ELECTRON LOSS AND CAPTURE BY kev HELIUM IONS IN VARIOUS GASES
|
|
|
- Derek James
- 5 years ago
- Views:
Transcription
1 SOVET PHYSCS JETP VOLUME 14, NUMBER 1 JANUARY, 196 ELECTRON LOSS AND CAPTURE BY kev HELUM ONS N VAROUS GASES L.. PVOVAR, V. M. TUBAEV, and M. T. NOVKOV Khar'kov Physico-Technical nstitute, Academy of Sciences, Ukrainian S.S.R. Submitted to JETP editor February 7, 1961 J. Exptl. Theoret. Phys. (U.S.S.R.) 41, 6-31 (July, 1961) The cross sections for electron loss u 1 and electron capture u 10 by singly charged helium ions in single collisions in gases have been measured. The relative amounts of He 0, He+, and He + ions in beams of equilibrium composition traversing the gases have also been measured. The cross section for electron capture by doubly charged helium ions u 1 and the cross section for electron loss by helium atoms u 01 are estimated from the ratios of these components. The gas targets consisted of helium, nitrogen, argon, and krypton. The measurements were performed for ion energies between 00 and 100 kev. 1. NTRODUCTON A number of studies have been devoted to chargeexchange processes involving helium atoms and ions in collisions with gas molecules. A summary of the basic experimental results is given in the review article of Allison. [11 This review article gives the results of experimental investigations on the loss and capture of electrons by helium ions and atoms in collisions with molecules of H, N, 0, He, Ne, Ar, Kr, and Xe in the kev interval and with molecules of H, He, and air up to an energy of 40 kev. The article also gives separate data for the charge exchange of a particles in air obtained at high energies by Rutherford. Extenl?ion of the investigations in the direction of higher energies is of interest from the viewpoint of further development of the theory of atomic collisions. n the present article, we describe the results of measurements of the effective cross section for the loss and capture of an electron by helium ions and also the results of measurements of collisions between helium particle beams of equilibrium composition and H and N molecules and He, Ar, and Kr atoms in the kev interval.. APPARATUS AND EXPERMENTAL METHOD n this experiment, we used the arrangement employed by us earlier [1 for the measurement of the cross sections for the dissociation of molecular ions of hydrogen. The method of measurement was basically the same. 0 The primary beam of single charged helium ions produced by an electrostatic accelerator was separated by a mass monochromator and passed through the collision chamber. The He+ and He+ components which were formed were separated by an electrostatic analyzer and entered Faraday cups. The currents were measured by EMU-3 vacuum-tube electrometers. The intensity of the beam of neutral He 0 particles was recorded with a detector which measured the secondary electron emission current arising from the bombardment of copper foil by the fast helium atoms. The basic design of this detector is the same as that described by Stier, Barnett, and Evans. [JJ The detector was calibrated with an auxiliary detector consisting of a thin copper foil and a copperconstantan thermocouple mounted at the center of the foil (on the rear side). This foil too was bornbarded by the He 0 beam. Since the difference in temperature at the ends of the thermocouple did not exceed 0.1 C during the measurements, the thermal emf was proportional to the intensity of the beam of particles bombarding the foil. The size of the secondary electron emission current was measured with an EMU-3 vacuum-tube electrometer. The beam component currents were measured simultaneously, which made it possible to decrease the error caused by oscillations in the primary ion beam intensity. The measurements of the cross sections for electron capture u 10 and electron loss u 1 by singly charged helium ions were made by the mass-spectrographic method. The cross sections were calculated from the formulas
2 ELECT R 0 N L 0 S S AND CAPTURE BY k e v HELUM 0 N S 1 where N, N+, and N+ are the amounts of neutral atoms, singly charged ions, and doubly charged ions of helium in the beam traversing the collision chamber; n is the concentration of the gas atoms in the chamber; L is the effective length of the chamber; the subscript b indicates background. For each case, we plotted the ratio of the number of secondary particles to the number of primary particles as a function of nl. We determined the cross sections u 10 and u 1 from the linear part of these curves. n all cases, we checked the influence of the difference in the scattering of secondary and primary beam particles by means of diaphragms at the exit channel of the collision chamber. Under the geometrical conditions of our experiment (beam diameter mm and channel diameter 6. mm), the scattering had no influence on the size of the measured cross sections, within the limits of experimental error. The cross sections u 10 and u 1 were calculated as the mean of two or three independent measurements. The random errors of measurement did not exceed ± 1% for the cross sections u 1 and ± 18% for the cross sections u 10 The primary ion beam energy was determined to an accuracy of ± %. For the measurements of the equilibrium components of the beam, entrance and exit channels, each of diameter. mm and length 80 mm, were mounted in the collision chamber, which had an over-all length of 400 mm. The beam entered the collision chamber through a diaphragm 1.6 mm in diameter mounted in front of the entrance channel. The large diffusion resistances of the channels made it possible to use target thicknesses of about (3-) x molecules per cm At each value of the primary ion beam energy, a check was made to establish whether the charge distribution of the beam had attained a stationary value. To do this, we investigated the dependence of the size of the He 0, He+, and He + components of the beam on the gas pressure in the collision chamber close to the equilibrium composition. n the ion energy region studied by us, we could neglect the influence of the process of formation of negative helium ions [41 and assume that the above-mentioned three components were present in the 00 to kev energy interval and that effectively only two components, He+ and He +, were present in the to 100 kev interval, (1) () since at kev the He 0 is already present in the beam in quantities no greater than 6% of the total number of particles, and its content drops very quickly with an increase in energy. f we also neglect the influence of the two-electron loss and capture processes, then we can estimate the electron capture cross section u 1 and electron loss cross section u 01 from the following relations: [1J where u 1 and u 10 are the cross sections for electron loss and capture by singly charged helium ions; F Ocx:> F icx:> and F CX) are the relative contents of the He 0, He+, and He + components in a beam of equilibrium composition. n order to check whether differences in the scattering of the beam components affect their size, we changed the collision chamber channels. t turned out that, when the 6.-mm diam channels were replaced by.-mm diam channels, the ratios of the beam components remained unchanged within the limits of experimental error. The random errors in the measurement of the equilibrium composition were about ± % for the F icx:> and F CX) components and ± 11% for the F Ocx:> component. 3. RESULTS OF MEASUREMENTS AND DSCUS SON n this experiment, we measured the cross sections for electron capture and loss by singly charged helium ions in collisions with H, N molecules and He, Ar, and Kr atoms. We used the following gases as targets: hydrogen admitted through a palladium filter, helium with impurities not exceeding 0.1 %, nitrogen with 0.03% impurities, and argon and krypton with 0.1% impurities. The results of the measurements of equilibrium compositions in the helium beam are shown in the table. The equilibrium components of the helium beam in some gases have been measured and calculated by other authors in the 8- to 40-kev interval. The data of the present experiment are in satisfactory agreement with the data calculated by Allison [1J for hydrogen, helium, and air and with the measurements of Snitzer [1 in argon. At 00 kev, our data fit together with the data of Barnett and Stier[6J for hydrogen, helium, nitrogen, and argon. Owing to the small cross sections for electron capture by He+ and He + ions, the neutral component of the beam F 0 CX) did not attain equilibrium for the target thicknesses used at energies above kev (above 600 kev in the case of hydrogen and above 900 kev for the F icx:> component). (3)
3 PVOVAR, TUBAEV, and NOVKOV F ooo F 00 Hydrogen Helium il Nitrogen , o:o 0. OJ 700 (0.0) (0.010) i (0.006) (0.03) '> 1000 (0.004) (0.) (0.0) (0.00) (0.17) 0.88 (0.013) (0.006) (0.001) (0.1) 0,874 (0.01) ,68 (0.0037) 0, (0,001) (0.10) (0.007) (0.003) (0.001) (0.09) (0.00) (0.00) (0.07) 0.93 (0.003) ,877 Argon Krypton , , ! (0.006) (0.003) (0,004) 0, (0.00) (0.003) (0.001) ,9 (0.00) (0;001) (0.0016) 0, (0,001) ,96 (0.0014) The corresponding values of F oco and F leo are therefore shown in parentheses in the table. The results of the measurements of the cross sections for electron loss a 1 and electron capture a 10 as a function of the He+ ion energy are shown in Figs. 1-. Also shown in these figures are the values of the cross sections for electron capture and loss a 1 and a 01 calculated from formula (3). Wherever possible, we give for comparison the data of Allison et al, [lj who measured the corresponding cross sections in the kev interval in hydrogen, helium, and air. Our values are in satisfactory agreement with their data. As seen from the figures, the cross sections for electron loss a 1 and a 01 for all the gases studied by us increase with energy, attain a broad maximum, and then slowly decrease with a further increase in the energy of the He+ ions. The cross sections for electron capture a 10 and a 1 rapidly decrease monotonically with an increa~e in energy over the entire investigated interval. The values of the cross sections depend on the kind of target gas and increase with the atomic number of the target substance. Also shown in Fig. 1 are the values of the cross sections for electron capture by doubly charged helium ions in hydrogen as calculated by Schiff. [7J u, cm /atom ~ 7 ll. ~ il!jot J,z 10 a;cm /atom 16-~ ~ ~ t:f- ao... a,z "'-. "' ~, ~ a,, a,o- - "' "o ZOO BOO FG. 1. Cross section for electron loss and capture by helium ions in hydrogen. 6- values of a 10 from refe~ence 1, A - values of u ; from reference 1; dotted curve - according to data of reference 7. % a,, ""-.. ;... a,o /9!O BOO FG.. Cross section for electron loss and capture by helium ions in helium 6- values of a 10 from reference 1, A- values of u; from reference 1; dotted curve- according to data of reference 7.
