Investigation of M1 transitions of the ground-state configuration of In-like Tungsten
|
|
|
- Winfred Freeman
- 5 years ago
- Views:
Transcription
1 Investigation of M1 transitions of the ground-state configuration of In-like Tungsten W Li 1,2,3, J Xiao 1,2, Z Shi 1,2a, Z Fei 1,2b, R Zhao 1,2c, T Brage 3, S Huldt, R Hutton 1, 2 * and Y Zou 1,2 * 1 The Key lab of Applied Ion Beam Physics, Ministry of Education, China 2 Shanghai EBIT laboratory, Modern physics institute, Fudan University, Shanghai, China 3 Division of Mathematical Physics, Department of Physics, Lund University, Sweden Lund Observatory, Lund University, Sweden Corresponding author: * rhutton@fudan.edu.cn, zouym@fudan.edu.cn Abstract. Three visible lines of M1 transitions from In-like W were recorded using the Shanghai permanent magnet electron beam ion trap. The experimental wavelengths were measured as 93.8±0.15, ±0.13 and ±0.23 nm (vacuum wavelengths). These results are in good agreement with theoretical predictions obtained using large-scale Relativistic Many-Body Perturbation Theory, in the form of the Flexible Atomic Code. 1. Introduction a present address: Beijing Synchrotron Radiation Facility, Institute of High Energy Physics, Chinese Academy of Sciences, Beijing 10009, China b present address: Shanghai Institute of Applied Physics, Chinese Academy of Sciences, Shanghai , China c present address: Department of Radiotherapy, Shanghai Pulmonary Hospital, Tongji University School of Medicine, 20033, China Keywords: visible spectroscopy, tungsten, EBIT, metastable PACS number(s): Jc, Fw, aj There is a large demand for atomic data of different charge states of Tungsten, since it is considered as a strong candidate for coating material in the International Tokomak Experimental Reactor (ITER), especially in the divertor region. This is due to its excellent thermomechanical properties and its very low erosion under various physical and chemical conditions [1, 2]. Unfortunately, there is very little spectroscopic data available for tungsten in the charge states between W 6+ and W 28+ [3], which are important in the divertor region, where the electron temperature will be considerably lower than in the core plasma. The need for visible spectral lines from tungsten ions in all charge states for ITER diagnostics was pointed out by Skinner in 2006 []. Recently we have focused on the charge states of interest in the divertor region of ITER (W 6+ to W 28+ ) [5-10]. For W 25+, W 26+ and W 27+, which are systems with few f-electrons (between one and three), most visible lines will originate from magnetic dipole (M1) transitions between fine structure levels of the ground state configurations. W 27+ has a very simple ground state configuration consisting of only two levels, namely f 2 F5/2 and 2 F7/2 and just one
2 M1 transition, which we recently identified and analysed [5]. W 26+ is more complex having a f 2 ground state configuration. We reported in [6] on our identification of seven transitions between its levels, which made it possible to determine the excitation energy of seven of the thirteen levels in this configuration. The good agreement between theoretical and experimental values for W 27+ and W 26+ lends a strong support and confidence to our method for dealing with the few f-electron systems. In the work presented here we continue along the lines laid down in the earlier works [5, 6] and study M1 forbidden lines from W 25+, which is more complex having a ground configuration of f 3 with 1 fine structure levels. 2. Experiment The spectra were recorded using the Shanghai permanent magnet electron beam ion trap (SH-PermEBIT) [11, 12]. Tungsten was injected by the volatile compound W(CO)6 and collided with the electron beam in the drift tube of the EBIT to produce the highly charged ions W q+. The photons emitted from the trapped ions were focused by a biconvex lens to be recorded by an Andor SR-303i spectrometer equipped with a 1200 lines/mm grating and an Andor Newton CCD camera (Andor DU90P-BU2). Details of the experimental arrangement can be found in Ref. [5, 6] where we report on forbidden-line spectroscopy of W 27+ and W 26+, respectively. Similar to the method employed for W 26+, the W 25+ charge state was identified relying on the identification of the W 26+ and W 27+ tungsten ions in Ref [5, 6], namely, the first set of lines to appear as the beam energy was lowered from that required to produce the W 26+ ions were identified as being from the W 25+ ions. The identifications were also supported by the charge state being in accordance to the ionization potential and all three lines showing the same intensity dependence on the electron beam energy. The wavelength was calibrated by using lines from either a Hg lamp or a Fe hollow cathode lamp or the lines from background carbon and oxygen. 3. Calculational method For W 27+ and W 26+ ions, we found excellent agreement between our experimental results and calculations using both Multiconfiguration Dirac-Hartree-Fock method, in the form of the GRASP2K code [13], as well as Relativistic Many-Body Perturbation Theory, utilizing the RMBPT option of the FAC code [1]. It is clear that the RMBPT method is very well-suited for these isolated ground state configurations of highly ionized systems and in general this method converges faster than GRASP2K. The identification of these lines was again aided by calculations using both the GRASP2K [13] and the FAC [1] codes, but we focused mainly on the latter and only used GRASP2K to support the identifications. In the RMBPT approach, the Hilbert space of the full Hamiltonian is partitioned into two sub-spaces, labeled M and N. In the present work, the model space M contains only the d 10 f 3 ground configuration where correlation is included to all orders
3 through a configuration interaction (CI) expansion. The N sub-space contains the remaining configurations that are excited by single or double excitations from the M space whose contribution to correlation will be included to second-order perturbation theory. Detailed information about the theoretical method is given in Refs. [1, 15] and can also be found in our previous publications [6, 7, 10]. In order to investigate the convergence of the calculations, we define two maximal principal quantum numbers for the N sub-space: n1 for one-electron excitation and the first electron of the double excitations, and n2 for the second electron of the double excitations. We increase n1 and n2 step-by-step, respectively, to achieve convergence. First, we define n2 = 20 and increase n1 in steps of 10 from 20 to n1max, where the results have converged. Second, we set n1 = n1max and increase n2 in steps of 10 in the range 20 to n2max, again with converged results. The maximum orbital angular quantum number was always set to lmax = 15. Figure 1. Relative convergence of the lowest ten energy levels as the size of maximum principal quantum numbers of the virtual states in the N space (see text). δe is the difference in percentages of energy from the previous N space set. (a) n2max = 20 and increase n1max in steps of 10 from 20 to 70. (b) n1max = 70 and increase n2max in steps of 10 in the range 20 to 60. The y-axis is given in logarithmic scale.. Results and Discussions
4 Figure 2. A partial energy level diagram of In-like tungsten. The energy levels are taken from our RMBPT calculations. There are 1 energy levels in the ground state configuration of In-like tungsten where the highest levels are close to 28 ev. For convenience we only show levels up to 12 ev as the higher energy levels will not contribute to observable spectral lines. Solid (red) lines represent the M1 emission lines observed in this work Figure 1 shows the convergence study for the lowest ten excitation energies as a function of n1max and n2max, respectively, using the RMBPT method. In the first case, figure 1 (a), it shows the relative convergence with n1max=70 the excitation energy is less than 0.01%. For the second set of calculations, figure 1 (b) shows that for n2max=60 the convergence is close to 0.01% for I11/2 and to % for F3/2. The results are therefore well converged and the corresponding partial energy-level diagram is shown in figure 2, where the levels are ordered by their J values and shown with different colors (online). The three M1 emission lines observed in this work are represented by solid (red) lines, see below. The spectra were recorded for electron beam energies of 730 ev, 780 ev, 830 ev, 880 ev, 1100 ev, and 1180 ev and some of the recorded spectra are shown in figure 3. In these spectra, two lines are marked with arrows (red on-line). The line on the right hand side was recently identified as a W 26+ line [6,16]. The line at 93.8 ± 0.15 nm, is seen to appear at a lower electron beam energy and just above the ionization potential of W 2+ and is therefore assumed to be from W 25+. The experimental wavelength agrees with the prediction from our RMBPT calculation for the W 25+ ( I11/2 I9/2) line: nm. Table 1 shows the comparison of our measured and calculated wavelengths for the three W 25+ lines studied in this work. The wavelengths of the observed lines are transformed from air to vacuum values using the formula given by Morton [17]. We also compare our data to the calculated results by Radtke et al. using the HULLAC code [18]. The RMBPT calculation results show better agreement with the experimental values where the discrepancy was found to be 0.31%, 0.01% and 0.89%, respectively.
