March CUME Organic Chemistry
|
|
|
- Gloria Chase
- 5 years ago
- Views:
Transcription
1 March CUME Organic Chemistry Department of Chemistry, University of Missouri Columbia Saturday, March 21, O Clock, 221 Chemistry Dr. Rainer Glaser Announced Reading Circular Dichroism and Exciton-Coupled Circular Dichroism bi.re.frin.gence n [ISV] (1898): the refraction of light in an anisotropic material (as calcite) in two slightly different directions to form two rays -- bi.re.frin.gent adj di.chro.ism n (1819): the property of some crystals and solutions of absorbing one of two planepolarized components of transmitted light more strongly than the other; also: the property of exhibiting different colors by reflected or transmitted light--compare circular dichroism. circular dichroism n (ca. 1961) 1: the property (as of an optically active medium) of unequal absorption of right and left plane-polarized light so that the emergent light is elliptically polarized 2: a spectroscopic technique that makes use of circular dichroism. Definitions from Britannica Online. Search "circular dichroism" Britannica Online. < [Accessed 07 March 1998]. 1
2 Question 1. Polarized Light, Circular Birefringence and Circular Dichroism. (60 points) (a) In area 1, draw a schematic diagram of unpolarized light. In area 2, draw a schematic diagram of plane or linearly polarized light. In both of these cases, draw the E-field vector direction(s) and assume that you are looking along the direction the light is traveling (z-direction). In area 3, draw a linearly polarized light wave as a function of time assuming that the light travels in the z-direction (left to right) and is linearly polarized in the xz-plane. In area 4, draw a linearly polarized light wave as a function of time assuming that the light travels in the z-direction (left to right on paper) and is linearly polarized in the yz-plane. (8 points) Area 1 (2 points) Area 2 (2 points) Area 3 (2 points) Area 4 (2 points) 2
3 (b) Now let s turn to circularly polarized light. Some text follows from the text Organic Structural Spectroscopy by Lambert et al. (Prentice Hall, 1997). Fill in the blanks. Then read the legend to the Figure (adapted from Figure of the text by Lambert et al.) and create the four sketches that the figure legend describes. (12 points) (Lambert et al., p. 260): (linearly polarized, unpolarized) light might be decomposed into a pair of orthogonal polarization states that are important in understanding chiroptical phenomena: left and right circularly polarized light. In the case of circularly polarized light, the transverse vibrations [of the E-field vector] trace out a helix as a function of time. The helix may be either left-handed (top in Figure) or right-handed (bottom in Figure). Viewed in cross-section, that is, if an observer were situated on the z-axis looking toward the light source, the transverse vibrations would trace out a. Light whose electric field vector traces out a right-handed helical pattern is termed light. The cross-sectional appearance of (clockwise, counter-clockwise) rotation of the electric field is obtained by pushing the helix forward through a perpendicular plane without rotating it. In other words, the helix is moved forward, but it is not turned like a mechanical screw. (1 point for each blank, 4 total) (2 points) (2 points) (2 points) (2 points) Figure. Circularly polarized radiation. Left: The light wave as a function of time. Right: Cross section of the light wave. Light polarized (top) left circularly and (bottom) right circularly. 3
4 (c) The components E L and E R of left and right circularly polarized light are shown in area 1 for some linearly polarized light. First, draw the E-field resulting from vector addition of the components. Pay attention to direction and magnitude. In areas 2-6, draw the situations after E L and E R have rotated an additional thirty degrees each, that is, both components move at the same speed. (12 points) area 1 area 2 area 3 E L E R area 4 area 5 area 6 (d) Do as in (c) but assume that E L and E R move at different speeds. Assume that E L moves 45 degrees in the time E R requires to move 30 degrees. Again, draw the resulting E-field obtained by vector addition of the components. Pay attention to direction and magnitude. In areas 2 and 3, draw the situations after the components E L and E R have rotated two and three times longer. (6 points) area 1 area 2 area 3 State the overall effect resulting from this phenomenon of circular birefringence (2 points): 4
5 (e) Consider situation (c) and assume that E L and E R are absorbed equally as they travel though a material. State what happens to the direction of the plane of the polarized light and state what happens to the vector sum of the E L and E R as time progresses. State what type of polarized light comes out of the material. (7 points) (f) Consider situation (c) and assume that E L and E R are absorbed to a different degree (circular dichroism) as they travel though a material. State what happens to the direction of the plane of the polarized light and state what happens to the vector sum of the E L and E R as time progresses. State what type of polarized light comes out of the material. (7 points) (g) Consider situation (d) (circular birefringence) and assume that E L and E R are absorbed to a different degree (circular dichroism) as they travel though a material. State what happens to the direction of the plane of the polarized light and state what happens to the vector sum of the E L and E R as time progresses. State what type of polarized light comes out of the material. (6 points) 5
6 Question 2. Circular Birefringence and Circular Dichroism Quantities. (20 points) (a) Circular birefringence measurements aim at quantifying the difference in the speeds of left and right polarized light. Circular birefringence measures optical rotation. Write down the equation that defines the specific rotation and give its units. Write down the equation that defines molar rotation and gives its units. Specific rotation: (5 points) Molar rotation: (5 points) (b) Circular dichroism measurements aim at quantifying the difference in the absorbances of left and right polarized light. Most CD instruments measure angle of ellipticity θ vs. λ, which can be adjusted to read the differential absorbance, A = A L - A R. This quantity is related to the difference in molar absorptivity, ε = ε L - ε R. Write down the equation that links A and ε. Give units for every quantity that occurs in this equation. Then explain what the angle of ellipticity is (an illustration would be better than a long epic description). A = f( ε): (5 points) The angle of ellipticity, θ, mmmh, let s see... (5 points) 6
7 Question 3. Absorbance Spectra and CD Cotton Effects. (20 points) (a) Suppose you have a material that has an allowed electronic transition centered at a wavelength of λ = 500 nm. Draw schematic spectra as requested. In each case, the horizontal axis will be the wavelength axis and λ increases to the right (the usual convention). Assume that λ = 500 nm is just in the middle. For the vertical axis, provide the quantity that is being measured and indicate the sign (no need to give numbers and magnitudes, no need to give units). (3 points each box) The UV/Vis spectrum of the pure R-enantiomer. The CD spectrum of the pure R-enantiomer with positive Cotton effect (assumed in our case). The UV/Vis spectrum of the pure S enantiomer. The CD spectrum of the pure S-enantiomer. The UV/Vis spectrum of the racemate. The CD spectrum of the racemate. Why is the Cotton effect called the Cotton effect? (2 points) Named after F. Albert Cotton (the Cotton from the text Cotton & Wilkinson) The effect was first discovered in studies of cotton, the plant. Named after Aime Cotton, a French female physicist. Named after Aime Cotton, a French male physicist. 7
