Structure Prediction Methods. Fundamental Questions. Protein Structure Prediction. Protein Folding. The Protein Folding Problem MACGT...
|
|
|
- Daisy Owens
- 5 years ago
- Views:
Transcription
1 Tertary Structure of Proten Anfnsen s experments, late 1950 s through 1960 s Rbonuclease, an enzyme nvolved n cleavage of nuclec acds. Structure has a combnaton of α and β segments and four dsulfde brdges What are Dsulfde Brdges? Oxdaton Cys- Cys- Cys-S S-Cys Reducton Actve, natve structure BME s reducng agent Urea unfolds protens add β-mercaptoethanol (BME) HS-CH -CH -OH add urea, H N-C-NH Cys58 Cys110 Cys58 Cys110 O Denatured, nactve, random col, many conformatons Many Conformatons Many Conformatons Remove BME Remove urea Remove BME - Natve structure -fully actve - 4 dsulfde bond correct One Conformaton S S S S S S S Mxture of 105 dfferent conformatons, 1% actve S Remove urea One Conformaton 1
2 The Proten Foldng Problem Levnthal s paradox Consder a 100 resdue proten. If each resdue can take only 3 postons, there are = possble conformatons. If t takes s to convert from 1 structure to another, exhaustve search would take years! MACGT...? Gven a partcular sequence of amno acd resdues (prmary structure), what wll the tertary/quaternary structure of the resultng proten be? Proten Structure Predcton and Proten Foldng Proten Structure Predcton Fundamental Questons What s the structure of ths proten? Can be expermentally determned, today we know the structure of ~35,000 protens Can be predcted for some protens, usually n ~1 day on today's computers Proten Foldng: Fast Folders Tme Scale: 80 s 90 s 00 s 00 s 90 s 80 s ps ns µs µs ms sec Foldng MD Smulatons Foldng Experments Proten Foldng How does ths proten form ths structure? The process or mechansm of foldng Lmted expermental characterzaton Why does ths proten form ths structure? Why not some other fold? Why so quckly? -> Levnthal's Paradox: As there are an astronomcal number of conformatons possble, an unbased search would take too long for a proten to fold. Yet most protens fold n less than a second! Trp-cage, desgned mn-proten (0 aa): 4µs β-harpn of C-termnus of proten G (16 aa) : 6µs Engraled homeodoman (En-HD) (61 aa): ~7µs WW domans (38-44 aa): >4µs Fe(II) cytochrome b 56 (106 aa): extrapolated ~5µs B doman of proten A (58 aa): extrapolated ~8µs Structure Predcton Methods Homology Modelng 1 QQYTA KIKGR 11 TFRNE KELRD 1 FIEKF KGR Algorthm Secondary structure (only sequence) Homology modelng (usng related structure) Fold recognton Ab-nto 3D predcton Assumes smlar (homologous) sequences have very smlar tertary structures Basc structural framework s often the same (same secondary structure elements packed n the same way) Loop regons dffer Wde dfferences possble, even among closely related protens
3 Threadng Strateges for Proten Structure Predcton Gven: sequence of proten P wth unknown structure Database of known folds Fnd: Most plausble fold for P Evaluate qualty of such arrangement Places the resdues of unknown P along the backbone of a known structure and determne stablty of sde chans n that arrangement Comparatve Modelng Fold Recognton Ab Into 1. Identfy sequence homologs 1. Representaton as templates 1. Fold classfcaton. Force feld. Use sequence algnment to Method 3D-Profles 3. Global Optmzaton generate model 3. Improves wth data 4. Structure at global mnmum 3. Fll n unalgned regons 5. Can dscover new folds 4. Improves wth data 1. Requres > 5% sequence 1. Needs good number of protens n 1. Computatonally ntensve dentty each fold Drawbacks. Physcal modelng. Loops and sdechan. Crtcally dependent on scorng conformatons are crtcal functon Resoluton < 3 A 3-7 A > 5 A Tme to Compute < Day ~ Day >> Day Complementarty of the Methods X-ray crystallography- hghest resoluton structures; faster than NMR NMR- enables wdely varyng soluton condtons; characterzaton of motons and dynamc, weakly nteractng systems Computaton- fundamental understandng of structure, dynamcs and nteractons; models wthout experment; very fast The proten sequence contans all nformaton needed to create a correctly folded proten. Many protens fold spontaneously to ther natve structure Proten foldng s relatvely fast Chaperones speed up foldng, but do not alter the structure Forces drvng proten foldng It s beleved that hydrophobc collapse s a key drvng force for proten foldng Hydrophobc core Polar surface nteractng wth solvent Mnmum volume (no cavtes) Dsulfde bond formaton stablzes Hydrogen bonds Polar and electrostatc nteractons Four models that could account for the rapd rate of proten foldng durng bologcal proten synthess. - The Framework Model - The Nucleaton Model - The Molten Globule Model - Foldng Funnels Natve state s typcally only 5 to 10 kcal/mole more stable than the unfolded form 3
4 Framework Model Nucleaton Model Elements of Secondary Structure Formed Only MOST Stable Sec. Structure Formed Nucleaton Molten Globule Foldng Funnel Concept Unfolded, many conformatons More Compact Some secondary structural elements formed wth hydrophobc resdues nsde Many Possble Foldng Pathways to Get to Natve State Natve State, one conformaton Thermodynamcs of Proten Foldng Bond stretchng: sec. Elastc vbratons: sec. Rotatons of surface sdechans: sec. Hnge bendng: sec. Rotaton of bured sde chans: sec. Proten foldng: sec. Smulated foldng n 1 µsec; peptde n a box of water Free Energy Funnel Entropy and Enthalpy n Proten Foldng Unfolded Proten H, small, negatve S, large, postve G = H - T S bondng flexblty Folded Proten H, large, negatve S, small, postve Compensaton n entropy and enthalpy for proten Contrbuton of entropy of water molecules released upon foldng S of water s large and postve 4
5 Thermodynamcs of Proten Foldng G foldng =G folded -G unfolded = Natve state (N) C N Denatured state C C N N (H folded -H unfolded )-T(S folded -S unfolded )= H foldng -T S foldng G foldng H foldng -T S foldng Energy - unfolded folded Folded protens are hghly ordered S foldng negatve, so T S foldng s a postve quantty H foldng s a negatve quantty - enthalpy s favored n folded state. Total Gbbs free energy dfference s negatve folded state favoured N Sze of cavty n solvent ~6500Å S chan: sgnfcantly decreased, due to the well defned conformaton Non-bonded nteractons: ntra-molecular Compact structure G D N = H D N T S D N N C C S chan: large, due to the large number of dfferent conformatons Non-bonded nteractons: nter-molecular Non compact structure N Average sze of cavty n solvent :0,500Å C 1) ELECTROLYTE ADDITION - nterference wth the collod state ) INSOLUBLE SALT FORMATION - ProtenTrchloracetate 3) ORGANIC SOLVENTS - ETHANOL - nterferes wth the delectrc constant 4) HEAT DENATURATION - more energy n system (bonds break) 5) ph Factors that dsrupt the Natve state - destroys charge - destroys ablty to nteract wth water Thermodynamc Descrpton of Proten Foldng The natve and unfolded states are n equlbrum, the foldng reacton can be quantfed n terms of thermodynamcs. The equlbrum (N U) between the natve (N) and unfolded (U) states s defned by the equlbrum constant, K, as: K = [U]/[N] = K U The dfference n Gbbs free energy ( G) between the unfolded and natve states s then: G = -RT ln K For K u, a postve G ndcates that the natve state s more stable. 6) DESTRUCTION OF HYDROGEN BONDING - UREA - known H-bond dsrupter The free energy s composed of both enthalpc and entropc contrbutons: G = H - T S where H and S are the enthalpy and entropy change, respectvely, upon unfoldng. Thermal Unfoldng Snce H and S are strongly temperature-dependent, G s better expressed as: G = H 1 C P (T-T 1 ) - T [ S 1 C P ln(t/t 1 )] where the subscrpt 1 ndcates the value of H and S at a reference temperature, T 1, and C p s the specfc heat or heat-capacty change. Most protens denature reversbly allowng thermodynamc analyss. 1) ELECTROLYTE ADDITION - nterference wth the collod state ) INSOLUBLE SALT FORMATION - ProtenTrchloracetate 3) ORGANIC SOLVENTS - ETHANOL - nterferes wth the delectrc constant 4) HEAT DENATURATION - more energy n system (bonds break) 5) ph Factors that dsrupt the Natve state - destroys charge - destroys ablty to nteract wth water 6) DESTRUCTION OF HYDROGEN BONDING - UREA - known H-bond dsrupter 5
6 Solvng Proten Structures Only knds of technques allow one to get atomc resoluton pctures of macromolecules X-ray Crystallography (frst appled n Kendrew & Perutz) NMR Spectroscopy (frst appled n Ernst & Wuthrch) Structure Functon Structure Mechansm Structure Orgns/Evoluton Structure-based Drug Desgn Solvng the Proten Foldng Problem Ab Into Predcton Predctng the 3D structure wthout any pror knowledge Used when homology modellng or threadng have faled (no homologues are evdent) Equvalent to solvng the Proten Foldng Problem Stll a research problem Ab Into Foldng Two Central Problems Samplng conformatonal space ( ) The energy mnmum problem The Samplng Problem (Solutons) Lattce models, off-lattce models, smplfed chan methods The Energy Problem (Solutons) Threadng energes, packng assessment, topology assessment Problems n Proten Foldng Two key questons: Evaluaton how can we tell a correctly-folded proten from an ncorrectly folded proten? H-bonds, electrostatcs, hydrophobc effect, etc. Derve a functon, see how well t does on real protens Optmzaton once we get an evaluaton functon, can we optmze t? Smulated annealng/monte Carlo Interacton Covalent bonds Ionc Hydrogen bond Hydrophobc van der Waals Approx. bond strength n kj/mole > 00 (rangng up to 900) 0-40 ~5-0 ~ 8 ~ 4 Evaluaton of Proten Folds Emprcal potental functons Resdue-based: spatal relatonshps among resdues Stereochemstry-based: molecular nteractons (covalent, electrostatc, etc.) wth coeffcents Ab-nto potental functons Procheck, etc. Full molecular dynamcs Very computatonally expensve Polypeptdes Represented by a range of approaches or approxmatons ncludng: all atom representatons n cartesan space all atom representatons n dhedral space smplfed atomc versons n dhedral space tube/cylnder/rbbon representatons lattce models AMBER (Asssted Model Buldng wth Energy Refnement ) force feld 3 Vn Etotal = Kr ( r req ) Kθ ( θ θ eq ) [ cos( n )] 1 ω bonds angles dhedrals 1 atoms a b atoms qq j j r r 1 6 < < j ε r 6
