CHEMILUM NESCENCE FROM THE REACTION OF 2-METHYLENE-3-ACETYLOXAZ--ETC(U) AU 6 2 D STEFFE( A C MEBANE, G A SCHUSTER N00OSA 76-C 0745
|
|
|
- Frederick Malone
- 5 years ago
- Views:
Transcription
1 El CHEMILUM NESCENCE FROM THE REACTION OF 2-METHYLENE-3-ACETYLOXAZ--ETC(U) AU 6 2 D STEFFE( A C MEBANE, G A SCHUSTER N00OSA 76-C 0745
2 * R-M.~.. 11 unclassified SECURITY CLASSIFICATION OF THIS PAGE (When Dol. Eatwoo r REPORT DOCMENTATION PAGE -ZOR ng~ CMLEV ORM I. REPORT NUMBUR 12. OOVT ACCESSION NO: 3. RECIPIENT'S CATALOG NUMBER N C TITLE (men Subtile) S. TYPE OF REPORT 6 PERIOD COVERED S Chemiluminescence fromi the Reaction of 2- S Methylene-3-acetyloxazoline-4,5-dione with Hydrogen Peroxide -. Technical I. PERFORMING ONG. REPORT NUMBER 00 Daniel Steffek, Robert C. Mebane and N Gary B. Schuster 7. AUTHOR(s) I. CONTRACT ON GRANT NuNSEP1(s) 9PERFORMING ORGANIZATION NAME AND ADDRESS to. PROGRAM ELEMENT. PROJECT. TASK S. AREA 6 WORK UNIT NUMBERS Department of Chemistry 4! University of Illinois NR Urbana, IL I I. CONTROLLING OFFICE NAME AND ADDRESS 12. REPORT DATE S Chemistry Program, Materials Science Division August 20, 1982 Office of Naval Research, 800 N. Quincy Street IS. NUMBER OF PAGES Arlington, VA MONITORING AGENCY NAME & AOORIESS(it diferfroo t Controllng Ofice.) 16. DISTRIBUTION STATEMENT (of this Deport) IIS. SECURITY CLASS. (Of ftopoew Unclassified IS&. DECLASSIFICATIOM/DOWNGRADING SCHEDUJLE This document has been approved for public release and sale; its distribution is unlimited 17. DISTRIBUTION STATEMENT (of the abstrten terd In Block S.it different from Ror..t) 10. SUPPLEMENTARY NOTES chemilnuinescence cylcic peroxides AU 1982 CIEEL 20. JTRACT (Contnue an roe o *#do tf nfooeow, and Ifltit by Nock niinbt) I~ereaction of 2-methylen-3ceyoxazoline-4,-5-dione W) with hydrogen peroxide in the presence of a fluorescent aromatic hydrocarbon generates light. The mechanism for light generation was investigated and found to parallel that LU of oxalate esters. The reaction of dione(iy. catalyzed by added base, but the i... chemiluminescence intensity drops as the'base concentration increases. A Sreaction sequence is proposed consistent with the experimental observations.p DD D ON EDITION OFP I NOV 0S IS OSOLETE UNCLASSIFIED I/M SECURITY CLASSIFICATION OFP TWIS8 PASE (11beo. Dot 8~.U ameree
3 OFFICE OF NAVAL RESEARCH Contract N C-0745 Task No. NR TECHNICAL REPORT NO. N C Prepared for Publication in Journal of Organic Chemistry School of Chemical Sciences University of Illinois Urbana, Illinois August 20, 1982 Reproduction in whole or in part is permitted for any purposes of the United States Government Approved for Public Release; Distribution Unlimited. A Accession For NTIS l-- Al'- 7 7 ' -' J11 t: Di-.t Chemiluminescence from the Reaction of 2-Methylene-3-acetyloxazoline- 4,5-dione with Hydrogen Peroxide by Daniel Steffek, Robert C. Mebane and Gary B. Schuster 4 Noon-
4 Chemical reactions that generate visible light have been actively 1 investigated for the past 50 years. Recent research has revealed important details of the chemical and physical transformations required for 2 efficient light generation from these processes. Typically these reactions involve, as starting material or as a key intermediate,an organic peroxide. Conversion of the weak oxygen-oxygen bond of the peroxide to a much stronger carbon-oxygen double bond in a product carbonyl group in most cases supplies much of the energy required to form the requisite electronically excited state. The mechanism for conversion of the chemical potential energy of the peroxide to visible light depends on the details of the structure of the peroxide. One of the most efficient chemiluminescent systems yet discovered is based on the reaction of hydrogen peroxide with certain derivatives of oxalic acid in the presence of a suitable fluorescer. 3 ' 4 This reaction can generate light with an efficiency approaching 35%, and forms the basis for the popular Cyalume light sticks. For these reasons we were attracted to the recent report by Richter and Temme 6 of a new reaction of aliphatic imides with oxalyl chloride to give oxazolidinediones, eq 1. We set out to examine the possibility that these "derivatives" of oxalic 1 acid are capable of generating light. Herein we report the results of that investigation. H CH 3 *y r 0 -HCI- 0 I CH 3 CH 3 CI CI H (CH2 Oxazolidinedione was prepared according to the published procedure. When a solution of dione in dimethylphthalate containing 9,10-diphenylanthracene (DPA) is treated with hydrogen peroxide blue chemiluminescence characteristic of DPA fluorescence can be easily seen in a darkened room
5 with the unaided eye. Control experiments showed that all three ingredients are required to generate light. Thus the central question asked at the outset of this investigation can be answered in the affirmative. Dione is capable of generating chemiluminescence. We set out next to explore the details of the chemical transformations leading to light. The rate of the chemiluminescent reaction of is increased by added bases. We explored several potential catalysts for the reaction of with hydrogen peroxide and found that 2,6-lutidine gives clean and reproducible results. Addition of the lutidine to the reaction mixture has two effects on the chemiluminescent process. First, as the lutidine concentration is increased the observed pseudo first-order rate of decay of tf'e chemiluminescent reaction increases, Figure 1. Second, increasing the lutidine concentration from 5.2xlO-4 M to 5.2xlO-2 M leads to a decrease in the total chemiluminescent intensity obtained by a factor of about 15. Thus the lutidine must be effecting two parts of the reaction that leads eventually to light. The lutidine increases the rate of the slow step while inhibiting eventual generation of light. It should be noted that while lutidine does quench the fluorescence of DPA in dimethylphthalate this process alone is not sufficient to account for the decrease in light intensity. 7 In a similar fashion we examined the effect of hydrogen peroxide concentration on the rate of the chemiluminescent reaction of k and its light generating efficiency. This study revealed that the hydrogen peroxide and the base have parallel effects. Increasing the hydrogen peroxide concentration increased the rate of reaction while simultaneously decreasing the light output. Changing the fluorescer structure reveals some additional details about the light generating process. The aromatic hydrocarbon fluorescers such as DPA, rubrene, perylene and 9,10-diphenylethynylanthracene all give
6 reasonably intense chemiluminescence. However, 9,10-dibromoanthracene (DBA), which has been shown to be a useful diagnostic for the generation of triplet excited states, 8 gives no meaningful light above the low background level. This observation, similar to one we made earlier for diphenoyl peroxide cnemiluminescence, 9 is indicative of the operation of the chemically initiated electron-exchange luminescence (CIEEL) mechanism. 2 Investigations of the chemiluminescence of some other oxalic acid derivatives has revealed that the fluorescer catalyzes the decomposition of 4 a metastable intermediate. Thus addition of the fluorescer to the reaction mixture some time after the other reagents have been mixed still yields nearly as much light as is obtained when the fluorescer is present from the outset. Similar attempts to detect a metastable intermediate from dione were unsuccessful. Late addition of the fluorescer did not generate any light above that expected based on the known rate of reaction of the system components. The results of these investigations permit us to suggest, as a working hypothesis, the mechanism for the chemiluminescence of dione shown in Scheme 1. This mechanism is modeled after a similar proposal made by Scheme 1 0 H202 t [M I N jh00, LH A~ )LM %, 00 0~ I, 4-cyclization!, 6-cyclization 0 luoreser 0r-N 0C. 3 ' 0-0 \d.ch3 0 0-, 00 % N 3 CH 3 0 Chemi luminescence dark 14O2 Q 4 + HNCI CH dark 00-- OH )CH
