10. SPONTANEOUS EMISSION & MULTIPOLE INTERACTIONS
|
|
|
- Oliver Wilkinson
- 6 years ago
- Views:
Transcription
1 P4533 Fall 1995 (Zorn) Atoms in the Radiation Field, 2 page SPONTANEOUS EMISSION & MULTIPOLE INTERACTIONS In this chapter we address two of the questions that remain as we build a theoretical description of atoms in a radiation field. In Sec we discuss the origin of spontaneous emission, and In Sec we explore the use of multipole expansions to understand why the electric dipole interaction dominates the atom-field coupling Why Excited Atoms Decay A Principle Of Equal Probability The common observation is that an excited atom tends to decay (e.g. n=2 n=1) with the spontaneous emission of a photon, but that the opposite process, spontaneous absorption (n=1 n=2), does not happen. We would like to understand why this is so on the basis of general principles. Of course it is possible to say something like "atoms naturally seek their lowest energy level", but this is neither intellectually satisfying nor really correct. Rather we will approach the atom+field problem on the basis of a general statistical principle that can be applied to a wide variety of situations ranging from the simple throw of dice to the intricate coupling of an atom to a field. This principle can be summarized in a somewhat oversimplified manner as follows: Suppose we have defined a physical system of interest In our mind's eye we now consider a large number (an ensemble) of identical systems, all of which have been prepared in the same way. Let the individual system have N different configurations (k = 1,2,3...N) in which it might be found under the conditions of preparation. We will assume that each system has an observable g (with values g k ) that identifies the system's configuration. For the moment suppose that there is no a priori reason to prefer any one of the configurations over any another. Now do a sequence of measurements of the quantity g on randomly selected members of the ensemble. The principle asserts that (under these conditions) all values of g k will occur with equal frequency. This principle does not seem remarkable at first glance, but it underlies equilibrium statistical physics in a way that permits one do to real calculations. Its generalizations lead to the Liouville theorem and to the thermodynamic concepts of entropy and free energy. We bring it up here so that we can understand why spontaneous emission is common and why Einstein could ignore spontaneous absorption in his phenomenological description of radiative processes.
2 P4533 Fall 1995 (Zorn) Atoms in the Radiation Field, 2 page 10.2 A Coupled Oscillator Model For The Atom + Field System We now build on the oscillator representation of the electromagnetic field. OBJECTIVE: To build a conceptual model based on coupled oscillators that replicates the important features of the atom in a radiation field. To show that an oscillator formalism can be used to represent the atom + field system in an intuitive way, and To show that this formalism provides a basis for understanding: (1) Why an atom in an excited state tends to decay to its ground state by spontaneous emission, (2) Why the reverse process [spontaneous absorption] is not likely, (3) That the Heisenberg uncertainty δe δt>h (perhaps applied in the form δν δ >1 for the analysis of spectra) is not an additional postulate of quantum mechanics but instead follows as a consequence of this conventional model of the atom+field system. Basics of Coupled Oscillators: Two entities having fairly similar oscillation frequencies find it relatively easy to exchange energy even if the coupling between them is weak. A simple example is provided by two pendulums of essentially equal length that are weakly coupled in some manner(for example by being hung from the same not-so-rigid support). If one pendulum is set in motion while the other is at rest, the second gradually picks up oscillation amplitude while the first dies down; the energy then flows back from the second to the first and this slow cycle repeats over and over until mechanical losses cause the oscillations in both to die out. This situation in classical mechanics has a close parallel in quantum mechanics that will prove useful for our model of the interaction of an atom (regarded as a micro-object with a dominant resonance frequency) with the matching Fourier components of the electromagnetic radiation field. Oscillator #1 Oscillator #2 Time Fig 10.1 Coupled oscillator amplitudes: #1 initially excited, #2 initially at rest
3 P4533 Fall 1995 (Zorn) Atoms in the Radiation Field, 2 page 10.3 The simplest atom+field model is one that uses a system with only two energy levels, separated by frequency ν, to represent the atom and a single, quantized harmonic oscillator (with equally spaced levels separated by the same frequency ν) to represent the field. The two oscillators systems are weakly coupled. We begin at time t=0 with the atom in its upper energy state and the field in its lowest state, and then gives its energy to the electromagnetic field after characteristic time T, but the field soon gives the energy back to the atom which returns to the excited state after time 2T. The cycle then continues, with the back&forth flow of energy between field and atom, much as is shown in Fig This entire set of oscillators represents the field "Atom" coupling This example has only one field oscillator in resonance with the atom Fig Oscillator model for case where atom resonates with only one oscillator Atomic spectral line cavity modes Fig Spectral diagram when atomic line is narrower than cavity mode spacing; atom resonates with only one cavity mode. Although one can create the situation of Figs. (10.2) and (10.3) by placing an atom into a small resonant cavity of extraordinarily high Q, this model with only one active field mode does not show the usual behavior of the ordinary atom+field system. We need to allow for participation of more field modes.
4 P4533 Fall 1995 (Zorn) Atoms in the Radiation Field, 2 page 10.4 More representative of actual situations is the model where the cavity is large enough so that many modes of the electromagnetic field are essentially resonant with the atomic transition. If there are M such modes, we can regard the composite system as one of M+1 weakly coupled oscillators, where all but one are in their lowest state at time t=0. The weak coupling allows the energy to be transferred from the initially excited oscillator to another oscillator in a characteristic time T. This passage of the excitation among the M+1 oscillators continues in random fashion, much as the passage of the little ball in a roulette wheel. M is often a rather large number and, as a consequence of this sharing of the quantum among all the field oscillators and the single atom, it is extremely unlikely that the atom will be again discovered in its excited state. This entire set of oscillators represents the field "Atom" Coupled this subset has M members; they are all very nearly in resonance with the atom Fig Oscillator model for case where atom resonates with many cavity modes Atomic spectral line cavity modes Fig Spectral diagram when atomic line spans many cavity modes In presenting diagrams and analogies to illustrate the spirit of this model, we use the icon of a pendulum to represent an oscillator with one characteristic frequency. So a pendulum symbol will represent not only the 2-level atom but also a mode of the electromagnetic field. We suggest the variation in oscillator frequencies by drawing the pendulum cords of slightly different length. (Note that we tacitly assume that the characteristic time for energy transfer between oscillators is much longer than the period (2π/ω ) of any of the oscillators.)
5 P4533 Fall 1995 (Zorn) Atoms in the Radiation Field, 2 page 10.5 Can we predict the evolution of the system after t=0? Suppose we begin at t=0 with the atom in an excited state but with no excitation in any mode of the field (all n k =0). The system at time t=0 is approximately described by: Ψ(t = 0) = n atom =1 n k field = 0 = 1,0,0,0,0,0,0,L (10.1) But this state is not stable because the quantum numbers of the modes will change at a rate that depends on the strength of the coupling between modes. In principle, it would seem that the amplitude of each oscillator at time t is predictable from a knowledge of the amplitudes at time t=0. In practice, given the inevitable residual randomness in most experimental situations, one predicts that the initiallyconcentrated probability amplitude diffuses equally and rapidly so each oscillator finally has its own small share ε.. Because there are so many field modes that are energetically accessible, the distribution of probability soon evolves to one that is heavily concentrated in the field and hardly at all in the atom: Ψ(0) = 1,0,0,0,0,0,0,L t>>τ Ψ(t) = ε, ε,ε,ε,ε,ε,ε,l (10.2) Density Of Modes The overlap of the spectral line with many cavity modes provides the opportunity for excitation to wander away from the atom. The density of such modes (the number of cavity modes per frequency interval dν, reckoned at large mode numbers) goes up with the cube of the frequency in an ordinary, three-dimensional cavity. This provides a theoretical justification for the proportionality of the lifetime to λ 3, as deduced by Einstein on thermodynamic grounds. Harmonic Oscillator representation for field is not optional: To model the atom we just needed an entity that would absorb/emit radiation at a relatively well defined frequency. In this discussion we are concerned almost entirely with the ground state and a single excited state of the atom, so we could have used any potential with at least one bound state for our atom model. On the other hand, the choice of a harmonic oscillator representation is essential for our treatment of the field. We know that the energy per mode is directly proportional to the number of photons in the mode. Since all those photons have the same frequency, the mode energy levels are equally spaced. So our representative for a field mode must also have equally spaced energy levels, and the this is found only in the simple harmonic oscillator. We have no alternative.
