Selective Paraffin Removal from Olefin/Paraffin Mixtures by Adsorption to Aluminum Methylphosphate-α: A Molecular Simulation Study
|
|
|
- Marjory Harvey
- 5 years ago
- Views:
Transcription
1 Selectve Paraffn Removal from Olefn/Paraffn Mxtures by Adsorpton to Alumnum Methylphosphate-α: A Molecular Smulaton Study Maake C. Kroon 1,3, Carmelo Herdes and Lourdes F. Vega,3* 1 Process Equpment, Department of Process & Energy, Delft Unversty of Technology, Leeghwaterstraat 44, 68 CA Delft, The Netherlands Insttut de Cenca de Materals de Barcelona, Conseo Superor de Investgacones Centífcas, Campus de la UAB, 8193 Bellaterra, Barcelona, Span 3 MATGAS Research Center and Ar Products and Chemcals, Campus de la UAB, 8193 Bellaterra, Barcelona, Span E-mal: vegal@matgas.com; fax: In ths work the selectve paraffn removal from olefn/paraffn mxtures by adsorpton to the hybrd norganc-organc materal alumnum methylphosphonate polymorph alpha s nvestgated usng Grand Canoncal Monte Carlo smulatons. Ths materal selectvely adsorbs the paraffn from ethane/ethylene mxtures as well as ethyl chlorde/vnyl chlorde mxtures. Therefore, the key factor determnng the selectvty s found to be the molecular nteracton between the methyl group of ths materal and the methyl group of the paraffn. Introducton The olefn/paraffn separaton s the most energy ntensve separaton carred out n the petrochemcal ndustry. Currently, hgh-pressure dstllaton s used as purfcaton method, whch s energy-ntensve and expensve. A cheaper, more effcent, less energy-ntensve and cleaner method for separatng olefns from paraffns would be very desrable. One of these alternatve separaton methods s adsorpton, whch s stll n the research-phase. Commonly, t s the olefn that s preferentally adsorbed usng ths separaton method, because of the strong nteracton between the unsaturated bond of the olefn and a metal on on the surface of the adsorbent materal, formng a π-complex. Because the olefn s preferably adsorbed, t s the paraffn that s easly obtaned n ts pure form. Unfortunately, n ndustral processes the desred product s the olefn (raw materal for polymers) and not the paraffn. Therefore, adsorpton s not an opton to replace the current dstllaton columns (yet). However, when t s the paraffn that s selectvely adsorbed, the olefn can be easly obtaned n ts pure form, whch would make the adsorpton process a very attractve alternatve. Recently, for the frst tme, an adsorpton materal was found that was able to selectvely adsorb the paraffn nstead of the olefn. Ths research nvolved the selectve absorpton of ethyl chlorde compared to vnyl chlorde usng the hybrd norganc-organc materal alumnum methylphosphonate polymorph alpha (AlMePO-α) [1]. The study was performed by a combned expermental-molecular smulaton approach. However, t s stll unclear why AlMePO-α selectvely adsorbs the ethyl chlorde compared to the vnyl chlorde, and whether ths materal s also suted for other olefn/paraffn separatons. Here, we wll use molecular smulatons n order to understand the key factors determnng the selectve paraffn adsorpton by AlMePO-α (molecular nteractons, sterc effects, rgdty) and to nvestgate the sutablty of ths materal for other olefn/paraffn separatons. The molecular model used for the materal s the same developed n prevous work [], whle approprate force felds are 1
2 used for the dfferent olefns and paraffns under consderaton. As an example, the selectve adsorpton of ethane from ethane/ethylene mxtures by AlMePO-α s nvestgated. Methodology Molecular models: The AlMePO-α materal s modeled as consstng of sngle Lennard-Jones spheres for the ndvdual atoms, where the Lennard-Jones parameters of the Al, P and O atoms were taken from Schumacher et al. [3] and the methyl groups are represented as sngle Lennard-Jones stes located at the postons of the C-atom []. The parameters used n ths work are gven n table 1. The smulaton cell conssts of 96 alumnum atoms, 144 phosphorus atoms, 43 oxygen atoms, and 144 methyl groups. Table 1: Lennard-Jones parameters of AlMePO-α used n the smulatons [,3] Ste σ (nm) ε/k B (K) Al - - P - - O CH Both ethane and ethylene are modeled usng three dfferent force felds: two unted-atom molecular models.e. the OPLS-UA model [4] and the TraPPE model [5], and a two-center Lennard-Jones force feld wth pont quadrupole potental.e. the CLJQ model [6,7]. The frst two models wth fxed bond lengths (l ) between the unted-atom groups (CH 3 or CH ) only use the Lennard-Jones ntermolecular potental (φ LJ ) for the non-bonded nteractons, where ε/k B and σ are model parameters [4,5]: 1 6 σ = = σ φ LJ = 4ε (1) 1 1 r r The CLJQ model descrbes the total flud-flud potental as a sum of the Lennard-Jones nteractons (φ LJ ) and the quadrupolar nteractons (φ QQ ), where the quadrupole moment of the flud (Q) s an addtonal model parameter located at the geometrc center of the molecule: 3 Q φ = [ 1 5( c + c ) 15c c + ( c 5c c ) ] () QQ wth: 4 ( 4πε ) r 5 ab c = cosθ (3) c = cosθ (4) c = cos θ cosθ + snθ snθ cosφ (5)
3 where and refer to the LJ stes on the molecules, r s the dstance between the centers of molecules and, r ab s the center-to-center dstance amongst molecules, θ and θ are the polar angles of the molecular axs wth respect to a lne onng the molecular centers, φ s the dfference n azmuthal angles and ε s the vacuum permttvty. The LJ parameters for the CH 3 groups of ethane and the CH groups of ethylene for the three dfferent models and the quadrupole moment for the CLJQ model are shown n table. Table : Force felds and parameters for ethane and ethylene used n the smulatons [4-7] Component Force feld Parameters ε/k B (K) σ (Å) l (Å) Q (B) Ethane OPLS-UA TraPPE CLJQ Ethylene OPLS-UA TraPPE CLJQ Smulaton detals: Wth Grand Canoncal Monte Carlo (GCMC) smulatons, the number of molecules adsorbed versus the actvty s obtaned. In order to change the actvty nto pressure, the vral equaton of state n terms of actvty s used [8]: p p = ξξ + ( ρ ξ ) ( ρ ) + ξ ξ ξ (6) where ξ and ρ are the actvty and densty of the saturaton state pont of pure ethane or ethylene at the needed temperature. The state ponts were calculated from bulk smulatons and can be found n table 3. Table 3: State ponts of ethane and ethylene at dfferent temperatures Component T (K) P (kpa) ρ (mol/l) ξ (Å -3 ) Ethane Ethylene The postons of the atoms of the adsorbent materal were fxed durng the smulatons. Ths mples that the sold-sold nteractons do not affect the calculatons, as they cancel out when energetcally comparng smulated confguratons. Hence, only sold-flud and flud-flud nteractons were calculated. The Lorentz-Berthelot mxng rules were used to calculate the Lennard-Jones parameters between stes of dfferent types. The smulatons requred x1 7 confguratons to reach the equlbrum. Average propertes were calculated over blocks wth 5x1 5 confguratons once the equlbrum was reached. The flud-flud potental was cut at r c = 6 σ ff as recommended by Duque and Vega [9]. 3
