POGIL EXERCISE 05 How Are Electrons Distributed Around the Nucleus of the Atom?
|
|
|
- Donald Cole
- 5 years ago
- Views:
Transcription
1 RUN TIME: 50 MIN POGIL 5 Page 1 of 6 POGIL EXERCISE 05 How Are Electrons Distributed Around the Nucleus of the Atom? Each member should locate his/her role in Table 1 and assume his or her role at this time. The new manager takes charge of the POGIL folder and hands out the GRF and RRF to the appropriate members. The new recorder should record the names of the group members on the new GRF and the Reflector should record the start time on her/his form. Table 1. Group Member Role Assignments GROUP TYPE -> GROUPS OF THREE GROUPS OF FOUR MEMBER NO. -> Manager + + Reporter + + Recorder + + Reflector + + Technician Encourager + + SFUC + * + * The manager should direct the members to use two minutes to read the following material. OBSERVATION I. Niels Bohr did some Nobel Prize thinking that was eventually substantiated experimentally about the question as to the location of electrons around the nucleus of an atom. As you already know from a previous POGIL, electrons cannot be viewed as maintaining a planer (flat) orbit around the nucleus. His conclusion was that electrons were arranged in shells around the nucleus. He referred to these shells as principal energy levels. Eventually, seven shells were found or inferred to exist in nature; they were numbered one through seven from near the nucleus (1) to the exterior (7). The shells also increase in energy from one to seven. The outer most shell coincides with the period number of the element in the periodic chart. This means that all elements in Period 1 have only one shell; all elements in period 2 have two shells; etc. You can visualize shells as spheres surrounding the nucleus. 1. What is the maximum number of shells known? 2. How many periods are there in the periodic chart? 3. What is your conclusion from the data of Items 1 and 2? +5 MIN
2 RUN TIME: 50 MIN POGIL 5 Page 2 of 6 4. How many principle energy levels does mercury (Hg) have? Give a reason for your answer. 5. Which element has the most energetic electrons, calcium (Ca) or Iodine (I)? Give a reason for your answer. OBSERVATION II. Electrons are arranged in shells, subshells and orbitals. The orbital is the basic unit structure of electron distribution; each orbital can hold a maximum of 2 electrons. The next level of organization is the subshell. There are four types of subshells designated by the letters: s, p, d, and f. Each subshell contains a specific number of orbitals: The s subshell contains a maximum of one orbital and therefore can hold a maximum of two electrons. The p subshell can contain up to three orbitals for a maximum of six electrons. The d subshell can contain up to five orbitals holding a maximum of ten electrons. Finally, the f subshell can have up to seven orbitals potentially holding 14 electrons. The s and p subshells of the outermost shell (the period number in which the element is located) are the electrons most often involved in chemical reactions. These electrons are called valence electrons and it is these electrons that are diagramed in Lewis diagrams. The other electrons in the d and f subshells are known as core electrons and are not diagramed in the Lewis diagram of the atom since they play a secondary role in the chemical reactivity of the element. The largest structure in electron configuration scheme is known as the shell which constitute the principal energy levels within the atom. Shells are numbered and this number is the period number in which the element falls on the periodic chart. 6. Draw a concept map in Figure I that describes the relationship between orbitals, subshells and shells of an atom (beginning with the smallest moving to the largest). When you re finished, submit your diagram to the instructor for validation. 7. Table 2 summarizes the known electronic configuration of atoms by telling you what type of subshells are present in each shell (period). Complete Table 3 using the information in Observation II and Table What group of elements has the same atomic number as the last cell of each row of Table 3? +25 MIN
3 RUN TIME: 50 MIN POGIL 5 Page 3 of 6 Figure I. Diagram of Electron Configuration Relationships Table 2. Electron Configuration of Atoms Principle PERIOD Contains these Subshells E. Level NUMBER s p d f 1 1 YES NO NO NO 2 2 YES YES NO NO 3 3 YES YES NO NO 4 4 YES YES YES NO 5 5 YES YES YES NO 6 6 YES YES YES YES 7 7 YES NO YES YES +25 MIN
4 RUN TIME: 50 MIN POGIL 5 Page 4 of 6 Table 3. Maximum Electron Capacity of All Known Elements. PERIOD (SHELL) Maximum Number of Electrons in MAXIMUM # E - IN NUMBER s Orbitals p Orbitals d Orbitals f Orbitals SHELL a ATOM b a Maximum number of electrons in shell is equal to sum of values in Columns 2-5. b Maximum number of electrons in Atom for Period 1 is equal to the number if electrons in the shell; for Period 2 the maximum number of electrons in the atom is equal the maximum number in Period 1 + the maximum number in Period 2; etc. Recorders should have the instructor to examine your completed Table 3. After approval, continue processing with Item Arrange the following terms in increasing order of the maximum number of electrons each can hold: orbital, shell, subshell. 10. What special name is given to the outer most s and p electrons? 11. What special name is given to all the other electrons of the atoms? 12. What is the total number of electrons that can be accommodated in s and p orbitals? OBSERVATION III. The electrons of an atom do not add randomly to the various orbitals, shells and subshells. Electrons occupy a certain orbital, subshell, and shell based on the energy they contain. You can determine the electronic configuration of any element by finding the element on the periodic chart (Figure II). 13. Using Figure II as modified by the mini-lecture provided by the instructor, write the Noble Gas configuration for the elements listed in Period 6 of the Periodic Chart by completing Table MIN
5 RUN TIME: 50 MIN POGIL 5 Page 5 of 6 Figure II. Electron Configuration Related to the Periodic Chart EXERCISE END. Managers should collect the GRF, the Recorders POGIL and the RRF, paperclip them together, and place in the left pocket of the folder. The folder should be closed and left on the table. +50 MIN
6 RUN TIME: 50 MIN POGIL 5 Page 6 of 6 Table 4. Electron Configuration and Lewis Diagrams of a Selection of Period 6 Elements. Element Symbol Atomic No. 55 Nobel Gas Electron Configuration Lewis Diagram Gd 64 [Xe] 54 6s 2 4f min
POGIL EXERCISE 04 Anatomy and Physiology of the Atom
 RUN TIME: 60 MIN POGIL 4 Page 1 of 8 POGIL EXERCISE 04 Anatomy and Physiology of the Atom Each member should assume his or her role at this time. The new manager takes charge of the POGIL folder and hands
RUN TIME: 60 MIN POGIL 4 Page 1 of 8 POGIL EXERCISE 04 Anatomy and Physiology of the Atom Each member should assume his or her role at this time. The new manager takes charge of the POGIL folder and hands
POGIL EXERCISE 15 Why and How Do Atoms React with Each Other?