8 Question 4. Exciton Coupling and Bisignate Cotton Effects. (20 points) (a) If two chromophores X and Y have very similar absorption bands (X = Y or X Y) and if these two chromophores are in (relatively) close proximity (even 1000 pm can be close in this case), then the bichromophoric composite system will exhibit an exciton splitting (Davydov spitting). As the consequence of this interaction between the excited states, the one absorption (or the two almost identical absorptions) of the individual chromophores X and Y now will split into one transition at higher and one transition at lower energy. Draw a diagram that illustrates this situation. (5 points) (b) What are the effects on the UV/Vis and the CD spectra? The UV/Vis band will (narrow, broaden) compared to that of the band of X or Y alone. More change occurs in the CD spectrum since the signs of the Cotton effects of the two transitions will be opposite and a bisignate shape of the CD curve occurs. One speaks of positive exciton chirality if the Cotton effects at (longer, shorter) wavelength is positive. (The exciton chirality can be related to absolute chirality via the so-called exciton chirality rules.) As with Question 3 (a), draw schematic spectra as requested. (15 points) UV/Vis spectrum of one chromophore X: UV/Vis spectrum of two excitoncoupled chromophores X: CD spectrum of two excitoncoupled chromophores X with positive exciton chirality. 8
9 Question 5. Exciton Coupled CD Spectra of Retinal and Derivatives. (50 points) Absolute Sense of Twist of the C12-C13 Bond of the Retinal Chromophore in Bovine Rhodopsin on Exciton-Coupled CD Spectra of 11,12-Dihydroretinal Analogues. Qiang Tan, Jihong Lou, Babak Borhan, Elena Karnaukhova, Nina Berova, and Koji Nakanishi Angew. Chem. Int. Ed. Engl. 1997, 36, Figure 1 shows the structure of the 11-cisretinal chromophore in native rhodoposin. The UV/Vis and CD spectra are shown of bovin rhodopsin in octylglucoside. (a) Looking at the CD spectrum, one realizes that there are two bands that show Cotton effects: One band at nm shows a (positive, negative) Cotton effect and the other band at nm shows a (positive, negative) Cotton effect. The simple fact that bovin rhodopsin shows a CD spectrum indicates that the chromophore is (chiral, achiral). Realize that there are (give number) chirality centers in A, (give number) chirality centers in B, and (give number) chirality centers in C. (Ignore the attachment to the imine-n. Whatever chirality centers might be in there, they all are far away as far as the present discussion is concerned.) (8 points) (b) Experiments show that the α-band vanishes when planes B and C are forced to co-planarity. Experiments also show that the β-band vanishes when planes A and B are forced to co-planarity. (6 pts) Explain. 9
10 (c) Consider butadiene. Butadiene forms two stable conformers. The most stable conformation is (cis, trans, gauche) and the other conformation is (cis, trans, gauche). Considering this information, one might have expected that planes A and B (are, are not) coplanar and that planes B and C (are, are not) co-planar. (6 points) (d) To determine the absolute direction of the twist around the C12-C13 bond, Nakanishi et al. wanted to disrupt the conjugates polyene by a single bond so that the two polyene moieties flanking the single bond would couple. The sign of the resulting bisignate couplet would reflect the twist in a nonempirical manner. This expectation was illustrated by the figure which is reproduced below without its caption. Explain this reasoning. State what is expected to couple. State why it is reasonable to expect such coupling. State why the drawn configurations are expected to give the bisignate spectra shown. For each of the spectra state whether they show positive or negative exciton chirality. Use precise language as much as possible. (20 points) 10
11 11
12 (e) Nakanishi et al. incorporated the (protonated Schiff base of the) 11,12-dihydroretinal into opsin and measured the CD spectrum. The triene absorbs at 255 nm. The protonated Schiff base absorbs at 270. The CD spectrum shows negative exciton coupling with a maximum at 275 nm and a minimum at 295 nm. Provided that 11,12-dihydroretinal adopts essentially the same conformation as that of 11-cisretinal in rhodopsin, the exciton coupling allows one to deduce the absolute stereochemistry. Show the structure with the correct conformation and argue briefly for it. (10 points) 12
13 13
4. Circular Dichroism - Spectroscopy
 4. Circular Dichroism - Spectroscopy The optical rotatory dispersion (ORD) and the circular dichroism (CD) are special variations of absorption spectroscopy in the UV and VIS region of the spectrum. The
4. Circular Dichroism - Spectroscopy The optical rotatory dispersion (ORD) and the circular dichroism (CD) are special variations of absorption spectroscopy in the UV and VIS region of the spectrum. The
Chemistry 416 Spectroscopy Winter Semester 1996 Dr. Rainer Glaser
 Chemistry 416 Spectroscopy Winter Semester 1996 Dr. Rainer Glaser First 1-our Examination UV/Vis Spectroscopy Friday, February 9, 1996, 8:40-9:30 Name: Max Yours Question 1 10 Question 2 10 Question 3
Chemistry 416 Spectroscopy Winter Semester 1996 Dr. Rainer Glaser First 1-our Examination UV/Vis Spectroscopy Friday, February 9, 1996, 8:40-9:30 Name: Max Yours Question 1 10 Question 2 10 Question 3
Circular Dichroism & Optical Rotatory Dispersion. Proteins (KCsa) Polysaccharides (agarose) DNA CHEM 305. Many biomolecules are α-helical!
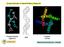 Circular Dichroism & Optical Rotatory Dispersion Polysaccharides (agarose) DNA Proteins (KCsa) Many biomolecules are α-helical! How can we measure the amount and changes in amount of helical structure
Circular Dichroism & Optical Rotatory Dispersion Polysaccharides (agarose) DNA Proteins (KCsa) Many biomolecules are α-helical! How can we measure the amount and changes in amount of helical structure
Lab #13: Polarization
 Lab #13: Polarization Introduction In this experiment we will investigate various properties associated with polarized light. We will study both its generation and application. Real world applications
Lab #13: Polarization Introduction In this experiment we will investigate various properties associated with polarized light. We will study both its generation and application. Real world applications
Chiroptical Spectroscopy
 Chiroptical Spectroscopy Theory and Applications in Organic Chemistry Lecture 2: Polarized light Masters Level Class (181 041) Mondays, 8.15-9.45 am, NC 02/99 Wednesdays, 10.15-11.45 am, NC 02/99 28 Electromagnetic
Chiroptical Spectroscopy Theory and Applications in Organic Chemistry Lecture 2: Polarized light Masters Level Class (181 041) Mondays, 8.15-9.45 am, NC 02/99 Wednesdays, 10.15-11.45 am, NC 02/99 28 Electromagnetic
Polarization Mode Dispersion
 Unit-7: Polarization Mode Dispersion https://sites.google.com/a/faculty.muet.edu.pk/abdullatif Department of Telecommunication, MUET UET Jamshoro 1 Goos Hänchen Shift The Goos-Hänchen effect is a phenomenon
Unit-7: Polarization Mode Dispersion https://sites.google.com/a/faculty.muet.edu.pk/abdullatif Department of Telecommunication, MUET UET Jamshoro 1 Goos Hänchen Shift The Goos-Hänchen effect is a phenomenon
Circular Dichroism. Principles and Applications VCH. Koji Nakanishi, Nina Berova, and Robert W. Woody EDITED BY
 Circular Dichroism Principles and Applications EDITED BY Koji Nakanishi, Nina Berova, and Robert W. Woody VCH Contents Contributors xv Chapter 1 Circular Dichroism: An Introduction 1 Günther Snatzke 1.1
Circular Dichroism Principles and Applications EDITED BY Koji Nakanishi, Nina Berova, and Robert W. Woody VCH Contents Contributors xv Chapter 1 Circular Dichroism: An Introduction 1 Günther Snatzke 1.1
Light for which the orientation of the electric field is constant although its magnitude and sign vary in time.
