pharmaceutical industry- drug discovery
|
|
|
- Gladys Porter
- 5 years ago
- Views:
Transcription
1 ombinatorial hemistry: molecular diversity "Synthesis and pplications of Small Molecule Libraries." Thompson, L..; llman, J.. hem. ev.,, -00. "esign, Synthesis, and valuation of Small-Molecule Libraries. llman, J.. cc. hem. es.,, -. ombinatorial hemistry, icholas K. Terrett, xford University Press, London, pharmaceutical industry- drug discovery Lead mpd optimization (synthesis) rug 0 years $ million $ $ $ Lead identification: literature (open & patent) nature (natural products) areful optimization of a lead structure via chemical synthesis methyl-ethyl-butyl-futile game umber of marketable drugs per compounds that undergo preliminary biological testing 0,000 ational drug design ombinatorial chemistry Peptides: poor bioavailability, poor transport, easily metabolized: poor drug candidates. owever, they are the natural substrates for many enzymes and receptors (drug targets) and have well-defined conformations: excellent lead compounds nzymes: converts a substrate to a distinct product eceptor: binds a ligand (no reaction), causing a chain of physiochemical events leading to a pharmacological response. gonist: substance that interacts (binds) with a receptor and elicits an observable response ntagonist: substances inhibits the affect of an agonist, but has no biological activity of its own
2 xample of molecular diversity: tetra-peptide: consider only the 0 natural amino acids (L-series) 0 = 0,000 different tetra-peptides! now include the -amino acids (0 L + = ) =. million different tetra-peptides!! now include 0 unnatural amino acids = million different tetra-peptides!!!! ombinatorial chemistry: method by which a family (library) of related compounds (structurally & synthetically) can be prepared and evaluated (screened) or multi-step synthesis, one must use solid-phase synthetic approach in order to expedite purification of intermediates Split synthesis (mixture libraries) combine, mix,split combine, mix,split
3 0 Split synthesis (con t) 0 Why not? eactivity of the coupling reaction may be different and this could bias the library Split synthesis approach, all compounds are equally represented (each coupling is individually controlled),,,,,, etc
4 econvolution of the library via biological activity and sub-library re-synthesis iological activity of the library select library - - iological activity of the library select library iological activity of the library select library Lead mpd
5 Problems: deconvolution of the library can be labor intensive can be fooled by low concentrations of highly active compounds activity is dependent upon the compound and its concentration activity observed is the the combined activity of the entire library dvantage: can synthesize a very large number of compounds very quickly and relatively easily depending on the chemistry. ncoded Libraries ) ) combine, mix,split ) ) combine, mix,split ) )
6 ncoded libaries (con t) leave the marker and analyze the code. ach unique marker codes for a unique peptide sequence ) ) ) ) ) ) : = = = = = = = = = ) linker- --M ) linker-code-- linker linker M code library ode (-0%) library region and coding region must be orthogonally protected ) piperidine ) add linker linker code M ) + )add code linker linker code ode M Synthesis of an encoded library
7 sub-library tag pool tag x y eading the code selectively remove the code from the resin hydrolyze and analyze + + n-ead ssay for eceptor inding using a luorescently Labeled eceptor ) dd labeled receptor labelled with fluorescent tag ) allow to bind to ligands on the beads ) wash to remove unbound receptor eads
8 Spatially ddressable, Parallel Synthesis 0 Spatially ddressable, Parallel Synthesis (con t)
9 Spatially addressable, parallel libraries dvantage: synthesizing pure compounds, no need to deconvolute the library isadvantage libraries tend to be much smaller iversomer apparatus cc. hem. es.,, -
10 nhibitors sp rg Val Tyr le is Pro Phe is Leu Val le is sn! angiotensinogen! inhibitors aspartyl protease renin sp rg Val Tyr le is Pro Phe is Leu! angiotensin! (little biological activity)! Zn + protease angiotensin converting enzyme () sp rg Val Tyr le is Pro Phe! angiotensin! (vasoconstriction high blood pressure)! iovan S aptopril Proteases: catalyzes the hydrolysis of peptide bonds protease +. Serine protease. ysteine protease. spartyl protease. Zinc (metallo) protease : zinc protease ( no x-ray or M structure), compared to carboxypeptidase or thermolysin important catalytic groups: lu-0, is-, is- (catalytic triad)
11 inhibitor Library r r g, t g r,-dipole g r Z,-diploar cycloaddition r = -, -, - ( ), - Ph Z l r Z r Z r = Z = tu S aptopril ( nhibitor) l = cs l cs l cs l 0 cmpds, each are multiple stereoisomers cs Ph K i ~ 0 pm (x better than aptopril Parallel Synthesis of a enzodiazepine Library ' M ' M ) ' ) + ) base ) X cleave from solid support ' ' enzodiazepine l l Valium (diazapam) Librium (chlordiazepoxide)
12 -aminobenzophenones Parallel Synthesis of a enzodiazepine Library α-amino acids alkylating agents - l r r (-aminobenzophenone) x (amino acids) x (alkyl halides) = compounds holecystikinin (K)! K-: sp-tyr(s)-met-ly-trp-met-sp-phe-! β-turn Mimics: (i+) (i+) (i+) (i) Linchpin Scaffold + β-turn + + S n( ) ( )n
13 ombinatorial synthesis of linchpin β-turn mimic r, T r S n S-tu S n S-tu ) M, T ) piperidine n S S-tu r, T n S S-tu r ) Ph P ) cyclization ) n S valuated for Somatostatin receptor binding! n=, x = x = 0 = β-turn mimics! 0
به نام خدا. New topics in. organic chemistry. Dr Morteza Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran
 به نام خدا New topics in 2 organic chemistry Dr Morteza Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Combinatorial chemistry 1 http://www.combichemistry.com/
به نام خدا New topics in 2 organic chemistry Dr Morteza Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Combinatorial chemistry 1 http://www.combichemistry.com/
Ping-Chiang Lyu. Institute of Bioinformatics and Structural Biology, Department of Life Science, National Tsing Hua University.
 Pharmacophore-based Drug design Ping-Chiang Lyu Institute of Bioinformatics and Structural Biology, Department of Life Science, National Tsing Hua University 96/08/07 Outline Part I: Analysis The analytical
Pharmacophore-based Drug design Ping-Chiang Lyu Institute of Bioinformatics and Structural Biology, Department of Life Science, National Tsing Hua University 96/08/07 Outline Part I: Analysis The analytical
EMPIRICAL VS. RATIONAL METHODS OF DISCOVERING NEW DRUGS
 EMPIRICAL VS. RATIONAL METHODS OF DISCOVERING NEW DRUGS PETER GUND Pharmacopeia Inc., CN 5350 Princeton, NJ 08543, USA pgund@pharmacop.com Empirical and theoretical approaches to drug discovery have often
EMPIRICAL VS. RATIONAL METHODS OF DISCOVERING NEW DRUGS PETER GUND Pharmacopeia Inc., CN 5350 Princeton, NJ 08543, USA pgund@pharmacop.com Empirical and theoretical approaches to drug discovery have often
Chemistry 224 Bioorganic Chemistry Friday, Sept. 29, This Exam is closed book and closed notes. Please show all your work!
 page 1 of 6 hemistry 224 ame Bioorganic hemistry Friday, ept. 29, 2000 Exam 1 100 points This Exam is closed book and closed notes Please show all your work! tereochemistry counts as indicated! eatness
page 1 of 6 hemistry 224 ame Bioorganic hemistry Friday, ept. 29, 2000 Exam 1 100 points This Exam is closed book and closed notes Please show all your work! tereochemistry counts as indicated! eatness
Chemistry Problem Set #9 Due on Thursday 11/15/18 in class.