7 The hydrophobc zpper effect: Lattce Models Scorng Lattce Models H/P model scorng: count noncovalent hydrophobc nteractons. Ken Dll ~ 1997 Sometmes: Penalze for bured polar or surface hydrophobc resdues Fold Optmzaton Smple lattce models (HP-models) Two types of resdues: hydrophobc and polar -D or 3-D lattce The only force s hydrophobc collapse Score = number of H H contacts 3.5Å A Smple D Lattce Lattce Foldng Buld a n x m matrx (a D array) Choose an arbtrary pont as your N termnal resdue (start resdue) Add or subtract 1 from the x or y poston of the start resdue Check to see f the new pont (resdue) s off the lattce or s already occuped Evaluate the energy Go to step 3 and repeat untl done Lattce Algorthm Red = hydrophobc Blue = hydrophlc If Red s near empty space E = E1 If Blue s near empty space E = E-1 If Red s near another Red E = E-1 If Blue s near another Blue E = E0 If Blue s near Red E = E0 7
8 More Complex Lattces What can we do wth lattce models? For smaller polypeptdes, exhaustve search can be used Lookng at the best fold, even n such a smple model, can teach us nterestng thngs about the proten foldng process More realstc models 1.45 A Hgher resoluton lattces (45 lattce, etc.) Off-lattce models Local moves Optmzaton/search methods and φ/ψ representatons Greedy search Graph theoretcal methods Monte Carlo, smulated annealng, etc. Non-Lattce Models Non-Lattce Models Res H C R H 3.5 Å Wth a more realstc off-lattce model, we need a better energy functon to evaluate a conformaton (fold). Theoretcal force feld: G = G van der Waals G h-bonds G solvent G coulomb 1.53 Å C 1.3 Å N 1.00 Å Emprcal force felds O 1.4 Å 1.47 Å C Res 1 R H Energy Terms Covalent r q φ Bondng Terms: bond stretch Harmonc Potental Stretchng K r (r -r j ) Noncovalent r van der Waals A /r 6 -B /r 1 Bendng K θ (θ - θ j ) r Coulomb q q j /4πεr Torsonal K φ (1 cos(nφ j )) r H-bond C /r 10 -D /r 1 Most often Harmonc 1 V ( bond kr r r = 0) bonds Morse Potental for dssocaton studes V Morse = bonds D e Two new parameters: D: dssocaton energy a: wdth of the potental well a( r r0 ) [ 1] Vbond D Vmorse r 0 bond length Morse Potental r 0 D bond length 8
9 Bondng Terms: angle bendng Bondng Terms: Torsons Harmonc Potental Most often Harmonc V angle 1 = k θ ( θ θ angles 0 ) CHARMM force feld s Urey-Bradley angle term: V UB Vangle θ 0 angle 1 = kub ( s s0 ) Ths UB term s only found n CHARMM UB force feld to optmze the ft to vbratonal spectra. s: the 1,3-dstance. Torson energy: rotaton about a bond (dhedral angles) U torson Vn = [1 cos( nφ δ )] j torsons -j-k-l φ k l Vn: force constant n: perodcty of the angle ( determnes how many peaks and wells n the potental, often from 1-6 ) δ: phase of the angle (often 0º or 180º) Mackerell et al. J. Phys. Chem. B 10, 3586, 1998 Bondng Terms: Improper Torsons Non-bonded Terms Coulomb Potental Improper torson s not a regular torson angle. It s used to descrbe the energy of out-of-plane motons. It s often necessary for planar groups, such as sp hybrdzed carbons n carbonyl groups and n aromatc rngs, because the normal torson terms descrbed above s not suffcent to mantan the planarty (ω~0). or U U mproper mproper V o = [1 cos(ω 180 )] mproper = mproper kw ( ω ω ) 0 j k -j-k-l ω l Electrostatc nteractons (Coulomb s Law) 1 qq j Velec = 4πε < j r Lennard-Jones nteractons 1 6 σ < σ VLJ = 4ε 1 6 j r r Combnaton Rules for LJ 1 ε = εε σ ( ) j = σ σ j σ = Velec VLJ σ σ j ~1/r par dstance LJ Potental par dstance r/sgma 1-4 Non-bonded Interactons Non-bonded exclusons 1- and 1-3 nteractons excluded 1-4 nteractons partally excluded 1-4 nteracton scalngs OPLSAA scales by 0.5 for both electrostatc and LJ AMBER94 scales 0.5 for LJ and 1/1. for electrostatc nteracton CHARMM has specal 1,4-terms j Even though they are non-bonded nteractons, 1-4 terms are often calculated along wth bonded terms. k l The hydrophobc effect The free energy gan from buryng a hydrophobc group s proportonal to the surface area bured 9
10 Lnear relaton between the solvent accessble surface area and the transfer free energy of amno acds Accessble Surface Area G transfer = -γ ASA γ = 0.05 cal/å Accessble Surface Area QHTAWCLTSEQHTAAVIWDCETPGKQNGAYQEDCA HHHHHHCCEEEEEEEEEEECCHHHHHHHCCCCCCC Solvent Probe Reentrant Surface Accessble Surface Van der Waals Surface QHTAWCLTSEQHTAAVIWDCETPGKQNGAYQEDCAMD BBPPBEEEEEPBPBPBPBBPEEEPBPEPEEEEEEEEE Accessble Surface Area Calculatons Force Felds: Typcal Energy Functons DSSP - Database of Secondary Structures for Protens (swft.embl-hedelberg.de/dssp) Connolly Molecular Surface Home Page Naccess Home Page ASA Parallelzaton Proten Structure Database U = bonds angles torsons mproper elec LJ 1 kr ( r r0 ) 1 kθ ( θ θ0) Vn [1 cos( nφ δ )] V ( mproper torson) q q r A [ r j 1 B ] 6 r Bond stretches Angle bendng Torsonal rotaton Improper torson (sp) Electrostatc nteracton Lennard-Jones nteracton 10
11 Whch Force Feld to Use? Most popular force felds: CHARMM, AMBER and OPLSAA OPLSAA(000): Probably the best avalable force feld for condensed-phase smulaton of peptdes. Work to develop parameterzaton that wll nclude broader classes of drug-lke molecules s ongong. GB/SA solvaton energes are good. MMFF: An excellent force feld for bopolymers and many drug-lke organc molecules that do not have parameters n other force felds. AMBER*/OPLS*: Good force felds for bopolymers and carbohydrates; many parameters were added n MacroModel whch extend the scope of ths force feld to a number of mportant organc functonal groups. GB/SA solvaton energes range from moderate (AMBER*) to good (OPLS*). AMBER94: An excellent force feld for protens and nuclec acds. However, there are no extensons for non-standard resdues or organc molecules, also there s a alpha-helx tendency for protens. AMBER99 fxes ths helx problem to some degree, but not completely. MM*/MM3*: Excellent force felds for hydrocarbons and molecules wth sngle or remotely spaced functonal groups. GB/SA solvaton energes tend to be poor relatve to those calculated wth other force felds. CHARMM: Good general purpose force feld for protens and nuclec acds. A bt weak for drug-lke organc molecules. GROMOS96: Good general purpose force feld for protens, partcularly good for free energy perturbatons due to soft-core potentals. Weak for reproducng solvaton free energes of organc molecules and small peptdes. Force Feld Parameterzaton Equlbrum bond dstances and angles: X-ray crystallography Bond and angle force constants: vbratonal spectra, normal mode calculatons wth QM Dhedral angle parameters: dffcult to measure drectly expermentally; ft to QM calculatons for rotatons around a bond wth other motons fxed Atom charges: ft to expermental lqud propertes, ESP charge fttng to reproduce electrostatc potentals of hgh level QM, X-ray crystallographc electron densty Lennard-Jones parameters: often most dffcult to determne, ft to expermental lqud propertes, ntermolecular energy fttng Applcatons NMR or X-ray structure refnement Proten structure predcton Proten foldng knetcs and mechancs Conformatonal dynamcs Global optmzaton DNA/RNA smulatons Membrane protens/lpd layers smulatons Delectrc constant NH 3 ε water = 80 Partal Charges -O-C 0.4 C = O N - H 0. O ε vacuum = 1 ε proten nteror = -10 -O-C NH 3 O ε water, salt > O H H If you know the poston of every partal charge (ncludng water), you do not need a delectrc constant Delectrc constant NH 3 ε water = 80 -O-C O ε vacuum = 1 ε proten nteror = -10 -O-C NH 3 O ε water, salt > 80 a Dpole - Monopole Interactons q θ q- dpole r r 1 r q o Dpole - Dpole Interactons monopole U = Σ U (n) = U 1 U U = 1/4πε o (q/r 1 -q/r ) = q/4πε o (r 1 -r / r 1 r ) f r >> a then r -r 1 ~ a cos θ and r 1 r = r U = qa/4πεo (cos θ/ r ) U s a functon of θ and r. If you rotate around the dpole axs, there s no change n the value of U ε 10 ε 80 The electrostatc potental at any pont relatve to fxed known charges even n the presence of moble charges usng the Posson - Boltzmann Equaton E = -µ a µ b / εr 3 E = -µ a µ b / εr 3 µ = dpole moment = Zd water = 1.85 D peptde bond = 3.5 D retnal = 15 D Interacton energy s dependent on orentaton and dstance 11
