The weathering of biotite in some Aberdeenshire soils
|
|
|
- Phyllis Willis
- 6 years ago
- Views:
Transcription
1 1080 The weathering of biotite in some Aberdeenshire soils By M. J. WILSOn, B.Sc., Ph.D. The Macaulay Institute for Soil Research, Craigiebuckler, Aberdeen [Taken as read 9 June 1966] Summary. The weathering of biotite in some freely drained soils developed from a biotite-rich quartz gabbro near Strathdon, Aberdeenshire, has been studied. Using a combination of X-ray, chemical, differential thermal, infra-red, and optical techniques, it was found that the biotite weathers to an intergrade aluminous vermiculite-chlorite together with zones of kaolinite and gibbsite. The crystallographic axes of biotite and included kaolinite are common and parallel and it is proposed that both kaolinite and gibbsite zones crystallized epitaxially within opened out cleavage spaces of the host biotite. The development of kaolinite and gibbsite in this manner could have occurred relatively quickly and the significance of this with regard to generalized pedogenic weathering reactions of biotite is discussed. I T has long been known that biotite releases potassium into the soil during the course of natural weathering although the associated mineralogical changes have remained obscure until fairly recently. Early studies of this problem appeared to indicate that biotite weathered towards a kaolinitic end-product. Thus, Denison et al. (1929), in a major study pursued mainly by chemical and optical means, came to this conclusion and Kerr (1930) described a weathered granodiorite in which biotite had changed to kaolinite via a gel-like stage. Later investigators found that biotite also weathered to hydrobiotite (Wager, 1945), vermiculite (Walker, 1949), and montmorillonite (MacEwan, 1953) and it became clear that biotite weathered to different end-products under different weathering environments. In a more recent study, Kato (1965) showed that biotite in a freely drained Japanese soil decomposed mainly to trioctahedral aluminium vermiculite and also partly to kaolinite and gibbsite. A brief preliminary account of the weathered biotites studied here (Wilson, 1966) showed them to consist of a 14 A intergradient aluminous vermiculite-chlorite and kaolinite. Materials and methods Source of material. The weathered biotite used in this study was collected from three soils from the Strathdon area, Aberdeenshire. Each of these soils is freely drained and appears to be directly related to the
2 WEATHERING OF BIOTITE 1081 same parent material, namely a biotite-rich quartz gabbro. This is a medium-grained holocrystalline rock whose major mineral constituents include labradorite, pyroxene, amphibole, biotite, and quartz. The pyroxene occurs as colourless, well-shaped prisms and basal plates and frequently shows various stages of alteration to amphibole. It may be surrounded by an amphibole rim or may be almost entirely replaced, the only sign of the original mineral being in the typical pyroxene shape, or in a small central core. Both augite and hypersthene are present, the former being dominant. The amphibole is usually pale green in colour, slightly or non-pleochroic, and is occasionally fibrous. It has a small or straight extinction angle and when it replaces pyroxenes it assumes a variety of orientations. X-ray powder data for the separated amphibole show a good fit for tremolite. The biotite occurs as large, deep reddish brown plates. The mafic minerals are enclosed within an unaltered subhedral mosaic of labradorite with minor amounts of interstitial quartz. Petrographically, this rock bears a close resemblance to the quartz gabbro of the Haddo House district described by Read (1935) as an uncontaminated rock showing evidence of end-stage modification and autometamorphism. The locations of the three soils sampled are: Soil 1, Near the 125~-ft spot height on the west side of the minor road connecting the B973 road with Kirkton of Glen Buchat. Soil 2. Approximately 1000 yards SW of Belnaboth, about 1300 ft above sea level at the head of a small stream. Soil 3. On roadside just south of Tornagawn. (NJ/372145) (NJ/357156) (NJ/343146) The sedentary nature of Soil 1 is clear in the field since the underlying parent rock may be directly observed. The parent materials of Soils 2 and 3 could not be seen but a mineral examination indicates their uncontaminated nature. Microscopic study of the heavy and light sand fractions of the three soils showed little difference in the nature or proportions of their component minerals, although the light fraction of Soil 3 contains a little more quartz than those of Soils 1 and 2. Methods of Investigation. Weathered biotite is very abundant in all the light, fine sand fractions (50 to 200/~) and pure separates could be made with a magnetic separator, or simply by placing the sample on a stiff piece of paper and gently tapping so that the more rounded grains roll free leaving behind the platy biotite. The purity of each separate
3 1082 ~. J. WILSOn" On was always checked by examination under the binocular microscope. X-ray powder photographs were obtained using a Raymax diffraction unit (Co-Ka radiation with an iron filter) and 9-cm precision powder cameras. Single crystal photographs were taken using a Unicam S. 35 single crystal Weissenberg goniometer (Cu-Ka radiation with a nickel filter) the mineral having been previously correctly oriented by observing the interference figure in convergent light. Differential thermal curves were obtained in the apparatus described by Mitchell and Mackenzie (1959) using a heating rate of 10 ~ C per minute and infra-red spectra using a Grubb-Parsons S. 4 double beam spectrometer. The cation-exchange capacities of the weathered biotites were determined by the method of Mackenzie (1951). Silica and alumina were determined colorimetrically. Results X-ray powder data for separated weathered biotite flakes between 50 and 200 ~ in diameter from Soils 1, 2, and 3 are shown in table I, together with measurements of the fresh biotite from the parent rock of Soil 1. The results in this table are characteristic of many samples of weathered biotite taken from different parts of the three soil profiles. Several features of these powder patterns are of interest and these will be discussed in more detail. Almost invariably a first order reflection occurs between 13-8 and 14.6 _~ and at first it was considered that this spacing indicated the conversion of biotite to vermiculite, which has been shown to occur by Walker (1949). However, this interpretation was not confirmed by the behaviour of the mineral on heating or on potassium saturation. Walker (1961) showed that true vermiculite collapses to about 10 A on heating to 300~ for several hours. Such heating of the weathered biotite, followed by immediate sealing in a glass capillary tube to prevent rehydration, induced collapse only to about 11"50 A. Again, vermiculite derived from the weathering of biotite should collapse easily to 10 A on potassium saturation (Weaver, 1958), but the weathered flakes failed to collapse below 13"0 _~ following this treatment. These results indicated the possible presence of interlayer contaminants, in particular non-exchangeable interlayer aluminium, which has been shown to inhibit lattice contraction (Rich and Obenshain, 1955). In order to extract interlayer aluminium the weathered flakes were treated with boiling 0"5 N NaOH for about l0 rain and subsequently re-examined by X-rays. NaOH treatment brings about a significant
4 WEATKEPJNG OF :RIOTITE 1083 TABLE I. X-ray powder dat,~ for fresh and weathered biotites from soils 1, 2, and 3 Fresh Biotite Weathered ]Motite 1 Weathered Biotite 2 Weathered Biotite 3 d I d I 14.5~ vs 138~ vs 10'15 I vs m 7"20 m 7.3 s 4'85 m 4-~5 s 4'62 w 4"65 w 4.63 m 4'42 w 4"40 s vb 4'20 ~'w 4"05 vw 3.75 m 3'60 ms 3.55 s 3"35 vs 3"24 w 3-17 wb 3.06 w 2"92 wb 2-88 m 2-82 m 2"75 s 2.62 s 2.64 m 2.64 ms 2.53 s 2,56 ms 2.49 m 2"44 s 2"39 m 2.39 ms 2"27 w 2'23 m 2"20 ins 2'05 ms 2-00 w 1-91 ms 1.72 ms 1-68 Yw 1.74 vw 1-67 ms 1.69 vw 1.63 u 1.65 w 1.61 vw 1.54 s 1.54 ms 1.54 ms 1"49 w 1"49 nl d ~ s 7"3 ms b 4.75 m 4.48 ms bd 3"88 w 3-55 ms 3-36 m 3-14 w 2.57 m 2.34 m 2.23 w 2-01 w 1.82 vw m difference in the behaviour of the mineral on heating to 300 ~ C and on K-saturation. Before NaOH treatment After NaOH treatment Sample no Untreated K-saturated Heated to 300 ~ C Similar results were also obtained using the Na2C0 a method of Follett et al. (1965) for the dissolution of amorphous alumina and silica. The weight percentage total of A120 ~ extracted by this method ranged from 2.58 ~ to 5.40 ~ and clearly much of this must have been located in interlayer positions. The amount of silica extracted varied from 2.92 ~ to 4-67 %. Jackson (1963) has summarized recent work showing that interlayer atumination is a fairly common occurrence in expanded 2:1 layer silicates of soil clays. The interlayer positions of these silicates may be partially filled with positively charged aquo-hydroxoaluminium ions, e.g. A16(OH)12(OH2)12 8+, thus creating a heterogeneous intergrade between true 2 : 1 and 2 : 1 : 1 silicates. It seems probable that the weathered biotite has been partly converted to such an
5 1084 M.J. WILSON ON intergrade, the expanded interlayer positions being occupied by islands of hydroxo-aquo-aluminium, possibly associated with silica, and that it may be regarded, therefore, as an intergrade aluminous vermiculitechlorite. Table I also shows the regular occurrence of a line at about 7.3 ~_, which persists on heating to 300 ~ C indicating that it is not a second order of the 1t/~ line, together with what appears to be a dioctahedral 060 line at 1.49 A. These lines have been observed for powders and single flakes. Heating to 600 ~ C for several hours causes them to disappear, whilst the trioctahedral 060 line associated with biotite remains unaffected. This appears to indicate the presence of a kaolinite mineral within the biotite. As to the nature of the kaolinite mineral, the slightly high spacing of the first order reflection, 7"3 -~ as opposed to 7-15 A, suggests the possibility of halloysite. The potassium acetate method of Wada (1961)was used in order to test this possibility. Flakes from Soil 3, which showed a strong line at 7"3 A, and samples of pure kaolinite and halloysite were ground with potassium acetate. The pure minerals and the kaolinite mineral in the biotite complexed with the potassium acetate and expanded to 14. When the samples were washed with water and air dried at 65 ~o R.H. the halloysite collapsed to 10 A whilst the kaolinite and the kaolinite in the biotite collapsed to 7-1 and 7"3 J~ respectively. It is apparent, therefore, that the behaviour of the kaolinite mineral is more like that of kaolinite than halloysite. One reason for the 001 spacing being at 7"3 ~ may be the possible presence of a small number of interstratified 10 A biotite units within tile kaolinite crystals. Finally, the X-ray powder data for the weathered biotites frequently show a line at about 4-8 A. This is characteristic of gibbsite and suggests the presence of this mineral within the biotite flakes and differential thermal and infra-red results support this interpretation. The powder pattern of the biotites of Soil 3 does not contain a line at 4-8 J~ indicating the absence of gibbsite in these flakes. To recapitulate, therefore, using a combination of X-ray diffraction analysis and auxiliary chemical treatments it has been shown that biotite may weather to a complex 14 A intergrade aluminous vermiculitechlorite, with kaolinite and gibbsite. X-ray single crystal results. In order to investigate the orientation relationship of the kaolinite zones in the weathered biotite a normal beam equatorial Weissenberg photograph rotated about the a-axis was taken of a weathered flake from Soil 2. All the reflections appear as broadened elongated streaks probably indicating some degree of internal
