Multiservice Tactics, Techniques, and Procedures for Nuclear, Biological, and Chemical (NBC) Protection
|
|
|
- Deborah Hopkins
- 5 years ago
- Views:
Transcription
1 FM /MCWP /NTTP /AFTTP(I) C1 Change 1 Headquarters Department of the Army United States Marine Corps United States Navy United States Air Force Washington, DC, 31 December 2009 Multiservice Tactics, Techniques, and Procedures for Nuclear, Biological, and Chemical (NBC) Protection 1. Change Field Manual (FM) /Marine Corps Warfighting Publication (MCWP) /Navy Tactics, Techniques, and Procedures (NTTP) /Air Force Tactics, Techniques, and Procedures (Interservice) (AFTTP[I]) , 2 June 2003, as follows: Remove old pages: Insert new pages: E-3 and E-4 E-3 and E-4 2. This change is based on the Edgewood Chemical Biological Center s Toxic Industrial Chemical Assessment of Nuclear, Biological, and Chemical (NBC) Filter Performance (ECBC- TR-093). It was originally thought that the protection afforded to personnel using chemical, biological, radiological, and nuclear (CBRN) filters against fuming nitric acid was effective. However, recent reports have shown that the protection afforded to personnel using CBRN filters against fuming nitric acid is poor. 3. A bar ( ) marks new or changed material. 4. File this transmittal sheet in front of the publication. DISTRIBUTION RESTRICTION: Approved for public release; distribution is unlimited.
2 FM /MCWP /NTTP /AFTTP(I) C1 31 December 2009 By Order of the Secretary of the Army: GEORGE W. CASEY, JR. General, United States Army Chief of Staff Official: JOYCE E. MORROW Administrative Assistant to the Secretary of the Army DISTRIBUTION: Active Army, Army National Guard, and U.S. Army Reserve: To be distributed in accordance with the initial distribution number , requirements for FM By Order of the Secretary of the Air Force MAX E. KIRSCHBAUM Colonel, USAF Commander Headquarters, Air Force Civil Engineer Support Agency Air Force Distribution: F Marine Corps PCN: PIN:
3 Table E-1. Protection Afforded by NBC Filters for Selected TIC High Hazard Medium Hazard Low Hazard ammonia - P acetone cyanohydrin - M allyl isothiocyanate - E arsine - E acrolein - P arsenic trichloride - M boron trichloride - E acrylonitrile - P bromine - P boron trifluoride - E allyl alcohol - M bromine chloride - M carbon disulfide - P allyl amine - P bromine pentafluoride - M chlorine - E allyl chlorocarbonate - M bromine trifluoride - M diborane - E boron tribromide - M carbonyl fluoride - P ethylene oxide - P carbon monoxide - P chlorine pentafluoride - M fluorine - E carbonyl sulfide - P chlorine trifluoride - M formaldehyde - P chloroacetone - M chloroacetaldehyde - M hydrogen bromide - E chloroacetonitrile - M chloroacetyl chloride - M hydrogen chloride - E chlorosulfonic acid - E cyanogen - E hydrogen cyanide - E crotonaldehyde - M diphenylmethane-4 diisocyanate - E hydrogen fluoride - E diketene - M ethyl chloroformate - M hydrogen sulfide - E 1,2-dimethyl hydrazine - P ethyl chlorothioformate - E nitric acid, fuming - P dimethyl sulfate - E ethylene imine - P phosgene - E ethylene dibromide - M ethylphosphonothioicdichloride - E phosphorus trichloride - E hydrogen selenide - P ethyl phosphonous dichloride - M sulfur dioxide - E iron pentacarbonyl - M hexachlorocyclopentadiene - E sulfuric acid - E methanesulfonyl chloride - E hydrogen iodide - P tungsten hexafluoride - E methyl bromide - P isobutyl chloroformate - M methyl chloroformate - P isopropyl chloroformite - M methyl chlorosilane - P n-butyl chloroformate - M methyl hydrazine - M nitric oxide - P methyl isocyanate - P n-propyl chloroformate - M methyl mercaptan - P isopropyl - P n-butyl isocyanate - M parathion - E nitrogen dioxide - P perchloromethyl mercaptan - E phosphine - M sec-butyl chloroformate - M trichloroacetyl chloride - M sulfuryl fluoride - P phosphorus oxychloride - M tert-butyl isocyanate - M phosphorus pentafluoride - P tetraethyl lead - E selenium hexafluoride - E tetraethyl pyrophosphate - E silicon tetrafluoride - P tetramethyl lead - M stibine - P toluene 2,4-diisocyanate - E sulfur trioxide - M toluene 2,6-diisocyanate - E sulfuryl chloride - P tellurium hexafluoride - P tert-octyl mercaptan - E titanium tetrachloride - E trifluoroacetyl chloride - P Legend: Filter Effective (E); Marginally (M); Poor (P). b. The listing of the TIC (Table E-1) represents a broad range of physical and chemical properties. Chemical families consist of halides (fluoride, chloride, bromide, and iodide), cyanides, cyanates, amines, oxides of carbon and nitrogen, ketones, aldehydes, esters, phosphates, thiols, and heavy metals (lead and titanium). This information shows that about 75 percent of the chemicals have a vapor pressure above 10 mm mercury, a state at which the strength of physical adsorption is reduced more rapidly on activated carbon and is a greater concern with respect to desorption. c. Table E-1 provides, in summary form, assessment results of the protection afforded by NBC filters to the selected TICs. The filter assessment indicated that many of the TIC could be effectively removed by the filter (effective), minimally removed by the filter (poor), or partially removed by the filter 31 December 2009 FM /MCWP /NTTP /AFTTP(I) , C1 E-3
4 (marginal). However, variables, such as being near the explosive and meteorological conditions, could affect the assessment. Several of the TICs were effectively removed by NBC filters; however, almost equal numbers were assessed as performing poorly (P) or marginally (M). d. Table E-1 provides data to only support unit planning. For example, this data could be used to support risk assessments based on IPB evaluations (e.g., types of TIC found in an AOI) furnished by the intelligence officer or the staff surgeon. However, military units (except for special-purpose units like EOD or HAZMAT response teams) lack the capability to detect most TIC; the unit response to TIC incidents/accidents remains as described in Chapter II. Note: This summary only addresses several of the TIC that represent an aerosol hazard and are produced in large quantities. There are many other TIC that present other hazards such as flammability and oxygen depletion, etc. Consult the technical references for specific information on TIC hazards, safety considerations, and other applicable emergency response guidelines. E-4 FM /MCWP /NTTP /AFTTP(I) , C1 31 December 2009
