There is basically one simple mechanism for all electrophilic aromatic substitutions:
|
|
|
- Helen Stafford
- 6 years ago
- Views:
Transcription
1 Substitution Reactions of Aromatic Compounds Simple alkenes tend to undergo addition reactions: The elements of the reagent (HBr or Br2) are simply added to the starting material. This is called, unsurprisingly, an addition reaction. Aromatic compounds do not react in this manner; and it usually takes a catalyst to initiate reaction with halogens: What we have done in this case is substitute a bromine atom for a hydrogen atom. Hence the reaction is termed an aromatic substitution. Because the benzene ring is quite electron-rich, it almost always behaves as the nucleophile in a reaction - which means that the substitution on benzene occurs by the addition of an electrophile to benzene; thus, the reactions are termed electrophilic aromatic substitution : There is basically one simple mechanism for all electrophilic aromatic substitutions: The benzene acts as a nucleophile, attacking the electrophile with a pair of its π-electrons. This initial step destroys the aromaticity of the molecule! The resulting positive charge is delocalized over the ortho and para positions. The conjugate base of the initial electrophile then assists in removing the now extraneous proton, and restores aromaticity. Note that addition of the conjugate base to the cyclohexadienyl cation (an addition reaction) does not occur because the addition reaction is much less exothermic than the rearomatization reaction and cannot compete with it. Because all electrophilic aromatic substitutions proceed in this way, the only thing that matters is the preparation of a reactive electrophile. Why a reactive electrophile? As you can see, the first step of the reaction involves destroying aromaticity. In order to do this, there must be a 1
2 significant energetic driving force. This driving force comes in the form of a very reactive (unhappy) electrophile. How are such electrophiles generated? Halogenation As you can imagine, halogens bearing a positive charge are particularly reactive. We will focus on preparing halogen electrophiles from Br, Cl and I. Bromine: Allowing bromine to react with iron metal first generates FeBr3, which then interacts with the remaining Br2 to form a highly Lewis acidic system that is capable of reacting with weakly nucleophilic aromatic compounds: The reaction proceeds by the mechanism shown below to give brominated benzene: Chlorine: The same chemistry shown for bromine also works with chlorine togenerate Cl+. A mixture of benzene, chlorine and iron(iii)chloride yields the chlorobenzene: Iodine: It is a little more difficult to make iodine sufficiently electrophilic. For relatively activated compounds, where a mild source of I+ is required, copper salts are often used as a catalyst: Reaction then proceeds by the standard mechanism, with I+ as the electrophile, to give iodinated benzenes. What good are aromatic halides? The halogens are excellent synthetic handles they can be easily converted into other functional groups. For 2
3 example, bromobenzenes can be turned into Grignard reagents, and then reacted with aldehydes, ketones, etc... Nitration We can also make a highly electrophilic form of NO2 (from HNO3/H2SO4), the nitronium ion: Which can then react with aromatic compounds via the standard mechanism to give nitrated aromatics... Why nitrate aromatics? There are a couple of good reasons to nitrate things. The first is in the manufacture of explosives highly nitrated organic molecules are frequently used as explosives (trinitrotoluene (TNT), nitroglycerine, etc.). The second reason is that nitro-groups are generally easy to reduce to amines. And since it is nearly impossible to make an amine electrophilic (in order to add it to an aromatic ring under electrophilic aromatic substitution conditions), aromatic nitro compounds are about the only precursors to aromatic amines: 3
4 Sulfonation Just as with nitration, it is easy to make a solution of highly electrophilic SO3 (SO3/H2SO4). This solution can be used to sulfonate aromatic compounds: Why is this a useful reaction? First of all, the reaction is REVERSIBLE, especially at high temperature! Cook it up in hot AQUEOUS acid, and the SO3 group falls right off again. Second, aromatic sulfonic acids were used as the first antibiotics - the so-called sulfa drugs, such as sulfanilamide. Friedel-Crafts Reactions: This reaction comes in two flavors - alkylation and acylation. Alkylation first: The basic premise of this reaction is the electrophilic addition of alkyl groups to an aromatic ring. The general scheme: whether a true carbocation is formed depends on the nature of the R group A simple example: There are a number of drawbacks to this reaction: 1) Does not work at all on aromatic rings with de-activating groups (nitro, any carbonyl, cyano) attached. 2) Because alkyl groups are activating, over-alkylation is a significant problem. 3) Because a carbocationic intermediate is involved, rearrangements tend to take place. For example, 4
5 However, Friedel & Crafts came up with another reaction without so many drawbacks. The Friedel-Crafts acylation generally proceeds without complication (through an acylium ion): For example: Note: the only significant restriction is that we still can t have any deactivating groups on the ring. Over-acylation is not a problem because an acyl group is de-activating. Therefore only one acyl group can add! There is also no problem with rearrangements. This is a very efficient reaction! Substituent Effects Most of what we ve talked about so-far has involve the addition of a compound to an unsubstituted benzene ring. What of additions to a substituted ring? Let s summarize: 1. Activating groups Because the benzene ring s electrons are acting as the nucleophile in all of the above reactions, rings substituted with strong electron-donating groups 5
6 (particularly π-electron donating) are considered activated - they often will react without a catalyst! Some examples of activating groups are: OH, OR, NH2, NR2, Alkyl. Oxygen and nitrogen-based activating groups increase reactivity by a resonance effect: As you can see, the very nature of the activation requires ortho-para direction! Alkyl groups work somewhat differently. They are not as strong at activating the ring, and their main function is stabilizing the positive charge formed after the attack on the electrophile: 2. The Halogens: The halogens are in a class by themselves, and behave in an unusual manner. They are VERY electron withdrawing, and thus DE-ACTIVATE the ring towards electrophilic substitution. However, their multiple lone-pairs are able to stabilize the cation formed after electrophilic addition, and thus direct ortho-para: 3. Meta-Directing De-activators. Strongly electron-withdrawing groups de-activate the ring towards electrophilic substitution. Examples of such groups are: Ester (COOR), Acid (COOH), Aldehyde (CHO), Nitro (NO2), Ketone (or acyl, COR), Cyano (CN). However, with a strong enough electrophile, the rings often will react. 6
7 However, in order to avoid putting the charge that develops (after nucleophilic attack on an electrophile) on the carbon attached to the electron-withdrawing group, the incoming electrophile must attach to the meta carbons (see below). Note that the charge cannot be delocalized onto the carbon containing the acyl group (also note - the charge delocalizes over all of the ortho and para positions!) Other considerations: 1. Sterics is particularly a problem with bulky ortho-para directors - the ortho positions are blocked, forcing addition to the para positions: Multi-functional compounds: READ Sections carefully What if the ring has several substituents? Your text has a rather detailed description of what to do, so here I ll just supplement that data. 1) If the effects of two substituents point to reactivity at one particular carbon, you re in luck! That s where the electrophile will go (providing the sterics are not horrible). 2) The strongest directing group almost always prevails (e.g. OH over alkyl, NR 2 over Br, etc). The order of precedence is: a) Strong o,p directors (OR, NR 2 ) b) Alkyl groups and halogen c) All meta-directors. 4) And of course, keep a close eye on the steric environment. Birch reduction. a) Li, liquid NH3 (or Na/NH 3 ) converts benzene into 1,4-cyclohexadiene: 7