7 Rauhut for the chemiluminescence of oxalate esters. 4 In the first, and ratelimiting, step hydroperoxide anion adds to the oxazolinedione opening the ring to generate first an enolate anion and then the peroxycarboxylate anion. The rate of this reaction should be sensitive to the lutidine concentration since it depends upon the amount of hydroperoxide present. The fate of the ring opened intermediate probably determines the chemiluminescence yield. Three reasonable alternative reactions are suggested; 1,4-cyclization leading to a dioxetane intermediate is the most likely route to the observed chemiluminescence, 1,6-cyclization, and base catalyzed addition of a second equivalent of hydrogen peroxide, can compete with the dioxetane route and do not lead to light. The base catalysis of the reaction rate and inhibiton of chemiluminescence are consistent with this proposal. Also, the absence of a metastable intermediate is reasonably accounted for since reversal of the 1,4- cycloaddition gives the dark routes another opportunity to consume the intermediate. We have qualitatively measured the light generating efficiency of the reaction of 1 and find it to be several orders of magnitude less than the most efficient oxalate esters, However, no attempt has yet been made to optimize the structure of thp. oxazolinedione to increase light output. Acknowledgement: This work was supported by the Office of Naval Research.... _
8 t 5 rimental Section General. Solvents were purified by distillation before use. Fluorescers were chromatographed on alumina and then recrystallized. The chemiluminescence measurements were performed by the photon counting method at room 10 temperature using the apparatus previously described. T ca emiluminescence measurement. A solution of oxazolinedione in dimethylphthalate was added to a solution of the fluorescer and hydrogen peroxide, also in dimethylphthalate, so that the final volume was ca. 2.0 ml. Typical concentrations of these reagents in this solution were 1, 5.5xi0 4 M; fluorescer, 3.5x10-4 M; H ' 0.4 M. For the lutidine catalyzed runs was the base also present in the peroxide solution. The light output from these solutions were monitored for ca. 4 half-lives at room Lemperature. I', L on--
9 1. Albrecht, H. 0. Z. Physik. Chem., 1928, 136, Schuster, G. B.; Schmidt, S. P. Adv. Phys. Org. Chem., 1982, 18, Chandross, E. A. Tetrahedron Lett., 1963, Rauhut, M. M. Accts. Chem. Res., 1969, 2, Tseng, S.-S.; Mohan, A. G.; Haines, L. G.; Lourdes, S. U.; Rauhut, M. M. J. Org. Chem., 1979, 44, Richter, R.; Temme, G. H. J. Org. Chem., 1981, 46, The lifetime of DPA is ca. 9 ns, thus quenching would have to occur at a rate far above the diffusion limit for the lutidine to quench DPA fluorescence to the extent observed. 8. Vassil'ev, R. F. Ptog. React. Kinet., 1967, 4, 305. Wilson, T.; Schaap, A. P. J. Amer. Chem. Soc., 1971, 93, Koo, J.-Y.; Schuster, G. B. J. Amer. Chem. Soc., 1977, 99, 6107, ibid., 1978, 10_0, Smith, J. P.; Schrock, A. K.; Schuster, G. B. J. Amer. Chem. Soc., 1982, 104, ~ 3
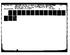
10 _.illli r LUTIDINE M
11 Captions for Figure Figure 1 The effect of added lutidine on the observed first order rate constant for decay of chemiluminescence from in dimethylphthalate at room temperature. The catalytic rate constant is 5.2xlO - s O *1 1:
12 SP472-3/Al 472 :GAN:716:dde 78u No. TECH!JICAL REPORT DISTRIBUTION LIST, GEN Copies No. Co.ie Office of Naval Research U.S. Army Research Office Aztn: Code 472 Attn: CRD-AA-IP 800 North Quincy Street P.O. Box 1211 Arlington, Virginia Research Triangle Park, N.C O R Branch Office Naval Ocean Systems Center Attn: Dr. George Sandoz Attn: Mr. Joe McCartney 536 S. Clark Street San Diego, California Chicago, Illinois Naval Weapons Center 0%," Area Office Attn: Dr. A. B. Amster, Attn: Scientific Dept. Chemistry Division 715 Broadway China Lake, California New York, tew York Naval Civil Engineering Laboratory ON!I Western Regional Office Attn: Dr. R. W. Drisko 1030 East Green Street Port lueneme, California Pasadena, California O:P Eastern/Central Regional Office Department of Physics & Chemistry Naval Postgraduate School Atcn: Dr. L. It. Peebles Monterey, California Euilding 114, Section D 666 Summer Street Dr. A. L. S1afkosky Poston, Massachusetts Scientific Advisor Commandant of the Marine Corps Director, Naval Research Laboratory (Code RD-1) Attn: Code 6100 Washington, D.C a1hington, D.C Office of Naval Research The Assistant Secretary Attn: Dr. Richard S. Miller of the Navy (RE&S) 800 N. Quincy Street :; partnent of the Navy Arlington, Virginia Pom 4E736, Pentagon V:ashington, D.C Naval Ship Research and Development Center Commander, Naval Air Systems Command Attn: Dr. G. Bosmajian, Applied Attn: Code 310C (H. Rosenwasser) Chemistry Division De.partment of the Navy Annapolis, Maryland Uashington, D.C Naval Ocean Systems Center Defense Technical Information Center Attn: Dr. S. Yamamoto, Harine Building 5, Cameron Station Sciences Division Alexandria, Virginia San Diego, California Dr. Fred Saalfeld Mr. John Boyle Chemistry Division, Code 6100 Materials Branch "aval Research Laboratory Naval Ship Engineering Center Washington, D.C Philadelphia, Pennsylvania bo --- C-
13 5P4 SP4723/A378u4 7-3/A3472:AN :ddc TECHNICAL REPORT DISTRIBUTION LIST, GEN No. Copies Dr. Rudolph J. Harcus of fice of Naval Research Scientific Liaison Group American Embassy APO San Francisco :,r. James Kelley DTNSRDC Code 2803 Annapolis, naryland V1 2
14 SP482-3/A23 472:GAN:716:ddc 78u TECHNICAL REPORT DISTRIBUTION LIST, 051A No. Copies No. Copies Dr. H. A. El-Sayed Dr. H. Rauhut Department of Chemistry Chemical Research Division University of California, American Cyanamid Company Los Angeles Bound Brook, New Jersey Los Angeles, California Dr. J. I. Zink Dr. E. R. Bernstein Department of Chemistry Department of Chemistry University of California, Colorado State University Los Angeles Fort Collins, Colorado Los Angeles, California Dr. C. A. Heller Dr. D. Haarer Naval Weapons Center IBH Code 6059 San Jose Research Center China Lake, California Cottle Road San Jose, California Dr. J. 1 R. MacDonald Chemistry Division Dr. John Cooper Naval Research Laboratory Code 6130 Code 6110 Naval Research Laboratory Washington, D.C Washington, D.C G. B. Schuster Dr. William M. Jackson *Cheol Depart Department of Chemistry Universit linois Howard University Ur,' llinois 1 1 Washington, DC Dr. A. Adamson Dr. George E. Walraffen Department of Chemistry Department of Chemistry University of Southern Howard University California Washington, DC Los Angeles, California Dr. M. S. Wrighton Department of Chemistry Massachusetts Institute of Technology Cambridge, Hassachusetts
15
Ulm 1.00 &L6 MICROCOPY RESOLUTION4 TEST CHART. 4-_IU44k 1URE-AU OF STANOARM-19WA - -W ~ -. EW W W.~ %
 ND-R193 591 A ROTATIONAL ISOMERIC STATE MODEL FOR THE POLYCARBONATE 1/'1 OF 22' - BIS (4-H.. (U) MASSACHUSETTS INST OF TECH CAMIRIDGE DEPT OF CHEMICAL ENOINEE.. A HtITNII( ET AL. UNCLASSIFIED MEu"M. 31
ND-R193 591 A ROTATIONAL ISOMERIC STATE MODEL FOR THE POLYCARBONATE 1/'1 OF 22' - BIS (4-H.. (U) MASSACHUSETTS INST OF TECH CAMIRIDGE DEPT OF CHEMICAL ENOINEE.. A HtITNII( ET AL. UNCLASSIFIED MEu"M. 31
\\IIl\\l/\\ll\\\I/ (\I\\\Il\\\lI\/I/III
 AD- A2 3 9 152 \\IIl\\l/\\ll\\\I/ (\I\\\Il\\\lI\/I/III OFFICE OF NAVAL RESEARCH Contract N00014-85-K-0772 Work Unit No. 4326-808 TECHNICAL REPORT NO. 139 A Ferracarborane Analogue to [Fp]-. Synthesis and
AD- A2 3 9 152 \\IIl\\l/\\ll\\\I/ (\I\\\Il\\\lI\/I/III OFFICE OF NAVAL RESEARCH Contract N00014-85-K-0772 Work Unit No. 4326-808 TECHNICAL REPORT NO. 139 A Ferracarborane Analogue to [Fp]-. Synthesis and
AD-A FATION PAGE cmb No SUPPLEMENTARY NOTESlIII lllll II ili JUN PRICE CODE
 Form Approved AD-A252 270 FATION PAGE cmb No. 0704-0188 ge I~ uour oer resorse. including tile time for reviewing instructions. searchng existing cata sourcf-.. 'r, P 'llecicon o0 of information Send Commintsr
Form Approved AD-A252 270 FATION PAGE cmb No. 0704-0188 ge I~ uour oer resorse. including tile time for reviewing instructions. searchng existing cata sourcf-.. 'r, P 'llecicon o0 of information Send Commintsr
I~ma. A. V. Tobolsy and V. D. Gupta MAY Princeton, New Jersey. Technical Report No. 34. The Office of Naval Research
 TEXTILE RESEARCH INSTITUTE 0 Princeton, New Jersey Technical Report No. 34 to The on Contract No. Nonr-09000 and Nonr-09001 THE MODULUS OF POLYETHYLENE by D DOC MC A. V. Tobolsy and V. D. Gupta MAY1 2
TEXTILE RESEARCH INSTITUTE 0 Princeton, New Jersey Technical Report No. 34 to The on Contract No. Nonr-09000 and Nonr-09001 THE MODULUS OF POLYETHYLENE by D DOC MC A. V. Tobolsy and V. D. Gupta MAY1 2
