som Yang a, Young Gi Hong a, Sarah P. Mitchell b,c, Matthew P. rown b,c, and
Varicose and cheerio collaborate with pebble to mediate semaphorin-1a reverse signaling in Drosophila Sangyun Jeong a,1, Da-som Yang a, Young Gi Hong a, Sarah P. Mitchell b,c, Matthew P. rown b,c, and
Why Flies? stages of embryogenesis. The Fly in History
 The Fly in History 1859 Darwin 1866 Mendel c. 1890 Driesch, Roux (experimental embryology) 1900 rediscovery of Mendel (birth of genetics) 1910 first mutant (white) (Morgan) 1913 first genetic map (Sturtevant
The Fly in History 1859 Darwin 1866 Mendel c. 1890 Driesch, Roux (experimental embryology) 1900 rediscovery of Mendel (birth of genetics) 1910 first mutant (white) (Morgan) 1913 first genetic map (Sturtevant
SUPPLEMENTARY INFORMATION
 Supplementary Notes Downregulation of atlastin does not affect secretory traffic We investigated whether Atlastin might play a role in secretory traffic. Traffic impairment results in disruption of Golgi
Supplementary Notes Downregulation of atlastin does not affect secretory traffic We investigated whether Atlastin might play a role in secretory traffic. Traffic impairment results in disruption of Golgi
CIE Biology A-level Topic 15: Control and coordination
 CIE Biology A-level Topic 15: Control and coordination Notes Neuron structure The nerve cells called neurones play an important role in coordinating communication within the nervous system. The structure
CIE Biology A-level Topic 15: Control and coordination Notes Neuron structure The nerve cells called neurones play an important role in coordinating communication within the nervous system. The structure
Ral mediates activity-dependent growth of postsynaptic membranes via recruitment of the exocyst
 The EMBO Journal (2013) 32, 2039 2055 www.embojournal.org Ral mediates activity-dependent growth of postsynaptic membranes via recruitment of the exocyst THE EMBO JOURNAL Rita O Teodoro 1,2, *, Gulc in
The EMBO Journal (2013) 32, 2039 2055 www.embojournal.org Ral mediates activity-dependent growth of postsynaptic membranes via recruitment of the exocyst THE EMBO JOURNAL Rita O Teodoro 1,2, *, Gulc in
Over-expression screen in Drosophila identifies neuronal roles of GSK-3β/shaggy as. a regulator of AP-1 dependent developmental plasticity
 Genetics: Published Articles Ahead of Print, published on October 1, 2008 as 10.1534/genetics.107.085555 Over-expression screen in Drosophila identifies neuronal roles of GSK-3β/shaggy as a regulator of
Genetics: Published Articles Ahead of Print, published on October 1, 2008 as 10.1534/genetics.107.085555 Over-expression screen in Drosophila identifies neuronal roles of GSK-3β/shaggy as a regulator of
Neural development its all connected
 Neural development its all connected How do you build a complex nervous system? How do you build a complex nervous system? 1. Learn how tissue is instructed to become nervous system. Neural induction 2.
Neural development its all connected How do you build a complex nervous system? How do you build a complex nervous system? 1. Learn how tissue is instructed to become nervous system. Neural induction 2.
Biology EOC Review Study Questions
 Biology EOC Review Study Questions Microscopes and Characteristics of Life 1. How do you calculate total magnification on a compound light microscope? 2. What is the basic building block of all living
Biology EOC Review Study Questions Microscopes and Characteristics of Life 1. How do you calculate total magnification on a compound light microscope? 2. What is the basic building block of all living
Evolution of Transcription factor function: Homeotic (Hox) proteins
 Evolution of Transcription factor function: Homeotic (Hox) proteins Hox proteins regulate morphology in cellular zones on the anterior-posterior axis of embryos via the activation/repression of unknown
Evolution of Transcription factor function: Homeotic (Hox) proteins Hox proteins regulate morphology in cellular zones on the anterior-posterior axis of embryos via the activation/repression of unknown
Lecture 7. Development of the Fruit Fly Drosophila
 BIOLOGY 205/SECTION 7 DEVELOPMENT- LILJEGREN Lecture 7 Development of the Fruit Fly Drosophila 1. The fruit fly- a highly successful, specialized organism a. Quick life cycle includes three larval stages
BIOLOGY 205/SECTION 7 DEVELOPMENT- LILJEGREN Lecture 7 Development of the Fruit Fly Drosophila 1. The fruit fly- a highly successful, specialized organism a. Quick life cycle includes three larval stages
Introduction Principles of Signaling and Organization p. 3 Signaling in Simple Neuronal Circuits p. 4 Organization of the Retina p.