 L e c t u r e 8 1 Polarization Polarized light Light for which the orientation of the electric field is constant although its magnitude and sign vary in time. Imagine two harmonic, linearly polarized light
L e c t u r e 8 1 Polarization Polarized light Light for which the orientation of the electric field is constant although its magnitude and sign vary in time. Imagine two harmonic, linearly polarized light
Reflection = EM strikes a boundary between two media differing in η and bounces back
 Reflection = EM strikes a boundary between two media differing in η and bounces back Incident ray θ 1 θ 2 Reflected ray Medium 1 (air) η = 1.00 Medium 2 (glass) η = 1.50 Specular reflection = situation
Reflection = EM strikes a boundary between two media differing in η and bounces back Incident ray θ 1 θ 2 Reflected ray Medium 1 (air) η = 1.00 Medium 2 (glass) η = 1.50 Specular reflection = situation
Chiroptical Spectroscopy
 Chiroptical Spectroscopy Theory and Applications in Organic Chemistry Lecture 1: It all started with a piece of quartz. Masters Level Class (181 041) Mondays, 8.15-9.45 am, C 02/99 Wednesdays, 10.15-11.45
Chiroptical Spectroscopy Theory and Applications in Organic Chemistry Lecture 1: It all started with a piece of quartz. Masters Level Class (181 041) Mondays, 8.15-9.45 am, C 02/99 Wednesdays, 10.15-11.45
POLARISATION. We have not really discussed the direction of the Electric field other that that it is perpendicular to the direction of motion.
 POLARISATION Light is a transverse electromagnetic wave. We have not really discussed the direction of the Electric field other that that it is perpendicular to the direction of motion. If the E field
POLARISATION Light is a transverse electromagnetic wave. We have not really discussed the direction of the Electric field other that that it is perpendicular to the direction of motion. If the E field
Electromagnetic wave energy & polarization
 Phys 0 Lecture 6 Electromagnetic wave energy & polarization Today we will... Learn about properties p of electromagnetic waves Energy density & intensity Polarization linear, circular, unpolarized Apply
Phys 0 Lecture 6 Electromagnetic wave energy & polarization Today we will... Learn about properties p of electromagnetic waves Energy density & intensity Polarization linear, circular, unpolarized Apply
Polarization of Light and Birefringence of Materials
 Polarization of Light and Birefringence of Materials Ajit Balagopal (Team Members Karunanand Ogirala, Hui Shen) ECE 614- PHOTONIC INFORMATION PROCESSING LABORATORY Abstract-- In this project, we study
Polarization of Light and Birefringence of Materials Ajit Balagopal (Team Members Karunanand Ogirala, Hui Shen) ECE 614- PHOTONIC INFORMATION PROCESSING LABORATORY Abstract-- In this project, we study
Structure of Coordination Compounds
 Chapter 22 COORDINATION CHEMISTRY (Part II) Dr. Al Saadi 1 Structure of Coordination Compounds The geometry of coordination compounds plays a significant role in determining their properties. The structure
Chapter 22 COORDINATION CHEMISTRY (Part II) Dr. Al Saadi 1 Structure of Coordination Compounds The geometry of coordination compounds plays a significant role in determining their properties. The structure
POLARIZATION FUNDAMENTAL OPTICS POLARIZATION STATES 1. CARTESIAN REPRESENTATION 2. CIRCULAR REPRESENTATION. Polarization. marketplace.idexop.
 POLARIZATION POLARIZATION STATS Four numbers are required to describe a single plane wave Fourier component traveling in the + z direction. These can be thought of as the amplitude and phase shift of the
POLARIZATION POLARIZATION STATS Four numbers are required to describe a single plane wave Fourier component traveling in the + z direction. These can be thought of as the amplitude and phase shift of the
Review of Circular Dichroism
 Review of Circular Dichroism Absorption of photons by molecules requires that electrons in the molecule be able to absorb the energy of the photon and also its momentum (energy is conserved and momentum
Review of Circular Dichroism Absorption of photons by molecules requires that electrons in the molecule be able to absorb the energy of the photon and also its momentum (energy is conserved and momentum
Chemistry 304B, Spring 1999 Lecture 5 1. UV Spectroscopy:
 Chemistry 304B, Spring 1999 Lecture 5 1 Ultraviolet spectroscopy; UV Spectroscopy: Infrared spectroscopy; Nuclear magnetic resonance spectroscopy General basis of spectroscopy: Shine light at a collection
Chemistry 304B, Spring 1999 Lecture 5 1 Ultraviolet spectroscopy; UV Spectroscopy: Infrared spectroscopy; Nuclear magnetic resonance spectroscopy General basis of spectroscopy: Shine light at a collection
Waves & Oscillations
 Physics 42200 Waves & Oscillations Lecture 32 Polarization of Light Spring 2015 Semester Matthew Jones Types of Polarization Light propagating through different materials: One polarization component can
Physics 42200 Waves & Oscillations Lecture 32 Polarization of Light Spring 2015 Semester Matthew Jones Types of Polarization Light propagating through different materials: One polarization component can
A very brief history of the study of light
 1. Sir Isaac Newton 1672: A very brief history of the study of light Showed that the component colors of the visible portion of white light can be separated through a prism, which acts to bend the light
1. Sir Isaac Newton 1672: A very brief history of the study of light Showed that the component colors of the visible portion of white light can be separated through a prism, which acts to bend the light
Chiroptical Spectroscopy
 Chiroptical Spectroscopy Theory and Applications in Organic Chemistry Lecture 3: (Crash course in) Theory of optical activity Masters Level Class (181 041) Mondays, 8.15-9.45 am, NC 02/99 Wednesdays, 10.15-11.45
Chiroptical Spectroscopy Theory and Applications in Organic Chemistry Lecture 3: (Crash course in) Theory of optical activity Masters Level Class (181 041) Mondays, 8.15-9.45 am, NC 02/99 Wednesdays, 10.15-11.45
Topic 4: Waves 4.3 Wave characteristics
 Guidance: Students will be expected to calculate the resultant of two waves or pulses both graphically and algebraically Methods of polarization will be restricted to the use of polarizing filters and
Guidance: Students will be expected to calculate the resultant of two waves or pulses both graphically and algebraically Methods of polarization will be restricted to the use of polarizing filters and
Skoog Chapter 6 Introduction to Spectrometric Methods
 Skoog Chapter 6 Introduction to Spectrometric Methods General Properties of Electromagnetic Radiation (EM) Wave Properties of EM Quantum Mechanical Properties of EM Quantitative Aspects of Spectrochemical
Skoog Chapter 6 Introduction to Spectrometric Methods General Properties of Electromagnetic Radiation (EM) Wave Properties of EM Quantum Mechanical Properties of EM Quantitative Aspects of Spectrochemical
Optics and Optical Design. Chapter 6: Polarization Optics. Lectures 11 13
 Optics and Optical Design Chapter 6: Polarization Optics Lectures 11 13 Cord Arnold / Anne L Huillier Polarization of Light Arbitrary wave vs. paraxial wave One component in x direction y x z Components
Optics and Optical Design Chapter 6: Polarization Optics Lectures 11 13 Cord Arnold / Anne L Huillier Polarization of Light Arbitrary wave vs. paraxial wave One component in x direction y x z Components
polarisation of Light
 Basic concepts to understand polarisation of Light Polarization of Light Nature of light: light waves are transverse in nature i. e. the waves propagates in a direction perpendicular to the direction of
Basic concepts to understand polarisation of Light Polarization of Light Nature of light: light waves are transverse in nature i. e. the waves propagates in a direction perpendicular to the direction of
Lecture 4: Polarisation of light, introduction
 Lecture 4: Polarisation of light, introduction Lecture aims to explain: 1. Light as a transverse electro-magnetic wave 2. Importance of polarisation of light 3. Linearly polarised light 4. Natural light
Lecture 4: Polarisation of light, introduction Lecture aims to explain: 1. Light as a transverse electro-magnetic wave 2. Importance of polarisation of light 3. Linearly polarised light 4. Natural light
240 Chem. Stereochemistry. Chapter 5
 240 Chem Stereochemistry Chapter 5 1 Isomerism Isomers are different compounds that have the same molecular formula. Constitutional isomers are isomers that differ because their atoms are connected in
240 Chem Stereochemistry Chapter 5 1 Isomerism Isomers are different compounds that have the same molecular formula. Constitutional isomers are isomers that differ because their atoms are connected in
NAWAB SHAH ALAM KHAN COLLEGE OF ENGINEERING & TECHNOLOGY UNIT II-a POLARISATION
 NAWAB SHAH ALAM KHAN COLLEGE OF ENGINEERING & TECHNOLOGY UNIT II-a 1 POLARISATION SYLLABUS :Polarization: Introduction, Malus s law, double refraction, Nicol prism, Quarter wave and half wave plates. 1.