 RUN TIME: 65 MIN POGIL 15 Page 1 of 8 POGIL EXERCISE 15 Why and How Do Atoms React with Each Other? Each member should assume his or her role at this time. The new manager takes charge of the POGIL folder
RUN TIME: 65 MIN POGIL 15 Page 1 of 8 POGIL EXERCISE 15 Why and How Do Atoms React with Each Other? Each member should assume his or her role at this time. The new manager takes charge of the POGIL folder
RUN TIME: 50 MIN POGIL 10 Page 1 of 5
 RUN TIME: 50 MIN POGIL 10 Page 1 of 5 POGIL EXERCISE 10 What Is a Mole and Why Do Chemists Need It? Each member should assume his or her role at this time. The new manager takes charge of the POGIL folder
RUN TIME: 50 MIN POGIL 10 Page 1 of 5 POGIL EXERCISE 10 What Is a Mole and Why Do Chemists Need It? Each member should assume his or her role at this time. The new manager takes charge of the POGIL folder
The Periodic Table & Electronic Structure
 Periodic Table The Periodic Table & Electronic Structure Is there an overall reason that the elements are arranged in the order that they are? Yes! We ve seen that the elements are arranged in order of
Periodic Table The Periodic Table & Electronic Structure Is there an overall reason that the elements are arranged in the order that they are? Yes! We ve seen that the elements are arranged in order of
POGIL EXERCISE 13 All about Oxidation States, Oxidation and Reduction
 +5 MIN RUN TIME: 85 MIN POGIL 13: Page 1 of 7 POGIL EXERCISE 13 All about Oxidation States, Oxidation and Reduction Each member should locate his/her role in Table 1 and assume his or her role at this
+5 MIN RUN TIME: 85 MIN POGIL 13: Page 1 of 7 POGIL EXERCISE 13 All about Oxidation States, Oxidation and Reduction Each member should locate his/her role in Table 1 and assume his or her role at this
UNIT 2 PART 1: ELECTRONS
 UNIT 2 PART 1: ELECTRONS Electrons in an Atom Bohr s Model: Electrons resided in an allowed orbit. Quantum Mechanics Model: Probability of finding an electron in an area around the nucleus. This area around
UNIT 2 PART 1: ELECTRONS Electrons in an Atom Bohr s Model: Electrons resided in an allowed orbit. Quantum Mechanics Model: Probability of finding an electron in an area around the nucleus. This area around
Electron Configurations: Assigning each electron in an atom to the energy level and sublevel it occupies in the atom. Number of Electrons
 First some terms and more information about the structure of the atom: 1) Energy level is no longer an orbit but more like a boundary or maximum distance from the nucleus that electrons occupy. 1, 2, 3
First some terms and more information about the structure of the atom: 1) Energy level is no longer an orbit but more like a boundary or maximum distance from the nucleus that electrons occupy. 1, 2, 3
The Particle Theory of Matter
 Identifying Elements TEKS: 8.5B - identify that protons determine an element s identity and valence electrons determine its chemical properties, including reactivity. 8.5C interpret the arrangement of
Identifying Elements TEKS: 8.5B - identify that protons determine an element s identity and valence electrons determine its chemical properties, including reactivity. 8.5C interpret the arrangement of
Chapter 9. Blimps, Balloons, and Models for the Atom. Electrons in Atoms and the Periodic Table. Hindenburg. Properties of Elements Hydrogen Atoms
 Chapter 9 Electrons in Atoms and the Periodic Table Blimps, Balloons, and Models for the Atom Hindenburg Blimps, Balloons, and Models for the Atom Properties of Elements Hydrogen Atoms Helium Atoms 1 Blimps,
Chapter 9 Electrons in Atoms and the Periodic Table Blimps, Balloons, and Models for the Atom Hindenburg Blimps, Balloons, and Models for the Atom Properties of Elements Hydrogen Atoms Helium Atoms 1 Blimps,
Bohr Models are NOT Boring! How to Draw Bohr Diagrams
 Bohr Models are NOT Boring! How to Draw Bohr Diagrams 1) Find the element on the periodic table. 2)Determine the number of electrons--it is the same as the atomic number. 3)This is how many electrons you
Bohr Models are NOT Boring! How to Draw Bohr Diagrams 1) Find the element on the periodic table. 2)Determine the number of electrons--it is the same as the atomic number. 3)This is how many electrons you
LIMITATIONS OF RUTHERFORD S ATOMIC MODEL
 ELECTRONS IN ATOMS LIMITATIONS OF RUTHERFORD S ATOMIC MODEL Did not explain the chemical properties of atoms For example, it could not explain why metals or compounds of metals give off characteristic
ELECTRONS IN ATOMS LIMITATIONS OF RUTHERFORD S ATOMIC MODEL Did not explain the chemical properties of atoms For example, it could not explain why metals or compounds of metals give off characteristic
Chemistry Vocabulary. These vocabulary words appear on the Chemistry CBA in addition to being tested on the Chemistry Vocabulary Test.
 Chemistry Vocabulary These vocabulary words appear on the Chemistry CBA in addition to being tested on the Chemistry Vocabulary Test. atom the smallest unit of an element that still represents that element.
Chemistry Vocabulary These vocabulary words appear on the Chemistry CBA in addition to being tested on the Chemistry Vocabulary Test. atom the smallest unit of an element that still represents that element.
8.5C: Periodic Table
 Reflect Suppose you wanted to organize your locker at school. How could you separate and arrange everything in an organized way? You could place the books, notebooks, and folders on a shelf that is separate
Reflect Suppose you wanted to organize your locker at school. How could you separate and arrange everything in an organized way? You could place the books, notebooks, and folders on a shelf that is separate
POGIL EXERCISE 12 How Can You Predict Chemical Reactions?
