Lesson 4. Temperature change
|
|
|
- Franklin Casey
- 5 years ago
- Views:
Transcription
1 Lesson 4 Temperature change T E A C H E R G U I D E Lesson summary Students meet scientist Jason Williams, an industrial chemist who designs the materials and processes for making solar cells. He explains that during the summers, Antarctic days are very long, sometimes lasting a couple of weeks or more. That makes solar energy an abundant natural resource near the South Pole. Solar cells convert energy from the sun into electrical energy. Converting energy from one form to another is an important process. In the activity, students will conduct two chemical reactions that convert chemical energy into thermal energy. Key concepts + A change in temperature is a clue that a chemical reaction may have occurred. + When the temperature increases during a chemical reaction, it is called an exothermic reaction. + When the temperature decreases during a chemical reaction, it is called an endothermic reaction. + It takes energy to break chemical bonds in the reactants. + Energy is released when chemical bonds form in the products. Safety Be sure you and the students wear properly fitting goggles. Read and follow all safety warnings on the labels of the sodium bicarbonate, citric acid, calcium chloride, and universal indicator containers. Also follow the warnings on the packaging of the foot warmer. Have students wash their hands after the activity. Proper disposal At the end of the lesson, have students pour their used solutions in a waste container. The resulting solution from the final demo should be placed in this container, too. Then dispose of all of this liquid waste down the drain or according to local regulations. The left-over sodium bicarbonate, citric acid, and calcium chloride powders can be disposed of with the classroom trash. The used foot warmer may be disposed of with the classroom trash. Wipe up spills with paper towels and dispose of them with the classroom trash. 1
2 Introduction 1. Introduce students to a scientist and his work. Distribute the student activity sheets for Lesson 4. In this lesson, students meet industrial chemist Jason Williams. He develops the materials that make up solar cells. He explains that there is an abundant supply of sunshine during the Antarctic summers. Because scientists work on the continent in the summer, there is also a higher demand for electricity. Solar panels convert energy from the sun into electrical energy. Students will conduct two chemical reactions that explore the conversion of chemical energy into thermal energy. Teacher demonstration 2. Do a demonstration to introduce students to the idea that the temperature can increase dramatically in some chemical reactions. Show students a hand warmer. Tell them these products work because of a chemical reaction that begins as soon as the package is opened. One of the hand warmer s main ingredients is very fine iron powder. Moisture and oxygen from the air react with the iron and release heat as part of a chemical reaction. This reaction is basically the same as rusting, but it happens much faster. You will need + 1 hand warmer Preparation instructions + Open the packaging for the hand warmer about fifteen minutes before you meet the students. If using scissors to open the package, be sure that you do not accidentally cut the hand warmer. Procedure 1. Pass the hand warmer around so that students can feel that it is warm. Expected results The hand warmer will begin to warm up slowly but will remain warm for a few hours. Tell students that there is always a change in temperature during chemical reactions. Sometimes they are dramatic, like with the hand warmer. Other times, the change is so slight that you might not notice it. 2
3 to predict + Do you think it is possible for the temperature of a chemical reaction to go down? Students may not be sure at this point, but the temperature can increase or decrease during a chemical reaction. Explain that if the temperature goes up, the reaction is called an exothermic reaction. More heat energy exits. If the temperature goes down, the reaction is called an endothermic reaction. More heat energy goes in to make the chemical reaction happen. + Is the chemical reaction between the iron filings and the oxygen and water in the air an endothermic or exothermic reaction? Exothermic. Student activity 3. Have students use a thermometer to observe the temperature changes in two different chemical reactions. Review with students how to read a thermometer. Let students know that they will measure both the starting temperature and either the highest or lowest temperature reached using the Celsius scale. Activity summary Information on how to conduct this activity is included on the student activity sheet. Students will monitor temperature changes during two different chemical reactions. The temperature decreases during one chemical reaction and increases in the other. Preparation instructions + Use the pre-made stickers to label one set of portion cups citric acid, calcium chloride, and sodium bicarbonate for each group. If you conducted lessons 1 and 3, reuse these labeled cups. + Place 1 teaspoon of citric acid, calcium chloride, and sodium bicarbonate in their labeled cups. + Use scissors to clip the bottom of the plastic backing off each thermometer so that the bottom of the plastic lines up with the bottom of the red bulb. This way, the amount of liquid used in the activity will cover the bulb of the thermometer completely. 3
4 Expected results + Chemical reaction between citric acid solution and sodium bicarbonate There will be bubbling, and the temperature will decrease. + Chemical reaction between sodium bicarbonate solution and calcium chloride There will be bubbling, and the temperature will increase. Have groups share their starting and final temperature for each reaction. Also have them classify each as either an endothermic or exothermic reaction. The temperature decreases in endothermic reactions and increases in exothermic reactions. Student activity 4. Have students conduct the chemistry challenge by adjusting reactants to get the temperature between 40 and 50 C. + What do you think will happen if you used more calcium chloride in the exothermic reaction? Increasing the amount of the calcium chloride will cause the temperature to increase more. Activity summary Students should refer to their activity sheet for guidance on how to conduct this activity. They will conduct the exothermic reaction again but this time with the goal of getting the highest temperature somewhere between 40 and 50 C. They will record the amount of calcium chloride used and the highest temperature reached. You may wish to limit either the time or the number of trials students conduct. Expected results Adding more calcium chloride will cause a greater increase in temperature. Class discussion 5. Discuss the big chemistry idea to help students understand what makes a chemical reaction either endothermic or exothermic. The big chemistry idea on the student activity sheet revisits the idea that in a chemical reaction atoms or groups of atoms in the reactants rearrange and re-bond to form the products. It also describes endothermic and exothermic reactions in terms of the energy used and released as bonds between atoms break and form. Tell students that when the temperature of a chemical reaction increases, the reaction is called an exothermic reaction. The first part of the word, exo, means out or out of and thermic has to do with heat or energy. So an exothermic reaction means that more energy goes out or is released by the reaction than goes into it. This leaves the reaction mixture at a higher temperature. 4