4 In order to compare wth prevous expermental and smulaton data on ethyl chlorde/vnyl chlorde adsorpton, the number of ethane/ethylene molecules adsorbed was converted to the amount of ethane/ethylene adsorbed on AlMePO-α n excess to what s present n the bulk. Results and Dscusson In fgure 1 the smulated sotherms for ethane wth the three dfferent models are shown at, and. The error bars are so small that they fall wthn the symbols representng the data ponts. It can be seen that the three models gve smlar results. OPLS-UA TraPPE CLJQ Fgure 1: Adsorpton sotherms of ethane n AlMePO-α at, and usng the OPLS- UA model, thetrappe model and the CLJQ model Fgure shows the smulated sotherms for ethylene wth the three dfferent models at K, and. Agan, the three models gve smlar results. OPLS-UA TraPPE CLJQ Fgure : Adsorpton sotherms of ethylene n AlMePO-α at, and usng the OPLS- UA model, thetrappe model and the CLJQ model When comparng fgures 1 and, t can be notced that the adsorpton of pure ethane s always hgher than the adsorpton of pure ethylene, so preferental adsorpton of the paraffn by AlMePO-α s also observed for ethane/ethylene mxtures. Fgure 3 compares the adsorpton of ethane and ethylene at, and usng the OPLS-UA model. It can be notced that the adsorpton of both ethane and ethylene s hgher at lower temperatures, whereas the selectvty to ethane (=paraffn) adsorpton s hgher at hgher temperatures. The optmal separaton temperature s therefore a trade-off between a hgher overall adsorpton at lower temperatures and a hgher selectvty at hgher temperatures. 4
5 . ethane ethylene ethane ethylene ethane ethylene Fgure 3: Adsorpton sotherms of pure ethane and ethylene at, and usng the OPLS-UA model The obtaned sotherms for ethane and ethylene were also compared to the sotherms determned prevously for ethyl chlorde and vnyl chlorde [1], whch are shown n fgure 4. It can be seen that the adsorpton of pure ethyl chlorde s also always hgher than the adsorpton of pure vnyl chlorde. Also, the dfference n adsorpton between ethyl chlorde and vnyl chlorde s ncreasng wth ncreasng temperature, although ths dfference seems a lttle larger compared to the dfference n adsorpton between ethane and ethylene. However, the maxmum amount of both ethyl chlorde (1.5 ) and vnyl chlorde (.9 ) adsorbed s lower than the maxmum amount of both ethane (1.4 ) and ethylene (5 ) adsorbed. Thus, both ethane and ethylene adsorb better to AlMePO-α than ethyl chlorde and vnyl chlorde. Fgure 4: Adsorpton sotherms of pure ethylene chlorde and vnyl chlorde at K (left), (mddle) and (rght) [1]. Ethyl chlorde expermental data (o), ethyl chlorde smulated results ( ), vnyl chlorde expermental data ( ), and vnyl chlorde smulated results ( ) Because AlMePO-α shows selectvty for paraffn adsorpton for both ethane/ethylene mxtures and for ethyl chlorde/vnyl chlorde mxtures, t s expected that the molecular nteracton between the materal and the paraffn s the key factor determnng the selectvty and not the sterc effects, as ethyl chlorde and vnyl chlorde are much larger than ethane and ethylene. Ths also follows from the snapshots shows n fgure 5, where the ethane, ethylene, ethyl chlorde and vnyl chlorde molecules arrange exactly n the same way n the materal AlMePO-α, wth the methyl groups of the AlMePO-α drected towards the methyl groups of ethane and ethyl chlorde. 5
6 Fgure 5: Snapshots at maxmum loadng for ethane (upper left), ethylene (below left), ethyl chlorde (upper rght) and vnyl chlorde (below rght) Conclusons The materal AlMePO-α shows selectve paraffn adsorpton for both ethane/ethylene mxtures and ethyl chlorde/vnyl chlorde mxtures. Therefore, the key factor determnng the selectvty s found to be the molecular nteracton between the methyl group of ths materal and the methyl group of the paraffn. Acknowledgements Ths work s partally funded by the Spansh government (CTQ5-96/PPQ) and the Catalan government (SGR5-88). Supercomputng tme from CESCA and MATGAS AIE s also acknowledged. We are grateful to A. Valente, Z. Ln and J.A.P. Couthno for ther contrbuton to ths work and for provdng the expermental data. References [1] Herdes, C., Valente, A., Ln, Z., Rocha, J., Coutnho, J. A. P., Medna, F., Vega, L. F., Langmur, Vol. 3, 7, p. 799 [] Herdes, C., Ln, Z., Valente, A., Coutnho, J. A. P., Vega, L. F., Langmur, Vol., 7, p. 397 [3] Schumacher, C., Gonzalez, J., Wrght, P. A., Seaton, N. A., Phys. Chem. Chem. Phys., Vol. 7, 5, p. 351 [4] Jorgensen, W. L., Madura, J. D., Swenson, C. J., J. Am. Chem. Soc., Vol. 16, 1984, p [5] Martn, M. G., Sepmann, J. I., J. Phys. Chem. B, Vol. 1, 1998, p. 569 [6] Curbelo, S., Müller, E. A., Ads. Sc. Technol., Vol. 3, 5, p. 855 [7] Vrabec, J., Stoll, J., Hasse, H., J. Phys. Chem. B, Vol. 15, 1, p. 116 [8] Hansen, J.-P., McDonald, I. R., Theory of Smple Lquds, nd ed., Academc Press: London, UK, 1986 [9] Duque, D., Vega, L. F., J. Chem. Phys., Vol. 11, 4, p
Assignment 4. Adsorption Isotherms
 Insttute of Process Engneerng Assgnment 4. Adsorpton Isotherms Part A: Compettve adsorpton of methane and ethane In large scale adsorpton processes, more than one compound from a mxture of gases get adsorbed,
Insttute of Process Engneerng Assgnment 4. Adsorpton Isotherms Part A: Compettve adsorpton of methane and ethane In large scale adsorpton processes, more than one compound from a mxture of gases get adsorbed,
modeling of equilibrium and dynamic multi-component adsorption in a two-layered fixed bed for purification of hydrogen from methane reforming products
 modelng of equlbrum and dynamc mult-component adsorpton n a two-layered fxed bed for purfcaton of hydrogen from methane reformng products Mohammad A. Ebrahm, Mahmood R. G. Arsalan, Shohreh Fatem * Laboratory
modelng of equlbrum and dynamc mult-component adsorpton n a two-layered fxed bed for purfcaton of hydrogen from methane reformng products Mohammad A. Ebrahm, Mahmood R. G. Arsalan, Shohreh Fatem * Laboratory
Electrical double layer: revisit based on boundary conditions
 Electrcal double layer: revst based on boundary condtons Jong U. Km Department of Electrcal and Computer Engneerng, Texas A&M Unversty College Staton, TX 77843-318, USA Abstract The electrcal double layer
Electrcal double layer: revst based on boundary condtons Jong U. Km Department of Electrcal and Computer Engneerng, Texas A&M Unversty College Staton, TX 77843-318, USA Abstract The electrcal double layer
Understanding The Solubility Of Gases In Alkyl- Imidazolium Based Ionic Liquids By A Molecular Modelling Approach.
 Understandng The Solublty Of Gases In Alkyl- Imdazolum Based Ionc Lquds By A Molecular Modellng Approach. Jord S. Andreu 1,2, Lourdes F. Vega 1,2 *, 1 Insttut de Cenca de Materals de Barcelona, Consejo
Understandng The Solublty Of Gases In Alkyl- Imdazolum Based Ionc Lquds By A Molecular Modellng Approach. Jord S. Andreu 1,2, Lourdes F. Vega 1,2 *, 1 Insttut de Cenca de Materals de Barcelona, Consejo
Supplementary Notes for Chapter 9 Mixture Thermodynamics
 Supplementary Notes for Chapter 9 Mxture Thermodynamcs Key ponts Nne major topcs of Chapter 9 are revewed below: 1. Notaton and operatonal equatons for mxtures 2. PVTN EOSs for mxtures 3. General effects
Supplementary Notes for Chapter 9 Mxture Thermodynamcs Key ponts Nne major topcs of Chapter 9 are revewed below: 1. Notaton and operatonal equatons for mxtures 2. PVTN EOSs for mxtures 3. General effects
A Modulated Hydrothermal (MHT) Approach for the Facile. Synthesis of UiO-66-Type MOFs
 Supplementary Informaton A Modulated Hydrothermal (MHT) Approach for the Facle Synthess of UO-66-Type MOFs Zhgang Hu, Yongwu Peng, Zx Kang, Yuhong Qan, and Dan Zhao * Department of Chemcal and Bomolecular
Supplementary Informaton A Modulated Hydrothermal (MHT) Approach for the Facle Synthess of UO-66-Type MOFs Zhgang Hu, Yongwu Peng, Zx Kang, Yuhong Qan, and Dan Zhao * Department of Chemcal and Bomolecular
Introduction to Vapor/Liquid Equilibrium, part 2. Raoult s Law:
 CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
One-sided finite-difference approximations suitable for use with Richardson extrapolation