' -A ON COLOR POLYNOMIALS OF FIBONACCI GRAPHS(U) GEORGIA /1 UNIV ATHENS DEPT OF CHEMISTRY S EL-BASIL it RUG 87 TR-52 N K-8365
 ' -A184 133 ON COLOR POLYNOMIALS OF FIBONACCI GRAPHS(U) GEORGIA /1 UNIV ATHENS DEPT OF CHEMISTRY S EL-BASIL it RUG 87 TR-52 N88814-84-K-8365 UNCLASSIFIED F/G 12/1 NL l, lull 11111 I L I Ill- I1 I2.2 I
' -A184 133 ON COLOR POLYNOMIALS OF FIBONACCI GRAPHS(U) GEORGIA /1 UNIV ATHENS DEPT OF CHEMISTRY S EL-BASIL it RUG 87 TR-52 N88814-84-K-8365 UNCLASSIFIED F/G 12/1 NL l, lull 11111 I L I Ill- I1 I2.2 I
1W3. MICROCOPY RESOLUTION TEST CHART NATLONAL BUIREAU Of STANDARDS A...-.-
 AD-R149 745 NOVEL PHOTO-THERMAL TECHNIQUE FOR FLOW AND SPECTROSCOPIC MEASUREMENTS IN A DENSE AEROSOL STREAM (U) IBM RESEARCH LAB SAN JOSE CA H SONTAG ET AL. UNCLASSIFIED 28 DEC 84 TR-16 Nee8i4-83-C-0i70
AD-R149 745 NOVEL PHOTO-THERMAL TECHNIQUE FOR FLOW AND SPECTROSCOPIC MEASUREMENTS IN A DENSE AEROSOL STREAM (U) IBM RESEARCH LAB SAN JOSE CA H SONTAG ET AL. UNCLASSIFIED 28 DEC 84 TR-16 Nee8i4-83-C-0i70
K-0DI. flflflflflflflflflii
 flflflflflflflflflii AD-Ass7 364 TEXAS UNIV AT AUSTIN DEPT OF CHEMISTRY F/6 7/4 " POTOCATALYTIC PRODUCTION OF HYDROGEN FROM WATER AND TEXAS LIBN-ETC(U) JUM 80 S SATO, J M WHITE NOOOl,-75-C-0922 END K-0DI
flflflflflflflflflii AD-Ass7 364 TEXAS UNIV AT AUSTIN DEPT OF CHEMISTRY F/6 7/4 " POTOCATALYTIC PRODUCTION OF HYDROGEN FROM WATER AND TEXAS LIBN-ETC(U) JUM 80 S SATO, J M WHITE NOOOl,-75-C-0922 END K-0DI
Catalytic Oxidation of CW Agents Using H O in 2 2 Ionic Liquids
 Catalytic Oxidation of CW Agents Using H O in 2 2 Ionic Liquids William M. Nelson,, PhD IL Waste Management & Research Center 2003 Joint Service Scientific Conference on Chemical & Biological Defense Research
Catalytic Oxidation of CW Agents Using H O in 2 2 Ionic Liquids William M. Nelson,, PhD IL Waste Management & Research Center 2003 Joint Service Scientific Conference on Chemical & Biological Defense Research
THREE-DIMENSIONAL STRUCTURE AND EVOLUTION OF PROPAGATING DISTURBANCES IN THE MARINE LAYER OFF THE U.S
 THREE-DIMENSIONAL STRUCTURE AND EVOLUTION OF PROPAGATING DISTURBANCES IN THE MARINE LAYER OFF THE U.S. WEST COAST: ANALYSIS OF 1996 AIRCRAFT OBSERVATION LONG TERM GOALS John M. Bane, Jr. Department of
THREE-DIMENSIONAL STRUCTURE AND EVOLUTION OF PROPAGATING DISTURBANCES IN THE MARINE LAYER OFF THE U.S. WEST COAST: ANALYSIS OF 1996 AIRCRAFT OBSERVATION LONG TERM GOALS John M. Bane, Jr. Department of
8kb IRT CORP SAN DIEGO CALIF F/S 20/8 MOLECLJt.AR BEAM STUDIES OF LOW ENERGY REACTIONS. (U) OCT 78 B H NEYNABER, S V TANG
 r AD A059 8kb IRT CORP SAN DIEGO CALIF F/S 20/8 MOLECLJt.AR BEAM STUDIES OF LOW ENERGY REACTIONS. (U) OCT 78 B H NEYNABER, S V TANG N0001U 7k C OO11 UNCLASSIFIED IRT 8105 008 NL I END! 9845 I DAtt A1 onc
r AD A059 8kb IRT CORP SAN DIEGO CALIF F/S 20/8 MOLECLJt.AR BEAM STUDIES OF LOW ENERGY REACTIONS. (U) OCT 78 B H NEYNABER, S V TANG N0001U 7k C OO11 UNCLASSIFIED IRT 8105 008 NL I END! 9845 I DAtt A1 onc
128lll=*A [_ 2. MICROCOPY RLSOLUrION ILST CHART. NATONAT IIT If P, TANDAgllh Ap
 AO.AilI 400 DEFENSE MAPPING AGENCY HYDROGRAPHIC/ TOPOGRAPHIC CENT--ETC F/G 8/5 THE DEFENSE MAPPING AGENCY HYDROGRAPHIC/TOPOGRAPHIC CENTER GED--ETC(U) FEB 82 R B BERUFF UNCLASSIFIED NL LmEIl 128lll=*A0
AO.AilI 400 DEFENSE MAPPING AGENCY HYDROGRAPHIC/ TOPOGRAPHIC CENT--ETC F/G 8/5 THE DEFENSE MAPPING AGENCY HYDROGRAPHIC/TOPOGRAPHIC CENTER GED--ETC(U) FEB 82 R B BERUFF UNCLASSIFIED NL LmEIl 128lll=*A0
, i, ~. '~~~* a F- WI W U V U S S S S S S It
 AD-A194 365 REACTION DYNAMICS ON SEMICONDUCTOR SURFACES(U) h RENSSELAER POLYTECHNIC INST TROY NY DEPT OF MATERIALS ENGINEERING J B HUDSON 29 FEB 86 NOB8e4-86-K-0259 UNCLASSIFIED F/G 7/4 NI @MonossonE ,
AD-A194 365 REACTION DYNAMICS ON SEMICONDUCTOR SURFACES(U) h RENSSELAER POLYTECHNIC INST TROY NY DEPT OF MATERIALS ENGINEERING J B HUDSON 29 FEB 86 NOB8e4-86-K-0259 UNCLASSIFIED F/G 7/4 NI @MonossonE ,
UNCLASSIFIED AD DEFENSE DOCUMENTATION CENTER FOR SCIENTIFIC AND TECHNICAL INFORMATION CAMERON STATION, ALEXANDRIA, VIRGINIA UNCLASSIFIED
 UNCLASSIFIED AD 437890 DEFENSE DOCUMENTATION CENTER FOR SCIENTIFIC AND TECHNICAL INFORMATION CAMERON STATION, ALEXANDRIA, VIRGINIA UNCLASSIFIED NOTICE: When government or other drawings, specifications
UNCLASSIFIED AD 437890 DEFENSE DOCUMENTATION CENTER FOR SCIENTIFIC AND TECHNICAL INFORMATION CAMERON STATION, ALEXANDRIA, VIRGINIA UNCLASSIFIED NOTICE: When government or other drawings, specifications
A0A TEXAS UNIV AT AUSTIN DEPT OF CHEMISTRY F/G 7/5 PHOTOASSISTED WATER-GAS SHIFT REACTION ON PLATINIZED T7TANIAI T--ETCIUI
 AA113 878 TEXAS UNIV AT AUSTIN DEPT OF CHEMISTRY F/G 7/5 PHOTOASSISTED WATER-GAS SHIFT REACTION ON PLATINIZED T7TANIAI T--ETCIUI APR 82 S FANG. 8 CHEN..J M WHITE N14-75-C-922 UNCLASSIFIED NL OFFICE OF
AA113 878 TEXAS UNIV AT AUSTIN DEPT OF CHEMISTRY F/G 7/5 PHOTOASSISTED WATER-GAS SHIFT REACTION ON PLATINIZED T7TANIAI T--ETCIUI APR 82 S FANG. 8 CHEN..J M WHITE N14-75-C-922 UNCLASSIFIED NL OFFICE OF
Predicting Tropical Cyclone Formation and Structure Change
 Predicting Tropical Cyclone Formation and Structure Change Patrick A. Harr Department of Meteorology Naval Postgraduate School Monterey, CA 93943-5114 Telephone: (831)656-3787 FAX:(831)656-3061 email:
Predicting Tropical Cyclone Formation and Structure Change Patrick A. Harr Department of Meteorology Naval Postgraduate School Monterey, CA 93943-5114 Telephone: (831)656-3787 FAX:(831)656-3061 email:
F/6 7/4 I I ANGELES AND INHOMOGENEOUS LINEWIDTNS OF T--ETC (UI MR 81 J R MMOAN, E P CHOCK, W D HOPEWELL N C-0602 WICLASSIFIED TR-37 N
 F/6 7/4 I I A-0A7 614 CAIFORNIA ON THE ORIGINS UNIY OF LOS HOMOGENEOUS ANGELES AND INHOMOGENEOUS LINEWIDTNS OF T--ETC (UI MR 81 J R MMOAN, E P CHOCK, W D HOPEWELL N0001-75-C-0602 WICLASSIFIED TR-37 N LEVEL
F/6 7/4 I I A-0A7 614 CAIFORNIA ON THE ORIGINS UNIY OF LOS HOMOGENEOUS ANGELES AND INHOMOGENEOUS LINEWIDTNS OF T--ETC (UI MR 81 J R MMOAN, E P CHOCK, W D HOPEWELL N0001-75-C-0602 WICLASSIFIED TR-37 N LEVEL
Volume 6 Water Surface Profiles
 A United States Contribution to the International Hydrological Decade HEC-IHD-0600 Hydrologic Engineering Methods For Water Resources Development Volume 6 Water Surface Profiles July 1975 Approved for
A United States Contribution to the International Hydrological Decade HEC-IHD-0600 Hydrologic Engineering Methods For Water Resources Development Volume 6 Water Surface Profiles July 1975 Approved for
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Analyst. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Electrogenerated Chemiluminescence at 9,10-Diphenylanthracene/
Electronic Supplementary Material (ESI) for Analyst. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Electrogenerated Chemiluminescence at 9,10-Diphenylanthracene/
REPORT DOCUMENTATION PAGE. Theoretical Study on Nano-Catalyst Burn Rate. Yoshiyuki Kawazoe (Tohoku Univ) N/A AOARD UNIT APO AP
 REPORT DOCUMENTATION PAGE Form Approved OMB No. 0704-0188 The public reporting burden for this collection of information is estimated to average 1 hour per response, including the time for reviewing instructions,
REPORT DOCUMENTATION PAGE Form Approved OMB No. 0704-0188 The public reporting burden for this collection of information is estimated to average 1 hour per response, including the time for reviewing instructions,
UNCLASSIFIED SECONDARY I ON MASS SPECTROMETRIC IMAGE DEPTH PROFILE ANALYSIS D--ETCCUI SEP 82 G H MORRISON. P K CHU, A C HARRIS NOO0014AOC-0538 TR-9
 UNCLASSIFIED SECONDARY I ON MASS SPECTROMETRIC IMAGE DEPTH PROFILE ANALYSIS D--ETCCUI SEP 82 G H MORRISON. P K CHU, A C HARRIS NOO0014AOC-0538 TR-9 OFFICE OF NAVAL RESEARCH Contract N00014-80-C-0538 Task
UNCLASSIFIED SECONDARY I ON MASS SPECTROMETRIC IMAGE DEPTH PROFILE ANALYSIS D--ETCCUI SEP 82 G H MORRISON. P K CHU, A C HARRIS NOO0014AOC-0538 TR-9 OFFICE OF NAVAL RESEARCH Contract N00014-80-C-0538 Task
Parametric Models of NIR Transmission and Reflectivity Spectra for Dyed Fabrics
 Naval Research Laboratory Washington, DC 20375-5320 NRL/MR/5708--15-9629 Parametric Models of NIR Transmission and Reflectivity Spectra for Dyed Fabrics D. Aiken S. Ramsey T. Mayo Signature Technology
Naval Research Laboratory Washington, DC 20375-5320 NRL/MR/5708--15-9629 Parametric Models of NIR Transmission and Reflectivity Spectra for Dyed Fabrics D. Aiken S. Ramsey T. Mayo Signature Technology