4 E L E C T R 0 N L 0 S S AND C A P T U R E BY k e v H E L U M 0 N S 3 a; cm /atom c--- u... o---,- "" ~"':;;-- 6 " > Oot v Ot "' ~ ~ ~ '\,... ~ ' "" ~to H... _l_ ~ FG. 3. Cross section for electron loss and capture by helium ions in nitrogen.!::. -data from reference 1 for a; 0 in air, A - data from reference 1 for a; in air. i measurement of the equilibrium components F 100 and F oo. Schiff [ 7 J also calculated the cross sections for electron capture a 10 by He+ ions in helium by means of the Born approximation. The corresponding values of a 10 are shown in Fig. by the dotted curve. The theoretical curve systematically lies 0-40% below the experimental curve; the difference between them increases with energy. We note that this position of the theoretical curve with respect to the experimental curve of Fig. was predicted by Schiff for ion velocities v > v 0 (v 0 = q /ti), which occurs in this case. Since we measured the cross sections for ion velocities up to v ~ 4v 0, it is of interest to compare the data obtained in the velocity interval between v "' 3v 0 and v "' 4v 0 with the qualitative results obtained by Bohr for a 1 and a 1 in the case of light ions at velocities v» v 0 [BJ Assuming that the nucleus and the electron of the target atom act independently on the ion, Bohr obtained the following expression for light atoms: For substances of larger atomic number ( Z 1 :s zf 3 ), where the electrons and nucleus of the target atom do not act independently, (4) FG. 4. Cross section for electron loss and capture by helium ions in argon., a, cm jatom 1g- ts !'\ '"' ~ - ""' '-n. (lot - ty,,-. - a,, --..._,... -o... o,o to ta FG.. Cross section for electron loss and capture by helium ions in krypton. The theoretical and experimental curves in the kev interval are in good agreement. t should be kept in mind, however, that such close agreement can be more or less accidental, if only because the experimental values of a 1 are given with a large error-this includes both the error of measurement of a 1 and the error of v1 ~ volv. () For electron capture by an a particle in the case of atoms that are not very light, Bohr obtained the expression The experimental results do not strictly follow these functions of the ion velocity, but for H and He the decrease in the cross sections for electron loss a 1 takes place more rapidly than for N, Ar, and Kr, which is in qualitative agreement with Bohr's conclusions. The dependence of a 1 on the ion velocity obtained in this experiment for N, Ar, and Kr differs from expression (6) in the value of the exponent. The corresponding expressions can be written as follows: for nitrogen, a 1 "' (v 0 /v ) 6 9 ; for argon, a 1 "' (v 0 /v ) 6 7 ; and for krypton, a 1 "' (v 0 /v)4.7. We note that Barnett and Reynolds[9J obtained similar results for hydrogen ion energies up to 1000 kev. For nitrogen, the exponent turned out to be "'6. and for argon n conclusion, the authors consider it their duty to express their gratitude to Academician A. K. Val'ter for his interest in and attention to this work. (6)
5 4 PVOVAR, TUBAEV, and NOVKOV 1 S. K. Allison, Revs. Modern Phys. 30, 1137 (198). Pi vovar, Tubaev, and Novikov, JE TP 40, 34 (1961). Soviet Phys. JETP 13, 3 (1961). 3 Stier, Barnett, and Evans, Phys. Rev. 96, 973 (194). 4Dukel'skii, Afrosimov, and Fedorenko, JETP 30, 79 (196), Soviet Phys. JETP 3, 764 (196). E. Snitzer, Phys. Rev. 89, 137 (193). 6 C. F. Barnett and P.M. Stier, Phys. Rev. 109, 38 (198). 7 H. Schiff, Can. J. Phys. 3, 393 (194). 8 N. Bohr, The Passage of Atomic Particles through Matter, Kgl. Danske Videnskab. Selskab, Mat.-fys. Medd 18, No. 8 (1948). 9 c. F. Barnett and H. K. Reynolds, Phys. Rev. 109, 3 (198). Translated by E. Marquit 6
IONIZATION OF GASES BY He IONS WITH ENERGY MeV
 SOVIET PHYSICS JE TP VOLUME 27, NUMBER 5 NOVEMBER, 1968 IONIZATION OF GASES BY He IONS WITH ENERGY 0.2-1.8 MeV L. I. PIVOVAR, Yu. Z. LEVCHENKO, and A. N. GRIGOR'EV Physico-technical Institute, Ukrainian
SOVIET PHYSICS JE TP VOLUME 27, NUMBER 5 NOVEMBER, 1968 IONIZATION OF GASES BY He IONS WITH ENERGY 0.2-1.8 MeV L. I. PIVOVAR, Yu. Z. LEVCHENKO, and A. N. GRIGOR'EV Physico-technical Institute, Ukrainian
FORMATION OF HIGHLY EXCITED HYDROGEN ATOMS BY PROTON CHARGE EXCHANGE IN GASES
 SOVET PHYSCS JETP VOLUME 20, NUMBER 4 APRL, 1965 FORMATON OF HGHLY EXCTED HYDROGEN ATOMS BY PROTON CHARGE EXCHANGE N GASES R. N. L'N, B.. KKAN, V. A. OPARN, E. S. SOLOV'EV, and N. V. FEDORENKO A. F. offe
SOVET PHYSCS JETP VOLUME 20, NUMBER 4 APRL, 1965 FORMATON OF HGHLY EXCTED HYDROGEN ATOMS BY PROTON CHARGE EXCHANGE N GASES R. N. L'N, B.. KKAN, V. A. OPARN, E. S. SOLOV'EV, and N. V. FEDORENKO A. F. offe
EXPERIMENTAL STUDY OF ELECTRON LOSS BY MULTIPLY CHARGED IONS IN GASES
 SOVET PHYSCS JETP VOLUME 1, NUMBER 1 JULY, 196 EXPERMENTAL STUDY OF ELECTRON LOSS BY MULTPLY CHARGED ONS N GASES. S. DMTREV, V. S. NKOLAEV, L. N. FATEEVA, and Ya. A. TEPLOVA nstitute of Nuclear Physics,
SOVET PHYSCS JETP VOLUME 1, NUMBER 1 JULY, 196 EXPERMENTAL STUDY OF ELECTRON LOSS BY MULTPLY CHARGED ONS N GASES. S. DMTREV, V. S. NKOLAEV, L. N. FATEEVA, and Ya. A. TEPLOVA nstitute of Nuclear Physics,
MANY-PHOTON IONIZATION OF THE XENON ATOM BY RUBY LASER RADIATION
 SOVIET PHYSICS JETP VOLUME 23, NUMBER 1 JULY, 1966 MANY-PHOTON IONIZATION OF THE XENON ATOM BY RUBY LASER RADIATION G. S. VORONOV and N. B. DELONE P. N. Lebedev Physics Institute, Academy of Sciences,
SOVIET PHYSICS JETP VOLUME 23, NUMBER 1 JULY, 1966 MANY-PHOTON IONIZATION OF THE XENON ATOM BY RUBY LASER RADIATION G. S. VORONOV and N. B. DELONE P. N. Lebedev Physics Institute, Academy of Sciences,
CHARGE EXCHANGE OF PROTONS IN INERT GASES INVOLVING THE FORMATION OF FAST HYDROGEN ATOMS IN THE 2s AND 2p STATES
 SOVET PHYSCS JETP VOLUME 23, NUMBER 3 SEPTEMBER, 1966 CHARGE EXCHANGE OF PROTONS N NERT GASES NVOLVNG THE FORMATON OF FAST HYDROGEN ATOMS N THE 2s AND 2p STATES E. P. ANDREEV, V. A. ANKUDNOV, and S. V.
