Solutes & Water Chapter 4
|
|
|
- Martin Fletcher
- 5 years ago
- Views:
Transcription
1 4 th Lecture, 23 Jan 2009 Vertebrate Physiology ECOL 437 (MCB/VetSci 437) Univ. of Arizona, spring 2009 Solutes & Water Chapter 4 Kevin Bonine & Kevin Oh 1. Finish Molecules, Membranes, etc. 2. Solutes & Water 1 Housekeeping, 23 January 2009 Readings Today: Chapters 2 & 4 Mon 26 Jan: Chapters 4 & 11 Wed 28 Jan: Chapter 11 LAB Wed 28 Jan: Bisbal & Specker 1991 (see website for links to papers) Fri 30 Jan: Chs 11 & 12 Lab discussion leaders: 04 Feb 1pm Carlos?, Michelle 3pm Maria, Jay Lab discussion leaders: 28 Jan 1pm Steve, Ami 3pm Ty, George 2 1
2 TODAY 3 TODAY! 4 2
3 Vertebrate Physiology 437 Chapter 4 Movement of Solutes and Water 5 What are the different ways to get substances across membranes? 6 3
4 Movement Across Membranes 1. Passive Diffusion (= simple diffusion) 2. Passive Transport (= facilitated diffusion) 3. Active Transport Transport (pore or carrier) may be highly selective 417 Randall et al How does a channel act selectively? 8 4
5 Ion Channels Ion selectivity Leaky channels (e.g., K) Voltagegated channels (e.g., Na, K, Ca) Ligandgated channels Mechanically activated channels, etc. charge ease of dehydration size 429 Randall et al Movement Across Membranes 1. Passive Diffusion (= simple diffusion) nonpolar/nonelectrolyte lipid soluble (steroid hormones) few H bonds ~smaller size rate depends on [ ] gradient No saturation 417 Randall et al
6 Diffusion Fick Equation: J = D C 1 C 2 X J = net rate of diffusion per unit area D = diffusion coefficient (depends on permeability and Temp) C 1 C 2 = [gradient] X = distance separating C1 from C2 11 Origin of Circulatory Systems! Hill et al
7 Movement Across Membranes 1. Passive Diffusion (= simple diffusion) 2. Passive Transport (= facilitated diffusion) Down Electrochemical gradient A. pore B. carrier mediated pores show some saturation, but not as much as carriers Randall et al Movement Across Membranes 1. Passive Diffusion (= simple diffusion) 2. Passive Transport (= facilitated diffusion) 3. Active Transport (1 o, 2 o ) 417 Randall et al Randall et al Na/K ATPase Pump 14 7
8 El Nino lack of food Starvation Animals may lose 15% body length bone absorption Only adult vertebrate known to regularly shrink (astronauts?) Largest animals die natural selection vs. sexual selection (Most efficient salt glands known in reptiles) Galapagos Marine Iguana (Iguanidae) high cost of salt excretion Amblyrhynchus cristatus 17 K.E.Bonine 2004 Reptilian Salt Glands 18 8
9 In Freshwater Hill et al (See Bisbal & Specker 1991) Hill et al
10 Membrane Selectivity (Channels) 429 Randall et al Charge, ease of dehydration, size Diffusion nonpolar/nonelectrolyte lipophilic few H bonds smaller size Transport rates depend on 1. electrochemical gradient 2. # carriers/pores Randall et al Movement Across Membranes 420 Randall et al Randall et al How is this related to the early test for diabetes?? 22 10
11 Movement Across Membranes 422 Randall et al Movement Across Membranes 426 Randall et al How would you describe this movement across membrane? 24 11
12 424 Randall et al Na/K ATPase Pump 25 Movement Across Membranes How does glucose cross membranes? Most tissues: Passive transport down [ ] gradient via carrier proteins In gut: 2 o active to move Glu against [ ] gradient into blood from food 26 12
13 Hill et al Leinhard et al
14 Osmotic Properties of Cells and Relative Ion Concentrations K Ca Ca 412 Randall et al Na K Cl Cl Na Permeabilities K >> Na ; Cl A (includes proteins, phosphate groups, etc.) 29 Electrogenic vs. Electroneutral? Hill et al
15 Ion Gradients as an Energy Source CET example: Metabolism Electron Transport Chain ATP creation energy currency Glucose > Glycolysis > Pyruvic Acid > Krebs Cycle > NADH2, FADH2 > Electron Transport Chain > Oxygen final electron acceptor Result: H2O, Proton Gradient > Oxidative Phosphorylation > ATP 1 Move molecules (see p.170 Hill 2008) 2 Electrical Signalling 3 Chemiosmotic Energy Transduction 427 Randall et al Randall et al Just add water How does water move across membranes? Aquaporins (Diffusion) 32 15
16 Movement Across Membranes Iso Hypo osmotic Hyper In specific tissues and cells: Iso Hypo tonic Hyper Movement of water 414 Randall et al Osmotic Properties of Cells and Relative Ion Concentrations K Ca Ca Na K Cl Cl Na 412 Randall et al Hypertonic Cell Contents 416 Randall et al
17 Colligative Properties Osmotic Pressure Freezing Point Water Vapor Pressure (boiling point; evaporation) Hill et al Hill et al
18 Osmolarity 6 x osmolar solution (Osm) has 1 Avogadro s number of dissolved particles/ liter solvent 1 milliosmolar solution (mosm) has Avogadro s number of dissolved particles/ liter solvent 37 What osmolarity do you get if you add 6 x molecules of glucose to a liter of water? What osmolarity do you get if you add 6 x molecules of table salt to a liter of water? NaCl (strong electrolyte) 38 18
19 Osmotic Pressure Vs. Hydrostatic Pressure Hill et al Difference in osmotic potential Rate of Osmosis = K П 1 П 2 X Proportionality Coefficient (~ permeability and temp) Distance between solutions 40 19
20 Electrochemical equilibrium Fig 3.6, Hill et al Movement Across Membranes Electrochemical Gradient 1. Electrical gradient 2. Concentration gradient K K Electrochemical equilibrium Equilibrium potential (E x in mv) Na when [X] gradient = electrical gradient Na 42 20
21 Equilibrium potential (E x in mv) Every ion s goal in life is to make the membrane potential equal its own equilibrium potential (E x in mv) 43 21
Lecture 04, 01 Sept 2005 Chapters 2, 3, and 10. Vertebrate Physiology ECOL 437 (aka MCB 437, VetSci 437) University of Arizona Fall 2005
 Lecture 04, 01 Sept 2005 Chapters 2, 3, and 10 Vertebrate Physiology ECOL 437 (aka MCB 437, VetSci 437) University of Arizona Fall 2005 instr: Kevin Bonine t.a.: Kristen Potter 1 Vertebrate Physiology
Lecture 04, 01 Sept 2005 Chapters 2, 3, and 10 Vertebrate Physiology ECOL 437 (aka MCB 437, VetSci 437) University of Arizona Fall 2005 instr: Kevin Bonine t.a.: Kristen Potter 1 Vertebrate Physiology
Lecture 04, 01 Sept 2005 Chapters 2, 3, and 10. Vertebrate Physiology ECOL 437 (aka MCB 437, VetSci 437) University of Arizona Fall 2005
 Lecture 04, 01 Sept 2005 Chapters 2, 3, and 10 Vertebrate Physiology ECOL 437 (aka MCB 437, VetSci 437) University of Arizona Fall 2005 instr: Kevin Bonine t.a.: Kristen Potter Vertebrate Physiology 437
Lecture 04, 01 Sept 2005 Chapters 2, 3, and 10 Vertebrate Physiology ECOL 437 (aka MCB 437, VetSci 437) University of Arizona Fall 2005 instr: Kevin Bonine t.a.: Kristen Potter Vertebrate Physiology 437
Lecture 04, 04 Sept 2003 Chapters 4 and 5. Vertebrate Physiology ECOL 437 University of Arizona Fall instr: Kevin Bonine t.a.