 Journal of Computatonal Physcs 219 (2006) 13 20 Short note One-sded fnte-dfference approxmatons sutable for use wth Rchardson extrapolaton Kumar Rahul, S.N. Bhattacharyya * Department of Mechancal Engneerng,
Journal of Computatonal Physcs 219 (2006) 13 20 Short note One-sded fnte-dfference approxmatons sutable for use wth Rchardson extrapolaton Kumar Rahul, S.N. Bhattacharyya * Department of Mechancal Engneerng,
Grand canonical Monte Carlo simulations of bulk electrolytes and calcium channels
 Grand canoncal Monte Carlo smulatons of bulk electrolytes and calcum channels Thess of Ph.D. dssertaton Prepared by: Attla Malascs M.Sc. n Chemstry Supervsor: Dr. Dezső Boda Unversty of Pannona Insttute
Grand canoncal Monte Carlo smulatons of bulk electrolytes and calcum channels Thess of Ph.D. dssertaton Prepared by: Attla Malascs M.Sc. n Chemstry Supervsor: Dr. Dezső Boda Unversty of Pannona Insttute
CHEMICAL ENGINEERING
 Postal Correspondence GATE & PSUs -MT To Buy Postal Correspondence Packages call at 0-9990657855 1 TABLE OF CONTENT S. No. Ttle Page no. 1. Introducton 3 2. Dffuson 10 3. Dryng and Humdfcaton 24 4. Absorpton
Postal Correspondence GATE & PSUs -MT To Buy Postal Correspondence Packages call at 0-9990657855 1 TABLE OF CONTENT S. No. Ttle Page no. 1. Introducton 3 2. Dffuson 10 3. Dryng and Humdfcaton 24 4. Absorpton
THE EFFECT OF TORSIONAL RIGIDITY BETWEEN ELEMENTS ON FREE VIBRATIONS OF A TELESCOPIC HYDRAULIC CYLINDER SUBJECTED TO EULER S LOAD
 Journal of Appled Mathematcs and Computatonal Mechancs 7, 6(3), 7- www.amcm.pcz.pl p-issn 99-9965 DOI:.75/jamcm.7.3. e-issn 353-588 THE EFFECT OF TORSIONAL RIGIDITY BETWEEN ELEMENTS ON FREE VIBRATIONS
Journal of Appled Mathematcs and Computatonal Mechancs 7, 6(3), 7- www.amcm.pcz.pl p-issn 99-9965 DOI:.75/jamcm.7.3. e-issn 353-588 THE EFFECT OF TORSIONAL RIGIDITY BETWEEN ELEMENTS ON FREE VIBRATIONS
Lecture. Polymer Thermodynamics 0331 L Chemical Potential
 Prof. Dr. rer. nat. habl. S. Enders Faculty III for Process Scence Insttute of Chemcal Engneerng Department of Thermodynamcs Lecture Polymer Thermodynamcs 033 L 337 3. Chemcal Potental Polymer Thermodynamcs
Prof. Dr. rer. nat. habl. S. Enders Faculty III for Process Scence Insttute of Chemcal Engneerng Department of Thermodynamcs Lecture Polymer Thermodynamcs 033 L 337 3. Chemcal Potental Polymer Thermodynamcs
Energy, Entropy, and Availability Balances Phase Equilibria. Nonideal Thermodynamic Property Models. Selecting an Appropriate Model
 Lecture 4. Thermodynamcs [Ch. 2] Energy, Entropy, and Avalablty Balances Phase Equlbra - Fugactes and actvty coeffcents -K-values Nondeal Thermodynamc Property Models - P-v-T equaton-of-state models -
Lecture 4. Thermodynamcs [Ch. 2] Energy, Entropy, and Avalablty Balances Phase Equlbra - Fugactes and actvty coeffcents -K-values Nondeal Thermodynamc Property Models - P-v-T equaton-of-state models -
Economics 101. Lecture 4 - Equilibrium and Efficiency
 Economcs 0 Lecture 4 - Equlbrum and Effcency Intro As dscussed n the prevous lecture, we wll now move from an envronment where we looed at consumers mang decsons n solaton to analyzng economes full of
Economcs 0 Lecture 4 - Equlbrum and Effcency Intro As dscussed n the prevous lecture, we wll now move from an envronment where we looed at consumers mang decsons n solaton to analyzng economes full of
Mass Transfer Processes
 Mass Transfer Processes S. Majd Hassanzadeh Department of Earth Scences Faculty of Geoscences Utrecht Unversty Outlne: 1. Measures of Concentraton 2. Volatlzaton and Dssoluton 3. Adsorpton Processes 4.
Mass Transfer Processes S. Majd Hassanzadeh Department of Earth Scences Faculty of Geoscences Utrecht Unversty Outlne: 1. Measures of Concentraton 2. Volatlzaton and Dssoluton 3. Adsorpton Processes 4.
χ x B E (c) Figure 2.1.1: (a) a material particle in a body, (b) a place in space, (c) a configuration of the body
 Secton.. Moton.. The Materal Body and Moton hyscal materals n the real world are modeled usng an abstract mathematcal entty called a body. Ths body conssts of an nfnte number of materal partcles. Shown
Secton.. Moton.. The Materal Body and Moton hyscal materals n the real world are modeled usng an abstract mathematcal entty called a body. Ths body conssts of an nfnte number of materal partcles. Shown
Solution Thermodynamics
 Soluton hermodynamcs usng Wagner Notaton by Stanley. Howard Department of aterals and etallurgcal Engneerng South Dakota School of nes and echnology Rapd Cty, SD 57701 January 7, 001 Soluton hermodynamcs
Soluton hermodynamcs usng Wagner Notaton by Stanley. Howard Department of aterals and etallurgcal Engneerng South Dakota School of nes and echnology Rapd Cty, SD 57701 January 7, 001 Soluton hermodynamcs
Non-Ideality Through Fugacity and Activity
 Non-Idealty Through Fugacty and Actvty S. Patel Deartment of Chemstry and Bochemstry, Unversty of Delaware, Newark, Delaware 19716, USA Corresondng author. E-mal: saatel@udel.edu 1 I. FUGACITY In ths dscusson,
Non-Idealty Through Fugacty and Actvty S. Patel Deartment of Chemstry and Bochemstry, Unversty of Delaware, Newark, Delaware 19716, USA Corresondng author. E-mal: saatel@udel.edu 1 I. FUGACITY In ths dscusson,
Amplification and Relaxation of Electron Spin Polarization in Semiconductor Devices
 Amplfcaton and Relaxaton of Electron Spn Polarzaton n Semconductor Devces Yury V. Pershn and Vladmr Prvman Center for Quantum Devce Technology, Clarkson Unversty, Potsdam, New York 13699-570, USA Spn Relaxaton
Amplfcaton and Relaxaton of Electron Spn Polarzaton n Semconductor Devces Yury V. Pershn and Vladmr Prvman Center for Quantum Devce Technology, Clarkson Unversty, Potsdam, New York 13699-570, USA Spn Relaxaton
University of Washington Department of Chemistry Chemistry 452/456 Summer Quarter 2014
 Lecture 16 8/4/14 Unversty o Washngton Department o Chemstry Chemstry 452/456 Summer Quarter 214. Real Vapors and Fugacty Henry s Law accounts or the propertes o extremely dlute soluton. s shown n Fgure
Lecture 16 8/4/14 Unversty o Washngton Department o Chemstry Chemstry 452/456 Summer Quarter 214. Real Vapors and Fugacty Henry s Law accounts or the propertes o extremely dlute soluton. s shown n Fgure
Lecture 4. Macrostates and Microstates (Ch. 2 )
 Lecture 4. Macrostates and Mcrostates (Ch. ) The past three lectures: we have learned about thermal energy, how t s stored at the mcroscopc level, and how t can be transferred from one system to another.
Lecture 4. Macrostates and Mcrostates (Ch. ) The past three lectures: we have learned about thermal energy, how t s stored at the mcroscopc level, and how t can be transferred from one system to another.
Equation of State Modeling of Phase Equilibrium in the Low-Density Polyethylene Process
 Equaton of State Modelng of Phase Equlbrum n the Low-Densty Polyethylene Process H. Orbey, C. P. Boks, and C. C. Chen Ind. Eng. Chem. Res. 1998, 37, 4481-4491 Yong Soo Km Thermodynamcs & Propertes Lab.
Equaton of State Modelng of Phase Equlbrum n the Low-Densty Polyethylene Process H. Orbey, C. P. Boks, and C. C. Chen Ind. Eng. Chem. Res. 1998, 37, 4481-4491 Yong Soo Km Thermodynamcs & Propertes Lab.
EPR Paradox and the Physical Meaning of an Experiment in Quantum Mechanics. Vesselin C. Noninski
 EPR Paradox and the Physcal Meanng of an Experment n Quantum Mechancs Vesseln C Nonnsk vesselnnonnsk@verzonnet Abstract It s shown that there s one purely determnstc outcome when measurement s made on
EPR Paradox and the Physcal Meanng of an Experment n Quantum Mechancs Vesseln C Nonnsk vesselnnonnsk@verzonnet Abstract It s shown that there s one purely determnstc outcome when measurement s made on
( ) 1/ 2. ( P SO2 )( P O2 ) 1/ 2.
