Metallocenes WILEY-VCH. Volume 2. Synthesis Reactivity Applications. Edited by Antonio Togni and Ronald L. Halterman
|
|
|
- Hugo Johnson
- 5 years ago
- Views:
Transcription
1 Metallocenes Volume 2 Synthesis Reactivity Applications Edited by Antonio Togni and Ronald L. Halterman WILEY-VCH Weinheim New York Chichester Brisbane Singapore Toronto /
2 Preface V Volume 1 Synthesis and Reactivity 1 Main Group Metallocenes P. Jutzi and N. Burford 1.1 Abstract Introduction Synthetic Procedures Reactions Involving Cyclopentadienes Salt Metathesis Reactions Disproportionation, Comproportionation and Decomposition Halide Ion Abstraction Substitution onto the Cp Ligand Frame Reduction Addition of Cp Anions Characterization Features Structural Types Sandwich Complexes Half Sandwich Complexes /i-rc-cyclopentadienyl Molecular Complexes ju-rc-cyclopentadienyl Super-Sandwich Polymers Dimers Clusters Cp-Element Orbital Interactions Orbital Interaction Model for Half Sandwich Complexes Orbital Interaction Model for Sandwich Complexes Metallocenes of the Alkali Metals 23 /
3 VI Contents Monomers and Polymers Lithocene and Sodocene Anions Metallocenes of the Alkaline Earth Metals Beryllocene Alkaline Earth Sandwich Metallocenes Half Sandwich Metallocenes of the Group 2 Elements Metallocenes of the Group 13 Elements Half Sandwich Monomers, Dimers, Oligomers and Polymers Clusters Metallocenes Involving Group 13 Elements in Oxidation State in Heteroatom Clusters Metallocenes of the Group 14 Elements Group 14 Sandwich Complexes Half Sandwich Complexes of Group 14 Elements Tricyclopentadienyl Complexes Reaction Chemistry Metallocenes of the Pnictogens Factors Governing the Structure of Multicyclopentadienyl Complexes Conclusions Acknowledgments 46 References 47 2 Lanthanocenes F. T. Edelmann 2.1 Introduction Lanthanide(II) Metallocenes Lanthanide(III) Metallocenes Lanthanide(III) Metallocenes Containing Ln-Group 17 Element Bonds Lanthanide(III) Metallocenes Containing Ln-Group 16 Element Bonds Lanthanide(III) Metallocenes Containing Ln-Group 15 Element Bonds Lanthanide(III) Metallocenes Containing Ln-Group 14 Element Bonds Lanthanide(III) Metallocenes Containing Ln-Hydrogen Bonds Conclusion and Perspective Acknowledgments 104 References 104
4 VII 3 Group 3 Metallocenes P. J. Chirik and J. E. Bercaw 3.1 Introduction Synthesis of Group 3 Metallocenes Scandium Yttrium and Lanthanum Group 3 Metallocenes for the Activation of Hydrocarbons Group 3 Metallocenes as Homogeneous Olefin Polymerization Catalysts Reactions of Group 3 Metallocenes with Acetylenes Reactions of Group 3 Metallocenes with Group 6 Compounds Reactions of Group 3 Metallocenes with Other Small Molecules Applications of Group 3 Metallocenes in Organic Synthesis Photophysics of Group 3 Metallocenes Alternatives of Cyclopentadienyl Ligands Concluding Remarks 147 References Titanocenes R. Beckhaus 4.1 Introduction Bonding Modes of Cyclopentadienyl Ligands in Titanium Complexes Synthesis of Titanocene Complexes Sources of the Titanocene Fragment as a Versatile Building Block Electronic Properties of Titanocenes Titanocene Complexes in Low Oxidation States Substituents on the Cp Rings Ligands in the Coordination Sphere of Titanocenes Olefins Acetylenes Carbonyls Phosphines Hydrides Titanium Ligand Multiple Bonds Carbene Complexes Complexes Containing Ti-X Double Bonds (X: N, P, Chalcogenides) Metallocene Complexes of the Type [Cp 2 TiL 2 ]
5 Aqua-Titanocene Complexe Titanocene Complexes with Highly Fluorinated Ligand Systems Further Complexes with [Titanocene] 2 " 1 " (d ) and [Titanocene]" 1 " (d 1 ) Units Reactions in the Coordination Sphere of Titanocenes The free Titanocenes Reactions of Ti-C a Bonds Reductive Elimination Oxidative Addition a-h Elimination ß-U Elimination Reactions of Ti=C Double Bonds Reactions with Electrophiles C-H Activation Reactions on [L n Ti=C] Fragments [2+1] Additions Cycloaddition Reactions with Acetylenes Synthesis and Reactivity of Metallaoxetanes Synthesis of Heterodinuclear Carbene Complexes with a Titanaoxetane Substructure Synthesis and Reactivity of Heterotitanacyclobutenes and-butanes Structure and Reactivity Pattern for small Titanacycles Dimetallic Compounds Derived from Ti=C Species Reactions Involving the Cp Ligand Analytical Data Structural Data NMR Data l H NMR Data C NMR Data Ti NMR Data UV vis Spectra Selected Applications of Titanocene Complexes C-C Coupling Reactions Dimerization of Ethylene Head to Tail Coupling Reactions of Acetylenes Cyclization Reactions Stoichiometric and Catalytic Reactions of Carbenoid Metal Complexes of Electron-Deficient Transition Metals Carbonyl Olefinations with Tebbe, Grubbs, and Petasis Reagents Ene Reactions Catalytic Reactions Cp-Equivalents Acknowledgments 230 References 230
6 IX 5 Zirconocenes E. Negishi andj.-l. Montchamp 5.1 Introduction Preparation of Cp 2 Zr Compounds Transmetallation Hydrozirconation Oxidative Addition Oxidative Coupling and Complexation Reactions and Synthetic Applications of Monoorganylzirconocene Derivatives Protonolysis, Halogenolysis, Oxidation, and Related Carbon-Heteroatom Bond Formation Reactions Protonolysis and Deuterolysis Halogenolysis Oxidation Carbon-Carbon Bond Formation via Polar Reactions of RZrCp 2 Cl with Carbon Electrophiles Reactions of Allylzirconocenes with Aldehydes Reactions of Cationic Alkyl- and Alkenylzirconocenes with Aldehydes, Epoxides, Ketones, and Nitriles Carbon-Carbon Bond Formation via Migratory Insertion Reactions of RZrCp 2 Cl Carbonylation Isonitrile Insertion Other Migratory Insertion Reactions Stoichiometric Carbozirconation of Monoorganylzirconocenes Reactions of Monoorganylzirconocenes via Transmetallation Stoichiometric Conversion of Monoorganylzirconocenes into Other Organometals Reactions of Monoorganylzirconocenes Catalyzed by Metal Complexes Organometallic Reactions Catalyzed by Zirconocene Derivatives Zirconium-Catalyzed Carboalumination and Carbozincation of Alkynes Zirconium-Catalyzed Hydroalumination and Hydroboration of Alkenes and Alkynes Zirconium-Catalyzed Enantioselective Carboalumination of Alkenes Reactions Involving n-complexes of Zirconocenes or Three- Membered Zirconacycles Formation of n-complexes of Zirconocenes 284