 Introduction Principles of Signaling and Organization p. 3 Signaling in Simple Neuronal Circuits p. 4 Organization of the Retina p. 5 Signaling in Nerve Cells p. 9 Cellular and Molecular Biology of Neurons
Introduction Principles of Signaling and Organization p. 3 Signaling in Simple Neuronal Circuits p. 4 Organization of the Retina p. 5 Signaling in Nerve Cells p. 9 Cellular and Molecular Biology of Neurons
The Transmembrane Semaphorin Sema I Is Required in Drosophila for Embryonic Motor and CNS Axon Guidance
 Neuron, Vol. 20, 207 220, February, 1998, Copyright 1998 by Cell Press The Transmembrane Semaphorin Sema I Is Required in Drosophila for Embryonic Motor and CNS Axon Guidance Hung-Hsiang Yu, Houmam H.
Neuron, Vol. 20, 207 220, February, 1998, Copyright 1998 by Cell Press The Transmembrane Semaphorin Sema I Is Required in Drosophila for Embryonic Motor and CNS Axon Guidance Hung-Hsiang Yu, Houmam H.
MOLECULAR CONTROL OF EMBRYONIC PATTERN FORMATION
 MOLECULAR CONTROL OF EMBRYONIC PATTERN FORMATION Drosophila is the best understood of all developmental systems, especially at the genetic level, and although it is an invertebrate it has had an enormous
MOLECULAR CONTROL OF EMBRYONIC PATTERN FORMATION Drosophila is the best understood of all developmental systems, especially at the genetic level, and although it is an invertebrate it has had an enormous
Supplemental Data. Wang et al. (2014). Plant Cell /tpc
 Supplemental Figure1: Mock and NPA-treated tomato plants. (A) NPA treated tomato (cv. Moneymaker) developed a pin-like inflorescence (arrowhead). (B) Comparison of first and second leaves from mock and
Supplemental Figure1: Mock and NPA-treated tomato plants. (A) NPA treated tomato (cv. Moneymaker) developed a pin-like inflorescence (arrowhead). (B) Comparison of first and second leaves from mock and
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1121356/dc1 Supporting Online Material for Polar PIN Localization Directs Auxin Flow in Plants Justyna Wiśniewska, Jian Xu, Daniela Seifertová, Philip B. Brewer, Kamil
www.sciencemag.org/cgi/content/full/1121356/dc1 Supporting Online Material for Polar PIN Localization Directs Auxin Flow in Plants Justyna Wiśniewska, Jian Xu, Daniela Seifertová, Philip B. Brewer, Kamil
Supplementary Figure 1: To test the role of mir-17~92 in orthologous genetic model of ADPKD, we generated Ksp/Cre;Pkd1 F/F (Pkd1-KO) and Ksp/Cre;Pkd1
 Supplementary Figure 1: To test the role of mir-17~92 in orthologous genetic model of ADPKD, we generated Ksp/Cre;Pkd1 F/F (Pkd1-KO) and Ksp/Cre;Pkd1 F/F ;mir-17~92 F/F (Pkd1-miR-17~92KO) mice. (A) Q-PCR
Supplementary Figure 1: To test the role of mir-17~92 in orthologous genetic model of ADPKD, we generated Ksp/Cre;Pkd1 F/F (Pkd1-KO) and Ksp/Cre;Pkd1 F/F ;mir-17~92 F/F (Pkd1-miR-17~92KO) mice. (A) Q-PCR
Early consolidation of development and physiology of an identified presynaptic nerve terminal
 Laviolette and Stewart BMC Neuroscience 2013, 14:124 RESEARCH ARTICLE Open Access Early consolidation of development and physiology of an identified presynaptic nerve terminal Matthew Laviolette and Bryan
Laviolette and Stewart BMC Neuroscience 2013, 14:124 RESEARCH ARTICLE Open Access Early consolidation of development and physiology of an identified presynaptic nerve terminal Matthew Laviolette and Bryan
Chapter 18 Regulation of Gene Expression