 Chemistry 391 - Problem Set #9 Due on Thursday 11/15/18 in class. Name 1. There is a real enzyme called cocaine esterase that is produced in bacteria that live at the base of the coca plant. The enzyme
Chemistry 391 - Problem Set #9 Due on Thursday 11/15/18 in class. Name 1. There is a real enzyme called cocaine esterase that is produced in bacteria that live at the base of the coca plant. The enzyme
From Friday s material
 5.111 Lecture 35 35.1 Kinetics Topic: Catalysis Chapter 13 (Section 13.14-13.15) From Friday s material Le Chatelier's Principle - when a stress is applied to a system in equilibrium, the equilibrium tends
5.111 Lecture 35 35.1 Kinetics Topic: Catalysis Chapter 13 (Section 13.14-13.15) From Friday s material Le Chatelier's Principle - when a stress is applied to a system in equilibrium, the equilibrium tends
Combinatorial Chemistry Technology
 ugo Kubinyi, www.kubinyi.de ombinatorial hemistry Technology ugo Kubinyi Germany E-Mail kubinyi@t-online.de omepage www.kubinyi.de ugo Kubinyi, www.kubinyi.de ombinatorial Diversity in ature 20 natural
ugo Kubinyi, www.kubinyi.de ombinatorial hemistry Technology ugo Kubinyi Germany E-Mail kubinyi@t-online.de omepage www.kubinyi.de ugo Kubinyi, www.kubinyi.de ombinatorial Diversity in ature 20 natural
Principles of Drug Design
 Advanced Medicinal Chemistry II Principles of Drug Design Tentative Course Outline Instructors: Longqin Hu and John Kerrigan Direct questions and enquiries to the Course Coordinator: Longqin Hu I. Introduction
Advanced Medicinal Chemistry II Principles of Drug Design Tentative Course Outline Instructors: Longqin Hu and John Kerrigan Direct questions and enquiries to the Course Coordinator: Longqin Hu I. Introduction
Module No. 31: Peptide Synthesis: Definition, Methodology & applications
 PAPER 9: TECHNIQUES USED IN MOLECULAR BIOPHYSICS I Module No. 31: Peptide Synthesis: Definition, Methodology & applications Objectives: 1. Introduction 2. Synthesis of peptide 2.1. N-terminal protected
PAPER 9: TECHNIQUES USED IN MOLECULAR BIOPHYSICS I Module No. 31: Peptide Synthesis: Definition, Methodology & applications Objectives: 1. Introduction 2. Synthesis of peptide 2.1. N-terminal protected
7.012 Problem Set 1. i) What are two main differences between prokaryotic cells and eukaryotic cells?
 ame 7.01 Problem Set 1 Section Question 1 a) What are the four major types of biological molecules discussed in lecture? Give one important function of each type of biological molecule in the cell? b)
ame 7.01 Problem Set 1 Section Question 1 a) What are the four major types of biological molecules discussed in lecture? Give one important function of each type of biological molecule in the cell? b)
Early Stages of Drug Discovery in the Pharmaceutical Industry
 Early Stages of Drug Discovery in the Pharmaceutical Industry Daniel Seeliger / Jan Kriegl, Discovery Research, Boehringer Ingelheim September 29, 2016 Historical Drug Discovery From Accidential Discovery
Early Stages of Drug Discovery in the Pharmaceutical Industry Daniel Seeliger / Jan Kriegl, Discovery Research, Boehringer Ingelheim September 29, 2016 Historical Drug Discovery From Accidential Discovery
Syllabus. 1. Occurrence and Functions of Peptides in Nature and Every Day Life hormones, neurotransmitters, therapeutics, artificial sweetener,
 Syllabus 1. ccurrence and Functions of Peptides in ature and Every Day Life hormones, neurotransmitters, therapeutics, artificial sweetener, 2. Peptide Synthesis a) spartam: Properties of amino acids;
Syllabus 1. ccurrence and Functions of Peptides in ature and Every Day Life hormones, neurotransmitters, therapeutics, artificial sweetener, 2. Peptide Synthesis a) spartam: Properties of amino acids;
COMBINATORIAL CHEMISTRY: CURRENT APPROACH
 COMBINATORIAL CHEMISTRY: CURRENT APPROACH Dwivedi A. 1, Sitoke A. 2, Joshi V. 3, Akhtar A.K. 4* and Chaturvedi M. 1, NRI Institute of Pharmaceutical Sciences, Bhopal, M.P.-India 2, SRM College of Pharmacy,
COMBINATORIAL CHEMISTRY: CURRENT APPROACH Dwivedi A. 1, Sitoke A. 2, Joshi V. 3, Akhtar A.K. 4* and Chaturvedi M. 1, NRI Institute of Pharmaceutical Sciences, Bhopal, M.P.-India 2, SRM College of Pharmacy,
It s the amino acids!
 Catalytic Mechanisms HOW do enzymes do their job? Reducing activation energy sure, but HOW does an enzyme catalysis reduce the energy barrier ΔG? Remember: The rate of a chemical reaction of substrate
Catalytic Mechanisms HOW do enzymes do their job? Reducing activation energy sure, but HOW does an enzyme catalysis reduce the energy barrier ΔG? Remember: The rate of a chemical reaction of substrate
Receptor Based Drug Design (1)
 Induced Fit Model For more than 100 years, the behaviour of enzymes had been explained by the "lock-and-key" mechanism developed by pioneering German chemist Emil Fischer. Fischer thought that the chemicals
Induced Fit Model For more than 100 years, the behaviour of enzymes had been explained by the "lock-and-key" mechanism developed by pioneering German chemist Emil Fischer. Fischer thought that the chemicals
COMBINATORIAL CHEMISTRY IN A HISTORICAL PERSPECTIVE
 NUE FEATURE T R A N S F O R M I N G C H A L L E N G E S I N T O M E D I C I N E Nuevolution Feature no. 1 October 2015 Technical Information COMBINATORIAL CHEMISTRY IN A HISTORICAL PERSPECTIVE A PROMISING
NUE FEATURE T R A N S F O R M I N G C H A L L E N G E S I N T O M E D I C I N E Nuevolution Feature no. 1 October 2015 Technical Information COMBINATORIAL CHEMISTRY IN A HISTORICAL PERSPECTIVE A PROMISING
-1- HO H O H. β-d-galactopyranose (A) OMe CH 3 I. MeO. Ag 2 O. O Me HNO 3. OAc (CH 3 CO) 2 O. AcO. pyridine. O Ac. NaBH 4 H 2 O. MeOH. dry HCl.