7 X Contents Stoichiometric Reactions of 71-Complexes of Zirconocenes (or Three-Membered Zirconacycles) Protonolysis and Halogenolysis Ring Expansion Reactions Regioisomerization, Stereoisomerization, cr-bond Metathesis, and Other Stoichiometric Reactions of Three-Membered Zirconacycles Stoichiometric Reactions of Five-Membered Zirconacycles Catalytic Reactions Involving Zirconacycles and Related Derivatives Zirconium-Catalyzed Carbomagnesiation Reactions Involving C-H Activation Zirconium-Catalyzed Carboalumination of Alkynes via C-H Activation Other Zirconium-Catalyzed Reactions Involving Cp 2 Zr(II) Complexes Conclusion 310 References Group 5 and Group 6 Metallocenes P. Royo and E. Ryan 6.1 Introduction Type A Metallocene Compounds Neutral Metallocenes Group 5 Metallocenes [MCp 2 ] Group 6 Metallocenes [MCp 2 ] Ionic Mettalocenes Group 5 Cationic Metallocenes [MCp 2 ] Group 5 Anionic Metallocenes [MCp 2 ]" Group 6 Cationic Metallocenes [MCp 2 ] Group 6 Anionic Metallocenes [MCp 2 ]' Type B-Bent bis(cyclopentadienyl) Metal Complexes with Mono-Functional Single-Atom Donor Monoanionic and Neutral Ligands Complexes with One Coordinated Ligand Neutral Group 6 [MCp 2 L] Metal Complexes Anionic Group 5 [MCp 2 L]" and Group 6 [MCp 2 X]" Metal Complexes Neutral Group 5 [MCp 2 L] and Group 6 [MCp 2 X] Metal Complexes 336
8 XI Anionic Group 5 [MCp2X]- and Cationic Group 6 [MCp 2 L] + Metal Complexes Neutral d 2 Vanadium Compounds [VCp 2 X] Neutral d 2 Niobium and Tantalum Compounds [(Nb,Ta)Cp 2 X] Cationic Group 5 d 2 Metal Compounds [MCp 2 L] Cationic Group 6 d 2 Metal Compounds [MCp 2 X] Complexes with Two Coordinated Ligands Neutral Group 5 d 2 Metal Compounds [MCp 2 XL] Cationic Group 5 d 2 Metal Compounds [MCp 2 L 2 ] Anionic Group 5 d 2 Metal Compounds [MCp 2 X 2 ]" Neutral Group 6 d 2 Metal Compounds [MCp 2 X 2 ] Cationic Group 6 d 2 Metal Compounds [MCp 2 XL] Dicationic Group 6 d 2 Metal Compounds [MCp 2 L 2 ] Neutral Group 5 d 1 Metal Compounds [MCp 2 X 2 ] Cationic Group 5 d 1 Metal Compounds [MCp 2 XL] +, [MCp 2 L 2 ] Cationic Group 6 d 1 Metal Compounds [MCp 2 X 2 ] Cationic Group 5 d Metal Compounds [MCp 2 X 2 ] +, [MCp 2 XL] Dicationic Group 6 d Metal Compounds [MCp 2 X 2 ] Complexes with Three Coordinated Ligands Neutral Group 5 d Metal Compounds [MCp 2 X 3 ] Cationic Group 5 d Metal Compounds [MCp 2 X 2 L] +, [MCp 2 XL 2 ] 2+ and [MCp 2 L 3 ] Cationic Group 6 d Metal Compounds [MCp 2 X 3 ] Type C - Complexes with Single-Atom Donor Dianionic Ligands Electron Compounds Neutral Group 5 d Metal Compounds [MCp 2 EX] Cationic Group 5 d Metal Compounds[MCp 2 EL] Neutral Group 6 d 2 Metal Compounds [MCp 2 E] Cationic Group 6 d Metal Compounds [MCp 2 EX] Electron Compounds Neutral d 1 Vanadium Compounds [VCp 2 E] Type D-Complexes with J7 2 -Side-Bonded Ligands Electron Compounds Neutral Group 5 d 2 Metal Compounds [MCp 2 (r7 2 -X-L)] Neutral Group 5 Metal Complexes [MCp 2 X(?7 2 -C-C)] or [MCp 2 X(?7 2 -C-Y)] (Y = N, O, or S) Anionic Group 5 d 2 Metal Compounds [MCp 2 (?7 2 -C,Y)] + and [MCp 2 X(77 2 -C,Y)] Neutral Group 6 d 2 Metal Compounds [MCp 2 (r7 2 -X-X)] Cationic Group 6 d 2 Metal Compounds [MCp 2 (?7 2 -X-L)] Neutral Group 5 d Metal Compounds [MCp 2 (?7 2 -X-X)Z] Cationic Group 5 d Metal Compounds [MCp^L]* and [MCp 2 (?7 2 -X-L)Z] + 401
9 Electron Compounds Neutral d 1 Vanadium Compounds [VCp 2 (r? 2 -X-X)] Niobium Compounds [NbCp 2 (T7 2 -L-L)] and [NbCp 2 (T7 2 -C,Y)] 404 References Half-Sandwich Complexes as Metallocene Analogs J. Okuda and T. Eberle 7.1 Introduction Complexes with Linked Amido-Cyclopentadienyl Ligands Synthesis Hydrido and Alkyl Complexes Reactivity Structure Complexes with Polydentate Amido-Cyclopentadienyl Ligands Complexes with two Amido-Cyclopentadienyl Ligands Complexes with Linked Alkoxo-Cyclopentadienyl Ligands Catalysis Polymerization of Ethylene, a-olefins and Dienes Hydrogenation and Hydroboration Conclusion 450 References Synthesis of Chirai Titanocene and Zirconocene Dichlorides R. L. Halterman 8.1 Introduction Ligand Chirality and Stereoisomeric Metal Complexes Unbridged Ligands Cyclopentadienes Indenes Bridged Ligands with Nonstereogenic Bridging Groups Bis(cyclopentadienes) Bis(indenes) Bis(fluorenes) Mixed Ligands Bridged Ligands with Stereogenic Bridging Groups Unbridged Metal Complexes Bis(cyclopentadienyl)metals 513
10 XIII Bis(indenes) Bis(fluorenes) Mixed Ligands Metal Complexes with Nonstereogenic Bridging Groups Bis(cyclopentadienyl)metals Bis(indenes) Bis(fluorenes) Mixed Ligands Metal Complexes with Stereogenic Bridging Groups 531 References 539
THE ORGANOMETALLIC CHEMISTRY OF THE TRANSITION METALS
 THE ORGANOMETALLIC CHEMISTRY OF THE TRANSITION METALS Second Edition ROBERT H. CRABTREE Yale University New Haven, Connecticut A Wiley-Interscience Publication JOHN WILEY & SONS New York / Chichester /
THE ORGANOMETALLIC CHEMISTRY OF THE TRANSITION METALS Second Edition ROBERT H. CRABTREE Yale University New Haven, Connecticut A Wiley-Interscience Publication JOHN WILEY & SONS New York / Chichester /
CONTENTS PART I STRUCTURES OF THE TRANSITION-METAL COMPLEXES
 CONTENTS Introduction... 1 1. Organization of the text... 1 2. Frontiers of organometallic chemistry... 2 3. Situation of the book with respect to teaching... 2 4. Reference books and other selected references...
CONTENTS Introduction... 1 1. Organization of the text... 1 2. Frontiers of organometallic chemistry... 2 3. Situation of the book with respect to teaching... 2 4. Reference books and other selected references...
Chem 634. Introduction to Transition Metal Catalysis. Reading: Heg Ch 1 2 CS-B 7.1, , 11.3 Grossman Ch 6
 Chem 634 Introduction to Transition etal Catalysis eading: eg Ch 1 2 CS-B 7.1, 8.2 8.3, 11.3 Grossman Ch 6 Announcements Problem Set 1 due Thurs, 9/24 at beginning of class ffice our: Wed, 10:30-12, 220
Chem 634 Introduction to Transition etal Catalysis eading: eg Ch 1 2 CS-B 7.1, 8.2 8.3, 11.3 Grossman Ch 6 Announcements Problem Set 1 due Thurs, 9/24 at beginning of class ffice our: Wed, 10:30-12, 220
Basic Organometallic Chemistry : Concepts, Syntheses, and Applications of Transition Metals. Table Of Contents: Foreword
 Basic Organometallic Chemistry : Concepts, Syntheses, and Applications of Transition Metals Table Of Contents: Foreword v Preface vii List of abbreviations ix Chapter 1 Introduction 1 (15) 1.1 What is
Basic Organometallic Chemistry : Concepts, Syntheses, and Applications of Transition Metals Table Of Contents: Foreword v Preface vii List of abbreviations ix Chapter 1 Introduction 1 (15) 1.1 What is
Reaction chemistry of complexes Three general forms: 1. Reactions involving the gain and loss of ligands a. Ligand Dissoc. and Assoc. (Bala) b.
