Single-molecule covalent chemistry in a protein nanoreactor
|
|
|
- Bryan Summers
- 5 years ago
- Views:
Transcription
1 Single-molecule covalent chemistry in a protein nanoreactor Summer School on Biosensing Jacobs University Bremen 28th June 2009 Laboratory of Hagan Bayley University of Oxford
2 We can do lots of things with nanopores: stochastic sensing engineered pores adapters polymer fishing lines polymer analysis counting/ identification sequencing [?] relatively little work on covalent chemistry
3 Utility in sensing There are lots of reactive analytes: chemical warfare agents pesticides anticancer drugs onions
4 Chemistry at the single-molecule level: using a transmembrane protein pore as a nanoreactor α-hemolysin pore as a nanoreactor favorable dimensions
5 Current measurements are made by single channel electrical recording in planar bilayers Perturbations of the ionic current tell us what is happening inside the pore, we see individual interactions or reaction steps for single molecules cis V V = 40 mv 700 ps = 30 pa 1 M NaCl trans In this talk: various V, salt, filtering, timescales etc.
6 Protein nanopore detection provides kinetic information τ on = 1/k on [A] τ off = 1/k off K d = k off / k on The signal contains kinetic information with ~10 µsec resolution under favorable circumstances
7 Single molecule covalent chemistry inside the pore irreversible reactions reversible covalent chemistry irreversible chemistry with turnover limits and future prospects
8 Advantages: short-lived intermediates in any step minor pathways fast reactions to ~10 µsec no tags required, e.g. fluorophores high reagent concentrations possible good control over temperature, ph, salt
9 Single molecule covalent chemistry inside the pore irreversible reactions reversible covalent chemistry irreversible chemistry with turnover
10 Chemistry of caged proteins Loudwig, S. and Bayley, H. Light-activated proteins: an overview, pp , in Dynamic studies in biology: phototriggers, photoswitches and caged biomolecules, M. Goeldner and R. Givens, eds., Wiley-VCH Verlag, Weinheim, (2005)
11 hν
12
13
14 lifetime of carbamate fast step, after slow step pk a = 6.4
15 Chemistry of caged proteins slow! Good for cell biology Too slow for e.g. protein folding
16 Single molecule covalent chemistry inside the pore irreversible reactions reversible covalent chemistry irreversible chemistry with turnover
17 Azobenzene isomerization at the single-molecule level Loudwig, S. and Bayley, H. J. Am. Chem. Soc. 128, (2006)
18 Reactive toxic molecules - examples, chemical warfare agents Lewisite VX nerve gas mustard gas
19 Arsenic(III) as a poison environment (skin disease, cancer) therapeutic agents (Ehrlich, trypanosomiasis, leukemia) chemical weapons (Lewisite) rat poison, murder (As 2 O 3 - tasteless)
20 Arsenic(III) as a poison environment (skin disease, cancer) therapeutic agents (Ehrlich, trypanosomiasis, leukemia) chemical weapons (Lewisite) rat poison, murder (As 2 O 3 - tasteless)
21 Organoarsenic(III) chemistry
22 Organoarsenic(III) chemistry
23 αhl pore containing a single cysteine projecting into the channel lumen P SH
24 4-sulfophenylarsine oxide reacts reversibly with P SH
25 Chiral arsenic adducts 250 P SH count B A (G137C) 1 (WT) Conductance (pa) rare transition, overrepresented here
26 kaon = (14+1) x 103 M-1s-1 kbon = ( ) x103 M-1s-1 kaoff = ( ) s-1 kboff = ( ) s-1 kab = ( ) s-1 kba = ( )s-1 kaon kab kboff=( )x103 M-1s-3 kbon kba kaoff=( )x103 M-1s-3
27 Single molecule covalent chemistry inside the pore irreversible reactions reversible covalent chemistry irreversible chemistry with turnover
28
29 fast step, after slow step
30 rate of cyclization step
31 X
32 Polymerization monitored step by step cf.
33
34 How far can this technology be pushed? reaction network (stereochemistry) isotope effect
35 Stereochemistry of nucleophilic reaction at As P 1 SH 5 A S As OH R 1 2 B OH S As D S As R 2 S R C S As S R 1 R 1 R 2
36 P 1 SH 5 A S As OH R 1 2 B OH S As D S As R 2 S R C S As S R 1 R 1 R 2
37 1 P SH 17 D A S As S As OH R2 S R 1 OH S As S As 8 R 6 1 R 2 S E 2 14 S As S R 3 R F B 5 16 C R 3 S S As 10 R 1 R 1 R 1
38 Complex reaction network
39 .. allowed and forbidden transitions... P HO As HO SO 3 SH R 1 = SO 3 A S As OH R 1 B OH S As H 2 N R 2 = O OH D S As O OH R 2 S R 1 S As S C R 2 R 1 R 1 R 3 = O HO H 2 N O O NH NH E S As S R 3 R 1 S R 3 S As F R 1
40 ... set limits on the stereochemistry... inversion, retention, racemization...
41 Measuring an isotope effect
42
43
44
45 single molecule- lifetime of intermediate: τ* D /τ* H = (k -1,H + k 2,H )/(k -1,D + k 2,D ) bulk: k obs-h /k obs-d = (k 1H k 2H /k 1D k 2D )(k -1,D + k 2,D )/(k -1,H + k 2,H ) for k 2 relatively large (fast second step) τ* D /τ* H = (k 2,H )/(k 2,D )... see full isotope effect k obs-h /k obs-d ~ 1 because = k 1H /k 1D ~ k -1,D /k -1,H ~ 1 (and are rate limiting)
46
47 Next level issues/problems: Is water in the pore like bulk water? 400 molecules in the barrel Protein surface effects Are reactants driven into the pore? charged reactants neutral reactants- electroosmosis Can large flexible molecules get in? note that for a sensor all this does not matter
48 Is water in the pore like bulk water?- probably radii constriction
49 X-ray structures of mutant αhl βcd complexes M. Montoya & E. Gouaux
50 Are charged reactants driven into the pore? - yes, but effect can be small at low applied potentials K d ( V) = K d (0)e-z F δ V/RT
51 Are neutral reactants driven into the pore by electrosmosis? Yes- can reduce current and therefore EOF by manipulating applied potential and ion selectivity
52 Can large flexible molecules get in?- They pay a price (i) POLYMER'-SH + N S S N POLYMER'- S S N (ii) POLYMER'- S S N + HS-ALPHA HL POLYMER'-S- S-ALPHA HL Π = [PEG] pore /[PEG] bath = e -N(a/D) 5/3 for an infinitely long tube (de Gennes) ln k = -N(a/D) 5/3 + ln k[peg] bath k, measured rate constant D, diameter pore a, persistence length of polymer N, number of repeats in polymer
53 Expanding the applicability of nanopore technology to covalent chemistry temperature ph solvents chemistry other than SH range of accessible rate constants (noise) more precise measurements
54 Protein pores at high temperatures α-hemolysin, leukocidin and OmpG are stable at high temperatures and show no sharp transitions in conductance
55 (M113F/K147N) 7, βcd, adamantane-1-carboxylate
56
57 next: reactive side chains
58 QuickTime and a TIFF (Uncompressed) decompressor are needed to see this picture. visit our web site QuickTime and a TIFF (Uncompressed) decompressor are needed to see this picture. QuickTime and a TIFF (Uncompressed) decompressor are needed to see this picture. QuickTime and a TIFF (Uncompressed) decompressor are needed to see this picture. Former single-molecule chemists: Seong-Ho Shin Tudor Luchian Sandra Loudwig Mackay Steffenson Haichen Wu Oxford group: Arijit Banerjee Marc Boudreau Stephen Cheley Timmy Choi Anne Hammerstein Leon Harrington Marsi Henricus Andrew Heron Deanpen Japrung Jochen Klingelhoefer Lingbing Kong Wen-wu Li Siran Lu Giovanni Maglia Rethi Madathil Ellina Mikhailova Kunal Mehta Sarah Rogers Pinky Raychaudhuri James Reeve Dvir Rotem Maria Salichou Tanuj Sapra Samantha Shanley David Stoddart Ruhma Syeda Emma Wallace
59
Stochastic sensing: from metal ions to DNA sequencing, and beyond
 Stochastic sensing: from metal ions to DNA sequencing, and beyond 3 rd READNA Symposium PRBB Auditorium Barcelona 28th September 2012 Laboratory of Hagan Bayley University of Oxford.. G... T... A.....