8 Na/ liq. NH 3 etc EtOH EtO-H H H etc Na H H H H EtO-H etc H H b) Alkyl and alkoxy groups prefer to end up on the C(sp2) of the cyclohexadiene product. c) CO2H group prefers to end up on the C(sp3) of the cyclohexadiene product. Reactions on the Aromatic Side Chain: Alkyl groups attached to a benzene ring are surprisingly reactive. The hydrogens on a carbon atom attached to the benzene ring are called benzylic. Notice a similarity to allylic systems? Benzylic hydrogens show a reactivity similar to allylic hydrogens: They can be brominated with NBS, for example: 8
9 Aromatic compounds with benzylic hydrogens are also susceptible to oxidative degradation: Potassium permanganate (KMnO4) decomposes these to the benzoic acid (caution -you loose the WHOLE side-chain on this one!): Note - if there are NO benzylic hydrogens (last example), the benzylic position is not oxidized (in this case, only the alcohol on the side chain gets oxidized. Hydrogenation: With a sufficiently active catalyst, it is possible to hydrogenate the aromatic system to a completely saturated system. Because aromatic chemistry is so rich & diverse, this is one method to prepare highly substituted cyclohexanes: It is also extremely easy to hydrogenate a side-chain. As a simple example, take ethyl cinnamate: Along with hydrogenation, we can also do a different kind of reduction a 9
10 deoxygenation! This reaction ONLY works on Aryl Ketones (R H), and is a PERFECT way to turn a Friedel-Crafts Acylation into an Alkylation: NOTE ALSO THAT a Clemmenson reduction using Zn(Hg), HCl or Wolf- Kishner reduction using N 2 H 4, KOH, will accomplished the same thing. End of Material for Exam 2 Palladium-mediated Reactions of Aromatic Compounds Palladium chemistry is dominated by two oxidation states 0 and +2. Typical Pd(0) sources include: Heck reaction The Heck Reaction is one of the most synthetically useful palladium-catalyzed reactions and is very efficient; it couples together a halide or triflate compound with an alkene to form a new alkene. Pd(0) is electron rich and will undergo what is termed oxidative addition reaction with suitable R X substrates, such as aryl halides (Ar X) or triflates (Ar OTf; TfO- = CF3SO3-), to form a Pd(II) complex. Note that in this reaction R -X can be aryl, vinyl, or any alkyl group without β hydrogens on an sp 3 carbon (we ll see why in a moment); and X = halide or triflate. The Pd-C sigma bond is very reactive, especially toward carbon-carbon π bonds: 10
11 Note that the choice of (R X) substrates is limited to aryl, heteroaryl, vinylic, and benzylic halides or triflates. This is because the presence of an sp 3 carbon in the β position carrying a hydrogen rapidly results in β-hydride elimination. The alkene can be mono- or disubstituted and can be electron rich, -poor, or neutral; the base need not be strong and can be Et3N, NaOAc, or aqueous Na2CO3. For example, Electron withdrawing groups such as esters promote excellent regioselectivity in favor of terminal attack: In contrast, electron donating groups, such as ethers, lead to attack at the end of the alkene substituted by oxygen to produce in this case the 1,1- disubstituted product 11
12 By the way, Pd(0) is generated in-situ in the above reaction by: Another common route to Pd(0) is: ligand H exchange Et 3 N + PdL 2 Cl 2 N Et 2 Cl PdL 2 Cl!-H elimination HPdL 2 Cl Pd 0 L 2 reductive elimination More Metal-Catalyzed Cross-Coupling Reactions i) C C bond-forming reactions The overall reaction is: R 1 X: R 1 must not have β-hydrogens that can eliminate hence aryl, vinyl, allyl, benzyl, and polyfluoroalkyl halides and triflates are typical. R 2 M: R 2 can be almost anything including examples with β-hydrogens; this is because reductive coupling is faster than β-h elimination. M = MgX, ZnX, Cu, SnR2, AlMe2, B(OR)2, etc. 12
13 R 1 X a) ArCl + RMgCl + cat. (Ph 3 P) 2 NiCl 2 ArR (Kumada coupling) b) ArX + RM + cat. Pd(0) + phosphine ligand ArR (M = SnBu 3 or SnMe 3, B(OH) 2, ZnCl) (Stille, Suzuki, Negishi couplings) c) ArX + CO + MeOH + base + cat. Pd(0) + phosphine ligand ArCO 2 Me d) ArX + RC CH + base + cat. CuI + cat. Pd(0) + phosphine ligand ArC CR (Sonogashira coupling). In general, use C(sp2) with B(OH)2, C(sp2) or C(sp3) with Sn or Zn. C(sp3) can be primary, secondary, or tertiary no rearrangements, unlike Friedel Crafts chemistry! These are the most widely used methods these days for attaching C(sp3) and C(sp2) to aromatic rings. Disconnection: Ar R Ar Br + R M. (1) If R is C C, M = H is best. (2) If R is C=C, M = B(OH)2 is best. (3) If R is C C, M = ZnCl or SnMe3 is best. Friedel Crafts alkylation is also an option (but watch for rearrangements when working forward!). (4) If in doubt, use M = SnMe3, which works with all kinds of C. (5) If R is C=O, use Friedel Crafts acylation! more on Stille Coupling Reaction the moist widely used cross-coupling reaction. For example, - the Stille reaction maybe coupled with cabonylation. For example, when the normal Stille reaction is carried out in a CO saturated solution, under 1 atm CO pressure, excellent yield of the carbonylated product can be obtained. For example, 13
14 ii) C N and C O bond-forming reactions (Buchwald Hartwig couplings) a) ArX + ROH + base + cat. Pd(0) + phosphine ligand ArOR b) ArX + R2NH + base + cat. Pd(0) + phosphine ligand ArNR2 Note: Cannot do these reactions by SN2 or SN1! Now, we have many ways to make ArCO2H! a) ArH + X2 + Lewis acid ArX + Mg ArMgX + CO2 ArCO2H. b) ArH + X2 + Lewis acid ArX + CuCN ArCN + aq. NaOH ArCO2H. c) ArH + X2 + Lewis acid ArX + CO + MeOH + cat. Pd(0) + phosphine ligand ArCO2Me ArCOOH (ester hydrolysis). d) ArH + CH3Cl + AlCl3 ArCH3 + KMnO4 ArCO2H. 14
16. Chemistry of Benzene: Electrophilic Aromatic Substitution. Based on McMurry s Organic Chemistry, 7 th edition
 16. Chemistry of Benzene: Electrophilic Aromatic Substitution Based on McMurry s Organic Chemistry, 7 th edition Substitution Reactions of Benzene and Its Derivatives Benzene is aromatic: a cyclic conjugated
16. Chemistry of Benzene: Electrophilic Aromatic Substitution Based on McMurry s Organic Chemistry, 7 th edition Substitution Reactions of Benzene and Its Derivatives Benzene is aromatic: a cyclic conjugated
Electrophilic Aromatic Substitution. Dr. Mishu Singh Department of chemistry Maharana Pratap Govt.P.G.College Hardoi
 Electrophilic Aromatic Substitution Dr. Mishu Singh Department of chemistry Maharana Pratap Govt.P.G.College Hardoi 1 Recall the electophilic addition of HBr (or Br2) to alkenes H + nu cleophile H Br H
Electrophilic Aromatic Substitution Dr. Mishu Singh Department of chemistry Maharana Pratap Govt.P.G.College Hardoi 1 Recall the electophilic addition of HBr (or Br2) to alkenes H + nu cleophile H Br H
Organic Chemistry. M. R. Naimi-Jamal. Faculty of Chemistry Iran University of Science & Technology
 Organic Chemistry M. R. Naimi-Jamal Faculty of Chemistry Iran University of Science & Technology Chapter 5-2. Chemistry of Benzene: Electrophilic Aromatic Substitution Based on McMurry s Organic Chemistry,
Organic Chemistry M. R. Naimi-Jamal Faculty of Chemistry Iran University of Science & Technology Chapter 5-2. Chemistry of Benzene: Electrophilic Aromatic Substitution Based on McMurry s Organic Chemistry,
Chemistry of Benzene: Electrophilic Aromatic Substitution
 Chemistry of Benzene: Electrophilic Aromatic Substitution Why this Chapter? Continuation of coverage of aromatic compounds in preceding chapter focus shift to understanding reactions Examine relationship
Chemistry of Benzene: Electrophilic Aromatic Substitution Why this Chapter? Continuation of coverage of aromatic compounds in preceding chapter focus shift to understanding reactions Examine relationship
12/27/2010. Chapter 15 Reactions of Aromatic Compounds
 Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen atom
Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen atom
Chapter 16 Chemistry of Benzene: Electrophilic Aromatic Substitution