 eaction chemistry of complexes Three general forms: 1. eactions involving the gain and loss of ligands a. Ligand Dissoc. and Assoc. (Bala) b. Oxidative Addition c. eductive Elimination d. Nucleophillic
eaction chemistry of complexes Three general forms: 1. eactions involving the gain and loss of ligands a. Ligand Dissoc. and Assoc. (Bala) b. Oxidative Addition c. eductive Elimination d. Nucleophillic
14-1 Reactions Involving Gain or Loss of Ligands Reactions Involving Modification of Ligands
 Organometallic Reaction and Catalysis 14-1 Reactions Involving Gain or Loss of Ligands 14-2 Reactions Involving Modification of Ligands 14-3 Organometallic Catalysts 14-4 Heterogeneous Catalysts Inorganic
Organometallic Reaction and Catalysis 14-1 Reactions Involving Gain or Loss of Ligands 14-2 Reactions Involving Modification of Ligands 14-3 Organometallic Catalysts 14-4 Heterogeneous Catalysts Inorganic
CHEM 251 (4 credits): Description
 CHEM 251 (4 credits): Intermediate Reactions of Nucleophiles and Electrophiles (Reactivity 2) Description: An understanding of chemical reactivity, initiated in Reactivity 1, is further developed based
CHEM 251 (4 credits): Intermediate Reactions of Nucleophiles and Electrophiles (Reactivity 2) Description: An understanding of chemical reactivity, initiated in Reactivity 1, is further developed based
ACTIVATION OF C H BONDS BY LOW-VALENT METAL COMPLEXES ( THE ORGANOMETALLIC CHEMISTRY )
 CHAPTER IV ACTIVATION OF C H BONDS BY LOW-VALENT METAL COMPLEXES ( THE ORGANOMETALLIC CHEMISTRY ) n the end of the 1960s the leading specialist in homogeneous catalysis Jack Halpern wrote [1]: to develop
CHAPTER IV ACTIVATION OF C H BONDS BY LOW-VALENT METAL COMPLEXES ( THE ORGANOMETALLIC CHEMISTRY ) n the end of the 1960s the leading specialist in homogeneous catalysis Jack Halpern wrote [1]: to develop
Detailed Course Content
 Detailed Course Content Chapter 1: Carbon Compounds and Chemical Bonds The Structural Theory of Organic Chemistry 4 Chemical Bonds: The Octet Rule 6 Lewis Structures 8 Formal Charge 11 Resonance 14 Quantum
Detailed Course Content Chapter 1: Carbon Compounds and Chemical Bonds The Structural Theory of Organic Chemistry 4 Chemical Bonds: The Octet Rule 6 Lewis Structures 8 Formal Charge 11 Resonance 14 Quantum
Contents. Preface XI Abbreviation XV List of Contributors XIX
 V Preface XI Abbreviation XV List of Contributors XIX 1 The Chemistry of Gold 1 M. Concepción Gimeno 1.1 Introduction 1 1.1.1 History 1 1.1.2 Chemical and Physical Properties 2 1.1.3 Theoretical Considerations
V Preface XI Abbreviation XV List of Contributors XIX 1 The Chemistry of Gold 1 M. Concepción Gimeno 1.1 Introduction 1 1.1.1 History 1 1.1.2 Chemical and Physical Properties 2 1.1.3 Theoretical Considerations
Carbenes and Carbene Complexes I Introduction
 Carbenes and Carbene Complexes I Introduction A very interesting (honest) class of radical-like molecules Steadily becoming more important as they find far more synthetic applications We will primarily
Carbenes and Carbene Complexes I Introduction A very interesting (honest) class of radical-like molecules Steadily becoming more important as they find far more synthetic applications We will primarily
ORGANIC CHEMISTRY. Fifth Edition. Stanley H. Pine
 ORGANIC CHEMISTRY Fifth Edition Stanley H. Pine Professor of Chemistry California State University, Los Angeles McGraw-Hill, Inc. New York St. Louis San Francisco Auckland Bogota Caracas Lisbon London
ORGANIC CHEMISTRY Fifth Edition Stanley H. Pine Professor of Chemistry California State University, Los Angeles McGraw-Hill, Inc. New York St. Louis San Francisco Auckland Bogota Caracas Lisbon London
O CH 3. Mn CH 3 OC C. 16eelimination
 igratory Insertion igratory Insertion/Elimination 1 A migratory insertion reaction is when a cisoidal anionic and neutral ligand on a metal complex couple together to generate a new coordinated anionic
igratory Insertion igratory Insertion/Elimination 1 A migratory insertion reaction is when a cisoidal anionic and neutral ligand on a metal complex couple together to generate a new coordinated anionic
A. Loupy, B.Tchoubar. Salt Effects in Organic and Organometallic Chemistry
 A. Loupy, B.Tchoubar Salt Effects in Organic and Organometallic Chemistry 1 Introduction - Classification of Specific Salt Effects 1 1.1 Specific Salt Effects Involving the Salt's Lewis Acid or Base Character
A. Loupy, B.Tchoubar Salt Effects in Organic and Organometallic Chemistry 1 Introduction - Classification of Specific Salt Effects 1 1.1 Specific Salt Effects Involving the Salt's Lewis Acid or Base Character
Insertion and elimination. Peter H.M. Budzelaar
 Peter H.. Budzelaar Insertion reactions If at a metal centre you have a) a σ-bound group (hydride, alkyl, aryl) b) a ligand containing a π-system (olefin, alkyne, C) the σ-bound group can migrate to the
Peter H.. Budzelaar Insertion reactions If at a metal centre you have a) a σ-bound group (hydride, alkyl, aryl) b) a ligand containing a π-system (olefin, alkyne, C) the σ-bound group can migrate to the
Nucleophilic attack on ligand
 Nucleophilic attack on ligand Nucleophile "substitutes" metal hapticity usually decreases xidation state mostly unchanged Competition: nucleophilic attack on metal usually leads to ligand substitution
Nucleophilic attack on ligand Nucleophile "substitutes" metal hapticity usually decreases xidation state mostly unchanged Competition: nucleophilic attack on metal usually leads to ligand substitution
R 2 R 4 Ln catalyst. This manuscript describes the methods for the synthesis and application of group 4 metallocene bis(trimethylsilyl)acetylene
 VII Abstracts 2011 p1 2.12.15 rganometallic Complexes of Scandium, Yttrium, and the Lanthanides P. Dissanayake, D. J. Averill, and M. J. Allen This manuscript is an update to the existing Science of Synthesis
VII Abstracts 2011 p1 2.12.15 rganometallic Complexes of Scandium, Yttrium, and the Lanthanides P. Dissanayake, D. J. Averill, and M. J. Allen This manuscript is an update to the existing Science of Synthesis
Carbenes and Olefin Metathesis
 arbenes and Olefin etathesis Peter H.. Budzelaar etal-carbon multiple bonds any transition metals form not only - single bonds but also = and (more rare) even bonds. omplexes containing an = bond are called
arbenes and Olefin etathesis Peter H.. Budzelaar etal-carbon multiple bonds any transition metals form not only - single bonds but also = and (more rare) even bonds. omplexes containing an = bond are called
Chem Selected Aspects of Main Group Chemistry
 Selected Aspects of Main Group Chemistry For the rest of the course, we will look at some aspects of the chemistry of main group compounds. The basic principles that you have learned concerning atoms,
Selected Aspects of Main Group Chemistry For the rest of the course, we will look at some aspects of the chemistry of main group compounds. The basic principles that you have learned concerning atoms,
Metal Hydrides, Alkyls, Aryls, and their Reactions
 Metal Hydrides, Alkyls, Aryls, and their Reactions A Primer on MO Theory σ-bonding in Organotransition Metal Complexes M-C Bond Energies in Organotransition Metal Complexes Thermodynamic Predictions
Metal Hydrides, Alkyls, Aryls, and their Reactions A Primer on MO Theory σ-bonding in Organotransition Metal Complexes M-C Bond Energies in Organotransition Metal Complexes Thermodynamic Predictions
https://cuvillier.de/de/shop/publications/766
 Jelena Jenter (Autor) Nitrogen Donor Ligands in the Coordination Chemistry of the are Earth and Alkaline Earth Metals Synthesis - Structures - Catalysis https://cuvillier.de/de/shop/publications/766 Copyright:
Jelena Jenter (Autor) Nitrogen Donor Ligands in the Coordination Chemistry of the are Earth and Alkaline Earth Metals Synthesis - Structures - Catalysis https://cuvillier.de/de/shop/publications/766 Copyright:
Repeated insertion. Multiple insertion leads to dimerization, oligomerization or polymerization. κ 1: mainly dimerization κ
 Repeated insertion ultiple insertion leads to dimerization, oligomerization or polymerization. k prop Et Key factor: k CT / k prop = κ κ 1: mainly dimerization κ 0.1-1.0: oligomerization (always mixtures)
Repeated insertion ultiple insertion leads to dimerization, oligomerization or polymerization. k prop Et Key factor: k CT / k prop = κ κ 1: mainly dimerization κ 0.1-1.0: oligomerization (always mixtures)
PAPER No. : Paper-9, Organic Chemistry-III (Reaction Mechanism-2) MODULE No. : Module-10, Hydroboration Reaction CHEMISTRY
 Subject Chemistry Paper No and Title Module No and Title Module Tag Paper-9, Organic Chemistry-III (Reaction Mechanism-2) Module-10, Hydroboration Reaction CHE_P9_M10 TABLE OF CONTENTS 1. Learning Outcomes
Subject Chemistry Paper No and Title Module No and Title Module Tag Paper-9, Organic Chemistry-III (Reaction Mechanism-2) Module-10, Hydroboration Reaction CHE_P9_M10 TABLE OF CONTENTS 1. Learning Outcomes
5.03, Inorganic Chemistry Prof. Daniel G. Nocera Lecture 4 Apr 11: Bent Metallocenes and Ziegler Natta Catalysis
 5.03, Inorganic hemistry Prof. Daniel G. Nocera Lecture 4 Apr 11: Bent Metallocenes and Ziegler Natta atalysis The electronic structure of organometallic complexes follows directly from the sandwich compounds
5.03, Inorganic hemistry Prof. Daniel G. Nocera Lecture 4 Apr 11: Bent Metallocenes and Ziegler Natta atalysis The electronic structure of organometallic complexes follows directly from the sandwich compounds
Organometallic Chemistry and Homogeneous Catalysis
 Organometallic Chemistry and Homogeneous Catalysis Dr. Alexey Zazybin Lecture N1 Kashiwa Campus, October 9, 2009 What compounds we can call organometallic compounds? Compounds containing direct metal-carbon
Organometallic Chemistry and Homogeneous Catalysis Dr. Alexey Zazybin Lecture N1 Kashiwa Campus, October 9, 2009 What compounds we can call organometallic compounds? Compounds containing direct metal-carbon
Keynotes in Organic Chemistry
 Keynotes in Organic Chemistry Second Edition ANDREW F. PARSONS Department of Chemistry, University of York, UK Wiley Contents Preface xi 1 Structure and bonding 1 1.1 Ionic versus covalent bonds 1 1.2
Keynotes in Organic Chemistry Second Edition ANDREW F. PARSONS Department of Chemistry, University of York, UK Wiley Contents Preface xi 1 Structure and bonding 1 1.1 Ionic versus covalent bonds 1 1.2
Zr-Catalyzed Carbometallation
 -Catalyzed Carbometallation C C C C ML n C C ML n ML n C C C C ML n ML n C C ML n Wipf Group esearch Topic Seminar Juan Arredondo November 13, 2004 Juan Arredondo @ Wipf Group 1 11/14/2004 Carbometallation
-Catalyzed Carbometallation C C C C ML n C C ML n ML n C C C C ML n ML n C C ML n Wipf Group esearch Topic Seminar Juan Arredondo November 13, 2004 Juan Arredondo @ Wipf Group 1 11/14/2004 Carbometallation
KOT 222 Organic Chemistry II
 KOT 222 Organic Chemistry II Course Objectives: 1) To introduce the chemistry of alcohols and ethers. 2) To study the chemistry of functional groups. 3) To learn the chemistry of aromatic compounds and
KOT 222 Organic Chemistry II Course Objectives: 1) To introduce the chemistry of alcohols and ethers. 2) To study the chemistry of functional groups. 3) To learn the chemistry of aromatic compounds and
Course 201N 1 st Semester Inorganic Chemistry Instructor: Jitendra K. Bera
 andout-9 ourse 201N 1 st Semester 2006-2007 Inorganic hemistry Instructor: Jitendra K. Bera ontents 3. rganometallic hemistry xidative Addition, Reductive Elimination, Migratory Insertion, Elimination
andout-9 ourse 201N 1 st Semester 2006-2007 Inorganic hemistry Instructor: Jitendra K. Bera ontents 3. rganometallic hemistry xidative Addition, Reductive Elimination, Migratory Insertion, Elimination
Inorganic Chemistry Year 3
 Inorganic Chemistry Year 3 Transition Metal Catalysis Eighteen Electron Rule 1.Get the number of the group that the metal is in (this will be the number of d electrons) 2.Add to this the charge 1.Negative
Inorganic Chemistry Year 3 Transition Metal Catalysis Eighteen Electron Rule 1.Get the number of the group that the metal is in (this will be the number of d electrons) 2.Add to this the charge 1.Negative
Julio Alvarez-Builla, Juan Jose Vaquero,
 Edited by Julio Alvarez-Builla, Juan Jose Vaquero, and Jose Barluenga Modern Heterocyclic Chemistry Volume 1 WILEY- VCH WILEY-VCH Verlag GmbH & Co. KG aa List of Contributors XV Volume 1 1 Heterocyclic
Edited by Julio Alvarez-Builla, Juan Jose Vaquero, and Jose Barluenga Modern Heterocyclic Chemistry Volume 1 WILEY- VCH WILEY-VCH Verlag GmbH & Co. KG aa List of Contributors XV Volume 1 1 Heterocyclic
HYDROCARBON CHEMISTRY
 HYDROCARBON CHEMISTRY George A. Olah Loker Hydrocarbon Research Institute and Department of Chemistry University of Southern California Los Angeles, California Ärpäd Molnär Department of Organic Chemistry
HYDROCARBON CHEMISTRY George A. Olah Loker Hydrocarbon Research Institute and Department of Chemistry University of Southern California Los Angeles, California Ärpäd Molnär Department of Organic Chemistry
ORGANIC - BROWN 8E CH ALKENES AND REACTIONS OF ALKENES
 !! www.clutchprep.com CONCEPT: ALKENES and ALKYNES Alkenes/Alkynes are named by adding the suffix modifier (- /- ) to the end of the root. Alkenes/alkynes receive in numbering alkanes Location is assigned
!! www.clutchprep.com CONCEPT: ALKENES and ALKYNES Alkenes/Alkynes are named by adding the suffix modifier (- /- ) to the end of the root. Alkenes/alkynes receive in numbering alkanes Location is assigned
Introduction. A1.1 (a) Shell number and number of subshells 1. A1.1 (b) Orbitals 2. A1.1 (c ) Orbital shapes (s, p & d) 2
 Preface Table of Contents Introduction i A1.1 (a) Shell number and number of subshells 1 A1.1 (b) Orbitals 2 A1.1 (c ) Orbital shapes (s, p & d) 2 A1.1 (d) Relative energies of s,p,d,f sub-shells 4 A 1.1
Preface Table of Contents Introduction i A1.1 (a) Shell number and number of subshells 1 A1.1 (b) Orbitals 2 A1.1 (c ) Orbital shapes (s, p & d) 2 A1.1 (d) Relative energies of s,p,d,f sub-shells 4 A 1.1
Page 1 of 9. Sessional Examination (November 2017) Max Marks: 20 Date: Time: One Hour. Model Answers
 Page 1 of 9 Sessional Examination (November 2017) Class: B. Pharm-II yr (III sem) Subject: Pharma Org. Chem-II Max Marks: 20 Date: 14.11.2017 Time: One Hour Model Answers Q. 1. Solve the following (ANY
Page 1 of 9 Sessional Examination (November 2017) Class: B. Pharm-II yr (III sem) Subject: Pharma Org. Chem-II Max Marks: 20 Date: 14.11.2017 Time: One Hour Model Answers Q. 1. Solve the following (ANY
Organic Chemistry 112 A B C - Syllabus Addendum for Prospective Teachers
 Chapter Organic Chemistry 112 A B C - Syllabus Addendum for Prospective Teachers Ch 1-Structure and bonding Ch 2-Polar covalent bonds: Acids and bases McMurry, J. (2004) Organic Chemistry 6 th Edition
Chapter Organic Chemistry 112 A B C - Syllabus Addendum for Prospective Teachers Ch 1-Structure and bonding Ch 2-Polar covalent bonds: Acids and bases McMurry, J. (2004) Organic Chemistry 6 th Edition
ORGANIC CHEMISTRY. Wiley STUDY GUIDE AND SOLUTIONS MANUAL TO ACCOMPANY ROBERT G. JOHNSON JON ANTILLA ELEVENTH EDITION. University of South Florida
 STUDY GUIDE AND SOLUTIONS MANUAL TO ACCOMPANY ORGANIC CHEMISTRY ELEVENTH EDITION T. W. GRAHAM SOLOMONS University of South Florida CRAIG B. FRYHLE Pacific Lutheran University SCOTT A. SNYDER Columbia University