NAWAB SHAH ALAM KHAN COLLEGE OF ENGINEERING & TECHNOLOGY UNIT II-a 1 POLARISATION SYLLABUS :Polarization: Introduction, Malus s law, double refraction, Nicol prism, Quarter wave and half wave plates. 1.
Physics I Keystone Institute Technology & Management Unit-II
 Un-polarized light Ordinary light is a collection of wave trains emitted by atoms or group of atoms with coherent time no longer than 10-8 second. Each wave train has different orientation and phase of
Un-polarized light Ordinary light is a collection of wave trains emitted by atoms or group of atoms with coherent time no longer than 10-8 second. Each wave train has different orientation and phase of
Chapter 5 Stereochemistry. Stereoisomers
 Chapter 5 Stereochemistry Stereoisomers Same bonding sequence Different arrangement in space Example: OOC-C=C-COO has two geometric (cis-trans) isomers: COO COO COO COO Stereochemistry Slide 5-2 1 Chirality
Chapter 5 Stereochemistry Stereoisomers Same bonding sequence Different arrangement in space Example: OOC-C=C-COO has two geometric (cis-trans) isomers: COO COO COO COO Stereochemistry Slide 5-2 1 Chirality
Quarter wave plates and Jones calculus for optical system
 2/11/16 Electromagnetic Processes In Dispersive Media, Lecture 6 1 Quarter wave plates and Jones calculus for optical system T. Johnson 2/11/16 Electromagnetic Processes In Dispersive Media, Lecture 6
2/11/16 Electromagnetic Processes In Dispersive Media, Lecture 6 1 Quarter wave plates and Jones calculus for optical system T. Johnson 2/11/16 Electromagnetic Processes In Dispersive Media, Lecture 6
( ) x10 8 m. The energy in a mole of 400 nm photons is calculated by: ' & sec( ) ( & % ) 6.022x10 23 photons' E = h! = hc & 6.
 Introduction to Spectroscopy Spectroscopic techniques are widely used to detect molecules, to measure the concentration of a species in solution, and to determine molecular structure. For proteins, most
Introduction to Spectroscopy Spectroscopic techniques are widely used to detect molecules, to measure the concentration of a species in solution, and to determine molecular structure. For proteins, most
118/218 FINAL TAKE HOME due Monday, March 19, Chemistry room 4129
 118/218 FINAL TAKE HOME due Monday, March 19, Chemistry room 4129 [1] In the stilbene photoisomerization paper on the website handouts page refer to Figure 3. (a) How does the wavelength and timing of
118/218 FINAL TAKE HOME due Monday, March 19, Chemistry room 4129 [1] In the stilbene photoisomerization paper on the website handouts page refer to Figure 3. (a) How does the wavelength and timing of
E The oscillating E-field defines the polarization of the wave. B
 This sheet is the lab document your TA will use to score your lab. It is to be turned in at the end of lab. To receive full credit you must use complete sentences and explain your reasoning. A. Describing
This sheet is the lab document your TA will use to score your lab. It is to be turned in at the end of lab. To receive full credit you must use complete sentences and explain your reasoning. A. Describing
MOLECULAR LIGHT SCATTERING AND OPTICAL ACTIVITY
 MOLECULAR LIGHT SCATTERING AND OPTICAL ACTIVITY Second edition, revised and enlarged LAURENCE D. BARRON, F.R.S.E. Gardiner Professor of Chemistry, University of Glasgow 122. CAMBRIDGE UNIVERSITY PRESS
MOLECULAR LIGHT SCATTERING AND OPTICAL ACTIVITY Second edition, revised and enlarged LAURENCE D. BARRON, F.R.S.E. Gardiner Professor of Chemistry, University of Glasgow 122. CAMBRIDGE UNIVERSITY PRESS
Preparation of Polythiophene Films Showing Optical Activity by Electrochemical Polymerisation in Cholesteric Liquid Crystal Containing Coumarine
 International Letters of Chemistry, Physics and Astronomy Online: 2015-01-26 ISSN: 2299-3843, Vol. 46, pp 42-47 doi:10.18052/www.scipress.com/ilcpa.46.42 2015 SciPress Ltd., Switzerland Preparation of
International Letters of Chemistry, Physics and Astronomy Online: 2015-01-26 ISSN: 2299-3843, Vol. 46, pp 42-47 doi:10.18052/www.scipress.com/ilcpa.46.42 2015 SciPress Ltd., Switzerland Preparation of
Group Theory: Matrix Representation & Consequences of Symmetry
 Group Theory: Matrix Representation & Consequences of Symmetry Matrix Representation of Group Theory Reducible and Irreducible Representations The Great Orthogonality Theorem The ive Rules The Standard
Group Theory: Matrix Representation & Consequences of Symmetry Matrix Representation of Group Theory Reducible and Irreducible Representations The Great Orthogonality Theorem The ive Rules The Standard
Absorption Spectra. ! Ti(H 2 O) 6 3+ appears purple (red + blue) because it absorbs green light at ~500 nm = ~20,000 cm 1.
 Absorption Spectra! Colors of transition metal complexes result from absorption of a small portion of the visible spectrum with transmission of the unabsorbed frequencies. Visible Spectra of [M(H 2 O)
Absorption Spectra! Colors of transition metal complexes result from absorption of a small portion of the visible spectrum with transmission of the unabsorbed frequencies. Visible Spectra of [M(H 2 O)
16. More About Polarization
 16. More About Polarization Polarization control Wave plates Circular polarizers Reflection & polarization Scattering & polarization Birefringent materials have more than one refractive index A special
16. More About Polarization Polarization control Wave plates Circular polarizers Reflection & polarization Scattering & polarization Birefringent materials have more than one refractive index A special
Optics and Optical Design. Chapter 6: Polarization Optics. Lectures 11-13
 Optics and Optical Design Chapter 6: Polarization Optics Lectures 11-13 Cord Arnold / Anne L Huillier Polarization of Light Arbitrary wave vs. paraxial wave One component in x-direction y x z Components
Optics and Optical Design Chapter 6: Polarization Optics Lectures 11-13 Cord Arnold / Anne L Huillier Polarization of Light Arbitrary wave vs. paraxial wave One component in x-direction y x z Components
Chemistry 123: Physical and Organic Chemistry Topic 1: Organic Chemistry
 Concept Check: Topic 1: Conformation Winter 2009 Page 112 Concept Check: Topic 1: Conformation Winter 2009 Page 113 1 STEREOCHEMISTRY Winter 2009 Page 114 We have already covered two kinds of isomerism:
Concept Check: Topic 1: Conformation Winter 2009 Page 112 Concept Check: Topic 1: Conformation Winter 2009 Page 113 1 STEREOCHEMISTRY Winter 2009 Page 114 We have already covered two kinds of isomerism:
Terms used in UV / Visible Spectroscopy
 Terms used in UV / Visible Spectroscopy Chromophore The part of a molecule responsible for imparting color, are called as chromospheres. OR The functional groups containing multiple bonds capable of absorbing
Terms used in UV / Visible Spectroscopy Chromophore The part of a molecule responsible for imparting color, are called as chromospheres. OR The functional groups containing multiple bonds capable of absorbing
CH 3 C 2 H 5. Tetrahedral Stereochemistry
 Ch 5 Tetrahedral Stereochemistry Enantiomers - Two non-superimposable mirror image molecules - They are stereoisomers with the same atoms and bonds, but different spatial geometries. - The two molecules
Ch 5 Tetrahedral Stereochemistry Enantiomers - Two non-superimposable mirror image molecules - They are stereoisomers with the same atoms and bonds, but different spatial geometries. - The two molecules
Chap. 5. Jones Calculus and Its Application to Birefringent Optical Systems
 Chap. 5. Jones Calculus and Its Application to Birefringent Optical Systems - The overall optical transmission through many optical components such as polarizers, EO modulators, filters, retardation plates.