SOVET PHYSCS JETP VOLUME 23, NUMBER 3 SEPTEMBER, 1966 CHARGE EXCHANGE OF PROTONS N NERT GASES NVOLVNG THE FORMATON OF FAST HYDROGEN ATOMS N THE 2s AND 2p STATES E. P. ANDREEV, V. A. ANKUDNOV, and S. V.
DOUBLE MOTT SCATTERING OF ELECTRONS SOVIET PHYSICS JETP VOLUME 17, NUMBER 4 OCTOBER, 1963
 SOVET PHYSCS JETP VOLUME 17, NUMBER 4 OCTOBER, 1963 DOUBLE MOTT SCATTERNG OF ELECTRONS L.A. MKAELYAN, A. A. BOROVOi, and E.. DENSOV nstitute of Atomic Energy, Academy of Sciences, U.S.S.R. Submitted to
SOVET PHYSCS JETP VOLUME 17, NUMBER 4 OCTOBER, 1963 DOUBLE MOTT SCATTERNG OF ELECTRONS L.A. MKAELYAN, A. A. BOROVOi, and E.. DENSOV nstitute of Atomic Energy, Academy of Sciences, U.S.S.R. Submitted to
THE MODERN VIEW OF ATOMIC STRUCTURE
 44 CHAPTER 2 Atoms, Molecules, and Ions GO FIGURE What is the charge on the particles that form the beam? Experiment Interpretation Incoming a particles Beam of a particles Source of a particles Nucleus
44 CHAPTER 2 Atoms, Molecules, and Ions GO FIGURE What is the charge on the particles that form the beam? Experiment Interpretation Incoming a particles Beam of a particles Source of a particles Nucleus
DEPOSITION OF THIN TiO 2 FILMS BY DC MAGNETRON SPUTTERING METHOD
 Chapter 4 DEPOSITION OF THIN TiO 2 FILMS BY DC MAGNETRON SPUTTERING METHOD 4.1 INTRODUCTION Sputter deposition process is another old technique being used in modern semiconductor industries. Sputtering
Chapter 4 DEPOSITION OF THIN TiO 2 FILMS BY DC MAGNETRON SPUTTERING METHOD 4.1 INTRODUCTION Sputter deposition process is another old technique being used in modern semiconductor industries. Sputtering
4. The mass of a proton is approximately equal to the mass of A an alpha particle C a positron. B a beta particle D a neutron
 1. Which particles have approximately the same mass? A an electron and an alpha particle B an electron and a proton C a neutron and an alpha particle D a neutron and a proton 2. Which phrase describes
1. Which particles have approximately the same mass? A an electron and an alpha particle B an electron and a proton C a neutron and an alpha particle D a neutron and a proton 2. Which phrase describes
CLASS 12th. Modern Physics-II
 CLASS 12th Modern Physics-II 1. Thomson s Atom Model From the study of discharge of electricity through gases, it became clear that an atom consists of positive and negative charges. As the atom is electrically
CLASS 12th Modern Physics-II 1. Thomson s Atom Model From the study of discharge of electricity through gases, it became clear that an atom consists of positive and negative charges. As the atom is electrically
EFFECTIVE CROSS SECTIONS FOR THE EXCHANGE EXCITATION OF ATOMS AND IONS BY ELECTRON IMPACT
 SOVET PHYSCS JETP VOLUME 25, NUMBER 1 JULY, 1967 EFFECTVE CROSS SECTONS FOR THE EXCHANGE EXCTATON OF ATOMS AND ONS BY ELECTRON MPACT. L. BEGMAN and L. A. VANSHTEN P. N. Lebedev Physics nstitute, Academy
SOVET PHYSCS JETP VOLUME 25, NUMBER 1 JULY, 1967 EFFECTVE CROSS SECTONS FOR THE EXCHANGE EXCTATON OF ATOMS AND ONS BY ELECTRON MPACT. L. BEGMAN and L. A. VANSHTEN P. N. Lebedev Physics nstitute, Academy
ON THE DOMAIN STRUCTURE OF UNIAXIAL FERRO MAGNETS
 SOVET PHYSCS JETP VOLUME 19, NUMBER 5 NOVEMBER, 1964 ON THE DOMAN STRUCTURE OF UNAXAL FERRO MAGNETS JAN KACZER Physical nstitute, Czechoslovak Academy of Sciences, Prague Submitted to JETP editor October
SOVET PHYSCS JETP VOLUME 19, NUMBER 5 NOVEMBER, 1964 ON THE DOMAN STRUCTURE OF UNAXAL FERRO MAGNETS JAN KACZER Physical nstitute, Czechoslovak Academy of Sciences, Prague Submitted to JETP editor October
1. Introduction. Etim I.P. 1, William E. S. 2, Ekwe S.O. 3. Licensed Under Creative Commons Attribution CC BY
 Alpha-Particle Spectroscopy and Ranges in Air Etim I.P. 1, William E. S. 2, Ekwe S.O. 3 1 Lecturer in the Department Of Physics, University of Calabar, Calabar, Nigeria 2 Concluded his Master program in
Alpha-Particle Spectroscopy and Ranges in Air Etim I.P. 1, William E. S. 2, Ekwe S.O. 3 1 Lecturer in the Department Of Physics, University of Calabar, Calabar, Nigeria 2 Concluded his Master program in
EXPERIMENTAL SETUP AND PROCEDURE
 NEUTRALIZATION OF H - IONS ON THE PLASMA TARGET V.A. Baturin *, P.A. Litvinov Institute of Applied Physics, National Academy of Science of Ukraine, 58 Petropavlovskaya St. Sumy, 40030 Ukraine, E-mail:
NEUTRALIZATION OF H - IONS ON THE PLASMA TARGET V.A. Baturin *, P.A. Litvinov Institute of Applied Physics, National Academy of Science of Ukraine, 58 Petropavlovskaya St. Sumy, 40030 Ukraine, E-mail:
AE 3051, Lab #16. Investigation of the Ideal Gas State Equation. By: George P. Burdell. Group E3
 AE 3051, Lab #16 Investigation of the Ideal Gas State Equation By: George P. Burdell Group E3 Summer Semester 000 Abstract The validity of the ideal gas equation of state was experimentally tested for
AE 3051, Lab #16 Investigation of the Ideal Gas State Equation By: George P. Burdell Group E3 Summer Semester 000 Abstract The validity of the ideal gas equation of state was experimentally tested for
MEASUREMENT OF LIFETIMES OF EXCITED STATES OF THE HYDROGEN ATOM
 SOVIET PHYSICS JETP VOLUME 21, NUMBER 1 JULY, 1965 MEASUREMENT OF LIFETIMES OF EXCITED STATES OF THE HYDROGEN ATOM V. A. ANKUDINOV, S. V. BOBASHEV, and E. P. ANDREEV A. F. Ioffe Physico-technical Institute,
SOVIET PHYSICS JETP VOLUME 21, NUMBER 1 JULY, 1965 MEASUREMENT OF LIFETIMES OF EXCITED STATES OF THE HYDROGEN ATOM V. A. ANKUDINOV, S. V. BOBASHEV, and E. P. ANDREEV A. F. Ioffe Physico-technical Institute,
Ionization and excitation of Rydberg states in collisions of fast metastable helium atoms with He, Ne, and N2
 onization and excitation of Rydberg states in collisions of fast metastable helium atoms with He, Ne, and N2 Yu. A. Kudryavtsev and V. V. Petrunin nstitute of Spectroscopy, Academy of Sciences of the USSR,
onization and excitation of Rydberg states in collisions of fast metastable helium atoms with He, Ne, and N2 Yu. A. Kudryavtsev and V. V. Petrunin nstitute of Spectroscopy, Academy of Sciences of the USSR,
Quadrupole Mass Spectrometry Concepts. Mass spectrometers for residual gas analysis: Intermediate Level Users Guide
 Quadrupole Mass Spectrometry Concepts Mass spectrometers for residual gas analysis: Intermediate Level Users Guide What does Residual Gas Analysis allow us to do? RGA is the examination of the molecular
Quadrupole Mass Spectrometry Concepts Mass spectrometers for residual gas analysis: Intermediate Level Users Guide What does Residual Gas Analysis allow us to do? RGA is the examination of the molecular
Courtesy of ESS and TheRGA web pages part of a series of application and theory notes for public use which are provided free of charge by ESS.
 ESS The RGA freenotes Theory page 1 of 14 RGA Theory Notes Courtesy of ESS and TheRGA web pages part of a series of application and theory notes for public use which are provided free of charge by ESS.
ESS The RGA freenotes Theory page 1 of 14 RGA Theory Notes Courtesy of ESS and TheRGA web pages part of a series of application and theory notes for public use which are provided free of charge by ESS.