 John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 16 Chemistry of Benzene: Electrophilic Aromatic Substitution Paul D. Adams University of Arkansas Substitution Reactions of Benzene and Its Derivatives
John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 16 Chemistry of Benzene: Electrophilic Aromatic Substitution Paul D. Adams University of Arkansas Substitution Reactions of Benzene and Its Derivatives
16. Chemistry of Benzene: Electrophilic Aromatic Substitution جانشینی الکتروندوستی آروماتیک شیمی آلی 2
 16. Chemistry of Benzene: Electrophilic Aromatic Substitution جانشینی الکتروندوستی آروماتیک شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based
16. Chemistry of Benzene: Electrophilic Aromatic Substitution جانشینی الکتروندوستی آروماتیک شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based
16. Chemistry of Benzene: Electrophilic Aromatic Substitution جانشینی الکتروندوستی آروماتیک شیمی آلی 2
 16. Chemistry of Benzene: Electrophilic Aromatic Substitution جانشینی الکتروندوستی آروماتیک شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based
16. Chemistry of Benzene: Electrophilic Aromatic Substitution جانشینی الکتروندوستی آروماتیک شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based
Organic Chemistry. Second Edition. Chapter 19 Aromatic Substitution Reactions. David Klein. Klein, Organic Chemistry 2e
 Organic Chemistry Second Edition David Klein Chapter 19 Aromatic Substitution Reactions Copyright 2015 John Wiley & Sons, Inc. All rights reserved. Klein, Organic Chemistry 2e 19.1 Introduction to Electrophilic
Organic Chemistry Second Edition David Klein Chapter 19 Aromatic Substitution Reactions Copyright 2015 John Wiley & Sons, Inc. All rights reserved. Klein, Organic Chemistry 2e 19.1 Introduction to Electrophilic
ORGANIC - BROWN 8E CH. 22- REACTIONS OF BENZENE AND ITS DERIVATIVES
 !! www.clutchprep.com CONCEPT: ELECTROPHILIC AROMATIC SUBSTITUTION GENERAL MECHANISM Benzene reacts with very few reagents. It DOES NOT undergo typical addition reactions. Why? If we can get benzene to
!! www.clutchprep.com CONCEPT: ELECTROPHILIC AROMATIC SUBSTITUTION GENERAL MECHANISM Benzene reacts with very few reagents. It DOES NOT undergo typical addition reactions. Why? If we can get benzene to
Ch 16 Electrophilic Aromatic Substitution
 Ch 16 Electrophilic Aromatic Substitution Mechanism - Aromatic rings typically undergo substitution, where an H is replaced with an electrophile (E+). - The rings do not typically undergo addition across
Ch 16 Electrophilic Aromatic Substitution Mechanism - Aromatic rings typically undergo substitution, where an H is replaced with an electrophile (E+). - The rings do not typically undergo addition across
Chapter 17. Reactions of Aromatic Compounds
 Chapter 17 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Although benzene s pi electrons are in a stable aromatic system, they are available to attack a strong electrophile to give
Chapter 17 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Although benzene s pi electrons are in a stable aromatic system, they are available to attack a strong electrophile to give
Chapter 15. Reactions of Aromatic Compounds. Electrophilic Aromatic Substitution on Arenes. The first step is the slow, rate-determining step
 Electrophilic Aromatic Substitution on Arenes Chapter 15 Reactions of Aromatic Compounds The characteristic reaction of aromatic rings is substitution initiated by an electrophile halogenation nitration
Electrophilic Aromatic Substitution on Arenes Chapter 15 Reactions of Aromatic Compounds The characteristic reaction of aromatic rings is substitution initiated by an electrophile halogenation nitration
Organic Chemistry, 7 L. G. Wade, Jr. Chapter , Prentice Hall
 Organic Chemistry, 7 th Edition L. G. Wade, Jr. Chapter 17 Reactions of Aromatic Compounds 2010, Prentice Hall Electrophilic Aromatic Substitution Although h benzene s pi electrons are in a stable aromatic
Organic Chemistry, 7 th Edition L. G. Wade, Jr. Chapter 17 Reactions of Aromatic Compounds 2010, Prentice Hall Electrophilic Aromatic Substitution Although h benzene s pi electrons are in a stable aromatic
Lecture Topics: I. Electrophilic Aromatic Substitution (EAS)
 Reactions of Aromatic Compounds Reading: Wade chapter 17, sections 17-1- 17-15 Study Problems: 17-44, 17-46, 17-47, 17-48, 17-51, 17-52, 17-53, 17-59, 17-61 Key Concepts and Skills: Predict and propose
Reactions of Aromatic Compounds Reading: Wade chapter 17, sections 17-1- 17-15 Study Problems: 17-44, 17-46, 17-47, 17-48, 17-51, 17-52, 17-53, 17-59, 17-61 Key Concepts and Skills: Predict and propose
Chapter 15 Reactions of Aromatic Compounds
 Chapter 15 1 Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen
Chapter 15 1 Chapter 15 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Arene (Ar-H) is the generic term for an aromatic hydrocarbon The aryl group (Ar) is derived by removal of a hydrogen
08. Chemistry of Benzene: Electrophilic Aromatic Substitution. Based on McMurry s Organic Chemistry, 6 th edition, Chapter 16
 08. Chemistry of Benzene: Electrophilic Aromatic Substitution Based on McMurry s Organic Chemistry, 6 th edition, Chapter 16 Benzene is a nucleophile p electrons make benzene nucleophile, like alkenes.
08. Chemistry of Benzene: Electrophilic Aromatic Substitution Based on McMurry s Organic Chemistry, 6 th edition, Chapter 16 Benzene is a nucleophile p electrons make benzene nucleophile, like alkenes.
11/30/ Substituent Effects in Electrophilic Substitutions. Substituent Effects in Electrophilic Substitutions
 Chapter 9 Problems: 9.1-29, 32-34, 36-37, 39-45, 48-56, 58-59, 61-69, 71-72. 9.8 Substituent effects in the electrophilic substitution of an aromatic ring Substituents affect the reactivity of the aromatic
Chapter 9 Problems: 9.1-29, 32-34, 36-37, 39-45, 48-56, 58-59, 61-69, 71-72. 9.8 Substituent effects in the electrophilic substitution of an aromatic ring Substituents affect the reactivity of the aromatic
Treatment of cyclooctatetrene with potassium gives you a dianion. Classify the starting material and product as aromatic, antiaromatic or
 Treatment of cyclooctatetrene with potassium gives you a dianion. Classify the starting material and product as aromatic, antiaromatic or nonaromatic? 1 2 Classify cyclononatetrene and it s various ions
Treatment of cyclooctatetrene with potassium gives you a dianion. Classify the starting material and product as aromatic, antiaromatic or nonaromatic? 1 2 Classify cyclononatetrene and it s various ions
Chapter 12. Reactions of Arenes: Electrophilic Aromatic Substitution. Class Notes. A. The method by which substituted benzenes are synthesized
 Chapter 12 Reactions of Arenes: Electrophilic Aromatic Substitution Chapter 12 suggested problems: 22, 23, 26, 27, 32, 33 Class Notes I. Electrophilic aromatic substitution reactions A. The method by which
Chapter 12 Reactions of Arenes: Electrophilic Aromatic Substitution Chapter 12 suggested problems: 22, 23, 26, 27, 32, 33 Class Notes I. Electrophilic aromatic substitution reactions A. The method by which
Ch.16 Chemistry of Benzene: Electrophilic Aromatic Substitution
 Ch.16 Chemistry of Benzene: Electrophilic Aromatic Substitution Electrophilic aromatic substitution: E + E + + Some electrophilic aromatic substitution: X N 2 S 3 R C R alogenation Nitration Sulfonation
Ch.16 Chemistry of Benzene: Electrophilic Aromatic Substitution Electrophilic aromatic substitution: E + E + + Some electrophilic aromatic substitution: X N 2 S 3 R C R alogenation Nitration Sulfonation
Synthesis Using Aromatic Materials
 Chapter 10 Synthesis Using Aromatic Materials ELECTROPHILIC AROMATIC SUBSTITUTION AND DIRECTED ORTHO METALATION Copyright 2018 by Nelson Education Limited 1 10.2 p Bonds Acting as Nucleophiles Copyright
Chapter 10 Synthesis Using Aromatic Materials ELECTROPHILIC AROMATIC SUBSTITUTION AND DIRECTED ORTHO METALATION Copyright 2018 by Nelson Education Limited 1 10.2 p Bonds Acting as Nucleophiles Copyright
Aromatic Compounds II
 2302272 Org Chem II Part I Lecture 2 Aromatic Compounds II Instructor: Dr. Tanatorn Khotavivattana E-mail: tanatorn.k@chula.ac.th Recommended Textbook: Chapter 17 in Organic Chemistry, 8 th Edition, L.
