DNS of transition in hypersonic boundary-layer flows including high-temperature gas effects
|
|
|
- Julius Brown
- 6 years ago
- Views:
Transcription
1 Center for Turbulence Research Annual Research Brefs DNS of transton n hypersonc boundary-layer flows ncludng hgh-temperature gas effects By C. Stemmer AND N. N. Mansour 1. Motvaton and Objectve Wnd-tunnel experments at hypersonc Mach numbers above 10 are extremely dffcult to undertake and facltes are lmted. Addtonally,the stagnaton condtons for free flght under atmospherc condtons can not be reproduced. Ths results n a lmted portablty of the wnd-tunnel results to atmospherc condtons. Therefore,numercal nvestgatons of hypersonc transton can be extremely valuable n developng an understandng of the transton process at hypersonc speeds. The objectve of ths effort s to develop an understandng of effects of nonequlbrum chemstry on transton. Our approach s to compare hypersonc transton on a flat plate under nonequlbrum chemcal and thermal condtons to hypersonc transton under equlbrum condtons. In the 1950 s and 60 s,a seres of hypersonc experments was conducted n free flght. The transton locaton could be found but no detals on the transtonal structures could be recorded n these experments see Schneder,1999,for a comprehensve revew of supersonc and hypersonc experments. Schneder also notes that the angles of attack of the test vehcles are uncertan. An ongong experment on transton at Ma = 21 n Novosbrsk,Russa Mronov & Maslov 2000,promses expermental verfcaton of the numercal fndngs to some extent. Further detaled experments on transton at hypersonc speeds cannot be expected n the near future. 2. Governng Equatons In order not to confuse the ndex notatons,the ndex refers to the speces 1-5 and no summaton s mpled on ths ndex,whereas the ndces j, k and l refer to the Cartesan drectons x, y and z and summaton from 1-3 s mpled. The contnuty equaton for chemcally-reactng compressble flows becomes ρ t + ρ u j + u D x,j = W, 2.1 j where W represents the speces producton terms see Eq and u D the dffuson veloctes see Eq Rewrtng ths equaton wth the speces concentratons rather than the denstes,t becomes ρ Dc Dt + ρ u D x,j =W, 2.2 j where the speces concentratons are gven by c = ρ ρ. 2.3
2 144 C. Stemmer & N. N. Mansour Note that snce c =1, 2.4 only 1 equatons have to be solved. The total mass s conserved ρ t + ρu j = The total momentum equatons are ρ Du j Dt = p + τ jk 2.6 x k wth uj τ jk = µ x k + u k The bulk vscosty s denoted by λ. The energy equaton for the total energy becomes ρ De + u ju j /2 Dt = q j + q vb j,j pu j,j + u k τ jk + + δ jk λu l,l. 2.7 ρ h u D,j,j 2.8 where e descrbes the nternal energy. The energy equaton for the vbratonal energy e vb n the case of vbratonal nonequlbrum s as follows e vb t + e vb u j + u D j = q vb j + Q T V + Q chem. 2.9 For the equlbrum case,the vbratonal temperature T vb s equal to the translatonal temperature T and eq s used wth T replacng T vb. The nternal energy for the complete system s a sum of the speces nternal energes takng nto account ther concentratons, e = c e The equlbrum nternal energy for one speces conssts of the translatonal,rotatonal and vbratonal energy and the heat of formaton. Note that atoms N and O delver no vbratonal and rotatonal contrbuton to the nternal energy e = e trans T +e rot T +e vb T vb + h f The nternal energy contrbutons from translaton,rotaton and vbraton are assembled through the specfc heats at constant volume as e = c trans v, T + c rot v, T + c vb v, T vb + h f The enthalpy s expressed as h = c trans p, T + c rot p, T + c vb p, + h f The nternal energy and enthalpy are connected through h = e + p ρ. 2.14
3 DNS of hypersonc transton 145 Mass Fracton c N O O N NO Temperature [K] Fgure 1. Composton of equlbrum ar at 1 atm. The flud s treated as an deal gas,where the followng equaton holds p = p = R ρ T M For the dffuson veloctes u D,Fck s law of dffuson s employed ρ u D j = ρd c, 2.16 where the dffuson coeffcent s ndependent of the speces. The translatonal and the vbratonal heat conducton s descrbed through Fourer s law q j = κ T, q vb j vb Tvb = κ Chemcal Modelng A fve speces N 2, O 2, N, O, NO model for ar wll be appled. The equlbrum composton for ar at constant pressure over temperature s shown n Fg. 1. The reacton rates k f and k b are modeled n an Arrhenus manner accordng to Park The model proposed by Park takes nto account the translatonal as well as the vbratonal temperature T vb for each speces. The vbratonal temperature descrbes the vbratonal relaxaton,whereas a translatonal temperature ncludes the rotatonal relaxaton,whch s assumed to take place nstantly. It only takes 9-12 molecule collsons for the rotatonal relaxaton to complete,whereas the vbratonal relaxaton takes 10 5 molecule collsons to reach a steady state the same order of magntude as for the chemcal relaxaton. The seventeen chemcal reactons thought to be suffcent for the modelng of ar under the condtons of nterest are as follows: The reacton partner M represents any of the
4 146 C. Stemmer & N. N. Mansour fve speces consdered; see Park,1989. N 2 +M N+N+M reac. 1 O 2 +M O+O+M reac. 2 NO+M N+O+M reac N 2 +O NO+N reac. 4 NO+O N+O 2 reac. 5 wth the producton terms M N2,M O2,M NO,M N andm O represent the speces masses : W N2 = M N2 R 1 + R 4 W O2 = M O2 R 2 R 5 W NO = M NO R 3 R 4 + R W N = M N 2R 1 R 3 R 4 R 5 W O = M O 2R 2 R 3 + R 4 + R 5 where R 1 = k f,1 ρn2 R 5 = k f,5 ρno M NO M N2 ρ M ρo2 ρ k f,2 M O2 M O + + k b,1 ρn M N 2 ρ M 2 ρo ρ k b,2 M O M R 2 = M R 3 = ρno ρ k f,3 + ρn k b,3 M NO M ρn2 ρo ρno R 4 = k f,4 + k b,4 M N2 M O ρo ρo2 + k b,5 M O2 M N ρn M NO M N ρn M N ρo ρ, M O and the forward reacton rates k f for the fve reactons consdered are M 2.20 k f,1 = TT vb 3/2 exp 59, 500/ TT vb for M = molecule k f,1 = TT vb 3/2 exp 59, 500/ TT vb for M = atom k f,2 = TT vb 8/5 exp 113, 200/ TT vb for M = molecule k f,2 = TT vb 8/5 exp 113, 200/ TT vb for M = atom k f,3 = exp 75, 500/ TT vb for M= N 2,O k f,3 = exp 75, 500/ TT vb for M= N,O,NO k f,4 = TT vb 1 exp 38, 370/ TT vb k f,5 = exp 19, 450/ TT vb. The backward reacton rates k b are calculated from the equlbrum rates through k b, = k f, /K eq, 2.22
5 DNS of hypersonc transton 147 The equlbrum rates are defned as K eq,1 =exp TT vb /10, log 10 10, 000/ TT vb , 000/ TT vb /TT vb K eq,2 =exp TT vb /10, log 10 10, 000/ TT vb , 000/ TT vb /TT vb K eq,3 =exp TT vb /10, log 10 10, 000/ TT vb , 000/ TT vb /TT vb 2.23 K eq,4 =exp TT vb /10, log 10 10, 000/ TT vb , 000/ TT vb /TT vb K eq,5 =exp TT vb /10, log 10 10, 000/ TT vb , 000/ TT vb /TT vb 2.2. Modelng of physcal and transport propertes The followng relatons are for a mxture of chemcally-reactng gases Specfc heat at constant volume The specfc heat at constant volume c v for atoms s descrbed through: c v, = c trans v, = 3 2 R The partal dervatves of the speces concentratons wth respect to the temperature are the contrbutons due to chemcal reactons. The specfc heat at constant volume c v for molecules Vncent & Kruger 1982 s made up as follows, where Θ vb c v, = c trans v, + c rot v, + c vb v, = 3 2 R + R + Θvb /T vb 2 e Θvb /T vb e Θvb /T vb 1 2 R, 2.25 s the characterstc temperature of vbraton of the molecular speces Specfc heat at constant pressure The specfc heat at constant pressure c p s descrbed by: c p, = c v, + R T Vscosty Blottner s formula wll be employed for the modelng of the vscosty Blottner,Johnson & Ells Ths approxmate formula s vald up to 10,000 K, far exceedng the temperature range of the flows nvestgated here. The coeffcents A µ,b µ and C µ are gven by Blottner et al. µ =0.1 exp [C µ + ln T B µ +lnt A µ ]. 2.27