Chap. 5. Jones Calculus and Its Application to Birefringent Optical Systems - The overall optical transmission through many optical components such as polarizers, EO modulators, filters, retardation plates.
DEGREE OF POLARIZATION VS. POINCARÉ SPHERE COVERAGE - WHICH IS NECESSARY TO MEASURE PDL ACCURATELY?
 DEGREE OF POLARIZATION VS. POINCARÉ SPHERE COVERAGE - WHICH IS NECESSARY TO MEASURE PDL ACCURATELY? DEGREE OF POLARIZATION VS. POINCARE SPHERE COVERAGE - WHICH IS NECESSARY TO MEASURE PDL ACCURATELY? Introduction
DEGREE OF POLARIZATION VS. POINCARÉ SPHERE COVERAGE - WHICH IS NECESSARY TO MEASURE PDL ACCURATELY? DEGREE OF POLARIZATION VS. POINCARE SPHERE COVERAGE - WHICH IS NECESSARY TO MEASURE PDL ACCURATELY? Introduction
Introduction to Polarization
 Phone: Ext 659, E-mail: hcchui@mail.ncku.edu.tw Fall/007 Introduction to Polarization Text Book: A Yariv and P Yeh, Photonics, Oxford (007) 1.6 Polarization States and Representations (Stokes Parameters
Phone: Ext 659, E-mail: hcchui@mail.ncku.edu.tw Fall/007 Introduction to Polarization Text Book: A Yariv and P Yeh, Photonics, Oxford (007) 1.6 Polarization States and Representations (Stokes Parameters
Waves & Oscillations
 Physics 42200 Waves & Oscillations Lecture 25 Propagation of Light Spring 2013 Semester Matthew Jones Midterm Exam: Date: Wednesday, March 6 th Time: 8:00 10:00 pm Room: PHYS 203 Material: French, chapters
Physics 42200 Waves & Oscillations Lecture 25 Propagation of Light Spring 2013 Semester Matthew Jones Midterm Exam: Date: Wednesday, March 6 th Time: 8:00 10:00 pm Room: PHYS 203 Material: French, chapters
Advanced Inorganic Chemistry
 Advanced Inorganic Chemistry Alireza Gorji Department of Chemistry, Yazd University Isomerism 20_441 Isomers (same formula but different properties) ایزومری Structural isomers (different bonds) Stereoisomers
Advanced Inorganic Chemistry Alireza Gorji Department of Chemistry, Yazd University Isomerism 20_441 Isomers (same formula but different properties) ایزومری Structural isomers (different bonds) Stereoisomers
Chapter 1 - The Nature of Light
 David J. Starling Penn State Hazleton PHYS 214 Electromagnetic radiation comes in many forms, differing only in wavelength, frequency or energy. Electromagnetic radiation comes in many forms, differing
David J. Starling Penn State Hazleton PHYS 214 Electromagnetic radiation comes in many forms, differing only in wavelength, frequency or energy. Electromagnetic radiation comes in many forms, differing
CHAPTER 4 TEST REVIEW
 IB PHYSICS Name: Period: Date: # Marks: 74 Raw Score: IB Curve: DEVIL PHYSICS BADDEST CLASS ON CAMPUS CHAPTER 4 TEST REVIEW 1. In which of the following regions of the electromagnetic spectrum is radiation
IB PHYSICS Name: Period: Date: # Marks: 74 Raw Score: IB Curve: DEVIL PHYSICS BADDEST CLASS ON CAMPUS CHAPTER 4 TEST REVIEW 1. In which of the following regions of the electromagnetic spectrum is radiation
Lecture 20 Optical Characterization 2
 Lecture 20 Optical Characterization 2 Schroder: Chapters 2, 7, 10 1/68 Announcements Homework 5/6: Is online now. Due Wednesday May 30th at 10:00am. I will return it the following Wednesday (6 th June).
Lecture 20 Optical Characterization 2 Schroder: Chapters 2, 7, 10 1/68 Announcements Homework 5/6: Is online now. Due Wednesday May 30th at 10:00am. I will return it the following Wednesday (6 th June).
All measurement has a limit of precision and accuracy, and this must be taken into account when evaluating experimental results.
 Chapter 11: Measurement and data processing and analysis 11.1 Uncertainty and error in measurement and results All measurement has a limit of precision and accuracy, and this must be taken into account
Chapter 11: Measurement and data processing and analysis 11.1 Uncertainty and error in measurement and results All measurement has a limit of precision and accuracy, and this must be taken into account
Conjugated Systems. With conjugated double bonds resonance structures can be drawn
 Conjugated Systems Double bonds in conjugation behave differently than isolated double bonds With conjugated double bonds resonance structures can be drawn With isolated double bonds cannot draw resonance
Conjugated Systems Double bonds in conjugation behave differently than isolated double bonds With conjugated double bonds resonance structures can be drawn With isolated double bonds cannot draw resonance
hand and delocalization on the other, can be instructively exemplified and extended
 Text Related to Segment 8.0 00 Claude E. Wintner The ideas developed up to this point, concerning stereochemistry on the one hand and delocalization on the other, can be instructively exemplified and extended
Text Related to Segment 8.0 00 Claude E. Wintner The ideas developed up to this point, concerning stereochemistry on the one hand and delocalization on the other, can be instructively exemplified and extended
Light as a Transverse Wave.
 Waves and Superposition (Keating Chapter 21) The ray model for light (i.e. light travels in straight lines) can be used to explain a lot of phenomena (like basic object and image formation and even aberrations)
Waves and Superposition (Keating Chapter 21) The ray model for light (i.e. light travels in straight lines) can be used to explain a lot of phenomena (like basic object and image formation and even aberrations)
OPSE FINAL EXAM Fall 2016 YOU MUST SHOW YOUR WORK. ANSWERS THAT ARE NOT JUSTIFIED WILL BE GIVEN ZERO CREDIT.