2302272 Org Chem II Part I Lecture 2 Aromatic Compounds II Instructor: Dr. Tanatorn Khotavivattana E-mail: tanatorn.k@chula.ac.th Recommended Textbook: Chapter 17 in Organic Chemistry, 8 th Edition, L.
Key ideas: In EAS, pi bond is Nu and undergoes addition.
 Objective 7. Apply addition and elimination concepts to predict electrophilic aromatic substitution reactions (EAS) of benzene and monosubstituted benzenes. Skills: Draw structure ID structural features
Objective 7. Apply addition and elimination concepts to predict electrophilic aromatic substitution reactions (EAS) of benzene and monosubstituted benzenes. Skills: Draw structure ID structural features
11/26/ Polycyclic Aromatic Compounds. Polycyclic Aromatic Compounds. Polycyclic Aromatic Compounds
 9.5 Polycyclic Aromatic Compounds The general concept of aromaticity can be extended to include polycyclic aromatic compounds Benzo[a]pyrene is one of the cancer-causing substances found in tobacco smoke
9.5 Polycyclic Aromatic Compounds The general concept of aromaticity can be extended to include polycyclic aromatic compounds Benzo[a]pyrene is one of the cancer-causing substances found in tobacco smoke
Chapter 17 Reactions of Aromatic Compounds. Electrophilic Aromatic Substitution
 Chapter 17 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Electrophile substitutes for a hydrogen on the benzene ring. Chapter 17: Aromatics 2-Reactions Slide 17-2 1 Mechanism Step
Chapter 17 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution Electrophile substitutes for a hydrogen on the benzene ring. Chapter 17: Aromatics 2-Reactions Slide 17-2 1 Mechanism Step
CHAPTER 16 - CHEMISTRY OF BENZENE: ELECTROPHILIC AROMATIC SUBSTITUTION
 CAPTR 16 - CMISTRY F BNZN: LCTRPILIC ARMATIC SUBSTITUTIN As stated in the previous chapter, benzene and other aromatic rings do not undergo electrophilic addition reactions of the simple alkenes but rather
CAPTR 16 - CMISTRY F BNZN: LCTRPILIC ARMATIC SUBSTITUTIN As stated in the previous chapter, benzene and other aromatic rings do not undergo electrophilic addition reactions of the simple alkenes but rather
Chapter 15. Reactions of Aromatic Compounds. 1. Electrophilic Aromatic Substitution Reactions
 hapter 15 eactions of Aromatic ompounds 1. Electrophilic Aromatic Substitution eactions v verall reaction reated by Professor William Tam & Dr. Phillis hang opyright S 3 2 S 4 S 3 2. A General Mechanism
hapter 15 eactions of Aromatic ompounds 1. Electrophilic Aromatic Substitution eactions v verall reaction reated by Professor William Tam & Dr. Phillis hang opyright S 3 2 S 4 S 3 2. A General Mechanism
Benzene and Aromatic Compounds. Chapter 15 Organic Chemistry, 8 th Edition John McMurry
 Benzene and Aromatic Compounds Chapter 15 Organic Chemistry, 8 th Edition John McMurry 1 Background Benzene (C 6 H 6 ) is the simplest aromatic hydrocarbon (or arene). Four degrees of unsaturation. It
Benzene and Aromatic Compounds Chapter 15 Organic Chemistry, 8 th Edition John McMurry 1 Background Benzene (C 6 H 6 ) is the simplest aromatic hydrocarbon (or arene). Four degrees of unsaturation. It
Chapter 17 Reactions of Aromatic Compounds
 rganic Chemistry, 6 th Edition L. G. Wade, Jr. Chapter 17 Reactions of Aromatic Compounds Jo Blackburn Richland College, Dallas, TX Dallas County Community College District 2006, Prentice all Electrophilic
rganic Chemistry, 6 th Edition L. G. Wade, Jr. Chapter 17 Reactions of Aromatic Compounds Jo Blackburn Richland College, Dallas, TX Dallas County Community College District 2006, Prentice all Electrophilic
C h a p t e r N i n e t e e n Aromatics II: Reactions of Benzene & Its Derivatives
 C h a p t e r N i n e t e e n Aromatics II: Reactions of Benzene & Its Derivatives Arenium ion from addition of tert-butyl cation to benzene (blue is δ+and red δ-) Note: Problems with italicized numbers
C h a p t e r N i n e t e e n Aromatics II: Reactions of Benzene & Its Derivatives Arenium ion from addition of tert-butyl cation to benzene (blue is δ+and red δ-) Note: Problems with italicized numbers
Benzene and Aromatic Compounds
 1 Background Benzene and Aromatic Compounds Benzene (C 6 H 6 ) is the simplest aromatic hydrocarbon (or arene). Benzene has four degrees of unsaturation, making it a highly unsaturated hydrocarbon. Whereas
1 Background Benzene and Aromatic Compounds Benzene (C 6 H 6 ) is the simplest aromatic hydrocarbon (or arene). Benzene has four degrees of unsaturation, making it a highly unsaturated hydrocarbon. Whereas
Chapter 13 Reactions of Arenes Electrophilic Aromatic Substitution
 . 13 hapter 13 eactions of Arenes lectrophilic Aromatic ubstitution lectrophiles add to aromatic rings in a fashion somewhat similar to the addition of electrophiles to alkenes. ecall: 3 4 Y 1 4 2 1 δ
. 13 hapter 13 eactions of Arenes lectrophilic Aromatic ubstitution lectrophiles add to aromatic rings in a fashion somewhat similar to the addition of electrophiles to alkenes. ecall: 3 4 Y 1 4 2 1 δ
Reactions of Aromatic Compounds. Aromatic compounds do not react like other alkenes. With an appropriate catalyst, however, benzene will react
 Reactions of Aromatic Compounds Aromatic compounds do not react like other alkenes 2 Fe 3 2 Does not form A major part of the problem for this reaction is the product has lost all aromatic stabilization,
Reactions of Aromatic Compounds Aromatic compounds do not react like other alkenes 2 Fe 3 2 Does not form A major part of the problem for this reaction is the product has lost all aromatic stabilization,
2016 Pearson Education, Inc. Isolated and Conjugated Dienes
 2016 Pearson Education, Inc. Isolated and Conjugated Dienes 2016 Pearson Education, Inc. Reactions of Isolated Dienes 2016 Pearson Education, Inc. The Mechanism Double Bonds can have Different Reactivities
2016 Pearson Education, Inc. Isolated and Conjugated Dienes 2016 Pearson Education, Inc. Reactions of Isolated Dienes 2016 Pearson Education, Inc. The Mechanism Double Bonds can have Different Reactivities
Chapter 5. Aromatic Compounds
 Chapter 5. Aromatic Compounds 5.1 Structure of Benzene: The Kekule Proposal Mid-1800s, benzene was known to have the molecular formula C 6 6. Benzene reacts with 2 in the presence of iron to give substitution
Chapter 5. Aromatic Compounds 5.1 Structure of Benzene: The Kekule Proposal Mid-1800s, benzene was known to have the molecular formula C 6 6. Benzene reacts with 2 in the presence of iron to give substitution
Benzenes & Aromatic Compounds
 Benzenes & Aromatic Compounds 1 Structure of Benzene H H C C C H C 6 H 6 H C C C H H A cyclic conjugate molecule Benzene is a colourless odourless liquid, boiling at 80 o C and melting at 5 o C. It is
Benzenes & Aromatic Compounds 1 Structure of Benzene H H C C C H C 6 H 6 H C C C H H A cyclic conjugate molecule Benzene is a colourless odourless liquid, boiling at 80 o C and melting at 5 o C. It is
CHEM Chapter 16. Chemistry of Benzene: Electrophilic Aromatic Substitution (homework) W