5 + A chemical reaction is endothermic when it takes more energy to break the bonds in the reactants than is released when the new bonds form to make the products. + A chemical reaction is exothermic when more energy is released when the new bonds form to make the products than is used to break the bonds in the reactants. + What do you know about the amount of energy required to break the bonds of the reactants compared to the amount of energy released when the products are formed? More energy was released when the bonds in the products formed than was required to break the bonds in the reactants. Teacher demonstration 6. Pour solutions from cup to cup and have students identify the clues of chemical change. In this demonstration, you will pour the entire contents of one cup into another that contains a small amount of citric acid, then sodium carbonate, then calcium chloride, and then citric acid again. As you pour the contents of one cup into the next, have students identify each of the clues of chemical change. Bring closure to all of the hands-on explorations students have conducted in this kit with this colorful demonstration. You will need + Goggles + Universal indicator + Citric acid + Sodium carbonate + Calcium chloride + Water + Thermometer + Small metric measuring cup Scoop + Small scoop + Small spoon + 5 clear plastic cups Spoon 5
6 Preparation instructions Line up 5 clear plastic cups. Place the following in each of 5 cups: + First cup 30 ml water and 30 drops of universal indicator + Second cup 1 scoop of citric acid + Third cup 1 spoon of sodium carbonate + Fourth cup 1 spoon of calcium chloride + Fifth cup 1 spoon of citric acid Procedure 1. Place the thermometer into the green indicator solution and make a note of the temperature. 2. Move the thermometer to the second cup and pour all of the green indicator solution into the cup. Swirl and make a note of the temperature. 3. Move the thermometer to the third cup and pour all of this solution into the next cup. Swirl and make a note of the temperature. 4. Move the thermometer to the fourth cup and pour all of this solution into the next cup. Swirl and make a note of the temperature. 5. Move the thermometer to the fifth cup and pour the entire contents of the cup into the next cup. Swirl and make a note of the temperature. Expected results + Cup 1 Solution is green. + Cup 2 Solution turns pink. + Cup 3 Solution turns purple. + Cup 4 Solution turns a chalky blue and the temperature increases. + Cup 5 Solution bubbles, turns orange, and becomes clear. + Which clues of chemical change did you observe? A color change, production of a gas, a slight change in temperature, and the formation of a precipitate. Application Have students read the real-world application on the student activity sheet. Explain that exothermic chemical reactions are used by military troops in remote locations so that they can have hot meals when they are in the field. Meals-Ready-to-Eat, or MREs, use a chemical reaction to warm up a pouch of food in about 10 minutes. 6
CO 2. Lesson 1. Production of a gas
 Lesson 1 Production of a gas T E A C H E R G U I D E CO 2 Lesson summary Students meet volcanologist Victor Helguson, who is studying the gases released by volcanoes in Iceland. Students conduct a chemical
Lesson 1 Production of a gas T E A C H E R G U I D E CO 2 Lesson summary Students meet volcanologist Victor Helguson, who is studying the gases released by volcanoes in Iceland. Students conduct a chemical
Lesson 2. Color change
 Lesson 2 Color change T E A C H E R G U I D E Lesson summary Students meet marine chemist Sera Tuikabe, who is studying ocean acidification in the water surrounding the Republic of the Fiji Islands. Students
Lesson 2 Color change T E A C H E R G U I D E Lesson summary Students meet marine chemist Sera Tuikabe, who is studying ocean acidification in the water surrounding the Republic of the Fiji Islands. Students
Chapter 6, Lesson 7: Energy Changes in Chemical Reactions
 Chapter 6, Lesson 7: Energy Changes in Chemical Reactions Key Concepts If two substances react and the temperature of the mixture decreases, the reaction is endothermic. If two substances react and the
Chapter 6, Lesson 7: Energy Changes in Chemical Reactions Key Concepts If two substances react and the temperature of the mixture decreases, the reaction is endothermic. If two substances react and the
Chapter 6, Lesson 9: Neutralizing Acids and Bases
 Chapter 6, Lesson 9: Neutralizing Acids and Bases Key Concepts ph is a measure of the concentration of H 3 O + ions in a solution. Adding an acid increases the concentration of H 3 O + ions in the solution.
Chapter 6, Lesson 9: Neutralizing Acids and Bases Key Concepts ph is a measure of the concentration of H 3 O + ions in a solution. Adding an acid increases the concentration of H 3 O + ions in the solution.
Chapter 6, Lesson 10: Carbon Dioxide Can Make a Solution Acidic
 Chapter 6, Lesson 10: Carbon Dioxide Can Make a Solution Acidic Key Concepts Carbon dioxide (CO 2 ) gas dissolved in water can cause water to become acidic. The acidity of water from dissolved CO 2 can
Chapter 6, Lesson 10: Carbon Dioxide Can Make a Solution Acidic Key Concepts Carbon dioxide (CO 2 ) gas dissolved in water can cause water to become acidic. The acidity of water from dissolved CO 2 can
Chemical Energy Conversions. Vanderbilt Student Volunteers for Science VINSE/VSVS Rural
 Chemical Energy Conversions Vanderbilt Student Volunteers for Science 2018-2019 VINSE/VSVS Rural Important!!! Please use this resource to reinforce your understanding of the lesson! Make sure you have
Chemical Energy Conversions Vanderbilt Student Volunteers for Science 2018-2019 VINSE/VSVS Rural Important!!! Please use this resource to reinforce your understanding of the lesson! Make sure you have
Activity Sheet Transferring thermal energy by dissolving salts
 Student Name: Date: Activity Sheet Transferring thermal energy by dissolving salts 1) Define Thermal energy and temperature in the boxes below. Thermal Energy Temperature Practice Experiment: Aim: To practice
Student Name: Date: Activity Sheet Transferring thermal energy by dissolving salts 1) Define Thermal energy and temperature in the boxes below. Thermal Energy Temperature Practice Experiment: Aim: To practice
Chapter 1, Lesson 3: The Ups and Downs of Thermometers
 Chapter 1, Lesson 3: The Ups and Downs of Thermometers Key Concepts The way a thermometer works is an example of heating and cooling a liquid. When heated, the molecules of the liquid in the thermometer
Chapter 1, Lesson 3: The Ups and Downs of Thermometers Key Concepts The way a thermometer works is an example of heating and cooling a liquid. When heated, the molecules of the liquid in the thermometer
Chapter 6, Lesson 1: What is a Chemical Reaction?