5 This page intentionally left blank.
6 This page intentionally left blank.
7
8 Marine Corps PCN: PIN:
Principal Investigator: Department: Laboratory room #s: Updated/revised: Chemical Name Amount Physical state Location Hazard Class
 Chemical Inventory Principal Investigator: Department: Laboratory room #s: Updated/revised: Chemical Name Amount Physical state Location Hazard Class 1 SUGGESTED SHELF STORAGE PATTERN - ORGANIC Organic
Chemical Inventory Principal Investigator: Department: Laboratory room #s: Updated/revised: Chemical Name Amount Physical state Location Hazard Class 1 SUGGESTED SHELF STORAGE PATTERN - ORGANIC Organic
Chemical Storage According to Compatibility
 Chemical Storage According to Compatibility To lessen risk of exposure to hazardous chemicals, all chemicals should be separated and stored according to hazard category and compatibility. *Storage Groups
Chemical Storage According to Compatibility To lessen risk of exposure to hazardous chemicals, all chemicals should be separated and stored according to hazard category and compatibility. *Storage Groups
CORROSIVES 1. Should be kept in acid resistant cabinets or on polyethylene trays. 2. Must never be stored on high shelves. 1
 Storing Chemicals (from Laboratory Safety Manual. June 1999) Proper chemical storage is extremely important in order to maximize personal safety with respect to chemical spills, chemical incompatibilities,
Storing Chemicals (from Laboratory Safety Manual. June 1999) Proper chemical storage is extremely important in order to maximize personal safety with respect to chemical spills, chemical incompatibilities,
Protection Level Afforded by the Canadian C7 Canister Against Single Large-Scale Release of Chlorine
 Of SR 68 UNC LA SSIF-1 DEFENCE RESEARCH ESTABLISHMENT SUFFIELD Protection Level Afforded by the Canadian C7 Canister Against Single Large-Scale Release of Chlorine By: S.H.C. Liang, E.C. Yee and S.J. Armour
Of SR 68 UNC LA SSIF-1 DEFENCE RESEARCH ESTABLISHMENT SUFFIELD Protection Level Afforded by the Canadian C7 Canister Against Single Large-Scale Release of Chlorine By: S.H.C. Liang, E.C. Yee and S.J. Armour
Safety Manual > Incompatible Chemicals Partial Listing
 Safety Manual > Incompatible Chemicals Partial Listing C. Incompatible Chemicals Partial Listing Chemical Incompatible Chemicals Acetic acid Chromic acid, nitric acid, permanganates, and peroxides Acetic
Safety Manual > Incompatible Chemicals Partial Listing C. Incompatible Chemicals Partial Listing Chemical Incompatible Chemicals Acetic acid Chromic acid, nitric acid, permanganates, and peroxides Acetic
INCOMPATIBILITY BETWEEN CHEMICALS
 INCOMPATIBILITY BETWEEN S CODE SHR 301 Date: April 2009 Revision: 02 Page: 1 of 5 INCOMPATIBILITY BETWEEN S Products that could react violently with each other should not be stored together, particularly
INCOMPATIBILITY BETWEEN S CODE SHR 301 Date: April 2009 Revision: 02 Page: 1 of 5 INCOMPATIBILITY BETWEEN S Products that could react violently with each other should not be stored together, particularly
Combustible Gas Catalytic Bead (0-100 %LEL) Part No FM Performance Certified 1,4 FM Performance Certified 1
 Sensor Data Sheet Document No. 365-2211-31 (Rev D) Combustible Gas Catalytic Bead (0-100 %LEL) Part No. 823-0211-31 FM Performance Certified 1,4 FM Performance Certified 1 Minimum Indicated Concentration...
Sensor Data Sheet Document No. 365-2211-31 (Rev D) Combustible Gas Catalytic Bead (0-100 %LEL) Part No. 823-0211-31 FM Performance Certified 1,4 FM Performance Certified 1 Minimum Indicated Concentration...
Glove Selection Chart
 Glove Selection Chart The following guide is a general guide for glove selection in relation to chemicals handled. The information presented here is believed to be accurate; however, we cannot guarantee
Glove Selection Chart The following guide is a general guide for glove selection in relation to chemicals handled. The information presented here is believed to be accurate; however, we cannot guarantee
Appendix 5: table of chemical compatibility for tubing
 Appendix 5: table of chemical compatibility for tubing KEY 1 Excellent 2 Good 3 Fair 4 Not recommended - no data Silicone PharMed Iso-Versinic PVC Acetaldehyde 3 4 4 2 Acetamide, 67% in w 1 2 4 4 Acetate
Appendix 5: table of chemical compatibility for tubing KEY 1 Excellent 2 Good 3 Fair 4 Not recommended - no data Silicone PharMed Iso-Versinic PVC Acetaldehyde 3 4 4 2 Acetamide, 67% in w 1 2 4 4 Acetate
Revised Permeation Values for DuPont Tychem SL
 Revised Permeation Values for DuPont Tychem SL Chemical Name Revised Permeation Values for DuPont Tychem SL (as of January, 2011) ACETIC ACID 64-19-7 >480 >480 ACETIC ANHYDRIDE 108-24-7 48 48 ACETONE 67-64-1
Revised Permeation Values for DuPont Tychem SL Chemical Name Revised Permeation Values for DuPont Tychem SL (as of January, 2011) ACETIC ACID 64-19-7 >480 >480 ACETIC ANHYDRIDE 108-24-7 48 48 ACETONE 67-64-1
APPENDIX C CHEMICALS USED IN CLANDESTINE DRUG LABS
 APPENDIX C CHEMICALS USED IN CLANDESTINE DRUG LABS You must exercise extreme caution when ope at the scene of an illegal drug lab. Do not walk into, touch, or move chemicals or spilled material. Avoid
APPENDIX C CHEMICALS USED IN CLANDESTINE DRUG LABS You must exercise extreme caution when ope at the scene of an illegal drug lab. Do not walk into, touch, or move chemicals or spilled material. Avoid
LABORATORY SAFETY PLAN APPENDIX
 LABORATORY SAFETY PLAN APPENDIX APPENDIX 1 REQUIRED CEE LAB ACCESS FORMS CEE Laboratory User Form As a laboratory user in the Department of Civil and Environmental Engineering at Northeastern University,
LABORATORY SAFETY PLAN APPENDIX APPENDIX 1 REQUIRED CEE LAB ACCESS FORMS CEE Laboratory User Form As a laboratory user in the Department of Civil and Environmental Engineering at Northeastern University,
Chemical Storage Guidelines
 Storage Guidelines Do not store excess chemical containers on the benchtops, designate a storage location and return containers to that location after each use. Do not store chemicals in the fume hood.
Storage Guidelines Do not store excess chemical containers on the benchtops, designate a storage location and return containers to that location after each use. Do not store chemicals in the fume hood.
INSTRUCTIONS ON EVERY AP EXAM:
 Most Common Reaction Types: 1. Acid-base neutralization (both weak & strong) 2. Nonmetal and metal oxides with water 3. Active metals with water 4. Single replacement redox 5. Double replacement precipitation
Most Common Reaction Types: 1. Acid-base neutralization (both weak & strong) 2. Nonmetal and metal oxides with water 3. Active metals with water 4. Single replacement redox 5. Double replacement precipitation