12 Na Na Cl- Cl- Emprcal Force Felds and Molecular Mechancs -10 kcal/mol descrbe nteracton of atoms or groups substrate NH 3 -O-C O NH 3 -O-C O enzyme the parameters are emprcal,.e. they are dependent on others and have no drect ntrnsc meanng Examples: GROMOS96 (van Gusteren) CHARMM (M. Karplus) AMBER (Kollman) unfavorable - ordered water favorable - van der Waals - free water hydrophobc effect = ~50 cal/mol/å Example for a (very) smple Force Feld: ν = bonds angles torsons k k ( l l ) ( θ θ ) VN,0,0 ( 1 cos( nω γ )) 1 6 N N σ σ qq j = 4πε 1 j 1 r r 4πε 0r = Complete Energy Functon: p 1 H = kr ( r req) m atoms bond rotaton [ V (1 e 0 H bond AMBER (Asssted Model Buldng wth Energy Refnement ) force feld 3 Vn Etotal = Kr ( r req ) Kθ ( θ θ eq ) [ cos( n )] 1 ω bonds bond stretch vn [1 cos( nφ γ )] ' a( r r0 ) ) V ] 0 angles 0 S bond dhedrals 1 atoms a b atoms qq j j r r 1 6 < < j ε r bond angle bendng [ V (1 e non bonded A B [ 1 6 r r ' a( r r0 ) ) V ] qq j ] ε r 1 kθ ( θ θeq) 0 Sources of force parameters: Bonds, VdW, Electrostatc (for amno acds, nucleotdes only): AMBER: J. Am. Chem. Soc. 117, CHARMM: J. Comp. Chem. 4, H-bonds (Morse potental): Nuclec Acds Res. 0, Bophys. J. 66, Electrostatc parameters of organc molecules need to be computed ndvdually by usng specal software (such as Gaussan) 1
13 Concept of energy scale s Important for molecular modelng Energy Mnmzaton E = f(x) E s a functon of coordnates ether cartesan or nternal At mnmum the frst dervatves are zero and the second dervatves are all postve de = 0 dx d E 0 dx a mult dmensonal energy landscape Energy coord x Potental Energy Surface (PES) coord y Systematc Searchng explore the whole PES Stochastc Searchng fnd all low energy mnma by generatng startng conformaton wth random changes of rotatable dhedral angles (sometmes combned wth random perturbaton of the Cartesan coordnates) followed by mnmzaton Monte Carlo Smulatons generate a Boltzmann dstrbuted ensemble of conformatons, can estmate macroscopc thermodynamc propertes Molecular Dynamcs Smulates the tme dependent moton of the molecular system, can estmate macroscopc thermodynamc propertes Smulated Annealng Playng wth the temperature (T) n ether MD or MC smulatons to speed up search for low energy mnma Dstance geometry method for generatng conformatons that satsfy expermental constrants Systematc Searchng Molecular Mechancs - Energy Mnmzaton The energy of the system s mnmzed. The system tres to relax Typcally, the system relaxes to a local mnmum (LM). 13
14 Conformatonal Samplng Md-energy lower energy lowest energy hghest energy Treat Proten molecule as a set of balls (wth mass) connected by rgd rods and sprngs Rods and sprngs have emprcally determned force constants Hgh Energy Steepest Descent & Conjugate Gradents Low Energy Overhead Vew Mnmzaton Methods Sde Vew Energy surfaces for protens are complex hyperdmensonal spaces Bggest problem s overcomng local mnmum problem Smple methods (slow) to complex methods (fast) Monte Carlo Method Steepest Descent Conjugate Gradent Frequently used for energy mnmzaton of large (and small) molecules Ideal for calculatng mnma for complex (.e. non-lnear) surfaces or functons Both use dervatves to calculate the slope and drecton of the optmzaton path Both requre that the scorng or energy functon be dfferentable (smooth) Steepest Descent Conjugate Gradent Mnmzaton Hgh Energy Hgh Energy Low Energy Low Energy Makes small locally steep moves down gradent Includes nformaton about the pror hstory of path The steepest descent method uses the frst dervatve to determne the drecton towards the mnmum. 14
15 Monte Carlo Algorthm Monte Carlo Mnmzaton Generate a conformaton or algnment (a state) Calculate that state s energy or score If that state s energy s less than the prevous state accept that state and go back to step 1 If that state s energy s greater than the prevous state accept t f a randomly chosen number s < e -E/kT where E s the state energy otherwse reject t Go back to step 1 and repeat untl done Low Energy Hgh Energy Performs a progressve or drected random search Molecular Dynamcs (MD) In molecular dynamcs, energy s suppled to the system, typcally usng a constant temperature (.e. constant average knetc energy). Molecular Dynamcs (MD) Use Newtonan mechancs to calculate the net force and acceleraton experenced by each atom. Each atom s treated as a pont wth mass m and fxed charge q Determne the force F on each atom: r r d r r F = m = ν ( R) dt Use postons and acceleratons at tme t (and postons from t - δ t) to calculate new postons at tme t δ t Intal veloctes (v ) usng the Boltzmann dstrbuton at the gven temperature v = (m /πkt) 1/ exp (- m v /kt) Molecular dynamcs (MD) smulatons A determnstc method based on the soluton of Newton s equaton of moton F = m a for the th partcle; the acceleraton at each step s calculated from the negatve gradent of the overall potental, usng F = - grad V -= - V In molecular dynamcs forces are derved from a potental energy functon V, whch depend on the partcle coordnates: The problem of modellng a materal can therefore be restated as that of fndng a potental functon for that materal. Molecular dynamcs (MD) smulatons V = Σ k (energes of nteractons between and all other resdues k located wthn a cutoff dstance of R c from ) Dervatve of V wth respect to the poston vector r = (x, y, z ) T at each step a x ~ - V/ x a y ~ - V/ y a z ~- V/ z Non-Bonded Interacton Potentals Electrostatc nteractons of the form E k (es) = q q k /r k Van der Waals nteractons E (vdw) = - a k /r k 6 b k /r k 1 Bonded Interacton Potentals Bond stretchng E (bs) = (k bs /) (l l 0 ) Bond angle dstorton E (bad) = (k θ /) (θ θ 0 ) Bond torsonal rotaton E (tor) = (k φ /) f(cosφ ) 15
16 Molecular dynamcs (MD) smulatons The Verlet algorthm The most wdely used method of ntegratng the equatons of moton s that ntally adopted by Verlet [1967].The method s based on postons r(t), acceleratons a (t), and the postons r(t -δt) from the prevous step. The equaton for advancng the postons reads as r(tδt) = r(t)-r(t-δt) δt a(t) The veloctes do not appear at all. They have been elmnated by addton of the equatons obtaned by Taylor expanson about r(t): r(tδt) = r(t) δt v(t) (1/) δt a(t)... r(t-δt) = r(t) - δt v(t) (1/) δt a(t)- The veloctes are not needed to compute the trajectores, but they are useful for estmatng the knetc energy (and hence the total energy). They may be obtaned from the formula v(t)= [r(tδt)-r(t-δt)]/δt Trajectory fle: Durng molecular dynamcs (and energy mnmzaton) the coordnates (and veloctes) are saved at regular ntervals. Such a fle s called a trajectory fle. Water Models A recent revew lsted 46 dstnct models, so ndrectly ndcatng ther lack of success n quanttatvely reproducng the propertes of real water. They may, however, offer useful nsght nto water's behavor. Implct Solvent Models Water molecules are not ncluded as molecules, but represented by an extra potental on the solvent accessble surface. only 50% slower than vacuum calculatons Explct Solvent Models Water molecules are explctly ncluded as ndvdual molecules. Force Felds for water molecules are not trval... Computatonally expensve... Models types a, b and c are all planar whereas type d s almost tetrahedral ~10 tmes faster than explct water MD Perodc Boundary Condtons (PBC) Buldng peptdes usng Z matrces Perodc boundary condtons are used to smulate solvated systems or crystals. In solvated systems, PBC prevents that the solvent "evaporates n slco" dstance angle dhedral connectvty N H H H (end of fle) (1 means optmze, 0 means keep constant, -1 means vary accordng to a desgnated pattern) 16
17 PDB Representaton PDB Representaton contd. HETATM 1 C HETATM C HETATM 3 C HETATM 4 H HETATM 5 H HETATM 6 H HETATM 7 H HETATM 8 H HETATM 9 H HETATM 10 H HETATM 11 H contnued... (blue ndcates data columns not utlzed/recognzed by all software) CONECT CONECT CONECT CONECT 4 1 CONECT 5 1 CONECT 6 CONECT 7 CONECT 8 3 CONECT 9 3 CONECT 10 3 CONECT 11 1 END Atom types (AMBER) Bond Parameters Angle Parameters Torson Parameters 17
18 Improper Torsons Van der Waals (LJ) Parameters 1 6 σ < σ = 4ε 1 j r r VLJ 6 Atomc Partal Charges Typcal Tme Scales... Bond stretchng: Elastc vbratons: Rotatons of surface sdechans: Hnge bendng: Rotaton of bured sde chans: Proten foldng: sec sec sec sec sec sec. Tmescale n MD: A Typcal tmestep n MD s 1 fs (10-15 sec) (deally 1/10 of the hghest frequency vbraton) Ab nto proten foldng smulaton Physcal tme for smulaton 10 4 seconds Typcal tme-step sze seconds Number of MD tme steps Atoms n a typcal proten and water smulaton 3,000 Approxmate number of nteractons n force calculaton 10 9 Machne nstructons per force calculaton 1000 Total number of machne nstructons 10 3 BlueGene capacty (floatng pont operatons per second) 1 pentaflop (10 15 ) 18
19 AMBER: J. Am. Chem. Soc. 117,
MOLECULAR DYNAMICS ,..., What is it? 2 = i i
 MOLECULAR DYNAMICS What s t? d d x t 2 m 2 = F ( x 1,..., x N ) =1,,N r ( x1 ( t),..., x ( t)) = v = ( x& 1 ( t ),..., x& ( t )) N N What are some uses of molecular smulatons and modelng? Conformatonal
MOLECULAR DYNAMICS What s t? d d x t 2 m 2 = F ( x 1,..., x N ) =1,,N r ( x1 ( t),..., x ( t)) = v = ( x& 1 ( t ),..., x& ( t )) N N What are some uses of molecular smulatons and modelng? Conformatonal
Problem Points Score Total 100
 Physcs 450 Solutons of Sample Exam I Problem Ponts Score 1 8 15 3 17 4 0 5 0 Total 100 All wor must be shown n order to receve full credt. Wor must be legble and comprehensble wth answers clearly ndcated.
Physcs 450 Solutons of Sample Exam I Problem Ponts Score 1 8 15 3 17 4 0 5 0 Total 100 All wor must be shown n order to receve full credt. Wor must be legble and comprehensble wth answers clearly ndcated.