 -1-1. Draw structures for the products you would expect to obtain from reaction of β-dgalactopyranose () with each of the reagents below. Be sure to include all relevant stereochemistry. (40 pts). β-d-galactopyranose
-1-1. Draw structures for the products you would expect to obtain from reaction of β-dgalactopyranose () with each of the reagents below. Be sure to include all relevant stereochemistry. (40 pts). β-d-galactopyranose
Conformational search and QSAR (quantitative structure-activity relationships)
 Bio 5476 Lab Exercise 11.7.08 Instructor: Dan Kuster (d.kuster@gmail.com) Conformational search and QSAR (quantitative structure-activity relationships) Purpose: In this lab, you will use SYBYL to build,
Bio 5476 Lab Exercise 11.7.08 Instructor: Dan Kuster (d.kuster@gmail.com) Conformational search and QSAR (quantitative structure-activity relationships) Purpose: In this lab, you will use SYBYL to build,
NMR study of complexes between low molecular mass inhibitors and the West Nile virus NS2B-NS3 protease
 University of Wollongong Research Online Faculty of Science - Papers (Archive) Faculty of Science, Medicine and Health 2009 NMR study of complexes between low molecular mass inhibitors and the West Nile
University of Wollongong Research Online Faculty of Science - Papers (Archive) Faculty of Science, Medicine and Health 2009 NMR study of complexes between low molecular mass inhibitors and the West Nile
Aziridine Carboxylates, Carboxamides and Lactones: New Methods for Their Preparation and Their Transformation into α- and β-amino Acid Derivatives
 Molecules 2000, 5 293 Aziridine Carboxylates, Carboxamides and Lactones: ew Methods for Their Preparation and Their Transformation into α- and β-amino Acid Derivatives obert H. Dodd Institut de Chimie
Molecules 2000, 5 293 Aziridine Carboxylates, Carboxamides and Lactones: ew Methods for Their Preparation and Their Transformation into α- and β-amino Acid Derivatives obert H. Dodd Institut de Chimie
Advanced Certificate in Principles in Protein Structure. You will be given a start time with your exam instructions
 BIRKBECK COLLEGE (University of London) Advanced Certificate in Principles in Protein Structure MSc Structural Molecular Biology Date: Thursday, 1st September 2011 Time: 3 hours You will be given a start
BIRKBECK COLLEGE (University of London) Advanced Certificate in Principles in Protein Structure MSc Structural Molecular Biology Date: Thursday, 1st September 2011 Time: 3 hours You will be given a start
A. Reaction Mechanisms and Catalysis (1) proximity effect (2) acid-base catalysts (3) electrostatic (4) functional groups (5) structural flexibility
 (P&S Ch 5; Fer Ch 2, 9; Palm Ch 10,11; Zub Ch 9) A. Reaction Mechanisms and Catalysis (1) proximity effect (2) acid-base catalysts (3) electrostatic (4) functional groups (5) structural flexibility B.
(P&S Ch 5; Fer Ch 2, 9; Palm Ch 10,11; Zub Ch 9) A. Reaction Mechanisms and Catalysis (1) proximity effect (2) acid-base catalysts (3) electrostatic (4) functional groups (5) structural flexibility B.
Reading for today: Chapter 16 (selections from Sections A, B and C) Friday and Monday: Chapter 17 (Diffusion)
 Lecture 29 Enzymes Reading for today: Chapter 6 (selections from Sections, B and C) Friday and Monday: Chapter 7 (Diffusion) 4/3/6 Today s Goals Michaelis-Menten mechanism for simple enzyme reactions:
Lecture 29 Enzymes Reading for today: Chapter 6 (selections from Sections, B and C) Friday and Monday: Chapter 7 (Diffusion) 4/3/6 Today s Goals Michaelis-Menten mechanism for simple enzyme reactions:
Introduction to Chemoinformatics and Drug Discovery
 Introduction to Chemoinformatics and Drug Discovery Irene Kouskoumvekaki Associate Professor February 15 th, 2013 The Chemical Space There are atoms and space. Everything else is opinion. Democritus (ca.
Introduction to Chemoinformatics and Drug Discovery Irene Kouskoumvekaki Associate Professor February 15 th, 2013 The Chemical Space There are atoms and space. Everything else is opinion. Democritus (ca.
Principles of Drug Design
 (16:663:502) Instructors: Longqin Hu and John Kerrigan Direct questions and enquiries to the Course Coordinator: Longqin Hu For more current information, please check WebCT at https://webct.rutgers.edu
(16:663:502) Instructors: Longqin Hu and John Kerrigan Direct questions and enquiries to the Course Coordinator: Longqin Hu For more current information, please check WebCT at https://webct.rutgers.edu
Biological Macromolecules
 Introduction for Chem 493 Chemistry of Biological Macromolecules Dr. L. Luyt January 2008 Dr. L. Luyt Chem 493-2008 1 Biological macromolecules are the molecules of life allow for organization serve a
Introduction for Chem 493 Chemistry of Biological Macromolecules Dr. L. Luyt January 2008 Dr. L. Luyt Chem 493-2008 1 Biological macromolecules are the molecules of life allow for organization serve a
C a h p a t p e t r e r 6 E z n y z m y e m s
 Chapter 6 Enzymes 4. Examples of enzymatic reactions acid-base catalysis: give and take protons covalent catalysis: a transient covalent bond is formed between the enzyme and the substrate metal ion catalysis:
Chapter 6 Enzymes 4. Examples of enzymatic reactions acid-base catalysis: give and take protons covalent catalysis: a transient covalent bond is formed between the enzyme and the substrate metal ion catalysis:
Biochemistry 3100 Sample Problems Binding proteins, Kinetics & Catalysis
 (1) Draw an approximate denaturation curve for a typical blood protein (eg myoglobin) as a function of ph. (2) Myoglobin is a simple, single subunit binding protein that has an oxygen storage function
(1) Draw an approximate denaturation curve for a typical blood protein (eg myoglobin) as a function of ph. (2) Myoglobin is a simple, single subunit binding protein that has an oxygen storage function
Enzyme function: the transition state. Enzymes & Kinetics V: Mechanisms. Catalytic Reactions. Margaret A. Daugherty A B. Lecture 16: Fall 2003
 Lecture 16: Enzymes & Kinetics V: Mechanisms Margaret A. Daugherty Fall 2003 Enzyme function: the transition state Catalytic Reactions A B Catalysts (e.g. enzymes) act by lowering the transition state
Lecture 16: Enzymes & Kinetics V: Mechanisms Margaret A. Daugherty Fall 2003 Enzyme function: the transition state Catalytic Reactions A B Catalysts (e.g. enzymes) act by lowering the transition state
Catalytic Reactions. Intermediate State in Catalysis. Lecture 16: Catalyzed reaction. Uncatalyzed reaction. Enzymes & Kinetics V: Mechanisms
 Enzyme function: the transition state Catalytic Reactions Lecture 16: Enzymes & Kinetics V: Mechanisms Margaret A. Daugherty Fall 2003 A B Catalysts (e.g. enzymes) act by lowering the transition state
Enzyme function: the transition state Catalytic Reactions Lecture 16: Enzymes & Kinetics V: Mechanisms Margaret A. Daugherty Fall 2003 A B Catalysts (e.g. enzymes) act by lowering the transition state
Retrieving hits through in silico screening and expert assessment M. N. Drwal a,b and R. Griffith a
 Retrieving hits through in silico screening and expert assessment M.. Drwal a,b and R. Griffith a a: School of Medical Sciences/Pharmacology, USW, Sydney, Australia b: Charité Berlin, Germany Abstract:
Retrieving hits through in silico screening and expert assessment M.. Drwal a,b and R. Griffith a a: School of Medical Sciences/Pharmacology, USW, Sydney, Australia b: Charité Berlin, Germany Abstract:
Patrick, An Introduction to Medicinal Chemistry 5e Chapter 7 Enzymes as drug targets. 1) The structures of isoleucine and valine are as follows.