 Chapter 6, Lesson 1: What is a Chemical Reaction? Key Concepts: A physical change, such as a state change or dissolving, does not create a new substance, but a chemical change does. In a chemical reaction,
Chapter 6, Lesson 1: What is a Chemical Reaction? Key Concepts: A physical change, such as a state change or dissolving, does not create a new substance, but a chemical change does. In a chemical reaction,
2/22/2019 NEW UNIT! Chemical Interactions. Atomic Basics #19
 NEW UNIT! Chemical Interactions Atomic Basics #19 1 Vocabulary: Matter: Anything that has mass and takes up space. Atom: the smallest particle of matter. Element: A pure substance made up of only one type
NEW UNIT! Chemical Interactions Atomic Basics #19 1 Vocabulary: Matter: Anything that has mass and takes up space. Atom: the smallest particle of matter. Element: A pure substance made up of only one type
Reactions that Produce Heat
 Chemical Dominoes Activity 7 Reactions that Produce Heat GOALS In this activity you will: Explain the effect of particle size and use of a catalyst on reaction rate. Represent energy changes graphically.
Chemical Dominoes Activity 7 Reactions that Produce Heat GOALS In this activity you will: Explain the effect of particle size and use of a catalyst on reaction rate. Represent energy changes graphically.
Chemical Reaction Lab Bagged Chemical Reactions
 Learning Target: The student experiments and determines that the rates of reaction among atoms and molecules depend on the concentration, pressure, and temperature of the reactants and the presence or
Learning Target: The student experiments and determines that the rates of reaction among atoms and molecules depend on the concentration, pressure, and temperature of the reactants and the presence or
In your activity, you will use a chemical reaction and a filter to remove some very small dissolved particles.
 Lesson 3 Forming a precipitate S T U D E N T A C T I V I T Y S H E E T Name: Meet the scientist Country: Republic of Peru Scientist: Adriana Garcia Sera, from Fiji, told you about the problems of ocean
Lesson 3 Forming a precipitate S T U D E N T A C T I V I T Y S H E E T Name: Meet the scientist Country: Republic of Peru Scientist: Adriana Garcia Sera, from Fiji, told you about the problems of ocean
Section 1: What is a Chemical Reaction
 Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
MiSP CHEMICAL REACTIONS, L3 Teacher Guide. Introduction
 MiSP CHEMICAL REACTIONS, L3 Teacher Guide Introduction This weeklong unit should be included with other chemistry content teaching and learning. It is designed to follow Intermediate Level Science Core
MiSP CHEMICAL REACTIONS, L3 Teacher Guide Introduction This weeklong unit should be included with other chemistry content teaching and learning. It is designed to follow Intermediate Level Science Core
Chapter 5, Lesson 1: Water is a Polar Molecule
 Chapter 5, Lesson 1: Water is a Polar Molecule Key Concepts The water molecule, as a whole, has 10 protons and 10 electrons, so it is neutral. In a water molecule, the oxygen atom and hydrogen atoms share
Chapter 5, Lesson 1: Water is a Polar Molecule Key Concepts The water molecule, as a whole, has 10 protons and 10 electrons, so it is neutral. In a water molecule, the oxygen atom and hydrogen atoms share
Post-Show. Chemistry. Periodic Table of the Elements. After the Show. Traveling Science Shows
 Traveling Science Shows Post-Show Chemistry After the Show We recently presented a Chemistry show at your school, and thought you and your students might like to continue investigating this topic. The
Traveling Science Shows Post-Show Chemistry After the Show We recently presented a Chemistry show at your school, and thought you and your students might like to continue investigating this topic. The
Chemical Reactions. Teachers Guide.
 Chemical Reactions. Teachers Guide. Introduction: Almost everything you will ever see in this universe is made of atoms. Each different type of atom is known as a chemical element there are currently 118
Chemical Reactions. Teachers Guide. Introduction: Almost everything you will ever see in this universe is made of atoms. Each different type of atom is known as a chemical element there are currently 118
Physical & Chemical PROPERTIES
 Name Test Date Hour Group Chem #4 Notebook Properties & Changes LEARNING TARGETS I can give examples of physical and chemical properties. I can give examples of physical and chemical changes. I can identify
Name Test Date Hour Group Chem #4 Notebook Properties & Changes LEARNING TARGETS I can give examples of physical and chemical properties. I can give examples of physical and chemical changes. I can identify
COPYRIGHT FOUNTAINHEAD PRESS
 Calorimetry: Heats of Solution Objective: Use calorimetric measurements to determine heats of solution of two ionic compounds. Materials: Solid ammonium nitrate (NH 4 NO 3 ) and anhydrous calcium chloride
Calorimetry: Heats of Solution Objective: Use calorimetric measurements to determine heats of solution of two ionic compounds. Materials: Solid ammonium nitrate (NH 4 NO 3 ) and anhydrous calcium chloride
The Hand Warmer Design Challenge: Where Does the Heat Come From?
 The Hand Warmer Design Challenge: Where Does the Heat Come From? LSNED Learn Something New Every Day About Sharing and Contributions Interesting Facts Science In Your Mittens: The Chemistry Of Hand Warmers
The Hand Warmer Design Challenge: Where Does the Heat Come From? LSNED Learn Something New Every Day About Sharing and Contributions Interesting Facts Science In Your Mittens: The Chemistry Of Hand Warmers
Chapter 5, Lesson 2 Surface Tension
 Chapter 5, Lesson 2 Surface Tension Key Concepts The attraction of molecules at the surface of a liquid is called surface tension. The polarity of water molecules can help explain why water has a strong
Chapter 5, Lesson 2 Surface Tension Key Concepts The attraction of molecules at the surface of a liquid is called surface tension. The polarity of water molecules can help explain why water has a strong
What Do You Think? Investigate GOALS
 Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations about the combinations of materials
Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations about the combinations of materials
Today is: Monday, October 17th
 Today is: Monday, October 17th Get out your homework! 10/17/2016 #motivationmonday (This is Week 11 Warm Ups!) 1. It s a new quarter, which means a fresh start! What can you do to stay motivated this quarter?
Today is: Monday, October 17th Get out your homework! 10/17/2016 #motivationmonday (This is Week 11 Warm Ups!) 1. It s a new quarter, which means a fresh start! What can you do to stay motivated this quarter?
