Determination of Resonance Stabilization Energy of Benzene Jonathan Smith (With Small Adaptations by Amanda Nienow)
|
|
|
- Kelly Howard
- 6 years ago
- Views:
Transcription
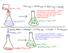
1 Determination of Resonance Stabilization Energy of Benzene Jonathan Smith (With Small Adaptations by Amanda Nienow) Abstract In this investigation we will measure the heat evolved during the combustion of an organic compound in the presence of excess oxygen under constant volume conditions. We will determine the caloric content of a piece of candy as well as determine ΔUcombustion for 1,5,9-trans, trans, cis-1,5,9-cyclododecatriene. The latter information will help us determine the resonance energy of benzene. Related Readings 1. Halpern, A. M. Experimental Physical Chemistry: A Laboratory Textbook. 2 nd Ed. Prentice Hall: Upper Saddle River, NJ. [Available in Nobel 107] 2. McQuarrie, D.A., Simon, J.D., Physical Chemistry: A Molecular Approach. University Science Books: Sausalito, CA. Chapter 19. Background In this investigation we will measure the heat evolved during the combustion of an organic compound in the presence of excess oxygen under constant volume conditions. The heat evolved, q, thus equals ΔU rather than ΔH. Upon determination of ΔU combustion we can then determine a wide range of properties of the compounds examined. ΔH f is readily determined from ΔU combustion and can be used to determine reaction energetics. In examining 1,5,9-trans, trans, cis-1,5,9-cyclododecatriene [ ] (CDDT) we can determine the resonance energy of benzene from the thermodynamics of the following theoretical reaction. C6H 6( g) (benzene) + C 6 H 12 (g) (cyclohexane) C 12 H 18 (g) (CDDT) + E resonance The reason this works is because both the reactants and products have the same number of each type of chemical bond (C-C, H-C, C=C). This type of reaction is called an isodesmic reaction and does not have to correspond to an actual chemical reaction but is useful in constructing a thermodynamic cycle which gives us insight into the relative stability of compounds. Isodesmic reactions produce better theoretical values because of the conservation of each type of bond. In this case the difference between reactants and products is the resonance energy of benzene. How do we get these values from bomb calorimetry? We compute the heats of formation for CDDT from our combustion of this compound and we use accepted values for benzene and cyclohexane. We need to convert from enthalpy to energy as well. Figure 1. CDDT
2 One of your pre-lab exercises is to draw a structure of CDDT and indicate through a thermodynamic cycle how this leads to the resonance energy of benzene. In this thermodynamic cycle, the heat of vaporization of CDDT will be needed. A relationship between log(p vap ) and 1/T has been established 1 and from this relationship ΔH vap can be determined (e.g., see the handout for the Vapor Pressure of a Pure Substance lab) log 10 ( P vap ) = (1) T The combustion is carried out in a pressure vessel (bomb) immersed in a water bath. The heat evolved is transferred to the bomb and water bath, resulting in a readily measured temperature rise. A second body of water, called the jacket, surrounds the water bath. Its temperature is adjusted to be equal or nearly equal to that of the water bath in order to eliminate or at least minimize heat losses from the bath. The calorimeter may be operated adiabatically by varying the jacket temperature during the experiment (by adjusting the relative flow rates of hot and cold water) so as to keep pace with the rising temperature of the inner water bath. Alternatively, the calorimeter may be operated isothermally by keeping the jacket temperature constant. You should approximate adiabatic conditions in this experiment. The heat evolved during the combustion of an unknown, q v, is related to the temperature rise by q v = ΔU combustion =ΔU c = C s (T 2 T 1 ) (2) where C s is the combined heat capacity in Joules per degree of the calorimeter pressure vessel (bomb), the water in the heat bath, and the walls of the water bath. We will measure this temperature rise with a thermometer in tandem with a computer-based acquisition. The heat evolved is the sum of the internal energy of combustion of the sample, ΔU c, plus the heat of formation of HNO 3 from N 2, ΔU f, (Equation (3) below) plus the heat of combustion of the fuse wire. Therefore, to find ΔU c, corrections need to be made for these other sources of energy. The process for these corrections will be discussed in the procedure section below. (Note that Eq (2) doesn t account for these processes.) N 2( g) + H 2 O(l) + 5/2O 2 (g) 2HNO 3 (g) (3) (Note: Many texts use the symbol E for internal energy U. The use of H for enthalpy is fairly universal. The word "heat" can be associated with q, ΔH, or ΔU. So be careful when consulting the literature.) From ΔU c one can find ΔH combustion. To do this, assume that all gases (from the combustion reaction of the sample) behave ideally and use equation 4. Use T f for T. (For the candy samples, you may not be able to find ΔH c since you don t have a balanced chemical reation.) ΔH c = ΔU c + Δn RT g (4) Pre-lab Exercises: 1. Obtain a piece of candy of your choice to combust in the bomb calorimeter. Those with a small water content and containing only carbohydrates work best. Some that have been run successfully in the past include chocolate bars, M&M s, Reese s Pieces, and Lifesavers. 2. Draw a structure of CDDT and indicate through a thermodynamic cycle how this leads to the resonance energy of benzene.
3 3. Read Experiment 5, Halpern available in Nobel 107 (a substitute, albeit not as specific for this experiment, is McQuarrie and Simon, Ch 19). 4. Answer the following in your notebook: A student combusts 1.0 g benzoic acid in a bomb calorimeter and finds the length of wire has decreased 2.7 cm and that 5 ml 0.1 M NaOH is needed to neutralize the liquid in the bomb. The initial temperature was C and the final temperature was C. What is C s? Safety Precautions: 1. Always wear safety goggles. 2. A gas cylinder containing O2 at high pressure is used in this experiment. Be sure the cylinder is securely attached to a strong foundation. Also, never fill the bomb with a pressure greater than 30 atm. 3. Do not use samples sizes greater than 1.1 g (to prevent too much heat from being produced. 4. Follow the steps given below in the exact order given, and take extreme care when igniting the bomb. Method/Procedure: Overview To determine C s (the heat capacity of the system), a sample of benzoic acid is burned because it has a well characterized heat of combustion and serves as a standard (ΔU c = kj g -1 ). The length of the fuse wire is measured before and after combustion [ΔU = 9.6 J (cm wire consumed)]. The bomb is rinsed with water after combustion, and the washings are titrated with 0.1 M NaOH to determine the amount of acid (assumed all HNO 3 ) present. (ΔU f ( HNO 3 ) is 5.77 J per ml of 0.1 M base used.) If ΔΤ is the temperature rise during the combustion of w b grams of benzoic acid, and combustion of d cm of wire, and the acid titration is V ml of 0.1 M alkali, then 26420w + 9.6d V C s = (5) ΔT After determining C s, you will be ready to measure t he heat of combustion of several compounds to determine some of their properties based on this standardization. Bomb calorimetry is also used to determine the caloric content of food, so you should choose some type of food to determine how many "calories" it contains, i.e. measure its heat of combustion (1 kcal = 1 food "calorie"). The internal energy of combustion (ΔU c ) for an unknown (on a per gram basis) can be computed using Equation (6). Cs ΔT 9.6d 5.77V Δ U C = (6) ws where w s is the weight of the sample; all other definitions from equation 5 hold. The internal energy of combustion computed from Equation (6) is without sign, but of course the heat of combustion must be negative since all combustions are exothermic. Be wary of significant figure conventions and convert values from kj/gram to kj/mole in consulting the literature and in comparing texts. You should report your internal energy of combustion as a negative quantity. We will determine the heat of combustion (ΔU c ) for 1,5,9-trans, trans, cis-1,5,9- cyclododecatriene and a piece of candy of your choice. The heat of combustion