Stochastic sensing: from metal ions to DNA sequencing, and beyond 3 rd READNA Symposium PRBB Auditorium Barcelona 28th September 2012 Laboratory of Hagan Bayley University of Oxford.. G... T... A.....
Single molecule investigations of the phdependent interaction between nanoparticles and an a-hemolysin protein pore
 Single molecule investigations of the phdependent interaction between nanoparticles and an a-hemolysin protein pore Dr. Alina ASANDEI The Science Department of Alexandru Ioan Cuza University Iasi 2012
Single molecule investigations of the phdependent interaction between nanoparticles and an a-hemolysin protein pore Dr. Alina ASANDEI The Science Department of Alexandru Ioan Cuza University Iasi 2012
Role of hydration in nanopore sensing: Hofmeister series, preferential solvation, osmotic stress, and all that
 Role of hydration in nanopore sensing: Hofmeister series, preferential solvation, osmotic stress, and all that Sergey M. Bezrukov Laboratory of Physical and Structural Biology, NICHD National Institutes
Role of hydration in nanopore sensing: Hofmeister series, preferential solvation, osmotic stress, and all that Sergey M. Bezrukov Laboratory of Physical and Structural Biology, NICHD National Institutes
Supporting Information
 Supporting Information Wiley-VCH 2005 69451 Weinheim, Germany Single protein pores containing molecular adapters at high temperatures Xiao-feng Kang, Li-Qun Gu, Stephen Cheley, and Hagan Bayley* [*] Hagan
Supporting Information Wiley-VCH 2005 69451 Weinheim, Germany Single protein pores containing molecular adapters at high temperatures Xiao-feng Kang, Li-Qun Gu, Stephen Cheley, and Hagan Bayley* [*] Hagan
Leader: Deborah Course: CHEM 178 Instructor: Bonaccorsi/Vela Date: 2/6/18 + H 2 CHF 3. a. Express the rate law in terms of m, n, and k.
 EXAM I REVIEW KEY Leader: Deborah Course: CHEM 178 Instructor: Bonaccorsi/Vela Date: 2/6/18 1. Given the following reaction: CF 4 + H 2 CHF 3 + HF a. Express the rate law in terms of m, n, and k. Rate
EXAM I REVIEW KEY Leader: Deborah Course: CHEM 178 Instructor: Bonaccorsi/Vela Date: 2/6/18 1. Given the following reaction: CF 4 + H 2 CHF 3 + HF a. Express the rate law in terms of m, n, and k. Rate
Chapter 6 Ionic Reactions-Nucleophilic Substitution and Elimination Reactions of Alkyl Halides"
 Chapter 6 Ionic Reactions-Nucleophilic Substitution and Elimination Reactions of Alkyl Halides" t Introduction" The polarity of a carbon-halogen bond leads to the carbon having a partial positive charge"
Chapter 6 Ionic Reactions-Nucleophilic Substitution and Elimination Reactions of Alkyl Halides" t Introduction" The polarity of a carbon-halogen bond leads to the carbon having a partial positive charge"
Glendale Community College Chemistry 105 Exam. 3 Lecture Notes Chapters 6 & 7
 Sevada Chamras, Ph.D. Glendale Community College Chemistry 105 Exam. 3 Lecture Notes Chapters 6 & 7 Description: Examples: 3 Major Types of Organic Halides: 1. Alkyl Halides: Chapter 6 (Part 1/2) : Alkyl
Sevada Chamras, Ph.D. Glendale Community College Chemistry 105 Exam. 3 Lecture Notes Chapters 6 & 7 Description: Examples: 3 Major Types of Organic Halides: 1. Alkyl Halides: Chapter 6 (Part 1/2) : Alkyl
A. Reaction Mechanisms and Catalysis (1) proximity effect (2) acid-base catalysts (3) electrostatic (4) functional groups (5) structural flexibility
 (P&S Ch 5; Fer Ch 2, 9; Palm Ch 10,11; Zub Ch 9) A. Reaction Mechanisms and Catalysis (1) proximity effect (2) acid-base catalysts (3) electrostatic (4) functional groups (5) structural flexibility B.
(P&S Ch 5; Fer Ch 2, 9; Palm Ch 10,11; Zub Ch 9) A. Reaction Mechanisms and Catalysis (1) proximity effect (2) acid-base catalysts (3) electrostatic (4) functional groups (5) structural flexibility B.
Organic Chemistry 112 A B C - Syllabus Addendum for Prospective Teachers
 Chapter Organic Chemistry 112 A B C - Syllabus Addendum for Prospective Teachers Ch 1-Structure and bonding Ch 2-Polar covalent bonds: Acids and bases McMurry, J. (2004) Organic Chemistry 6 th Edition
Chapter Organic Chemistry 112 A B C - Syllabus Addendum for Prospective Teachers Ch 1-Structure and bonding Ch 2-Polar covalent bonds: Acids and bases McMurry, J. (2004) Organic Chemistry 6 th Edition
Protein Nanopores with Covalently Attached Molecular Adapters
 Published on Web 11/30/2007 Protein Nanopores with Covalently Attached Molecular Adapters Hai-Chen Wu, Yann Astier, Giovanni Maglia, Ellina Mikhailova, and Hagan Bayley* Department of Chemistry, UniVersity
Published on Web 11/30/2007 Protein Nanopores with Covalently Attached Molecular Adapters Hai-Chen Wu, Yann Astier, Giovanni Maglia, Ellina Mikhailova, and Hagan Bayley* Department of Chemistry, UniVersity
5.03, Inorganic Chemistry Prof. Daniel G. Nocera Lecture 15 Apr 11: Substitution Reactions and the Trans Effect
 5.03, Inorganic Chemistry Prof. Daniel G. ocera Lecture 15 Apr 11: Substitution Reactions and the Trans Effect A substitution reaction is one in which an existing ligand on a metal center is replaced by
5.03, Inorganic Chemistry Prof. Daniel G. ocera Lecture 15 Apr 11: Substitution Reactions and the Trans Effect A substitution reaction is one in which an existing ligand on a metal center is replaced by
11. Reactions of Alkyl Halides: Nucleophilic Substitutions and Eliminations
 11. Reactions of Alkyl Halides: Nucleophilic Substitutions and Eliminations Based on McMurry s Organic Chemistry, 6 th edition 2003 Ronald Kluger Department of Chemistry University of Toronto Alkyl Halides
11. Reactions of Alkyl Halides: Nucleophilic Substitutions and Eliminations Based on McMurry s Organic Chemistry, 6 th edition 2003 Ronald Kluger Department of Chemistry University of Toronto Alkyl Halides
Enzyme Catalysis & Biotechnology
 L28-1 Enzyme Catalysis & Biotechnology Bovine Pancreatic RNase A Biochemistry, Life, and all that L28-2 A brief word about biochemistry traditionally, chemical engineers used organic and inorganic chemistry
L28-1 Enzyme Catalysis & Biotechnology Bovine Pancreatic RNase A Biochemistry, Life, and all that L28-2 A brief word about biochemistry traditionally, chemical engineers used organic and inorganic chemistry
Catalysis Lectures W.H. Green 5.68J/10.652J Spring Handouts: Norskov et al., J. Catalysis Imbihl and Ertl, Chem. Rev. (partial) Homework
 Catalysis Lectures W.H. Green 5.68J/10.652J Spring 2003 Handouts: Norskov et al., J. Catalysis Imbihl and Ertl, Chem. Rev. (partial) Homework Major points: 1) Why reactions have barriers, and how catalysts
Catalysis Lectures W.H. Green 5.68J/10.652J Spring 2003 Handouts: Norskov et al., J. Catalysis Imbihl and Ertl, Chem. Rev. (partial) Homework Major points: 1) Why reactions have barriers, and how catalysts
QuickTime and a TIFF (Uncompressed) decompressor are needed to see this picture. Organic Chemistry. QuickTime and a are needed to see this picture.