5 Table 1. Computed rates (A), in s -1, from present FAC-code and Wavelength (λ), in nm, for three different transitions in W 25+, from calculations using the FAC (present calculations) and the HULLAC [18] codes and observed in the SH-PermEBIT. Transition A (s -1 ) λrmbpt (nm) λobserved (nm) λhullac (nm) I11/2 I9/ ± I13/2 I11/ ± H9/2 I9/ ± Figure 3. Visible spectra of tungsten obtained at the SH-PermEBIT, for different beam energies. The left line at 93.8 nm marked by an arrow is the I11/2 I9/2 transition in W 25+ while the right one, also marked by an arrow, is the 3 F3 3 F2 transition in W 26+. For all spectra shown here the beam current was about 5 ma. As we pointed out above, W 25+ has a complicated f 3 ground configuration with the 1 energy levels shown in figure 2. It might therefore at first glance be surprising that not very many lines are observed. There are however three reasons for the lack of observed spectral lines referring to figure 2. First, since there are many levels in this configuration, the excited levels can decay to several different lower levels, leading to possible low intensities in any single branch. Second, the ground state has a high J-value of 9/2 and therefore low-lying levels with total angular momentum of either 3/2 or 5/2 cannot decay to it by M1 transitions. As an example the F3/2 can only decay radiatively via a high order forbidden transition giving a lifetime in the order of years. This type of level will then decay via collisions, even in very dilute plasma as in an EBIT. In table 2, we give the lowest ten excitation energies and their corresponding
6 computed radiative lifetime. A third reason for the lack of lines in our spectra is that other levels can decay to the lowest J = 9/2 level, but will produce lines outside the range of our spectrometer. Table 2. Excitation energies E, lifetime τ of the lowest ten energy levels of the f 3 ground state configuration of In-like W from our RMBPT calculation. The lifetime of F3/2 is confirmed by using the MCDHF calculation and big scale Relativistic Configuration Interaction (RCI) calculation. Label E (cm -1 ) τ (ms) Label E(cm -1 ) τ (ms) I9/2 0 F5/ I11/ I15/ F3/ yr S3/ I13/ F7/ H9/ G5/ Conclusion In conclusion, the SH-PermEBIT operates very well in the required electron energy range, and three M1 spectral lines of In-like W have been observed for the first time. The wavelengths were all in excellent agreement (The discrepancies were within 1%.) with our RMBPT result which lends strong support to the identifications. One of these three M1 transitions the I11/2 I9/2, is very strong and may be a good candidate to use in ITER diagnostics in the visible wavelength range. Acknowledgements This work was supported by the Chinese National Fusion Project for ITER No. 2015GB117000, Shanghai Leading Academic Discipline Project No. B107 as well as the Swedish Science Council (Vetenskapsrådet). We would like to thank Professor Gordon Berry for carefully reading the manuscript and for offering very useful comments. We are grateful to the Nordic Center at Fudan University for continued support to our exchange between Fudan and Lund Universities. References [1] Phillips V 2006 Phys. Scr. T [2] Pütterich T, Neu R, Biedermann C, Radtke R and ASDEX Upgrade Team 2005 J. Phys. B: At. Mol. Opt. Phys [3] Kramida A E and Shirai T 2009 At. Data Nucl. Data Tables [] Skinner C H 2008 Can. J. Phys. 86(1) 285 [5] Fei Z, Zhao R, Shi Z, Xiao J, Qiu M, Grumer J, Andersson M, Brage T, Hutton R,
7 and Zou Y 2012 Phys. Rev. A [6] Fei Z, Li W, Du W, Shi Z, Zhao R, Xiao J, Brage T, Huldt S, Hutton R and Zou Y 201 Phys. Rev. A [7]Qiu M et al 201 J. Phys. B: At. Mol. Opt. Phys. 7, [8] Zhao Z, Qiu M, Zhao R, Li W, Guo X, Xiao J, Zou Y and Hutton R 2015 J. Phys. B: At. Mol. Opt. Phys [9] Li W, Shi Z, Yang Y, Xiao J, Brage T, Hutton R and Zou Y 2015 Phys. Rev. A [10] Qiu M, Li W, Zhao Z, Yang Y, Xiao J, Brage T, Hutton R and Zou Y 2015 J. Phys. B: At. Mol. Opt. Phys [11] Xiao J, Fei Z, Jin X, Lu D, Shen Y, Liljeby L, Hutton R, Zou Y 2011 Phys. Scr., T [12] Xiao J, Fei Z, Yang Y, Jin X, Lu D, Shen Y, Liljeby L, Hutton R, and Zou Y 2012 Rev. Sci. Instrum [13] Jönsson P, He X, Fischer C F and Grant I P 2007 Comput. Phys. Commun [1] Gu M F 2008 Can. J. Phys [15] Gu M F, Tomer H, Ehud B, and Steven M K 2006 Astrophys. J [16] Ding X B, Koike F, Murakami I, Kato D, Sakaue H A, Dong C Z, Nakamura N, Komatsu A and Sakoda J 2011 J. Phys. B: At. Mol. Opt. Phys 1500 [17] Morton D C 1991 Astrophys. J. Suppl. S [18] Radtke R, Biedermann C, Fussmann G, Schwob J L, Mandelbaum P, and Doron R 2007 At Plas Mater Interac Data Fusion 13 5
W 25+ More Complex, W 25+, which is In-like. There are now 41 fine structure levels belonging to the ground state,
 W 25+ More Complex, W 25+, which is In-like. There are now 41 fine structure levels belonging to the ground state, A partial energy level diagram of In-like tungsten. The energy levels are taken from our
W 25+ More Complex, W 25+, which is In-like. There are now 41 fine structure levels belonging to the ground state, A partial energy level diagram of In-like tungsten. The energy levels are taken from our
Trapped in Shanghai Spectroscopy with the Shanghai Electron Beam Ion Traps
 Trapped in Shanghai Spectroscopy with the Shanghai Electron Beam Ion Traps Roger Hutton! "#on behalf of the Shanghai EBIT laboratory Modern Physics Institute, Fudan University, Shanghai China Outline Where
Trapped in Shanghai Spectroscopy with the Shanghai Electron Beam Ion Traps Roger Hutton! "#on behalf of the Shanghai EBIT laboratory Modern Physics Institute, Fudan University, Shanghai China Outline Where
Energy levels and radiative data for Kr-like W 38+ from MCDHF and RMBPT calculations
 Energy levels and radiative data for Kr-like W 8+ from MCDHF and RMBPT calculations XueLing Guo 1,2,, Jon Grumer 1, Tomas Brage 1, Ran Si 2,, ChongYang Chen 2,, Per Jönsson 4, Kai Wang 5,6, Jun Yan 6,7,
Energy levels and radiative data for Kr-like W 8+ from MCDHF and RMBPT calculations XueLing Guo 1,2,, Jon Grumer 1, Tomas Brage 1, Ran Si 2,, ChongYang Chen 2,, Per Jönsson 4, Kai Wang 5,6, Jun Yan 6,7,
arxiv: v1 [physics.atom-ph] 2 Dec 2015
![arxiv: v1 [physics.atom-ph] 2 Dec 2015 arxiv: v1 [physics.atom-ph] 2 Dec 2015](/thumbs/94/118268126.jpg) J. Phys. B: At. Mol. Opt. Phys. arxiv:1512.657v1 [physics.atom-ph] 2 Dec 215 Theoretical investigation of spectroscopic properties of W 26+ in EBIT plasma V. Jonauskas, A. Kynienė, P. Rynkun, S. Kučas,
J. Phys. B: At. Mol. Opt. Phys. arxiv:1512.657v1 [physics.atom-ph] 2 Dec 215 Theoretical investigation of spectroscopic properties of W 26+ in EBIT plasma V. Jonauskas, A. Kynienė, P. Rynkun, S. Kučas,
NIST Research on Spectroscopy and Collisional-Radiative Modeling of Highly-Charged Ions of Tungsten
 NIST Research on Spectroscopy and Collisional-Radiative Modeling of Highly-Charged Ions of Tungsten Yuri Ralchenko National Institute of Standards and Technology Gaithersburg, USA Vienna, Austria, Dec
NIST Research on Spectroscopy and Collisional-Radiative Modeling of Highly-Charged Ions of Tungsten Yuri Ralchenko National Institute of Standards and Technology Gaithersburg, USA Vienna, Austria, Dec
An import year!