 Chapter 18 Regulation of Gene Expression Differential gene expression Every somatic cell in an individual organism contains the same genetic information and replicated from the same original fertilized
Chapter 18 Regulation of Gene Expression Differential gene expression Every somatic cell in an individual organism contains the same genetic information and replicated from the same original fertilized
Neurons and Nervous Systems
 34 Neurons and Nervous Systems Concept 34.1 Nervous Systems Consist of Neurons and Glia Nervous systems have two categories of cells: Neurons, or nerve cells, are excitable they generate and transmit electrical
34 Neurons and Nervous Systems Concept 34.1 Nervous Systems Consist of Neurons and Glia Nervous systems have two categories of cells: Neurons, or nerve cells, are excitable they generate and transmit electrical
downstream (0.8 kb) homologous sequences to the genomic locus of DIC. A DIC mutant strain (ro- 6
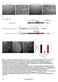 A B C D ts Figure S1 Generation of DIC- mcherry expressing N.crassa strain. A. N. crassa colony morphology. When a cot1 (top, left panel) strain is grown at permissive temperature (25 C), it exhibits straight
A B C D ts Figure S1 Generation of DIC- mcherry expressing N.crassa strain. A. N. crassa colony morphology. When a cot1 (top, left panel) strain is grown at permissive temperature (25 C), it exhibits straight
Fig. S1. Expression pattern of moody-gal4 in third instar. Maximum projection illustrating a dissected moody-gal4>ngfp L3 larva stained for Repo
 Fig. S1. Expression pattern of moody-gal4 in third instar. Maximum projection illustrating a dissected moody-gal4>ngfp L3 larva stained for Repo (magenta), Fas2 (blue) and GFP (green) in overview (A) and
Fig. S1. Expression pattern of moody-gal4 in third instar. Maximum projection illustrating a dissected moody-gal4>ngfp L3 larva stained for Repo (magenta), Fas2 (blue) and GFP (green) in overview (A) and
Supplemental table S7.
 Supplemental table S7. GO terms significantly enriched in significantly up-regulated genes of the microarray. K: number of genes from the input cluster in the given category. F: number of total genes in
Supplemental table S7. GO terms significantly enriched in significantly up-regulated genes of the microarray. K: number of genes from the input cluster in the given category. F: number of total genes in
Shavenbaby Couples Patterning to Epidermal Cell Shape Control. Chanut-Delalande H, Fernandes I, Roch F, Payre F, Plaza S (2006) PLoS Biol 4(9): e290
 Shavenbaby Couples Patterning to Epidermal Cell Shape Control. Chanut-Delalande H, Fernandes I, Roch F, Payre F, Plaza S (2006) PLoS Biol 4(9): e290 Question (from Introduction): How does svb control the
Shavenbaby Couples Patterning to Epidermal Cell Shape Control. Chanut-Delalande H, Fernandes I, Roch F, Payre F, Plaza S (2006) PLoS Biol 4(9): e290 Question (from Introduction): How does svb control the
IN the Drosophila ventral nerve cord, there are 20
 Copyright Ó 2007 by the Genetics Society of America DOI: 10.1534/genetics.107.075085 Regulation of Axon Guidance by Slit and Netrin Signaling in the Drosophila Ventral Nerve Cord Krishna Moorthi Bhat,
Copyright Ó 2007 by the Genetics Society of America DOI: 10.1534/genetics.107.075085 Regulation of Axon Guidance by Slit and Netrin Signaling in the Drosophila Ventral Nerve Cord Krishna Moorthi Bhat,