 CHEM 2425. Chapter 16. Chemistry of Benzene: Electrophilic Aromatic Substitution (homework) W Short Answer Exhibit 16-1 MATCH a structure or term from the following list with each description below. Place
CHEM 2425. Chapter 16. Chemistry of Benzene: Electrophilic Aromatic Substitution (homework) W Short Answer Exhibit 16-1 MATCH a structure or term from the following list with each description below. Place
Chapter 19: Benzene and Aromatic Substitution Reactions [Sections: 18.2, 18.6; ]
![Chapter 19: Benzene and Aromatic Substitution Reactions [Sections: 18.2, 18.6; ] Chapter 19: Benzene and Aromatic Substitution Reactions [Sections: 18.2, 18.6; ]](/thumbs/96/128021536.jpg) Chapter 19: Benzene and Aromatic Substitution eactions [Sections: 18.2, 18.6; 19.1-19.12] omenclature of Substituted Benzenes i. Monosubstituted Benzenes C 2 C 3 ii. Disubstituted Benzenes X X X Y Y Y
Chapter 19: Benzene and Aromatic Substitution eactions [Sections: 18.2, 18.6; 19.1-19.12] omenclature of Substituted Benzenes i. Monosubstituted Benzenes C 2 C 3 ii. Disubstituted Benzenes X X X Y Y Y
Fundamentals of Organic Chemistry
 Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 3. AROMATIC HYDROCARBONS Aromatic
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 3. AROMATIC HYDROCARBONS Aromatic
Chemistry 204: Benzene and Aromaticity
 Chemistry 204: Benzene and Aromaticity Structure of and Bonding in Benzene benzene, C 6 H 6, was first isolated in 1825 (Michael Faraday), but it was not until more than 100 years later that an adequate
Chemistry 204: Benzene and Aromaticity Structure of and Bonding in Benzene benzene, C 6 H 6, was first isolated in 1825 (Michael Faraday), but it was not until more than 100 years later that an adequate
4. AROMATIC COMPOUNDS
 BOOKS 1) Organic Chemistry Structure and Function, K. Peter C. Vollhardt, Neil Schore, 6th Edition 2) Organic Chemistry, T. W. Graham Solomons, Craig B. Fryhle 3) Organic Chemistry: A Short Course, H.
BOOKS 1) Organic Chemistry Structure and Function, K. Peter C. Vollhardt, Neil Schore, 6th Edition 2) Organic Chemistry, T. W. Graham Solomons, Craig B. Fryhle 3) Organic Chemistry: A Short Course, H.
There are two main electronic effects that substituents can exert:
 Substituent Effects There are two main electronic effects that substituents can exert: RESONANCE effects are those that occur through the π system and can be represented by resonance structures. These
Substituent Effects There are two main electronic effects that substituents can exert: RESONANCE effects are those that occur through the π system and can be represented by resonance structures. These
Chapter 16- Chemistry of Benzene: Electrophilic Aromatic Substitution
 Chapter 16- Chemistry of Benzene: Electrophilic Aromatic Substitution Ashley Piekarski, Ph.D. Substitution Reactions of Benzene and Its Derivatives Benzene is aroma%c What does aromatic mean? Reac9ons
Chapter 16- Chemistry of Benzene: Electrophilic Aromatic Substitution Ashley Piekarski, Ph.D. Substitution Reactions of Benzene and Its Derivatives Benzene is aroma%c What does aromatic mean? Reac9ons
Chapter 17 Aromati ti S u stit tit t u i tion Reactions
 Chapter 17 Aromatic Substitution Reactions 1 17.1 Mechanism for Electricphilic Aromatic Substitution Arenium ion resonance stabilization 2 Example 1. Example 2. 3 Example 2. Mechanism of the nitration
Chapter 17 Aromatic Substitution Reactions 1 17.1 Mechanism for Electricphilic Aromatic Substitution Arenium ion resonance stabilization 2 Example 1. Example 2. 3 Example 2. Mechanism of the nitration
Electrophilic Aromatic Substitution
 Chem 263 ct. 8, 2013 lectrophilic Aromatic Substitution Benzene appears to be a remarkably stable and unreactive compared to alkenes, such as cyclohexene or ethylene, or even alkanes, such as cyclohexane
Chem 263 ct. 8, 2013 lectrophilic Aromatic Substitution Benzene appears to be a remarkably stable and unreactive compared to alkenes, such as cyclohexene or ethylene, or even alkanes, such as cyclohexane
Examples of Substituted Benzenes
 Organic Chemistry 5 th Edition Paula Yurkanis Bruice Examples of Substituted Benzenes Chapter 15 Reactions of Substituted Benzenes Irene Lee Case Western Reserve University Cleveland, OH 2007, Prentice
Organic Chemistry 5 th Edition Paula Yurkanis Bruice Examples of Substituted Benzenes Chapter 15 Reactions of Substituted Benzenes Irene Lee Case Western Reserve University Cleveland, OH 2007, Prentice
SURVEY ON ARYL COMPOUNDS
 Journal of Plastic and Polymer Technology (JPPT) Vol. 1, Issue 1, Jun 2015, 111-132 TJPRC Pvt. Ltd SURVEY ON ARYL COMPOUNDS NAGHAM MAHMOOD ALJAMALI Organic Chemistry, Department of Chemistry, College of
Journal of Plastic and Polymer Technology (JPPT) Vol. 1, Issue 1, Jun 2015, 111-132 TJPRC Pvt. Ltd SURVEY ON ARYL COMPOUNDS NAGHAM MAHMOOD ALJAMALI Organic Chemistry, Department of Chemistry, College of
Chapter 17: Reactions of Aromatic Compounds
 1 Chapter 17: Reactions of Aromatic Compounds I. Introduction to Electrophilic Aromatic Substitution (EAS) A. General Mechanism II. Reactions of Electrophilic Aromatic Substitution A. Halogenation (E =
1 Chapter 17: Reactions of Aromatic Compounds I. Introduction to Electrophilic Aromatic Substitution (EAS) A. General Mechanism II. Reactions of Electrophilic Aromatic Substitution A. Halogenation (E =
Chapter 16. Chemistry of Benzene: Electrophilic Aromatic Substitution. Reactivity of Benzene
 hapter 16 hemistry of Benzene: Electrophilic Aromatic Substitution Reactivity of Benzene - stabilization due to aromaticity makes benzene significantly less reactive than isolated alkenes 2 no reaction
hapter 16 hemistry of Benzene: Electrophilic Aromatic Substitution Reactivity of Benzene - stabilization due to aromaticity makes benzene significantly less reactive than isolated alkenes 2 no reaction
CHEMISTRY 263 HOME WORK
 Lecture Topics: CHEMISTRY 263 HOME WORK Module7: Hydrogenation of Alkenes Hydrogenation - syn and anti- addition - hydrogenation of alkynes - synthesis of cis-alkenes -synthesis of trans-alkenes Text sections:
Lecture Topics: CHEMISTRY 263 HOME WORK Module7: Hydrogenation of Alkenes Hydrogenation - syn and anti- addition - hydrogenation of alkynes - synthesis of cis-alkenes -synthesis of trans-alkenes Text sections:
Module9. Nuclear Magnetic Resonance Spectroscopy Nuclear Magnetic Resonance (NMR) spectroscopy - Chemical shift - Integration of signal area
 1 CHEMISTRY 263 HOME WORK Lecture Topics: Module7. Hydrogenation of Alkenes The Function of the Catalyst - Syn and anti- addition Hydrogenation of Alkynes - Syn- addition of hydrogen: Synthesis of cis-alkenes
1 CHEMISTRY 263 HOME WORK Lecture Topics: Module7. Hydrogenation of Alkenes The Function of the Catalyst - Syn and anti- addition Hydrogenation of Alkynes - Syn- addition of hydrogen: Synthesis of cis-alkenes
Chapter 24. Amines. Based on McMurry s Organic Chemistry, 7 th edition