6 148 C. Stemmer & N. N. Mansour Thermal conductvty The speces thermal conductvtes are descrbed employng Eucken s correcton,gven as Hrschfelder,Curtss & Brd 1964: κ = µ 5 2 ctrans v, + c rot v,, κ vb = µ c vb v, Mxng rules for vscosty and thermal conductvty The mxng rule n a mxture of gases,accordng to Wlke 1950,s n x µ µ mx n j=1 x 2.29 jφ j wth =1 [ 1+µ /µ j 1/2 M j /M 1/4] 2 Φ j = 8 + 8M /M j 1/2 and x = c /M n j=1 c j/m j. The same formula apples for the thermal conductvtes,replacng the vscosty µ by the thermal conductvty k. Further detals of the physcal modelng can be found,for example,n Sarma Dffuson coeffcent A constant Schmdt number Sc = 0.5 s assumed Hudson 1996 whch yelds for the dffuson coeffcent: D = µ ρsc = 2 µ 2.30 ρ Translatonal-vbratonal energy exchange Vbratonal energy s present only n the molecular speces N 2,O 2 and NO,whch are all modeled as harmonc oscllators. Therefore the followng equatons are vald. In case of the ncorporaton of anharmonc oscllatory molecules lke CO 2,dfferent relaxaton and energy expressons have to be appled Vncent & Kruger The translatonal-vbratonal energy exchange s descrbed through a Landau-Teller relaxaton model Vncent & Kruger 1982 as, Q T V = e vb,eq T e vb T vb c, 2.31 τ where the relaxaton tmes are determned for each speces as τ = 1 p C 1 expc 2 /T 1/3, 2.32 and the nonequlbrum vbratonal energy depends on the vbratonal temperature as e vb = Θvb /T vb e Θvb The equlbrum value for the vbratonal energy e vb,eq T replacng T vb. /T vb 1 R T vb follows the same expresson,wth
7 DNS of hypersonc transton 149 β arctan1/ma chem. reac. effects shock Ma>>1 b.l. δ/x Ma 2 / Re x Fgure 2. Schematc of shock locaton and boundary-layer edge for hypersonc boundary layers on a flat plate, showng dependence on Mach number The chemcal source term n Eq. 2.9 s expressed as the sum over the vbratonal nternal energy multpled wth the producton terms: Q chem = c e vb W Future Work A spatal fnte-dfference DNS code wll be appled on a Cartesan three-dmensonal grd on a flat plate. The code wll ncorporate a shock-capturng technque,snce the shock provoked by the flat-plate leadng edge s the major source of nonequlbrum. For the hgh Mach numbers,the locaton of the shock and the boundary-layer edge,whch s the area of lnear nstablty for hypersonc flows,merge,and the chemcal and thermal nonequlbrum n ths regon s expected to nfluence transton to a large extent Fg. 2; see also Anderson,1989. For the flght condtons nvestgated,the data n Fg. 3 are relevant. At a speed of V =5.9 Km/s,dssocaton of ntrogen and oxygen can be expected. For an alttude of h = 25 Km,chemcal and thermal equlbrum wll persst at a Mach number Ma = 20. At an alttude of about h = 100 Km Ma=20.8,full nonequlbrum condtons are present. Condtons are chosen such that onzaton wll not take place. Ths choce s consstent wth the return path of the shuttle as t enters the athmosphere.
8 150 C. Stemmer & N. N. Mansour Fgure 3. Flow regmes and thermochemcal phenomena n the stagnaton regon of a 30.5 cm radus sphere flyng n ar Gupta et al REFERENCES Anderson, J. D Hypersonc and and Hgh Temperature Gas Dynamcs. AIAA publcaton. Blottner, F. G., Johnson, M. & Ells, M Chemcally reactng vscous flow program for mult-component gas mxtures. Sanda Natl. Laboratores,SC-RR Gupta, R. N., Yos, M. J., Thompson, R. A. & Lee, K.-P A revew of reacton rates and thermodynamc and transport ropertes for an 11-speces ar Model for chemcal and thermal nonequlbrum calculatons to 30,000K. NASA RP Hrschfelder, J. O., Curtss, C. F. & Brd, R. A Molecular Theory of Gases and Lquds. Wley & Sons,New York. Hudson, M. J Lnear Stablty of Hypersonc Flows n Thermal and Chemcal Nonequlbrum. Ph.D. Thess,North Carolna State Unversty,Ralegh,NC. Mronov, S. G. & Maslov, A. A.,Expermental study of secondary stablty n a hypersonc shock layer on a flat plate. J. Flud Mech. 412, Park, C A revew of reacton rates n hgh temperature ar. AIAA Paper Sarma,G.S.R.2000 Physco-chemcal modelng n hypersonc flow smulaton. Prog. Aerospace Sc. 36, Schneder, S. P Flght data for boundary-layer transton at hypersonc and supersonc speeds. J. Spacecraft and Rockets 36,8-20. Vncent, W. G. & Kruger. C. H Introducton to Physcal Gas Dynamcs. Kreger,Malabar,FL. Wlke, S. P A Vscosty Equaton for Gas Mxtures. J. Comp. Phys. 18,
Internal energy excitation and dissociation of molecular nitrogen in a compressing flow
 Center for Turbulence Research Annual Research Brefs 29 59 Internal energy exctaton and dssocaton of molecular ntrogen n a compressng flow By T. E. Magn, M. Panes, A. Bourdon, R. Jaffe AND D. Schwenke
Center for Turbulence Research Annual Research Brefs 29 59 Internal energy exctaton and dssocaton of molecular ntrogen n a compressng flow By T. E. Magn, M. Panes, A. Bourdon, R. Jaffe AND D. Schwenke
Basic concept of reactive flows. Basic concept of reactive flows Combustion Mixing and reaction in high viscous fluid Application of Chaos
 Introducton to Toshhsa Ueda School of Scence for Open and Envronmental Systems Keo Unversty, Japan Combuston Mxng and reacton n hgh vscous flud Applcaton of Chaos Keo Unversty 1 Keo Unversty 2 What s reactve
Introducton to Toshhsa Ueda School of Scence for Open and Envronmental Systems Keo Unversty, Japan Combuston Mxng and reacton n hgh vscous flud Applcaton of Chaos Keo Unversty 1 Keo Unversty 2 What s reactve
Numerical Heat and Mass Transfer
 Master degree n Mechancal Engneerng Numercal Heat and Mass Transfer 06-Fnte-Dfference Method (One-dmensonal, steady state heat conducton) Fausto Arpno f.arpno@uncas.t Introducton Why we use models and
Master degree n Mechancal Engneerng Numercal Heat and Mass Transfer 06-Fnte-Dfference Method (One-dmensonal, steady state heat conducton) Fausto Arpno f.arpno@uncas.t Introducton Why we use models and
Module 1 : The equation of continuity. Lecture 1: Equation of Continuity
 1 Module 1 : The equaton of contnuty Lecture 1: Equaton of Contnuty 2 Advanced Heat and Mass Transfer: Modules 1. THE EQUATION OF CONTINUITY : Lectures 1-6 () () () (v) (v) Overall Mass Balance Momentum
1 Module 1 : The equaton of contnuty Lecture 1: Equaton of Contnuty 2 Advanced Heat and Mass Transfer: Modules 1. THE EQUATION OF CONTINUITY : Lectures 1-6 () () () (v) (v) Overall Mass Balance Momentum
( ) 1/ 2. ( P SO2 )( P O2 ) 1/ 2.
 Chemstry 360 Dr. Jean M. Standard Problem Set 9 Solutons. The followng chemcal reacton converts sulfur doxde to sulfur troxde. SO ( g) + O ( g) SO 3 ( l). (a.) Wrte the expresson for K eq for ths reacton.
Chemstry 360 Dr. Jean M. Standard Problem Set 9 Solutons. The followng chemcal reacton converts sulfur doxde to sulfur troxde. SO ( g) + O ( g) SO 3 ( l). (a.) Wrte the expresson for K eq for ths reacton.