1. How many moles of hydrogen are needed to completely react with 2.00 moles of nitrogen?
 Stoichiometry Mole-to-Mole 1. How many moles of hydrogen are needed to completely react with 2.00 moles of nitrogen? N 2 + H 2 NH 3 2. If 5.50 moles of calcium carbide (CaC 2 ) reacts with an excess of
Stoichiometry Mole-to-Mole 1. How many moles of hydrogen are needed to completely react with 2.00 moles of nitrogen? N 2 + H 2 NH 3 2. If 5.50 moles of calcium carbide (CaC 2 ) reacts with an excess of
BGSU s Guide to Chemical Storage
 BGSU s Guide to Chemical Storage Environmental Health & Safety 372-2171 Chemical Storage Guidlines General Guidelines Store dry chemicals on shelves. Store flammables together. Use approved flammable storage
BGSU s Guide to Chemical Storage Environmental Health & Safety 372-2171 Chemical Storage Guidlines General Guidelines Store dry chemicals on shelves. Store flammables together. Use approved flammable storage
Write the name or formula for:
 Do Now Date: Tuesday, November 2, 2015 Objective: Name and write formulas for ionic and molecular (covalent) compounds. Write the name or formula for: K 2 SO 4 NaNO 3 Calcium Hydroxide Tuesday, November
Do Now Date: Tuesday, November 2, 2015 Objective: Name and write formulas for ionic and molecular (covalent) compounds. Write the name or formula for: K 2 SO 4 NaNO 3 Calcium Hydroxide Tuesday, November
Prioritization of the Percutaneous Hazard of Industrial Chemicals
 Naval Research Laboratory Washington, DC 20375-5320 NRL/FR/6364--11-10,213 Prioritization of the Percutaneous Hazard of Industrial Chemicals Thomas E. Sutto Materials and Sensors Branch Materials Science
Naval Research Laboratory Washington, DC 20375-5320 NRL/FR/6364--11-10,213 Prioritization of the Percutaneous Hazard of Industrial Chemicals Thomas E. Sutto Materials and Sensors Branch Materials Science
HAZARDOUS MATERIALS SURVEY. Note: List ALL virgin stock materials in current and projected inventories LAB NAME: PI: ADDRESS:
 HAZARDOUS MATERIALS SURVEY Note: List ALL virgin stock materials in current and projected inventories LAB NAME: PI: ADDRESS: Please read each definition of the following Hazardous Materials carefully and
HAZARDOUS MATERIALS SURVEY Note: List ALL virgin stock materials in current and projected inventories LAB NAME: PI: ADDRESS: Please read each definition of the following Hazardous Materials carefully and
Watering Systems. Water and Chemical Reference Guide. Instruction # IM /06
 Watering Systems. Water and Chemical Reference Guide Instruction # IM-030-06 07/06 Water quality Below are Lubing s recommendations for drinking water quality when using our watering systems: Description
Watering Systems. Water and Chemical Reference Guide Instruction # IM-030-06 07/06 Water quality Below are Lubing s recommendations for drinking water quality when using our watering systems: Description
4.2 Covalent Compounds: Formulas and Names LEARNING OBJECTIVES
 4.2 Covalent Compounds: Formulas and Names LEARNING OBJECTIVES 1. Determine the chemical formula of a simple covalent compound from its name. 2. Determine the name of a simple covalent compound from its
4.2 Covalent Compounds: Formulas and Names LEARNING OBJECTIVES 1. Determine the chemical formula of a simple covalent compound from its name. 2. Determine the name of a simple covalent compound from its
Naming Compounds. Part One: Naming Ionic Compounds. Identifying Ionic Compounds
 Naming Compounds Naming compounds is an important part of chemistry. Most compounds fall in to one of three categories ionic compounds, molecular compounds, or acids. Part One: Naming Ionic Compounds Identifying
Naming Compounds Naming compounds is an important part of chemistry. Most compounds fall in to one of three categories ionic compounds, molecular compounds, or acids. Part One: Naming Ionic Compounds Identifying
Environmental Health & Safety Policy Manual
 Environmental Health & Safety Policy Manual Issue Date: 5/24/2010 Updated: 10/1/2012 Policy # EHS - 200.07 Chemical Procurement, Inventory and Security Policy 1.0 PURPOSE To implement accountability and
Environmental Health & Safety Policy Manual Issue Date: 5/24/2010 Updated: 10/1/2012 Policy # EHS - 200.07 Chemical Procurement, Inventory and Security Policy 1.0 PURPOSE To implement accountability and
How Big Are Atoms? Five-hundred-million gold atoms lined up side-byside would form a line as long as a $10 bill.
 How Big Are Atoms? Five-hundred-million gold atoms lined up side-byside would form a line as long as a $10 bill. Look around your home and you ll be amazed at the variety of chemicals in your cupboards
How Big Are Atoms? Five-hundred-million gold atoms lined up side-byside would form a line as long as a $10 bill. Look around your home and you ll be amazed at the variety of chemicals in your cupboards
Passive Sampling Badges: Dosimeter (TWA) Tubes: 102 Solution Tubes: 103 Detector Tubes & Pumps:
 Passive Sampling Badges: 100-101 Dosimeter (TWA) Tubes: 102 Solution Tubes: 103 Detector Tubes & Pumps: 104-119 3M Passive Air Monitoring Badges 3M Air Monitors are simple and effective devices that collect
Passive Sampling Badges: 100-101 Dosimeter (TWA) Tubes: 102 Solution Tubes: 103 Detector Tubes & Pumps: 104-119 3M Passive Air Monitoring Badges 3M Air Monitors are simple and effective devices that collect
Office of Research Safety Laboratory Safety Manual 806 Chemical Storage and Handling
 Office of Research Safety Laboratory Safety Manual 806 Chemical Storage and Handling 1. Proper chemical storage and transport is extremely important in order to maximize personal safety with respect to
Office of Research Safety Laboratory Safety Manual 806 Chemical Storage and Handling 1. Proper chemical storage and transport is extremely important in order to maximize personal safety with respect to
Technical/Application Article 01 Version 1.2
 Technical/Application Article 01 Version 1.2 th 25 January 2017 WRH What is a PID? Introduction PID is the abbreviation for Photo-Ionization Detector. A PID is a hand-held, personal, or fixed wallmounted
Technical/Application Article 01 Version 1.2 th 25 January 2017 WRH What is a PID? Introduction PID is the abbreviation for Photo-Ionization Detector. A PID is a hand-held, personal, or fixed wallmounted
PRODUCT DATA SHEET PDS A88_E
 Plugs Suitable for A B C D Ref. Quantity Box/Bag (mm) (mm) (mm) (mm) TCP5 M12R + Pg7R M12R 4,5 8,5 10,8 4,5 3.000/100 TCP10 Pg9R Pg9R 6 12 12 4,5 2.000/100 TCP12 M12 + Pg7 M12 + Pg7 M16R + Pg11R M16R +
Plugs Suitable for A B C D Ref. Quantity Box/Bag (mm) (mm) (mm) (mm) TCP5 M12R + Pg7R M12R 4,5 8,5 10,8 4,5 3.000/100 TCP10 Pg9R Pg9R 6 12 12 4,5 2.000/100 TCP12 M12 + Pg7 M12 + Pg7 M16R + Pg11R M16R +
DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE NAME
 DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE NAME - The name of an ionic compound is made of the names of the CATION and ANION in the compound. - To get the FORMULA, you must figure out the SMALLEST
DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE NAME - The name of an ionic compound is made of the names of the CATION and ANION in the compound. - To get the FORMULA, you must figure out the SMALLEST
Chemistry Chapter 7 Test
 Chemistry Chapter 7 Test Multiple Choice Identify the choice that best completes the statement or answers the question. 1. A chemical formula includes the symbols of the elements in the compound and subscripts
Chemistry Chapter 7 Test Multiple Choice Identify the choice that best completes the statement or answers the question. 1. A chemical formula includes the symbols of the elements in the compound and subscripts
Field Behavior of NBC Agents
 /AFM 105-7 FMFM 7-11H Field Behavior of NBC Agents Pub Date 11-86 PCN 139 711800 00 FMFM 7-11-H AFM 105-7 3 NOVEMBER 1986 By Order of the Secretaries of the Army and Air Force: Official: R. L. DILWORTH
/AFM 105-7 FMFM 7-11H Field Behavior of NBC Agents Pub Date 11-86 PCN 139 711800 00 FMFM 7-11-H AFM 105-7 3 NOVEMBER 1986 By Order of the Secretaries of the Army and Air Force: Official: R. L. DILWORTH
Formula of a Compound
 Name Formula of a Compound 1. Useful only if it correctly represents the substance. 2. The composition is determined in chemical analysis. 3. The formula then is derived by atomic theory and chemical bonding
Name Formula of a Compound 1. Useful only if it correctly represents the substance. 2. The composition is determined in chemical analysis. 3. The formula then is derived by atomic theory and chemical bonding
Chapter Six Chemical Names and Formulas WS C U1C6
 Chapter Six Chemical Names and Formulas WS C U1C6 Name Period Section 6.1 Part I: Matching. Match the definition with the term that best correlates to it. No definition will be used more than once. 1.