 Lecture 04, 04 Sept 2003 Chapters 4 and 5 Vertebrate Physiology ECOL 437 University of Arizona Fall 2003 instr: Kevin Bonine t.a.: Bret Pasch Vertebrate Physiology 437 1. Membranes (CH4) 2. Nervous System
Lecture 04, 04 Sept 2003 Chapters 4 and 5 Vertebrate Physiology ECOL 437 University of Arizona Fall 2003 instr: Kevin Bonine t.a.: Bret Pasch Vertebrate Physiology 437 1. Membranes (CH4) 2. Nervous System
What are neurons for?
 5 th & 6 th Lectures Mon 26 & Wed 28 Jan 2009 Vertebrate Physiology ECOL 437 (MCB/VetSci 437) Univ. of Arizona, spring 2009 Kevin Bonine & Kevin Oh 1. Finish Solutes Water 2. Neurons Neurons Chapter 11
5 th & 6 th Lectures Mon 26 & Wed 28 Jan 2009 Vertebrate Physiology ECOL 437 (MCB/VetSci 437) Univ. of Arizona, spring 2009 Kevin Bonine & Kevin Oh 1. Finish Solutes Water 2. Neurons Neurons Chapter 11
Housekeeping, 26 January 2009
 5 th & 6 th Lectures Mon 26 & Wed 28 Jan 2009 Vertebrate Physiology ECOL 437 (MCB/VetSci 437) Univ. of Arizona, spring 2009 Neurons Chapter 11 Kevin Bonine & Kevin Oh 1. Finish Solutes + Water 2. Neurons
5 th & 6 th Lectures Mon 26 & Wed 28 Jan 2009 Vertebrate Physiology ECOL 437 (MCB/VetSci 437) Univ. of Arizona, spring 2009 Neurons Chapter 11 Kevin Bonine & Kevin Oh 1. Finish Solutes + Water 2. Neurons
Neurons. 5 th & 6 th Lectures Mon 26 & Wed 28 Jan Finish Solutes + Water. 2. Neurons. Chapter 11
 5 th & 6 th Lectures Mon 26 & Wed 28 Jan 2009 Vertebrate Physiology ECOL 437 (MCB/VetSci 437) Univ. of Arizona, spring 2009 Neurons Chapter 11 Kevin Bonine & Kevin Oh 1. Finish Solutes + Water 2. Neurons
5 th & 6 th Lectures Mon 26 & Wed 28 Jan 2009 Vertebrate Physiology ECOL 437 (MCB/VetSci 437) Univ. of Arizona, spring 2009 Neurons Chapter 11 Kevin Bonine & Kevin Oh 1. Finish Solutes + Water 2. Neurons
Chapter 3 Part 1! 10 th ed.: pp ! 11 th ed.: pp !! Cellular Transport Mechanisms! The Cell Cycle!
 Chapter 3 Part 1! 10 th ed.: pp. 87 105! 11 th ed.: pp. 90 107!! Cellular Transport Mechanisms! The Cell Cycle! Transport Processes: Passive and Active (1 of 2)! 1. Passive transport! Does not use ATP!
Chapter 3 Part 1! 10 th ed.: pp. 87 105! 11 th ed.: pp. 90 107!! Cellular Transport Mechanisms! The Cell Cycle! Transport Processes: Passive and Active (1 of 2)! 1. Passive transport! Does not use ATP!
Chapter 3 Part 1! 10 th ed.: pp ! 11 th ed.: pp !! Cellular Transport Mechanisms! The Cell Cycle!
 Chapter 3 Part 1! 10 th ed.: pp. 87 105! 11 th ed.: pp. 90 107!! Cellular Transport Mechanisms! The Cell Cycle! Transport Processes: Passive and Active (1 of 2)! 1. Passive transport! Does not use ATP!
Chapter 3 Part 1! 10 th ed.: pp. 87 105! 11 th ed.: pp. 90 107!! Cellular Transport Mechanisms! The Cell Cycle! Transport Processes: Passive and Active (1 of 2)! 1. Passive transport! Does not use ATP!
The following question(s) were incorrectly answered.