4 determination for cis-1,5,9-trans, trans, cis-1,5,9-cyclododecatriene will allow us to investigate the resonance energy of benzene based on the consideration of a thermodynamic cycle. We will also find ΔH c for CDDT. We will determine the caloric content of the candy we examine, so you should choose some type of food to determine how many "calories" it contains, i.e. measure its heat of combustion (1 kcal = 1 food "calorie"). The link between combustion and our own use of the energy is that the respiration process involves the same net chemical reaction equation. Steps: Follow the steps given below exactly in the correct sequence. As always, wear your safety goggles. 1. Weigh out a sample of benzoic acid or other substance. The sample size must not exceed 1.1 g. Larger samples would evolve more than the calorimeter maximum of 10,000 cal and might cause an explosion. 2. Form the sample into a pellet using the press. It is probably not necessary to form your food sample into a pellet if it is already in compact form or a liquid. 3. Attach a measured length of fuse wire (about cm) between the electrodes. See p. 3 of the Parr Instruction Manual. Let the wire touch the pellet. 4. Place 1 ml of water in the bomb. 5. Carefully lower the head onto the bomb. Turn the screw cap down hand tight; never use a wrench. 6. If this is your first sample, ask your lab instructor/ta for help with the oxygen filling described in the next few steps. 7. Remove the inlet valve thumb nut from the bomb. Attach the filling connection from the oxygen tank. Draw tight by hand (no wrench). 8. Close the control valve (black knob) on the tank regulator. 9. Crack open (1/4 turn) the main valve on the oxygen tank. 10. Slowly open tile control valve. Let the bomb pressure increase slowly to 25 atm. Close the control valve. NEVER CHARGE TO MORE THAN 30 ATM. 11. Relieve the pressure in the connecting tube by flipping the relief valve (underneath the control valve). 12. Remove connecting tube from bomb. (The bomb has a one-way filling valve which automatically closes.) 13. Fill a 2-liter volumetric flask with distilled water. This should be 1-2 C below room temperature. Pour it into the oval bucket. 14. Submerge the bomb in the bucket, attaching the electric cable. 15. Close the calorimeter cover. Carefully lower the thermometers. Make sure both stirrer shafts are properly engaged. 16. Turn on the stirrer motor. (You may need to manually start the shaft wheel turning). 17. Take several temperature readings at 30 s intervals until the inner bath temperature is nearly constant. 18. Fire the fuse wire by pushing the button, and hold on until the light goes out. Never hold it down for more than a few seconds. 19. Continue to read the inner bath temperature at 30 s intervals after combustion. Examine a plot of temperature vs. time (or your recording of temperature over
5 time). After firing the bomb the temperature will start to climb and then you should record a fair amount of data to verify that the temperature has leveled out. 20. Take out the bomb. Slowly relieve the pressure by turning tile needle valve with the key. 21. Unscrew the cap. Measure the length of wire left. Rinse the bomb, and titrate the washings with standardized NaOH. 22. You should do at least two runs for each sample (a total of six runs two each for benzoic acid, cis-1,5,9-cyclododecatriene, and your candy). Do a third run for as many of the samples as you have time. Report/Analysis Follow the general guidelines for writing a lab report. The experimental section does not need to include all 23 steps outlined above, but do report with enough detail that someone familiar with bomb calorimetry could repeat this experiment. Determine the heat capacity of the system, and the heat of combustion for 1,5,9-trans, trans, cis-1,5,9- cyclododecatriene and a piece of candy of your choice. Use the heat of combustion determination for 1,5,9-trans, trans, cis-1,5,9-cyclododecatriene to investigate the resonance energy of benzene based on the consideration of a thermodynamic cycle and to determine ΔH C. In addition, determine the caloric content of the candy you examined (i.e. measure its heat of combustion and use the conversion 1 kcal = 1 food "calorie"). Discuss your determination of the food calories and how it relates to the packaging of the candy. If possible, compare your results to literature or other such published data. Include a discussion on error analysis (major sources, how to reduce, limitations, etc). References 1. Rauh, H. J.; Geyer, W.; Schmidt, H.; Geisler, G. Z. Phys. Chemie (Liepzip) 1973, 253, Halpern, A. M. Experimental Physical Chemistry, second ed.; Prentice Hall: Upper Saddle River, NJ, McQuarrie, D.A., Simon, J.D., Physical Chemistry: A Molecular Approach. University Science Books: Sausalito, CA. Chapter Parr Bomb Calorimeters,
Heat of Combustion. Parr oxygen bomb calorimeter (or equivalent), pellet press, thermometer (0.01 C), fuse wire.
 Heat of Combustion PURPOSE The purposes of this experiment are to determine the heats of combustion of several related substances using a bomb calorimeter and relate differences in heats of combustion
Heat of Combustion PURPOSE The purposes of this experiment are to determine the heats of combustion of several related substances using a bomb calorimeter and relate differences in heats of combustion
Resonance Energy of Benzene
 Resonance Energy of Benzene Purpose The purpose of this experiment is to evaluate the resonance energy of benzene by calorimetry. The heat of combustion of a compound will be measured with a bomb calorimeter.
Resonance Energy of Benzene Purpose The purpose of this experiment is to evaluate the resonance energy of benzene by calorimetry. The heat of combustion of a compound will be measured with a bomb calorimeter.
Bomb Calorimetry: Heat of Combustion of Naphthalene
 Bomb Calorimetry: Heat of Combustion of Naphthalene Most tabulated H values of highly exothermic reactions come from bomb calorimeter experiments. Heats of combustion are most common, in which the combustible
Bomb Calorimetry: Heat of Combustion of Naphthalene Most tabulated H values of highly exothermic reactions come from bomb calorimeter experiments. Heats of combustion are most common, in which the combustible
The Enthalpy of Formation of Camphor by Bomb Calorimetry 1
 The Enthalpy of Formation of Camphor by Bomb Calorimetry 1 Purpose: The enthalpy of combustion of camphor will be determined in a bomb calorimeter. The enthalpy of formation of camphor will be calculated
The Enthalpy of Formation of Camphor by Bomb Calorimetry 1 Purpose: The enthalpy of combustion of camphor will be determined in a bomb calorimeter. The enthalpy of formation of camphor will be calculated
Experiment 1: Adiabatic Bomb Calorimeter (Dated: August 25, 2009)
 Experiment 1: Adiabatic Bomb Calorimeter (Dated: August 25, 2009) I. INTRODUCTION Heat released in a chemical reaction can be determined experimentally by using an adiabatic calorimeter. The reaction must
Experiment 1: Adiabatic Bomb Calorimeter (Dated: August 25, 2009) I. INTRODUCTION Heat released in a chemical reaction can be determined experimentally by using an adiabatic calorimeter. The reaction must
Bomb Calorimetry. Read the lab thoroughly. Answer the pre-lab questions that appear at the end of this lab exercise.
 Experiment 12 Bomb Calorimetry Pre-Lab Assignment Before coming to lab: Read the lab thoroughly. Answer the pre-lab questions that appear at the end of this lab exercise. Purpose A bomb calorimeter will
Experiment 12 Bomb Calorimetry Pre-Lab Assignment Before coming to lab: Read the lab thoroughly. Answer the pre-lab questions that appear at the end of this lab exercise. Purpose A bomb calorimeter will
4 BOMB CALORIMETRY. Chapter 4. Bomb Calorimetry 24 I. PREPARATION
 Chapter 4. Bomb Calorimetry 24 4 BOMB CALORIMETRY I. PREPARATION Read "Principles of Calorimetry" in Ch. VI of Shoemaker, Garland, and Nibler (SGN, pp. 145-151), as well as Experiment 6 (pp. 152-158).
Chapter 4. Bomb Calorimetry 24 4 BOMB CALORIMETRY I. PREPARATION Read "Principles of Calorimetry" in Ch. VI of Shoemaker, Garland, and Nibler (SGN, pp. 145-151), as well as Experiment 6 (pp. 152-158).