Student Notes. Chemical Reactions LINK
 LCPS Core Experience Chemical Reactions Student Notes OBJECTIVES Students will: investigate the relationship between reactants and products. investigate an exothermic reaction. investigate an endothermic
LCPS Core Experience Chemical Reactions Student Notes OBJECTIVES Students will: investigate the relationship between reactants and products. investigate an exothermic reaction. investigate an endothermic
Characteristics of Chemical Change
 Section 2 Characteristics of Chemical Change What Do You See? Learning Outcomes In this section you will Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Section 2 Characteristics of Chemical Change What Do You See? Learning Outcomes In this section you will Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Section 1 Forming New Substances
 Section 1 Forming New Substances Key Concept During chemical reactions, atoms rearrange to form new substances that have different properties than the original substances had. What You Will Learn Four
Section 1 Forming New Substances Key Concept During chemical reactions, atoms rearrange to form new substances that have different properties than the original substances had. What You Will Learn Four
Chapter 5, Lesson 5 Using Dissolving to Identify an Unknown
 Chapter 5, Lesson 5 Using Dissolving to Identify an Unknown Key Concepts Different substances are made from different atoms, ions, or molecules, which interact with water in different ways. Since dissolving
Chapter 5, Lesson 5 Using Dissolving to Identify an Unknown Key Concepts Different substances are made from different atoms, ions, or molecules, which interact with water in different ways. Since dissolving
Experiment 8 - Chemical Changes
 Experiment 8 - Chemical Changes When a chemical change occurs, the chemicals that you start with are changed into different chemicals. We know when this happens because the new chemicals have different
Experiment 8 - Chemical Changes When a chemical change occurs, the chemicals that you start with are changed into different chemicals. We know when this happens because the new chemicals have different
STEMscopedia: PHYSICAL AND CHEMICAL PROPERTIES AND CHANGES 8P1CD
 Reflect STEMscopedia: PHYSICAL AND CHEMICAL 8P1CD What do ice cream, root beer, and carbon dioxide gas have in common? Not only do these ingredients combine to make a good treat on a hot summer day, but
Reflect STEMscopedia: PHYSICAL AND CHEMICAL 8P1CD What do ice cream, root beer, and carbon dioxide gas have in common? Not only do these ingredients combine to make a good treat on a hot summer day, but
PREPARE FOR THE ACTIVITY. Activity Sheet Chapter 6, Lesson 8 ph and Color Change
 Activity Sheet Chapter 6, Lesson 8 ph and Color Change Name Date DEMONSTRATION 1. Your teacher poured green universal indicator into each of two cups. What does the change in color of the indicator solution
Activity Sheet Chapter 6, Lesson 8 ph and Color Change Name Date DEMONSTRATION 1. Your teacher poured green universal indicator into each of two cups. What does the change in color of the indicator solution
EXPERIMENT. for a Weak Acid. Determination of K a
 EXPERIMENT Determination of K a for a Weak Acid Hands-On Labs, Inc. Version 42-0151-00-02 Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before you begin.
EXPERIMENT Determination of K a for a Weak Acid Hands-On Labs, Inc. Version 42-0151-00-02 Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before you begin.
LEVEL ZERO VOICE CATALYST (10 minutes, individual work): 1. Counting atoms
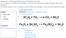 Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
Assignment 4 Chemical Detectives at the Molecular Level LO: To explain the differences between chemical and physical changes through observable evidence. EQ: What evidence can be used to tell the difference
Identifying Solids 1-2 KEY CONCEPTS AND PROCESS SKILLS KEY VOCABULARY ACTIVITY OVERVIEW L A B O R ATO R Y A-69
 Identifying Solids 40- to 1-2 50-minute sessions ACTIVITY OVERVIEW 7 L A B O R ATO R Y Students conduct tests on the solids separated from the mixture to gain information about the physical and chemical
Identifying Solids 40- to 1-2 50-minute sessions ACTIVITY OVERVIEW 7 L A B O R ATO R Y Students conduct tests on the solids separated from the mixture to gain information about the physical and chemical
Science Is A Verb! Elementary. Part 2 LET S DO IT! ISBN
 LET S DO IT! Science Is A Verb! Elementary Part 2 ISBN 978-1-847003-58-4 Contents INTRODUCTION Lab Title What Happens When Steel Rusts? Students know that during chemical reactions the atoms in the reactants
LET S DO IT! Science Is A Verb! Elementary Part 2 ISBN 978-1-847003-58-4 Contents INTRODUCTION Lab Title What Happens When Steel Rusts? Students know that during chemical reactions the atoms in the reactants
Physical and Chemical Changes Or How Do You Know When You ve Made Something New?
 Introduction Or How Do You Know When You ve Made Something New? Remember that all matter has characteristic physical and chemical properties. Matter can also undergo physical and chemical changes. How
Introduction Or How Do You Know When You ve Made Something New? Remember that all matter has characteristic physical and chemical properties. Matter can also undergo physical and chemical changes. How
Energy Changes in Chemical Reactions
 Energy Changes in Chemical Reactions Author(s): Ashley Colvin, Yunus Kinkhabwala, Prof. Song Lin, Jonathan Neff, & Greg Sauer Date Created: October 2016 Subject: Chemistry Grade Level: Middle School Standards:
Energy Changes in Chemical Reactions Author(s): Ashley Colvin, Yunus Kinkhabwala, Prof. Song Lin, Jonathan Neff, & Greg Sauer Date Created: October 2016 Subject: Chemistry Grade Level: Middle School Standards:
1 Forming New Substances
 CHAPTER 9 1 Forming New Substances SECTION Chemical Reactions BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a What are exothermic reactions? What are
CHAPTER 9 1 Forming New Substances SECTION Chemical Reactions BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a What are exothermic reactions? What are
Experiment 7 Can You Slow It Down?
 Experiment 7 Can You Slow It Down? OUTCOMES After completing this experiment, the student should be able to: tell which factors influence the reaction rate and how they influence the rate. change the temperature
Experiment 7 Can You Slow It Down? OUTCOMES After completing this experiment, the student should be able to: tell which factors influence the reaction rate and how they influence the rate. change the temperature
Chapter 5, Lesson 1: Water is a Polar Molecule
 Chapter 5, Lesson 1: Water is a Polar Molecule Key Concepts The water molecule, as a whole, has 10 protons and 10 electrons, so it is neutral. In a water molecule, the oxygen atom and hydrogen atoms share
Chapter 5, Lesson 1: Water is a Polar Molecule Key Concepts The water molecule, as a whole, has 10 protons and 10 electrons, so it is neutral. In a water molecule, the oxygen atom and hydrogen atoms share
Ocean Acidification in a Cup Materials
 Ocean Acidification in a Cup Ocean acidification is a problem that humans will have to deal with as we release more and more carbon dioxide into the atmosphere. This activity demonstrates how water can
Ocean Acidification in a Cup Ocean acidification is a problem that humans will have to deal with as we release more and more carbon dioxide into the atmosphere. This activity demonstrates how water can
Activity Title: It s Either Very Hot or Very Cold Up There!