6 P4533 Fall 1995 (Zorn) Atoms in the Radiation Field, 2 page 10.6 Spontaneous Absorption Is Unlikely Under the hypothesized circumstances (with J.1 and only one quantum of available energy in the entire system) the atom, once de-excited via "spontaneous" decay, is very unlikely to be found again in its excited state. Therefore we regard "spontaneous" absorption (finding the atom in the excited state sometime after it had first decayed to the ground state) as a statistically insignificant process in our theory of atom-radiation interactions. Stimulated Absorption is Probable if Field is Excited: The argument presented so far has been based on the field being quiet (n k = 0 for all k) at t=0, and this is not too bad an approximation when one has a modest intensity of field inside a cavity of decent size; the occupation number per mode is then very low. However the description is significantly modified if any field modes near the atomic resonance are substantially excited, for example by shining a laser on the system. In that case the field has many photons in the relevant frequency range, and the atom has a good chance of acquiring a unit of that energy. One predicts theoretically (and confirms experimentally) that the an atom once in the ground state will be found in the excited state with a probability that depends on the intensity of the radiation at the relevant frequency. This is the quantum theoretic basis for Einstein's phenomenological description [Eqs. (9.1) and (9.2)] of the rate of stimulated and spontaneous transitions between two atomic levels: dn = N dt 1 ( t) B ν ρ( ν) and dn dt = N 2 t [ ( )] (10.3) ( ) A 21 + B 21 ρ ν 21 An excited atom is noticeable The presence of an excited atom within an apparatus can be quite noticeable, whereas a radiation field with one quantum of energy has many more (but to the experimentalist, usually equivalent) ways of holding the quantum of energy than does the atom. So random process will usually end up with the atom de-excited and the field excited in one of its modes. It its way, this is analogous to playing a slot machine with many wheels instead of the usual three... only one of all the possible configurations (each with its own minuscule probability) gets our attention.
7 P4533 Fall 1995 (Zorn) Atoms in the Radiation Field, 2 page Describing the Atom-Field Interaction with Multipoles Suppose an atom finds itself in an electrical environment described by the scalar potential V(x). This potential might be produced by a distant source of radiation or by a nearby set of electrodes connected to sources of voltage. Or in a collision experiment the field might be produced by an oncoming charged particle. We want to describe the energy that arises from the atom's being exposed to this field. If the atom were considered as a classical system comprised of point charges qn at locations xn in this potential, the energy could be written as. q n V ( x n ). n This can be generalized to an atom described as a classical, continuous distribution of charge (x). And we can write a similar form for an atom with a total charge e distributed with charge density Ψ * ( x) Ψ( x). { = q n V x n ρ( x) V( x)dx Ψ * ( x) V( x) Ψ( x)dx (10.4) W Energy n ( ) Classical, with discrete charges Classical, with continuous charge distribution ρ(x) quantum mechanical, with charge distribution given by Ψ * Ψ It will be convenient to expand the externally-applied potential V(x) in a Taylor series: V( x) = V x 0 x ( ) + V = V 0 + V x x + 0 ( x x 0 ) + x o 1 2 V 2! x V 2! x 2 x x 0 x o x 2 + L ( ) 2 + L (10.5a,b) where the second form applies when we make the reasonable choice of taking x =0 at the center of mass of the atom. The classical field-atom interaction energy is then: constant W = ρ(x) } 6 constant constant 74 8 V V 1 2 V + 0 x x + 0 2! x 2 x 2 + L 0 dx (10.6) We consider a one-dimensional situation here in order to present the physics of the atomfield interaction with minimum of algebraic clutter. It is obvious that the theory will usually be needed in its three-dimensional form, and then often in spherical coordinates. We can discuss this more realistic case with greater ease somewhat later, after we have developed our use of spherical harmonics and the theory of angular momentum..
8 P4533 Fall 1995 (Zorn) Atoms in the Radiation Field, 2 page 10.8 Note that the derivatives of V are evaluated at x=0 and therefore can be taken out from under the integrals W{ = V 0ρ(x) dx field-atom interaction energy monopole term netcharge on atom V x ρ(x) x dx E-field at the origin dipole term electric dipole moment of the atom quadrupole term V 2! x 2 ρ(x) x 2 dx (10.7) E-field gradient at origin electric quadrupole moment of the atom This is the beginning (and the most important part) of a multipole expansion for the energy of interaction between a field and a charge distribution. The terms in this expansion (10.7) involve a separation of variables, so to speak, since they are products in which one term describes the field, the other, the atom: characteristics of the field as given by the potential and its gradients characteristics of the atom as given by its multipole moments The first (monopole) term in the series describes the situation to first order as though a point charge were situated at the origin of coordinates. The subsequent terms then provide corrections and additions. In many cases it is sufficient to describe the system as though it had only a monopole interaction. But often (particularly if the net charge is zero) we are also forced to acknowledge the presence of the dipole interaction. We can usually (but not always) neglect the quadrupole interaction. The multipole moment description is very typical of physics, first using a coarse approximation and then adding more and more refinements to get better and better agreement between experiment and theory. Example of Linear Multipole: In the figure below, we show an arbitrary distribution of charge (point charges of both signs) along the x-axis that can be represented by a superposition of the monopole, dipole and quadrupole moments. The monopole, dipole, and quadrupole should all be placed on the same x-axis to make this synthesis x=0 x MONOPOLE APPROXIMATION x=0 x ORIGINAL LINEAR CHARGE DISTRIBUTION x DIPOLE CONTRIBUTION QUADRUPOLE CONTRIBUTION x Fig 10.6 Charge distribution along a line and its resolution into multipole components To first order, this is a point charge of +2. Upon closer inspection we find that the original distribution has properties of a dipole moment pointing to the left, and of a spreading of net of (+) charge as a quadrupole moment. x=0
9 P4533 Fall 1995 (Zorn) Atoms in the Radiation Field, 2 page 10.9 The equivalent to (10.7) written out using the quantum state functions to express the charge distribution is: W = ev 0 Ψ * Ψ dx + e V x Ψ * xψ dx 0 + e 1 2 V 2! x 2 0 Ψ * x 2 Ψ dx + L(10.8) We see from expression (10.8) that: I. The monopole term ev 0 Ψ * Ψ dx has the same magnitude irrespective of the state of the atom since Ψ * Ψ dx = 1 by normalization of the state function. In an electrically neutral atom, the interaction energy of the electrons is precisely offset by the energy from the interaction of the protons, so the monopole term will vanish. II. The dipole term vanishes unless a) the state Ψ (x) is a superposition that contains terms of both (+) and (-) parity. b) the electric field has a finite value at x=0 Comments Applied Field induces a dipole moment: Isolated atoms have spherical symmetry and thus have no intrinsic electric dipole moment, however if one applies an external electric field, that field couples to and distorts the charge distribution; the field induces a dipole moment, and associated with this polarization of the atom there is a finite dipole interaction energy. Dipole Interaction usually most important: If an atom is subjected to an oscillating field where the wave-length (λ) is much larger than the dimensions of the atom, then the field (even though oscillating in time) is essentially homogenous over the volume of the atom at any given instant. Thus the field E = V/ x is appreciable for situations involving ultraviolet, visible, infrared, and microwave radiation. (But the field gradient is very small: E/ x = 2 V/ x 2 ~ 0, so quadrupole processes are less important.) For this reason, atomic radiative processes at wavelengths longer than the extreme uv are almost completely governed by the dipole interaction. The formulation of the dipole interaction as given here enables us to can derive the selection rules and other statements about the relative probabilities for various radiative transitions. Collision Experiment may Involve High-order multipoles: If the atom is subject to collisions from incoming charged particles, then the atom may experience an electric field that is not at all homogeneous. In that case, both the field and its gradients may be of substantial magnitude and, as a consequence, the quadrupole term of the multipole series may be relatively large.