 Answers to end-of-chapter questions 1) The structures of isoleucine and valine are as follows. 2 C 2 2 C 2 3 C C 3 3 C C 3 Isoleucine Valine Isoleucine has a larger side chain than valine, and so there
Answers to end-of-chapter questions 1) The structures of isoleucine and valine are as follows. 2 C 2 2 C 2 3 C C 3 3 C C 3 Isoleucine Valine Isoleucine has a larger side chain than valine, and so there
4 Examples of enzymes
 Catalysis 1 4 Examples of enzymes Adding water to a substrate: Serine proteases. Carbonic anhydrase. Restrictions Endonuclease. Transfer of a Phosphoryl group from ATP to a nucleotide. Nucleoside monophosphate
Catalysis 1 4 Examples of enzymes Adding water to a substrate: Serine proteases. Carbonic anhydrase. Restrictions Endonuclease. Transfer of a Phosphoryl group from ATP to a nucleotide. Nucleoside monophosphate
Biochemistry 462a - Enzyme Kinetics Reading - Chapter 8 Practice problems - Chapter 8: (not yet assigned); Enzymes extra problems
 Biochemistry 462a - Enzyme Kinetics Reading - Chapter 8 Practice problems - Chapter 8: (not yet assigned); Enzymes extra problems Introduction Enzymes are Biological Catalysis A catalyst is a substance
Biochemistry 462a - Enzyme Kinetics Reading - Chapter 8 Practice problems - Chapter 8: (not yet assigned); Enzymes extra problems Introduction Enzymes are Biological Catalysis A catalyst is a substance
SYSTEMATIC SEARCH AND ANGIOTENSIN CONVERTING ENZYME INHIBITORS Ligand-Based SAR
 SYSTEMATIC SEARCH AND ANGIOTENSIN CONVERTING ENZYME INHIBITORS Ligand-Based SAR Garland R. Marshall Sapienza Universita di Roma October 27, 2014 The Pharmacophore Concept (Active-site Mapping) MOST THERAPEUTIC
SYSTEMATIC SEARCH AND ANGIOTENSIN CONVERTING ENZYME INHIBITORS Ligand-Based SAR Garland R. Marshall Sapienza Universita di Roma October 27, 2014 The Pharmacophore Concept (Active-site Mapping) MOST THERAPEUTIC
Central Dogma. modifications genome transcriptome proteome
 entral Dogma DA ma protein post-translational modifications genome transcriptome proteome 83 ierarchy of Protein Structure 20 Amino Acids There are 20 n possible sequences for a protein of n residues!
entral Dogma DA ma protein post-translational modifications genome transcriptome proteome 83 ierarchy of Protein Structure 20 Amino Acids There are 20 n possible sequences for a protein of n residues!
Read more about Pauling and more scientists at: Profiles in Science, The National Library of Medicine, profiles.nlm.nih.gov
 2018 Biochemistry 110 California Institute of Technology Lecture 2: Principles of Protein Structure Linus Pauling (1901-1994) began his studies at Caltech in 1922 and was directed by Arthur Amos oyes to
2018 Biochemistry 110 California Institute of Technology Lecture 2: Principles of Protein Structure Linus Pauling (1901-1994) began his studies at Caltech in 1922 and was directed by Arthur Amos oyes to
CH MEDICINAL CHEMISTRY
 CH 458 - MEDICINAL CHEMISTRY SPRING 2011 M: 5:15pm-8 pm Sci-1-089 Prerequisite: Organic Chemistry II (Chem 254 or Chem 252, or equivalent transfer course) Instructor: Dr. Bela Torok Room S-1-132, Science
CH 458 - MEDICINAL CHEMISTRY SPRING 2011 M: 5:15pm-8 pm Sci-1-089 Prerequisite: Organic Chemistry II (Chem 254 or Chem 252, or equivalent transfer course) Instructor: Dr. Bela Torok Room S-1-132, Science
Chapter 8: An Introduction to Metabolism
 Chapter 8: An Introduction to Metabolism Key Concepts 8.1 An organism s metabolism transforms matter and energy, subject to the laws of thermodynamics 8.2 The free-energy change of a reaction tells us
Chapter 8: An Introduction to Metabolism Key Concepts 8.1 An organism s metabolism transforms matter and energy, subject to the laws of thermodynamics 8.2 The free-energy change of a reaction tells us
CHEM J-9 June 2014
 CEM1611 2014-J-9 June 2014 Alanine (ala) and lysine (lys) are two amino acids with the structures given below as Fischer projections. The pk a values of the conjugate acid forms of the different functional
CEM1611 2014-J-9 June 2014 Alanine (ala) and lysine (lys) are two amino acids with the structures given below as Fischer projections. The pk a values of the conjugate acid forms of the different functional
Structural biology and drug design: An overview
 Structural biology and drug design: An overview livier Taboureau Assitant professor Chemoinformatics group-cbs-dtu otab@cbs.dtu.dk Drug discovery Drug and drug design A drug is a key molecule involved
Structural biology and drug design: An overview livier Taboureau Assitant professor Chemoinformatics group-cbs-dtu otab@cbs.dtu.dk Drug discovery Drug and drug design A drug is a key molecule involved
A) at equilibrium B) endergonic C) endothermic D) exergonic E) exothermic.
 CHEM 2770: Elements of Biochemistry Mid Term EXAMINATION VERSION A Date: October 29, 2014 Instructor: H. Perreault Location: 172 Schultz Time: 4 or 6 pm. Duration: 1 hour Instructions Please mark the Answer
CHEM 2770: Elements of Biochemistry Mid Term EXAMINATION VERSION A Date: October 29, 2014 Instructor: H. Perreault Location: 172 Schultz Time: 4 or 6 pm. Duration: 1 hour Instructions Please mark the Answer
Dynamic Combinatorial Chemistry in the identification of new host guest interactions: proof of principle
 Dynamic Combinatorial Chemistry in the identification of new host guest interactions: proof of principle ick Paras MacMillan Group Meeting ctober 17, 2001 Lead References: Lehn, J.-M.; Eliseev, A. V. Science
Dynamic Combinatorial Chemistry in the identification of new host guest interactions: proof of principle ick Paras MacMillan Group Meeting ctober 17, 2001 Lead References: Lehn, J.-M.; Eliseev, A. V. Science
OXFORD H i g h e r E d u c a t i o n Oxford University Press, All rights reserved.