 1759.. An import year! Some Historic and Current Aspects of Plasma Diagnostics Using Atomic Spectroscopy Roger Hutton and Yaming Zou Shanghai Electron Beam Ion Trap Laboratory Modern Physics Institute,
1759.. An import year! Some Historic and Current Aspects of Plasma Diagnostics Using Atomic Spectroscopy Roger Hutton and Yaming Zou Shanghai Electron Beam Ion Trap Laboratory Modern Physics Institute,
EUV spectra from the NIST EBIT
 EUV spectra from the NIST EBIT D. Kilbane and G. O Sullivan Atomic and Molecular Plasma Physics group, UCD, Ireland J. D. Gillaspy, Yu. Ralchenko and J. Reader National Institute of Standards and Technology,
EUV spectra from the NIST EBIT D. Kilbane and G. O Sullivan Atomic and Molecular Plasma Physics group, UCD, Ireland J. D. Gillaspy, Yu. Ralchenko and J. Reader National Institute of Standards and Technology,
Direct Observation of the M1 Transition between the Ground Term Fine Structure Levels of W VIII
 Article Direct Observation of the M1 Transition between the Ground Term Fine Structure Levels of W VIII Momoe Mita 1, Hiroyuki A. Sakaue 2, Daiji Kato 2,3, Izumi Murakami 2,3 and Nobuyuki Nakamura 1, *
Article Direct Observation of the M1 Transition between the Ground Term Fine Structure Levels of W VIII Momoe Mita 1, Hiroyuki A. Sakaue 2, Daiji Kato 2,3, Izumi Murakami 2,3 and Nobuyuki Nakamura 1, *
Extreme ultraviolet spectroscopy of highly charged argon ions at the Berlin EBIT
 Extreme ultraviolet spectroscopy of highly charged argon ions at the Berlin EBIT C. Biedermann, R. Radtke, G. Fussmann, F.I. Allen Institut für Physik der Humboldt-Universität zu Berlin, Lehrstuhl Plasmaphysik,
Extreme ultraviolet spectroscopy of highly charged argon ions at the Berlin EBIT C. Biedermann, R. Radtke, G. Fussmann, F.I. Allen Institut für Physik der Humboldt-Universität zu Berlin, Lehrstuhl Plasmaphysik,
Cascade emission in electron beam ion trap plasma of W 25+ ion
 2 3 4 5 6 7 8 Cascade emission in electron beam ion trap plasma of W 25+ ion V. Jonauskas a,, T. Pütterich b, S. Kučas a, Š. Masysa, A. Kynienė a, G. Gaigalas a, R. Kisielius a, L. Radžiūtė a, P. Rynkun
2 3 4 5 6 7 8 Cascade emission in electron beam ion trap plasma of W 25+ ion V. Jonauskas a,, T. Pütterich b, S. Kučas a, Š. Masysa, A. Kynienė a, G. Gaigalas a, R. Kisielius a, L. Radžiūtė a, P. Rynkun
The Extreme Ultraviolet Emissions of W 23+ (4f 5 )
 The Extreme Ultraviolet Emissions of W 23+ (4f 5 ) T Pütterich, V. Jonauskas, R Neu, R Dux and ASDEX Upgrade Team Max Planck Institut für Plasmaphysik, EURATOM Association, D 85748 Garching, Germany Institute
The Extreme Ultraviolet Emissions of W 23+ (4f 5 ) T Pütterich, V. Jonauskas, R Neu, R Dux and ASDEX Upgrade Team Max Planck Institut für Plasmaphysik, EURATOM Association, D 85748 Garching, Germany Institute
Energy levels and radiative rates for Ne-like ions from Cu to Ga
 Pramana J. Phys. (2017) 89:79 DOI 10.1007/s12043-017-1469-x Indian Academy of Sciences Energy levels and radiative rates for Ne-like ions from Cu to Ga NARENDRA SINGH and SUNNY AGGARWAL Department of Physics,
Pramana J. Phys. (2017) 89:79 DOI 10.1007/s12043-017-1469-x Indian Academy of Sciences Energy levels and radiative rates for Ne-like ions from Cu to Ga NARENDRA SINGH and SUNNY AGGARWAL Department of Physics,
Observation of Tungsten Line Emissions in Wavelength Range of Å in Large Helical Device )
 Observation of Tungsten Line Emissions in Wavelength Range of 10-500 Å in Large Helical Device ) Yang LIU 1), Shigeru MORITA 1,2), Tetsutarou OISHI 1,2), Motoshi GOTO 1,2) and Xianli HUANG 2) 1) Department
Observation of Tungsten Line Emissions in Wavelength Range of 10-500 Å in Large Helical Device ) Yang LIU 1), Shigeru MORITA 1,2), Tetsutarou OISHI 1,2), Motoshi GOTO 1,2) and Xianli HUANG 2) 1) Department
Relativistic Calculations for Be-like Iron
 Commun. Theor. Phys. (Beijing, China) 50 (2008) pp. 468 472 Chinese Physical Society Vol. 50, No. 2, August 15, 2008 Relativistic Calculations for Be-like Iron YANG Jian-Hui, 1 LI Ping, 2, ZHANG Jian-Ping,
Commun. Theor. Phys. (Beijing, China) 50 (2008) pp. 468 472 Chinese Physical Society Vol. 50, No. 2, August 15, 2008 Relativistic Calculations for Be-like Iron YANG Jian-Hui, 1 LI Ping, 2, ZHANG Jian-Ping,
Plasma-Related Atomic Physics with an Electron Beam Ion Trap
 Plasma-Related Atomic Physics with an Electron Beam Ion Trap Nobuyuki NAKAMURA Institute for Laser Science, The University of Electro-Communications, Tokyo 182-8585, Japan (Received 20 May 2013 / Accepted
Plasma-Related Atomic Physics with an Electron Beam Ion Trap Nobuyuki NAKAMURA Institute for Laser Science, The University of Electro-Communications, Tokyo 182-8585, Japan (Received 20 May 2013 / Accepted
Evaluation and Comparison of the Configuration Interaction Calculations for Complex Atoms
 Atoms 2014, 2, 1-14; doi:10.3390/atoms2010001 OPEN ACCESS atoms ISSN 2218-2004 www.mdpi.com/journal/atoms Article Evaluation and Comparison of the Configuration Interaction Calculations for Complex Atoms
Atoms 2014, 2, 1-14; doi:10.3390/atoms2010001 OPEN ACCESS atoms ISSN 2218-2004 www.mdpi.com/journal/atoms Article Evaluation and Comparison of the Configuration Interaction Calculations for Complex Atoms
Atomic Data for Lowly-Charged Tungsten Ions
 Atomic Data for Lowly-Charged Tungsten Ions Patrick Palmeri patrick.palmeri@umons.ac.be ADAS 2012 (Cadarache, France) 1 Outline Introduction HFR+CPOL Method Results: W 0, W 3-5+ Conclusions & Perspectives
Atomic Data for Lowly-Charged Tungsten Ions Patrick Palmeri patrick.palmeri@umons.ac.be ADAS 2012 (Cadarache, France) 1 Outline Introduction HFR+CPOL Method Results: W 0, W 3-5+ Conclusions & Perspectives
This work was performed under the auspices of the U.S. Department of Energy by Lawrence Livermore National Laboratory under contract
 This work was performed under the auspices of the U.S. Department of Energy by under contract DE-AC52-7NA27344. Lawrence Livermore National Security, LLC The ITER tokamak Tungsten (W) is attractive as
This work was performed under the auspices of the U.S. Department of Energy by under contract DE-AC52-7NA27344. Lawrence Livermore National Security, LLC The ITER tokamak Tungsten (W) is attractive as
Fine Structure Calculations of Atomic Data for Ar XVI
 Journal of Modern Physics, 2015, 6, 1609-1630 Published Online September 2015 in SciRes. http://www.scirp.org/journal/jmp http://dx.doi.org/10.4236/jmp.2015.611163 Fine Structure Calculations of Atomic
Journal of Modern Physics, 2015, 6, 1609-1630 Published Online September 2015 in SciRes. http://www.scirp.org/journal/jmp http://dx.doi.org/10.4236/jmp.2015.611163 Fine Structure Calculations of Atomic