 QuickTime and a TIFF (Uncompressed) decompressor are needed to see this picture. Organic Chemistry QuickTime and a TIFF (Uncompressed) decompressor are needed to see this picture. Organic Chemistry Has
QuickTime and a TIFF (Uncompressed) decompressor are needed to see this picture. Organic Chemistry QuickTime and a TIFF (Uncompressed) decompressor are needed to see this picture. Organic Chemistry Has
(1) Solvent interactions Water is a ligand itself so it can become involved in the chemistry: Overall reaction: [L 5 MX] + Y [L 5 MY] + X
![(1) Solvent interactions Water is a ligand itself so it can become involved in the chemistry: Overall reaction: [L 5 MX] + Y [L 5 MY] + X (1) Solvent interactions Water is a ligand itself so it can become involved in the chemistry: Overall reaction: [L 5 MX] + Y [L 5 MY] + X](/thumbs/75/72042422.jpg) (1) Solvent interactions Water is a ligand itself so it can become involved in the chemistry: Overall reaction: [L 5 MX] + Y [L 5 MY] + X 270 1.(slow) [L 5 MX] + H 2 O [L 5 M(H 2 O)] + X 2. (fast) [L 5
(1) Solvent interactions Water is a ligand itself so it can become involved in the chemistry: Overall reaction: [L 5 MX] + Y [L 5 MY] + X 270 1.(slow) [L 5 MX] + H 2 O [L 5 M(H 2 O)] + X 2. (fast) [L 5
Chapter 11, Part 1: Polar substitution reactions involving alkyl halides
 hapter 11, Part 1: Polar substitution reactions involving alkyl halides Overview: The nature of alkyl halides and other groups with electrophilic sp 3 hybridized leads them to react with nucleophiles and
hapter 11, Part 1: Polar substitution reactions involving alkyl halides Overview: The nature of alkyl halides and other groups with electrophilic sp 3 hybridized leads them to react with nucleophiles and
CHAPTER 7. Further Reactions of Haloalkanes: Unimolecular Substitution and Pathways of Elimination
 CHAPTER 7 Further Reactions of Haloalkanes: Unimolecular Substitution and Pathways of Elimination 7-1 Solvolysis of Tertiary and Secondary Haloalkanes The rate of S N 2 reactions decrease dramatically
CHAPTER 7 Further Reactions of Haloalkanes: Unimolecular Substitution and Pathways of Elimination 7-1 Solvolysis of Tertiary and Secondary Haloalkanes The rate of S N 2 reactions decrease dramatically
Homework - Review of Chem 2310
 omework - Review of Chem 2310 Chapter 1 - Atoms and Molecules Name 1. What is organic chemistry? 2. Why is there an entire one year course devoted to the study of organic compounds? 3. Give 4 examples
omework - Review of Chem 2310 Chapter 1 - Atoms and Molecules Name 1. What is organic chemistry? 2. Why is there an entire one year course devoted to the study of organic compounds? 3. Give 4 examples
Chemistry 123: Physical and Organic Chemistry Topic 1: Organic Chemistry
 Concept Check: Topic 1: Conformation Winter 2009 Page 112 Concept Check: Topic 1: Conformation Winter 2009 Page 113 1 STEREOCHEMISTRY Winter 2009 Page 114 We have already covered two kinds of isomerism:
Concept Check: Topic 1: Conformation Winter 2009 Page 112 Concept Check: Topic 1: Conformation Winter 2009 Page 113 1 STEREOCHEMISTRY Winter 2009 Page 114 We have already covered two kinds of isomerism:
Name Biology Chapter 2 Note-taking worksheet
 Name Biology Chapter 2 Note-taking worksheet The Nature of Matter 1. Life depends on Atoms 1. The study of chemistry starts with the basic unit of matter, the. 2. The atom was first used by the Greek philosopher
Name Biology Chapter 2 Note-taking worksheet The Nature of Matter 1. Life depends on Atoms 1. The study of chemistry starts with the basic unit of matter, the. 2. The atom was first used by the Greek philosopher
Course Goals for CHEM 202
 Course Goals for CHEM 202 Students will use their understanding of chemical bonding and energetics to predict and explain changes in enthalpy, entropy, and free energy for a variety of processes and reactions.
Course Goals for CHEM 202 Students will use their understanding of chemical bonding and energetics to predict and explain changes in enthalpy, entropy, and free energy for a variety of processes and reactions.