 CLOSED BOOK. Equation Sheet is provided. YOU MUST SHOW YOUR WORK. ANSWERS THAT ARE NOT JUSTIFIED WILL BE GIVEN ZERO CREDIT. ALL NUMERICAL ANSERS MUST HAVE UNITS INDICATED. (Except dimensionless units like
CLOSED BOOK. Equation Sheet is provided. YOU MUST SHOW YOUR WORK. ANSWERS THAT ARE NOT JUSTIFIED WILL BE GIVEN ZERO CREDIT. ALL NUMERICAL ANSERS MUST HAVE UNITS INDICATED. (Except dimensionless units like
6. A solution of red Kool-Aid transmits light at a wavelength range of nm.
 I. Multiple Choice (15 pts) 1. FRET stands for a. Fluorescence Recovery Electron Transfer b. Fluorescence Resonance Energy Transfer c. Fluorescence Recovery Energy Transfer 2. Fluorescence involves the
I. Multiple Choice (15 pts) 1. FRET stands for a. Fluorescence Recovery Electron Transfer b. Fluorescence Resonance Energy Transfer c. Fluorescence Recovery Energy Transfer 2. Fluorescence involves the
Polarization of Light
 Polarization of Light A light beam which has all of the wave oscillations in a single plane of space is said to have total plane polarization. Light with an equal amount of oscillations in all directions
Polarization of Light A light beam which has all of the wave oscillations in a single plane of space is said to have total plane polarization. Light with an equal amount of oscillations in all directions
Chapter 33: ELECTROMAGNETIC WAVES 559
 Chapter 33: ELECTROMAGNETIC WAVES 1 Select the correct statement: A ultraviolet light has a longer wavelength than infrared B blue light has a higher frequency than x rays C radio waves have higher frequency
Chapter 33: ELECTROMAGNETIC WAVES 1 Select the correct statement: A ultraviolet light has a longer wavelength than infrared B blue light has a higher frequency than x rays C radio waves have higher frequency
Visible and IR Absorption Spectroscopy. Andrew Rouff and Kyle Chau
 Visible and IR Absorption Spectroscopy Andrew Rouff and Kyle Chau The Basics wavelength= (λ) original intensity= Ι o sample slab thickness= dl Final intensity= I f ε = molar extinction coefficient -di=
Visible and IR Absorption Spectroscopy Andrew Rouff and Kyle Chau The Basics wavelength= (λ) original intensity= Ι o sample slab thickness= dl Final intensity= I f ε = molar extinction coefficient -di=
17.1 Classes of Dienes
 17.1 Classes of Dienes There are three categories for dienes: Cumulated: pi bonds are adjacent. Conjugated: pi bonds are separated by exactly ONE single bond. Isolated: pi bonds are separated by any distance
17.1 Classes of Dienes There are three categories for dienes: Cumulated: pi bonds are adjacent. Conjugated: pi bonds are separated by exactly ONE single bond. Isolated: pi bonds are separated by any distance
PH 222-2C Fall Electromagnetic Waves Lectures Chapter 33 (Halliday/Resnick/Walker, Fundamentals of Physics 8 th edition)
 PH 222-2C Fall 2012 Electromagnetic Waves Lectures 21-22 Chapter 33 (Halliday/Resnick/Walker, Fundamentals of Physics 8 th edition) 1 Chapter 33 Electromagnetic Waves Today s information age is based almost
PH 222-2C Fall 2012 Electromagnetic Waves Lectures 21-22 Chapter 33 (Halliday/Resnick/Walker, Fundamentals of Physics 8 th edition) 1 Chapter 33 Electromagnetic Waves Today s information age is based almost
Design and Development of a Smartphone Based Visible Spectrophotometer for Analytical Applications
 Design and Development of a Smartphone Based Visible Spectrophotometer for Analytical Applications Bedanta Kr. Deka, D. Thakuria, H. Bora and S. Banerjee # Department of Physicis, B. Borooah College, Ulubari,
Design and Development of a Smartphone Based Visible Spectrophotometer for Analytical Applications Bedanta Kr. Deka, D. Thakuria, H. Bora and S. Banerjee # Department of Physicis, B. Borooah College, Ulubari,
Jones calculus for optical system
 2/14/17 Electromagnetic Processes In Dispersive Media, Lecture 6 1 Jones calculus for optical system T. Johnson Key concepts in the course so far What is meant by an electro-magnetic response? What characterises
2/14/17 Electromagnetic Processes In Dispersive Media, Lecture 6 1 Jones calculus for optical system T. Johnson Key concepts in the course so far What is meant by an electro-magnetic response? What characterises
Calculate a rate given a species concentration change.
 Kinetics Define a rate for a given process. Change in concentration of a reagent with time. A rate is always positive, and is usually referred to with only magnitude (i.e. no sign) Reaction rates can be
Kinetics Define a rate for a given process. Change in concentration of a reagent with time. A rate is always positive, and is usually referred to with only magnitude (i.e. no sign) Reaction rates can be
Electromagnetic Waves. Chapter 33 (Halliday/Resnick/Walker, Fundamentals of Physics 8 th edition)
 PH 222-3A Spring 2007 Electromagnetic Waves Lecture 22 Chapter 33 (Halliday/Resnick/Walker, Fundamentals of Physics 8 th edition) 1 Chapter 33 Electromagnetic Waves Today s information age is based almost
PH 222-3A Spring 2007 Electromagnetic Waves Lecture 22 Chapter 33 (Halliday/Resnick/Walker, Fundamentals of Physics 8 th edition) 1 Chapter 33 Electromagnetic Waves Today s information age is based almost
To be covered (and why) Spectroscopy of Proteins. UV-Vis Absorption. UV-Vis Absorption. Spectra
 To be covered (and why) Spectroscopy of Proteins General considerations UV-Vis Absorption quantitation Fluorescence hydrophobicity Foldedness FT-Infrared Foldedness ircular Dichroism Foldedness NMR (a
To be covered (and why) Spectroscopy of Proteins General considerations UV-Vis Absorption quantitation Fluorescence hydrophobicity Foldedness FT-Infrared Foldedness ircular Dichroism Foldedness NMR (a
4Types of Isomers. 1. Structural Isomers/(Constitutional) 2. Geometric Isomers/(Cis/Trans) 3. Optical Isomers A. Enantiomers B.