 Chemstry 360 Dr. Jean M. Standard Problem Set 9 Solutons. The followng chemcal reacton converts sulfur doxde to sulfur troxde. SO ( g) + O ( g) SO 3 ( l). (a.) Wrte the expresson for K eq for ths reacton.
Chemstry 360 Dr. Jean M. Standard Problem Set 9 Solutons. The followng chemcal reacton converts sulfur doxde to sulfur troxde. SO ( g) + O ( g) SO 3 ( l). (a.) Wrte the expresson for K eq for ths reacton.
Kernel Methods and SVMs Extension
 Kernel Methods and SVMs Extenson The purpose of ths document s to revew materal covered n Machne Learnng 1 Supervsed Learnng regardng support vector machnes (SVMs). Ths document also provdes a general
Kernel Methods and SVMs Extenson The purpose of ths document s to revew materal covered n Machne Learnng 1 Supervsed Learnng regardng support vector machnes (SVMs). Ths document also provdes a general
Army Ants Tunneling for Classical Simulations
 Electronc Supplementary Materal (ESI) for Chemcal Scence. Ths journal s The Royal Socety of Chemstry 2014 electronc supplementary nformaton (ESI) for Chemcal Scence Army Ants Tunnelng for Classcal Smulatons
Electronc Supplementary Materal (ESI) for Chemcal Scence. Ths journal s The Royal Socety of Chemstry 2014 electronc supplementary nformaton (ESI) for Chemcal Scence Army Ants Tunnelng for Classcal Smulatons
Structure and Drive Paul A. Jensen Copyright July 20, 2003
 Structure and Drve Paul A. Jensen Copyrght July 20, 2003 A system s made up of several operatons wth flow passng between them. The structure of the system descrbes the flow paths from nputs to outputs.
Structure and Drve Paul A. Jensen Copyrght July 20, 2003 A system s made up of several operatons wth flow passng between them. The structure of the system descrbes the flow paths from nputs to outputs.
) is the unite step-function, which signifies that the second term of the right-hand side of the
 Casmr nteracton of excted meda n electromagnetc felds Yury Sherkunov Introducton The long-range electrc dpole nteracton between an excted atom and a ground-state atom s consdered n ref. [1,] wth the help
Casmr nteracton of excted meda n electromagnetc felds Yury Sherkunov Introducton The long-range electrc dpole nteracton between an excted atom and a ground-state atom s consdered n ref. [1,] wth the help
Dummy variables in multiple variable regression model
 WESS Econometrcs (Handout ) Dummy varables n multple varable regresson model. Addtve dummy varables In the prevous handout we consdered the followng regresson model: y x 2x2 k xk,, 2,, n and we nterpreted
WESS Econometrcs (Handout ) Dummy varables n multple varable regresson model. Addtve dummy varables In the prevous handout we consdered the followng regresson model: y x 2x2 k xk,, 2,, n and we nterpreted
DUE: WEDS FEB 21ST 2018
 HOMEWORK # 1: FINITE DIFFERENCES IN ONE DIMENSION DUE: WEDS FEB 21ST 2018 1. Theory Beam bendng s a classcal engneerng analyss. The tradtonal soluton technque makes smplfyng assumptons such as a constant
HOMEWORK # 1: FINITE DIFFERENCES IN ONE DIMENSION DUE: WEDS FEB 21ST 2018 1. Theory Beam bendng s a classcal engneerng analyss. The tradtonal soluton technque makes smplfyng assumptons such as a constant
π e ax2 dx = x 2 e ax2 dx or x 3 e ax2 dx = 1 x 4 e ax2 dx = 3 π 8a 5/2 (a) We are considering the Maxwell velocity distribution function: 2πτ/m
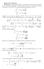 Homework Solutons Problem In solvng ths problem, we wll need to calculate some moments of the Gaussan dstrbuton. The brute-force method s to ntegrate by parts but there s a nce trck. The followng ntegrals
Homework Solutons Problem In solvng ths problem, we wll need to calculate some moments of the Gaussan dstrbuton. The brute-force method s to ntegrate by parts but there s a nce trck. The followng ntegrals
An Interactive Optimisation Tool for Allocation Problems
 An Interactve Optmsaton ool for Allocaton Problems Fredr Bonäs, Joam Westerlund and apo Westerlund Process Desgn Laboratory, Faculty of echnology, Åbo Aadem Unversty, uru 20500, Fnland hs paper presents
An Interactve Optmsaton ool for Allocaton Problems Fredr Bonäs, Joam Westerlund and apo Westerlund Process Desgn Laboratory, Faculty of echnology, Åbo Aadem Unversty, uru 20500, Fnland hs paper presents
Physics 5153 Classical Mechanics. Principle of Virtual Work-1
 P. Guterrez 1 Introducton Physcs 5153 Classcal Mechancs Prncple of Vrtual Work The frst varatonal prncple we encounter n mechancs s the prncple of vrtual work. It establshes the equlbrum condton of a mechancal
P. Guterrez 1 Introducton Physcs 5153 Classcal Mechancs Prncple of Vrtual Work The frst varatonal prncple we encounter n mechancs s the prncple of vrtual work. It establshes the equlbrum condton of a mechancal
Solutions of execrises
 Solutons of execrses 1) XPS spectrum analyss The fgure below shows an XPS spectrum measured on the surface of a clean nsoluble homo-polyether. Usng the formulas and tables n ths document, answer the followng
Solutons of execrses 1) XPS spectrum analyss The fgure below shows an XPS spectrum measured on the surface of a clean nsoluble homo-polyether. Usng the formulas and tables n ths document, answer the followng
Influence Of Operating Conditions To The Effectiveness Of Extractive Distillation Columns
 Influence Of Operatng Condtons To The Effectveness Of Extractve Dstllaton Columns N.A. Vyazmna Moscov State Unversty Of Envrnmental Engneerng, Department Of Chemcal Engneerng Ul. Staraya Basmannaya 21/4,
Influence Of Operatng Condtons To The Effectveness Of Extractve Dstllaton Columns N.A. Vyazmna Moscov State Unversty Of Envrnmental Engneerng, Department Of Chemcal Engneerng Ul. Staraya Basmannaya 21/4,
Lecture 12. Transport in Membranes (2)
 Lecture 12. Transport n embranes (2) odule Flow Patterns - Perfect mxng - Countercurrent flow - Cocurrent flow - Crossflow embrane Cascades External ass-transfer Resstances Concentraton Polarzaton and
Lecture 12. Transport n embranes (2) odule Flow Patterns - Perfect mxng - Countercurrent flow - Cocurrent flow - Crossflow embrane Cascades External ass-transfer Resstances Concentraton Polarzaton and
Constitutive Modelling of Superplastic AA-5083
 TECHNISCHE MECHANIK, 3, -5, (01, 1-6 submtted: September 19, 011 Consttutve Modellng of Superplastc AA-5083 G. Gulano In ths study a fast procedure for determnng the constants of superplastc 5083 Al alloy
TECHNISCHE MECHANIK, 3, -5, (01, 1-6 submtted: September 19, 011 Consttutve Modellng of Superplastc AA-5083 G. Gulano In ths study a fast procedure for determnng the constants of superplastc 5083 Al alloy
Physics 607 Exam 1. ( ) = 1, Γ( z +1) = zγ( z) x n e x2 dx = 1. e x2
 Physcs 607 Exam 1 Please be well-organzed, and show all sgnfcant steps clearly n all problems. You are graded on your wor, so please do not just wrte down answers wth no explanaton! Do all your wor on
Physcs 607 Exam 1 Please be well-organzed, and show all sgnfcant steps clearly n all problems. You are graded on your wor, so please do not just wrte down answers wth no explanaton! Do all your wor on
Department of Statistics University of Toronto STA305H1S / 1004 HS Design and Analysis of Experiments Term Test - Winter Solution
 Department of Statstcs Unversty of Toronto STA35HS / HS Desgn and Analyss of Experments Term Test - Wnter - Soluton February, Last Name: Frst Name: Student Number: Instructons: Tme: hours. Ads: a non-programmable
Department of Statstcs Unversty of Toronto STA35HS / HS Desgn and Analyss of Experments Term Test - Wnter - Soluton February, Last Name: Frst Name: Student Number: Instructons: Tme: hours. Ads: a non-programmable
Brownian-Dynamics Simulation of Colloidal Suspensions with Kob-Andersen Type Lennard-Jones Potentials 1
 Brownan-Dynamcs Smulaton of Collodal Suspensons wth Kob-Andersen Type Lennard-Jones Potentals 1 Yuto KIMURA 2 and Mcho TOKUYAMA 3 Summary Extensve Brownan-dynamcs smulatons of bnary collodal suspenton
Brownan-Dynamcs Smulaton of Collodal Suspensons wth Kob-Andersen Type Lennard-Jones Potentals 1 Yuto KIMURA 2 and Mcho TOKUYAMA 3 Summary Extensve Brownan-dynamcs smulatons of bnary collodal suspenton
Physics 181. Particle Systems