Q e E i /k B. i i i i
 Water and Aqueous Solutons 3. Lattce Model of a Flud Lattce Models Lattce models provde a mnmalst, or coarse-graned, framework for descrbng the translatonal, rotatonal, and conformatonal degrees of freedom
Water and Aqueous Solutons 3. Lattce Model of a Flud Lattce Models Lattce models provde a mnmalst, or coarse-graned, framework for descrbng the translatonal, rotatonal, and conformatonal degrees of freedom
Introduction to Vapor/Liquid Equilibrium, part 2. Raoult s Law:
 CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
CE304, Sprng 2004 Lecture 4 Introducton to Vapor/Lqud Equlbrum, part 2 Raoult s Law: The smplest model that allows us do VLE calculatons s obtaned when we assume that the vapor phase s an deal gas, and
Thermodynamics General
 Thermodynamcs General Lecture 1 Lecture 1 s devoted to establshng buldng blocks for dscussng thermodynamcs. In addton, the equaton of state wll be establshed. I. Buldng blocks for thermodynamcs A. Dmensons,
Thermodynamcs General Lecture 1 Lecture 1 s devoted to establshng buldng blocks for dscussng thermodynamcs. In addton, the equaton of state wll be establshed. I. Buldng blocks for thermodynamcs A. Dmensons,
5.60 Thermodynamics & Kinetics Spring 2008
 MIT OpenCourseWare http://ocw.mt.edu 5.60 Thermodynamcs & Knetcs Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.60 Sprng 2008 Lecture #29 page 1
MIT OpenCourseWare http://ocw.mt.edu 5.60 Thermodynamcs & Knetcs Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.60 Sprng 2008 Lecture #29 page 1
Lecture 7: Boltzmann distribution & Thermodynamics of mixing
 Prof. Tbbtt Lecture 7 etworks & Gels Lecture 7: Boltzmann dstrbuton & Thermodynamcs of mxng 1 Suggested readng Prof. Mark W. Tbbtt ETH Zürch 13 März 018 Molecular Drvng Forces Dll and Bromberg: Chapters
Prof. Tbbtt Lecture 7 etworks & Gels Lecture 7: Boltzmann dstrbuton & Thermodynamcs of mxng 1 Suggested readng Prof. Mark W. Tbbtt ETH Zürch 13 März 018 Molecular Drvng Forces Dll and Bromberg: Chapters
CHAPTER 5 NUMERICAL EVALUATION OF DYNAMIC RESPONSE
 CHAPTER 5 NUMERICAL EVALUATION OF DYNAMIC RESPONSE Analytcal soluton s usually not possble when exctaton vares arbtrarly wth tme or f the system s nonlnear. Such problems can be solved by numercal tmesteppng
CHAPTER 5 NUMERICAL EVALUATION OF DYNAMIC RESPONSE Analytcal soluton s usually not possble when exctaton vares arbtrarly wth tme or f the system s nonlnear. Such problems can be solved by numercal tmesteppng
Energy, Entropy, and Availability Balances Phase Equilibria. Nonideal Thermodynamic Property Models. Selecting an Appropriate Model
 Lecture 4. Thermodynamcs [Ch. 2] Energy, Entropy, and Avalablty Balances Phase Equlbra - Fugactes and actvty coeffcents -K-values Nondeal Thermodynamc Property Models - P-v-T equaton-of-state models -
Lecture 4. Thermodynamcs [Ch. 2] Energy, Entropy, and Avalablty Balances Phase Equlbra - Fugactes and actvty coeffcents -K-values Nondeal Thermodynamc Property Models - P-v-T equaton-of-state models -
Numerical Heat and Mass Transfer
 Master degree n Mechancal Engneerng Numercal Heat and Mass Transfer 06-Fnte-Dfference Method (One-dmensonal, steady state heat conducton) Fausto Arpno f.arpno@uncas.t Introducton Why we use models and
Master degree n Mechancal Engneerng Numercal Heat and Mass Transfer 06-Fnte-Dfference Method (One-dmensonal, steady state heat conducton) Fausto Arpno f.arpno@uncas.t Introducton Why we use models and
Lecture 4. Macrostates and Microstates (Ch. 2 )
 Lecture 4. Macrostates and Mcrostates (Ch. ) The past three lectures: we have learned about thermal energy, how t s stored at the mcroscopc level, and how t can be transferred from one system to another.
Lecture 4. Macrostates and Mcrostates (Ch. ) The past three lectures: we have learned about thermal energy, how t s stored at the mcroscopc level, and how t can be transferred from one system to another.
Module 3 LOSSY IMAGE COMPRESSION SYSTEMS. Version 2 ECE IIT, Kharagpur
 Module 3 LOSSY IMAGE COMPRESSION SYSTEMS Verson ECE IIT, Kharagpur Lesson 6 Theory of Quantzaton Verson ECE IIT, Kharagpur Instructonal Objectves At the end of ths lesson, the students should be able to:
Module 3 LOSSY IMAGE COMPRESSION SYSTEMS Verson ECE IIT, Kharagpur Lesson 6 Theory of Quantzaton Verson ECE IIT, Kharagpur Instructonal Objectves At the end of ths lesson, the students should be able to:
Lecture 14: Forces and Stresses
 The Nuts and Bolts of Frst-Prncples Smulaton Lecture 14: Forces and Stresses Durham, 6th-13th December 2001 CASTEP Developers Group wth support from the ESF ψ k Network Overvew of Lecture Why bother? Theoretcal
The Nuts and Bolts of Frst-Prncples Smulaton Lecture 14: Forces and Stresses Durham, 6th-13th December 2001 CASTEP Developers Group wth support from the ESF ψ k Network Overvew of Lecture Why bother? Theoretcal
Chapter 13: Multiple Regression
 Chapter 13: Multple Regresson 13.1 Developng the multple-regresson Model The general model can be descrbed as: It smplfes for two ndependent varables: The sample ft parameter b 0, b 1, and b are used to
Chapter 13: Multple Regresson 13.1 Developng the multple-regresson Model The general model can be descrbed as: It smplfes for two ndependent varables: The sample ft parameter b 0, b 1, and b are used to
Supporting Materials
 Chrstopher Lockhart and Dmtr K. Klmov Molecular nteractons of Alzhemer's bomarker FDDNP wth A peptde Supportng Materals Parameterzaton of FDDNP molecule: CHARMM General Force Feld (CGenFF verson 2b6) was
Chrstopher Lockhart and Dmtr K. Klmov Molecular nteractons of Alzhemer's bomarker FDDNP wth A peptde Supportng Materals Parameterzaton of FDDNP molecule: CHARMM General Force Feld (CGenFF verson 2b6) was
Supporting Information
 Supportng Informaton The neural network f n Eq. 1 s gven by: f x l = ReLU W atom x l + b atom, 2 where ReLU s the element-wse rectfed lnear unt, 21.e., ReLUx = max0, x, W atom R d d s the weght matrx to
Supportng Informaton The neural network f n Eq. 1 s gven by: f x l = ReLU W atom x l + b atom, 2 where ReLU s the element-wse rectfed lnear unt, 21.e., ReLUx = max0, x, W atom R d d s the weght matrx to
5.04, Principles of Inorganic Chemistry II MIT Department of Chemistry Lecture 32: Vibrational Spectroscopy and the IR
 5.0, Prncples of Inorganc Chemstry II MIT Department of Chemstry Lecture 3: Vbratonal Spectroscopy and the IR Vbratonal spectroscopy s confned to the 00-5000 cm - spectral regon. The absorpton of a photon
5.0, Prncples of Inorganc Chemstry II MIT Department of Chemstry Lecture 3: Vbratonal Spectroscopy and the IR Vbratonal spectroscopy s confned to the 00-5000 cm - spectral regon. The absorpton of a photon
STATISTICAL MECHANICS
 STATISTICAL MECHANICS Thermal Energy Recall that KE can always be separated nto 2 terms: KE system = 1 2 M 2 total v CM KE nternal Rgd-body rotaton and elastc / sound waves Use smplfyng assumptons KE of
STATISTICAL MECHANICS Thermal Energy Recall that KE can always be separated nto 2 terms: KE system = 1 2 M 2 total v CM KE nternal Rgd-body rotaton and elastc / sound waves Use smplfyng assumptons KE of
NUMERICAL DIFFERENTIATION
 NUMERICAL DIFFERENTIATION 1 Introducton Dfferentaton s a method to compute the rate at whch a dependent output y changes wth respect to the change n the ndependent nput x. Ths rate of change s called the
NUMERICAL DIFFERENTIATION 1 Introducton Dfferentaton s a method to compute the rate at whch a dependent output y changes wth respect to the change n the ndependent nput x. Ths rate of change s called the
First Law: A body at rest remains at rest, a body in motion continues to move at constant velocity, unless acted upon by an external force.