 Chapter 24. Amines Based on McMurry s Organic Chemistry, 7 th edition Amines Organic Nitrogen Compounds Organic derivatives of ammonia, NH 3, Nitrogen atom with a lone pair of electrons, making amines
Chapter 24. Amines Based on McMurry s Organic Chemistry, 7 th edition Amines Organic Nitrogen Compounds Organic derivatives of ammonia, NH 3, Nitrogen atom with a lone pair of electrons, making amines
Chapter 17 Reactions of Aromatic Compounds
 Chapter 17 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution o General reaction - an electrophile replaces a hydrogen Electrons of pi system attack strong electrophile, generating resonancestabilized
Chapter 17 Reactions of Aromatic Compounds Electrophilic Aromatic Substitution o General reaction - an electrophile replaces a hydrogen Electrons of pi system attack strong electrophile, generating resonancestabilized
Electrophilic Aromatic Substitution
 Chem 263 Sept 29, 2016 lectrophilic Aromatic Substitution Benzene appears to be a remarkably stable (36 kcal/mole more) and unreactive compared to alkenes, such as cyclohexene or ethylene, or even alkanes,
Chem 263 Sept 29, 2016 lectrophilic Aromatic Substitution Benzene appears to be a remarkably stable (36 kcal/mole more) and unreactive compared to alkenes, such as cyclohexene or ethylene, or even alkanes,
H 2 SO 4 Ar-NO 2 + H2O
 Phenyl group: Shorthand for phenyl: Ph, C 6 5,. An aryl group is an aromatic group: phenyl, substituted phenyl, or other aromatic group. Shorthand: Ar Generalized electrophilic aromatic substitution: E
Phenyl group: Shorthand for phenyl: Ph, C 6 5,. An aryl group is an aromatic group: phenyl, substituted phenyl, or other aromatic group. Shorthand: Ar Generalized electrophilic aromatic substitution: E
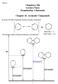
5, Organic Chemistry-II (Reaction Mechanism-1)
 Subject Chemistry Paper No and Title Module No and Title Module Tag 5, Organic Chemistry-II (Reaction Mechanism-1) 28, Arenium ion mechanism in electrophilic aromatic substitution, orientation and reactivity,
Subject Chemistry Paper No and Title Module No and Title Module Tag 5, Organic Chemistry-II (Reaction Mechanism-1) 28, Arenium ion mechanism in electrophilic aromatic substitution, orientation and reactivity,
Electrophilic Aromatic Substitution
 Lecture 12 Electrophilic Aromatic Substitution E E February 22, 2018 Electrophilic Aromatic Substitution Electrophilic aromatic substitution: a reaction in which a hydrogen atom on an aromatic ring is
Lecture 12 Electrophilic Aromatic Substitution E E February 22, 2018 Electrophilic Aromatic Substitution Electrophilic aromatic substitution: a reaction in which a hydrogen atom on an aromatic ring is
Reactions of Benzene Reactions of Benzene 1
 Reactions of Benzene Reactions of Benzene 1 2 Halogenation of Benzene v Benzene does not react with Br 2 or Cl 2 unless a Lewis acid is present (a catalytic amount is usually enough) 3 v Mechanism v Mechanism
Reactions of Benzene Reactions of Benzene 1 2 Halogenation of Benzene v Benzene does not react with Br 2 or Cl 2 unless a Lewis acid is present (a catalytic amount is usually enough) 3 v Mechanism v Mechanism
William H. Brown & Christopher S. Foote
 William. Brown & Christopher S. Foote Requests for permission to make copies of any part of the work should be mailed to:permissions Department, arcourt Brace & Company, 6277 Sea arbor Drive, Orlando,
William. Brown & Christopher S. Foote Requests for permission to make copies of any part of the work should be mailed to:permissions Department, arcourt Brace & Company, 6277 Sea arbor Drive, Orlando,
Electrophilic Aromatic Substitution (Aromatic compounds) Ar-H = aromatic compound 1. Nitration Ar-H + HNO 3, H 2 SO 4 Ar-NO 2 + H 2 O 2.
 Electrophilic Aromatic Substitution (Aromatic compounds) Ar- = aromatic compound 1. Nitration Ar- + NO 3, 2 SO 4 Ar- + 2 O 2. Sulfonation Ar- + 2 SO 4, SO 3 Ar-SO 3 + 2 O 3. alogenation Ar- + X 2, Fe Ar-X
Electrophilic Aromatic Substitution (Aromatic compounds) Ar- = aromatic compound 1. Nitration Ar- + NO 3, 2 SO 4 Ar- + 2 O 2. Sulfonation Ar- + 2 SO 4, SO 3 Ar-SO 3 + 2 O 3. alogenation Ar- + X 2, Fe Ar-X
Chapter 19: Aromatic Substitution Reactions
 Chem A225 Notes Page 52 Chapter 19: Aromatic Substitution Reactions Topic One: lectrophilic Aromatic Substitution I. Introduction to lectrophilic Aromatic Substitution (AS) A. eneral Reaction Pattern B.
Chem A225 Notes Page 52 Chapter 19: Aromatic Substitution Reactions Topic One: lectrophilic Aromatic Substitution I. Introduction to lectrophilic Aromatic Substitution (AS) A. eneral Reaction Pattern B.
BENZENE & AROMATIC COMPOUNDS
 BENZENE & AROMATIC COMPOUNDS Dr. Zainab M Almarhoon 2 Learning Objectives By the end of chapter four the students will: Understand the resonance description of structure of benzene Understand the hybridization
BENZENE & AROMATIC COMPOUNDS Dr. Zainab M Almarhoon 2 Learning Objectives By the end of chapter four the students will: Understand the resonance description of structure of benzene Understand the hybridization
The now-banned diet drug fen-phen is a mixture of two synthetic substituted benzene: fenfluramine and phentermine.
 The now-banned diet drug fen-phen is a mixture of two synthetic substituted benzene: fenfluramine and phentermine. Chemists have synthesized compounds with structures similar to adrenaline, producing amphetamine.
The now-banned diet drug fen-phen is a mixture of two synthetic substituted benzene: fenfluramine and phentermine. Chemists have synthesized compounds with structures similar to adrenaline, producing amphetamine.
15.10 Effect of Substituents on Reactivity and Orientation
 15.10 ffect of Substituents on Reactivity and Orientation Z NO 3 2 SO 4 Z Z Z + + o- p- m- Z O Me CN o(%) 40 59 30 6 17 p(%) 60 37 69
15.10 ffect of Substituents on Reactivity and Orientation Z NO 3 2 SO 4 Z Z Z + + o- p- m- Z O Me CN o(%) 40 59 30 6 17 p(%) 60 37 69
Aryl Halides. Structure
 Aryl Halides Structure Aryl halides are compounds containing halogen attached directly to an aromatic ring. They have the general formula ArX, where Ar is phenyl, substituted phenyl. X= F,Cl,Br,I An aryl
Aryl Halides Structure Aryl halides are compounds containing halogen attached directly to an aromatic ring. They have the general formula ArX, where Ar is phenyl, substituted phenyl. X= F,Cl,Br,I An aryl
Loudon Chapter 23 Review: Amines Jacquie Richardson, CU Boulder Last updated 4/22/2018
 This chapter is about the chemistry of nitrogen. We ve seen it before in several places, but now we can look at several reactions that are specific to nitrogen. Amines can be subdivided based on how many
This chapter is about the chemistry of nitrogen. We ve seen it before in several places, but now we can look at several reactions that are specific to nitrogen. Amines can be subdivided based on how many
But in organic terms: Oxidation: loss of H 2 ; addition of O or O 2 ; addition of X 2 (halogens).