Chapter Six Chemical Names and Formulas WS C U1C6 Name Period Section 6.1 Part I: Matching. Match the definition with the term that best correlates to it. No definition will be used more than once. 1.
PREVENTING HAZARDOUS WASTE FINES
 University of Medicine and Dentistry of New Jersey EOHSS FACTSHEET PREVENTING HAZARDOUS WASTE FINES In the last few years institutions such as MIT, University of New Hampshire, Yale, Stanford, and Boston
University of Medicine and Dentistry of New Jersey EOHSS FACTSHEET PREVENTING HAZARDOUS WASTE FINES In the last few years institutions such as MIT, University of New Hampshire, Yale, Stanford, and Boston
Available chemicals from the catalog (the starting sources of carbon compounds will continually decrease as we learn new reactions.
 ucleophilic ubstitution & Elimination Chemistry Beauchamp 1 Available chemicals from the catalog (the starting sources of carbon compounds will continually decrease as we learn new reactions. ources of
ucleophilic ubstitution & Elimination Chemistry Beauchamp 1 Available chemicals from the catalog (the starting sources of carbon compounds will continually decrease as we learn new reactions. ources of
Chemical Safety. Peter Yeung D.G. Manager. Safety Office The University of Hong Kong
 Chemical Safety Peter Yeung D.G. Manager Safety Office The University of Hong Kong Lesson Learned 1. Laboratory fire 2. Chemical spill 3. Incompatibles in waste disposal Lab. Fire Sodium/Solvent Chemical
Chemical Safety Peter Yeung D.G. Manager Safety Office The University of Hong Kong Lesson Learned 1. Laboratory fire 2. Chemical spill 3. Incompatibles in waste disposal Lab. Fire Sodium/Solvent Chemical
Appendix D. Comparison of OSHA and EPA Lists of Highly Hazardous Chemicals and Regulated Substances Acetaldehyde. Acrolein [2-Propenal]
![Appendix D. Comparison of OSHA and EPA Lists of Highly Hazardous Chemicals and Regulated Substances Acetaldehyde. Acrolein [2-Propenal] Appendix D. Comparison of OSHA and EPA Lists of Highly Hazardous Chemicals and Regulated Substances Acetaldehyde. Acrolein [2-Propenal]](/thumbs/90/103489389.jpg) Appendix D Comparison of OSHA and EPA Lists of Highly Hazardous Chemicals and egulated Substances 75-07-0 Acetaldehyde 107-02-8 Acrolein [2-Propenal], 107-13-1 Acrylonitrile [ 2 -Propenenitrile ] 814-68-6
Appendix D Comparison of OSHA and EPA Lists of Highly Hazardous Chemicals and egulated Substances 75-07-0 Acetaldehyde 107-02-8 Acrolein [2-Propenal], 107-13-1 Acrylonitrile [ 2 -Propenenitrile ] 814-68-6
Nomenclature. Common Names. Common Names COMPOUNDS FORMED FROM IONS. Binary Ionic Compounds
 PO 4 3- phosphate ion C 2 H 3 O 2 - acetate ion World of Chemistry: Chapter 4 Nomenclature HC 2 H 3 O 2 Acetic Acid Common Names Common Names A lot of chemicals have common lot of chemicals have common
PO 4 3- phosphate ion C 2 H 3 O 2 - acetate ion World of Chemistry: Chapter 4 Nomenclature HC 2 H 3 O 2 Acetic Acid Common Names Common Names A lot of chemicals have common lot of chemicals have common
Nomenclature Naming Ionic Compounds Worksheet #1
 Naming Ionic Compounds Worksheet #1 In forming ionic compounds with non-metals, the transition metals often exhibit more than one valence. For example, in the reaction between iron and chlorine, two products
Naming Ionic Compounds Worksheet #1 In forming ionic compounds with non-metals, the transition metals often exhibit more than one valence. For example, in the reaction between iron and chlorine, two products
WESTERN CONNECTICUT STATE UNIVERSITY CHEMICAL STORAGE AND COMPATIBILITY GUIDELINES PROCEDURE S-118
 WESTERN CONNECTICUT STATE UNIVERSITY CHEMICAL STORAGE AND COMPATIBILITY GUIDELINES PROCEDURE S-118 Issued 6/4/02 Please direct any questions or comments about the applicability of this document to Luigi
WESTERN CONNECTICUT STATE UNIVERSITY CHEMICAL STORAGE AND COMPATIBILITY GUIDELINES PROCEDURE S-118 Issued 6/4/02 Please direct any questions or comments about the applicability of this document to Luigi
500 SERIES CHEMICAL RESISTANCE GUIDE
 500 SERIES CHEMICAL RESISTANCE GUIDE INTRODUCTION The Stonchem 500 Series Chemical Resistance Guide is designed to aid in the proper selection of material for every job application. Exposure to over 250
500 SERIES CHEMICAL RESISTANCE GUIDE INTRODUCTION The Stonchem 500 Series Chemical Resistance Guide is designed to aid in the proper selection of material for every job application. Exposure to over 250
Chemical Polyurethane Polyethelyne PVC Nylon 11 Kynar
 ACETIC ACID, GLACIAL 4 2 4-1 ACETIC ACID, 30% 4 1 4 2 1 ACETONE 4 2 4 1 4 ACETYLENE 1 4 1 1 1 ALKAZENE 4 - - - - ALUMINUM CHLORIDE (aq) 3 2 1-1 ALUMINUM NITRATE (aq) 3-2 - 1 AMMONIA ANGYDROUS 4 2 1-4 AMMONIA
ACETIC ACID, GLACIAL 4 2 4-1 ACETIC ACID, 30% 4 1 4 2 1 ACETONE 4 2 4 1 4 ACETYLENE 1 4 1 1 1 ALKAZENE 4 - - - - ALUMINUM CHLORIDE (aq) 3 2 1-1 ALUMINUM NITRATE (aq) 3-2 - 1 AMMONIA ANGYDROUS 4 2 1-4 AMMONIA
Assessing Unknown Odors and Chemicals
 Treat yourself to better rental service. Thank You for Attending Today s Webinar: Assessing Unknown Odors and Chemicals Your Host Matt DeLacluyse Operations Mgr RAECO Rents mattd@raecorents.com Featured
Treat yourself to better rental service. Thank You for Attending Today s Webinar: Assessing Unknown Odors and Chemicals Your Host Matt DeLacluyse Operations Mgr RAECO Rents mattd@raecorents.com Featured
Summer Assignment Part 2
 Summer Assignment Part 2 Name: 1. Metric Conversions. Remember 1 cm 3 = 1 ml 1 L = 1 dm 3 ITEM GIVEN METRIC UNIT DESIRED METRIC UNIT A 8.43 cm mm B 2.41 x 10 2 cm m C 294.5 nm cm D 1.445 x 10 4 m km E
Summer Assignment Part 2 Name: 1. Metric Conversions. Remember 1 cm 3 = 1 ml 1 L = 1 dm 3 ITEM GIVEN METRIC UNIT DESIRED METRIC UNIT A 8.43 cm mm B 2.41 x 10 2 cm m C 294.5 nm cm D 1.445 x 10 4 m km E
(U) Indicators for Terrorist Use of Toxic Industrial Chemicals
 FEDERAL BUREAU OF INVESTIGATION INTELLIGENCE BULLETIN Weapons of Mass Destruction Directorate 22 March 2007 (U) Indicators for Terrorist Use of Toxic Industrial Chemicals (U) THIS INTELLIGENCE BULLETIN
FEDERAL BUREAU OF INVESTIGATION INTELLIGENCE BULLETIN Weapons of Mass Destruction Directorate 22 March 2007 (U) Indicators for Terrorist Use of Toxic Industrial Chemicals (U) THIS INTELLIGENCE BULLETIN
Nomenclature. Naming Compounds
 Nomenclature Naming Compounds Ionic Compounds Metal bonding with non-metal One atom gains electrons, one atom loses electrons Exist as ions with full highest energy levels. Are held together in a giant
Nomenclature Naming Compounds Ionic Compounds Metal bonding with non-metal One atom gains electrons, one atom loses electrons Exist as ions with full highest energy levels. Are held together in a giant