BSc. II 3 rd Semester. Submitted By Dr. Sangita Nohria Associate Professor PGGCG-11 Chandigarh 1
 BSc. II 3 rd Semester Submitted By Dr. Sangita Nohria Associate Professor PGGCG-11 Chandigarh 1 Introduction to Alkyl Halides Alkyl halides are organic molecules containing a halogen atom bonded to an
BSc. II 3 rd Semester Submitted By Dr. Sangita Nohria Associate Professor PGGCG-11 Chandigarh 1 Introduction to Alkyl Halides Alkyl halides are organic molecules containing a halogen atom bonded to an
UNIT 4 REVISION CHECKLIST CHEM 4 AS Chemistry
 UNIT 4 REVISION CHECKLIST CHEM 4 AS Chemistry Topic 4.1 Kinetics a) Define the terms: rate of a reaction, rate constant, order of reaction and overall order of reaction b) Deduce the orders of reaction
UNIT 4 REVISION CHECKLIST CHEM 4 AS Chemistry Topic 4.1 Kinetics a) Define the terms: rate of a reaction, rate constant, order of reaction and overall order of reaction b) Deduce the orders of reaction
The Chemistry of Life 2007-
 The Chemistry of Life 2007- Why are we studying chemistry? Chemistry is the foundation of Biology The World of Elements H NaMg K Ca C N O P S Different kinds of atoms = elements Life requires ~25 chemical
The Chemistry of Life 2007- Why are we studying chemistry? Chemistry is the foundation of Biology The World of Elements H NaMg K Ca C N O P S Different kinds of atoms = elements Life requires ~25 chemical
Basic Organic Chemistry Course code : CHEM (Pre-requisites : CHEM 11122)
 Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
PRACTICE PROBLEMS UNIT 8
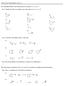 PRACTICE PROBLEMS UNIT 8 8A. Identify halides and carbocations as being 1 o, 2 o, or 3 o 8A.1 assify the following halides and carbocations as 1 o, 2 o, or 3 o. 8A.2 Circle the most stable cation in each
PRACTICE PROBLEMS UNIT 8 8A. Identify halides and carbocations as being 1 o, 2 o, or 3 o 8A.1 assify the following halides and carbocations as 1 o, 2 o, or 3 o. 8A.2 Circle the most stable cation in each
MATTER AND THE ENVIRONMENT. Environmental Science Chapter 3 Lesson 1 Textbook pgs
 MATTER AND THE ENVIRONMENT Environmental Science Chapter 3 Lesson 1 Textbook pgs. 64-71 ATOMS The atom is the most basic unit of matter. Atoms are incredibly small. Placed side by side, 100 million atoms
MATTER AND THE ENVIRONMENT Environmental Science Chapter 3 Lesson 1 Textbook pgs. 64-71 ATOMS The atom is the most basic unit of matter. Atoms are incredibly small. Placed side by side, 100 million atoms
Organometallic Rections 1: Reactions at the Metal
 E Organometallic Rections 1: Reactions at the Metal Three major classes of reactions: 1 Ligand Substitution associative (cf. S N 2) dissociative (cf. S N 1) interchange (not dealt with in this course)
E Organometallic Rections 1: Reactions at the Metal Three major classes of reactions: 1 Ligand Substitution associative (cf. S N 2) dissociative (cf. S N 1) interchange (not dealt with in this course)
LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor
 LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor Note: Adequate space is given for each answer. Questions that require a brief explanation should
LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor Note: Adequate space is given for each answer. Questions that require a brief explanation should
Subject Overview Curriculum pathway
 Subject Overview Curriculum pathway Course Summary Course: A Level Chemistry Overall Summary Unit / Module Exam / Controlled % of course UMS allocation Marks available UMS / RAW mark grade boundaries from
Subject Overview Curriculum pathway Course Summary Course: A Level Chemistry Overall Summary Unit / Module Exam / Controlled % of course UMS allocation Marks available UMS / RAW mark grade boundaries from
A primary hydrogen deuterium isotope effect observed at the single-molecule level
 Correction notice Nature Chemistry doi:10.1038/nchem.821 (2010) A primary hydrogen deuterium isotope effect observed at the single-molecule level Siran Lu, Wen-Wu Li, Dvir Rotem, Ellina Mikhailova and
Correction notice Nature Chemistry doi:10.1038/nchem.821 (2010) A primary hydrogen deuterium isotope effect observed at the single-molecule level Siran Lu, Wen-Wu Li, Dvir Rotem, Ellina Mikhailova and
Homework - I. Hand draw the structure of an eukaryotic cell with all organelles clearly drawn and describe the function of each organelle.
 Homework - I Hand draw the structure of an eukaryotic cell with all organelles clearly drawn and describe the function of each organelle. Please get a book and read the chapter related to cell structure!!!
Homework - I Hand draw the structure of an eukaryotic cell with all organelles clearly drawn and describe the function of each organelle. Please get a book and read the chapter related to cell structure!!!
What are radicals? H. Cl. Chapter 10 Radical Reactions. Production of radicals. Reactions of radicals. Electronic structure of methyl radical
 What are radicals? Radicals are intermediates with an unpaired electron Chapter 10 Radical Reactions H. Cl. Hydrogen radical Chlorine radical Methyl radical Often called free radicals Formed by homolytic
What are radicals? Radicals are intermediates with an unpaired electron Chapter 10 Radical Reactions H. Cl. Hydrogen radical Chlorine radical Methyl radical Often called free radicals Formed by homolytic
Elements and Isotopes
 Section 2-1 Notes Atoms Life depends on chemistry. The basic unit of matter is the atom. Atoms are incredibly small The subatomic particles that make up atoms are protons, neutrons, and electrons. Parts
Section 2-1 Notes Atoms Life depends on chemistry. The basic unit of matter is the atom. Atoms are incredibly small The subatomic particles that make up atoms are protons, neutrons, and electrons. Parts
THE CHEMISTRY OF LIFE
 THE CHEMISTRY OF LIFE ATOMS All living things are made up of matter Atoms are the smallest unit of matter Made up of 3 subatomic particles: 1. Protons- positively charged, found in the nucleus, has mass
THE CHEMISTRY OF LIFE ATOMS All living things are made up of matter Atoms are the smallest unit of matter Made up of 3 subatomic particles: 1. Protons- positively charged, found in the nucleus, has mass
Chemistry of Life. Chapter Two
 Chemistry of Life Chapter Two 1 Biology and Chemistry Biology = study of life Chemistry = study of matter and the changes it undergoes Matter anything that takes up space and has mass Life is made up of
Chemistry of Life Chapter Two 1 Biology and Chemistry Biology = study of life Chemistry = study of matter and the changes it undergoes Matter anything that takes up space and has mass Life is made up of
Electrochemistry & Redox. Voltaic Cells. Electrochemical Cells
 Electrochemistry & Redox An oxidation-reduction (redox) reaction involves the transfer of electrons from the reducing agent to the oxidising agent. OXIDATION - is the LOSS of electrons REDUCTION - is the
Electrochemistry & Redox An oxidation-reduction (redox) reaction involves the transfer of electrons from the reducing agent to the oxidising agent. OXIDATION - is the LOSS of electrons REDUCTION - is the
Chem 3A - Practice Midterm I. Note: This is a slightly modified version of the first midterm exam from Chem 112A Fall 2012
 Chem 3A - Practice Midterm I Note: This is a slightly modified version of the first midterm exam from Chem 112A Fall 2012 Please provide all answers in the space provided. You are not allowed to use a
Chem 3A - Practice Midterm I Note: This is a slightly modified version of the first midterm exam from Chem 112A Fall 2012 Please provide all answers in the space provided. You are not allowed to use a
Exam I Answer Key: Summer 2006, Semester C
 1. Which of the following tripeptides would migrate most rapidly towards the negative electrode if electrophoresis is carried out at ph 3.0? a. gly-gly-gly b. glu-glu-asp c. lys-glu-lys d. val-asn-lys
1. Which of the following tripeptides would migrate most rapidly towards the negative electrode if electrophoresis is carried out at ph 3.0? a. gly-gly-gly b. glu-glu-asp c. lys-glu-lys d. val-asn-lys
The Reactions of Mitomycin C with Dithiols III. Mysterious Reactions
 The Reactions of Mitomycin C with Dithiols III. Mysterious Reactions Manuel M. Paz Departmento de Química Orgánica, Universidade de Santiago de Compostela, Facultade de Ciencias, Campus de Lugo 272 Lugo,
The Reactions of Mitomycin C with Dithiols III. Mysterious Reactions Manuel M. Paz Departmento de Química Orgánica, Universidade de Santiago de Compostela, Facultade de Ciencias, Campus de Lugo 272 Lugo,
Name: Class: Date: ID: A
 Name: Class: _ Date: _ ID: A Ch 2 Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Isotopes are atoms of the same element with the same number of
Name: Class: _ Date: _ ID: A Ch 2 Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Isotopes are atoms of the same element with the same number of
UN-STIRRED LAYERS AT THE BIOMEMBRANE-AQUEOUS SOLUTION INTERFACE EVIDENCED BY ION TRANSPORT MEASUREMENTS THROUGH PORE-FORMING PROTEINS
 Journal of Optoelectronics and Advanced Materials Vol. 7, No. 2, April 2005, p. 897-902 UN-STIRRED LAYERS AT THE BIOMEMBRANE-AQUEOUS SOLUTION INTERFACE EVIDENCED BY ION TRANSPORT MEASUREMENTS THROUGH PORE-FORMING
Journal of Optoelectronics and Advanced Materials Vol. 7, No. 2, April 2005, p. 897-902 UN-STIRRED LAYERS AT THE BIOMEMBRANE-AQUEOUS SOLUTION INTERFACE EVIDENCED BY ION TRANSPORT MEASUREMENTS THROUGH PORE-FORMING
2013 W. H. Freeman and Company. 6 Enzymes
 2013 W. H. Freeman and Company 6 Enzymes CHAPTER 6 Enzymes Key topics about enzyme function: Physiological significance of enzymes Origin of catalytic power of enzymes Chemical mechanisms of catalysis
2013 W. H. Freeman and Company 6 Enzymes CHAPTER 6 Enzymes Key topics about enzyme function: Physiological significance of enzymes Origin of catalytic power of enzymes Chemical mechanisms of catalysis
Modeling Biomolecular Systems II. BME 540 David Sept
 Modeling Biomolecular Systems II BME 540 David Sept Introduction Why do we perform simulations? What makes simulations possible? How do we perform simulations? What types of things/systems do we simulate?