How bad is that snack anyway?
 Physical Sciences 11 Experiment 1 How bad is that snack anyway? Monday, 2/10 Wednesday, 2/12 Science Center Room 117 Please read this entire document and complete the attached prelab before your lab. This
Physical Sciences 11 Experiment 1 How bad is that snack anyway? Monday, 2/10 Wednesday, 2/12 Science Center Room 117 Please read this entire document and complete the attached prelab before your lab. This
PROPULSION LAB MANUAL
 PROPULSION LAB MANUAL Measurement of Calorific Value of a Solid Fuel Sample using a Bomb Calorimeter DEPARTMENT OF AEROSPACE ENGINEERING Indian Institute of Technology Kharagpur CONTENTS 1. Introduction
PROPULSION LAB MANUAL Measurement of Calorific Value of a Solid Fuel Sample using a Bomb Calorimeter DEPARTMENT OF AEROSPACE ENGINEERING Indian Institute of Technology Kharagpur CONTENTS 1. Introduction
Lab 2. Molar enthalpy of combustion
 Lab 2. Molar enthalpy of combustion Introduction Chemical reactions like burning an organic compound typically produce heat. In this experiment you will be measuring the amount of heat produced by burning
Lab 2. Molar enthalpy of combustion Introduction Chemical reactions like burning an organic compound typically produce heat. In this experiment you will be measuring the amount of heat produced by burning
1. State in your own terms what is the first law of thermodynamics, a closed system, an isolated system, surroundings, heat, work, and energy.
 Worksheet 1 1. State in your own terms what is the first law of thermodynamics, a closed system, an isolated system, surroundings, heat, work, and energy. The first law of thermodynamics is the conservation
Worksheet 1 1. State in your own terms what is the first law of thermodynamics, a closed system, an isolated system, surroundings, heat, work, and energy. The first law of thermodynamics is the conservation
Thermochemistry/Calorimetry. Determination of the enthalpy of combustion with a calorimetric bomb LEC 02. What you need:
 LEC 02 Thermochemistry/Calorimetry with a calorimetric bomb What you can learn about 1st law of thermodynamics Hess law Enthalpy of combustion Enthalpy of formation Heat capacity Principle and tasks The
LEC 02 Thermochemistry/Calorimetry with a calorimetric bomb What you can learn about 1st law of thermodynamics Hess law Enthalpy of combustion Enthalpy of formation Heat capacity Principle and tasks The
MEEN 3242 MEE LAB II
 MEEN 3242 MEE LAB II Experiment Report Lab # 2 Bomb Calorimeter Group Members: Hugo Diaz Reed Greenwood Juan Orona Alan Sanoja Micheal Shackelford Date: September 27, 2013 DEPARTMENT OF MECHANICAL AND
MEEN 3242 MEE LAB II Experiment Report Lab # 2 Bomb Calorimeter Group Members: Hugo Diaz Reed Greenwood Juan Orona Alan Sanoja Micheal Shackelford Date: September 27, 2013 DEPARTMENT OF MECHANICAL AND
AC : PC-BASED MEASUREMENT OF THE HEAT OF COMBUSTION OF A SOLID FUEL USING OXYGEN BOMB CALORIMETER
 AC 2007-2839: PC-BASED MEASUREMENT OF THE HEAT OF COMBUSTION OF A SOLID FUEL USING OXYGEN BOMB CALORIMETER Ramesh Prasad, University of New Brunswick-St. John Ramesh C. Prasad, Professor of Mechanical
AC 2007-2839: PC-BASED MEASUREMENT OF THE HEAT OF COMBUSTION OF A SOLID FUEL USING OXYGEN BOMB CALORIMETER Ramesh Prasad, University of New Brunswick-St. John Ramesh C. Prasad, Professor of Mechanical
Chapter 5. Thermochemistry
 Chapter 5 Thermochemistry Dr. A. Al-Saadi 1 Preview Introduction to thermochemistry: Potential energy and kinetic energy. Chemical energy. Internal energy, work and heat. Exothermic vs. endothermic reactions.
Chapter 5 Thermochemistry Dr. A. Al-Saadi 1 Preview Introduction to thermochemistry: Potential energy and kinetic energy. Chemical energy. Internal energy, work and heat. Exothermic vs. endothermic reactions.
Chapter 8 Thermochemistry
 William L Masterton Cecile N. Hurley http://academic.cengage.com/chemistry/masterton Chapter 8 Thermochemistry Edward J. Neth University of Connecticut Outline 1. Principles of heat flow 2. Measurement
William L Masterton Cecile N. Hurley http://academic.cengage.com/chemistry/masterton Chapter 8 Thermochemistry Edward J. Neth University of Connecticut Outline 1. Principles of heat flow 2. Measurement
11B, 11E Temperature and heat are related but not identical.
 Thermochemistry Key Terms thermochemistry heat thermochemical equation calorimeter specific heat molar enthalpy of formation temperature enthalpy change enthalpy of combustion joule enthalpy of reaction
Thermochemistry Key Terms thermochemistry heat thermochemical equation calorimeter specific heat molar enthalpy of formation temperature enthalpy change enthalpy of combustion joule enthalpy of reaction
ENTHALPY OF FORMATION OF MgO
 ENTHALPY OF FORMATION OF MgO ELECTRONIC LABORATORY NOTEBOOK (ELN) INSTRUCTIONS All work for this experiment must be recorded, attached, or answered in the ELN. Create a pre & inlab page in the Experiment
ENTHALPY OF FORMATION OF MgO ELECTRONIC LABORATORY NOTEBOOK (ELN) INSTRUCTIONS All work for this experiment must be recorded, attached, or answered in the ELN. Create a pre & inlab page in the Experiment
Chapter 5 Thermochemistry. 許富銀 ( Hsu Fu-Yin)
 Chapter 5 Thermochemistry 許富銀 ( Hsu Fu-Yin) 1 Thermodynamics The study of energy and its transformations is known as thermodynamics The relationships between chemical reactions and energy changes that
Chapter 5 Thermochemistry 許富銀 ( Hsu Fu-Yin) 1 Thermodynamics The study of energy and its transformations is known as thermodynamics The relationships between chemical reactions and energy changes that
THE ENERGY OF PHASE CHANGES
 C H E M I S T R Y 1 5 0 Chemistry for Engineers THE ENERGY OF PHASE CHANGES DEPARTMENT OF CHEMISTRY UNIVERSITY OF KANSAS The Energy of Phase Changes Introduction Consider heating a solid: as the solid
C H E M I S T R Y 1 5 0 Chemistry for Engineers THE ENERGY OF PHASE CHANGES DEPARTMENT OF CHEMISTRY UNIVERSITY OF KANSAS The Energy of Phase Changes Introduction Consider heating a solid: as the solid
Chapter 5 THERMO. THERMO chemistry. 5.4 Enthalpy of Reactions 5.5 Calorimetry 5.6 Hess s Law 5.7 Enthalpies of Formation
 Chapter 5 THERMO THERMO chemistry 5.4 Enthalpy of Reactions 5.5 Calorimetry 5.6 Hess s Law 5.7 Enthalpies of Formation Chemical Equations 1 st WRITE the Chemical Equation 2 nd BALANCE the Chemical Equation
Chapter 5 THERMO THERMO chemistry 5.4 Enthalpy of Reactions 5.5 Calorimetry 5.6 Hess s Law 5.7 Enthalpies of Formation Chemical Equations 1 st WRITE the Chemical Equation 2 nd BALANCE the Chemical Equation
Types of Energy Calorimetry q = mc T Thermochemical Equations Hess s Law Spontaneity, Entropy, Gibb s Free energy
 Unit 7: Energy Outline Types of Energy Calorimetry q = mc T Thermochemical Equations Hess s Law Spontaneity, Entropy, Gibb s Free energy Energy Energy is the ability to do work or produce heat. The energy
Unit 7: Energy Outline Types of Energy Calorimetry q = mc T Thermochemical Equations Hess s Law Spontaneity, Entropy, Gibb s Free energy Energy Energy is the ability to do work or produce heat. The energy
Physical Chemistry Laboratory [CHEM 335] BOMB CALORIMETRY Adriana Biney and Silmilly Toribio
![Physical Chemistry Laboratory [CHEM 335] BOMB CALORIMETRY Adriana Biney and Silmilly Toribio Physical Chemistry Laboratory [CHEM 335] BOMB CALORIMETRY Adriana Biney and Silmilly Toribio](/thumbs/87/96015679.jpg) Physical Chemistry Laboratory [CHEM 335] BOMB CALORIMETRY Adriana Biney and Silmilly Toribio ABSTRACT In this experiment a Parr 1341 Plain Jacket Oxygen Bomb Calorimeter was employed to determine the enthalpy