Thermodynamics General
 Thermodynamcs General Lecture 1 Lecture 1 s devoted to establshng buldng blocks for dscussng thermodynamcs. In addton, the equaton of state wll be establshed. I. Buldng blocks for thermodynamcs A. Dmensons,
Thermodynamcs General Lecture 1 Lecture 1 s devoted to establshng buldng blocks for dscussng thermodynamcs. In addton, the equaton of state wll be establshed. I. Buldng blocks for thermodynamcs A. Dmensons,
Appendix II Summary of Important Equations
 W. M. Whte Geochemstry Equatons of State: Ideal GasLaw: Coeffcent of Thermal Expanson: Compressblty: Van der Waals Equaton: The Laws of Thermdynamcs: Frst Law: Appendx II Summary of Important Equatons
W. M. Whte Geochemstry Equatons of State: Ideal GasLaw: Coeffcent of Thermal Expanson: Compressblty: Van der Waals Equaton: The Laws of Thermdynamcs: Frst Law: Appendx II Summary of Important Equatons
Open Systems: Chemical Potential and Partial Molar Quantities Chemical Potential
 Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
The ChemSep Book. Harry A. Kooijman Consultant. Ross Taylor Clarkson University, Potsdam, New York University of Twente, Enschede, The Netherlands
 The ChemSep Book Harry A. Koojman Consultant Ross Taylor Clarkson Unversty, Potsdam, New York Unversty of Twente, Enschede, The Netherlands Lbr Books on Demand www.bod.de Copyrght c 2000 by H.A. Koojman
The ChemSep Book Harry A. Koojman Consultant Ross Taylor Clarkson Unversty, Potsdam, New York Unversty of Twente, Enschede, The Netherlands Lbr Books on Demand www.bod.de Copyrght c 2000 by H.A. Koojman
Transfer Functions. Convenient representation of a linear, dynamic model. A transfer function (TF) relates one input and one output: ( ) system
 Transfer Functons Convenent representaton of a lnear, dynamc model. A transfer functon (TF) relates one nput and one output: x t X s y t system Y s The followng termnology s used: x y nput output forcng
Transfer Functons Convenent representaton of a lnear, dynamc model. A transfer functon (TF) relates one nput and one output: x t X s y t system Y s The followng termnology s used: x y nput output forcng
and Statistical Mechanics Material Properties
 Statstcal Mechancs and Materal Propertes By Kuno TAKAHASHI Tokyo Insttute of Technology, Tokyo 15-855, JAPA Phone/Fax +81-3-5734-3915 takahak@de.ttech.ac.jp http://www.de.ttech.ac.jp/~kt-lab/ Only for
Statstcal Mechancs and Materal Propertes By Kuno TAKAHASHI Tokyo Insttute of Technology, Tokyo 15-855, JAPA Phone/Fax +81-3-5734-3915 takahak@de.ttech.ac.jp http://www.de.ttech.ac.jp/~kt-lab/ Only for
Problem Points Score Total 100
 Physcs 450 Solutons of Sample Exam I Problem Ponts Score 1 8 15 3 17 4 0 5 0 Total 100 All wor must be shown n order to receve full credt. Wor must be legble and comprehensble wth answers clearly ndcated.
Physcs 450 Solutons of Sample Exam I Problem Ponts Score 1 8 15 3 17 4 0 5 0 Total 100 All wor must be shown n order to receve full credt. Wor must be legble and comprehensble wth answers clearly ndcated.
Introduction to Vapor/Liquid Equilibrium, part 2. Raoult s Law:
 CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
Solution Thermodynamics
 Soluton hermodynamcs usng Wagner Notaton by Stanley. Howard Department of aterals and etallurgcal Engneerng South Dakota School of nes and echnology Rapd Cty, SD 57701 January 7, 001 Soluton hermodynamcs
Soluton hermodynamcs usng Wagner Notaton by Stanley. Howard Department of aterals and etallurgcal Engneerng South Dakota School of nes and echnology Rapd Cty, SD 57701 January 7, 001 Soluton hermodynamcs
modeling of equilibrium and dynamic multi-component adsorption in a two-layered fixed bed for purification of hydrogen from methane reforming products
 modelng of equlbrum and dynamc mult-component adsorpton n a two-layered fxed bed for purfcaton of hydrogen from methane reformng products Mohammad A. Ebrahm, Mahmood R. G. Arsalan, Shohreh Fatem * Laboratory
modelng of equlbrum and dynamc mult-component adsorpton n a two-layered fxed bed for purfcaton of hydrogen from methane reformng products Mohammad A. Ebrahm, Mahmood R. G. Arsalan, Shohreh Fatem * Laboratory
Appendix B. The Finite Difference Scheme
 140 APPENDIXES Appendx B. The Fnte Dfference Scheme In ths appendx we present numercal technques whch are used to approxmate solutons of system 3.1 3.3. A comprehensve treatment of theoretcal and mplementaton
140 APPENDIXES Appendx B. The Fnte Dfference Scheme In ths appendx we present numercal technques whch are used to approxmate solutons of system 3.1 3.3. A comprehensve treatment of theoretcal and mplementaton
Airflow and Contaminant Simulation with CONTAM
 Arflow and Contamnant Smulaton wth CONTAM George Walton, NIST CHAMPS Developers Workshop Syracuse Unversty June 19, 2006 Network Analogy Electrc Ppe, Duct & Ar Wre Ppe, Duct, or Openng Juncton Juncton
Arflow and Contamnant Smulaton wth CONTAM George Walton, NIST CHAMPS Developers Workshop Syracuse Unversty June 19, 2006 Network Analogy Electrc Ppe, Duct & Ar Wre Ppe, Duct, or Openng Juncton Juncton
If two volatile and miscible liquids are combined to form a solution, Raoult s law is not obeyed. Use the experimental data in Table 9.
 9.9 Real Solutons Exhbt Devatons from Raoult s Law If two volatle and mscble lquds are combned to form a soluton, Raoult s law s not obeyed. Use the expermental data n Table 9.3: Physcal Chemstry 00 Pearson
9.9 Real Solutons Exhbt Devatons from Raoult s Law If two volatle and mscble lquds are combned to form a soluton, Raoult s law s not obeyed. Use the expermental data n Table 9.3: Physcal Chemstry 00 Pearson
Turbulent Flow. Turbulent Flow
 http://www.youtube.com/watch?v=xoll2kedog&feature=related http://br.youtube.com/watch?v=7kkftgx2any http://br.youtube.com/watch?v=vqhxihpvcvu 1. Caothc fluctuatons wth a wde range of frequences and
http://www.youtube.com/watch?v=xoll2kedog&feature=related http://br.youtube.com/watch?v=7kkftgx2any http://br.youtube.com/watch?v=vqhxihpvcvu 1. Caothc fluctuatons wth a wde range of frequences and
5.62 Physical Chemistry II Spring 2008
 MIT OpenCourseWare http://ocw.mt.edu 5.62 Physcal Chemstry II Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.62 Sprng 2008 Lecture 34 Page Transton
MIT OpenCourseWare http://ocw.mt.edu 5.62 Physcal Chemstry II Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.62 Sprng 2008 Lecture 34 Page Transton
A quote of the week (or camel of the week): There is no expedience to which a man will not go to avoid the labor of thinking. Thomas A.