6 WEATHERING OF BIOTITE 1085 disorder. The observed 001 and 0k0 reflections can be attributed to a combination of weathered biotite, kaolinite, and gibbsite. To check this interpretation the flake was heated to 600 ~ C for several hours and another zero layer photograph taken. Table II shows that those reflections attributed to kaolinite and gibbsite have disappeared. The 00l TABLn IL X-ray single crystal data for weathered biotite flake from Soil 2 Weathered biotite flalce dok0 I Mineral d0o / I Mineral 4.60 s Biotite 4.80 m Gibbsite 2.23 w Kaolinite 3.50 s Kaolinite 1-54 vs Biotite 3.02 w Biotite (broad) 2-17 ms Biotite 2.05 ms Biotite 1.44 w 1-30 w Weathered biotite fla~e heated at 600 ~ C 4.67 ms Biotite 3.08 s Biotite 1-54 s Biotite 2"33 vw Biotite 1-86 vw Biotite 1.54 w Biotite 1.32 w Biotite reflections of the heated flake now fall into a series of orders indicating collapse of the weathered biotite along the c-axis to 9.3 A. Along the b-axis the 2"23 X 040 kaolinite reflection vanishes on heating, whilst the 1.54 A reflection, previously very diffuse and so obscuring a separate dioctahedral reflection at 1.49 A, becomes much more sharply defined. These results appear to indicate that the crystallographic axes of the biotite and included kaolinite zones are common and parallel. The gibbsite is probably similarly oriented. Differential thermal curves for weathered biotites collected from the upper and lower parts of the soil profiles are shown in fig. 1. It is clear that the curves of the biotites from Soils 1 and 2 are similar--with the two most prominent endothermic reactions at about 275~ and between 480 ~ C and 500 ~ C. In addition there are weak endothermic effects at about 600~ and 800~ in some of the samples. Almost certainly the gibbsite present in the flakes is responsible for the reactions at about 275~ whilst the major endotherm is probably due to the dehydroxylation of kaolinite. The biotites from Soil 3 produce a different thermal curve. Here the gibbsite reaction is absent, thus confirming the X-ray data. The flakes from the upper part of soil profile 3 show a
7 1086 ~. J. WILSON ON series of reactions characteristic of kaolinite and here again it may be noted that the powder pattern of this material showed a strong line at 7"3 _~. Biotite from the lower part of this soil profile lacks the high temperature exothermic reaction of kaolinite but shows a large endothermie peak at 815 ~ C. The reason for this is unknown. a, d, ~ ~ _. / ~ J ~ ~ " f. ~ ' ~ 484 log FIG. 1. Differential thermal analysis curves for: a, unweathered biotite; b to g, weathered biotites, from: b and c, Soil l; d and e, Soil 2;f and g, Soil 3. For each soil, the first curve is for biotite from the upper horizon, the second for biotite from the lower horizon. The infra-red spectra of weathered biotites from Soils 2 and 3 and of the fresh biotite are shown in fig. 2. The biotite from Soil 2 contains a well-crystallized kaolinite with OH-stretching absorption bands at 3697, 3669, 3652, and 3620 cm -1 (Russell and Farmer, 1964) and also gibbsite with OH bands at 3536 and 34:79 cm -1. A more poorly crystallized kaolinite is present in the biotite from Soil 3, as indicated by the absence of the 3669 and 3652 cm -1 bands, but gibbsite was not detected. The latter sample also has a rather broad OH-stretching band near 3575 cm -1, probably due to chloritic interlayers shown to be present in some flakes by X-ray evidence. Both weathered biotites show a rather broad low-frequency OH-stretching band of the type that has been associated with allolohanes (Mitchell et al., 1964). The cation-exchange capacities of the weathered biotite flakes from the
8 WEATHERING OF BIOTITE I A ~ ~82 C. 2"6 2" " p FIo. 2. Infra-red absorption curves for: A, weathered biotite from Soil 2; ~, weathered biotite from Soil 3; c, fresh biotite. upper and lower parts of all three soil profiles vary between 23.2 and 69"6 m. eq/100 g. Soil No. 1 Soil No. 2 Soil ~To " 34.2 m. eq " 24.9 m. eq. 3-6" 23.2 m. eq. 20~24" " " 69.6 Since the cation-exchange capacity of unweathered biotite is only 9 m.eq/]00 g it appears that weathering has induced a considerable increase. Probably in view of their abundance the weathered biotites account for a large proportion of the total soil exchange capacity. It
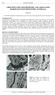
9 1088 M.J. w;lso:~ ON may be noted that the cation-exchange capacity of the weathered flakes from Soils 2 and 3 shows a marked increase in the lower horizons, but more work is needed before it can be established whether this is a feature of general significance. OTtical data. The weathered biotites were studied optically in the light fine sand fractions and also in slides cut from impregnated soils. Study by the former method is confined to the preferentially oriented (001) cleavage surfaces. However, sections cut from impregnated soil blocks do not suffer this disadvantage and here the biotite may be studied in a variety of attitudes. The (001) cleavage sections are usually pale yellowish brown, nonpleochroic and of low birefringence. The fi refractive indices of the weathered flakes are considerably lower than the indices of the fresh unweathered biotite. The fresh biotite had fi 1-625, while the weathered samples had: Soil 1, to 1-588; Soil 2, to 1.583; Soil 3, to This accords with the general findings of many previous workers (e.g. Denison et al., 1929; Walker, 1949). In convergent light the cleavage flakes show a biaxial negative interference figure with a small optic axial angle ranging from 5 ~ to 15 ~ approximately. The figure may often be distorted and its definition poor. The surfaces of the biotites frequently show micromorphological features comparable to those described by Raman and Jackson (1965) in their electron-optical study of mica surface morphology during laboratory weathering. These include various discontinuities such as curling and scrolling of mica layers and edges and fractures oriented at 120 ~ angles (plate XIII, a-d). From plate XIII, e, which shows the characteristic appearance of many of the mica flakes between crossed nicols, it is clear that different parts of the same crystal are in different optical orientations. This probably results from the folded and discontinuous nature of the mica layers and perhaps also from the more acute internal disorder arising from the complex mosaic weathering of mica as envisaged by Jackson (1965). The edges of the weathered biotites are seen in thin sections of impregnated soil blocks. The most prominent feature is the way the flakes are split and opened out along the cleavage planes (pl. XIII, f and g). Occasionally, these spaces are occupied by a mineral showing low relief and birefringence and a length-slow orientation. Almost certainly these are the phases of kaolinite or gibbsite or both detected by X-ray diffraction, d.t.a., and infra-red analysis, which may now be referred to as zones. Because of their low birefringence these zones may be more easily
10 WEATHERING OF BIOTITE 1089 discerned with the gypsum plate, which, with the coincidence of the slow vibration directions, causes them to stand out as vivid blue streaks in sharp contrast to the biotite. Invariably the slow directions of the zones and of the biotite coincide indicating the controlling influence of the host mineral. Discussion From the results quoted it is clear that the weathering of biotite involves expansion in the c direction, interlayer alumination, formation of gibbsite zones, and formation of kaolinite zones. The natural weathering of biotite to vermiculite involving a lattice expansion from 10 to 14 A was shown by Walker (1949). This transformation proceeds essentially through the replacement of interlayer potassium by other ions and by water, together with such changes as oxidation of octahedral iron. Jackson (1963a and 1963b) has described the way in which interlayer spaces of expanded 2:1 layer silicates in soil clays may be aluminated and the interlayer alumination of the weathered biotites described above may have proceeded in a comparable manner. Removal of potassium with concurrent lattice expansion provides negative surfaces on to which positively charged hydroxo-aquo-aluminium ions become attached so binding the sheets together. Eventual polymerization of this complex then creates an intergrade vermiculite-chlorite. In this particular instance the aluminium is probably derived from hydrolysis of the labradorite of the parent material--which would also result in the release of some silica--and indeed microscopic examination of this mineral in the fine sand fraction clearly shows features characteristic of solution. Edges invariably present an etched or ragged appearance, usually due to differential solution of the feldspar along adjacent twin lamellae. The formation of gibbsite zones within the biotite Crystals may be favoured by the tendency of the biotite to split and open out during the course of weathering (P1. XIII, f and g). It is significant in this respect that Slaughter and Milne (1960) were able to synthesize gibbsite, attached to one surface only, in the interlayer spaces of swollen montmorillonite. Barnhisel and Rich (1963) also achieved the formation of gibbsite in the laboratory under acid conditions from aluminiuminterlayered montmorillonite. Thus it seems likely that the splitting of the biotite flakes during weathering presents fresh surfaces for the organization and precipitation of introduced hydroxy-aluminium together with the space in which discrete zones of gibbsite can grow.