Physical Chemistry Laboratory [CHEM 335] BOMB CALORIMETRY Adriana Biney and Silmilly Toribio ABSTRACT In this experiment a Parr 1341 Plain Jacket Oxygen Bomb Calorimeter was employed to determine the enthalpy
Calorimetry. Summary. Contents
 Summary Calorimetry Calorimetry can be used to determine the heat flow in chemical reactions, to measure heats of solvation, characterize phase transitions and ligand binding to proteins. Theoretical predictions
Summary Calorimetry Calorimetry can be used to determine the heat flow in chemical reactions, to measure heats of solvation, characterize phase transitions and ligand binding to proteins. Theoretical predictions
Calorimetry Measurements of Fusion, Hydration and Neutralization - Hess Law
 Calorimetry Measurements of Fusion, Hydration and Neutralization - Hess Law EXPERIMENT 9 Prepared by Edward L. Brown, Lee University and Verrill M. Norwood, Cleveland State Community College To become
Calorimetry Measurements of Fusion, Hydration and Neutralization - Hess Law EXPERIMENT 9 Prepared by Edward L. Brown, Lee University and Verrill M. Norwood, Cleveland State Community College To become
Hess' Law: Calorimetry
 Exercise 9 Page 1 Illinois Central College CHEMISTRY 130 Name: Hess' Law: Calorimetry Objectives The objectives of this experiment are to... - measure the heats of reaction for two chemical reactions.
Exercise 9 Page 1 Illinois Central College CHEMISTRY 130 Name: Hess' Law: Calorimetry Objectives The objectives of this experiment are to... - measure the heats of reaction for two chemical reactions.
Apply the ideal gas law (PV = nrt) to experimentally determine the number of moles of carbon dioxide gas generated
 Teacher Information Ideal Gas Law Objectives Determine the number of moles of carbon dioxide gas generated during a reaction between hydrochloric acid and sodium bicarbonate. Through this investigation,
Teacher Information Ideal Gas Law Objectives Determine the number of moles of carbon dioxide gas generated during a reaction between hydrochloric acid and sodium bicarbonate. Through this investigation,
Name: Section: Score: /10 PRE LABORATORY ASSIGNMENT EXPERIMENT 7
 Name: Section: Score: /10 PRE LABORATORY ASSIGNMENT EXPERIMENT 7 1. Is the sign of Δ r H for an exothermic reaction positive or negative? Why? 2. When 4.21 grams of potassium hydroxide are added to 250.
Name: Section: Score: /10 PRE LABORATORY ASSIGNMENT EXPERIMENT 7 1. Is the sign of Δ r H for an exothermic reaction positive or negative? Why? 2. When 4.21 grams of potassium hydroxide are added to 250.
Chapter 8 Thermochemistry: Chemical Energy. Chemical Thermodynamics
 Chapter 8 Thermochemistry: Chemical Energy Chapter 8 1 Chemical Thermodynamics Chemical Thermodynamics is the study of the energetics of a chemical reaction. Thermodynamics deals with the absorption or
Chapter 8 Thermochemistry: Chemical Energy Chapter 8 1 Chemical Thermodynamics Chemical Thermodynamics is the study of the energetics of a chemical reaction. Thermodynamics deals with the absorption or
Enthalpies of Reaction
 Enthalpies of Reaction Enthalpy is an extensive property Magnitude of H is directly related to the amount of reactant used up in a process. CH 4 (g) + 2O 2 (g) CO 2 (g) + 2H 2 O(l) H = 890 kj 2CH 4 (g)
Enthalpies of Reaction Enthalpy is an extensive property Magnitude of H is directly related to the amount of reactant used up in a process. CH 4 (g) + 2O 2 (g) CO 2 (g) + 2H 2 O(l) H = 890 kj 2CH 4 (g)
Thermochemistry Chapter 8
 Thermochemistry Chapter 8 Thermochemistry First law of thermochemistry: Internal energy of an isolated system is constant; energy cannot be created or destroyed; however, energy can be converted to different
Thermochemistry Chapter 8 Thermochemistry First law of thermochemistry: Internal energy of an isolated system is constant; energy cannot be created or destroyed; however, energy can be converted to different
The Nature of Energy. Chapter Six: Kinetic vs. Potential Energy. Energy and Work. Temperature vs. Heat
 The Nature of Energy Chapter Six: THERMOCHEMISTRY Thermodynamics is the study of energy and its transformations. Thermochemistry is the study of the relationship between chemical reactions and energy changes
The Nature of Energy Chapter Six: THERMOCHEMISTRY Thermodynamics is the study of energy and its transformations. Thermochemistry is the study of the relationship between chemical reactions and energy changes
Designing a Hand Warmer AP* Chemistry Big Idea 5, Investigation 12 An Advanced Inquiry Lab
 Introduction Designing a Hand Warmer AP* Chemistry Big Idea 5, Investigation 12 An Advanced Inquiry Lab Catalog No. AP7654 Publication No. 7654 WEB Put your chemistry skills to commercial use! From instant
Introduction Designing a Hand Warmer AP* Chemistry Big Idea 5, Investigation 12 An Advanced Inquiry Lab Catalog No. AP7654 Publication No. 7654 WEB Put your chemistry skills to commercial use! From instant
Chapter 6 Thermochemistry 許富銀
 Chapter 6 Thermochemistry 許富銀 6.1 Chemical Hand Warmers Thermochemistry: the study of the relationships between chemistry and energy Hand warmers use the oxidation of iron as the exothermic reaction: Nature
Chapter 6 Thermochemistry 許富銀 6.1 Chemical Hand Warmers Thermochemistry: the study of the relationships between chemistry and energy Hand warmers use the oxidation of iron as the exothermic reaction: Nature
8.6 The Thermodynamic Standard State
 8.6 The Thermodynamic Standard State The value of H reported for a reaction depends on the number of moles of reactants...or how much matter is contained in the system C 3 H 8 (g) + 5O 2 (g) > 3CO 2 (g)
8.6 The Thermodynamic Standard State The value of H reported for a reaction depends on the number of moles of reactants...or how much matter is contained in the system C 3 H 8 (g) + 5O 2 (g) > 3CO 2 (g)
COPYRIGHT FOUNTAINHEAD PRESS
 Calorimetry: Heats of Solution Objective: Use calorimetric measurements to determine heats of solution of two ionic compounds. Materials: Solid ammonium nitrate (NH 4 NO 3 ) and anhydrous calcium chloride
Calorimetry: Heats of Solution Objective: Use calorimetric measurements to determine heats of solution of two ionic compounds. Materials: Solid ammonium nitrate (NH 4 NO 3 ) and anhydrous calcium chloride
Lecture Outline. 5.1 The Nature of Energy. Kinetic Energy and Potential Energy. 1 mv
 Chapter 5. Thermochemistry Common Student Misconceptions Students confuse power and energy. Students confuse heat with temperature. Students fail to note that the first law of thermodynamics is the law
Chapter 5. Thermochemistry Common Student Misconceptions Students confuse power and energy. Students confuse heat with temperature. Students fail to note that the first law of thermodynamics is the law
COMBUSTION OF FUEL 12:57:42
 COMBUSTION OF FUEL The burning of fuel in presence of air is known as combustion. It is a chemical reaction taking place between fuel and oxygen at temperature above ignition temperature. Heat is released
COMBUSTION OF FUEL The burning of fuel in presence of air is known as combustion. It is a chemical reaction taking place between fuel and oxygen at temperature above ignition temperature. Heat is released
Thermochemistry: Calorimetry and Hess s Law
 Thermochemistry: Calorimetry and Hess s Law Some chemical reactions are endothermic and proceed with absorption of heat while others are exothermic and proceed with an evolution of heat. The magnitude