INFRARED SPECTROSCOPY OF HYDROGEN CYANIDE IN SOLID PARAHYDROGEN (BRIEFING CHARTS)
 AFRL-MN-EG-TP-2006-7403 INFRARED SPECTROSCOPY OF HYDROGEN CYANIDE IN SOLID PARAHYDROGEN (BRIEFING CHARTS) C. Michael Lindsay, National Research Council, Post Doctoral Research Associate Mario E. Fajardo
AFRL-MN-EG-TP-2006-7403 INFRARED SPECTROSCOPY OF HYDROGEN CYANIDE IN SOLID PARAHYDROGEN (BRIEFING CHARTS) C. Michael Lindsay, National Research Council, Post Doctoral Research Associate Mario E. Fajardo
Real-Time Environmental Information Network and Analysis System (REINAS)
 Calhoun: The NPS Institutional Archive Faculty and Researcher Publications Faculty and Researcher Publications 1998-09 Real-Time Environmental Information Network and Analysis System (REINAS) Nuss, Wendell
Calhoun: The NPS Institutional Archive Faculty and Researcher Publications Faculty and Researcher Publications 1998-09 Real-Time Environmental Information Network and Analysis System (REINAS) Nuss, Wendell
7 Naval Research Laboratory OTIC
 Rockwell International Corporation 1049 Camino Dos Rios Rockwell P.O. Box 1085 Thousand Oaks, California 91360 International C (805) 373-4545 AD-A220 233 March 18, 1986 In Reply Refer To: SC86-533 7 Naval
Rockwell International Corporation 1049 Camino Dos Rios Rockwell P.O. Box 1085 Thousand Oaks, California 91360 International C (805) 373-4545 AD-A220 233 March 18, 1986 In Reply Refer To: SC86-533 7 Naval
ADl-AO PUDUE UMIV LAFAYETTE IN DEPT OF CHEMISTRY F/6 7/4
 ADl-AO84 712 PUDUE UMIV LAFAYETTE IN DEPT OF CHEMISTRY F/6 7/4 UCLASSIFIED - PHIOTOCHEMICAL CONSEQUENCES OF INTERCHROMOPHORIC INTERACTICNS.(Ul MAY 80 H MORRISON OAA629-79 0 0009 ARO-13088.9-C NRL 6-SO
ADl-AO84 712 PUDUE UMIV LAFAYETTE IN DEPT OF CHEMISTRY F/6 7/4 UCLASSIFIED - PHIOTOCHEMICAL CONSEQUENCES OF INTERCHROMOPHORIC INTERACTICNS.(Ul MAY 80 H MORRISON OAA629-79 0 0009 ARO-13088.9-C NRL 6-SO
FINAL REPORT. Richard H. Clarke Department of Chemistry Boston University. February 25, U.S. Army Research Office DAAG K-0124
 9" f W.-F '..1. SPECTROSCOPIC INVESTIGATIONS OF ORGANIC MOLECULES ON GRAPHITE SURFACES FINAL REPORT CO I Richard H. Clarke Department of Chemistry Boston University February 25, 1986 V- - U.S. Army Research
9" f W.-F '..1. SPECTROSCOPIC INVESTIGATIONS OF ORGANIC MOLECULES ON GRAPHITE SURFACES FINAL REPORT CO I Richard H. Clarke Department of Chemistry Boston University February 25, 1986 V- - U.S. Army Research
MICROCOPY RESOLUIION IESI CHARI. NAMfNA[ IC~ ANtn
 A0-A107 459 NEW YORK( UNIV NY COURANT INST OF MATHEMATICAL SCIENCE-S F/6 20/11 RESEARCH IN C ONTI NUUM MECANICS- C-30 SIIDNOV 81 F JOHN N5001V76C-130NL CHNCS( UNLASIIE 14"' '2."8 MICROCOPY RESOLUIION IESI
A0-A107 459 NEW YORK( UNIV NY COURANT INST OF MATHEMATICAL SCIENCE-S F/6 20/11 RESEARCH IN C ONTI NUUM MECANICS- C-30 SIIDNOV 81 F JOHN N5001V76C-130NL CHNCS( UNLASIIE 14"' '2."8 MICROCOPY RESOLUIION IESI
7 AD-AlIft 398 VERMONT UNIV BURLINGTON DEPT OF CHEMISTRY F/B 7/3 A REINVESTIGATION OF THE REACTIONS OF ENOLATE ANIONS WITH CYCLO--ETC(U)
 7 AD-AlIft 398 VERMONT UNIV BURLINGTON DEPT OF CHEMISTRY F/B 7/3 A REINVESTIGATION OF THE REACTIONS OF ENOLATE ANIONS WITH CYCLO--ETC(U) h UCSEP &I C W ALLEN, K RANACHANRAN, R P BRIGHT N0OOV-77-C-0605
7 AD-AlIft 398 VERMONT UNIV BURLINGTON DEPT OF CHEMISTRY F/B 7/3 A REINVESTIGATION OF THE REACTIONS OF ENOLATE ANIONS WITH CYCLO--ETC(U) h UCSEP &I C W ALLEN, K RANACHANRAN, R P BRIGHT N0OOV-77-C-0605
Toward a Better Understanding of Ocean-Wave-Typhoon Interactions in the Western Pacific Ocean
 DISTRIBUTION STATEMENT A. Approved for public release; distribution is unlimited. Toward a Better Understanding of Ocean-Wave-Typhoon Interactions in the Western Pacific Ocean Shenn-Yu Chao Horn Point
DISTRIBUTION STATEMENT A. Approved for public release; distribution is unlimited. Toward a Better Understanding of Ocean-Wave-Typhoon Interactions in the Western Pacific Ocean Shenn-Yu Chao Horn Point
REGENERATION OF SPENT ADSORBENTS USING ADVANCED OXIDATION (PREPRINT)
 AL/EQ-TP-1993-0307 REGENERATION OF SPENT ADSORBENTS USING ADVANCED OXIDATION (PREPRINT) John T. Mourand, John C. Crittenden, David W. Hand, David L. Perram, Sawang Notthakun Department of Chemical Engineering
AL/EQ-TP-1993-0307 REGENERATION OF SPENT ADSORBENTS USING ADVANCED OXIDATION (PREPRINT) John T. Mourand, John C. Crittenden, David W. Hand, David L. Perram, Sawang Notthakun Department of Chemical Engineering
The Radiological Hazard of Plutonium Isotopes and Specific Plutonium Mixtures
 LA-13011 The Radiological Hazard of Plutonium Isotopes and Specific Plutonium Mixtures Los Alamos NATIONAL LABORATORY Los Alamos National Laboratory is operated by the University of California for the
LA-13011 The Radiological Hazard of Plutonium Isotopes and Specific Plutonium Mixtures Los Alamos NATIONAL LABORATORY Los Alamos National Laboratory is operated by the University of California for the
THE UNIVERSITY OF CONNECTICUT SCHOOL OF ENGINEERING 11E MOTION OF A PLANE EVAPORATION FRONT IN A SUPERHEATED LIQUID R. S. BRAND
 THE UNIVERSITY OF CONNECTICUT Q SCHOOL OF ENGINEERING q 7 TECHNICAL REPORT NO. 2 11E MOTION OF A PLANE EVAPORATION FRONT IN A SUPERHEATED LIQUID by R. S. BRAND MECHANICAL ENGINEERING DEPARTMENT This research
THE UNIVERSITY OF CONNECTICUT Q SCHOOL OF ENGINEERING q 7 TECHNICAL REPORT NO. 2 11E MOTION OF A PLANE EVAPORATION FRONT IN A SUPERHEATED LIQUID by R. S. BRAND MECHANICAL ENGINEERING DEPARTMENT This research
geeeo. I UNCLASSIFIED NO 814-B5-K-88 F/G 716 NL SCENCESECTION L F HANCOCK ET AL 81 AUG 87 TR-i
 -AiS4 819 PROTON ABSTRACTION AS A ROUTE TO CONDUCTIVE POLYMERS 1/1 (U) PENNSYLVANIA STATE UNIV UNIVERSITY PARK PA POLYMER SCENCESECTION L F HANCOCK ET AL 81 AUG 87 TR-i I UNCLASSIFIED NO 814-B5-K-88 F/G
-AiS4 819 PROTON ABSTRACTION AS A ROUTE TO CONDUCTIVE POLYMERS 1/1 (U) PENNSYLVANIA STATE UNIV UNIVERSITY PARK PA POLYMER SCENCESECTION L F HANCOCK ET AL 81 AUG 87 TR-i I UNCLASSIFIED NO 814-B5-K-88 F/G
I~~. Iill. JW lg. L3. IInn. ,. Lk 2l I1-0 -I125 A MICROCOPY RESOLUTION TEST CHART q. -ANDA'PDS, lq. r. 7Y 147
 -A182 516 TORSIONAL NOTION IN AROMATIC MOLECULES CONFORMATIONAL ANALYSIS OF METHYL- (U) COLORADO STATE UNIV FORT COLLINS DEPT OF CHEMISTRY P J BREEN ET AL MAY 87 UNCLASSIFIED TR-38 NOM 14-79-C-8647 F/G
-A182 516 TORSIONAL NOTION IN AROMATIC MOLECULES CONFORMATIONAL ANALYSIS OF METHYL- (U) COLORADO STATE UNIV FORT COLLINS DEPT OF CHEMISTRY P J BREEN ET AL MAY 87 UNCLASSIFIED TR-38 NOM 14-79-C-8647 F/G
DEPARTMENT OF THE ARMY WATERWAYS EXPERIMENT STATION. CORPS OF ENGINEERS P. 0. BOX 631 VICKSBURG. MISSISSIPPI 39180
 DEPARTMENT OF THE ARMY WATERWAYS EXPERIMENT STATION. CORPS OF ENGINEERS P. 0. BOX 631 VICKSBURG. MISSISSIPPI 39180 IN REPLY REF6R TO: WESYV 31 July 1978 SUBJECT: Transmittal of Technical Report D-78-34
DEPARTMENT OF THE ARMY WATERWAYS EXPERIMENT STATION. CORPS OF ENGINEERS P. 0. BOX 631 VICKSBURG. MISSISSIPPI 39180 IN REPLY REF6R TO: WESYV 31 July 1978 SUBJECT: Transmittal of Technical Report D-78-34
UNCLASSIFIED F/S 28/iS NI
 AD-R158 6680 POSSIBLE he HRNISMS OF RTOM TRANSFER IN SCANNING / TUNNELING MICROSCOPY(U) CHICAGO UNIV IL JAMES FRANCK INST R GONER JUL 85 N@814-77-C-88:18 UNCLASSIFIED F/S 28/iS NI 11111o 1.0.02 IL25L 1386
AD-R158 6680 POSSIBLE he HRNISMS OF RTOM TRANSFER IN SCANNING / TUNNELING MICROSCOPY(U) CHICAGO UNIV IL JAMES FRANCK INST R GONER JUL 85 N@814-77-C-88:18 UNCLASSIFIED F/S 28/iS NI 11111o 1.0.02 IL25L 1386
IiII II I I 1. III~i AD-A t \> NOV E THERMALLY REGENERATIVE FUEL CELLS OCTOBER 1991 CONTRACT N C-0071
 AD-A242 900 0- IiII II I I 1 III~i THERMALLY REGENERATIVE FUEL CELLS CONTRACT N00014-89-C-0071 ' J A) \> NOV 2719910 E FINAL REPORT OCTOBER 1991 PREPARED FOR: OFFICE OF NAVAL RESEARCH DEPARTMENT OF THE
AD-A242 900 0- IiII II I I 1 III~i THERMALLY REGENERATIVE FUEL CELLS CONTRACT N00014-89-C-0071 ' J A) \> NOV 2719910 E FINAL REPORT OCTOBER 1991 PREPARED FOR: OFFICE OF NAVAL RESEARCH DEPARTMENT OF THE
Eu... p UNCLASSIFIED ARO-i72672-EG DAAO2-8-C-i42 F/G 21/2 NL. D-A INVESTIGATION OF THE KINETICS OF CATALYZED COMBUSTION i/i
 I D-A142 153 INVESTIGATION OF THE KINETICS OF CATALYZED COMBUSTION i/i OF FUEL(U) UNITED TECHNOLOGIES RESEARCH CENTER EAST I HARTFORD CT P J IARTENEY 04 MAY 84 UTRC/955279-i p UNCLASSIFIED ARO-i72672-EG