Modeling Biomolecular Systems II BME 540 David Sept Introduction Why do we perform simulations? What makes simulations possible? How do we perform simulations? What types of things/systems do we simulate?
Introduction to Chemical Reactions. Chapter 6
 Introduction to Chemical Reactions Chapter 6 Instructional Goals 1. Given the reactants and product in a chemical reaction, the student will be able to write and balance chemical equations. 2. Identify
Introduction to Chemical Reactions Chapter 6 Instructional Goals 1. Given the reactants and product in a chemical reaction, the student will be able to write and balance chemical equations. 2. Identify
An Overview of Organic Reactions. Reaction types: Classification by outcome Most reactions produce changes in the functional group of the reactants:
 An Overview of Organic Reactions Reaction types: Classification by outcome Most reactions produce changes in the functional group of the reactants: 1. Addition (forward) Gain of atoms across a bond Example:
An Overview of Organic Reactions Reaction types: Classification by outcome Most reactions produce changes in the functional group of the reactants: 1. Addition (forward) Gain of atoms across a bond Example:
CHM 152 Final Exam Review
 CHM 152 Final Exam Review Kinetics Chapter 12 End-of-Chapter Suggested problems: 1, 2, 3, 4, 6, 7, 9, 11, 13, 14, 15, 17, 19, 21, 25, 29, 31, 33 (graphing), 37, 39, 41, 47, 51, 53, 57, 63, 67, 68, 69,
CHM 152 Final Exam Review Kinetics Chapter 12 End-of-Chapter Suggested problems: 1, 2, 3, 4, 6, 7, 9, 11, 13, 14, 15, 17, 19, 21, 25, 29, 31, 33 (graphing), 37, 39, 41, 47, 51, 53, 57, 63, 67, 68, 69,
Kinetics. Consider an irreversible unimolecular reaction k. -d[a]/dt = k[a] Can also describe in terms of appearance of B.
![Kinetics. Consider an irreversible unimolecular reaction k. -d[a]/dt = k[a] Can also describe in terms of appearance of B. Kinetics. Consider an irreversible unimolecular reaction k. -d[a]/dt = k[a] Can also describe in terms of appearance of B.](/thumbs/72/67129702.jpg) Kinetic data gives insight into reaction mechanisms kinetic analysis will describe a relationship between concentrations of all chemical species before the rate determining step in a given reaction and
Kinetic data gives insight into reaction mechanisms kinetic analysis will describe a relationship between concentrations of all chemical species before the rate determining step in a given reaction and
EXAMINATION 2 Chemistry 3A
 000402 1 EXAMINATION 2 Chemistry 3A Name: Print first name before second! Use capital letters! SID #: Peter Vollhardt April 11, 2017 GSI (if you are taking Chem 3AL): Please provide the following information
000402 1 EXAMINATION 2 Chemistry 3A Name: Print first name before second! Use capital letters! SID #: Peter Vollhardt April 11, 2017 GSI (if you are taking Chem 3AL): Please provide the following information
Week 4. Even harder stuff!
 Week 4 Even harder stuff! Focus: SN1 and SN2 Two organic reactions Learn about two basic pathways for how these reactions happen Focus on stereochemistry Focus: SN1 and SN2 You need a couple of things
Week 4 Even harder stuff! Focus: SN1 and SN2 Two organic reactions Learn about two basic pathways for how these reactions happen Focus on stereochemistry Focus: SN1 and SN2 You need a couple of things
Principles of Enzyme Catalysis Arthur L. Haas, Ph.D. Department of Biochemistry and Molecular Biology
 Principles of Enzyme Catalysis Arthur L. Haas, Ph.D. Department of Biochemistry and Molecular Biology Review: Garrett and Grisham, Enzyme Specificity and Regulation (Chapt. 13) and Mechanisms of Enzyme
Principles of Enzyme Catalysis Arthur L. Haas, Ph.D. Department of Biochemistry and Molecular Biology Review: Garrett and Grisham, Enzyme Specificity and Regulation (Chapt. 13) and Mechanisms of Enzyme
Atoms, Elements, Atoms, Elements, Compounds and Mixtures. Compounds and Mixtures. Atoms and the Periodic Table. Atoms and the.
 Atoms, Elements, Compounds and Mixtures Explain how fractional distillation can be used to separate a mixture. 1 Atoms, Elements, Compounds and Mixtures Fractional distillation is used to separate components
Atoms, Elements, Compounds and Mixtures Explain how fractional distillation can be used to separate a mixture. 1 Atoms, Elements, Compounds and Mixtures Fractional distillation is used to separate components
CH 253, Fall Question Points Possible Points Received
 CH 253, Fall 2000 Exam #3 - M. Schwartz November 17, 2000 Name (Print Clearly) Question Points Possible Points Received 1 20 2 15 3 8 4 5 5 4 6 6 7 14 8 8 9 5 10 21 Subtotal 106 Extra Credit 2 Total 108
CH 253, Fall 2000 Exam #3 - M. Schwartz November 17, 2000 Name (Print Clearly) Question Points Possible Points Received 1 20 2 15 3 8 4 5 5 4 6 6 7 14 8 8 9 5 10 21 Subtotal 106 Extra Credit 2 Total 108
Chapter 8. Substitution reactions of Alkyl Halides
 Chapter 8. Substitution reactions of Alkyl Halides There are two types of possible reaction in organic compounds in which sp 3 carbon is bonded to an electronegative atom or group (ex, halides) 1. Substitution
Chapter 8. Substitution reactions of Alkyl Halides There are two types of possible reaction in organic compounds in which sp 3 carbon is bonded to an electronegative atom or group (ex, halides) 1. Substitution
6. Reaction Chemistry
 6. Reaction Chemistry 6.1 Chemical Elements 6.2 Chemical Bonding 6.3 Chemical Reactions 6.4 Thermodynamics 6.5 Properties of Water 6.6 Important Biomolecules 6.1 Chemical Elements It is common for elements
6. Reaction Chemistry 6.1 Chemical Elements 6.2 Chemical Bonding 6.3 Chemical Reactions 6.4 Thermodynamics 6.5 Properties of Water 6.6 Important Biomolecules 6.1 Chemical Elements It is common for elements
Chem 3719 Klein Chapter Practice Problems
 Chem 379 Klein Chapter Practice Problems Dr. Peter Norris, 208 Klein Chapter Problems : Review of General Chemistry. Draw viable structures for molecules with the following molecular formulae. Remember
Chem 379 Klein Chapter Practice Problems Dr. Peter Norris, 208 Klein Chapter Problems : Review of General Chemistry. Draw viable structures for molecules with the following molecular formulae. Remember
Essential Organic Chemistry. Chapter 9
 Essential Organic Chemistry Paula Yurkanis Bruice Chapter 9 Substitution and Elimination Reactions of Alkyl Halides 9.1 How Alkyl Halides React Substitution Reactions One group takes the place of another.