Advanced Optical Communications Prof. R. K. Shevgaonkar Department of Electrical Engineering Indian Institute of Technology, Bombay
 Advanced Optical Communications Prof. R. K. Shevgaonkar Department of Electrical Engineering Indian Institute of Technology, Bombay Lecture No. # 15 Laser - I In the last lecture, we discussed various
Advanced Optical Communications Prof. R. K. Shevgaonkar Department of Electrical Engineering Indian Institute of Technology, Bombay Lecture No. # 15 Laser - I In the last lecture, we discussed various
Spontaneous Emission, Stimulated Emission, and Absorption
 Chapter Six Spontaneous Emission, Stimulated Emission, and Absorption In this chapter, we review the general principles governing absorption and emission of radiation by absorbers with quantized energy
Chapter Six Spontaneous Emission, Stimulated Emission, and Absorption In this chapter, we review the general principles governing absorption and emission of radiation by absorbers with quantized energy
MODERN OPTICS. P47 Optics: Unit 9
 MODERN OPTICS P47 Optics: Unit 9 Course Outline Unit 1: Electromagnetic Waves Unit 2: Interaction with Matter Unit 3: Geometric Optics Unit 4: Superposition of Waves Unit 5: Polarization Unit 6: Interference
MODERN OPTICS P47 Optics: Unit 9 Course Outline Unit 1: Electromagnetic Waves Unit 2: Interaction with Matter Unit 3: Geometric Optics Unit 4: Superposition of Waves Unit 5: Polarization Unit 6: Interference
Molecular spectroscopy
 Molecular spectroscopy Origin of spectral lines = absorption, emission and scattering of a photon when the energy of a molecule changes: rad( ) M M * rad( ' ) ' v' 0 0 absorption( ) emission ( ) scattering
Molecular spectroscopy Origin of spectral lines = absorption, emission and scattering of a photon when the energy of a molecule changes: rad( ) M M * rad( ' ) ' v' 0 0 absorption( ) emission ( ) scattering
Theory of optically thin emission line spectroscopy
 Theory of optically thin emission line spectroscopy 1 Important definitions In general the spectrum of a source consists of a continuum and several line components. Processes which give raise to the continuous
Theory of optically thin emission line spectroscopy 1 Important definitions In general the spectrum of a source consists of a continuum and several line components. Processes which give raise to the continuous
9. RADIATIVE PROCESSES I -- ATOMS AND LIGHT
 QM P453 F95 (Zorn) Atoms in the Radiation Field, I page 9.1 9. RADIATIVE PROCESSES I -- ATOMS AND LIGHT We have discussed the field as a system and described it in terms of excitations of its normal modes.
QM P453 F95 (Zorn) Atoms in the Radiation Field, I page 9.1 9. RADIATIVE PROCESSES I -- ATOMS AND LIGHT We have discussed the field as a system and described it in terms of excitations of its normal modes.
8 Wavefunctions - Schrödinger s Equation
 8 Wavefunctions - Schrödinger s Equation So far we have considered only free particles - i.e. particles whose energy consists entirely of its kinetic energy. In general, however, a particle moves under
8 Wavefunctions - Schrödinger s Equation So far we have considered only free particles - i.e. particles whose energy consists entirely of its kinetic energy. In general, however, a particle moves under
CHAPTER 13 Molecular Spectroscopy 2: Electronic Transitions
 CHAPTER 13 Molecular Spectroscopy 2: Electronic Transitions I. General Features of Electronic spectroscopy. A. Visible and ultraviolet photons excite electronic state transitions. ε photon = 120 to 1200
CHAPTER 13 Molecular Spectroscopy 2: Electronic Transitions I. General Features of Electronic spectroscopy. A. Visible and ultraviolet photons excite electronic state transitions. ε photon = 120 to 1200
NPTEL/IITM. Molecular Spectroscopy Lectures 1 & 2. Prof.K. Mangala Sunder Page 1 of 15. Topics. Part I : Introductory concepts Topics
 Molecular Spectroscopy Lectures 1 & 2 Part I : Introductory concepts Topics Why spectroscopy? Introduction to electromagnetic radiation Interaction of radiation with matter What are spectra? Beer-Lambert
Molecular Spectroscopy Lectures 1 & 2 Part I : Introductory concepts Topics Why spectroscopy? Introduction to electromagnetic radiation Interaction of radiation with matter What are spectra? Beer-Lambert
ATOMIC AND LASER SPECTROSCOPY
 ALAN CORNEY ATOMIC AND LASER SPECTROSCOPY CLARENDON PRESS OXFORD 1977 Contents 1. INTRODUCTION 1.1. Planck's radiation law. 1 1.2. The photoelectric effect 4 1.3. Early atomic spectroscopy 5 1.4. The postulates
ALAN CORNEY ATOMIC AND LASER SPECTROSCOPY CLARENDON PRESS OXFORD 1977 Contents 1. INTRODUCTION 1.1. Planck's radiation law. 1 1.2. The photoelectric effect 4 1.3. Early atomic spectroscopy 5 1.4. The postulates
PAPER No. : 8 (PHYSICAL SPECTROSCOPY) MODULE No. : 5 (TRANSITION PROBABILITIES AND TRANSITION DIPOLE MOMENT. OVERVIEW OF SELECTION RULES)
 Subject Chemistry Paper No and Title Module No and Title Module Tag 8 and Physical Spectroscopy 5 and Transition probabilities and transition dipole moment, Overview of selection rules CHE_P8_M5 TABLE
Subject Chemistry Paper No and Title Module No and Title Module Tag 8 and Physical Spectroscopy 5 and Transition probabilities and transition dipole moment, Overview of selection rules CHE_P8_M5 TABLE
Introduction. Chapter The Purpose of Statistical Mechanics
 Chapter 1 Introduction 1.1 The Purpose of Statistical Mechanics Statistical Mechanics is the mechanics developed to treat a collection of a large number of atoms or particles. Such a collection is, for
Chapter 1 Introduction 1.1 The Purpose of Statistical Mechanics Statistical Mechanics is the mechanics developed to treat a collection of a large number of atoms or particles. Such a collection is, for
- HH Photons and Planck Radiation Law Hsiu-Hau Lin (Oct 23, 2012)
 - HH64 - Photons and Planck Radiation Law Hsiu-Hau Lin hsiuhau.lin@gmail.com (Oct 23, 212) Quantum theory begins with the Planck radiation law for thermal radiation at di erent frequencies, u! = ~ 2 c
- HH64 - Photons and Planck Radiation Law Hsiu-Hau Lin hsiuhau.lin@gmail.com (Oct 23, 212) Quantum theory begins with the Planck radiation law for thermal radiation at di erent frequencies, u! = ~ 2 c
CHM Physical Chemistry II Chapter 12 - Supplementary Material. 1. Einstein A and B coefficients
 CHM 3411 - Physical Chemistry II Chapter 12 - Supplementary Material 1. Einstein A and B coefficients Consider two singly degenerate states in an atom, molecule, or ion, with wavefunctions 1 (for the lower
CHM 3411 - Physical Chemistry II Chapter 12 - Supplementary Material 1. Einstein A and B coefficients Consider two singly degenerate states in an atom, molecule, or ion, with wavefunctions 1 (for the lower
Chapter 39. Particles Behaving as Waves
 Chapter 39 Particles Behaving as Waves 39.1 Electron Waves Light has a dual nature. Light exhibits both wave and particle characteristics. Louis de Broglie postulated in 1924 that if nature is symmetric,
Chapter 39 Particles Behaving as Waves 39.1 Electron Waves Light has a dual nature. Light exhibits both wave and particle characteristics. Louis de Broglie postulated in 1924 that if nature is symmetric,
Introduction to Sources: Radiative Processes and Population Inversion in Atoms, Molecules, and Semiconductors Atoms and Molecules
 OPTI 500 DEF, Spring 2012, Lecture 2 Introduction to Sources: Radiative Processes and Population Inversion in Atoms, Molecules, and Semiconductors Atoms and Molecules Energy Levels Every atom or molecule
OPTI 500 DEF, Spring 2012, Lecture 2 Introduction to Sources: Radiative Processes and Population Inversion in Atoms, Molecules, and Semiconductors Atoms and Molecules Energy Levels Every atom or molecule
14. Structure of Nuclei
 14. Structure of Nuclei Particle and Nuclear Physics Dr. Tina Potter Dr. Tina Potter 14. Structure of Nuclei 1 In this section... Magic Numbers The Nuclear Shell Model Excited States Dr. Tina Potter 14.