11 1090 ~. J. WILSON ON Kaolinite zones could grow in the biotite crystal in a like manner. On the other hand, a topotactic weathering transformation of expanded 2:1 layer silicate clays has been suggested by several workers. For example, Glenn et al. (1960) considered that montmorillonite weathered to kaolinite via the intermediate stage of intergradient vermiculitechlorite. Such a transformation involves complete interlayer alumination of the montmorillonite and also inversion of silica tetrahedra. The evolution of kaolinite is thus envisaged as proceeding along a 2:1-2:2-1:1 pathway. A similar topotactie change from biotite to kaolinite, however, would be drastic since it would involve: formation of a complete gibbsite sheet in the interlayer spaces; conversion of the octahedral layer from tri- to dioctahedral with the expulsion of iron and nlagncsium and the introduction of aluminium; introduction of Si 4+ ions into the tetrahedral layer; and tetrahedral inversion. Furthermore, these changes would occur only at particular parts of the biotite structure. In general the building up of kaolinite zones by epitaxial crystallization af[ords a more acceptable explanation. Thus initial weathering opens out the biotite flakes along the cleavage planes. The exposed surfaces then act as templates that control the orientation of the kaolinite during subsequent growth presumably from an alumina-silica gel. The precise chemical and physical conditions that could induce such a crystallization are unknown and recent work by De Kimpe et al. (1964) indicates kaolinite synthesis at low temperatures and pressures to be exceptionally difficult and that it is essential for six-fold co-ordinated aluminium to be stabilized; in the presence of silica this Can only be achieved at low pi-i. The same authors also found that seeding an alumina-silica gel with crystalline muscovite brought about an appreciable yield of kaolinite, although this occurs only at slightly elevated temperatures and pressures. In a general analysis of the pedogenic weathering of micas, including biotite, Jackson (1963 a, b) proposed the following sequence of reactions, most of which occur in the clay-size fraction of the soil : Pedogenic Mica -~ Yermiculite -~ 2 : 1-2 : 2 14 _~ ~ A1 ---* Kaolinite (Biotite) intergrade Chlorite and Halloysite The above sequence implies that change to each succeeding phase is complete. However, it has been shown previously that single crystals of the weathered biotite up to 1 mm in diameter may contain several
12 WEATHERING O:F BIOTITE 1091 different phases and that therefore the reaction outlined above is inapplicable in this instance. Although the exact relationship between aluminium interlayering and kaolinite and gibbsite zonation in biotites is unknown, it seems probable that their formation was relatively rapid and contemporaneous. Thus biotite weathered in this manner has been found in only slightly disintegrated rock where visually the flakes look fairly fresh. Provided chemical conditions are suitable, therefore, there seems to be no reason why clay minerals such as kaolinite or gibbsite, which are usually indicators of relatively intense weathering, should not crystallize epitaxially almost at the inception of the decomposition of a mineral. The breakdown of the Strathdon biotite may be envisaged as occurring as follows : the first stage involves expansion of the biotite to vermiculite with some concurrent interlayer alumination; subsequently, there is a build up of interlayer aluminium thereby creating an aluminous intergrade vermiculite-chlorite, together with splitting along the cleavage planes permitting epitaxial crystallization of kaolinite and gibbsite. Further weathering could result in the destruction of the intergradient mineral leaving pseudomorphs of kaolinite and gibbsite; it is noteworthy that kaolinite pseudomorphs after biotite have been found by the writer in the coarse sand fraction of Soil 3. These reactions may thus be formalized: Biotite (4) -> Vermiculite (8) Intergrade vermiculite-chlorite (9) --> Intergrade vermiculite-chlorite (9) --> Kaolinite (10) Kaolinite (10) Gibbsite (11) Gibbsite (11) where the bracketed numbers represent weathering indexes as proposed by Jackson et al. (1948). Kaolinite and gibbsite are the final products of weathering because relatively they are the most resistant. Conclusions The present study has shown by a variety of techniques that the biotites in some freely drained soils near Strathdon, Aberdeenshire, weather to a complex polycomponent assemblage of aluminous vermiculite-chlorite with zones of kaolinite and gibbsite. Since flakes of this nature up to 1 mm in diameter have been found in only slightly disintegrated rock it is considered that these changes occurred relatively
13 TYLER 1092 M.J. WILSON ON quickly. The kaolinite and gibbsite zones crystallized epitaxially within cleavage cracks and do not represent intensely altered parts of the biotite structure. Walker (1949) showed that under intermediate drain- age conditions biotite changed to vermiculite whilst MacEwan (1953) discovered a biotite-to-montmorillonite transformation in some poorly drained soils. It is possible that the changes described above are characteristic of a freely drained environment, although the precise conditions which favour such weathering are at present unknown. Aclcnowledgements. The writer wishes to thank Mr. W. A. Mitchell and Mr. A. P. Thomson for much help and advice during the course of this investigation. He is also grateful to Mr. A. P. Thomson for the X-ray determinations, to Mr. A. C. Birnie for the differential thermal results, to Dr. V. C. Farmer for obtaining and interpreting the infra-red spectra, and to Mr. B. F. L. Smith for silica and alumina determinations. Thanks are also due to Mr. E. L. Birse who drew the writer's attention to these soils in the first place. References BARN~ISEL (R. I.) and RICH (C. I.), Proc. Soil Sci. Soc. Amer., vol. 27, p DENISON (I. A.), FRY (W. H.), and GILn (P. L.), Teeh. Bull. U.S. Dept. Agric. no. 128, p. 1. DE KIMrE (C.), GASTUC~E (M. C.), and BRINDLEY (G. W.), Amer. Min., vol. 49, p. 1. FOLLETT (E. A. C.), MITCI~ELL (B. D.), McHARDu (W. J.), and SMIT~ (B. F. L.), Clay Min. Bull., vol. 6, p. 23. GLENN (1%. C.), JACKSON (M. L.), HOLE (F. D.), and LE~ (G. B.), Clays and Clay Minerals, 8th Conf., Pergamon Press, London, p. 63. JACKSON (M. L.), 1963a. Proc. Soil Sci. Soc. Amer., vol. 27, p b. Clays and Clay Minerals, llth Conf., Pergamon Press, New York, p. 29. Soil Sci., vol. 99, p HSEuNo (Y.), COREY (1%. B.), EVANS (E. J.), and VAN DEN H~UV~L (R. C.), Proc. Soil Sci. Soc. Amer., vol. 16, p. 3. (S. A.), WILLIS (A. L.), BOURBEAU (G. A.), and PENNINGTON (1%. P.), Journ. Phys. and Colloid. Chem., vol. 52, p KATO (Y.), Soil Sci. and Plant Nutrition, vol. 11, p. 30. KERR (P. F.), Amer. Min., vol. 15, p MAcEwAN (D. M. C.), Clay Min. Bull., vol. 2, p MACKENZIE (1%. C.), Journ. Colloid Sci., vol. 6, p MITC]~ELL (B. D.), FARMER (V. C.), and McHARDY (W. H.), Advances in Agronomy, vol. 16, p and MACKENZIE (R. C.), t959. Clay Min. Bull., vol. 4, p %AM~_N (K. V.) and JACKSON (M. L.), Proc. Soil Sci. Soc. Amer., vol. 29, p %EAD (H. H.), Quart. Journ. Geol. Soc., vol. 91, p %Icg (C. I.) and OBE~S~AIN (S. S.), Proc. Soil Sci. Soe. Amer., vol. 19, p %USSELL (J. D.) and FARMER (V. C.), Clay Min. Bull., vol. 5, p SLAUGHTER (M.) and MILNE (I. H.), Proc. 7th Confl Clays and Clay Min., pp WADA (K.), Amer. Min., vol. 46, p. 78. WAOER (L. 1%.), Proc. Yorks. Geol. Soc., vol. 23, p WALKER (G. F.), Min. Mag., vol. 28, p. 693.
14 WEATHERING OF BIOTITE 1093 WALKER (G. F.), 'Vermiculite Minerals' in 'The X-ray Identification and Crystal Structures of Clay Minerals'. Edited by G. Brown. Min. Soc., London, p WEAVER (C. E.), Amer. Min., vol. 43, p WILson (M, J.), Nature, vol. 210, p [Manuscript received 25 March 1966] EXPLANATION OF PLATE XIII FIO. a. Weathered biotite from Soil 1 showing curled edges and typical surface morphology. 650 FIo. b. Weathered biotit~soil 1--showing surface scrolling of layers FIG. c. Weathered biotite--soil 2--showing surface irregularities and cracks FIG. d. Weathered biotite--soil 2--showing scrolls, surface irregularities and cracks at 120 ~ angles FIG. e. As above--characteristic appearance of weathered biotites between crossed nicols. FiG. f. Weathered biotite--soil 2--split and opened out along cleavage planes FIG. g. Weathered biotite Soil 2--showing exfoliated appearance. 250, crossed nicols. 3z
15 MIN. MAG. VOL. 35 Plate X I I I ]~. J. WILSON." WEATHERING OF BIOTITE
THE USE OF PIPERIDINE AS AN AID TO CLAY-MINERAL IDENTIFICATION
 THE USE OF PIPERIDINE AS AN AID TO CLAY-MINERAL IDENTIFICATION By J. M. OADES* and W. N. TOWNSEND Department of Agriculture, The University of Leeds. [Received 30th August, 1962] ABSTRACT It is suggested
THE USE OF PIPERIDINE AS AN AID TO CLAY-MINERAL IDENTIFICATION By J. M. OADES* and W. N. TOWNSEND Department of Agriculture, The University of Leeds. [Received 30th August, 1962] ABSTRACT It is suggested
MINERAL CONTENT AND DISTRIBUTION AS INDEXES OF WEATHERING IN THE OMEGA AND AHMEEK SOILS OF NORTHERN WISCONSIN
 MINERAL CONTENT AND DISTRIBUTION AS INDEXES OF WEATHERING IN THE OMEGA AND AHMEEK SOILS OF NORTHERN WISCONSIN By L. D. WHITTIG 1 AND M. L. JACKSON University of Wisconsin, Madison, Wisconsin ABSTRACT Quantitative
MINERAL CONTENT AND DISTRIBUTION AS INDEXES OF WEATHERING IN THE OMEGA AND AHMEEK SOILS OF NORTHERN WISCONSIN By L. D. WHITTIG 1 AND M. L. JACKSON University of Wisconsin, Madison, Wisconsin ABSTRACT Quantitative
FROM A BIOTITE-HORNBLENDE ROCK
 Clay Minerals (1970) 8, 435. A STUDY OF WEATHERING IN A SOIL DERIVED FROM A BIOTITE-HORNBLENDE ROCK II. THE WEATHERING OF HORNBLENDE M. J. WILSON ANt) V. C. FARMER The Macaulay Institute for Soil Research,
Clay Minerals (1970) 8, 435. A STUDY OF WEATHERING IN A SOIL DERIVED FROM A BIOTITE-HORNBLENDE ROCK II. THE WEATHERING OF HORNBLENDE M. J. WILSON ANt) V. C. FARMER The Macaulay Institute for Soil Research,
The effect of isomorphous substitutions on the intensities of (OO1) reflections of mica- and chlorite-type structures.