 Reactions of Alcohols Alcohols are versatile organic compounds since they undergo a wide variety of transformations the majority of which are either oxidation or reduction type reactions. Normally: Oxidation
Reactions of Alcohols Alcohols are versatile organic compounds since they undergo a wide variety of transformations the majority of which are either oxidation or reduction type reactions. Normally: Oxidation
Class XII: Chemistry Chapter 13: Amines Top concepts
 Class XII: Chemistry Chapter 13: Amines Top concepts 1. Amines are regarded as derivatives of ammonia in which one, two or all three hydrogen atoms are replaced by alkyl or aryl group 2. Classification
Class XII: Chemistry Chapter 13: Amines Top concepts 1. Amines are regarded as derivatives of ammonia in which one, two or all three hydrogen atoms are replaced by alkyl or aryl group 2. Classification
Chapter 10: Carboxylic Acids and Their Derivatives
 Chapter 10: Carboxylic Acids and Their Derivatives The back of the white willow tree (Salix alba) is a source of salicylic acid which is used to make aspirin (acetylsalicylic acid) The functional group
Chapter 10: Carboxylic Acids and Their Derivatives The back of the white willow tree (Salix alba) is a source of salicylic acid which is used to make aspirin (acetylsalicylic acid) The functional group
Amines. Amines are organic compounds containing a nitrogen functionality. primary secondary tertiary quaternary
 Amines Amines are organic compounds containing a nitrogen functionality Depending upon the number of alkyl, or aryl, groups attached to nitrogen determines its classification, or order 2 primary secondary
Amines Amines are organic compounds containing a nitrogen functionality Depending upon the number of alkyl, or aryl, groups attached to nitrogen determines its classification, or order 2 primary secondary
REACTIONS OF AROMATIC COMPOUNDS
 A STUDENT SHOULD BE ABLE TO: REACTIONS OF AROMATIC COMPOUNDS 1. Predict the product(s) of Electrophilic aromatic substitution (EAS): halogenation, sulfonation, nitration, Friedel- Crafts alkylation and
A STUDENT SHOULD BE ABLE TO: REACTIONS OF AROMATIC COMPOUNDS 1. Predict the product(s) of Electrophilic aromatic substitution (EAS): halogenation, sulfonation, nitration, Friedel- Crafts alkylation and
75. A This is a Markovnikov addition reaction. In these reactions, the pielectrons in the alkene act as a nucleophile. The strongest electrophile will
 71. B SN2 stands for substitution nucleophilic bimolecular. This means that there is a bimolecular rate-determining step. Therefore, the reaction will follow second-order kinetics based on the collision
71. B SN2 stands for substitution nucleophilic bimolecular. This means that there is a bimolecular rate-determining step. Therefore, the reaction will follow second-order kinetics based on the collision
240 Chem. Aromatic Compounds. Chapter 6
 240 Chem Aromatic Compounds Chapter 6 1 The expressing aromatic compounds came to mean benzene and derivatives of benzene. Structure of Benzene: Resonance Description C 6 H 6 1.It contains a six-membered
240 Chem Aromatic Compounds Chapter 6 1 The expressing aromatic compounds came to mean benzene and derivatives of benzene. Structure of Benzene: Resonance Description C 6 H 6 1.It contains a six-membered
Synthesis of Nitriles a. dehydration of 1 amides using POCl 3 : b. SN2 reaction of cyanide ion on halides:
 I. Nitriles Nitriles consist of the CN functional group, and are linear with sp hybridization on C and N. Nitriles are non-basic at nitrogen, since the lone pair exists in an sp orbital (50% s character
I. Nitriles Nitriles consist of the CN functional group, and are linear with sp hybridization on C and N. Nitriles are non-basic at nitrogen, since the lone pair exists in an sp orbital (50% s character
Mechanisms. . CCl2 F + Cl.
 Mechanisms 1) Free radical substitution Alkane à halogenoalkane Initiation: Propagation: Termination: Overall: 2) Ozone depletion UV light breaks the C Cl bond releasing chlorine radical CFCl 3 F à. CCl2
Mechanisms 1) Free radical substitution Alkane à halogenoalkane Initiation: Propagation: Termination: Overall: 2) Ozone depletion UV light breaks the C Cl bond releasing chlorine radical CFCl 3 F à. CCl2
ALCOHOLS AND PHENOLS
 ALCOHOLS AND PHENOLS ALCOHOLS AND PHENOLS Alcohols contain an OH group connected to a a saturated C (sp3) They are important solvents and synthesis intermediates Phenols contain an OH group connected to
ALCOHOLS AND PHENOLS ALCOHOLS AND PHENOLS Alcohols contain an OH group connected to a a saturated C (sp3) They are important solvents and synthesis intermediates Phenols contain an OH group connected to
Organic Chemistry Lecture 2 - Hydrocarbons, Alcohols, Substitutions
 ALKANES Water-insoluble, low density C-C single bonds Higher MW -> higher BP, higher MP Branching -> lower BP, higher MP Forms cycloalkanes which can have ring strain Cyclohexane: chair vs. boat configuration
ALKANES Water-insoluble, low density C-C single bonds Higher MW -> higher BP, higher MP Branching -> lower BP, higher MP Forms cycloalkanes which can have ring strain Cyclohexane: chair vs. boat configuration
Ch 19 Aldehydes and Ketones
 Ch 19 Aldehydes and Ketones Aldehydes (RCHO), with the exception of formaldehyde (H 2 CO), are compounds with both an H and an organic group attached to a carbonyl. Ketones (R 2 CO) are compounds with
Ch 19 Aldehydes and Ketones Aldehydes (RCHO), with the exception of formaldehyde (H 2 CO), are compounds with both an H and an organic group attached to a carbonyl. Ketones (R 2 CO) are compounds with
Learning Guide for Chapter 18 - Aromatic Compounds II
 Learning Guide for Chapter 18 Aromatic Compounds. lectrophilic aromatic substitution ntroduction Mechanism Reagents and Products lectrophiles ffects of stituents FriedelCrafts alkylation and acylation
Learning Guide for Chapter 18 Aromatic Compounds. lectrophilic aromatic substitution ntroduction Mechanism Reagents and Products lectrophiles ffects of stituents FriedelCrafts alkylation and acylation
Lecture 27 Organic Chemistry 1
 CHEM 232 rganic Chemistry I at Chicago Lecture 27 rganic Chemistry 1 Professor Duncan Wardrop April 20, 2010 1 Self Test Question Nitrosonium (not nitronium) cations can be generated by treating sodium
CHEM 232 rganic Chemistry I at Chicago Lecture 27 rganic Chemistry 1 Professor Duncan Wardrop April 20, 2010 1 Self Test Question Nitrosonium (not nitronium) cations can be generated by treating sodium
CHEM 242 REACTIONS OF ARENES: CHAP 12 ASSIGN ELECTROPHILIC AROMATIC SUBSTITUTION A B C D E
 CHEM 242 REACTIONS OF ARENES: CHAP 12 ASSIGN ELECTROPHILIC AROMATIC SUBSTITUTION 1. Consider carefully the mechanism of the following electrophilic aromatic substitution reaction and indicate which of
CHEM 242 REACTIONS OF ARENES: CHAP 12 ASSIGN ELECTROPHILIC AROMATIC SUBSTITUTION 1. Consider carefully the mechanism of the following electrophilic aromatic substitution reaction and indicate which of
Chapter 4: Aromatic Compounds. Bitter almonds are the source of the aromatic compound benzaldehyde
 Chapter 4: Aromatic Compounds Bitter almonds are the source of the aromatic compound benzaldehyde Sources of Benzene Benzene, C 6 H 6, is the parent hydrocarbon of the especially stable compounds known
Chapter 4: Aromatic Compounds Bitter almonds are the source of the aromatic compound benzaldehyde Sources of Benzene Benzene, C 6 H 6, is the parent hydrocarbon of the especially stable compounds known
Ch 20 Carboxylic Acids and Nitriles
 Ch 20 Carboxylic Acids and Nitriles Carboxylic Acids (RCO 2 H) are compounds with an OH attached to a carbonyl. Nitriles (RC N) are compounds a carbon-nitrogen triple bond. Naming Carboxylic Acids 1. Replace
Ch 20 Carboxylic Acids and Nitriles Carboxylic Acids (RCO 2 H) are compounds with an OH attached to a carbonyl. Nitriles (RC N) are compounds a carbon-nitrogen triple bond. Naming Carboxylic Acids 1. Replace
CHEMISTRY. Module No and Title Module-, Electrophilic Aromatic Substitution: The ortho/para ipso attack, orientation in other ring systems.