PHYSICS FORM 5 PHYSICS OF THE ATOM
 The Shell Model and The Periodic Table PHYSICS The Rutherford-Bohr model of the atom shed light on the arrangement of the periodic table of the elements. The orbits which electrons were allowed to occupy
The Shell Model and The Periodic Table PHYSICS The Rutherford-Bohr model of the atom shed light on the arrangement of the periodic table of the elements. The orbits which electrons were allowed to occupy
Low Energy Nuclear Fusion Reactions in Solids
 Kasagi, J., et al. Low Energy Nuclear Fusion Reactions in Solids. in 8th International Conference on Cold Fusion. 2000. Lerici (La Spezia), Italy: Italian Physical Society, Bologna, Italy. Low Energy Nuclear
Kasagi, J., et al. Low Energy Nuclear Fusion Reactions in Solids. in 8th International Conference on Cold Fusion. 2000. Lerici (La Spezia), Italy: Italian Physical Society, Bologna, Italy. Low Energy Nuclear
Activity # 2. Name. Date due. Assignment on Atomic Structure
 Activity # 2 10 Name Date Date due Assignment on Atomic Structure NOTE: This assignment is based on material on the Power Point called Atomic Structure, as well as pages 167-173 in the Science Probe textbook.
Activity # 2 10 Name Date Date due Assignment on Atomic Structure NOTE: This assignment is based on material on the Power Point called Atomic Structure, as well as pages 167-173 in the Science Probe textbook.
Ionic and Covalent Bonds
 Ionic and Covalent Bonds Section #2 Downloadable at: http://tekim.undip.ac.id/staf/istadi Compounds: Introduction to Bonding The noble gases - helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe),
Ionic and Covalent Bonds Section #2 Downloadable at: http://tekim.undip.ac.id/staf/istadi Compounds: Introduction to Bonding The noble gases - helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe),
= : K A
 Atoms and Nuclei. State two limitations of JJ Thomson s model of atom. 2. Write the SI unit for activity of a radioactive substance. 3. What observations led JJ Thomson to conclusion that all atoms have
Atoms and Nuclei. State two limitations of JJ Thomson s model of atom. 2. Write the SI unit for activity of a radioactive substance. 3. What observations led JJ Thomson to conclusion that all atoms have
Ionic and Covalent Bonds
 Ionic and Covalent Bonds Downloaded at http://www.istadi.net Section #2 1 2 1 Compounds: Introduction to Bonding The noble gases - helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon
Ionic and Covalent Bonds Downloaded at http://www.istadi.net Section #2 1 2 1 Compounds: Introduction to Bonding The noble gases - helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon
ICPMS Doherty Lecture 1
 ICPMS Doherty Lecture 1 Mass Spectrometry This material provides some background on how to measure isotope abundances by means of mass spectrometry. Mass spectrometers create and separate ionized atoms
ICPMS Doherty Lecture 1 Mass Spectrometry This material provides some background on how to measure isotope abundances by means of mass spectrometry. Mass spectrometers create and separate ionized atoms
Physics 30 Modern Physics Unit: Atomic Basics
 Physics 30 Modern Physics Unit: Atomic Basics Models of the Atom The Greeks believed that if you kept dividing matter into smaller and smaller pieces, you would eventually come to a bit of matter that
Physics 30 Modern Physics Unit: Atomic Basics Models of the Atom The Greeks believed that if you kept dividing matter into smaller and smaller pieces, you would eventually come to a bit of matter that
The Franck-Hertz Experiment David Ward The College of Charleston Phys 370/Experimental Physics Spring 1997
 The Franck-Hertz Experiment David Ward The College of Charleston Phys 370/Experimental Physics Spring 1997 Abstract One of the most important experiments supporting quantum theory is the Franck- Hertz
The Franck-Hertz Experiment David Ward The College of Charleston Phys 370/Experimental Physics Spring 1997 Abstract One of the most important experiments supporting quantum theory is the Franck- Hertz
FISSION OF SILVER BY HIGH-ENERGY PROTONS
 ELASTC SCATTERNG OF 3 -Mev PONS BY DEUTERONS 21 n conclusion, the authors wish to thank Prof.. M. Frank and. a. Barit for help in this work, G. M. Vagradov for useful discussion, and also the group of
ELASTC SCATTERNG OF 3 -Mev PONS BY DEUTERONS 21 n conclusion, the authors wish to thank Prof.. M. Frank and. a. Barit for help in this work, G. M. Vagradov for useful discussion, and also the group of
Teacher: Mr. gerraputa. Name: Base your answer to the question on the information below. Given the electron dot diagram:
 Teacher: Mr. gerraputa Print Close Name: 1. Given the electron dot diagram: The valence electrons represented by the electron dot diagram could be those of atoms in Group 1. 13 3. 3 2. 15 4. 16 2. Which
Teacher: Mr. gerraputa Print Close Name: 1. Given the electron dot diagram: The valence electrons represented by the electron dot diagram could be those of atoms in Group 1. 13 3. 3 2. 15 4. 16 2. Which
INTEGRAL AND SPECTRAL CHARACTERISTICS OF ATON STATIONARY PLASMA THRUSTER OPERATING ON KRYPTON AND XENON
 1 INTEGRAL AND SPECTRAL CHARACTERISTICS OF ATON STATIONARY PLASMA THRUSTER OPERATING ON KRYPTON AND XENON A.I.Bugrova, A.I.Morozov *, A.S.Lipatov, A.M.Bishaev, V.K.Kharchevnikov, M.V.Kozintseva. Moscow
1 INTEGRAL AND SPECTRAL CHARACTERISTICS OF ATON STATIONARY PLASMA THRUSTER OPERATING ON KRYPTON AND XENON A.I.Bugrova, A.I.Morozov *, A.S.Lipatov, A.M.Bishaev, V.K.Kharchevnikov, M.V.Kozintseva. Moscow
EXPERIMENTAL INVESTIGATION OF THE EXCITATION OF Ar II AND Kr II IN ELECTRON-ION COLLISIONS
 SOVET PHYSCS JETP VOLUME 36, NUMBER 6 JUNE, 1973 EXPERMENTAL NVESTGATON OF THE EXCTATON OF Ar AND Kr N ELECTRON-ON COLLSONS. P. ZAPESOCHNYl, A.. MRE, A.. DASHCHENKO, V. S. VUKSTCH, F. F. DANCH and V. A.
SOVET PHYSCS JETP VOLUME 36, NUMBER 6 JUNE, 1973 EXPERMENTAL NVESTGATON OF THE EXCTATON OF Ar AND Kr N ELECTRON-ON COLLSONS. P. ZAPESOCHNYl, A.. MRE, A.. DASHCHENKO, V. S. VUKSTCH, F. F. DANCH and V. A.
INTRODUCTION Strained Silicon Monochromator Magnesium Housing Windows for Monochromator Shutter and Collimator Fission Detector HOPG Monochromator
 Design for a Four-Blade Neutron Interferometer INTRODUCTION Strained Silicon Monochromator The neutron beam used for this interferometer is separated from the NIST reactor's main beam using a strained
Design for a Four-Blade Neutron Interferometer INTRODUCTION Strained Silicon Monochromator The neutron beam used for this interferometer is separated from the NIST reactor's main beam using a strained
Auger decay of excited Ar projectiles emerging from carbon foils
 J. Phys. B: Atom. Molec. Phys., Vol. 9, No. 15, 1976. Printed in Great Britain. @ 1976 LETTER TO THE EDITOR Auger decay of excited Ar projectiles emerging from carbon foils R A Baragiola?, P Ziem and N
J. Phys. B: Atom. Molec. Phys., Vol. 9, No. 15, 1976. Printed in Great Britain. @ 1976 LETTER TO THE EDITOR Auger decay of excited Ar projectiles emerging from carbon foils R A Baragiola?, P Ziem and N
3 - Atomic Absorption Spectroscopy
 3 - Atomic Absorption Spectroscopy Introduction Atomic-absorption (AA) spectroscopy uses the absorption of light to measure the concentration of gas-phase atoms. Since samples are usually liquids or solids,
3 - Atomic Absorption Spectroscopy Introduction Atomic-absorption (AA) spectroscopy uses the absorption of light to measure the concentration of gas-phase atoms. Since samples are usually liquids or solids,
Energy loss of alpha particles - Prelab questions
 Energy loss of alpha particles - Prelab questions 1. Write down the decay path from 226 Ra to 206 Pb. Show the intermediate nuclides and the nuclear reactions which cause each transformation (α/β ± decay).
Energy loss of alpha particles - Prelab questions 1. Write down the decay path from 226 Ra to 206 Pb. Show the intermediate nuclides and the nuclear reactions which cause each transformation (α/β ± decay).