14. Structure of Nuclei Particle and Nuclear Physics Dr. Tina Potter Dr. Tina Potter 14. Structure of Nuclei 1 In this section... Magic Numbers The Nuclear Shell Model Excited States Dr. Tina Potter 14.
Fundamentals of Spectroscopy for Optical Remote Sensing. Course Outline 2009
 Fundamentals of Spectroscopy for Optical Remote Sensing Course Outline 2009 Part I. Fundamentals of Quantum Mechanics Chapter 1. Concepts of Quantum and Experimental Facts 1.1. Blackbody Radiation and
Fundamentals of Spectroscopy for Optical Remote Sensing Course Outline 2009 Part I. Fundamentals of Quantum Mechanics Chapter 1. Concepts of Quantum and Experimental Facts 1.1. Blackbody Radiation and
Supplement: Statistical Physics
 Supplement: Statistical Physics Fitting in a Box. Counting momentum states with momentum q and de Broglie wavelength λ = h q = 2π h q In a discrete volume L 3 there is a discrete set of states that satisfy
Supplement: Statistical Physics Fitting in a Box. Counting momentum states with momentum q and de Broglie wavelength λ = h q = 2π h q In a discrete volume L 3 there is a discrete set of states that satisfy
Appendix A. The Particle in a Box: A Demonstration of Quantum Mechanical Principles for a Simple, One-Dimensional, One-Electron Model System
 Appendix A The Particle in a Box: A Demonstration of Quantum Mechanical Principles for a Simple, One-Dimensional, One-Electron Model System Real quantum mechanical systems have the tendency to become mathematically
Appendix A The Particle in a Box: A Demonstration of Quantum Mechanical Principles for a Simple, One-Dimensional, One-Electron Model System Real quantum mechanical systems have the tendency to become mathematically
Physics 221 Lecture 31 Line Radiation from Atoms and Molecules March 31, 1999
 Physics 221 Lecture 31 Line Radiation from Atoms and Molecules March 31, 1999 Reading Meyer-Arendt, Ch. 20; Möller, Ch. 15; Yariv, Ch.. Demonstrations Analyzing lineshapes from emission and absorption
Physics 221 Lecture 31 Line Radiation from Atoms and Molecules March 31, 1999 Reading Meyer-Arendt, Ch. 20; Möller, Ch. 15; Yariv, Ch.. Demonstrations Analyzing lineshapes from emission and absorption
Spectral Resolution. Spectral resolution is a measure of the ability to separate nearby features in wavelength space.
 Spectral Resolution Spectral resolution is a measure of the ability to separate nearby features in wavelength space. R, minimum wavelength separation of two resolved features. Delta lambda often set to
Spectral Resolution Spectral resolution is a measure of the ability to separate nearby features in wavelength space. R, minimum wavelength separation of two resolved features. Delta lambda often set to
24/ Rayleigh and Raman scattering. Stokes and anti-stokes lines. Rotational Raman spectroscopy. Polarizability ellipsoid. Selection rules.
 Subject Chemistry Paper No and Title Module No and Title Module Tag 8/ Physical Spectroscopy 24/ Rayleigh and Raman scattering. Stokes and anti-stokes lines. Rotational Raman spectroscopy. Polarizability
Subject Chemistry Paper No and Title Module No and Title Module Tag 8/ Physical Spectroscopy 24/ Rayleigh and Raman scattering. Stokes and anti-stokes lines. Rotational Raman spectroscopy. Polarizability
Quantum Mechanics-I Prof. Dr. S. Lakshmi Bala Department of Physics Indian Institute of Technology, Madras. Lecture - 21 Square-Integrable Functions
 Quantum Mechanics-I Prof. Dr. S. Lakshmi Bala Department of Physics Indian Institute of Technology, Madras Lecture - 21 Square-Integrable Functions (Refer Slide Time: 00:06) (Refer Slide Time: 00:14) We
Quantum Mechanics-I Prof. Dr. S. Lakshmi Bala Department of Physics Indian Institute of Technology, Madras Lecture - 21 Square-Integrable Functions (Refer Slide Time: 00:06) (Refer Slide Time: 00:14) We
Chem 3502/4502 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 2006 Christopher J. Cramer. Lecture 10, February 10, / 4
 Chem 350/450 Physical Chemistry II (Quantum Mechanics 3 Credits Spring Semester 006 Christopher J. Cramer Lecture 10, February 10, 006 Solved Homework We are asked to find and for the first two
Chem 350/450 Physical Chemistry II (Quantum Mechanics 3 Credits Spring Semester 006 Christopher J. Cramer Lecture 10, February 10, 006 Solved Homework We are asked to find and for the first two
Atomic Structure and Processes
 Chapter 5 Atomic Structure and Processes 5.1 Elementary atomic structure Bohr Orbits correspond to principal quantum number n. Hydrogen atom energy levels where the Rydberg energy is R y = m e ( e E n
Chapter 5 Atomic Structure and Processes 5.1 Elementary atomic structure Bohr Orbits correspond to principal quantum number n. Hydrogen atom energy levels where the Rydberg energy is R y = m e ( e E n
is the minimum stopping potential for which the current between the plates reduces to zero.
 Module 1 :Quantum Mechanics Chapter 2 : Introduction to Quantum ideas Introduction to Quantum ideas We will now consider some experiments and their implications, which introduce us to quantum ideas. The
Module 1 :Quantum Mechanics Chapter 2 : Introduction to Quantum ideas Introduction to Quantum ideas We will now consider some experiments and their implications, which introduce us to quantum ideas. The
Lasers PH 645/ OSE 645/ EE 613 Summer 2010 Section 1: T/Th 2:45-4:45 PM Engineering Building 240
 Lasers PH 645/ OSE 645/ EE 613 Summer 2010 Section 1: T/Th 2:45-4:45 PM Engineering Building 240 John D. Williams, Ph.D. Department of Electrical and Computer Engineering 406 Optics Building - UAHuntsville,
Lasers PH 645/ OSE 645/ EE 613 Summer 2010 Section 1: T/Th 2:45-4:45 PM Engineering Building 240 John D. Williams, Ph.D. Department of Electrical and Computer Engineering 406 Optics Building - UAHuntsville,
List of Comprehensive Exams Topics
 List of Comprehensive Exams Topics Mechanics 1. Basic Mechanics Newton s laws and conservation laws, the virial theorem 2. The Lagrangian and Hamiltonian Formalism The Lagrange formalism and the principle
List of Comprehensive Exams Topics Mechanics 1. Basic Mechanics Newton s laws and conservation laws, the virial theorem 2. The Lagrangian and Hamiltonian Formalism The Lagrange formalism and the principle
Dept. of Physics, MIT Manipal 1
 Chapter 1: Optics 1. In the phenomenon of interference, there is A Annihilation of light energy B Addition of energy C Redistribution energy D Creation of energy 2. Interference fringes are obtained using
Chapter 1: Optics 1. In the phenomenon of interference, there is A Annihilation of light energy B Addition of energy C Redistribution energy D Creation of energy 2. Interference fringes are obtained using
General Chemistry by Ebbing and Gammon, 8th Edition
 Chem 1045 General Chemistry by Ebbing and Gammon, 8th Edition George W.J. Kenney, Jr Last Update: 26-Mar-2009 Chapter 7: Quantum Theory of the Atom These Notes are to SUPPLIMENT the Text, They do NOT Replace
Chem 1045 General Chemistry by Ebbing and Gammon, 8th Edition George W.J. Kenney, Jr Last Update: 26-Mar-2009 Chapter 7: Quantum Theory of the Atom These Notes are to SUPPLIMENT the Text, They do NOT Replace
Addition of Opacities and Absorption
 Addition of Opacities and Absorption If the only way photons could interact was via simple scattering, there would be no blackbodies. We ll go into that in much more detail in the next lecture, but the
Addition of Opacities and Absorption If the only way photons could interact was via simple scattering, there would be no blackbodies. We ll go into that in much more detail in the next lecture, but the
( ) x10 8 m. The energy in a mole of 400 nm photons is calculated by: ' & sec( ) ( & % ) 6.022x10 23 photons' E = h! = hc & 6.