arxiv: v2 [physics.atom-ph] 10 Oct 2013
![arxiv: v2 [physics.atom-ph] 10 Oct 2013 arxiv: v2 [physics.atom-ph] 10 Oct 2013](/thumbs/94/118719103.jpg) Cascade emission in electron beam ion trap plasma Valdas Jonauskas a, Šarūnas Masysa, Aušra Kynienė a, Gediminas Gaigalas a arxiv:1210.5742v2 [physics.atom-ph] 10 Oct 2013 a Institute of Theoretical Physics
Cascade emission in electron beam ion trap plasma Valdas Jonauskas a, Šarūnas Masysa, Aušra Kynienė a, Gediminas Gaigalas a arxiv:1210.5742v2 [physics.atom-ph] 10 Oct 2013 a Institute of Theoretical Physics
Academic Editor: Joseph Reader Received: 21 December 2016; Accepted:19 January 2017; Published: 27 January 2017
 atoms Article JJ2LSJ Transformation and Unique Labeling for Energy Levels Gediminas Gaigalas 1, *, Charlotte Froese Fischer 2, Pavel Rynkun 1 and Per Jönsson 3 1 Institute of Theoretical Physics and Astronomy,
atoms Article JJ2LSJ Transformation and Unique Labeling for Energy Levels Gediminas Gaigalas 1, *, Charlotte Froese Fischer 2, Pavel Rynkun 1 and Per Jönsson 3 1 Institute of Theoretical Physics and Astronomy,
Study of absorption and re-emission processes in a ternary liquid scintillation system *
 CPC(HEP & NP), 2010, 34(11): 1724 1728 Chinese Physics C Vol. 34, No. 11, Nov., 2010 Study of absorption and re-emission processes in a ternary liquid scintillation system * XIAO Hua-Lin( ) 1;1) LI Xiao-Bo(
CPC(HEP & NP), 2010, 34(11): 1724 1728 Chinese Physics C Vol. 34, No. 11, Nov., 2010 Study of absorption and re-emission processes in a ternary liquid scintillation system * XIAO Hua-Lin( ) 1;1) LI Xiao-Bo(
Application of atomic data to quantitative analysis of tungsten spectra on EAST tokamak
 Technical Meeting on Uncertainty Assessment and Benchmark Experiments for Atomic and Molecular Data for Fusion Applications, 19-21 December 2016, Vienna, Austria Application of atomic data to quantitative
Technical Meeting on Uncertainty Assessment and Benchmark Experiments for Atomic and Molecular Data for Fusion Applications, 19-21 December 2016, Vienna, Austria Application of atomic data to quantitative
Academic Editor: Joseph Reader Received: 25 November 2016; Accepted: 6 January 2017; Published: 12 January 2017
 atoms Article Combining Multiconfiguration and Perturbation Methods: Perturbative Estimates of Core Core Electron Correlation Contributions to Excitation Energies in Mg-Like Iron Stefan Gustafsson 1, Per
atoms Article Combining Multiconfiguration and Perturbation Methods: Perturbative Estimates of Core Core Electron Correlation Contributions to Excitation Energies in Mg-Like Iron Stefan Gustafsson 1, Per
Plasma Spectroscopy Inferences from Line Emission
 Plasma Spectroscopy Inferences from Line Emission Ø From line λ, can determine element, ionization state, and energy levels involved Ø From line shape, can determine bulk and thermal velocity and often
Plasma Spectroscopy Inferences from Line Emission Ø From line λ, can determine element, ionization state, and energy levels involved Ø From line shape, can determine bulk and thermal velocity and often
Ab-initio Calculations for Forbidden M1/E2 Decay Rates in Ti XIX ion
 EJTP 3, No. 11 (2006) 111 122 Electronic Journal of Theoretical Physics Ab-initio Calculations for Forbidden M1/E2 Decay Rates in Ti XIX ion A. Farrag Physics Department,Faculty of Science, Cairo University,
EJTP 3, No. 11 (2006) 111 122 Electronic Journal of Theoretical Physics Ab-initio Calculations for Forbidden M1/E2 Decay Rates in Ti XIX ion A. Farrag Physics Department,Faculty of Science, Cairo University,
Analyzing Line Emission Spectra viewed through a Spectroscope using a Smartphone
 Energy (ev) Analyzing Line Emission Spectra viewed through a Spectroscope using a Smartphone Eugene T. Smith, PhD Goals: 1. Calibrate spectroscope using mercury emission source or fluorescent bulb. 2.
Energy (ev) Analyzing Line Emission Spectra viewed through a Spectroscope using a Smartphone Eugene T. Smith, PhD Goals: 1. Calibrate spectroscope using mercury emission source or fluorescent bulb. 2.
Probing magnetic fields in the solar corona using MITs MITs Magnetic-field Induced Transitions. Jon Grumer
 Probing magnetic fields in the solar corona using MITs MITs Magnetic-field Induced Transitions Jon Grumer COMPAS - Division of Mathematical Physics @ Lund University jon.grumer@teorfys.lu.se ICAMDATA /
Probing magnetic fields in the solar corona using MITs MITs Magnetic-field Induced Transitions Jon Grumer COMPAS - Division of Mathematical Physics @ Lund University jon.grumer@teorfys.lu.se ICAMDATA /
Investigation of Water Fragments
 National Nuclear Research University MEPhI Federal State Autonomous Institution for Higher Education 31 Kashirskoe shosse 115409 Moscow, Russia VAT registration number, 7724068140 REG. No 1037739366477
National Nuclear Research University MEPhI Federal State Autonomous Institution for Higher Education 31 Kashirskoe shosse 115409 Moscow, Russia VAT registration number, 7724068140 REG. No 1037739366477
arxiv: v1 [physics.atm-clus] 11 Jun 2014
![arxiv: v1 [physics.atm-clus] 11 Jun 2014 arxiv: v1 [physics.atm-clus] 11 Jun 2014](/thumbs/89/97995797.jpg) Energy levels, radiative rates, and lifetimes for transitions in W LVIII Kanti M. Aggarwal and Francis P. Keenan arxiv:1406.2838v1 [physics.atm-clus] 11 Jun 2014 Astrophysics Research Centre, School of
Energy levels, radiative rates, and lifetimes for transitions in W LVIII Kanti M. Aggarwal and Francis P. Keenan arxiv:1406.2838v1 [physics.atm-clus] 11 Jun 2014 Astrophysics Research Centre, School of
Radiative rates of transitions from the 2s2p 3 5 S 2 level of neutral carbon
 Radiative rates of transitions from the 2s2p 3 5 S 2 level of neutral carbon K Haris 1 and A Kramida National Institute of Standards and Technology, Gaithersburg, MD 20899, USA E-mail: haris.kunari@nist.gov
Radiative rates of transitions from the 2s2p 3 5 S 2 level of neutral carbon K Haris 1 and A Kramida National Institute of Standards and Technology, Gaithersburg, MD 20899, USA E-mail: haris.kunari@nist.gov
Impurity accumulation in the main plasma and radiation processes in the divetor plasma of JT-60U
 1 EX/P4-25 Impurity accumulation in the main plasma and radiation processes in the divetor plasma of JT-6U T. Nakano, H. Kubo, N. Asakura, K. Shimizu and S. Higashijima Japan Atomic Energy Agency, Naka,
1 EX/P4-25 Impurity accumulation in the main plasma and radiation processes in the divetor plasma of JT-6U T. Nakano, H. Kubo, N. Asakura, K. Shimizu and S. Higashijima Japan Atomic Energy Agency, Naka,
Electron temperature is the temperature that describes, through Maxwell's law, the kinetic energy distribution of the free electrons.