 RUN TIME: 60 MIN POGIL 12 Page 1 of 7 POGIL EXERCISE 12 How Can You Predict Chemical Reactions? Each member should assume his or her role at this time. The new manager takes charge of the POGIL folder
RUN TIME: 60 MIN POGIL 12 Page 1 of 7 POGIL EXERCISE 12 How Can You Predict Chemical Reactions? Each member should assume his or her role at this time. The new manager takes charge of the POGIL folder
The Structure of the Atom
 The Structure of the Atom 1 The Atom as Matter Dalton s atomic theory had problems It considered atoms to be hard, indivisible particles Did not explain why atoms reacted Also did not explain why atoms
The Structure of the Atom 1 The Atom as Matter Dalton s atomic theory had problems It considered atoms to be hard, indivisible particles Did not explain why atoms reacted Also did not explain why atoms
Chem I - Wed, 9/16/15
 Chem I - Wed, 9/16/15 Do Now Complete the back of worksheet 4.5 Homework Pennium Lab if not Finished E- Config POGIL Agenda Return Papers Wks 4.5 Pennium Lab Electron Config Chapter 5 Quantum Theory and
Chem I - Wed, 9/16/15 Do Now Complete the back of worksheet 4.5 Homework Pennium Lab if not Finished E- Config POGIL Agenda Return Papers Wks 4.5 Pennium Lab Electron Config Chapter 5 Quantum Theory and
UNIT (2) ATOMS AND ELEMENTS
 UNIT (2) ATOMS AND ELEMENTS 2.1 Elements An element is a fundamental substance that cannot be broken down by chemical means into simpler substances. Each element is represented by an abbreviation called
UNIT (2) ATOMS AND ELEMENTS 2.1 Elements An element is a fundamental substance that cannot be broken down by chemical means into simpler substances. Each element is represented by an abbreviation called
UNIT 2 CHEMISTRY IN ACTION. Mr.Yeung
 UNIT 2 CHEMISTRY IN ACTION Mr.Yeung ATOMS REVIEW Check out the Scale of the Universe http://htwins.net/scale2/ Keep the following questions in mind: 1. Are cells bigger or smaller than atoms? 2. Generally
UNIT 2 CHEMISTRY IN ACTION Mr.Yeung ATOMS REVIEW Check out the Scale of the Universe http://htwins.net/scale2/ Keep the following questions in mind: 1. Are cells bigger or smaller than atoms? 2. Generally
Use the Venn Diagram to compare and contrast the Bohr Model of the atom with the Quantum Mechanical Model of atom
 Use the Venn Diagram to compare and contrast the Bohr Model of the atom with the Quantum Mechanical Model of atom Bohr Model Quantum Model Energy level Atomic orbital Quantum Atomic number Quantum mechanical
Use the Venn Diagram to compare and contrast the Bohr Model of the atom with the Quantum Mechanical Model of atom Bohr Model Quantum Model Energy level Atomic orbital Quantum Atomic number Quantum mechanical
A bit of review. Atoms are made of 3 different SUB-ATOMIC PARTICLES: 1. ELECTRONS 2. PROTONS 3. NEUTRONS
 Chemistry in Action A bit of review Chemistry is the study of MATTER and ENERGY. Matter is anything that has MASS. All matter is made of super small particles called ATOMS. Atoms are made of 3 different
Chemistry in Action A bit of review Chemistry is the study of MATTER and ENERGY. Matter is anything that has MASS. All matter is made of super small particles called ATOMS. Atoms are made of 3 different
Sample Exercise 6.1 Concepts of Wavelength and Frequency
 Sample Exercise 6.1 Concepts of Wavelength and Frequency Two electromagnetic waves are represented in the margin. (a) Which wave has the higher frequency? (b) If one wave represents visible light and the
Sample Exercise 6.1 Concepts of Wavelength and Frequency Two electromagnetic waves are represented in the margin. (a) Which wave has the higher frequency? (b) If one wave represents visible light and the
Structure of the Atom. Intext Exercise 1
 What are canal rays? Intext Exercise 1 Canal rays are positively charged radiations. These rays consist of positively charged particles known as protons. They were discovered by Goldstein in 16. If an
What are canal rays? Intext Exercise 1 Canal rays are positively charged radiations. These rays consist of positively charged particles known as protons. They were discovered by Goldstein in 16. If an
The Periodic Table. Atoms, Elements, and the Periodic Table
 Atoms, Elements, and the Periodic Table Element: a pure substance that cannot be broken down into simpler substances by a chemical reaction. Each element is identified by a one- or two-letter symbol. Elements
Atoms, Elements, and the Periodic Table Element: a pure substance that cannot be broken down into simpler substances by a chemical reaction. Each element is identified by a one- or two-letter symbol. Elements
2.3 Periodic Table and Atomic Theory
 2.3 Periodic Table and Atomic Theory https://www.khanacademy.org/science/biology/chemistry-- of-life/electron-shells-and-orbitals/v/orbitals https://www.youtube.com/watch?v=ghan8xzq-d8 2.3 Periodic Table
2.3 Periodic Table and Atomic Theory https://www.khanacademy.org/science/biology/chemistry-- of-life/electron-shells-and-orbitals/v/orbitals https://www.youtube.com/watch?v=ghan8xzq-d8 2.3 Periodic Table
Student Exploration: Electron Configuration
 Name: Date: Student Exploration: Electron Configuration Vocabulary: atomic number, atomic radius, Aufbau principle, chemical family, diagonal rule, electron configuration, Hund s rule, orbital, Pauli exclusion
Name: Date: Student Exploration: Electron Configuration Vocabulary: atomic number, atomic radius, Aufbau principle, chemical family, diagonal rule, electron configuration, Hund s rule, orbital, Pauli exclusion
CHEMISTRY 9 REVIEW & INTRO TO CHEMISTRY 10. Section 4.1: Atomic Theory and Bonding
 1 CHEMISTRY 9 REVIEW & INTRO TO CHEMISTRY 10 Section 4.1: Atomic Theory and Bonding ATOMS AND COMPOUNDS An atom is the smallest particle of an element that still has the properties of that element An atom
1 CHEMISTRY 9 REVIEW & INTRO TO CHEMISTRY 10 Section 4.1: Atomic Theory and Bonding ATOMS AND COMPOUNDS An atom is the smallest particle of an element that still has the properties of that element An atom
Question 1: What are canal rays? Answer: Canal rays are positively charged radiations. These rays consist of positively charged particles known as
 Question 1: What are canal rays? Canal rays are positively charged radiations. These rays consist of positively charged particles known as protons. They were discovered by Goldstein in 1886. Question 2:
Question 1: What are canal rays? Canal rays are positively charged radiations. These rays consist of positively charged particles known as protons. They were discovered by Goldstein in 1886. Question 2:
Test Review # 4. Chemistry: Form TR4-9A
 Chemistry: Form TR4-9A REVIEW Name Date Period Test Review # 4 Location of electrons. Electrons are in regions of the atom known as orbitals, which are found in subdivisions of the principal energy levels
Chemistry: Form TR4-9A REVIEW Name Date Period Test Review # 4 Location of electrons. Electrons are in regions of the atom known as orbitals, which are found in subdivisions of the principal energy levels
Chapter 6. Electronic. Electronic Structure of Atoms Pearson Education
 Chapter 6 Laser: step-like energy transition 6.1 The Wave Nature of Light 6.2 Quantized Energy and Photons 6.3 Line Spectra and the Bohr Model 6.4 The Wave Behavior of Matter 6.5 Quantum Mechanics and