PHITS calculation of the radiation field in HIMAC BIO
 PHITS calculation of the radiation field in HIMAC BIO Ondřej Ploc, Yukio Uchihori, Hisashi Kitamura, Lembit Sihver National Institute of Radiological Sciences, Chiba, Japan Nuclear Physics Institute, Prague,
PHITS calculation of the radiation field in HIMAC BIO Ondřej Ploc, Yukio Uchihori, Hisashi Kitamura, Lembit Sihver National Institute of Radiological Sciences, Chiba, Japan Nuclear Physics Institute, Prague,
Multicusp Sources for Ion Beam Lithography Applications
 LBL-3 6645 UC-406 Multicusp Sources for Ion Beam Lithography Applications K.N. Leung, P. H e n, W.B. Kunkel, Y. Lee, L. Perkins, D. Pickard, M. Sarstedt, M. Weber, and M.D. Williams Accelerator and Fusion
LBL-3 6645 UC-406 Multicusp Sources for Ion Beam Lithography Applications K.N. Leung, P. H e n, W.B. Kunkel, Y. Lee, L. Perkins, D. Pickard, M. Sarstedt, M. Weber, and M.D. Williams Accelerator and Fusion
- A spark is passed through the Argon in the presence of the RF field of the coil to initiate the plasma
 THE PLASMA Inductively Coupled Plasma Mass Spectrometry (ICP-MS) What is a Plasma? - The magnetic field created by a RF (radio frequency) coil produces a current within a stream of Argon (Ar) gas, which
THE PLASMA Inductively Coupled Plasma Mass Spectrometry (ICP-MS) What is a Plasma? - The magnetic field created by a RF (radio frequency) coil produces a current within a stream of Argon (Ar) gas, which
Ion Implanter Cyclotron Apparatus System
 Ion Implanter Cyclotron Apparatus System A. Latuszyñski, K. Pyszniak, A. DroŸdziel, D. M¹czka Institute of Physics, Maria Curie-Sk³odowska University, Lublin, Poland Abstract In this paper the authors
Ion Implanter Cyclotron Apparatus System A. Latuszyñski, K. Pyszniak, A. DroŸdziel, D. M¹czka Institute of Physics, Maria Curie-Sk³odowska University, Lublin, Poland Abstract In this paper the authors
Lawrence Berkeley National Laboratory Recent Work
 Lawrence Berkeley National Laboratory Recent Work Title CHARGE DISTRIBUTIONS OF OXYGEN AND NEON IONS PASSING THROUGH GASES Permalink https://escholarship.org/uc/item/7rh3w981 Authors Hubbard, Edward L.
Lawrence Berkeley National Laboratory Recent Work Title CHARGE DISTRIBUTIONS OF OXYGEN AND NEON IONS PASSING THROUGH GASES Permalink https://escholarship.org/uc/item/7rh3w981 Authors Hubbard, Edward L.
Authors, Title, Journal Citation and Comments. Reynolds, H. K. Dunbar, D. N. F. Wenzel, W. A. Whaling, W.
 Year Authors, Title, Journal and Comments 1944-Gray 1578 Gray, L. H. 1944 'The Ionization Method of Measuring Neutron Energy' Proc. Comb. Phil. Soc., 40, 72-102 (1944) Comment : S. H, He (.25-8 MeV) ->
Year Authors, Title, Journal and Comments 1944-Gray 1578 Gray, L. H. 1944 'The Ionization Method of Measuring Neutron Energy' Proc. Comb. Phil. Soc., 40, 72-102 (1944) Comment : S. H, He (.25-8 MeV) ->
Stopping power for MeV 12 C ions in solids
 Nuclear Instruments and Methods in Physics Research B 35 (998) 69±74 Stopping power for MeV C ions in solids Zheng Tao, Lu Xiting *, Zhai Yongjun, Xia Zonghuang, Shen Dingyu, Wang Xuemei, Zhao Qiang Department
Nuclear Instruments and Methods in Physics Research B 35 (998) 69±74 Stopping power for MeV C ions in solids Zheng Tao, Lu Xiting *, Zhai Yongjun, Xia Zonghuang, Shen Dingyu, Wang Xuemei, Zhao Qiang Department
LIMITATIONS OF RUTHERFORD S ATOMIC MODEL
 ELECTRONS IN ATOMS LIMITATIONS OF RUTHERFORD S ATOMIC MODEL Did not explain the chemical properties of atoms For example, it could not explain why metals or compounds of metals give off characteristic
ELECTRONS IN ATOMS LIMITATIONS OF RUTHERFORD S ATOMIC MODEL Did not explain the chemical properties of atoms For example, it could not explain why metals or compounds of metals give off characteristic
(Total for Question = 5 marks) PhysicsAndMathsTutor.com
 1 Rutherford designed an experiment to see what happened when alpha particles were directed at a piece of gold foil. Summarise the observations and state the conclusions Rutherford reached about the structure
1 Rutherford designed an experiment to see what happened when alpha particles were directed at a piece of gold foil. Summarise the observations and state the conclusions Rutherford reached about the structure
MS482 Materials Characterization ( 재료분석 ) Lecture Note 5: RBS
 2016 Fall Semester MS482 Materials Characterization ( 재료분석 ) Lecture Note 5: RBS Byungha Shin Dept. of MSE, KAIST 1 Course Information Syllabus 1. Overview of various characterization techniques (1 lecture)
2016 Fall Semester MS482 Materials Characterization ( 재료분석 ) Lecture Note 5: RBS Byungha Shin Dept. of MSE, KAIST 1 Course Information Syllabus 1. Overview of various characterization techniques (1 lecture)
Particle Nature of Matter. Chapter 4
 Particle Nature of Matter Chapter 4 Modern physics When my grandfather was born, atoms were just an idea. That year, 1897, was marked by the discovery of the electron by J.J. Thomson. The nuclear model
Particle Nature of Matter Chapter 4 Modern physics When my grandfather was born, atoms were just an idea. That year, 1897, was marked by the discovery of the electron by J.J. Thomson. The nuclear model
INTERMOLECULAR FORCES
 INTERMOLECULAR FORCES Their Origin and Determination By GEOFFREY C. MAITLAND Senior Research Scientist Schlumberger Cambridge Research, Cambridge MAURICE RIGBY Lecturer in the Department of Chemistry King's
INTERMOLECULAR FORCES Their Origin and Determination By GEOFFREY C. MAITLAND Senior Research Scientist Schlumberger Cambridge Research, Cambridge MAURICE RIGBY Lecturer in the Department of Chemistry King's
ELEMENT2 High Resolution- ICP-MS INSTRUMENT OVERVIEW
 ELEMENT2 High Resolution- ICP-MS INSTRUMENT OVERVIEW Inductively Coupled Plasma Mass Spectrometry (ICP-MS) What is a Plasma? - The magnetic field created by a RF (radio frequency) coil produces
ELEMENT2 High Resolution- ICP-MS INSTRUMENT OVERVIEW Inductively Coupled Plasma Mass Spectrometry (ICP-MS) What is a Plasma? - The magnetic field created by a RF (radio frequency) coil produces
Radioactivity. General Physics II PHYS 111. King Saud University College of Applied Studies and Community Service Department of Natural Sciences
 King Saud University College of Applied Studies and Community Service Department of Natural Sciences Radioactivity General Physics II PHYS 111 Nouf Alkathran nalkathran@ksu.edu.sa Outline Radioactive Decay
King Saud University College of Applied Studies and Community Service Department of Natural Sciences Radioactivity General Physics II PHYS 111 Nouf Alkathran nalkathran@ksu.edu.sa Outline Radioactive Decay
Atoms and Periodic Table Unit Notes
 1 Atoms and Periodic Table Unit Notes Name: (DO NOT LOSE!) Rutherford s gold foil experiment An Atom is the smallest part of an element which can take part in a chemical reaction 2 The atom consists of
1 Atoms and Periodic Table Unit Notes Name: (DO NOT LOSE!) Rutherford s gold foil experiment An Atom is the smallest part of an element which can take part in a chemical reaction 2 The atom consists of
R. Clark, D. Humbert, K. Sheikh Nuclear Data Section
 Calculation of Atomic Data for Plasma Modeling: Introduction and Atomic Structure Part 1 R. Clark, D. Humbert, K. Sheikh Nuclear Data Section Overview Plasmas in fusion research Data needs for plasma modeling
Calculation of Atomic Data for Plasma Modeling: Introduction and Atomic Structure Part 1 R. Clark, D. Humbert, K. Sheikh Nuclear Data Section Overview Plasmas in fusion research Data needs for plasma modeling
Name Date Due Test Day! Unit 1: Atomic Theory. Pretest Practice K +
 ! 2017-2018 Name Date Due Test Day! Unit 1: Atomic Theory Pretest Practice 1.) Complete the following table: Work space (if needed)! 2.) What do the superscripts and subscript represent in the symbol below?