 Introduction to Spectroscopy Spectroscopic techniques are widely used to detect molecules, to measure the concentration of a species in solution, and to determine molecular structure. For proteins, most
Introduction to Spectroscopy Spectroscopic techniques are widely used to detect molecules, to measure the concentration of a species in solution, and to determine molecular structure. For proteins, most
CHAPTER 2: POSTULATES OF QUANTUM MECHANICS
 CHAPTER 2: POSTULATES OF QUANTUM MECHANICS Basics of Quantum Mechanics - Why Quantum Physics? - Classical mechanics (Newton's mechanics) and Maxwell's equations (electromagnetics theory) can explain MACROSCOPIC
CHAPTER 2: POSTULATES OF QUANTUM MECHANICS Basics of Quantum Mechanics - Why Quantum Physics? - Classical mechanics (Newton's mechanics) and Maxwell's equations (electromagnetics theory) can explain MACROSCOPIC
Quantum Mechanics: Discretization of Energy and Wave-Particle Duality
 Quantum Mechanics: Discretization of Energy and Wave-Particle Duality 15th March 2010 1 Introduction Classical physics (derived from the ideas of Newton, Hamilton, Lagrange and other early physicists)
Quantum Mechanics: Discretization of Energy and Wave-Particle Duality 15th March 2010 1 Introduction Classical physics (derived from the ideas of Newton, Hamilton, Lagrange and other early physicists)
Infrared Spectroscopy
 Infrared Spectroscopy The Interaction of Light with Matter Electric fields apply forces to charges, according to F = qe In an electric field, a positive charge will experience a force, but a negative charge
Infrared Spectroscopy The Interaction of Light with Matter Electric fields apply forces to charges, according to F = qe In an electric field, a positive charge will experience a force, but a negative charge
Ch 7 Quantum Theory of the Atom (light and atomic structure)
 Ch 7 Quantum Theory of the Atom (light and atomic structure) Electromagnetic Radiation - Electromagnetic radiation consists of oscillations in electric and magnetic fields. The oscillations can be described
Ch 7 Quantum Theory of the Atom (light and atomic structure) Electromagnetic Radiation - Electromagnetic radiation consists of oscillations in electric and magnetic fields. The oscillations can be described
FIBER OPTICS. Prof. R.K. Shevgaonkar. Department of Electrical Engineering. Indian Institute of Technology, Bombay. Lecture: 15. Optical Sources-LASER
 FIBER OPTICS Prof. R.K. Shevgaonkar Department of Electrical Engineering Indian Institute of Technology, Bombay Lecture: 15 Optical Sources-LASER Fiber Optics, Prof. R.K. Shevgaonkar, Dept. of Electrical
FIBER OPTICS Prof. R.K. Shevgaonkar Department of Electrical Engineering Indian Institute of Technology, Bombay Lecture: 15 Optical Sources-LASER Fiber Optics, Prof. R.K. Shevgaonkar, Dept. of Electrical
Today: general condition for threshold operation physics of atomic, vibrational, rotational gain media intro to the Lorentz model
 Today: general condition for threshold operation physics of atomic, vibrational, rotational gain media intro to the Lorentz model Laser operation Simplified energy conversion processes in a laser medium:
Today: general condition for threshold operation physics of atomic, vibrational, rotational gain media intro to the Lorentz model Laser operation Simplified energy conversion processes in a laser medium:
B2.III Revision notes: quantum physics
 B.III Revision notes: quantum physics Dr D.M.Lucas, TT 0 These notes give a summary of most of the Quantum part of this course, to complement Prof. Ewart s notes on Atomic Structure, and Prof. Hooker s
B.III Revision notes: quantum physics Dr D.M.Lucas, TT 0 These notes give a summary of most of the Quantum part of this course, to complement Prof. Ewart s notes on Atomic Structure, and Prof. Hooker s
Introduction to Quantum Mechanics Prof. Manoj Kumar Harbola Department of Physics Indian Institute of Technology, Kanpur
 Introduction to Quantum Mechanics Prof. Manoj Kumar Harbola Department of Physics Indian Institute of Technology, Kanpur Lecture - 06 Stimulated emission and amplification of light in a LASER (Refer Slide
Introduction to Quantum Mechanics Prof. Manoj Kumar Harbola Department of Physics Indian Institute of Technology, Kanpur Lecture - 06 Stimulated emission and amplification of light in a LASER (Refer Slide
QUANTUM MECHANICS. Franz Schwabl. Translated by Ronald Kates. ff Springer
 Franz Schwabl QUANTUM MECHANICS Translated by Ronald Kates Second Revised Edition With 122Figures, 16Tables, Numerous Worked Examples, and 126 Problems ff Springer Contents 1. Historical and Experimental
Franz Schwabl QUANTUM MECHANICS Translated by Ronald Kates Second Revised Edition With 122Figures, 16Tables, Numerous Worked Examples, and 126 Problems ff Springer Contents 1. Historical and Experimental
Laser Types Two main types depending on time operation Continuous Wave (CW) Pulsed operation Pulsed is easier, CW more useful
 What Makes a Laser Light Amplification by Stimulated Emission of Radiation Main Requirements of the Laser Laser Gain Medium (provides the light amplification) Optical Resonator Cavity (greatly increase
What Makes a Laser Light Amplification by Stimulated Emission of Radiation Main Requirements of the Laser Laser Gain Medium (provides the light amplification) Optical Resonator Cavity (greatly increase
Quantum Physics and General Relativity
 Quantum Physics and General Relativity The self maintained electric potential of the accelerating charges equivalent with the General Relativity space-time curvature, and since it is true on the quantum
Quantum Physics and General Relativity The self maintained electric potential of the accelerating charges equivalent with the General Relativity space-time curvature, and since it is true on the quantum
Mechanics, Heat, Oscillations and Waves Prof. V. Balakrishnan Department of Physics Indian Institute of Technology, Madras
 Mechanics, Heat, Oscillations and Waves Prof. V. Balakrishnan Department of Physics Indian Institute of Technology, Madras Lecture - 21 Central Potential and Central Force Ready now to take up the idea
Mechanics, Heat, Oscillations and Waves Prof. V. Balakrishnan Department of Physics Indian Institute of Technology, Madras Lecture - 21 Central Potential and Central Force Ready now to take up the idea
MOLECULAR SPECTROSCOPY
 MOLECULAR SPECTROSCOPY First Edition Jeanne L. McHale University of Idaho PRENTICE HALL, Upper Saddle River, New Jersey 07458 CONTENTS PREFACE xiii 1 INTRODUCTION AND REVIEW 1 1.1 Historical Perspective
MOLECULAR SPECTROSCOPY First Edition Jeanne L. McHale University of Idaho PRENTICE HALL, Upper Saddle River, New Jersey 07458 CONTENTS PREFACE xiii 1 INTRODUCTION AND REVIEW 1 1.1 Historical Perspective
QUANTUM MECHANICS AND MOLECULAR SPECTROSCOPY
 QUANTUM MECHANICS AND MOLECULAR SPECTROSCOPY CHEM 330 B. O. Owaga Classical physics Classical physics is based on three assumptions i. Predicts precise trajectory for particles with precisely specified
QUANTUM MECHANICS AND MOLECULAR SPECTROSCOPY CHEM 330 B. O. Owaga Classical physics Classical physics is based on three assumptions i. Predicts precise trajectory for particles with precisely specified
Model Answer (Paper code: AR-7112) M. Sc. (Physics) IV Semester Paper I: Laser Physics and Spectroscopy
 Model Answer (Paper code: AR-7112) M. Sc. (Physics) IV Semester Paper I: Laser Physics and Spectroscopy Section I Q1. Answer (i) (b) (ii) (d) (iii) (c) (iv) (c) (v) (a) (vi) (b) (vii) (b) (viii) (a) (ix)
Model Answer (Paper code: AR-7112) M. Sc. (Physics) IV Semester Paper I: Laser Physics and Spectroscopy Section I Q1. Answer (i) (b) (ii) (d) (iii) (c) (iv) (c) (v) (a) (vi) (b) (vii) (b) (viii) (a) (ix)
Physics 9e/Cutnell. correlated to the. College Board AP Physics 2 Course Objectives
 correlated to the College Board AP Physics 2 Course Objectives Big Idea 1: Objects and systems have properties such as mass and charge. Systems may have internal structure. Enduring Understanding 1.A:
correlated to the College Board AP Physics 2 Course Objectives Big Idea 1: Objects and systems have properties such as mass and charge. Systems may have internal structure. Enduring Understanding 1.A:
The Photoelectric Effect
 Stellar Astrophysics: The Interaction of Light and Matter The Photoelectric Effect Methods of electron emission Thermionic emission: Application of heat allows electrons to gain enough energy to escape
Stellar Astrophysics: The Interaction of Light and Matter The Photoelectric Effect Methods of electron emission Thermionic emission: Application of heat allows electrons to gain enough energy to escape
Comments to Atkins: Physical chemistry, 7th edition.