 10.3.1.1 Excitation and radiation of spectra 10.3.1.1.1 Plasmas A plasma of the type occurring in spectrochemical radiation sources may be described as a gas which is at least partly ionized and contains
10.3.1.1 Excitation and radiation of spectra 10.3.1.1.1 Plasmas A plasma of the type occurring in spectrochemical radiation sources may be described as a gas which is at least partly ionized and contains
Atomic spectra of one and two-electron systems
 Atomic spectra of one and two-electron systems Key Words Term symbol, Selection rule, Fine structure, Atomic spectra, Sodium D-line, Hund s rules, Russell-Saunders coupling, j-j coupling, Spin-orbit coupling,
Atomic spectra of one and two-electron systems Key Words Term symbol, Selection rule, Fine structure, Atomic spectra, Sodium D-line, Hund s rules, Russell-Saunders coupling, j-j coupling, Spin-orbit coupling,
Observation of Atomic Spectra
 Observation of Atomic Spectra Introduction In this experiment you will observe and measure the wavelengths of different colors of light emitted by atoms. You will first observe light emitted from excited
Observation of Atomic Spectra Introduction In this experiment you will observe and measure the wavelengths of different colors of light emitted by atoms. You will first observe light emitted from excited
Two-electron systems
 Two-electron systems Laboratory exercise for FYSC11 Instructor: Hampus Nilsson hampus.nilsson@astro.lu.se Lund Observatory Lund University September 12, 2016 Goal In this laboration we will make use of
Two-electron systems Laboratory exercise for FYSC11 Instructor: Hampus Nilsson hampus.nilsson@astro.lu.se Lund Observatory Lund University September 12, 2016 Goal In this laboration we will make use of
Experiment #9. Atomic Emission Spectroscopy
 Introduction Experiment #9. Atomic Emission Spectroscopy Spectroscopy is the study of the interaction of light with matter. This interaction can be in the form of the absorption or the emission of electromagnetic
Introduction Experiment #9. Atomic Emission Spectroscopy Spectroscopy is the study of the interaction of light with matter. This interaction can be in the form of the absorption or the emission of electromagnetic
Chapter 28. Atomic Physics
 Chapter 28 Atomic Physics Quantum Numbers and Atomic Structure The characteristic wavelengths emitted by a hot gas can be understood using quantum numbers. No two electrons can have the same set of quantum
Chapter 28 Atomic Physics Quantum Numbers and Atomic Structure The characteristic wavelengths emitted by a hot gas can be understood using quantum numbers. No two electrons can have the same set of quantum
プラズマ光源関連イオンの EBIT による分光
 プラズマ光源関連イオンの EBIT による分光 Emission spectroscopy of multiply charged ions related to plasma light sources with an EBIT 大橋隼人, 八釼純治, 坂上裕之 *, 中村信行 レーザー新世代研究センター, 電気通信大学 * 核融合科学研究所 2012 年度原子分子データ応用フォーラムセミナー 2012
プラズマ光源関連イオンの EBIT による分光 Emission spectroscopy of multiply charged ions related to plasma light sources with an EBIT 大橋隼人, 八釼純治, 坂上裕之 *, 中村信行 レーザー新世代研究センター, 電気通信大学 * 核融合科学研究所 2012 年度原子分子データ応用フォーラムセミナー 2012
The Structure of the Atom
 CHAPTER 5 The Structure of the Atom 5.4 Light and Spectroscopy 460 370 BC 1808 1870 1897 1910 1925 Today Democritus Atomism Dalton Modern atomic theory Crookes Cathode rays Thomson Discovery of the electron
CHAPTER 5 The Structure of the Atom 5.4 Light and Spectroscopy 460 370 BC 1808 1870 1897 1910 1925 Today Democritus Atomism Dalton Modern atomic theory Crookes Cathode rays Thomson Discovery of the electron
Radiative data in the Zr I spectrum obtained by Laser Induced Fluorescence
 Radiative data in the Zr I spectrum obtained by Laser Induced Fluorescence G. Malcheva 1, R. Mayo 2, M. Ortiz 2, J. Ruiz 2, L. Engström 3, H. Lundberg 3, H. Nilsson 4, P. Quinet 5,6, É. Biémont 5,6 and
Radiative data in the Zr I spectrum obtained by Laser Induced Fluorescence G. Malcheva 1, R. Mayo 2, M. Ortiz 2, J. Ruiz 2, L. Engström 3, H. Lundberg 3, H. Nilsson 4, P. Quinet 5,6, É. Biémont 5,6 and
Lab 10: Spectroscopy & the Hydrogen Atom Phy208 Fall 2008
 Lab 10: Spectroscopy & the Hydrogen Atom Phy208 Fall 2008 Name Section This sheet is the lab document your TA will use to score your lab. It is to be turned in at the end of lab. To receive full credit
Lab 10: Spectroscopy & the Hydrogen Atom Phy208 Fall 2008 Name Section This sheet is the lab document your TA will use to score your lab. It is to be turned in at the end of lab. To receive full credit
Identification and Plasma Diagnostics Study of Extreme Ultraviolet Transitions in Highly Charged Yttrium
 atoms Article Identification and Plasma Diagnostics Study of Extreme Ultraviolet Transitions in Highly Charged Yttrium Roshani Silwal 1,2,, Endre Takacs 1,2, Joan M. Dreiling 2, John D. Gillaspy 2,3 and
atoms Article Identification and Plasma Diagnostics Study of Extreme Ultraviolet Transitions in Highly Charged Yttrium Roshani Silwal 1,2,, Endre Takacs 1,2, Joan M. Dreiling 2, John D. Gillaspy 2,3 and
Atomic Spectroscopy. Objectives
 Atomic Spectroscopy Name Objectives explain the difference between emission and absorption spectra calculate the energy of orbits in the Bohr model of hydrogen calculate E for energy transitions in the
Atomic Spectroscopy Name Objectives explain the difference between emission and absorption spectra calculate the energy of orbits in the Bohr model of hydrogen calculate E for energy transitions in the
PHYS General Physics II Lab The Balmer Series for Hydrogen Source. c = speed of light = 3 x 10 8 m/s
 PHYS 1040 - General Physics II Lab The Balmer Series for Hydrogen Source Purpose: The purpose of this experiment is to analyze the emission of light from a hydrogen source and measure and the wavelengths
PHYS 1040 - General Physics II Lab The Balmer Series for Hydrogen Source Purpose: The purpose of this experiment is to analyze the emission of light from a hydrogen source and measure and the wavelengths
Extreme-ultraviolet emissivity from Xe 8+ to Xe 12+ by using a detailed line-by-line method
 Eur. Phys. J. D (21) DOI: 1.114/epjd/e21-192-6 Regular Article THE EUROPEAN PHYSICAL JOURNAL D Extreme-ultraviolet emissivity from Xe 8+ to Xe 12+ by using a detailed line-by-line method J. Zeng a, C.
Eur. Phys. J. D (21) DOI: 1.114/epjd/e21-192-6 Regular Article THE EUROPEAN PHYSICAL JOURNAL D Extreme-ultraviolet emissivity from Xe 8+ to Xe 12+ by using a detailed line-by-line method J. Zeng a, C.
where n = (an integer) =
 5.111 Lecture Summary #5 Readings for today: Section 1.3 (1.6 in 3 rd ed) Atomic Spectra, Section 1.7 up to equation 9b (1.5 up to eq. 8b in 3 rd ed) Wavefunctions and Energy Levels, Section 1.8 (1.7 in
5.111 Lecture Summary #5 Readings for today: Section 1.3 (1.6 in 3 rd ed) Atomic Spectra, Section 1.7 up to equation 9b (1.5 up to eq. 8b in 3 rd ed) Wavefunctions and Energy Levels, Section 1.8 (1.7 in
Rostohar, D; Derkatch, A; Hartman, Henrik; Johansson, Sveneric; Lundberg, Hans; Mannervik, S; Norlin, L-O; Royen, P; Schmitt, A
 Lifetime measurements of Metastable States in Fe+ Rostohar, D; Derkatch, A; Hartman, Henrik; Johansson, Sveneric; Lundberg, Hans; Mannervik, S; Norlin, L-O; Royen, P; Schmitt, A Published in: Physical
Lifetime measurements of Metastable States in Fe+ Rostohar, D; Derkatch, A; Hartman, Henrik; Johansson, Sveneric; Lundberg, Hans; Mannervik, S; Norlin, L-O; Royen, P; Schmitt, A Published in: Physical
high temp ( K) Chapter 20: Atomic Spectroscopy
 high temp (2000-6000K) Chapter 20: Atomic Spectroscopy 20-1. An Overview Most compounds Atoms in gas phase high temp (2000-6000K) (AES) (AAS) (AFS) sample Mass-to-charge (ICP-MS) Atomic Absorption experiment
high temp (2000-6000K) Chapter 20: Atomic Spectroscopy 20-1. An Overview Most compounds Atoms in gas phase high temp (2000-6000K) (AES) (AAS) (AFS) sample Mass-to-charge (ICP-MS) Atomic Absorption experiment
Lab 5: Spectroscopy & the Hydrogen Atom Phy248 Spring 2009
 Lab 5: Spectroscopy & the Hydrogen Atom Phy248 Spring 2009 Name Section Return this spreadsheet to your TA that will use it to score your lab. To receive full credit you must use complete sentences and
Lab 5: Spectroscopy & the Hydrogen Atom Phy248 Spring 2009 Name Section Return this spreadsheet to your TA that will use it to score your lab. To receive full credit you must use complete sentences and
Production of HCI with an electron beam ion trap
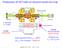 Production of HCI with an electron beam ion trap I=450 ma E= 5 kev axially: electrodes radially: electron beam space charge total trap potential U trap 200 V (U trap ion charge) 10000 ev 15000 A/cm 2 n
Production of HCI with an electron beam ion trap I=450 ma E= 5 kev axially: electrodes radially: electron beam space charge total trap potential U trap 200 V (U trap ion charge) 10000 ev 15000 A/cm 2 n
Comparison of hollow cathode and Penning discharges for metastable He production
 INSTITUTE OF PHYSICS PUBLISHING Plasma Sources Sci. Technol. 11 (2002) 426 430 Comparison of hollow cathode and Penning discharges for metastable He production PLASMA SOURCES SCIENCE AND TECHNOLOGY PII:
INSTITUTE OF PHYSICS PUBLISHING Plasma Sources Sci. Technol. 11 (2002) 426 430 Comparison of hollow cathode and Penning discharges for metastable He production PLASMA SOURCES SCIENCE AND TECHNOLOGY PII:
Experiment 7: Spectrum of the Hydrogen Atom
 Experiment 7: Spectrum of the Hydrogen Nate Saffold nas2173@columbia.edu Office Hour: Mondays, 5:30-6:30PM INTRO TO EXPERIMENTAL PHYS-LAB 1493/1494/2699 Introduction The physics behind: The spectrum of
Experiment 7: Spectrum of the Hydrogen Nate Saffold nas2173@columbia.edu Office Hour: Mondays, 5:30-6:30PM INTRO TO EXPERIMENTAL PHYS-LAB 1493/1494/2699 Introduction The physics behind: The spectrum of
Canadian Journal of Physics. Energy, fine structure, hyperfine structure, and transitions for the high-lying multi-excited 4Pe,o states of B-like ions
 Energy, fine structure, hyperfine structure, and transitions for the high-lying multi-excited 4Pe,o states of B-like ions Journal: Canadian Journal of Physics Manuscript ID cjp-2-69.r3 Manuscript Type:
Energy, fine structure, hyperfine structure, and transitions for the high-lying multi-excited 4Pe,o states of B-like ions Journal: Canadian Journal of Physics Manuscript ID cjp-2-69.r3 Manuscript Type:
The Grating Spectrometer and Atomic Spectra
 PHY 192 Grating Spectrometer 1 The Grating Spectrometer and Atomic Spectra Introduction In the previous experiment diffraction and interference were discussed and at the end a diffraction grating was introduced.