 657 The effect of isomorphous substitutions on the intensities of (OO1) reflections of mica- and chlorite-type structures. By GEORGE BROWN', B.Sc. Pedology Department, Rothamsted Experimental Station,
657 The effect of isomorphous substitutions on the intensities of (OO1) reflections of mica- and chlorite-type structures. By GEORGE BROWN', B.Sc. Pedology Department, Rothamsted Experimental Station,
Trioctahedral minerals in the soil-clays of north-east Scotland.
 72 Trioctahedral minerals in the soil-clays of north-east Scotland. By G. F. WALKER, B.Sc., Ph.D. Macaulay Institute for Soil Research, Aberdeen. [Read at the meeting of the Clay Minerals Group, November
72 Trioctahedral minerals in the soil-clays of north-east Scotland. By G. F. WALKER, B.Sc., Ph.D. Macaulay Institute for Soil Research, Aberdeen. [Read at the meeting of the Clay Minerals Group, November
Copyright SOIL STRUCTURE and CLAY MINERALS
 SOIL STRUCTURE and CLAY MINERALS Soil Structure Structure of a soil may be defined as the mode of arrangement of soil grains relative to each other and the forces acting between them to hold them in their
SOIL STRUCTURE and CLAY MINERALS Soil Structure Structure of a soil may be defined as the mode of arrangement of soil grains relative to each other and the forces acting between them to hold them in their
Soil Colloidal Chemistry. Compiled and Edited by Dr. Syed Ismail, Marthwada Agril. University Parbhani,MS, India
 Soil Colloidal Chemistry Compiled and Edited by Dr. Syed Ismail, Marthwada Agril. University Parbhani,MS, India 1 The Colloidal Fraction Introduction What is a colloid? Why this is important in understanding
Soil Colloidal Chemistry Compiled and Edited by Dr. Syed Ismail, Marthwada Agril. University Parbhani,MS, India 1 The Colloidal Fraction Introduction What is a colloid? Why this is important in understanding
Lecture 6. Physical Properties. Solid Phase. Particle Composition
 Lecture 6 Physical Properties Solid Phase Particle Composition 1 Questions What are tetrahedrons and octahedrons? How do silica tetrahedra bonds affect mineral weathering? Difference between primary and
Lecture 6 Physical Properties Solid Phase Particle Composition 1 Questions What are tetrahedrons and octahedrons? How do silica tetrahedra bonds affect mineral weathering? Difference between primary and
GY-343 Petrology Petrographic Microscope Laboratory
 Introduction to the Petrographic Microscope In this laboratory you will be using the petrographic microscope to analyze thin sections of various types of igneous rocks. You will be assigned a thin section
Introduction to the Petrographic Microscope In this laboratory you will be using the petrographic microscope to analyze thin sections of various types of igneous rocks. You will be assigned a thin section
muscovite PART 4 SHEET SILICATES
 muscovite PART 4 SHEET SILICATES SHEET SILICATES = PHYLLOSILICATES Phyllon = leaf Large group of mineral including many common minerals: muscovite, biotite, serpentine, chlorite, talc, clay minerals Structure:
muscovite PART 4 SHEET SILICATES SHEET SILICATES = PHYLLOSILICATES Phyllon = leaf Large group of mineral including many common minerals: muscovite, biotite, serpentine, chlorite, talc, clay minerals Structure:
Soil Mechanics Prof. B.V.S. Viswanadham Department of Civil Engineering Indian Institute of Technology, Bombay Lecture 3
 Soil Mechanics Prof. B.V.S. Viswanadham Department of Civil Engineering Indian Institute of Technology, Bombay Lecture 3 In the previous lecture we have studied about definitions of volumetric ratios and
Soil Mechanics Prof. B.V.S. Viswanadham Department of Civil Engineering Indian Institute of Technology, Bombay Lecture 3 In the previous lecture we have studied about definitions of volumetric ratios and
ANOMALIES IN TILE ETHYLENE GLYCOL SOLVA- TION TECHNIQUE USED IN X-RAY DIFFRACTION * ABSTRACT
 ANOMALIES IN TILE ETHYLENE GLYCOL SOLVA- TION TECHNIQUE USED IN X-RAY DIFFRACTION * G. W. KUNZE Agricultural and lv[echanical College of Texas ABSTRACT X-ray diffraction results are presented to show that
ANOMALIES IN TILE ETHYLENE GLYCOL SOLVA- TION TECHNIQUE USED IN X-RAY DIFFRACTION * G. W. KUNZE Agricultural and lv[echanical College of Texas ABSTRACT X-ray diffraction results are presented to show that
Earth Materials II Review Optical Mineralogy and Igneous Minerals
 Earth Materials II Review Optical Mineralogy and Igneous Minerals Refractive Index and Angle of Refraction Refractive Index(R. I. ) = velocity of light in a vacuum velocity of light in a medium The refractive
Earth Materials II Review Optical Mineralogy and Igneous Minerals Refractive Index and Angle of Refraction Refractive Index(R. I. ) = velocity of light in a vacuum velocity of light in a medium The refractive
9/4/2015. Feldspars White, pink, variable Clays White perfect Quartz Colourless, white, red, None
 ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks Chapter 3.0: Weathering & soils Chapter 4.0: Geological
ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks Chapter 3.0: Weathering & soils Chapter 4.0: Geological
23/9/2013 ENGINEERING GEOLOGY. Chapter 2: Rock classification:
 ENGINEERING GEOLOGY Chapter 2: Rock classification: ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks
ENGINEERING GEOLOGY Chapter 2: Rock classification: ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks
Weathering and Erosion
 Weathering and Erosion Weathering the disintegration and decomposition of material at the surface Erosion the transportation of weathered material by water, wind, or ice Weathering Two kinds of weathering
Weathering and Erosion Weathering the disintegration and decomposition of material at the surface Erosion the transportation of weathered material by water, wind, or ice Weathering Two kinds of weathering
Minerals II: Physical Properties and Crystal Forms. From:
 Minerals II: Physical Properties and Crystal Forms From: http://webmineral.com/data/rhodochrosite.shtml The Physical Properties of Minerals Color Streak Luster Hardness External Crystal Form Cleavage The
Minerals II: Physical Properties and Crystal Forms From: http://webmineral.com/data/rhodochrosite.shtml The Physical Properties of Minerals Color Streak Luster Hardness External Crystal Form Cleavage The
Igneous petrology EOSC 321
 Igneous petrology EOSC 321 Laboratory 2: Determination of plagioclase composition. Mafic and intermediate plutonic rocks Learning Goals. After this Lab, you should be able: Determine plagioclase composition
Igneous petrology EOSC 321 Laboratory 2: Determination of plagioclase composition. Mafic and intermediate plutonic rocks Learning Goals. After this Lab, you should be able: Determine plagioclase composition
Biaxial Minerals This document last updated on 27-Oct-2014
 1 of 18 10/27/2014 1:10 PM EENS 2110 Tulane University Biaxial Minerals Mineralogy Prof. Stephen A. Nelson This document last updated on 27-Oct-2014 All minerals that crystallize in the orthorhombic, monoclinic,
1 of 18 10/27/2014 1:10 PM EENS 2110 Tulane University Biaxial Minerals Mineralogy Prof. Stephen A. Nelson This document last updated on 27-Oct-2014 All minerals that crystallize in the orthorhombic, monoclinic,
LAB 2: SILICATE MINERALS
 GEOLOGY 640: Geology through Global Arts and Artifacts LAB 2: SILICATE MINERALS FRAMEWORK SILICATES The framework silicates quartz and feldspar are the most common minerals in Earth s crust. Quartz (SiO
GEOLOGY 640: Geology through Global Arts and Artifacts LAB 2: SILICATE MINERALS FRAMEWORK SILICATES The framework silicates quartz and feldspar are the most common minerals in Earth s crust. Quartz (SiO
Pyroxenes (Mg, Fe 2+ ) 2 Si 2 O 6 (monoclinic) and. MgSiO 3 FeSiO 3 (orthorhombic) Structure (Figure 2 of handout)
 Pyroxenes (Mg, Fe 2+ ) 2 Si 2 O 6 (monoclinic) and 20 MgSiO 3 FeSiO 3 (orthorhombic) Structure (Figure 2 of handout) Chain silicate eg Diopside Mg and Fe ions link SiO 3 chains The chain runs up and down
Pyroxenes (Mg, Fe 2+ ) 2 Si 2 O 6 (monoclinic) and 20 MgSiO 3 FeSiO 3 (orthorhombic) Structure (Figure 2 of handout) Chain silicate eg Diopside Mg and Fe ions link SiO 3 chains The chain runs up and down
THERMAL TRANSFORMATIONS OF PYROPHYLLITE AND TALC AS REVEALED BY X-RAY AND ELECTRON DIFFRACTION STUDIES*
 THERMAL TRANSFORMATIONS OF PYROPHYLLITE AND TALC AS REVEALED BY X-RAY AND ELECTRON DIFFRACTION STUDIES* by M. NAKAHIRA AND T. KATO Pennsylvania State University, University Park, Pa., U.S.A. and University
THERMAL TRANSFORMATIONS OF PYROPHYLLITE AND TALC AS REVEALED BY X-RAY AND ELECTRON DIFFRACTION STUDIES* by M. NAKAHIRA AND T. KATO Pennsylvania State University, University Park, Pa., U.S.A. and University
Ch. 4 - Clay Minerals, Rock Classification Page 1. Learning Objectives. Wednesday, January 26, 2011
 Ch. 4 - Clay Minerals, Rock Classification Page 1 Learning Objectives Ch. 4 - Clay Minerals, Rock Classification Page 2 Symbols Ch. 4 - Clay Minerals, Rock Classification Page 3 Clay Minerals and Structure
Ch. 4 - Clay Minerals, Rock Classification Page 1 Learning Objectives Ch. 4 - Clay Minerals, Rock Classification Page 2 Symbols Ch. 4 - Clay Minerals, Rock Classification Page 3 Clay Minerals and Structure
EESC 4701: Igneous and Metamorphic Petrology IGNEOUS MINERALS LAB 1 HANDOUT
 EESC 4701: Igneous and Metamorphic Petrology IGNEOUS MINERALS LAB 1 HANDOUT Sources: Cornell EAS302 lab, UMass Lowell 89.301 Mineralogy, LHRIC.org The Petrographic Microscope As you know, light is an electromagnetic
EESC 4701: Igneous and Metamorphic Petrology IGNEOUS MINERALS LAB 1 HANDOUT Sources: Cornell EAS302 lab, UMass Lowell 89.301 Mineralogy, LHRIC.org The Petrographic Microscope As you know, light is an electromagnetic
Why study Weathering?