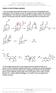 Patrick, An Introduction to dicinal hemistry 4e Answers to end-of-chapter questions 1) The mechanism below shows the release of one molecule of formaldehyde from methenamine. The mechanism can then be
Patrick, An Introduction to dicinal hemistry 4e Answers to end-of-chapter questions 1) The mechanism below shows the release of one molecule of formaldehyde from methenamine. The mechanism can then be
Chemistry Chapter 22
 hemistry 2100 hapter 22 Proteins Proteins serve many functions, including the following. 1. Structure: ollagen and keratin are the chief constituents of skin, bone, hair, and nails. 2. atalysts: Virtually
hemistry 2100 hapter 22 Proteins Proteins serve many functions, including the following. 1. Structure: ollagen and keratin are the chief constituents of skin, bone, hair, and nails. 2. atalysts: Virtually
AMRI COMPOUND LIBRARY CONSORTIUM: A NOVEL WAY TO FILL YOUR DRUG PIPELINE
 AMRI COMPOUD LIBRARY COSORTIUM: A OVEL WAY TO FILL YOUR DRUG PIPELIE Muralikrishna Valluri, PhD & Douglas B. Kitchen, PhD Summary The creation of high-quality, innovative small molecule leads is a continual
AMRI COMPOUD LIBRARY COSORTIUM: A OVEL WAY TO FILL YOUR DRUG PIPELIE Muralikrishna Valluri, PhD & Douglas B. Kitchen, PhD Summary The creation of high-quality, innovative small molecule leads is a continual
2015 AP Biology Unit 2 PRETEST- Introduction to the Cell and Biochemistry
 Name: Class: _ Date: _ 2015 AP Biology Unit 2 PRETEST- Introduction to the Cell and Biochemistry Multiple Choice Identify the choice that best completes the statement or answers the question. 1) In what
Name: Class: _ Date: _ 2015 AP Biology Unit 2 PRETEST- Introduction to the Cell and Biochemistry Multiple Choice Identify the choice that best completes the statement or answers the question. 1) In what
Exam I Answer Key: Summer 2006, Semester C
 1. Which of the following tripeptides would migrate most rapidly towards the negative electrode if electrophoresis is carried out at ph 3.0? a. gly-gly-gly b. glu-glu-asp c. lys-glu-lys d. val-asn-lys
1. Which of the following tripeptides would migrate most rapidly towards the negative electrode if electrophoresis is carried out at ph 3.0? a. gly-gly-gly b. glu-glu-asp c. lys-glu-lys d. val-asn-lys
Mechanisms of catalysis
 Mechanisms of catalysis Proximity and orientation effects Proximity: Reaction between bound molecules doesn't require an improbable collision of 2 molecules -- they're already in "contact" (increases the
Mechanisms of catalysis Proximity and orientation effects Proximity: Reaction between bound molecules doesn't require an improbable collision of 2 molecules -- they're already in "contact" (increases the
Catalysis. Instructor: Dr. Tsung-Lin Li Genomics Research Center Academia Sinica
 Catalysis Instructor: Dr. Tsung-Lin Li Genomics Research Center Academia Sinica References: Biochemistry" by Donald Voet and Judith G. Voet Biochemistry" by Christopher K. Mathews, K. E. Van Hold and Kevin
Catalysis Instructor: Dr. Tsung-Lin Li Genomics Research Center Academia Sinica References: Biochemistry" by Donald Voet and Judith G. Voet Biochemistry" by Christopher K. Mathews, K. E. Van Hold and Kevin
CHM 251 Organic Chemistry 1
 CM 251 rganic Chemistry 1 NAME Fall 2008 omework #2 Due: Tuesday, ctober 7, 2008 at 7 am Print out this assignment and complete all of your work on these pages. Question #1 Rank the following alkenes in
CM 251 rganic Chemistry 1 NAME Fall 2008 omework #2 Due: Tuesday, ctober 7, 2008 at 7 am Print out this assignment and complete all of your work on these pages. Question #1 Rank the following alkenes in
An Introduction to Metabolism
 An Introduction to Metabolism I. All of an organism=s chemical reactions taken together is called metabolism. A. Metabolic pathways begin with a specific molecule, which is then altered in a series of
An Introduction to Metabolism I. All of an organism=s chemical reactions taken together is called metabolism. A. Metabolic pathways begin with a specific molecule, which is then altered in a series of
Supplementary Figure 3 a. Structural comparison between the two determined structures for the IL 23:MA12 complex. The overall RMSD between the two
 Supplementary Figure 1. Biopanningg and clone enrichment of Alphabody binders against human IL 23. Positive clones in i phage ELISA with optical density (OD) 3 times higher than background are shown for
Supplementary Figure 1. Biopanningg and clone enrichment of Alphabody binders against human IL 23. Positive clones in i phage ELISA with optical density (OD) 3 times higher than background are shown for
Chapters 5-6 Enzymes. Catalyst: A substance that speeds up the rate of a chemical reaction but is not itself consumed.
 hapters 56 Enzymes atalyst: A substance that speeds up the rate of a chemical reaction but is not itself consumed. Most biological catalysts are proteins but some RA are catalysts too. e.g. Peptide bonds
hapters 56 Enzymes atalyst: A substance that speeds up the rate of a chemical reaction but is not itself consumed. Most biological catalysts are proteins but some RA are catalysts too. e.g. Peptide bonds
Chapter 6. Ground Rules Of Metabolism
 Chapter 6 Ground Rules Of Metabolism Alcohol Dehydrogenase An enzyme Breaks down ethanol and other toxic alcohols Allows humans to drink Metabolism Is the totality of an organism s chemical reactions Arises
Chapter 6 Ground Rules Of Metabolism Alcohol Dehydrogenase An enzyme Breaks down ethanol and other toxic alcohols Allows humans to drink Metabolism Is the totality of an organism s chemical reactions Arises
CHAPTER 8. An Introduction to Metabolism
 CHAPTER 8 An Introduction to Metabolism WHAT YOU NEED TO KNOW: Examples of endergonic and exergonic reactions. The key role of ATP in energy coupling. That enzymes work by lowering the energy of activation.
CHAPTER 8 An Introduction to Metabolism WHAT YOU NEED TO KNOW: Examples of endergonic and exergonic reactions. The key role of ATP in energy coupling. That enzymes work by lowering the energy of activation.