 Name: Marcie Joseph Module: Cells & chemistry Test topic/animation: My animations/all animations Test type: Multiple choice Score: 48/50 Percent correct: 96% The following question(s) were incorrectly
Name: Marcie Joseph Module: Cells & chemistry Test topic/animation: My animations/all animations Test type: Multiple choice Score: 48/50 Percent correct: 96% The following question(s) were incorrectly
1. Neurons & Action Potentials
 Lecture 6, 30 Jan 2008 Vertebrate Physiology ECOL 437 (MCB/VetSci 437) Univ. of Arizona, spring 2008 Kevin Bonine & Kevin Oh 1. Intro Nervous System Fxn (slides 32-60 from Mon 28 Jan; Ch10) 2. Neurons
Lecture 6, 30 Jan 2008 Vertebrate Physiology ECOL 437 (MCB/VetSci 437) Univ. of Arizona, spring 2008 Kevin Bonine & Kevin Oh 1. Intro Nervous System Fxn (slides 32-60 from Mon 28 Jan; Ch10) 2. Neurons
Membranes 2: Transportation
 Membranes 2: Transportation Steven E. Massey, Ph.D. Associate Professor Bioinformatics Department of Biology University of Puerto Rico Río Piedras Office & Lab: NCN#343B Tel: 787-764-0000 ext. 7798 E-mail:
Membranes 2: Transportation Steven E. Massey, Ph.D. Associate Professor Bioinformatics Department of Biology University of Puerto Rico Río Piedras Office & Lab: NCN#343B Tel: 787-764-0000 ext. 7798 E-mail:
Lecture 3 13/11/2018
 Lecture 3 13/11/2018 1 Plasma membrane ALL cells have a cell membrane made of proteins and lipids. protein channel Cell Membrane Layer 1 Layer 2 lipid bilayer protein pump Lipid bilayer allows water, carbon
Lecture 3 13/11/2018 1 Plasma membrane ALL cells have a cell membrane made of proteins and lipids. protein channel Cell Membrane Layer 1 Layer 2 lipid bilayer protein pump Lipid bilayer allows water, carbon
Chapter 7-3 Cells and Their Environment
 Chapter 7-3 Cells and Their Environment 7-3 Passive Transport Passive transport-the movement of substances across the cell membrane without using NRG Concentration Gradient-difference in concentration
Chapter 7-3 Cells and Their Environment 7-3 Passive Transport Passive transport-the movement of substances across the cell membrane without using NRG Concentration Gradient-difference in concentration
Transporters and Membrane Motors Nov 15, 2007
 BtuB OM vitamin B12 transporter F O F 1 ATP synthase Human multiple drug resistance transporter P-glycoprotein Transporters and Membrane Motors Nov 15, 2007 Transport and membrane motors Concentrations
BtuB OM vitamin B12 transporter F O F 1 ATP synthase Human multiple drug resistance transporter P-glycoprotein Transporters and Membrane Motors Nov 15, 2007 Transport and membrane motors Concentrations
Upcoming Events of Interest
 Ecosystems 31 January 2007 9th class meeting Sky Islands El Nino Ecosystems 31 Jan READINGS: Sky Islands, El Nino (on website) EO Wilson book chapter Friday 02 Feb: EO Wilson book chapter (same as above)
Ecosystems 31 January 2007 9th class meeting Sky Islands El Nino Ecosystems 31 Jan READINGS: Sky Islands, El Nino (on website) EO Wilson book chapter Friday 02 Feb: EO Wilson book chapter (same as above)
Electrical Properties of the Membrane
 BIOE 2520 Electrical Properties of the Membrane Reading: Chapter 11 of Alberts et al. Stephen Smith, Ph.D. 433 Biotech Center shs46@pitt.edu Permeability of Lipid membrane Lipid bilayer is virtually impermeable
BIOE 2520 Electrical Properties of the Membrane Reading: Chapter 11 of Alberts et al. Stephen Smith, Ph.D. 433 Biotech Center shs46@pitt.edu Permeability of Lipid membrane Lipid bilayer is virtually impermeable
9.1 Mixtures and Solutions
 9.1 Mixtures and Solutions Heterogeneous mixture: : A nonuniform mixture that has regions of different composition. Homogeneous mixture: : A uniform mixture that has the same composition throughout. Solution:
9.1 Mixtures and Solutions Heterogeneous mixture: : A nonuniform mixture that has regions of different composition. Homogeneous mixture: : A uniform mixture that has the same composition throughout. Solution:
Slide 1. Slide 2. Slide 3. Colligative Properties. Compounds in Aqueous Solution. Rules for Net Ionic Equations. Rule
 Slide 1 Colligative Properties Slide 2 Compounds in Aqueous Solution Dissociation - The separation of ions that occurs when an ionic compound dissolves Precipitation Reactions - A chemical reaction in
Slide 1 Colligative Properties Slide 2 Compounds in Aqueous Solution Dissociation - The separation of ions that occurs when an ionic compound dissolves Precipitation Reactions - A chemical reaction in
Lecture 02, 28 Aug 2003 Chapters 2, 3, and 4. Vertebrate Physiology ECOL 437 University of Arizona Fall instr: Kevin Bonine t.a.
 Lecture 02, 28 Aug 2003 Chapters 2, 3, and 4 Vertebrate Physiology ECOL 437 University of Arizona Fall 2003 instr: Kevin Bonine t.a.: Bret Pasch Vertebrate Physiology 437 1. 3x5 cards, Syllabus, Photos
Lecture 02, 28 Aug 2003 Chapters 2, 3, and 4 Vertebrate Physiology ECOL 437 University of Arizona Fall 2003 instr: Kevin Bonine t.a.: Bret Pasch Vertebrate Physiology 437 1. 3x5 cards, Syllabus, Photos
Ch. 3: Cells & Their Environment
 Ch. 3: Cells & Their Environment OBJECTIVES: 1. Understand cell membrane permeability 2. To recognize different types of cellular transport (passive vs active) 3. To understand membrane potential and action
Ch. 3: Cells & Their Environment OBJECTIVES: 1. Understand cell membrane permeability 2. To recognize different types of cellular transport (passive vs active) 3. To understand membrane potential and action
Membrane transport 1. Summary
 Membrane transport 1. Summary A. Simple diffusion 1) Diffusion by electrochemical gradient no energy required 2) No channel or carrier (or transporter protein) is needed B. Passive transport (= Facilitated
Membrane transport 1. Summary A. Simple diffusion 1) Diffusion by electrochemical gradient no energy required 2) No channel or carrier (or transporter protein) is needed B. Passive transport (= Facilitated
Cellular Transport and the Cell Cycle
 Chapter 8 Cellular Transport and the Cell Cycle In your textbook, read about osmosis: diffusion of water. Reinforcement and Study Guide Section 8.1 Cellular Transport Complete the table by checking the
Chapter 8 Cellular Transport and the Cell Cycle In your textbook, read about osmosis: diffusion of water. Reinforcement and Study Guide Section 8.1 Cellular Transport Complete the table by checking the
Cellular Respiration. The mechanism of creating cellular energy. Thursday, 11 October, 12
 Cellular Respiration The mechanism of creating cellular energy What do we know?? What do we know?? Grade 5 - Food --> Energy What do we know?? Grade 5 - Food --> Energy Grade 10 - glu. + O2 --> CO2 + H20
Cellular Respiration The mechanism of creating cellular energy What do we know?? What do we know?? Grade 5 - Food --> Energy What do we know?? Grade 5 - Food --> Energy Grade 10 - glu. + O2 --> CO2 + H20
Movement across the Cell Membrane. AP Biology
 Movement across the Cell Membrane The diffusion of solutes across a synthetic membrane Molecules of dye WATER Membrane (cross section) Net diffusion Net diffusion Equilibrium (a) Diffusion of one solute
Movement across the Cell Membrane The diffusion of solutes across a synthetic membrane Molecules of dye WATER Membrane (cross section) Net diffusion Net diffusion Equilibrium (a) Diffusion of one solute
TRANSPORT ACROSS MEMBRANE
 TRANSPORT ACROSS MEMBRANE The plasma membrane functions to isolate the inside of the cell from its environment, but isolation is not complete. A large number of molecules constantly transit between the
TRANSPORT ACROSS MEMBRANE The plasma membrane functions to isolate the inside of the cell from its environment, but isolation is not complete. A large number of molecules constantly transit between the
State state describe
 Warm-Up State the products of the light-dependent reaction of photosynthesis, state which product has chemical energy, and describe how that product is made. KREBS ETC FADH 2 Glucose Pyruvate H 2 O NADH
Warm-Up State the products of the light-dependent reaction of photosynthesis, state which product has chemical energy, and describe how that product is made. KREBS ETC FADH 2 Glucose Pyruvate H 2 O NADH
Unit 3: Cell Energy Guided Notes
 Enzymes Unit 3: Cell Energy Guided Notes 1 We get energy from the food we eat by breaking apart the chemical bonds where food is stored. energy is in the bonds, energy is the energy we use to do things.