Essential Organic Chemistry Paula Yurkanis Bruice Chapter 9 Substitution and Elimination Reactions of Alkyl Halides 9.1 How Alkyl Halides React Substitution Reactions One group takes the place of another.
MODULE No. 24: Solution Kinetics Part - III
 Subject Paper No and Title Module No and Title Module Tag 6 and PHYSICAL CHEMISTRY-II (Statistical 24 and Solution Kinetics - III CHE_P6_M24 TABLE OF CONTENTS 1. Learning outcomes 2. Introduction 3. Primary
Subject Paper No and Title Module No and Title Module Tag 6 and PHYSICAL CHEMISTRY-II (Statistical 24 and Solution Kinetics - III CHE_P6_M24 TABLE OF CONTENTS 1. Learning outcomes 2. Introduction 3. Primary
Organic Reactions Susbstitution S N. Dr. Sapna Gupta
 Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
Chapter 9. Nucleophilic Substitution and ß-Elimination
 Chapter 9 Nucleophilic Substitution and ß-Elimination Nucleophilic Substitution Nucleophile: From the Greek meaning nucleus loving. A molecule or ion that donates a pair of electrons to another atom or
Chapter 9 Nucleophilic Substitution and ß-Elimination Nucleophilic Substitution Nucleophile: From the Greek meaning nucleus loving. A molecule or ion that donates a pair of electrons to another atom or
Mechanism: We therefore describe the mechanism as a S N 2 mechanism.
 OH Mechanism: This is known as a bimolecular reaction as there are 2 molecules involved in the slow step. We therefore describe the mechanism as a S N 2 mechanism. We see an inversion in this process around
OH Mechanism: This is known as a bimolecular reaction as there are 2 molecules involved in the slow step. We therefore describe the mechanism as a S N 2 mechanism. We see an inversion in this process around
Lab Activity 9: Introduction to Organic Chemical Reactivity, Lab 5 Prelab, Reflux
 Lab Activity 9: Introduction to Organic Chemical Reactivity, Lab 5 Prelab, Reflux Objectives 1. Identify structural features (pi bonds, bond polarity, lone pairs) of a compound 2. Determine whether a structural
Lab Activity 9: Introduction to Organic Chemical Reactivity, Lab 5 Prelab, Reflux Objectives 1. Identify structural features (pi bonds, bond polarity, lone pairs) of a compound 2. Determine whether a structural
CD Basis Set of Spectra that is used is that derived from comparing the spectra of globular proteins whose secondary structures are known from X-ray
 CD Basis Set of Spectra that is used is that derived from comparing the spectra of globular proteins whose secondary structures are known from X-ray crystallography An example of the use of CD Modeling
CD Basis Set of Spectra that is used is that derived from comparing the spectra of globular proteins whose secondary structures are known from X-ray crystallography An example of the use of CD Modeling
Course Information. Instructor Information
 Jordan University of Science and Technology Department of Chemistry Course Syllabus Fall 2018/2019 Course Information Course Number: CHEM 108 Course Name: General and Organic Chemistry Credit Hours: 4
Jordan University of Science and Technology Department of Chemistry Course Syllabus Fall 2018/2019 Course Information Course Number: CHEM 108 Course Name: General and Organic Chemistry Credit Hours: 4
Initials: 1. Chem 633: Advanced Organic Chemistry 2016 Final Exam
 Initials: 1 ame: Chem 633: Advanced rganic Chemistry 2016 Final Exam This exam is closed note, closed book. Please answer the following questions clearly and concisely. In general, use pictures and less
Initials: 1 ame: Chem 633: Advanced rganic Chemistry 2016 Final Exam This exam is closed note, closed book. Please answer the following questions clearly and concisely. In general, use pictures and less
c cm 3 d. a, b, and c c ng d g
 Mr. Stone Honors Biology Practice Test 1. Which one is equal to 1.5 10 3 ml? a. 1.5 10-1 L b. 150 10 0 ml c. 1.5 10 3 cm 3 d. a, b, and c e. b and c only 2. Which one is NOT equal to 2.74 10 2 milligrams?
Mr. Stone Honors Biology Practice Test 1. Which one is equal to 1.5 10 3 ml? a. 1.5 10-1 L b. 150 10 0 ml c. 1.5 10 3 cm 3 d. a, b, and c e. b and c only 2. Which one is NOT equal to 2.74 10 2 milligrams?
What comes now. Remember general trends and Periodic Table. Chapter 8 Hydrogen. CHEM 462 Wednesday, Nov. 3 T. Hughbanks
 Chapter 8 Hydrogen CHEM 462 Wednesday, Nov. 3 T. Hughbanks What comes now From this point on, we will be discussing more descriptive chemistry - this requires integration of the principles covered up to
Chapter 8 Hydrogen CHEM 462 Wednesday, Nov. 3 T. Hughbanks What comes now From this point on, we will be discussing more descriptive chemistry - this requires integration of the principles covered up to
1 Chemistry Notes Dr. Reeves Science Class (This was me when I had hair.)
 1 Chemistry Notes Dr. Reeves Science Class (This was me when I had hair.) Table of Contents Introduction Slide 5 Topics of Discussion Slide 6 Periodic Table Slide 10 Elements Slide 15 Chemical Formulas
1 Chemistry Notes Dr. Reeves Science Class (This was me when I had hair.) Table of Contents Introduction Slide 5 Topics of Discussion Slide 6 Periodic Table Slide 10 Elements Slide 15 Chemical Formulas
initial rate M s x 10-6 [B] M x x 10-6
![initial rate M s x 10-6 [B] M x x 10-6 initial rate M s x 10-6 [B] M x x 10-6](/thumbs/86/93476064.jpg) 1. Consider the reaction below: C 6 H 12 O 6 (s) + 12 O 2 (g) 6 CO 2 (g) + 6 H 2 O(g) Which of the following is an incorrect expression of the rate? 1. rate = (Δ[H 2 O] / 6 Δt) 2. rate = -(Δ[O 2 ] / 12
1. Consider the reaction below: C 6 H 12 O 6 (s) + 12 O 2 (g) 6 CO 2 (g) + 6 H 2 O(g) Which of the following is an incorrect expression of the rate? 1. rate = (Δ[H 2 O] / 6 Δt) 2. rate = -(Δ[O 2 ] / 12
Dental Biochemistry EXAM I
 Dental Biochemistry EXAM I August 29, 2005 In the reaction below: CH 3 -CH 2 OH -~ ethanol CH 3 -CHO acetaldehyde A. acetoacetate is being produced B. ethanol is being oxidized to acetaldehyde C. acetaldehyde
Dental Biochemistry EXAM I August 29, 2005 In the reaction below: CH 3 -CH 2 OH -~ ethanol CH 3 -CHO acetaldehyde A. acetoacetate is being produced B. ethanol is being oxidized to acetaldehyde C. acetaldehyde
Chemistry Final Study Guide KEY. 3. Define physical changes. A change in any physical property of a substance, not in the substance itself.