PHY 192 Grating Spectrometer 1 The Grating Spectrometer and Atomic Spectra Introduction In the previous experiment diffraction and interference were discussed and at the end a diffraction grating was introduced.
Estimating the plasma flow in a recombining plasma from
 Paper P3-38 Estimating the plasma flow in a recombining plasma from the H α emission U. Wenzel a, M. Goto b a Max-Planck-Institut für Plasmaphysik (IPP) b National Institute for Fusion Science, Toki 509-5292,
Paper P3-38 Estimating the plasma flow in a recombining plasma from the H α emission U. Wenzel a, M. Goto b a Max-Planck-Institut für Plasmaphysik (IPP) b National Institute for Fusion Science, Toki 509-5292,
The Spectrophotometer and Atomic Spectra of Hydrogen Physics 246
 The Spectrophotometer and Atomic Spectra of Hydrogen Physics 46 Introduction: When heated sufficiently, most elements emit light. With a spectrometer, the emitted light can be broken down into its various
The Spectrophotometer and Atomic Spectra of Hydrogen Physics 46 Introduction: When heated sufficiently, most elements emit light. With a spectrometer, the emitted light can be broken down into its various
Chapter 28 Atomic Physics
 Chapter 28 Atomic Physics GOALS After you have mastered the contents of this chapter, you will be able to achieve the following goals: Definitions Define each of the following terms and use it in an operational
Chapter 28 Atomic Physics GOALS After you have mastered the contents of this chapter, you will be able to achieve the following goals: Definitions Define each of the following terms and use it in an operational
Atomic structure and dynamics
 Atomic structure and dynamics -- need and requirements for accurate atomic calculations Analysis and interpretation of optical and x-ray spectra (astro physics) Isotope shifts and hyperfine structures
Atomic structure and dynamics -- need and requirements for accurate atomic calculations Analysis and interpretation of optical and x-ray spectra (astro physics) Isotope shifts and hyperfine structures
Line ratios and wavelengths of helium-like argon n = 2 satellite transitions and resonance lines
 1 Line ratios and wavelengths of helium-like argon n = 2 satellite transitions and resonance lines C. Biedermann a, R. Radtke a, and K. Fournier b a Max-Planck-Institut für Plasmaphysik, Bereich Plasmadiagnostik,
1 Line ratios and wavelengths of helium-like argon n = 2 satellite transitions and resonance lines C. Biedermann a, R. Radtke a, and K. Fournier b a Max-Planck-Institut für Plasmaphysik, Bereich Plasmadiagnostik,
Atomic fluorescence. The intensity of a transition line can be described with a transition probability inversely
 Atomic fluorescence 1. Introduction Transitions in multi-electron atoms Energy levels of the single-electron hydrogen atom are well-described by EE nn = RR nn2, where RR = 13.6 eeee is the Rydberg constant.
Atomic fluorescence 1. Introduction Transitions in multi-electron atoms Energy levels of the single-electron hydrogen atom are well-described by EE nn = RR nn2, where RR = 13.6 eeee is the Rydberg constant.
James Maxwell ( )
 From Atoms To Stars James Maxwell (1831 1879) Finalized the work of others on electricity and magnetism. He formulated Maxwell Equations for the electromagnetic field. His equations predicted the existence
From Atoms To Stars James Maxwell (1831 1879) Finalized the work of others on electricity and magnetism. He formulated Maxwell Equations for the electromagnetic field. His equations predicted the existence
EMISSION SPECTROSCOPY
 IFM The Department of Physics, Chemistry and Biology LAB 57 EMISSION SPECTROSCOPY NAME PERSONAL NUMBER DATE APPROVED I. OBJECTIVES - Understand the principle of atomic emission spectra. - Know how to acquire
IFM The Department of Physics, Chemistry and Biology LAB 57 EMISSION SPECTROSCOPY NAME PERSONAL NUMBER DATE APPROVED I. OBJECTIVES - Understand the principle of atomic emission spectra. - Know how to acquire
Isotopic effect of Cl + 2 rovibronic spectra in the A X system
 Vol 18 No 7, July 009 c 009 Chin. Phys. Soc. 1674-1056/009/1807)/74-05 Chinese Physics B and IOP Publishing Ltd Isotopic effect of Cl + rovibronic spectra in the A X system Wu Ling ) a)c), Yang Xiao-Hua
Vol 18 No 7, July 009 c 009 Chin. Phys. Soc. 1674-1056/009/1807)/74-05 Chinese Physics B and IOP Publishing Ltd Isotopic effect of Cl + rovibronic spectra in the A X system Wu Ling ) a)c), Yang Xiao-Hua
A more comprehensive theory was needed. 1925, Schrödinger and Heisenberg separately worked out a new theory Quantum Mechanics.
 Ch28 Quantum Mechanics of Atoms Bohr s model was very successful to explain line spectra and the ionization energy for hydrogen. However, it also had many limitations: It was not able to predict the line
Ch28 Quantum Mechanics of Atoms Bohr s model was very successful to explain line spectra and the ionization energy for hydrogen. However, it also had many limitations: It was not able to predict the line
The Grating Spectrometer and Atomic Spectra
 PHY 192 Grating Spectrometer Spring 2012 1 The Grating Spectrometer and Atomic Spectra Introduction In the previous experiment diffraction and interference were discussed and at the end a diffraction grating
PHY 192 Grating Spectrometer Spring 2012 1 The Grating Spectrometer and Atomic Spectra Introduction In the previous experiment diffraction and interference were discussed and at the end a diffraction grating
Lecture 0. NC State University
 Chemistry 736 Lecture 0 Overview NC State University Overview of Spectroscopy Electronic states and energies Transitions between states Absorption and emission Electronic spectroscopy Instrumentation Concepts
Chemistry 736 Lecture 0 Overview NC State University Overview of Spectroscopy Electronic states and energies Transitions between states Absorption and emission Electronic spectroscopy Instrumentation Concepts
A fluorescent tube is filled with mercury vapour at low pressure. After mercury atoms have been excited they emit photons.