Enzymes Unit 3: Cell Energy Guided Notes 1 We get energy from the food we eat by breaking apart the chemical bonds where food is stored. energy is in the bonds, energy is the energy we use to do things.
Chemistry of Life Cells & Bioprocesses CRT Review
 Chemistry of Life Cells & Bioprocesses CRT Review Chapter 2: The Chemistry of Life macromolecules - The four types of macromolecules are carbohydrates, lipids, nucleic acids, and proteins Types of Macromolecules
Chemistry of Life Cells & Bioprocesses CRT Review Chapter 2: The Chemistry of Life macromolecules - The four types of macromolecules are carbohydrates, lipids, nucleic acids, and proteins Types of Macromolecules
CELL BIOLOGY - CLUTCH CH. 9 - TRANSPORT ACROSS MEMBRANES.
 !! www.clutchprep.com K + K + K + K + CELL BIOLOGY - CLUTCH CONCEPT: PRINCIPLES OF TRANSMEMBRANE TRANSPORT Membranes and Gradients Cells must be able to communicate across their membrane barriers to materials
!! www.clutchprep.com K + K + K + K + CELL BIOLOGY - CLUTCH CONCEPT: PRINCIPLES OF TRANSMEMBRANE TRANSPORT Membranes and Gradients Cells must be able to communicate across their membrane barriers to materials
Lecture 31: Solutions 5
 Lecture 31: Solutions 5 Read: BLB 13.5 13.6 HW: BLB 13:9,58,61,67,69,75 Sup13:12 18 Know: colligative properties vapor pressure lowering boiling point elevation freezing point depression osmotic pressure
Lecture 31: Solutions 5 Read: BLB 13.5 13.6 HW: BLB 13:9,58,61,67,69,75 Sup13:12 18 Know: colligative properties vapor pressure lowering boiling point elevation freezing point depression osmotic pressure
All organisms require a constant expenditure of energy to maintain the living state - "LIFE".
 CELLULAR RESPIRATION All organisms require a constant expenditure of energy to maintain the living state - "LIFE". Where does the energy come from and how is it made available for life? With rare exception,
CELLULAR RESPIRATION All organisms require a constant expenditure of energy to maintain the living state - "LIFE". Where does the energy come from and how is it made available for life? With rare exception,
Chapter Outline. The Living Cell. The Cell Theory. The Nature and Variety of Cells. Cell theory. Observing Cells: The Microscope
 Chapter Outline The Living Cell Chapter 21 The Nature and Variety of Cells How Does a Cell Work? Metabolism: Energy and Life Cell Division Great Idea: Life is based on chemistry, and chemistry takes place
Chapter Outline The Living Cell Chapter 21 The Nature and Variety of Cells How Does a Cell Work? Metabolism: Energy and Life Cell Division Great Idea: Life is based on chemistry, and chemistry takes place
Osmolarity, Tonicity, and Equivalents Study Guide & Practice. 1. Osmolarity
 Osmolarity, Tonicity, and Equivalents Study Guide & Practice 1. Osmolarity Recall that the osmotic pressure of a solution is created by solutes dissolved in a solvent, and the dissolved solutes create
Osmolarity, Tonicity, and Equivalents Study Guide & Practice 1. Osmolarity Recall that the osmotic pressure of a solution is created by solutes dissolved in a solvent, and the dissolved solutes create
Membrane Physiology. Dr. Hiwa Shafiq Oct-18 1
 Membrane Physiology Dr. Hiwa Shafiq 22-10-2018 29-Oct-18 1 Chemical compositions of extracellular and intracellular fluids. 29-Oct-18 2 Transport through the cell membrane occurs by one of two basic processes:
Membrane Physiology Dr. Hiwa Shafiq 22-10-2018 29-Oct-18 1 Chemical compositions of extracellular and intracellular fluids. 29-Oct-18 2 Transport through the cell membrane occurs by one of two basic processes:
2002NSC Human Physiology Semester Summary
 2002NSC Human Physiology Semester Summary Griffith University, Nathan Campus Semester 1, 2014 Topics include: - Diffusion, Membranes & Action Potentials - Fundamentals of the Nervous System - Neuroanatomy
2002NSC Human Physiology Semester Summary Griffith University, Nathan Campus Semester 1, 2014 Topics include: - Diffusion, Membranes & Action Potentials - Fundamentals of the Nervous System - Neuroanatomy
7.02 Colligative Properties
 7.02 Colligative Properties Changes in solvent properties due to impurities Colloidal suspensions or dispersions scatter light, a phenomenon known as the Tyndall effect. (a) Dust in the air scatters the
7.02 Colligative Properties Changes in solvent properties due to impurities Colloidal suspensions or dispersions scatter light, a phenomenon known as the Tyndall effect. (a) Dust in the air scatters the
Biology September 2015 Exam One FORM G KEY
 Biology 251 17 September 2015 Exam One FORM G KEY PRINT YOUR NAME AND ID NUMBER in the space that is provided on the answer sheet, and then blacken the letter boxes below the corresponding letters of your
Biology 251 17 September 2015 Exam One FORM G KEY PRINT YOUR NAME AND ID NUMBER in the space that is provided on the answer sheet, and then blacken the letter boxes below the corresponding letters of your
Biology September 2015 Exam One FORM W KEY
 Biology 251 17 September 2015 Exam One FORM W KEY PRINT YOUR NAME AND ID NUMBER in the space that is provided on the answer sheet, and then blacken the letter boxes below the corresponding letters of your
Biology 251 17 September 2015 Exam One FORM W KEY PRINT YOUR NAME AND ID NUMBER in the space that is provided on the answer sheet, and then blacken the letter boxes below the corresponding letters of your
Metabolism. Fermentation vs. Respiration. End products of fermentations are waste products and not fully.
 Outline: Metabolism Part I: Fermentations Part II: Respiration Part III: Metabolic Diversity Learning objectives are: Learn about respiratory metabolism, ATP generation by respiration linked (oxidative)
Outline: Metabolism Part I: Fermentations Part II: Respiration Part III: Metabolic Diversity Learning objectives are: Learn about respiratory metabolism, ATP generation by respiration linked (oxidative)
Chem Lecture 9 Pumps and Channels Part 1
 Chem 45 - Lecture 9 Pumps and Channels Part 1 Question of the Day: What two factors about a molecule influence the change in its free energy as it moves across a membrane? Membrane proteins function as
Chem 45 - Lecture 9 Pumps and Channels Part 1 Question of the Day: What two factors about a molecule influence the change in its free energy as it moves across a membrane? Membrane proteins function as
BIO 210 Chapter 4 Physiology of Cells. By Beth Wyatt, Jack Bagwell, & John McGill. Introduction
 BIO 210 Chapter 4 Physiology of Cells By Beth Wyatt, Jack Bagwell, & John McGill Introduction The living must exchange materials with the nonliving. How does this happen? Cell transport Two major types
BIO 210 Chapter 4 Physiology of Cells By Beth Wyatt, Jack Bagwell, & John McGill Introduction The living must exchange materials with the nonliving. How does this happen? Cell transport Two major types
ALE 24. Colligative Properties (Part 2)
 Name Chem 162, Section: Group Number: ALE 24. Colligative Properties (Part 2) (Reference: 13.6 Silberberg 5 th edition) Why is calcium chloride spread on highways in the North during the Winter? The Model:
Name Chem 162, Section: Group Number: ALE 24. Colligative Properties (Part 2) (Reference: 13.6 Silberberg 5 th edition) Why is calcium chloride spread on highways in the North during the Winter? The Model:
Endocrine Physiology. Introduction to Endocrine Principles
 Endocrine Physiology Introduction to Endocrine Principles There are TWO major groups of hormones Peptide and protein hormones Amine hormones Peptide and protein hormones act through cell membrane receptors
Endocrine Physiology Introduction to Endocrine Principles There are TWO major groups of hormones Peptide and protein hormones Amine hormones Peptide and protein hormones act through cell membrane receptors
Moving Materials Across The Cell Membrane
 1. Active transport: movement of large molecules across the cell membrane that uses the cell's energy. 2. Diffusion:the movement of molecules from an area of high concentration to an area of low concentration.