 4Types of Isomers 1. Structural Isomers/(Constitutional) 2. Geometric Isomers/(Cis/Trans) 3. Optical Isomers A. Enantiomers B. Diastereomers 4Types of Isomers C 4 10 C 4 10 O O O O O O O O O O O O C 3
4Types of Isomers 1. Structural Isomers/(Constitutional) 2. Geometric Isomers/(Cis/Trans) 3. Optical Isomers A. Enantiomers B. Diastereomers 4Types of Isomers C 4 10 C 4 10 O O O O O O O O O O O O C 3
William H. Brown & Christopher S. Foote
 Requests for permission to make copies of any part of the work should be mailed to:permissions Department, Harcourt Brace & Company, 6277 Sea Harbor Drive, Orlando, Florida 32887-6777 William H. Brown
Requests for permission to make copies of any part of the work should be mailed to:permissions Department, Harcourt Brace & Company, 6277 Sea Harbor Drive, Orlando, Florida 32887-6777 William H. Brown
Physics 313: Laboratory 8 - Polarization of Light Electric Fields
 Physics 313: Laboratory 8 - Polarization of Light Electric Fields Introduction: The electric fields that compose light have a magnitude, phase, and direction. The oscillating phase of the field and the
Physics 313: Laboratory 8 - Polarization of Light Electric Fields Introduction: The electric fields that compose light have a magnitude, phase, and direction. The oscillating phase of the field and the
Wave Propagation in Uniaxial Media. Reflection and Transmission at Interfaces
 Lecture 5: Crystal Optics Outline 1 Homogeneous, Anisotropic Media 2 Crystals 3 Plane Waves in Anisotropic Media 4 Wave Propagation in Uniaxial Media 5 Reflection and Transmission at Interfaces Christoph
Lecture 5: Crystal Optics Outline 1 Homogeneous, Anisotropic Media 2 Crystals 3 Plane Waves in Anisotropic Media 4 Wave Propagation in Uniaxial Media 5 Reflection and Transmission at Interfaces Christoph
CHAPTER 5. Stereoisomers
 CHAPTER 5 Stereoisomers We have already covered two kinds of isomerism: Constitutional Isomers (structural isomers) Stereoisomers Examples of Constitutional Isomers: Examples of Stereoisomers: Another
CHAPTER 5 Stereoisomers We have already covered two kinds of isomerism: Constitutional Isomers (structural isomers) Stereoisomers Examples of Constitutional Isomers: Examples of Stereoisomers: Another
Circular Dichroism. For students of HI Computational Structural Biology
 T H E U N I V E R S I T Y of T E X A S S C H O O L O F H E A L T H I N F O R M A T I O N S C I E N C E S A T H O U S T O N Circular Dichroism For students of HI 6001-125 Computational Structural Biology
T H E U N I V E R S I T Y of T E X A S S C H O O L O F H E A L T H I N F O R M A T I O N S C I E N C E S A T H O U S T O N Circular Dichroism For students of HI 6001-125 Computational Structural Biology
Lecture 4: Anisotropic Media. Dichroism. Optical Activity. Faraday Effect in Transparent Media. Stress Birefringence. Form Birefringence
 Lecture 4: Anisotropic Media Outline Dichroism Optical Activity 3 Faraday Effect in Transparent Media 4 Stress Birefringence 5 Form Birefringence 6 Electro-Optics Dichroism some materials exhibit different
Lecture 4: Anisotropic Media Outline Dichroism Optical Activity 3 Faraday Effect in Transparent Media 4 Stress Birefringence 5 Form Birefringence 6 Electro-Optics Dichroism some materials exhibit different
The Fundamentals of Spectroscopy: Theory BUILDING BETTER SCIENCE AGILENT AND YOU
 The Fundamentals of Spectroscopy: Theory BUILDING BETTER SCIENCE AGILENT AND YOU 1 Agilent is committed to the educational community and is willing to provide access to company-owned material. This slide
The Fundamentals of Spectroscopy: Theory BUILDING BETTER SCIENCE AGILENT AND YOU 1 Agilent is committed to the educational community and is willing to provide access to company-owned material. This slide
Physics 214 Course Overview
 Physics 214 Course Overview Lecturer: Mike Kagan Course topics Electromagnetic waves Optics Thin lenses Interference Diffraction Relativity Photons Matter waves Black Holes EM waves Intensity Polarization
Physics 214 Course Overview Lecturer: Mike Kagan Course topics Electromagnetic waves Optics Thin lenses Interference Diffraction Relativity Photons Matter waves Black Holes EM waves Intensity Polarization
Optics Polarization. Lana Sheridan. June 20, De Anza College
 Optics Polarization Lana Sheridan De Anza College June 20, 2018 Last time interference from thin films Newton s rings Overview the interferometer and gravitational waves polarization birefringence 7 Michelson
Optics Polarization Lana Sheridan De Anza College June 20, 2018 Last time interference from thin films Newton s rings Overview the interferometer and gravitational waves polarization birefringence 7 Michelson
Stereochemistry. 3-dimensional Aspects of Tetrahedral Atoms
 Stereochemistry 3-dimensional Aspects of Tetrahedral Atoms Chiral Entire molecules or simply atoms that do not possess a plane of symmetry are called chiral. Conversely, the term achiral is applied to
Stereochemistry 3-dimensional Aspects of Tetrahedral Atoms Chiral Entire molecules or simply atoms that do not possess a plane of symmetry are called chiral. Conversely, the term achiral is applied to
J- vs. H-type assembly: pentamethine cyanine (Cy5) as near IR chiroptical. reporter
 S1 Supporting Information: J- vs. H-type assembly: pentamethine cyanine (Cy5) as near IR chiroptical reporter Larysa I. Markova, a,b Vladimir L. Malinovskii, a Leonid D. Patsenker b and Robert Häner* a
S1 Supporting Information: J- vs. H-type assembly: pentamethine cyanine (Cy5) as near IR chiroptical reporter Larysa I. Markova, a,b Vladimir L. Malinovskii, a Leonid D. Patsenker b and Robert Häner* a
Light. November 101 Lect 11 1
 Light What is light? To start, what are the observed properties of light? Describe the intrinsic properties of light light by itself. Later, what are the interactions of light? What happens when light
Light What is light? To start, what are the observed properties of light? Describe the intrinsic properties of light light by itself. Later, what are the interactions of light? What happens when light
ANSWER KEY PAGE 1 of 11
 ANSWER KEY PAGE 1 of 11 UNIVERSITY OF MANITOBA DEPARTMENT OF CHEMISTRY CHEM 3390 STRUCTURAL TRANSFORMATIONS IN ORGANIC CHEMISTRY FINAL EXAMINATION Dr. Phil Hultin Tuesday December 13, 2011 9:00 am. NAME:
ANSWER KEY PAGE 1 of 11 UNIVERSITY OF MANITOBA DEPARTMENT OF CHEMISTRY CHEM 3390 STRUCTURAL TRANSFORMATIONS IN ORGANIC CHEMISTRY FINAL EXAMINATION Dr. Phil Hultin Tuesday December 13, 2011 9:00 am. NAME:
Model Answer (Paper code: AR-7112) M. Sc. (Physics) IV Semester Paper I: Laser Physics and Spectroscopy
 Model Answer (Paper code: AR-7112) M. Sc. (Physics) IV Semester Paper I: Laser Physics and Spectroscopy Section I Q1. Answer (i) (b) (ii) (d) (iii) (c) (iv) (c) (v) (a) (vi) (b) (vii) (b) (viii) (a) (ix)
Model Answer (Paper code: AR-7112) M. Sc. (Physics) IV Semester Paper I: Laser Physics and Spectroscopy Section I Q1. Answer (i) (b) (ii) (d) (iii) (c) (iv) (c) (v) (a) (vi) (b) (vii) (b) (viii) (a) (ix)
CHEM 261 Oct 11, Diastereomers. Enantiomers. Pheromones: from Greek pherein horman meaning to carry excitement. Discovered by Adolf Butenanot.
 EM 26 ct, 208 REALL: is Trans Trans Diastereomers Enantiomers Enantiomers have opposite stereochemistry at every stereocenter (chiral center) Diastereomers are all stereoisomers that are not enantiomers
EM 26 ct, 208 REALL: is Trans Trans Diastereomers Enantiomers Enantiomers have opposite stereochemistry at every stereocenter (chiral center) Diastereomers are all stereoisomers that are not enantiomers
a) Write the mechanism of Friedel-Crafts alkylation of ethyl benzene to give 1,4- diethylbenzene. Show all arrow pushing.
 a) Write the mechanism of Friedel-Crafts alkylation of ethyl benzene to give 1,4- diethylbenzene. Show all arrow pushing. Br AlBr 3 b) Using resonance and inductive effects, explain why an ethyl group
a) Write the mechanism of Friedel-Crafts alkylation of ethyl benzene to give 1,4- diethylbenzene. Show all arrow pushing. Br AlBr 3 b) Using resonance and inductive effects, explain why an ethyl group
Jones vector & matrices
 Jones vector & matrices Department of Physics 1 Matrix treatment of polarization Consider a light ray with an instantaneous E-vector as shown y E k, t = xe x (k, t) + ye y k, t E y E x x E x = E 0x e i
Jones vector & matrices Department of Physics 1 Matrix treatment of polarization Consider a light ray with an instantaneous E-vector as shown y E k, t = xe x (k, t) + ye y k, t E y E x x E x = E 0x e i
Types of Molecular Vibrations
 Important concepts in IR spectroscopy Vibrations that result in change of dipole moment give rise to IR absorptions. The oscillating electric field of the radiation couples with the molecular vibration
Important concepts in IR spectroscopy Vibrations that result in change of dipole moment give rise to IR absorptions. The oscillating electric field of the radiation couples with the molecular vibration
OPSE FINAL EXAM Fall 2015 YOU MUST SHOW YOUR WORK. ANSWERS THAT ARE NOT JUSTIFIED WILL BE GIVEN ZERO CREDIT.