 Grades 3-5 Teacher Pages Activity Title: It s Either Very Hot or Very Cold Up There! Activity Objective(s): In this activity, and the follow-up activity next week, teams will design and conduct experiments
Grades 3-5 Teacher Pages Activity Title: It s Either Very Hot or Very Cold Up There! Activity Objective(s): In this activity, and the follow-up activity next week, teams will design and conduct experiments
Activity 6.5 From gas to liquid to solid
 Activity 6.5 This activity is an extension of Activity 6.4a in which ice is used to make a container cold. As in Activity 6.4a, this activity will work only with sufficient water vapor in the air. Here,
Activity 6.5 This activity is an extension of Activity 6.4a in which ice is used to make a container cold. As in Activity 6.4a, this activity will work only with sufficient water vapor in the air. Here,
Energy Transformations Activities Students engage in different activities to observe energy changing form!
 Energy Transformations Activities Students engage in different activities to observe energy changing form! Name: Energy Labs and Activities Activity 1: Static Electricity Glue this side down Procedure:
Energy Transformations Activities Students engage in different activities to observe energy changing form! Name: Energy Labs and Activities Activity 1: Static Electricity Glue this side down Procedure:
Student Exploration: Chemical Changes
 Name: Date: Student Exploration: Chemical Changes Vocabulary: acid, base, catalyst, chemical change, coefficient, conservation of matter, decomposition, dissolve, double replacement, endothermic, exothermic,
Name: Date: Student Exploration: Chemical Changes Vocabulary: acid, base, catalyst, chemical change, coefficient, conservation of matter, decomposition, dissolve, double replacement, endothermic, exothermic,
The effects of sodium chloride on the Boiling Point of Dihydrogen Monoxide
 The effects of sodium chloride on the Boiling Point of Dihydrogen Monoxide Background: Dissolving a solute in a liquid can change its properties. These are called Colligative Properties, which are properties
The effects of sodium chloride on the Boiling Point of Dihydrogen Monoxide Background: Dissolving a solute in a liquid can change its properties. These are called Colligative Properties, which are properties
CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED)
 CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED) Overview: Students investigate chemical reactions using given substances. Students identify chemical reactions in their daily lives. Objectives: The student
CHEMICAL REACTION IN A BAGGY (MODIFIED FOR ADEED) Overview: Students investigate chemical reactions using given substances. Students identify chemical reactions in their daily lives. Objectives: The student
Chapter 1, Lesson 3: The Ups and Downs of Thermometers
 Chapter 1, Lesson 3: The Ups and Downs of Thermometers Key Concepts The way a thermometer works is an example of heating and cooling a liquid. When heated, the molecules of the liquid in the thermometer
Chapter 1, Lesson 3: The Ups and Downs of Thermometers Key Concepts The way a thermometer works is an example of heating and cooling a liquid. When heated, the molecules of the liquid in the thermometer
Predict the effect of increased competition for abiotic and biotic resources on a food web. colored pencils graph paper ruler
 Edit File QUICK LAB Effect of Abiotic and Biotic Factors No organism exists in isolation. Organisms depend on and compete for the abiotic, or non-living, factors in its environment. For example, organisms
Edit File QUICK LAB Effect of Abiotic and Biotic Factors No organism exists in isolation. Organisms depend on and compete for the abiotic, or non-living, factors in its environment. For example, organisms
Standards 8.5.c. I know chemical reactions usually liberate or absorbs heat.
 6 March 2013 Give an example of a physical change and a chemical change, and then describe how they are different from the other. Explain your answer in 2-3 sentences. Standards 8.5.c. I know chemical
6 March 2013 Give an example of a physical change and a chemical change, and then describe how they are different from the other. Explain your answer in 2-3 sentences. Standards 8.5.c. I know chemical
Chemical Matrix of Acids and Bases
 Chemical Matrix of Acids and Bases Overview:If you love the idea of mixing up chemicals and dream of having your own mad science lab, this one is for you. You are going to mix up solids and liquids in
Chemical Matrix of Acids and Bases Overview:If you love the idea of mixing up chemicals and dream of having your own mad science lab, this one is for you. You are going to mix up solids and liquids in
Lesson 1: What are chemical changes?
 Lesson 1 Summary Lesson 1: What are chemical changes? Vocabulary Use with pp. 375 377 physical change a change in which the material keeps its identify chemical change a change in which one substance or
Lesson 1 Summary Lesson 1: What are chemical changes? Vocabulary Use with pp. 375 377 physical change a change in which the material keeps its identify chemical change a change in which one substance or
3. Kira makes 93 greeting cards for a craft fair. She sells the cards in packs of 5. How many full packs of greeting cards does Kira make?
 Chapter 4 Test 1. 2. 3. Kira makes 93 greeting cards for a craft fair. She sells the cards in packs of 5. How many full packs of greeting cards does Kira make? packs Work Space: 5. A kennel is moving 160
Chapter 4 Test 1. 2. 3. Kira makes 93 greeting cards for a craft fair. She sells the cards in packs of 5. How many full packs of greeting cards does Kira make? packs Work Space: 5. A kennel is moving 160
The Next Generation Science Standards (NGSS)
 The Next Generation Science Standards (NGSS) CHAPTER 6, LESSON 1: WHAT IS A CHEMICAL REACTION? MS-PS1-2. Analyze and interpret data on the properties of substances before and after the substances interact
The Next Generation Science Standards (NGSS) CHAPTER 6, LESSON 1: WHAT IS A CHEMICAL REACTION? MS-PS1-2. Analyze and interpret data on the properties of substances before and after the substances interact
Titration with an Acid and a Base
 Skills Practice Titration with an Acid and a Base Titration is a process in which you determine the concentration of a solution by measuring what volume of that solution is needed to react completely with
Skills Practice Titration with an Acid and a Base Titration is a process in which you determine the concentration of a solution by measuring what volume of that solution is needed to react completely with
Experiment 17 It s A Gas and More!
 Energy Energy Experiment 17 It s A Gas and More! OUTCOMES After completing this lab activity, the student should be able to: explain a simple method for distinguishing carbon dioxide gas from oxygen gas.
Energy Energy Experiment 17 It s A Gas and More! OUTCOMES After completing this lab activity, the student should be able to: explain a simple method for distinguishing carbon dioxide gas from oxygen gas.