 A quote of the week (or camel of the week): here s no expedence to whch a man wll not go to avod the labor of thnkng. homas A. Edson Hess law. Algorthm S Select a reacton, possbly contanng specfc compounds
A quote of the week (or camel of the week): here s no expedence to whch a man wll not go to avod the labor of thnkng. homas A. Edson Hess law. Algorthm S Select a reacton, possbly contanng specfc compounds
CHEMICAL REACTIONS AND DIFFUSION
 CHEMICAL REACTIONS AND DIFFUSION A.K.A. NETWORK THERMODYNAMICS BACKGROUND Classcal thermodynamcs descrbes equlbrum states. Non-equlbrum thermodynamcs descrbes steady states. Network thermodynamcs descrbes
CHEMICAL REACTIONS AND DIFFUSION A.K.A. NETWORK THERMODYNAMICS BACKGROUND Classcal thermodynamcs descrbes equlbrum states. Non-equlbrum thermodynamcs descrbes steady states. Network thermodynamcs descrbes
Numerical Study of Propane-Air Mixture Combustion in a Burner Element
 Defect and Dffuson Forum Vols. 73-76 (8 pp 144-149 onlne at http://www.scentfc.net (8 Trans Tech Publcatons, Swtzerland Onlne avalable snce 8/Feb/11 Numercal Study of Propane-Ar Mxture Combuston n a Burner
Defect and Dffuson Forum Vols. 73-76 (8 pp 144-149 onlne at http://www.scentfc.net (8 Trans Tech Publcatons, Swtzerland Onlne avalable snce 8/Feb/11 Numercal Study of Propane-Ar Mxture Combuston n a Burner
Normally, in one phase reservoir simulation we would deal with one of the following fluid systems:
 TPG4160 Reservor Smulaton 2017 page 1 of 9 ONE-DIMENSIONAL, ONE-PHASE RESERVOIR SIMULATION Flud systems The term sngle phase apples to any system wth only one phase present n the reservor In some cases
TPG4160 Reservor Smulaton 2017 page 1 of 9 ONE-DIMENSIONAL, ONE-PHASE RESERVOIR SIMULATION Flud systems The term sngle phase apples to any system wth only one phase present n the reservor In some cases
...Thermodynamics. If Clausius Clapeyron fails. l T (v 2 v 1 ) = 0/0 Second order phase transition ( S, v = 0)
 If Clausus Clapeyron fals ( ) dp dt pb =...Thermodynamcs l T (v 2 v 1 ) = 0/0 Second order phase transton ( S, v = 0) ( ) dp = c P,1 c P,2 dt Tv(β 1 β 2 ) Two phases ntermngled Ferromagnet (Excess spn-up
If Clausus Clapeyron fals ( ) dp dt pb =...Thermodynamcs l T (v 2 v 1 ) = 0/0 Second order phase transton ( S, v = 0) ( ) dp = c P,1 c P,2 dt Tv(β 1 β 2 ) Two phases ntermngled Ferromagnet (Excess spn-up
Three-Phase Distillation in Packed Towers: Short-Cut Modelling and Parameter Tuning
 European Symposum on Computer Arded Aded Process Engneerng 15 L. Pugjaner and A. Espuña (Edtors) 2005 Elsever Scence B.V. All rghts reserved. Three-Phase Dstllaton n Packed Towers: Short-Cut Modellng and
European Symposum on Computer Arded Aded Process Engneerng 15 L. Pugjaner and A. Espuña (Edtors) 2005 Elsever Scence B.V. All rghts reserved. Three-Phase Dstllaton n Packed Towers: Short-Cut Modellng and
THE VIBRATIONS OF MOLECULES II THE CARBON DIOXIDE MOLECULE Student Instructions
 THE VIBRATIONS OF MOLECULES II THE CARBON DIOXIDE MOLECULE Student Instructons by George Hardgrove Chemstry Department St. Olaf College Northfeld, MN 55057 hardgrov@lars.acc.stolaf.edu Copyrght George
THE VIBRATIONS OF MOLECULES II THE CARBON DIOXIDE MOLECULE Student Instructons by George Hardgrove Chemstry Department St. Olaf College Northfeld, MN 55057 hardgrov@lars.acc.stolaf.edu Copyrght George
Numerical Transient Heat Conduction Experiment
 Numercal ransent Heat Conducton Experment OBJECIVE 1. o demonstrate the basc prncples of conducton heat transfer.. o show how the thermal conductvty of a sold can be measured. 3. o demonstrate the use
Numercal ransent Heat Conducton Experment OBJECIVE 1. o demonstrate the basc prncples of conducton heat transfer.. o show how the thermal conductvty of a sold can be measured. 3. o demonstrate the use
Supplementary Notes for Chapter 9 Mixture Thermodynamics
 Supplementary Notes for Chapter 9 Mxture Thermodynamcs Key ponts Nne major topcs of Chapter 9 are revewed below: 1. Notaton and operatonal equatons for mxtures 2. PVTN EOSs for mxtures 3. General effects
Supplementary Notes for Chapter 9 Mxture Thermodynamcs Key ponts Nne major topcs of Chapter 9 are revewed below: 1. Notaton and operatonal equatons for mxtures 2. PVTN EOSs for mxtures 3. General effects
Lecture 5.8 Flux Vector Splitting
 Lecture 5.8 Flux Vector Splttng 1 Flux Vector Splttng The vector E n (5.7.) can be rewrtten as E = AU (5.8.1) (wth A as gven n (5.7.4) or (5.7.6) ) whenever, the equaton of state s of the separable form
Lecture 5.8 Flux Vector Splttng 1 Flux Vector Splttng The vector E n (5.7.) can be rewrtten as E = AU (5.8.1) (wth A as gven n (5.7.4) or (5.7.6) ) whenever, the equaton of state s of the separable form
Lecture. Polymer Thermodynamics 0331 L Chemical Potential
 Prof. Dr. rer. nat. habl. S. Enders Faculty III for Process Scence Insttute of Chemcal Engneerng Department of Thermodynamcs Lecture Polymer Thermodynamcs 033 L 337 3. Chemcal Potental Polymer Thermodynamcs
Prof. Dr. rer. nat. habl. S. Enders Faculty III for Process Scence Insttute of Chemcal Engneerng Department of Thermodynamcs Lecture Polymer Thermodynamcs 033 L 337 3. Chemcal Potental Polymer Thermodynamcs
5.04, Principles of Inorganic Chemistry II MIT Department of Chemistry Lecture 32: Vibrational Spectroscopy and the IR
 5.0, Prncples of Inorganc Chemstry II MIT Department of Chemstry Lecture 3: Vbratonal Spectroscopy and the IR Vbratonal spectroscopy s confned to the 00-5000 cm - spectral regon. The absorpton of a photon
5.0, Prncples of Inorganc Chemstry II MIT Department of Chemstry Lecture 3: Vbratonal Spectroscopy and the IR Vbratonal spectroscopy s confned to the 00-5000 cm - spectral regon. The absorpton of a photon
A PROCEDURE FOR SIMULATING THE NONLINEAR CONDUCTION HEAT TRANSFER IN A BODY WITH TEMPERATURE DEPENDENT THERMAL CONDUCTIVITY.
 Proceedngs of the th Brazlan Congress of Thermal Scences and Engneerng -- ENCIT 006 Braz. Soc. of Mechancal Scences and Engneerng -- ABCM, Curtba, Brazl,- Dec. 5-8, 006 A PROCEDURE FOR SIMULATING THE NONLINEAR
Proceedngs of the th Brazlan Congress of Thermal Scences and Engneerng -- ENCIT 006 Braz. Soc. of Mechancal Scences and Engneerng -- ABCM, Curtba, Brazl,- Dec. 5-8, 006 A PROCEDURE FOR SIMULATING THE NONLINEAR
Turbulent Nonpremixed Flames
 School of Aerospace Engneerng Turbulent Nonpremxed Flames Jerry Setzman. 5 Mole Fracton.15.1.5 CH4 HO HCO x 1 Temperature Methane Flame.1..3 Dstance (cm) 15 1 5 Temperature (K) TurbulentNonpremxed -1 School
School of Aerospace Engneerng Turbulent Nonpremxed Flames Jerry Setzman. 5 Mole Fracton.15.1.5 CH4 HO HCO x 1 Temperature Methane Flame.1..3 Dstance (cm) 15 1 5 Temperature (K) TurbulentNonpremxed -1 School
CHEMICAL ENGINEERING
 Postal Correspondence GATE & PSUs -MT To Buy Postal Correspondence Packages call at 0-9990657855 1 TABLE OF CONTENT S. No. Ttle Page no. 1. Introducton 3 2. Dffuson 10 3. Dryng and Humdfcaton 24 4. Absorpton
Postal Correspondence GATE & PSUs -MT To Buy Postal Correspondence Packages call at 0-9990657855 1 TABLE OF CONTENT S. No. Ttle Page no. 1. Introducton 3 2. Dffuson 10 3. Dryng and Humdfcaton 24 4. Absorpton