ing equilibrium i Dynamics? simulations on AchE and Implications for Edwin Kamau Protein Science (2008). 17: /29/08
 Induced-fit d or Pre-existin ing equilibrium i Dynamics? Lessons from Protein Crystallography yand MD simulations on AchE and Implications for Structure-based Drug De esign Xu Y. et al. Protein Science
Induced-fit d or Pre-existin ing equilibrium i Dynamics? Lessons from Protein Crystallography yand MD simulations on AchE and Implications for Structure-based Drug De esign Xu Y. et al. Protein Science
Lecture 11: Enzymes: Kinetics [PDF] Reading: Berg, Tymoczko & Stryer, Chapter 8, pp
![Lecture 11: Enzymes: Kinetics [PDF] Reading: Berg, Tymoczko & Stryer, Chapter 8, pp Lecture 11: Enzymes: Kinetics [PDF] Reading: Berg, Tymoczko & Stryer, Chapter 8, pp](/thumbs/74/70848503.jpg) Lecture 11: Enzymes: Kinetics [PDF] Reading: Berg, Tymoczko & Stryer, Chapter 8, pp. 216-225 Updated on: 2/4/07 at 9:00 pm Key Concepts Kinetics is the study of reaction rates. Study of enzyme kinetics
Lecture 11: Enzymes: Kinetics [PDF] Reading: Berg, Tymoczko & Stryer, Chapter 8, pp. 216-225 Updated on: 2/4/07 at 9:00 pm Key Concepts Kinetics is the study of reaction rates. Study of enzyme kinetics
COMBINATORIAL COMPOUND LIBRARIES FOR DRUG DISCOVERY: AN ONGOING CHALLENGE
 CMBIATRIA CMPUD IBRARIES FR DRUG DISCVER: A GIG CAEGE. Mario Geysen, Frank Schoenen, David Wagner and Richard Wagner Almost 2 years of combinatorial chemistry have emphasized the power of numbers, a key
CMBIATRIA CMPUD IBRARIES FR DRUG DISCVER: A GIG CAEGE. Mario Geysen, Frank Schoenen, David Wagner and Richard Wagner Almost 2 years of combinatorial chemistry have emphasized the power of numbers, a key
Abstract Process Economics Program Report 232 CHIRAL INTERMEDIATES (March 2001)
 Abstract Process Economics Program Report 232 CHIRAL INTERMEDIATES (March 2001) Chiral chemicals are a unique class of compounds that, although chemically identical, exist as mirror images of each other
Abstract Process Economics Program Report 232 CHIRAL INTERMEDIATES (March 2001) Chiral chemicals are a unique class of compounds that, although chemically identical, exist as mirror images of each other
CHEMISTRY (CHE) CHE 104 General Descriptive Chemistry II 3
 Chemistry (CHE) 1 CHEMISTRY (CHE) CHE 101 Introductory Chemistry 3 Survey of fundamentals of measurement, molecular structure, reactivity, and organic chemistry; applications to textiles, environmental,
Chemistry (CHE) 1 CHEMISTRY (CHE) CHE 101 Introductory Chemistry 3 Survey of fundamentals of measurement, molecular structure, reactivity, and organic chemistry; applications to textiles, environmental,
4. What is the general expression Keq (the equilibrium constant) in terms of product and reactant concentration? tell us about the enzyme.
 Section 8 Enzyme Kinetics Pre-Activity Assignment 1. Produce a reading log for the sections in your text that discuss the Michaelis-Menten equation and including kcat. 2. Focus on the derivation of the
Section 8 Enzyme Kinetics Pre-Activity Assignment 1. Produce a reading log for the sections in your text that discuss the Michaelis-Menten equation and including kcat. 2. Focus on the derivation of the
THE UNIVERSITY OF MANITOBA. PAPER NO: _1_ LOCATION: 173 Robert Schultz Theatre PAGE NO: 1 of 5 DEPARTMENT & COURSE NO: CHEM / MBIO 2770 TIME: 1 HOUR
 THE UNIVERSITY OF MANITOBA 1 November 1, 2016 Mid-Term EXAMINATION PAPER NO: _1_ LOCATION: 173 Robert Schultz Theatre PAGE NO: 1 of 5 DEPARTMENT & COURSE NO: CHEM / MBIO 2770 TIME: 1 HOUR EXAMINATION:
THE UNIVERSITY OF MANITOBA 1 November 1, 2016 Mid-Term EXAMINATION PAPER NO: _1_ LOCATION: 173 Robert Schultz Theatre PAGE NO: 1 of 5 DEPARTMENT & COURSE NO: CHEM / MBIO 2770 TIME: 1 HOUR EXAMINATION:
Resins and solid phase anchors in the organic chemistry
 Resins and solid phase anchors in the organic chemistry Overview 1963: 1. Resins Merrifield used chloromethylated-nitrated copolymer of styrene and divinylbenene 1 st cross-linked polystyrene resinds bead
Resins and solid phase anchors in the organic chemistry Overview 1963: 1. Resins Merrifield used chloromethylated-nitrated copolymer of styrene and divinylbenene 1 st cross-linked polystyrene resinds bead
Solid phase peptide synthesis (SPPS)
 Tapio Nevalainen Drug synthesis II 2012 Solid phase peptide synthesis (SPPS) Solid phase peptide synthesis (SPPS) R. Bruce Merrifield, Professor at Rockefeller University, has been awarded the Nobel Prize
Tapio Nevalainen Drug synthesis II 2012 Solid phase peptide synthesis (SPPS) Solid phase peptide synthesis (SPPS) R. Bruce Merrifield, Professor at Rockefeller University, has been awarded the Nobel Prize
Introduction. OntoChem
 Introduction ntochem Providing drug discovery knowledge & small molecules... Supporting the task of medicinal chemistry Allows selecting best possible small molecule starting point From target to leads
Introduction ntochem Providing drug discovery knowledge & small molecules... Supporting the task of medicinal chemistry Allows selecting best possible small molecule starting point From target to leads
In silico pharmacology for drug discovery
 In silico pharmacology for drug discovery In silico drug design In silico methods can contribute to drug targets identification through application of bionformatics tools. Currently, the application of
In silico pharmacology for drug discovery In silico drug design In silico methods can contribute to drug targets identification through application of bionformatics tools. Currently, the application of
Theophylline (TH), the structure of which is presented below, is a bronchial-dilator used for the treatment of asthma.
 1 EXAM SCIETIIC CULTURE CEMISTRY PRBLEM 1: Theophylline (T), the structure of which is presented below, is a bronchial-dilator used for the treatment of asthma. 3 C 7 1 3 9 1.1 Structural study and acid-base
1 EXAM SCIETIIC CULTURE CEMISTRY PRBLEM 1: Theophylline (T), the structure of which is presented below, is a bronchial-dilator used for the treatment of asthma. 3 C 7 1 3 9 1.1 Structural study and acid-base
Final Chem 4511/6501 Spring 2011 May 5, 2011 b Name
 Key 1) [10 points] In RNA, G commonly forms a wobble pair with U. a) Draw a G-U wobble base pair, include riboses and 5 phosphates. b) Label the major groove and the minor groove. c) Label the atoms of
Key 1) [10 points] In RNA, G commonly forms a wobble pair with U. a) Draw a G-U wobble base pair, include riboses and 5 phosphates. b) Label the major groove and the minor groove. c) Label the atoms of
Study of Non-Covalent Complexes by ESI-MS. By Quinn Tays
 Study of Non-Covalent Complexes by ESI-MS By Quinn Tays History Overview Background Electrospray Ionization How it is used in study of noncovalent interactions Uses of the Technique Types of molecules
Study of Non-Covalent Complexes by ESI-MS By Quinn Tays History Overview Background Electrospray Ionization How it is used in study of noncovalent interactions Uses of the Technique Types of molecules
Energy and Cellular Metabolism
 1 Chapter 4 About This Chapter Energy and Cellular Metabolism 2 Energy in biological systems Chemical reactions Enzymes Metabolism Figure 4.1 Energy transfer in the environment Table 4.1 Properties of
1 Chapter 4 About This Chapter Energy and Cellular Metabolism 2 Energy in biological systems Chemical reactions Enzymes Metabolism Figure 4.1 Energy transfer in the environment Table 4.1 Properties of
BIOLOGY 10/11/2014. An Introduction to Metabolism. Outline. Overview: The Energy of Life
 8 An Introduction to Metabolism CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson Outline I. Forms of Energy II. Laws of Thermodynamics III. Energy and metabolism IV. ATP V. Enzymes
8 An Introduction to Metabolism CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson Outline I. Forms of Energy II. Laws of Thermodynamics III. Energy and metabolism IV. ATP V. Enzymes
CHEMISTRY Topic #4: Building Block Oriented Synthesis Fall 2017 Dr. Susan Findlay
 CHEMISTRY 4000 Topic #4: Building Block Oriented Synthesis Fall 2017 Dr. Susan Findlay When to Choose a Building Block Approach While all retrosynthetic analyses must end with identification of starting
CHEMISTRY 4000 Topic #4: Building Block Oriented Synthesis Fall 2017 Dr. Susan Findlay When to Choose a Building Block Approach While all retrosynthetic analyses must end with identification of starting
NH 2. Biochemistry I, Fall Term Sept 9, Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter
 Biochemistry I, Fall Term Sept 9, 2005 Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter 3.1-3.4. Key Terms: ptical Activity, Chirality Peptide bond Condensation reaction ydrolysis
Biochemistry I, Fall Term Sept 9, 2005 Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter 3.1-3.4. Key Terms: ptical Activity, Chirality Peptide bond Condensation reaction ydrolysis
Affinity labels for studying enzyme active sites. Irreversible Enzyme Inhibition. Inhibition of serine protease with DFP
 Irreversible Enzyme Inhibition Irreversible inhibitors form stable covalent bonds with the enzyme (e.g. alkylation or acylation of an active site side chain) There are many naturally-occurring and synthetic
Irreversible Enzyme Inhibition Irreversible inhibitors form stable covalent bonds with the enzyme (e.g. alkylation or acylation of an active site side chain) There are many naturally-occurring and synthetic
MITOCW enzyme_kinetics
 MITOCW enzyme_kinetics In beer and wine production, enzymes in yeast aid the conversion of sugar into ethanol. Enzymes are used in cheese-making to degrade proteins in milk, changing their solubility,
MITOCW enzyme_kinetics In beer and wine production, enzymes in yeast aid the conversion of sugar into ethanol. Enzymes are used in cheese-making to degrade proteins in milk, changing their solubility,
Under strongly acidic conditions at ph = 1 every functional group in phosphoserine that can pick up a proton, does.