STUDY GUIDE AND SOLUTIONS MANUAL TO ACCOMPANY ORGANIC CHEMISTRY ELEVENTH EDITION T. W. GRAHAM SOLOMONS University of South Florida CRAIG B. FRYHLE Pacific Lutheran University SCOTT A. SNYDER Columbia University
Catalysis & Sustainable Processes
 Catalysis & Sustainable Processes The Polymers Story 8 lectures http://www.kcpc.usyd.edu.au/cem3113.html username: chem3 password: carbon12 Lecturer: Associate Professor Sébastien Perrier s.perrier@chem.usyd.edu.au;
Catalysis & Sustainable Processes The Polymers Story 8 lectures http://www.kcpc.usyd.edu.au/cem3113.html username: chem3 password: carbon12 Lecturer: Associate Professor Sébastien Perrier s.perrier@chem.usyd.edu.au;
Organic Chemistry Laboratory Summer Lecture 6 Transition metal organometallic chemistry and catalysis July
 344 Organic Chemistry Laboratory Summer 2013 Lecture 6 Transition metal organometallic chemistry and catalysis July 30 2013 Summary of Grignard lecture Organometallic chemistry - the chemistry of compounds
344 Organic Chemistry Laboratory Summer 2013 Lecture 6 Transition metal organometallic chemistry and catalysis July 30 2013 Summary of Grignard lecture Organometallic chemistry - the chemistry of compounds
Module 6 : General properties of Transition Metal Organometallic Complexes. Lecture 2 : Synthesis and Stability. Objectives
 Module 6 : General properties of Transition Metal Organometallic Complexes Lecture 2 : Synthesis and Stability Objectives In this lecture you will learn the following Understand the role lead by ligands
Module 6 : General properties of Transition Metal Organometallic Complexes Lecture 2 : Synthesis and Stability Objectives In this lecture you will learn the following Understand the role lead by ligands
Transition Metal Chemistry
 Transition Metal Chemistry 2 2011.12.2 Ⅰ Fundamental Organometallic Reactions Following four reactions are important formal reaction patterns in organotransition metal complexes, which would conveniently
Transition Metal Chemistry 2 2011.12.2 Ⅰ Fundamental Organometallic Reactions Following four reactions are important formal reaction patterns in organotransition metal complexes, which would conveniently
H Organometallic Catalysis in Industry
 H Organometallic Catalysis in Industry Some terminology: Catalytic cycles: a circular path meant to show productive reactions, in order, that lead from the catalytically active species and its reaction
H Organometallic Catalysis in Industry Some terminology: Catalytic cycles: a circular path meant to show productive reactions, in order, that lead from the catalytically active species and its reaction
LIGAND DESIGN CARBENES. Fischer carbenes (B) have a heteroatom substituent on the alpha carbon atom.
 There are two main classes of carbene ligands Alkylidene (or Schrock carbene) ligands (A) have one or two alkyl or aryl substituents on the alpha carbon atom. Fischer carbenes (B) have a heteroatom substituent
There are two main classes of carbene ligands Alkylidene (or Schrock carbene) ligands (A) have one or two alkyl or aryl substituents on the alpha carbon atom. Fischer carbenes (B) have a heteroatom substituent
Inorganic Chemistry GARY L. MIESSLER DONALD A. TARR. St. Olaf College Northfield, Minnesota
 Inorganic Chemistry GARY L. MIESSLER DONALD A. TARR St. Olaf College Northfield, Minnesota Contents PREFACE xiii 1 INTRODUCTION TO INORGANIC CHEMISTRY 1 1-1 What Is Inorganic Chemistry? 1 1-2 Contrasts
Inorganic Chemistry GARY L. MIESSLER DONALD A. TARR St. Olaf College Northfield, Minnesota Contents PREFACE xiii 1 INTRODUCTION TO INORGANIC CHEMISTRY 1 1-1 What Is Inorganic Chemistry? 1 1-2 Contrasts
Catalytic Chemistry. Bruce C. Gates. John Wiley & Sons, Inc. New York Chichester Brisbane Toronto Singapore. University of Delaware ^.'-'.
 : s / ; '.... ;. : : ^.'-'. Catalytic Chemistry Bruce C. Gates University of Delaware John Wiley & Sons, Inc. New York Chichester Brisbane Toronto Singapore Contents List of Notation xix 1 INTRODUCTION
: s / ; '.... ;. : : ^.'-'. Catalytic Chemistry Bruce C. Gates University of Delaware John Wiley & Sons, Inc. New York Chichester Brisbane Toronto Singapore Contents List of Notation xix 1 INTRODUCTION
Lecture 01 Week - 01 History of Organometallic Compounds
 Transition Metal Organometallic Chemistry: Principles to Applications Prof. Prasenjit Ghosh Department of Chemistry Indian Institute of Technology, Bombay Lecture 01 Week - 01 History of Organometallic
Transition Metal Organometallic Chemistry: Principles to Applications Prof. Prasenjit Ghosh Department of Chemistry Indian Institute of Technology, Bombay Lecture 01 Week - 01 History of Organometallic
Alcohol Synthesis. Dr. Sapna Gupta
 Alcohol Synthesis Dr. Sapna Gupta Synthesis of Alcohols Alcohols can be synthesized from several functional groups. Nucleophilic substitution of O - on alkyl halide ydration of alkenes water in acid solution
Alcohol Synthesis Dr. Sapna Gupta Synthesis of Alcohols Alcohols can be synthesized from several functional groups. Nucleophilic substitution of O - on alkyl halide ydration of alkenes water in acid solution
Organometallic Chemistry and Homogeneous Catalysis
 Organometallic Chemistry and Homogeneous Catalysis Dr. Alexey Zazybin Lecture N6 Kashiwa Campus, November 27, 2009 Group VIB: Cr, Mo, W -Oxidation states from -2 to +6 -While +2 and +3 for Cr are quite
Organometallic Chemistry and Homogeneous Catalysis Dr. Alexey Zazybin Lecture N6 Kashiwa Campus, November 27, 2009 Group VIB: Cr, Mo, W -Oxidation states from -2 to +6 -While +2 and +3 for Cr are quite
π bonded ligands alkene complexes alkyne complexes allyl complexes diene complexes cyclopentadienyl complexes arene complexes metallacycles
 π bonded ligands alkene complexes alkyne complexes allyl complexes diene complexes cyclopentadienyl complexes arene complexes metallacycles M Transition metal alkene complexes The report in 1825 by William
π bonded ligands alkene complexes alkyne complexes allyl complexes diene complexes cyclopentadienyl complexes arene complexes metallacycles M Transition metal alkene complexes The report in 1825 by William
Organometallic Chemistry and Homogeneous Catalysis
 rganometallic hemistry and omogeneous atalysis Dr. Alexey Zazybin Lecture N8 Kashiwa ampus, December 11, 2009 Types of reactions in the coordination sphere of T 3. Reductive elimination X-L n -Y L n +
rganometallic hemistry and omogeneous atalysis Dr. Alexey Zazybin Lecture N8 Kashiwa ampus, December 11, 2009 Types of reactions in the coordination sphere of T 3. Reductive elimination X-L n -Y L n +
CHEMISTRY 263 HOME WORK
 Lecture Topics: CHEMISTRY 263 HOME WORK Module7: Hydrogenation of Alkenes Hydrogenation - syn and anti- addition - hydrogenation of alkynes - synthesis of cis-alkenes -synthesis of trans-alkenes Text sections:
Lecture Topics: CHEMISTRY 263 HOME WORK Module7: Hydrogenation of Alkenes Hydrogenation - syn and anti- addition - hydrogenation of alkynes - synthesis of cis-alkenes -synthesis of trans-alkenes Text sections:
Learning Guide for Chapter 13 - Alkynes
 Learning Guide for Chapter 13 - Alkynes I. Introduction to Alkynes - p 1 II. Natural ccurrence and Uses of Alkynes - p 5 III. Physical Properties of Alkynes - p 7 IV. Spectroscopy of Alkynes - p 7 V. Nomenclature
Learning Guide for Chapter 13 - Alkynes I. Introduction to Alkynes - p 1 II. Natural ccurrence and Uses of Alkynes - p 5 III. Physical Properties of Alkynes - p 7 IV. Spectroscopy of Alkynes - p 7 V. Nomenclature
JEFFERSON COLLEGE COURSE SYLLABUS CHM200 ORGANIC CHEMISTRY I. 5 Credit Hours. Prepared by: Richard A. Pierce