 Comments to Atkins: Physical chemistry, 7th edition. Chapter 16: p. 483, Eq. (16.1). The definition that the wave number is the inverse of the wave length should be used. That is much smarter. p. 483-484.
Comments to Atkins: Physical chemistry, 7th edition. Chapter 16: p. 483, Eq. (16.1). The definition that the wave number is the inverse of the wave length should be used. That is much smarter. p. 483-484.
Energy levels and atomic structures lectures chapter one
 Structure of Atom An atom is the smallest constituent unit of ordinary matter that has the properties of a element. Every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Atoms are
Structure of Atom An atom is the smallest constituent unit of ordinary matter that has the properties of a element. Every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Atoms are
Problem 1. Hyperfine Emission from Neutral Hydrogen
 Ay 201 Radiative Processes Problem Set 4 Solutions Linda Strubbe and Eugene Chiang October 2, 2003 Problem 1. Hyperfine Emission from Neutral Hydrogen This problem is an exercise in learning more astronomy
Ay 201 Radiative Processes Problem Set 4 Solutions Linda Strubbe and Eugene Chiang October 2, 2003 Problem 1. Hyperfine Emission from Neutral Hydrogen This problem is an exercise in learning more astronomy
AP PHYSICS 2 FRAMEWORKS
 1 AP PHYSICS 2 FRAMEWORKS Big Ideas Essential Knowledge Science Practices Enduring Knowledge Learning Objectives ELECTRIC FORCE, FIELD AND POTENTIAL Static Electricity; Electric Charge and its Conservation
1 AP PHYSICS 2 FRAMEWORKS Big Ideas Essential Knowledge Science Practices Enduring Knowledge Learning Objectives ELECTRIC FORCE, FIELD AND POTENTIAL Static Electricity; Electric Charge and its Conservation
Quantum Mechanics & Atomic Structure (Chapter 11)
 Quantum Mechanics & Atomic Structure (Chapter 11) Quantum mechanics: Microscopic theory of light & matter at molecular scale and smaller. Atoms and radiation (light) have both wave-like and particlelike
Quantum Mechanics & Atomic Structure (Chapter 11) Quantum mechanics: Microscopic theory of light & matter at molecular scale and smaller. Atoms and radiation (light) have both wave-like and particlelike
CHAPTER 13 LECTURE NOTES
 CHAPTER 13 LECTURE NOTES Spectroscopy is concerned with the measurement of (a) the wavelengths (or frequencies) at which molecules absorb/emit energy, and (b) the amount of radiation absorbed at these
CHAPTER 13 LECTURE NOTES Spectroscopy is concerned with the measurement of (a) the wavelengths (or frequencies) at which molecules absorb/emit energy, and (b) the amount of radiation absorbed at these
Rotation and vibration of Molecules
 Rotation and vibration of Molecules Overview of the two lectures... 2 General remarks on spectroscopy... 2 Beer-Lambert law for photoabsorption... 3 Einstein s coefficients... 4 Limits of resolution...
Rotation and vibration of Molecules Overview of the two lectures... 2 General remarks on spectroscopy... 2 Beer-Lambert law for photoabsorption... 3 Einstein s coefficients... 4 Limits of resolution...
Teaching philosophy. learn it, know it! Learn it 5-times and you know it Read (& simple question) Lecture Problem set
 Learn it 5-times and you know it Read (& simple question) Lecture Problem set Teaching philosophy Review/work-problems for Mid-term exam Review/re-work for Final exam Hand in homework every Monday (1 per
Learn it 5-times and you know it Read (& simple question) Lecture Problem set Teaching philosophy Review/work-problems for Mid-term exam Review/re-work for Final exam Hand in homework every Monday (1 per
Chemistry 3502/4502. Final Exam Part I. May 14, 2005
 Advocacy chit Chemistry 350/450 Final Exam Part I May 4, 005. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle
Advocacy chit Chemistry 350/450 Final Exam Part I May 4, 005. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle
1 Longitudinal modes of a laser cavity
 Adrian Down May 01, 2006 1 Longitudinal modes of a laser cavity 1.1 Resonant modes For the moment, imagine a laser cavity as a set of plane mirrors separated by a distance d. We will return to the specific
Adrian Down May 01, 2006 1 Longitudinal modes of a laser cavity 1.1 Resonant modes For the moment, imagine a laser cavity as a set of plane mirrors separated by a distance d. We will return to the specific
What are Lasers? Light Amplification by Stimulated Emission of Radiation LASER Light emitted at very narrow wavelength bands (monochromatic) Light
 What are Lasers? What are Lasers? Light Amplification by Stimulated Emission of Radiation LASER Light emitted at very narrow wavelength bands (monochromatic) Light emitted in a directed beam Light is coherenent
What are Lasers? What are Lasers? Light Amplification by Stimulated Emission of Radiation LASER Light emitted at very narrow wavelength bands (monochromatic) Light emitted in a directed beam Light is coherenent
Particle nature of light & Quantization
 Particle nature of light & Quantization A quantity is quantized if its possible values are limited to a discrete set. An example from classical physics is the allowed frequencies of standing waves on a
Particle nature of light & Quantization A quantity is quantized if its possible values are limited to a discrete set. An example from classical physics is the allowed frequencies of standing waves on a
PHYS 172: Modern Mechanics Fall 2009
 PHYS 172: Modern Mechanics Fall 2009 Lecture 14 Energy Quantization Read 7.1 7.9 Reading Question: Ch. 7, Secs 1-5 A simple model for the hydrogen atom treats the electron as a particle in circular orbit
PHYS 172: Modern Mechanics Fall 2009 Lecture 14 Energy Quantization Read 7.1 7.9 Reading Question: Ch. 7, Secs 1-5 A simple model for the hydrogen atom treats the electron as a particle in circular orbit
Collisionally Excited Spectral Lines (Cont d) Diffuse Universe -- C. L. Martin
 Collisionally Excited Spectral Lines (Cont d) Please Note: Contrast the collisionally excited lines with the H and He lines in the Orion Nebula spectrum. Preview: Pure Recombination Lines Recombination
Collisionally Excited Spectral Lines (Cont d) Please Note: Contrast the collisionally excited lines with the H and He lines in the Orion Nebula spectrum. Preview: Pure Recombination Lines Recombination
What Makes a Laser Light Amplification by Stimulated Emission of Radiation Main Requirements of the Laser Laser Gain Medium (provides the light
 What Makes a Laser Light Amplification by Stimulated Emission of Radiation Main Requirements of the Laser Laser Gain Medium (provides the light amplification) Optical Resonator Cavity (greatly increase
What Makes a Laser Light Amplification by Stimulated Emission of Radiation Main Requirements of the Laser Laser Gain Medium (provides the light amplification) Optical Resonator Cavity (greatly increase
Conclusion. 109m Ag isomer showed that there is no such broadening. Because one can hardly
 Conclusion This small book presents a description of the results of studies performed over many years by our research group, which, in the best period, included 15 physicists and laboratory assistants
Conclusion This small book presents a description of the results of studies performed over many years by our research group, which, in the best period, included 15 physicists and laboratory assistants
Stellar Astrophysics: The Interaction of Light and Matter
 Stellar Astrophysics: The Interaction of Light and Matter The Photoelectric Effect Methods of electron emission Thermionic emission: Application of heat allows electrons to gain enough energy to escape
Stellar Astrophysics: The Interaction of Light and Matter The Photoelectric Effect Methods of electron emission Thermionic emission: Application of heat allows electrons to gain enough energy to escape
Review of the Formalism of Quantum Mechanics
 Review of the Formalism of Quantum Mechanics The postulates of quantum mechanics are often stated in textbooks. There are two main properties of physics upon which these postulates are based: 1)the probability
Review of the Formalism of Quantum Mechanics The postulates of quantum mechanics are often stated in textbooks. There are two main properties of physics upon which these postulates are based: 1)the probability
IV. Classical Statistical Mechanics
 IV. Classical Statistical Mechanics IV.A General Definitions Statistical Mechanics is a probabilistic approach to equilibrium macroscopic properties of large numbers of degrees of freedom. As discussed
IV. Classical Statistical Mechanics IV.A General Definitions Statistical Mechanics is a probabilistic approach to equilibrium macroscopic properties of large numbers of degrees of freedom. As discussed
Semiconductor Physics and Devices
 Introduction to Quantum Mechanics In order to understand the current-voltage characteristics, we need some knowledge of electron behavior in semiconductor when the electron is subjected to various potential
Introduction to Quantum Mechanics In order to understand the current-voltage characteristics, we need some knowledge of electron behavior in semiconductor when the electron is subjected to various potential
Which of the following classes of electromagnetic waves will not ionise neutral atoms?