PRODUCT DATA SHEET PDS A112_E. Metric thread M 1.5 pitch CEI EN CEI EN 50262
 reduced cable entry Metric thread M 1.5 pitch CEI EN 60423 CEI EN 50262 Ref. P Fixing A B C L Light Hole min-max Spanner min-max Quantity Grey (mm) (mm) (mm) (mm) (mm) 1910S.M16N M16x1,5 16,5 4-7,5 19
reduced cable entry Metric thread M 1.5 pitch CEI EN 60423 CEI EN 50262 Ref. P Fixing A B C L Light Hole min-max Spanner min-max Quantity Grey (mm) (mm) (mm) (mm) (mm) 1910S.M16N M16x1,5 16,5 4-7,5 19
NAMING IONIC COMPOUNDS
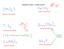 NAMING IONIC COMPOUNDS iron(ii) carbonate ammonium sulfide titanium (IV) sulfide barium phosphate Spelling matters! calcium nitrate sodium phosphide DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE
NAMING IONIC COMPOUNDS iron(ii) carbonate ammonium sulfide titanium (IV) sulfide barium phosphate Spelling matters! calcium nitrate sodium phosphide DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE
HSW NORM-JECT SYRINGES CHEMICAL RESISTANCE CHART
 A Beer + + Acetic acid (10-60%) + O Benzaldehyde + O Acetic acid (1-10%) + + Benzene O - Acetic acid (80-100%) + O Benzene sulfonic acid + + Acetic anhydride + + Benzoic acid + Acetone + + Bismuth carbonate
A Beer + + Acetic acid (10-60%) + O Benzaldehyde + O Acetic acid (1-10%) + + Benzene O - Acetic acid (80-100%) + O Benzene sulfonic acid + + Acetic anhydride + + Benzoic acid + Acetone + + Bismuth carbonate
Naming Chemical Compounds
 Naming Chemical Compounds Naming compounds is an important part of chemistry. Most compounds fall into one of four categories Ionic Compounds, Molecular Compounds, Acids and Bases, and Hydrates Part One:
Naming Chemical Compounds Naming compounds is an important part of chemistry. Most compounds fall into one of four categories Ionic Compounds, Molecular Compounds, Acids and Bases, and Hydrates Part One:
Chemical Resistance Properties of Anko Tubing
 Chemical Resistance Properties of Anko Tubing E = Excellent G = Good F = Fair X = Not Recommended U = Untested Environment, % Conc.* w = Water alc = Alcohol * If concentration is not indicated, assume
Chemical Resistance Properties of Anko Tubing E = Excellent G = Good F = Fair X = Not Recommended U = Untested Environment, % Conc.* w = Water alc = Alcohol * If concentration is not indicated, assume
Notes: Molar Mass, Percent Composition, Mole Calculations, and Empirical/Molecular Formulas
 Notes: Molar Mass, Percent Composition, Mole Calculations, and Empirical/Molecular Formulas In Chemistry, a Mole is: the unit that measures the amount of a substance - equals 6.022 x 10 23 particles of
Notes: Molar Mass, Percent Composition, Mole Calculations, and Empirical/Molecular Formulas In Chemistry, a Mole is: the unit that measures the amount of a substance - equals 6.022 x 10 23 particles of
National Institute of Justice
 U.S. Department of Justice Office of Justice Programs National Institute of Justice National Institute of Justice Law Enforcement and Corrections Standards and Testing Program Guide for the Selection of
U.S. Department of Justice Office of Justice Programs National Institute of Justice National Institute of Justice Law Enforcement and Corrections Standards and Testing Program Guide for the Selection of
Compound Names and Formulas Activity
 Name: KEY Class Period: Compound Names and Formulas Activity Part 1 Instructions: Study the following compound formulas and their corresponding names. Then answer the questions below. Questions: 1. What
Name: KEY Class Period: Compound Names and Formulas Activity Part 1 Instructions: Study the following compound formulas and their corresponding names. Then answer the questions below. Questions: 1. What
CHEMICAL RESISTANCE DATA FOR POLYETHYLENES
 CHEMICAL RESISTANCE DATA FOR POLYETHYLENES [ R = Resistant LR= Low Resistance NR= Not Resistant] 23 o C 60 o C 23 o C 60 o C Acetaldehyde(100%) LR NR LR NR Acetic acid (10%) R R R R Acetic acid (50%) R
CHEMICAL RESISTANCE DATA FOR POLYETHYLENES [ R = Resistant LR= Low Resistance NR= Not Resistant] 23 o C 60 o C 23 o C 60 o C Acetaldehyde(100%) LR NR LR NR Acetic acid (10%) R R R R Acetic acid (50%) R
AP Chemistry Summer Assignment
 AP Chemistry Summer Assignment Due Date: Thursday, September 1 st, 2011 Directions: Show all of your work for full credit. Include units and labels. Record answers to the correct number of significant
AP Chemistry Summer Assignment Due Date: Thursday, September 1 st, 2011 Directions: Show all of your work for full credit. Include units and labels. Record answers to the correct number of significant
Forming Compounds - Homework Package SNC2D1 Grade 10 Academic Science
 Forming Compounds - Homework Package SNC2D1 Grade 10 Academic Science Name: Date: 1. The Formation of Ions complete the following table. Group Example & Bohr-Rutherford diagram # of valence electrons Lose
Forming Compounds - Homework Package SNC2D1 Grade 10 Academic Science Name: Date: 1. The Formation of Ions complete the following table. Group Example & Bohr-Rutherford diagram # of valence electrons Lose
actual yield (p. 372) excess reagent (p. 369) mole-mole relationship for ag bw: x mol G b mol W a mol G xb a mol W Organizing Information
 12 Study Guide 12 Study Guide Study Tip Prioritize Schedule your time realistically. Stick to your deadlines. If your class subscribes to the Interactive Textbook with ChemASAP, your students can go online
12 Study Guide 12 Study Guide Study Tip Prioritize Schedule your time realistically. Stick to your deadlines. If your class subscribes to the Interactive Textbook with ChemASAP, your students can go online
Immaculate Conception High School Summer Assignment Chemistry Honors. * Ms. Lusamba if you have any questions:
 Immaculate Conception High School Summer Assignment Chemistry Honors * Email Ms. Lusamba if you have any questions: plusamba@ichspride.org Directions : This assignment consists of four sections, each of
Immaculate Conception High School Summer Assignment Chemistry Honors * Email Ms. Lusamba if you have any questions: plusamba@ichspride.org Directions : This assignment consists of four sections, each of
Pellistor Relative Responses
 Pellistor Relative Responses PELLISTOR RELATIVE RESPONSES SGX Sensortech are often asked for relative response figures when using the company s range of pellistors for non-straight-chain hydrocarbons and
Pellistor Relative Responses PELLISTOR RELATIVE RESPONSES SGX Sensortech are often asked for relative response figures when using the company s range of pellistors for non-straight-chain hydrocarbons and
Nomenclature. Ex. For sodium the oxidation number is +1. For oxygen the oxidation number is -2.