3. Be able to derive the chemical equilibrium constants from statistical mechanics.
 Lecture #17 1 Lecture 17 Objectves: 1. Notaton of chemcal reactons 2. General equlbrum 3. Be able to derve the chemcal equlbrum constants from statstcal mechancs. 4. Identfy how nondeal behavor can be
Lecture #17 1 Lecture 17 Objectves: 1. Notaton of chemcal reactons 2. General equlbrum 3. Be able to derve the chemcal equlbrum constants from statstcal mechancs. 4. Identfy how nondeal behavor can be
Process Modeling. Improving or understanding chemical process operation is a major objective for developing a dynamic process model
 Process Modelng Improvng or understandng chemcal process operaton s a major objectve for developng a dynamc process model Balance equatons Steady-state balance equatons mass or energy mass or energy enterng
Process Modelng Improvng or understandng chemcal process operaton s a major objectve for developng a dynamc process model Balance equatons Steady-state balance equatons mass or energy mass or energy enterng
Lecture 12. Modeling of Turbulent Combustion
 Lecture 12. Modelng of Turbulent Combuston X.S. Ba Modelng of TC Content drect numercal smulaton (DNS) Statstcal approach (RANS) Modelng of turbulent non-premxed flames Modelng of turbulent premxed flames
Lecture 12. Modelng of Turbulent Combuston X.S. Ba Modelng of TC Content drect numercal smulaton (DNS) Statstcal approach (RANS) Modelng of turbulent non-premxed flames Modelng of turbulent premxed flames
Thermal-Fluids I. Chapter 18 Transient heat conduction. Dr. Primal Fernando Ph: (850)
 hermal-fluds I Chapter 18 ransent heat conducton Dr. Prmal Fernando prmal@eng.fsu.edu Ph: (850) 410-6323 1 ransent heat conducton In general, he temperature of a body vares wth tme as well as poston. In
hermal-fluds I Chapter 18 ransent heat conducton Dr. Prmal Fernando prmal@eng.fsu.edu Ph: (850) 410-6323 1 ransent heat conducton In general, he temperature of a body vares wth tme as well as poston. In
Pressure Measurements Laboratory
 Lab # Pressure Measurements Laboratory Objectves:. To get hands-on experences on how to make pressure (surface pressure, statc pressure and total pressure nsde flow) measurements usng conventonal pressuremeasurng
Lab # Pressure Measurements Laboratory Objectves:. To get hands-on experences on how to make pressure (surface pressure, statc pressure and total pressure nsde flow) measurements usng conventonal pressuremeasurng
Physics 240: Worksheet 30 Name:
 (1) One mole of an deal monatomc gas doubles ts temperature and doubles ts volume. What s the change n entropy of the gas? () 1 kg of ce at 0 0 C melts to become water at 0 0 C. What s the change n entropy
(1) One mole of an deal monatomc gas doubles ts temperature and doubles ts volume. What s the change n entropy of the gas? () 1 kg of ce at 0 0 C melts to become water at 0 0 C. What s the change n entropy
Mass and Heat Diffusion in Ternary Polymer Solutions: A Classical Irreversible Thermodynamics Approach
 Mass and Heat ffuson n Ternary Polymer Solutons: A Classcal Irreversble Thermodynamcs Approach S. Shams Es-hagh and M. Cakmak * epartment of Polymer Engneerng, 5 S. Forge St., The Unversty of Akron, Akron,
Mass and Heat ffuson n Ternary Polymer Solutons: A Classcal Irreversble Thermodynamcs Approach S. Shams Es-hagh and M. Cakmak * epartment of Polymer Engneerng, 5 S. Forge St., The Unversty of Akron, Akron,
Energy, Entropy, and Availability Balances Phase Equilibria. Nonideal Thermodynamic Property Models. Selecting an Appropriate Model
 Lecture 4. Thermodynamcs [Ch. 2] Energy, Entropy, and Avalablty Balances Phase Equlbra - Fugactes and actvty coeffcents -K-values Nondeal Thermodynamc Property Models - P-v-T equaton-of-state models -
Lecture 4. Thermodynamcs [Ch. 2] Energy, Entropy, and Avalablty Balances Phase Equlbra - Fugactes and actvty coeffcents -K-values Nondeal Thermodynamc Property Models - P-v-T equaton-of-state models -
THE IGNITION PARAMETER - A quantification of the probability of ignition
 THE IGNITION PARAMETER - A quantfcaton of the probablty of ton INFUB9-2011 Topc: Modellng of fundamental processes Man author Nels Bjarne K. Rasmussen Dansh Gas Technology Centre (DGC) NBR@dgc.dk Co-author
THE IGNITION PARAMETER - A quantfcaton of the probablty of ton INFUB9-2011 Topc: Modellng of fundamental processes Man author Nels Bjarne K. Rasmussen Dansh Gas Technology Centre (DGC) NBR@dgc.dk Co-author
4.2 Chemical Driving Force
 4.2. CHEMICL DRIVING FORCE 103 4.2 Chemcal Drvng Force second effect of a chemcal concentraton gradent on dffuson s to change the nature of the drvng force. Ths s because dffuson changes the bondng n a
4.2. CHEMICL DRIVING FORCE 103 4.2 Chemcal Drvng Force second effect of a chemcal concentraton gradent on dffuson s to change the nature of the drvng force. Ths s because dffuson changes the bondng n a
GENERAL EQUATIONS OF PHYSICO-CHEMICAL
 GENERAL EQUATIONS OF PHYSICO-CHEMICAL PROCESSES Causes and conons for the evoluton of a system... 1 Integral formulaton of balance equatons... 2 Dfferental formulaton of balance equatons... 3 Boundary
GENERAL EQUATIONS OF PHYSICO-CHEMICAL PROCESSES Causes and conons for the evoluton of a system... 1 Integral formulaton of balance equatons... 2 Dfferental formulaton of balance equatons... 3 Boundary
COMPOSITE BEAM WITH WEAK SHEAR CONNECTION SUBJECTED TO THERMAL LOAD
 COMPOSITE BEAM WITH WEAK SHEAR CONNECTION SUBJECTED TO THERMAL LOAD Ákos Jósef Lengyel, István Ecsed Assstant Lecturer, Professor of Mechancs, Insttute of Appled Mechancs, Unversty of Mskolc, Mskolc-Egyetemváros,
COMPOSITE BEAM WITH WEAK SHEAR CONNECTION SUBJECTED TO THERMAL LOAD Ákos Jósef Lengyel, István Ecsed Assstant Lecturer, Professor of Mechancs, Insttute of Appled Mechancs, Unversty of Mskolc, Mskolc-Egyetemváros,
University of Washington Department of Chemistry Chemistry 452/456 Summer Quarter 2014
 Lecture 16 8/4/14 Unversty o Washngton Department o Chemstry Chemstry 452/456 Summer Quarter 214. Real Vapors and Fugacty Henry s Law accounts or the propertes o extremely dlute soluton. s shown n Fgure
Lecture 16 8/4/14 Unversty o Washngton Department o Chemstry Chemstry 452/456 Summer Quarter 214. Real Vapors and Fugacty Henry s Law accounts or the propertes o extremely dlute soluton. s shown n Fgure
5.60 Thermodynamics & Kinetics Spring 2008
 MIT OpenCourseWare http://ocw.mt.edu 5.60 Thermodynamcs & Knetcs Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.60 Sprng 2008 Lecture #29 page 1
MIT OpenCourseWare http://ocw.mt.edu 5.60 Thermodynamcs & Knetcs Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.60 Sprng 2008 Lecture #29 page 1
A large scale tsunami run-up simulation and numerical evaluation of fluid force during tsunami by using a particle method
 A large scale tsunam run-up smulaton and numercal evaluaton of flud force durng tsunam by usng a partcle method *Mtsuteru Asa 1), Shoch Tanabe 2) and Masaharu Isshk 3) 1), 2) Department of Cvl Engneerng,
A large scale tsunam run-up smulaton and numercal evaluaton of flud force durng tsunam by usng a partcle method *Mtsuteru Asa 1), Shoch Tanabe 2) and Masaharu Isshk 3) 1), 2) Department of Cvl Engneerng,
is the calculated value of the dependent variable at point i. The best parameters have values that minimize the squares of the errors
 Multple Lnear and Polynomal Regresson wth Statstcal Analyss Gven a set of data of measured (or observed) values of a dependent varable: y versus n ndependent varables x 1, x, x n, multple lnear regresson
Multple Lnear and Polynomal Regresson wth Statstcal Analyss Gven a set of data of measured (or observed) values of a dependent varable: y versus n ndependent varables x 1, x, x n, multple lnear regresson
Wilbur and Ague 4 WILBUR AND AGUE; APPENDIX DR1. Two-dimensional chemical maps as well as chemical profiles were done at 15 kv using
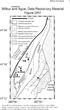 DR2006139 Wlbur and Ague 4 WILBUR AND AGUE; APPENDIX DR1 MINERAL ANALYSES Two-dmensonal chemcal maps as well as chemcal profles were done at 15 kv usng the JEOL JXA-8600 electron mcroprobe at Yale Unversty