 CLOSED BOOK. Equation Sheet is provided. YOU MUST SHOW YOUR WORK. ANSWERS THAT ARE NOT JUSTIFIED WILL BE GIVEN ZERO CREDIT. ALL NUMERICAL ANSERS MUST HAVE UNITS INDICATED. (Except dimensionless units like
CLOSED BOOK. Equation Sheet is provided. YOU MUST SHOW YOUR WORK. ANSWERS THAT ARE NOT JUSTIFIED WILL BE GIVEN ZERO CREDIT. ALL NUMERICAL ANSERS MUST HAVE UNITS INDICATED. (Except dimensionless units like
Electromagnetic fields and waves
 Electromagnetic fields and waves Maxwell s rainbow Outline Maxwell s equations Plane waves Pulses and group velocity Polarization of light Transmission and reflection at an interface Macroscopic Maxwell
Electromagnetic fields and waves Maxwell s rainbow Outline Maxwell s equations Plane waves Pulses and group velocity Polarization of light Transmission and reflection at an interface Macroscopic Maxwell
Organic Cume. Solvent Polarity Indicators
 Organic Cume University of Missouri-Columbia June 1995 Dr. Rainer Glaser Solvent Polarity Indicators Suggested Reading: Reichardt, C. Chem. Rev. 1994, 94, 2319-2358. Max. Question 1 24 Question 2 34 Question
Organic Cume University of Missouri-Columbia June 1995 Dr. Rainer Glaser Solvent Polarity Indicators Suggested Reading: Reichardt, C. Chem. Rev. 1994, 94, 2319-2358. Max. Question 1 24 Question 2 34 Question
Polarization of light
 Laboratory#8 Phys4480/5480 Dr. Cristian Bahrim Polarization of light Light is a transverse electromagnetic wave (EM) which travels due to an electric field and a magnetic field oscillating in phase and
Laboratory#8 Phys4480/5480 Dr. Cristian Bahrim Polarization of light Light is a transverse electromagnetic wave (EM) which travels due to an electric field and a magnetic field oscillating in phase and
Chapter 33. Electromagnetic Waves
 Chapter 33 Electromagnetic Waves Today s information age is based almost entirely on the physics of electromagnetic waves. The connection between electric and magnetic fields to produce light is own of
Chapter 33 Electromagnetic Waves Today s information age is based almost entirely on the physics of electromagnetic waves. The connection between electric and magnetic fields to produce light is own of
Conjugated Dienes and Ultraviolet Spectroscopy
 Conjugated Dienes and Ultraviolet Spectroscopy Key Words Conjugated Diene Resonance Structures Dienophiles Concerted Reaction Pericyclic Reaction Cycloaddition Reaction Bridged Bicyclic Compound Cyclic
Conjugated Dienes and Ultraviolet Spectroscopy Key Words Conjugated Diene Resonance Structures Dienophiles Concerted Reaction Pericyclic Reaction Cycloaddition Reaction Bridged Bicyclic Compound Cyclic
ECE 185 ELECTRO-OPTIC MODULATION OF LIGHT
 ECE 185 ELECTRO-OPTIC MODULATION OF LIGHT I. Objective: To study the Pockels electro-optic (EO) effect, and the property of light propagation in anisotropic medium, especially polarization-rotation effects.
ECE 185 ELECTRO-OPTIC MODULATION OF LIGHT I. Objective: To study the Pockels electro-optic (EO) effect, and the property of light propagation in anisotropic medium, especially polarization-rotation effects.
Practical KK transformation and anisotropy
 Optical spectroscopy in materials science 6. Practical KK transformation and anisotropy Kamarás Katalin MTA Wigner FK kamaras.katalin@wigner.mta.hu Optical spectroscopy in materials science 6. 1 Optical
Optical spectroscopy in materials science 6. Practical KK transformation and anisotropy Kamarás Katalin MTA Wigner FK kamaras.katalin@wigner.mta.hu Optical spectroscopy in materials science 6. 1 Optical
A high-accuracy universal polarimeter study of optical anisotropy and optical activity in laminated collagen membranes
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information (ESI) A high-accuracy universal polarimeter study of optical
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information (ESI) A high-accuracy universal polarimeter study of optical
Name: Student Number: University of Manitoba - Department of Chemistry CHEM Introductory Organic Chemistry II - Term Test 1
 Name: Student Number: University of Manitoba - Department of Chemistry CHEM 2220 - Introductory Organic Chemistry II - Term Test 1 Thursday, February 11, 2016; 7-9 PM This is a 2-hour test, marked out
Name: Student Number: University of Manitoba - Department of Chemistry CHEM 2220 - Introductory Organic Chemistry II - Term Test 1 Thursday, February 11, 2016; 7-9 PM This is a 2-hour test, marked out
Lab 8: L-6, Polarization Lab Worksheet
 Lab 8: L-6, Polarization Lab Worksheet Name This sheet is the lab document your TA will use to score your lab. It is to be turned in at the end of lab. To receive full credit you must use complete sentences
Lab 8: L-6, Polarization Lab Worksheet Name This sheet is the lab document your TA will use to score your lab. It is to be turned in at the end of lab. To receive full credit you must use complete sentences
Electromagnetic Waves
 May 7, 2008 1 1 J.D.Jackson, Classical Electrodynamics, 2nd Edition, Section 7 Maxwell Equations In a region of space where there are no free sources (ρ = 0, J = 0), Maxwell s equations reduce to a simple
May 7, 2008 1 1 J.D.Jackson, Classical Electrodynamics, 2nd Edition, Section 7 Maxwell Equations In a region of space where there are no free sources (ρ = 0, J = 0), Maxwell s equations reduce to a simple
Light Waves and Polarization
 Light Waves and Polarization Xavier Fernando Ryerson Communications Lab http://www.ee.ryerson.ca/~fernando The Nature of Light There are three theories explain the nature of light: Quantum Theory Light
Light Waves and Polarization Xavier Fernando Ryerson Communications Lab http://www.ee.ryerson.ca/~fernando The Nature of Light There are three theories explain the nature of light: Quantum Theory Light
Retinal Proteins (Rhodopsins) Vision, Bioenergetics, Phototaxis. Bacteriorhodopsin s Photocycle. Bacteriorhodopsin s Photocycle
 Molecular chanisms of Photoactivation and Spectral Tuning in Retinal Proteins Emad Tajkhorshid Theoretical and Computational Biophysics Group Beckman Institute University of Illinois at Urbana-Champaign
Molecular chanisms of Photoactivation and Spectral Tuning in Retinal Proteins Emad Tajkhorshid Theoretical and Computational Biophysics Group Beckman Institute University of Illinois at Urbana-Champaign