 Nomenclature 1. BONDING CAPACITY (VALENCE) The number of bonds an atom can make. For a Cation, the bonding capacity is the number of electrons lost to become stable. For an Anion, the bonding capacity
Nomenclature 1. BONDING CAPACITY (VALENCE) The number of bonds an atom can make. For a Cation, the bonding capacity is the number of electrons lost to become stable. For an Anion, the bonding capacity
Training on identification of hazardous activities March 2010, Skopje Annex I of the Convention and Annex I of the SEVESO II Directive
 Training on identification of hazardous activities 23-24 March 2010, Skopje Annex I of the Convention and Annex I of the SEVESO II Directive Suzana Milutinović Ministry of Environment and Spatial Planning
Training on identification of hazardous activities 23-24 March 2010, Skopje Annex I of the Convention and Annex I of the SEVESO II Directive Suzana Milutinović Ministry of Environment and Spatial Planning
Dräger simultaneous test sets Dräger-Tubes
 Dräger simultaneous test sets Dräger-Tubes Risks and potential dangers at a glance: The SimultanTest Sets allow the measurement of five different gases at the same time. ST-1996-2005 02 Dräger simultaneous
Dräger simultaneous test sets Dräger-Tubes Risks and potential dangers at a glance: The SimultanTest Sets allow the measurement of five different gases at the same time. ST-1996-2005 02 Dräger simultaneous
Binary Molecular Compounds
 Binary Molecular Compounds What guidelines are used to write the name and formula of a binary molecular compound? Recall that binary ionic compounds are composed of the ions of two elements, a metal and
Binary Molecular Compounds What guidelines are used to write the name and formula of a binary molecular compound? Recall that binary ionic compounds are composed of the ions of two elements, a metal and
3 Chemical Equilibrium
 Aubrey High School AP Chemistry 3 Chemical Equilibrium Name Period Date / / 3.1 Problems Chemical Analysis 1. Write the equilibrium constant expressions for the following reactions. How are they related
Aubrey High School AP Chemistry 3 Chemical Equilibrium Name Period Date / / 3.1 Problems Chemical Analysis 1. Write the equilibrium constant expressions for the following reactions. How are they related
Naming Compounds Quiz A
 Naming Compounds Quiz A 1. How can you quickly tell if a compound is molecular or ionic? Give an example of each kind. 2. Name these compounds: NaBr, MgS, AlN, Na 2 S, Mg 3 P 2, AlF 3. 3. Name these compounds:
Naming Compounds Quiz A 1. How can you quickly tell if a compound is molecular or ionic? Give an example of each kind. 2. Name these compounds: NaBr, MgS, AlN, Na 2 S, Mg 3 P 2, AlF 3. 3. Name these compounds:
NAMING IONIC COMPOUNDS. ammonium sulfide. iron(ii) carbonate. barium phosphate. titanium(iv) sulfide. Spelling matters!
 67 NAMING IONIC COMPOUNDS ammonium sulfide iron(ii) carbonate titanium(iv) sulfide barium phosphate Spelling matters! barium phosphide 68 DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE NAME - The
67 NAMING IONIC COMPOUNDS ammonium sulfide iron(ii) carbonate titanium(iv) sulfide barium phosphate Spelling matters! barium phosphide 68 DETERMINING THE FORMULA OF AN IONIC COMPOUND FROM THE NAME - The
Safe Installation and Use of the NanoRam Immersion Probe
 Safe Installation and Use of the NanoRam Immersion Probe Description: NanoRam Immersion Probe - 12 inch length; Immersion Probe Disposable Protective Sleeve Model Number: NR2-IMP; NR2-IMPS Part Number:
Safe Installation and Use of the NanoRam Immersion Probe Description: NanoRam Immersion Probe - 12 inch length; Immersion Probe Disposable Protective Sleeve Model Number: NR2-IMP; NR2-IMPS Part Number:
The ions/polyatomic ions, solubility rules, and activity series will need to be memorized.