DR2006139 Wlbur and Ague 4 WILBUR AND AGUE; APPENDIX DR1 MINERAL ANALYSES Two-dmensonal chemcal maps as well as chemcal profles were done at 15 kv usng the JEOL JXA-8600 electron mcroprobe at Yale Unversty
Electrical double layer: revisit based on boundary conditions
 Electrcal double layer: revst based on boundary condtons Jong U. Km Department of Electrcal and Computer Engneerng, Texas A&M Unversty College Staton, TX 77843-318, USA Abstract The electrcal double layer
Electrcal double layer: revst based on boundary condtons Jong U. Km Department of Electrcal and Computer Engneerng, Texas A&M Unversty College Staton, TX 77843-318, USA Abstract The electrcal double layer
NAME and Section No. it is found that 0.6 mol of O
 NAME and Secton No. Chemstry 391 Fall 7 Exam III KEY 1. (3 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). In the reacton 3O O3 t s found that.6 mol of O are consumed. Fnd
NAME and Secton No. Chemstry 391 Fall 7 Exam III KEY 1. (3 Ponts) ***Do 5 out of 6***(If 6 are done only the frst 5 wll be graded)*** a). In the reacton 3O O3 t s found that.6 mol of O are consumed. Fnd
Week3, Chapter 4. Position and Displacement. Motion in Two Dimensions. Instantaneous Velocity. Average Velocity
 Week3, Chapter 4 Moton n Two Dmensons Lecture Quz A partcle confned to moton along the x axs moves wth constant acceleraton from x =.0 m to x = 8.0 m durng a 1-s tme nterval. The velocty of the partcle
Week3, Chapter 4 Moton n Two Dmensons Lecture Quz A partcle confned to moton along the x axs moves wth constant acceleraton from x =.0 m to x = 8.0 m durng a 1-s tme nterval. The velocty of the partcle
Physics 5153 Classical Mechanics. D Alembert s Principle and The Lagrangian-1
 P. Guterrez Physcs 5153 Classcal Mechancs D Alembert s Prncple and The Lagrangan 1 Introducton The prncple of vrtual work provdes a method of solvng problems of statc equlbrum wthout havng to consder the
P. Guterrez Physcs 5153 Classcal Mechancs D Alembert s Prncple and The Lagrangan 1 Introducton The prncple of vrtual work provdes a method of solvng problems of statc equlbrum wthout havng to consder the
The Advanced URANUS Navier-Stokes Code for the Simulation of Nonequilibrium Re-entry Flows
 Trans. JSASS Space Tech. Japan Vol. 7, No. sts26, pp. Pe_15-Pe_24, 29 The Advanced URANUS Naver-Stokes Code for the Smulaton of Nonequlbrum Re-entry Flows By Markus FERTIG 1 and Georg HERDRICH 1 1 Insttut
Trans. JSASS Space Tech. Japan Vol. 7, No. sts26, pp. Pe_15-Pe_24, 29 The Advanced URANUS Naver-Stokes Code for the Smulaton of Nonequlbrum Re-entry Flows By Markus FERTIG 1 and Georg HERDRICH 1 1 Insttut
A STUDY ON THE PHYSICS OF SUPERSONIC MIXING FIELD WITH INJECTION AT DIFFERENT ANGLES
 Proceedngs of the Internatonal Conference on Mechancal Engneerng 23 (ICME23) 26-28 December 23 Dhaka Bangladesh ICME3-FL-18 A STUDY ON THE PHYSICS OF SUPERSONIC MIXING FIELD WITH INJECTION AT DIFFERENT
Proceedngs of the Internatonal Conference on Mechancal Engneerng 23 (ICME23) 26-28 December 23 Dhaka Bangladesh ICME3-FL-18 A STUDY ON THE PHYSICS OF SUPERSONIC MIXING FIELD WITH INJECTION AT DIFFERENT
THE EFFECT OF TORSIONAL RIGIDITY BETWEEN ELEMENTS ON FREE VIBRATIONS OF A TELESCOPIC HYDRAULIC CYLINDER SUBJECTED TO EULER S LOAD
 Journal of Appled Mathematcs and Computatonal Mechancs 7, 6(3), 7- www.amcm.pcz.pl p-issn 99-9965 DOI:.75/jamcm.7.3. e-issn 353-588 THE EFFECT OF TORSIONAL RIGIDITY BETWEEN ELEMENTS ON FREE VIBRATIONS
Journal of Appled Mathematcs and Computatonal Mechancs 7, 6(3), 7- www.amcm.pcz.pl p-issn 99-9965 DOI:.75/jamcm.7.3. e-issn 353-588 THE EFFECT OF TORSIONAL RIGIDITY BETWEEN ELEMENTS ON FREE VIBRATIONS
Thermo-Calc Software. Modelling Multicomponent Precipitation Kinetics with CALPHAD-Based Tools. EUROMAT2013, September 8-13, 2013 Sevilla, Spain
 Modellng Multcomponent Precptaton Knetcs wth CALPHAD-Based Tools Kasheng Wu 1, Gustaf Sterner 2, Qng Chen 2, Åke Jansson 2, Paul Mason 1, Johan Bratberg 2 and Anders Engström 2 1 Inc., 2 AB EUROMAT2013,
Modellng Multcomponent Precptaton Knetcs wth CALPHAD-Based Tools Kasheng Wu 1, Gustaf Sterner 2, Qng Chen 2, Åke Jansson 2, Paul Mason 1, Johan Bratberg 2 and Anders Engström 2 1 Inc., 2 AB EUROMAT2013,
Be true to your work, your word, and your friend.
 Chemstry 13 NT Be true to your work, your word, and your frend. Henry Davd Thoreau 1 Chem 13 NT Chemcal Equlbrum Module Usng the Equlbrum Constant Interpretng the Equlbrum Constant Predctng the Drecton
Chemstry 13 NT Be true to your work, your word, and your frend. Henry Davd Thoreau 1 Chem 13 NT Chemcal Equlbrum Module Usng the Equlbrum Constant Interpretng the Equlbrum Constant Predctng the Drecton
Rigid body simulation
 Rgd bod smulaton Rgd bod smulaton Once we consder an object wth spacal etent, partcle sstem smulaton s no longer suffcent Problems Problems Unconstraned sstem rotatonal moton torques and angular momentum
Rgd bod smulaton Rgd bod smulaton Once we consder an object wth spacal etent, partcle sstem smulaton s no longer suffcent Problems Problems Unconstraned sstem rotatonal moton torques and angular momentum
between standard Gibbs free energies of formation for products and reactants, ΔG! R = ν i ΔG f,i, we
 hermodynamcs, Statstcal hermodynamcs, and Knetcs 4 th Edton,. Engel & P. ed Ch. 6 Part Answers to Selected Problems Q6.. Q6.4. If ξ =0. mole at equlbrum, the reacton s not ery far along. hus, there would
hermodynamcs, Statstcal hermodynamcs, and Knetcs 4 th Edton,. Engel & P. ed Ch. 6 Part Answers to Selected Problems Q6.. Q6.4. If ξ =0. mole at equlbrum, the reacton s not ery far along. hus, there would
Frequency dependence of the permittivity
 Frequency dependence of the permttvty February 7, 016 In materals, the delectrc constant and permeablty are actually frequency dependent. Ths does not affect our results for sngle frequency modes, but
Frequency dependence of the permttvty February 7, 016 In materals, the delectrc constant and permeablty are actually frequency dependent. Ths does not affect our results for sngle frequency modes, but
2 Finite difference basics
 Numersche Methoden 1, WS 11/12 B.J.P. Kaus 2 Fnte dfference bascs Consder the one- The bascs of the fnte dfference method are best understood wth an example. dmensonal transent heat conducton equaton T
Numersche Methoden 1, WS 11/12 B.J.P. Kaus 2 Fnte dfference bascs Consder the one- The bascs of the fnte dfference method are best understood wth an example. dmensonal transent heat conducton equaton T
Osmotic pressure and protein binding
 Osmotc pressure and proten bndng Igor R. Kuznetsov, KochLab Symposum talk 5/15/09 Today we take a closer look at one of the soluton thermodynamcs key ponts from Steve s presentaton. Here t s: d[ln(k off
Osmotc pressure and proten bndng Igor R. Kuznetsov, KochLab Symposum talk 5/15/09 Today we take a closer look at one of the soluton thermodynamcs key ponts from Steve s presentaton. Here t s: d[ln(k off
Physics 141. Lecture 14. Frank L. H. Wolfs Department of Physics and Astronomy, University of Rochester, Lecture 14, Page 1
 Physcs 141. Lecture 14. Frank L. H. Wolfs Department of Physcs and Astronomy, Unversty of Rochester, Lecture 14, Page 1 Physcs 141. Lecture 14. Course Informaton: Lab report # 3. Exam # 2. Mult-Partcle
Physcs 141. Lecture 14. Frank L. H. Wolfs Department of Physcs and Astronomy, Unversty of Rochester, Lecture 14, Page 1 Physcs 141. Lecture 14. Course Informaton: Lab report # 3. Exam # 2. Mult-Partcle
Mass Transfer Processes
 Mass Transfer Processes S. Majd Hassanzadeh Department of Earth Scences Faculty of Geoscences Utrecht Unversty Outlne: 1. Measures of Concentraton 2. Volatlzaton and Dssoluton 3. Adsorpton Processes 4.
Mass Transfer Processes S. Majd Hassanzadeh Department of Earth Scences Faculty of Geoscences Utrecht Unversty Outlne: 1. Measures of Concentraton 2. Volatlzaton and Dssoluton 3. Adsorpton Processes 4.