 Chemistry Final Study Guide KEY Unit 2: Matter & Its Properties, Lesson 1: Physical and Chemical Properties & Changes 1. Define physical properties. The characteristics of a substance that can be observed
Chemistry Final Study Guide KEY Unit 2: Matter & Its Properties, Lesson 1: Physical and Chemical Properties & Changes 1. Define physical properties. The characteristics of a substance that can be observed
Due Date: First day of school if you miss the first day of school, you must send a scanned/pdf copy to Mr. Mejia:
 Name: Date: AP Chemistry Summer Assignment Due Date: First day of school if you miss the first day of school, you must send a scanned/pdf copy to Mr. Mejia: jmejia@cboek12.org Assessment: Within the first
Name: Date: AP Chemistry Summer Assignment Due Date: First day of school if you miss the first day of school, you must send a scanned/pdf copy to Mr. Mejia: jmejia@cboek12.org Assessment: Within the first
Chemistry 5.07SC Biological Chemistry I Fall Semester, 2013
 Chemistry 5.07SC Biological Chemistry I Fall Semester, 2013 Lecture 9 Biochemical Transformations I. Carbon-carbon bond forming and cleaving reactions in Biology (see the Lexicon). Enzymes catalyze a limited
Chemistry 5.07SC Biological Chemistry I Fall Semester, 2013 Lecture 9 Biochemical Transformations I. Carbon-carbon bond forming and cleaving reactions in Biology (see the Lexicon). Enzymes catalyze a limited
Multiple Choice Identify the letter of the choice that best completes the statement or answers the question.
 CP Chem Review 2 Matching Match each item with the correct statement below. a. activated complex d. activation energy b. reaction rate e. free energy c. inhibitor 1. the minimum energy colliding particles
CP Chem Review 2 Matching Match each item with the correct statement below. a. activated complex d. activation energy b. reaction rate e. free energy c. inhibitor 1. the minimum energy colliding particles
Lec.1 Chemistry Of Water
 Lec.1 Chemistry Of Water Biochemistry & Medicine Biochemistry can be defined as the science concerned with the chemical basis of life. Biochemistry can be described as the science concerned with the chemical
Lec.1 Chemistry Of Water Biochemistry & Medicine Biochemistry can be defined as the science concerned with the chemical basis of life. Biochemistry can be described as the science concerned with the chemical
BIOCHEMISTRY NOTES - UNIT 2-
 BIOCHEMISTRY NOTES - UNIT 2- ATOMS - the basic unit of matter. Contains subatomic particles o (+ charge) o (no charge/neutral) o (- charge) Protons and neutrons have about the same mass. Electrons are
BIOCHEMISTRY NOTES - UNIT 2- ATOMS - the basic unit of matter. Contains subatomic particles o (+ charge) o (no charge/neutral) o (- charge) Protons and neutrons have about the same mass. Electrons are
Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions
 Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions Halogen compounds are important for several reasons. Simple alkyl and aryl halides, especially chlorides and bromides, are versatile
Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions Halogen compounds are important for several reasons. Simple alkyl and aryl halides, especially chlorides and bromides, are versatile
10. Organohalides. Based on McMurry s Organic Chemistry, 7 th edition
 10. Organohalides Based on McMurry s Organic Chemistry, 7 th edition What Is an Alkyl Halide An organic compound containing at least one carbonhalogen bond (C-X) X (F, Cl, Br, I) replaces H Can contain
10. Organohalides Based on McMurry s Organic Chemistry, 7 th edition What Is an Alkyl Halide An organic compound containing at least one carbonhalogen bond (C-X) X (F, Cl, Br, I) replaces H Can contain
Analyzing Ion channel Simulations
 Analyzing Ion channel Simulations (Neher and Sakmann, Scientific American 1992) Single channel current (Heurteaux et al, EMBO 2004) Computational Patch Clamp (Molecular Dynamics) Atoms move according to
Analyzing Ion channel Simulations (Neher and Sakmann, Scientific American 1992) Single channel current (Heurteaux et al, EMBO 2004) Computational Patch Clamp (Molecular Dynamics) Atoms move according to
LAB 05 Enzyme Action
 LAB 05 Enzyme Action Objectives: Name the substrate and products of the peroxidase-catalyzed reaction. To understand the terms: enzyme, activation energy, active site, ph, and denaturation. Distinguish
LAB 05 Enzyme Action Objectives: Name the substrate and products of the peroxidase-catalyzed reaction. To understand the terms: enzyme, activation energy, active site, ph, and denaturation. Distinguish
Problem Set 1
 2006 7.012 Problem Set 1 Due before 5 PM on FRIDAY, September 15, 2006. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. For each of the following parts, pick
2006 7.012 Problem Set 1 Due before 5 PM on FRIDAY, September 15, 2006. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. For each of the following parts, pick
Chapter-2 (Page 22-37) Physical and Chemical Properties of Water
 Chapter-2 (Page 22-37) Physical and Chemical Properties of Water Introduction About 70% of the mass of the human body is water. Water is central to biochemistry for the following reasons: 1- Biological
Chapter-2 (Page 22-37) Physical and Chemical Properties of Water Introduction About 70% of the mass of the human body is water. Water is central to biochemistry for the following reasons: 1- Biological
Chem ORGANIC CHEMISTRY I
 ORGANIC CHEMISTRY I CHEM 221 /4 02 Final Examination April 20, 2005 0900-1200 Dr. Cerrie ROGERS x x periodic table & pk a data table provided non-programmable calculators allowed molecular model kits allowed
ORGANIC CHEMISTRY I CHEM 221 /4 02 Final Examination April 20, 2005 0900-1200 Dr. Cerrie ROGERS x x periodic table & pk a data table provided non-programmable calculators allowed molecular model kits allowed
for sodium ion (Na + )
 3.4 Unit 2 Chemistry 2 Throughout this unit candidates will be expected to write word equations for reactions specified. Higher tier candidates will also be expected to write and balance symbol equations
3.4 Unit 2 Chemistry 2 Throughout this unit candidates will be expected to write word equations for reactions specified. Higher tier candidates will also be expected to write and balance symbol equations
WESTLAKE HIGH SCHOOL BIOLOGY SUMMER ASSIGNMENT
 WESTLAKE HIGH SCHOOL BIOLOGY SUMMER ASSIGNMENT Dear Future Biology Student, Hello! The Biology Team at Westlake High School is pleased to know that you are going to be a part of Biology in August! This
WESTLAKE HIGH SCHOOL BIOLOGY SUMMER ASSIGNMENT Dear Future Biology Student, Hello! The Biology Team at Westlake High School is pleased to know that you are going to be a part of Biology in August! This
Chapter 14: Chemical Kinetics II. Chem 102 Dr. Eloranta
 Chapter 14: Chemical Kinetics II Chem 102 Dr. Eloranta Rate Laws If you are familiar with calculus Experiments would allow you to determine the reaction order and rate constant, but what if you wanted
Chapter 14: Chemical Kinetics II Chem 102 Dr. Eloranta Rate Laws If you are familiar with calculus Experiments would allow you to determine the reaction order and rate constant, but what if you wanted
5. Draw the alkene that gives the product shown, and specify its stereochemistry. (2 points)
 JSPERSE HEM 350 TEST 3 VERSIN 1 h. 7 Structure and Synthesis of lkenes h. 8 Reactions of lkenes 1 1. How many elements of unsaturation are in the formula 6 H 9 N 2? (3 points) a. 0 b. 1 c. 2 d. 3 e. 4
JSPERSE HEM 350 TEST 3 VERSIN 1 h. 7 Structure and Synthesis of lkenes h. 8 Reactions of lkenes 1 1. How many elements of unsaturation are in the formula 6 H 9 N 2? (3 points) a. 0 b. 1 c. 2 d. 3 e. 4
Stereochemistry. In organic chemistry, subtle differences in spatial arrangements can give rise to prominent effects.