 AP Chemistry Summer Assignment 2012 The summer assignment is designed to help you practice: - writing chemical formulas, names, and chemical equations - reaction types - predicting reaction products -
AP Chemistry Summer Assignment 2012 The summer assignment is designed to help you practice: - writing chemical formulas, names, and chemical equations - reaction types - predicting reaction products -
CfE Higher Chemistry. Unit 1: Chemical Changes and Structure. Intermolecular forces
 CfE Higher Chemistry Unit 1: Chemical Changes and Structure Intermolecular forces 05/09/2017 Van der Waal s Forces and London Dispersion Forces 05/09/2017 Learning Outcomes : I can explain the difference
CfE Higher Chemistry Unit 1: Chemical Changes and Structure Intermolecular forces 05/09/2017 Van der Waal s Forces and London Dispersion Forces 05/09/2017 Learning Outcomes : I can explain the difference
Chemical Storage Guidelines Page 1 of 10
 Guidelines Page 1 of 10 1.0 PURPOSE: Proper storage is needed to minimize the hazards associated with accidentally mixing incompatible chemicals. Due to the diverse individual properties of chemicals that
Guidelines Page 1 of 10 1.0 PURPOSE: Proper storage is needed to minimize the hazards associated with accidentally mixing incompatible chemicals. Due to the diverse individual properties of chemicals that
Chemical Permeation Guide
 Chemical Permeation Guide What is permeation? Guidance on Chemical Permeation and Penetration Permeation is the process by which a potentially hazardous chemical moves through a material on a molecular
Chemical Permeation Guide What is permeation? Guidance on Chemical Permeation and Penetration Permeation is the process by which a potentially hazardous chemical moves through a material on a molecular
WRITING FORMULAS: MOLAR MASS, %COMPOSITION, EMPIRICAL AND MOLECULAR FORMULAS
 WRITING FORMULAS: MOLAR MASS, %COMPOSITION, EMPIRICAL AND MOLECULAR FORMULAS REVIEW: Polyatomic ions, writing names from formulas, oxidation number rules I. WRITING FORMULAS FROM NAMES: A. Rules: 1. Know
WRITING FORMULAS: MOLAR MASS, %COMPOSITION, EMPIRICAL AND MOLECULAR FORMULAS REVIEW: Polyatomic ions, writing names from formulas, oxidation number rules I. WRITING FORMULAS FROM NAMES: A. Rules: 1. Know
Philippine Regulations on Chemicals The Global Summit on Chemical Safety and Security April 2016, Kielce, Poland
 Philippine Regulations on Chemicals The Global Summit on Chemical Safety and Security 18-20 April 2016, Kielce, Poland Health ENGR. JEAN C. BORROMEO Supervising Environmental Management Specialist DENR
Philippine Regulations on Chemicals The Global Summit on Chemical Safety and Security 18-20 April 2016, Kielce, Poland Health ENGR. JEAN C. BORROMEO Supervising Environmental Management Specialist DENR
Carbon Compounds. Chemical Bonding Part 2
 Carbon Compounds Chemical Bonding Part 2 Introduction to Functional Groups: Alkanes! Alkanes Compounds that contain only carbons and hydrogens, with no double or triple bonds.! Alkyl Groups A part of a
Carbon Compounds Chemical Bonding Part 2 Introduction to Functional Groups: Alkanes! Alkanes Compounds that contain only carbons and hydrogens, with no double or triple bonds.! Alkyl Groups A part of a
Chemical Nomenclature
 Chemical Nomenclature! The first names for chemicals were common names: Sugar, quicklime, Epsom salts, milk of magnesia, gypsom, laughing gas Simple, but not practical, the tell us little about the chemicals
Chemical Nomenclature! The first names for chemicals were common names: Sugar, quicklime, Epsom salts, milk of magnesia, gypsom, laughing gas Simple, but not practical, the tell us little about the chemicals
Chapter 12. Answers to Questions. 1. (a) nitrogen dioxide (b) hydrogen sulfide (c) hydrogen chloride
 Chapter 12 Answers to Questions 1. (a) nitrogen dioxide (b) hydrogen sulfide (c) hydrogen chloride 2. (a) hydrogen cyanide (b) dinitrogen oxide (c) ammonia 3. X = sulfur, Y = oxygen, Z = sulfur dioxide
Chapter 12 Answers to Questions 1. (a) nitrogen dioxide (b) hydrogen sulfide (c) hydrogen chloride 2. (a) hydrogen cyanide (b) dinitrogen oxide (c) ammonia 3. X = sulfur, Y = oxygen, Z = sulfur dioxide
- Some MOLECULES can gain or lose electrons to form CATIONS or ANIONS. These are called POLYATOMIC IONS
 63 POLYATOMIC IONS - Some MOLECULES can gain or lose electrons to form CATIONS or ANIONS. These are called POLYATOMIC IONS - Polyatomic ions form ionic compounds in the same way that single-element ions
63 POLYATOMIC IONS - Some MOLECULES can gain or lose electrons to form CATIONS or ANIONS. These are called POLYATOMIC IONS - Polyatomic ions form ionic compounds in the same way that single-element ions
Teaching innovatively (with focus on ICT) and its impact on the quality of education LESSON PLAN CHEMISTRY
 Teaching innovatively (with focus on ICT) and its impact on the quality of education LESSON PLAN CHEMISTRY Teacher: Fazilet TURNA Level: 9th Grade Unit: Substance and Its Features Topic: Naming Compounds
Teaching innovatively (with focus on ICT) and its impact on the quality of education LESSON PLAN CHEMISTRY Teacher: Fazilet TURNA Level: 9th Grade Unit: Substance and Its Features Topic: Naming Compounds
ACETONE. PRODUCT IDENTIFICATION CAS NO EINECS NO MOL WT H.S. CODE Oral rat LD50: 5800 mg/kg
 ACETONE www.pawarchemicals.com PRODUCT IDENTIFICATION CAS NO 67-64-1 EINECS NO. 200-662-2 FORMULA (CH3)2C=O MOL WT. 58.08 H.S. CODE 2914.11 TOXICITY SYNONYMS Oral rat LD50: 5800 mg/kg Dimethyl ketone;
ACETONE www.pawarchemicals.com PRODUCT IDENTIFICATION CAS NO 67-64-1 EINECS NO. 200-662-2 FORMULA (CH3)2C=O MOL WT. 58.08 H.S. CODE 2914.11 TOXICITY SYNONYMS Oral rat LD50: 5800 mg/kg Dimethyl ketone;
Unit 4. Inorganic formulation
 Index Unit 4. Inorganic formulation 1. STICHIMETRIC NAMES F BINARY CMPUNDS...2 1.1.. With multiplying prefix...2 1.2.- With oxidation numbers...3 1.3.- With charge number...5 2. Special names...6 Chemistry
Index Unit 4. Inorganic formulation 1. STICHIMETRIC NAMES F BINARY CMPUNDS...2 1.1.. With multiplying prefix...2 1.2.- With oxidation numbers...3 1.3.- With charge number...5 2. Special names...6 Chemistry
Bohr Diagrams/Lewis Diagrams. Naming Ionic Compounds
 Bohr Diagrams/Lewis Diagrams Which subatomic particles are located in the nucleus? Protons and neutrons What is the difference between a Bohr Diagram and a Lewis Diagram? Lewis diagram shows only valence
Bohr Diagrams/Lewis Diagrams Which subatomic particles are located in the nucleus? Protons and neutrons What is the difference between a Bohr Diagram and a Lewis Diagram? Lewis diagram shows only valence
Name: Class: Date: 10. How many neutrons does europium-151 have? What is the isotope s mass number? Number of neutrons = mass number atomic number
 Name: Class: Date: Unit Review Sheet Short Answer Give the number of protons, electrons, and neutrons in each of the following atoms. Atomic number = number of protons = number of electrons Number of neutrons
Name: Class: Date: Unit Review Sheet Short Answer Give the number of protons, electrons, and neutrons in each of the following atoms. Atomic number = number of protons = number of electrons Number of neutrons
Chemical Resistance of PVC Products
 The mechanism of chemical attack on thermoplastics in general, and PVC sheets in particular, differs significantly from the mechanism of corrosion of metals. Corrosion of metals results in a gradual loss
The mechanism of chemical attack on thermoplastics in general, and PVC sheets in particular, differs significantly from the mechanism of corrosion of metals. Corrosion of metals results in a gradual loss
Chapter 2 Problems.notebook. September 28, Sep 24 7:34 PM. Sep 24 7:33 PM. Sep 24 7:35 PM. Sep 24 7:35 PM. Sep 24 7:36 PM.