1. Active transport: movement of large molecules across the cell membrane that uses the cell's energy. 2. Diffusion:the movement of molecules from an area of high concentration to an area of low concentration.
Chapter 9. Solutions
 Chapter 9 Solutions Water Properties Polar O more electronegative, partial negative charge H less electronegative, partial positive charge Bent structure, 2 lone pairs on oxygen Participates in HYDROGEN
Chapter 9 Solutions Water Properties Polar O more electronegative, partial negative charge H less electronegative, partial positive charge Bent structure, 2 lone pairs on oxygen Participates in HYDROGEN
Cellular Respiration Stage 4: Electron Transport Chain
 Cellular Respiration Stage 4: Electron Transport Chain 2006-2007 Cellular respiration What s the point? The point is to make ATP! ATP 2006-2007 ATP accounting so far Glycolysis 2 ATP Kreb s cycle 2 ATP
Cellular Respiration Stage 4: Electron Transport Chain 2006-2007 Cellular respiration What s the point? The point is to make ATP! ATP 2006-2007 ATP accounting so far Glycolysis 2 ATP Kreb s cycle 2 ATP
Chapter 4: Energy From the sun to you in two easy steps
 Chapter 4: Energy From the sun to you in two easy steps Lectures by Mark Manteuffel, St. Louis Community College Learning Objectives Understand and be able to explain the following: How energy flows from
Chapter 4: Energy From the sun to you in two easy steps Lectures by Mark Manteuffel, St. Louis Community College Learning Objectives Understand and be able to explain the following: How energy flows from
Life 21 - Aerobic respiration Raven & Johnson Chapter 9 (parts)
 1 Life 21 - Aerobic respiration Raven & Johnson Chapter 9 (parts) Objectives 1: Describe the overall action of the Krebs cycle in generating ATP, NADH and FADH 2 from acetyl-coa 2: Understand the generation
1 Life 21 - Aerobic respiration Raven & Johnson Chapter 9 (parts) Objectives 1: Describe the overall action of the Krebs cycle in generating ATP, NADH and FADH 2 from acetyl-coa 2: Understand the generation
Housekeeping, 14 January 2009 LAB BEGINS TODAY
 Lecture 1, 14 Jan 2009 Chapter 1 Vertebrate Physiology ECOL 437 (MCB/VetSci 437) Univ. of Arizona, spring 2009 Kevin Bonine & Kevin Oh 1. Syllabus 2. Vertebrate Physiology Integration Structure/Function
Lecture 1, 14 Jan 2009 Chapter 1 Vertebrate Physiology ECOL 437 (MCB/VetSci 437) Univ. of Arizona, spring 2009 Kevin Bonine & Kevin Oh 1. Syllabus 2. Vertebrate Physiology Integration Structure/Function
Lecture 1 (1/24) (Collins)
 Lecture 1 (1/24) (Collins) Physiology The study of how cells interact with their environment to obtain the things requires for life (water, salts, heat, etc.) Exchange systems Systems that allow for the
Lecture 1 (1/24) (Collins) Physiology The study of how cells interact with their environment to obtain the things requires for life (water, salts, heat, etc.) Exchange systems Systems that allow for the
Properties of Solutions
 Properties of Solutions The Solution Process A solution is a homogeneous mixture of solute and solvent. Solutions may be gases, liquids, or solids. Each substance present is a component of the solution.
Properties of Solutions The Solution Process A solution is a homogeneous mixture of solute and solvent. Solutions may be gases, liquids, or solids. Each substance present is a component of the solution.
Lecture Series 9 Cellular Pathways That Harvest Chemical Energy
 Lecture Series 9 Cellular Pathways That Harvest Chemical Energy Reading Assignments Review Chapter 3 Energy, Catalysis, & Biosynthesis Read Chapter 13 How Cells obtain Energy from Food Read Chapter 14
Lecture Series 9 Cellular Pathways That Harvest Chemical Energy Reading Assignments Review Chapter 3 Energy, Catalysis, & Biosynthesis Read Chapter 13 How Cells obtain Energy from Food Read Chapter 14
Universality of sensory-response systems
 excite.org(anism): Electrical Signaling Universality of sensory-response systems Three step process: sensation-integration-response Bacterial chemotaxis Madigan et al. Fig. 8.24 Rick Stewart (CBMG) Human
excite.org(anism): Electrical Signaling Universality of sensory-response systems Three step process: sensation-integration-response Bacterial chemotaxis Madigan et al. Fig. 8.24 Rick Stewart (CBMG) Human
Physics of biological membranes, diffusion, osmosis Dr. László Nagy
 Physics of biological membranes, diffusion, osmosis Dr. László Nagy -Metabolic processes and transport processes. - Macrotransport : transport of large amount of material : through vessel systems : in
Physics of biological membranes, diffusion, osmosis Dr. László Nagy -Metabolic processes and transport processes. - Macrotransport : transport of large amount of material : through vessel systems : in
Photosynthesis and Cellular Respiration
 Name Date Class CHAPTER 5 TEST PREP PRETEST Photosynthesis and Cellular Respiration In the space provided, write the letter of the term or phrase that best completes each statement or best answers each
Name Date Class CHAPTER 5 TEST PREP PRETEST Photosynthesis and Cellular Respiration In the space provided, write the letter of the term or phrase that best completes each statement or best answers each
Solutions. Solutions Overview
 Solutions Chapter 9 Solutions Overview Terminology Units of Concentration Dilutions Colligative Properties 1 Terminology Solution- A homogenous mixture of two or more substances (ions or small molecules)
Solutions Chapter 9 Solutions Overview Terminology Units of Concentration Dilutions Colligative Properties 1 Terminology Solution- A homogenous mixture of two or more substances (ions or small molecules)
Renal handling of substances. Dr.Charushila Rukadikar Assistance Professor Physiology
 Renal handling of substances Dr.Charushila Rukadikar Assistance Professor Physiology GENERAL PRINCIPLES OF RENAL TUBULAR TRANSPORT Transport mechanisms across cell membrane 1) Passive transport i. Diffusion
Renal handling of substances Dr.Charushila Rukadikar Assistance Professor Physiology GENERAL PRINCIPLES OF RENAL TUBULAR TRANSPORT Transport mechanisms across cell membrane 1) Passive transport i. Diffusion
Main idea of this lecture:
 Ac#ve Transport Main idea of this lecture: How do molecules, big and small, get in OR out of a cell? 2 Main ways: Passive Transport (Does not require energy) Lecture 1 Ac=ve Transport (Requires energy)