 Secton 1. Dynamcs (Newton s Laws of Moton) Two approaches: 1) Gven all the forces actng on a body, predct the subsequent (changes n) moton. 2) Gven the (changes n) moton of a body, nfer what forces act
Secton 1. Dynamcs (Newton s Laws of Moton) Two approaches: 1) Gven all the forces actng on a body, predct the subsequent (changes n) moton. 2) Gven the (changes n) moton of a body, nfer what forces act
Generalized Linear Methods
 Generalzed Lnear Methods 1 Introducton In the Ensemble Methods the general dea s that usng a combnaton of several weak learner one could make a better learner. More formally, assume that we have a set
Generalzed Lnear Methods 1 Introducton In the Ensemble Methods the general dea s that usng a combnaton of several weak learner one could make a better learner. More formally, assume that we have a set
Transfer Functions. Convenient representation of a linear, dynamic model. A transfer function (TF) relates one input and one output: ( ) system
 Transfer Functons Convenent representaton of a lnear, dynamc model. A transfer functon (TF) relates one nput and one output: x t X s y t system Y s The followng termnology s used: x y nput output forcng
Transfer Functons Convenent representaton of a lnear, dynamc model. A transfer functon (TF) relates one nput and one output: x t X s y t system Y s The followng termnology s used: x y nput output forcng
Markov Chain Monte Carlo Lecture 6
 where (x 1,..., x N ) X N, N s called the populaton sze, f(x) f (x) for at least one {1, 2,..., N}, and those dfferent from f(x) are called the tral dstrbutons n terms of mportance samplng. Dfferent ways
where (x 1,..., x N ) X N, N s called the populaton sze, f(x) f (x) for at least one {1, 2,..., N}, and those dfferent from f(x) are called the tral dstrbutons n terms of mportance samplng. Dfferent ways
Amplification and Relaxation of Electron Spin Polarization in Semiconductor Devices
 Amplfcaton and Relaxaton of Electron Spn Polarzaton n Semconductor Devces Yury V. Pershn and Vladmr Prvman Center for Quantum Devce Technology, Clarkson Unversty, Potsdam, New York 13699-570, USA Spn Relaxaton
Amplfcaton and Relaxaton of Electron Spn Polarzaton n Semconductor Devces Yury V. Pershn and Vladmr Prvman Center for Quantum Devce Technology, Clarkson Unversty, Potsdam, New York 13699-570, USA Spn Relaxaton
CSci 6974 and ECSE 6966 Math. Tech. for Vision, Graphics and Robotics Lecture 21, April 17, 2006 Estimating A Plane Homography
 CSc 6974 and ECSE 6966 Math. Tech. for Vson, Graphcs and Robotcs Lecture 21, Aprl 17, 2006 Estmatng A Plane Homography Overvew We contnue wth a dscusson of the major ssues, usng estmaton of plane projectve
CSc 6974 and ECSE 6966 Math. Tech. for Vson, Graphcs and Robotcs Lecture 21, Aprl 17, 2006 Estmatng A Plane Homography Overvew We contnue wth a dscusson of the major ssues, usng estmaton of plane projectve
Week3, Chapter 4. Position and Displacement. Motion in Two Dimensions. Instantaneous Velocity. Average Velocity
 Week3, Chapter 4 Moton n Two Dmensons Lecture Quz A partcle confned to moton along the x axs moves wth constant acceleraton from x =.0 m to x = 8.0 m durng a 1-s tme nterval. The velocty of the partcle
Week3, Chapter 4 Moton n Two Dmensons Lecture Quz A partcle confned to moton along the x axs moves wth constant acceleraton from x =.0 m to x = 8.0 m durng a 1-s tme nterval. The velocty of the partcle
The Polarizable Pseudo-Particles Solvent Model
 LAMBE, UMR 8587 The Polarzable Pseudo-Partcles Solvent Model Nathale Basdevant, Tap Ha Duong, Danel Borgs LAMBE, Unversté d Evry and CNRS (UMR 8587) Ateler CFBo, 14 Ma 006 Solvaton: Some ssues Solvaton:
LAMBE, UMR 8587 The Polarzable Pseudo-Partcles Solvent Model Nathale Basdevant, Tap Ha Duong, Danel Borgs LAMBE, Unversté d Evry and CNRS (UMR 8587) Ateler CFBo, 14 Ma 006 Solvaton: Some ssues Solvaton:
Wilbur and Ague 4 WILBUR AND AGUE; APPENDIX DR1. Two-dimensional chemical maps as well as chemical profiles were done at 15 kv using
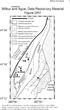 DR2006139 Wlbur and Ague 4 WILBUR AND AGUE; APPENDIX DR1 MINERAL ANALYSES Two-dmensonal chemcal maps as well as chemcal profles were done at 15 kv usng the JEOL JXA-8600 electron mcroprobe at Yale Unversty
DR2006139 Wlbur and Ague 4 WILBUR AND AGUE; APPENDIX DR1 MINERAL ANALYSES Two-dmensonal chemcal maps as well as chemcal profles were done at 15 kv usng the JEOL JXA-8600 electron mcroprobe at Yale Unversty
4DVAR, according to the name, is a four-dimensional variational method.
 4D-Varatonal Data Assmlaton (4D-Var) 4DVAR, accordng to the name, s a four-dmensonal varatonal method. 4D-Var s actually a drect generalzaton of 3D-Var to handle observatons that are dstrbuted n tme. The
4D-Varatonal Data Assmlaton (4D-Var) 4DVAR, accordng to the name, s a four-dmensonal varatonal method. 4D-Var s actually a drect generalzaton of 3D-Var to handle observatons that are dstrbuted n tme. The
THE VIBRATIONS OF MOLECULES II THE CARBON DIOXIDE MOLECULE Student Instructions
 THE VIBRATIONS OF MOLECULES II THE CARBON DIOXIDE MOLECULE Student Instructons by George Hardgrove Chemstry Department St. Olaf College Northfeld, MN 55057 hardgrov@lars.acc.stolaf.edu Copyrght George
THE VIBRATIONS OF MOLECULES II THE CARBON DIOXIDE MOLECULE Student Instructons by George Hardgrove Chemstry Department St. Olaf College Northfeld, MN 55057 hardgrov@lars.acc.stolaf.edu Copyrght George
Monte Carlo method II
 Course MP3 Lecture 5 14/11/2006 Monte Carlo method II How to put some real physcs nto the Monte Carlo method Dr James Ellott 5.1 Monte Carlo method revsted In lecture 4, we ntroduced the Monte Carlo (MC)
Course MP3 Lecture 5 14/11/2006 Monte Carlo method II How to put some real physcs nto the Monte Carlo method Dr James Ellott 5.1 Monte Carlo method revsted In lecture 4, we ntroduced the Monte Carlo (MC)
Chapter 5. Solution of System of Linear Equations. Module No. 6. Solution of Inconsistent and Ill Conditioned Systems
 Numercal Analyss by Dr. Anta Pal Assstant Professor Department of Mathematcs Natonal Insttute of Technology Durgapur Durgapur-713209 emal: anta.bue@gmal.com 1 . Chapter 5 Soluton of System of Lnear Equatons
Numercal Analyss by Dr. Anta Pal Assstant Professor Department of Mathematcs Natonal Insttute of Technology Durgapur Durgapur-713209 emal: anta.bue@gmal.com 1 . Chapter 5 Soluton of System of Lnear Equatons
Kernel Methods and SVMs Extension
 Kernel Methods and SVMs Extenson The purpose of ths document s to revew materal covered n Machne Learnng 1 Supervsed Learnng regardng support vector machnes (SVMs). Ths document also provdes a general
Kernel Methods and SVMs Extenson The purpose of ths document s to revew materal covered n Machne Learnng 1 Supervsed Learnng regardng support vector machnes (SVMs). Ths document also provdes a general
Supplementary Notes for Chapter 9 Mixture Thermodynamics
 Supplementary Notes for Chapter 9 Mxture Thermodynamcs Key ponts Nne major topcs of Chapter 9 are revewed below: 1. Notaton and operatonal equatons for mxtures 2. PVTN EOSs for mxtures 3. General effects
Supplementary Notes for Chapter 9 Mxture Thermodynamcs Key ponts Nne major topcs of Chapter 9 are revewed below: 1. Notaton and operatonal equatons for mxtures 2. PVTN EOSs for mxtures 3. General effects
Physics 181. Particle Systems
 Physcs 181 Partcle Systems Overvew In these notes we dscuss the varables approprate to the descrpton of systems of partcles, ther defntons, ther relatons, and ther conservatons laws. We consder a system
Physcs 181 Partcle Systems Overvew In these notes we dscuss the varables approprate to the descrpton of systems of partcles, ther defntons, ther relatons, and ther conservatons laws. We consder a system
Grand canonical Monte Carlo simulations of bulk electrolytes and calcium channels
 Grand canoncal Monte Carlo smulatons of bulk electrolytes and calcum channels Thess of Ph.D. dssertaton Prepared by: Attla Malascs M.Sc. n Chemstry Supervsor: Dr. Dezső Boda Unversty of Pannona Insttute
Grand canoncal Monte Carlo smulatons of bulk electrolytes and calcum channels Thess of Ph.D. dssertaton Prepared by: Attla Malascs M.Sc. n Chemstry Supervsor: Dr. Dezső Boda Unversty of Pannona Insttute
Linear Approximation with Regularization and Moving Least Squares
 Lnear Approxmaton wth Regularzaton and Movng Least Squares Igor Grešovn May 007 Revson 4.6 (Revson : March 004). 5 4 3 0.5 3 3.5 4 Contents: Lnear Fttng...4. Weghted Least Squares n Functon Approxmaton...
Lnear Approxmaton wth Regularzaton and Movng Least Squares Igor Grešovn May 007 Revson 4.6 (Revson : March 004). 5 4 3 0.5 3 3.5 4 Contents: Lnear Fttng...4. Weghted Least Squares n Functon Approxmaton...
Folding Proteins with Both Alpha and Beta Structures in a Reduced Model
 Foldng Protens wth Both Alpha and Beta Structures n a Reduced Model Nan-Yow Chen Department of Physcs, Natonal Tsng Hua Unversty, Hsnchu, Tawan, Republc of Chna Advsor: Chung-Yu Mou Department of Physcs,
Foldng Protens wth Both Alpha and Beta Structures n a Reduced Model Nan-Yow Chen Department of Physcs, Natonal Tsng Hua Unversty, Hsnchu, Tawan, Republc of Chna Advsor: Chung-Yu Mou Department of Physcs,
Some Comments on Accelerating Convergence of Iterative Sequences Using Direct Inversion of the Iterative Subspace (DIIS)
 Some Comments on Acceleratng Convergence of Iteratve Sequences Usng Drect Inverson of the Iteratve Subspace (DIIS) C. Davd Sherrll School of Chemstry and Bochemstry Georga Insttute of Technology May 1998
Some Comments on Acceleratng Convergence of Iteratve Sequences Usng Drect Inverson of the Iteratve Subspace (DIIS) C. Davd Sherrll School of Chemstry and Bochemstry Georga Insttute of Technology May 1998
Lecture 12: Discrete Laplacian
 Lecture 12: Dscrete Laplacan Scrbe: Tanye Lu Our goal s to come up wth a dscrete verson of Laplacan operator for trangulated surfaces, so that we can use t n practce to solve related problems We are mostly
Lecture 12: Dscrete Laplacan Scrbe: Tanye Lu Our goal s to come up wth a dscrete verson of Laplacan operator for trangulated surfaces, so that we can use t n practce to solve related problems We are mostly
5.62 Physical Chemistry II Spring 2008
 MIT OpenCourseWare http://ocw.mt.edu 5.62 Physcal Chemstry II Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.62 Sprng 2008 Lecture 34 Page Transton
MIT OpenCourseWare http://ocw.mt.edu 5.62 Physcal Chemstry II Sprng 2008 For nformaton about ctng these materals or our Terms of Use, vst: http://ocw.mt.edu/terms. 5.62 Sprng 2008 Lecture 34 Page Transton
ReaxFF Potential Functions
 ReaxFF Potental Functons Supportng nformaton for the manuscrpt A ReaxFF Reactve Force Feld for Molecular Dynamcs Smulatons of Hydrocarbon Oxdaton by Kmberly Chenoweth Adr CT van Dun and Wllam A Goddard
ReaxFF Potental Functons Supportng nformaton for the manuscrpt A ReaxFF Reactve Force Feld for Molecular Dynamcs Smulatons of Hydrocarbon Oxdaton by Kmberly Chenoweth Adr CT van Dun and Wllam A Goddard
Moments of Inertia. and reminds us of the analogous equation for linear momentum p= mv, which is of the form. The kinetic energy of the body is.