 Physcs 181 Partcle Systems Overvew In these notes we dscuss the varables approprate to the descrpton of systems of partcles, ther defntons, ther relatons, and ther conservatons laws. We consder a system
Physcs 181 Partcle Systems Overvew In these notes we dscuss the varables approprate to the descrpton of systems of partcles, ther defntons, ther relatons, and ther conservatons laws. We consder a system
10.34 Numerical Methods Applied to Chemical Engineering Fall Homework #3: Systems of Nonlinear Equations and Optimization
 10.34 Numercal Methods Appled to Chemcal Engneerng Fall 2015 Homework #3: Systems of Nonlnear Equatons and Optmzaton Problem 1 (30 ponts). A (homogeneous) azeotrope s a composton of a multcomponent mxture
10.34 Numercal Methods Appled to Chemcal Engneerng Fall 2015 Homework #3: Systems of Nonlnear Equatons and Optmzaton Problem 1 (30 ponts). A (homogeneous) azeotrope s a composton of a multcomponent mxture
Electronic Quantum Monte Carlo Calculations of Energies and Atomic Forces for Diatomic and Polyatomic Molecules
 RESERVE HIS SPACE Electronc Quantum Monte Carlo Calculatons of Energes and Atomc Forces for Datomc and Polyatomc Molecules Myung Won Lee 1, Massmo Mella 2, and Andrew M. Rappe 1,* 1 he Maknen heoretcal
RESERVE HIS SPACE Electronc Quantum Monte Carlo Calculatons of Energes and Atomc Forces for Datomc and Polyatomc Molecules Myung Won Lee 1, Massmo Mella 2, and Andrew M. Rappe 1,* 1 he Maknen heoretcal
Supporting Information
 Supportng Informaton Water structure at the ar-aqueous nterface of dvalent caton and ntrate solutons Man Xu, Rck Spnney, Heather C. Allen* allen@chemstry.oho-state.edu Fresnel factors and spectra normalzaton
Supportng Informaton Water structure at the ar-aqueous nterface of dvalent caton and ntrate solutons Man Xu, Rck Spnney, Heather C. Allen* allen@chemstry.oho-state.edu Fresnel factors and spectra normalzaton
KINETICS OF GAS HYDRATE FORMATION FROM PYROLYSIS GAS IN WATER-IN-OIL EMULSION SYSTEM
 Proceedngs of the 7th Internatonal Conference on Gas Hydrates (ICGH 211), Ednburgh, Scotland, Unted Kngdom, July 17-21, 211. KINETICS OF GAS HYDRATE FORMATION FROM PYROLYSIS GAS IN WATER-IN-OIL EMULSION
Proceedngs of the 7th Internatonal Conference on Gas Hydrates (ICGH 211), Ednburgh, Scotland, Unted Kngdom, July 17-21, 211. KINETICS OF GAS HYDRATE FORMATION FROM PYROLYSIS GAS IN WATER-IN-OIL EMULSION
General Thermodynamics for Process Simulation. Dr. Jungho Cho, Professor Department of Chemical Engineering Dong Yang University
 General Thermodynamcs for Process Smulaton Dr. Jungho Cho, Professor Department of Chemcal Engneerng Dong Yang Unversty Four Crtera for Equlbra μ = μ v Stuaton α T = T β α β P = P l μ = μ l1 l 2 Thermal
General Thermodynamcs for Process Smulaton Dr. Jungho Cho, Professor Department of Chemcal Engneerng Dong Yang Unversty Four Crtera for Equlbra μ = μ v Stuaton α T = T β α β P = P l μ = μ l1 l 2 Thermal
EVALUATION OF THE VISCO-ELASTIC PROPERTIES IN ASPHALT RUBBER AND CONVENTIONAL MIXES
 EVALUATION OF THE VISCO-ELASTIC PROPERTIES IN ASPHALT RUBBER AND CONVENTIONAL MIXES Manuel J. C. Mnhoto Polytechnc Insttute of Bragança, Bragança, Portugal E-mal: mnhoto@pb.pt Paulo A. A. Perera and Jorge
EVALUATION OF THE VISCO-ELASTIC PROPERTIES IN ASPHALT RUBBER AND CONVENTIONAL MIXES Manuel J. C. Mnhoto Polytechnc Insttute of Bragança, Bragança, Portugal E-mal: mnhoto@pb.pt Paulo A. A. Perera and Jorge
DESIGN OPTIMIZATION OF CFRP RECTANGULAR BOX SUBJECTED TO ARBITRARY LOADINGS
 Munch, Germany, 26-30 th June 2016 1 DESIGN OPTIMIZATION OF CFRP RECTANGULAR BOX SUBJECTED TO ARBITRARY LOADINGS Q.T. Guo 1*, Z.Y. L 1, T. Ohor 1 and J. Takahash 1 1 Department of Systems Innovaton, School
Munch, Germany, 26-30 th June 2016 1 DESIGN OPTIMIZATION OF CFRP RECTANGULAR BOX SUBJECTED TO ARBITRARY LOADINGS Q.T. Guo 1*, Z.Y. L 1, T. Ohor 1 and J. Takahash 1 1 Department of Systems Innovaton, School
ESA modelling and cycle design
 ESA modellng and cycle desgn WP and WP 5 Unversty of Belgrade MATESA Dssemnaton day, Oslo 16.6.016 Motvaton Develop rgorous 3D models (CFD) to understand the processes, examne the nfluence of condtons
ESA modellng and cycle desgn WP and WP 5 Unversty of Belgrade MATESA Dssemnaton day, Oslo 16.6.016 Motvaton Develop rgorous 3D models (CFD) to understand the processes, examne the nfluence of condtons
Section 8.3 Polar Form of Complex Numbers
 80 Chapter 8 Secton 8 Polar Form of Complex Numbers From prevous classes, you may have encountered magnary numbers the square roots of negatve numbers and, more generally, complex numbers whch are the
80 Chapter 8 Secton 8 Polar Form of Complex Numbers From prevous classes, you may have encountered magnary numbers the square roots of negatve numbers and, more generally, complex numbers whch are the
Lecture 7: Boltzmann distribution & Thermodynamics of mixing
 Prof. Tbbtt Lecture 7 etworks & Gels Lecture 7: Boltzmann dstrbuton & Thermodynamcs of mxng 1 Suggested readng Prof. Mark W. Tbbtt ETH Zürch 13 März 018 Molecular Drvng Forces Dll and Bromberg: Chapters
Prof. Tbbtt Lecture 7 etworks & Gels Lecture 7: Boltzmann dstrbuton & Thermodynamcs of mxng 1 Suggested readng Prof. Mark W. Tbbtt ETH Zürch 13 März 018 Molecular Drvng Forces Dll and Bromberg: Chapters
Week 9 Chapter 10 Section 1-5
 Week 9 Chapter 10 Secton 1-5 Rotaton Rgd Object A rgd object s one that s nondeformable The relatve locatons of all partcles makng up the object reman constant All real objects are deformable to some extent,
Week 9 Chapter 10 Secton 1-5 Rotaton Rgd Object A rgd object s one that s nondeformable The relatve locatons of all partcles makng up the object reman constant All real objects are deformable to some extent,
Phase equilibria for the oxygen-water system up to elevated temperatures and pressures
 Phase equlbra for the oxygen-water system up to elevated temperatures and pressures Xaoyan J 1, 2, Xaohua Lu 2, Jnyue Yan 1,3* 1 Department of Chemcal Engneerng and Technology / Energy Processes, Royal
Phase equlbra for the oxygen-water system up to elevated temperatures and pressures Xaoyan J 1, 2, Xaohua Lu 2, Jnyue Yan 1,3* 1 Department of Chemcal Engneerng and Technology / Energy Processes, Royal
Non-Commercial Use Only
 Plottng P-x-y dagram for bnary system Acetone/water at temperatures 25,100,and 200 C usng UNIFAC method and comparng t wth expermental results. Unfac Method: The UNIFAC method s based on the UNIQUAC equaton,
Plottng P-x-y dagram for bnary system Acetone/water at temperatures 25,100,and 200 C usng UNIFAC method and comparng t wth expermental results. Unfac Method: The UNIFAC method s based on the UNIQUAC equaton,
Lecture 14: Forces and Stresses
 The Nuts and Bolts of Frst-Prncples Smulaton Lecture 14: Forces and Stresses Durham, 6th-13th December 2001 CASTEP Developers Group wth support from the ESF ψ k Network Overvew of Lecture Why bother? Theoretcal
The Nuts and Bolts of Frst-Prncples Smulaton Lecture 14: Forces and Stresses Durham, 6th-13th December 2001 CASTEP Developers Group wth support from the ESF ψ k Network Overvew of Lecture Why bother? Theoretcal
THE VIBRATIONS OF MOLECULES II THE CARBON DIOXIDE MOLECULE Student Instructions
 THE VIBRATIONS OF MOLECULES II THE CARBON DIOXIDE MOLECULE Student Instructons by George Hardgrove Chemstry Department St. Olaf College Northfeld, MN 55057 hardgrov@lars.acc.stolaf.edu Copyrght George
THE VIBRATIONS OF MOLECULES II THE CARBON DIOXIDE MOLECULE Student Instructons by George Hardgrove Chemstry Department St. Olaf College Northfeld, MN 55057 hardgrov@lars.acc.stolaf.edu Copyrght George
On the correction of the h-index for career length
 1 On the correcton of the h-ndex for career length by L. Egghe Unverstet Hasselt (UHasselt), Campus Depenbeek, Agoralaan, B-3590 Depenbeek, Belgum 1 and Unverstet Antwerpen (UA), IBW, Stadscampus, Venusstraat
1 On the correcton of the h-ndex for career length by L. Egghe Unverstet Hasselt (UHasselt), Campus Depenbeek, Agoralaan, B-3590 Depenbeek, Belgum 1 and Unverstet Antwerpen (UA), IBW, Stadscampus, Venusstraat
The optimal delay of the second test is therefore approximately 210 hours earlier than =2.