Visco-Rubber Elastic Model for Pressure Sensitive Adhesive
 Vsco-Rubber Elastc Model for Pressure Senstve Adhesve Kazuhsa Maeda, Shgenobu Okazawa, Koj Nshgch and Takash Iwamoto Abstract A materal model to descrbe large deformaton of pressure senstve adhesve (PSA
Vsco-Rubber Elastc Model for Pressure Senstve Adhesve Kazuhsa Maeda, Shgenobu Okazawa, Koj Nshgch and Takash Iwamoto Abstract A materal model to descrbe large deformaton of pressure senstve adhesve (PSA
A Self-Consistent Gibbs Excess Mixing Rule for Cubic Equations of State: derivation and fugacity coefficients
 A Self-Consstent Gbbs Excess Mxng Rule for Cubc Equatons of State: dervaton and fugacty coeffcents Paula B. Staudt, Rafael de P. Soares Departamento de Engenhara Químca, Escola de Engenhara, Unversdade
A Self-Consstent Gbbs Excess Mxng Rule for Cubc Equatons of State: dervaton and fugacty coeffcents Paula B. Staudt, Rafael de P. Soares Departamento de Engenhara Químca, Escola de Engenhara, Unversdade
Applied Nuclear Physics (Fall 2004) Lecture 23 (12/3/04) Nuclear Reactions: Energetics and Compound Nucleus
 .101 Appled Nuclear Physcs (Fall 004) Lecture 3 (1/3/04) Nuclear Reactons: Energetcs and Compound Nucleus References: W. E. Meyerhof, Elements of Nuclear Physcs (McGraw-Hll, New York, 1967), Chap 5. Among
.101 Appled Nuclear Physcs (Fall 004) Lecture 3 (1/3/04) Nuclear Reactons: Energetcs and Compound Nucleus References: W. E. Meyerhof, Elements of Nuclear Physcs (McGraw-Hll, New York, 1967), Chap 5. Among
Global Sensitivity. Tuesday 20 th February, 2018
 Global Senstvty Tuesday 2 th February, 28 ) Local Senstvty Most senstvty analyses [] are based on local estmates of senstvty, typcally by expandng the response n a Taylor seres about some specfc values
Global Senstvty Tuesday 2 th February, 28 ) Local Senstvty Most senstvty analyses [] are based on local estmates of senstvty, typcally by expandng the response n a Taylor seres about some specfc values
State-resolved models of chemical reactions for non-equilibrium flow simulations
 DOI: 10.1009/EUCASS2017-111 7 TH EUROPEAN CONFERENCE FOR AERONAUTICS AND AEROSPACE SCIENCES EUCASS State-resolved models of chemcal reactons for non-equlbrum flow smulatons E.V.Kustova and A.S.Savelev
DOI: 10.1009/EUCASS2017-111 7 TH EUROPEAN CONFERENCE FOR AERONAUTICS AND AEROSPACE SCIENCES EUCASS State-resolved models of chemcal reactons for non-equlbrum flow smulatons E.V.Kustova and A.S.Savelev
Implicit Integration Henyey Method
 Implct Integraton Henyey Method In realstc stellar evoluton codes nstead of a drect ntegraton usng for example the Runge-Kutta method one employs an teratve mplct technque. Ths s because the structure
Implct Integraton Henyey Method In realstc stellar evoluton codes nstead of a drect ntegraton usng for example the Runge-Kutta method one employs an teratve mplct technque. Ths s because the structure
Chemical Equilibrium. Chapter 6 Spontaneity of Reactive Mixtures (gases) Taking into account there are many types of work that a sysem can perform
 Ths chapter deals wth chemcal reactons (system) wth lttle or no consderaton on the surroundngs. Chemcal Equlbrum Chapter 6 Spontanety of eactve Mxtures (gases) eactants generatng products would proceed
Ths chapter deals wth chemcal reactons (system) wth lttle or no consderaton on the surroundngs. Chemcal Equlbrum Chapter 6 Spontanety of eactve Mxtures (gases) eactants generatng products would proceed
Mathematical Preparations
 1 Introducton Mathematcal Preparatons The theory of relatvty was developed to explan experments whch studed the propagaton of electromagnetc radaton n movng coordnate systems. Wthn expermental error the
1 Introducton Mathematcal Preparatons The theory of relatvty was developed to explan experments whch studed the propagaton of electromagnetc radaton n movng coordnate systems. Wthn expermental error the
The Discretization Process
 FMIA F Moukalled L Mangan M Darwsh An Advanced Introducton wth OpenFOAM and Matlab Ths textbook explores both the theoretcal foundaton of the Fnte Volume Method (FVM) and ts applcatons n Computatonal Flud
FMIA F Moukalled L Mangan M Darwsh An Advanced Introducton wth OpenFOAM and Matlab Ths textbook explores both the theoretcal foundaton of the Fnte Volume Method (FVM) and ts applcatons n Computatonal Flud
EN40: Dynamics and Vibrations. Homework 4: Work, Energy and Linear Momentum Due Friday March 1 st
 EN40: Dynamcs and bratons Homework 4: Work, Energy and Lnear Momentum Due Frday March 1 st School of Engneerng Brown Unversty 1. The fgure (from ths publcaton) shows the energy per unt area requred to
EN40: Dynamcs and bratons Homework 4: Work, Energy and Lnear Momentum Due Frday March 1 st School of Engneerng Brown Unversty 1. The fgure (from ths publcaton) shows the energy per unt area requred to
SUPPLEMENTARY INFORMATION
 do: 0.08/nature09 I. Resonant absorpton of XUV pulses n Kr + usng the reduced densty matrx approach The quantum beats nvestgated n ths paper are the result of nterference between two exctaton paths of
do: 0.08/nature09 I. Resonant absorpton of XUV pulses n Kr + usng the reduced densty matrx approach The quantum beats nvestgated n ths paper are the result of nterference between two exctaton paths of
The Multiple Classical Linear Regression Model (CLRM): Specification and Assumptions. 1. Introduction
 ECONOMICS 5* -- NOTE (Summary) ECON 5* -- NOTE The Multple Classcal Lnear Regresson Model (CLRM): Specfcaton and Assumptons. Introducton CLRM stands for the Classcal Lnear Regresson Model. The CLRM s also
ECONOMICS 5* -- NOTE (Summary) ECON 5* -- NOTE The Multple Classcal Lnear Regresson Model (CLRM): Specfcaton and Assumptons. Introducton CLRM stands for the Classcal Lnear Regresson Model. The CLRM s also
Electrostatic Potential from Transmembrane Currents
 Electrostatc Potental from Transmembrane Currents Let s assume that the current densty j(r, t) s ohmc;.e., lnearly proportonal to the electrc feld E(r, t): j = σ c (r)e (1) wth conductvty σ c = σ c (r).
Electrostatc Potental from Transmembrane Currents Let s assume that the current densty j(r, t) s ohmc;.e., lnearly proportonal to the electrc feld E(r, t): j = σ c (r)e (1) wth conductvty σ c = σ c (r).