 Why study Weathering? Weathering process of disintegrating solid rock & producing loose debris To understand geol process (like hydrologic systems) and how landscapes evolve (topo maps, landforms) Weathering
Why study Weathering? Weathering process of disintegrating solid rock & producing loose debris To understand geol process (like hydrologic systems) and how landscapes evolve (topo maps, landforms) Weathering
Report on samples from the Great Basin Science Sample and Records Library
 Jonathan G. Price, Ph.D. State Geologist and Director Nevada Bureau of Mines and Geology Office telephone: 775-784-6691 extension 5 1664 North Virginia Street Home telephone: 775-329-8011 University of
Jonathan G. Price, Ph.D. State Geologist and Director Nevada Bureau of Mines and Geology Office telephone: 775-784-6691 extension 5 1664 North Virginia Street Home telephone: 775-329-8011 University of
Objectives of this Lab. Introduction. The Petrographic Microscope
 Geological Sciences 101 Lab #9 Introduction to Petrology Objectives of this Lab 1. Understand how the minerals and textures of rocks reflect the processes by which they were formed. 2. Understand how rocks
Geological Sciences 101 Lab #9 Introduction to Petrology Objectives of this Lab 1. Understand how the minerals and textures of rocks reflect the processes by which they were formed. 2. Understand how rocks
The effect of grinding on micas. I. Muscovite.
 178 I The effect of grinding on micas. I. Muscovite. By ROBERT C. MACKENZIE, B.Sc., Ph.D., A.R.I.C. and ANGELA A. MILNE, B.Sc., Ph.D. The Macaulay Institute for Soil Research, Craigiebuckler, Aberdeen.
178 I The effect of grinding on micas. I. Muscovite. By ROBERT C. MACKENZIE, B.Sc., Ph.D., A.R.I.C. and ANGELA A. MILNE, B.Sc., Ph.D. The Macaulay Institute for Soil Research, Craigiebuckler, Aberdeen.
Atoms, Molecules and Minerals
 Atoms, Molecules and Minerals Atoms Matter The smallest unit of an element that retain its properties Molecules - a small orderly group of atoms that possess specific properties - H 2 O Small nucleus surrounded
Atoms, Molecules and Minerals Atoms Matter The smallest unit of an element that retain its properties Molecules - a small orderly group of atoms that possess specific properties - H 2 O Small nucleus surrounded
HETEROGENEITY IN MONTMORILLONITE. JAMES L. MCATEE, JR. Baroid Division, National Lead Co., Houston, Texas
 HETEROGENEITY IN MONTMORILLONITE By JAMES L. MCATEE, JR. Baroid Division, National Lead Co., Houston, Texas ABSTRACT X-ray diffraction patterns and cation-exchange data are presented for centrifuged Wyoming
HETEROGENEITY IN MONTMORILLONITE By JAMES L. MCATEE, JR. Baroid Division, National Lead Co., Houston, Texas ABSTRACT X-ray diffraction patterns and cation-exchange data are presented for centrifuged Wyoming
THE DECOMPOSITION PRODUCTS OF ANORTHITE ATTACKED BY PURE WATER AT ELEVATED TEMPERA- TURES AND PRESSURE
 THE DECOMPOSITION PRODUCTS OF ANORTHITE ATTACKED BY PURE WATER AT ELEVATED TEMPERA- TURES AND PRESSURE By A. F. FREDERICKSON AND J. E. Cox, JR., Washington University, St. Louis, Mo. ABSTRACT The behavior
THE DECOMPOSITION PRODUCTS OF ANORTHITE ATTACKED BY PURE WATER AT ELEVATED TEMPERA- TURES AND PRESSURE By A. F. FREDERICKSON AND J. E. Cox, JR., Washington University, St. Louis, Mo. ABSTRACT The behavior
Sedimentary Geology. Strat and Sed, Ch. 1 1
 Sedimentary Geology Strat and Sed, Ch. 1 1 Sedimentology vs. Stratigraphy Sedimentology is the study of the origin and classification of sediments and sedimentary rocks Mostly the physical and chemical
Sedimentary Geology Strat and Sed, Ch. 1 1 Sedimentology vs. Stratigraphy Sedimentology is the study of the origin and classification of sediments and sedimentary rocks Mostly the physical and chemical
Weathering and Clast Production Geol 113
 Weathering and Clast Production Geol 113 Goals: To understand the production of clasts from the weathering of rocks. Materials: rock and sediment samples from the two localities, sediment grain size chart,
Weathering and Clast Production Geol 113 Goals: To understand the production of clasts from the weathering of rocks. Materials: rock and sediment samples from the two localities, sediment grain size chart,
Scanning Electron Microscopic Study of Formation. of Gibbsite From Plagioclase
 Papers of the Institute for Thermal Spring Research. Okavarna University, No. 45, pp. 1l~24 (976) Scanning Electron Microscopic Study of Formation of Gibbsite From Plagioclase --The clay minerals in the
Papers of the Institute for Thermal Spring Research. Okavarna University, No. 45, pp. 1l~24 (976) Scanning Electron Microscopic Study of Formation of Gibbsite From Plagioclase --The clay minerals in the
This work follows the international standard nomenclature (IUGS) in naming the
 CHAPTER FIVE: PETROGRAPHY This work follows the international standard nomenclature (IUGS) in naming the different Platreef rock types. It should be noted that new lithologies not described in chapter
CHAPTER FIVE: PETROGRAPHY This work follows the international standard nomenclature (IUGS) in naming the different Platreef rock types. It should be noted that new lithologies not described in chapter
Name Petrology Spring 2006
 Igneous rocks lab Part I Due Tuesday 3/7 Igneous rock classification and textures For each of the rocks below, describe the texture, determine whether the rock is plutonic or volcanic, and describe its
Igneous rocks lab Part I Due Tuesday 3/7 Igneous rock classification and textures For each of the rocks below, describe the texture, determine whether the rock is plutonic or volcanic, and describe its
Clay Science 9, (1996)
 Clay Science 9, 335-345 (1996) ALTERATION OF MICA AND CHLORITE IN PADDY SOILS DERIVED FROM TRIASSIC AND JURASSIC SEDIMENTS YASUO KITAGAWA and KATSUHIKO ITAMI Fukui Prefectural University, Matsuoka, Fukui
Clay Science 9, 335-345 (1996) ALTERATION OF MICA AND CHLORITE IN PADDY SOILS DERIVED FROM TRIASSIC AND JURASSIC SEDIMENTS YASUO KITAGAWA and KATSUHIKO ITAMI Fukui Prefectural University, Matsuoka, Fukui
WHY DOES HALLOYSITE ROLL?--A NEW MODEL
 Clays and Clay Minerals, Vol. 44, No. 2, 191 196, 1996. WHY DOES HALLOYSITE ROLL?--A NEW MODEL BALBIR SINGH Centre for Microscopy and Microanalysis, The University of Queensland Brisbane, Qld. 4072, Australia
Clays and Clay Minerals, Vol. 44, No. 2, 191 196, 1996. WHY DOES HALLOYSITE ROLL?--A NEW MODEL BALBIR SINGH Centre for Microscopy and Microanalysis, The University of Queensland Brisbane, Qld. 4072, Australia
The Nucleus. Protons. Positive electrical charge The number of protons in the nucleus determines the atomic number
 Matter Atoms The smallest unit of an element that retain its properties Small nucleus surrounded by a cloud of electrons The nucleus contains protons and neutrons The Nucleus Protons Positive electrical
Matter Atoms The smallest unit of an element that retain its properties Small nucleus surrounded by a cloud of electrons The nucleus contains protons and neutrons The Nucleus Protons Positive electrical
The Lithosphere. Definition
 10/14/2014 www.komar.de The Lithosphere Ben Sullivan, Assistant Professor NRES 765, Biogeochemistry October 14th, 2014 Contact: bsullivan@cabnr.unr.edu Definition io9.com tedquarters.net Lithos = rocky;
10/14/2014 www.komar.de The Lithosphere Ben Sullivan, Assistant Professor NRES 765, Biogeochemistry October 14th, 2014 Contact: bsullivan@cabnr.unr.edu Definition io9.com tedquarters.net Lithos = rocky;
Mineral Characterization and Crystalline Nature of Quartz in Ponnaiyar River Sediments, Tamilnadu, India
 American-Eurasian Journal of Scientific Research 4 (2): 03-07 2009 ISSN 88-6785 IDOSI Publications 2009 Mineral Characterization and Crystalline Nature of Quartz in Ponnaiyar River Sediments Tamilnadu
American-Eurasian Journal of Scientific Research 4 (2): 03-07 2009 ISSN 88-6785 IDOSI Publications 2009 Mineral Characterization and Crystalline Nature of Quartz in Ponnaiyar River Sediments Tamilnadu
ELECTRON SPIN RESONANCE STUDIES OF MONTMORILLONITES
 Clay Minerals (1985) 20, 281-290 ELECTRON SPIN RESONANCE STUDIES OF MONTMORILLONITES C. CRACIUN AND AURELIA MEGHEA* Institutul de Cercethri pentru Pedologie ~i A grochimie and *Institutul Politeehnie Bueure~ti,
Clay Minerals (1985) 20, 281-290 ELECTRON SPIN RESONANCE STUDIES OF MONTMORILLONITES C. CRACIUN AND AURELIA MEGHEA* Institutul de Cercethri pentru Pedologie ~i A grochimie and *Institutul Politeehnie Bueure~ti,
Tikrit University. College of Engineering Civil engineering Department SOIL PROPERTES. Soil Mechanics. 3 rd Class Lecture notes Up Copyrights 2016
 Tikrit University SOIL PROPERTES College of Engineering Civil engineering Department Soil Mechanics 3 rd Class Lecture notes Up Copyrights 2016 1-Soil Composition -Solids -Water -Air 2-Soil Phases -Dry
Tikrit University SOIL PROPERTES College of Engineering Civil engineering Department Soil Mechanics 3 rd Class Lecture notes Up Copyrights 2016 1-Soil Composition -Solids -Water -Air 2-Soil Phases -Dry
GROUNDWATER GEOCHEMISTRY AND ASSOCIATED HARDPANS IN SOUTHWESTERN AUSTRALIA
 254 GROUNDWATER GEOCHEMISTRY AND ASSOCIATED HARDPANS IN SOUTHWESTERN AUSTRALIA Sam Lee CRC LEME, Department of Applied Geology, Curtin University of Technology, Bentley, Western Australia, 6485 INTRODUCTION
254 GROUNDWATER GEOCHEMISTRY AND ASSOCIATED HARDPANS IN SOUTHWESTERN AUSTRALIA Sam Lee CRC LEME, Department of Applied Geology, Curtin University of Technology, Bentley, Western Australia, 6485 INTRODUCTION
Sedimentation & Stratigraphy. Lab 3: Heavy Mineral Analysis Using the Franz Magnetic Separator
 Sedimentation & Stratigraphy Name I. Introduction Lab 3: Heavy Mineral Analysis Using the Franz Magnetic Separator In Lab 2, we used sand size-distributions to determine if the sediments were derived from
Sedimentation & Stratigraphy Name I. Introduction Lab 3: Heavy Mineral Analysis Using the Franz Magnetic Separator In Lab 2, we used sand size-distributions to determine if the sediments were derived from
THE FORMATION OF IDDINGSITE. A. B. Eowanls, Uniaersity of M elbourne, Auslralia.