Thermochemistry: Calorimetry and Hess s Law Some chemical reactions are endothermic and proceed with absorption of heat while others are exothermic and proceed with an evolution of heat. The magnitude
CHEM 1105 S10 March 11 & 14, 2014
 CHEM 1105 S10 March 11 & 14, 2014 Today s topics: Thermochemistry (Chapter 6) Basic definitions Calorimetry Enthalpy Thermochemical equations Calculating heats of reaction Hess s Law Energy and Heat Some
CHEM 1105 S10 March 11 & 14, 2014 Today s topics: Thermochemistry (Chapter 6) Basic definitions Calorimetry Enthalpy Thermochemical equations Calculating heats of reaction Hess s Law Energy and Heat Some
Chapter 17 Thermochemistry
 Chapter 17 Thermochemistry Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Explain how energy, heat, and work are related. 2 Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Classify
Chapter 17 Thermochemistry Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Explain how energy, heat, and work are related. 2 Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Classify
THERMOCHEMISTRY & DEFINITIONS
 THERMOCHEMISTRY & DEFINITIONS Thermochemistry is the study of the study of relationships between chemistry and energy. All chemical changes and many physical changes involve exchange of energy with the
THERMOCHEMISTRY & DEFINITIONS Thermochemistry is the study of the study of relationships between chemistry and energy. All chemical changes and many physical changes involve exchange of energy with the
Section 9: Thermodynamics and Energy
 Section 9: Thermodynamics and Energy The following maps the videos in this section to the Texas Essential Knowledge and Skills for Science TAC 112.35(c). 9.01 Law of Conservation of Energy Chemistry (11)(A)
Section 9: Thermodynamics and Energy The following maps the videos in this section to the Texas Essential Knowledge and Skills for Science TAC 112.35(c). 9.01 Law of Conservation of Energy Chemistry (11)(A)
AP* Chemistry THERMOCHEMISTRY
 AP* Chemistry THERMOCHEMISTRY Terms for you to learn that will make this unit understandable: Energy (E) the ability to do work or produce heat ; the sum of all potential and kinetic energy in a system
AP* Chemistry THERMOCHEMISTRY Terms for you to learn that will make this unit understandable: Energy (E) the ability to do work or produce heat ; the sum of all potential and kinetic energy in a system
Experiment 12 Determination of an Enthalpy of Reaction, Using Hess s Law
 Experiment 12 Determination of an Enthalpy of Reaction, Using Hess s Law Object: To measure the standard heat of formation, f, of MgO (s), and to become familiar with calorimetry as a toll for measuring
Experiment 12 Determination of an Enthalpy of Reaction, Using Hess s Law Object: To measure the standard heat of formation, f, of MgO (s), and to become familiar with calorimetry as a toll for measuring
Chapter 6: Thermochemistry
 Chapter 6: Thermochemistry Section 6.1: Introduction to Thermochemistry Thermochemistry refers to the study of heat flow or heat energy in a chemical reaction. In a study of Thermochemistry the chemical
Chapter 6: Thermochemistry Section 6.1: Introduction to Thermochemistry Thermochemistry refers to the study of heat flow or heat energy in a chemical reaction. In a study of Thermochemistry the chemical
EXPERIMENT 5. THE ENTHALPY OF FORMATION OF CAMPHOR BY BOMB CALORIMETRY 1
 EXPERIMENT 5. THE ENTHALPY OF FORMATION OF CAMPHOR BY BOMB CALORIMETRY 1 The enthalpies of combustion and formation for camphor will be determined using a bomb calorimeter and tabulated values for the
EXPERIMENT 5. THE ENTHALPY OF FORMATION OF CAMPHOR BY BOMB CALORIMETRY 1 The enthalpies of combustion and formation for camphor will be determined using a bomb calorimeter and tabulated values for the
HEATS OF REACTION EXPERIMENT
 16 Text Reference Section 11.4 HEATS OF REACTION EXPERIMENT PURPOSE To measure the heats of reaction for three related exothermic reactions and to verify Hess s law of heat summation. Time Required 50
16 Text Reference Section 11.4 HEATS OF REACTION EXPERIMENT PURPOSE To measure the heats of reaction for three related exothermic reactions and to verify Hess s law of heat summation. Time Required 50
Quantities in Chemical Reactions
 Quantities in Chemical Reactions 6-1 6.1 The Meaning of a Balanced Equation C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O (g) The balanced equation tells us: 1 molecule of propane reacts with 5 molecules of
Quantities in Chemical Reactions 6-1 6.1 The Meaning of a Balanced Equation C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O (g) The balanced equation tells us: 1 molecule of propane reacts with 5 molecules of
Name Date Class THE FLOW OF ENERGY HEAT AND WORK
 17.1 THE FLOW OF ENERGY HEAT AND WORK Section Review Objectives Explain the relationship between energy, heat, and work Distinguish between exothermic and endothermic processes Distinguish between heat
17.1 THE FLOW OF ENERGY HEAT AND WORK Section Review Objectives Explain the relationship between energy, heat, and work Distinguish between exothermic and endothermic processes Distinguish between heat
Chemistry: The Central Science. Chapter 5: Thermochemistry
 Chemistry: The Central Science Chapter 5: Thermochemistry Study of energy and its transformations is called thermodynamics Portion of thermodynamics that involves the relationships between chemical and
Chemistry: The Central Science Chapter 5: Thermochemistry Study of energy and its transformations is called thermodynamics Portion of thermodynamics that involves the relationships between chemical and
Thermochemistry: Heat and Chemical Change
 Thermochemistry: Heat and Chemical Change 1 Heat or Thermal Energy (q) Heat is a form of energy Is heat the same as temperature? Heat flows between two objects at different temperatures. Hot Cold 2 Chemical
Thermochemistry: Heat and Chemical Change 1 Heat or Thermal Energy (q) Heat is a form of energy Is heat the same as temperature? Heat flows between two objects at different temperatures. Hot Cold 2 Chemical
Thermochemistry is the study of the relationships between chemical reactions and energy changes involving heat.
 CHEM134- F18 Dr. Al- Qaisi Chapter 06: Thermodynamics Thermochemistry is the study of the relationships between chemical reactions and energy changes involving heat. Energy is anything that has the capacity
CHEM134- F18 Dr. Al- Qaisi Chapter 06: Thermodynamics Thermochemistry is the study of the relationships between chemical reactions and energy changes involving heat. Energy is anything that has the capacity
Chapter 5. Thermochemistry
 Chapter 5 Thermochemistry Energy Thermodynamics Study of the relationship between heat, work, and other forms of energy Thermochemistry A branch of thermodynamics Focuses on the study of heat given off
Chapter 5 Thermochemistry Energy Thermodynamics Study of the relationship between heat, work, and other forms of energy Thermochemistry A branch of thermodynamics Focuses on the study of heat given off
Chapter 5 Thermochemistry
 Chapter 5 Thermochemistry Learning Outcomes: Interconvert energy units Distinguish between the system and the surroundings in thermodynamics Calculate internal energy from heat and work and state sign
Chapter 5 Thermochemistry Learning Outcomes: Interconvert energy units Distinguish between the system and the surroundings in thermodynamics Calculate internal energy from heat and work and state sign
If neither volume nor pressure of the system changes, w = 0 and ΔE = q = ΔH. The change in internal energy is equal to the change in enthalpy.