 Subject Chemistry Paper No and Title Paper-5, Organic Chemistry-II Module No and Title Module-, Electrophilic Aromatic Substitution: The ortho/para Module Tag CHE_P5_M29 TABLE OF CONTENTS 1. Learning Outcomes
Subject Chemistry Paper No and Title Paper-5, Organic Chemistry-II Module No and Title Module-, Electrophilic Aromatic Substitution: The ortho/para Module Tag CHE_P5_M29 TABLE OF CONTENTS 1. Learning Outcomes
More Nomenclature: Common Names for Selected Aromatic Groups. Aryl = Ar = aromatic group. It is a broad term, and includes any aromatic rings.
 More Nomenclature: Common Names for Selected Aromatic Groups Phenyl group = or Ph = C 6 H 5 = Aryl = Ar = aromatic group. It is a broad term, and includes any aromatic rings. Benzyl = Bn = It has a -CH
More Nomenclature: Common Names for Selected Aromatic Groups Phenyl group = or Ph = C 6 H 5 = Aryl = Ar = aromatic group. It is a broad term, and includes any aromatic rings. Benzyl = Bn = It has a -CH
MCAT Organic Chemistry Problem Drill 10: Aldehydes and Ketones
 MCAT rganic Chemistry Problem Drill 10: Aldehydes and Ketones Question No. 1 of 10 Question 1. Which of the following is not a physical property of aldehydes and ketones? Question #01 (A) Hydrogen bonding
MCAT rganic Chemistry Problem Drill 10: Aldehydes and Ketones Question No. 1 of 10 Question 1. Which of the following is not a physical property of aldehydes and ketones? Question #01 (A) Hydrogen bonding
Lecture 3: Aldehydes and ketones
 Lecture 3: Aldehydes and ketones I want to start by talking about the mechanism of hydroboration/ oxidation, which is a way to get alcohols from alkenes. This gives the anti-markovnikov product, primarily
Lecture 3: Aldehydes and ketones I want to start by talking about the mechanism of hydroboration/ oxidation, which is a way to get alcohols from alkenes. This gives the anti-markovnikov product, primarily
Chapter 16: Aromatic Compounds
 Chamras Chemistry 106 Lecture otes xamination 2 Materials Chapter 16: Aromatic Compounds Benzene, the Most Commonly Known Aromatic Compound: The aromatic nature of benzene stabilizes it 36 kcal.mol 1.
Chamras Chemistry 106 Lecture otes xamination 2 Materials Chapter 16: Aromatic Compounds Benzene, the Most Commonly Known Aromatic Compound: The aromatic nature of benzene stabilizes it 36 kcal.mol 1.
I5 ELECTROPHILIC SUBSTITUTIONS OF
 Section I Aromatic chemistry I5 ELECTPILIC SUBSTITUTINS F MN-SUBSTITUTED AMATIC INGS Key Notes ortho, meta and para substitution Substituent effect eaction profile Activating groups inductive o/p Deactivating
Section I Aromatic chemistry I5 ELECTPILIC SUBSTITUTINS F MN-SUBSTITUTED AMATIC INGS Key Notes ortho, meta and para substitution Substituent effect eaction profile Activating groups inductive o/p Deactivating
Chapter 9 Aldehydes and Ketones Excluded Sections:
 Chapter 9 Aldehydes and Ketones Excluded Sections: 9.14-9.19 Aldehydes and ketones are found in many fragrant odors of many fruits, fine perfumes, hormones etc. some examples are listed below. Aldehydes
Chapter 9 Aldehydes and Ketones Excluded Sections: 9.14-9.19 Aldehydes and ketones are found in many fragrant odors of many fruits, fine perfumes, hormones etc. some examples are listed below. Aldehydes
BENZENE AND AROMATIC COMPOUNDS
 BENZENE AND AROMATIC COMPOUNDS The discovery of benzene: 1825 - Michael Faraday, empirical formula of C 1834 - Eilhard Mitscherlich synthesized benzin from gum benzoin, empirical formula C Aromatic The
BENZENE AND AROMATIC COMPOUNDS The discovery of benzene: 1825 - Michael Faraday, empirical formula of C 1834 - Eilhard Mitscherlich synthesized benzin from gum benzoin, empirical formula C Aromatic The
Frost Circles a Great Trick
 Aromatics Frost Circles a Great Trick Inscribe a polygon of the same number of sides as the ring to be examined such that one of the vertices is at the bottom of the ring The relative energies of the MOs
Aromatics Frost Circles a Great Trick Inscribe a polygon of the same number of sides as the ring to be examined such that one of the vertices is at the bottom of the ring The relative energies of the MOs
Alkyl phenyl ketones are usually named by adding the acyl group as prefix to phenone.
 Aldehydes, Ketones and Carboxylic Acids Nomenclature of aldehydes and ketones Aldehydes: Often called by their common names instead of IUPAC names. Ketones: Derived by naming two alkyl or aryl groups bonded
Aldehydes, Ketones and Carboxylic Acids Nomenclature of aldehydes and ketones Aldehydes: Often called by their common names instead of IUPAC names. Ketones: Derived by naming two alkyl or aryl groups bonded
AMINES. 3. Secondary When two hydrogen atoms are replaced by two alkyl or aryl groups.
 AMINES Amine may be regarded as derivative of ammonia formed by replacement of one or more hydrogen atoms by corresponding number of alkyl or aryl group CLASSIFICATION 1. Ammonia 2. Primary amine 3. Secondary
AMINES Amine may be regarded as derivative of ammonia formed by replacement of one or more hydrogen atoms by corresponding number of alkyl or aryl group CLASSIFICATION 1. Ammonia 2. Primary amine 3. Secondary
2. Examining the infrared spectrum of a compound allows us to:
 CHEM 204 2010 Ass. 1 Problem 1. The amount of energy in infrared light corresponds to: a. the amount of energy needed to promote one electron from a bonding to an antibonding molecular orbital b. the amount
CHEM 204 2010 Ass. 1 Problem 1. The amount of energy in infrared light corresponds to: a. the amount of energy needed to promote one electron from a bonding to an antibonding molecular orbital b. the amount
Chapter 23 Phenols CH. 23. Nomenclature. The OH group takes precedence as the parent phenol.
 CH. 23 Chapter 23 Phenols Nomenclature The OH group takes precedence as the parent phenol. Carboxyl and acyl groups take precedence over the OH group. The OH group is a strong electron-donating group through
CH. 23 Chapter 23 Phenols Nomenclature The OH group takes precedence as the parent phenol. Carboxyl and acyl groups take precedence over the OH group. The OH group is a strong electron-donating group through
Carboxylic Acids and Nitriles
 Carboxylic Acids and Nitriles Why this Chapter? Carboxylic acids present in many industrial processes and most biological processes They are the starting materials from which other acyl derivatives are
Carboxylic Acids and Nitriles Why this Chapter? Carboxylic acids present in many industrial processes and most biological processes They are the starting materials from which other acyl derivatives are
Chapter 25: The Chemistry of Life: Organic and Biological Chemistry
 Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
The C-X bond gets longerand weakergoing down the periodic table.
 Chapter 10: Organohalides Organic molecules containing halogen atoms (X) bonded to carbon are useful compounds in synthesis and on their own. 10.2 Structure of alkyl halides The C-X bond gets longerand
Chapter 10: Organohalides Organic molecules containing halogen atoms (X) bonded to carbon are useful compounds in synthesis and on their own. 10.2 Structure of alkyl halides The C-X bond gets longerand