 THE FORMATION OF IDDINGSITE A. B. Eowanls, Uniaersity of M elbourne, Auslralia. h.rrnooucrton fn their comprehensive paper on the origin, occurrence, composition and physical properties of the mineral
THE FORMATION OF IDDINGSITE A. B. Eowanls, Uniaersity of M elbourne, Auslralia. h.rrnooucrton fn their comprehensive paper on the origin, occurrence, composition and physical properties of the mineral
OLIVINES, PYROXENES, AND AMPHIBOLES PLEOCHROISM, INTERFERENCE COLORS AND EXTINCTION ANGLES
 GLY 4200C Lab Exercise 12 Pleochroism OLIVINES, PYROXENES, AND AMPHIBOLES PLEOCHROISM, INTERFERENCE COLORS AND EXTINCTION ANGLES When minerals are viewed under PP they may show color. Many minerals are
GLY 4200C Lab Exercise 12 Pleochroism OLIVINES, PYROXENES, AND AMPHIBOLES PLEOCHROISM, INTERFERENCE COLORS AND EXTINCTION ANGLES When minerals are viewed under PP they may show color. Many minerals are
Mechanical Weathering
 Weathering is the disintegration and decomposition of material at or near the surface. Erosion is the incorporation and transportation of material by a mobile agent, usually water, wind, or ice. Geologists
Weathering is the disintegration and decomposition of material at or near the surface. Erosion is the incorporation and transportation of material by a mobile agent, usually water, wind, or ice. Geologists
CLAY MINERAL DISTRIBUTION IN THE HIAWATHA SANDY SOILS OF NORTHERN WISCONSIN 1
 CLAY MINERAL DISTRIBUTION IN THE HIAWATHA SANDY SOILS OF NORTHERN WISCONSIN 1 By B. E. BROWN AND M. L. JACKSON Department of Soils, University of Wisconsin, Madison ABSTRACT Mineralogical analyses were
CLAY MINERAL DISTRIBUTION IN THE HIAWATHA SANDY SOILS OF NORTHERN WISCONSIN 1 By B. E. BROWN AND M. L. JACKSON Department of Soils, University of Wisconsin, Madison ABSTRACT Mineralogical analyses were
Chapter 6. Weathering, Erosion, and Soil
 Chapter 6 Weathering, Erosion, and Soil Introduction Rocks and minerals disintegrate and decompose by the processes of physical and chemical weathering. This breakdown occurs because the parent material
Chapter 6 Weathering, Erosion, and Soil Introduction Rocks and minerals disintegrate and decompose by the processes of physical and chemical weathering. This breakdown occurs because the parent material
GEOL Lab 11 (Metamorphic Rocks in Hand Sample and Thin Section)
 GEOL 333 - Lab 11 (Metamorphic Rocks in Hand Sample and Thin Section) Introduction - Metamorphic rock forms from any pre-existing rock that undergoes changes due to intense heat and pressure without melting.
GEOL 333 - Lab 11 (Metamorphic Rocks in Hand Sample and Thin Section) Introduction - Metamorphic rock forms from any pre-existing rock that undergoes changes due to intense heat and pressure without melting.
Igneous petrology EOSC 321 Laboratory 1: Ultramafic plutonic and volcanic rocks
 1 Igneous petrology EOSC 321 Laboratory 1: Ultramafic plutonic and volcanic rocks Material Needed: a) Microscope, b) Glossary of rock names and textures (see Pages 24-25 and 43 of Winter); c) Lab1 Manual
1 Igneous petrology EOSC 321 Laboratory 1: Ultramafic plutonic and volcanic rocks Material Needed: a) Microscope, b) Glossary of rock names and textures (see Pages 24-25 and 43 of Winter); c) Lab1 Manual
CLAY MINERAL FORMATION IN SEA WATER BY SUBMARINE WEATHERING OF K-FELDSPAR by
 CLAY MINERAL FORMATION IN SEA WATER BY SUBMARINE WEATHERING OF K-FELDSPAR by R. W. REx and B. D. MART~* Chevron Research Company, La Habra, California and U,S. Navy Oceanographic Office, Washington, D.C.
CLAY MINERAL FORMATION IN SEA WATER BY SUBMARINE WEATHERING OF K-FELDSPAR by R. W. REx and B. D. MART~* Chevron Research Company, La Habra, California and U,S. Navy Oceanographic Office, Washington, D.C.
CHAPTER 3.3: METAMORPHIC ROCKS
 CHAPTER 3.3: METAMORPHIC ROCKS Introduction Metamorphism - the process of changes in texture and mineralogy of pre-existing rock due to changes in temperature and/or pressure. Metamorphic means change
CHAPTER 3.3: METAMORPHIC ROCKS Introduction Metamorphism - the process of changes in texture and mineralogy of pre-existing rock due to changes in temperature and/or pressure. Metamorphic means change
Student Name: College: Grade:
 Student Name: College: Grade: Physical Geology Laboratory IGNEOUS MINERALS AND ROCKS IDENTIFICATION - INTRODUCTION & PURPOSE: In this lab you will learn to identify igneous rocks in hand samples from their
Student Name: College: Grade: Physical Geology Laboratory IGNEOUS MINERALS AND ROCKS IDENTIFICATION - INTRODUCTION & PURPOSE: In this lab you will learn to identify igneous rocks in hand samples from their
 26. MIXED-LAYER ILLITE/MONTMORILLONITE CLAYS FROM SITES 146 AND 149 Herman E. Roberson, State University of New York, Binghamton, New York INTRODUCTION The purpose of this report is to describe the clay
26. MIXED-LAYER ILLITE/MONTMORILLONITE CLAYS FROM SITES 146 AND 149 Herman E. Roberson, State University of New York, Binghamton, New York INTRODUCTION The purpose of this report is to describe the clay
Earth: An Introduction to Physical Geology Weathering and Soil
 Chapter 6 Lecture Earth: An Introduction to Physical Geology Eleventh Edition Weathering and Soil Tarbuck and Lutgens Weathering Weathering involves the physical breakdown and chemical alteration of rock
Chapter 6 Lecture Earth: An Introduction to Physical Geology Eleventh Edition Weathering and Soil Tarbuck and Lutgens Weathering Weathering involves the physical breakdown and chemical alteration of rock
A few more details on clays, Soil Colloids and their properties. What expandable clays do to surface area. Smectite. Kaolinite.
 A few more details on clays, Soil Colloids and their properties What expandable clays do to surface area Kaolinite Smectite Size 0.5-5 µm External surface 10-30 m 2 /g Internal surface - Size 0.1-1 µm
A few more details on clays, Soil Colloids and their properties What expandable clays do to surface area Kaolinite Smectite Size 0.5-5 µm External surface 10-30 m 2 /g Internal surface - Size 0.1-1 µm
Analysis of Clays and Soils by XRD
 Analysis of Clays and Soils by XRD I. Introduction Proper sample preparation is one of the most important requirements in the analysis of powder samples by X-ray diffraction (XRD). This statement is especially
Analysis of Clays and Soils by XRD I. Introduction Proper sample preparation is one of the most important requirements in the analysis of powder samples by X-ray diffraction (XRD). This statement is especially
Engineering Geology Prof. Debasis Roy Department of Civil Engineering Indian Institute of Technology, Kharagpur. Lecture - 13 Weathering
 Engineering Geology Prof. Debasis Roy Department of Civil Engineering Indian Institute of Technology, Kharagpur Lecture - 13 Weathering Hello everyone and welcome back. Today s lesson will be on weathering.
Engineering Geology Prof. Debasis Roy Department of Civil Engineering Indian Institute of Technology, Kharagpur Lecture - 13 Weathering Hello everyone and welcome back. Today s lesson will be on weathering.
Igneous petrology EOSC 321 Laboratory 8: Intermediate and Felsic Volcanic Rocks. Pyroclastic Rocks
 321 Lab 8 Instructor: L. Porritt - 1 - Igneous petrology EOSC 321 Laboratory 8: Intermediate and Felsic Volcanic Rocks. Pyroclastic Rocks Learning Goals. After this Lab, you should be able: Identify fine-grained
321 Lab 8 Instructor: L. Porritt - 1 - Igneous petrology EOSC 321 Laboratory 8: Intermediate and Felsic Volcanic Rocks. Pyroclastic Rocks Learning Goals. After this Lab, you should be able: Identify fine-grained
CHEMICAL, PHYSICAL, AND MINERALOGICAL PROPERTIES OF CERTAIN SOIL PROFILES IN THE LOWER MISSISSIPPI DELTA B. N. DRISKELL ABSTRACT
 CHEMICAL, PHYSICAL, AND MINERALOGICAL PROPERTIES OF CERTAIN SOIL PROFILES IN THE LOWER MISSISSIPPI DELTA B. N. DRISKELL Louisiana State University ABSTRACT The soils of the lower Mississippi Delta are
CHEMICAL, PHYSICAL, AND MINERALOGICAL PROPERTIES OF CERTAIN SOIL PROFILES IN THE LOWER MISSISSIPPI DELTA B. N. DRISKELL Louisiana State University ABSTRACT The soils of the lower Mississippi Delta are
Review - Unit 2 - Rocks and Minerals
 Review - Unit 2 - Rocks and Minerals Base your answers to questions 1 and 2 on the diagram below, which shows the results of three different physical tests, A, B, and C, that were performed on a mineral.