 1 In an experiment to demonstrate the photoelectric effect, a charged metal plate is illuminated with light from different sources. The plate loses its charge when an ultraviolet light source is used but
1 In an experiment to demonstrate the photoelectric effect, a charged metal plate is illuminated with light from different sources. The plate loses its charge when an ultraviolet light source is used but
The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of hydrogen atom.
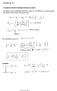 Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
Lecture 6 - spectroscopy
 Lecture 6 - spectroscopy 1 Light Electromagnetic radiation can be thought of as either a wave or as a particle (particle/wave duality). For scattering of light by particles, air, and surfaces, wave theory
Lecture 6 - spectroscopy 1 Light Electromagnetic radiation can be thought of as either a wave or as a particle (particle/wave duality). For scattering of light by particles, air, and surfaces, wave theory
V( x) = V( 0) + dv. V( x) = 1 2
 Spectroscopy 1: rotational and vibrational spectra The vibrations of diatomic molecules Molecular vibrations Consider a typical potential energy curve for a diatomic molecule. In regions close to R e (at
Spectroscopy 1: rotational and vibrational spectra The vibrations of diatomic molecules Molecular vibrations Consider a typical potential energy curve for a diatomic molecule. In regions close to R e (at
RFSS: Lecture 6 Gamma Decay
 RFSS: Lecture 6 Gamma Decay Readings: Modern Nuclear Chemistry, Chap. 9; Nuclear and Radiochemistry, Chapter 3 Energetics Decay Types Transition Probabilities Internal Conversion Angular Correlations Moessbauer
RFSS: Lecture 6 Gamma Decay Readings: Modern Nuclear Chemistry, Chap. 9; Nuclear and Radiochemistry, Chapter 3 Energetics Decay Types Transition Probabilities Internal Conversion Angular Correlations Moessbauer
Radiating Dipoles in Quantum Mechanics
 Radiating Dipoles in Quantum Mechanics Chapter 14 P. J. Grandinetti Chem. 4300 Oct 27, 2017 P. J. Grandinetti (Chem. 4300) Radiating Dipoles in Quantum Mechanics Oct 27, 2017 1 / 26 P. J. Grandinetti (Chem.
Radiating Dipoles in Quantum Mechanics Chapter 14 P. J. Grandinetti Chem. 4300 Oct 27, 2017 P. J. Grandinetti (Chem. 4300) Radiating Dipoles in Quantum Mechanics Oct 27, 2017 1 / 26 P. J. Grandinetti (Chem.
Chapter 7 Atomic Structure -1 Quantum Model of Atom. Dr. Sapna Gupta
 Chapter 7 Atomic Structure -1 Quantum Model of Atom Dr. Sapna Gupta The Electromagnetic Spectrum The electromagnetic spectrum includes many different types of radiation which travel in waves. Visible light
Chapter 7 Atomic Structure -1 Quantum Model of Atom Dr. Sapna Gupta The Electromagnetic Spectrum The electromagnetic spectrum includes many different types of radiation which travel in waves. Visible light
A few principles of classical and quantum mechanics
 A few principles of classical and quantum mechanics The classical approach: In classical mechanics, we usually (but not exclusively) solve Newton s nd law of motion relating the acceleration a of the system
A few principles of classical and quantum mechanics The classical approach: In classical mechanics, we usually (but not exclusively) solve Newton s nd law of motion relating the acceleration a of the system
Non-stationary States and Electric Dipole Transitions
 Pre-Lab Lecture II Non-stationary States and Electric Dipole Transitions You will recall that the wavefunction for any system is calculated in general from the time-dependent Schrödinger equation ĤΨ(x,t)=i
Pre-Lab Lecture II Non-stationary States and Electric Dipole Transitions You will recall that the wavefunction for any system is calculated in general from the time-dependent Schrödinger equation ĤΨ(x,t)=i
Chemistry 3502/4502. Final Exam Part I. May 14, 2005
 Chemistry 3502/4502 Final Exam Part I May 14, 2005 1. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle (e) The
Chemistry 3502/4502 Final Exam Part I May 14, 2005 1. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle (e) The
Quantum Electronics/Laser Physics Chapter 4 Line Shapes and Line Widths
 Quantum Electronics/Laser Physics Chapter 4 Line Shapes and Line Widths 4.1 The Natural Line Shape 4.2 Collisional Broadening 4.3 Doppler Broadening 4.4 Einstein Treatment of Stimulated Processes Width
Quantum Electronics/Laser Physics Chapter 4 Line Shapes and Line Widths 4.1 The Natural Line Shape 4.2 Collisional Broadening 4.3 Doppler Broadening 4.4 Einstein Treatment of Stimulated Processes Width
Lecture 10. Lidar Effective Cross-Section vs. Convolution
 Lecture 10. Lidar Effective Cross-Section vs. Convolution q Introduction q Convolution in Lineshape Determination -- Voigt Lineshape (Lorentzian Gaussian) q Effective Cross Section for Single Isotope --
Lecture 10. Lidar Effective Cross-Section vs. Convolution q Introduction q Convolution in Lineshape Determination -- Voigt Lineshape (Lorentzian Gaussian) q Effective Cross Section for Single Isotope --
What are Lasers? Light Amplification by Stimulated Emission of Radiation LASER Light emitted at very narrow wavelength bands (monochromatic) Light
 What are Lasers? What are Lasers? Light Amplification by Stimulated Emission of Radiation LASER Light emitted at very narrow wavelength bands (monochromatic) Light emitted in a directed beam Light is coherenent
What are Lasers? What are Lasers? Light Amplification by Stimulated Emission of Radiation LASER Light emitted at very narrow wavelength bands (monochromatic) Light emitted in a directed beam Light is coherenent
MOLECULAR ENERGY LEVELS DR IMRANA ASHRAF
 MOLECULAR ENERGY LEVELS DR IMRANA ASHRAF OUTLINE q q q q MOLECULE MOLECULAR ORBITAL THEORY MOLECULAR TRANSITIONS INTERACTION OF RADIATION WITH MATTER q TYPES OF MOLECULAR ENERGY LEVELS q MOLECULE q In
MOLECULAR ENERGY LEVELS DR IMRANA ASHRAF OUTLINE q q q q MOLECULE MOLECULAR ORBITAL THEORY MOLECULAR TRANSITIONS INTERACTION OF RADIATION WITH MATTER q TYPES OF MOLECULAR ENERGY LEVELS q MOLECULE q In
The Particle in a Box
 Page 324 Lecture 17: Relation of Particle in a Box Eigenstates to Position and Momentum Eigenstates General Considerations on Bound States and Quantization Continuity Equation for Probability Date Given:
Page 324 Lecture 17: Relation of Particle in a Box Eigenstates to Position and Momentum Eigenstates General Considerations on Bound States and Quantization Continuity Equation for Probability Date Given:
Dielectrics. Lecture 20: Electromagnetic Theory. Professor D. K. Ghosh, Physics Department, I.I.T., Bombay
 What are dielectrics? Dielectrics Lecture 20: Electromagnetic Theory Professor D. K. Ghosh, Physics Department, I.I.T., Bombay So far we have been discussing electrostatics in either vacuum or in a conductor.