 Moments of Inerta Suppose a body s movng on a crcular path wth constant speed Let s consder two quanttes: the body s angular momentum L about the center of the crcle, and ts knetc energy T How are these
Moments of Inerta Suppose a body s movng on a crcular path wth constant speed Let s consder two quanttes: the body s angular momentum L about the center of the crcle, and ts knetc energy T How are these
EPR Paradox and the Physical Meaning of an Experiment in Quantum Mechanics. Vesselin C. Noninski
 EPR Paradox and the Physcal Meanng of an Experment n Quantum Mechancs Vesseln C Nonnsk vesselnnonnsk@verzonnet Abstract It s shown that there s one purely determnstc outcome when measurement s made on
EPR Paradox and the Physcal Meanng of an Experment n Quantum Mechancs Vesseln C Nonnsk vesselnnonnsk@verzonnet Abstract It s shown that there s one purely determnstc outcome when measurement s made on
Physics 5153 Classical Mechanics. Principle of Virtual Work-1
 P. Guterrez 1 Introducton Physcs 5153 Classcal Mechancs Prncple of Vrtual Work The frst varatonal prncple we encounter n mechancs s the prncple of vrtual work. It establshes the equlbrum condton of a mechancal
P. Guterrez 1 Introducton Physcs 5153 Classcal Mechancs Prncple of Vrtual Work The frst varatonal prncple we encounter n mechancs s the prncple of vrtual work. It establshes the equlbrum condton of a mechancal
Modeling of Dynamic Systems
 Modelng of Dynamc Systems Ref: Control System Engneerng Norman Nse : Chapters & 3 Chapter objectves : Revew the Laplace transform Learn how to fnd a mathematcal model, called a transfer functon Learn how
Modelng of Dynamc Systems Ref: Control System Engneerng Norman Nse : Chapters & 3 Chapter objectves : Revew the Laplace transform Learn how to fnd a mathematcal model, called a transfer functon Learn how
Problem Set 9 Solutions
 Desgn and Analyss of Algorthms May 4, 2015 Massachusetts Insttute of Technology 6.046J/18.410J Profs. Erk Demane, Srn Devadas, and Nancy Lynch Problem Set 9 Solutons Problem Set 9 Solutons Ths problem
Desgn and Analyss of Algorthms May 4, 2015 Massachusetts Insttute of Technology 6.046J/18.410J Profs. Erk Demane, Srn Devadas, and Nancy Lynch Problem Set 9 Solutons Problem Set 9 Solutons Ths problem
PHYS 705: Classical Mechanics. Newtonian Mechanics
 1 PHYS 705: Classcal Mechancs Newtonan Mechancs Quck Revew of Newtonan Mechancs Basc Descrpton: -An dealzed pont partcle or a system of pont partcles n an nertal reference frame [Rgd bodes (ch. 5 later)]
1 PHYS 705: Classcal Mechancs Newtonan Mechancs Quck Revew of Newtonan Mechancs Basc Descrpton: -An dealzed pont partcle or a system of pont partcles n an nertal reference frame [Rgd bodes (ch. 5 later)]
Week 8: Chapter 9. Linear Momentum. Newton Law and Momentum. Linear Momentum, cont. Conservation of Linear Momentum. Conservation of Momentum, 2
 Lnear omentum Week 8: Chapter 9 Lnear omentum and Collsons The lnear momentum of a partcle, or an object that can be modeled as a partcle, of mass m movng wth a velocty v s defned to be the product of
Lnear omentum Week 8: Chapter 9 Lnear omentum and Collsons The lnear momentum of a partcle, or an object that can be modeled as a partcle, of mass m movng wth a velocty v s defned to be the product of
Computational Fluid Dynamics. Smoothed Particle Hydrodynamics. Simulations. Smoothing Kernels and Basis of SPH
 Computatonal Flud Dynamcs If you want to learn a bt more of the math behnd flud dynamcs, read my prevous post about the Naver- Stokes equatons and Newtonan fluds. The equatons derved n the post are the
Computatonal Flud Dynamcs If you want to learn a bt more of the math behnd flud dynamcs, read my prevous post about the Naver- Stokes equatons and Newtonan fluds. The equatons derved n the post are the
Lecture Notes on Linear Regression
 Lecture Notes on Lnear Regresson Feng L fl@sdueducn Shandong Unversty, Chna Lnear Regresson Problem In regresson problem, we am at predct a contnuous target value gven an nput feature vector We assume
Lecture Notes on Lnear Regresson Feng L fl@sdueducn Shandong Unversty, Chna Lnear Regresson Problem In regresson problem, we am at predct a contnuous target value gven an nput feature vector We assume
Probabilistic method to determine electron correlation energy
 Probablstc method to determne electron elaton energy T.R.S. Prasanna Department of Metallurgcal Engneerng and Materals Scence Indan Insttute of Technology, Bombay Mumba 400076 Inda A new method to determne
Probablstc method to determne electron elaton energy T.R.S. Prasanna Department of Metallurgcal Engneerng and Materals Scence Indan Insttute of Technology, Bombay Mumba 400076 Inda A new method to determne
Temperature. Chapter Heat Engine
 Chapter 3 Temperature In prevous chapters of these notes we ntroduced the Prncple of Maxmum ntropy as a technque for estmatng probablty dstrbutons consstent wth constrants. In Chapter 9 we dscussed the
Chapter 3 Temperature In prevous chapters of these notes we ntroduced the Prncple of Maxmum ntropy as a technque for estmatng probablty dstrbutons consstent wth constrants. In Chapter 9 we dscussed the
...Thermodynamics. If Clausius Clapeyron fails. l T (v 2 v 1 ) = 0/0 Second order phase transition ( S, v = 0)
 If Clausus Clapeyron fals ( ) dp dt pb =...Thermodynamcs l T (v 2 v 1 ) = 0/0 Second order phase transton ( S, v = 0) ( ) dp = c P,1 c P,2 dt Tv(β 1 β 2 ) Two phases ntermngled Ferromagnet (Excess spn-up
If Clausus Clapeyron fals ( ) dp dt pb =...Thermodynamcs l T (v 2 v 1 ) = 0/0 Second order phase transton ( S, v = 0) ( ) dp = c P,1 c P,2 dt Tv(β 1 β 2 ) Two phases ntermngled Ferromagnet (Excess spn-up
Rigid body simulation
 Rgd bod smulaton Rgd bod smulaton Once we consder an object wth spacal etent, partcle sstem smulaton s no longer suffcent Problems Problems Unconstraned sstem rotatonal moton torques and angular momentum
Rgd bod smulaton Rgd bod smulaton Once we consder an object wth spacal etent, partcle sstem smulaton s no longer suffcent Problems Problems Unconstraned sstem rotatonal moton torques and angular momentum
The Feynman path integral
 The Feynman path ntegral Aprl 3, 205 Hesenberg and Schrödnger pctures The Schrödnger wave functon places the tme dependence of a physcal system n the state, ψ, t, where the state s a vector n Hlbert space
The Feynman path ntegral Aprl 3, 205 Hesenberg and Schrödnger pctures The Schrödnger wave functon places the tme dependence of a physcal system n the state, ψ, t, where the state s a vector n Hlbert space
ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM
 ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM An elastc wave s a deformaton of the body that travels throughout the body n all drectons. We can examne the deformaton over a perod of tme by fxng our look
ELASTIC WAVE PROPAGATION IN A CONTINUOUS MEDIUM An elastc wave s a deformaton of the body that travels throughout the body n all drectons. We can examne the deformaton over a perod of tme by fxng our look
Note 10. Modeling and Simulation of Dynamic Systems
 Lecture Notes of ME 475: Introducton to Mechatroncs Note 0 Modelng and Smulaton of Dynamc Systems Department of Mechancal Engneerng, Unversty Of Saskatchewan, 57 Campus Drve, Saskatoon, SK S7N 5A9, Canada
Lecture Notes of ME 475: Introducton to Mechatroncs Note 0 Modelng and Smulaton of Dynamc Systems Department of Mechancal Engneerng, Unversty Of Saskatchewan, 57 Campus Drve, Saskatoon, SK S7N 5A9, Canada
SOLUTION/EXAMPLES. Contact during the exam: phone: , EXAM TBT4135 BIOPOLYMERS. 14 December Time:
 1 NRWEGIN UNIVERSITY F SCIENCE ND TECHNLGY DEPRTMENT F BITECHNLGY Professor Bjørn E. Chrstensen, Department of Botechnology Contact durng the exam: phone: 73593327, 92634016 EXM TBT4135 BIPLYMERS 14 December
1 NRWEGIN UNIVERSITY F SCIENCE ND TECHNLGY DEPRTMENT F BITECHNLGY Professor Bjørn E. Chrstensen, Department of Botechnology Contact durng the exam: phone: 73593327, 92634016 EXM TBT4135 BIPLYMERS 14 December
Week 9 Chapter 10 Section 1-5
 Week 9 Chapter 10 Secton 1-5 Rotaton Rgd Object A rgd object s one that s nondeformable The relatve locatons of all partcles makng up the object reman constant All real objects are deformable to some extent,
Week 9 Chapter 10 Secton 1-5 Rotaton Rgd Object A rgd object s one that s nondeformable The relatve locatons of all partcles makng up the object reman constant All real objects are deformable to some extent,
Prof. Dr. I. Nasser Phys 630, T Aug-15 One_dimensional_Ising_Model
 EXACT OE-DIMESIOAL ISIG MODEL The one-dmensonal Isng model conssts of a chan of spns, each spn nteractng only wth ts two nearest neghbors. The smple Isng problem n one dmenson can be solved drectly n several
EXACT OE-DIMESIOAL ISIG MODEL The one-dmensonal Isng model conssts of a chan of spns, each spn nteractng only wth ts two nearest neghbors. The smple Isng problem n one dmenson can be solved drectly n several
where the sums are over the partcle labels. In general H = p2 2m + V s(r ) V j = V nt (jr, r j j) (5) where V s s the sngle-partcle potental and V nt
 Physcs 543 Quantum Mechancs II Fall 998 Hartree-Fock and the Self-consstent Feld Varatonal Methods In the dscusson of statonary perturbaton theory, I mentoned brey the dea of varatonal approxmaton schemes.