Review - Unit 2 - Rocks and Minerals Base your answers to questions 1 and 2 on the diagram below, which shows the results of three different physical tests, A, B, and C, that were performed on a mineral.
WEATHERING. Turning Rock to Sediment and Solutions 10/22/2012
 WEATHERING Turning Rock to Sediment and Solutions Igneous rocks form at high temperatures; at the Earth s surface they are chemically unstable and will begin to disintegrate and decompose in a process
WEATHERING Turning Rock to Sediment and Solutions Igneous rocks form at high temperatures; at the Earth s surface they are chemically unstable and will begin to disintegrate and decompose in a process
ESS 439 Igneous Petrology/Optical Mineralogy
 1 Lab # 3: Biaxial minerals ESS 439 Igneous Petrology/Optical Mineralogy In this laboratory session we will introduce the concepts of the Biaxial Indicatrix, Optic Axes, Vibration Directions and Ray Paths,
1 Lab # 3: Biaxial minerals ESS 439 Igneous Petrology/Optical Mineralogy In this laboratory session we will introduce the concepts of the Biaxial Indicatrix, Optic Axes, Vibration Directions and Ray Paths,
Feldspars. Structure. The feldspars are by far the most abundant group of minerals and are found in igneous, metamorphic and many sedimentary rocks.
 Feldspars The feldspars are by far the most abundant group of minerals and are found in igneous, metamorphic and many sedimentary rocks. Structure Felsdpars are framework silicates where each silica tetrahedra
Feldspars The feldspars are by far the most abundant group of minerals and are found in igneous, metamorphic and many sedimentary rocks. Structure Felsdpars are framework silicates where each silica tetrahedra
Mechanism of formation of Jbrsterite and enstatite from serpentine
 189 Mechanism of formation of Jbrsterite and enstatite from serpentine By G. W. BRNDLEY, M.Sc., Ph.D., and RYozo HAYAM, M.S. Materials Research Laboratory, The Pennsylvania State University, University
189 Mechanism of formation of Jbrsterite and enstatite from serpentine By G. W. BRNDLEY, M.Sc., Ph.D., and RYozo HAYAM, M.S. Materials Research Laboratory, The Pennsylvania State University, University
Geol 5310 (Spr 09) Lab 1 Review of Optical Mineralogy (9/9/09) Due Date: Wed., September 16.
 Geol 5310 (Spr 09) Lab 1 Review of Optical Mineralogy (9/9/09) Due Date: Wed., September 16. Name_ Score /36 Objective: Re-familiarize yourself with the optical properties of minerals and concepts like,
Geol 5310 (Spr 09) Lab 1 Review of Optical Mineralogy (9/9/09) Due Date: Wed., September 16. Name_ Score /36 Objective: Re-familiarize yourself with the optical properties of minerals and concepts like,
Uniaxial Minerals Descriptions
 Uniaxial Minerals Descriptions Look at 6 uniaxial minerals Quartz Nepheline Calcite Apatite Tourmaline Zircon Examine composition, relief, colour, form, cleavage, twinning, birefringence, occurrence Quartz
Uniaxial Minerals Descriptions Look at 6 uniaxial minerals Quartz Nepheline Calcite Apatite Tourmaline Zircon Examine composition, relief, colour, form, cleavage, twinning, birefringence, occurrence Quartz
GEOLOGY 333 LAB 5. Light Mechanics
 GEOLOGY 333 LAB 5 OPTICAL MICROSCOPY & MINERALS IN THIN SECTION Light Mechanics Light Waves: Visible light travels in waves, which have measurable wavelengths, frequencies, and velocities Wavelength (
GEOLOGY 333 LAB 5 OPTICAL MICROSCOPY & MINERALS IN THIN SECTION Light Mechanics Light Waves: Visible light travels in waves, which have measurable wavelengths, frequencies, and velocities Wavelength (
Silicates. The most common group of minerals forming the silicate Earth
 Silicates The most common group of minerals forming the silicate Earth 25% of all minerals (~1000) 40% of rock forming minerals 90% of earth s crust i.e those minerals you are likely to find ~100 of earth
Silicates The most common group of minerals forming the silicate Earth 25% of all minerals (~1000) 40% of rock forming minerals 90% of earth s crust i.e those minerals you are likely to find ~100 of earth
NOTE TOSUDITE CRYSTALLIZATION IN THE KAOLINIZED GRANITIC CUPOLA OF MONTEBRAS, CREUSE, FRANCE
 Clay Minerals (1986) 21, 225-230 225 NOTE TOSUDITE CRYSTALLIZATION IN THE KAOLINIZED GRANITIC CUPOLA OF MONTEBRAS, CREUSE, FRANCE Albite, muscovite granite and greisens of the Montebras cupola, Creuse,
Clay Minerals (1986) 21, 225-230 225 NOTE TOSUDITE CRYSTALLIZATION IN THE KAOLINIZED GRANITIC CUPOLA OF MONTEBRAS, CREUSE, FRANCE Albite, muscovite granite and greisens of the Montebras cupola, Creuse,
EESC 4701: Igneous and Metamorphic Petrology METAMORPHIC ROCKS LAB 8 HANDOUT
 Sources: Caltech, Cornell, UCSC, TAMIU Introduction EESC 4701: Igneous and Metamorphic Petrology METAMORPHIC ROCKS LAB 8 HANDOUT Metamorphism is the process by which physical and chemical changes in a
Sources: Caltech, Cornell, UCSC, TAMIU Introduction EESC 4701: Igneous and Metamorphic Petrology METAMORPHIC ROCKS LAB 8 HANDOUT Metamorphism is the process by which physical and chemical changes in a
CERAMIC MATERIALS I. Asst. Prof. Dr. Ayşe KALEMTAŞ
 CERAMIC MATERIALS I akalemtas@mu.edu.tr, akalemtas@gmail.com, Phone: 211 19 17 Metallurgical and Materials Engineering Department Traditional Ceramics Clay products Main Components Clay Feldspar Silica
CERAMIC MATERIALS I akalemtas@mu.edu.tr, akalemtas@gmail.com, Phone: 211 19 17 Metallurgical and Materials Engineering Department Traditional Ceramics Clay products Main Components Clay Feldspar Silica
MICROSTRUCTURAL DEVELOPMENT IN FIRE DAMAGED CONCRETE - A CASE STUDY
 MICROSTRUCTURAL DEVELOPMENT IN FIRE DAMAGED CONCRETE - A CASE STUDY V P Chatterjee, S K Chaturvedi, S K Gupta, M Kumar and P S Sharma National Council for Cement and Building Materials, Ballabgarh, Haryana,
MICROSTRUCTURAL DEVELOPMENT IN FIRE DAMAGED CONCRETE - A CASE STUDY V P Chatterjee, S K Chaturvedi, S K Gupta, M Kumar and P S Sharma National Council for Cement and Building Materials, Ballabgarh, Haryana,
NATURE AND ORIGIN OF AN ALUMINOUS VERMICULITIC WEATHERING PRODUCT IN ACID SOILS FROM UPLAND CATCHMENTS IN SCOTLAND
 Clay Minerals (1990) 25, 467-475 NATURE AND ORIGIN OF AN ALUMINOUS VERMICULITIC WEATHERING PRODUCT IN ACID SOILS FROM UPLAND CATCHMENTS IN SCOTLAND D. C. BAIN, A. MELLOR* AND M. J. WILSON The Macaulay
Clay Minerals (1990) 25, 467-475 NATURE AND ORIGIN OF AN ALUMINOUS VERMICULITIC WEATHERING PRODUCT IN ACID SOILS FROM UPLAND CATCHMENTS IN SCOTLAND D. C. BAIN, A. MELLOR* AND M. J. WILSON The Macaulay
Part 1 Soil Its Nature and Origin
 Part 1 Soil Its Nature and Origin Soil is essentially a natural body of mineral and organic constituents produced by solid material recycling during a myriad of complex processes of solid crust modifications,
Part 1 Soil Its Nature and Origin Soil is essentially a natural body of mineral and organic constituents produced by solid material recycling during a myriad of complex processes of solid crust modifications,
2. Which mineral is white or colorless, has a hardness of 2.5, and splits with cubic cleavage? 1. calcite 3. pyrite 2. halite 4.
 RM2 JRQ Mr. Chase Name Date: 1. Base your answer on the information, table, and photographs (see image) and on your knowledge of Earth science. "Herkimer Diamonds" Gem-quality "Herkimer Diamonds" are hexagonal-shaped
RM2 JRQ Mr. Chase Name Date: 1. Base your answer on the information, table, and photographs (see image) and on your knowledge of Earth science. "Herkimer Diamonds" Gem-quality "Herkimer Diamonds" are hexagonal-shaped
CHLORITIZED WEATHERING PRODUCTS OF A NEW ENGLAND GLACIAL TILL
 CHLORITIZED WEATHERING PRODUCTS OF A NEW ENGLAND GLACIAL TILL R. M. QUIGLEY~ AND R. T. MARTIN Soil Engineering Division, Massachusetts Institute of Technology, Cambridge, Massachusetts ABSTRACT The clay
CHLORITIZED WEATHERING PRODUCTS OF A NEW ENGLAND GLACIAL TILL R. M. QUIGLEY~ AND R. T. MARTIN Soil Engineering Division, Massachusetts Institute of Technology, Cambridge, Massachusetts ABSTRACT The clay
J. PETROVIČ. Institute of Inorganic Chemistry, Slovak Academy of Sciences, Bratislava 9. Received March 13, 1969
 Isomorphous Substitution of Aluminium for Silicon in Tobermoritic Structure. II. The Mixtures Prepared from Different Starting Materials and from Gels Containing Aluminium Ion J. PETROVIČ Institute of
Isomorphous Substitution of Aluminium for Silicon in Tobermoritic Structure. II. The Mixtures Prepared from Different Starting Materials and from Gels Containing Aluminium Ion J. PETROVIČ Institute of
Optical Mineralogy in a Nutshell
 Optical Mineralogy in a Nutshell Use of the petrographic microscope Slides borrowed/adapted from Jane Selverstone (University of New Mexico) and John Winter (Whitman College) Why use the petrographic microscope?