What are dielectrics? Dielectrics Lecture 20: Electromagnetic Theory Professor D. K. Ghosh, Physics Department, I.I.T., Bombay So far we have been discussing electrostatics in either vacuum or in a conductor.
84 My God, He Plays Dice! Chapter 12. Irreversibility. This chapter on the web informationphilosopher.com/problems/reversibility
 84 My God, He Plays Dice! This chapter on the web informationphilosopher.com/problems/reversibility Microscopic In the 1870 s, Ludwig Boltzmann developed his transport equation and his dynamical H-theorem
84 My God, He Plays Dice! This chapter on the web informationphilosopher.com/problems/reversibility Microscopic In the 1870 s, Ludwig Boltzmann developed his transport equation and his dynamical H-theorem
Low Emittance Machines
 Advanced Accelerator Physics Course RHUL, Egham, UK September 2017 Low Emittance Machines Part 1: Beam Dynamics with Synchrotron Radiation Andy Wolski The Cockcroft Institute, and the University of Liverpool,
Advanced Accelerator Physics Course RHUL, Egham, UK September 2017 Low Emittance Machines Part 1: Beam Dynamics with Synchrotron Radiation Andy Wolski The Cockcroft Institute, and the University of Liverpool,
Optical Spectroscopy 1 1. Absorption spectroscopy (UV/vis)
 Optical Spectroscopy 1 1. Absorption spectroscopy (UV/vis) 2 2. Circular dichroism (optical activity) CD / ORD 3 3. Fluorescence spectroscopy and energy transfer Electromagnetic Spectrum Electronic Molecular
Optical Spectroscopy 1 1. Absorption spectroscopy (UV/vis) 2 2. Circular dichroism (optical activity) CD / ORD 3 3. Fluorescence spectroscopy and energy transfer Electromagnetic Spectrum Electronic Molecular
Laser Cooling of Gallium. Lauren Rutherford
 Laser Cooling of Gallium Lauren Rutherford Laser Cooling Cooling mechanism depends on conservation of momentum during absorption and emission of radiation Incoming photons Net momentum transfer to atom
Laser Cooling of Gallium Lauren Rutherford Laser Cooling Cooling mechanism depends on conservation of momentum during absorption and emission of radiation Incoming photons Net momentum transfer to atom
2) The energy of a photon of light is proportional to its frequency and proportional to its wavelength.
 Advanced Chemistry Chapter 13 Review Name Per Show all work Wave Properties 1) Which one of the following is correct? A) ν + λ = c B) ν λ = c C) ν = cλ D) λ = c ν E) νλ = c 2) The energy of a photon of
Advanced Chemistry Chapter 13 Review Name Per Show all work Wave Properties 1) Which one of the following is correct? A) ν + λ = c B) ν λ = c C) ν = cλ D) λ = c ν E) νλ = c 2) The energy of a photon of
de = j ν dvdωdtdν. (1)
 Transfer Equation and Blackbodies Initial questions: There are sources in the centers of some galaxies that are extraordinarily bright in microwaves. What s going on? The brightest galaxies in the universe
Transfer Equation and Blackbodies Initial questions: There are sources in the centers of some galaxies that are extraordinarily bright in microwaves. What s going on? The brightest galaxies in the universe
 Math Questions for the 2011 PhD Qualifier Exam 1. Evaluate the following definite integral 3" 4 where! ( x) is the Dirac! - function. # " 4 [ ( )] dx x 2! cos x 2. Consider the differential equation dx
Math Questions for the 2011 PhD Qualifier Exam 1. Evaluate the following definite integral 3" 4 where! ( x) is the Dirac! - function. # " 4 [ ( )] dx x 2! cos x 2. Consider the differential equation dx
van Quantum tot Molecuul
 10 HC10: Molecular and vibrational spectroscopy van Quantum tot Molecuul Dr Juan Rojo VU Amsterdam and Nikhef Theory Group http://www.juanrojo.com/ j.rojo@vu.nl Molecular and Vibrational Spectroscopy Based
10 HC10: Molecular and vibrational spectroscopy van Quantum tot Molecuul Dr Juan Rojo VU Amsterdam and Nikhef Theory Group http://www.juanrojo.com/ j.rojo@vu.nl Molecular and Vibrational Spectroscopy Based
CHAPTER 8 The Quantum Theory of Motion
 I. Translational motion. CHAPTER 8 The Quantum Theory of Motion A. Single particle in free space, 1-D. 1. Schrodinger eqn H ψ = Eψ! 2 2m d 2 dx 2 ψ = Eψ ; no boundary conditions 2. General solution: ψ
I. Translational motion. CHAPTER 8 The Quantum Theory of Motion A. Single particle in free space, 1-D. 1. Schrodinger eqn H ψ = Eψ! 2 2m d 2 dx 2 ψ = Eψ ; no boundary conditions 2. General solution: ψ
6. Molecular structure and spectroscopy I
 6. Molecular structure and spectroscopy I 1 6. Molecular structure and spectroscopy I 1 molecular spectroscopy introduction 2 light-matter interaction 6.1 molecular spectroscopy introduction 2 Molecular
6. Molecular structure and spectroscopy I 1 6. Molecular structure and spectroscopy I 1 molecular spectroscopy introduction 2 light-matter interaction 6.1 molecular spectroscopy introduction 2 Molecular
International Physics Course Entrance Examination Questions
 International Physics Course Entrance Examination Questions (May 2010) Please answer the four questions from Problem 1 to Problem 4. You can use as many answer sheets you need. Your name, question numbers
International Physics Course Entrance Examination Questions (May 2010) Please answer the four questions from Problem 1 to Problem 4. You can use as many answer sheets you need. Your name, question numbers
Saturation Absorption Spectroscopy of Rubidium Atom
 Saturation Absorption Spectroscopy of Rubidium Atom Jayash Panigrahi August 17, 2013 Abstract Saturated absorption spectroscopy has various application in laser cooling which have many relevant uses in
Saturation Absorption Spectroscopy of Rubidium Atom Jayash Panigrahi August 17, 2013 Abstract Saturated absorption spectroscopy has various application in laser cooling which have many relevant uses in
Topics for the Qualifying Examination
 Topics for the Qualifying Examination Quantum Mechanics I and II 1. Quantum kinematics and dynamics 1.1 Postulates of Quantum Mechanics. 1.2 Configuration space vs. Hilbert space, wave function vs. state
Topics for the Qualifying Examination Quantum Mechanics I and II 1. Quantum kinematics and dynamics 1.1 Postulates of Quantum Mechanics. 1.2 Configuration space vs. Hilbert space, wave function vs. state
Rb, which had been compressed to a density of 1013
 Modern Physics Study Questions for the Spring 2018 Departmental Exam December 3, 2017 1. An electron is initially at rest in a uniform electric field E in the negative y direction and a uniform magnetic
Modern Physics Study Questions for the Spring 2018 Departmental Exam December 3, 2017 1. An electron is initially at rest in a uniform electric field E in the negative y direction and a uniform magnetic