Chapter 6 Laser: step-like energy transition 6.1 The Wave Nature of Light 6.2 Quantized Energy and Photons 6.3 Line Spectra and the Bohr Model 6.4 The Wave Behavior of Matter 6.5 Quantum Mechanics and
Ionic Bond Proton. Cation Electron. Valence Electrons Atomic mass. Octet Rule Isotope
 Atoms and Ions Test Study Guide Physical Science Ms. Rowlen 2017 Know these definitions. Atom Ionic Bond Proton Ion Neutron Cation Electron Anion Atomic number Valence Electrons Atomic mass Octet Rule
Atoms and Ions Test Study Guide Physical Science Ms. Rowlen 2017 Know these definitions. Atom Ionic Bond Proton Ion Neutron Cation Electron Anion Atomic number Valence Electrons Atomic mass Octet Rule
Chapter 2 Atoms and Elements. Electromagnetic Radiation. Electromagnetic Spectrum. Electron Energy Levels. 2.6 Electron Energy Levels
 Chapter 2 Atoms and Elements Electromagnetic Radiation 2.6 Electron Energy Levels Electromagnetic radiation Consists of energy particles called photons that travel as waves. Includes low energy particles
Chapter 2 Atoms and Elements Electromagnetic Radiation 2.6 Electron Energy Levels Electromagnetic radiation Consists of energy particles called photons that travel as waves. Includes low energy particles
Atomic Structure and the Periodic Table
 Structure and the Structure Atoms are made of three particles: 1. Protons = positive 2. Neutrons = neutral 3. Electrons = negative The protons and neutrons are in the center of the atom (the nucleus) Electrons
Structure and the Structure Atoms are made of three particles: 1. Protons = positive 2. Neutrons = neutral 3. Electrons = negative The protons and neutrons are in the center of the atom (the nucleus) Electrons
Atoms & Atomic Structure
 Atoms & Atomic Structure - An atom is the smallest piece of an element - are composed of three types of sub-atomic particles: protons, neutrons & electrons Nucleus = central portion of atom - small and
Atoms & Atomic Structure - An atom is the smallest piece of an element - are composed of three types of sub-atomic particles: protons, neutrons & electrons Nucleus = central portion of atom - small and
CHEM 120 Spring 2017 Introduction to Inorganic Chemistry Instructor Dr. Upali Siriwardane (Ph.D. Ohio State)
 CHEM 120 Spring 2017 Introduction to Inorganic Chemistry Instructor Dr. Upali Siriwardane (Ph.D. Ohio State) E-mail: upali@latech.edu Office: 311 Carson Taylor Hall ; Phone: 318-257-4941; Office Hours:
CHEM 120 Spring 2017 Introduction to Inorganic Chemistry Instructor Dr. Upali Siriwardane (Ph.D. Ohio State) E-mail: upali@latech.edu Office: 311 Carson Taylor Hall ; Phone: 318-257-4941; Office Hours:
The Atom. The Atom and The Periodic Table of Elements. Evolution of Atomic Theory
 The Atom The Atom and The Periodic Table of Elements An atom is the smallest particle of matter. It cannot be divided chemically An atom is very small: A sheet of paper is about a million atoms thick Because
The Atom The Atom and The Periodic Table of Elements An atom is the smallest particle of matter. It cannot be divided chemically An atom is very small: A sheet of paper is about a million atoms thick Because
LABELING ELECTRONS IN ATOMS
 Date: Name: LABELING ELECTRONS IN ATOMS The location of each electron in an atom is determined by a few different factors. Each factor is represented by a QUANTUM NUMBER. Prediction: What do you think
Date: Name: LABELING ELECTRONS IN ATOMS The location of each electron in an atom is determined by a few different factors. Each factor is represented by a QUANTUM NUMBER. Prediction: What do you think
PRETEST ON CHAPTER 1 ATOMIC STRUCTURE &THE PERIODIC TABLE OF ELEMENTS
 Name Date Mark: /65 = % PRETEST ON CHAPTER 1 ATOMIC STRUCTURE &THE PERIODIC TABLE OF ELEMENTS Part 1 MULTIPLE CHOICES Answer all the questions on the multiple choice sheet provided at the end 1) Oxygen
Name Date Mark: /65 = % PRETEST ON CHAPTER 1 ATOMIC STRUCTURE &THE PERIODIC TABLE OF ELEMENTS Part 1 MULTIPLE CHOICES Answer all the questions on the multiple choice sheet provided at the end 1) Oxygen
Atomic Structure: Isotopes and the Nucleus. There are 3 subatomic particles that are the building block of atoms.
 Atoms & Elements Part 1: Atomic Structure: Isotopes and the Nucleus Atoms are the building blocks of matter. What is the definition of matter? There are 3 subatomic particles that are the building block
Atoms & Elements Part 1: Atomic Structure: Isotopes and the Nucleus Atoms are the building blocks of matter. What is the definition of matter? There are 3 subatomic particles that are the building block
Unit 6: Chemistry Test 1 Study Guide
 Name: Due Date: Unit 6: Chemistry Test 1 Study Guide Date: Test Date: Unit 6 Important Topics: Please review the concepts on this study guide as well as any other notes/worksheets from this unit. I. Aim
Name: Due Date: Unit 6: Chemistry Test 1 Study Guide Date: Test Date: Unit 6 Important Topics: Please review the concepts on this study guide as well as any other notes/worksheets from this unit. I. Aim
When I lecture we will add more info, so leave spaces in your notes
 Title and Highlight Topic: EQ: Date Reflect Question: Reflect on the material by asking a question (its not suppose to be answered from notes) NOTES: Write out the notes from my website. Use different
Title and Highlight Topic: EQ: Date Reflect Question: Reflect on the material by asking a question (its not suppose to be answered from notes) NOTES: Write out the notes from my website. Use different
Agenda. Warm Up. Objective. Electron Configurations. Where are the e- s in an atom? Quantum numbers can tell us. Principle Quantum Number (n) 10/14/14
 Warm Up Agenda Worksheet: Orbital shapes Review: Quantum numbers (PAP) (Quiz next class) Lots of handouts Whiteboards: Quantum numbers practice (PAP) Turn in Flame test Lab Coloring: periodic table s,
Warm Up Agenda Worksheet: Orbital shapes Review: Quantum numbers (PAP) (Quiz next class) Lots of handouts Whiteboards: Quantum numbers practice (PAP) Turn in Flame test Lab Coloring: periodic table s,
Electrons, Energy, & the Electromagnetic Spectrum Notes
 Electrons, Energy, & the Electromagnetic Spectrum Notes Bohr Model Diagram Interpretation What form of EM radiation is released when an electron in a hydrogen atom falls from the 5 th energy level to the
Electrons, Energy, & the Electromagnetic Spectrum Notes Bohr Model Diagram Interpretation What form of EM radiation is released when an electron in a hydrogen atom falls from the 5 th energy level to the
POGIL 6 Key Periodic Table Trends (Part 2)
 Honors Chem Block Name POGIL 6 Key Periodic Table Trends (Part 2) is a measure of the ability of an atom s nucleus to attract electrons from a different atom within a covalent bond. A higher electronegativity
Honors Chem Block Name POGIL 6 Key Periodic Table Trends (Part 2) is a measure of the ability of an atom s nucleus to attract electrons from a different atom within a covalent bond. A higher electronegativity
Chapter 11 Prep Test CLASS SET!!!! Matching
 CLASS SET!!!! ID: A Chapter 11 Prep Test Matching Match each item with the correct statement below. A electronegativity D period B ionization energy E transition metal C atomic radius F group 1 type of