 Stereochemistry This is study of the 3 dimensional arrangement in space of molecules. In organic chemistry, subtle differences in spatial arrangements can give rise to prominent effects. E.g. the isomers
Stereochemistry This is study of the 3 dimensional arrangement in space of molecules. In organic chemistry, subtle differences in spatial arrangements can give rise to prominent effects. E.g. the isomers
Computational Studies of the Photoreceptor Rhodopsin. Scott E. Feller Wabash College
 Computational Studies of the Photoreceptor Rhodopsin Scott E. Feller Wabash College Rhodopsin Photocycle Dark-adapted Rhodopsin hn Isomerize retinal Photorhodopsin ~200 fs Bathorhodopsin Meta-II ms timescale
Computational Studies of the Photoreceptor Rhodopsin Scott E. Feller Wabash College Rhodopsin Photocycle Dark-adapted Rhodopsin hn Isomerize retinal Photorhodopsin ~200 fs Bathorhodopsin Meta-II ms timescale
Chem 112A: Final Exam
 Chem 112A: Final Exam December 14th, 2011 Please provide all answers in the spaces provided. You are not allowed to use a calculator for this exam, but you may use molecular model kits. nly cyclohexane
Chem 112A: Final Exam December 14th, 2011 Please provide all answers in the spaces provided. You are not allowed to use a calculator for this exam, but you may use molecular model kits. nly cyclohexane
Chemical Principles for Biochemistry
 Although this is an introductory course in biochemistry, a certain background in chemistry is assumed. This section outlines certain principles and enumerates those terms and structures it is assumed that
Although this is an introductory course in biochemistry, a certain background in chemistry is assumed. This section outlines certain principles and enumerates those terms and structures it is assumed that
Reaction mechanism 1/17/19. HCl. HCl. 3 radical can form here. Br 2. hint!
 If the reagents or reaction conditions favor radicals, then it is likely to be a radical reaction. Look for hints like peroxide, AIBN, NBS, hn (UV light). 3 radical can form here Reaction mechanism How
If the reagents or reaction conditions favor radicals, then it is likely to be a radical reaction. Look for hints like peroxide, AIBN, NBS, hn (UV light). 3 radical can form here Reaction mechanism How
Lecture 11 - Stoichiometry. Lecture 11 - Introduction. Lecture 11 - The Mole. Lecture 11 - The Mole. Lecture 11 - The Mole
 Chem 103, Section F0F Unit IV - Stoichiometry of Formulas and Equations Lecture 11 The concept of a mole, which is a very large group of atoms or molecules Determining the formulas for a compound Stoichiometry
Chem 103, Section F0F Unit IV - Stoichiometry of Formulas and Equations Lecture 11 The concept of a mole, which is a very large group of atoms or molecules Determining the formulas for a compound Stoichiometry
Advanced Inorganic Chemistry. Alireza Gorji Department of Chemistry, Yazd University. Introduction.
 Advanced Inorganic Chemistry Alireza Gorji Department of Chemistry, Yazd University Introduction 2 1 Kinetics vs. Thermodynamics Thermodynamics Kinetics G = H -T S G = H -T S G = -RTlnK G= -RTlnk Large
Advanced Inorganic Chemistry Alireza Gorji Department of Chemistry, Yazd University Introduction 2 1 Kinetics vs. Thermodynamics Thermodynamics Kinetics G = H -T S G = H -T S G = -RTlnK G= -RTlnk Large
PAPER No. : 5; Organic Chemistry-II MODULE No. : 13; Mixed S N 1 and S N 2 Reactions
 Subject Chemistry Paper No and Title Module No and Title Module Tag 5; Organic Chemistry-II 13; Mixed S N 1 and S N 2 Reactions CHE_P5_M13 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction 3. Nature
Subject Chemistry Paper No and Title Module No and Title Module Tag 5; Organic Chemistry-II 13; Mixed S N 1 and S N 2 Reactions CHE_P5_M13 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction 3. Nature
Saba Al Fayoumi. Tamer Barakat. Dr. Mamoun Ahram + Dr. Diala Abu-Hassan
 1 Saba Al Fayoumi Tamer Barakat Dr. Mamoun Ahram + Dr. Diala Abu-Hassan What is BIOCHEMISTRY??? Biochemistry = understanding life Chemical reactions are what makes an organism (An organism is simply atoms
1 Saba Al Fayoumi Tamer Barakat Dr. Mamoun Ahram + Dr. Diala Abu-Hassan What is BIOCHEMISTRY??? Biochemistry = understanding life Chemical reactions are what makes an organism (An organism is simply atoms
Name Date. Chapter 2 - Chemistry Guide Microbiology (MCB 2010C) Part 1
 Name Date Chapter 2 - Chemistry Guide Microbiology (MCB 2010C) Part 1 The study of biology in the 21 st century is actually the study of biochemistry. In order to be successful in this course, it is important
Name Date Chapter 2 - Chemistry Guide Microbiology (MCB 2010C) Part 1 The study of biology in the 21 st century is actually the study of biochemistry. In order to be successful in this course, it is important
enol unstable; can't be isolated
 Name 1 ke alkenes, alkynes are stabilized by alkyl groups, so internal alkynes (i.e. disubstituted alkynes) are generally more stable than terminal alkynes (i.e. monosubstituted alkynes). With this in
Name 1 ke alkenes, alkynes are stabilized by alkyl groups, so internal alkynes (i.e. disubstituted alkynes) are generally more stable than terminal alkynes (i.e. monosubstituted alkynes). With this in
Chemistry 2000 Lecture 14: Redox reactions
 Chemistry 2000 Lecture 14: Redox reactions Marc R. Roussel February 8, 2018 Marc R. Roussel Chemistry 2000 Lecture 14: Redox reactions February 8, 2018 1 / 12 Review: Oxidation states The oxidation state
Chemistry 2000 Lecture 14: Redox reactions Marc R. Roussel February 8, 2018 Marc R. Roussel Chemistry 2000 Lecture 14: Redox reactions February 8, 2018 1 / 12 Review: Oxidation states The oxidation state