Physcs 543 Quantum Mechancs II Fall 998 Hartree-Fock and the Self-consstent Feld Varatonal Methods In the dscusson of statonary perturbaton theory, I mentoned brey the dea of varatonal approxmaton schemes.
Gouy-Chapman model (1910) The double layer is not as compact as in Helmholtz rigid layer.
 CHE465/865, 6-3, Lecture 1, 7 nd Sep., 6 Gouy-Chapman model (191) The double layer s not as compact as n Helmholtz rgd layer. Consder thermal motons of ons: Tendency to ncrease the entropy and make the
CHE465/865, 6-3, Lecture 1, 7 nd Sep., 6 Gouy-Chapman model (191) The double layer s not as compact as n Helmholtz rgd layer. Consder thermal motons of ons: Tendency to ncrease the entropy and make the
Feature Selection & Dynamic Tracking F&P Textbook New: Ch 11, Old: Ch 17 Guido Gerig CS 6320, Spring 2013
 Feature Selecton & Dynamc Trackng F&P Textbook New: Ch 11, Old: Ch 17 Gudo Gerg CS 6320, Sprng 2013 Credts: Materal Greg Welch & Gary Bshop, UNC Chapel Hll, some sldes modfed from J.M. Frahm/ M. Pollefeys,
Feature Selecton & Dynamc Trackng F&P Textbook New: Ch 11, Old: Ch 17 Gudo Gerg CS 6320, Sprng 2013 Credts: Materal Greg Welch & Gary Bshop, UNC Chapel Hll, some sldes modfed from J.M. Frahm/ M. Pollefeys,
2 Finite difference basics
 Numersche Methoden 1, WS 11/12 B.J.P. Kaus 2 Fnte dfference bascs Consder the one- The bascs of the fnte dfference method are best understood wth an example. dmensonal transent heat conducton equaton T
Numersche Methoden 1, WS 11/12 B.J.P. Kaus 2 Fnte dfference bascs Consder the one- The bascs of the fnte dfference method are best understood wth an example. dmensonal transent heat conducton equaton T
Feature Selection: Part 1
 CSE 546: Machne Learnng Lecture 5 Feature Selecton: Part 1 Instructor: Sham Kakade 1 Regresson n the hgh dmensonal settng How do we learn when the number of features d s greater than the sample sze n?
CSE 546: Machne Learnng Lecture 5 Feature Selecton: Part 1 Instructor: Sham Kakade 1 Regresson n the hgh dmensonal settng How do we learn when the number of features d s greater than the sample sze n?
Solutions of execrises
 Solutons of execrses 1) XPS spectrum analyss The fgure below shows an XPS spectrum measured on the surface of a clean nsoluble homo-polyether. Usng the formulas and tables n ths document, answer the followng
Solutons of execrses 1) XPS spectrum analyss The fgure below shows an XPS spectrum measured on the surface of a clean nsoluble homo-polyether. Usng the formulas and tables n ths document, answer the followng
NP-Completeness : Proofs
 NP-Completeness : Proofs Proof Methods A method to show a decson problem Π NP-complete s as follows. (1) Show Π NP. (2) Choose an NP-complete problem Π. (3) Show Π Π. A method to show an optmzaton problem
NP-Completeness : Proofs Proof Methods A method to show a decson problem Π NP-complete s as follows. (1) Show Π NP. (2) Choose an NP-complete problem Π. (3) Show Π Π. A method to show an optmzaton problem
Frequency dependence of the permittivity
 Frequency dependence of the permttvty February 7, 016 In materals, the delectrc constant and permeablty are actually frequency dependent. Ths does not affect our results for sngle frequency modes, but
Frequency dependence of the permttvty February 7, 016 In materals, the delectrc constant and permeablty are actually frequency dependent. Ths does not affect our results for sngle frequency modes, but
ENGN 40 Dynamics and Vibrations Homework # 7 Due: Friday, April 15
 NGN 40 ynamcs and Vbratons Homework # 7 ue: Frday, Aprl 15 1. Consder a concal hostng drum used n the mnng ndustry to host a mass up/down. A cable of dameter d has the mass connected at one end and s wound/unwound
NGN 40 ynamcs and Vbratons Homework # 7 ue: Frday, Aprl 15 1. Consder a concal hostng drum used n the mnng ndustry to host a mass up/down. A cable of dameter d has the mass connected at one end and s wound/unwound
1. Mean-Field Theory. 2. Bjerrum length
 1. Mean-Feld Theory Contnuum models lke the Posson-Nernst-Planck equatons are mean-feld approxmatons whch descrbe how dscrete ons are affected by the mean concentratons c and potental φ. Each on mgrates
1. Mean-Feld Theory Contnuum models lke the Posson-Nernst-Planck equatons are mean-feld approxmatons whch descrbe how dscrete ons are affected by the mean concentratons c and potental φ. Each on mgrates
is the calculated value of the dependent variable at point i. The best parameters have values that minimize the squares of the errors
 Multple Lnear and Polynomal Regresson wth Statstcal Analyss Gven a set of data of measured (or observed) values of a dependent varable: y versus n ndependent varables x 1, x, x n, multple lnear regresson
Multple Lnear and Polynomal Regresson wth Statstcal Analyss Gven a set of data of measured (or observed) values of a dependent varable: y versus n ndependent varables x 1, x, x n, multple lnear regresson
Tracking with Kalman Filter
 Trackng wth Kalman Flter Scott T. Acton Vrgna Image and Vdeo Analyss (VIVA), Charles L. Brown Department of Electrcal and Computer Engneerng Department of Bomedcal Engneerng Unversty of Vrgna, Charlottesvlle,
Trackng wth Kalman Flter Scott T. Acton Vrgna Image and Vdeo Analyss (VIVA), Charles L. Brown Department of Electrcal and Computer Engneerng Department of Bomedcal Engneerng Unversty of Vrgna, Charlottesvlle,
Global Sensitivity. Tuesday 20 th February, 2018
 Global Senstvty Tuesday 2 th February, 28 ) Local Senstvty Most senstvty analyses [] are based on local estmates of senstvty, typcally by expandng the response n a Taylor seres about some specfc values
Global Senstvty Tuesday 2 th February, 28 ) Local Senstvty Most senstvty analyses [] are based on local estmates of senstvty, typcally by expandng the response n a Taylor seres about some specfc values
Composite Hypotheses testing
 Composte ypotheses testng In many hypothess testng problems there are many possble dstrbutons that can occur under each of the hypotheses. The output of the source s a set of parameters (ponts n a parameter
Composte ypotheses testng In many hypothess testng problems there are many possble dstrbutons that can occur under each of the hypotheses. The output of the source s a set of parameters (ponts n a parameter
Physics 5153 Classical Mechanics. D Alembert s Principle and The Lagrangian-1
 P. Guterrez Physcs 5153 Classcal Mechancs D Alembert s Prncple and The Lagrangan 1 Introducton The prncple of vrtual work provdes a method of solvng problems of statc equlbrum wthout havng to consder the
P. Guterrez Physcs 5153 Classcal Mechancs D Alembert s Prncple and The Lagrangan 1 Introducton The prncple of vrtual work provdes a method of solvng problems of statc equlbrum wthout havng to consder the
Difference Equations
 Dfference Equatons c Jan Vrbk 1 Bascs Suppose a sequence of numbers, say a 0,a 1,a,a 3,... s defned by a certan general relatonshp between, say, three consecutve values of the sequence, e.g. a + +3a +1
Dfference Equatons c Jan Vrbk 1 Bascs Suppose a sequence of numbers, say a 0,a 1,a,a 3,... s defned by a certan general relatonshp between, say, three consecutve values of the sequence, e.g. a + +3a +1
Georgia Tech PHYS 6124 Mathematical Methods of Physics I
 Georga Tech PHYS 624 Mathematcal Methods of Physcs I Instructor: Predrag Cvtanovć Fall semester 202 Homework Set #7 due October 30 202 == show all your work for maxmum credt == put labels ttle legends
Georga Tech PHYS 624 Mathematcal Methods of Physcs I Instructor: Predrag Cvtanovć Fall semester 202 Homework Set #7 due October 30 202 == show all your work for maxmum credt == put labels ttle legends
University of Washington Department of Chemistry Chemistry 453 Winter Quarter 2015
 Lecture 2. 1/07/15-1/09/15 Unversty of Washngton Department of Chemstry Chemstry 453 Wnter Quarter 2015 We are not talkng about truth. We are talkng about somethng that seems lke truth. The truth we want
Lecture 2. 1/07/15-1/09/15 Unversty of Washngton Department of Chemstry Chemstry 453 Wnter Quarter 2015 We are not talkng about truth. We are talkng about somethng that seems lke truth. The truth we want
3.1 Expectation of Functions of Several Random Variables. )' be a k-dimensional discrete or continuous random vector, with joint PMF p (, E X E X1 E X
 Statstcs 1: Probablty Theory II 37 3 EPECTATION OF SEVERAL RANDOM VARIABLES As n Probablty Theory I, the nterest n most stuatons les not on the actual dstrbuton of a random vector, but rather on a number
Statstcs 1: Probablty Theory II 37 3 EPECTATION OF SEVERAL RANDOM VARIABLES As n Probablty Theory I, the nterest n most stuatons les not on the actual dstrbuton of a random vector, but rather on a number
ECEN 5005 Crystals, Nanocrystals and Device Applications Class 19 Group Theory For Crystals
 ECEN 5005 Crystals, Nanocrystals and Devce Applcatons Class 9 Group Theory For Crystals Dee Dagram Radatve Transton Probablty Wgner-Ecart Theorem Selecton Rule Dee Dagram Expermentally determned energy
ECEN 5005 Crystals, Nanocrystals and Devce Applcatons Class 9 Group Theory For Crystals Dee Dagram Radatve Transton Probablty Wgner-Ecart Theorem Selecton Rule Dee Dagram Expermentally determned energy