CLASS SET!!!! ID: A Chapter 11 Prep Test Matching Match each item with the correct statement below. A electronegativity D period B ionization energy E transition metal C atomic radius F group 1 type of
Name. Date. Handout 3. Drawing Electron Shell Diagrams
 1 Supplemental Instruction Biology 1441 Spring 2007 SI Leader: Syed Abbas Name Date Handout 3. Drawing Electron Shell Diagrams Electron shell diagrams help us visualize how electrons are arranged around
1 Supplemental Instruction Biology 1441 Spring 2007 SI Leader: Syed Abbas Name Date Handout 3. Drawing Electron Shell Diagrams Electron shell diagrams help us visualize how electrons are arranged around
H!!!! = E! Lecture 7 - Atomic Structure. Chem 103, Section F0F Unit II - Quantum Theory and Atomic Structure Lecture 7. Lecture 7 - Introduction
 Chem 103, Section F0F Unit II - Quantum Theory and Atomic Structure Lecture 7 Lecture 7 - Atomic Structure Reading in Silberberg - Chapter 7, Section 4 The Qunatum-Mechanical Model of the Atom The Quantum
Chem 103, Section F0F Unit II - Quantum Theory and Atomic Structure Lecture 7 Lecture 7 - Atomic Structure Reading in Silberberg - Chapter 7, Section 4 The Qunatum-Mechanical Model of the Atom The Quantum
Chemistry 101 Chapter 11 Modern Atomic Theory
 Chemistry 101 Chapter 11 Modern Atomic Theory Electromagnetic radiation: energy can be transmitted from one place to another by lightmore properly called electromagnetic radiation. Many kinds of electromagnetic
Chemistry 101 Chapter 11 Modern Atomic Theory Electromagnetic radiation: energy can be transmitted from one place to another by lightmore properly called electromagnetic radiation. Many kinds of electromagnetic
4.1 Atomic Theory and Bonding
 4.1 Atomic Theory and Bonding An atom is the that still has the 50 million atoms, = An atom = (s) + (s) + (s) Atoms join together. A compound is a that is composed of combined in a. and are atoms/elements;
4.1 Atomic Theory and Bonding An atom is the that still has the 50 million atoms, = An atom = (s) + (s) + (s) Atoms join together. A compound is a that is composed of combined in a. and are atoms/elements;
Photocopied/printed notes can not be used during the Unit Notebook Check in class.
 These notes are posted on my site for the following reasons: for students to copy in their own hand-writing in order to complete their class notes if student did not have enough time in class if student
These notes are posted on my site for the following reasons: for students to copy in their own hand-writing in order to complete their class notes if student did not have enough time in class if student
Of The Following Cannot Accommodate More Than An Octet Of Electrons
 Of The Following Cannot Accommodate More Than An Octet Of Electrons This is most common example of exceptions to the octet rule. their empty d orbitals to accommodate additional electrons. A case where
Of The Following Cannot Accommodate More Than An Octet Of Electrons This is most common example of exceptions to the octet rule. their empty d orbitals to accommodate additional electrons. A case where
Mendeleev s Periodic Law
 Mendeleev s Periodic Law Periodic Law When the elements are arranged in order of increasing atomic mass, certain sets of properties recur periodically. Mendeleev s Periodic Law allows us to predict what
Mendeleev s Periodic Law Periodic Law When the elements are arranged in order of increasing atomic mass, certain sets of properties recur periodically. Mendeleev s Periodic Law allows us to predict what
Atomic Structure & the Periodic Table
 Atomic Structure & the Periodic Table Basic Definitions smallest unit of an element that retains the properties of that element Atoms are made up of several subatomic particles called,, and Protons, Neutrons,
Atomic Structure & the Periodic Table Basic Definitions smallest unit of an element that retains the properties of that element Atoms are made up of several subatomic particles called,, and Protons, Neutrons,
POGIL EXERCISE 16 Concentration Terms and There Use
 RUN TIME = 80 MIN POGIL 16 Page 1 of 10 POGIL EXERCISE 16 Concentration Terms and There Use Each member should locate his/her role in Table 1 and assume his or her role at this time. The new manager takes
RUN TIME = 80 MIN POGIL 16 Page 1 of 10 POGIL EXERCISE 16 Concentration Terms and There Use Each member should locate his/her role in Table 1 and assume his or her role at this time. The new manager takes
Chapter 4: Structure of the Atom Science
 Chapter 4: Structure of the Atom Science 1. What are canal rays? Canal rays are positively charged radiations that can pass through perforated cathode plate. These rays consist of positively charged particles
Chapter 4: Structure of the Atom Science 1. What are canal rays? Canal rays are positively charged radiations that can pass through perforated cathode plate. These rays consist of positively charged particles
Chapter 3: Electron Structure and the Periodic Law
 PERIODIC LAW This is a statement about the behavior of the elements when they are arranged in a specific order. In its present form the statement is: Elements with similar chemical properties occur at
PERIODIC LAW This is a statement about the behavior of the elements when they are arranged in a specific order. In its present form the statement is: Elements with similar chemical properties occur at
Matter. Atoms and Elements
 Matter Atoms and Elements Chemistry: The study of matter and its changes So, what is matter and what isn t? Matter isn t: Energy in the form of waves-- Light, Electricity, Heat, Magnetic Fields, Sound,
Matter Atoms and Elements Chemistry: The study of matter and its changes So, what is matter and what isn t? Matter isn t: Energy in the form of waves-- Light, Electricity, Heat, Magnetic Fields, Sound,
Unit 8: Atomic Theory. Quantum Mechanics
 Unit 8: Atomic Theory Quantum Mechanics 1 Unit 8: Atomic Theory 1. Historical Views of the Atom 2. The 'New' Look Atom 3. Electron Configurations 4. Electron Configurations & the Periodic Table 5. Quantum
Unit 8: Atomic Theory Quantum Mechanics 1 Unit 8: Atomic Theory 1. Historical Views of the Atom 2. The 'New' Look Atom 3. Electron Configurations 4. Electron Configurations & the Periodic Table 5. Quantum
6.4 Electronic Structure of Atoms (Electron Configurations)
 Chapter 6 Electronic Structure and Periodic Properties of Elements 317 Orbital n l m l degeneracy Radial nodes (no.) 4f 4 3 7 0 4p 4 1 3 2 7f 7 3 7 3 5d 5 2 5 2 Check Your Learning How many orbitals have
Chapter 6 Electronic Structure and Periodic Properties of Elements 317 Orbital n l m l degeneracy Radial nodes (no.) 4f 4 3 7 0 4p 4 1 3 2 7f 7 3 7 3 5d 5 2 5 2 Check Your Learning How many orbitals have
9/13/2011. The Greek Philosophers. Atomic Structure & The Periodic Table. Dalton s Atomic Theory. J. J. Thomson. Thomson s Experiment
 Atomic Structure & The Periodic Table The Greek Philosophers Democritus believed that all matter is made up of tiny particles that could not be divided Aristotle -- thought that matter was made of only
Atomic Structure & The Periodic Table The Greek Philosophers Democritus believed that all matter is made up of tiny particles that could not be divided Aristotle -- thought that matter was made of only
POGIL LAB EXERCISE 15 HOW DO YOU STANDARDIZE AN ACID AND BASE?