 5 Thermochemistry Visualizing Concepts 5.1 The book s potential energy is due to the opposition of gravity by an object of mass m at a distance d above the surface of the earth. Kinetic energy is due to
5 Thermochemistry Visualizing Concepts 5.1 The book s potential energy is due to the opposition of gravity by an object of mass m at a distance d above the surface of the earth. Kinetic energy is due to
Thermochemistry. Energy. 1st Law of Thermodynamics. Enthalpy / Calorimetry. Enthalpy of Formation
 Thermochemistry Energy 1st Law of Thermodynamics Enthalpy / Calorimetry Hess' Law Enthalpy of Formation The Nature of Energy Kinetic Energy and Potential Energy Kinetic energy is the energy of motion:
Thermochemistry Energy 1st Law of Thermodynamics Enthalpy / Calorimetry Hess' Law Enthalpy of Formation The Nature of Energy Kinetic Energy and Potential Energy Kinetic energy is the energy of motion:
Thermochemistry: Energy Flow and Chemical Reactions
 Thermochemistry: Energy Flow and Chemical Reactions Outline thermodynamics internal energy definition, first law enthalpy definition, energy diagrams, calorimetry, theoretical calculation (heats of formation
Thermochemistry: Energy Flow and Chemical Reactions Outline thermodynamics internal energy definition, first law enthalpy definition, energy diagrams, calorimetry, theoretical calculation (heats of formation
Chapter 6. Energy Thermodynamics
 Chapter 6 Energy Thermodynamics 1 Energy is... The ability to do work. Conserved. made of heat and work. a state function. independent of the path, or how you get from point A to B. Work is a force acting
Chapter 6 Energy Thermodynamics 1 Energy is... The ability to do work. Conserved. made of heat and work. a state function. independent of the path, or how you get from point A to B. Work is a force acting
ENTHALPY, INTERNAL ENERGY, AND CHEMICAL REACTIONS: AN OUTLINE FOR CHEM 101A
 ENTHALPY, INTERNAL ENERGY, AND CHEMICAL REACTIONS: AN OUTLINE FOR CHEM 101A PART 1: KEY TERMS AND SYMBOLS IN THERMOCHEMISTRY System and surroundings When we talk about any kind of change, such as a chemical
ENTHALPY, INTERNAL ENERGY, AND CHEMICAL REACTIONS: AN OUTLINE FOR CHEM 101A PART 1: KEY TERMS AND SYMBOLS IN THERMOCHEMISTRY System and surroundings When we talk about any kind of change, such as a chemical
= (25.0 g)(0.137 J/g C)[61.2 C - (-31.4 C)] = 317 J (= kj)
![= (25.0 g)(0.137 J/g C)[61.2 C - (-31.4 C)] = 317 J (= kj) = (25.0 g)(0.137 J/g C)[61.2 C - (-31.4 C)] = 317 J (= kj)](/thumbs/89/101004910.jpg) CHEM 101A ARMSTRONG SOLUTIONS TO TOPIC D PROBLEMS 1) For all problems involving energy, you may give your answer in either joules or kilojoules, unless the problem specifies a unit. (In general, though,
CHEM 101A ARMSTRONG SOLUTIONS TO TOPIC D PROBLEMS 1) For all problems involving energy, you may give your answer in either joules or kilojoules, unless the problem specifies a unit. (In general, though,
Thermochemistry. Energy. 1st Law of Thermodynamics. Enthalpy / Calorimetry. Enthalpy of Formation
 THERMOCHEMISTRY Thermochemistry Energy 1st Law of Thermodynamics Enthalpy / Calorimetry Hess' Law Enthalpy of Formation The Nature of Energy Kinetic Energy and Potential Energy Kinetic energy is the energy
THERMOCHEMISTRY Thermochemistry Energy 1st Law of Thermodynamics Enthalpy / Calorimetry Hess' Law Enthalpy of Formation The Nature of Energy Kinetic Energy and Potential Energy Kinetic energy is the energy
Thermochemistry-Part 1
 Brad Collins Thermochemistry-Part 1 Chapter 7 Thermochemistry Thermodynamics: The study of energy Thermochemistry: The study of energy in chemical reactions Energy: The capacity to do work Work = force
Brad Collins Thermochemistry-Part 1 Chapter 7 Thermochemistry Thermodynamics: The study of energy Thermochemistry: The study of energy in chemical reactions Energy: The capacity to do work Work = force
Quantities in Chemical Reactions
 Quantities in Chemical Reactions 6-1 6.1 The Meaning of a Balanced Equation C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O (g) The balanced equation tells us: 1 molecule of propane reacts with 5 molecules of
Quantities in Chemical Reactions 6-1 6.1 The Meaning of a Balanced Equation C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O (g) The balanced equation tells us: 1 molecule of propane reacts with 5 molecules of
AP CHEMISTRY. Unit 5 Thermochemistry. Jeff Venables Northwestern High School
 AP CHEMISTRY Unit 5 Thermochemistry Jeff Venables Northwestern High School Kinetic Energy and Potential Energy Kinetic energy - the energy of motion: Ek = 1 mv 2 Potential energy - the energy an object
AP CHEMISTRY Unit 5 Thermochemistry Jeff Venables Northwestern High School Kinetic Energy and Potential Energy Kinetic energy - the energy of motion: Ek = 1 mv 2 Potential energy - the energy an object
June Which is a closed system? (A) burning candle (B) halogen lightbulb (C) hot water in a sink (D) ripening banana
 June 2005 28. Which is a closed system? burning candle halogen lightbulb hot water in a sink ripening banana 29. Which involves the greatest energy change? chemical reaction nuclear reaction phase change
June 2005 28. Which is a closed system? burning candle halogen lightbulb hot water in a sink ripening banana 29. Which involves the greatest energy change? chemical reaction nuclear reaction phase change
DETERMINING AND USING H
 DETERMINING AND USING H INTRODUCTION CHANGES IN CHEMISTRY Chemistry is the science that studies matter and the changes it undergoes. Changes are divided into two categories: physical and chemical. During
DETERMINING AND USING H INTRODUCTION CHANGES IN CHEMISTRY Chemistry is the science that studies matter and the changes it undergoes. Changes are divided into two categories: physical and chemical. During
Thermodynamics Enthalpy of Reaction and Hess s Law
 P.O. Box 219 Batavia, Illinois 60510 1-800-452-1261 flinn@flinnsci.com Visit our website at: www.flinnsci.com 2003 Flinn Scientific, Inc. All Rights Reserved. Your Safer Source for Science Supplies Thermodynamics
P.O. Box 219 Batavia, Illinois 60510 1-800-452-1261 flinn@flinnsci.com Visit our website at: www.flinnsci.com 2003 Flinn Scientific, Inc. All Rights Reserved. Your Safer Source for Science Supplies Thermodynamics
CHAPTER 3 THE FIRST LAW OF THERMODYNAM- ICS
 CHAPTER 3 THE FIRST LAW OF THERMODYNAM- ICS Introduction In this chapter, we discuss the First Law of Thermodynamics (energy cannot be created or destroyed). In our discussion, we will define some important
CHAPTER 3 THE FIRST LAW OF THERMODYNAM- ICS Introduction In this chapter, we discuss the First Law of Thermodynamics (energy cannot be created or destroyed). In our discussion, we will define some important
This reaction is ENDOTHERMIC. Energy is being transferred from the room/flask/etc. (the SURROUNDINGS) to the reaction itself (the SYSTEM).