 THE IEC 61508 FORMULAS 223 The optmal delay of the second test s therefore approxmately 210 hours earler than =2. 8.4 The IEC 61508 Formulas IEC 61508-6 provdes approxmaton formulas for the PF for smple
THE IEC 61508 FORMULAS 223 The optmal delay of the second test s therefore approxmately 210 hours earler than =2. 8.4 The IEC 61508 Formulas IEC 61508-6 provdes approxmaton formulas for the PF for smple
Name: SID: Discussion Session:
 Name: SID: Dscusson Sesson: Chemcal Engneerng Thermodynamcs 141 -- Fall 007 Thursday, November 15, 007 Mdterm II SOLUTIONS - 70 mnutes 110 Ponts Total Closed Book and Notes (0 ponts) 1. Evaluate whether
Name: SID: Dscusson Sesson: Chemcal Engneerng Thermodynamcs 141 -- Fall 007 Thursday, November 15, 007 Mdterm II SOLUTIONS - 70 mnutes 110 Ponts Total Closed Book and Notes (0 ponts) 1. Evaluate whether
Gravitational Acceleration: A case of constant acceleration (approx. 2 hr.) (6/7/11)
 Gravtatonal Acceleraton: A case of constant acceleraton (approx. hr.) (6/7/11) Introducton The gravtatonal force s one of the fundamental forces of nature. Under the nfluence of ths force all objects havng
Gravtatonal Acceleraton: A case of constant acceleraton (approx. hr.) (6/7/11) Introducton The gravtatonal force s one of the fundamental forces of nature. Under the nfluence of ths force all objects havng
Temperature. Chapter Heat Engine
 Chapter 3 Temperature In prevous chapters of these notes we ntroduced the Prncple of Maxmum ntropy as a technque for estmatng probablty dstrbutons consstent wth constrants. In Chapter 9 we dscussed the
Chapter 3 Temperature In prevous chapters of these notes we ntroduced the Prncple of Maxmum ntropy as a technque for estmatng probablty dstrbutons consstent wth constrants. In Chapter 9 we dscussed the
Inductance Calculation for Conductors of Arbitrary Shape
 CRYO/02/028 Aprl 5, 2002 Inductance Calculaton for Conductors of Arbtrary Shape L. Bottura Dstrbuton: Internal Summary In ths note we descrbe a method for the numercal calculaton of nductances among conductors
CRYO/02/028 Aprl 5, 2002 Inductance Calculaton for Conductors of Arbtrary Shape L. Bottura Dstrbuton: Internal Summary In ths note we descrbe a method for the numercal calculaton of nductances among conductors
NMT EE 589 & UNM ME 482/582 ROBOT ENGINEERING. Dr. Stephen Bruder NMT EE 589 & UNM ME 482/582
 NMT EE 589 & UNM ME 482/582 ROBOT ENGINEERING Dr. Stephen Bruder NMT EE 589 & UNM ME 482/582 4. Moton Knematcs 4.2 Angular Velocty Knematcs Summary From the last lecture we concluded that: If the jonts
NMT EE 589 & UNM ME 482/582 ROBOT ENGINEERING Dr. Stephen Bruder NMT EE 589 & UNM ME 482/582 4. Moton Knematcs 4.2 Angular Velocty Knematcs Summary From the last lecture we concluded that: If the jonts
Please review the following statement: I certify that I have not given unauthorized aid nor have I received aid in the completion of this exam.
 ME 270 Sprng 2017 Exam 1 NAME (Last, Frst): Please revew the followng statement: I certfy that I have not gven unauthorzed ad nor have I receved ad n the completon of ths exam. Sgnature: Instructor s Name
ME 270 Sprng 2017 Exam 1 NAME (Last, Frst): Please revew the followng statement: I certfy that I have not gven unauthorzed ad nor have I receved ad n the completon of ths exam. Sgnature: Instructor s Name
MODEL OF HYDROPNEUMATIC THREE POINT HITCH
 ENINEERIN FR RUR DEVEPMENT Jelgava, 3.-4.05.03. MDE F YDRPNEUMTI TREE PINT IT Jans acekls-bertmans, Erks Kronbergs atva Unversty of grculture jans.lacekls@llu.lv, erks.kronbergs@llu.lv bstract. Ths paper
ENINEERIN FR RUR DEVEPMENT Jelgava, 3.-4.05.03. MDE F YDRPNEUMTI TREE PINT IT Jans acekls-bertmans, Erks Kronbergs atva Unversty of grculture jans.lacekls@llu.lv, erks.kronbergs@llu.lv bstract. Ths paper
Gasometric Determination of NaHCO 3 in a Mixture
 60 50 40 0 0 5 15 25 35 40 Temperature ( o C) 9/28/16 Gasometrc Determnaton of NaHCO 3 n a Mxture apor Pressure (mm Hg) apor Pressure of Water 1 NaHCO 3 (s) + H + (aq) Na + (aq) + H 2 O (l) + CO 2 (g)
60 50 40 0 0 5 15 25 35 40 Temperature ( o C) 9/28/16 Gasometrc Determnaton of NaHCO 3 n a Mxture apor Pressure (mm Hg) apor Pressure of Water 1 NaHCO 3 (s) + H + (aq) Na + (aq) + H 2 O (l) + CO 2 (g)
Global Sensitivity. Tuesday 20 th February, 2018
 Global Senstvty Tuesday 2 th February, 28 ) Local Senstvty Most senstvty analyses [] are based on local estmates of senstvty, typcally by expandng the response n a Taylor seres about some specfc values
Global Senstvty Tuesday 2 th February, 28 ) Local Senstvty Most senstvty analyses [] are based on local estmates of senstvty, typcally by expandng the response n a Taylor seres about some specfc values
ANSWERS. Problem 1. and the moment generating function (mgf) by. defined for any real t. Use this to show that E( U) var( U)
 Econ 413 Exam 13 H ANSWERS Settet er nndelt 9 deloppgaver, A,B,C, som alle anbefales å telle lkt for å gøre det ltt lettere å stå. Svar er gtt . Unfortunately, there s a prntng error n the hnt of
Econ 413 Exam 13 H ANSWERS Settet er nndelt 9 deloppgaver, A,B,C, som alle anbefales å telle lkt for å gøre det ltt lettere å stå. Svar er gtt . Unfortunately, there s a prntng error n the hnt of
Experiment 1 Mass, volume and density
 Experment 1 Mass, volume and densty Purpose 1. Famlarze wth basc measurement tools such as verner calper, mcrometer, and laboratory balance. 2. Learn how to use the concepts of sgnfcant fgures, expermental
Experment 1 Mass, volume and densty Purpose 1. Famlarze wth basc measurement tools such as verner calper, mcrometer, and laboratory balance. 2. Learn how to use the concepts of sgnfcant fgures, expermental
ADSORPTION EQUILIBRIUM OF LIGHT HYDROCARBON MIXTURES BY MONTE CARLO SIMULATION
 Brazlan Journal of Chemcal Engneerng ISSN 0104-6632 Prnted n Brazl www.abeq.org.br/bjche Vol. 24, No. 04, pp. 597-610 October - December, 2007 ADSORPTION EQUILIBRIUM OF LIGHT HYDROCARBON MIXTURES BY MONTE