I D-A142 153 INVESTIGATION OF THE KINETICS OF CATALYZED COMBUSTION i/i OF FUEL(U) UNITED TECHNOLOGIES RESEARCH CENTER EAST I HARTFORD CT P J IARTENEY 04 MAY 84 UTRC/955279-i p UNCLASSIFIED ARO-i72672-EG
FRACTAL CONCEPTS AND THE ANALYSIS OF ATMOSPHERIC PROCESSES
 1 FRACTAL CONCEPTS AND THE ANALYSIS OF ATMOSPHERIC PROCESSES Robert L. Street Environmental Fluid Mechanics Laboratory Department of Civil Engineering Stanford University Stanford, CA 94305-4020 650-723-4969;
1 FRACTAL CONCEPTS AND THE ANALYSIS OF ATMOSPHERIC PROCESSES Robert L. Street Environmental Fluid Mechanics Laboratory Department of Civil Engineering Stanford University Stanford, CA 94305-4020 650-723-4969;
OF NAVAL RESEARCH. Technical Report No. 87. Prepared for Publication. in the.. v.. il and/or -- c Spocial Journal of Elactroanalytical Chemistry it
 ,~OFFICE OF NAVAL RESEARCH Contract N00014-86-K-0556 Technical Report No. 87. Th., Combined Influence of Solution Resistance and Charge-Transfer Kinetics on Microelectrode Cyclic Voltammetry oo i Acee'ic
,~OFFICE OF NAVAL RESEARCH Contract N00014-86-K-0556 Technical Report No. 87. Th., Combined Influence of Solution Resistance and Charge-Transfer Kinetics on Microelectrode Cyclic Voltammetry oo i Acee'ic
ELECTRON BEAM AND PULSED CORONA PROCESSING OF VOLATILE ORGANIC COMPOUNDS AND NITROGEN OXIDES
 ELECTRON BEAM AND PULSED CORONA PROCESSING OF VOLATILE ORGANIC COMPOUNDS AND NITROGEN OXIDES B. M. Penetrante, M. C. Hsiao, J. N. Bardsley, B. T. Merritt, G. E. Vogtlin and P. H. Wallman Lawrence Livermore
ELECTRON BEAM AND PULSED CORONA PROCESSING OF VOLATILE ORGANIC COMPOUNDS AND NITROGEN OXIDES B. M. Penetrante, M. C. Hsiao, J. N. Bardsley, B. T. Merritt, G. E. Vogtlin and P. H. Wallman Lawrence Livermore
The chemiluminescent peroxyoxalate system: state of the art almost 50 years from its discovery
 The chemiluminescent peroxyoxalate system: state of the art almost 50 years from its discovery Luiz Francisco M. L. Ciscato, a,c* Felipe A. Augusto, a Dieter Weiss, b Fernando H. Bartoloni, c Steffen Albrecht
The chemiluminescent peroxyoxalate system: state of the art almost 50 years from its discovery Luiz Francisco M. L. Ciscato, a,c* Felipe A. Augusto, a Dieter Weiss, b Fernando H. Bartoloni, c Steffen Albrecht
IMPROVED SYNTHESIS AND REACTION CHEMISTRY OF FN 3
 IMPROVED SYNTHESIS AND REACTION CHEMISTRY OF FN 3 William W. Wilson,, Karl O. Christe, Ashwani Vij, Ralf Haiges ERC, Inc and Propellants Branch, Propulsion Directorate, Air Force Research Laboratory, Edwards
IMPROVED SYNTHESIS AND REACTION CHEMISTRY OF FN 3 William W. Wilson,, Karl O. Christe, Ashwani Vij, Ralf Haiges ERC, Inc and Propellants Branch, Propulsion Directorate, Air Force Research Laboratory, Edwards
1.0~ O. 1*1.8.2IIII II5 uuu11~ I 1 I6 MICROCOPY RESOLUTION TEST CHART NATIONAL BUREAU OF STANDARDS 1963-A
 6AD-Ai5@ 148 NUMERICAL ALGORITHMS AND SOFTWARE FOR MATRIX.RICCATI i/i EGUATIONS(U) UNIVERSITY OF SOUTHERN CALIFORNIA LOS ANGELES A J LAUB 25 OCT 84 RlRO-i8289 15-MAl UNCLASSIFIED MEOMONEE OAAG29-i-K-8i3i
6AD-Ai5@ 148 NUMERICAL ALGORITHMS AND SOFTWARE FOR MATRIX.RICCATI i/i EGUATIONS(U) UNIVERSITY OF SOUTHERN CALIFORNIA LOS ANGELES A J LAUB 25 OCT 84 RlRO-i8289 15-MAl UNCLASSIFIED MEOMONEE OAAG29-i-K-8i3i
FUNDAMENTAL STUDIES OF THE CATALYTIC IGNITION PROCESS
 FUNDAMENTAL STUDIES OF THE CATALYTIC IGNITION PROCESS Final Report KLK411 N08-03 National Institute for Advanced Transportation Technology University of Idaho Robert Lounsbury; Katrina Leichliter; and
FUNDAMENTAL STUDIES OF THE CATALYTIC IGNITION PROCESS Final Report KLK411 N08-03 National Institute for Advanced Transportation Technology University of Idaho Robert Lounsbury; Katrina Leichliter; and
OFFICE OF NAVAL RESEARCH. Contract Nonr-4756(07) Task No. NR Determination of the Rate Constants
 OFFICE OF NAVAL RESEARCH a Contract Nonr-4756(07) Task No. NR 015-816 CD O I - J > HI l! Determination of the Rate Constants of the Intersyntem Crossing Processes to the Individual Multlplets of the Lowest
OFFICE OF NAVAL RESEARCH a Contract Nonr-4756(07) Task No. NR 015-816 CD O I - J > HI l! Determination of the Rate Constants of the Intersyntem Crossing Processes to the Individual Multlplets of the Lowest
REPORT DOCUMENTATION PAGE
 REPORT DOCUMENTATION PAGE Form Approved OMB NO. 0704-0188 Public Reporting burden for this collection of information is estimated to average 1 hour per response, including the time for reviewing instructions,
REPORT DOCUMENTATION PAGE Form Approved OMB NO. 0704-0188 Public Reporting burden for this collection of information is estimated to average 1 hour per response, including the time for reviewing instructions,
AIRFOIL DESIGN PROCEDURE A MODIFIED THEODORSEN E-FUNCTION. by Raymond L. Barger. Langley Research Center NASA TECHNICAL NOTE NASA TN D-7741
 NASA TECHNICAL NOTE NASA TN D-7741 AND 1- I- 6~7 (NASA-TN-D-7741) A MODIFIED THEODORSEN N74-33428 EPSILON-FUNCTION AIRFOIL DESIGN PROCEDURE (NASA) 19 p BC $3.00 CSCL 01A Unclas H1/01 48910 rr A MODIFIED
NASA TECHNICAL NOTE NASA TN D-7741 AND 1- I- 6~7 (NASA-TN-D-7741) A MODIFIED THEODORSEN N74-33428 EPSILON-FUNCTION AIRFOIL DESIGN PROCEDURE (NASA) 19 p BC $3.00 CSCL 01A Unclas H1/01 48910 rr A MODIFIED
Second Interim Report NTIS GRA&I. 0 Michael Grttzel, Ph.D. By. Professor of Chemistry. June 20, United States Army
 D FILE COPY CATALYTIC AGENT DEGRADATION ON OXIDE LI 8 AND IN MCROHETEROGENEOUS SOLU'ION SYSTEMS cj Accession For Second Interim Report NTIS GRA&I by DTIC TAB Unannounced Justification 0 Michael Grttzel,
D FILE COPY CATALYTIC AGENT DEGRADATION ON OXIDE LI 8 AND IN MCROHETEROGENEOUS SOLU'ION SYSTEMS cj Accession For Second Interim Report NTIS GRA&I by DTIC TAB Unannounced Justification 0 Michael Grttzel,
I UNCLASSIFIED ONR-TR-2 NL
 I AD-A083 615 WASHINGTON UNIV SEATTLE DEPT OF ELECTRICAL ENGINEERING F/G 4/1 I MULTIPLE SC ATTERING EFFECTS ON TRANSMISSION THROUGH THE ATMOSPH-ETC(U) IAPR GO0 A I SHIMARU NOOGI#-78C50723 I UNCLASSIFIED
I AD-A083 615 WASHINGTON UNIV SEATTLE DEPT OF ELECTRICAL ENGINEERING F/G 4/1 I MULTIPLE SC ATTERING EFFECTS ON TRANSMISSION THROUGH THE ATMOSPH-ETC(U) IAPR GO0 A I SHIMARU NOOGI#-78C50723 I UNCLASSIFIED
00 Lfl. Technical Report No. 8 OFFICE OF NAVAL RESEARCH. Contract NO J R & T Code I
 00 Lfl 00 0 o OFFICE OF NAVAL RESEARCH Contract NO0014-87-J-11 18 R & T Code 413301I Technical Report No. 8 A Raman Spectroscopic and Electrochemical Study on Photoinduced Crystal Growth of DABCO Polylodide
00 Lfl 00 0 o OFFICE OF NAVAL RESEARCH Contract NO0014-87-J-11 18 R & T Code 413301I Technical Report No. 8 A Raman Spectroscopic and Electrochemical Study on Photoinduced Crystal Growth of DABCO Polylodide
EEEJ AA CHICAGO UNIV ILL DEPT OF CHEMISTRY F/6 7/4 ION-ION NEUTRALIZATION PROCESSES AND THEIR CONSEQUENCES. (U)
 AA099 05 CHICAGO UNIV ILL DEPT OF CHEMISTRY F/6 7/4 ION-ION NEUTRALIZATION PROCESSES AND THEIR CONSEQUENCES. (U) EEEJ MA Y 81 R S BERRY DAAG29RO0-K-0016 UNCLASSIFIED 880 169451C NL UNCLASS!FIED, SECUI4ITY
AA099 05 CHICAGO UNIV ILL DEPT OF CHEMISTRY F/6 7/4 ION-ION NEUTRALIZATION PROCESSES AND THEIR CONSEQUENCES. (U) EEEJ MA Y 81 R S BERRY DAAG29RO0-K-0016 UNCLASSIFIED 880 169451C NL UNCLASS!FIED, SECUI4ITY
Chemiluminescence-Generating Nanoreactor Formulation for Near- Infrared Imaging of Hydrogen Peroxide and Glucose Level In Vivo
 Supporting Information Advanced Functional Materials Chemiluminescence-Generating Nanoreactor Formulation for Near- Infrared Imaging of Hydrogen Peroxide and Glucose Level In Vivo Chang-Keun Lim, 1 Yong-Deok
Supporting Information Advanced Functional Materials Chemiluminescence-Generating Nanoreactor Formulation for Near- Infrared Imaging of Hydrogen Peroxide and Glucose Level In Vivo Chang-Keun Lim, 1 Yong-Deok
Report Documentation Page
 Report Documentation Page Form Approved OMB No. 0704-0188 Public reporting burden for the collection of information is estimated to average 1 hour per response, including the time for reviewing instructions,
Report Documentation Page Form Approved OMB No. 0704-0188 Public reporting burden for the collection of information is estimated to average 1 hour per response, including the time for reviewing instructions,
ILLINOIS STATE WATER SURVEY at the University of Illinois Urbana, Illinois
 ILLINOIS STATE WATER SURVEY at the University of Illinois Urbana, Illinois INVESTIGATION OF THE QUANTITATIVE DETERMINATION OF POINT AND AREAL PRECIPITATION BY RADAR ECHO MEASUREMENTS Interim Report No.