Optical Mineralogy in a Nutshell Use of the petrographic microscope Slides borrowed/adapted from Jane Selverstone (University of New Mexico) and John Winter (Whitman College) Why use the petrographic microscope?
Practice Questions for Lecture 5 Geology 1200
 Practice Questions for Lecture 5 Geology 1200 Use these questions to test your knowledge of Lecture5. The exams will be similar in format, except that they will deal with more than one chapter, and will
Practice Questions for Lecture 5 Geology 1200 Use these questions to test your knowledge of Lecture5. The exams will be similar in format, except that they will deal with more than one chapter, and will
ESS Minerals. Lee. 1. The table below shows some properties of four different minerals.
 Name: ESS Minerals Pd. 1. The table below shows some properties of four different minerals. The minerals listed in the table are varieties of which mineral? (A) garnet (B) magnetite (C) olivine (D) quartz
Name: ESS Minerals Pd. 1. The table below shows some properties of four different minerals. The minerals listed in the table are varieties of which mineral? (A) garnet (B) magnetite (C) olivine (D) quartz
Matter and Minerals. Earth 9 th edition Chapter 3 Minerals: summary in haiku form "Mineral" defined: natural, inorganic, solid (and two more).
 1 2 Matter and Minerals Earth 9 th edition Chapter 3 Minerals: summary in haiku form "Mineral" defined: natural, inorganic, solid (and two more). continued... 3 4 5 6 7 8 9 10 11 12 13 14 Also crystalline,
1 2 Matter and Minerals Earth 9 th edition Chapter 3 Minerals: summary in haiku form "Mineral" defined: natural, inorganic, solid (and two more). continued... 3 4 5 6 7 8 9 10 11 12 13 14 Also crystalline,
GY 302: Crystallography & Mineralogy
 UNIVERSITY OF SOUTH ALABAMA GY 302: Crystallography & Mineralogy Lecture 7b: Optical Mineralogy Instructor: Dr. Douglas Haywick Last Time 1. Properties of light 2. Minerals and light transmission Light
UNIVERSITY OF SOUTH ALABAMA GY 302: Crystallography & Mineralogy Lecture 7b: Optical Mineralogy Instructor: Dr. Douglas Haywick Last Time 1. Properties of light 2. Minerals and light transmission Light
Physical Geology 101 Laboratory MINERALS II Silicate and Carbonate Rock-Forming Minerals
 Student Name: College: Grade: Physical Geology 101 Laboratory MINERALS II Silicate and Carbonate Rock-Forming Minerals I. INTRODUCTION: The purpose of this lab is you will improve your mineral identification
Student Name: College: Grade: Physical Geology 101 Laboratory MINERALS II Silicate and Carbonate Rock-Forming Minerals I. INTRODUCTION: The purpose of this lab is you will improve your mineral identification
And the study of mineral the branch in geology is termed as mineralogy. (Refer Slide Time: 0:29)
 Earth Sciences for Civil Engineering Professor Javed N Malik Department of Earth Sciences Indian Institute of Technology Kanpur Module 2 Lecture No 6 Rock-Forming Minerals and their Properties (Part-2)
Earth Sciences for Civil Engineering Professor Javed N Malik Department of Earth Sciences Indian Institute of Technology Kanpur Module 2 Lecture No 6 Rock-Forming Minerals and their Properties (Part-2)
Last Time. GY 302: Crystallography & Mineralogy. Polymorphs & Polymorphism. Other Crystal Structures. Other Crystal Structures. This Week s Agenda
 UNIVERSITY OF SOUTH ALABAMA Last Time GY 302: Crystallography & Mineralogy 1. Polymorphs and Polymorphism 2. Pseudomorphs and other definitions 3. Crystal Habit Lecture 7: Optical Mineralogy (two day lecture?)
UNIVERSITY OF SOUTH ALABAMA Last Time GY 302: Crystallography & Mineralogy 1. Polymorphs and Polymorphism 2. Pseudomorphs and other definitions 3. Crystal Habit Lecture 7: Optical Mineralogy (two day lecture?)
Petrographic Investigation of Two Float Samples from the Goldstake Property, Northern Ontario. Prepared for: Mr. Robert Dillman
 Petrographic Investigation of Two Float Samples from the Goldstake Property, Northern Ontario Prepared for: Mr. Robert Dillman by Jim Renaud Renaud Geological Consulting Ltd. 21272 Denfield Road London,
Petrographic Investigation of Two Float Samples from the Goldstake Property, Northern Ontario Prepared for: Mr. Robert Dillman by Jim Renaud Renaud Geological Consulting Ltd. 21272 Denfield Road London,
X-RAY STUDIES OF THE ALTERATION OF SODA FELDSPAW ABSTRACT
 X-RAY STUDIES OF THE ALTERATION OF SODA FELDSPAW By G. W. BRINDLEY AND E. W. RADOSLOVICH Department of Ceramic Technology, The Pennsylvania State University, University Park, Pennsylvania ABSTRACT Studies
X-RAY STUDIES OF THE ALTERATION OF SODA FELDSPAW By G. W. BRINDLEY AND E. W. RADOSLOVICH Department of Ceramic Technology, The Pennsylvania State University, University Park, Pennsylvania ABSTRACT Studies
Wearing Down Landforms
 Wearing Down Landforms Denudation Denudation has also been called gradation. It is the Natural loss of soil and rock debris, blown away by wind or washed away by running water, laying bare the rock below.
Wearing Down Landforms Denudation Denudation has also been called gradation. It is the Natural loss of soil and rock debris, blown away by wind or washed away by running water, laying bare the rock below.
Chapter I Basic Characteristics of Soils
 Chapter I Basic Characteristics of Soils Outline 1. The Nature of Soils (section 1.1 Craig) 2. Soil Texture (section 1.1 Craig) 3. Grain Size and Grain Size Distribution (section 1.2 Craig) 4. Particle
Chapter I Basic Characteristics of Soils Outline 1. The Nature of Soils (section 1.1 Craig) 2. Soil Texture (section 1.1 Craig) 3. Grain Size and Grain Size Distribution (section 1.2 Craig) 4. Particle
Surname. Number OXFORD CAMBRIDGE AND RSA EXAMINATIONS ADVANCED SUBSIDIARY GCE F792 GEOLOGY. Rocks Processes and Products
 Candidate Forename Centre Number Candidate Surname Candidate Number OXFORD CAMBRIDGE AND RSA EXAMINATIONS ADVANCED SUBSIDIARY GCE F792 GEOLOGY Rocks Processes and Products WEDNESDAY 20 MAY 2009: Afternoon
Candidate Forename Centre Number Candidate Surname Candidate Number OXFORD CAMBRIDGE AND RSA EXAMINATIONS ADVANCED SUBSIDIARY GCE F792 GEOLOGY Rocks Processes and Products WEDNESDAY 20 MAY 2009: Afternoon
Matter and Minerals Earth: Chapter Pearson Education, Inc.
 Matter and Minerals Earth: Chapter 3 Minerals: Building Blocks of Rocks By definition a mineral is: Naturally occurring An inorganic solid Ordered internal molecular structure Definite chemical composition
Matter and Minerals Earth: Chapter 3 Minerals: Building Blocks of Rocks By definition a mineral is: Naturally occurring An inorganic solid Ordered internal molecular structure Definite chemical composition
THIS IS A NEW SPECIFICATION
 THIS IS A NEW SPECIFICATION ADVANCED SUBSIDIARY GCE GEOLOGY Rocks Processes and Products F792 * OCE / 11038 * Candidates answer on the question paper OCR Supplied Materials: None Other Materials Required:
THIS IS A NEW SPECIFICATION ADVANCED SUBSIDIARY GCE GEOLOGY Rocks Processes and Products F792 * OCE / 11038 * Candidates answer on the question paper OCR Supplied Materials: None Other Materials Required:
Igneous Rock Classification, Processes and Identification Physical Geology GEOL 100
 Igneous Rock Classification, Processes and Identification Physical Geology GEOL 100 Ray Rector - Instructor Major Concepts 1) Igneous rocks form directly from the crystallization of a magma or lava 2)
Igneous Rock Classification, Processes and Identification Physical Geology GEOL 100 Ray Rector - Instructor Major Concepts 1) Igneous rocks form directly from the crystallization of a magma or lava 2)
A Technique for Microscopical Soil Examinations
 A Technique for Microscopical Soil Examinations Skip Palenik Microtrace LLC Elgin, IL USA www.microtracescientific.com Edmond Locard Preliminary Separation Color (dry) Sieving Low Power Microscopy Seeds,
A Technique for Microscopical Soil Examinations Skip Palenik Microtrace LLC Elgin, IL USA www.microtracescientific.com Edmond Locard Preliminary Separation Color (dry) Sieving Low Power Microscopy Seeds,
Igneous petrology EOSC 321
 Igneous petrology EOSC 321 Laboratory 1: Review of optical properties of minerals. Ultramafic plutonic and volcanic rocks Material Needed: a) Microscope, b) Glossary of rock names and textures (see Pages
Igneous petrology EOSC 321 Laboratory 1: Review of optical properties of minerals. Ultramafic plutonic and volcanic rocks Material Needed: a) Microscope, b) Glossary of rock names and textures (see Pages
Crust Elements. Elements of Earth. Minerals. Crystals. Interconnected Rocks and minerals Interior processes Erosion and deposition Water and air
 Emily and Megan Earth System Science Interconnected Rocks and minerals Interior processes Erosion and deposition Water and air Made of atoms Earth is mostly iron, by weight Elements of Earth Made of atoms
Emily and Megan Earth System Science Interconnected Rocks and minerals Interior processes Erosion and deposition Water and air Made of atoms Earth is mostly iron, by weight Elements of Earth Made of atoms
Weathering. Weathering: is the process which breaks rocks into smaller bits. A. Physical or mechanical weathering
 Weathering Weathering: is the process which breaks rocks into smaller bits. Types of Weathering: A. Physical or mechanical weathering B. Chemical weathering C. Biological weathering 1 Mechanical or Physical
Weathering Weathering: is the process which breaks rocks into smaller bits. Types of Weathering: A. Physical or mechanical weathering B. Chemical weathering C. Biological weathering 1 Mechanical or Physical