 1. The radius of a Cl nucleus is 4.0 fm (femtometer), and the radius of a Cl atom is 100 pm (picometer). (1 fm = 1 X 10 15m; 1 pm = 1 X 10 12m). c) How many times larger is the volume of the atom than
1. The radius of a Cl nucleus is 4.0 fm (femtometer), and the radius of a Cl atom is 100 pm (picometer). (1 fm = 1 X 10 15m; 1 pm = 1 X 10 12m). c) How many times larger is the volume of the atom than
Figure 1. I - Mg 2+ HS Chemistry POGIL Activity. metal with a non-metal with a with a
 HS Chemistry PGIL Activity Topic: Naming & Formula Writing 1(BW) Why? In this activity we will address the question: How do the smallest particles of matter connect to each other and how do we represent
HS Chemistry PGIL Activity Topic: Naming & Formula Writing 1(BW) Why? In this activity we will address the question: How do the smallest particles of matter connect to each other and how do we represent
This exam will be given over 2 days. Part 1: Objectives 1-13 Part 2: Objectives 14-24
 Name Hour January Exam Practice A This exam will be given over 2 days. Part 1: Objectives 1-13 Part 2: Objectives 14-24 This practice exam will be graded in the exam portion of the grade book. Objectives
Name Hour January Exam Practice A This exam will be given over 2 days. Part 1: Objectives 1-13 Part 2: Objectives 14-24 This practice exam will be graded in the exam portion of the grade book. Objectives
Naming Compounds Tutorial and Worksheet
 Naming Compounds Tutorial and Worksheet Since we use different methods in naming binary covalent (molecular) compounds and ionic compounds, the first step in naming or writing the formula of a compound
Naming Compounds Tutorial and Worksheet Since we use different methods in naming binary covalent (molecular) compounds and ionic compounds, the first step in naming or writing the formula of a compound
DETECTOR TUBES AND PUMPS
 DETECTOR TUBES AND PUMPS 08-00-02 FEATURES Quick and inexpensive to use. A reliable method of testing more than 170 hazardous gases and vapors. Kwik-Draw Deluxe and Gas Tester II pumps offer automatic
DETECTOR TUBES AND PUMPS 08-00-02 FEATURES Quick and inexpensive to use. A reliable method of testing more than 170 hazardous gases and vapors. Kwik-Draw Deluxe and Gas Tester II pumps offer automatic
Understanding the Threat from Common Toxic Chemicals. George R. Famini, PhD Director Chemical Security Analysis Center
 Understanding the Threat from Common Toxic Chemicals George R. Famini, PhD Director Chemical Security Analysis Center CSAC is a Force Multiplier for the Homeland Security Enterprise Core Capability Generate
Understanding the Threat from Common Toxic Chemicals George R. Famini, PhD Director Chemical Security Analysis Center CSAC is a Force Multiplier for the Homeland Security Enterprise Core Capability Generate
Honors Chemistry Review for Semester One Final Exam
 Honors Chemistry Review for Semester One Final Exam Chapter 1 What is chemistry? Branches of chemistry Scientific method 1. What is chemistry? 2. Which measurement depends on gravitational force mass or
Honors Chemistry Review for Semester One Final Exam Chapter 1 What is chemistry? Branches of chemistry Scientific method 1. What is chemistry? 2. Which measurement depends on gravitational force mass or
CHAPTER 7 CHEMICAL FORMULAS AND BONDING
 Name Date Period CHAPTER 7 CHEMICAL FORMULAS AND BONDING IONIC COMPOUNDS 1. What are the two types of bonds that will be discussed in this chapter? 2. How do ionic bonds form? 3. What do you call the positive
Name Date Period CHAPTER 7 CHEMICAL FORMULAS AND BONDING IONIC COMPOUNDS 1. What are the two types of bonds that will be discussed in this chapter? 2. How do ionic bonds form? 3. What do you call the positive
Chemistry in Action. Gr. 10 Work Booklet. Name:
 Chemistry in Action Gr. 10 Work Booklet Name: Determining the Number of Subatomic Particles Element name Chemical symbol # protons # neutrons # electrons atomic number atomic mass 7 7 5 6 5 1 0 1 Ca 9
Chemistry in Action Gr. 10 Work Booklet Name: Determining the Number of Subatomic Particles Element name Chemical symbol # protons # neutrons # electrons atomic number atomic mass 7 7 5 6 5 1 0 1 Ca 9
Multiple Choice Identify the choice that best completes the statement or answers the question.
 Covalent Practice CP November 2015 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Bromine, atomic number 35, belongs to Group 17. How many electrons does
Covalent Practice CP November 2015 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Bromine, atomic number 35, belongs to Group 17. How many electrons does
CHEMICAL RESISTANCE LIST
 CHEMICAL RESISTANCE LIST The information provided is designed to be used as a guide and all information must be checked on a project basis. Data based upon room temperatures. Elevated temperatures will
CHEMICAL RESISTANCE LIST The information provided is designed to be used as a guide and all information must be checked on a project basis. Data based upon room temperatures. Elevated temperatures will
2681 Parleys Way. Suite 201. Salt Lake City, UT AccuSense FAQ. May 2010
 2681 Parleys Way Suite 201 Salt Lake City, UT 84109 801 746 7888 www.seertechnology.com AccuSense FAQ May 2010 AccuSense Frequently Asked Questions (FAQs) 1. What is AccuSense? AccuSense is a laboratory
2681 Parleys Way Suite 201 Salt Lake City, UT 84109 801 746 7888 www.seertechnology.com AccuSense FAQ May 2010 AccuSense Frequently Asked Questions (FAQs) 1. What is AccuSense? AccuSense is a laboratory
Synthesis of Nitriles a. dehydration of 1 amides using POCl 3 : b. SN2 reaction of cyanide ion on halides:
 I. Nitriles Nitriles consist of the CN functional group, and are linear with sp hybridization on C and N. Nitriles are non-basic at nitrogen, since the lone pair exists in an sp orbital (50% s character
I. Nitriles Nitriles consist of the CN functional group, and are linear with sp hybridization on C and N. Nitriles are non-basic at nitrogen, since the lone pair exists in an sp orbital (50% s character
CHEMISTRY 135 REVISION OF NAMES, FORMULAE AND EQUATIONS
 CHEMISTRY 135 REVISION OF NAMES, FORMULAE AND EQUATIONS Answer the following questions as if you were going to hand them in for marking. You will be tested on them in class time. 1) Give the correct chemical
CHEMISTRY 135 REVISION OF NAMES, FORMULAE AND EQUATIONS Answer the following questions as if you were going to hand them in for marking. You will be tested on them in class time. 1) Give the correct chemical
Fluid Compatibility Table
 Acetaldehyde C A A A B D C A A Acetate Solvents A B A A D D A A Acetic Acid, crude C B A A D D A A Acetic Acid, pure D B A A D D A A Acetic Acid, 10% C B A A B D D A A Acetic Acid, 80% C B A A C D D A
Acetaldehyde C A A A B D C A A Acetate Solvents A B A A D D A A Acetic Acid, crude C B A A D D A A Acetic Acid, pure D B A A D D A A Acetic Acid, 10% C B A A B D D A A Acetic Acid, 80% C B A A C D D A
SNC2D CHEMISTRY 2/18/2013. CHEMICAL REACTIONS L Molecular Compounds (P ) Molecular Compounds. Molecular Compounds
 SNC2D CHEMISTRY CHEMICAL REACTIONS L (P.165-168) RECALL! The atoms in molecular compounds share electrons to form the stable electron arrangements of the closest noble gas. As a result, the molecules they
SNC2D CHEMISTRY CHEMICAL REACTIONS L (P.165-168) RECALL! The atoms in molecular compounds share electrons to form the stable electron arrangements of the closest noble gas. As a result, the molecules they