 JEFFERSON COLLEGE COURSE SYLLABUS CHM200 ORGANIC CHEMISTRY I 5 Credit Hours Prepared by: Richard A. Pierce Revised Date: January 2008 by Ryan H. Groeneman Arts & Science Education Dr. Mindy Selsor, Dean
JEFFERSON COLLEGE COURSE SYLLABUS CHM200 ORGANIC CHEMISTRY I 5 Credit Hours Prepared by: Richard A. Pierce Revised Date: January 2008 by Ryan H. Groeneman Arts & Science Education Dr. Mindy Selsor, Dean
Chapter 8 Alkenes and Alkynes II: Addition Reactions
 Chapter 8 Alkenes and Alkynes II: Addition Reactions Introduction: Additions to Alkenes Generally the reaction is exothermic because one π and one σ bond are converted to two σ bonds The π electrons of
Chapter 8 Alkenes and Alkynes II: Addition Reactions Introduction: Additions to Alkenes Generally the reaction is exothermic because one π and one σ bond are converted to two σ bonds The π electrons of
NOT TO BE REMOVED FROM THE EXAMINATION HALL
 A copy of the Level III (FHEQ Level 6) Equation and Data Sheet booklet is provided. The use of hand-held, battery-operated, electronic calculators will be permitted subject to the regulations governing
A copy of the Level III (FHEQ Level 6) Equation and Data Sheet booklet is provided. The use of hand-held, battery-operated, electronic calculators will be permitted subject to the regulations governing
CATALYSIS MULTICATALYST SYSTEM IN ASYMMETRIC. Wiley. Department of Chemistry
 MULTICATALYST SYSTEM IN ASYMMETRIC CATALYSIS JIAN ZHOU Shanghai Key Laboratory of Green Chemistry and Chemical Processes Department of Chemistry East China Normal University Shanghai, China Wiley Preface
MULTICATALYST SYSTEM IN ASYMMETRIC CATALYSIS JIAN ZHOU Shanghai Key Laboratory of Green Chemistry and Chemical Processes Department of Chemistry East China Normal University Shanghai, China Wiley Preface
PART 2. -BONDED ORGANOMETALLICS
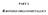 PART 2. -BODED ORGAOETALLIS V() 6 r() 6 o() 6 W() 6 Fe() 5 Ru() 5 Os() 5 i() 4 Figure 1. Binary, ononuclear etal arbonyls n n O Fe O O Fe O o O o n 2 () 10 Fe 2 () 9 o 2 () 8 Fe Fe Fe Ir() 3 Os Os Os o
PART 2. -BODED ORGAOETALLIS V() 6 r() 6 o() 6 W() 6 Fe() 5 Ru() 5 Os() 5 i() 4 Figure 1. Binary, ononuclear etal arbonyls n n O Fe O O Fe O o O o n 2 () 10 Fe 2 () 9 o 2 () 8 Fe Fe Fe Ir() 3 Os Os Os o
Loudon Chapter 18 Review: Vinyl/Aryl Reactivity Jacquie Richardson, CU Boulder Last updated 2/21/2016
 Chapter 18 covers leaving groups that are directly attached to double-bonded sp 2 carbons. These molecules don t do most of the regular alkyl halide chemistry from Ch. 9 (S N1/ S N2/E1), but they can do
Chapter 18 covers leaving groups that are directly attached to double-bonded sp 2 carbons. These molecules don t do most of the regular alkyl halide chemistry from Ch. 9 (S N1/ S N2/E1), but they can do
Spring Term 2012 Dr. Williams (309 Zurn, ex 2386)
 Chemistry 242 Organic Chemistry II Spring Term 2012 Dr. Williams (309 Zurn, ex 2386) Web Page: http://math.mercyhurst.edu/~jwilliams/ jwilliams@mercyhurst.edu (or just visit Department web site and look
Chemistry 242 Organic Chemistry II Spring Term 2012 Dr. Williams (309 Zurn, ex 2386) Web Page: http://math.mercyhurst.edu/~jwilliams/ jwilliams@mercyhurst.edu (or just visit Department web site and look
Chapter 8 Alkenes and Alkynes II: Addition Reactions. Alkenes are electron rich. Additions to Alkenes
 Additions to Alkenes Chapter 8 Alkenes and Alkynes II: Addition Reactions Generally the reaction is exothermic because one p and one s bond are converted to two s bonds Alkenes are electron rich The carbocation
Additions to Alkenes Chapter 8 Alkenes and Alkynes II: Addition Reactions Generally the reaction is exothermic because one p and one s bond are converted to two s bonds Alkenes are electron rich The carbocation
CHEM 261 HOME WORK Lecture Topics: MODULE 1: The Basics: Bonding and Molecular Structure Text Sections (N0 1.9, 9-11) Homework: Chapter 1:
 CHEM 261 HOME WORK Lecture Topics: MODULE 1: The Basics: Bonding and Molecular Structure Atomic Structure - Valence Electrons Chemical Bonds: The Octet Rule - Ionic bond - Covalent bond How to write Lewis
CHEM 261 HOME WORK Lecture Topics: MODULE 1: The Basics: Bonding and Molecular Structure Atomic Structure - Valence Electrons Chemical Bonds: The Octet Rule - Ionic bond - Covalent bond How to write Lewis
1. Determine the oxidation state of the metal centre and count the number of electrons.
 Exercise sheet : Organometallic chemistry Gunnar Bachem 1. Determine the oxidation state of the metal centre and count the number of electrons. 2. The metal fragment 1 reacts with the amine to give a carbene
Exercise sheet : Organometallic chemistry Gunnar Bachem 1. Determine the oxidation state of the metal centre and count the number of electrons. 2. The metal fragment 1 reacts with the amine to give a carbene
14.11 Alkane Synthesis Using Organocopper Reagents
 14.11 Alkane Synthesis Using Organocopper Reagents Lithium Dialkylcuprates Lithium dialkylcuprates are useful synthetic reagents. They are prepared from alkyllithiums and a copper(i) halide. 2RLi + CuX
14.11 Alkane Synthesis Using Organocopper Reagents Lithium Dialkylcuprates Lithium dialkylcuprates are useful synthetic reagents. They are prepared from alkyllithiums and a copper(i) halide. 2RLi + CuX
JEFFERSON COLLEGE COURSE SYLLABUS CHM201 ORGANIC CHEMISTRY II. 5 Credit Hours. Prepared by: Richard A. Pierce
 JEFFERSON COLLEGE COURSE SYLLABUS CHM201 ORGANIC CHEMISTRY II 5 Credit Hours Prepared by: Richard A. Pierce Revised Date: January 2008 by Ryan H. Groeneman Arts & Science Education Dr. Mindy Selsor, Dean
JEFFERSON COLLEGE COURSE SYLLABUS CHM201 ORGANIC CHEMISTRY II 5 Credit Hours Prepared by: Richard A. Pierce Revised Date: January 2008 by Ryan H. Groeneman Arts & Science Education Dr. Mindy Selsor, Dean
Organometallic Chemistry 0/ the Transition Elements
 Organometallic Chemistry 0/ the Transition Elements MODERN INORGANIC CHEMISTRY Series Editor: John P. Fackler, Jr. Texas A&M University CARBON-FUNCTlONAL ORGANOSILICON COMPOUNDS Edited by Vac1av Chvalovsky
Organometallic Chemistry 0/ the Transition Elements MODERN INORGANIC CHEMISTRY Series Editor: John P. Fackler, Jr. Texas A&M University CARBON-FUNCTlONAL ORGANOSILICON COMPOUNDS Edited by Vac1av Chvalovsky
Organometallic Catalysis
 Organometallic Catalysis The catalysts we will study are termed homogeneous catalysts as they are dissolved in th e same solvent as the substrate. In contrast, heterogeneous catalysts, such as palladium
Organometallic Catalysis The catalysts we will study are termed homogeneous catalysts as they are dissolved in th e same solvent as the substrate. In contrast, heterogeneous catalysts, such as palladium
Chapter 16. Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group. Physical Properties of Aldehydes and Ketones. Synthesis of Aldehydes
 Nomenclature of Aldehydes and Ketones Chapter 16 Aldehydes and Ketones I. Aldehydes replace the -e of the parent alkane with -al The functional group needs no number Nucleophilic Addition to the Carbonyl
Nomenclature of Aldehydes and Ketones Chapter 16 Aldehydes and Ketones I. Aldehydes replace the -e of the parent alkane with -al The functional group needs no number Nucleophilic Addition to the Carbonyl
N-Heterocyclic Carbenes (NHCs)
 N-Heterocyclic Carbenes (NHCs) In contrast to Fischer and Schrock type carbenes NHCs are extremely stable, inert ligands when complexed to a metal centre. Similar to phosphine ligands they are electronically
N-Heterocyclic Carbenes (NHCs) In contrast to Fischer and Schrock type carbenes NHCs are extremely stable, inert ligands when complexed to a metal centre. Similar to phosphine ligands they are electronically
Answers To Chapter 7 Problems.