More Chemical Changes
 Activity 2 More Chemical Changes Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Activity 2 More Chemical Changes Activity 2 More Chemical Changes GOALS In this activity you will: Observe several typical examples of evidence that a chemical change is occurring. Make generalizations
Pre-Lab Read the entire laboratory assignment. Answer all pre-lab questions before beginning the lab.
 Name: Date: Pd: Lab Partner: Lab # 13: Types of Reactions, Predicting Products of Chemical Reactions Lab Accelerated Chemistry 1 Introduction: If you examine your bicycle after it has been left out in
Name: Date: Pd: Lab Partner: Lab # 13: Types of Reactions, Predicting Products of Chemical Reactions Lab Accelerated Chemistry 1 Introduction: If you examine your bicycle after it has been left out in
AP Chemistry: Designing an Effective Hand Warmer Student Guide INTRODUCTION
 AP Chemistry: Designing an Effective Hand Warmer Student Guide INTRODUCTION AP and the Advanced Placement Program are registered trademarks of the College Entrance Examination Board. The activity and materials
AP Chemistry: Designing an Effective Hand Warmer Student Guide INTRODUCTION AP and the Advanced Placement Program are registered trademarks of the College Entrance Examination Board. The activity and materials
Released Science Inquiry Task A Cool Investigation
 Date: Your Name: Released Science Inquiry Task A Cool Investigation 07 Grade Student Test Booklet Directions: Science You will be reading a story and analyzing the data provided to answer a set of questions.
Date: Your Name: Released Science Inquiry Task A Cool Investigation 07 Grade Student Test Booklet Directions: Science You will be reading a story and analyzing the data provided to answer a set of questions.
EXPERIMENT. Titration for Acetic Acid in Vinegar
 EXPERIMENT Titration for Acetic Acid in Vinegar Hands-On Labs, Inc. Version 42-0208-00-02 Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before you begin.
EXPERIMENT Titration for Acetic Acid in Vinegar Hands-On Labs, Inc. Version 42-0208-00-02 Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before you begin.
The Eight Solution Problem Exploring Reactions of Aqueous Ionic Compounds
 The Eight Solution Problem Exploring Reactions of Aqueous Ionic Compounds About this Lesson This activity allows students to mix a variety of known ionic solutions while making careful observations. After
The Eight Solution Problem Exploring Reactions of Aqueous Ionic Compounds About this Lesson This activity allows students to mix a variety of known ionic solutions while making careful observations. After
Chapter 4, Lesson 4: Energy Levels, Electrons, and Covalent Bonding
 Chapter 4, Lesson 4: Energy Levels, Electrons, and Covalent Bonding Key Concepts The electrons on the outermost energy level of the atom are called valence electrons. The valence electrons are involved
Chapter 4, Lesson 4: Energy Levels, Electrons, and Covalent Bonding Key Concepts The electrons on the outermost energy level of the atom are called valence electrons. The valence electrons are involved
Virtual Library Lesson: Oobleck, Gloop, and Glurch
 Oobleck, Gloop, and Glurch Lesson Overview Throughout this lesson, students will use inquiry skills to identify states of matter, describe physical properties, and modify the recipe to change physical
Oobleck, Gloop, and Glurch Lesson Overview Throughout this lesson, students will use inquiry skills to identify states of matter, describe physical properties, and modify the recipe to change physical
Matter Lesson 2. Learning Goal 3: I can describe the differences between physical and chemical changes of matter.
 Matter Lesson 2 Learning Goal 2: I can describe the differences between intensive physical properties, extensive physical properties, and chemical properties of matter. Learning Goal 3: I can describe
Matter Lesson 2 Learning Goal 2: I can describe the differences between intensive physical properties, extensive physical properties, and chemical properties of matter. Learning Goal 3: I can describe
Chemistry: classifying chemical and physical changes in various materials/substances
 Chemistry: classifying chemical and physical changes in various materials/substances Nikki Schilling, Ames, St. Paul Mn Based on original activity from Cool Chemistry Concoctions by Joe Rhatigan and Veronika
Chemistry: classifying chemical and physical changes in various materials/substances Nikki Schilling, Ames, St. Paul Mn Based on original activity from Cool Chemistry Concoctions by Joe Rhatigan and Veronika
o Test tube In this experiment, you ll be observing the signs of chemical reactions. These include the following:
 Experiment: Chemical Reactions & Chemical s Objective In this experiment, students perform a variety of chemical reactions. For each reaction, student identify the signs that a reaction has occurred, write
Experiment: Chemical Reactions & Chemical s Objective In this experiment, students perform a variety of chemical reactions. For each reaction, student identify the signs that a reaction has occurred, write
Apply the ideal gas law (PV = nrt) to experimentally determine the number of moles of carbon dioxide gas generated
 Teacher Information Ideal Gas Law Objectives Determine the number of moles of carbon dioxide gas generated during a reaction between hydrochloric acid and sodium bicarbonate. Through this investigation,
Teacher Information Ideal Gas Law Objectives Determine the number of moles of carbon dioxide gas generated during a reaction between hydrochloric acid and sodium bicarbonate. Through this investigation,
Reactivity of Halide Ions
 Microscale Reactivity of Halide Ions The four halide salts used in this experiment are found in your body. Although sodium fluoride is poisonous, trace amounts seem to be beneficial to humans in the prevention
Microscale Reactivity of Halide Ions The four halide salts used in this experiment are found in your body. Although sodium fluoride is poisonous, trace amounts seem to be beneficial to humans in the prevention
Shifts in Equilibrium: Le Châtelier s Principle
 6 Shifts in Equilibrium: Le Châtelier s Principle Introduction Whenever a chemical reaction occurs, the reverse reaction can also occur. As the original reactants, on the left side of the equation, react
6 Shifts in Equilibrium: Le Châtelier s Principle Introduction Whenever a chemical reaction occurs, the reverse reaction can also occur. As the original reactants, on the left side of the equation, react
Classifying Chemical Reactions: Lab Directions
 Classifying Chemical Reactions: Lab Directions Please Return Background: The power of chemical reactions to transform our lives is visible all around us in our homes, in our cars, even in our bodies. Chemists
Classifying Chemical Reactions: Lab Directions Please Return Background: The power of chemical reactions to transform our lives is visible all around us in our homes, in our cars, even in our bodies. Chemists
Chemical Equilibrium and Le Chatlier s Principle
 MiraCosta College Introductory Chemistry Laboratory Chemical Equilibrium and Le Chatlier s Principle EXPERIMENTAL TASK Examine a number of chemical reaction systems at equilibrium, predict the shifts they
MiraCosta College Introductory Chemistry Laboratory Chemical Equilibrium and Le Chatlier s Principle EXPERIMENTAL TASK Examine a number of chemical reaction systems at equilibrium, predict the shifts they
Name: Block : Date: (Textbook Chapter 9.4) Rate of reaction or reaction rate is how quickly or slowly reactants turn into products.