Brazlan Journal of Chemcal Engneerng ISSN 0104-6632 Prnted n Brazl www.abeq.org.br/bjche Vol. 24, No. 04, pp. 597-610 October - December, 2007 ADSORPTION EQUILIBRIUM OF LIGHT HYDROCARBON MIXTURES BY MONTE
ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM
 ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM An elastc wave s a deformaton of the body that travels throughout the body n all drectons. We can examne the deformaton over a perod of tme by fxng our look
ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM An elastc wave s a deformaton of the body that travels throughout the body n all drectons. We can examne the deformaton over a perod of tme by fxng our look
Electrochemical Equilibrium Electromotive Force
 CHM465/865, 24-3, Lecture 5-7, 2 th Sep., 24 lectrochemcal qulbrum lectromotve Force Relaton between chemcal and electrc drvng forces lectrochemcal system at constant T and p: consder Gbbs free energy
CHM465/865, 24-3, Lecture 5-7, 2 th Sep., 24 lectrochemcal qulbrum lectromotve Force Relaton between chemcal and electrc drvng forces lectrochemcal system at constant T and p: consder Gbbs free energy
Chapter 2. Electrode/electrolyte interface: ----Structure and properties
 Chapter 2 Electrode/electrolyte nterface: ----Structure and propertes Electrochemcal reactons are nterfacal reactons, the structure and propertes of electrode / electrolytc soluton nterface greatly nfluences
Chapter 2 Electrode/electrolyte nterface: ----Structure and propertes Electrochemcal reactons are nterfacal reactons, the structure and propertes of electrode / electrolytc soluton nterface greatly nfluences
Adiabatic Sorption of Ammonia-Water System and Depicting in p-t-x Diagram
 Adabatc Sorpton of Ammona-Water System and Depctng n p-t-x Dagram J. POSPISIL, Z. SKALA Faculty of Mechancal Engneerng Brno Unversty of Technology Techncka 2, Brno 61669 CZECH REPUBLIC Abstract: - Absorpton
Adabatc Sorpton of Ammona-Water System and Depctng n p-t-x Dagram J. POSPISIL, Z. SKALA Faculty of Mechancal Engneerng Brno Unversty of Technology Techncka 2, Brno 61669 CZECH REPUBLIC Abstract: - Absorpton
Adsorption: A gas or gases from a mixture of gases or a liquid (or liquids) from a mixture of liquids is bound physically to the surface of a solid.
 Searatons n Chemcal Engneerng Searatons (gas from a mxture of gases, lquds from a mxture of lquds, solds from a soluton of solds n lquds, dssolved gases from lquds, solvents from gases artally/comletely
Searatons n Chemcal Engneerng Searatons (gas from a mxture of gases, lquds from a mxture of lquds, solds from a soluton of solds n lquds, dssolved gases from lquds, solvents from gases artally/comletely
Open Systems: Chemical Potential and Partial Molar Quantities Chemical Potential
 Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
Open Systems: Chemcal Potental and Partal Molar Quanttes Chemcal Potental For closed systems, we have derved the followng relatonshps: du = TdS pdv dh = TdS + Vdp da = SdT pdv dg = VdP SdT For open systems,
2.If the concentration is of bulk phenomenon, it is absorption. (e.g) Absorption of Ink on the surface of a chalk.
 183101 EGIEERIG CHEMISTRY UIT 3 SURFACE CHEMISTRY TERMS AD DEFIITIOS: 1. Adsorpton: Concentraton of lqud or gaseous molecules over the surface of a sold materal s known as adsorpton. It s a surface phenomenon.
183101 EGIEERIG CHEMISTRY UIT 3 SURFACE CHEMISTRY TERMS AD DEFIITIOS: 1. Adsorpton: Concentraton of lqud or gaseous molecules over the surface of a sold materal s known as adsorpton. It s a surface phenomenon.
If two volatile and miscible liquids are combined to form a solution, Raoult s law is not obeyed. Use the experimental data in Table 9.
 9.9 Real Solutons Exhbt Devatons from Raoult s Law If two volatle and mscble lquds are combned to form a soluton, Raoult s law s not obeyed. Use the expermental data n Table 9.3: Physcal Chemstry 00 Pearson
9.9 Real Solutons Exhbt Devatons from Raoult s Law If two volatle and mscble lquds are combned to form a soluton, Raoult s law s not obeyed. Use the expermental data n Table 9.3: Physcal Chemstry 00 Pearson
The ChemSep Book. Harry A. Kooijman Consultant. Ross Taylor Clarkson University, Potsdam, New York University of Twente, Enschede, The Netherlands
 The ChemSep Book Harry A. Koojman Consultant Ross Taylor Clarkson Unversty, Potsdam, New York Unversty of Twente, Enschede, The Netherlands Lbr Books on Demand www.bod.de Copyrght c 2000 by H.A. Koojman
The ChemSep Book Harry A. Koojman Consultant Ross Taylor Clarkson Unversty, Potsdam, New York Unversty of Twente, Enschede, The Netherlands Lbr Books on Demand www.bod.de Copyrght c 2000 by H.A. Koojman
Lecture 4: November 17, Part 1 Single Buffer Management
 Lecturer: Ad Rosén Algorthms for the anagement of Networs Fall 2003-2004 Lecture 4: November 7, 2003 Scrbe: Guy Grebla Part Sngle Buffer anagement In the prevous lecture we taled about the Combned Input
Lecturer: Ad Rosén Algorthms for the anagement of Networs Fall 2003-2004 Lecture 4: November 7, 2003 Scrbe: Guy Grebla Part Sngle Buffer anagement In the prevous lecture we taled about the Combned Input
CONTRAST ENHANCEMENT FOR MIMIMUM MEAN BRIGHTNESS ERROR FROM HISTOGRAM PARTITIONING INTRODUCTION
 CONTRAST ENHANCEMENT FOR MIMIMUM MEAN BRIGHTNESS ERROR FROM HISTOGRAM PARTITIONING N. Phanthuna 1,2, F. Cheevasuvt 2 and S. Chtwong 2 1 Department of Electrcal Engneerng, Faculty of Engneerng Rajamangala
CONTRAST ENHANCEMENT FOR MIMIMUM MEAN BRIGHTNESS ERROR FROM HISTOGRAM PARTITIONING N. Phanthuna 1,2, F. Cheevasuvt 2 and S. Chtwong 2 1 Department of Electrcal Engneerng, Faculty of Engneerng Rajamangala
Please review the following statement: I certify that I have not given unauthorized aid nor have I received aid in the completion of this exam.