! 2017-2018 Name Date Due Test Day! Unit 1: Atomic Theory Pretest Practice 1.) Complete the following table: Work space (if needed)! 2.) What do the superscripts and subscript represent in the symbol below?
atomos is a Greek word which means indivisible
 The History of Atomic Theory i.e. the history of the development of thought about what an atom is. 1st timeframe: around 5 B.C. : This was the time of the Ancient Greeks (in Athens, Greece). During the
The History of Atomic Theory i.e. the history of the development of thought about what an atom is. 1st timeframe: around 5 B.C. : This was the time of the Ancient Greeks (in Athens, Greece). During the
Introduction to Elemental Analysis
 Introduction to Elemental Analysis A dust storm charges across the Sahara desert, whipping up sand and blowing it west, towards the ocean. Meanwhile, in the middle of the Atlantic Ocean, a team of scientist
Introduction to Elemental Analysis A dust storm charges across the Sahara desert, whipping up sand and blowing it west, towards the ocean. Meanwhile, in the middle of the Atlantic Ocean, a team of scientist
v = E B FXA 2008 UNIT G485 Module Magnetic Fields BQv = EQ THE MASS SPECTROMETER
 UNIT G485 Module 1 5.1.2 Magnetic Fields 11 Thus, in order for the particle to suffer NO DEFLECTION and so exit the device at Y : From which : MAGNETIC FORCE UP = ELECTRIC FORCE DOWN BQv = EQ THE MASS
UNIT G485 Module 1 5.1.2 Magnetic Fields 11 Thus, in order for the particle to suffer NO DEFLECTION and so exit the device at Y : From which : MAGNETIC FORCE UP = ELECTRIC FORCE DOWN BQv = EQ THE MASS
Lecture 22 Ion Beam Techniques
 Lecture 22 Ion Beam Techniques Schroder: Chapter 11.3 1/44 Announcements Homework 6/6: Will be online on later today. Due Wednesday June 6th at 10:00am. I will return it at the final exam (14 th June).
Lecture 22 Ion Beam Techniques Schroder: Chapter 11.3 1/44 Announcements Homework 6/6: Will be online on later today. Due Wednesday June 6th at 10:00am. I will return it at the final exam (14 th June).
Differential Cross Section Measurements in Ion-molecule Collisions
 Differential Cross Section Measurements in Ion-molecule Collisions Song Cheng Department of Physics and Astronomy, University of Toledo, Toledo, Ohio 43606 A 14 m long beam line dedicated to study very
Differential Cross Section Measurements in Ion-molecule Collisions Song Cheng Department of Physics and Astronomy, University of Toledo, Toledo, Ohio 43606 A 14 m long beam line dedicated to study very
Assessment of the Azimuthal Homogeneity of the Neutral Gas in a Hall Effect Thruster using Electron Beam Fluorescence
 Assessment of the Azimuthal Homogeneity of the Neutral Gas in a Hall Effect Thruster using Electron Beam Fluorescence IEPC-2015-91059 / ISTS-2015-b-91059 Presented at Joint Conference of 30th International
Assessment of the Azimuthal Homogeneity of the Neutral Gas in a Hall Effect Thruster using Electron Beam Fluorescence IEPC-2015-91059 / ISTS-2015-b-91059 Presented at Joint Conference of 30th International
QUANTUM MECHANICAL THEORY OF THE EQUATION OF STATE FOR POTASSIUM, ALUMINUM, AND IRON
 SOVIET PHYSICS JETP VOLUME 24, NUMBER 1 JANUARY, 1967 QUANTUM MECHANICAL THEORY OF THE EQUATION OF STATE FOR POTASSIUM, ALUMINUM, AND IRON G. M. GANDEL'MAN Submitted to JETP editor February 28, 1966 J.
SOVIET PHYSICS JETP VOLUME 24, NUMBER 1 JANUARY, 1967 QUANTUM MECHANICAL THEORY OF THE EQUATION OF STATE FOR POTASSIUM, ALUMINUM, AND IRON G. M. GANDEL'MAN Submitted to JETP editor February 28, 1966 J.
Developing the Periodic Table
 Developing the Periodic Table Early Element Classification Mendeleev s First Periodic Table Mendeleev s First Periodic Table Mendeleev s Periodic Table Arranged by increasing atomic mass Some elements
Developing the Periodic Table Early Element Classification Mendeleev s First Periodic Table Mendeleev s First Periodic Table Mendeleev s Periodic Table Arranged by increasing atomic mass Some elements
DESIGN OF A WIEN FILTER AND MEASUREMENT OF LONGITUDINAL POLARIZATION OF BETA PARTICLES BY S. S. ABHYANKAR AND M. R. BHIDAY
 DESIGN OF A WIEN FILTER AND MEASUREMENT OF LONGITUDINAL POLARIZATION OF BETA PARTICLES BY S. S. ABHYANKAR AND M. R. BHIDAY (Department of Physics, University of Poona, Poona-7, India) Received January
DESIGN OF A WIEN FILTER AND MEASUREMENT OF LONGITUDINAL POLARIZATION OF BETA PARTICLES BY S. S. ABHYANKAR AND M. R. BHIDAY (Department of Physics, University of Poona, Poona-7, India) Received January
EXAMINATION QUESTIONS (6)
 1. What is a beta-particle? A a helium nucleus B a high-energy electron C four protons D two neutrons EXAMINATION QUESTIONS (6) 2. The diagram shows part of a circuit used to switch street lamps on and
1. What is a beta-particle? A a helium nucleus B a high-energy electron C four protons D two neutrons EXAMINATION QUESTIONS (6) 2. The diagram shows part of a circuit used to switch street lamps on and
THE BIG IDEA: ELECTRONS AND THE STRUCTURE OF ATOMS. BONDING AND INTERACTIONS.
 HONORS CHEMISTRY - CHAPTER 9 CHEMICAL NAMES AND FORMULAS OBJECTIVES AND NOTES - V18 NAME: DATE: PAGE: THE BIG IDEA: ELECTRONS AND THE STRUCTURE OF ATOMS. BONDING AND INTERACTIONS. Essential Questions 1.
HONORS CHEMISTRY - CHAPTER 9 CHEMICAL NAMES AND FORMULAS OBJECTIVES AND NOTES - V18 NAME: DATE: PAGE: THE BIG IDEA: ELECTRONS AND THE STRUCTURE OF ATOMS. BONDING AND INTERACTIONS. Essential Questions 1.
X-RAY SCATTERING AND MOSELEY S LAW. OBJECTIVE: To investigate Moseley s law using X-ray absorption and to observe X- ray scattering.
 X-RAY SCATTERING AND MOSELEY S LAW OBJECTIVE: To investigate Moseley s law using X-ray absorption and to observe X- ray scattering. READING: Krane, Section 8.5. BACKGROUND: In 1913, Henry Moseley measured
X-RAY SCATTERING AND MOSELEY S LAW OBJECTIVE: To investigate Moseley s law using X-ray absorption and to observe X- ray scattering. READING: Krane, Section 8.5. BACKGROUND: In 1913, Henry Moseley measured
ELECTROMAGNETIC WAVES
 VISUAL PHYSICS ONLINE MODULE 7 NATURE OF LIGHT ELECTROMAGNETIC WAVES SPECTRA PRODUCED BY DISCHARGE TUBES CATHODE RAYS (electron beams) Streams of electrons (negatively charged particles) observed in vacuum
VISUAL PHYSICS ONLINE MODULE 7 NATURE OF LIGHT ELECTROMAGNETIC WAVES SPECTRA PRODUCED BY DISCHARGE TUBES CATHODE RAYS (electron beams) Streams of electrons (negatively charged particles) observed in vacuum
Development of a High-Speed VUV Camera System for 2-Dimensional Imaging of Edge Turbulent Structure in the LHD
 Development of a High-Speed VUV Camera System for 2-Dimensional Imaging of Edge Turbulent Structure in the LHD Masaki TAKEUCHI, Satoshi OHDACHI and LHD experimental group National Institute for Fusion
Development of a High-Speed VUV Camera System for 2-Dimensional Imaging of Edge Turbulent Structure in the LHD Masaki TAKEUCHI, Satoshi OHDACHI and LHD experimental group National Institute for Fusion
Nuclear Reactions A Z. Radioactivity, Spontaneous Decay: Nuclear Reaction, Induced Process: x + X Y + y + Q Q > 0. Exothermic Endothermic
 Radioactivity, Spontaneous Decay: Nuclear Reactions A Z 4 P D+ He + Q A 4 Z 2 Q > 0 Nuclear Reaction, Induced Process: x + X Y + y + Q Q = ( m + m m m ) c 2 x X Y y Q > 0 Q < 0 Exothermic Endothermic 2
Radioactivity, Spontaneous Decay: Nuclear Reactions A Z 4 P D+ He + Q A 4 Z 2 Q > 0 Nuclear Reaction, Induced Process: x + X Y + y + Q Q = ( m + m m m ) c 2 x X Y y Q > 0 Q < 0 Exothermic Endothermic 2
i ~ ~ f- i : I DIFFUSION OF METASTABLE HELIUM ATOMS IN HELIUM GAS AT LOW TEMPERATURES ~... SOVIET PHYSICS JETP VOLUME 26, NUMBER 3 MARCH, 1968
 SOVET PHYSCS JETP VOLUME 26, NUMBER 3 MARCH, 1968 DFFUSON OF METASTABLE HELUM ATOMS N HELUM GAS AT LOW TEMPERATURES. Ya. FUGOL' and P. L. PAKHOMOV Physico-technical nstitute of Low Temperatures, Academy
SOVET PHYSCS JETP VOLUME 26, NUMBER 3 MARCH, 1968 DFFUSON OF METASTABLE HELUM ATOMS N HELUM GAS AT LOW TEMPERATURES. Ya. FUGOL' and P. L. PAKHOMOV Physico-technical nstitute of Low Temperatures, Academy
International Atomic Energy Agency, Vienna, Austria. Charge Transfer in Collisions of Ions with atoms and molecules.