 1. (48 pts) a. L-Phosphoserine is a modified amino acid that is generated in proteins by phosphorylation of serine residues. The amino acid side chain has two acidic protons, which exhibit different pk
1. (48 pts) a. L-Phosphoserine is a modified amino acid that is generated in proteins by phosphorylation of serine residues. The amino acid side chain has two acidic protons, which exhibit different pk
An Introduction to Metabolism. Chapter 8
 An Introduction to Metabolism Chapter 8 METABOLISM I. Introduction All of an organism s chemical reactions Thousands of reactions in a cell Example: digest starch use sugar for energy and to build new
An Introduction to Metabolism Chapter 8 METABOLISM I. Introduction All of an organism s chemical reactions Thousands of reactions in a cell Example: digest starch use sugar for energy and to build new
Metabolism and enzymes
 Metabolism and enzymes 4-11-16 What is a chemical reaction? A chemical reaction is a process that forms or breaks the chemical bonds that hold atoms together Chemical reactions convert one set of chemical
Metabolism and enzymes 4-11-16 What is a chemical reaction? A chemical reaction is a process that forms or breaks the chemical bonds that hold atoms together Chemical reactions convert one set of chemical
Answers to Chapter 4 (in-text & asterisked problems)
 Introduction to ioganic hemistry and hemical iology 1 nswers to hapter 4 (intext & asterisked problems) nswer 41 The bulky tertbutyl group has a strong equatial preference, the following two confmations
Introduction to ioganic hemistry and hemical iology 1 nswers to hapter 4 (intext & asterisked problems) nswer 41 The bulky tertbutyl group has a strong equatial preference, the following two confmations
A Plausible Model Correlates Prebiotic Peptide Synthesis with. Primordial Genetic Code
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 A Plausible Model Correlates Prebiotic Peptide Synthesis with Primordial Genetic Code Jianxi Ying,
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 A Plausible Model Correlates Prebiotic Peptide Synthesis with Primordial Genetic Code Jianxi Ying,
Bioengineering & Bioinformatics Summer Institute, Dept. Computational Biology, University of Pittsburgh, PGH, PA
 Pharmacophore Model Development for the Identification of Novel Acetylcholinesterase Inhibitors Edwin Kamau Dept Chem & Biochem Kennesa State Uni ersit Kennesa GA 30144 Dept. Chem. & Biochem. Kennesaw
Pharmacophore Model Development for the Identification of Novel Acetylcholinesterase Inhibitors Edwin Kamau Dept Chem & Biochem Kennesa State Uni ersit Kennesa GA 30144 Dept. Chem. & Biochem. Kennesaw
Chapter 5 Volume 2 Operation of enzymes- The nutcracker mechanism *
 Chapter 5, Vol. 2, Operation of enzymes 2-5-1 Chapter 5 Volume 2 Operation of enzymes- The nutcracker mechanism * Fortunately Dorovska-Taran and Martnek with their respective coworkers measured the steady-state
Chapter 5, Vol. 2, Operation of enzymes 2-5-1 Chapter 5 Volume 2 Operation of enzymes- The nutcracker mechanism * Fortunately Dorovska-Taran and Martnek with their respective coworkers measured the steady-state
Solutions In each case, the chirality center has the R configuration
 CAPTER 25 669 Solutions 25.1. In each case, the chirality center has the R configuration. C C 2 2 C 3 C(C 3 ) 2 D-Alanine D-Valine 25.2. 2 2 S 2 d) 2 25.3. Pro,, Trp, Tyr, and is, Trp, Tyr, and is Arg,
CAPTER 25 669 Solutions 25.1. In each case, the chirality center has the R configuration. C C 2 2 C 3 C(C 3 ) 2 D-Alanine D-Valine 25.2. 2 2 S 2 d) 2 25.3. Pro,, Trp, Tyr, and is, Trp, Tyr, and is Arg,
Lecture 15: Enzymes & Kinetics. Mechanisms ROLE OF THE TRANSITION STATE. H-O-H + Cl - H-O δ- H Cl δ- HO - + H-Cl. Margaret A. Daugherty.