ILLINOIS STATE WATER SURVEY at the University of Illinois Urbana, Illinois INVESTIGATION OF THE QUANTITATIVE DETERMINATION OF POINT AND AREAL PRECIPITATION BY RADAR ECHO MEASUREMENTS Interim Report No.
EE77Eh I DR? 4 GNRTO OF PHSHRU ESE PHOSPHORUS ANIDE RND L/I PHOSPHINE DERIVATIVE.. (U) DUKE UNIV DURHAM NC DEPT OF
 I DR? 4 GNRTO OF PHSHRU ESE PHOSPHORUS ANIDE RND L/I I PHOSPHINE DERIVATIVE.. (U) DUKE UNIV DURHAM NC DEPT OF EE77Eh CHEMISTRY L D QUIN V7 OCT 86 ARO-22945. 7-CH UNCLASSIFIED DRLO3-86-K-6628 F/G 7/3 NL
I DR? 4 GNRTO OF PHSHRU ESE PHOSPHORUS ANIDE RND L/I I PHOSPHINE DERIVATIVE.. (U) DUKE UNIV DURHAM NC DEPT OF EE77Eh CHEMISTRY L D QUIN V7 OCT 86 ARO-22945. 7-CH UNCLASSIFIED DRLO3-86-K-6628 F/G 7/3 NL
Sediment Acoustics. Award #: N Thrust Category: High-Frequency LONG-TERM GOAL
 Sediment Acoustics Robert D. Stoll Lamont-Doherty Earth Observatory of Columbia University Palisades, New York 10964 phone: (845) 365 8392 fax: (845) 365 8179 email: stoll@ldeo.columbia.edu Award #: N00014-94-1-0258
Sediment Acoustics Robert D. Stoll Lamont-Doherty Earth Observatory of Columbia University Palisades, New York 10964 phone: (845) 365 8392 fax: (845) 365 8179 email: stoll@ldeo.columbia.edu Award #: N00014-94-1-0258
REPORT DOCUMENTATION PAGE
 REPORT DOCUMENTATION PAGE Form Approved OMB No. 0704-0188 Public reporting burden for this collection of information is estimated to average 1 hour per response, including the time for reviewing instructions,
REPORT DOCUMENTATION PAGE Form Approved OMB No. 0704-0188 Public reporting burden for this collection of information is estimated to average 1 hour per response, including the time for reviewing instructions,
UNIVERSITY OF ILLINOIS - URBANA, ILLINOIS
 UNIVERSITY OF ILLINOIS - URBANA, ILLINOIS STATE MODELS OF ACTIVE RLC NETWORKS Ahmet Dervisoglu REPORT R - 237 DECEMBER,1964 COORDINATED SCIENCE LABORATORY UNIVERSITY OF ILLINOIS URBANA, ILLINOIS C o n
UNIVERSITY OF ILLINOIS - URBANA, ILLINOIS STATE MODELS OF ACTIVE RLC NETWORKS Ahmet Dervisoglu REPORT R - 237 DECEMBER,1964 COORDINATED SCIENCE LABORATORY UNIVERSITY OF ILLINOIS URBANA, ILLINOIS C o n
Quantitation and Ratio Determination of Uranium Isotopes in Water and Soil Using Inductively Coupled Plasma Mass Spectrometry (ICP-MS)
 Quantitation and Ratio Determination of Uranium Isotopes in Water and Soil Using Inductively Coupled Plasma Mass Spectrometry (ICP-MS) D.N. Kurk, T.E. Beegle, S.C. Spence and R.J. Swatski Report Documentation
Quantitation and Ratio Determination of Uranium Isotopes in Water and Soil Using Inductively Coupled Plasma Mass Spectrometry (ICP-MS) D.N. Kurk, T.E. Beegle, S.C. Spence and R.J. Swatski Report Documentation
AFRL-RW-EG-TP
 AFRL-RW-EG-TP-2011-018 Relational Information Space for Dynamic Systems Robert A. Murphey Air Force Research Laboratory, Munitions Directorate AFRL/RWG 101 West Eglin Boulevard Eglin AFB, FL 32542-6810
AFRL-RW-EG-TP-2011-018 Relational Information Space for Dynamic Systems Robert A. Murphey Air Force Research Laboratory, Munitions Directorate AFRL/RWG 101 West Eglin Boulevard Eglin AFB, FL 32542-6810
KULTIWAVELENGTH LASER PROPAGATION STUDY ~ III. Quarterly Progress Report No. h March 16, June 15, J. Richard Kerr
 CO o KULTIWAVELENGTH LASER PROPAGATION STUDY ~ III i> Quarterly Progress Report No. h March 16, 1971 - June 15, 1971 J. Richard Kerr Oregon Graduate Center for Study and Research Portland, Oregon July,
CO o KULTIWAVELENGTH LASER PROPAGATION STUDY ~ III i> Quarterly Progress Report No. h March 16, 1971 - June 15, 1971 J. Richard Kerr Oregon Graduate Center for Study and Research Portland, Oregon July,
UNIVERSITY OF CALIFORNIA, SAN DIEGO SCRIPPS INSTITUTION OF OCEANOGRAPHY VISIBILITY LABORATORY SAN DIEGO, CALIFORNIA 92152
 UNIVERSITY OF CALIFORNIA, SAN DIEGO SCRIPPS INSTITUTION OF OCEANOGRAPHY VISIBILITY LABORATORY SAN DIEGO, CALIFORNIA 92152 ATMOSPHERIC OPTICAL MEASUREMENTS IN THE VICINITY OF CRATER LAKE, OREGON. PART I.
UNIVERSITY OF CALIFORNIA, SAN DIEGO SCRIPPS INSTITUTION OF OCEANOGRAPHY VISIBILITY LABORATORY SAN DIEGO, CALIFORNIA 92152 ATMOSPHERIC OPTICAL MEASUREMENTS IN THE VICINITY OF CRATER LAKE, OREGON. PART I.
SECURITY CLASSIFICATION OF TWI",A,'- Form Approved. ICUMENTATION'PAGE OMB No b. RESTRICTIVE MARKINGS OF
 SECURITY CLASSIFICATION OF TWI",A,'- Form Approved ICUMENTATION'PAGE OMB No. 0704-0188 AD- A207 216 1b. RESTRICTIVE MARKINGS OF 3. DISTRIBUTION/AVAILABILITY OF REPORT Approved for puli c release b. DECLASSiFICATION
SECURITY CLASSIFICATION OF TWI",A,'- Form Approved ICUMENTATION'PAGE OMB No. 0704-0188 AD- A207 216 1b. RESTRICTIVE MARKINGS OF 3. DISTRIBUTION/AVAILABILITY OF REPORT Approved for puli c release b. DECLASSiFICATION
UNCLASSIFIED ADL DEFENSE DOCUMENTATION CENTER FOR SCIENTIFIC AND TECHNICAL INFORMATION CAMERON STATION ALEXANDRIA. VIRGINIA UNCLASSIFIED
 UNCLASSIFIED ADL 4 5 2981 DEFENSE DOCUMENTATION CENTER FOR SCIENTIFIC AND TECHNICAL INFORMATION CAMERON STATION ALEXANDRIA. VIRGINIA UNCLASSIFIED NOTICE: When goverment or other drawings, specifications
UNCLASSIFIED ADL 4 5 2981 DEFENSE DOCUMENTATION CENTER FOR SCIENTIFIC AND TECHNICAL INFORMATION CAMERON STATION ALEXANDRIA. VIRGINIA UNCLASSIFIED NOTICE: When goverment or other drawings, specifications
NAVAL POSTGRADUATE SCHOOL
 NPS55-87-015 NAVAL POSTGRADUATE SCHOOL Monterey, California AN OPTIMAL BRANCH-AND-BOUND PROCEDURE FOR THE CONSTRAINED PATH, MOVING TARGET SEARCH PROBLEM JAMES N. EAGLE JAMES R. YEE DECEMBER 1987 Approved
NPS55-87-015 NAVAL POSTGRADUATE SCHOOL Monterey, California AN OPTIMAL BRANCH-AND-BOUND PROCEDURE FOR THE CONSTRAINED PATH, MOVING TARGET SEARCH PROBLEM JAMES N. EAGLE JAMES R. YEE DECEMBER 1987 Approved
DISTRIBUTION A: Distribution approved for public release.