 Answers To Chapter Problems.. Most of the Chapter problems appear as end-of-chapter problems in later chapters.. The first reaction is an ene reaction. When light shines on in the presence of light and
Answers To Chapter Problems.. Most of the Chapter problems appear as end-of-chapter problems in later chapters.. The first reaction is an ene reaction. When light shines on in the presence of light and
Module9. Nuclear Magnetic Resonance Spectroscopy Nuclear Magnetic Resonance (NMR) spectroscopy - Chemical shift - Integration of signal area
 1 CHEMISTRY 263 HOME WORK Lecture Topics: Module7. Hydrogenation of Alkenes The Function of the Catalyst - Syn and anti- addition Hydrogenation of Alkynes - Syn- addition of hydrogen: Synthesis of cis-alkenes
1 CHEMISTRY 263 HOME WORK Lecture Topics: Module7. Hydrogenation of Alkenes The Function of the Catalyst - Syn and anti- addition Hydrogenation of Alkynes - Syn- addition of hydrogen: Synthesis of cis-alkenes
REALLY, REALLY STRONG BASES. DO NOT FORGET THIS!!!!!
 CHEM 345 Problem Set 4 Key Grignard (RMgX) Problem Set You will be using Grignard reagents throughout this course to make carbon-carbon bonds. To use them effectively, it will require some knowledge from
CHEM 345 Problem Set 4 Key Grignard (RMgX) Problem Set You will be using Grignard reagents throughout this course to make carbon-carbon bonds. To use them effectively, it will require some knowledge from
D. X. Hu Towards Catalytic Enantioselective Halogenation of Alkenes Burns Group
 D. X. Hu Towards Catalytic Enantioselective Halogenation of Alkenes Burns Group Literature Review Organic Synthesis 10, 20, 50 Years from Now? Catalytic Enantioselective Halogenation October 6 th, 2012
D. X. Hu Towards Catalytic Enantioselective Halogenation of Alkenes Burns Group Literature Review Organic Synthesis 10, 20, 50 Years from Now? Catalytic Enantioselective Halogenation October 6 th, 2012
Catalysis by Group IV Elements CHEM 966 (Tunge) Good reference: Titanium and Zirconium in Organic Synthesis Ilan Marek Ed., 2002.
 Catalysis by Group IV Elements CEM 966 (Tunge) Good reference: Titanium and Zirconium in rganic Synthesis Ilan Marek Ed., 2002. Electronegativity: Ti(1.54); (1.33); f(1.3) Much of the chemistry is dominated
Catalysis by Group IV Elements CEM 966 (Tunge) Good reference: Titanium and Zirconium in rganic Synthesis Ilan Marek Ed., 2002. Electronegativity: Ti(1.54); (1.33); f(1.3) Much of the chemistry is dominated
Ketones and Aldehydes Reading Study Problems Key Concepts and Skills Lecture Topics: Structure of Ketones and Aldehydes Structure:
 Ketones and Aldehydes Reading: Wade chapter 18, sections 18-1- 18-21 Study Problems: 18-43, 18-44,18-50, 18-51, 18-52, 18-59, 18-60, 18-62, 18-64, 18-72. Key Concepts and Skills: Interpret the IR, NMR,
Ketones and Aldehydes Reading: Wade chapter 18, sections 18-1- 18-21 Study Problems: 18-43, 18-44,18-50, 18-51, 18-52, 18-59, 18-60, 18-62, 18-64, 18-72. Key Concepts and Skills: Interpret the IR, NMR,
Chem Carbenoids and Related Species
 Chem 59-651 Carbenoids and elated Species In contrast to carbenes, the heavier group 14 analogues and numerous group 15 analogues have been known for considerably longer. Probably the most important difference
Chem 59-651 Carbenoids and elated Species In contrast to carbenes, the heavier group 14 analogues and numerous group 15 analogues have been known for considerably longer. Probably the most important difference
5.03 In-Class Exam 3
 5.03 In-Class Exam 3 Christopher C. Cummins April 9, 2010 Instructions Clearly write your name at the top of this front page, but otherwise do not write on this front page as it will be used for scoring.
5.03 In-Class Exam 3 Christopher C. Cummins April 9, 2010 Instructions Clearly write your name at the top of this front page, but otherwise do not write on this front page as it will be used for scoring.
Chapter 16 Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group
 Chapter 16 Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
Chapter 16 Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
Organometallics Study Meeting
 rganometallics Study eeting 04/21/2011.itsunuma 1. rystal field theory(ft) and ligand field theory(lft) FT: interaction between positively charged metal cation and negative charge on the non-bonding electrons
rganometallics Study eeting 04/21/2011.itsunuma 1. rystal field theory(ft) and ligand field theory(lft) FT: interaction between positively charged metal cation and negative charge on the non-bonding electrons
Carboxylic Acids and Nitriles
 Carboxylic Acids and Nitriles Why this Chapter? Carboxylic acids present in many industrial processes and most biological processes They are the starting materials from which other acyl derivatives are
Carboxylic Acids and Nitriles Why this Chapter? Carboxylic acids present in many industrial processes and most biological processes They are the starting materials from which other acyl derivatives are
Advanced Organic FOURTH. Part A: Structure and Mechanisms
 Advanced Organic FOURTH Chemistry EDITION Part A: Structure and Mechanisms Advanced Organic Chemistry PART A: Structure and Mechanisms PART B: Reactions and Synthesis Advanced Organic Chemistry FOURTH
Advanced Organic FOURTH Chemistry EDITION Part A: Structure and Mechanisms Advanced Organic Chemistry PART A: Structure and Mechanisms PART B: Reactions and Synthesis Advanced Organic Chemistry FOURTH
Metallocene Catalysts for Ethylene Polymerization
 etallocene Catalysts for Ethylene Polymerization Dr. Syriac J. Palackal* and Dr. Atieh Abu Raqabah Sabic R&D Riyadh, Kingdom of Saudi Arabia ABSTRACT etallocene catalysts are the latest addition to the
etallocene Catalysts for Ethylene Polymerization Dr. Syriac J. Palackal* and Dr. Atieh Abu Raqabah Sabic R&D Riyadh, Kingdom of Saudi Arabia ABSTRACT etallocene catalysts are the latest addition to the
Topic 9. Aldehydes & Ketones
 Chemistry 2213a Fall 2012 Western University Topic 9. Aldehydes & Ketones A. Structure and Nomenclature The carbonyl group is present in aldehydes and ketones and is the most important group in bio-organic
Chemistry 2213a Fall 2012 Western University Topic 9. Aldehydes & Ketones A. Structure and Nomenclature The carbonyl group is present in aldehydes and ketones and is the most important group in bio-organic
Electrophilic Carbenes
 Electrophilic Carbenes The reaction of so-called stabilized diazo compounds with late transition metals produces a metal carbene intermediate that is electrophilic. The most common catalysts are Cu(I)
Electrophilic Carbenes The reaction of so-called stabilized diazo compounds with late transition metals produces a metal carbene intermediate that is electrophilic. The most common catalysts are Cu(I)
Contents. 1 Matter: Its Properties and Measurement 1. 2 Atoms and the Atomic Theory Chemical Compounds Chemical Reactions 111
 Ed: Pls provide art About the Authors Preface xvii xvi 1 Matter: Its Properties and Measurement 1 1-1 The Scientific Method 2 1-2 Properties of Matter 4 1-3 Classification of Matter 5 1-4 Measurement of
Ed: Pls provide art About the Authors Preface xvii xvi 1 Matter: Its Properties and Measurement 1 1-1 The Scientific Method 2 1-2 Properties of Matter 4 1-3 Classification of Matter 5 1-4 Measurement of
Course 201N 1 st Semester Inorganic Chemistry Instructor: Jitendra K. Bera
 andout-8 ourse 201N 1 st Semester 2006-2007 Inorganic hemistry Instructor: Jitendra K. Bera ontents 3. Organometallic hemistry yclopentadienyl, Alkyl and Alkene yclopentadienyl p The cyclopentadienyl ligand
andout-8 ourse 201N 1 st Semester 2006-2007 Inorganic hemistry Instructor: Jitendra K. Bera ontents 3. Organometallic hemistry yclopentadienyl, Alkyl and Alkene yclopentadienyl p The cyclopentadienyl ligand
Chapter 5. Reactions of Alkenes and Alkynes
 Learning objectives: Chapter 5. Reactions of Alkenes and Alkynes 1. Differentiate primary, secondary, and tertiary carbocations, and recognize the order of stability for these carbocations. 2. Identify
Learning objectives: Chapter 5. Reactions of Alkenes and Alkynes 1. Differentiate primary, secondary, and tertiary carbocations, and recognize the order of stability for these carbocations. 2. Identify
ORGANIC - CLUTCH CH ADDITION REACTIONS.