 International Centre for Theoretical Physics (ICTP), Trieste, Italy International Atomic Energy Agency, Vienna, Austria Training Workshop on Atomic and Molecular Data for Fusion Energy Research Charge
International Centre for Theoretical Physics (ICTP), Trieste, Italy International Atomic Energy Agency, Vienna, Austria Training Workshop on Atomic and Molecular Data for Fusion Energy Research Charge
Time to develop a model
 ATOMIC THEORY ONCE UPON A TIME People have been fascinated with matter for a long time. What is matter? What is all this stuff around us made of? Can it be broken down? Are there different types of matter?
ATOMIC THEORY ONCE UPON A TIME People have been fascinated with matter for a long time. What is matter? What is all this stuff around us made of? Can it be broken down? Are there different types of matter?
ION Pumps for UHV Systems, Synchrotrons & Particle Accelerators. Mauro Audi, Academic, Government & Research Marketing Manager
 ION Pumps for UHV Systems, Synchrotrons & Particle Accelerators Mauro Audi, Academic, Government & Research Marketing Manager ION Pumps Agilent Technologies 1957-59 Varian Associates invents the first
ION Pumps for UHV Systems, Synchrotrons & Particle Accelerators Mauro Audi, Academic, Government & Research Marketing Manager ION Pumps Agilent Technologies 1957-59 Varian Associates invents the first
(ii) Compare the movement and arrangement of the molecules in solid nitrogen to those in nitrogen gas.
 1 Kinetic theory explains the properties of matter in terms of the arrangement and movement of particles. (a) Nitrogen is a gas at room temperature. Nitrogen molecules, N 2, are spread far apart and move
1 Kinetic theory explains the properties of matter in terms of the arrangement and movement of particles. (a) Nitrogen is a gas at room temperature. Nitrogen molecules, N 2, are spread far apart and move
Chapter 13: Partial Pressure Analysis
 Chapter 13: Partial Pressure Analysis Previously several types of sub-atmospheric pressure gauge were described. All of these gauges share one common feature: they report the total gas pressure. Partial
Chapter 13: Partial Pressure Analysis Previously several types of sub-atmospheric pressure gauge were described. All of these gauges share one common feature: they report the total gas pressure. Partial
Single Electron Capture in Slow Collisions of Doubly Charged Ions with Dinuclear Molecules
 Int. J. Mol. Sci. 2002, 3, 209-219 International Journal of Molecular Sciences ISSN 1422-0067 2002 by MDPI www.mdpi.org/ijms/ Single Electron Capture in Slow Collisions of Doubly Charged Ions with Dinuclear
Int. J. Mol. Sci. 2002, 3, 209-219 International Journal of Molecular Sciences ISSN 1422-0067 2002 by MDPI www.mdpi.org/ijms/ Single Electron Capture in Slow Collisions of Doubly Charged Ions with Dinuclear
Unsteady MHD Couette Flow with Heat Transfer in the Presence of Uniform Suction and Injection
 Mechanics and Mechanical Engineering Vol. 12, No. 2 (2008) 165 176 c Technical University of Lodz Unsteady MHD Couette Flow with Heat Transfer in the Presence of Uniform Suction and Injection Hazem A.
Mechanics and Mechanical Engineering Vol. 12, No. 2 (2008) 165 176 c Technical University of Lodz Unsteady MHD Couette Flow with Heat Transfer in the Presence of Uniform Suction and Injection Hazem A.
Transverse momentum of ionized atoms and diatomic molecules acquired in collisions with fast highly-charged heavy ion. V. Horvat and R. L.
 Transverse momentum of ionized atoms and diatomic molecules acquired in collisions with fast highly-charged heavy ion V. Horvat and R. L. Watson The momenta of ions and electrons emerging from collisions
Transverse momentum of ionized atoms and diatomic molecules acquired in collisions with fast highly-charged heavy ion V. Horvat and R. L. Watson The momenta of ions and electrons emerging from collisions
Chem Homework Set Answers
 Chem 310 th 4 Homework Set Answers 1. Cyclohexanone has a strong infrared absorption peak at a wavelength of 5.86 µm. (a) Convert the wavelength to wavenumber.!6!1 8* = 1/8 = (1/5.86 µm)(1 µm/10 m)(1 m/100
Chem 310 th 4 Homework Set Answers 1. Cyclohexanone has a strong infrared absorption peak at a wavelength of 5.86 µm. (a) Convert the wavelength to wavenumber.!6!1 8* = 1/8 = (1/5.86 µm)(1 µm/10 m)(1 m/100
FXA Candidates should be able to :
 1 Candidates should be able to : INTRODUCTION Describe qualitatively the alpha-particle scattering experiment and the evidence this provides for the existence, charge and small size of the nucleus. Describe
1 Candidates should be able to : INTRODUCTION Describe qualitatively the alpha-particle scattering experiment and the evidence this provides for the existence, charge and small size of the nucleus. Describe
Association of H + with H 2 at Low Temperatures
 WDS'11 Proceedings of Contributed Papers, Part II, 175 179, 011. ISBN 978--7378-185-9 MATFYZPRESS Association of with at Low Temperatures I. Zymak, P. Jusko, S. Roučka, D. Mulin, R. Plašil, and J. Glosík
WDS'11 Proceedings of Contributed Papers, Part II, 175 179, 011. ISBN 978--7378-185-9 MATFYZPRESS Association of with at Low Temperatures I. Zymak, P. Jusko, S. Roučka, D. Mulin, R. Plašil, and J. Glosík
Chapter 4. Table of Contents. Section 1 The Development of a New Atomic Model. Section 2 The Quantum Model of the Atom
 Arrangement of Electrons in Atoms Table of Contents Section 1 The Development of a New Atomic Model Section 2 The Quantum Model of the Atom Section 3 Electron Configurations Section 1 The Development of
Arrangement of Electrons in Atoms Table of Contents Section 1 The Development of a New Atomic Model Section 2 The Quantum Model of the Atom Section 3 Electron Configurations Section 1 The Development of
THE NATURE OF THE ATOM. alpha particle source
 chapter THE NATURE OF THE ATOM www.tutor-homework.com (for tutoring, homework help, or help with online classes) Section 30.1 Rutherford Scattering and the Nuclear Atom 1. Which model of atomic structure
chapter THE NATURE OF THE ATOM www.tutor-homework.com (for tutoring, homework help, or help with online classes) Section 30.1 Rutherford Scattering and the Nuclear Atom 1. Which model of atomic structure
ATOMIC WORLD P.1. ejected photoelectrons. current amplifier. photomultiplier tube (PMT)
 ATOMIC WORLD P. HKAL PAPER I 0 8 The metal Caesium has a work function of.08 ev. Given: Planck constant h = 6.63 0 34 J s, charge of an electron e =.60 0 9 C (a) (i) Calculate the longest wavelength of
ATOMIC WORLD P. HKAL PAPER I 0 8 The metal Caesium has a work function of.08 ev. Given: Planck constant h = 6.63 0 34 J s, charge of an electron e =.60 0 9 C (a) (i) Calculate the longest wavelength of
Safety Co-ordinator : Patrick Walden, TRIUMF, ext : 7340
 Safety Report for experiment E1104 : Beamtime Schedule 113 16-31 July 2008 Study of halo effects in the Scattering of 11 Li with heavy targets at energies around the Coulomb Barrier Experiment Leaders
Safety Report for experiment E1104 : Beamtime Schedule 113 16-31 July 2008 Study of halo effects in the Scattering of 11 Li with heavy targets at energies around the Coulomb Barrier Experiment Leaders
Chemistry 311: Instrumentation Analysis Topic 2: Atomic Spectroscopy. Chemistry 311: Instrumentation Analysis Topic 2: Atomic Spectroscopy
 Topic 2b: X-ray Fluorescence Spectrometry Text: Chapter 12 Rouessac (1 week) 4.0 X-ray Fluorescence Download, read and understand EPA method 6010C ICP-OES Winter 2009 Page 1 Atomic X-ray Spectrometry Fundamental