Ac#ve Transport Main idea of this lecture: How do molecules, big and small, get in OR out of a cell? 2 Main ways: Passive Transport (Does not require energy) Lecture 1 Ac=ve Transport (Requires energy)
Pathways that Harvest and Store Chemical Energy
 6 Pathways that Harvest and Store Chemical Energy Energy is stored in chemical bonds and can be released and transformed by metabolic pathways. Chemical energy available to do work is termed free energy
6 Pathways that Harvest and Store Chemical Energy Energy is stored in chemical bonds and can be released and transformed by metabolic pathways. Chemical energy available to do work is termed free energy
Forms of stored energy in cells
 Forms of stored energy in cells Electrochemical gradients Covalent bonds (ATP) Reducing power (NADH) During photosynthesis, respiration and glycolysis these forms of energy are converted from one to another
Forms of stored energy in cells Electrochemical gradients Covalent bonds (ATP) Reducing power (NADH) During photosynthesis, respiration and glycolysis these forms of energy are converted from one to another
COLLIGATIVE PROPERTIES
 COLLIGATIVE PROPERTIES Depend on the number of solute particles in solution but not on the identity of the solute Vapor pressure lowering Boiling point elevation Freezing point depression Osmotic pressure
COLLIGATIVE PROPERTIES Depend on the number of solute particles in solution but not on the identity of the solute Vapor pressure lowering Boiling point elevation Freezing point depression Osmotic pressure
CELL SIGNALLING and MEMBRANE TRANSPORT. Mark Louie D. Lopez Department of Biology College of Science Polytechnic University of the Philippines
 CELL SIGNALLING and MEMBRANE TRANSPORT Mark Louie D. Lopez Department of Biology College of Science Polytechnic University of the Philippines GENERIC SIGNALLING PATHWAY CELL RESPONSE TO SIGNALS CELL RESPONSE
CELL SIGNALLING and MEMBRANE TRANSPORT Mark Louie D. Lopez Department of Biology College of Science Polytechnic University of the Philippines GENERIC SIGNALLING PATHWAY CELL RESPONSE TO SIGNALS CELL RESPONSE
Glycolysis: Acetyl-CoA synthesis (Intermediate Step) and Krebs Cycle Electron Transport Chain (ETC):
 Name: Bio AP Lab: Cell Respiration (Modified from Carolina Cell Respiration & AP Biology Investigative Labs) BACKGROUND: You are probably familiar with photosynthesis, the process that plants use to harness
Name: Bio AP Lab: Cell Respiration (Modified from Carolina Cell Respiration & AP Biology Investigative Labs) BACKGROUND: You are probably familiar with photosynthesis, the process that plants use to harness
Solution Concentrations CHAPTER OUTLINE
 Chapter 8B Solution Concentrations CHAPTER OUTLINE Concentration Units Mass Percent Using Percent Concentration Molarity Using Molarity Dilution Osmolarity Tonicity of Solutions 2 CONCENTRATION UNITS The
Chapter 8B Solution Concentrations CHAPTER OUTLINE Concentration Units Mass Percent Using Percent Concentration Molarity Using Molarity Dilution Osmolarity Tonicity of Solutions 2 CONCENTRATION UNITS The
Hole s Human Anatomy and Physiology Tenth Edition. Chapter 2
 PowerPoint Lecture Outlines to accompany Hole s Human Anatomy and Physiology Tenth Edition Shier w Butler w Lewis Chapter 2 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction
PowerPoint Lecture Outlines to accompany Hole s Human Anatomy and Physiology Tenth Edition Shier w Butler w Lewis Chapter 2 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction
Water, water everywhere,; not a drop to drink. Consumption resulting from how environment inhabited Deforestation disrupts water cycle
 Chapter 3 Water: The Matrix of Life Overview n n n Water, water everywhere,; not a drop to drink Only 3% of world s water is fresh How has this happened Consumption resulting from how environment inhabited
Chapter 3 Water: The Matrix of Life Overview n n n Water, water everywhere,; not a drop to drink Only 3% of world s water is fresh How has this happened Consumption resulting from how environment inhabited
Colligative Properties
 Colligative Properties! Consider three beakers: " 50.0 g of ice " 50.0 g of ice + 0.15 moles NaCl " 50.0 g of ice + 0.15 moles sugar (sucrose)! What will the freezing temperature of each beaker be? " Beaker
Colligative Properties! Consider three beakers: " 50.0 g of ice " 50.0 g of ice + 0.15 moles NaCl " 50.0 g of ice + 0.15 moles sugar (sucrose)! What will the freezing temperature of each beaker be? " Beaker
Properties of Solutions. Overview of factors affecting solubility Ways of expressing concentration Physical properties of solutions
 Properties of Solutions Overview of factors affecting solubility Ways of expressing concentration Physical properties of solutions Learning objectives Define terms solute, solvent and solution Distinguish
Properties of Solutions Overview of factors affecting solubility Ways of expressing concentration Physical properties of solutions Learning objectives Define terms solute, solvent and solution Distinguish
Cell Structure and Function Unit 4
 Cell Structure and Function Unit 4 Definition of Cell A cell is the smallest unit that is capable of performing life functions. RECALL... Levels of Organization! Why do we call them cells? In 1665, Robert
Cell Structure and Function Unit 4 Definition of Cell A cell is the smallest unit that is capable of performing life functions. RECALL... Levels of Organization! Why do we call them cells? In 1665, Robert
OT Exam 1, August 9, 2002 Page 1 of 8. Occupational Therapy Physiology, Summer Examination 1. August 9, 2002
 Page 1 of 8 Occupational Therapy Physiology, Summer 2002 Examination 1 August 9, 2002 Dr. Heckman's section is questions 1-6 and each question is worth 5 points for a total of 30 points. Dr. Driska's section
Page 1 of 8 Occupational Therapy Physiology, Summer 2002 Examination 1 August 9, 2002 Dr. Heckman's section is questions 1-6 and each question is worth 5 points for a total of 30 points. Dr. Driska's section
Organisms are made up of specialized cells.
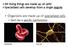 All living things are made up of cells! Specialized cells develop from a single zygote Organisms are made up of specialized cells. Each has a specific job/function red blood cell nerve cell Zygotes (fertilized
All living things are made up of cells! Specialized cells develop from a single zygote Organisms are made up of specialized cells. Each has a specific job/function red blood cell nerve cell Zygotes (fertilized
A solution is a homogeneous mixture of two or more substances.