 POGIL LAB EXERCISE 15 HOW DO YOU STANDARDIZE AN ACID AND BASE? POGIL LAB 15 Page 1 of 10 Each member should assume his or her role at this time. The new manager takes charge of the POGIL folder and hands
POGIL LAB EXERCISE 15 HOW DO YOU STANDARDIZE AN ACID AND BASE? POGIL LAB 15 Page 1 of 10 Each member should assume his or her role at this time. The new manager takes charge of the POGIL folder and hands
E J The electron s energy difference between the second and third levels is J. = J
 The wavelength of light emitted is 654 nm. = c f c 3.00 10 8 m/s f c 3.00 108 m 1s 6.54 10 7 m f 4.59 4.59 10 14 z 1 s 10 14 The frequency of the light emitted is 4.59 10 14 z. E hf h 6.63 10 34 J/z E
The wavelength of light emitted is 654 nm. = c f c 3.00 10 8 m/s f c 3.00 108 m 1s 6.54 10 7 m f 4.59 4.59 10 14 z 1 s 10 14 The frequency of the light emitted is 4.59 10 14 z. E hf h 6.63 10 34 J/z E
Atomic Model and Periodic Table Test Review
 Atomic Model and Periodic Table Test Review A. Give the family name for each description. 1. I have 1 electron on my outer shell. 2. One of the elements has 35 protons. 3. I have 2 electrons on my outer
Atomic Model and Periodic Table Test Review A. Give the family name for each description. 1. I have 1 electron on my outer shell. 2. One of the elements has 35 protons. 3. I have 2 electrons on my outer
Name: Date: Period: ELECTRON ORBITAL ACTIVITY
 ELECTRON ORBITAL ACTIVITY Background Information: The arrangement of electrons within the orbitals of an atom is known as the electron configuration. The most stable arrangement is called the ground-state
ELECTRON ORBITAL ACTIVITY Background Information: The arrangement of electrons within the orbitals of an atom is known as the electron configuration. The most stable arrangement is called the ground-state
Model 1: Orbitals. 1. What is an atomic orbital? What are the four different orbitals?
 Name: Date: Period: POGIL: Electron Configuration and Orbitals Model 1: Orbitals The quantum mechanical model determines the allowed energies an electron can have and how likely it is to find the electron
Name: Date: Period: POGIL: Electron Configuration and Orbitals Model 1: Orbitals The quantum mechanical model determines the allowed energies an electron can have and how likely it is to find the electron
Chapter 6 Electronic Structure of Atoms
 Chapter 6. Electronic Structure of Atoms NOTE: Review your notes from Honors or regular Chemistry for the sequence of atomic models and the evidence that allowed scientists to change the model. If you
Chapter 6. Electronic Structure of Atoms NOTE: Review your notes from Honors or regular Chemistry for the sequence of atomic models and the evidence that allowed scientists to change the model. If you
Review 6: Modern Atomic Theory. Copyright Cengage Learning. All rights reserved. 11 1
 Review 6: Modern Atomic Theory Copyright Cengage Learning. All rights reserved. 11 1 The concept of the nuclear atom left unanswered questions about 1. why the nucleus is so dense. 2. why the negative
Review 6: Modern Atomic Theory Copyright Cengage Learning. All rights reserved. 11 1 The concept of the nuclear atom left unanswered questions about 1. why the nucleus is so dense. 2. why the negative
Unit 8: Atomic Theory. Quantum Mechanics
 Unit 8: Atomic Theory Quantum Mechanics 1 Unit 8: Atomic Theory 1. Historical Views of the Atom 2. The 'New' Look Atom 3. Electron Configurations 4. Electron Configurations & the Periodic Table 5. Quantum
Unit 8: Atomic Theory Quantum Mechanics 1 Unit 8: Atomic Theory 1. Historical Views of the Atom 2. The 'New' Look Atom 3. Electron Configurations 4. Electron Configurations & the Periodic Table 5. Quantum
Wave-Mechanical Model of the Atom. Aim: To write orbital notation and electron configurations representing the wave mechanical model of the atom.
 Wave-Mechanical Model of the Atom Aim: To write orbital notation and electron configurations representing the wave mechanical model of the atom. Wave-Mechanical Model of the Atom Atoms contain protons,
Wave-Mechanical Model of the Atom Aim: To write orbital notation and electron configurations representing the wave mechanical model of the atom. Wave-Mechanical Model of the Atom Atoms contain protons,
Remember Bohr s Explanation: Energy Levels of Hydrogen: The Electronic Structure of the Atom 11/28/2011
 The Electronic Structure of the Atom Bohr based his theory on his experiments with hydrogen he found that when energy is added to a sample of hydrogen, energy is absorbed and reemitted as light When passed
The Electronic Structure of the Atom Bohr based his theory on his experiments with hydrogen he found that when energy is added to a sample of hydrogen, energy is absorbed and reemitted as light When passed
Electron Configurations
 Relate the electron configuration of an element to its valence electron(s) and its position on the periodic table. Electron Configurations Outcomes: Write electron configurations for elements of the periodic
Relate the electron configuration of an element to its valence electron(s) and its position on the periodic table. Electron Configurations Outcomes: Write electron configurations for elements of the periodic
Edexcel Chemistry A-level
 Edexcel Chemistry A-level Topic 1 - Atomic Structure and Periodic Table Flashcards What was stated in Dalton s atomic theory? (4) What was stated in Dalton s atomic theory? Atoms are tiny particles made
Edexcel Chemistry A-level Topic 1 - Atomic Structure and Periodic Table Flashcards What was stated in Dalton s atomic theory? (4) What was stated in Dalton s atomic theory? Atoms are tiny particles made
Period Table Worksheet 1
 Period Table Worksheet. While doing a research project, you noted the following information about five elements. Element A :. is a solid;. conducts electricity;. has electrons in its outermost shell;.
Period Table Worksheet. While doing a research project, you noted the following information about five elements. Element A :. is a solid;. conducts electricity;. has electrons in its outermost shell;.