 Q1.(a) A fluorescent tube is filled with mercury vapour at low pressure. After mercury atoms have been excited they emit photons. In which part of the electromagnetic spectrum are these photons? What is
Q1.(a) A fluorescent tube is filled with mercury vapour at low pressure. After mercury atoms have been excited they emit photons. In which part of the electromagnetic spectrum are these photons? What is
Calculation of the Isotope Shifts on 5S 1/2 4D 3/2,5/2 Transitions of 87,88 Sr +
 Commun. Theor. Phys. (Beijing, China) 37 (22) pp 76 7 c International Academic Publishers Vol. 37, No. 6, June 5, 22 Calculation of the Isotope Shifts on 5S /2 4D 3/2,5/2 Transitions of 87,88 Sr + LI Yong,,2
Commun. Theor. Phys. (Beijing, China) 37 (22) pp 76 7 c International Academic Publishers Vol. 37, No. 6, June 5, 22 Calculation of the Isotope Shifts on 5S /2 4D 3/2,5/2 Transitions of 87,88 Sr + LI Yong,,2
Oscillator strengths and E1 radiative rates for Ca-like titanium, Ti III
 Int. J. New. Hor. Phys. 2, No. 1, 25-31 (2015) 25 International Journal of New Horizons in Physics http://dx.doi.org/10.12785/ijnhp/020105 Oscillator strengths and E1 radiative rates for Ca-like titanium,
Int. J. New. Hor. Phys. 2, No. 1, 25-31 (2015) 25 International Journal of New Horizons in Physics http://dx.doi.org/10.12785/ijnhp/020105 Oscillator strengths and E1 radiative rates for Ca-like titanium,
Shape Coexistence and Band Termination in Doubly Magic Nucleus 40 Ca
 Commun. Theor. Phys. (Beijing, China) 43 (2005) pp. 509 514 c International Academic Publishers Vol. 43, No. 3, March 15, 2005 Shape Coexistence and Band Termination in Doubly Magic Nucleus 40 Ca DONG
Commun. Theor. Phys. (Beijing, China) 43 (2005) pp. 509 514 c International Academic Publishers Vol. 43, No. 3, March 15, 2005 Shape Coexistence and Band Termination in Doubly Magic Nucleus 40 Ca DONG
Theoretical study on the K α transition properties of F-like ions
 J. At. Mol. Sci. doi: 10.4208/jams.013010.022010a Vol. 1, No. 2, pp. 134-142 May 2010 Theoretical study on the K α transition properties of F-like ions X. L. Wang, J. J. Wan, Y. J. Wang, and C. Z. Dong
J. At. Mol. Sci. doi: 10.4208/jams.013010.022010a Vol. 1, No. 2, pp. 134-142 May 2010 Theoretical study on the K α transition properties of F-like ions X. L. Wang, J. J. Wan, Y. J. Wang, and C. Z. Dong
General Physics (PHY 2140)
 General Physics (PHY 140) Lecture 33 Modern Physics Atomic Physics Atomic spectra Bohr s theory of hydrogen http://www.physics.wayne.edu/~apetrov/phy140/ Chapter 8 1 Lightning Review Last lecture: 1. Atomic
General Physics (PHY 140) Lecture 33 Modern Physics Atomic Physics Atomic spectra Bohr s theory of hydrogen http://www.physics.wayne.edu/~apetrov/phy140/ Chapter 8 1 Lightning Review Last lecture: 1. Atomic
Time-dependent kinetics model for a helium discharge plasma
 J. Phys. B: At. Mol. Opt. Phys. 32 (1999) 1001 1008. Printed in the UK PII: S0953-4075(99)97893-8 Time-dependent kinetics model for a helium discharge plasma J Abdallah Jr, N Palmer, W Gekelman, J Maggs
J. Phys. B: At. Mol. Opt. Phys. 32 (1999) 1001 1008. Printed in the UK PII: S0953-4075(99)97893-8 Time-dependent kinetics model for a helium discharge plasma J Abdallah Jr, N Palmer, W Gekelman, J Maggs
Because light behaves like a wave, we can describe it in one of two ways by its wavelength or by its frequency.
 Light We can use different terms to describe light: Color Wavelength Frequency Light is composed of electromagnetic waves that travel through some medium. The properties of the medium determine how light
Light We can use different terms to describe light: Color Wavelength Frequency Light is composed of electromagnetic waves that travel through some medium. The properties of the medium determine how light
CSUS Department of Chemistry Experiment 9 Chem. 1A
 CSUS Department of Chemistry xperiment 9 Chem. 1A xp. 9 PR-Lab ASSIGNMNT Name: Lab Section (1) Use equation (2) [see the discussion on the next page] to calculate the energies of the ten lowest states
CSUS Department of Chemistry xperiment 9 Chem. 1A xp. 9 PR-Lab ASSIGNMNT Name: Lab Section (1) Use equation (2) [see the discussion on the next page] to calculate the energies of the ten lowest states
The LANL atomic kinetics modeling effort and its application to W plasmas
 The LANL atomic kinetics modeling effort and its application to W plasmas James Colgan, Joseph Abdallah, Jr., Christopher Fontes, Honglin Zhang Los Alamos National Laboratory IAEA CRP December 2010 jcolgan@lanl.gov
The LANL atomic kinetics modeling effort and its application to W plasmas James Colgan, Joseph Abdallah, Jr., Christopher Fontes, Honglin Zhang Los Alamos National Laboratory IAEA CRP December 2010 jcolgan@lanl.gov
Atomic and Molecular Data Activities at NIFS in
 21 th DCN meeting, IAEA, Sep. 7-9, 2011 Atomic and Molecular Data Activities at NIFS in 2009 2011 Izumi Murakami Atomic and Molecular Processes Section, Fusion Systems Research Division, Department of
21 th DCN meeting, IAEA, Sep. 7-9, 2011 Atomic and Molecular Data Activities at NIFS in 2009 2011 Izumi Murakami Atomic and Molecular Processes Section, Fusion Systems Research Division, Department of
Introduction. Procedure and Data
 Introduction The spectrum is the entire range over which some measurable property of a physical system of phenomenon can vary. Systems that have spectrums include sound frequency, electromagnetic radiation
Introduction The spectrum is the entire range over which some measurable property of a physical system of phenomenon can vary. Systems that have spectrums include sound frequency, electromagnetic radiation
UNIT : QUANTUM THEORY AND THE ATOM
 Name St.No. Date(YY/MM/DD) / / Section UNIT 102-10: QUANTUM THEORY AND THE ATOM OBJECTIVES Atomic Spectra for Hydrogen, Mercury and Neon. 1. To observe various atomic spectra with a diffraction grating
Name St.No. Date(YY/MM/DD) / / Section UNIT 102-10: QUANTUM THEORY AND THE ATOM OBJECTIVES Atomic Spectra for Hydrogen, Mercury and Neon. 1. To observe various atomic spectra with a diffraction grating
Relativistic multichannel treatment of autoionization Rydberg series of 4s 2 nf(n = 4 23)J π = (7/2) for scandium
 Vol 17 No 6, June 2008 c 2008 Chin. Phys. Soc. 1674-1056/2008/17(06)/2027-06 Chinese Physics B and IOP Publishing Ltd Relativistic multichannel treatment of autoionization Rydberg series of 4s 2 nf(n =
Vol 17 No 6, June 2008 c 2008 Chin. Phys. Soc. 1674-1056/2008/17(06)/2027-06 Chinese Physics B and IOP Publishing Ltd Relativistic multichannel treatment of autoionization Rydberg series of 4s 2 nf(n =
Survey of EUV Impurity Line Spectra and EUV Bremsstrahlung Continuum in LHD )
 Plasma and Fusion Research: Regular Articles Volume 6, 2402078 (2011) Survey of EUV Impurity Line Spectra and EUV Bremsstrahlung Continuum in LHD ) Chunfeng DONG, Shigeru MORITA 1), Malay Bikas CHOWDHURI
Plasma and Fusion Research: Regular Articles Volume 6, 2402078 (2011) Survey of EUV Impurity Line Spectra and EUV Bremsstrahlung Continuum in LHD ) Chunfeng DONG, Shigeru MORITA 1), Malay Bikas CHOWDHURI
Emission Spectrum of Atomic Gases. Prelab Questions
 Emission Spectrum of Atomic Gases Prelab Questions Before this coming to this lab, please review your text for the physics of the spectrum of visible light and of diffraction grating spectrometer.. Which
Emission Spectrum of Atomic Gases Prelab Questions Before this coming to this lab, please review your text for the physics of the spectrum of visible light and of diffraction grating spectrometer.. Which
First Experiments with the Greifswald EBIT
 First Experiments with the Greifswald EBIT B Schabinger 1, C Biedermann 2, S Gierke 1, G Marx 1, R Radtke 1,2 and L Schweikhard 1 1 Ernst Moritz Arndt University of Greifswald, Institute of Physics, Felix-Hausdorff-Str.
First Experiments with the Greifswald EBIT B Schabinger 1, C Biedermann 2, S Gierke 1, G Marx 1, R Radtke 1,2 and L Schweikhard 1 1 Ernst Moritz Arndt University of Greifswald, Institute of Physics, Felix-Hausdorff-Str.
Introduction to the Diagnosis of Magnetically Confined Thermonuclear Plasma
 Introduction to the Diagnosis of Magnetically Confined Thermonuclear Plasma Core diagnostics II: Bolometry and Soft X-rays J. Arturo Alonso Laboratorio Nacional de Fusión EURATOM-CIEMAT E6 P2.10 arturo.alonso@ciemat.es
Introduction to the Diagnosis of Magnetically Confined Thermonuclear Plasma Core diagnostics II: Bolometry and Soft X-rays J. Arturo Alonso Laboratorio Nacional de Fusión EURATOM-CIEMAT E6 P2.10 arturo.alonso@ciemat.es
Plasma Spectroscopy in ISTTOK
 Plasma Spectroscopy in ISTTOK J. Figueiredo 1, R. B. Gomes 1, T. Pereira 1, H. Fernandes 1, A. Sharakovski 2 1 Associação EURATOM/IST, Centro de Fusão Nuclear, IST, 1049-001 Lisboa, Portugal 2 Association
Plasma Spectroscopy in ISTTOK J. Figueiredo 1, R. B. Gomes 1, T. Pereira 1, H. Fernandes 1, A. Sharakovski 2 1 Associação EURATOM/IST, Centro de Fusão Nuclear, IST, 1049-001 Lisboa, Portugal 2 Association
Complete the following. Clearly mark your answers. YOU MUST SHOW YOUR WORK TO RECEIVE CREDIT.