 151 This reaction is EXOTHERMIC. Energy is transferred from the reactants and products (the SYSTEM) to the water in the flask, the flask, etc. (the SURROUNDINGS) This reaction is ENDOTHERMIC. Energy is
151 This reaction is EXOTHERMIC. Energy is transferred from the reactants and products (the SYSTEM) to the water in the flask, the flask, etc. (the SURROUNDINGS) This reaction is ENDOTHERMIC. Energy is
Gravity is a force which keeps us stuck to the earth. The Electrostatic force attracts electrons to protons in an atom.
 Energy Relations in Chemistry: Thermochemistry The Nature of Energy Sugar you eat is "combusted" by your body to produce CO 2 and H 2 O. During this process energy is also released. This energy is used
Energy Relations in Chemistry: Thermochemistry The Nature of Energy Sugar you eat is "combusted" by your body to produce CO 2 and H 2 O. During this process energy is also released. This energy is used
Chapter 5 Thermochemistry
 Chapter 5 Thermochemistry Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Explain how energy, heat, and work are related. 2 Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Classify
Chapter 5 Thermochemistry Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Explain how energy, heat, and work are related. 2 Section 17.1 The Flow of Energy Heat and Work OBJECTIVES: Classify
CHEMISTRY. Chapter 5 Thermochemistry
 CHEMISTRY The Central Science 8 th Edition Chapter 5 Thermochemistry Dr. Kozet YAPSAKLI The Nature of Energy Kinetic and Potential Energy Potential energy can be converted into kinetic energy. E p = mgh
CHEMISTRY The Central Science 8 th Edition Chapter 5 Thermochemistry Dr. Kozet YAPSAKLI The Nature of Energy Kinetic and Potential Energy Potential energy can be converted into kinetic energy. E p = mgh
Calorimetry: Heat of Solution
 Calorimetry: Heat of Solution When a substance undergoes a change in temperature, the quantity of heat lost or gained can be calculated using the following relationship: (heat) = m s T (1) The specific
Calorimetry: Heat of Solution When a substance undergoes a change in temperature, the quantity of heat lost or gained can be calculated using the following relationship: (heat) = m s T (1) The specific
Experiment #12. Enthalpy of Neutralization
 Experiment #12. Enthalpy of Neutralization Introduction In the course of most physical processes and chemical reactions there is a change in energy. In chemistry what is normally measured is ΔH (enthalpy
Experiment #12. Enthalpy of Neutralization Introduction In the course of most physical processes and chemical reactions there is a change in energy. In chemistry what is normally measured is ΔH (enthalpy
Standardizing a Solution of Sodium Hydroxide. Evaluation copy
 Standardizing a Solution of Sodium Hydroxide Computer 6 It is often necessary to test a solution of unknown concentration with a solution of a known, precise concentration. The process of determining the
Standardizing a Solution of Sodium Hydroxide Computer 6 It is often necessary to test a solution of unknown concentration with a solution of a known, precise concentration. The process of determining the
High School Science Chemistry Unit 12 Exemplar Lesson 02: Heat Energy in Chemical Reactions
 Science Unit: 12 Lesson: 02 Suggested Duration: 7 days High School Science Unit 12 Exemplar Lesson 02: Heat Energy in Chemical Reactions This lesson is one approach to teaching the State Standards associated
Science Unit: 12 Lesson: 02 Suggested Duration: 7 days High School Science Unit 12 Exemplar Lesson 02: Heat Energy in Chemical Reactions This lesson is one approach to teaching the State Standards associated
CHEMISTRY 130 General Chemistry I. Thermochemistry
 CHEMISTRY 130 General Chemistry I Thermochemistry The burning of a match, shown above [1], is a chemical reaction between oxygen and sulfur. [2] Intuitively, we know that this reaction releases heat enough
CHEMISTRY 130 General Chemistry I Thermochemistry The burning of a match, shown above [1], is a chemical reaction between oxygen and sulfur. [2] Intuitively, we know that this reaction releases heat enough
not to be republished NCERT MOST of the reactions are carried out at atmospheric pressure, hence THERMOCHEMICAL MEASUREMENT UNIT-3
 UNIT-3 THERMOCHEMICAL MEASUREMENT MOST of the reactions are carried out at atmospheric pressure, hence heat changes noted for these reactions are enthalpy changes. Enthalpy changes are directly related
UNIT-3 THERMOCHEMICAL MEASUREMENT MOST of the reactions are carried out at atmospheric pressure, hence heat changes noted for these reactions are enthalpy changes. Enthalpy changes are directly related
Thermochemistry. Using Heats of Reaction - Hess s Law - Standard Enthalpies of Formation - Fuels Foods, Commercial Fuels, and Rocket Fuels
 Thermochemistry Understanding Heats of Reaction - Energy and Its Units - Heat of Reaction - Enthalpy and Enthalpy Change - Thermochemical Equations - Applying Stoichiometry to Heats of Reaction - Measuring
Thermochemistry Understanding Heats of Reaction - Energy and Its Units - Heat of Reaction - Enthalpy and Enthalpy Change - Thermochemical Equations - Applying Stoichiometry to Heats of Reaction - Measuring
Warm up. 1) What is the conjugate acid of NH 3? 2) What is the conjugate base of HNO 2? 3) If the ph is 9.2, what is the [H 3 O + ], poh, and [OH - ]?
![Warm up. 1) What is the conjugate acid of NH 3? 2) What is the conjugate base of HNO 2? 3) If the ph is 9.2, what is the [H 3 O + ], poh, and [OH - ]? Warm up. 1) What is the conjugate acid of NH 3? 2) What is the conjugate base of HNO 2? 3) If the ph is 9.2, what is the [H 3 O + ], poh, and [OH - ]?](/thumbs/78/77877522.jpg) Warm up 1) What is the conjugate acid of NH 3? 2) What is the conjugate base of HNO 2? 3) If the ph is 9.2, what is the [H 3 O + ], poh, and [OH - ]? 4) What is the concentration of H 2 SO 4 if 30.1 ml
Warm up 1) What is the conjugate acid of NH 3? 2) What is the conjugate base of HNO 2? 3) If the ph is 9.2, what is the [H 3 O + ], poh, and [OH - ]? 4) What is the concentration of H 2 SO 4 if 30.1 ml
s Traditionally, we use the calorie as a unit of energy. The nutritional Calorie, Cal = 1000 cal. Kinetic Energy and Potential Energy
 AP Chemistry: Thermochemistry Lecture Outline 5.1 The Nature of Energy Thermodynamics is the study of energy and its transformations. Thermochemistry is the study of the relationships between chemical
AP Chemistry: Thermochemistry Lecture Outline 5.1 The Nature of Energy Thermodynamics is the study of energy and its transformations. Thermochemistry is the study of the relationships between chemical
Chemistry. Friday, March 30 th Monday, April 9 th, 2018
 Chemistry Friday, March 30 th Monday, April 9 th, 2018 Do-Now: BrainPOP: Heat 1. Write down today s FLT 2. Distinguish between exothermic and endothermic processes. 3. What is the specific heat of water?
Chemistry Friday, March 30 th Monday, April 9 th, 2018 Do-Now: BrainPOP: Heat 1. Write down today s FLT 2. Distinguish between exothermic and endothermic processes. 3. What is the specific heat of water?