Mathematical Preparations
 1 Introducton Mathematcal Preparatons The theory of relatvty was developed to explan experments whch studed the propagaton of electromagnetc radaton n movng coordnate systems. Wthn expermental error the
1 Introducton Mathematcal Preparatons The theory of relatvty was developed to explan experments whch studed the propagaton of electromagnetc radaton n movng coordnate systems. Wthn expermental error the
Markov Chain Monte Carlo (MCMC), Gibbs Sampling, Metropolis Algorithms, and Simulated Annealing Bioinformatics Course Supplement
 Markov Chan Monte Carlo MCMC, Gbbs Samplng, Metropols Algorthms, and Smulated Annealng 2001 Bonformatcs Course Supplement SNU Bontellgence Lab http://bsnuackr/ Outlne! Markov Chan Monte Carlo MCMC! Metropols-Hastngs
Markov Chan Monte Carlo MCMC, Gbbs Samplng, Metropols Algorthms, and Smulated Annealng 2001 Bonformatcs Course Supplement SNU Bontellgence Lab http://bsnuackr/ Outlne! Markov Chan Monte Carlo MCMC! Metropols-Hastngs
Nice plotting of proteins II
 Nce plottng of protens II Fnal remark regardng effcency: It s possble to wrte the Newton representaton n a way that can be computed effcently, usng smlar bracketng that we made for the frst representaton
Nce plottng of protens II Fnal remark regardng effcency: It s possble to wrte the Newton representaton n a way that can be computed effcently, usng smlar bracketng that we made for the frst representaton
Chapter 8 Indicator Variables
 Chapter 8 Indcator Varables In general, e explanatory varables n any regresson analyss are assumed to be quanttatve n nature. For example, e varables lke temperature, dstance, age etc. are quanttatve n
Chapter 8 Indcator Varables In general, e explanatory varables n any regresson analyss are assumed to be quanttatve n nature. For example, e varables lke temperature, dstance, age etc. are quanttatve n
Workshop: Approximating energies and wave functions Quantum aspects of physical chemistry
 Workshop: Approxmatng energes and wave functons Quantum aspects of physcal chemstry http://quantum.bu.edu/pltl/6/6.pdf Last updated Thursday, November 7, 25 7:9:5-5: Copyrght 25 Dan Dll (dan@bu.edu) Department
Workshop: Approxmatng energes and wave functons Quantum aspects of physcal chemstry http://quantum.bu.edu/pltl/6/6.pdf Last updated Thursday, November 7, 25 7:9:5-5: Copyrght 25 Dan Dll (dan@bu.edu) Department
STATISTICAL MECHANICAL ENSEMBLES 1 MICROSCOPIC AND MACROSCOPIC VARIABLES PHASE SPACE ENSEMBLES. CHE 524 A. Panagiotopoulos 1
 CHE 54 A. Panagotopoulos STATSTCAL MECHACAL ESEMBLES MCROSCOPC AD MACROSCOPC ARABLES The central queston n Statstcal Mechancs can be phrased as follows: f partcles (atoms, molecules, electrons, nucle,
CHE 54 A. Panagotopoulos STATSTCAL MECHACAL ESEMBLES MCROSCOPC AD MACROSCOPC ARABLES The central queston n Statstcal Mechancs can be phrased as follows: f partcles (atoms, molecules, electrons, nucle,
( ) 1/ 2. ( P SO2 )( P O2 ) 1/ 2.
 Chemstry 360 Dr. Jean M. Standard Problem Set 9 Solutons. The followng chemcal reacton converts sulfur doxde to sulfur troxde. SO ( g) + O ( g) SO 3 ( l). (a.) Wrte the expresson for K eq for ths reacton.
Chemstry 360 Dr. Jean M. Standard Problem Set 9 Solutons. The followng chemcal reacton converts sulfur doxde to sulfur troxde. SO ( g) + O ( g) SO 3 ( l). (a.) Wrte the expresson for K eq for ths reacton.
PHYS 705: Classical Mechanics. Calculus of Variations II
 1 PHYS 705: Classcal Mechancs Calculus of Varatons II 2 Calculus of Varatons: Generalzaton (no constrant yet) Suppose now that F depends on several dependent varables : We need to fnd such that has a statonary
1 PHYS 705: Classcal Mechancs Calculus of Varatons II 2 Calculus of Varatons: Generalzaton (no constrant yet) Suppose now that F depends on several dependent varables : We need to fnd such that has a statonary
Robert Eisberg Second edition CH 09 Multielectron atoms ground states and x-ray excitations
 Quantum Physcs 量 理 Robert Esberg Second edton CH 09 Multelectron atoms ground states and x-ray exctatons 9-01 By gong through the procedure ndcated n the text, develop the tme-ndependent Schroednger equaton
Quantum Physcs 量 理 Robert Esberg Second edton CH 09 Multelectron atoms ground states and x-ray exctatons 9-01 By gong through the procedure ndcated n the text, develop the tme-ndependent Schroednger equaton
Second Order Analysis
 Second Order Analyss In the prevous classes we looked at a method that determnes the load correspondng to a state of bfurcaton equlbrum of a perfect frame by egenvalye analyss The system was assumed to
Second Order Analyss In the prevous classes we looked at a method that determnes the load correspondng to a state of bfurcaton equlbrum of a perfect frame by egenvalye analyss The system was assumed to
Chapter 11: Simple Linear Regression and Correlation
 Chapter 11: Smple Lnear Regresson and Correlaton 11-1 Emprcal Models 11-2 Smple Lnear Regresson 11-3 Propertes of the Least Squares Estmators 11-4 Hypothess Test n Smple Lnear Regresson 11-4.1 Use of t-tests
Chapter 11: Smple Lnear Regresson and Correlaton 11-1 Emprcal Models 11-2 Smple Lnear Regresson 11-3 Propertes of the Least Squares Estmators 11-4 Hypothess Test n Smple Lnear Regresson 11-4.1 Use of t-tests
THERMAL DISTRIBUTION IN THE HCL SPECTRUM OBJECTIVE
 ame: THERMAL DISTRIBUTIO I THE HCL SPECTRUM OBJECTIVE To nvestgate a system s thermal dstrbuton n dscrete states; specfcally, determne HCl gas temperature from the relatve occupatons of ts rotatonal states.
ame: THERMAL DISTRIBUTIO I THE HCL SPECTRUM OBJECTIVE To nvestgate a system s thermal dstrbuton n dscrete states; specfcally, determne HCl gas temperature from the relatve occupatons of ts rotatonal states.
Image Processing for Bubble Detection in Microfluidics
 Image Processng for Bubble Detecton n Mcrofludcs Introducton Chen Fang Mechancal Engneerng Department Stanford Unverst Startng from recentl ears, mcrofludcs devces have been wdel used to buld the bomedcal
Image Processng for Bubble Detecton n Mcrofludcs Introducton Chen Fang Mechancal Engneerng Department Stanford Unverst Startng from recentl ears, mcrofludcs devces have been wdel used to buld the bomedcal
Errors for Linear Systems
 Errors for Lnear Systems When we solve a lnear system Ax b we often do not know A and b exactly, but have only approxmatons  and ˆb avalable. Then the best thng we can do s to solve ˆx ˆb exactly whch
Errors for Lnear Systems When we solve a lnear system Ax b we often do not know A and b exactly, but have only approxmatons  and ˆb avalable. Then the best thng we can do s to solve ˆx ˆb exactly whch
Physics 141. Lecture 14. Frank L. H. Wolfs Department of Physics and Astronomy, University of Rochester, Lecture 14, Page 1
 Physcs 141. Lecture 14. Frank L. H. Wolfs Department of Physcs and Astronomy, Unversty of Rochester, Lecture 14, Page 1 Physcs 141. Lecture 14. Course Informaton: Lab report # 3. Exam # 2. Mult-Partcle
Physcs 141. Lecture 14. Frank L. H. Wolfs Department of Physcs and Astronomy, Unversty of Rochester, Lecture 14, Page 1 Physcs 141. Lecture 14. Course Informaton: Lab report # 3. Exam # 2. Mult-Partcle
This chapter illustrates the idea that all properties of the homogeneous electron gas (HEG) can be calculated from electron density.
 1 Unform Electron Gas Ths chapter llustrates the dea that all propertes of the homogeneous electron gas (HEG) can be calculated from electron densty. Intutve Representaton of Densty Electron densty n s
1 Unform Electron Gas Ths chapter llustrates the dea that all propertes of the homogeneous electron gas (HEG) can be calculated from electron densty. Intutve Representaton of Densty Electron densty n s
Chapter 9: Statistical Inference and the Relationship between Two Variables
 Chapter 9: Statstcal Inference and the Relatonshp between Two Varables Key Words The Regresson Model The Sample Regresson Equaton The Pearson Correlaton Coeffcent Learnng Outcomes After studyng ths chapter,
Chapter 9: Statstcal Inference and the Relatonshp between Two Varables Key Words The Regresson Model The Sample Regresson Equaton The Pearson Correlaton Coeffcent Learnng Outcomes After studyng ths chapter,
Boostrapaggregating (Bagging)
 Boostrapaggregatng (Baggng) An ensemble meta-algorthm desgned to mprove the stablty and accuracy of machne learnng algorthms Can be used n both regresson and classfcaton Reduces varance and helps to avod
Boostrapaggregatng (Baggng) An ensemble meta-algorthm desgned to mprove the stablty and accuracy of machne learnng algorthms Can be used n both regresson and classfcaton Reduces varance and helps to avod
Chapter Newton s Method
 Chapter 9. Newton s Method After readng ths chapter, you should be able to:. Understand how Newton s method s dfferent from the Golden Secton Search method. Understand how Newton s method works 3. Solve
Chapter 9. Newton s Method After readng ths chapter, you should be able to:. Understand how Newton s method s dfferent from the Golden Secton Search method. Understand how Newton s method works 3. Solve