Irregular vibrations in multi-mass discrete-continuous systems torsionally deformed
 (2) 4 48 Irregular vbratons n mult-mass dscrete-contnuous systems torsonally deformed Abstract In the paper rregular vbratons of dscrete-contnuous systems consstng of an arbtrary number rgd bodes connected
(2) 4 48 Irregular vbratons n mult-mass dscrete-contnuous systems torsonally deformed Abstract In the paper rregular vbratons of dscrete-contnuous systems consstng of an arbtrary number rgd bodes connected
(Online First)A Lattice Boltzmann Scheme for Diffusion Equation in Spherical Coordinate
 Internatonal Journal of Mathematcs and Systems Scence (018) Volume 1 do:10.494/jmss.v1.815 (Onlne Frst)A Lattce Boltzmann Scheme for Dffuson Equaton n Sphercal Coordnate Debabrata Datta 1 *, T K Pal 1
Internatonal Journal of Mathematcs and Systems Scence (018) Volume 1 do:10.494/jmss.v1.815 (Onlne Frst)A Lattce Boltzmann Scheme for Dffuson Equaton n Sphercal Coordnate Debabrata Datta 1 *, T K Pal 1
Physics 5153 Classical Mechanics. Principle of Virtual Work-1
 P. Guterrez 1 Introducton Physcs 5153 Classcal Mechancs Prncple of Vrtual Work The frst varatonal prncple we encounter n mechancs s the prncple of vrtual work. It establshes the equlbrum condton of a mechancal
P. Guterrez 1 Introducton Physcs 5153 Classcal Mechancs Prncple of Vrtual Work The frst varatonal prncple we encounter n mechancs s the prncple of vrtual work. It establshes the equlbrum condton of a mechancal
DETERMINATION OF TEMPERATURE DISTRIBUTION FOR ANNULAR FINS WITH TEMPERATURE DEPENDENT THERMAL CONDUCTIVITY BY HPM
 Ganj, Z. Z., et al.: Determnaton of Temperature Dstrbuton for S111 DETERMINATION OF TEMPERATURE DISTRIBUTION FOR ANNULAR FINS WITH TEMPERATURE DEPENDENT THERMAL CONDUCTIVITY BY HPM by Davood Domr GANJI
Ganj, Z. Z., et al.: Determnaton of Temperature Dstrbuton for S111 DETERMINATION OF TEMPERATURE DISTRIBUTION FOR ANNULAR FINS WITH TEMPERATURE DEPENDENT THERMAL CONDUCTIVITY BY HPM by Davood Domr GANJI
GeoSteamNet: 2. STEAM FLOW SIMULATION IN A PIPELINE
 PROCEEDINGS, Thrty-Ffth Workshop on Geothermal Reservor Engneerng Stanford Unversty, Stanford, Calforna, February 1-3, 010 SGP-TR-188 GeoSteamNet:. STEAM FLOW SIMULATION IN A PIPELINE Mahendra P. Verma
PROCEEDINGS, Thrty-Ffth Workshop on Geothermal Reservor Engneerng Stanford Unversty, Stanford, Calforna, February 1-3, 010 SGP-TR-188 GeoSteamNet:. STEAM FLOW SIMULATION IN A PIPELINE Mahendra P. Verma
Gasometric Determination of NaHCO 3 in a Mixture
 60 50 40 0 0 5 15 25 35 40 Temperature ( o C) 9/28/16 Gasometrc Determnaton of NaHCO 3 n a Mxture apor Pressure (mm Hg) apor Pressure of Water 1 NaHCO 3 (s) + H + (aq) Na + (aq) + H 2 O (l) + CO 2 (g)
60 50 40 0 0 5 15 25 35 40 Temperature ( o C) 9/28/16 Gasometrc Determnaton of NaHCO 3 n a Mxture apor Pressure (mm Hg) apor Pressure of Water 1 NaHCO 3 (s) + H + (aq) Na + (aq) + H 2 O (l) + CO 2 (g)
Solution Thermodynamics
 CH2351 Chemcal Engneerng Thermodynamcs II Unt I, II www.msubbu.n Soluton Thermodynamcs www.msubbu.n Dr. M. Subramanan Assocate Professor Department of Chemcal Engneerng Sr Svasubramanya Nadar College of
CH2351 Chemcal Engneerng Thermodynamcs II Unt I, II www.msubbu.n Soluton Thermodynamcs www.msubbu.n Dr. M. Subramanan Assocate Professor Department of Chemcal Engneerng Sr Svasubramanya Nadar College of
Thermodynamics II. Department of Chemical Engineering. Prof. Kim, Jong Hak
 Thermodynamcs II Department of Chemcal Engneerng Prof. Km, Jong Hak Soluton Thermodynamcs : theory Obectve : lay the theoretcal foundaton for applcatons of thermodynamcs to gas mxture and lqud soluton
Thermodynamcs II Department of Chemcal Engneerng Prof. Km, Jong Hak Soluton Thermodynamcs : theory Obectve : lay the theoretcal foundaton for applcatons of thermodynamcs to gas mxture and lqud soluton
Publication 2006/01. Transport Equations in Incompressible. Lars Davidson
 Publcaton 2006/01 Transport Equatons n Incompressble URANS and LES Lars Davdson Dvson of Flud Dynamcs Department of Appled Mechancs Chalmers Unversty of Technology Göteborg, Sweden, May 2006 Transport
Publcaton 2006/01 Transport Equatons n Incompressble URANS and LES Lars Davdson Dvson of Flud Dynamcs Department of Appled Mechancs Chalmers Unversty of Technology Göteborg, Sweden, May 2006 Transport
Flow equations To simulate the flow, the Navier-Stokes system that includes continuity and momentum equations is solved
 Smulaton of nose generaton and propagaton caused by the turbulent flow around bluff bodes Zamotn Krll e-mal: krart@gmal.com, cq: 958886 Summary Accurate predctons of nose generaton and spread n turbulent
Smulaton of nose generaton and propagaton caused by the turbulent flow around bluff bodes Zamotn Krll e-mal: krart@gmal.com, cq: 958886 Summary Accurate predctons of nose generaton and spread n turbulent
Estimation of the composition of the liquid and vapor streams exiting a flash unit with a supercritical component
 Department of Energ oltecnco d Mlano Va Lambruschn - 05 MILANO Eercses of Fundamentals of Chemcal rocesses rof. Ganpero Gropp Eercse 8 Estmaton of the composton of the lqud and vapor streams etng a unt
Department of Energ oltecnco d Mlano Va Lambruschn - 05 MILANO Eercses of Fundamentals of Chemcal rocesses rof. Ganpero Gropp Eercse 8 Estmaton of the composton of the lqud and vapor streams etng a unt
Modeling of Dynamic Systems
 Modelng of Dynamc Systems Ref: Control System Engneerng Norman Nse : Chapters & 3 Chapter objectves : Revew the Laplace transform Learn how to fnd a mathematcal model, called a transfer functon Learn how
Modelng of Dynamc Systems Ref: Control System Engneerng Norman Nse : Chapters & 3 Chapter objectves : Revew the Laplace transform Learn how to fnd a mathematcal model, called a transfer functon Learn how
Diffusion Mass Transfer
 Dffuson Mass Transfer General onsderatons Mass transfer refers to mass n transt due to a speces concentraton gradent n a mture. Must have a mture of two or more speces for mass transfer to occur. The speces
Dffuson Mass Transfer General onsderatons Mass transfer refers to mass n transt due to a speces concentraton gradent n a mture. Must have a mture of two or more speces for mass transfer to occur. The speces
The Two-scale Finite Element Errors Analysis for One Class of Thermoelastic Problem in Periodic Composites
 7 Asa-Pacfc Engneerng Technology Conference (APETC 7) ISBN: 978--6595-443- The Two-scale Fnte Element Errors Analyss for One Class of Thermoelastc Problem n Perodc Compostes Xaoun Deng Mngxang Deng ABSTRACT
7 Asa-Pacfc Engneerng Technology Conference (APETC 7) ISBN: 978--6595-443- The Two-scale Fnte Element Errors Analyss for One Class of Thermoelastc Problem n Perodc Compostes Xaoun Deng Mngxang Deng ABSTRACT
Turbulence classification of load data by the frequency and severity of wind gusts. Oscar Moñux, DEWI GmbH Kevin Bleibler, DEWI GmbH
 Turbulence classfcaton of load data by the frequency and severty of wnd gusts Introducton Oscar Moñux, DEWI GmbH Kevn Blebler, DEWI GmbH Durng the wnd turbne developng process, one of the most mportant
Turbulence classfcaton of load data by the frequency and severty of wnd gusts Introducton Oscar Moñux, DEWI GmbH Kevn Blebler, DEWI GmbH Durng the wnd turbne developng process, one of the most mportant
Name ID # For relatively dilute aqueous solutions the molality and molarity are approximately equal.
 Name ID # 1 CHEMISTRY 212, Lect. Sect. 002 Dr. G. L. Roberts Exam #1/Sprng 2000 Thursday, February 24, 2000 CLOSED BOOK EXM No notes or books allowed. Calculators may be used. tomc masses of nterest are
Name ID # 1 CHEMISTRY 212, Lect. Sect. 002 Dr. G. L. Roberts Exam #1/Sprng 2000 Thursday, February 24, 2000 CLOSED BOOK EXM No notes or books allowed. Calculators may be used. tomc masses of nterest are
STUDY ON TWO PHASE FLOW IN MICRO CHANNEL BASED ON EXPERI- MENTS AND NUMERICAL EXAMINATIONS
 Blucher Mechancal Engneerng Proceedngs May 0, vol., num. www.proceedngs.blucher.com.br/evento/0wccm STUDY ON TWO PHASE FLOW IN MICRO CHANNEL BASED ON EXPERI- MENTS AND NUMERICAL EXAMINATIONS Takahko Kurahash,
Blucher Mechancal Engneerng Proceedngs May 0, vol., num. www.proceedngs.blucher.com.br/evento/0wccm STUDY ON TWO PHASE FLOW IN MICRO CHANNEL BASED ON EXPERI- MENTS AND NUMERICAL EXAMINATIONS Takahko Kurahash,
Chapter 13: Multiple Regression
 Chapter 13: Multple Regresson 13.1 Developng the multple-regresson Model The general model can be descrbed as: It smplfes for two ndependent varables: The sample ft parameter b 0, b 1, and b are used to
Chapter 13: Multple Regresson 13.1 Developng the multple-regresson Model The general model can be descrbed as: It smplfes for two ndependent varables: The sample ft parameter b 0, b 1, and b are used to
Brownian-Dynamics Simulation of Colloidal Suspensions with Kob-Andersen Type Lennard-Jones Potentials 1
 Brownan-Dynamcs Smulaton of Collodal Suspensons wth Kob-Andersen Type Lennard-Jones Potentals 1 Yuto KIMURA 2 and Mcho TOKUYAMA 3 Summary Extensve Brownan-dynamcs smulatons of bnary collodal suspenton
Brownan-Dynamcs Smulaton of Collodal Suspensons wth Kob-Andersen Type Lennard-Jones Potentals 1 Yuto KIMURA 2 and Mcho TOKUYAMA 3 Summary Extensve Brownan-dynamcs smulatons of bnary collodal suspenton