 !! www.clutchprep.com CONCEPT: GENERAL MECHANISM Addition reactions are ones in which 1 bond is broken and 2 new bonds are formed. They are the inverse of reactions EXAMPLE: Provide the mechanism for the
!! www.clutchprep.com CONCEPT: GENERAL MECHANISM Addition reactions are ones in which 1 bond is broken and 2 new bonds are formed. They are the inverse of reactions EXAMPLE: Provide the mechanism for the
N-Heterocyclic Carbenes (NHCs)
 N-Heterocyclic Carbenes (NHCs) In contrast to Fischer and Schrock type carbenes NHCs are extremely stable, inert ligands when complexed to a metal centre. Similar to phosphine ligands they are electronically
N-Heterocyclic Carbenes (NHCs) In contrast to Fischer and Schrock type carbenes NHCs are extremely stable, inert ligands when complexed to a metal centre. Similar to phosphine ligands they are electronically
AQUEOUS ORGANOMETALLIC CATALYSIS
 AQUEOUS ORGANOMETALLIC CATALYSIS Catalysis by Metal Complexes Volume 23 Editors: B. R. James, The University of British Columbia, Vancouver, Canada P. W. N. M. van Leeuwen, University of Amsterdam, The
AQUEOUS ORGANOMETALLIC CATALYSIS Catalysis by Metal Complexes Volume 23 Editors: B. R. James, The University of British Columbia, Vancouver, Canada P. W. N. M. van Leeuwen, University of Amsterdam, The
Oxidative Addition and Reductive Elimination
 xidative Addition and Reductive Elimination red elim coord 2 ox add ins Peter.. Budzelaar xidative Addition Basic reaction: n + X Y n X Y The new -X and -Y bonds are formed using: the electron pair of
xidative Addition and Reductive Elimination red elim coord 2 ox add ins Peter.. Budzelaar xidative Addition Basic reaction: n + X Y n X Y The new -X and -Y bonds are formed using: the electron pair of
Alkenes (Olefins) Chapters 7 & 8 Organic Chemistry, 8 th Edition John McMurry
 Alkenes (Olefins) Chapters 7 & 8 Organic Chemistry, 8 th Edition John McMurry 1 Structure and Bonding 2 Structure and Bonding Rotation around the C=C bond is restricted 90 rotation The p orbitals are orthogonal
Alkenes (Olefins) Chapters 7 & 8 Organic Chemistry, 8 th Edition John McMurry 1 Structure and Bonding 2 Structure and Bonding Rotation around the C=C bond is restricted 90 rotation The p orbitals are orthogonal
JEFFERSON COLLEGE COURSE SYLLABUS CHM200 ORGANIC CHEMISTRY I. 5 Credit Hours. Prepared by: Richard A. Pierce. Revised by: Sean Birke October, 2013
 JEFFERSON COLLEGE COURSE SYLLABUS CHM200 ORGANIC CHEMISTRY I 5 Credit Hours Prepared by: Richard A. Pierce Revised by: Sean Birke October, 2013 Ms. Linda Abernathy, Math, Science & Business Division Chair
JEFFERSON COLLEGE COURSE SYLLABUS CHM200 ORGANIC CHEMISTRY I 5 Credit Hours Prepared by: Richard A. Pierce Revised by: Sean Birke October, 2013 Ms. Linda Abernathy, Math, Science & Business Division Chair
Introduction to Organometallic Chemistry. Stability of organometallic reagents
 Introduction to rganometallic hemistry. rganometallic chemistry is the chemistry of compounds that contain M- bonds, where M is either a main group metal or a transition metal.. ne common feature of all
Introduction to rganometallic hemistry. rganometallic chemistry is the chemistry of compounds that contain M- bonds, where M is either a main group metal or a transition metal.. ne common feature of all
ST. JOSEPH S COLLEGE OF ARTS & SCIENCE (AUTONOMOUS) ST. JOSEPH S COLLEGE ROAD, CUDDALORE CH101T ORGANIC CHEMISTRY I (SEMESTER-I)
 UNIT I 1. The hybridization involved in the formation of acetylene is a) sp b) sp 2 c) sp 3 d) sp 3 d 2. The IUPAC name of is 1. 3-hexene b) 4-hexene c) 3-hexyne d) 4-hexyne 3. -------- is the type of
UNIT I 1. The hybridization involved in the formation of acetylene is a) sp b) sp 2 c) sp 3 d) sp 3 d 2. The IUPAC name of is 1. 3-hexene b) 4-hexene c) 3-hexyne d) 4-hexyne 3. -------- is the type of
N-HETEROCYCLIC CARBENES: STRUCTURE AND PROPERTIES
 N-HETEROCYCLIC CARBENES: STRUCTURE AND PROPERTIES Zachery Matesich 24 February 2015 Roadmap 2 Introduction Synthetic Methods History of NHCs Properties of NHCs Nature of the carbene Structural properties
N-HETEROCYCLIC CARBENES: STRUCTURE AND PROPERTIES Zachery Matesich 24 February 2015 Roadmap 2 Introduction Synthetic Methods History of NHCs Properties of NHCs Nature of the carbene Structural properties
DEPARTMENT: Chemistry
 CODE: CHEM 203 TITLE: Organic Chemistry I INSTITUTE: STEM DEPARTMENT: Chemistry COURSE DESCRIPTION: Students will apply many concepts from general chemistry to a study of organic chemistry. They will be
CODE: CHEM 203 TITLE: Organic Chemistry I INSTITUTE: STEM DEPARTMENT: Chemistry COURSE DESCRIPTION: Students will apply many concepts from general chemistry to a study of organic chemistry. They will be
1. Radical Substitution on Alkanes. 2. Radical Substitution with Alkenes. 3. Electrophilic Addition
 1. Radical Substitution on Alkanes Only Cl and Br are useful at the laboratory level. Alkane reactivity: tertiary > secondary > primary > methyl Numbers below products give their relative yield. Relative
1. Radical Substitution on Alkanes Only Cl and Br are useful at the laboratory level. Alkane reactivity: tertiary > secondary > primary > methyl Numbers below products give their relative yield. Relative
Chapter 16 Aldehydes and Ketones I Nucleophilic Addition to the Carbonyl Group
 Chapter 16 Aldehydes and Ketones I Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
Chapter 16 Aldehydes and Ketones I Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
Alkynes Nomenclature of Alkynes
 Chapter 7 Alkynes Alkynes - hydrocarbons containing a carbon-carbon triple bond (2 bonds) Acyclic alkanes = C n H 2n+2 Alkenes and cyclic alkanes = C n H 2n Alkynes (and cyclic alkenes) = C n H 2n-2 The
Chapter 7 Alkynes Alkynes - hydrocarbons containing a carbon-carbon triple bond (2 bonds) Acyclic alkanes = C n H 2n+2 Alkenes and cyclic alkanes = C n H 2n Alkynes (and cyclic alkenes) = C n H 2n-2 The
p Bonds as Electrophiles
 Chapter 7 p Bonds as Electrophiles REACTIONS OF CARBONYLS AND RELATED FUNCTIONAL GROUPS Copyright 2018 by Nelson Education Limited 1 7.2.1 Orbital structure of the carbonyl group Because oxygen is more
Chapter 7 p Bonds as Electrophiles REACTIONS OF CARBONYLS AND RELATED FUNCTIONAL GROUPS Copyright 2018 by Nelson Education Limited 1 7.2.1 Orbital structure of the carbonyl group Because oxygen is more