 NME (Last, Frst): Please revew the followng statement: I certfy that I have not gven unauthorzed ad nor have I receved ad n the completon of ths exam. Sgnature: INSTRUCTIONS Begn each problem n the space
NME (Last, Frst): Please revew the followng statement: I certfy that I have not gven unauthorzed ad nor have I receved ad n the completon of ths exam. Sgnature: INSTRUCTIONS Begn each problem n the space
Frequency dependence of the permittivity
 Frequency dependence of the permttvty February 7, 016 In materals, the delectrc constant and permeablty are actually frequency dependent. Ths does not affect our results for sngle frequency modes, but
Frequency dependence of the permttvty February 7, 016 In materals, the delectrc constant and permeablty are actually frequency dependent. Ths does not affect our results for sngle frequency modes, but
Graph Reconstruction by Permutations
 Graph Reconstructon by Permutatons Perre Ille and Wllam Kocay* Insttut de Mathémathques de Lumny CNRS UMR 6206 163 avenue de Lumny, Case 907 13288 Marselle Cedex 9, France e-mal: lle@ml.unv-mrs.fr Computer
Graph Reconstructon by Permutatons Perre Ille and Wllam Kocay* Insttut de Mathémathques de Lumny CNRS UMR 6206 163 avenue de Lumny, Case 907 13288 Marselle Cedex 9, France e-mal: lle@ml.unv-mrs.fr Computer
CHAPTER IV RESEARCH FINDING AND DISCUSSIONS
 CHAPTER IV RESEARCH FINDING AND DISCUSSIONS A. Descrpton of Research Fndng. The Implementaton of Learnng Havng ganed the whole needed data, the researcher then dd analyss whch refers to the statstcal data
CHAPTER IV RESEARCH FINDING AND DISCUSSIONS A. Descrpton of Research Fndng. The Implementaton of Learnng Havng ganed the whole needed data, the researcher then dd analyss whch refers to the statstcal data
Markov Chain Monte Carlo Lecture 6
 where (x 1,..., x N ) X N, N s called the populaton sze, f(x) f (x) for at least one {1, 2,..., N}, and those dfferent from f(x) are called the tral dstrbutons n terms of mportance samplng. Dfferent ways
where (x 1,..., x N ) X N, N s called the populaton sze, f(x) f (x) for at least one {1, 2,..., N}, and those dfferent from f(x) are called the tral dstrbutons n terms of mportance samplng. Dfferent ways
A novel mathematical model of formulation design of emulsion explosive
 J. Iran. Chem. Res. 1 (008) 33-40 Journal of the Iranan Chemcal Research IAU-ARAK www.au-jcr.com A novel mathematcal model of formulaton desgn of emulson explosve Mng Lu *, Qfa Lu Chemcal Engneerng College,
J. Iran. Chem. Res. 1 (008) 33-40 Journal of the Iranan Chemcal Research IAU-ARAK www.au-jcr.com A novel mathematcal model of formulaton desgn of emulson explosve Mng Lu *, Qfa Lu Chemcal Engneerng College,
Monte Carlo method II
 Course MP3 Lecture 5 14/11/2006 Monte Carlo method II How to put some real physcs nto the Monte Carlo method Dr James Ellott 5.1 Monte Carlo method revsted In lecture 4, we ntroduced the Monte Carlo (MC)
Course MP3 Lecture 5 14/11/2006 Monte Carlo method II How to put some real physcs nto the Monte Carlo method Dr James Ellott 5.1 Monte Carlo method revsted In lecture 4, we ntroduced the Monte Carlo (MC)
Problem Set 9 Solutions
 Desgn and Analyss of Algorthms May 4, 2015 Massachusetts Insttute of Technology 6.046J/18.410J Profs. Erk Demane, Srn Devadas, and Nancy Lynch Problem Set 9 Solutons Problem Set 9 Solutons Ths problem
Desgn and Analyss of Algorthms May 4, 2015 Massachusetts Insttute of Technology 6.046J/18.410J Profs. Erk Demane, Srn Devadas, and Nancy Lynch Problem Set 9 Solutons Problem Set 9 Solutons Ths problem
University of Washington Department of Chemistry Chemistry 453 Winter Quarter 2015
 Lecture 2. 1/07/15-1/09/15 Unversty of Washngton Department of Chemstry Chemstry 453 Wnter Quarter 2015 We are not talkng about truth. We are talkng about somethng that seems lke truth. The truth we want
Lecture 2. 1/07/15-1/09/15 Unversty of Washngton Department of Chemstry Chemstry 453 Wnter Quarter 2015 We are not talkng about truth. We are talkng about somethng that seems lke truth. The truth we want
NUMERICAL DIFFERENTIATION
 NUMERICAL DIFFERENTIATION 1 Introducton Dfferentaton s a method to compute the rate at whch a dependent output y changes wth respect to the change n the ndependent nput x. Ths rate of change s called the
NUMERICAL DIFFERENTIATION 1 Introducton Dfferentaton s a method to compute the rate at whch a dependent output y changes wth respect to the change n the ndependent nput x. Ths rate of change s called the
MAGNETISM MAGNETIC DIPOLES
 MAGNETISM We now turn to magnetsm. Ths has actually been used for longer than electrcty. People were usng compasses to sal around the Medterranean Sea several hundred years BC. However t was not understood
MAGNETISM We now turn to magnetsm. Ths has actually been used for longer than electrcty. People were usng compasses to sal around the Medterranean Sea several hundred years BC. However t was not understood
Simulated Power of the Discrete Cramér-von Mises Goodness-of-Fit Tests
 Smulated of the Cramér-von Mses Goodness-of-Ft Tests Steele, M., Chaselng, J. and 3 Hurst, C. School of Mathematcal and Physcal Scences, James Cook Unversty, Australan School of Envronmental Studes, Grffth
Smulated of the Cramér-von Mses Goodness-of-Ft Tests Steele, M., Chaselng, J. and 3 Hurst, C. School of Mathematcal and Physcal Scences, James Cook Unversty, Australan School of Envronmental Studes, Grffth
Thermal Desorption in Pure Hexane and Hexane/Butane Mixtures on Graphite
 Thermal Desorpton n Pure Hexane and Hexane/Butane Mxtures on Graphte Cary L. Pnt Department of Physcs Unversty of Northern Iowa Cedar Falls, Iowa 564-5 USA Receved: November 9, 24 Accepted: November 29,
Thermal Desorpton n Pure Hexane and Hexane/Butane Mxtures on Graphte Cary L. Pnt Department of Physcs Unversty of Northern Iowa Cedar Falls, Iowa 564-5 USA Receved: November 9, 24 Accepted: November 29,
On Adaptive Control of Simulated Moving Bed Plants. Plants Using Comsol s Simulink Interface. Speaker: Marco Fütterer
 daptve Smulated Movng ed Plants Usng Comsol s Smulnk Interfae Speaker: Maro Fütterer Insttut für utomatserungstehnk Otto-von-Guerke Unverstät Unverstätsplatz, D-39106 Magdeburg Germany e-mal: maro.fuetterer@ovgu.de
daptve Smulated Movng ed Plants Usng Comsol s Smulnk Interfae Speaker: Maro Fütterer Insttut für utomatserungstehnk Otto-von-Guerke Unverstät Unverstätsplatz, D-39106 Magdeburg Germany e-mal: maro.fuetterer@ovgu.de
Investigation of a New Monte Carlo Method for the Transitional Gas Flow
 Investgaton of a New Monte Carlo Method for the Transtonal Gas Flow X. Luo and Chr. Day Karlsruhe Insttute of Technology(KIT) Insttute for Techncal Physcs 7602 Karlsruhe Germany Abstract. The Drect Smulaton
Investgaton of a New Monte Carlo Method for the Transtonal Gas Flow X. Luo and Chr. Day Karlsruhe Insttute of Technology(KIT) Insttute for Techncal Physcs 7602 Karlsruhe Germany Abstract. The Drect Smulaton
y i x P vap 10 A T SOLUTION TO HOMEWORK #7 #Problem
 SOLUTION TO HOMEWORK #7 #roblem 1 10.1-1 a. In order to solve ths problem, we need to know what happens at the bubble pont; at ths pont, the frst bubble s formed, so we can assume that all of the number
SOLUTION TO HOMEWORK #7 #roblem 1 10.1-1 a. In order to solve ths problem, we need to know what happens at the bubble pont; at ths pont, the frst bubble s formed, so we can assume that all of the number
STUDY ON TWO PHASE FLOW IN MICRO CHANNEL BASED ON EXPERI- MENTS AND NUMERICAL EXAMINATIONS
 Blucher Mechancal Engneerng Proceedngs May 0, vol., num. www.proceedngs.blucher.com.br/evento/0wccm STUDY ON TWO PHASE FLOW IN MICRO CHANNEL BASED ON EXPERI- MENTS AND NUMERICAL EXAMINATIONS Takahko Kurahash,
Blucher Mechancal Engneerng Proceedngs May 0, vol., num. www.proceedngs.blucher.com.br/evento/0wccm STUDY ON TWO PHASE FLOW IN MICRO CHANNEL BASED ON EXPERI- MENTS AND NUMERICAL EXAMINATIONS Takahko Kurahash,
Physics 114 Exam 2 Fall 2014 Solutions. Name:
 Physcs 114 Exam Fall 014 Name: For gradng purposes (do not wrte here): Queston 1. 1... 3. 3. Problem Answer each of the followng questons. Ponts for each queston are ndcated n red. Unless otherwse ndcated,
Physcs 114 Exam Fall 014 Name: For gradng purposes (do not wrte here): Queston 1. 1... 3. 3. Problem Answer each of the followng questons. Ponts for each queston are ndcated n red. Unless otherwse ndcated,
#64. ΔS for Isothermal Mixing of Ideal Gases
 #64 Carnot Heat Engne ΔS for Isothermal Mxng of Ideal Gases ds = S dt + S T V V S = P V T T V PV = nrt, P T ds = v T = nr V dv V nr V V = nrln V V = - nrln V V ΔS ΔS ΔS for Isothermal Mxng for Ideal Gases
#64 Carnot Heat Engne ΔS for Isothermal Mxng of Ideal Gases ds = S dt + S T V V S = P V T T V PV = nrt, P T ds = v T = nr V dv V nr V V = nrln V V = - nrln V V ΔS ΔS ΔS for Isothermal Mxng for Ideal Gases