 Science 10: Chemistry! Factors Affecting Reaction Rates (Textbook Chapter 94) Rate of reaction or reaction rate is how quickly or slowly reactants turn into products reaction rate = A slow reaction has
Science 10: Chemistry! Factors Affecting Reaction Rates (Textbook Chapter 94) Rate of reaction or reaction rate is how quickly or slowly reactants turn into products reaction rate = A slow reaction has
The student s results are shown in the table below. Time / minutes Time / minutes
 Q1.(a) Anhydrous calcium chloride is not used as a commercial de-icer because it reacts with water. The reaction with water is exothermic and causes handling problems. A student weighed out 1.00 g of anhydrous
Q1.(a) Anhydrous calcium chloride is not used as a commercial de-icer because it reacts with water. The reaction with water is exothermic and causes handling problems. A student weighed out 1.00 g of anhydrous
Chapter 2, Lesson 1: Heat, Temperature, and Conduction
 Chapter 2, Lesson 1: Heat, Temperature, and Conduction Key Concepts Adding energy (heating) atoms and molecules increases their motion, resulting in an increase in temperature. Removing energy (cooling)
Chapter 2, Lesson 1: Heat, Temperature, and Conduction Key Concepts Adding energy (heating) atoms and molecules increases their motion, resulting in an increase in temperature. Removing energy (cooling)
Experiment#1 Beer s Law: Absorption Spectroscopy of Cobalt(II)
 : Absorption Spectroscopy of Cobalt(II) OBJECTIVES In successfully completing this lab you will: prepare a stock solution using a volumetric flask; use a UV/Visible spectrometer to measure an absorption
: Absorption Spectroscopy of Cobalt(II) OBJECTIVES In successfully completing this lab you will: prepare a stock solution using a volumetric flask; use a UV/Visible spectrometer to measure an absorption
Separating the Mixture
 Separating the Mixture 40- to 1 50-minute session ACTIVITY OVERVIEW I N V E S T 5 I O N I G AT Students perform their procedures written in Activity 3, A Plan to Separate the Mixture, to physically separate
Separating the Mixture 40- to 1 50-minute session ACTIVITY OVERVIEW I N V E S T 5 I O N I G AT Students perform their procedures written in Activity 3, A Plan to Separate the Mixture, to physically separate
Substances and Mixtures:Separating a Mixture into Its Components
 MiraCosta College Introductory Chemistry Laboratory Substances and Mixtures:Separating a Mixture into Its Components EXPERIMENTAL TASK To separate a mixture of calcium carbonate, iron and sodium chloride
MiraCosta College Introductory Chemistry Laboratory Substances and Mixtures:Separating a Mixture into Its Components EXPERIMENTAL TASK To separate a mixture of calcium carbonate, iron and sodium chloride
Experiment 6 Shifts in Equilibrium: Le Châtelier s Principle
 Experiment 6 Shifts in Equilibrium: Le Châtelier s Principle Introduction Whenever a chemical reaction occurs, the reverse reaction can also occur. As the original reactants, on the left side of the equation,
Experiment 6 Shifts in Equilibrium: Le Châtelier s Principle Introduction Whenever a chemical reaction occurs, the reverse reaction can also occur. As the original reactants, on the left side of the equation,
CALORIMETRY: Heat of Fusion of Ice
 Pre-Lab Discussion CALORIMETRY: Heat of Fusion of Ice When a chemical or physical change takes place, heat is either given off or absorbed That is, the change is either exothermic or endothermic It is
Pre-Lab Discussion CALORIMETRY: Heat of Fusion of Ice When a chemical or physical change takes place, heat is either given off or absorbed That is, the change is either exothermic or endothermic It is
Bellevue College CHEM& 121 Experiment: Stoichiometric Analysis of an Antacid 1
 Experiment: Stoichiometric Analysis of an Antacid 1 Introduction In this lab, you will use the concept of stoichiometry to solve two sequential problems. First, you will try to determine the products of
Experiment: Stoichiometric Analysis of an Antacid 1 Introduction In this lab, you will use the concept of stoichiometry to solve two sequential problems. First, you will try to determine the products of
CHM101 Lab Measurements and Conversions Grading Rubric
 CHM101 Lab Measurements and Conversions Grading Rubric Name Team Name Criteria Points possible Points earned Lab Performance Printed lab handout and rubric was brought to lab 3 Safety and proper waste
CHM101 Lab Measurements and Conversions Grading Rubric Name Team Name Criteria Points possible Points earned Lab Performance Printed lab handout and rubric was brought to lab 3 Safety and proper waste
LAB 1B: THE SCIENTIFIC METHOD: MAKING OBSERVATIONS & DESIGNING EXPERIMENTS
 LAB 1B: THE SCIENTIFIC METHOD: MAKING OBSERVATIONS & DESIGNING EPERIMENTS PURPOSE: To practice the scientific method of observation, hypothesis, experimentation, & conclusion. SAFETY CONCERNS: Always wear
LAB 1B: THE SCIENTIFIC METHOD: MAKING OBSERVATIONS & DESIGNING EPERIMENTS PURPOSE: To practice the scientific method of observation, hypothesis, experimentation, & conclusion. SAFETY CONCERNS: Always wear
Experiment 14 - Heats of Reactions
 Experiment 14 - Heats of Reactions If a chemical reaction is carried out inside a calorimeter, the heat evolved or absorbed by the reaction can be determined. A calorimeter is an insulated container, and
Experiment 14 - Heats of Reactions If a chemical reaction is carried out inside a calorimeter, the heat evolved or absorbed by the reaction can be determined. A calorimeter is an insulated container, and
Experiment 6 Alcohols and Phenols
 Experiment 6 Alcohols and Phenols Alcohols are organic molecules that contain a hydroxyl (-) group. Phenols are molecules that contain an group that is directly attached to a benzene ring. Alcohols can
Experiment 6 Alcohols and Phenols Alcohols are organic molecules that contain a hydroxyl (-) group. Phenols are molecules that contain an group that is directly attached to a benzene ring. Alcohols can
MONDAY (12/12) TUESDAY (12/13) WEDNESDAY (12/14) THURSDAY (12/15) FRIDAY (12/16) Making Acid Rain (a lab) Quiz
 Homework Activities Name: Date: Period: This week, we will be using our knowledge of acids and bases and studying how acids, specifically acid rain, affect our lives and our environment. We will also end
Homework Activities Name: Date: Period: This week, we will be using our knowledge of acids and bases and studying how acids, specifically acid rain, affect our lives and our environment. We will also end
Page 1 / 12. Chemistry Exam. Name: Matter Properties, Structure. Question 1 (1 point) The atomic number of an atom is. A. The mass of the atom.