 UNIT (5) SOLUTIONS A solution is a homogeneous mixture of two or more substances. 5.1 Terminology Solute and Solvent A simple solution has two components, a solute, and a solvent. The substance in smaller
UNIT (5) SOLUTIONS A solution is a homogeneous mixture of two or more substances. 5.1 Terminology Solute and Solvent A simple solution has two components, a solute, and a solvent. The substance in smaller
CBSE Quick Revision Notes (Class-11 Biology) CHAPTER-11 TRANSPORT IN PLANTS
 CBSE Quick Revision Notes (Class-11 Biology) CHAPTER-11 TRANSPORT IN PLANTS Plant transport various substance like gases, minerals, water, hormones, photosynthetes and organic solutes to short distance
CBSE Quick Revision Notes (Class-11 Biology) CHAPTER-11 TRANSPORT IN PLANTS Plant transport various substance like gases, minerals, water, hormones, photosynthetes and organic solutes to short distance
Metabolism Review. A. Top 10
 A. Top 10 Metabolism Review 1. Energy production through chemiosmosis a. pumping of H+ ions onto one side of a membrane through protein pumps in an Electron Transport Chain (ETC) b. flow of H+ ions across
A. Top 10 Metabolism Review 1. Energy production through chemiosmosis a. pumping of H+ ions onto one side of a membrane through protein pumps in an Electron Transport Chain (ETC) b. flow of H+ ions across
solubility solubilities that increase with increasing temperature
 Solubility The concentration of the solute in a saturated solution is the solubility of the solute About 95% of all ionic compounds have aqueous solubilities that increase with increasing temperature Temperature
Solubility The concentration of the solute in a saturated solution is the solubility of the solute About 95% of all ionic compounds have aqueous solubilities that increase with increasing temperature Temperature
Clicker Question. Clicker Question
 Which organelle provides a cell with protection? A. Mitochondria B. Cell membrane C. Nucleus D. Chloroplast This organelle uses sunlight in order to make glucose. A. Chloroplast B. Mitochondria C. Golgi
Which organelle provides a cell with protection? A. Mitochondria B. Cell membrane C. Nucleus D. Chloroplast This organelle uses sunlight in order to make glucose. A. Chloroplast B. Mitochondria C. Golgi
Energy Transformation. Metabolism = total chemical reactions in cells.
 Energy Transformation Metabolism = total chemical reactions in cells. metabole = change Metabolism is concerned with managing the material and energy resources of the cell -Catabolism -Anabolism -Catabolism
Energy Transformation Metabolism = total chemical reactions in cells. metabole = change Metabolism is concerned with managing the material and energy resources of the cell -Catabolism -Anabolism -Catabolism
Cellular Transportation & Respiration
 Cellular Transportation & Respiration Passive Transport A cell membrane is semiperamble, which means that it allows only certain substances to enter or leave a cell. Passive transport is the movement of
Cellular Transportation & Respiration Passive Transport A cell membrane is semiperamble, which means that it allows only certain substances to enter or leave a cell. Passive transport is the movement of
Chapter 12.4 Colligative Properties of Solutions Objectives List and define the colligative properties of solutions. Relate the values of colligative
 Chapter 12.4 Colligative Properties of Solutions Objectives List and define the colligative properties of solutions. Relate the values of colligative properties to the concentrations of solutions. Calculate
Chapter 12.4 Colligative Properties of Solutions Objectives List and define the colligative properties of solutions. Relate the values of colligative properties to the concentrations of solutions. Calculate
Copy into Note Packet and Return to Teacher
 Copy into Note Packet and Return to Teacher Section 1: Nature of Matter Objectives: Differentiate between atoms and elements. Analyze how compounds are formed. Distinguish between covalent bonds, hydrogen
Copy into Note Packet and Return to Teacher Section 1: Nature of Matter Objectives: Differentiate between atoms and elements. Analyze how compounds are formed. Distinguish between covalent bonds, hydrogen
Alaa' Alajrami. Hussam Twaissi. Mohammad khatatbeh
 6 Alaa' Alajrami Hussam Twaissi Mohammad khatatbeh Keep smiling stay beautiful,,, We will continue talking about cell membrane and what cause the potential across it. * Proton depended carrier: (secondary
6 Alaa' Alajrami Hussam Twaissi Mohammad khatatbeh Keep smiling stay beautiful,,, We will continue talking about cell membrane and what cause the potential across it. * Proton depended carrier: (secondary
Introduction to Physiology II: Control of Cell Volume and Membrane Potential
 Introduction to Physiology II: Control of Cell Volume and Membrane Potential J. P. Keener Mathematics Department Math Physiology p.1/23 Basic Problem The cell is full of stuff: Proteins, ions, fats, etc.
Introduction to Physiology II: Control of Cell Volume and Membrane Potential J. P. Keener Mathematics Department Math Physiology p.1/23 Basic Problem The cell is full of stuff: Proteins, ions, fats, etc.
Physical Pharmacy. Solutions. Khalid T Maaroof MSc. Pharmaceutical sciences School of pharmacy Pharmaceutics department
 Physical Pharmacy Solutions Khalid T Maaroof MSc. Pharmaceutical sciences School of pharmacy Pharmaceutics department 10/31/2015 Online access: bit.ly/physicalpharmacy 1 Mixtures a combination of two or
Physical Pharmacy Solutions Khalid T Maaroof MSc. Pharmaceutical sciences School of pharmacy Pharmaceutics department 10/31/2015 Online access: bit.ly/physicalpharmacy 1 Mixtures a combination of two or
Basic Chemistry. Chemistry Review. Bio 250: Anatomy & Physiology
 Basic Chemistry Bio 250: Anatomy & Physiology Chemistry Review It is going to be your responsibility to review the basic principles of chemistry you learned in BIO 101 This basic set of notes will help
Basic Chemistry Bio 250: Anatomy & Physiology Chemistry Review It is going to be your responsibility to review the basic principles of chemistry you learned in BIO 101 This basic set of notes will help
Membrane Structure and Function POGIL
 Why? Membrane Structure and Function POGIL Advertisements for sports drinks, such as Gatorade, Powerade, and Vitaminwater seem to be everywhere. All of these drinks are supposed to help your body recover
Why? Membrane Structure and Function POGIL Advertisements for sports drinks, such as Gatorade, Powerade, and Vitaminwater seem to be everywhere. All of these drinks are supposed to help your body recover
Water and solutions. Prof. Ramune Morkuniene, Biochemistry Dept., LUHS
 Water and solutions Prof. Ramune Morkuniene, Biochemistry Dept., LUHS Characteristics of water molecule Hydrophylic, hydrophobic and amphipatic compounds Types of real solutions Electrolytes and non- electrolytes