 AFRL-AFOSR-UK-TR-2016-0035 Solar Wind Earth Exchange Project 140200 Steven Sembay UNIVERSITY OF LEICESTER 10/28/2016 Final Report DISTRIBUTION A: Distribution approved for public release. Air Force Research
AFRL-AFOSR-UK-TR-2016-0035 Solar Wind Earth Exchange Project 140200 Steven Sembay UNIVERSITY OF LEICESTER 10/28/2016 Final Report DISTRIBUTION A: Distribution approved for public release. Air Force Research
Crowd Behavior Modeling in COMBAT XXI
 Crowd Behavior Modeling in COMBAT XXI Imre Balogh MOVES Research Associate Professor ilbalogh@nps.edu July 2010 831-656-7582 http://movesinstitute.org Report Documentation Page Form Approved OMB No. 0704-0188
Crowd Behavior Modeling in COMBAT XXI Imre Balogh MOVES Research Associate Professor ilbalogh@nps.edu July 2010 831-656-7582 http://movesinstitute.org Report Documentation Page Form Approved OMB No. 0704-0188
An Examination of 3D Environmental Variability on Broadband Acoustic Propagation Near the Mid-Atlantic Bight
 An Examination of 3D Environmental Variability on Broadband Acoustic Propagation Near the Mid-Atlantic Bight Kevin B. Smith Code PH/Sk, Department of Physics Naval Postgraduate School Monterey, CA 93943
An Examination of 3D Environmental Variability on Broadband Acoustic Propagation Near the Mid-Atlantic Bight Kevin B. Smith Code PH/Sk, Department of Physics Naval Postgraduate School Monterey, CA 93943
Report Documentation Page
 Spectroscopic Challenges of Photoionized Plasmas ASP Conference Series, Vol. XXX, 2 Gary Ferland and Daniel Wolf Savin, eds. Photoionization Modeling of Infrared Fine-Structure Lines in Luminous Galaxies
Spectroscopic Challenges of Photoionized Plasmas ASP Conference Series, Vol. XXX, 2 Gary Ferland and Daniel Wolf Savin, eds. Photoionization Modeling of Infrared Fine-Structure Lines in Luminous Galaxies
LAGRANGIAN MEASUREMENTS OF EDDY CHARACTERISTICS IN THE CALIFORNIA CURRENT
 LAGRANGIAN MEASUREMENTS OF EDDY CHARACTERISTICS IN THE CALIFORNIA CURRENT Robert C. Beardsley, Kenneth H. Brink, Richard Limeburner Clark Laboratory, Mail Stop 21 Woods Hole Oceanographic Institution Woods
LAGRANGIAN MEASUREMENTS OF EDDY CHARACTERISTICS IN THE CALIFORNIA CURRENT Robert C. Beardsley, Kenneth H. Brink, Richard Limeburner Clark Laboratory, Mail Stop 21 Woods Hole Oceanographic Institution Woods
1SEP 01 J S LANGER AFOSR
 AD-A113 871 CARNEGIE-MELLON UNIV PITTSBURGH PA DEPT OF PHYSICS PG2/ I PH4ASE TRANSFORMATIONS AND NONEQUILIBRIUM INTERFACES.CW) 1SEP 01 J S LANGER AFOSR-80-0034 UNCLASSIFIED AFOSR-TR-82-0307 NI. Unclassified
AD-A113 871 CARNEGIE-MELLON UNIV PITTSBURGH PA DEPT OF PHYSICS PG2/ I PH4ASE TRANSFORMATIONS AND NONEQUILIBRIUM INTERFACES.CW) 1SEP 01 J S LANGER AFOSR-80-0034 UNCLASSIFIED AFOSR-TR-82-0307 NI. Unclassified
Metrology Experiment for Engineering Students: Platinum Resistance Temperature Detector
 Session 1359 Metrology Experiment for Engineering Students: Platinum Resistance Temperature Detector Svetlana Avramov-Zamurovic, Carl Wick, Robert DeMoyer United States Naval Academy Abstract This paper
Session 1359 Metrology Experiment for Engineering Students: Platinum Resistance Temperature Detector Svetlana Avramov-Zamurovic, Carl Wick, Robert DeMoyer United States Naval Academy Abstract This paper
Evolution of Tropical Cyclone Characteristics
 Evolution of Tropical Cyclone Characteristics Patrick A. Harr Department of Meteorology Naval Postgraduate School Monterey, CA 93943-5114 Telephone : (831) 656-3787 FAX: (831) 656-3061 email: paharr@nps.navy.mil
Evolution of Tropical Cyclone Characteristics Patrick A. Harr Department of Meteorology Naval Postgraduate School Monterey, CA 93943-5114 Telephone : (831) 656-3787 FAX: (831) 656-3061 email: paharr@nps.navy.mil
R&T Code b41c011dar. Technical Report No. 40. Synthesis, Characterization, and Electropolymerization of Ferrocene Substituted Anilines.
 AD-A240 866 SE.E T E R SEP 2 0 1991 "r r OFFICE OF NAVAL RESEARCH Contract No. N00014-88-K-0732 R&T Code b41c011dar Technical Report No. 40 Synthesis, Characterization, and Electropolymerization of Ferrocene
AD-A240 866 SE.E T E R SEP 2 0 1991 "r r OFFICE OF NAVAL RESEARCH Contract No. N00014-88-K-0732 R&T Code b41c011dar Technical Report No. 40 Synthesis, Characterization, and Electropolymerization of Ferrocene
CHEMICAL KINETICS C.H. BAMFORD C.F.H. TIPPER WSSSKUH EDITED BY
 CHEMICAL KINETICS EDITED BY C.H. BAMFORD M.A., Ph.D., Sc.D. (Cantab.), F.R.I.C., F.R.S. Campbell-Brown Professor of Industrial Chemistry, Uniuersity of Liverpool AND C.F.H. TIPPER Ph.D. (Bristol), D.Sc.
CHEMICAL KINETICS EDITED BY C.H. BAMFORD M.A., Ph.D., Sc.D. (Cantab.), F.R.I.C., F.R.S. Campbell-Brown Professor of Industrial Chemistry, Uniuersity of Liverpool AND C.F.H. TIPPER Ph.D. (Bristol), D.Sc.
DISTRIBUTION A: Distribution approved for public release.
 AFRL-AFOSR-JP-TR-2017-0059 Nanostructured catalytic hybrid materials Kap Seung Yang UNIVERSITY INDUSTRY LIAISON OF CHONNAM NATIONAL UN 08/28/2017 Final Report DISTRIBUTION A: Distribution approved for
AFRL-AFOSR-JP-TR-2017-0059 Nanostructured catalytic hybrid materials Kap Seung Yang UNIVERSITY INDUSTRY LIAISON OF CHONNAM NATIONAL UN 08/28/2017 Final Report DISTRIBUTION A: Distribution approved for
Alcohols Oxidation by oxygen O 2 in presence of vanadoheteropolyacid (H 5 PMo 10 V 2 O 40 ) as green catalyst
 International Journal of ChemTech Research CODEN( USA): IJCRGG ISSN : 0974-4290 Vol.2, No.3, pp 1666-1672, July-Sept 2010 Alcohols Oxidation by oxygen O 2 in presence of vanadoheteropolyacid (H 5 PMo 10
International Journal of ChemTech Research CODEN( USA): IJCRGG ISSN : 0974-4290 Vol.2, No.3, pp 1666-1672, July-Sept 2010 Alcohols Oxidation by oxygen O 2 in presence of vanadoheteropolyacid (H 5 PMo 10
Singlet Oxygen. Laura Calvo Parra Denmark Group Meeting February 21, 2017
 Singlet Oxygen Laura Calvo Parra Denmark Group Meeting February 21, 2017 Presentation Outline I. Introduction II. Electronic transitions III. Photosensitizers IV. Schenk-Ene reaction V. [4+2] and [2+2]
Singlet Oxygen Laura Calvo Parra Denmark Group Meeting February 21, 2017 Presentation Outline I. Introduction II. Electronic transitions III. Photosensitizers IV. Schenk-Ene reaction V. [4+2] and [2+2]
UNCLASSIFIED AD DEFENSE DOCUMENTATION CENTER FOR SCIENTIFIC AND TECHNICAL INFORMATION CAMERON STATION, ALEXANDRIA, VIRGINIA UNCLASSIFIED
 UNCLASSIFIED AD 409 815 DEFENSE DOCUMENTATION CENTER FOR SCIENTIFIC AND TECHNICAL INFORMATION CAMERON STATION, ALEXANDRIA, VIRGINIA UNCLASSIFIED NOTICE: When government or other drawings, specifications
UNCLASSIFIED AD 409 815 DEFENSE DOCUMENTATION CENTER FOR SCIENTIFIC AND TECHNICAL INFORMATION CAMERON STATION, ALEXANDRIA, VIRGINIA UNCLASSIFIED NOTICE: When government or other drawings, specifications
o J4 % o% 3.2. "w*, ',-'. %). %!- A 1.4. WIN F -,, a'p..
 AD-MI94 316 THE ELECTROMAGNETIC SPECTRUM - THE INFORMATION MINE(U) 1/1 ARIZONA UNIV TUCSON DEPT OF CHEMISTRY DENTON ET AL. 64 FEB 9B TR-60 900014-96-KC-0316 2UNcLfS S7IDFG 65M mmhhllhh % o% o J4 3.2. "w*,
AD-MI94 316 THE ELECTROMAGNETIC SPECTRUM - THE INFORMATION MINE(U) 1/1 ARIZONA UNIV TUCSON DEPT OF CHEMISTRY DENTON ET AL. 64 FEB 9B TR-60 900014-96-KC-0316 2UNcLfS S7IDFG 65M mmhhllhh % o% o J4 3.2. "w*,
C-) ISUMMARY REPORT LINEAR AND NONLINEAR ULTRASONIC INTERACTIONS ON LIQUID-SOLID BOUNDARIES
 AD-AI13 682 GEORGETOWN UF4IV WASHINGTON DC DEPT OF PHYSICS F/G 20/1 LINEAR AND NONLINEAR ULTRASONIC INTERACTIONS OH LIQUID-SOLID 9O--ETC(U) APR 82 A S WATER UNCLASSIFIED SUUS-042-S N0OOIA-78-C-0584 NL
AD-AI13 682 GEORGETOWN UF4IV WASHINGTON DC DEPT OF PHYSICS F/G 20/1 LINEAR AND NONLINEAR ULTRASONIC INTERACTIONS OH LIQUID-SOLID 9O--ETC(U) APR 82 A S WATER UNCLASSIFIED SUUS-042-S N0OOIA-78-C-0584 NL
DISYRI'3UTED BY: National Technical Information Service U. S. DEPARTMENT OF COMMERCE 5282 Port Royal Road, Springfield Va.