Topic 2b: X-ray Fluorescence Spectrometry Text: Chapter 12 Rouessac (1 week) 4.0 X-ray Fluorescence Download, read and understand EPA method 6010C ICP-OES Winter 2009 Page 1 Atomic X-ray Spectrometry Fundamental
NUCLEAR AND PARTICLE PHYSICS (PH242) PARTICLE PHYSICS
 NUCLEAR AND PARTICLE PHYSICS (PH242) PARTICLE PHYSICS History of Elementary Particles THE CLASSICAL ERA (1897-1932) Elementary particle physics was born in 1897 with J.J. Thomson s discovery of the ELECTRONS
NUCLEAR AND PARTICLE PHYSICS (PH242) PARTICLE PHYSICS History of Elementary Particles THE CLASSICAL ERA (1897-1932) Elementary particle physics was born in 1897 with J.J. Thomson s discovery of the ELECTRONS
What is a theory? An organized system of accepted knowledge that applies in a variety of circumstances to explain a specific set of phenomena
 Atomic Structure What is a theory? An organized system of accepted knowledge that applies in a variety of circumstances to explain a specific set of phenomena Early Theories Democritus: 4 B.C.: atom He
Atomic Structure What is a theory? An organized system of accepted knowledge that applies in a variety of circumstances to explain a specific set of phenomena Early Theories Democritus: 4 B.C.: atom He
 Title Stopping Powers of Al, Ni, Cu, Ag, Alpha Particles Author(s) Ishiwari, Ryutaro; Shiomi, Naoko; K Fumiko Citation Bulletin of the Institute for Chemi University (1977), 55(1): 68-73 Issue Date 1977-03-31
Title Stopping Powers of Al, Ni, Cu, Ag, Alpha Particles Author(s) Ishiwari, Ryutaro; Shiomi, Naoko; K Fumiko Citation Bulletin of the Institute for Chemi University (1977), 55(1): 68-73 Issue Date 1977-03-31
Sergey O. Tverdokhlebov
 IEPC-93-232 2140 Study of Double-Stage Anode Layer Thruster Using Inert Gases Sergey O. Tverdokhlebov Central Research Institute of Machine Building Kaliningrad (Moscow Region), Russia Abstract Pt - total
IEPC-93-232 2140 Study of Double-Stage Anode Layer Thruster Using Inert Gases Sergey O. Tverdokhlebov Central Research Institute of Machine Building Kaliningrad (Moscow Region), Russia Abstract Pt - total
Regents review Atomic & periodic
 2011-2012 1. The diagram below represents the nucleus of an atom. What are the atomic number and mass number of this atom? A) The atomic number is 9 and the mass number is 19. B) The atomic number is 9
2011-2012 1. The diagram below represents the nucleus of an atom. What are the atomic number and mass number of this atom? A) The atomic number is 9 and the mass number is 19. B) The atomic number is 9
CHM 213 (INORGANIC CHEMISTRY): Applications of Standard Reduction Potentials. Compiled by. Dr. A.O. Oladebeye
 CHM 213 (INORGANIC CHEMISTRY): Applications of Standard Reduction Potentials Compiled by Dr. A.O. Oladebeye Department of Chemistry University of Medical Sciences, Ondo, Nigeria Electrochemical Cell Electrochemical
CHM 213 (INORGANIC CHEMISTRY): Applications of Standard Reduction Potentials Compiled by Dr. A.O. Oladebeye Department of Chemistry University of Medical Sciences, Ondo, Nigeria Electrochemical Cell Electrochemical
Alex M Imai, Y. Ohta and A. Itoh Department of Nuclear Engineering, Kyoto University
 Alex M Imai, Y. Ohta and A. Itoh Department of Nuclear Engineering, Kyoto University Joint IAEA-NFRI Technical Meeting on Data Evaluation for Atomic, Molecular and Plasma-Material Interaction Processes
Alex M Imai, Y. Ohta and A. Itoh Department of Nuclear Engineering, Kyoto University Joint IAEA-NFRI Technical Meeting on Data Evaluation for Atomic, Molecular and Plasma-Material Interaction Processes
Chapter 3: Electron Structure and the Periodic Law
 Chapter 3: Electron Structure and the Periodic Law PERIODIC LAW This is a statement about the behavior of the elements when they are arranged in a specific order. In its present form the statement is:
Chapter 3: Electron Structure and the Periodic Law PERIODIC LAW This is a statement about the behavior of the elements when they are arranged in a specific order. In its present form the statement is:
SECTION A Quantum Physics and Atom Models
 AP Physics Multiple Choice Practice Modern Physics SECTION A Quantum Physics and Atom Models 1. Light of a single frequency falls on a photoelectric material but no electrons are emitted. Electrons may
AP Physics Multiple Choice Practice Modern Physics SECTION A Quantum Physics and Atom Models 1. Light of a single frequency falls on a photoelectric material but no electrons are emitted. Electrons may
The effect of self-absorption in hollow cathode lamp on its temperature
 Plasma Science and Applications (ICPSA 2013) International Journal of Modern Physics: Conference Series Vol. 32 (2014) 1460349 (9 pages) The Author DOI: 10.1142/S2010194514603494 The effect of self-absorption
Plasma Science and Applications (ICPSA 2013) International Journal of Modern Physics: Conference Series Vol. 32 (2014) 1460349 (9 pages) The Author DOI: 10.1142/S2010194514603494 The effect of self-absorption
IMPORTANT NOTES ANIL TUTORIALS PREVIOUS YEARS QUESTIONS OTHER IMPORTANT QUESTIONS
 Structure of The Atom IMPORTANT NOTES 1. Experiments on static electricity have proved that seemingly electrically neutral matter consists of electrically charged particles, such that positive charges
Structure of The Atom IMPORTANT NOTES 1. Experiments on static electricity have proved that seemingly electrically neutral matter consists of electrically charged particles, such that positive charges
Principles of Chemistry: A Molecular Approach, 3e (Tro) Chapter 2 Atoms and Elements
 Principles of Chemistry: A Molecular Approach, 3e (Tro) Chapter 2 Atoms and Elements 1) Which of the following is an example of the law of multiple proportions? A) A sample of chlorine is found to contain
Principles of Chemistry: A Molecular Approach, 3e (Tro) Chapter 2 Atoms and Elements 1) Which of the following is an example of the law of multiple proportions? A) A sample of chlorine is found to contain
Mass Analyzers. Principles of the three most common types magnetic sector, quadrupole and time of flight - will be discussed herein.
 Mass Analyzers After the production of ions in ion sources, the next critical step in mass spectrometry is to separate these gas phase ions according to their mass-to-charge ratio (m/z). Ions are extracted
Mass Analyzers After the production of ions in ion sources, the next critical step in mass spectrometry is to separate these gas phase ions according to their mass-to-charge ratio (m/z). Ions are extracted
Introduction to the Q Trap LC/MS/MS System
 www.ietltd.com Proudly serving laboratories worldwide since 1979 CALL +1.847.913.0777 for Refurbished & Certified Lab Equipment ABI Q Trap LC/MS/MS Introduction to the Q Trap LC/MS/MS System The Q Trap
www.ietltd.com Proudly serving laboratories worldwide since 1979 CALL +1.847.913.0777 for Refurbished & Certified Lab Equipment ABI Q Trap LC/MS/MS Introduction to the Q Trap LC/MS/MS System The Q Trap
Production of HCI with an electron beam ion trap
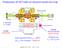 Production of HCI with an electron beam ion trap I=450 ma E= 5 kev axially: electrodes radially: electron beam space charge total trap potential U trap 200 V (U trap ion charge) 10000 ev 15000 A/cm 2 n
Production of HCI with an electron beam ion trap I=450 ma E= 5 kev axially: electrodes radially: electron beam space charge total trap potential U trap 200 V (U trap ion charge) 10000 ev 15000 A/cm 2 n