Lab #9- Calorimetry/Thermochemistry to the Rescue
 Chesapeake Campus Chemistry 111 Laboratory Lab #9- Calorimetry/Thermochemistry to the Rescue Objectives Determine whether a reaction is endothermic or exothermic. Determine the best ionic compound of to
Chesapeake Campus Chemistry 111 Laboratory Lab #9- Calorimetry/Thermochemistry to the Rescue Objectives Determine whether a reaction is endothermic or exothermic. Determine the best ionic compound of to
Thermochemistry. Chapter Energy Storage and Reference. 4.2 Chemical Rxns. 4.3 Hess s law Overview
 Chapter 4 Thermochemistry 4.1 Energy Storage and Reference Thermochemistry is the study of the energy released when the atomic-level associations are changed. A chemical reaction (rxn) is one where these
Chapter 4 Thermochemistry 4.1 Energy Storage and Reference Thermochemistry is the study of the energy released when the atomic-level associations are changed. A chemical reaction (rxn) is one where these
Stoichiometry Rockets
 Stoichiometry Rockets The objective of this lab is to to: calculate the needed volume of fuel to react with a given volume of gas and result in a productive explosion determine the heat of the reaction
Stoichiometry Rockets The objective of this lab is to to: calculate the needed volume of fuel to react with a given volume of gas and result in a productive explosion determine the heat of the reaction
LECTURE 4 Variation of enthalpy with temperature
 LECTURE 4 Variation of enthalpy with temperature So far, we can only work at 25 C. Like c v we define a constant pressure heat capacity, c p, as the amount of heat energy needed to raise the temperature
LECTURE 4 Variation of enthalpy with temperature So far, we can only work at 25 C. Like c v we define a constant pressure heat capacity, c p, as the amount of heat energy needed to raise the temperature
Name: Chemistry 103 Laboratory University of Massachusetts Boston HEATS OF REACTION PRELAB ASSIGNMENT
 Name: Chemistry 103 Laboratory University of Massachusetts Boston HEATS OF REACTION PRELAB ASSIGNMENT Chemical and physical changes usually involve the absorption or liberation of heat, given the symbol
Name: Chemistry 103 Laboratory University of Massachusetts Boston HEATS OF REACTION PRELAB ASSIGNMENT Chemical and physical changes usually involve the absorption or liberation of heat, given the symbol
Chesapeake Campus Chemistry 111 Laboratory
 Chesapeake Campus Chemistry 111 Laboratory Objectives Calculate molar mass using the ideal gas law and laboratory data. Determine the identity of an unknown from a list of choices. Determine how sources
Chesapeake Campus Chemistry 111 Laboratory Objectives Calculate molar mass using the ideal gas law and laboratory data. Determine the identity of an unknown from a list of choices. Determine how sources
Acid-Base Titration. Evaluation copy
 Acid-Base Titration Computer 7 A titration is a process used to determine the volume of a solution that is needed to react with a given amount of another substance. In this experiment, your goal is to
Acid-Base Titration Computer 7 A titration is a process used to determine the volume of a solution that is needed to react with a given amount of another substance. In this experiment, your goal is to
Is NOT just ΔG = (-) It IS ΔG = -2070kJ which means it is really spontaneous.
 LECTURE 21. THERMODYNAMICS LET S GET QUANTITATIVE Today we will examine the quantitative side of thermodynamics, while actually coming up with numerical values for ΔH, ΔS, etc. Up to this point we have
LECTURE 21. THERMODYNAMICS LET S GET QUANTITATIVE Today we will examine the quantitative side of thermodynamics, while actually coming up with numerical values for ΔH, ΔS, etc. Up to this point we have
Energy Changes in Chemical Reactions
 Chapter Energy Changes in Chemical Reactions 10.1 Energy and Energy Changes 10.2 Introduction to Thermodynamics States and State Functions The First Law of Thermodynamics Work and Heat 10.3 Enthalpy Reactions
Chapter Energy Changes in Chemical Reactions 10.1 Energy and Energy Changes 10.2 Introduction to Thermodynamics States and State Functions The First Law of Thermodynamics Work and Heat 10.3 Enthalpy Reactions
1.4 Energetics. N Goalby chemrevise.org 1. Standard Enthalpy Change of Formation. Standard Enthalpy Change of Combustion
 1.4 Energetics Definition: Enthalpy change is the amount of heat energy taken in or given out during any change in a system provided the pressure is constant. In an exothermic change energy is transferred
1.4 Energetics Definition: Enthalpy change is the amount of heat energy taken in or given out during any change in a system provided the pressure is constant. In an exothermic change energy is transferred
Observations of Container. Hot Same Size. Hot Same Size. Hot Same Size. Observations of Container. Cold Expanded. Cold Expanded.
 Chapter 9: Phenomena Phenomena: Below is data from three different reactions carried out with three different amounts of reactants. All reactants were carried out in expandable/contractable containers
Chapter 9: Phenomena Phenomena: Below is data from three different reactions carried out with three different amounts of reactants. All reactants were carried out in expandable/contractable containers
Experiment 2 Heat of Combustion: Magnesium
 Experiment 2 Heat of Combustion: Magnesium Purpose Hess s Law states that when are going from a particular set of reactants to a particular set of products, the heat of reaction is the same whether the
Experiment 2 Heat of Combustion: Magnesium Purpose Hess s Law states that when are going from a particular set of reactants to a particular set of products, the heat of reaction is the same whether the
Chapter 5 Thermochemistry
 Chapter 5 Thermochemistry Energy -Very much a chemistry topic Every chemical change has an accompanying change of. Combustion of fossil fuels The discharging a battery Metabolism of foods If we are to
Chapter 5 Thermochemistry Energy -Very much a chemistry topic Every chemical change has an accompanying change of. Combustion of fossil fuels The discharging a battery Metabolism of foods If we are to
Sample Exercise 5.1 Describing and Calculating Energy Changes
 Sample Exercise 5.1 Describing and Calculating Energy Changes A bowler lifts a 5.4-kg (12-lb) bowling ball from ground level to a height of 1.6 m (5.2 feet) and then drops the ball back to the ground.
Sample Exercise 5.1 Describing and Calculating Energy Changes A bowler lifts a 5.4-kg (12-lb) bowling ball from ground level to a height of 1.6 m (5.2 feet) and then drops the ball back to the ground.
Thermodynamics C Test
 Northern Regional: January 19 th, 2019 Thermodynamics C Test Name(s): Team Name: School Name: Team Number: Rank: Score: Science Olympiad North Florida Regional at the University of Florida Thermodynamics
Northern Regional: January 19 th, 2019 Thermodynamics C Test Name(s): Team Name: School Name: Team Number: Rank: Score: Science Olympiad North Florida Regional at the University of Florida Thermodynamics
measure ΔT in water to get q = q surroundings and use q system = q surroundings
 example using water: Calculate the amount of energy required to heat 95.0 g of water from 22.5 C to 95.5 C. q = s m ΔT ( C (4.184 J g 1 C 1 ) (95.0 g) (73.0 = = 2.90 x 10 4 J or 29.0 kj Constant Pressure
example using water: Calculate the amount of energy required to heat 95.0 g of water from 22.5 C to 95.5 C. q = s m ΔT ( C (4.184 J g 1 C 1 ) (95.0 g) (73.0 = = 2.90 x 10 4 J or 29.0 kj Constant Pressure
Thermochemistry/Calorimetry. Determination of the enthalpy of vaporization of liquids LEC 02. What you need: What you can learn about
 LEC 02 Thermochemistry/Calorimetry Determination of the enthalpy of vaporization of liquids What you can learn about Enthalpy of vaporisation Entropy of vaporisation Trouton s rule Calorimetry Heat capacity
LEC 02 Thermochemistry/Calorimetry Determination of the enthalpy of vaporization of liquids What you can learn about Enthalpy of vaporisation Entropy of vaporisation Trouton s rule Calorimetry Heat capacity