4.1.1 A simple model of the atom, symbols, relative atomic mass, electronic charge and isotopes
 4.1 Atomic structure and the periodic table The periodic table provides chemists with a structured organisation of the known chemical elements from which they can make sense of their physical and chemical
4.1 Atomic structure and the periodic table The periodic table provides chemists with a structured organisation of the known chemical elements from which they can make sense of their physical and chemical
Unit Two: Elements & Matter. February 1, 2016
 Unit Two: Elements & Matter February 1, 2016 Warm-Up: 2/1/2016 1. Fill in the following information: Atomic Symbol Ca 2+ Atomic Number Proton Neutron Electron 34 36 Mass Num. 2. Identify which family the
Unit Two: Elements & Matter February 1, 2016 Warm-Up: 2/1/2016 1. Fill in the following information: Atomic Symbol Ca 2+ Atomic Number Proton Neutron Electron 34 36 Mass Num. 2. Identify which family the
Science 10 Chapter 4 Atomic Theory Explains the Formation of Compounds
 What is a pure substance? -contains only 1 kind of matter What are the 2 categories of pure substances? -elements -compounds What is an element? -a pure substance that cannot be broken down into simpler
What is a pure substance? -contains only 1 kind of matter What are the 2 categories of pure substances? -elements -compounds What is an element? -a pure substance that cannot be broken down into simpler
The Electronic Structures of Atoms Electromagnetic Radiation The wavelength of electromagnetic radiation has the symbol λ.
 CHAPTER 7 Atomic Structure Chapter 8 Atomic Electron Configurations and Periodicity 1 The Electronic Structures of Atoms Electromagnetic Radiation The wavelength of electromagnetic radiation has the symbol
CHAPTER 7 Atomic Structure Chapter 8 Atomic Electron Configurations and Periodicity 1 The Electronic Structures of Atoms Electromagnetic Radiation The wavelength of electromagnetic radiation has the symbol
Electrons in the outermost s and p orbitals. These are the electrons most often involved in bonding.
 Electrons in the outermost s and p orbitals. These are the electrons most often involved in bonding. The organization of electrons in an atom or ion, from the lowest energy orbital ( s ) to the highest
Electrons in the outermost s and p orbitals. These are the electrons most often involved in bonding. The organization of electrons in an atom or ion, from the lowest energy orbital ( s ) to the highest
Early Chemistry. Early Chemists only believed in 1 element: Dirt. Later Chemists believed in 4 elements:
 Early Chemistry Early Chemists only believed in 1 element: Dirt Later Chemists believed in 4 elements: Air Earth Fire Water Various combinations of these produced various compounds Atomic Structure All
Early Chemistry Early Chemists only believed in 1 element: Dirt Later Chemists believed in 4 elements: Air Earth Fire Water Various combinations of these produced various compounds Atomic Structure All
Keep protons in the nucleus from repelling each other. Atomic Number Mass Number Atomic Mass number of protons determines identity of atom
 Module 1 - The Atom page 1 of 5 A: Matter & Energy Matter The Has, can be weighed, takes up space Made of Energy Makes the stuff do things, like move or glow No mass, doesn t take up space B: Atomic Structure
Module 1 - The Atom page 1 of 5 A: Matter & Energy Matter The Has, can be weighed, takes up space Made of Energy Makes the stuff do things, like move or glow No mass, doesn t take up space B: Atomic Structure
ELECTRONIC STRUCTURE OF ATOMS
 ELECTRONIC STRUCTURE OF ATOMS Electron Spin The electron: spins around its own axis acts as an tiny magnet (any moving electrical charge creates a magnetic field around itself) can spin in either of 2
ELECTRONIC STRUCTURE OF ATOMS Electron Spin The electron: spins around its own axis acts as an tiny magnet (any moving electrical charge creates a magnetic field around itself) can spin in either of 2
Friday November 4. Wave-Particle Duality
 Friday November 4 Entry Task: Take the wave problems from Wednesday Learning Target: I can begin to identify the properties of light as either a particle or a wave Wave-Particle Duality Pre-19 th century
Friday November 4 Entry Task: Take the wave problems from Wednesday Learning Target: I can begin to identify the properties of light as either a particle or a wave Wave-Particle Duality Pre-19 th century
A1: Atomic Structure Worksheet (Goals 1 3, Chapter 4)
 Unit 3 Assignment Packet Name: Period: A1: Atomic Structure Worksheet (Goals 1 3, Chapter 4) 1. Democritus, who lived in Greece during the 4 th century B.C., suggested that is made up of tiny particles
Unit 3 Assignment Packet Name: Period: A1: Atomic Structure Worksheet (Goals 1 3, Chapter 4) 1. Democritus, who lived in Greece during the 4 th century B.C., suggested that is made up of tiny particles
Chemistry B11 Chapter 3 Atoms
 Chapter 3 Atoms Element: is a substance that consists of identical atoms (hydrogen, oxygen, and Iron). 116 elements are known (88 occur in nature and chemist have made the others in the lab). Compound:
Chapter 3 Atoms Element: is a substance that consists of identical atoms (hydrogen, oxygen, and Iron). 116 elements are known (88 occur in nature and chemist have made the others in the lab). Compound:
Chapter 9 Periodic Law The structure of molecules and describing reactions
 Chapter 9 Periodic Law The structure of molecules and describing reactions Reading Assignment: Read the entire chapter. Homework: see the web site for homework. http://web.fccj.org/~smilczan/psc/homework7_11.htm
Chapter 9 Periodic Law The structure of molecules and describing reactions Reading Assignment: Read the entire chapter. Homework: see the web site for homework. http://web.fccj.org/~smilczan/psc/homework7_11.htm
Chemistry 11. Unit 8 Atoms and the Periodic Table Part II Electronic Structure of Atoms
 Chemistry 11 Unit 8 Atoms and the Periodic Table Part II Electronic Structure of Atoms 2 1. Atomic number and atomic mass In the previous section, we have seen that from 50 to 100 years after Dalton proposed
Chemistry 11 Unit 8 Atoms and the Periodic Table Part II Electronic Structure of Atoms 2 1. Atomic number and atomic mass In the previous section, we have seen that from 50 to 100 years after Dalton proposed
Noble Gas Config. Period Block (s, p, d, f) Group
 LOCATION OF ELEMENTS WORKSHEET Noble Gas Config. Period Block (s, p, d, f) Group 1 [Ne] 3s 2 3p 2 2 [Ar] 4s 2 3d 10 4p 6 3 [Xe] 6s 2 4 [Kr] 5s 2 4d 10 5p 5 5 [Ar] 4s 2 3d 10 4p 1 6 [He] 2s 2 2p 3 7 [Kr]