 CHEM 322 Name Exam 3 Spring 2013 Complete the following. Clearly mark your answers. YOU MUST SHOW YOUR WORK TO RECEIVE CREDIT. Warm-up (3 points each). 1. In Raman Spectroscopy, molecules are promoted
CHEM 322 Name Exam 3 Spring 2013 Complete the following. Clearly mark your answers. YOU MUST SHOW YOUR WORK TO RECEIVE CREDIT. Warm-up (3 points each). 1. In Raman Spectroscopy, molecules are promoted
Impurity transport analysis & preparation of W injection experiments on KSTAR
 Impurity transport analysis & preparation of W injection experiments on KSTAR J. H. Hong, H. Y. Lee, S. H. Lee, S. Jang, J. Jang, T. Jeon, H. Lee, and W. Choe ( ) S. G. Lee, C. R. Seon, J. Kim, ( ) 마스터부제목스타일편집
Impurity transport analysis & preparation of W injection experiments on KSTAR J. H. Hong, H. Y. Lee, S. H. Lee, S. Jang, J. Jang, T. Jeon, H. Lee, and W. Choe ( ) S. G. Lee, C. R. Seon, J. Kim, ( ) 마스터부제목스타일편집
The trap states in the Sr 2 MgSi 2 O 7 and (Sr,Ca)MgSi 2 O 7 long afterglow phosphor activated by Eu 2+ and Dy 3+
 Journal of Alloys and Compounds 387 (2005) 65 69 The trap states in the Sr 2 MgSi 2 O 7 and (Sr,Ca)MgSi 2 O 7 long afterglow phosphor activated by Eu 2+ and Dy 3+ Bo Liu a,, Chaoshu Shi a,b, Min Yin a,
Journal of Alloys and Compounds 387 (2005) 65 69 The trap states in the Sr 2 MgSi 2 O 7 and (Sr,Ca)MgSi 2 O 7 long afterglow phosphor activated by Eu 2+ and Dy 3+ Bo Liu a,, Chaoshu Shi a,b, Min Yin a,
Validation of spectral MSE for Alcator C-Mod and for ITER
 Validation of spectral MSE for Alcator C-Mod and for ITER K.T. Liao 1, W.L. Rowan 1, R.T. Mumgaard 2, Bob Granetz 2, Steve Scott 3, Fred Levinton 4, Howard Yuh 4, O. Marchuk 5, Y. Ralchenko 6 1 The University
Validation of spectral MSE for Alcator C-Mod and for ITER K.T. Liao 1, W.L. Rowan 1, R.T. Mumgaard 2, Bob Granetz 2, Steve Scott 3, Fred Levinton 4, Howard Yuh 4, O. Marchuk 5, Y. Ralchenko 6 1 The University
High-Resolution Survey of the Visible Spectrum of NiF by Fourier Transform Spectroscopy
 Journal of Molecular Spectroscopy 214, 152 174 (2002) doi:10.1006/jmsp.2002.8567 High-Resolution Survey of the Visible Spectrum of NiF by Fourier Transform Spectroscopy Y. Krouti, T. Hirao,,1 C. Dufour,
Journal of Molecular Spectroscopy 214, 152 174 (2002) doi:10.1006/jmsp.2002.8567 High-Resolution Survey of the Visible Spectrum of NiF by Fourier Transform Spectroscopy Y. Krouti, T. Hirao,,1 C. Dufour,
Sharif University of Technology Physics Department. Modern Physics Spring 2016 Prof. Akhavan
 Sharif University of Technology Physics Department Modern Physics Spring 2016 Prof. Akhavan Problems Set #5. Due on: 03 th of April / 15 th of Farvardin. 1 Blackbody Radiation. (Required text book is Modern
Sharif University of Technology Physics Department Modern Physics Spring 2016 Prof. Akhavan Problems Set #5. Due on: 03 th of April / 15 th of Farvardin. 1 Blackbody Radiation. (Required text book is Modern
Formation of High-b ECH Plasma and Inward Particle Diffusion in RT-1
 J Fusion Energ (2010) 29:553 557 DOI 10.1007/s10894-010-9327-6 ORIGINAL RESEARCH Formation of High-b ECH Plasma and Inward Particle Diffusion in RT-1 H. Saitoh Z. Yoshida J. Morikawa Y. Yano T. Mizushima
J Fusion Energ (2010) 29:553 557 DOI 10.1007/s10894-010-9327-6 ORIGINAL RESEARCH Formation of High-b ECH Plasma and Inward Particle Diffusion in RT-1 H. Saitoh Z. Yoshida J. Morikawa Y. Yano T. Mizushima
Vacuum ultraviolet 5d-4f luminescence of Gd 3+ and Lu 3+ ions in fluoride matrices
 Vacuum ultraviolet 5d-4f luminescence of Gd 3+ and Lu 3+ ions in fluoride matrices M. Kirm, 1 G. Stryganyuk, 2,3 S. Vielhauer, 1 G. Zimmerer, 2,3 V.N. Makhov, 1,4 B.Z. Malkin, 5 O.V. Solovyev, 5 R.Yu.
Vacuum ultraviolet 5d-4f luminescence of Gd 3+ and Lu 3+ ions in fluoride matrices M. Kirm, 1 G. Stryganyuk, 2,3 S. Vielhauer, 1 G. Zimmerer, 2,3 V.N. Makhov, 1,4 B.Z. Malkin, 5 O.V. Solovyev, 5 R.Yu.
Any first year text, sections on atomic structure, spectral lines and spectrometers
 Physics 33 Experiment 5 Atomic Spectra References Any first year text, sections on atomic structure, spectral lines and spectrometers Any modern physics text, eg F.K. Richtmeyer, E.H. Kennard and J.N.
Physics 33 Experiment 5 Atomic Spectra References Any first year text, sections on atomic structure, spectral lines and spectrometers Any modern physics text, eg F.K. Richtmeyer, E.H. Kennard and J.N.
ONE-ELECTRON AND TWO-ELECTRON SPECTRA
 ONE-ELECTRON AND TWO-ELECTRON SPECTRA (A) FINE STRUCTURE AND ONE-ELECTRON SPECTRUM PRINCIPLE AND TASK The well-known spectral lines of He are used for calibrating the diffraction spectrometer. The wavelengths
ONE-ELECTRON AND TWO-ELECTRON SPECTRA (A) FINE STRUCTURE AND ONE-ELECTRON SPECTRUM PRINCIPLE AND TASK The well-known spectral lines of He are used for calibrating the diffraction spectrometer. The wavelengths
ATOMIC PHYSICS. history/cosmology/tools/ tools-spectroscopy.htm CHAPTER 9 - FROM SPECTROSCOPY TO ATOMS
 ATOMIC PHYSICS http://www.aip.org/ history/cosmology/tools/ tools-spectroscopy.htm CHAPTER 9 - FROM SPECTROSCOPY TO ATOMS What We Will Study Basics of electromagnetic radiation - The AC generator, again
ATOMIC PHYSICS http://www.aip.org/ history/cosmology/tools/ tools-spectroscopy.htm CHAPTER 9 - FROM SPECTROSCOPY TO ATOMS What We Will Study Basics of electromagnetic radiation - The AC generator, again
Chapter 5 Electrons In Atoms
 Chapter 5 Electrons In Atoms 5.1 Revising the Atomic Model 5.2 Electron Arrangement in Atoms 5.3 Atomic Emission Spectra and the Quantum Mechanical Model 1 Copyright Pearson Education, Inc., or its affiliates.
Chapter 5 Electrons In Atoms 5.1 Revising the Atomic Model 5.2 Electron Arrangement in Atoms 5.3 Atomic Emission Spectra and the Quantum Mechanical Model 1 Copyright Pearson Education, Inc., or its affiliates.
[2] (b) An electron is accelerated from rest through a potential difference of 300 V.
![[2] (b) An electron is accelerated from rest through a potential difference of 300 V. [2] (b) An electron is accelerated from rest through a potential difference of 300 V.](/thumbs/89/98791036.jpg) 1 (a) In atomic physics electron energies are often stated in electronvolts (ev) Define the electronvolt. State its value in joule.. [2] (b) An electron is accelerated from rest through a potential difference
1 (a) In atomic physics electron energies are often stated in electronvolts (ev) Define the electronvolt. State its value in joule.. [2] (b) An electron is accelerated from rest through a potential difference
Atomic and Molecular Data Activities for Fusion Research in JAEA. T. Nakano Japan Atomic Energy Agency
 "Technical Aspects of Atomic and Molecular Data Processing and Exchange" (20th Meeting of the Atomic and Molecular Data Centres and ALADDIN Network), 7-9 September 2009, IAEA HQ, Vienna, Austria Atomic
"Technical Aspects of Atomic and Molecular Data Processing and Exchange" (20th Meeting of the Atomic and Molecular Data Centres and ALADDIN Network), 7-9 September 2009, IAEA HQ, Vienna, Austria Atomic