Water and solutions Prof. Ramune Morkuniene, Biochemistry Dept., LUHS Characteristics of water molecule Hydrophylic, hydrophobic and amphipatic compounds Types of real solutions Electrolytes and non- electrolytes
Chapter 11 Problems: 11, 15, 18, 20-23, 30, 32-35, 39, 41, 43, 45, 47, 49-51, 53, 55-57, 59-61, 63, 65, 67, 70, 71, 74, 75, 78, 81, 85, 86, 93
 Chapter 11 Problems: 11, 15, 18, 20-23, 30, 32-35, 39, 41, 43, 45, 47, 49-51, 53, 55-57, 59-61, 63, 65, 67, 70, 71, 74, 75, 78, 81, 85, 86, 93 Chapter 11 Properties of Solutions Types of mixtures: homogenous
Chapter 11 Problems: 11, 15, 18, 20-23, 30, 32-35, 39, 41, 43, 45, 47, 49-51, 53, 55-57, 59-61, 63, 65, 67, 70, 71, 74, 75, 78, 81, 85, 86, 93 Chapter 11 Properties of Solutions Types of mixtures: homogenous
Advanced Higher Biology. Unit 1- Cells and Proteins 2c) Membrane Proteins
 Advanced Higher Biology Unit 1- Cells and Proteins 2c) Membrane Proteins Membrane Structure Phospholipid bilayer Transmembrane protein Integral protein Movement of Molecules Across Membranes Phospholipid
Advanced Higher Biology Unit 1- Cells and Proteins 2c) Membrane Proteins Membrane Structure Phospholipid bilayer Transmembrane protein Integral protein Movement of Molecules Across Membranes Phospholipid
Chapter 11. Properties of Solutions Solutions
 Chapter 11. Properties of Solutions Solutions Homogeneous Mixture 1 Solution Composition Equivalent moles of solute (mol) Acid-Base reaction Molarity (M) = liter of solution (L) 1 eq: the quantity of acid
Chapter 11. Properties of Solutions Solutions Homogeneous Mixture 1 Solution Composition Equivalent moles of solute (mol) Acid-Base reaction Molarity (M) = liter of solution (L) 1 eq: the quantity of acid
Chapter 13. Properties of Solutions. Lecture Presentation. John D. Bookstaver St. Charles Community College Cottleville, MO
 Lecture Presentation Chapter 13 Properties of John D. Bookstaver St. Charles Community College Cottleville, MO are homogeneous mixtures of two or more pure substances. In a solution, the solute is dispersed
Lecture Presentation Chapter 13 Properties of John D. Bookstaver St. Charles Community College Cottleville, MO are homogeneous mixtures of two or more pure substances. In a solution, the solute is dispersed
Thinking Like a Chemist About Solution Properties UNIT 5 DAY 4
 UNIT5-DAY4-LaB1230 Page 1 UNIT5-DAY4-LaB1230 Wednesday, January 23, 2013 5:43 PM Thinking Like a Chemist About Solution Properties UNIT 5 DAY 4 What are we going to learn today? Thinking Like a Chemist
UNIT5-DAY4-LaB1230 Page 1 UNIT5-DAY4-LaB1230 Wednesday, January 23, 2013 5:43 PM Thinking Like a Chemist About Solution Properties UNIT 5 DAY 4 What are we going to learn today? Thinking Like a Chemist
Chapter 11 Properties of Solutions
 Chapter 11 Properties of Solutions Solutions Homogeneous mixtures of two or more substances Composition is uniform throughout the sample No chemical reaction between the components of the mixture Solvents
Chapter 11 Properties of Solutions Solutions Homogeneous mixtures of two or more substances Composition is uniform throughout the sample No chemical reaction between the components of the mixture Solvents
Where does Physical Chemistry fit into your course in Dentistry?
 Where does Physical Chemistry fit into your course in Dentistry? Acidogenic bacteria in dental plaque can rapidly metabolise certain carbohydrates to acid endproducts. In the mouth, the resultant change
Where does Physical Chemistry fit into your course in Dentistry? Acidogenic bacteria in dental plaque can rapidly metabolise certain carbohydrates to acid endproducts. In the mouth, the resultant change
Chemical Basis of Life
 Chemical Basis of Life Jan 30 11:42 AM In order to understand digestion and nutrition, we need some basic biochemistry Chemistry studies the composition of matter and its changes as well as the change
Chemical Basis of Life Jan 30 11:42 AM In order to understand digestion and nutrition, we need some basic biochemistry Chemistry studies the composition of matter and its changes as well as the change
REVIEW 2: CELLS & CELL COMMUNICATION. A. Top 10 If you learned anything from this unit, you should have learned:
 Name AP Biology REVIEW 2: CELLS & CELL COMMUNICATION A. Top 10 If you learned anything from this unit, you should have learned: 1. Prokaryotes vs. eukaryotes No internal membranes vs. membrane-bound organelles
Name AP Biology REVIEW 2: CELLS & CELL COMMUNICATION A. Top 10 If you learned anything from this unit, you should have learned: 1. Prokaryotes vs. eukaryotes No internal membranes vs. membrane-bound organelles
A + B = C C + D = E E + F = A
 Photosynthesis - Plants obtain energy directly from the sun - Organisms that do this are autotrophs (make their own food from inorganic forms) - Photosynthesis is a series of chemical reactions where the
Photosynthesis - Plants obtain energy directly from the sun - Organisms that do this are autotrophs (make their own food from inorganic forms) - Photosynthesis is a series of chemical reactions where the
Hole s Human Anatomy and Physiology Eleventh Edition. Chapter 2
 Hole s Human Anatomy and Physiology Eleventh Edition Shier Butler Lewis Chapter 2 1 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. CHAPTER 2 CHEMICAL BASIS OF
Hole s Human Anatomy and Physiology Eleventh Edition Shier Butler Lewis Chapter 2 1 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. CHAPTER 2 CHEMICAL BASIS OF
METABOLISM. What is metabolism? Categories of metabolic reactions. Total of all chemical reactions occurring within the body
 METABOLISM What is metabolism? METABOLISM Total of all chemical reactions occurring within the body Categories of metabolic reactions Catabolic reactions Degradation pathways Anabolic reactions Synthesis
METABOLISM What is metabolism? METABOLISM Total of all chemical reactions occurring within the body Categories of metabolic reactions Catabolic reactions Degradation pathways Anabolic reactions Synthesis
Cell Energy Notes ATP THE ENDOSYMBIOTIC THEORY. CELL ENERGY Cells usable source of is called ATP stands for. Name Per
 Cell Energy Notes Name Per THE ENDOSYMBIOTIC THEORY The Endosymbiotic theory is the idea that a long time ago, engulfed other prokaryotic cells by. This resulted in the first First proposed by Explains
Cell Energy Notes Name Per THE ENDOSYMBIOTIC THEORY The Endosymbiotic theory is the idea that a long time ago, engulfed other prokaryotic cells by. This resulted in the first First proposed by Explains
CELL STRUCTURE AND FUNCTION NOTES
 CELL STRUCTURE AND FUNCTION NOTES Cell Theory 1. All living things 2. Cells are the. 3. Cells are produced All Living things have cells a. Unicellular b. Multicellular Types of Cells a. Prokaryote: i.
CELL STRUCTURE AND FUNCTION NOTES Cell Theory 1. All living things 2. Cells are the. 3. Cells are produced All Living things have cells a. Unicellular b. Multicellular Types of Cells a. Prokaryote: i.