 AD-753 391 CHEMICAL MODIFICATIONS OF SILICA SURFACES Manfred J. D. Low New York University Prepared for: Army Research Office (Durham) 12 November 1972 DISYRI'3UTED BY: National Technical Information Service
AD-753 391 CHEMICAL MODIFICATIONS OF SILICA SURFACES Manfred J. D. Low New York University Prepared for: Army Research Office (Durham) 12 November 1972 DISYRI'3UTED BY: National Technical Information Service
Experimental and Theoretical Studies of Ice-Albedo Feedback Processes in the Arctic Basin
 LONG TERM GOALS Experimental and Theoretical Studies of Ice-Albedo Feedback Processes in the Arctic Basin D.K. Perovich J.A. Richter-Menge W.B. Tucker III M. Sturm U. S. Army Cold Regions Research and
LONG TERM GOALS Experimental and Theoretical Studies of Ice-Albedo Feedback Processes in the Arctic Basin D.K. Perovich J.A. Richter-Menge W.B. Tucker III M. Sturm U. S. Army Cold Regions Research and
DEPARTMENT: Chemistry
 CODE CHEM 204 TITLE: Organic Chemistry II INSTITUTE: STEM DEPARTMENT: Chemistry COURSE DESCRIPTION: A continuation of CHEM-203, students will extend their studies into topics including aromatic hydrocarbons,
CODE CHEM 204 TITLE: Organic Chemistry II INSTITUTE: STEM DEPARTMENT: Chemistry COURSE DESCRIPTION: A continuation of CHEM-203, students will extend their studies into topics including aromatic hydrocarbons,
IUNCLSSIME N E F/G 11?2 NL
 AD-RI58 658 BORON-NITROGEN POLYNERS(U) ULTRASYSTENS INC IRVINE CA III 1 K L PACIOREK ET AL. 01 JUL 85 SN-2822-F NS9914-82-C-9482 IUNCLSSIME N E F/G 11?2 NL 1.0 16 2.: w1111342 1.8 11111125 1111.4 L MICROCOPY
AD-RI58 658 BORON-NITROGEN POLYNERS(U) ULTRASYSTENS INC IRVINE CA III 1 K L PACIOREK ET AL. 01 JUL 85 SN-2822-F NS9914-82-C-9482 IUNCLSSIME N E F/G 11?2 NL 1.0 16 2.: w1111342 1.8 11111125 1111.4 L MICROCOPY
Chemiluminescence A Toast to Chemistry Chemiluminescence
 Chemiluminescence A Toast to Chemistry Chemiluminescence SCIENTIFIC Introduction Chemiluminescence demonstrations are popular with students and teachers alike. This activity demonstrates basic chemiluminescence
Chemiluminescence A Toast to Chemistry Chemiluminescence SCIENTIFIC Introduction Chemiluminescence demonstrations are popular with students and teachers alike. This activity demonstrates basic chemiluminescence
Comparative Analysis of Flood Routing Methods
 US Army Corps of Engineers Hydrologic Engineering Center Comparative Analysis of Flood Routing Methods September 1980 Approved for Public Release. Distribution Unlimited. RD-24 REPORT DOCUMENTATION PAGE
US Army Corps of Engineers Hydrologic Engineering Center Comparative Analysis of Flood Routing Methods September 1980 Approved for Public Release. Distribution Unlimited. RD-24 REPORT DOCUMENTATION PAGE
NAVAL POSTGRADUATE SCHOOL
 NPS-PH-07-001 NAVAL POSTGRADUATE SCHOOL MONTEREY, CALIFORNIA Correction to attenuation treatment in the Monterey- Miami Parabolic Equation Model by Kevin B. Smith Michael A. Wolfson A. Vincent van Leijen
NPS-PH-07-001 NAVAL POSTGRADUATE SCHOOL MONTEREY, CALIFORNIA Correction to attenuation treatment in the Monterey- Miami Parabolic Equation Model by Kevin B. Smith Michael A. Wolfson A. Vincent van Leijen
Exact Solution of a Constrained. Optimization Problem in Thermoelectric Cooling
 Applied Mathematical Sciences, Vol., 8, no. 4, 77-86 Exact Solution of a Constrained Optimization Problem in Thermoelectric Cooling Hongyun Wang Department of Applied Mathematics and Statistics University
Applied Mathematical Sciences, Vol., 8, no. 4, 77-86 Exact Solution of a Constrained Optimization Problem in Thermoelectric Cooling Hongyun Wang Department of Applied Mathematics and Statistics University
Reminder: These notes are meant to supplement, not replace, the textbook and lab manual. Electrophilic Aromatic Substitution notes
 Reminder: These notes are meant to supplement, not replace, the textbook and lab manual. Electrophilic Aromatic Substitution notes History and Application: The rate of a reaction directly impacts the commercial
Reminder: These notes are meant to supplement, not replace, the textbook and lab manual. Electrophilic Aromatic Substitution notes History and Application: The rate of a reaction directly impacts the commercial
Atovaquone: An Antipneumocystic Agent
 Atovaquone: An Antipneumocystic Agent Atovaquone is a pharmaceutical compound marketed in the United States under different combinations to prevent and treat pneumocystosis and malaria. In a report from
Atovaquone: An Antipneumocystic Agent Atovaquone is a pharmaceutical compound marketed in the United States under different combinations to prevent and treat pneumocystosis and malaria. In a report from
7 AD-A CALIFORNIA UNIV SANTA BARBARAF/ / HYDROGENATION OF CO ON ALUMINA SUPPORTED METALS: A TUNN!ELING SP-ETC (U)
 7 AD-A093 285 CALIFORNIA UNIV SANTA BARBARAF/ / HYDROGENATION OF CO ON ALUMINA SUPPORTED METALS: A TUNN!ELING SP-ETC (U) UNLSIID -DEC A0 R E ROEKER, P HANSMA, KASKA NO01D4 8-C 0OD ED %ECU.AITY CLASSIsVIC&Ttc.%
7 AD-A093 285 CALIFORNIA UNIV SANTA BARBARAF/ / HYDROGENATION OF CO ON ALUMINA SUPPORTED METALS: A TUNN!ELING SP-ETC (U) UNLSIID -DEC A0 R E ROEKER, P HANSMA, KASKA NO01D4 8-C 0OD ED %ECU.AITY CLASSIsVIC&Ttc.%
OFFICE OF NAVAL RESEARCH. Prepared for Publication. in the
 OFFICE OF NAVAL RESEARCH Task No. NR 056-607 / EHNICAL REST 0 16 u 1 I- "-.......,d CHARGE-TRANSFER INTERACTIONS BETWEEN TRANSITION METAL HEXAFLUORIDES AND XENON'. by J. D./Webb M E. R./Bernstein\ Prepared
OFFICE OF NAVAL RESEARCH Task No. NR 056-607 / EHNICAL REST 0 16 u 1 I- "-.......,d CHARGE-TRANSFER INTERACTIONS BETWEEN TRANSITION METAL HEXAFLUORIDES AND XENON'. by J. D./Webb M E. R./Bernstein\ Prepared
Extension of the BLT Equation to Incorporate Electromagnetic Field Propagation
 Extension of the BLT Equation to Incorporate Electromagnetic Field Propagation Fredrick M. Tesche Chalmers M. Butler Holcombe Department of Electrical and Computer Engineering 336 Fluor Daniel EIB Clemson
Extension of the BLT Equation to Incorporate Electromagnetic Field Propagation Fredrick M. Tesche Chalmers M. Butler Holcombe Department of Electrical and Computer Engineering 336 Fluor Daniel EIB Clemson
TO Approved for public release, distribution unlimited
 UNCLASSIFIED AD NUMBER AD463752 NEW LIMITATION CHANGE TO Approved for public release, distribution unlimited FROM Distribution authorized to U.S. Gov't. agencies and their contractors; Administrative/Operational
UNCLASSIFIED AD NUMBER AD463752 NEW LIMITATION CHANGE TO Approved for public release, distribution unlimited FROM Distribution authorized to U.S. Gov't. agencies and their contractors; Administrative/Operational
Earth Radii Used in Numerical Weather Models
 Naval Research Laboratory Monterey, CA 93943-5502 NRLMiRI7543--05-8888 Earth Radii Used in Numerical Weather Models Louis A. I-IEMBREE Meteorological Application Development Branch Marine Meteorology Division
Naval Research Laboratory Monterey, CA 93943-5502 NRLMiRI7543--05-8888 Earth Radii Used in Numerical Weather Models Louis A. I-IEMBREE Meteorological Application Development Branch Marine Meteorology Division
Aldehydes and Ketones : Aldol Reactions
 Aldehydes and Ketones : Aldol Reactions The Acidity of the a Hydrogens of Carbonyl Compounds: Enolate Anions Hydrogens on carbons a to carbonyls are unusually acidic The resulting anion is stabilized by
Aldehydes and Ketones : Aldol Reactions The Acidity of the a Hydrogens of Carbonyl Compounds: Enolate Anions Hydrogens on carbons a to carbonyls are unusually acidic The resulting anion is stabilized by
Supplementary Figure 1 XRD and Raman spectrum characterization of GQDs. a, XRD pattern of GQDs. b, Raman spectrum of GQDs, the appearance of the mode
 Supplementary Figure 1 XRD and Raman spectrum characterization of GQDs. a, XRD pattern of GQDs. b, Raman spectrum of GQDs, the appearance of the mode at 1456 cm -1 is not fully understood. Nevertheless,
Supplementary Figure 1 XRD and Raman spectrum characterization of GQDs. a, XRD pattern of GQDs. b, Raman spectrum of GQDs, the appearance of the mode at 1456 cm -1 is not fully understood. Nevertheless,
AD -A PAGE RMB No gathetriflg af *, t OnifrmatiOn sed commenti regaraing this.jdtn estimate or any other *%w ct of this
 1 form PAGE Approved AD -A272 994 4 PAGE RMB No. 0704-0188 gathetriflg af *, t OnifrmatiOn sed commenti regaraing this.jdtn estimate or any other *%w ct of this "a'oiiet'on of ;tton.4eacort i e ri S"rces.
1 form PAGE Approved AD -A272 994 4 PAGE RMB No. 0704-0188 gathetriflg af *, t OnifrmatiOn sed commenti regaraing this.jdtn estimate or any other *%w ct of this "a'oiiet'on of ;tton.4eacort i e ri S"rces.