 Lecture 15: Enzymes & Kinetics Mechanisms Margaret A. Daugherty Fall 2004 ROLE OF THE TRANSITION STATE Consider the reaction: H-O-H + Cl - H-O δ- H Cl δ- HO - + H-Cl Reactants Transition state Products
Lecture 15: Enzymes & Kinetics Mechanisms Margaret A. Daugherty Fall 2004 ROLE OF THE TRANSITION STATE Consider the reaction: H-O-H + Cl - H-O δ- H Cl δ- HO - + H-Cl Reactants Transition state Products
Fluorine in Peptide and Protein Engineering
 Fluorine in Peptide and Protein Engineering Rita Fernandes Porto, February 11 th 2016 Supervisor: Prof. Dr. Beate Koksch 1 Fluorine a unique element for molecule design The most abundant halogen in earth
Fluorine in Peptide and Protein Engineering Rita Fernandes Porto, February 11 th 2016 Supervisor: Prof. Dr. Beate Koksch 1 Fluorine a unique element for molecule design The most abundant halogen in earth
UNIT 6 PART 3 *REGULATION USING OPERONS* Hillis Textbook, CH 11
 UNIT 6 PART 3 *REGULATION USING OPERONS* Hillis Textbook, CH 11 REVIEW: Signals that Start and Stop Transcription and Translation BUT, HOW DO CELLS CONTROL WHICH GENES ARE EXPRESSED AND WHEN? First of
UNIT 6 PART 3 *REGULATION USING OPERONS* Hillis Textbook, CH 11 REVIEW: Signals that Start and Stop Transcription and Translation BUT, HOW DO CELLS CONTROL WHICH GENES ARE EXPRESSED AND WHEN? First of
FRAUNHOFER IME SCREENINGPORT
 FRAUNHOFER IME SCREENINGPORT Design of screening projects General remarks Introduction Screening is done to identify new chemical substances against molecular mechanisms of a disease It is a question of
FRAUNHOFER IME SCREENINGPORT Design of screening projects General remarks Introduction Screening is done to identify new chemical substances against molecular mechanisms of a disease It is a question of
Computational Biology 1
 Computational Biology 1 Protein Function & nzyme inetics Guna Rajagopal, Bioinformatics Institute, guna@bii.a-star.edu.sg References : Molecular Biology of the Cell, 4 th d. Alberts et. al. Pg. 129 190
Computational Biology 1 Protein Function & nzyme inetics Guna Rajagopal, Bioinformatics Institute, guna@bii.a-star.edu.sg References : Molecular Biology of the Cell, 4 th d. Alberts et. al. Pg. 129 190
Lecture # 3, 4 Selecting a Catalyst (Non-Kinetic Parameters), Review of Enzyme Kinetics, Selectivity, ph and Temperature Effects
 1.492 - Integrated Chemical Engineering (ICE Topics: Biocatalysis MIT Chemical Engineering Department Instructor: Professor Kristala Prather Fall 24 Lecture # 3, 4 Selecting a Catalyst (Non-Kinetic Parameters,
1.492 - Integrated Chemical Engineering (ICE Topics: Biocatalysis MIT Chemical Engineering Department Instructor: Professor Kristala Prather Fall 24 Lecture # 3, 4 Selecting a Catalyst (Non-Kinetic Parameters,
Homogeneous Catalysis - B. List
 omogeneous Catalysis - B. List 2.2.2 Research Area "rganocatalytic Asymmetric α-alkylation of Aldehydes" (B. List) Involved:. Vignola, A. Majeed Seayad bjective: α-alkylations of carbonyl compounds are
omogeneous Catalysis - B. List 2.2.2 Research Area "rganocatalytic Asymmetric α-alkylation of Aldehydes" (B. List) Involved:. Vignola, A. Majeed Seayad bjective: α-alkylations of carbonyl compounds are
Chem 250 Evening Exam 2
 Page 1 of 10 Evening Exam 2 ame:: Chem 250 Evening Exam 2 This exam is composed of 40 questions. As discussed in the course syllabus, honesty and integrity are absolute essentials for this class. In fairness
Page 1 of 10 Evening Exam 2 ame:: Chem 250 Evening Exam 2 This exam is composed of 40 questions. As discussed in the course syllabus, honesty and integrity are absolute essentials for this class. In fairness
MEDCHEM 562 Kent Kunze Lecture 2. Physicochemical Properties of Drugs and Drug Disposition. Stereochemistry (Double the trouble. or double the fun?
 12 MEDCHEM 562 Kent Kunze Lecture 2 Physicochemical Properties of Drugs and Drug Disposition Stereochemistry (Double the trouble. or double the fun?) The vast majority of drugs contain at least one stereo-center
12 MEDCHEM 562 Kent Kunze Lecture 2 Physicochemical Properties of Drugs and Drug Disposition Stereochemistry (Double the trouble. or double the fun?) The vast majority of drugs contain at least one stereo-center
MEDCHEM 562 Kent Kunze Lecture 2. Physicochemical Properties of Drugs and Drug Disposition. Stereochemistry (Double the trouble. or double the fun?
 15 MEDCHEM 562 Kent Kunze Lecture 2 Physicochemical Properties of Drugs and Drug Disposition Stereochemistry (Double the trouble. or double the fun?) The vast majority of drugs contain at least one stereo-center
15 MEDCHEM 562 Kent Kunze Lecture 2 Physicochemical Properties of Drugs and Drug Disposition Stereochemistry (Double the trouble. or double the fun?) The vast majority of drugs contain at least one stereo-center
Chapter 4: Amino Acids
 Chapter 4: Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. lipid polysaccharide enzyme 1940s 1980s. Lipids membrane 1960s. Polysaccharide Are energy metabolites and many of
Chapter 4: Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. lipid polysaccharide enzyme 1940s 1980s. Lipids membrane 1960s. Polysaccharide Are energy metabolites and many of
1. Draw the structure of oxazolone formed upon activation of N-Acetylvaline
 Peptide quiz 1 (5 questions for 10 minutes) 10 points max 1. Draw the structure of oxazolone formed upon activation of -Acetylvaline 3 C 3 C 2. The following DKP (1) is prepared by cyclisation of dipeptide
Peptide quiz 1 (5 questions for 10 minutes) 10 points max 1. Draw the structure of oxazolone formed upon activation of -Acetylvaline 3 C 3 C 2. The following DKP (1) is prepared by cyclisation of dipeptide
7.012 Problem Set 1 Solutions
 ame TA Section 7.012 Problem Set 1 Solutions Your answers to this problem set must be inserted into the large wooden box on wheels outside 68120 by 4:30 PM, Thursday, September 15. Problem sets will not
ame TA Section 7.012 Problem Set 1 Solutions Your answers to this problem set must be inserted into the large wooden box on wheels outside 68120 by 4:30 PM, Thursday, September 15. Problem sets will not
Problem Set 7: Stereochemistry-ANSWER KEY
 Problem Set 7: Stereochemistry-ANSWER KEY Chemistry 260 Organic Chemistry 1. The answer is (2). Circled isomers have a stereogenic carbon (*) and hence a stereogenic centre. C 2 C 2 C 2 C 2 C 2 C 2 C 2
Problem Set 7: Stereochemistry-ANSWER KEY Chemistry 260 Organic Chemistry 1. The answer is (2). Circled isomers have a stereogenic carbon (*) and hence a stereogenic centre. C 2 C 2 C 2 C 2 C 2 C 2 C 2
Life Sciences 1a Lecture Slides Set 10 Fall Prof. David R. Liu. Lecture Readings. Required: Lecture Notes McMurray p , O NH
 Life ciences 1a Lecture lides et 10 Fall 2006-2007 Prof. David R. Liu Lectures 17-18: The molecular basis of drug-protein binding: IV protease inhibitors 1. Drug development and its impact on IV-infected
Life ciences 1a Lecture lides et 10 Fall 2006-2007 Prof. David R. Liu Lectures 17-18: The molecular basis of drug-protein binding: IV protease inhibitors 1. Drug development and its impact on IV-infected
Biochemistry 3300 Problems (and Solutions) Metabolism I
 (1) Provide a reasonable systematic name for an enzyme that catalyzes the following reaction: fructose + ATP > fructose-1 phosphate + ADP (2) The IUBMB has a developed a set of rules for classifying enzymes
(1) Provide a reasonable systematic name for an enzyme that catalyzes the following reaction: fructose + ATP > fructose-1 phosphate + ADP (2) The IUBMB has a developed a set of rules for classifying enzymes