LOCATION OF ELEMENTS WORKSHEET Noble Gas Config. Period Block (s, p, d, f) Group 1 [Ne] 3s 2 3p 2 2 [Ar] 4s 2 3d 10 4p 6 3 [Xe] 6s 2 4 [Kr] 5s 2 4d 10 5p 5 5 [Ar] 4s 2 3d 10 4p 1 6 [He] 2s 2 2p 3 7 [Kr]
Quantum Theory and Electron Configurations
 Chapter 5 Chapter 5 Quantum Theory and Electron Configurations It s all about color In terms of atomic models, so far: Dalton (1803) = Tiny, solid particle Thomson (1897) = Plum Pudding model Electrons
Chapter 5 Chapter 5 Quantum Theory and Electron Configurations It s all about color In terms of atomic models, so far: Dalton (1803) = Tiny, solid particle Thomson (1897) = Plum Pudding model Electrons
Chapter 9: Elements are the Building blocks of Life
 Chapter 9: Elements are the Building blocks of Life Section 9.1- Elements and the Periodic Table Keep Scale in mind Animation: http://htwins.net/scale2/ I. ELEMENTS All matter is made up of one or more
Chapter 9: Elements are the Building blocks of Life Section 9.1- Elements and the Periodic Table Keep Scale in mind Animation: http://htwins.net/scale2/ I. ELEMENTS All matter is made up of one or more
Unit 1 Atomic Theory
 Unit 1 Atomic Theory 1.0 You are expected to be already familiar with. Ionic nomenclature (binary, polyatomic, multivalency) Covalent nomenclature Writing chemical formulas for ionic and covalent compounds
Unit 1 Atomic Theory 1.0 You are expected to be already familiar with. Ionic nomenclature (binary, polyatomic, multivalency) Covalent nomenclature Writing chemical formulas for ionic and covalent compounds
Test Topics: Periodic Table, Atomic Theory, Physical/Chemical Properties, Atom, Isotopes, Average Atomic Mass
 Elemental Properties Review Worksheet Test Topics: Periodic Table, Atomic Theory, Physical/Chemical Properties, Atom, Isotopes, Average Atomic Mass Periodic Table 1. List the element symbols for the following
Elemental Properties Review Worksheet Test Topics: Periodic Table, Atomic Theory, Physical/Chemical Properties, Atom, Isotopes, Average Atomic Mass Periodic Table 1. List the element symbols for the following
What are the isotopes of hydrogen? H, 2 H, and 3 H
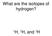 What are the isotopes of hydrogen? 1 H, 2 H, and 3 H The average mass of a boron atom is 10.81. Assuming you were able to isolate only one boron atom, the chance that you would randomly get one with a
What are the isotopes of hydrogen? 1 H, 2 H, and 3 H The average mass of a boron atom is 10.81. Assuming you were able to isolate only one boron atom, the chance that you would randomly get one with a
ATOMIC MODELS AND ELECTRON BEHAVIOR. Chelsea I Academic Chemistry
 ATOMIC MODELS AND ELECTRON BEHAVIOR Chelsea I Academic Chemistry Proposed by Neils Bohr, a Danish physicist in 1913 Hydrogen atoms have energy states, the lowest state being the ground state (n=1) When
ATOMIC MODELS AND ELECTRON BEHAVIOR Chelsea I Academic Chemistry Proposed by Neils Bohr, a Danish physicist in 1913 Hydrogen atoms have energy states, the lowest state being the ground state (n=1) When
5. N. 9. Cl 2. Pb. 6. Ag. c. 4f d. 3d
 Brief Instructions An electron configuration is a method of indicating the arrangement of electrons about a nucleus. A typical electron configuration consists of numbers, letters, and superscripts with
Brief Instructions An electron configuration is a method of indicating the arrangement of electrons about a nucleus. A typical electron configuration consists of numbers, letters, and superscripts with
Unit 2: Atomic Structure Practice Packet
 Unit 2: Atomic Structure Practice Packet Dalton s Model: hard sphere model 1. I can describe John Dalton s contribution to our Thomson s Experiment: cathode ray experiment 2. I can describe JJ Thomson
Unit 2: Atomic Structure Practice Packet Dalton s Model: hard sphere model 1. I can describe John Dalton s contribution to our Thomson s Experiment: cathode ray experiment 2. I can describe JJ Thomson
CHEM 1305: Introductory Chemistry
 CHEM 1305: Introductory Chemistry The Periodic Table From Chapter 5 Textbook Introductory Chemistry: Concepts and Critical Thinking Seventh Edition by Charles H. Corwin Classification of Elements By 1870,
CHEM 1305: Introductory Chemistry The Periodic Table From Chapter 5 Textbook Introductory Chemistry: Concepts and Critical Thinking Seventh Edition by Charles H. Corwin Classification of Elements By 1870,
Periodic Table of Elements. Matter: Properties and Changes
 Periodic Table of Elements Matter: Properties and Changes Warm up Instructions: 1. Use the periodic table of elements to looks for the letters in your first or last name to write your name in elements.
Periodic Table of Elements Matter: Properties and Changes Warm up Instructions: 1. Use the periodic table of elements to looks for the letters in your first or last name to write your name in elements.
MODULE-21 TRENDS IN THE MODERN PERIODIC TABLE
 MODULE-21 TRENDS IN THE MODERN PERIODIC TABLE Valency is defined as the number of electrons an atom requires to lose, gain, or share in order to complete its valence shell to attain the stable noble gas
MODULE-21 TRENDS IN THE MODERN PERIODIC TABLE Valency is defined as the number of electrons an atom requires to lose, gain, or share in order to complete its valence shell to attain the stable noble gas
Atomic Theory. Quantum Mechanics
 Atomic Theory Quantum Mechanics Quantum Mechanics The ol solar system model of the atom does have some practical uses It tells us that protons and neutrons are in the nucleus, and electrons are in orbitals
Atomic Theory Quantum Mechanics Quantum Mechanics The ol solar system model of the atom does have some practical uses It tells us that protons and neutrons are in the nucleus, and electrons are in orbitals
Electron Configuration
 Electron Configuration Orbitals Remember that orbitals simply represent an area where the electron is most likely to be found. Formally, orbitals are defined using four quantum numbers Orbitals have particular
Electron Configuration Orbitals Remember that orbitals simply represent an area where the electron is most likely to be found. Formally, orbitals are defined using four quantum numbers Orbitals have particular
Unit 4B- Electron Configuration- Guided Notes
 Unit 4B- Electron Configuration- Guided Notes Atomic Structure Electrons are arranged in or around the nucleus of an atom o First shell can hold a maximum of electrons o Second shell can hold a maximum
Unit 4B- Electron Configuration- Guided Notes Atomic Structure Electrons are arranged in or around the nucleus of an atom o First shell can hold a maximum of electrons o Second shell can hold a maximum