 Chemistry Exam Matter Properties, Structure Name: Question 1 (1 point) The atomic number of an atom is A. The mass of the atom. B. The number of protons added to the number of neutrons in the nucleus.
Chemistry Exam Matter Properties, Structure Name: Question 1 (1 point) The atomic number of an atom is A. The mass of the atom. B. The number of protons added to the number of neutrons in the nucleus.
Chemical Bonds. MATERIALS 24-well microplate calcium chloride candle citric acid conductivity tester ethanol gloves iron ring lab apron
 Microscale Chemical Bonds Chemical compounds are combinations of atoms held together by chemical bonds. These chemical bonds are of two basic types ionic and covalent. Ionic bonds result when one or more
Microscale Chemical Bonds Chemical compounds are combinations of atoms held together by chemical bonds. These chemical bonds are of two basic types ionic and covalent. Ionic bonds result when one or more
Alka Seltzer Lab: Reaction rate - Factors that affect the rate of chemical reactions
 Alka Seltzer Lab: Reaction rate - Factors that affect the rate of chemical reactions Materials Lead Scribe Reporter/Questioner Clean-up WRITE ALL NOTES / DATA / OBSERVATIONS ON YOUR DATA SHEET The rate
Alka Seltzer Lab: Reaction rate - Factors that affect the rate of chemical reactions Materials Lead Scribe Reporter/Questioner Clean-up WRITE ALL NOTES / DATA / OBSERVATIONS ON YOUR DATA SHEET The rate
Name Date Period Molecular Nature of Water
 Name Date Period Molecular Nature of Water Purpose: To determine how water molecules react using molecular models and Lab demos. Materials: I cup of 12 water molecules (red & white), 1 Na (blue), 1 Cl
Name Date Period Molecular Nature of Water Purpose: To determine how water molecules react using molecular models and Lab demos. Materials: I cup of 12 water molecules (red & white), 1 Na (blue), 1 Cl
Chapter 2, Lesson 5: Changing State Melting
 Chapter 2, Lesson 5: Changing State Melting Key Concepts Melting is a process that causes a substance to change from a solid to a liquid. Melting occurs when the molecules of a solid speed up enough that
Chapter 2, Lesson 5: Changing State Melting Key Concepts Melting is a process that causes a substance to change from a solid to a liquid. Melting occurs when the molecules of a solid speed up enough that
This article describes a two-part
 34 Students Learn Exothermic and Endothermic Processes BY EVELYN GRAY This article describes a two-part investigation exploring endothermic and exothermic processes. During the first part of the investigation,
34 Students Learn Exothermic and Endothermic Processes BY EVELYN GRAY This article describes a two-part investigation exploring endothermic and exothermic processes. During the first part of the investigation,
EXPERIMENT. Stoichiometry of a Precipitation Reaction
 EXPERIMENT Stoichiometry of a Precipitation Reaction Hands-On Labs, Inc. Version 42-0201-00-02 Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before
EXPERIMENT Stoichiometry of a Precipitation Reaction Hands-On Labs, Inc. Version 42-0201-00-02 Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before
Exploring Acids & Bases
 Food Explorations Lab: Exploring Acids & Bases STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will use cabbage juice indicator to determine if two unknown samples are acids or
Food Explorations Lab: Exploring Acids & Bases STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will use cabbage juice indicator to determine if two unknown samples are acids or
Percentage of Acetic Acid in Vinegar
 Microscale Percentage of Acetic Acid in Vinegar When sweet apple cider is fermented in the absence of oxygen, the product is an acid, vinegar. Most commercial vinegars are made by fermentation, but some,
Microscale Percentage of Acetic Acid in Vinegar When sweet apple cider is fermented in the absence of oxygen, the product is an acid, vinegar. Most commercial vinegars are made by fermentation, but some,
Lesson Plan: Stearic Acid
 Lesson Plan: Stearic Acid Created by: In this lesson, students investigate how stearic acid undergoes a 2014 AACT Middle School phase change from solid to liquid and back from liquid to solid. Content
Lesson Plan: Stearic Acid Created by: In this lesson, students investigate how stearic acid undergoes a 2014 AACT Middle School phase change from solid to liquid and back from liquid to solid. Content
Working in the Chemistry Laboratory
 Working in the Chemistry Laboratory Accelerated Chemistry I Introduction: One of the most important components of your chemistry course is the laboratory experience. Perhaps you have done experiments in
Working in the Chemistry Laboratory Accelerated Chemistry I Introduction: One of the most important components of your chemistry course is the laboratory experience. Perhaps you have done experiments in
HEATS OF REACTION EXPERIMENT
 16 Text Reference Section 11.4 HEATS OF REACTION EXPERIMENT PURPOSE To measure the heats of reaction for three related exothermic reactions and to verify Hess s law of heat summation. Time Required 50
16 Text Reference Section 11.4 HEATS OF REACTION EXPERIMENT PURPOSE To measure the heats of reaction for three related exothermic reactions and to verify Hess s law of heat summation. Time Required 50
GCSE. Chemistry Practical Manual CCEA GCSE TEACHER GUIDANCE. Unit 3: Practical Skills
 GCSE CCEA GCSE TEACHER GUIDANCE Chemistry Practical Manual Unit 3: Practical Skills C2: Investigate the reactions of acids, including temperature changes that occur For first teaching from September 2017
GCSE CCEA GCSE TEACHER GUIDANCE Chemistry Practical Manual Unit 3: Practical Skills C2: Investigate the reactions of acids, including temperature changes that occur For first teaching from September 2017
A Chemical Clock. 5. Consider each of the following questions regarding data and measurements:
 A Chemical Clock Things to Consider 1. What are the three major objectives of this experiment? What methods will you try using to achieve each of these three objectives? 2. What is difference between reaction
A Chemical Clock Things to Consider 1. What are the three major objectives of this experiment? What methods will you try using to achieve each of these three objectives? 2. What is difference between reaction
